- 1Division of Comparative Pathology, Department of Pathology and Laboratory Medicine, Miller School of Medicine, University of Miami, Miami, FL, USA
- 2Service de Parasitologie—Mycologie—Médecine tropicale, Centre Hospitalier Universitaire de Tours, Tours, France
- 3Centre d'Etude des Pathologies Respiratoires (CEPR) Institut National de la Santé et de la Recherche Médicale U1100/Équipe 3, Université François-Rabelais, Tours, France
Invasive aspergillosis has been studied in laboratory by the means of plethora of distinct animal models. They were developed to address pathophysiology, therapy, diagnosis, or miscellaneous other concerns associated. However, there are great discrepancies regarding all the experimental variables of animal models, and a thorough focus on them is needed. This systematic review completed a comprehensive bibliographic analysis specifically-based on the technical features of rodent models infected with Aspergillus fumigatus. Out the 800 articles reviewed, it was shown that mice remained the preferred model (85.8% of the referenced reports), above rats (10.8%), and guinea pigs (3.8%). Three quarters of the models involved immunocompromised status, mainly by steroids (44.4%) and/or alkylating drugs (42.9%), but only 27.7% were reported to receive antibiotic prophylaxis to prevent from bacterial infection. Injection of spores (30.0%) and inhalation/deposition into respiratory airways (66.9%) were the most used routes for experimental inoculation. Overall, more than 230 distinct A. fumigatus strains were used in models. Of all the published studies, 18.4% did not mention usage of any diagnostic tool, like histopathology or mycological culture, to control correct implementation of the disease and to measure outcome. In light of these findings, a consensus discussion should be engaged to establish a minimum standardization, although this may not be consistently suitable for addressing all the specific aspects of invasive aspergillosis.
Introduction
Aspergillosis is an airborne fungal infection due to ubiquitous molds belonging to the genus Aspergillus. In human medicine, Aspergillus fumigatus is the main species involved in aspergillosis with isolation in more than 80% of the clinical samples with positive culture, regardless the context (Desoubeaux et al., 2014a). A. fumigatus is present in the environment, especially in air, water, plants, and soil. When its spores are inhaled, it may be responsible for a wide-range of distinct clinical entities, but invasive aspergillosis—which is primarily reported in immunocompromised individuals—remains the most feared because of its high mortality rates ranging from 30 to 100% (Lortholary et al., 2011; Bitar et al., 2014).
As both basic and clinical knowledge about invasive aspergillosis is limited, laboratory models of the disease are needed. In spite of recent major advances (Sable et al., 2008; Brown, 2011; Steele and Wormley, 2012; Wüthrich et al., 2012; Drew et al., 2013; Lanternier et al., 2013), there are still many concerns to be addressed: for example, why a particular strain is more virulent than another (Becker et al., 2006)? How to prevent a contamination? Which route of drug administration to be privileged to cure the infection (Becker et al., 2002b)? And why a diagnostic tool is better than another one in such context (Becker et al., 2000, 2002a)? All these scenarii are very complex and for such purposes, development of animal models seem more valuable research tools than in vitro experiments, especially because they span the gap between the bench and the clinic bed. Theoretically, animal models mimic, as closely as possible, the clinical course and the symptoms of the disease as observed in human patients. Also they are assumed to be more easily repeatable, less expensive, and potentially more readily and quickly provide reliable scientific responses than clinical trials. Unfortunately for animal models studying invasive aspergillosis (Mahajan et al., 1978; Ghori and Edgar, 1979; Chaudhary and Singh, 1983; Chaudhary et al., 1988; Chilvers et al., 1989; Andriole et al., 1992; Kurtz et al., 1995; Leenders et al., 1996; Richard et al., 1996; Cicogna et al., 1997; Kirkpatrick et al., 2000b; Clemons and Stevens, 2005; Gavaldà et al., 2005; Lewis and Wiederhold, 2005; Patterson, 2005; Chandenier et al., 2009), heterogeneity has always been great regarding their technical variables, like the species or strains to be used, the animal sex and weight, the immunosuppressive regimen, the route of experimental infection, the fungal inoculum size, and the methods to assess fungal burden (Hohl, 2014). Thus, it currently does not exist any consensus for a unique animal model. However, one can notice that rodents have been mostly used so far, because they are of small size, inexpensive, easy-to-handle, and the ready availability of reagents and methods (Andriole et al., 1992; Clemons and Stevens, 2005; Lewis and Wiederhold, 2005; Patterson, 2005; Paulussen et al., 2014).
Therefore, it is now critical for animal models to be well-defined (Clemons and Stevens, 2005), and efforts to choose the best one(s) are required before a possible standardization. For such a purpose, we decided to complete a comprehensive overview of all the published reports that dealt with models of invasive aspergillosis. Within the text, and in order to perform a personal criticizing analysis, we sometimes subjectively placed emphasis on some studies that were thought to be interesting for providing specific and relevant information. To circumvent confounding bias, we restricted our study to A. fumigatus infection in rodent species. We took this opportunity to address most of the current pending issues. They applied to harmonization of the technical features and experimental settings, and to the following questions: what these assays are used for, how the results derived from them should be interpreted, and what philosophy or ethics should be considered.
Materials and Methods
Search Criteria
A systematic literature review was performed using a rigorous search strategy in the PubMed database for English language literature published up to October 2016, based on the following MeSH terms: [(“Aspergillus fumigatus”[Mesh]) OR (((“Aspergillosis”[Mesh]) OR (“Invasive Pulmonary Aspergillosis”[Mesh]) OR (“Pulmonary Aspergillosis”[Mesh])) NOT (“Aspergillosis, Allergic Bronchopulmonary”[Mesh])) AND ((“Models, Animal”[Mesh]) OR ((“Rodentia”[Mesh]) OR (“Mice”[Mesh]) OR (“Rats”[Mesh]) OR (“Guinea Pigs”[Mesh])) OR (“Cricetinae”[Mesh])) NOT (“Rabbits”[Mesh]) NOT (“Birds”[Mesh])]. Then, the authors exhaustively reviewed the retained articles. For each, they thoroughly focused on the pivotal experimental parameters and the major technical features that are assumed to likely influence the results (Schmidt, 2002; Clemons and Stevens, 2005; Lewis and Wiederhold, 2005; Patterson, 2005; Capilla et al., 2007): rodent species and strains as well as their weight and sex, the immunosuppressive regimen they underwent, the A. fumigatus strain(s) and the fungal inoculum used for the experimental challenge, the route of inoculation, the clinical, and biological parameters to follow up to assert correct implementation of the disease and its monitoring (Figure 1).
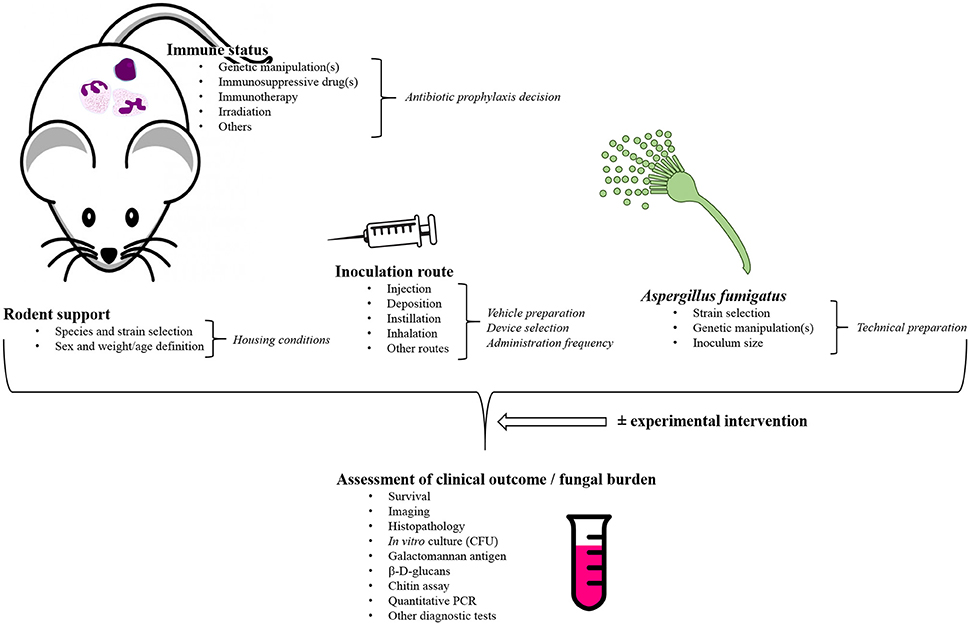
Figure 1. General overview of the pivotal study parameters that require to be considered when developing a rodent model of invasive aspergillosis caused by Aspergillus fumigatus. This scheme is probably not exhaustive, but it outlines the major variables that are mostly thought to be critical for model development in laboratories. CFU, Colony-forming unit(s); PCR, Polymerase chain reaction.
Analysis
Statistical analyses were performed using XLStat v.2014.6.04® software (Addinsoft, Paris, France). The α-risk was adjusted at 0.05.
Results
Number of Publications and Addressed Topics
Our electronic search about rodent models of invasive aspergillosis due to A. fumigatus retrieved 1,435 publications. Out of them, 91 were excluded since not written in English. Sixteen were not included because not accessible. After thorough reviewing, a total of 800 articles were finally retained for complete analysis (Figure 2). The first paper about rodent model of aspergillosis was published in 1967 (Ford and Friedman, 1967). More than three quarters have been written after the year 2000 (Figure 3).
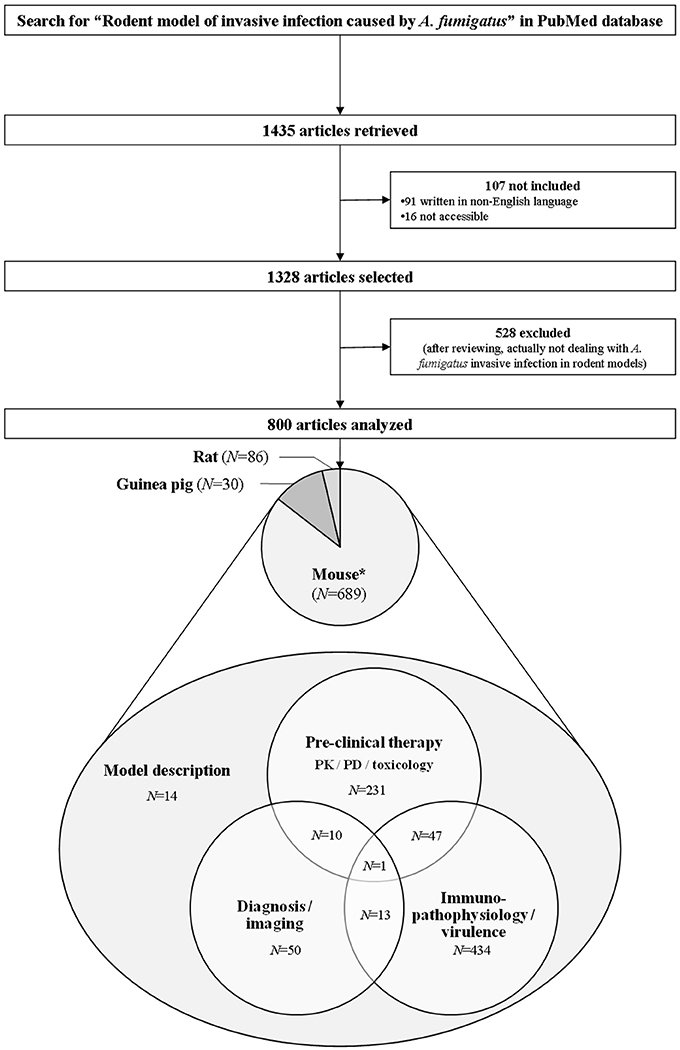
Figure 2. Flow chart of the bibliometric study. The research was completed in PubMed up to October 2016 according to the criteria reported in the Section Materials and Methods. Scientific reports, oral communications, and posters were not addressed in this study. N, Number; PD, Pharmacodynamics; PK, Pharmacokinetics. *Five articles reported the use of several rodent species at a time: two papers with mice plus rats simultaneously, and three with mice plus guinea pigs.
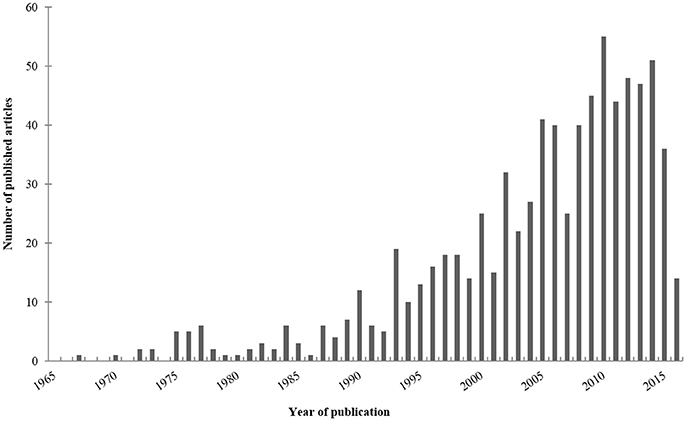
Figure 3. Published articles per year. For this bar chart, have been only taken into account the articles about rodent models of invasive aspergillosis due to Aspergillus fumigatus retrieved in PubMed up to October 2016, according to the criteria reported in the Section Material and Methods.
Key objectives of rodent models of invasive aspergillosis and their major topics are summarized in Table 1. A large majority of all the analyzed articles were dedicated to immunopathology of invasive aspergillosis and study of the fungal virulence, e.g., disease transmission, innate and acquired host-response, genes and proteins involved in fungal invasion, susceptibility to infection: 61.9% addressed this topic vs. 36.1 and 9.3% which were rather devoted to pre-clinical therapy [pharmacology/pharmacokinetics/toxicology, and also several vaccine assays (Clemons et al., 2014) and role of surgery (Habicht et al., 2002)], and to diagnosis/imaging approaches (Yang et al., 2009), respectively. Of note, the analyzed articles frequently covered several research fields at a time: for instance, 10 manuscripts dealt with both diagnosis/imaging and pre-clinical therapy simultaneously, 13 overlapped both diagnosis/imaging and immune-pathophysiology/virulence, and 47 addressed therapeutic and immuno-pathophysiology/virulence concerns within the same studies (Supplementary Material 1). Only a few were purely descriptive, and were thus intended to describe a new model of aspergillosis but with no mention of neither therapeutic assays associated, nor assessment of diagnostic/imaging tests nor immune-pathophysiological studies (Walzl et al., 1987; Dixon et al., 1989; Jensen and Hau, 1990a,b; Jensen and Schønheyder, 1993; Nawada et al., 1996; Yonezawa et al., 2000; Chiller et al., 2002; Sheppard et al., 2004; Steinbach et al., 2004; Zimmerli et al., 2007; Chandenier et al., 2009; Desoubeaux and Chandenier, 2012; Herbst et al., 2013; Leleu et al., 2013a; Zhang et al., 2013; Alcazar-Fuoli et al., 2015). Some of them reported the benefit of new devices to induce the experimental disease (Steinbach et al., 2004; Chandenier et al., 2009; Desoubeaux and Chandenier, 2012; Leleu et al., 2013a), others were focused on the experimental description of particular aspergillosis forms like cerebral aspergillosis (Chiller et al., 2002; Zimmerli et al., 2007) or invasive fungal rhino-sinusitis (Zhang et al., 2013). A few articles described models that offered the opportunity to study aspergillosis in very specific context, like solid-organ transplantation (Herbst et al., 2013) or during bacterial superinfection (Yonezawa et al., 2000). Interestingly, Jensen et al. addressed the clinical consequences for the fetus when the mouse mother was infected with A. fumigatus during pregnancy (Jensen and Hau, 1990a,b).

Table 1. Main general objectives for rodent models of invasive aspergillosis due to Aspergillus fumigatus.
General Description of the Various Rodent Models: Species and Strains, Weight and Sex
Overall, high variability was noticed regarding the rodent species (Figure 1, Table 2). Mice were used in 86.1% of the selected works, vs. 10.8 and 3.8% for rats and guinea pigs, respectively. Sometimes two of these species were tested concurrently in the same study (Reichard et al., 1997; Odds et al., 1998; Niwano et al., 1999; Hanazawa et al., 2000; Dufresne et al., 2012), like Odds et al. that reported evaluation of possible correlation between in vitro antifungal susceptibilities and treatment outcomes in both mice and guinea pigs (Odds et al., 1998), or Dufresne et al. who established that point-of-care diagnosis-based on urinary galactomannan-(GM)-like antigens detection is feasible in both mice and guinea pigs (Dufresne et al., 2012). To date, hamsters have not been tested as a model of invasive aspergillosis.
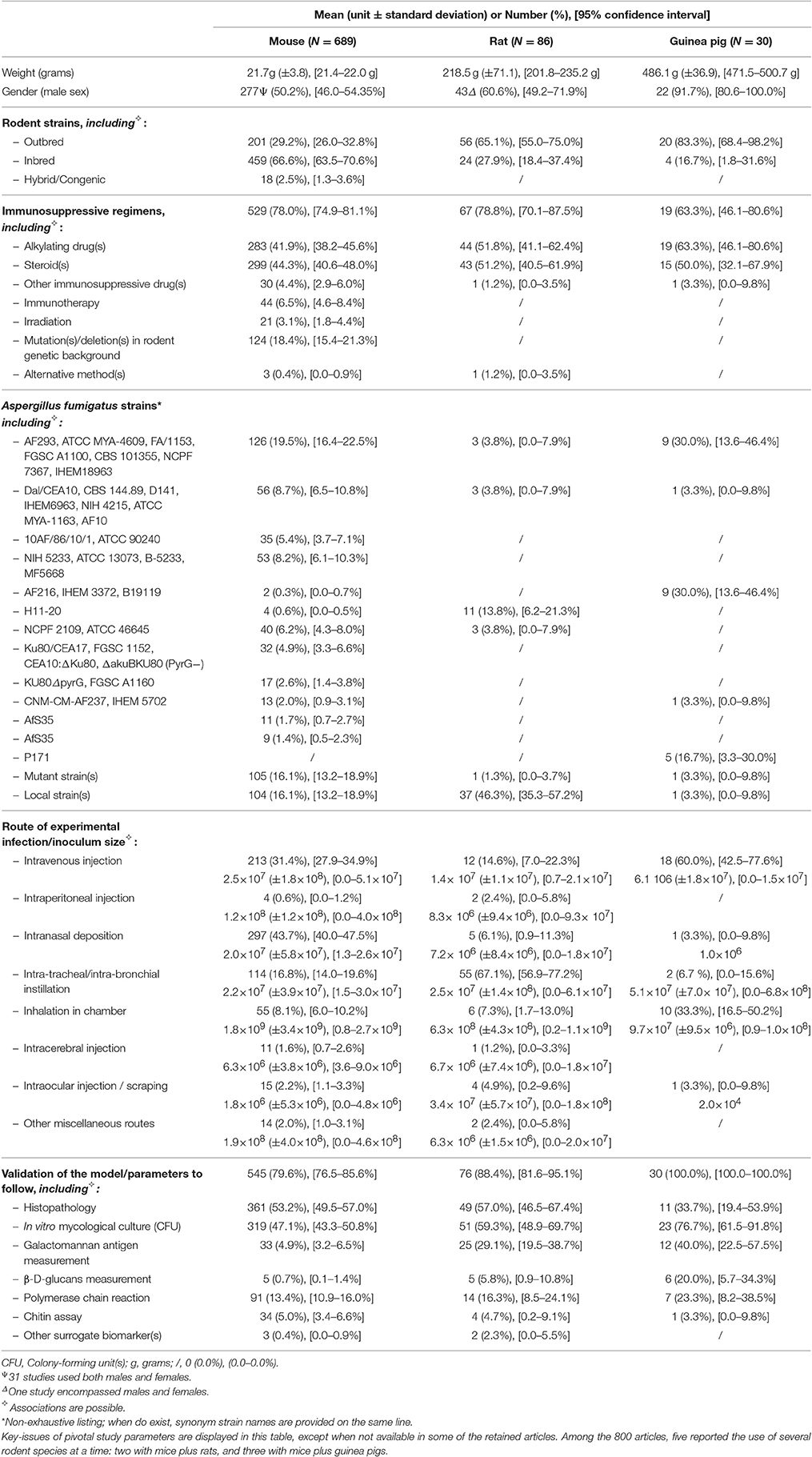
Table 2. Overall description of all the published rodent models of invasive aspergillosis due to Aspergillus fumigatus.
There were significant differences regarding the body weight of the rodents at time of experimental inoculation. For example, weight ranged from 9 g for very young mice (Li et al., 2014) to around 45 g, when they lived up to 42 months (Khosravi et al., 2012). For rats, it spanned from 26.5 g for 11 day old-pups (Zimmerli et al., 2007) to almost 400 g for oldest animals (Sivak et al., 2004a,b; Risovic et al., 2007; Wasan et al., 2007, 2009). Weight for guinea pigs was more homogeneous: 486.1 g ± 36.9. While animal sex was not specified in 152 articles, males were reported to be used in 42.6% of all the selected papers, and females in 41.9%. Both sexes were used without differentiation in 32 works (Supplementary Material 1).
Inbred rodents were more frequently used than outbred, 60.5 vs. 34.5% respectively. Among inbred mice, BALB/c, C57BL/6, and ICR/HaJ were reported in 209, 161, and 55 articles, respectively (Supplementary Material 2), i.e., 30.3, 23.5, and 8.2% of all the experimental studies using mice. When addressing chronic granulomatous disease, C57BL/6 strain was used to induce mutation in gp91phox or gp47phox genes in order to generate deficiency of the oxidative burst in phagocytic cells. A lot of other inbred mouse strains were only sporadically employed, like DBA/2J, CF-1, albino DDY—mainly in Japanese facilities—, or 129/Sv mice, these latter being also particularly useful for production of targeted mutations (Supplementary Material 2). Regarding the outbred strains, CD-1 and albino Swiss Webster mice were the most used, in 122 and 49 papers (Supplementary Material 2), i.e., 17.9 and 7.0% of the reported experiments in this rodent species. Outbred Swiss OF1 and NMRI mice were less employed (Supplementary Material 2), this latter being mostly tested in pharmacology and toxicology studies. Hybrid and congenic mice were generated in 2.5% of the labs using mouse species as animal models of invasive aspergillosis by mating two inbred strains and backcrossing their descendants over several generations. For instance, BALB/c mice were coupled with DBA/2 to get CD2F1 hybrids, or to generate mutation in order to mimic chronic granulomatous disease in B6.129S2 strains (Supplementary Material 2).
For studies with rats and guinea pigs, outbred strains were more often used than inbred ones: Sprague-Dawley represented 51.2% of the reports dealing with rats, while Pirbright white Dunkin-Hartley were associated with 76.7% of assays in guinea pig (Supplementary Material 2). These two strains were judged as excellent multipurpose models for safety and efficacy testing, as were also outbred albino Wistar and albino-CD rats that were tested in 14 and 1 works, respectively. For inbred rats, RP-strain albino, Lewis, Dark Agouti, and albino Oxford strains were used in 15, 3, 3, and 2 articles, respectively (Supplementary Material 2). The last three ones were considered as amenable supports for addressing immunology and inflammation response.
Selection of the Immunosuppressive Regimen
Induction of immunosuppression in rodents was reported in 617 publications, i.e., 78.0, 78.8, and 63.3% of articles that included mice, rats, and guinea pigs, respectively (Table 2). It was not the case for some very specific models, especially those for which experimental infection was achieved by intravenous (IV) route (models based on IV route represented 79.5% of the works with no immunosuppressive regimen), or when only local course of invasive aspergillosis was expected like ocular infection (Jie et al., 2009; Zhong et al., 2009, 2016; Ren et al., 2010; Guo et al., 2012; Gresnigt et al., 2014; Taylor et al., 2014a; Jiang et al., 2015, 2016; Li et al., 2015; Xu et al., 2015; Zhao et al., 2015).
Steroids were used in 44.4% articles, especially by the means of subcutaneous (sc) injections of cortisone or triamcinolone acetonide. Dexamethasone was less employed (Baisakh et al., 1975; Bartroli et al., 1998; Clemons et al., 2000b; Ullmann et al., 2007; Zimmerli et al., 2007; Morisse et al., 2012, 2013), sometimes given in drinking water (Meulbroek et al., 2003; García et al., 2006). Prednisolone and methylprednisolone were only rarely administered (Corbel and Eades, 1977; Yamakami et al., 1996; Hashimoto et al., 1998; Paris et al., 2003; McCulloch et al., 2009, 2012; Zhao et al., 2010; Rebong et al., 2011; Alsaadi et al., 2012; Zhao and Perlin, 2013; Zhang et al., 2014). Many dosages were tested for steroids, either in a single administration or in repeated applications. Cortisone sc 100–200 mg/Kg 3 times a week (tiw) for the 2 weeks before experimental infection was quite common in mice.
Alkylating drugs were reported to be used in 42.9% papers, and in 24.9% in association with steroids. Intraperitoneal injection of cyclophosphamide at a 150 mg/Kg tiw dosage the week before infection was mostly used in mice. Perifosine (Bonifazi et al., 2010), busulphan (Baisakh et al., 1975; Stein et al., 2013), and nitrogen mustards (Schaffner et al., 1982; Schaffner and Frick, 1985; Schaffner and Böhler, 1993) were also tested, but all of them primarily in former works.
Other immunosuppressive medicines were used, like 5-fluorouracil (Hata et al., 1996a,b, 2011; Wallace et al., 1997; Graybill et al., 1998, 2003a; BitMansour et al., 2005; Stojanovic et al., 2011; Salas et al., 2013), tacrolimus (FK506; High and Washburn, 1997; Herbst et al., 2013, 2015; Shirkhani et al., 2015), cyclosporine A (Polak-Wyss, 1991; High and Washburn, 1997), mitotic poisons (Baisakh et al., 1975; Balloy et al., 2005a,b; Loussert et al., 2010; Hein et al., 2015). Tunicamycin was employed once because of its anti-Natural Killer (NK) cell property (Maheshwari et al., 1988), and liposomal dichloromethylene diphosphonate (DMPD) because it decreases macrophages in liver and spleen (Moonis et al., 1994). Interestingly, 5 mg gold sodium thiomalate were injected 1 h before A. fumigatus inoculation in mice by Williams et al. (1981).
Forty-five articles recorded usage of immunotherapy. For 24 of them, anti-neutrophil Ly6 (Gr-1) rat IgG2b MAb57 antibody (clone RB6-8C5) was used on the basis of its property to react with mouse Ly-6G, i.e., a 21–25 kDa protein also known as the myeloid differentiation antigen Gr-1, for 24 of them (Supplementary Material 3). Some other antibodies, like anti-asialo GM1 and PK136, were injected to specifically study response due to NK cells (Maheshwari et al., 1988; Tandon et al., 1988; Morrison et al., 2003) or to target CD4+ and/or CD8+ T-cell lymphocytes (Corbel and Eades, 1977; Carvalho et al., 2012; Cruz et al., 2013). Some models described genetic rough depletion of all B-cell (Montagnoli et al., 2003) and T-cell lymphocytes (Maheshwari et al., 1988; Tandon et al., 1988).
Besides in 21 studies, 6–9 Gy irradiation was enforced to rodents in order to mimic total blood-cells depletion during bone marrow transplantation (Supplementary Material 3).
Genetic mutations were sometimes induced in some targeted rodent genes, when studying role of particular immunological pathways, for instance those involving the cytokines, receptors, proteins, enzymes associated with immune response, and activation of immunoglobulins, like MyD88, Card9, Rag1 and Rag2, PTX3, TRIF, and many others (Supplementary Material 3). Mutations in gp91phox or gp47phox genes were generated to mimic chronic granulomatous disease (Supplementary Material 3). Notably, an original model of surfactant protein (SP)-deficient mice was used to show that absence of SP-D protein made rodents more susceptible to invasive aspergillosis than the absence of SP-A (Brieland et al., 2001).
Alternative procedures were developed to allow invasive aspergillosis in very original opportunistic contexts, like septic infections following caecal ligation and puncture (Benjamim et al., 2003, 2005), as well as cancerous disease using 1.0 × 106 S-180 tumor cells suspension (Okawa et al., 2002), and rhinosinusitis after unilateral nasal obstruction with sponges (Zhang et al., 2013). In addition to all these abovementioned methods, rodents were sometimes fed with low-diet hypo-protein regimen to mimic the malnourished status of the weakest human patients that are usually infected with A. fumigatus in hospitals (Miyazaki et al., 1993; Mitsutake et al., 1995; Chandenier et al., 2009; Lo Giudice et al., 2010; Desoubeaux and Chandenier, 2012; Desoubeaux et al., 2014b).
