- 1MOA Laboratory for Risk Assessment of Quality and Safety of Livestock and Poultry Products, Huazhong Agricultural University, Wuhan, China
- 2Hubei Collaborative Innovation Center for Animal Nutrition and Feed Safety, Huazhong Agricultural University, Wuhan, China
- 3National Reference Laboratory of Veterinary Drug Residues and MOA Key Laboratory for Detection of Veterinary Drug Residues, Huazhong Agricultural University, Wuhan, China
Salmonella spp. can indirectly infect humans via transfer from animals and animal-derived food products, and thereby cause potentially fatal diseases. Therefore, gaining an understanding of Salmonella infection in farm animals is increasingly important. The aim of this study was to identify the distribution of serotypes in Salmonella samples isolated from chickens (n = 837), pigs (n = 930), and dairy cows (n = 418) in central China (Henan, Hubei, and Hunan provinces) in 2010–2011, and investigate the susceptibility of strains to antimicrobial agents. Salmonella isolates were identified by PCR amplification of the invA gene, serotypes were determined by using a slide agglutination test for O and H antigens, and susceptibility to 24 antimicrobials was tested using the agar dilution method. In total, 248 Salmonella strains were identified: 105, 105, and 38 from chickens, dairy cows, and pigs, respectively. Additionally, 209 strains were identified in diseased pigs from the Huazhong Agricultural University veterinary hospital. Among these 457 strains, the dominant serotypes were Typhimurium in serogroup B, IIIb in serogroup C, and Enteritidis in serogroup D. In antimicrobial susceptibility tests, 41.14% of Salmonella spp. were susceptible to all antimicrobial agents, 48.14% were resistant to at least one, and 34.72% were resistant to more than three classes. Strains were highly resistant to sulfamethoxazole-trimethoprim (39.61%), nalidixic acid (39.17%), doxycycline (28.22%), and tetracycline (27.58%). Resistance to cephalosporins and fluoroquinolones ranged from 5.25 to 7.44% and 19.04 to 24.51%, respectively. Among penicillin-resistant and cephalosporin-resistant strains, 25 isolates produced extended-spectrum β-lactamases (ESBLs). The multidrug-resistant and ESBL-producing Salmonella strains identified in healthy animals here will present a challenge for veterinary medicine and farm animal husbandry, and could also pose a threat to public health. The level of antibiotic resistance observed in this study further highlights the need for careful and selective use of antibiotics.
Introduction
Salmonella spp. are a common source of foodborne diseases that cause morbidity and mortality worldwide (Chiu et al., 2010). Treating Salmonella infection in humans is expensive; for example, in the USA, it causes illness in ~1.2 million patients annually, resulting in estimated medical costs of $365 million (Centers for Disease Control and Prevention, 2014). In the European Union (EU), salmonellosis is the second most commonly reported gastrointestinal infection, with a confirmed case rate of 20.4 cases per 100,000 individuals in 2011 (European Centre for Disease Prevention and Control, 2013). In China, Salmonella causes an estimated 22.2% of foodborne diseases (Wang et al., 2007), and salmonellosis ranks fourth among the most prevalent foodborne diseases caused by microbial agents (Zhu et al., 2012). Many Salmonella serotypes exist, with >2600 serovars classified based on the reactivity of antisera to O and H antigens (Stevens et al., 2009), and ~292 identified in China between the 1980s and the end of 2000 (Yang, 2010). Salmonella also affects farm animals, and infections on farms can cause substantial economic damage in relation to, for example, loss of poultry stocks and costly animal husbandry. Numerous serotypes of isolated Salmonella have been found to overlap between farm animals and humans (Alcaine et al., 2006). Indeed, Salmonella not only directly infects humans but also causes indirect infections via transfer from animals and animal-derived food products such as pork and milk.
The use of antimicrobials is important for the control and treatment of Salmonella. However, since the early 1990s, antimicrobial- and multidrug-resistant Salmonella strains have emerged, leading to treatment failure. Researchers have reported a link between the use of antimicrobials in food animals and the emergence of antimicrobial resistance in pathogenic bacteria (Gong et al., 2013). Multidrug-resistant bacteria pose a severe threat to public health, particularly those that are resistant to β-lactams and fluoroquinolones (Lai et al., 2014). The increasing number of multidrug-resistant Salmonella strains is a global concern, with some countries and international organizations creating surveillance systems which include collaboration between human health, veterinary, and food-related sectors to monitor the spread of these and other foodborne bacteria. Examples include the Danish Integrated Antimicrobial Resistance Monitoring and Research Program, the European Food Safety Authority, the National Antimicrobial Resistance Monitoring System in the USA, and the Global Foodborne Infections Network run by the World Health Organization. These surveillance systems are also employed to monitor antimicrobial resistance, antimicrobial consumption in livestock, and serotype distribution, and data describing the current trend of increasing resistance to multiple drugs has been made available (European Centre for Disease Prevention and Control, 2013). In contrast, surveillance reports are unavailable in China; however, the National Monitoring Network for Bacteria of Animal Origin was launched as a surveillance system in 2008. Travel, migration, and the distribution of food between countries can also contribute to the spread of foodborne diseases and multidrug-resistant bacteria. Therefore, monitoring the distribution of Salmonella serotypes and levels of antibiotic resistance in animals and animal-food products is also important for maintaining safe travel and the commercial trade in food animals (Lai et al., 2014; Russell et al., 2014).
In central China, Henan, Hubei, and Hunan provinces are important residential and trade centers. These three areas are also the main producers of animal-derived foods in China; in 2012, their output of meat, eggs, and milk accounted for 19.14%, 19.45%, and 9.17% of total Chinese production, respectively (China Agriculture Statistical Report, 2012). Previous studies documented a phenotype for Salmonella serovars that was discovered in chickens and pigs, and in animal-derived foods, in China (Gong et al., 2013; Li et al., 2013; Lai et al., 2014; Wang et al., 2014). However, specific knowledge of the Salmonella serovars that exist in dairy cows and other farm animals in central China is currently lacking. Therefore, the aim of this study was to evaluate the prevalence of Salmonella in chickens, pigs, and dairy cows on farms in central China. Additionally, the diversity of Salmonella serovars on these farms was identified, and their susceptibility and resistance to antimicrobial agents was investigated. A diverse range of serotypes was observed in healthy and diseased farm animals, and several multidrug-resistant and extended-spectrum β-lactamase (ESBL)-producing Salmonella strains were identified.
Materials and Methods
Samples and Salmonella Isolation
Cloacal and anal swabs were collected from healthy animals on farms in Henan, Hubei, and Hunan provinces between March 2010 and July 2011. Each sampling site was visited only once. In total, 2185 samples were collected in a random manner from chickens (n = 837), pigs (n = 930), and dairy cows (n = 418). Farms were chosen based on their scale with the following requirements: for pigs, annual sales were >10,000 heads; for chickens, the breeding stock was >100,000 heads; and for dairy cows, the breeding stock was >1000 heads. The owners of each farm gave permission for swab samples to be to collected. The animals from which samples were extracted remained alive, did not undergo any surgery, and were not administered any drugs. Therefore, ethical approval was not required for the study because the sampling process did not harm the animals; however, their distress was considered and minimized at all times. All collected samples were stored at 4 °C and cultured at least 24 h before the isolation experiments were conducted.
