- Department of Neurology, The First Affiliated Hospital, Sun Yat-Sen University, Guangzhou, China
Background: Gelsemium elegans (G. elegans) is a toxic plant indigenous to Southeast Asia. It is highly poisonous due to its strong respiratory depressive effect. However, G. elegans poisoning cases have not been summarized comprehensively and are rarely reported in English journals. Furthermore, none of the present reports present prognosis in detail.
Case presentation: A 26-year-old female was found comatose at home and brought to the hospital with deep coma, hypoxia, and acidosis. After mechanical ventilation for hours, the patient recovered from coma with sequelae of impaired short-term memory, disorientation, and childish behaviors. Brain magnetic resonance imaging (MRI) showed bilateral hippocampus and basal ganglia damage due to hypoxia. During 8 months of follow-up, both her symptoms and brain MRI scan improved significantly.
Conclusion: G. elegans is highly toxic. Although patients may die within 30 min due to its strong respiratory depressive effect, they can survive with timely respiratory support and enjoy gradual improvement without delayed postanoxic encephalopathy.
Background
Gelsemium elegans (G. elegans) is one of three species of Gelsemium, a genus of flowering plants in the Gelsemiaceae family. It is indigenous to Southeast Asia and can be specifically found in southeast China, India, Indonesia, Laos, Malaysia, north Myanmar, north Thailand, and Vietnam. G. elegans is an evergreen shrub, with long stems up to 12 m, and grows as a twining vine, interweaving with other surrounding vegetation. It may interweave with other edible plants or be mistaken for various lookalike therapeutic herbs, leading to inadvertent consumption and poisoning. G. elegans is highly poisonous due to its strong neurological and respiratory depressive effects (1). Oral administration of crude extracts of G. elegans at doses of 10, 15, 20, and 25 mg/kg caused death in 11, 50, 72, and 100%, respectively, of test mice (2). The high concentration of alkaloids appears to be responsible for the toxic effects of the plant. To date, a total of 121 alkaloids have been found in Gelsemium, and gelsenicine is the most toxic alkaloid in G. elegans (LD50 ~0.128 mg/kg mice, intraperitoneally; 0.26 mg/kg rat, intraperitoneally; and 0.15 mg/kg rat, intravenously), whereas koumine is the most abundant alkaloid and exhibits mild toxicity (LD50 ~100 mg/kg mice, intraperitoneally) (3). In contrast to an intensive study of G. elegans on phytochemistry, G. elegans poisoning cases have not been summarized comprehensively and are rarely reported in English journals. Furthermore, none of the present reports presents prognosis in detail. Here, we report a case of G. elegans poisoning.
Case Presentation
The patient, an educated 26-year-old Teochew woman, was found unconscious in her bedroom around midnight. She was immediately sent to the nearest emergency room. At arrival, she had a Glasgow coma scale score 3/15, respiratory rate 36 breaths/min, pupils 6 mm in diameter without light reaction, heart rate 112 beats/min, blood pressure 152/100 mmHg, and SpO2 36%. Intubation was carried out immediately, and she was placed on mechanical ventilation; half an hour later, arterial blood gas analysis revealed pH 7.22, PaO2 35 mmHg, PaCO2 57.5 mmHg, SO2 55%, lactate 5.0 mmol/l, HCO3 22.8 mmol/l, SB −3.8 mmol/l, and AB −5.1 mmol/l. She regained consciousness and was weaned from mechanical ventilation with normal arterial blood gas analysis results 6 h later. After the first aid, her vital signs were normal, and her condition did not fluctuate or deteriorate. She continued to be hospitalized at her local medical center for 11 days but failed to identify the cause of the coma. To find out the cause of the coma, she was sent to our hospital.
At arrival in our department, she presented with euphoria and childish behaviors. She was unable to recall how she sank into coma. Her past medical history was unremarkable. Physical examination was only notable for disorientation to time and place and impaired short-term memory. Results of routine blood, serum, urine, and stool tests and cerebrospinal fluid analysis were unremarkable. The Mini-Mental State Examination (MMSE) score was 23/30. Brain computed tomography scan was normal. Cerebral magnetic resonance imaging (MRI) showed abnormal signals from bilateral globus pallidus and asymmetry of the hippocampus larger on the left side (Figure 1). Magnetic resonance spectroscopy (MRS) showed that the values of NAA/(Cho + Cr) at bilateral hippocampus were less than normal in both sides but more prominent in the left.
