- 1Institute of Health Food, Zhejiang Academy of Medical Sciences, Hangzhou, China
- 2Institute of Preventive Veterinary Medicine and Zhejiang Provincial Key Laboratory of Preventive Veterinary Medicine, Zhejiang University, Hangzhou, China
Type VI secretion system (T6SS) is a macromolecular transenvelope machine encoded within the genomes of several proteobacteria species. Vibrio parahaemolyticus contains two putative T6SS systems, VpT6SS1 and VpT6SS2, both contributing to adherence to Caco-2 and/or HeLa cells. However, it remains unknown if these systems are involved in cellular responses. In order to exclude the effects of other virulence factors known to induce cytotoxicity or autophagy, a triple deletion mutant dTTT (with deletion of tdh, and T3SS1 and T3SS2 structural protein genes) was used as the parent strain to construct deletion mutants of T6SS genes. The mutant dTTT-ΔicmF2, but not dTTT-ΔicmF1, reduced autophagic response upon 4 h of infection of the macrophage. Further attempt was made to search for the possible effector proteins that might be responsible for direct induction of autophagy by deletion of the genes encoding Hcp2 and VgrG2, two putative translocons of T6SS2 of V. parahaemolyticus. Deletion of either hcp2 or vgrG2 did reduce the autophagic response. However, increased LC3-II lipidation was seen only in the macrophage cells transfected with pVgrG2, but not with pHcp2. Chloroquinine treatment increased accumulation of LC3-II, suggesting that VgrG2 enhanced autophagic flux. The fact that vgrG2 deletion led to reduced level of intracellular cAMP suggests a possible role of cAMP signaling in autophagic responses to the bacterium. We conclude that VgrG2 of V. parahaemolyticus induces autophagy in macrophages.
Introduction
Vibrio parahaemolyticus is one of the leading causes of human foodborne gastroenteritis in China (about one-third of the foodborne outbreaks reported between 1991 and 2001) due to consumption of raw or under-cooked seafood (Liu et al., 2004). Early studies have linked gastroenteritis to the presence of thermostable direct hemolysin (TDH), TDH-related hemolysin (TRH) and two sets of type III secretion systems (T3SS1 and T3SS2), which are able to induce general cytotoxicity or enterotoxicity to host cells (Kaper et al., 1984; Park et al., 2004; Yeung and Boor, 2004; Okada et al., 2009).
Type VI secretion system is a macromolecular transenvelope machine encoded within the genomes of several proteobacterial species (Mougous et al., 2006; Pukatzki et al., 2007; Bingle et al., 2008; Bernard et al., 2010). The system contains 13–20 proteins [Intracellular multiplication Factor (IcmF)-associated homologous proteins, IAHP] coded by the gene cluster (Boyer et al., 2009). Deletion of icmF associated proteins did not affect expression of the translocon proteins but prevents their translocation (Pukatzki et al., 2006; Suarez et al., 2008). The T6SSs of Vibrio cholerae, Pseudomonas aeruginosa, Aeromonas hydrophila, and Vibrio anguillarum were found to participate in pathogenicity: adhesion to epithelial cells, cytotoxicity, resistance to phagocytosis, tolerance to stress sensing, and replication inside the host cells (Mougous et al., 2006; Pukatzki et al., 2006; Zheng and Leung, 2007; Suarez et al., 2008; Weber et al., 2009; Jani and Cotter, 2010). Of the two sets of putative T6SS in V. parahaemolyticus (VpT6SS), we found that VpT6SS1 is present in majority of clinical isolates (90.9%), but less in environmental or food isolates (25.0%) while VpT6SS2 exists in all isolates, and both systems contribute different aspects of adherence to Caco-2 and/or HeLa cells (Yu et al., 2012).
Autophagy acts as an intracellular surveillance system to monitor and trap invading pathogens and influence both the innate and adaptive immune responses (Burdette et al., 2009b; Deretic and Levine, 2009). For most intracellular bacteria, host cells use autophagy to prevent cytoplasmic replication or invasion of intracellular pathogens by engulfing the pathogens in autophagic vesicles and targeting them to lysosomes (Levine and Deretic, 2007). In extracellular bacteria like Vibrio spp, secreted proteins are involved in autophagy (Gutierrez et al., 2007). With V. parahaemolyticus, VP1680 (VopQ) and VP1659 (two secretion proteins of T3SS1) are found to induce autophagy accompanied with cytotoxicity, disruption of actin structure, and cell death (Burdette et al., 2009a; Zhou et al., 2010). By examining LC3 lipidation and EGFP-LC3 punctation in macrophages infected with V. parahaemolyticus, we provide the first evidence that the VgrG2, a translocon of VpT6SS2, induces autophagy.
Materials and Methods
Bacterial Strains and Plasmids
The bacterial strains and plasmids used in this study are listed in Table 1. V. parahaemolyticus strain HZ is a clinical isolate from the Zhejiang Provincial Center for Disease Control and Prevention, Zhejiang, China. Escherichia coli strains DH5α, BL21, and CC118λpir were used for general manipulation of plasmids, prokaryotic expression of proteins, and mobilization of plasmids into V. parahaemolyticus, respectively. The bacterial strains were grown at 37°C in Luria-Bertani (LB) broth (E. coli) or LB broth supplemented with 3% NaCl (V. parahaemolyticus). LB agar supplemented with 3% NaCl, 10 μg/ml chloramphenicol, and 25 μg/ml polymyxin was used for screening mutant strains. The culture media were supplemented, where appropriate, with the following antibiotics: chloramphenicol (Cm, 10 μg/ml), ampicillin (Amp, 100 μg/ml), and kanamycin (Kan, 50 μg/ml).
Plasmid Construction
The mammalian expression vector pcDNA-egfp and pcDNA-egfp-lc3b were constructed from pcDNA3.1 (Invitrogen) in our laboratory (Zhu et al., 2012). To construct pHcp2 and pVgrG2 in pcDNA3.1 background for expression of these proteins fused with GFP (Table 1), egfp was PCR-amplified from pcDNA-egfp by primers GFP-F/R, and genes hcp2 and vgrG2 were from V. parahemolyticus strain HZ amplified by primers hcp2-F/R and vgrG2-F/R, respectively. The egfp-hcp2 and egfp-vgrG2 fusion fragments were obtained by overlap PCR using primers GFP-F/hcp2-R and GFP-F/vgrG2-R, respectively, and cloned into the multiple cloning site of pcDNA3.1. The above primers are listed in Table 2. All constructs were confirmed by DNA sequencing.
Homologous Recombination
In-frame gene deletion of vgrG2 was generated by sacB-based allelic exchange as described previously (Park et al., 2004; Yu et al., 2012). Briefly, PCR amplification was performed to generate the upstream and downstream fragments of the vgrG2 gene (using respective primer pair vgrG2-A/B and vgrG2-C/D, Table 2). Overlap PCR was performed to construct a fragment with deletion of the vgrG2 gene using the primer pair vgrG2-A/D. The fragment was cloned into pMD18T vector (Takara) and then subcloned into the suicide vector pYAK1 that contains the sacB gene conferring sensitivity to sucrose. The recombinant plasmid was introduced into E. coli CC118λpir and then mated with V. parahaemolyticus dTTT (strain HZ with in-frame deletion of tdh, vcrD1, and vcrD2) as the parent strain. The resulting mutant strains were screened using selective LB agar as specified above.
