- 1Faculty of Chinese Medicine, Macau University of Science and Technology, Taipa, Macau SAR, China
- 2Mammary Department, Shuguang Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, China
- 3Faculty of Chinese Medicine, Shanghai University of Traditional Chinese Medicine, Shanghai, China
Objective: In this study, we retrospectively analyzed the relationship between PRL level and serum inflammatory and immune markers in patients with granulomatous lobular mastitis (GLM) and analyzed the effect of bromocriptine treatment on serum inflammatory and immune markers in patients with GLM. These analyses were conducted to illustrate that PRL is not only an endocrine hormone but also an immune factor, thereby providing evidence that GLM is an autoimmune disease.
Methods: We conducted a retrospective analysis of GLM cases admitted between 2023 and 2024. Clinical features were compared between patients with differential prolactin (PRL) levels using nonparametric tests, with concomitant documentation of prevalent clinical manifestations. Spearman’s rank correlation was employed to assess associations between PRL concentrations and clinical characteristics/serum biomarkers. To evaluate bromocriptine’s therapeutic efficacy, a propensity score-matched (PSM) cohort was established. Longitudinal serological changes were analyzed using nonparametric statistical methods for paired comparisons.
Results: Elevated prolactin levels significantly correlated with lesion size (p<0.05). Patients with abnormal PRL exhibited lower 6-month cure rates compared to those with normal levels (93.1% vs. 100%, p=0.02). Baseline-PRL positively associated with neutrophil counts (NE#), Immunoglobulin E (IgE), and ceruloplasmin (CER) (all p<0.05). After treatment, baseline-PRL remained linked to elevated neutrophils, C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), T4 lymphocyte percentage (CD4+ T cells), and IgE, but inversely correlated with lymphocytes (all p<0.05). Propensity-matched analysis (17 bromocriptine-treated vs. 13 no-treated) revealed reduced ESR, downregulated major histocompatibility complex class II (MHC-II) expression, and increased interleukin 4 (IL4), interleukin 5 (IL5), and regulatory T cell percentage (Treg%) levels in the treatment group (all p<0.05).
Conclusions: Our findings suggest prolactin may act as an immunomodulatory factor in GLM, potentially influencing T/B-cell immunity and inflammatory cytokine recruitment. Additionally, the observed correlation between prolactin and ceruloplasmin positions ceruloplasmin as a candidate biomarker for GLM, though further validation in independent cohorts is required.
1 Introduction
Granulomatous lobular mastitis, a benign chronic inflammatory disorder of mammary glandular tissue (1), demonstrates a prevalence of 3.5% in reproductive-aged populations (2). This condition predominantly manifests 3–5 years postpartum, persisting for 5–15 months (extending to 24 months in refractory cases) (3), with documented recurrence rates spanning 24.8-59% (4–6). While GLM is non-lethal, its severe symptomatology, protracted disease course, and high recurrence rates impose substantial physical and psychological burdens on patients (7). Although the precise etiology and pathogenesis remain undefined, GLM demonstrates strong epidemiological associations with pregnancy and hyperprolactinemia (1). The clinical characteristics of mammary inflammation, erythema nodosum, and arthritis, coupled with its enigmatic pathogenesis, have led to the prevailing hypothesis that GLM is an autoimmune entity. Prolactin, a pituitary-derived polypeptide hormone traditionally associated with lactogenesis and mammary gland development, demonstrates pleiotropic regulatory functions in both physiological and pathological contexts. Emerging evidence reveals PRL’s dual immunoregulatory capacity, functioning both as an endocrine hormone and a cytokine capable of precipitating immune hyperactivation (8–10). Clinically, bromocriptine (a dopamine D2 receptor agonist) serves dual therapeutic roles: as first-line management for hyperprolactinemia and as an immunomodulatory adjunct in GLM through PRL suppression. The demonstrated clinical efficacy of PRL normalization in GLM treatment provides compelling indirect evidence of their pathobiological interconnection.
This study retrospectively analyzed clinical and laboratory data of recent patients to investigate prolactin’s correlations with etiological factors, local symptoms, and serum biomarkers in GLM. We further examined the prognostic implications of baseline PRL levels on postoperative inflammatory/immune responses and evaluated bromocriptine’s therapeutic effects on immunoinflammatory parameters, aiming to investigate the potential association of PRL with these responses and to provide evidence supporting the hypothesis that GLM may be an autoimmune disease.
2 Patients and methods
2.1 Clinical data
Clinical records were retrospectively collected from patients hospitalized at Shuguang Hospital, Shanghai University of Traditional Chinese Medicine between January 2023 and December 2024.