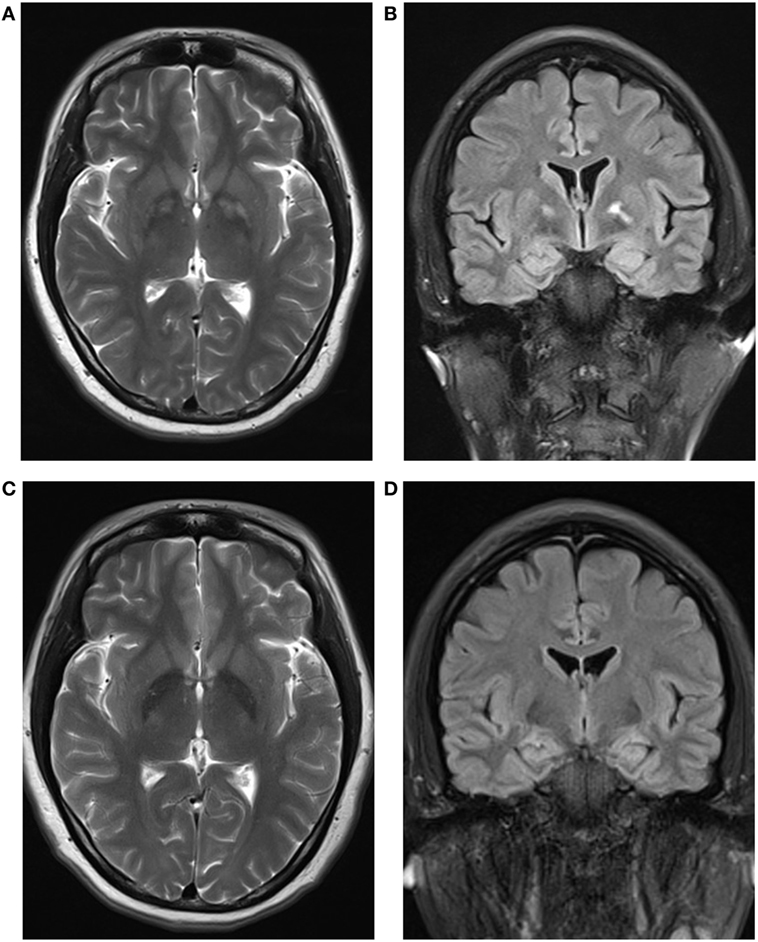
Figure 1. (A,C) Axial T2-weighted magnetic resonance imaging (MRI) slices showing symmetrical hyperintensity in the globus pallidus at 17 days (A) and 3.5 months (C) after poisoning, demonstrating a reduction in globus pallidus hyperintensity. (B,D) Coronal fluid-attenuated inversion recovery MRI slices at 17 days (B) showing asymmetry of the hippocampus larger on the left side and improved symmetry at 3.5 months (D) after poisoning.
History was taken in detail several times; finally, her husband recalled that there was a bottle of broth of herbs at her bedside table. The herbs looked like G. elegans. Samples of interest taken in the scene were then analyzed and toxic Gelsemium alkaloids were detected by the China National Analytical Center of the Chinese Academy of Sciences. Therefore, diagnosis of G. elegans poisoning was established.
Gradually, euphoria and childish behaviors wore off within 1 month. However, she presented with depressed mood and did not respond when asked whether she ingested the broth of G. elegans. She was discharged on the 36th day after onset. On the first follow-up, 3.5 months after intoxication, we evaluated her with Hamilton Depression Rating Scale (24 items), and the score was 23. During this evaluation, the patient informed us that she ingested the broth of G. elegans the evening of initial presentation, and she knew it was toxic. An antidepressant (escitalopram) was added. The MMSE score at that time was 26/30, with short-term memory improved, but the orientation ability to time and place was still impaired. MRI showed that the range of abnormal signals of bilateral globus pallidus was reduced (Figure 1). The symmetry of the hippocampus was also improved in the MRI scan (Figure 1), but MRS still showed NAA/(Cho + Cr) < 0.75. On the second follow-up, 8 months after intoxication, our patient was able to work, although she felt much more challenge than before. The score of Hamilton Depression Rating Scale (24 items) declined to 19. The MMSE score was 28/30, with mild impairment of orientation ability to time and place. We added the Montreal Cognitive Assessment this time, which detected short-term memory impairment and disorientation more sensitively of our patient, and the score was 25/30. MRI and MRS were quite similar to that of the first follow-up.
