- 1Department of Pulmonology, Southwest Hospital, Third Military Medical University, Chongqing, China
- 2State Key Laboratory of Pathogen and Biosecurity, Beijing Institute of Microbiology and Epidemiology, Beijing, China
- 3Department of Pharmacy, Southwest Hospital, Third Military Medical University, Chongqing, China
Pseudomonas aeruginosa strain 10265 was recovered from a patient with pneumonia in a Chinese public hospital, and it displays the carbapenem resistance phenotype due to the acquisition of a non-conjugative but mobilizable IncP-6-type plasmid p10265-KPC. p10265-KPC carries a Tn5563-borne defective mer locus, and a novel ΔISEc33-associated blaKPC-2 gene cluster without paired inverted repeats and paired direct repeats at both ends. Mobilization of this ΔISEc33-associated element in p10265-KPC would be attributed to homologous recombination-based insertion of a foreign structure Tn3-ISApu1-orf7-ISApu2- ISKpn27-ΔblaTEM-1-blaKPC-2-ΔISKpn6- korC-orf6-klcA-ΔrepB into a pre-existent intact ISEc33, making ISEc33 truncated at the 3′ end. The previously reported pCOL-1 represents the first sequenced KPC-producing IncP-6 plasmid, while p10265-KPC is the second one. These two plasmids carry two distinct blaKPC-2 gene clusters, which are inserted into the different sites of the IncP-6 backbone and have different evolutionary histories of assembly and mobilization. This is the first report of identification of the IncP-6-type resistance plasmid in China.
Introduction
Klebsiella pneumoniae carbapenamases (KPCs) were initially discovered in USA in 1996, and they have disseminated worldwide among Enterobacteriaceae, Pseudomonas and Acinetobacter with K. pneumoniae being the most predominate species (Munoz-Price et al., 2013; Chen et al., 2014b). At least 23 KPC protein variants (KPC-2 to KPC-24; KPC-1 is essentially identical to KPC-2) have been identified1. The blaKPC genes are typically present on plasmids, varying in size, genetic structure and incompatibility group (e.g., IncFII, FIA, I2, A/C, N, X, R, P, U, W, L/M, and ColE; Munoz-Price et al., 2013; Chen et al., 2014b). Chromosomal location of blaKPC has also been evidenced in Pseudomonas aeruginosa, indicating that the blaKPC genes can be integrated into host genome (Villegas et al., 2007; Cuzon et al., 2011). KPC-producing bacteria are resistant to almost all β-lactams and often to other classes of antibiotics (such as aminoglycosides, quinolones, and tetracyclines), resulting from the co-selection of additional resistance determinants encoded by insertion sequence-, integron- and transposon-associated mobile gene clusters and, thereby, leaves few or no antimicrobial treatment options (Munoz-Price et al., 2013; Chen et al., 2014b).
At least thirteen incompatibility groups, IncP-1 to IncP-7 and IncP-9 to IncP-14, have been recognized for the plasmids found in the natural isolates of Pseudomonas, and about one third of these plasmids characterized belong to the IncP-2 group which typically have a size > 300 kb (Sagai et al., 1976; Boronin, 1992). Plasmids belonging to IncP-2, IncP-5, IncP-7, IncP-10, IncP-12, and IncP-13 incompatibility groups have a narrow host range and cannot be transferred from Pseudomonas to Escherichia coli, while the other IncP types especially including IncP-1, IncP-4, and IncP-6 seem to have a broad host range (Sagai et al., 1976; Boronin, 1992; Xiong et al., 2013). IncP-1 corresponds to IncP in the E. coli plasmid classification system, and plasmids of this group can transfer and replicate virtually in all Gram-negative bacteria, contributing to the spread of antibiotic and heavy metal resistance (Popowska and Krawczyk-Balska, 2013).
The IncP-6 plasmids are capable of replicating in both E. coli (where there are assigned into the IncG group) and Pseudomonas. Only a few IncP-6 plasmids, such as Rms149 from P. aeruginosa (Haines et al., 2005), pCOL-1 from P. aeruginosa (Naas et al., 2013) and pRIO-5 from Serratia marcescens (Bonnin et al., 2012), have been fully sequenced, and all these plasmid have acquired various mobile genetic structures harboring resistance markers. In addition, pRSB105 from an uncultured eubacterium represents a mosaic plasmid that carries the IncP-6 backbone as well as the Rep1 replicon module, most likely contributing to the extension of plasmid’s host range (Schluter et al., 2007).
Data presented here reveal that P. aeruginosa strain 10265 harbors a novel IncP-6 resistance plasmid p10265-KPC. The complete sequence of p10265-KPC was determined and compared with other sequenced IncP-6 plasmids. p10265-KPC carries a novel ΔISEc33-associated blaKPC-2 gene cluster as well as a Tn5563-borne defective mercury resistance (mer) gene locus, providing further insights into drug resistance mechanism of the KPC-encoding IncP-6 plasmids.
Materials and Methods
Bacterial species was identified using Bruker MALDI Biotyper (Bruker Daltonics, Bremen, Germany) and 16S rRNA gene sequencing (Frank et al., 2008). The major acquired carbapenemase and extended-spectrum β-lactamase genes were detected by PCR, followed by amplicon sequencing on ABI 3730 Sequencer (Applied Biosystems, Foster City, CA, USA; Chen et al., 2015). The experimental protocols were approved by the Ethics Committee of the Third Military Medical University.
