- 1The James Hutton Institute, Dundee, United Kingdom
- 2NatureScot, Aberdeen, United Kingdom
- 3The James Hutton Institute, Aberdeen, United Kingdom
Muirburn is a land management practice of upland Calluna vulgaris-dominated moorland landscapes that uses prescribed burning to maintain vegetation diversity, enhance the nutritional value of vegetation for livestock, and support game management. While the impacts of Muirburn are well characterized for aboveground diversity, there is a lack of equivalent data on the impacts of Muirburn on soil biodiversity. To address this, the impact of Muirburn on soil nematode communities was assessed over a 12-month period. Muirburn had no effect on soil nematode abundance, community structure, species richness, or functional group composition. However, irrespective of fire, there were temporal effects on nematode community structure likely driven by five genera representing several nematode functional groups: Bitylenchus, Filenchus, Plectus, Mesorhabditis, and Thonus. Nematode species richness also exhibited a temporal effect, although arguably this could have been driven by the final sampling time point. The relative composition of nematode functional groups also varied temporally. Results from this study contribute to addressing the significant knowledge deficit regarding the impacts of Muirburn practices on soil biodiversity and help to inform future best practice guidance for the prescribed burning of upland moorland.
1 Introduction
The iconic upland landscape of Scotland delivers multiple ecosystem benefits and is of international conservation importance (1). In part, this landscape is managed through the highly prescribed process of Muirburn (2), i.e., the rotational controlled burning of vegetation in moorland areas. These moorland areas are dominated by Calluna vulgaris and subject to rotational Muirburn. The management aims of this are to support a) red deer and sheep grazing by promoting new plant growth, b) structural plant diversity, and c) conservation of open moorland areas, providing forage and cover for economically important game birds such as red grouse (Lagopus lagopus scotica) (3–5). Notwithstanding the adoption of best practice, the outcomes of moorland burning are highly variable and driven by several factors, including pre-fire vegetation composition and age, fire severity, post-fire management, and natural regeneration pathways (6–8).
In stark contrast to the depth of available data on the effects of prescribed moorland burning on aboveground diversity (5, 9), there is a paucity of similar data regarding the effects on soil biota (10). While the focus on aboveground diversity is likely to be driven by end-user needs, the knowledge deficit regarding the impact on soil biota is crucial given that most soil processes are driven by soil biota (11), and the interactions between different soil biota confer a range of functional outcomes (12, 13), which in turn benefit aboveground plant communities (14). This reflects the wider paucity of data on the effects of fire on soil biota (15).
Nematodes are one of the most abundant soil taxa globally (16) and have key roles in, for example, soil nutrient cycling, which can enhance plant growth (17, 18), but their function is dependent upon complex interactions with other soil biota (19) and the availability of nutrients, which may be modified post-fire (20). Their functional role can be inferred by their trophic position. Nematodes feature in all major trophic levels of the soil food web (21) and have pivotal roles in processing organic nutrients (17) and the control of soil microorganism populations (22–24), with critical roles in regulating carbon and nutrient dynamics within and across landscapes (19), and are a good indicator of biological activity in soils (25).
The few studies on the effect of wildfires on soil nematodes have reported inconsistent results. For example, along a 3,000-km north–south transect, at an ecoregion scale, ground fires in different forest types had no impact on overall nematode abundance; however, this masked an increased abundance of bacterivorous nematodes, offset by a reduction in the abundance of fungivorous and herbivorous nematodes (26). In contrast, a short-term (≤1 year) reduction in overall nematode abundance was reported in fire-affected forest soils compared to controls linked to fire severity (27). Although the abundance of bacterivorous nematodes increased, the abundance of herbivorous and omnivorous nematodes decreased. Recently, it was reported that wildfire greatly reduced total nematode abundance in a Mediterranean pine forest and resulted in a shift from a fungivore- to bacterivore/herbivore-dominant system compared to unburnt areas (28). The effects of wildfire have been reported to negatively impact total nematode abundance for at least 15 years compared to unburnt areas in an area of natural vegetation dominated by hawthorn and Berberis (29). Nevertheless, total nematode abundance was >90% greater in annual burnt vs. unburnt plots after 32 years in tallgrass prairie (30). However, immediately after a semi-arid grassland wildfire, bacterivorous, fungivorous, and herbivorous nematode abundances were reduced compared to those in unburnt areas, with recovery rates differing by trophic group (31). This contrasts with an increase in total nematode and herbivore abundance because of prescribed burning of snow-tussock grassland (32) and an increase in total nematode abundance and richness at the genus level after fire in an old-field grassland compared to unburnt controls (33).
A meta-analysis assessing belowground community responses to fire (34) concluded that fire reduces overall nematode abundance by 88%. However, these authors caveated this result due to the small sample size of relevant studies. Fire in a Mediterranean forest resulted in a lower nematode trophic diversity, alpha diversity, and richness (at genus level) in burnt forest soils compared to unburnt forest soils, with effects on abundance being limited to a few nematode genera (35). Prescribed fires either had no impact on nematode abundance in a pine forest (36) or had short-term effects on abundance dependent on vegetation cover (juniper vs. yucca) and soil moisture in an arid savanna (37). The effects of fire on nematode communities may therefore be highly system dependent, making it important that we understand the effects of burning in upland moorland systems subject to Muirburn.
To understand the impact, if any, of Muirburn on soil nematode communities, paired transects representing burnt and unburnt plots were temporally sampled over a 1-year period. Given that upland moorlands are characterized by high water availability and that Muirburn is a prescribed short-term event, it was hypothesized that Muirburn would have no significant impact on the overall abundance of soil nematode communities but may have short-term impacts on certain specific functional (trophic) groups.
2 Materials and methods
Two adjacent study sites were sampled, and these were located on Birnie (56.90982 N, 2.54861 W at 350–400 m a.s.l) and Thorter (56.91888 N, 2.57264 W at 390–430 m a.s.l) Hills, which form part of the Glensaugh Research Farm, Aberdeenshire, UK, managed by The James Hutton Institute. The study sites had a predominantly south-facing aspect, with the dominant soil being podzol and the dominant vegetation being moorland heather, C. vulgaris. Parallel transects, burnt and unburnt, were established on both hills in early spring using a replicated “Before-After-Control-Impact” (BACI) design with soil samples taken at points along the transects. The experiment was conducted in a spatially discrete area chosen for the same typology, aspect, and uniformity in ground cover to remove any potential issue with vegetation combustion rate. Moreover, each patch was subjected to the same duration of burning to mitigate differential fire intensity.
