1
Division of Systems Neuroscience, Tohoku University Graduate School of Life Sciences, Sendai, Japan
2
Kavli Institute for Systems Neuroscience and Centre for the Biology of Memory, Norwegian University of Science and Technology, Trondheim, Norway
Using recombinant rabies virus (RV), we developed a dual transsynaptic retrograde tracing technique in the rat central nervous system. Two strains of recombinant RV, injected into two separate loci of the brain, were taken up through axon terminals and carried retrogradely and transsynaptically from neuron to neuron. Each viral strain expresses a unique marker in infected neurons. Therefore, neurons that project transsynaptically to two brain loci can be detected by double-labeling. In this review, we will introduce the advantage of dual viral tracing by recombinant RV, and will also address some potential weaknesses of this technique. Although false negative results may arise due to interference between two strains of RV, the ability of the recombinant RV to visualize the morphology of the infected cell and to infect primates in addition to rodents will make this technique a potential tool to provide new insights into the complex organization of brain networks.
The central nervous system (CNS) is composed of multiple, interconnected neural circuits. One way to elucidate the complex wiring of the brain is to use tracer substances (dyes or proteins), which are taken up by neurons and carried by axonal transport. In cases where the tracer is transported retrogradely, such as Fast Blue, Diamidino Yellow or Fluorogold, neurons projecting to the injected site will be labeled. Such methods potentially visualize the complete set of converging inputs to a particular brain region. The combination of two retrograde tracers which can be distinguished, for example by different fluorescence wavelength (Kuypers et al., 1980
), is referred to as double retrograde labeling
. This method is useful to assess whether a particular brain region has divergent projections to several regions. Moreover, one can reveal whether the projections originate from single neurons (divergent projection) or from nearby distinct neurons (parallel projection). These tracing techniques have provided us remarkable insights into the afferent and efferent connections of brain regions.
Since the early 1990’s, viruses have also been used to visualize neuronal connections (Kuypers and Ugolini, 1990
). Unlike most conventional chemical retrograde tracers, viral retrograde tracers reliably move from one neuron to another neuron mainly via synapses. More importantly, they replicate in infected neurons, resulting in strong amplification of signals. These features enable precise tracing of chains of neurons connected via synapses (Figure 1
). A variety of neurotropic viruses have been used for such transsynaptic tracing
, namely, herpes simplex virus type 1 (HSV-1), pseudorabies (PRV), and rabies virus
(RV). Among these viruses, the Bartha strain of PRV (PRV-Bartha) and the challenge virus standard (CVS) of RV are used often as transsynaptic retrograde tracers (for reviews, see Aston-Jones and Card, 2000
; Kelly and Strick, 2000
).
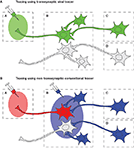
Figure 1. Schematic diagram of the comparison of viral and conventional tracers in tracing neural circuits which converge to Region A. (A) By injecting a retrograde viral tracer into Region A, the virus will first infect the neurons that directly project to the injection site (first-order neuron) in Region B, and then spread further to the synaptically connected upstream neurons (second-order neuron) in Region C. Therefore, transsynaptic labeling enables us to trace chains of specific neurons, such as projection neurons which interconnect distinct brain regions or interneurons which modulate the activity of such projection neurons. (B) By using conventional non-transsynaptic tracers, several experiments with different injection sites must be conducted in order to reveal the afferent neural circuits: injection to Region A to detect first-order neurons, and injection to Region B to detect second-order neurons. However, in this way, neurons that are not connected with the circuit A may also be stained, such as the neuron in Region D.
To further elucidate the complicated neural networks in the CNS, such as the interaction of multiple neural circuits, a method which combines double retrograde labeling with viral transsynaptic tracing would be useful. The idea of this method, dual viral tracing, is explained in Figure 2
. Two strains of recombinant viruses are used as transsynaptic tracers, each of which expresses a unique marker protein. With this method, two different neuronal circuits can be labeled distinctly, and a single neuron, from which the two diverging circuits are originating, can be visualized by co-expression of two marker proteins (double labeling). In our original paper (Ohara et al., 2009
), we described dual viral tracing in the rat CNS using recombinant strains of RV. In this article, we will review the reported use of various dual viral tracer approaches, the advantages of dual viral tracing using RV recombinants and point out potential weaknesses and promises of the latter method.
