- 1Doctor of Philosophy Program in Applied Microbiology (International Program) in Faculty of Science, Chiang Mai University, Chiang Mai, Thailand
- 2Graduate School, Chiang Mai University, Chiang Mai, Thailand
- 3Department of Biology, Faculty of Science, Chiang Mai University, Chiang Mai, Thailand
- 4Center of Excellence in Bioresources for Agriculture, Industry and Medicine, Chiang Mai University, Chiang Mai, Thailand
Recently, there is an urgent need for new drugs due to the emergence of drug resistant pathogenic microorganisms and new infectious diseases. Members of phylum Actinobacteria are promising source of bioactive compounds notably antibiotics. The search for such new compounds has shifted to extreme or underexplored environments to increase the possibility of discovery. Cave ecosystems have attracted interest of the research community because of their unique characteristics and the microbiome residing inside including actinobacteria. At the time of writing, 47 species in 30 genera of actinobacteria were reported from cave and cave related habitats. Novel and promising bioactive compounds have been isolated and characterized. This mini-review focuses on the diversity of cultivable actinobacteria in cave and cave-related environments, and their bioactive metabolites from 1999 to 2018.
Introduction
Caves are generally regarded as any natural underground chamber that is large enough for human entrance. They can be classified based on type of rock and formation method. The most common types of caves are limestone and other calcareous rocks (Northup and Lavoie, 2001). Though caves have been studied for hundreds of years, their microbiome are generally underexplored and overlooked. Caves are attracting the interests of microbiologists, in terms of microbial diversity, during the past decade (Laiz et al., 1999; Barton et al., 2004; Barton, 2006). It is believed that microbes collected from pristine sites that are unexplored or rarely visited by humans are likely to be novel taxa or strains which produce unique beneficial chemical compounds. Market demand for new drugs is on the rise due to the emergence of new diseases and drug resistant pathogens (Genilloud, 2017; Kemung et al., 2018; Takahashi and Nakashima, 2018). With a combination of unique conditions including high humidity, relatively low and stable temperature, and low nutrients, caves are expected to harbor novel microorganisms with biotechnological benefits. Members of actinobacteria are reported to be a dominant microbial population in several cave ecosystems (Groth and Saiz-Jimenez, 1999; Cheeptham et al., 2013; Tomczyk-Zak and Zielenkiewicz, 2016; Ghosh et al., 2017).
Actinobacteria are large group of high G+C Gram positive bacteria (Barka et al., 2016). They are regarded as the most prolific source of bioactive compounds in particular commercially available antibiotics. Actinobacteria produce approximately two-thirds of all know antibiotics in the market, most of these are from members of the genus Streptomyces (Barka et al., 2016). Several members of diverse actinobacterial taxa were also found to produce wide range of other biologically active compounds, for examples antibacterial, anticancer, or antifungal drugs (Barka et al., 2016; Genilloud, 2017; Castro et al., 2018; Takahashi and Nakashima, 2018). Isolation of actinobacteria from unique natural habitats is of interest to avoid re-isolation of strains that produce known bioactive metabolites and usually lead to highly diverse actinobacterial communities. The present mini-review provides evidence that actinobacteria from caves are expected to be a good source for drug discovery (Yücel and Yamac, 2010; Cheeptham et al., 2013; Kay et al., 2013; Ghosh et al., 2017; Riquelme et al., 2017).
Selective Isolation of Cave Actinobacteria
In the past decade, there are many reports on the discovery of novel actinobacteria in cave habitats. Successful isolation of actinobacteria from caves depend largely on factors of (1) media composition (Kim et al., 1998) (2) culture condition, and (3) pretreatment methods (Kim et al., 1998; Nakaew et al., 2009a,b; Duangmal et al., 2012; Niyomvong et al., 2012; Velikonja et al., 2014; Fang et al., 2017b; Adam et al., 2018). Media used for the isolation of cave actinobacteria range from routine cultivation media such as International Streptomyces Project medium 2 (yeast malt extract agar, ISP2) or tryptic soy agar (TSA) to selective media including humic acid vitamin agar (HV), starch casein agar (SC), starch casein nitrate agar (SCN), peptone-yeast extract/brain-heart infusion medium (PY-BHI), R2A medium, actinomycete isolation agar (AI), and Gauze's medium No.1. Moreover, isolation media that mimic the conditions of low concentration nutrients in caves such as tap water agar, 1/100 ISP2 and oligotrophic medium (M5) were also successfully used for the isolation of actinobacteria. (Lee et al., 2000b; Velikonja et al., 2014; Covington et al., 2018; Passari et al., 2018). High concentration of nutrients in standard cultivation media were reported to cause cell death in cave-associated bacteria due to osmotic stress (Barton, 2006; Ghosh et al., 2017).
Two important culture conditions for actinobacteria isolation are incubation temperature and incubation time. Four incubation temperatures (5°, 13°, 20°, and 28°C) were used for the isolation of soil bacteria including actinobacteria from three caves in Northern Spain (Laiz et al., 2003). The incubation temperature of 5°C was used to represent cave temperature and target psychrotrophs, 28°C as laboratory incubation temperature and 20°C as intermediate temperature between cave and laboratory conditions. The highest number of actinobacterial isolates (mostly sporoactinomycetes) was obtained at 28°C followed by 13°, 20° and 5°C, respectively. However, a higher diversity was observed from 13°C than 28°C. Therefore, these authors concluded that the isolation of actinobacteria is a temperature-dependent process. In addition, longer incubation time was successfully used to promote the recovery of slow-growing actinobacteria (Laiz et al., 2003).
Pretreatment, both chemical and physical methods are generally useful for isolation of various actinobacterial species. Physical pretreatments involve the use of air drying, moist heat, dry heat and electromagnetic wave. Moist heating (water bath at 50°C for 5–6 min) is useful for eliminating of fast growing bacteria (Niyomvong et al., 2012; Velikonja et al., 2014). Dry heating at 120°C for 1 h is effective in reducing number of unwanted bacteria and found to be an effective method for isolation of members of the genera Dactylosporangium, Streptosporangium and Microbispora, while growth of streptomycetes was limited (Jiang et al., 2016). In addition, dry heating with or without phenol treatment resulted in a reduction of bacteria and heat-labile Streptomyces, thus heat resistant rare actinobacteria were readily isolated (Kim et al., 1998; Nakaew et al., 2009a; Niyomvong et al., 2012). However, these treatments also affect the number of viable actinobacteria (Niyomvong et al., 2012). Pretreatment using microwave irradiation was effective for the isolation of rare actinobacteria (Niyomvong et al., 2012) and capable of inducing spore germination in some species of Streptomyces, Nocardia, Streptosporangium, Lentzea, Micromonospora, and Micropolyspora (currently transferred to Nocardia) (Bulina et al., 1997; Wang et al., 2013; Velikonja et al., 2014). For chemical pretreatment, the type and concentration of calcium salts are important for the isolation of actinobacteria (Fang et al., 2017b). Selective media supplemented with CaCO3 yield higher actinobacterial count than those supplemented with CaCl2 and (CH3COO)2Ca. The concentration of these three salts, at low concentration (0.1 and 0.01% (w/v) yield higher CFU of actinobacteria than in its absence or at high concentration. Calcium is important for environmental stress tolerance in actinobacteria because calcium forms a compound with dipicolinic acid as calcium dipicolinate and acts as secondary stabilizing agent for spore against environmental stress (Moir and Smith, 1990).
