- 1Food Animal Health Research Program, Ohio Agricultural Research and Development Center, The Ohio State University, Wooster, OH, USA
- 2VPH-Biotec Global Consortium
- 3Faculty of Veterinary Medicine, Sokoine University of Agriculture, Morogoro, Tanzania
- 4Department of Veterinary Preventive Medicine, College of Veterinary Medicine, The Ohio State University, Columbus, OH, USA
Foodborne Campylobacter infections pose a serious threat to public health worldwide. However, the occurrence and characteristics of Campylobacter in food animals and products remain largely unknown in Tanzania. The objective of this study was to determine the prevalence, antibiotic resistance, and genetic profiles (sequence types, STs) of Campylobacter isolated from feces of pigs and dairy and beef cattle in Tanzania. Overall, 259 (~30%) of 864 samples were positive for Campylobacter spp, which were detected in 32.5, 35.4, and 19.6% of the pig, dairy, and beef cattle samples, respectively. Multiplex PCR analysis identified 64.5 and 29.3% of the Campylobacter isolates as C. coli and C. jejuni, respectively. The majority (91.9%) of the isolates from pig samples were identified as C. coli, while C. jejuni accounted for 65.5% of the isolates from cattle. Antimicrobial susceptibility testing using the disk diffusion assay and the broth microdilution method revealed resistance to: ampicillin (Amp) (70.3% and 75.7%, respectively), gentamicin (Gen) (1.8% and 12.6%), streptomycin (Str) (65.8 and 74.8%), erythromycin (Ery) (41.4 and 48.7%), tetracycline (Tet) (18.9 and 23.4%), and ciprofloxacin (Cip) (14.4 and 7.2%). Resistance to nalidixic acid (Nal) (39.6%), azithromycin (Azm) (13.5%), and chloramphenicol (Chl) (4.5%) was determined using the disk diffusion assay only, while resistance to tylosin (Tyl) (38.7%) was quantified using the broth microdilution method. Multilocus sequence typing of 111 Campylobacter isolates resulted in the identification of 48 STs (26 C. jejuni and 22 C. coli) of which seven were novel (six C. jejuni and one C. coli). Taken together, this study revealed the high prevalence, genetic diversity and antimicrobial resistance of Campylobacter in important food animals in Tanzania, which highlights the urgent need for the surveillance and control of Campylobacter in this country.
Introduction
Campylobacter spp. are among the most common etiological agents of foodborne bacterial gastroenteritis in humans worldwide, accounting for an estimated 500 million infections per year globally (Ruiz-Palacios, 2007; WHO, 2013). The number of reported cases of campylobacteriosis is high in developed countries (Scallan et al., 2011; EFSA and ECDC, 2014), while the disease remains under reported in developing countries due to the absence of regular surveillance programs (Coker et al., 2002). In Tanzania, Campylobacter has been reported to affect up to 20% of children under 5 years old (Jacob et al., 2011; Deogratias et al., 2014). Campylobacter is increasingly becoming a major problem in Sub-Sahara Africa where the number of infections is predicted to double by the year 2020 (Coker et al., 2002). Furthermore, deficiencies in food safety regulations and limited epidemiological data in many African countries, including Tanzania, hamper the assessment, surveillance, and control of Campylobacter infections. Therefore, in these countries, studies that address the occurrence and antimicrobial resistance of Campylobacter in food animals are of paramount importance.
Campylobacter is zoonotic and various food animals including poultry, pigs, and cattle are implicated as important reservoirs (Man, 2011; Sahin et al., 2015). Recent studies have shown that the contributions of non-poultry associated Campylobacter to human infections are considerable and warrant investigation (Ragimbeau et al., 2008; Wilson et al., 2008). For example, it was shown that cases of human Campylobacter infections can be attributed equally to cattle and poultry sources in certain countries (Wilson et al., 2008; de Haan et al., 2010). Studies from different countries reported that Campylobacter prevalence can range from 12 to 100% in dairy herds (Gilpin et al., 2008; Ragimbeau et al., 2008; Pradhan et al., 2009; Sanad et al., 2011), 5.4 to 83% in beef cattle (Salihu et al., 2009; Haruna et al., 2013), and 46 to 100% in pigs (Saenz et al., 2000; Pezzotti et al., 2003; Payot et al., 2004; Litrup et al., 2007; Mdegela et al., 2011). While Campylobacter jejuni can constitute the predominant species isolated from cattle (Ragimbeau et al., 2008; Sanad et al., 2011), Campylobacter coli is more frequently isolated from pigs (Saenz et al., 2000; Mdegela et al., 2011). In addition to potential contamination of milk and carcasses at slaughter, Campylobacter colonization of cattle and pigs pose a serious risk for contamination of surface and sub-surface water during disposal of abattoir effluents and animal slurries. This might further contribute to the transmission of Campylobacter to other food animals or directly to humans (Minihan et al., 2004; Devane et al., 2005; Garrett et al., 2007). Consequently, defining the role of cattle and pigs as reservoirs for these pathogens might be important for understanding the epidemiology of Campylobacter in Tanzania.
Most Campylobacter infections in humans are self-limiting and do not require antimicrobial therapy. However, in systemic infections or in immunocompromised individuals, erythromycin (Ery) and fluoroquinolones are used as the drugs of choice. However, studies have reported an increase in the resistance of Campylobacter to various antimicrobials, including the drugs of choice (Van Looveren et al., 2001; Chen et al., 2010; Cody et al., 2010; Rozynek et al., 2010). The rise in antimicrobial resistant Campylobacter has been linked to the use of antimicrobials in veterinary medicine and in farming practices (White et al., 2002; Zhu et al., 2006). While antimicrobial resistant Campylobacter has been reported worldwide, the situation might be more severe in developing countries where there is widespread and largely uncontrolled use of antimicrobials (Byarugaba, 2004; Kariuki, 2010). This is particularly important, because some of the resistant isolates have been suspected to spread from food animals to humans (Rozynek et al., 2010). Therefore, analysis of antimicrobial resistance of Campylobacter isolated from food animals in developing countries is needed to better manage cognate infections and mitigate emergence of antimicrobial resistant strains. Subsequently, in this study, we investigated the prevalence, antimicrobial resistance, and genetic diversity of Campylobacter isolated from pigs, dairy, and beef cattle from three different geographical regions in Tanzania.
Materials and Methods
Geographical Locations and Sample Collection
A cross-sectional sampling was conducted between April 2013 and March 2014. Samples were collected from pigs, dairy, and beef cattle from three geographically distinct regions of Tanzania, namely Arusha in Northern Tanzania: Iringa in Southwestern Tanzania, and Morogoro in Eastern Tanzania. These locations were strategically selected, because they are among the regions that hold the largest populations of farmed animals in Tanzania. A total of 864 samples: pig feces (n = 458), beef cattle feces (n = 214), and dairy cattle feces (n = 192) were collected from the three regions [Arusha (n = 189; 82 pig, 47 dairy, and 60 beef samples), Iringa (n = 150; 66 pig, 32 dairy, and 52 beef samples), and Morogoro (n = 525; 310 pig, 113 dairy, and 102 beef samples)]. Beef cattle feces samples (10 g) were randomly and aseptically collected from the colon during slaughter at the abattoirs. Similarly, 10 g of fresh pen-floor fecal samples were aseptically collected from dairy cattle and pigs on farms. Samples were placed on ice and immediately shipped to the laboratory for Campylobacter isolation. All samples were processed within 24–48 h after collection.
Isolation of Campylobacter from Fecal Samples
Campylobacter was isolated as described previously (Kashoma et al., 2014, 2015). Approximately 2 g of feces were suspended in 9 mL of maximum recovery diluent (MRD) (Neogen, USA). One milliliters of the suspension was added to 9 mL of Preston broth containing Campylobacter growth and selective supplements (SR0117E and SR0232; Oxoid, England). The suspensions were then incubated at 42°C for 48 h in airtight jars containing the Campy Pouch system (Becton Dickinson and Co., Maryland, USA) to generate microaerobic conditions. After incubation, 100 μL of the suspension was spread onto a modified charcoal cefoperazone deoxycholate agar (mCCDA) plate (Oxoid) containing a Campylobacter selective supplement (SR0155E, Oxoid) and incubated for 48 h at 42°C under microaerobic conditions. Three to five colonies suspected as Campylobacter were selected from each mCCDA plate and further purified using Muller-Hinton (MH; Difco, MD) agar plates containing a Campylobacter selective supplement (SR0117E, Oxoid). Pure cultures were stored at −80°C in MH broth supplemented with 30% glycerol (vol/vol) until further analysis.
