- 1Institute for Stroke and Dementia Research, University Hospital, LMU Munich, Munich, Germany
- 2Neurology Unit, Stroke Unit, Arcispedale Santa Maria Nuova, Azienda Unità Sanitaria Locale-IRCCS Reggio Emilia, Reggio Emilia, Italy
- 3MVZ Labor PD Dr. Volkmann und Kollegen, Gesellschaft Bürgerlichen Rechts, Karlsruhe, Germany
- 4Klinik für Neurologie, SLK-Kliniken Heilbronn GmbH, Heilbronn, Germany
- 5Institut und Poliklinik für Neuroradiologie, University Hospital Carl Gustav Carus, Dresden, Germany
Background: Cortical superficial siderosis (cSS) represents a key neuroimaging marker of cerebral amyloid angiopathy (CAA) that is associated with intracranial hemorrhages and cognitive impairment. Nevertheless, the association between cSS and core cerebrospinal fluid (CSF) biomarkers for dementia remain unclear.
Methods: One hundred and one patients with probable (79%, 80/101) or possible (21%, 21/101) CAA according to the modified Boston criteria and mild cognitive impairment according to Petersen criteria were prospectively included between 2011 and 2016. CSF analyses of ß-amyloid 42, ß-amyloid 40, total tau and phosphorylated tau were performed using sandwich-type enzyme-linked immunosorbent-assay. All patients received MRI and Mini-Mental-State Examination (MMSE). Logistic regression analysis was used to adjust for possible confounders.
Results: cSS was present in 61% (62/101). Of those, 53% (33/62) had disseminated cSS and 47% (29/62) focal cSS. ß-amyloid 42 was lower in patients with cSS than in patients without cSS (OR 0.2; 95% CI 0.08–0.6; p = 0.0052) and lower in patients with disseminated cSS than in those with focal cSS (OR 0.02; 95% CI 0.003–0.2; p = 0.00057). Presence of cSS had no association with regard to ß-amyloid 40, total tau and phosphorylated tau.
Conclusions: Our results demonstrate that the presence and extent of cSS are associated with reduced CSF ß-amyloid 42 levels. Further studies are needed to investigate the underlying mechanisms of this association.
Introduction
Cerebral amyloid angiopathy (CAA)—characterized by the deposition of ß-amyloid in the walls of leptomeningeal vessels—is a common cerebral small vessel disease and a major cause of intracerebral hemorrhage in the elderly (1–3). Furthermore, it has become evident that CAA is associated with cognitive impairment (4). Specifically, it has been shown that CAA patients perform worse in executive functioning, perceptual speed, and episodic memory compared with normative values (5).
The various CAA-related MRI structural lesions essentially represent vascular-mediated brain damage, rather than abnormalities of CAA-laden vessels themselves. CSF biomarkers that are typically altered in AD [ß-amyloid 42 (Aß42), ß-amyloid 40 (Aß40), total tau (t-tau) and phosphorylated tau (p-tau)] may offer another approach to understand underlying mechanisms and courses of patients with CAA (6). While Aß42 can be detected in both senile plaques of AD patients and capillaries of CAA patients, Aß40 deposition is supposed to appear preferentially in the walls of leptomeningeal arteries of CAA patients (7, 8).
More recently, it has been suggested that CAA-related cognitive impairment is more pronounced in a subgroup of CAA patients that demonstrate the MRI marker “cortical superficial siderosis” (cSS), which most likely reflect blood residues in the subarachnoid space (9). In line with this, patients from memory clinic populations have a higher cSS prevalence of 2–6% (10–12) compared to the general population at around 1% (13).
We hypothesized that differences between cSS positive and cSS negative patients might also be reflected in different CSF biomarker profiles. Since cSS patients are regarded to be of a higher risk for future neurovascular events and for cognitive dysfunction, we hypothesized that cSS positive patients would demonstrate lower levels of Aß40 and Aß42 together with elevated tau levels. Hence, we analyzed the core CSF biomarker profile in a prospective cohort of patients with mild cognitive impairment and the diagnosis of a possible or probable CAA.