Novel Actinobacterial Taxa
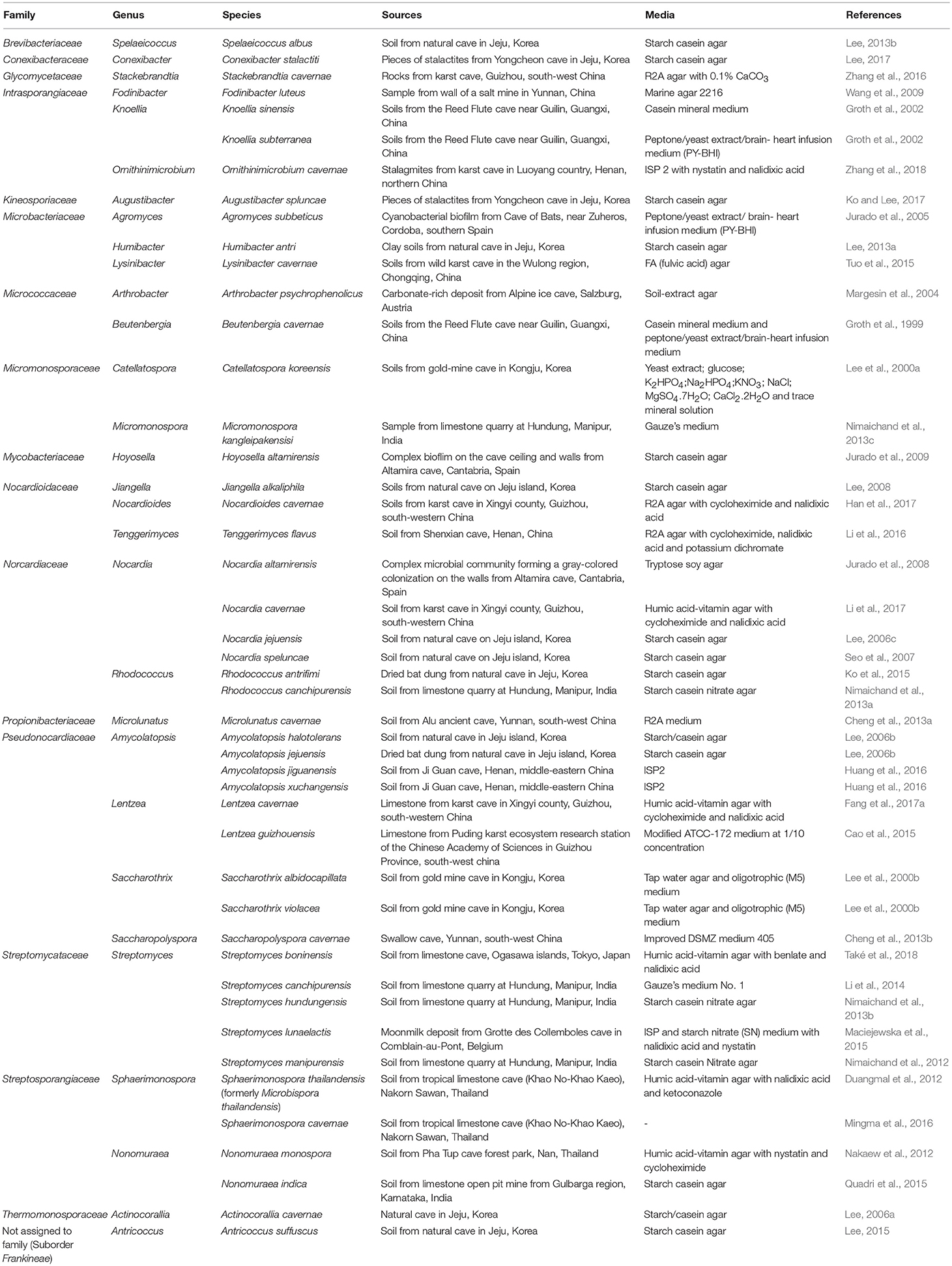
Several novel actinobacterial taxa isolated from caves and cave related habitats during the period of 20 years from 1999 to 2018 were summarized in Table 1. In total, 47 species within 30 genera were described including 7 novel genera. The highest number of novel species was from genus Streptomyces (5) followed by Amycolatopsis (4) and Nocardia (4). The majority of these novel actinobacteria were isolated from cave soils including 6 novel genera, Antricoccus, Beutenbergia, Knoellia, Lysinibacter Spelaeicoccus and Sphaerimonospora. Only the genus Hoyosella was recovered from complex biofilm on the ceiling and wall of Altamira cave, Spain. The extreme conditions within the caves are expected to create stress for the inhabitant microorganisms at the genetic level, paving the way for the evolution of new species and their novel metabolites (Tawari and Gupta, 2013). Therefore, caves are considered as an attractive source for the isolation of novel actinobacterial taxa.
Most species were isolated from selective media that were designed for the isolation of actinobacteria such as humic acid vitamin agar, starch casein agar, starch casein nitrate agar. However, some novel species were isolated using general cultivation media such as ISP2 media (Amycolatopsis jiguanensis and A. xuchangensis) and TSA (Nocardia altamirensis). In addition, low nutrient media (tap water agar and oligotrophic M5 media) were preferable for the isolation of Saccharothrix violacea and S. albidocapillata. Most novel species were incubated at 28°–30°C for 1–6 weeks. However, Arthrobacter psychrophenolicus was isolated at 4°C, this may be because this species originated from Alpine ice cave in Salzburg, Austria (Margesin et al., 2004). Lysinibacter cavernae was isolated at 15°C from soil in a wild karst cave in the Wulong region, Chongqing, China (Tuo et al., 2015). Streptomyces lunaelactis was isolated at 17°C from a moonmilk deposit in the Grotte des Collemboles cave in Belgium (Maciejewska et al., 2015).
Pretreatment procedures were also useful for isolation of some novel species. For example, Microbispora thailandensis was isolated from soil pretreated with microwave radiation at a frequency of 2460 MHz and power setting of 100 W for 45 s (Duangmal et al., 2012). Nonomuraea monospora was isolated from soil treated with phenol (Nakaew et al., 2012). Streptomyces manipurensis was isolated from soil supplemented with 0.1 g of CaCO3 for 1 day to prevent the growth of fast growing bacteria (Nimaichand et al., 2012).