The procedure for culture and isolation was based on standard laboratory protocol (WHO Global Foodborne Infections Network, 2010). Briefly, the samples were mixed with 5 mL of 0.85% saline solution for 30 min, and 1 mL of this mixture solution was added to buffered peptone water (BPW; Hopebiol, Qingdao, China) at a volume ratio of 1:10. The resultant mixture was incubated for 10 h at 37 °C for pre-enrichment. Approximately 500 μL of this BPW mixture was then added to 5 mL selenite cystine broth (Hopebiol, Qingdao, China) and incubated at 37 °C for 20 h. The broth was streaked onto CHROMagar Salmonella (CHROMagar, France) and incubated at 37 °C for 24–48 h. Potential Salmonella colonies were transferred into Luria–Bertani agar for purification and enrichment, and then incubated for 20 h at 37 °C to facilitate identification.
Additionally, 209 strains of Salmonella were isolated from samples collected from diseased pigs in the Huazhong Agricultural University veterinary hospital (HAUvh) between 2008 and 2010.
Salmonella Identification
Biochemical testing and the invA gene were used to confirm the identity of isolates with typical Salmonella phenotypes. Biochemical testing was performed using a biochemical tube (Hangzhou Microbe Reagent Co., Ltd., China), and the results were interpreted based on Bergey's Manual of Systematic Bacteriology (Garrity et al., 2004). The invA gene was amplified by polymerase chain reaction (PCR) (Malorny et al., 2003). Positive results were randomly selected for sequencing. The obtained sequences were compared with the same gene registered in GenBank by using BLAST.
Determination of Salmonella Serogroup and Serotype
The serogroup and serovars of the Salmonella isolates were determined by slide agglutination tests with O antigen and H antigen antiserums obtained from Lansheng, Lanzhou Institute of Biology, China. The results were interpreted according to the Kauffmann-White scheme (Grimont and Weill, 2007).
Antimicrobial Susceptibility Testing
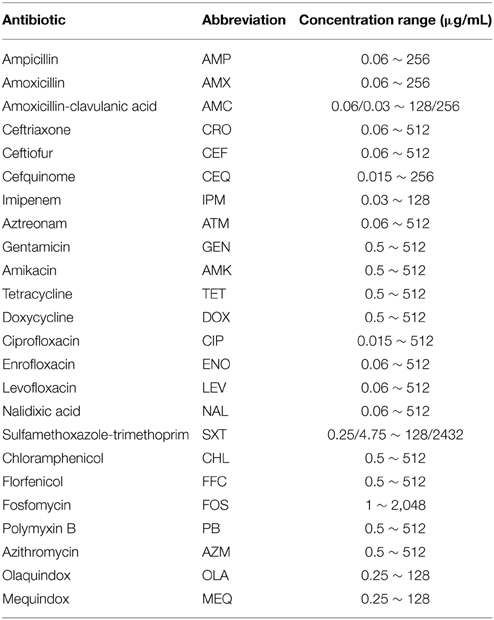
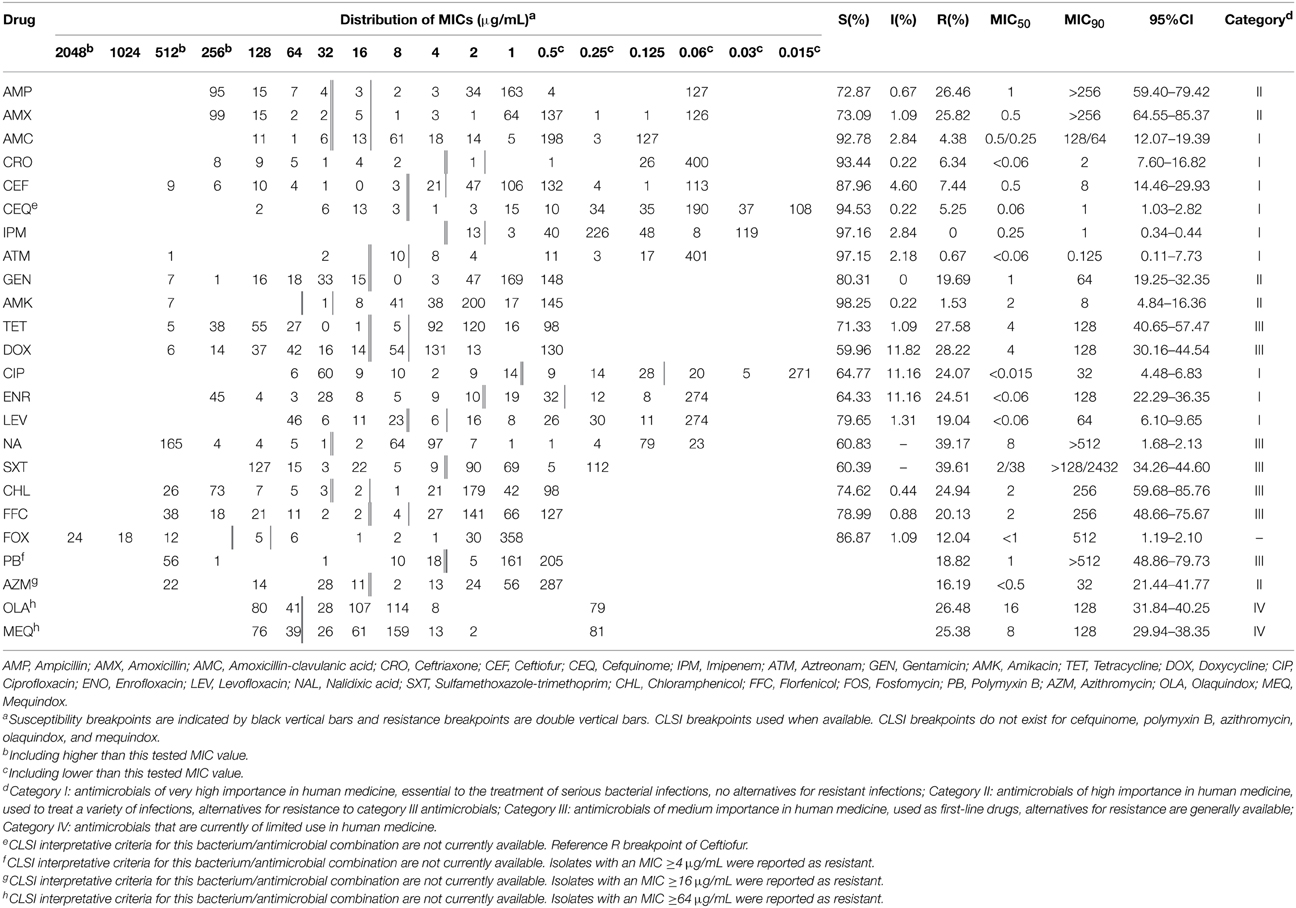
The agar dilution method with Mueller-Hinton agar (Oxoid Ltd., England) was used to test the susceptibility of Salmonella to 24 antibiotics (Table 1). These antimicrobials were classified based on their importance to human medicine (Government of Canada, 2005). Escherichia coli ATCC 25922 was used as the control microorganism. The results of the tests for ampicillin (AMP), amoxicillin (AMX), amoxicillin-clavulanic acid (AMC), ceftriaxone (CRO), imipenem (IPM), aztreonam (ATM), gentamicin (GEN), amikacin (AMK), tetracycline (TET), doxycycline (DOX), ciprofloxacin (CIP), levofloxacin (LEV), nalidixic acid (NAL), sulfamethoxazole-trimethoprim (SXT), chloramphenicol (CHL), and fosfomycin (FOS) were interpreted based on CLSI M100-S22 (Clinical and Laboratory Standards Institute, 2012), whereas those for ceftiofur (CEF), enrofloxacin (ENO), and florfenicol (FFC) were interpreted based on CLSI M31-A3 (Clinical and Laboratory Standards Institute, 2008). Interpretive CLSI criteria were not available for cefquinome (CEQ), polymyxin B (PB), azithromycin (AZM), olaquindox (OLA), and mequindox (MEQ); therefore, results with the following minimum inhibitory concentration (MIC) values were considered resistant for these antibiotics: CEQ ≥8 μg/mL (using CEF as a reference); PB ≥4 μg/mL (Kwa et al., 2007); AZM ≥16 μg/mL (Sjölund-Karlsson et al., 2011); OLA and MEQ ≥64 μg/mL (Sørensen et al., 2003).
