- 1Life and Environmental Sciences and Sierra Nevada Research Institute, University of California at Merced, Merced, CA, USA
- 2Monterey Bay Aquarium Research Institute, Moss Landing, CA, USA
Microbial communities play central roles in ocean biogeochemical cycles, and are particularly important in in oceanic oxygen minimum zones (OMZs). However, the key carbon, nitrogen, and sulfur (S) cycling processes catalyzed by OMZ microbial communities are poorly constrained spatially, temporally, and with regard to the different microbial groups involved. Here we sample across dissolved oxygen (DO) gradients in the oceans’ largest OMZ by volume—the eastern tropical North Pacific ocean, or ETNP—and quantify 16S rRNA and functional gene transcripts to detect and constrain the activity of different S-cycling groups. Based on gene expression profiles, putative dissimilatory sulfite reductase (dsrA) genes are actively expressed within the ETNP OMZ. dsrA expression was limited almost entirely to samples with elevated nitrite concentrations, consistent with previous observations in the Eastern Tropical South Pacific OMZ. dsrA and ‘reverse’ dissimilatory sulfite reductase (rdsrA) genes are related and the associated enzymes are known to operate in either direction—reducing or oxidizing different S compounds. We found that rdsrA genes and soxB genes were expressed in the same samples, suggestive of active S cycling in the ETNP OMZ. These data provide potential thresholds for S cycling in OMZs that closely mimic recent predictions, and indicate that S cycling may be broadly relevant in OMZs.
Introduction
Large areas of the ocean are characterized by low oxygen concentrations. These naturally occurring oxygen minimum zones (OMZs) are regions of pelagic ocean where dissolved oxygen (DO) concentrations are depleted below 20 μmol kg-1 due to microbial respiration (Helly and Levin, 2004; Paulmier and Ruiz-Pino, 2009; Gilly et al., 2013). OMZs are often associated with coastal and equatorial upwelling regions and the attendant high levels of microbial respiration driven by elevated primary production rates. OMZs are dominated by microbial communities, as much of the macrofauna of the pelagic ocean is unable to survive extended hypoxia (Diaz and Rosenberg, 2008; Gilly et al., 2013). At the same time, the total volume of OMZ waters is growing, their upper boundaries are vertically shoaling, and the degree of anoxia is intensifying within the cores of OMZs (Stramma et al., 2008; Gilly et al., 2013). These changes are collectively referred to as ‘ocean deoxygenation,’ and are driven by rising ocean temperatures that reduce oxygen solubility and increase water column stratification—both of which deplete DO concentrations at depth (Keeling et al., 2010).
The oxygen gradients found in OMZs create a chemically complex habitat that can sustain many types of microbial metabolism (Wright et al., 2012). While OMZs are well-known sites of nitrogen (N) cycling (Lam and Kuypers, 2011), recent work indicates that ‘cryptic’ sulfur (S) cycling occurs within the water column of the Eastern Tropical South Pacific Ocean (ETSP; Canfield et al., 2010). S metabolism is profoundly ancient, and genes related to S oxidation and reduction are dispersed throughout the Bacterial and Archaeal domains (Ghosh and Dam, 2009). S-cycling microorganisms, in turn, are distributed throughout a wide range of habitats—from marine sediments and eutrophic ponds, to animal guts and volcanic hot springs—where S exists in a range of chemical forms and oxidation states (Muyzer and Stams, 2008). However, S cycling was previously thought to occur in a relatively limited capacity in the ocean, specifically within anoxic basins such as the Black Sea, and ocean sediments (Canfield, 1989; Jørgensen et al., 1991). Active S cycling in the water column of open ocean OMZs has several implications for ocean biology and biogeochemistry, including the fact that hydrogen sulfide is toxic to large organisms (Lavik et al., 2009). The availability of different forms of S alters competitive and cooperative interactions among microbial groups and processes, and has an important bearing on organic matter remineralization and N cycling in OMZs (Johnston et al., 2014).
At the same time, several characteristic and abundant bacterial groups have been identified in OMZs and implicated in S cycling. This includes the γ-proteobacteria SUP05 and ARCTIC96BD-19 clades (Walsh et al., 2009), the δ-proteobacteria SAR324 (Swan et al., 2011; Sheik et al., 2014), and the Marine Group A (SAR406 clade; Wright et al., 2014). These groups typically comprise large percentages of OMZ microbial communities, although their specific niches are still ill-defined. Based on genomes and metagenomes, SUP05, ARCTIC96BD-19, and SAR324 are now believed to have chemolithoautotrophic metabolisms that utilize sulfide and thiosulfate, as well as oxidized N, as sources of energy (Walsh et al., 2009; Canfield et al., 2010; Swan et al., 2011; Sheik et al., 2014). The oxidation of S using N (nitrate or nitrite) is known as ‘chemoautotrophic denitrification’ and is distinct from conventional, heterotrophic denitrification (oxidation of organic carbon using nitrate or nitrite), potentially with higher energy yields (Lam and Kuypers, 2011). This process plays a dual biogeochemical role by removing toxic sulfide (Lavik et al., 2009) while concurrently converting N to gaseous forms that may be ‘lost’ from the system. Marine Group A bacteria have also been implicated in S cycling, potentially reducing S in the water column (Wright et al., 2014). However, these broader groups are each made up many operational taxonomic units (OTUs) that are heterogeneously distributed within OMZs (Wright et al., 2012); whether these represent different ecotypes with distinct biogeochemical roles and ecological niches is also unknown.
We used quantitative PCR (qPCR) of 16S rRNA and functional genes to determine the distribution, relative abundance, and potential activity of these S-utilizing bacteria across the oxycline of the eastern tropical North Pacific OMZ. These samples were collected before cryptic S cycling was identified in the ETSP; however, archived DNA and cDNA offer the possibility of examining S-cycling processes based on gene expression patterns. In the ETSP, S was reduced and oxidized so rapidly that it was not easily detectable based on chemical measurements, yet transcriptomics provided a way to track S cycling (Canfield et al., 2010). We apply this approach along oxygen gradients present in the Eastern Tropical North Pacific (ETNP)—the ocean’s largest OMZ by volume (Paulmier and Ruiz-Pino, 2009)—to determine (1) whether S cycling may occur, and (2) how it is distributed along environmental gradients.
Materials and Methods
Sample Collection and DNA and RNA Extraction
Samples were collected aboard the R/V New Horizon in the Gulf of California (GOC) and ETNP in July–August 2008 (Figure 1). DO concentrations were measured using a Seabird oxygen sensor on the CTD and were corrected based on Winkler titrations (r2 = 0.997, n = 187 for the cruise). DNA/RNA sample collection depths were selected to capture the transition from hypoxic to suboxic conditions in the OMZ across 4–6 depths spaced every 10–20 m. Additional details of sampling are provided in Beman et al. (2012, 2013), Beman and Carolan (2013).
