- 1Molecular Parasitology and Vector Biology Laboratory, Department of Biological Sciences, Birla Institute of Technology and Science, Pilani, India
- 2Department of Zoology, Chaudhary Bansi Lal University, Bhiwani, India
- 3Department of Biotechnology, Chaudhary Bansi Lal University, Bhiwani, India
Anopheles mosquito midgut harbors a diverse group of endogenous bacteria that grow extensively after the blood feeding and help in food digestion and nutrition in many ways. Although, the growth of endogenous bacteria is regulated by various factors, however, the robust antibacterial immune reactions are generally suppressed in this body compartment by a heme peroxidase HPX15 crosslinked mucins barrier. This barrier is formed on the luminal side of the midgut and blocks the direct interactions and recognition of bacteria or their elicitors by the immune reactive midgut epithelium. We hypothesized that in the absence of HPX15, an increased load of exogenous bacteria will enormously induce the mosquito midgut immunity and this situation in turn, can easily regulate mosquito-pathogen interactions. In this study, we found that the blood feeding induced AsHPX15 gene in Anopheles stephensi midgut and promoted the growth of endogenous as well as exogenous fed bacteria. In addition, the mosquito midgut also efficiently regulated the number of these bacteria through the induction of classical Toll and Imd immune pathways. In case of AsHPX15 silenced midguts, the growth of midgut bacteria was largely reduced through the induction of nitric oxide synthase (NOS) gene, a downstream effector molecule of the JAK/STAT pathway. Interestingly, no significant induction of the classical immune pathways was observed in these midguts. Importantly, the NOS is a well known negative regulator of Plasmodium development, thus, we proposed that the induction of diverged immune pathways in the absence of HPX15 mediated midgut barrier might be one of the strategies to manipulate the vectorial capacity of Anopheles mosquito.
Introduction
The tropical region of the world pays a heavy toll to malaria in terms of death and economic loss. World Health Organization reported 214 million cases and 438,000 deaths from malaria in 2015 (WHO, 2015). This situation certainly warrants attention to develop effective methods for malaria prevention. A number of strategies such as, discovery of effective vaccine/drug against malaria parasite Plasmodium and numerous vector control measures are worth mentioning here. However, the worldwide reporting of drug resistance in Plasmodium is also the major challenge in this field (Farooq and Mahajan, 2004).
Arresting Plasmodium development inside the mosquito host and developing transmission blocking strategies may also be promising in controlling malaria spread among the human hosts. Plasmodium completes its sexual life cycle in mosquito and asexual cycle continues in human host. Mosquito midgut is the first organ where mosquito-parasite interactions are initiated. In the midgut, Plasmodium undergoes a series of complex developmental transitions and majority of parasites are killed during this process (Ghosh et al., 2000; Sinden and Billingsley, 2001; Pradel, 2007). It is of note that the number of Plasmodium is regulated by the ingested blood factors as well as mosquito innate immunity (Lensen et al., 1998; Michel and Kafatos, 2005; Vlachou et al., 2005; Ramiro et al., 2011; Simon et al., 2013).
Several studies revealed that wild type or laboratory-reared mosquito midgut is housed by a variety of bacteria (Minard et al., 2013; Kajla et al., 2015a) and their interaction with different development stages of Plasmodium also determines the mosquito cycle of the parasite (Touré et al., 2000; Dong et al., 2009; Cirimotich et al., 2011b; Wang et al., 2011; Boissière et al., 2012; Moreno-García et al., 2014). In addition, the type and number of bacteria also vary during different development stages and habitat of the mosquito (Wang et al., 2011; Minard et al., 2013; Kajla et al., 2015a). Researchers exploited these findings to identify the mosquito gut-specific symbionts that can regulate Plasmodium development (Boissière et al., 2012). Gram-negative bacteria (for example, Serratia marcescens, Enterobacter species such as E. amnigenus, E. cloacae, and E. sakazaki) have been identified in anophelines midgut that inhibit Plasmodium falciparum or P. vivax sporogonic development. Mosquitoes treatment with antibiotics reduced the number of midgut bacteria that, in turn, reversed their effect on Plasmodium development (Pumpuni et al., 1993, 1996; Beier et al., 1994; Gonzalez-Ceron et al., 2003; Cirimotich et al., 2011a). Interestingly, in an artificial feeding, the co-infection of bacteria with Plasmodium in field collected or lab-reared mosquitoes also reduced the number of developing parasites (Dong et al., 2009; Cirimotich et al., 2011a). Thus, understanding the mechanisms behind these regulations will provide an excellent tool to develop Plasmodium control strategies. Earlier reports indicated that the bacteria suppressed Plasmodium infection through numerous mechanisms. For example, various enzymes and toxins produced by bacteria or the mosquito immunity or reactive oxygen species (ROS) induced against bacteria might kill the parasite (Pumpuni et al., 1993; Azambuja et al., 2005; Dong et al., 2009; Cirimotich et al., 2011a).
It is noteworthy to mention that the growth of midgut endogenous bacteria is induced extensively after the blood feeding. In addition, the feeding of exogenous bacteria supplemented blood also increases bacterial burden in the mosquito midgut. However, the midgut has a remarkable capacity to manage the midgut environment in a way to minimize the deleterious effects of the increased bacterial load (Warr et al., 2008; Dong et al., 2009; Gupta et al., 2009; Meister et al., 2009; Kajla et al., 2015a). Recent studies in A. gambiae mosquito identified a mechanism that modulates the innate immunity against bolus bacteria (Kumar et al., 2010; Kajla et al., 2015a). In these mosquitoes, a heme peroxidase AgHPX15 catalyzes the crosslinking of a mucin layer at the luminal surface of the midgut epithelium. This crosslinked mucin acts like a barrier to block the interaction of bolus bacteria or bacterial elicitors with the immune reactive midgut epithelium. This mechanism also provides a protected environment for the development of Plasmodium inside the midgut. Interestingly, AgHPX15 silencing resulted in induction of key antibacterial immune genes and decreased bacterial load in the midgut (Kumar et al., 2010; Kajla et al., 2015a). In addition, the reduction of Plasmodium development was also evident in these silenced midguts. It is also noteworthy to mention that HPX15 is a unique anopheline-lineage specific gene and its orthologs are absent in insects and human, however, they are present in the genome of nineteen worldwide distributed species of Anopheles mosquito and share 65–99% amino acids identity (Kajla et al., 2015b, 2016a).
These facts prompted us to develop a hypothesis that an increased load of exogenous bacteria in HPX15 silenced anopheline midguts will markedly induce the mosquito immunity due to the uninterrupted interactions of bacterial elicitors with the immune reactive midgut epithelium. Exploitation of this mechanism will be helpful to manipulate the vectorial capacity of the insect host. Thus, in the present study, we analyzed the mechanism of immunomodulation in the AsHPX15 silenced Anopheles stephensi midguts to understand the overview of microbial interactions with the mosquito defense system in this body compartment.
Materials and Methods
Rearing of Mosquitoes
Anopheles stephensi mosquitoes were reared in insectory at 28°C, 80% relative humidity (RH) and 12 h light:dark cycles as described before (Kajla et al., 2016b). Larvae were fed on a mixture of dog food (PetLovers Crunch), fish food (Toya), and tetramine (in 1:1:1 ratio). Adults were fed on 10% sucrose solution ad labitum. To maintain mosquito colony, adult females were also fed on anesthetized mice and the egg laid by them were collected in moist conditions. The use of animals in the study was approved by the Institutional animal ethics committee.
Bacteria Feeding to the Mosquitoes and Collection of Tissues
Wild type Escherichia coli (MTCC 40) and Micrococcus luteus (MTCC 106) bacteria were obtained from the Microbial Type Culture Collection (MTCC), Institute of Microbial Technology (IMTECH), Chandigarh, India and used throughout the study. Bacteria were grown separately in LB media following standard protocols (Gupta et al., 2009). The growth of bacteria in culture was estimated by reading the absorbance at 600 nm. 1.5 ml of each bacterium culture (A600 = 0.5) was mixed and centrifuged at 2300 × g for 5 min. The cell pellet was washed twice with Ashburner’s PBS (3 mM Sodium chloride, 7 mM Disodium hydrogen phosphate, and 3 mM Sodium dihydrogen phosphate, pH 7.2) and finally suspended in 3 ml fresh mouse blood (109 bacterial cells/ml blood). Overnight starved adult A. stephensi females were allowed to feed on mouse blood supplemented with or without bacterial suspension through an artificial membrane feeder as before (Dong et al., 2006; Gupta et al., 2009; Kumar et al., 2010). After different time of blood feeding the mosquito midguts were dissected, collected in RNAlater and stored at -80°C. Sugar fed midguts served as controls and were also collected in the same way.
RNA Isolation, cDNA Synthesis, and Real Time PCR
Total RNA was isolated from the midgut tissues using the RNAeasy Mini kit (Qiagen, Cat no. 74104) and following manufacturer’s instructions. First-strand cDNA was synthesized using Quantitech reverse transcription kit (Qiagen, Cat no. 205311). The expression of various genes (as mentioned in the Table 1) was analyzed through qPCR using Sybr Green Master Mix (Biorad Cat no. 170-8882) and normalized against the constitutively expressed S7 ribosomal protein gene as before (Kajla et al., 2016a). The PCR was initiated with denaturation at 95°C for 3 min followed by 35 additional cycles at 94°C for 10 s, 57°C for 30 s and 72°C for 50 s. The final extension was carried at 72°C for 10 min and ΔΔCt method was used to calculate the relative expressions of the target genes in the samples against the controls as discussed before (Livak and Schmittgen, 2001).
dsRNA Synthesis and Gene Silencing
LacZ (218bp) or AsHPX15 (428bp) was cloned in TOPO 2.1 vector and PCR amplified using M13F, 5′GTAAAACG ACGGCCAGT3′ and T7-M13R, 5′CTCGAGTAATACGAC TCACTATAGGGCAGGAAACAGCTATGAC3′ primers with the following conditions; 94°C for 5 min, 40 cycles of 94°C for 30 s, 55°C for 30 s, 72°C for 30 s, and final extension was carried at 72°C for 10 min. Amplicons were purified with the QIAquick PCR Purification Kit (Qiagen, Cat No 28104). PCR-purified amplicons tailed with T7 promoter sequences were used to synthesize dsRNA with the MEGAscript kit (Ambion) following the manufacturer’s instructions. For dsRNA mediated interference (RNAi), 1–2 days old female mosquitoes were injected with 69 nl of 3 μg/μl dsLacZ (controls) or dsAsHPX15 (silenced) RNA into their thoraxes using a nanojector (Drummond, Broomall, PA, USA). Five days after the injection, mosquitoes were allowed to feed on mouse blood supplemented with or without 109 bacterial cells/ml as discussed above. Tissue collection and gene specific PCR was carried as mentioned above.
Statistical Analysis of the Data
All the data were expressed as mean ± standard deviation. Statistical significance between test and respective controls was analyzed by t-test using GraphPad Prism 5.0 software (Motulsky, 1999). The data with P < 0.05 was considered significant.