Identification of Campylobacter Species Using PCR
For PCR analysis, bacterial DNA lysates were prepared from fresh pure Campylobacter cultures using the boiling method as previously described (Kashoma et al., 2014, 2015). In cases where no PCR products were detected, template DNA was prepared using QIAamp DNA Mini Kit (QIAGEN, Hilden, Germany) according to the manufacturer's instructions. Confirmation and speciation of putative Campylobacter was performed by multiplex-PCR (mPCR) as described previously (Linton et al., 1997; Denis et al., 1999; Yamazaki-Matsune et al., 2007). Isolates that were positive for the genus-specific PCR but negative for the C. coli and C. jejuni-specific PCR were designated as other thermophilic campylobacters (OTC). C. jejuni 81–176 (wild-type strain) and C. coli (ATCC 33559) were used as positive controls, while standard-grade laboratory water was used as a no template (negative) control.
Antimicrobial Susceptibility Analysis
Antimicrobial resistance analysis was performed on 111 Campylobacter isolates (42 C. jejuni and 69 C. coli) that were randomly selected to represent different animal hosts and geographical locations. The antimicrobials tested are representatives of the drugs used for humans and in the animal industry in Tanzania (Kashoma et al., 2015; Komba et al., 2015). The analysis was conducted using the Kirby-Bauer disk diffusion and the broth microdilution methods as described previously (Luber et al., 2003; Lehtopolku et al., 2012; Kashoma et al., 2015). Both tests were performed in accordance to the recommendations of the Clinical Laboratory Standards Institute (CLSI, 2012). The results were interpreted as susceptible, intermediately resistant, or resistant according to the CLSI (2012) or the ROSCO MIC for veterinary isolates (ROSCO, 2007) guidelines (Kashoma et al., 2015). Multi-drug Resistance (MDR) was defined as resistance to three or more antimicrobial agents (Hakanen et al., 2003).
In the Kirby-Bauer disk diffusion test, nine antimicrobial agents (Oxoid, UK) were tested at the following concentrations: 10 μg ampicillin (Amp), 5 μg ciprofloxacin (Cip), 15 μg Ery, 30 μg nalidixic acid (Nal), 10 μg streptomycin (Str), 30 μg tetracycline (Tet), 15 μg azithromycin (Azm), 10 μg gentamicin (Gen), and 30 μg chloramphenicol (Chl). The antimicrobial discs were placed on the surface of culture plates and the diameter of the zone of inhibition was measured after 24 h of microaerobic incubation at 42°C.
For the broth microdilution test and the determination of the minimum inhibitory concentration (MIC), 96-well plates containing two-fold serial dilutions of the antimicrobial agents were used as described previously (Ge et al., 2013; Kashoma et al., 2015). The antimicrobial agents tested included Amp, Cip, Ery, Gen, tylosin (Tyl), Str, and Tet. MIC values were defined as the lowest concentration of an antimicrobial agent that produced no visible growth and the results were confirmed spectrophotometrically using a microplate reader (Multiskan® Spectrum, Thermo Scientific, USA) (Ge et al., 2013). In both assays, C. jejuni 81–176 and C. coli (ATCC 33559) were used as positive control strains.
Multilocus Sequence Typing (MLST) Analysis
In order to determine the genetic diversity of the Campylobacter isolates and their relationship to existing clonal complexes (CC) and sequence types (STs), 111 isolates (42 C. jejuni and 69 C. coli) which were also tested for antimicrobial resistance were analyzed by MLST as described previously (Dingle et al., 2001; Sanad et al., 2011). Briefly, loci from seven housekeeping genes (aspA, glnA, gltA, glyA, pgm, tkt, and uncA) were amplified using PCR and specific primers (Dingle et al., 2001). The size of the amplicons was confirmed using agarose gel electrophoresis. PCR products were then treated with ExoSAP (Affymetrix Inc., USA), sequenced in both directions. The forward and reverse sequences were aligned using ClustalW (www.ebi.ac.uk/clustalw), allele profiles were determined by BLAST analysis using the single-locus query function, while STs were assigned using the allele profile query function available in the MLST Campylobacter database (http://pubmlst.org/campylobacter). STs were then traced to their respective CC using BURST (http://pubmlst.org/).
Statistical Analysis
The prevalence and antimicrobial resistance of Campylobacter from pigs, dairy, and beef cattle in all three regions were compared using the Chi-squared (χ2) test. A value of P < 0.05 was considered statistically significant. Agreement between the two antimicrobial resistance tests was determined using the Kappa statistic (Luber et al., 2003). A Kappa value of 100% indicates total agreement between the classifiers.
Results
The Prevalence of Campylobacter in Feces Sampled from Cattle and Pigs
Campylobacter were detected in 259 (30%) of 864 fecal samples and were distributed as follows: a total of 149 (32.5%), 68 (35.4%), and 42 (19.6%) isolates were retrieved from pig, dairy, and beef cattle fecal samples, respectively. C. jejuni and C. coli constituted the majority of the isolates and were detected in 76 (8.8%) and 166 (19.3%) of the samples, respectively (Table 1). There was no significant difference (P>0.05) in the total number of C. jejuni isolated from fecal samples obtained from either dairy (21.9%) or the beef cattle (14.0%). Significantly (P < 0.0001) more C. coli were isolated from pigs (29.9%) in comparison to cattle (7.4%), while C. jejuni was significantly higher (P < 0.0001) in cattle (17.7%) than in pig (0.9%) samples. Furthermore, the occurrence of Campylobacter in animals varied according to geographic location of sampling sites. Specifically, Campylobacter were retrieved from 34.1% of all fecal samples collected from Morogoro, which was significantly higher (P < 0.05) than those from Arusha (27.5%) and Iringa (18.7%), respectively (Table 1). Moreover, total C. coli prevalence was significantly higher (P < 0.05) in Morogoro (5.9%) than in Arusha (1.7%) or Iringa (1.9%). The combined prevalence of C. jejuni in beef and dairy cattle feces from Morogoro (23.2%; 50 C. jejuni retrieved from 215 fecal samples) was significantly higher (P < 0.05) in comparison to that from Arusha (12.2%) and Iringa (10.7%), respectively. However, no significant difference (P>0.05) in the prevalence of C. jejuni in beef and dairy cattle feces was observed between Arusha and Iringa.
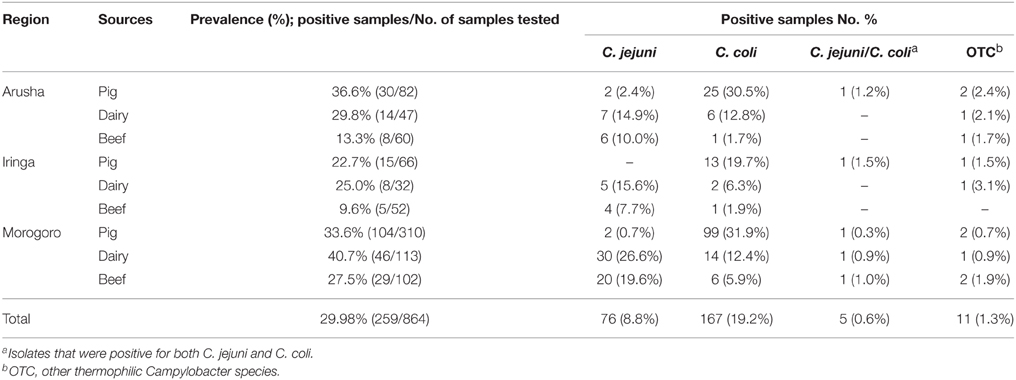
Table 1. The prevalence of Campylobacter species in pig, dairy, and beef cattle feces collected from three geographical locations in Tanzania.