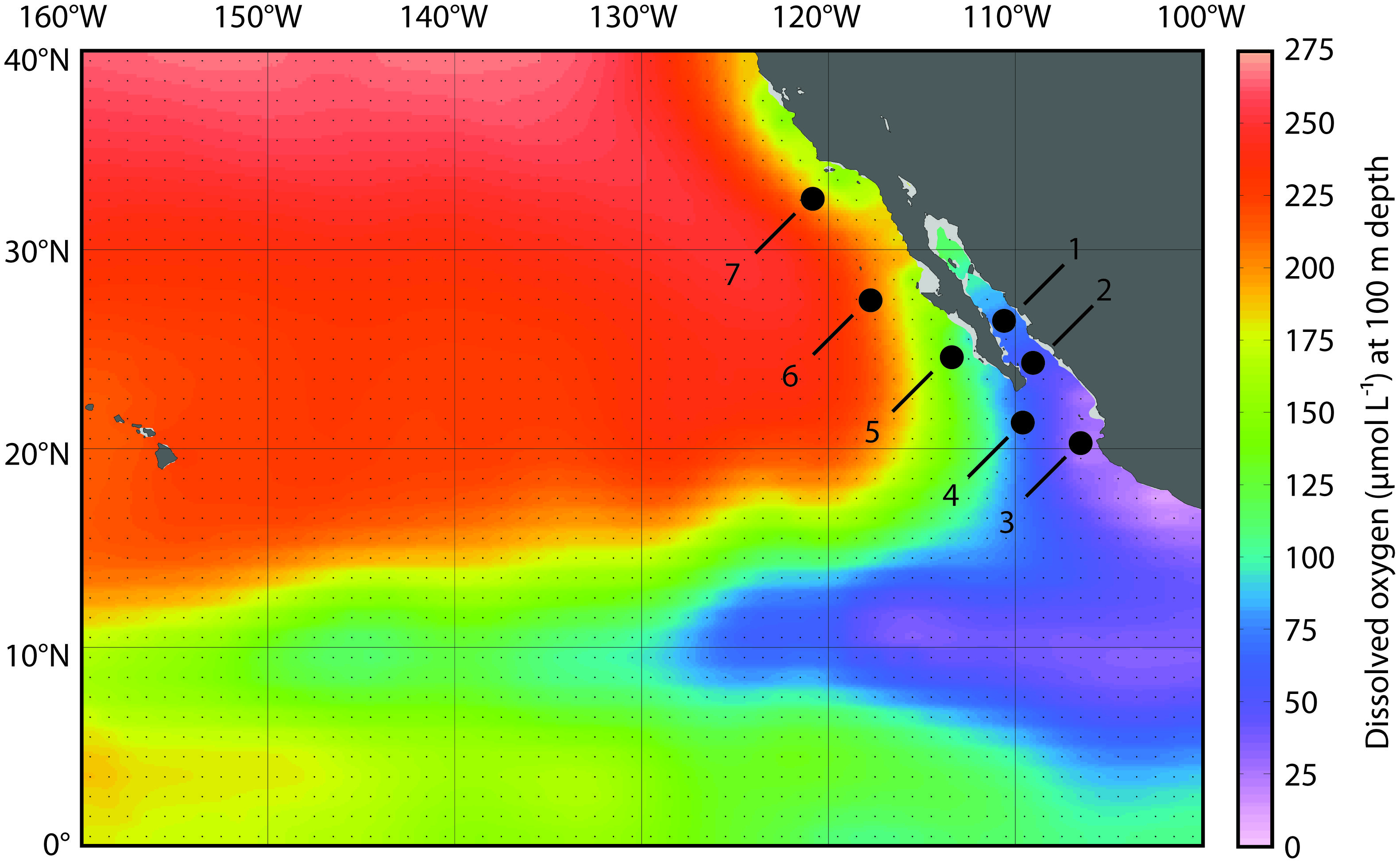
FIGURE 1. Map of sampling stations in the eastern tropical North Pacific Ocean (ETNP). Station locations are plotted on dissolved oxygen (DO) concentrations (μmol L-1) at 100 m depth from the World Ocean Atlas (plotted in Ocean Data View).
At each depth, two sets of 2 L samples were filtered through separate 25 mm diameter 0.2 μm Suppor filters (Pall Corporation, Port Washington, NY, USA) using a peristaltic pump. Filters were flash frozen in liquid nitrogen and stored at -80°C until DNA and RNA extraction, with one set of filters dedicated to each. Details of DNA extraction were reported in Beman et al. (2012, 2013). Details of RNA extraction are reported in Beman and Carolan (2013). In brief, RNA was extracted using the Qiagen RNeasy kit following Church et al. (2005), and treated with DNase to remove carry-over DNA; cDNA was generated from extracted RNA using the Invitrogen SuperScript III Reverse Transcriptase kit (Life Technologies Corporation, Carlsbad, CA, USA) following the manufacturer’s instructions; remaining RNA was removed through incubation with RNase H (Invitrogen).
Quantitative PCR and PCR
Quantitative PCR assays were performed on a Stragene MX3005P (Agilent Technologies, LA Jolla, CA, USA) using the following reaction chemistry: 12.5 μL SYBR Premix F (Epicentre Biotechnologies, Madison, WI, USA), 2 μL MgCl2, 1 μL of each primer, 0.25 μL AmpliTaq polymerase (Applied Biosystems, Life Technologies Corporation, Carlsbad, CA, USA), 40 ng μL-1 BSA, and 1 ng DNA in a final volume of 25 μL. Details of assays used are provided in Table 1. All assays were taken directly from the literature, and used standards that were synthesized using GeneArt Gene Synthesis (Life Technologies Corporation, Carlsbad, CA, USA). This included 16S genes from Chromatiales and SUP05 bacteria, as well as (reverse) dissimilatory sulfite reductase (dsrA and rdsrA) genes, and the thiosulfate-oxidizing enzyme system (soxB) gene. qPCR efficiencies and R2 for standard curves are also provided in Table 1. The efficiency of the Chromatiales assay is on the high end of acceptability for qPCR and likely stems from the use of a universal primer in this assay. For some assays, we tested only for presence/absence or expression/lack of expression within samples, and did not quantify gene abundances (see below). No efficiencies or r2 values are reported for these assays, as we used positive controls but did not generate a standard curve.

TABLE 1. Details of quantitative PCR (qPCR) assays, including the primers used, the reference for the qPCR protocol, and the efficiency and r2 of standard curves.