Results
Growth Pattern of Exogenous Fed Bacteria in Mosquito Midgut
Mosquito midgut is housed by a diverged group of naturally acquired (endogenous) bacteria. The majority of these bacteria are Gram-negative and their growth is induced after blood feeding (Rani et al., 2009; Wang et al., 2011; Boissière et al., 2012; Chavshin et al., 2012). In parallel, the blood feeding also induces the formation of peritrophic matrix, an acellular barrier that separates blood bolus from the midgut epithelium (Kumar et al., 2010; Injera et al., 2013). Additionally, a heme peroxidase HPX15 crosslinked mucin barrier on the luminal surface of the epithelium also help to maintain a low immunity zone in the blood fed midgut to support the growth of endogenous bacteria (Kumar et al., 2010; Kajla et al., 2015a). We were interested to understand the relationship between the growth of exogenous fed bacteria and midgut immune responses in the presence of A. stephensi AsHPX15 crosslinked mucin barrier. For that, the adult females were fed on blood supplemented without or with a mixture of Gram+ (M. luteus) and Gram- (E. coli) bacteria and the growth kinetics of midgut bacteria was analyzed. Results presented in Figure 1 revealed that the relative levels of 16S rRNA for endogenous bacteria increased 25-fold after 3 h of blood feeding when compared to the sugar fed midguts (p = 0.010). Afterward, the relative levels of endogenous bacteria were 1900, 133, and 270 times after 6, 12, and 18 h of post blood feeding, respectively, against the sugar fed controls (Figure 1). This showed significant reduction in the levels of endogenous bacteria at 12 h (p = 0.0008) and 18 h (p = 0.0011) when compared to the 6 h blood fed midguts. Furthermore, the endogenous bacteria levels increased 7000 times in 24 h blood fed midguts in comparison to the sugar fed midguts (Figure 1). These results are in agreement with previous findings where an increase in endogenous bacteria was observed after 6 h of blood feeding and that further followed the similar growth pattern (Luckhart et al., 1998; Kumar et al., 2010).
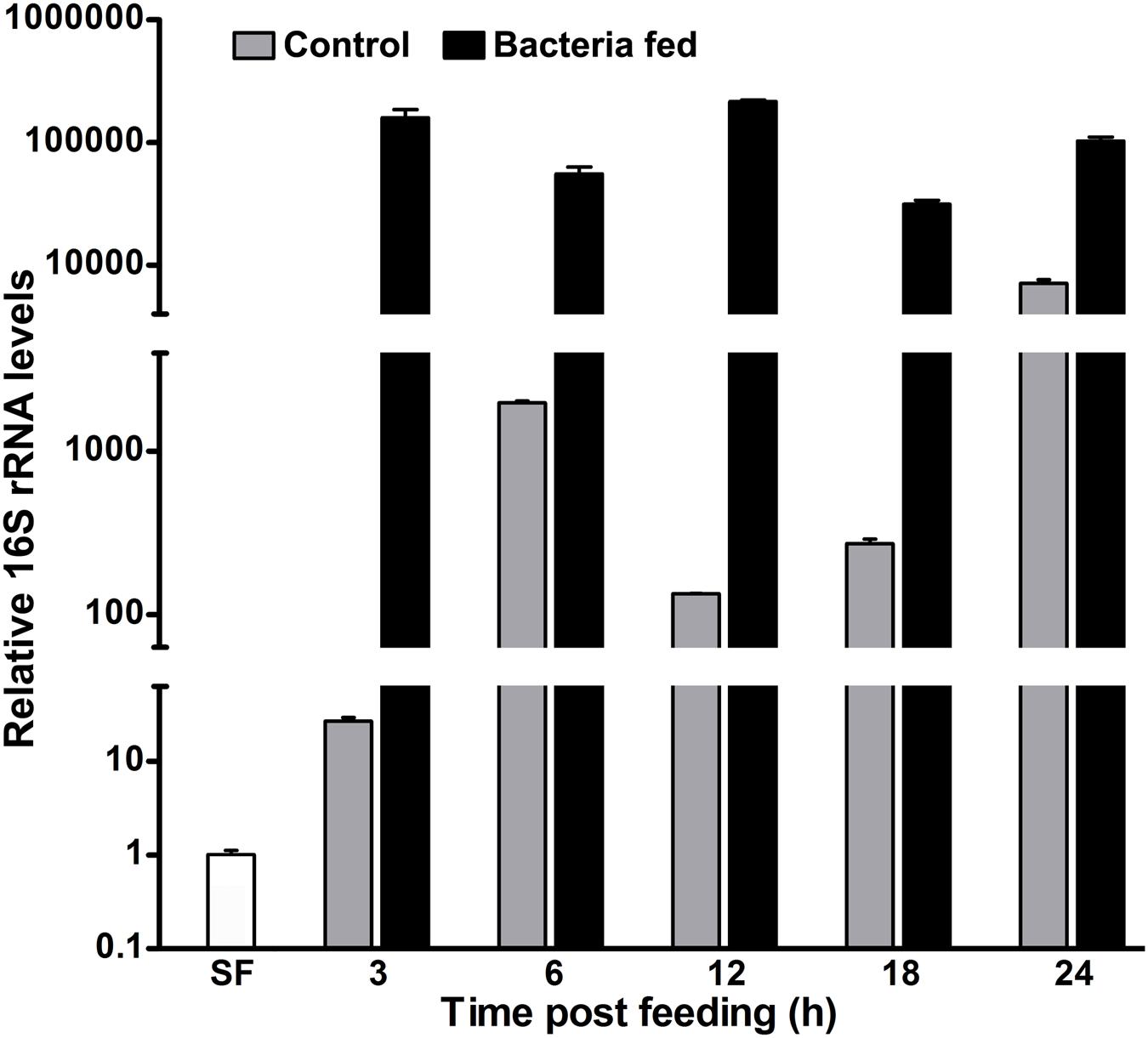
FIGURE 1. The kinetics of 16S rRNA levels in blood fed mosquito midguts. Mosquitoes were fed either on blood alone (controls) or supplemented with a mixture of M. luteus and E. coli bacteria (109 cells/ml blood). The relative levels of 16S rRNA were analyzed in the pool of mosquito midguts collected at different time intervals after the feeding and represented here in log10 scale. SF represents the 16S rRNA levels of sugar fed midguts.
Parallelly, in case of exogenous bacteria fed midguts, the relative levels of 16S rRNA were 150,000-fold at 3 h in comparison to the sugar fed midguts (Figure 1). Interestingly, the relative 16S rRNA levels were indifferent at 6 h (p = 0.0645) and 12 h (p = 0.1696), respectively, when compared to 3 h post bacteria fed midguts. However, the expression levels of 16S rRNA gene at 18 h were reduced fivefold (p = 0.0041) against 3 h post fed midguts (Figure 1). Thus, these altered levels of 16S rRNA at different time points indicated that the growth of bacteria is regulated rhythmically, most probably by the midgut immunity.
It is noteworthy to mention that in our study, we fed a large amount of bacteria (109 bacterial cells/ml blood) to the mosquitoes. The comparative levels of 16S rRNA revealed that the exogenous bacteria fed midguts have 28- and 15-fold higher bacteria load at 6 and 24 h, respectively, when compared to the just blood fed midguts (Figure 1). The reduction in the folds ratio at 24 h seems to be due to the decreased levels of 16S rRNA in exogenous bacteria fed midguts and increased levels of endogenous bacteria in the corresponding blood fed controls. This might indicate that the midgut environment is particularly favorable for the growth of endogenous bacteria in comparison to the exogenous fed microbes.
AsHPX15 Gene is Induced in Exogenous Bacteria Fed Midguts
Previously, we found that the heme peroxidase HPX15 gene is highly induced in A. gambiae or A. stephensi blood fed midguts (Kumar et al., 2004; Kajla et al., 2016a). This molecule is important to suppress the immune activation and facilitating the growth of endogenous bacteria in the blood fed midguts (Kumar et al., 2010; Kajla et al., 2015a). We also analyzed the expression kinetics of AsHPX15 gene in those midguts that are mentioned in Figure 1. We found that AsHPX15 gene was induced 23- and 27-fold at 6 and 12 h post blood feeding, respectively, when compared to the sugar fed midguts (Figure 2). The induction of AsHPX15 gene in 6 h post bacteria supplemented blood fed midguts was similar to the blood fed controls (p = 0.6485). However, its expression levels were reduced drastically at 12 and 18 h in bacteria fed midguts against blood fed controls (p = 0.0018 for 12 h and p = 0.0038 for 18 h). Interestingly, the relative AsHPX15 mRNA levels were higher in 3 and 24 h post bacteria fed midguts against the blood fed controls. Thus, the heavy load of exogenous bacteria in the midgut had no effect on the early phase of AsHPX15 gene expression (Figure 2). This may be simply due to the regulation of this gene by ecdysone hormone, which is induced in blood fed mosquitoes as reported before (Kajla et al., 2016a). We also believed that AsHPX15 catalyzed mucin barrier might be completed or its formation started within the first 12 h of post blood feeding because the presence of mucin and AgHPX15 protein is detected at the luminal surface of A. gambiae midgut epithelium by this time (Kumar et al., 2010; Injera et al., 2013). On the other hand, if the mucin barrier formation is completed and it suppresses the midgut antibacterial immunity, then the over growth of exogenous bacteria might be lethal for the mosquito. However, we found that the mortality in blood fed controls and exogenous bacteria fed mosquitoes was indifferent. This indicated that mosquito gut immunity is capable of counteracting the increased load of bacteria without compromising its survival.
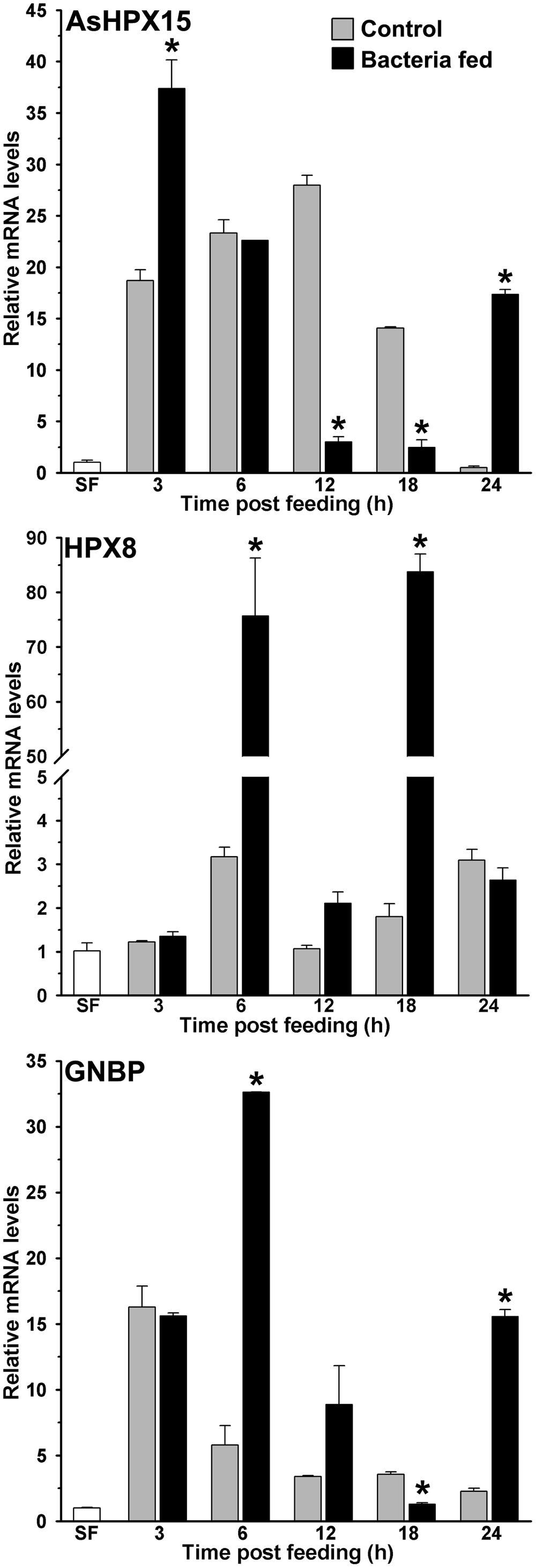
FIGURE 2. Expression kinetics of heme peroxidases and pattern recognition receptor (PRR) in bacteria fed midguts. The kinetics of relative mRNA levels of AsHPX15, HPX8, or GNBP gene were analyzed in the blood alone (controls) or supplemented with a mixture of M. luteus and E. coli bacteria fed midguts. SF represents the mRNA levels of these genes in sugar fed midguts. Significant differences in the gene expressions between control and bacteria fed midguts are denoted by asterisks.