Bioactive Compounds From Cave Actinobacteria
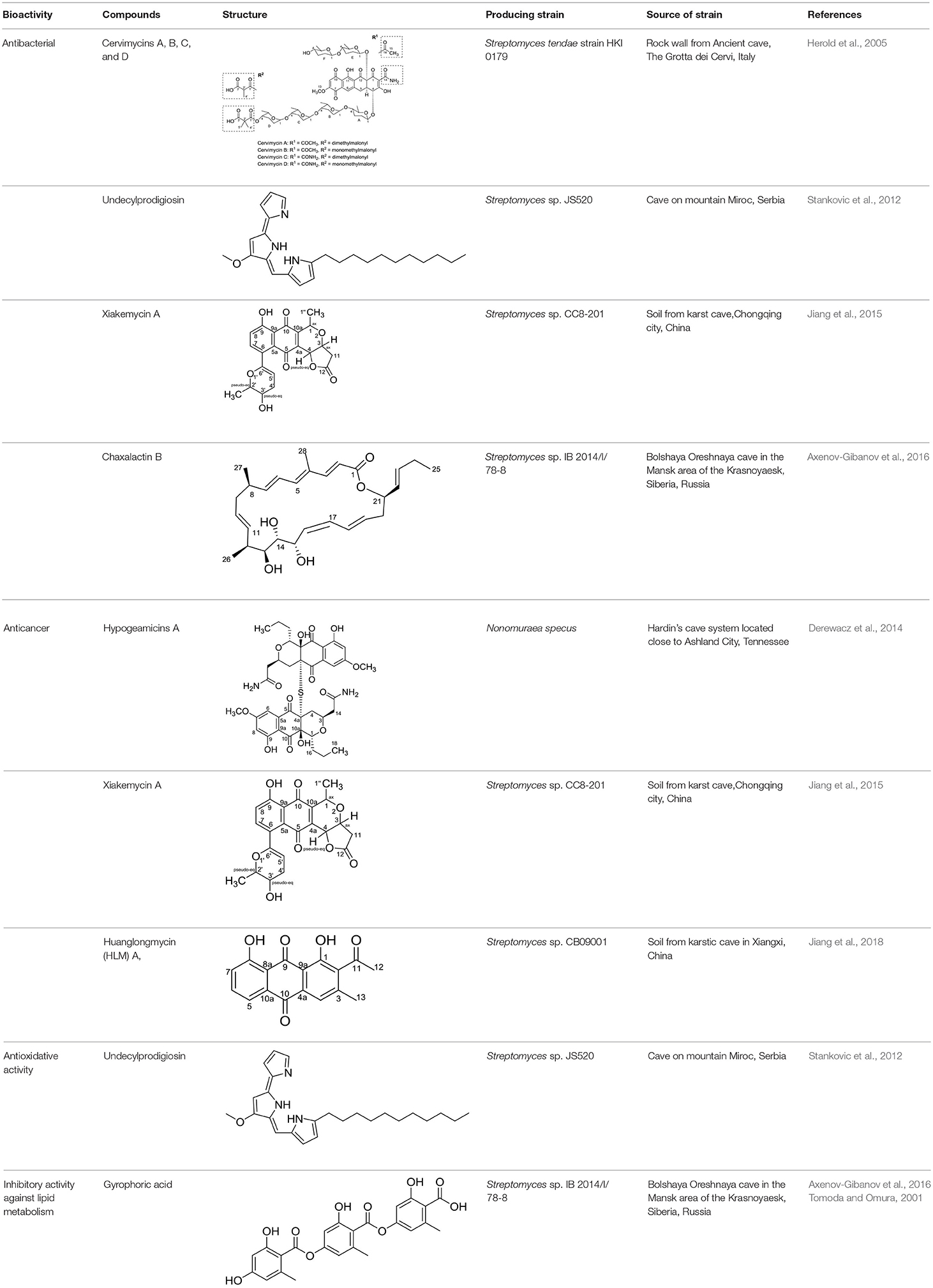
Caves are extreme habitats with low nutrient, temperature and light intensity but have high humidity (Schabereiter-Gurtner et al., 2002). These unique characteristics may promote the production of bioactive substances in particular antibiotics by actinobacteria (Nakaew et al., 2009a). Bioactive metabolites from cave associated actinobacteria have been purified, their structure elucidated and reported in recent years (Table 2). These compounds mostly displayed anti-bacterial and/or anti-cancer activities. The most prolific producer is members of the genus Streptomyces.
Cervimycin A, B, C, and D were produced from Streptomyces tendae strain HKI 0179, isolated from a rock wall in an ancient cave, the Grotta dei Cervi in Italy. Cervimycins A and B are novel polyketide glycosides. However, cervimycin C and D have the same structure as known compounds A2121-3 and A2121-2. Cervimycins A-D are highly active against Gram positive bacteria (B. subtilis and S. aureus) and multi-drug-resistant S. aureus (MRSA), vancomycin-resistant Enterococcus faecalis (VRE) and efflux-resistant S. aureus EfS4 (Herold et al., 2005).
Xiakemycin A is a novel pyranonaphthoquinone (PNQ) antibiotics produced by Streptomyces sp. CC8-201 from remote karst soil in China. Xiakemycin A showed strong inhibitory activities against Gram positive bacteria (S. aureus, S. epidermidis, E. faecalis, and E. faecium) and cytotoxic against numerous cancer cell lines (human lung cancer A549 cells, breast cancer MCF-7 cells, hepatoma HepG-2 cell, cervical cancer HeLa cells, colon carcinoma HCT-116 cell p53 wt cells, neuroblastoma SH-SY5Y cells, and human prostate cancer PC-3) (Jiang et al., 2015).
Hypogeamicins A, B, C, and D were produced by Nonomuraea specus isolated from Hardin's cave system in Tennessee, USA. Hypogeamicin A showed cytotoxicity to colon cancer cell line TCT-1 while hypogeamicin B-D were active against B. subtilis with no cytotoxicity to TCT-1. However, hypogeamicin B-D are not as potent as erythromycin and gentamicin in terms of antimicrobial activity against B. subtilis (Derewacz et al., 2014).
Huanglongmycin A, B, and C are aromatic polyketides from Streptomyces sp. CB09001, isolated from karstic cave soil of Xiangxi, China. Huanglongmycin A showed a weak anti-Gram negative bacteria (Pseudomonas aeruginosa and Escherichia coli) and moderate cytotoxicity against A549 lung cancer cell line. Huanglongmycin B has weak antibacterial activity against S. aureus and multi-drug-resistant S. aureus (MRSA). Huanglongmycin C showed neither antibacterial nor anticancer activities (Jiang et al., 2018). Undecylprodigiosin was produced by Streptomyces sp. JS520 isolated from sediments in cave in the mountain Miroc, Serbia. Undecylprodigiosin is a deep red pigment with antibacterial activity against Micrococcus luteus, B. subtilis, and C. albicans. Moreover, undecylprodigiosin also showed antioxidative and UV-protective properties (Stankovic et al., 2012).
Four known compounds with bioactivity (cyclodysidin D, chaxalactin B, stylissazole B, and gyrophoric acid) were reported to produce by Streptomyces sp. IB 2014/I/ 78-8 from moonmilk speleothem of Bolshaya Oreshnaya cave in Siberia (Axenov-Gibanov et al., 2016). Cyclodysidin D is previously reported in marine sponge, Dysidea tupha associated Streptomyces sp. RV 15. This compound showed no activity against bacteria, fungi and parasites (Abdelmohsen et al., 2014). Chaxalactin B was produced from Streptomyces sp. C34 from a hyper-arid soil samples collected from the Atacama Desert, Chile. This compound has strong activity against Gram positive bacteria (Castro et al., 2018). Stylissazole B was isolated from the marine sponge Stylissa carteri collected in the Solomon islands but no report on bioactivity (Patel et al., 2010). Gyrophoric acid isolated from Humicola sp. FO-2942 is an inhibitor of diacylglycerol acyltransferase and a lipid-lowering agent (Inokoshi et al., 2010).