Results and Discussion
Oxygen Gradients
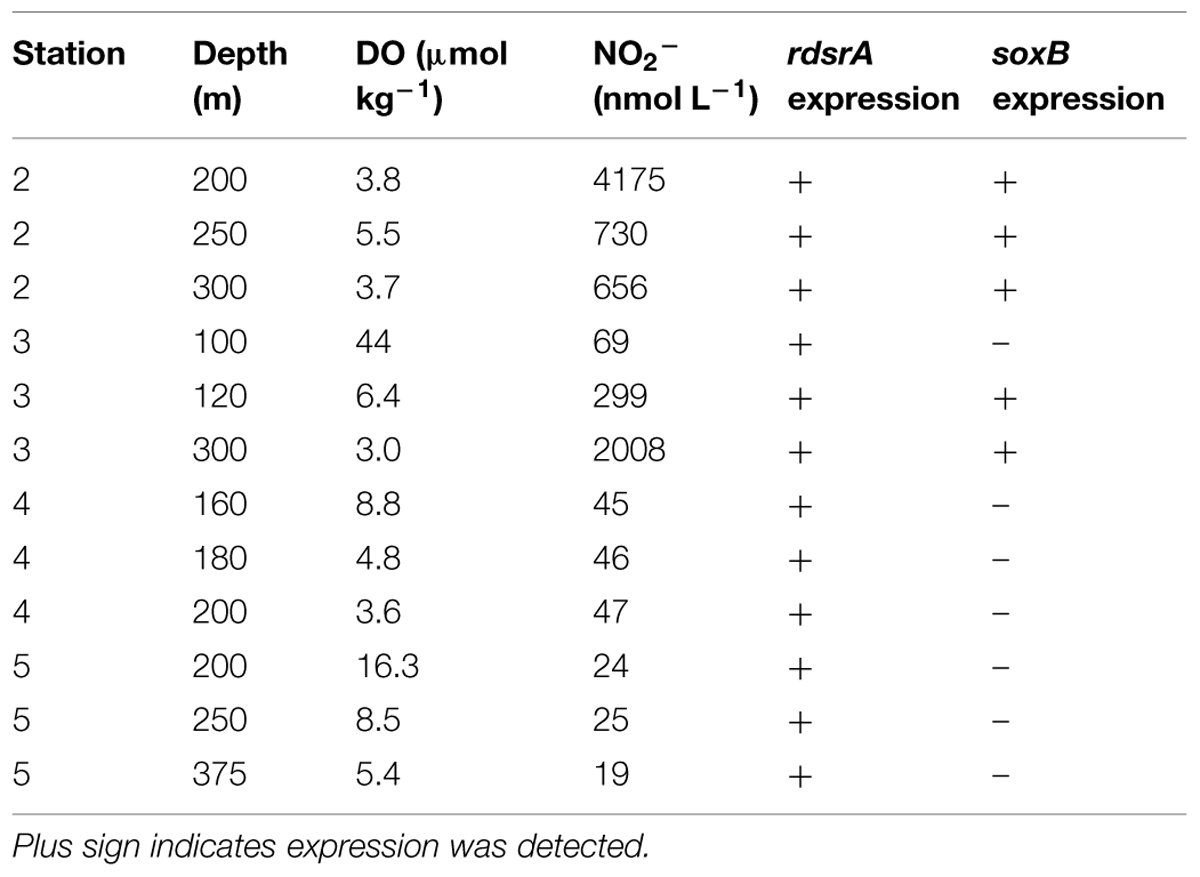
Dissolved oxygen concentrations decline with depth in the ocean, and in OMZ regions they drop below 20 μmol kg-1. In the ETNP, these low DO values typically span several 100 m of the water column; they extend laterally from the coast of Mexico and Central America across the Pacific Ocean, north along the coast of Baja California, and also extend into the GOC (the body of water defined by the Baja California peninsula and mainland Mexico). We sampled a series of seven stations in the ETNP and GOC (Figure 1A) that extend across this DO gradient: station 1 was located in the Carmen Basin of the GOC, station 2 was located near the mouth of the GOC, and station 3 was located in the core of the ETNP OMZ—where DO concentrations declined rapidly with depth and dropped to 20 μmol kg-1 by 111 m (Figure 2). Four additional stations extended north from the tip of Baja (station 4) to several 100 km off the coast of San Diego, California (station 7). We previously sequenced 16S rRNA genes using 454 pyrosequencing to characterize microbial communities in the upper water column and OMZ (Beman and Carolan, 2013); here we focus on the four stations (number 2–5) that have a well-developed OMZ.
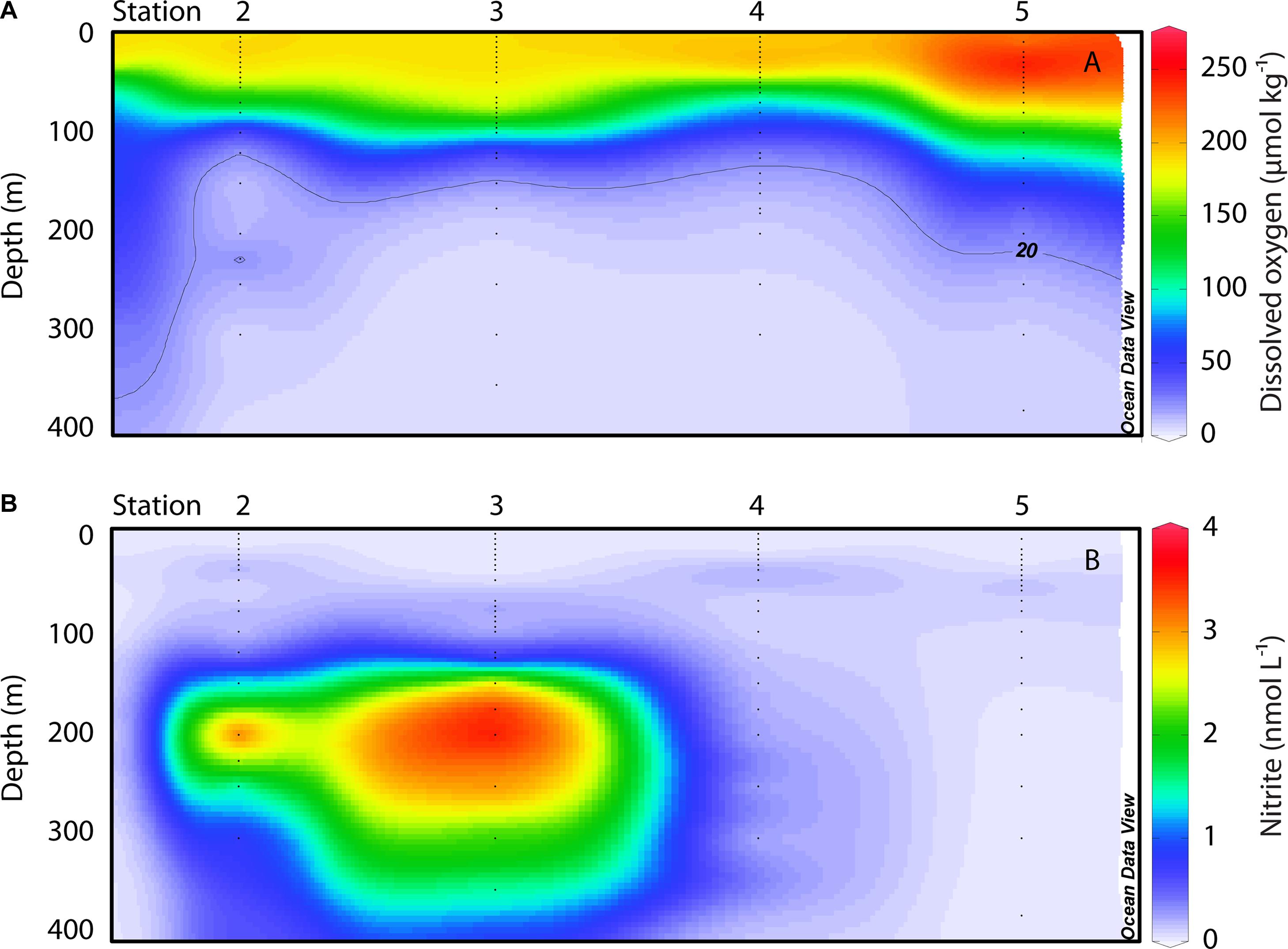
FIGURE 2. Oceanographic sections of (A) DO (μmol kg-1) and (B) nitrite (nmol L-1). The location of individual sampling stations is indicated along the horizontal axis; vertical axes display depth down to 400 m; color scales show concentrations. Small black dots denote the depths of bottle samples collected for nitrite measurements, and discrete oxygen data for the same depths were extracted from the Winkler-corrected CTD data. Data were gridded in Ocean Data View.
