- 1Faculty of Health, York University, Toronto, ON, Canada
- 2OpenLab, University Health Network (UHN), Toronto, ON, Canada
- 3Perley Health Rideau Veretans’ Health Centre, Ottawa, ON, Canada
Background: Due to the high prevalence of post-traumatic stress disorder (PTSD) among veterans, as this population ages, they are more likely to develop dementia and exhibit behavioral and psychological symptoms of dementia (BPSD), including responsive behaviors. BPSDs are linked to adverse clinical outcomes, hospitalization, and earlier mortality and are directly related to increased cost and burden of care. In long-term care institutions, residents' behaviors such as physical (striking out, biting, grabbing, etc.) and/or verbal (cursing, screaming, etc.) reactions are associated with higher staff burnout levels which contribute to absenteeism, high turnover, low engagement, and elevated risk of patient abuse or neglect. Despite their limited effectiveness and association with hastening of cognitive and physical decline, medications (neuroleptic/sedating drugs) are commonly used for people with dementia who exhibit responsive behaviors. In long-term care settings, more than 30% of veterans with high-care needs and 20% of those with low-care needs are prescribed drugs to manage their symptoms and behaviors. There is growing pressure from the medical community to engage in non-pharmacological strategies as the first-line of treatment to reduce BPSDs. Virtual reality (VR) presents a unique opportunity to transport people away from environmental factors that amplify feelings of loneliness, boredom, and discomfort, which are known to trigger responsive behaviors, into natural calming settings (such as a peaceful lake, or a colorful forest). Using immersive VR as a non-pharmacological therapy has been piloted with frail older adults in both community and acute-care settings with promising results. However, to date, there have been no rigorous longitudinal studies of VR therapy in long-term care, in particular, studies that evaluate its potential to reduce responsive behaviors related to triggering events. The current study provided novel opportunities for Perley and Rideau Veterans' Health Centre (Perley Health), which has prioritized reducing resident responsive behaviors and maintaining a healthy workforce.
Objective: The purpose of this study was to evaluate the feasibility and potential benefits of introducing VR therapy in a veterans' long-term care health center, with the main goal of reducing responsive behaviors for veterans living with dementia, including responsive behaviors related to experiencing physical and emotional pain. This includes evaluating VR therapy with respect to acceptability, comfort, enjoyment, relaxation, and its ability to promote reminiscence. Of special interest was the impact of VR therapy in cases where responsive behaviors were triggered by a predictable environmental event (e.g., bathing, toileting, etc.).
Methods: This was a prospective, longitudinal, non-randomized interventional study that employed convenience sampling. Veterans residing in this long-term care setting who exhibited responsive behaviors were recruited and grouped into two categories according to how they usually exhibited responsive behaviors: Group T—responsive behaviors were triggered by known activities or events in a relatively predictable way (e.g., sundowning, wound care), and Group S—initiation of responsive behaviors did not follow specific predictable patterns. Residents in both groups received the VR therapy intervention, which consisted of watching 360° VR video footage of natural and social scenes using an Oculus Go head-mounted-display. Group T received “targeted” VR therapy sessions occurring just before or during events that could trigger responsive behaviors (e.g., before bathing). Group S received “scheduled” VR therapy sessions akin to other recreational activities (e.g., at a mutually convenient time during the day). Intended data collection consisted of baseline scores from validated tools including the Pain Assessment for Advanced Dementia (PAINAD), Resident Assessment Instrument-Minimum Data Set 2.0 (RAI-MDS), and Palliative Performance Scale and daily clinical progress notes extracted from patients' electronic records during the study period, as well as intervention data-collection tool comprising a quantitative survey (for residents' feedback when possible) and qualitative structured observations during the intervention by recreational therapists (RTs). Also described are the changes implemented to data collection and analyses as a number of methodological challenges arose during the study.
Results: Thirty-three veterans (mean age 91.6 years, SD 5.9) with varying degrees of cognitive impairment: 3% (1/33) borderline intact, 15% (5/33) mild impairment, 61% (20/33) moderate impairment, 12% (4/33) moderately severe impairment, and 6% (2/33) severe impairment participated in the study. The number of sessions per participant ranged from 2 to 6, with an average of 3.3 (SD = 1) sessions per participant. A total of 111 VR therapy sessions took place, 98 of which were scheduled (88%) and 13 were targeted (12%). The RTs reported that targeted sessions were particularly difficult to conduct due to staffing/resource constraints. In 61% (68/111) of all sessions, no responsive behaviors were observed during, or soon after, the VR therapy, and no pro re nata (PRN) medications had to be administered during the sessions. In 46% (6/13) of targeted sessions, participants did not exhibit responsive behaviors usually triggered by a specific environmental event. The majority (63%, 70/111) of participants found the technology comfortable, and in 47% (52/111) of sessions, the RTs reported that VR therapy made the resident feel good or better than they felt before the session. In 33% (37/111) of all sessions, residents reminisced about the past and in 67% (74/111) of sessions residents reported wanting to try VR again.
Conclusion: Findings indicate that VR therapy is overall acceptable and enjoyable for veterans living with dementia with varying degrees of cognitive and physical impairments. Staff at the veterans' center continued to use scheduled VR therapy as a recreational tool beyond the study period. Notwithstanding the difficulties in administering targeted sessions, there was observational evidence of the potential to reduce environmentally triggered responsive behaviors; this warrants further exploration of approaches to improve protocol feasibility in support of studying treatment effectiveness. Finally, manufacturers and providers of VR therapy should consider ways in which content, equipment, and administration can be customized and optimized for this particularly frail and diverse population.
Background
The prevalence of post-traumatic stress disorder (PTSD) among veterans was estimated at 23% in a meta-analysis involving close to five million veterans examined in 33 studies (Fulton et al., 2015). The world’s aging population brings increasing numbers of individuals with behavioural and psychological symptoms of dementia (BPSDs), particularly in nursing home settings (Finkel et al., 1996). Aging veterans with PTSD are more than twice as likely to develop dementia and compared with those without PTSD (Günak et al., 2020; Qureshi et al., 2010; Yaffe et al., 2010), and have been found to exhibit more significant BPSDs (Pinciotti et al., 2017). Responsive behaviors are often exhibited by people with dementia (PwD) and can include physical (striking out, biting, grabbing etc.) and/or verbal (cursing, screaming, etc.) aggression, agitation (pacing nervously, excessive physical movement), and restlessness (Alzheimer's Society of Canada, 2019; Pelletier and Landreville, 2007). These behaviors can show patterns related to time of day, such as sundowning, and/or can be triggered by particular care or treatment activities such as dressing changes (e.g., wound care), or personal care (e.g., bathing).
Some studies have noted a relationship between higher levels of staff stress and prevalence of responsive behaviors such as aggression and screaming (Edvardsson et al., 2008; Miyamoto et al., 2010); staff stress levels are related to higher levels of burnout, absenteeism, turnover, risk of resident neglect, and lower levels of job satisfaction (Islam et al., 2017). Further, an important implication of unmanaged responsive behaviors surrounds limits to participation in meaningful activities that are conducive to the health of individuals with dementia. Responsive behaviors often lead to reciprocal challenges with interpersonal interaction which can result in poorer provision of care and separation of the individual with responsive behaviors from other residents in long-term care (Erkes et al., 2021).
Among current means to manage BPSD are pharmacological interventions (neuroleptic/sedating medications) and physical barriers and restraints (alarms, locks, Buxton chairs, tethers). These interventions are associated with negative physical and mental consequences for patients, such as skin trauma, muscle atrophy, anxiety, distress, and aggressive behavior (Gastmans and Milisen, 2006; Agens, 2010). Their use in older adult patient populations poses ethical issues and raises feelings of unhappiness among staff (Gunawardena and Smithard, 2019). In long-term care settings, more than 30% of veterans with high-care needs and 20% with low-care needs are prescribed drugs to manage their symptoms and behaviors (University of South Australia, 2007).
New approaches are clearly needed, and many non-pharmacological strategies have been attempted with varying levels of success (ONeil et al., 2011; Meyer and O'Keefe, 2018; Erkes et al., 2021). There is growing pressure from the medical community to engage in non-pharmacological strategies as the first line of treatment to reduce BPSDs when possible (National Collaborating Centre for Mental Health, 2007; Guideline Adaptation Committee, 2016; Dyer et al., 2017). Given the promising effects of music therapy and arts-based therapy, researchers have suggested a similar exploration of the application of virtual reality (VR) technology for dementia care (Rusted et al., 2006; García-Betances et al., 2015; Wang and Li, 2016; Gómez-Romero et al., 2017; Zhang et al., 2017; D'Cunha et al., 2019; Dermody et al., 2020; Kishita et al., 2020).
VR presents a unique opportunity to transport people away from environmental factors that amplify feelings (e.g., loneliness, boredom, and discomfort) known to trigger responsive behaviors into natural calming settings (e.g., a peaceful lake, a colorful forest, etc.). A VR head-mounted device (HMD) seamlessly replaces the viewer's real environment with rich sights and sounds, generating a three-dimensional simulated world displayed all around them. The technology has demonstrated good feasibility and potential to benefit frail older adults, including populations with dementia (Appel et al., 2020a; Appel et al., 2020b; Appel et al., 2021a). VR interventions have already been adopted or incorporated as a means to screen for mild cognitive impairment, improve cognitive training, practice activities of daily living, and promote wellbeing more broadly in PwD (Beason-Held et al., 2013; D'Cunha et al., 2019; Liu et al., 2019; Clay et al., 2020; Matsangidou et al., 2020). VR has also been used as a tool to help build empathy in providers and the general public towards those living with dementia and their caregivers (Wijma et al., 2017).