Methods
Subjects
Screening took place in the outpatient clinics of the university hospitals in Reggio Emilia (Italy) and LMU Munich (Germany). Three hundred and forty-five patients with subjective cognitive impairment and the MRI based suspicion of a CAA were screened. Of those 71% (244/345) did not undergo lumbar puncture because of (a) normal objective cognitive testing (n = 134), (b) patient decline (n = 90) or (c) intake of oral anticoagulation (n = 20). The remaining 101 patients entered the final analysis. Seventy-nine percent (80/101) had a probable CAA and 21% (21/101) a possible CAA according to the modified Boston criteria (2). All patients in the final analysis had the diagnosis of mild cognitive impairment based on the Petersen criteria (14). The reasons for the initial presentation at the outpatient clinics were distributed as follows: past intracranial hemorrhage 34% (34/101), memory complaints 39% (39/101), past ischemic stroke or TIA 14% (14/101), transient focal neurological episodes (TFNE) or presumed focal seizure 4% (4/101), dizziness, headache or other unspecific neurological complaints 10% (10/101). Patients not included into the study did not differ in terms of age gender and cardiovascular risk factors (Supplementary Table 3). Informed consent were obtained from each patient according to the Declaration of Helsinki. Ethics approval was obtained from the regional ethics board.
MRI
MR images in Munich were acquired on a 3 Tesla MRI scanner (Signa HDxt, GE Healthcare), in Reggio Emilia on a 1.5 Tesla scanner (Achieva, Philips Healthcare). Cortical superficial siderosis was identified on T2*-weighted gradient-echo sequence (GRE) or susceptibility-weighted imaging (SWI) by two trained raters (FAW and CC). Disagreement in two cases was resolved by consensus read (interrater κ = 0.97). Cerebral microbleeds (CMB) were defined according to the STRIVE criteria (15). CMB were manually marked on lesion masks, which were then normalized to 1 mm Montreal Neurological Institute (MNI) standard space and automatically rated as described before (16). CMB were categorized in 4 groups (0 CMB, 1 CMB, 2–4 CMB and ≥5 CMB) according to the recent literature (17, 18).
CSF
CSF samples were obtained by lumbar puncture within 6 weeks after MRI, collected in 10 mL polypropylene tubes and centrifuged within 2 h, and then frozen until analysis. Aß42, Aß40, t-tau and p-tau were measured using sandwich type enzyme linked immunosorbent assays; Ab42 was measured with Innotest ß-amyloid (42), Ab40 was measured with Innotest ß-amyloid (40), t-tau with Innotest hTau-Ag, and p-tau with Innotest Phospho-tau (181 P) (Innogenetics, 9,052 Gent, Belgium, www.fujirebio-europe.com). The unit used for biomarkers is pg/mL.
The inter lot variation coefficient between the centers were Aß42 5.15%, Aß40 7.87%, t-tau 14.01%, p-tau 16.2% and Aß42 5.7%, Aß40 8.2%, t-tau 7.5, p-tau 16.4. Further, there were no significant differences between the median levels of core CFS markers between the two centers, indicating a good comparability of the CSF assays (Supplementary Table 4).
Statistical Analysis
Baseline characteristics and risk factors were compared using chi-square tests, Fisher's exact tests and student's t-tests where appropriate. Logistic regression analyses corrected for age, sex and CMB category was performed using the cSS groups as dependent variables. To use the binary cSS status as the dependent variable in the logistic regression, the analysis was inverted. All CSF marker levels were log-transformed for regression analysis and are presented as median with interquartile range to account for the non-normal distribution. Logistic regression analyses with MMSE as the dependent variable were performed using logistic regression corrected for age, sex, hypertension, diabetes and hypercholesterinemia. MRI field strength and T2*-weighted method (GRE vs. SWI) were tested as a variable in univariate analysis. Therefore, it was not included into the logistic regression model. The relationship between cSS and the categories of CMB and CSF markers was tested using ordinal logistic regression under a proportional odds assumption. For the additional model, we applied a backward stepwise regression analysis optimizing on Akaike Information Criterion (AIC) to statistically select for relevant covariates. As a result, hypertension was selected as the only parameter. All analyses was performed using R (R version 3.5.1: A language and environment for statistical computing; R Foundation for Statistical Computing, Vienna, Austria).
