- 1Department of Neurology, Hwa Mei Hospital, University of Chinese Academy of Science, Ningbo, China
- 2Ningbo Institute of Life and Health Industry, University of Chinese Academy of Science, Ningbo, China
- 3Department of Neurology, Longyan First Affiliated Hospital, Fujian Medical University, Longyan, China
- 4Department of Rehabilitation, Hwa Mei Hospital, University of Chinese Academy of Science, Ningbo, China
Background: Otolin-1 is an inner ear-specific protein that is exclusively expressed in otoconia and vestibule and cochlea cells. Recent investigations reported that otolin-1 can cross the blood-labyrinthine barrier and that the levels in serum well-reflected otolith status. Serum otolin-1 levels in patients with benign paroxysmal positional vertigo (BPPV) are significantly elevated compared with healthy controls. We aimed to explore whether otolin-1 can also serve as a biomarker for predicting BPPV recurrence.
Method: Patients at our institution with new-onset of idiopathic BPPV between May, 2017 and May, 2018 were recruited and followed up for 2 years. All demographic data of the patients were collected, and serum levels of otolin-1 and other laboratory indicators were measured and compared according to the recurrence status.
Results: A total of 74 patients, who met the inclusion criteria were enrolled in this study, of which 27 (36.5%) patients had suffered one or more episodes of recurrence after undergoing canal repositioning treatments during the study. The serum levels of otolin-1 in patients with recurrent BPPV were significantly higher than those in patients without recurrent BPPV (363.9 vs. 309.8 pg/ml, p = 0.001). In multivariate analysis comparing the second to fourth quartiles (Q2–Q4) against the first quartile (Q1) of otolin-1, the level of otolin-1 in Q4 could significantly predict BPPV recurrence, and the odds ratio (OR) was elevated by approximately 812% (OR = 9.12; 95% confidence interval [CI]: 1.44–57.9; p = 0.019).
Conclusion: High serum levels of otolin-1 were associated with an increased risk of BPPV recurrence, and further investigation is required to confirm this association and clarify the exact mechanism.
Introduction
Benign paroxysmal positional vertigo (BPPV) is the most common cause of peripheral vertigo and affects nearly 17% of patients with dizziness or vertigo. The typical clinical presentation of transient vertigo attacks with characteristic nystagmus is provoked by head position changes (1, 2). Canalith repositioning manoeuvers (CRMs) are the first-line treatments for BPPV, and symptoms can be relieved in more than 95% of cases (3). Additionally, BPPV cases will resolve spontaneously within a few weeks. However, BPPV sometimes recurs with a reported recurrence rate ranging from 7 to 50%, depending on the clinical setting, which results in anxiety and poor quality of life in patients (4, 5). Several studies have explored the risk factor-associated recurrence of BPPV, but the conclusions are conflicting (6, 7). Therefore, exploring the potential markers that predict the recurrence of BPPV may have an important value for the integrated management of BPPV.
Otolin-1 is a scaffolding glycoprotein whose messenger RNA (mRNA) is exclusively expressed in otoconia and cells of semicircular canal cristae, organ of Corti, and marginal cells of the stria vascularis (8–10). Since Parham et al. first reported that otolin-1 can be detected in peripheral blood, some investigations have been carried out and confirmed that serum levels of otolin-1 in patients with BPPV were significantly elevated compared with those in healthy controls (11–14). In addition, the levels of otolin-1 in healthy people increase with age, which is consistent with the age-related demineralization of otoconia and the increased prevalence of BPPV with age (15).
All these studies provided proof of concept for using the inner specific protein otolin-1 as a circulatory biomarker for otoconia degeneration. We speculate that the more severe the otoconia degeneration is, the higher the level of otolin-1. In people with higher levels of otolin-1, otoconia might be more easily exfoliated from the maculae and tends to cause vertigo attacks (16). To date, no studies in the literature have corroborated the recurrence of BPPV with the levels of inner-specific proteins. In this study, we took an initial step to further investigate whether the serum level of otolin-1 can serve as a biomarker for predicting BPPV recurrence events.
