- 1State Key Laboratory of Environmental Chemistry and Ecotoxicology, Research Center for Eco-Environmental Sciences, Chinese Academy of Sciences, Beijing, China
- 2College of Resources and Environment, University of Chinese Academy of Sciences, Beijing, China
- 3Department of Municipal and Environmental Engineering, School of Civil Engineering, Beijing Jiaotong University, Beijing, China
Numerous genes are involved in various strategies to resist toxic arsenic (As). However, the As resistance strategy in genus Pantoea is poorly understood. In this study, a comparative genome analysis of 23 Pantoea genomes was conducted. Two vertical genetic arsC-like genes without any contribution to As resistance were found to exist in the 23 Pantoea strains. Besides the two arsC-like genes, As resistance gene clusters arsRBC or arsRBCH were found in 15 Pantoea genomes. These ars clusters were found to be acquired by horizontal gene transfer (HGT) from sources related to Franconibacter helveticus, Serratia marcescens, and Citrobacter freundii. During the history of evolution, the ars clusters were acquired more than once in some species, and were lost in some strains, producing strains without As resistance capability. This study revealed the organization, distribution and the complex evolutionary history of As resistance genes in Pantoea spp.. The insights gained in this study improved our understanding on the As resistance strategy of Pantoea spp. and its roles in the biogeochemical cycling of As.
Introduction
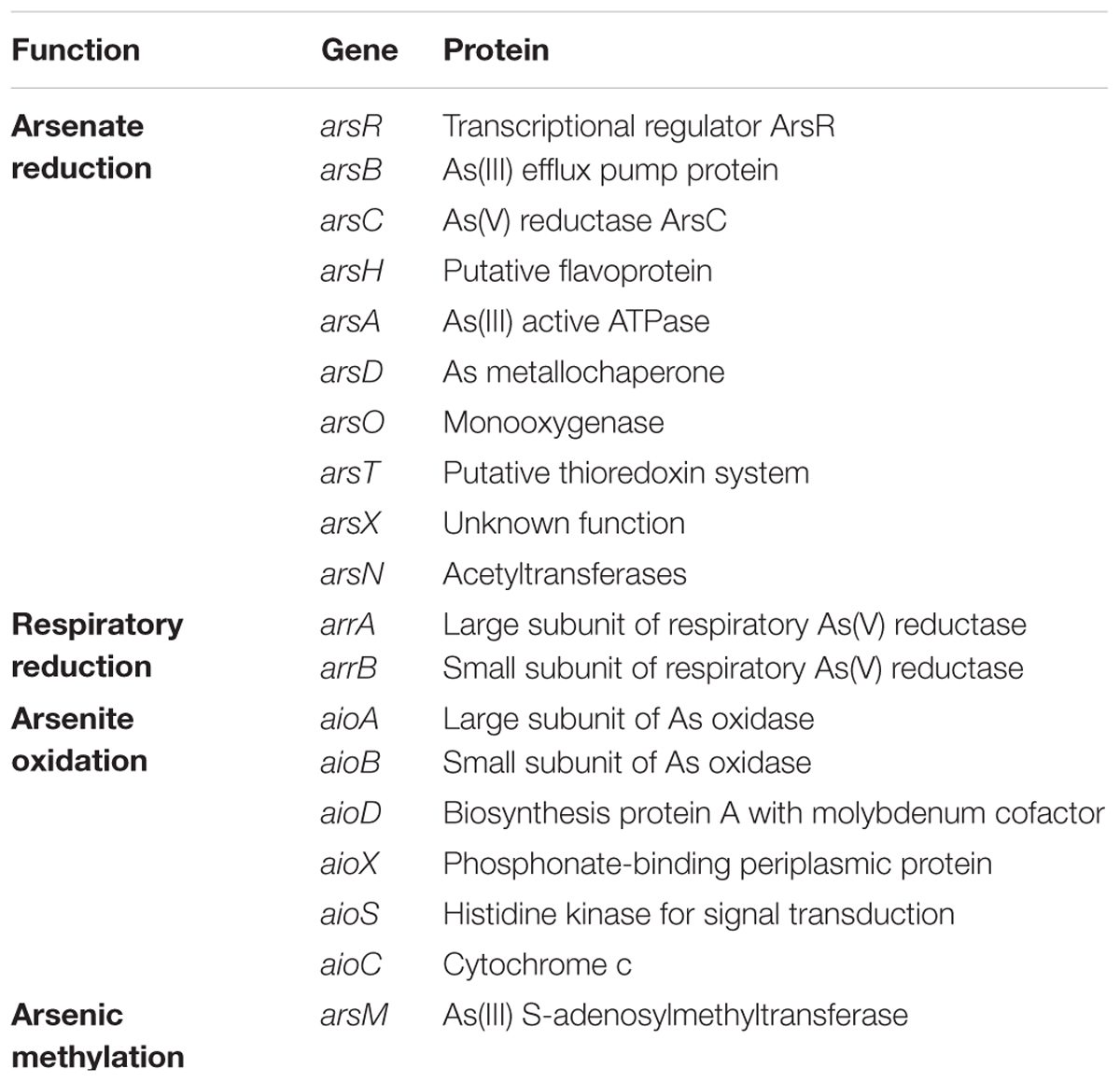
Arsenic (As), one of the earliest known toxic elements, occurs naturally worldwide (Smith et al., 2002). To adapt to habitats with elevated As, microbes have evolved dynamic resistance mechanisms. The most ubiquitous and important strategy of As resistance is to reduce As(V) to As(III) and extrude it using ars operons with various genomic configurations in specific bacterial strains (Páezespino et al., 2009). The core genes of ars systems, however, are arsR (encoding the transcriptional repressor ArsR), arsB (encoding the arsenite efflux pump ArsB) and arsC (encoding arsenate reductase ArsC) (Xu et al., 1998). Besides this detoxification mechanism using ars systems, some strains possess the mechanism of As methylation-demethylation, changing inorganic As into organic forms using a distinct gene arsM (Qin et al., 2006; Zhao et al., 2015). Some strains are able to oxidize As(III) to As(V), which involve membrane-associated proteins, AoxAB (Levin and Tal, 2003; Ghosh et al., 2014). Some strains are able to reduce As(V) to As(III) with ArrAB as part of their respiratory processes transferring electrons to As and producing the energy for strains (Saltikov and Newman, 2003). The reported genes related to the strategy of As resistance are listed in Table 1.
In traditional molecular biology research, As resistance traits are revealed primarily based on the cultivation of a specific strain, and it is impossible to study the As strategy of all strains in a genus. Nevertheless, understanding of such traits in all strains of a genus is sometime more desirable. Gaining this knowledge is no longer a challenge with the explosive development of high-throughput sequencing technology. The genomic sequence of a strain contains nearly all of the genetic information. Therefore, fundamental knowledge such as the phylogenetic, the genetic traits of As resistance and its evolutionary history can be obtained through comparative genomic analysis (Arsène-Ploetze et al., 2010; Colston et al., 2014). So here we use genomic information of Pantoea spp. and compared these genomes to explore and predict the strategy of As resistance and their evolutionary patterns in genus Pantoea as an example.
Pantoea is a genus of Gram-negative, facultative anaerobic bacteria. This genus belongs to gamma Proteobacteria, family Gammaproteobacteria, and was recently separated from the genus Enterobacter (Gavini et al., 1989). Currently, the genus contains 26 species1. Members of this genus are found in various environmental matrices (Meng et al., 1995; Zhang and Birch, 1997; Rezzonico et al., 2009). In 2013, the strain Pantoea sp. IMH was an isolate that reported firstly as the strain having the As resistance capability within Pantoea species (Wu et al., 2013). Further, we sequenced the genome of Pantoea sp. IMH and found two ars clusters (arsR1B1C1H1 and arsR2B2C2H2) co-contributing to its As resistance (Tian and Jing, 2014; Wang et al., 2016). However, the evolutionary history and genetic traits of As resistance in genus Pantoea are not fully understood.
Herein, we present the first study of the genetic traits of As resistance in Pantoea spp., as well as their evolutionary history. Two vertically transmitted arsC-like genes without any contribution to As resistance were found to exist in the 23 Pantoea strains. Besides these two arsC-like genes, As resistance gene clusters arsRBC or arsRBCH were found in 15 Pantoea genomes. These ars clusters were acquired by horizontal gene transfer (HGT) from sources related to Franconibacter helveticus, Serratia marcescens, and Citrobacter freundii. The insights gained in this study improve our understanding on the complex evolutionary history of As resistance genes and their roles in the biogeochemical cycling of As.