Results
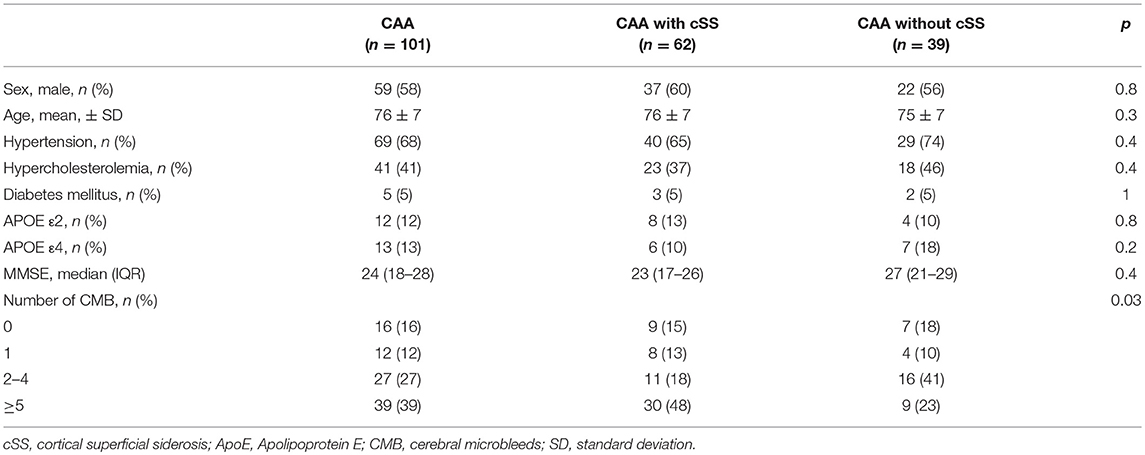
CSS was present in 61% (62/101). Patients with and without cSS were well balanced with regard to age, sex, and cardiovascular risk factors. MRI field strength and T2*-weighted method (GRE vs SWI) did not show any significant effect on the presence and extent of cSS (all p > 0.4). Detailed baseline characteristics are shown in Tables 1, 2.
Presence of cSS and MMSE
CAA patients with cSS had numerically lower median MMSE values than CAA patients without cSS (23 vs. 27, Table 1). However, this difference did not reach statistical significance, neither without adjustment (0.4), nor in a model adjusting for sex, age and cardiovascular risk factors (p = 0.05) nor in a model adjusting for hypertension only (p = 0.1).
Presence of cSS and CSF Markers
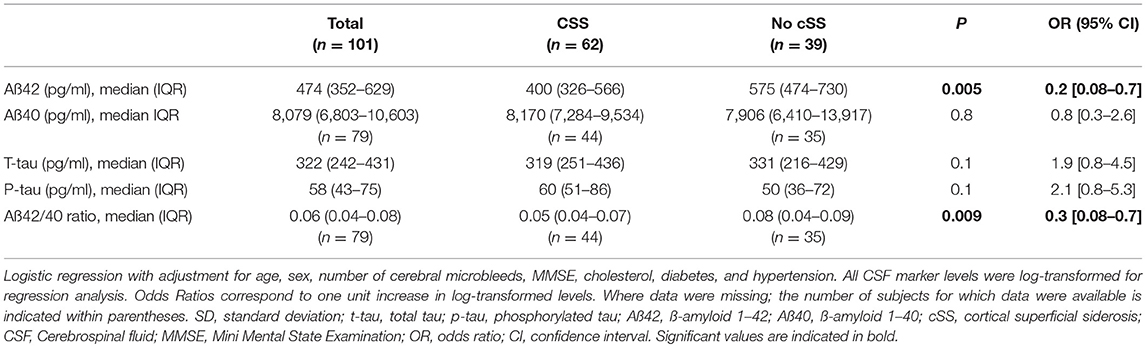
In logistic regression analyses, Aß42 was significantly lower in patients with cSS compared to those without (OR 0.2; 95% CI 0.08–0.7; p = 0.0052, Figure 1 and Table 3). Further, the Aß42/40 ratio was significant lower in patients with cSS than in patients without cSS (OR 0.3; 95% CI 0.08–0.7; p = 0.0091). Patients with and without cSS did not show significantly different CSF levels of Aß40 (OR 0.8; 95% CI 0.3–2.6; p = 0.77), t-tau (OR 1.9; 95% CI 0.8–4.5; p = 0.12), and p-tau (OR 2.1; 95% CI 0.8–5.3; p = 0.12).