Preparation of Protein Samples From Bacterial Supernatants and Pellets
Secreted proteins from the parent and mutant V. parahaemolyticus strains were prepared from the supernatant samples of cultures grown for 16 h at 28°C in LB broth. The samples were passed through a 0.2 μm pore-size syringe filter and precipitated by adding trichloroacetic acid to a final concentration of 10% (vol/vol). The proteins were collected by centrifugation at 15,000 g for 30 min at 4°C. The precipitates were solubilized in 40 μl 0.1M NaOH, and 10 μl of 5x SDS-PAGE loading buffer was added prior to SDS-PAGE with 10% polyacrylamide. For separation of T6SS proteins associated with the bacterial cells, V. parahaemolyticus cultures were pelleted by centrifugation, and the pellets were resuspended in 10 mM phosphate buffered saline pH 7.2 (PBS, 100 mg wet weight pellet per ml). A volume of 160 μl was then mixed with 10 μl of 5X SDS-PAGE loading buffer, and the mixtures were heat-treated for 5 min in a boiling water-bath to release proteins from the bacterial cells before SDS-PAGE.
Cells Culture, Bacterial Infection, and Vector Transfection
Murine RAW264.7 macrophage cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM, Gibco) supplemented with 10% new-born calf serum, L-glutamine (1%), penicillin G (100 U/ml), and streptomycin (100 μg/ml).
The macrophage cells were infected with mid-log phase cultures (3–4 h) of V. parahaemolyticus wild-type strain (WT) HZ, and single or multiple deletion mutants at multiplicity of infection (MOI) of 10 at 37°C and 5% CO2. Infection was allowed to proceed for 0.5 h and the supernatant was then removed. The cell monolayers were rinsed with sterile PBS to remove unattached bacteria and then incubated in the presence of fresh medium for 2 or 4 h at 37°C and 5% CO2 for cytotoxicity assay or SDS-PAGE and immunoblotting as described below. Rapamycin treated (0.5 μM, Merck) or mock-infected cells were included as controls (here and in other relevant experiments).
The cells stably expressing GFP-LC3B on the coverslips were infected with the strain dTTT or dTTT-ΔicmF2 at MOI of 10. The remaining procedure is the same as above. At the end of 4 h incubation, the cell monolayers were subjected for confocal microscopic imaging.
The macrophage cells were transfected with pcDNA-egfp, pHcp2, or pVgrG2 using lipofectamine 2000 (Invitrogen) for transient expression of the target proteins. The transfection medium was replaced 6 h post-transfection by complete medium containing G418 (0.5 mg/ml), and the cells were incubated for 24 h at 37°C and 5% CO2 for analysis of LC3 lipidation by immunoblotting.
For examination of the effects of 3-methyladenine (3-MA) and chloroquinine (CQ) on autophagic response, the RAW264.7 cells were first treated with these agents (10 mM 3-MA, and 5 mM CQ) for 1 h before they were transfected with vectors pHcp2, pVgrG2, and pcDNA-gfp as described above. The cell pellets were then collected for SDS-PAGE/Western blotting for LC3-II and β-actin.
Analysis of cAMP in Macrophages Infected with Vibrio Parahaemolyticus Strains
For measurement of cAMP within macrophages infected with the bacterial strains, the RAW264.7 cell monolayers were infected with the parent and mutant strains (triple deletion mutant dTTT with additional deletion of hcp2 or vgrG2, namely dTTT-Δhcp2 or dTTT-ΔvgrG2; about 10 MOI). After 4 h of incubation at 37°C and 5% CO2, the cell monolayers were washed twice with PBS and lysed with deionized H2O for 10 min followed by repeated pipetting and vortex-mixing in Eppendorf tubes. The cell lysates were subjected to centrifugation as above and the supernatant samples were collected for analysis of intracellular cAMP using the ELISA-based cAMP assay kit (detection range from 0.5 to 6 nM, lot No HL30028, Shanghai Haling Biol. Technol. Co., Ltd., China) according to the manufacturer’s instruction.
SDS-PAGE and Immunoblotting
After infection or transfection for indicated times, all cell samples were lysed for 10 min in ice cold lysis buffer [50 mM Tris-HCl pH 8.0, 140 mM NaCl, 1.5 mM MgCl2, 0.5% NP-40 with complete protease inhibitor cocktail (Roche)]. Cell debris was pelleted by centrifugation and clear supernatants transferred to new tubes. Protein concentration was measured by BCA protein assay kit (MultiSciences, Hangzhou, China). Protein samples were boiled for 5 min in the presence of 5x SDS-PAGE loading buffer.
Proteins on the gels were electro-transferred onto an Immobilon-P membrane (Millipore). The membranes were blocked with 5% skim milk in tris-buffered saline (20 mM Tris, 137 mM NaCl, pH 7.6) containing 0.05% Tween 20 and probed for 1 h with rabbit anti-Hcp2 or rabbit anti-VgrG2 polyclonal antibodies (Yu et al., 2012), rabbit anti-LC3 polyclonal IgG (Sigma–Aldrich) or anti-β-actin monoclonal IgG (MultiSciences, Hangzhou, China) for 3 h at room temperature. The blots were then probed with goat anti-rabbit or anti-mouse horse-radish peroxidase-labeled antibodies (KPL), and developed by the SuperSignala West Pico Chemiluminescent Substrate (Pierce) according to the manufacturer’s instruction.
Confocal Microscopy
Cells infected as above were washed with PBS, fixed and permeabilized with 80% cold acetone in PBS at -20°C for 20 min, and washed again with PBS. Cells were then counterstained with 4′,6-diamidino-2-phenylindole (DAPI) nucleic acid stain (Invitrogen). Fluorescence was observed under a laser scanning confocal microscope (Leica TCS SP5, Munich, Germany). The average number of EGFP-LC3 punctae per cell from at least 60 cells per sample was counted (Mizushima et al., 2010).
Cytotoxicity Assay
The culture supernatants from infected macrophage cells as above were collected for lactate dehydrogenase activity (LDH) using CytoTox 96 non-radioactive cytotoxicity assay (Promega).
Statistical Analysis
All data, where appropriate, are mean ± SD from three independent experiments and analyzed by Student’s t-test.