Materials and Methods
Phylogenetic Analysis
Phylogenetic trees of Pantoea species were constructed based on 100 single-copy core proteins shared by 23 Pantoea genomes and the genome of Tatumella sp. NML 06-3099 according to the following three methods: maximum likelihood (ML), neighbor joining (NJ), and Bayesian inference (BI). ML and NJ trees were computed by applying models with 1,000 bootstrap replicates and uniform rates in MEGA5 (Tamura, 2011). Multiple alignments of amino acid sequences were carried out by ClustalW, and the CONSEL program was used to select the best model of the trees (Shimodaira and Hasegawa, 2001; Thompson et al., 2002). The BI tree was generated using the MrBayes package with mixed models (Ronquist et al., 2012). The NJ tree of concatenated arsRBC homologs was generated according to the same method described above. MEGA5 or FigTree v.1.3.12 was used to illustrate the constructed trees.
Average Nucleotide Identity (ANI)
Assembled contigs were reconstituted from the RAST-generated GenBank files for 23 genomes by using the seqret function of the EMBOSS package (Rice et al., 2000). These 23 genomes were treated in the same manner to ensure that any biases were consistent across the entire dataset. JSpecies1.2.1 was used to analyze these contig sets for the ANI and tetramer usage patterns, using default parameters (Richter and Rosselló-Móra, 2009).
Comparative Genomics
All of the orthologous pairs between Pantoea test genomes were identified by Pan Genome Analysis Pipeline (PGAP) (Zhao et al., 2012). The common dataset of shared genes among test strains was defined as their core genome. The total set of genes with test genomes was defined as the pan genome. The set of genes in each strain not shared with other strains was defined as the unique genes. The details of the strains used are listed in Supplementary Table S1.
Construction of the Recombinant Plasmids and Escherichia coli Strains
A 3.86 kb BamHI-XbaI DNA fragment containing the complete ars1 cluster of Pantoea stewartii S301 (promoter region, 342 bp upstream of the start codon ATG of arsR, the contiguous four genes arsR1B1C1H1 and 281 bp upstream of the start codon ATG of arsH) was PCR amplified with primers Ars1-F and Ars1-R (Supplementary Table S2). A 3.43 kb BamHI-XbaI DNA fragment containing the complete ars2 gene cluster of P. agglomerans Tx10 (a 280 bp region downstream of the stop codon TAA of arsC2 and the contiguous ten genes arsR2B2C2H2 and 328 bp downstream of the stop codon TAA of arsH2) was PCR amplified with primers Ars2-F and Ars2-R (Supplementary Table S2).
An 860 bp BamHI-XbaI DNA fragment containing the complete arsC1-like gene of P. stewartii DC283 (promoter region, 221 bp upstream of the start codon ATG of arsC1-like gene, arsC1-like and 209 bp downstream of the stop codon TTA of arsC1-like gene) was PCR amplified with primers ArsC1-like-F and ArsC1-like-R (Supplementary Table S2). A 942 bp BamHI-XbaI DNA fragment containing the complete arsC2-like gene of P. stewartii DC283 (promoter region, 265 bp upstream of the start codon ATG of arsC2-like gene, arsC2-like and 236 bp downstream of the stop codon TTA of arsC2-like gene) was PCR amplified with primers ArsC2-like-F and ArsC2-like-R (Supplementary Table S2).
The above PCR products were ligated to the BamHI-XbaI site of plasmid pUC18, yielding plasmids pUC18-ars1, pUC18-ars2, pUC18-arsC1-like, and pUC18-arsC2-like. Then the plasmids were transferred to E. coli AW3110, yielding the recombinant E. coli AW3110-ars1, E. coli AW3110-ars2, E. coli AW3110-arsC1-like and E. coli AW3110-arsC2-like strains, respectively.
Strains, Plasmids, and Culture Conditions
The strains and plasmids used in this work are summarized in Supplementary Table S3. E. coli and Pantoea strains were grown in LB medium (per liter contains: 10 g tryptone, 5 g yeast, and 10 g NaCl) or LB plates (LB medium with w/v 1.5% agar) at 30°C. When appropriate, antibiotics were added at the following concentration: 100 μg/mL ampicillin. Resistance to As species was tested by plating serial dilutions of cultures of each strain onto agar plates containing filtered sodium arsenate (Na3AsO4).
Results
Genomic Features
To date, 26 species have been reported in genus Pantoea and strains of nine species (P. ananatis, P. agglomerans, P. stewartii, P. vagans, P. dispersa, P. septica, P. rodasii, P. rwandensis, and P. anthophila) have been sequenced3. To study the genetic traits and phylogenetic history of As resistance in genus Pantoea, 23 strains were chosen, containing two to three standard strains sequenced in each species and five unidentified strains (Supplementary Table S1). A summary of features for these 23 sequenced genomes is listed in Supplementary Table S1. The G+C contents of the 23 genomes range from 53.4 to 59.1. These genomes vary in size by approximately 1.6 mega-bases in average (ranging from 4.02 to 5.68 Mb) with coding sequence (CDS) numbers ranging from 3580 to 8894, indicating a substantial strain-to-strain variation.
Strain-Specific and Core Genes
To reveal the genomic features specific to each strain, we identified all orthologous pairs between the tested Pantoea genomes using PGAP. Our analysis of the total of 23 genomes revealed that a pan genome contains 48,207 putative protein-coding genes in the genus Pantoea. Out of these 48,207 genes, 10,896 (22.6%) were represented in the specific genomes of Pantoea spp., suggesting some frequency of horizontal gene acquisition from other taxa. The number of specific genes ranges from 131 to 1,285, with the smallest encoded by P. vagans C9-1 and the largest identified in P. agglomerans Tx10 (Supplementary Figure S1). The cluster of orthologous groups (COG) assignments reveal that a higher proportion of strain-specific genes in most of the strains can be assigned to the K (transcription), L (DNA replication), and M (cell wall/membrane/envelope biogenesis) categories (Supplementary Figure S2).
In contrast to the pan-genome, the core genome of Pantoea spp. contains 1,994 putative protein-coding genes, which represents 38.8–56.1% of the repertoire of protein coding genes of each strain, illustrating a small degree of genomic diversity in this group of bacteria (Supplementary Figure S1). The genomic analysis agrees with the fact that Pantoea strains are consistent in morphological and physiological appearance. Furthermore, the COG assignment results show that these core genes are in different functional categories (Supplementary Figure S3). In fact, the percentage of genes in each functional category remains rather similar (with an average divergence of 8.6%). This is consistent with an earlier report that larger prokaryotic genomes preferentially accumulate genes directly or indirectly involved in metabolism (Konstantinidis and Tiedje, 2004). These genes support a broader metabolic diversity, which, in turn, would improve the ecological success of Pantoea under more diverse environmental conditions.
Phylogenic Analyses
To associate the distribution of As resistance genes in Pantoea spp. with their phylogenetic affiliation, we constructed the phylogeny tree of the 23 Pantoea spp. based on 16S rRNA gene sequences using NJ methods rooted by Tatumella sp. NML 06-3099 (Supplementary Figure S9). This phylogenetic tree showed that the strains in the same species reported were grouped together except strain 848PVAG. At the same time, we constructed the phylogeny of the 23 genomes based on concatenation of the 100 core genes that are present as single copies in a genome using the ML method and rooted by Tatumella sp. NML 06-3099 (Figure 1). The phylogenetic trees, inferred using BI and NJ methods (Supplementary Figures S4, S5), were congruent with the ML phylogenetic tree. These trees show that some strains in different species reported were grouped together, such as, FF5 and 848PVAG, ZBG6, GB1, MP2, and Tx10. The phylogeny based on the 100 core genes that are present as single copies in a genome showed a good correlation with that of 16S rRNA gene sequences, except for three strains ZBG6, GB1, and 299R. These results suggested that there were mistakes in the classification of Pantoea spp.. Further identification of the phylogenetic status of these strains was carried out as follows.
