- 1Department of Cardiac Surgery, Shanghai Chest Hospital, Shanghai Jiao Tong University, Shanghai, China
- 2Department of Pharmacy, Shanghai Chest Hospital, Shanghai Jiao Tong University, Shanghai, China
- 3Department of Cardiovascular Surgery, Mount Sinai Hospital, Icahn School of Medicine at Mount Sinai, New York, NY, United States
- 4Department of Radiology, Shanghai Chest Hospital, Shanghai Jiao Tong University, Shanghai, China
Objective: By assessing the normal dimensions and the relationship between the aortic root and leaflets in Chinese population, the objective of this three-dimensional computed tomography (3DCT)-based study was to establish a matching reference for leaflets and aortic root for aortic valve (AV) repair.
Method: Electrocardiogram-gated multi-detector CT was performed on 168 Chinese participants with a normal aortic valve. Measurements of the aortic annuli and leaflets were obtained. The correlations between and the ratios of the specific root and leaflet measurements were analyzed. The references for the leaflet and root dimensions were suggested based on geometric height (gH) using a linear regression equation. The utility of the ratios was tested with CT images of 15 patients who underwent aortic valve repair.
Result: The mean annulus diameter (AD), sino-tubular junction (STJ) diameter, geometric height (gH), effective height (eH), free margin length (FML), commissural height (ComH), inter-commissural distance (ICD), and coaptation height (CH) were 22.4 ± 1.7 mm, 27.3 ± 2, 0.4 mm, 15.5 ± 1.7 mm, 8.9 ± 1.2 mm, 32.0 ± 3.4 mm, 17.9 ± 1.9 mm, 23.1 ± 2.3 mm, and 3.1 ± 0.6 mm, respectively. The gH/AD, FML/ICD, and eH/ComH ratios were 0.69 ± 0.07, 1.38 ± 0.08, and 0.50 ± 0.07, respectively. The gH correlated with all other leaflet and root measurements (P < 0.01), whereas the FML demonstrated a better correlation with ICD compared with gH (R2 = 0.75, and R2 = 0.37, respectively). The FML/ICD and eH/ComH ratios might be used to assess leaflet-root mismatch and post-repair leaflet billowing.
Conclusion: The normal aortic valve measurements based on 3DCT revealed a specific relationship between the root and leaflets; and this will guide the development of an objective method of aortic valve repair.
Introduction
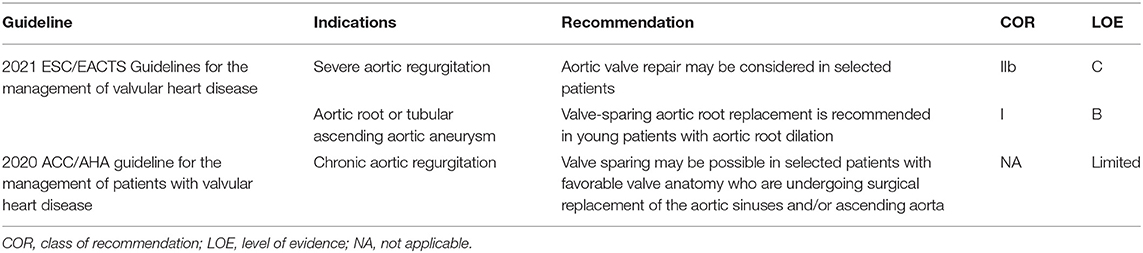
Aortic valve repair and valve-sparing aortic root procedures have become increasingly popular interventions for tricuspid and bicuspid valve anomalies (Table 1). Unlike the matured surgical technique of mitral valve repair, the standardization and the reproducibility of surgical approaches to aortic valve (AV) repair remain controversial, as they are technically demanding and the surgical principles for re-establishing the natural correlation between the dimensions of all valve components vary with the different institutions (1–5). Given that the dimensions of the leaflets change with aortic root dilatation, correcting the dilated root based on normal value alone, without restoring an appropriate leaflet-root interrelationship, may, otherwise, induce post-repair leaflet billowing (6). Thus, a profound understanding of normal AV anatomy and setting quantitative methods to evaluate leaflet-root mismatch in aortic regurgitation (AR) are of vital importance for AV repair.
Aortic valve is a three-dimensional (3D) anatomical structure and functional apparatus that consists of root annuli (basal ring and STJ) and leaflets. A deep understanding of the 3D configuration of AV is a prerequisite for determining the causes of AR and the surgical approach to AV repair (7–9). In clinical practice, echocardiography is the major imaging modality for evaluating the AV anatomy, and it can be operated at the bedside and is easy to use. However, the spatial resolution of echocardiography is low, and because of software limitation of the company, several crucial parameters cannot be obtained as desired by the operator. Multi-detector 3D computed tomography (3DCT) provides images with high spatial resolution, and customized software is available for post-processing, which enables the visualization and evaluation of specific 3D valve parameters. Therefore, given the above advantages, the 3DCT has been established as the gold standard for pre-operative trans-catheter aortic valve implantation planning (10, 11).
In this study, we aimed to explore the anatomy of the AV as a functional apparatus of the aortic root and leaflets using 3DCT in a large cohort of healthy Chinese. We also aimed to more comprehensively analyze the spatial and functional relationship among key AV components, set the reference for leaflet/root mismatch, and establish critical information about CT applications in patients with AR and aortic root aneurysm related to the indications for AV repair technique.
Materials and Methods
Study Population
The CT datasets obtained from adults who visited the Cardiovascular Health Screening Center between 2017 and 2018 were retrospectively analyzed in this study. The inclusive criteria were the following: (1) patients were confirmed to have normal AV function by transthoracic echocardiogram; (2) patients underwent ECG-gated multi-detector CT with contrast enhancement; (3) patients' age > 18 years; (4) the demographic information of the patient were available and complete. Patients with diseases that altered blood flow patterns across the AV or the anatomy of the aortic root (e.g., heart valve and congenital heart diseases, uncontrolled hypertension, Marfan syndrome) were excluded. Two experienced radiologists examined the dataset independently. Patient will only be enrolled in the study when both physicians reached the agreement on a structurally and functionally normal AV with normal size aortic roots.
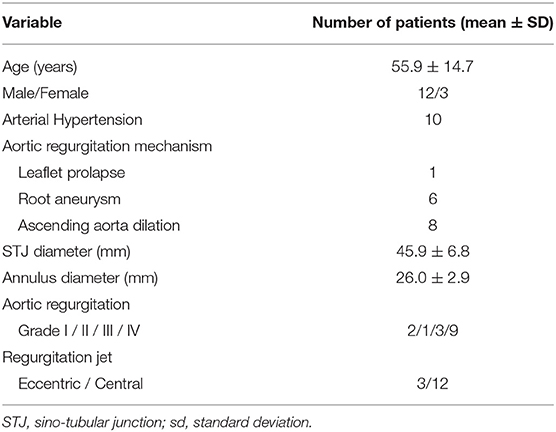
As a result, the study included 168 adults, 87 men and 81 women. The mean age was 50.2 ± 15.1 years (range, 26–77 years). The enrolled participants were divided into four groups according to age: group 1, 20–35 years (n = 39); group 2, 35–50 years (n = 43); group 3, 50 to 65 years (n = 50); and group 4, 65 to 80 years (n = 36). Additionally, from 2017 to 2018, a total of 15 patients with AR underwent AV repair with or without root replacement; both pre-operative and post-operative CT images were analyzed. Baseline characteristics are presented (Table 2). The Institutional Review Board (IRB) or equivalent ethics committee of the Shanghai Chest Hospital approved the study protocol and publication of data. Patient written consent for the publication of the study data was waived by the IRB, as the research involved no more than minimal risk to subjects.