Plasmid electroporation or conjugal transfer was performed with E. coli TOP10 (LacZ-, resistant to streptomycin and tetracycline) and EC600 (LacZ-, resistant to nalidixic acid and rifampicin) being used as recipient for selection of blaKPC-positive electroporants or transconjugants, respectively (Chen et al., 2015). Transfer of the blaKPC gene on the plasmid was determined by S1-PFGE and Southern blot hybridization (Lee et al., 2012; Chen et al., 2015).
Activity of Ambler class A/B/D carbapenemases in bacterial cell extracts was determined by CarbaNP test (Dortet et al., 2012) with modifications (Chen et al., 2015). Bacterial antimicrobial susceptibility was tested by VITEK 2 (BioMérieux Vitek, Hazelwood, MO, USA) and interpreted as per Clinical and Laboratory Standards Institute guidelines (Twenty-Fourth Informational Supplement M10-S24, 2014).
Plasmid DNA was isolated from E. coli electroporant using Qiagen large construct kit (Qiagen, Hilden, Germany), and sequenced by whole-genome shotgun strategy in combination with Illumina HiSeq 2500 (Illumina, San Diego, CA, USA) sequencing technology. The contigs were assembled with Velvet, and the gaps were filled through combinatorial PCR and Sanger sequencing on ABI 3730 Sequencer. The genes were predicted with GeneMarkSTM and further annotated by BLASTP and BLASTN against UniProt and NR databases. Gene organization diagrams were drawn with Inkscape2. The complete sequence of p10265-KPC was submitted to GenBank under accession number KU578314.
Results and Discussion
Case Report
In September 2010, an 81-year-old male with hemafecia visited a public hospital in Beijing of China, and the progressive symptoms of fever, couch, and pulmonary infection were observed after hospitalization. The patient had the underlying diseases hypertension, diabetes, multiple cerebral infarction, and chronic renal insufficiency. The patient received long-term hospital care under the hospital and was bed-ridden with indwelling catheter. His symptoms of hemafecia were generally well controlled during hospitalization, but he started to suffer from the recurrent urinary tract infections since August 2013. About 2 weeks later, bacterial colonies were observed after cultivation of the urine specimens on the Mueller-Hinton agar, and the bacterial isolate designated 10265 was identified as P. aeruginosa. Based on the antimicrobial susceptibility test results, the patient received the intravenous administration with amikacin, and his symptoms associated with urinary tract infections progressively disappeared.
Overview of Plasmid p10265-KPC
Screening for the blaGES, blaKPC, blaSME, blaIMI, blaBIC, blaIMP, blaV IM, blaNDM, blaTMB, blaFIM, blaSPM, blaDIM, blaGIM, blaSIM, blaAIM, blaSMB, blaOXA, blaCTX-M, blaTEM, blaSHV, blaGES, blaPER, blaV EB, and blaOXA genes by PCR (Chen et al., 2015) indicated the presence of only blaKPC-2 (but not any of the other bla genes tested) in strain 10265. Electroporation of the plasmid DNA of strain 10265 into E. coli TOP10 generated a blaKPC-positive electroporant 10265-KPC-TOP10, but repeated attempts of plasmid conjugal transfer with E. coli EC600 being used as recipient and strain 10265 as donor failed to obtain a blaKPC-positive E. coli transconjugant. S1-PFGE followed by Southern hybridization (Chen et al., 2015) indicated the presence of a ∼40 kb plasmid, being able to hybridize with a blaKPC-specific probe (Lee et al., 2012), in both 10265 and 10265-KPC-TOP10 (data not shown).10265 and 10265-KPC-TOP10 but not TOP10 have the class A carbapenemase activity (data not shown). Strain 10265 is resistant to all the β-lactam, β-lactamase inhibitor, fluoroquinolone, and sulfonamide drugs tested but remains susceptible to aminoglycosides, while 10265-KPC-TOP10 is resistant to β-lactams and β-lactamase inhibitors but remains susceptible to all the other drugs (Table 1). Taken together, strain 10265 harbors a non-conjugative plasmid p10265-KPC, which carries the blaKPC-2 gene to mediate the resistance to β-lactams including monobactam and carbapenems.

TABLE 1. Antimicrobial resistance phenotypes of Pseudomonas aeruginosa and Escherichia coli with plasmid p10265-KPC.
The complete sequence of p10265-KPC, recovered from the 10265-KPC-TOP10 strain, was determined with a mean coverage of 124, resulting in a circular plasmid sequence of 38,939 bp with an average G + C content of 58.2% (Figure 1). Sequence annotation generated a total of 41 predicted open reading frames. The modular structure of p10265-KPC is divided into the backbone [especially including the regions for plasmid replication (repA) and stability (parABC and mob)], and three separate accessory modules (a novel blaKPC-2 gene cluster, Tn5563, and ISPa19) inserted at the different sites of the backbone.
