- 1Institute for Biological Sciences, University of Rostock, Rostock, Germany
- 2Biocenter, Botanical Institute and Cluster of Excellence on Plant Sciences, University of Cologne, Cologne, Germany
- 3Department of Plant and Environmental Sciences, Copenhagen Plant Science Centre, University of Copenhagen, Taastrup, Denmark
- 4Institute of Plant Sciences, University of Graz, Graz, Austria
Trichoderma species are present in many ecosystems and some strains have the ability to reduce the severity of plant diseases by activating various defense pathways via specific biologically active signaling molecules. Hence we investigated the effects of low molecular weight volatile compounds of Trichoderma asperellum IsmT5 on Arabidopsis thaliana. During co-cultivation of T. asperellum IsmT5 without physical contact to A. thaliana we observed smaller but vital and robust plants. The exposed plants exhibit increased trichome numbers, accumulation of defense-related compounds such as H2O2, anthocyanin, camalexin, and increased expression of defense-related genes. We conclude that A. thaliana perceives the Trichoderma volatiles as stress compounds and subsequently initiates multilayered adaptations including activation of signaling cascades to withstand this environmental influence. The prominent headspace volatile of T. asperellum IsmT5 was identified to be 6-pentyl-α-pyrone (6PP), which was solely applied to A. thaliana to verify the growth and defense reactions. Most noticeable is that A. thaliana preexposed to 6PP showed significantly reduced symptoms when challenged with Botrytis cinerea and Alternaria brassicicola, indicating that defense-activated plants subsequently became more resistant to pathogen attack. Together, these results support that products that are based on Trichoderma volatiles have the potential being a useful biocontrol agent in agriculture.
Introduction
The genus Trichoderma (telemorph Hypocrea) includes cosmopolitan soil-borne species, some of them are saprophytes and are frequently isolated from soil and wood as well as plant litter (Błaszczyk et al., 2011). Other species were detected inside of root tissues of many plants as opportunistic, avirulent symbionts (Harman et al., 2004). Trichoderma longibraciatum, an example of a human pathogen, was isolated from infected tissues of immunocompromised individuals, who suffered opportunistic infections (Kuhls et al., 1999). Together, this reflects a large distribution and a pronounced adaptability of this genus to live in different habitats. Furthermore, Trichoderma species play an important role in the health of an ecosystem (Klein and Eveleigh, 1998) and since at least the 1930s species are known and used as biocontrol agents (plant growth promoting fungi) to reduce the severity of plant diseases (Weindling, 1932) and subsequently increase yields (Harman et al., 2004; Lorito et al., 2010). These beneficial effects were related to the control of deleterious soil microflora, the degradation of toxic compounds, the direct stimulation of root development by the production of phytohormones, enhanced solubility and subsequent increase in the availability of phosphorus and several micronutrients due to the presence of Trichoderma (Altomare et al., 1999; Gravel et al., 2007; Bae et al., 2009; Contreras-Cornejo et al., 2009; Martínez-Medina et al., 2011; Vos et al., 2015). Some effective Trichoderma strains were shown to produce a variety of microbe-associated molecular patterns (MAMPs) (Vinale et al., 2012). The first recognized MAMP was identified as an ethylene induced xylanase 2 (Xyn2/Eix) which is a potent elicitor of plant defense responses in specific tobacco and tomato cultivars (Rotblat et al., 2002). Plants colonized by Trichoderma species, or treated by e.g., cellulases, 18-mer peptaibols, harzianolide, and harzianopyridone, provide resistance to a wide variety of pathogenic microorganisms (Hermosa et al., 2012). According to Harman et al. (2004) each of the above mentioned activation process begins with the colonization of the plant roots by Trichoderma spp. Trichoderma species that are able to establish such interactions, induce massive changes in their transcriptome and metabolism (Reino et al., 2008; Brotman et al., 2010) and such metabolites have been found not only to directly inhibit the growth of pathogenic microorganisms but also increase disease resistance by triggering the defense system in plants (induced systemic resistance, ISR) (overview Vos et al., 2015). Furthermore, metabolite-pretreated plants responded to a pathogen attack much faster or more intensively (Verhagen et al., 2004; Shoresh et al., 2010; Verhage et al., 2010; Hermosa et al., 2012), a mechanism known as priming (Conrath, 2011).
The application of compounds originating from biological sources (biological control agents) and the development of novel sustainable crop protection strategies to reduce the usage of pesticides, bactericides and fungicides in agriculture are increasingly demanded by consumers. Trichoderma has the potential to find broad application, because it is already used due to its high mycoparasitic and antibiotic potential against different plant pathogens (review Vos et al., 2015). More than 60% of the registered biopesticides are based on Trichoderma (Verma et al., 2007).
We propose in a new and innovative hypothesis that volatile compounds emitted by Trichoderma facilitate the distribution of beneficial effects over long distances and that they may contribute to the improvement of plant growth. In the past decade progress has been made in understanding the role of microbial volatiles in multitrophic interactions and their potential functions (e.g., Kai et al., 2007, 2009; Vespermann et al., 2007; Minerdi et al., 2009; Wenke et al., 2010; Blom et al., 2011; Junker and Tholl, 2013; Naznin et al., 2013; D'Alessandro et al., 2014; Piechulla and Degenhardt, 2014). At present ca. 10,000 procaryots were identified, but only less than 500 bacterial and fungal species have been investigated regarding their potential to emit VOCs (Effmert et al., 2012; Lemfack et al., 2013). VOCs belong to different chemical classes, e.g., mono- and sesquiterpenes, alcohols, ketones, lactones, esters, thioalcohols, thioesters, and cyclohexanes (Splivallo et al., 2011; Kramer and Abraham, 2012; Lemfack et al., 2013). Due to their economical importance the volatile profiles of the prominent truffles were studied in detail and it was shown that the volatiles of Tuber borchii, Tuber indicum, and Tuber melanosporum inhibit leaf growth and root development of A. thaliana (Splivallo et al., 2007). Many fungi produce 1-octeno-3-ol, which enhances plant resistance to the necrotrophic fungus Botrytis cinerea by inducing defense signaling cascades (Kishimoto et al., 2007; Contreras-Cornejo et al., 2014). Volatiles of Alternaria alternata, Penicillium charlesii, and Penicillium aurantiogriseum promote growth and starch accumulation in several plant species (Ezquer et al., 2010).
As mentioned above, Trichoderma has numerous ways of indirectly enhancing plant growth (Vos et al., 2015) however so far very limited information is available upon volatile-based interactions in the context of plant growth promotion. Trichoderma viride for example stimulated the growth of A. thaliana in the absence of direct physical contact and increased lateral root formation and established early-flowering phenotypes (Hung et al., 2013). It was also shown that volatiles of Trichoderma act antibiotically against pathogenic fungi and thereby confer plant growth promotion (Vinale et al., 2008a). These hints stimulated us to screen several Trichoderma species for VOCs-mediated effects and that subsequently one specific strain was chosen to investigate thoroughly the morphological, physiological and molecular alterations in A. thaliana upon co-cultivation with Trichoderma asperellum IsmT5. These studies included the monitoring of growth and defense reactions in the plants under normal and challenged conditions.
Material and Methods
Biological Materials
The wild type Arabidopsis thaliana (Col-0) was used in all experiments. Seeds were kindly provided by Dr. Zhonglin Mou (Microbiology Department, University of Florida, Gainesville, FL, USA). Transgenic lines were obtained from: DR5::GUS from Dr. Zsuzsanna Kolbert (Department of Plant Biology, Faculty of Science and Informatics, University of Szeged, Szeged, Hungary), PDF1.2::GUS from Dr. Anja van Dijken (Utrecht University, The Netherlands), pYUC8::GUS from Dr. Stephan Pollmann (Metabolomics Unit at the Center of Plant Biotechnology and Genomics, Madrid, Spain), and PR1::GUS from the European Arabidopsis Stock Centre (UK; http://arabidopsis.info/).
Trichoderma species used in this study were isolated from different locations in Egypt during January 2005–January 2006. T. asperellum IsmT5 was isolated from the rhizosphere of maize cultivated in Ismailia, Trichoderma harzianum was isolated from okra roots cultivated in Serabium village, and Trichoderma spp. was isolated from soil in Sinai (Wadi El Arbeen). T. asperellum IsmT5 Samuels, Lieckf. and Nirenberg was identified and deposited at The Centraalbureau voor Schimmelcultures (Applied and Industrial Mycology/Identification Service CBS-KNAW Fungal Biodiversity Centre, Utrecht, The Netherlands) under accession number CBS 137093 (Data sheets 1 and 2 in Supplementary Material).
Alternaria brassicicola was obtained from Dr. Eckehard Koch (Julius-Kühn-Institute, Braunschweig, Germany) and Botrytis cinerea from Dr. Andreas v. Tiedemann (University of Göttingen, Germany).
Trichoderma—Plant Co-cultivation
The effects of volatiles of Trichoderma sp. on plant growth were tested in a closed and open co-cultivation system (Figure S1A). A. thaliana seeds were surface sterilized (1 min 70% ethanol, 5 min 5% calcium hypochlorite, rinsed four times with sterilized distilled water) and cultivated on MS medium (Murashige and Skoog, 1962). The seeds were vernalized for 3 days at 4°C in the absence of light and then four seedlings were transferred to glass jars containing solidified MS medium in slant position for 3 days. At this time a disc (∅ 0.5 cm) of a 7 days old Trichoderma culture grown on nutrient broth agar (30 g glucose; 2 g NaNO3; 1 g KH2PO4; 1 g yeast extract; 2 g peptone; 0.5 g KCl; 0.5 g MgSO4·7H2O; 8 mg CaCl2·6H2O; 1 mg ZnSO4·7H2O; 10 mg FeSO4·7H2O per liter) (Bonnarme et al., 1997) at 20°C was introduced into a small beaker containing 20 ml broth agar. The small beaker was placed into the jar without any physical contact to the plants or MS agar. The jar was placed in 3 cm distance to the plants. The seedlings were exposed to Trichoderma volatiles for 9 days at 24°C and 84 μmolm−2s−1 of light at a 16/8 h light/dark cycle. Under these growth conditions Trichoderma did not produce spores and fungal growth was not observed in control experiments in MS agar. Fresh weight, leaf area and root length were determined four times (technical replicates). The experiments were repeated three times (biological replicates).
Analysis of Anthocyanins
The accumulation of anthocyanins in Arabidopsis seedlings was determined after 9 days of co-cultivation with T. asperellum IsmT5 applying the method of Neff and Chory (1998) with some modifications. At least two samples of 100 mg VOCs-exposed seedlings in comparison with control plants were incubated overnight in 150 μL of methanol acidified with 1% HCl (w/v). After the addition of 100 μL of distilled water, anthocyanins were separated from chlorophylls with 250 μL of chloroform. The absorbance of the aqueous phase was measured at 535 and 657 nm. The relative amount of anthocyanins per 100 mg of fresh weight was calculated by the equation A535–A657 × 100.