ECG-Gated 3DCT Measurements
All 3DCT data were systematically analyzed using the Osirix software (Version 9.5.1 Geneva, Swiss). Multiplane reconstruction was performed to visualize and measure dimensions of key AV elements outlined by Hagendorff et al. (12). The anatomic structures and the parameters were mainly categorized into two parts from a geometric and functional perspective and measured by two study investigators independently. A video shows how these AV elements were reconstructed and measured using 3DCT images with detailed workflow (Videos 1–8 and Video legends 1–8).
Aortic Root Configuration
Two-dimensional measurements of the aortic root were made at end-diastole (80% RR interval) at these levels of the functional annuli: (1) annulus or basal ring (determined by the transverse plane crossing the nadirs of the leaflets, Video 1) and (2) sino-tubular junction (STJ, determined by the narrowest plane connecting the sinus and ascending aorta, Video 8). The following parameters were obtained: annulus and STJ surface area, annulus diameter (AD) and STJ diameter (derived using the surface area under the assumption that the annulus and STJ were circular), STJ area to annulus area ratio (STJ/Annulus area), and STJ diameter to annulus diameter ratio (STJ/AD) (Figures 1A,B).
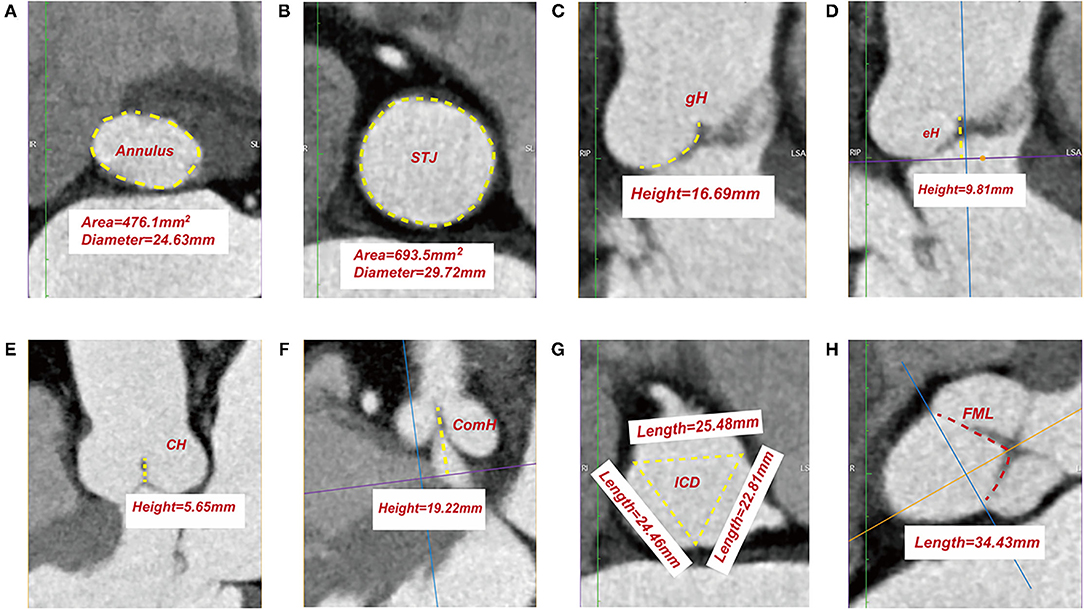
Figure 1. Measurements of leaflet and root parameters in normal aortic valve by three-dimensional reconstruction of ECG-gated computed tomography. Surface area and diameter of annulus (A). Surface area and diameter of sino-tubular junction (B). Measurement of geometric height (C). Measurement of effective height (D). Measurement of coaptation height (E). Measurement of commissure height (F). Measurement of inter-commissural distance (G). Measurement of free margin length (H). STJ, sino-tubular junction; gH, geometric height; eH, effective height; CH, coaptation height; ComH, commissure height; ICD, inter-commissural distance; FML, free margin length.
Leaflet Geometry
Leaflet geometries were only assessed at end-diastole (80% RR interval), during which the image artifact resulting from the vibration of the leaflets is least, and the closure of the AV is best evaluated with leaflets fully pressurized by the diastolic pressure. The leaflet parameters investigated are as follows (most definitions and nomenclatures are by Hagendorff et al.) (12): geometric height (gH), referring to the distance between the curved length of the respective cusp during diastole from the aortic insertion in the nadir of the sinus to the central part of the free margin (Video 2); effective height (eH), referring to the difference in height between the annular plane and the free margin of each cusp during diastole (Video 3); coaptation height (CH, Video 4); free margin length (FML, Video 7); commissure height (ComH), referring to the distance between the commissural apex and the annular plane (Video 5); inter-commissural distance (ICD), referring to the distance between the two adjacent commissural apices (Video 6) (Figures 1C–H).
Leaflet-Root Ratios
The ratios of the average gH to the calculated AD (gH/AD), average FML (gH/FML), average eH (eH/gH), and calculated STJ diameter (gH/STJ); the ratio of the average FML to the average ICD (FML/ICD); and the ratio of the average eH to the average ComH (eH/ComH) of the leaflets were determined in our study to demonstrate the relationship between the leaflet and root geometries.
Pre- and Post-repair CT Images of 15 Patients With AR
All 15 patients enrolled for analysis underwent ECG-gated 3DCT within 1 month prior to the AV repair; CT scan was repeated to obtain post-repair images between post-operative days 7 and 10. The aforementioned measurements and leaflet-root ratios were obtained to evaluate pre-operative AR mechanism and to assess the post-repair outcome.
Statistical Analyses
Continuous variables were assessed using the Kolmogorov-Smirnov test for normal distribution and presented as the mean ± standard deviation. Independent-sample t-test or one-way ANOVA was used to compare the means of the groups. Linear regression was performed to calculate the Pearson correlation coefficient (R), and a p < 0.05 was considered statistically significant. Linear regression equations were constructed using gH as the variable and the other parameters as dependent variables. The R statistical software version 3.6.1 was used to perform all the statistical analyses. For all the parameters derived from the 3DCT measurements, the two independent observers achieved inter-observer variability of 0.909–0.987 (intra-class correlation coefficients) and an intra-observer variability of 0.920–0.989. All measurements were <10% of coefficient of variation (range, 4.2–9.1%).