Methods
This study was approved by our institutional review board (protocol number KY-2017-014) and performed in accordance with the tenets of the Declaration of Helsinki. Informed consent was obtained from all participants. Patients who were diagnosed with de novo idiopathic BPPV at the Department of Neurology and Emergency, Hwa Mei Hospital, University of Chinese Academy of Science from May 2017 to May 2018 were included. The detailed recruitment and exclusion criteria were described in previous study (12). Since informed consent was obtained, morning fasting blood samples were collected, routine laboratory indexes were assessed, and the remaining serum was stored at −80°C until further analysis. Serum levels of otolin-1 were assessed by a human enzyme-linked immunosorbent assay kit (category number: QY-E03713; QAYEE-BIO, Shanghai, China).
Patients were treated with appropriate CRM and re-evaluated after 7 days until the symptoms and nystagmus were absent. Once the patient's vertigo attack occurred, he or she was asked to return to our hospital to be re-examined. If the interval between the onset of two symptoms was more than 1 month, it was defined as a recurrence. Patients were followed up regularly by telephone or as outpatient at 1, 3, 6, 12, and 24 months. All the data that were recorded included age, sex, lifestyle habits, ongoing health problems, medication history, affected semicircular canal, onset time, interval between blood collection and symptom onset, recurrence rate, and laboratory indicators.
Statistical Analysis
Statistical analysis was performed using SPSS 22.0 (SPSS Inc., Chicago, IL, USA). Continuous variables following normal distributions are expressed as mean ± standard deviation, and those that did not follow a normal distribution are expressed as the median and interquartile range (IQR). Categorical variables are presented as the number of cases and percentage. Comparison between categorical variables was performed using a t-test, the chi-square test, Fisher's test, or Mann-Whitney U-test. Multivariable logistic regression was performed to evaluate the recurrence risk factors for BPPV patients. Multivariate analysis was used to assess recurrent BPPV according to otolin-1 quartiles (the lowest quartile [Q1] was used as the reference). A receiver operating characteristic (ROC) curve was generated to evaluate the ability of serum otolin-1 levels to predict recurrent BPPV and no recurrent BPPV. Values of p < 0.05 were considered statistically significant.
Results
Demographics and Clinical Characteristics of the Subjects
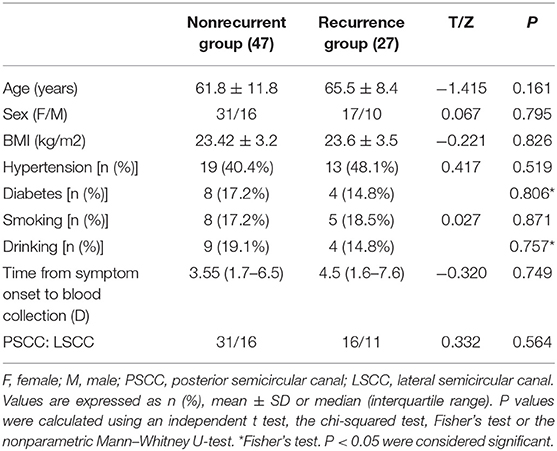
A total of 78 patients diagnosed with de novo idiopathic BPPV were included in the study, while 4 patients were eliminated from the analysis due to loss during the follow-up. There were 27 (36.5%) patients who experienced recurrence after CRM treatment, of whom 18 patients (66.7%) suffered one episode of recurrence, while 7 patients (25.9%) suffered two episodes of recurrence and 2 patients (7.4%) suffered three or more episodes of recurrence during the follow-up. The general demographics of these patients are compared in Table 1 according to the number of recurrence episodes.
