- 1Division of Dermatology, Department of Medicine, Khon Kaen University, Khon Kaen, Thailand
- 2Department of Internal Medicine, Texas Tech University Health Sciences Center, Lubbock, TX, United States
- 3Division of Infectious Disease and Tropical Medicine, Department of Medicine, Khon Kaen University, Khon Kaen, Thailand
- 4Division of Dermatology, Department of Internal Medicine, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
- 5Division of Infectious Diseases and Tropical Medicine, Department of Internal Medicine, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
- 6Center for Clinical Epidemiology and Clinical Statistics, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
- 7Department of Biomedical Informatics and Clinical Epidemiology, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
- 8Pharmacoepidemiology and Statistics Research Center (PESRC), Faculty of Pharmacy, Chiang, Mai University, Chiang Mai, Thailand
Introduction: Cutaneous manifestations in adult-onset immunodeficiency (AOID) resulting from anti-interferon-gamma autoantibody (AIGA) are prevalent and can be classified into infective and reactive disorders. To date, no clinical studies have specifically examined pustular reaction in patients with AOID. This study aimed to provide an original characterization of the clinical manifestations associated with pustular reaction in AOID and to compare these features with those observed in a clinically similar entity, generalized pustular psoriasis (GPP).
Methods: A retrospective study was conducted between January 2014 and June 2023 at the dermatology clinics of Maharaj Nakorn Chiang Mai Hospital, Chiang Mai and Srinagarind Hospital, Khon Kaen, Thailand. The study included adult patients diagnosed with AOID presenting with pustular reaction, defined as pinhead-sized pustules on an erythematous base occurring any area of skin, as well as those diagnosed with GPP. Cases with pustular drug eruption and with non-sterile pustules were excluded. Data analysis was performed using both univariable and multivariable statistical methods.
Results: Total of 64 patients diagnosed with AOID who subsequently developed a pustular reaction were included in the study. Clinically, the cutaneous manifestations were characterized by discrete, pinhead-sized pustules distributed on erythematous bases, predominantly affecting the trunk and extremities. A concomitant infection was identified in 54 patients (84.4%). Rapid-growing mycobacteria represented the most frequently identified pathogens, with lymph nodes being the most commonly involved anatomical site. Comparative analysis with a cohort of 77 patients diagnosed with GPP elucidated distinct clinical hallmarks differentiating the two entities. Lymphadenopathy, hepatomegaly, and splenomegaly emerged as distinguishing features more frequently associated with AOID-related pustular reaction. In contrast, the presence of geographic tongue and nail involvement was more characteristic of GPP. In instances where these pathognomonic features were absent, a multivariable predictive model was developed to aid in diagnostic differentiation. This model incorporated the presence of concomitant infections, elevated serum globulin levels, and increased alkaline phosphatase levels.
Conclusion: Among patients presenting with sterile pustular eruptions, the presence of lymphadenopathy, hepatomegaly, and splenomegaly served as perfect clinical predictors of an underlying pustular reaction associated with AOID. In cases where these hallmark features were absent, a predictive algorithm incorporating the presence of a concomitant infections, serum globulin concentration, and alkaline phosphatase level demonstrated robust utility in estimating the likelihood of AOID-associated pustular reaction.
1 Introduction
Adult-onset immunodeficiency (AOID) due to anti-interferon-gamma autoantibodies (AIGA) is an increasingly recognized form of acquired immunodeficiency, frequently associated with cutaneous manifestations (1, 2). AOID is characterized by the presence of high-titer, neutralizing autoantibodies directed against interferon-gamma (IFN-γ), a key cytokine in host defense against intracellular pathogens. The condition primarily affects individuals of Southeast and East Asian descent, implicating potential genetic predisposition in its pathogenesis (1, 3). Nevertheless, the typical age of onset—around the fifth decade of life—suggests that environmental factors may also contribute significantly to disease development.