Results
Normal Aortic Valve
The measurements, with their ranges and means, for the normal AV are reported based on root configuration and leaflet geometries, and the data are stratified by age and gender.
For the normal AV, the average annulus area was 396.1 ± 60.3 mm2 (range, 278.3–516.0 mm2), the area-derived AD was 22.4 ± 1.7 mm (range, 18.8–25.6 mm), the average STJ area was 590.5± 101.7 mm2 (range, 349.8–771.9 mm2), the area-derived STJ diameter was 27.3 ± 2.4 mm (range, 21.1–31.4 mm), the STJ/annulus area ratio was 1.5 ± 0.2 (range, 1.0–2.1), the STJ/AD ratio was 1.2 ± 0.1 (range, 1–1.4), the average gH was 15.5 ± 1.7 mm (range, 11.0–21.6 mm) with the highest mean gH (16.2 ± 1.6 mm) in the non-coronary (NC) leaflet and the shortest in the right coronary (RC) leaflet (14.9 ± 1.7 mm), the mean eH was 8.9 ± 1.2 mm (range, 6.29–12.0 mm), and the three leaflets showed no significant difference (p = 0.867). The mean FML and ICD, all being longest in the RC and shortest in the left coronary (LC) leaflet, were 32.0 ± 3.4 mm (range, 21.3–40.7 mm) and 23.1 ± 2.3 mm (range, 17.4–28.2 mm), respectively. The mean ComH, being longest between the NC and RC leaflets, was 17.9 ± 1.7 mm (range, 13.0–23.7 mm). The mean CH, being longest for NC leaflets, was 3.1 ± 0.6 mm (range, 1.33–5.26 mm) (Supplementary Tables S1, S2).
Specific ratios were established to elucidate the spatial relationship between the leaflets and root geometries; the average gH/AD, gH/FML, eH/gH, FML/ICD, and eH/ComH were 0.69 ± 0.07, 0.49 ± 0.06, 0.58 ± 0.07, 0.57 ± 0.05, 1.38 ± 0.08, and 0.50 ± 0.07, respectively. The age groups showed no statistically significant difference (Supplementary Table S3). The absolute values of the root and leaflet parameters were significantly greater in men than in women at all levels (p < 0.001), but the same leaflet-root ratios were maintained. Although no significant differences were found among the age groups for most parameters, the trends of greater STJ/annulus ratios were observed along with an increase in age (p < 0.01) (Supplementary Table S4).
All the measured leaflet and root dimensions showed positive correlations with gH (p < 0.0001), whereas FML demonstrated a better correlation with ICD compared with gH (R2 = 0.75, R2 = 0.37). eH was also positively correlated with ComH but not as well as with gH (R2 = 0.16, R2 = 0.44) (Figures 2, 3). Based on the gH ranging from 12 to 25 mm, we calculated the related leaflet and root parameters using linear regression equations (Supplementary Table S5).
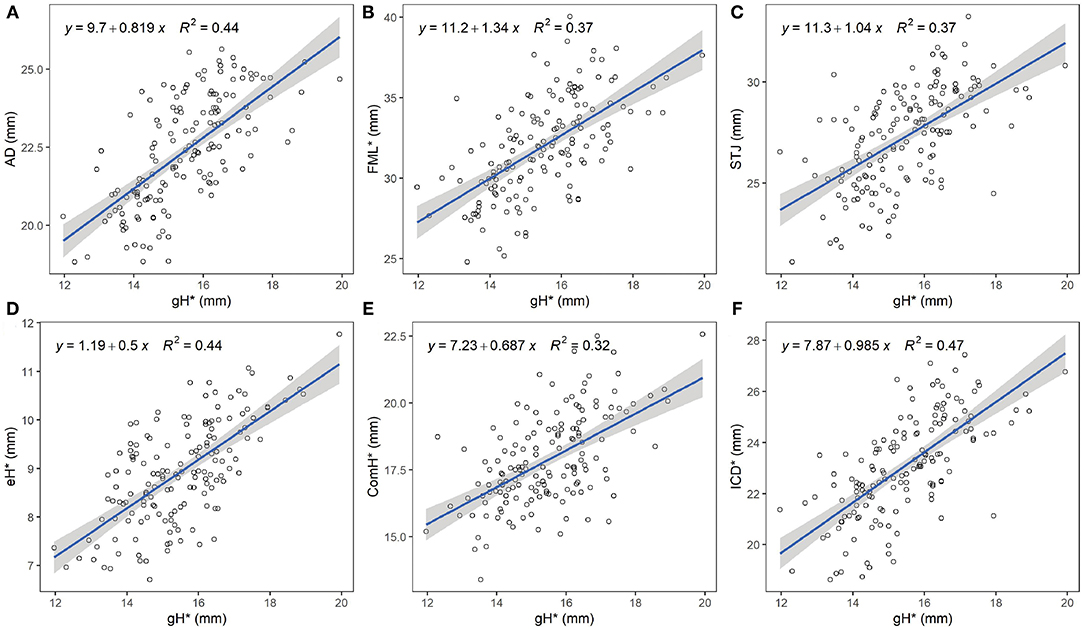
Figure 2. Relationship between geometric height and aortic valve measures. The blue line represents the best fit linear regression model of the data points. The shaded gray area represents the 95% confidence level interval for predictions from each linear model. (A) Linear regression between gH and AD. (B) Linear regression between gH and FML. (C) Linear regression between gH and STJ area-derived diameter. (D) Linear regression between gH and eH. (E) Linear regression between gH and ComH (F) Linear regression between gH and ICD. gH, geometric height; AD, annular diameter; FML, free margin length; STJ, sino-tubular junction; eH, effective height; ComH, commissural height; ICD, inter-commissural distance. *, average measures of three leaflets.
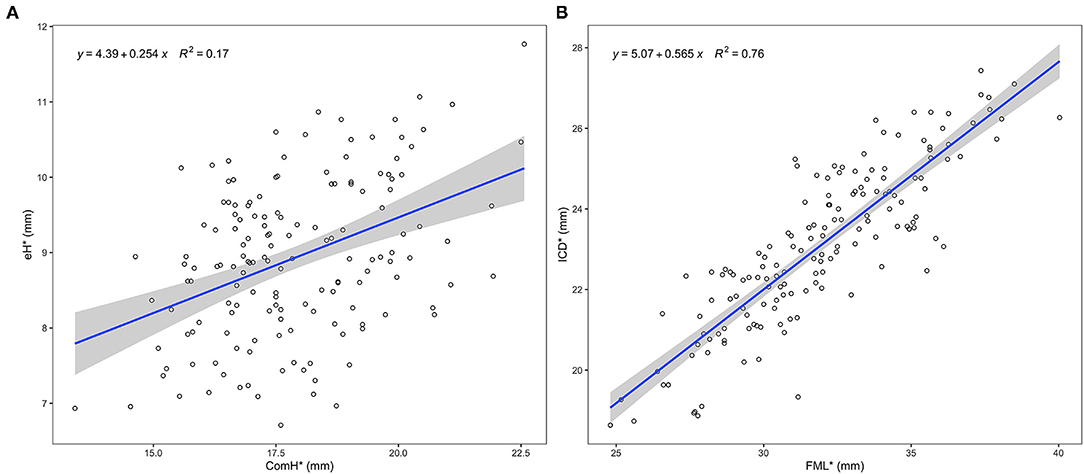
Figure 3. Relationship between specific leaflet and root measures. The blue line represents the best fit linear regression model of the data points. The shaded gray area represents the 95% confidence level interval for predictions from each linear model. (A) Linear regression between ComH and eH. (B) Linear regression between FML and ICD. eH, effective height; ComH, commissural height; FML, free margin length; ICD, inter-commissural distance; *, average measures of three leaflets.