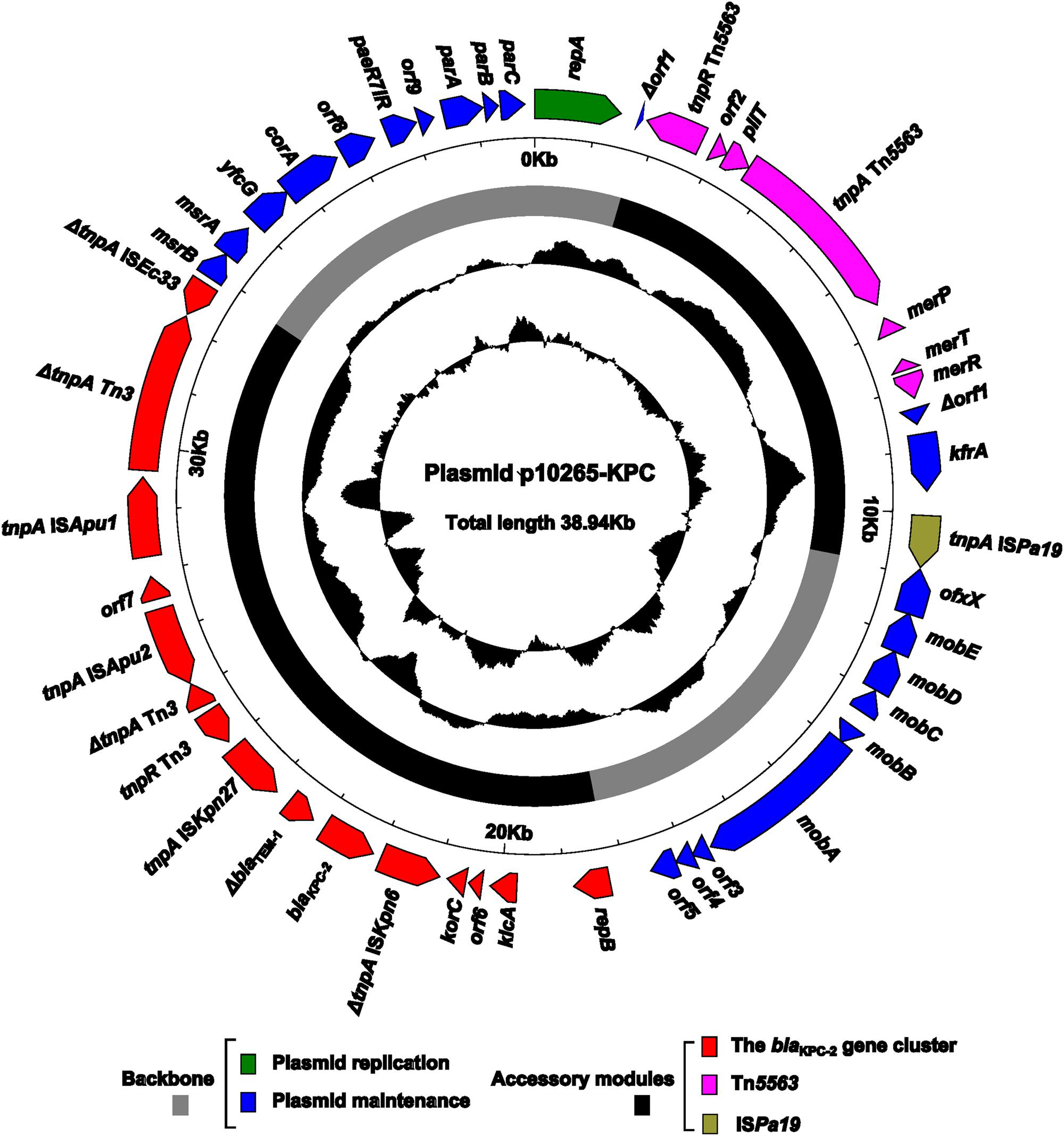
FIGURE 1. Schematic maps of p10265-KPC. Genes are denoted by arrows and colored based on gene function classification. The innermost two circles indicate the GC-Skew [(G-C)/(G + C)] and the GC content.
Accessory Modules of p10265-KPC
The rapid spread of blaKPC genes in European and American countries is linked to their location in a Tn3-family unit transposon Tn4401 with at least eight designated isoforms a to g and a separate d (Chen et al., 2012; Bryant et al., 2013; Chmelnitsky et al., 2014). Tn4401b is the prototype one and has a modular structure tnpRA (transposition core module)-ISKpn7-blaKPC-ISKpn6, which is delimited by two 39 bp inverted repeats (IRs), and the other isoforms result from distinct deletion or insertion events occurring at the different sites of Tn4401b (Chen et al., 2012; Bryant et al., 2013; Chmelnitsky et al., 2014).
In China, Tn4401 is rarely found (Ho et al., 2013) and, instead, a core module Tn3-ISKpn27-blaKPC-ΔISKpn6 is frequently identified as the blaKPC platform (Shen et al., 2009; Chen et al., 2014a,c; Li et al., 2015). For generation of the above core module, an intact ISKpn27 is inserted into the 3′-end of Tn3, and then the resulting Tn3-ISKpn27 is connected with blaKPC-2 and ΔISKpn6 (Figure 2). This core module is imbedded in two major classes of transposons (Wang et al., 2015), the Tn1722-based unit transposons [e.g., pKP048 (Shen et al., 2009)] and the IS26-based composite transposons [e.g., pKPC-LKEc (Chen et al., 2014c)].
