- 1Shandong University of Traditional Chinese Medicine, Jinan, China
- 2The Affiliated Hospital of Shandong University of Traditional Chinese Medicine, Jinan, China
Objective: This study aimed to evaluate the efficacy of acupuncture in improving ovulation rates in women with polycystic ovary syndrome (PCOS) and to identify optimal dosage parameters, including the number of acupoints, treatment frequency, and session duration, using integrated pairwise meta-analysis, network meta-analysis (NMA), and model-based dose-response modeling.
Methods: Nine databases were searched up to January 2025, yielding 43 randomized controlled trials (RCTs) involving 4,827 participants that compared acupuncture with sham acupuncture, pharmacotherapy, or conventional therapy control group. Pairwise meta-analysis, NMA, and model-based dose-response modeling were performed.
Results: Acupuncture alone significantly increased ovulation rates compared with sham acupuncture (RR = 1.15, 95% CI: 1.04-1.27) and pharmacotherapy (RR = 1.11, 95% CI: 1.04-1.20). Additionally, acupuncture combined with herbal medicine outperformed pharmacotherapy (RR = 1.27, 95% CI: 1.12-1.43). NMA ranked acupuncture combined with herbal medicine as the most effective intervention (SUCRA = 97.8%). Dose-response modeling identified the following optimal protocols: for acupuncture alone, 30 minutes per session, 29 acupoints, three sessions per week for 24 weeks; and for combined therapy, 19 minutes per session, 26 acupoints, four sessions per week for 24 weeks.
Conclusion: Acupuncture is an effective non-pharmacological intervention for PCOS-related ovulatory dysfunction, with its efficacy dependent on precise dosing parameters. These findings highlight the need for standardized protocols in future trials to validate dose-response thresholds and to optimize personalized treatment strategies.
Systematic review registration: www.crd.york.ac.uk, identifier PROSPERO (CRD420250651353).
1 Introduction
Polycystic ovary syndrome (PCOS), a complex endocrine disorder affecting 4-21% of reproductive-aged women globally, is diagnosed using the Rotterdam criteria, which require at least two of the following: hyperandrogenism (clinical or biochemical), irregular menstrual cycles, and polycystic ovarian morphology (1, 2). The disorder’s multifactorial etiology encompasses genetic polymorphisms (e.g., CYP gene family variants), environmental exposures to endocrine-disrupting chemicals [EDCs, including bisphenol A (BPA), dichlorodiphenyltrichloroethane (DDT), and mercury], high-calorie dietary intake, physical inactivity, and gut microbiota dysbiosis (3, 4). The triad of insulin resistance (IR), gonadotropin dysregulation, and chronic low-grade inflammation mechanistically drives PCOS pathogenesis. IR, affecting approximately 70% of patients, leads to compensatory hyperinsulinemia, which stimulates ovarian androgen secretion while suppressing hepatic sex hormone-binding globulin (SHBG) synthesis. Concurrently, aberrant lipid metabolism exacerbates IR, establishing a self-perpetuating cycle of metabolic dysfunction (5, 6). Gonadotropin dysregulation manifests as increased pulsatile secretion of gonadotropin-releasing hormone (GnRH), elevating the luteinizing hormone (LH)/follicle-stimulating hormone (FSH) ratio. This hormonal imbalance promotes excessive androgen production by ovarian theca cells, contributing to hyperandrogenemia (7). Finally, dysregulated immune cells and elevated inflammatory cytokines in serum and ovarian tissues induce systemic low-grade chronic inflammation (SLCI), which synergistically interacts with obesity, hyperandrogenemia, and IR to amplify metabolic and reproductive dysfunction (8). Collectively, these mechanisms perpetuate the heterogeneous clinical manifestations of PCOS.
Clinically, these pathophysiological disturbances manifest as obesity (present in 53-74% of cases) and hyperandrogenemia (in approximately 60% of cases). Furthermore, PCOS increases the risk of several comorbid conditions, including a 2.87-fold higher risk of type 2 diabetes (95% CI: 1.37-6.01), a 1.72-fold higher risk of hypertension (95% CI: 1.43-2.07), an approximately 10-fold higher risk of obstructive sleep apnea (95% CI: 3.90-23.26), and a 1.68-fold higher risk of cardiometabolic diseases (95% CI: 1.26-2.23) (9–11). Oral contraceptives are the most commonly prescribed therapy for PCOS. Clomiphene citrate, a non-steroidal selective estrogen receptor modulator (SERM), and letrozole, an aromatase inhibitor, are widely used to induce ovulation. Additionally, metformin is employed to ameliorate IR in patients with PCOS (12). However, due to adverse effects and poor adherence associated with oral pharmacotherapies (13–15), the exploration of alternative therapeutic approaches has become imperative.
Due to the limited efficacy and risks of conventional pharmacological treatments, non-pharmacological approaches like acupuncture have attracted growing interest for their potential to improve reproductive and metabolic dysfunction in PCOS via multimodal mechanisms (16). Rooted in traditional Chinese medicine (TCM) theory, acupuncture aims to restore balance to the “Kidney-Tiangui-Chongren” axis, a conceptual framework corresponding to dysregulation of the hypothalamic-pituitary-ovarian (HPO) axis in biomedical terms (17). Recent studies show that electroacupuncture downregulates ovarian Alas2 expression, modulates autophagy, and regulates the kisspeptin-GnRH/LH axis, thereby alleviating clinical symptoms of PCOS (18–20). However, some studies report nonsignificant therapeutic effects of acupuncture, likely due to methodological limitations in trial design, such as acupoint selection, treatment duration, control group allocation, and suboptimal statistical analyses (21).
While current evidence supports the therapeutic potential of acupuncture in PCOS, its dose-response relationships remain underexplored. Existing systematic reviews have predominantly focused on pairwise comparisons between acupuncture and single comparator interventions, without comprehensively assessing the efficacy hierarchy of acupuncture, pharmacological therapies, and their combined regimens in improving ovulation rates. To address these limitations, this study employs a dual analytical framework combining conventional pairwise meta-analysis with network meta-analysis and model-based dose-response modeling. This approach aims to hierarchically rank therapeutic efficacy and elucidate nonlinear relationships between key acupuncture dosage parameters (acupoint quantity, treatment frequency, and duration) and ovulation outcomes. The findings are expected to optimize acupuncture protocols by identifying thresholds for maximal benefit, thereby guiding clinical practice and informing randomized controlled (RCT) design for PCOS management.
2 Materials and methods
In accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, this study was designed to follow established protocols for systematic reviews and meta-analyses. The methodology complied with the PRISMA Statement for standard pairwise meta-analysis, while components related to the NMA were guided by the corresponding PRISMA extension. Both frameworks were implemented rigorously to ensure methodological consistency and strict adherence to contemporary academic standards. The study protocol was prospectively registered with PROSPERO (CRD420250651353).
2.1 Eligibility criteria and outcomes
This systematic review included studies comparing acupuncture with sham acupuncture, conventional Western medications (e.g., metformin, letrozole, clomiphene), or conventional therapy control groups in patients with PCOS. Conventional therapy control groups were defined as those receiving the same adjunct therapies alone, serving as comparators for interventions combining acupuncture with these therapies. Adjunct treatments included Western medications (e.g., metformin, letrozole, clomiphene) and conventional Chinese herbal medicine. Additionally, comparisons between acupuncture combined with herbal medicine and conventional Western drugs were conducted to explore the synergistic effects consistent with the TCM principle of integrating acupuncture and herbal therapy. Studies were included if they met the following criteria: (1) RCTs published in English or Chinese; (2) participants diagnosed with PCOS according to the 2003 Rotterdam consensus criteria (22); (3) intervention groups receiving acupuncture alone or combined with herbal medicine; (4) control groups receiving sham acupuncture or conventional pharmacological treatments; and (5) Reporting ovulation rate as a primary or secondary outcome measure.
The exclusion criteria were as follows: (1) studies involving participants with concurrent endocrine or reproductive disorders, including hyperprolactinemia, thyroid dysfunction, premature ovarian failure, androgen-secreting adrenal or ovarian tumors, hypothalamic or pituitary amenorrhea, or other conditions associated with hyperandrogenism, menstrual irregularities, or ovulatory dysfunction; (2) non-randomized studies, including reviews, animal or in vitro studies, conference abstracts, meta-analyses, retrospective analyses, case reports, or non-primary research; (3) studies with fewer than 20 participants; (4) duplicate publications; and (5) studies with incomplete abstract information or inaccessible raw data.
The primary outcome measure was the ovulation rate, calculated as follows: Ovulation Rate (%) = (Number of Ovulatory Cycles ÷ Total Observed Cycles) × 100%. Secondary outcomes included levels of sex hormones (FSH, LH, and testosterone) and body mass index (BMI).
2.2 Data sources and searches
Five English databases (i.e., PubMed, Web of Science, Embase, Cochrane Library, and Ovid Medline) and four Chinese databases (CNKI, WanFang, VIP, and CBM) from their inception to 17 January 2025 were searched. Relevant terminology related to acupuncture and PCOS was first identified through the MeSH database of the National Center for Biotechnology Information (NCBI) to establish standardized descriptors and entry terms. When multiple MeSH terms appeared, selections were made based on conceptual relevance to the study objectives. The search strategy was then refined across databases to enhance retrieval precision, using truncation to capture lexical variations and removing redundant terms to streamline the algorithm. This process aimed to improve methodological rigor and reproducibility (see Supplementary File 1).