2.2 Diagnostic criteria
All cases were pathologically diagnosed with GLM according to the criteria defined in “Management of GLM: An international multidisciplinary consensus (2021 edition)” (1). As per the consensus:”GLM is characterized histologically as non-caseating granulomatous lesions with epithelioid histiocytes and multinucleated giant cells, located in the center of the lobes. The surrounding tissue is mainly infiltrated by neutrophils, lymphocytes, plasma cells and a small number of eosinophils. The lesions can be multifocal and form micro-abscesses and vary in size.
2.3 Inclusion criteria
1. Biological females aged ≥18 years;
2. Histopathological confirmed GLM diagnosis (per institutional protocols);
3. Hospitalized patients receiving standardized care protocols with: Complete clinical documentation (symptom progression, physical findings); Comprehensive laboratory profiles (pre/post-operative inflammatory and immune biomarkers).
2.4 Exclusion criteria
1. Patients with incomplete medical records or missing essential laboratory data;
2. Individuals with concurrent severe comorbidities or undergoing therapies that may confound study outcomes;
3. Pregnant and lactating women.
2.5 Therapeutic protocol
All enrolled patients underwent protocol-defined surgical interventions coupled with standardized postoperative care. Participants in the bromocriptine cohort received supplemental perioperative administration of bromocriptine mesylate (2.5 mg orally twice daily) as adjuvant therapy. The control group received no adjuvant pharmacological therapy.
2.6 Data collection
Baseline clinical characteristics of the enrolled patients were documented, encompassing age, body mass index (BMI), postpartum and lactation periods, trauma and menstrual history, as well as smoking habits. Disease-related symptoms such as fever, leg erythema, arthritis, breast mass size, skin changes, suppuration, medication, and duration were also recorded. Additionally, serum laboratory data were obtained upon admission and after 14 days of treatment, including complete blood count, ESR, CRP levels, lymphocyte subsets, interleukins, and immunoglobulin. Follow-up assessments were conducted at 2-,3- and 6-month post-treatment to monitor the recovery progress of the patients.
2.7 Propensity score matching
PSM was performed to balance baseline characteristics between bromocriptine-treated and untreated cohorts, using 1:1 nearest-neighbor matching with a caliper width of 0.2 SD. Covariates included age, BMI, disease duration, lesion size, ulceration number, quadrant involvement, febrile status, erythema severity grading, trauma history, and pretreatment prolactin levels.
2.8 Statistical methods
Statistical analyses were performed using SPSS 23.0 and Origin-Pro 2025. Continuous variables with non-normal distribution (assessed by Shapiro-Wilk test) are presented as median (interquartile range) and analyzed using Mann-Whitney U/Kruskal-Wallis tests. Categorical variables are expressed as frequencies (%) with Pearson’s χ² or Fisher’s exact tests for comparisons. Spearman’s rank correlation coefficient was employed for bivariate analyses. Statistical significance was defined as two-tailed p<0.05.
2.9 Flowchart
The flow chart of this study is shown in Figure 1.
Figure 1. The flowchart of prolactin and bromocriptine on serum markers in Granulomatous Lobular Mastitis.
3 Results
3.1 Demographic and clinical characteristics
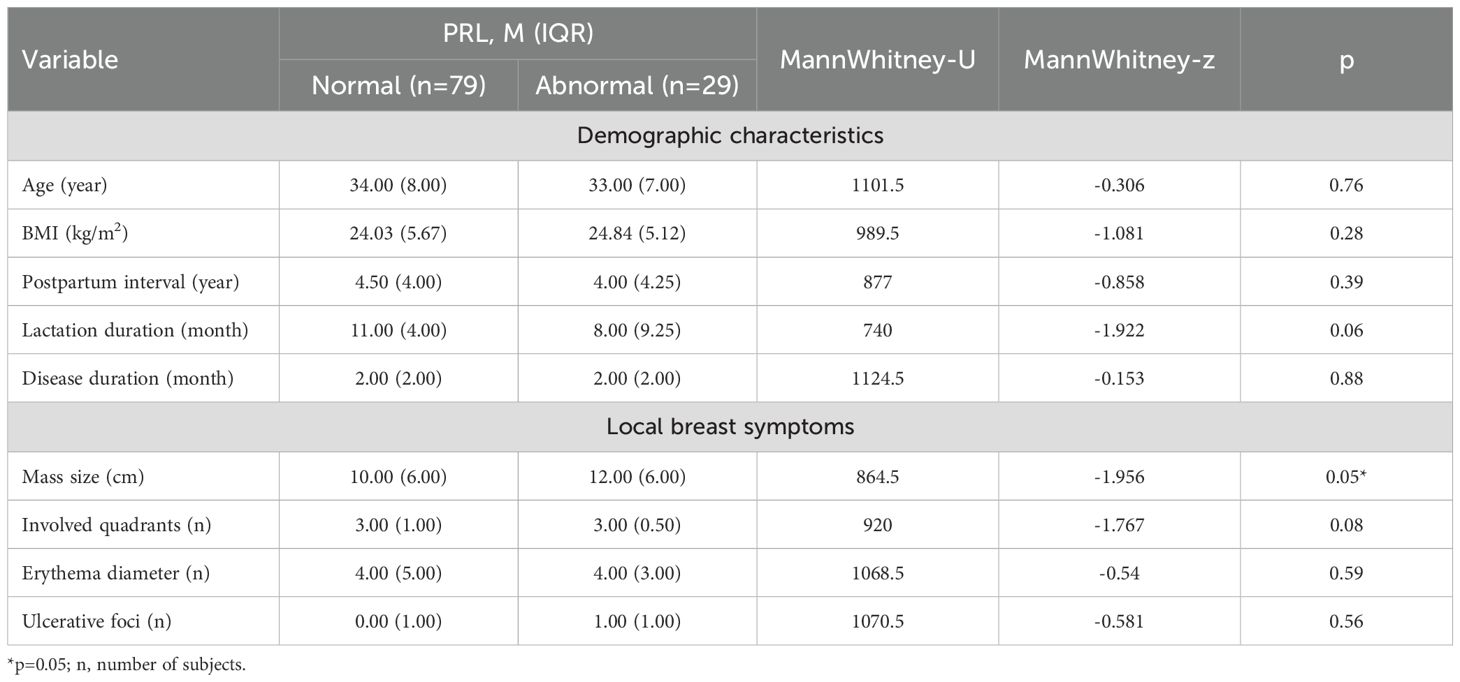
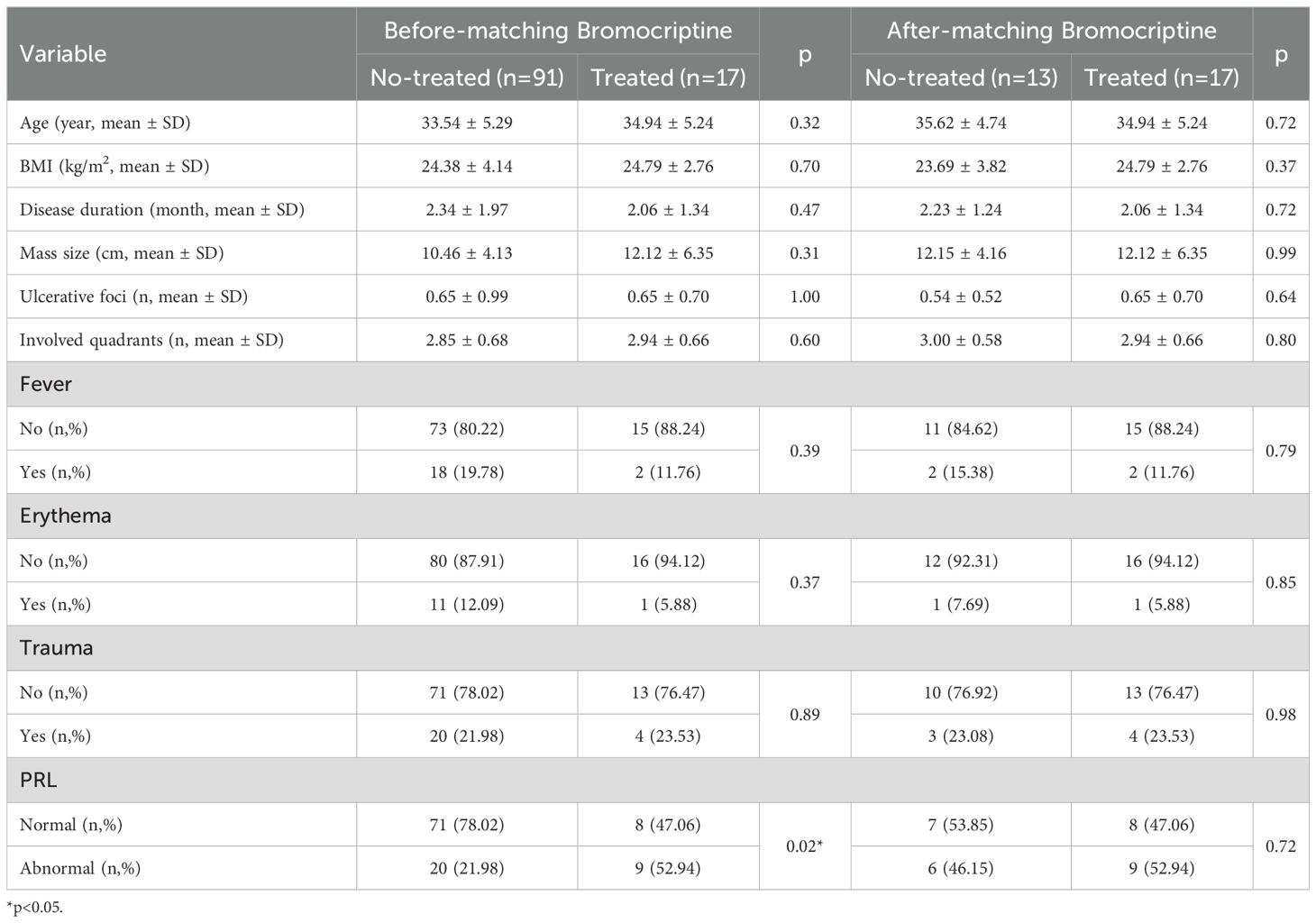
A total of 108 women meeting the inclusion criteria were included in the study, as detailed in Table 1. These women had a mean (SD) age of 33.76 (5.28) years and a BMI of 24.44 (3.95) kg/m². Stratified by serum PRL status, both groups had similar median BMI values close to the upper limit of normal (24.00 kg/m²), a median postpartum interval of 4 years, lesion sizes larger than 10 cm, median lactation duration exceeding 6 months, and disease duration surpassing 2 months. Notably, the group with PRL dysregulation exhibited significantly larger breast lesions compared to the other group (median 12 cm vs. 10 cm, p=0.05) (Table 1).
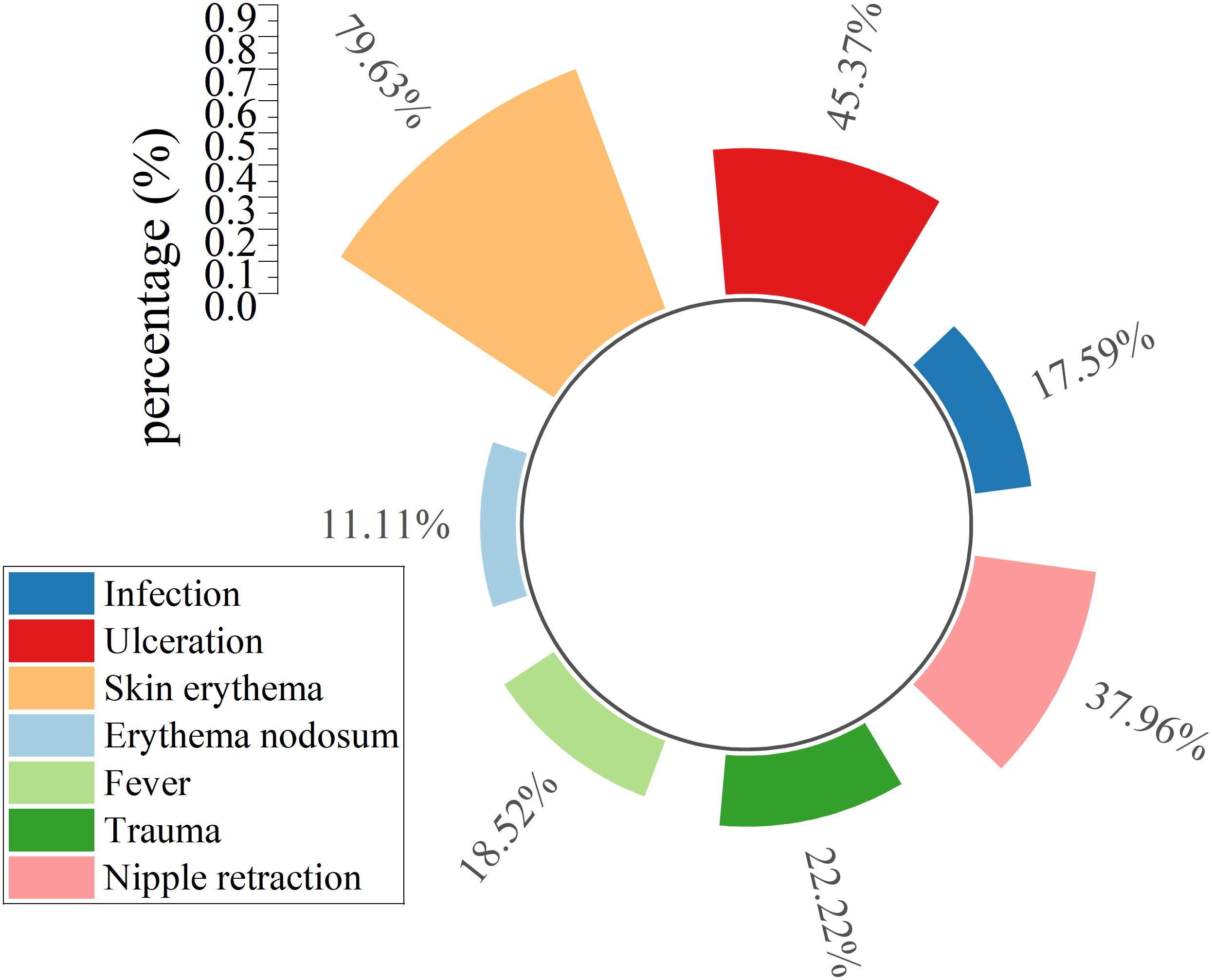
Local breast manifestations (excluding masses) and systemic symptoms were systematically cataloged in Figure 2. Local breast findings including erythema (79.63%), ulceration (45.37%), and nipple retraction (37.96%) demonstrated significantly higher prevalence than systemic manifestations such as fever (18.52%) and erythema nodosum (11.11%). Bacterial infections (17.59%) as the second least frequent complication throughout clinical management, marginally exceeding erythema nodosum (11.11%) (Figure 2).
3.2 Correlation between baseline-PRL and GLM
3.2.1 Baseline-PRL levels and prognosis
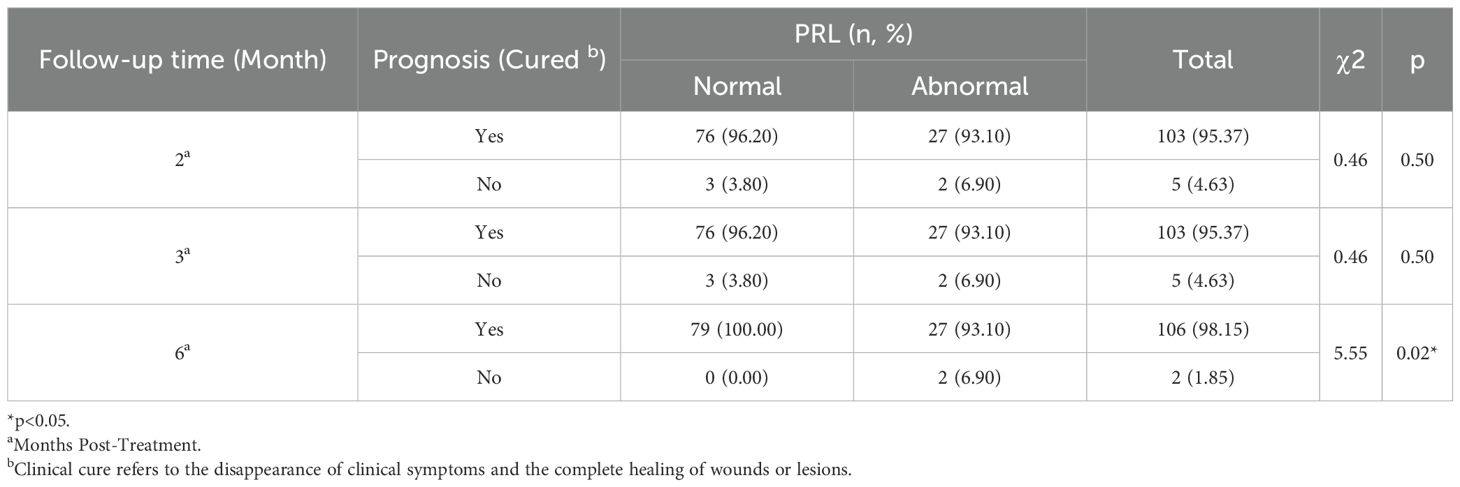
Longitudinal follow-up evaluations were conducted at 2-, 3-, and 6-month post-treatment and systematically examined (Table 2). Patients exhibiting prolactin dysregulation showed notably lower rates of remission at the 6-month follow-up in comparison to patients with normal prolactin levels (Table 2).
