Abstract
The microRNAs (miRNAs) function as a key regulator in many biological processes through post-transcriptional suppression of messenger RNAs. Recent advancements have revealed that miRNAs are involved in many biological functions of cells. Not only host cells, but also some viruses encode miRNAs in their genomes. Viral miRNAs regulate cell proliferation, differentiation, apoptosis, and the cell cycle to establish infection and produce viral progeny. Particularly, miRNAs encoded by herpes virus families play integral roles in persistent viral infection either by regulation of metabolic processes or the immune response of host cells. The life-long persistent infection of gamma herpes virus subfamilies, such as Epstein-Barr virus and Kaposi's sarcoma-associated herpesvirus, induces host cells to malignant transformation. The unbalanced metabolic processes and evasion from host immune surveillance by viral miRNAs are induced either by direct targeting of key proteins or indirect regulation of multiple signaling pathways. We provide an overview of the pathogenic roles of viral miRNAs in cellular metabolism and immune responses during herpesvirus infection.
Introduction
The microRNAs (miRNAs) are small non-coding RNAs consisting of 19–23 nucleotides. The primary miRNA in the nucleus is cleaved into smaller pre-miRNA consisting of around 70 nucleotides with a hairpin structure. The pre-miRNA is then exported to the cytoplasm, where it is cleaved by Dicer to form mature miRNA. The miRNA is incorporated into the RNA-induced silencing complex (RISC), which contains the essential endonuclease Argonaute 2 (Ago2). The miRNA-RISC interacts with the 3′ untranslated region (UTR) in mRNA. This complex suppresses target gene expression through the translational repression or induction of mRNA deadenylation (Winter et al., 2009). Cellular miRNAs play important parts in the regulation of cellular pathways, of which dysregulation has been linked to many disorders including cancer. Viral miRNAs were reported originally by Pfeffer et al. (2004), and now many DNA viruses are known to contain miRNAs in their genomes. More than 200 viral miRNAs are currently identified mainly in the herpesvirus family (Table 1).
Table 1
| Viruses | miRNAs | Targets | Target sites (Upper: Target, Lower: miRNA) | Function | References |
|---|---|---|---|---|---|
| EBV | miR-BART16 | LMP1 |  | Latent membrane protein that mimics CD40 and induces NF-κB activation | Lo et al., 2007 |
 | |||||
| miR-BART17-5p | LMP1 | 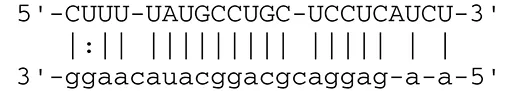 | Latent membrane protein that mimics CD40 and induces NF-κB activation | Lo et al., 2007 | |
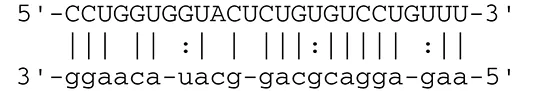 | |||||
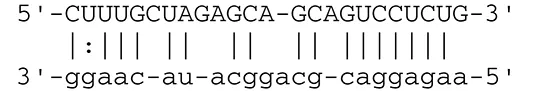 | |||||
| miR-BART1-5p | LMP1 | 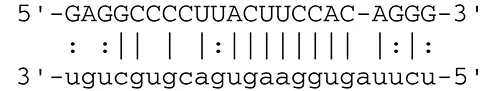 | Latent membrane protein that mimics CD40 and induces NF-κB activation | Lo et al., 2007 | |
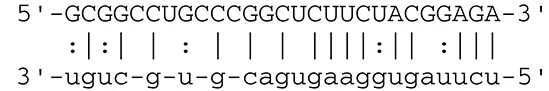 | |||||
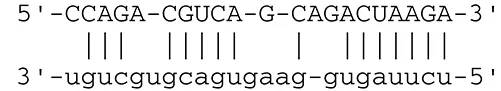 | |||||
| miR-BART22 | LMP2A | 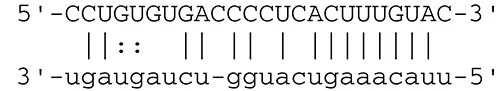 | Latent membrane protein that mimics B cell receptor | Lung et al., 2009 | |
| miR-BART2-5p | BALF5 |  | Viral DNA polymerase in lytic infection | Barth et al., 2008 | |
| miR-BART2-5p | MICB | 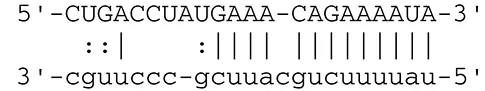 | Cellular stress-induced ligand for (NKG2D type II receptor NK cell, CD8 αβT cell) | Nachmani et al., 2009 | |
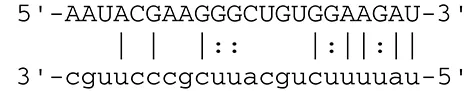 | |||||
| miR-BART3 | IPO7 | 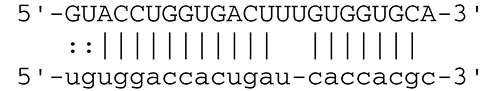 | Nuclear import receptor relating to innate immunity | Dolken et al., 2010 | |
| miR-BART15 | NLRP3 |  | Component of inflammasome | Haneklaus et al., 2012 | |
| miR-BART20-5p | BZLF1 | 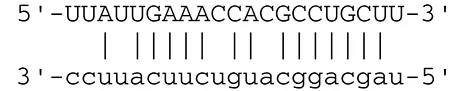 | Immediate-early genes that induce lytic replication | Jung et al., 2014 | |
| BRLF1 |  | ||||
| miR-BART6-5p | Dicer1 | 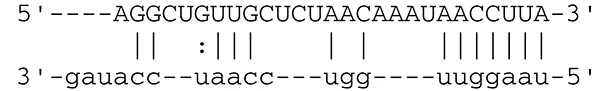 | RNase III family enzyme generating miRNA from pre-miRNA | Iizasa et al., 2010 | |
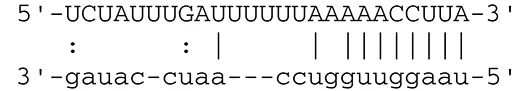 | |||||
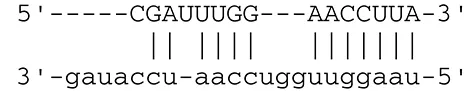 | |||||
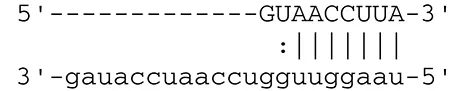 | |||||
| miR-BHRF1-3 | CXCL11 | Not defined in the reference | Chemoattractant factor for activated T cells (chemokine) | Xia et al., 2008 | |
| (I-TAC) | |||||
| miR-BART1 | PHGDH | Not defined in the reference | Central metabolite involved in synthesis of L-serine, cystine, and glycine | Ye et al., 2013 | |
| EHD1 | Not defined in the reference | Regulator of endosomal transport of plasma membrane proteins | |||
| PTEN |  | Tumor suppressor gene antagonizing the PI3K/AKT pathway | Cai et al., 2015 | ||
 | |||||
| miR-BART1-5p,3,4,5,6,7,8,10,18-3p | TGF-β | Not defined in the reference | Metabolic reprogramming | Wan et al., 2015 | |
| Pathway | Not defined in the reference | Regulation of Warburg metabolism | |||
| Wnt pathway | |||||
