- 1Research Group Language Cycles, Max Planck Institute for Human Cognitive and Brain Sciences, Leipzig, Germany
- 2Clinic for Phoniatrics and Pedaudiology, University Hospital Münster, Münster, Germany
- 3Center for Biomedical Imaging and Neuromodulation, Nathan Kline Institute, Orangeburg, NY, United States
- 4Department of Psychiatry and Psychotherapy, Philipps-University Marburg, Marburg, Germany
Deficits in language production and comprehension are characteristic of schizophrenia. To date, it remains unclear whether these deficits arise from dysfunctional linguistic knowledge, or dysfunctional predictions derived from the linguistic context. Alternatively, the deficits could be a result of dysfunctional neural tracking of auditory information resulting in decreased auditory information fidelity and even distorted information. Here, we discuss possible ways for clinical neuroscientists to employ neural tracking methodology to independently characterize deficiencies on the auditory–sensory and abstract linguistic levels. This might lead to a mechanistic understanding of the deficits underlying language related disorder(s) in schizophrenia. We propose to combine naturalistic stimulation, measures of speech–brain synchronization, and computational modeling of abstract linguistic knowledge and predictions. These independent but likely interacting assessments may be exploited for an objective and differential diagnosis of schizophrenia, as well as a better understanding of the disorder on the functional level—illustrating the potential of neural tracking methodology as translational tool in a range of psychotic populations.
Introduction
Schizophrenia is characterized by language deficits ranging from lower acoustic and phonetic levels to higher semantic and syntactic levels that are highly functionally relevant (Bleuler, 1950; Chaika, 1990; DeLisi, 2001; Covington et al., 2005). Among others, key features of patients’ speech include flattened prosody, simplified syntax, and loosened semantic associations (for review, see Andreasen, 1979; Kircher et al., 2014). In comprehension, correspondingly, patients are impaired in the processing of linguistic information at these levels (Leitman et al., 2005; Mohammad and DeLisi, 2013; Javitt and Sweet, 2015; Moro et al., 2015). In addition, major symptoms of schizophrenia such as auditory hallucinations and delusions are thought to be closely related to impaired speech perception and language comprehension (Brown and Kuperberg, 2015). To date, the neuropathology of language impairments in schizophrenia remains unclear (Miller and Isard, 1963; Morice and Delahunty, 1996; Crow, 1998; DeLisi, 2001; Bagner et al., 2003; Li et al., 2009; Brown and Kuperberg, 2015; Hirano et al., 2020). In this paper, we suggest that the understanding of language deficits in schizophrenia could benefit from analyzing neural oscillations with neural tracking methodology. Oscillations can be aligned to speech and this alignment seems to be guided by attention, especially in “cocktail party” settings (e.g., Vander Ghinst et al., 2016; for review, see Lakatos et al., 2019a)1. Therefore, we propose that multi-scale (on phrasal/syllable temporal scales) oscillatory alignment provides a novel tool for assessing language dysfunctions on various linguistic levels.
Our Hypothesis and Theory article considers three main issues: In the first part, we discuss possible ways to address deficits in auditory perception and speech tracking as such. In the second part, we delineate probable relationships between prosodic–syntactic deficits and altered delta-band oscillations. In the third part, we hypothesize that impaired semantics in schizophrenia could result from altered beta–gamma coupling. In the last section, we discuss how to pursue these hypotheses by combining naturalistic experimental paradigms with methodology that assesses the exogenous neural tracking of auditory–phonetic information and the endogenous generation of abstract linguistic information.
Auditory Perception and Speech Tracking: Impaired Theta-Band Oscillations
Deficits in auditory processing could underlie a range of language-related symptoms in schizophrenia (Javitt and Freedman, 2015). Such deficits do not include hearing per se (McKay et al., 2000; Javitt, 2009), as detection thresholds, sensitivity to loudness, spatial localization, and the P1/N1 complex in the event-related brain potential (ERP) for isolated sounds are all intact (Javitt and Freedman, 2015). Yet, patients show reduced auditory mismatch negativities (MMN; for review, see Näätänen and Kähkönen, 2008; Todd et al., 2013; Michie et al., 2016) to both speech and non-speech sounds (Kasai et al., 2002, 2003). In addition, P1/N1 difference waves are altered under repetition priming and sensory gating (Freedman et al., 1987; Adler et al., 1998; Patterson et al., 2008), and for perception of words differing in lexicality (Hirano et al., 2008). Moreover, the N1 difference between self-produced and presented auditory stimuli has been found abnormal in experiments investigating potential deficits in corollary discharge and efference copy during auditory speech processing (Ford et al., 2001, 2007a, b). Apart from the MMN, P1, and N1 components, a reduction of the P300 in oddball paradigms is a robust neural marker of schizophrenia (Ford et al., 1994, 2008; Higashima et al., 2003). In sum, despite the fact that isolated sounds appear to be processed normally in schizophrenia, earlier behavioral and ERP evidence suggests that patients with schizophrenia are impaired in a range of auditory processes—whenever patient’s perception of tones or speech involves top-down influences—thus suggesting predictive coding rather than audition impairments in schizophrenia (Adams et al., 2013; Sterzer et al., 2018; Howes et al., 2020; Smith et al., 2021).
In spite of these rather robust results, the altered difference ERPs calculated from stimulus train of simple tones or speech sounds (i.e., MMN and P300) are hard to dissociate from altered oscillatory activity due to the fact that oscillatory phase reset contributes heavily to ERPs (Klimesch et al., 2007; Ding and Simon, 2014; Obleser and Kayser, 2019; Haegens, 2020). It has been argued that the MMN represents a mainly theta-band phase reset that occurs in the extragranular layers of the auditory cortex; moreover, the MMN is vulnerable to blockage of the N-Methyl-D-Aspartate Receptor (Lakatos et al., 2019b). NMDA receptor related deficits are hypothesized to underlie a range of symptoms as well as auditory deficits in schizophrenia (Kort et al., 2017; Corlett et al., 2018; Javitt et al., 2020). In addition, altered P300 responses in schizophrenia were observed in parallel with altered theta-band oscillations (Ford et al., 2008). In sum, this initial evidence leaves it open whether aberrant theta oscillations are impaired independently of evoked responses as shown in the ERPs, and thus they both might contribute to auditory and language deficits in schizophrenia.
Here, we propose that neural tracking methodology that employs naturalistics paradigms should allow researchers and clinicians to better focus on theta-band oscillations without the confounding ERP alterations elicited by controlled experiments. In particular, this contrasts with oddball experiments and the associated MMN and P300 alterations. Theta-band oscillations are thought to phase-lock to the acoustic edges of syllables, aiding their segmentation or even identification (Luo and Poeppel, 2007; Howard and Poeppel, 2012; Gross et al., 2013; Peelle et al., 2013; Doelling et al., 2014). Furthermore, processing of phonemes in context (e.g., labeling of phonetic features, predicting the likelihood of upcoming phonemes) can be investigated together with envelope tracking with state-of-the-art multivariate analysis techniques, by close examination of low-frequency oscillations including the theta band (e.g., Di Liberto et al., 2015, 2019; Daube et al., 2019) and the delta band (see below). To date, however, only few recent studies have reported altered theta-band power and phase aberrence in schizophrenia, which used simple tones delivered as part of traditional oddball and gating paradigms (Lakatos et al., 2013; Kantrowitz et al., 2016; Lee et al., 2017). Thus, investigating potential impairments of theta-band oscillations during neural tracking of naturalistic speech could contribute to extant research, and could provide an unique window into understanding the neuropathology of language deficits in schizophrenia. Further potential of the combined use of computational modeling and neural tracking methodology to dissociate acoustic, phonological, and linguistic processing is provided below.
Prosody and Syntax: Abnormal Delta-Band Oscillations?
Auditory processing deficits in schizophrenia are considered as reflecting “up-stream” functions, such as prosody (Javitt, 2009). Flattened prosody in production is a negative symptom of schizophrenia, characterized by reduced modulation of fundamental frequency and amplitude, utterances that are shortened and less variable in duration, and pauses that are longer and more variable (Alpert et al., 1989, 2000; Covington et al., 2005). Such language production related indices can classify schizophrenia incidence with high accuracy (Püschel et al., 1998; Rapcan et al., 2010; Martínez-Sánchez et al., 2015) and may help to detect risk (Cibelli et al., 2017). In comprehension, patients struggle to infer emotions and communicative intentions from prosody (e.g., Pawełczyk et al., 2018a). While this is sometimes discussed as epiphenomenal to impaired emotion reception (Murphy and Cutting, 1990; Mitchell and Crow, 2005; Hoekert et al., 2007; Lin et al., 2018), flattened prosody occurs also when emotional vocabulary is intact (Alpert et al., 2000). Moreover, auditory deficits in prosody perception predict most variance associated with impaired comprehension of emotional prosody (Leitman et al., 2005; Dondé et al., 2017), and the inference of emotion is improved by prosody training (Lado-Codesido et al., 2019).
In addition to prosody, syntactic impairments have been observed. Syntactic rules serve to decode the propositional relationships amongst words in speech (cf. Martin, 2020). Patients with schizophrenia do not reliably detect syntactic errors (Moro et al., 2015), and their working memory benefits less from syntactic structure (i.e., no sentence superiority effect; Bonhage et al., 2017; Li et al., 2018). Alternatively, syntactic rules might be intact, but their top-down influence on perception is temporally distorted (Rochester et al., 1973). In healthy populations, syntactic boundaries (e.g., clause endings) influence perception, such that acoustic events that are experimentally displaced from a boundary are perceptually “dragged toward it” (Fodor and Bever, 1965). This effect appears to be altered in patients (Rochester et al., 1973). A temporal deficit would be also consistent with reports of turn-taking deficits (Sichlinger et al., 2019). Healthy speakers tend to indicate turn-giving with prosodic markings (Levinson, 2016), but patients often fail to do so (Bellani et al., 2009; Colle et al., 2013; Pawełczyk et al., 2018b). In particular, the timing of turn-giving is affected, such that variance in utterance duration decreases and variance in pause duration increases (Alpert et al., 2000).
We propose that the respective neural counterparts of prosodic and syntactic symptoms could be assessed by focusing on delta-band oscillations (i.e., 0.5–4 Hz; Buzsaki, 2006; Güntekin and Başar, 2016; Figure 1), as the time scale of these neural oscillations can be clearly linked to speech structure (Giraud and Poeppel, 2012; Ding et al., 2016). While delta-band oscillations are certainly altered in schizophrenia (for review, see Başar and Güntekin, 2008; Ford et al., 2008; Doege et al., 2010; Lakatos et al., 2013), there is no unitary link with the above symptoms yet. We propose that establishing such a link would support differential diagnosis of the underlying disorder, and it could also contribute to the ongoing struggle for a dissociation of the functional roles of delta-band oscillations in prosody, syntax, and timing (Lakatos et al., 2008; Ghitza, 2017; Meyer et al., 2017, 2019).