Antimicrobial Susceptibility of the C. jejuni and C. coli Isolates
Analysis of the Kirby-Bauer disk diffusion assay showed that 106 of the 111 isolates (95.5%) were resistant to one or more antimicrobial agents, whereas five (4.5%) isolates were pan-susceptible to all antimicrobials tested (Table 2). Twenty-one isolates (18.9%; six C. jejuni and 15 C. coli) were resistant to a single antimicrobial agent and 22 isolates (19.8%; 10 C. jejuni and 12 C. coli) showed resistance to two antimicrobial agents (Table 2). Sixty-three (56.8%) of all isolates (24 C. jejuni and 39 C. coli) were classified as MDR. Of the MDR isolates, 38 (60.3%) were from pig, 15 (23.8%) from dairy, and 10 (15.9%) from beef cattle feces. Two C. jejuni isolates recovered from dairy cattle were resistant to Gen, whereas 9.0% of all isolates (four C. jejuni and six C. coli) were resistant to Chl (Table 2). Approximately 14.4% (six C. jejuni and 10 C. coli) and 13.5% (six C. jejuni and nine C. coli) of the isolates were resistant to Cip and Azm, respectively (Tables 2, 4). Twenty-one (18.9%) isolates were shown to be resistant to Tet, 46 (41.4%) to Ery, and 44 (39.6%) to Nal. In addition, 65.8% (34 C. jejuni and 39 C. coli) and 70.3% (28 C. jejuni and 50 C. coli) of isolates were resistant to Str and Amp, respectively (Tables 2, 4). While resistance to Str was significantly higher (P < 0.05) in isolates recovered from cattle in comparison to those from pigs, there were no significant differences (P>0.05) in resistance associated with the remaining antimicrobials.
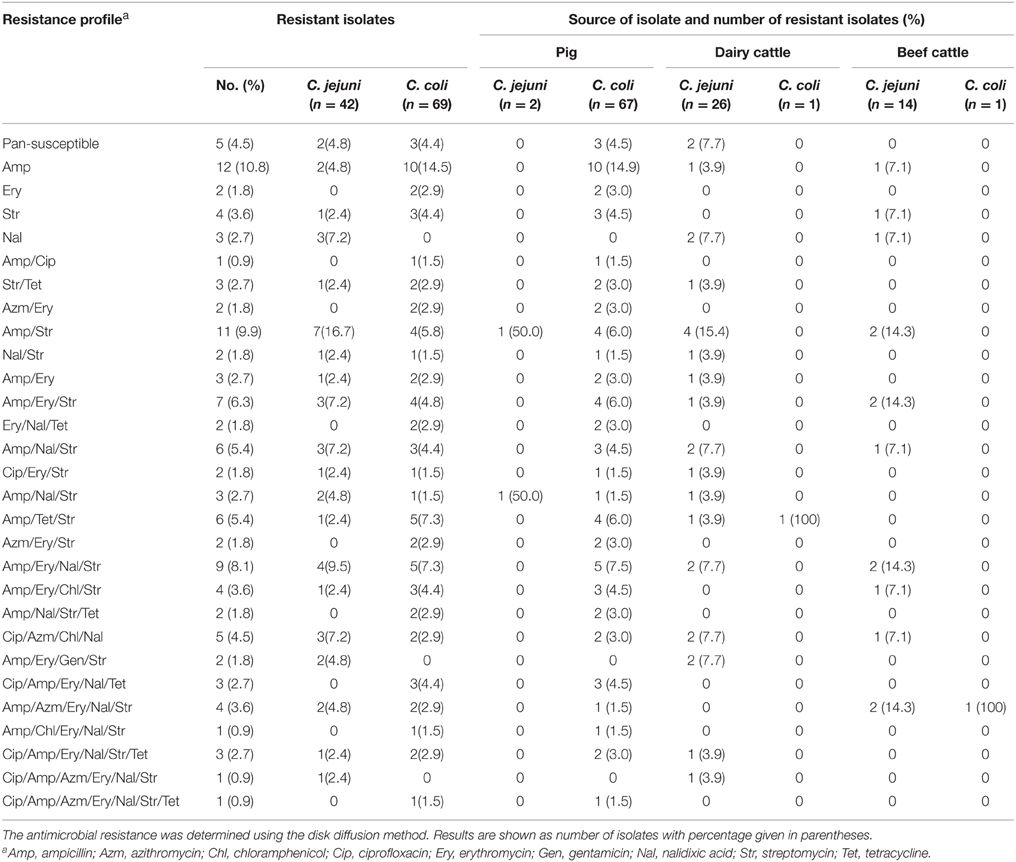
Table 2. Antimicrobial resistance of C. jejuni and C. coli isolated from pig, dairy, and beef cattle samples.
The analysis of antimicrobial resistance by the broth microdilution method revealed that the majority (94.6%) were resistant to at least one antimicrobial agent, while six (5.4%) isolates were pan-susceptible. The Campylobacter isolates displayed resistance most frequently to Amp (75.7%) and less frequently to Cip (7.2%) (Tables 3, 4). In comparison to C. jejuni, significantly more (P < 0.05) C. coli isolates displayed resistance to Str and Amp regardless of the source of the isolates. Seventeen of 42 C. jejuni (40.5%) isolates were resistant to three or more antimicrobials, while, 69.6% (48/69) of C. coli isolates were resistant to three or more antimicrobials. Approximately 2.4 and 28.6% of C. jejuni isolates were resistant to Cip and Gen, respectively (Table 3). Additionally, 10.2% and 2.9% of C. coli strains were resistant to Cip and Gen, respectively. While the number of C. coli isolates resistant to Gen was significantly different (P < 0.01) in comparison to that of C. jejuni, there was no significant difference with respect to resistance to Cip. The number of C. jejuni (42.9%) isolates resistant to Tyl was not significantly different in comparison to that of C. coli (36.2%) (P>0.05). However, the number of C. jejuni (19.1%) isolates resistant to Ery was significantly different in comparison to that of C. coli (66.7%) (P < 0.0007).
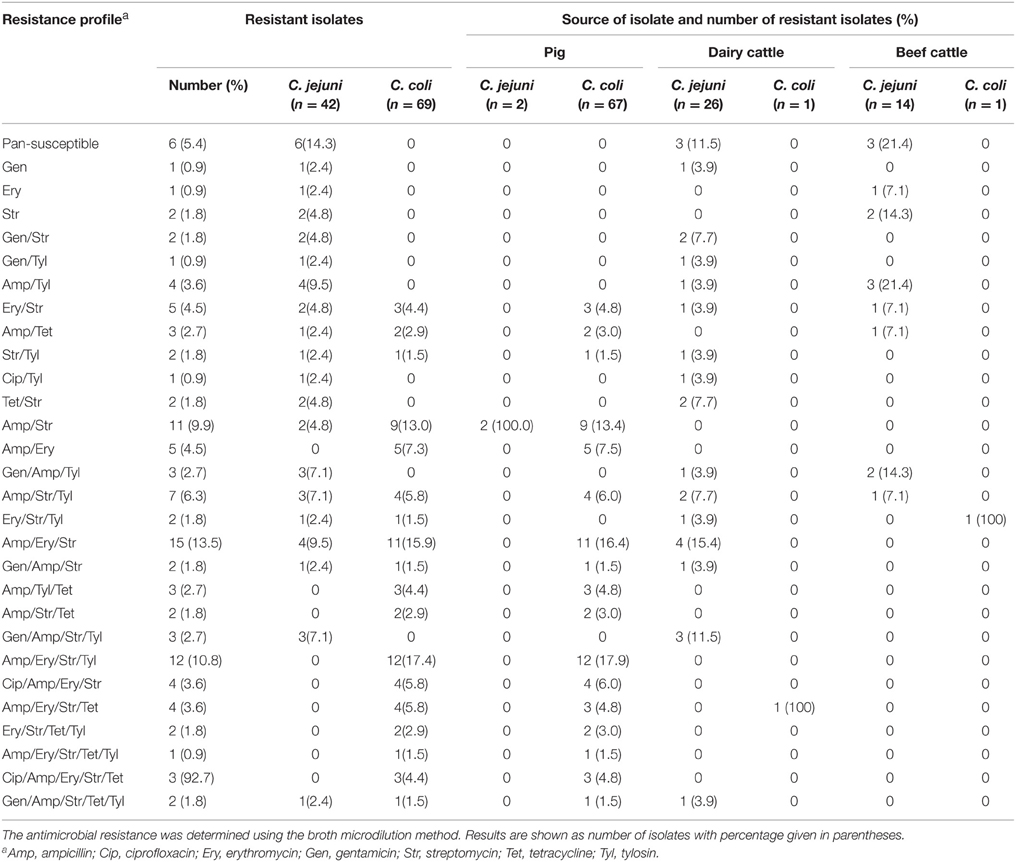
Table 3. Antimicrobial resistance of C. jejuni and C. coli isolated from pig, dairy, and beef cattle samples.
