1
Department of Biochemistry and Molecular Genetics, Research Institute for Neurological Diseases and Geriatrics, Kyoto Prefectural University of Medicine, Japan
2
Department of Biophysics, Graduate School of Science, Kyoto University, Japan
3
Institute for Comprehensive Medical Science, Fujita Health University, Japan
4
Frontier Technology Center, Kyoto University Graduate School of Medicine, Japan
5
Japan Science and Technology Agency, BIRD & CREST, Japan
Anxiety is an instinct that may have developed to promote adaptive survival by evading unnecessary danger. However, excessive anxiety is disruptive and can be a basic disorder of other psychiatric diseases such as depression. The KF-1, a ubiquitin ligase located on the endoplasmic reticulum (ER), may prevent excessive anxiety; kf-1−/− mice exhibit selectively elevated anxiety-like behavior against light or heights. It is surmised that KF-1 degrades some target proteins, responsible for promoting anxiety, through the ER-associated degradation pathway, similar to Parkin in Parkinson’s disease (PD). Parkin, another ER-ubiquitin ligase, prevents the degeneration of dopaminergic neurons by degrading the target proteins responsible for PD. Molecular phylogenetic studies have revealed that the prototype of kf-1 appeared in the very early phase of animal evolution but was lost, unlike parkin, in the lineage leading up to Drosophila. Therefore, kf-1−/− mice may be a powerful tool for elucidating the molecular mechanisms involved in emotional regulation, and for screening novel anxiolytic/antidepressant compounds.
Anxiety, or learned fear, is not necessarily harmful to everyday life but, rather, is a natural ability that may have arose to evade unnecessary dangers. However, excessive anxiety is debilitating or disadvantageous for life as it reduces behavioral activities necessary for adaptation. Moreover, anxiety can be a core symptom of various mental/behavioral disorders, such as major depressive disorders, obsessive-compulsive disorders, panic disorder, adaptive disorder, post-traumatic stress disorder, social withdrawal disorder, and various phobias. Patients with anxiety/depression interpret circumstantial incidences, including episodes, comments, and expressions, in a negative way. The interpretation leads individuals to enhanced capture or delusions caused by potential signs of danger. This suggests that a system of negative and positive regulation of the emotional expression may have developed under the evolutionary constraint. Such a system would be most apparent in highly social animals with relatively little reproduction per generation, like humans (Darwin, 1872
). Indeed, there is evidence that the amygdala is responsiblefor the expression of anxiety or fear, and the prefrontal cortex plays a role in fear extinction by regulating the amygdala-mediated expression of fear (see Bishop, 2007
). Although the molecular mechanisms underlying negative and positive regulation of the anxiety are not fully understood, many genes have been reported to affect anxiety or fear (Chen et al., 1994
; Gogos et al., 1998
; Heisler et al., 1998
; Holmes et al., 2003
; Miyakawa et al., 1994
, 2003
; Nakajima et al., 2008
; Yamasaki et al., 2008
). Among these, the genes related to the serotonergic system are seen to be of special interest. For example, human genomic studies of anxiety/depression have focused on genes related to the monoaminergic neurotransmission of serotonin receptors (5-HT1A) and transporters (5-HTT) (see Levinson, 2006
; Uher and McGuffin, 2008
). Genetic studies using gene-targeting techniques have revealed that knockout mice lacking either 5-ht1A or 5-htt exhibit significantly increased anxiety-like behaviors (Heisler et al., 1998
; Holmes et al., 2003
). However, proteins that interact directly with neurotransmitters seem to function downstream, rather than upstream, of the serotonergic pathway.
Anxiety/depression is the most common psychiatric disease seen in patients irrespective of nations, societies, and religions. Consequently, pharmaceutical companies have made extensive worldwide efforts to develop anxiolytic/antidepressant drugs, particularly serotonergic compounds such as selective serotonin reuptake inhibitor (SSRI) and serotonin noradrenalin reuptake inhibitor (SNRI) based on the monoamine hypothesis. The hypothesis assumes that the pathogenesis of depression is caused by the depletion of monoamines such as serotonin in the brain. This assumption has however not been proven in the last 60 years. Testing systems have been developed for rodents to measure behavioral despair and to screen serotonergic drugs. These include the forced swim test and the tail suspension test. Compounds that elevate monoamine levels in the brain reduce the despair-like behavior or immobility time of animals under fearful conditions (Porsolt et al., 1978
; Steru et al., 1985
). However, approximately one-third of the patients with anxiety/depression do not respond to the serotonergic drugs, suggesting that despair-like behavior in rodents does not precisely represent anxiety/depression in humans, from a pharmacological point of view. Therefore, it is desirable to have some genetic animal models that display excessive anxiety-like behavior specifically without affecting despair-like behavior, as observed in the kf-1−/− mice (Tsujimura et al., 2008
), to look for novel anxiolytic/antidepressant drugs that are effective in patients who do not respond to serotonergic drugs.