Results
T6SS2 Induces Autophagy in Macrophage Cells
Our preliminary data indicate that there was no apparent autophagic response of the macrophage cells in 2-h infection with V. parahaemolyticus, either the WT strain or its triple deletion mutant strain dTTT void of tdh, vcrD1, and vcrD2 with or without additional deletion of icmF1 and icmF2 (structural genes of VpT6SS1 and VpT6SS2, respectively; data now shown). When infection continued up to 4 h, T3SS1 was found to be involved in autophagy shown as reduced LC3-II transformation in the macrophage cells infected with ΔvcrD1 deletion mutant (Figure 1A), as previously reported (Burdette et al., 2009a; Zhou et al., 2010). In cells infected with the WT strain or its mutants Δtdh and ΔvcrD1, significant cytotoxic effect at 4 h was seen possibly due to T3SS1. To exclude the confounding effects of cytotoxicity, the triple deletion mutant (dTTT, in-frame deletion of tdh, vcrD1, and vcrD2) was used as the parent strain to examine the possible role of VpT6SS in autophagy after further deletion of both icmF1 and icmF2.
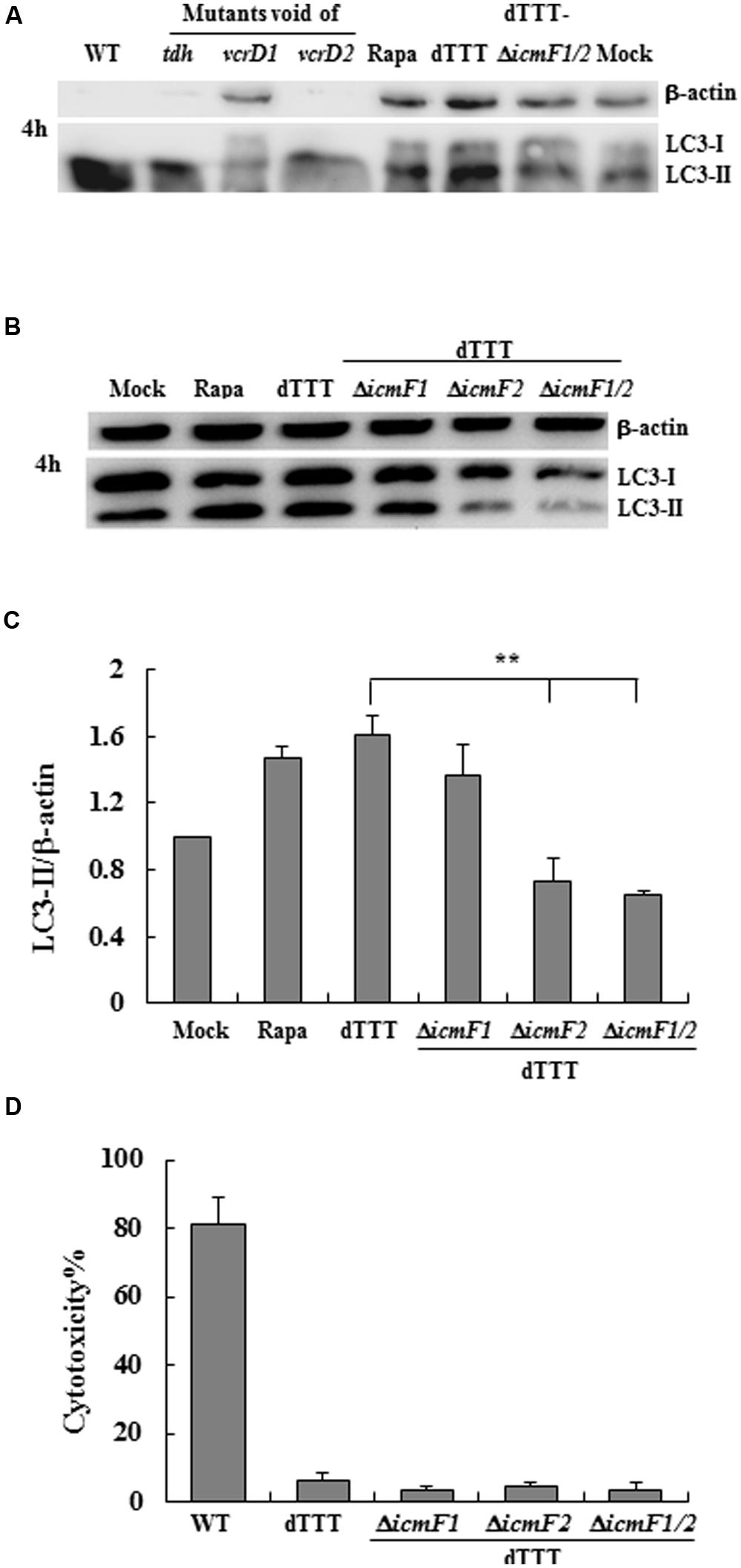
FIGURE 1. Vibrio parahaemolyticus T6SS2 induces autophagy of macrophage cells without causing apparent cytotoxicity. (A,B) Autophagy of macrophage cells was measured by LC3-II accumulation after infection for 4 h with the wild-type strain (WT), its triple deletion mutant dTTT or dTTT with further deletion of T6SS1 and T6SS2 structural genes. Rapamycin-treated (Rapa) and mock-infected (Mock) cells were used as positive and negative controls. (C) LC3-II to β-actin ratio (mean ± SD) of three independent experiments as shown in (B; **P < 0.01). (D) Cytotoxicity to macrophages infected as above was measured by LDH release.
Initial experiments with the multiple deletion strain did show reduced LC3 lipidation upon further deletion of both icmF1 and icmF2 [dTTT-ΔicmF1/2, as compared with its parent strain dTTT or rapamycin positive control (Figure 1A)]. To explore which VpT6SS system triggered autophagy of macrophages, further deletion of icmF1 or icmF2 was attempted from the triple mutant strain dTTT. Figures 1B,C shows that LC3 lipidation was significantly reduced in dTTT-ΔicmF1/2 or dTTT-ΔicmF2, but not dTTT-ΔicmF1 (P < 0.01, as compared with its parent strain dTTT; Figure 1C), indicating that VpT6SS2 might be involved in the autophagic response. TDH and/or T3SS, but not VpT6SS contribute to cytotoxicity as revealed by lactate dehydrogenase release (Figure 1D). To further confirm whether VpT6SS2 was indeed related to autophagy and induce the formation of autophagosomes, the EGFP-LC3-expressing macrophage cells were infected with V. parahaemolyticus dTTT and its mutant dTTT-ΔicmF2. Figure 2 shows that the mutant dTTT-ΔicmF2 exhibited significantly reduced number of cells containing punctae of EGFP-LC3 (i.e., autophagosome-like vesicles), as compared with its parent strain dTTT (P < 0.01).