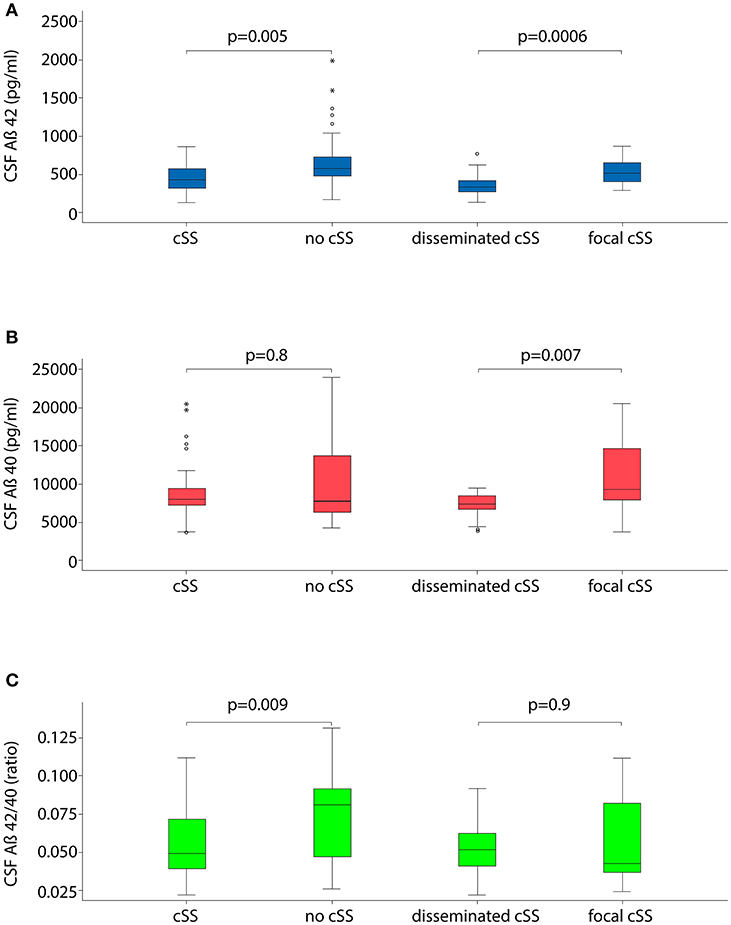
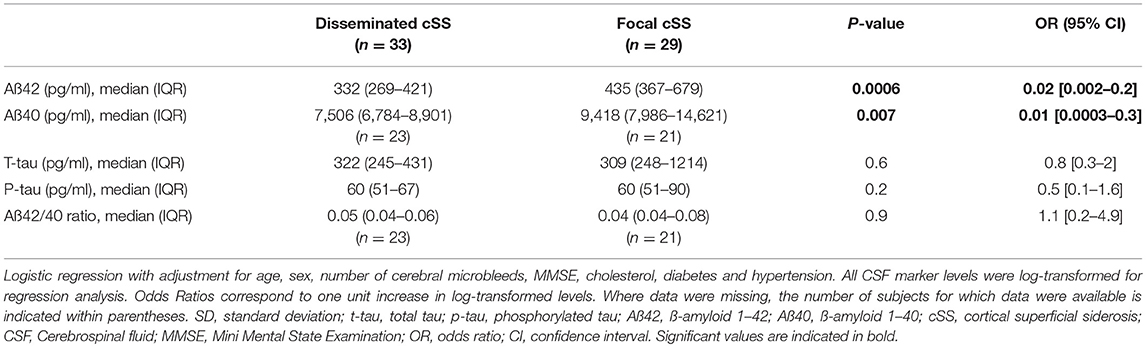
Figure 1. Boxplots illustrating cerebrospinal fluid (CSF) markers for dementia stratified for the presence and extent of cortical superficial siderosis (cSS). (A) CSF level of Aß42; (B) CSF level of Aß40; and (C) ratio of Aß42 to Aß40.
Extent of cSS and CSF Markers
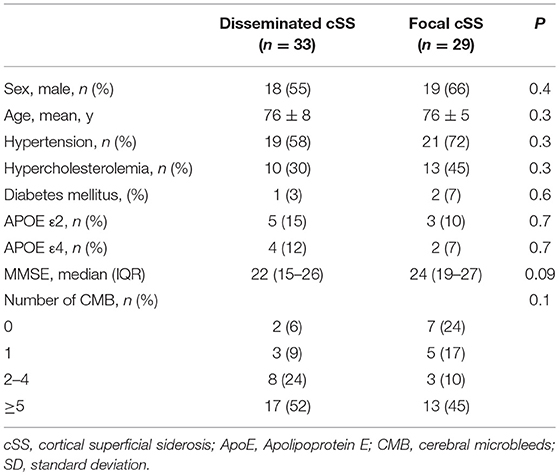
Out of the overall cohort, 33% (33/101) had disseminated (i. e. affecting >3 cortical sulci) cSS and 29% (29/101) had focal (affecting ≤ 3 sulci) cSS. CSF levels of Aß42 and Aß40 were significantly lower in patients with disseminated cSS than in focal cSS (OR 0.02; 95% CI 0.002–0.2; p = 0.00057 and OR 0.01; 95% CI 0.0003–0.3; p = 0.0069, please see Figure 1 and Table 4).