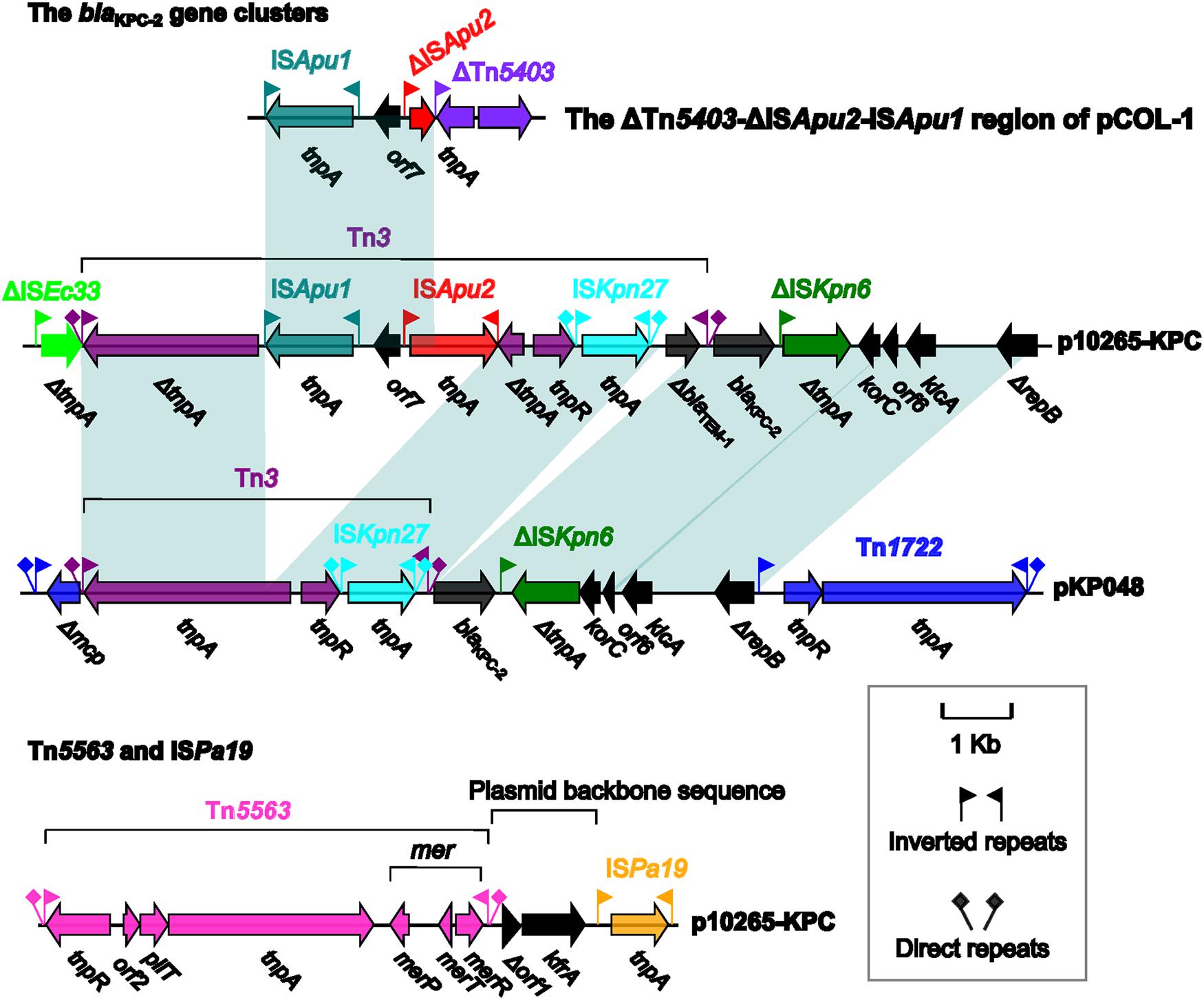
FIGURE 2. Accessory modules of p10265-KPC and comparison with related genetic contents. Genes are denoted by arrows and colored based on gene function classification. Shading regions denote shared regions of homology (>95% nucleotide similarity).
The Tn1722-based transposon in pKP048 (Shen et al., 2009) looks like a prototype one, and it is generated from the insertion of ISKpn27-blaKPC-2-ΔISKpn6-korC-orf6- klcA-ΔrepB into the mcp gene of the cryptic transposon Tn1722, making mcp to be truncated; moreover, it is bounded by 39 bp IRs and further flanked by 5 bp direct repeats (DRs: target site duplications which are usually the signature of a transposition event) at both ends (Figure 2). Before being captured by Tn1722, the above mentioned core module ISKpn27-blaKPC-2-ΔISKpn6 is connected with a gene cluster korC-orf6-klcA-ΔrepB.
In p10265-KPC, the primary genetic content Tn3-ISKpn27-blaKPC-2-ΔISKpn6 -korC-orf6-klcA-ΔrepB is also found but it has undergone two major evolutionary events: (i) the insertion of a ΔblaTEM-1 gene between ISKpn27 and the Tn3 IRR (IR right), and (ii) the insertion of ISApu1-orf7-ISApu2 into the Tn3 tnpA gene, disrupting it into two separate parts (Figure 2). The resulting structure is then connected with ΔISEc33 to finally constitute a 15.1 kb ΔISEc33-associated element ΔISEc33-Tn3-ISApu1-orf7-ISApu2-ISKpn27-ΔblaTEM-1-blaKPC-2-ΔISKpn6-korC- orf6-klcA-ΔrepB (Figure 2). In contrast to the Tn1722-based unit transposons, the ΔISEc33-associated element is not bracketed by IRs and DRs. In addition, the Tn3 in this ΔISEc33-associated element is heavily fragmented due to insertion of various elements and most likely defective in the activity of transposition. The mobilization of this ΔISEc33-associated element in p10265-KPC would attribute to homologous recombination-based insertion of a foreign element Tn3-ISApu1-orf7-ISApu2-ISKpn27-ΔblaTEM-1-blaKPC-2-ΔISKpn6-korC-orf6-klcA-ΔrepB into a pre-existent intact ISEc33 element (making it truncated at 3′ end), rather than resulting from a transposition event of the whole ΔISEc33-associated element followed by the deletion of its adjacent extremities removing IR and DR sequences.