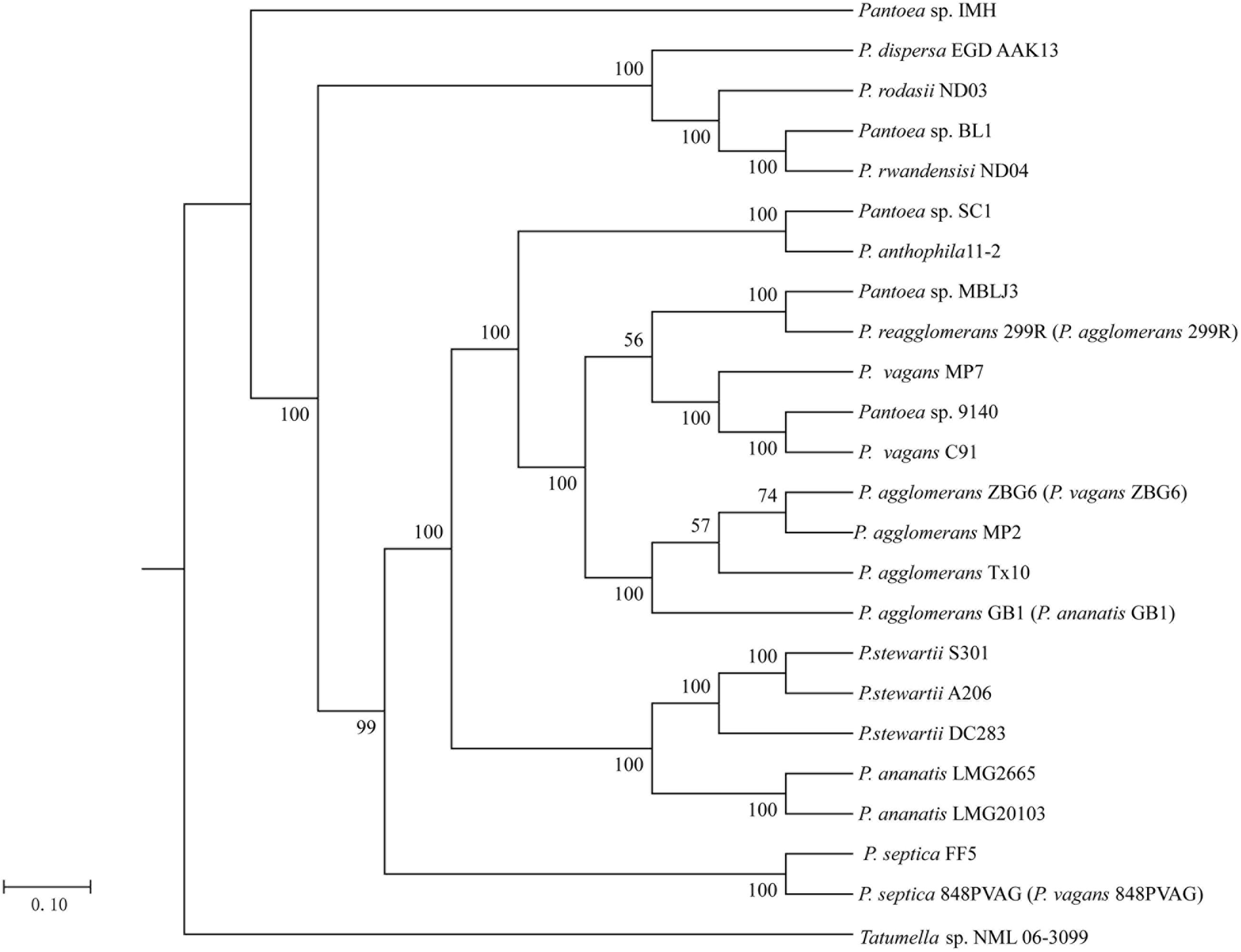
FIGURE 1. Phylogenetic relationship of 23 Pantoea strains. Maximum likelihood (ML) phylogenetic tree was constructed based on 100 single-copy core proteins shared by 23 genomes and an out-group Tatumella sp. NML 06-3099.
The information gained from the phylogenetic analysis provides an important depiction of the evolutionary relationship between different strains, but it does not translate directly into the overall similarity of the genomes, which is usually determined through the DNA-DNA hybridization (DDH). Herein, ANI approach was used to overcome the difficulty of conventional laboratory-based DDH in evaluating the genomic similarity of bacteria (Richter and Rosselló-Móra, 2009). The ANI results justified the conclusion of phylogenetic analysis. As shown in Figure 2, 23 strains were classified into 12 species based on their ANI ≥ 96%. For examples, LMG2665 and LMG20103 resulted in a higher ANI (99.3%), suggesting that they belong to the same species (P. ananatis). Strain 9140 and C91 resulted in a higher ANI (98.6%), suggesting that they belong to the same species (P. vagans). It was noteworthy that Panotea sp. IMH represented a novel species for the ANI ≥ 96% between IMH and other strains.
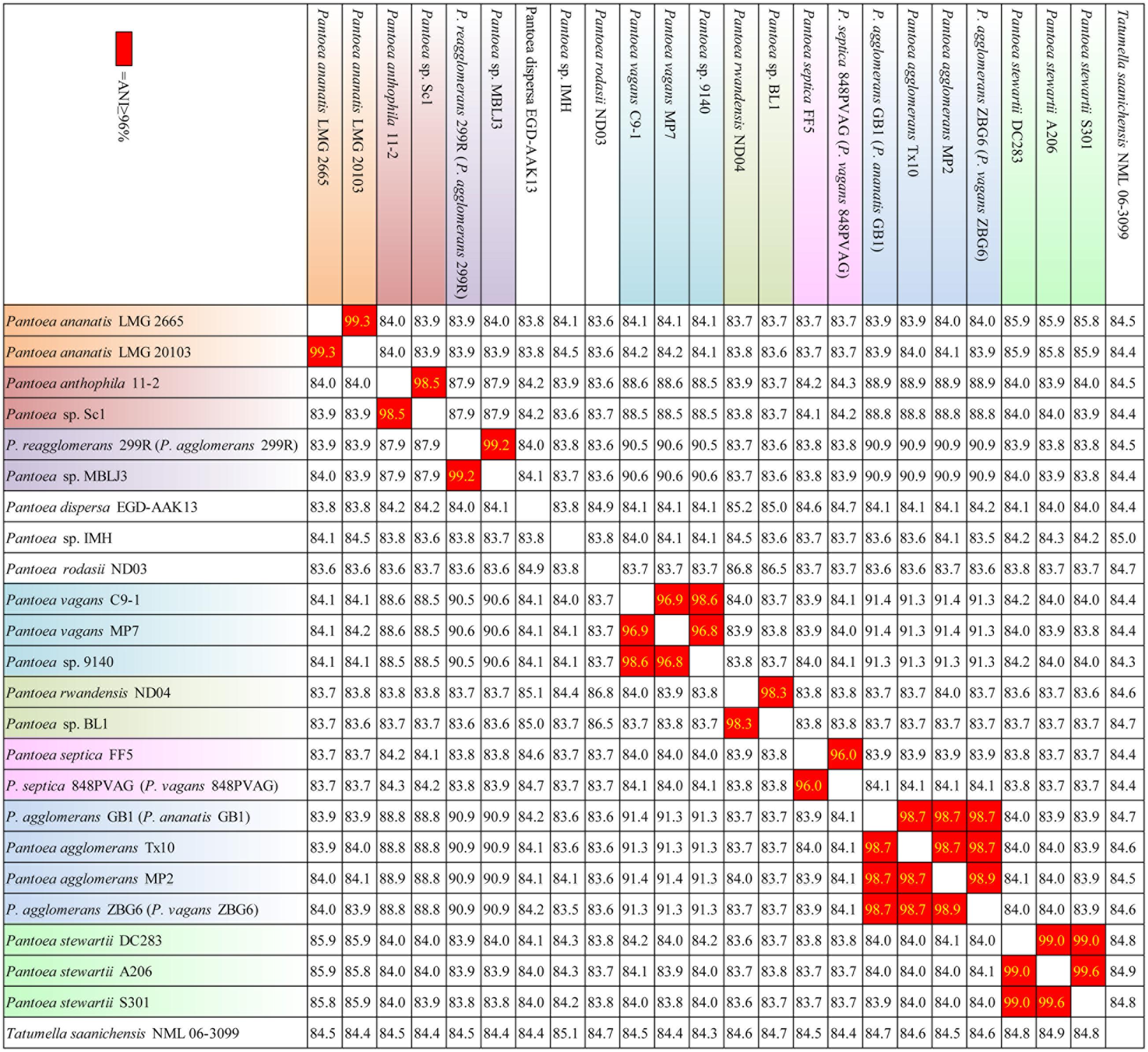
FIGURE 2. Average Nucleotide Identity (ANI) (%) based on whole genome alignments. ANI values are colored red according to historical species cutoff value (≥96%). Strains in one species are marked out the same color.