The Antibacterial Heme Peroxidase HPX8 is Induced in Exogenous Bacteria Fed Midguts
Our results revealed that the numbers of exogenously fed bacteria not only increase in the mosquito midgut, it is also regulated at different time points (Figure 1). Thus, we hypothesized that some of the immune genes might be controlling the overgrowth of bacteria in these midguts. To test this concept, we analyzed the expression of a heme peroxidase HPX8, which has been reported to be an antibacterial gene in A. gambiae (Kumar et al., 2010). Results presented in Figure 2 revealed that HPX8 mRNA is induced 25- and 42-fold at 6 and 18 h, respectively, in bacteria fed midguts against the blood fed controls (p = 0.0206 for 6 h and p = 0.0015 for 18 h), however, at other time points, HPX8 mRNA levels in the test and controls were indifferent. This induction pattern of HPX8 gene (Figure 2) corroborated with the bacterial 16S rRNA levels in these midguts (Figure 1). These findings indicated that HPX8 might be one of the candidate genes that participate in the midgut antibacterial immunity.
Classical Immune Pathways are Induced in Exogenous Bacteria Fed Midguts
In insects, Gram-negative binding protein (GNBP) acts like the pattern recognition receptor (PRR) and triggers classical innate immunity through Toll or Imd pathway against Gram-positive or Gram-negative bacteria, respectively (Lemaitre et al., 1995; Michel et al., 2001; Osta et al., 2004; Waterhouse et al., 2007). In anopheline mosquitoes, several isoforms of GNBP have been identified (Dimopoulos et al., 1998; Christophides et al., 2002; Osta et al., 2004; Warr et al., 2008) thus, we designed a set of common primers to detect the expression of any given isoform of GNBP in our study (primer sequences are provided in Table 1). Our analyses revealed that mRNA levels of GNBP were induced ∼17-fold after 3 h of blood feeding when compared to the sugar fed midguts and that remained unaffected by the presence of bacteria in the blood (Figure 2). However, at 6 and 12 h post bacteria feeding, the relative mRNA levels of GNBP were six and two folds higher, respectively, against the normal blood fed controls (Figure 2). Furthermore, the relative mRNA levels of GNBP were seven folds higher in 24 h post bacteria fed midguts in comparison to the blood fed controls (Figure 2, p = 0.0019). These findings indicated that GNBP induction represents two episodes, an early event around 6 h and a late phase expression around 24 h post bacteria feeding. Thus, to further explore the immune regulatory mechanism that balances the mosquito midgut immunity against exogenous bacteria, these two time points were further analyzed in detail.
We studied the induction of Toll and Imd pathways in the above-mentioned samples those were collected at early 6 h and late 24 h post feeding. The gene expression analyses revealed that the Toll mRNA levels were ∼three folds higher in the 6 h post bacteria fed midguts in comparison to the blood fed controls (Figure 3, p = 0.036). However, the induction of this gene was indifferent at 24 h in these midguts (p = 0.563). In addition, we also analyzed the expression of gambicin in these samples. Gambicin is an effector antimicrobial peptide, which is regulated by both Toll and Imd pathways (Vizioli et al., 2001). The results presented in Figure 3 revealed that gambicin was induced 10-folds higher in 6 h post bacteria fed midguts when compared to the blood fed controls (p = 0.004). Although, gambicin expression is further induced in 24 h post bacteria fed midguts, however, these levels were similar to the blood fed controls (p = 0.294). Because gambicin is effective against both Gram+ and Gram- bacteria, therefore, its expression at early and late time points corroborate with the increased number of midgut bacteria (compare Figures 1 and 3). In conclusion, the induction of classical Toll and Imd immune pathways regulate the growth of bacteria in blood fed midguts.
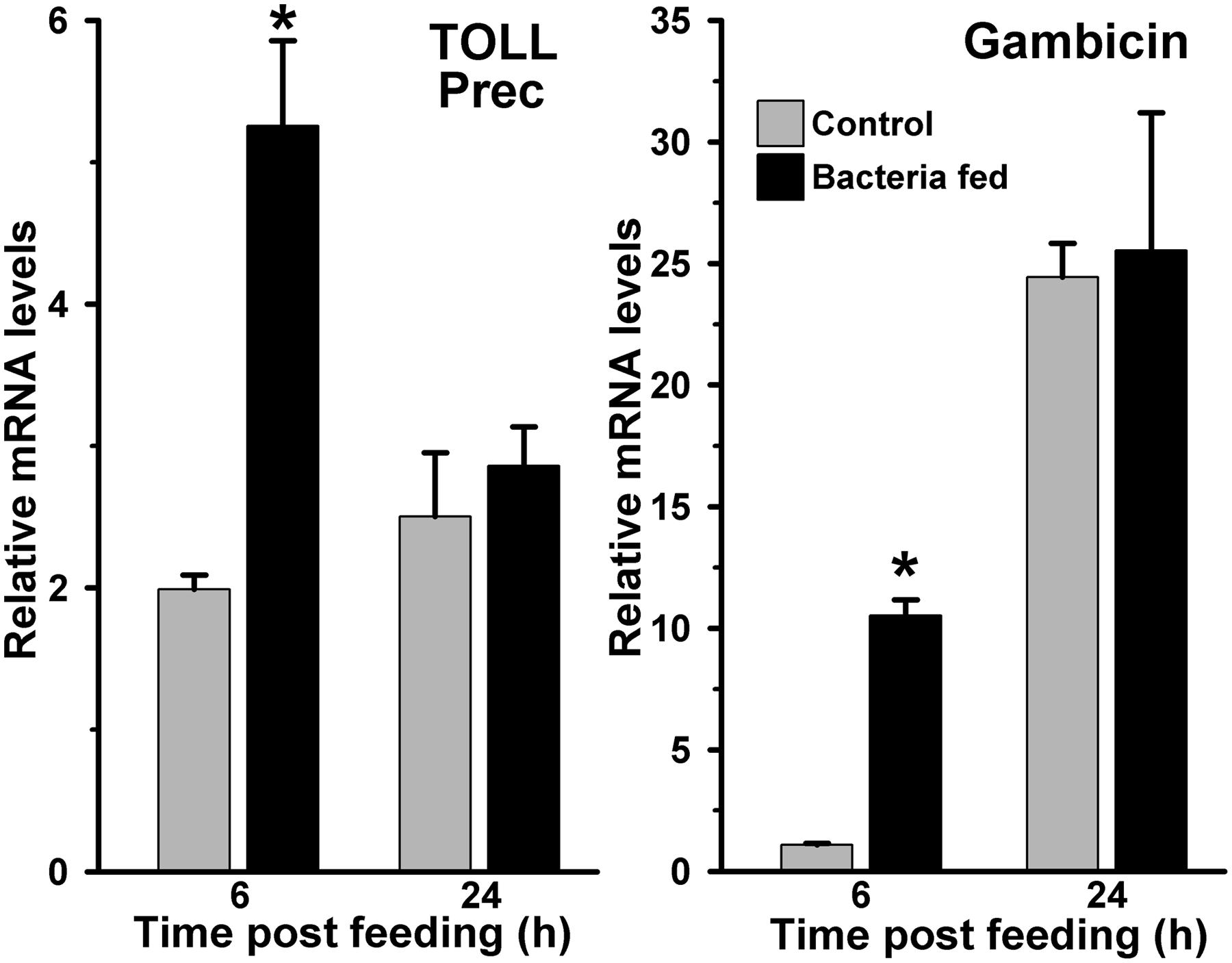
FIGURE 3. Expression of Toll and gambicin immune genes in bacteria fed Anopheles stephensi midguts. mRNA levels of midgut Toll (Toll precursor) or gambicin gene were analyzed at 6 and 24 h after feeding the blood alone (controls) or supplemented with a mixture of E. coli and M. luteus. Relative levels of mRNA are presented against the sugar fed midguts. Significant differences in the gene expression levels are denoted by an asterisk.
JAK/STAT Pathway is Not Induced in Exogenous Bacteria Fed Midguts
Another mosquito immune pathway, known as JAK/STAT, is also reported to be involved in gut immunity against a variety of pathogens (Gupta et al., 2009; Kumar et al., 2010). In this pathway, the receptor mediated activation of tyrosine kinases (JAKs) initiates the phosphorylation of cytosolic STAT (Signal transducer and activator of transcription) proteins. The phosphorylated STATs make dimer and upon translocation into the nucleus, they regulate the expression of numerous immune genes. One of such STAT-mediated effector gene nitric oxide synthase (NOS) is reported to play a crucial role in controlling the pathogenic development in Anopheles mosquito (Gupta et al., 2009; Kumar et al., 2010; Bahia et al., 2011). We were also interested to understand the activation of STAT pathway in exogenous bacteria fed midguts. Results presented in Figure 4 revealed that the NOS mRNA levels in 6 and 24 h post bacteria fed midguts were indifferent from the blood fed midguts (p = 0.399 at 6 h and p = 0.123 at 24 h). Collectively, these results suggested that NOS is not induced in the midgut against exogenous fed bacteria. Thus, we believed that the STAT pathway does not participate in A. stephensi midgut antibacterial immunity and confirms the previous findings as reported in other mosquitoes (Agaisse and Perrimon, 2004; Gupta et al., 2009; Kumar et al., 2010).