Soil cores were collected using a grass plot sampler (internal diam. 2.3 cm; Eijkelkamp, Giesbeek, The Netherlands) to a depth of 10 cm. A 1-kg composite soil sample was collected from each sampled area, with each composite sample consisting of approximately 20 cores taken randomly along a standard “W” pattern (38). For both burnt and unburnt areas, samples were collected before burning (t = 0), immediately after burning (t = 0.1), and 3, 10, 20, 35, 60, 120, 240, and 360 days thereafter, with 20 replicates of both burnt and unburnt samples, representing a total of 400 samples. Soil samples were stored at 4°C prior to processing. Nematodes were extracted from a 200-g subsample of each soil sample (39) using a modified Baermann funnel method (40). After ca. 48 h, extracted nematodes were collected in 20 mL of water and left to settle for ca. 2 h. Thereafter, water was decanted to leave a ca. 2-mL volume.
Environmental [air and soil (10-cm depth) temperature and precipitation] data (Supplementary Figure S1) were sourced from the Birnie Burn weather station (ca. 250 m a.s.l.) that is part of the UK Environmental Change Network located approximately 2 km from the experimental sites.
Nematode community composition was assessed using directed Terminal restriction fragment length polymorphism (T-RFLP) (24), with methods used as previously reported for nematode DNA extraction, sample processing, and PCR conditions (41).
Differences in nematode community structure were visualized using principal coordinates analysis (PCoA) and alternative Gower distances. Differences in beta diversity between burnt and unburnt plots were tested using PERMANOVA with alternative Gower distances used for T-RFLP data, plot used as a constraining variable (strata), and a default of 999 permutations. Pairwise dissimilarities were calculated between paired burnt and unburnt plots for each time point. To account for the repeated measures design, the resulting values were compared using a linear mixed-effects model with plot pair as a random factor.
Species richness was taken to be the number of different terminal restriction fragments (T-RFs) present. A linear mixed-effects model with plot as a random factor and time since burning as an independent variable was used to assess the effect of burning and time on species richness. Models were run separately for burnt and unburnt plots, and an additional model that only used time points from 0 to 60 days since burning was used to assess the short-term impacts of fire on species richness.
Nematode taxonomy and nematode feeding groups (42) were assigned to T-RFs (24). Where a T-RF could not be robustly taxonomically identified, it was classified as “unknown”. Differences in the relative fluorescence of nematode feeding groups were tested using a linear mixed-effects model as described above. In addition, the effects of soil temperature, dry temperature, and rainfall were tested for each of the feeding groups. Differences in the composition of feeding groups were tested using a PERMANOVA, again with plot as a blocking variable. All statistics were carried out in the R statistical environment (43), distance matrix calculations and PERMANOVA were carried out in the package vegan (44), PCoA was calculated in the package ape (45), and linear mixed-effects models were carried out using the package nlme v. 3.1-160 (46).
3 Results
Prescribed fire (Muirburn) did not result in a significant change in total fluorescence, a proxy for nematode abundance between burnt and unburnt treatments (p = 0.426; Figure 1). However, there were temporal effects within both burnt and unburnt treatments. Over the duration of the study, and including all sampling time points, herbivorous nematodes decreased in relative abundance under both burnt (p < 0.001, AIC = −233.67) and unburnt (p = 0.04, AIC = −170.281) treatments (Figure 1a). Similarly, omnivore relative abundance decreased but only under the influence of burning (Figure 1c, p < 0.001, AIC = −217.97). In contrast, bacterivorous nematodes increased in relative abundance (Figure 1b, p = 0.03, AIC = −120.89) in burnt but not unburnt plots (p = 0.469). Predatory nematodes (Figure 1d) did not exhibit any temporal effects under either the burnt (p = 0.283) or unburnt treatment (p = 0.543). Soil moisture showed a significant difference between burnt and unburnt plots (p < 0.01) and over time (p < 0.01).
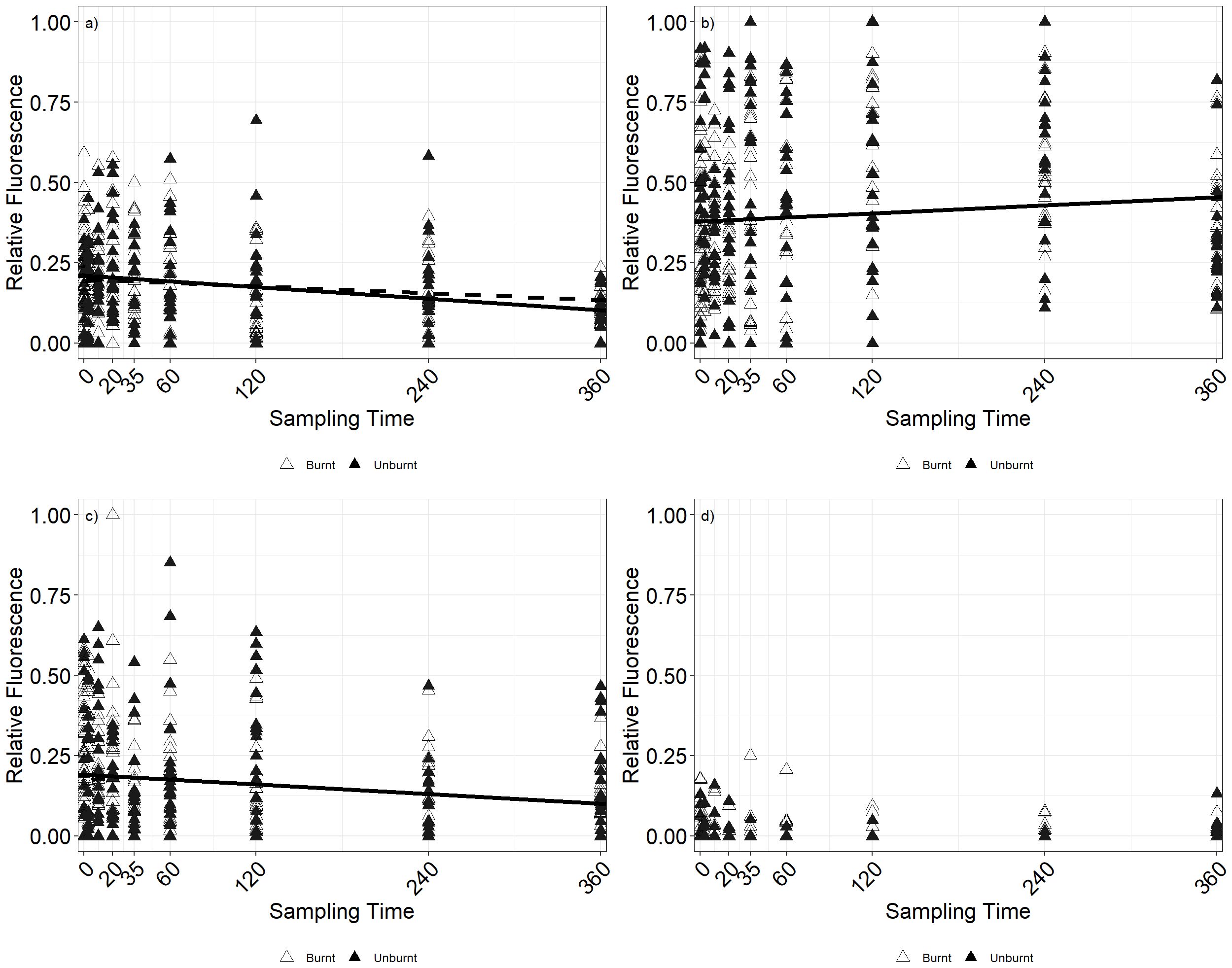
Figure 1. Change in relative fluorescence over time for (a) herbivores, (b) bacterivores, (c) omnivores, and (d) predators. Lines of best fit as tested using generalized linear model (GLM) with plot as a random factor are shown for burnt plots (solid line) and unburnt plots (dotted line). Lines are omitted where there was no significant relationship between time and relative fluorescence.