Bioactivity of Uncharacterized Compounds
Several cave actinobacteria have been screened for their biological activity such as antibacterial, anticancer and antifungal. However, no pure compound and their structure were reported in these studies. The screening of only bioactivity without the structure elucidation of bioactive metabolites may not useful for the discovery of new antibiotics (Hug et al., 2018). Nevertheless, these findings provide evidence which supports the potential of cave actinobacteria to be exploited for novel bioactive compounds.
Turkish karstic caves were reported to harbor actinobacteria, for which 62% of the isolates, were active against several microbial pathogens (Gram positive bacteria, Gram negative bacteria, yeast, and filamentous fungi). Streptomyces sp. 1492 had strong activity against clinical strains of MRSA, VRE, and Acinetobacter baumanii (Yücel and Yamac, 2010). Streptomyces E9 isolated from Helmcken Falls cave in British Columbia could inhibit the growth of Paenibacillus larvae, a causative agent of American foulbrood disease in honeybees (Kay et al., 2013). A moonmilk speleothems of limestone caves, Grotte des Collemboles in Belgium were investigated for antimicrobial producing cultivable actinobacteria. A collection of obtained Streptomyces displayed strong inhibitory activity against Gram positive and Gram negative bacteria (Maciejewska et al., 2016). In a study of cultivable actinobacteria from Azores volcanic caves in Portugal, 18.1% of 148 actinobacterial isolates have antibacterial activity against at least one of the following bacteria: Salmonella typhimurium, E. coli, P. aeruginosa, Proteus sp., Listeria monocytogenes, L. innocua, and S. aureus. Most of the active isolates belong to the genus Streptomyces (S. nojiriensis, S. spiroverticillatus, S. avidinii, and S. mauvecolor) followed by Arthrobacter (Riquelme et al., 2017). A total of 40 taxa belonging to the genera Agromyces, Amycolatopsis, Kocuria, Micrococcus, Micromonospora, Nocardia, Streptomyces, and Rhodococcus were recovered from moonmilk deposits inside the Grotte des Collemboles, Belgium. Antimicrobial activity was found in isolated strains against Gram positive bacteria (87%) and Gram negative bacteria (59%) (Adam et al., 2018). Sixteen isolates of Streptomyces spp. from Chaabe cave in Algeria were screened for their antimicrobial activity using agar cylinder method. All of them showed strong anti-Gram positive (S. aureus, M. luteus, L. monocytogenes, and B. subtilis) activity (Belyagoubi et al., 2018).
For anticancer activity, a rare actinobacterium Spirillospora albida strain CMU-PNK470 was isolated from Phanangkhoi cave in northern Thailand (Nakaew et al., 2009a). This bacterium showed activity against human small lung cancer cell (NCI-H1870) with an IC50 value of 10.18 μg/ml. Similarly, Nonomurea roseola strain PT708 isolated from Phatup cave forest park in northern Thailand was tested positive for anticancer activity against human oral cavity cancer (KB) and human small lung cancer cells (NCI-H187) (Nakaew et al., 2009b). Moreover, these two strains are also active against some Gram positive pathogenic bacteria (B. cereus, MRSA, and Paenibacillus larvae).
Some examples of antifungal activity from cave actinobacteria have been reported. Antagonistic Streptomyces, Micromonospora, Streptosporangium, and Dactylosporangium were isolated from five caves (Cheondong, Kosoo, Nadong, Seonglyu, and Ssangyong) in Korea (Kim et al., 1998). They showed activity against at least one of plant pathogenic fungi (Alternaria solani, Colletotrichum gloeosporioides, Fusarium oxysporum f.sp. lycopersici, Magnaporthe grisea, Phytophthora capsici, and Rhizoctonia solani). Similarly, members of genera Streptomyces and Janibacter isolated from limestone deposit sites in Hundung, Manipur, India were reported to show anticandidal and biocontrol activities against rice fungal pathogens (Curvularia oryzae, F. oxysporum, Helminthosporum oryzae, Pyricularia oryzae, R. pryzae-sativae, and R. solani) as well as antibacterial activity (Nimaichand et al., 2015). However, Amycolatopsis, Rhodococcus, and Pseudonocardia isolates showed only biocontrol activity against rice fungal pathogen. Recently, five Streptomyces spp. from Chaabe cave in Algeria was reported to produce non-polyenic antifungal substances active against C. albicans (Belyagoubi et al., 2018).
Conclusion and Future Perspectives
Emerging and re-emerging infectious diseases are threatening human society at an alarming rate. It is a call of emergency to find an effective cure for these pathogens. Actinobacteria are proving again to be prolific producers of promising bioactive compounds with widely application. Cave and karst environments are underexplored microbiologically and should not be overlooked for the search and discovery of novel actinobacteria and their chemical diversity of useful compounds. It is evident from this mini-review that cave environments harbor novel and diverse actinobacteria (Table 1). These actinobacteria offer a rich source of bioactive compounds as exemplified in Table 2. We opine that in order to explore cave actinobacteria to their full potential, 2 major research area must be addressed. The first area of research should deal with the ability to isolate and cultivate actinobacteria of interest. It is well-accepted that most microorganisms could not be cultivated in laboratory. The isolation and cultivation of bioactive producing actinobacteria under laboratory conditions represent the first challenge. Currently, the isolation strategy specifically for cave actinobacteria is lacking. There is still an urgent need for an improved selective isolation to target specific actinobacterial taxa of interest and extended our ability to tap into the majority of these uncultivable bacteria. Modification of growth conditions and use of new culturing methods were proposed for cultivation of previously uncultivable microorganisms (Pham and Kim, 2012). A combination of enrichment techniques including heat-pretreatments of samples, adjusting media pH and calcium salts supplements were effectively applied to isolate rare actinobacteria from karstic caves (Fang et al., 2017b).
The advancement of next generation sequencing and accumulation of high quality whole genome data provide a powerful tool and useful information to support the search for novel bioactive metabolites for drug development. Currently, these genome data of actinobacteria reveal the presence of several biosynthetic gene clusters of secondary metabolites and reaffirm status of actinobacteria as prolific producers of bioactive compounds. However, these gene clusters are not normally expressed under laboratory conditions. Many secondary metabolites encoded by these gene clusters remain unidentified in fermentation broth (Scherlach and Hertweck, 2009; Ren et al., 2017). Therefore, the second challenge lies in our ability to activate these silent gene clusters. Recently, specific biological and chemical stimuli namely exposure to antibiotics, metals and mixed microbial culture, were successfully employed to activate secondary metabolites production in cave actinobacteria (Covington et al., 2018). Evidently, the study on cave actinobacteria and their bioactive compounds is still at an early stage. There still remains room for further study to guarantee cave actinobacteria as producers of new bioactive compounds for the benefit of human well-being.