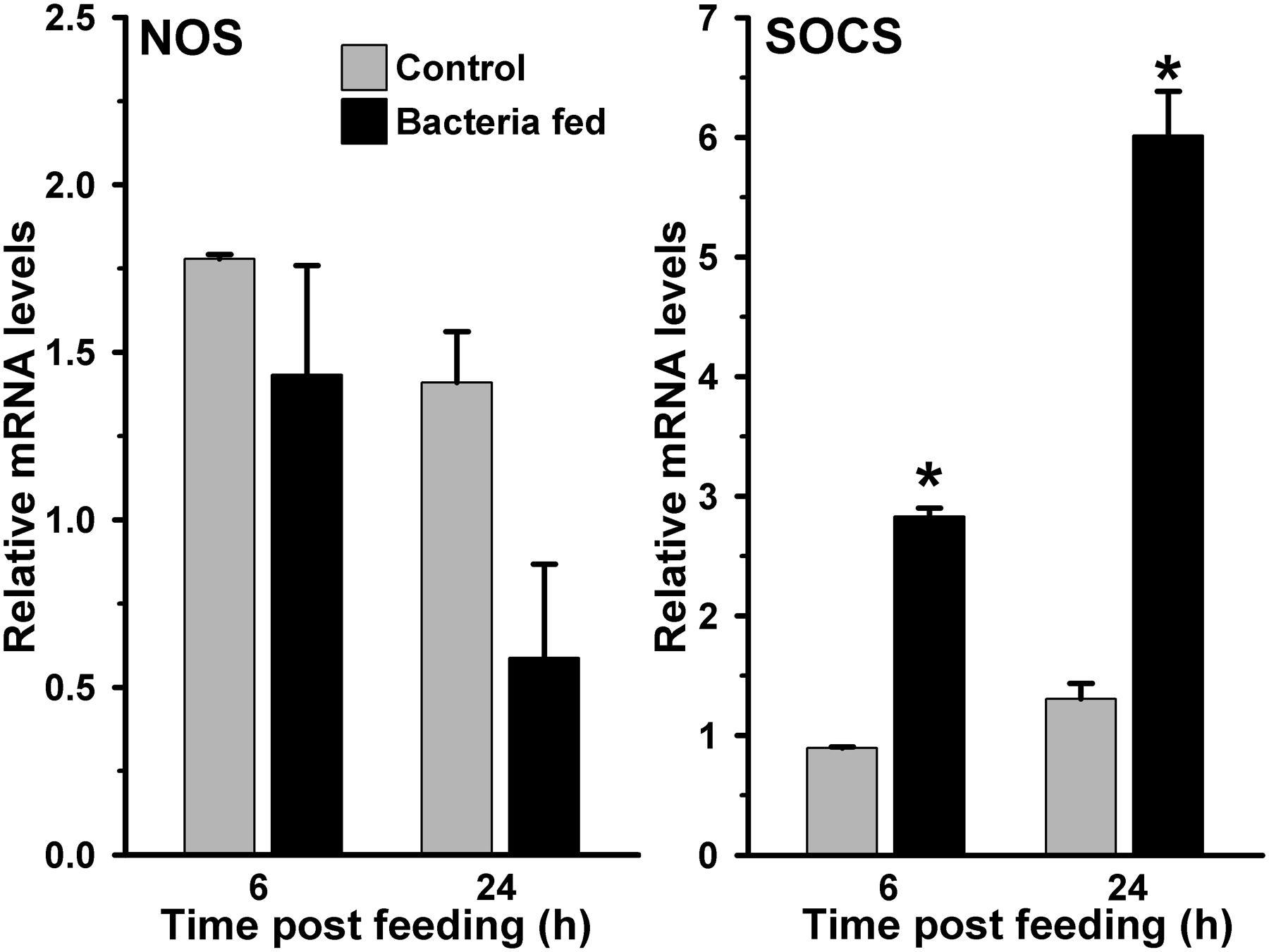
FIGURE 4. Expression of NOS and SOCS immune genes in bacteria fed A. stephensi midguts. Midgut mRNA levels of NOS or SOCS gene were analyzed at 6 and 24 h after feeding the blood alone (controls) or supplemented with a mixture of E. coli and M. luteus. Relative levels of mRNA are presented against the sugar fed midguts. Significant differences in the gene expression levels are shown by asterisks.
We hypothesized that either the STAT pathway in above-mentioned midguts remain uninduced or it is suppressed by the bacteria through some specific mechanism. For that, we analyzed the expression of the suppressor of cytokine signaling (SOCS) gene, which is a downstream feedback repressor of the STAT pathway (Wormald and Hilton, 2004; Gupta et al., 2009; Dhawan et al., 2015). Interestingly, the SOCS is induced ∼four and ∼five folds at 6 and 24 h, respectively, in the bacteria fed midguts against blood fed controls (Figure 4, p = 0.001 for 6 h and p = 0.006 for 24 h). These results revealed that the induction of SOCS gene in bacteria fed midguts might be responsible for inhibiting the activation of STAT pathway and NOS expression in a way similar to other systems (Zhang et al., 2011; Demirel et al., 2013; Maehr et al., 2014).
AsHPX15 Silencing Suppressed the Growth of Exogenous Bacteria in the Midgut
We were interested to understand the mosquito midgut immune regulation against high load of the exogenous bacteria when HPX15 crosslinked mucin barrier formation is suppressed. For that, we silenced AsHPX15 gene through dsRNA-mediated interference as discussed in Materials and Methods. Silenced mosquitoes were fed on blood supplemented with bacteria (a mixture of M. luteus and E. coli). The midguts were collected at early 6 h and late 24 h post feeding to analyze the expression profiling of the immune genes as mentioned above. Our analysis of AsHPX15 mRNA levels in controls and silenced midguts revealed that this gene was silenced in the range of 80–90% (Figure 5). The relative 16S rRNA levels in controls and silenced midguts were similar at 6 h post feeding (p = 0.053). However, at 24 h post feeding there was 63% reduction in the 16S rRNA levels in the silenced midguts against the unsilenced controls (Figure 5, p = 0.0004). Thus, AsHPX15 silencing reduces the overall growth of bacteria in the midgut bolus that might include the endogenous as well as exogenous bacteria. These findings are in partial agreement with previous reports where silencing of A. stephensi AsHPX15 ortholog drastically reduced the growth of endogenous bacteria in A. gambiae midguts (Kumar et al., 2010; Kajla et al., 2015a).
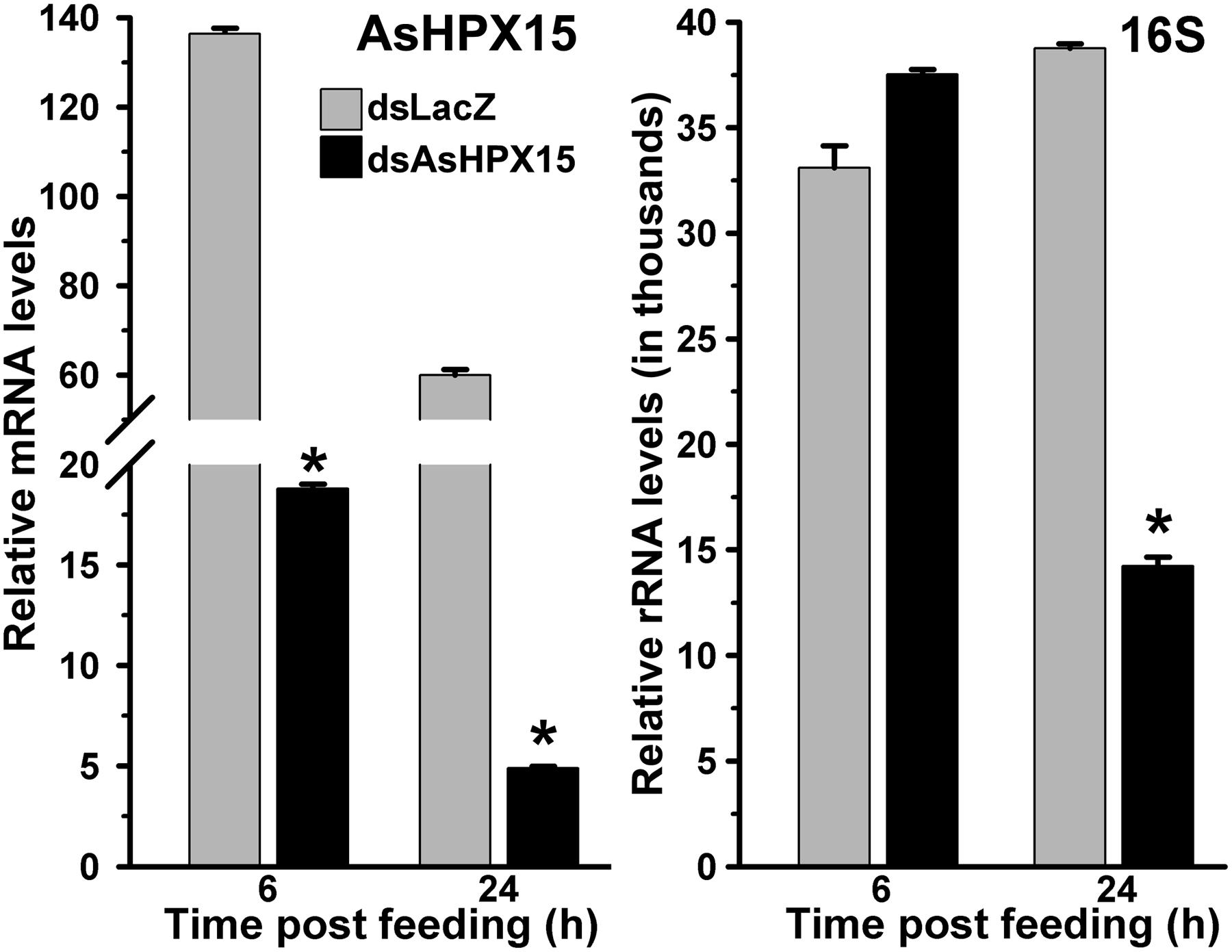
FIGURE 5. Relative levels of AsHPX15 mRNA and 16S rRNA in AsHPX15 silenced and bacteria fed midguts. Mosquitoes injected with dsLacZ (controls) or dsAsHPX15 (silenced) RNA were fed on bacteria supplemented blood and relative levels of AsHPX15 mRNA and 16S rRNA were analyzed in their midguts at different time intervals after feeding. Relative mRNA levels are presented against the sugar fed midguts. Significant differences are shown by asterisks.
Classical Antibacterial Immune Genes are Suppressed in AsHPX15 Silenced Midguts
To understand the immune regulation of bacteria in the AsHPX15 silenced midguts, we analyzed the expression profile of important classical immune pathways. Results presented in Figure 6 revealed that the mRNA levels of the PRR GNBP were reduced significantly in silenced midguts at 6 h (p = 0.005) and 24 h (p = 0.0111) post bacteria feeding, when compared to their respective unsilenced controls. Thus, the reduced levels of GNBP expression in silenced midguts might reflect the compromised situation for the recognition and induction of classical immune pathways. This fact was further confirmed after analyzing the gene expression of Toll and Imd pathways in these samples. We found that the levels of Toll mRNA were indifferent in silenced and control midguts after 6 h of bacteria feeding (Figure 6, p = 0.123). However, Toll mRNA levels were suppressed seven folds in the silenced midguts than controls at 24 h post bacteria feeding (p = 0.0209). These results revealed that Toll pathway is not induced in AsHPX15 silenced midguts neither at early nor at late stage of post bacteria feeding. Furthermore, we analyzed the expression of gambicin in these AsHPX15 silenced samples. We observed that the expression of gambicin gene was significantly downregulated in the silenced midguts after 6 h post feeding against controls (Figure 6, p = 0.023). Interestingly, at 24 h post feeding the expression of gambicin gene was induced ∼three folds in silenced midguts in comparison to the controls (p = 0.004). These findings suggested that the induction of gambicin in 24 h silenced midguts might be regulated by some other mechanism(s) that is different from classical Toll and Imd pathways.