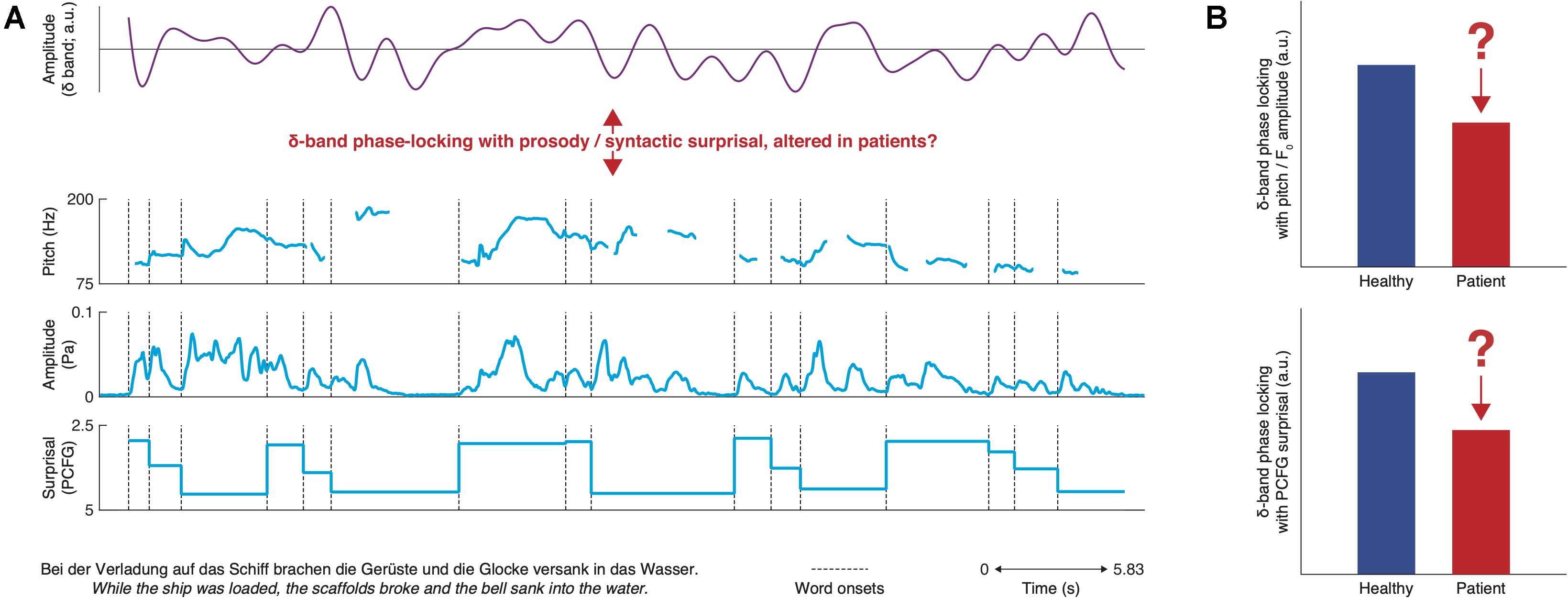
Figure 1. Overview of hypotheses for prosodic and syntactic deficits. (A) top: synthetic delta-band oscillation; bottom: frequency– and amplitude modulations corresponding to the pitch track as well as computational-linguistic measures of the application of syntactic rules for the example sentence at the bottom. It is hypothesized that delta-band phase-locking is impaired in patients with schizophrenia and that depending on the underlying disorder, this abnormality could be restricted to either prosody or syntax. For demonstration, a strongly phase-locked delta-band oscillation was generated via a randomization procedure. (B) Hypotheses as bar charts: depending on the underlying deficit, either phase-locking to prosody or syntax should be impaired in patients.
On the one hand, in healthy subjects, delta-band oscillations synchronize with prosody (Bourguignon et al., 2013; Gross et al., 2013; Mai et al., 2016; Molinaro et al., 2016), the perception of which is impaired in schizophrenia (e.g., Dondé et al., 2017). On the other hand, delta-band frequencies match the rate of occurrence of syntactic phrases and sentences (Ding et al., 2016) and delta-band phases are aligned to syntactic structure (Brennan and Martin, 2020) and information content (Meyer and Gumbert, 2018), independently to prosody (Meyer et al., 2017). Healthy subjects show increased delta-band power during working memory encoding of syntactically structured relative to unstructured word sequences (Bonhage et al., 2017). In contrast, working memory encoding in schizophrenia patients does not benefit much from syntactic structure (Li et al., 2018).
Instead of prosodic and syntactic deficits as such, abnormal delta-band oscillations could also indicate an underlying timing deficit. Lakatos et al. (2013) observed reduced delta-band phase alignment (measured by inter-trial phase coherence) in patients across the isochronous trials of an auditory oddball experiment. The authors interpret this as indicating deficient temporal prediction, mediated by the alignment of oscillatory brain activity to external stimulus timing (i.e., oscillatory entrainment). They also demonstrate that the lack of phase alignment is associated with reduced behavioral performance and correlates with clinical symptoms. This interpretation of their results stems from prior work related to the role of delta-band oscillations in temporal prediction (Lakatos et al., 2008; Stefanics et al., 2010; Arnal et al., 2014; Breska and Deouell, 2017; Jones et al., 2017; Rimmele et al., 2018; Donhauser and Baillet, 2020). Specifically, Stefanics et al. (2010) observed enhanced auditory target detection during specific phase intervals that were elicited through prior rhythmic stimulation (cf. Henry and Obleser, 2012; Hickok et al., 2015). Delta-band phase is an imprint of the neuronal excitability of auditory regions (e.g., Lakatos et al., 2008). Delta-band oscillations could thus likely serve prediction by preallocating excitability and functional connectivity within relevant brain circuits to the expected onsets of upcoming stimuli (e.g., Lakatos et al., 2008, 2009). Our recent results speak in favor of a link between the involvement of the delta band in syntactic processing and its involvement in temporal prediction. We found that the delta phase is not just generally correlated with syntax, but it is more specifically correlated with the strength of syntactic predictions (Hale, 2001; Levy, 2008; Meyer and Gumbert, 2018).
Semantics: Impaired Predictive Coding in the Beta- and Gamma-Bands?
Semantic impairments in schizophrenia are less controversial than prosodic and syntactic impairments. Patients commonly display hyperactivation of lexical-semantic associations. While healthy individuals associate lion with tiger but not with stripes, patients with schizophrenia may do so. Accordingly, patients produce words that are less directly related to their intended message (Bleuler, 1950). For example, they might complain about their chest pain by saying I wonder if my box is broken (Chaika, 1990). Correspondingly in comprehension, patients show enhanced semantic priming effects (Spitzer et al., 1994; Weisbrod et al., 1998; Kreher et al., 2009). However, depending on the task under study, patients may also exhibit a more restricted semantic network than healthy subjects during comprehension (Kreher et al., 2009). The comprehension deficits manifest beyond the word level, that is, real-world objects and events are commonly associated with special and negative meaning, a defining feature of delusions.
Most electrophysiological literature on semantic comprehension deficits in schizophrenia has exploited the N400 component of the evoked response (ERP), typically manipulating the semantic/discourse fit between a target word and its preceding word/sentence context (Kutas and Hillyard, 1980; Hagoort et al., 2004; Nieuwland and Van Berkum, 2006; Lau et al., 2008; Kutas and Federmeier, 2011). These studies have offered valuable insights into how word– and sentence-level semantics are disrupted vs. preserved in schizophrenia (Mohammad and DeLisi, 2013; Kiang and Gerritsen, 2019).
Word-level semantic processing in schizophrenia is most commonly investigated via priming paradigms. Yet, the literature is inconsistent in terms of whether the priming-N400 effects are enhanced or reduced in patients (Mathalon et al., 2002, 2010; Salisbury, 2008; Kuperberg et al., 2019; Sharpe et al., 2020). This discrepancy may result from impairments at different levels of the linguistic hierarchy. Patients may be impaired in lexical access (Kuperberg et al., 2019), may suffer from reduced or enhanced semantic activation (Titone et al., 2000; Mathalon et al., 2010), or may fail to derive predictions from the word context (Sharpe et al., 2020). Of note, the prediction failure account accords with results from sentence-level N400 studies: Whereas the N400 reflecting semantic retrieval and integration seems to be unaffected in schizophrenia (Kuperberg et al., 2006), converging evidence has shown that patients are unable to utilize contextual information to suppress irrelevant meanings of a target word, for example, when comprehending a homophone (Sitnikova et al., 2002). Hence, it has been proposed that semantic deficits in schizophrenia may originate from a general inability to integrate and update predictions of higher linguistic levels (e.g., context) with lower-level semantic inputs (Brown and Kuperberg, 2015). However, as it remains unclear whether the N400 indexes prediction, prediction error, or a combination of both (Kutas and Federmeier, 2011; Bornkessel-Schlesewsky and Schlesewsky, 2019; He et al., 2020; Kuperberg et al., 2020; Nieuwland et al., 2020), it also remains unresolved whether linguistic prediction or prediction error underlies semantic impairments in schizophrenia.
With the equivocal interpretation of N400 alterations in mind, we propose to investigate semantic deficits by examining neural oscillations in the beta– and gamma-bands (for a possible relationship between the N400 and delta-band oscillations, see Roehm et al., 2007). The maintenance of semantic top-down predictions has been associated with beta-band power, whereas gamma-band power reflects the integration with bottom-up semantic input (Lewis and Bastiaansen, 2015; Lewis et al., 2015; Meyer, 2017). In healthy populations, at the semantic level, the sensitivity of beta–gamma-band power has been reported in a series of studies (Hagoort et al., 2004; Wang et al., 2012a, b, 2018; Kielar et al., 2014, 2015). Notably, as the majority of these studies have leveraged the classic semantic violation paradigm, despite a theoretical dissociation, it remains controversial how beta– and gamma-bands map to prediction or prediction error during sentence-level processing (for review, see Prystauka and Lewis, 2019). We thus suggest a naturalistic approach (Figure 2), allowing for the dissociation of prediction and error at the single-word level, as well as an independent comparison between healthy and clinical groups. In healthy participants, beta-band power decreases for more precise prediction; for decreased error, gamma-band power increases accordingly. In addition, when predicted and incoming information match, cross-frequency coupling between the beta and gamma bands would increase (Roopun et al., 2008; e.g., Engel and Fries, 2010; Chao et al., 2018). The beta–gamma interplay offers a promising candidate mechanism that bridges predictive and integrative semantic processes. It also forms the basis for a plausible unifying theory linking predictive deficits in schizophrenia across functional domains outside of language. In the sensory domain, our previous work has shown that gamma power is less modulated for schizophrenia in response to prediction error on the acoustic level (Lakatos et al., 2013). In a similar vein, effects for predictive beta modulation in schizophrenia has been reported when patients are engaged in social interactive games (Billeke et al., 2015). Oscillations across frequency ranges appear to be coupled (Lakatos et al., 2005; Canolty et al., 2006; Canolty and Knight, 2010). Thus, it is worthwhile to investigate the cross-frequency dynamics (e.g., phase–amplitude coupling) in schizophrenia (Kirihara et al., 2012; Hirano et al., 2018) during language processing and speech perception to examine, for example, if impaired beta–gamma oscillations will impact delta oscillatory tracking and vice versa.