3.2.2 Correlations between baseline-PRL levels and inflammatory markers
Significant correlations emerged between baseline prolactin levels and inflammatory indices (Figure 3): positive associations with neutrophilic markers (both NE% and NE#) contrasted with negative correlation to lymphocyte percentage (LY%) (all p<0.05, Spearman’s test).
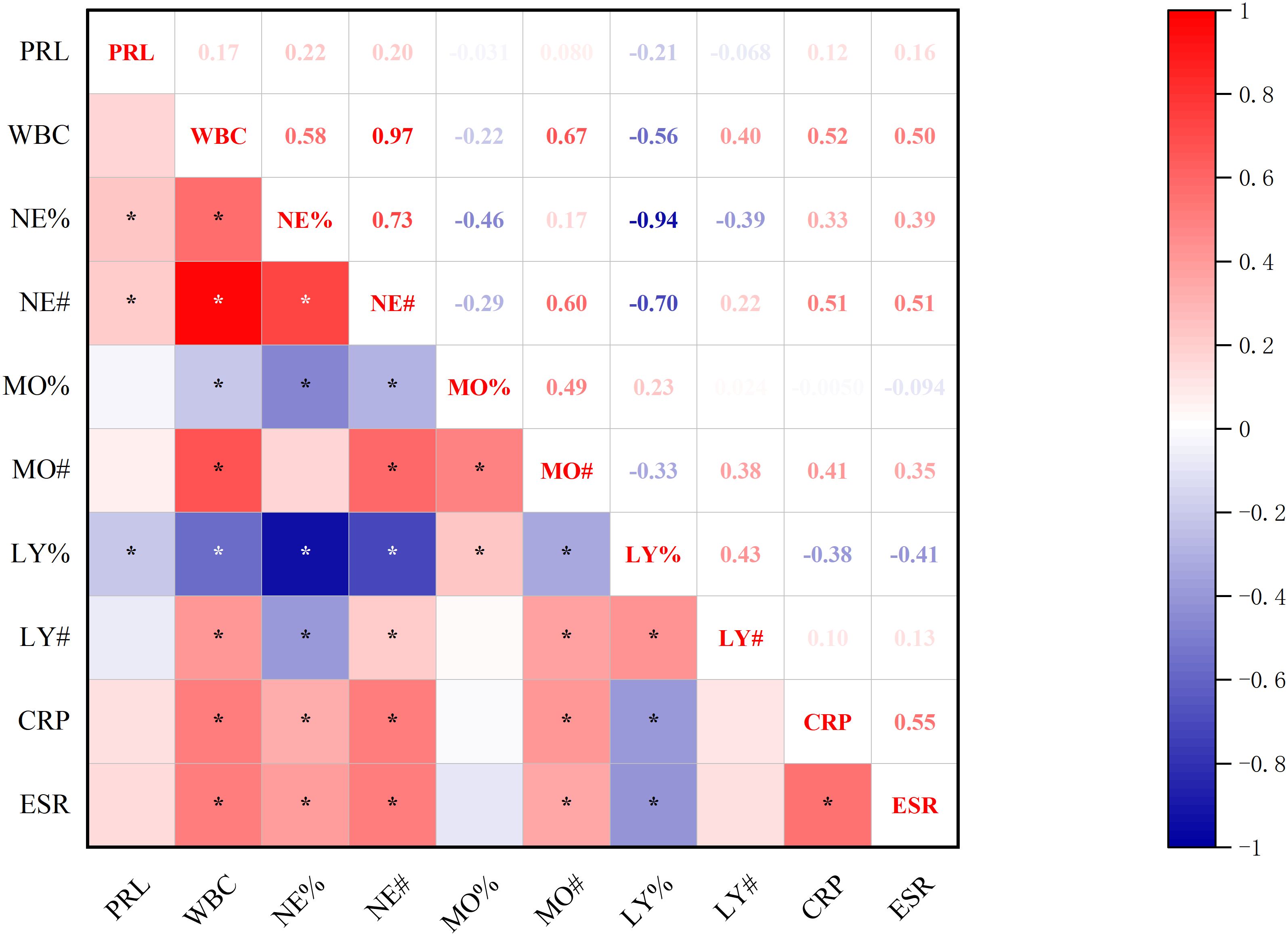
Figure 3. Correlations between baseline-PRL Levels and inflammatory markers. *p<0.05; PRL, prolactin blood level; WBC, white blood cell count; NE%, neutrophil percentage; NE#, neutrophil count; MO%, monocyte percentage; MO#, monocyte count; LY%, lymphocyte percentage; LY#, lymphocyte count; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate.
3.2.3 Correlations between baseline-PRL levels and immunoglobulins
Significant correlations emerged between baseline prolactin levels and immunological markers (Figure 4): positive associations were observed with IgE and CER. Furthermore, CER demonstrated additional correlations with IgM and complement system components (CH50), while IgE showed a positive association with IgG. PRL was not significantly associated with Interleukin and lymphocyte subsets (all p<0.05, Spearman’s test).
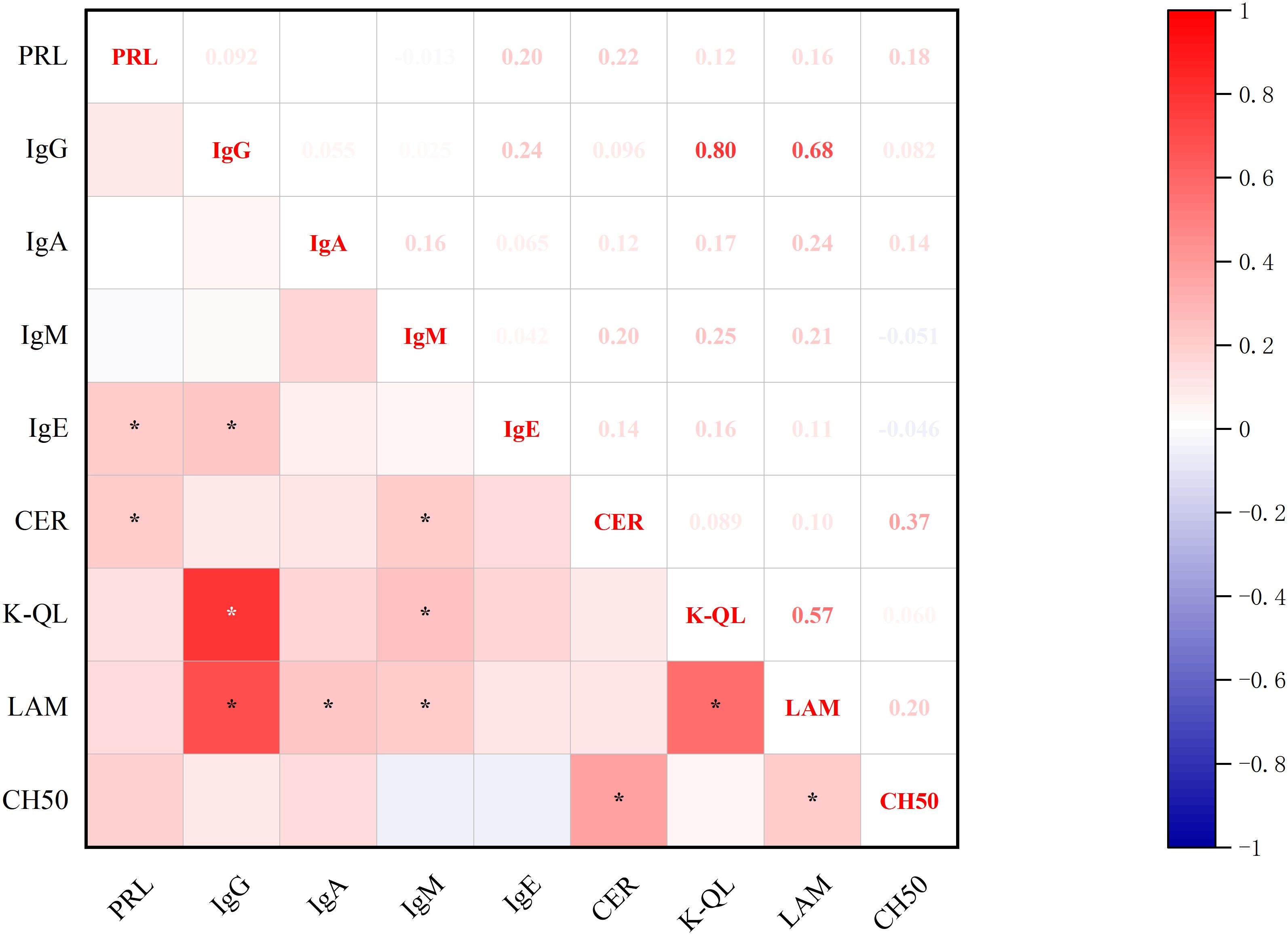
Figure 4. Correlations between PRL Levels and immunoglobulin. *p<0.05; PRL, prolactin blood level; IgG, immunoglobulin G; IgA, immunoglobulin A; IgM, immunoglobulin M; IgE, immunoglobulin E; CER, ceruloplasmin; K-QL, Kappa light chain; LAM, Lymphocyte Activation Markers; CH50, Complement system components.