CMB and CSF Marker
Eighty-four percent (85/101) of all patients had any CMB. Using Chi-Square test, there was a significant difference regarding the number of CMB in patients with and without cSS stratified for groups (p = 0.03). While patients with cSS less often had 2–4 CMB than patients without cSS (18%, 11/62 and 41%, 16/39), cSS patients were overrepresented in the group of >5 CMB (48%, 30/62 and 23%, 9/39). The number of CMB did not differ between patients with disseminated and focal cSS (p = 0.1).
In ordinal logistic regression analysis adjusting for age, sex and cardiovascular risk factors, there was no significant correlation between the number of CMB and all tested CSF parameters (all p > 0.05).
cSS and CSF Markers in Subgroups
We repeated the logistic regression analyses restricted to patients with probable CAA according to the modified Boston criteria (Supplementary Tables 1, 2). In that analysis, Aß42 remained lower in patients with cSS compared to those without (OR 0.3; 95% CI 0.07–0.9; p = 0.05). Further, the Aß42/40 ratio was significantly lower in patients with cSS than in patients without cSS (OR 0.3; 95% CI 0.07–0.9; p = 0.04). Also patients with disseminated cSS still showed significantly lower levels of Aß42 (OR 0.02; 95% CI 0.002–0.3; p = 0.003) and lower levels of Aß40 (OR 0.0004; 95% CI 0.0000006–0.3; p = 0.01).
Discussion
The main results from this cohort study of 101 patients with a possible or probable CAA and CSF samples are that (1) CSF Aß42 is significantly lower in patients with cSS than in patients without cSS, and (2) CSF Aß42 is significantly lower in patients with disseminated cSS (>3 sulci) than in CAA patients with focal cSS.
The current results indicate that both the presence and the extent of cSS are associated with lower Aß42 CSF levels. Since accumulating evidence suggests that the presence and extent of cSS might be a marker of CAA severity with higher recurrence rates of intracerebral hemorrhage (3, 16), it may be possible that Aß42 may also act as a biomarker of CAA severity. Previous data on Aß42 CSF levels in patients with CAA are relatively rare and limited in power. A recent meta-analysis on the topic identified five heterogeneous CAA cohorts that included overall 60 patients (6).
The underlying mechanism of the presumed association between cSS and Aß42 remain to be elucidated. However, our results are consistent with neuropathological studies that found Aß42 trapped in the cerebral vessels of CAA patients and therefore hampers the transport of Aß42 toward the cerebrospinal fluid. Also, in vitro data and PET studies suggest, that low Aβ42 in CSF of CAA patients is related to ß-amyloid deposition in the vessel walls and the adjacent brain parenchyma (19, 20). Further, experimental studies and neuropathological findings have demonstrated a high co-incidence between CAA and Alzheimer's disease (AD) (21, 22). Therefore, it may be speculated that the demonstrated reduced Aß42 levels may partly be explained by a higher rate of a co-incident AD in patients with cSS. However, we did not detect higher tau levels in cSS patients contradicting this hypothesis. Hence neuropathological studies are needed to disentangle the complex interplay between cSS and AD.
Patients with disseminated cSS had significantly lower Aß40 levels than those with focal cSS in our cohort. We are not aware of any other study that investigated the extent of cSS and its association with Aß40. However, this result is in line with a case series of 15 patients with hereditary cerebral hemorrhage with amyloidosis–Dutch type (HCHWA-D) that detected decreased CSF Aß40 levels in HCHWA-D patients in comparison to healthy controls (23). Further, it is in accordance with neuropathological reports demonstrating deposition of Aß40 within the vessel walls of CAA patients (19). Since patients with disseminated cSS most probably represent a group of more severely affected CAA patients, they may harbor more Aß40 depositions within the vessel wall and consecutively demonstrate lower CSF Aß40 levels.