As VR technology becomes increasingly accessible, it provides a deployable and scalable solution across different healthcare settings such as acute care hospitals, rehabilitation institutions, long-term care facilities, and in the home. A 2021 scoping review on the topic of VR as a means to promote wellbeing in PwD found that no studies to date have focused specifically on the use of VR in managing pain, and only a few studies vaguely addressed its impact on responsive behaviors in this group (Appel et al., 2021b). Furthermore, nearly half of studies had 10 or fewer participants, and only four studies reported using 360° VR films (i.e., as opposed to two-dimensional films or computer graphics) (Appel et al., 2021a).
Exposure to natural environments (seeing greenery, hearing natural sounds of the outdoors) has been shown to decrease anxiety and physiological distress (White et al., 2019). VR therapy that uses 360° films can provide residents in long-term care with the opportunity to explore a variety of outdoor scenery and other personally meaningful and realistic content. This pilot project was designed to address the gap in knowledge regarding the feasibility and the outcomes of using 360° VR films as means to reduce responsive behaviors for veterans and residents in a long-term care center. Factors related to the potential of VR therapy to improve wellbeing through promoting enjoyment, engagement, relaxation, and reminiscence were also investigated.
Aims/Objectives
The purpose of this study was to evaluate the feasibility and potential benefits of introducing VR therapy to veterans and residents in a long-term care health center, with the main goal of reducing responsive behaviors for individuals living with dementia. This included evaluating VR therapy with respect to managing responsive behaviors and pain, as well as to measure its acceptability and participant enjoyment, relaxation, and reminiscence during therapy. The study focused on two distinct types of VR therapy administration:
Targeted sessions (Group T) were conducted shortly before or during special care/treatment activities (e.g., wound dressing change, bathing, toileting) known to trigger responsive behaviors in certain residents to observe the effect of VR therapy on direct/immediate management of these behaviors.
Scheduled sessions (Group S) were conducted with residents during recreational activity time, to evaluate the effect of VR therapy on frequency and/or intensity of responsive behaviors and its potential to distract from physical and emotional pain/discomfort and promote relaxation and stimulation.
Methods
This was a prospective, longitudinal, open, non-randomized interventional clinical trial based on convenience sampling. It employed a mixed-methods design, consisting of quantitative and qualitative outcomes. The study design included data collection from sources such as electronic patient records, pre–post intervention surveys, validated instruments, structured observations, and progress notes.
Design
The study design consisted of VR therapy sessions conducted in two distinct conditions and categorized accordingly in the following groups: Group T—Targeted VR therapy, and Group S—Scheduled VR therapy.
Targeted VR therapy sessions were conducted prior to, or during, special care procedures (e.g., wound dressing change, bathing, toileting) known to trigger responsive behaviors. Participants were assigned to this group based on a documented history of behaviors with identifiable triggers. Participants in this group took part in at least one triggered VR therapy session.
Scheduled VR therapy sessions were conducted by trained recreation therapists/recreation professionals (RTs) during recreational activity programming time or using time available between residents' treatments, appointments, visits, or other activities. Participants assigned to this group took part in at least one scheduled VR therapy session and could choose to participate in additional sessions thereafter.
Setting
Participants were recruited between January and December of 2019 from a long-term care home in Canada. The Perley and Rideau Veterans' Health Centre (Perley Health) is one of the largest long-term care homes in Ontario. The Perley Health campus includes 450 long-term care beds, a Seniors Village with 139 independent-living apartments for seniors, and many clinical, therapeutic, and recreational services available on site. The Centre of Excellence in Frailty-Informed Care (CoE), established at the Perley Health in 2019, conducts applied research and identifies, develops, and shares best practices in the care of older adults living with frailty. Perley Health is home to 230 veteran residents. The Resident Assessment Instrument-Minimum Data Set (RAI-MDS) data from the fourth quarter of 2019 indicated that 66.7% of veteran residents at the Perley Health had a diagnosis of dementia. Physical responsiveness was observed in 17.3% of the total veteran population. Moreover, 21% of veteran residents were recorded with previous mental health history (Canadian Institute for Health Information, 2019), and according to The RAI-MDS data, 15% of Perley Health veteran residents experienced worsened behavioral symptoms in 2019 (Canadian Institute for Health Information, 2019).
Ethics approval for the pilot study was obtained from York University's Research Ethics Board (reference number Ethics Protocol #2756).
Recruitment and Consent
Recruitment of study participants was based on convenience sampling. Healthcare professionals at the Perley and Rideau Veterans' Health Centre identified residents who met the eligibility criteria from the group of residents under their care at the time. An informational flyer was produced to help recruit participants and provide details to personal caregivers (see Supplementary Material S1). Potential participants were initially approached by a member of their circle of care and asked about their interest in the study. Those who responded positively had a follow-up meeting with the study research coordinator (RC). Prospective participants able to provide informed consent met in person with the RC who explained the study, its risks and benefits, and the consent process in detail. For prospective participants unable to provide informed consent (based on a healthcare provider's formal assessment), the RC contacted the resident's substitute decision maker (SDM) via phone, explained the study and the consent process in detail, and requested verbal consent which was documented in the resident's chart.
Inclusion/Exclusion Criteria
Inclusion criteria required that study participants were adults over 18 years old, could communicate in English, were clinically diagnosed with dementia, and exhibited responsive behaviors. Residents were excluded from the study if they presented with any of the following: vision impairment at a level that would make it difficult/impossible for them to see the VR films; open wounds or skin conditions on the face; chronic neck pain/injury that would make it unsafe to wear the VR headset; pacemakers or implanted defibrillators; or a known history of seizures.
Participants
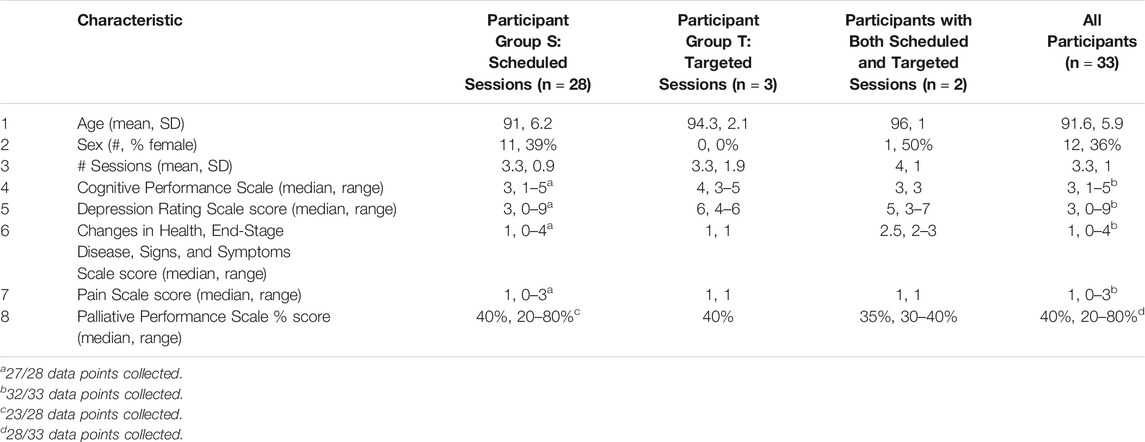
A total of 33 veterans (12 female) with an average age of 91.6 years (SD 5.9) participated in the study. Baseline assessment included the Palliative Performance Scale (Victoria Hospice Society, 2006), as well as metrics drawn from a subset of the Resident Assessment Instrument-Home Care (RAI-HC) Describing Outcome Scales (Canadian Institute for Health Informatics, 2013) used by Perley Health. Participants had varying degrees of cognitive impairment as assessed by Cognitive Impairment Scale (CPS) scores: 3% (1/33) borderline intact, 15% (5/33) mild impairment, 61% (20/33) moderate impairment, 12% (4/33) moderately severe impairment, 6% (2/33) severe impairment, and 0% (0/33) very severe impairment participated in the study, and 3% (1/33) had no data available. The median daily Pain Scale score was 1 with scores ranging from 0 to 3 and higher scores indicating a greater level of pain. The median Depression Rating Scale (DRS) score was 3 with scores ranging from 0 to 9; a score of 3 or more on the DRS suggests potential or actual depression. The median Changes in Health, End-Stage Disease, Signs, and Symptoms Scale (CHESS) score was 1 with scores ranging from 0 to 4 and higher scores indicating greater medical complexity. The median Palliative Performance Scale score was 40% with scores ranging from 20% (totally bed bound, extensive disease, complete reliance for self-care, minimal food intake to sips, fully conscious with varied levels of confusion) to 80% (full ambulation, some evidence of disease, normal or reduced food intake, normal activity with effort, self-reliant for self-care, fully conscious). Table 1 describes participant demographics and baseline clinical data.
