- 1Department of Microbiology, Immunology and Biochemistry, University of Tennessee Health Science Center, Memphis, TN, USA
- 2Department of Pharmaceutical Sciences, University of Tennessee Health Science Center, Memphis, TN, USA
- 3Chemical Biology and Therapeutics Department, St. Jude Children's Research Hospital, Memphis, TN, USA
The conventional method for quantitating Mycobacterium tuberculosis (Mtb) in vitro and in vivo relies on bacterial colony forming unit (CFU) enumeration on agar plates. Due to the slow growth rate of Mtb, it takes 3–6 weeks to observe visible colonies on agar plates. Imaging technologies that are capable of quickly quantitating both active and dormant tubercle bacilli in vitro and in vivo would accelerate research toward the development of anti-TB chemotherapies and vaccines. We have developed a fluorescent probe that can directly label the Mtb cell wall components. The fluorescent probe, designated as DLF-1, has a strong affinity to the D-Ala-D-Ala unit of the late peptidoglycan intermediates in the bacterial cell wall. We demonstrate that DLF-1 is capable of detecting Mtb in both the actively replicating and dormant states in vitro at 100 nM without inhibiting bacterial growth. The DLF-1 fluorescence signal correlated well with CFU of the labeled bacteria (R2 = 1 and 0.99 for actively replicating and dormant Mtb, respectively). DLF-1 can also quantitate labeled Mtb inside of cells. The utility of DLF-1 probe to quantitate Mtb was successfully applied to identify genes critical for cell invasion. In conclusion, this novel near infrared imaging probe provides a powerful new tool for enumerating Mtb with potential future use in bacterial virulence study.
Introduction
With more than three deaths per minute, tuberculosis (TB) is a major public health threat. Approximately 9 million new cases of TB are identified per year, with 1.5 million deaths related to TB (WHO, 2015), making TB the greatest cause of mortality due to a bacterial pathogen. The majority of individuals infected with Mtb develop latent TB infection (LTBI) where bacteria and the host have established equilibrium without causing apparent symptoms in the host. In LTBI, Mtb can persist in host lungs or other tissues for months to decades without replication. Approximately one third of the world population has been latently infected by Mtb (Sudre et al., 1992). Because LTBI can be reactivated upon immune suppression, the infected individuals serve as a huge reservoir of active TB in the community. Eliminating latent TB cases would help to reduce active TB significantly.
Research on TB has been impeded by the slow growth rate of Mycobacterium tuberculosis (Mtb), the major causative pathogen of TB. Conventionally, quantification of Mtb in vitro and in vivo relies on counting bacterial colony forming units (CFU) on agar plates. Mtb divides every 15–20 h. It takes several weeks for Mtb to form visible colonies on solid agar plates (Glickman and Jacobs, 2001; Smith, 2003; Zumla et al., 2013). Imaging technologies that are capable of quickly quantitating tubercle bacilli in vitro and in vivo would greatly accelerate research toward the development of anti-TB chemotherapies and vaccines.
Both bioluminescent and fluorescent protein labeled Mycobacterium spp. have been developed for quantitating bacteria in TB research (Andreu et al., 2010, 2012, 2013; Carroll et al., 2010; Kong et al., 2010; Zelmer et al., 2012; Zhang et al., 2012; Ollinger et al., 2013; Singh et al., 2014; Vocat et al., 2015), a number of which have been applied to in vivo imaging of M. tuberculosis infection (Andreu et al., 2010, 2013; Kong et al., 2010; Zelmer et al., 2012; Zhang et al., 2012). By using non-invasive fluorescent-imaging technologies as laboratory tools to track Mtb in vitro and in animals, infections can be visualized in real-time. Although recombinant reporter strains have been used for imaging Mtb infection (Andrew and Roberts, 1993; Cooksey et al., 1993; Arain et al., 1996; Hickey et al., 1996; Heuts et al., 2009) in vitro and in vivo, expression of a foreign gene can impact bacterial fitness (Coulson et al., 1994; Wendland and Bumann, 2002; Rang et al., 2003) or sometimes even be toxic to bacteria (Andreu et al., 2010).
In this study, we developed a fluorescent-imaging strategy that labels macromolecular components of the TB cell wall. This long wavelength fluorescent-imaging probe designated as direct labeling fluorescence-1 (DLF-1), is shown to be highly quantitative. DLF-1 is capable of detecting Mtb in both actively replicating and non-replicating dormant states in vitro. DLF-1 can also quantitate labeled Mtb harbored in cells. In addition, we have successfully verified that DLF-1 can be applied to identify bacterial genes critical for cell entry. This novel imaging probe provides a powerful new tool for quantitating actively replicating and dormant Mtb in vitro.
Materials and Methods
Synthesis of DLF-1
Et3N (15.2 mL, 0.109 mmol), oxyma (4.7 mg, 0.0328 mmol), and EDCI (6.3 mg, 0.0328 mmol) were added to a stirred solution of vancomycin (32.5 mg, 0.0219 mmol) and Boc-Gly-OH (4.6 mg, 0.0263 mmol) in DMF (0.2 mL). After being stirred for 22 h at room temperature, the reaction mixture was filtered. The filtrate was purified by reverse phase HPLC to afford Boc-Glycyl-vancomycin (85% yield). 0.1 mL of 4.0 M HCl (dioxane solution) was then added to a stirred solution of Boc-Glycyl-vancomycin (3.9 mg, 2.39 mmol) in dioxane (0.1 mL). After 4 h at room temperature, all volatiles were removed under reduced pressure to afford HCl-Glycyl-vancomycin. Et3N (1.0 mL, 7.32 mmol) and cyanin5.5 NHS ester (0.52 mg, 0.732 mmol) in DMF (0.1 mL) were added to a stirred solution of HCl-Glycyl-vancomycin in DMF (0.1 mL). After 6 h at room temperature, all volatiles were removed under reduced pressure to afford the crude DLF-1. This was purified by reverse phase HPLC to afford DLF-1 (>95% purity).
Verification of DLF-1 Binding to PG Using Isothermal Titration Calorimetry (ITC)
Binding of bacterial PG peptide to vancomycin or DLF-1 was monitored using an ITC200 (Microcal, Piscataway, NJ). Vancomycin hydrochloride from Streptomyces orientalis (32 μM final concentration, V2002, Sigma) or DLF-1 (7 μM final) was diluted in 1X PBS pH 7.4 (21-040-CM, Corning), and 204 μL was loaded into the cell. Experiments contained 1% DMSO (BP-231, Fisher) in both syringe and cell, to aid solubility. Peptide Ac-L-Lys-D-Ala-D-Ala was dissolved in the same buffer (600 or 100 μM, respectively), and 39.4 μL was loaded in the syringe. For each binding, three separate experiments were conducted at 25°C (298.15K). Injections were performed with serial injections of peptide; first, one injection of 1 μL, followed by 19 incremental injections of 2 μL, at 120 s intervals. Data from the first injection was excluded, due to pre-equilibration mixing between the contents of cell and syringe at the syringe tip. Peak areas were integrated, normalized, and then fitted by non-linear regression using the independent sites model in Origin™ (version 2.3.6, Microcal, Piscataway, NJ).
Bacterial Growth Curves in the Presence or Absence of DLF-1
The mycobacterium strains (M. smegmatis, M. bovis BCG, and Mtb) were started from −80°C stocks. For M. bovis BCG, and Mtb, cultures growth in the presence or absence of DLF-1 were performed in 7H9 broth (Difco, Detroit, MI) supplemented with 0.5% glycerol, 10% oleic acid dextrose complex without catalase and 0.05% Tween 80 (MOAD/T medium) at 37°C without shaking. The bacterial cultures were then started in the presence of different concentrations of DLF-1 (0, 100 nM, 1 μM) and monitored OD over time in a 96-well plate with a Tecan multimode reader (Infinite 200 Pro, Tecan) at absorbance 600 nm with bandwidth 9 nm and number of flashes as 25. OD was measured daily for M. bovis BCG and Mtb to plot growth curves. For M. smegmatis, culture was set up in a 96-well plate, and the plate was kept in the reader at 37°C. OD was measured hourly with automatic shaking for 10 min before OD measurement. The Supplementary Figure 1 shows the correlation between the OD values measured with the Tecan microplate reader at 600 nm and the OD value in a cuvette measured with commonly used ThermoScientific Genesys 20 spectrophotometer (ThermoFisher Scientific) at 600 nm, which helps convert the OD values of the Tecan microplate reader into OD values in a cuvette measured with the Genesys 20.