Analysis of Chlorophyll Content
Total chlorophylls were extracted in 1 mL of 80% aqueous acetone containing 2.5 mM sodium phosphate buffer (pH 7.8) to minimize conversion of chlorophylls into phaeophytins. The suspension was centrifuged at 4°C at 2500 rpm for 10 min. For each experiment, at least two groups of 100 mg of seedlings (after 9 days of co-cultivation) were used and fluorescence was determined using a spectrometer (Ultrospec 3000, Pharmacia). The absorbance at different wavelength (480, 646, 647, 652, 663, 664, and 750 nm) was measured. The absolute amount of chlorophyll was calculated using the extinction coefficient indicated by Porra et al. (1989). Concentration of chlorophyll, expressed as μg/mg was determined by the following equations:
Determination of Trichome Density
Trichome number was measured upon the appearance of the first true fully expanded rosette leaf produced by treated and untreated plants (after 9 days of co-cultivation). The target leaf was first removed from the plant and traced. The leaf area was measured using a leaf area meter (AM 300, ADC BioScientific, Hoddesdon Herts, UK) and the adaxial trichome number was determined under a dissecting microscope. The trichome density was calculated as trichome number per leaf area (number/cm2).
Reactive Oxygen Species (ROS)
H2O2
Hydrogen peroxide content of treated and untreated A. thaliana seedlings (after 9 days of co-cultivation) was measured spectrophotometrically after reaction with potassium iodide (KI) according to Chakrabarty et al. (2009). The reaction mixture consisted of 0.5 ml supernatant of 0.1% trichloroacetic acid (TCA) seedling extract, 0.5 ml of 100 mM K-phosphate buffer (pH 7), and 2 ml reagent (1 M KI, w/v in fresh double-distilled water). 0.5 ml of 0.1% TCA served as control. After 1 h of incubation in darkness at room temperature, the absorbance was measured at 390 nm. The amount of hydrogen peroxide was calculated using a standard curve prepared with known concentrations of H2O2.
Viability of Roots
Root activity is an indirect indicator of tissue viability, and can be determined by using 2, 3, 5-triphenyl tetrazolium chloride (TTC) (Shen et al., 1991). Viable (respiring) tissue reduces TTC to red-colored triphenyl formazan by accepting electrons from the mitochondrial electron transport chain (Comas et al., 2000). Thus, a decrease in root activity is an indication of reduced respiration and reduced viability often resulting from tissue damage. Briefly, 50 mg of freshly harvested root tissue (after 9 days of co-cultivation) was treated with 5 ml of 0.4% TTC solution (w/v) and 5 ml of 0.067 M phosphate buffer (pH 7.4). This mixture was incubated at 40°C for 3 h followed by the addition of 2 ml 2 M H2SO4. Thereafter, roots were ground in 10 ml ethyl acetate to extract red triphenyl formazan. Its concentration was measured spectrophotometrically at 485 nm and expressed as A485 g−1 h−1.
Quantification of Phytohormones and Camalexin
Determination/quantification of the phytohormones ABA, indole-3-acetic acid (IAA), JA, SA, and the phytoalexin camalexin was performed after 9 days of co-cultivation according to Großkinsky et al. (2014) with slight modifications. Briefly, volatile exposed and control seedlings were frozen and ground in liquid nitrogen; 200 mg per sample were extracted with 80% methanol and internal standards were added for the quantification of camalexin (6-fluoroindole-3-carboxyaldehyde; Sigma-Aldrich, Steinheim, Germany) and the phytohormones (deuterium-labeled hormones; Olchemim Ltd, Olomouc, Czech Republic). The extracts were directly subjected to HPLC analysis for camalexin (Ultimate 3000; Dionex, Sunnyvale, USA). For phytohormone determination, methanol extracts were passed through Chromafix C18-columns (Macherey-Nagel, Düren, Germany), completely dried (Integrated SpeedVac® Concentrator System AES1000; Savant Instruments Inc., Holbrook, USA), resuspended in 20% methanol, passed through Chromafil PES-20/25 filters (Macherey-Nagel, Düren, Germany) and subjected to UHPLC-MS/MS (Thermo Scientific; Waltham, USA) analyses.
Quantification of Glucosinolates
Freeze-dried exposed (after 9 days of co-cultivation) and control seedlings (100 mg) were transferred into 2 mL reaction tubes and lyophylised. The isolation and analysis of GSL content was performed by using the desulpho-GSL method on an ultra-performance liquid chromatography (UPLC) device (Waters, Eschborn) as described in Frerigmann et al. (2012).
GUS Assay
Transgenic Arabidopsis lines carrying a GUS reporter system were used in this study. After 9 days of co-cultivation, the expression patterns of the ß-glucuronidase activity were elucidated by histochemical staining using at least 10 exposed and control seedlings, each (Jefferson et al., 1987). In order to visualize the GUS activity, whole seedlings were incubated in a solution of 1 mM X-gluc (5-bromo-4-chloro-3-indolyl-ß-D-glucuronic acid), 0.1 M phosphate buffer (pH 7.0), 10 mM EDTA, 0.1% (v/v) Triton X-100, and 1 mM K3Fe(CN)6 overnight at 37°C. Subsequently, seedlings were incubated in buffer and fixed for at least 20 min in a mixture of 5% (v/v) formaldehyde, 5% (v/v) acetic acid, and 20% ethanol (v/v), followed by an additional incubation in 50% (v/v) ethanol. The seedlings were preserved in formaldehyde containing 80% (v/v) ethanol till microscopic examination. Images of the plants were recorded with a digital camera Ricoh Cx4 (Ricoh, Tokyo, Japan).
Head Space Collection and GC/MS Analysis of Trichoderma VOCs
In a glass Petri dish of 9 cm diameter, a disc (∅ 0.5 cm) from a 7 days old T. asperellum IsmT5 culture was inoculated on 20 ml of broth agar (2.2). The Petri dish was placed into the incubation chamber of a slightly modified airflow collection system (Figure S1B) (Kai et al., 2007). Charcoal-purified, sterile humidified air was pushed through a closed system. Volatiles present in the headspace were carried along and finally trapped in a column containing 40 mg Super Q as trapping material (Alltech Associates, Deerfield, Illinois, USA). The airstream of the pump (Gardner Denver, Puchheim, Germany) was adjusted to a constant flow of 0.6 l min−1. The volatiles were collected in 24 h intervals during the incubation period of up to 9 days. Volatiles were eluted from the trapping material with 300 μl dichloromethane. For quantitation, 10 μl of nonyl acetate (5 ng) was added as an internal standard. Samples were analyzed using the Shimadzu GC/MSQP5000 (Kyoto, Japan) equipped with a DB5-MS column (60 m × 0.25 mm × 0.25 μm; J&W Scientific, Folsom, California, USA). Splitless injection of 1 μl sample was performed at 200°C with a sampling time of 2 min using a CTC autosampler (CTC Analytics, Zwingen, Switzerland). The initial column temperature was set at 35°C, followed by a ramp of 10°C min−1 up to 280°C with a final hold for 15 min at 280°C. Helium was used as the carrier gas at a flow rate of 1.1 ml min−1. Ionization was performed at 70 eV and mass spectra were obtained using the scan modus (2 scans per second, total ion count, 40–280 m/z). Confirmation of structure assignments was done by comparison of mass spectra and retention times with those of available standards, with literature data, as well as by comparison with spectra covered by the NIST107 (version 1998) library, and by comparison of Kovats indices. Experiments were replicated at least three times.
Effect of 6PP on Arabidopsis
To investigate effects of 6PP (Sigma-Aldrich, Steinheim, Germany) on Arabidopsis, three concentrations (0.5, 1, and 2 mM) of the pure compound were applied. Briefly, 1 ml of 6PP was dissolved in sterilized distilled water and then applied to a small glass tube that was fixed in a 9 cm Petri dish distant to 3 days old seedlings. Petri dishes were sealed and incubated for 9 days at 24°C in vertical position. Fresh weight and root length were measured. The experiment was replicated four times.
The potential of 6PP in reducing disease severity of phytopathogenic fungi such as A. brassicicola and B. cinerea on A. thaliana was also tested. 2 mM 6PP (concentration equals the concentration emitted by T. asperellum IsmT5) were applied into the soil where 4 weeks old A. thaliana grew. After 24 h, four plant leaves in each glass jar were challenged with the pathogens. 5 μl of a spore suspension (5 × 106 spores/ml) of B. cinerea or A. brassiciola were spotted onto the leaves. The percentage of disease severity (lesion size) was measured 5 days post infection.
RT–PCR
Total RNA was extracted from leaves (100 mg FW) of exposed (after 9 days of co-cultivation) and control plants. After 9 days of volatile treatment with 2 mM 6PP (see Effect of 6PP on Arabidopsis) or co-cultivation with T. asperellum IsmT5 (see Trichoderma—Plant Co-cultivation) seedlings were harvested, cells were homogenized by grinding with mortar and pestle in liquid nitrogen and RNA was enriched using the Nucleospin kit (Machery-Nagel, Düren, Germany). mRNA was reverse-transcribed into cDNA using the primers (Table S1) and SuperScript reverse transcriptase (Thermo Scientific Maxima Reverse Transcriptase) according to the protocol of the manufacturer. Gene-specific primers (Table S1, obtained from Life Technologies, Carlsbad, USA) were used to amplify respective genes via PCR (Hippauf et al., 2010). The ubiquitin gene (AT4G05320) served as internal control.
Statistics
F-statistics of ANOVA was used to calculate significance in all experiments. Each F-statistic is a ratio of mean squares. The numerator is the mean square for the term. The denominator is chosen such that the expected value of the numerator mean square differs from the expected value of the denominator mean square only by the effect of interest. The effect for a random term is represented by the variance component of the term. The effect for a fixed term is represented by the sum of squares of the model components associated with that term divided by its degrees of freedom. Therefore, a high F-statistic indicates a significant effect (Moreira et al., 2013).
Results
Trichoderma Volatiles Alter Morphology and Physiology of Arabidopsis
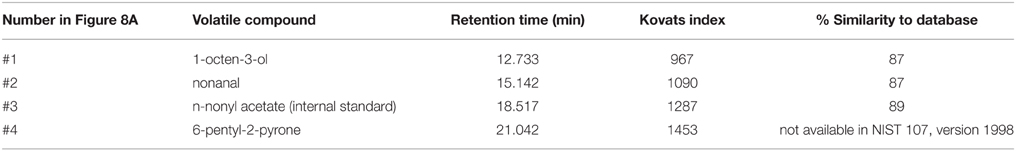
To investigate the impact of Trichoderma volatile compounds on plant growth, a simple and reliable co-cultivation system was established (Figure S1A). After 9 days of co-cultivation and concurrent exposure various parameters were investigated. First, we determined how growth and development of A. thaliana were influenced by three different Trichoderma isolates (Figure 1). The three isolates originated from different locations in Egypt. While Trichoderma sp. and T. harzianum volatiles did not influence the morphology and habitus of A. thaliana, exposure of seedlings to T. asperellum IsmT5 volatiles reduced fresh weight, root length, and leaf area by ca. 40, 60, and 50%, respectively (Figures 1A–C). Thus, T. asperellum IsmT5 volatiles were investigated in more detail.