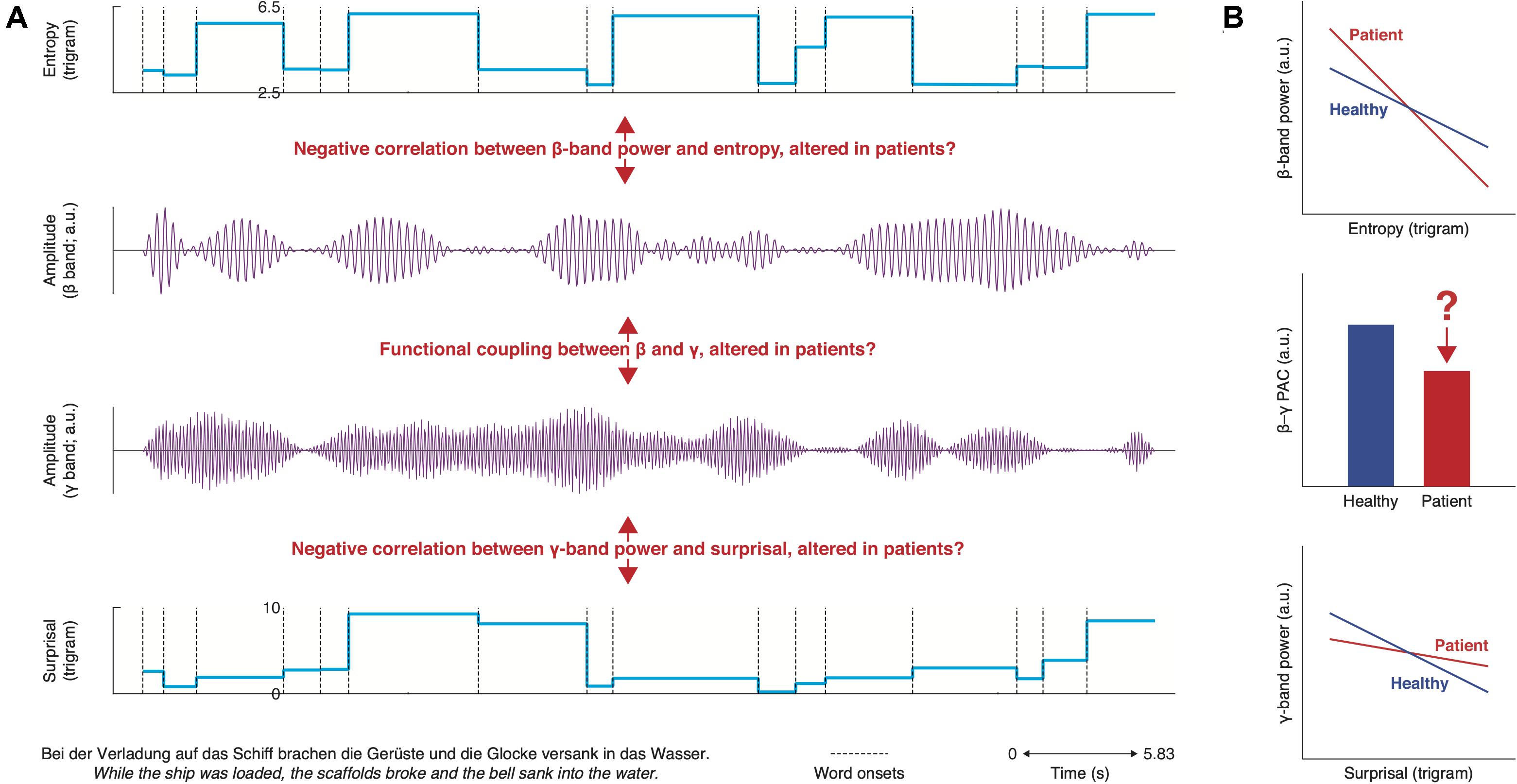
Figure 2. Overview of hypotheses for semantic deficits. (A) top: word-by-word entropy time course for example sentence (bottom left); middle: synthetic beta– and gamma-band time courses; bottom: word-by-word surprisal time course for example sentence (bottom left). We hypothesize that the relationship between beta-band power and entropy as well as between gamma-band power and surprisal is abnormal in schizophrenia patients. Additionally, phase–amplitude coupling of the beta and gamma band might be disturbed. These effects will likely differ amongst schizophrenia subgroups (e.g., hallucinators vs. non-hallucinators). (B) Summary of hypotheses.
A particular focus on dysfunctional beta– and gamma-band oscillations has the additional potential of providing a theoretical explanation of core symptoms of schizophrenia, such as auditory hallucinations and delusions, and on how these symptoms, in turn, impact upon sensory tracking and linguistic prediction. Impairments of prediction in schizophrenia can be nuanced (Sterzer et al., 2018): it has been proposed that auditory hallucinations may derive from overly precise (stronger) prediction (Corlett et al., 2018; Heinz et al., 2019), whereas delusions are related to imprecise (weaker) prediction (Stuke et al., 2018), even if both symptoms often co-occur. We propose that physiologically, hallucinations and delusions are perpetrated by stronger vs. weaker synchronization of brain activity correspondingly in certain frequency bands.
More importantly, both stronger and weaker predictions may occur at hierarchically different levels of sensory and higher cognitive processes in schizophrenia, and may be subject to interaction across levels (Horga et al., 2014; Teufel et al., 2015; Alderson-Day et al., 2017; Powers et al., 2017), indicating the importance of changes in functional connectivity. In the language domain, word-level priming N400 deficits is proposed to be related to delusion severity, thus may support impaired semantic prediction (Kiang and Gerritsen, 2019). In relation to neural oscillations, it has been reported that prestimulus beta-band phase is inversely related to hallucination severity when patients produce speech and listen to the speech sound that they have produced (Ford et al., 2007b). Although the authors did not report power modulation, this study might be an indication of potential link between auditory hallucinations and the beta-band phase in terms of aberrant prediction across comprehension and production of speech (Wang et al., 2012a; Piai et al., 2014; Lewis et al., 2015). Moreover, the most replicated oscillatory correlate of auditory hallucinations is reported in the literature investigating auditory steady-state responses: Gamma-band (usually 40Hz) power and inter-trial phase coherence has been shown to correlate well with hallucination severity (Spencer et al., 2008; Mulert et al., 2011). Notably, steady-state responses reflect a mixture of stimulus-specific evoked responses and the resonant response of the sensory cortices. Therefore, they may not be interpreted on a par with endogenous gamma oscillations (Duecker et al., 2020). However, the strong correlation between the gamma-band responses and auditory hallucinations, together with the reported beta alterations, suggest that both frequency bands are valuable candidates of evaluating dysfunctional predictive coding from a phenomenological perspective. Apart from semantic processing, recent studies employing naturalistic approaches suggest that auditory processing in the form of speech tracking or phonemic prediction—as reflected by low-frequency oscillations in the theta and delta bands—may be subject to top-down influence such as semantic or contextual prediction (Broderick et al., 2019; Heilbron et al., 2020). These emerging studies are prime examples of how naturalistic approaches might directly contribute to the underlying neuropathology of auditory hallucinations in schizophrenia: Instead of observing generally modulated semantic prediction (beta) and auditory tracking (theta) for non-hallucinating patients, we propose to investigate how these processes are enhanced or reduced in hallucinating patients, and how are the functional coupling between beta-theta bands altered in hallucinations.
Toward Naturalistic Experiments for Schizophrenia Research
Most electrophysiological studies on language deficits in schizophrenia employed controlled factorial designs that used isolated sentences or word pairs. These studies have provided valuable insights into the neuropathology of schizophrenia, but face limitations. First, repetitive experimental procedures limit ecological validity (Brennan, 2016; Willems et al., 2016; Hamilton and Huth, 2018; Hasson et al., 2018; Kandylaki and Bornkessel-Schlesewsky, 2019; Shamay-Tsoory and Mendelsohn, 2019). Second, the typical isochronous presentation of words and sentences (e.g., oddball paradigms, rapid serial visual presentation, RSVP) triggers sequences of evoked responses that have the potential to mask oscillatory activity (Meyer et al., 2020; Poeppel and Teng, 2020); note that this advantage only pertains to those evoked components that are genuine to oddball designs (e.g., P300, see above) and RSVP designs (e.g., repetitive visual onset responses). Third, factorial subtraction approach (e.g., standard – deviant, congruent – incongruent) does not allow straightforward dissociations between acoustic–phonetic and abstract linguistic processes (e.g., Nieuwland et al., 2020). Finally, it is difficult to measure interactions across linguistic levels with factorial approaches (Brown and Kuperberg, 2015; Sterzer et al., 2018).
We thus propose to address language deficits in schizophrenia with naturalistic experiments using ecologically-valid language stimuli (Hamilton and Huth, 2018; Kandylaki and Bornkessel-Schlesewsky, 2019). In such naturalistic experiments, participants are presented with entire narratives (e.g., Stehwien et al., 2020). This enhances feasibility under the temporal and monetary constraints of clinical research while still increasing statistical power and flexibility beyond factorial designs. Narratives also allow the analysis of neural tracking of acoustic and phonetic modulations at the sampling rate of the electrophysiological recording or phonetic–phonological annotation (e.g., Gross et al., 2013; Bastos and Schoffelen, 2015; Di Liberto et al., 2015; Daube et al., 2019). This in turn allows researchers to directly address the above hypothesis on dysfunctional theta-band tracking that we presented in our first scenario above. In parallel, multiple levels of word-by-word/phrase-by-phrase linguistic processing can be analyzed through domain-specific metrics derived by computational-linguistic modeling (e.g., Hale, 2001, 2016; Levy, 2008; Frank et al., 2015; Brennan, 2016). Emerging studies have approached naturalistic story comprehension to investigate language processing in healthy aging (Broderick et al., 2020; Cuevas et al., 2020). In schizophrenia research, naturalistic experiments were proposed for the study of social dysfunctions (Leong and Schilbach, 2019; Brandi et al., 2020). In the language domain, an eye-tracking study using a visual-world paradigm has looked into the impact of higher-level discourse on ambiguity resolution (Rabagliati et al., 2019). We have recently investigated patient’s processing of multimodal stories (i.e., auditory story, manual gestures) using functional magnetic resonance imaging, showing that manual gestures can enhance patients’ reduced semantic activation in a left fronto-temporal network (Cuevas et al., 2019; Cuevas et al., in preparation).