To assess whether the final sampling time point (T360) was the primary driver of the temporal variability in the nematode community, the analysis was repeated with the removal of data from T360. As with the inclusion of the T360 data, there were no statistically supported differences (bacterivore, p = 0.093; herbivore, p = 0.578; omnivore, p = 0.589; predator, p = 0.060) between burnt and unburnt treatments, with temporal differences only occurring within the respective burnt and unburnt treatments. However, two differences were noted compared to when T360 data were included: herbivorous nematodes no longer exhibited temporal variability in relative abundance within unburnt plots (p = 0.844), and bacterivorous nematodes now showed a small but significant increase temporally under unburnt plots (p = 0.02, AIC = −109.40).
While nematode species richness (Figure 2b) temporally increased within both burnt (p < 0.01, AIC = 1,080.37) and unburnt treatments (p < 0.01, AIC = 994.67), like nematode relative abundance, there was no impact of burning (p = 0.138) on species richness. Constraining the analysis to only the early sampling time points (T0–T60, Figure 2a) to determine whether the later time points were driving species richness resulted in no temporal variability under the burnt treatment (p = 0.629) and slight temporal variation under the unburnt treatment (p = 0.04, AIC = 610.911).
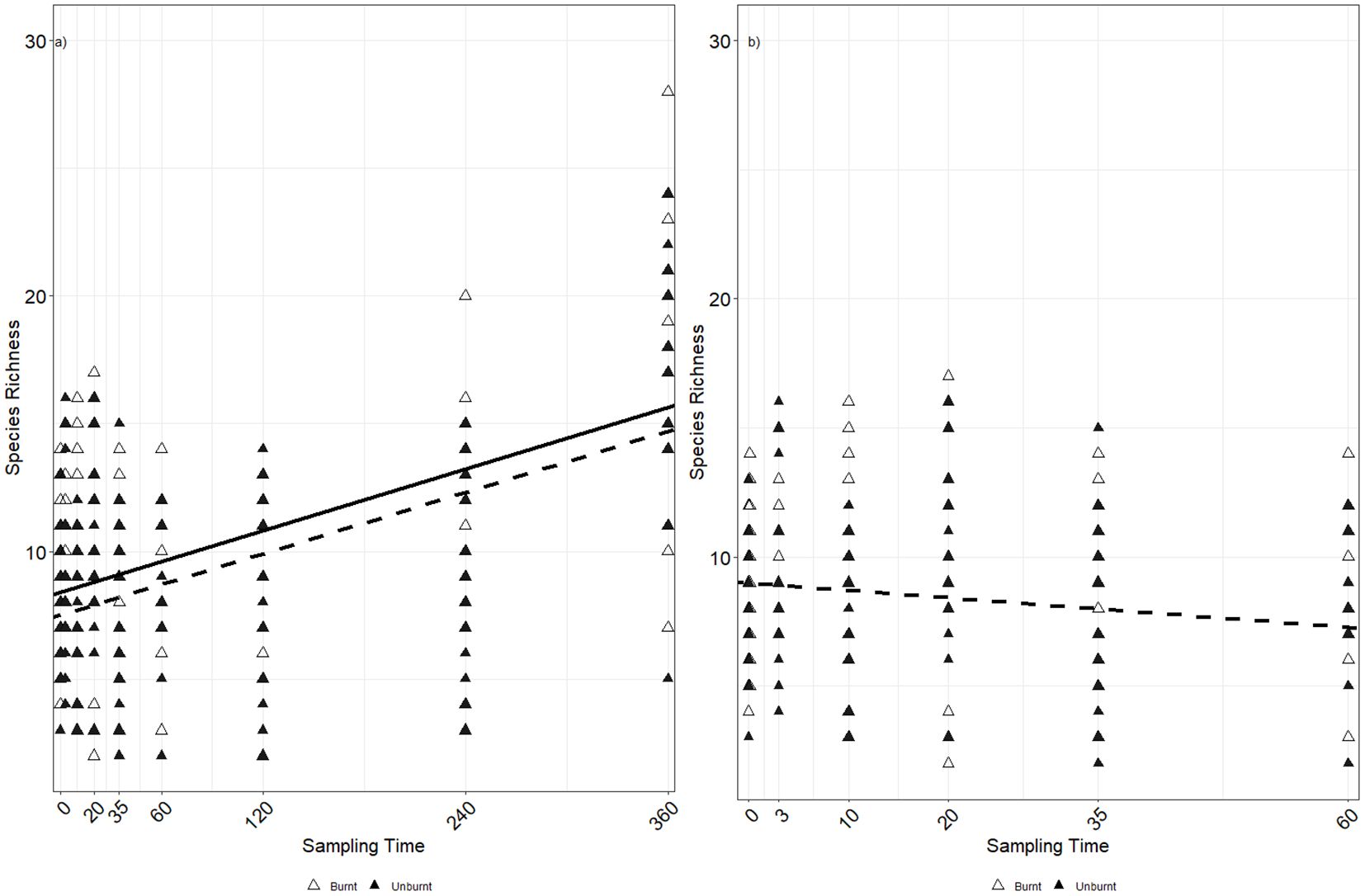
Figure 2. Change in nematode species richness over time in burnt and unburnt plots for (a) T0–T60 and (b) all time points. Lines of best fit as tested using GLM with plot as a random factor are shown for burnt plots (solid line) and unburnt plots (dotted line). Lines are omitted where there was no significant relationship between time and species richness.