| miR-BHRF1 | SUMO pathyway | Not defined in the reference | Post-translational modification involved in various cellular processes | Callegari et al., 2014 | |
| KSHV | miR-K12-7 | MICB |  | Cellular stress-induced ligand for NKG2D type II receptor (NK cell, CD8 αβT cell) | Nachmani et al., 2009 |
| KSHV miRNAs | EGLN2 | Not defined in the reference | Enzyme involved in the hypoxia-inducible factor signaling pathways | Yogev et al., 2014 | |
| HSPA9 | Not defined in the reference | Key protein in mitochondrial import machinery | |||
| HCMV | miR-UL112 | MICB | Not defined in the reference | Cellular stress-induced ligand for NKG2D type II receptor (NK cell, CD8 αβT cell, γδT cell) | Nachmani et al., 2010 |
| miR-US4-1 | ERAP1 |  | Aminopeptidase 1 processing protein to form HLA class I binding peptide | Kim et al., 2011 | |
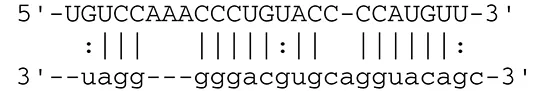 | |||||
| miR-UL148D | CCL5 (RANTES) | 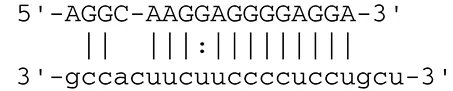 | Chemoattractant factor for memory T cells and eosinophils (chemokine) | Kim et al., 2012 | |
| miR-UL112-3p | TLR2 | 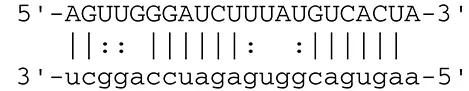 | Receptor for peptidoglycan (lipopolysaccharide) activating inflammation | Landais et al., 2015 | |
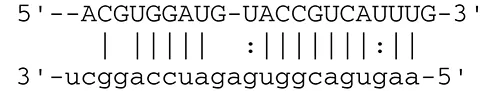 | |||||
| miR-UL112-3p | IKKA, IKKB | Not defined in the reference | Inducer of NF-κB signal, upregulation of Glut3 by mediating aerobic glycolysis | Hancock et al., 2017 | |
| miR-US5-1 | IKKA, IKKB | Not defined in the reference | Inducer of NF-κB signal, upregulation of Glut3 by mediating aerobic glycolysis | ||
| HSV-1 | miR-H8 | PIGT |  | Component of GPI transamidase complex | Enk et al., 2016 |
| miR-H6 | ICP4 |  | Transactivator of lytic infection-associated genes | Umbach et al., 2008 | |
| miR-H2 | ICP0 |  | E3 ubiquitin ligase that activates viral gene transcription | Umbach et al., 2008 | |
| miR-H3 | ICP34.5 |  | Neurovirulence factor that inactivates autophagy | Umbach et al., 2008 | |
| miR-H4 | ICP34.5 |  | Neurovirulence factor that inactivates autophagy | Umbach et al., 2008 | |
| miR-H4-5p | p16INK4A |  | Tumor suppressor gene and repressor of the PI3K/AKT pathway | Zhao et al., 2015 | |
The role of herpes virus-encoded miRNAs in immune evasion and cancer metabolism.
White or gray backgrounds relate to immune evasion, and black backgrounds relate to cancer metabolism. KSHV, Kaposi's sarcoma-associated herpesvirus; HCMV, human cytomegalovirus; HSV, herpes simplex virus.
The first report of viral miRNA was described in the herpesvirus family, especially in Epstein-Barr virus (EBV) strain B95-8 having a 12-kb deletion in BamHI-A rightward transcripts (BART) region (Pfeffer et al., 2004). Subsequent studies revealed that EBV encodes 44 different mature BART miRNAs and 4 mature BamHI-H rightward open reading frame 1 (BHRF1) miRNAs (Cai et al., 2006; Grundhoff et al., 2006; Zhu et al., 2009) (Figure 1). Rhesus lymphocryptovirus encodes miRNAs, which are orthologs of EBV miRNAs and evolutionarily conserved (Cai et al., 2006). Kaposi's sarcoma-associated herpesvirus (KSHV), the etiologic agent of Kaposi's sarcoma, encodes 13 precursor (pre)-miRNAs. These KSHV miRNAs are located in the KSHV latency-associated region (Pfeffer et al., 2005; Grundhoff et al., 2006). By sharing target genes, these KSHV miRNAs function as analogs of cellular oncomirs (Skalsky et al., 2007; Gottwein et al., 2011; Manzano et al., 2013).
Figure 1
This mini-review summarizes the role of viral miRNAs in cellular metabolism and the immune system of hosts to understand the pathologies of viral infection. Although viral miRNAs have also been detected in JC virus and human immunodeficiency virus, we mainly discuss miRNAs from Herpesviridae.
Pathological roles of EBV miRNAs
EBV-encoded miRNAs
EBV is associated with many tumors, including lymphoma and epithelial carcinomas (Young and Murray, 2003; Luo et al., 2005). Detection of EBV-encoded small non-coding RNAs (EBERs) by in situ hybridization is used as a diagnostic hallmark of EBV infection in tumor cells. EBERs can be detected in a variety of tumors, such as nasal NK/T-cell lymphoma, post-transplant lymphoma, Burkitt's lymphoma, Hodgkin's disease, diffuse large B-cell lymphoma, nasopharyngeal carcinoma (NPC), and gastric carcinoma (Delecluse et al., 2007). Following primary infection, EBV establishes latent infection. Three latency types (I, II, III) are defined depending on the pattern of expression in viral genes (Middeldorp et al., 2003). BART miRNAs are expressed in all latency types, whereas BHRF1 miRNAs are expressed only in type III latency. BHRF1 transcripts encode four mature BHRF1 miRNAs. BART transcripts have two clusters, cluster 1 and cluster 2, which generate 44 mature BART miRNAs (Figure 1) (Pfeffer et al., 2004; Cai et al., 2006; Zhu et al., 2009). EBV B95-8 strain has a 12-kb deletion and lacks most of the BART miRNAs (Baer et al., 1984).