In order to prevent any undesirable bacterial infection during experiments, antibiotics were reported to be administered in 181 works, that means in only 27.7% of the studies in which immunosuppression was induced. Variability of the drug(s) to be used for such a purpose was very high. Because they are cheap, cyclins, and especially tetracycline at a dosage of 1 mg/mL in drinking water, were mostly provided to rodents (Supplementary Material 4). Quinolones, usually administered in gavage food or in drinking water like enrofloxacin at a 5–8 mg/kg daily dosage, were second choices (Supplementary Material 4). They were sometimes dispensed in association with sulfamethoxazole-trimethoprim (Chang et al., 2004; Ito et al., 2006), oral cyclins (Aufauvre-Brown et al., 1997; Brown et al., 2000; Balloy et al., 2005b; McDonagh et al., 2008; Herbst et al., 2013, 2015; Huber and Bignell, 2014), or intra-muscular (IM) teicoplanin (van Vianen et al., 2006; Petrik et al., 2010, 2012, 2014; Verwer et al., 2013). Cephalosporins were a quite frequently-used alternative (Supplementary Material 4): ceftazidime was sc- or IM-injected at a 50 mg/Kg daily dosage. Oral and IM usage of aminosides was rarer (Supplementary Material 4). Injectable glycopeptides and carbapenems were sporadically used to prevent infections due to multi-resistant staphylococci and enterobacteriae (Melchers et al., 1994; Hashimoto et al., 1998; Yonezawa et al., 2000; Benjamim et al., 2005; Steinbach et al., 2006; Tansho et al., 2006; van Vianen et al., 2006; Cramer et al., 2008; Pinchai et al., 2009; Rivera et al., 2009; Petrik et al., 2010, 2012, 2014; Grahl et al., 2011; Martinez et al., 2013; Verwer et al., 2013). Likewise, multi-antibiotic associations were sometimes reported, like the following scheme: ciprofloxacin (660 mg/L) and polymyxin B (100 mg/L) in water, plus IM amoxicillin (40 mg/kg/day) and a single shot of IM gentamicin (6 mg/kg) at time of experimental infection (Leenders et al., 1996; Dams et al., 1999; Becker et al., 2000, 2002a,b, 2003, 2006; Van Etten et al., 2000; Ruijgrok et al., 2001, 2005, 2006). Interestingly, a few authors preconized to dilute halogenated derivatives, like chlorine or iodine, as disinfectants in drinking water (Corbel and Eades, 1977; Xu et al., 2015). To note, six studies clearly asserted that antibiotic usage was not required, because of low incidence rate of opportunistic bacterial infection that they had noticed in the past works (de Repentigny et al., 1993; Kretschmar et al., 2001; Lewis et al., 2002; Chaturvedi et al., 2005; Iannitti et al., 2013; Speth et al., 2013).
Choice of the Aspergillus fumigatus Strain
Overall, 230 distinct A. fumigatus strains were reported in the literature for experimental infection (Table 2). The analysis was made difficult because of the global use of synonyms and unofficial alternative strain names.
AF293, also known as ATCC MYA-4609, FA/1153, FGSC A1100, CBS 101355, NCPF 7367, or IHEM18963, was the most used strain, including sometimes its derivatives like the fluffy variant (Ben-Ami et al., 2010b) or the Af293.1 (Bok et al., 2005, 2006; Tsitsigiannis et al., 2005; Cramer et al., 2006; Romano et al., 2006; Dagenais et al., 2008; Ma et al., 2008; Qiao et al., 2008; Lee et al., 2009; Han et al., 2010; Leal et al., 2010, 2012, 2013; Jhingran et al., 2012; Sekonyela et al., 2013; Taylor et al., 2014b; Kerr et al., 2016) and Af293.6 mutants (Dagenais et al., 2008; Leal et al., 2012; Sekonyela et al., 2013; Kerr et al., 2016), in 18.3% of the experimental infections (Supplementary Material 5). It was followed by Dal/CEA10, also referred to as CBS 144.89, D141, IHEM6963, NIH 4215, ATCC MYA-1163, and AF10, in 8.0% of the cases (Supplementary Material 5). Ku80 strains, like CEA10: ΔKu80, also named Ku80/CEA17 or FGSC 1152, which is one of Dal/CEA10 derivate, have been largely used because they increased homologous recombination for gene replacement. There are Ku80 pyrG+ and pyrG– strains, and the ones that are auxotrophic mutants in the uracil/uridine pathway express attenuate virulence. H11–20 strain was chosen in 15 works, but mostly in rat models (Supplementary Material 5), as it was first isolated from rat dying of spontaneously-acquired aspergillosis while on steroid treatment.
Some A. fumigatus strains were selected for their very specific features like bioluminescent AfC3 (Brock et al., 2008; Ibrahim-Granet et al., 2010; Fekkar et al., 2012; Jouvion et al., 2012; Morisse et al., 2012) or Af 2/7/1 (Galiger et al., 2013; Savers et al., 2016). Among other examples, one should notice for instance AF91 (also maned NCPF 7100, IHEM 13936, or J960180) which expresses attenuated virulence (Denning et al., 1997a,b; Dannaoui et al., 1999; Overdijk et al., 1999; Warn et al., 2003, 2006, 2010; Paisley et al., 2005), or EMFR S678P that resists to echinocandins (Miyazaki et al., 1993; Lepak et al., 2013a,b,c) and AZN 58 for which flucytosin is not active (Verweij et al., 2008). V28–77 and V59–73 are azole-resistant strains that were employed to assess impact of M220I mutation and G54 substitution in the gene coding Cyp51A upon the pharmacokinetics and pharmacodynamics properties of voriconazole (Mavridou et al., 2010a,b; Seyedmousavi et al., 2015).
In addition, 105 articles reported usage of reference strains but genetically modified by specific mutation/deletion, like ΔgliA (Cramer et al., 2006; Kupfahl et al., 2006; Sugui et al., 2007a; Chiang et al., 2008) and ΔgliP (Wang et al., 2014), ΔchsC (Mellado et al., 1996; Aufauvre-Brown et al., 1997) and chsG (Mellado et al., 1996), ΔsebA (Dinamarco et al., 2012a,b), ΔcatA (Paris et al., 2003; Ben-Ami et al., 2010a; Ben-Ami and Kontoyiannis, 2012; Leal et al., 2013), or ΔaspB (Vargas-Muñiz et al., 2015). They were usually tested in virulence studies that focused on fungal factors like proteins of the cell wall integrity, elastase, and other miscellaneous proteases (Kothary et al., 1984; Kolattukudy et al., 1993; Frosco et al., 1994), catalases (Paris et al., 2003), phospholipase, toxins (Paris et al., 1993; Bok et al., 2006; Cramer et al., 2006; Sugui et al., 2007a,b; Gravelat et al., 2008; Ben-Ami et al., 2009; Wang et al., 2014), adhesins, restrictocins (Smith et al., 1993), conidial pigments like melanin (Jahn et al., 1997; Langfelder et al., 1998), histidine kinase (Bartroli et al., 1998; Clemons et al., 2002; Du et al., 2002), calcineurine (Steinbach et al., 2006; Juvvadi et al., 2013), alkaline protease (Monod et al., 1993; Tang et al., 1993; Jaton-Ogay et al., 1994; Smith et al., 1994), and chitin synthase (Mellado et al., 1996; Aufauvre-Brown et al., 1997).
Moreover, 141 manuscripts reported usage of miscellaneous strains that had been locally isolated and that were not referenced in official collections (Supplementary Material 5).
Implementation of the Experimental Infection
Size of the fungal inoculum for the experimental infection varied greatly (Table 2), depending especially on the A. fumigatus strain(s) selected (Johnson et al., 2000; Takemoto et al., 2004, 2006; Cacciapuoti et al., 2006; Warn et al., 2006, 2010; Mavridou et al., 2010a,b), the aims of the study [e.g., Gao et al., doubled the inoculum when studying histopathology (Gao et al., 1997), whereas O'Hanlon et al. increased it 6-fold for the same purpose (O'Hanlon et al., 2011)], as well as the immunocompromised status of the animals (globally, the more immunocompromised are the rodents, the lower inoculum is needed to induce aspergillosis in them) and the route of administration (Mehrad et al., 1999; Cenci et al., 2000; BitMansour and Brown, 2002; Bozza et al., 2003; Chang et al., 2004; Takemoto et al., 2004, 2006; Grahl et al., 2012; Slesiona et al., 2012; Juvvadi et al., 2013; Wharton et al., 2015).
For instance, inoculum deposited into the rodents was particularly low when A. fumigatus spores were aerosolized into an inhalation chamber, like the acrylic Hinners' chamber described by Steinbach et al. and the large-scale inhalational Madison chamber: around 1.0 × 104 conidia per animal, although a larger fungal suspension was needed to generate the inoculum (Steinbach et al., 2004, 2006; Cramer et al., 2008; Pinchai et al., 2009; Leleu et al., 2013a,b; Lamoth et al., 2014a,b): the mean size was ~1.4 × 109 conidia/mL, extreme values ranging from 1.0 × 103 (Ahmad et al., 2014) to 1.2 × 1012/mL (Chiang et al., 2006; Gravelat et al., 2008; Evans et al., 2010a; Ibrahim et al., 2011), in a 10–40 mL-suspension (mean: 12 mL) spread during 25 min to 1 h, with a flow rate of 100–200 kPa (1–2 bar). Out of all the articles retained for analysis, 8.8% used such devices (Supplementary Material 6). In 304 works, the intranasal route was privileged. Out of them, 297 used mice, which represented 43.7% of all the articles dealing with this rodent species: an average of 2.0 × 107 A. fumigatus spores were instilled in nares (min: 3.0 × 103—max: 5.0 × 108). In contrast, the intranasal route was more rarely reported for rats (mean inoculum size: 7.2 × 106, min: 5.0 × 103—max: 2.0 × 107; Hachem et al., 2006; Morisse et al., 2012, 2013; Zhang et al., 2013; Yan et al., 2014), in part because their respiratory apparatus is longer and their alveoli harder to reach. Instead in them, the bronchial-tracheal instillation was primarily chosen in 67.1% of the papers (Supplementary Material 6): the mean inoculum size was 2.5 × 107 spores, and spanned from 1.0 × 103 (Khan et al., 2008) to 1.0 × 109 (Land et al., 1989), whereas it was quite close to this in mice undergoing the same bronchial-tracheal procedure, 2.2 × 107 (min: 1.5 × 102—max: 2.0 × 107; Björgvinsdóttir et al., 1997; Bozza et al., 2002b, 2003; Garlanda et al., 2002; Montagnoli et al., 2003). In most of the cases, spores suspension was most often instilled into the trachea or directly into the lungs after tracheotomy or small thoracotomy (Habicht et al., 2002). In some studies, a cannula was passed into the trachea through the mouth, and then one of the lung lobes (Balloy et al., 2005b; Ruijgrok et al., 2005; Nagasaki et al., 2009). Passing through the upper airways, the MicroSprayer® Aerosolizer device was proven to generate an air-dispersed controlled cloud of conidia in bronchial-tracheal apparatus of rats, mimicking then the real pathophysiology of airborne A. fumigatus exposure (Chandenier et al., 2009; Desoubeaux and Chandenier, 2012; Desoubeaux et al., 2014b). To our knowledge, only one research facility employed bronchial-tracheal route in guinea pigs (Chandrasekar et al., 2000, 2004).
IV route was used to generate rapid fungal dissemination through bloodstream. It usually required a smaller A. fumigatus inoculum, regardless the immunocompromised status of the rodents: 2.3 × 107 spores in average, extremes ranging from 5.0 × 101 (Cutsem et al., 1993) to 2.5 × 109 (Bowman et al., 2001), and its usage was reported in 30.0% of the selected articles of this review. Jugular vein (Sivak et al., 2004a,b; Risovic et al., 2007), femoral vein (Wong et al., 1989), and penis vein (Overdijk et al., 1996; Reichard et al., 1997; Odds et al., 1998) were generally chosen for such a purpose in large rodent species, while lateral tail vein was privileged in mice (Odds et al., 1998). Retro-orbital vein was an alternative, but has become less frequent in recent years for ethical considerations (te Dorsthorst et al., 2005; Verweij et al., 2008; Wagener et al., 2008; Mouton et al., 2009; Dirr et al., 2010; Kotz et al., 2010; Heesemann et al., 2011). To provide free movement for the animal and to make easier the IV administration, Odds et al. and Meerpoel et al. connected a catheter to the ligated jugular vein via a proprietary swivel device (Odds et al., 2000; Meerpoel et al., 2010). To induce endocarditis in guinea pigs, Martin et al. injected 1.0 × 104 spores in the internal carotid, passing a catheter through the left auricle and just through the mitral valve (Martin et al., 1997).
Alternative route of experimental infection were sometimes studied to address some very specific forms of invasive aspergillosis, like local eye invasion during endophthalmitis or ulcerative keratitis, and cerebral infection (Supplementary Material 6). For ocular infection, the corneal epithelium was abraded by needle (Carrion et al., 2013) or totally removed with a paracentesis knife that perforated cuts perpendicular to each other (Ren et al., 2010), then the damaged region was either smeared with conidia (Zhong et al., 2016) or directly injected (Zhong et al., 2009); but in both situations, great difficulties were encountered to reliably adjust the inoculum size. For the models of cerebral aspergillosis, the investigators injected A. fumigatus spores directly into the brain resulting in local high tissue burdens (Chiller et al., 2002). For instance, Mozzala et al. used a 0.1 mL glass micro-syringe associated with a 27-gauge disposable needle to go through the central area of the frontal bone of mice to a depth of 2 mm (Mazzolla et al., 1991). In rats, Zimmerli et al. proposed to reach the cisterna magna using an innovative procedure that does not produce structural brain damage. It appeared to be well-tolerated by all animals as no mortality was observed during the first 12 h after injection (Zimmerli et al., 2007). For a nephritis model, 1.0 × 103 spores were injected into the intra-medullar kidney (Walzl et al., 1987; Schaude et al., 1990). Cutaneous abscesses were generated in murine soft tissues by sc injections, mainly into the thigh after fur removal (Lupetti et al., 2002; Ruiz-Cabello et al., 2002; Donat et al., 2012), or by intradermal injections into the ears (Petersen et al., 2002; Goebel et al., 2005; Stein et al., 2013).
Altogether, the frequency of spore administration to induce experimental infection was generally based on a single application, except for some specific studies that performed daily inoculations over 2 or 3 days (or even over a longer period; Smith et al., 1994; Cenci et al., 1998, 1999, 2000, 2001, 2002; Del Sero et al., 1999; Bozza et al., 2002a; Du et al., 2002; Bellocchio et al., 2004a,b, 2005; Gaziano et al., 2004; Mellado et al., 2005; Shao et al., 2005a; Mazaki et al., 2006; Zelante et al., 2007, 2009, 2015; D'Angelo et al., 2009; Morton et al., 2010, 2012; Moretti et al., 2012, 2014; Zhang et al., 2013; Hein et al., 2015), or repeated spaced administrations (Smith, 1972, 1973, 1977; Turner et al., 1975a,b; Morton et al., 2012; Turner et al., 1976; Lehmann and White, 1976; de Repentigny et al., 1993; Cenci et al., 1997; Mazaki et al., 2006; Fei et al., 2011; Templeton et al., 2011; Alcazar-Fuoli et al., 2015; Savers et al., 2016), sometimes in order to induce immune protection (Centeno-Lima et al., 2002) or to enhance the infection yields, particularly for inhalational models in chamber (Buskirk et al., 2014).
Validation of the Model and Outcome Parameters to Follow Up
Overall, 81.6% of the studied literature reported usage of at least one test to confirm that the experimental infection has been correctly implemented in rodents or to assess the fungal burden (Table 2), and 38.6% noticed the concomitant usage of two or more distinct techniques. Histopathology and in vitro mycological culture have been more largely described, in 52.8 and 49.0% articles, respectively, sometimes concomitantly in 25.6% of the cases (Supplementary Material 7).
More recent methods include the detection of GM, (1 → 3)β-D-glucans, and fungal DNA by polymerase chain reaction (PCR; Supplementary Material 7). In 4.9% of the animal studies, the chitin assay was used for assessment of fungal burden in fluids or homogenized tissues (Supplementary Material 7).
Some investigators analyzed the host immune response as outcome measure (Morgenstern et al., 1997; Cenci et al., 1998, 1999; Duong et al., 1998; Brieland et al., 2001; Shao et al., 2005b; Steele et al., 2005; Bonnett et al., 2006; Montagnoli et al., 2006; Cornish et al., 2008; Herbst et al., 2013; Kasahara et al., 2016; Renshaw et al., 2016). For instance in bronchial-alveolar lavage (BAL) fluids, they measured cytokine concentrations through ELISA assays, and addressed representation changes of lymphocyte cells or phagocytic cells by flow cytometry. NADPH-oxidative pathway was also investigated as markers for evolution of the infection (Aratani et al., 2002; Philippe et al., 2003; Cornish et al., 2008; Stein et al., 2013; Prüfer et al., 2014; Röhm et al., 2014).
Detection of anti-Aspergillus antibodies was sometimes performed (Turner et al., 1975b, 1976; Naik et al., 2011). In human medicine it is nonetheless not considered as a biomarker of invasive infection, but rather of chronic or allergic aspergillosis. To attest of the consequences of invasive infection, blood urea nitrogen, creatinine, serum glutamic pyruvic transaminase (ALAT), and serum glutamic oxaloacetic transaminase (ASAT) in serum were measured as indirect but unspecific surrogate endpoints (Singh et al., 2014).
Discussion
Animal models of invasive aspergillosis have been developed to make the link between in vitro experiments and clinical trials. They have been used extensively to study various aspects of pathogenesis, innate and acquired host-response, disease transmission, diagnostic tools assessment, and preclinical therapy during aspergillosis (Clemons and Stevens, 2005, 2006a,b). Theoretically, a perfect unique model, i.e., highly reproducible, economical and standardized, should be expected to address reproducibly all these issues (Najvar et al., 2004; Patterson, 2005). This present overview attempted to summarize all the technical parameters reported in the literature about rodent models of invasive infection caused by A. fumigatus, but was not intended to detail the wealth of insight gained from them into the pathogenis, host immune response, diagnosis, and treatment, and the reader is invited to refer to other publications (Sable et al., 2008; Brown, 2011; Steele and Wormley, 2012; Wüthrich et al., 2012; Drew et al., 2013; Lanternier et al., 2013). Overall, it highlighted great variations regarding all the experimental settings of the lab models. Actually, such heterogeneity has always existed in disease models in other systems (Maarman et al., 2013; Golden et al., 2015), and this is rather a general problem in research and not exclusive to aspergillosis, but herein, the variables were both related to the host and the pathogen factors, as well as to the route of infection and the size of fungal inoculum. These discrepancies likely resulted in great differences on the study results. For instance, combination of caspofungin (an echinocandin drug) and liposomal amphotericin B (a polyen), as well as the association of caspofungin with amphotericin B lipid complexes, was shown to have no significantly enhanced activity in a cerebral model of aspergillosis (Luque et al., 2003; Clemons et al., 2005; Imai et al., 2005), whereas in contrast, micafungin (another echinocandin) had reduced activity and even possible enhanced drug toxicity with triamcinolone against pulmonary disease in steroid-suppressed mice (Clemons and Stevens, 2006c). In this latter model, it was also found that the combination of micafungin and itraconazole was antagonistic, highlighting a decrease in efficacy (Clemons and Stevens, 2006c), while it was not in a systemic model (Luque et al., 2003). Investigating the pathogenesis of gliotoxin-producing and non-producing isogenic strains of A. fumigatus, a series of studies demonstrated that the secondary metabolite contributes to virulence in a non-neutropenic murine model of disease, through its effects on NF-κB-dependent host cell apoptosis and on phagocyte NADPH oxidase function, but not in neutropenic murine models (Kupfahl et al., 2006; Sugui et al., 2007b; Spikes et al., 2008). Beyond these few examples, several other studies also demonstrated that drugs may have different therapeutic effects and virulence factors have distinct impacts depending on the chosen model (Graybill et al., 2003a). Thus, a minimum standardization seems necessary to reliably compare the results between laboratories.
First, choice regarding the rodent species and strain is critical to ensure correct reproducibility (Mitsutake et al., 1995; Durrant et al., 2011; Mirkov et al., 2014). Of course, murine models have predominated for most investigators over the years. Regarding their respective physiology, it is indeed acknowledged that mice and humans have similarities in organ systems, biochemistries, pathologies, and even in their two genomes that both encompass ~30,000 genes and for which the proportion with no homology between them is < 1% (Mouse Genome Sequencing Consortium et al., 2002). Moreover, the limited body size of mice allows usage of a relatively large number of animals to be tested simultaneously under identical conditions, which is of course relevant to perform statistical analysis. In addition, many commercial reagents and kits are readily available to aid in studies. Furthermore, in the light of all the genetically-defined mouse strains that are currently available, scientists have the great possibility to select the most appropriate host factors they need for mimicking specific clinical situations and to generate an infection according to a well-defined pattern (Pollock et al., 1995; Morgenstern et al., 1997; Dennis et al., 2006; Lengerova et al., 2012; Leleu et al., 2013b). For instance, BALB/c mice are a well-known general multipurpose model allowing studying for infectious diseases, while C57BL/6 strain represents permissive background for maximal expression of most mutations, like those in genes coding for cytokines, toll-like receptors (TLRs), Dectin-1 or other receptors/proteins associated with immune response (Steele et al., 2005; Carrion et al., 2013; Herbst et al., 2013, 2015; Leal et al., 2013; Shepardson et al., 2013; Bozza et al., 2014; Espinosa et al., 2014; Moretti et al., 2014; Taylor et al., 2014b; Caffrey et al., 2015; Jhingran et al., 2015; Karki et al., 2015; Wharton et al., 2015; Zelante et al., 2015; Kasahara et al., 2016; Savers et al., 2016). Usage of genetically-deficient knockout (KO) mice provided new insights upon the pathophysiology of invasive aspergillosis (Deepe et al., 2000). For example, it proved that Interleukin- (IL-)6, IL-12 and interferon-γ (INFγ) were protective factors against A. fumigatus. In contrast, IL-10 and IL-4 deficiency made respective KO-mice more resistant to infection (Cenci et al., 1998, 2001; Del Sero et al., 1999; Clemons et al., 2000a): neutralization of IL-10 was reported to up-regulate production of nitric oxide, contributing to an effective fungicidal (Romani et al., 1994), and IL-4 cured 70% of infected mice when administered exogenously while protecting them from a second lethal challenge (Cenci et al., 1997). Importantly, KO-models also showed that IL-17, as well as TLR-4 and TLR-2 are of great importance in the innate response against A. fumigatus. For instance when they are TLR-2 KO, mice had low tumor necrosis factor-(TNF)-α and IL-12 rates, as well as reduced survival and higher fungal burdens in the tissues than competent mice (Bellocchio et al., 2004a; Balloy et al., 2005b). In a near future, it is expected that the improvement of all the molecular tools will be able to provide more and more genetically modified rodent strains. It is also noteworthy that AKR/J, C57BL/6, 129/SvJ, and BALB/c inbred strains were shown to be more resistant to A. fumigatus infection than MRL/MPJ and NZW/LacJ mice which were themselves more resistant than DBA/2 (Zaas et al., 2008). By the way, these latter are complement deficient, and their susceptibility to invasive aspergillosis sheds light on the role of complement in host defense against the fungus. One of the advantages of all the aforementioned inbred strains relies in that they express less host genetic variability (Festing, 2010), although some slight differences have been raised up for DBA/2 and BALB/c strains between Great Britain and U.S.A. (Hector et al., 1990). However, one could argue that invasive aspergillosis actually occurs in genetically-not defined human patients which probably exhibit great genomic heterogeneity (Goldman and Osmani, 2007). In contrast, outbred strains are genetically randomized, and their phenotypic background is not totally controllable (Chia et al., 2005). As evidenced by several examples with Albino Swiss Webster before the year 2000 (Sandhu et al., 1970, 1976; Smith, 1972, 1973; Baisakh et al., 1975; Lehmann and White, 1975, 1976, 1978; Ghosh et al., 1977; White, 1977; Saeed and Hay, 1981; Polak, 1982, 1987; Polak et al., 1982; Van Cutsem et al., 1984, 1987; Dixon, 1987; Maheshwari et al., 1988; Tandon et al., 1988; Dixon et al., 1989; Hector et al., 1990; Clark et al., 1991; Polak-Wyss, 1991; Paris et al., 1993; Thau et al., 1994; Wiederhold et al., 2004; Lewis et al., 2005), they should only be considered during drug development for pre-clinical screening studies, when specific host factors are not assumed to be critical (Clemons and Stevens, 2006a). In contrast, outbred mice are not suited to precise pharmacology/pharmacokinetics studies, because for example their gut mucosae enable too rapid metabolism for azole drugs (Sugar and Liu, 2000; MacCallum and Odds, 2002; Graybill et al., 2003b). Although the mouse strains are less expensive, use of bigger animals, like guinea pigs, have the advantage to allow serial sampling, like repeated blood sampling as well as BAL. Moreover, guinea pigs do not express an acute infection pattern, and thus likely allow reducing the number of animals to be used by reducing rapid mortality (Riera et al., 1983; Capilla and Guarro, 2004). Doses and antifungal regimens can also be easily monitored and modified (Reichard et al., 1997; Chandrasekar et al., 2004; Clemons and Stevens, 2005; Vallor et al., 2008; Wiederhold et al., 2009, 2013, 2015; Dufresne et al., 2012; Hooper et al., 2012; Kirkpatrick et al., 2012; Lengerova et al., 2012; Jambunathan et al., 2013; Zhao et al., 2015; White et al., 2016), especially for addressing clinical efficacy and pharmacodynamics/pharmacokinetics of echinocandin or azoles (Van Cutsem et al., 1989, 1990; Cutsem et al., 1993; Arrese et al., 1994; Overdijk et al., 1996, 1999; Reichard et al., 1997; Odds et al., 1998, 2000; Kirkpatrick et al., 2000b, 2002, 2006; Loeffler et al., 2002; MacCallum et al., 2005; Meerpoel et al., 2010). Indeed, the in vivo metabolism in guinea pigs is thought to be comparable to this in humans because of a slow clearance (Graybill et al., 2003b). Using an interesting endocarditis model in guinea pigs (Martin et al., 1997), some investigators were able to highlight the superiority of voriconazole over itraconazole to cure aspergillosis. Nonetheless, in spite of all their valuables features, guinea pigs have been poorly used so far, maybe because they express complex social structure and are stressed under unfamiliar environments or the experimental manipulations (Hennessy, 1999). In such a context, rats may be considered as an interesting compromise between mice and guinea pigs. Rats have been most often used to study invasive aspergillosis with initial pulmonary course (Habicht et al., 2002; Chandenier et al., 2009; Desoubeaux and Chandenier, 2012; Desoubeaux et al., 2014b). Rabbit models (Kurup, 1984; Komadina et al., 1985; Longman and Martin, 1987; Patterson et al., 1988, 1989; Singh et al., 1990; Berenguer et al., 1995; Walsh et al., 1995; Mylonakis et al., 1997; Kirkpatrick et al., 2000a; Roberts et al., 2000; Petraitiene et al., 2004; Clemons and Stevens, 2005; Hao et al., 2008; Petraitis et al., 2009), as well as those developed in birds (Ghori and Edgar, 1979; Chaudhary et al., 1988; Suleiman et al., 2012; Melloul et al., 2014) or in other animals like cows (Jensen et al., 1996), sheep (Corbel et al., 1973; Boase et al., 2011), monkeys (Mahajan et al., 1978), or invertebrates (Lionakis et al., 2005; Chamilos et al., 2010; Cheema and Christians, 2011; Lionakis and Kontoyiannis, 2012; Gomez-Lopez et al., 2014) have also been tested. Invertebrates like Drosophila melanogaster, Danio rerio, Caenorhabditis elegans, and Galleria mellonella were shown to be useful to study drug distribution, toxicology and metabolic stability, but their highly-simplistic physiology is far much different from that in humans (Giacomotto and Ségalat, 2010). In contrast, work in rabbits appeared relevant, mainly because this species is highly susceptible to infection, and allows serial sampling and easy administration of drugs (Schmidt, 2002). The ability to visualize anatomic details by computed tomography is particularly advantageous to check the progression of focal aspergillosis (Walsh et al., 1995; Petraitiene et al., 2002). However this animal model is more expensive, requires specific facility for this husbandry, and is limited by the availability of a few immunological and biomolecular reagents. There is also a lack of genetically-defined rabbit strains (Capilla et al., 2007).
Another point to thoroughly discuss is the immunosuppressive regimen to administer to the rodents. Since many human patients with invasive aspergillosis are rendered immunocompromised by prior exposure to cytotoxic chemotherapy and/or steroids (Bitar et al., 2014), most of the animal models have included one or both of these medications in order to facilitate correct implementation of A. fumigatus disease. Cytotoxic agents, like alkylating drugs e.g., cyclophosphamide or cytosine arabinoside, bind to DNA during cellular replication and thus induce profound neutropenia (Johnson et al., 2000; Chandenier et al., 2009; Desoubeaux and Chandenier, 2012). The histological and radiological features of models treated with alkylating drugs were very close to those of profoundly neutropenic infected patients, like those undergoing leukemia (Chandenier et al., 2009): Fungal growth, dissemination, and destruction of parenchymal architecture by invasive hyphae is the primary mechanism of tissue injury and death. However, the neutropenic models are currently becoming less relevant, since the characteristics of human patients infected with A. fumigatus are progressively changing and have less such traditional risks for invasive disease (Upton et al., 2007; Lortholary et al., 2011). Steroids use in mice showed distinct pattern of pathogenesis in comparison with neutrophil-depleting drugs: in steroid-treated rodents, fungal growth is significantly reduced in comparison to chemotherapy-treated animals, and the massive influx of functionally impaired leukocytes triggers dysregulated responses associated with tissue damage, hypoxia, and immunopathology (Balloy et al., 2005a; Grahl et al., 2011). Indeed, steroids affect alveolar macrophage function, and thus reduce the first barrier to pulmonary infection. They also impact T- and B-cell lymphocytes, and they decrease the production of cytokines, which compromises the adaptive immune response against invasive aspergillosis (Tang et al., 1993). In experimental works, dexamethasone was less effective than hydrocortisone or triamcinolone to induce aspergillosis (Marr et al., 2004). Convincingly, the combination of both alkylating and steroid drugs seems valuable to increase lethality in infected animals (Dixon et al., 1989). Some non-neutropenic immunocompromised models have been developed with cyclosporine A and/or steroids to stimulate long-term post-engraftment immune-impaired conditions of bone marrow transplant recipients (Lengerova et al., 2012; Leleu et al., 2013b), while persistent neutropenic supports may be generated by neutrophil-depleting monoclonal antibodies, like IgG2b MAb57 antibody (Mehrad et al., 2002; Richie et al., 2007a,b; Park et al., 2010; Kapp et al., 2014; O'Dea et al., 2014). Regardless of method, a regular monitoring of leucocytes count is recommended to ensure a correct achievement of the immunocompromised status (Stephens-Romero et al., 2005). However, it is important to underline herein that any experimental immunosuppression can affect the host-response to infection, and de facto enhances complexity of the model understanding. These effects should be clearly defined, as they actually impact the final conclusions of the study (Balloy et al., 2005a). For instance although nude mice have no mature T-cell lymphocytes, their macrophages were described as being at a higher basal state of activation, and they had increased numbers of NK cells (Cheers and Waller, 1975), so that they experienced potential paradoxical resistance during early during infection. Therefore, it is imperative not to extrapolate data to other susceptible or non-susceptible host states in the absence of experimental confirmation.