In p10265-KPC, the 6.5 kb transposon Tn5563 is located upstream of ISPa19, with two consecutive backbone genes orf6 and kfrA as the interval between Tn5563 and ISPa19. ISPa19 contains the single transposase gene tnpA, and this insertion sequence is bordered by 18 bp IRs; this insertion sequence was initially described in plasmid Rms149 (Haines et al., 2005). Tn5563, belonging to the Tn3 subgroup of the Tn3 family transposons, was initially identified in plasmid pRA2 from P. alcaligenes and organized sequentially as tnpR (resolvase), orf2 (hypothetical protein), pliT (PilT domain-containing protein), tnpA, merP (mercuric transport protein periplasmic componen), merT (mercuric transport protein), and merR (mercuric resistance operon regulatory protein); this gene cluster is bounded by 39 bp IRs and further flanked by 5 bp (this study) or 7 bp DRs at both ends (Yeo et al., 1998). Various truncated versions of Tn5563 are also found in pOZ176 (Xiong et al., 2013) and pUM505 (Ramirez-Diaz et al., 2011) from P. aeruginosa. The Tn5563 element of p10265-KPC differs from the prototype Tn5563 of pRA2 with a 286 bp insertion (nucleotide position 7270 to 7555) between merP and merT. The above observations indicate the frequent inter-plasmid transmission of Tn5563 among Pseudomonas species.
Replication and Maintenance Determinants of p10265-KPC
Plasmid p10265-KPC belongs to the IncP-6 incompatibility group because it carries three partition genes parABC and a downstream replicase gene repA, which constitute an IncP6-type consecutive par-rep gene cluster. The parA gene encodes an ATPase, whereas parB and parC encode auxiliary partition proteins. The repA gene and the parABC locus of p10265-KPC show >97% and >99%, respectively, nucleotide sequence identity with the three IncP-6 plasmids Rms149, pRIO-5, and pCOL-1. The Rms149 RepA has been shown to confer the plasmid’s replication ability in E. coli, P. aeruginosa, and P. putida (Haines et al., 2005), while the pRIO-5 RepA is able to replicate in S. marcescens and Acinetobacter baumannii but not in P. aeruginosa (Bonnin et al., 2012). The parABC locus of Rms149 is known to promote the plasmid mobilization in E. coli (Haines et al., 2005).
All of p10265-KPC, Rms149, pRIO-5, and pCOL-1 contain a 5.6 kb MOBP family mobilization module (Francia et al., 2004), which is composed of six genes mobA (relaxase/primase fusion protein), mobB (oriT recognition-like protein), mobC (relaxosome protein), and mobD and mobE (auxiliary proteins). The study with Rms149 denotes that the mob gene cluster is functional for the plasmid mobilization in E. coli (Haines et al., 2005). In addition, the above mob gene clusters are similar to those of the IncQ plasmids pTF-FC2 and pTC-F14; as shown by the genetic analysis using these two plasmids, the minimal region essential for mobilization are mobA, mobB, and mobC, while mobD and mobE are non-essential but together they greatly enhance the mobilization frequency (van Zyl et al., 2003).
Genomic Comparison of p10265-KPC with pCOL-1
pCOL-1 represents the first sequenced KPC-producing IncP-6 plasmid (Naas et al., 2013), while p10265-KPC is the second one. The par-rep regions for partition-replication and the mob gene modules for mobilization are conserved in the p10265-KPC and pCOL-1 backbones (Figure 3), allowing these two plasmids to transfer if a conjugative plasmid is also present in the cell. The p10265-KPC and pCOL-1 backbones lack the conjugal transfer gene regions, which is in accordance with the fact that these two plasmids are unable to self-transfer their drug resistance markers via conjugation. An inversion of the backbone gene cluster msrB-msrA-yfcG-corA-orf8 in p10265-KPC turns it into Δorf8-corA′-yfcG-msrA-msrB in pCOL-1 with a truncation of orf8 and, moreover, corA becomes a pseudo gene with the accumulation of multiple indels.
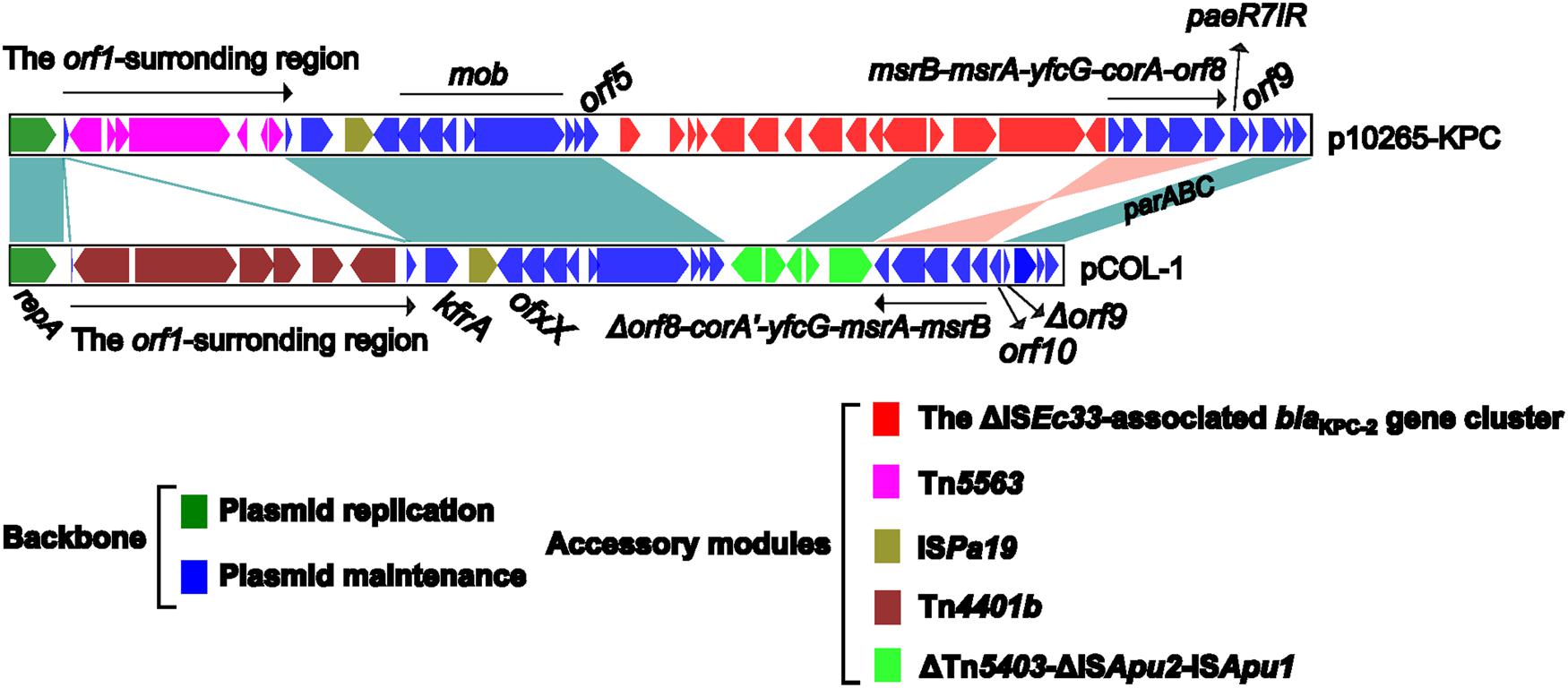