The majority (85%, 28/33) of participants took part in scheduled study sessions, and 9% (3/33) took part in targeted sessions. Two participants (2/33, 6%) completed both scheduled and targeted sessions throughout their stay. Their demographics do not differ from the rest of the participants in any significant way. There were no significant differences at baseline (one-way ANOVA at 0.05 significance) between participants assigned to scheduled sessions (Group S) or to targeted sessions (Group T) or those who received both targeted and scheduled sessions in terms of average age, sex, number of sessions completed, cognitive impairment, frailty, pain frequency and intensity, or depression. Table 1 describes participant demographics and baseline clinical data by study group.
VR Therapy Intervention
Therapy was administered by seven RTs who were the main point of contact for participants, instructed participants on the technology, and provided assistance throughout therapy sessions. The RTs also coordinated scheduling of targeted sessions with health care practitioners (HCPs) (i.e., nurses/nurse aides/personal support workers) who would be caring for participants in Group T at the time of triggering events.
For both scheduled and targeted sessions, the VR therapy intervention consisted of participants watching one or more short 360° VR films (1–3 min each), using a VR HMD (or headset). Participants were sitting on a swivel chair (allowing them to rotate around in a full circle), a wheelchair, or a safe standard chair or were sitting or lying down on their bed in the presence of the RT conducting the session.
The VR equipment consisted of an Oculus Go VR HMD, selected due to its simplicity, fidelity of experience, relative affordability, portability (mobile and wearable), and ease-of-use (stand-alone requiring no external hardware). The Oculus Go features a built-in head tracking module which greatly improves the motion latency, reducing the risk of simulator sickness due to motion lag. Figure 1 depicts a veteran wearing an Oculus Go VR headset.
The VR therapy experiences consisted of a collection of short 360° VR films including various nature scenes (rocky lakeshore, sunny forest, dense forest, floating icebergs, and sunny beach). The films were specially designed for persons with dementia and contained calming scenes with no first-person movement to mitigate simulator sickness. Figure 2 shows a screenshot from two of the nature scenes.

FIGURE 2. 2D screen capture of VR nature scenes (Scene 2: Open field with foliage and Scene 5: Aquamarine beach).
The RTs assisted with creating and selecting additional tailored content for participants by filming 360° videos of local areas of interest to the participants (e.g., Parliament Hill at night with holiday lighting) and showing residents additional nature and animal-themed YouTube videos or a Google Street View walk-through. RTs were provided with guidance by the research team on how to create and select VR content (i.e., no first-person movement, minimal changes in sound and lighting) to decrease the chance of simulator sickness.
Data Collection
Quantitative and qualitative data were collected from two sources:
1) Electronic patient record (EPR): participants' medical records were used to obtain baseline demographics (age, sex) and clinical data (cognitive capacity, depression, frailty, and pain). Baseline measures were based on validated instruments from the RAI-MDS 2.0 (Canadian Institute for Health Information, 2018): including the Cognitive Performance Scale, Depression Rating Scale, and Pain Scale. Additionally, Palliative Performance Scale scores were collected. Health care providers' progress notes from the study period were examined for non-structured qualitative feedback and observations during the VR sessions.
2) The intervention data-collection tool, which comprised four sections to gather data on the outcome measures (see Supplementary Material S2 for the complete document):
a) Open-ended questions for the RT to answer relating to the outcomes of interest: pain and responsive behaviors.
b) A structured observation guide (5-point Likert scale, open-ended comments, Yes/No) completed by RTs to record their perceptions of VR's impact on a resident during a session. This included perceptions of enjoyment, relaxation, and discomfort determined through participants' facial expressions, gestures, and vocalizations during and immediately after the VR session.
c) The Pain Assessment for Advanced Dementia (PAINAD) Scale (Warden et al., 2003) (administered prior to sessions).
d) Open-ended questions for the participant to answer relating to the outcomes of interest: acceptability, enjoyment, and willingness to use VR again. When participants were unable to provide feedback, the RTs recorded their impressions of residents' reactions when possible. Given their familiarity with the residents, RTs were confident in assessing participants' experiences of relaxation, distress, and (dis)comfort.
Outcome Measures
A) The impact of VR therapy on responsive behaviors was evaluated based on the occurrences of responsive behaviors and the frequency of administering pro re nata (PRN) pain medications (e.g., acetaminophen, hydromorphone, morphine, etc.) during and immediately after the sessions. Information captured from the ongoing progress notes recorded during the study period also served to validate the impact on responsive behaviors based on the need for medication/altered care during the study period.
B) The impact of VR therapy on physical and/or emotional pain was determined by RT report on subjective participant feedback during sessions and RT observations of participant's pain level immediately preceding and during the intervention, using the PAINAD scale.
C) Acceptability of the technology was determined by the duration of exposure and ability to tolerate the headset, resident responses to questions about feeling good, (dis)liking the experience, or finding the devices (un)comfortable, as well as RTs' observations of actions like trying to remove the headset.
D) Enjoyment during/from VR therapy was operationalized through positive vocalizations (oohs, ahs, giggling, or saying), engagement with/in VR (gestures: pointing, lifting feet, moving hands), actions indicating relaxation (muscle relaxation in the face or body, deep exhaling), and participants' responses to questions such as “Did you like what you just saw in VR?” as well as their willingness and desire to try VR again.
E) Reminiscence was operationalized as instances of participant recollection of past events, places, or activities initiated by viewing the VR experiences, as recorded and documented by the RTs.
Analysis
Demographic and baseline data were reported by participants, whereas descriptive statistics drawn from the quantitative survey data (relating to the outcomes of interest: acceptability, enjoyment, reminiscence, pain, and responsive behaviors) were reported by VR therapy session. The analysis of the quantitative survey data was performed by session type (scheduled or targeted), with the goal of determining the impact of VR therapy for each condition. Progress notes (unstructured text) extracted from the EPR were imported into NVivo 11 (QSR International, LLC) and underwent thematic analysis using grounded theory and the constant comparison method. Findings from these qualitative observations were used to validate and expand on the quantitative results.
Results
Entire raw dataset if requested. Not currently appended as a Supplementary Material.
Sessions
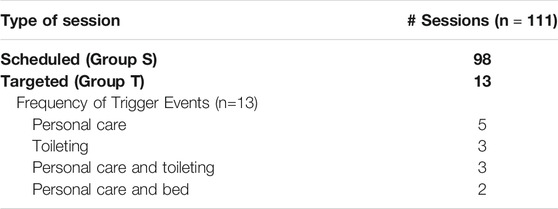
The most common triggers were events related to personal care, which contributed to most (77%, 10/13) instances of the targeted sessions. Table 2 provides the list of trigger events and their observed frequencies in the study.
A total of 111 VR therapy sessions took place, of which 88% (98/111) were scheduled and conducted as recreational activities (Group S), and 12% (13/111) were targeted and conducted at times of a trigger activity that predicted responsive behaviors (Group T). All participants (100%, 33/33) chose to participate in at least a second VR therapy session when given the opportunity (range = 2–5 sessions per participant).
Managing Responsive Behaviors
There was no need to administer PRN pain/sedating medication to resident participants during any of the VR therapy study sessions (n = 111). In the majority of sessions (61%, 68/111), no responsive behaviors were observed during or shortly after VR therapy. In 34% (38/111) of sessions, at least one responsive behavior was observed. The responsive behaviors identified in these 38 sessions were categorized as follows: wandering (63%, 24/38), agitation (21%, 8/38), negative affect (e.g., sadness, worriedness, etc.) (8%, 3/38), a combination of wandering and negative affect (5%, 2/38), and related to the activity of packing their belongings (3%, 1/38). In a minority of sessions, RTs were not sure if a responsive behavior occurred (4%, 4/111) or did not fill out a response (1%, 1/111). In 46% (6/13) of targeted sessions, participants did not exhibit the responsive behaviors usually triggered by the identified environmental event (e.g., bathing, wound dressing, etc.).
A more nuanced depiction of the impact of VR on behaviors can be drawn from some of the anecdotes documented in the HCP/RT notes. For example, when commenting on the level of agitation experienced by a resident, it was reported of the scheduled VR session that “His face looked calmer [than] when we started and he had a smile. He was not smiling when we started […] A regular PSW commented how calm and happy he looked. A regular RPN also commented [on] how calm and happy [the] resident looked.” A number of HCP notes described a positive impact on residents that were wandering when VR was administered; for example, one note reported: “Resident was [approached] crying, and ended in a good mood” similarly, another note reports “became engaged in VR … did relax for about 30 min and then fell asleep.” One HCP note captured multiple sessions with the same resident and reported that by the second session, “[the resident] remembered from the day before and was happy to have the visit. Had VR therapy for 3 min and appeared to really enjoy it. She was then in a good mood, and it seemed to have raised her spirits. When I came back 5 min later, the resident was still smiling, in good spirits.” Overall qualitative analyses of the notes indicated that HCP/RTs observed many positive experiences administering VR therapy in both targeted and scheduled sessions and felt VR therapy was effective in some cases for redirecting symptoms of aggression, improving mood, and boosting alertness.
Managing Pain
Participant pain level was assessed before the VR session using the PAINAD instrument, where scores range from 0 = no pain, 1–3 = mild pain; 4–6 = moderate pain; and 7–10 = severe pain. See Figure 3. No PRN medications (e.g., acetaminophen, hydromorphone, morphine, etc.) were required or administered for pain during any of the VR therapy sessions. Participants in the targeted sessions had higher PAINAD scores on average (M = 1.54) than those in scheduled sessions (M = 0.78) when the session began.