The MMSE values of patients with cSS were numerically lower than in patients without cSS as hypothesized by previous findings that detected a higher prevalence of cSS patients in memory clinics (10, 11). However, this difference did not reach statistical significance. Hence, the results of the current study prevent meaningful conclusions on the extent and etiology of cognitive impairment in patients with cSS. Longitudinal studies with repeated extensive neuropsychological testing and cSS lesion-volume mapping are needed to disentangle the complex interplay of vascular pathology and ß-amyloid-pathology in cSS and CAA.
Of note, there was no consistent association between the presence of cSS and Aß40 in the current study, which may relate to the experimental finding that Aß42 deposits earlier in the vessel wall than Aß40 (23–25). Alternatively, it might be speculated from the odds ratio of 0.8 that slight Aß40 CSF levels differences are present, but the association did not reach statistical significance due to a lack of statistical power. We did not formally compare patients with focal cSS and no cSS due to the small sample sized within subgroups, but it also may be speculated that the effect of lower Aß40 values in cSS is primarily driven by the subgroup of patients with disseminated cSS rather than those with focal cSS. Further studies with larger sample size are needed to fully capture the effect of cSS subgroups and Aß40.
Interestingly, our results did not show an association between the number of CMB and reduced levels of Aß42 or Aß40 in contrast to other reports (23, 26, 27). This discrepancy may partly be related to differences in cohort characteristics since the prevalence of CMB was very high with 84% in this cohort resulting in a potential ceiling effect of the association.
Our study has several strengths including the prospective design, the standardized CSF sampling and analysis as well as the central MR imaging. Limitations include the use of two different MR scanners with different field strengths (3.0 or 1.5 Tesla), that may have had an impact on the number of CMB and potentially the extent of cSS. However, we included MRI field strength and the effect of T2*-weighted method (GRE vs. SWI) as a variable in univariate analysis were it did not show a significant association with CSF markers which makes a strong effect very unlikely. Further, there are no pathological proven CAA cases included. However, 79% of patients were diagnosed with probable CAA according to the modified Boston criteria, which have demonstrated an excellent sensitivity and specificity for CAA (2). Additionally, we repeated the logistic regression analysis on core CSF markers restricted to the subgroup of patients fulfilling the diagnosis of probable CAA with stable results. Finally, differences in medical history between patients might have confounded results in CSF analysis. However, in the backward stepwise regression analysis, only hypertension resulted relevant and a subgroup analysis stratified for differences in medical history was regarded not meaningful due to the limited sample sizes.
Conclusions
The present study demonstrates for the first time that presence and extent of cSS are associated with lower Aß42 CSF levels. Our data suggest that Aß42 may act as an additional marker of CAA severity. Nevertheless, further studies are needed to proof these findings and to investigate its underlying mechanisms.
Data Availability
All datasets generated for this study are included in the manuscript and/or the Supplementary Files.
Consent for Publication
All authors approved the final manuscript and consent to its publication.
Ethics Statement
Informed consent was obtained from each patient according to the Declaration of Helsinki. Ethics approval was obtained from the regional ethics board (Ethikkommission LMU Munich). Written informed consent was obtained from all patients.
Author Contributions
CC and FW designed the study. CC, MZ, DJ, and VS acquired clinical, neuropsychological and CSF data. RM analyzed the data. AK, JL, CO, and MD interpreted the manuscript. CC and FW interpreted and drafted the manuscript. FW supervised and revised the manuscript. All authors read and approved the final manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fneur.2019.00439/full#supplementary-material
Abbreviations
CAA, Cerebral amyloid angiopathy; cSS, Cortical superficial siderosis; CSF, Cerebrospinal fluid; Aß42, ß-amyloid 42; Aß40, ß-amyloid 40; t-tau, total tau; p-tau, phosphorylated tau; MMSE, Mini-Mental State Examination; AD, Alzheimer's disease; CMB, Cerebral microbleeds; SD, Standard deviation; OR, odds ratio; CI, confidence interval; y, years; DM, Diabetes mellitus; ApoE, Apolipoprotein E; IQR, interquartile range; HCHWA-D, hereditary cerebral hemorrhage with amyloidosis of the Dutch type.