FIGURE 3. Linear comparison of sequenced plasmids p10265-KPC and pCOL-1. Genes are denoted by arrows and colored based on gene function classification. Shading regions denote shared regions of homology (>95% nucleotide similarity).
A 206 bp fragment comprising 12 copies of tandem repeat of GCGCCTGCCTTTGAGTA is inserted into the repA-orf1 intergenic region of pCOL-1 relative to p10265-KPC (Figures 3 and 4). The open reading frame orf1 is disrupted at two distinct sites, respectively, by the insertion of two different accessory elements, namely a 10 kb blaKPC-2-carrying Tn4401b in pCOL-1 and a mer locus-carrying Tn5563 in p10265-KPC (Figures 3 and 4).

FIGURE 4. orf1-surrounding regions. Shown are the orf1-surrounding DNA regions of p10265-KPC and pCOL-1 with inserted genetic contents.
pCOL-1 and p10265-KPC share the accessory element ISPa19, which is inserted between the backbone genes kfrA and ofxX (Figure 3). Downstream of the backbone gene orf5 of p10265-KPC and pCOL-1 are two distinct inserted accessory regions, namely the ΔISEc33-associated blaKPC-2 gene cluster and a 4.3 kb ΔTn5403-ΔISApu2-ISApu1 region (showing sequence similarity to the ΔISEc33-associated element), respectively (Figures 2 and 3).
Remarkably, pCOL-1 and p10265-KPC carry two distinct blaKPC-2 gene clusters, Tn4401b and the ΔISEc33-associated element, respectively; these two gene clusters are inserted at two different sites of the IncP-6 backbone and seem to have completely different evolutionary histories of genetic assembly and transposition.
Conclusion
Plasmid p10265-KPC is a novel IncP-6 resistance plasmid from P. aeruginosa, and it carries the IncP-6-type replication, partition and mobilization systems and a novel ΔISEc33-associated blaKPC-2 gene cluster accounting for carbapenem resistance. The ΔISEc33-associated element has a complex mosaic structure, which is genetically related to the previously characterized Tn1722-based blaKPC-2-carrring unit transposons (Wang et al., 2015). Besides blaKPC-2, p10265-KPC still carries a truncated blaTEM-1 gene in the blaKPC-2 gene cluster and an incomplete mer locus in Tn5563, both of which are thought defective to mediate the corresponding resistance phenotypes. KPC-2 appears to be the sole determinant of antimicrobial resistance for p10265-KPC. Epidemiological study and routine surveillance of p10265-KPC-like plasmids in China is needed.
The IncP-6 resistance plasmids are not commonly found in the natural isolates of P. aeruginosa. The accumulating complete sequences of the IncP-6 plasmids would make it possible to chart their evolutionary history and to draw the inferences about the processes that lead to these complex plasmid genomes. As seen from p10265-KPC, Rms149, pRIO-5, and pCOL-1, the IncP-6 plasmid backbones are able to integrate a wide variety of foreign genetic contents through transposition or homologous recombination mediated by the transposable elements such as insertion sequences, transposons and integrons. The IncP-6 plasmids are quite unusual in having a relatively small backbone but carrying a large amount of accessory modules which are mainly composed of mobile genetic elements and resistance determinants. It is worth elucidating whether there are specific mechanisms associated with the IncP-6 plasmids to promote the involvement of themselves in the complex processes of acquisition of foreign genetic contents.
Author Contributions
DZ, FS, and XZ designed the study. XD, WX, JF, WL, GL, HW, and FS performed experiments. DZ, XD, and FS analyzed data. XD, WX, JF, WL, GL, HW, and FS contributed reagents, materials and analysis tools. DZ, XD, FS, and XZ wrote this manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This work was funded by the Chongqing Application and Development Program (cstc2014yykfA110021), the National Basic Research Program of China (2015CB554202), and the National High-Tech Research and Development Program of China (2014AA021402).