MIC of DLF-1 against M. smegmatis
Minimum inhibitory concentration of DLF-1 against M. smegmatis at which 90% of strains were inhibited (MIC90) was determined using the standard broth microdilution method in 96-well microtiter plates as described previously with minor modifications (Swenson et al., 1982). Briefly, serial doubling dilutions of DLF-1 were prepared in MOAD broth to achieve final concentrations ranging from 8 to 0.125 μg/ml. M. smegmatis was added to each well with a final inoculum of ~105 CFU/ml. The plate was incubated at 37°C. OD600 was measured at 48 h with a Tecan microplate reader. The wells with M. smegmatis alone were controls.
Direct Labeling Bacteria with DLF-1
Bacterial cultures were stopped when they reached the logarithmic growth phase (OD600, 0.4~0.6). For each strain of studied bacteria, a series of dilutions were made starting from OD600 of 1.0 measured with Genesys 20 in growth medium. Bacteria cells were centrifuged at 9000 rpm for 5 min, and cell pellets were washed with 1X PBS twice and finally resuspended in 1X PBS containing 100 nM DLF-1 for 3 h on ice (for fast-growing M. smegmatis) or at room temperature (for M. bovis BCG and Mtb CDC1551). After incubation with DLF-1, bacterial cells were centrifuged (9000 rpm, 5 min), washed once with 1X PBS (10,000 rpm, 10 min) to remove unbound DLF-1, and were finally resuspended in equal volumes of 1X PBS solutions. Each sample was added to a fluorescence 96-well microplate, and fluorescence intensity of DLF-1 was measured with a multimode fluorescence plate reader (InfiniteR 200 PRO, Tecan) at excitation 680 nm and emission 710 nm. Each strain of bacteria was also titrated to enumerate CFU. For determination of threshold of detection, we optimized the condition of the Tecan reader, with gain value as 150, excitation 670 nm and emission 700 nm. For cell infection, a series of concentrations of a tdTomato-expressing Mtb strain were incubated with 100 nM DLF-1, then were used to infect THP-1 macrophages and A549 human lung epithelial cells. Fluorescence was measured 3-h post infection by the Tecan plate reader.
Fluorescent Microscopy of DLF-1 Labeled Mtb Inside Cells
A tdTomato expressing Mtb strain (Erdman-pJDC60) was applied to infect cells after labeling bacteria with DLF-1. This strain was cultured in MOAD/T medium with addition of 25 ug/ml kanamycin until it reaches the logarithmic phase (OD600, 0.4~0.6). A549, or THP-1 cells were grown on the coverslips in 24-well cell culture plates at a concentration of 2 × 105 cells/well. For THP-1, phorbol 12-myristate 13-acetate (PMA) was used to stimulate the cells for 3 days. The cells were then infected with bacteria at 37°C for 3 h. After bacterial infection, extracellular bacteria were aspirated, and the infected cells were washed with 1X PBS, treated with amikacin (200 μg/ml) for additional 2 h to eliminate extracellular bacteria as described previously (Mehta et al., 1996; Napier et al., 2011). Bacterium infected cells were then fixed with 4% freshly made paraformaldehyde for 10 min at room temperature. After washing with 1X PBS for three times, the fixed infected cells were permeabilized with 0.2% Tween-80/PBS (v/v) solution for 5 min, washed again with 1X PBS for three times, and then stained with DAPI solution (300 nM) for 5 min in the dark. After another three-time washing with 1X PBS, the coverslips containing bacterium infected cells were taken out and mounted to glass slides for fluorescence microscopy analysis with a deconvolution microscope (Carl Zeiss). For imaging of DLF-1 labeled dormant Mtb inside macrophages, a Mtb CDC1551 strain carrying a genome integrated tdTomato gene (CDC1551-pYK13) was cultured in a modified Wayne non-replicating persistence model. THP-1 cells seeded in a 24-well plate containing cover slips were stimulated with PMA for 3 days, and then infected with the DLF-1 (100 nM) labeled, non-replicating td-Tomato-expressing Mtb. After bacterial infection, the same protocols of DAPI staining, slide mounting, and microscopy imaging were conducted. Images were taken with a 63X oil lens.
Construction of a Mtb Strain Carrying a Genome-Integrated tdTomato Gene
We constructed a tdTomato-expressing Mtb in a more stable manner by site-specifically integrating a single copy tdTomato into the mycobacterial chromosome, using a mycobacterial plasmid containing the phage attachment site attP and the L5 integrase (int) gene from the mycobacteriophage L5 genome, as has been shown by others for bioluminescent reporters (Andreu et al., 2010, 2013). To construct this vector, we began with replacing the GFP gene in pFJS8, an E. coli-mycobacterium shuttle plasmid containing the L5 promoter (Miltner et al., 2005), with a fragment from pDONR222 (Invitrogen) containing a suicide ccdB gene and a chloramphenicol resistance gene (CmR) at restriction sites of HindIII/KpnI. In this way, the expressions of ccdB and CmR are under a L5 promoter. We then cut the L5-ccdB- CmR fragment from the new vector at XhoI and KpnI and ligated it into XhoI/KpnI digested pYUB412, a mycobacterial integrating cosmid vector (Bange et al., 1999). The tdTomato gene was then PCR amplified from pRSETB-tdTomato using an up-stream primer containing HindIII site at 5′ end and a down-stream primer containing KpnI site at 5′ end (F: 5′-TATAAAGCTTGGATCCATGGTGAGCAAGG-3′; R: 5′-TATAGGTACCGAATTCTTACTTGTACAGCTCGTCC-3′). The purified PCR product was then digested with HindIII and KpnI and ligated into the HindIII/KpnI digested pYUB412 containing L5-ccdB- CmR. The new plasmid is designated as pYK13. After transformation of the pYK13 into CDC1551, we selected colonies with hygromycin selective agar plates, and confirmed the success of integration of tdTomato into Mtb genome by PCR amplifying the tdTomato and hygromycin resistance genes, and by the presence of tdTomato specific fluorescence.
Non-replicating Persistence Model
We modified the Wayne non-replicating persistence (hypoxic shift-down) model by substituting Dubos with 7H9 (containing 0.05% Tween 80 and 10% OAD) and using the Mtb strain expressing genome-integrated tdTomato (CDC1551-pYK13). Mtb cultures were set up at 105–106 cfu/ml in 12 × 75 mm tubes in a total volume of 3.0 mL (head space ratio, HSR = 0.5). Each tube contained a 13 × 4 mm diameter stir bar (Fisher, PA, USA). Cultures were incubated in a 37°C incubator on a stir plate with 200 rpm. Some cultures also contained Methylene Blue at a concentration of 1.5 mg/L to be used as an indicator of oxygen depletion. An additional tube of Methylene Blue without Mtb was used as a color reference. When the Methylene Blue-containing tube turned colorless, cultures were incubated for an additional week (total 18–22 days), and then were used for DLF-1 fluorescence labeling. A sample of the culture was carefully removed from the middle portion of each tube by pipette to avoid disturbing the pellicle on the surface or the sediment on the bottom. Optical density reading (600 nm) of the cultures was determined. A series of dilutions were made and then centrifuged (9000 rpm, 5 min), washed once with 1X PBS/T, and then resuspended in 1X PBS. DLF-1 labeling of the dormant cultures were performed similarly as for labeling of other bacteria before. The Mtb CDC1551 strain carrying a genome-integrated tdTomato gene was cultured in the Wayne model to reach dormant state, diluted in various concentrations, and then labeled with DLF-1. Fluorescent intensity of DLF-1 labeled dormant Mtb was plotted against the concentration of bacteria or concentration of DLF-1, respectively. All conditions were set up in triplicates. Samples of dilutions were taken for CFU enumeration on 7H10 agar plates in duplicates. Colonies were enumerated at 4–6 weeks following inoculation.
Using DLF-1 Labeling to Identify Genes Important for Cell Invasion
Mtb transposon mutant of plcC, CDC1551, ΔhbhA and MT103 strains were cultured in MOAD/T medium. Bacterial cells grown to middle-logarithmic phase (OD600nm, 0.4~0.6) were harvested, labeled with 100 nM DLF-1 at room temperature for 3 h, washed and suspended in antibiotic free A549 cell culture medium. A549 cells were cultured and seeded to 96-well fluorescent microplates (5 × 104/well) 1 day before experiment. DLF-1 labeled Mtb mutants and wild type control strains were used to infect A549 cells at a MOI of 50. Fluorescence of the microplates was measured to quantitate bacteria loaded initially. After 3 h of infection, infected cells were washed once with 1X PBS, then added cell culture medium containing 200 μg/ml amikacin and incubated for another 2 h to remove extracellular bacteria. After aspirating amikacin containing medium and washed with PBS solution, fluorescence of the microplates were measured to quantitate intracellular bacteria. Bacterial infection rate of each strain was calculated as the average fluorescence intensity at the end of infection divided by the average fluorescence at the beginning. Bacterial infection ratio of mutant to wild type control is calculated as the mutant infection rate divided by that of wild type strain. Bacterial samples from the initial and end of infection were taken and plated for CFU analysis.