2.3 Selection of studies and data extraction
Two reviewers (W.J.L. and Z.C.Y.) independently screened all abstracts and full-text articles, with discrepancies resolved through iterative consensus discussions. Data extraction was conducted using prespecified standardized forms to systematically capture authorship, publication year, trial design, participant characteristics (e.g., age, diagnostic criteria), intervention protocols, follow-up duration, and primary and secondary outcome measures. Persistent discrepancies during data extraction were adjudicated by a third reviewer (W.J.) to ensure methodological rigor.
2.4 Risk of bias and certainty of evidence
Two reviewers (W.J.L. and Z.C.Y.) independently assessed the risk of bias in the included trials. The updated version of the ROB 2.0 assessment tool recommended by the Cochrane Collaboration was used, which covers five domains: (1) randomization process, (2) deviations from intended interventions, (3) missing outcome data, (4) measurement of outcomes, and (5) selection of reported results (23) (see Supplementary File 2). The quality of evidence for the pairwise meta-analyses was assessed using the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) framework (24). Downgrading factors considered included risk of bias, inconsistency, indirectness, imprecision, and publication bias (25) (see Supplementary File 11). The Confidence in NMA (CINeMA) framework was applied to evaluate the certainty of evidence derived from the NMA (26). This framework assessed six methodological domains: (1) within-study bias, (2) reporting bias (including publication bias and selective outcome reporting), (3) applicability of evidence, (4) precision of estimates, (5) between-study heterogeneity, and (6) network inconsistency. CINeMA employed a three-tier classification system (low risk, some concerns, or high risk) to assess methodological rigor within each domain. These domain-specific evaluations were subsequently aggregated to derive a four-tier confidence categorization (high, moderate, low, or very low) for all comparative treatment effects (27).
2.5 Assessment of acupuncture dose
Dosage quantification was determined using four predefined criteria (28), with thresholds established based on the median distribution of parameters observed in the included studies, ensuring a balanced stratification for analysis: (1) number of acupoints per session: >13 acupoints (high dose) vs. ≤13 acupoints (low dose); (2) Deqi response: presence (high dose) vs. absence (low dose) of the characteristic needling sensation; (3) treatment frequency: >4 sessions/week (high dose) vs. ≤4 sessions/week (low dose); and (4) intervention duration: >12 weeks (high dose) vs. ≤12 weeks (low dose).A scoring system assigned +1 point for each high-dose criterion and -1 point for each low-dose criterion. Cumulative scores categorized interventions into three groups: (1) high dose: aggregate scores of +2 to +4; (2) moderate dose: scores of -1 to +1; and (3) low dose: aggregate scores of -4 to -2. Two independent reviewers evaluated each criterion, with unresolved discrepancies adjudicated by a third reviewer. To mitigate bias, reviewers were blinded to each other’s assessments throughout the process.
2.6 Data analysis
2.6.1 Pairwise meta-analysis
Meta-analyses for dichotomous outcomes (ovulation rate) were conducted using the “meta” package in R (version 4.4.2). Heterogeneity was assessed using the I2 statistic, with p < 0.05 or I2 > 50% indicating statistically significant heterogeneity. A random-effects model was selected when I2 < 50%; otherwise, a fixed-effects model was applied to ensure robustness. Sensitivity analysis was performed using the ‘metainf’ function with a leave-one-out approach to evaluate the stability of the results. Additionally, all pairwise comparisons required at least two studies for inclusion. Covariates including age, number of acupoints, acupuncture frequency, acupuncture type, retaining time, and acupuncture dose were incorporated into univariate meta-regression and subgroup analyses to explore their influence on outcomes. For analyses involving ≥10 studies, publication bias was assessed using Egger’s test and funnel plots. Symmetrical funnel plots with p > 0.05 were interpreted as indicating no significant publication bias (see Supplementary File 3).
2.6.2 NMA
NMA was conducted using the “netmeta” package in R (version 4.4.2). Heterogeneity was evaluated using the I2 statistic, with a random-effects model selected if p < 0.05 or I2 > 50%; otherwise, a fixed-effects model was applied. Given the complexity of evidence structures in NMA, inconsistency between direct and indirect evidence was assessed using the node-splitting method in addition to heterogeneity testing. This method evaluates the agreement between direct and indirect evidence at specific split nodes, with p-values > 0.05 indicating no significant inconsistency. Node-split analyses were performed to quantify inconsistency for each comparison. Interventions were ranked based on the surface under the cumulative ranking curve (SUCRA) to evaluate their efficacy in improving ovulation rates in patients with PCOS. Additionally, NMA meta-regression using the “gemtc” package examined the influence of covariates, including age, number of acupoints, acupuncture frequency, acupuncture type, treatment duration, acupuncture dose, and needle retention time, on outcomes and heterogeneity.
2.6.3 Exploratory acupuncture dose-response analysis
Dose-response analysis was conducted using the “MBNMAdose” package in R (version 4.4.2). This Bayesian dose-response modeling framework, termed Model-Based NMA for Dose-Response Relationships (MBNMA), integrates multiple dose levels into a unified structure by simulating diverse dose-response functions. By modeling dose-response relationships, this approach connects otherwise disconnected evidence networks and enhances the precision of treatment effect estimates (29).
To validate the consistency assumption of the model, a NMA model was fitted that does not assume consistency, exclusively modeling the direct relative effects between each treatment arm and the reference treatment within individual studies. The results of this NMA model were compared with those derived from the Unrelated Mean Effects (UME) model. A discrepancy between direct and indirect evidence within the network would indicate that the consistency assumption should be reconsidered (30).
The Deviance Information Criterion (DIC) values of spline functions, log-linear functions, and Emax functions, each modeled under both random-effects and fixed-effects frameworks, were compared to identify the optimal functional form balancing goodness-of-fit and model complexity. Covariates, including the number of acupoints, acupuncture frequency, and dose group classification, were incorporated into the analysis.
3 Results
3.1 Studies selection and characteristics of include trails
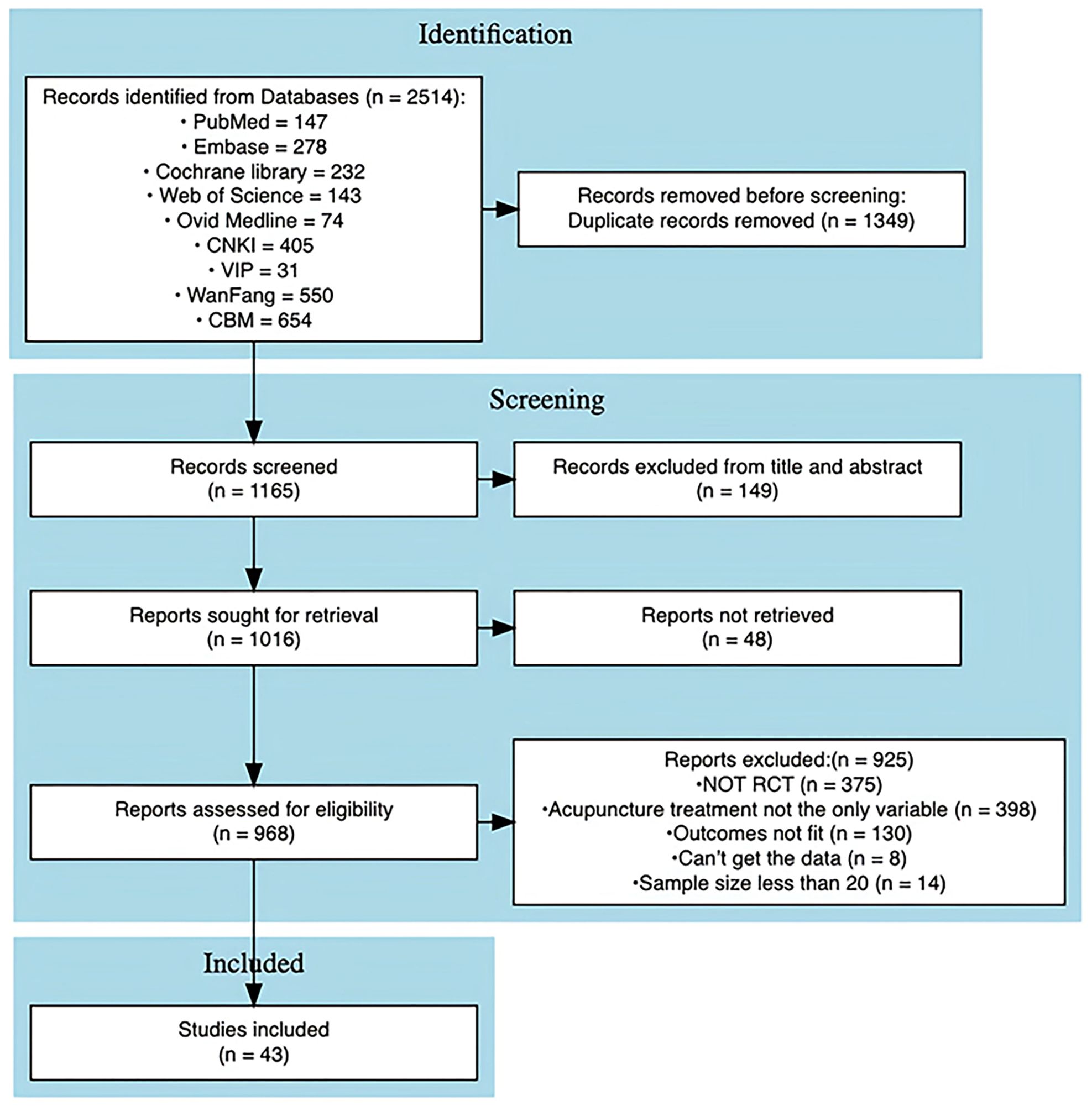
A total of 2,514 studies were identified through database searches. After removing 1,349 duplicate records, 149 studies were excluded following title and abstract screening. Of the remaining 1,016 studies, 925 were excluded after independent full-text review, and 48 studies (31–73) were unavailable for retrieval. Ultimately, 43 studies were included in the final analysis. Reasons for exclusion at each screening stage are detailed in Figure 1. The included studies were published between 2005 and 2024, with 40 studies (approximately 93.0%) published within the last decade (2015-2024), indicating the currency of the evidence. The mean age of participants across the included studies was 27.82 years. All studies reported ovulation rate as an outcome measure, with 7 studies reporting BMI, 25 reporting FSH, 30 reporting LH, and 14 reporting the LH/FSH ratio. Among the 35 studies that included conventional pharmacological treatments as a comparator group, the specific Western medications varied. Clomiphene citrate was the most frequently used medication, appearing in 19 studies (54.3%), either alone or in combination with other drugs. Letrozole was included in 7 studies (20.0%), Diane-35 (ethinyl estradiol/cyproterone acetate) in 8 studies (22.9%), and metformin in 6 studies (17.1%). Detailed characteristics of the included trials are provided in Supplementary File 4.