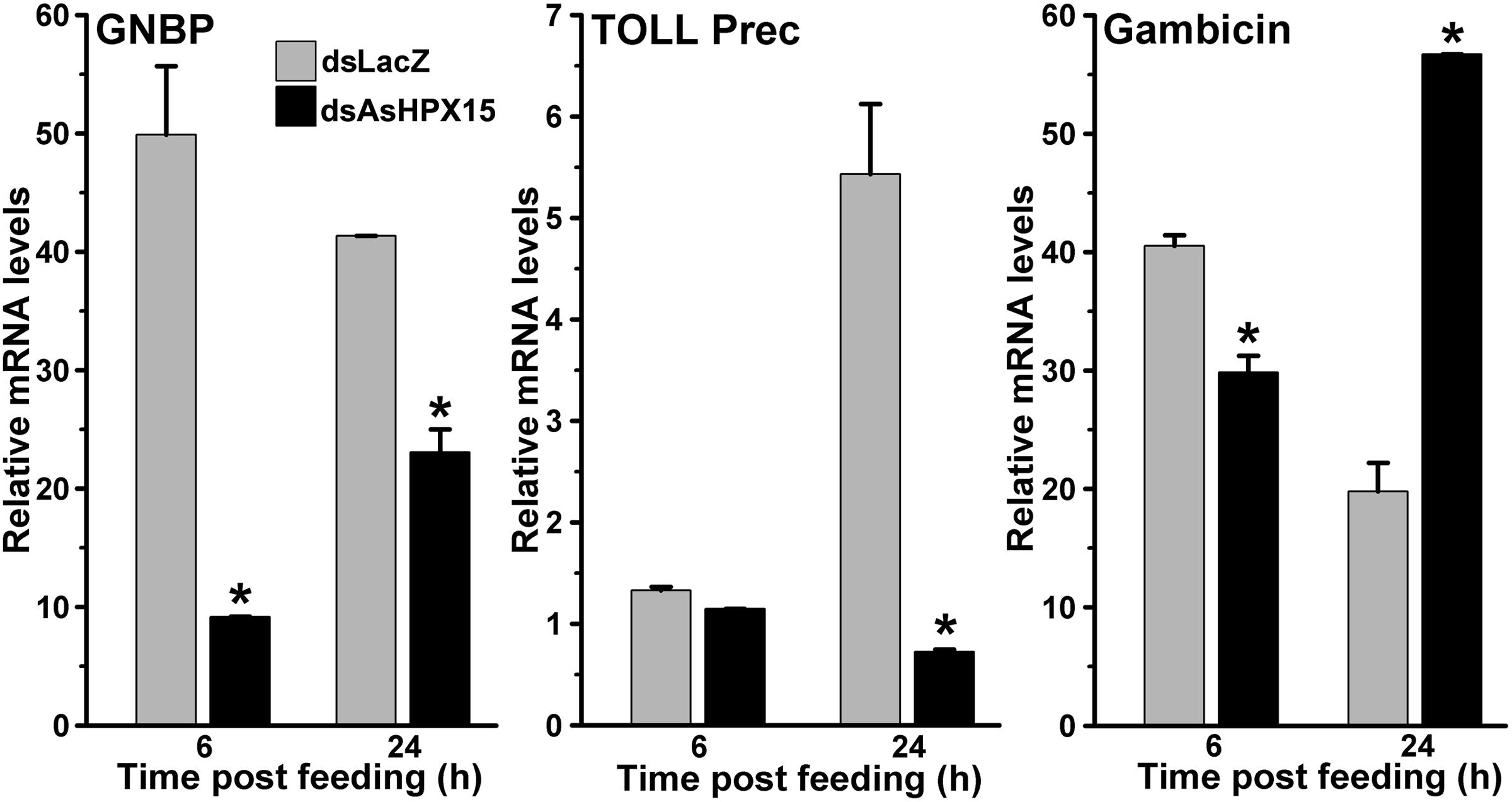
FIGURE 6. Expression of classical immune pathway genes in AsHPX15 silenced and bacteria fed midguts. Mosquitoes injected with dsLacZ (controls) or dsAsHPX15 (silenced) RNA were fed on bacteria supplemented blood and relative mRNA levels of GNBP, Toll (Toll precursor) or gambicin gene were analyzed in their midguts after 6 and 24 h post feeding. Relative levels of mRNA are presented against the sugar fed midguts. Significant differences between dsLacZ and dsAsHPX15 samples are shown by an asterisk.
JAK/STAT Pathway is Induced against Exogenous Bacteria in AsHPX15 Silenced Midguts
We further examined the JAK/STAT pathway genes in AsHPX15 silenced midguts to understand the involvement of this pathway in antibacterial immunity. For that, we compared the expression of STAT pathway genes in silenced or sham treated mosquito midguts after bacteria supplemented blood feeding. Results presented in Figure 7 revealed that NOS and SOCS genes were induced 15- and 3-folds, respectively, in the silenced midguts after 6 h of bacteria feeding when compared to the controls (p = 0.0002 for NOS and p < 0.0001 for SOCS expression). However, the expressions of both these genes in the silenced midguts were reduced 4- and 1.3-folds, respectively, after 24 h post feeding against unsilenced controls (p < 0.0001 for NOS and p = 0.068 for SOCS expression). This pattern of NOS and SOCS expression indicated the activation of STAT pathway in silenced midguts. Interestingly, the profile of NOS expression also corroborated with the levels of bacteria in the silenced midguts (Comparing Figures 5 and 7). We believed that the induced NOS during the early hours (6 h, Figure 7) might be responsible for reducing the levels of bacteria at later time points (24 h, Figure 5) as this gene is reported to play an antibacterial role in mosquitoes as well as other insects (Hillyer and Estévez-Lao, 2010). These results collectively revealed that STAT pathway is actively engaged in antibacterial immunity in the AsHPX15 silenced A. stephensi midguts. This effect might be due to the reduction of AsHPX15 catalyzed mucin barrier crosslinking and that, in turn, allows direct interaction of bacteria or bacterial elicitors with immune reactive midgut epithelium.
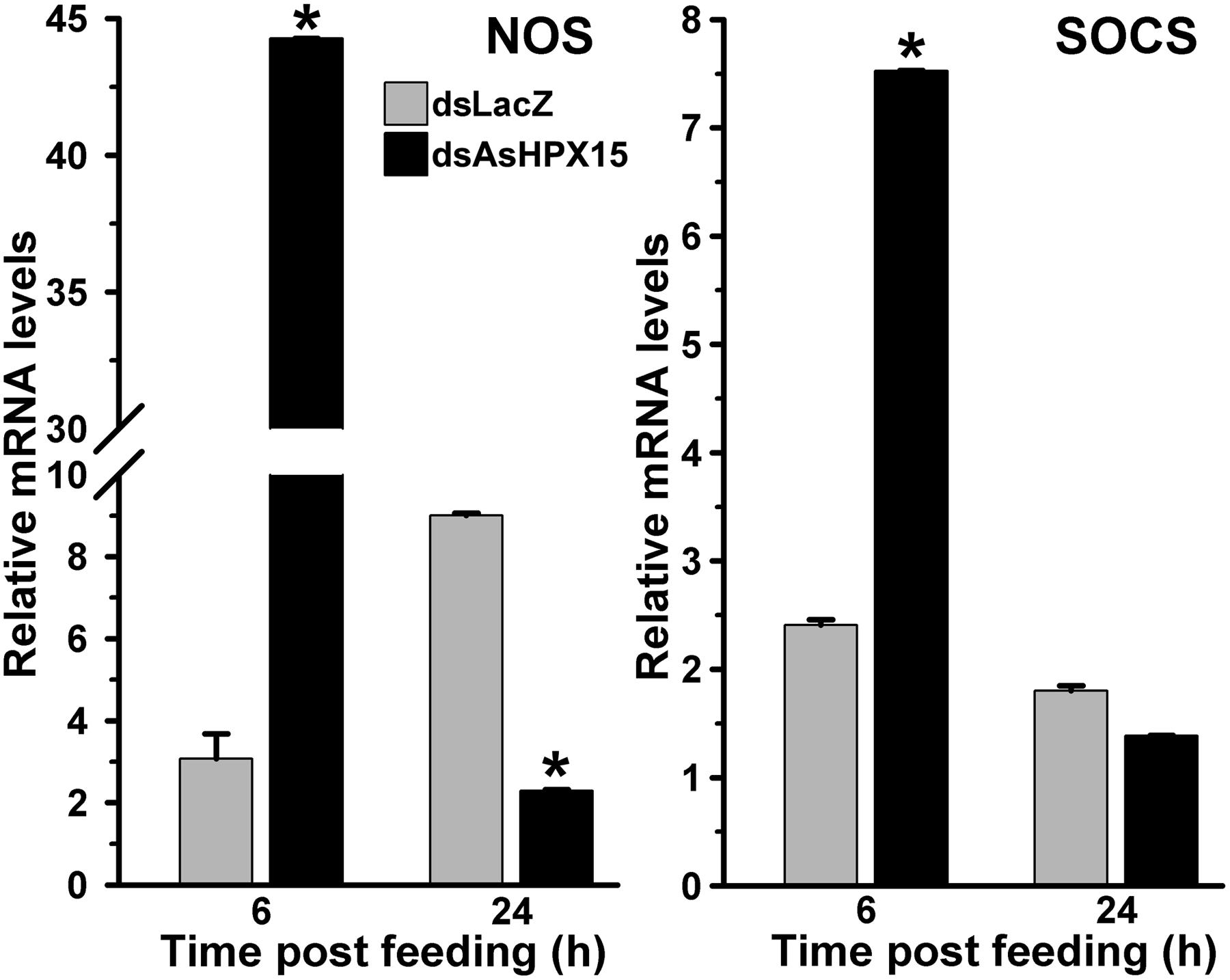
FIGURE 7. Relative levels of NOS and SOCS mRNA in AsHPX15 silenced and bacteria fed midguts. Mosquitoes injected with dsLacZ (controls) or dsAsHPX15 (silenced) RNA were fed on bacteria supplemented blood and relative levels of NOS or SOCS mRNA were analyzed in their midguts at different time intervals after feeding. Relative levels of mRNA are presented against the sugar fed midguts. Significant differences are denoted by an asterisk.
The Antibacterial Peroxidase HPX8 is Suppressed in AsHPX15 Silenced Midguts
As, we found before that HPX8, an antibacterial peroxidase, is induced after the exogenous bacteria feeding (Figure 2). We analyzed the expression of this gene to understand its role in regulating the bacterial load when AsHPX15 mediated physical barrier formation is interrupted. Results presented in Figure 8 revealed that in dsLacZ injected control midguts, HPX8 gene was induced 650-fold after 24 h of exogenous bacteria feeding against sugar fed midguts. However, its expression was reduced ∼40 times in AsHPX15 silenced midguts when compared to the unsilenced controls (Figure 8). Moreover, the expression levels of HPX8 gene in controls and silenced midguts at 6 h post feeding were indifferent (p = 0.078).