The gene for KF-1 was originally discovered in connection with Alzheimer’s disease (AD). Genetic studies of familial AD (FAD) made significant progress in 1990s. In this period, three genes encoding β-amyloid precursor protein, pressenilin-1 (PS1), and presenilin-2 (PS2) were identified as the causative genes for FAD (see Hashimoto-Gotoh et al., 2006
; Lundkvist and Näslund, 2007
), and two other genes were found expressed more frequently in the frontal cortex of an AD patient than a non-AD subject (Yasojima et al., 1997
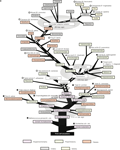
). One of the later two genes, gfap, was a known gene encoding glial fibrillary acidic protein, and the other was a novel gene, named kf-1. The kf-1 gene is expressed most prominently in the brain and more or less throughout the entire body, but least, if any, in the liver, which implies that kf-1 may not be a housekeeping gene. Human kf-1 has four exons ranging over approximately 30 kb, and is mapped to chromosome 2p11.2. The protein structure deduced from the cDNA sequences has revealed that human KF-1 protein (GenBank Acc No. BAA19739) consists of 685 amino acids, and contains a possible leader peptide (amino acid positions 1–19), two membrane-spanning segments (326–345 and 366–380), and a RING-H2 finger motif
(621–662) close to the C-terminus (Figure 1
A). KF-1-like proteins have been found in other animals including fish (Danio rerio), lancelet (Blanchistoma floridae), sea urchin (Strongylocentrotus purpuratus), sea anemone (Nematostella vectensis), and tablet animals (Trichoplax adhaerens) (Figure 1
A). Molecular phylogenetic studies suggest that genes for the animal and vertebral proteins may be kf-1 orthologues (Figures 1
B,C, respectively). Unexpectedly, KF-1 homologues do not exist in insects (Drosophila melanogaster) and thread worms (Caenorhabditis elegans, and C. briggsae), even though it is likely that the prototype gene appeared in the very early phase of animal evolution, before the separation of Placozoa and Eumetazoa (Miller and Ball, 2008
; Srivastava et al., 2008
). Homologues of the prototype do not exist in prokaryotes, plants, or fungi, but are found first in one of the most primitive multicellular animals, such as tablet animals (Schubert, 1993
) (Figure 1
D). The results may be consistent with the report based on the EST analysis that some genes, formerly thought to be vertebrate inventions, must have been present in the common metazoan ancestor (Kortschak et al., 2003
). Choanoflagellates (Monosiga brevicollis, and M. ovata), supposedly one of the most closely related unicellular protists to animals (King et al., 2008
), do not possess kf-1 (Figure 1
D).
Figure 1. Molecular phylogenetic studies of the kf-1 gene. (A) The alignment of KF-1 protein sequences. The sequence identities (%) given at the ends were obtained against human sequences minus the regions after the RING-H2 finger motif, namely after amino acid position 621 in human KF-1. Asterisks show the alignment sites used for inferring the molecular phylogenetic tree
in (B). The two transmembrane regions of human KF-1 are indicated by ‘=’ and the RING-H2 finger motif for zinc-binding is indicated by ‘#’. The number given at the right side indicates the amino acid position of the C-terminal amino acid in each line. The alignment positions, including gaps, are given on the top right of each row. Hs, Dr, Bf, Sp, Nv, and Ta denote Homo sapiens, Danio rerio, Blanchistoma floridae, Strongylocentrotus purpuratus, Nematostella vectensis and Trichoplax adhaerens, respectively. The gene identification (GI) numbers are 1945615, 52219054, 210102052, 115660659, 156406687, and 196009364 in the respective species. Blast Expect values (blastp with BLOSUM62 for Matrix) of Dr, Bf, Sp, Nv, and Ta against Hs are E < 10−130, E = 2 × 10−130, E = 3 × 10−117, E = 3 × 10−104, and E = 3 × 10−57, respectively. CS denotes consensus amino acid. (B) The molecular phylogenetic tree for animal KF-1. The tree is inferred by the genetic algorithm-based maximum likelihood method (Katoh et al., 2001
). The evolutionary rate heterogeneity among the sites was taken into account by assuming Yang’s discrete gamma model (Yang, 1994