3.2 Risk of bias and certainty of evidence
Supplementary File 5 summarizes the risk of bias assessment for the included trials. Three studies (7%) were rated as “low risk” across all domains. Twelve studies (28%) raised “some concerns” in the randomization process due to insufficiently described allocation concealment, while 28 studies (65%) exhibited deficiencies in the blinding of participants and outcome assessors. One study was categorized as “some concerns” due to attrition rates between 10% and 20%. Four studies (9%) that were registered in clinical trial registries with publicly accessible protocols were classified as “low risk.” The CINeMA evaluation indicated high certainty of evidence for only a minority of comparisons (e.g., acupuncture plus medicine vs. sham acupuncture). Most comparisons were rated as very low certainty due to within-study bias, heterogeneity, or imprecision, highlighting the limited reliability of the existing evidence base. In the pairwise meta-analyses, the GRADE framework was employed to appraise the quality of evidence. The findings indicate that most evidence supporting ovulation rate outcomes was of high quality. However, moderate-quality evidence was noted in comparisons of acupuncture combined with Chinese herbal medicine versus conventional pharmaceuticals, and acupuncture versus routine treatment controls. These downgrades were primarily due to wide confidence intervals in the former and potential publication bias in the latter (Supplementary File 12). Conversely, when applying the CINeMA framework to assess evidence certainty in network meta-analyses, most comparisons were downgraded to very low certainty. This discrepancy reflects the fundamental differences between GRADE and CINeMA: GRADE focuses on the internal validity of direct comparisons, while CINeMA evaluates the certainty across the entire evidence network, incorporating both direct and indirect evidence. High heterogeneity, potential inconsistencies, and uncertainties in indirect comparison pathways were the primary factors contributing to the overall low certainty of network evidence, even when individual direct comparisons were of relatively high quality.
3.2.1 Acupuncture dose
The number of acupoints used across studies ranged from 2 to 29, with the majority (72%, 31 trials) employing 11–17 acupoints. All studies explicitly confirmed the presence of deqi. Acupuncture frequency varied from once weekly to once daily, with most trials (70%, 30 trials) administering either three or seven sessions per week. The intervention duration spanned 4 to 24 weeks, with the majority (72%, 31 trials) utilizing a 12-week protocol. Based on predefined criteria, the acupuncture dosages in the included studies were categorized as high dose (21%, 9 trials), moderate dose (58%, 25 trials), and low dose (21%, 9 trials).
3.3 Pairwise meta-analysis
The conventional pairwise meta-analysis comparing acupuncture and control demonstrated the following findings for ovulation rate: acupuncture was associated with a 1.15 times higher ovulation rate than sham acupuncture (RR = 1.15, 95% CI: 1.04-1.27); acupuncture showed a 1.11 times higher ovulation rate compared to medicine (RR = 1.11, 95% CI: 1.04-1.20); acupuncture combined with Chinese herbal medicine yielded a 1.27 times higher ovulation rate than Western medicine alone (RR = 1.27, 95% CI: 1.12-1.43); and acupuncture exhibited a 1.23 times higher ovulation rate compared to conventional therapy control groups (RR = 1.23, 95% CI: 1.17-1.30) (Figure 2). Acupuncture combined with Chinese medicine resulted in significantly greater reductions in LH levels relative to conventional Western medicine (SMD = 1.49, 95% CI: 0.73-2.25). Compared to conventional therapy control groups, acupuncture interventions demonstrated statistically significant improvements in the following outcomes: LH levels (SMD = 1.17, 95% CI: 0.71-1.62), LH/FSH ratio (SMD = 0.91, 95% CI: 0.08-1.74), T levels (SMD = 0.71, 95% CI: 0.31-1.10), and BMI (MD = 2.81, 95% CI: 1.65-3.97) (Supplementary File 6).
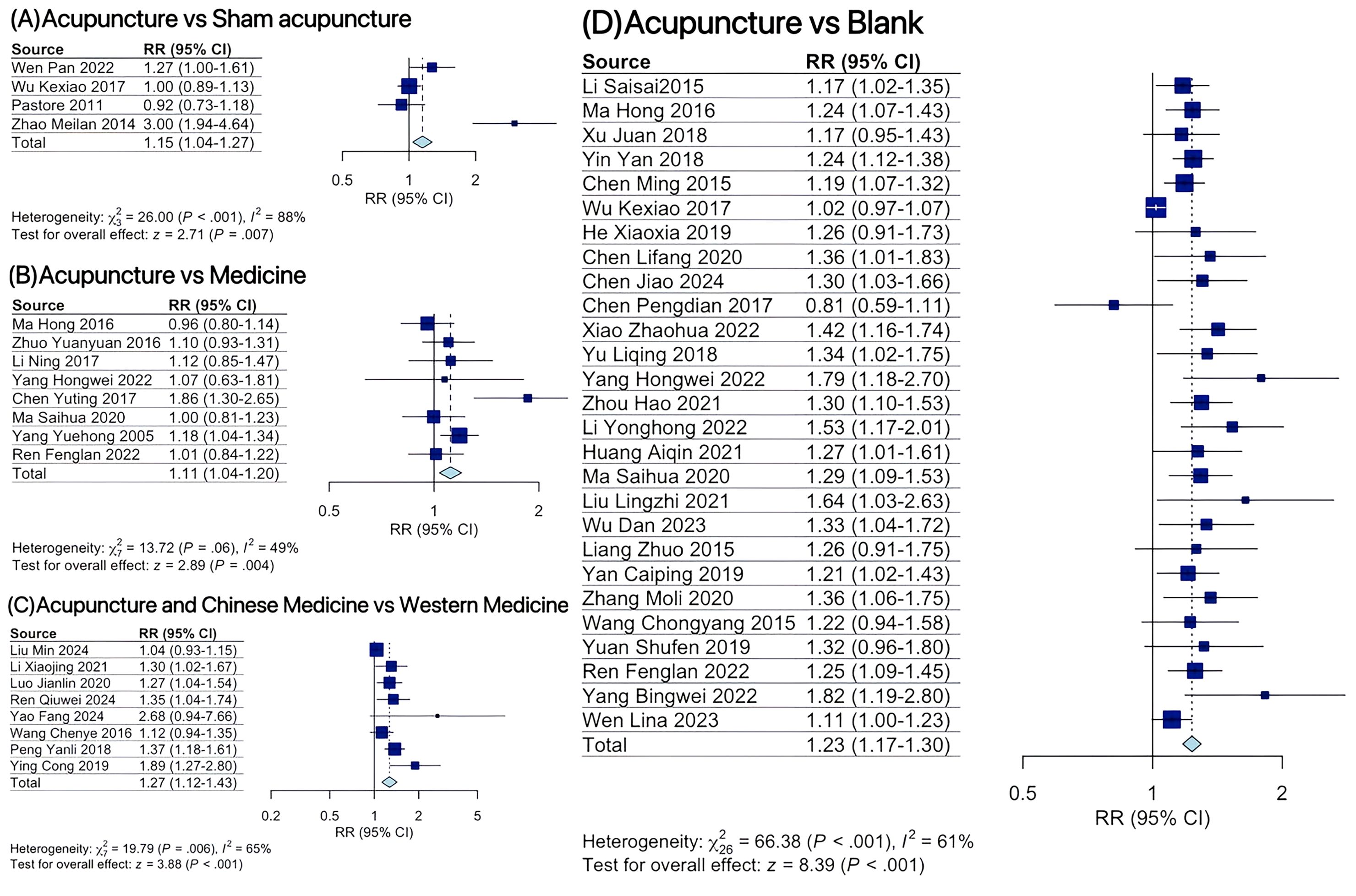
Figure 2. Forest plot for ovulation rate at the end of treatment. (A) Acupuncture versus sham acupuncture; (B) Acupuncture versus Medicine; (C) Acupuncture and Chinese Medicine versus Western Medicine; (D) Acupuncture versus blank.
Sensitivity analysis demonstrated that the direction and statistical significance of the pooled effect estimates remained consistent upon exclusion of any single study, indicating robust findings (Supplementary File 7).