The functional impairment of IFN-γ signaling results in marked susceptibility to opportunistic infections (OIs), mirroring the immunological deficits observed in acquired immunodeficiency syndrome (AIDS). The most frequently encountered OIs in AOID include non-tuberculous mycobacteria (NTM), dimorphic fungi, and non-typhoidal Salmonella species. Lymph nodes are the most commonly affected anatomical sites, followed by pulmonary, osseous, and cutaneous involvement (4).
Cutaneous manifestations are not uncommon in AOID and may serve as important clinical clues to the underlying immunologic defect. These dermatologic findings can be broadly classified into two categories: infectious and reactive dermatoses. Infectious skin conditions include viral infections such as herpes simplex and varicella-zoster virus, cutaneous involvement by NTM, and invasive dimorphic fungal infections, notably Talaromyces marneffei and Histoplasma capsulatum (5). Among the reactive dermatoses, neutrophilic dermatoses are the most prominent, with Sweet syndrome being the most frequently reported presentation. Other neutrophilic conditions observed in this context include pustular eruptions and panniculitis (6–11).
Pustular eruptions encompass a broad differential diagnosis, including classic entities such as generalized pustular psoriasis (GPP), drug-induced pustular eruptions, and subcorneal pustular dermatosis (12). Despite the growing recognition of neutrophilic dermatoses as reactive cutaneous manifestations in AOID, pustular eruptions in this context remain inadequately characterized. Key aspects—including detailed rash morphology, systemic associations, laboratory parameters, and patterns of concomitant infection— are not yet fully elucidated. A clearer understanding of these features is essential to improve diagnostic accuracy and inform optimal management strategies. This study aimed to comprehensively characterize the clinical profiles of AOID patients presenting with pustular eruptions and to compare these findings with those observed in patients with GPP. Furthermore, we propose a novel predictive scoring system to aid in the diagnosis of pustular reaction associated with AOID, offering a clinically applicable tool with high discriminative performance.
2 Material and methods
2.1 Subjects and study design
We performed a retrospective review of clinical data in adult patients (aged >18 years old) with pustular reaction in AOID and GPP in the dermatology clinics of Maharaj Nakorn Chiang Mai Hospital, Chiang Mai, and Srinagarind Hospital, Khon Kaen, Thailand, from January 2014 to June 2023. The diagnosis of AIGA associated with AOID was made according to the following criteria: (i) the positive AIGA using dot enzyme-linked immunosorbent assay (ELISA) (Maharaj Nakorn Chiang Mai Hospital) (13) or via inhibitory ELISA (Srinagarind Hospital) (14); (ii) exclusion of other immunocompromised conditions, including human immunodeficiency virus (HIV), malignancies, or immunosuppressive therapy; and (iii) presence of typical concomitant opportunistic infection resulting from impaired IFN-γ function. Diagnosis of GPP was made by board-certified dermatologists followed the definition proposed in the European Consensus Statement (15). We excluded cases with pustular drug eruption and with non-sterile pustules (evidence of microorganism in pustules from microbiological test). Cases with pustular reaction concomitant with Sweet syndrome were excluded.
We conducted a review of medical records for patients with pustular reactions who attended the dermatology clinic. Clinical manifestations were carefully assessed by the investigators to confirm the diagnosis of pustular reaction in the context of AOID. Demographic data, rash morphology, associated systemic findings, and relevant laboratory results were documented.
Concomitant opportunistic infections in patients with AOID were recorded, including the type of microorganisms, organ involvement, and their temporal relationship to the onset of the pustular reaction. The onset of concomitant infections was defined as the date on which the first clinical symptom or sign of infection—such as fever or lymphadenopathy—was noted.
For patients who experienced multiple episodes of pustular reactions, all episodes were documented. Data collection included both static demographic information for each patient and dynamic records detailing each episode of pustular reaction.
2.2 Ethical approval
This study was approved by the Ethics Committee of the Faculty of Medicine, Chiang Mai University (MED-2566-09405) and Khon Kaen University Ethics Committee for Human Research (HE661088).
