- 1Department of Gynecology, Beijing Obstetrics and Gynecology Hospital, Capital Medical University, Beijing, China
- 2Ultrastructural Pathology Center, Peking University First Hospital, Beijing, China
- 3Laboratory of Electron Microscopy, Department of Gynecology, Minimally Invasive Center, Beijing Obstetrics and Gynecology Hospital, Capital Medical University, Beijing, China
A comprehensive knowledge of the vaginal ecosystem is critical for the development of successful approaches to the treatment of infections. The role of Lactobacilli in preventing vulvovaginal candidiasis (VVC) is controversial. In this study, we investigated the therapeutic effects and mechanism of Lactobacillus crispatus or delbrueckii on vaginitis caused by Candida albicans in a Sprague–Dawley rat model. A microbiological evaluation was performed by Gram staining and fungal colonies were enumerated. The antifungal efficacy of the two Lactobacillus strains was assessed by hematoxylin and eosin (HE) staining, transmission electron microscopy (TEM), immunohistochemical detection of interferon-γ (IFN-γ), interleukin (IL)-4, IL-17, and epithelial-derived IgG (RP125). Our in vitro results showed that the inhibitory activity against Candida colony-forming unit (CFU) counts was demonstrated by the two Lactobacillus strains (P < 0.001). Our results indicated that Lactobacillus administration played an indispensable role in maintaining the immune homeostasis, and decreasing the Th1/Th2 ratio (IFN-γ/IL-4) by regulating the epithelial secretion of cytokines that inhibit epithelial proinflammatory cytokine release, while increasing epithelial-derived IgG expression (P < 0.05), suggesting antibody-mediated protection. Our results implicate L. crispatus and L. delbrueckii as a potential adjunct biotherapeutic agent in women with VVC, especially for those with drug resistance, adverse effects or contraindications when using antifungal agents. Further large, long-term, well-planned clinical studies should be performed in clinical practice to determine their clinical potential of L. crispatus and L. delbrueckii as an adjunct treatment for VVC.
Introduction
It is now well accepted that the microbiota present in the human body can impact immunity, physiology, and health (Song et al., 2018). It has been postulated that the unique vaginal microbiome has evolved to perform the dual roles of disease resistance and obstetric protection (Amabebe and Anumba, 2018). Moreover, infections can arise from imbalances in the highly diverse vaginal microbiota (e.g., VVC). VVC, caused primarily by Candida, is the second-most common vaginal infecti and is associated with vulval discomfort or and pain (Dovnik et al., 2015). The recommended standard therapy for vaginal C. albicans infections is antifungal therapy, consisting of oral or intravaginal azole, or triazole drugs (Sobel and Sobel, 2018). However, prolonged use of antibiotic administration increases recurrence rates, probably due to an inability to re-establish the normal Lactobacillus-dominated vaginal flora.
In recent years there has been growing interest in the therapeutic use of probiotics. Lactobacillus species in general are recommended as a new strategy, which is increasing in popularity based on accumulating evidence for their effectiveness in restoring normal microbial function and preventing urogenital infections (Amabebe and Anumba, 2018). Probiotic use has been associated with a significant reduction in the recurrence of VVC (Palacios et al., 2016). First introduced in 1974 by Parker (Fuller, 1999), the word “probiotic” is used to describe live microorganisms that provide a benefit to the recipient when administered in sufficient quantities (World Health Organization and Food and Agriculture Organization of the United Nations, 2006).
The vaginal microbiota in most healthy women of reproductive-age is dominated by protective Lactobacillus species that are thought to reinforce the defense against invasion and colonization by pathogenic microorganisms (Melgaço et al., 2018). In women with vaginal infections, such as BV and VVC, the dominance of these Lactobacilli is compromised (Sobel, 2007). Consequently, intravaginal probiotics of the Lactobacillus genus are common and act beneficially in a number of ways. In addition to their function as a barrier to vaginal colonization by harmful microorganisms (Kim and Park, 2017), these species maintain an acidic intravaginal microflora by generating lactic acid via carbohydrate decomposition (Cadieux et al., 2002), inhibit catalase-negative anaerobic organisms by production of hydrogen peroxide (H2O2) as a source of free radicals (Lamont, 2003) and produce antimicrobial peptides, such as bacteriocin-like substances and biosurfactants (Witkin et al., 2013). In addition, Lactobacillus species induce anti-inflammatory immune responses in the host via the NF-κB pathway (Santos et al., 2018).
More than 20 species of Lactobacilli have been detected in the vagina, of which L. crispatus is the most prevalent in healthy Asian women and has been widely investigated as a vaginal probiotic due to its strong antimicrobial activity (Ravel et al., 2011).
Lactobacillus crispatus confers protection on the epithelial barrier against injury and inflammation by regulating epithelial cell function (Anton et al., 2018). Our previous study showed that L. crispatus attenuates the virulence of C. albicans, modulates the secretion of cytokines and chemokines, and enhances the immune response of VK2/E6E7 cells in vitro (Niu et al., 2017). However, there are no commercially available vaginal preparations containing L. crispatus in the Chinese market. Live Lactobacillus Capsule for Vaginal Use, containing L. delbrueckii (Neimenggu Wanzeshuangqi Pharmaceutical Co., Ltd.) is the only commercial preparation of Lactobacillus that is currently available. This preparation has been introduced to inhibit the cytotoxic effects and adhesion of pathogenic organisms (Banerjee et al., 2009). L. delbrueckii produces large amounts of H2O2 and has been reported to inhibit C. albicans more effectively than many other vaginally derived strains found in healthy women (Strus et al., 2005). Due to the close anatomic relationship with the vagina, the anus is convenient for the migration of gut organisms, including oral administration of probiotic Lactobacillus strains (e.g., yogurt drinks), to the vagina. Passive transfer of Lactobacilli to the vagina from the rectum may be an important consideration in the delivery of probiotics in foods and dietary supplements (Buggio et al., 2019). Intake of oral probiotics containing Lactobacillus strains improved the recovery rate and symptoms of patients with vaginal infections and tended to improve the vaginal microbial pattern (Laue et al., 2018).
Considering the proven health promoting effects of L. crispatus, investigations of the efficacy of topical Lactobacillus strains on treatment of VVC are warranted. To date, few, if any, studies have assessed the anti-Candida activities and probiotic properties of Lactobacillus strains in VVC. Therefore, the objective of this study was to investigate and compare the effects of (1) L. crispatus as the predominant vaginal bacteria in humans, and (2) L. delbrueckii, which is contained in the only commercially available vaginal preparation of Lactobacillus strains, to prevent the invasion of C. albicans, additionally focusing on the immune response of the host vaginal epithelium in a rat model.
Materials and Methods
Microorganism and Growth Conditions
Lactobacillus crispatus [American Type Culture Collection (ATCC) 33820] was routinely cultured anaerobically at 37°C in de Man, Rogosa and Sharpe broth (Becton Dickinson, Cockeysville, MD, United States). After overnight culture, cells were collected by centrifugation (10,000 × g, 10 min, 5°C), washed twice, and resuspended in 0.9% saline solution at 3 × 108 CFU/ml. The vaginal Lactobacillus (Dingjunsheng®; Neimenggu Wanzeshuangqi Pharmaceutical Co., Ltd.) probiotic preparation consisted of 0.25 g capsules containing approximately 0.25 × 106 CFU of live L. delbrueckii. Suspensions were formulated daily in saline solution to achieve approximately 3 × 108 CFU/ml. L. crispatus or L. delbrueckii suspensions were administered intravaginally to each animal once daily at 3 × 108 CFU/ml.
Candida albicans strains (ATCC-11006) were cultured to the third generation on Sabouraud dextrose agar (SDA; Becton Dickinson) at 37°C for 72 h and harvested. After resuspension in 0.9% saline solution, the cells were counted using a hemocytometer (Hausser Scientific; Horsham, PA, United States) and adjusted to a final concentration of 1 × 105 CFU/ml.
Animals
Female Sprague–Dawley rats (n = 24, 210–240 g), obtained from Beijing Vital River Laboratory Animal Technology Co. Ltd. (Beijing, China), were maintained in cages under a 12-h light/ 12-h dark cycle with free access to commercial food and water.