Only a few articles out of all the literature actually mentioned anti-opportunistic antibiotic prophylaxis, when rodents underwent immunosuppressive regimen, although undesirable bacterial infections had been shown to hamper and precede correct development of experimental fungal infection (Clemons et al., 2006a). Thus, every effort to prevent them appears worthwhile (Schmidt, 2002). Specific pathogen-free animals represent a valuable option, but their cost makes them less available, especially for the research facilities that are not equipped with adapted sterile housing conditions. Instead, three antibiotic schemes have been mainly used, alone or in combination: cyclins or quinolones provided through the beverage (Yu et al., 1990; Pollock et al., 1995; Cenci et al., 1997; Martin et al., 1997; Niwano et al., 1999; Morisse et al., 2012; Herbst et al., 2013), and sc ceftazidime (Kirkpatrick et al., 2012; Lengerova et al., 2012; White et al., 2016). The oral route may actually be considered as only suboptimal as it results in variable exposure. Indeed, drinking water which contains antibiotics tends to be progressively less intake by rodents as their infection progresses (Chandenier et al., 2009), and oral bio-availability is low for the above-mentioned antibiotics (Cunha et al., 1982). Likewise, injection of third generation-cephalosporin with large antibiotic spectrum have effects upon cytokine expression and disruption to normal microflora in the host. Thus, ceftazidime, which displays anti-Pseudomonas and anti-Enterobacteriaceae activities, may play a deleterious role when studying pathophysiology during aspergillosis (Cramer et al., 2008; Pinchai et al., 2009).
Regarding the route of experimental challenge, IV inoculation primarily induces overwhelming systemic disseminated infection in rodents (Kirkpatrick et al., 2000b; Seyedmousavi et al., 2013). It is probably the easiest route to standardize, because all the A. fumigatus inoculum is directly and entirely injected into the bloodstream through an accessible vein. Liquid fungal suspension can be precisely quantified and calibrated for minimal inter-experimental variability. Thus, IV route evidenced an excellent infection/dose-to-mortality ratio, and did not systematically require a preceding immunosuppression of the animals (Schmidt, 2002): without immunosuppressive regimen, an inoculum seized around 1.0 × 107 A. fumigatus conidia per mouse was largely enough to lead to an acute and reproducible infection (Paulussen et al., 2014, 2015). On the contrary, administration of cyclophosphamide or cortisone in such a context led to higher variation in clinical outcome. Thus in immunocompetent guinea pigs, the IV route has been particularly useful in pre-clinical therapeutic studies, as well as in assessing the kinetics of diagnostic markers (Kirkpatrick et al., 2000b), meanwhile reducing the number of animal experiments (Kirkpatrick et al., 2013). However, the IV route is of course estimated as an unnatural process, especially because this experimental inoculation procedure does not recapitulate the real one during aspergillosis bypassing mucosal host defense, and because it involves unusual organs, like kidneys (Andriole et al., 1992). Likewise, intra-abdominal infection should not be privileged, since it is not clinically relevant, and rodents often resist to this artificial route of inoculation. In comparison, experimental challenge through the respiratory airways consistently mimic the natural entry into the human body and leads first to the development of invasive aspergillosis in lungs (Andriole et al., 1992; Sheppard et al., 2004, 2006; Steinbach et al., 2004). Nevertheless, it systematically requires a prior immunosuppression to be reproducible (Denning et al., 1995), except for models simulating chronic granulomatous disease (Morgenstern et al., 1997; Philippe et al., 2003). It is harder to standardize because the number of fungal elements arriving in lung tissue is generally uncertain, and all the animals do not react stereotypically (Sheppard et al., 2004; Steinbach et al., 2004). Consequently, respiratory challenge requires a higher number of rodents to be statistically relevant (Latgé, 1999). Besides, organ tropism remains a little bit different from the primary tissues that are usually infected in human patients: substantial renal involvement is high following pulmonary infection in mice (Clemons and Stevens, 2005). Given that they allow good histopathologic correlation with the human disease and reproducible colony-forming unit(s) (CFU) counts (Sheppard et al., 2004), inhalational models in hermetic chamber have been largely promoted for better recapitulating course of natural exposure (Andriole et al., 1992; Patterson, 2005), but they require a heavy inoculum (typically 12 mL of 109 conidia/mL spread during 40 min before a 1h-subsequent exposure). Using a particle counter, it was evidenced that < 5% of generated particles were bigger than 5μm, which means that nebulization was likely efficacy enough to disperse A. fumigatus spores and avoid agglomerates (Leleu et al., 2013a,b). Tween and triton detergents have been commonly used to prepare fungal cell suspensions (Stephens-Romero et al., 2005). Some assays attempted to measure the amount of fungus that was actually inhaled by the challenged animals, and results were contradictory: Bretz et al. estimated the intake inoculum at 3.4 × 106 in mouse lungs, when a 1.0 × 108 A. fumigatus conidia/mL suspension was aerosolized during 90 s by pumping air into the flask using a 60 mL-syringe (Bretz et al., 2008). Ibrahim et al. estimated a mean inhalation of only 2.1 × 103 conidia, when 1.2 × 1010 were nebulized in the chamber (Ibrahim et al., 2010, 2011). To improve the infection rate, Evans et al. suggested to supplement room air with 5% CO2 to promote maximal ventilation and homogeneous exposure throughout the lungs (Evans et al., 2010a,b). Some large-scale devices, like the Madison chamber, now offer the possibility to simultaneously deliver an accurate infectious inoculum by aerosol to an extensive number of rodents, but also to larger species (McMurray, 2001). Buskirk et al. described an interesting device based on an acoustical generator to chronically deliver dry fungal powder aerosols to mice housed in a nose-only exposure chamber. It works for 2 h twice a week during 1 month (Buskirk et al., 2014). Older inhalational processes, like the 1 min-forced exposure over A. fumigatus culture in flask, appear obsolete: although this insufflation technique recapitulates human infection faithfully since conidia are not solubilized in solutions that typically contain detergents (Stephens-Romero et al., 2005), it remains hard to control as animals were dying rapidly within 3–4 days following infectious challenge (Le Conte et al., 1992). Furthermore, the number of rodents that can be fitted into the specialized inhalation chamber is modest. Instead of the inhalation route, the intranasal deposition of A. fumigatus droplets close to the nares may be considered valuable, as theoretically allowing a more controllable intake by the rodents. Unfortunately through this procedure, about only 10% of the deposited fungal load was estimated to reach the lungs (Markaryan et al., 1994). Besides, evaluation by qPCR showed that the intranasal route actually generated smaller fungal burdens with higher standard deviation, and less homogenous pneumonia (Steinbach et al., 2004), and so fungal lesions were likely to arise in larger airways rather than in alveoli (Tang et al., 1993; Shibuya et al., 1999; Steinbach et al., 2004). Thus, some investigators suggested to introduce 5 μL saline serum into the nostrils with the objective of drawing out some of the spores which could possibly have been left in this area (García et al., 2006), and/or to place the rodents in semi-vertical position just after the experimental infection (Bakker-Woudenberg, 2003). Likewise, Lepak et al. recommended a pulmonary aspiration following the intranasal deposition in order to drive the spore suspension toward the lung alveoli (Lepak et al., 2013a,b,c). This procedure produced invasive aspergillosis in more than 90% of animals for which mortality was 100% by 72–96 h post-infection, when not treated. In such a context, the bronchial-tracheal instillation appears as a relevant alternative to inhalational and intranasal models, but so far it usually required a minor surgical procedure to expose the trachea for injection below a small incision (Bakker-Woudenberg, 2003; Clemons and Stevens, 2005; Goldman and Osmani, 2007). In order to enhance dispersion of fungal suspension into the lungs, some studies reported that rodents were mechanically ventilated following the bronchial-tracheal deposition, using a respirator for 2 min (Prüfer et al., 2014). Instead, alternative techniques that minimize surgery seem interesting, like oropharyngeal aspiration (Amarsaikhan et al., 2014; Shepardson et al., 2014; Sugui et al., 2014). Some investigators suggested a spore delivery into the caudal oropharynx of anesthetized rodents, in which normal breathing resulted in fluid aspiration into the lungs (Sugui et al., 2010, 2011, 2012, 2014; Faro-Trindade et al., 2012; Liu et al., 2013; Amarsaikhan et al., 2014; Lilly et al., 2014; O'Dea et al., 2014; Röhm et al., 2014; Shepardson et al., 2014). Animals were suspended by their upper incisors from a suture thread on a 90° inclined board, and their tongue was gently extended to prevent them to swallow during the experimental infection, then the chest was gently compressed and released just after deposition of liquid (Vethanayagam et al., 2011; Grimm et al., 2013). To better target the lungs, some authors described utility of visual guidance into the trachea (Rayamajhi et al., 2011). Original devices, like the MicroSprayer® aerosolizer, that generate a cloud of A. fumigatus spores directly into the trachea is even more valuable to get closer to the human disease. They do not require highly-seasoned personnel (Goldman and Osmani, 2007; Chandenier et al., 2009; Desoubeaux and Chandenier, 2012). In addition to the respiratory and the IV challenge, miscellaneous alternative routes of infection were developed to address some very specific forms of invasive aspergillosis, like cerebral aspergillosis (Chiller et al., 2002, 2003; Zimmerli et al., 2007) which is probably the most common extra-pulmonary site of infection in human medicine and that results in more than 80% mortality. Such models of intra-cranial inoculation do not have to receive systematic exogenous immunosuppression. Although this is not the natural route by which people usually acquire cerebral aspergillosis, the histopathological lesions and cellular host-response were very similar to the observations in human infection (Chiller et al., 2002), describing development of abscesses and necrotic areas in brain and subsequent infectious foci through bloodstream dissemination (Kleinschmidt-DeMasters, 2002). These models were primarily helpful to show the benefit of combination therapies, and also to evidence that higher dosages of an antifungal drug are not always more curative (Clemons et al., 2005, 2006b; Imai et al., 2005; Singh et al., 2005; Clemons and Stevens, 2006c).
This review noticed great variability regarding the name of A. fumigatus strain(s) to be used in experimental models. This choice has probable important consequences on the conclusions that were drawn from the assays, because host responses to individual strains likely differ in magnitude and in quality (Rizzetto et al., 2013): actually, no one could legitimately extend his finding to other works because each strain owns its particularities. However, the actual need to choose of a unique strain for all the rodent models is still a debatable issue today. For instance, one could imagine that selecting an hypo-virulent strain to induce lower mortality is more convenient to study diagnostic tools benefits at early stage and over the course of the disease, but in the other hand, one could address easily the overall survival in pre-clinical therapeutic assays, when mortality rates is almost 100% with an hyper-virulent strain. Globally, investigators always privileged the usage of referenced A. fumigatus strains that had been first isolated from clinical samples during invasive aspergillosis course in patients. For instance, AFB62 (Sugui et al., 2011, 2014; Losada et al., 2015), TIMM 2920 (Tansho et al., 2006), AF210 also named as NCPF 7101 or 2.06013 (Denning et al., 1997a,b; Verweij et al., 1998; Johnson et al., 2000; Paisley et al., 2005), IFM 4942 (Yamada et al., 1993), or BMU 01200 were isolated in hematopoietic stem cell transplants (Sun et al., 2012; Zhang et al., 2015), as well as A22 and AF65 (also referred to as NCPF 7097 or ATCC® MYA772 for the latter) were collected in lung biopsy from immunocompromised patients (Denning et al., 1997a; Verweij et al., 1998; Denning and Warn, 1999; Johnson et al., 2000; Paisley et al., 2005; Speth et al., 2013). Likewise, Zhao et al. relevantly used a A. fumigatus strain that came from fungal endophthalmitis for their work about eye aspergillosis (Zhao et al., 2015). In contrast, it was more questionable when were used A. fumigatus strains that had not been isolated in a context of invasive aspergillosis, like MF13 first found in a sputum secondary to aspergilloma (Mitsutake et al., 1995; Otsubo et al., 1998, 1999; Kakeya et al., 2008; Takazono et al., 2009) or CBS 100079 in a human ear (Sarfati et al., 2002). Likewise, Af285 had been isolated from the sputum of a patient suffering from allergic aspergillosis, but not invasive form (Madan et al., 2001, 2010; Kaur et al., 2007; Singh et al., 2009). Sometimes, the strains were derivative from the veterinary medicine, since they had been first isolated in chicken or pigeons (Van Cutsem et al., 1984). However, in the light of its historical widespread usage and as it was sequenced first (Nierman et al., 2005), AF293, also referred to as ATCC MYA-4609, FA/1153, FGSC A1100, CBS 101355, NCPF 7367, or IHEM18963, and its derivatives appears of course as the most standard strain, although it is also known to express less virulence than other ones. Dal/CEA10, also named CBS 144.89, D141, IHEM6963, NIH 4215, ATCC MYA-1163, FGSC A1163, or AF10, has also been largely used. It is regrettable that only few studies thoroughly addressed variations in virulence among distinct A. fumigatus strains (Hanson et al., 1995). Comparison with other Aspergillus species like A. flavus, A. terreus, A. niger, and A. nidulans should be even harder: great inter-species discrepancies are supposed, as it was preliminary illustrated through the in vitro/in vivo correlation that was satisfactory for amphotericin B in a murine model infected with A. terreus, but bad for both A. flavus and A. fumigatus (Johnson et al., 2000; Mosquera et al., 2001).
In rodent models, another concern of variability is the size of A. fumigatus inoculum to be used for the experimental challenge. It was evidenced a dose-dependent correlation regarding the number of conidia in the inoculum with the severity of infection, regardless of the rodent model and the A. fumigatus strain (Dixon et al., 1989; Hector et al., 1990; Chiller et al., 2002; Clemons et al., 2000a, 2002), but our bibliographic analysis highlighted very large variations, ranging for instance from 1.0 × 102 (Waldorf et al., 1984) to 1.0 × 109 (Graybill et al., 1998) for mice infected by intranasal route. In addition, culture conditions and pre-infection technical steps are also great challenges for harmonization. Incubation temperature and humidity, timing of culture in plate, diluent to be used, and method for conidia counting in the suspension are variable parameters that change a lot depending on the protocols. Such various practices may have some consequences: for example, one could propose the consequences on the A. fumigatus virulence of a long time culture-period vs. a short time culture-period.
Best methods for fungal burden assessment and outcome evaluation in infected rodents still remains controversial (Bowman et al., 2001; Balloy et al., 2005a; Imai et al., 2005; Singh et al., 2005). Of course, the straightforward parameter for disease progression remains the overall mortality, but ethical committees currently encourage using alternative endpoints. Easy to implement for most of the labs, in vitro mycological culture is a seducing semi-quantitative approach. Practically, homogenized tissues and centrifuged fluids are serially diluted and spread on agar plates, and thereafter number of CFU is counted on each after a specified incubation time. Culture was shown to be roughly indicative of fungal burden (Graybill et al., 1983), and only of viable fungus. It does not scale linearly with hyphal burden in infected tissues. Importantly, CFU count cannot discriminate between persistent and active infective lesions in tissues. Likewise, grinding the A. fumigatus hyphae during tissue pre-processing can produce artificially-increased CFU count [(Bowman et al., 2001; Kirkpatrick et al., 2002); unlike yeasts (te Dorsthorst et al., 2005), a large fungal mass of tangled hyphae cannot be distinguished from single-cell conidial forms when cultivated (Latgé, 1999)]. At the opposite, disruption of the organs can kill viable fungus, and thus leads to underestimation (Graybill, 2000). Therefore, for an enhanced reproducibility, it appears critical to adjust the CFU count to gram body weight or fluid volume. This unit is likely more appropriate than CFU per organ or total CFU per animal. By the way, it was surprising to note that Hummel et al. and Fidan et al. attempted to perform blood cultures (Hummel et al., 2004; Fidan et al., 2008), whereas it is well-known that Aspergillus genus never grows in blood culture bottles. In order to study disease progression and diagnosis (Becker et al., 2000; Loeffler et al., 2002), detection of GM antigen and β-D-glucans is probably useful to make correlation of animal data with clinical results. Mitsutake et al. underlined that the elevation in levels of β-D-glucans increased in correlation with the elevation of GM antigen titres, and thus is reliably measurable during experimental aspergillosis (Mitsutake et al., 1995). Detection in blood may provide multiple endpoints of assessment when repeated samplings are performed, especially in bigger species (Kirkpatrick et al., 2012; White et al., 2016). GM antigen and β-D-glucans may also be tested in BAL fluids (Jambunathan et al., 2013). Nevertheless, GM and β-D-glucans measurement globally remains quite expensive, and its interpretation is still difficult in rodents, since the positive cut-off were validated only in human samples so far (Becker et al., 2000). Consequently, further studies are requested to rule on the pre-clinical use of these surrogate biomarkers, and to determine standardized interpretive values and how their diagnostic results would be best used. As an alternative indicator of fungal burden, the chitin assay was tested several times, but it is old and not indicative whether the organism present is still viable, as it allows quantification of an inactive component of the cell wall deposited within infected tissues (Lehmann and White, 1975; Bowman et al., 2001; Balloy et al., 2005a). This method is also more labor-intensive than GM and β-D-glucans testing. Very sensitive, qPCR using the 18S rRNA gene as target has been more recently applied to the determination of A. fumigatus burden in the tissues, but it requires specialized costly equipment and reagents that labs with limited resources can't afford. qPCR seems correlated to CFU count (Bowman et al., 2001; Loeffler et al., 2002; Singh et al., 2005; Lengerova et al., 2012), although some investigators reported that it is less suitable than the latter to confirm therapeutic effects of antifungal drugs. Indeed, qPCR is too much subtle to detect actual changes in fungal load, while mycological culture is able to indicate the presence of a limited amount of residual organism (Singh et al., 2005). Furthermore, qPCR cannot provide information about the viability of the fungal elements, especially because no one really knows the clearance timing of DNA from dead A. fumigatus (Vallor et al., 2008; Lengerova et al., 2012). In this context, histopathology is still considered as the reference standard to prove infection (Goldman and Osmani, 2007; Desoubeaux et al., 2014a). In addition to the observation of fungal elements, slides examination provides greatly detailed insights about the inflammation process and the extent of infection. Nonetheless, one should notice the overall lack of technical details that are provided in publications for the correct achievement of slides preparation and for a complete assessment of tissue invasion. For instance for histopathology in lungs, only a few authors thoroughly described how they processed: Becker et al. specified that every lung has to be cut at three levels ±1 mm apart. According to their recommendations, two adjacent sections were obtained at every level: one should be stained with haematoxylin and eosin and the other with Grocott-Gomori's methenamine silver (Becker et al., 2003). Baistrocchi et al. recommended to perform serial step sections of 5 μm, taken at 80 μm intervals and stained with Periodic acid-Schiff: a minimum of five sections of each lung has to be examined for all animals in each experiment in order to ensure 100 lesions were detected in the group displaying the highest level of infection (Baistrocchi et al., 2016). Additional information were also provided by Panepinto et al. when they explained how histopathological lesion areas were measured by using ScionImage® analysis software (Panepinto et al., 2003). Among the unusual miscellaneous methods for monitoring the course of infection, some investigators reported how useful could be in vivo imaging techniques based on bioluminescent A. fumigatus strain (Brock et al., 2008; Ibrahim-Granet et al., 2010; Fekkar et al., 2012; Jouvion et al., 2012), or antibody-guided positron emission tomography and magnetic resonance imaging (Rolle et al., 2016). The former requires luciferin as exogenous substrate, while the latter needs particular caution for radiation protection. Recently, the expanding availability of immunological reagents to monitor the recruitment and functional activation of immune cells informs immune-pathophysiology studies aiming to identify the molecular and cellular basis of antifungal immunity. ELISA assays measure the production of cytokines and other inflammatory mediators, while flow cytometry can quantify host leukocyte populations that reside in or are recruited to portals of infection. Interestingly, other authors made specific focus on clinical endpoint scales that are far less expensive to measure, and allow refinement and reducing euthanasia procedures: modifications of the respiratory function during pulmonary aspergillosis (Becker et al., 2006), body temperatures changes (Adamson et al., 2013), or behavior alteration and weight loss ≥20% baseline (Chandenier et al., 2009; Desoubeaux and Chandenier, 2012). All these surrogate endpoints mandated by ethic committees need to be clearly clarified and defined on quantitative terms whenever possible, to decide a correct timing for euthanasia (Morton and Griffiths, 1985; Carstens and Moberg, 2000). For example, a validated grid evaluates twice daily the discomfort level for each animal according to a scale which scores from 1 to 6 on the basis of appearance and physiological behavior changes (Becker et al., 2006; Chandenier et al., 2009; Desoubeaux and Chandenier, 2012), reaction to stimuli, and other readily-available parameters (Adamson et al., 2013): empirically as an example, score 1, no discomfort; score 2, minor discomfort; score 3, poor discomfort; score 4, serious discomfort; score 5, severe discomfort; score 6, death (Morton and Griffiths, 1985; Chandenier et al., 2009; Desoubeaux et al., 2014b).
The richness of experience accumulated over time for rodent models of invasive aspergillosis has demonstrated its utility for reliably reflecting what happens clinically in humans, when for instance addressing its immuno-pathophysiology, predicting clinical efficacy, pharmacology and toxicity of antifungal drugs, and studying diagnostic predictive value of the biomarkers. Unfortunately, it appeared in this exhaustive review that there is obviously no consensus to develop the ideal model of aspergillosis. Indeed as it is also the case for other models of infectious diseases, no single model of invasive aspergillosis is currently able to answer all questions, and each has its own limitations. Besides, we have shown that distinct rodent models can provide different—and even contradictory—results, depending on the context for which they are developed. For this reason, substantial variability from one experiment to the next has to be largely minimized in a near future, firstly by defining the minimum criteria to ensure for a reliable and reproducible support. According to us, and as it is the most consensual, usage of male mice immunocompromised by both alkylating drug and steroids, and challenged through respiratory airways in inhalation chamber, by deposition into the nares, or by a non-invasive device, seems the most relevant (Figure 4). Rats remain good alternative for tracheal inoculation. This attempt at is a preliminary critical step to tend toward standardization. It aims to subsequently allow high-quality in vivo experiments and direct comparisons of data between centers, especially since the scientific community working on A. fumigatus infection continues to grow over time.
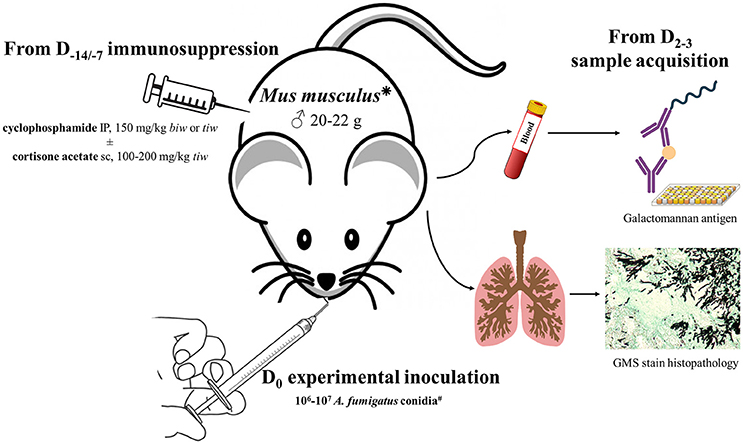
Figure 4. Summary of recommendations for further studies involving animal models of invasive aspergillosis. Suggestions for standardization are based on objective analysis of all the published literature faced with the authors' personal experience. In light of all the benefits due to their small size, their costs and the large availability of reagents dedicated to, mice should be privileged. Within a 1–2 week-long period, immunocompromised status is easily achieved by repeated injections of low-cost drugs, like alkylating agents or steroids. Thereafter, a tracheo-pulmonary challenge is recommended by non-invasive device, like MicroSprayer® aerosolizer, allowing accurate control of the fungal inoculum. In such a context, no more than 107 A. fumigatus conidia are usually needed to yield 90–100% infection rates. Generally, onset of clinical signs occurs within 48–72 h after the experimental inoculation. After that time, the challenged animals start becoming moribund from aspergillosis (ruffled fur, decreased defecation, lethargy, anorexia, weight loss, ataxia, various pulmonary signs, gross bleeding …). Although death remains the major clinical outcome, primarily in therapeutic assays, alternative endpoints may be assessed to estimate the fungal load while refining the animal welfare: nowadays, galactomannan antigen determination in blood and histopathology in lungs appear reliable and largely validated, in comparison with other surrogate biomarkers. *For therapeutic assays: outbred mouse strains like Albino Swiss Webster and CD-1; for pharmacology-pharmacokinetics and toxicology studies: outbred mouse strains like Swiss OF1 and NMRI; for immuno-pathophysiology: inbred mouse strains like C57BL/6, BALB/c, DBA2, 129/Sv, and CD2F1; for general purposes: C57BL/6 and BALB/c. #Referenced A. fumigatus strains should be privileged for inoculation, especially those that have been widely used so far, like AF293 and Dal/CEA10. Otherwise, it makes sense to use strains that were initially isolated in a relevant context of invasive aspergillosis. Environmental strains or local unreferenced strains should be avoided, because they don't allow large-scale reliable comparison. biw, Twice a week; D, Day (D0 being the date of experimental challenge); GMS, Grocott-Gomori methenamine silver staining; IP, Intraperitoneal; ♂, Male; sc, Subcutanous; tiw, Thrice a week.
Author Contributions
GD designed and performed the bibliographic analysis. GD drafted the manuscript and the figures. CC reviewed the manuscript.
Funding
This work was supported by none. The authors did not receive any specific research funding for this study.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors would like to thank Louis Calder Memorial Library, Miller School of Medicine, University of Miami, for having large access to all the articles referenced in this study.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fmicb.2017.00841/full#supplementary-material
References
Adamson, T. W., Diaz-Arevalo, D., Gonzalez, T. M., Liu, X., and Kalkum, M. (2013). Hypothermic endpoint for an intranasal invasive pulmonary aspergillosis mouse model. Comp. Med. 63, 477–481.
Ahmad, S., Al-Shaikh, A. A., and Khan, Z. (2014). Development of a novel inhalational model of invasive pulmonary aspergillosis in rats and comparative evaluation of three biomarkers for its diagnosis. PLoS ONE 9:e100524. doi: 10.1371/journal.pone.0100524
Alcazar-Fuoli, L., Buitrago, M., Gomez-Lopez, A., and Mellado, E. (2015). An alternative host model of a mixed fungal infection by azole susceptible and resistant Aspergillus spp. strains. Virulence 6, 376–384. doi: 10.1080/21505594.2015.1025192
Alsaadi, M., Italia, J. L., Mullen, A. B., Ravi Kumar, M. N. V., Candlish, A. A., Williams, R. A. M., et al. (2012). The efficacy of aerosol treatment with non-ionic surfactant vesicles containing amphotericin B in rodent models of leishmaniasis and pulmonary aspergillosis infection. J. Control. Release Off. J. Control. Release Soc. 160, 685–691. doi: 10.1016/j.jconrel.2012.04.004
Amarsaikhan, N., O'Dea, E. M., Tsoggerel, A., Owegi, H., Gillenwater, J., and Templeton, S. P. (2014). Isolate-dependent growth, virulence, and cell wall composition in the human pathogen Aspergillus fumigatus. PLoS ONE 9:e100430. doi: 10.1371/journal.pone.0100430
Andriole, V. T., Miniter, P., George, D., Kordick, D., and Patterson, T. F. (1992). Animal models: usefulness for studies of fungal pathogenesis and drug efficacy in aspergillosis. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 14(Suppl. 1), S134–S138. doi: 10.1093/clinids/14.Supplement_1.S134
Aratani, Y., Kura, F., Watanabe, H., Akagawa, H., Takano, Y., Suzuki, K., et al. (2002). Relative contributions of myeloperoxidase and NADPH-oxidase to the early host defense against pulmonary infections with Candida albicans and Aspergillus fumigatus. Med. Mycol. 40, 557–563. doi: 10.1080/mmy.40.6.557.563
Arrese, J. E., Delvenne, P., Van Cutsem, J., Piérard-Franchimont, C., and Piérard, G. E. (1994). Experimental aspergillosis in guinea pigs: influence of itraconazole on fungaemia and invasive fungal growth. Mycoses 37, 117–122. doi: 10.1111/j.1439-0507.1994.tb00786.x
Aufauvre-Brown, A., Mellado, E., Gow, N. A., and Holden, D. W. (1997). Aspergillus fumigatus chsE: a gene related to CHS3 of Saccharomyces cerevisiae and important for hyphal growth and conidiophore development but not pathogenicity. Fungal Genet. Biol. 21, 141–152. doi: 10.1006/fgbi.1997.0959
Baisakh, K. M., Agarwal, D. S., Iyengar, B., and Bhatia, V. N. (1975). Experimental aspergillosis and phycomycosis in mice. Indian J. Med. Res. 63, 1716–1731.
Baistrocchi, S. R., Lee, M. J., Lehoux, M., Ralph, B., Snarr, B. D., Robitaille, R., et al. (2016). Posaconazole-loaded leukocytes as a novel treatment strategy targeting invasive pulmonary aspergillosis. J. Infect. Dis. doi: 10.1093/infdis/jiw513. [Epub ahead of print].