2.3 Statistical analysis
A statistical analysis was conducted using StataNow 18.5 (StataCorp, College Station, Texas, USA) and R version 4.1.2. Data cleaning procedures, including the handling of missing values, removal of duplicates, and standardization of units, were performed prior to analysis. Descriptive statistics included the mean with standard deviation (SD) for continuous data and percentages for categorical data. Independent t-tests were used to compare continuous variables, while Fisher’s exact test was employed for categorical data. Statistical significance was defined as a p-value of less than 0.05.
Variables with more than 50% missing data were excluded from model development. For the development of a predictive equation to estimate the probability of pustular reaction in AOID, variables with significant associations (p< 0.05) in the univariable analysis were included in the multivariable logistic regression. Due to the binary outcome and sparse data, Firth’s logistic regression was used. Missing data were handled with multiple imputations using chained equations (MICE). Variables identified as perfect predictors of the outcome were excluded from model development to avoid exacerbating the sparse data problem. Additionally, patients with these pathognomonic features were not included, as their diagnosis could be easily made, and they were not the target population for our model. We applied backward stepwise elimination, beginning with the full model and sequentially removing variables with p-values greater than 0.05, to identify the most parsimonious set of predictors that remained statistically significant. A predictive model to estimate the probability of pustular reaction was