) and an optimized shape parameter alpha of 1.93. Highly homologous sites without gap sites (404 amino acid sites) were selected for the comparison as indicated by asterisks in (A). The numbers at each branching node represent the bootstrap probability. The branching orders within the ellipse are not significant. The shaded region is expanded in (C) for vertebral KF-1. GI numbers are given in (A). (C) The maximum likelihood tree for vertebral KF-1, which corresponds to the shaded region in (B). Highly homologous sites without gap sites within the vertebrate sequences (580 amino acid sites; not shown) were used for tree inference. The optimized shape parameter alpha is 0.43. The numbers at the branching nodes are the bootstrap probabilities. GI numbers are given in (A) for H. sapiens and D. rerio, and 2058263, 126305140, 71896439, and 20126691 for M. musculus, M. domestica, G. gallus, and X. laevis, respectively. (D) Presence/absence, generation, and transmission of kf-1 on the evolutionary tree of life. The tree is essentially the same as the one, that was previously presented (Hashimoto-Gotoh et al., 2003
), with minor modifications and corrections except for the genes concerned. ‘?’ with bi-arrows and two open stars or crosses denotes uncertainty of the precise position of generation or loss of kf-1, respectively. Circles or crosses represent species in which the presence or absence of kf-1 has been confirmed in the Gen Bank database on January 29, 2009. In the case of sea squirt (Ciona intestinalis), the presence of kf-1 is represented by a triangle as only fragmented amino acid sequences have been found similar to those of KF-1. This is probably due to an incomplete determination of genomic sequences. Detailed data on which this illustration is essentially based are given in (A), (B), and (C).
The KF-1 protein is an E3 ubiquitin ligase that may modulate the cellular protein levels of its unknown substrate(s) (Lorick et al., 1999
). The expression of kf-1 is also increased in the frontal cortex and hippocampus after chronic administration of SSRI in rats (Yamada et al., 2000
). Furthermore, rat kf-1 expression is elevated after physical antidepressant treatments, such as electroconvulsive therapy (Nishioka et al., 2003
), and repetitive transcranial magnetic stimulation (Kudo et al., 2005
). This implies that the up-regulation of kf-1 expression is associated with some physiological responses to antidepressant treatments rather than being caused by a chemical reaction to serotonergic compounds such as SSRIs. It was, however, not clear whether this was a result, cause, or coincidental side effect of antidepressive processes.
As a possible correlation between KF-1 and AD has been suggested (Yasojima et al., 1997
), and as depression can occur early in the course of AD in ps1 mutation carriers unaware of their genetic status (Ringman et al., 2004
), the first working hypothesis was that presenilins could be KF-1 substrates in the endoplasmic reticulum (ER) associated degradation (ERAD) pathway (see Carvalho et al., 2006
; Lorick et al., 2006
). Therefore, the intracellular localization of KF-1 was examined in comparison to that of presenilins known to be located on ER (Kovacs et al., 1996
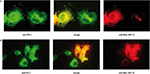
). The results have revealed that KF-1 is co- localized with both PS1 and PS2 (Figures 2
A,B and Tsujimura et al., 2008
), which implies that KF-1 may in fact be an ER-ubiquitin ligase. The co-localization of KF-1 with other ER markers, such as Der-1 and VCP, has also been observed (Maruyama et al., 2008
).
Figure 2. Immunostaining of Xenopus KF-1 tagged with the Myc epitope and PSα or PSβ co-expressed in COS-1 cells. (A) Proteins were stained with anti-Myc antibody probed with rhodamin conjugated anti-mouse IgG for KF-1 (red) and anti-PSα (Xenopus PS1 homologue) probed with fluorescein isothiocyanate (FITC) conjugated to anti-rabbit IgG for PSα (green). Images of ‘anti Myc (KF-1)’ and ‘anti PSα’ are merged (‘merge’). (B) The same as (A) except PSβ (Xenopus PS2 homologue) was used instead of PSα. KF-1 is co-localized with either PSα or PSβ as indicated in yellow or orange (‘merge’).