Strains MP2, Tx10, GB1, and ZBG6 which grouped together were identified as strains of P. agglomerans. Meanwhile, this result confirms the synonymy of P. FF5 and 848 PVAG (P. vagans), and suggests that 299R is not a member of species P. agglomerans. In agreement with the phylogenetic analysis, our ANI results indicate that there are mistakes in the classification of strain 299R, 848PVAG, GB1, and ZBG6. This mis-classification was also reported in other genus and generally corrected with the advance in technology (Goris et al., 2007). To associate the distribution of As-related genes with their phylogenetic affiliation, in this article below we renamed strain 299R to P. reagglomerans 299R (P. agglomerans 299R), 848PVAG to P. septica 848PVAG (P. vagans 848PVAG), GB1 to P. agglomerans GB1 (P. ananatis GB1), and ZBG6 to P. agglomerans ZBG6 (P. vagans ZBG6).
Distribution and Organization of As-Related Genes in Pantoea Genomes
Only As resistance genes (ars genes) including arsR, arsB, arsC, and arsH were detected in most Pantoea genomes (Supplementary Table S4 and Figure 3). The arsC gene encoding arsenate reductase is involved in the transformation of As(V) to As(III), which is then excreted by the As efflux pump ArsB encoded by the arsB gene. Nevertheless, aio, arr, and arsM were not found in Pantoea genomes, suggesting that cytoplasmic As(V) reduction and As(III) extrusion are the As resistance strategy used in genus Pantoea spp.. This mechanism benefits the bacteria itself, though it enhances the toxicity to the surrounding environment.
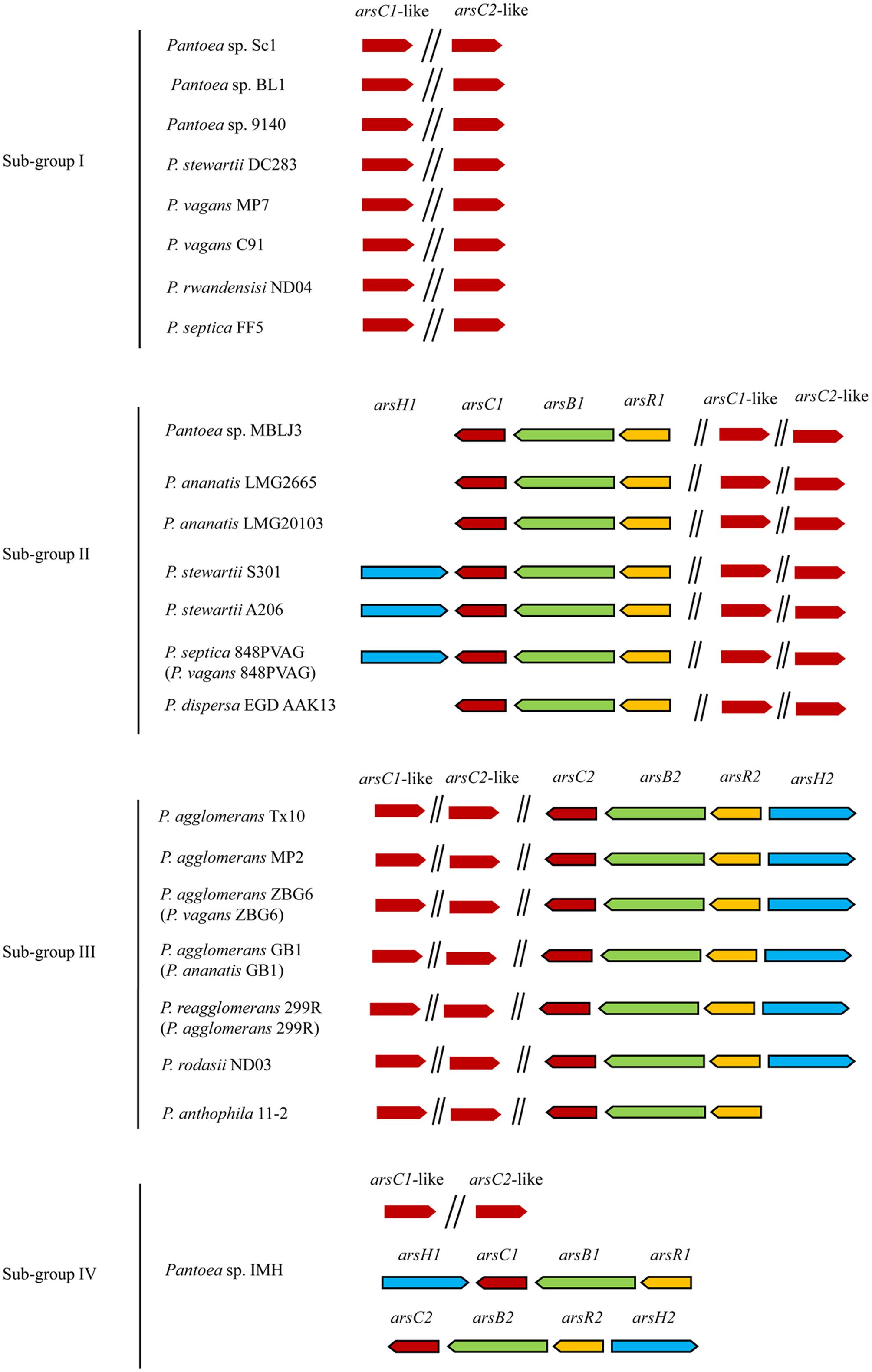
FIGURE 3. Distribution and organization of ars genes and arsC-like genes in 23 Pantoea strains. arsC, arsB, arsR, arsH, and arsC-like genes are marked with different colors. There are only arsC-like genes in Sub-group I, arsR1B1C1 or arsR1B1C1H1 in Sub-group II, arsR2B2C2 or arsR2B2C2H2 in Sub-group III, and both arsR1B1C1H1 and arsR2B2C2H2 in Sub-group IV.
The ars genes in a genome are prone to group together as ars clusters (arsRBC and arsRBCH). Although comparison of the COG assignments of 23 genomes revealed that the DNA sequences between homologous genes within these ars clusters are conserved, some variations exist in DNA sequences, which can be divided into two sub-groups (ars1 and ars2) (Figure 3). Unlike the two ars clusters in Pantoea sp. IMH, only one ars cluster, either ars1 or ars2, was observed in other strains (Supplementary Table S2 and Figure 3). The ars gene clusters generally exhibited more than 80% identity within each sub-group and about 54% identity between two sub-groups. Actually, the ars clusters were not detected in eight strains including Sc1, BL1, 9140, DC283, MP7, C91, ND04, and FF5. Moreover, two arsC-like genes with only 25% homology (arsC1-like and arsC2-like) were found in the 23 genomes. Based on the different ars genes distributions, the 23 strains were categorized into four sub-groups and discussed as follows. The overall distribution and organization of As resistance genes in 23 Pantoea strains are summarized in Figure 3.
Evolution and the Origin of ars Clusters
The distribution and organization of ars genes in Pantoea raise a question as to their evolution. The deviant G+C content is used as a detect method of HGT (Ochman et al., 2000; Xie et al., 2014). We detected the G+C content of ars clusters and their corresponding genomes. The results showed that the G+C contents of the ars1 clusters are higher than those of the genomes in Pantoea strains (56.3–57.8 vs. 53.4–54.7) except P. septica 848PVAG (P. vagans 848PVAG) and P. dispersa EGD-AAK13; the G+C contents of the ars2 clusters are lower than those of the genomes in Pantoea strains (50.6–52.4 vs. 53.7–58.8), showing variation of G+C content between clusters and the corresponding genomes. These results indicated that these ars clusters may be acquired in Pantoea strains by HGT (Supplementary Table S4 and Figure S6). To further elucidate the evolution of the ars gene clusters, we compared the chromosomal regions flanking the ars gene clusters among the 23 Pantoea strains and found that the genes in the upstream and downstream regions were conserved among strains of the same species (Figure 4). For example, the DNA polymerase V subunit UmuC gene and adenosine deaminase gene in the upstream and Cd(II)/Pb(II)-responsive transcriptional regulator gene and ATPase P gene in the downstream are conserved for the ars clusters in the strain Tx10, MP2, ZBG6, and GB1 within the species P. agglomerans strains. The same species strains share the same insertion sites, whereas the different species’ strains result in different insertion sites, suggesting that ars clusters may be acquired more than once.

FIGURE 4. Synteny of the chromosomal regions flanking the ars gene cluster among each species-group.