Author Contributions
PR contributed data for selective isolation, novel taxa, bioactive metabolites and Tables 1, 2. WP conceived the idea, wrote, and revised the whole manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This research work was supported by Center of Excellence on Biodiversity (BDC), Office of Higher Education Commission (Project BDC-PG3-161005) and partially supported by Chiang Mai University. PR is grateful to Graduate School, Chiang Mai University for TA/RA scholarship for the academic year 2018–2019.
References
Abdelmohsen, U. R., Bayar, K., and Hentschel, U. (2014). Diversity, abundance and natural products of marine sponge-associatd actinomycetes. Nat. Prod. Rep. 31, 381–399. doi: 10.1039/C3NP70111E
Adam, D., Maciejewska, M., Naôm, é, A., Martinet, L., Coppieters, W., Karim, L., et al. (2018). Isolation, characterization, and antibacterial activity of hard-to-culture actinobacteria from cave moonmilk deposits. Antibiotics 7:28. doi: 10.3390/antibiotics7020028
Axenov-Gibanov, D., Voytsekhovskaya, I. V., Tokovenko, B. T., Protasov, E. S., Gamaiunov, S. V., Rabets, Y. V., et al. (2016). Actinobacteria isolated from an underground lake moonmilk speleothem from the biggest conglomeratic karstic cave in Siberia as sources of novel biologically active compounds. PLoS ONE 11:e0149216. doi: 10.1371/journal.pone.0149216
Barka, E. A., Vatsa, P., Sanchez, L., Gaveau-Vaillant, N., Jacquard, C., Klenk, H.-P., et al. (2016). Taxonomy, physiology, and natural products of actinobacteria. Microbiol. Mol. Biol. Rev. 80, 1–43. doi: 10.1128/MMBR.00019-15
Barton, A. H., Taylor, M. R., and Pace, N. R. (2004). Molecular phylogenetic analysis of a bacterial community in an oligotrophic cave environment. Geomicrobiol. J. 21, 11–20. doi: 10.1080/01490450490253428
Barton, H. A. (2006). Introduction to cave microbiology: a review for the non-specialist. J. Cave Karst Stud. 68, 43–54.
Belyagoubi, L., Belyagoubi-Benhammou, N., Jurada, V., Dupont, J., Lacost, S., Djebbah, F., et al. (2018). Antimicrobial activities of culturable microorganisms (actinomycetes and fungi) isolated from Chaabe cave, Algeria. Int. J. Speleol. 47, 189–199. doi: 10.5038/1827-806X.47.2.2148
Bulina, T. I., Alferova, I. V., and Terkhova, L. P. (1997). A novel approach to isolation of actinomycetes involving irradiation of soil samples with microwaves. Microbiology 66, 231–234.
Cao, C. L., Zhou, X. Q., Qin, S., Tao, F. X., Jiang, J. H., and Lian, B. (2015). Lentzea guizhouensis sp. nov., a novel lithophilous actinobacterium isolated from limestone from the karst area, Guizhou, China. Antonie van Leeuwenhoek 108, 1365–1372. doi: 10.1007/s10482-015-0589-x
Castro, J. F., Razmilic, V., Gomez-Escribano, J. P., Andrews, B., Asenjo, J., and Bibb, M. (2018). The ‘gifted’ actinomycetes Streptomyces leeuwenhoekii. Antonie van Leeuwenhoek 111, 1433–1488. doi: 10.1007/s10482-018-1034-8
Cheeptham, N., Sadoway, T., Rule, D., Watson, K., Moote, P., Soliman, L. C., et al. (2013). Cure from the cave: volcanic cave actinomycetes and their potential in drug discovery. Int. J. Speleol. 42, 35–47. doi: 10.5038/1827-806X.42.1.5
Cheng, J., Chen, W., Zhang, B. H., Nimaichand, S., Zhou, E. M., Lu, X. H., et al. (2013a). Microlunatus cavernae sp. nov., a novel actinobacterium isolated from Alu ancient cave, Yunnan, south-west China. Antonie van Leeuwenhoek 104, 95–101. doi: 10.1007/s10482-013-9929-x
Cheng, J., Zhang, Y. G., Chen, W., Li, L., Zhang, D. F., Wang, H. F., et al. (2013b). Saccharopolyspora cavernae sp. nov., a novel actinomycete isolated from the Swallow cave in Yunnan, south-west China. Antonie van Leeuwenhoek 104, 837–843. doi: 10.1007/s10482-013-9996-z
Covington, B. C., Spraggins, J. M., Ynigez-Gutierrez, A. E., Hylton, Z. B., and Bachmann, B. O. (2018). Response of secondary metabolism of hypogean actinobacterial genera to chemical and biological stimuli. Appl. Environ. Microbiol. 84, e01125–e01118. doi: 10.1128/AEM.01125-18
Derewacz, D. K., McNees, C. R., Scalmani, G., Covington, C. L., Shanmugam, G., Marnett, L. J., et al. (2014). Structure and stereochemical determination of hypogeamicins from a cave-derived actinomycete. J. Nat. Prod. 77, 1759–1763. doi: 10.1021/np400742p
Duangmal, K., Mingma, R., Pathom-aree, W., Niyomvong, N., Inahashi, Y., Matsumoto, A., et al. (2012). Microbispora thailandensis sp. nov., an actinomycete isolate from cave soil. J. Antibiot. 65, 491–494. doi: 10.1038/ja.2012.57
Fang, B. Z., Han, M. X., Liu, L., Zhang, Z. T., Liu, W. L., Shen, J. T., et al. (2017a). Lentzea covernae sp. nov., an actinobacterium isolated from a karst cave sample, and emended description of the genus Lentzea. Int. J. Syst. Evol. Microbiol. 67, 2357–2362. doi: 10.1099/ijsem.0.002364
Fang, B. Z., Nimaichand, S., Han, M. X., Jiao, J. Y., Cheng, J., Wei, D. Q., et al. (2017b). Insights on the effects of heat pretreatment, pH, and calcium salts on isolation of rare Actinobacteria from karstic caves. Front. Microbiol. 8:1535. doi: 10.3389/fmicb.2017.01535
Genilloud, O. (2017). Actinomycetes: still a source of novel antibiotics. Nat. Prod. Rep. 34, 1203–1232. doi: 10.1039/C7NP00026J
Ghosh, S., Kuisiene, N., and Cheeptham, N. (2017). The cave microbiome as a source for drug discovery: reality or pipe dream? Biochem. Pharmacol. 134, 18–34. doi: 10.1016/j.bcp.2016.11.018
Groth, I., and Saiz-Jimenez, C. (1999). Actinomycetes in hypogean environments. Geomicrobiol. J. 16, 1–8. doi: 10.1080/014904599270703