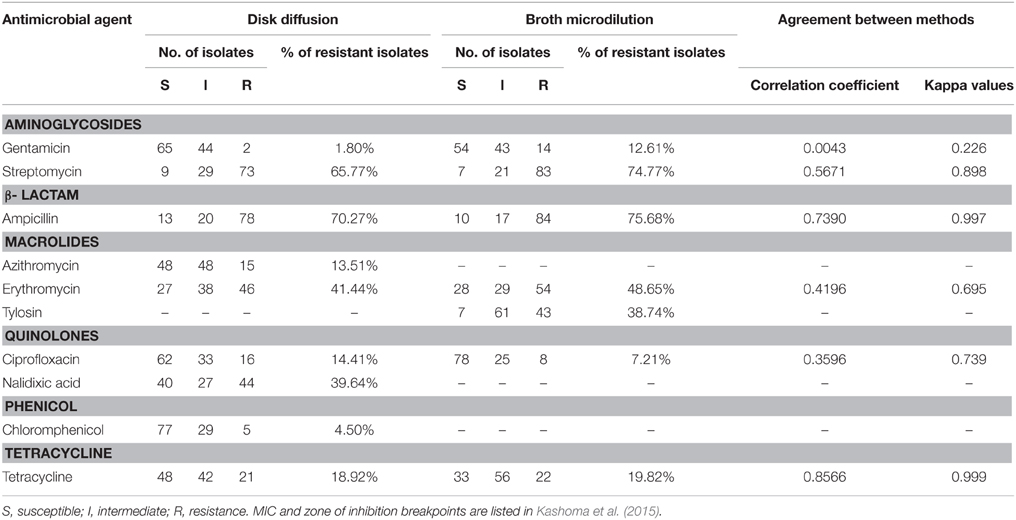
Table 4. Comparison of antimicrobial resistance of Campylobacter spp. identified by disk diffusion and broth microdilution methods.
Using the broth microdilution and disk diffusion methods, similar results (P>0.05) were obtained for five out of six antimicrobial agents (Cip, Str, Amp, Ery, Tet) tested (Table 4). The correlation coefficients between the results obtained from the two methods were 0.0043 for Gen, 0.3596 for Cip, 0.4196 for Ery, 0.5671 for Str, 0.8566 for Tet, and 0.7390 for Amp. Additionally analysis using the Kappa statistics showed that the results obtained using the two tests were mostly in high agreement. The Kappa values were 0.739 for Cip, 0.695 for Ery, 0.898 for Str, 0.977 for Amp, and 0.999 for Tet. Lower agreement was only found for Gen (Kappa = 0.226) (Table 4).
MLST Analysis of C. jejuni and C. coli Isolates
MLST was performed to determine the genetic diversity and clonal origins of the Campylobacter isolates that were tested for antimicrobial resistance. A total of 48 different STs were identified for the 111 Campylobacter isolates. The C. jejuni isolates (n = 42) were classified into 26 unique STs of which six (STs 7690, 7697, 7698, 7699, 7700, and 7701) were novel. These novel STs were distributed as follows: three were identified from dairy cattle, two from beef, and one ST from pigs. Nineteen C. jejuni STs were assigned to a previously described clonal complex (CC 828), whereas seven STs (including the six new STs) belonged to an undefined clonal complex (Table 5). The C. coli isolates (n = 69) were classified into 22 STs (Table 5). Two C. coli isolates were assigned to a new ST (ST 7683). Fifty two C. coli isolates (18 STs) were assigned to a previously described clonal complex (CC 828), while 17 isolates (four STs) belonged to an undefined clonal complex. Overall, these findings imply the occurrence of diverse strains, with majority of STs appearing to not overlap between sources and geographical locations. Only the C. coli ST 2713 was identified in two geographical locations (Arusha and Iringa), whereas ST 4309 occurred in pig and dairy samples.
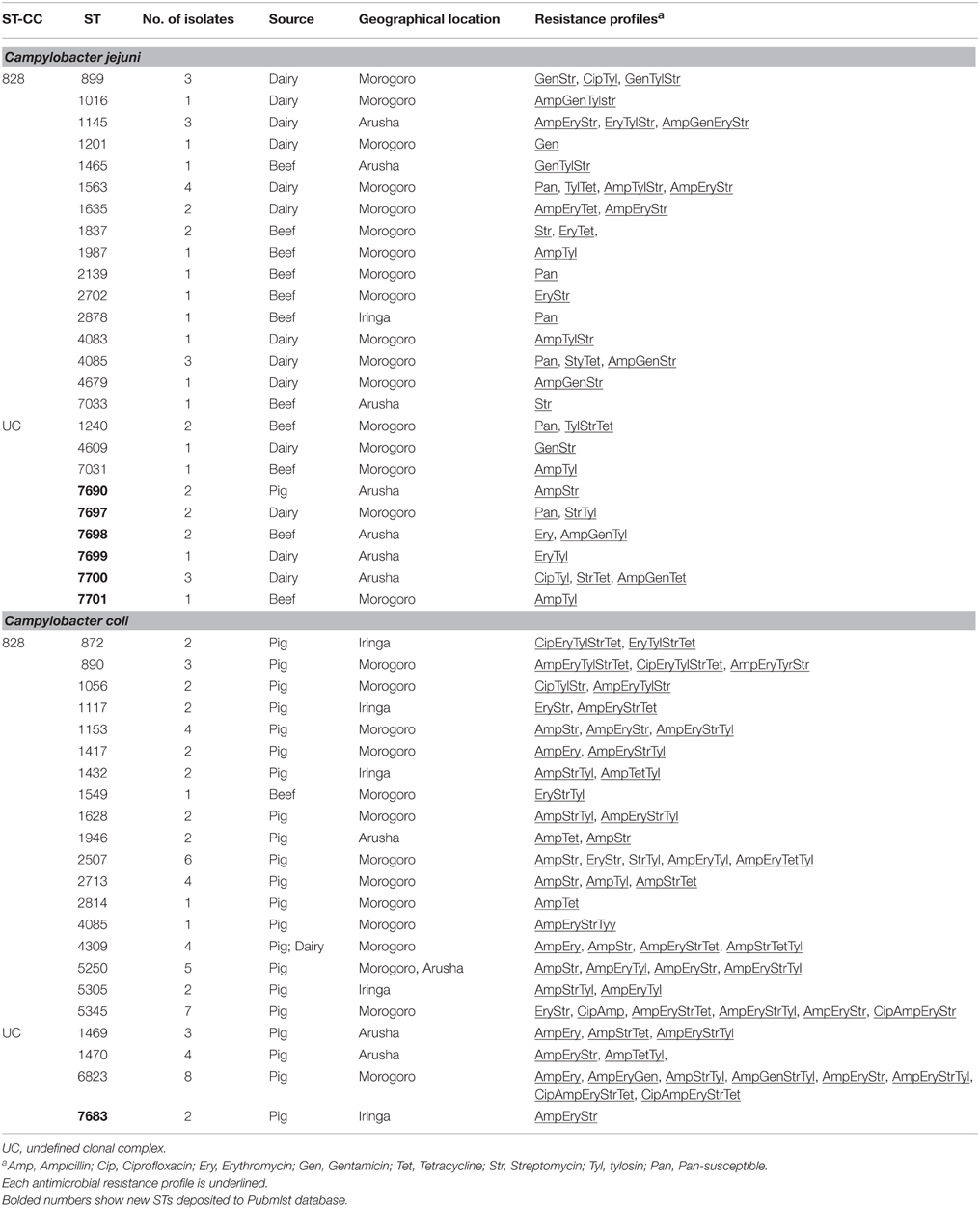
Table 5. Distribution of clonal complexes, sequence types and antimicrobial resistance profiles (Broth microdilution assay) of C. jejuni and C. coli from pig, dairy, and beef cattle in Tanzania.