Irrespective of fire, there was a significant change in nematode community structure over time [Fdf (9,368) = 4.417, p < 0.001]. Five nematode genera representing three different functional groups (38)—Bitylenchus (herbivore), Filenchus (herbivore), Plectus (bacterivore), Mesorhabditis (bacterivore), and Thonus (omnivore/predator)—had the highest loadings along PCoA axis 1, suggesting that these genera were driving the differences in nematode community structure between time points (Figure 3a). Pairwise dissimilarities between paired burnt and unburnt plots did not differ with time (p = 0.417) (Figure 4). Analysing the data at the resolution of nematode trophic groups highlighted the strength but opposing influences of bacterivores, herbivores, and omnivores (Figure 3b).
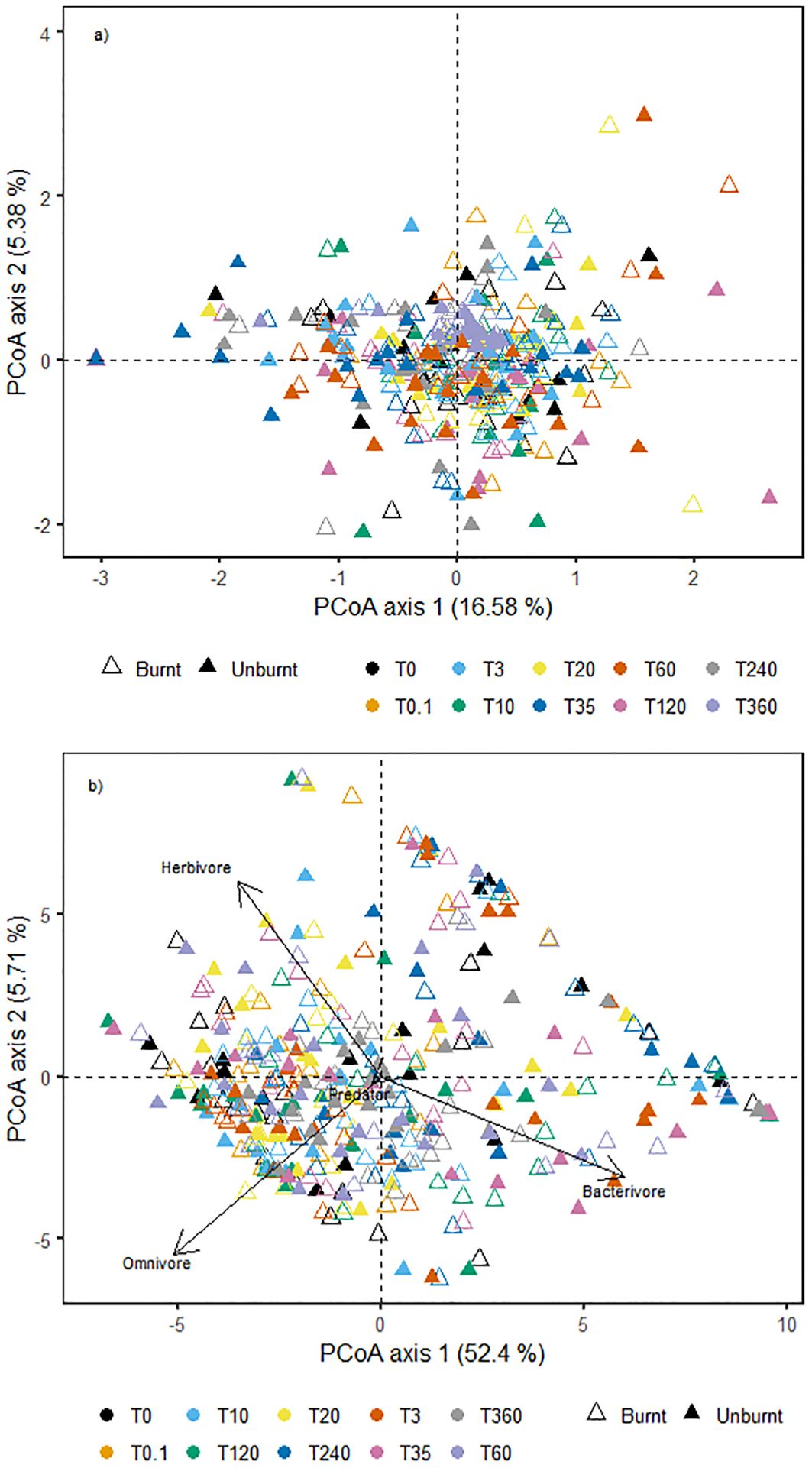
Figure 3. PCoA plot of nematode community composition for all samples at all time points by (a) individual T-RF and (b) nematode trophic groups, with lines representing strength of influence. PCoA, principal coordinates analysis.
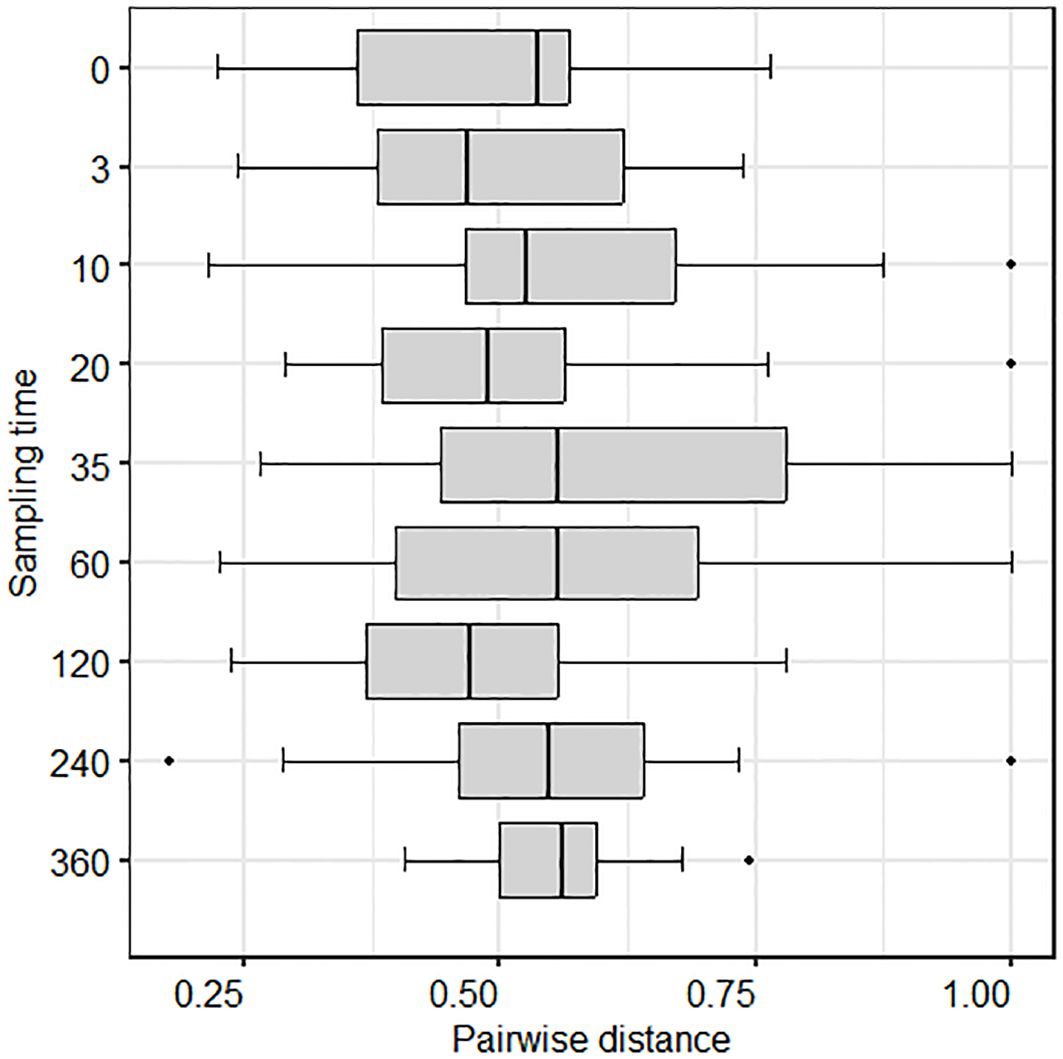
Figure 4. Change in pairwise distances (alternative Gower) over time between burnt and unburnt plots.