Groth, I., Schumann, P., Schuetze, B., Augsten, K., Kramer, I., and Stackebrandt, E. (1999). Beutenbergia cavernae gen. nov., sp. nov., an L-lysine-containing actinomycete isolated from a cave. Int. J. Syst. Bacteriol. 49, 1733–1740. doi: 10.1099/00207713-49-4-1733
Groth, I., Schumann, P., Schutze, B., Augsten, K., and Stackebrandt, E. (2002). Knoellia sinensis gen. nov., sp. nov. and Knoellia subterranea sp. nov., two novel actinocacteria isolated from a cave. Int. J. Syst. Evol. Microbiol. 52, 77–84. doi: 10.1099/00207713-52-1-77
Han, M. X., Fang, B. Z., Tian, Y., Zhang, W. Q., Jiao, J. Y., Liu, L., et al. (2017). Nocardioides cavernae sp. nov., an actinobacterium isolated from a karst cave. Int. J. Syst. Evol. Microbiol. 67, 633–639. doi: 10.1099/ijsem.0.001676
Herold, K., Gollmick, F. A., Groth, I., Roth, M., Menzel, K. D., Mollmann, U., et al. (2005). Cervimycin A-D: a polyketide glycoside complex from a cave bacterium can defeat vancomycin resistance. Chem. Eur. J. 11, 5523–5530. doi: 10.1002/chem.200500320
Huang, J. R., Ming, H., Li, S., Zhao, Z. L., Meng, X. L., Zhang, J. X., et al. (2016). Amycolatopsis xuchangensis sp. nov. and Amycolatopsis jiguanensis sp. nov., isolated from soil. Antonie van Leeuwenhoek 109, 1423–1431. doi: 10.1007/s10482-016-0742-1
Hug, J. J., Bader, C. D., Remskar, M., Cirnski, K., and Muller, R. (2018). Concepts and methods to access novel antibiotics from actinomycetes. Antibiotics 7:44. doi: 10.3390/antibiotics7020044
Inokoshi, J., Takagi, Y., Uchida, R., Masuma, R., Omure, S., and Tomoda, H. (2010). Production of a new type of amidepsine with a sugar moiety by static fermentation of Humicola sp. FO-2942. J. Antibiot. 63, 9–16. doi: 10.1038/ja.2009.110
Jiang, L., Pu, H., Xiang, J., Su, M., Yan, X., Yang, D., et al. (2018). Huanglongmycin A-C, cytotoxic polyketides biosynthesized by a putative type II polyketide synthase from Streptomyces sp. CB09001. Front. Chem. 6:254. doi: 10.3389/fchem.2018.00254
Jiang, Y., Li., Q., Chen, X., and Jiang, C. (2016). “Isolation and cultivation methods of Actinobacteria” in Actinobacteria- Basics and Biotechnological Applications, ed. D. Dhanasekaran (Rijeka: InTech), 39–57.
Jiang, Z. K., Guo, L., Chen, C., Liu, S. W., Zhang, L., Dai, S. J., et al. (2015). Xiakemycin A, a novel pyranonaphthoquinone antibiotic, produced by the Streptomyces sp. CC8-201 from the soil of a karst cave. J. Antibiot. 68, 771–774. doi: 10.1038/ja.2015.70
Jurado, V., Kroppenstedt, R. M., Jimenez, C. S., Klenk, H.-P., Mounieé, D., Laiz, L., et al. (2009). Hoyosella altamirensis gen. nov., sp. nov., a new member of the order Actinomycetales isolated from a cave bioflim. Int. J. Syst. Evol. Microbiol. 59, 3105–3110. doi: 10.1099/ijs.0.008664-0
Jurado, V., Boiron, P., Kroppenstedt, R. M., Laurent, F., Couble, A., Laiz, L., et al. (2008). Nocardia altamirensis sp. nov., isolated from Altamira cave, Cantabria, Spain. Int. J. Syst. Evol. Microbiol. 58, 2210–2214. doi: 10.1099/ijs.0.65482-0
Jurado, V., Groth, I., Gonzalez, J. M., Laiz, L., and Jimenez, C. S. (2005). Agromyces subbeticus sp. nov., isolated from a cave in southern Spain. Int. J. Syst. Evol. Microbiol. 55, 1897–1901. doi: 10.1099/ijs.0.63637-0
Kay, S., Pathom-aree, W., and Cheeptham, N. (2013). Screening of volcanic cave actinomycetes for antimicrobial activity against Paenibacillus larvae, a honey bee pathogen. Chiang Mai J. Sci. 40. 26–33.
Kemung, H. M., Tan, L. T. H., Khan, T. M., Chan, K. G., Pusparajah, P., Goh, B. H., et al. (2018). Streptomyces as a prominent resource of future anti-MRSA drugs. Front. Microbiol. 9:2221. doi: 10.3389/fmicb.2018.02221
Kim, B. S., Lee, J. Y., and Hwang, B. K. (1998). Diversity of actinomycetes antagonistic to plant pathogenic fungi in cave and sea-mud soils of Korea. J. Microbiol. 36, 86–92.
Ko, D. H., and Lee, S. D. (2017). Augustibacter spluncae sp. nov., isolated from a lava cave stalactite. Int. J. Syst. Evol. Microbiol. 67, 3283–3288. doi: 10.1099/ijsem.0.002108
Ko, K. S., Kim, Y., Seong, C. N., and Lee, S. D. (2015). Rhodococcus antrifimi sp. nov., isolated from dried bat dung of a cave. Int. J. Syst. Evol. Microbiol. 65, 4043–4048. doi: 10.1099/ijsem.0.000534
Laiz, L., Gonzalez-Delvalle, M., Hermosin, B., Ortiz-Martinez, A., and Saiz-Jimenez, C. (2003). Isolation of cave bacteria and substrate utilization at different temperature. Geomicrobiol. J. 20, 479–489. doi: 10.1080/713851125
Laiz, L., Groth, I., Gonzalez, I., and Saiz-Jimenez, C. (1999). Microbiological study of the dripping waters in Altamira cave (Santillana del mar, Spain). J. Microbiol. Methods 36, 129–138. doi: 10.1016/S0167-7012(99)00018-4
Lee, S. D. (2006a). Actinocorallia cavernae sp. nov., isolated from a natural cave in Jeju, Korea. Int. J. Syst. Evol. Microbiol. 56, 1085–1088. doi: 10.1099/ijs.0.63895-0
Lee, S. D. (2006b). Amycolatopsis jejuensis sp. nov. and Amycolatopsis halotolerans sp. nov., novel actinomycetes isolated from a natural cave. Int. J. Syst. Evol. Microbiol. 56, 549–553. doi: 10.1099/ijs.0.63881-0
Lee, S. D. (2006c). Nocardia jejuensis sp. nov., novel actinomycetes isolated from a natural cave on Jeju island, Republic of Korea. Int. J. Syst. Evol. Microbiol. 56, 559–562. doi: 10.1099/ijs.0.63866-0
Lee, S. D. (2008). Jiangella alkaliphila sp. nov., an actinobacterium isolated from a cave. Int. J. Syst. Evol. Microbiol. 58, 1176–1179. doi: 10.1099/ijs.0.65479-0
Lee, S. D. (2013a). Humibacter antri sp. nov., an actinobactrium isolated from a natural cave, and emended description of the genus Humibacter. Int. J. Syst. Evol. Microbiol. 63, 4315–4319. doi: 10.1099/ijs.0.050708-0
