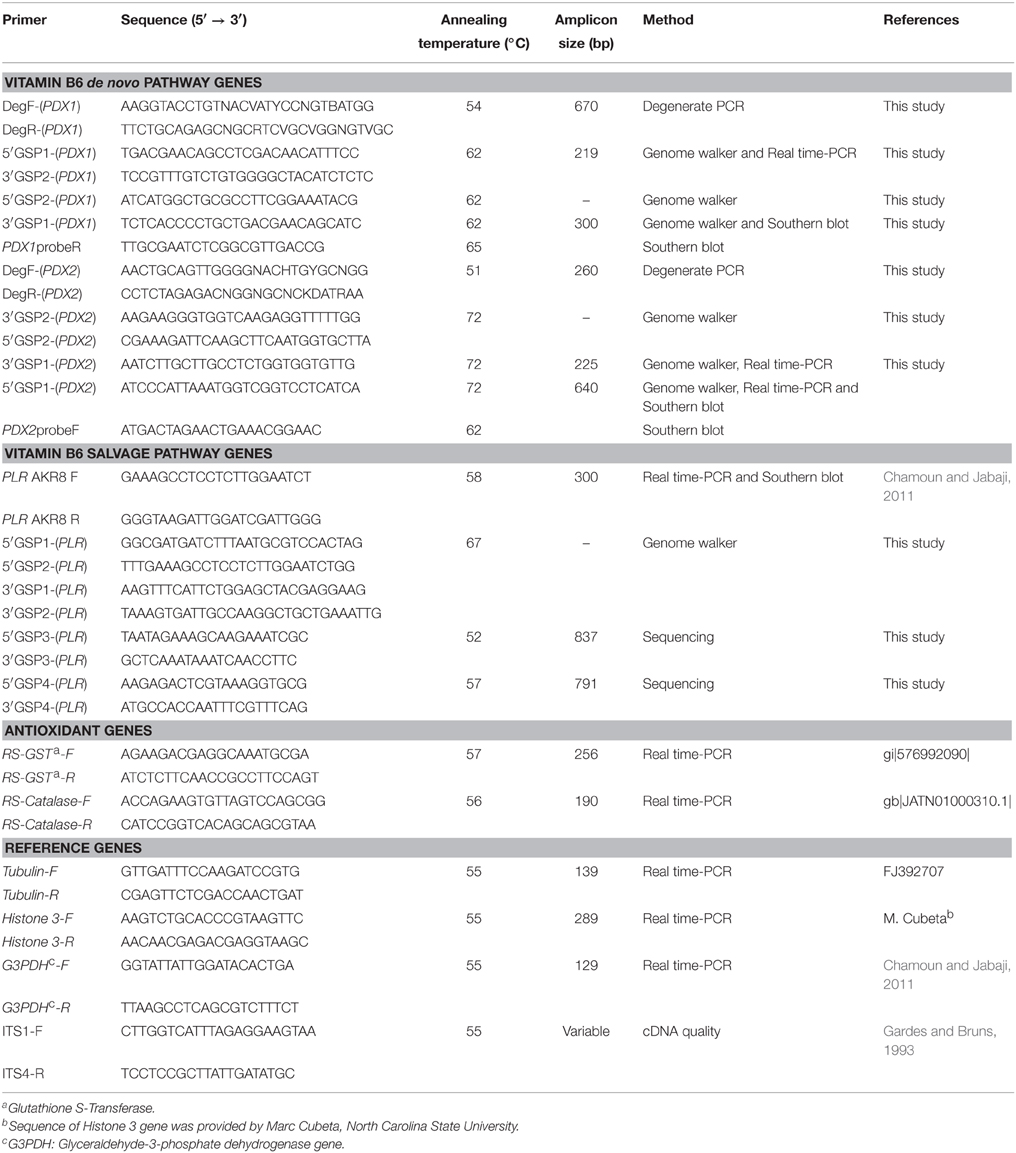
- 1Department of Plant Science, McGill University, Ste-Anne-de-Bellevue, QC, Canada
- 2Department of Plant Pathology, Ohio State University, Columbus, OH, USA
Vitamin B6 is recognized as an important cofactor required for numerous metabolic enzymes, and has been shown to act as an antioxidant and play a role in stress responses. It can be synthesized through two different routes: salvage and de novo pathways. However, little is known about the possible function of the vitamin B6 pathways in the fungal plant pathogen Rhizoctonia solani. Using genome walking, the de novo biosynthetic pathway genes; RsolPDX1 and RsolPDX2 and the salvage biosynthetic pathway gene, RsolPLR were sequenced. The predicted amino acid sequences of the three genes had high degrees of similarity to other fungal PDX1, PDX2, and PLR proteins and are closely related to other R. solani anastomosis groups. We also examined their regulation when subjected to reactive oxygen species (ROS) stress inducers, the superoxide generator paraquat, or H2O2, and compared it to the well-known antioxidant genes, catalase and glutathione-S-transferase (GST). The genes were differentially regulated with transcript levels as high as 33 fold depending on the gene and type of stress reflecting differences in the type of damage induced by ROS. Exogenous addition of the vitamers PN or PLP in culture medium significantly induced the transcription of the vitamin B6 de novo encoding genes as early as 0.5 hour post treatment (HPT). On the other hand, transcription of RsolPLR was vitamer-specific; a down regulation upon supplementation of PN and upregulation with PLP. Our results suggest that accumulation of ROS in R. solani mycelia is linked to transcriptional regulation of the three genes and implicate the vitamin B6 biosynthesis machinery in R. solani, similar to catalases and GST, as an antioxidant stress protector against oxidative stress.
Introduction
Vitamin B6 is a collective term that refers to a group of six vitamers: pyridoxal (PL), pyridoxine (PN), pyridoxamine, and their phosphorylated derivatives (PLP, PNP, PMP) (Fitzpatrick et al., 2012; Vanderschuren et al., 2013). In plants, fungi and prokaryotes, vitamin B6 vitamers are produced via the de novo biosynthetic pathway that ultimately leads to the synthesis of the most active form pyridoxal 5′-phosphate (PLP) via a heterodimer complex made up of two pyridoxal biosynthesis proteins that belong to highly conserved protein families (PDX1 and PDX2) (Mittenhuber, 2001; Raschle et al., 2005; Fitzpatrick et al., 2007). Of all the vitamers, PLP is critically important because it is essential as a cofactor for over 140 chemical reactions (Percudani and Peracchi, 2003; Roje, 2007; Hellmann and Mooney, 2010). In addition to the de novo pathway, a conserved “salvage pathway” is found in all organisms (González et al., 2007; Herrero et al., 2011; Rueschhoff et al., 2012). Reactions in the salvage pathway include the reduction of PL to PN, which is carried out by pyridoxal reductase (PLR), a downstream enzyme in the vitamin B6 biosynthesis pathway (Morita et al., 2004; Herrero et al., 2011), and the conversion of PN into PNP which is performed by PNP oxidase generating at the end of its pathway PLP (González et al., 2007; Sang et al., 2007).
In recent years, vitamin B6 has been identified as a potent antioxidant with a high ability to quench reactive oxygen species (ROS), resulting in an antioxidant capacity that rivals that of tocopherols or ascorbic acid, and may play a role in stress responses in fungi and plants (Mooney et al., 2009; Vanderschuren et al., 2013). The antioxidant properties of vitamin B6 was originally reported in the fungal pathogen Cercospora nicotianae, by providing resistance to cercosporin, a singlet oxygen generating toxin (Ehrenshaft et al., 1999; Bilski et al., 2000; Daub and Ehrenshaft, 2000). This novel characteristic of vitamin B6 as a ROS scavenger and its ability to increase resistance to biotic and abiotic stresses have been demonstrated in plant-microbe interaction studies (Danon et al., 2005; Denslow et al., 2005). Supplementation with PM could delay or decrease pathogen-induced leaf necrosis (Denslow et al., 2005) while PN could protect Arabidopsis flu mutant, which releases singlet oxygen in plastids, from cell death (Danon et al., 2005).
Studies on vitamin B6 metabolism and regulation are limited to a few fungi, (Ehrenshaft et al., 1999; Osmani et al., 1999; Benabdellah et al., 2009) including one plant pathogenic fungus, C. nicotianae. Necrotrophic fungi are successful pathogens that are able to overcome or suppress an array of complex ROS-mediated plant defenses (Chung, 2012). The relative sensitivity of necrotrophic plant pathogens to ROS is likely determined by the effectiveness of their own ROS detoxification ability. To survive under aerobic conditions, fungi must possess detoxification systems such as NADPH oxidase (NOX) complex that can effectively scavenge ROS, maintain reduced redox states within subcellular microenvironments, and repair ROS-triggered damage (Heller and Tudzynski, 2011; Chung, 2012).