The comparison among these stations provides systematic gradients in DO and nitrite (NO2-) concentrations with depth and between stations. DO reaches lower absolute values and the OMZ generally thickens, from north to south. OMZ waters (DO <20 μmol kg-1) were located at 91 m depth at station 2 (with an intrusion of water with slightly higher DO concentrations at 220–255 m), 111 m at station 3, 121 m at station 4, and 178 m at station 5. However, a recent review by Ulloa et al. (2012) distinguishes between typical OMZs and what they refer to as ‘anoxic marine zones’ (AMZs), where DO concentrations drop below 50 nM. They note these anoxic waters are also characterized by the accumulation of NO2- in secondary nitrite maxima. In the ETNP, prominent secondary NO2- maxima were observed at stations 2 and 3 in OMZ waters (Figure 2). NO2- concentrations at these depths were an order of magnitude greater than in the primary nitrite maximum (PNM) at the base of the euphotic zone, reaching up to 4.18 μmol L-1 in the OMZ at station 2 and 4.24 μmol L-1 in the OMZ at station 3. This feature extended from ∼125–500 m at stations 2 and 3. In contrast, station 4 had low DO at depth, but NO2- concentrations did not rise above 114 nM; at station 5, OMZ waters were found starting at 178 m, and the NO2- concentration did not rise above 30 nM. Low DO waters define OMZs and allow for anaerobic N cycling to proceed; while it was previously thought that sulfate would only be exploited when oxidized N species were sufficiently depleted, recent evidence from the Eastern Tropical South Pacific OMZ indicates that a cryptic S cycle may occur in AMZs (Canfield et al., 2010; Ulloa et al., 2012). Our biogeochemical data indicate that stations 4 and 5 represent OMZs, whereas stations 2 and 3 represent AMZs.
Abundance and Activity of Sulfur Cycling Bacterial Groups
Along these gradients we quantified specific groups and genes and also examined their activity: amplification of particular genes in DNA indicates the presence of a particular group or gene, whereas amplification in cDNA indicates that a particular group is actively growing or a particular gene is actively expressed. Our previous sequencing data indicated relatively high abundances of Chromatiales (Beman and Carolan, 2013), or purple-sulfur bacteria, a group capable of either chemo- or photo-trophic growth under anoxia (Imhoff, 2005). In anoxic aquatic ecosystems, Chromatiales are typically found in a narrow band where sufficient amounts of both sulfide and light are found; sulfide and light almost invariably display opposing gradients with depth, but can together sustain anoxygenic photosynthesis (Imhoff, 2001; Overmann, 2001).
We quantified Chromatiales 16S rRNA to examine the distribution of these organisms with depth and from station to station. Chromatiales were readily detected in DNA and were present in the ETNP at all four stations, but did not appear to be highly active based on 16S rRNA genes in the cDNA fraction. Chromatiales 16S rRNA genes were detected at one depth at stations 2 and 5, and two depths at stations 3 and 4, and were most abundant at stations 2 and 3 (Figure 3). In contrast, 16S rRNA was not detected in most cDNA samples, and typically showed 1–2 orders of magnitude lower values than 16S rRNA genes in DNA. The ratio of 16S rRNA in cDNA versus DNA ranged from 0 to 0.36. Chromatiales 16S rRNA was most abundant at 120 m at station 3, but light levels at this depth—and in all other samples with Chromatiales 16S rRNA expression—were extremely low, and Chromatiales are most likely not photosynthetically active at these depths.