The relative composition of nematode functional groups varied temporally [Fdf (1,9) = 3.08, p < 0.001] but not as a result of prescribed fire (Table 1). Although several nematode functional groups varied over time [herbivore, Fdf (1,9) = 3.11, p < 0.001; omnivore, Fdf (1,9) = 2.40, p < 0.05; bacterivore, Fdf (1,9) = 3.55, p < 0.001], there was no consistent temporal pattern. Moreover, with two exceptions, the environmental variables, air and soil temperature (10-cm depth) and precipitation, had no impact on nematode trophic groups (Table 1). However, soil temperature at 10-cm depth significantly increased bacterivore relative abundance (p < 0.001), and precipitation slightly increased herbivore relative abundance (p = 0.015). The potential of predatory nematodes being a biological driver within the system was checked with a significant inverse relationship observed between predatory and bacterivore nematodes (p < 0.001), but not between predatory and either herbivore (p = 0.487) or omnivore nematodes (p = 0.089).
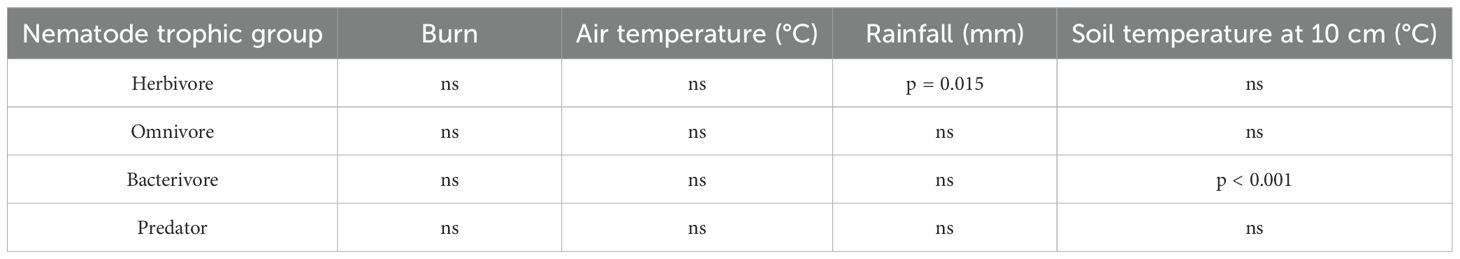
Table 1. Effect of burning and environmental drivers on the relative abundance of nematode trophic groups.
4 Discussion
Nematodes have been rarely studied in UK moorland soils, and to date, studies have been either observational (47) or associated with the impact of land use change (48). To our knowledge, this is the first study to consider the impact of prescribed burning (Muirburn) on nematodes of moorland soil.
Prescribed fire adhering to Muirburn principles (2) had no effect on soil nematode abundance, community structure, species richness, or functional group composition in an upland moorland landscape. Thus, our hypothesis that Muirburn would have a limited impact on some or all nematode functional (trophic) groups is rejected.
This contrasts with the effects of prescribed fire in a 3-year study system that led to an increase in the proportion of bacterial feeding nematodes in unburnt compared to burnt patches in a juniper-dominated savanna (37), although this was influenced by a strong temporal component. Furthermore, total nematode abundance was the greatest in unburnt compared to burnt patches, especially during cool seasons (37). In a Pinus forest study (49), a short-term (6 weeks) effect of prescribed burning was noted, with the abundance of omnivores and predators increasing and herbivore abundance unchanged, whereas in grassland systems, prescribed fire increased herbivore abundance (32, 33) compared to unburnt controls.
In the current study, soil nematode community composition and richness were temporally driven, although in the case of bacterivores in unburnt plots, this was driven by an increase in their relative abundance at the final sampling time point, 360 days after the imposition of Muirburn had the greatest influence. Temporal variability of soil nematode communities due to a range of (a)biotic factors is well recognized (50–52), and in the context of prescribed fire, such factors have included changes in soil organic matter (36); loss of tree canopies, litter accumulation, and shrub foliage, affecting soil temperature and moisture (37); modified interactions with other soil biota (15, 53); and increased soil N (33).
While this study was able to detect changes in the relative abundance of the different feeding groups, it was unable to determine the drivers, with only bacterivores and herbivores showing a relationship with any of the measured climatic variables, and this relationship only had a small effect on the relative abundance of these feeding groups. Precipitation, previously reported to drive changes in nematode abundance and community structure (54–56), had a limited effect in this study, with only the relative abundance of herbivores marginally increasing. This is likely due to the radically different systems of forest, prairie grasslands, and semi-arid steppe compared to year-round saturated moorland soil, as in this study, and thus, moisture was likely not a stress to the system. Moreover, precipitation has a reported range of effects on nematodes. As in a recent study of nematodes under soybean cultivation in Brazil, precipitation had no effect on nematodes (57). In the same study (57), air temperature was significantly correlated to nematode community structure in line with previous studies under a range of land use types (58, 59). In contrast, air temperature had no effect on nematode trophic groups in this study, perhaps due to the exposed nature of the study sites. However, in the current study, soil temperature at 10-cm depth resulted in an increase in bacterivorous nematodes, which concurs with previous studies under grassland (60) and controlled conditions (59). Moreover, soil temperature was reported to have a similar differential effect on nematode trophic groups, which varied under land use type (61).