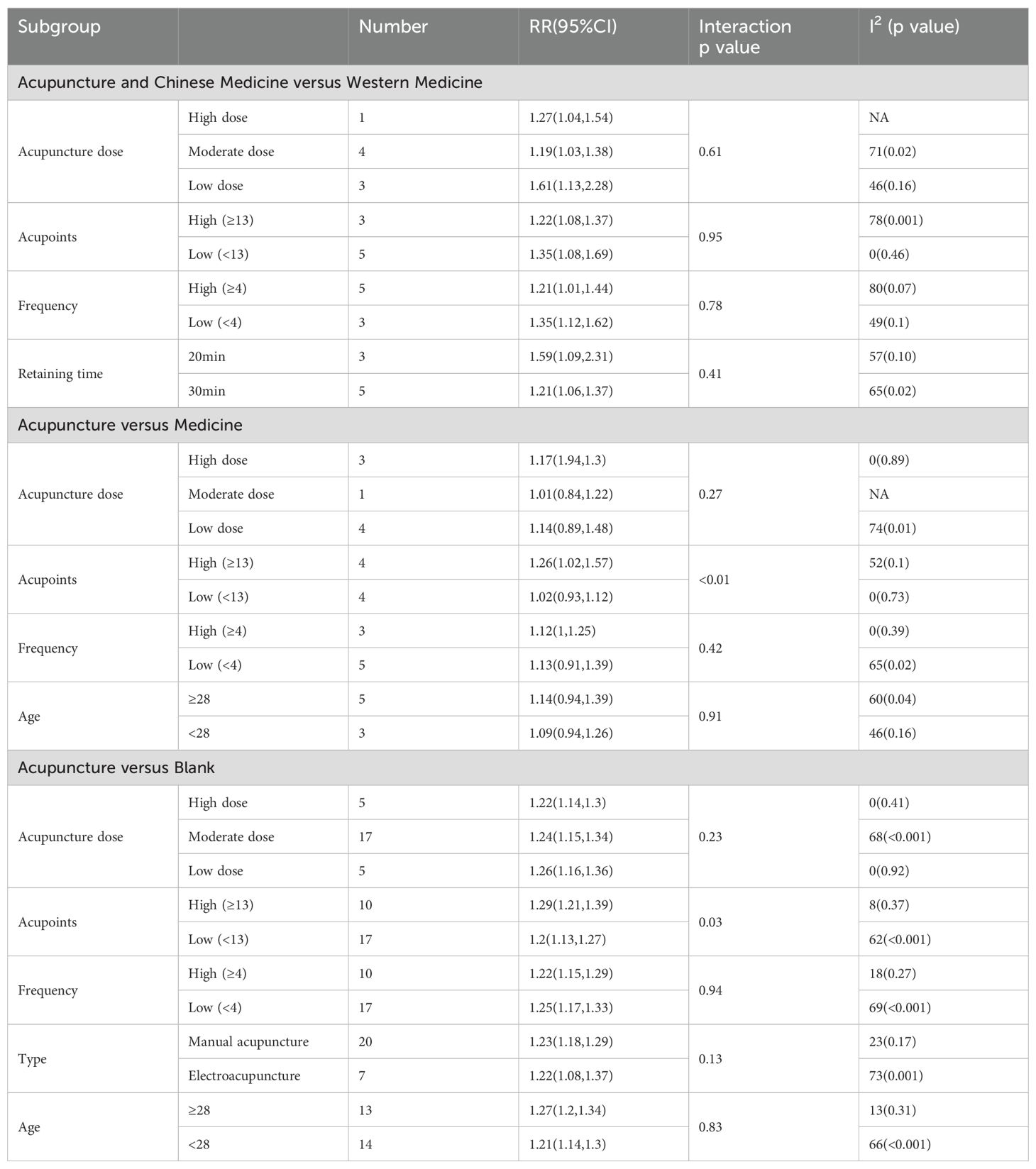
In most regression and subgroup analyses, covariates including mean patient age, number of acupoints, acupuncture frequency, acupuncture type, and needle retention time did not demonstrate statistically significant effects on outcomes (Table 1, Supplementary File 8). However, when compared to medicine, the high-acupoint group (RR = 1.26, 95% CI: 1.02-1.57) showed significantly greater improvement in ovulation rates among patients with PCOS than the low-acupoint group (RR = 1.02, 95% CI: 0.93-1.12; interaction p < 0.01). Similarly, compared to conventional therapy control groups, the high-acupoint group (RR = 1.29, 95% CI: 1.21-1.39) exhibited a more pronounced improvement in ovulation rates than the low-acupoint group (RR = 1.20, 95% CI: 1.13-1.27; interaction p = 0.03). These findings identify acupuncture dosage parameters, specifically the number of acupoints and acupuncture frequency, as principal contributors to heterogeneity. In comparisons of acupuncture combined with herbal medicine versus Western pharmacotherapy, high-acupoint protocols demonstrated substantial heterogeneity (I2 = 78%), whereas low-acupoint protocols exhibited negligible heterogeneity (I2 = 0%). For acupuncture versus pharmacotherapy alone, low-acupoint, high-dose, and high-frequency regimens showed minimal heterogeneity (I2 = 0%), while high-acupoint, low-dose, and low-frequency regimens displayed significant heterogeneity (I2 = 52%, 74%, and 65%, respectively). In comparisons of acupuncture versus conventional therapy control groups, moderate-dose regimens demonstrated intermediate heterogeneity (I2 = 68%), whereas high- and low-dose regimens were homogeneous (I2 = 0% for both).
3.4 NMA
The network diagram (Supplementary File 13) incorporated 43 studies evaluating 10 interventions. Node sizes are proportional to the number of included studies, while edge thickness corresponds to the number of trials providing direct comparisons between interventions.
Supplementary File 15 demonstrates that, compared to sham acupuncture, both acupuncture combined with medicine (RR = 1.45, 95% CI: 1.22-1.72) and acupuncture alone (RR = 1.21, 95% CI: 1.03-1.42) significantly improved ovulation rates in patients with PCOS. In contrast, interventions involving Chinese herbal medicine alone or pharmacotherapy (e.g., letrozole) did not reach statistical significance. SUCRA values (Supplementary File 9) indicated that acupuncture combined with medicine had the highest cumulative ranking probability (97.8%), followed by sham acupuncture combined with medicine (80.6%), suggesting that acupuncture combined with herbal medicine may represent the optimal intervention for improving ovulation rates.
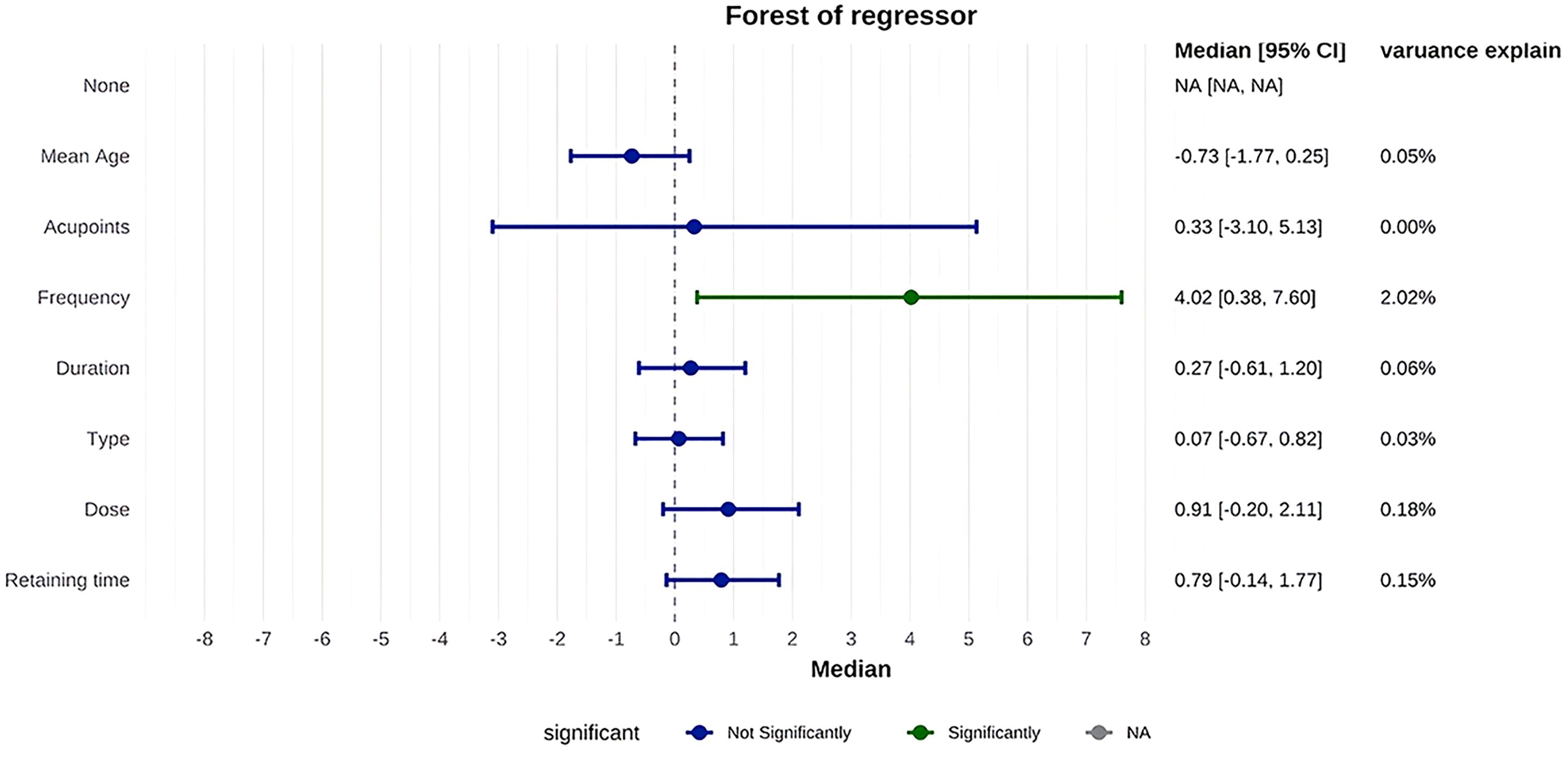
Network meta-regression identified acupuncture frequency and dosage as the primary sources of heterogeneity. Acupuncture frequency (β = 3.77, 95% CI: 1.52-6.21) and dosage (β = 1.41, 95% CI: 0.68-2.20) exhibited significant positive associations with ovulation rates, while mean age, number of acupoints, needle retention time, and total treatment duration showed no statistically significant effects (Figure 3).