Association of Sequence Types (STs) with Antimicrobial Resistance Pattern
The majority of C. jejuni STs (21 STs; 37 isolates) were resistant to multiple antimicrobials, whereas two STs (ST 2139 and ST 2878) were susceptible to all antimicrobials tested by the broth microdilution method (Table 5). Almost all the C. jejuni isolates that belonged to undefined CC were resistant to either one or two antimicrobials. In general, C. coli isolates showed more diverse antimicrobial resistance profiles in comparison to C. jejuni isolates (Table 5). With the exception of ST 2814, which was represented by one isolate that was resistant to two antimicrobial agents, the majority of STs included isolates with different resistance profiles despite belonging to the same ST (Table 5). Mostly, C. coli STs were significantly associated with MDR (P < 0.05) with the majority (86.7%) of isolates in the undefined complex being resistant to three or more antimicrobials.
Discussion
We assessed the prevalence, antimicrobial resistance and genetic diversity of Campylobacter isolated from pigs, dairy, and beef cattle in Tanzania, where animal husbandry is a very important agricultural activity (MLDF, 2014). In this study, the prevalence of Campylobacter in dairy and beef cattle samples was similar to the findings reported in other countries (Gilpin et al., 2008; Ragimbeau et al., 2008; Pradhan et al., 2009; Salihu et al., 2009; Sanad et al., 2011). Furthermore, the overall recovery rate of Campylobacter from cattle was significantly different among the three sampling location/regions. While the prevalence of C. coli in the pig samples in this study was in agreement with previous reports (Saenz et al., 2000; Jensen et al., 2006), it was also slightly lower than those reported in many other studies (Saenz et al., 2000; Pezzotti et al., 2003; Payot et al., 2004; Mdegela et al., 2011). The differences in C. jejuni and/or C. coli prevalence within Tanzania and between countries may be due to several factors, including farming and slaughtering practices, geographical locations, or other risk factors, including the concentration of the farms in each location and their proximity to other livestock such as poultry (Moore et al., 2002; Humphrey et al., 2007; Sahin et al., 2015).
Both the agar disk diffusion and the broth microdilution methods have commonly been used to determine antimicrobial resistance in Campylobacter (Miflin et al., 2007; Senok et al., 2007). Because of the importance of the implications associated with reporting antimicrobial resistance in human food-borne pathogens, in this study two methods were used in order to generate robust analysis and reduce methodological bias. Our results showed an agreement between these two methods for most of the antimicrobials tested (Table 4), suggesting that either method was generally adequate for analyzing antimicrobial resistance. However, in certain cases such as Gen where the agreement between the two methods was low, multiple susceptibility testing approaches must be adopted and results must be interpreted cautiously to account for methodological variability. The comparison between the two methods is also particularly important for facilitating research conducted in resource limited countries like Tanzania, because it allows the selection of a suitable method for antimicrobial resistance analysis without unnecessary expenditures.
A considerable number of Campylobacter isolates in this study was resistant to macrolides (Ery, Tyl, and Azm) (Tables 2, 3). Macrolides such as Tyl are extensively used in Tanzania as therapeutic agents for treatment of cattle respiratory conditions (Kashoma et al., 2015). The use of Tyl in animals for the purpose of either treatment or growth promotion contributes to the selection of resistant Campylobacter strains to macrolides including Ery (Juntunen et al., 2010). These observations suggest a possible association between the heavy use of Tyl in Tanzania and the increase in the resistance Campylobacter to Tyl and Ery observed in this study. Furthermore, the high resistance to macrolides (Ery) in Campylobacter isolated from humans in Tanzania (Komba et al., 2015) highlights the need for understanding the impact of the use of antimicrobials in animal agriculture on the rise of resistant pathogens in food animals and humans. This further emphasizes the need for Campylobacter surveillance and control studies in Tanzania.
In this study, a relatively high level of resistance to Nal (47.6% C. jejuni and 34.8% C. coli) was observed, while the number of Cip resistant Campylobacter isolates (11.7% and 7.2% using the disk diffusion and broth microdilution methods, respectively) was relatively low (Tables 2, 4). The wide-spread resistance to Nal corroborated reports on Campylobacter from different food animals/products in other countries (Taremi et al., 2006; Bostan et al., 2009; Dabiri et al., 2014), while Cip resistance was generally lower than elsewhere (Taremi et al., 2006; Wieczorek and Osek, 2013). Furthermore, in a previous study on beef carcasses and raw milk in Tanzania (Kashoma et al., 2015), it was observed that C. jejuni (65.7%) and C. coli (63.2%) isolates were resistant to Nal, while 9.3–11.8% were resistant to Cip. The resistance to Cip in isolates originating from food animals/products in Tanzania is of concern, because this antimicrobial is used for treatment of human campylobacteriosis. Currently, the factors that are promoting Cip resistance in Tanzanian food-associated isolates are not clear, especially because it is known that this antimicrobial is not commonly used in animal agriculture in Tanzania. However, enrofloxacin is licensed for therapeutic use in poultry against colibacilliosis, pasteurellosis, and mycoplasmosis (Mubito et al., 2014), which may result in the selection of resistance of Campylobacter to fluoroquinolones. These resistant isolates might be transmitted to other food animals that are in the proximity of poultry via a variety of vehicles, including common farm workers and/or flies or other critters (McDermott et al., 2002; Van Boven et al., 2003; Stapleton et al., 2010). Furthermore, it is possible that Cip resistant isolates might have originated from humans where this antimicrobial is commonly used. This might be supported by a recent report showing that a relatively high percentage (22.1%) of human Campylobacter isolates were resistant to Cip in Tanzania (Komba et al., 2015). Regardless, the determination of factors that select for Cip resistant isolates will require further investigation.
In this study, a range of resistance (low to high number of isolates) was observed for different antimicrobials. Specifically, a relatively low number of isolates were resistant to Gen (1.8–12.6% of isolates depending on the testing method) and Chl (6.3%), respectively. Generally, Chl and Gen resistance in Campylobacter is known to be low (Fallon et al., 2003; Kassa et al., 2007). Furthermore, a previous study in Tanzania showed that 13% and 11.8% of the Campylobacter isolated from beef carcasses and raw milk were resistant to Chl and Gen, respectively (Kashoma et al., 2015). Since Tets are widely used in humans and as therapeutics or feed additives in livestock and poultry in Tanzania, a high number of Tet resistant isolates was expected. For example, Komba et al. (2015) reported 76.5% and 57.1% of human-associated C. jejuni and C. coli were resistant to Tet in Tanzania. However, a moderate resistance to Tet (19.8% of the isolates) was observed in this study. The reasons behind this are currently not clear and require further analysis. In contrast, Campylobacter can inherently display resistance to β-lactams (including Amp) (Engberg et al., 2006; Li et al., 2007). Consequently, the high resistance to Amp observed in this study was not surprising.
In recent years multidrug resistant Campylobacter strains have been increasingly reported worldwide, which is now recognized as a major emerging public health concern. The numbers of multidrug resistant isolates (40% C. jejuni and 69.9% C. coli) are comparable to those reported in other countries (Van Looveren et al., 2001; Pezzotti et al., 2003; Wieczorek and Osek, 2013). Furthermore, analysis of human-associated Campylobacter in Tanzania showed that 77.9% of the isolates were resistant to more than six of the tested antimicrobials, while 19.9% were resistant to all tested antimicrobials (n = 12) (Komba et al., 2015). While the contribution of food animal-associated isolates to the MDR in human isolates is currently unknown, this is a point of serious concern that suggests that Tanzania, like other countries, has to devise stringent control and regulatory measures to reduce MDR isolates in the food chain.