Results
DLF-1 is a High Affinity Stoichiometric Probe for the D-Ala-D-Ala Motif of Bacterial Peptidoglycan (PG)
The new fluorescent probe was designed to bind to the terminal peptide D-Ala-D-Ala of the PG precursor. DLF-1 was synthesized by conjugating fluorescent Cy5.5 with glycyl-vancomycin (Figure 1). Isothermal Titration Calorimetry (ITC) was applied to verify that DLF-1 binds to the D-Ala-D-Ala motif of PG. ITC measures the heat release (or absorption) associated with a binding event, offering an orthogonal, non-fluorescence based, validation of binding (Pierce et al., 1999). ITC shows that DLF-1 binds stoichiometrically (N = 0.99) to the peptide Ac-L-Lys-D-Ala-D-Ala (AcKAA) in PBS (Figure 2) with high affinity (Kd 2.3 μM). The binding affinity for DLF-1 is similar to parent molecule vancomycin (2.5 μM, Supplementary Table 1) and in line with prior studies (Leavitt and Freire, 2001; Rekharsky et al., 2006). These results suggest that DLF-1 has suitable biochemical target binding properties to advance to microbial profiling.
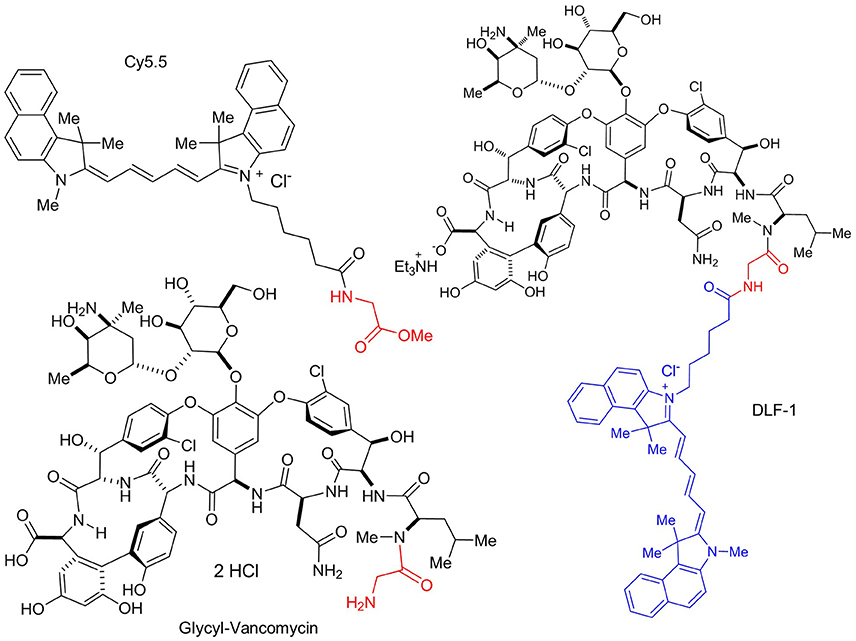
Figure 1. Structure of DLF-1. DLF-1 is synthesized by conjugating glycyl-vancomycin with a near infrared dye Cy5.5.
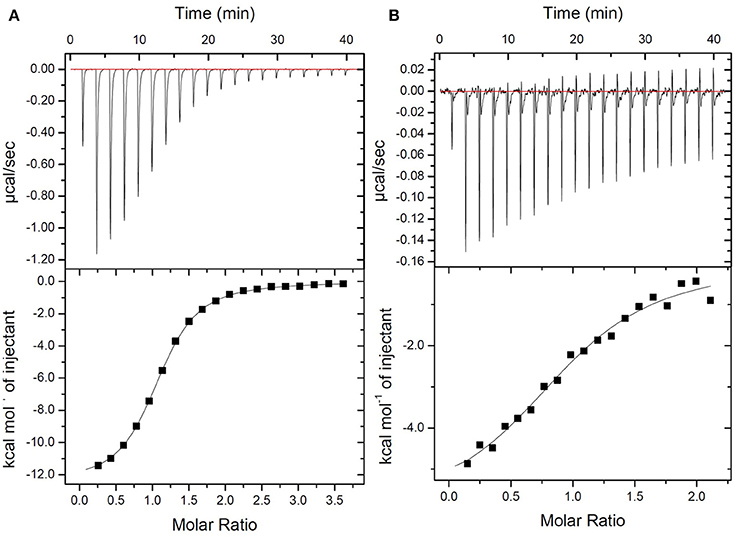
Figure 2. Thermodynamic evaluation of Ac-L-Lys-D-Ala-D-Ala-OH binding to DLF-1. Top: Raw curve of power vs. time. Bottom: Fit of integrated peak areas (heat released per injection) used to determine the thermodynamic parameters presented in Supplementary Table 1. (A) Representative ITC data measuring the thermodynamics of binding of peptide to Vancomycin. Peptide (600 μM) was titrated into a solution containing vancomycin (32 μM) in 1X PBS at 25°C. (B) Representative ITC data measuring the thermodynamics of binding of peptide to DLF-1. One percent of DMSO was added to control and test titrations, as it increased solubility of DLF-1.
DLF-1 Does Not Inhibit Growth of Bacteria at the Working Concentration
In order to label actively growing bacteria, chemical probes should not affect bacterial growth. We tested whether DLF-1 labeling inhibits growths of M. smegmatis, M. bovis bacillus Calmette-Guérin (BCG) and Mtb CDC1551. Two concentrations of DLF-1 were used to incubate with bacteria grown in liquid media. The growth curves of bacteria incubated with two concentrations of DLF-1 were compared with those without DLF-1. One μM of DLF-1 in culture media significantly inhibited growth of M. smegmatis, while having much less of effect on growth of M. bovis BCG and Mtb CDC1551. When 100 nM DLF-1 was used, no significant effect on growth was observed for Mtb (Figure 3). The MIC90 for DLF-1 against M. smegmatis was determined as 1 μg/ml (~0.45 μM) by broth microdilution method. We then decided to use 100 nM of DLF-1 for the remaining bacterial labeling.
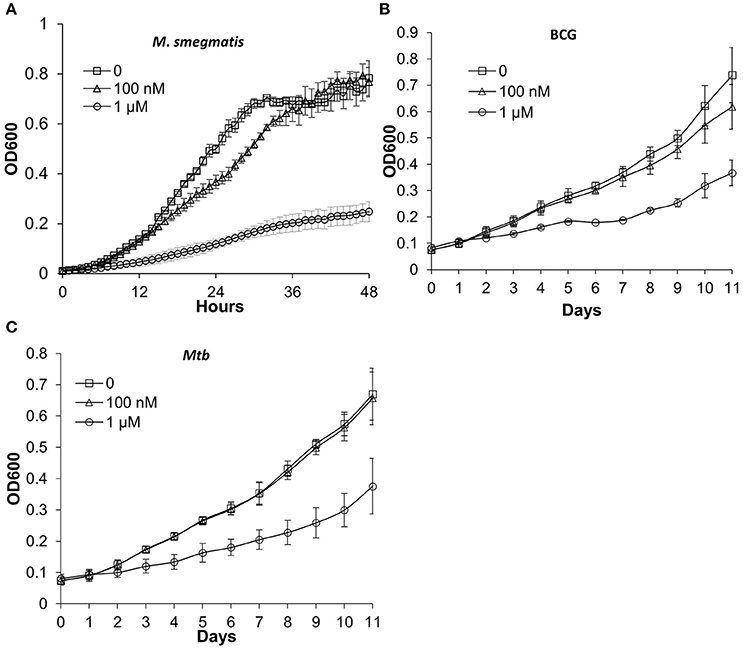
Figure 3. Mycobacterial growth curves in the presence or absence of DLF-1. (A–C) represent the growth curve of each mycobacterium in media containing 0, 100 nM, 1 μM of DLF-1, respectively. (A) Mycobacterium smegmatis; (B) Mycobacterium bovis BCG; (C) Mycobacterium tuberculosis CDC1551. Error bars represent standard errors of means calculated based on results from three independent experiments.