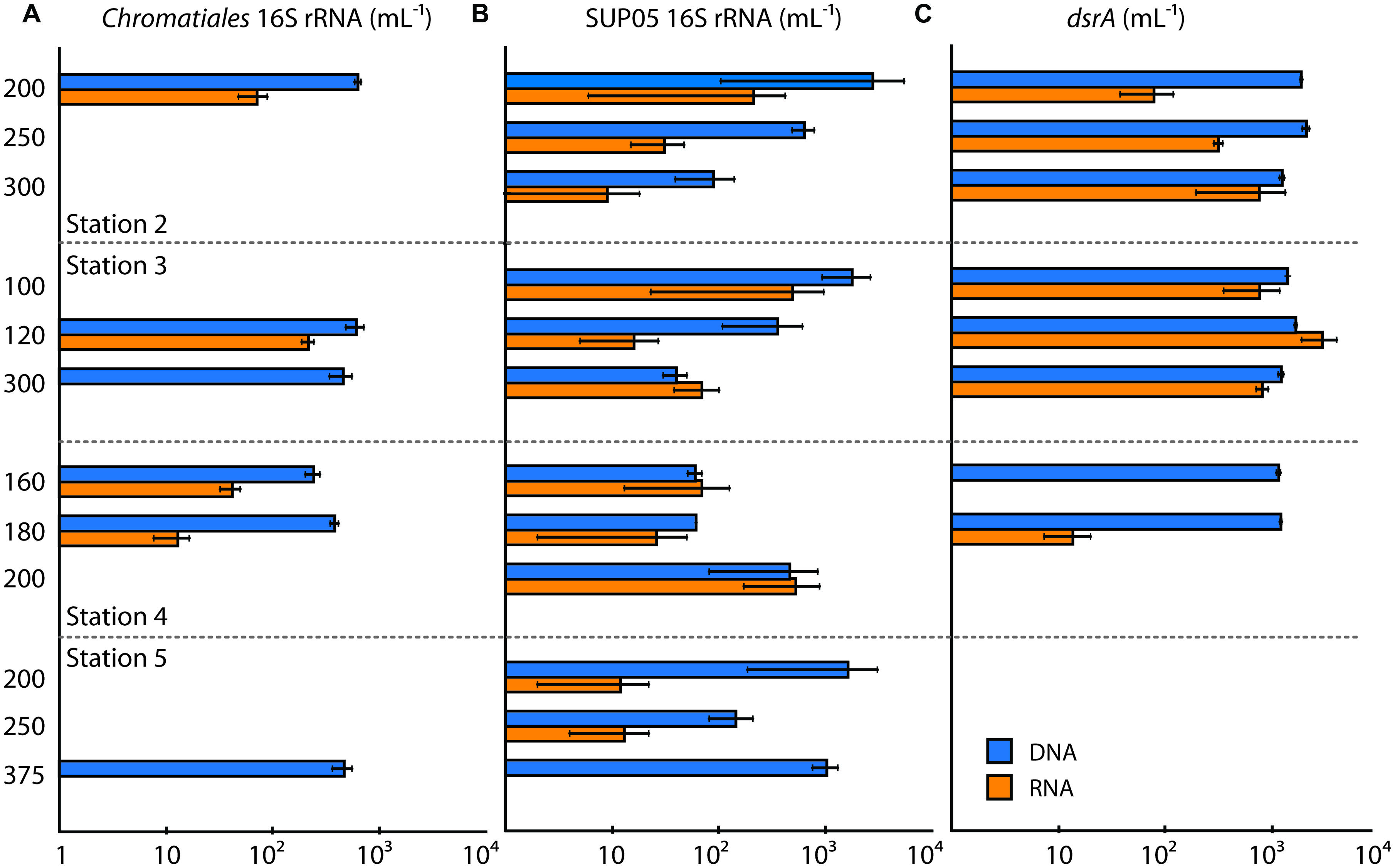
FIGURE 3. Quantitative PCR (qPCR) data for (A) Chromatiales 16S rRNA, (B) SUP05 16S rRNA, and (C) dsrA genes and transcripts (expressed per mL). Station and depth of sample collected are shown, and gene or transcript abundances are displayed along the horizontal axes (log scale). Abundances in DNA are shown in blue, and abundances in RNA (i.e., cDNA) are shown in orange. Error bars denote SD of the triplicate measurement.
We also quantified SUP05 Gammaproteobacteria, which are abundant in hypoxic and anoxic waters worldwide (Walsh et al., 2009), and have been implicated in the ‘detoxification’ of sulfide plumes in OMZs/AMZs (Lavik et al., 2009; Schunck et al., 2013). In these cases, SUP05 and other S-oxidizing bacteria may ‘bloom’ at levels of up to 105 cells ml-1. SUP05 were detected at all stations in the ETNP but were present in low abundance (<3000 16 rRNA genes ml-1; Figure 3). SUP05 were least abundant at station 4, and were present at higher levels at station 5—however, SUP05 were much less active at Station 5 than their numbers would suggest, and 16S rRNA was barely detectable in cDNA. At station 4, 16S rRNA profiles closely tracked 16S rRNA genes, showing comparable numbers and an increase with depth. In contrast, SUP05 were most abundant at stations 2 and 3—where there is a well-developed OMZ and AMZ—but were comparatively less active—with levels of 16S rRNA in cDNA that were 1–2 orders of magnitude lower than 16S rRNA genes in DNA. A key exception to this pattern was 300 m depth at station 3, where 16S rRNA in cDNA exceeded DNA values. This sample is characterized by low DO and high NO2-, and the relatively high levels of SUP05 activity are consistent with a role in chemoautotrophic denitrification—i.e., the oxidization of reduced S compounds using nitrate/NO2- (Walsh et al., 2009).
Despite these relatively high levels of 16S rRNA, the general pattern at stations 2 and 3 was declining SUP05 abundance and activity with increasing depth and NO2- concentrations, and decreasing DO concentrations. This is consistent with surveys of the Black Sea and Baltic Sea, where SUP05-related organisms occur at highest abundance at the interface of oxic and anoxic waters (Glaubitz et al., 2013). There was a surprising amount of variation in DO and NO2- concentrations where SUP05 were most abundant and active (at 200 m at station 2, 100 m at station 3, and 200 m at station 4; Figure 4). Stations 2 and 4 had low DO (<4 μmol kg-1) at 200 m, whereas DO concentrations where higher at 100 m at station 3; NO2- concentrations were elevated at 200 m at station 4 (>4000 nM), and were lower at 200 m at station 4 and 100 m at station 3. This is consistent with the idea that SUP05 have considerable metabolic diversity and may tolerate a range of geochemical conditions (Walsh et al., 2009; Swan et al., 2011; Murillo et al., 2014). In the ETSP, for example, SUP05 are active throughout much of the water column, and their metagenomes show evidence for S and N metabolism, as well as mixotrophy (Murillo et al., 2014).