then derived. The area under the receiver operating characteristic (AuROC) curve was calculated to assess discriminative ability. Calibration was evaluated using a calibration plot, expected-to-observed ratio (E:O), calibration slope, and calibration-in-the-large (CITL). The optimal probability cut-off points were determined using Youden’s method.
3 Results
3.1 Clinical profile of patients with pustular reaction in AOID and GPP
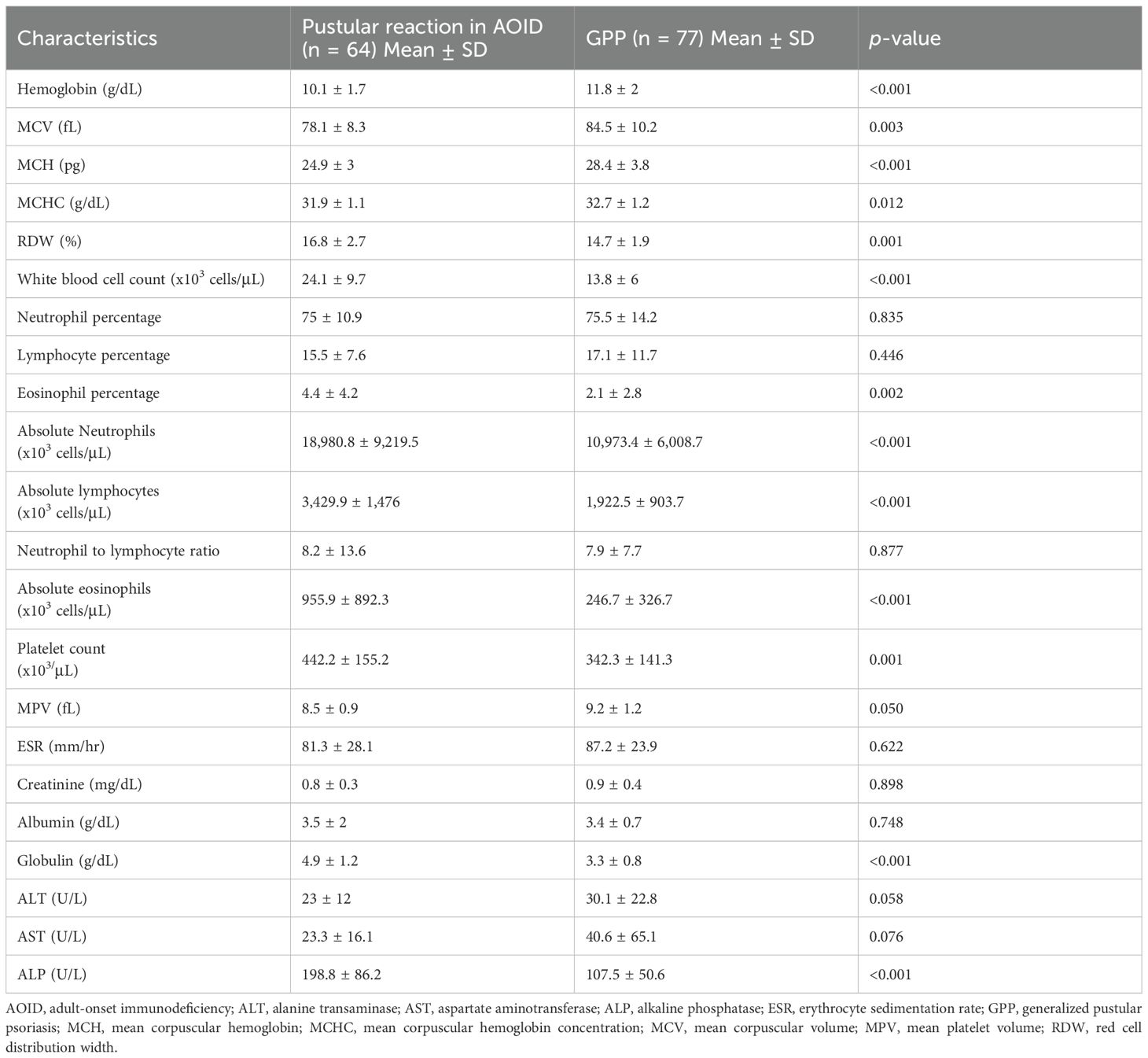
A total of 64 AOID patients with pustular reaction were included in the study (Table 1, Supplementary Figure S1). There was a slight predominance of females among the patients, with a mean age of 53.2 years. Recurrence of pustular reaction was observed in 18 patients (28.1%). Discrete, pinhead-sized pustules on an erythematous base are characteristic, occasionally coalescing (Figure 