Bakker-Woudenberg, I. A. J. M. (2003). Experimental models of pulmonary infection. J. Microbiol. Methods 54, 295–313. doi: 10.1016/S0167-7012(03)00118-0
Balloy, V., Huerre, M., Latgé, J.-P., and Chignard, M. (2005a). Differences in patterns of infection and inflammation for corticosteroid treatment and chemotherapy in experimental invasive pulmonary aspergillosis. Infect. Immun. 73, 494–503. doi: 10.1128/IAI.73.1.494-503.2005
Balloy, V., Si-Tahar, M., Takeuchi, O., Philippe, B., Nahori, M.-A., Tanguy, M., et al. (2005b). Involvement of toll-like receptor 2 in experimental invasive pulmonary aspergillosis. Infect. Immun. 73, 5420–5425. doi: 10.1128/IAI.73.9.5420-5425.2005
Bartroli, J., Turmo, E., Algueró, M., Boncompte, E., Vericat, M. L., Conte, L., et al. (1998). New azole antifungals. 2. Synthesis and antifungal activity of heterocyclecarboxamide derivatives of 3-amino-2-aryl-1-azolyl-2-butanol. J. Med. Chem. 41, 1855–1868.
Becker, M. J., Dams, E. T. M., de Marie, S., Oyen, W. J. G., Boerman, O. C., Fens, M. H. A. M., et al. (2002a). Scintigraphic imaging using 99mTc-labeled PEG liposomes allows early detection of experimental invasive pulmonary aspergillosis in neutropenic rats. Nucl. Med. Biol. 29, 177–184. doi: 10.1016/S0969-8051(01)00291-8
Becker, M. J., De Marie, S., Fens, M. H. A. M., Haitsma, J. J., Verbrugh, H. A., Lachmann, B., et al. (2006). Pathophysiology of unilateral pulmonary aspergillosis in an experimental rat model. Med. Mycol. Off. Publ. Int. Soc. Hum. Anim. Mycol. 44, 133–139. doi: 10.1080/13693780500271749
Becker, M. J., de Marie, S., Fens, M. H. A. M., Hop, W. C. J., Verbrugh, H. A., and Bakker-Woudenberg, I. A. J. M. (2002b). Enhanced antifungal efficacy in experimental invasive pulmonary aspergillosis by combination of AmBisome with Fungizone as assessed by several parameters of antifungal response. J. Antimicrob. Chemother. 49, 813–820.
Becker, M. J., de Marie, S., Fens, M. H. A. M., Verbrugh, H. A., and Bakker-Woudenberg, I. A. J. M. (2003). Effect of amphotericin B treatment on kinetics of cytokines and parameters of fungal load in neutropenic rats with invasive pulmonary aspergillosis. J. Antimicrob. Chemother. 52, 428–434. doi: 10.1093/jac/dkg367
Becker, M. J., de Marie, S., Willemse, D., Verbrugh, H. A., and Bakker-Woudenberg, I. A. (2000). Quantitative galactomannan detection is superior to PCR in diagnosing and monitoring invasive pulmonary aspergillosis in an experimental rat model. J. Clin. Microbiol. 38, 1434–1438.
Bellocchio, S., Gaziano, R., Bozza, S., Rossi, G., Montagnoli, C., Perruccio, K., et al. (2005). Liposomal amphotericin B activates antifungal resistance with reduced toxicity by diverting Toll-like receptor signalling from TLR-2 to TLR-4. J. Antimicrob. Chemother. 55, 214–222. doi: 10.1093/jac/dkh542
Bellocchio, S., Montagnoli, C., Bozza, S., Gaziano, R., Rossi, G., Mambula, S. S., et al. (2004a). The contribution of the Toll-like/IL-1 receptor superfamily to innate and adaptive immunity to fungal pathogens in vivo. J. Immunol. 172, 3059–3069.
Bellocchio, S., Moretti, S., Perruccio, K., Fallarino, F., Bozza, S., Montagnoli, C., et al. (2004b). TLRs govern neutrophil activity in aspergillosis. J. Immunol. 173, 7406–7415.
Ben-Ami, R., and Kontoyiannis, D. P. (2012). A nonlethal murine cutaneous model of invasive aspergillosis. Methods Mol. Biol. 845, 569–582. doi: 10.1007/978-1-61779-539-8_42
Ben-Ami, R., Lewis, R. E., Leventakos, K., and Kontoyiannis, D. P. (2009). Aspergillus fumigatus inhibits angiogenesis through the production of gliotoxin and other secondary metabolites. Blood 114, 5393–5399. doi: 10.1182/blood-2009-07-231209
Ben-Ami, R., Lewis, R. E., Leventakos, K., Latgé, J.-P., and Kontoyiannis, D. P. (2010a). Cutaneous model of invasive aspergillosis. Antimicrob. Agents Chemother. 54, 1848–1854. doi: 10.1128/AAC.01504-09
Ben-Ami, R., Varga, V., Lewis, R. E., May, G. S., Nierman, W. C., and Kontoyiannis, D. P. (2010b). Characterization of a 5-azacytidine-induced developmental Aspergillus fumigatus variant. Virulence 1, 164–173. doi: 10.4161/viru.1.3.11750
Benjamim, C. F., Hogaboam, C. M., Lukacs, N. W., and Kunkel, S. L. (2003). Septic mice are susceptible to pulmonary aspergillosis. Am. J. Pathol. 163, 2605–2617. doi: 10.1016/S0002-9440(10)63615-2
Benjamim, C. F., Lundy, S. K., Lukacs, N. W., Hogaboam, C. M., and Kunkel, S. L. (2005). Reversal of long-term sepsis-induced immunosuppression by dendritic cells. Blood 105, 3588–3595. doi: 10.1182/blood-2004-08-3251
Berenguer, J., Allende, M. C., Lee, J. W., Garrett, K., Lyman, C., Ali, N. M., et al. (1995). Pathogenesis of pulmonary aspergillosis. Granulocytopenia versus cyclosporine and methylprednisolone-induced immunosuppression. Am. J. Respir. Crit. Care Med. 152, 1079–1086. doi: 10.1164/ajrccm.152.3.7663787
Bitar, D., Lortholary, O., Le Strat, Y., Nicolau, J., Coignard, B., Tattevin, P., et al. (2014). Population-based analysis of invasive fungal infections, France, 2001-2010. Emerging Infect. Dis. 20, 1149–1155. doi: 10.3201/eid2007.140087
BitMansour, A., and Brown, J. M. Y. (2002). Prophylactic administration of liposomal amphotericin B is superior to treatment in a murine model of invasive aspergillosis after hematopoietic cell transplantation. J. Infect. Dis. 186, 134–137. doi: 10.1086/341294
BitMansour, A., Cao, T. M., Chao, S., Shashidhar, S., and Brown, J. M. Y. (2005). Single infusion of myeloid progenitors reduces death from Aspergillus fumigatus following chemotherapy-induced neutropenia. Blood 105, 3535–3537. doi: 10.1182/blood-2004-07-2676
Björgvinsdóttir, H., Ding, C., Pech, N., Gifford, M. A., Li, L. L., and Dinauer, M. C. (1997). Retroviral-mediated gene transfer of gp91phox into bone marrow cells rescues defect in host defense against Aspergillus fumigatus in murine X-linked chronic granulomatous disease. Blood 89, 41–48.
Boase, S., Valentine, R., Singhal, D., Tan, L. W., and Wormald, P.-J. (2011). A sheep model to investigate the role of fungal biofilms in sinusitis: fungal and bacterial synergy. Int. Forum Allergy Rhinol. 1, 340–347. doi: 10.1002/alr.20066
Bok, J. W., Balajee, S. A., Marr, K. A., Andes, D., Nielsen, K. F., Frisvad, J. C., et al. (2005). LaeA, a regulator of morphogenetic fungal virulence factors. Eukaryotic Cell 4, 1574–1582. doi: 10.1128/EC.4.9.1574-1582.2005
Bok, J. W., Chung, D., Balajee, S. A., Marr, K. A., Andes, D., Nielsen, K. F., et al. (2006). GliZ, a transcriptional regulator of gliotoxin biosynthesis, contributes to Aspergillus fumigatus virulence. Infect. Immun. 74, 6761–6768. doi: 10.1128/IAI.00780-06
Bonifazi, P., D'Angelo, C., Zagarella, S., Zelante, T., Bozza, S., De Luca, A., et al. (2010). Intranasally delivered siRNA targeting PI3K/Akt/mTOR inflammatory pathways protects from aspergillosis. Mucosal Immunol. 3, 193–205. doi: 10.1038/mi.2009.130
Bonnett, C. R., Cornish, E. J., Harmsen, A. G., and Burritt, J. B. (2006). Early neutrophil recruitment and aggregation in the murine lung inhibit germination of Aspergillus fumigatus conidia. Infect. Immun. 74, 6528–6539. doi: 10.1128/IAI.00909-06
Bowman, J. C., Abruzzo, G. K., Anderson, J. W., Flattery, A. M., Gill, C. J., Pikounis, V. B., et al. (2001). Quantitative PCR assay to measure Aspergillus fumigatus burden in a murine model of disseminated aspergillosis: demonstration of efficacy of caspofungin acetate. Antimicrob. Agents Chemother. 45, 3474–3481. doi: 10.1128/AAC.45.12.3474-3481.2001
Bozza, S., Campo, S., Arseni, B., Inforzato, A., Ragnar, L., Bottazzi, B., et al. (2014). PTX3 binds MD-2 and promotes TRIF-dependent immune protection in aspergillosis. J. Immunol. 193, 2340–2348. doi: 10.4049/jimmunol.1400814
Bozza, S., Gaziano, R., Lipford, G. B., Montagnoli, C., Bacci, A., Di Francesco, P., et al. (2002a). Vaccination of mice against invasive aspergillosis with recombinant Aspergillus proteins and CpG oligodeoxynucleotides as adjuvants. Microbes Infect. Inst. Pasteur 4, 1281–1290.
Bozza, S., Gaziano, R., Spreca, A., Bacci, A., Montagnoli, C., di Francesco, P., et al. (2002b). Dendritic cells transport conidia and hyphae of Aspergillus fumigatus from the airways to the draining lymph nodes and initiate disparate Th responses to the fungus. J. Immunol. 168, 1362–1371.
Bozza, S., Perruccio, K., Montagnoli, C., Gaziano, R., Bellocchio, S., Burchielli, E., et al. (2003). A dendritic cell vaccine against invasive aspergillosis in allogeneic hematopoietic transplantation. Blood 102, 3807–3814. doi: 10.1182/blood-2003-03-0748
Bretz, C., Gersuk, G., Knoblaugh, S., Chaudhary, N., Randolph-Habecker, J., Hackman, R. C., et al. (2008). MyD88 signaling contributes to early pulmonary responses to Aspergillus fumigatus. Infect. Immun. 76, 952–958. doi: 10.1128/IAI.00927-07
Brieland, J. K., Jackson, C., Menzel, F., Loebenberg, D., Cacciapuoti, A., Halpern, J., et al. (2001). Cytokine networking in lungs of immunocompetent mice in response to inhaled Aspergillus fumigatus. Infect. Immun. 69, 1554–1560. doi: 10.1128/IAI.69.3.1554-1560.2001
Brock, M., Jouvion, G., Droin-Bergère, S., Dussurget, O., Nicola, M.-A., and Ibrahim-Granet, O. (2008). Bioluminescent Aspergillus fumigatus, a new tool for drug efficiency testing and in vivo monitoring of invasive aspergillosis. Appl. Environ. Microbiol. 74, 7023–7035. doi: 10.1128/AEM.01288-08
Brown, G. D. (2011). Innate antifungal immunity: the key role of phagocytes. Annu. Rev. Immunol. 29, 1–21. doi: 10.1146/annurev-immunol-030409-101229
Brown, J. S., Aufauvre-Brown, A., Brown, J., Jennings, J. M., Arst, H., and Holden, D. W. (2000). Signature-tagged and directed mutagenesis identify PABA synthetase as essential for Aspergillus fumigatus pathogenicity. Mol. Microbiol. 36, 1371–1380. doi: 10.1046/j.1365-2958.2000.01953.x
Buskirk, A. D., Green, B. J., Lemons, A. R., Nayak, A. P., Goldsmith, W. T., Kashon, M. L., et al. (2014). A murine inhalation model to characterize pulmonary exposure to dry Aspergillus fumigatus conidia. PLoS ONE 9:e109855. doi: 10.1371/journal.pone.0109855
Cacciapuoti, A., Halpern, J., Mendrick, C., Norris, C., Patel, R., and Loebenberg, D. (2006). Interaction between posaconazole and caspofungin in concomitant treatment of mice with systemic Aspergillus infection. Antimicrob. Agents Chemother. 50, 2587–2590. doi: 10.1128/AAC.00829-05
Caffrey, A. K., Lehmann, M. M., Zickovich, J. M., Espinosa, V., Shepardson, K. M., Watschke, C. P., et al. (2015). IL-1α signaling is critical for leukocyte recruitment after pulmonary Aspergillus fumigatus challenge. PLoS Pathog. 11:e1004625. doi: 10.1371/journal.ppat.1004625
Capilla, J., and Guarro, J. (2004). Correlation between in vitro susceptibility of Scedosporium apiospermum to voriconazole and in vivo outcome of scedosporiosis in guinea pigs. Antimicrob. Agents Chemother. 48, 4009–4011. doi: 10.1128/AAC.48.10.4009-4011.2004
Capilla, J., Clemons, K. V., and Stevens, D. A. (2007). Animal models: an important tool in mycology. Med. Mycol. 45, 657–684. doi: 10.1080/13693780701644140
Carrion, S. d. J., Leal, S. M. Jr., Ghannoum, M. A., Aimanianda, V., Latgé, J.-P., and Pearlman, E. (2013). The RodA hydrophobin on Aspergillus fumigatus spores masks dectin-1- and dectin-2-dependent responses and enhances fungal survival in vivo. J. Immunol. 191, 2581–2588. doi: 10.4049/jimmunol.1300748
Carstens, E., and Moberg, G. P. (2000). Recognizing pain and distress in laboratory animals. ILAR J. 41, 62–71. doi: 10.1093/ilar.41.2.62
Carvalho, A., De Luca, A., Bozza, S., Cunha, C., D'Angelo, C., Moretti, S., et al. (2012). TLR3 essentially promotes protective class I-restricted memory CD8+ T-cell responses to Aspergillus fumigatus in hematopoietic transplanted patients. Blood 119, 967–977. doi: 10.1182/blood-2011-06-362582
Cenci, E., Mencacci, A., Bacci, A., Bistoni, F., Kurup, V. P., and Romani, L. (2000). T cell vaccination in mice with invasive pulmonary aspergillosis. J. Immunol. 165, 381–388. doi: 10.4049/jimmunol.165.1.381
Cenci, E., Mencacci, A., Casagrande, A., Mosci, P., Bistoni, F., and Romani, L. (2001). Impaired antifungal effector activity but not inflammatory cell recruitment in interleukin-6-deficient mice with invasive pulmonary aspergillosis. J. Infect. Dis. 184, 610–617. doi: 10.1086/322793
Cenci, E., Mencacci, A., Del Sero, G., Bacci, A., Montagnoli, C., d'Ostiani, C. F., et al. (1999). Interleukin-4 causes susceptibility to invasive pulmonary aspergillosis through suppression of protective type I responses. J. Infect. Dis. 180, 1957–1968.
Cenci, E., Mencacci, A., Fè d'Ostiani, C., Del Sero, G., Mosci, P., Montagnoli, C., et al. (1998). Cytokine- and T helper-dependent lung mucosal immunity in mice with invasive pulmonary aspergillosis. J. Infect. Dis. 178, 1750–1760.
Cenci, E., Mencacci, A., Spreca, A., Montagnoli, C., Bacci, A., Perruccio, K., et al. (2002). Protection of killer antiidiotypic antibodies against early invasive aspergillosis in a murine model of allogeneic T-cell-depleted bone marrow transplantation. Infect. Immun. 70, 2375–2382. doi: 10.1128/IAI.70.5.2375-2382.2002
Cenci, E., Perito, S., Enssle, K. H., Mosci, P., Latgé, J. P., Romani, L., et al. (1997). Th1 and Th2 cytokines in mice with invasive aspergillosis. Infect. Immun. 65, 564–570.
Centeno-Lima, S., Silveira, H., Casimiro, C., Aguiar, P., and do Rosário, V. E. (2002). Kinetics of cytokine expression in mice with invasive aspergillosis: lethal infection and protection. FEMS Immunol. Med. Microbiol. 32, 167–173. doi: 10.1111/j.1574-695X.2002.tb00549.x
Chamilos, G., Bignell, E. M., Schrettl, M., Lewis, R. E., Leventakos, K., May, G. S., et al. (2010). Exploring the concordance of Aspergillus fumigatus pathogenicity in mice and Toll-deficient flies. Med. Mycol. 48, 506–510. doi: 10.3109/13693780903225813
Chandenier, J., Bernard, S., Montharu, J., Bailly, E., Fetissof, F., de Monte, M., et al. (2009). The utility of a nebulised intra-tracheal rat model of invasive pulmonary aspergillosis. Mycoses 52, 239–245. doi: 10.1111/j.1439-0507.2009.01695.x
Chandrasekar, P. H., Cutright, J. L., and Manavathu, E. K. (2004). Efficacy of voriconazole plus amphotericin B or micafungin in a guinea-pig model of invasive pulmonary aspergillosis. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 10, 925–928. doi: 10.1111/j.1469-0691.2004.00958.x
Chandrasekar, P. H., Cutright, J., and Manavathu, E. (2000). Efficacy of voriconazole against invasive pulmonary aspergillosis in a guinea-pig model. J. Antimicrob. Chemother. 45, 673–676. doi: 10.1093/jac/45.5.673
Chang, Y. C., Tsai, H.-F., Karos, M., and Kwon-Chung, K. J. (2004). THTA, a thermotolerance gene of Aspergillus fumigatus. Fungal Genet. Biol. 41, 888–896. doi: 10.1016/j.fgb.2004.06.004
Chaturvedi, A. K., Kavishwar, A., Shiva Keshava, G. B., and Shukla, P. K. (2005). Monoclonal immunoglobulin G1 directed against Aspergillus fumigatus cell wall glycoprotein protects against experimental murine aspergillosis. Clin. Diagn. Lab. Immunol. 12, 1063–1068. doi: 10.1128/cdli.12.9.1063-1068.2005
Chaudhary, B., and Singh, B. (1983). Pathogenicity of Aspergillus fumigatus in chicks, guinea fowl, rabbits and mice. Mykosen 26, 421–429. doi: 10.1111/j.1439-0507.1983.tb03230.x
Chaudhary, S. K., Sadana, J. R., and Pruthi, A. K. (1988). Sequential pathological studies in Japanese quails infected experimentally with Aspergillus fumigatus. Mycopathologia 103, 157–166. doi: 10.1007/BF00436815
Cheema, M. S., and Christians, J. K. (2011). Virulence in an insect model differs between mating types in Aspergillus fumigatus. Med. Mycol. 49, 202–207. doi: 10.3109/13693786.2010.512301
Cheers, C., and Waller, R. (1975). Activated macrophages in congenitally athymic “nude mice” and in lethally irradiate mice. J. Immunol. 115, 844–847.
Chia, R., Achilli, F., Festing, M. F. W., and Fisher, E. M. C. (2005). The origins and uses of mouse outbred stocks. Nat. Genet. 37, 1181–1186. doi: 10.1038/ng1665
Chiang, L. Y., Ejzykowicz, D. E., Tian, Z.-Q., Katz, L., and Filler, S. G. (2006). Efficacy of ambruticin analogs in a murine model of invasive pulmonary aspergillosis. Antimicrob. Agents Chemother. 50, 3464–3466. doi: 10.1128/AAC.00558-06
Chiang, L. Y., Sheppard, D. C., Gravelat, F. N., Patterson, T. F., and Filler, S. G. (2008). Aspergillus fumigatus stimulates leukocyte adhesion molecules and cytokine production by endothelial cells in vitro and during invasive pulmonary disease. Infect. Immun. 76, 3429–3438. doi: 10.1128/IAI.01510-07
Chiller, T. M., Luque, J. C., Sobel, R. A., Farrokhshad, K., Clemons, K. V., and Stevens, D. A. (2002). Development of a murine model of cerebral aspergillosis. J. Infect. Dis. 186, 574–577. doi: 10.1086/341567
Chiller, T. M., Sobel, R. A., Luque, J. C., Clemons, K. V., and Stevens, D. A. (2003). Efficacy of amphotericin B or itraconazole in a murine model of central nervous system Aspergillus infection. Antimicrob. Agents Chemother. 47, 813–815. doi: 10.1128/AAC.47.2.813-815.2003
Chilvers, E. R., Spreadbury, C. L., and Cohen, J. (1989). Bronchoalveolar lavage in an immunosuppressed rabbit model of invasive pulmonary aspergillosis. Mycopathologia 108, 163–171. doi: 10.1007/BF00436221
Cicogna, C. E., White, M. H., Bernard, E. M., Ishimura, T., Sun, M., Tong, W. P., et al. (1997). Efficacy of prophylactic aerosol amphotericin B lipid complex in a rat model of pulmonary aspergillosis. Antimicrob. Agents Chemother. 41, 259–261.
Clark, J. M., Whitney, R. R., Olsen, S. J., George, R. J., Swerdel, M. R., Kunselman, L., et al. (1991). Amphotericin B lipid complex therapy of experimental fungal infections in mice. Antimicrob. Agents Chemother. 35, 615–621. doi: 10.1128/AAC.35.4.615
Clemons, K. V., and Stevens, D. A. (2005). The contribution of animal models of aspergillosis to understanding pathogenesis, therapy and virulence. Med. Mycol. Off. Publ. Int. Soc. Hum. Anim. Mycol. 43(Suppl. 1), S101–S110. doi: 10.1080/13693780500051919
Clemons, K. V., and Stevens, D. A. (2006a). Animal models of Aspergillus infection in preclinical trials, diagnostics and pharmacodynamics: what can we learn from them? Med. Mycol. 44, S119–S126. doi: 10.1080/13693780600871174
Clemons, K. V., and Stevens, D. A. (2006b). Animal models testing monotherapy versus combination antifungal therapy: lessons learned and future directions. Curr. Opin. Infect. Dis. 19, 360–364. doi: 10.1097/01.qco.0000235163.70678.59
Clemons, K. V., and Stevens, D. A. (2006c). Efficacy of micafungin alone or in combination against experimental pulmonary aspergillosis. Med. Mycol. 44, 69–73.
Clemons, K. V., Espiritu, M., Parmar, R., and Stevens, D. A. (2005). Comparative efficacies of conventional amphotericin b, liposomal amphotericin B (AmBisome), caspofungin, micafungin, and voriconazole alone and in combination against experimental murine central nervous system aspergillosis. Antimicrob. Agents Chemother. 49, 4867–4875. doi: 10.1128/AAC.49.12.4867-4875.2005
Clemons, K. V., Gonzalez, G. M., Singh, G., Imai, J., Espiritu, M., Parmar, R., et al. (2006a). Development of an orogastrointestinal mucosal model of candidiasis with dissemination to visceral organs. Antimicrob. Agents Chemother. 50, 2650–2657. doi: 10.1128/AAC.00530-06
Clemons, K. V., Grunig, G., Sobel, R. A., Mirels, L. F., Rennick, D. M., and Stevens, D. A. (2000a). Role of IL-10 in invasive aspergillosis: increased resistance of IL-10 gene knockout mice to lethal systemic aspergillosis. Clin. Exp. Immunol. 122, 186–191.
Clemons, K. V., Martinez, M., Chen, V., Liu, M., Yoon, H. J., and Stevens, D. A. (2014). Protection against experimental aspergillosis by heat-killed yeast is not antibody dependent. Med. Mycol. 52, 422–426. doi: 10.1093/mmy/myt015
Clemons, K. V., Miller, T. K., Selitrennikoff, C. P., and Stevens, D. A. (2002). fos-1, a putative histidine kinase as a virulence factor for systemic aspergillosis. Med. Mycol. 40, 259–262. doi: 10.1080/mmy.40.3.259.262
Clemons, K. V., Parmar, R., Martinez, M., and Stevens, D. A. (2006b). Efficacy of Abelcet alone, or in combination therapy, against experimental central nervous system aspergillosis. J. Antimicrob. Chemother. 58, 466–469. doi: 10.1093/jac/dkl236
Clemons, K. V., Sobel, R. A., and Stevens, D. A. (2000b). Toxicity of LY303366, an echinocandin antifungal, in mice pretreated with glucocorticoids. Antimicrob. Agents Chemother. 44, 378–381.
Corbel, M. J., and Eades, S. M. (1977). Examination of the effect of age and acquired immunity on the susceptibility of mice to infection with Aspergillus fumigatus. Mycopathologia 60, 79–85. doi: 10.1007/BF00490376
Corbel, M. J., Pepin, G. A., and Millar, P. G. (1973). The serological response to Aspergillus fumigatus in experimental mycotic abortion in sheep. J. Med. Microbiol. 6, 539–548. doi: 10.1099/00222615-6-4-539
Cornish, E. J., Hurtgen, B. J., McInnerney, K., Burritt, N. L., Taylor, R. M., Jarvis, J. N., et al. (2008). Reduced nicotinamide adenine dinucleotide phosphate oxidase-independent resistance to Aspergillus fumigatus in alveolar macrophages. J. Immunol. 180, 6854–6867. doi: 10.4049/jimmunol.180.10.6854
Cramer, R. A., Gamcsik, M. P., Brooking, R. M., Najvar, L. K., Kirkpatrick, W. R., Patterson, T. F., et al. (2006). Disruption of a nonribosomal peptide synthetase in Aspergillus fumigatus eliminates gliotoxin production. Eukaryotic Cell 5, 972–980. doi: 10.1128/EC.00049-06
Cramer, R. A., Perfect, B. Z., Pinchai, N., Park, S., Perlin, D. S., Asfaw, Y. G., et al. (2008). Calcineurin target CrzA regulates conidial germination, hyphal growth, and pathogenesis of Aspergillus fumigatus. Eukaryotic Cell 7, 1085–1097. doi: 10.1128/EC.00086-08
Cruz, C. R. Y., Lam, S., Hanley, P. J., Bear, A. S., Langston, C., Cohen, A. J., et al. (2013). Robust T cell responses to aspergillosis in chronic granulomatous disease: implications for immunotherapy. Clin. Exp. Immunol. 174, 89–96. doi: 10.1111/cei.12156
Cunha, B. A., Comer, J. B., and Jonas, M. (1982). The tetracyclines. Med. Clin. North Am. 66, 293–302. doi: 10.1016/S0025-7125(16)31461-4
Cutsem, J. V., Meulemans, L., Gerven, F. V., and Stynen, D. (1993). Effect of tissue invasion and treatment with itraconazole or amphotericin B on galactomannan levels in plasma of guinea-pigs with experimental invasive aspergillosis. J. Med. Vet. Mycol. 31, 315–324. doi: 10.1080/02681219380000381
Dagenais, T. R. T., Chung, D., Giles, S. S., Hull, C. M., Andes, D., and Keller, N. P. (2008). Defects in conidiophore development and conidium-macrophage interactions in a dioxygenase mutant of Aspergillus fumigatus. Infect. Immun. 76, 3214–3220. doi: 10.1128/IAI.00009-08
D'Angelo, C., De Luca, A., Zelante, T., Bonifazi, P., Moretti, S., Giovannini, G., et al. (2009). Exogenous pentraxin 3 restores antifungal resistance and restrains inflammation in murine chronic granulomatous disease. J. Immunol. 183, 4609–4618. doi: 10.4049/jimmunol.0900345
Dams, E. T., Becker, M. J., Oyen, W. J., Boerman, O. C., Storm, G., Laverman, P., et al. (1999). Scintigraphic imaging of bacterial and fungal infection in granulocytopenic rats. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 40, 2066–2072.
Dannaoui, E., Borel, E., Persat, F., Monier, M. F., and Piens, M. A. (1999). In-vivo itraconazole resistance of Aspergillus fumigatus in systemic murine aspergillosis. EBGA Network. European research group on Biotypes and Genotypes of Aspergillus fumigatus. J. Med. Microbiol. 48, 1087–1093. doi: 10.1099/00222615-48-12-1087
de Repentigny, L., Petitbois, S., Boushira, M., Michaliszyn, E., Sénéchal, S., Gendron, N., et al. (1993). Acquired immunity in experimental murine aspergillosis is mediated by macrophages. Infect. Immun. 61, 3791–3802.
Deepe, G. S., Romani, L., Calich, V. L., Huffnagle, G., Arruda, C., Molinari-Madlum, E. E., et al. (2000). Knockout mice as experimental models of virulence. Med. Mycol. 38(Suppl. 1), 87–98. doi: 10.1080/744118735
Del Sero, G., Mencacci, A., Cenci, E., d'Ostiani, C. F., Montagnoli, C., Bacci, A., et al. (1999). Antifungal type 1 responses are upregulated in IL-10-deficient mice. Microbes Infect. Inst. Pasteur 1, 1169–1180.
Denning, D. W., and Warn, P. (1999). Dose range evaluation of liposomal nystatin and comparisons with amphotericin B and amphotericin B lipid complex in temporarily neutropenic mice infected with an isolate of Aspergillus fumigatus with reduced susceptibility to amphotericin B. Antimicrob. Agents Chemother. 43, 2592–2599.
Denning, D. W., Hall, L., Jackson, M., and Hollis, S. (1995). Efficacy of D0870 compared with those of itraconazole and amphotericin B in two murine models of invasive aspergillosis. Antimicrob. Agents Chemother. 39, 1809–1814. doi: 10.1128/AAC.39.8.1809
Denning, D. W., Radford, S. A., Oakley, K. L., Hall, L., Johnson, E. M., and Warnock, D. W. (1997a). Correlation between in-vitro susceptibility testing to itraconazole and in-vivo outcome of Aspergillus fumigatus infection. J. Antimicrob. Chemother. 40, 401–414.
Denning, D. W., Venkateswarlu, K., Oakley, K. L., Anderson, M. J., Manning, N. J., Stevens, D. A., et al. (1997b). Itraconazole resistance in Aspergillus fumigatus. Antimicrob. Agents Chemother. 41, 1364–1368.