For the second scenario outlined above, the investigation of impaired syntactic and prosodic processing in schizophrenia, a naturalistic approach would allow for a dissociation of the previously proposed prosodic and syntactic deficits. To approximate prosody, the audio stimulus would be low-pass filtered to yield those frequency modulations that correspond to pitch changes (Meyer et al., 2017; Meyer and Gumbert, 2018). Alternatively, the speech envelope could be low-pass filtered, yielding pitch amplitude modulations (Bourguignon et al., 2013; e.g., Gross et al., 2013; Mai et al., 2016). To concurrently model the application of syntactic rules, computational-linguistic modeling would be employed. In electrophysiological research on healthy populations, parsers that operationalize probabilistic context-free grammars are used frequently (e.g., Roark et al., 2009; Frank et al., 2015; Meyer and Gumbert, 2018; Vassileiou et al., 2018). Such algorithms are trained on large corpora annotated with part-of-speech labels and syntactic structures, enabling subsequent annotation of the narrative used for stimulation. Information theory is then applied to quantify syntactic processing difficulty (Shannon, 1948; Hale, 2001, 2016). The prosodic and syntactic regressors would then be related statistically to the electrophysiological data. For prosody, this could be achieved using a variant of speech–brain coupling methodology (for review, see Bastos and Schoffelen, 2015; Poeppel and Teng, 2020). For syntax, time-resolved multiple regression (Sassenhagen, 2019) or multivariate temporal response functions (mTRF, Crosse et al., 2016) could be used, alternatively also allowing to include both prosody and syntax within a single statistical model.
At the semantic level, as sentences unfold in a word-by-word manner, making semantic predictions of a word based on its prior context is equal to having some degree of certainty about the future (i.e., predictive coding). When instead an improbable event occurs, the prediction turns out to be an error. In computational approaches, the probability at which a listener is able to predict the meaning of the next word of the narrative from the preceding passage is measured by word-level entropy, and the corresponding prediction error when encountered a word is parameterized as word-level surprisal. Essentially, both indices can be derived from the conditional probability of word forms as calculated by standard toolboxes (Stolcke, 2002; Roark et al., 2009; Frank et al., 2015; Willems et al., 2016). Word-level entropy and surprisal can then be regressed against power of band-pass filtered continuous EEG in the beta– and gamma-bands via time-resolved multiple regression or the mTRF (Crosse et al., 2016; Ehinger and Dimigen, 2019; Sassenhagen, 2019). Individual beta coefficients would be then directly compared between patients and healthy controls, revealing if semantic prediction or prediction error are impaired in schizophrenia. In addition, the impact of major schizophrenia symptoms (e.g., auditory hallucinations) on semantic-level predictive coding could be evaluated via a comparison between patients with or without auditory hallucinations.
Importantly, group differences in the respective correlations between entropy/surprisal and beta–gamma-band power would readily define candidate frequencies and time windows to address the hypothesis of abnormal phase–amplitude coupling between the beta and gamma bands in schizophrenia (e.g., Bastos and Schoffelen, 2015; Hyafil et al., 2015). While translational application of the naturalistic approach has been initially employed in autism research (Brennan et al., 2018), its value for schizophrenia research awaits validation.
Conclusion
We have outlined the potential of studying neural tracking in the functional characterization of linguistic deficits in schizophrenia. In our view, two threads should be followed: First, deficient theta-band tracking of syllables should be assessed as part of the “routine ERP analyses” in schizophrenia. While the exact mechanisms of theta-band tracking are still being debated, it is clear that it reflects both bottom-up and top-down mechanisms that might be altered in patients. Second, the previously proposed relationship between delta-band oscillations, prosody, syntax, and temporal prediction may help to study the corresponding deficits in schizophrenia in a hypothesis-driven manner, with the potential to dissociate underlying electrophysiological dysfunction(s). Third, the general role of the beta–gamma interplay in the generation and evaluation of predictions may be fruitful in elucidating the electrophysiological dysfunction(s) that correspond to contextual–semantic symptoms. While both threads connect well with the literature, the direct link between frequency bands and linguistic dysfunctions may be overly simplistic. While often neural oscillations are assigned to specific functions or oscillatory deficits are linked to specific deficits, we believe that since these are coupled across both spatial and temporal scales, they should be evaluated in unison in relation to the naturalistic paradigms we propose.
Data Availability Statement
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author/s.
Author Contributions
LM, PL, and YH wrote the manuscript. All authors contributed to the article and approved the submitted version.
Funding
LM was supported by the Max Planck Research Group Language Cycles. PL was supported by NIH grants R01DC012947, R01MH109289, and P50MH109429. YH was supported by the Deutsche Forschungsgemeinschaft grant HE8029/2-1.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
- ^ Note that our manuscript is restricted to hypotheses on auditory processes genuine to speech processing, as well as on higher-level linguistic processes. The independent modulation of these processes by attention and working memory, both of which are impaired in schizophrenia (Braff, 1993; Carter et al., 1998), is beyond the scope of the current article.
References
Adams, R. A., Stephan, K. E., Brown, H. R., Frith, C. D., and Friston, K. J. (2013). The computational anatomy of psychosis. Front. Psychiatry 4:47. doi: 10.3389/fpsyt.2013.00047
Adler, L. E., Olincy, A., Waldo, M., Harris, J. G., Griffith, J., Stevens, K., et al. (1998). Schizophrenia, sensory gating, and nicotinic receptors. Schizophr. Bull. 24, 189–202. doi: 10.1093/oxfordjournals.schbul.a033320
Alderson-Day, B., Lima, C. F., Evans, S., Krishnan, S., Shanmugalingam, P., Fernyhough, C., et al. (2017). Distinct processing of ambiguous speech in people with non-clinical auditory verbal hallucinations. Brain 140, 2475–2489. doi: 10.1093/brain/awx206
Alpert, M., Rosen, A., Welkowitz, J., Sobin, C., and Borod, J. C. (1989). Vocal acoustic correlates of flat affect in schizophrenia: similarity to Parkinson’s disease and right hemisphere disease and contrast with depression. Br. J. Psychiatry 154, 51–56. doi: 10.1192/S0007125000295780
Alpert, M., Rosenberg, S. D., Pouget, E. R., and Shaw, R. J. (2000). Prosody and lexical accuracy in flat affect schizophrenia. Psychiatry Res. 97, 107–118. doi: 10.1016/S0165-1781(00)00231-6
Andreasen, N. C. (1979). Thought, language, and communication disorders. I. Clinical assessment, definition of terms, and evaluation of their reliability. Arch. Gen. Psychiatry 36, 1315–1321. doi: 10.1001/archpsyc.1979.01780120045006
Arnal, L. H., Doelling, K. B., and Poeppel, D. (2014). Delta–beta coupled oscillations underlie temporal prediction accuracy. Cereb. Cortex 25, 3077–3085. doi: 10.1093/cercor/bhu103
Bagner, D. M., Melinder, M. R. D., and Barch, D. M. (2003). Language comprehension and working memory language comprehension and working memory deficits in patients with schizophrenia. Schizophr. Res. 60, 299–309. doi: 10.1016/S0920-9964(02)00280-3
Başar, E., and Güntekin, B. (2008). A review of brain oscillations in cognitive disorders and the role of neurotransmitters. Brain Res. 1235, 172–193. doi: 10.1016/j.brainres.2008.06.103
Bastos, A. M., and Schoffelen, J.-M. (2015). A tutorial review of functional connectivity analysis methods and their interpretational pitfalls. Front. Syst. Neurosci. 9:175. doi: 10.3389/fnsys.2015.00175
Bellani, M., Perlini, C., and Brambilla, P. (2009). Language disturbances in schizophrenia. Epidemiol. Psichiatr. Soc. 18, 314–317. doi: 10.1017/S1121189X00000270
Billeke, P., Armijo, A., Castillo, D., López, T., Zamorano, F., Cosmelli, D., et al. (2015). Paradoxical expectation: oscillatory brain activity reveals social interaction impairment in schizophrenia. Biol. Psychiatry 78, 421–431. doi: 10.1016/j.biopsych.2015.02.012
Bonhage, C. E., Meyer, L., Gruber, T., Friederici, A. D., and Mueller, J. L. (2017). Oscillatory EEG dynamics underlying automatic chunking during sentence processing. Neuroimage 152, 647–657. doi: 10.1016/j.neuroimage.2017.03.018
Bornkessel-Schlesewsky, I., and Schlesewsky, M. (2019). Toward a neurobiologically plausible model of language-related, negative event-related potentials. Front. Psychol. 10:298. doi: 10.3389/fpsyg.2019.00298
Bourguignon, M., De Tiège, X., de Beeck, M. O., Ligot, N., Paquier, P., Van Bogaert, P., et al. (2013). The pace of prosodic phrasing couples the listener’s cortex to the reader’s voice. Hum. Brain Mapp. 34, 314–326. doi: 10.1002/hbm.21442
Braff, D. L. (1993). Information processing and attention dysfunctions in schizophrenia. Schizophr. Bull. 19, 233–259. doi: 10.1093/schbul/19.2.233
Brandi, M.-L., Kaifel, D., Lahnakoski, J. M., and Schilbach, L. (2020). A naturalistic paradigm simulating gaze-based social interactions for the investigation of social agency. Behav. Res. Methods 52, 1044–1055. doi: 10.3758/s13428-019-01299-x
Brennan, J. (2016). Naturalistic sentence comprehension in the brain. Lang. Linguist. Compass 10, 299–313. doi: 10.1111/lnc3.12198
Brennan, J. R., Lajiness-O’Neill, R., Bowyer, S., Kovelman, I., and Hale, J. T. (2018). Predictive sentence comprehension during story-listening in autism spectrum disorder. Lang. Cogn. Neurosci. 34, 428–439. doi: 10.1080/23273798.2018.1560483
Brennan, J. R., and Martin, A. E. (2020). Phase synchronization varies systematically with linguistic structure composition. Philos. Trans. R. Soc. Lond. B Biol. Sci. 375:20190305. doi: 10.1098/rstb.2019.0305
Breska, A., and Deouell, L. Y. (2017). Neural mechanisms of rhythm-based temporal prediction: delta phase-locking reflects temporal predictability but not rhythmic entrainment. PLoS Biol. 15:e2001665. doi: 10.1371/journal.pbio.2001665
Broderick, M. P., Anderson, A. J., and Lalor, E. C. (2019). Semantic context enhances the early auditory encoding of natural speech. J. Neurosci. 39, 7564–7575. doi: 10.1523/JNEUROSCI.0584-19.2019
Broderick, M. P., Di Liberto, G. P., Anderson, A. J., Rofes, A., and Lalor, E. C. (2020). Dissociable electrophysiological measures of natural language processing reveal differences in comprehension strategy in healthy ageing. bioRxiv [Preprint]. doi: 10.1101/2020.04.17.046201
Brown, M., and Kuperberg, G. R. (2015). A hierarchical generative framework of language processing: linking language perception, interpretation, and production abnormalities in schizophrenia. Front. Hum. Neurosci. 9:611. doi: 10.3389/fnhum.2015.00643
Buzsaki, G. (2006). Rhythms of the Brain. Oxford: Oxford University Press. doi: 10.1093/acprof:oso/9780195301069.001.0001
Canolty, R. T., Edwards, E., Dalal, S. S., Soltani, M., Nagarajan, S. S., Kirsch, H. E., et al. (2006). High gamma power is phase-locked to theta oscillations in human neocortex. Science 313, 1626–1628. doi: 10.1126/science.1128115
Canolty, R. T., and Knight, R. T. (2010). The functional role of cross-frequency coupling. Trends Cogn. Sci. 14, 506–515. doi: 10.1016/j.tics.2010.09.001
Carter, C. S., Perlstein, W., Ganguli, R., Brar, J., Mintun, M., and Cohen, J. D. (1998). Functional hypofrontality and working memory dysfunction in schizophrenia. Am. J. Psychiatry 155, 1285–1287. doi: 10.1176/ajp.155.9.1285
Chaika, E. O. (1990). Understanding Psychotic Speech: Beyond Freud and Chomsky. Springfield, IL: Charles C Thomas Publisher.