References
1. Wollenweber FA, Opherk C, Zedde M, Catak C, Malik R, Duering M, et al. Prognostic relevance of cortical superficial siderosis in cerebral amyloid angiopathy. Neurology. (2019) 92:e792–801. doi: 10.1212/WNL.0000000000006956
2. Linn J, Halpin A, Demaerel P, Ruhland J, Giese AD, Dichgans M, et al. Prevalence of superficial siderosis in patients with cerebral amyloid angiopathy. Neurology. (2010) 74:1346–50. doi: 10.1212/WNL.0b013e3181dad605
3. Charidimou A, Linn J, Vernooij MW, Opherk C, Akoudad S, Baron JC, et al. Cortical superficial siderosis: detection and clinical significance in cerebral amyloid angiopathy and related conditions. Brain. (2015) 138:2126–39. doi: 10.1093/brain/awv162
4. Wermer MJH, Greenberg SM. The growing clinical spectrum of cerebral amyloid angiopathy. Curr Opin Neurol. (2018) 31:28–35. doi: 10.1097/WCO.0000000000000510
5. Case NF, Charlton A, Zwiers A, Batool S, McCreary CR, Hogan DB, et al. Cerebral amyloid angiopathy is associated with executive dysfunction and mild cognitive impairment. Stroke. (2016) 47:2010–6. doi: 10.1161/STROKEAHA.116.012999
6. Charidimou A, Friedrich JO, Greenberg SM, Viswanathan A. Core cerebrospinal fluid biomarker profile in cerebral amyloid angiopathy: a meta-analysis. Neurology. (2018) 90:e754–62. doi: 10.1212/WNL.0000000000005030
7. Herzig MC, Winkler DT, Burgermeister P, Pfeifer M, Kohler E, Schmidt SD, et al. Abeta is targeted to the vasculature in a mouse model of hereditary cerebral hemorrhage with amyloidosis. Nat Neurosci. (2004) 7:954–60. doi: 10.1038/nn1302
8. Thal DR, Griffin WS, de Vos RA, Ghebremedhin E. Cerebral amyloid angiopathy and its relationship to Alzheimer's disease. Acta Neuropathol. (2008) 115:599–609. doi: 10.1007/s00401-008-0366-2
9. Lummel N, Wollenweber FA, Demaerel P, Bochmann K, Malik R, Opherk C, et al. Clinical spectrum, underlying etiologies and radiological characteristics of cortical superficial siderosis. J Neurol. (2015) 262:1455–62. doi: 10.1007/s00415-015-7736-1
10. Wollenweber FA, Buerger K, Mueller C, Ertl-Wagner B, Malik R, Dichgans M, et al. Prevalence of cortical superficial siderosis in patients with cognitive impairment. J Neurol. (2014) 261:277–82. doi: 10.1007/s00415-013-7181-y
11. Zonneveld HI, Goos JD, Wattjes MP, Prins ND, Scheltens P, van der Flier WM, et al. Prevalence of cortical superficial siderosis in a memory clinic population. Neurology. (2014) 82:698–704. doi: 10.1212/WNL.0000000000000150
12. Inoue Y, Nakajima M, Uetani H, Hirai T, Ueda M, Kitajima M, et al. Diagnostic significance of cortical superficial siderosis for Alzheimer disease in patients with cognitive impairment. AJNR Am J Neuroradiol. (2016) 37:223–7. doi: 10.3174/ajnr.A4496
13. Vernooij MW, Ikram MA, Hofman A, Krestin GP, Breteler MM, van der Lugt A. Superficial siderosis in the general population. Neurology. (2009) 73:202–5. doi: 10.1212/WNL.0b013e3181ae7c5e
14. Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med. (2004) 256:183–94. doi: 10.1111/j.1365-2796.2004.01388.x
15. Wardlaw JM, Smith EE, Biessels GJ, Cordonnier C, Fazekas F, Frayne R, et al. Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol. (2013) 12:822–38. doi: 10.1016/S1474-4422(13)70124-8
16. Wollenweber FA, Baykara E, Zedde M, Gesierich B, Achmuller M, Jouvent E, et al. Cortical superficial siderosis in different types of cerebral small vessel disease. Stroke. (2017) 48:1404–7. doi: 10.1161/STROKEAHA.117.016833