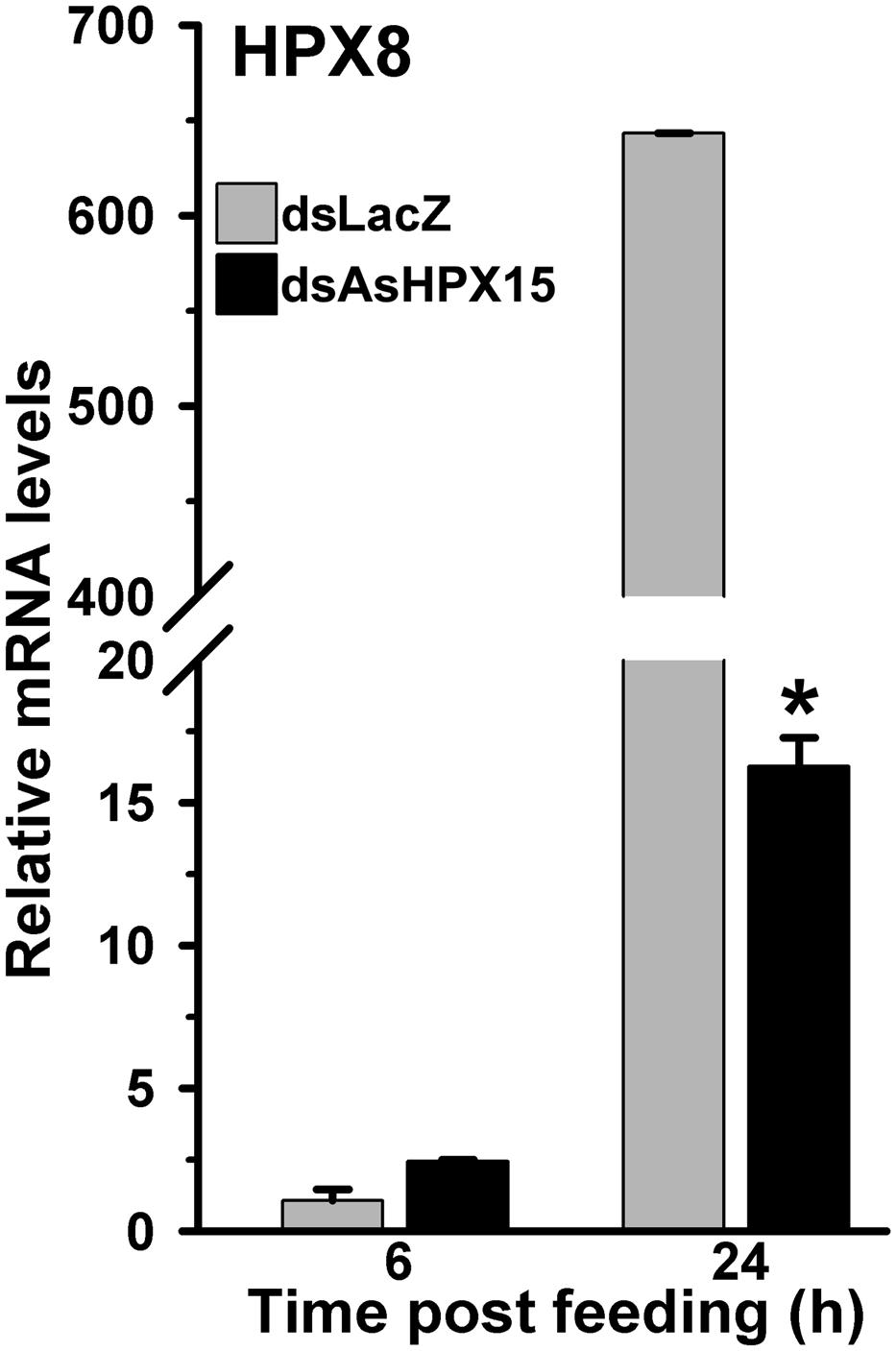
FIGURE 8. Expression of HPX8 gene in AsHPX15 silenced and bacteria fed midguts. Mosquitoes injected with dsLacZ (controls) or dsAsHPX15 (silenced) RNA were fed on bacteria supplemented blood and relative levels of HPX8 mRNA in their midguts were analyzed at different time points after feeding. Relative levels of mRNA are presented against the sugar fed midguts. Significant differences are shown by an asterisk.
These results collectively indicated that the classical antibacterial pathways are least effective in controlling the heavy load of exogenous bacteria in AsHPX15 silenced midguts. However, the induced NOS (an effector gene of the JAK/STAT pathway) and gambicin seem to be the major determinants of the antibacterial immunity in these midguts.
Discussion
The mosquito midgut is an organ for food digestion and immunity. It is an excellent system to understand the interaction of innate immunity, exogenous food and endogenous natural microbial habitats. It is noteworthy to mention that a fine balance between the innate immunity and digestion process is required to drive the physiological functions of the midgut over the immune reactivity against bolus antigens. For that, mosquito midgut is equipped with a number of mechanisms (Kajla et al., 2015a). Among these mechanisms, the formation of a heme peroxidase HPX15 crosslinked mucin barrier is extremely important (Kumar et al., 2010). This mucin barrier does not allow the interaction and recognition of bolus bacteria with the immune reactive midgut epithelium and thus, protects them from the immune attack. As a result of that food digestion or other important physiological processes are driven forward without the induction of midgut immunity (Kajla et al., 2015a). Our data also supported this concept that the endogenous as well as exogenous fed bacteria easily grow in the midgut lumen and AsHPX15 gene is induced after feeding the normal or bacteria supplemented blood (Figures 1 and 2). Interestingly, we observed the reduction of AsHPX15 gene expression in 12 h post bacteria fed midguts (Figure 2). This effect might be due to the SOCS mediated suppression of STAT pathway (Figure 4). The presence of STAT binding site in the regulatory region of AsHPX15 gene supported this assumption (Kajla et al., 2016a). We also believed that the suppression of AsHPX15 gene expression in bacteria fed midguts might be least effective on the mucin barrier formation because the luminal secretion of mucins and localization of HPX15 protein occur by this time, which is well evident in the case of A. gambiae midguts (Kumar et al., 2010; Injera et al., 2013).
Importantly, the number of midgut bacteria does not increase continuously; however, there is variability in their growth pattern (Figure 1). It may be due to the selection and growth of specific types of midgut bacteria at certain time points or all types of bacteria have variable growth patterns at different time of post blood feeding. Moreover, these assumptions warrant further investigations. On the other hand, the midgut immunity might also play an important role in regulating the growth of bacteria at certain moments therefore, some of the classical immune genes such as, HPX8, GNBP, Toll and gambicin are induced at 6 h after bacteria supplemented blood feeding (Figures 2 and 3). However, in case of bacteria-free blood fed control midguts, the induction of these genes is comparatively minimal to facilitate the growth of endogenous bacteria. Thus, the mosquito midgut system might determine the degree and strength of immune activation on the basis of bacterial or antigenic load in blood bolus. These findings may also explain the previous observations by other researchers where feeding the exogenous bacteria with Plasmodium infected blood reduced the parasite development (Pumpuni et al., 1993, 1996; Gonzalez-Ceron et al., 2003; Dong et al., 2009). We believed that the negative regulation of Plasmodium development in the presence of exogenous bacteria might be due to an early induction of midgut immunity as we observed in Figure 7 because some of these immune pathways are also known to participate in antiplasmodial immunity (Cirimotich et al., 2011a; Bahia et al., 2014).
Our previous findings revealed that the anopheline heme peroxidase HPX15 is a unique lineage-specific gene and its role in the modulation of mosquito midgut immunity can be targeted to control Plasmodium development (Kajla et al., 2014, 2015b, 2016a). Thus, we thought to combine our present knowledge of exogenous bacteria induced immunity with AsHPX15 gene silencing to understand the additive effect of these two events on mosquito midgut immunity. We found that AsHPX15 silencing induced innate immunity to suppress the bacterial load in the silenced midguts and capable of regulating the high load of exogenous fed bacteria (Figure 5). These findings are in agreement with the previous study where silencing of AsHPX15 ortholog in A. gambiae suppressed the growth of endogenous bacteria through the induction of bacteria-specific classical immunity (Kumar et al., 2010). These effects are simply due to the disruption of mucin barrier in AsHPX15 silenced midguts and that leads to the recognition of the bolus bacteria by the immune reactive midgut epithelial cells (Kumar et al., 2010).
We observed that the regulation of exogenous fed bacteria in the mosquito midgut is due to the activation of classical Toll and Imd immune pathways when AsHPX15 mediated mucin barrier is present (Figures 2 and 3). The silencing of AsHPX15 gene and thus, reducing the formation of mucin barrier, might alter the number of bacteria in the midgut. In AsHPX15 silenced midguts the classical immune pathways are mostly ineffective (Figure 6) and the bacteria levels are regulated by other stronger immune mechanisms. Thus, NOS is activated at early hours in AsHPX15 silenced midguts to control the heavy load of bacteria (that includes exogenous as well as endogenous bacteria) (Figure 7). In addition, the induction of SOCS gene in parallel to the NOS might again indicate the activation as well as balanced regulation of JAK/STAT pathway in AsHPX15 silenced midguts (Figure 7). Our findings also revealed that the levels of endogenous bacteria are very less in comparison to the exogenous fed bacteria (Figure 1) thus, NOS is induced in AsHPX15 silenced midguts to control the outsized load of these bacteria (Figure 7). In addition, the induction of gambicin, a classical immune pathway gene, in AsHPX15 silenced midguts might indicate its regulation through JAK/STAT pathway as suggested by other researchers (Buchon et al., 2009; Cheng et al., 2016).
Immune regulation of a huge exogenous bacterial load in the AsHPX15 gene silenced midguts revealed that there is a circumstantial activation of antibacterial immunity. In other words, classical immune pathways are most active against increasing load of bacteria when AsHPX15 gene is expressed (Figures 2 and 3), however, STAT pathway predominates as antibacterial in the absence of AsHPX15 gene expression (Figure 7). This might indicate that the bacterial load and its interaction with the midgut epithelium boosts more effective immune molecule to manage the microbial homeostasis. Thus, we proposed a model as shown in Figure 9 that AsHPX15 helps in the formation of mucin barrier in A. stephensi midgut. This mucin barrier blocks the direct interactions of bolus bacteria with the immune reactive gut epithelial cells. However, the increased growth of bacteria or their elicitors activate the classical Toll and Imd pathways that maintain gut bacterial homeostasis. On the other hand, in AsHPX15 silenced midguts the formation of mucin barrier is compromised and bacterial elicitors activate the midgut epithelial immunity. As a result, midgut epithelial cells mount a strong immune response through the activation of STAT pathway to regulate the growth of midgut bacteria and protecting the mosquito against its deleterious effects (Figure 9).