DLF-1 Can Be Used to Accurately Quantitate Mycobacteria In vitro
We have validated the application of DLF-1 to detection of actively replicating Mtb in vitro, in comparison with fast-growing M. smegmatis, and slow-growing M. bovis BCG. The correlation between fluorescent signal and bacterial numbers was evaluated after DLF-1 labeling of each Mycobacterium spp. Fluorescence generated from DLF-1 labeling linearly correlated with bacterial CFUs in all tested mycobacterial strains (Figure 4). The lowest number of Mtb within the linear range is 5.3 × 104 (P < 0.05, Figure 4D).
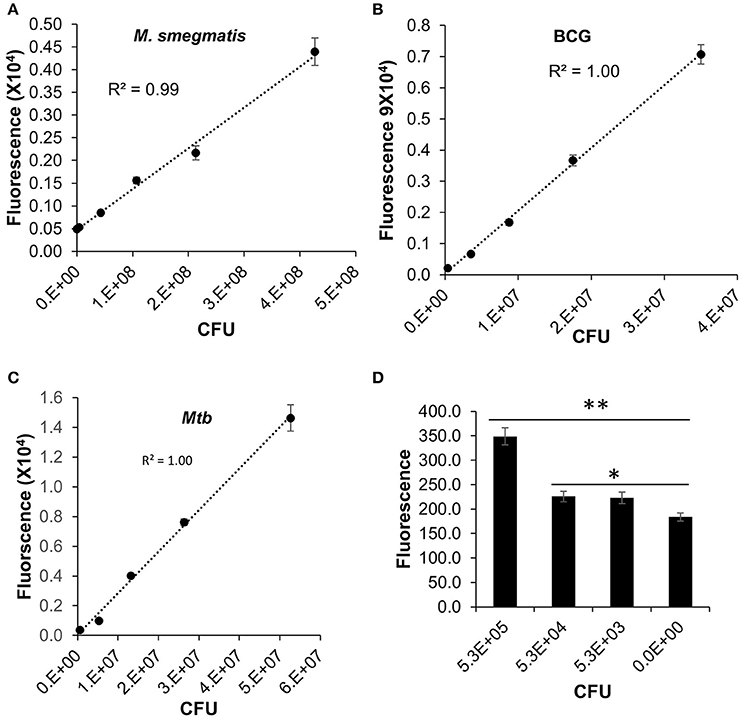
Figure 4. Direct labeling of different mycobacteria with DLF-1. Fluorescent intensities of DLF-1 labeled bacteria were plotted against the numbers of bacteria. (A–C) represent plots for M. Smegmatis, M. bovis BCG, and Mtb CDC1551, respectively. Linear trend lines with R-squares are also displayed for each plot. Linear range: M. smegmatis 4.3 × 105−4.3 × 107; M. bovis BCG 3.5 × 104−3.5 × 107; and Mtb 5.3 × 104−5.3 × 107. (D) Minimum number of Mtb within the detectable linear range. **P < 0.01; *P < 0.05. Error bars represent standard errors of means calculated based on results from three independent experiments.
We also examined the threshold for differentiation of DLF-1 labeled Mtb from the same number of Mtb without DLF-1 labeling using an optimal combination of excitation and emission wavelengths and gain value set up in the multimode reader. An Mtb strain carrying a plasmid expressing tdTomato was used (Kong et al., 2016), and the tdTomato fluorescence served as an internal control for DLF-1 labeling. When measured at the optimal excitation and emission wavelengths of the Cy5.5 fluorophore on DLF-1, DLF-1 labeled Mtb showed significantly higher fluorescence than the bacteria without labeling at each dilution of bacteria (Supplementary Figure 2A). When measured at the appropriate wavelengths for tdTomato, there was no such a difference between the two groups at any dilution of bacteria, suggesting there are the same bacterial numbers in the two groups at each dilution (Supplementary Figure 2B). The threshold of detection of Mtb for DLF-1 is ~103 CFU using the multimode plate reader with the optimal wavelengths and gain value described in Materials and Methods (P < 0.05). Although this condition can differentiate 103 CFU from the non-labeled control, the detected fluorescence is not correlated with CFU very well (R2 = 0.75).
DLF-1 Labeling Can Quantitate Mtb Inside of Cells
We examined if DLF-1 could be applied to image Mtb in cell infection models. A tdTomato-expressing Mtb strain was incubated with DLF-1 and infected THP-1 macrophages and A549 human lung epithelial cells. Fluorescence was detected from MOI = 10 in both cell lines (Figure 5A), and the fluorescence measured from DLF-1 labeled Mtb increased as MOIs rose. The fluorescent intensity of endogenous tdTomato among different MOI groups in A549 or THP-1 cells was measured in parallel with fluorescence of DLF-1. Cell infection with DLF-1 labeled Mtb was further verified by fluorescent microscopy. Slides with fixed infected cells were imaged with a fluorescent deconvolution microscope using pseudo colors: tdTomato-green, Cy5.5-red and DAPI-blue. As shown in Figure 5B, tdTomato and Cy5.5 are co-localized in cytoplasm of THP-1 and A549, which confirmed that DLF-1 can be applied to detect labeled Mtb inside of cells.
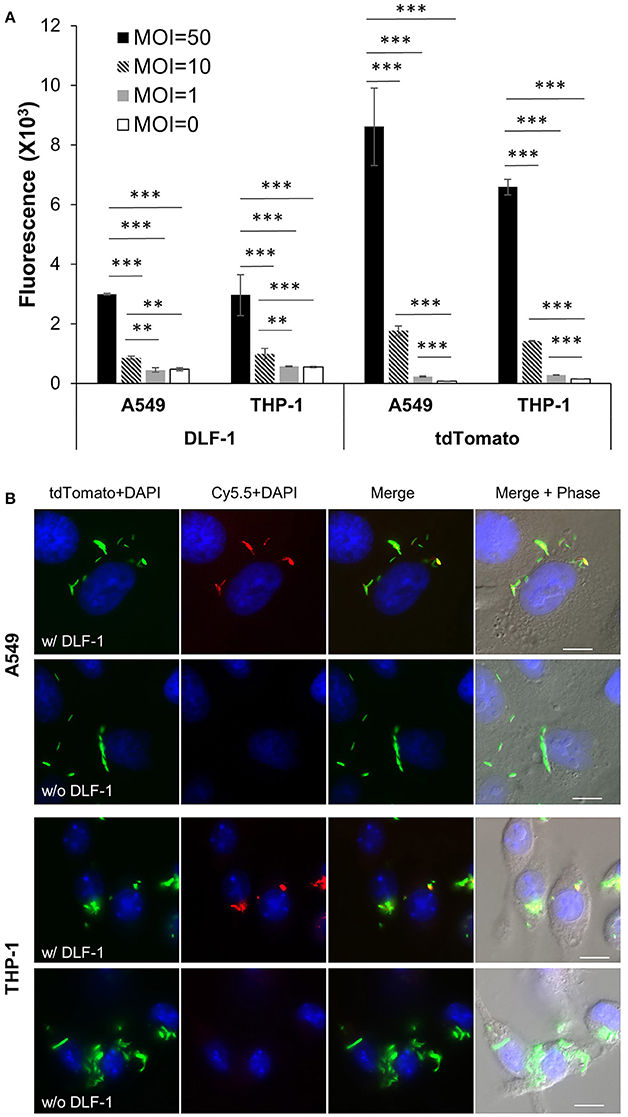
Figure 5. Imaging infection of human epithelial cell line (A549) and human macrophage cell line (THP-1) with DLF-1 (100 nM) labeled Mtb. (A) Fluorescence detected from cells infected with DLF-1 labeled Mtb at different MOIs in vitro. Fluorescent intensities were measured for the labeled DLF-1 (Cy5.5) and for the endogenous fluorescent tdTomato protein, respectively. Error bars represent standard errors of means calculated based on results from three independent experiments. One-way ANOVA test was conducted for assessing overall differences among groups, and Turkey's multiple comparison tests were applied to assess differences between two groups. **P < 0.01; and ***P < 0.001. (B) Images of DLF-1 labeled Mtb inside of cells. THP-1 and A549 were infected with a tdTomato-expressing Mtb strain which has been labeled with DLF-1. Red, pseudo-colored DLF-1; Green, pseudo-colored tdTomato; and Blue, DAPI. White scale bars represent 10 μm.