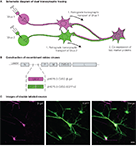
Figure 2. Dual viral tracing using genetically modified virus. (A) Neurons which have a divergent projection to two brain regions will be infected by retrograde transsynaptic transport of the two recombinant viruses, and will be double-labeled by the co-expression of the two marker proteins. (B) Two RV recombinants were generated for dual viral tracing by inserting different marker genes encoding either β-gal or EGFP. (C) Neuron double-labeled by β-gal (magenta, left panel) and EGFP (green, center panel) can be seen in white in the merged image (right panel). Scale bar = 20 μm.
Dual viral tracing has been introduced by Jansen et al. (1995)
using recombinant strains of PRV-Bartha. PRV-Bartha is an attenuated strain of PRV which transports along the axon in a retrograde direction. Unlike other α-herpesviruses (HSV-1 and PRV), PRV-Bartha is less likely to induce cell lysis and therefore will hardly infect anatomically non-connected nearby cells by local spread of the virus. Because of these features, this virus is widely used as a transsynaptic retrograde tracer in rodents. Jansen et al. (1995)
used two recombinants of PRV-Bartha which express unique markers [gC viral glycoprotein and β galactosidase (β-gal)] to address a longstanding issue: whether there are neurons which regulate the functions of two distinct autonomic systems. The two recombinant strains were injected, respectively, into one of two peripheral organs, adrenal gland and stellate ganglion (the major sympathetic source that innervates the heart). The distribution of double labeled neurons in the hypothalamus and brainstem demonstrated the existence of neurons which innervate these two peripheral structures. However, the rate of successful double infection in this first study was very low (8 out of 256 rats) since the virus strains used in this study were nonisogenic strains that differ in their invasive properties. The strain with stronger invasive properties spreads within neural circuits faster than the other strain and prevents the other strain from subsequently infecting the same neuron (Kim et al., 1999
). The low rate of successful double infection due to this interference problem (viral interference
) has been settled partially by using two isogenic PRV recombinants which present similar invasive properties (Cano et al., 2004
). Dual viral tracing with these isogenic strains, which express either β-gal or enhanced green fluorescent protein (EGFP) as a marker protein, has been used to identify brainstem neurons that control the simultaneous activation of multiple muscles that are involved in certain actions, such as respiration and emesis (Billig et al., 2000
). This method has also been used to demonstrate the existence of neurons which simultaneously coordinate the activations of both motor and autonomic systems (Krout et al., 2003
; Kerman et al., 2006
).
In the original paper (Ohara et al., 2009
), we used recombinant strains of RV instead of the previously used PRV recombinants. Similar to PRV-Bartha, RV transports along the axon exclusively in the retrograde direction and spread to neurons that are synaptically connected (Iwasaki and Clark, 1975
; Charlton and Casey, 1979
). However, there are several differences in the infection properties in these two viruses. Unlike PRV infection, RV selectively infects neurons, not glia cells. Furthermore, RV does not induce cellular lysis and subsequent local viral spread (Ugolini, 1995
). One further prominent characteristic of RV is that it can infect primates in addition to rodents whereas PRV cannot (Callaway, 2008
). These features make RV a powerful tool to reveal the complicated connections within the CNS especially in non-human primates, such as connections of the basal ganglia, cerebellum and cerebral cortex (Kelly and Strick, 2003
; Hoshi et al., 2005
), or specifically in the visual pathway (Nassi and Callaway, 2006
).