3.5 Exploratory acupuncture dose-response
Our analysis identified distinct dose-response relationships for acupuncture monotherapy and acupuncture combined with medicine. Both modalities exhibited positive associations between acupoint quantity and treatment duration with ovulation rates. Notably, acupuncture frequency followed a W-shaped dose-response curve in monotherapy, whereas combination therapy demonstrated an inverted U-shaped relationship between treatment duration and efficacy. Regarding needle retention time, acupuncture exhibited a U-shaped dose-response curve, while combination therapy demonstrated an inverted U-shaped pattern. Maximal efficacy for acupoint quantity occurred at 29 acupoints in acupuncture (RR = 0.32, 95% CI: 0.21-0.45; SD = 0.06) and at 26 acupoints in combination therapy (RR = 0.64, 95% CI: 0.51-0.74; SD = 0.06). Similarly, both therapeutic regimens showed peak efficacy at 24 weeks (acupuncture: RR = 0.23, 95% CI: 0.04-0.55; SD = 0.14; combination therapy: RR = 0.42, 95% CI: 0.09-0.78; SD = 0.18). Optimal acupuncture frequency diverged between approaches, peaking at 3.3 sessions/week for acupuncture (RR = 0.33, 95% CI: 0.27-0.40; SD = 0.03) and at 4.1 sessions/week for combination therapy (RR = 0.51, 95% CI: 0.43-0.59; SD = 0.04). The optimal needle retention time was 30 minutes for acupuncture (RR = 0.24, 95% CI: 0.21-0.27; SD = 0.02) and 19 minutes for combination therapy (RR = 0.53, 95% CI: 0.41-0.65; SD = 0.06) (Figure 4, Supplementary File 10).
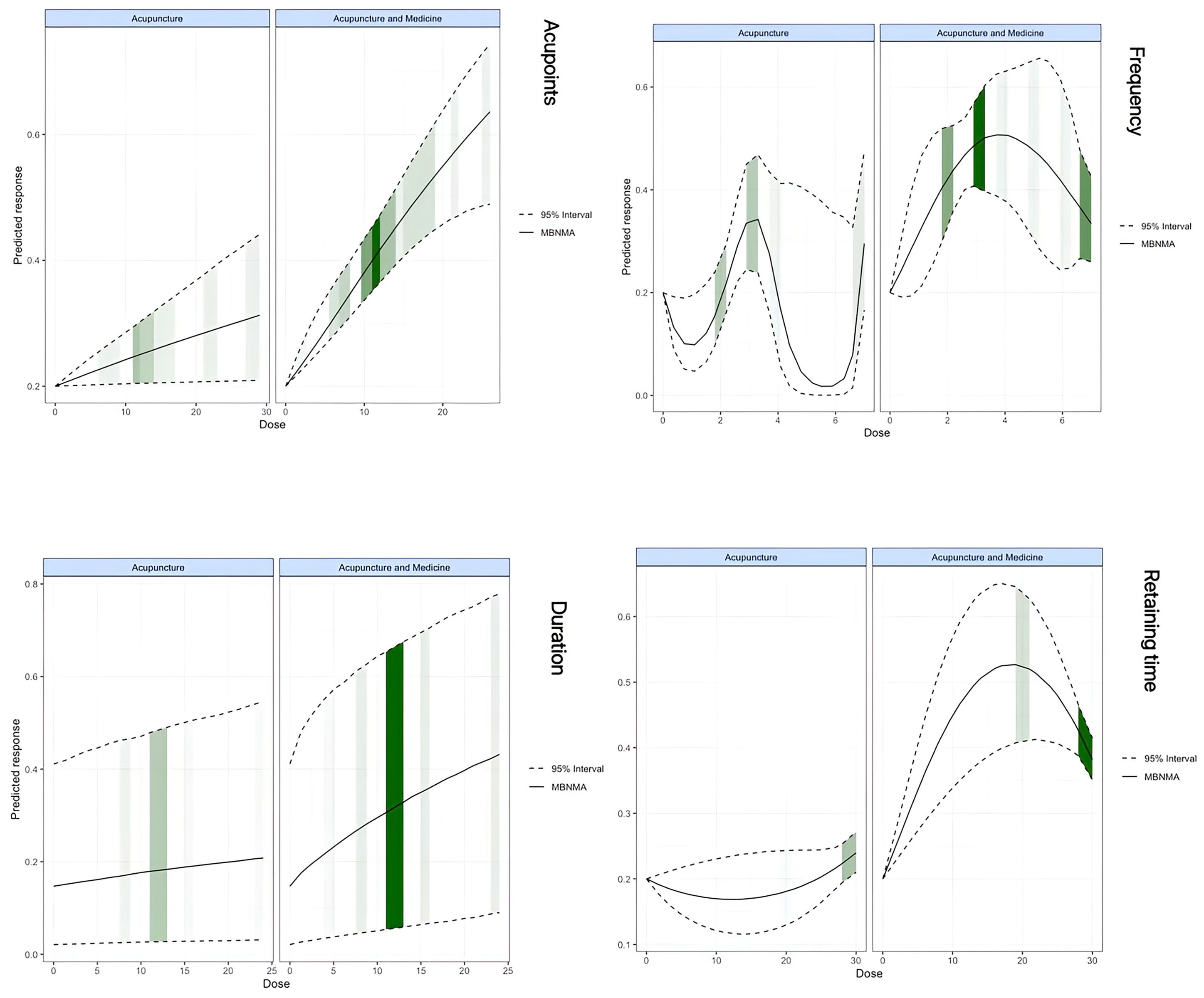
Figure 4. Dose-response relationships of acupuncture and acupuncture-medicine with acupoints, frequency, duration.
4 Discussion
4.1 Principal findings
To our knowledge, this is the most recent and comprehensive systematic review and meta-analysis evaluating the efficacy of acupuncture for treating PCOS while considering acupuncture type, dose, number of acupoints, frequency, and needle retention time. Our analysis included 43 RCTs involving 4,827 participants. The findings demonstrate that both acupuncture alone and acupuncture combined with pharmacotherapy significantly improve ovulation rates compared to pharmacotherapy alone or sham acupuncture, with both conventional meta-analysis and NMA confirming the superior efficacy of acupuncture-based interventions.
Specifically, acupuncture demonstrates superior efficacy in improving ovulation rates compared to sham acupuncture or pharmacotherapy alone in patients with PCOS, although it does not exhibit statistically significant effects on sex hormone levels or BMI. In contrast, compared to conventional therapy control groups, acupuncture not only enhances ovulation rates but also modulates sex hormone levels and BMI. Acupuncture combined with herbal medicine demonstrates superior efficacy to Western pharmacotherapy in improving ovulation rates and LH levels, with no significant effects on FSH levels or the LH/FSH ratio. Network meta-analysis further reveals that combining acupuncture with pharmacotherapy enhances ovulation outcomes compared to pharmacotherapy alone. Mechanistically, acupuncture may ameliorate ovulatory dysfunction by downregulating LncMEG3, inhibiting the PI3K/AKT/mTOR pathway, and reducing granulosa cell autophagy (74). Furthermore, acupuncture alleviates follicular arrest in PCOS rats by decreasing the overexpression of AMH and normalizing the imbalance between FSH and AMH in granulosa cells (75).
Subgroup analyses revealed that high-acupoint protocols (≥13 acupoints) demonstrated greater efficacy in improving ovulation rates compared to low-acupoint protocols (<13 acupoints) when compared against pharmacotherapy alone or conventional therapy control groups. Acupuncture dosage parameters, particularly acupoint number and stimulation frequency, emerged as primary sources of heterogeneity in pairwise meta-analyses. Network meta-regression confirmed these as key contributors, indicating a clear dose-response relationship between dosage variability and therapeutic outcomes. Exploratory analyses identified optimal efficacy with 30-minute needle retention and 29 acupoints per session, administered three times weekly for 24 weeks. In contrast, acupuncture combined with pharmacotherapy was most effective with 19-minute retention, 26 acupoints per session, and four sessions per week over the same duration. Acupuncture time-dose parameters, particularly frequency and needle retention duration, exhibit a threshold effect, highlighting the critical role of stimulus duration in modulating biochemical signaling. This effect promotes adequate neurochemical release and receptor activation, potentially influencing gene expression and cellular autophagy (20, 76).