Recurrence Risk Factor Analysis
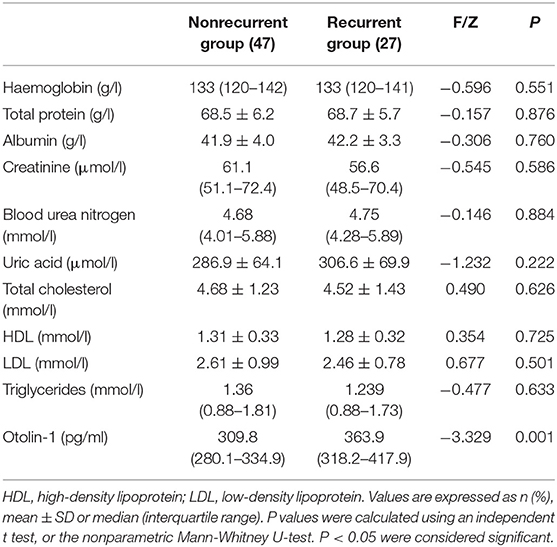
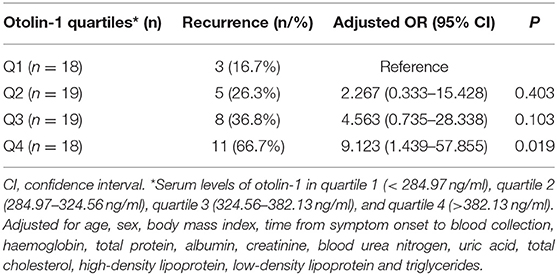
There were no statistical differences in the age distribution, sex ratio, body mass index, lifestyle habits, clinical history, time from symptom onset to blood collection, types of semicircular canal involved, or laboratory results such as hemoglobin, blood creatinine and urea nitrogen, uric acid, liver function, and lipid profiles between the groups (Table 2). Higher serum levels of otolin-1 were observed in patients with recurrent BPPV than in those patients without recurrence (363.9 [IQR: 318.2–417.9] pg/ml vs. 309.8 [280.1–334.9] pg/ml; Z = −3.329; p = 0.001). Multiple logistic regression analyses showed that the serum level of otolin-1 was an independent risk factor for the recurrence of BPPV (OR: 1.004, 95% CI: 1.000–1.007; p = 0.043) (Supplementary Material). The recurrence rate of BPPV across otolin-1 quartiles, ranges from 16.7% (Q1) to 66.7% (Q4). In multivariate analysis models, comparing the second, third, and fourth quartiles (Q2, Q3, and Q4) of otolin-1 levels against Q1, only the levels of otolin-1 in Q4 were correlated with recurrent BPPV (OR: 9.123; 95% CI: 1.44–57.9; p = 0.019) (Table 3). A serum otolin-1 value of 334.7 pg/ml was shown to aid in the prediction of BPPV recurrence, with a sensitivity of 70.4%, a specificity of 74.5%, and an area under the cure of 0.724 (95% CI: 0.602–0.847; Figure 1).
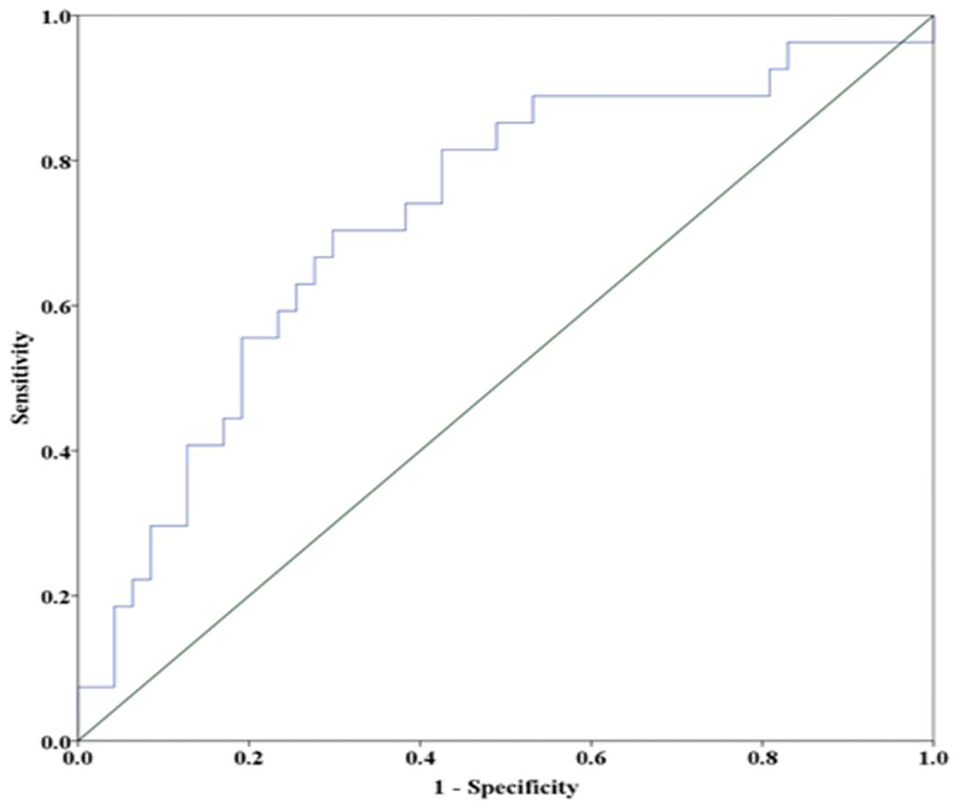
Figure 1. Receiver operator characteristic analysis of otolin-1 for predicting the recurrence BPPV. A cut off value of 334.7 pg/ml provides a sensitivity of 70.4%, a specificity of 74.5%, and an area under the curve of 0.724 (95% CI = 0.602–0.847).
Discussion
From a clinical perspective, biomarkers for investigating otologic disorders are very helpful and can enable prediction of disease, therapeutic targeting, and response. Otolin-1, an inner ear-specific protein, is a potential circulatory biomarker candidate for otoconia degeneration. Our study investigated the correlation between the serum level of otolin-1 and recurrence events in patients with BPPV. This study showed the following: 1) Serum levels of otolin-1 in patients with recurrent BPPV were significantly higher than those in patients with non-recurrent BPPV; 2) elevated serum levels of otolin-1 were associated with a higher risk of recurrent BPPV, suggesting that otolin-1 may be a risk factor for the recurrence of BPPV.