The five genera driving the temporal changes in community structure in this study represented several functional groups, thus suggesting that potentially multiple factors had a role in shaping nematode communities, although these taxa did not lead to burnt and unburnt plots separating on the PCoA, suggesting that they were important in both burnt and unburnt plots. The dominant nematode functional groups in this study were bacterivores and herbivores, which is consistent with a meta-analysis of data collected from a broad spectrum of global biomes (16).
While the time of sampling was the main cause of any differences, there was a potential relationship between time and burning with such temporal trends. When data from T360 were included, there were significant differences in the relative abundance of omnivores and bacterivores in burnt but not unburnt plots. However, re-analysing the data without the T360 data yielded additional significant differences for bacterivores, but not omnivores, from unburnt plots. Therefore, it is clear that the drivers of change under Muirburn are complex. A review of the impacts of fire on soil communities (15) concluded that indirect rather than direct effects of fire had a greater impact on nematodes and especially so in systems where soil moisture is limited. Thus, the lack of any impact of Muirburn practices on soil nematode communities in this study may be due to the high soil moisture content, which is characteristic of the upland systems where Muirburn is deployed, and the timing of prescribed burning, which the Muirburn Code states should occur in winter/early spring to mitigate habitat damage, and reduces the potential of collateral ignition of nearby moss/peat (10, 62). Moreover, a sampling depth of 10 cm, such as used in this study, was considered to have a potential dilution effect, i.e., an insufficient depth profile to generate discernible fire effects (63).
Alternatively, notwithstanding that nematodes are considered of utility as an integrated measure of the functional state of soils (25), they may be a poor choice of soil taxa to study regarding the impact of fire, given their resilience to abiotic extremes, including heat (64). Moreover, given small-scale heterogeneity effects on soil biota (65), future studies characterizing microhabitat properties may be a promising approach (66). This study, for the first time, systematically evaluates the impact of Muirburn on soil nematode communities and finds that the temporal dynamics of soil nematode communities outweigh the effects of fire, providing important references for future fire management practices.
Data availability statement
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found below: https://www.ebi.ac.uk/ena, ERX4144080-ERX4144317.
Ethics statement
The manuscript presents research on animals that do not require ethical approval for their study.
Author contributions
RN: Writing – review & editing, Validation, Funding acquisition, Project administration, Supervision, Writing – original draft, Conceptualization, Resources. DK: Methodology, Investigation, Writing – review & editing. RH: Methodology, Writing – review & editing, Investigation. DF: Methodology, Investigation, Writing – review & editing. MG: Data curation, Visualization, Validation, Writing – review & editing, Formal analysis.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. The study was funded by the Scottish Government Rural and Environment Science and Analytical Services (RESAS) through Underpinning National Capacity and Healthy Soils for a Green Recovery, as part of the 2016–2022 and 2022–2027 Strategic Research Programmes.
Acknowledgments
We thank colleagues at The James Hutton Institute sequencing service for the provision of the T-RFLP data.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fsoil.2025.1619114/full#supplementary-material
References
1. Thompson DBA, MacDonald AJ, Marsden JH, and Galbraith CA. Upland heather moorland in Great Britain: A review of international importance, vegetation change and some objectives for nature conservation. Biol Conserv. (1995) 71:163–78. doi: 10.1016/0006-3207(94)00043-P
2. The Muirburn Code. Inverness: Scottish Natural Heritage. In: Management of moorland by burning and cutting. Perth: NatureScot. (2017). p. 24.
4. Robertson PA, Park KJ, and Barton AF. Loss of heather Calluna vulgaris moorland in the Scottish uplands: the role of red grouse Lagopus lagopus scoticus management. Wildlife Biol. (2001) 7:11–6. doi: 10.2981/wlb.2001.004
5. Grant MC, Mallord J, Stephen L, and Thompson PS. The costs and benefits of grouse moor management to biodiversity and aspects of the wider environment: a review. RSPB Research Report 43. Sandy: Royal Society for the Protection of Birds (2012).
6. Tucker G. Review of the impacts of heather and grassland burning in the uplands on soils, hydrology and biodiversity. English Nature Research Report No. 550. Peterborough: English Nature (2003).
7. Davies GM, Legg CJ, Smith AA, and MacDonald AJ. Fire rate of spread in Calluna vulgaris-dominated moorlands. J Appl Ecol. (2009) 46:1054–63. doi: 10.1111/j.1365-2664.2009.01681.x
8. Davies GM, Smith AA, MacDonald AJ, Bakker JD, and Legg CJ. Fire intensity, fire severity and ecosystem response in heathlands: factors affecting the regeneration of Calluna vulgaris. J Appl Ecol. (2010) 47:356–65. doi: 10.1111/j.1365-2664.2010.01774.x
9. Milligan G, Rose RJ, O’Reilly J, and Marrs RH. Effects of rotational prescribed burning and sheep grazing on moorland plant communities: results from a 60-year intervention experiment. Land Degrad Dev. (2018) 29:1397–412. doi: 10.1002/ldr.2953
10. Brooker R, Hester A, Newey S, and Pakeman R. Socio-economic and biodiversity impacts of driven grouse moors in Scotland: Part 2 Biodiversity impacts of driven grouse moors in Scotland. Edinburgh: Scottish Government. (2018), 65.