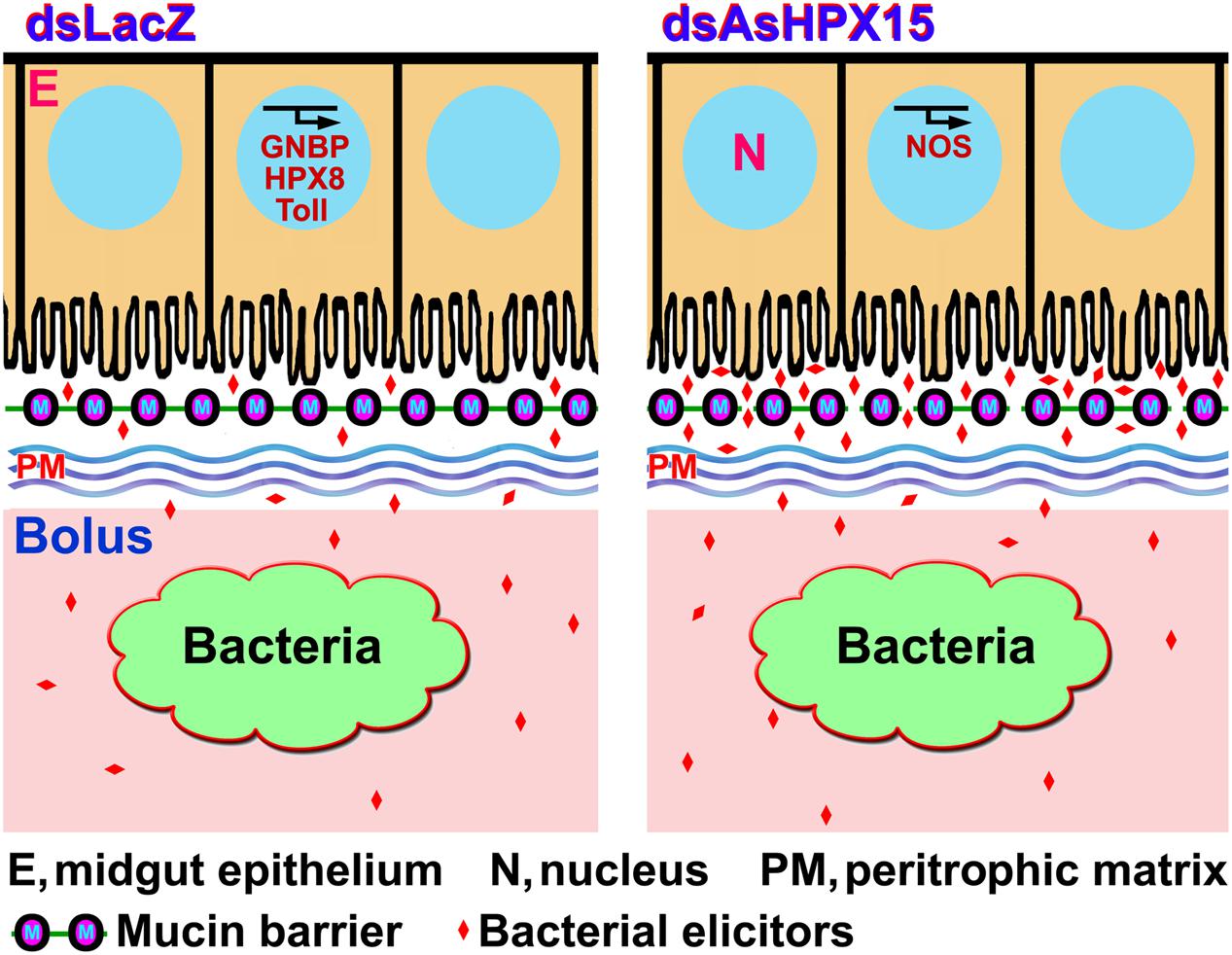
FIGURE 9. Model of A. stephensi midgut immunity in the presence or absence of AsHPX15 gene. AsHPX15 catalyzes the formation of mucin layer that blocks the direct interaction of lumen bacteria or bacterial elicitors with midgut epithelium. The increased load of bacteria induces classical immune pathways such as, GNBP, Toll and an antibacterial heme peroxidase HPX8 to regulate bacterial numbers. AsHPX15 silencing reduces the formation of mucin barrier that allows direct interaction of lumen bacteria or their elicitors with midgut epithelium. In this condition, STAT pathway effector molecule NOS is induced to regulate the numbers of midgut bacteria.
Based on our findings, we emphasized that manipulation of AsHPX15 gene in A. stephensi modulates the immune system and co-infection of Plasmodium with exogenous bacteria will result in the negative regulation of the parasite development through induction of specific immune pathways. Because HPX15 is highly conserved gene exclusively among anophelines (Kajla et al., 2015b, 2016a), then the proposed idea might be applicable for other worldwide distributed malaria vectors as a common strategy.
Author Contributions
MK, LG, and SK designed experiments for this work. MK, TC, PK, KG, and RD collected samples to perform experiments. MK, TC, PK, KG, RD, LG, and SK performed AsHPX15 and bacterial kinetics experiments and analyzed the induction of different immune genes. MK, TC, PK, LG, and SK performed AsHPX15 silencing experiments. MK, LG, and SK analyzed the data and wrote the manuscript with input from all authors. All authors read and approved the manuscript.
Funding
MK and KG are the recipient of Basic Science Research (BSR) fellowship from University Grants Commission (UGC), India. PK, RD, and TC would like to acknowledge the Department of Science and Technology (DST), Government of India for providing the fellowship. This work was supported by the research grants from the Department of Science and Technology (DST), Government of India (grant number SR/SO/HS-0131/2010).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors wish to thank the laboratory staff for maintaining the insectory, Institute administration for providing all other facilities to accomplish the research and central animal facility (CAF) for providing mice that were used in this study.
References
Agaisse, H., and Perrimon, N. (2004). The roles of JAK/STAT signaling in Drosophila immune responses. Immunol. Rev. 198, 72–82. doi: 10.1111/j.0105-2896.2004.0133.x
Azambuja, P., Garcia, E. S., and Ratcliffe, N. A. (2005). Gut microbiota and parasite transmission by insect vectors. Trends Parasitol. 21, 568–572. doi: 10.1016/j.pt.2005.09.011
Bahia, A. C., Dong, Y., Blumberg, B. J., Mlambo, G., Tripathi, A., BenMarzouk-Hidalgo, O. J., et al. (2014). Exploring Anopheles gut bacteria for Plasmodium blocking activity. Environ. Microbiol. 16, 2980–2994. doi: 10.1111/1462-2920.12381
Bahia, A. C., Kubota, M. S., Tempone, A. J., Araujo, H. R., Guedes, B. A., Orfano, A. S., et al. (2011). The JAK-STAT pathway controls Plasmodium vivax load in early stages of Anopheles aquasalis infection. PLoS Negl. Trop. Dis. 5:e1317. doi: 10.1371/journal.pntd.0001317
Beier, M. S., Pumpuni, C. B., Beier, J. C., and Davis, J. R. (1994). Effects of para-aminobenzoic acid, insulin, and gentamycin on Plasmodium falciparum development in anopheline mosquitoes (Diptera: Culicidae). J. Med. Entomol. 31, 561–565. doi: 10.1093/jmedent/31.4.561
Boissière, A., Tchioffo, M. T., Bachar, D., Abate, L., Marie, A., Nsango, S. E., et al. (2012). Midgut microbiota of the malaria mosquito vector Anopheles gambiae and interactions with Plasmodium falciparum infection. PLoS Patho. 8:e1002742. doi: 10.1371/journal.ppat.1002742
Buchon, N., Broderick, N. A., Poidevin, M., Pradervand, S., and Lemaitre, B. (2009). Drosophila intestinal response to bacterial infection: activation of host defense and stem cell proliferation. Cell Host Microbe 5, 200–211. doi: 10.1016/j.chom.2009.01.003
Chavshin, A. R., Oshaghi, M. A., Vatandoost, H., Pourmand, M. R., Raeisi, A., Enayati, A. A., et al. (2012). Identification of bacterial microflora in the midgut of the larvae and adult of wild caught Anopheles stephensi: a step toward finding suitable paratransgenesis candidates. Acta Trop. 121, 129–134. doi: 10.1016/j.actatropica.2011.10.015
Cheng, G., Liu, Y., Wang, P., and Xiao, X. (2016). Mosquito defense strategies against viral infection. Trends Parasitol. 32, 177–186. doi: 10.1016/j.pt.2015.09.009
Christophides, G. K., Zdobnov, E., Barillas-Mury, C., Birney, E., Blandin, S., Blass, C., et al. (2002). Immunity-related genes and gene families in Anopheles gambiae. Science 298, 159–165. doi: 10.1126/science.1077136
Cirimotich, C. M., Dong, Y., Clayton, A. M., Sandiford, S. L., Souza-Neto, J. A., Mulenga, M., et al. (2011a). Natural microbe-mediated refractoriness to Plasmodium infection in Anopheles gambiae. Science 332, 855–858. doi: 10.1126/science.1201618
Cirimotich, C. M., Ramirez, J. L., and Dimopoulos, G. (2011b). Native microbiota shape insect vector competence for human pathogens. Cell Host Microbe 10, 307–310. doi: 10.1016/j.chom.2011.09.006
Demirel, I., Säve, S., Kruse, R., and Persson, K. (2013). Expression of suppressor of cytokine signalling 3 (SOCS3) in human bladder epithelial cells infected with uropathogenic Escherichia coli. APMIS 121, 158–167. doi: 10.1111/j.1600-0463.2012.02951.x
Dhawan, R., Gupta, K., Kajla, M., Kumar, S., Gakhar, S. K., Kakani, P., et al. (2015). Molecular characterization of SOCS gene and its expression analysis on Plasmodium berghei infection in Anopheles culicifacies. Acta Trop. 152, 170–175. doi: 10.1371/journal.pntd.0001317
Dimopoulos, G., Seeley, D., Wolf, A., and Kafatos, F. C. (1998). Malaria infection of the mosquito Anopheles gambiae activates immune-responsive genes during critical transition stages of the parasite life cycle. EMBO J. 17, 6115–6123. doi: 10.1093/emboj/17.21.6115
Dong, Y., Aguilar, R., Xi, Z., Warr, E., Mongin, E., and Dimopoulos, G. (2006). Anopheles gambiae immune responses to human and rodent Plasmodium parasite species. PLoS Patho. 2:e52. doi: 10.1371/journal.ppat.0020052
Dong, Y., Manfredini, F., and Dimopoulos, G. (2009). Implication of the mosquito midgut microbiota in the defense against malaria parasites. PLoS Patho. 5:e1000423. doi: 10.1371/journal.ppat.1000423
Ghosh, A., Edwards, M. J., and Jacobs-Lorena, M. (2000). The journey of the malaria parasite in the mosquito: hopes for the new century. Parasitol. Today 16, 196–201. doi: 10.1016/S0169-4758(99)01626-9
Gonzalez-Ceron, L., Santillan, F., Rodriguez, M. H., Mendez, D., and Hernandez-Avila, J. E. (2003). Bacteria in midguts of field-collected Anopheles albimanus block Plasmodium vivax sporogonic development. J. Med. Entomol. 40, 371–374. doi: 10.1603/0022-2585-40.3.371
Gupta, L., Molina-Cruz, A., Kumar, S., Rodrigues, J., Dixit, R., Zamora, R. E., et al. (2009). The STAT pathway mediates late-phase immunity against Plasmodium in the mosquito Anopheles gambiae. Cell Host Microbe. 5, 498–507. doi: 10.1016/j.chom.2009.04.003
Hillyer, J. F., and Estévez-Lao, T. Y. (2010). Nitric oxide is an essential component of the hemocyte-mediated mosquito immune response against bacteria. Dev. Comp. Immunol. 34, 141–149. doi: 10.1016/j.dci.2009.08.014
Injera, W. E., Kabiru, E. W., Gicheru, M. M., Githure, J. I., and Beier, J. C. (2013). Immunopathological features developing in the mosquito midgut after feeding on Anopheles gambiae Mucin-1/Interleukin-12 cDNA immunized mice. Int. J. Morphol. 31, 329–337. doi: 10.4067/S0717-95022013000100051
Kajla, M., Bhattacharya, K., Gupta, K., Banerjee, U., Kakani, P., Gupta, L., et al. (2016b). Identification of temperature induced larvicidal efficacy of Agave angustifolia against Aedes, Culex and Anopheles larvae. Front. Public Health 3:286. doi: 10.3389/fpubh.2015.00286
Kajla, M., Gupta, K., Gupta, L., and Kumar, S. (2015a). A fine-tuned management between physiology and immunity maintains the gut microbiota in insects. Biochem. Physiol. 4, 182. doi: 10.4172/2168-9652.1000182
Kajla, M., Gupta, K., Kakani, P., Dhawan, R., Choudhury, T. P., Gupta, L., et al. (2015b). Identification of an Anopheles lineage-specific unique heme peroxidase HPX15: a plausible candidate for arresting malaria parasite development. J. Phylogen. Evolution. Biol. 3:160. doi: 10.4172/2329-9002.1000160