MLST analysis classified the tested isolates into 48 STs. Almost 71.2% of the isolates were assigned to known CC, while 28.8% could not be traced to known lineages. These findings highlight the diversity of Campylobacter genotypes and suggest that certain food-animal associated isolates might have evolved and adapted to Tanzanian farm animals and/or farming practices. In this study, the majority of C. coli and C. jejuni isolates belonged to ST 828 clonal complex, which is associated mainly with isolates from agricultural and environmental sources and human clinical cases (Sheppard et al., 2010). Also, researchers have reported the presence of progenitor strains of the ST 828 complex in human, swine, poultry, and cattle from different parts of the world (Dingle et al., 2005; Miller et al., 2006; Sanad et al., 2011; Kashoma et al., 2014). This is important, because the contributions of various possible sources of infection, including food animals, to the burden of human campylobacteriosis in Tanzania is not clearly defined and require further investigations.
Conclusions
In the present study, a high prevalence and genotypic diversity of Campylobacter from pigs, dairy, and beef cattle in Tanzania was observed. The majority of the Campylobacter isolates examined were resistant to multiple antimicrobials, which was confirmed using two different methods Therefore, prudent use of antimicrobials in veterinary/farming practices remains essential to reduce the pool of antimicrobial resistant pathogens that might impact human health. Collectively, this study highlights the need for continuous efforts to control Campylobacter colonization in farm animals in order to limit the transmission of these pathogens to humans.
Author Contributions
GR, RK, and IPK conceived the study. IPK, IIK, AK, and BK performed the experiments. IPK, IIK, WG, RK and GR analyzed the data. IPK, IIK, and GR wrote the manuscript. All authors read and approved the final manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This work was supported by the WHO Advisory Group on Integrated Surveillance of Antimicrobial Resistance (AGISAR), the NIH Fogarty fellowship program on Molecular Epidemiology and Key Issues in Food-borne Pathogens in Eastern Africa, the United States Agency for International Development (USAID) through the Feed the Future Tanzania-based Innovative Agricultural Research Initiative (iAGRI) and funds from Ohio Agricultural Research and Development Center (OARDC), The Ohio State University.
References
Bostan, K., Aydin, A., and Ang, M. K. (2009). Prevalence and antibiotic susceptibility of thermophilic Campylobacter species on beef, mutton, and chicken carcasses in Istanbul, Turkey. Microb. Drug Resist. 15, 143–149. doi: 10.1089/mdr.2009.0894
Byarugaba, D. K. (2004). A view on antmicrobial resistance in developing countries and responsible risk factors. Int. J. Antimicrob. Agents 24, 105–110. doi: 10.1016/j.ijantimicag.2004.02.015
Chen, X., Naren, G. W., Wu, C. M., Wang, Y., Dai, L., Xia, L. N., et al. (2010). Prevalence and antimicrobial resistance of Campylobacter isolates in broilers from China. Vet. Microbiol. 144, 133–139. doi: 10.1016/j.vetmic.2009.12.035
Clinical Laboratory Standards Institute (CLSI) (2012). Performance Standards for Antimicrobial Susceptibility Testing; Twenty-second Informational Supplement M11-S22. Wayne, PA: Clinical and Laboratory Standards Institute.
Cody, A. J., Colles, F. M., Sheppard, S. K., and Maiden, M. C. J. (2010). Where does Campylobacter come from? A molecular odyssey. Adv. Exp. Med. Biol. 659, 47–56. doi: 10.1007/978-1-4419-0981-7_4
Coker, A. O., Isokpehi, R. D., Thomas, B. N., Amisu, K. O., and Obi, C. L. (2002). Human campylobacteriosis in developing countries. Emerg. Infect. Dis. 8, 237–243. doi: 10.3201/eid0803.010233
Dabiri, H., Aghamohammad, S., Goudarzi, H., Noori, M., Hedayati, M. A., and Ghoreyshiamiri, S. M. (2014). Prevalence and antibiotic susceptibility of Campylobacter species isolated from chicken and beef meat. Int. J. Enteric Pathog. 2, e17087. doi: 10.17795/ijep2322-5866
de Haan, C. P., Kivisto, R., and Hakkinen, M. (2010). Multilocus sequence types of Finnish bovine Campylobacter jejuni isolates and their attribution to human infections. BMC Microbiol. 10:200. doi: 10.1186/1471-2180-10-200
Denis, M., Soumet, C., Rivoal, K., Ermel, G., Blivet, D., Salvat, G., et al. (1999). Development of a m-PCR for simultaneous identification of Campylobacter jejuni and C. coli. Lett. Appl. Microbiol. 29, 406–410. doi: 10.1046/j.1472-765X.1999.00658.x
Deogratias, A., Mushi, M. F., Paterno, L., Tappe, D., Seni, J., Kabymera, R., et al. (2014). Prevalence and determinants of Campylobacter infection among under five children with acute watery diarrhea in Mwanza, North Tanzania. Arch. Public Health 72:17. doi: 10.1186/2049-3258-72-17
Devane, M. L., Nicol, C., Ball, A., Klena, J. D., Scholes, P., Hudson, J. A., et al. (2005). The occurrence of Campylobacter subtypes in environmental reservoirs and potential transmission routes. J. Appl. Microbiol. 98, 980–990. doi: 10.1111/j.1365-2672.2005.02541.x
Dingle, K. E., Colles, F. M., Falush, D., and Maiden, M. C. J. (2005). Sequence typing and comparison of population biology of Campylobacter coli and Campylobacter jejuni. J. Clin. Microbiol. 43, 340–347. doi: 10.1128/JCM.43.1.340-347.2005
Dingle, K. E., Colles, F. M., Wareing, D. R. A., Ure, R., and Fox, A. J. (2001). Multilocus sequence typing system for Campylobacter jejuni. J. Clin. Microbiol. 39, 14–23. doi: 10.1128/JCM.39.1.14-23.2001
Engberg, J., Keelan, M., Gerner-Smidt, P., and Taylor, D. E. (2006). “Chapter 16: Antimicrobial resistance in Campylobacter,” in Antimicrobial Resistance in Bacteria of Animal Origin, ed F. M. Aarestrup (Washington, DC: ASM Press), 269–292.