Dennis, C. G., Greco, W. R., Brun, Y., Youn, R., Slocum, H. K., Bernacki, R. J., et al. (2006). Effect of amphotericin B and micafungin combination on survival, histopathology, and fungal burden in experimental aspergillosis in the p47phox-/- mouse model of chronic granulomatous disease. Antimicrob. Agents Chemother. 50, 422–427. doi: 10.1128/AAC.50.2.422-427.2006
Desoubeaux, G., and Chandenier, J. (2012). A nebulized intra-tracheal rat model of invasive pulmonary aspergillosis. Methods Mol. Biol. 845, 511–518. doi: 10.1007/978-1-61779-539-8_36
Desoubeaux, G., Bailly, É., and Chandenier, J. (2014a). Diagnosis of invasive pulmonary aspergillosis: updates and recommendations. Méd. Mal. Infect. 44, 89–101.
Desoubeaux, G., Jourdan, M.-L., Valera, L., Jardin, B., Hem, S., Caille, A., et al. (2014b). Proteomic demonstration of the recurrent presence of inter-alpha-inhibitor H4 heavy-chain during aspergillosis induced in an animal model. Int. J. Med. Microbiol. 304, 327–338. doi: 10.1016/j.ijmm.2013.11.015
Dinamarco, T. M., Almeida, R. S., de Castro, P. A., Brown, N. A., dos Reis, T. F., Ramalho, L. N. Z., et al. (2012a). Molecular characterization of the putative transcription factor SebA involved in virulence in Aspergillus fumigatus. Eukaryot. Cell 11, 518–531.
Dinamarco, T. M., Freitas, F. Z., Almeida, R. S., Brown, N. A., dos Reis, T. F., Ramalho, L. N. Z., et al. (2012b). Functional characterization of an Aspergillus fumigatus calcium transporter (PmcA) that is essential for fungal infection. PLoS ONE 7:e37591. doi: 10.1371/journal.pone.0037591
Dirr, F., Echtenacher, B., Heesemann, J., Hoffmann, P., Ebel, F., and Wagener, J. (2010). AfMkk2 is required for cell wall integrity signaling, adhesion, and full virulence of the human pathogen Aspergillus fumigatus. Int. J. Med. Microbiol. 300, 496–502. doi: 10.1016/j.ijmm.2010.03.001
Dixon, D. M. (1987). In vivo models: evaluating antifungal agents. Methods Find. Exp. Clin. Pharmacol. 9, 729–738.
Dixon, D. M., Polak, A., and Walsh, T. J. (1989). Fungus dose-dependent primary pulmonary aspergillosis in immunosuppressed mice. Infect. Immun. 57, 1452–1456.
Donat, S., Hasenberg, M., Schäfer, T., Ohlsen, K., Gunzer, M., Einsele, H., et al. (2012). Surface display of Gaussia princeps luciferase allows sensitive fungal pathogen detection during cutaneous aspergillosis. Virulence 3, 51–61. doi: 10.4161/viru.3.1.18799
Drew, R. H., Townsend, M. L., Pound, M. W., Johnson, S. W., and Perfect, J. R. (2013). Recent advances in the treatment of life-threatening, invasive fungal infections. Expert Opin. Pharmacother. 14, 2361–2374. doi: 10.1517/14656566.2013.838217
Du, C., Li, R., Ma, S., and Wang, D. (2002). Cloning of Aspergillus fumigatus histidine kinase gene fragment and its expression during invasive infection. Mycopathologia 153, 5–10. doi: 10.1023/A:1015294930331
Dufresne, S. F., Datta, K., Li, X., Dadachova, E., Staab, J. F., Patterson, T. F., et al. (2012). Detection of urinary excreted fungal galactomannan-like antigens for diagnosis of invasive aspergillosis. PLoS ONE 7:e42736. doi: 10.1371/journal.pone.0042736
Duong, M., Ouellet, N., Simard, M., Bergeron, Y., Olivier, M., and Bergeron, M. G. (1998). Kinetic study of host defense and inflammatory response to Aspergillus fumigatus in steroid-induced immunosuppressed mice. J. Infect. Dis. 178, 1472–1482. doi: 10.1086/314425
Durrant, C., Tayem, H., Yalcin, B., Cleak, J., Goodstadt, L., de Villena, F. P.-M., et al. (2011). Collaborative Cross mice and their power to map host susceptibility to Aspergillus fumigatus infection. Genome Res. 21, 1239–1248. doi: 10.1101/gr.118786.110
Espinosa, V., Jhingran, A., Dutta, O., Kasahara, S., Donnelly, R., Du, P., et al. (2014). Inflammatory monocytes orchestrate innate antifungal immunity in the lung. PLoS Pathog. 10:e1003940. doi: 10.1371/journal.ppat.1003940
Evans, S. E., Scott, B. L., Clement, C. G., Larson, D. T., Kontoyiannis, D., Lewis, R. E., et al. (2010a). Stimulated innate resistance of lung epithelium protects mice broadly against bacteria and fungi. Am. J. Respir. Cell Mol. Biol. 42, 40–50.
Evans, S. E., Tuvim, M. J., Zhang, J., Larson, D. T., García, C. D., Martinez-Pro, S., et al. (2010b). Host lung gene expression patterns predict infectious etiology in a mouse model of pneumonia. Respir. Res. 11:101. doi: 10.1186/1465-9921-11-101
Faro-Trindade, I., Willment, J. A., Kerrigan, A. M., Redelinghuys, P., Hadebe, S., Reid, D. M., et al. (2012). Characterisation of innate fungal recognition in the lung. PLoS ONE 7:e35675. doi: 10.1371/journal.pone.0035675
Fei, M., Bhatia, S., Oriss, T. B., Yarlagadda, M., Khare, A., Akira, S., et al. (2011). TNF-alpha from inflammatory dendritic cells (DCs) regulates lung IL-17A/IL-5 levels and neutrophilia versus eosinophilia during persistent fungal infection. Proc. Natl. Acad. Sci. U.S.A. 108, 5360–5365. doi: 10.1073/pnas.1015476108
Fekkar, A., Pionneau, C., Brossas, J. Y., Marinach-Patrice, C., Snounou, G., Brock, M., et al. (2012). DIGE enables the detection of a putative serum biomarker of fungal origin in a mouse model of invasive aspergillosis. J. Proteomics 75, 2536–2549. doi: 10.1016/j.jprot.2012.01.040
Festing, M. F. W. (2010). Inbred strains should replace outbred stocks in toxicology, safety testing, and drug development. Toxicol. Pathol. 38, 681–690. doi: 10.1177/0192623310373776
Fidan, I., Kalkanci, A., Bolat, S., Yesilyurt, E., Erdal, B., Yolbakan, S., et al. (2008). Expression of the surface antigens of lymphocytes and the levels of cytokines in mice infected with Aspergillus fumigatus. J. Infect. Dev. Ctries. 2, 34–39. doi: 10.3855/jidc.319
Ford, S., and Friedman, L. (1967). Experimental study of the pathogenicity of aspergilli for mice. J. Bacteriol. 94, 928–933.
Frosco, M. B., Chase, T., and Macmillan, J. D. (1994). The effect of elastase-specific monoclonal and polyclonal antibodies on the virulence of Aspergillus fumigatus in immunocompromised mice. Mycopathologia 125, 65–76. doi: 10.1007/BF01371095
Galiger, C., Brock, M., Jouvion, G., Savers, A., Parlato, M., and Ibrahim-Granet, O. (2013). Assessment of efficacy of antifungals against Aspergillus fumigatus: value of real-time bioluminescence imaging. Antimicrob. Agents Chemother. 57, 3046–3059. doi: 10.1128/AAC.01660-12
Gao, J. L., Wynn, T. A., Chang, Y., Lee, E. J., Broxmeyer, H. E., Cooper, S., et al. (1997). Impaired host defense, hematopoiesis, granulomatous inflammation and type 1-type 2 cytokine balance in mice lacking CC chemokine receptor 1. J. Exp. Med. 185, 1959–1968. doi: 10.1084/jem.185.11.1959
García, M. E., Caballero, J., Blanco, I., Cruzado, M., Costas, E., and Blanco, J. L. (2006). Changes in the elastase activity and colonization ability of Aspergillus fumigatus after successive inoculations in mice. Rev. Iberoam. Micol. 23, 221–223. doi: 10.1016/S1130-1406(06)70048-1
Garlanda, C., Hirsch, E., Bozza, S., Salustri, A., De Acetis, M., Nota, R., et al. (2002). Non-redundant role of the long pentraxin PTX3 in anti-fungal innate immune response. Nature 420, 182–186. doi: 10.1038/nature01195
Gavaldà, J., Martín, M.-T., López, P., Gomis, X., Ramírez, J.-L., Rodríguez, D., et al. (2005). Efficacy of nebulized liposomal amphotericin B in treatment of experimental pulmonary aspergillosis. Antimicrob. Agents Chemother. 49, 3028–3030. doi: 10.1128/AAC.49.7.3028-3030.2005
Gaziano, R., Bozza, S., Bellocchio, S., Perruccio, K., Montagnoli, C., Pitzurra, L., et al. (2004). Anti-Aspergillus fumigatus efficacy of pentraxin 3 alone and in combination with antifungals. Antimicrob. Agents Chemother. 48, 4414–4421. doi: 10.1128/AAC.48.11.4414-4421.2004
Ghori, H. M., and Edgar, S. A. (1979). Comparative susceptibility and effect of mild Aspergillus fumigatus infection on three strains of chickens. Poult. Sci. 58, 14–17. doi: 10.3382/ps.0580014
Ghosh, S. K., Mehta, P. K., Patel, J. G., Kashyap, S. K., and Chatterjee, S. K. (1977). Histochemical changes in adrenals of Swiss mice on intraperitoneal administration of Aspergillus spores. Cell. Mol. Biol. Cyto Enzymol. 22, 65–71.
Giacomotto, J., and Ségalat, L. (2010). High-throughput screening and small animal models, where are we? Br. J. Pharmacol. 160, 204–216. doi: 10.1111/j.1476-5381.2010.00725.x
Goebel, W. S., Mark, L. A., Billings, S. D., Meyers, J. L., Pech, N., Travers, J. B., et al. (2005). Gene correction reduces cutaneous inflammation and granuloma formation in murine X-linked chronic granulomatous disease. J. Invest. Dermatol. 125, 705–710. doi: 10.1111/j.0022-202X.2005.23908.x
Golden, J. W., Hammerbeck, C. D., Mucker, E. M., and Brocato, R. L. (2015). Animal models for the study of rodent-borne hemorrhagic fever viruses: arenaviruses and hantaviruses. Biomed. Res. Int. 2015:793257. doi: 10.1155/2015/793257
Goldman, G. H., and Osmani, S. A. (2007). The Aspergilli: Genomics, Medical Aspects, Biotechnology, and Research Methods. Boca Raton, FL: CRC Press. doi: 10.1201/9781420008517
Gomez-Lopez, A., Forastiero, A., Cendejas-Bueno, E., Gregson, L., Mellado, E., Howard, S. J., et al. (2014). An invertebrate model to evaluate virulence in Aspergillus fumigatus: the role of azole resistance. Med. Mycol. 52, 311–319. doi: 10.1093/mmy/myt022
Grahl, N., Dinamarco, T. M., Willger, S. D., Goldman, G. H., and Cramer, R. A. (2012). Aspergillus fumigatus mitochondrial electron transport chain mediates oxidative stress homeostasis, hypoxia responses and fungal pathogenesis. Mol. Microbiol. 84, 383–399. doi: 10.1111/j.1365-2958.2012.08034.x
Grahl, N., Puttikamonkul, S., Macdonald, J. M., Gamcsik, M. P., Ngo, L. Y., Hohl, T. M., et al. (2011). In vivo hypoxia and a fungal alcohol dehydrogenase influence the pathogenesis of invasive pulmonary aspergillosis. PLoS Pathog. 7:e1002145. doi: 10.1371/journal.ppat.1002145
Gravelat, F. N., Doedt, T., Chiang, L. Y., Liu, H., Filler, S. G., Patterson, T. F., et al. (2008). In vivo analysis of Aspergillus fumigatus developmental gene expression determined by real-time reverse transcription-PCR. Infect. Immun. 76, 3632–3639. doi: 10.1128/IAI.01483-07
Graybill, J. R. (2000). The role of murine models in the development of antifungal therapy for systemic mycoses. Drug Resist. 3, 364–383. doi: 10.1054/drup.2000.0171
Graybill, J. R., Bocanegra, R., Gonzalez, G. M., and Najvar, L. K. (2003a). Combination antifungal therapy of murine aspergillosis: liposomal amphotericin B and micafungin. J. Antimicrob. Chemother. 52, 656–662. doi: 10.1093/jac/dkg425
Graybill, J. R., Bocanegra, R., Najvar, L. K., Loebenberg, D., and Luther, M. F. (1998). Granulocyte colony-stimulating factor and azole antifungal therapy in murine aspergillosis: role of immune suppression. Antimicrob. Agents Chemother. 42, 2467–2473.
Graybill, J. R., Kaster, S. R., and Drutz, D. J. (1983). Treatment of experimental murine aspergillosis with BAY n7133. J. Infect. Dis. 148, 898–906. doi: 10.1093/infdis/148.5.898
Graybill, J. R., Najvar, L. K., Gonzalez, G. M., Hernandez, S., and Bocanegra, R. (2003b). Improving the mouse model for studying the efficacy of voriconazole. J. Antimicrob. Chemother. 51, 1373–1376. doi: 10.1093/jac/dkg261
Gresnigt, M. S., Bozza, S., Becker, K. L., Joosten, L. A. B., Abdollahi-Roodsaz, S., van der Berg, W. B., et al. (2014). A polysaccharide virulence factor from Aspergillus fumigatus elicits anti-inflammatory effects through induction of Interleukin-1 receptor antagonist. PLoS Pathog. 10:e1003936. doi: 10.1371/journal.ppat.1003936
Grimm, M. J., Vethanayagam, R. R., Almyroudis, N. G., Dennis, C. G., Khan, A. N. H., D'Auria, A. C., et al. (2013). Monocyte- and macrophage-targeted NADPH oxidase mediates antifungal host defense and regulation of acute inflammation in mice. J. Immunol. 190, 4175–4184. doi: 10.4049/jimmunol.1202800
Guo, H., Gao, J., and Wu, X. (2012). Toll-like receptor 2 siRNA suppresses corneal inflammation and attenuates Aspergillus fumigatus keratitis in rats. Immunol. Cell Biol. 90, 352–357. doi: 10.1038/icb.2011.49
Habicht, J. M., Preiss, M., Passweg, J., Dalquen, P., Matt, P., Adler, H., et al. (2002). Invasive pulmonary aspergillosis: effects of early resection in a neutropenic rat model. Eur. J. Cardio Thorac. Surg. Off. J. Eur. Assoc. Cardio Thorac. Surg. 22, 728–732. doi: 10.1016/S1010-7940(02)00467-0
Hachem, R., Bahna, P., Hanna, H., Stephens, L. C., and Raad, I. (2006). EDTA as an adjunct antifungal agent for invasive pulmonary aspergillosis in a rodent model. Antimicrob. Agents Chemother. 50, 1823–1827. doi: 10.1128/AAC.50.5.1823-1827.2006
Han, K.-H., Chun, Y.-H., Figueiredo, B. d. C., Soriani, F. M., Savoldi, M., Almeida, A., et al. (2010). The conserved and divergent roles of carbonic anhydrases in the filamentous fungi Aspergillus fumigatus and Aspergillus nidulans. Mol. Microbiol. 75, 1372–1388. doi: 10.1111/j.1365-2958.2010.07058.x
Hanazawa, R., Murayama, S. Y., and Yamaguchi, H. (2000). In-situ detection of Aspergillus fumigatus. J. Med. Microbiol. 49, 285–290. doi: 10.1099/0022-1317-49-3-285
Hanson, L. H., Clemons, K. V., Denning, D. W., and Stevens, D. A. (1995). Efficacy of oral saperconazole in systemic murine aspergillosis. J. Med. Vet. Mycol. 33, 311–317.
Hao, W., Pan, Y.-X., Ding, Y.-Q., Xiao, S., Yin, K., Wang, Y.-D., et al. (2008). Well-characterized monoclonal antibodies against cell wall antigen of Aspergillus species improve immunoassay specificity and sensitivity. Clin. Vaccine Immunol. 15, 194–202. doi: 10.1128/CVI.00362-07
Hashimoto, A., Yamakami, Y., Kamberi, P., Yamagata, E., Karashima, R., Nagaoka, H., et al. (1998). Comparison of PCR, (1–>3)-beta-D-glucan and galactomannan assays in sera of rats with experimental invasive aspergillosis. J. Clin. Lab. Anal. 12, 257–262. doi: 10.1002/(SICI)1098-2825(1998)12:5<257::AID-JCLA1>;3.0.CO;2-3
Hata, K., Horii, T., Miyazaki, M., Watanabe, N.-A., Okubo, M., Sonoda, J., et al. (2011). Efficacy of oral E1210, a new broad-spectrum antifungal with a novel mechanism of action, in murine models of candidiasis, aspergillosis, and fusariosis. Antimicrob. Agents Chemother. 55, 4543–4551. doi: 10.1128/AAC.00366-11
Hata, K., Kimura, J., Miki, H., Toyosawa, T., Moriyama, M., and Katsu, K. (1996a). Efficacy of ER-30346, a novel oral triazole antifungal agent, in experimental models of aspergillosis, candidiasis, and cryptococcosis. Antimicrob. Agents Chemother. 40, 2243–2247.
Hata, K., Kimura, J., Miki, H., Toyosawa, T., Nakamura, T., and Katsu, K. (1996b). In vitro and in vivo antifungal activities of ER-30346, a novel oral triazole with a broad antifungal spectrum. Antimicrob. Agents Chemother. 40, 2237–2242.
Hector, R. F., Yee, E., and Collins, M. S. (1990). Use of DBA/2N mice in models of systemic candidiasis and pulmonary and systemic aspergillosis. Infect. Immun. 58, 1476–1478.
Heesemann, L., Kotz, A., Echtenacher, B., Broniszewska, M., Routier, F., Hoffmann, P., et al. (2011). Studies on galactofuranose-containing glycostructures of the pathogenic mold Aspergillus fumigatus. Int. J. Med. Microbiol. 301, 523–530. doi: 10.1016/j.ijmm.2011.02.003
Hein, K. Z., Takahashi, H., Tsumori, T., Yasui, Y., Nanjoh, Y., Toga, T., et al. (2015). Disulphide-reduced psoriasin is a human apoptosis-inducing broad-spectrum fungicide. Proc. Natl. Acad. Sci. U.S.A. 112, 13039–13044. doi: 10.1073/pnas.1511197112
Hennessy, M. B. (1999). Social influences on endocrine activity in guinea pigs, with comparisons to findings in nonhuman primates. Neurosci. Biobehav. Rev. 23, 687–698. doi: 10.1016/S0149-7634(99)00013-5
Herbst, S., Shah, A., Carby, M., Chusney, G., Kikkeri, N., Dorling, A., et al. (2013). A new and clinically relevant murine model of solid-organ transplant aspergillosis. Dis. Model. Mech. 6, 643–651. doi: 10.1242/dmm.010330
Herbst, S., Shah, A., Mazon Moya, M., Marzola, V., Jensen, B., Reed, A., et al. (2015). Phagocytosis-dependent activation of a TLR9-BTK-calcineurin-NFAT pathway co-ordinates innate immunity to Aspergillus fumigatus. EMBO Mol. Med. 7, 240–258. doi: 10.15252/emmm.201404556
High, K. P., and Washburn, R. G. (1997). Invasive aspergillosis in mice immunosuppressed with cyclosporin A, tacrolimus (FK506), or sirolimus (rapamycin). J. Infect. Dis. 175, 222–225. doi: 10.1093/infdis/175.1.222
Hohl, T. M. (2014). Overview of vertebrate animal models of fungal infection. J. Immunol. Methods 410, 100–112. doi: 10.1016/j.jim.2014.03.022
Hooper, D. G., Bolton, V. E., Sutton, J. S., Guilford, F. T., Straus, D. C., Najvar, L. K., et al. (2012). Assessment of Aspergillus fumigatus in guinea pig bronchoalveolar lavages and pulmonary tissue by culture and realtime polymerase chain reaction studies. Int. J. Mol. Sci. 13, 726–736. doi: 10.3390/ijms13010726
Huber, F., and Bignell, E. (2014). Distribution, expression and expansion of Aspergillus fumigatus LINE-like retrotransposon populations in clinical and environmental isolates. Fungal Genet. Biol. 64, 36–44. doi: 10.1016/j.fgb.2014.01.002
Hummel, M., Baust, C., Kretschmar, M., Nichterlein, T., Schleiermacher, D., Spiess, B., et al. (2004). Detection of Aspergillus DNA by a nested PCR assay is superior to blood culture in an experimental murine model of invasive aspergillosis. J. Med. Microbiol. 53, 803–806. doi: 10.1099/jmm.0.45545-0
Iannitti, R. G., Casagrande, A., De Luca, A., Cunha, C., Sorci, G., Riuzzi, F., et al. (2013). Hypoxia promotes danger-mediated inflammation via receptor for advanced glycation end products in cystic fibrosis. Am. J. Respir. Crit. Care Med. 188, 1338–1350. doi: 10.1164/rccm.201305-0986OC
Ibrahim, A. S., Gebremariam, T., French, S. W., Edwards, J. E., and Spellberg, B. (2010). The iron chelator deferasirox enhances liposomal amphotericin B efficacy in treating murine invasive pulmonary aspergillosis. J. Antimicrob. Chemother. 65, 289–292. doi: 10.1093/jac/dkp426
Ibrahim, A. S., Gebremariam, T., Luo, G., Fu, Y., French, S. W., Edwards, J. E., et al. (2011). Combination therapy of murine mucormycosis or aspergillosis with iron chelation, polyenes, and echinocandins. Antimicrob. Agents Chemother. 55, 1768–1770. doi: 10.1128/AAC.01577-10
Ibrahim-Granet, O., Jouvion, G., Hohl, T. M., Droin-Bergère, S., Philippart, F., Kim, O. Y., et al. (2010). In vivo bioluminescence imaging and histopathopathologic analysis reveal distinct roles for resident and recruited immune effector cells in defense against invasive aspergillosis. BMC Microbiol. 10:105. doi: 10.1186/1471-2180-10-105
Imai, J., Singh, G., Fernandez, B., Clemons, K. V., and Stevens, D. A. (2005). Efficacy of Abelcet and caspofungin, alone or in combination, against CNS aspergillosis in a murine model. J. Antimicrob. Chemother. 56, 166–171. doi: 10.1093/jac/dki178
Ito, J. I., Lyons, J. M., Hong, T. B., Tamae, D., Liu, Y.-K., Wilczynski, S. P., et al. (2006). Vaccinations with recombinant variants of Aspergillus fumigatus allergen Asp f 3 protect mice against invasive aspergillosis. Infect. Immun. 74, 5075–5084. doi: 10.1128/IAI.00815-06
Jahn, B., Koch, A., Schmidt, A., Wanner, G., Gehringer, H., Bhakdi, S., et al. (1997). Isolation and characterization of a pigmentless-conidium mutant of Aspergillus fumigatus with altered conidial surface and reduced virulence. Infect. Immun. 65, 5110–5117.
Jambunathan, K., Watson, D. S., Najvar, L. K., Wiederhold, N. P., Kirkpatrick, W. R., Patterson, T. F., et al. (2013). Prolyl endopeptidase activity in bronchoalveolar lavage fluid: a novel diagnostic biomarker in a guinea pig model of invasive pulmonary aspergillosis. Med. Mycol. 51, 592–602. doi: 10.3109/13693786.2012.761360
Jaton-Ogay, K., Paris, S., Huerre, M., Quadroni, M., Falchetto, R., Togni, G., et al. (1994). Cloning and disruption of the gene encoding an extracellular metalloprotease of Aspergillus fumigatus. Mol. Microbiol. 14, 917–928. doi: 10.1111/j.1365-2958.1994.tb01327.x
Jensen, H. E., and Hau, J. (1990a). A murine model for the study of the impact of Aspergillus fumigatus inoculation on the foeto-placental unit. Mycopathologia 112, 11–18.
Jensen, H. E., and Hau, J. (1990b). Murine mycotic placentitis produced by intravenous inoculation of conidia from Aspergillus fumigatus. Vivo Athens Greece 4, 247–252.
Jensen, H. E., and Schønheyder, H. (1993). Experimental murine mycotic placentitis and abortion. A potent animal model (short communication). J. Exp. Anim. Sci. 35, 155–160.
Jensen, H. E., Halbaek, B., Lind, P., Krogh, H. V., and Frandsen, P. L. (1996). Development of murine monoclonal antibodies for the immunohistochemical diagnosis of systemic bovine aspergillosis. J. Vet. Diagn. Investig. Off. Publ. Am. Assoc. Vet. Lab. Diagn. Inc 8, 68–75.
Jhingran, A., Kasahara, S., Shepardson, K. M., Junecko, B. A. F., Heung, L. J., Kumasaka, D. K., et al. (2015). Compartment-specific and sequential role of MyD88 and CARD9 in chemokine induction and innate defense during respiratory fungal infection. PLoS Pathog. 11:e1004589. doi: 10.1371/journal.ppat.1004589
Jhingran, A., Mar, K. B., Kumasaka, D. K., Knoblaugh, S. E., Ngo, L. Y., Segal, B. H., et al. (2012). Tracing conidial fate and measuring host cell antifungal activity using a reporter of microbial viability in the lung. Cell Rep. 2, 1762–1773. doi: 10.1016/j.celrep.2012.10.026
Jiang, N., Zhao, G., Lin, J., Hu, L., Che, C., Li, C., et al. (2015). Indoleamine 2,3-dioxygenase is involved in the inflammation response of corneal epithelial Cclls to Aspergillus fumigatus infections. PLoS ONE 10:e0137423. doi: 10.1371/journal.pone.0137423
Jiang, N., Zhao, G.-Q., Lin, J., Hu, L.-T., Che, C.-Y., Li, C., et al. (2016). Expression of indoleamine 2,3-dioxygenase in a murine model of Aspergillus fumigatus keratitis. Int. J. Ophthalmol. 9, 491–496. doi: 10.18240/ijo.2016.04.03
Jie, Z., Wu, X., and Yu, F.-S. X. (2009). Activation of Toll-like receptors 2 and 4 in Aspergillus fumigatus keratitis. Innate Immun. 15, 155–168. doi: 10.1177/1753425908101521
Johnson, E. M., Oakley, K. L., Radford, S. A., Moore, C. B., Warn, P., Warnock, D. W., et al. (2000). Lack of correlation of in vitro amphotericin B susceptibility testing with outcome in a murine model of Aspergillus infection. J. Antimicrob. Chemother. 45, 85–93. doi: 10.1093/jac/45.1.85
Jouvion, G., Brock, M., Droin-Bergère, S., and Ibrahim-Granet, O. (2012). Duality of liver and kidney lesions after systemic infection of immunosuppressed and immunocompetent mice with Aspergillus fumigatus. Virulence 3, 43–50. doi: 10.4161/viru.3.1.18654
Juvvadi, P. R., Gehrke, C., Fortwendel, J. R., Lamoth, F., Soderblom, E. J., Cook, E. C., et al. (2013). Phosphorylation of Calcineurin at a novel serine-proline rich region orchestrates hyphal growth and virulence in Aspergillus fumigatus. PLoS Pathog. 9:e1003564. doi: 10.1371/journal.ppat.1003564
Kakeya, H., Miyazaki, Y., Senda, H., Kobayashi, T., Seki, M., Izumikawa, K., et al. (2008). Efficacy of SPK-843, a novel polyene antifungal, in comparison with amphotericin B, liposomal amphotericin B, and micafungin against murine pulmonary aspergillosis. Antimicrob. Agents Chemother. 52, 1868–1870. doi: 10.1128/AAC.01369-07
Kapp, K., Prüfer, S., Michel, C. S., Habermeier, A., Luckner-Minden, C., Giese, T., et al. (2014). Granulocyte functions are independent of arginine availability. J. Leukoc. Biol. 96, 1047–1053. doi: 10.1189/jlb.3AB0214-082R
Karki, R., Man, S. M., Malireddi, R. K. S., Gurung, P., Vogel, P., Lamkanfi, M., et al. (2015). Concerted activation of the AIM2 and NLRP3 inflammasomes orchestrates host protection against Aspergillus infection. Cell Host Microbe 17, 357–368. doi: 10.1016/j.chom.2015.01.006
Kasahara, S., Jhingran, A., Dhingra, S., Salem, A., Cramer, R. A., and Hohl, T. M. (2016). Role of Granulocyte-Macrophage Colony-Stimulating Factor signaling in regulating neutrophil antifungal activity and the oxidative burst during respiratory fungal challenge. J. Infect. Dis. 213, 1289–1298. doi: 10.1093/infdis/jiw054
Kaur, S., Gupta, V. K., Thiel, S., Sarma, P. U., and Madan, T. (2007). Protective role of mannan-binding lectin in a murine model of invasive pulmonary aspergillosis. Clin. Exp. Immunol. 148, 382–389. doi: 10.1111/j.1365-2249.2007.03351.x
Kerr, S. C., Fischer, G. J., Sinha, M., McCabe, O., Palmer, J. M., Choera, T., et al. (2016). FleA expression in Aspergillus fumigatus is recognized by fucosylated structures on mucins and macrophages to prevent lung infection. PLoS Pathog. 12:e1005555. doi: 10.1371/journal.ppat.1005555
Khan, Z. U., Ahmad, S., and Theyyathel, A. M. (2008). Detection of Aspergillus fumigatus-specific DNA, (1-3)-beta-D-glucan and galactomannan in serum and bronchoalveolar lavage specimens of experimentally infected rats. Mycoses 51, 129–135. doi: 10.1111/j.1439-0507.2007.01461.x
Khosravi, A. R., Mahdavi Omran, S., Shokri, H., Lotfi, A., and Moosavi, Z. (2012). Importance of elastase production in development of invasive aspergillosis. J. Mycol. Médicale 22, 167–172. doi: 10.1016/j.mycmed.2012.03.002
Kirkpatrick, W. R., Coco, B. J., and Patterson, T. F. (2006). Sequential or combination antifungal therapy with voriconazole and liposomal amphotericin B in a Guinea pig model of invasive aspergillosis. Antimicrob. Agents Chemother. 50, 1567–1569. doi: 10.1128/AAC.50.4.1567-1569.2006
Kirkpatrick, W. R., McAtee, R. K., Fothergill, A. W., Loebenberg, D., Rinaldi, M. G., and Patterson, T. F. (2000a). Efficacy of SCH56592 in a rabbit model of invasive aspergillosis. Antimicrob. Agents Chemother. 44, 780–782. doi: 10.1128/AAC.44.3.780-782.2000
Kirkpatrick, W. R., McAtee, R. K., Fothergill, A. W., Rinaldi, M. G., and Patterson, T. F. (2000b). Efficacy of voriconazole in a guinea pig model of disseminated invasive aspergillosis. Antimicrob. Agents Chemother. 44, 2865–2868. doi: 10.1128/AAC.44.10.2865-2868.2000
Kirkpatrick, W. R., Najvar, L. K., Vallor, A. C., Wiederhold, N. P., Bocanegra, R., Pfeiffer, J., et al. (2012). Prophylactic efficacy of single dose pulmonary administration of amphotericin B inhalation powder in a guinea pig model of invasive pulmonary aspergillosis. J. Antimicrob. Chemother. 67, 970–976. doi: 10.1093/jac/dkr567
Kirkpatrick, W. R., Perea, S., Coco, B. J., and Patterson, T. F. (2002). Efficacy of caspofungin alone and in combination with voriconazole in a Guinea pig model of invasive aspergillosis. Antimicrob. Agents Chemother. 46, 2564–2568.