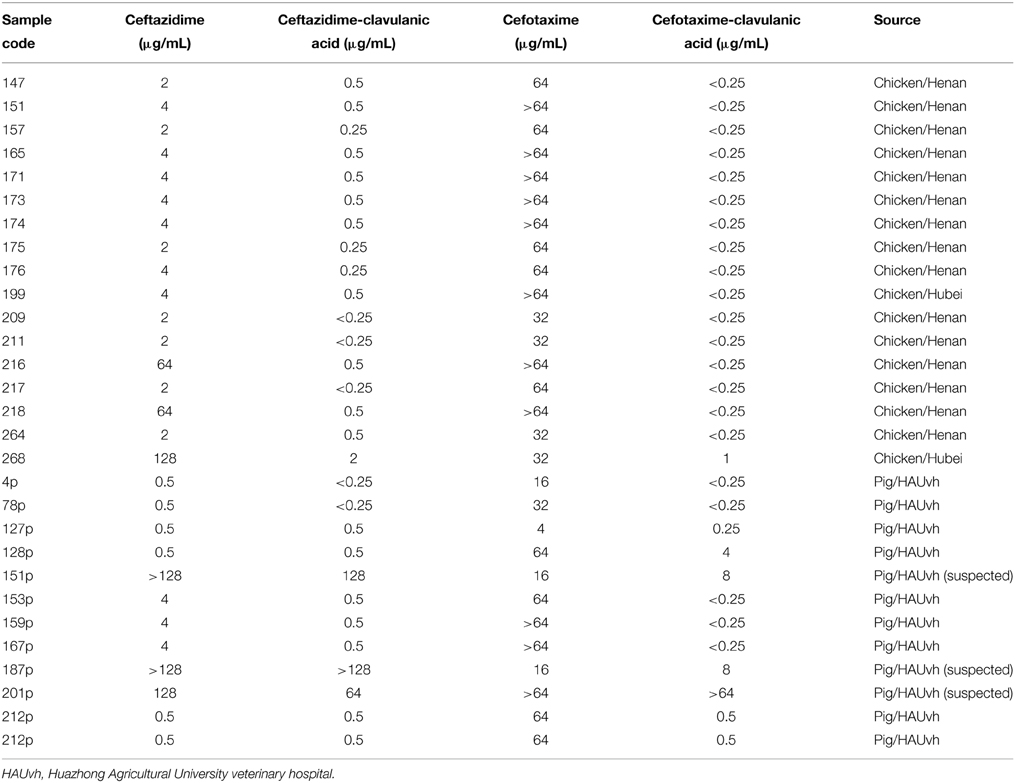
Strains that were resistant to AMP, AMX, ATM, and/or CRO were examined for ESBLs by using the MIC values of ceftazidime, ceftazime-clavulanic acid, cefotaxime, and cefotaxime-clavulanic acid. Results were interpreted based on CLSI M100-S22 (Clinical and Laboratory Standards Institute, 2012), and Klebsiella pneumoniae ATCC 700603 and E. coli ATCC 25922 were used as the control organisms.
Statistical Analysis
The isolated strains were categorized as sensitive (S), intermediary (I), or resistant (R) based on the MIC values and the CLSI interpretive criteria. MIC50 and MIC90 were calculated using previously described methods (Schwarz et al., 2010), and 95% confidence intervals were calculated using SPSS 16.0 software (IBM, USA).
Results
Salmonella Isolates and Serotypes
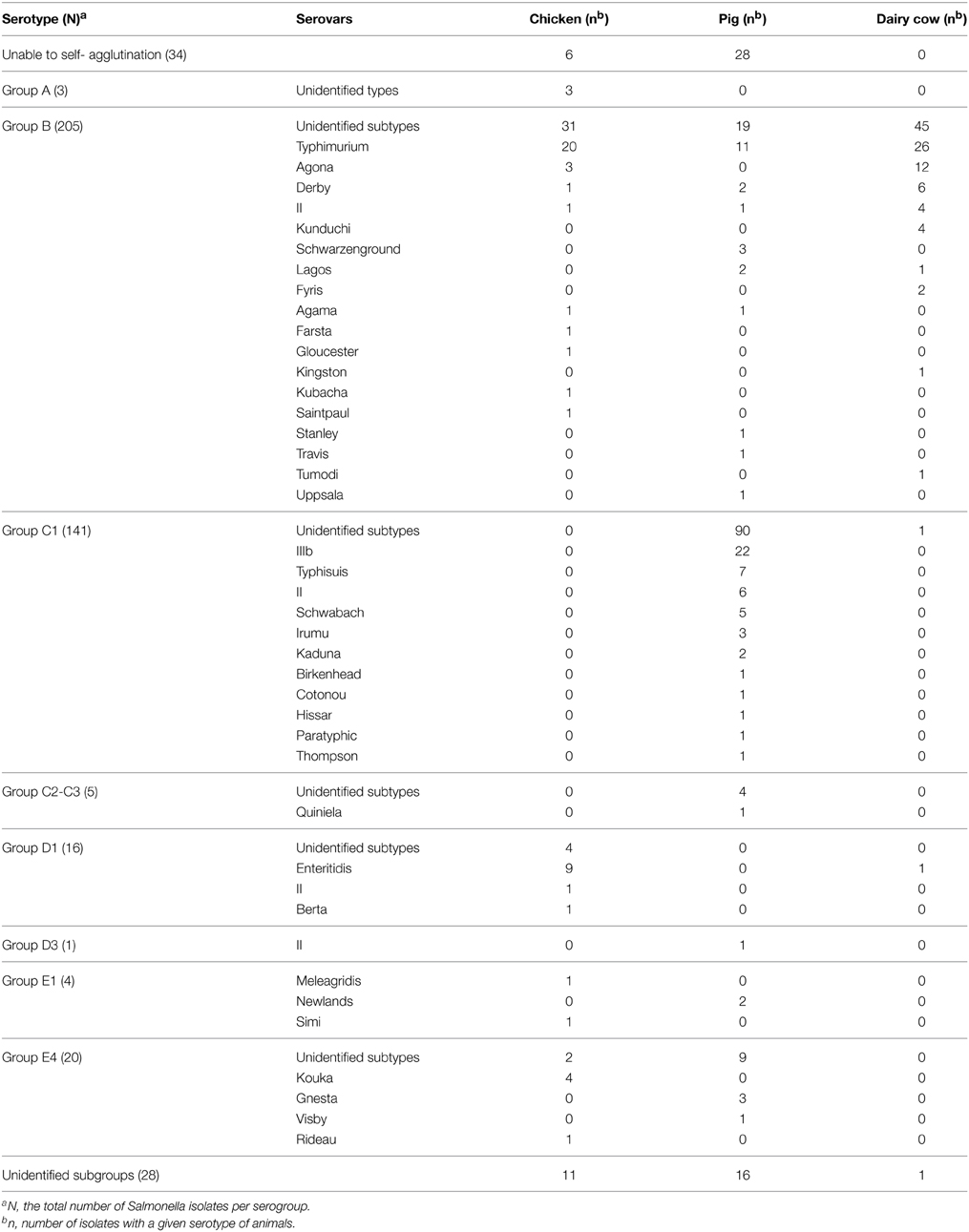
In total, 248 bacterial isolates taken from healthy chickens, pigs, and dairy cows, and 209 isolates from diseased pigs, were verified as Salmonella spp., and 457 strains identified as serotypes were tested for antimicrobial susceptibility. In all samples taken from healthy animals, the prevalence of Salmonella spp. was 11.35% (248/2185). The specific prevalence of Salmonella spp. in chickens, pigs, and dairy cows was 12.55% (105/837), 4.09% (38/930), and 25.12% (105/418), respectively.