The soil fungus Rhizoctonia solani Kühn (teleomorph Thanatephorus cucumeris, Frank, Donk) is a generalist, necrotrophic pathogen with a wide host range, causing damping-off of seedlings, root crown, stem rots, and sheath blight diseases of plants (Ogoshi, 1996; Sneh, 1996). Basal resistance to Rhizoctonia diseases in several crops is correlated with ROS-scavenging mechanisms such as hydrogen peroxide (H2O2) production, enhanced peroxidases (POX), and superoxide dismutase (SOD) activities, transcriptional regulation of NOX, regulation of several metabolites in the phenylpropanoid and vitamin B6 biosynthetic pathways, accumulation of oxidized fatty acids, and increased levels of cell wall bound phenolics (Taheri and Tarighi, 2011; Aliferis and Jabaji, 2012; Foley et al., 2013; Nikraftar et al., 2013; Aliferis et al., 2014). However, data on ROS-scavenging systems in R. solani is very limited. To date, evidence that R. solani genes of the vitamin B6 pathway are upregulated in response to biotic stress has been reported (Morissette et al., 2008; Chamoun and Jabaji, 2011; Gkarmiri et al., 2015). Parasitized hyphae and sclerotia of R. solani by the mycoparasite Stachybotrys elegans have displayed a substantial up-regulation in the transcription of the gene encoding PLR (Chamoun and Jabaji, 2011). Other vitamin B6 biosynthetic encoding genes such as pyridoxal-5-phosphatases and transaminases were recently reported to be upregulated in R. solani in response to antagonistic plant associated bacteria (Gkarmiri et al., 2015).
With the aim of gaining insight into the possible implication of ROS on vitamin B6 regulation in R. solani, we report on the characterization of three vitamin B6 genes, RsolPDX1, and RsolPDX2 from the vitamin B6 de novo pathway and RsolPLR from the salvage pathway. The characterized genes are homologs of R. solani and other known fungal vitamin B6 genes. R. solani vitamin B6 de novo pathway genes were up-regulated by the superoxide generator paraquat but not by H2O2 whereas RsolPLR was up-regulated by both chemicals. This displays a unique response of these genes according to the type of oxidative stress induced by ROS generating chemicals.
Materials and Methods
Fungal Strains, Media, and Culture Conditions
Starter culture of R. solani Kühn AG3 (isolate Rs114, ATCC 10183) was grown on potato dextrose agar (PDA; Difco, Detroit, USA) at 24°C for 5 days. For the isolation of vitamin B6 related genes (RsolPDX1, RsolPDX2, RsolPLR) from R. solani, plugs (5 mm) from starter cultures were placed in the center of fresh PDA plates, overlaid with cellophane membranes (500 PUT; UCB, North Augusta, USA), and grown for 7 days at 24°C. Cultures of Fusarium oxysporum (ATCC 60860) and Trichoderma virens (DAOM 169262) were grown similarly to R. solani and used as controls.
Experimental Setup for Oxidative Stress
To investigate the role of R. solani in detoxification/homeostasis of ROS, the relative transcript abundance of vitamin B6 genes (RsolPDX1, RsolPDX2, and RsolPLR) was monitored over time when R. solani was subjected to different oxidative stress inducers or when exogenous additions of vitamin B6 vitamers were added. For this purpose, R. solani plugs (5 mm) were grown in Petri plates containing 15 mL of half-strength potato dextrose broth (PDB) (PDB; Difco, Detroit, USA) for 3 days at 24°C. Subsequently, the PDB media in R. solani cultures was removed and substituted with 15 mL of fresh PDB amended with one of the following stress inducers: 5 mM of H2O2 (Sigma, Toronto, ON, Canada), 7.5 mM of phenylacetic acid (PAA) (Sigma) and 4 mM of the superoxide generator paraquat (N,N′-dimethyl-4,4′-bipyridinium dichloride), (Syngenta Crop Protection, Toronto, ON, Canada), or with the vitamin B6 vitamers: 0.01 g L−1 pyridoxine (PN) (Sigma), and 0.01 g L−1 pyridoxal-5′-phosphate (PLP) (Sigma). No amendment was added to the half-strength PDB media in the control plates. Oxidant sensitivity and effective concentrations (EC50) for H2O2 and paraquat were determined by obtaining dose-response curves and the concentration which resulted in near 50% inhibition of R. solani growth at 72 hours post treatment (HPT) (Wang et al., 2011), whereas the concentration of applied PAA was selected based on previous reports (Bartz et al., 2012). The vitamin B6 vitamers were chosen based on optimized concentrations that do not inhibit the growth of R. solani.
Comparison of the relative transcript abundance of RsolPDX1, RsolPDX2, and RsolPLR genes with the well-established antioxidants glutathione S-transferase (GST) and catalase genes was performed using specific primers designed in this study (Table 1). All experiments were conducted with three biological replicates and two technical replicates per treatment or control. Mycelia from treatments and controls were harvested at different HPT that spanned from 0.5 to 72 h depending on the type of the stress inducer, flash-frozen in liquid nitrogen, and processed for RNA extraction. Additional culture plates of R. solani exposed to H2O2 and paraquat were kept for light and fluorescence microscopy.
Nucleic Acids Extraction
Total genomic DNA was isolated from 100 mg of ground tissue of R. solani, F. oxysporum, and T. virens using the DNeasy Plant Mini Kit™ (Qiagen, Toronto, ON, Canada). Total RNA was isolated from 100 mg of ground R. solani mycelia exposed to various stress treatments and from the control using the RNeasy Plant Mini Kit™ (Qiagen) and treated with RNase-free DNaseI™ (Qiagen) according to the manufacturer's recommendations. The concentration and purity of RNA were assessed by spectrophotometry with ND1000 (NanoDrop, Wilmington, Delaware), while RNA quality was verified on 1.2% (w/v) formaldehyde-agarose gel electrophoresis. A total of 500 ng RNA was reverse transcribed using the Quantitect Reverse transcriptase kit™ (Qiagen).
Genome Library Construction and Manipulation of the Vitamin B6 Biosynthesis Genes RsolPDX1, RsolPDX2, and RsolPLR
Degenerate primer pairs, DegF-(PDX1)/DegR-(PDX1), and DegF-(PDX2)/DegR-(PDX2), were designed from the alignment of eight protein sequences of PDX1 (Figure 1A) and 11 protein sequences of PDX2 (Figure 1B) belonging to different fungi, respectively. Primers design was based on the conserved domains in PDX1 and PDX2 (Figures 1A,B). The primer pairs were also tested on the genomic DNA of F. oxysporum (ATCC 60860), and T. virens (DAOM 169262) since both fungi are known to harbor both genes, PDX1 (F. oxysporum Genbank accession number ENH63312) and PDX2 (T. virens Genbank accession number EHK18113), respectively. The degenerate primers were used in PCR reactions to amplify putative products of PDX1 and PDX2 from genomic DNA of R. solani.

Figure 1. Amino acid sequence alignment of the PDX1 (A) and PDX2 (B) proteins belonging to various fungi and plants. The GenBank accession numbers of the PDX1 proteins (A) are as follows: Piriformospora inidica (CAFZ01000024), Cercospora nicotianae (AF0356191825), Emericella nidulans (AF133101.1), Arthroderma gypseum (315049582), Neurospora crassa (AAK07850.1), Magnaporthe oryzae (389628343), Pyrenophora tritici-repentis (189190153), Aspergillus terreusNIH262 (115433602), Arabidopsis thaliana (145360746). The GenBank accession numbers of the PDX2 proteins (B) are as follows: Cercospora nicotianae (AF294268.1), Aspergillus nidulans (AF363613.1), Candida albicans (68468776), Neurospora crassa (12802355), Mycosphaerella populorum (gi 453085089), Saccharomyces cerevisiae Sno1p (296147212), Saccharomyces cerevisiae Sno2p (296147449), Saccharomyces cerevisiae Sno3p (296144372), Fusarium oxysporum (gi 475662976), Arabidopsis thaliana (30697380), Brachypodium distachyon (357113803). The multiple sequence alignment was done using BioEdit v7.2.0. Identical amino acid residues in all the sequences are highlighted DegF: Degenerate forward primer, DegR: Degenerate reverse primer. Boxes represent the location of degenerate primers used in PCR.
For the construction of Genome-walking libraries, R. solani AG3 (Rs114) genomic DNA was digested with four restriction enzymes (EcoRV, DraI, PvuII, and StuI) following the manufacturer's recommendations (Clontech, CA, USA). For Genome-walking PCR reactions, a set of gene-specific primers (PDX1-GSP, PDX2-GSP, PLR-GSP: 5′GSP1, 5′GSP2, 3′GSP1, and 3′GSP2; Table 1) were designed based on the genomic DNA sequence of the putative products for PDX1 and PDX2, and on R. solani mRNA sequence (Genbank accession number EU008744.1) for PLR. Specific PCR fragments were obtained, purified using the QIAquick PCR Purification Kit (Qiagen), and sub-cloned in pDrive cloning vector (Qiagen). Positive clones were sequenced and assembled in a contig using BioEdit v7.2.0 (Hall, 1999) and then blasted on NCBI for homology and identity confirmation. The full sequences of RsolPDX1, RsolPDX2, and RsolPLR genes were annotated and submitted to GenBank database (KF620111, KF620112, and KJ395592), respectively.