Kirkpatrick, W. R., Wiederhold, N. P., Najvar, L. K., and Patterson, T. F. (2013). Animal models in mycology: what have we learned over the past 30 years. Curr. Fungal Infect. Rep. 7, 68–78. doi: 10.1007/s12281-012-0126-6
Kleinschmidt-DeMasters, B. K. (2002). Central nervous system aspergillosis: a 20-year retrospective series. Hum. Pathol. 33, 116–124. doi: 10.1053/hupa.2002.30186
Kolattukudy, P. E., Lee, J. D., Rogers, L. M., Zimmerman, P., Ceselski, S., Fox, B., et al. (1993). Evidence for possible involvement of an elastolytic serine protease in aspergillosis. Infect. Immun. 61, 2357–2368.
Komadina, T. G., Wilkes, T. D., Shock, J. P., Ulmer, W. C., Jackson, J., and Bradsher, R. W. (1985). Treatment of Aspergillus fumigatus keratitis in rabbits with oral and topical ketoconazole. Am. J. Ophthalmol. 99, 476–479. doi: 10.1016/0002-9394(85)90017-0
Kothary, M. H., Chase, T., and Macmillan, J. D. (1984). Correlation of elastase production by some strains of Aspergillus fumigatus with ability to cause pulmonary invasive aspergillosis in mice. Infect. Immun. 43, 320–325.
Kotz, A., Wagener, J., Engel, J., Routier, F., Echtenacher, B., Pich, A., et al. (2010). The mitA gene of Aspergillus fumigatus is required for mannosylation of inositol-phosphorylceramide, but is dispensable for pathogenicity. Fungal Genet. Biol. 47, 169–178. doi: 10.1016/j.fgb.2009.10.001
Kretschmar, M., Buchheidt, D., Hof, H., and Nichterlein, T. (2001). Galactomannan enzyme immunoassay for monitoring systemic infection with Aspergillus fumigatus in mice. Diagn. Microbiol. Infect. Dis. 41, 107–112. doi: 10.1016/S0732-8893(01)00282-6
Kupfahl, C., Heinekamp, T., Geginat, G., Ruppert, T., Härtl, A., Hof, H., et al. (2006). Deletion of the gliP gene of Aspergillus fumigatus results in loss of gliotoxin production but has no effect on virulence of the fungus in a low-dose mouse infection model. Mol. Microbiol. 62, 292–302. doi: 10.1111/j.1365-2958.2006.05373.x
Kurtz, M. B., Bernard, E. M., Edwards, F. F., Marrinan, J. A., Dropinski, J., Douglas, C. M., et al. (1995). Aerosol and parenteral pneumocandins are effective in a rat model of pulmonary aspergillosis. Antimicrob. Agents Chemother. 39, 1784–1789. doi: 10.1128/AAC.39.8.1784
Kurup, V. P. (1984). Interaction of Aspergillus fumigatus spores and pulmonary alveolar macrophages of rabbits. Immunobiology 166, 53–61. doi: 10.1016/S0171-2985(84)80143-6
Lamoth, F., Juvvadi, P. R., Gehrke, C., Asfaw, Y. G., and Steinbach, W. J. (2014a). Transcriptional activation of heat shock protein 90 mediated via a proximal promoter region as trigger of caspofungin resistance in Aspergillus fumigatus. J. Infect. Dis. 209, 473–481. doi: 10.1093/infdis/jit530
Lamoth, F., Juvvadi, P. R., Soderblom, E. J., Moseley, M. A., Asfaw, Y. G., and Steinbach, W. J. (2014b). Identification of a key lysine residue in heat shock protein 90 required for azole and echinocandin resistance in Aspergillus fumigatus. Antimicrob. Agents Chemother. 58, 1889–1896. doi: 10.1128/AAC.02286-13
Land, C. J., Sostarić, B., Fuchs, R., Lundström, H., and Hult, K. (1989). Intratracheal exposure of rats to Aspergillus fumigatus spores isolated from sawmills in Sweden. Appl. Environ. Microbiol. 55, 2856–2860.
Langfelder, K., Jahn, B., Gehringer, H., Schmidt, A., Wanner, G., and Brakhage, A. A. (1998). Identification of a polyketide synthase gene (pksP) of Aspergillus fumigatus involved in conidial pigment biosynthesis and virulence. Med. Microbiol. Immunol. 187, 79–89. doi: 10.1007/s004300050077
Lanternier, F., Cypowyj, S., Picard, C., Bustamante, J., Lortholary, O., Casanova, J.-L., et al. (2013). Primary immunodeficiencies underlying fungal infections. Curr. Opin. Pediatr. 25, 736–747. doi: 10.1097/mop.0000000000000031
Le Conte, P., Joly, V., Saint-Julien, L., Gillardin, J. M., Carbon, C., and Yeni, P. (1992). Tissue distribution and antifungal effect of liposomal itraconazole in experimental cryptococcosis and pulmonary aspergillosis. Am. Rev. Respir. Dis. 145, 424–429. doi: 10.1164/ajrccm/145.2_Pt_1.424
Leal, S. M., Cowden, S., Hsia, Y.-C., Ghannoum, M. A., Momany, M., and Pearlman, E. (2010). Distinct roles for Dectin-1 and TLR4 in the pathogenesis of Aspergillus fumigatus keratitis. PLoS Pathog. 6:e1000976. doi: 10.1371/journal.ppat.1000976
Leal, S. M., Roy, S., Vareechon, C., Carrion, S. d., Clark, H., Lopez-Berges, M. S., et al. (2013). Targeting iron acquisition blocks infection with the fungal pathogens Aspergillus fumigatus and Fusarium oxysporum. PLoS Pathog. 9:e1003436. doi: 10.1371/journal.ppat.1003436
Leal, S. M., Vareechon, C., Cowden, S., Cobb, B. A., Latgé, J.-P., Momany, M., et al. (2012). Fungal antioxidant pathways promote survival against neutrophils during infection. J. Clin. Invest. 122, 2482–2498. doi: 10.1172/JCI63239
Lee, I., Oh, J.-H., Shwab, E. K., Dagenais, T. R. T., Andes, D., and Keller, N. P. (2009). HdaA, a class 2 histone deacetylase of Aspergillus fumigatus, affects germination and secondary metabolite production. Fungal Genet. Biol. 46, 782–790. doi: 10.1016/j.fgb.2009.06.007
Leenders, A. C., de Marie, S., ten Kate, M. T., Bakker-Woudenberg, I. A., and Verbrugh, H. A. (1996). Liposomal amphotericin B (AmBisome) reduces dissemination of infection as compared with amphotericin B deoxycholate (Fungizone) in a rate model of pulmonary aspergillosis. J. Antimicrob. Chemother. 38, 215–225. doi: 10.1093/jac/38.2.215
Lehmann, P. F., and White, L. O. (1975). Chitin assay used to demonstrate renal localization and cortisone-enhanced growth of Aspergillus fumigatus mycelium in mice. Infect. Immun. 12, 987–992.
Lehmann, P. F., and White, L. O. (1976). Acquired immunity to Aspergillus fumigatus. Infect. Immun. 13, 1296–1298.
Lehmann, P. F., and White, L. O. (1978). Rapid germination of Aspergillus fumigatus conidia in mouse kidneys and a kidney extract. Sabouraudia 16, 203–209. doi: 10.1080/00362177885380271
Leleu, C., Menotti, J., Meneceur, P., Choukri, F., Sulahian, A., Garin, Y. J.-F., et al. (2013a). Bayesian development of a dose-response model for Aspergillus fumigatus and invasive aspergillosis. Risk Anal. Off. Publ. Soc. Risk Anal. 33, 1441–1453. doi: 10.1111/risa.12007
Leleu, C., Menotti, J., Meneceur, P., Choukri, F., Sulahian, A., Garin, Y. J.-F., et al. (2013b). Efficacy of liposomal amphotericin B for prophylaxis of acute or reactivation models of invasive pulmonary aspergillosis. Mycoses 56, 241–249. doi: 10.1111/myc.12011
Lengerova, M., Kocmanova, I., Racil, Z., Hrncirova, K., Pospisilova, S., Mayer, J., et al. (2012). Detection and measurement of fungal burden in a guinea pig model of invasive pulmonary aspergillosis by novel quantitative nested real-time PCR compared with galactomannan and (1,3)-β-D-glucan detection. J. Clin. Microbiol. 50, 602–608. doi: 10.1128/JCM.05356-11
Lepak, A. J., Marchillo, K., VanHecker, J., and Andes, D. R. (2013a). Impact of in vivo triazole and echinocandin combination therapy for invasive pulmonary aspergillosis: enhanced efficacy against Cyp51 mutant isolates. Antimicrob. Agents Chemother. 57, 5438–5447. doi: 10.1128/AAC.00833-13
Lepak, A. J., Marchillo, K., Vanhecker, J., and Andes, D. R. (2013b). Isavuconazole (BAL4815) pharmacodynamic target determination in an in vivo murine model of invasive pulmonary aspergillosis against wild-type and cyp51 mutant isolates of Aspergillus fumigatus. Antimicrob. Agents Chemother. 57, 6284–6289. doi: 10.1128/AAC.01355-13
Lepak, A. J., Marchillo, K., Vanhecker, J., and Andes, D. R. (2013c). Posaconazole pharmacodynamic target determination against wild-type and Cyp51 mutant isolates of Aspergillus fumigatus in an in vivo model of invasive pulmonary aspergillosis. Antimicrob. Agents Chemother. 57, 579–585. doi: 10.1128/AAC.01279-12
Lewis, R. E., and Wiederhold, N. P. (2005). Murine model of invasive aspergillosis. Methods Mol. Med. 118, 129–142. doi: 10.1385/1-59259-943-5:129
Lewis, R. E., Prince, R. A., Chi, J., and Kontoyiannis, D. P. (2002). Itraconazole preexposure attenuates the efficacy of subsequent amphotericin B therapy in a murine model of acute invasive pulmonary aspergillosis. Antimicrob. Agents Chemother. 46, 3208–3214. doi: 10.1128/AAC.46.10.3208-3214.2002
Lewis, R. E., Wiederhold, N. P., Chi, J., Han, X. Y., Komanduri, K. V., Kontoyiannis, D. P., et al. (2005). Detection of gliotoxin in experimental and human aspergillosis. Infect. Immun. 73, 635–637. doi: 10.1128/IAI.73.1.635-637.2005
Li, C., Zhao, G., Che, C., Lin, J., Li, N., Hu, L., et al. (2015). The role of LOX-1 in innate immunity to Aspergillus fumigatus in corneal epithelial cells. Invest. Ophthalmol. Vis. Sci. 56, 3593–3603. doi: 10.1167/iovs.14-15989
Li, P., Xu, X., Cao, E., Yu, B., Li, W., Fan, M., et al. (2014). Vitamin D deficiency causes defective resistance to Aspergillus fumigatus in mice via aggravated and sustained inflammation. PLoS ONE 9:e99805. doi: 10.1371/journal.pone.0099805
Lilly, L. M., Scopel, M., Nelson, M. P., Burg, A. R., Dunaway, C. W., and Steele, C. (2014). Eosinophil deficiency compromises lung defense against Aspergillus fumigatus. Infect. Immun. 82, 1315–1325. doi: 10.1128/IAI.01172-13
Lionakis, M. S., and Kontoyiannis, D. P. (2012). Drosophila melanogaster as a model organism for invasive aspergillosis. Methods Mol. Biol. 845, 455–468. doi: 10.1007/978-1-61779-539-8_32
Lionakis, M. S., Lewis, R. E., May, G. S., Wiederhold, N. P., Albert, N. D., Halder, G., et al. (2005). Toll-deficient Drosophila flies as a fast, high-throughput model for the study of antifungal drug efficacy against invasive aspergillosis and Aspergillus virulence. J. Infect. Dis. 191, 1188–1195. doi: 10.1086/428587
Liu, W., Yan, M., Sugui, J. A., Li, H., Xu, C., Joo, J., et al. (2013). Olfm4 deletion enhances defense against Staphylococcus aureus in chronic granulomatous disease. J. Clin. Invest. 123, 3751–3755. doi: 10.1172/JCI68453
Lo Giudice, P., Campo, S., Verdoliva, A., Rivieccio, V., Borsini, F., De Santis, R., et al. (2010). Efficacy of PTX3 in a rat model of invasive aspergillosis. Antimicrob. Agents Chemother. 54, 4513–4515. doi: 10.1128/AAC.00674-10
Loeffler, J., Kloepfer, K., Hebart, H., Najvar, L., Graybill, J. R., Kirkpatrick, W. R., et al. (2002). Polymerase chain reaction detection of aspergillus DNA in experimental models of invasive aspergillosis. J. Infect. Dis. 185, 1203–1206. doi: 10.1086/339824
Longman, L. P., and Martin, M. V. (1987). A comparison of the efficacy of itraconazole, amphotericin B and 5-fluorocytosine in the treatment of Aspergillus fumigatus endocarditis in the rabbit. J. Antimicrob. Chemother. 20, 719–724. doi: 10.1093/jac/20.5.719
Lortholary, O., Gangneux, J.-P., Sitbon, K., Lebeau, B., de Monbrison, F., Le Strat, Y., et al. (2011). Epidemiological trends in invasive aspergillosis in France: the SAIF network (2005-2007). Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 17, 1882–1889. doi: 10.1111/j.1469-0691.2011.03548.x
Losada, L., Sugui, J. A., Eckhaus, M. A., Chang, Y. C., Mounaud, S., Figat, A., et al. (2015). Genetic analysis using an isogenic mating pair of Aspergillus fumigatus identifies azole resistance genes and lack of MAT locus's role in virulence. PLoS Pathog. 11:e1004834. doi: 10.1371/journal.ppat.1004834
Loussert, C., Schmitt, C., Prevost, M.-C., Balloy, V., Fadel, E., Philippe, B., et al. (2010). In vivo biofilm composition of Aspergillus fumigatus. Cell. Microbiol. 12, 405–410. doi: 10.1111/j.1462-5822.2009.01409.x
Lupetti, A., Welling, M. M., Mazzi, U., Nibbering, P. H., and Pauwels, E. K. J. (2002). Technetium-99m labelled fluconazole and antimicrobial peptides for imaging of Candida albicans and Aspergillus fumigatus infections. Eur. J. Nucl. Med. Mol. Imaging 29, 674–679. doi: 10.1007/s00259-001-0760-7
Luque, J. C., Clemons, K. V., and Stevens, D. A. (2003). Efficacy of micafungin alone or in combination against systemic murine aspergillosis. Antimicrob. Agents Chemother. 47, 1452–1455. doi: 10.1128/AAC.47.4.1452-1455.2003
Ma, Y., Qiao, J., Liu, W., Wan, Z., Wang, X., Calderone, R., et al. (2008). The sho1 sensor regulates growth, morphology, and oxidant adaptation in Aspergillus fumigatus but is not essential for development of invasive pulmonary aspergillosis. Infect. Immun. 76, 1695–1701. doi: 10.1128/IAI.01507-07
Maarman, G., Lecour, S., Butrous, G., Thienemann, F., and Sliwa, K. (2013). A comprehensive review: the evolution of animal models in pulmonary hypertension research; are we there yet? Pulm. Circ. 3, 739–756. doi: 10.1086/674770
MacCallum, D. M., and Odds, F. C. (2002). Influence of grapefruit juice on itraconazole plasma levels in mice and guinea pigs. J. Antimicrob. Chemother. 50, 219–224. doi: 10.1093/jac/dkf103
MacCallum, D. M., Whyte, J. A., and Odds, F. C. (2005). Efficacy of caspofungin and voriconazole combinations in experimental aspergillosis. Antimicrob. Agents Chemother. 49, 3697–3701. doi: 10.1128/AAC.49.9.3697-3701.2005
Madan, T., Kishore, U., Singh, M., Strong, P., Clark, H., Hussain, E. M., et al. (2001). Surfactant proteins A and D protect mice against pulmonary hypersensitivity induced by Aspergillus fumigatus antigens and allergens. J. Clin. Invest. 107, 467–475. doi: 10.1172/JCI10124
Madan, T., Reid, K. B. M., Clark, H., Singh, M., Nayak, A., Sarma, P. U., et al. (2010). Susceptibility of mice genetically deficient in SP-A or SP-D gene to invasive pulmonary aspergillosis. Mol. Immunol. 47, 1923–1930. doi: 10.1016/j.molimm.2010.02.027
Mahajan, V. M., Dayal, Y., Patra, C. P., and Bhatia, I. M. (1978). Experimental aspergillosis in monkeys. Sabouraudia 16, 199–201. doi: 10.1080/00362177885380261
Maheshwari, R. K., Tandon, R. N., Feuillette, A. R., Mahouy, G., Badillet, G., and Friedman, R. M. (1988). Interferon inhibits Aspergillus fumigatus growth in mice: an activity against an extracellular infection. J. Interferon Res. 8, 35–44. doi: 10.1089/jir.1988.8.35
Markaryan, A., Morozova, I., Yu, H., and Kolattukudy, P. E. (1994). Purification and characterization of an elastinolytic metalloprotease from Aspergillus fumigatus and immunoelectron microscopic evidence of secretion of this enzyme by the fungus invading the murine lung. Infect. Immun. 62, 2149–2157.
Marr, K. A., Balajee, S. A., McLaughlin, L., Tabouret, M., Bentsen, C., and Walsh, T. J. (2004). Detection of galactomannan antigenemia by enzyme immunoassay for the diagnosis of invasive aspergillosis: variables that affect performance. J. Infect. Dis. 190, 641–649. doi: 10.1086/422009
Martin, M. V., Yates, J., and Hitchcock, C. A. (1997). Comparison of voriconazole (UK-109,496) and itraconazole in prevention and treatment of Aspergillus fumigatus endocarditis in guinea pigs. Antimicrob. Agents Chemother. 41, 13–16.
Martinez, M., Chen, V., Tong, A.-J., Hamilton, K., Clemons, K. V., and Stevens, D. A. (2013). Experimental evidence that granulocyte transfusions are efficacious in treatment of neutropenic hosts with pulmonary aspergillosis. Antimicrob. Agents Chemother. 57, 1882–1887. doi: 10.1128/AAC.02533-12
Mavridou, E., Brüggemann, R. J. M., Melchers, W. J. G., Mouton, J. W., and Verweij, P. E. (2010a). Efficacy of posaconazole against three clinical Aspergillus fumigatus isolates with mutations in the cyp51A gene. Antimicrob. Agents Chemother. 54, 860–865. doi: 10.1128/AAC.00931-09
Mavridou, E., Bruggemann, R. J. M., Melchers, W. J. G., Verweij, P. E., and Mouton, J. W. (2010b). Impact of cyp51A mutations on the pharmacokinetic and pharmacodynamic properties of voriconazole in a murine model of disseminated aspergillosis. Antimicrob. Agents Chemother. 54, 4758–4764. doi: 10.1128/AAC.00606-10
Mazaki, Y., Hashimoto, S., Tsujimura, T., Morishige, M., Hashimoto, A., Aritake, K., et al. (2006). Neutrophil direction sensing and superoxide production linked by the GTPase-activating protein GIT2. Nat. Immunol. 7, 724–731. doi: 10.1038/ni1349
Mazzolla, R., Barluzzi, R., Romani, L., Mosci, P., and Bistoni, F. (1991). Anti-Candida resistance in the mouse brain and effect of intracerebral administration of interleukin 1. J. Gen. Microbiol. 137, 1799–1804. doi: 10.1099/00221287-137-8-1799
McCulloch, E., Ramage, G., Jones, B., Warn, P., Kirkpatrick, W. R., Patterson, T. F., et al. (2009). Don't throw your blood clots away: use of blood clot may improve sensitivity of PCR diagnosis in invasive aspergillosis. J. Clin. Pathol. 62, 539–541. doi: 10.1136/jcp.2008.063321
McCulloch, E., Ramage, G., Rajendran, R., Lappin, D. F., Jones, B., Warn, P., et al. (2012). Antifungal treatment affects the laboratory diagnosis of invasive aspergillosis. J. Clin. Pathol. 65, 83–86. doi: 10.1136/jcp.2011.090464
McDonagh, A., Fedorova, N. D., Crabtree, J., Yu, Y., Kim, S., Chen, D., et al. (2008). Sub-telomere directed gene expression during initiation of invasive aspergillosis. PLoS Pathog. 4:e1000154. doi: 10.1371/journal.ppat.1000154
McMurray, D. N. (2001). Disease model: pulmonary tuberculosis. Trends Mol. Med. 7, 135–137. doi: 10.1016/S1471-4914(00)01901-8
Meerpoel, L., Heeres, J., Backx, L. J. J., Van der Veken, L. J. E., Hendrickx, R., Corens, D., et al. (2010). Synthesis and in vitro and in vivo antifungal activity of the hydroxy metabolites of saperconazole. ChemMedChem 5, 757–769. doi: 10.1002/cmdc.201000040
Mehrad, B., Strieter, R. M., and Standiford, T. J. (1999). Role of TNF-alpha in pulmonary host defense in murine invasive aspergillosis. J. Immunol. 162, 1633–1640.
Mehrad, B., Wiekowski, M., Morrison, B. E., Chen, S.-C., Coronel, E. C., Manfra, D. J., et al. (2002). Transient lung-specific expression of the chemokine KC improves outcome in invasive aspergillosis. Am. J. Respir. Crit. Care Med. 166, 1263–1268. doi: 10.1164/rccm.200204-367OC
Melchers, W. J., Verweij, P. E., van den Hurk, P., van Belkum, A., De Pauw, B. E., Hoogkamp-Korstanje, J. A., et al. (1994). General primer-mediated PCR for detection of Aspergillus species. J. Clin. Microbiol. 32, 1710–1717.
Mellado, E., Aufauvre-Brown, A., Gow, N. A., and Holden, D. W. (1996). The Aspergillus fumigatus chsC and chsG genes encode class III chitin synthases with different functions. Mol. Microbiol. 20, 667–679. doi: 10.1046/j.1365-2958.1996.5571084.x
Mellado, E., Garcia-Effron, G., Buitrago, M. J., Alcazar-Fuoli, L., Cuenca-Estrella, M., and Rodriguez-Tudela, J. L. (2005). Targeted gene disruption of the 14-alpha sterol demethylase (cyp51A) in Aspergillus fumigatus and its role in azole drug susceptibility. Antimicrob. Agents Chemother. 49, 2536–2538. doi: 10.1128/AAC.49.6.2536-2538.2005
Melloul, E., Thierry, S., Durand, B., Cordonnier, N., Desoubeaux, G., Chandenier, J., et al. (2014). Assessment of Aspergillus fumigatus burden in lungs of intratracheally-challenged turkeys (Meleagris gallopavo) by quantitative PCR, galactomannan enzyme immunoassay, and quantitative culture. Comp. Immunol. Microbiol. Infect. Dis. 37, 271–279. doi: 10.1016/j.cimid.2014.07.005
Meulbroek, J. A., Nilius, A. M., Li, Q., Wang, W., Hasvold, L., Steiner, B., et al. (2003). In vivo characterization of A-192411: a novel fungicidal lipopeptide (II). Bioorg. Med. Chem. Lett. 13, 495–497. doi: 10.1016/S0960-894X(02)00979-4
Mirkov, I., El-Muzghi, A. A. M., Djokic, J., Ninkov, M., Popov Aleksandrov, A., Glamoclija, J., et al. (2014). Pulmonary immune responses to Aspergillus fumigatus in rats. Biomed. Environ. Sci. 27, 684–694. doi: 10.3967/bes2014.104
Mitsutake, K., Kohno, S., Miyazaki, T., Yamamoto, Y., Yanagihara, K., Kakeya, H., et al. (1995). Detection of (1-3)-beta-D-glucan in a rat model of aspergillosis. J. Clin. Lab. Anal. 9, 119–122. doi: 10.1002/jcla.1860090208
Miyazaki, H. M., Kohno, S., Miyazaki, Y., Mitsutake, K., Tomono, K., Kaku, M., et al. (1993). Efficacy of intravenous itraconazole against experimental pulmonary aspergillosis. Antimicrob. Agents Chemother. 37, 2762–2765. doi: 10.1128/AAC.37.12.2762
Monod, M., Paris, S., Sarfati, J., Jaton-Ogay, K., Ave, P., and Latgé, J. P. (1993). Virulence of alkaline protease-deficient mutants of Aspergillus fumigatus. FEMS Microbiol. Lett. 106, 39–46. doi: 10.1111/j.1574-6968.1993.tb05932.x
Montagnoli, C., Bozza, S., Bacci, A., Gaziano, R., Mosci, P., Morschhäuser, J., et al. (2003). A role for antibodies in the generation of memory antifungal immunity. Eur. J. Immunol. 33, 1193–1204. doi: 10.1002/eji.200323790
Montagnoli, C., Fallarino, F., Gaziano, R., Bozza, S., Bellocchio, S., Zelante, T., et al. (2006). Immunity and tolerance to Aspergillus involve functionally distinct regulatory T cells and tryptophan catabolism. J. Immunol. 176, 1712–1723. doi: 10.4049/jimmunol.176.3.1712
Moonis, M., Ahmad, I., and Bachhawat, B. K. (1994). Effect of elimination of phagocytic cells by liposomal dichloromethylene diphosphonate on aspergillosis virulence and toxicity of liposomal amphotericin B in mice. J. Antimicrob. Chemother. 33, 571–583. doi: 10.1093/jac/33.3.571
Moretti, S., Bozza, S., D'Angelo, C., Casagrande, A., Della Fazia, M. A., Pitzurra, L., et al. (2012). Role of innate immune receptors in paradoxical caspofungin activity in vivo in preclinical aspergillosis. Antimicrob. Agents Chemother. 56, 4268–4276. doi: 10.1128/AAC.05198-11
Moretti, S., Bozza, S., Massi-Benedetti, C., Prezioso, L., Rossetti, E., Romani, L., et al. (2014). An immunomodulatory activity of micafungin in preclinical aspergillosis. J. Antimicrob. Chemother. 69, 1065–1074. doi: 10.1093/jac/dkt457
Morgenstern, D. E., Gifford, M. A., Li, L. L., Doerschuk, C. M., and Dinauer, M. C. (1997). Absence of respiratory burst in X-linked chronic granulomatous disease mice leads to abnormalities in both host defense and inflammatory response to Aspergillus fumigatus. J. Exp. Med. 185, 207–218. doi: 10.1084/jem.185.2.207
Morisse, H., Heyman, L., Salaün, M., Favennec, L., Picquenot, J. M., Bohn, P., et al. (2012). In vivo and in situ imaging of experimental invasive pulmonary aspergillosis using fibered confocal fluorescence microscopy. Med. Mycol. 50, 386–395. doi: 10.3109/13693786.2011.617788
Morisse, H., Heyman, L., Salaün, M., Favennec, L., Picquenot, J. M., Bohn, P., et al. (2013). In vivo molecular microimaging of pulmonary aspergillosis. Med. Mycol. 51, 352–360. doi: 10.3109/13693786.2012.729138
Morrison, B. E., Park, S. J., Mooney, J. M., and Mehrad, B. (2003). Chemokine-mediated recruitment of NK cells is a critical host defense mechanism in invasive aspergillosis. J. Clin. Invest. 112, 1862–1870. doi: 10.1172/JCI18125
Morton, C. O., Loeffler, J., De Luca, A., Frost, S., Kenny, C., Duval, S., et al. (2010). Dynamics of extracellular release of Aspergillus fumigatus DNA and galactomannan during growth in blood and serum. J. Med. Microbiol. 59, 408–413. doi: 10.1099/jmm.0.017418-0
Morton, C. O., de Luca, A., Romani, L., and Rogers, T. R. (2012). RT-qPCR detection of Aspergillus fumigatus RNA in vitro and in a murine model of invasive aspergillosis utilizing the PAXgene® and Tempus™ RNA stabilization systems. Med. Mycol. 50, 661–666. doi: 10.3109/13693786.2011.652200
Morton, D. B., and Griffiths, P. H. (1985). Guidelines on the recognition of pain, distress and discomfort in experimental animals and an hypothesis for assessment. Vet. Rec. 116, 431–436. doi: 10.1136/vr.116.16.431
Mosquera, J., Warn, P. A., Morrissey, J., Moore, C. B., Gil-Lamaignere, C., and Denning, D. W. (2001). Susceptibility testing of Aspergillus flavus: inoculum dependence with itraconazole and lack of correlation between susceptibility to amphotericin B in vitro and outcome in vivo. Antimicrob. Agents Chemother. 45, 1456–1462. doi: 10.1128/AAC.45.5.1456-1462.2001
Mouse Genome Sequencing Consortium, Waterston, R. H., Lindblad-Toh, K., Birney, E., Rogers, J., Abril, J. F., et al. (2002). Initial sequencing and comparative analysis of the mouse genome. Nature 420, 520–562. doi: 10.1038/nature01262
Mouton, J. W., te Dorsthorst, D. T. A., Meis, J. F. G. M., and Verweij, P. E. (2009). Dose-response relationships of three amphotericin B formulations in a non-neutropenic murine model of invasive aspergillosis. Med. Mycol. 47, 802–807. doi: 10.3109/13693780802672644
Mylonakis, E., Chalevelakis, G., Saroglou, G., Danias, P., Argyropoulou, A. D., Paniara, O., et al. (1997). Efficacy of deoxycholate amphotericin B and unilamellar liposomal amphotericin B in prophylaxis of experimental Aspergillus fumigatus endocarditis. Mayo Clin. Proc. 72, 1022–1027. doi: 10.4065/72.11.1022
Nagasaki, Y., Eriguchi, Y., Uchida, Y., Miyake, N., Maehara, Y., Kadowaki, M., et al. (2009). Combination therapy with micafungin and amphotericin B for invasive pulmonary aspergillosis in an immunocompromised mouse model. J. Antimicrob. Chemother. 64, 379–382. doi: 10.1093/jac/dkp175
Naik, S. R., Thakare, V. N., Desai, S. K., and Rahalkar, P. R. (2011). Study of immunological aspects of aspergillosis in mice and effect of polyene macrolide antibiotic (SJA-95) and IFN-γ: a possible role of IFN-γ as an adjunct in antifungal therapy. Immunol. Lett. 141, 68–73. doi: 10.1016/j.imlet.2011.07.008
Najvar, L. K., Cacciapuoti, A., Hernandez, S., Halpern, J., Bocanegra, R., Gurnani, M., et al. (2004). Activity of posaconazole combined with amphotericin B against Aspergillus flavus infection in mice: comparative studies in two laboratories. Antimicrob. Agents Chemother. 48, 758–764. doi: 10.1128/AAC.48.3.758-764.2004
Nawada, R., Amitani, R., Tanaka, E., Niimi, A., Suzuki, K., Murayama, T., et al. (1996). Murine model of invasive pulmonary aspergillosis following an earlier stage, noninvasive Aspergillus infection. J. Clin. Microbiol. 34, 1433–1439.