4.2 Comparisons with previous studies
Over the past five years, two pairwise meta-analyses (77, 78) and one NMA (79) have examined the effects of acupuncture on PCOS. Wu Tianyu et al. (77) found that acupuncture combined with metformin significantly reduced LH levels, LH/FSH ratios, and T levels compared to metformin alone. Acupuncture combined with Chinese herbal medicine was more effective than metformin in reducing LH levels, consistent with findings comparing acupuncture to conventional therapy controls and acupuncture-herbal combinations to Western pharmacotherapy. However, the absence of sham acupuncture, herbal medicine alone, clomiphene, letrozole, and other comparators in this meta-analysis limits its utility for guiding decisions between acupuncture and alternative therapies. Unlike prior studies that focused mainly on acupoint frequency, this analysis highlights acupuncture dose, including acupoint number, stimulation frequency, and treatment duration, offering novel evidence to inform protocol standardization. Chen Xin et al. (78) reported that acupuncture combined with metformin improves ovulation rates more effectively than metformin alone, consistent with our results. Yang Lijie et al. (79) conducted an NMA identifying acupuncture combined with clomiphene as the most effective intervention for improving ovulation rates, which also corroborates our conclusions. Additionally, our exploratory dose-response analyses identified three sessions per week as the optimal acupuncture frequency for enhancing ovulation rates, aligning with Yang Lijie et al.’s findings supporting every-other-day administration.
4.3 Strengths and limitations
This study represents the most comprehensive evaluation to date of acupuncture’s therapeutic potential in PCOS, combining traditional pairwise meta-analysis, NMA, and exploratory dose-response modeling. Its methodological rigor and novel analytical framework offer several advantages. First, the integration of pairwise meta-analysis and NMA enables hierarchical ranking of therapeutic interventions by synthesizing direct and indirect evidence, enhancing result reliability while addressing gaps in prior reviews limited to pairwise comparisons. Second, MBNMA elucidates nonlinear relationships between key acupuncture dosage parameters (number of acupoints, frequency, duration) and ovulation outcomes, providing actionable thresholds for optimizing clinical protocols. Third, rigorous subgroup and meta-regression analyses identified acupuncture dosage as a critical source of heterogeneity, resolving inconsistencies in prior studies by demonstrating superior ovulation rates with high-acupoint protocols compared to low-acupoint regimens. Finally, the application of the CINeMA framework enhanced transparency in assessing evidence certainty.
However, this study has several limitations. First, although the exploratory dose-response modeling is innovative, its reliance on Bayesian assumptions may oversimplify the complex biological interactions between acupuncture parameters and physiological responses. Second, substantial heterogeneity across trials reduced the certainty of evidence to low or very low for many comparisons. Third, the acupuncture dosage scoring tool, a non-weighted composite of acupoint quantity, deqi response, treatment frequency, and duration, is based on Chinese clinical standards, which may limit its applicability to studies conducted in other regions. Fourth, comparisons involving acupuncture combined with herbal medicine versus Western medicine complicate the attribution of efficacy solely to acupuncture. While these results support the benefits of integrated Traditional Chinese Medicine approaches, they reflect synergistic or additive effects rather than acupuncture’s isolated impact, potentially overestimating its standalone efficacy. Lastly, the limited number of included trials precluded funnel plot analysis, restricting the ability to assess publication bias comprehensively.
4.4 Perspectives
Current research has rarely focused on optimizing acupuncture protocols and parameter selection, including acupuncture type and dosage. Although our subgroup analyses revealed no differences between electroacupuncture and manual acupuncture based on interaction p-values, acupuncture dose and its key parameters demonstrated significant impacts on treatment outcomes. Therefore, future RCTs should place greater emphasis on standardizing acupuncture dosage. Additionally, retention time is a critical yet long-overlooked parameter. While most current RCTs default to a 30-minute retention period, our subgroup analyses demonstrated higher effect sizes for ovulation rate improvement in the 20-minute retention group. Future studies should design head-to-head RCTs (e.g., 20 vs. 30 vs. 45 minutes) incorporating functional near-infrared spectroscopy (fNIRS) to dynamically monitor ovarian hemodynamic changes and define optimal retention windows. Concurrently, the interaction between retention time and other parameters warrants exploration. For example, it remains unclear whether high acupoint counts combined with shorter retention times yield synergistic effects or whether specific acupoints (e.g., Zigong [CV4], Sanyinjiao [SP6]) require differential retention durations for maximal efficacy. Furthermore, clinical RCTs should prioritize rigorous blinding implementation. Although the procedural nature of acupuncture poses challenges for practitioner blinding, blinding of participants and outcome assessors should be consistently enforced to minimize bias and enhance methodological rigor.
5 Conclusion
Acupuncture serves as an effective non-pharmacological intervention for addressing PCOS-related ovulatory dysfunction. Furthermore, this study identifies acupuncture dosage and parameter selection as critical determinants of therapeutic efficacy and predicts their optimal thresholds. Future research should prioritize high-quality, dose-finding RCTs to validate these dose-response thresholds and refine personalized therapeutic algorithms.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Author contributions
JLW: Writing – original draft, Software, Formal analysis, Methodology. ZS: Writing – original draft, Visualization. CZ: Writing – original draft, Validation. CX: Data curation, Writing – original draft. HB: Project administration, Writing – original draft. JY: Writing – original draft. JW: Resources, Conceptualization, Writing – review & editing, Funding acquisition.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. This work was supported by the Capital Health Research and Development of Special Fund (2024-1-4371), entitled “Effect evaluation of acupuncture for polycystic ovary syndrome”.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fendo.2025.1610338/full#supplementary-material
References
1. Lizneva D, Suturina L, Walker W, Brakta S, Gavrilova-Jordan L, and Azziz R. Criteria, prevalence, and phenotypes of polycystic ovary syndrome. Fertil Steril. (2016) 106:6–15. doi: 10.1016/j.fertnstert.2016.05.003
2. Joham AE, Norman RJ, Stener-Victorin E, Legro RS, Franks S, Moran LJ, et al. Polycystic ovary syndrome. Lancet Diabetes Endocrinol. (2022) 10:668–80. doi: 10.1016/S2213-8587(22)00163-2
3. Dilliyappan S, Kumar AS, Venkatesalu S, Palaniyandi T, Baskar G, Sivaji A, et al. Polycystic ovary syndrome: Recent research and therapeutic advancements. Life Sci. (2024) 359:123221. doi: 10.1016/j.lfs.2024.123221
4. Wu J, Wang K, Qi X, Zhou S, Zhao S, Lu M, et al. The intestinal fungus Aspergillus tubingensis promotes polycystic ovary syndrome through a secondary metabolite. Cell Host Microbe. (2025) 33:119–136.e11. doi: 10.1016/j.chom.2024.12.006
5. Goodarzi MO, Dumesic DA, Chazenbalk G, and Azziz R. Polycystic ovary syndrome: etiology, pathogenesis and diagnosis. Nat Rev Endocrinol. (2011) 7:219–31. doi: 10.1038/nrendo.2010.217
6. Diamanti-Kandarakis E and Dunaif A. Insulin resistance and the polycystic ovary syndrome revisited: an update on mechanisms and implications. Endocr Rev. (2012) 33:981–1030. doi: 10.1210/er.2011-1034
7. Helvaci N and Yildiz BO. Polycystic ovary syndrome as a metabolic disease. Nat Rev Endocrinol. (2025) 21:230–44. doi: 10.1038/s41574-024-01057-w
8. Deng H, Chen Y, Xing J, Zhang N, and Xu L. Systematic low-grade chronic inflammation and intrinsic mechanisms in polycystic ovary syndrome. Front Immunol. (2024) 15:1470283. doi: 10.3389/fimmu.2024.1470283
9. Amisi CA. Markers of insulin resistance in polycystic ovary syndrome women: An update. World J Diabetes. (2022) 13:129–49. doi: 10.4239/wjd.v13.i3.129
10. Tay CT, Mousa A, Vyas A, Pattuwage L, Tehrani FR, and Teede H. 2023 International evidence-Based polycystic ovary syndrome guideline update: insights from a systematic review and meta-Analysis on elevated clinical cardiovascular disease in polycystic ovary syndrome. J Am Heart Assoc. (2024) 13:e033572. doi: 10.1161/JAHA.123.033572
11. Dason ES, Koshkina O, Chan C, and Sobel M. Diagnosis and management of polycystic ovarian syndrome. CMAJ. (2024) 196:E85–94. doi: 10.1503/cmaj.231251
12. Stańczak NA, Grywalska E, and Dudzińska E. The latest reports and treatment methods on polycystic ovary syndrome. Ann Med. (2024) 56:2357737. doi: 10.1080/07853890.2024.2357737