Genomic Southern Hybridization
Specific probes for each of the RsolPDX1, RsolPDX2, and RsolPLR genes were constructed by amplifying genomic DNA fragments of R. solani using an appropriate set of primer pairs (Table 1). The amplified products were DIG-labeled using the DIG-High Prime DNA Labeling and Detection Starter Kit I (Roche Applied Science, QC, Canada). The genomic DNA of R. solani was digested with an appropriate set of restriction enzymes [(EcoRI, PstI, and HindIII for PDX1), (EcoRI, PstI, and KpnI for PDX2), (EcoRI, PstI, HindIII, and XhoI for PLR)], electrophoresed on a 1% agarose gel, transferred onto Nylon Hybond N+ membrane (Roche Applied Science), and UV cross-linked. Hybridization was performed following the procedure of Chamoun et al. (2013).
Phylogenetic Analysis
Multiple sequence alignments of RsolPDX1, RsolPDX2, and RsolPLR cDNA with other members of PDX1, PDX2, and PLR from selected fungi (Ascomycetes and Basidiomycetes) and plants were conducted using Clustal Omega (Sievers and Higgins, 2014). The tree was constructed using MEGA 6.0 and generated by the maximum likelihood method based on the General Time Reversible model with 1000 Bootstrap replicates (Tamura et al., 2011).
Quantitative RT-PCR
QRT-PCR assays were conducted on five target genes RsolPDX1, RsolPDX2, RsolPLR, GST, and catalase, and three reference genes G3PDH, Histone-3, and β-Tubulin (Table 1) using Stratagene Mx3000 (Stratagene, Cedar Creek, USA). Each template and negative control had three biological replicates with two technical replicates included in each run. PCR assays were performed as previously described (Chamoun et al., 2013) using the appropriate annealing temperature for each primer pair (Table 1). The relative transcript abundance levels of the genes were calculated according to (Zhao and Fernald, 2005) and normalized against the reference genes showing the lowest minimal variation and coefficient of variation (CV) across all treatments. The relative transcript abundance of the genes was tested for significance between treatments and controls at each harvesting time point by Two-way analysis of variance (ANOVA) and, when appropriate, for least significant differences (LSD) at P < 0.05 using the SPSS statistical package v. 22.0, (IBM Corp., Armonk, NY, USA).
Optical and Fluorescence Microscopy
To associate changes in the transcript abundance of the genes with the ROS accumulation in R. solani to the different stress inducers, control and treated-mycelia with H2O2 (5 mM) or paraquat (4 mM) were viewed under a light and fluorescent microscope at 3 and 24 HPT. Images were digitally documented with the Moticam 2300 digital camera (GENEQ Inc. Montreal, Quebec) for light microscopy. For ROS detection, R. solani mycelia were incubated with 10 μM of 2′,7′-dichlorodihydrofluorescein diacetate (H2DCF-DA), a specific ROS molecular-detection probe, in half-strength PDB for 30 min. Mycelia were then washed with pre-warmed (28°C) half-strength PDB for 30 min to remove the non-internalized probe and were examined under Zeiss SteREO Discovery.V20 fluorescence microscope (Carl Zeiss Canada Ltd., Toronto, Ontario, Canada) at 3 and 24 HPT. Fluorescence intensity emitted from individual cells was measured using an excitation wavelength of 470 nm.
Results
R. solani Genes RsolPDX1, RsolPDX2, and RsolPLR are Members of the Vitamin B6 Biosynthetic Pathway
The designed primer pairs DegF/R-(PDX1) and DegF/R-(PDX2) successfully amplified PCR products of 671 and 263 bp in size, respectively which were confirmed by sequencing to be PDX1 and PDX2. Subsequently, the gene specific primers (PDX1-GSP, PDX2-GSP, PLR-GSP) (Table 1) were used to obtain the whole sequences of RsolPDX1, RsolPDX2, and RsolPLR using the Genome-walking technique. Initially, only partial sequence was obtained for PLR, hence, additional PLR-GSP primers (Table 1) were designed based on genome sequence of R. solani AG3 (strain Rhs1AP) to get the complete sequence. PCR products for each of the three genes were assembled into contigs and their sequence analysis led to the identification of the vitamin B6 genes belonging to the de novo pathway in R. solani, RsolPDX1 (accession number KF620111), and RsolPDX2 (accession number KF620112), and to a identification of RsolPLR (accession number KJ395592) belonging to the vitamin B6 salvage pathway. The organization of the three genes and their features are depicted in Figures 2–4.

Figure 2. Complete sequence of RsolPDX1 (gene accession number KF620111). Introns are written in italic in the gene sequence. The signature motif of the PDX1 protein family is underlined. Amino acids known to be essential for enzyme activity are shadowed. Asterisk indicates end codon. Dotted line indicates southern blot probe.
The full length of the RsolPDX1 and RsolPDX2 sequences obtained during our analyses are 1640 and 1178 bp, respectively. The open reading frame of RsolPDX1 consists of 1402 nucleotides encoded from 61 to 1463 bp, while the ORF of RsolPDX2 consists of 980 nucleotides encoded from 99 to 1079 bp. Both genes are interrupted with several introns (Figures 2, 3). The predicted protein sequences of RsolPDX1 and RsolPDX2 consist of 322 and 248 amino acid residues with a molecular weight of 33.65 or 26.78 kDa and a calculated isoelectric point (pI) of 6.625 or 5.815, respectively. The signature motif of the PDX1 protein family (LPVVSFAAGGVATPADAA) and conserved amino acids (D, M, K, and GTG) were identified in PDX1 (Figure 2). Similarly, the signature motif of the PDX2 protein family (C-H-E) which is essential for the protein activity in addition to other conserved amino acids (i.e., PGGEST, FIRAP, and FHPE) are highlighted in PDX2 (Figure 3).

Figure 3. Complete sequence of RsolPDX2 (gene accession number KF620112). Introns are written in italic in the gene sequence. The signature motif of the PDX2 protein family C-H-E (known to be essential for enzyme activity) is underlined. Conserved amino acids are shadowed. Asterisk indicates end codon. Dotted line indicates southern blot probe.
The obtained full length of the RsolPLR sequence is 1837 bp and its sequence analysis revealed that it belongs to the aldo_keto_reductase family. RsolPLR ORF is encoded from 50 to 1627 bp and is interrupted by several introns whereas conserved amino acids (i.e., AR, SE, E, and YSPLG) in PLR proteins are highlighted (Figure 4). RsolPLR gene encodes a putative polypeptide of 337 amino acid residues with a molecular weight of 36.92 kDa and a calculated isoelectric point (pI) of 5.89 (Figure 4).

Figure 4. Complete sequence of RsolPLR (gene accession number KJ395592). Introns are written in italic in the gene sequence. The four residues that compose the Aldo_keto_reductase conserved catalytic tetrad are underlined. Conserved amino acids are shadowed. Asterisk indicates end codon. Dotted line indicates southern blot probe.
Gene Copies of RsolPDX1, RsolPDX2, and RsolPLR
Gene copies of RsolPDX1, RsolPDX2, and RsolPLR in the genome of R. solani were estimated by Southern blot analysis (Figure 5). Under high stringency conditions of hybridization, RsolPDX1, and RsolPLR probes hybridized to more than one high molecular fragment, indicating that two copies are present for each of the genes. On the other hand, RsolPDX2 is shown to be present as single copy as revealed by the hybridization of its corresponding probe to a single fragment (Figure 5).
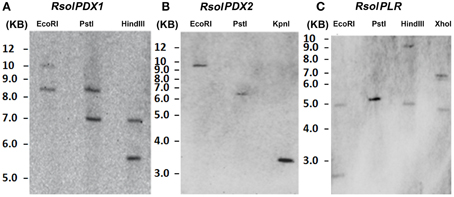
Figure 5. Southern blot analysis showing gene copy number of RsolPDX1 (A), RsolPDX2 (B), and RsolPLR (C) in R. solani AG3.
RsolPDX1, RsolPDX2, and RsolPLR are Phylogenetically Related to Homologs of the Vitamin B6 Genes
The phylogenetic tree of PDX1, PDX2, and PLR shows a complete separation between the three families of vitamin B6 biosynthesis genes. Within each gene family, Basidiomycete, and Ascomycete sequences have clustered in separate clades. Candida albicans and Saccharomyces cerevisiae were clustered in a separate clade for each gene. PDX1, PDX2, and PLR sequences of different R. solani anastomosis groups are consistently clustered together in a separate clade (Figure 6). Within each of the two families, RsolPDX1/2 are most closely related to PDX1/2 of Rhs1AP, another strain of R. solani AG3. Finally, within the aldo-keto reductase superfamily, RsolPLR gene is closely clustered to the PLR gene of R. solani AG1-IA, the rice sheath blight pathogen (Figure 6).