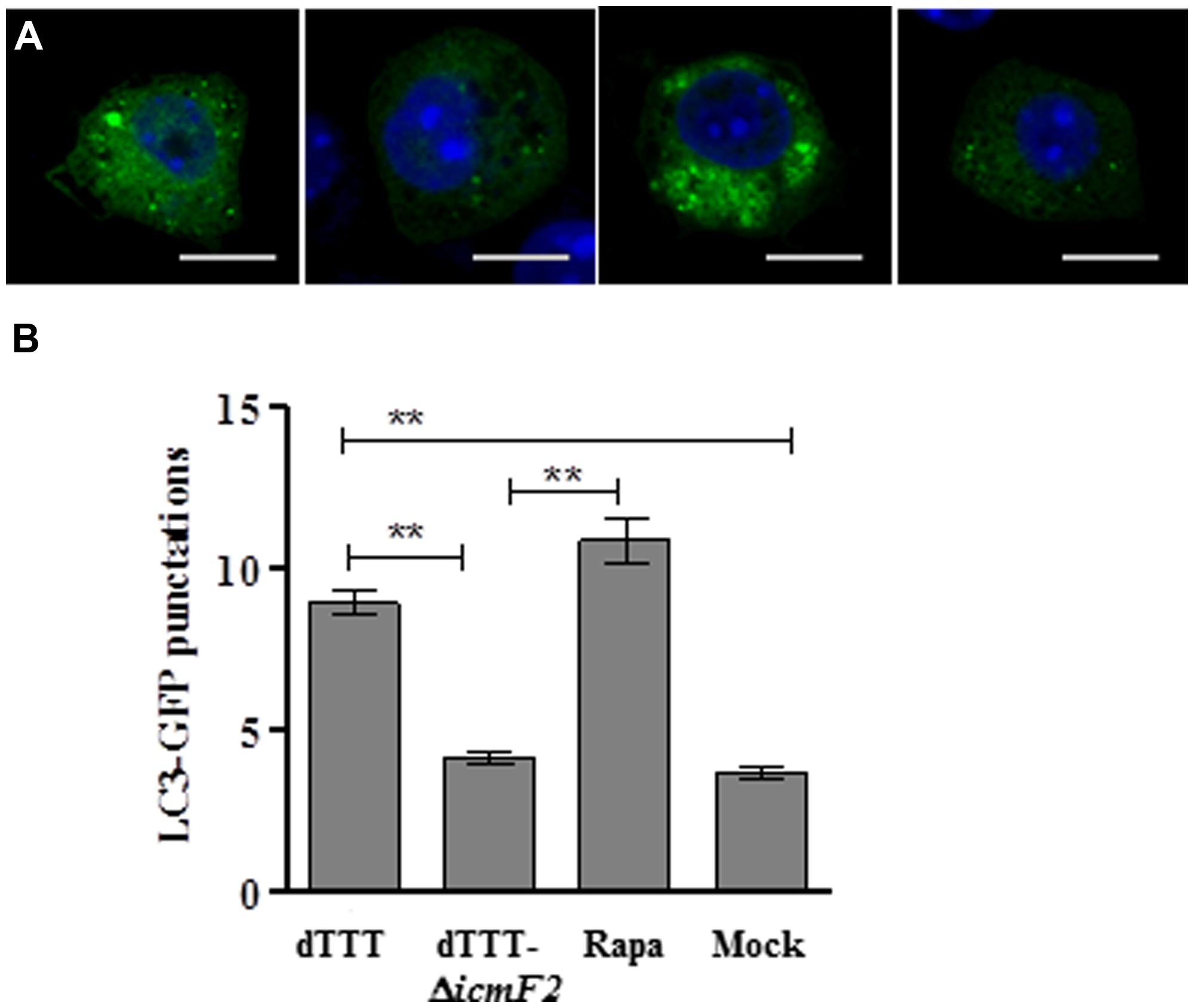
FIGURE 2. Vibrio parahaemolyticus T6SS2 induces autophagosome formation in macrophages. (A) Formation of autophagosome vesicles shown as green punctae in EGFP-LC3 expressing macrophage cells infected with strain dTTT and its icmF2 deletion mutant. Rapamycin-treated (Rapa) and mock-infected (Mock) cells were used as positive and negative controls (scale bar: 10 μm). (B) Mean ± SD of EGFP-LC3-II punctae per cell from 60 cells of three independent experiments as represented in (A; **P < 0.01).
VgrG2 of VpT6SS2 is the Effector Protein Devoting to Autophagy
A number of Gram-negative bacteria use T6SS to infect eukaryotic cells by its effectors, translocon, or secretion proteins (Cascales, 2008; Pukatzki et al., 2009). We paid attention to Hcp2 and VgrG2, two known translocon family proteins of VpT6SS2 (Yu et al., 2012). Deletion of either hcp2 or vgrG2 decreased the ratio of LC3-II to β-actin in cells infected with the mutant strains in comparison with its parent strain dTTT (Figure 3, P < 0.05), indicating that Hcp2, VgrG2, or even other secretion proteins translocated by them might contribute to autophagy in the macrophage cells.
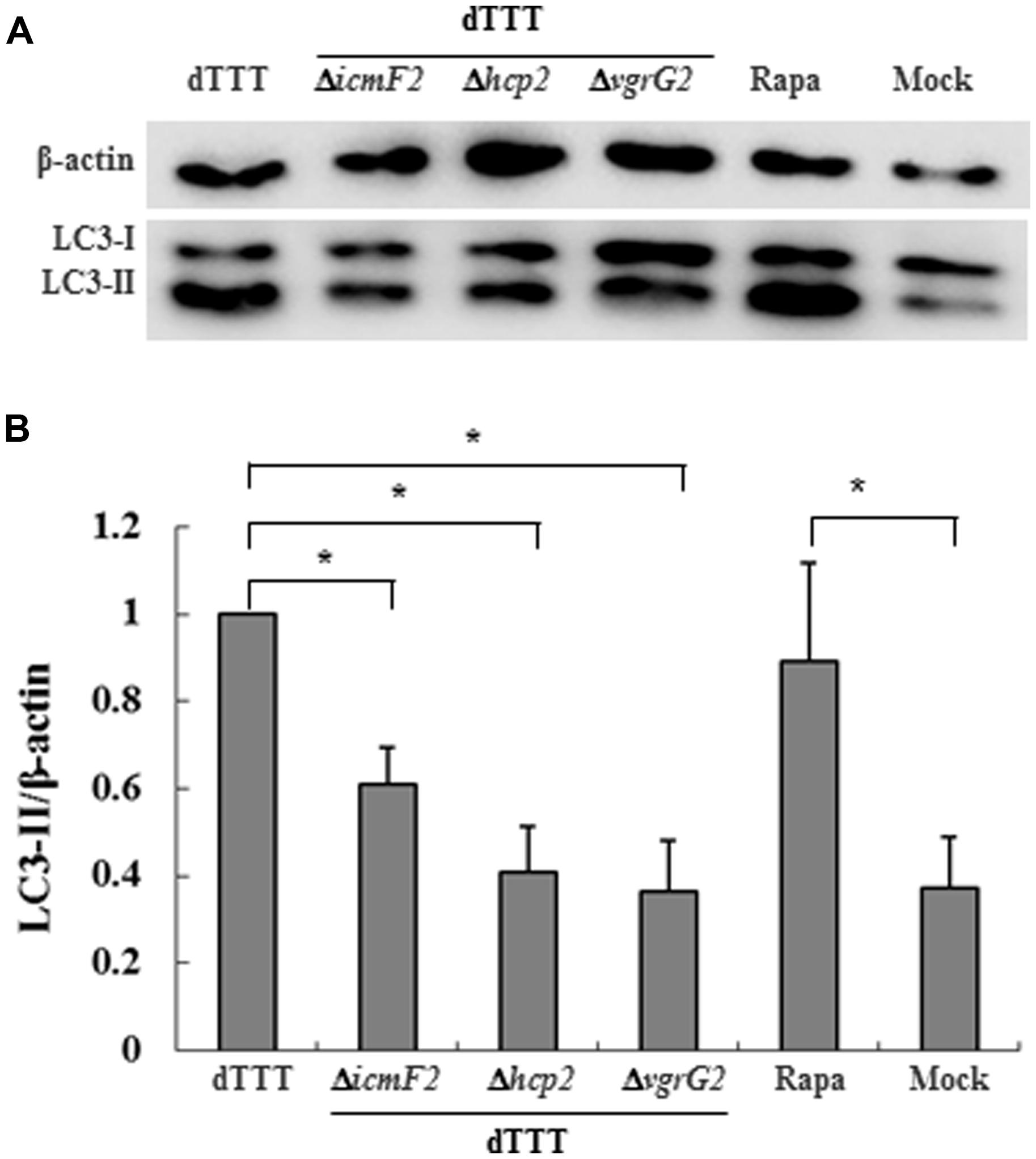
FIGURE 3. Vibrio parahaemolyticus T6SS2 translocons might be involved in autophagic response of macrophages. (A) LC3-II accumulation in cells infected for 4 h with strain dTTT and its mutants. Rapamycin-treated (Rapa) and mock-infected (Mock) cells were used as positive and negative controls. (B) Ratios of LC3-II to β-actin were calculated from ‘A’ and reported as mean ± SD of three independent experiments with the dTTT ratio normalized to 1.0 (*P < 0.05).