3.2.4 Correlations between baseline-PRL levels and clinical features
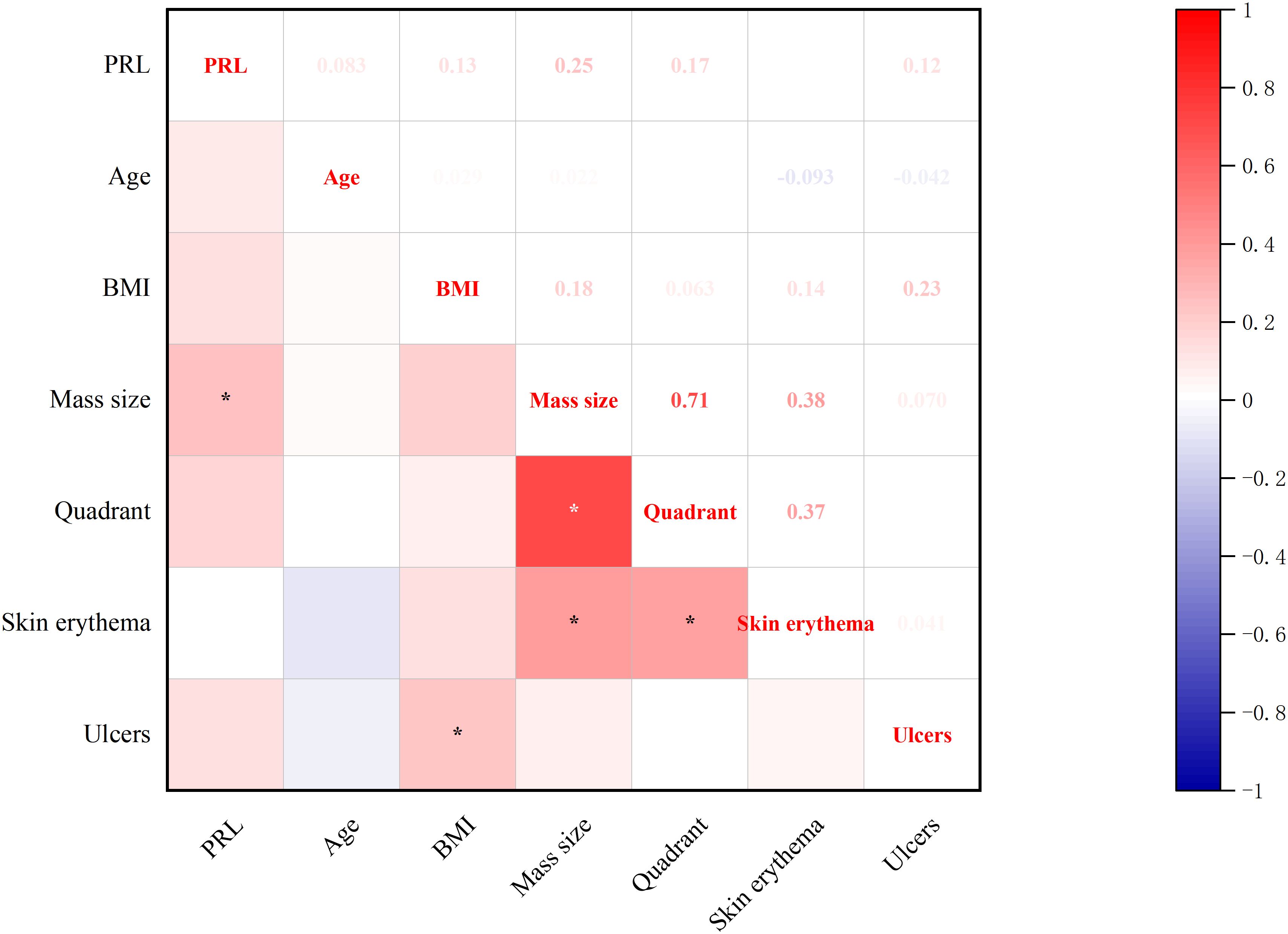
Significant correlations emerged between baseline prolactin levels and clinical features (Figure 5): direct associations were observed with breast mass size. Concurrently, BMI showed positive correlations with ulcers which present suppurative breast lesion development (all p<0.05, Spearman’s test).
3.3 Correlations between baseline-PRL and GLM serum markers after treatment
3.3.1 Correlations between baseline-PRL and post-treatment inflammatory makers
Significant correlations were observed between baseline-PRL levels and post-treatment inflammatory markers, including complete blood count parameters, CRP, ESR (Figure 6). PRL demonstrated positive associations with NE%, CRP, and ESR, while showing a negative correlation with lymphocyte (both LY# and LY%) (all p<0.05, Spearman’s test).
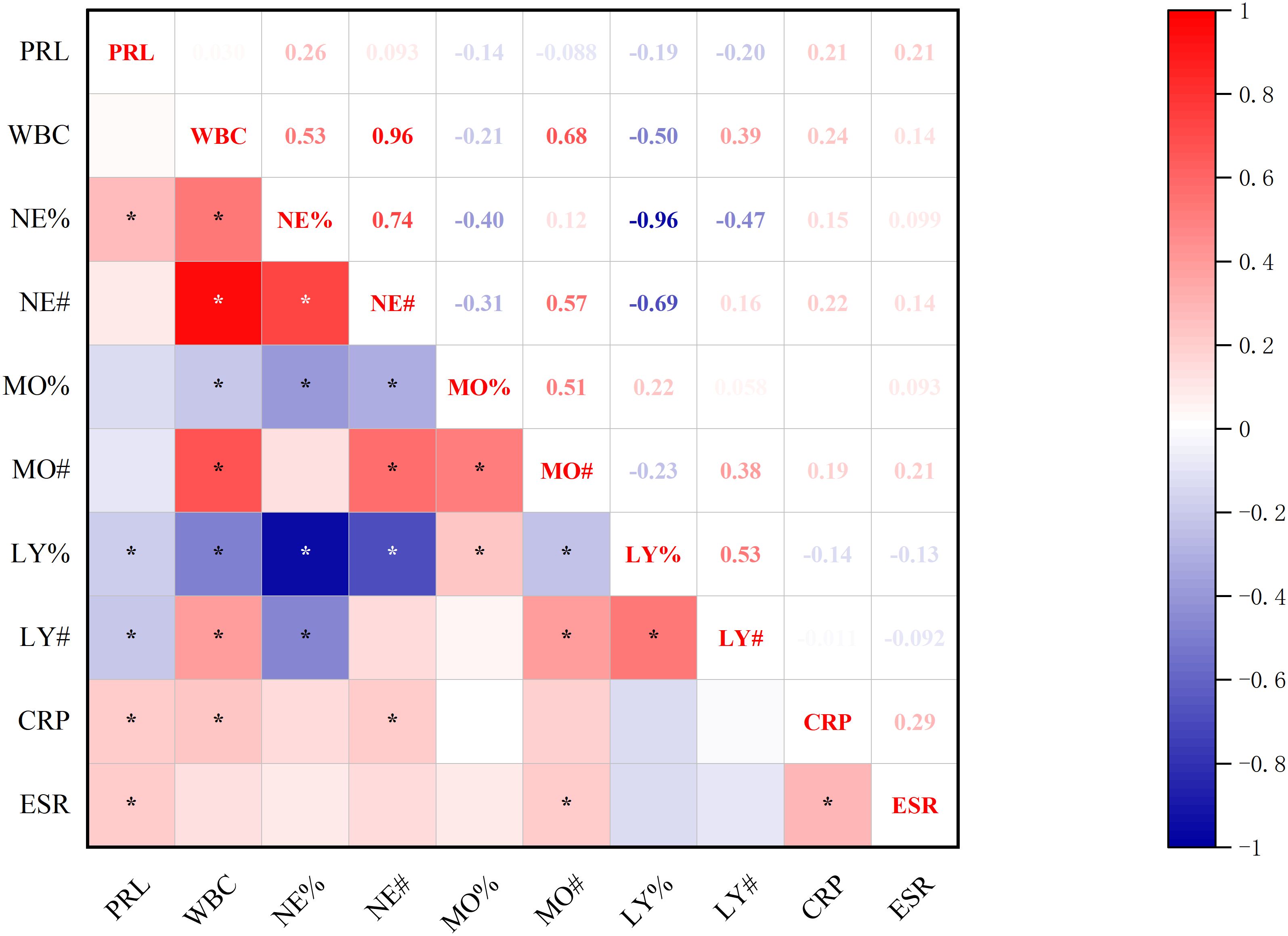
Figure 6. Correlation between baseline-PRL and post-treatment inflammatory makers. *p<0.05; PRL, prolactin blood level; WBC, white blood cell count; NE%, neutrophil percentage; NE#, neutrophil count; MO%, monocyte percentage; MO#, monocyte count; LY%, lymphocyte percentage; LY#, lymphocyte count; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate.
3.3.2 Correlations between baseline-PRL and post-treatment immune molecules
Significant correlations emerged between baseline-PRL levels and post-treatment immune profiles (Figures 7, 8). PRL demonstrated positive associations with CD4+ T% and IgE. Furthermore, CD4+ T% showed positive correlations with CD3+ T%, CD2+, Treg%, and CD4+/CD8+, while negative correlations with CD16+ NK%, CD8+ T%, and CD8+CD38+ (all p<0.05, Spearman’s test).
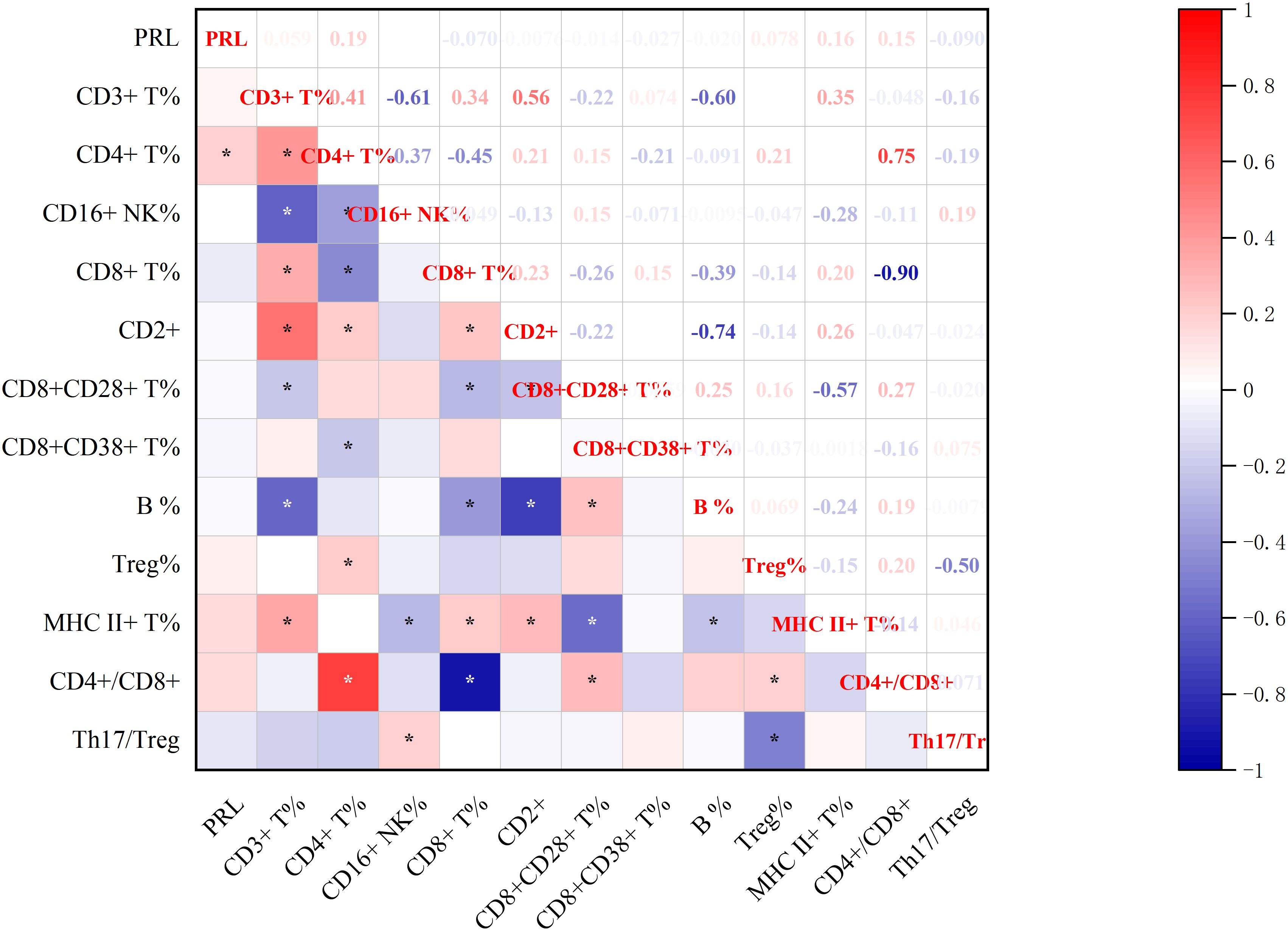
Figure 7. Correlation between baseline-PRL and post-treatment Lymphocyte Subsets.*p<0.05; CD3+ T%, total T lymphocyte percentage; CD4+ T%, T4 lymphocyte percentage; CD16+ NK%, NK lymphocyte percentage ; CD8+ T%, T8 lymphocyte percentage; CD2+, leukocyte function-associated antigen; CD8+CD28+ T%, CD28+ T8 lymphocytes ; CD8+CD38+ T%,CD38+ T8 lymphocytes ; CD19+ B%, B lymphocyte percentage; Treg%, regulatory T cell percentage; MHC II+ T%, MHC-II T cells; CD4+/CD8+,CD4/CD8 T4/T8 lymphocyte ratio; Th17/Treg, helper-T 17/ regulatory T cell radio.