Acceptability and Comfort
Although the lengths of time the participants wore the HMD was not precisely documented, analysis of the progress notes provided some insights. The duration of VR experiences fell into two broad categories: 1) brief exposure, e.g., 2–5 s, which indicated the participant was willing to try VR (i.e., raising the headset to their eyes and glancing very briefly), and 2) longer exposure, e.g., over 5 min, which constituted sufficient time to “experience” VR and engage with the content. The longest documented exposure was 35 min, during a targeted session, described in the progress notes as follows:
Resident had headset on throughout care. Halfway through, the headset fell off the resident, and the RT asked the resident if they would like it back on, and the resident stated yes. Headset remained on throughout 30 min of care and 5 min after. Resident was humming a few times through the session as well as looking around at the fall forest video he was watching. Resident was calm throughout. Resident did not have any behaviors.
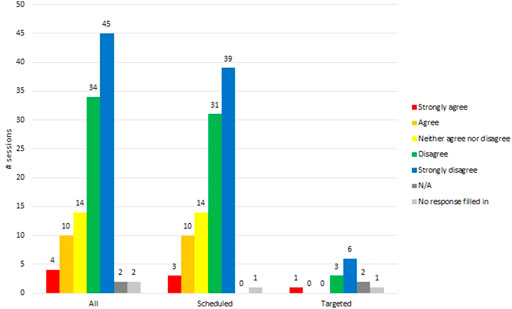
In the majority (71%, 79/111) of sessions, RTs selected either strongly disagree (45/111) or disagree (34/111), in response to the question: resident “disliked VR (they try to remove the HMD, they vocalize negatively.” See Figure 4.
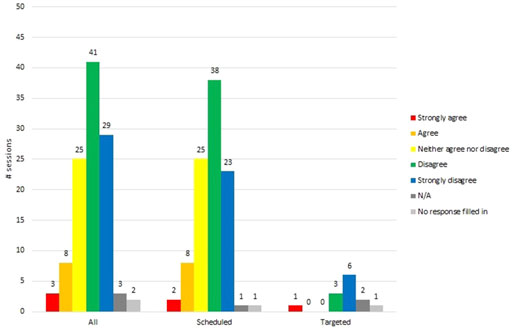
In 63% (70/111) of sessions, participants found the VR HMD comfortable. This was based on RTs selecting strongly disagree (29/111) or disagree (41/111) to the question: was the resident “uncomfortable in VR?” See Figure 5.
The clinical progress notes revealed several reasons for discomfort experienced by some participants, notably the elastics used to secure the HMD on the head, poor image focus, and difficulty accommodating glasses under the HMD. In one case it appeared that the tension from the strap was bothersome: “Resident accepted the headset, but did not allow the RT to put the elastic band around his head. The RT put the headset up to his face.” In another instance the elastics pulled on a residents' hair: “Resident agreed to try [the] VR headset. RT put it up to his eyes; this did not bother him. RT then tried to place the head strap over the resident's head. It pulled his hair and made the resident uncomfortable.” These challenges were overcome with strategies such as the RT holding the headset up to the resident's eyes without placing it on their head. RTs reported that residents generally had the ability to handle the headset on their own. One note recorded that the “Resident's duration of use has shown to increase in comparison to previous sessions. Resident would on occasion lift the headset up to peek out from the headset but would place the headset back on his own head.” In another session “The resident held onto the headset (no straps were used) and looked up, down, and from side to side. No prompting was needed. When [the] resident pulled the headset back from his eyes, they were wide and bright. Resident was enjoying the experience. When [the] session was over [the] resident was in a good mood and appreciated the visit.”
In terms of mobility, progress notes indicate that many residents were able to move their head and neck to make use of the 360° visuals. In some cases, residents continued to ambulate (under supervision) with the HMD on, as noted in these examples: “During both attempts resident was able to self-propel his wheelchair in the hallway”; “Resident was pointing and moving her head and chair around to see more of the video. She said she felt like they were really there.”
Another theme that emerged from the progress notes was about the helpfulness of involvement of informal caregivers in the provision of VR therapy. For example, in one report, the “POA tried many times over the 30 min visit to get her father to open [his] eyes to look at the VR. She was successful twice, but for very short periods of time. The POA then suggested trying a session in the evening and after his bath as he is more alert then.”
Enjoyment From the Experience
Almost half (47%, 52/111) of residents were perceived to feel good or better from watching VR. See Table 3 for RTs reported answers to the question “Did VR therapy make the resident feel good or better?” than they did before the session. Likewise, interviewed RTs indicated that VR therapy did not induce more negative moods and often improved residents' moods in the moment and after the session (Appel et al., 2021b).
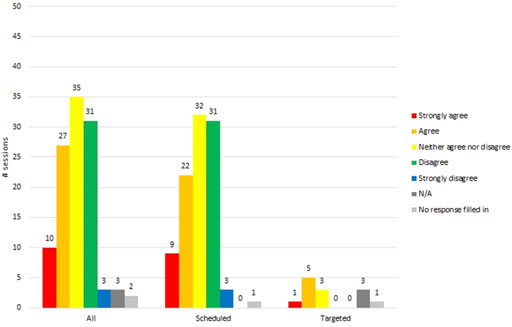
In 33% (37/111) of sessions, RTs strongly agreed (10/111) or agreed (27/111) that VR “helped the resident relax (muscle relaxation in the face or body, deep exhaling)” (see Figure 6).
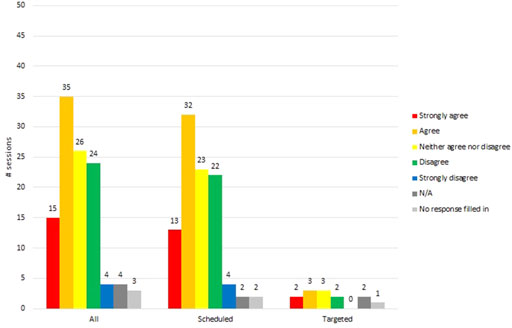
Many (45%, 50/111) VR sessions produced signs of enjoyment, as determined from RTs selecting strongly agree (15/111) or agree (35/111) to the question: resident “enjoyed VR (vocalization: oohs, ahs, giggling, or saying)?” See Figure 7.
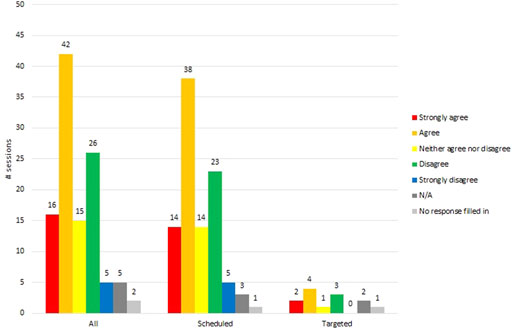
The majority (52%, 58/111) of sessions saw participants engaged with VR through gestures, pointing, and body movement, as determined from RTs selecting strongly agree (16/111) or agree (42/111) to the question: resident “engaged with VR (gestures: pointing, lifting feet, moving hands)?” (see Figure 8).
The progress notes documented several examples of physical displays of relaxation and enjoyment, such as “RT showed the resident a slow-moving slideshow of dogs. She really enjoyed it. Resident was sitting in her recliner. Her feet started on the floor. She then crossed her legs, then started rocking in her chair. Very relaxed. Resident did have her hands on the sides of the headset but talked about and pointed at the different images.” Another note reported, “His face looked calmer than when we started, and he had a smile. He was not smiling when we started. Writer thanked the resident for his time. A regular [personal support worker] commented how calm and happy he looked. A regular [registered practical nurse] also commented on how calm and happy the resident looked.”
Progress notes revealed that VR therapy could contribute to enjoyment by creating opportunities for residents to converse with each other about what they saw and experienced in VR. For example, one note stated that the “Resident enjoyed the videos. He was quick to look at each one. Then a co-resident who was also part of the study joined us at the table. The headset was shared between residents and they discussed what they saw. The first resident spoke about the headset like they had seen it before and was aware of it when it was placed on the other resident.” In a similar scenario, two co-residents were sitting at a table while one was experiencing VR; the progress note records: “This time different videos were shown. Now that there was conversation between three of us, the resident was more interested in the headset and what was on it. She found the session more interesting this time.” Some participants found VR experiences so entertaining that they said they would promote the program: “Resident was happy after the session. Writer brought her back to her room. She said she would tell the others she saw cows today.”
VR Content
In over three quarters (76%, 84/111) of sessions, residents responded “Yes” to the question “Did you like what you just saw in VR?” In only 8% (9/111) of sessions, residents responded, or RTs interpreted their response as a “No,” to this question; cited reasons included a brief viewing period (22%, 2/9), disinterest (22%, 2/9), disliking the visual content (22%, 2/9), blurry vision (11%, 1/9), or an unspecified reason (11%, 1/9).
Data from the progress notes supported the observational and survey data with rich commentary. For example, in one report, the “Resident responded positively to the beach and forest footage.” Resident tried to grab at things while the headset was on and voiced “This is something, isn't it?” Many residents commented on what they saw; one example was noted as follows: Each time resident engaged in conversation on what he saw, looked around, and when looking at the sunny beach resident attempted to put on his sunglasses as he stated “It is a bright sunny day” Even when describing the visuals was a cognitive challenge for some participants, they still appeared to enjoy the experience “She was amazed at what she saw. She described the cows she was looking at but could not think of the word ‘cow’ to describe them. The same happened when showing her a video with a rabbit in snow. Resident enjoyed the video and time spent together.” For some, the experience was even memorable from one session to the next, as was reported in this note: RT got the VR headset and asked resident if he wanted to “see the movie in the headset?” Resident voiced “I've seen that before; I know it's good!”