To further examine if Hcp2 or VgrG2 of VpT6SS devotes to autophagy, plasmids pHcp2, and pVgrG2 were made based on pcDNA-egfp (Zhu et al., 2012) to express EGFP-Hcp2 and EGFP-VgrG2 in macrophage cells after transfection for 24 h. We found that VgrG2, but not Hcp2, increased the conversion from LC3-I to LC3-II with significant elevation of LC3-II/β-actin ratio (P < 0.05) in comparison with the mock pcDNA-egfp (Figures 4A,B).
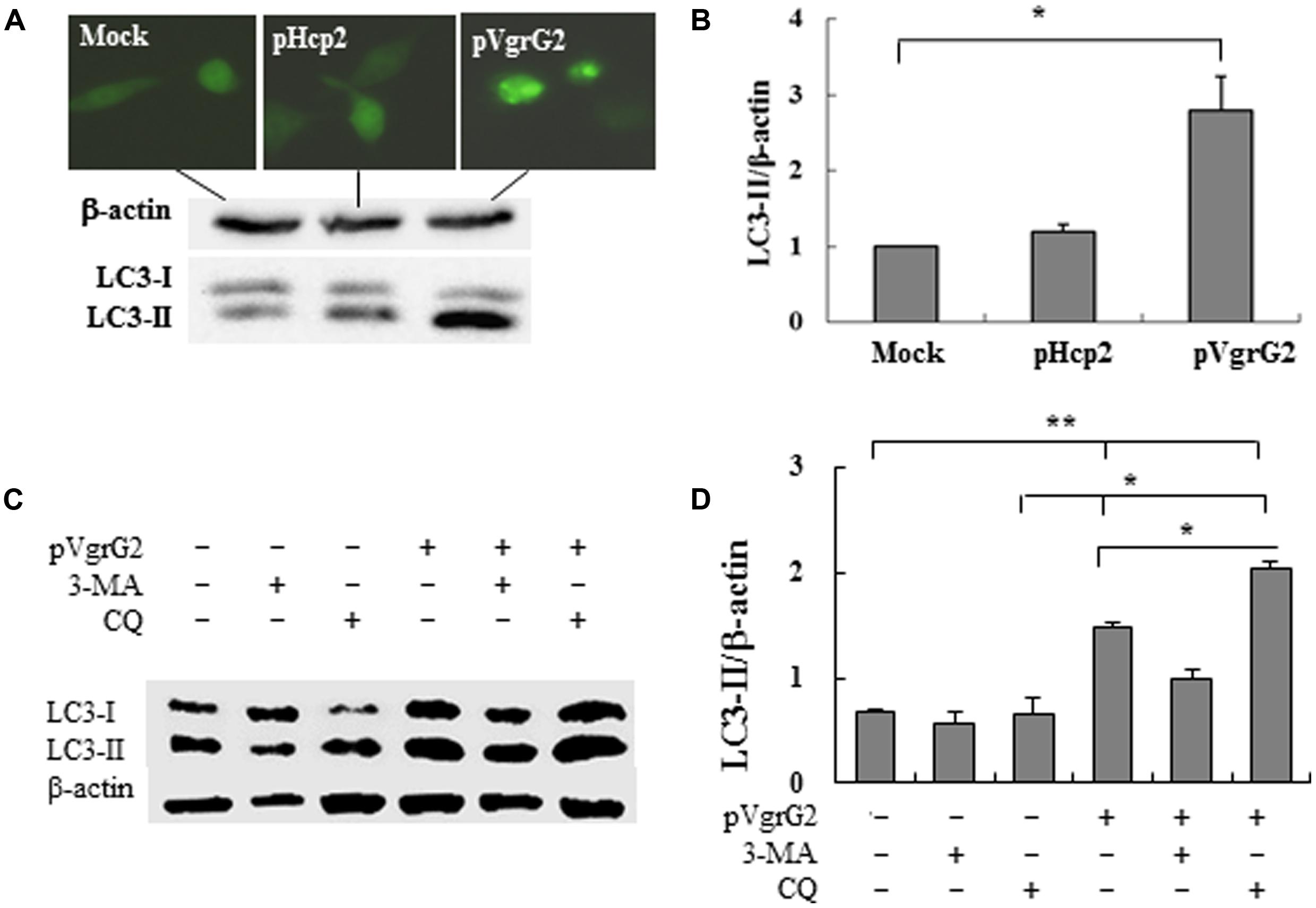
FIGURE 4. Vibrio parahaemolyticus T6SS2 VgrG2 is the effector protein inducing autophagy of macrophages. (A) Hcp2 and VgrG2 protein were expressed in macrophage cells as the EGFP fusion proteins. LC3-II accumulation was measured by immunoblotting. (B) Ratios of LC3-II to β-actin of ‘A’ were calculated and reported as mean ± SD of three independent experiments with the ratio of Mock cells (transfected with the control vector pcDNA-egfp) normalized to 1.0. (C) Macrophage cells were pretreated for 1 h with 3-methyl adenine (3-MA) or chloroquinine (CQ) and then transfected with pVgrG2 or pcDNA-egfp for analysis of LC3-II by Western blotting. (D) Ratios of LC3-II to β-actin were calculated and reported as mean ± SEM of three independent experiments. *P < 0.05; **P < 0.01.