DLF-1 Can Label Non-replicating Dormant Mtb In vitro
In order to study molecular mechanisms involved in the onset of latency and/or reactivation, several in vitro and in vivo models have been developed to mimic LTBI in human. The Wayne model developed in 1996 is a well-characterized in vitro model based on hypoxic growth of Mtb (Wayne and Hayes, 1996). In this model, tubercle bacilli are cultured in an undisturbed liquid medium and exposed to oxygen depletion by a way of a completely sealed culture container. Mtb undergo an orderly process of metabolic shutdown, and develop resistance to rifampicin and isoniazid, but are sensitive to the anaerobic bactericide metronidazole (Wayne and Lin, 1982; Wayne, 1994; Wayne and Hayes, 1996). To validate DLF-1 in detecting dormant Mtb, we labeled dormant Mtb generated by the Wayne model with DLF-1.
An Mtb CDC1551 strain with a genome-integrated tdTomato gene was used in the Wayne model to generate dormant Mtb. With this strain, fluorescence of tdTomato can serve as an internal positive control for DLF-1 labeling. The dormant state of Mtb from this model was confirmed by verifying drug tolerance. After Mtb reached dormant state, 0.1 μg/ml rifampin was added in the culture for 3 days. The proportion of survived dormant Mtb reached 23.5 ± 4.8% (calculated as CFU of treated/CFU of untreated), which is consistent with previously reported results (Wayne and Hayes, 1996). With the same dose of rifampin, 97.5% of actively replicating Mtb was killed (Supplementary Table 2).
After the hypoxic dormant bacilli were obtained, cultures were washed with PBS, diluted into a series of concentrations, and incubated with DLF-1. The bacteria were plated for CFU enumeration. The fluorescent signal from labeled bacteria was measured with a multimode plate reader, using Cy5.5 and tdTomato wavelengths, respectively. Correlation between DLF-1 fluorescence and DLF-1 concentrations was analyzed first to determine dose-response relationship of DLF-1 labeling (Supplementary Figure 3A). The measured fluorescence increased as the concentration of DLF-1 went higher. Correlation between fluorescence and bacterial numbers was also analyzed using DLF-1 and tdTomato wavelengths, respectively (Figure 6A and Supplementary Figure 3B). Both DLF-1 specific fluorescence and tdTomato fluorescence correlated very well with bacterial CFUs. The threshold of detection for DLF-1 labeled dormant Mtb was determined as 3.1 × 105 CFU (Figure 6B). DLF-1 labeling dormant Mtb was also confirmed with fluorescent microscopy using a deconvolution microscope as shown in Figure 6C for bacterial smear and in Figure 6D for intracellular dormant Mtb in THP-1 cells. The red pseudo color from DLF-1 co-localized with the green pseudo color from tdTomato very well both in smear slides and in cell infection slides, which confirms that DLF-1 is capable of labeling dormant Mtb, and the fluorescent signal can be measured when bacteria are inside of cells.
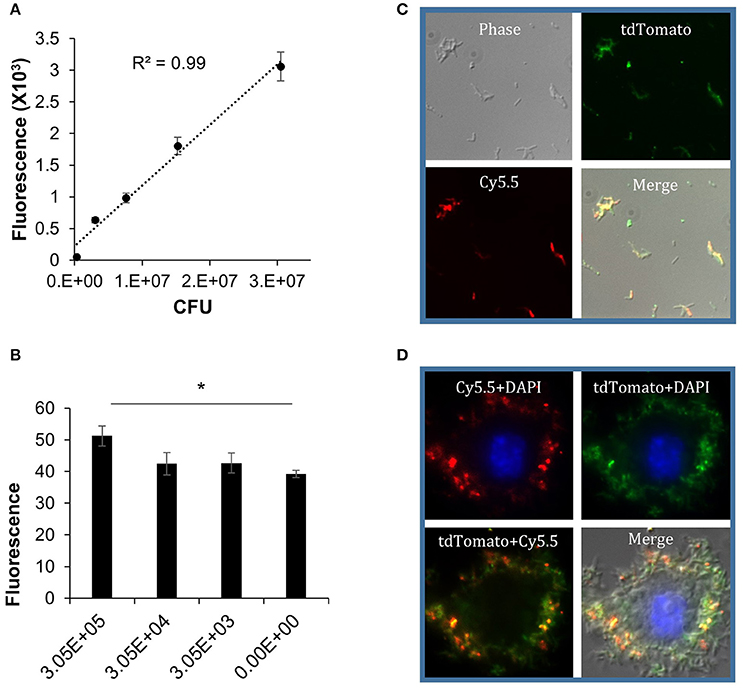
Figure 6. DLF-1 labeling of dormant Mtb cultured in Wayne model. (A) Correlation between fluorescence measured with DLF-1 wavelengths and bacterial numbers of dormant Mtb. Mtb was labeled with DLF-1 at 100 nM. (B) Threshold detection of DLF-1 labeling dormant Mtb in culture. *P < 0.05. (C) DLF-1 (100 nM) labeling dormant Mtb smeared on slides, and imaged with a fluorescent microscope. (D) Microscopy images of the DLF-1 (100 nM) labeled dormant Mtb strain carrying a genome-integrated tdTomato gene inside of THP-1 macrophages. Red, Cy5.5; Green, tdTomato; Blue, DAPI stained nuclei.
DLF-1 Can Be Applied to Screen for Mtb Genes Critical for Cell Invasion In vitro
Conventionally, identification of Mtb virulence genes relies on comparison of bacterial CFUs between mutant and wild type strains under specific conditions. DLF-1 labeling has great potential to replace CFU enumeration in vitro. As DLF-1 labeling can quantitate labeled Mtb inside cells, we set out to identify bacterial genes that are critical for cell entry. Previous studies have reported that the heparin-binding hemagglutinin (HbhA) of Mtb plays an important role in infecting epithelial cells (Pethe et al., 2001; Mueller-Ortiz et al., 2002; Parra et al., 2004; Choi et al., 2013). HbhA was initially identified in Mtb and M. bovis BCG (Menozzi et al., 1996, 1998). It is located on the surface of the mycobacterium and mediates binding of the bacillus to epithelial cells and fibroblasts (Menozzi et al., 1996). Deletion of hbhA (ΔhbhA) leads to reduced adherence of bacteria to epithelial cells but shows no effect on adherence to macrophages (Pethe et al., 2001). We evaluated entry rates of ΔhbhA and the wild type Mt103 strain in A549 human lung epithelial cells using DLF-1 labeling, in comparison with CFU enumeration. Infection ratios of ΔhbhA to the wild type strain Mt103 at a MOI of 50 was <1 based on both DLF-1 fluorescent intensity and CFU results (Table 1). The infection ratio calculated using DLF-1 fluorescent intensity was consistent with the infection ratio calculated using CFU, which suggests that DLF-1 can be applied to screen bacterial genes that are critical for cell infection, and the ΔhbhA strain can serve as a positive strain for this purpose.

Table 1. Bacterial cell infection ratios of mutants to the parental wild type strains calculated using DLF-1 labeling fluorescence and CFU, respectively.
Phospholipases are important bacterial virulence factors (Titball, 1998). Among them, phospholipase C (Plc) is important for replicating and cell-to-cell spread in mouse peritoneal macrophages for Listeria monocytogenes (Camilli et al., 1991). The Plc of Clostridium perfringenes is also named α toxin, which is haemolytic and suppresses neutrophil respiratory burst by disturbing host cell protein kinase C signaling (Terada et al., 1999). Mtb has four Plc genes, designated plcA, B, C, and D. The plcA, B, and C loci are clustered, while plcD is in a separated region in the chromosome of Mtb CDC1551. Expression of plc genes are upregulated at the 24-h post infection in macrophage infections, but deletions of plc genes do not impair growth in the human macrophage cell line THP-1. The plcABC triple mutant and plcABCD quadruple mutants are both attenuated in the late phase of mouse infection, indicating plc genes are important for Mtb survival in hosts (Raynaud et al., 2002).
We obtained the plcC mutant from BEI Resources (Manassas, VA) and examined its cell infection rate, in comparison to the infection rate of wild type CDC1551 in A549 cells. The bacterial infection ratio of plcC mutant to wide type CDC1551 was 0.7 ± 0.1 based on DLF-1 fluorescence intensity, and was 0.5 ± 0.2 based on CFU enumeration (Table 1). These results indicate that plcC is an important gene for Mtb entering alveolar epithelial cells. The detailed mechanism of how plcC helps bacilli infect epithelial cells remains to be explored.