Otoconia are dense crystals composed of calcium carbonate and an organic matrix, which are synthesized during embryonic development and completely calcified on the seventh postnatal day. Otoconial degeneration and dislodged otoconia falling into the canal are the leading causes of BPPV (17–19). Dislodged otoconia can also be dissolved in the endolymph, and some matrix proteins may be reabsorbed and released into circulation (20). Due to the limitations of current technology, it is difficult to observe otoconia by radiographic testing in real time and even more difficult to study vital specimens. Thus, it is important to explore potential biomarkers for otoconia-related diseases. Therefore, an increasing number of studies have focused on the metabolism of otoconial matrix proteins.
The otoconia organic matrix is composed of a variety of proteins, the chief constituents of which are otoconin-90 and otolin-1. Otolin-1 is a secreted glycoprotein whose mRNA expression is restricted to the support cells of the vestibular maculae, semicircular canal cristae, and organ of Corti and marginal cells of the stria vascularis (10, 21). Parham et al. first reported that otolin-1 could be detected in serum and that the levels were higher in patients with BPPV than in healthy controls (11). Since BPPV in some of these patients was not in the acute stage, only one-third of the serum otolin-1 values of the patients were higher than those in the control range. Since then, a series of studies have been carried out to explore whether otolin-1 has any value in the diagnosis and prognosis of otolith-related diseases. Dogan et al. (22) reported that the levels of serum otolin-1 were significantly increased in patients who had undergone mastoidectomy. Other studies conducted by Irugu et al. (13) and Yadav et al. (14) showed that the serum levels of otolin-1 in patients with BPPV were significantly higher than those in healthy controls. Recently, Naples et al. (23) in a pilot study evaluated a role for prestin and otolin-1 as biomarkers to differentiate Meniere's disease (MD) from vestibular migraine (VM). Prestin and otolin-1 levels were not significantly elevated in MD patients compared to VM patients. Thus, their role in differentiating MD from VM remains to be elucidated in future studies. Previous studies found that the levels of otolin-1 in peripheral blood increase with age, which is consistent with scanning electron microscopy findings of age related degeneration of otoconia and the increased prevalence of BPPV with aged. The changes in serum levels of otolin-1 reported in these studies reflected the processes of breakdown and degradation of otoconia. Similarly, a study found that otoconin-90 could be detected in peripheral blood and the levels in patients with BPPV were significantly higher than those in healthy controls. Otoconin-90 blood levels showed a high positive correlation with age, reflecting the process of otoconia degradation with age (24). All these studies suggested that otolin-1, an otoconia marker, has great clinical application prospects.