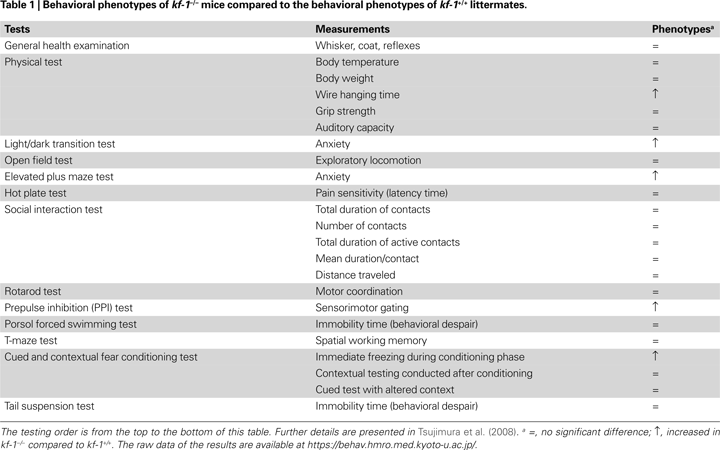
Although mouse kf-1 is expressed in many tissues, particularly in the brain, the lack of kf-1 does not lead to any abnormalities in appearance or behaviors, including body weight, reproductive capability, exploratory locomotion, nociception, social behavior, motor coordination, behavioral despair, spatial working memory, and context memory (Table 1
).
The exception is that kf-1−/− mice display a pronounced increase in anxiety-like behavior in the light/dark transition test (stay time in light compartment: p = 0.0004; number of transitions: p = 0.0392) (Tsujimura et al., 2008
). Consistently, the mice also show ‘timidity-like’ responses under stressful situations, such as prolonged wire-hanging time, enhanced immediate freezing with an aversive foot shock, and decreased locomotor activity at heights (Table 1 and Tsujimura et al., 2008
). As significant differences are not observed in body weight and grip strength, and in general exploratory locomotion, the ‘timidity-like’ responses are likely to be related to psychological but not physical alterations in kf-1−/− mice. This implies that KF-1 plays a role in emotional control by suppressing anxiety at least in mice. In this context, it is of particular interest to note that a number of SNP
s are reported in the coding and non-coding exonic regions of human kf-1 according to the NCBI’s SNP database, and some are supposed to substantially modulate KF-1 activity in homozygous or dual heterozygous carriers; for example, rs35921467 resulting in a frame shift at amino acid position 180, rs17857046 and rs17853383 in critical amino acid substitutions (S to P and P to H) at 251 and 502, respectively, and rs11695337 in a termination codon at 626.
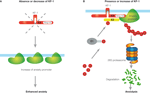
As KF-1 is an E3 ubiquitin ligase located on ER, KF-1 may be responsible for conducting the ERAD pathway, in which some factors promoting anxiety may be targeted. Therefore, the absence or reduction of KF-1 activity may result in ‘enhanced anxiety’ by increasing putative anxiety promoters (Figure 3
A). Regarding serotonergic metabolisms, it is not clear at the moment whether or how the ERAD pathway directed by KF-1 is associated with the serotonergic pathway, and it has to be elucidated in future. However, KF-1’s targets for proteasomal degradation should be neither the serotonin receptor (5-HT1A) nor the transporter (5-HTT), because a lack of these proteins increases anxiety-like behaviors (Heisler et al., 1998
; Holmes et al., 2003
).
Figure 3. Schematic representation of the anxiety suppressor model through the ERAD pathway. (A) When KF-1 is absent or decreases, the putative anxiety promoter will be increased on the ER, resulting in ‘enhanced anxiety’. (B) When KF-1 is present or increases, the putative anxiety promoter will be degraded or lost due to 26S proteasomal degradation through the KF-1 mediating ERAD pathway, resulting in ‘anxiolysis’. ‘Ub’ represents the ubiquitin molecule. ‘E1’, ‘E2’, and ‘E3’ represent the ubiquitin-activating enzyme, ubiquitin-conjugating enzyme, and ubiquitin ligase, respectively.