Discussion
Bacterial cell walls are organized with layers of PG located immediately outside of the cytoplasmic membranes (Scheffers and Pinho, 2005; Shih and Rothfield, 2006; Silhavy et al., 2010). PG is composed of a polysaccharide backbone along with alternating N-acetylmuramic acid and N-acetylglucosamine residues. These chains are then cross-linked via D-Ala-D-Ala motifs by transpeptidases (penicillin binding proteins; Scheffers and Pinho, 2005; Shih and Rothfield, 2006; Silhavy et al., 2010). The peptide cross links are essential for providing structural integrity and rigidity to the bacterial cell wall (Höltje, 1998; Shih and Rothfield, 2006). Almost all bacterial cell walls contain PG, but not all of them have the same structures. Gram-positive bacteria have a thicker PG layer than Gram-negative bacteria (Shockman and Barrett, 1983; Beveridge, 1999; Shih and Rothfield, 2006; Silhavy et al., 2010). Like Gram-negative bacteria, the PG cell wall layer of mycobacteria lies below an outer membrane, comprised of arabinogalactan (AG), and mycolic acids (MA), which are covalently linked together to form a MA-AG-PG complex (Hett and Rubin, 2008). This MA-AG-PG complex is the essential core of the mycobacterial cell wall and often the target of many drugs used to combat mycobacteria.
Glycopeptide antibiotics such as vancomycin, teicoplanin, and telavancin act by binding to the terminal peptide D-Ala-D-Ala of PG precursors, and are widely used in the treatment of infections caused by Gram-positive bacteria, especially MRSA (Reynolds, 1989; Finch and Eliopoulos, 2005; Pace and Yang, 2006; Butler et al., 2014). Thus, the DLF-1 probe could be used to label Gram-positive bacteria. Vancomycin and its analogs can bind to the mycobacterial cell wall components PG and/or lipid II (MIC of vancomycin against Mtb: 2 μg/ml) (Lambert, 2002; Dinesh et al., 2013; Soetaert et al., 2015). To tag vancomycin, fluorescent molecules must be introduced at sites that do not interfere with its ability to bind to PG precursors. Vancomycin has two amine groups that are amenable to chemical modification. The amine located on the disaccharide is more chemically reactive (Kahne et al., 2005), and as the disaccharide is not involved in D-Ala-D-Ala binding, we attached Cy5.5 to this amine group to produce our probe, DLF-1. We verified that DLF-1 has a high affinity for D-Ala-D-Ala independently using isothermal titration calorimetry (ITC). Because this novel fluorescent labeling probe has vancomycin as an anchor, it is critical to confirm that DLF-1 does not affect bacterial growth at the working concentration for imaging. We have verified that 100 nM of DLF-1 does not affect Mtb growth.
Fluorescently labeled glycopeptide antibiotics have been used: (1) to detect bacterial infections in vivo (van Oosten et al., 2013), (2) to study the genes that control the biosynthesis of peptidoglycan (Tiyanont et al., 2006), and (3) to investigate cell wall synthesis in B. subtilis (Daniel and Errington, 2003). Recently, a study demonstrated that a fluorescent dye IR800CW conjugated with vancomycin can be applied to image Staphylococcus aureus infection in mice with a high specificity for detection of Gram-positive bacterial infections (van Oosten et al., 2013). We explored the application of the DLF-1 in detecting active replicating and dormant Mtb, and found that DLF-1 can effectively label both actively replicating and dormant Mtb. Furthermore, we developed an approach to use DLF-1 labeled Mtb to quickly screen for bacterial genes that are important for infectious and nonphagocytic cells. Because the DLF-1 imaging method does not require a recombinant strain, it can be directly applied to an established mutant library to study bacterial genes in vitro. This application of DLF-1 has been verified by using the ΔhbhA mutant of Mt103 strain. It is well-known that HbhA is critical for Mtb infection of epithelial cells (Pethe et al., 2001; Mueller-Ortiz et al., 2002; Parra et al., 2004; Choi et al., 2013). CFU enumeration results confirm that deletion of the hbhA gene did impair bacterial infection in A549 cells, as reported previously (Pethe et al., 2001). Using ΔhbhA as a positive control, we have found by DLF-1 labeling and CFU enumeration that another gene of Mtb, plcC, is also important for bacterial entry of epithelial cells. To our knowledge, this is the first report showing that plcC plays an important role for infecting epithelial cells. The fact of mutant of hbhA or plcC had reduced entry rates but was still able to enter epithelial cells indicates that Mtb has other genes that compensate function of HbhA or PlcC. As lung epithelial cells are the first barriers to Mtb infection, how Mtb enters epithelial cells is very critical for establishment of lung infection at earlier stages. It remains to further explore the detailed mechanism of plcC in lung infection and if the other three plc genes of Mtb share similar function.
The novel fluorescent imaging method described here provides a useful substitute for traditional time-consuming, labor-intensive approaches of quantitating bacterial numbers in TB research in vitro. However, at current stage, DLF-1 cannot be directly applied to TB diagnostic assays as it is not a mycobacterial specific probe. It remains to be investigated if DLF-1 is capable of in vivo imaging of Mtb infection in live animal models and ex vivo imaging of infected organs. The dye Cy5.5 on DLF-1 makes it ideal for in vivo imaging, as its excitation and emission wavelengths are within the “near infrared window,” in which the absorbance coefficients of hemoglobin water and hemoglobin have less absorbance, so excitation light can penetrate through the animal without significant loss (Weissleder, 2001). In the future, we will validate the sensitivity, reproducibility and accuracy of DLF-1, by quantitating bacterial numbers in Mtb infection in vivo. We will also evaluate application of this imaging technique to quantification of drug response in anti-TB therapy. As DLF-1 labels both viable bacteria and dead ones, it can be utilized together with other techniques that measure viable bacteria only, such as fluorescence protein expressing strains, to evaluate bactericidal activities in laboratory. The dead bacterial number could be calculated as the number of bacteria estimated by DLF-1 labeling subtracting the number of bacteria estimated by measuring fluorescent intensity of the fluorescence protein expressing strain. In summary, this strategy could be applied to preclinical screening therapies in a more time-efficient manner, and be used to real-time quantitate bacterial load while studying bacterial genes and host immune factors involved in active TB or LTBI. The success of these studies would accelerate screening for anti-TB therapy in animal models and enhance studies of bacterial and host factors in TB.
Author Contributions
Conceived and designed the experiments: DY, FD, KM, RL, MK, and YK. Performed the experiments: DY, FD, KM, and YK. Analyzed the data: DY, FD, KM, RL, MK, and YK. Contributed reagents/materials/analysis tools: RL, MK, and YK. Wrote the manuscript: DY, FD, and YK.
Funding
This work was supported by the National Heart, Lung, And Blood Institute of the National Institutes of Health under award number R21HL115463, University of Tennessee Health Science Center start-up funding, and the American Lebanese Syrian Associated Charities, St. Jude Children's Research Hospital.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The plasmids and mycobacteria strains were kindly provided by Dr. Jeffrey D. Cirillo. The hbhA mutant strain was originally from Dr. Camille Locht's laboratory. We would like to thank Dr. William Shadrick of St. Jude Children's Research Hospital, for invaluable help with ITC and manuscript preparation.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fmicb.2016.02021/full#supplementary-material
Abbreviations
TB, tuberculosis; Mtb, Mycobacterium tuberculosis; CFU, colony forming unit; ITC, isothermal titration calorimetry; PBS, Phosphate buffered saline; MOI, Multiplicity of infection; PG, peptidoglycan; AG', arabinogalactan; MA, Mycolic acid; D-Ala-D-Ala, D-alanyl-D-alanine.
References
Andreu, N., Fletcher, T., Krishnan, N., Wiles, S., and Robertson, B. D. (2012). Rapid measurement of antituberculosis drug activity in vitro and in macrophages using bioluminescence. J. Antimicrob. Chemother. 67, 404–414. doi: 10.1093/jac/dkr472
Andreu, N., Zelmer, A., Fletcher, T., Elkington, P. T., Ward, T. H., Ripoll, J., et al. (2010). Optimisation of bioluminescent reporters for use with mycobacteria. PLoS ONE 5:e10777. doi: 10.1371/journal.pone.0010777
Andreu, N., Zelmer, A., Sampson, S. L., Ikeh, M., Bancroft, G. J., Schaible, U. E., et al. (2013). Rapid in vivo assessment of drug efficacy against Mycobacterium tuberculosis using an improved firefly luciferase. J. Antimicrob. Chemother. 68, 2118–2127. doi: 10.1093/jac/dkt155
Andrew, P. W., and Roberts, I. S. (1993). Construction of a bioluminescent mycobacterium and its use for assay of antimycobacterial agents. J. Clin. Microbiol. 31, 2251–2254.