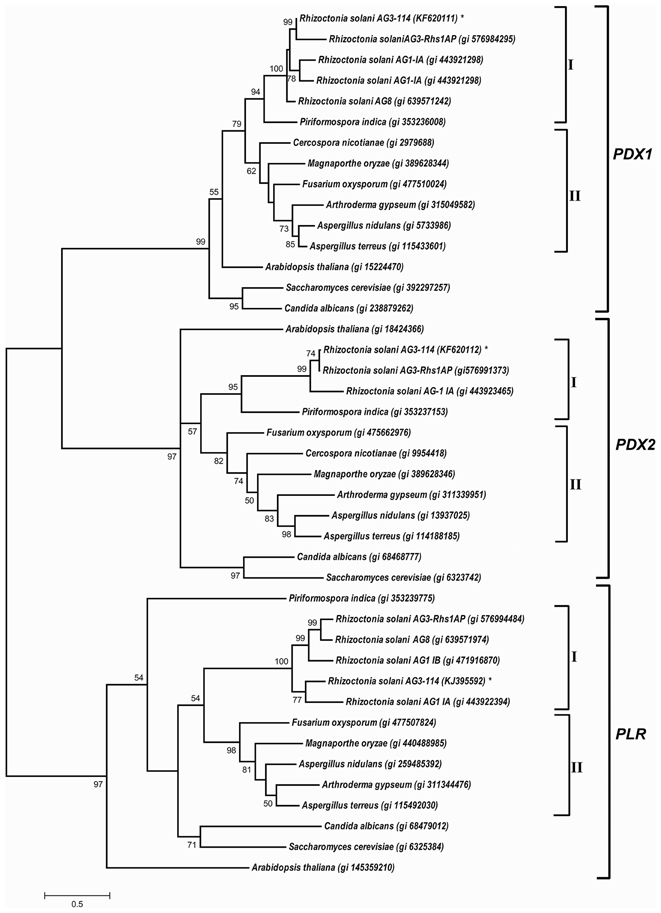
Figure 6. A phylogenetic tree based on the similarity among cDNA sequences of PDX1, PDX2, and PLR from selected fungi and plants. The phylogenetic relationship was generated from multiple sequence alignments using Clustal Omega and the tree was constructed using the program MEGA 6.0, and generated by the maximum likelihood method (Tamura et al., 2011). NCBI accession numbers are listed next to the species names. Asterisk indicates R. solani AG3- 114 cDNA sequences of RsolPDX1, RsolPDX2, and RsolPLR. Bootstrap scores (1000 replicates) are shown at nodes only when they are higher than 50%. I: Basidiomycetes, II: Ascomycetes.
R. solani Genes of the Vitamin B6 de novo and Salvage Pathways are Differentially Regulated by Different Stress Inducers
To provide insight into the regulation of R. solani vitamin B6 biosynthesis genes exposed to different stress inducers, and investigate whether they could play a role in homeostasis of ROS, the relative transcript abundances of RsolPDX1, RsolPDX2, and RsolPLR genes were assessed in R. solani at 24 and 72 HPT with paraquat and PAA and also at 0.5 and 3 HPT with H2O2.
Paraquat
Compared to control cultures, the relative transcript abundance of RsolPDX1 and RsolPDX2 has significantly increased at both time points with a higher fold increase of 6.4 and 9.9 in RsolPDX1 and RsolPDX2, 24 HTP, respectively (Figures 7A,D). Likewise, there was a significant increase in relative transcript abundance level of RsolPLR at both HPT with a notable fold increase of 32.8 (Figure 7G).
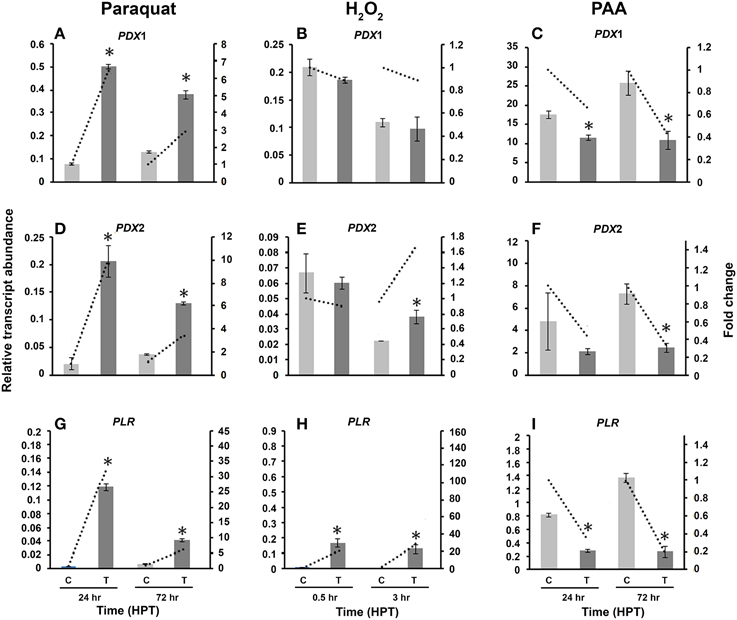
Figure 7. Effect of abiotic stress on R. solani AG3 vitamin B6 genes. Regulation of RsolPDX1 (A–C), RsolPDX2 (D–F), and RsolPLR (G–I) transcripts when grown in PDB amended with paraquat (4 mM) (A,D,G), hydrogen peroxide (H2O2) (5 mM) (B,E,H) or phenylacetic acid (PAA) (7.5 mM) (C,F,I). The relative transcript abundance of gene expression was normalized with appropriate housekeeping genes (G3PDH for paraquat and H2O2; Histone and Tubulin for PAA). C, control; R. solani grown without stress inducers. T, treatment with stress inducer. Asterisk indicates significant relative transcript abundance between the control and interaction at each time point using Least Significant Difference test (P < 0.05). Bars represent the average relative transcript abundance of three biological replicates ± standard deviation. Dotted line represents fold change which was calculated by normalization of treatment samples with appropriate controls at each corresponding time point.
H2O2
Exposure of R. solani mycelia to H2O2 had no effect on RsolPDX1 (Figure 7B) but an effect on RsolPDX2 at 3 HPT with a slight fold increase (1.68 fold; Figure 7E). Importantly H2O2 caused a notable increase in RsolPLR relative transcript abundance at 0.5 and 3 HPT with a substantial fold increase of 27.8 fold at 3 HPT (Figure 7H).
PAA
To obtain further insights into the putative activities of vitamin B6, we also assessed whether the relative transcripts abundance of RsolPDX1, RsolPDX2, and RsolPLR are regulated by PAA, an antimicrobial and antioxidant compound produced by R. solani AG3 (Bartz et al., 2012). The three genes were mostly down-regulated in the range of 5.0–1.5 fold at 24 and 72 HPT, respectively (Figures 7C,F,I).
The Antioxidant Encoding Genes GST and Catalase are Transcriptionally Regulated in R. solani by Abiotic Stress
To confirm the presence of oxidative stress in R. solani, the activity of other well-known antioxidant genes like GST and catalase was compared to the activity of the vitamin B6 genes under the same abiotic stress conditions (Figure 8). GST was significantly induced by paraquat and H2O2 (Figures 8A,B) with an 8.6 fold and 4.2 increase at 24 and 72 HPT, respectively. Paraquat significantly upregulated catalase at both time periods, with a highest increase of 3.9 fold at 72 HPT (Figure 8C). However, exposure to H2O2 caused a downregulation of 5.9 fold decrease as compared to the control at 3 HPT (Figure 8D).