Ubiquitination
plays an essential regulatory role in all critical eukaryotic cellular processes. Proteasomal degradation through the ERAD pathway is not an exception. It has been well established that these processes play an important role in a variety of human somatic diseases, ranging from cancer, viral infection, diabetes, and inflammation to muscle wastage and neurodegenerative disorders (see Kostova et al., 2007
; Petroski, 2008
). However, there are no reports on the animal specific ubiquitin ligase, which is responsible for emotional control or mental disorders. Concerning neurodegenerative disorders, Parkin, another ER-ubiquitin ligase, plays a key role in preventing the degeneration of dopaminergic neurons in Parkinson’s disease (PD). A recessive mutation in parkin is responsible for the neurodegenerative disorder known as the autosomal recessive juvenile form of Parkinsonism, the most common form of familial PD (Kitada et al., 1998
). Human parkin with 12 exons ranging over approximately 1.5 Mb is located on chromosome 6q25.2.27, and encodes an E3 ubiquitin ligase, consisting of 465 amino acids, with two RING finger motifs. Parkin substrates have been identified being degraded through the ERAD pathway, such as Pael-R, CDCrel-1, α-Syn, and synphilin-1 (Yang et al., 2003
). The accumulation of these proteins in dopaminergic neurons leads to ER stress-induced apoptosis, resulting in PD (see von Coelln et al., 2004
). Promoting their degradation would aid PD patients by preventing neurodegeneration. In fact, the neuronal synthesis of the Parkin substrate Pael-R has been shown to cause a loss of dopaminergic cells in transgenic Drosophila models. The co-expression of parkin results in the degradation of Pael-R and nearly completely blocks neurodegeneration, whereas interfering with the function of endogenous Drosophila Parkin promotes Pael-R accumulation and augments its toxicity (Yang et al., 2003
). Hence, the over-expression of parkin has emerged as a powerful approach to PD with complementary effects to approaches described for the use of neurotrophic factors against PD (Ulusoy and Kirik, 2008
). An analogy could be made by having such Drosophila models to study the role of KF-1 in anxiety/depression. However, the analogy may not be feasible as no evidence is available for the involvement of neuronal cell death in anxiety/depression, and kf-1 is absent in Drosophila (Figure 1
D). It should be noted that kf-1−/− mice have increased PPI
values (Table 1
), which implies that at least one of the KF-1 target proteins is involved in regulation of sensorimotor gating
. In such cases, over-expression of kf-1 might cause serious side effects as the PPI values are usually reduced in schizophrenic patients. Despite this, one cannot rule out the possibility of kf-1 up-regulation as an anxiolytic/antidepressant treatment.
Instead, kf-1−/− mice provide a powerful tool for some pharmacological approaches to anxiety/depression. For example, by using the mice, one can identify the putative anxiety promoters that are degraded through the ERAD pathway mediated by KF-1 and are responsible for the sensitivity to potential signs of danger or to stressful situations (Figures 3
A,B). KF-1 substrates acting as anxiety promoter may be found by the computed differential screening in 2-dimensional gel electrophoresis (see Marengo et al., 2008
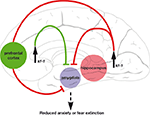
) of cell homogenates derived from the frontal cortex or hippocampus of kf-1−/− and kf-1+/+ mice. As the expression of kf-1 is up-regulated in the frontal cortex and hippocampus after antidepressant treatments, it is likely that the anxiolytic/antidepressive effect of KF-1 functions primarily in these tissues and that their inhibitory signals are transmitted to the amygdala to prevent the manifestation of anxiety or fear (Figure 4
). This proposal may be highly hypothetical and hence has to be elucidated in future studies. To conclude, examining kf-1-/- mice in a simple light/dark transition test is an effective method of screening novel anxiolytic/ antidepressant drugs that inhibit the putative anxiety promoters.
Figure 4. A circuitry presentation of the anxiety suppressor model by the up-regulation of kf-1 expression. The circuitry model was constructed by considering the fear extinction neural model (Sotres-Bayon et al., 2006
). The anxiolytic pathway after kf-1 up-regulation is incorporated into the model.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
We are grateful to Drs. M. Matsuki, and K. Yamanishi for their collaborations during this work, and Messrs. N. Matsumoto and T. Aramaki, and Drs. H. Yamagishi, S. Clapcote, and M. Nakagawa for their support and helpful advices to our work. THG thanks Drs. T. Mizuno, K. Yasojima, Y. Ariga, Y. Fukumaki, C. Mitchell and M. Miyata for valuable discussions and comments. NI is a recipient of Grant-in-Aid for Scientific Research on Priority Area “Comparative Genomics” from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
SNP: DNA sequence variation, called single nucleotide polymorphism, occurring on a single nucleotide in the genome among individual members of a species or between paired chromosomes within an individual. ‘SNP (pronounced snip)’ was defined initially as that with a minor allele frequency of ≥1%, but this definition is rather artificial and not meaningful.
Ubiquitination: Ubiquitination (or ubiquitylation) is the post-translational tagging reaction mediated by E3 ubiquitin ligase to a protein with one or more highly conserved peptide molecules called ‘ubiquitin’ consisting of 76 amino acids. Ubiquitination targets the substrate protein for proteasomal degradation.