Nierman, W. C., Pain, A., Anderson, M. J., Wortman, J. R., Kim, H. S., Arroyo, J., et al. (2005). Genomic sequence of the pathogenic and allergenic filamentous fungus Aspergillus fumigatus. Nature 438, 1151–1156. doi: 10.1038/nature04332
Niwano, Y., Kuzuhara, N., Goto, Y., Munechika, Y., Kodama, H., Kanai, K., et al. (1999). Efficacy of NND-502, a novel imidazole antimycotic agent, in experimental models of Candida albicans and Aspergillus fumigatus infections. Int. J. Antimicrob. Agents 12, 221–228. doi: 10.1016/S0924-8579(99)00076-X
O'Dea, E. M., Amarsaikhan, N., Li, H., Downey, J., Steele, E., Van Dyken, S. J., et al. (2014). Eosinophils are recruited in response to chitin exposure and enhance Th2-mediated immune pathology in Aspergillus fumigatus infection. Infect. Immun. 82, 3199–3205. doi: 10.1128/IAI.01990-14
O'Hanlon, K. A., Cairns, T., Stack, D., Schrettl, M., Bignell, E. M., Kavanagh, K., et al. (2011). Targeted disruption of nonribosomal peptide synthetase pes3 augments the virulence of Aspergillus fumigatus. Infect. Immun. 79, 3978–3992. doi: 10.1128/IAI.00192-11
Odds, F. C., Oris, M., Van Dorsselaer, P., and Van Gerven, F. (2000). Activities of an intravenous formulation of itraconazole in experimental disseminated Aspergillus, Candida, and Cryptococcus infections. Antimicrob. Agents Chemother. 44, 3180–3183. doi: 10.1128/AAC.44.11.3180-3183.2000
Odds, F. C., Van Gerven, F., Espinel-Ingroff, A., Bartlett, M. S., Ghannoum, M. A., Lancaster, M. V., et al. (1998). Evaluation of possible correlations between antifungal susceptibilities of filamentous fungi in vitro and antifungal treatment outcomes in animal infection models. Antimicrob. Agents Chemother. 42, 282–288.
Okawa, Y., Murata, Y., Suzuki, M., Ito, M., Hotchi, M., and Suzuki, S. (2002). Delayed lethal response to Aspergillus fumigatus infection in sarcoma 180 tumor-bearing mice. FEMS Immunol. Med. Microbiol. 34, 113–117. doi: 10.1111/j.1574-695X.2002.tb00611.x
Otsubo, T., Maesaki, S., Hossain, M. A., Yamamoto, Y., Tomono, K., Tashiro, T., et al. (1999). In vitro and in vivo activities of NS-718, a new lipid nanosphere incorporating amphotericin B, against Aspergillus fumigatus. Antimicrob. Agents Chemother. 43, 471–475.
Otsubo, T., Maruyama, K., Maesaki, S., Miyazaki, Y., Tanaka, E., Takizawa, T., et al. (1998). Long-circulating immunoliposomal amphotericin B against invasive pulmonary aspergillosis in mice. Antimicrob. Agents Chemother. 42, 40–44.
Overdijk, B., Van Steijn, G. J., and Odds, F. C. (1996). Chitinase levels in guinea pig blood are increased after systemic infection with Aspergillus fumigatus. Glycobiology 6, 627–634. doi: 10.1093/glycob/6.6.627
Overdijk, B., Van Steijn, G. J., and Odds, F. C. (1999). Distribution of chitinase in guinea pig tissues and increases in levels of this enzyme after systemic infection with Aspergillus fumigatus. Microbiol. Read. Engl. 145(Pt 1), 259–269. doi: 10.1099/13500872-145-1-259
Paisley, D., Robson, G. D., and Denning, D. W. (2005). Correlation between in vitro growth rate and in vivo virulence in Aspergillus fumigatus. Med. Mycol. 43, 397–401. doi: 10.1080/13693780400005866
Panepinto, J. C., Oliver, B. G., Fortwendel, J. R., Smith, D. L. H., Askew, D. S., and Rhodes, J. C. (2003). Deletion of the Aspergillus fumigatus gene encoding the Ras-related protein RhbA reduces virulence in a model of invasive pulmonary aspergillosis. Infect. Immun. 71, 2819–2826. doi: 10.1128/IAI.71.5.2819-2826.2003
Paris, S., Monod, M., Diaquin, M., Lamy, B., Arruda, L. K., Punt, P. J., et al. (1993). A transformant of Aspergillus fumigatus deficient in the antigenic cytotoxin ASPFI. FEMS Microbiol. Lett. 111, 31–36. doi: 10.1111/j.1574-6968.1993.tb06357.x
Paris, S., Wysong, D., Debeaupuis, J.-P., Shibuya, K., Philippe, B., Diamond, R. D., et al. (2003). Catalases of Aspergillus fumigatus. Infect. Immun. 71, 3551–3562. doi: 10.1128/IAI.71.6.3551-3562.2003
Park, S. J., Burdick, M. D., Brix, W. K., Stoler, M. H., Askew, D. S., Strieter, R. M., et al. (2010). Neutropenia enhances lung dendritic cell recruitment in response to Aspergillus via a cytokine-to-chemokine amplification loop. J. Immunol. 185, 6190–6197. doi: 10.4049/jimmunol.1002064
Patterson, T. F. (2005). The future of animal models of invasive aspergillosis. Med. Mycol. Off. Publ. Int. Soc. Hum. Anim. Mycol. 43(Suppl. 1), S115–S119. doi: 10.1080/13693780400029429
Patterson, T. F., Miniter, P., Dijkstra, J., Szoka, F. C., Ryan, J. L., and Andriole, V. T. (1989). Treatment of experimental invasive aspergillosis with novel amphotericin B/cholesterol-sulfate complexes. J. Infect. Dis. 159, 717–724. doi: 10.1093/infdis/159.4.717
Patterson, T. F., Miniter, P., Ryan, J. L., and Andriole, V. T. (1988). Effect of immunosuppression and amphotericin B on Aspergillus antigenemia in an experimental model. J. Infect. Dis. 158, 415–422. doi: 10.1093/infdis/158.2.415
Paulussen, C., Boulet, G. A. V., Cos, P., Delputte, P., and Maes, L. J. R. M. (2014). Animal models of invasive aspergillosis for drug discovery. Drug Discov. Today 19, 1380–1386. doi: 10.1016/j.drudis.2014.06.006
Paulussen, C., Boulet, G., Bosschaerts, T., Cos, P., Fortin, A., and Maes, L. (2015). Efficacy of oleylphosphocholine (OlPC) in vitro and in a mouse model of invasive aspergillosis. Mycoses 58, 127–132. doi: 10.1111/myc.12286
Petersen, J. E., Hiran, T. S., Goebel, W. S., Johnson, C., Murphy, R. C., Azmi, F. H., et al. (2002). Enhanced cutaneous inflammatory reactions to Aspergillus fumigatus in a murine model of chronic granulomatous disease. J. Invest. Dermatol. 118, 424–429. doi: 10.1046/j.0022-202x.2001.01691.x
Petraitiene, R., Petraitis, V., Groll, A. H., Sein, T., Schaufele, R. L., Francesconi, A., et al. (2002). Antifungal efficacy of caspofungin (MK-0991) in experimental pulmonary aspergillosis in persistently neutropenic rabbits: pharmacokinetics, drug disposition, and relationship to galactomannan antigenemia. Antimicrob. Agents Chemother. 46, 12–23. doi: 10.1128/AAC.46.1.12-23.2002
Petraitiene, R., Petraitis, V., Lyman, C. A., Groll, A. H., Mickiene, D., Peter, J., et al. (2004). Efficacy, safety, and plasma pharmacokinetics of escalating dosages of intravenously administered ravuconazole lysine phosphoester for treatment of experimental pulmonary aspergillosis in persistently neutropenic rabbits. Antimicrob. Agents Chemother. 48, 1188–1196. doi: 10.1128/AAC.48.4.1188-1196.2004
Petraitis, V., Petraitiene, R., Hope, W. W., Meletiadis, J., Mickiene, D., Hughes, J. E., et al. (2009). Combination therapy in treatment of experimental pulmonary aspergillosis: in vitro and in vivo correlations of the concentration- and dose- dependent interactions between anidulafungin and voriconazole by Bliss independence drug interaction analysis. Antimicrob. Agents Chemother. 53, 2382–2391. doi: 10.1128/AAC.00329-09
Petrik, M., Franssen, G. M., Haas, H., Laverman, P., Hörtnagl, C., Schrettl, M., et al. (2012). Preclinical evaluation of two 68Ga-siderophores as potential radiopharmaceuticals for Aspergillus fumigatus infection imaging. Eur. J. Nucl. Med. Mol. Imaging 39, 1175–1183. doi: 10.1007/s00259-012-2110-3
Petrik, M., Haas, H., Dobrozemsky, G., Lass-Flörl, C., Helbok, A., Blatzer, M., et al. (2010). 68Ga-siderophores for PET imaging of invasive pulmonary aspergillosis: proof of principle. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 51, 639–645. doi: 10.2967/jnumed.109.072462
Petrik, M., Haas, H., Laverman, P., Schrettl, M., Franssen, G. M., Blatzer, M., et al. (2014). 68Ga-triacetylfusarinine C and 68Ga-ferrioxamine E for Aspergillus infection imaging: uptake specificity in various microorganisms. Mol. Imaging Biol. 16, 102–108. doi: 10.1007/s11307-013-0654-7
Philippe, B., Ibrahim-Granet, O., Prévost, M. C., Gougerot-Pocidalo, M. A., Sanchez Perez, M., Van der Meeren, A., et al. (2003). Killing of Aspergillus fumigatus by alveolar macrophages is mediated by reactive oxidant intermediates. Infect. Immun. 71, 3034–3042. doi: 10.1128/IAI.71.6.3034-3042.2003
Pinchai, N., Perfect, B. Z., Juvvadi, P. R., Fortwendel, J. R., Cramer, R. A., Asfaw, Y. G., et al. (2009). Aspergillus fumigatus calcipressin CbpA is involved in hyphal growth and calcium homeostasis. Eukaryot. Cell 8, 511–519. doi: 10.1128/EC.00336-08
Polak, A. (1982). Oxiconazole, a new imidazole derivative. Evaluation of antifungal activity in vitro and in vivo. Arzneimittelforschung 32, 17–24.
Polak, A. (1987). Combination therapy of experimental candidiasis, cryptococcosis, aspergillosis and wangiellosis in mice. Chemotherapy 33, 381–395. doi: 10.1159/000238524
Polak, A., Scholer, H. J., and Wall, M. (1982). Combination therapy of experimental candidiasis, cryptococcosis and aspergillosis in mice. Chemotherapy 28, 461–479. doi: 10.1159/000238138
Polak-Wyss, A. (1991). Protective effect of human granulocyte colony-stimulating factor (hG-CSF) on Cryptococcus and Aspergillus infections in normal and immunosuppressed mice. Mycoses 34, 205–215. doi: 10.1111/j.1439-0507.1991.tb00645.x
Pollock, J. D., Williams, D. A., Gifford, M. A., Li, L. L., Du, X., Fisherman, J., et al. (1995). Mouse model of X-linked chronic granulomatous disease, an inherited defect in phagocyte superoxide production. Nat. Genet. 9, 202–209. doi: 10.1038/ng0295-202
Prüfer, S., Weber, M., Stein, P., Bosmann, M., Stassen, M., Kreft, A., et al. (2014). Oxidative burst and neutrophil elastase contribute to clearance of Aspergillus fumigatus pneumonia in mice. Immunobiology 219, 87–96. doi: 10.1016/j.imbio.2013.08.010
Qiao, J., Kontoyiannis, D. P., Calderone, R., Li, D., Ma, Y., Wan, Z., et al. (2008). Afyap1, encoding a bZip transcriptional factor of Aspergillus fumigatus, contributes to oxidative stress response but is not essential to the virulence of this pathogen in mice immunosuppressed by cyclophosphamide and triamcinolone. Med. Mycol. 46, 773–782. doi: 10.1080/13693780802054215
Rayamajhi, M., Redente, E. F., Condon, T. V., Gonzalez-Juarrero, M., Riches, D. W. H., and Lenz, L. L. (2011). Non-surgical intratracheal instillation of mice with analysis of lungs and lung draining lymph nodes by flow cytometry. J. Vis. Exp. 51:e2702. doi: 10.3791/2702
Rebong, R. A., Santaella, R. M., Goldhagen, B. E., Majka, C. P., Perfect, J. R., Steinbach, W. J., et al. (2011). Polyhexamethylene biguanide and calcineurin inhibitors as novel antifungal treatments for Aspergillus keratitis. Invest. Ophthalmol. Vis. Sci. 52, 7309–7315. doi: 10.1167/iovs.11-7739
Reichard, U., Monod, M., Odds, F., and Rüchel, R. (1997). Virulence of an aspergillopepsin-deficient mutant of Aspergillus fumigatus and evidence for another aspartic proteinase linked to the fungal cell wall. J. Med. Vet. Mycol. Bi-Mon. Publ. Int. Soc. Hum. Anim. Mycol. 35, 189–196.
Ren, S., Zhang, F., Li, C., Jia, C., Li, S., Xi, H., et al. (2010). Selection of housekeeping genes for use in quantitative reverse transcription PCR assays on the murine cornea. Mol. Vis. 16, 1076–1086.
Renshaw, H., Vargas-Muñiz, J. M., Richards, A. D., Asfaw, Y. G., Juvvadi, P. R., and Steinbach, W. J. (2016). Distinct roles of myosins in Aspergillus fumigatus hyphal growth and pathogenesis. Infect. Immun. 84, 1556–1564. doi: 10.1128/IAI.01190-15
Richard, J. L., Dvorak, T. J., and Ross, P. F. (1996). Natural occurrence of gliotoxin in turkeys infected with Aspergillus fumigatus, Fresenius. Mycopathologia 134, 167–170. doi: 10.1007/BF00436725
Richie, D. L., Fuller, K. K., Fortwendel, J., Miley, M. D., McCarthy, J. W., Feldmesser, M., et al. (2007a). Unexpected link between metal ion deficiency and autophagy in Aspergillus fumigatus. Eukaryot. Cell 6, 2437–2447. doi: 10.1128/EC.00224-07
Richie, D. L., Miley, M. D., Bhabhra, R., Robson, G. D., Rhodes, J. C., and Askew, D. S. (2007b). The Aspergillus fumigatus metacaspases CasA and CasB facilitate growth under conditions of endoplasmic reticulum stress. Mol. Microbiol. 63, 591–604. doi: 10.1111/j.1365-2958.2006.05534.x
Riera, C. M., Masih, D. T., and Nobile, R. (1983). Experimental cryptococcosis in guinea pigs. Mycopathologia 82, 179–184. doi: 10.1007/BF00439224
Risovic, V., Rosland, M., Sivak, O., Wasan, K. M., and Bartlett, K. (2007). Assessing the antifungal activity of a new oral lipid-based amphotericin B formulation following administration to rats infected with Aspergillus fumigatus. Drug Dev. Ind. Pharm. 33, 703–707. doi: 10.1080/03639040601077349
Rivera, A., Collins, N., Stephan, M. T., Lipuma, L., Leiner, I., and Pamer, E. G. (2009). Aberrant tissue localization of fungus-specific CD4+ T cells in IL-10-deficient mice. J. Immunol. 183, 631–641. doi: 10.4049/jimmunol.0900396
Rizzetto, L., Giovannini, G., Bromley, M., Bowyer, P., Romani, L., and Cavalieri, D. (2013). Strain dependent variation of immune responses to A. fumigatus: definition of pathogenic species. PLoS ONE 8:e56651. doi: 10.1371/journal.pone.0056651
Roberts, J., Schock, K., Marino, S., and Andriole, V. T. (2000). Efficacies of two new antifungal agents, the triazole ravuconazole and the echinocandin LY-303366, in an experimental model of invasive aspergillosis. Antimicrob. Agents Chemother. 44, 3381–3388. doi: 10.1128/AAC.44.12.3381-3388.2000
Röhm, M., Grimm, M. J., D'Auria, A. C., Almyroudis, N. G., Segal, B. H., and Urban, C. F. (2014). NADPH oxidase promotes neutrophil extracellular trap formation in pulmonary aspergillosis. Infect. Immun. 82, 1766–1777. doi: 10.1128/IAI.00096-14
Rolle, A.-M., Hasenberg, M., Thornton, C. R., Solouk-Saran, D., Männ, L., Weski, J., et al. (2016). ImmunoPET/MR imaging allows specific detection of Aspergillus fumigatus lung infection in vivo. Proc. Natl. Acad. Sci. U.S.A. 113, E1026–E1033. doi: 10.1073/pnas.1518836113
Romani, L., Puccetti, P., Mencacci, A., Cenci, E., Spaccapelo, R., Tonnetti, L., et al. (1994). Neutralization of IL-10 up-regulates nitric oxide production and protects susceptible mice from challenge with Candida albicans. J. Immunol. 152, 3514–3521.
Romano, J., Nimrod, G., Ben-Tal, N., Shadkchan, Y., Baruch, K., Sharon, H., et al. (2006). Disruption of the Aspergillus fumigatus ECM33 homologue results in rapid conidial germination, antifungal resistance and hypervirulence. Microbiol. Read. Engl. 152, 1919–1928. doi: 10.1099/mic.0.28936-0
Ruijgrok, E. J., Fens, M. H. A. M., Bakker-Woudenberg, I. A. J. M., van Etten, E. W. M., and Vulto, A. G. (2006). Nebulized amphotericin B combined with intravenous amphotericin B in rats with severe invasive pulmonary aspergillosis. Antimicrob. Agents Chemother. 50, 1852–1854. doi: 10.1128/AAC.50.5.1852-1854.2006
Ruijgrok, E. J., Fens, M. H. A., Bakker-Woudenberg, I. A. J. M., van Etten, E. W. M., and Vulto, A. G. (2005). Nebulization of four commercially available amphotericin B formulations in persistently granulocytopenic rats with invasive pulmonary aspergillosis: evidence for long-term biological activity. J. Pharm. Pharmacol. 57, 1289–1295. doi: 10.1211/jpp.57.10.0007
Ruijgrok, E. J., Vulto, A. G., and Van Etten, E. W. (2001). Efficacy of aerosolized amphotericin B desoxycholate and liposomal amphotericin B in the treatment of invasive pulmonary aspergillosis in severely immunocompromised rats. J. Antimicrob. Chemother. 48, 89–95. doi: 10.1093/jac/48.1.89
Ruiz-Cabello, J., Regadera, J., Santisteban, C., Graña, M., Pérez de Alejo, R., Echave, I., et al. (2002). Monitoring acute inflammatory processes in mouse muscle by MR imaging and spectroscopy: a comparison with pathological results. NMR Biomed. 15, 204–214. doi: 10.1002/nbm.761
Sable, C. A., Strohmaier, K. M., and Chodakewitz, J. A. (2008). Advances in antifungal therapy. Annu. Rev. Med. 59, 361–379. doi: 10.1146/annurev.med.59.062906.071602
Saeed, E. N., and Hay, R. J. (1981). Immunoperoxidase staining in the recognition of Aspergillus infections. Histopathology 5, 437–444. doi: 10.1111/j.1365-2559.1981.tb01804.x
Salas, V., Pastor, F. J., Calvo, E., Sutton, D. A., Fothergill, A. W., and Guarro, J. (2013). Evaluation of the in vitro activity of voriconazole as predictive of in vivo outcome in a murine Aspergillus fumigatus infection model. Antimicrob. Agents Chemother. 57, 1404–1408. doi: 10.1128/AAC.01331-12
Sandhu, D. K., Sandhu, R. S., Damodaran, V. N., and Randhawa, H. S. (1970). Effect of cortisone on bronchopulmonary aspergillosis in mice exposed to spores of various Aspergillus species. Sabouraudia 8, 32–38. doi: 10.1080/00362177085190601
Sandhu, D. K., Sandhu, R. S., Khan, Z. U., and Damodaran, V. N. (1976). Conditional virulence of a p-aminobenzoic acid-requiring mutant of Aspergillus fumigatus. Infect. Immun. 13, 527–532.
Sarfati, J., Diaquin, M., Debeaupuis, J. P., Schmidt, A., Lecaque, D., Beauvais, A., et al. (2002). A new experimental murine aspergillosis model to identify strains of Aspergillus fumigatus with reduced virulence. Nihon Ishinkin Gakkai Zasshi 43, 203–213. doi: 10.3314/jjmm.43.203
Savers, A., Rasid, O., Parlato, M., Brock, M., Jouvion, G., Ryffel, B., et al. (2016). Infection-mediated priming of phagocytes protects against lethal secondary Aspergillus fumigatus challenge. PLoS ONE 11:e0153829. doi: 10.1371/journal.pone.0153829
Schaffner, A., and Böhler, A. (1993). Amphotericin B refractory aspergillosis after itraconazole: evidence for significant antagonism. Mycoses 36, 421–424. doi: 10.1111/j.1439-0507.1993.tb00732.x
Schaffner, A., and Frick, P. G. (1985). The effect of ketoconazole on amphotericin B in a model of disseminated aspergillosis. J. Infect. Dis. 151, 902–910. doi: 10.1093/infdis/151.5.902
Schaffner, A., Douglas, H., and Braude, A. (1982). Selective protection against conidia by mononuclear and against mycelia by polymorphonuclear phagocytes in resistance to Aspergillus. Observations on these two lines of defense in vivo and in vitro with human and mouse phagocytes. J. Clin. Invest. 69, 617–631. doi: 10.1172/JCI110489
Schaude, M., Petranyi, G., Ackerbauer, H., Meingassner, J. G., and Mieth, H. (1990). Preclinical antimycotic activity of SDZ 89-485: a new orally and topically effective triazole. J. Med. Vet. Mycol. Bi-Mon. Publ. Int. Soc. Hum. Anim. Mycol. 28, 445–454.
Schmidt, A. (2002). Animal models of aspergillosis - also useful for vaccination strategies? Mycoses 45, 38–40.
Sekonyela, R., Palmer, J. M., Bok, J.-W., Jain, S., Berthier, E., Forseth, R., et al. (2013). RsmA regulates Aspergillus fumigatus gliotoxin cluster metabolites including cyclo(L-Phe-L-Ser), a potential new diagnostic marker for invasive aspergillosis. PLoS ONE 8:e62591. doi: 10.1371/journal.pone.0062591
Seyedmousavi, S., Brüggemann, R. J. M., Meis, J. F., Melchers, W. J. G., Verweij, P. E., and Mouton, J. W. (2015). Pharmacodynamics of isavuconazole in an Aspergillus fumigatus mouse infection model. Antimicrob. Agents Chemother. 59, 2855–2866. doi: 10.1128/AAC.04907-14
Seyedmousavi, S., Melchers, W. J. G., Mouton, J. W., and Verweij, P. E. (2013). Pharmacodynamics and dose-response relationships of liposomal amphotericin B against different azole-resistant Aspergillus fumigatus isolates in a murine model of disseminated aspergillosis. Antimicrob. Agents Chemother. 57, 1866–1871. doi: 10.1128/AAC.02226-12
Shao, C., Qu, J., He, L., Zhang, Y., Wang, J., Wang, Y., et al. (2005a). Transient overexpression of gamma interferon promotes Aspergillus clearance in invasive pulmonary aspergillosis. Clin. Exp. Immunol. 142, 233–241. doi: 10.1111/j.1365-2249.2005.02828.x
Shao, C., Qu, J., He, L., Zhang, Y., Wang, J., Zhou, H., et al. (2005b). Dendritic cells transduced with an adenovirus vector encoding interleukin-12 are a potent vaccine for invasive pulmonary aspergillosis. Genes Immun. 6, 103–114. doi: 10.1038/sj.gene.6364167
Shepardson, K. M., Jhingran, A., Caffrey, A., Obar, J. J., Suratt, B. T., Berwin, B. L., et al. (2014). Myeloid derived hypoxia inducible factor 1-alpha is required for protection against pulmonary Aspergillus fumigatus infection. PLoS Pathog. 10:e1004378. doi: 10.1371/journal.ppat.1004378
Shepardson, K. M., Ngo, L. Y., Aimanianda, V., Latgé, J.-P., Barker, B. M., Blosser, S. J., et al. (2013). Hypoxia enhances innate immune activation to Aspergillus fumigatus through cell wall modulation. Microbes Infect. Inst. Pasteur 15, 259–269. doi: 10.1016/j.micinf.2012.11.010
Sheppard, D. C., Graybill, J. R., Najvar, L. K., Chiang, L. Y., Doedt, T., Kirkpatrick, W. R., et al. (2006). Standardization of an experimental murine model of invasive pulmonary aspergillosis. Antimicrob. Agents Chemother. 50, 3501–3503. doi: 10.1128/AAC.00787-06
Sheppard, D. C., Rieg, G., Chiang, L. Y., Filler, S. G., Edwards, J. E. Jr., and Ibrahim, A. S. (2004). Novel inhalational murine model of invasive pulmonary aspergillosis. Antimicrob. Agents Chemother. 48, 1908–1911. doi: 10.1128/AAC.48.5.1908-1911.2004
Shibuya, K., Takaoka, M., Uchida, K., Wakayama, M., Yamaguchi, H., Takahashi, K., et al. (1999). Histopathology of experimental invasive pulmonary aspergillosis in rats: pathological comparison of pulmonary lesions induced by specific virulent factor deficient mutants. Microb. Pathog. 27, 123–131. doi: 10.1006/mpat.1999.0288
Shirkhani, K., Teo, I., Armstrong-James, D., and Shaunak, S. (2015). Nebulised amphotericin B-polymethacrylic acid nanoparticle prophylaxis prevents invasive aspergillosis. Nanomedicine Nanotechnol. Biol. Med. 11, 1217–1226. doi: 10.1016/j.nano.2015.02.012
Singh, G., Imai, J., Clemons, K. V., and Stevens, D. A. (2005). Efficacy of caspofungin against central nervous system Aspergillus fumigatus infection in mice determined by TaqMan PCR and CFU methods. Antimicrob. Agents Chemother. 49, 1369–1376. doi: 10.1128/AAC.49.4.1369-1376.2005
Singh, M., Madan, T., Waters, P., Sonar, S., Singh, S. K., Kamran, M. F., et al. (2009). Therapeutic effects of recombinant forms of full-length and truncated human surfactant protein D in a murine model of invasive pulmonary aspergillosis. Mol. Immunol. 46, 2363–2369. doi: 10.1016/j.molimm.2009.03.019
Singh, S. M., Sharma, S., and Chatterjee, P. K. (1990). Clinical and experimental mycotic keratitis caused by Aspergillus terreus and the effect of subconjunctival oxiconazole treatment in the animal model. Mycopathologia 112, 127–137. doi: 10.1007/BF00436642
Singh, S., Dabur, R., Gatne, M. M., Singh, B., Gupta, S., Pawar, S., et al. (2014). In vivo efficacy of a synthetic coumarin derivative in a murine model of aspergillosis. PLoS ONE 9:e103039. doi: 10.1371/journal.pone.0103039
Sivak, O., Bartlett, K., and Wasan, K. M. (2004a). Heat-treated Fungizone retains amphotericin B antifungal activity without renal toxicity in rats infected with Aspergillus fumigatus. Pharm. Res. 21, 1564–1566. doi: 10.1023/B:PHAM.0000041449.46054.b4
Sivak, O., Bartlett, K., Risovic, V., Choo, E., Marra, F., Batty, D. S., et al. (2004b). Assessing the antifungal activity and toxicity profile of amphotericin B lipid complex (ABLC; Abelcet) in combination with caspofungin in experimental systemic aspergillosis. J. Pharm. Sci. 93, 1382–1389.