In over half (52%, 44/84) of the sessions where residents responded that they liked what they saw in VR, they further elaborated on what they liked about it. Visual content (e.g., animals, landscapes, nature, people) was by far cited most frequently (82%, 36/44), followed by elements of the experience (e.g., realism, reminiscence) (14%, 6/44), and emotions that were elicited (e.g., interest, engagement, calmness, happiness) (11%, 5/44).
In two-thirds (67%, 74/111) of the sessions, when asked, the residents explicitly stated they wanted to try VR therapy again. In a minority of sessions, residents noted uncertainty (22%, 24/111) or declines (5%, 6/111) towards trying VR therapy again. When residents declined, no explanations were given as to why. When they were “not sure,” cited reasons included: resident being unimpressed/disinterested (29%, 7/24), finding the HMD too heavy (4% 1/24), afternoon sessions being less favorable than the morning sessions (4% 1/24), and the resident only wanting a social visit (4% 1/24).
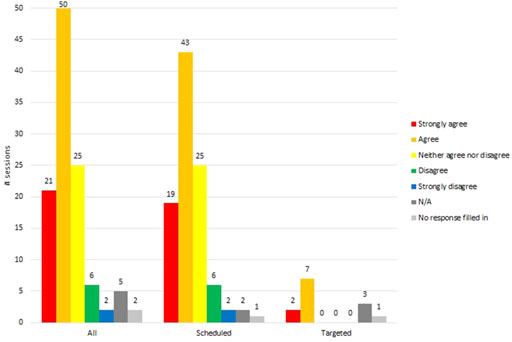
When asked of the RTs, their perception of residents' willingness to try VR again produced similar results, with almost two-thirds (64%, 71/111) of RTs selecting strongly agree (21/111) or agree (50/111) to the statement “Resident would like to try VR again” (see Figure 9). Session notes indicated that residents' overall level of interest in VR therapy increased when the content was personally relevant to the resident and that the RTs were able to use the headset flexibly during sessions in order to show videos to participants that would be personally meaningful (e.g., specific animals, fall leaves).
Reminiscence
In a third (33%, 37/111) of sessions, residents appeared to reminisce during and/or after VR therapy, which was observed in both the scheduled and the targeted sessions; this led RTs to consider VR therapy to be an effective conversation aid and prompting tool. Reminiscence was observed more frequently during the scheduled sessions (35%, 34/98) than the targeted sessions (23%, 3/13).
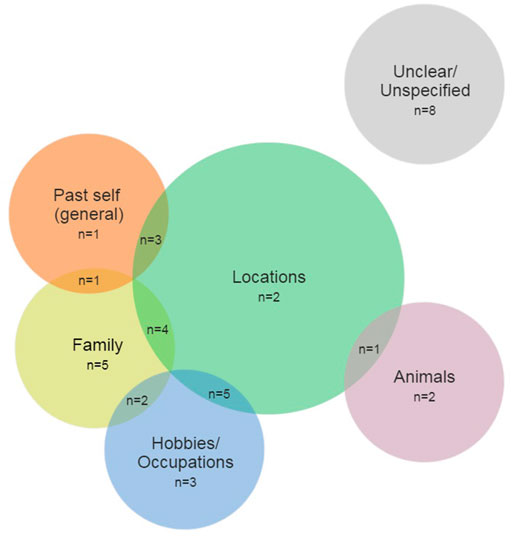
From most to least frequent, the emergent reminiscence themes were mentions of locations (15), family (12), hobbies/occupations (10), their past self in general (5), and animals (3). Of the 37 sessions where reminiscence occurred, participants typically (43%, 16/37) reminisced about two of the aforementioned themes. Figure 10 outlines frequency and co-occurrence of the emergent themes.
Findings from the progress notes mirrored the data collected during the sessions. Two of the most common reminiscence themes were mentions of family and aspects of childhood. The narratives shared by participants appear relatively detailed and specific, indicating that VR was a good trigger of memories. For example, one progress note stated “She viewed videos of the water and of cows eating in a barn. Resident compared the headset to a camera and taking pictures. This started her talking about her son-in-law and how he takes pictures.” In another example, it was noted “We then talked about the plants in her room and how one of her daughters has a passion for plants like her.” In terms of childhood memories, narratives were usually triggered by common scenery, like a farm or beach. “Resident started to share memories about taking an old horse to school as a child. Resident grew up on a farm.” Another participant “mentioned he grew up on a farm in Prince Edward County. The RT is also from this area, and this sparked a conversation about the area, his family, the farm he grew up on, and the ferries and bridges needed to get to the area.” In yet another example, the note stated “The warm southern beaches [she saw in VR]… reminded her of where she grew up. Resident then told a story of her father bringing her to a beach as a child. The resident then commented on people using the beaches to harvest coconuts and how this was destroying the natural beauty of the beach. Resident was happy with the visit and said she wished she had some money of her own to purchase a headset.”
Challenges With Implementing the Protocol
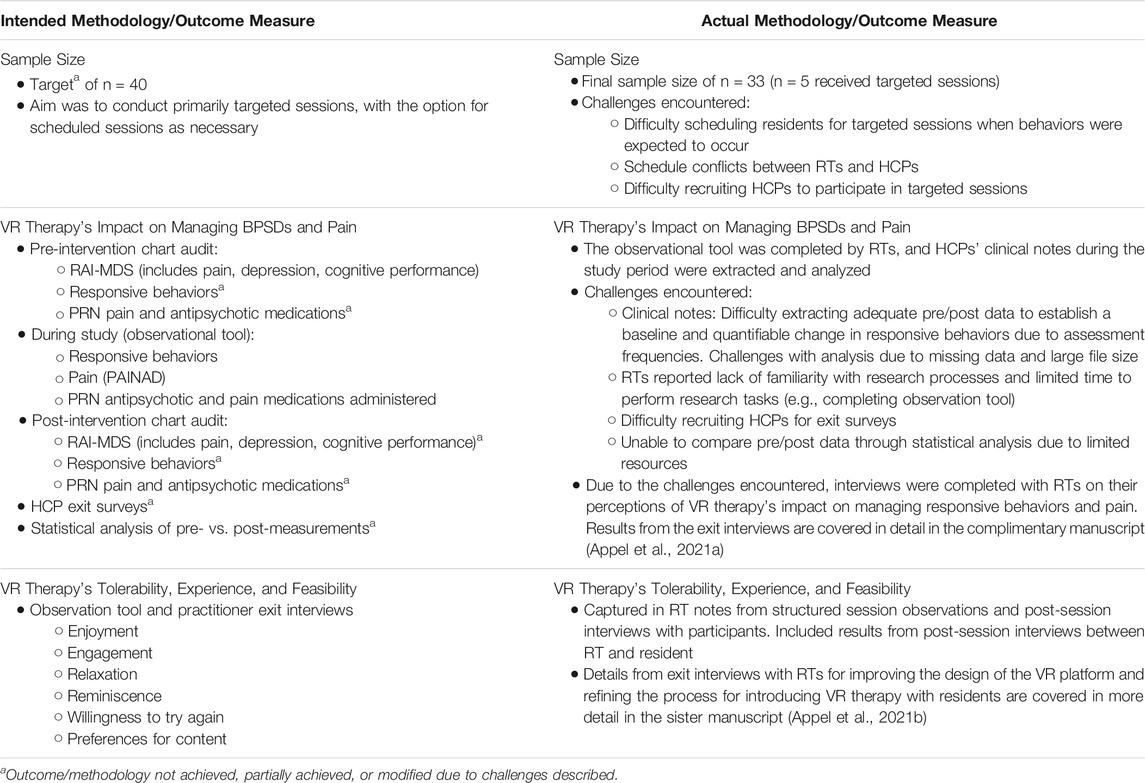
The key challenge that arose during this study was a lack of required personnel available at the time of a triggered session resulting in significantly fewer targeted VR therapy sessions than expected. While scheduled sessions involved only the RT and booking the session depended on the patient's and the RT's available times, targeted sessions required involvement and coordination of schedules for both the RT and the HCP, in addition to the triggering event timing constraint. Furthermore, responsive behaviors were not found to be as predictable as originally anticipated; eight participants were discharged from the study prior to receiving treatment, as clinical staff no longer observed consistent responsive behaviors. Data collection procedures were adapted during the study in order to accommodate the institutional workflow and processes.
Table 4 below highlights discrepancies between the intended and actual outcome measures and methodology. It is worthy of note that a separate and complementary study (Appel et al., 2021a), which consisted of in-depth interviews at the end of the trial, provides a detailed account of the RTs' perspectives and personal challenges, as well as their recommendations to help ensure a successful process and technical environment in future studies.
Discussion
In this study we aimed to evaluate the feasibility and potential benefits of introducing VR therapy to manage responsive behaviors in residents with dementia at a veterans' long-term care health center. More specifically, we looked at VR therapy in (1) “targeted sessions” concurrent with events known to trigger responsive behaviors, and (2) “scheduled sessions” conducted during recreational time.