Cohort of 15 AR Patients With Pre- and Post-repair CT Images
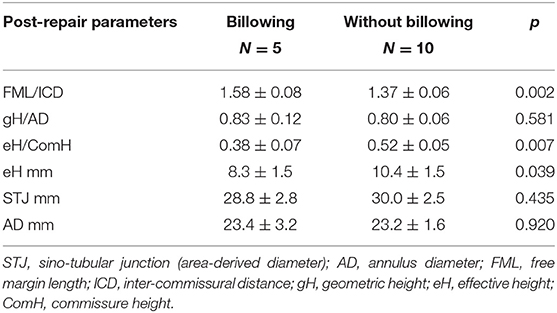
The three patients with leaflet prolapse causing eccentric AR jet were identified by bent leaflet belly in the pre-repair CT images, which was not presented in the other 12 patients with central jet (Figure 4). After AV repair, leaflet billowing without prolapse defined by altered coaptation configuration from normal reversed “Y” to reversed “T” shape in the post repair CT images was found in five patients (Figure 5). The post-operative echocardiogram demonstrated no more than mild AR in all 15 patients. The detailed valve parameters and leaflet-root ratios based on CT images were measured and calculated in pre- and post-repair groups (Table 3).
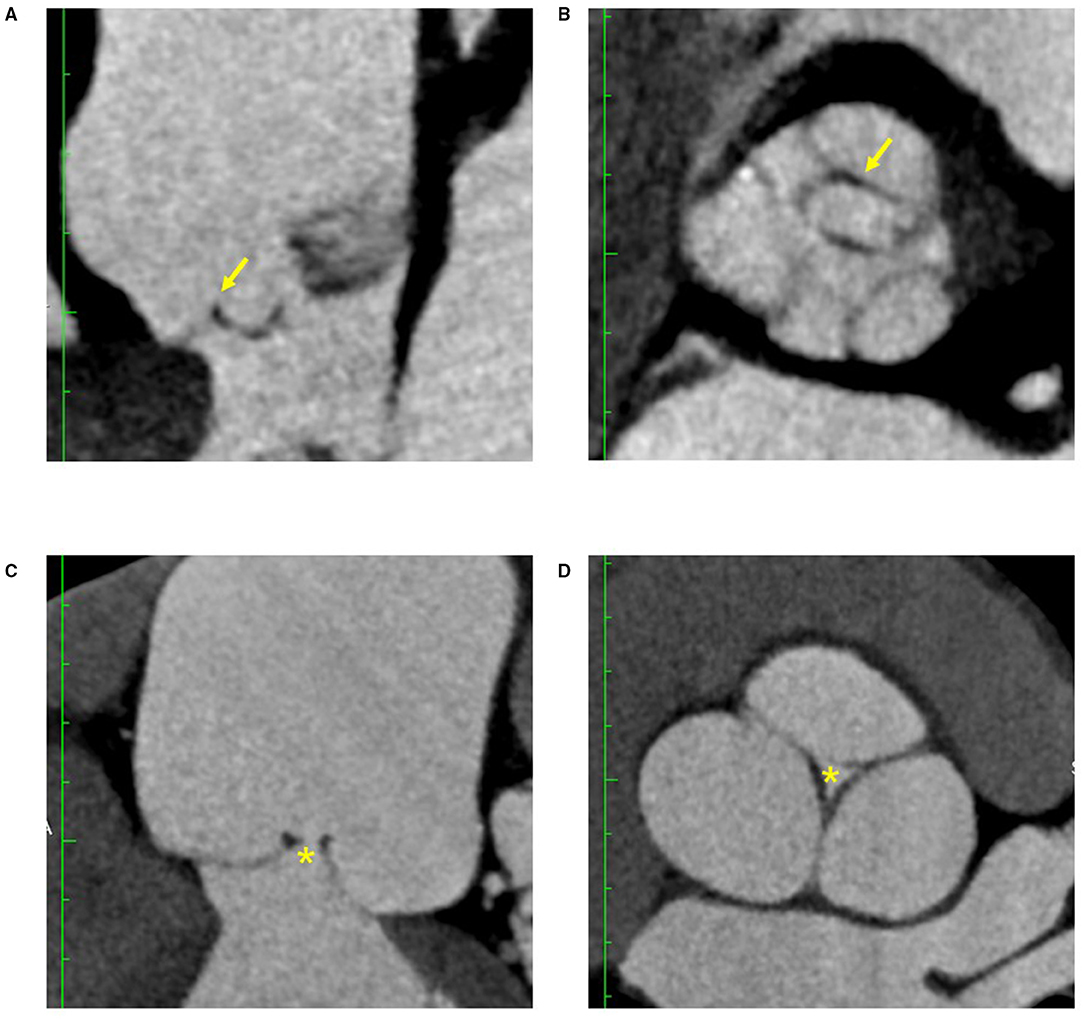
Figure 4. CT features of AR with eccentric jet and central jet. Bent leaflet belly indicated by arrows were both identified in long axis (A) and short axis (B) of reconstructed CT images in AR with eccentric jet. Loss of coaptation indicated by asterisks were both identified in long axis (C) and short axis (D) of the reconstructed CT images in AR with central jet. AR, aortic regurgitation.