Slesiona, S., Gressler, M., Mihlan, M., Zaehle, C., Schaller, M., Barz, D., et al. (2012). Persistence versus escape: Aspergillus terreus and Aspergillus fumigatus employ different strategies during interactions with macrophages. PLoS ONE 7:e31223. doi: 10.1371/journal.pone.0031223
Smith, G. R. (1972). Experimental aspergillosis in mice: aspects of resistance. J. Hyg. 70, 741–754. doi: 10.1017/S0022172400022580
Smith, G. R. (1973). Effect of sub-lethal treatment with formalin on the germination of Aspergillus fumigatus spores. J. Hyg. 71, 745–753. doi: 10.1017/S0022172400023007
Smith, G. R. (1977). Aspergillus fumigatus: a possible relationship between spore size and virulence for mice. J. Gen. Microbiol. 102, 413–415. doi: 10.1099/00221287-102-2-413
Smith, J. M., Davies, J. E., and Holden, D. W. (1993). Construction and pathogenicity of Aspergillus fumigatus mutants that do not produce the ribotoxin restrictocin. Mol. Microbiol. 9, 1071–1077. doi: 10.1111/j.1365-2958.1993.tb01236.x
Smith, J. M., Tang, C. M., Van Noorden, S., and Holden, D. W. (1994). Virulence of Aspergillus fumigatus double mutants lacking restriction and an alkaline protease in a low-dose model of invasive pulmonary aspergillosis. Infect. Immun. 62, 5247–5254.
Speth, C., Hagleitner, M., Ott, H. W., Würzner, R., Lass-Flörl, C., and Rambach, G. (2013). Aspergillus fumigatus activates thrombocytes by secretion of soluble compounds. J. Infect. Dis. 207, 823–833. doi: 10.1093/infdis/jis743
Spikes, S., Xu, R., Nguyen, C. K., Chamilos, G., Kontoyiannis, D. P., Jacobson, R. H., et al. (2008). Gliotoxin production in Aspergillus fumigatus contributes to host-specific differences in virulence. J. Infect. Dis. 197, 479–486. doi: 10.1086/525044
Steele, C., and Wormley, F. L. (2012). Immunology of fungal infections: lessons learned from animal models. Curr. Opin. Microbiol. 15, 413–419. doi: 10.1016/j.mib.2012.05.017
Steele, C., Rapaka, R. R., Metz, A., Pop, S. M., Williams, D. L., Gordon, S., et al. (2005). The beta-glucan receptor dectin-1 recognizes specific morphologies of Aspergillus fumigatus. PLoS Pathog. 1:e42. doi: 10.1371/journal.ppat.0010042
Stein, S., Scholz, S., Schwäble, J., Sadat, M. A., Modlich, U., Schultze-Strasser, S., et al. (2013). From bench to bedside: preclinical evaluation of a self-inactivating gammaretroviral vector for the gene therapy of X-linked chronic granulomatous disease. Hum. Gene Ther. Clin. Dev. 24, 86–98. doi: 10.1089/humc.2013.019
Steinbach, W. J., Benjamin, D. K. Jr., Trasi, S. A., Miller, J. L., Schell, W. A., Zaas, A. K., et al. (2004). Value of an inhalational model of invasive aspergillosis. Med. Mycol. Off. Publ. Int. Soc. Hum. Anim. Mycol. 42, 417–425.
Steinbach, W. J., Cramer, R. A., Perfect, B. Z., Asfaw, Y. G., Sauer, T. C., Najvar, L. K., et al. (2006). Calcineurin controls growth, morphology, and pathogenicity in Aspergillus fumigatus. Eukaryot. Cell 5, 1091–1103. doi: 10.1128/EC.00139-06
Stephens-Romero, S. D., Mednick, A. J., and Feldmesser, M. (2005). The pathogenesis of fatal outcome in murine pulmonary aspergillosis depends on the neutrophil depletion strategy. Infect. Immun. 73, 114–125. doi: 10.1128/IAI.73.1.114-125.2005
Stojanovic, I., Mirkov, I., Kataranovski, M., Glamoclija, J., and Stosic-Grujicic, S. (2011). A role for macrophage migration inhibitory factor in protective immunity against Aspergillus fumigatus. Immunobiology 216, 1018–1027. doi: 10.1016/j.imbio.2011.03.005
Sugar, A. M., and Liu, X. P. (2000). Effect of grapefruit juice on serum voriconazole concentrations in the mouse. Med. Mycol. 38, 209–212. doi: 10.1080/mmy.38.3.209.212
Sugui, J. A., Losada, L., Wang, W., Varga, J., Ngamskulrungroj, P., Abu-Asab, M., et al. (2011). Identification and characterization of an Aspergillus fumigatus “supermater” pair. mBio 2:e00234-11. doi: 10.1128/mBio.00234-11
Sugui, J. A., Pardo, J., Chang, Y. C., Müllbacher, A., Zarember, K. A., Galvez, E. M., et al. (2007a). Role of laeA in the regulation of alb1, gliP, conidial morphology, and virulence in Aspergillus fumigatus. Eukaryot. Cell 6, 1552–1561. doi: 10.1128/EC.00140-07
Sugui, J. A., Pardo, J., Chang, Y. C., Zarember, K. A., Nardone, G., Galvez, E. M., et al. (2007b). Gliotoxin is a virulence factor of Aspergillus fumigatus: gliP deletion attenuates virulence in mice immunosuppressed with hydrocortisone. Eukaryot. Cell 6, 1562–1569. doi: 10.1128/EC.00141-07
Sugui, J. A., Peterson, S. W., Clark, L. P., Nardone, G., Folio, L., Riedlinger, G., et al. (2012). Aspergillus tanneri sp. nov., a new pathogen that causes invasive disease refractory to antifungal therapy. J. Clin. Microbiol. 50, 3309–3317. doi: 10.1128/JCM.01509-12
Sugui, J. A., Peterson, S. W., Figat, A., Hansen, B., Samson, R. A., Mellado, E., et al. (2014). Genetic relatedness versus biological compatibility between Aspergillus fumigatus and related species. J. Clin. Microbiol. 52, 3707–3721. doi: 10.1128/JCM.01704-14
Sugui, J. A., Vinh, D. C., Nardone, G., Shea, Y. R., Chang, Y. C., Zelazny, A. M., et al. (2010). Neosartorya udagawae (Aspergillus udagawae), an emerging agent of aspergillosis: how different is it from Aspergillus fumigatus? J. Clin. Microbiol. 48, 220–228. doi: 10.1128/JCM.01556-09
Suleiman, M. M., Duncan, N., Eloff, J. N., and Naidoo, V. (2012). A controlled study to determine the efficacy of Loxostylis alata (Anacardiaceae) in the treatment of aspergillus in a chicken (Gallus domesticus) model in comparison to ketoconazole. BMC Vet. Res. 8:210. doi: 10.1186/1746-6148-8-210
Sun, Z., Zhu, P., Li, L., Wan, Z., Zhao, Z., and Li, R. (2012). Adoptive immunity mediated by HLA-A*0201 restricted Asp f16 peptides-specific CD8+ T cells against Aspergillus fumigatus infection. Eur. J. Clin. Microbiol. Infect. Dis. Off. Publ. Eur. Soc. Clin. Microbiol. 31, 3089–3096. doi: 10.1007/s10096-012-1670-2
Takazono, T., Izumikawa, K., Mihara, T., Kosai, K., Saijo, T., Imamura, Y., et al. (2009). Efficacy of combination antifungal therapy with intraperitoneally administered micafungin and aerosolized liposomal amphotericin B against murine invasive pulmonary aspergillosis. Antimicrob. Agents Chemother. 53, 3508–3510. doi: 10.1128/AAC.00285-09
Takemoto, K., Yamamoto, Y., Ueda, Y., Sumita, Y., Yoshida, K., and Niki, Y. (2004). Comparative studies on the efficacy of AmBisome and Fungizone in a mouse model of disseminated aspergillosis. J. Antimicrob. Chemother. 53, 311–317. doi: 10.1093/jac/dkh055
Takemoto, K., Yamamoto, Y., Ueda, Y., Sumita, Y., Yoshida, K., and Niki, Y. (2006). Comparative study on the efficacy of AmBisome and Fungizone in a mouse model of pulmonary aspergillosis. J. Antimicrob. Chemother. 57, 724–731. doi: 10.1093/jac/dkl005
Tandon, R. N., Feuillette, A. R., Mahouy, G., Badillet, G., Friedman, R. M., and Maheshwari, R. K. (1988). Interferon protects mice against an extracellular infection of Aspergillus fumigatus. Ann. N.Y. Acad. Sci. 544, 409–411. doi: 10.1111/j.1749-6632.1988.tb40437.x
Tang, C. M., Cohen, J., Krausz, T., Van Noorden, S., and Holden, D. W. (1993). The alkaline protease of Aspergillus fumigatus is not a virulence determinant in two murine models of invasive pulmonary aspergillosis. Infect. Immun. 61, 1650–1656.
Tansho, S., Abe, S., Ishibashi, H., Torii, S., Otani, H., Ono, Y., et al. (2006). Efficacy of intravenous itraconazole against invasive pulmonary aspergillosis in neutropenic mice. J. Infect. Chemother. Off. J. Jpn. Soc. Chemother. 12, 355–362.
Taylor, P. R., Leal, S. M., Sun, Y., and Pearlman, E. (2014a). Aspergillus and Fusarium corneal infections are regulated by Th17 cells and IL-17-producing neutrophils. J. Immunol. 192, 3319–3327. doi: 10.4049/jimmunol.1302235
Taylor, P. R., Roy, S., Leal, S. M., Sun, Y., Howell, S. J., Cobb, B. A., et al. (2014b). Activation of neutrophils by autocrine IL-17A-IL-17RC interactions during fungal infection is regulated by IL-6, IL-23, RORγt and dectin-2. Nat. Immunol. 15, 143–151. doi: 10.1038/ni.2797
te Dorsthorst, D. T. A., Verweij, P. E., Meis, J. F. G. M., and Mouton, J. W. (2005). Efficacy and pharmacodynamics of flucytosine monotherapy in a nonneutropenic murine model of invasive aspergillosis. Antimicrob. Agents Chemother. 49, 4220–4226. doi: 10.1128/AAC.49.10.4220-4226.2005
Templeton, S. P., Buskirk, A. D., Law, B., Green, B. J., and Beezhold, D. H. (2011). Role of germination in murine airway CD8+ T-cell responses to Aspergillus conidia. PLoS ONE 6:e18777. doi: 10.1371/journal.pone.0018777
Thau, N., Monod, M., Crestani, B., Rolland, C., Tronchin, G., Latgé, J. P., et al. (1994). Rodletless mutants of Aspergillus fumigatus. Infect. Immun. 62, 4380–4388.
Tsitsigiannis, D. I., Bok, J.-W., Andes, D., Nielsen, K. F., Frisvad, J. C., and Keller, N. P. (2005). Aspergillus cyclooxygenase-like enzymes are associated with prostaglandin production and virulence. Infect. Immun. 73, 4548–4559. doi: 10.1128/IAI.73.8.4548-4559.2005
Turner, K. J., Hackshaw, R., Papadimitriou, J., and Perrott, J. (1976). The pathogenesis of experimental pulmonary aspergillosis in normal and cortisone-treated rats. J. Pathol. 118, 65–73. doi: 10.1002/path.1711180202
Turner, K. J., Hackshaw, R., Papadimitriou, J., Wetherall, J. D., and Perrott, J. (1975a). Experimental aspergillosis in rats infected via intraperitoneal and subcutaneous routes. Immunology 29, 55–66.
Turner, K. J., Papadimitriou, J., Hackshaw, R., and Wetherall, J. D. (1975b). Experimental aspergillosis in normal rats infected intravenously. J. Reticuloendothel. Soc. 17, 300–312.
Ullmann, A. J., Krammes, E., Sommer, S., Buschmann, I., Jahn-Muehl, B., Cacciapuoti, A., et al. (2007). Efficacy of posaconazole and amphotericin B in experimental invasive pulmonary aspergillosis in dexamethasone immunosuppressed rats. J. Antimicrob. Chemother. 60, 1080–1084. doi: 10.1093/jac/dkm328
Upton, A., Kirby, K. A., Carpenter, P., Boeckh, M., and Marr, K. A. (2007). Invasive aspergillosis following hematopoietic cell transplantation: outcomes and prognostic factors associated with mortality. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 44, 531–540. doi: 10.1086/510592
Vallor, A. C., Kirkpatrick, W. R., Najvar, L. K., Bocanegra, R., Kinney, M. C., Fothergill, A. W., et al. (2008). Assessment of Aspergillus fumigatus burden in pulmonary tissue of guinea pigs by quantitative PCR, galactomannan enzyme immunoassay, and quantitative culture. Antimicrob. Agents Chemother. 52, 2593–2598. doi: 10.1128/AAC.00276-08
Van Cutsem, J., Meulemans, L., Van Gerven, F., and Stynen, D. (1990). Detection of circulating galactomannan by Pastorex Aspergillus in experimental invasive aspergillosis. Mycoses 33, 61–69.
Van Cutsem, J., Van Gerven, F., and Janssen, P. A. (1987). Activity of orally, topically, and parenterally administered itraconazole in the treatment of superficial and deep mycoses: animal models. Rev. Infect. Dis. 9(Suppl. 1), S15–S32. doi: 10.1093/clinids/9.supplement_1.s15
Van Cutsem, J., Van Gerven, F., and Janssen, P. A. (1989). Oral and parenteral therapy with saperconazole (R 66905) of invasive aspergillosis in normal and immunocompromised animals. Antimicrob. Agents Chemother. 33, 2063–2068. doi: 10.1128/AAC.33.12.2063
Van Cutsem, J., Van Gerven, F., Van de Ven, M. A., Borgers, M., and Janssen, P. A. (1984). Itraconazole, a new triazole that is orally active in aspergillosis. Antimicrob. Agents Chemother. 26, 527–534. doi: 10.1128/AAC.26.4.527
Van Etten, E. W., Stearne-Cullen, L. E., ten Kate, M., and Bakker-Woudenberg, I. A. (2000). Efficacy of liposomal amphotericin B with prolonged circulation in blood in treatment of severe pulmonary aspergillosis in leukopenic rats. Antimicrob. Agents Chemother. 44, 540–545. doi: 10.1128/AAC.44.3.540-545.2000
van Vianen, W., de Marie, S., ten Kate, M. T., Mathot, R. A. A., and Bakker-Woudenberg, I. A. J. M. (2006). Caspofungin: antifungal activity in vitro, pharmacokinetics, and effects on fungal load and animal survival in neutropenic rats with invasive pulmonary aspergillosis. J. Antimicrob. Chemother. 57, 732–740. doi: 10.1093/jac/dkl015
Vargas-Muñiz, J. M., Renshaw, H., Richards, A. D., Lamoth, F., Soderblom, E. J., Moseley, M. A., et al. (2015). The Aspergillus fumigatus septins play pleiotropic roles in septation, conidiation, and cell wall stress, but are dispensable for virulence. Fungal Genet. Biol. 81, 41–51. doi: 10.1016/j.fgb.2015.05.014
Verweij, P. E., Oakley, K. L., Morrissey, J., Morrissey, G., and Denning, D. W. (1998). Efficacy of LY303366 against amphotericin B-susceptible and -resistant Aspergillus fumigatus in a murine model of invasive aspergillosis. Antimicrob. Agents Chemother. 42, 873–878.
Verweij, P. E., Te Dorsthorst, D. T. A., Janssen, W. H. P., Meis, J. F. G. M., and Mouton, J. W. (2008). In vitro activities at pH 5.0 and pH 7.0 and in vivo efficacy of flucytosine against Aspergillus fumigatus. Antimicrob. Agents Chemother. 52, 4483–4485. doi: 10.1128/AAC.00491-08
Verwer, P. E. B., ten Kate, M. T., Falcone, F. H., Morroll, S., Verbrugh, H. A., Bakker-Woudenberg, I. A. J. M., et al. (2013). Evidence supporting a role for mammalian chitinases in efficacy of caspofungin against experimental aspergillosis in immunocompromised rats. PLoS ONE 8:e75848. doi: 10.1371/journal.pone.0075848
Vethanayagam, R. R., Almyroudis, N. G., Grimm, M. J., Lewandowski, D. C., Pham, C. T. N., Blackwell, T. S., et al. (2011). Role of NADPH oxidase versus neutrophil proteases in antimicrobial host defense. PLoS ONE 6:e28149. doi: 10.1371/journal.pone.0028149
Wagener, J., Echtenacher, B., Rohde, M., Kotz, A., Krappmann, S., Heesemann, J., et al. (2008). The putative alpha-1,2-mannosyltransferase AfMnt1 of the opportunistic fungal pathogen Aspergillus fumigatus is required for cell wall stability and full virulence. Eukaryot. Cell 7, 1661–1673. doi: 10.1128/EC.00221-08
Waldorf, A. R., Levitz, S. M., and Diamond, R. D. (1984). In vivo bronchoalveolar macrophage defense against Rhizopus oryzae and Aspergillus fumigatus. J. Infect. Dis. 150, 752–760. doi: 10.1093/infdis/150.5.752
Wallace, T. L., Paetznick, V., Cossum, P. A., Lopez-Berestein, G., Rex, J. H., and Anaissie, E. (1997). Activity of liposomal nystatin against disseminated Aspergillus fumigatus infection in neutropenic mice. Antimicrob. Agents Chemother. 41, 2238–2243.
Walsh, T. J., Garrett, K., Feurerstein, E., Girton, M., Allende, M., Bacher, J., et al. (1995). Therapeutic monitoring of experimental invasive pulmonary aspergillosis by ultrafast computerized tomography, a novel, noninvasive method for measuring responses to antifungal therapy. Antimicrob. Agents Chemother. 39, 1065–1069. doi: 10.1128/AAC.39.5.1065
Walzl, H. L., Ackerbauer, H., Meingassner, J. G., and Mieth, H. (1987). Histopathology of organ lesions in mice after an intravenous or intratracheal or intrarenal infection with Aspergillus fumigatus. Mykosen 30, 10–18. doi: 10.1111/j.1439-0507.1987.tb03819.x
Wang, D.-N., Toyotome, T., Muraosa, Y., Watanabe, A., Wuren, T., Bunsupa, S., et al. (2014). GliA in Aspergillus fumigatus is required for its tolerance to gliotoxin and affects the amount of extracellular and intracellular gliotoxin. Med. Mycol. 52, 506–518. doi: 10.1093/mmy/myu007
Warn, P. A., Morrissey, G., Morrissey, J., and Denning, D. W. (2003). Activity of micafungin (FK463) against an itraconazole-resistant strain of Aspergillus fumigatus and a strain of Aspergillus terreus demonstrating in vivo resistance to amphotericin B. J. Antimicrob. Chemother. 51, 913–919. doi: 10.1093/jac/dkg185
Warn, P. A., Sharp, A., Morrissey, G., and Denning, D. W. (2010). Activity of aminocandin (IP960; HMR3270) compared with amphotericin B, itraconazole, caspofungin and micafungin in neutropenic murine models of disseminated infection caused by itraconazole-susceptible and -resistant strains of Aspergillus fumigatus. Int. J. Antimicrob. Agents 35, 146–151. doi: 10.1016/j.ijantimicag.2009.09.029
Warn, P. A., Sharp, A., Mosquera, J., Spickermann, J., Schmitt-Hoffmann, A., Heep, M., et al. (2006). Comparative in vivo activity of BAL4815, the active component of the prodrug BAL8557, in a neutropenic murine model of disseminated Aspergillus flavus. J. Antimicrob. Chemother. 58, 1198–1207. doi: 10.1093/jac/dkl396
Wasan, E. K., Bartlett, K., Gershkovich, P., Sivak, O., Banno, B., Wong, Z., et al. (2009). Development and characterization of oral lipid-based amphotericin B formulations with enhanced drug solubility, stability and antifungal activity in rats infected with Aspergillus fumigatus or Candida albicans. Int. J. Pharm. 372, 76–84. doi: 10.1016/j.ijpharm.2009.01.003
Wasan, K. M., Sivak, O., Rosland, M., Risovic, V., and Bartlett, K. (2007). Assessing the antifungal activity, pharmacokinetics, and tissue distribution of amphotericin B following the administration of Abelcet and AmBisome in combination with caspofungin to rats infected with Aspergillus fumigatus. J. Pharm. Sci. 96, 1737–1747. doi: 10.1002/jps.20801
Wharton, R. E., Stefanov, E. K., King, R. G., and Kearney, J. F. (2015). Antibodies generated against Streptococci protect in a mouse model of disseminated aspergillosis. J. Immunol. 194, 4387–4396. doi: 10.4049/jimmunol.1401940
White, L. O. (1977). Germination of Aspergillus fumigatus conidia in the lungs of normal and cortisone-treated mice. Sabouraudia 15, 37–41. doi: 10.1080/00362177785190071
White, P. L., Wiederhold, N. P., Loeffler, J., Najvar, L. K., Melchers, W., Herrera, M., et al. (2016). Comparison of nonculture blood-based tests for diagnosing invasive aspergillosis in an animal model. J. Clin. Microbiol. 54, 960–966. doi: 10.1128/JCM.03233-15
Wiederhold, N. P., Kontoyiannis, D. P., Chi, J., Prince, R. A., Tam, V. H., and Lewis, R. E. (2004). Pharmacodynamics of caspofungin in a murine model of invasive pulmonary aspergillosis: evidence of concentration-dependent activity. J. Infect. Dis. 190, 1464–1471. doi: 10.1086/424465
Wiederhold, N. P., Najvar, L. K., Bocanegra, R., Kirkpatrick, W. R., Patterson, T. F., and Thornton, C. R. (2013). Interlaboratory and interstudy reproducibility of a novel lateral-flow device and influence of antifungal therapy on detection of invasive pulmonary aspergillosis. J. Clin. Microbiol. 51, 459–465. doi: 10.1128/JCM.02142-12
Wiederhold, N. P., Najvar, L. K., Matsumoto, S., Bocanegra, R. A., Herrera, M. L., Wickes, B. L., et al. (2015). Efficacy of the investigational echinocandin ASP9726 in a guinea pig model of invasive pulmonary aspergillosis. Antimicrob. Agents Chemother. 59, 2875–2881. doi: 10.1128/AAC.04857-14
Wiederhold, N. P., Thornton, C. R., Najvar, L. K., Kirkpatrick, W. R., Bocanegra, R., and Patterson, T. F. (2009). Comparison of lateral flow technology and galactomannan and (1->3)-beta-D-glucan assays for detection of invasive pulmonary aspergillosis. Clin. Vaccine Immunol. 16, 1844–1846. doi: 10.1128/CVI.00268-09
Williams, D. M., Weiner, M. H., and Drutz, D. J. (1981). Immunologic studies of disseminated infection with Aspergillus fumigatus in the nude mouse. J. Infect. Dis. 143, 726–733. doi: 10.1093/infdis/143.5.726
Wong, B., Brauer, K. L., Tsai, R. R., and Jayasimhulu, K. (1989). Increased amounts of the Aspergillus metabolite D-mannitol in tissue and serum of rats with experimental aspergillosis. J. Infect. Dis. 160, 95–103. doi: 10.1093/infdis/160.1.95
Wüthrich, M., Deepe, G. S., and Klein, B. (2012). Adaptive immunity to fungi. Annu. Rev. Immunol. 30, 115–148. doi: 10.1146/annurev-immunol-020711-074958
Xu, Q., Zhao, G., Lin, J., Wang, Q., Hu, L., and Jiang, Z. (2015). Role of Dectin-1 in the innate immune response of rat corneal epithelial cells to Aspergillus fumigatus. BMC Ophthalmol. 15:126. doi: 10.1186/s12886-015-0112-1
Yamada, H., Tsuda, T., Watanabe, T., Ohashi, M., Murakami, K., and Mochizuki, H. (1993). In vitro and in vivo antifungal activities of D0870, a new triazole agent. Antimicrob. Agents Chemother. 37, 2412–2417. doi: 10.1128/AAC.37.11.2412
Yamakami, Y., Hashimoto, A., Tokimatsu, I., and Nasu, M. (1996). PCR detection of DNA specific for Aspergillus species in serum of patients with invasive aspergillosis. J. Clin. Microbiol. 34, 2464–2468.
Yan, Y., Zhao, Z., Wan, H., Wu, R., Fang, J., and Liu, H. (2014). A novel fungus concentration-dependent rat model for acute invasive fungal rhinosinusitis: an experimental study. BMC Infect. Dis. 14:3856. doi: 10.1186/s12879-014-0713-y
Yang, Z., Kontoyiannis, D. P., Wen, X., Xiong, C., Zhang, R., Albert, N. D., et al. (2009). Gamma scintigraphy imaging of murine invasive pulmonary aspergillosis with a (111)In-labeled cyclic peptide. Nucl. Med. Biol. 36, 259–266. doi: 10.1016/j.nucmedbio.2008.12.004
Yonezawa, M., Sugiyama, H., Kizawa, K., Hori, R., Mitsuyama, J., Araki, H., et al. (2000). A new model of pulmonary superinfection with Aspergillus fumigatus and Pseudomonas aeruginosa in mice. J. Infect. Chemother. Off. J. Jpn. Soc. Chemother. 6, 155–161. doi: 10.1007/s101560000005
Yu, B., Niki, Y., and Armstrong, D. (1990). Use of immunoblotting to detect Aspergillus fumigatus antigen in sera and urines of rats with experimental invasive aspergillosis. J. Clin. Microbiol. 28, 1575–1579.
Zaas, A. K., Liao, G., Chien, J. W., Weinberg, C., Shore, D., Giles, S. S., et al. (2008). Plasminogen alleles influence susceptibility to invasive aspergillosis. PLoS Genet. 4:e1000101. doi: 10.1371/journal.pgen.1000101
Zelante, T., Bozza, S., De Luca, A., D'Angelo, C., Bonifazi, P., Moretti, S., et al. (2009). Th17 cells in the setting of Aspergillus infection and pathology. Med. Mycol. 47(Suppl. 1), S162–S169. doi: 10.1080/13693780802140766
Zelante, T., De Luca, A., Bonifazi, P., Montagnoli, C., Bozza, S., Moretti, S., et al. (2007). IL-23 and the Th17 pathway promote inflammation and impair antifungal immune resistance. Eur. J. Immunol. 37, 2695–2706. doi: 10.1002/eji.200737409
Zelante, T., Wong, A. Y. W., Ping, T. J., Chen, J., Sumatoh, H. R., Viganò, E., et al. (2015). CD103(+) dendritic cells control Th17 cell function in the lung. Cell Rep. 12, 1789–1801. doi: 10.1016/j.celrep.2015.08.030
Zhang, C., Kong, Q., Cai, Z., Liu, F., Chen, P., Song, J., et al. (2015). The newly nonsporulated characterization of an Aspergillus fumigatus isolate from an immunocompetent patient and its clinic indication. Fungal Genet. Biol. 81, 250–260. doi: 10.1016/j.fgb.2015.03.001
Zhang, F., An, Y., Li, Z., and Zhao, C. (2013). A novel model of invasive fungal rhinosinusitis in rats. Am. J. Rhinol. Allergy 27, 361–366. doi: 10.2500/ajra.2013.27.3953
Zhang, M., Su, X., Sun, W.-K., Chen, F., Xu, X.-Y., and Shi, Y. (2014). Efficacy of the combination of voriconazole and caspofungin in experimental pulmonary aspergillosis by different Aspergillus species. Mycopathologia 177, 11–18. doi: 10.1007/s11046-013-9719-z
Zhao, J., Cheng, Y., Song, X., Wang, C., Su, G., and Liu, Z. (2015). A comparative treatment study of intravitreal voriconazole and liposomal amphotericin B in an Aspergillus fumigatus endophthalmitis model. Invest. Ophthalmol. Vis. Sci. 56, 7369–7376. doi: 10.1167/iovs.15-17266
Zhao, Y., and Perlin, D. S. (2013). Quantitative detection of Aspergillus spp. by real-time nucleic acid sequence-based amplification. Methods Mol. Biol. 968, 83–92. doi: 10.1007/978-1-62703-257-5_6
Zhao, Y., Park, S., Warn, P., Shrief, R., Harrison, E., and Perlin, D. S. (2010). Detection of Aspergillus fumigatus in a rat model of invasive pulmonary aspergillosis by real-time nucleic acid sequence-based amplification. J. Clin. Microbiol. 48, 1378–1383. doi: 10.1128/JCM.02214-09
Zhong, J., Huang, W., Deng, Q., Wu, M., Jiang, H., Lin, X., et al. (2016). Inhibition of TREM-1 and Dectin-1 alleviates the severity of fungal keratitis by modulating innate immune responses. PLoS ONE 11:e0150114. doi: 10.1371/journal.pone.0150114
Zhong, W., Yin, H., and Xie, L. (2009). Expression and potential role of major inflammatory cytokines in experimental keratomycosis. Mol. Vis. 15, 1303–1311.
Keywords: invasive aspergillosis, Aspergillus fumigatus, rodent models, mice, experimental infection
Citation: Desoubeaux G and Cray C (2017) Rodent Models of Invasive Aspergillosis due to Aspergillus fumigatus: Still a Long Path toward Standardization. Front. Microbiol. 8:841. doi: 10.3389/fmicb.2017.00841
Received: 17 March 2017; Accepted: 24 April 2017;
Published: 16 May 2017.
Edited by:
Miguel Cacho Teixeira, Universidade de Lisboa, PortugalReviewed by:
Anna Dongari-Bagtzoglou, University of Connecticut Health Center, USAIsabel M. Miranda, University of Porto, Portugal
Copyright © 2017 Desoubeaux and Cray. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Guillaume Desoubeaux, Z3VpbGxhdW1lLmRlc291YmVhdXhAdW5pdi10b3Vycy5mcg==
 Guillaume Desoubeaux
Guillaume Desoubeaux Carolyn Cray1
Carolyn Cray1