Chao, Z. C., Takaura, K., Wang, L., Fujii, N., and Dehaene, S. (2018). Large-scale cortical networks for hierarchical prediction and prediction error in the primate brain. Neuron 100, 1252–1266.e3. doi: 10.1016/j.neuron.2018.10.004
Cibelli, E., Cole, J., Mittal, V., and Goldrick, M. (2017). Prosodic cues to psychosis risk. J. Acoust. Soc. Am. 141, 3700–3700. doi: 10.1121/1.4988072
Colle, L., Angeleri, R., Vallana, M., Sacco, K., Bara, B. G., and Bosco, F. M. (2013). Understanding the communicative impairments in schizophrenia: a preliminary study. J. Commun. Disord. 46, 294–308. doi: 10.1016/j.jcomdis.2013.01.003
Corlett, P. R., Horga, G., Fletcher, P. C., Alderson-Day, B., Schmack, K., and Powers, A. R. (2018). Hallucinations and strong priors. Trends Cogn. Sci 23, 114–127. doi: 10.1016/j.tics.2018.12.001
Covington, M. A., He, C., Brown, C., Naçi, L., McClain, J. T., Fjordbak, B. S., et al. (2005). Schizophrenia and the structure of language: the linguist’s view. Schizophr. Res. 77, 85–98. doi: 10.1016/j.schres.2005.01.016
Crosse, M. J., Di Liberto, G. M., Bednar, A., and Lalor, E. C. (2016). The multivariate temporal response function (mTRF) toolbox: a MATLAB toolbox for relating neural signals to continuous stimuli. Front. Hum. Neurosci. 10:604. doi: 10.3389/fnhum.2016.00604
Crow, T. J. (1998). Nuclear schizophrenic symptoms as a window on the relationship between thought and speech. Br. J. Psychiatry 173, 303–309. doi: 10.1192/bjp.173.4.303
Cuevas, P., He, Y., Billino, J., Kozasa, E., and Straube, B. (2020). Age-related effects on the neural processing of semantic complexity in a continuous narrative: modulation by gestures already present in young to middle-aged adults. Neuropsychologia 151:107725. doi: 10.1016/j.neuropsychologia.2020.107725
Cuevas, P., Steines, M., He, Y., Nagels, A., Culham, J., and Straube, B. (2019). The facilitative effect of gestures on the neural processing of semantic complexity in a continuous narrative. Neuroimage 195, 38–47. doi: 10.1016/j.neuroimage.2019.03.054
Daube, C., Ince, R. A. A., and Gross, J. (2019). Simple acoustic features can explain phoneme-based predictions of cortical responses to speech. Curr. Biol. 29, 1924–1937.e9. doi: 10.1016/j.cub.2019.04.067
DeLisi, L. E. (2001). Speech disorder in schizophrenia: review of the literature and exploration of its relation to the uniquely human capacity for language. Schizophr. Bull. 27, 481–496. doi: 10.1093/oxfordjournals.schbul.a006889
Di Liberto, G. M., Di Liberto, G. M., O’Sullivan, J. A., and Lalor, E. C. (2015). Low-frequency cortical entrainment to speech reflects phoneme-level processing. Curr. Biol. 25, 2457–2465. doi: 10.1016/j.cub.2015.08.030
Di Liberto, G. M., Wong, D., Melnik, G. A., and de Cheveigné, A. (2019). Low-frequency cortical responses to natural speech reflect probabilistic phonotactics. Neuroimage 196, 237–247. doi: 10.1016/j.neuroimage.2019.04.037
Ding, N., Melloni, L., Zhang, H., Tian, X., and Poeppel, D. (2016). Cortical tracking of hierarchical linguistic structures in connected speech. Nat. Neurosci. 19, 158–164. doi: 10.1038/nn.4186
Ding, N., and Simon, J. Z. (2014). Cortical entrainment to continuous speech: functional roles and interpretations. Front. Hum. Neurosci. 8:311. doi: 10.3389/fnhum.2014.00311
Doege, K., Jansen, M., Mallikarjun, P., Liddle, E. B., and Liddle, P. F. (2010). How much does phase resetting contribute to event-related EEG abnormalities in schizophrenia? Neurosci. Lett. 481, 1–5. doi: 10.1016/j.neulet.2010.06.008
Doelling, K. B., Arnal, L. H., Ghitza, O., and Poeppel, D. (2014). Acoustic landmarks drive delta-theta oscillations to enable speech comprehension by facilitating perceptual parsing. Neuroimage 85(Pt 2), 761–768. doi: 10.1016/j.neuroimage.2013.06.035
Dondé, C., Luck, D., Grot, S., Leitman, D. I., Brunelin, J., and Haesebaert, F. (2017). Tone-matching ability in patients with schizophrenia: a systematic review and meta-analysis. Schizophr. Res. 181, 94–99. doi: 10.1016/j.schres.2016.10.009
Donhauser, P. W., and Baillet, S. (2020). Two distinct neural timescales for predictive speech processing. Neuron 105, 385–393.e9. doi: 10.1016/j.neuron.2019.10.019
Duecker, K., Gutteling, T. P., Herrmann, C. S., and Jensen, O. (2020). No evidence for entrainment: endogenous gamma oscillations and responses to rhythmic visual stimulation coexist in visual cortex. bioRxiv [Preprint]. doi: 10.1101/2020.09.02.279497
Ehinger, B. V., and Dimigen, O. (2019). Unfold: an integrated toolbox for overlap correction, non-linear modeling, and regression-based EEG analysis. PeerJ 7:e7838. doi: 10.7717/peerj.7838
Engel, A. K., and Fries, P. (2010). Beta-band oscillations—signalling the status quo? Curr. Opin. Neurobiol. 20, 156–165. doi: 10.1016/j.conb.2010.02.015
Fodor, J. A., and Bever, T. G. (1965). The psychological reality of linguistic segments. J. Verb. Learn. Verb. Behav. 4, 414–420. doi: 10.1016/S0022-5371(65)80081-0
Ford, J. M., Gray, M., Faustman, W. O., Roach, B. J., and Mathalon, D. H. (2007a). Dissecting corollary discharge dysfunction in schizophrenia. Psychophysiology 44, 522–529. doi: 10.1111/j.1469-8986.2007.00533.x
Ford, J. M., Mathalon, D. H., Heinks, T., Kalba, S., Faustman, W. O., and Roth, W. T. (2001). Neurophysiological evidence of corollary discharge dysfunction in schizophrenia. Am. J. Psychiatry 158, 2069–2071. doi: 10.1176/appi.ajp.158.12.2069
Ford, J. M., Roach, B. J., Faustman, W. O., and Mathalon, D. H. (2007b). Synch before you speak: auditory hallucinations in schizophrenia. Am. J. Psychiatry 164, 458–466. doi: 10.1176/ajp.2007.164.3.458
Ford, J. M., Roach, B. J., Hoffman, R. S., and Mathalon, D. H. (2008). The dependence of P300 amplitude on gamma synchrony breaks down in schizophrenia. Brain Res. 1235, 133–142. doi: 10.1016/j.brainres.2008.06.048
Ford, J. M., White, P., Lim, K. O., and Pfefferbaum, A. (1994). Schizophrenics have fewer and smaller P300s: a single-trial analysis. Biol. Psychiatry 35, 96–103. doi: 10.1016/0006-3223(94)91198-3
Frank, S. L., Otten, L. J., Galli, G., and Vigliocco, G. (2015). The ERP response to the amount of information conveyed by words in sentences. Brain Lang. 140, 1–11. doi: 10.1016/j.bandl.2014.10.006
Freedman, R., Adler, L. E., Gerhardt, G. A., Waldo, M., Baker, N., Rose, G. M., et al. (1987). Neurobiological studies of sensory gating in schizophrenia. Schizophr. Bull. 13, 669–678. doi: 10.1093/schbul/13.4.669
Ghitza, O. (2017). Acoustic-driven delta rhythms as prosodic markers. Null 32, 545–561. doi: 10.1080/23273798.2016.1232419
Giraud, A.-L., and Poeppel, D. (2012). Cortical oscillations and speech processing: emerging computational principles and operations. Nat. Neurosci. 15, 511–517. doi: 10.1038/nn.3063
Gross, J., Hoogenboom, N., Thut, G., Schyns, P., Panzeri, S., Belin, P., et al. (2013). Speech rhythms and multiplexed oscillatory sensory coding in the human brain. PLoS Biol. 11:e1001752. doi: 10.1371/journal.pbio.1001752
Güntekin, B., and Başar, E. (2016). Review of evoked and event-related delta responses in the human brain. Int. J. Psychophysiol. 103, 43–52. doi: 10.1016/j.ijpsycho.2015.02.001
Haegens, S. (2020). Entrainment revisited: a commentary on Meyer, Sun, and Martin (2020). Lang. Cogn. Neurosci. 35, 1119–1123. doi: 10.1080/23273798.2020.1758335
Hagoort, P., Hald, L., Bastiaansen, M., and Petersson, K.-M. (2004). Integration of word meaning and world knowledge in language comprehension. Science 304, 438–441. doi: 10.1126/science.1095455
Hale, J. (2001). “A probabilistic earley parser as a psycholinguistic model,” in Proceedings of the Second Meeting of the North American Chapter of the Association for Computational Linguistics on Language Technologies NAACL ’01, (Stroudsburg, PA, USA: Association for Computational Linguistics), 1–8. doi: 10.3115/1073336.1073357
Hale, J. (2016). Information-theoretical complexity metrics. Lang. Linguist. Compass 10, 397–412. doi: 10.1111/lnc3.12196
Hamilton, L. S., and Huth, A. G. (2018). The revolution will not be controlled: natural stimuli in speech neuroscience. Lang. Cogn. Neurosci. 35, 573–582. doi: 10.1080/23273798.2018.1499946
Hasson, U., Egidi, G., Marelli, M., and Willems, R. M. (2018). Grounding the neurobiology of language in first principles: The necessity of non-language-centric explanations for language comprehension. Cognition 180, 135–157. doi: 10.1016/j.cognition.2018.06.018
He, Y., Luell, S., Muralikrishnan, R., Straube, B., and Nagels, A. (2020). Gesture’s body orientation modulates the N400 for visual sentences primed by gestures. Hum. Brain Mapp. 41, 4901–4911. doi: 10.1002/hbm.25166
Heilbron, M., Armeni, K., Schoffelen, J. M., Hagoort, P., and de Lange, F. P. (2020). A hierarchy of linguistic predictions during natural language comprehension. bioRxiv. doi: 10.1101/2020.12.03.410399