BART miRNAs are expressed more strongly in EBV-associated epithelial cells than in B lymphocytes (Chen et al., 2010). BART transcripts have two TATA-less promoter regions, designated P1 and P2 (Sadler and Raab-Traub, 1995). Although P1 supports substantial activity in epithelial cells and B lymphocytes, P2 exhibits strong activity only in epithelial cells. Several transcription factors, important for cellular metabolism and immunity, regulate BART promoter activity (Chen et al., 2005). P1 activity is negatively regulated by interferon regulatory factor 5 (IRF5) and IRF7, and P2 activity is positively regulated by c-Myc and CCAAT-enhancer-binding protein (C/EBP) family members. Relative expression between positive and negative transcription factors in EBV-infected cells probably controls the expression level of BART miRNAs. BHRF1 miRNAs are generated as part of the Cp- and/or Wp-initiated EBNA transcripts in cells showing latency III infection (Amoroso et al., 2011). The expression of miR-BHRF1-1 absolutely depends on Cp/Wp activity, because it locates at the 5′ UTR of BHRF1 mRNA and overlaps with the EBV replication-activated BHRF1 promoter (Amoroso et al., 2011). However, because miR-BHRF1-2 and miR-BHRF1-3 locate at the 3′ UTR, they are strongly expressed by an alternative promoter for lytic BHRF1 transcript during lytic replication (Kelly et al., 2009; Xing and Kieff, 2011). The expression of BART miRNAs inversely correlates with the methylation status of the promoter in EBV-infected B lymphocytes. Promoter methylation may be important in regulating the expression of BART and BHRF1 miRNAs (Kim do et al., 2011).
EBV miRNAs are transferred to adjacent cells via exosomes (Pegtel et al., 2010). The secreted viral miRNAs modify the expression of target genes in recipient cells (Haneklaus et al., 2012). Some BART miRNAs show distinctive expression between cells and exosomes. MiR-BART7 is expressed more abundantly in exosomes than in EBV-positive NPC cells (Meckes et al., 2010). However, miR-BART8-5p is expressed less abundantly in exosomes than in lymphoblastoid cell lines (Hoshina et al., 2016). These findings indicate that some EBV miRNAs are selectively packaged and transported into recipient cells via exosomes.
Immune evasion by EBV miRNAs
EBV can establish either latent infection or lytic replication (Kenney and Mertz, 2014). In tumor cells, EBV usually maintains latent infection rather than entering into lytic replication (Cohen, 2000). During latent infection, EBV transcribes viral miRNAs to escape from the host immune system by targeting both cellular and viral genes. Lowered expression of viral proteins enables infected cells to escape antigenic recognition by the host immune system. Expression of latent membrane protein 1 (LMP1) and LMP2A is downregulated by BART cluster 1 miRNAs (miR-BART16, 17-5p, and 1-5p) (Lo et al., 2007) and miR-BART22 (Lung et al., 2009), respectively. LMP1 and LMP2A are oncogenic viral proteins that promote EBV-positive malignancies by engaging a number of signal pathways, such as the NFκB, JNK/p38-SAPK, PI3K/Akt, ERK-MAPK, and JAK/STAT pathways, followed by subsequent induction of morphological and phenotypic alterations (Young and Rickinson, 2004). LMP1 and LMP2A alter the host immune system by cooperating with environmental and host genetic factors (Dawson et al., 2012).
Viral miRNAs help maintain latent infection by expressing limited numbers of viral genes that allow EBV to evade host immune surveillance. MiR-BART20-5p suppresses lytic replication by targeting EBV immediate-early genes BZLF1 and BRLF1 (Jung et al., 2014), key regulators of the expression of EBV proteins and the production of progeny virus (Pattle and Farrell, 2006). Downregulation of Dicer by miR-BART6-5p reduces the expression of BZLF1, BRLF1, and both EBNA2 and LMP1 latent proteins in C666-1, an EBV-positive NPC cell line (Iizasa et al., 2010). Likewise, miR-BART2-5p blocks lytic replication by inhibiting expression of viral DNA polymerase BALF5 (Barth et al., 2008).
EBV miRNAs can also block immune response by reducing cytokines, chemokines, and T-cell stimulatory molecules. MiR-BHRF1-3 modulates host interferon (IFN) response by targeting CXCL11, an IFN-inducible T-cell-attracting chemokine (Xia et al., 2008). Suppression of MHC class I-related chain B (MICB) by miR-BART2-5p protects EBV-infected cells from attack by NK cells and T cells. MICB is also a target gene for other herpesvirus miRNAs, including KSHV-encoded miR-K12-1 and human cytomegalovirus (HCMV)-encoded miR-UL112 (Nachmani et al., 2009). Importin 7 (IPO7), a receptor for importing transcription factors into the nucleus, has important function for innate immunity, because loss of IPO7 in macrophages inhibits interleukin (IL)-6 secretion (Yang et al., 2009). IPO7 is a putative target for miR-BART1-3p and miR-BART3 (Dolken et al., 2010). MiR-BART15 also reduces IL-1β production from the inflammasome by targeting NLRP3 (NLR family, pyrin domain containing 3; also known as cryopyrin) at the same 3′ UTR region that host miR-223 recognizes (Haneklaus et al., 2012). These findings suggest that EBV miRNAs inhibit immune response by multiple mechanisms.
EBV miRNAs in cancer metabolism
Compared with normal cells, cancer cells increase metabolic autonomy, which uses nutrients and promotes metabolic processes of the host cells to support proliferation. Recent studies showed that several EBV miRNAs regulate cellular metabolic processes. MiR-BART1 is one of the metabolic regulators strongly expressed in NPC. The expression levels of phosphoglycerate dehydrogenase (PHGDH) and EH domain-containing protein 1 (EHD1) were significantly upregulated by miR-BART1 expression in an NPC cell line, CNE1 (Ye et al., 2013). PHGDH is important for the synthesis of serine and glycine, a central metabolite for a variety of biosynthetic pathways, by removing 3-phosphoglycerate during glycolysis (DeBerardinis, 2011). EHD1 regulates endosomal transport of plasma membrane proteins such as transferrin receptor and ß1 integrin (Jovic et al., 2007).
Wan et al. reported that 9 EBV miRNAs (miR-BART1-5p, 3, 4, 5, 6, 7, 8, 10, and 18-3p) are highly expressed in NPC tissues. Genome pathway analysis indicated that upregulated EBV miRNAs mainly target transforming growth factor β (TGF-β) and Wnt signaling pathways (Wan et al., 2015), which are involved in many biological processes during oncogenesis, including reprogramming of tumor cell bioenergetics (Sherwood, 2015) and the bioenergetic shift toward catabolism (Guido et al., 2012). Previous studies showed that both pathways are dysregulated in NPC (Zeng et al., 2007; Chen et al., 2009). Consistent with this result, the TGF-β signaling pathway is suppressed by upregulation of miR-BHRF1, because miR-BHRF1 targets the small ubiquitin-like modifier-regulated component SMAD3 and the transcription co-regulators JUN and FOS (Callegari et al., 2014). Recently, target genes for EBV miRNAs were identified by high-throughput sequencing of RNA isolated by the methods of crosslinking immunoprecipitation (HITS-CLIP) and photoactivatable ribonucleoside-enhanced crosslinking and immunoprecipitation (PAR-CLIP). These EBV miRNA targets include many components of the Wnt signaling pathway (Riley et al., 2012; Skalsky et al., 2012).