Experimental Candidal Vaginitis
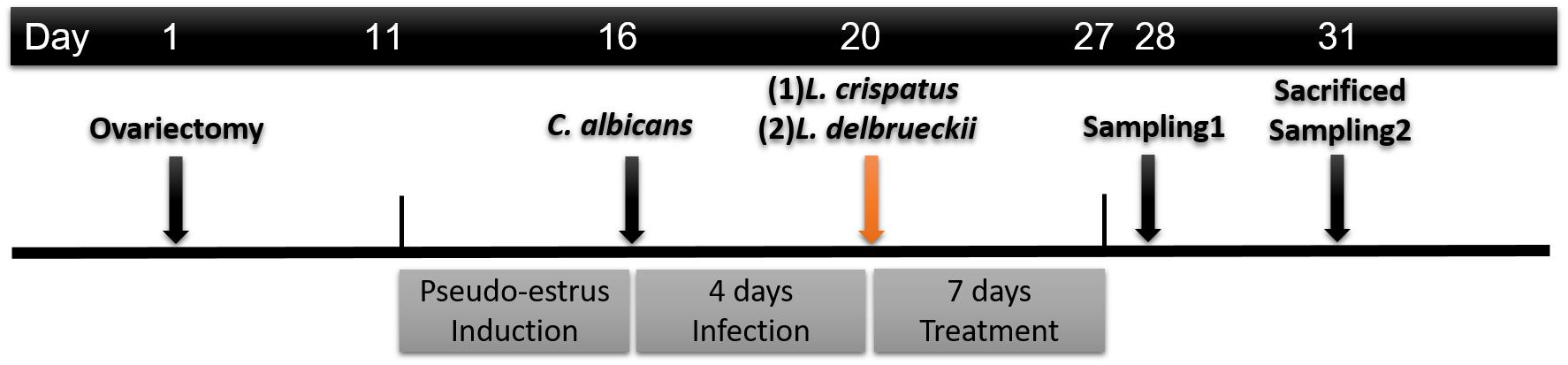
The timeline of the experimental animal treatment and sample collection is shown in Figure 1. Prior to infection, animals received subcutaneous injection of 0.5 mg estradiol benzoate (Estradiolo, Amsa Farmaceutici, Rome, Italy) at 2-day intervals until the end of the experiment to induce immunosuppression (Zhang et al., 2018). The hormonal activity was assessed by analysis of the enucleated epithelial cells in vaginal fluid smears verified under a light microscope; enucleated cornified cells indicated the pseudo-estrus phase (Mandl, 1951). The rats were inoculated intravaginally with a yeast suspension of 1 × 108 yeasts/mL (in 150 mL of sterile saline solution) of washed C. albicans blastoconidia. Four days after inoculation but before treatment, samples were obtained from the vaginal cavity to confirm C. albicans infection by Gram staining of the yeast/hyphae-like form and vaginal cells observed under light microscopy.
The rats were randomly divided into four groups (n = 6 rats per group). At 24 h after the initial infection, rats were randomly divided into following four groups (n = 6 rats per group); (a) uninfected (Control, vaginal inoculation with 0.9% saline solution); (b) infected (Model, vaginal inoculation with 0.9% saline solution); (c) infected but received intravaginal L. crispatus (approximately 3.0 × 108 CFU/mL) once a day for 7 days; and (d) infected but received intravaginal L. delbrueckii (approximately 3.0 × 108 CFU/mL) once a day for 7 days. At 1 (Day 28) and 4 days (Day 31) after the 7-day treatment period, a microbiological evaluation was performed by Gram staining. The C. albicans load in vaginal lavage (two sequential 500 μl volumes of sterile saline) was determined by the CFU assay on SDA. The antifungal effects were analyzed by enumerating the CFU/mL within the samples. All rats were anesthetized with an intraperitoneal injection of 100 mg/kg of ketamine hydrochloride and euthanized. Whole-vagina samples were harvested.
Histological Analysis
Following immersion in formalin, vaginal samples were cut into 4-μm thick sections and stained using a HE staining kit (Beyotime Biotechnology, Beijing, China) according to the manufacturer’s instructions. The tissues were then dehydrated by immersion in a gradient alcohol series followed by xylene clearing. After sealing with a neutral balsam, the tissues were evaluated for pathological changes under a microscope (Olympus).
Immunohistochemistry
Sections were incubated overnight at 4°C with the following primary detection antibodies: anti-rat-IFN-γ (1:100), anti-rat-IL-4 (1:200), anti-rat-IL-17 (1:50) (all rabbit polyclonal, Cloud-Clone, United States), and anti-IgG (RP215, provided by the Immunology Department, Peking University Health Science Center; 1:100). Sections were then incubated with horseradish peroxidase-conjugated anti-rabbit Ig (Zhongshan Golden Bridge Biotechnology) and immunoreactivity was developed using the 3,3′-diaminobenzidine (DAB) substrate system. Negative control sections were incubated with PBS instead of the primary antibody. Semi-quantitative analysis was performed by an experienced pathologist based on IRS (Dereci et al., 2017) calculated by multiplying SP scores: 0, <10%; 1, 10–25%; 2, 26–50%; 3, 51–75%; 4, 76–100%) and SI scores: 0, negative; 1, mild; 2, moderate; 3, severe).
Transmission Electron Microscopy (TEM) Examination
Vaginal tissue pieces were removed, fixed (3% glutaraldehyde and 1% osmium tetroxide), dehydrated in a graded ethanol series and embedded in a PON812 resin (SPI, West Chester, PA, United States). Tissues were. then stained with 5% uranyl acetate and lead citrate. Ultrastructural changes in the vaginal tissues were observed under a JEM 1230 TEM (JEOL Co., Hitachi Ltd., Tokyo, Japan) and photographed.
Desmosomes within the epithelium were identified and enumerated by two independent investigators according to a previously described method (Capocelli et al., 2015). Desmosomes were identified and quantified as discrete, linear hyper-densities along the outer cellular membrane that confer structural integrity to tissues. Additional morphological features of the epithelia were identified independently by two investigators.
Statistical Analysis
All data were analyzed using statistical analysis software SPSS 13.0. Data represent the mean ± standard deviation (n = 6 animals per group) of three independent experiments. Quantitative variables were tested for normal distribution and data were compared using a single-factor analysis of variance (ANOVA) using Windows software (SPSS Inc., Chicago, IL, United States). Fisher’s LSD test was used to evaluate differences between two groups. P < 0.05 was considered to indicate statistical significance.
Results
L. crispatus and L. delbrueckii Protect Against C. albicans in VVC Rats
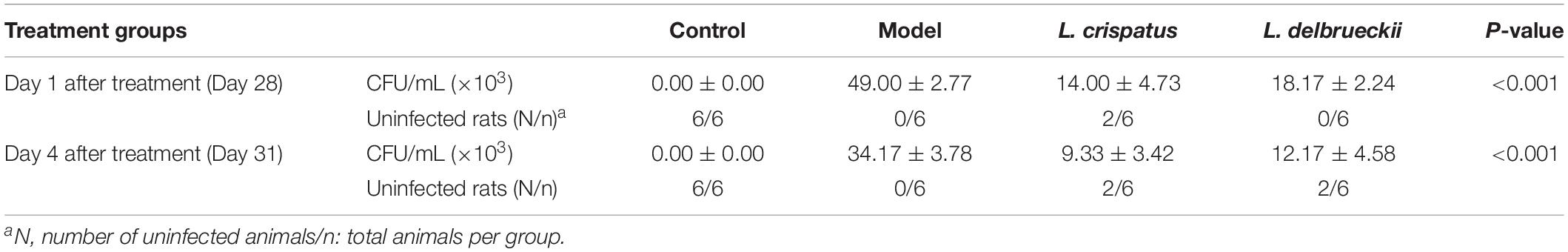
To compare the antifungal effects of L. crispatus and L. delbrueckii, rats were infected intravaginally with the mucosal C. albicans. At 1 and 4 days after the last dose, samples were obtained from all animals, and assessed by Gram staining. The results showed that vaginal administration of L. crispatus or L. delbrueckii exerted similarly, weak anti-C. albicans vaginitis effects. Among the untreated Model rats that remained infected, all vaginal swabs tested positive for C. albicans, with the epithelial cells being penetrated by the hypha (Day 31, Figure 2A). However, two of the six rats with positive vaginal swabs were Gram stain-negative after receiving intravaginal L. crispatus or L. delbrueckii (Day 31), suggesting less than half of the infected rats were cured. The negative pathogen conversion rate of L. crispatus at 1 (Day 28) and 4 days (Day 31) after treatment reached 33.3% (2/6), and 33.3% (2/6), respectively, and there were no significant differences compared with the negative pathogen conversion rate of the corresponding L. delbrueckii groups [0% (0/6) at 1 day (Day 28) and 33.3% at 4 days (Day 31) after treatment; P > 0.05; Figures 2B–D].