Kajla, M., Kakani, P., Choudhury, T. P., Gupta, K., Dhawan, R., Gakhar, S. K., et al. (2014). Characterization of anopheline unique peroxidase and its role in the regulation of Plasmodium development. Malar. J. 13:49. doi: 10.1186/1475-2875-13-S1-P49
Kajla, M., Kakani, P., Gupta, K., Choudhury, T. P., Gupta, L., and Kumar, S. (2016a). Characterization and expression analysis of gene encoding heme peroxidase HPX15 in major Indian malaria vector Anopheles stephensi (Diptera: Culicidae). Acta Trop. 158, 107–116. doi: 10.1016/j.actatropica.2016.02.028
Kumar, S., Gupta, L., Han, Y. S., and Barillas-Mury, C. (2004). Inducible peroxidases mediate nitration of Anopheles midgut cells undergoing apoptosis in response to Plasmodium invasion. J. Biol. Chem. 279, 53475–53482. doi: 10.1074/jbc.M409905200
Kumar, S., Molina-Cruz, A., Gupta, L., Rodrigues, J., and Barillas-Mury, C. (2010). A peroxidase/dual oxidase system modulates midgut epithelial immunity in Anopheles gambiae. Science 327, 1644–1648. doi: 10.1126/science.1184008
Lemaitre, B., Kromer-Metzger, E., Michaut, L., Nicolas, E., Meister, M., Georgel, P., et al. (1995). A recessive mutation, immune deficiency (Imd), defines two distinct control pathways in the Drosophila host defense. Proc. Natl. Acad. Sci. U.S.A. 92, 9465–9469. doi: 10.1073/pnas.92.21.9465
Lensen, A., Mulder, L., Tchuinkam, T., Willemsen, L., Eling, W., and Sauerwein, R. (1998). Mechanisms that reduce transmission of Plasmodium falciparum malaria in semi immune and non-immune persons. J. Infect. Dis. 177, 1358–1363. doi: 10.1086/515263
Livak, K. J., and Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C (T)) Method. Methods 25, 402–408. doi: 10.1006/meth.2001.1262
Luckhart, S., Vodovotz, Y., Cui, L., and Rosenberg, R. (1998). The mosquito Anopheles stephensi limits malaria parasite development with inducible synthesis of nitric oxide. Proc. Natl. Acad. Sci U.S.A. 12, 5700–5705. doi: 10.1073/pnas.95.10.5700
Maehr, T., Vecino, J. L., Wadsworth, S., Wang, T., and Secombes, C. J. (2014). Four CISH paralogues are present in rainbow trout Oncorhynchus mykiss: differential expression and modulation during immune responses and development. Mol. Immunol. 62, 186–198. doi: 10.1016/j.molimm.2014.06.021
Meister, S., Agianian, B., Turlure, F., Relogio, A., Morlais, I., Kafatos, F. C., et al. (2009). Anopheles gambiae PGRPLC-mediated defense against bacteria modulates infections with malaria parasites. PLoS Patho. 5:e1000542. doi: 10.1371/journal.ppat.1000542
Michel, K., and Kafatos, F. C. (2005). Mosquito immunity against Plasmodium. Insect Biochem. Mol. Biol. 35, 677–689. doi: 10.1016/j.ibmb.2005.02.009
Michel, T., Reichhart, J. M., Hoffmann, J. A., and Royet, J. (2001). Drosophila Toll is activated by Gram-positive bacteria through a circulating peptidoglycan recognition protein. Nature 414, 756–759. doi: 10.1038/414756a
Minard, G., Mavingui, P., and Moro, C. V. (2013). Diversity and function of bacterial microbiota in the mosquito holobiont. Parasit. Vectors 6, 146. doi: 10.1186/1756-3305-6-146
Moreno-García, M., Recio-Tótoro, B., Claudio-Piedras, F., and Lanz-Mendoza, H. (2014). Injury and immune response: applying the danger theory to mosquitoes. Front. Plant Sci. 5:451. doi: 10.3389/fpls.2014.00451
Osta, M. A., Christophides, G. K., Vlachou, D., and Kafatos, F. C. (2004). Innate immunity in the malaria vector Anopheles gambiae: comparative and functional genomics. J. Exp. Biol. 207, 2551–2563. doi: 10.1242/jeb.01066
Pradel, G. (2007). Proteins of the malaria parasite sexual stages: expression, function and potential for transmission blocking strategies. Parasitology 134, 1911–1929. doi: 10.1017/S0031182007003381
Pumpuni, C. B., Beier, M. S., Nataro, J. P., Guers, L. D., and Davis, J. R. (1993). Plasmodium falciparum: inhibition of sporogonic development in Anopheles stephensi by Gram-negative bacteria. Exp. Parasitol. 77, 195–199. doi: 10.1006/expr.1993.1076
Pumpuni, C. B., DeMaio, J., Kent, M., Davis, J. R., and Beier, J. C. (1996). Bacterial population dynamics in three anopheline species: the impact on Plasmodium sporogonic development. Am. J. Trop. Med. Hyg. 54, 214–218.
Ramiro, R. S., Alpedrinha, J., Carter, L., Gardner, A., and Reece, S. E. (2011). Sex and death: the effects of innate immune factors on the sexual reproduction of malaria parasites. PLoS Pathog. 7:e1001309. doi: 10.1371/journal.ppat.1001309
Rani, A., Sharma, A., Rajagopal, R., Adak, T., and Bhatnagar, R. K. (2009). Bacterial diversity analysis of larvae and adult midgut microflora using culture-dependent and culture-independent methods in lab-reared and field-collected Anopheles stephensi-an Asian malarial vector. BMC Microbiol. 9:96. doi: 10.1186/1471-2180-9-96
Simon, N., Lasonder, E., Scheuermayer, M., Kuehn, A., Tews, S., Fischer, R., et al. (2013). Malaria parasites co-opt human factor H to prevent complement-mediated lysis in the mosquito midgut. Cell Host Microbe 13, 29–41. doi: 10.1016/j.chom.2012.11.013
Sinden, R. E., and Billingsley, P. F. (2001). Plasmodium invasion of mosquito cells: hawk or dove? Trends Parasitol. 17, 209–212. doi: 10.1016/S1471-4922(01)01928-6
Touré, A. M., Mackey, A. J., Wang, Z. X., and Beier, J. C. (2000). Bactericidal effects of sugar-fed antibiotics on resident midgut bacteria of newly emerged anopheline mosquitoes (Diptera: Culicidae). J. Med. Entomol. 37, 246–249. doi: 10.1603/0022-2585-37.2.246
Vijay, S., Rawat, M., Adak, T., Dixit, R., Nanda, N., Srivastava, H., et al. (2011). Parasite killing in malaria non-vector mosquito Anopheles culicifacies species B: implication of nitric oxide synthase upregulation. PLoS ONE 6:e18400. doi: 10.1371/journal.pone.0018400
Vizioli, J., Bulet, P., Hoffmann, J. A., Kafatos, F. C., Müller, H. M., and Dimopoulos, G. (2001). Gambicin: a novel immune responsive antimicrobial peptide from the malaria vector Anopheles gambiae. Proc. Natl. Acad. Sci. U.S.A. 98, 12630–12635. doi: 10.1073/pnas.221466798
Vlachou, D., Schlegelmilch, T., Christophides, G. K., and Kafatos, F. C. (2005). Functional genomic analysis of midgut epithelial responses in Anopheles during Plasmodium invasion. Curr. Biol. 15, 1185–1195. doi: 10.1016/j.cub.2005.06.044
Wang, Y., Gilbreath, T. M., Kukutla, P., Yan, G., and Xu, J. (2011). Dynamic gut microbiome across life history of the malaria mosquito Anopheles gambiae in Kenya. PLoS ONE 6:e24767. doi: 10.1371/journal.pone.0024767
Warr, E., Das, S., Dong, Y., and Dimopoulos, G. (2008). The Gram-negative bacteria-binding protein gene family: its role in the innate immune system of Anopheles gambiae and in anti-Plasmodium defence. Insect Mol. Biol. 17, 39–51. doi: 10.1111/j.1365-2583.2008.00778.x
Waterhouse, R. M., Kriventseva, E. V., Meister, S., Xi, Z., Alvarez, K. S., Bartholomay, L. C., et al. (2007). Evolutionary dynamics of immune-related genes and pathways in disease-vector mosquitoes. Science 316, 1738–1743. doi: 10.1126/science.1139862
WHO (2015). WHO Global Malaria Programme. World Malaria Report: 2015. Geneva: World Health Organization.
Wormald, S., and Hilton, D. J. (2004). Inhibitors of cytokine signal transduction. J. Biol. Chem. 279, 821–824. doi: 10.1074/jbc.R300030200
Keywords: Anopheles stephensi, peroxidases, HPX15, mucin barrier, midgut bacteria, Plasmodium, innate immunity, vectorial capacity
Citation: Kajla M, Choudhury TP, Kakani P, Gupta K, Dhawan R, Gupta L and Kumar S (2016) Silencing of Anopheles stephensi Heme Peroxidase HPX15 Activates Diverse Immune Pathways to Regulate the Growth of Midgut Bacteria. Front. Microbiol. 7:1351. doi: 10.3389/fmicb.2016.01351
Received: 04 July 2016; Accepted: 16 August 2016;
Published: 31 August 2016.
Edited by:
Alexandre Morrot, Federal University of Rio de Janeiro, BrazilReviewed by:
Rajnikant Dixit, National Institute of Malaria Research, IndiaYeon Soo Han, Chonnam National University, South Korea
Copyright © 2016 Kajla, Choudhury, Kakani, Gupta, Dhawan, Gupta and Kumar. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sanjeev Kumar, c2FuamVldkBwaWxhbmkuYml0cy1waWxhbmkuYWMuaW4= c2FuamVldmxhbGl0YUBob3RtYWlsLmNvbQ==
 Mithilesh Kajla
Mithilesh Kajla Tania P. Choudhury1
Tania P. Choudhury1 Parik Kakani
Parik Kakani Kuldeep Gupta
Kuldeep Gupta Rini Dhawan
Rini Dhawan Lalita Gupta
Lalita Gupta Sanjeev Kumar
Sanjeev Kumar