Arain, T. M., Resconi, A. E., Hickey, M. J., and Stover, C. K. (1996). Bioluminescence screening in vitro (Bio-Siv) assays for high-volume antimycobacterial drug discovery. Antimicrob. Agents Chemother. 40, 1536–1541.
Bange, F. C., Collins, F. M., and Jacobs, W. R. Jr. (1999). Survival of mice infected with Mycobacterium smegmatis containing large DNA fragments from Mycobacterium tuberculosis. Tuber. Lung Dis. 79, 171–180. doi: 10.1054/tuld.1998.0201
Beveridge, T. J. (1999). Structures of gram-negative cell walls and their derived membrane vesicles. J. Bacteriol. 181, 4725–4733.
Butler, M. S., Hansford, K. A., Blaskovich, M. A., Halai, R., and Cooper, M. A. (2014). Glycopeptide antibiotics: back to the future. J. Antibiot. 67, 631–644. doi: 10.1038/ja.2014.111
Camilli, A., Goldfine, H., and Portnoy, D. A. (1991). Listeria monocytogenes mutants lacking phosphatidylinositol-specific phospholipase C are avirulent. J. Exp. Med. 173, 751–754. doi: 10.1084/jem.173.3.751
Carroll, P., Schreuder, L. J., Muwanguzi-Karugaba, J., Wiles, S., Robertson, B. D., Ripoll, J., et al. (2010). Sensitive detection of gene expression in mycobacteria under replicating and non-replicating conditions using optimized far-red reporters. PLoS ONE 5:e9823. doi: 10.1371/journal.pone.0009823
Choi, J. A., Lim, Y. J., Cho, S. N., Lee, J. H., Jeong, J. A., Kim, E. J., et al. (2013). Mycobacterial HBHA induces endoplasmic reticulum stress-mediated apoptosis through the generation of reactive oxygen species and cytosolic Ca2+ in murine macrophage RAW 264.7 cells. Cell Death Dis. 4, e957. doi: 10.1038/cddis.2013.489
Cooksey, R. C., Crawford, J. T., Jacobs, W. R. Jr., and Shinnick, T. M. (1993). A rapid method for screening antimicrobial agents for activities against a strain of Mycobacterium tuberculosis expressing firefly luciferase. Antimicrob. Agents Chemother. 37, 1348–1352. doi: 10.1128/AAC.37.6.1348
Coulson, N. M., Fulop, M., and Titball, R. W. (1994). Effect of different plasmids on colonization of mouse tissues by the aromatic amino acid dependent Salmonella typhimurium SL 3261. Microb. Pathog. 16, 305–311. doi: 10.1006/mpat.1994.1031
Daniel, R. A., and Errington, J. (2003). Control of cell morphogenesis in bacteria: two distinct ways to make a rod-shaped cell. Cell 113, 767–776. doi: 10.1016/S0092-8674(03)00421-5
Dinesh, N., Sharma, S., and Balganesh, M. (2013). Involvement of efflux pumps in the resistance to peptidoglycan synthesis inhibitors in Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 57, 1941–1943. doi: 10.1128/AAC.01957-12
Finch, R. G., and Eliopoulos, G. M. (2005). Safety and efficacy of glycopeptide antibiotics. J. Antimicrob. Chemother. 55(Suppl. 2), ii5–ii13. doi: 10.1093/jac/dki004
Glickman, M. S., and Jacobs, W. R. Jr. (2001). Microbial pathogenesis of Mycobacterium tuberculosis: dawn of a discipline. Cell 104, 477–485. doi: 10.1016/S0092-8674(01)00236-7
Hett, E. C., and Rubin, E. J. (2008). Bacterial growth and cell division: a mycobacterial perspective. Microbiol. Mol. Biol. Rev. 72, 126–156. doi: 10.1128/MMBR.00028-07
Heuts, F., Carow, B., Wigzell, H., and Rottenberg, M. E. (2009). Use of non-invasive bioluminescent imaging to assess mycobacterial dissemination in mice, treatment with bactericidal drugs and protective immunity. Microbes Infect. 11, 1114–1121. doi: 10.1016/j.micinf.2009.08.005
Hickey, M. J., Arain, T. M., Shawar, R. M., Humble, D. J., Langhorne, M. H., Morgenroth, J. N., et al. (1996). Luciferase in vivo expression technology: use of recombinant mycobacterial reporter strains to evaluate antimycobacterial activity in mice. Antimicrob. Agents Chemother. 40, 400–407.
Höltje, J. V. (1998). Growth of the stress-bearing and shape-maintaining murein sacculus of Escherichia coli. Microbiol. Mol. Biol. Rev. 62, 181–203.
Kahne, D., Leimkuhler, C., Lu, W., and Walsh, C. (2005). Glycopeptide and lipoglycopeptide antibiotics. Chem. Rev. 105, 425–448. doi: 10.1021/cr030103a
Kong, Y., Yang, D., Cirillo, S. L., Li, S., Akin, A., Francis, K. P., et al. (2016). Application of fluorescent protein expressing strains to evaluation of anti-tuberculosis therapeutic efficacy in vitro and in vivo. PLoS ONE 11:e0149972. doi: 10.1371/journal.pone.0149972
Kong, Y., Yao, H., Ren, H., Subbian, S., Cirillo, S. L., Sacchettini, J. C., et al. (2010). Imaging tuberculosis with endogenous beta-lactamase reporter enzyme fluorescence in live mice. Proc. Natl. Acad. Sci. U.S.A. 107, 12239–12244. doi: 10.1073/pnas.1000643107
Lambert, P. A. (2002). Cellular impermeability and uptake of biocides and antibiotics in Gram-positive bacteria and mycobacteria. J. Appl. Microbiol. 92(Suppl. 46S–54S). doi: 10.1046/j.1365-2672.92.5s1.7.x
Leavitt, S., and Freire, E. (2001). Direct measurement of protein binding energetics by isothermal titration calorimetry. Curr. Opin. Struct. Biol. 11, 560–566. doi: 10.1016/S0959-440X(00)00248-7
Mehta, P. K., King, C. H., White, E. H., Murtagh, J. J. Jr., and Quinn, F. D. (1996). Comparison of in vitro models for the study of Mycobacterium tuberculosis invasion and intracellular replication. Infect. Immun. 64, 2673–2679.