European Food Safety Authority (EFSA) European Centre for Disease Prevention Control (ECDC) (2014). The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2012. EFSA J. 12:3547. doi: 10.2903/j.efsa.2014.3547
Fallon, R., O'sullivan, N., Maher, M., and Carroll, C. (2003). Antimicrobial resistance of Campylobacter jejuni and Campylobacter coli isolates from broiler chickens isolated at an Irish poultry processing plant. Lett. Appl. Microbiol. 36, 277–281. doi: 10.1046/j.1472-765X.2003.01308.x
Garrett, N., Devane, M. L., Hudson, J. A., Nicol, C., Ball, A., Klena, J. D., et al. (2007). Statistical comparison of Campylobacter jejuni subtypes from human cases and environmental sources. J. Appl. Microbiol. 103, 2113–2121. doi: 10.1111/j.1365-2672.2007.03437.x
Ge, B., Wang, F., Sjölund-Karlsson, M., and McDermott, P. F. (2013). Antimicrobial resistance in Campylobacter. Susceptibility testing methods and resistance trends. J. Microbiol. Methods 95, 57–67. doi: 10.1016/j.mimet.2013.06.021
Gilpin, B. J., Thorrold, B., Scholes, P., Longhurst, R. D., Devane, M., Nicol, C., et al. (2008). Comparison of Campylobacter jejuni genotypes from dairy cattle and human sources from the Matamata-Piako District of New Zealand. J. Appl. Microbiol. 105, 1354–1360. doi: 10.1111/j.1365-2672.2008.03863.x
Hakanen, A. J., Lehtopolku, M., Siitonen, A., Huovinen, P., and Kotilainen, P. (2003). Multidrug resistance in Campylobacter jejuni strains collected from Finnish patients during 1995-2000. J. Antimicrob. Chemother. 52, 1035–1039. doi: 10.1093/jac/dkg489
Haruna, M., Sasaki, Y., Murakami, M., Mori, T., Asai, T., Ito, K., et al. (2013). Prevalence and antimicrobial resistance of Campylobacter isolates from beef cattle and pigs in Japan. J. Vet. Med. Sci. 75, 625–628. doi: 10.1292/jvms.12-0432
Humphrey, T., O'Brien, S., and Madsen, M. (2007). Campylobacters as zoonotic pathogens: a food production perspective. Int. J. Food. Microbiol. 117, 237–257. doi: 10.1016/j.ijfoodmicro.2007.01.006
Jacob, P., Mdegela, R. H., and Nonga, H. E. (2011). Comparison of Cape Town and Skirrow's Campylobacter isolation protocols in humans and broilers in Morogoro, Tanzania. Trop. Anim. Health Prod. 43, 1007–1013. doi: 10.1007/s11250-011-9799-z
Jensen, A. N., Dalsgaard, A., Baggesen, D. L., and Nielsen, E. M. (2006). The occurrence and characterization of Campylobacter jejuni and C. coli in organic pigs and their outdoor environment. Vet. Microbiol. 116, 96–105. doi: 10.1016/j.vetmic.2006.03.006
Juntunen, P., Heiska, H., Olkkola, S., Myllyniemi, A. L., and Hänninen, M. L. (2010). Antimicrobial resistance in Campylobacter coli selected by tylosin treatment at a pig farm. Vet. Microbiol. 146, 90–97. doi: 10.1016/j.vetmic.2010.04.027
Kariuki, S. (2010). “Chapter 11: Antimicrobial resistance in enteric pathogens in developing countries,” in Antimicrobial Resistance in Developing Countries, eds A. J. Sosa, D. K. Byarugaba, C. F. Amábile-Cuevas, P. Hsueh, S. Kariuki, and I. N. Okeke (New York, NY: Springer Publishing Co. press) 177–197.
Kashoma, I. P. B., Kassem, I. I., John, J., Kessy, B., Gebreyes, W., Kazwala, R. R., et al. (2015). Prevalence and antimicrobial resistance of campylobacter isolated from dressed beef carcasses and raw milk in Tanzania. Microb. Drug Resist. doi: 10.1089/mdr.2015.0079. [Epub ahead of print].
Kashoma, I. P. B., Kumar, A., Sanad, Y. M., Gebreyes, W., Kazwala, R. R., Garabed, R., et al. (2014). Phenotypic and genotypic diversity of thermophilic Campylobacter spp. in commercial Turkey flocks: a longitudinal study. Foodborne Pathog. Dis. 11, 850–860. doi: 10.1089/fpd.2014.1794
Kassa, T., Gebre-Selassie, S., and Asrat, D. (2007). Antimicrobial susceptibility patterns of thermotolerant Campylobacter strains isolated from food animals in Ethiopia. Vet. Microbiol. 119, 82–87. doi: 10.1016/j.vetmic.2006.08.011
Komba, E. V. G., Mdegela, R. H. P., Msoffe, L. M., Nielsen, L. N., and Ingmer, H. (2015). Prevalence, antimicrobial resistance and risk factors for thermophilic Campylobacter infections in symptomatic and asymptomatic humans in Tanzania. Zoonoses Public Health. 62, 557–568. doi: 10.1111/zph.12185
Lehtopolku, M., Kotilainen, P., Puukka, P., Nakari, U., Siitonen, A., Eerola, E., et al. (2012). Inaccuracy of the disk diffusion method compared with the agar dilution method for susceptibility testing of Campylobacter spp. J. Clin. Microbiol. 50, 52–56. doi: 10.1128/JCM.01090-11
Li, X. Z., Mehrotra, M., Ghimire, S., and Adewoye, L. (2007). β-lactam resistance and β-lactamases in bacteria of animal origin. Vet. Microbiol. 121, 197–214. doi: 10.1016/j.vetmic.2007.01.015
Linton, D., Lawson, A. J., Owen, R. J., and Stanley, J. (1997). PCR detection, identification to species level, and fingerprinting of Campylobacter jejuni and Campylobacter coli direct from diarrheic samples. J. Clin. Microbiol. 35, 2568–2572.
Litrup, E., Torpdahl, M., and Nielsen, E. M. (2007). Multilocus sequence typing performed on Campylobacter coli isolates from humans, broilers, pigs and cattle originating in Denmark. J. Appl. Microbiol. 103, 210–218. doi: 10.1111/j.1365-2672.2006.03214.x
Luber, P., Bartelt, E., Genschow, E., Wagner, J., and Hahn, H. (2003). Comparison of broth microdilution, E test, and Agar Dilution methods for antibiotic susceptibility testing of Campylobacter jejuni and Campylobacter coli. J. Clin. Microbiol. 41, 1062–1068. doi: 10.1128/JCM.41.3.1062-1068.2003
Man, S. M. (2011). The clinical importance of emerging Campylobacter species. Nat. Rev. Gastroenterol. Hepatol. 8, 669–685. doi: 10.1038/nrgastro.2011.191
McDermott, P. F., Bodeis, S. M., English, L. L., White, D. G., Walker, R. D., Zhao, S., et al. (2002). Ciprofloxacin resistance in Campylobacter jejuni evolves rapidly in chickens treated with fluoroquinolones. J. Infect. Dis. 185, 837–840. doi: 10.1086/339195
Mdegela, R. H., Laurence, K., Jacob, P., and Nonga, H. E. (2011). Occurrences of thermophilic Campylobacter in pigs slaughtered at Morogoro slaughter slabs, Tanzania. Trop. Anim. Health Prod. 43, 83–87. doi: 10.1007/s11250-010-9657-4
Miflin, J. K., Templeton, J. M., and Blackall, P. J. (2007). Antibiotic resistance in Campylobacter jejuni and Campylobacter coli isolated from poultry in the South-East Queensland region. J. Antimicrob. Chemother. 59, 775–778. doi: 10.1093/jac/dkm024
Miller, W. G., Englen, M. D., Kathariou, S., Wesley, I. V., Wang, G., Pittenger-Alley, L., et al. (2006). Identification of host-associated alleles by multilocus sequence typing of Campylobacter coli strains from food animals. Microbiology 152, 245–255. doi: 10.1099/mic.0.28348-0
Minihan, D., Whyte, P., O'Mahony, M., Fanning, S., McGill, K., and Collins, J. D. (2004). Campylobacter spp. in Irish feedlot cattle: a longitudinal study involving pre-harvest and harvest phases of the food chain. J. Vet. Med. B 51, 28–33. doi: 10.1046/j.1439-0450.2003.00722.x
Ministry of Livestock Development Fisheries (MLDF) (2014). The United republic of Tanzania. Budget speech. Dodoma. Available online at: www.mifugouvuvi.go.tz/
Moore, J. E., Garcia, M. M., and Madden, R. H. (2002). Subspecies characterization of porcine Campylobacter coli and Campylobacter jejuni by multilocus enzyme electrophoresis typing. Vet. Res. Commun. 26, 1–9. doi: 10.1023/A:1013308903157
Mubito, E. P., Shahada, F., Kimanya, M. E., and Buza, J. J. (2014). Antimicrobial use in the poultry industry in Dar-es-Salaam, Tanzania and public health implications. Am. J. Res. Commun. 2, 51–63.