We developed a recombinant RV which was derived from a vaccine strain of RV (HEP-Flury) except that the glycoprotein (G) gene was taken from the CVS strain (Inoue et al., 2004
). This recombinant virus (rHEP5.0-CVSG) effectively propagated transsynaptically in a retrograde direction. Furthermore, the morphological features of the infected neurons were clearly visualized by using antibodies against the expressed marker protein, such as EGFP or β-gal. We used this recombinant RV and achieved dual transsynaptic tracing in the entorhinal-hippocampal network of the rat. The entorhinal cortex and hippocampal region are known to contribute to encoding, consolidation and retrieval of declarative memories. The afferent and efferent connections within these regions have been well studied (for an overview, see Witter and Amaral, 2004
). Briefly, the areas of hippocampus, including the dentate gyrus, areas CA3, CA1 and the entorhinal cortex (EC), exhibit a series of connections that are ideal to test the potential of our recombinant strains of viruses for single and double labeling (Figure 3
A). Axons of pyramidal cells in the hippocampal CA3 region projects bilaterally to CA1 (Swanson et al., 1980
; Laurberg and Sorensen, 1981
; Li et al., 1994
). In turn, CA1 pyramidal cells project to EC, and with the exception of those in the most dorsal tip of CA1, all neurons project strictly ipsilaterally (van Groen and Wyss, 1990
). On the basis of this established anatomy, we hypothesized that a single CA3 cell influences EC bilaterally, with only one CA1 synapse in between. This inferred connectivity, however, has never been demonstrated experimentally. Our new dual viral tracing technique would be perfectly suited to confirm this connection in a straightforward way.
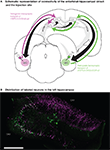
Figure 3. Experimental design of dual viral tracing in the rat entorhinal-hippocampal circuit. (A) rHEP5.0-CVSG- β-gal and rHEP5.0-CVSG-EGFPx2 were injected to the left and right MEC, respectively. The viruses, transsynaptically transported from the bilateral MEC will go through CA1 and will first meet in the hippocampal CA3 region. (B) In field CA1, neurons are labeled only by the virus (rHEP5.0-CVSG- β-gal) injected into the ipsilateral MEC, whereas β-gal (magenta) and GFP (green) labels intermingle in CA3 region. Note that there are double-labeled neurons (white) in CA3. MEC, medial entorhinal cortex. Scale bar = 500 μm.
We created two recombinant strains of virus, each expressing one of two distinct marker proteins, β-gal and EGFP (pHEP5.0-CVSG- β-gal and pHEP5.0-CVSG-EGFPx2), and injected each of these two strains to the left and right medial entorhinal cortex (MEC) separately. 6 days after injection, which is an approximate time required for the recombinant virus to infect the second-order upstream neurons, many labeled neurons were observed in the dorsal hippocampus. CA1 neurons were predominantly labeled by the recombinant strain that was injected into the ipsilateral MEC, whereas β-gal and EGFP labels intermingled in the bilateral CA3 region. 12.5–21.7% of the labeled CA3 neurons were double-labeled (Figure 3
B). In some double-labeled neurons, the level of expression of at least one of the two marker proteins was high enough to visualize the morphological feature of the neuron. The results demonstrate that single neurons in CA3 are connected to MEC bilaterally, with only one synaptic relay at the CA1. In view of the known organization of this neuronal chain, the present finding was expected, but is the first direct demonstration of this transsynaptic connection from individual CA3 neurons to MEC bilaterally. This study also demonstrated the potential use of this dual viral tracing method using RV recombinants to reveal the complicated circuits in the CNS.
Although the dual viral tracing method using RV recombinants is powerful in dissecting multiple circuits, a number of issues should be considered. First, not all cell types or connections can be infected. Viral specificity occurs in case of granule cells in the dentate gyrus, which cannot be infected by RV (Jackson and Reimer, 1989
), and CA3 pyramidal cells will not be infected through synapses of Schaffer collaterals by PRV-Bartha (Sik et al., 2006
). Second, it is important to assess the possibility of multiple transsynaptic routes between two brain structures. An efficient way is to carry out single viral tracing experiments first and to compare the results with established connectivity. In case of the connections between CA3 and EC, single tracing experiments indicated that a possible connection mediated by the lateral septum (Witter and Amaral, 2004
) was an unlikely explanation for the reported transsynaptic labeling in CA3 following injections in EC.