Discussion
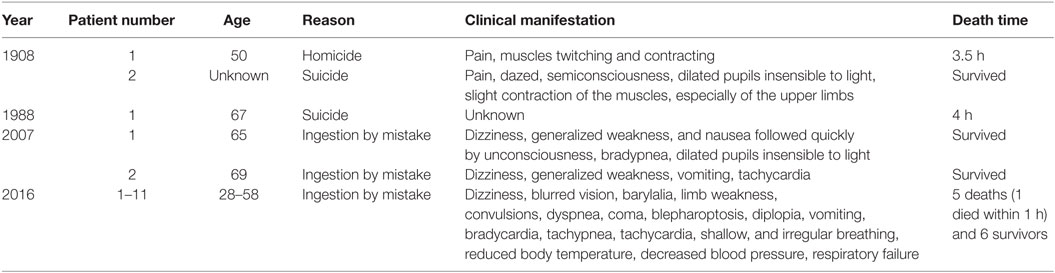
Reports of G. elegans poisoning in English are rare. To our knowledge, there are only four papers that involved the subject of G. elegans poisoning (summarized in Table 1) (1, 4–6). However, using the Chinese name of G. elegans as the search keyword, we found another 28 papers (7–34) that reported G. elegans poisoning cases in Wanfang Data, a major Chinese academic paper database of China. Most of these cases are from collective food poisoning accidents due to mistaking G. elegans as non-toxic herbs (7, 8, 11–13, 15, 16, 22, 23, 25–29). The most common reason for G. elegans poisoning is mistaken ingestion, as its morphology is similar to some non-toxic Chinese medicinal herbs (1, 6). The second most common reason is suicide (10, 12, 14, 17, 18). Some other rare causes include homicide (19) and using G. elegans to treat disease without caution (14, 30).
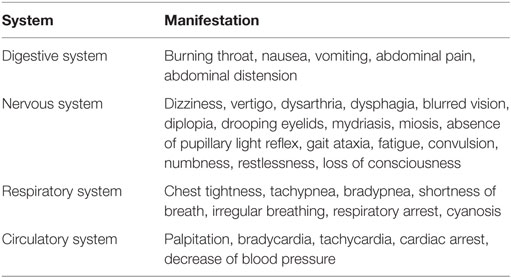
The clinical features of G. elegans poisoning can be summarized as follows. First, the incubation period is very short, with onset of symptoms mostly occurring within 20 min after ingestion (27). The shortest onset of symptoms after ingestion recorded is 5 min (28) and the longest is 2 h (29). In addition, the external application of G. elegans to treat skin disease can also be harmful, with an incubation period of less than 1 h (30). Second, in most cases, the initial manifestations of G. elegans poisoning are gastrointestinal abnormalities followed by neurological abnormalities and respiratory depression. However, it is indeed multifarious (Table 2 lists the common manifestations being mentioned in the literature) and may vary according to the dose of ingestion. In those who ingest higher doses, neurological abnormalities and respiratory failure will be more prominent and earlier to present (14) and even cover up gastrointestinal manifestations. Third, among the multifarious clinical manifestations of G. elegans poisoning, respiratory depression is the most prominent and the main cause of death. Patients may die within 30 min (13) to 2.5 h (27) due to respiratory failure. Although the chemical composition of G. elegans is complex, the alkaloids appear to be responsible for the toxic effects of the plant (3). Animal studies demonstrate that many alkaloids possess respiratory depressive effects (2, 35), such as gelsemicine, sempervirine, koumicine, koumine, kouminicine, and kounidine. Fourth, G. elegans poisoning can also lead to the dysfunction of the heart, kidneys, and/or liver, but they are uncommon. Long QT syndrome, torsade de pointes, and third-degree atrioventricular block are reported (9, 14, 16, 21, 26, 32). The overall incidence of arrhythmia is less than 10%. Given the fact that cardiovascular disorders appear after respiratory arrest, it seems the alkaloids of G. elegans affect the cardiovascular system at a lesser extent, which differs from aconitine or daturine intoxication (16). It is also proposed that G. elegans ingestion may lead to direct renal injury, manifesting as oliguria, anuria, and progressive elevation of blood urea nitrogen and creatinine on the second or third day of intoxication (28). Hepatic function may be affected as well: for about 14% patients, jaundice and/or elevation of alanine transaminase and/or aspartate transaminase may occur several days after intoxication (28).