BPPV is a benign disease with a high-resolution rate, although it is prone to relapse, which leads to anxiety and poor quality of life in patients and increases the risk of falling due to imbalance (25, 26). Several studies have attempted to clarify relationships between age, diabetes, hypertension, delayed BPPV treatment using CRM, multiple canal involvement, osteoporosis, vitamin D deficiency, and the recurrence of BPPV, but the risk factor-associated recurrence remains elusive (4, 6, 7). Therefore, studying the risk factors for the recurrence of BPPV is imperative for better relapse prevention. The inner ear-specific protein, otolin-1, served as a candidate biomarker in circulation for the diagnosis of BPPV, but there have been no relevant studies investigating whether the serum level of otolin-1 can predict BPPV recurrence events.
Previously, we conducted a study and reported that the serum levels of otolin-1 in active episodes of patients with BPPV were significantly higher than those in healthy controls (324.55 vs. 259.54 pg/ml, p < 0.001), and ROC analysis showed that serum otolin-1 cut off value of 299.45 pg/ml could discriminate patients with BPPV from healthy controls with a sensitivity of 67.9% and a specificity of 72.7% (12). During the two-year follow-up period, 27 (36.5%) patients experienced recurrence after initial CRM treatment, which was consistent with previous studies that reported the recurrence rate (27). We found that the levels of otolin-1 in patients with BPPV relapse were significantly higher than those in patients without relapse, and high levels of otolin-1 were shown to be a risk factor for the recurrence of BPPV. These findings suggest that otolin-1 may also serve as a biomarker for BPPV recurrence and has some clinical value in the prediction of recurrence in BPPV patients.
This study has some limitations. First, it was a small-scale, single-center, observational study with a relatively short follow-up time, and the association between otolin-1 levels in circulation and recurrence of BPPV was only suggested. Second, we measured the serum levels of otolin-1 at only one time point in patients who initially presented with symptoms at the time of recruitment to the study. The last is also the primary limitation of the research. To date, there is no information on otolin-1 temporal evolution during the metabolic process of otoconia, as we cannot obtain inner ear tissues in real time and lack a proper animal model. Therefore, research on otolin-1 as a clinical biomarker for BPPV is still in the preliminary stages. Many studies should be conducted to clarify the metabolic process of otolith matrix proteins, which could provide experimental support for the possibility of utilizing otolin-1 as a biomarker for BPPV.
Conclusion
Elevated serum levels of otolin-1 were associated with an increased risk of recurrent BPPV, but additional work is needed to establish its value and clarify the exact mechanism.
Data Availability Statement
The datasets analyzed in this article are available upon request to:d3VfeXVucWluQDEyNi5jb20=.
Ethics Statement
The studies involving human participants were reviewed and approved by Hwa Mei Hospital, University of Chinese Academy of Science review board (Protocol Number KY-2017-014). The patients/participants provided their written informed consent to participate in this study.
Author Contributions
ZF, ZH, WH, XLu, XLi, MZ, and WY included, followed, and recorded patients data. ZF and ZH checked medical records and wrote the article. YW conceived and led the work. All authors contributed to the article and approved the submitted version.