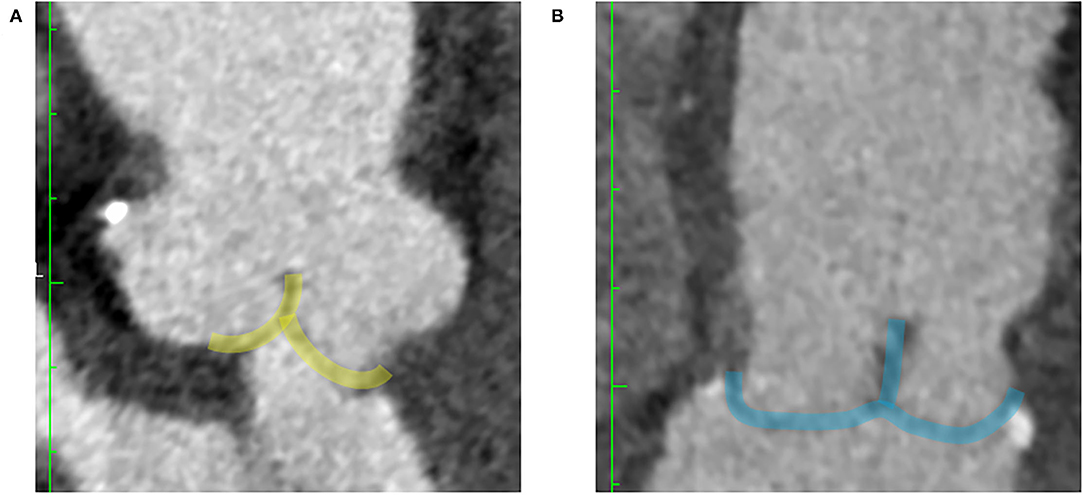
Figure 5. CT features of post-repair valve coaptations. The shaded green area delineated the post-repair coaptation without leaflet billowing in a reversed Y form (A). The shaded blue area delineated the post-repair coaptation with leaflet billowing in a reversed T form (B).
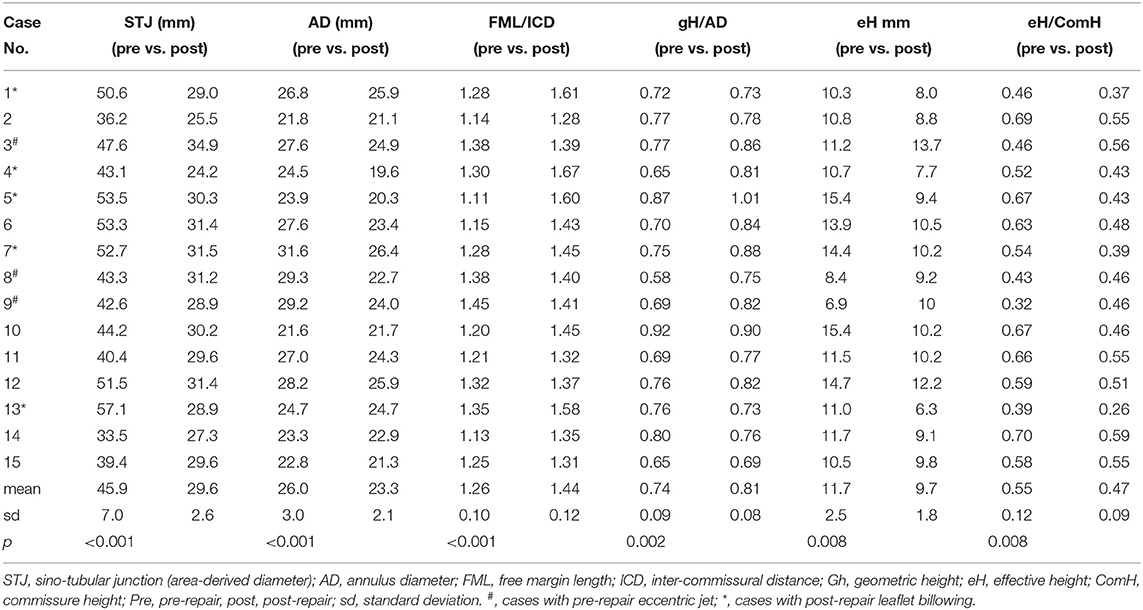
Table 3. Statistical comparison and detailed pre- and post-operative leaflet-root parameters of 15 aortic valve repair cases.
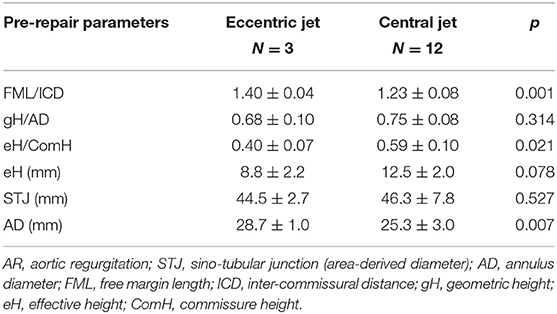
The valve parameters and leaflet-root ratios were markedly different between pre- and post-repair group. The 15 patients were divided into two groups based on classification of AR jet (eccentric vs. central) pre-operatively, and they were categorized into two groups according to post-repair leaflet billowing post-operatively.
Pre-operatively, the patients with central jet showed significantly smaller FML/ICD ratio than patients with eccentric jet (1.23 ± 0.08 vs. 1.40 ± 0.04, p = 0.001). Both eH and eH/ComH were smaller in the eccentric jet group, but significance was only observed for eH/ComH (p = 0.021). Meanwhile, eccentric jet group showed significantly larger AD than central jet group (28.7 ± 1 vs. 25.3 ± 3 mm, p = 0.007) (Table 4).
Post-operative FML/ICD ratios were much larger in the patients showing post-repair leaflet billowing than in those without billowing (1.58 ± 0.08 vs. 1.37 ± 0.06, p = 0.002). Both eH and eH/ComH were smaller in the billowing group (Table 5).
Discussion
Our study aimed to comprehensively explore the anatomy of normal tricuspid AV with 3DCT, with a focus on the relationship between leaflet geometry and functional root annuli dimension in vivo. CT data were also collected on a cohort of 15 patients who underwent AV repair to assess potential implication of specific leaflet-root relationship. Several studies have already reported the dimensions of normal AV based on ex vivo evaluations of the root specimens (13–15), without physiologic blood pressurization; thus, the leaflet-root relationship has not been thoroughly established and requires further clarification. The development of cardiac CT and dedicated software for post-processing allows for a comprehensive 3D assessment of AV in vivo with improved spatiotemporal resolution (16). Moreover, these measurements can be acquired robustly with proposed workflow, and both built-in CT workstation and commercialized software can be utilized with minimal measurement variations.
The configurations of the leaflet and root of the normal aortic valve are asymmetric. Consistent with the findings of previous studies (17, 18), leaflet asymmetry was also confirmed in our study. We took a further step with 3DCT analysis; we took more measurements of the leaflets in the normal AV and found that they were asymmetrical at multiple geometric levels. The shortest gH and the longest FML of the RC leaflet make it slender than the NC and LC leaflets, which could be the cause of the more frequent prolapse of the RC leaflet in AR. On the other hand, the longest ICD of the RC leaflet may be responsible for ensuring an eH that is equivalent to those of the NC and LC leaflets, as revealed by our study. This suggested that a specific leaflet-root ratio, which was evaluated but not fully validated by small a sample study by De Kerchove et al. (15), may exist at the STJ level to maintain valve competency. Similarly, the ComHs were also uneven, being highest between the NC and RC leaflets and lowest between the NC and LC leaflets, and this resulted in the tilting of the aortic root with non-parallel STJ and basal ring (19). Based on the above in vivo 3DCT findings, the current AV repair strategy does not seem to consider the natural AV asymmetry by positioning the ComHs symmetrically, both circumferentially and longitudinally, within the Dacron graft during valve-sparing root procedures.