17. Charidimou A, Boulouis G, Roongpiboonsopit D, Auriel E, Pasi M, Haley K, et al. Cortical superficial siderosis multifocality in cerebral amyloid angiopathy: a prospective study. Neurology. (2017) 89:2128–35. doi: 10.1212/WNL.0000000000004665
18. Charidimou A, Martinez-Ramirez S, Reijmer YD, Oliveira-Filho J, Lauer A, Roongpiboonsopit D, et al. Total magnetic resonance imaging burden of small vessel disease in cerebral amyloid angiopathy: an imaging-pathologic study of concept validation. JAMA Neurol. (2016) 73:994–1001. doi: 10.1001/jamaneurol.2016.0832
19. Vinters HV. Emerging concepts in Alzheimer's disease. Annu Rev Pathol. (2015) 10:291–319. doi: 10.1146/annurev-pathol-020712-163927
20. Na HK, Park JH, Kim JH, Kim HJ, Kim ST, Werring DJ, et al. Cortical superficial siderosis: a marker of vascular amyloid in patients with cognitive impairment. Neurology. (2015) 84:849–55. doi: 10.1212/WNL.0000000000001288
21. Thal DR, Ghebremedhin E, Orantes M, Wiestler OD. Vascular pathology in Alzheimer disease: correlation of cerebral amyloid angiopathy and arteriosclerosis/lipohyalinosis with cognitive decline. J Neuropathol Exp Neurol. (2003) 62:1287–301. doi: 10.1093/jnen/62.12.1287
22. Verbeek MM, Kremer BP, Rikkert MO, Van Domburg PH, Skehan ME, Greenberg SM. Cerebrospinal fluid amyloid beta(40) is decreased in cerebral amyloid angiopathy. Ann Neurol. (2009) 66:245–9. doi: 10.1002/ana.21694
23. van Etten ES, Verbeek MM, van der Grond J, Zielman R, van Rooden S, van Zwet EW, et al. beta-amyloid in CSF: biomarker for preclinical cerebral amyloid angiopathy. Neurology. (2017) 88:169–76. doi: 10.1212/WNL.0000000000003486
24. Shinkai Y, Yoshimura M, Ito Y, Odaka A, Suzuki N, Yanagisawa K, et al. Amyloid beta-proteins 1-40 and 1-42(43) in the soluble fraction of extra- and intracranial blood vessels. Ann Neurol. (1995) 38:421–8. doi: 10.1002/ana.410380312
25. Natte R, Yamaguchi H, Maat-Schieman ML, Prins FA, Neeskens P, Roos RA, et al. Ultrastructural evidence of early non-fibrillar Abeta42 in the capillary basement membrane of patients with hereditary cerebral hemorrhage with amyloidosis, Dutch type. Acta Neuropathol. (1999) 98:577–82. doi: 10.1007/s004010051121
26. Kester MI, Goos JD, Teunissen CE, Benedictus MR, Bouwman FH, Wattjes MP, et al. Associations between cerebral small-vessel disease and Alzheimer disease pathology as measured by cerebrospinal fluid biomarkers. JAMA Neurol. (2014) 71:855–62. doi: 10.1001/jamaneurol.2014.754
Keywords: cerebral amyloid angiopathy, cortical superficial siderosis, cerebrospinal fluid, cerebral microbleeds, neuroimaging
Citation: Catak C, Zedde M, Malik R, Janowitz D, Soric V, Seegerer A, Krebs A, Düring M, Opherk C, Linn J and Wollenweber FA (2019) Decreased CSF Levels of ß-Amyloid in Patients With Cortical Superficial Siderosis. Front. Neurol. 10:439. doi: 10.3389/fneur.2019.00439
Received: 22 February 2019; Accepted: 10 April 2019;
Published: 26 April 2019.
Edited by:
Eric Jouvent, Université Sorbonne Paris Cité, FranceReviewed by:
Alessandro Biffi, Harvard Medical School, United StatesYael D. Reijmer, University Medical Center Utrecht, Netherlands
Copyright © 2019 Catak, Zedde, Malik, Janowitz, Soric, Seegerer, Krebs, Düring, Opherk, Linn and Wollenweber. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Frank A. Wollenweber, ZnJhbmsud29sbGVud2ViZXJAbWVkLnVuaS1tdWVuY2hlbi5kZQ==
†These authors have contributed equally to this work as co-first authors
 Cihan Catak
Cihan Catak Marialuisa Zedde2†
Marialuisa Zedde2† Rainer Malik
Rainer Malik Alexander Krebs
Alexander Krebs Marco Düring
Marco Düring Frank A. Wollenweber
Frank A. Wollenweber