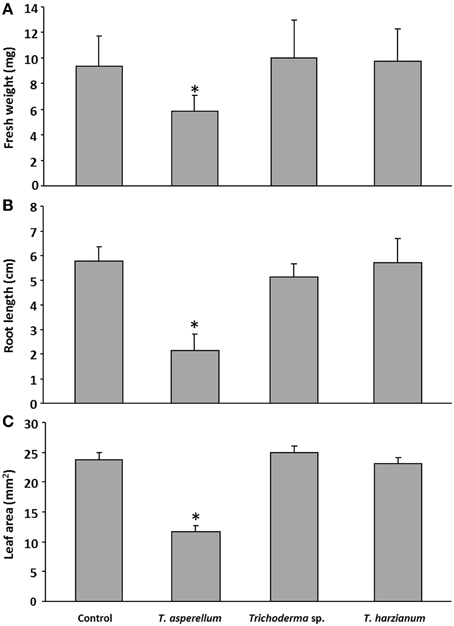
Figure 1. Effect of volatiles of different Trichoderma isolates on the growth of Arabidopsis thaliana. Seedlings and fungi were co-cultivated for 9 days at 24°C and 84 μmol m−2s−1 of light at a 16/8 h light/dark cycle. Fresh weight (A), root length (B), and leaf area (C) were measured (four technical replicates). The experiments were repeated three times (biological replicates). Non-exposed control plants were also investigated, n = 12, error bars indicate SD, *P < 0.05.
The plants treated with T. asperellum IsmT5 volatiles appeared robust and showed no wilt or other detrimental symptoms (Figures 2A,B). Interestingly, the leaves of such exposed seedlings appeared to be much darker. Spectrophotometric analyses revealed a ca. three-fold higher accumulation of anthocyanin pigments in the co-cultivated seedlings compared to controls (Figure 2C). Since anthocyanin pigmentation plays a role in plant protection, e.g., forming a photoprotective screen in vegetative tissues and functioning as antimicrobial agent and feeding deterrent in the defense responses (Winkel-Shirley, 2001; Steyn et al., 2002) we further investigated morphological and physiological parameters of plant defense in A. thaliana. Microscopic examination of the leaf surface showed that trichome density was significantly increased by 47% in the leaves exposed to T. asperellum IsmT5 volatiles (Figure 3).
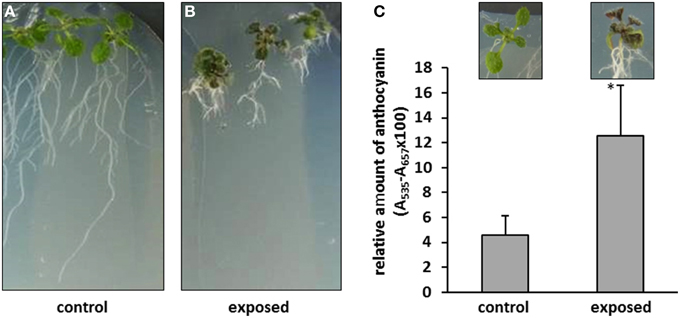
Figure 2. Phenotype and anthocyanin accumulation in Arabidopsis thaliana exposed to Trichoderma asperellum volatiles. Co-cultivation was performed for 9 days at 24°C and 84 μmol m−2s−1 of light at a 16/8 h light/dark cycle. Stunted but robust Arabidopsis seedlings were obtained after Trichoderma volatile exposure, control (A), and exposed seedlings (B). (C) Accumulation of anthocyanin in Arabidopsis seedlings co-cultivated with Trichoderma asperellum IsmT5: Anthocyanins were extracted from control (left) and exposed plants (right) and measured at 657 nm. Concentrations were calculated from three independent experiments and four technical replicates, n = 12, error bars indicate SD, *P < 0.05.
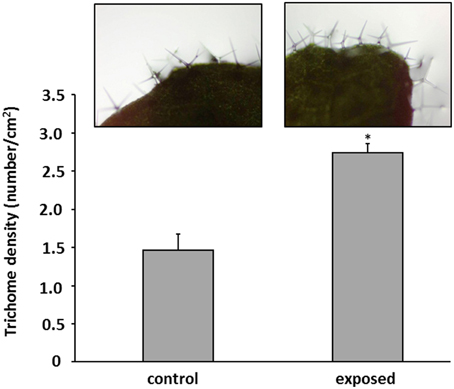
Figure 3. Trichomes on leaves of Arabidopsis thaliana exposed to Trichoderma asperellum volatiles. Co-cultivation was performed for 9 days at 24°C and 84 μmol m−2s−1 of light at a 16/8 h light/dark cycle. Trichomes were counted and leaf area was measured to determine trichome density (number per cm2) of control (left) and exposed plants (right). Density was calculated from five leaves, error bars indicate SD, *P < 0.05.
Reactive oxygen species (ROS) are small molecules such as hydrogen peroxide (H2O2), superoxide anion (O), and hydroxyl radical (•OH) and are one of the earliest signals that activate plant defense responses (Singh et al., 2010). We examined H2O2 accumulation after 9 days of co-cultivation of T. asperellum IsmT5 with A. thaliana. The level of H2O2 was increased four-fold in the exposed plants compared to control plants (Figure 4A). Camalexin is a phytoalexin, which usually accumulates after pathogen attack or after treatment with abiotic elicitors such as UV or silver nitrate (Glawischnig, 2007). Camalexin accumulation increased by 97% in A. thaliana upon fungal volatile exposure (Figure 4B, Figure S2). Furthermore, tissue viability of volatile exposed seedlings of A. thaliana was investigated by determining mitochondrial respiration activity. Figure 4C documents a ca. 40% higher respiration activity in plants treated with T. asperellum IsmT5 volatiles referring to an increased viability in exposed seedlings. In summary, A. thaliana plants exposed to Trichoderma volatiles manifest improved survival strategies and defense responses.
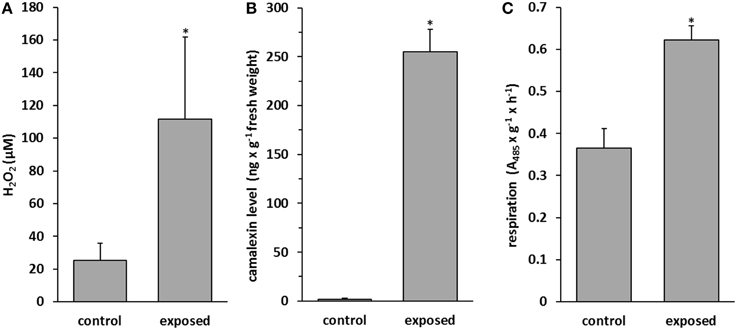
Figure 4. Determination of defense molecules in Arabidopsis thaliana exposed to Trichoderma asperellum volatiles. Co-cultivation was performed for 9 days at 24°C and 84 μmol m−2s−1 of light at a 16/8 h light/dark cycle. H2O2 (A), camalexin (B), and activity of respiration chain in roots (C) were measured in control and volatile exposed seedlings. Parameters were obtained from two independent experiments and five leaves, error bars indicate SD, *P < 0.05.
Trichoderma Volatiles Modulate Phytohormone Levels in Arabidopsis
Phytohormones are well known to regulate growth and development of plants and several plant hormones also act as central players in triggering the plant immune signaling network (Howe and Jander, 2008; Bari and Jones, 2009; Katagiri and Tsuda, 2010; Vos et al., 2015). Very little is known how the presence of volatiles is translated or mediated via phytohormone depending signaling cascades. Here we addressed the main signaling pathways. The levels of three prominent phytohormones were examined in control and T. asperellum IsmT5 volatile-exposed seedlings. The results showed that in co-cultured plants salicylic acid (SA) was increased by 61% and abscisic acid (ABA) by 40% (Figures 5A,C). No significant differences were recorded for other hormones, e.g., jasmonic acid (JA) (Figure 5B). Apparently the signal transduction chains that stimulate SA and ABA accumulation were selectively activated by the fungal volatiles leading to increased phytohormone levels.
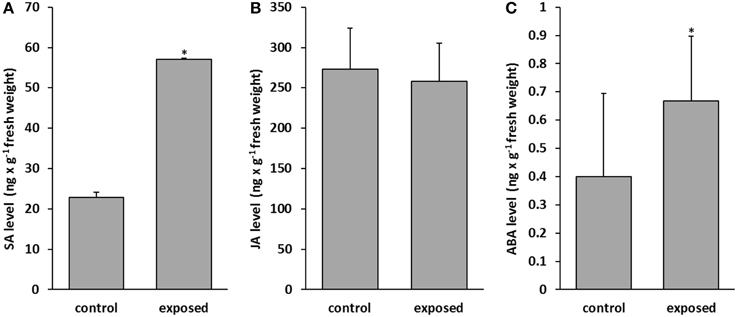
Figure 5. Determination of phytohormones in Arabidopsis thaliana exposed to Trichoderma asperellum volatiles. Co-cultivation was performed for 9 days at 24°C and 84 μmol m−2s−1 of light at a 16/8 h light/dark cycle. Salicylic acid (A), jasmonic acid (B), and abscisic acid (C) were measured in control (left column) and volatile exposed (right column) seedlings. Parameters were obtained from two independent experiments and five leaves, error bars indicate SD, *P < 0.05.
Trichoderma Volatiles Stimulate Gene Expression in Arabidopsis thaliana
In order to investigate whether T. asperellum IsmT5 volatiles cause gene activation in Arabidopsis, we used four transgenic lines with promoter-uidA chimeric gene constructs related: (i) to IAA responses (DR5::GUS and YUC8::GUS), (ii) to the transcriptional activation of indolic glucosinolates (MYB51::GUS), (iii) plant defensin (PDF1.2::GUS), and (iv) pathogenesis related protein 1 (PR1::GUS). In exposed seedlings all four promoters were activated and a distinct blue coloration compared to control plants was observed (Figure 6, Figure S3). The promoter of the DR5 gene was primarily activated at the rim of the leaves (Figure 6A), while the PR1 promoter revealed gus activation in a zone near the base of the leaf (Figure 6B). Interestingly, the MYB51 promoter was activated in the trichomes and epidermal tissue (Figure 6C). PDF 1.2::GUS and YUC8::GUS were expressed in whole leaves of the exposed seedlings (Figures S3A,B). In summary, Arabidopsis plants respond to T. asperellum IsmT5 volatiles by activating genes that are involved in phytohormone biosynthesis and plant defense.