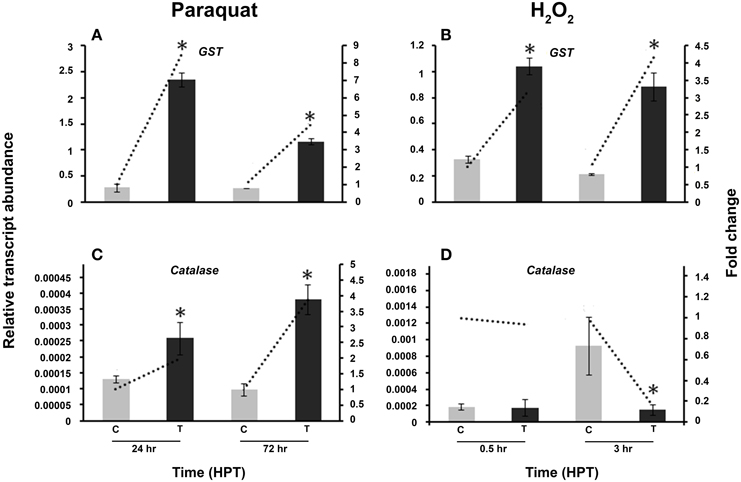
Figure 8. Effect of abiotic stress on R. solani AG3 glutathione S-Transferase (GST) and catalase genes. Regulation of R. solani AG3 GST (A,B), and catalase (C,D) transcripts when grown in PDB amended with paraquat (4 mM) (A,C) or hydrogen peroxide (H2O2) (5 mM) (B,D). The relative transcript abundance of gene expression was normalized with G3PDH. C, control; R. solani grown without stress inducers. T, treatment with stress inducer. HPT, hours post treatment. Asterisk indicates significant relative transcript abundance between the control and interaction of each time point using Least Significant Difference test (P < 0.05). Bars represent the average relative transcript abundance of three biological replicates ± standard deviation. Dotted line represents fold change which was calculated by normalization of treatment samples with appropriate controls at each corresponding time point.
R. solani Vitamin B6 Genes are Regulated by External Supplementation of Vitamin B6 Vitamers
To understand the role of vitamin B6 vitamers on the transcriptional regulation of vitamin B6 genes, R. solani was grown in PDB amended with either PN or PLP. No growth differences were observed between the control and the treatments (data not shown). Exogenous addition of PN, induced the regulation of both RsolPDX1 and RsolPDX2 (6.1 and 7.7 fold, respectively) at 3 HPT (Figures 9A,C), caused a drop to near basal level values at 24 HPT (1.7 and 1.1 fold, respectively), followed by significant down-regulation (16.7 and 9.1 fold, respectively) at 72 HPT (Figures 9A,C). PN caused a decrease in RsolPLR relative transcript abundance at all time points (Figure 9E). The amendment of growth medium with PLP, induced the expression of RsolPDX1 and RsolPDX2 at 24 HPT followed by a drop to the basal level at 72 HPT (Figures 9B,D). There was a substantial increase in transcript relative abundance of RsolPLR 3 HPT (2.5 fold) and 24 HPT (6.7 fold) when PLP was added exogenously (Figure 9F).
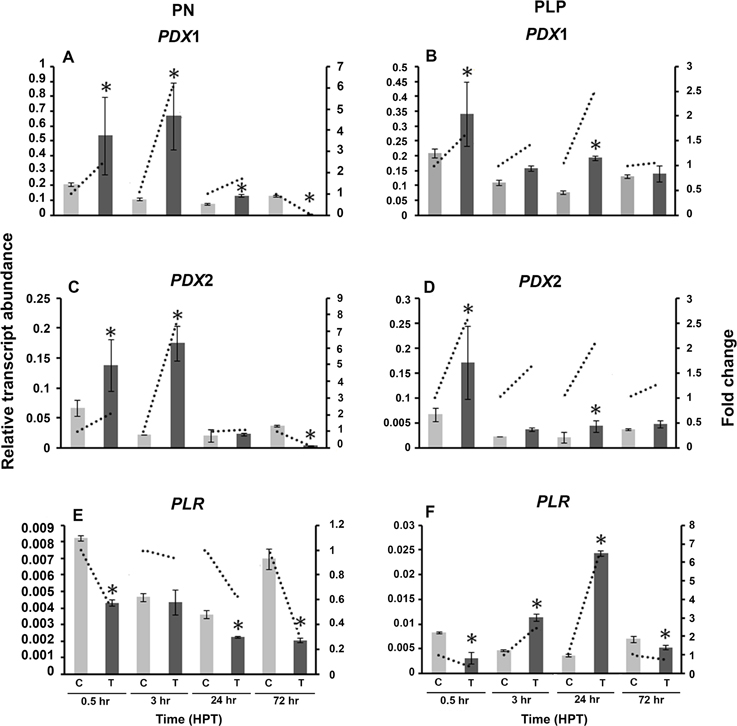
Figure 9. Effect of Vitamin B6 vitamers (PN: pyridoxine or PLP: pyridoxal 5′-phosphate) on R. solani AG3 vitamin B6 genes. Regulation of RsolPDX1 (A,B), RsolPDX2 (C,D), and RsolPLR (E,F) transcripts when grown in PDB amended with PN (0.01 g/L) (A, C, E) or PLP (0.01 g/L) (B, D, F). The relative transcript abundance of gene expression was normalized with G3PDH. C, control; R. solani grown without stress inducers. T, treatment with vitamers. HPT, hours post treatment. Asterisk indicates significant relative transcript abundance between the control and interaction of each time point using Least Significant Difference test (P < 0.05). Bars represent the average relative transcript abundance of three biological replicates ± standard deviation. Dotted line represents fold change which was calculated by normalization of treatment samples with appropriate controls at each corresponding time point.
Accumulation of ROS in R. solani Mycelia is Linked to Transcriptional Regulation of RsolPDX1, RsolPDX2, and RsolPLR
To determine whether the increase in vitamin B6 transcripts in the paraquat or H2O2-treated mycelia are linked to an accumulation of ROS in the fungal hyphae, the presence of these chemical species was estimated by adding H2DCF-DA to the growing media in which the fungus was exposed to 3 and 24 h under H2O2 and paraquat stress, respectively. An intense green fluorescence, corresponding to the oxidative form of H2DCF-DA, was detected in both treatments (Figures 10G–L). Hyphae of the untreated control plates did not display any green fluorescence (Figures 10A–F). R. solani growth was reduced by the addition of paraquat and H2O2 as compared to the control treatment (Figures 11A,C,E). Additionally, H2O2 and paraquat-treated hyphae showed increased levels of vacuolarization and compaction (Figures 11D,F) when compared to the control (Figure 11B) leading to loss of viability. These data confirm that exposure of the fungus to paraquat or H2O2 induces an oxidative stress in the fungal hyphae and could be related to the oxidative status of the fungus.
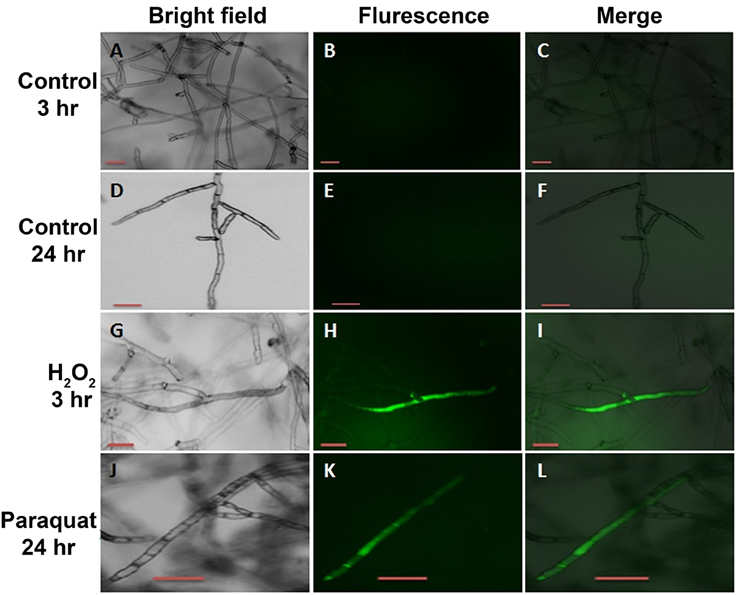
Figure 10. Intracellular production of ROS in R. solani AG3 mycelia grown for 3 days before exposure to 4 mM paraquat or 5 mM H2O2. The production of ROS was visualized by fluorescence microscopy using the ROS-sensitive probe H2DCF-DA. Control mycelia with no evidence of endogenous ROS production at 3 HPT (A–C) or at HPT 24 (D–F). Mycelia grown in PDB amended with 5 mM H2O2 at 3 HPT (G–I) or 4 mM paraquat at 24 HPT (J–L). Bar = 50 μm.
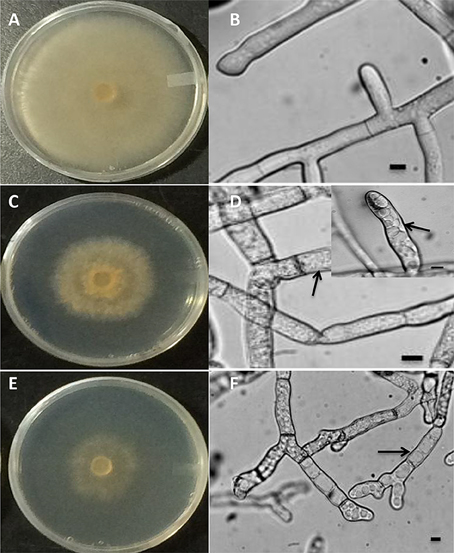
Figure 11. Macroscopic (A,C,E) and microscopic (B,D,F) images of R. solani AG3 mycelia grown for 3 days before exposure to abiotic stress. Control mycelia in PDB at 3 HPT or at 24 HPT showing normal hyphal cellular organization (A,B). Mycelia grown in PDB amended with 5 mM H2O2 at 3 HPT (C,D, inset) or 4 mM paraquat at 72 HPT (E,F) displaying intracellular disorganization, vacuolarization, and loss of cytoplasm (arrows). Bar = 10 μm.