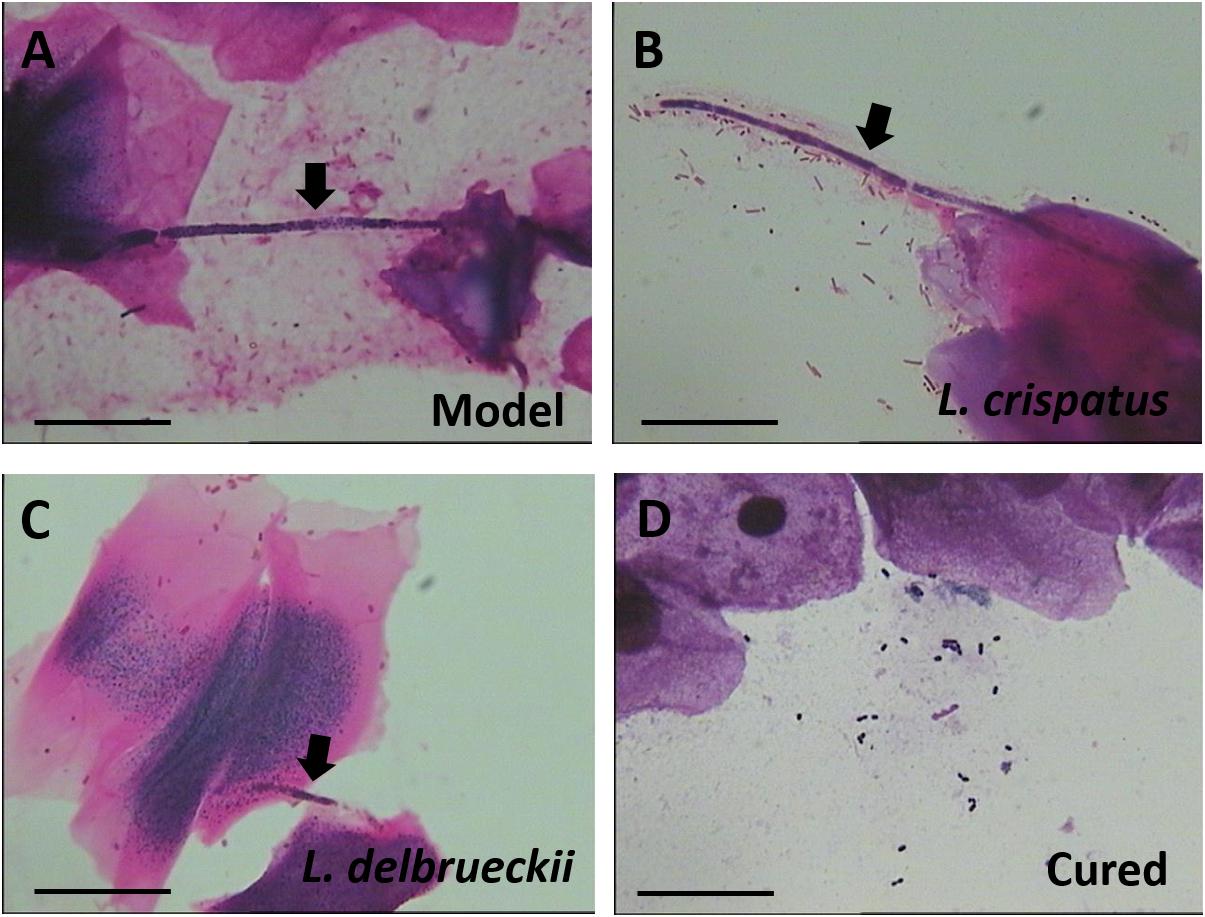
Figure 2. Gram staining of a vaginal swab from Model (A) and Lactobacilli-treated rats (B–D). Candida albicans hypha (black arrow) adhering to vaginal epithelial cells. The probiotic Lactobacilli surround and inhibit the growth of C. albicans hyphae. Scale bar = 20 μm, magnification × 1,000. Model (without treatment); L. crispatus treated (3.0 × 108 CFU for 7 days, Gram stain-positive); L. delbrueckii treated (3.0 × 108 CFU for 7 days, Gram stain-positive); and Cured (3.0 × 108 CFU of L. crispatus for 7 days, Gram stain-negative).
Next, we assessed the antifungal effects of intravaginal L. crispatus or L. delbrueckii on the fungal burden by quantifying the colony formation (Figure 3). At baseline, Control rats were free of C. albicans. The mean CFU/mL for C. albicans at Day 28 was 49.00 ± 2.77 × 103 in the Model group at Day 28, and was followed by a slow decline in vaginal Candida burden to 34.17 ± 3.78 × 103 at Day 31. One day after the treatment (Day 28), the mean CFUs in the L. crispatus (14.00 ± 4.73 × 103) and L. delbrueckii (18.17 ± 2.24 × 103) groups were significantly lower than those in the Model group (Figure 3A and Table 1; P < 0.001). Four days after the treatment (Day 31), the mean CFUs in the L. crispatus (9.33 ± 3.42 × 103), and L. delbrueckii (12.17 ± 4.58 × 103) groups were significantly lower than those the Model group (Figure 3B and Table 1; P < 0.001). There were no significant differences in the mean CFUs in the L. crispatus, and L. delbrueckii groups at one (Day 28) or 4 day (Day 31) after the treatment (P > 0.05).
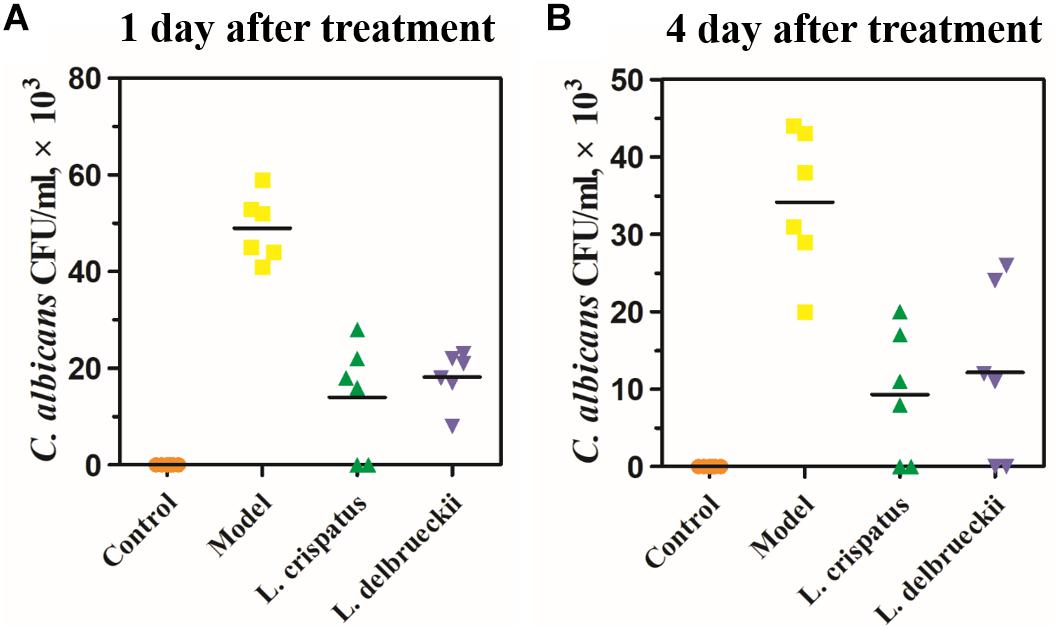
Figure 3. The number of colony-forming units (CFU)/mL of samples of vaginal tissue obtained from rats (n = 6 rats/group) at 1 day (A) and 4 days (B) after treatment and incubated on Sabouraud dextrose agar. Uninfected female Sprague-Dawley rats were used as controls. Model rats were infected with 1 × 108 CFU/ml C. albicans. Control (uninfected); Model: infected with 1 × 108 CFU/ml C. albicans;L. crispatus treated: infected and treated with L. crispatus 3.0 × 108 CFU for 7 days; L. delbrueckiitreated: infected and treated with L. delbrueckii 3.0 × 108 CFU for 7 days.
Morphometric Analysis
We performed HE staining and TEM analyses of anatomical and ultrastructural changes to evaluate the probiotic properties of the two Lactobacillus strains. The epithelium and mucosa in uninfected Control animals appeared normal, with no signs of inflammation, and a thick layer of stratum corneum was observed in the vaginal canal. All Model animals presented abnormal vaginal discharge and tissue swelling, as well as severe inflammation of the epithelium following Candida infection. HE staining revealed serious damage to the vaginal mucosa, with epithelial cell exfoliation and inflammatory cell infiltration, following infection with C. albicans (Figure 4A). Compared to the Model animals mucosa, the animals treated with L. crispatus or L. delbrueckii for 7 days showed a significant alleviation of inflammation and damage to the vaginal epithelial mucosa. In addition, an almost complete restoration of the mucosa was observed although some inflammatory cells persisted on the surface of the mucosa and in the lamina propria (Figures 4B,C). Several damaged tissues were observed with a complete restoration of a healthy vaginal mucosa (Figure 4D).