Interestingly, as shown in Figure 4, the flanking regions of the ars gene clusters in strain P. stewartii S301 and P. stewartii A206 were homologous to the corresponding regions of strain P. stewartii DC283; the same phenomenon was found in strain P. septica 848PVAG (P. vagans 848PVAG) and P. septica FF5, and strain P. anthophila 11-2 and Pantoea sp. Sc1. This result suggests that ars clusters may be lost in P. stewartii DC283, P. septica FF5, and Pantoea sp. Sc1.
To gain insights into the origin of ars genes clusters in Pantoea, a NJ phylogenetic tree was constructed based on the ArsRBC protein sequences. As shown in Figure 5, the strains possessing ars1 and ars2 clusters form separate groups. Notably, the phylogeny reveals that the ars1 and ars clusters of F. helveticus were sister groups, and ars2 grouped to ars clusters of S. marcescens and C. freundii. These results imply that the ars1 cluster may be acquired via HGT from F. helveticus, and ars2 from S. marcescens and C. freundii in early evolutionary history.
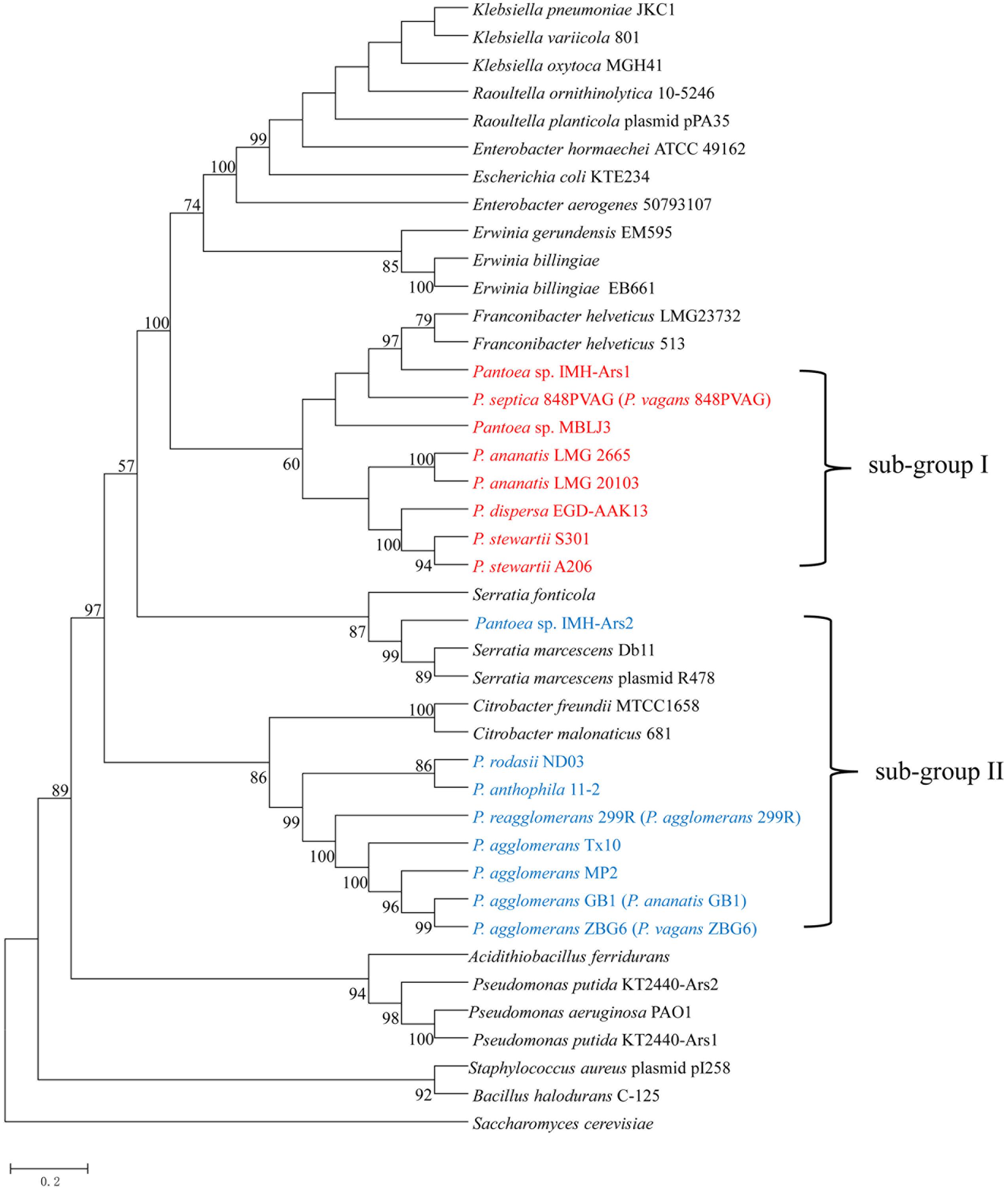
FIGURE 5. Neighbor joining phylogenetic tree of the ArsRBC protein sequences derived from Pantoea spp. strains and other representative species. A total of 1,000 bootstrap replicates was made, and bootstrap values are indicated at each node. ars1 cluster grouped together marked in red, ars2 cluster formed in another group marked in blue.
Two arsC-Like Genes in Pantoea
Our studies reveal that two arsC-like genes (arsC1-like and arsC2-like) are found in the 23 genomes with just 25% homology (Figure 3). Our phylogenetic analysis showed that the ArsC-like sequences formed distinct groups, which were clearly divergent from conventional arsenate reductase (Figure 6). It was reported that Cys-12, Arg-60, Arg-94, and Arg-107 were four conserved residues of the ArsC protein in the process of arsenic resistance (Gladysheva et al., 1996). Cys-12 was identified as a catalytic residue and was activated by nearby residues Arg-60, Arg-94, and Arg-107 (Martin et al., 2001). Alignment analysis of arsC and arsC-like genes shows that Cys-12 and Arg-94 residues were conserved, but residues Arg-107 and Arg-60 in two ArsC-like proteins were not conserved (Supplementary Figure S8). These results suggest that these two arsC-like genes are not involved in the As resistance.
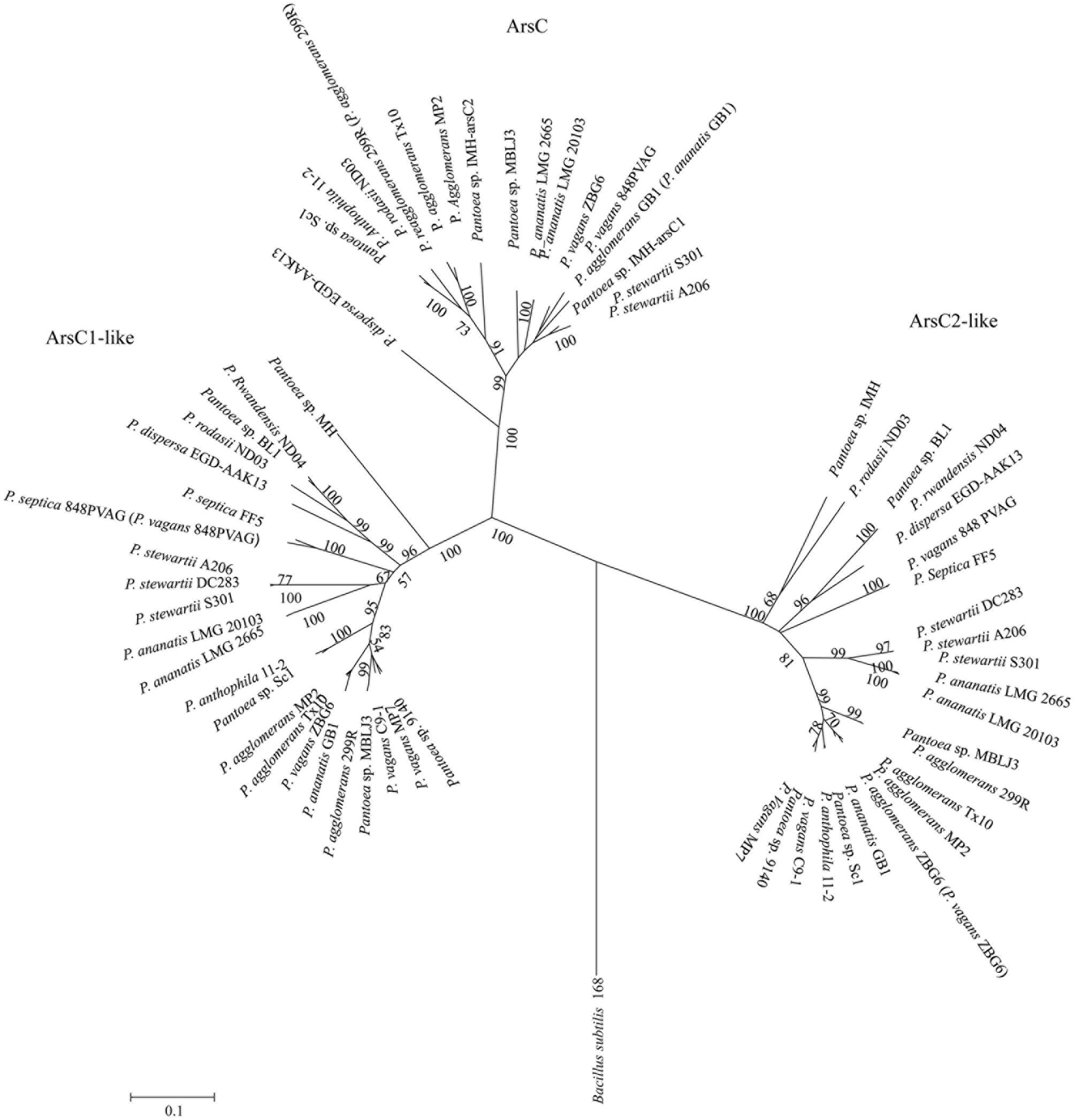
FIGURE 6. Neighbor-joining tree based on ArsC/ArsC-like proteins. ArsC/ArsC-like sequences were derived from 23 Pantoea strains and the representative microorganisms. ArsC sequence of Bacillus subtilis 168 was used as an out group.