Footnotes
References
Bonnin, R. A., Poirel, L., Sampaio, J. L., and Nordmann, P. (2012). Complete sequence of broad-host-range plasmid pRIO-5 harboring the extended-spectrum-β-lactamase gene blaBES-1. Antimicrob. Agents Chemother. 56, 1116–1119. doi: 10.1128/AAC.00480-11
Boronin, A. M. (1992). Diversity of Pseudomonas plasmids: to what extent? FEMS Microbiol. Lett. 100, 461–467. doi: 10.1111/j.1574-6968.1992.tb05740.x
Bryant, K. A., Van Schooneveld, T. C., Thapa, I., Bastola, D., Williams, L. O., Safranek, T. J., et al. (2013). KPC-4 Is encoded within a truncated Tn4401 in an IncL/M plasmid, pNE1280, isolated from Enterobacter cloacae and Serratia marcescens. Antimicrob. Agents Chemother. 57, 37–41. doi: 10.1128/AAC.01062-12
Chen, L., Chavda, K. D., Mediavilla, J. R., Jacobs, M. R., Levi, M. H., Bonomo, R. A., et al. (2012). Partial excision of blaKPC from Tn4401 in carbapenem-resistant Klebsiella pneumoniae. Antimicrob. Agents Chemother. 56, 1635–1638. doi: 10.1128/AAC.06182-11
Chen, L., Hu, H., Chavda, K. D., Zhao, S., Liu, R., Liang, H., et al. (2014a). Complete sequence of a KPC-producing IncN multidrug-resistant plasmid from an epidemic Escherichia coli sequence type 131 strain in China. Antimicrob. Agents Chemother. 58, 2422–2425. doi: 10.1128/AAC.02587-13
Chen, L., Mathema, B., Chavda, K. D., Deleo, F. R., Bonomo, R. A., and Kreiswirth, B. N. (2014b). Carbapenemase-producing Klebsiella pneumoniae: molecular and genetic decoding. Trends Microbiol. 22, 686–696. doi: 10.1016/j.tim.2014.09.003
Chen, Y. T., Lin, J. C., Fung, C. P., Lu, P. L., Chuang, Y. C., Wu, T. L., et al. (2014c). KPC-2-encoding plasmids from Escherichia coli and Klebsiella pneumoniae in Taiwan. J. Antimicrob. Chemother. 69, 628–631. doi: 10.1093/jac/dkt409
Chen, Z., Li, H., Feng, J., Li, Y., Chen, X., Guo, X., et al. (2015). NDM-1 encoded by a pNDM-BJ01-like plasmid p3SP-NDM in clinical Enterobacter aerogenes. Front. Microbiol. 6:294. doi: 10.3389/fmicb.2015.00294
Chmelnitsky, I., Shklyar, M., Leavitt, A., Sadovsky, E., Navon-Venezia, S., Ben Dalak, M., et al. (2014). Mix and match of KPC-2 encoding plasmids in Enterobacteriaceae-comparative genomics. Diagn. Microbiol. Infect. Dis. 79, 255–260. doi: 10.1016/j.diagmicrobio.2014.03.008
Cuzon, G., Naas, T., Villegas, M. V., Correa, A., Quinn, J. P., and Nordmann, P. (2011). Wide dissemination of Pseudomonas aeruginosa producing β-lactamase blaKPC-2 gene in Colombia. Antimicrob. Agents Chemother. 55, 5350–5353. doi: 10.1128/AAC.00297-11
Dortet, L., Poirel, L., and Nordmann, P. (2012). Rapid identification of carbapenemase types in Enterobacteriaceae and Pseudomonas spp. by using a biochemical test. Antimicrob. Agents Chemother. 56, 6437–6440. doi: 10.1128/AAC.01395-12
Francia, M. V., Varsaki, A., Garcillan-Barcia, M. P., Latorre, A., Drainas, C., and De La Cruz, F. (2004). A classification scheme for mobilization regions of bacterial plasmids. FEMS Microbiol. Rev. 28, 79–100. doi: 10.1016/j.femsre.2003.09.001
Frank, J. A., Reich, C. I., Sharma, S., Weisbaum, J. S., Wilson, B. A., and Olsen, G. J. (2008). Critical evaluation of two primers commonly used for amplification of bacterial 16S rRNA genes. Appl. Environ. Microbiol. 74, 2461–2470. doi: 10.1128/AEM.02272-07
Haines, A. S., Jones, K., Cheung, M., and Thomas, C. M. (2005). The IncP-6 plasmid Rms149 consists of a small mobilizable backbone with multiple large insertions. J. Bacteriol. 187, 4728–4738. doi: 10.1128/JB.187.14.4728-4738.2005
Ho, P. L., Cheung, Y. Y., Lo, W. U., Li, Z., Chow, K. H., Lin, C. H., et al. (2013). Molecular characterization of an atypical IncX3 plasmid pKPC-NY79 carrying blaKPC-2 in a Klebsiella pneumoniae. Curr. Microbiol. 67, 493–498. doi: 10.1007/s00284-013-0398-2
Lee, C. M., Liao, C. H., Lee, W. S., Liu, Y. C., Mu, J. J., Lee, M. C., et al. (2012). Outbreak of Klebsiella pneumoniae carbapenemase-2-producing K. pneumoniae sequence type 11 in Taiwan in 2011. Antimicrob. Agents Chemother. 56, 5016–5022. doi: 10.1128/AAC.00878-12