Lee, S. D. (2013b). Spelaeicoccus albus gen. nov., sp. nov., an actinobacterium isolated from a natural cave. Int. J. Syst. Evol. Microbiol. 63, 3958–3963. doi: 10.1099/ijs.0.050732-0
Lee, S. D. (2015). Antricoccus suffuscus gen. nov., sp. nov., isolated from a natural cave. Int. J. Syst. Evol. Microbiol. 65, 4410–4416. doi: 10.1099/ijsem.0.000588
Lee, S. D. (2017). Conexibacter stalactiti sp. nov., isolated from stalactites in a lava cave and emended description of the genus Conexibacter. Int. J. Syst. Evol. Microbiol. 67, 3214–3218. doi: 10.1099/ijsem.0.002083
Lee, S. D., Kang, S. O., and Hah, Y. C. (2000a). Catellatospora koreensis sp. nov., a novel actinomycete isolated from a gold-mine cave. Int. J. Syst. Evol. Microbiol. 50, 1103–1111. doi: 10.1099/00207713-50-3-1103
Lee, S. D., Kim, E. S., Roe, J. H., Kim, J. H., Kang, S. O., and Hah, Y. C. (2000b). Saccharothrix violacea sp. nov., isolated from a gold mine cave, and Saccharothrix albidocapillata comb. nov. Int. J. Syst. Evol. Microbiol. 50, 1315–1323. doi: 10.1099/00207713-50-3-1315
Li, Q. Q., Han, M. X., Fang, B. Z., Jiao, J. Y., Liu, L., Yang, Z. W., et al. (2017). Nocardia cavernae sp. nov., a novel actinobacterium isolated from a karst cave sample. Int. J. Syst. Evol. Microbiol. 67, 2998–3003. doi: 10.1099/ijsem.0.002072
Li, W. J., Nimaichand, S., Jiang, Z., Liu, M. J., Khieu, T. N., Kim, C. J., et al. (2014). Streptomyces canchipurensis sp. nov., isolated from a limestone habitat. Antonie van Leeuwenhoek 106, 1119–1126. doi: 10.1007/s10482-014-0281-6
Li, X. J., Dai, S. J., Liu, S. W., Liu, J. M., Chen, L., Hu, L., et al. (2016). Tenggerimyces flavus sp. nov., isolated from soil in a karst cave, and emended description of the genus Tenggerimyces. Int. J. Syst. Evol. Microbiol. 66, 1499–1505. doi: 10.1099/ijsem.0.000908
Maciejewska, M., Adam, D., Martinet, L., Naome, A., Calusiriska, M., Delfosse, P., et al. (2016). A phenotypic and genotypic analysis of the antimicrobial potential of cultivable Streptomyces isolated from cave moonmilk deposits. Front. Microbiol. 7:1455. doi: 10.3389/fmicb.2016.01455
Maciejewska, M., Pessi, I. S., Arias, A. A., Noirfalise, P., Luis, G., Ongena, M., et al. (2015). Streptomyces lunaelactis sp. nov., a novel ferroverdin A-producing Streptomyces species isolated from a moonmilk speleothem. Antonie van Leeuwenhoek 107, 519–531. doi: 10.1007/s10482-014-0348-4
Margesin, R., Schumann, P., Spröer, C., and Gounot, A. M. (2004). Arthrobacter psychrophenolicus sp. nov., isolated from an alpine ice cave. Int. J. Syst. Evol. Microbiol. 54, 2067–2072. doi: 10.1099/ijs.0.63124-0
Mingma, R., Duangmal, K., Také, Inahashi, Y., Ōmura, S., Takahashi, Y., and Matsumoto, A. (2016). Proposal of Sphaerimonospora cavernae gen. nov., sp. nov. and transfer of Microbispora mesophila (Zhang et al., 1984). Sphaerimonospora mesophila comb. nov. and Microbispora thailandensis (Duangmal et al., 2012) to Sphaerimonospora thailandensis comb. nov. Int. J. Syst. Evol. Microbiol. 66, 1735–1744. doi: 10.1099/ijsem.0.000935
Moir, A., and Smith, D. A. (1990). The genetics of bacterial spore germination. Annu. Rev. Microbiol. 44, 531–553. doi: 10.1146/annurev.mi.44.100190.002531
Nakaew, N., Pathom-aree, W., and Lumyong, S. (2009a). First record of the isolation, identification and biological activity of a new strain of Spirillospora albida from Thai cave soils. Actinomycetologica 23, 1–7. doi: 10.3209/saj.SAJ230102
Nakaew, N., Pathom-aree, W., and Lumyong, S. (2009b). Generic diversity of rare actinomycetes from Thai cave soils and their possible use as new bioactive compounds. Actinomycetologica 23, 21–26. doi: 10.3209/saj.SAJ230201
Nakaew, N., Sungthong, R., Yokota, A., and Lumyong, S. (2012). Nonomuraea monospora sp. nov., an actinomycete isolated from cave soil in Thailand, and emended description of the genus Nonomuraea. Int. J. Syst. Evol. Microbiol. 62, 3007–3012. doi: 10.1099/ijs.0.035220-0
Nimaichand, S., Devi, A. M., Tamreihao, K., Ningthoujam, D. S., and Li, W.-J. (2015). Actinobacterial diversity in limestone deposit sites in Hundung, Manipur (India) and their antimicrobial activities. Front. Microbiol 6:413. doi: 10.3389/fmicb.2015.00413
Nimaichand, S., Sanasam, S., Zheng, L.-Q., Zhu, W.-Y., Yang, L.-L., Tang, S.-K., et al. (2013a). Rhodococcus canchipurensis sp. nov., an actinomycete isolated from a limestone deposit site. Int. J. Syst. Evol. Microbiol. 63, 114–118. doi: 10.1099/ijs.0.036087-0
Nimaichand, S., Tamrihao, K., Yang, L. L., Zhu, W. Y., Zhang, Y. G., Li, L., et al. (2013b). Streptomyces hundungensis sp. nov., a novel actinomycete with antifungal activity and plant growth promoting traits. J. Antibiot. 66, 205–209. doi: 10.1038/ja.2012.119
Nimaichand, S., Zhang, Y.-G., Cheng, J., Li, L., Zhang, D.-F., Zhou, E.-M., et al. (2013c). Micromonospora kangleipakensis sp. nov., isolated from a sample of limestone quarry. Int. J. Syst. Evol. Microbiol. 63, 4546–4551. doi: 10.1099/ijs.0.052746-0
Nimaichand, S., Zhu, W. Y., Yang, L. L., Ming, H., Nie, G. X., Tang, S. K., et al. (2012). Streptomyces manipurensis sp. nov., a novel actinomycete isolated from a limestone deposit site in Manipur, India. Antonie van Leeuwenhoek 102, 133–139. doi: 10.1007/s10482-012-9720-4
Niyomvong, N., Pathom-aree, W., Thamchaipenet, A., and Duangmal, K. (2012). Actinomycetes from tropical limestone caves. Chiang Mai J. Sci. 39, 373–388.