Funding
This study was supported by Ningbo Medical Key Discipline (Grant No. B12), Ningbo Natural Science Foundation (Grant No. 202003N4240), and Hwa Mei Foundation (Grant No. 2021HMKY 30).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fneur.2022.841677/full#supplementary-material
References
1. Editorial Board of Chinese Journal of Otorhinolaryngology Head and Neck Surgery. Guideline of diagnosis and treatment of benign paroxysmal positional vertigo (in Chinese). Chin J Otorhinolaryngol Head Neck Surg. (2017) 52:173–7. doi: 10.3760/cma.j.issn.1673-0860.2017.03.003
2. Kim HJ, Park J, Kim JS, Kim HJ. Update on benign paroxysmal positional vertigo. J Neurol. (2021) 268:1995–2000. doi: 10.1007/s00415-020-10314-7
3. Mandalà M, Salerni L, Nuti D., Mandalà M. Benign paroxysmal positional vertigo treatment: a practical update. Curr Treat Options Neurol. (2019) 21:66. doi: 10.1007/s11940-019-0606-x
4. Chen J, Zhang S, Cui K, Liu C. Chen J. Risk factors for benign paroxysmal positional vertigo recurrence:a systematic review and meta-analysis. J Neuro. (2021) 268:4117–27. doi: 10.1007/s00415-020-10175-0
5. Lindell E, Kollén L, Johansson M, Karlsson T, Rydén L, Falk Erhag H, et al. Benign paroxysmal positional vertigo, dizziness, and health-related quality of life among older adults in a population-based setting. Eur Arch Otorhinolaryngol. (2021) 278:1637–44. doi: 10.1007/s00405-020-06357-1
6. Sfakianaki I, Binos P, Karkos P, Dimas GG, Psillas G. Risk factors for recurrence of benign paroxysmal positional vertigo. A clinical review. J Clin Med. (2021) 10:4372. doi: 10.3390/jcm10194372
7. Zhu CT, Zhao XQ, Ju Y, Wang Y, Chen MM, Cui Y. Clinical characteristics and risk factors for the recurrence of benign paroxysmal positional vertigo. Front. Neurol. (2019) 10:1190. doi: 10.3389/fneur.2019.01190
8. Hołubowicz R, Ozyhar A, Dobryszycki P., Hołubowicz R, et al. Molecular mechanism of calcium induced trimerization of C1q-like domain of otolin-1 from human and zebrafish. Sci Rep. (2021) 11:12778. doi: 10.1038/s41598-021-92129-8
9. Hołubowicz R, Wojtas M, Taube M, Kozak M, Ozyhar A, Dobryszycki P. Effect of calcium ions on structure and stability of the C1q-like domain of otolin-1 from human and zebrafish. FEBS J. (2017) 284:4278–97. doi: 10.1111/febs.14308
10. Mulry E, Parham K. Inner ear proteins as potential biomarkers. Otol Neurotol. (2020) 41:145–52. doi: 10.1097/MAO.0000000000002466
11. Parham K, Sacks D, Bixby C, Fall P. Inner ear protein as a biomarker in circulation? Otolaryngol Head Neck Surg. (2014) 151:1038–40. doi: 10.1177/0194599814551127
12. Wu Y, Han W, Yan W, Lu X, Zhou M, Li L, et al. Increased otolin-1 in serum as a potential biomarker for idiopathic benign paroxysmal positional vertigo episodes. Front Neurol. (2020) 11:367. doi: 10.3389/fneur.2020.00367
13. Irugu DVK, Singh A, Yadav H, Verma H, Kumar R, Abraham RA, et al. Serum otolin-1 as a biomarker for benign paroxysmal positional vertigo: a case-control study. J Laryngol Otol. (2021) 135:589–92. doi: 10.1017/S0022215121001274
14. Yadav H, Irugu D, Ramakrishanan L, Singh A, Abraham R, Sikka K, et al. An evaluation of serum otolin-1 and vitamin-D in benign paroxysmal positional vertigo. J Vestib Res. (2021) 31:433–40. doi: 10.3233/VES-201601