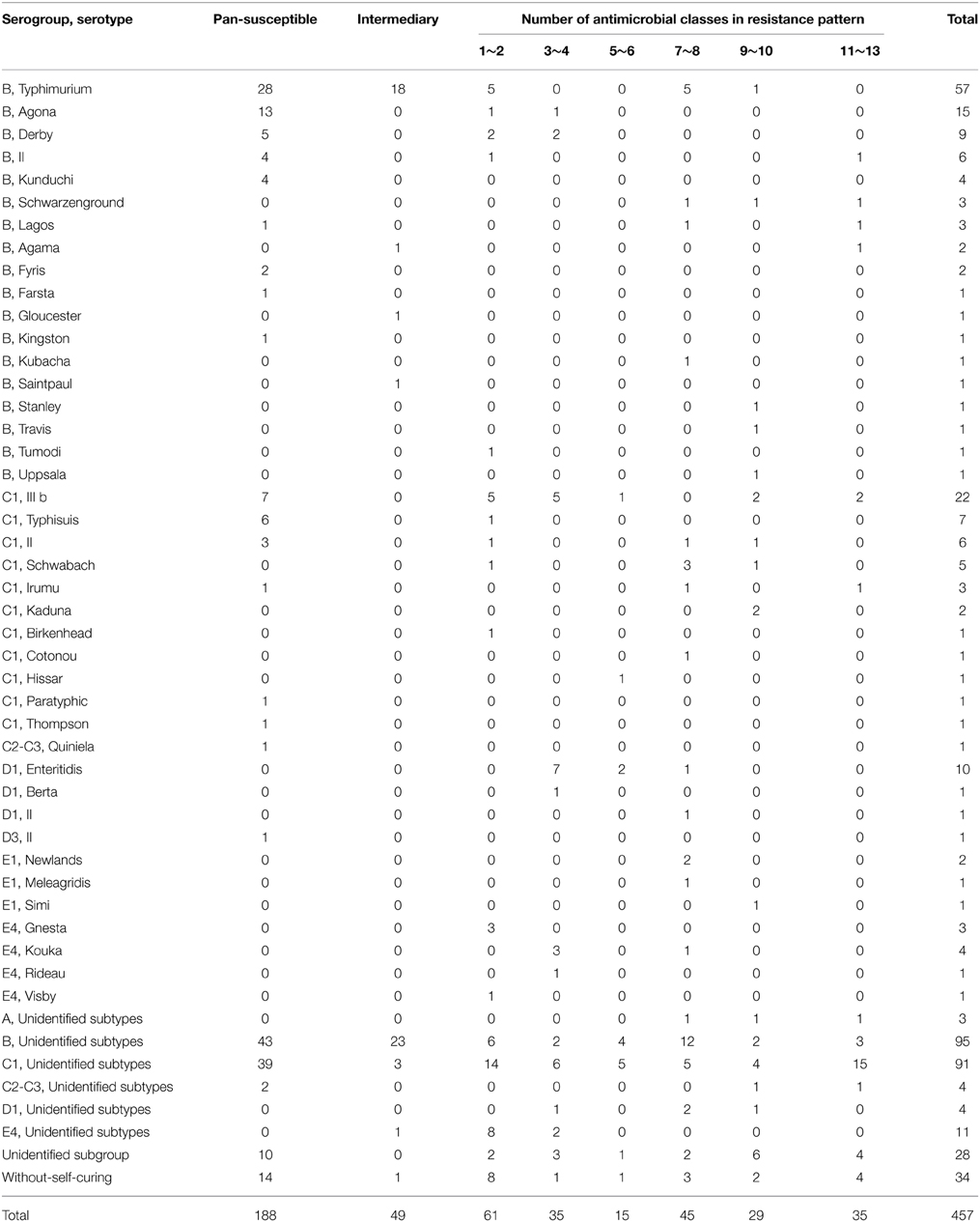
After serotyping, all isolates were divided into three groups (Table 2). Thirty-four isolates did not cluster for self-agglutination in sterile saline. Twenty-eight isolates could self-agglutination in sterile saline, but such activity was not observed with O antigen. The serogroups of the remaining 395 strains were determined. Eight distinct serogroups and 41 distinct serotypes were identified, as shown in Table 2. Group B (n = 205, 44.86%) and group C1 (n = 141, 30.85%) were identified as the dominant serogroups. Typhimurium 12.47% (n = 57) and IIIb 4.81% (n = 22) were the dominant serovars. Detailed serotypes could not be determined for 208 Salmonella isolates using the available H antigen in the different serogroups. According to the species of animal tested, the dominant serotypes were Typhimurium and Enteritidis in chickens, IIIb and Typhimurium in pigs, and Typhimurium and Agona in dairy cows (Table 2).
Antimicrobial Susceptibility Testing
Results of antimicrobial susceptibility testing are summarized in Table 3. Among the 457 isolates, 188 (41.14%) were susceptible to all tested antimicrobials and 220 (48.14%) were resistant to at least one antibiotic. All strains showed susceptibility or intermediate susceptibility to imipenem. Strains were most commonly resistant to nalidixic acid (39.17%), sulfamethoxazole-trimethoprim (39.61%), doxycycline (28.22%), and tetracycline (27.58%). They also showed resistance to all specialist drugs used only in veterinary medicine, including enrofloxacin (24.51%), florfenicol (20.13%), ceftiofur (7.44%), and cefquinome (5.25%), and to drugs used in both veterinary and human medicine, such as ciprofloxacin (24.07%), gentamicin (19.69%), ceftriaxone (6.34%), and amoxicillin/clavulanate (4.38%). Resistance to azithromycin, which is one of the macrolides often used against gram-positive bacteria, was expressed at 16.19%. Although they are rarely used in clinical applications, resistance to polymyxin B (18.82%) and fosfomycin (12.04%) was also observed.
When antibiotic resistance was analyzed according to animal species, the resistance rates of Salmonella were different for the majority of the drugs tested (Table 4). Excluding imipenem, resistance to the other 23 drugs ranged from 1.90 to 49.52% in chickens and 0.40 to 48.18% in pigs. In comparison, Salmonella from dairy cows showed resistance to 12 types of drug, and resistance rates ranged from 0.95 to 10.48%.