Northup, D. E., and Lavoie, K. H. (2001). Geomicrobiology of caves: a review. Geomicrobiol. J. 18, 199–222. doi: 10.1080/01490450152467750
Passari, A. K., Misha, V. K., Gupta, V. K., and Singh, B. P. (2018). “Chapter 1: methods used for the recovery of culturable endophytic actinobacteria: an overview,” in Actinobacteria: Diversity and Biotechnological Applications: New and Future Developments in Microbial Biotechnology and Bioengineering, eds B. P. Singh, A. K. Passari, and V. K. Gupta (Amsterdam: Elsevier), 1–11.
Patel, K., Laille, R., Martin, M. T., Tilvi, S., Moriou, C., Gallard, J. F., et al. (2010). Unprecedented stylissazoles A-C from Stylissa carteri: another dimension for marine pyrrole-2-aminoimadazole metabolite diversity. Angew. Chem. Int. Ed. 49, 4775–4779. doi: 10.1002/anie.201000444
Pham, V. H., and Kim, J. (2012). Cultivation of unculturable soil bacteria. Trends Biotechnol. 30, 475–484. doi: 10.1016/j.tibtech.2012.05.007
Quadri, S. R., Tian, X. P., Zhang, J., Li, J., Nie, G. X., Tang, S. K., et al. (2015). Nonomuraea indica sp. nov., novel actinomycetes isolated from lime-stone open pit mine, India. J. Antibiot. 68, 491–495. doi: 10.1038/ja.2015.24
Ren, H., Wang, B., and Zhao, H. (2017). Breaking the silence: New strategies for discovering novel natural products. Curr. Opin. Biotechnol. 48, 21–27. doi: 10.1016/j.copbio.2017.02.008
Riquelme, C., Dapkevicius, M. L. E., Miller, A. Z., Charlop-Powers, Z., Brady, S., Mason, C., et al. (2017). Biotechnological potential of actinobacteria from Canadian and Azorean volcanic caves. Appl. Microbiol. Biotechnol. 101, 843–857. doi: 10.1007/s00253-016-7932-7
Schabereiter-Gurtner, C., Saiz-Jimenez„, C., Pinar, G., Lubitz, W., and Röllke, S. (2002). Altamira cave paleolithic paintings harbor partly unknown bacterial communities. FEMS Microbiol. Lett. 211, 7–11. doi: 10.1111/j.1574-6968.2002.tb11195.x
Scherlach, K., and Hertweck, C. (2009). Triggering cryptic natural product biosynthesis in microorganisms. Org. Biomol. Chem. 7, 1753–1760. doi: 10.1039/b821578b
Seo, J. P., Yun, Y. W., and Lee, S. D. (2007). Nocardia speluncae sp. nov., isolated from a cave. Int. J. Syst. Evol. Microbiol. 57, 2932–2935. doi: 10.1099/ijs.0.65085-0
Stankovic, N., Radulovic, V., Petkovic, M., Vuckovic, I., Jadranin, M., Vasiljevic, B., et al. (2012). Streptomyces sp. JS520 produces exceptionally high quantities of undecylprodigiosin with antibacterial, antioxidative, and UV-protective properties. Appl. Microbiol. Biotechnol. 96, 1217–1231. doi: 10.1007/s00253-012-4237-3
Takahashi, Y., and Nakashima, T. (2018). Actinomycetes, an inexhaustible source of naturally occurring antibiotics. Antibiotics 7:45. doi: 10.3390/antibiotics7020045
Také, A., Inahashi, Y., Ōmura, S., Takahashi, Y., and Matsumoto, A. (2018). Streptomyces boninensis sp. nov., isolated from soil from a limestone cave in the Ogasawara islands. Int. J. Syst. Evol. Microbiol. 68, 1795–1799. doi: 10.1099/ijsem.0.002753
Tawari, K., and Gupta, R. K. (2013). Diversity and isolation of rare actinomyctes: an overview. Crit. Rev. Microbiol. 39, 256–294. doi: 10.3109/1040841X.2012.709819
Tomczyk-Zak, K., and Zielenkiewicz, U. (2016). Microbial diversity in caves. Geomicrobiol. J. 33, 20–38. doi: 10.1080/01490451.2014.1003341
Tomoda, H., and Omura, S. (2001). “Screening for inhibitors of lipid metabolism,” in Enzyme Technologies for Pharmaceutical and Biotechnological Applications, eds. H.A. Kirst, W.K. Yeh, M.J. Zmijewski, Jr. (New York, NY: Marcel Dekker Inc), 343–378.
Tuo, L., Liu, S. W., Liu, J. M., Zhang, Y. Q., Jiang, Z. K., Liu, X. F., et al. (2015). Lysinibacter cavernae gen. nov., sp. nov., a new member of the family Microbacteriaceae isolated from a karst cave. Int. J. Syst. Evol. Microbiol. 65, 3305–3312. doi: 10.1099/ijsem.0.000415
Velikonja, B. H., Tkave, R., and Pasic, L. (2014). Diversity of cultivable bacteria involved in the formation of macroscopic microscopic microbial colonies (cave silver) on the walls of a cave in Slovenia. Int. J. Speleol. 43, 45–46. doi: 10.5038/1827-806X.43.1.5
Wang, D. S., Xue, Q. H., Zhu, W. J., Zhao, J., Duan, J. L., and Shen, G. H. (2013). Microwave irradiation is a useful tool for improving isolation of actinomycetes from soil. Microbiology 82, 102–110. doi: 10.1134/S0026261712060161
Wang, Z. G., Wang, Y. X., Liu, J. H., Chen, Y. G., Zhang, X. X., Wen, M. L., et al. (2009). Fodinibacter luteus gen. nov., sp. nov., an actinobactrium isolated from a salt mine. Int. J. Syst. Evol. Microbiol. 59, 2185–2190. doi: 10.1099/ijs.0.006882-0
Yücel, S., and Yamac, M. (2010). Selection of Streptomyces isolates from Turkish karstic caves against antibiotic resistant microorganisms. Pak. J. Pharm. Sci. 23, 1–6.
Zhang, L. Y., Ming, H., Meng, X. L., Fang, B. Z., Jiao, J. Y., Nimaichand, S., et al. (2018). Ornithinimicrobium cavernae sp. nov., an actinobactrium isolated from a karst cave. Antonie van Leeuwenhoek. 112, 179–186. doi: 10.1007/s10482-018-1141-6
Keywords: actinobacteria, cave, karst, bioactive compounds, diversity, antimicrobial, anticancer, natural products
Citation: Rangseekaew P and Pathom-aree W (2019) Cave Actinobacteria as Producers of Bioactive Metabolites. Front. Microbiol. 10:387. doi: 10.3389/fmicb.2019.00387
Received: 15 November 2018; Accepted: 13 February 2019;
Published: 22 March 2019.
Edited by:
Learn-Han Lee, Monash University Malaysia, MalaysiaReviewed by:
Sheng Qin, Jiangsu Normal University, ChinaJoachim Wink, Helmholtz Center for Infection Research, Helmholtz Association of German Research Centers (HZ), Germany
Copyright © 2019 Rangseekaew and Pathom-aree. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Wasu Pathom-aree, d2FzdS5wQGNtdS5hYy50aA==
 Pharada Rangseekaew
Pharada Rangseekaew Wasu Pathom-aree
Wasu Pathom-aree