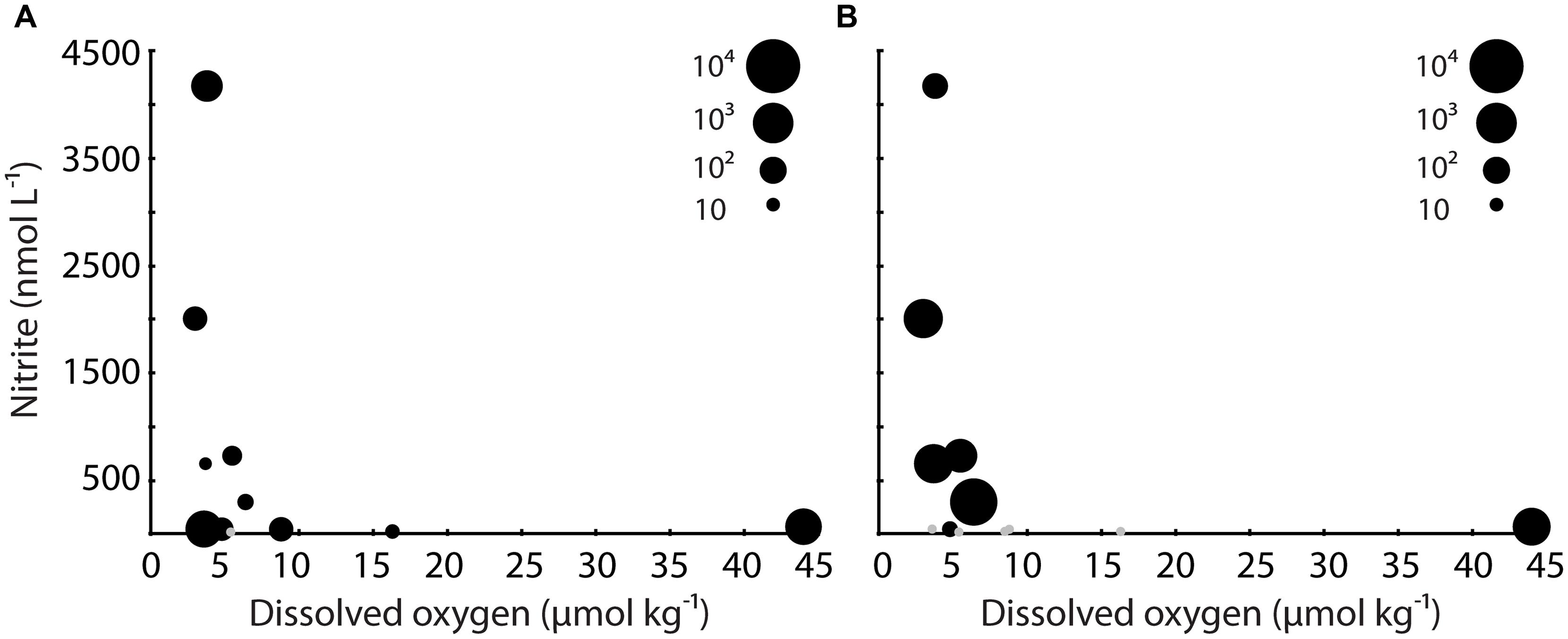
FIGURE 4. Bubble plot of qPCR data for (A) SUP05 16S rRNA and (B) dsrA plotted versus DO and nitrite. The size of the bubbles is proportional to level of expression. Gray datapoints indicate that no expression was detected.
Expression of Functional Genes involved in Sulfur and Nitrogen Cycling
To determine if a cryptic S cycle was possible in the ETNP OMZ, we quantified the DNA and cDNA gene copies for the dsrA gene, a hallmark of sulfate-reducing bacteria. The primers we used a specifically designed to target sulfate-reducing bacteria (Kondo et al., 2004), however, the dsrA gene is closely related to the rdsrA gene found within Chlorobi, Alpha-, Beta-, and Gammaproteobacteria (Loy et al., 2009). dsrA and rdsrA are known to act on sulfite, sulfate, and elemental S, allowing for diverse S metabolism (Ghosh and Dam, 2009), and may function in either the reductive or oxidative direction. dsrA expression is therefore indicative of S cycling, but in-depth omics and geochemical analyses are required to determine which S-cycling pathways are active at any given time.
Given the need for anaerobic conditions and the idea that NO2- is indicative of true anoxia (Ulloa et al., 2012), we expected that dsrA expression would be greatest in OMZ waters with high NO2- concentrations at stations 2 and 3. dsrA genes and transcripts were in fact most common and abundant at stations 2 and 3, supporting this hypothesis (Figure 3). dsrA genes were detected at 160 and 180 m at station 4 and were not detected at station 5. However, dsrA was expressed only at very low levels at 180 m at station 4, suggesting little to no activity outside of ‘AMZ’ conditions in the ETNP. These findings are similar to those of Stewart et al. (2012) in the ETSP, who found that rdsrA/dsrA genes were expressed throughout the OMZ, with the greatest proportional representation in the core of the OMZ. However, they identified ca. 90% of these transcripts as oxidative, rdsrA transcripts rather than reductive dsrA transcripts.
At station 2 in the ETNP, we found similar levels of dsrA genes throughout the water column (in DNA), but observed a pronounced increase in dsrA transcripts with increasing depth. This trend was not observed at station 3: instead, highest levels of expression occurred at 120 m depth, and dsrA was also expressed at 100 m, where DO was 44 μmol kg-1. There are several possible explanations for this, the first being that this is a spurious datapoint, as the remaining data all support the idea that dsrA is only expressed under low DO and elevated NO2. (Figure 3). However, there are several alternative explanations. First, DO declined rapidly with depth at station 3, dropping from 190 μmol kg-1 at 85 m depth to<20 μmol kg-1 at 111 m. Along this sharp oxygen gradient, microorganisms may experience a range of DO concentrations through time as supply and demand vary, or as they are transported through the water column. Second, sulfate reduction could occur on particles within anoxic microsites. Earlier research suggested that this might be a common phenomenon (Shanks and Reeder, 1993). It is also possible that this particular sample—or some proportion of the expressed dsrA genes—reflects S oxidation, rather than reduction.
To gain greater insight into S oxidation, we used qPCR to detect rdsrA using primers that target Alpha- and Gammaproteobacterial S-oxidizers, such as SUP05 and related lineages (Lavik et al., 2009). In our hands, we were not able to generate high quality PCR product (based on dissociation curves) and we report these data only in terms of presence/absence—i.e., was the gene detected and expressed, or not. rdsrA genes and transcripts were detected in all OMZ and AMZ samples collected in this study (Table 2). These findings are consistent with the presence and activity of SUP05 in these samples, as well as the large numbers of ARCTIC96BD-19 and SAR324 sequences recovered in our earlier work (Beman and Carolan, 2013). We also screened samples for the soxB, a gene that encodes for part of a thiosulfate oxidizing enzyme complex and is found in many bacterial lineages (Meyer et al., 2007; Ghosh and Dam, 2009). In contrast to rdsrA, soxB was only expressed at 200, 250, and 300 m depth at station 2 and 120 and 300 m at 3 (Table 2). These samples are all characterized by DO concentrations of 6.5 μmol kg-1 or lower and NO2- concentrations of 299 nM or greater, suggesting that soxB expression is confined to AMZ waters in the ETNP. Our previous analysis of nitrate reductase genes (narG and napA) also showed that narG, napA, or both, were expressed where DO was low and NO2- was elevated (presumably due to nitrate reduction to NO2-; Beman et al., 2013). S oxidation and conventional denitrification could occur independently of one another, but coincident expression of S oxidation and N reduction genes suggests that chemoautotrophic denitrification may occur in the ETNP.