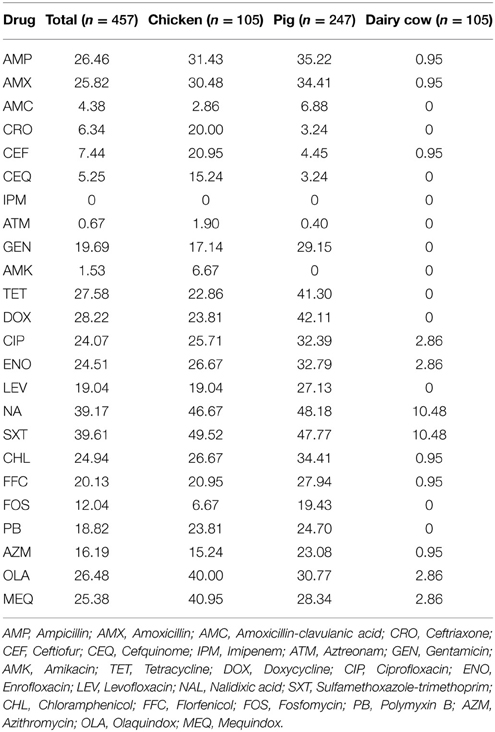
Table 4. Antibiotic resistance of Salmonella isolated from different food-producing animals unit: %.
In total, 159 strains (34.72%) exhibited varying degrees of multidrug resistance (MDR) (Table 5), defined as resistance to at least three different classes of antimicrobials. The Salmonella spp. that exhibited MDR consisted of 104 strains from pigs (including 15 and 89 strains from healthy and diseased pigs, respectively), 51 strains from healthy chickens, and 4 strains from healthy dairy cows. MDR isolates showed diverse resistance to different classes of antibiotics, but were most frequently resistant to penicillins, fluoroquinolones, and quinolones (each occurring 11 times). β-lactam/β-lactamase inhibitor combinations appeared with the lowest frequency (five times) in Salmonella strains with MDR.
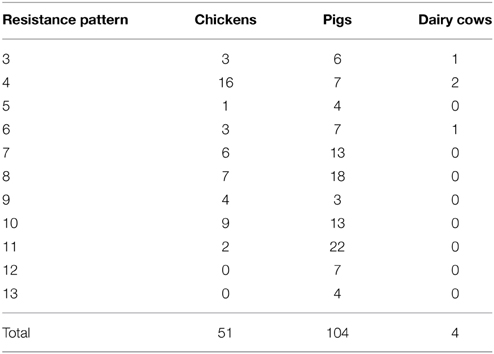
Table 5. The number of multidrug-resistance Salmonella strains identified in sampled animals unit: strain.
Based on analysis of MIC values and CLSI, 25 of the 457 isolates (5.47%) were confirmed as ESBL producers, with three suspected strains (Table 6). The ESBL-producing strains were obtained from different animals and areas as follows: two strains from chickens in Hubei, 15 strains from chickens in Henan, and eight strains from diseased pigs. Because the differences in MIC values between CAZ, CAZ/C and/or CTX, and CTX/C were not =3-fold based on CLSI, three strains from diseased pigs were suspected to be ESBL-producing strains.
The Salmonella serotypes of isolates differed in terms of their sensitivity to drugs (Table 7). Among all drug-sensitive isolates (n = 188), 80 isolates were identified in 17 specific serotypes. Typhimurium (n = 28) was the major serotype. Isolates of Salmonella strains with MDR showed 26 specific serotypes and the major serotypes were Enteritidis and IIIb (both n = 10). All serotypes were resistant to nalidixic acid, but differences were observed in the resistance profiles of Salmonella serotypes that exhibited MDR. The most common resistance profiles in Salmonella IIIb (n = 10) and Enteritidis isolates (n = 10) were AMP-AMX-SXT-NAL and SXT-OLA-MEQ-NAL, respectively. The resistance profile was AMP-AMX-TET-DOX-ENO-SXT-CHL-NAL in Typhimurium (n = 6), TET-DOX-CIP-ENO-SXT-CHL-FFC-NAL in Schwabach (n = 4), and PB-OLA-MEQ-NAL in Kouka (n = 4).
Discussion
The prevalence of Salmonella spp. in the center of China were in agreement with those obtained from Sichuan province farm animals (Li et al., 2013), but differed from other areas. The results were higher than in EU (European Food Safety Authority, 2014), where chicken 2.7%, pig 6.3%, cattle 2.4%, but were lower than in USA (National Antimicrobial Resistance Monitoring System, 2011), where chicken 52.56% ~ 47.95%, pig 10.34% ~ 8.79%, cattle 23.02% ~ 33.20%. Differences in isolation rates can be interpreted based on differences in region, sample types, collection seasons, culture methods, isolation methodologies, culture media, and local environmental conditions. For example, salmonellosis cases increased over the summer months, peaking in August and September, began decreasing thereafter (European Centre for Disease Prevention and Control, 2013). Therefore, a global perspective should ideally be adopted during sample collection, isolate separation, and, in particular, long-term monitoring.
The prevalence of Salmonella serotypes differed among animals and regions. Serogroup B and C1 were dominant, this result was consistent with in USA (Antunes et al., 2011). Serotype differ from other study. The dominant in chicken, pig, dairy cattle were Typhimurium and Enteritidis, IIIb and Typhimurium, Typhimurium and Agona in this study, respectively. The dominant serotype from chicken were Derby and Typhimurium in Sichuan province (Li et al., 2013), from pig were Enteritidis Indianain in Shandong province (Lai et al., 2014). The dominant from chicken, pig, cattle were Kentucky and Enteritidis, Adelaide and Johannesburg, Montevideo and Dublin in USA (National Antimicrobial Resistance Monitoring System, 2011); and from chicken, pig, cattle were Enteritidis and Infantis, Typhimurium and Typhimurium (monophasic), Typhimurium and Dublin in EU (European Food Safety Authority, 2014). This difference suggests serogroups and serotypes are varying according to geographical regions, and diversities and complexities. Certain serovars can emerge within a country or region for a certain period and then disappear with no evident cause or intervention. For instance, compared with data in 2010, the number of Enteritidis and Typhimurium decreased by 6% and 9% in 2011, respectively, (European Centre for Disease Prevention and Control, 2013); similar, compared with results in 2009, Enteritidis decreased, even disappeared from chicken and pig in 2012 from Shandong province (Lai et al., 2014).
The Salmonella serotypes isolated from farm animals overlapped with those that cause illnesses in humans, further highlighting the fact that Salmonella could be transmitted from animals to humans via the food chain (Alcaine et al., 2006; de Jong et al., 2009), such as Typhimurium and Enteritidis had identified from human in Henan and Hubei provinces (Cui et al., 2009; Xia et al., 2009). Some researchers have suggested that the differences in the dominant serotypes between animals and humans might be due to variations in their pathogenicity and corresponding resistance profiles (Volf et al., 2010), the strain containing Typhimurium and Enteritidis could acquired resistance to a large number of different antimicrobial compounds. At least two different serotypes existed from multiple-two strains to multiple-eleven strains, especially Enteritidis and Typhimurium, which were similar with other study from animals (Li et al., 2013; European Food Safety Authority, 2014; Lai et al., 2014), also with human (Cui et al., 2009; Xia et al., 2009). This may be related to the presence of the genetic structure known as Salmonella genomic island 1 (SGI1) and resistance genes (European Food Safety Authority, 2014). Therefore, monitoring of Salmonella should preferably focus more strongly on serovars that could, for example, be involved in a large outbreak.