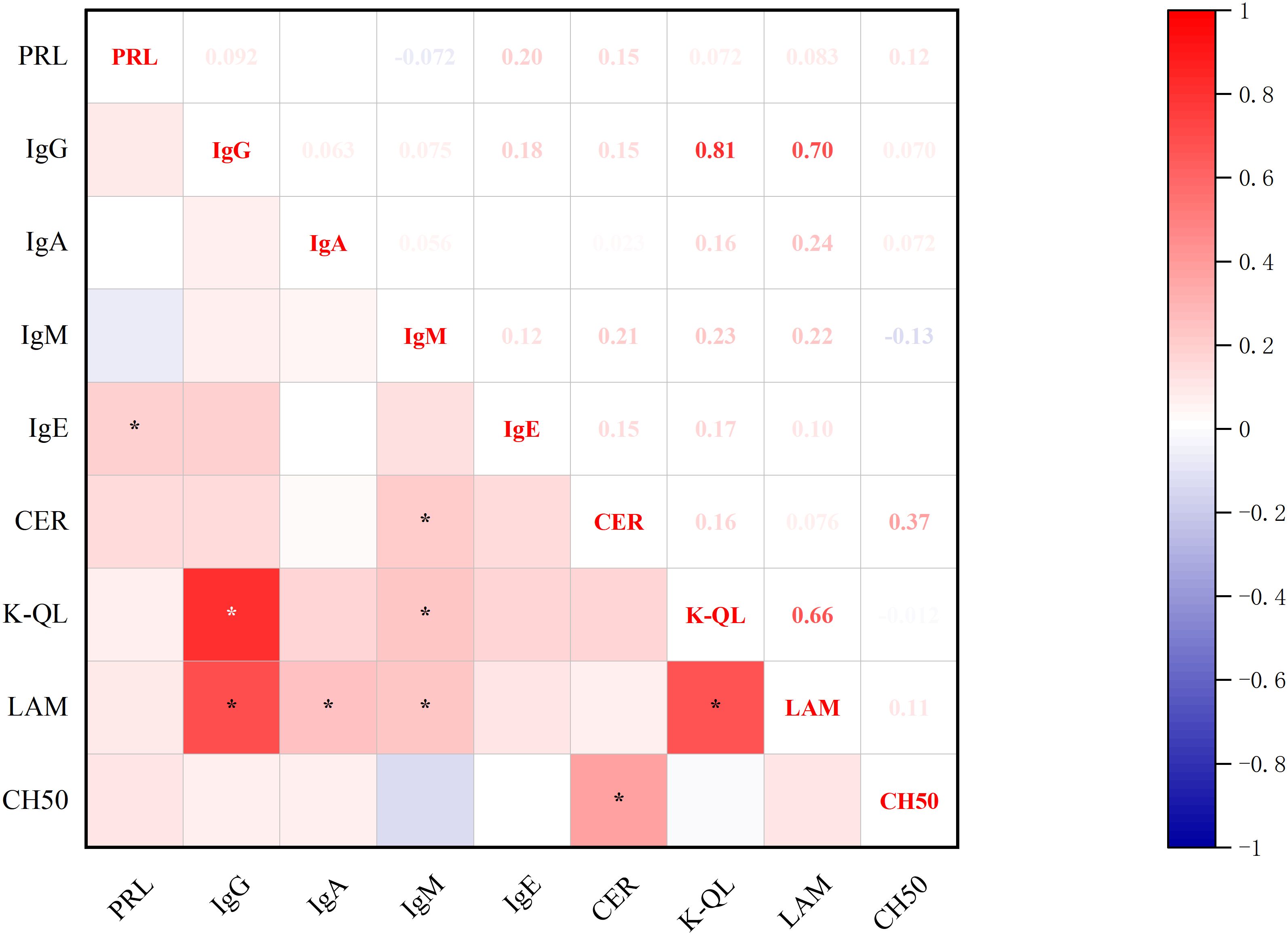
Figure 8. Correlations between baseline-PRL and post-treatment immunoglobulins. *p<0.05; PRL, prolactin blood level; IgG, immunoglobulin G; IgA, immunoglobulin A; IgM, immunoglobulin M; IgE, immunoglobulin E; CER, ceruloplasmin; K-QL, kappa light chain; LAM, lymphocyte activation markers; CH50, complement system components.
3.4 Propensity-matched analysis of the effect of bromocriptine on GLM
To elucidate the PRL-GLM interaction, we conducted PSM using clinical features in the study cohort. Patients were stratified into bromocriptine-treated (n=17) and untreated control (n=13) groups based on therapeutic regimen (Table 3).
3.4.1 Effect of bromocriptine on inflammatory markers
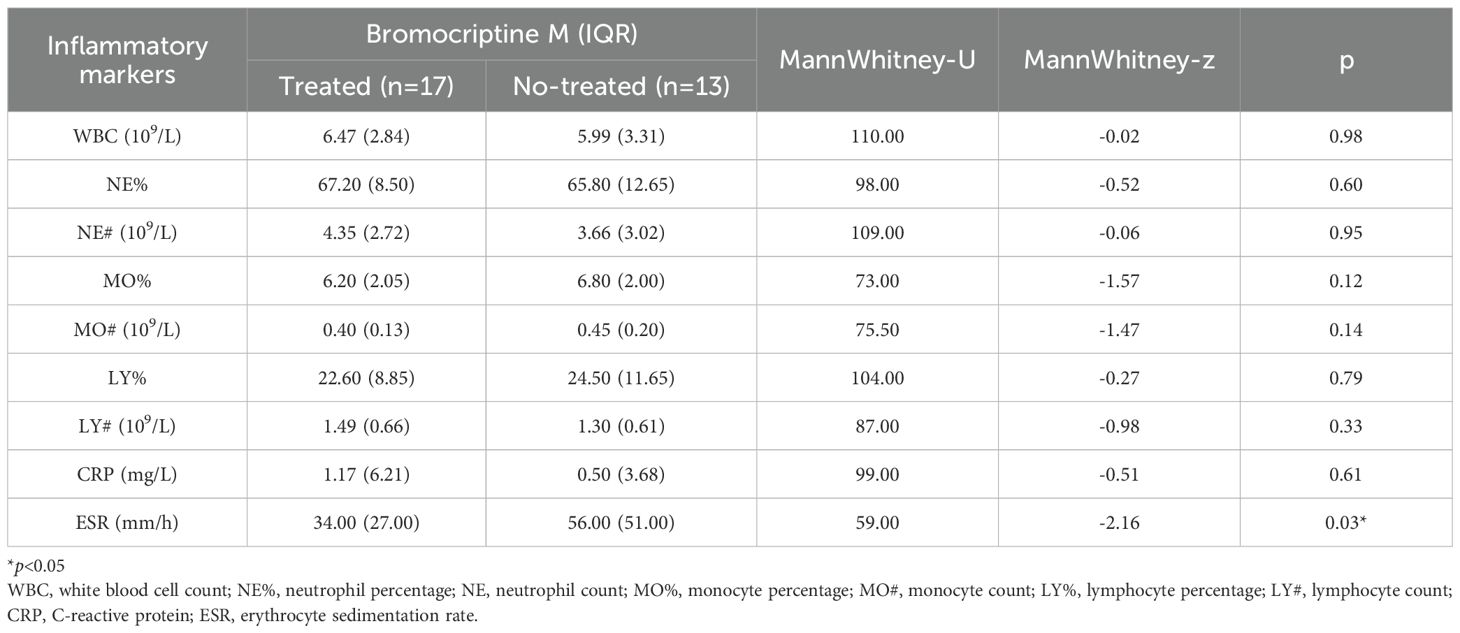
In the propensity-matched cohort, the bromocriptine-treated group demonstrated significantly lower prolactin levels and ESR compared to untreated controls (Table 4).