To explore the evolution of these two arsC-like genes in Pantoea, molecular phylogenetic analysis, molecular conservation, and linear representation analysis were used (Rice and Lampson, 1995; Nelson et al., 1999; Brochier-Armanet and Forterre, 2006; Dagan et al., 2008). The comparative analysis showed that the two arsC-like genes are conserved in all of the 23 (Supplementary Figure S7). Phylogenetic analysis showed that arsC1-like and arsC2-like genes were grouped together, respectively (Figure 6). These results suggested that the two arsC-like genes evolved with a possible evolutionary scenario of that there is a common ancestor. Further, we compared the flanking regions of the two arsC-like genes. Interestingly, two genes in the upstream (the uracil phosphoribosyl transferase genes and uracil/xanthine transporter genes) and two genes in the downstream (sulfur reduction protein DsrE and GntR family transcriptional regulator genes) are conserved for arsC1-like genes. The two genes in the upstream (DNA-binding response regulator genes and multidrug efflux RND transporter permease genes) and two genes in the downstream (succinyl-diaminopimelate desuccinylase genes and membrane protein genes) are also conserved for arsC2-like genes (Supplementary Figure S7). This observation also suggests that arsC1-like and arsC2-like genes were the vertical genetic genes in the genus Pantoea. Possibility, they may have been the main As resistance contributors in early times and later had evolved with deviance.
Functional Analysis of ars Gene and arsC-Like Genes
To verify that the ars gene clusters are the contributors to the As resistance, the ars1 cluster with its promoter from P. stewartii S301, a representative strain with the ars1 cluster, and the ars2 cluster with its promoter from P. agglomerans Tx10, a representative strain with the ars2 cluster, were PCR amplified and then ligated into vector pUC18 and further transferred to E. coli AW3110 (without any As resistance genes). The growth of the yielded recombinant E. coli strains E. coli-ars1 and E. coli-ars2, was tested in 5 mM concentration As(V). As shown in Figure 7, both E. coli-ars1 and E. coli-ars2 survived in 5 mM As(V), and E. coli-ars1 grew better than E. coli-ars2. This result suggests that both the ars1 and ars2 clusters enabled E. coli AW3110 to resist As, and ars1 seemed to have a more effective As resistance capability than ars2.
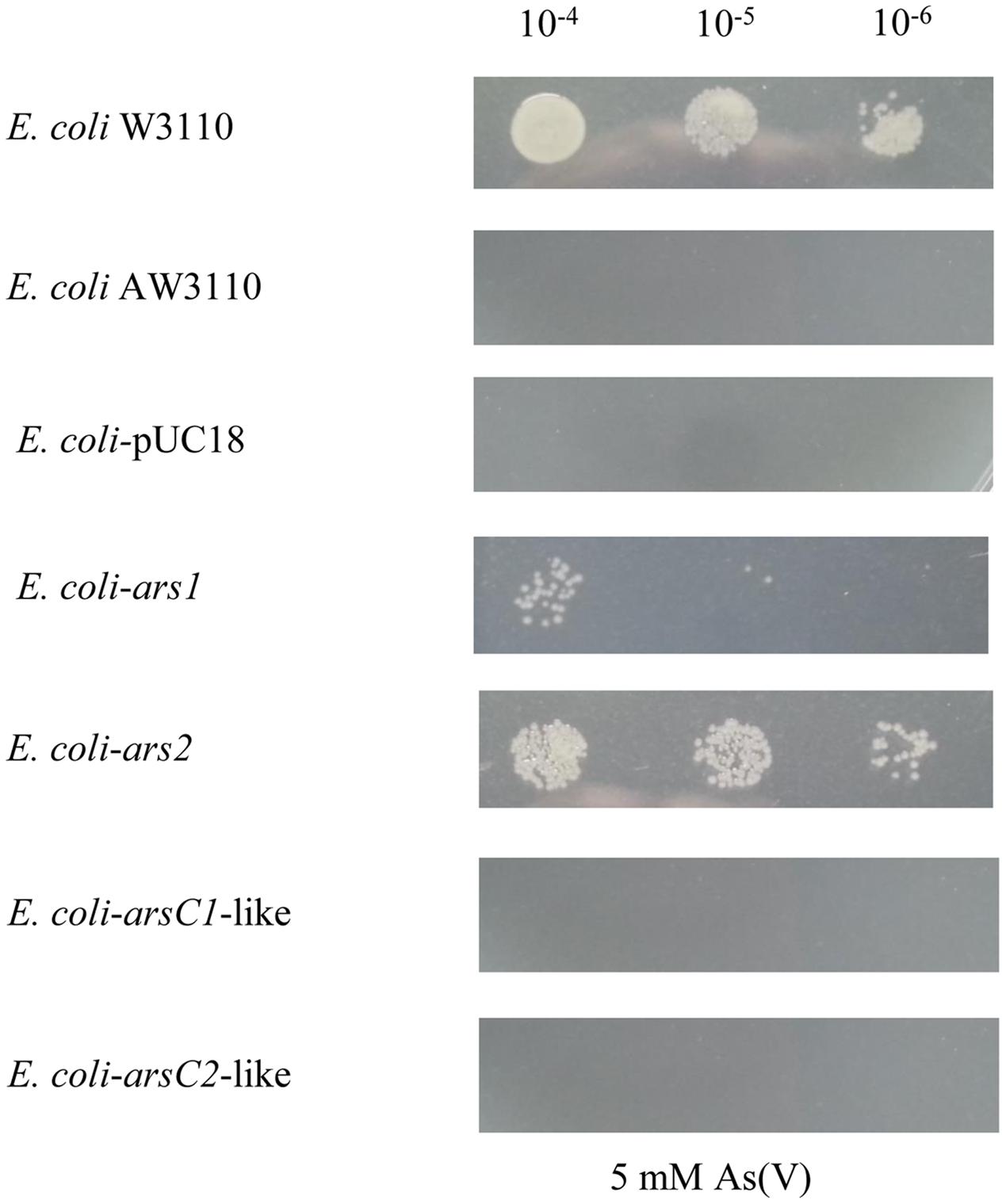
FIGURE 7. The heterologous expression of ars1, ars2, arsC1-like and arsC2-like in E. coli AW3110. The pUC18-ars1, pUC18-ars2, pUC18-arsC1-like and pUC18-arsC2-like plasmids (Supplementary Table S3) were transformed, along with the empty vector pUC18 in Escherichia coli AW3110. E. coli W3110 (with arsRBC cluster) as a control. Serial 5 μl dilutions of the strains were then plated on LB agar medium with 5 mM As(V). Pictures were taken after 48 h at 30°C.
To test the functions of arsC1-like and arsC2-like genes, the arsC1-like gene and arsC2-like gene with their promoters from P. stewartii DC283 were PCR amplified and then ligated into vector pUC18 and further transferred to E. coli AW3110. As shown in Figure 7, neither the arsC1-like nor arsC2-like gene enables E. coli AW3110 to resist As. In line with the alignment result, the function analysis demonstrates that these two arsC-like genes do not contribute to As resistance.