The PI3K/AKT/mTOR pathway is a critical regulator in cell survival, growth, protein synthesis, and glucose metabolism (Yap et al., 2008). Moreover, the AKT pathway promotes the expression of genes involved in glycolysis and lipid genesis (Wullschleger et al., 2006). In NPC cells, miR-BART1 significantly reduces phosphatase and tensin homolog (PTEN) expression while increasing the phosphorylation level of pAKT, pFAK, p130Cas, pShc, and pERK1/2 (Cai et al., 2015). PTEN inhibits the PI3K/AKT pathway by dephosphorylating PIP3 and increasing PIP2, resulting in a reduction of membrane recruitment of AKTs (Rafalski and Brunet, 2011). Thus, miR-BART1 activates migration, invasion, and metastasis of NPC cells via suppression of PTEN (Cai et al., 2015).
MiR-BART7 is expressed strongly in NPC cells and promotes cell proliferation, migration, and invasion. Pathway analysis indicates that the expression level of various genes is altered by the expression of miR-BART7. These target genes belong to the signaling pathway of calcium and the immune system, ionotropic glutamate receptor, ATP-binding cassette transporters, nuclear receptors in lipid metabolism and toxicity, the TGF-ß-signaling pathway, and metabolism of lipids and lipoproteins (Chan et al., 2012). Therefore, the aberrant expression of EBV miRNAs may contribute to metabolic abnormality and oncogenesis in EBV-infected cells by unbalancing various signaling pathways.
miRNAs in other herpesviruses
Herpesvirus-encoded miRNAs
Herpesviruses other than EBV and KSHV also encode miRNAs. Herpes simplex virus (HSV) has two serotypes, HSV-1 and HSV-2, which infect oral or genital mucosa. The latency-associated transcript functioning as a primary miRNA precursor is exclusively expressed during latent infection (Wagner et al., 1988; Umbach et al., 2008). Since the first report in 2006, 27 mature miRNA sequences have been identified in the HSV-1 genome (Cui et al., 2006; Jurak et al., 2010). Similarly, HSV-2 encodes 24 mature miRNAs (Umbach et al., 2010). Several miRNAs are conserved between HSV-1 and HSV-2, especially in their seed regions. These viral miRNAs have analogous functions in immune evasion and virus propagation (Jurak et al., 2010; Umbach et al., 2010).
Unlike other herpesviruses, HCMV miRNAs are not clustered in latent transcripts, but are distributed throughout the viral genome (Buck et al., 2007). Currently, 26 mature HCMV miRNA sequences are uploaded on miRBase (http://www.mirbase.org). HCMV miRNAs target multiple genes related to immune response, cell cycle control, and vesicle trafficking (Hook et al., 2014).
Varicella-zoster virus (VZV) is a pathogenic human virus that causes chicken pox and shingles. Unlike other herpesviruses examined, although many small RNA sequencing studies have been performed, VZV miRNAs have not been identified yet (Umbach et al., 2009).
Human herpesvirus 6 (HHV-6), with its two variants HHV-6A and HHV-6B, is a ubiquitous pathogen in general human populations. Both are very closely related, with nearly 90% homology at the genomic level. Deep sequencing of small RNA species identified a small non-coding RNA with the characteristics of a viral miRNA from cells harboring HHV-6A. Growth analyses of mutant viruses revealed that miR-U86 directly impacts lytic replication by targeting HHV-6A immediate-early gene U86 (Nukui et al., 2015). HHV-6B encodes four pre-miRNAs expressed from direct repeat regions located at either side of the genome. HHV-6B miR-Ro6-2 is a seed ortholog of host miR-582-5p, which targets SMAD3 to downregulate TGF-β. HHV-6B miRNAs also have the potential to regulate viral replication (Tuddenham et al., 2012).
HHV-7 is a ubiquitous T-lymphotropic virus infecting most humans. As with VZV, HHV-7-encoded miRNAs have not yet been identified (Louten et al., 2015).
Herpes viral miRNAs in immune evasion
EBV miR-BART2-5p and KSHV miR-K12-7 target the 3′ UTR of MICB at different locations (Nachmani et al., 2009). HCMV miR-UL112 and cellular miR-376a synergistically downregulate MICB expression and subsequently help the virus evade innate immune recognition (Nachmani et al., 2010). Recognition of HCMV-infected cells by cytotoxic T lymphocytes is impaired due to the reduced expression of aminopeptidase ERAP1 by HCMV miR-US4-1 (Kim et al., 2011). In human fibroblast cells, HCMV clinical strain-specific miR-UL148D was shown to block the human chemokine RANTES, which attracts immune cells during inflammation and the immune response (Kim et al., 2012).
HSV-1 miR-H8 targets the glycosylphosphatidylinositol gene, which results in reduced expression of several immune-modulating proteins, viral expansion, and viral evasion from natural killer cell elimination (Enk et al., 2016). HSV miRNAs also target viral genes to maintain latency and suppress immune function. HSV-1 miR-H6 and miR-H2 reduce infected cell polypeptide 4 (ICP4) and ICP0, respectively (Umbach et al., 2008; Duan et al., 2012). Both miR-H3 and miR-H4 target ICP34.5 mRNA (Umbach et al., 2008). HSV-2 miRNAs also contribute to latency and immune evasion similarly to HSV-1 miRNAs because of the close homology of these two viruses.
In KSHV-infected primary effusion lymphoma cells, KSHV miR-K1 inhibits viral lytic replication by targeting the 3′ UTR of IκBα protein, an inhibitor of the NFκB complexes. Enhanced NFκB activity evades host immune system and promotes cell survival (Lei et al., 2010). The KSHV miRNA cluster also represses a network of targets associated with STAT3 and suppresses STAT3 activation upon IL-6 treatment. KSHV miR-K6-5 targets the 3′ UTR of PKCδ, a Ser/Thr kinase that phosphorylates and activates STAT3. KSHV miR-K921 also targets a second Ser/Tr kinase, IRAK1. Repression of BIRC5, a transcriptional target of STAT3, by KSHV miR-K12-5 promotes KSHV infection. These multiple KSHV miRNAs that repress STAT3 can weaken the innate immune responses to type-I interferons and inhibit the induction of antiviral genes, such as IRF1, IFITM1, and ISG15 (Ramalingam and Ziegelbauer, 2017).