15. Tabtabai R, Haynes L, Kuchel GA, Parham K. Age-related increase in blood levels of Otolin-1 in humans. Otol Neurotol. (2017) 38:865–9 doi: 10.1097/MAO.0000000000001426
16. Jang YS, Hwang CH, Shin JY, Bae WY, Kim LS. Age-related changes on the morphology of the otoconia. Laryngoscope. (2006) 116:996–1001. doi: 10.1097/01.mlg.0000217238.84401.03
17. Moreland KT, Hong M, Lu W, Rowley CW, Ornitz DM, De Yoreo JJ, et al. In vitro calcite crystal morphology is modulated by otoconial proteins otolin-1 and otoconin-90. PLoS ONE. (2014) 9:e95333. doi: 10.1371/journal.pone.0095333
18. Andrade LR, Lins U, Farina M, Kachar B, Thalmann R. Immunogold TEM of otoconin 90 and otolin−1 relevance to mineralization of otoconia, and pathogenesis of benign positional vertigo. Hear Res. (2012) 292:14–25 doi: 10.1016/j.heares.2012.07.003
19. Lunderg YW, Xu Y, Thiessen KD, Kramer KL. Mechanisms of otoconia and otolith development. Dev Dyn. (2015) 244:239–53. doi: 10.1002/dvdy.24195
20. Zucca G, Valli S, Valli P, Perin P, Mira E. Why do benign paroxysmal positional vertigo episodes recover spontaneously? J Vestib Res. (1998) 8:325–9. doi: 10.3233/VES-1998-8404
21. Hołubowicz R, Ozyhar A, Dobryszycki P. Natural mutations affect structure and function of gC1q domain of otolin-1. Int J Mol Sci. (2021) 22:9085. doi: 10.3390/ijms22169085
22. Dogan M, Sahin M, Kurtulmuş Y. Otolin-1, as a potential marker for inner ear trauma after mastoidectomy. J Int Adv Otol. (2019) 15:200–3. doi: 10.5152/iao.2019.5155
23. Naples JG, Soda Drew, Rahman K, Ruckenstein MJ, Parham K. Evaluating the role of otologic biomarkers to differentiate Meniere's disease and vestibular migraine. Ear Hear. (2021). doi: 10.1097/AUD.0000000000001123. [Epub ahead of print].
24. Bi J, Liu B, Zhang Y, Zhou Q. Study on the bone metabolism indices and otoconin- 90 in benign paroxysmal positional vertigo. Otol Neurotol. (2021) 42:e744–9. doi: 10.1097/MAO.0000000000003087
25. Gunes A, Yuzbasioglu Y. Effects of treatment on anxiety levels among patients with benign paroxysmal positional vertigo. Eur Arch Otorhinolaryngol. (2019) 276:711–8. doi: 10.1007/s00405-019-05297-9
26. Jumani K, Powell J. Benign paroxysmal positional vertigo: management and its impact on falls. Ann Otol Rhinol Laryngol. (2017) 126:602–5. doi: 10.1177/0003489417718847
Keywords: benign paroxysmal positional vertigo, otolin-1, recurrence, canalith repositioning procedure, biomarker
Citation: Fan Z, Hu Z, Han W, Lu X, Liu X, Zhou M, Yan W and Wu Y (2022) High Serum Levels of Otolin-1 in Patients With Benign Paroxysmal Positional Vertigo Predict Recurrence. Front. Neurol. 13:841677. doi: 10.3389/fneur.2022.841677
Received: 22 December 2021; Accepted: 27 January 2022;
Published: 14 March 2022.
Edited by:
Michael Strupp, Ludwig Maximilian University of Munich, GermanyReviewed by:
Takeshi Tsutsumi, Tokyo Medical and Dental University, JapanJeremy Hornibrook, University of Canterbury, New Zealand
Copyright © 2022 Fan, Hu, Han, Lu, Liu, Zhou, Yan and Wu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yunqin Wu, d3VfeXVucWluQDEyNi5jb20=
†These authors have contributed equally to this work
 Zhenyi Fan
Zhenyi Fan Zhizhou Hu
Zhizhou Hu Weiwei Han
Weiwei Han Xiaoxiong Lu
Xiaoxiong Lu Xiaoxia Liu1,2
Xiaoxia Liu1,2 Yunqin Wu
Yunqin Wu