The diagnostic process for G. elegans intoxication may be time consuming and could likely involve forensic investigation (10). Gelsemium alkaloids can be detected in the urine, suggesting that urinary gelsemine is a practical marker of Gelsemium exposure in human subjects (36). Because of the relatively short half-life of Gelsemium alkaloids (37), urine specimens need to be collected in a timely manner.
Gastric lavage, activated charcoal, and cathartics are all used for gastrointestinal decontamination in the treatment of G. elegans poisoning. The large majority of the reported literature considers gastrointestinal decontamination as the main focus of treatment (12, 17, 19, 21, 33). However, more recent studies have pointed out that there is an increased risk of aspiration associated with these practices. Sudden respiratory arrest may occur during gastrointestinal decontamination procedures, which cannot be treated easily in time to rescue the patient successfully. Therefore, a protected airway, either when the patient is alert with intact airway reflexes or under intubation, is essential prior to the initiation of any such procedure. In reality, however, owing to the very rapid absorption of Gelsemium alkaloids from the gastrointestinal tract, the value of gastrointestinal decontamination is in itself questionable. As stated earlier, patients are likely to die within 1 h (1) or even 30 min (13) due to respiratory arrest. Therefore, we believe that the rule of treatment is to closely monitor respiration and to provide respiratory support as soon as possible when necessary. To enhance poison elimination, many authors adopt hemodialysis as an effective therapy (21, 22, 33), although it is not well proven or documented in this particular context. The safety of hemodialysis is non-controversial among authors and the likelihood of liver or kidney injury as an effect of Gelsemium poisoning justifies its application (28). In our case, the major intervention was timely respiratory support. After the acute phase, we tried hyperbaric oxygen therapy (HBOT) on the advice of HBOT experts. However, this therapy lacked satisfactory evidence to begin with and, perhaps unsurprisingly, failed to produce significant effects.
There is scant information regarding the prognosis of G. elegans poisoning in previous case reports; these either summarize the case as “recovered and discharged” (7) or “became seriously ill and experienced no symptom relief after medical treatment” (1). The fact is that nearly all cases are reported by emergency-room doctors who are quite unlikely to reevaluate these patients. As mentioned earlier, our patient gradually returned to baseline with some residual dysfunction (e.g., minimal short-term memory impairment and disorientation). There is a need for more observations to define the prognosis of G. elegans poisoning.
Conclusion
Gelsemium elegans is highly toxic. Although patients may die within 30 min due to its strong respiratory depressive effect, they can survive with timely respiratory support and enjoy gradual improvement without delayed postanoxic encephalopathy.
Ethics Statement
No investigation or intervention was performed outside routine clinical care for this patient. As this is a case report, without experimental intervention into routine care, no formal research ethics approval is required. Written, fully informed consent was given and recorded from the patient.
Author Contributions
ZZ, LW, YL, HC, and WZ were involved in the workup of the patient, planning and conducting investigations, and providing clinical care. They reviewed and revised the manuscript and approved the final manuscript as submitted. ZZ, YZ, XF, and WZ planned the case report, drafted the initial manuscript, reviewed and revised the manuscript, and approved the final manuscript as submitted.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewers, SW and MR, handling editor declared their shared affiliation, and the handling editor states that the process nevertheless met the standards of a fair and objective review.
Acknowledgments
The authors would like to thank the patient and her family for participating in this case report.
Funding
This report was supported by the National Natural Science Foundation of China (grant number 81371260), the National Key Clinical Department, the National Key Discipline, and Guangdong Provincial Key Laboratory for Diagnosis and Treatment of Major Neurological Diseases.
References
1. Xiang H, Zhou YJ, Huang PL, Yu CN, Liu J, Liu LY, et al. Lethal poisoning with Gelsemium elegans in Guizhou, China. Public Health (2016) 136:185–7. doi:10.1016/j.puhe.2016.02.031
2. Rujjanawate C, Kanjanapothi D, Panthong A. Pharmacological effect and toxicity of alkaloids from Gelsemium elegans Benth. J Ethnopharmacol (2003) 89(1):91–5. doi:10.1016/S0378-8741(03)00267-8
3. Jin GL, Su YP, Liu M, Xu Y, Yang J, Liao KJ, et al. Medicinal plants of the genus Gelsemium (Gelsemiaceae, Gentianales) – a review of their phytochemistry, pharmacology, toxicology and traditional use. J Ethnopharmacol (2014) 152(1):33–52. doi:10.1016/j.jep.2014.01.003
4. John Bell LRCP. Note on a case of poisoning by Gelsemium. Lancet (1908) 4410(171):717. doi:10.1016/S0140-6736(00)67315-4
5. Zhang YG, Huang GZ. Poisoning by toxic plants in China. Report of 19 autopsy cases. Am J Forensic Med Pathol (1988) 4(9):313–9.