Herpes viral miRNAs in cancer metabolism
Several core cellular metabolic pathways are significantly altered by herpesvirus infection and the expression of viral miRNAs (Sanchez and Lagunoff, 2015). HSV-1 miR-H4-5p directly targets cyclin-dependent kinase inhibitor 2A (p16) mRNA in neuroblastoma cell lines. Suppression of miR-H4-5p inhibits cell proliferation, invasion, and progression of the cell cycle via the p16-mediated PI3K-AKT signaling pathway (Zhao et al., 2015). Likewise, HCMV miR-UL112-3p modulates the TLR/IRAK1/NFκB signaling pathway by targeting Toll-like receptor 2 mRNA (Landais et al., 2015). NFκB signaling is known to upregulate the expression of Glut3 in p53-deficient cells (Kawauchi et al., 2008), suggesting that inhibition of NFκB signaling by miR-UL112-3p and miR-US5-1 might suppress aerobic glycolysis (Hancock et al., 2017).
KSHV has been shown to alter host cell energy metabolism by concurrent regulation of two independent pathways. First, KSHV miRNAs stabilize and activate transcription factor HIF1α, a master regulator of cell metabolism, by targeting hypoxia-inducible factor prolyl hydroxylase, EGLN2. Second, downregulation of the mitochondrial heat shock protein A9 (HSPA9) by KSHV miRNAs reduces mitochondrial copy numbers and enhances anaerobic glycolysis (Warburg effect). Downregulation of EGLN2 and HSPA9 allows cell proliferation in a low oxygen condition (Yogev et al., 2014). KSHV miR-K12-11 and miR-K12-3 prevent lytic reactivation by reducing the expression of cellular transcription factors MYB, C/EBPα, and Ets-1, which are reported as activators of the RTA promoter (Plaisance-Bonstaff et al., 2014).
Murine gammaherpesvirus 68 (MHV-68), a natural pathogen of wild rodents, encodes for 14 pre-miRNAs. All MHV-68 miRNAs are located downstream of viral tRNA-like elements and transcribed by RNA polymerase III. Recent research showed that an MHV-68 mutant lacking the expression of all miRNAs results in a higher viral genomic load in the spleen. This report shows that MHV-68 miRNAs contribute to the maintenance of latency in vivo (Steer et al., 2016).
The generation and analysis of mutant viruses revealed that MHV-68 miRNAs are dispensable for short-term virus replication but are important for the establishment of lifelong infection in memory B cells. Furthermore, a lack of miRNA expression results in the complete attenuation of lethal disease in a virus-induced pneumonia model, demonstrating a key role for the viral miRNAs in pathogenesis (Feldman et al., 2014).
Similar to EBERs in EBV, MHV-68 encodes non-coding RNAs called TMERs (tRNA-miRNA-encoded RNAs), which are highly expressed in latently infected cells. TMERs harbor a predicted tRNA-like element and two downstream pre-miRNA hairpins and are processed by tRNase Z instead of Drosha, similar to cellular non-coding tRNAs, to generate mature miRNAs. Analysis of individual TMER mutant viruses has shown TMER4 to be a key mediator of virus dissemination. Interestingly, TMER4 miRNA seed sequence mutants do not compromise TMER4 function. These results demonstrate a crucial miRNA-independent function of TMER4 in hematogenous dissemination and the establishment of peripheral latency (Feldman et al., 2016).
Summary and conclusions
Viral miRNAs play important roles in cancer development and progression by modulating immune response and metabolic circuits. Although further study is necessary to understand the pathogenic significance of viral miRNAs, viral miRNAs can be applied for the diagnosis of cancer, identification of drug targets, and therapeutic use.
Statements
Author contributions
HK wrote the manuscript. SF and YK assisted in creating the table. HI and HY edited the paper and contributed financial assistance.
Funding
This study was supported by KAKENHI (Grant-in-Aid for Scientific Research) from the Ministry of Education, Culture, Sports, Science, and Technology (HI: 26460465, and HY: 16H05843), and a Health Labor Sciences Research Grant from the Ministry of Health Labor and Welfare, Japan (17fk0310105h0001).
Acknowledgments
The authors thank Ms. Sayuri Hamada for her support in the preparation of the figure.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1
AmorosoR.FitzsimmonsL.ThomasW. A.KellyG. L.RoweM.BellA. I. (2011). Quantitative studies of Epstein-Barr virus-encoded microRNAs provide novel insights into their regulation. J. Virol.85, 996–1010. 10.1128/jvi.01528-10
2
BaerR.BankierA. T.BigginM. D.DeiningerP. L.FarrellP. J.GibsonT. J.et al. (1984). DNA sequence and expression of the B95-8 Epstein-Barr virus genome. Nature310, 207–211. 10.1038/310207a0
3
BarthS.PfuhlT.MamianiA.EhsesC.RoemerK.KremmerE.et al. (2008). Epstein-Barr virus-encoded microRNA miR-BART2 down-regulates the viral DNA polymerase BALF5. Nucleic Acids Res.36, 666–675. 10.1093/nar/gkm1080
4
BuckA. H.Santoyo-LopezJ.RobertsonK. A.KumarD. S.ReczkoM.GhazalP. (2007). Discrete clusters of virus-encoded micrornas are associated with complementary strands of the genome and the 7.2-kilobase stable intron in murine cytomegalovirus. J. Virol.81, 13761–13770. 10.1128/jvi.01290-07
5
CaiL.YeY.JiangQ.ChenY.LyuX.LiJ.et al. (2015). Epstein-Barr virus-encoded microRNA BART1 induces tumour metastasis by regulating PTEN-dependent pathways in nasopharyngeal carcinoma. Nat. Commun.6:7353. 10.1038/ncomms8353
6
CaiX.SchaferA.LuS.BilelloJ.DesrosiersR.EdwardsR.et al. (2006). Epstein-Barr virus microRNAs are evolutionarily conserved and differentially expressed. PLoS Pathog.2:e23. 10.1371/journal.ppat.0020023
7
CallegariS.GastaldelloS.FaridaniO. R.MasucciM. G. (2014). Epstein-Barr virus encoded microRNAs target SUMO-regulated cellular functions. FEBS J.281, 4935–4950. 10.1111/febs.13040
8
ChanJ. Y.GaoW.HoW. K.WeiW. I.WongT. S. (2012). Overexpression of Epstein-Barr virus-encoded microRNA-BART7 in undifferentiated nasopharyngeal carcinoma. Anticancer Res.32, 3201–3210.