The use of these single tracing experiments also allowed assessing the temporal progression of the two viral strains in the targeted circuit, which is essential to realize efficient double-labeling. As shown in the PRV studies, false negative results may arise in dual viral tracing: neurons which innervate the two injection regions may not be double-labeled due to viral interference. We have demonstrated the effect of this interference on double labeling in an in vitro preparation, and showed that the efficiency of double labeling decreases as the interval between the two infections increases, in accordance with previous report using PRV recombinants (Banfield et al., 2003
). Thus, for efficient double labeling using viral tracers, two strains of viruses must infect a neuron with only a few hours difference. To achieve this, we must consider the time required for the two recombinant strains to infect the neurons of interest. This difference is determined by the propagation speed of the two strains and by the infection pathways, such as the number of synapses, the length of the axon, and the strength of connections (Lycke and Tsiang, 1987
; Ugolini, 1995
). Therefore, in order to realize efficient double-labeling the timing of the two injections should be adjusted, taking into account the specifics of each of the two strains such that the two viral strains reach the neurons of interest within a short time period.
Dual transsynaptic tracing using recombinant RV is a powerful method since RV specifically infects neural circuits in a retrograde manner in a broad range of animals including rodents and primates. Although there still are some problems in the use of this method as described above, recent developments in genetic techniques regarding RV provides the hint of solutions to these problems. Wickersham et al. (2007)
reported an elegant technique to label neurons that project to a single, genetically targeted neuron in vitro by applying two modifications to RV: (1) deleting the G gene from the genome, and (2) providing the virus with an avian virus envelope (EnvA). This modified virus can only infect specific neurons expressing the EnvA receptor (TVA), and spread transsynaptically to directly connected neurons only if the infected cell is also transfected with G gene. This strategy may be used to restrict the infection pathway of the virus to a certain route and will facilitate the interpretation of the transsynaptic labeling.
Genetic modification may also provide a solution to the problem of false negative results due to viral interference. A previous study in Vesicular stomatitis virus, which belongs to the same family, Rhabdoviridae, as RV, has shown that the G protein interferes with the viral uptake of a subsequent virus (Whitaker-Dowling et al., 1983
). Therefore, recombinant RV with low G protein expression may diminish the effect of viral interference, but right now this problem has not been solved. We must further realize that quantitative analysis is difficult in dual viral tracing since the results may underestimate the real number of collateralized neurons, due to interference. On the good side, those neurons that express both marker proteins undoubtedly have anatomical connections to both of the infected target areas. Furthermore, since our recombinant RV can reveal the morphological characteristics of the labeled neurons, the double-labeled neuron can be morphologically typed. The method can also be combined with antibody staining against neural cell markers to classify the cell in more detail. It may also be interesting to combine this method with anterograde tracing, which visualizes inputs from a certain region to the virally-infected neurons. This will allow qualitative and quantitative analyses using confocal- and potentially electron-microscopy. We thus expect that this dual viral tracing with recombinant RV, for which we provided proof of principle in the entorhinal-hippocampal system, will be of use to untangle complex connections in the CNS and to visualize the complex architectures that underlie a variety of brain functions.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
This work was supported by Grant-in-Aid for Scientific Research on Priority Areas (Integrative Brain Research) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
Rabies virus: Nonsegmented negative-strand RNA-virus which belongs to the genus Lyssavirus of the family Rhabdoviridae. Rabies virus infects hosts at the periphery and moves to the central nervous system via retrograde axonal and transsynaptic transport. This virus finally causes fatal disease in human and animals.
Viral interference: An interference mechanism that is established after primary infection, preventing the infected cell from being superinfected by the same or a closely related virus. This inhibition mechanism may occur at various stages of the viral life cycle, such as surface binding, endocytosis, and subsequent replication steps.
Ohara, S., Inoue, K., Yamada, M., Yamawaki, T., Koganezawa, N., Tsutsui, K., Witter, M. P., and Iijima, T. (2009). Dual transneuronal tracing in the rat entorhinal-hippocampal circuit by intracerebral injection of recombinant rabies virus vectors. Front. Neuroanat. 3, 1. doi: 10.3389/neuro.05.001.2009.