6. Fung HT, Lam KK, Lam SK, Wong OF, Kam CW. Two cases of Gelsemium elegans Benth. poisoning. Hong Kong J Emerg Med (2007) 4(14):221–4.
7. Xiao J, Wang L, Liu YM, Ma WE. Analysis of diagnosis and treatment of eight cases of Gelsemium elegans Benth. poisoning. Med Front (2016) 6(3):357.
8. Mo GY, Han ZY, Ruan AH. Investigation of a case of Gelsemium elegans poisoning due to ingestion of rice wine. China Health Care Nutr (2015) 1(5):130, 133.
9. Cao W-Z, Zhang T, Zhong D-C, Chen R. Long QT syndrome caused by Gelsemium elegans poisoning in a case. Med Inf (2015) 28(16):332. doi:10.3969/j.issn.1006-1959.2015.16.509
10. Li L, Zeng Y. Forensic analysis of a case of suicide by using Gelsemium elegans. Guangdong Gongan Keji (2015) 23(2):74–5.
11. Liu ZC, Song XM. Experience of comprehensive treatment of 8 cases of acute Gelsemium elegans poisoning. Int Med Health Guid News (2014) 20(3):391–2. doi:10.3760/cma.j.issn.1007-1245.2014.03.034
12. Nie XM, Mai WL, Huang CL. Pre-hospital first aid and nursing of Gelsemium elegans poisoning. Med Forum (2014) 18(21):2876–7.
13. Lin XJ, Lu FJ, Lu CH. Investigation of a case of Gelsemium elegans poisoning caused by ingestion of herb medicinal liquor. World Health Digest (2013) 10(3):89–90. doi:10.3969/j.issn.1672-5085.2013.03.084
14. Wen J. Clinical analysis of 18 cases of Gelsemium elegans poisoning. Med Forum (2013) 17(28):3764–5. doi:10.3969/j.issn.1672-1721.2013.28.061
15. Long J, Zhou J, Liang Z. Investigation and analysis of a case of food poisoning caused by eating Gelsemium elegans. Pract Prev Med (2013) 20(12):1472–3. doi:10.3969/j.issn.1006-3110.2013.12.023
16. Meng D, Luo Y. The experience and lessons of rescuing 8 cases of Gelsemium elegans poisoning. Chin Commun Doctors (2012) 14(16):315. doi:10.3969/j.issn.1007-614x.2012.16.309
17. Li W. Clinical analysis of 78 cases of Gelsemium elegans poisoning. Med Inf (2011) 24(9):5872–3.
18. Chen R. The experience of rescuing respiratory and cardiac arrest caused by Gelsemium elegans poisoning treatment. China Pract Med (2011) 6(19):195–6. doi:10.3969/j.issn.1673-7555.2011.19.149
19. Yue-nan S, Zeng W, Huang W-C. Preventive tracheal intubation in the emergency treatment of Gelsemium elegans poisoning. Int Med Health Guid News (2009) 15(23):22–4. doi:10.3760/cma.j.issn.1007-1245.2009.23.008
20. Wei L, Li H. A report of two deaths caused by Gelsemium elegans poisoning. J Appl Prev Med (2009) 15(4):245. doi:10.3969/j.issn.1673-758X.2009.04.028
21. Pan G. Rescue of 8 cases of Gelsemium elegans poisoning. China Pharm (2008) 17(21):56–7. doi:10.3969/j.issn.1006-4931.2008.21.039
22. Lu Z, Deng J, Chen Z, Lu X, Feng J, Zeng F. Survey of a poisoning in college students due to accidental drinking Gelsemium elegances Benth’s flower tea. China Trop Med (2007) 7(1):147–8. doi:10.3969/j.issn.1009-9727.2007.01.074
23. Liu J. The treatment of collective Gelsemium elegans poisoning. J Dali Univ (2007) 6(z1):189. doi:10.3969/j.issn.1672-2345.2007.z1.085
24. Lu S, Su X, Huang Z. A case report of rescuing respiratory arrest due to Gelsemium elegans poisoning. Acta Medicinae Sinica (2006) 19(2):184. doi:10.3969/j.issn.1008-2409.2006.02.121