3.4.2 Effect of bromocriptine on immune molecules
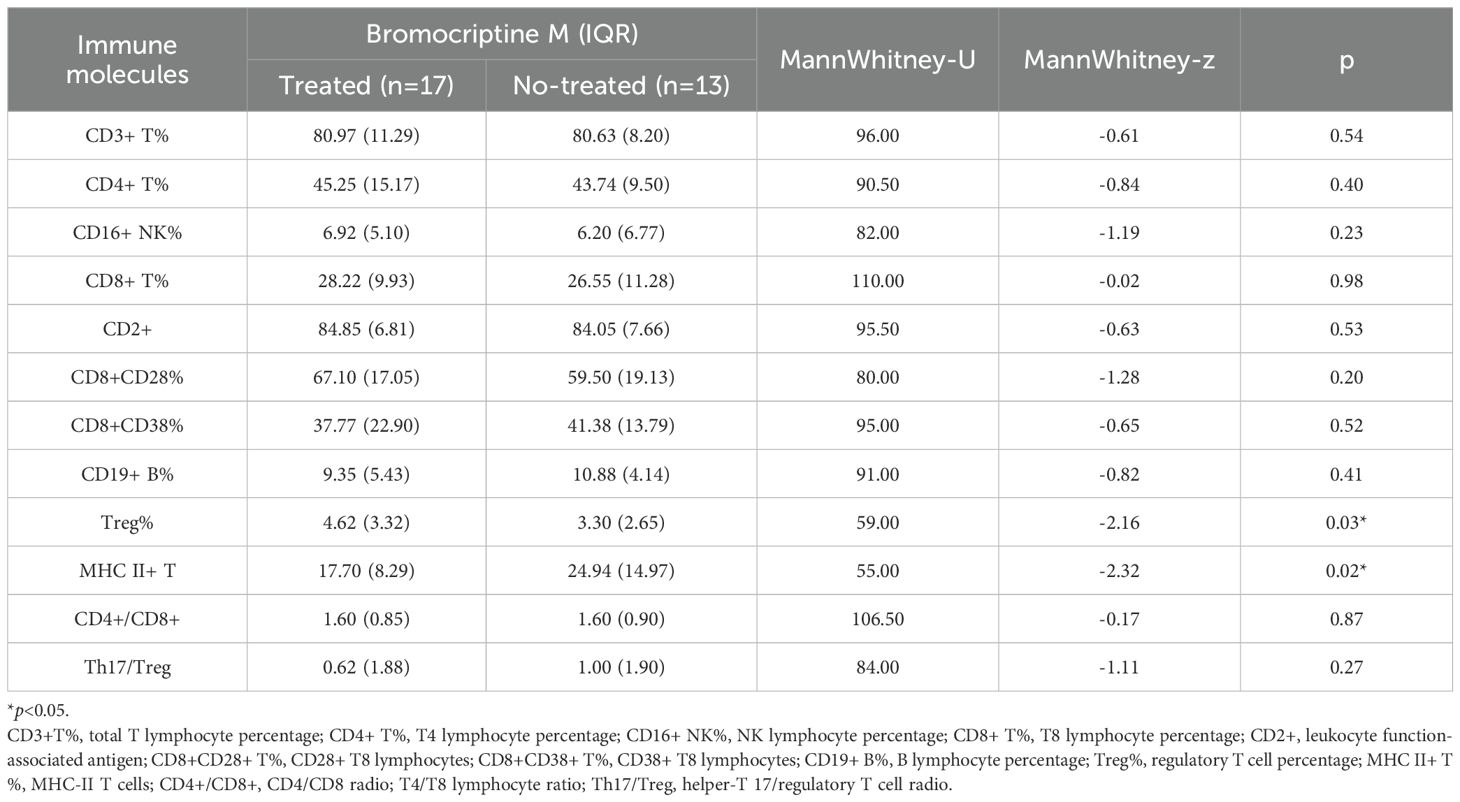
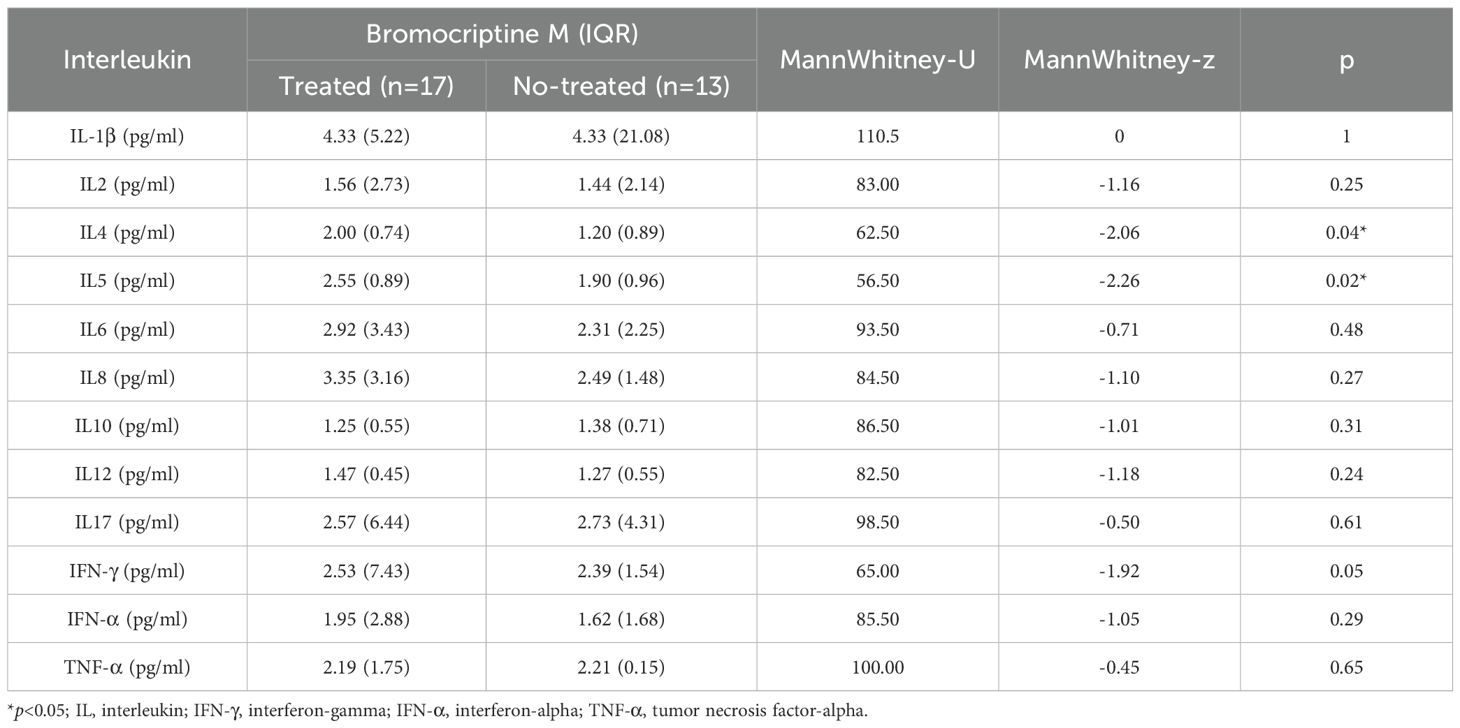
In the propensity-matched cohort, the bromocriptine-treated group demonstrated significantly elevated Treg%, IL4, and IL5 compared to untreated controls. Conversely, MHC-II+T cells expression was markedly reduced in the treatment group (Tables 5, 6).
4 Discussion
4.1 GLM: a persistent therapeutic conundrum
Granulomatous lobular mastitis, a benign chronic inflammatory breast condition predominantly affecting women of childbearing age (median 33 years, typically 4 years postpartum), demonstrates prolonged clinical courses (≤2 years) and high recurrence rates (24.8-59%) (1, 3–6). Our surgical cohort exhibited severe disease manifestations: median lesion size 10 cm, multi-quadrant involvement (≥2 quadrants), and suppuration (up to 6 episodes). Localized breast symptoms significantly outweighed systemic manifestations, with bacterial infections implicated in only 17.59% of cases, substantially limiting antibiotic efficacy. These findings underscore the dual burden of GLM - physically debilitating presentations compounded by profound psychosocial distress (7). The urgent need persists for pathogenesis elucidation, diagnostic biomarker development, and targeted therapeutic strategies to mitigate diagnostic delays and optimize clinical management.
4.2 PRL dysregulation in GLM progression
Amidst etiological controversies, pregnancy and hyperprolactinemia emerge as the most substantiated GLM correlates (1, 11, 12), demonstrating stronger pathogenic relevance than menstrual irregularities, nipple deformity or obesity (13–17). Emerging evidence consistently implicates prolactin dysregulation in GLM pathogenesis and progression (1). Patients with GLM demonstrate significantly elevated PRL levels compared to healthy controls (18), with lactation-associated hyperprolactinemia identified as a primary risk factor through its promotion of ductal secretion abnormalities (17). Notably, intracranial lesion-induced PRL elevation may directly contribute to GLM development (19), underscoring the importance of routine PRL screening in clinical evaluation. Longitudinal observations further suggest that dynamic changes in PRL levels before and after treatment correlate with recurrence risk (20), highlighting the prognostic significance of PRL monitoring. These collective findings establish PRL modulation as a critical component in comprehensive GLM management strategies (21). Our study shows that PRL abnormalities may affect cure rates 6 months after treatment. Meanwhile, our findings demonstrate that patients with PRL dysregulation present significantly larger breast lesions, with a positive correlation observed between PRL levels and lesion dimensions, reinforcing the pathophysiological link between hyperprolactinemia and disease severity. Importantly, lesion size emerged as a key prognostic indicator: larger lesions correlated with intensified pain perception, broader anatomical involvement, and prolonged recovery duration, consistent with established clinical patterns. Our study provides a foundation for further investigation into the association between PRL and disease severity.
4.3 Correlation between PRL and inflammatory markers
Histopathological evaluation reveals GLM is characterized by dense inflammatory cell infiltration surrounding mammary lobules, predominantly comprising neutrophils, lymphocytes, and plasma cells (22). While neutrophils initiate acute inflammatory responses, their excessive accumulation disrupts resolution pathways, perpetuating tissue damage. This persistent inflammatory milieu, potentially modulated by PRL dysregulation, demonstrates dynamic cytokine fluctuations that may fuel disease chronicity (23, 24). It has been found that PRL promotes dendritic cell maturation, enhances Th1-mediated immunity (Th1 immunity), and affects neutrophil function by increasing pro-inflammatory factors (25). Our findings reveal intensified neutrophil recruitment in patients with PRL abnormalities, aligning with GLM’s characteristic histopathological patterns (22). Notably, sustained neutrophilic involvement post-surgery, coupled with positive correlations between PRL levels and inflammatory markers (CRP and ESR), underscores PRL’s role in amplifying inflammatory intensity. Meantime, the consistent inverse relationship between PRL concentrations and lymphocyte proportions across treatment phases suggests PRL is associated with immune imbalance.
To our knowledge, our study is the first to report a positive correlation between prolactin and ceruloplasmin in GLM. As a multicopper oxidase, CER serves as a biomarker of systemic inflammation, oxidative stress, and copper/iron metabolism (26). The association between CER and oxidative stress is mechanistically linked to IL-6 induction, particularly in lipopolysaccharide (LPS)-driven pathogenesis (27, 28). Prior evidence supports CER’s involvement in autoimmune disorders such as rheumatoid arthritis and lupus erythematosus, providing a pathogenic analogy for GLM’s potential autoimmune etiology (29, 30). Notably, serum CER reflects systemic copper/iron metabolism, and recent studies have established a link between GLM and iron dysregulation: bacterial LPS underlies GLM pathogenesis via iron depletion, mammary cell death, and subsequent IL-6 up-regulation (27, 28). Furthermore, CER is implicated in immune processes, with evidence suggesting its association with immune dysregulation. In studies exploring CER as a prognostic biomarker in breast cancer, CER levels were correlated with T-cell activity and macrophage polarization, demonstrating positive associations with M0/M1 infiltration and negative correlations with M2 macrophages (27, 28). Our findings demonstrate a correlation between CER and PRL, which may further underscore the association of CER with immune dysregulation while positioning CER as a candidate disease-specific diagnostic biomarker for GLM.
4.4 Correlation between PRL and T-cell Immunity
Lymphocyte subsets encompass functionally distinct T cells, B cells, and NK cells in peripheral blood, defined by unique surface markers. These populations collaboratively regulate immune defense, homeostasis, and surveillance through specialized roles in adaptive and innate immunity. Prolactin can affect the expression of T-bet gene by affecting CD4+ T cells, and activated T-bet can further enhance the expression of CD4+ T cells. CD4+ T cells have been associated with GLM severity and exhibit dynamic changes during therapy (16, 31, 32). Our study revealed a positive correlation between baseline-PRL levels and post-treatment CD4+ T cells, suggesting that PRL fluctuations may modulate CD4+ T cell dynamics and trigger downstream immune cascades.