RAW264.7 cells were pretreated with P13-kinase inhibitors 3-MA and lysosome-phagosome fusion inhibitors chloroquinine. 3-MA is competent in its ability to inhibit induction of autophagy by rapamycin, a TOR kinase inhibitor, and well-characterized inducer of autophagy (Burdette et al., 2009a). The VgrG2-induced autophagy appears to be dependent of PI3-kinase activation because treatment with 3-MA repressed VgrG2-induced LC3-II accumulation (Figures 4C,D). Chloroquinine is a inhibitor of lysosome–phagosome fusion, which blocks degradation of LC3-II (Menzies et al., 2012). Chloroquinine treatment increased LC3-II lipidation in VgrG2-expressing cells, suggesting that VgrG2 enhanced the autophagic flux. These results indicate that VgrG2 of V. parahaemolyticus induces autophagy by targeting the initial events of autophagic signaling.
Besides, Cyclic AMP is known to activate AMPK which might contribute to autophagy due to repression of mTOR (Shaw, 2009). Figure 5 indicates that the ΔvgrG2 mutant infected cells showed similar level of intracellular cAMP to the mock-infected cells, but had significantly lower level of intracellular cAMP than its parent strain, suggesting that V. parahaemolyticus vgrG2 might activate the cAMP signaling pathway to induce the autophagic response.
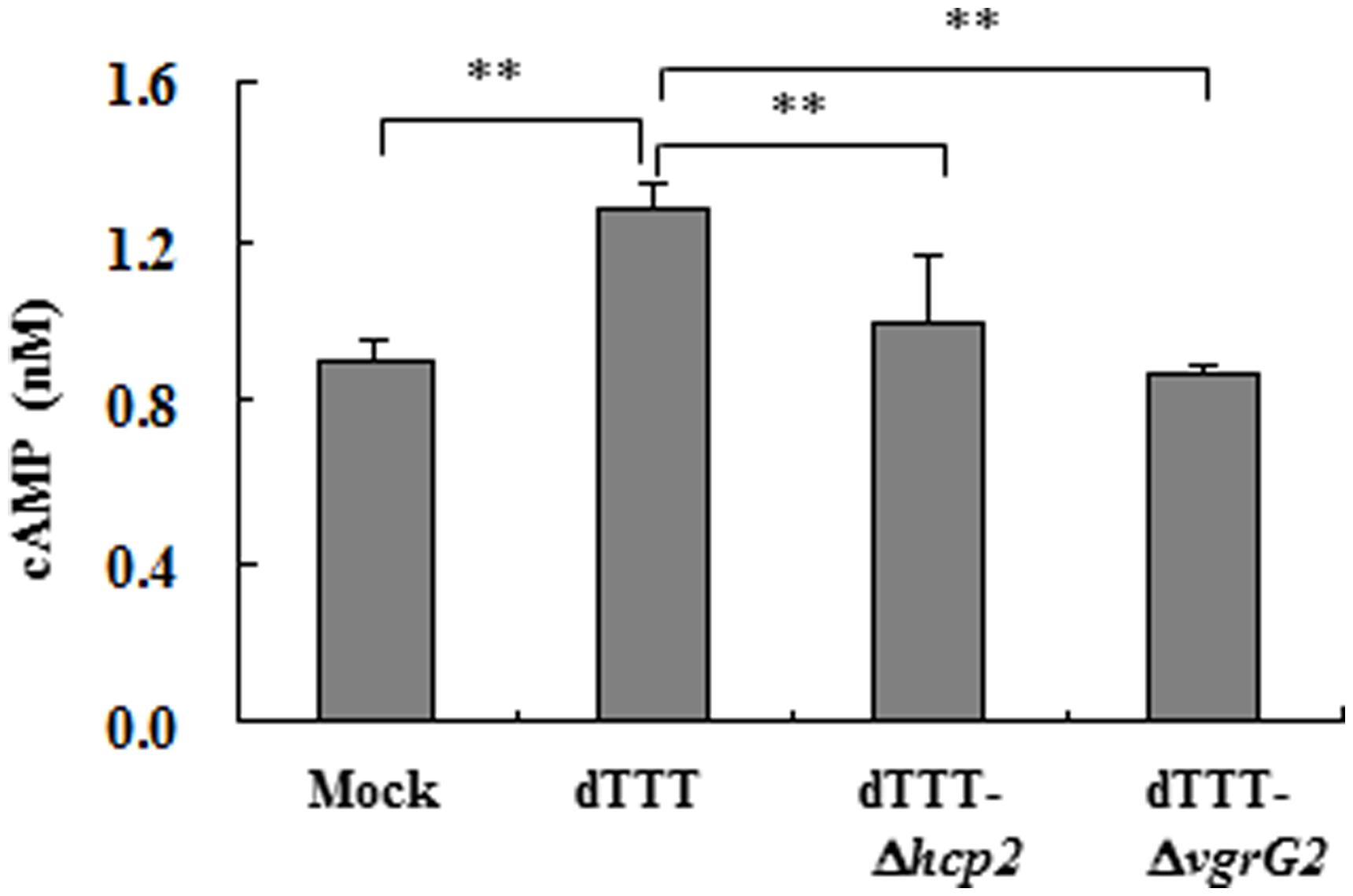
FIGURE 5. Intracellular cAMP of macrophages infected with V. parahaemolyticus vgrG2 deletion mutant was significantly reduced as compared with its parent triple deletion strain dTTT. The confluent RAW264.7 cell monolayers were infected with bacterial strains, each in triplicate wells, for 4 h. The culture supernatants were then removed, and the cell monolayers washed and lysed for cAMP determination. Each datum point represents mean ± SD of triplicate wells (**P < 0.01).
Discussion
Bacterial T6SS is a newly found secretion system which could be involved in pathogenesis and environment adaptation (Schwarz et al., 2010). Our previous study reveals that both T6SS1 and T6SS2 of V. parahaemolyticus contribute to adherence to mammalian cells (Yu et al., 2012). Here, we further show that T6SS2 of V. parahaemolyticus is involved in autophagic response of macrophages RAW264.7.