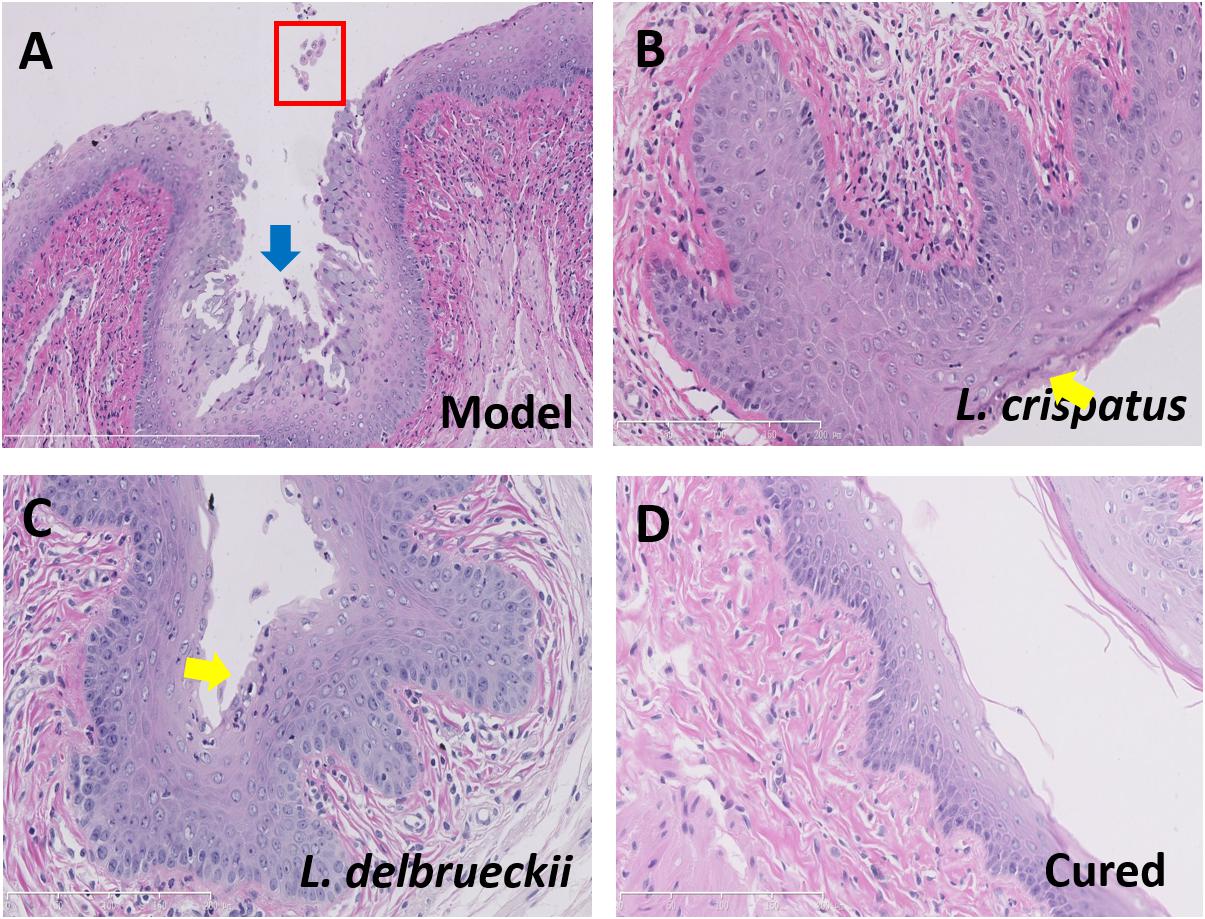
Figure 4. Histology of vaginal tissue sections from Model (A) and Lactobacilli-treated rats (B–D), stained with hematoxylin-eosin. Damaged vaginal epithelium (blue arrow), exfoliated vaginal epithelial cells (red box), and neutrophils (yellow arrow) infiltrating the vaginal epithelium are shown. Scale bar = 50 μm. Model (without treatment);L. crispatus-treated (3.0 × 108 CFU for 7 days, with neutrophil infiltration); L. delbrueckii-treated (3.0 × 108 CFU for 7 days, with neutrophil infiltration); and Cured (3.0 × 108 CFU of L. crispatus for 7 day, without neutrophil infiltration).
L. crispatus and L. delbrueckii Promote the Integrity of the Ultrastructural Morphology of the Vaginal Mucosal Barrier
Under TEM observation, the normal vaginal mucosal barrier consists of tightly packed stratified squamous non-keratinized epithelial cells (i.e., desmosomes, as rigid plaques that maintain tissue integrity and intercellular adhesion) with numerous intact finger-like and/or bubble-like pseudopods or microvilli on their surface (Figures 5A,B). The usual structure of desmosomes is clearly identified by the presence of outer and inner dense plaques on the cytoplasmic side of the plasma membrane. After C. albicans infection, neutrophils and spores infiltrating the vaginal epithelium and lamina propria were observed. In accordance with a previous study (Harmon and Green, 2013), histological evaluation showed that the intracellular spaces were enlarged, desmosomal junctions were significantly impaired and blurred, and cytoplasmic vacuolization was present in non-keratinocytes and intercellular disjunctions (Figures 5C,D). The number of desmosomes per cell was significantly reduced in epithelia after challenge compared with that in normal epithelia (43.67 ± 8.43 vs. 12.33 ± 4.32, P < 0.0001). Intracellularly, mitochondria exhibited swelling, as well as crest deformation and dissolution. Furthermore, compared with the Model group, we observed a marked ultrastructural improvement in the vaginal epithelium following treatment with either of the Lactobacillus strains, with significant reductions in the numbers of adhesive yeast, desmosome-like junctions and hyphal forms. The morphology of the mitochondria was normal or showed only slight swelling in the L. crispatus (Figures 5E,F) or L. delbrueckii groups (Figures 5G,H) when compared with that in the Model groups. The number of desmosomes in the L. crispatus group was significantly increased compared with that in the Model group (34.67 ± 7.61, P < 0.0001; Figure 6). Moreover, the number of desmosomes in the L. delbrueckii group (37.83 ± 6.97) was almost the same as that observed in normal epithelia (Figure 6; P = 0.443). These observations indicated that Lactobacillus induced a partial restoration of the mucosa.
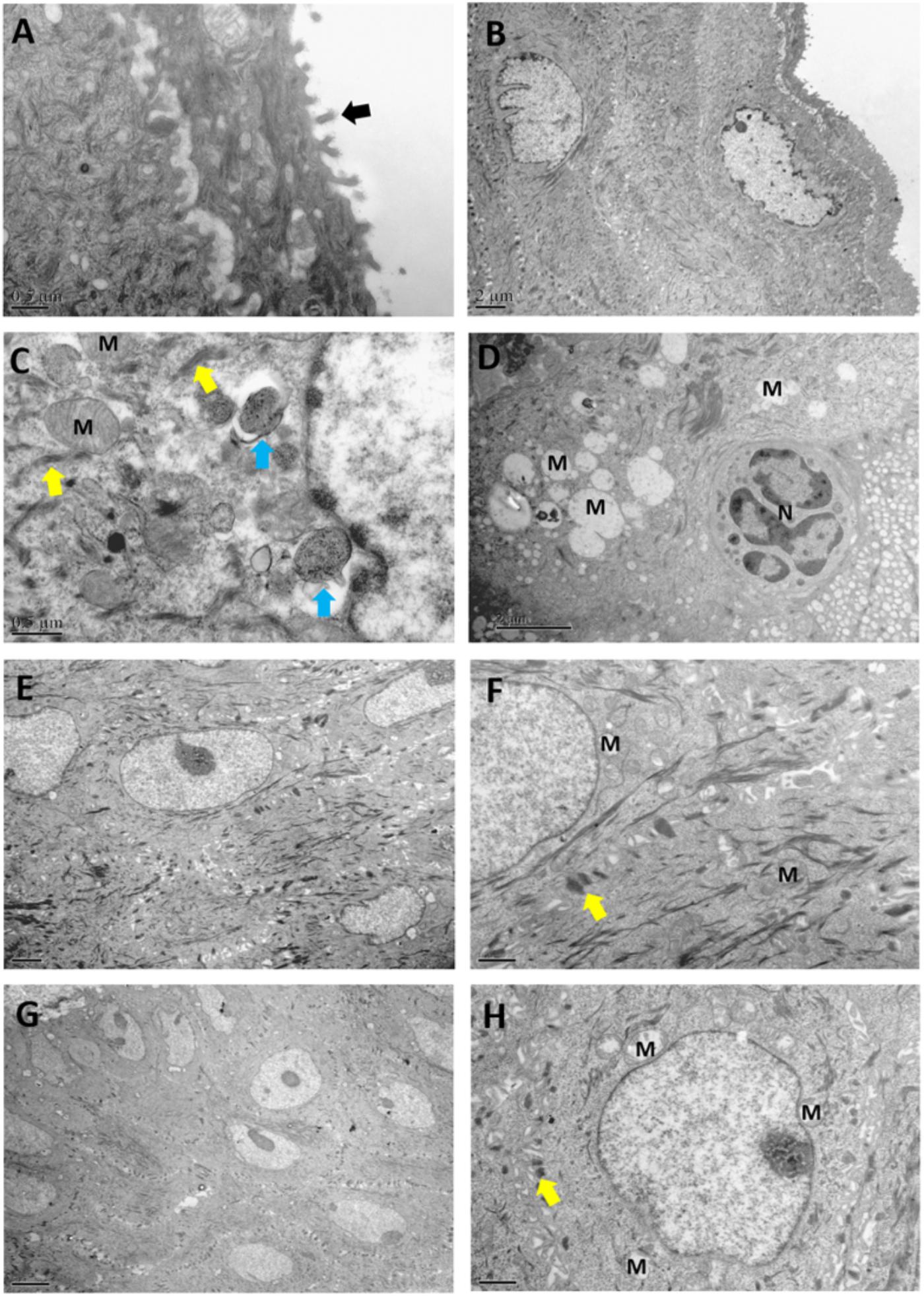
Figure 5. Ultrastructural changes in the vaginal mucosa of rats observed by transmission electron microscopy. Control, uninfected (A,B); Model, infected with 1 × 108 CFU/ml C. albicans (C,D); L. crispatus treated, infected and treated with L. crispatus 3.0 × 108 CFU for 7 days (E,F); L. delbrueckii treated, infected and treated with L. delbrueckii 3.0 × 108 CFU for 7 days (G,H). Rats in the Control group show normal vaginal morphology with a non-keratinized stratified squamous epithelium and intact microvilli (black arrow) extended by vaginal epithelial cells. Yeast cells (blue arrow) invading the superficial layers of the vaginal mucosa, with injured desmosome-like junctions (yellow arrow) and swollen mitochondria (M) after infection with 1 × 108 CFU/ml C. albicans.