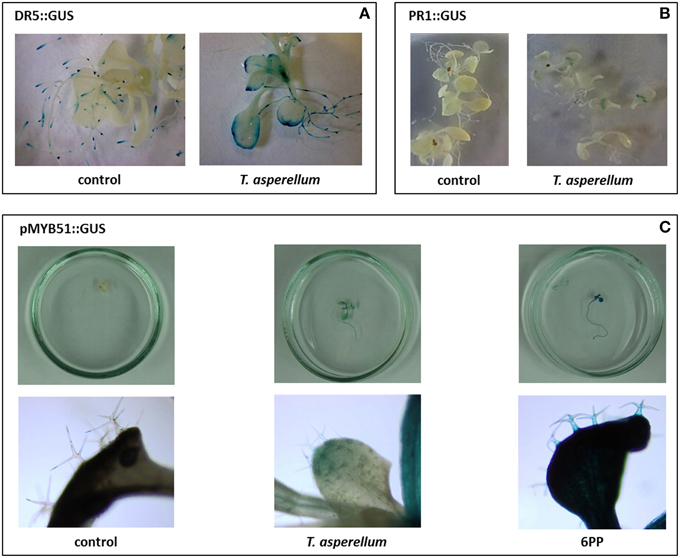
Figure 6. Expression of genes in leaves of transgenic Arabidopsis thaliana lines exposed to Trichoderma asperellum volatiles. Transgenic cell lines equipped with promoter gus constructs were co-cultivated with Trichoderma asperellum IsmT5. Promoters originated from the DR5 (indole biosynthesis) (A) and PR1 (pathogenesis related protein 1) (B), and MYB51 (transcription factor involved in trichome production) (C). Co-cultivation was performed for 9 days at 24°C and 84 μmol m−2s−1 of light at a 16/8 h light/dark cycle. Glucuronidase assay was performed in control and volatile exposed seedlings. Blue color indicates the expression of the uid gene in the tissue.
Analysis of Volatile Profiles of Trichoderma Species
The results described above demonstrate that volatiles released by T. asperellum IsmT5 caused changes at the morphological, physiological, and transcriptional level of A. thaliana. Since the volatile profile of T. asperellum IsmT5 was so far unknown, we collected and analyzed headspace volatiles (Figure S1B). In the GC chromatogram three peaks could be identified (Figure S4A). The contribution of the peak area of peak number #4 reflects ca. 90% of the total volatile emission. By comparison of mass spectra, retention times, and Kovats indices, the compound of peak #1 was tentatively identified as 1-octen-3-ol, #2 as nonanal and #4 as 6PP (Table 1). Because of its abundance, we focused our future work on compound #4. Its chemical identity was verified by comparison with the commercially available 6PP (Figures S4C,D). The dynamics of volatile emission was recorded in 24 h intervals throughout of 10 days (Figure S5). A maximum of 6PP accumulation (450 ng/μl = 2.7 mM) was reached at day 8 of cultivation.
Effects of 6-pentyl-α-pyrone on Arabidopsis thaliana
After identification of the major volatile compound released by T. asperellum IsmT5, we determined which concentration of 6PP caused morphological alterations in A. thaliana. Two milliliters of 0.5 mM, 1 mM, and 2 mM were placed in a vial next to Arabidospsis seedlings (Figure 7A). Exposure to 2 mM 6PP revealed ca. 50% reduction of fresh weight and root length (Figures 7B,C, respectively). Surprisingly, four aliphatic and three indole glucosinolates were also reduced upon 6PP and Trichoderma volatile application (Figure 7D; Figures S6A,B). To our knowledge it was shown for the first time that a volatile compound influenced glucosinolate levels in plants.
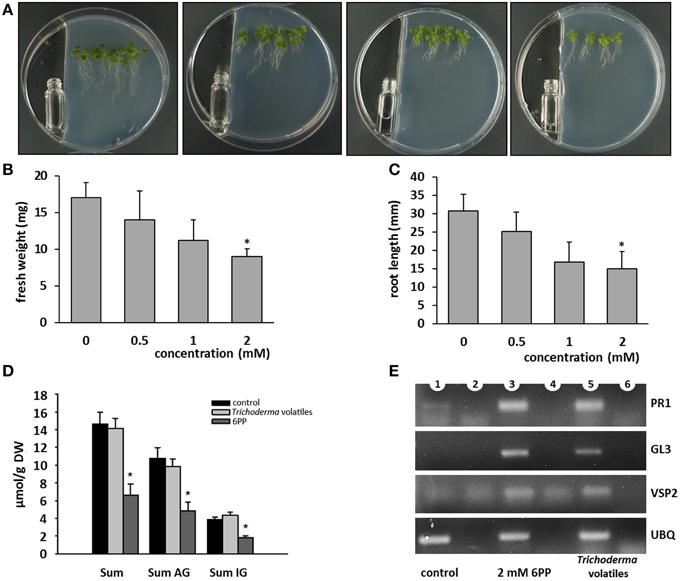
Figure 7. Effects of 6-pentyl-α-pyrone (6PP) on the growth, glucosinolate level, and defense gene expression of Arabidopsis thaliana. One ml of 6PP of different concentrations were filled into sterile glass vials and positioned next to 3 days old A. thaliana seedlings growing on MS medium (A). After 9 days fresh weight (B) and root length (C) were recorded. The experiment was repeated for three times and four seedlings were placed on the agar, n = 3, error bars indicate SD, *P < 0.05. (D): 3 days old A. thaliana seedlings were co-cultivated with T. asperellum IsmT5 (see Trichoderma—Plant Co-cultivation) or exposed to 6PP (see Effect of 6PP on Arabidopsis). After 9 days of co-cultivation or after 9 days of 6PP application whole seedlings were harvested and glucosinolates were extracted and analyzed by HPLC (see Quantification of Glucosinolates). AG, aliphatic glucosinolates; IG, indolic glucosinolates. n = 3, error bars indicate SD, *P < 0.05. (E): Via RT-PCR the expression of defense genes were analyzed: PR1, pathogenesis related protein 1; GL3, transcription factor GLABRA 3; VSP2, vegetative storage protein. Expression of ubiquitin (UBQ) was used for standardization. Controls in lane 2, 4, 6: RT-PCR without polymerase.
We furthermore investigated the effects of T. asperellum IsmT5 volatiles and 6PP on the expression (RT-PCR) of defense related genes in exposed Arabidopsis seedlings. Gene expression of the SA-induced pathogen related protein PR-1, the transcription factor involved in trichome formation GL3, and VSP2 activated by ethylene was clearly induced upon both treatments indicating that various defense genes were up-regulated upon volatile exposure (Figure 7E).
To further investigate whether the volatile compound 6PP is a plant defense inducer, we performed the following experiment: Arabidopsis plants were pretreated with 2 mM 6PP for 24 h followed by either the application of a spore suspension of the phytopathogenic fungus A. brassicicola or B. cinerea. After 5 days of incubation the lesion sizes were recorded. In both treatments the symptom sizes were reduced by ca. 40% and ca. 60%, respectively (Figure 8). We concluded that the exposure to 6PP induced resistance against fungal pathogens in Arabidopsis. A direct effect of 6PP on A. brassicicola was also investigated and spore germination was significantly reduced by 2 mM 6PP (Figure S7).
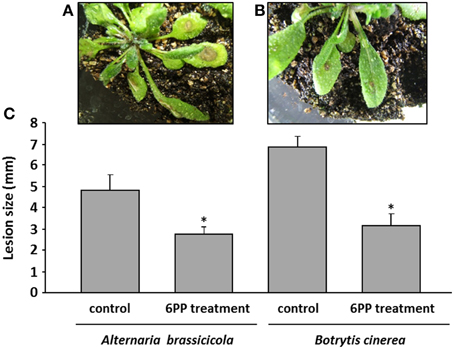
Figure 8. Development of disease symptoms in Arabidopsis thaliana leaves after preincubation with 6-pentyl-α-pyrone (6PP). Four weeks old Arabidopsis plants were grown in glass containers (see Effect of 6PP on Arabidopsis). Twenty milliliters of 2 mM 6PP were applied to the soil of each glass jar. The jar was covered with a lid and sealed with Parafilm®. Twenty-four hours later 50 μl of Botrytis cinerea spore suspension or of Alternaria brassicicola (each 5 × 106 spores/ml) were applied to the leaves. Representative examples of A. thaliana plants treated with a spore suspension of B. cinerea are shown in the upper panel (A): without preexposure to 6PP = control; (B): with 6PP preexposure. Lesions are visible as brown spots on the leaves. At the 5th day after inoculation the diameter of the lesions were recorded and calculated (C). The experiment was repeated for three times, each with four plants, n = 3, error bars indicate SD, *P < 0.05.
Discussion
The goal was to study morphological and physiological reactions of A. thaliana upon exposure to volatiles of four Trichoderma species. Only T. asperellum IsmT5 showed significant effects on plant growth and was therefore selected for further investigations. The fungal volatiles did not kill the plant but initiated several plant defense responses like the production of ROS, increased trichome number, accumulation of anthocyanins, and the phytoalexin camalexin. Furthermore, the expression of e.g., SA pathway depending defense genes were increased. We analyzed the headspace volatiles of T. asperellum IsmT5 and identified the most prominent VOC in the spectrum to be 6PP, which has a strong coconut-like aroma. Here we demonstrated a dual function of 6PP, (i) preincubation of A. thaliana with 6PP improved its resistance to the phytopathogenic fungi Botrytis and Alternaria (indirect action) and (ii) reduction of spore germination of Alternaria (direct action). These results support that Trichoderma volatiles have the potential being a useful biocontrol agent in agriculture.
Test System
Several co-cultivation systems were developed to test effects of fungal volatiles on plant growth. Naznin et al. (2013) and Hung et al. (2013) used airtight cultivation systems and observed growth promotion of tobacco and A. thaliana by Phoma and T. viride, respectively. We prepared slanted agar medium in glass jars for plant growth and inserted a beaker with fungi to avoid direct contact of both organisms (Figure S1A). This method had several advantages, e.g., easy and rapid inspection of the phenotypic responses of the roots and shoots during exposure to microbial volatiles, and no limitation to use only seedlings. We tested two alternative ways to cover the jars, either laying the lid on the jar however without sealing with Parafilm (closed system), or a funnel with sterile glass wool at the tip was placed upside down (open system) (Figure S1A). We preferred to use the open system (i) because it reflects the natural situation more closely, and (ii) to overcome the problem of CO2 accumulation due to metabolic activity of the microorganism, since it was recently shown that CO2 accumulated to 10-fold higher levels compared to ambient levels in closed containers (Kai and Piechulla, 2009).