CD4+ T cells differentiate into functionally distinct subsets (Th1, Th2, Th17, Treg) under specific stimuli. Emerging evidence highlights Th17 cells as key players in granulomatous pathogenesis, with Th17/Treg imbalance being mechanistically linked to autoimmune disorders and directly associated with granuloma formation in GLM (33–35). Treg cells counterbalance inflammatory responses through TGF-β and IL-10 secretion (10, 36). Intriguingly, while baseline-PRL showed no direct correlation with Treg%, its positive association with CD4+ T%, which correlated positively with Treg%, suggests that PRL may indirectly modulate Treg activity via CD4+ T cells intermediaries. Our PSM analysis further demonstrated elevated Treg% post-treatment, indicating PRL suppression enhances Treg-mediated immunoregulation. These findings collectively suggest PRL may constrain Treg activation, thereby exacerbating GLM-related immune dysregulation.
Additionally as a pivotal mediator of inflammatory cell-T cell communication, MHC-II interacts with surface receptors on antigen-presenting cells including dendritic cells, macrophages, and B lymphocytes (37, 38).In our outcomes, bromocriptine induced PRL reduction coincided with diminished MHC-II expression in treated patients, suggesting a potential interplay between PRL signaling and antigen presentation pathways. This observation, coupled with parallel declines in CRP and ESR, implies PRL-mediated immunomodulation may involve MHC-II-related mechanisms.
4.5 Correlation between PRL and B-cell immunity
The immune pathogenesis of GLM involves both innate and adaptive immunity (10, 18, 39, 40). B cells differentiate into plasma cells that secrete immunoglobulins such as IgG and IgE. Notably, prolactin receptors (PRLR) are expressed on B cells at various developmental stages, and dysregulated PRL levels can alter B-cell proliferation and immune function (41). Our findings demonstrate a persistent positive correlation between PRL and IgE levels, even postoperatively. The characteristic histopathological feature of plasma cell infiltration in GLM further confirms adaptive immune activation. Given that plasma cell abundance reflects tissue inflammation severity, consistent with the observed PRL inflammation marker correlations. We propose that PRL may contribute to GLM pathogenesis by modulating B-cell activity.
In our PSM cohort, PRL suppression therapy led to significant increases in IL-4 and IL-5 levels, indicating Th2-mediated immunity (Th2 immune) activation. This finding aligns with prior studies demonstrating that PRL modulates Th2 immune responses to influence disease progression (42), thereby supporting both the “PRL-Th2 drives GLM pathogenesis” hypothesis and providing new evidence for the “Th1/Th2 imbalance contributes to GLM development” theory (32). Notably, IL-4 plays a unique role in tissue repair: it promotes healing by inducing macrophage M2 polarization and stimulating the secretion of anti-inflammatory factors such as IL-10 (38). The observed elevation of IL-4 post-bromocriptine treatment in our study likely reflects dynamic reprogramming of macrophage polarization states, which strongly aligns with the mechanistic model that “macrophage phenotype switching regulates GLM progression and resolution.
4.6 PRL: a dual-function hormone and immune cytokine
Prolactin, a multifunctional molecule with a molecular weight of approximately 23 kDa, is secreted predominantly by the pituitary gland but can also be locally synthesized in extra pituitary tissues, including peripheral immune cells and mammary glands (8). The PRLR is widely expressed on the surface of diverse immune cells, such as lymphocytes (particularly natural killer cell subsets), macrophages, monocytes, granulocytes, and thymic epithelial cells, providing a structural basis for PRL’s direct regulation of immune activities (9, 43, 44). By binding to PRLR, PRL activates the Janus kinase 2/signal transducer and activator of transcription 5 (JAK2/STAT5) signaling pathway, which not only enhances Th1/Th17 responses and suppresses Treg function but also modulates NK cell cytotoxicity and macrophage polarization (8, 10, 43–47). Notably, patients with GLM often exhibit systemic manifestations such as arthritis and lower limb erythema, clinical features that closely resemble those of classic autoimmune diseases (10, 44). This observation suggests that PRL may serve as a key regulatory factor linking localized inflammation in GLM to systemic immune dysregulation. The broad distribution of PRLR underscores PRL’s unique role as an immunoendocrine bridge molecule, transcending its traditional perception as a lactation associated hormone (8, 43, 44).
Our study demonstrates that PRL directly modulates inflammatory and immune responses in GLM, as evidenced by its significant positive correlations with neutrophil activity and IgE proliferation (8, 44). In the PSM cohort, PRL suppression therapy reduced MHC-II expression while enhancing Treg, IL4 and IL5 differentiation, indicating PRL’s regulatory role in immune signaling and tolerance (10, 42). These findings provide novel insights into the persistent inflammatory microenvironment of GLM.
5 Conclusion
The etiology and pathogenesis of GLM remain incompletely understood, yet our findings strongly support the emerging autoimmune hypothesis by repositioning prolactin as a multifunctional immunomodulator rather than a conventional endocrine hormone. Clinically, PRL levels correlated with lesion size, neutrophil infiltration, CER, post-treatment CD4+ T cells and IgE levels, suggesting its dual role in both innate and adaptive immune dysregulation. Through PSM analysis, bromocriptine-induced PRL suppression was associated with reduced inflammatory markers (ESR and MHC-II) and enhanced Treg/Th2 responses (elevated Tregs, IL-4, IL-5). These observations reveal PRL’s central role in orchestrating immune homeostasis, where its dysregulation may perpetuate GLM pathology through intertwined inflammatory and immunoregulatory pathways. Our work not only provides clinical validation for PRL-driven autoimmunity in GLM but also highlights its potential as a therapeutic target for rebalancing immune tolerance. Additionally, we first report CER dysregulation in GLM, which provides a new perspective for GLM research.
5.1 Limitations
This study has several limitations. First, while our results demonstrate correlations between PRL and inflammatory/immune factors, these relationships require validation through basic research to elucidate the underlying mechanisms and molecular targets. Second, the retrospective design and hospital-based case selection may limit the robustness of certain findings—for instance, the association between lactation duration and PRL levels showed borderline significance (P=0.06). Although PSM mitigated confounding effects, its efficacy was constrained by the original cohort size, underscoring the need for larger-scale studies with prospective designs to enhance scientific validity. Additionally, the 2-week post-treatment observation window likely captured only a transient snapshot of immunological changes rather than the complete trajectory of GLM resolution. These limitations highlight the necessity of prospective multicenter studies with extended follow-up periods and standardized protocols to fully characterize PRL’s mechanistic role in GLM pathogenesis.
5.2 Future perspectives
Despite these limitations, our findings provide a crucial foundation for advancing GLM research and clinical management. Building on this work, future studies should prioritize expanding sample sizes through multi-center collaborations to enhance statistical validity. Integrating the emerging concept of GLM staging systems could enable mechanistic exploration across disease phases, offering insights into stage-specific pathophysiology. Prospective longitudinal designs are needed to clarify causal relationships between PRL dynamics and GLM progression. Furthermore, experimental models (e.g., immune cell co-cultures and GLM animal models) could unravel PRL’s direct immunomodulatory effects.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The studies involving humans were approved by the Ethics Committee of Shuguang Hospital affiliated to Shanghai University of Traditional Chinese Medicine (approval number 2024-1491-074-01, issued on 10 April 2024). The studies were conducted in accordance with the local legislation and institutional requirements. The human samples used in this study were acquired from a by-product of routine care or industry. Written informed consent for participation was not required from the participants or the participants’ legal guardians/next of kin in accordance with the national legislation and institutional requirements.
Author contributions
BL: Writing – review & editing, Writing – original draft, Data curation, Formal analysis, Visualization. WQ: Formal analysis, Writing – original draft, Writing – review & editing. WC: Supervision, Conceptualization, Writing – review & editing. HW: Conceptualization, Methodology, Writing – review & editing. YF: Funding acquisition, Writing – review & editing. XW: Resources, Writing – review & editing. JF: Investigation, Writing – original draft. QG: Project administration, Writing – original draft. SS: Data curation, Writing – original draft. JS: Data curation, Visualization, Writing – original draft.
Funding
The author(s) declare that no financial support was received for the research, and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer YY declared a shared parent affiliation with the authors WQ, HW, QW YF, JF, QG, SS, JS to the handling editor at the time of the review.
Generative AI statement
The author(s) declare that Generative AI was used in the creation of this manuscript. This manuscript is the original work of our research team. AI was solely utilized for language translation to enhance clarity. All data and viewpoints are the product of our team’s efforts, with AI not involved in research design, data analysis, or interpretation. We affirm the integrity and originality of our research contributions.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Yuan QQ, Xiao SX, Farouk O, Du YT, Sheybani F, Tan QT, et al. Management of granulomatous lobular mastitis: An international multidisciplinary consensus (2021 edition). Mil Med Res. (2022) 9:20. doi: 10.1186/s40779-022-00380-5
2. Martinez-Ramos D, Simon-Monterde L, Suelves-Piqueres C, Queralt-Martin R, Granel-Villach L, Laguna-Sastre JM, et al. Idiopathic granulomatous mastitis: A systematic review of 3060 patients. Breast J. (2019) 25:1245–50. doi: 10.1111/tbj.13446
3. Hua C, Li F, Shi Y, Xu Y, Zhu M, Wang Y, et al. Long-term outcomes of traditional Chinese medicine in the treatment of granulomatous lobular mastitis: A two-year follow-up study on recurrence and new occurrence rates with analysis of risk factors. J Inflamm Res. (2024) 17:7389–99. doi: 10.2147/JIR.S485589
4. Azizi A, Prasath V, Canner J, Gharib M, Sadat Fattahi A, Naser Forghani M, et al. Idiopathic granulomatous mastitis: Management and predictors of recurrence in 474 patients. Breast J. (2020) 26:1358–62. doi: 10.1111/tbj.13822
5. Aghajanzadeh M, Hassanzadeh R, Alizadeh Sefat S, Alavi A, Hemmati H, Esmaeili Delshad MS, et al. Granulomatous mastitis: Presentations, diagnosis, treatment and outcome in 206 patients from the north of iran. Breast. (2015) 24:456–60. doi: 10.1016/j.breast.2015.04.003
6. Kayadibi Y, Ucar N, Akan YN, Kaya MF, Yildirim E, Kurt SA, et al. Magnetic resonance imaging findings associated with recurrence in idiopathic granulomatous mastitis. Clin Imaging. (2022) 84:47–53. doi: 10.1016/j.clinimag.2022.01.010
7. Wang YT, Tang YQ, Xu JY, and Bian XM. The disease experience of women with granulomatous lobular mastitis. A Qual Interv Study J Inflamm Res. (2023) 16:4347–61. doi: 10.2147/JIR.S419347
8. Borba V, Carrera-Bastos P, Zandman-Goddard G, Lucia A, and Shoenfeld Y. Prolactin’s paradox: Friend, foe, or both in immune regulation? Autoimmun Rev. (2024) 23:103643. doi: 10.1016/j.autrev.2024.103643
9. Borba VV, Zandman-Goddard G, and Shoenfeld Y. Prolactin and autoimmunity: The hormone as an inflammatory cytokine. Best Pract Res Clin Endocrinol Metab. (2019) 33:101324. doi: 10.1016/j.beem.2019.101324
10. Zhou Y, Gong J, Deng X, Shen L, and Liu L. Novel insights: Crosstalk with non-puerperal mastitis and immunity. Front Immunol. (2024) 15:1431681. doi: 10.3389/fimmu.2024.1431681
11. Dilaveri C, Degnim A, Lee C, DeSimone D, Moldoveanu D, and Ghosh K. Idiopathic granulomatous mastitis. Breast J. (2024) 2024:6693720. doi: 10.1155/2024/6693720
12. Oltean HN, Soliman AS, Omar OS, Youssef TF, Karkouri M, Abdel-Aziz A, et al. Risk factors for chronic mastitis in morocco and egypt. Int J Inflam. (2013) 2013:184921. doi: 10.1155/2013/184921
13. Uysal E, Soran A, and Sezgin E. Granulomatous Mastitis Study Group. Factors related to recurrence of idiopathic granulomatous mastitis: What do we learn from a multicentre study? ANZ J Surg. (2018) 88:635–9.