Based on our MIC analysis, antibiotic-resistant bacteria, including multidrug-resistant bacteria, appeared in both healthy and diseased farm animals. These results were similar with other study (Gong et al., 2013; Li et al., 2013; Lai et al., 2014). One potential explanation is the use of antibiotics during breeding. Disease control and prevention in China during the breeding process mainly depends on the use of antibiotics (Gong et al., 2013). This arguably irrational use of antibiotics has contributed to the emergence of multidrug-resistant bacteria under selective antimicrobial pressure. Huazhong area (Henan, Hubei, and Hunan provinces) is one of the more concentrated areas of farm animals (China Agriculture Statistical Report, 2012), the demands and uses of drugs are huge, then inevitable there would be unreasonable use of drugs. These drugs, or the raw materials of the drugs, are inexpensive and easy to obtain; they have been widely used on animals in China. However, some steps have been taken to prevent the emergence of drug-resistant bacteria and promote food safety and public health with the Ministry of Agriculture of the People's Republic of China having formulated plans to ban or reduce the use of specific antimicrobials (Ministry of Agriculture of the People's Republic of China, 2002).
After the therapeutic effects of antimicrobials were confirmed in humans in the mid-1940s, they were soon introduced to veterinary medicine (McEwen, 2006). Even though some drugs were exclusively designed for veterinary use, the compounds administered are identical or very similar to those used in human medicine because they belong to the same antimicrobial classes (Heuer et al., 2009) such as β-lactams, cephalosporins, aminoglycosides, macrolides, tetracycline, sulfonamides, fluoroquinolones. Most of the antibiotic resistance to different classes has appeared in humans, animals, and/or animal products. Ceftriaxone had showed resistance rates ≤13% and 4% from imported foods and humans (Akiyama and Khan, 2012; Wong et al., 2014). Similar, gentamicin had shown resistance in animals and humans (Lai et al., 2014; Wong et al., 2014). Overlapping resistance not only can lead to treatment failure, but also have negative consequences in both humans and animals.
ESBL-producing Salmonella strains were isolated from chickens and pigs in different areas in this study. Currently, the resistance genes were still on attempting to identify and sequencing from the samples. The emergence of producing-ESBLs bacteria in animals and animal products is of particular concern to public health, it allow bacteria to become resistant to a wide variety of penicillins and cephalosporins. ESBL-producing bacteria have appeared in poultry, pig, and cattle farms (Horton et al., 2011; European Food Safety Authority, 2014). In Salmonella, acquisition of resistance genes was likely to have occurred by conjugation, usually with other Enterobacteriaceae through the transfer of plasmids (European Food Safety Authority, 2014). Bacteria that develop resistance via ESBLs could become a reservoir of resistance genes, which may enter the food chain (European Food Safety Authority, 2012). When antibiotic resistance occurs during infection, the remaining treatment option is usually an antibiotic from the carbapenem family. While carbapenems were previously drugs of last resort, their use is now also contributing to resistance (Centers for Disease Control and Prevention, 2014). They are used to treat highly resistant infections in humans, and are not used in food-producing animals. However, carbapenemase-producing organisms have been isolated from farm animals (Fischer et al., 2012). In China, monitoring strategies do not currently contain testing and identification of ESBL- or AmpC-producing organisms in isolates from animals and/or animal-derived food products. It suggest that this should be established within surveillance procedures to anticipate possible changes in the status of ESBL enzymes.
Salmonella isolates showed multiple-resistance in results, it suggested the emergence of co-resistance and/or cross-resistance (Cantón and Ruiz-Garbajosa, 2011). For example, amikacin had showed low levels of resistance (0.67%); this may be due to cross-resistance among aztreonam, ciprofloxacin, and β-lactams (Livermore, 2002). Co-resistance to fluoroquinolones and third- and fourth-generation cephalosporins has also been identified in Salmonella isolated from humans (Zhang et al., 2014). Considering these results, the prudent use of antibacterial agents should be strongly recommended in clinical, veterinary, and agricultural settings in order to preserve antibiotic activity and avoid the development of cross-resistance.
In previous studies, researchers have demonstrated the relationship between the increased prevalence of antimicrobial-resistant bacteria and (a) the increased use of antimicrobials in human and veterinary medicine, (b) greater movement of people and animals, and (c) increased industrialization (Cheng et al., 2012). Huazhong is central to China; this area not only meets the needs of its own people via the production of animals and food but also supplies other areas and imports animals and food from other regions. This movement is a potential contributor to the spread of antibiotic-resistant bacteria. The environment itself also contains a variety of bacteria that potentially represent an immense pool of antibiotic-resistance genes, which if transferred between bacteria could cause human and animal disease (D'Costa et al., 2006). For example, wild animals that are carriers of antimicrobial-resistant bacteria could pass on colonies of resistant strains when they encounter humans, farming areas, and/or waste (Literak et al., 2011). A limitation of our study was that the samples were only collected from farm animals. For complete consideration in a surveillance system, samples should also be collected from, for example, the farm environment and farm workers.
Despite bans and legislation, some antibiotics are still routinely fed to livestock prophylactically to increase profits and to limit potential bacterial infections in stressed and crowded livestock (Ndi and Barton, 2011). Antibiotic-resistant bacteria will inevitably follow wherever antimicrobials are used; therefore, a coordinated multi-disciplinary approach will be required to address this issue (Smith et al., 2009). Various programs around the world have been established to monitor and control antibiotic-resistant bacteria. Recently, an official document, “National Medium- and Long-Term Planning for Prevention and Control of Animal Epidemics (2012 to 2020),” (General Office of State Council of China, 2012) was issued in China. The government outlined plans to control diseases by detection and purification using a medium- or long-term program, with the aim of reducing the antimicrobials used in animals and in symptomatic treatment. In future, the safest way to prevent the problem may be reducing antimicrobial usage with strict policies and legislation to control the development and spread of resistant bacteria in animals.
Funding
This research is supported by the Ministry of Science and Technology of the People's Republic of China (2012 BAK01B02), National Natural Science Foundation of China (31101856/31302143), Grants from National Basic Research program of China (2013CB127200), National Key Technology R&D Program (2012BAK01B00), Fundamental Research Funds for the Central Universities (2662015PY035) and project supported by the morning program of Wuhan in China (2015070404010191).
Author Contributions
XK, HH, MD, YW, and ZY designed the study. XK, HH, IA, and ZL collected the antimicrobial susceptibility data. XK, HH, YW, and ZL collected the serotype data. XK, HH, MD, and ZY analyzed the data. XK, HH, IA, and YW wrote the manuscript. XK, HH, MD, and ZY revised manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Professor Aizhen Guo, who provided some of the Salmonella spp. isolates from diseased pigs.