Heinz, A., Murray, G. K., Schlagenhauf, F., Sterzer, P., Grace, A. A., and Waltz, J. A. (2019). Towards a unifying cognitive, neurophysiological, and computational neuroscience account of schizophrenia. Schizophr. Bull. 45, 1092–1100. doi: 10.1093/schbul/sby154
Henry, M. J., and Obleser, J. (2012). Frequency modulation entrains slow neural oscillations and optimizes human listening behavior. Proc. Natl. Acad. Sci. U.S.A. 109, 20095–20100. doi: 10.1073/pnas.1213390109
Hickok, G., Farahbod, H., and Saberi, K. (2015). The rhythm of perception. Psychol. Sci. 26, 1006–1013. doi: 10.1177/0956797615576533
Higashima, M., Nagasawa, T., Kawasaki, Y., Oka, T., Sakai, N., Tsukada, T., et al. (2003). Auditory P300 amplitude as a state marker for positive symptoms in schizophrenia: cross-sectional and retrospective longitudinal studies. Schizophr. Res. 59, 147–157. doi: 10.1016/S0920-9964(01)00397-8
Hirano, S., Hirano, Y., Maekawa, T., Obayashi, C., Oribe, N., Kuroki, T., et al. (2008). Abnormal neural oscillatory activity to speech sounds in schizophrenia: a magnetoencephalography study. J. Neurosci. 28, 4897–4903. doi: 10.1523/JNEUROSCI.5031-07.2008
Hirano, S., Nakhnikian, A., Hirano, Y., Oribe, N., Kanba, S., Onitsuka, T., et al. (2018). Phase-amplitude coupling of the electroencephalogram in the auditory cortex in schizophrenia. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 3, 69–76. doi: 10.1016/j.bpsc.2017.09.001
Hirano, S., Spencer, K. M., Onitsuka, T., and Hirano, Y. (2020). Language-related neurophysiological deficits in schizophrenia. Clin. EEG Neurosci. 51, 222–233. doi: 10.1177/1550059419886686
Hoekert, M., Kahn, R. S., Pijnenborg, M., and Aleman, A. (2007). Impaired recognition and expression of emotional prosody in schizophrenia: review and meta-analysis. Schizophr. Res. 96, 135–145. doi: 10.1016/j.schres.2007.07.023
Horga, G., Schatz, K. C., Abi-Dargham, A., and Peterson, B. S. (2014). Deficits in predictive coding underlie hallucinations in schizophrenia. J. Neurosci. 34, 8072–8082. doi: 10.1523/JNEUROSCI.0200-14.2014
Howard, M. F., and Poeppel, D. (2012). The neuromagnetic response to spoken sentences: co-modulation of theta band amplitude and phase. Neuroimage 60, 2118–2127. doi: 10.1016/j.neuroimage.2012.02.028
Howes, O. D., Hird, E. J., Adams, R. A., Corlett, P. R., and McGuire, P. (2020). Aberrant salience, information processing, and dopaminergic signaling in people at clinical high risk for psychosis. Biol. Psychiatry 88, 304–314. doi: 10.1016/j.biopsych.2020.03.012
Hyafil, A., Giraud, A.-L., Fontolan, L., and Gutkin, B. (2015). Neural cross-frequency coupling: connecting architectures, mechanisms, and functions. Trends Neurosci. 38, 725–740. doi: 10.1016/j.tins.2015.09.001
Javitt, D. C. (2009). When doors of perception close: bottom-up models of disrupted cognition in schizophrenia. Annu. Rev. Clin. Psychol. 5, 249–275. doi: 10.1146/annurev.clinpsy.032408.153502
Javitt, D. C., and Freedman, R. (2015). Sensory processing dysfunction in the personal experience and neuronal machinery of schizophrenia. Am. J. Psychiatry 172, 17–31. doi: 10.1176/appi.ajp.2014.13121691
Javitt, D. C., Siegel, S. J., Spencer, K. M., Mathalon, D. H., Hong, L. E., Martinez, A., et al. (2020). A roadmap for development of neuro-oscillations as translational biomarkers for treatment development in neuropsychopharmacology. Neuropsychopharmacology 45, 1411–1422. doi: 10.1038/s41386-020-0697-9
Javitt, D. C., and Sweet, R. A. (2015). Auditory dysfunction in schizophrenia: integrating clinical and basic features. Nat. Rev. Neurosci. 16, 535–550. doi: 10.1038/nrn4002
Jones, A., Hsu, Y.-F., Granjon, L., and Waszak, F. (2017). Temporal expectancies driven by self- and externally generated rhythms. Neuroimage 156, 352–362. doi: 10.1016/j.neuroimage.2017.05.042
Kandylaki, K. D., and Bornkessel-Schlesewsky, I. (2019). From story comprehension to the neurobiology of language. Lang. Cogn. Neurosci. 34, 405–410. doi: 10.1080/23273798.2019.1584679
Kantrowitz, J. T., Epstein, M. L., Beggel, O., Rohrig, S., Lehrfeld, J. M., Revheim, N., et al. (2016). Neurophysiological mechanisms of cortical plasticity impairments in schizophrenia and modulation by the NMDA receptor agonist D-serine. Brain 139, 3281–3295. doi: 10.1093/brain/aww262
Kasai, K., Nakagome, K., Itoh, K., Koshida, I., Hata, A., Iwanami, A., et al. (2002). Impaired cortical network for preattentive detection of change in speech sounds in schizophrenia: a high-resolution event-related potential study. Am. J. Psychiatry 159, 546–553. doi: 10.1176/appi.ajp.159.4.546
Kasai, K., Shenton, M. E., Salisbury, D. F., Hirayasu, Y., Lee, C.-U., Ciszewski, A. A., et al. (2003). Progressive decrease of left superior temporal gyrus gray matter volume in patients with first-episode schizophrenia. Am. J. Psychiatry 160, 156–164. doi: 10.1176/appi.ajp.160.1.156
Kiang, M., and Gerritsen, C. J. (2019). The N400 event-related brain potential response: a window on deficits in predicting meaning in schizophrenia. Int. J. Psychophysiol. 145, 65–69. doi: 10.1016/j.ijpsycho.2019.04.005
Kielar, A., Meltzer, J. A., Moreno, S., Alain, C., and Bialystok, E. (2014). Oscillatory responses to semantic and syntactic violations. J. Cogn. Neurosci. 26, 2840–2862. doi: 10.1162/jocn_a_00670
Kielar, A., Panamsky, L., Links, K. A., and Meltzer, J. A. (2015). Localization of electrophysiological responses to semantic and syntactic anomalies in language comprehension with MEG. Neuroimage 105, 507–524. doi: 10.1016/j.neuroimage.2014.11.016
Kircher, T., Krug, A., Stratmann, M., Ghazi, S., Schales, C., Frauenheim, M., et al. (2014). A rating scale for the assessment of objective and subjective formal Thought and Language Disorder (TALD). Schizophr. Res. 160, 216–221. doi: 10.1016/j.schres.2014.10.024
Kirihara, K., Rissling, A. J., Swerdlow, N. R., Braff, D. L., and Light, G. A. (2012). Hierarchical organization of gamma and theta oscillatory dynamics in schizophrenia. Biol. Psychiatry 71, 873–880. doi: 10.1016/j.biopsych.2012.01.016
Klimesch, W., Sauseng, P., Hanslmayr, S., Gruber, W., and Freunberger, R. (2007). Event-related phase reorganization may explain evoked neural dynamics. Neurosci. Biobehav. Rev. 31, 1003–1016. doi: 10.1016/j.neubiorev.2007.03.005
Kort, N. S., Ford, J. M., Roach, B. J., Gunduz-Bruce, H., Krystal, J. H., Jaeger, J., et al. (2017). Role of N-Methyl-D-aspartate receptors in action-based predictive coding deficits in schizophrenia. Biol. Psychiatry 81, 514–524. doi: 10.1016/j.biopsych.2016.06.019
Kreher, D. A., Goff, D., and Kuperberg, G. R. (2009). Why all the confusion? Experimental task explains discrepant semantic priming effects in schizophrenia under “automatic” conditions: evidence from event-related potentials. Schizophr. Res. 111, 174–181. doi: 10.1016/j.schres.2009.03.013
Kuperberg, G. R., Brothers, T., and Wlotko, E. W. (2020). A tale of two positivities and the N400: distinct neural signatures are evoked by confirmed and violated predictions at different levels of representation. J. Cogn. Neurosci. 32, 12–35. doi: 10.1162/jocn_a_01465
Kuperberg, G. R., Sitnikova, T., Goff, D., and Holcomb, P. J. (2006). Making sense of sentences in schizophrenia: electrophysiological evidence for abnormal interactions between semantic and syntactic processing. J. Abnorm. Psychol. 115, 251–265. doi: 10.1037/0021-843X.115.2.251
Kuperberg, G. R., Weber, K., Delaney-Busch, N., Ustine, C., Stillerman, B., Hämäläinen, M., et al. (2019). Multimodal neuroimaging evidence for looser lexico-semantic networks in schizophrenia:Evidence from masked indirect semantic priming. Neuropsychologia 124, 337–349. doi: 10.1016/j.neuropsychologia.2018.10.024
Kutas, M., and Federmeier, K. D. (2011). Thirty years and counting: finding meaning in the N400 component of the event-related brain potential (ERP). Annu. Rev. Psychol. 62, 621–647. doi: 10.1146/annurev.psych.093008.131123
Kutas, M., and Hillyard, S. A. (1980). Reading senseless sentences: brain potentials reflect semantic incongruity. Science 207, 203–205. doi: 10.1126/science.7350657
Lado-Codesido, M., Pérez, C. M., Mateos, R., Olivares, J. M., and Caballero, A. G. (2019). Improving emotion recognition in schizophrenia with “VOICES”: an on-line prosodic self-training. PLoS One 14:e0210816. doi: 10.1371/journal.pone.0210816
Lakatos, P., Gross, J., and Thut, G. (2019a). A new unifying account of the roles of neuronal entrainment. Curr. Biol. 29, R890–R905. doi: 10.1016/j.cub.2019.07.075
Lakatos, P., Karmos, G., Mehta, A. D., Ulbert, I., and Schroeder, C. E. (2008). Entrainment of neuronal oscillations as a mechanism of attentional selection. Science 320, 110–113. doi: 10.1126/science.1154735
Lakatos, P., O’Connell, M. N., Barczak, A., McGinnis, T., Neymotin, S., Schroeder, C. E., et al. (2019b). The thalamocortical circuit of auditory mismatch negativity. Biol. Psychiatry 87, 770–780. doi: 10.1016/j.biopsych.2019.10.029