Menozzi, F. D., Bischoff, R., Fort, E., Brennan, M. J., and Locht, C. (1998). Molecular characterization of the mycobacterial heparin-binding hemagglutinin, a mycobacterial adhesin. Proc. Natl. Acad. Sci. U.S.A. 95, 12625–12630. doi: 10.1073/pnas.95.21.12625
Menozzi, F. D., Rouse, J. H., Alavi, M., Laude-Sharp, M., Muller, J., Bischoff, R., et al. (1996). Identification of a heparin-binding hemagglutinin present in mycobacteria. J. Exp. Med. 184, 993–1001. doi: 10.1084/jem.184.3.993
Miltner, E., Daroogheh, K., Mehta, P. K., Cirillo, S. L., Cirillo, J. D., and Bermudez, L. E. (2005). Identification of Mycobacterium avium genes that affect invasion of the intestinal epithelium. Infect. Immun. 73, 4214–4221. doi: 10.1128/IAI.73.7.4214-4221.2005
Mueller-Ortiz, S. L., Sepulveda, E., Olsen, M. R., Jagannath, C., Wanger, A. R., and Norris, S. J. (2002). Decreased infectivity despite unaltered C3 binding by a DeltahbhA mutant of Mycobacterium tuberculosis. Infect. Immun. 70, 6751–6760. doi: 10.1128/IAI.70.12.6751-6760.2002
Napier, R. J., Rafi, W., Cheruvu, M., Powell, K. R., Zaunbrecher, M. A., Bornmann, W., et al. (2011). Imatinib-sensitive tyrosine kinases regulate mycobacterial pathogenesis and represent therapeutic targets against tuberculosis. Cell Host Microbe 10, 475–485. doi: 10.1016/j.chom.2011.09.010
Ollinger, J., Bailey, M. A., Moraski, G. C., Casey, A., Florio, S., Alling, T., et al. (2013). A dual read-out assay to evaluate the potency of compounds active against Mycobacterium tuberculosis. PLoS ONE 8:e60531. doi: 10.1371/journal.pone.0060531
Pace, J. L., and Yang, G. (2006). Glycopeptides: update on an old successful antibiotic class. Biochem. Pharmacol. 71, 968–980. doi: 10.1016/j.bcp.2005.12.005
Parra, M., Pickett, T., Delogu, G., Dheenadhayalan, V., Debrie, A. S., Locht, C., et al. (2004). The mycobacterial heparin-binding hemagglutinin is a protective antigen in the mouse aerosol challenge model of tuberculosis. Infect. Immun. 72, 6799–6805. doi: 10.1128/IAI.72.12.6799-6805.2004
Pethe, K., Alonso, S., Biet, F., Delogu, G., Brennan, M. J., Locht, C., et al. (2001). The heparin-binding haemagglutinin of M. tuberculosis is required for extrapulmonary dissemination. Nature 412, 190–194. doi: 10.1038/35084083
Pierce, M. M., Raman, C. S., and Nall, B. T. (1999). Isothermal titration calorimetry of protein-protein interactions. Methods 19, 213–221. doi: 10.1006/meth.1999.0852
Rang, C., Galen, J. E., Kaper, J. B., and Chao, L. (2003). Fitness cost of the green fluorescent protein in gastrointestinal bacteria. Can. J. Microbiol. 49, 531–537. doi: 10.1139/w03-072
Raynaud, C., Guilhot, C., Rauzier, J., Bordat, Y., Pelicic, V., Manganelli, R., et al. (2002). Phospholipases C are involved in the virulence of Mycobacterium tuberculosis. Mol. Microbiol. 45, 203–217. doi: 10.1046/j.1365-2958.2002.03009.x
Rekharsky, M. V., Yamamura, H., Inoue, C., Kawai, M., Osaka, I., Arakawa, R., et al. (2006). Chiral recognition in cucurbituril cavities. J. Am. Chem. Soc. 128, 14871–14880. doi: 10.1021/ja063323p
Reynolds, P. E. (1989). Structure, biochemistry and mechanism of action of glycopeptide antibiotics. Eur. J. Clin. Microbiol. Infect. Dis. 8, 943–950. doi: 10.1007/BF01967563
Scheffers, D. J., and Pinho, M. G. (2005). Bacterial cell wall synthesis: new insights from localization studies. Microbiol. Mol. Biol. Rev. 69, 585–607. doi: 10.1128/MMBR.69.4.585-607.2005
Shih, Y. L., and Rothfield, L. (2006). The bacterial cytoskeleton. Microbiol. Mol. Biol. Rev. 70, 729–754. doi: 10.1128/MMBR.00017-06
Shockman, G. D., and Barrett, J. F. (1983). Structure, function, and assembly of cell walls of gram-positive bacteria. Annu. Rev. Microbiol. 37, 501–527. doi: 10.1146/annurev.mi.37.100183.002441
Silhavy, T. J., Kahne, D., and Walker, S. (2010). The bacterial cell envelope. Cold Spring Harb. Perspect. Biol. 2:a000414. doi: 10.1101/cshperspect.a000414
Singh, V., Biswas, R. K., and Singh, B. N. (2014). Double recombinant Mycobacterium bovis BCG strain for screening of primary and rationale-based antimycobacterial compounds. Antimicrob. Agents Chemother. 58, 1389–1396. doi: 10.1128/AAC.01301-13
Smith, I. (2003). Mycobacterium tuberculosis pathogenesis and molecular determinants of virulence. Clin. Microbiol. Rev. 16, 463–496. doi: 10.1128/CMR.16.3.463-496.2003
Soetaert, K., Rens, C., Wang, X. M., De Bruyn, J., Lanéelle, M. A., Laval, F., et al. (2015). Increased vancomycin susceptibility in mycobacteria: a new approach to identify synergistic activity against multidrug-resistant mycobacteria. Antimicrob. Agents Chemother. 59, 5057–5060. doi: 10.1128/AAC.04856-14
Sudre, P., ten Dam, G., and Kochi, A. (1992). Tuberculosis - a global overview of the situation today. Bull. World Health Organ. 70, 149–159.
Swenson, J. M., Thornsberry, C., and Silcox, V. A. (1982). Rapidly growing mycobacteria: testing of susceptibility to 34 antimicrobial agents by broth microdilution. Antimicrob. Agents Chemother. 22, 186–192. doi: 10.1128/AAC.22.2.186
Terada, L. S., Johansen, K. A., Nowbar, S., Vasil, A. I., and Vasil, M. L. (1999). Pseudomonas aeruginosa hemolytic phospholipase C suppresses neutrophil respiratory burst activity. Infect. Immun. 67, 2371–2376.
Tiyanont, K., Doan, T., Lazarus, M. B., Fang, X., Rudner, D. Z., and Walker, S. (2006). Imaging peptidoglycan biosynthesis in Bacillus subtilis with fluorescent antibiotics. Proc. Natl. Acad. Sci. U.S.A. 103, 11033–11038. doi: 10.1073/pnas.0600829103
van Oosten, M., Schäfer, T., Gazendam, J. A., Ohlsen, K., Tsompanidou, E., de Goffau, M. C., et al. (2013). Real-time in vivo imaging of invasive- and biomaterial-associated bacterial infections using fluorescently labelled vancomycin. Nat. Commun. 4, 2584. doi: 10.1038/ncomms3584
Vocat, A., Hartkoorn, R. C., Lechartier, B., Zhang, M., Dhar, N., Cole, S. T., et al. (2015). Bioluminescence for assessing drug potency against nonreplicating Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 59, 4012–4019. doi: 10.1128/AAC.00528-15
Wayne, L. G. (1994). Dormancy of Mycobacterium tuberculosis and latency of disease. Eur. J. Clin. Microbiol. Infect. Dis. 13, 908–914. doi: 10.1007/BF02111491
Wayne, L. G., and Hayes, L. G. (1996). An in vitro model for sequential study of shiftdown of Mycobacterium tuberculosis through two stages of nonreplicating persistence. Infect. Immun. 64, 2062–2069.
Wayne, L. G., and Lin, K. Y. (1982). Glyoxylate metabolism and adaptation of Mycobacterium tuberculosis to survival under anaerobic conditions. Infect. Immun. 37, 1042–1049.
Weissleder, R. (2001). A clearer vision for in vivo imaging. Nat. Biotechnol. 19, 316–317. doi: 10.1038/86684
Wendland, M., and Bumann, D. (2002). Optimization of GFP levels for analyzing Salmonella gene expression during an infection. FEBS Lett. 521, 105–108. doi: 10.1016/S0014-5793(02)02834-X
Zelmer, A., Carroll, P., Andreu, N., Hagens, K., Mahlo, J., Redinger, N., et al. (2012). A new in vivo model to test anti-tuberculosis drugs using fluorescence imaging. J. Antimicrob. Chemother. 67, 1948–1960. doi: 10.1093/jac/dks161
Zhang, T., Li, S. Y., and Nuermberger, E. L. (2012). Autoluminescent Mycobacterium tuberculosis for rapid, real-time, non-invasive assessment of drug and vaccine efficacy. PLoS ONE 7:e29774. doi: 10.1371/journal.pone.0029774
Keywords: Mycobacterium tuberculosis, dormant M. tuberculosis, fluorescence, imaging
Citation: Yang D, Ding F, Mitachi K, Kurosu M, Lee RE and Kong Y (2016) A Fluorescent Probe for Detecting Mycobacterium tuberculosis and Identifying Genes Critical for Cell Entry. Front. Microbiol. 7:2021. doi: 10.3389/fmicb.2016.02021
Received: 10 August 2016; Accepted: 02 December 2016;
Published: 20 December 2016.
Edited by:
Michael Benedik, Texas A&M University, USAReviewed by:
Javier Bezos, Complutense University of Madrid, SpainAnthony Baughn, University of Minnesota, USA
Kurt L. Krause, University of Otago, New Zealand
Copyright © 2016 Yang, Ding, Mitachi, Kurosu, Lee and Kong. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ying Kong, eWtvbmczQHV0aHNjLmVkdQ==
†Present Address: Feng Ding, Department of Physiology, School of Basic Medical Sciences, Wenzhou Medical University-Chashan Campus, Wenzhou, China
‡These authors have contributed equally to this work.
 Dong Yang1‡
Dong Yang1‡ Feng Ding
Feng Ding Michio Kurosu
Michio Kurosu Richard E. Lee
Richard E. Lee Ying Kong
Ying Kong