Three different Trichoderma strains/isolates were investigated regarding their effects on Arabidopsis seedlings. Interestingly, the strains induced different phenotypic alterations in A. thaliana. The volatiles of T. asperellum IsmT5 influenced the plants negatively, which was manifested in the inhibition of primary root, reduction in size of leaves and fresh weight. In contrast, plant growth promotions due to Trichoderma volatiles have been observed by Hung et al. (2013) and Contreras-Cornejo et al. (2014). These contradictory results are most likely due to the different (open vs. closed) test systems used.
Headspace Analysis and 6-pentyl-α-pyrone Application
The defense responses initiated in A. thaliana upon Trichoderma volatile exposure prompted us to investigate the nature of the bioactive compound. Identification of headspace volatiles indicated different chemical classes i.e., alcohols, alkanes, and pyrones (Table 1). Similar compound diversity (25 different compounds) was also previously reported for Trichoderma atroviride (Stoppacher et al., 2010). Very prominent levels of 6PP were detected in the headspace of T. asperellum IsmT5. This compound and other α-pyrone analogs have been detected in cultures of T. viride (Collins and Halim, 1972), T. harzianum (Claydon et al., 1987), T. koningii (Simon et al., 1988), T. atroviride (Reithner et al., 2005), T. citrinoviride and T. hamatum, (Jeleñ et al., 2013), and recently in T. asperellum 328 (Wickel et al., 2013).
Lactones are generally very pleasant and potent flavor compounds (Kapfer et al., 1989) and a variety of microorganisms perform de novo lactone biosynthesis (Tressl et al., 1978). 6PP has a coconut-like aroma and it was known for long time that this odor appeared during Trichoderma cultivation (Bisby, 1939; Rifai, 1969). The emission of 6PP by T. asperellum IsmT5 reached levels of up to 450 ng/μl (= 2.7 mM), similar concentrations were reported by Kalyani et al. (2000) and Serrano-Carreón et al. (2004). A systematic optimization of growth conditions improved the production of 6PP by three orders of magnitude in T. atroviride (Oda et al., 2009). This was a success because of its use as a perfume in food and cosmetic industries. Beside the technological application it is well known that many natural lactones have antibacterial, antifungal or anti-inflammatory biological activity (Claydon et al., 1987; Simon et al., 1988; Cooney and Lauren, 1999; Pezet et al., 1999; Romero-Guido et al., 2011).
Plant Defense Reactions
The interaction of Trichoderma with a plant was thought to start by colonization of the outer root layers, resulting in the induction of resistance mechanisms to prevent further colonization (Yedidia et al., 1999; Harman et al., 2004; Mukherjee et al., 2013; Vos et al., 2015). Here we demonstrate that a volatile based interaction between A. thaliana and T. asperellum IsmT5 also exists, which might occur prior to physical contact. Although many evidences exist that Trichoderma activates plant immunity and development through different mechanisms, it was so far unknown whether microbial VOCs play a role in any of these plant defense processes (Contreras-Cornejo et al., 2014).
Here it was shown for the first time that Arabidopsis exposed to T. asperellum IsmT5 volatiles doubled its trichome number (Figure 3). Trichome formation in plants is a general defense strategy primarily developed to hinder landing, moving, and penetration of insects and other organisms on plant surfaces. The presented results indicate that the volatiles are apparently perceived as an intervening organismal interaction. While detailed investigations are needed to understand the underlying mechanisms related to these morphological alterations T. asperellum IsmT5 volatiles also induced typical plant defense responses at the physiological level, such as increasing the H2O2 level in leaves (Figure 4A). Similar observations were recorded by Splivallo et al. (2007). They found that truffle volatiles induced an oxidative burst in Arabidopsis. Increase of ROS in A. thaliana was also found upon bacterial volatile exposure (Wenke et al., 2012). Contradictory results of H2O2 production induced by Trichoderma volatiles were reported by Hung et al. (2013) and Contreras-Cornejo et al. (2014). Another early response in pathogen defense is the oxidation by peroxidases, subsequently reducing the amount of oxygen diffusing from the roots into the surrounding environment (Tiwari et al., 2002). This mechanism helps plant roots to avoid the uptake of toxic materials and thereby provides protection (Singh et al., 2007). The roots of volatile exposed plants showed increase in such root activity (Figure 4C). Similar results were recorded in rice and of seedlings of Ageratina adenophora (Zhang et al., 2012). Since ROS trigger many downstream processes leading to a dynamic defense responses characterized by inhibition of the growth of invaders through phytoalexin formation, callose deposition, strengthening of cell walls, synthesis of secondary metabolites and pathogenesis related (PR) proteins (Xu et al., 2008; Vinale et al., 2008b; Shoresh et al., 2010), we hypothesize that T. asperellum IsmT5 volatiles are perceived as oxidative stress and thus inducing alterations in the antioxidant enzyme machinery and accumulation of other protective substances in A. thaliana. A striking observation was the dark coloration of Trichoderma volatiles exposed leaves (Figure 2C) resulting from anthocyanin accumulation. Such accelerated anthocyanin accumulation due to volatile stress has to our knowledge not yet been reported although these results fit very well to the known function of anthocyanin to act as antimicrobial agents and feeding deterrents (Winkel-Shirley, 2001; Steyn et al., 2002; Shin et al., 2013). Furthermore, camalexin known as an important phytoalexin of Arabidopsis and an integral part of the plant defense system in A. thaliana (Glawischnig, 2007) was significantly up-regulated during volatiles exposure (Figure 4B). As other phytoalexins, camalexin production can be elicited by bacterial and fungal phytopathogens (as well as abiotic stress) and possesses antimicrobial activity (Großkinsky et al., 2012). Contreras-Cornejo et al. (2011) proved that Arabidopsis seedlings colonized with T. virens or T. atroviride accumulated high levels of camalexin. Camalexin deficient mutants such as pad3 (encodes last step in phytoalexin biosynthesis) displayed enhanced susceptibility to B. cinerea (Ferrari et al., 2003), and in A. thaliana treated with T. atroviride pad3 was up-regulated in roots and leaves (Salas-Marina et al., 2011). In this study the accumulation of camalexin due to T. asperellum IsmT5 volatile exposure highlights a new aspect and showed that not only by direct contact between plant and Trichoderma camalexin is induced but also by air borne signals. Glucosinolates play central roles in plant/biotic interactions and they are important determinants for plant fitness in the field. Hydrolysis products of glucosinolates are active against a wide variety of organisms, such as insects, plants, fungi, and bacteria (Vaughn, 1999). In this study we analyzed the levels of glucosinolates in Arabidopsis plants co-cultivated with T. asperellum IsmT5 or exposed to the fungal volatile 6PP. Our results demonstrate that 6PP caused a significant reduction in the accumulation of glucosinolates, while levels were not significantly altered in plants co-cultivated with T. asperellum IsmT5 (Figure 7D). Whereas the catabolism of glucosinolates and de novo biosynthesis is well balanced in plants co-cultivated with T. asperellum IsmT5, the degradation seems to be more dominant in 6PP-treated plants. Apparently, other volatiles of T. asperellum IsmT5 spectrum prevent a reduction of the essential glucosinolates and consequently without weakening the plant defense system. The function of Trp-derived indole glucosinolates in Arabidopsis immunity was validated with infection phenotypes of cyp81F2 and penetration2 (pen2) mutants. Analyses of loss-of-function mutant of pen2 (an alternative myrosinase) suggested that PEN2-mediated glucosinolate metabolism is important for pre-invasive resistance, while camalexin is contributing at the post-invasive stage of immunity (Lipka et al., 2005; Hiruma et al., 2010; Sanchez-Vallet et al., 2010; Schlaeppi et al., 2010). These observations are also in agreement with the activation of the SA pathway of T. asperellum Ism5 co-cultivated plants as shown in (Figure 5) and MYB51 has been previously reported to be important regulator of glucosinolates at SA-signaling (Frerigmann and Gigolashvili, 2014). We therefore expected the increase in the levels of pathogen related genes such as DR5::GUS, YUC8::GUS, PDF1.2::GUS, PR1::GUS, MYB51::GUS (Figure 6 and Figure S3). These results further supported that fungal volatiles were perceived as stress, which initiated defense processes and improved plant immunity.
The defense network in plants is regulated by the action of plant hormones via two main mechanisms, systemic acquired resistance (involvement of SA) and induced systemic resistance (regulated by ethylene and JA). It became clear that an intensive interplay between hormone signaling pathways exists, which effectively determines the response to specific types of invader. The activation of phytohormone signaling cascades by direct interactions between Trichoderma spp. and A. thaliana or other plants was already demonstrated to trigger JA or SA dependent systemic resistance (Contreras-Cornejo et al., 2011; Salas-Marina et al., 2011; Velázquez-Robledo et al., 2011; Yoshioka et al., 2012; Vos et al., 2015), however volatile based activations add a new facette to such strategies. While SA and ABA accumulated in A. thaliana exposed to T. asperellum IsmT5 volatiles, levels of JA were not altered (Figure 5). Why, in contrast, the marker gene for the JA/ethylene mediated signaling in A. thaliana, PDF 1.2a, was expressed in transgenic lines (Figure S3) could be explained by a species-specific activation of the signaling cascades as found in T. hamatum T382 and T. asperelloides (Mathys et al., 2012; Brotman et al., 2013).