Discussion
In this study, we characterized for the first time the PDX1/PDX2 genes of the de novo component and the PLR gene of the salvage component of the vitamin B6 biosynthetic pathway in the plant pathogenic fungus Rhizocotnia solani AG3 (Rs114) and provided the evidence for their transcriptional regulation under different stress inducers. We also show that these genes might play a role in oxidative stress protection in R. solani.
Sequences of the vitamin B6 genes are similar to those found in other organisms. The predicted amino acid sequences of the three genes showed high degree of similarity to other fungal PDX1, PDX2, and PLR proteins, and their close relatedness to other fungal homologs was confirmed (Ehrenshaft et al., 1998; Nakano et al., 1999; Ehrenshaft and Daub, 2001; Ellis, 2002; Barski et al., 2008; Benabdellah et al., 2009). The release of the draft genome sequence of R. solani AG3 (strain Rhs1AP) (Cubeta et al., 2014) further confirmed the genes' annotations. The presence of conserved amino acids suggests the ability of PDX1-PDX2 complex formation that is crucial for vitamin B6 biosynthesis (Belitsky, 2004; Raschle et al., 2005). Interestingly, there is variation in the number of introns present in various vitamin B6 de novo biosynthesis genes. As opposed to the PDX1 or PDX2 homologs identified in C. nicotianae and Glomus intraradices which are intronless (Ehrenshaft et al., 1998; Ehrenshaft and Daub, 2001; Benabdellah et al., 2009), the three vitamin B6 genes in R. solani AG3 (Rs114) contain introns similar to PyroA (PDX1) gene of Aspergillus nidulans (Osmani et al., 1999). From an evolutionary perspective, the presence of introns is advantageous because it provides organisms with diversity of proteins through alternative splicing or because introns might possess elements that are involved in regulation of gene expression (Kalsotra and Cooper, 2011; Yang et al., 2013).
Southern blot analysis indicated that two copies are present for RsolPDX1 and RsolPLR, most likely as a result of gene duplication, and a single copy is present for RsolPDX2 in R. solani AG-3 (Rs114). While most other fungi contain a single copy of those genes (Ehrenshaft et al., 1998; Osmani et al., 1999; Ehrenshaft and Daub, 2001; Benabdellah et al., 2009), S. cerevisiae, SNZ (PDX1 homolog), and SNO (PDX2 homolog) genes were shown to have three members each with only SNZ1 and SNO1 being implicated in vitamin B6 biosynthesis (Padilla et al., 1998; Rodríguez-Navarro et al., 2002). Only one copy of each of the three genes was found in the draft genome sequence of R. solani AG3 (strain Rhs1AP). This could be an indication that the PDX1, PDX2, and PLR gene copy numbers differ among the various R. solani strains, although this will require subsequent validation once higher quality genome assemblies are released. Our Southern hybridization provides only preliminary evidence of gene copy number. We don't exclude the possibility of having additional copies due to high molecular size of the fragments obtained. Much remains to be determined whether the two copies of RsolPDX1 and RsolPLR are both functional or one of them is a pseudogene.
To date, evidence that R. solani genes of the vitamin B6 pathway are upregulated in response to biotic stress has been reported (Morissette et al., 2008; Chamoun and Jabaji, 2011; Gkarmiri et al., 2015). However, to the best of our knowledge, little is known of the physiological responses of R. solani to various oxidative stress and also no studies exist on transcriptional regulation of genes in the vitamin B6 biosynthetic pathway, nor on their role in oxidative stress alleviation in R. solani experiencing abiotic stress.
The increased formation of ROS in hyphal cells can induce oxidative stress and damage to DNA, RNA, protein, and lipids, leading to the loss of cell viability (Apel and Hirt, 2004; Sharma et al., 2012). Our results clearly showed that paraquat and H2O2 induced ROS formation in hyphal cells of R. solani leading to growth reduction and loss of viability of hyphal cells. These observations were linked to the regulation of vitamin B6 encoding genes and their collective possible role as oxidative stress alleviation. Our findings indicate exposure of R. solani to paraquat upregulates RsolPDX1 and RsolPDX2 expression possibly suggesting a role of vitamin B6 in protection of R. solani against superoxide anions. In other organisms, gene regulation studies have continually connected vitamin B6 to oxidative stress and noted an increased transcriptional activity of vitamin B6 de novo pathway genes in response to oxidative stress. As examples, GintPDX1, the Glomus intrarradices and SNZ1, the S. cerevisiae PDX1 homolog and PDX2 of S. pombe showed increased transcript accumulation upon treatments with paraquat and H2O2, respectively (Lee et al., 1995; Chen et al., 2003; Benabdellah et al., 2009).
Furthermore, we show evidence that the vitamin B6 salvage pathway appears to be involved in the oxidative stress response in R. solani. RsolPLR transcript levels were up-regulated by both paraquat and H2O2 at all time points. This finding is in agreement with the transcriptional increase of PLR in S. pombe after 1 h of H2O2 treatment (Chen et al., 2003) and with the role of PLR in PN catalysis (Morita et al., 2004; Sang et al., 2007). The reason for the differential transcriptional regulation of RsolPDX1 and RsolPDX2 and RsolPLR by H2O2 and superoxide radicals is unknown, and may reflect differences in the type of damage induced by ROS.
To overcome oxidative damage, all living organisms have developed antioxidant systems to efficiently quench ROS excess and to keep ROS production and scavenging systems in check (Sharma et al., 2012). Maintaining cellular ROS homeostatis is achieved by the production of non-enzymatic antioxidants such as GST and enzymatic antioxidants such as catalases. Therefore, studying the regulation of GST and catalase in stressed cultures of R. solani is direct confirmation that these genes play a role in oxidative stress protection. Similar to vitamin B6 encoding genes, differential upregulation of GST and catalase was observed in response to the type of stressor, although the fold increase in vitamin B6 encoding genes was substantially higher. These results may indicate that vitamin B6, via the expression of RsolPDX1 and RsolPDX2 and RsolPLR, has better ability than glutathione to decrease ROS levels (Ehrenshaft et al., 1999).
The role of RsolPDX1 and RsolPDX2 and RsolPLR in vitamin B6 biosynthesis suggests a possible regulation of their expression by vitamin B6 and the expression of the homologs is dependent on vitamin B6 availability. Surprisingly in other biological systems, regulation of PDX1 expression was not affected by the addition of vitamin B6 (Rodríguez-Navarro et al., 2002; Benabdellah et al., 2009). The authors claimed that this independence is related to the high constitutive expression levels that may mask the effect of the absence of the vitamin. In our study, both PN and PLP vitamers up-regulated the vitamin B6 de novo genes, RsolPDX1, and RsolPDX2, in R. solani. Given the involvement of the pyridoxine encoding de novo genes in vitamin B6 biosynthesis, and the central role of PLP as a cofactor, a regulatory switch based on the amount of PN and PLP is likely to exist (Mooney et al., 2009), it is very plausible that vitamin B6 vitamers may act as regulators. Alternatively, the addition of vitamers might be perceived by R. solani as signal indicating stress leading to upregulation of the de novo vitamin B6 genes at the onset of treatment. In a similar fashion, plants supplemented with the well-known antioxidants riboflavin (vitamin B2) or thiamine (vitamin B1) developed systemic resistance to bacterial or fungal infections along with increased transcription of PR related genes (Dong and Beer, 2000; Ahn et al., 2005). As for the decrease in the transcript abundance levels of both RsolPDX1 and RsolPDX2 at 72 HPT might be due to conversion of PN into PNP and eventually to PLP, resulting in decrease of substrate availability (Zhao and Winkler, 1995; Rueschhoff et al., 2012). In this regard mechanisms of how vitamers regulate the de novo vitamin B6 genes remain to be explored further.
Contrary to RsolPDX1 and RsolPDX2, the transcriptional response of RsolPLR was vitamer specific; a down regulation in response to PN and up regulation in response to PLP. These results were expected since PN is the product of the reaction catalyzed by PLR (Herrero et al., 2011) causing a decrease in relative transcript abundance of RsolPLR. Increase in transcript levels may be due to the conversion of PLP to PL, the substrate for PLR (Zhao and Winkler, 1995; Herrero et al., 2011).