Lakatos, P., O’Connell, M. N., Barczak, A., Mills, A., Javitt, D. C., and Schroeder, C. E. (2009). The leading sense: supramodal control of neurophysiological context by attention. Neuron 64, 419–430. doi: 10.1016/j.neuron.2009.10.014
Lakatos, P., Schroeder, C. E., Leitman, D. I., and Javitt, D. C. (2013). Predictive suppression of cortical excitability and its deficit in schizophrenia. J. Neurosci. 33, 11692–11702. doi: 10.1523/JNEUROSCI.0010-13.2013
Lakatos, P., Shah, A. S., Knuth, K. H., Ulbert, I., Karmos, G., and Schroeder, C. E. (2005). An oscillatory hierarchy controlling neuronal excitability and stimulus processing in the auditory cortex. J. Neurophysiol. 94, 1904–1911. doi: 10.1152/jn.00263.2005
Lau, E. F., Phillips, C., and Poeppel, D. (2008). A cortical network for semantics: (de)constructing the N400. Nat. Rev. Neurosci. 9, 920–933. doi: 10.1038/nrn2532
Lee, M., Sehatpour, P., Hoptman, M. J., Lakatos, P., Dias, E. C., Kantrowitz, J. T., et al. (2017). Neural mechanisms of mismatch negativity dysfunction in schizophrenia. Mol. Psychiatry 22, 1585–1593. doi: 10.1038/mp.2017.3
Leitman, D. I., Foxe, J. J., Butler, P. D., Saperstein, A., Revheim, N., and Javitt, D. C. (2005). Sensory contributions to impaired prosodic processing in schizophrenia. Biol. Psychiatry 58, 56–61. doi: 10.1016/j.biopsych.2005.02.034
Leong, V., and Schilbach, L. (2019). The promise of two-person neuroscience for developmental psychiatry: using interaction-based sociometrics to identify disorders of social interaction. Br. J. Psychiatry 215, 636–638. doi: 10.1192/bjp.2019.73
Levinson, S. C. (2016). Turn-taking in human communication – origins and implications for language processing. Trends Cogn. Sci. 20, 6–14. doi: 10.1016/j.tics.2015.10.010
Levy, R. (2008). Expectation-based syntactic comprehension. Cognition 106, 1126–1177. doi: 10.1016/j.cognition.2007.05.006
Lewis, A. G., and Bastiaansen, M. (2015). A predictive coding framework for rapid neural dynamics during sentence-level language comprehension. Cortex 68, 155–168. doi: 10.1016/j.cortex.2015.02.014
Lewis, A. G., Wang, L., and Bastiaansen, M. (2015). Fast oscillatory dynamics during language comprehension: unification versus maintenance and prediction? Brain Lang. 148, 51–63. doi: 10.1016/j.bandl.2015.01.003
Li, A. W. Y., Viñas-Guasch, N., Hui, C. L. M., Chang, W.-C., Chan, S. K. W., Lee, E. H. M., et al. (2018). Verbal working memory in schizophrenia: the role of syntax in facilitating serial recall. Schizophr. Res. 192, 294–299. doi: 10.1016/j.schres.2017.04.008
Li, X., Branch, C. A., and DeLisi, L. E. (2009). Language pathway abnormalities in schizophrenia: a review of fMRI and other imaging studies. Curr. Opin. Psychiatry 22, 131–139. doi: 10.1097/YCO.0b013e328324bc43
Lin, Y., Ding, H., and Zhang, Y. (2018). Emotional prosody processing in schizophrenic patients: a selective review and meta-analysis. J. Clin. Med. Res. 7:363. doi: 10.3390/jcm7100363
Luo, H., and Poeppel, D. (2007). Phase patterns of neuronal responses reliably discriminate speech in human auditory cortex. Neuron 54, 1001–1010. doi: 10.1016/j.neuron.2007.06.004
Mai, G., Minett, J. W., and Wang, W. S.-Y. (2016). Delta, theta, beta, and gamma brain oscillations index levels of auditory sentence processing. Neuroimage 133, 516–528. doi: 10.1016/j.neuroimage.2016.02.064
Martin, A. E. (2020). A compositional neural architecture for language. J. Cogn. Neurosci. 32, 1407–1427. doi: 10.1162/jocn_a_01552
Martínez-Sánchez, F., Muela-Martínez, J. A., Cortés-Soto, P., García Meilán, J. J., Vera Ferrándiz, J. A., Egea Caparrós, A., et al. (2015). Can the acoustic analysis of expressive prosody discriminate schizophrenia? Span. J. Psychol. 18:E86. doi: 10.1017/sjp.2015.85
Mathalon, D. H., Faustman, W. O., and Ford, J. M. (2002). N400 and automatic semantic processing abnormalities in patients with schizophrenia. Arch. Gen. Psychiatry 59, 641–648. doi: 10.1001/archpsyc.59.7.641
Mathalon, D. H., Roach, B. J., and Ford, J. M. (2010). Automatic semantic priming abnormalities in schizophrenia. Int. J. Psychophysiol. 75, 157–166. doi: 10.1016/j.ijpsycho.2009.12.003
McKay, C. M., Headlam, D. M., and Copolov, D. L. (2000). Central auditory processing in patients with auditory hallucinations. Am. J. Psychiatry 157, 759–766. doi: 10.1176/appi.ajp.157.5.759
Meyer, L. (2017). The neural oscillations of speech processing and language comprehension: state of the art and emerging mechanisms. Eur. J. Neurosci. 28:3958.
Meyer, L., and Gumbert, M. (2018). Synchronization of electrophysiological responses with speech benefits syntactic information processing. J. Cogn. Neurosci. 30, 1066–1074. doi: 10.1162/jocn_a_01236
Meyer, L., Henry, M. J., Gaston, P., Schmuck, N., and Friederici, A. D. (2017). Linguistic bias modulates interpretation of speech via neural delta-band oscillations. Cereb. Cortex 27, 4293–4302. doi: 10.1093/cercor/bhw228
Meyer, L., Sun, Y., and Martin, A. E. (2019). Synchronous, but not entrained: exogenous and endogenous cortical rhythms of speech and language processing. Lang. Cogn. Neurosci. 35, 1089–1099. doi: 10.1080/23273798.2019.1693050
Meyer, L., Sun, Y., and Martin, A. E. (2020). Entraining” to speech, generating language? Lang. Cogn. Neurosci. 35, 1138–1148. doi: 10.1080/23273798.2020.1827155
Michie, P. T., Malmierca, M. S., Harms, L., and Todd, J. (2016). The neurobiology of MMN and implications for schizophrenia. Biol. Psychol. 116, 90–97. doi: 10.1016/j.biopsycho.2016.01.011
Miller, G. A., and Isard, S. (1963). Some perceptual consequences of linguistic rules. J. Verb. Learn. Verb. Behav. 2, 217–228. doi: 10.1016/S0022-5371(63)80087-0
Mitchell, R. L. C., and Crow, T. J. (2005). Right hemisphere language functions and schizophrenia: the forgotten hemisphere? Brain 128, 963–978. doi: 10.1093/brain/awh466
Mohammad, O. M., and DeLisi, L. E. (2013). N400 in schizophrenia patients. Curr. Opin. Psychiatry 26, 196–207. doi: 10.1097/YCO.0b013e32835d9e56
Molinaro, N., Lizarazu, M., Lallier, M., Bourguignon, M., and Carreiras, M. (2016). Out-of-synchrony speech entrainment in developmental dyslexia. Hum. Brain Mapp. 37, 2767–2783. doi: 10.1002/hbm.23206
Morice, R., and Delahunty, A. (1996). Frontal/executive impairments in schizophrenia. Schizophr. Bull. 22, 125–137. doi: 10.1093/schbul/22.1.125
Moro, A., Bambini, V., Bosia, M., Anselmetti, S., Riccaboni, R., Cappa, S. F., et al. (2015). Detecting syntactic and semantic anomalies in schizophrenia. Neuropsychologia 79, 147–157. doi: 10.1016/j.neuropsychologia.2015.10.030
Mulert, C., Kirsch, V., Pascual-Marqui, R., McCarley, R. W., and Spencer, K. M. (2011). Long-range synchrony of gamma oscillations and auditory hallucination symptoms in schizophrenia. Int. J. Psychophysiol. 79, 55–63. doi: 10.1016/j.ijpsycho.2010.08.004
Murphy, D., and Cutting, J. (1990). Prosodic comprehension and expression in schizophrenia. J. Neurol. Neurosurg. Psychiatry 53, 727–730. doi: 10.1136/jnnp.53.9.727
Näätänen, R., and Kähkönen, S. (2008). Central auditory dysfunction in schizophrenia as revealed by the mismatch negativity (MMN) and its magnetic equivalent MMNm: a review. Int. J. Neuropsychopharmacol. 12, 125. doi: 10.1017/S1461145708009322
Nieuwland, M. S., Barr, D. J., Bartolozzi, F., Busch-Moreno, S., Darley, E., Donaldson, D. I., et al. (2020). Dissociable effects of prediction and integration during language comprehension: evidence from a large-scale study using brain potentials. Philos. Trans. R. Soc. Lond. B Biol. Sci. 375:20180522. doi: 10.1098/rstb.2018.0522
Nieuwland, M. S., and Van Berkum, J. J. A. (2006). When peanuts fall in love: N400 evidence for the power of discourse. J. Cogn. Neurosci. 18, 1098–1111. doi: 10.1162/jocn.2006.18.7.1098
Obleser, J., and Kayser, C. (2019). Neural entrainment and attentional selection in the listening brain. Trends Cogn. Sci. 23, 913–926. doi: 10.1016/j.tics.2019.08.004
Patterson, J. V., Hetrick, W. P., Boutros, N. N., Jin, Y., Sandman, C., Stern, H., et al. (2008). P50 sensory gating ratios in schizophrenics and controls: a review and data analysis. Psychiatry Res. 158, 226–247. doi: 10.1016/j.psychres.2007.02.009
Pawełczyk, A., Kotlicka-Antczak, M., Łojek, E., Ruszpel, A., and Pawełczyk, T. (2018a). Schizophrenia patients have higher-order language and extralinguistic impairments. Schizophr. Res. 192, 274–280. doi: 10.1016/j.schres.2017.04.030
Pawełczyk, A., Łojek, E., Żurner, N., Gawłowska-Sawosz, M., and Pawełczyk, T. (2018b). Higher-order language dysfunctions as a possible neurolinguistic endophenotype for schizophrenia: Evidence from patients and their unaffected first degree relatives. Psychiatry Res. 267, 63–72. doi: 10.1016/j.psychres.2018.05.070