The presented results show that A. thaliana perceives the Trichoderma volatiles as stress compounds and subsequently initiates multilayered (morphological, physiological, and gene expression level) adaptations and activations of signaling cascades to withstand this environmental influence. This hypothesis was supported when the major volatile compound of the VOC spectrum of T. asperellum Ism5 was identified and A. thaliana preincubated with the pure compound 6PP and challenged with the phytopathogenic fungi Botrytis and Alternaria. The leaves showed significantly less necrotic symptoms compared to untreated plants (Figure 8). Consequently it was concluded that the volatiles of T. asperellum IsmT5 or 6PP activate the accumulation of typical defense molecules such as ROS, camalexin, anthocyanins, and the SA dependent plant hormone pathways and subsequently defense-activated plants become more resistant to pathogen attack and exhibited smaller lesions. Vinale et al. (2008a) also reported a reduction of disease symptoms in pea, tomato, and canola seedlings after addition of purified secondary metabolites. This effect correlated with elevated expression of chitinase, PR1 protein, and endochitinase. Maize plants growing in soil that was drenched with 6PP for 4 days prior to inoculation with Fusarium moniliforme showed considerable suppression of seedling blight compared to untreated controls (El-Hasan and Buchenauer, 2009). Cutler et al. (1986) were the first who recorded inhibitory effects on wheat coleoptiles. In addition, 6PP acts directly on spore germination of A. brassicicola (Figure S7), also demonstrated by Intana and Chamswarng (2007) and Yenjit et al. (2008) for A. brassicicola and Phythium aphanidermatum, respectively. Taken together, volatile metabolites from T. asperelleum IsmT5 (6PP) are involved in direct and indirect interactions between fungi and plants thereby have the potential to influence biocontrol processes (Cottier and Mühlschlegel, 2012). Deciphering the signaling cascades in plants that are induced by Trichoderma volatiles is a future challenge. So far, to the best of our knowledge, only one example is known which showed that a WRKY transcription factor is part of the signaling cascade in bacterial volatile plant interactions (Wenke et al., 2012). In the future, further investigations at the molecular level are required to shed light on the role of Trichoderma volatiles, especially 6PP, and bacterial volatiles in activation of defense mechanisms in plants to improve plant resistance and to design plant protection systems.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors thank Dr. Zoughlin Mou (University of Florida, Gainsville, USA) for Arabidopsis thaliana Col-0, Dr. Eckehard Koch (Julius-Kühn-Institute, Braunschweig, Germany) for Alternaria brassicicola, Dr. Andreas v. Tiedemann (University of Göttingen, Germany) for Botrytis cinerea, Dr. Zsuzsanna Kolbert (University of Szeged, Hungary) for DR5::GUS, Dr. Anja van Dijken (Plant-Microbe Interactions, Utrecht University) for PDF1,2::GUS, Dr. Stephan Pollmann (Metabolomics Unit at the Center of Plant Biotechnology and Genomics, Madrid, Spain) for pYUC8::GUS for kindly sending different biological materials. We thank Dr. Henning Frerigman (University of Cologne) for the UPLC analysis of glucosinolates, Dr. Hartwig Pfeifhofer for supporting the camalexin and phytohormone analyses (University of Graz), and Dr. Uta Effmert for preparing the figures and preparing the reference list. DKG is funded by the Individual Postdoctoral Grant No. 4093-00255 of the Danish Council for Independent Research, Danish Ministry of Higher Education and Science. We appreciate the contribution of the Egyptian Ministery of Higher Education and the DAAD for supporting MK, and the University of Rostock for financial support to BP.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fmicb.2015.00995
Abbreviations
VOCs, Volatile organic compounds; 6PP, 6-pentyl-α-pyrone; gus, glucoronidase; ROS, reactive oxygen species, SA, salicylic acid; JA, jasmonic acid; ABA, abscisic acid.
References
Altomare, C., Norvell, W. A., Bjorkman, T., and Harman, G. E. (1999). Solubilization of phosphates and micronutrients by the plant-growth-promoting and biocontrol fungus Trichoderma harzianum Rifai 1295–22. Appl. Environ. Microbiol. 65, 2926–2933.
Bae, H., Sicher, R. C., Kim, M. S., Kim, S. H., and Strem, M. D. (2009). The beneficial endophyte Trichoderma hamatum isolate DIS 219b promotes growth and delays the onset of the drought response in Theobroma cacao. J. Exp. Bot. 60, 3279–3295. doi: 10.1093/jxb/erp165
Bari, R., and Jones, J. D. G. (2009). Role of plant hormones in plant defence responses. Plant Mol. Biol. 69, 473–488. doi: 10.1007/s11103-008-9435-0
Bisby, G. R. (1939). Trichoderma viride Pers. ex Fries, and notes on Hypocrea. Trans. Br. Mycol. Soc. 23, 149–168. doi: 10.1016/S0007-1536(39)80020-1
Błaszczyk, L., Popiel, D., Chełkowski, J., Koczyk, G., Samuels, G. J., Sobieralski, K., et al. (2011). Species diversity of Trichoderma in Poland. J. Appl. Genetics. 52, 233–243. doi: 10.1007/s13353-011-0039-z
Blom, D., Fabbri, C., Connor, E. C., Schiestl, F. P., Klauser, D. R., Boller, T., et al. (2011). Production of plant growth modulating volatiles is widespread among rhizosphere bacteria and strongly depends on culture conditions. Environ. Microbiol. 13, 3047–3058. doi: 10.1111/j.1462-2920.2011.02582.x
Bonnarme, P., Djian, A., Latrasse, A., Féron, G., Giniès, C., Durand, A., et al. (1997). Production of 6-pentyl-α-pyrone by Trichoderma sp. from vegetable oils. J. Biotechnol. 56, 143–150. doi: 10.1016/S0168-1656(97)00108-9
Brotman, Y., Kapuganti, J. G., and Viterbo, A. (2010). Trichoderma. Curr. Biol. 20, 390–391. doi: 10.1016/j.cub.2010.02.042
Brotman, Y., Landau, U., Cuadros-Inostroza, Á., Takayuki, T., Fernie, A. R., Chet, I., et al. (2013). Trichoderma-plant root colonization: escaping early plant defense responses and activation of the antioxidant machinery for saline stress tolerance. PLoS Pathog. 9:1003221. doi: 10.1371/journal.ppat.1003221
Chakrabarty, D., Verma, A. K., and Datta, S. K. (2009). Oxidative stress and antioxidant activity as the basis of senescence in Hemerocallis (day lily) flowers. J. Hortic. For. 1, 113–119.
Claydon, N., Allan, M., Hanson, J. R., and Avent, A. G. (1987). Antifungal alkyl pyrones of Trichoderma harzianum. Trans. Br. Mycol. Soc. 88, 503–513. doi: 10.1016/S0007-1536(87)80034-7
Collins, R. P., and Halim, A. F. (1972). Characterization of the major aroma constituent of the fungus Trichoderma viride. J. Agric. Food Chem. 20, 437–438. doi: 10.1021/jf60180a010
Comas, L. H., Eissenstat, D. M., and Lakso, A. N. (2000). Assessing root death and root system dynamics in a study of grape canopy pruning. New Phytol. 147, 171–178. doi: 10.1046/j.1469-8137.2000.00679.x
Conrath, U. (2011). Molecular aspects of defence priming. Trends Plant Sci. 16, 524–531. doi: 10.1016/j.tplants.2011.06.004
Contreras-Cornejo, H. A., Macias-Rodriguez, L., Beltran-Peña, E., Herrera-Estrella, A., and Lopez-Bucio, J. (2011). Trichoderma-induced plant immunity likely involves both hormonal- and camalexin dependent mechanisms in Arabidopsis thaliana and confers resistance against necrotrophic fungus Botrytis cinerea. Plant Signal. Behav. 6, 1554. doi: 10.4161/psb.6.10.17443
Contreras-Cornejo, H. A., Macías-Rodríguez, L., Cortés-Penagos, C., and López-Bucio, J. (2009). Trichoderma virens, a plant beneficial fungus, enhances biomass production and promotes lateral root growth through an auxin-dependent mechanism in Arabidopsis. Plant Physiol. 149, 1579–1592. doi: 10.1104/pp.108.130369
Contreras-Cornejo, H. A., Macías-Rodríguez, L., Herrera-Estrella, A., and López-Bucio, J. (2014). The 4-phosphopantetheinyl transferase of Trichoderma virens plays a role in plant protection against Botrytis cinerea through volatile organic compound emission. Plant Soil 379, 261–274. doi: 10.1007/s11104-014-2069-x
Cooney, J. M., and Lauren, D. R. (1999). Biotransformation of the Trichoderma metabolite 6-n-pentyl-2H-pyran-2-one (6PAP) by selected fungal isolates. J. Nat. Prod. 62, 681–683. doi: 10.1021/np980349o
Cottier, F., and Mühlschlegel, F. A. (2012). Communication in Fungi. Int. J. Microbiol. 2012:351832. doi: 10.1155/2012/351832
Cutler, H. G., Cox, R. H., Crumley, F. G., and Cole, P. D. (1986). 6-Phenyl-α-pyrone from Trichoderma harzianum: its plant growth inhibitory and antimicrobial properties. Agric. Biol. Chem. 50, 2943–2945. doi: 10.1271/bbb1961.50.2943
D'Alessandro, M., Erb, M., Ton, J., Brandenburg, A., Karlen, D., Zopfi, J., et al. (2014). Volatiles produced by soil-borne endophytic bacteria increase plant pathogen resistance and affect tritrophic interactions. Plant Cell Environ. 37, 813–826. doi: 10.1111/pce.12220
Effmert, U., Kalderás, J., Warnke, R., and Piechulla, B. (2012). Volatile mediated interactions between bacteria and fungi in soil. J. Chem. Ecol. 38, 665–703. doi: 10.1007/s10886-012-0135-5
El-Hasan, A., and Buchenauer, H. (2009). Actions of 6-pentyl-alpha-pyrone in controlling seedling blight incited by Fusarium moniliforme and inducing defence responses in maize. J. Phytopathol. 157, 697–707. doi: 10.1111/j.1439-0434.2009.01565.x
Ezquer, I., Li, J., Ovecka, M., Baroja-Fernández, E., Jose Muñoz, F., Montero, M., et al. (2010). Microbial volatile emissions promote accumulation of exceptionally high levels of starch in leaves in mono- and dicotyledonous plants. Plant Cell Physiol. 51, 1674–1693. doi: 10.1093/pcp/pcq126
Ferrari, S., Plotnikova, J. M., De Lorenzo, G., and Ausubel, F. M. (2003). Arabidopsis local resistance to Botrytis cinerea involves salicylic acid and camalexin and requires EDS4 and PAD2, but not SID2, EDS5 or PAD4. Plant J. 35, 193–205. doi: 10.1046/j.1365-313X.2003.01794.x
Frerigmann, H., Böttcher, C., Baatout, D., and Gigolashvili, T. (2012). Glucosinolates are produced in trichomes of Arabidopsis thaliana. Front. Plant Sci. 3:242. doi: 10.3389/fpls.2012.00242
Frerigmann, H., and Gigolashvili, T. (2014). MYB34, MYB51, and MYB122 distinctly regulate indolic glucosinolate biosynthesis in Arabidopsis thaliana. Mol. Plant 7, 814–828. doi: 10.1093/mp/ssu004
Gravel, V., Antoun, H., and Tweddell, R. J. (2007). Growth stimulation and fruit yield improvement of greenhouse tomato plants by inoculation with Pseudomonas putida or Trichoderma atroviride: possible role of indole acetic acid (IAA). Soil Biol. Biochem. 39, 1968–1977. doi: 10.1016/j.soilbio.2007.02.015
Großkinsky, D. K., Albacete, A., Jammer, A., Krbez, P., van der Graaff, E., Pfeifhofer, H., et al. (2014). A rapid phytohormone and phytoalexin screening method for physiological phenotyping. Mol. Plant 7, 1053–1056. doi: 10.1093/mp/ssu015
Großkinsky, D. K., van der Graaff, E., and Roitsch, T. (2012). Phytoalexin transgenics in crop protection – fairy tale with a happy end? Plant Sci. 195, 54–70. doi: 10.1016/j.plantsci.2012.06.008
Harman, G. E., Howell, C. R., Viterbo, A., Chet, I., and Lorito, M. (2004). Trichoderma species: opportunistic, avirulent plant symbionts. Nat. Rev. Microbiol. 2, 43–56. doi: 10.1038/nrmicro797
Hermosa, R., Viterbo, A., Chet, I., and Monte, E. (2012). Plant-beneficial effects of Trichoderma and of its genes. Microbiology 158, 17–25. doi: 10.1099/mic.0.052274-0
Hippauf, F., Michalsky, E., Huang, R., Preissner, R., Barkman, T. J., and Piechulla, B. (2010). Enzymatic, expression and structural divergences among carboxyl O-methyltransferases after gene duplication and speciation in Nicotiana. Plant Mol. Biol. 72, 311–330. doi: 10.1007/s11103-009-9572-0
Hiruma, K., Onozawa-Komori, M., Takahashi, F., Asakura, M., Bednarek, P., Okuno, T., et al. (2010). Entry mode-dependent function of an indole glucosinolate pathway in Arabidopsis for nonhost resistance against anthracnose pathogens. Plant Cell 22, 2429–2443. doi: 10.1105/tpc.110.074344
Howe, G. A., and Jander, G. (2008). Plant immunity to insect herbivores. Annu. Rev. Plant Biol. 59, 41–66. doi: 10.1146/annurev.arplant.59.032607.092825
Hung, R., Lee, S., and Bennett, J. W. (2013). Arabidopsis thaliana as a model system for testing the effect of Trichoderma volatile organic compounds. Fungal Ecol. 6, 19–26. doi: 10.1016/j.funeco.2012.09.005
Intana, W., and Chamswarng, C. (2007). Control of Chinese-kale damping-off caused by Pythium aphanidermatum by antifungal metabolites of Trichoderma virens. Songklanakarin J. Sci. Technol. 29, 919–927.