In the study population sample, we observed only positive or neutral outcomes from VR therapy in terms of acceptability and its potential to reduce responsive behaviors, assist with pain management, and support enjoyment, relaxation, and reminiscence.
Scheduled VR therapy appears to meet feasibility requirements for long-term care residents with dementia, as staff at Perley Health continued to include VR therapy in their toolkit of resources beyond the study period and extended the offering to residents outside of the dementia ward. The program was interrupted during COVID but was preparing to resume in the fall of 2021.
Conversely, staff at Perley Health have not continued administering targeted sessions. This result is consistent with findings from Brimelow et al. (2020) who also experienced challenges with staffing, timing, and resident agitation when attempting to use VR as a diversion therapy for responsive behaviors that were already in progress. However, in spite of a number of challenges with implementing the protocol, there were positive outcomes from the targeted VR therapy sessions conducted in this study. A clear distinction must be made between the difficulty with administering a treatment/therapy and its potential to benefit the patient. Once participants in triggered sessions were wearing the headset, VR therapy appeared to have a pronounced calming effect in several of the participants. This is a critical preliminary finding that warrants further investigation into this non-pharmacological VR therapy administered at triggering events, since it can bear significant impact on how care is delivered for this population.
Given that much of the difficulty in evaluating triggered VR-sessions was associated with assembling sufficient staff at the appropriate time, an alternative way to overcome this specific aspect in future evaluations would be in home-based settings. Informal caregivers (e.g., family members) are generally dedicated to the care of one individual and do not encounter the same challenges with timing and scheduling as a multi-staff long-term care residence.
Previous studies have reported promising findings in the use of VR for frail older adults and for those with cognitive impairment (Appel et al., 2021c; D'Cunha et al., 2019; Silva et al., 2019). The current study examines, extends, and refines these findings in a few key ways. Participants in this study were veteran residents of a long-term care health center. In this population there is a high prevalence of PTSD, dementia, and responsive behaviors—significant causes of increased caregiver burden, distress, and PwD institutionalization. Participants had a documented diagnosis of dementia and history of responsive behaviors.
The lack of responsive behaviors or administration of pain medication during or immediately after the 111 VR therapy sessions leads to some assumptions. For scheduled sessions (n = 98), we can assume that this finding suggests VR therapy is unlikely to induce responsive behaviors. For targeted sessions (n = 13), where responsive behaviors were typically expected during the environmental event, we can assume that this finding suggests VR therapy could have reduced the frequency and/or severity of environmentally triggered responsive behaviors. While on average all participants enjoyed VR, it is of note that participants in the targeted sessions showed signs of relaxation during VR therapy more frequently. As scheduled sessions generally occurred when participants were not in a distressed mood and did not exhibit responsive behaviors, we can assume that there was less opportunity (or need) for relaxation interventions in this group.
On the other hand, reminiscence tended to occur more frequently during the scheduled sessions (35%, 34/98) than the targeted sessions (23%, 3/13). A possible explanation for this is that when VR experiences are used as a means of distraction to help manage responsive reactions during an environmental trigger event, participants may not be in a state of mind to feel removed enough from their real-world sensory experiences to allow for reminiscence.
In terms of acceptability, our findings mirror what was reported in previous studies with similar populations; newer models of VR headsets (i.e., Oculus Go) are feasible for use with older adults with dementia (including moderate and severe cognitive impairment) who exhibit responsive behaviors (Appel et al., 2021d; Brimelow et al., 2020). Feedback regarding the comfort of the HMD was generally positive, however, some aspects can be improved/customized to suit the unique needs of this population.
In line with previous studies (D'Cunha et al., 2019; Paletta et al., 2020), our findings suggest VR therapy is enjoyable for many PwD in long-term care. As regular participation in enjoyable activities is associated with better psychological and physical functioning in the general population (Pressman et al., 2009), our findings also support the notion that VR therapy has potential to have positive effects on general wellbeing and quality of life. In the majority (67%, 74/111) of sessions, participants stated their desire to experience VR therapy again, and notably, when given the opportunity, all participants engaged in VR therapy at least a second time. Furthermore, nearly half of participants (45%, 50/111) showed clear signs of enjoyment or felt better after VR therapy than before (52/111, 47%). These results are meaningful in comparison to pharmacological-based therapies trials; in a review of 10 placebo-controlled trials of cholinesterase inhibitors (CIs, a drug class encompassing four of the five approved drugs for mild-to-moderate stage Alzheimer's dementia), Casey et al. (2010) found that only approximately half of participants showed an immediate response.
Lastly, results demonstrated the value of capturing the support, experiences, opinions, and suggestions of RTs who work regularly with the residents to help design tailored VR therapy and content. Animals and landscapes were the types of content most frequently enjoyed by residents, and the latter was most often mentioned as a theme of reminiscence. However, preferences for content were unique, and content that was personally relevant was observed to relate to more positive experiences with VR. Incorporating participation and input from informal caregivers (family and friends) was also found to be helpful; informal caregivers are often the best supports and advocates for their loved ones with dementia, and similar to the RTs, they are able to help determine in which situations VR therapy may be useful and to what extent behaviors are being managed.
Recommendations
We experienced a number of challenges with implementing the protocol that we hope will serve to assist researchers in designing studies to further evaluate the impact of targeted VR therapy on responsive behaviors in individuals with dementia. First, our findings highlight the importance of researchers conducting studies in situ dedicating significant effort towards designing protocols that take into account existing staff work-flows and familiarity conducting research. In long-term care, co-design of study protocol with the HCPs involved in the study could be seen as a way to mitigate potential challenges with scheduling and with HCP willingness to participate in research activities.
Given the potential impacts on quality of life for both individuals with dementia and their caregivers, examining VR therapy's broader impact on responsive behaviors and wellbeing will be essential to understanding its effectiveness. In this study, residents participated in two to five sessions of VR therapy, and while “in the moment” improvements in mood and anxiety levels were apparent for many residents, we were not able to measure the impact of VR therapy exposure outside of sessions. When measuring the impact of VR therapy, researchers should consider the appropriateness of certain measures, even validated questionnaires (e.g., the Neuropsychiatric Inventory, which is administered every 4–6 weeks) (Connor et al., 2008), given that changes due to VR are likely to be observed over relatively short periods of time by a rotating collection of professional caregivers. Relatedly, it would be of value to explore “dosing” of VR therapy (i.e., the impact of different lengths and frequencies of exposure) and the duration or “washout period” of the benefits of VR therapy.
Additionally, future studies should focus on evaluating the effect of VR therapy on responsive behaviors triggered by different causes (e.g., emotional distress, physical pain, initiation of care, etc.). Our findings suggest that VR therapy may be more beneficial for managing physical pain compared to emotional pain, and this will need to be investigated systematically.
In terms of hardware, we found a number of areas where HMDs can be improved to better suit this population. Pressure on the forehead, worsened by use of the soft elastic straps, caused distress in some participants, and many RTs preferred to administer VR therapy by holding the headset up to the participants' eyes. Hardware designers should consider developing lighter-weight HMDs, increasing video resolution, and improving the mechanism to affix the headset (i.e., an alternative to soft elastic straps).
Regarding the software, the VR library used for this study did not cover the broad range of participant interests observed (e.g., passive park scenes versus active scenes featuring animals, personally relevant locations), though RTs were able to use the headset flexibly to find videos (of which some were not 360°) to show participants. VR film makers should consider creating libraries of vetted experiences for this population that are both safe (e.g., not over-stimulating, unlikely to induce simulator sickness, etc.) and engaging, covering a broad set of interests. Future studies should also systematically examine the library to see what features of videos are most effective in this population for reducing responsive behaviors, helping to manage pain, and for promoting quality of life. Another avenue to explore is the value of personalized content and shared experiences (multiple people simultaneously watching the same virtual environment).
Finally, many PwD experience a significant decline in communication abilities as their symptoms progress. Barriers in communication can impair relationships with caregivers, leading to loneliness, social isolation, and poor quality of life for both parties. As a significant number of participants reminisced during the VR therapy sessions, there is opportunity to explore the use of this technology as a tool for facilitating communication between PwD and caregivers. Given the potential impacts on quality of life for both individuals with dementia and their caregivers, future studies should focus on evaluating the effect of VR therapy on the wellbeing of caregivers with relation to the therapy's impact on reducing responsive behaviors.
Limitations
The results of this study should be interpreted with caution due to limitations in the data. The research team was aware of some of these methodological shortcomings when proposing the trial and attempted to work within what was feasible given the available institutional resources.
An important limitation is that a number of study metrics were not captured directly before and after the intervention (i.e., baseline and follow-up data on responsive behaviors and medications administered at triggering events, PAINAD) making it difficult to infer causal conclusions about the impact of VR on these outcomes. Additionally, this study did not include a comparison intervention, and the impact of VR (i.e., changes in participant behaviors and emotions) was based on RTs' observations, with associated bias. Participants in this study were not blind to treatment assignment, a limitation often present in e-health trials. Also, e-health trials, including the current one, often collect a large number of outcomes which increases the risk of Type I error. Moreover, it is possible that some of the VR therapy benefits observed were affected by the social contact with the RT, and we were not able to demonstrate an advantage over other similar recreation-based interventions (e.g., watching 360° videos on an iPad, music therapy).