References
Akiyama, T., and Khan, A. A. (2012). Molecular characterization of strains of fluoroquinolone-resistant Salmonella enterica serovar Schwarzengrund carrying multidrug resistance isolated from imported foods. J. Antimicrob. Chemother. 67, 101–110. doi: 10.1093/jac/dkr414
Alcaine, S. D., Soyer, Y., Wamick, L. D., Su, W. L., Sukhnanand, S., Richards, J., et al. (2006). Multilocus sequence typing supports the hypothesis that cow-and human-associated Salmonella isolates represent distinct and overlapping populations. Appl. Environ. Microbiol. 72, 7575–7585. doi: 10.1128/AEM.01174-06
Antunes, P., Mourao, J., Pestana, N., and Peixe, L. (2011). Leakage of emerging clinically relevant multidrug-resistant Salmonella clones from pig farms. J. Antimicrob. Chemother. 66, 2028–2032. doi: 10.1093/jac/dkr228
Cantón, R., and Ruiz-Garbajosa, P. (2011). Co-resistance: an opportunity for the bacteria and resistance genes. Curr. Opin. Pharmacol. 11, 477–485. doi: 10.1016/j.coph.2011.07.007
Centers for Disease Control Prevention (CDC). (2014). Antibiotic Resistance Threats in the United States, 2013. 73. Available online at: http://www.cdc.gov/drugresistance/threat-report-2013/pdf/ar-threats-2013-508.pdf
Cheng, A. C., Turnidge, J., Collignon, P., Looke, D., Barton, M., and Gottlieb, T. (2012). Control of fluoroquinolone resistance through successful regulation, Australia. Emerg. Infect. Dis. 18, 1453–1460. doi: 10.3201/eid1809.111515
China Agriculture Statistical Report. (2012). Ministry of Agriculture. Beijing: PRC. China Agriculture Press.
Chiu, L. H., Chiu, C. H., Horn, Y. M., Chiou, C. S., Lee, C. Y., Yeh, C. M., et al. (2010). Characterization of 13 multi-drug resistant Salmonella serovars from different broiler chickens associated with those of human isolates. BMC Microbiol. 10:86. doi: 10.1186/1471-2180-10-86
Clinical Laboratory Standards Institute. (2008). Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated from Animals; Approved Standard-Third Edition. Wayne, PA: CLSI (Informational Supplement, M31-A3).
Clinical Laboratory Standards Institute. (2012). Performance Standards for Antimicrobial Susceptibility Testing; Twenty-second Informational Supplement. Wayne, PA: CLSI (Twenty-Two Informational Supplement M100-S22).
Cui, S., Li, J., Sun, Z., Hu, C., Jin, S., Li, F., et al. (2009). Characterization of Salmonella enterica isolates from infants and toddlers in Wuhan, China. J. Antimicrob. Chemother. 63, 87–94. doi: 10.1093/jac/dkn452
D'Costa, V. M., McGrann, K. M., Hughes, D. W., and Wright, G. D. (2006). Sampling the antibiotic resistome. Science 311, 374–377. doi: 10.1126/science.1120800
de Jong, A., Bywater, R., Butty, P., Deroover, E., Godinho, K., Klein, U., et al. (2009). A pan-European survey of antimicrobial susceptibility towards human-use antimicrobial drugs among zoonotic and commensal enteric bacteria isolated from healthy food-producing animals. J. Antimicrob. Chemother. 63, 733–744. doi: 10.1093/jac/dkp012
European Centre for Disease Prevention Control. (2013). Annual Epidemiological Report Reporting on 2011 Surveillance Data and 2012 Epidemic Intelligence Data. 103–108. Available online at: http://www.ecdc.europa.eu/en/publications/_layouts/forms/Publication_DispForm.aspx?List=4f55ad51-4aed-4d32-b960-af70113dbb90&ID=989
European Food Safety Authority. (2012). Technical specifications on the harmonised monitoring and reporting of antimicrobial resistance in Salmonella, Campylobacter and indicator Escherichia coli and Enterococcus spp. bacteria transmitted through food. EFSA J. 10:2742. doi: 10.2903/j.efsa.2012.2742
European Food Safety Authority. (2014). The European Union summary report on antimicrobial resistance in zoonotic and indicator bacteria from humans, animals and food in 2012. EFSA J. 12:3590. doi: 10.2903/j.efsa.2014.3590
Fischer, J., Rodriguez, I., Schmoger, S., Friese, A., Roesler, U., Helmuth, R., et al. (2012). Escherichia coli producing VIM-1 carbapenemase isolated on a pig farm. J. Antimicrob. Chemother. 67, 1793–1795. doi: 10.1093/jac/dks108
Garrity, G. M., Bell, J. A., and Lilburn, T. G. (2004). Bergey's Manual of Systematic Bacteriology, 2nd Edn. New York, NY: Springer.
General Office of State Council of China. (2012). National Medium and Long-term Planning for Prevention and Control of Animal Epidemics (2012-2020), notice no 31. Available online at: http://www.gov.cn/gongbao/content/2012/content_2152427.htm
Gong, J., Xu, M., Zhu, C., Miao, J., Liu, X., Xu, B., et al. (2013). Antimicrobial resistance, presence of integrons and biofilm formation of Salmonella Pullorum isolates from eastern China (1962-2010). Avian Pathol. 42, 290–294. doi: 10.1080/03079457.2013.788129
Government of Canada. (2005). Current Thinking on Risk Management Measures to Address Antimicrobial Resistance Associated with the Use of Antimicrobial Agents in Food-producing Animals. ON: Health Canada, Veterinary Drugs Directorate. Available online at: http://www.hc-sc.gc.ca/dhp-mps/alt_formats/hpfb-dgpsa/pdf/vet/amr-ram_rep-rap_06_05-eng.pdf
Grimont, P. A. D., and Weill, F.-X. (2007). Antigenic Formulae of the Salmonella Serovars 9th Edn. Paris: WHO collaborating Centre for Reference and Research on Salmonella, Institut Pasteur.
Heuer, O. E., Kruse, H., Grave, K., Collignon, P., Karunasagar, I., and Angulo, F. J. (2009). Human health consequences of use of antimicrobial agents in aquaculture. Clin. Infect. Dis. 49, 1248–1253. doi: 10.1086/605667
Horton, R. A., Randall, L. P., Snary, E. L., Cockrem, H., Lotz, S., Wearing, H., et al. (2011). Fecal carriage and shedding density of CTX-M extended-spectrum beta-lactamase-producing Escherichia coli in cattle, chickens, and pigs: implications for environmental contamination and food production. Appl. Environ. Microbiol. 77, 3715–3719. doi: 10.1128/AEM.02831-10
Kwa, A., Kasiakou, S. K., Tam, V. H., and Falagas, M. E. (2007). Polymyxin B: similarities to and differences from colistin (polymyxin E). Expert. Rev. Anti. Infect. Ther. 5, 811–821. doi: 10.1586/14787210.5.5.811
Lai, J., Wu, C., Wu, C., Qi, J., Wang, Y., Wang, H., et al. (2014). Serotype distribution and antibiotic resistance of Salmonella in food-producing animals in Shandong province of China, 2009-2012. Int. J. Food Microbiol. 180, 30–38. doi: 10.1016/j.ijfoodmicro.2014.03.030
Li, R., Lai, J., Wang, Y., Liu, S., Li, Y., Liu, K., et al. (2013). Prevalence and characterization of Salmonella species isolated from pigs, ducks and chickens in Sichuan Province, China. Int. J. Food Microbiol. 163, 14–18. doi: 10.1016/j.ijfoodmicro.2013.01.020
Literak, I., Petro, R., Dolejska, M., Gruberova, E., Dobiasova, H., Petr, J., et al. (2011). Antimicrobial resistance in fecal Escherichia coli isolates from healthy urban children of two age groups in relation to their antibiotic therapy. Antimicrob. Agents Chemother. 55, 3005–3007. doi: 10.1128/AAC.01724-10
Livermore, D. M. (2002). Multiple mechanisms of antimicrobial resistance in Pseudomonas aeruginosa: our worst nightmare? Clin. Infect. Dis. 34, 634–640. doi: 10.1086/338782
Malorny, B., Hoorfar, J., Bunge, C., and Helmuth, R. (2003). Multicenter validation of the analytical accuracy of Salmonella PCR: towards an international standard. Appl. Environ. Microbiol. 69, 290–296. doi: 10.1128/AEM.69.1.290-296.2003
McEwen, S. A. (2006). Antibiotic use in animal agriculture: what have we learned and where are we going? Anim. Biotechnol. 17, 239–250. doi: 10.1080/10495390600957233
Ministry of Agriculture of the People's Republic of China. (2002). List of Bans for Veterinary Drugs and Other Compounds in Animal Food. Available online at: http://www.moa.gov.cn/zwllm/tzgg/gg/201104/t20110422_1976324.htm
National Antimicrobial Resistance Monitoring System. (2011). Animal Arm Annual Report. Available online at: http://ars.usda.gov/SP2UserFiles/Place/60400520/NARMS/NARMS2009/NARMSAnimalArm2009.pdf
Ndi, O., and Barton, M. (2011). “Antibiotic resistance in animals—The Australian perspective,” in Antimicrobial Resistance in the Environment, eds P. L. Keenand and M. H. M. M. Montforts (New York, NY: Wiley-Blackwell), 265–290.