11. Bardgett RD and van der Putten WH. Belowground biodiversity and ecosystem functioning. Nature. (2014) 515:505–11. doi: 10.1038/nature13855
12. Creamer RE, Hannula SE, van Leeuwen JP, Stone D, Rutgers M, Schmelz RM, et al. Ecological network analysis reveals the inter-connection between soil biodiversity and ecosystem function as effected by land use across Europe. Appl Soil Ecol. (2016) 97:112–24. doi: 10.1016/j.apsoil.2015.08.006
13. Manning P, van der Plas F, Soliveres S, Allan E, Maestre FT, Mace G, et al. Redefining ecosystem multifunctionality. Nat Ecol Evol. (2018) 2:427–36. doi: 10.1038/s41559-017-0461-7
14. Cortois R, Schröder-Georgi T, Weigelt A, van der Putten WH, and De Deyn GB. Plant–soil feedbacks: role of plant functional group and plant traits. J Ecol. (2016) 104:1608–17. doi: 10.1111/1365-2745.12643
15. Certini G, Moya D, Lucas-Borja ME, and Mastrolonardo G. The impact of fire on soil-dwelling biota: A review. For Ecol Manag. (2021) 488:118989. doi: 10.1016/j.foreco.2021.118989
16. van den Hoogen J, Geisen S, Routh D, Ferris H, Traunspurger W, Wardle DA, et al. Soil nematode abundance and functional group composition at a global scale. Nature. (2019) 572:194–8. doi: 10.1038/s41586-019-1418-6
17. Paterson E, Neilson R, Midwood AJ, Osborne SM, Sim A, Thornton B, et al. Altered food web structure and C-flux pathways associated with mineralisation of organic amendments to agricultural soil. Appl Soil Ecol. (2011) 48:107–16. doi: 10.1016/j.apsoil.2011.04.006
18. Gebremikael MT, Steel H, Buchan D, Bert W, and de Neve S. Nematodes enhance plant growth and nutrient uptake under C and N-rich conditions. Sci Rep. (2016) 6:32862. doi: 10.1038/srep32862
19. Mezeli MM, Page S, George TS, Neilson R, Mead A, Blackwell MSA, et al. Using a meta-analysis approach to understand complexity in soil biodiversity and phosphorus acquisition in plants. Soil Biol Biochem. (2020) 142:e107695. doi: 10.1016/j.soilbio.2019.107695
20. Pellegrini AFA, Hobbie SE, Reich PB, Jumpponen A, Brookshire ENJ, Caprio AC, et al. Repeated fire shifts carbon and nitrogen cycling by changing plant inputs and soil decomposition across ecosystems. Ecol Monogr. (2020) 90:e01409.
21. Wilschut RA and Geisen S. Nematodes as drivers of plant performance in natural systems. Trends Plant Sci. (2021) 26:237–47. doi: 10.1016/j.tplants.2020.10.006
22. Ingham RE, Trofymow JA, Ingham ER, and Coleman DC. Interactions of bacteria, fungi, and their nematode grazers: effects on nutrient cycling and plant growth. Ecol Monogr. (1985) 55:119–40. doi: 10.2307/1942528
23. Ferris H. Contribution of nematodes to the structure and function of the soil food web. J Nematol. (2010) 42:63–7.
24. Neilson R, Caul S, Fraser F, King D, Mitchell S, Roberts DM, et al. Microbial community size is a potential predictor of nematode functional group in limed grasslands. Appl Soil Ecol. (2020) 156:e103702. doi: 10.1016/j.apsoil.2020.103702
25. Ritz K and Trudgill DL. Utility of nematode community analysis as an integrated measure of the functional state of soils: perspectives and challenges. Plant Soil. (1999) 212:1–11. doi: 10.1023/A:1004673027625
26. Butenko KO, Gongalsky KB, Korobushkin DI, Ekschmitt K, and Zaitsev AS. Forest fires alter the trophic structure of soil nematode communities. Soil Biol Biochem. (2017) 109:107–17. doi: 10.1016/j.soilbio.2017.02.006
27. Renčo M, Adámek M, Jílková V, and Devetter M. Post-fire recovery of soil nematode communities depends on fire severity. Diversity. (2022) 14:1116. doi: 10.3390/d14121116
28. Papapostolou A, Kekelis P, Zafeiriou I, Gasparatos D, and Monokrousos N. Post-fire recovery of nematode communities along a slope gradient in a pine forest. Environ Monit Assess. (2024) 196:864. doi: 10.1007/s10661-024-13037-8
29. Kooch Y, Nouraei A, Wu D, Francaviglia R, and Frouz J. The effect of fire disturbance on the dynamics of soil physical, chemical, and biological properties over time in a semi-arid region. Appl Soil Ecol. (2024) 202:105568. doi: 10.1016/j.apsoil.2024.105568
30. Todd TC, Blair JM, and Callaham MA. Nematode communities respond more to N enrichment than to plant community changes over decades in tallgrass prairie. Appl Soil Ecol. (2025) 210:106096. doi: 10.1016/j.apsoil.2025.106096
31. Bastow J. The impacts of a wildfire in a semiarid grassland on soil nematode abundances over 4 years. Biol Fert Soils. (2020) 56:675–85. doi: 10.1007/s00374-020-01441-4
32. Yeates GW and Lee WG. Burning in a New Zealand snow-tussock grassland: effects on vegetation and soil fauna. New Zeal J Ecol. (1997) 21:73–9.
33. Song M, McDaniel MD, Zhu C, Lin F, and Zhang Y. Prescribed fire alters nematode communities in an old-field grassland. Ecol Evol. (2023) 13:e9977. doi: 10.1002/ece3.9977
34. Pressler Y, Moore JC, and Cotrufo MF. Belowground community responses to fire: meta-analysis reveals contrasting responses of soil microorganisms and mesofauna. Oikos. (2019) 128:309–27. doi: 10.1111/oik.05738
35. Pen-Mouratov S, Ginzburg O, Whitford WG, and Steinberger Y. Forest fire modifies soil free-living nematode communities in the Biriya woodland of northern Israel. Zool Stud. (2012) 51:1018–26.
36. Gibson KS, Johnson NC, Laturno C, Parmenter RP, and Antoninka A. Abundance of mites, but not of collembolans or nematodes, is reduced by restoration of a Pinus ponderosa forest with thinning, mastication, and prescribed fire. Trees For People. (2022) 7:100190. doi: 10.1016/j.tfp.2022.100190
37. Whitford WG, Pen-Mouratov S, and Steinberger Y. The effects of prescribed fire on soil nematodes in an arid juniper savanna. Open J Ecol. (2014) 4:66–75. doi: 10.4236/oje.2014.42009
38. Marshall B, Boag B, McNicol JW, and Neilson R. A comparison of the spatial distribution of three plant-parasitic nematode species at three different scales. Nematologica. (1998) 44:303–20.
39. Wiesel L, Daniell TJ, King D, and Neilson R. Determination of the optimal soil sample size to accurately characterise nematode communities in soil. Soil Biol Biochem. (2015) 80:89–91. doi: 10.1016/j.soilbio.2014.09.026
40. Brown DJF and Boag B. An examination of methods used to extract virus-vector nematodes (Nematoda: Longidoridae and Trichodoridae) from soil samples. Nematol medit. (1988) 16:93–9.
41. Donn S, Neilson R, Griffiths BS, and Daniell TJ. A novel molecular approach for rapid assessment of soil nematode assemblages – variation, validation and potential applications. Methods Ecol Evol. (2012) 3:12–23. doi: 10.1111/j.2041-210X.2011.00145.x
42. Yeates GW, Bongers T, de Goede RGM, Freckman DW, and Georgieva SS. Feeding habits in soil nematode families and genera - an outline for soil ecologists. J Nematol. (1993) 25:315–31.