Payot, S., Avrain, L., Magras, C., Praud, K., Cloeckaert, A., and Chaslus-Dancla, E. (2004). Relative contribution of target gene mutation and efflux to fluoroquinolone and erythromycin resistance, in French poultry and pig isolates of Campylobacter coli. Int. J. Antimicrob. Agents 23, 468–472. doi: 10.1016/j.ijantimicag.2003.12.008
Pezzotti, G., Serafin, A., Luzzi, I., Mioni, R., Milan, M., and Perin, R. (2003). Occurrence and resistance to antibiotics of Campylobacter jejuni and Campylobacter coli in animals and meat in northeastern Italy. Int. J. Food Microbiol. 82, 281–287. doi: 10.1016/S0168-1605(02)00314-8
Pradhan, A. K., Van Kessel, J. S., Karns, J. S., Wolfgang, D. R., Hovingh, E., Nelen, K. A., et al. (2009). Dynamics of endemic infectious diseases of animal and human importance on three dairy herds in the northeastern United States. J. Dairy Sci. 92, 1811–1825. doi: 10.3168/jds.2008-1486
Ragimbeau, C., Schneider, F., Losch, S., Even, J., and Mossong, J. (2008). Multilocus sequence typing, pulsed-field gel electrophoresis, and fla short variable region typing of clonal complexes of Campylobacter jejuni strains of human, bovine, and poultry origins in Luxembourg. Appl. Environ. Microbiol. 74, 7715–7722. doi: 10.1128/AEM.00865-08
ROSCO (2007). Veterinary Practice According to CLSI Breakpoints. Available online at: https://rosco.docontrol.com/admin/edit_submenu.aspmainmenu=fetchcrypt5D584AA0141E52DFDCBD31B6BE9D6934F6122C90C61A43AD748FF2403F136778037DBF4462606153AA84798AAB783C5FC31864538440A8E5B5027B5BBED4DD5F [Accessed on November 15, 2014].
Rozynek, E., Antos-Bielska, M., Dzierzanowska-Fangrat, K., Szczepanska, B., and Trafny, E. A. (2010). Genetic similarity of Campylobacter isolates in humans, food, and water sources in Central Poland. Foodborne Pathog. Dis. 7, 597–600. doi: 10.1089/fpd.2009.0407
Ruiz-Palacios, G. M. (2007). The health burden of Campylobacter infection and the impact of antimicrobial resistance: playing chicken. Clin. Infect. Dis. 44, 701–703. doi: 10.1086/509936
Saenz, Y., Zarazaga, M., Lantero, M., Gastanares, M. J., Baquero, F., and Torres, C. (2000). Antibiotic resistance in Campylobacter strains isolated from animals, foods, and humans in Spain in 1997-1998. Antimicrob. Agents Chemother. 44, 267–271. doi: 10.1128/AAC.44.2.267-271.2000
Sahin, O., Kassem, I. I., Shen, Z., Lin, J., Rajashekara, G., and Zhang, Q. (2015). Campylobacter in poultry: ecology and potential interventions. Avian Dis. 59, 185–200. doi: 10.1637/11072-032315-Review
Salihu, M. D., Abdulkadir, J. U., Oboegbulem, S. I., Egwu, G. O., Magiji, A. A., Lawal, M., et al. (2009). Isolation and prevalence of Campylobacter species in cattle from Sokoto State, Nigeria. Vet. Ital. 45, 501–505.
Sanad, Y. M., Kassem, I. I., Abley, M., Gebreyes, W., LeJeune, J. T., and Rajashekara, G. (2011). Genotypic and phenotypic properties of cattle-associated Campylobacter and their implications to public health in the USA. PLoS ONE 10:e25778. doi: 10.1371/journal.pone.0025778
Scallan, E., Griffin, P. M., Angulo, F. J., Tauxe, R. V., and Hoekstra, R. M. (2011). Foodborne illness acquired in the United States–unspecified agents. Emerg. Infect. Dis. 17, 16–22. doi: 10.3201/eid1701.P21101
Senok, A., Yousif, A., Mazi, W., Sharaf, E., Bindayna, K., Elnimael, A., et al. (2007). Pattern of antibiotic susceptibility in Campylobacter jejuni isolates of human and poultry origin. Jpn. J. Infect. Dis. 60, 1–4.
Sheppard, S. K., Dallas, J. F., Wilson, D. J., Strachan, N. J., McCarthy, N. D., Jolley, K. A., et al. (2010). Evolution of an agriculture-associated disease causing Campylobacter coli clade: evidence from national surveillance data in Scotland. PLoS ONE 15:e15708. doi: 10.1371/journal.pone.0015708
Stapleton, K., Cawthraw, S. A., Cooles, S. W., Coldham, N. G., La Ragione, R. M., Newell, D. G., et al. (2010). Selecting for development of fluoroquinolone resistance in a Campylobacter jejuni strain 81116 in chickens using various enrofloxacin treatment protocols. J. Appl. Microbiol. 109, 1132–1138. doi: 10.1111/j.1365-2672.2010.04734.x
Taremi, M., Mehdi, M., Dallal, S., Gachkar, L., MoezArdalan, S., Zolfagharian, K., et al. (2006). Prevalence and antimicrobial resistance of Campylobacter isolated from retail raw chicken and beef meat, Tehran, Iran. Int. J. Food Microbiol. 108, 401–403. doi: 10.1016/j.ijfoodmicro.2005.12.010
Van Boven, M., Veldman, K. T., de Jong, M. C., and Mevius, D. J. (2003). Rapid selection of quinolone resistance in Campylobacter jejuni but not in Escherichia coli in individually housed broilers. J. Antimicrob. Chemother. 52, 719–723. doi: 10.1093/jac/dkg402
Van Looveren, M., Daube, G., De Zutter, L., Dumont, J. M., Lammens, C., Wijdooghe, M., et al. (2001). Antimicrobial susceptibilities of Campylobacter strains isolated from food animals in Belgium. J. Antimicrob. Chemother. 48, 235–240. doi: 10.1093/jac/48.2.235
White, D., Zhao, S., Simjee, S., Wagner, D. D., and McDermott, P. F. (2002). Antimicrobial resistance of foodborne pathogens. Microb. Infect. 4, 405–412. doi: 10.1016/S1286-4579(02)01554-X
Wieczorek, K., and Osek, J. (2013). Characteristics and antimicrobial resistance of Campylobacter isolated from pig and cattle carcasses in Poland. Polish J. Vet. Sci. 16, 501–508. doi: 10.2478/pjvs-2013-0070
Wilson, D. J., Gabriel, E., Leatherbarrow, A. J., Cheesbrough, J., Gee, S., Bolton, E., et al. (2008). Tracing the source of campylobacteriosis. PLoS Genet. 4:e1000203. doi: 10.1371/journal.pgen.1000203
World Health Organization (WHO) (2013). The global view of Campylobacteriosis, Vol. 57. Report of expert consultation, World Health Organization, Utrecht. Available online at: http://www.who.int/iris/handle/10665/80751#sthash.4I6cXIMY.dpuf
Yamazaki-Matsune, W., Taguchi, M., Seto, K., Kawahara, R., Kawatsu, K., Kumeda, Y., et al. (2007). Development of a multiplex PCR assay for identification of Campylobacter coli, Campylobacter fetus, Campylobacter hyointestinalis subsp. hyointestinalis, Campylobacter jejuni, Campylobacter lari and Campylobacter upsaliensis. J. Med. Microbiol. 56, 1467–1473. doi: 10.1099/jmm.0.47363-0
Keywords: Campylobacter, MLST, food animals, food safety, antimicrobial resistance
Citation: Kashoma IP, Kassem II, Kumar A, Kessy BM, Gebreyes W, Kazwala RR and Rajashekara G (2015) Antimicrobial Resistance and Genotypic Diversity of Campylobacter Isolated from Pigs, Dairy, and Beef Cattle in Tanzania. Front. Microbiol. 6:1240. doi: 10.3389/fmicb.2015.01240
Received: 03 September 2015; Accepted: 26 October 2015;
Published: 12 November 2015.
Edited by:
Charles W. Knapp, University of Strathclyde, UKReviewed by:
Atte Von Wright, University of Eastern Finland, FinlandZhangqi Shen, Iowa State University, USA
Copyright © 2015 Kashoma, Kassem, Kumar, Kessy, Gebreyes, Kazwala and Rajashekara. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Gireesh Rajashekara, cmFqYXNoZWthcmEuMkBvc3UuZWR1
 Isaac P. Kashoma
Isaac P. Kashoma Issmat I. Kassem
Issmat I. Kassem Anand Kumar1
Anand Kumar1 Beda M. Kessy
Beda M. Kessy Gireesh Rajashekara
Gireesh Rajashekara