13. Tsiami AP, Goulis DG, Sotiriadis AI, and Kolibianakis EM. Higher ovulation rate with letrozole as compared with clomiphene citrate in infertile women with polycystic ovary syndrome: A systematic review and meta-analysis. Horm (athens Greece). (2021) 20:449–61. doi: 10.1007/s42000-021-00289-z
14. Pundir J, Achilli C, Bhide P, Sabatini L, Legro RS, Rombauts L, et al. Risk of foetal harm with letrozole use in fertility treatment: A systematic review and meta-analysis. Hum Reprod Update. (2021) 27:474–85. doi: 10.1093/humupd/dmaa055
15. Notaro ALG and Neto FTL. The use of metformin in women with polycystic ovary syndrome: An updated review. J Assist Reprod Genet. (2022) 39:573–9. doi: 10.1007/s10815-022-02429-9
16. Pundir J, Charles D, Sabatini L, Hiam D, Jitpiriyaroj S, Teede H, et al. Overview of systematic reviews of non-pharmacological interventions in women with polycystic ovary syndrome. Hum Reprod Update. (2019) 25:243–56. doi: 10.1093/humupd/dmy045
17. Zhou T, Wang F, Xu X, Zhu Y, Zhang R, Lee HW, et al. Non-pharmacological interventions of traditional Chinese medicine in treating polycystic ovary syndrome: A group consensus. Integr Med Res. (2024) 13:101093. doi: 10.1016/j.imr.2024.101093
18. Peng Y, Guo L, Gu A, Shi B, Ren Y, Cong J, et al. Electroacupuncture alleviates polycystic ovary syndrome-like symptoms through improving insulin resistance, mitochondrial dysfunction, and endoplasmic reticulum stress via enhancing autophagy in rats. Mol Med. (2020) 26:73. doi: 10.1186/s10020-020-00198-8
19. Cong J, Li M, Wang Y, Ma H, Yang X, Gao J, et al. Protective effects of electroacupuncture on polycystic ovary syndrome in rats: Down-regulating Alas2 to inhibit apoptosis, oxidative stress, and mitochondrial dysfunction in ovarian granulosa cells. Tissue Cell. (2023) 82:102090. doi: 10.1016/j.tice.2023.102090
20. Xu G, Zhao X, Li Z, Hu J, Li X, Li J, et al. Effects of electroacupuncture on the kisspeptin-gonadotropin-releasing hormone (GnRH) /luteinizing hormone (LH) neural circuit abnormalities and androgen receptor expression of kisspeptin/neurokinin B/dynorphin neurons in PCOS rats. J Ovarian Res. (2023) 16:15. doi: 10.1186/s13048-022-01078-x
21. Gang W-J and Jing X-H. Is acupuncture actually not effective for polycystic ovary syndrome?: Analysis of the trial published in jama. Chin J Integr Med. (2017) 23:883–6. doi: 10.1007/s11655-017-2831-z
22. Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod (oxf Engl). (2004) 19:41–7. doi: 10.1093/humrep/deh098
23. Cumpston M, Li T, Page MJ, Chandler J, Welch VA, Higgins JP, et al. Updated guidance for trusted systematic reviews: A new edition of the cochrane handbook for systematic reviews of interventions. Cochrane Database Syst Rev. (2019) 10:ED000142. doi: 10.1002/14651858.ED000142
24. Higgins JPT, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, et al. The cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ. (2011) 343:d5928. doi: 10.1136/bmj.d5928
25. Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ. (2008) 336:924–6. doi: 10.1136/bmj.39489.470347.AD
26. A GRADE working group approach for rating the quality of treatment effect estimates from network meta-analysis. BMJ (clin Res Ed). (2015) 350:h3326. doi: 10.1136/bmj.h3326
27. Papakonstantinou T, Nikolakopoulou A, Higgins JPT, Egger M, and Salanti G. CINeMA: Software for semiautomated assessment of the confidence in the results of network meta-analysis. Campbell Syst Rev. (2020) 16:e1080. doi: 10.1002/cl2.1080
28. Li YT, Li CL, Yang H, Huang L, Liu JJ, Zheng XY, et al. Correlation between acupuncture dose and pregnancy outcomes in women with polycystic ovary syndrome undergoing in vitro fertilization-embryo transfer: a systematic review. BMC Complement Med Ther. (2024) 24:407. doi: 10.1186/s12906-024-04695-9
29. Mawdsley D, Bennetts M, Dias S, Boucher M, and Welton N. Model-based network meta-analysis: A framework for evidence synthesis of clinical trial data. CPT: Pharmacometrics Syst Pharmacol. (2016) 5:393–401. doi: 10.1002/psp4.12091
30. Dias S, Welton NJ, Sutton AJ, Caldwell DM, Lu G, and Ades AE. Evidence synthesis for decision making 4: Inconsistency in networks of evidence based on randomized controlled trials. Med Decis Mak: Int J Soc Med Decis Mak. (2013) 33:641–56. doi: 10.1177/0272989X12455847
31. Peng YL, Shen Y, Luo L, and Hu YB. Effect of Bushen Huoxue recipe combined with electroacupuncture on endocrine hormone and pregnancy outcome in infertile patients with polycystic ovary syndrome. Guangxi Med J. (2018) 40:795–8.
32. He X, Fang Y, and Liu L. Randomized controlled study on the effect of different treatment regimens on endometrial receptivity in infertile patients with polycystic ovary syndrome. J Li-shizhen Traditional Chin Med. (2019) 30:2424–6.
33. Wang CH, Sun X, Ding C, and Shen Y. Effect of cangfu daotan decoction combined with acupuncture on glucose and lipid metabolism and ovulation rate in patients with obese polycystic ovary syndrome. Modern J Integrated Traditional Chin Western Med. (2016) 25:4056–8.
34. Yao F. Clinical observation of cangfu daotan pill combined with acupuncture in the treatment of polycystic ovary syndrome with spleen deficiency and phlegm dampness. J Pract Traditional Chin Med. (2024) 40:623–6.
35. Yu L, Cao L, Xie J, and Shi Y. Therapeutic effects on ovulation and reproduction promotion with acupuncture and clomiphene in polycystic ovary syndrome. Chin Acupuncture& Moxibustion. (2018) 38:263–8. doi: 10.13703/j.0255-2930.2018.03.009
36. Zhao M and Liang R. Randomized single-blind clinical controlled study of electroacupuncture for ovulation induction in polycystic ovary syndrome. Pract Clin J Integrated Traditional Chin Western Med. (2014) 14:66–8. doi: 10.13638/j.issn.1671-4040.2014.08.045
37. Zhuo Y, Wu J, Lin W, Pi M, Chen P, and Yang Z. The “regulatingconception-governorvessel”acupuncturemethodforinfertilityofpolycysticovarian syndrome. Chin Acupuncture& Moxibustion. (2016) 36:1237–41. doi: 10.13703/j.0255-2930.2016.12.002
38. Zhou H and Shao S. Clinical study on acupuncture method of regulating conception vessel and governor vessel combined with dingkun pills for polycystic ovarian disease syndrome with infertility. New Chin Med. (2021) 53:106–11. doi: 10.13457/j.cnki.jncm.2021.02.027
39. Li Y, Ke Y, Liu Y, Feng T, Li Y, Li H, et al. Efficacy of regulating ren meridian and dredging du meridian acupuncture combined with letrozole in the treatment of infertility caused by polycystic ovary syndrome with kidney-yang deficiency type and its effect on lipid metabolism and sex hormone levels. J Liaoning Univ Traditional Chin Med. (2022) 24:171–5. doi: 10.13194/j.issn.1673-842x.2022.07.038
40. Lin W, Pi M, Zhuo Y, and Yang Z. Observation on the effect of tiaoren tongdu acupuncture in the treatment of polycystic ovary syndrome infertility of kidney yang deficiency type. Guiding J Traditional Chin Med Pharm. (2018) 24:80–2. doi: 10.13862/j.cnki.cn43-1446/r.2018.04.029
41. Ma H, Quan X, Chen X, and Dong Y. Flyingneedlingtherapycombined withclomipheneforovulationfailureinpolycysticovarysyndrome:arandomizedcontrolledtrial. Chin Acupuncture Moxibustion. (2016) 36:1161–5. doi: 10.13703/j.0255-2930.2016.11.015
42. Chen P, Yang Z, Liu F, Yan B, and Qiu T. Clinical observation of Clomiphene Citrate Tablets plus conception and governor vessels acupuncture in the treatment for ovulation of polycystic ovarian syndrome. China Med Herald. (2017) 14:91–4.
43. Chen J, Fan H, Zeng J, Liu Y, Hu J, Zhou M, et al. Clinical efficacy observation of electroacupuncture cycle therapy combined with clomiphene citrate treatment based on the theory of ‘Ren zhu Baotai’ on polycystic ovary syndrome. China J Traditional Chin Med Pharm. (2024) 39:496–9.
44. Chen L. Efficacy of oral ethinyl estradiol cyproterone tablets combined with acupuncture in the treatment of polycystic ovary syndrome complicated with infertility. Contemp Med Symposium. (2020) 18:93–4.
45. Huang A. Effect of Lai ’s Tongyuan acupuncture combined with conventional western medicine on infertility patients with polycystic ovary syndrome. Med J Chin People’s Health. (2021) 33:91–3.
46. Wen L, Wang M, and Zhou Y. Effect of longsha open and close pivot needle method combined with Letrozole in the treatment of infertility with polycystic ovarian syndrome. Guiding J Traditional Chin Med Pharm. (2023) 20:117–20. doi: 10.20047/j.issn1673-7210.2023.30.24
47. Chen Y, Li Y, Luo J, Chen Y, and Zheng S. Clinical observation of tong yuan needling method plus letrozol and HCG for refractory sterility caused by polycystic ovary syndrome. Shanghai J Acupuncture Moxibustion. (2017) 36:692–6. doi: 10.13460/j.issn.1005-0957.2017.06.0692
48. Ma S, Dou Z, Song J, Liu S, Tian X, and Fu Y. Clinical observation of mind-refreshing and orifice-opening needling combined with western medication for polycystic ovary syndrome of kidney deficiency and liver depression syndrome. Shanghai J Acupuncture Moxibustion. (2020) 39:1128–32. doi: 10.13460/j.issn.1005-0957.2020.09.1128
49. Yang H, Zhao X, Wei M, and Liu X. Clinical study on yishen huatan prescription combined with acupuncture for infertility due to polycystic ovary syndrome of kidney deficiency with phlegm-dampness type. New Chin Med. (2022) 54:153–7. doi: 10.13457/j.cnki.jncm.2022.17.032
50. Liu L, Chen C, and He S. Effect of acupuncture on ovarian function and sex hormone levels in patients with polycystic ovary syndrome. Lab Med Clinic. (2021) 18:1596–8.