25. Dai Q. Electroencephalogram report of 6 cases of Gelsemium elegans poisoning. J Clin Electroneurophysiol (2006) 15(5):318. doi:10.3969/j.issn.1674-8972.2006.05.019
26. Liu G, Zhang C. Clinical analysis of 8 cases of Gelsemium elegans poisoning. Lingnan J Emerg Med (2006) 11(6):462. doi:10.3969/j.issn.1671-301X.2006.06.035
27. Liu H-Y. Survey of a food poisoning due to mistaken eating. China Trop Med (2006) 6(4):730. doi:10.3969/j.issn.1009-9727.2006.04.102
28. Chen S, Chen Y, Xu Z. A discussion of clinical characteristics and treatment of Gelsemium elegans poisoning. Chin J Crit Care Med (2006) 26(4):298–300. doi:10.3969/j.issn.1002-1949.2006.04.022
29. Chen P, Liu Q. Investigation of a case of poisoning due to ingestion of Gelsemium elegans. Strait J Prev Med (2006) 12(5):31–2. doi:10.3969/j.issn.1007-2705.2006.05.019
30. Xuan R. A case report of intoxication due to treatment of scabies by using Gelsemium elegans and realgar wine. J Emerg Tradit Chin Med (2005) 14(5):397. doi:10.3969/j.issn.1004-745X.2005.05.071
31. Ban X. Clinical analysis of 17 cases of Gelsemium elegans poisoning. Anthol Med (2005) 24(2):225–6. doi:10.3969/j.issn.1673-6575.2005.02.058
32. You S, Lian Z. Rescue 13 cases of Gelsemium elegans poisoning. Chin J Gen Pract (2004) 3(4):280. doi:10.3760/cma.j.issn.1671-7368.2004.04.043
33. Xiang Y, Qian S. A case report of a successful rescue of severe Gelsemium elegans poisoning. Guangdong Med J (2001) 22(9):776. doi:10.3969/j.issn.1001-9448.2001.09.080
34. Luo F. A report of 37 cases of Gelsemium elegans poisoning. Ningxia Med J (1999) 21(8):507. doi:10.3969/j.issn.1001-5949.1999.08.040
35. Yuan H, Wen L. Research advance in gelsemine and its rejuvenation in animals. Guangdong Feed (2002) 11(2):30–2. doi:10.3969/j.issn.1005-8613.2002.02.015
36. Lai CK, Chan YW. Confirmation of Gelsemium poisoning by targeted analysis of toxic Gelsemium alkaloids in urine. J Anal Toxicol (2009) 33(1):56–61. doi:10.1093/jat/33.1.56
37. Zhang S, Hu S, Yang X, Shen J, Zheng X, Huang K, et al. Development of a liquid chromatography with mass spectrometry method for the determination of gelsemine in rat plasma and tissue: application to a pharmacokinetic and tissue distribution study. J Sep Sci (2015) 38(6):936–42. doi:10.1002/jssc.201401168
Keywords: coma, Gelsemium elegans, poisoning, depressive disorder, suicide
Citation: Zhou Z, Wu L, Zhong YH, Fang XB, Liu YM, Chen HB and Zhang WX (2017) Gelsemium elegans Poisoning: A Case with 8 Months of Follow-up and Review of the Literature. Front. Neurol. 8:204. doi: 10.3389/fneur.2017.00204
Received: 14 October 2016; Accepted: 27 April 2017;
Published: 17 May 2017
Edited by:
Elisabeth Donahey, Loyola University Health System, USAReviewed by:
Megan A. Rech, Loyola University Medical Center, USAStephanie Lynn Bennett Wesling, Loyola University Medical Center, USA
Christoph Stretz, Yale School of Medicine, USA
Copyright: © 2017 Zhou, Wu, Zhong, Fang, Liu, Chen and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhang Weixi, d2VpeGl6aGFuZ0Bmb3htYWlsLmNvbQ==
 Zhou Zhou
Zhou Zhou Lei Wu
Lei Wu Yuhua Zhong
Yuhua Zhong Xiaobo Fang
Xiaobo Fang Yanmei Liu
Yanmei Liu Weixi Zhang
Weixi Zhang