Leaflet remodeling and adaption in aortic root dilation have been confirmed by multiple studies (16, 18, 20), and the gH and FML, as important leaflet sizing parameters, were found involved in this adaptive process. The size of the native leaflet indicated by gH is the major constraining factor for a successful AV repair without patch augmentation, which makes gH an important sizing parameter (21). Being another key element of the leaflet dimension, FML is closely associated with leaflet adaption and prolapse in the cases of root dilation with or without AR (15, 22, 23). Leaflet adaptions, along with root dilation, which commonly presents as the elongation of FML and an increase in gH (16, 20, 24), have not been fully addressed in AV repair due to frequent leaflet billowing after root replacement or annuloplasty despite CH restoration (6). Current AV repair strategies do not sufficiently consider the leaflet-root relationship, which may lead to over- or under-correction by targeting eH at a fixed value (eH > 9 mm) (25, 26). Over reduction of STJ in root dilation with an elongated FML may also require excessive leaflet plication resulting in a “large leaflet in a small root,” and long-term hemodynamics under these circumstances is not ideal. Furthermore, using gHs of <16 mm as a relative contraindication for AV repair may need reconsideration, given the small habitus in Asian group (21, 27). Therefore, we established the leaflet and root measurements based on gH using a linear regression equation to guide the AV repair and improve the practicality of the current study for a wider range of gH values (12–25 mm) (Table 5). Additionally, the better correlation of FML with ICD than with gH (Figure 5) implies that an independent approach may be necessary for free-margin plication and STJ reconstruction for better post-repair valve configurations. We hope that the development of dedicated sizers will improve the usefulness of the above measurement.
The classification of functional aortic leaflet-root abnormality was established by El Khoury to guide the assessment of AR mechanism and AV repair for the tricuspid AV (Table 6) (28). Based on our normal AV findings, we attempted to define these abnormalities as leaflet-root mismatches and suggest further divisions, based on the 3DCT measurements, into leaflet-STJ mismatch defined as FML/ICD ratio of 1.38 and leaflet-basal ring mismatch defined as gH/AD ratio of 0.69 derived from the mean value of normal population. The goal of AV repair is to achieve sufficient CH and adequate eH proportional to ComH defined as eH/ComH ratio of 0.5 (29, 30), which can be translated into restoring a normal gH/AD ratio by basal ring annuloplasty to correct annular ectasia and a normal FML/ICD ratio by combining FML plication and STJ reconstruction to correct leaflet prolapse and STJ dilation (Figure 6). Based on these normal ratios, we retrospectively analyzed the pre- and post-operative 3DCT images of 15 patients who underwent AV repair at our center without applying the matching methodology as suggested by the current study, and the results showed that the deviation from the normal reference values of the key leaflet-root ratios can be used for pre-operative AR assessment and post-operative review of the technique-related leaflet billowing.

Table 6. AV repair techniques based on the classification of AR with description of disease mechanisms.
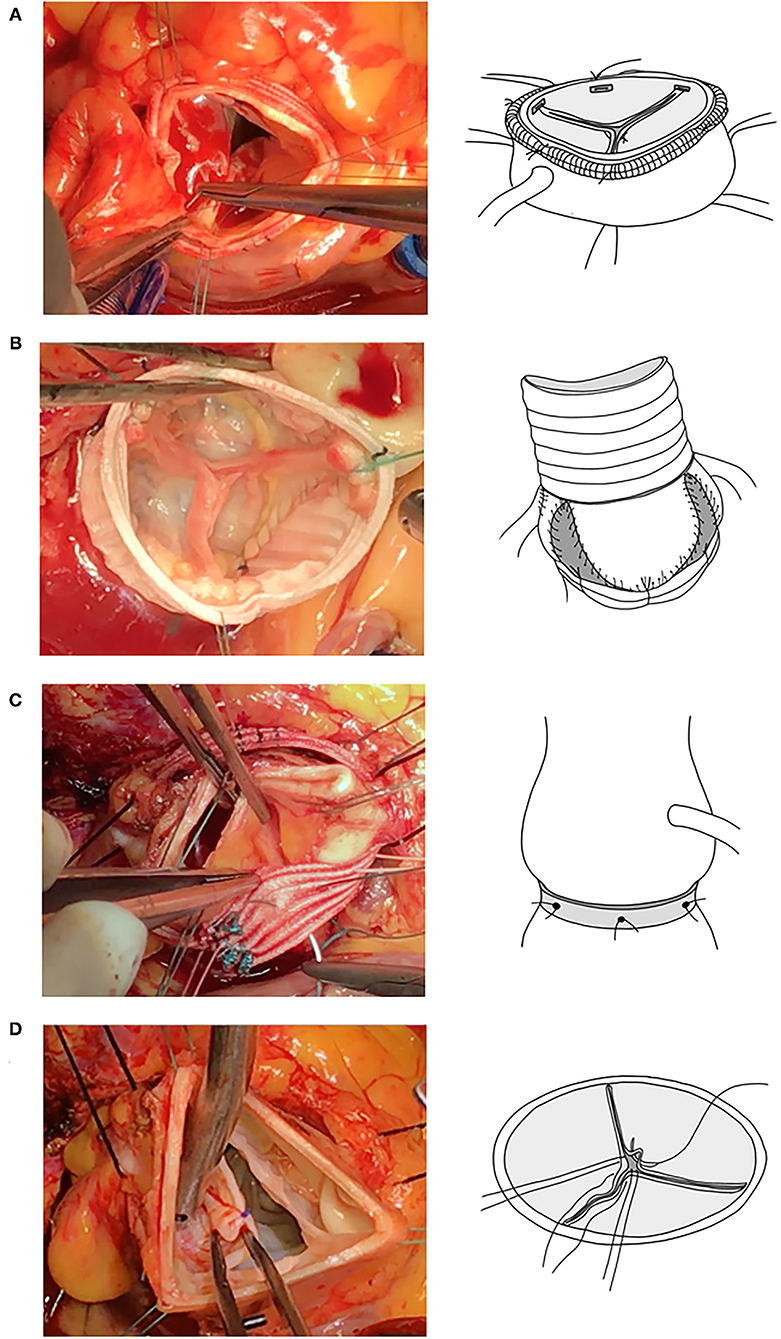
Figure 6. Important current AV repair techniques. From top downward (left column, surgical views; right column, illustrative figures), STJ remodeling (A), valve sparing root replacement (B), annuloplasty (C), and FML central plication (D). AV, aortic valve; STJ, sino-tubular junction; FML, free margin length.
When normal leaflet-root ratios were referred for pre-operative AR assessment, patients of our cohort with central AR featured a reduced FML/ICD ratio (1.28) and increased the eH/ComH ratio (0.59), probably indicating FML tethering and relative diastolic restrictive motion secondary to insufficient leaflet remodeling in relation to root dilatation. In contrast, the FML/ICD ratio (1.40) was increased and the eH/ComH ratio (0.4) was decreased in the case of eccentric AR resulted from leaflet prolapse, rather expressing excessive leaflet motion.