Russell, S. B., Emilio, E. D., Kevin, L., Winthrop, J. A., Lapidus, R. V., and Paul, R. C. (2014). Travel-associated antimicrobial drug-resistant nontyphoidal Salmonellae, 2004-2009. Emerg. Infect. Dis. 20, 603–611. doi: 10.3201/eid2004.131063
Schwarz, S., Silley, P., Simjee, S., Woodford, N., van Duijkeren, E., Johnson, A. P., et al. (2010). Assessing the antimicrobial susceptibility of bacteria obtained from animals. Vet. Microbiol. 141, 1–4. doi: 10.1016/j.vetmic.2009.12.013
Sjölund-Karlsson, M., Joyce, K., Blickenstaff, K., Ball, T., Haro, J., Medalla, F. M., et al. (2011). Antimicrobial susceptibility to azithromycin among Salmonella enterica isolates from the United States. Antimicrob. Agents Chemother. 55, 3985–3989. doi: 10.1128/AAC.00590-11
Smith, P. R., LeBreton, A., Horsberg, T. E., and Corsin, F. (2009). “Guidelines for antimicrobial use in aquaculture,” in Guide to Antimicrobial Use in Animals, eds L. Guardabassi, L. B. Jensen, and H. Kruse (Oxford: Blackwell Publishing Ltd), 207–218.
Sørensen, A. H., Hansen, L. H., Johannesen, E., and Sørensen, S. J. (2003). Conjugative plasmid conferring resistance to olaquindox. Antimicrob. Agents Chemother. 47, 798–799. doi: 10.1128/AAC.47.2.798-799.2003
Stevens, M. P., Humphrey, T. J., and Maskell, D. J. (2009). Molecular insights into farm animal and zoonotic Salmonella infections. Philos. Trans. R. Soc. Lond B Biol. Sci. 364, 2709–2723. doi: 10.1098/rstb.2009.0094
Volf, J., Havlickova, H., Hradecka, H., Ondrackova, P., Matiasovic, J., Faldyna, M., et al. (2010). Epidemiology and interaction of Salmonella enterica serovar Derby, Infantis and Typhimurium with porcine alveolar macrophages. Vet. Microbiol. 146, 105–110. doi: 10.1016/j.vetmic.2010.04.031
Wang, S. J., Duan, H. L., Zhang, W., and Li, J. (2007). Analysis of bacterial foodborne disease outbreaks in China between 1994 and 2005. FEMS Immunol. Med. Microbiol. 51, 8–13. doi: 10.1111/j.1574-695X.2007.00305.x
Wang, Y., Chen, Q., Cui, S. H., Xu, X., Zhu, J., Luo, H., et al. (2014). Enumeration and characterization of Salmonella isolates from retail chicken carcasses in Beijing, China. Foodborne Pathog Dis. 11, 126–132. doi: 10.1089/fpd.2013.1586
WHO Global Foodborne Infections Network. (2010). Laboratory Protocol, Isolation of Salmonella spp. from Food and Animal Faeces, 5th Edn. Available online at: http://www.antimicrobialresistance.dk/232-169-215-protocols.htm
Wong, M. H. Y., Yan, M., Chan, E. W. C., Biao, K., and Chen, S. (2014). Emergence of clinical Salmonella Typhimurium with concurrent resistance to ciprofloxacin, ceftriaxone and azithromycin. Antimicrob. Agents Chemother. 58, 3752–3756. doi: 10.1128/AAC.02770-13
Xia, S., Hendriksen, R. S., Xie, Z., Huang, L., Zhang, J., Guo, W., et al. (2009). Molecular characterization and antimicrobial susceptibility of Salmonella isolates from infections in humans inHenan Province, China. J. Clin. Microbiol. 47, 401–409. doi: 10.1128/JCM.01099-08
Yang, B. W. (2010). Prevalence and Characterization of Chicken Borne Salmonella Serovars in Eight Provinces and Cities of China. Ph.D. Master's Thesis. Northwest A&F University. 5. Available online at: http://210.27.80.82/opac/item.php?marc_no=0000477458
Zhang, J., Jin, H., Hu, J., Yuan, Z., Shi, W., Ran, L., et al. (2014). Serovars and antimicrobial resistance of non-typhoidal Salmonella from human patients in Shanghai, China, 2006–2010. Epidemiol Infect. 142, 826–832. doi: 10.1017/S0950268813001659
Keywords: Salmonella, farm animals, serotype, antibiotics, antimicrobial resistance
Citation: Kuang X, Hao H, Dai M, Wang Y, Ahmad I, Liu Z and Yuan Z (2015) Serotypes and antimicrobial susceptibility of Salmonella spp. isolated from farm animals in China. Front. Microbiol. 6:602. doi: 10.3389/fmicb.2015.00602
Received: 06 March 2015; Accepted: 01 June 2015;
Published: 22 June 2015.
Edited by:
Patrícia Poeta, University of Trás-os-Montes e Alto Douro, PortugalReviewed by:
Atte Von Wright, University of Eastern Finland, FinlandCarmen Losasso, Istituto Zooprofilattico Sperimentale delle Venezie, Italy
Copyright © 2015 Kuang, Hao, Dai, Wang, Ahmad, Liu and Yuan. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Haihong Hao, National Reference Laboratory of Veterinary Drug Residues and MOA Laboratory for Risk Assessment of Quality and Safety of Livestock and Poultry Products, Huazhong Agricultural University, No. 1, Shizhishan Street, Hongshan District, 430070 Wuhan, China,aGFvaGFpaG9uZ0BtYWlsLmh6YXUuZWR1LmNu;
Zonghui Yuan, National Reference Laboratory of Veterinary Drug Residues and MOA Key Laboratory for Detection of Veterinary Drug Residues, Huazhong Agricultural University, No. 1, Shizhishan Street, Hongshan District, 430070 Wuhan, China,eXVhbjU4MDJAbWFpbC5oemF1LmVkdS5jbg==
 Xiuhua Kuang
Xiuhua Kuang Haihong Hao
Haihong Hao Menghong Dai2,3
Menghong Dai2,3 Ijaz Ahmad
Ijaz Ahmad Zonghui Yuan
Zonghui Yuan