Jefferson, R. A., Kavanagh, T. A., and Bevan, M. W. (1987). GUS fusions: b-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J. 6, 3901–3907.
Jeleñ, H., Błaszczyk, L., Chełkowski, J., Rogowicz, K., and Strakowska, J. (2013). Formation of 6-n-pentyl-2H-pyran-2-one (6-PAP) and other volatiles by different Trichoderma species. Mycol. Prog. 13, 589–600. doi: 10.1007/s11557-013-0942-2
Junker, R. R., and Tholl, D. (2013). Volatile organic compound mediated interactions at the plant-microbe interface. J. Chem. Ecol. 39, 810–825. doi: 10.1007/s10886-013-0325-9
Kai, M., Effmert, U., Berg, G., and Piechulla, B. (2007). Volatiles of bacterial antagonists inhibit mycelial growth of the plant pathogen Rhizoctonia solani. Arch. Microbiol. 187, 351–360. doi: 10.1007/s00203-006-0199-0
Kai, M., Haustein, M., Molina, F., Petri, A., Scholz, B., and Piechulla, B. (2009). Bacterial volatiles and their action potential. Planta 81, 1001–1012. doi: 10.1007/s00253-008-1760-3
Kai, M., and Piechulla, B. (2009). Plant growth promotion due to rhizobacterial volatiles - an effect of CO2? FEBS Lett. 583, 3473–3477. doi: 10.1016/j.febslet.2009.09.053
Kalyani, A., Prapulla, S. G., and Karanth, N. G. (2000). Study on the production of 6-pentyl-alpha-pyrone using two methods of fermentation. Appl. Microbiol. Biotechnol. 53, 610–612. doi: 10.1007/s002530051665
Kapfer, G. F., Berger, R. G., and Drawert, F. (1989). Production of 4-decanolide by semicontinuous fermentation of Tyromyces sambuceus. Biotechnol. Lett. 11, 561–566. doi: 10.1007/BF01040036
Katagiri, F., and Tsuda, K. (2010). Understanding the plant immune system. Mol. Plant Microbe Interact. 12, 1531–1536. doi: 10.1094/MPMI-04-10-0099
Kishimoto, K., Matsui, K., Ozawa, R., and Takabayashi, J. (2007). Volatile 1-octen-3-ol induces a defensive response in Arabidopsis thaliana. J. Gen. Plant Pathol. 73, 35–37. doi: 10.1007/s10327-006-0314-8
Klein, D., and Eveleigh, D. E. (1998). “Ecology of Trichoderma,” in Trichoderma and Gliocaldium, Vol. 1: Basic Biology, Taxonomy and Genetics, eds C. P. Kubicek and G. E. Harman (London: Taylor and Francis), 57–73.
Kramer, R., and Abraham, W. R. (2012). Volatile sesquiterpenes from fungi: what are they good for? Phytochem. Rev. 11, 15–37. doi: 10.1007/s11101-011-9216-2
Kuhls, K., Lieckfeldt, E., Börner, T., and Guého, E. (1999). Molecular reidentification of human pathogenic Trichoderma isolates as Trichoderma longibrachiatum and Trichoderma citrinoviride. Med. Mycol. 37, 25–33. doi: 10.1080/02681219980000041
Lemfack, M. C., Nickel, J., Dunkel, M., Preissner, R., and Piechulla, B. (2013). mVOC: a database of microbial volatiles. Nucl. Acids Res. 42, D744–D748. doi: 10.1093/nar/gkt1250
Lipka, V., Dittgen, J., Bednarek, P., Bhat, R., Wiermer, M., and Stein, M. (2005). Pre- and postinvasion defenses both contribute to nonhost resistance in Arabidopsis. Science 310, 1180–1183. doi: 10.1126/science.1119409
Lorito, M., Woo, S. L., Harman, G. E., and Monte, E. (2010). Translational research on Trichoderma: from ‘omics to the field. Annu. Rev. Phytopathol. 48, 395–417. doi: 10.1146/annurev-phyto-073009-114314
Martínez-Medina, A., Roldán, A., and Pascual, J. A. (2011). Interaction between arbuscular mycorrhizal fungi and Trichoderma harzianum under conventional and low input fertilization field condition in melon crops: growth response and Fusarium wilt biocontrol. Appl. Soil Ecol. 47, 98–105. doi: 10.1016/j.apsoil.2010.11.010
Mathys, J., De Cremer, K., Timmermans, P., Van Kerckhove, S., Lievens, B., and Vanhaecke, M. (2012). Genome-wide characterization of ISR induced in Arabidopsis thaliana by Trichoderm ahamatum T382 against Botrytis cinerea infection. Front. Plant Sci. 3:108. doi: 10.3389/fpls.2012.00108
Minerdi, D., Bossi, S., Gullino, M. L., and Garibaldi, A. (2009). Volatile organic compounds: a potential direct long-distance mechanism for antagonistic action of Fusarium oxysporum strain MSA 35. Environ. Microbiol. 11, 844–854. doi: 10.1111/j.1462-2920.2008.01805.x
Moreira, E. E., Mexia, J. T., and Minder, C. E. (2013). F tests with random samples size. Theory and Applications. Stat. Probab. Lett. 83, 1520–1526. doi: 10.1016/j.spl.2013.02.020
Mukherjee, P. K., Horwitz, B. A., Herrera-Estrella, A., Schmoll, M., and Kenerley, C. M. (2013). Trichoderma research in the genome era. Annu. Rev. Phytopathol. 51, 105–129. doi: 10.1146/annurev-phyto-082712-102353
Murashige, T., and Skoog, F. (1962). A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plantarum 15, 473–497. doi: 10.1111/j.1399-3054.1962.tb08052.x
Naznin, H. A., Kimura, M., Miyazawa, M., and Hyakumachi, M. (2013). Analysis of volatile organic compounds emitted by plant growth-promoting fungus Phoma sp. GS8-3 for growth promotion effects on tobacco. Microbes Environ. 28, 42–49. doi: 10.1264/jsme2.ME12085
Neff, M. M., and Chory, J. (1998). Genetic interactions between phyochrome A, phytochrome B, and cryptochrome 1 during Arabidopsis development. Plant Physiol. 118, 27–35. doi: 10.1104/pp.118.1.27
Oda, S., Isshiki, K., and Ohashi, S. (2009). Production of 6-pentyl-α-pyrone with Trichoderma atroviride and its mutant in a novel extractive liquid-surface immobilization (Ext-LSI) system. Process Biochem. 44, 625–630. doi: 10.1016/j.procbio.2009.01.017
Pezet, R., Pont, V., and Tabacchi, R. (1999). Simple analysis of 6-pentyl-α-pyrone, a major antifungal metabolite of Trichoderma spp., useful for testing the antagonistic activity of these fungi. Phytochem. Analysis 10, 285–288.
Piechulla, B., and Degenhardt, J. (2014). The emerging importance of microbial volatile organic compounds. Plant Cell Environ. 37, 811–812. doi: 10.1111/pce.12254
Porra, R. J., Thompson, W. A., and Kriedemann, P. E. (1989). Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim Biophys. Acta Bioenerg. 975, 384–394. doi: 10.1016/S0005-2728(89)80347-0
Reino, J. L., Guerrero, R. F., Hernandez-Galan, R., and and Collado, I. G. (2008). Secondary metabolites from species of the biocontrol agent Trichoderma. Phytochem. Rev. 7, 89–123. doi: 10.1007/s11101-006-9032-2
Reithner, B., Brunner, K., Schuhmacher, R., Peissl, P., Seidl, V., Krska, R., et al. (2005). The G protein α subunit Tga1 of Trichoderma atroviride is involved in chitinase formation and differential production of antifungal metabolites. Fungal Genet. Biol. 42, 749–760. doi: 10.1016/j.fgb.2005.04.009
Romero-Guido, C., Belo, I., Ta, T. M., Cao-Hoang, L., Alchihab, M., Gomes, N., et al. (2011). Biochemistry of lactone formation in yeast and fungi and its utilisation for the production of flavour and fragrance compounds. Appl. Microbiol. Biotechnol. 89, 535–547. doi: 10.1007/s00253-010-2945-0
Rotblat, B., Enshell-Seijffers, D., Gershoni, J. M., Schuster, S., and Avni, A. (2002). Identification of an essential component of the elicitation active site of the EIX protein elicitor. Plant J. 32, 1049–1055. doi: 10.1046/j.1365-313X.2002.01490.x
Salas-Marina, M. A., Silva-Flores, M. A., Uresti-Rivera, E. E., Castro-Longoria, E., Herrera-Estrella, A., and Casas-Flores, S. (2011). Colonization of Arabidopsis roots by Trichoderma atroviride promotes growth and enhances systemic disease resistance through jasmonic acid/ethylene and salicylic acid pathways. Eur. J. Plant Pathol. 131, 15–26. doi: 10.1007/s10658-011-9782-6
Sanchez-Vallet, A., Ramos, B., Bednarek, P., López, G., Pislewska-Bednarek, M., and Schulze-Lefert, P. (2010). Tryptophan-derived secondary metabolites in Arabidopsis thaliana confer nonhost resistance to necrotrophic Plectosphaerella cucumerina fungi. Plant J. 63, 115–127. doi: 10.1111/j.1365-313X.2010.04224.x
Schlaeppi, K., Abou-Mansour, E., Buchala, A., and Mauch, F. (2010). Disease resistance of Arabidopsis to Phytophthora brassicae is established by the sequential action of indole glucosinolates and camalexin. Plant J. 62, 840–851. doi: 10.1111/j.1365-313X.2010.04197.x
Serrano-Carreón, L., Flores, C., Rodríguez, B., and Galindo, E. (2004). Rhizoctonia solani, an elicitor of 6-pentyl-α-pyrone production by Trichoderma harzianum in a two liquid phases, extractive fermentation system. Biotechnol. Lett. 26, 1403–1406. doi: 10.1023/B:BILE.0000045640.71840.b5
Shen, H. C., Zhou, W. J., Xi, H. F., and Ye, Q. F. (1991). A preliminary study of physiological and yield effects of paclobutrazol on Brassica napus. Acta Agric. Univ. Zhejiang 17, 423–426.