In terms of assessment of post-repair leaflet billowing, it was featured by a significantly increased FML/ICD ratio (1.58) and a reduced eH/ComH ratio (0.38). This phenomenon could be due to the over-reduction of STJ which led to leaflet redundancy. Although central plication of free margin was performed routinely to restore normal eH, the post-repair eH was still insufficient for the dilated root to reach the eH/ComH ratio of 0.5. In contrast, it can be seen from those cases without post-repair leaflet billowing that no leaflet-root mismatch was defined by altered ratios; both FML/ICD (1.37) and eH/ComH (0.52) ratios were comparable to normal population.
Study Limitation
We did not investigate the impact of age and body surface area on AV configuration, as have been discussed extensively in the literature (7, 14, 31–33), although they have important effects on AV repair. Because of the small habitus of our cohort compared with that of the western population, the gH measurements did not cover the 22–25 mm range, which limits the application of the gH-based ratio. The extreme gH measurements (<12 mm) also need to be validated. The calculated leaflet and root parameter reference values for AV repair also require further validation. The retrospective approach to analysis of the application of leaflet-root ratio and the low number of patients in a small cohort of 15 patients were the major limitations. The analyzed dataset of normal AV valves was retrospectively enrolled by two independent physicians, based on the CT images and medical records of the patients. While the data might be biased, they can still reflect leaflet-root relationships as potential reference for AV repair, especially when normal anatomy is absent. Extensive application of CT to perioperative root assessment could also be limited due to ionizing radiation. Comparative studies of the pre- and post-AV repair 3DCT images of AR patients with large samples are needed for further exploration. All measurements were performed based on 3DCT without validation by direct comparison via surgical inspection.
Conclusion
We described the normal dimensions of the aortic root configuration and leaflet geometry in the tricuspid AV based on 3DCT findings. Moreover, the findings from this study on the relationship between the leaflet and root with further investigations into the key geometrical features of valve closure may help improve the techniques for restoring normal functional anatomic relationships of pathologically altered aortic roots. A dedicated sizing parameter for assessing key functional elements with proper indexation, such as gH-indexed AD, FML-indexed STJ, and ComH-indexed eH, may be developed to standardize leaflet repair for root pathology.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics Statement
The studies involving human participants were reviewed and approved by the Institutional Review Board (IRB) or Equivalent Ethics Committee of the Shanghai Chest Hospital. Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements.
Author Contributions
TY, DZ, and IE-H had the idea for the study design and critically corrected the manuscript. QN and YS were involved in data collection and data verification. TY and HW drafted the manuscript. All authors read and approved the final manuscript.
Funding
The study was supported by the grant from Shanghai Municipal Commission of Health and Family Planning (20184Y0076 to TY) and the grant from Shanghai Hospital Development Center (SHDC12020124 to DZ).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
We would like to thank ZJ for her contribution in graphic illustrations.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fcvm.2021.731440/full#supplementary-material
Abbreviations
AV, aortic valve; AD, annulus diameter; AR, aortic regurgitation; CH, coaptation height; ComH, commissure height; 3DCT, three-dimensional computed tomography; eH, effective height; FML, free margin length; gH, geometric height; ICD, inter-commissural distance; LC, left coronary leaflet; NC, non-coronary leaflet; RC, right coronary leaflet; STJ, Sino-tubular junction.
References
1. Lansac E, Di Centa I, Sleilaty G, Lejeune S, Khelil N, Berrebi A, et al. Long-term results of external aortic ring annuloplasty for aortic valve repair. Eur J Cardiothorac Surg. (2016) 50:350–60. doi: 10.1093/ejcts/ezw070
2. Schneider U, Hofmann C, Aicher D, Takahashi H, Miura Y, Schafers HJ. Suture annuloplasty significantly improves the durability of bicuspid aortic valve repair. Ann Thorac Surg. (2017) 103:504–10. doi: 10.1016/j.athoracsur.2016.06.072
3. Zeeshan A, Idrees JJ, Johnston DR, Rajeswaran J, Roselli EE, Soltesz EG, et al. Durability of aortic valve cusp repair with and without annular support. Ann Thorac Surg. (2018) 105:739–48. doi: 10.1016/j.athoracsur.2017.09.019
4. Mastrobuoni S, De Kerchove L, Navarra E, Watremez C, Vancraeynest D, Rubay J, et al. Long-term experience with valve-sparing reimplantation technique for the treatment of aortic aneurysm and aortic regurgitation. J Thorac Cardiovasc Surg. (2019) 158:14–23. doi: 10.1016/j.jtcvs.2018.10.155
5. De Kerchove L, Mastrobuoni S, Boodhwani M, Astarci P, Rubay J, Poncelet A, et al. The role of annular dimension and annuloplasty in tricuspid aortic valve repair. Eur J Cardiothorac Surg. (2016) 49:428–37. doi: 10.1093/ejcts/ezv050
6. Ridley C, Sohmer B, Vallabhajosyula P, Augoustides JGT. Aortic leaflet billowing as a risk factor for repair failure after aortic valve repair. J Cardiothorac Vasc Anesth. (2017) 31:1001–6. doi: 10.1053/j.jvca.2017.02.019
7. Kunzelman KS, Grande KJ, David TE, Cochran RP, Verrier ED. Aortic root and valve relationships. Impact on surgical repair. J Thorac Cardiovasc Surg. (1994) 107:162–70. doi: 10.1016/S0022-5223(94)70465-1
8. De Kerchove L, Jashari R, Boodhwani M, Duy KT, Lengele B, Gianello P, et al. Surgical anatomy of the aortic root: implication for valve-sparing reimplantation and aortic valve annuloplasty. J Thorac Cardiovasc Surg. (2015) 149:425–33. doi: 10.1016/j.jtcvs.2014.09.042
9. De Paulis R, Salica A. Surgical anatomy of the aortic valve and root-implications for valve repair. Ann Cardiothorac Surg. (2019) 8:313–21. doi: 10.21037/acs.2019.04.16
10. Tops LF, Wood DA, Delgado V, Schuijf JD, Mayo JR, Pasupati S, et al. Noninvasive evaluation of the aortic root with multislice computed tomography implications for transcatheter aortic valve replacement. JACC Cardiovasc Imaging. (2008) 1:321–30. doi: 10.1016/j.jcmg.2007.12.006
11. Achenbach S, Delgado V, Hausleiter J, Schoenhagen P, Min JK, Leipsic JA. SCCT expert consensus document on computed tomography imaging before transcatheter aortic valve implantation (TAVI)/transcatheter aortic valve replacement (TAVR). J Cardiovasc Comput Tomogr. (2012) 6:366–80. doi: 10.1016/j.jcct.2012.11.002
12. Hagendorff A, Evangelista A, Fehske W, Schafers HJ. Improvement in the assessment of aortic valve and aortic aneurysm repair by 3-dimensional echocardiography. JACC Cardiovasc Imaging. (2019) 12:2225–44. doi: 10.1016/j.jcmg.2018.06.032
13. Vollebergh FE, Becker AE. Minor congenital variations of cusp size in tricuspid aortic valves. Possible link with isolated aortic stenosis. Br Heart J. (1977) 39:1006–11. doi: 10.1136/hrt.39.9.1006
14. Silver M. A., Roberts W. C. (1985). Detailed anatomy of the normally functioning aortic valve in hearts of normal and increased weight. Am J Cardiol. 55, 454–461. doi: 10.1016/0002-9149(85)90393-5
15. De Kerchove L, Momeni M, Aphram G, Watremez C, Bollen X, Jashari R, et al. Free margin length and coaptation surface area in normal tricuspid aortic valve: an anatomical study. Eur J Cardiothorac Surg. (2018) 53:1040–8. doi: 10.1093/ejcts/ezx456
16. Kim DH, Handschumacher MD, Levine RA, Sun BJ, Jang JY, Yang DH, et al. Aortic valve adaptation to aortic root dilatation: insights into the mechanism of functional aortic regurgitation from 3-dimensional cardiac computed tomography. Circ Cardiovasc Imaging. (2014) 7:828–35. doi: 10.1161/CIRCIMAGING.113.001976
17. Choo SJ, Mcrae G, Olomon JP, St George G, Davis W, Burleson-Bowles CL, et al. Aortic root geometry: pattern of differences between leaflets and sinuses of Valsalva. J Heart Valve Dis. (1999) 8:407–15.