Conclusion
RsolPDX1, RsolPDX2, and RsolPLR represent three important components of the vitamin B6 pathway in R. solani AG3 (Rs114). These genes are differentially regulated upon various types of oxidative stress, such as paraquat and H2O2. Taken together, the indirect participation of the de novo (RsolPDX1 and RsolPDX2) and salvage (RsolPLR) vitamin B6 genes in the oxidative stress response of the plant pathogenic fungus R. solani is strongly suggested. In yeasts, gene disruption and over expression have been valuable in delineating the biological role of PDX1 in fungi. Comparable approaches should prove equally valuable to define the role of R. solani vitamin B6 genes.
Author Contributions
JS, RC, and SJ conceived, designed, and executed the experiments. EG helped in the execution of certain experiments. JS and RC analyzed the data. JS, RC, and SJ contributed to the writing of the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The work was supported by a research grant to SJ from the Natural Sciences and Engineering Research Council of Canada (NSERC-Discovery, grant number 137135-13). The authors thank J.-B. Charron for the use of Zeiss SteREO Discovery.V20 microscope.
References
Ahn, I.-P., Kim, S., and Lee, Y.-H. (2005). Vitamin B1 functions as an activator of plant disease resistance. Plant Physiol. 138, 1505–1515. doi: 10.1104/pp.104.058693
Aliferis, K. A., Faubert, D., and Jabaji, S. (2014). A metabolic profiling strategy for the dissection of plant defense against fungal pathogens. PLoS ONE 9:e111930. doi: 10.1371/journal.pone.0111930
Aliferis, K. A., and Jabaji, S. (2012). FT-ICR/MS and GC-EI/MS metabolomics networking unravels global potato sprout's responses to Rhizoctonia solani infection. PLoS ONE 7:e42576. doi: 10.1371/journal.pone.0042576
Apel, K., and Hirt, H. (2004). Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 55, 373–399. doi: 10.1146/annurev.arplant.55.031903.141701
Barski, O. A., Tipparaju, S. M., and Bhatnagar, A. (2008). The aldo-keto reductase superfamily and its role in drug metabolism and detoxification. Drug Metab. Rev. 40, 553–624. doi: 10.1080/03602530802431439
Bartz, F. E., Glassbrook, N. J., Danehower, D. A., and Cubeta, M. A. (2012). Elucidating the role of the phenylacetic acid metabolic complex in the pathogenic activity of Rhizoctonia solani anastomosis group 3. Mycologia 104, 793–803. doi: 10.3852/11-084
Belitsky, B. R. (2004). Physical and enzymological interaction of Bacillus subtilis proteins required for de novo pyridoxal 5′-phosphate biosynthesis. J. Bacteriol. 186, 1191–1196. doi: 10.1128/JB.186.4.1191-1196.2004
Benabdellah, K., Azcón-Aguilar, C., Valderas, A., Speziga, D., Fitzpatrick, T. B., and Ferrol, N. (2009). GintPDX1 encodes a protein involved in vitamin B6 biosynthesis that is up-regulated by oxidative stress in the arbuscular mycorrhizal fungus Glomus intraradices. New Phytol. 184, 682–693. doi: 10.1111/j.1469-8137.2009.02978.x
Bilski, P., Li, M. Y., Ehrenshaft, M., Daub, M. E., and Chignell, C. (2000). Vitamin B6 (pyridoxine) and its derivatives are efficient singlet oxygen quenchers and potential fungal antioxidants. Photochem. Photobiol. 71, 129–134. doi: 10.1562/0031-8655(2000)0710129SIPVBP2.0.CO2
Chamoun, R., Aliferis, K. A., and Jabaji, S. H. (2013). Characterization and transcriptional regulation of Stachybotrys elegans mitogen-activated-protein kinase gene smkA following mycoparasitism and starvation conditions. Curr. Genet. 59, 43–54. doi: 10.1007/s00294-012-0386-2
Chamoun, R., and Jabaji, S. (2011). Expression of genes of Rhizoctonia solani and the biocontrol Stachybotrys elegans during mycoparasitism of hyphae and sclerotia. Mycologia 103, 483–493. doi: 10.3852/10-235
Chen, D., Toone, W. M., Mata, J., Lyne, R., Burns, G., Kivinen, K., et al. (2003). Global transcriptional responses of fission yeast to environmental stress. Mol. Biol. Cell 14, 214–229. doi: 10.1091/mbc.E02-08-0499
Chung, K.-R. (2012). Stress response and pathogenicity of the necrotrophic fungal pathogen Alternaria alternata. Scientifica 2012:635431. doi: 10.6064/2012/635431
Cubeta, M. A., Thomas, E., Dean, R. A., Jabaji, S., Neate, S. M., Tavantzis, S., et al. (2014). Draft genome sequence of the plant-pathogenic soil fungus Rhizoctonia solani anastomosis group 3 strain Rhs1AP. Genome Announc. 2, e01072–e01014. doi: 10.1128/genomeA.01072-14
Danon, A., Miersch, O., Felix, G., Camp, R. G., and Apel, K. (2005). Concurrent activation of cell death−regulating signaling pathways by singlet oxygen in Arabidopsis thaliana. Plant J. 41, 68–80. doi: 10.1111/j.1365-313X.2004.02276.x
Daub, M. E., and Ehrenshaft, M. (2000). The photoactivated Cercospora toxin cercosporin: contributions to plant disease and fundamental biology. Annu. Rev. Phytopathol. 38, 461–490. doi: 10.1146/annurev.phyto.38.1.461
Denslow, S. A., Walls, A. A., and Daub, M. E. (2005). Regulation of biosynthetic genes and antioxidant properties of vitamin B6 vitamers during plant defense responses. Physiol. Mol. Plant Path. 66, 244–255. doi: 10.1016/j.pmpp.2005.09.004
Dong, H., and Beer, S. (2000). Riboflavin induces disease resistance in plants by activating a novel signal transduction pathway. Phytopathology 90, 801–811. doi: 10.1094/PHYTO.2000.90.8.801
Ehrenshaft, M., Bilski, P., Li, M. Y., Chignell, C. F., and Daub, M. E. (1999). A highly conserved sequence is a novel gene involved in de novo vitamin B6 biosynthesis. Proc. Natl. Acad. Sci. U.S.A. 96, 9374–9378. doi: 10.1073/pnas.96.16.9374
Ehrenshaft, M., and Daub, M. E. (2001). Isolation of PDX2, a second novel gene in the pyridoxine biosynthesis pathway of eukaryotes, archaebacteria, and a subset of eubacteria. J. Bacteriol. 183, 3383–3390. doi: 10.1128/JB.183.11.3383-3390.2001
Ehrenshaft, M., Jenns, A. E., Chung, K. R., and Daub, M. E. (1998). SOR1, a gene required for photosensitizer and singlet oxygen resistance in Cercospora fungi is highly conserved in divergent organisms. Mol. Cell 1, 603–609. doi: 10.1016/S1097-2765(00)80060-X
Ellis, E. M. (2002). Microbial aldo-keto reductases. FEMS Microbiol. Lett. 216, 123–131. doi: 10.1111/j.1574-6968.2002.tb11425.x
Fitzpatrick, T. B., Amrhein, N., Kappes, B., Macheroux, P., Tews, I., and Raschle, T. (2007). Two independent routes of de novo vitamin B6 biosynthesis: not that different after all. J. Biochem. 407, 1–13. doi: 10.1042/BJ20070765
Fitzpatrick, T. B., Basset, G. J., Borel, P., Carrari, F., DellaPenna, D., Fraser, P. D., et al. (2012). Vitamin deficiencies in humans: can plant science help? Plant Cell Online 24, 395–414. doi: 10.1105/tpc.111.093120
Foley, R. C., Gleason, C. A., Anderson, J. P., Hamann, T., and Singh, K. B. (2013). Genetic and genomic analysis of Rhizoctonia solani interactions with Arabidopsis; evidence of resistance mediated through NADPH oxidases. PLoS ONE 8:e56814. doi: 10.1371/journal.pone.0056814
Gardes, M., and Bruns, T. D. (1993). ITS primers with enhanced specificity for basidiomycetes−application to the identification of mycorrhizae and rusts. Mol. Ecol. 2, 113–118. doi: 10.1111/j.1365-294X.1993.tb00005.x
Gkarmiri, K., Finlay, R. D., Alström, S., Thomas, E., Cubeta, M. A., and Högberg, N. (2015). Transcriptomic changes in the plant pathogenic fungus Rhizoctonia solani AG-3 in response to the antagonistic bacteria Serratia proteamaculans and Serratia plymuthica. BMC Genomics 16:630. doi: 10.1186/s12864-015-1758-z
González, E., Danehower, D., and Daub, M. E. (2007). Vitamer levels, stress response, enzyme activity, and gene regulation of Arabidopsis lines mutant in the pyridoxine/pyridoxamine 5′-phosphate oxidase (PDX3) and the pyridoxal kinase (SOS4) genes involved in the vitamin B6 salvage pathway. Plant Physiol. 145, 985–996. doi: 10.1104/pp.107.105189
Hall, T. A. (1999). “BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT,” in Nucleic Acids Symposium Series, 95–98.