Discussion
Pantoea is a genus with 26 members identified by DDH, a gold standard for prokaryotic species identification. However, laboratory-based DDH results may be irreproducible, and vary depending on the reannealing temperature (Gevers et al., 2005). With the rapid development in technology and decline in sequencing cost, promising new measurements such as ANI are being developed to evaluate the genomic similarity of bacteria (Richter and Rosselló-Móra, 2009). In this study, we identified the 23 Pantoea spp. phylogenetic status using ANI, together with phylogenetic trees based on concatenated sequences of the 100 core genes (Figures 1, 2). Our results showed that strain 299R, 848PVAG, GB1, and ZBG6 were misnamed. We reclassified strain 299R to P. reagglomerans 299R (P. agglomerans 299R), 848PVAG to P. septica 848PVAG (P. vagans 848PVAG), GB1 to P. agglomerans GB1 (P. ananatis GB1), and ZBG6 to P. agglomerans ZBG6 (P. vagans ZBG6). Our study provided data from genus Pantoea with a complex and controversial taxonomy and demonstrated the accuracy of a bioinformatics approach, such as ANI, to identify new species and to correct erroneous identifications from previous studies.
A previous study suggested that the ars system is a widespread As resistance mechanism (Páezespino et al., 2009). Pantoea sp. IMH was found to resist As by means of ars clusters. arsRBC is located on the large universal Pantoea plasmids of four stains including P. agglomerans E325, P. agglomerans MP2, P. eucalyptiαB, and P. anthophila Sc1 (Maayer et al., 2012). This information leads to the hypothesis that plasmids may be involved in the evolution of As resistance mechanism by ars genes in Pantoea spp. However, there are untouched questions such as what are the mechanisms of the other Pantoea spp. and what is the evolutionary history of genetic elements involved in the As resistance? To answer these questions, we collected the genome sequences of 23 strains in nine species in NCBI (P. ananatis, P. agglomerans, P. stewartii, P. vagans, P. dispersa, P. septica, P. rodasii, P. rwandensis, and P. anthophila). The sequencing results provided us with mass genomic information to detect the presence and the locations of As-related genes in Pantoea spp. Our study for the first time systematically analyzed the As resistance genes and revealed the As resistance traits in genus Pantoea. Our research provided the definitive evidence that that As resistance strategy in Pantoea spp. only involved the detoxification mechanism through ars clusters, not the respiratory reduction mechanism through arr clusters. This detoxification strategy was obtained by HGT. This conclusion can likely to be extended to most bacteria. We speculate that evolutionarily ancient microbes were exposed to As surroundings on ancient earth (Oremland et al., 2009). To overcome the As-induced selection pressure, microbes evolved ars genes in their genomes for survival by HGT. Therefore, ars has very early origins and represents a widespread As resistance mechanism.
Two scattered arsC-like genes exist in each genome of the 23 Pantoea strains, but they exhibited no functional As resistance. It is rare for arsC-like genes to show no As resistance capabilities (Butcher et al., 2000; Saltikov and Newman, 2003). Compared to functional protein ArsC, residues Arg-107 and Arg-60 of ArsC-like protein were variant (Supplementary Figure S8). We speculate that in early times, the ancestor of Pantoea spp. evolved the arsC gene to resist As, but later evolved with deviance during adaption to As-free niches, and thus retained non-functional arsC-like genes in some genomes.
The ars genes are abundant and tend to organize in typical arsRBC cluster structures (Figure 3). Apart from these operons, arsRBCH operons are widely observed. In genus Pantoea, these kinds of structures were anticipated, for these strains descended from a recent common ancestor. Our study suggests that ars clusters may be acquired by HGT from F. helveticus, S. marcescens, and C. freundii strains. This is consistent with recent literature showing that bacterial As resistance and transformation was a trait acquired via HGT, driven by adaptation to habitats containing As (Cai et al., 2009; Villegas-Torres et al., 2011). Interestingly, ars clusters are absent in some strains, suggesting that some microbes may have lost their As resistance genes during adaption to As-free niches. In addition, the number of As resistance genes in strains isolated from As-rich environments is much higher than in strains from other environments (Macur et al., 2004; Sutton et al., 2009). Compared to the evolutionary pattern of ars operons (Rosen, 1999), the evolution of As resistance genes (ars clusters) in Pantoea spp. involves a mix of HGT and loss, providing insight into the complex evolutionary history of As resistance.
Author Contributions
LW and CJ conceived and designed the study. LW performed the laboratory work and data analysis. LW, JW, and CJ drafted the tables and figures, and prepared the main manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We acknowledge the financial support of the National Basic Research Program of China (2015CB932003), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB14020302), and the National Natural Science Foundation of China (41373123, 41425016, 41503094, and 21321004). We thank Yongguan Zhu for the strain E. coli AW3110.
Data Accessibility
The NCBI accession numbers of 23 draft genome sequences of Pantoea are listed in Supplementary Table S1.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fmicb.2017.00471/full#supplementary-material
Footnotes
- ^ http://www.bacterio.net/pantoea.html
- ^ http://tree.bio.ed.ac.uk/software/figtree/
- ^ http://www.ncbi.nlm.nih.gov/genome/?term=pantoea
References
Arsène-Ploetze, F., Koechler, S., Marchal, M., Coppée, J. Y., Chandler, M., Bonnefoy, V., et al. (2010). Structure, function, and evolution of the Thiomonas spp. genome. PLoS Genet. 6:e1000859. doi: 10.1371/journal.pgen.1000859
Brochier-Armanet, C., and Forterre, P. (2006). Widespread distribution of archaeal reverse gyrase in thermophilic bacteria suggests a complex history of vertical inheritance and lateral gene transfers. Archaea 2, 83–93.
Butcher, B. G., Deane, S. M., and Rawlings, D. E. (2000). The chromosomal arsenic resistance genes of Thiobacillus ferrooxidans have an unusual arrangement and confer increased arsenic and antimony resistance to Escherichia coli. Appl. Environ. Microbiol. 66, 1826–1833. doi: 10.1128/AEM.66.5.1826-1833.2000
Cai, L., Liu, G., Rensing, C., and Wang, G. (2009). Genes involved in arsenic transformation and resistance associated with different levels of arsenic-contaminated soils. BMC Microbiol. 9:4. doi: 10.1186/1471-2180-9-4
Colston, S. M., Fullmer, M. S., Beka, L., Lamy, B., Gogarten, J. P., and Graf, J. (2014). Bioinformatic genome comparisons for taxonomic and phylogenetic assignments using aeromonas as a test case. MBio 5:e2136-14. doi: 10.1128/mBio.02136-14
Dagan, T., Artzyrandrup, Y., and Martin, W. (2008). Modular networks and cumulative impact of lateral transfer in prokaryote genome evolution. Proc. Natl. Acad. Sci. U.S.A. 105, 10039–10044. doi: 10.1073/pnas.0800679105
Gavini, F., Mergaert, J., Bej, A., Mielcarek, C., Izard, D., Kersters, K., et al. (1989). Transfer of Enterobacter agglomerans (Beijerinck 1888) Ewing and Fife 1972 to Pantoea gen. nov. as Pantoea agglomerans comb. nov. and description of Pantoea dispersa sp. nov. Int. J. Syst. Evol. Microbiol. 39, 337–345.
Gevers, D., Cohan, F. M., Lawrence, J. G., Spratt, B. G., Coenye, T., Feil, E. J., et al. (2005). Opinion: re-evaluating prokaryotic species. Nat. Rev. Microbiol. 3, 733–739. doi: 10.1038/nrmicro1236
Ghosh, D., Bhadury, P., and Routh, J. (2014). Diversity of arsenite oxidizing bacterial communities in arsenic-rich deltaic aquifers in West Bengal, India. Front. Microbiol. 5:602. doi: 10.3389/fmicb.2014.00602
Gladysheva, T., Liu, J., and Rosen, B. P. (1996). His-8 lowers the pKa of the essential Cys-12 residue of the ArsC arsenate reductase of plasmid R773. J. Biol. Chem. 271, 33256–33260. doi: 10.1074/jbc.271.52.33256
Goris, J., Konstantinidis, K. T., Klappenbach, J. A., Coenye, T., Vandamme, P., and Tiedje, J. M. (2007). DNA-DNA hybridization values and their relationship to whole-genome sequence similarities. Int. J. Syst. Evol. Microbiol. 57, 81–91. doi: 10.1099/ijs.0.64483-0
Konstantinidis, K. T., and Tiedje, J. M. (2004). Trends between gene content and genome size in prokaryotic species with larger genomes. Proc. Natl. Acad. Sci. U.S.A. 101, 3160–3165. doi: 10.1073/pnas.0308653100
Levin, D., and Tal, A. (2003). Arsenite oxidase aox genes from a metal-resistant beta-proteobacterium. J. Bacteriol. 185, 135–141. doi: 10.1186/1471-2164-11-709
Maayer, P. D., Chan, W. Y., Blom, J., Venter, S. N., Duffy, B., Smits, T. H. M., et al. (2012). The large universal Pantoea plasmid LPP-1 plays a major role in biological and ecological diversification. BMC Genomics 13:625. doi: 10.1186/1471-2164-13-625
Macur, R. E., Jackson, C. R., Botero, L. M., Mcdermott, T. R., and Inskeep, W. P. (2004). Bacterial populations associated with the oxidation and reduction of arsenic in an unsaturated soil. Environ. Sci. Technol. 38, 104–111.