51. Xu J and Zuo Y. Efficacy of acupuncture as adjunctive treatment on infertility patients with polycystic ovary syndrome. Chin Acupuncture& Moxibustion. (2018) 38:358–361, 363. doi: 10.13703/j.0255-2930.2018.04.004
52. Ying C. Clinical observation on acupuncture combined with shugan qutan sanyu decoction in the treatment of infertility caused by polycystic ovary syndrome. J Pract Traditional Chin Med. (2019) 35:914–5.
53. Liang Z. Effect of acupuncture combined with promoting ovulation treatment on pregnancy and ovulation rate of infecundity women with polycystic ovary syndrome of phlegm wet type. Modern J Integrated Traditional Chin Western Med. (2015) 24:1498–500, 1509.
54. Yan C, Fan C, Yan H, Zhang L, Zhong Y, and Zhou X. Clinical study of acupuncture combined with clomiphene to induce ovulation on follicular development and endometrium of patients with polycystic ovary syndrome. New Chin Med. (2019) 51:204–6. doi: 10.13457/j.cnki.jncm.2019.02.062
55. Zhang M, Wang X, Shan Z, and Dong B. Effects of acupuncture combined with western medication on endometrial thickness, ovulation rate and clinical pregnancy rate in infertile polycystic ovary syndrome patients. Shanghai J Acupuncture Moxibustion. (2020) 39:1133–7. doi: 10.13460/j.issn.1005-0957.2020.09.1133
56. Wang C. Clinical observation of acupuncture combined with clomiphene in the treatment of polycystic ovary syndrome. Asia-Pacific Traditional Med. (2015) 11:83–5.
57. Chen M, Zhang H, and Zhang Y. Clinical observation of acupuncture plus medication for obesity-type polycystic ovary syndrome. Shanghai J Acupuncture Moxibustion. (2015) 34:310–3. doi: 10.13460/j.issn.1005-0957.2015.04.0310
58. Wu D, Luo J, Pang Z, and You Z. Clinical effect of acupuncture on infertility of polycystic ovary syndrome with kidney deficiency and liver depression. China Modern Med. (2023) 30:126–30.
59. Yang Y, Hong J, Wei D, Chen X, and Lin J. Acupuncture on polycystic ovarian syndrome-induced infertility. J Guangdong Med Coll. (2005), 377–8.
60. Li S. Metformin and auxiliary acupuncture in the treatment of obese women infertility with polycystic ovary syndrome for 75 cases. Chin Med Modern Distance Educ China. (2015) 13:78–9.
61. Yuan S, Ye Y, and Wang T. Effect of acupuncture plus medication on ovary function in polycystic ovary syndrome due to kidney deficiency and blood stagnation. Shanghai J Acupuncture Moxibustion. (2019) 38:286–9. doi: 10.13460/j.issn.1005-0957.2019.03.0286
62. Ren F, Yang L, and Liu Y. Efficacy observation of acupuncture combined with medication for infertility in polycystic ovary syndrome of kidney deficiency and liver depression pattern. Shanghai J Acupuncture Moxibustion. (2022) 41:43–9. doi: 10.13460/j.issn.1005-0957.2022.01.0043
63. Yang B, Shi H, Zhou Y, Lu Q, and Yin X. Clinical observation on efficacy of acupuncture combined with traditional Chinese medicine for treating polycystic ovary syndrome patients with kidney Yang deficiency syndrome. Acad J Shanghai Univ Traditional Chin Med. (2022) 36:88–92. doi: 10.16306/j.1008-861x.2022.S1.022
64. Yin Y, Zhang Y, Zhang H, Jiang D, and Guo G. Clinical therapeutic effects of acupuncture combined with Chinese herbal medicine on infertility of polycystic ovary syndrome in the patients with ovulation induction with letrozole. Chin Acupuncture& Moxibustion. (2018) 38:27–32. doi: 10.13703/j.0255-2930.2018.01.006
65. Liu M. Observation on the curative effect of traditional Chinese medicine cycle adjustment method combined with acupuncture in the treatment of polycystic ovary syndrome with ovulation disorder infertility. J Pract Traditional Chin Med. (2024) 40:420–2.
66. Luo J. Clinical study on traditional Chinese medicine combined with acupuncture in the treatment of infertility with polycystic ovary syndrome. Friends Health. (2020) 46.
67. Xiao Z, Zhong H, and Zheng D. Clinical study of Ziyin Bushen Decoction combined with acupuncture therapy on patients with polycystic ovary syndrome of kidney yin deficiency type. Guangming J Chin Med. (2022) 37:4284–7.
68. Ren Q, Sun J, Cui S, and Chen D. Effects of Wuzi Xiezhuo decoction combined with acupuncture on glucose and lipid metabolism and ovulation rate in obese polycystic ovary syndrome. Chin Arch Traditional Chin Med. (2024), 1–7.
69. Li N and Zhang Y. Observation on the curative effect of Ziwuliuzhu acupoint opening method in the treatment of ovulation disorder of polycystic ovary syndrome. Beijing J Traditional Chin Med. (2017) 36:74–6. doi: 10.16025/j.1674-1307.2017.01.022
70. Li X, Zhang H, Zhang Z, and Guo B. Clinical observation of zuogui drink and huanglianEjiao decoction combined with acupuncture in treatment of infertility associated with polycystic ovary syndrome. J Guangzhou Univ Traditional Chin Med. (2021) 38:2145–51. doi: 10.13359/j.cnki.gzxbtcm.2021.10.017
71. Pan W, Li F, Wang Q, Huang Z, Yan Y, Zhao L, et al. A randomized sham-controlled trial of manual acupuncture for infertile women with polycystic ovary syndrome. Integr Med Res. (2022) 11:100830. doi: 10.1016/j.imr.2021.100830
72. Wu X-K, Stener-Victorin E, Kuang H-Y, Ma H-L, Gao J-S, Xie L-Z, et al. Effect of acupuncture and clomiphene in Chinese women with polycystic ovary syndrome. JAMA. (2017) 317:2502–14. doi: 10.1001/jama.2017.7217
73. Pastore LM, Williams CD, Jenkins J, and Patrie JT. True and sham acupuncture produced similar frequency of ovulation and improved LH to FSH ratios in women with polycystic ovary syndrome. J Clin Endocrinol Metab. (2011) 96:3143–50. doi: 10.1210/jc.2011-1126
74. Chen X, He H, Long B, Wei B, Yang P, Huang X, et al. Acupuncture regulates the apoptosis of ovarian granulosa cells in polycystic ovarian syndrome-related abnormal follicular development through LncMEG3-mediated inhibition of miR-21-3p. Biol Res. (2023) 56:31. doi: 10.1186/s40659-023-00441-6
75. Shi Y, Li L, Zhou J, Sun J, Chen L, Zhao J, et al. Efficacy of electroacupuncture in regulating the imbalance of AMH and FSH to improve follicle development and hyperandrogenism in PCOS rats. BioMed Pharmacother = BioMed Pharmacother. (2019) 113:108687. doi: 10.1016/j.biopha.2019.108687
76. Stener-Victorin E, Lundeberg T, Waldenström U, Manni L, Aloe L, Gunnarsson S, et al. Effects of electro-acupuncture on nerve growth factor and ovarian morphology in rats with experimentally induced polycystic ovaries. Biol Reprod. (2000) 63:1497–503. doi: 10.1095/biolreprod63.5.1497
77. Wu T, Liu Y, Kong F, Hu J, Liu Y, Yang J, et al. Improvement of endocrine and metabolic conditions in patients with polycystic ovary syndrome through acupuncture and its combined therapies: A systematic review and meta-analysis. Ann Med. (2025) 57:2477295. doi: 10.1080/07853890.2025.2477295
78. Chen X, Lan Y, Yang L, Liu Y, Li H, Zhu X, et al. Acupuncture combined with metformin versus metformin alone to improve pregnancy rate in polycystic ovary syndrome: A systematic review and meta-analysis. Front Endocrinol (Lausanne). (2022) 13:978280. doi: 10.3389/fendo.2022.978280
Keywords: acupuncture, polycystic ovary syndrome, ovulation, meta-analysis, dose-response analysis
Citation: Wei J, Shen Z, Zhao C, Xie C, Bai H, Yin J and Wang J (2025) Dose-response of acupuncture on ovulation rates in polycystic ovary syndrome: a meta-analysis and exploratory dose-response analysis. Front. Endocrinol. 16:1610338. doi: 10.3389/fendo.2025.1610338
Received: 11 April 2025; Accepted: 07 August 2025;
Published: 28 August 2025.
Edited by:
Biran Zhu, Hubei University of Chinese Medicine, ChinaReviewed by:
Xuesheng Ma, University of the Western Cape, South AfricaHuanfang Xu, Institute of Acupuncture and Moxibustion (WHO Collaborating Center for Traditional Medicine), China
Copyright © 2025 Wei, Shen, Zhao, Xie, Bai, Yin and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jian Wang, d2FuZ2ppYW5qN0AxMjYuY29t
 Jiale Wei
Jiale Wei Zheng Shen1
Zheng Shen1