18. Yang DH, Kim DH, Handschumacher MD, Levine RA, Kim JB, Sun BJ, et al. In vivo assessment of aortic root geometry in normal controls using 3D analysis of computed tomography. Eur Heart J Cardiovasc Imaging. (2017) 18:780–6. doi: 10.1093/ehjci/jew146
19. Lansac E, Lim HS, Shomura Y, Lim KH, Rice NT, Goetz WA, et al. Aortic root dynamics are asymmetric. J Heart Valve Dis. (2005) 14:400−7.
20. Thubrikar MJ, Labrosse MR, Zehr KJ, Robicsek F, Gong GG, Fowler BL. Aortic root dilatation may alter the dimensions of the valve leaflets. Eur J Cardiothorac Surg. (2005) 28:850–5. doi: 10.1016/j.ejcts.2005.09.012
21. Schafers HJ, Schmied W, Marom G, Aicher D. Cusp height in aortic valves. J Thorac Cardiovasc Surg. (2013) 146:269–74. doi: 10.1016/j.jtcvs.2012.06.053
22. De Kerchove L, Glineur D, Poncelet A, Boodhwani M, Rubay J, Dhoore W, et al. Repair of aortic leaflet prolapse: a ten-year experience. Eur J Cardiothorac Surg. (2008) 34:785–91. doi: 10.1016/j.ejcts.2008.06.030
23. Tamer S, Mastrobuoni S, Van Dyck M, Navarra E, Bollen X, Poncelet A, et al. Free margin length and geometric height in aortic root dilatation and leaflet prolapse: implications for aortic valve repair surgery. Eur J Cardiothorac Surg. (2020) 57:124–32. doi: 10.1093/ejcts/ezz132
24. Ballocca F, Ruggeri GM, Roscoe A, Thampinathan B, David TE, Lang RM, et al. Aortic root changes before and after surgery for chronic aortic dilatation: A 3D echocardiographic study. Echocardiography. (2019) 36:376–85. doi: 10.1111/echo.14216
25. Schafers HJ, Bierbach B, Aicher D. A new approach to the assessment of aortic cusp geometry. J Thorac Cardiovasc Surg. (2006) 132:436–8. doi: 10.1016/j.jtcvs.2006.04.032
26. Bierbach BO, Aicher D, Issa OA, Bomberg H, Graber S, Glombitza P, et al. Aortic root and cusp configuration determine aortic valve function. Eur J Cardiothorac Surg. (2010) 38:400–6. doi: 10.1016/j.ejcts.2010.01.060
27. Komiya T, Shimamoto T, Nonaka M, Matsuo T. Is small cusp size a limitation for aortic valve repair? Eur J Cardiothorac Surg. (2019) 56:497–502. doi: 10.1093/ejcts/ezz053
28. El Khoury G, Glineur D, Rubay J, Verhelst R, D'acoz Y, Poncelet A, et al. Functional classification of aortic root/valve abnormalities and their correlation with etiologies and surgical procedures. Curr Opin Cardiol. (2005) 20:115–21. doi: 10.1097/01.hco.0000153951.31887.a6
29. El Khoury G, De Kerchove L. Principles of aortic valve repair. J Thorac Cardiovasc Surg. (2013) 145:S26–29. doi: 10.1016/j.jtcvs.2012.11.071
30. Kari FA, Siepe M, Sievers HH, Beyersdorf F. Repair of the regurgitant bicuspid or tricuspid aortic valve: background, principles, and outcomes. Circulation. (2013) 128:854–63. doi: 10.1161/CIRCULATIONAHA.113.002475
31. Roman MJ, Devereux RB, Kramer-Fox R, O'loughlin J. Two-dimensional echocardiographic aortic root dimensions in normal children and adults. Am J Cardiol. (1989) 64:507–12. doi: 10.1016/0002-9149(89)90430-X
32. Zhu D, Zhao Q. Dynamic normal aortic root diameters: implications for aortic root reconstruction. Ann Thorac Surg. (2011) 91:485–9. doi: 10.1016/j.athoracsur.2010.10.058
Keywords: aortic root, aortic valve anatomy, leaflet-root mismatch, 3D computed tomography, aortic valve repair
Citation: Yang T, Wen H, El-Hamamsy I, Ni Q, Sun Y and Zhu D (2021) Relationship Between Leaflets and Root in Normal Aortic Valve Based on Computed Tomography Imaging: Implication for Aortic Valve Repair. Front. Cardiovasc. Med. 8:731440. doi: 10.3389/fcvm.2021.731440
Received: 27 June 2021; Accepted: 26 October 2021;
Published: 22 November 2021.
Edited by:
Daniel Morris, Charité Universitätsmedizin Berlin, GermanyReviewed by:
Francesco Sturla, IRCCS Policlinico San Donato, ItalyFilippo Cademartiri, Gabriele Monasterio Tuscany Foundation (CNR), Italy
Copyright © 2021 Yang, Wen, El-Hamamsy, Ni, Sun and Zhu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Tianyang Yang, eWFubmlreWFuZ3RpYW55YW5nQGhvdG1haWwuY29t; Dan Zhu, emh1ZGFuX21kQDEyNi5jb20=
†These authors have contributed equally to this work and share first authorship
 Tianyang Yang
Tianyang Yang Haini Wen
Haini Wen Ismail El-Hamamsy3
Ismail El-Hamamsy3