43. R Core Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing (2021). Available online at: https://www.R-project.org/.
44. Oksanen J, Simpson GL, Blanchet FG, Kindt R, Legendre P, Minchin PR, et al. Vegan: Community ecology package, version 2.6.4 (2022). Available online at: https://cran.r-project.org/web/packages/vegan/index.html.
45. Paradis E and Schliep K. ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics. (2019) 35:526–8. doi: 10.1093/bioinformatics/bty633
46. Pinheiro J, Bates D, and R Core Team. nlme: Linear and Nonlinear Mixed Effects Models. R package version 3.1-162 (2023). Available online at: https://CRAN.R-project.org/package=nlme.
47. Banage WB. The ecological importance of free-living soil nematodes with special reference to those of moorland soil. J Anim Ecol. (1963) 32:133–40. doi: 10.2307/2522
48. Keith AM, van der Wal R, Brooker RW, Osler GHR, Chapman SJ, and Burslem DFRP. Birch invasion of heather moorland increases nematode diversity and trophic complexity. Soil Biol Biochem. (2006) 38:3421–30. doi: 10.1016/j.soilbio.2006.05.013
49. McSorley R. Short-term effects of fire on the nematode community in a pine forest. Pedobiologia. (1993) 37:39–48. doi: 10.1016/S0031-4056(24)00084-2
50. Yeates GW. Effects of plants on nematode community structure. Annu Rev Phytopathol. (1999) 37:127–49. doi: 10.1146/annurev.phyto.37.1.127
51. Griffiths BS, Neilson R, and Bengough AG. Soil factors determined nematode community composition in a two-year pot experiment. Nematology. (2003) 5:889–97. doi: 10.1163/156854103773040808
52. Barrios E. Soil biota, ecosystem services and land productivity. Ecol Econ. (2007) 64:269–85. doi: 10.1016/j.ecolecon.2007.03.004
53. She R, Wang H-Q, Fornacca D, Li F-T, Zhang F, Yang Y-Q, et al. Independence effects of heat and ash on forest soil nematode-trapping fungi communities. Fire. (2023) 6:27. doi: 10.3390/fire6010027
54. Landesman WJ, Treonis AM, and Dighton J. Effects of a one-year rainfall manipulation on soil nematode abundances and community composition. Pedobiologia. (2011) 54:87–91. doi: 10.1016/j.pedobi.2010.10.002
55. Franco ALC, Guan P, Cui S, de Tomasel CM, Gherardi LA, Sala OE, et al. Precipitation effects on nematode diversity and carbon footprint across grasslands. Glob Change Biol. (2022) 28:2124–32. doi: 10.1111/gcb.16055
56. Li Y, Guo J, Wang X, Lian T, Yuan R, Feng J, et al. Precipitation addition during growing and non-growing seasons interactively affects the abundance of soil nematode communities in a semi-arid steppe. Catena. (2025) 249:108686. doi: 10.1016/j.catena.2024.108686
57. Martins do Rêgo Barros F, Neilson R, Giles M, Caul S, Pedrinho A, de Oliveira CMG, et al. Bacterial and nematode communities associated with soybean-cultivated soils from two native Brazilian biomes. Appl Soil Ecol. (2025) 212:106179. doi: 10.1016/j.apsoil.2025.106179
58. Pen-Mouratov S, He X, and Steinberger Y. Spatial distribution and trophic diversity of nematode populations under Acacia raddiana along a temperature gradient in the Negev Desert ecosystem. J Arid Environ. (2004) 56:339–55. doi: 10.1016/S0140-1963(03)00058-2
59. Zheng L, Wu S, Lu L, Li T, Liu Z, Li Z, et al. Unraveling the interaction effects of soil temperature and moisture on soil nematode community: A laboratory study. Euro J Soil Biol. (2023) 118:103537. doi: 10.1016/j.ejsobi.2023.103537
60. Papatheodorou EM, Argyropoulou MD, and Stamou GP. The effects of large- and small-scale differences in soil temperature and moisture on bacterial functional diversity and the community of bacterivorous nematodes. Appl Soil Ecol. (2004) 25:37–49. doi: 10.1016/S0929-1393(03)00100-8
61. Bakonyi G, Nagy P, Kovács-Láng E, Kovács E, Barabás S, Répási V, et al. Soil nematode community structure as affected by temperature and moisture in a temperate semiarid shrubland. Appl Soil Ecol. (2007) 37:31–40. doi: 10.1016/j.apsoil.2007.03.008
62. Fielding D, Newey S, Pakeman RJ, Miller D, Gagkas Z, Matthews K, et al. Limited spatial co-occurrence of wildfire and prescribed burning on moorlands in Scotland. Biol Conserv. (2024) 296:110700. doi: 10.1016/j.biocon.2024.110700
63. Pressler Y, Wilson G, Cotrufo MF, and Moore JC. Fire return interval influences soil food web structure and stability in an oak−pine savanna. Plant Soil. (2024). doi: 10.1007/s11104-024-06646-2
64. Thakur MP, Risch AC, and van der Putten WH. Biotic responses to climate extremes in terrestrial ecosystems. iScience. (2022) 25:104559. doi: 10.1016/j.isci.2022.104559
65. Nielsen UN, Osler GHR, Campbell CD, Neilson R, Burslem DFRP, and van der Wal R. The enigma of soil animal species diversity revisited: the role of small-scale heterogeneity. PloS One. (2010) 5:e11567. doi: 10.1371/journal.pone.0011567
Keywords: biodiversity, communities, disturbance, fire ecology, land management, soil biota, soil ecology, upland conservation
Citation: Neilson R, King D, Hooper R, Fielding DA and Giles ME (2025) Soil nematode community responses to prescribed burning in upland moorland: temporal dynamics prevail over fire effects. Front. Soil Sci. 5:1619114. doi: 10.3389/fsoil.2025.1619114
Received: 27 April 2025; Accepted: 05 August 2025;
Published: 29 August 2025.
Edited by:
Junqiang Zheng, Henan University, ChinaReviewed by:
Alasdair Noble, AgResearch Ltd, New ZealandShuaibing Wang, Yuxi Normal University, China
Chaosheng Luo, Yunnan Agricultural University, China
Copyright © 2025 Neilson, King, Hooper, Fielding and Giles. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Roy Neilson, cm95Lm5laWxzb25AaHV0dG9uLmFjLnVr
 Roy Neilson
Roy Neilson Dale King1
Dale King1 Madeline E. Giles
Madeline E. Giles