9
ChenH. C.ChenG. H.ChenY. H.LiaoW. L.LiuC. Y.ChangK. P.et al. (2009). MicroRNA deregulation and pathway alterations in nasopharyngeal carcinoma. Br. J. Cancer100, 1002–1011. 10.1038/sj.bjc.6604948
10
ChenH.HuangJ.WuF. Y.LiaoG.Hutt-FletcherL.HaywardS. D. (2005). Regulation of expression of the Epstein-Barr virus BamHI-A rightward transcripts. J. Virol.79, 1724–1733. 10.1128/JVI.79.3.1724-1733.2005
11
ChenS.-J.ChenG.-H.ChenY.-H.LiuC.-Y.ChangK.-P.ChangY.-S.et al. (2010). Characterization of Epstein-Barr virus miRNAome in nasopharyngeal carcinoma by deep sequencing. PLoS ONE5:e12745. 10.1371/journal.pone.0012745
12
CohenJ. I. (2000). Epstein-Barr virus infection. N. Engl. J. Med.343, 481–492. 10.1056/NEJM200008173430707
13
CuiC.GriffithsA.LiG.SilvaL. M.KramerM. F.GaasterlandT.et al. (2006). Prediction and identification of herpes simplex virus 1-encoded microRNAs. J. Virol.80, 5499–5508. 10.1128/jvi.00200-06
14
DawsonC. W.PortR. J.YoungL. S. (2012). The role of the EBV-encoded latent membrane proteins LMP1 and LMP2 in the pathogenesis of nasopharyngeal carcinoma (NPC). Semin. Cancer Biol.22, 144–153. 10.1016/j.semcancer.2012.01.004
15
DeBerardinisR. J. (2011). Serine metabolism: some tumors take the road less traveled. Cell Metab.14, 285–286. 10.1016/j.cmet.2011.08.004
16
DelecluseH. J.FeederleR.O'SullivanB.TaniereP. (2007). Epstein Barr virus-associated tumours: an update for the attention of the working pathologist. J. Clin. Pathol.60, 1358–1364. 10.1136/jcp.2006.044586
17
DolkenL.MaltererG.ErhardF.KotheS.FriedelC. C.SuffertG.et al. (2010). Systematic analysis of viral and cellular microRNA targets in cells latently infected with human gamma-herpesviruses by RISC immunoprecipitation assay. Cell Host Microbe7, 324–334. 10.1016/j.chom.2010.03.008
18
DuanF.LiaoJ.HuangQ.NieY.WuK. (2012). HSV-1 miR-H6 inhibits HSV-1 replication and IL-6 expression in human corneal epithelial cells in vitro. Clin. Dev. Immunol.2012:192791. 10.1155/2012/192791
19
EnkJ.LeviA.WeisblumY.YaminR.Charpak-AmikamY.WolfD. G.et al. (2016). HSV1 MicroRNA modulation of GPI anchoring and downstream immune evasion. Cell Rep.17, 949–956. 10.1016/j.celrep.2016.09.077
20
FeldmanE. R.KaraM.ColemanC. B.GrauK. R.OkoL. M.KruegerB. J.et al. (2014). Virus-encoded microRNAs facilitate gammaherpesvirus latency and pathogenesis in vivo. mBio5, e00981–e00914. 10.1128/mBio.00981-14
21
FeldmanE. R.KaraM.OkoL. M.GrauK. R.KruegerB. J.ZhangJ.et al. (2016). A gammaherpesvirus noncoding RNA is essential for hematogenous dissemination and establishment of peripheral latency. mSphere1, e00105–e0011510.1128/mSphere.00105-15
22
GottweinE.CorcoranD. L.MukherjeeN.SkalskyR. L.HafnerM.NusbaumJ. D.et al. (2011). Viral microRNA targetome of KSHV-infected primary effusion lymphoma cell lines. Cell Host Microbe10, 515–526. 10.1016/j.chom.2011.09.012
23
GrundhoffA.SullivanC. S.GanemD. (2006). A combined computational and microarray-based approach identifies novel microRNAs encoded by human gamma-herpesviruses. RNA12, 733–750. 10.1261/rna.2326106
24
GuidoC.Whitaker-MenezesD.CapparelliC.BallietR.LinZ.PestellR. G.et al. (2012). Metabolic reprogramming of cancer-associated fibroblasts by TGF-beta drives tumor growth: connecting TGF-beta signaling with “Warburg-like” cancer metabolism and L-lactate production. Cell Cycle11, 3019–3035. 10.4161/cc.21384
25
HancockM. H.HookL. M.MitchellJ.NelsonJ. A. (2017). Human cytomegalovirus MicroRNAs miR-US5-1 and miR-UL112-3p block proinflammatory cytokine production in response to nf-κb-activating factors through direct downregulation of IKKα and IKKβ. mBio8, e00109–00117. 10.1128/mBio.00109-17
26
HaneklausM.GerlicM.Kurowska-StolarskaM.RaineyA. A.PichD.McInnesI. B.et al. (2012). Cutting edge: miR-223 and EBV miR-BART15 regulate the NLRP3 inflammasome and IL-1beta production. J. Immunol.189, 3795–3799. 10.4049/jimmunol.1200312
27
HookL.HancockM.LandaisI.GrabskiR.BrittW.NelsonJ. A. (2014). Cytomegalovirus microRNAs. Curr. Opin. Virol.7, 40–46. 10.1016/j.coviro.2014.03.015
28
HoshinaS.SekizukaT.KataokaM.HasegawaH.HamadaH.KurodaM.et al. (2016). Profile of exosomal and intracellular microRNA in gamma-herpesvirus-infected lymphoma cell lines. PLoS ONE11:e0162574. 10.1371/journal.pone.0162574
29
IizasaH.WulffB.-E.AllaN.MaragkakisM.MegrawM.HatzigeorgiouA.et al. (2010). Editing of Epstein-Barr virus-encoded BART6 microRNAs controls their dicer targeting and consequently affects viral latency. J. Biol. Chem.285, 33358–33370. 10.1074/jbc.M110.138362
30
JovicM.NaslavskyN.RapaportD.HorowitzM.CaplanS. (2007). EHD1 regulates beta1 integrin endosomal transport: effects on focal adhesions, cell spreading and migration. J. Cell Sci.120, 802–814. 10.1242/jcs.03383
31
JungY. J.ChoiH.KimH.LeeS. K. (2014). miR-BART20-5p stabilizes Epstein-Barr virus latency by directly targeting BZLF1 and BRLF1. J. Virol.88, 9027–9037. 10.1128/jvi.00721-14
32
JurakI.KramerM. F.MellorJ. C.van LintA. L.RothF. P.KnipeD. M.et al. (2010). Numerous conserved and divergent microRNAs expressed by herpes simplex viruses 1 and 2. J. Virol.84, 4659–4672. 10.1128/jvi.02725-09
33
KawauchiK.ArakiK.TobiumeK.TanakaN. (2008). p53 regulates glucose metabolism through an IKK-NF-κB pathway and inhibits cell transformation. Nat. Cell. Biol.10, 611–618. 10.1038/ncb1724