1). Generalized involvement was found in approximately 60% of the patients. Large confluent pustules, resembling a “lake of pus”, were noted in 2 patients (Figure 2), while an annular configuration was observed in 1 patient (Figure 3). When compared to a control group of 77 patients with GPP, confluent pustules, annular configuration, geographic tongue, and nail involvement were less frequently observed in pustular reaction of AOID.
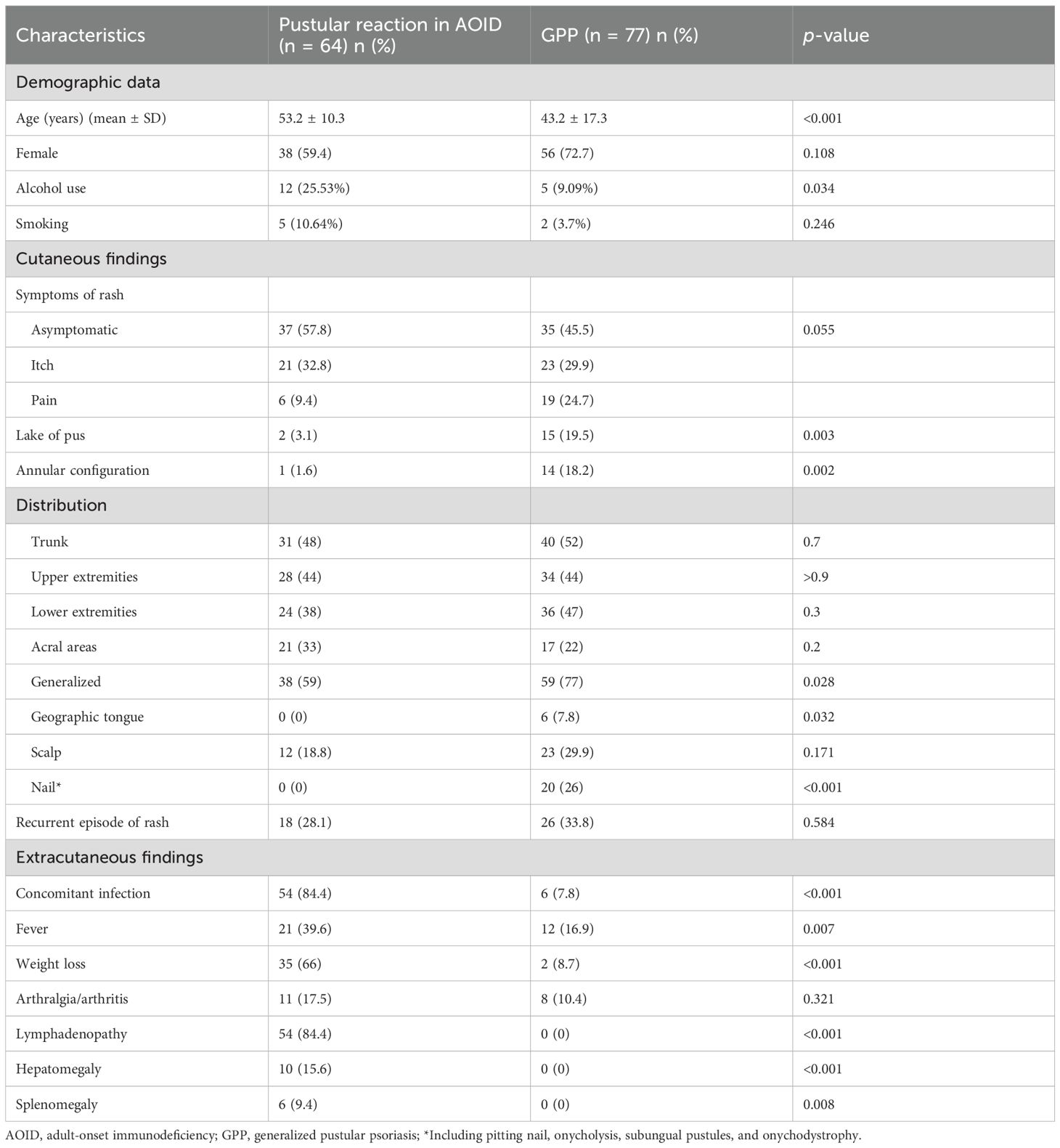
Table 1. Demographics and clinical characteristics of patients with pustular reaction in AOID and GPP.
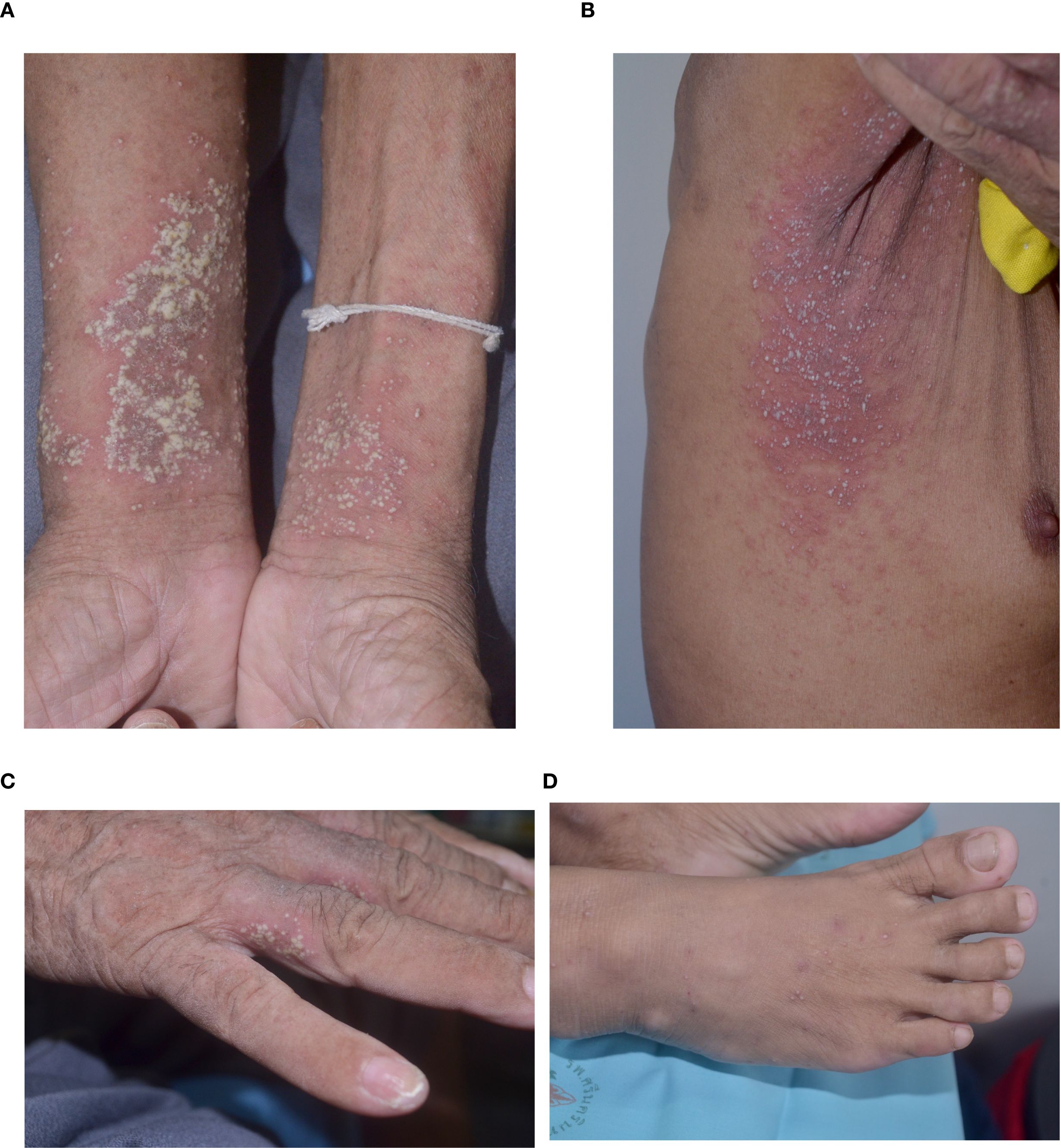
Figure 1. Morphology of lesions in pustular reaction in adult-onset immunodeficiency due to anti-interferon-gamma autoantibody (A) volar forearms; (B) axilla; (C) hand; (D) foot.
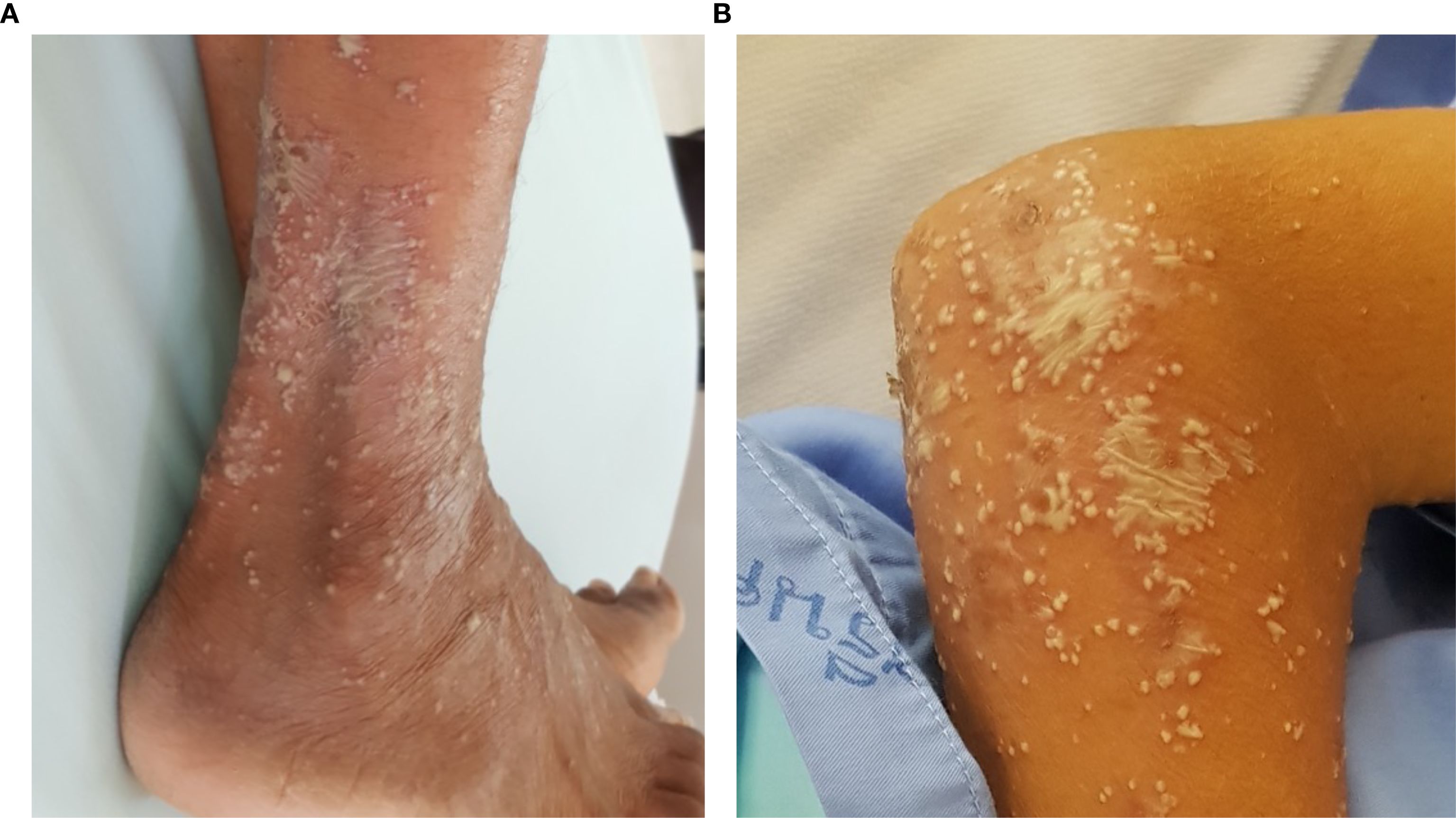