Assembled together, these data demonstrate that S cycling is active throughout the ETNP OMZ stations sampled in this study. The activity of SUP05 bacteria is consistent with anaerobic S oxidation in the OMZ, and expression of dsrA, rdsrA, and soxB indicates that several S-cycling pathways and processes may be active in the ETNP. Without definitive geochemical data, we cannot establish whether sulfate reduction and a cryptic S cycle are active in the ETNP, but this is one of at least three interpretations of our transcriptomic data. Recent analyses indicate that the clear separation between reductive dsrA gene sequences and oxidative rdsrA sequences allows for robust detection of these individual genes (Müller et al., 2014), and the dsrA primers that we used specifically target the reductive dsrA gene present in sulfate reducing bacteria (Kondo et al., 2004; Leloup et al., 2007; Müller et al., 2014). Alternatively, dsrA expression—along with rdsrA, and soxB—may capture S oxidation only. Given the expression patterns with depth and as a function of DO and nitrite, this is likely anaerobic S oxidation using oxidized N. This idea is supported by expression of nitrate reductase genes at the same depths (Beman et al., 2013). Chemoautotrophic denitrification has implications for N loss processes in the ETNP—which is still understudied compared with the ETSP and Arabian Sea, and is particularly relevant given the volume of the ETNP relative to the other OMZs. Finally, a third interpretation of our data is that they capture additional S-cycling processes beyond oxidation and reduction. For example, Schunck et al. (2013) raise the possibility of S disproportionation in the ETSP based on the joint expression of dsrA, soxB, and other S-cycling genes. Based on the SAR324 metagenome, Sheik et al. (2014) discuss lithoheterotrophy as a potentially important form of metabolism, and the SUP05 metagenome recovered from the ETSP also supports this idea (Murillo et al., 2014). These findings add layers of complexity to OMZ S cycling, such that significant geochemical and ‘omics research will be necessary to unravel the multiple forms of S metabolism and their connections to C and N cycling. Our data represent the first piece of evidence for S metabolism in the ETNP OMZ, and suggest that S cycling is prevalent in the ocean’s OMZs. Determining the significance of S cycling in terms of rates, lateral extent, and depth distribution in the ETNP will be key for understanding the biogeochemical and ecological implications of water column S cycling in the ETNP, and globally.
Author Contributions
JMB designed the study; JMS and JMB collected the samples; MTC performed the laboratory work; MTC and JMS contributed new reagents and analytical tools; MTC and JMB analyzed the data; MTC wrote the paper; JMS and JMB edited the paper.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Rachel Foster, Brian Popp, chief scientist Fred Prahl, and the officers and crew of the R/V New Horizon for assistance with sample collection, and thank Jason Smith and Susan Alford for assistance in the laboratory. This work was supported by US National Science Foundation award OCE-1034943.
References
Beman, J. M., and Carolan, M. T. (2013). Deoxygenation alters bacterial diversity and community composition in the ocean’s largest oxygen minimum zone. Nat. Commun. 4, 2705. doi: 10.1038/ncomms3705
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Beman, J. M., Popp, B. N., and Alford, S. E. (2012). Quanitifcaiton of ammonia oxidation rates and ammnia oxidizing archaea and bacteria at high resolution in the Gulf of California and the eastern tropical North Pacific. Limnol. Oceanogr. 57, 711–726. doi: 10.4319/lo.2012.57.3.0711
Beman, J. M., Shi, J. L., and Popp, B. N. (2013). Nitrite oxidation in the upper water column and oxygen minimum zone of the eastern tropical North Pacific Ocean. ISME J. 7, 2192–2205. doi: 10.1038/ismej.2013.96
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Canfield, D. E. (1989). Sulfate reduction and oxic respiration in marine sediments: implications for organic carbon preservation in euxinic environments. Deep Sea Res. A 38, 121–138. doi: 10.1016/0198-0149(89)90022-8
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Canfield, D. E., Stewart, F. J., Thamdrup, B., Brabandere, L. D., Dalsgaard, T., Delong, E. F., et al. (2010). A cryptic sulfur cycle in the oxygen minimum zone waters off the chilean coast. Science 330, 1375–1378. doi: 10.1126/science.1196889
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Church, M. J., Short, C. M., Jenkins, B. D., Karl, D. M., and Zehr, J. P. (2005). Temporal patterns of nitrogenase gene (nifH) expression in the oligotrophic North Pacific Ocean. Appl. Environ. Microbiol. 71, 5362–5370. doi: 10.1128/AEM.71.9.5362-5370.2005
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Coolen, M. J., and Overmann, J. (1998). Analysis of subfossil molecular remains of purple sulfur bacteria in a lake sediment. Appl. Environ. Microbiol. 64, 4513–4521.
Diaz, R. J., and Rosenberg, R. (2008). Spreading dead zones and consequences for marine ecosystems. Science 321, 926–929. doi: 10.1126/science.1156401
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Ghosh, W., and Dam, B. (2009). Biochemistry and molecular biology of lithotrophic sulfur Oxidation by taxonomically and ecologically diverse bacteria and archaea. FEMS Microbiol. Rev. 33, 999–1043. doi: 10.1111/j.1574-6976.2009.00187.x
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Gilly, W. F., Beman, J. M., Litvin, S. Y., and Robinson, B. M. (2013). Oceanographic and biological effects of shoaling of the oxygen minimum zone. Ann. Rev. Mar. Sci. 5, 393–420. doi: 10.1146/annurev-marine-120710-100849
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Glaubitz, S., Kießlich, K., Meeske, C., Labrenz, M., and Jürgens, K. (2013). SUP05 dominates the gammaproteobacterial sulfur oxidizer assemblages in pelagic redoxclines of the central Baltic and Black Seas. Appl. Environ. Microbiol. 79, 2767–2776. doi: 10.1128/AEM.03777-12
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Helly, J. J., and Levin, L. A. (2004). Global distribution of naturally occurring marine hypoxia on continental margins. Deep Sea Res. I 51, 1159–1168. doi: 10.1016/j.dsr.2004.03.009
Imhoff, J. F. (2001). True marine and halophilic anoxygenic phototrophic bacteria. Arch. Microbiol. 178, 243–254. doi: 10.1007/s002030100326
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Imhoff, J. F. (2005). “Chromatiales ord. nov,” in Bergey’s Manual of Systematic Bacteriology,” 2nd Edn, eds D. J. Brenner, N. R. Krieg, J. T. Staley, and G. M. Garrity (New York, NY: Springer).