34
KellyG. L.LongH. M.StylianouJ.ThomasW. A.LeeseA.BellA. I.et al. (2009). An Epstein-Barr virus anti-apoptotic protein constitutively expressed in transformed cells and implicated in burkitt lymphomagenesis: the Wp/BHRF1 link. PLoS Pathog.5:e1000341. 10.1371/journal.ppat.1000341
35
KenneyS. C.MertzJ. E. (2014). Regulation of the latent-lytic switch in Epstein-Barr virus. Semin. Cancer Biol.26, 60–68. 10.1016/j.semcancer.2014.01.002
36
Kim doN.SongY. J.LeeS. K. (2011). The role of promoter methylation in Epstein-Barr virus (EBV) microRNA expression in EBV-infected B cell lines. Exp. Mol. Med.43, 401–410. 10.3858/emm.2011.43.7.044
37
KimS.LeeS.ShinJ.KimY.EvnouchidouI.KimD.et al. (2011). Human cytomegalovirus microRNA miR-US4-1 inhibits CD8+ T cell responses by targeting the aminopeptidase ERAP1. Nat. Immunol.12, 984–991. 10.1038/ni.2097
38
KimY.LeeS.KimS.KimD.AhnJ. H.AhnK. (2012). Human cytomegalovirus clinical strain-specific microRNA miR-UL148D targets the human chemokine RANTES during infection. PLoS Pathog.8:e1002577. 10.1371/journal.ppat.1002577
39
LandaisI.PeltonC.StreblowD.DeFilippisV.McWeeneyS.NelsonJ. A. (2015). Human cytomegalovirus miR-UL112-3p targets TLR2 and modulates the TLR2/IRAK1/NFκB signaling pathway. PLoS Pathog.11:e1004881. 10.1371/journal.ppat.1004881
40
LeiX.BaiZ.YeF.XieJ.KimC. G.HuangY.et al. (2010). Regulation of NF-kappaB inhibitor IkappaBalpha and viral replication by a KSHV microRNA. Nat. Cell Biol.12, 193–199. 10.1038/ncb2019
41
LoA. K.ToK. F.LoK. W.LungR. W.HuiJ. W.LiaoG.et al. (2007). Modulation of LMP1 protein expression by EBV-encoded microRNAs. Proc. Natl. Acad. Sci. U.S.A.104, 16164–16169. 10.1073/pnas.0702896104
42
LoutenJ.BeachM.PalerminoK.WeeksM.HolensteinG. (2015). MicroRNAs expressed during viral infection: biomarker potential and therapeutic considerations. Biomark Insights10, 25–52. 10.4137/BMI.S29512
43
LungR. W.TongJ. H.SungY. M.LeungP. S.NgD. C.ChauS. L.et al. (2009). Modulation of LMP2A expression by a newly identified Epstein-Barr virus-encoded microRNA miR-BART22. Neoplasia11, 1174–1184. 10.1593/neo.09888
44
LuoB.WangY.WangX.-F.LiangH.YanL.-P.HuangB.-H.et al. (2005). Expression of Epstein-Barr virus genes in EBV-associated gastric carcinomas. World J. Gastroenterol.11, 629–633. 10.3748/wjg.v11.i5.629
45
ManzanoM.ShamulailatpamP.RajaA. N.GottweinE. (2013). Kaposi's sarcoma-associated herpesvirus encodes a mimic of cellular miR-23. J. Virol.87, 11821–11830. 10.1128/jvi.01692-13
46
MeckesD. G.Jr.ShairK. H.MarquitzA. R.KungC. P.EdwardsR. H.Raab-TraubN. (2010). Human tumor virus utilizes exosomes for intercellular communication. Proc. Natl. Acad. Sci. U.S.A.107, 20370–20375. 10.1073/pnas.1014194107
47
MiddeldorpJ. M.BrinkA. A.van den BruleA. J.MeijerC. J. (2003). Pathogenic roles for Epstein-Barr virus (EBV) gene products in EBV-associated proliferative disorders. Crit. Rev. Oncol. Hematol.45, 1–36. 10.1016/S1040-8428(02)00078-1
48
NachmaniD.LankryD.WolfD. G.MandelboimO. (2010). The human cytomegalovirus microRNA miR-UL112 acts synergistically with a cellular microRNA to escape immune elimination. Nat. Immunol.11, 806–813. 10.1038/ni.1916
49
NachmaniD.Stern-GinossarN.SaridR.MandelboimO. (2009). Diverse herpesvirus microRNAs target the stress-induced immune ligand MICB to escape recognition by natural killer cells. Cell Host Microbe5, 376–385. 10.1016/j.chom.2009.03.003
50
NukuiM.MoriY.MurphyE. A. (2015). A human herpesvirus 6A-encoded microRNA: role in viral lytic replication. J. Virol.89, 2615–2627. 10.1128/JVI.02007-14
51
PattleS.FarrellP. (2006). The role of Epstein-Barr virus in cancer. Expert Opin. Biol. Ther.6, 1193–1205. 10.1517/14712598.6.11.1193
52
PegtelD. M.CosmopoulosK.Thorley-LawsonD. A.van EijndhovenM. A.HopmansE. S.LindenbergJ. L.et al. (2010). Functional delivery of viral miRNAs via exosomes. Proc. Natl. Acad. Sci. U.S.A.107, 6328–6333. 10.1073/pnas.0914843107
53
PfefferS.SewerA.Lagos-QuintanaM.SheridanR.SanderC.GrasserF. A.et al. (2005). Identification of microRNAs of the herpesvirus family. Nat. Methods2, 269–276. 10.1038/nmeth746
54
PfefferS.ZavolanM.GrässerF.ChienM.RussoJ.JuJ.et al. (2004). Identification of virus-encoded microRNAs. Science304, 734–736. 10.1126/science.1096781
55
Plaisance-BonstaffK.ChoiH. S.BealsT.KruegerB. J.BossI. W.GayL. A.et al. (2014). KSHV miRNAs decrease expression of lytic genes in latently infected PEL and endothelial cells by targeting host transcription factors. Viruses6, 4005–4023. 10.3390/v6104005
56
RafalskiV. A.BrunetA. (2011). Energy metabolism in adult neural stem cell fate. Prog. Neurobiol.93, 182–203. 10.1016/j.pneurobio.2010.10.007
57
RamalingamD.ZiegelbauerJ. M. (2017). Viral microRNAs target a gene network, inhibit STAT activation, and suppress interferon responses. Sci. Rep.7:40813. 10.1038/srep40813
58
RileyK. J.RabinowitzG. S.YarioT. A.LunaJ. M.DarnellR. B.SteitzJ. A. (2012). EBV and human microRNAs co-target oncogenic and apoptotic viral and human genes during latency. EMBO J.31, 2207–2221. 10.1038/emboj.2012.63
59
SadlerR. H.Raab-TraubN. (1995). Structural analyses of the Epstein-Barr virus BamHI A transcripts. J. Virol.69, 1132–1141.