Peelle, J. E., Gross, J., and Davis, M. H. (2013). Phase-locked responses to speech in human auditory cortex are enhanced during comprehension. Cereb. Cortex 23, 1378–1387. doi: 10.1093/cercor/bhs118
Piai, V., Roelofs, A., and Maris, E. (2014). Oscillatory brain responses in spoken word production reflect lexical frequency and sentential constraint. Neuropsychologia 53, 146–156. doi: 10.1016/j.neuropsychologia.2013.11.014
Poeppel, D., and Teng, X. (2020). “Entrainment in human auditory cortex: mechanism and functions,” in The Senses: A Comprehensive Reference, 63–76. doi: 10.1016/b978-0-12-805408-6.00018-x
Powers, A. R., Mathys, C., and Corlett, P. R. (2017). Pavlovian conditioning–induced hallucinations result from overweighting of perceptual priors. Science 357, 596–600. doi: 10.1126/science.aan3458
Prystauka, Y., and Lewis, A. G. (2019). The power of neural oscillations to inform sentence comprehension: a linguistic perspective. Lang. Linguist. Compass 13:31. doi: 10.1111/lnc3.12347
Püschel, J., Stassen, H. H., Bomben, G., Scharfetter, C., and Hell, D. (1998). Speaking behavior and speech sound characteristics in acute schizophrenia. J. Psychiatr. Res. 32, 89–97. doi: 10.1016/S0022-3956(98)00046-6
Rabagliati, H., Delaney-Busch, N., Snedeker, J., and Kuperberg, G. (2019). Spared bottom-up but impaired top-down interactive effects during naturalistic language processing in schizophrenia: evidence from the visual-world paradigm. Psychol. Med. 49, 1335–1345. doi: 10.1017/S0033291718001952
Rapcan, V., D’Arcy, S., Yeap, S., Afzal, N., Thakore, J., and Reilly, R. B. (2010). Acoustic and temporal analysis of speech: a potential biomarker for schizophrenia. Med. Eng. Phys. 32, 1074–1079. doi: 10.1016/j.medengphy.2010.07.013
Rimmele, J. M., Morillon, B., Poeppel, D., and Arnal, L. H. (2018). Proactive sensing of periodic and aperiodic auditory patterns. Trends Cogn. Sci. 22, 870–882. doi: 10.1016/j.tics.2018.08.003
Roark, B., Bachrach, A., and Cardenas, C. (2009). “Deriving lexical and syntactic expectation-based measures for psycholinguistic modeling via incremental top-down parsing,” in Proceedings of the 2009 Conference on Empirical Methods in Natural Language Processing, Stroudsburg, PA. doi: 10.3115/1699510.1699553
Rochester, S. R., Harris, J., and Seeman, M. V. (1973). Sentence processing in schizophrenic listeners. J. Abnorm. Psychol. 82, 350–356. doi: 10.1037/h0035146
Roehm, D., Bornkessel-Schlesewsky, I., and Schlesewsky, M. (2007). The internal structure of the N400: Frequency characteristics of a language related ERP component. Chaos Complexity Lett. 2, 365–395.
Roopun, A. K., Kramer, M. A., Carracedo, L. M., Kaiser, M., Davies, C. H., Traub, R. D., et al. (2008). Period concatenation underlies interactions between gamma and beta rhythms in neocortex. Front. Cell. Neurosci. 2:1. doi: 10.3389/neuro.03.001.2008
Salisbury, D. F. (2008). Semantic activation and verbal working memory maintenance in schizophrenic thought disorder: insights from electrophysiology and lexical amibiguity. Clin. EEG Neurosci. 39, 103–107. doi: 10.1177/155005940803900217
Sassenhagen, J. (2019). How to analyse electrophysiological responses to naturalistic language with time-resolved multiple regression. Lang. Cogn. Neurosci. 34, 474–490. doi: 10.1080/23273798.2018.1502458
Shamay-Tsoory, S. G., and Mendelsohn, A. (2019). Real-life neuroscience: an ecological approach to brain and behavior research. Perspect. Psychol. Sci. 14, 841–859. doi: 10.1177/1745691619856350
Shannon, C. E. (1948). A mathematical theory of communication. Bell Syst. Tech. J. 27, 379–423. doi: 10.1002/j.1538-7305.1948.tb01338.x
Sharpe, V., Weber, K., and Kuperberg, G. R. (2020). Impairments in probabilistic prediction and bayesian learning can explain reduced neural semantic priming in schizophrenia. Schizophr. Bull. 46, 1558–1566. doi: 10.1093/schbul/sbaa069
Sichlinger, L., Cibelli, E., Goldrick, M., and Mittal, V. A. (2019). Clinical correlates of aberrant conversational turn-taking in youth at clinical high-risk for psychosis. Schizophr. Res. 204, 419–420. doi: 10.1016/j.schres.2018.08.009
Sitnikova, T., Salisbury, D. F., Kuperberg, G., and Holcomb, P. J. (2002). Electrophysiological insights into language processing in schizophrenia. Psychophysiology 39, 851–860. doi: 10.1111/1469-8986.3960851
Smith, R., Badcock, P., and Friston, K. J. (2021). Recent advances in the application of predictive coding and active inference models within clinical neuroscience. Psychiatry Clin. Neurosci. 75, 3–13. doi: 10.1111/pcn.13138
Spencer, K. M., Salisbury, D. F., Shenton, M. E., and McCarley, R. W. (2008). γ-Band auditory steady-state responses are impaired in first episode psychosis. Biol. Psychiatry 64, 369–375. doi: 10.1016/j.biopsych.2008.02.021
Spitzer, M., Weisker, I., Winter, M., and Maier, S. (1994). Semantic and phonological priming in schizophrenia. J. Abnorm. Psychol. 103, 485–494. doi: 10.1037/0021-843X.103.3.485
Stefanics, G., Hangya, B., Hernádi, I., and Winkler, I. (2010). Phase entrainment of human delta oscillations can mediate the effects of expectation on reaction speed. J. Neurosci. 30, 13578–13585. doi: 10.1523/JNEUROSCI.0703-10.2010
Stehwien, S., Henke, L., Hale, J., Brennan, J., and Meyer, L. (2020). “The little prince in 26 languages: towards a multilingual neuro-cognitive corpus,” in Proceedings of the Second Workshop on Linguistic and Neurocognitive Resources, Marseille, 43–49.
Sterzer, P., Adams, R. A., Fletcher, P., Frith, C., Lawrie, S. M., Muckli, L., et al. (2018). The predictive coding account of psychosis. Biol. Psychiatry 84, 634–643. doi: 10.1016/j.biopsych.2018.05.015
Stolcke, A. (2002). “SRILM-an extensible language modeling toolkit,” in Proceedings of the Seventh International Conference on Spoken Language Processing, Denver, CO.
Stuke, H., Weilnhammer, V. A., Sterzer, P., and Schmack, K. (2018). Delusion proneness is linked to a reduced usage of prior beliefs in perceptual decisions. Schizophr. Bull. 45, 80–86. doi: 10.1093/schbul/sbx189
Teufel, C., Subramaniam, N., Dobler, V., Perez, J., Finnemann, J., Mehta, P. R., et al. (2015). Shift toward prior knowledge confers a perceptual advantage in early psychosis and psychosis-prone healthy individuals. Proc. Natl. Acad. Sci. U.S.A. 112, 13401–13406. doi: 10.1073/pnas.1503916112
Titone, D., Levy, D. L., and Holzman, P. S. (2000). Contextual insensitivity in schizophrenic language processing: evidence from lexical ambiguity. J. Abnorm. Psychol. 109, 761–767. doi: 10.1037/0021-843X.109.4.761
Todd, J., Harms, L., Michie, P., and Schall, U. (2013). Mismatch negativity: translating the potential. Front. Psychiatry 4:171. doi: 10.3389/fpsyt.2013.00171
Vander Ghinst, M., Bourguignon, M., Op, de Beeck, M., Wens, V., Marty, B., et al. (2016). Left superior temporal gyrus is coupled to attended speech in a cocktail-party auditory scene. J. Neurosci. 36, 1596–1606. doi: 10.1523/JNEUROSCI.1730-15.2016
Vassileiou, B., Meyer, L., Beese, C., and Friederici, A. D. (2018). Alignment of alpha-band desynchronization with syntactic structure predicts successful sentence comprehension. Neuroimage 175, 286–296. doi: 10.1016/j.neuroimage.2018.04.008
Wang, L., Hagoort, P., and Jensen, O. (2018). Gamma oscillatory activity related to language prediction. J. Cogn. Neurosci. 30, 1075–1085. doi: 10.1162/jocn_a_01275
Wang, L., Jensen, O., van den Brink, D., Weder, N., Schoffelen, J.-M., Magyari, L., et al. (2012a). Beta oscillations relate to the N400m during language comprehension. Hum. Brain Mapp. 33, 2898–2912. doi: 10.1002/hbm.21410
Wang, L., Zhu, Z., and Bastiaansen, M. (2012b). Integration or predictability? A further specification of the functional role of gamma oscillations in language comprehension. Front. Psychol. 3:187. doi: 10.3389/fpsyg.2012.00187
Weisbrod, M., Maier, S., and Harig, S. (1998). Lateralised semantic and indirect semantic priming effects in people with schizophrenia. Br. J. Psychiatry 172, 142–146. doi: 10.1192/bjp.172.2.142
Keywords: neural tracking, neural oscillations, schizophrenia, electroencephalography, language comprehension, speech perception
Citation: Meyer L, Lakatos P and He Y (2021) Language Dysfunction in Schizophrenia: Assessing Neural Tracking to Characterize the Underlying Disorder(s)? Front. Neurosci. 15:640502. doi: 10.3389/fnins.2021.640502
Received: 11 December 2020; Accepted: 03 February 2021;
Published: 22 February 2021.
Edited by:
Giovanni M. Di Liberto, École Normale Supérieure, FranceReviewed by:
Edmund C Lalor, University of Rochester, United StatesYoji Hirano, Kyushu University, Japan
Copyright © 2021 Meyer, Lakatos and He. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yifei He, eWlmZWkuaGVAc3RhZmYudW5pLW1hcmJ1cmcuZGU=; eWlmZWkuaGVAbWVkLnVuaS1tYXJidXJnLmRl
 Lars Meyer
Lars Meyer Peter Lakatos3
Peter Lakatos3 Yifei He
Yifei He