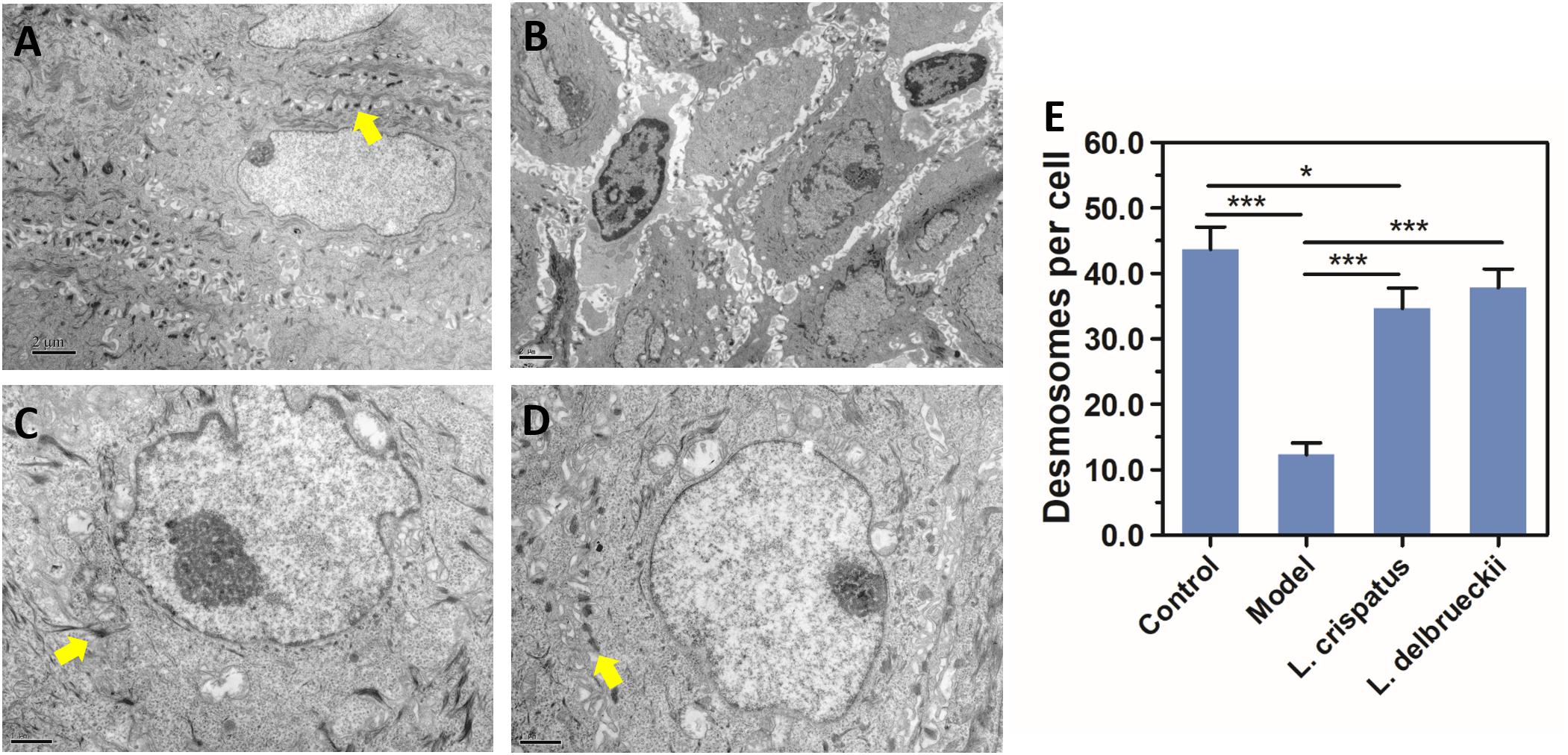
Figure 6. Desmosome quantification by TEM. Ultrastructure of desmosomes in the vaginal epithelium of rats observed by TEM. Control, uninfected (A); Model, infected with 1 × 108 CFU/ml C. albicans (B);L. crispatus treated, infected and treated with L. crispatus 3.0 × 108 CFU for 7 days (C); L. delbrueckiitreated, infected and treated with L. delbrueckii 3.0 × 108 CFU for 7 days (D). The desmosome-like junctions (yellow arrow) are significantly diminished, and the space between adjoining epithelial cells is widened in Model rats. Number of desmosomes per cell (E) in normal Control, Model, L. crispatus treated and L. delbrueckiitreated rats. Data represent the mean ± standard deviation (n = 6). *P < 0.05; **P < 0.01; ***P < 0.001.
L. crispatus and L. delbrueckii Modulate Cytokine Release by Vaginal Epithelial Tissue
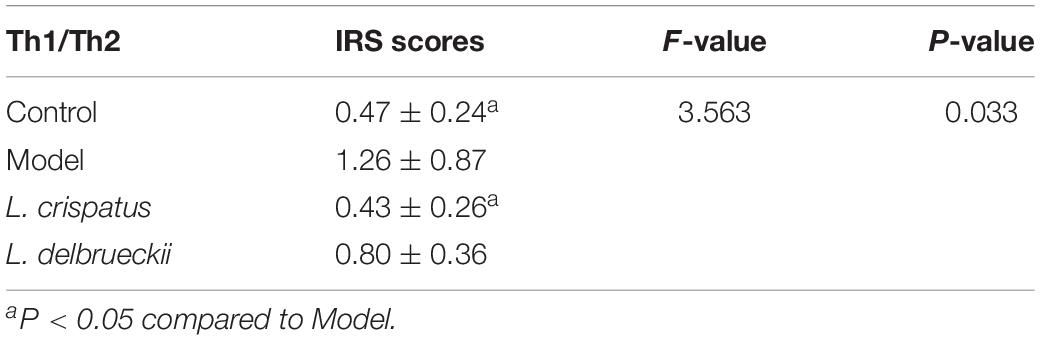
Our immunohistochemical studies revealed increased expression of IFN-γ (IRS: 4.17 ± 2.57 vs. 8.17 ± 2.57) and IL-17 (IRS: 4.67 ± 2.07 vs. 6.33 ± 2.88), indicating stimulation of Th 1 and Th 17-type immune responses in the vaginal epithelium following C. albicans infection. However, there were no significant differences in the IRS scores for IFN-γ (P = 0.055, Figure 7A), IL-4 (P = 0.082, Figure 7B), and IL-17 (P = 0.017, Figure 7C) among the groups. The IFN-γ/IL-4 ratio was calculated to reflect the Th1/Th2 balance (Table 2). When challenged with C. albicans, the IFN-γ/IL-4 ratio was significantly higher than that of the untreated animals (0.47 ± 0.24 vs. 1.26 ± 0.87, P = 0.012). In contrast, the ratio was significantly lower in the L. crispatus group following in vivo challenge (0.43 ± 0.26, P = 0.009) compared than that in the L. delbrueckii group (0.80 ± 0.36, P = 0.219), for which the ratio was similar to that of the normal tissue (P > 0.05).