60
SanchezE. L.LagunoffM. (2015). Viral activation of cellular metabolism. Virology 479–480, 609–618. 10.1016/j.virol.2015.02.038
61
SherwoodV. (2015). WNT signaling: an emerging mediator of cancer cell metabolism?Mol. Cell Biol.35, 2–10. 10.1128/MCB.00992-14
62
SkalskyR. L.CorcoranD. L.GottweinE.FrankC. L.KangD.HafnerM.et al. (2012). The viral and cellular microRNA targetome in lymphoblastoid cell lines. PLoS Pathog.8:e1002484. 10.1371/journal.ppat.1002484
63
SkalskyR. L.SamolsM. A.PlaisanceK. B.BossI. W.RivaA.LopezM. C.et al. (2007). Kaposi's sarcoma-associated herpesvirus encodes an ortholog of miR-155. J. Virol.81, 12836–12845. 10.1128/jvi.01804-07
64
SteerB.StrehleM.SattlerC.BundD.FlachB.StoegerT.et al. (2016). The small noncoding RNAs (sncRNAs) of murine gammaherpesvirus 68 (MHV-68) are involved in regulating the latent-to-lytic switch in vivo. Sci. Rep.6:32128. 10.1038/srep32128
65
TuddenhamL.JungJ. S.Chane-Woon-MingB.DolkenL.PfefferS. (2012). Small RNA deep sequencing identifies microRNAs and other small noncoding RNAs from human herpesvirus 6B. J. Virol.86, 1638–1649. 10.1128/JVI.05911-11
66
UmbachJ. L.KramerM. F.JurakI.KarnowskiH. W.CoenD. M.CullenB. R. (2008). MicroRNAs expressed by herpes simplex virus 1 during latent infection regulate viral mRNAs. Nature454, 780–783. 10.1038/nature07103
67
UmbachJ. L.NagelM. A.CohrsR. J.GildenD. H.CullenB. R. (2009). Analysis of human alphaherpesvirus microRNA expression in latently infected human trigeminal ganglia. J. Virol.83, 10677–10683. 10.1128/JVI.01185-09
68
UmbachJ. L.WangK.TangS.KrauseP. R.MontE. K.CohenJ. I.et al. (2010). Identification of viral microRNAs expressed in human sacral ganglia latently infected with herpes simplex virus 2. J. Virol.84, 1189–1192. 10.1128/jvi.01712-09
69
WagnerE. K.Devi-RaoG.FeldmanL. T.DobsonA. T.ZhangY. F.FlanaganW. M.et al. (1988). Physical characterization of the herpes simplex virus latency-associated transcript in neurons. J. Virol.62, 1194–1202.
70
WanX. X.YiH.QuJ. Q.HeQ. Y.XiaoZ. Q. (2015). Integrated analysis of the differential cellular and EBV miRNA expression profiles in microdissected nasopharyngeal carcinoma and non-cancerous nasopharyngeal tissues. Oncol. Rep.34, 2585–2601. 10.3892/or.2015.4237
71
WinterJ.JungS.KellerS.GregoryR.DiederichsS. (2009). Many roads to maturity: microRNA biogenesis pathways and their regulation. Nat. Cell Biol.11, 228–234. 10.1038/ncb0309-228
72
WullschlegerS.LoewithR.HallM. N. (2006). TOR signaling in growth and metabolism. Cell124, 471–484. 10.1016/j.cell.2006.01.016
73
XiaT.O'HaraA.AraujoI.BarretoJ.CarvalhoE.SapucaiaJ. B.et al. (2008). EBV microRNAs in primary lymphomas and targeting of CXCL-11 by ebv-mir-BHRF1-3. Cancer Res.68, 1436–1442. 10.1158/0008-5472.can-07-5126
74
XingL.KieffE. (2011). cis-Acting effects on RNA processing and Drosha cleavage prevent Epstein-Barr virus latency III BHRF1 expression. J. Virol.85, 8929–8939. 10.1128/jvi.00336-11
75
YangI. V.WadeC. M.KangH. M.AlperS.RutledgeH.LackfordB.et al. (2009). Identification of novel genes that mediate innate immunity using inbred mice. Genetics183, 1535–1544. 10.1534/genetics.109.107540
76
YapT. A.GarrettM. D.WaltonM. I.RaynaudF.de BonoJ. S.WorkmanP. (2008). Targeting the PI3K-AKT-mTOR pathway: progress, pitfalls, and promises. Curr. Opin. Pharmacol.8, 393–412. 10.1016/j.coph.2008.08.004
77
YeY.ZhouY.ZhangL.ChenY.LyuX.CaiL.et al. (2013). EBV-miR-BART1 is involved in regulating metabolism-associated genes in nasopharyngeal carcinoma. Biochem. Biophys. Res. Commun.436, 19–24. 10.1016/j.bbrc.2013.05.008
78
YogevO.LagosD.EnverT.BoshoffC. (2014). Kaposi's sarcoma herpesvirus microRNAs induce metabolic transformation of infected cells. PLoS Pathog.10:e1004400. 10.1371/journal.ppat.1004400
79
YoungL.MurrayP. (2003). Epstein-Barr virus and oncogenesis: from latent genes to tumours. Oncogene22, 5108–5121. 10.1038/sj.onc.1206556
80
YoungL.RickinsonA. (2004). Epstein-Barr virus: 40 years on. Nat. Rev. Cancer4, 757–768. 10.1038/nrc1452
81
ZengZ. Y.ZhouY. H.ZhangW. L.XiongW.FanS. Q.LiX. L.et al. (2007). Gene expression profiling of nasopharyngeal carcinoma reveals the abnormally regulated Wnt signaling pathway. Hum. Pathol.38, 120–133. 10.1016/j.humpath.2006.06.023
82
ZhaoH.ZhangC.HouG.SongJ. (2015). MicroRNA-H4-5p encoded by HSV-1 latency-associated transcript promotes cell proliferation, invasion and cell cycle progression via p16-mediated PI3K-Akt signaling pathway in SHSY5Y cells. Int. J. Clin. Exp. Med.8, 7526–7534.
83
ZhuJ.PfuhlT.MotschN.BarthS.NichollsJ.GrasserF.et al. (2009). Identification of novel Epstein-Barr virus microRNA genes from nasopharyngeal carcinomas. J. Virol.83, 3333–3341. 10.1128/JVI.01689-08
Summary
Keywords
microRNA, herpesvirus, oncogenesis, immune evasion, cell metabolism
Citation
Kim H, Iizasa H, Kanehiro Y, Fekadu S and Yoshiyama H (2017) Herpesviral microRNAs in Cellular Metabolism and Immune Responses. Front. Microbiol. 8:1318. doi: 10.3389/fmicb.2017.01318
Received
18 April 2017
Accepted
29 June 2017
Published
18 July 2017
Volume
8 - 2017
Edited by
Marina I. Arleevskaya, Kazan State Medical Academy, Russia
Reviewed by
Keiji Ueda, Osaka University, Japan; Laurie Krug, Stony Brook University, United States
Updates
Copyright
© 2017 Kim, Iizasa, Kanehiro, Fekadu and Yoshiyama.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hironori Yoshiyama yosiyama@med.shimane-u.ac.jp
This article was submitted to Microbial Immunology, a section of the journal Frontiers in Microbiology
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.