Vibrio parahaemolyticus contains several cytotoxic factors such as TDH and effector proteins of T3SS1 and T3SS2 which disrupt mammalian cell structure and lead to release of cellular contents (Burdette et al., 2008, 2009a; Zhou et al., 2010). In this study, we found the absence of β-actin bands and altered motility patterns of LC3 in cells infected with the WT, Δtdh and ΔvcrD2 strains, but not in the ΔvcrD1 mutant. This might indicate that T3SS1 contributed to loss of β-actin as a result of acute autophagy accompanied with cell wall disruption and release of cellular contents, as shown in previous studies (Burdette et al., 2009a,b; Zhou et al., 2010). Recent studies show that two T3SS1 effectors, VopQ and Vp1659, contribute to autophagy in eukaryotic cells (Burdette et al., 2009a; Zhou et al., 2010). In order to examine autophagy induction by T6SS independent of other factors and to avoid interference by cytotoxic effects, we constructed T6SS deletion mutants based on the triple deletion mutant void of tdh, vcrD1, and vcrD2 (strain dTTT) as the parent strain. It is clear that the triple deletion mutant dTTT and the mutant strains having further deletions of T6SS genes only showed marginal cytotoxicity as shown by LDH release.
Initially, we found that the multiple deletion mutant dTTT-ΔicmF2, but not dTTT-ΔicmF1, reduced autophagic response upon 4 h of infection of the macrophage, suggesting involvement of the putative T6SS2. Since icmF1 or icmF2 is the inner membrane protein of T6SS (Cascales, 2008), we attempted to search for the possible effector proteins that could be responsible for direct induction of autophagy by further deletion of the genes encoding Hcp2 and VgrG2, two putative translocons of T6SS2 seen in several bacterial species (Cascales, 2008) and also in T6SS2 of V. parahaemolyticus according to our previous study (Yu et al., 2012). Deletion of either hcp2 or vgrG2 did lessen the autophagic response as shown by decreased ratio of LC3-II to β-actin. However, increased LC3-II lipidation was seen only in the macrophage cells transfected with pVgrG2, but not with pHcp2. Therefore, we conclude that VgrG2, but not Hcp2, is the autophagy inducer of V. parahaemolyticus. Hcp might form as the tubular structure of VpT6SS for effector proteins including VgrG to translocate across the bacterial cell wall (Cascales and Cambillau, 2012). Deletion of hcp2 could apparently prevent VgrG2 translocation, leading to decreased LC3-II.
The fact that chloroquinine treatment caused accumulation of LC3-II indicates that VgrG2 could probably act on the upstream autophagic pathways. We also provide evidence that the cAMP signaling might be involved in autophagy induction by VgrG2 of V. parahaemolyticus since vgrG2 deletion led to reduced level of intracellular cAMP as compared with its parent strain. Different pathogens might explore distinct mechanisms in autophagic responses. Staphylococcus aureus was reported to enhance non-canonical autophagic response by its α-hemolysin in a PI3K/Beclin1-independent way, which is inhibitable by treating the cells with a permeable dibutyryl cAMP (a cAMP analog; Mestre and Colombo, 2012). They found that such inhibition was related to recruitment of Epac (Rap guanine nucleotide exchange factor/exchange protein activated by cAMP) and Rap2b via calpain activation. However, Chen et al. (2013) show that cAMP was necessary for resveratrol-induced PRKA-AMPK-SIRT1 activation of autophagy in human umbilical vein endothelial cells treated with this naturally occurring phytoalexin compound). Therefore, the possible mechanisms of cAMP signaling in V. parahaemolyticus VgrG2 induced autophagy require further investigation.
In conclusion, V. parahaemolyticus is clearly equipped with two arms of autophagy inducers, T3SS1 and T6SS2. However, T6SS2 induces autophagy without involving cytotoxic effect as seen in T3SS1.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Dr. T. Iida at Osaka University for providing the suicide vector pYAK1. This research is funded by Natural Science Foundation of China (31302120) and Zhejiang Provincial Natural Science Foundation of China (LQ13H90004).
References
Bernard, C. S., Brunet, Y. R., Gueguen, E., and Cascales, E. (2010). Nooks and crannies in type VI secretion regulation. J. Bacteriol. 192, 3850–3860. doi: 10.1128/JB.00370-10
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Bingle, L. E., Bailey, C. M., and Pallen, M. J. (2008). Type VI secretion: a beginner’s guide. Curr. Opin. Microbiol. 11, 3–8. doi: 10.1016/j.mib.2008.01.006
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Boyer, F., Fichant, G., Berthod, J., Vandenbrouck, Y., and Attree, I. (2009). Dissecting the bacterial type VI secretion system by a genome wide in silico analysis: what can be learned from available microbial genomic resources? BMC Genomics 10:104. doi: 10.1186/1471-2164-10-104
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Burdette, D. L., Seemann, J., and Orth, K. (2009a). Vibrio VopQ induces PI3-kinase-independent autophagy and antagonizes phagocytosis. Mol. Microbiol. 73, 639–649. doi: 10.1111/j.1365-2958.2009.06798.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Burdette, D. L., Yarbrough, M. L., and Orth, K. (2009b). Not without cause: Vibrio parahaemolyticus induces acute autophagy and cell death. Autophagy 5, 100–102. doi: 10.4161/auto.5.1.7264
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Burdette, D. L., Yarbrough, M. L., Orvedahl, A., Gilpin, C. J., and Orth, K. (2008). Vibrio parahaemolyticus orchestrates a multifaceted host cell infection by induction of autophagy, cell rounding, and then cell lysis. Proc. Natl. Acad. Sci. U.S.A. 105, 12497–12502. doi: 10.1073/pnas.0802773105
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Cascales, E. (2008). The type VI secretion toolkit. EMBO Rep. 9, 735–741. doi: 10.1038/embor.2008.131
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Cascales, E., and Cambillau, C. (2012). Structural biology of type VI secretion systems. Philos. Trans. R. Soc. Lond. B Biol. Sci. 367, 1102–1111. doi: 10.1098/rstb.2011.0209
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Chen, M. L., Yi, L., Jin, X., Liang, X. Y., Zhou, Y., Zhang, T.,et al. (2013). Resveratrol attenuates vascular endothelial inflammation by inducing autophagy through the cAMP signaling pathway. Autophagy 9, 2033–2045. doi: 10.4161/auto.26336
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Deretic, V., and Levine, B. (2009). Autophagy, immunity, and microbial adaptations. Cell Host Microbe 5, 527–549. doi: 10.1016/j.chom.2009.05.016
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Gutierrez, M. G., Saka, H. A., Chinen, I., Zoppino, F. C., Yoshimori, T., Bocco, J. L.,et al. (2007). Protective role of autophagy against Vibrio cholerae cytolysin, a pore-forming toxin from V. cholerae. Proc. Natl. Acad. Sci. U.S.A. 104, 1829–1834. doi: 10.1073/pnas.0601437104
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Jani, A. J., and Cotter, P. A. (2010). Type VI secretion: not just for pathogenesis anymore. Cell Host Microbe 8, 2–6. doi: 10.1016/j.chom.2010.06.012
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Kaper, J. B., Campen, R. K., Seidler, R. J., Baldini, M. M., and Falkow, S. (1984). Cloning of the thermostable direct or Kanagawa phenomenon-associated hemolysin of Vibrio parahaemolyticus. Infect. Immun. 45, 290–292.