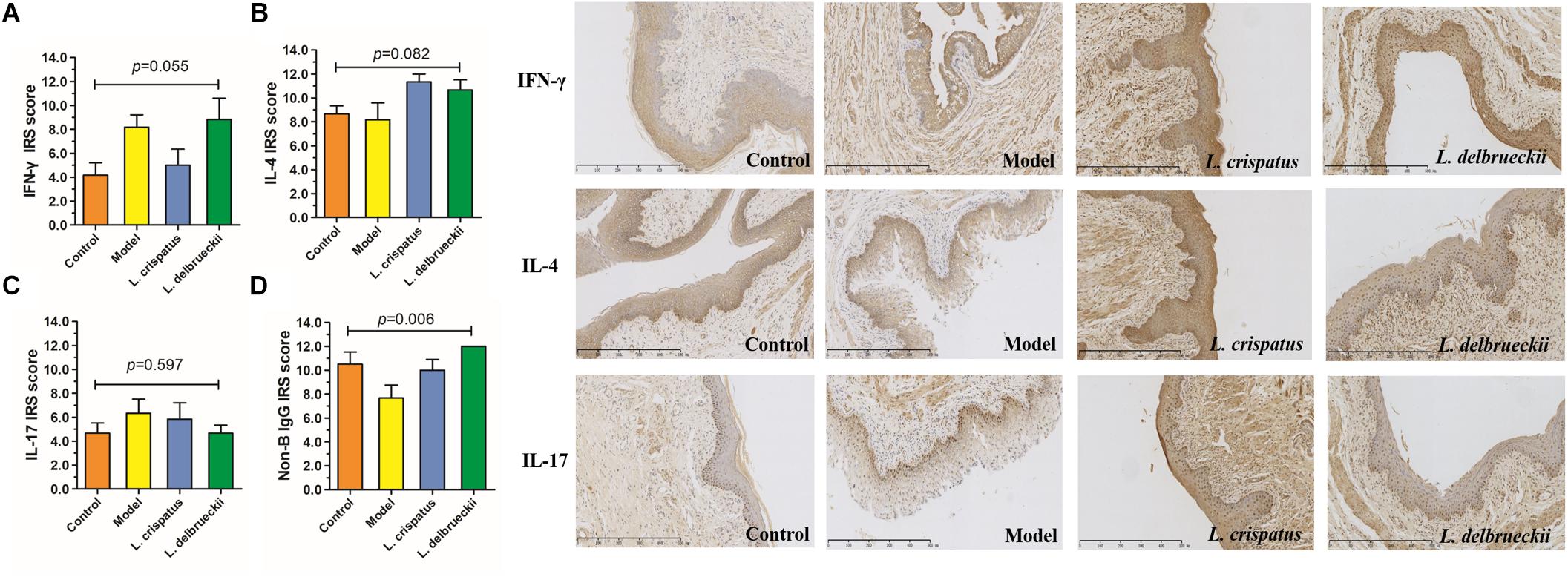
Figure 7. The expression of IFN-γ, IL-4 and IL-17 in vaginal tissues. Semi-quantitative analysis by immunoreactivity score (IRS) of IFN-γ, IL- 4 and IL-17 detected by immunohistochemistry. Control, uninfected (A); Model, infected with 1 × 108 CFU/ml C. albicans (B); L. crispatus treated, infected and treated with L. crispatus 3.0 × 108 CFU for 7 days (C); L. delbrueckii treated, infected and treated with L. delbrueckii 3.0 × 108 CFU for 7 days (D). Data represent the mean ± standard deviation (n = 6). Scale bar = 50 μm.
Although the vaginal mucosa acts a barrier against pathogenic invasion, there is no direct evidence that vaginally expressed Ig molecules contribute to antimicrobial activity. The monoclonal antibody RP215 binds specifically to a unique glycosylated epitope on non-B cell, epithelial-derived IgG. Strong R215-positive staining was detected in the cytoplasm of normal vaginal epithelial cells, while staining in the submucosa tissue was weak. There were significant differences in the IRS scores for epithelial-derived IgG among the groups (P = 0.006, Figure 7D). The IRS score for epithelial IgG secreted by the vaginal mucosa was 10.50 ± 2.51, which dropped sharply following infection (6.67 ± 3.27; P = 0.010). However, when treated with L. crispatus, the score was significantly increased (10.00 ± 2.19, P = 0.022) with a similar pattern observed in the L. delbrueckii group (12.00 ± 0.00, P = 0.153), which reached the baseline values (P > 0.05).
Discussion
Lactobacilli are Gram-positive rod-shaped, lactic acid producing bacteria which are mostly obligate and facultative anaerobes and predominantly found in the human genitourinary tract (Gupta et al., 1998). The Lactobacilli present in the vagina are important in preventing vaginal infections, such as BV and VVC, and invasion by pathogenic microorganisms, including human papillomavirus, and human immunodeficiency virus (Reid and Bocking, 2003). The currently available antimicrobial treatments for vaginal infections are associated with incomplete effectiveness leading to recurrent infections, drug resistance, and side-effects; therefore, alternative drugs are urgently required. In recent years, the use of Lactobacilli-derived probiotic bacteria has emerged as a novel strategy for the management of vaginal infections. However, investigations of the antifungal activities of probiotic strains are less common than investigations of their antibacterial activities (Chew et al., 2015). We have previously demonstrated that probiotic Lactobacilli can attenuate the virulence of C. albicans by modulating the secretion of cytokines and chemokines and enhancing the innate immune response in vitro (Niu et al., 2017). The functional characteristics of probiotics should be established based on both in vitro and in vivo studies. In the present study, intravaginal delivery of Lactobacillus strains for 7 days inhibited the in vivo growth of Candida species by between 60 and 70%. Thus, this animal model provides clear evidence of the fungicidal or candidacidal effects of the two probiotic Lactobacillus strains. Lopes et al. (2017) reported that L. delbrueckii showed no antimicrobial activity against pathogens, mainly due to the absence of organic acid production. However, in the present study, the antifungal effect of L. delbrueckii was similar to that of L. crispatus, which is believed to contribute to the control of vaginal microbiota by competing with other microflora for colonization of the vaginal epithelial cells (McLean and Rosenstein, 2000), as well as liberating H2O2 and other substances that mediate intracellular antifungal activity (Strus et al., 2006). These findings indicate the potential of these two probiotic Lactobacillus strains as an adjuvant to the mainstay in clinical treatment of VVC. However, the antifungal activity of probiotic Lactobacillus strains is very complex and requires further investigation (Ogunshe et al., 2011).
Maintaining the vaginal epithelial barrier is crucial as it maintains the balance between commensal microorganisms and the host. One of the potential health benefits of probiotics is the suppression of inflammatory responses (Isolauri et al., 2002). In our present study, upregulation of proinflammatory cytokines by C. albicans infection suggested T cell proliferation (IFN-γ and IL-17) and T cell activation (Bäuerl et al., 2013), while Lactobacillus treatment induced a partial reduction in the Th1/Th2 ratio. These observations suggest that both strains counteract the molecular events leading to T cell activation, as has been shown previously for the probiotic strains L. paracasei (Bäuerl et al., 2013), L. plantarum (Bäuerl et al., 2013), and L. casei (Carol et al., 2006). The two Lactobacillus strains used in this study, and particularly L. crispatus, were able to alter the cytokine profiles of vaginal epithelial cells, thus redirecting the pattern of immunity to a regulatory or tolerant mode that balances the Th1/Th2 ratio, and suppresses the proinflammatory cytokine production (e.g., IL17 and IFN-γ), although this difference was not statistically significant, which is in accordance with previous reports (Wasilewska et al., 2019). Decreased production of IFN-γ, a typical Th1 cytokine, could be a very interesting probiotic feature (Bäuerl et al., 2013). Although crucial for innate and adaptive immunity against intracellular pathogens, a prolonged distinct Th1-bias with a persistently high Th1/Th2 ratio leads to cell-mediated cytotoxicity, and inflammatory responses (Barousse et al., 2004; Ouyang et al., 2008). Taken together, these findings further support the hypothesis that Lactobacilli play an important role in maintaining the immune homeostasis by regulating cytokine secretion by epithelial cells (Bäuerl et al., 2013), and in this respect, L. crispatus treatment was more effective than L. delbrueckii. The sensing of, and signaling induced by, Lactobacilli require further investigation. Lactobacillus-derived proteins (Yan and Polk, 2012) and lactic acid induced acidification may be immunomodulatory functions that suppress the IFN-γ response of immune cells related to the Janus kinase/STAT signaling pathway and inhibit of the production of cytokines that mediate innate immunity (Fischer et al., 2007).
Previous studies have demonstrated that functional epithelial-derived IgG (RP215 positive) is secreted by vaginal epithelial cells in vitro. This finding challenges the classical concept that B cells are the only source of immunoglobulins (Li et al., 2016). These immunoglobulins appear to participate in skin innate immunity (Jiang et al., 2015) and may serve as natural antibodies (apparently the most controversial branch of humoral immunity), which not only recognize and neutralize antigenic microbial products (Amabebe and Anumba, 2018), but also provide long-term immunological memory that improves standard azole therapy efficacy and prevents relapse (Khasbiullina and Bovin, 2015). Our animal study confirmed our hypothesis that non-B IgG is expressed in the vaginal epithelium, a response that appears to be diminished by mycotic infections, but can be restored by Lactobacillus treatment. Further studies are required to ascertain the molecular mechanism by which Lactobacilli participate in the mucosal immunity in the vagina related to epithelial-derived IgG.
Another potential health benefit of Lactobacillus treatment is provided by the enhanced epithelial cell survival and barrier function (Yan and Polk, 2012; Yan et al., 2017). Ultrastructural analysis by electron microscopy verified several key features of in vivo vaginal epithelial tissue, such as the presence of desmosomes and mitochondria. In epithelial tissues, cells are anchored to one another via junctional complexes e.g., desmosomes, to maintain tissue integrity, and protect against the external environment (Green and Gaudry, 2000). Infection with pathogenic C. albicans not only significantly stimulates inflammatory cells, but also induces significant ultrastructural changes in the host cells, such as inhibited desmosome assembly, eventually leading to reduced adhesive strength (Kowalczyk and Green, 2013). Desmosomes, one of the predominant types of adhesive intercellular junctions in vertebrate tissues, are challenged by a variety of factors such as toxins, pathogens and inflammatory factors, potentially leading to tissue damage (Celentano and Cirillo, 2017). The damage to the epithelia, which could be the consequence of the inflammatory process itself, is related to disruption of this barrier, and passage of pathogenic microorganisms or molecules, an effect that is positively affected by probiotics, which stabilize the barrier function. There are several reports related to the effects of probiotic Lactobacilli on the “recovery” of the vaginal mucosa.