Finally, as conducting targeted sessions was resource-intensive, only a small subset of the participants (n = 5) were involved in targeted sessions where responsive behaviors were expected to occur. Insights and feedback received from RTs have indicated that their emotional reactions, such as hesitation to place the VR headset on someone frail and in distress, resulted in missed opportunities to fully evaluate the impact of targeted VR therapy. Nevertheless, small samples may still be appropriate for aims such as testing aspects of feasibility, provided that the sample is representative of the target population (Thabane et al., 2010; Moore et al., 2011). The present study was conducted in situ, and baseline data on the sample demonstrate that participants were representative of frail residents with dementia.
Conclusion
Findings indicate that VR therapy is acceptable and enjoyable for veterans living with dementia with varying degrees of cognitive and physical impairments. Staff at Perley Health continued to use scheduled VR therapy as a recreational tool beyond the study period. Despite difficulties in conducting targeted VR therapy sessions, there was observational evidence of potential to reduce environmentally triggered responsive behaviors in some residents. This warrants further investigation into both treatment effectiveness and measures to increase evaluation feasibility in long-term care settings. Finally, manufacturers and providers of VR therapy should consider ways in which content, equipment, and administration can be customized and optimized for this particularly frail population.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics Statement
The studies involving human participants were reviewed and approved by The Office of Research Ethics, York University. The patients/participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author Contributions
LA, EA, and LS conceived of the study and designed the protocol. EA, EK and SL conducted the data analysis. LA wrote the first draft, and it was reviewed and edited by all the authors.
Funding
The Centre for Aging and Brain Health Innovation generously awarded the Spark Research Grant to fund this study.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
We would like to thank the Centre for Brain Health and Innovation for their generous support. We are grateful to Codrin Talaba for working with our research team to film, edit, and provide the VR experiences for this study. Finally, we would like to acknowledge the Therapeutic Recreation and Creative Arts Program at the Perley Health for collaborating on this study and administering the VR sessions as well as the Perley Health Centre of Excellence for supporting this research.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/frvir.2021.724020/full#supplementary-material
References
Agens, J. (2010). Chemical and Physical Restraint Use in the Older Person. BJMP. 3, 302. Available at: https://www.bjmp.org/files/2010-3-1/bjmp-2010-3-1-302.pdf (Accessed March 31, 2021).
Alzheimer’s Society of Canada (2019). Conversations About: Dementia and Responsive Behaviours. Alzheimer Society of Canada. Available at: https://alzheimer.ca/sites/default/files/documents/conversations_dementia-and-responsive-behaviours.pdf.
Appel, L., Appel, E., Bogler, O., Wiseman, M., Cohen, L., Ein, N., et al. (2020a). Older Adults With Cognitive And/or Physical Impairments Can Benefit From Immersive Virtual Reality Experiences: A Feasibility Study. Front. Med. 6. doi:10.3389/fmed.2019.00329
Appel, L., Kisonas, E., Appel, E., Klein, J., Bartlett, D., Rosenberg, J., et al. (2020b). Introducing Virtual Reality Therapy for Inpatients with Dementia Admitted to an Acute Care Hospital: Learnings from a Pilot to Pave the Way to a Randomized Controlled Trial. Pilot Feasibility Stud. 6, 166. doi:10.1186/s40814-020-00708-9
Appel, L., Ali, S., Narang, T., Mozeson, K., Pasat, Z., Orchanian-Cheff, A., et al. (2021a). VR to Promote Wellbeing in Persons with Dementia: A Scoping Review. J. Rehabil. Assistive Tech. Eng. doi:10.1177/20556683211053952
Appel, L., Appel, E., Kisonas, E., and Lewis, S. (2021b). The Development of ObsRVR: Observational Instrument to Measure Reactions of People with Dementia Experiencing Virtual Reality [Conference presentation]Clinical Trials in Alzheimer's Disease congress. Boston, MA, United States. Available at: https://convention.apa.org/2019-video.
Appel, L., Appel, E., Kisonas, E., Pasat, Z., Mozeson, K., Vemulakonda, J., et al. (2021c). Virtual Reality for Veteran Relaxation (VR2) - Introducing VR-Therapy for Veterans With Dementia - Challenges and Rewards of the Therapists Behind the Scenes. Front. Virtual Real. 2 (2), 104. doi:10.3389/frvir.2021.720523
Appel, L., Kisonas, E., Appel, E., Klein, J., Bartlett, D., Rosenberg, J., et al. (2021d). Administering Virtual Reality Therapy to Manage Behavioral and Psychological Symptoms in Patients With Dementia Admitted to an Acute Care Hospital: Results of a Pilot Study. JMIR Form Res. 5, e22406. doi:10.2196/22406
Beason-Held, L. L., Goh, J. O., An, Y., Kraut, M. A., O'Brien, R. J., Ferrucci, L., et al. (2013). Changes in Brain Function Occur Years Before the Onset of Cognitive Impairment. J. Neurosci. 33, 18008–18014. doi:10.1523/jneurosci.1402-13.2013
Brimelow, R. E., Dawe, B., and Dissanayaka, N. (2020). Preliminary Research: Virtual Reality in Residential Aged Care to Reduce Apathy and Improve Mood. Cyberpsychology, Behav. Soc. Networking. 23, 165–170. doi:10.1089/cyber.2019.0286
Canadian Institute for Health Informatics (2013). Describing Outcome Scales (RAI-HC). [Adapted from InterRAI Corporation (1999)] Washington: InterRAI. Available at: https://www.cihi.ca/sites/default/files/document/outcome_rai-mds_2.0_en.pdf.
Canadian Institute for Health Information (2018). Continuing Care Reporting System. Ottawa: RAI-MDS Output Specifications, 2018–2019. Available at: https://www.cihi.ca/en/access-data-and-reports.
Canadian Institute for Health Information (2019). Internal RAI-MDS Report Ottawa: Canadian Institute for Health Information. unpublished.
Casey, D. A., Antimisiaris, D., and O'Brien, J. (2010). Drugs for Alzheimer's Disease: Are They Effective? P T: A peer-reviewed J. formulary Manag. 35 (4), 208–211.
Clay, F., Howett, D., Fitzgerald, J., Fletcher, P., Chan, D., and Price, A. (2020). Use of Immersive Virtual Reality in the Assessment and Treatment of Alzheimer's Disease: A Systematic Review. Jad. 75, 23–43. doi:10.3233/jad-191218
Connor, D. J., Sabbagh, M. N., and Cummings, J. L. (2008). Comment on Administration and Scoring of the Neuropsychiatric Inventory in Clinical Trials. Alzheimer's Demen. 4, 390–394. doi:10.1016/j.jalz.2008.09.002
D’Cunha, N. M., Nguyen, D., Naumovski, N., McKune, A. J., Kellett, J., Georgousopoulou, E. N., et al. (2019). A Mini-Review of Virtual Reality-Based Interventions to Promote Well-Being for People Living with Dementia and Mild Cognitive Impairment. Gerontology. 65 (4), 430–440. doi:10.1159/000500040
Dermody, G., Whitehead, L., Wilson, G., and Glass, C. (2020). The Role of Virtual Reality in Improving Health Outcomes for Community-Dwelling Older Adults: Systematic Review. J. Med. Internet Res. 22, 22. doi:10.2196/17331
Dyer, S. M., Harrison, S. L., Laver, K., Whitehead, C., and Crotty, M. (2017). An Overview of Systematic Reviews of Pharmacological and Non-Pharmacological Interventions for the Treatment of Behavioral and Psychological Symptoms of Dementia. Int. Psychogeriatr. 30, 295–309. doi:10.1017/s1041610217002344
Edvardsson, D., Sandman, P. O., Nay, R., and Karlsson, S. (2008). Associations Between the Working Characteristics of Nursing Staff and the Prevalence of Behavioral Symptoms in People With Dementia in Residential Care. Int. Psychogeriatr. 20, 764–776. doi:10.1017/s1041610208006716
Erkes, J., Camp, C. J., and Bayard, S. (2021). Don't Bother Trying, They Won't Do it! Effect of Responsive Behaviors on the Montessori Assessment System. Clin. Gerontologist., 1–8. doi:10.1080/07317115.2021.1924333
Finkel, S. I., and Cooler, C. (1996). Clinical Experiences And Methodologic Challenges In Conducting Trials In The Behavioural Disturbances Of Dementia. Intern. Psycho. Association 8 (2), 151–164. doi:10.1017/S1041610297003311
Fulton, J. J., Calhoun, P. S., Wagner, H. W., Schry, A. R., Hair, L. P., Elbogen, E., et al. (2015). The Prevalence Of Posttraumatic Stress Disorder In Operation Enduring Freedom/Operation Iraqi Freedom (OEF/OIF) Veterans: A Meta-Analysis. J. Anxiety Disor. 31, 98–107. doi:10.1016/j.janxdis.2015.02.003
García-Betances, R. I., Arredondo Waldmeyer, M. T., Fico, G., and Cabrera-Umpiérrez, M. F. (2015). A Succinct Overview of Virtual Reality Technology Use in Alzheimer's Disease. Front. Aging Neurosci. 7, 780. doi:10.3389/fnagi.2015.00080
Gastmans, C., and Milisen, K. (2006). Use of Physical Restraint in Nursing Homes: Clinical-Ethical Considerations. J. Med. Ethics. 32, 148–152. doi:10.1136/jme.2005.012708
Gómez-Romero, M., Jiménez-Palomares, M., Rodríguez-Mansilla, J., Flores-Nieto, A., Garrido-Ardila, E. M., and González-López-Arza, M. V. (2017). Benefits of Music Therapy on Behaviour Disorders in Subjects Diagnosed with Dementia: a Systematic Review. Neurología (English Edition). 32 (4), 253–263. doi:10.1016/j.nrleng.2014.11.003
Guideline Adaptation Committee (2016). Clinical Practice Guidelines and Principles of Care for People with Dementia Technical Report Volume 1. NHMRC Partnership Centre for Dealing with Cognitive and Related Functional Decline in Older People. Available at: https://cdpc.sydney.edu.au/wp-content/uploads/2019/06/dementia-guideline-technical-report-volume-1_Final_for-online-2.pdf (Accessed March 31, 2021).