Levine, B., and Deretic, V. (2007). Unveiling the roles of autophagy in innate and adaptive immunity. Nat. Rev. Immunol. 7, 767–777. doi: 10.1038/nri2161
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Liu, X., Chen, Y., Wang, X., and Ji, R. (2004). Foodborne disease outbreaks in China from 1992 to 2001 national foodborne disease surveillance system. Wei Sheng Yan Jiu 33, 725–727.
Menzies, F. M., Moreau, K., Puri, C., Renna, M., and Rubinsztein, D. C. (2012). Measurement of autophagic activity in mammalian cells. Curr. Protoc. Cell Biol. Chap. 15, Unit 1516. doi: 10.1002/0471143030.cb1516s54
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Mestre, M. B., and Colombo, M. I. (2012). cAMP and EPAC are key players in the regulation of the signal transduction pathway involved in the alpha-hemolysin autophagic response. PLoS Pathog. 8:e1002664. doi: 10.1371/journal.ppat.1002664
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Mizushima, N., Yoshimori, T., and Levine, B. (2010). Methods in mammalian autophagy research. Cell 140, 313–326. doi: 10.1016/j.cell.2010.01.028
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Mougous, J. D., Cuff, M. E., Raunser, S., Shen, A., Zhou, M., Gifford, C. A.,et al. (2006). A virulence locus of Pseudomonas aeruginosa encodes a protein secretion apparatus. Science 312, 1526–1530. doi: 10.1126/science.1128393
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Okada, N., Iida, T., Park, K. S., Goto, N., Yasunaga, T., Hiyoshi, H.,et al. (2009). Identification and characterization of a novel type III secretion system in trh-positive Vibrio parahaemolyticus strain TH3996 reveal genetic lineage and diversity of pathogenic machinery beyond the species level. Infect. Immun. 77, 904–913. doi: 10.1128/IAI.01184-08
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Park, K. S., Ono, T., Rokuda, M., Jang, M. H., Okada, K., Iida, T.,et al. (2004). Functional characterization of two type III secretion systems of Vibrio parahaemolyticus. Infect. Immun. 72, 6659–6665. doi: 10.1128/IAI.72.11.6659-6665.2004
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Pukatzki, S., Ma, A. T., Revel, A. T., Sturtevant, D., and Mekalanos, J. J. (2007). Type VI secretion system translocates a phage tail spike-like protein into target cells where it cross-links actin. Proc. Natl. Acad. Sci. U.S.A. 104, 15508–15513. doi: 10.1073/pnas.0706532104
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Pukatzki, S., Ma, A. T., Sturtevant, D., Krastins, B., Sarracino, D., Nelson, W. C.,et al. (2006). Identification of a conserved bacterial protein secretion system in Vibrio cholerae using the Dictyostelium host model system. Proc. Natl. Acad. Sci. U.S.A. 103, 1528–1533. doi: 10.1073/pnas.0510322103
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Pukatzki, S., Mcauley, S. B., and Miyata, S. T. (2009). The type VI secretion system: translocation of effectors and effector-domains. Curr. Opin. Microbiol. 12, 11–17. doi: 10.1016/j.mib.2008.11.010
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Schwarz, S., Hood, R. D., and Mougous, J. D. (2010). What is type VI secretion doing in all those bugs? Trends Microbiol. 18, 531–537. doi: 10.1016/j.tim.2010.09.001
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Shaw, R. J. (2009). LKB1 and AMP-activated protein kinase control of mTOR signalling and growth. Acta Physiol. (Oxf.) 196, 65–80. doi: 10.1111/j.1748-1716.2009.01972.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Suarez, G., Sierra, J. C., Sha, J., Wang, S., Erova, T. E., Fadl, A. A.,et al. (2008). Molecular characterization of a functional type VI secretion system from a clinical isolate of Aeromonas hydrophila. Microb. Pathog. 44, 344–361. doi: 10.1016/j.micpath.2007.10.005
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Weber, B., Hasic, M., Chen, C., Wai, S. N., and Milton, D. L. (2009). Type VI secretion modulates quorum sensing and stress response in Vibrio anguillarum. Environ. Microbiol. 11, 3018–3028. doi: 10.1111/j.1462-2920.2009.02005.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Yeung, P. S., and Boor, K. J. (2004). Epidemiology, pathogenesis, and prevention of foodborne Vibrio parahaemolyticus infections. Foodborne Pathog. Dis. 1, 74–88. doi: 10.1089/153531404323143594
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Yu, Y., Yang, H., Li, J., Zhang, P., Wu, B., Zhu, B.,et al. (2012). Putative type VI secretion systems of Vibrio parahaemolyticus contribute to adhesion to cultured cell monolayers. Arch. Microbiol. 194, 827–835. doi: 10.1007/s00203-012-0816-z
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Zheng, J., and Leung, K. Y. (2007). Dissection of a type VI secretion system in Edwardsiella tarda. Mol. Microbiol. 66, 1192–1206. doi: 10.1111/j.1365-2958.2007.05993.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Zhou, X., Konkel, M. E., and Call, D. R. (2010). Vp1659 is a Vibrio parahaemolyticus type III secretion system 1 protein that contributes to translocation of effector proteins needed to induce cytolysis, autophagy, and disruption of actin structure in HeLa cells. J. Bacteriol. 192, 3491–3502. doi: 10.1128/JB.01493-09
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Zhu, B., Xu, F., Li, J., Shuai, J., Li, X., and Fang, W. (2012). Porcine circovirus type 2 explores the autophagic machinery for replication in PK-15 cells. Virus Res. 163, 476–485. doi: 10.1016/j.virusres.2011.11.012
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Keywords: Vibrio parahaemolyticus, type VI secretion system, secretion system, effector proteins, autophagy
Citation: Yu Y, Fang L, Zhang Y, Sheng H and Fang W (2015) VgrG2 of type VI secretion system 2 of Vibrio parahaemolyticus induces autophagy in macrophages. Front. Microbiol. 6:168. doi: 10.3389/fmicb.2015.00168
Received: 22 December 2014; Accepted: 13 February 2015;
Published online: 02 March 2015.
Edited by:
Indrani Karunasagar, Karnataka Veterinary Animal and Fisheries Sciences University, IndiaReviewed by:
Pendru Raghunath, Dr. VRK Women’s Medical College, IndiaJessica L. Jones, United Stated Food and Drug Administration, USA
Romilio T. Espejo, Universidad de Chile, Chile
Copyright © 2015 Yu, Fang, Zhang, Sheng and Fang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Weihuan Fang, Institute of Preventive Veterinary Medicine and Zhejiang Provincial Key Laboratory of Preventive Veterinary Medicine, Zhejiang University, 388 Yuhangtang Road, Hangzhou 310058, China e-mail:d2hmYW5nQHpqdS5lZHUuY24=
†These authors have contributed equally to this work.
 Ying Yu
Ying Yu Lihua Fang
Lihua Fang Yan Zhang
Yan Zhang Hongxia Sheng
Hongxia Sheng Weihuan Fang
Weihuan Fang