Lactobacilli have been reported to decrease damage to the DNA of epithelial cells (Yeh et al., 2007), reduce epithelial permeability, and attenuate the inflammatory response (Ahrne and Hagslatt, 2011). Lactobacilli have also been reported to prevent the attachment of the pathogenic microorganism required to exert its effects on paracellular permeability through upregulating production of MUC3 mucin (Ahrne and Hagslatt, 2011) and ameliorate epithelial apoptosis by producing Lactobacillus-derived soluble proteins in an EGF receptor-dependent manner (Yan and Polk, 2012). Mitochondria are influenced by a multitude of vital signals involved in the regulation of energy metabolism and cellular survival. These signals promote or prevent cell survival by modulating mitochondrial function and structure (Sedlackova and Korolchuk, 2018). Fungal infections directly or indirectly impair mitochondrial ultrastructure, ultimately leading to destruction of the vaginal epithelium. However, in this study, we observed significant improvements in the ultrastructure of the vaginal epithelium and host cell mitochondria following the administration of Lactobacilli. Lactobacilli exert protective anti-oxidant effects by preventing mitochondrial-generated reactive oxygen species that induce membrane damage (Barbonetti et al., 2013).
This study provides evidence implicating Lactobacilli as a potential adjunct biotherapeutic agent in women with VVC, especially for those with drug resistance, adverse effects or contraindications when using antifungal agents. The effects of probiotic L. crispatus and L. delbrueckii in treating VVC are particularly noticeable in relation to changes to the vaginal mucosa that are microbiological, physiological and morphological in nature. In terms of antifungal, anti-inflammatory effects and mucosal repair, the effects of L. crispatus are similar to those of the commercial L. delbrueckii formulation. The promising results of this study highlighting the potential of Lactobacillus treatment of VVC in clinical practice require validation in well-designed clinical studies with larger sample sizes and long-term follow-up.
Ethics Statement
All animal experiments were performed according to the National Institutes of Health Guide for the care and Use of Laboratory Animals. The protocol was approved by Peking University First Hospital Ethics Committee (Permit No. #J201634). All animals were housed in the Animal Center Laboratory of Peking University First Hospital.
Author Contributions
ZL contributed to the main experimental conception and design. TL and XC performed the experiments. TL, XZ, and SW analyzed the data and contributed to reagents. TL wrote the manuscript. All authors approved the final version of the manuscript.
Funding
This work was supported by a grant from the National Natural Science Foundation of China (Grant Nos. 81771530 and 81801406) and Beijing Municipal Administration of Hospitals’ Youth Programme (Grant No. QML20181402).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors are grateful to Prof. Xiaoyan Qiu from Peking University Center for Human Disease Genomics for her kind donation of RP215 to support this study.
Abbreviations
ANOVA, one-way analysis of variance; ATCC, American type culture collection; BV, bacterial vaginosis; C. albicans, Candida albicans; CFU, colony-forming units; HE, hematoxylin and eosin; IFN, interferon; IL, interleukin; IRS, the immunoreactivity score; L. crispatus, Lactobacillus crispatus; LSD, least significant difference; MRS, de Man, Rogosa and Sharpe; SDA, sabouraud dextrose agar; SI, staining intensity; SP, staining percentage; TEM, transmission electron microscopy; Th, T helper; VVC, vulvovaginal candidiasis.
References
Ahrne, S., and Hagslatt, M. L. J. (2011). Effect of lactobacilli on paracellular permeability in the gut. Nutrients 3, 104–117. doi: 10.3390/nu3010104
Amabebe, E., and Anumba, D. O. C. (2018). The vaginal microenvironment: the physiologic role of Lactobacilli. Front. Med. 5:181. doi: 10.3389/fmed.2018.00181
Anton, L., Sierra, L. J., DeVine, A., Barila, G., Heiser, L., Brown, A. G., et al. (2018). Common cervicovaginal microbial supernatants alter cervical epithelial function: mechanisms by which Lactobacillus crispatus contributes to cervical health. Front. Microbiol. 9:2181. doi: 10.3389/fmicb.2018.02181
Banerjee, P., Merkel, G. J., and Bhunia, A. K. (2009). Lactobacillus delbrueckii ssp. bulgaricus B-30892 can inhibit cytotoxic effects and adhesion of pathogenic Clostridium difficile to Caco-2 cells. Gut. Pathog. 1:8. doi: 10.1186/1757-4749-1-8
Barbonetti, A., Vassallo, M. R., Cinque, B., Filipponi, S., Mastromarino, P., Cifone, M. G., et al. (2013). Soluble products of Escherichia coli induce mitochondrial dysfunction-related sperm membrane lipid peroxidation which is prevented by lactobacilli. PLoS. One 8:e83136. doi: 10.1371/journal.pone.0083136
Barousse, M. M., Van Der Pol, B. J., Fortenberry, D., Orr, D., and Fidel, P. L. Jr. (2004). Vaginal yeast colonisation, prevalence of vaginitis, and associated local immunity in adolescents. Sex. Transm. Infect. 80, 48–53.
Bäuerl, C., Llopis, M., Antolín, M., Monedero, V., Mata, M., Zúñiga, M., et al. (2013). Lactobacillus paracasei and Lactobacillus plantarum strains downregulate proinflammatory genes in an ex vivo system of cultured human colonic mucosa. Genes Nutr. 8, 165–180. doi: 10.1007/s12263-012-0301-y
Buggio, L., Somigliana, E., Borghi, A., and Vercellini, P. (2019). Probiotics and vaginal microecology: fact or fancy? BMC. Womens Health 19:25. doi: 10.1186/s12905-019-0723-4
Cadieux, P., Burton, J., Gardiner, G., Braunstein, I., Bruce, A. W., Kang, C. Y., et al. (2002). Lactobacillus strains and vaginal ecology. JAMA 287, 1940–1941.
Capocelli, K. E., Fernando, S. D., Menard-Katcher, C., Furuta, G. T., Masterson, J. C., and Wartchow, E. P. (2015). Ultrastructural features of eosinophilic oesophagitis: impact of treatment on desmosomes. J. Clin. Pathol. 68, 51–56. doi: 10.1136/jclinpath-2014-202586
Carol, M., Borruel, N., Antolin, M., Llopis, M., Casellas, F., Guarner, F., et al. (2006). Modulation of apoptosis in intestinal lymphocytes by a probiotic bacteria in Crohn’s disease. J. Leukoc. Biol. 79, 917–922. doi: 10.1189/jlb.0405188
Celentano, A., and Cirillo, N. (2017). Desmosomes in disease: a guide for clinicians. Oral. Dis. 23, 157–167. doi: 10.1111/odi.12527
Chew, S. Y., Cheah, Y. K., Seow, H. F., Sandai, D., and Than, L. T. (2015). Probiotic Lactobacillus rhamnosus GR-1 and Lactobacillus reuteri RC-14 exhibit strong antifungal effects against vulvovaginal candidiasis-causing Candida glabrata isolates. J. Appl. Microbiol. 118, 1180–1190. doi: 10.1111/jam.12772
Dereci, Ö., Akgün, Ş., Celasun, B., Öztürk, A., and Günhan, Ö (2017). Histological evaluation of the possible transformation of peripheral giant cell granuloma and peripheral ossifying fibroma: a preliminary study. Indian J. Pathol. Microbiol. 60, 15–20. doi: 10.4103/0377-4929.200032
Dovnik, A., Golle, A., Novak, D., Arko, D., and TakaČ, I. (2015). Treatment of vulvovaginal candidiasis: a review of the literature. Acta. Dermatovenerol. Alp. Pannonica Adriat. 24, 5–7.
Fischer, K., Hoffmann, P., Voelkl, S., Meidenbauer, N., Ammer, J., Edinger, M., et al. (2007). Inhibitory effect of tumor cell-derived lactic acid on human T cells. Blood 109, 3812–3819.
Fuller, R. (1999). “Probiotics for farm animals,” in Probiotics. A Critical Review, ed. Tannock, G. W. (Wymondham: Horizon Scientific Press), 15–22.
Green, K. J., and Gaudry, C. A. (2000). Are desmosomes more than tethers for intermediate filaments? Nat. Rev. Mol. Cell Biol. 1, 208–216.
Gupta, K., Stapleton, A. E., Hooton, T. M., Roberts, P. L., Fennell, C. L., and Stamm, W. E. (1998). Inverse association of H2O2-producing lactobacilli and vaginal Escherichia coli colonization in women with recurrent urinary tract infections. J. Infect. Dis. 178, 446–450. doi: 10.1086/515635
Harmon, R. M., and Green, K. J. (2013). Structural and functional diversity of desmosomes. Cell. Commun. Adhes. 20, 171–187. doi: 10.3109/15419061.2013.855204
Isolauri, E., Rautava, S., Kalliomäki, M., Kirjavainen, P., and Salminen, S. (2002). Probiotic research: learn from the evidence. Allergy 57, 1076–1077.