Günak, M., Billings, J., Carratu, E., Marchant, N., Favarato, G., and Orgeta, V. (2020). Post-Traumatic Stress Disorder As A Risk Factor For Dementia: Systematic Review And Meta-Analysis. British J. Psych. 217 (5), 600–608. doi:10.1192/bjp.2020.150
Gunawardena, R., and Smithard, D. G. (2019). The Attitudes towards the Use of Restraint and Restrictive Intervention Amongst Healthcare Staff on Acute Medical and Frailty Wards-A Brief Literature Review. Geriatrics. 4, 50. doi:10.3390/geriatrics4030050
Islam, M. S., Baker, C., Huxley, P., Russell, I. T., and Dennis, M. S. (2017). The Nature, Characteristics and Associations of Care home Staff Stress and Wellbeing: a National Survey. BMC Nurs. 16, 22. doi:10.1186/s12912-017-0216-4
Kishita, N., Backhouse, T., and Mioshi, E. (2020). Nonpharmacological Interventions to Improve Depression, Anxiety, and Quality of Life (QoL) in People with Dementia: an Overview of Systematic Reviews. J. Geriatr. Psychiatry Neurol. 33 (1), 28–41. doi:10.1177/0891988719856690
Liu, Y., Tan, W., Chen, C., Liu, C., Yang, J., and Zhang, Y. (2019). A Review of the Application of Virtual Reality Technology in the Diagnosis and Treatment of Cognitive Impairment. Front. Aging Neurosci. 11, 280. doi:10.3389/fnagi.2019.00280
Matsangidou, M., Schiza, E., Hadjiaros, M., Neokleous, K. C., Avraamides, M., Papayianni, E., et al. (2020). Dementia: I Am Physically Fading. Can Virtual Reality Help? Physical Training for People With Dementia in Confined Mental Health Units. Des. Approaches Supporting Tech. 12188, 366–382. doi:10.1007/978-3-030-49282-3_26
Meyer, C., and O’Keefe, F. (2018). Non-Pharmacological Interventions for People with Dementia: A Review of Reviews. Dementia. 19, 1927–1954. doi:10.1177/1471301218813234
Miyamoto, Y., Tachimori, H., and Ito, H. (2010). Formal Caregiver Burden in Dementia: Impact of Behavioral and Psychological Symptoms of Dementia and Activities of Daily Living. Geriatr. Nurs. 31, 246–253. doi:10.1016/j.gerinurse.2010.01.002
Moore, C. G., Carter, R. E., Nietert, P. J., and Stewart, P. W. (2011). Recommendations for Planning Pilot Studies in Clinical and Translational Research. Clin. translational Sci. 4 (5), 332–337. doi:10.1111/j.1752-8062.2011.00347.x
National Collaborating Centre for Mental Health (2007). Dementia: A NICE-SCIE Guideline on Supporting People with Dementia and Their Carers in Health and Social Care. London: British Psychological Society/Gaskell.
ONeil, M. E., Freeman, M., Christensen, V., Telerant, R., Addleman, A., and Kansagara, D. (2011). A Systematic Evidence Review of Non-pharmacological Interventions for Behavioral Symptoms of Dementia. Washington: Department of Veterans Affairs.
Paletta, L., Schüssler, S., Kober, S. E., Zweytik, E., Steiner, J., Andreu, J. P., et al. (2020). Virtual Reality-Based Mindfulness Training, Sensory Activation and Mental Assessment in Dementia Care. Alzheimer's Demen. 16 (Suppl. 7), e047344. doi:10.1002/alz.047344
Pelletier, I. C., and Landreville, P. (2007). Discomfort And Agitation In Older Adults With Dementia. BMC geriatrics 7, 27. doi:10.1186/1471-2318-7-27
Pinciotti, C., Bass, D., McCarthy, C., Judge, K. S., Wilson, N. L., Morgan, R. O., et al. (2017). Negative Consequences of Family Caregiving for Veterans With PTSD and Dementia. J. Nervous Mental Dis. 205 (106), 106–111. doi:10.1097/NMD.0000000000000560
Pressman, S. D., Matthews, K. A., Cohen, S., Martire, L. M., Scheier, M., Baum, A., et al. (2009). Association of Enjoyable Leisure Activities with Psychological and Physical Well-Being. Psychosomatic Med. 71 (7), 725–732. doi:10.1097/PSY.0b013e3181ad7978
Qureshi, S. U., Kimbrell, T., Pyne, J. M., Magruder, K. M., Hudson, T. J., Petersen, N. J., et al. (2010). Greater Prevalence and Incidence of Dementia in Older Veterans with Posttraumatic Stress Disorder. J. Am. Geriatr. Soc. 58, 1627–1633. doi:10.1111/j.1532-5415.2010.02977.x
Rusted, J., Sheppard, L., and Waller, D. (2006). A Multi-centre Randomized Control Group Trial on the Use of Art Therapy for Older People with Dementia. Group Anal. 39 (4), 517–536. doi:10.1177/0533316406071447
Santos Silva, R., Mol, A. M., and Ishitani, L. (2019). Virtual Reality for Older Users: a Systematic Literature Review. International Journal of Virtual Reality. 19, 11–25. doi:10.20870/ijvr.2019.19.1.2908
Thabane, L., Ma, J., Chu, R., Cheng, J., Ismaila, A., Rios, L. P., et al. (2010). A Tutorial on Pilot Studies: the What, Why and How. BMC Med. Res. Methodol. 10 (1). doi:10.1186/1471-2288-10-1
University of South Australia (2007). Antipsychotics in Dementia. Veterans' MATES. Available at: https://www.veteransmates.net.au/topic-12-therapeutic-brief (Accessed March 31, 2021).
Victoria Hospice Society (2006). Palliative Performance Scale (PPS). New York: Visiting Nurse Service of New York. Available at: https://www.vnsny.org/wp-content/uploads/2016/08/VNSNY-Palliative-Performance-Scale-PPS.pdf?pdf=/wp-content/uploads/2016/08/VNSNY-Palliative-Performance-Scale-PPS.pdf.
Wang, Q.-Y., and Li, D.-M. (2016). Advances in Art Therapy for Patients with Dementia. Chin. Nurs. Res. 3 (3), 105–108. doi:10.1016/j.cnre.2016.06.011
Warden, V., Hurley, A. C., and Volicer, L. (2003). Development and Psychometric Evaluation of the Pain Assessment in Advanced Dementia (PAINAD) Scale. J. Am. Med. Directors Assoc. 4 (1), 9–15. doi:10.1097/01.jam.0000043422.31640.f7
White, M. P., Alcock, I., Grellier, J., Wheeler, B. W., Hartig, T., Warber, S. L., et al. (2019). Spending at Least 120 minutes a Week in Nature Is Associated with Good Health and Wellbeing. Sci. Rep. 9, 7730. doi:10.1038/s41598-019-44097-3
Wijma, E. M., Veerbeek, M. A., Prins, M., Pot, A. M., and Willemse, B. M. (2017). A Virtual Reality Intervention to Improve the Understanding and Empathy for People with Dementia in Informal Caregivers: Results of a Pilot Study. Aging Ment. Health. 22, 1121–1129. doi:10.1080/13607863.2017.1348470
Yaffe, K., Vittinghoff, E., Lindquist, K., Barnes, D., Covinsky, K. E., Neylan, T., et al. (2010). Posttraumatic Stress Disorder and Risk of Dementia Among US Veterans. Arch. Gen. Psychiatry 67 (6), 608–613. doi:10.1001/archgenpsychiatry.2010.61
Keywords: virtual reality, dementia, veterans, behavioral and psychological symptoms in dementia, responsive behaviors, recreational therapist, non-pharmacological, behavioral and psychological symptoms
Citation: Appel L, Appel E, Kisonas E, Lewis S and Sheng LQ (2022) Virtual Reality for Veteran Relaxation: Can VR Therapy Help Veterans Living With Dementia Who Exhibit Responsive Behaviors?. Front. Virtual Real. 2:724020. doi: 10.3389/frvir.2021.724020
Received: 11 June 2021; Accepted: 15 December 2021;
Published: 11 February 2022.
Edited by:
Barry Peterson, VA Sierra Nevada Health Care System, United StatesReviewed by:
P. J. Standen, University of Nottingham, United KingdomEvelien Heyselaar, Radboud University, Netherlands
Copyright © 2022 Appel, Appel, Kisonas, Lewis and Sheng. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Lora Appel, bG9yYS5hcHBlbEB1aG4uY2E=
 Lora Appel
Lora Appel Eva Appel
Eva Appel Erika Kisonas2
Erika Kisonas2 Lacey Qing Sheng
Lacey Qing Sheng