Li, G., Zhang, Y., Bi, D., Shen, P., Ai, F., Liu, H., et al. (2015). First report of a clinical, multidrug-resistant Enterobacteriaceae isolate coharboring fosfomycin resistance gene fosA3 and carbapenemase gene blaKPC-2 on the same transposon, Tn1721. Antimicrob. Agents Chemother. 59, 338–343. doi: 10.1128/AAC.03061-14
Munoz-Price, L. S., Poirel, L., Bonomo, R. A., Schwaber, M. J., Daikos, G. L., Cormican, M., et al. (2013). Clinical epidemiology of the global expansion of Klebsiella pneumoniae carbapenemases. Lancet Infect. Dis. 13, 785–796. doi: 10.1016/S1473-3099(13)70190-7
Naas, T., Bonnin, R. A., Cuzon, G., Villegas, M. V., and Nordmann, P. (2013). Complete sequence of two KPC-harbouring plasmids from Pseudomonas aeruginosa. J. Antimicrob. Chemother. 68, 1757–1762. doi: 10.1093/jac/dkt094
Popowska, M., and Krawczyk-Balska, A. (2013). Broad-host-range IncP-1 plasmids and their resistance potential. Front. Microbiol. 4:44. doi: 10.3389/fmicb.2013.00044
Ramirez-Diaz, M. I., Diaz-Magana, A., Meza-Carmen, V., Johnstone, L., Cervantes, C., and Rensing, C. (2011). Nucleotide sequence of Pseudomonas aeruginosa conjugative plasmid pUM505 containing virulence and heavy-metal resistance genes. Plasmid 66, 7–18. doi: 10.1016/j.plasmid.2011.03.002
Sagai, H., Hasuda, K., Iyobe, S., Bryan, L. E., Holloway, B. W., and Mitsuhashi, S. (1976). Classification of R plasmids by incompatibility in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 10, 573–578. doi: 10.1128/AAC.10.4.573
Schluter, A., Szczepanowski, R., Kurz, N., Schneiker, S., Krahn, I., and Puhler, A. (2007). Erythromycin resistance-conferring plasmid pRSB105, isolated from a sewage treatment plant, harbors a new macrolide resistance determinant, an integron-containing Tn402-like element, and a large region of unknown function. Appl. Environ. Microbiol. 73, 1952–1960. doi: 10.1128/AEM.02159-06
Shen, P., Wei, Z., Jiang, Y., Du, X., Ji, S., Yu, Y., et al. (2009). Novel genetic environment of the carbapenem-hydrolyzing β-lactamase KPC-2 among Enterobacteriaceae in China. Antimicrob. Agents Chemother. 53, 4333–4338. doi: 10.1128/AAC.00260-09
van Zyl, L. J., Deane, S. M., and Rawlings, D. E. (2003). Analysis of the mobilization region of the broad-host-range IncQ-like plasmid pTC-F14 and its ability to interact with a related plasmid, pTF-FC2. J. Bacteriol. 185, 6104–6111. doi: 10.1128/JB.185.20.6104-6111.2003
Villegas, M. V., Lolans, K., Correa, A., Kattan, J. N., Lopez, J. A., and Quinn, J. P. (2007). First identification of Pseudomonas aeruginosa isolates producing a KPC-type carbapenem-hydrolyzing β-lactamase. Antimicrob. Agents Chemother. 51, 1553–1555. doi: 10.1128/AAC.01405-06
Wang, L., Fang, H., Feng, J., Yin, Z., Xie, X., Zhu, X., et al. (2015). Complete sequences of KPC-2-encoding plasmid p628-KPC and CTX-M-55-encoding p628-CTXM coexisted in Klebsiella pneumoniae. Front. Microbiol. 6:838. doi: 10.3389/fmicb.2015.00838
Xiong, J., Alexander, D. C., Ma, J. H., Deraspe, M., Low, D. E., Jamieson, F. B., et al. (2013). Complete sequence of pOZ176, a 500-kilobase IncP-2 plasmid encoding IMP-9-mediated carbapenem resistance, from outbreak isolate Pseudomonas aeruginosa 96. Antimicrob. Agents Chemother. 57, 3775–3782. doi: 10.1128/AAC.00423-13
Keywords: Pseudomonas aeruginosa, KPC-2, p10265-KPC, IncP-6
Citation: Dai X, Zhou D, Xiong W, Feng J, Luo W, Luo G, Wang H, Sun F and Zhou X (2016) The IncP-6 Plasmid p10265-KPC from Pseudomonas aeruginosa Carries a Novel ΔISEc33-Associated blaKPC-2 Gene Cluster. Front. Microbiol. 7:310. doi: 10.3389/fmicb.2016.00310
Received: 10 November 2015; Accepted: 25 February 2016;
Published: 10 March 2016.
Edited by:
Gilberto Igrejas, University of Trás-os-Montes and Alto Douro, PortugalReviewed by:
Christopher Morton Thomas, University of Birmingham, UKAlain Hartmann, Institut National de la Recherche Agronomique, France
Xian-Zhi Li, Health Canada, Canada
Copyright © 2016 Dai, Zhou, Xiong, Feng, Luo, Luo, Wang, Sun and Zhou. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiangdong Zhou, eGlhbmdkb25nemhvdUAxMjYuY29t; Fengjun Sun, ZmVuZ2pfc3VuQDE2My5jb20=
 Xiaotian Dai1
Xiaotian Dai1 Dongsheng Zhou
Dongsheng Zhou Fengjun Sun
Fengjun Sun