14. Co M, Cheng VCC, Wei J, Wong SCY, Chan SMS, Shek T, et al. Idiopathic granulomatous mastitis: A 10-year study from a multicentre clinical database. Pathology. (2018) 50:742–7. doi: 10.1016/j.pathol.2018.08.010
15. Zeng Y, Wang M, Gao X, Zhang D, Fu N, Zhao W, et al. Clinical characteristics of patients with granulomatous lobular mastitis associated with corynebacterium parakroppenstedtii infection and drug sensitivity analysis of the isolated strains. Ann Clin Microbiol Antimicrob. (2024) 23:95. doi: 10.1186/s12941-024-00755-7
16. Ge T, Sun P, Feng X, Gao X, Gao S, Wang T, et al. Clinical features and risk factors of bilateral granulomatous lobular mastitis. Med (Baltimore). (2024) 103:e37854. doi: 10.1097/MD.0000000000037854
17. Zeng Y, Zhang D, Fu N, Zhao W, Huang Q, Cui J, et al. Risk factors for granulomatous mastitis and establishment and validation of a clinical prediction model (nomogram). Risk Manag Healthc Policy. (2023) 16:2209–22. doi: 10.2147/RMHP.S431228
18. Esmaeil NK, Salih AM, Hammood ZD, Pshtiwan LRA, Abdullah AM, Kakamad FH, et al. Clinical, microbiological, immunological and hormonal profiles of patients with granulomatous mastitis. BioMed Rep. (2023) 18:41. doi: 10.3892/br.2023.1624
19. Nikolaev A, Blake CN, and Carlson DL. Association between hyperprolactinemia and granulomatous mastitis. Breast J. (2016) 22:224–31. doi: 10.1111/tbj.12552
20. Huang Y and Wu H. A retrospective analysis of recurrence risk factors for granulomatous lobular mastitis in 130 patients: More attention should be paied to prolactin level. Ann Palliat Med. (2021) 10:2824–31. doi: 10.21037/apm-20-1972
21. Tian C, Han X, Liu Z, Lv X, and Ning P. Management of granulomatous lobular mastitis and risk factors associated with recurrence. World J Surg. (2022) 46:2706–14. doi: 10.1007/s00268-022-06687-7
22. Cui L, Sun C, Guo J, Zhang X, and Liu S. Pathological manifestations of granulomatous lobular mastitis. Front Med. (2024) 11:1326587. doi: 10.3389/fmed.2024.1326587
23. Riaz B and Sohn S. Neutrophils in inflammatory diseases: Unraveling the impact of their derived molecules and heterogeneity. Cells. (2023) 12:2621. doi: 10.3390/cells12222621
24. Rasmi Y, Jalali L, Khalid S, Shokati A, Tyagi P, Ozturk A, et al. The effects of prolactin on the immune system, its relationship with the severity of COVID-19, and its potential immunomodulatory therapeutic effect. Cytokine. (2023) :169:156253. doi: 10.1016/j.cyto.2023.156253
25. Legorreta-Haquet MV, Santana-Sánchez P, Chávez-Sánchez L, and Chávez-Rueda AK. The effect of prolactin on immune cell subsets involved in SLE pathogenesis. Front Immunol. (2022) 13:1016427. doi: 10.3389/fimmu.2022.1016427
26. Orzheshkovskyi VV and Trishchynska MA. Ceruloplasmin: Its role in the physiological and pathological processes. Neurophysiology. (2019) 51:141–9. doi: 10.1007/s11062-019-09805-9
27. Chen F, Han B, Meng Y, Han Y, Liu B, Zhang B, et al. Ceruloplasmin correlates with immune infiltration and serves as a prognostic biomarker in breast cancer. Aging (Albany NY). (2021) 13:20438–67. doi: 10.18632/aging.203427
28. Khalaf MY, Mohammed AA, Mosa AA, Arif SH, and Mustafa JA. The correlation of antioxidant levels of breast cancer. Med (Baltimore). (2021) 100:e26878. doi: 10.1097/MD.0000000000026878
29. Liu Y, Zhu J, Xu L, Wang B, Lin W, and Luo Y. Copper regulation of immune response and potential implications for treating orthopedic disorders. Front Mol Biosci. (2022). Available online at: https://www.frontiersin.org/journals/molecular-biosciences/articles/10.3389/fmolb.2022.1065265/full (Accessed April 1, 2025).
30. Strecker D, Mierzecki A, and Radomska K. Copper levels in patients with rheumatoid arthritis. Ann Agric Environ Med. (2013) 20:312–6.
31. Tomio A, Schust DJ, Kawana K, Yasugi T, Kawana Y, Mahalingaiah S, et al. Prolactin can modulate CD4+ T-cell response through receptor-mediated alterations in the expression of T-bet. Immunol Cell Biol. (2008) 86:616–21. doi: 10.1038/icb.2008.29
32. Zheng B, Song J, Lu M, Chen C, and Sun S. Current research describing the role of CD4+ T lymphocyte subsets in the pathogenesis of granulomatous lobular mastitis. J Invest Surg. (2022) 35:1790–5. doi: 10.1080/08941939.2022.2090035
33. Krausgruber T, Redl A, Barreca D, Doberer K, Romanovskaia D, Dobnikar L, et al. Single-cell and spatial transcriptomics reveal aberrant lymphoid developmental programs driving granuloma formation. Immunity. (2023) 56:289–306. doi: 10.1016/j.immuni.2023.01.014
34. Waite JC and Skokos D. Th17 response and inflammatory autoimmune diseases. Int J Inflam. (2012) 2012:819467. doi: 10.1155/2012/819467
35. Cui H, Wang N, Li H, Bian Y, Wen W, Kong X, et al. The dynamic shifts of IL-10-producing Th17 and IL-17-producing treg in health and disease: A crosstalk between ancient “yin-yang” theory and modern immunology. Cell Commun Signal. (2024) 22:99. doi: 10.1186/s12964-024-01505-0
36. Palomares O, Martín-Fontecha M, Lauener R, Traidl-Hoffmann C, Cavkaytar O, Akdis M, et al. Regulatory T cells and immune regulation of allergic diseases: Roles of IL-10 and TGF-β. Genes Immun. (2014) 15:511–20. doi: 10.1038/gene.2014.45
37. Dixon AM and Roy S. Role of membrane environment and membrane-spanning protein regions in assembly and function of the class II major histocompatibility complex. Hum Immunol. (2019) 80:5–14. doi: 10.1016/j.humimm.2018.07.004
38. Yan L, Wang J, Cai X, Liou YC, Shen HM, Hao J, et al. Macrophage plasticity: Signaling pathways, tissue repair, and regeneration. MedComm. (2024) 5:e658. doi: 10.1002/mco2.658
39. Koksal H, Vatansev H, Artac H, and Kadoglou N. The clinical value of interleukins-8, -10, and -17 in idiopathic granulomatous mastitis. Clin Rheumatol. (2020) 39:1671–7. doi: 10.1007/s10067-020-04925-8
40. Cabioglu N, Cetin Aktas E, Emiroglu S, Tukenmez M, Ozkurt E, Muslumanoglu M, et al. Ozone therapy restores immune dysfunction in refractory idiopathic granulomatous mastitis as a novel potential therapeutic approach. Cell Biol Int. (2023) 47:228–37. doi: 10.1002/cbin.11953
41. Santana-Sánchez P, Vaquero-García R, Legorreta-Haquet MV, Chávez-Sánchez L, and Chávez-Rueda AK. Hormones and B-cell development in health and autoimmunity. Front Immunol. (2024) 15. Available online at: https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2024.1385501/full (Accessed March 20, 2025).
42. Ewerman L, Landberg E, Hellberg S, Hovland M, Sundin A, Jenmalm MC, et al. Immunomodulating effects depend on prolactin levels in patients with hyperprolactinemia. Horm Metab Res. (2020) 52:228–35. doi: 10.1055/a-1126-4272
43. Borba VV, Zandman-Goddard G, and Shoenfeld Y. Prolactin and autoimmunity. Front Immunol. (2018) 9:73. doi: 10.3389/fimmu.2018.00073
44. Lou Y, Xu H, Lu Z, Wang B, and Liu X. Immune regulation: A new strategy for traditional Chinese medicine-based treatment of granulomatous lobular mastitis. Front Immunol. (2024) 15:1494155. doi: 10.3389/fimmu.2024.1494155
45. Seif F, Khoshmirsafa M, Aazami H, Mohsenzadegan M, Sedighi G, and Bahar M. The role of JAK-STAT signaling pathway and its regulators in the fate of T helper cells. Cell Commun Signal. (2017) 15:23. doi: 10.1186/s12964-017-0177-y
46. Williams A, Hossack DJ, Thompson N, Sim YE, Wilson C, Schuch V, et al. Elevated levels of exogenous prolactin promote inflammation at the maternal-fetal interface via the JAK2/STAT5B signaling axis. Front Immunol. (2024) 15:1496610. doi: 10.3389/fimmu.2024.1496610
Keywords: granulomatous lobular mastitis, prolactin, immune, inflammatory, pathogenesis, ceruloplasmin
Citation: Liu B, Qu W, Feng Y, Wu X, Feng J, Gao Q, Shao S, Sun J, Wan H and Chen W (2025) Exploring the interplay of prolactin and bromocriptine on serum markers in granulomatous lobular mastitis. Front. Immunol. 16:1608875. doi: 10.3389/fimmu.2025.1608875
Received: 09 April 2025; Accepted: 21 July 2025;
Published: 29 August 2025.
Edited by:
Rosaria Talarico, ERN ReCONNET Coordination Team, ItalyReviewed by: Ruo Wang, Shanghai Jiao Tong University, China
Maria Victoria Legorreta-Haquet, Mexican Social Security Institute, Mexico
Yulian Yin, Shanghai University of Traditional Chinese Medicine, China
Copyright © 2025 Liu, Qu, Feng, Wu, Feng, Gao, Shao, Sun, Wan and Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Weiping Chen, Y2hlbndlaXBpbmdAbXVzdC5lZHUubW8=; Hua Wan, ZHJ3YW5odWFAMTYzLmNvbQ==
†These authors share first authorship
 Binheng Liu
Binheng Liu Wenchao Qu2†
Wenchao Qu2† Yi Feng
Yi Feng Xueqing Wu
Xueqing Wu