Martin, P., DeMel, S., Shi, J., Gladysheva, T., Gatti, D. L., Rosen, B. P., et al. (2001). Insights into the structure, solvation, and mechanism of ArsC arsenate reductase, a novel arsenic detoxification enzyme. Structure 9, 1071–1081. doi: 10.1016/S0969-2126(01)00672-4
Meng, M., Sun, W. Q., Geelhaar, L. A., Kumar, G., Patel, A. R., Payne, G. F., et al. (1995). Denitration of glycerol trinitrate by resting cells and cell extracts of Bacillus thuringiensis/cereus and Enterobacter agglomerans. Appl. Environ. Microbiol. 61, 2548–2553.
Nelson, K. E., Clayton, R. A., Gill, S. R., Gwinn, M. L., Dodson, R. J., Haft, D. H., et al. (1999). Evidence for lateral gene transfer between Archaea and Bacteria from genome sequence of Thermotoga maritima. Nature 399, 323–329. doi: 10.1038/20601
Ochman, H., Lawrence, J. G., and Groisman, E. A. (2000). Lateral gene transfer and the nature of bacterial innovation. Nature 405, 299–304. doi: 10.1038/35012500
Oremland, R. S., Saltikov, C. W., Wolfe-Simon, F., and Stolz, J. F. (2009). Arsenic in the evolution of earth and extraterrestrial ecosystems. Geomicrobiol. J. 26, 522–536. doi: 10.1080/01490450903102525
Páezespino, D., Tamames, J., De, L. V., and Cánovas, D. (2009). Microbial responses to environmental arsenic. Biometals 22, 117–130. doi: 10.1007/s10534-008-9195-y
Qin, J., Rosen, B. P., Zhang, Y., Wang, G., Franke, S., and Rensing, C. (2006). Arsenic detoxification and evolution of trimethylarsine gas by a microbial arsenite S-adenosylmethionine methyltransferase. Proc. Natl. Acad. Sci. U.S.A. 103, 2075–2080. doi: 10.1073/pnas.0506836103
Rezzonico, F., Smits, T. H., Montesinos, E., Frey, J. E., and Duffy, B. (2009). Genotypic comparison of Pantoea agglomerans plant and clinical strains. BMC Microbiol. 9:204. doi: 10.1186/1471-2180-9-204
Rice, P., Longden, I., and Bleasby, A. (2000). EMBOSS: the european molecular biology open software suite. Trends Genet. 16, 276–277. doi: 10.1016/S0168-9525(00)02024-2
Rice, S. A., and Lampson, B. C. (1995). Phylogenetic comparison of retron elements among the myxobacteria: evidence for vertical inheritance. J. Bacteriol. 177, 37–45. doi: 10.1128/jb.177.1.37-45.1995
Richter, M., and Rosselló-Móra, R. (2009). Shifting the genomic gold standard for the prokaryotic species definition. Proc. Natl. Acad. Sci. U.S.A. 106, 19126–19131. doi: 10.1073/pnas.0906412106
Ronquist, F., Teslenko, M., van, der Mark P., Ayres, D. L., Darling, A., Höhna, S., et al. (2012). MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 61, 539–542. doi: 10.1093/sysbio/sys029
Saltikov, C. W., and Newman, D. K. (2003). Genetic identification of a respiratory arsenate reductase. Proc. Natl. Acad. Sci. U.S.A. 100, 10983–10988. doi: 10.1073/pnas.1834303100
Shimodaira, H., and Hasegawa, M. (2001). CONSEL: for assessing the confidence of phylogenetic tree selection. Bioinformatics 17, 1246–1247. doi: 10.1093/bioinformatics/17.12.1246
Smith, A. H., Lopipero, P. A., Bates, M. N., and Steinmaus, C. M. (2002). Public health. Arsenic epidemiology and drinking water standards. Science 296, 2145–2146. doi: 10.1126/science.1072896
Sutton, N. B., van, der Kraan G. M., van, Loosdrecht M. C., Muyzer, G., Bruining, J., and Schotting, R. J. (2009). Characterization of geochemical constituents and bacterial populations associated with As mobilization in deep and shallow tube wells in Bangladesh. Water Res. 43, 1720–1730. doi: 10.1016/j.watres.2009.01.006
Tamura, K. (2011). MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 28, 2731–2739. doi: 10.1093/molbev/msr121
Thompson, J. D., Gibson, T. J., and Higgins, D. G. (2002). Multiple sequence alignment using ClustalW and ClustalX. Curr. Protoc. Bioinformatics Chater 2:Unit 2.3. doi: 10.1002/0471250953.bi0203s00
Tian, H., and Jing, C. (2014). Genome sequence of the aerobic arsenate-reducing bacterium Pantoea sp. strain IMH. Genome Announc. 2:e267-14. doi: 10.1128/genomeA.00267-14
Villegas-Torres, M. F., Bedoya-Reina, O. C., Salazar, C., Vives-Florez, M. J., and Dussan, J. (2011). Horizontal arsC gene transfer among microorganisms isolated from arsenic polluted soil. Int. Biodeter. Biodegradation 65, 147–152. doi: 10.1016/j.ibiod.2010.10.007
Wang, L., Zhuang, X., Zhuang, G., and Jing, C. (2016). Arsenic resistance strategy in Pantoea sp. IMH: organization, function and evolution of ars genes. Sci. Rep. 6:39195. doi: 10.1038/srep39195
Wu, Q., Du, J., Zhuang, G., and Jing, C. (2013). Bacillus sp. SXB and Pantoea sp. IMH, aerobic As(V)-reducing bacteria isolated from arsenic-contaminated soil. J. Appl. Microbiol. 114, 713–721. doi: 10.1111/jam.12093
Xie, J. B., Du, Z., Bai, L., Tian, C., Zhang, Y., Xie, J. Y., et al. (2014). Comparative genomic analysis of N2-fixing and non-N2-fixing Paenibacillus spp.: organization, evolution and expression of the nitrogen fixation genes. PLoS Genet. 10:e1004231. doi: 10.1371/journal.pgen.1004231
Xu, C., Zhou, T., Kuroda, M., and Rosen, B. P. (1998). Metalloid resistance mechanisms in prokaryotes. J. Biochem. 123, 16–23.
Zhang, L., and Birch, R. G. (1997). The gene for albicidin detoxification from Pantoea dispersa encodes an esterase and attenuates pathogenicity of Xanthomonas albilineans to sugarcane. Proc. Natl. Acad. Sci. U.S.A. 94, 9984–9989.
Zhao, C., Zhang, Y., Chan, Z., Chen, S., and Yang, S. (2015). Insights into arsenic multi-operons expression and resistance mechanisms in Rhodopseudomonas palustris CGA009. Front. Microbiol. 6:986. doi: 10.3389/fmicb.2015.00986
Keywords: comparative genomic, arsenic, Pantoea spp., arsenic resistance, ars genes
Citation: Wang L, Wang J and Jing C (2017) Comparative Genomic Analysis Reveals Organization, Function and Evolution of ars Genes in Pantoea spp. Front. Microbiol. 8:471. doi: 10.3389/fmicb.2017.00471
Received: 04 November 2016; Accepted: 07 March 2017;
Published: 21 March 2017.
Edited by:
Manuel Espinosa, Consejo Superior de Investigaciones Científicas (CSIC), SpainReviewed by:
Yunyoung Kwak, Kyungpook National University, South KoreaLukasz Drewniak, University of Warsaw, Poland
Ji-Hoon Lee, Chonbuk National University, South Korea
Copyright © 2017 Wang, Wang and Jing. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chuanyong Jing, Y3lqaW5nQHJjZWVzLmFjLmNu
 Liying Wang
Liying Wang Jin Wang
Jin Wang Chuanyong Jing
Chuanyong Jing