King, N., Westbrook, M. J., Young, S. L., Kuo, A., Abedin, M., Chapman, J., Fairclough, S., Hellsten, U., Isogai, Y., Letunic, I., Marr, M., Pincus, D., Putnam, N., Rokas, A., Wright, K. J., Zuzow, R., Dirks, W., Good, M., Goodstein, D., Lemons, D., Li, W., Lyons, J. B., Morris, A., Nichols, S., Richter, D. J., Salamov, A., Sequencing, J. G., Bork, P., Lim, W. A., Manning, G., Miller, W. T., McGinnis, W., Shapiro, H., Tjian, R., Grigoriev, I. V., and Rokhsar, D. (2008). The genome of the choanoflagellate Monosiga brevicollis and the origin of metazoans. Nature 451, 783–788.
Kovacs, D. M., Fausett, H. J., Page, K. J., Kim, T. W., Moir, R. D., Merriam, D. E., Hollister, R. D., Hallmark, O. G., Mancini, R., Felsenstein, K. M., Hyman, B. T., Tanzi, R. E., and Wasco, W. (1996). Alzheimer-associated presenilins 1 and 2: neuronal expression in brain and localization to intracellular membranes in mammalian cells. Nat. Med. 2, 224–229.
Petroski, M. D. (2008). The ubiquitin system, disease, and drug discovery. BMC Biochem. 9, S7. doi: 10.1186/1471-2091-9-S1-S7. Available at: http://www.pubmedcentral.nih.gov/picrender.fcgi?artid=2582801&blobtype=pdf
.
Ringman, J. M., Diaz-Olavarrieta, C., Rodriguez, Y., Chavez, M., Paz, F., Murrell, J., Macias, M. A., Hill, M., and Kawas, C. (2004). Female preclinical presenilin-1 mutation carriers unaware of their genetic status have higher levels of depression than their non-mutation carrying kin. J. Neurol. Neurosurg. Psychiatr. 75, 500–502.
Srivastava, M., Begovic, E., Chapman, J., Putnam, N. H., Hellsten, U., Kawashima, T., Kuo, A., Mitros, T., Salamov, A., Carpenter, M. L., Signorovitch, A. Y., Moreno, M. A., Kamm, K., Grimwood, J., Schmutz, J., Shapiro, H., Grigoriev, I. V., Buss, L. W., Schierwater, B., Dellaporta, S. L., and Rokhsar, D. S. (2008). The Trichoplax genome and the nature of placozoans. Nature 454, 955–960.
Tsujimura, A., Matsuki, M., Takao, K., Yamanishi, K., Miyakawa, T., and Hashimoto-Gotoh, T. (2008). Mice lacking the kf-1 gene exhibit increased anxiety – but not despair-like behavior. Front. Behav. Neurosci. 2, 4. doi: 10.3389/neuro.08.004.2008. Available at: http://www. frontiersin.org/ behavioralneuroscience/paper/10.3389/neuro.08/004.2008/pdf/
.
Yamada, M., Yamada, M., Yamazaki, S., Takahashi, K., Nishioka, G., Kudo, K., Ozawa, H., Yamada, S., Kiuchi, Y., Kamijima, K., Higuchi, T., and Momose, K. (2000). Identification of a novel gene with RING-H2 finger motif induced after chronic antidepressant treatment in rat brain. Biochem. Biophys. Res. Commun. 278, 150–157.
Yamasaki, N., Maekawa, M., Kobayashi, K., Kajii, Y., Maeda, J., Soma, M., Takao, K., Tanda, K., Ohira, K., Toyama, K., Kanzaki, K., Fukunaga, K., Sudo, Y., Ichinose, H., Ikeda, M., Iwata, N., Ozaki, N., Suzuki, H., Higuchi, M., Suhara, T., Yuasa, S., and Miyakawa, T. (2008). α-CaMKII deficiency causes immature dentate gyrus, a novel candidate endophenotype of psychiatric disorders. Mol. Brain 1, 6.
Yasojima, K., Tsujimura, A., Mizuno, T., Shigeyoshi, Y., Inazawa, J., Kikuno, R., Kuma, K., Ohkubo, K., Hosokawa, Y., Ibata, Y., Abe, T., Miyata, T., Matsubara, K., Nakajima, K., and Hashimoto-Gotoh, T. (1997). Cloning of human and mouse cDNAs encoding novel zinc finger proteins expressed in cerebellum and hippocampus. Biochem. Biophys. Res. Commun. 231, 481–487.