Heller, J., and Tudzynski, P. (2011). Reactive oxygen species in phytopathogenic fungi: signaling, development, and disease. Annu. Rev. Phytopathol. 49, 369–390. doi: 10.1146/annurev-phyto-072910-095355
Hellmann, H., and Mooney, S. (2010). Vitamin B6: a molecule for human health? Molecules 15, 442–459. doi: 10.3390/molecules15010442
Herrero, S., González, E., Gillikin, J. W., Vélëz, H., and Daub, M. E. (2011). Identification and characterization of a pyridoxal reductase involved in the vitamin B6 salvage pathway in Arabidopsis. Plant Mol. Biol. 76, 157–169. doi: 10.1007/s11103-011-9777-x
Kalsotra, A., and Cooper, T. A. (2011). Functional consequences of developmentally regulated alternative splicing. Nat. Rev. Genet. 12, 715–729. doi: 10.1038/nrg3052
Lee, J., Dawes, I. W., and Roe, J.-H. (1995). Adaptive response of Schizosaccharomyces pombe to hydrogen peroxide and menadione. Microbiology 141, 3127–3132. doi: 10.1099/13500872-141-12-3127
Mittenhuber, G. (2001). Phylogenetic analyses and comparative genomics of vitamin B6 (pyridoxine) and pyridoxal phosphate biosynthesis pathways. J. Mol. Microb. Biotech. 3, 1–20.
Mooney, S., Leuendorf, J.-E., Hendrickson, C., and Hellmann, H. (2009). Vitamin B6: a long known compound of surprising complexity. Molecules 14, 329–351. doi: 10.3390/molecules14010329
Morissette, D. C., Dauch, A., Beech, R., Masson, L., Brousseau, R., and Jabaji-Hare, S. (2008). Isolation of mycoparasitic-related transcripts by SSH during interaction of the mycoparasite Stachybotrys elegans with its host Rhizoctonia solani. Curr. Genet. 53, 67–80. doi: 10.1007/s00294-007-0166-6
Morita, T., Takegawa, K., and Yagi, T. (2004). Disruption of the plr1+ gene encoding pyridoxal reductase of Schizosaccharomyces pombe. J. Biochem. 135, 225–230. doi: 10.1093/jb/mvh026
Nakano, M., Morita, T., Yamamoto, T., Sano, H., Ashiuchi, M., Masui, R., et al. (1999). Purification, molecular cloning, and catalytic activity of Schizosaccharomyces pombe pyridoxal reductase a possible additional family in the aldo-keto reductase superfamily. J. Biol. Chem. 274, 23185–23190. doi: 10.1074/jbc.274.33.23185
Nikraftar, F., Taheri, P., Rastegar, M. F., and Tarighi, S. (2013). Tomato partial resistance to Rhizoctonia solani involves antioxidative defense mechanisms. Physiol. Mol. Plant Path. 81, 74–83. doi: 10.1016/j.pmpp.2012.11.004
Ogoshi, A. (1996). “Introduction—the genus Rhizoctonia,” in Rhizoctonia Species: Taxonomy, Molecular Biology, Ecology, Pathology and Disease Control, 3rd Edn, ed B. Sneh (Dordrecht: Springer), 1–9. doi: 10.1007/978-94-017-2901-7_1
Osmani, A. H., May, G. S., and Osmani, S. A. (1999). The extremely conserved pyroA gene of Aspergillus nidulans is required for pyridoxine synthesis and is required indirectly for resistance to photosensitizers. J. Biol. Chem. 274, 23565–23569. doi: 10.1074/jbc.274.33.23565
Padilla, P. A., Fuge, E. K., Crawford, M. E., Errett, A., and Werner-Washburne, M. (1998). The highly conserved, coregulated SNO and SNZ gene families in Saccharomyces cerevisiae respond to nutrient limitation. J. Bacteriol. 180, 5718–5726.
Percudani, R., and Peracchi, A. (2003). A genomic overview of pyridoxal-phosphate-dependent enzymes. EMBO Rep. 4, 850–854. doi: 10.1038/sj.embor.embor914
Raschle, T., Amrhein, N., and Fitzpatrick, T. B. (2005). On the two components of pyridoxal 5′-phosphate synthase from Bacillus subtilis. J. Biol. Chem. 280, 32291–32300. doi: 10.1074/jbc.M501356200
Rodríguez-Navarro, S., Llorente, B., Rodríguez-Manzaneque, M. T., Ramne, A., Uber, G., Marchesan, D., et al. (2002). Functional analysis of yeast gene families involved in metabolism of vitamins B1 and B6. Yeast 19, 1261–1276. doi: 10.1002/yea.916
Roje, S. (2007). Vitamin B biosynthesis in plants. Phytochemistry 68, 1904–1921. doi: 10.1016/j.phytochem.2007.03.038
Rueschhoff, E. E., Gillikin, J. W., Sederoff, H. W., and Daub, M. E. (2012). The SOS4 pyridoxal kinase is required for maintenance of vitamin B6 mediated processes in chloroplasts. Plant Physiol. Biochem. 63, 281–291. doi: 10.1016/j.plaphy.2012.12.003
Sang, Y., Barbosa, J. M., Wu, H., Locy, R. D., and Singh, N. K. (2007). Identification of a pyridoxine (pyridoxamine) 5′-phosphate oxidase from Arabidopsis thaliana. FEBS Lett. 581, 344–348. doi: 10.1016/j.febslet.2006.12.028
Sharma, P., Jha, A. B., Dubey, R. S., and Pessarakli, M. (2012). Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012:217037. doi: 10.1155/2012/217037
Sievers, F., and Higgins, D. G. (2014). “Clustal Omega, accurate alignment of very large numbers of sequences,” in Multiple Sequence Alignment Methods, ed D. J. Russell (New York, NY: Humana Press), 105–116. doi: 10.1007/978-1-62703-646-7_6
Sneh, B. (1996). Rhizoctonia Species: Taxonomy, Molecular Biology, Ecology, Pathology and Disease Control. Dordrecht: Springer. doi: 10.1007/978-94-017-2901-7
Taheri, P., and Tarighi, S. (2011). A survey on basal resistance and riboflavin-induced defense responses of sugar beet against Rhizoctonia solani. J. Plant Physiol. 168, 1114–1122. doi: 10.1016/j.jplph.2011.01.001
Tamura, K., Peterson, D., Peterson, N., Stecher, G., Nei, M., and Kumar, S. (2011). MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 28, 2731–2739. doi: 10.1093/molbev/msr121
Vanderschuren, H., Boycheva, S., Li, K.-T., Szydlowski, N., Gruissem, W., and Fitzpatrick, T. B. (2013). Strategies for vitamin B6 biofortification of plants: a dual role as a micronutrient and a stress protectant. Front. Plant Sci. 4:143. doi: 10.3389/fpls.2013.00143
Wang, S.-L., Liang, T.-W., and Yen, Y.-H. (2011). Bioconversion of chitin-containing wastes for the production of enzymes and bioactive materials. Carbohydr. Polym. 84, 732–742. doi: 10.1016/j.carbpol.2010.06.022
Yang, Y.-F., Zhu, T., and Niu, D.-K. (2013). Association of intron loss with high mutation rate in Arabidopsis: implications for genome size evolution. Genome Biol. Evol. 5, 723–733. doi: 10.1093/gbe/evt043
Zhao, G., and Winkler, M. E. (1995). Kinetic limitation and cellular amount of pyridoxine (pyridoxamine) 5′-phosphate oxidase of Escherichia coli K-12. J. Bacteriol. 177, 883–891.
Keywords: vitamin B6, de novo pathway, salvage pathway, Rhizoctonia solani, oxidative stress, abiotic stress, PDX genes, antioxidant genes
Citation: Samsatly J, Chamoun R, Gluck-Thaler E and Jabaji S (2016) Genes of the de novo and Salvage Biosynthesis Pathways of Vitamin B6 are Regulated under Oxidative Stress in the Plant Pathogen Rhizoctonia solani. Front. Microbiol. 6:1429. doi: 10.3389/fmicb.2015.01429
Received: 13 October 2015; Accepted: 30 November 2015;
Published: 05 January 2016.
Edited by:
Helio K. Takahashi, Universidade Federal de Sao Paulo, BrazilReviewed by:
Bjorn Sandrock, University of Marburg, GermanyMarcelo Tolmasky, California State University Fullerton, USA
Copyright © 2016 Samsatly, Chamoun, Gluck-Thaler and Jabaji. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Suha Jabaji, c3VoYS5qYWJhamlAbWNnaWxsLmNh
 Jamil Samsatly
Jamil Samsatly Rony Chamoun
Rony Chamoun Emile Gluck-Thaler
Emile Gluck-Thaler Suha Jabaji
Suha Jabaji