Johnston, D., Gill, B., Masterson, A., Beirne, E., Casciotti, K., Knapp, A., et al. (2014). Placing an upper limit on cryptic marine sulphur cycling. Nature 513, 530–533. doi: 10.1038/nature13698
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Jørgensen, B. B., Fusing, H., Wirsen, C. O., and Jannasen, H. W. (1991). Sulfide oxidation in the anoxic Black Sea chemocline. Deep Sea Res. A 38, S1083–S1103. doi: 10.1016/S0198-0149(10)80025-1
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Keeling, R. F., Kurtzinger, A., and Gruber, N. (2010). Ocean deoxygenation in a warming world. Ann. Rev. Mar. Sci. 2, 199–229. doi: 10.1146/annurev.marine.010908.163855
Kondo, R., Nedwell, D. B., Purdy, K. J., and Silva, S. Q. (2004). Detection and enumeration of sulphate-reducing bacteria in estuarine sediments by competitive PCR. Geomicrobiol. J. 21, 145–157. doi: 10.1080/01490450490275307
Lam, P., and Kuypers, M. M. M. (2011). Microbial nitrogen cycling processes in oxygen minimum zones. Annu. Rev. Marine Sci. 3, 317–345. doi: 10.1146/annurev-marine-120709-142814
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Lavik, G. T., Stuührmann, V., Brüchert, A., Van der Plas, V., Mohrholz, P., Lam, M., et al. (2009). Detoxification of sulphidic African shelf waters by blooming chemolithotrophs. Nature 457, 581–584. doi: 10.1038/nature07588
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Leloup, J., Loy, A., Knab, N. J., Borowski, C., Wagner, M., and Jørgensen, B. B. (2007). Diversity and abundance of sulfate-reducing microorganisms in the sulfate and methane zones of a marine sediment, Black Sea. Environ. Microbiol. 9, 131–142. doi: 10.1111/j.1462-2920.2006.01122.x
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Loy, A., Duller, S., Baranyi, C., Mushmann, M., Ott, J., Shanon, J., et al. (2009). Reverse dissimilatory sulfite reductase as a phylogenetic marker for a subgroup of sulfur oxidizing prokaryotes. Environ. Microbiol. 11, 289–299. doi: 10.1111/j.1462-2920.2008.01760.x
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Meyer, B., Imhoff, J. F., and Kuever, J. (2007). Molecular analysis of the distribution and phylogeny of the soxB gene among sulfur-oxidizing bacteria. Environ. Microbiol. 11, 2957–2977. doi: 10.1111/j.1462-2920.2007.01407.x
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Müller, A. L., Kjeldsen, K. U., Rattei, T., Pester, M., and Loy, A. (2014). Phylogenetic and environmental diversity of DsrAB-type dissimilatory (bi) sulfite reductases. ISME J. doi: 10.1038/ismej.2014.208 [Epub ahead of print].
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Murillo, A. A., Ramírez-Flandes, S., DeLong, E. F., and Ulloa, O. (2014). Enhanced metabolic versatility of planktonic sulfur-oxidizing γ-proteobacteria in an oxygen-deficient coastal ecosystem. Aquat. Microbiol. 1, 18. doi: 10.3389/fmars.2014.00018
Muyzer, G., and Stams, A. J. M. (2008). The ecology and biotechnology of sulphate-reducing bacteria. Nat. Rev. Microbiol. 6, 441–454. doi: 10.1038/nrmicro1892
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Overmann, J. (2001). Diversity and ecology of phototrophic sulfur bacteria. Microbiol. Today 28, 116–119.
Paulmier, A., and Ruiz-Pino, D. (2009). Oxygen minimum zones in the modern ocean. Prog. Oceanogr. 80, 113–128. doi: 10.1016/j.pocean.2008.08.001
Schunck, H., Lavik, G., Desai, D. K., Großkopf, T., Kalvelage, T., Löscher, C. R., et al. (2013). Giant hydrogen sulfide plume in the oxygen minimum zone off Peru supports chemolithoautotrophy. PLoS ONE 8:e68661. doi: 10.1371/journal.pone.0068661
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Shanks, A. L., and Reeder, M. L. (1993). Reducing microzones and sulfide production in marine snow. Mar. Ecol. Prog. Ser. 96, 43–47. doi: 10.3354/meps096043
Sheik, C. S., Jain, S., and Dick, G. J. (2014). Metabolic flexibility of enigmatic SAR324 revealed through metagenomics and metatranscriptomics. Environ. Microbiol. 16, 304–317. doi: 10.1111/1462-2920.12165
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Stewart, F. J., Ulloa, O., and DeLong, E. F. (2012). Microbial metatranscriptomics in a permanent marine oxygen minimum zone. Environ. Microbiol. 14, 23–40. doi: 10.1111/j.1462-2920.2010.02400.x
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Stramma, L., Johnson, G. C., Sprintall, J., and Mohrholz, V. (2008). Expanding oxygen minimum zones in the tropical oceans. Science 320, 655–658. doi: 10.1126/science.1153847
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Swan, B. K., Martinze-Garcia, M., Preston, C. M., Woyke, A. T., Lamy, D., Reinthaler, T., et al. (2011). Potential for chemolithoautotrophy among ubiquitous bacteria lineages in the dark ocean. Science 333, 1296–1300. doi: 10.1126/science.1203690
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Ulloa, O., Canfield, D. E., DeLong, E. F., Letelier, R. M., and Stewart, F. J. (2012). Microbial oceanography of anoxic oxygen minimum zones. Proc. Natl. Acad. Sci. U.S.A. 109, 15996–16003. doi: 10.1073/pnas.1205009109
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Walsh, D. A., Zaikova, E., Howes, C. G., Sung, Y. C., Wright, J. J., Tringe, S. G., et al. (2009). Metagenome of a versatile chemolithoautotroph from expanding oceanic dead zones. Science 326, 578–582. doi: 10.1126/science.1175309
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Wright, J. J., Knowar, K. M., and Hallam, S. J. (2012). Microbial ecology of expanding oxygen minimum zones. Nat. Rev. Microbiol. 10, 381–394. doi: 10.1038/nrmicro2778
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Wright, J. J., Mewis, K., Hanson, N. W., Konwar, K. M., Maas, K. R., and Hallam, S. J. (2014). Genomic properties of Marine Group A bacteria indicate a role in the marine sulfur cycle. ISME J. 8, 455–468. doi: 10.1038/ismej.2013.152
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Zaikova, E., Walsh, D. A., Stilwell, C. P., Mohn, W. W., Tortell, P. D., and Hallam, S. J. (2010). Microbial community dynamics in a seasonally anoxic fjord: Saanich Inlet, British Columbia. Environ. Microbiol. 12, 172–191. doi: 10.1111/j.1462-2920.2009.02058.x
PubMed Abstract | Full Text | CrossRef Full Text | Google Scholar
Keywords: sulfate reduction, sulfur oxidation, sulfur cyling, transcriptomics, oxygen minimum zone
Citation: Carolan MT, Smith JM and Beman JM (2015) Transcriptomic evidence for microbial sulfur cycling in the eastern tropical North Pacific oxygen minimum zone. Front. Microbiol. 6:334. doi: 10.3389/fmicb.2015.00334
Received: 06 February 2015; Accepted: 03 April 2015;
Published online: 11 May 2015
Edited by:
Hongyue Dang, Xiamen University, ChinaReviewed by:
Xiang Xiao, Shanghai JiaoTong University, ChinaFrank Stewart, Georgia Institute of Technology, USA
Copyright © 2015 Carolan, Smith and Beman. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: J. M. Beman, Life and Environmental Sciences and Sierra Nevada Research Institute, University of California at Merced, 5200 Lake Road, Merced, CA 95343, USAam1iZW1hbkBnbWFpbC5jb20=
 Molly T. Carolan
Molly T. Carolan JasonM. Smith
JasonM. Smith J. M. Beman
J. M. Beman