Jiang, D., Ge, J., Liao, Q., Ma, J., Liu, Y., Huang, J., et al. (2015). IgG and IgA with potential microbial-binding activity are expressed by normal human skin epidermal cells. Int. J. Mol. Sci. 16, 2574–2590. doi: 10.3390/ijms16022574
Khasbiullina, N. R., and Bovin, N. V. (2015). Hypotheses of the origin of natural antibodies: a glycobiologist’s opinion. Biochemistry 80, 820–835. doi: 10.1134/S0006297915070032
Kim, J. M., and Park, Y. J. (2017). Probiotics in the prevention and treatment of postmenopausal vaginal infections: review article. J. Menopausal. Med. 23, 139–145. doi: 10.6118/jmm.2017.23.3.139
Kowalczyk, A. P., and Green, K. J. (2013). Structure, function and regulation of desmosomes. Prog. Mol. Biol. Transl. Sci. 116, 95–118. doi: 10.1016/B978-0-12-394311-8.00005-4
Lamont, R. F. (2003). Infection in the prediction and antibiotics in the prevention of spontaneous preterm labour and preterm birth. BJOG 110, 71–75.
Laue, C., Papazova, E., Liesegang, A., Pannenbeckers, A., Arendarski, P., Linnerth, B., et al. (2018). Effect of a yoghurt drink containing Lactobacillus strains on bacterial vaginosis in women - a double-blind, randomised, controlled clinical pilot trial. Benef. Microb. 9, 35–50. doi: 10.3920/BM2017.0018
Li, T., Niu, X., Zhang, X., Wang, S., and Liu, Z. (2016). Baofukang suppository promotes the repair of vaginal epithelial cells in response to Candida albicans. AMB Exp. 6:109. doi: 10.1186/s13568-016-0281-1
Lopes, E. G., Moreira, D. A., Gullón, P., Gullón, B., Cardelle-Cobas, A., and Tavaria, F. K. (2017). Topical application of probiotics in skin: adhesion, antimicrobial and antibiofilm in vitro assays. J. Appl. Microbiol. 122, 450–461. doi: 10.1111/jam.13349
Mandl, A. M. (1951). The phases of the estrous cycle in the adult white rat. J. Exp. Biol. 28, 576–584.
McLean, N. W., and Rosenstein, I. J. (2000). Characterisation and selection of a Lactobacillus species to re-colonise the vagina of women with recurrent bacterial vaginosis. J. Med. Microbiol. 49, 543–552. doi: 10.1099/0022-1317-49-6-543
Melgaço, A. C. C., Blohem Pessoa, W. F., Freire, H. P., Evangelista de Almeida, M., Santos Barbosa, M., Passos Rezende, R. T., et al. (2018). Potential of maintaining a healthy vaginal environment by two Lactobacillus strains isolated from cocoa fermentation. Biomed. Res. Int. 2018:7571954. doi: 10.1155/2018/7571954
Niu, X. X., Li, T., Zhang, X., Wang, S. X., and Liu, Z. H. (2017). Lactobacillus crispatus modulates vaginal epithelial cell innate response to Candida albicans. Chin. Med. J. 130, 273–279. doi: 10.4103/0366-6999.198927
Ogunshe, A. A., Omotoso, M. A., and Bello, V. B. (2011). The in vitro antimicrobial activities of metabolites from Lactobacillus strains on Candida species implicated in Candida vaginitis. Malays. J. Med. Sci. 18, 13–25.
Ouyang, W., Chen, S., Liu, Z., Wu, Y., and Li, J. (2008). Local Th1/Th2 cytokine expression in experimental murine vaginal candidiasis. J. Huazhong. Univ. Sci. Technol. Med. Sci. 2008, 352–355.
Palacios, S., Espadaler, J., Fernández-Moya, J. M., Prieto, C., and Salas, N. (2016). Is it possible to prevent recurrent vulvovaginitis? The role of Lactobacillus plantarum I1001 (CECT7504). Eur. J. Clin. Microbiol. Infect. Dis. 35, 1701–1708.
Ravel, J., Gajer, P., Abdo, Z., Schneider, G. M., Koenig, S. S., McCulle, S. L., et al. (2011). Vaginal microbiome of reproductive-age women. Proc. Natl. Acad. Sci. U.S.A. 108, 4680–4687. doi: 10.1073/pnas.1002611107
Reid, G., and Bocking, A. (2003). The potential for probiotics to prevent bacterial vaginosis and preterm labor. Am. J. Obstet. Gynecol. 189, 1202–1208.
Santos, C. M. A., Pires, M. C. V., Leão, T. L., Silva, A. K. S., Miranda, L. S., Martins, F. S., et al. (2018). Anti-inflammatory effect of two Lactobacillus strains during infection with Gardnerella vaginalis and Candida albicans in a HeLa cell culture model. Microbiology 164, 349–358. doi: 10.1099/mic.0.000608
Sedlackova, L., and Korolchuk, V. I. (2018). Mitochondrial quality control as a key determinant of cell survival. Biochim. Biophys. Acta Mol. Cell. Res. 1866, 575–587. doi: 10.1016/j.bbamcr.2018.12.012
Sobel, J. D. (2007). Candidal vulvovaginitis. Lancet 369, 1961–1971. doi: 10.1016/S0140-6736(07)60917-9
Sobel, J. D., and Sobel, R. (2018). Current treatment options for vulvovaginal candidiasis caused by azole-resistant Candida species. Expert. Opin. Pharmacother. 19, 971–977. doi: 10.1080/14656566.2018.1476490
Song, J., Lang, F., Zhao, N., Guo, Y., and Zhang, H. (2018). Vaginal lactobacilli induce differentiation of monocytic precursors toward langerhans-like cells: in vitro evidence. Front. Immunol. 9:2437. doi: 10.3389/fimmu.2018.02437
Strus, M., Brzychczy-Włoch, M., Gosiewski, T., Kochan, P., and Heczko, P. B. (2006). The in vitro effect of hydrogen peroxide on vaginal microbial communities. FEMS Immunol. Med. Microbiol. 48, 56–63.
Strus, M., Brzychczy-Wloch, M., Kucharska, A., Gosiewski, T., and Heczko, P. B. (2005). Inhibitory activity of vaginal Lactobacillus bacteria on yeasts causing vulvovaginal candidiasis. Med. Dosw. Mikrobiol. 57, 7–17.
Wasilewska, E., Zlotkowska, D., and Wroblewska, B. (2019). Yogurt starter cultures of Streptococcus thermophilus and Lactobacillus bulgaricus ameliorate symptoms and modulate the immune response in a mouse model of dextran sulfate sodium-induced colitis. J. Dairy Sci. 102, 37–53. doi: 10.3168/jds.2018-14520
Witkin, S. S., Mendes-Soares, H., Linhares, I. M., Jayaram, A., Ledger, W. J., and Forney, L. J. (2013). Influence of vaginal bacteria and D-and L-lactic acid isomers on vaginal extracellular matrix metalloproteinase inducer: implications for protection against upper genital tract infections. mBio. 4:e460-13. doi: 10.1128/mBio.00460-13
World Health Organization, and Food and Agriculture Organization of the United Nations. (2006). Probiotics in Food: Health and Nutritional Properties and Guidelines for Evaluation. Rome, IT: World Health Organization, Food and Agriculture Organization of the United Nations.
Yan, F., Liu, L., Cao, H., Moore, D. J., Washington, M. K., Wang, B., et al. (2017). Neonatal colonization of mice with LGG promotes intestinal development and decreases susceptibility to colitis in adulthood. Mucosal. Immunol. 10, 117–127. doi: 10.1038/mi.2016.43
Yan, F., and Polk, D. B. (2012). Characterization of a probiotic-derived soluble protein which reveals a mechanism of preventive and treatment effects of probiotics on intestinal inflammatory diseases. Gut. Microb. 3, 25–28. doi: 10.4161/gmic.19245
Yeh, S. L., Lin, M. S., and Chen, H. L. (2007). Inhibitory effects of a soluble dietary fiber from Amorphophallus konjac on cytotoxicity and DNA damage induced by fecal water in Caco-2 cells. Planta. Med. 73, 1384–1388. doi: 10.1055/s-2007-990228
Keywords: Lactobacillus crispatus, Lactobacillus delbrueckii, vulvovaginal candidiasis, antifungal, microbiome
Citation: Li T, Liu Z, Zhang X, Chen X and Wang S (2019) Local Probiotic Lactobacillus crispatus and Lactobacillus delbrueckii Exhibit Strong Antifungal Effects Against Vulvovaginal Candidiasis in a Rat Model. Front. Microbiol. 10:1033. doi: 10.3389/fmicb.2019.01033
Received: 21 January 2019; Accepted: 24 April 2019;
Published: 08 May 2019.
Edited by:
Jack Wong, The Chinese University of Hong Kong, ChinaReviewed by:
Alex Galanis, Democritus University of Thrace, GreeceJose Antonio Reales-Calderon, Singapore Immunology Network (A*STAR), Singapore
Copyright © 2019 Li, Liu, Zhang, Chen and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhaohui Liu, MTczMDEyNTU0MjZAMTYzLmNvbQ==
 Ting Li
Ting Li Zhaohui Liu
Zhaohui Liu Xu Zhang
Xu Zhang Xi Chen3
Xi Chen3