Shin, D. H., Choi, M. G., Kim, K., Bang, G., Cho, M., Choi, S. B., et al. (2013). HY5 regulates anthocyanin biosynthesis by inducing the transcriptional activation of the MYB75/PAP1 transcription factor in Arabidopsis. FEBS Lett. 587, 1543–1547. doi: 10.1016/j.febslet.2013.03.037
Shoresh, M., Harman, G. E., and Mastouri, F. (2010). Induced systemic resistance and plant responses to fungal biocontrol agents. Annu. Rev. Phytopathol. 48, 21–43. doi: 10.1146/annurev-phyto-073009-114450
Simon, A., Dunlop, R. W., Ghisalberti, E. L., and Sivasithamparama, K. (1988). Trichoderma koningii produces a pyrone compound with antibiotic properties. Soil Biol. Biochem. 20, 263–264. doi: 10.1016/0038-0717(88)90050-8
Singh, H. B., Singh, B. N., Singh, S. P., and Nautiyal, C. S. (2010). Solid-state cultivation of Trichoderma harzianum NBRI-1055 for modulating natural antioxidants in soybean seed matrix. Bioresour. Technol. 101, 6444–6453. doi: 10.1016/j.biortech.2010.03.057
Singh, H. P., Batish, D. R., Kohli, R. K., and Arora, K. (2007). Arsenic-induced root growth inhibition in mung bean (Phaseolus aureus Roxb.) is due to oxidative stress resulting from enhanced lipid peroxidation. Plant Growth Regul. 53, 65–73. doi: 10.1007/s10725-007-9205-z
Splivallo, R., Novero, M., Bertea, C. M., Bossi, S., and Bonfante, P. (2007). Truffle volatiles inhibit growth and induce an oxidative burst in Arabidopsis thaliana. New Phytol. 175, 417–424. doi: 10.1111/j.1469-8137.2007.02141.x
Splivallo, R., Ottonello, S., Mello, A., and Karlovsky, P. (2011). Truffle volatiles: from chemical ecology to aroma biosynthesis. New Phytol. 189, 688–699. doi: 10.1111/j.1469-8137.2010.03523.x
Steyn, W. J., Wand, S. J. E., Holcroft, D. M., and Jacobs, G. (2002). Anthocyanins in vegetative tissues: a proposed unified function in photoprotection. New Phytol. 155, 349–361. doi: 10.1046/j.1469-8137.2002.00482.x
Stoppacher, N., Kluger, B., Zeilinger, S., Krska, R., and Schumacher, R. (2010). Identification and profiling of volatile metabolites of the biocontrol fungus Trichoderma atroviridae by HS-SPME-GC-.MS. J. Microbiol. Methods 81, 187–193. doi: 10.1016/j.mimet.2010.03.011
Tiwari, B. S., Belenghi, B., and Levine, A. (2002). Oxidative stress increased respiration and generation of reactive oxygen species, resulting in ATP depletion, opening of mitochondrial permeability transition, and programmed cell death. Plant Physiol. 128, 1271–1281. doi: 10.1104/pp.010999
Tressl, R., Friese, L., Fendesack, F., and Köppler, H. (1978). Gas chromatographic-mass spectrometric investigation of hop aroma constituents in beer. J. Agric. Food Chem. 26, 1422–1426. doi: 10.1021/jf60220a037
Vaughn, S. F. (1999). “Glucosinolates as natural pesticides,” in Principles and Practices in Plant Allelochemical Interactions, eds H. G. Cutler and S. J. Cutler (Boca Raton, FL: CRC Press), 81–91. doi: 10.1201/9781420048629.ch7
Velázquez-Robledo, R., Contreras-Cornejo, H. A., Macías-Rodríguez, L., Hernández-Morales, A., Aguirre, J., Casas-Flores, S., et al. (2011). Role of the 4-phosphopantetheinyl transferase of Trichoderma virens in secondary metabolism and induction of plant defense responses. Mol. Plant Microbe Interact. 24, 1459–1471. doi: 10.1094/MPMI-02-11-0045
Verhage, A., van Wees, S. C. M., and Pieterse, C. M. J. (2010). Plant immunity: it's the hormones talking, but what do they say? Plant Physiol. 154, 536–540. doi: 10.1104/pp.110.161570
Verhagen, B. W. M., Glazebrook, J., Zhu, T., Chang, H. S., Van Loon, L. C., and Pieterse, C. M. J. (2004). The transcriptome of rhizobacteria-induced systemic resistance in Arabidopsis. Mol. Plant Microbe Interact. 17, 895–908. doi: 10.1094/MPMI.2004.17.8.895
Verma, M., Brar, S., Tyagi, R., Surampalli, R., and Valero, J. (2007). Antagonistic fungi, Trichoderma spp.: panoply of biological control. Biochem. Eng. J. 37, 1–20. doi: 10.1016/j.bej.2007.05.012
Vespermann, A., Kai, M., and Piechulla, B. (2007). Rhizobacterial volatiles affect the growth of fungi and Arabidopsis thaliana. Appl. Environ. Microb. 73, 5639–5641. doi: 10.1128/AEM.01078-07
Vinale, F., Sivasithamparam, K., Ghisalberti, E. L., Marra, R., Barbetti, M. J., Li, H., et al. (2008a). A novel role for Trichoderma secondary metabolites in the interactions with plants. Physiol. Mol. Plant Pathol. 72, 80–86. doi: 10.1016/j.pmpp.2008.05.005
Vinale, F., Sivasithamparam, K., Ghisalberti, E. L., Marra, R., Woo, S. L., and Lorito, M. (2008b). Trichoderma–plant–pathogen interactions. Soil Biol. Biochem. 40, 1–10. doi: 10.1016/j.soilbio.2007.07.002
Vinale, F., Sivasithamparam, K., Ghisalberti, E. L., Ruocco, M., Woo, S. L., and Lorito, M. (2012). Trichoderma Secondary Metabolites that Affect Plant Metabolism. Nat. Prod. Commun. 7, 1545–1550.
Vos, C. M. F., De Cremer, K., Cammue, B. P. A., and De Coninck, B. (2015). The toolbox of Trichoderma spp. in biocontrol of Botrytis cinerea disease. Mol. Plant Pathol. 16, 400–412. doi: 10.1111/mpp.12189
Weindling, R. (1932). Trichoderma lignorum as a parasite of other soil fungi. Phytopathology 22, 837–845.
Wenke, K., Kai, M., and Piechulla, B. (2010). Belowground volatiles facilitate interactions between plant roots and soil organisms. Planta 231, 499–506. doi: 10.1007/s00425-009-1076-2
Wenke, K., Wanke, D., Kilian, J., Berendzen, K., Harter, K., and Piechulla, B. (2012). Volatiles of two growth-inhibiting rhizobacteria commonly engage AtWRKY18 function. Plant J. 70, 445–459. doi: 10.1111/j.1365-313X.2011.04891.x
Wickel, S. M., Citron, C. A., and Dickschat, J. S. (2013). 2H-Pyran-2-ones from Trichoderma viride and Trichoderma asperellum. Eur. J. Org. Chem. 2013, 2906–2913. doi: 10.1002/ejoc.201300049
Winkel-Shirley, B. (2001). Flavonoid biosynthesis: a colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol. 126, 485–493. doi: 10.1104/pp.126.2.485
Xu, X., Qin, G., and Tian, S. (2008). Effect of microbial biocontrol agents on alleviating oxidative damage of peach fruit subjected to fungal pathogen. Int. J. Food Microbiol. 126, 153–158. doi: 10.1016/j.ijfoodmicro.2008.05.019
Yedidia, I., Benhamou, N., and Chet, I. (1999). Induction of defense responses in cucumber plants (Cucumis sativus L.) by the Biocontrol agent Trichoderma harzianum. Appl. Environ. Microbiol. 65, 1061–1070.
Yenjit, P., Issarakraisila, M., Intana, W., Sattasalalchai, S., Suwanno, T., and Chantrapromma, K. (2008). Efficacy of extracted substances from the pericarp of Garcinia mangostana to control major diseases of tropical fruits in the laboratory. Acta Hortic. 787, 339–343. doi: 10.17660/actahortic.2008.787.42
Yoshioka, Y., Ichikawa, H., Naznin, H. A., Kogure, A., and Hyakumachi, M. (2012). Systemic resistance induced in Arabidopsis thaliana by Trichoderma asperellum SKT-1, a microbial pesticide of seedborne diseases of rice. Pest Manag. Sci. 68, 60–66. doi: 10.1002/ps.2220
Keywords: Trichoderma asperellum IsmT5, Botrytis cinerea, Alternaria brassicicola, Arabidopsis thaliana, mVOCs, 6-pentyl-α-pyrone, glucosinolates, camalexin
Citation: Kottb M, Gigolashvili T, Großkinsky DK and Piechulla B (2015) Trichoderma volatiles effecting Arabidopsis: from inhibition to protection against phytopathogenic fungi. Front. Microbiol. 6:995. doi: 10.3389/fmicb.2015.00995
Received: 18 June 2015; Accepted: 07 September 2015;
Published: 29 September 2015.
Edited by:
Paolina Garbeva, Netherlands Institute of Ecology (NIOO-KNAW), NetherlandsReviewed by:
Ruth Schmidt, Netherlands Institute of Ecology (NIOO-KNAW), NetherlandsOlaf Tyc, The Royal Netherlands Academy of Arts and Sciences, Netherlands
Tomislav Cernava, Graz University of Technology, Austria
Copyright © 2015 Kottb, Gigolashvili, Großkinsky and Piechulla. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Birgit Piechulla, Institute for Biological Sciences, University of Rostock, Albert-Einstein-Str. 3, 18059 Rostock, Germany,YmlyZ2l0LnBpZWNodWxsYUB1bmktcm9zdG9jay5kZQ==
†Present Address: Metwally Kottb, Botany Department, Faculty of Science, Suez Canal University, Ismailia, Egypt
 Metwally Kottb
Metwally Kottb Tamara Gigolashvili
Tamara Gigolashvili Dominik K. Großkinsky
Dominik K. Großkinsky Birgit Piechulla
Birgit Piechulla