Figure 2. Lake of pus mimicking generalized pustular psoriasis in 2 patients with pustular reaction in adult-onset immunodeficiency due to anti-interferon-gamma autoantibody (A) leg; (B) forearm.
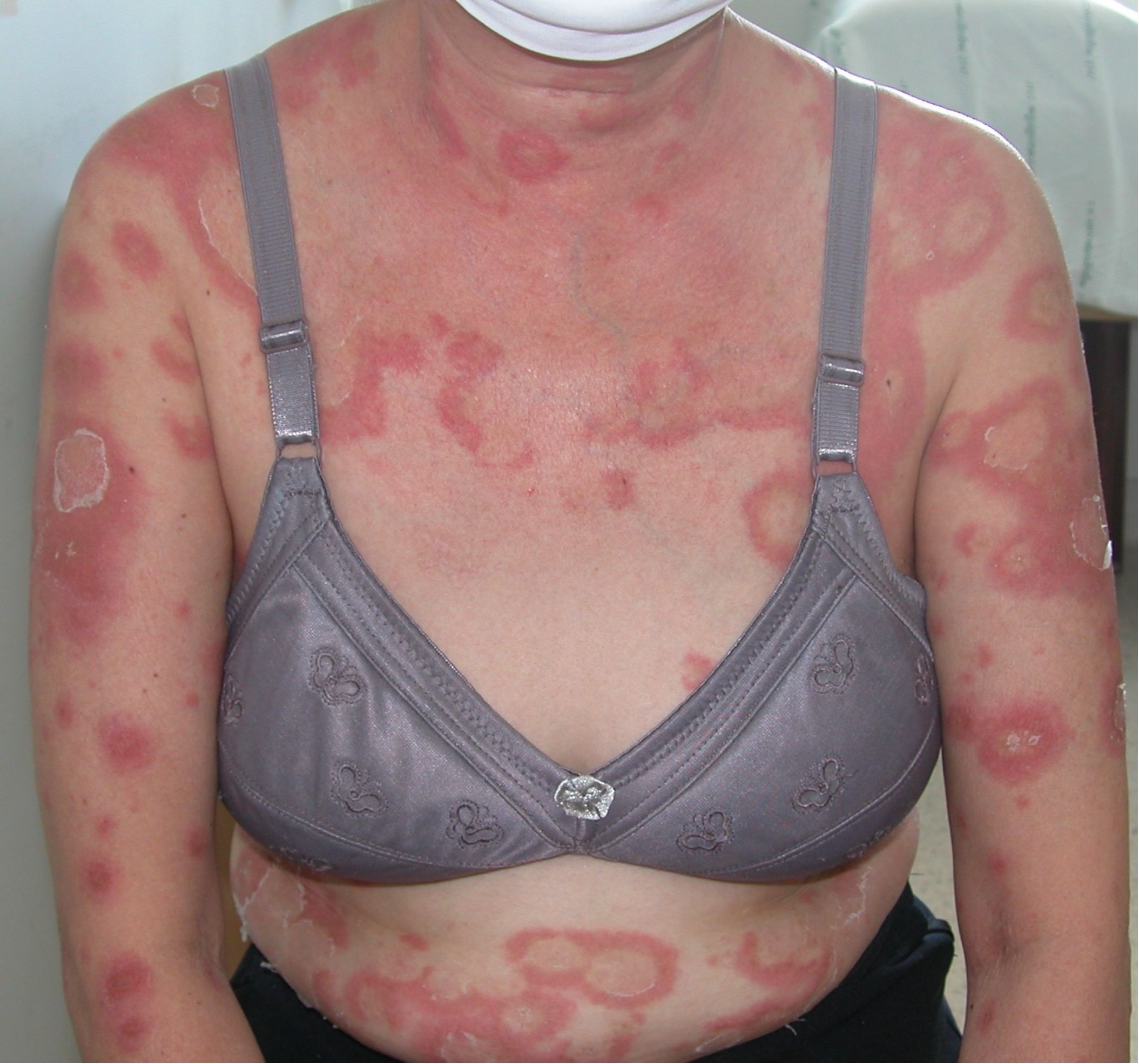
Figure 3. Annular configuration was observed in 1 patient with pustular reaction in adult-onset immunodeficiency due to anti-interferon-gamma autoantibody.
The most common extracutaneous findings were lymphadenopathy, followed by weight loss, fever, arthralgia/arthritis and hepatomegaly. Compared to GPP, all of these findings, with the exception of arthralgia/arthritis, were significantly more prevalent in the pustular reaction associated with AOID.
Interestingly, we identified several pathognomonic clinical features that serve as strong predictors for each disease. Lymphadenopathy, hepatomegaly, and splenomegaly are indicative of the pustular reaction associated with AOID, while geographic tongue and nail involvement are characteristic of GPP. A total of 61 patients in the cohort did not exhibit any of these pathognomonic features.
3.2 Laboratory profile of patients with pustular reaction in AOID and GPP
Anemia, leukocytosis, eosinophilia, thrombocytosis, hyperglobulinemia, and elevated alkaline phosphatase were significantly more common in the pustular reaction associated with AOID compared to GPP (Table 2).
3.3 Concomitant infections of patients with pustular reaction in AOID and GPP
Concomitant infections were identified in 54 cases (84.4%) among AOID patients with pustular reaction (Table 3). A total of 79 episodes of pustular reaction associated with concomitant infections were documented. In the majority of episodes (n = 64), the infection preceded the onset of the pustular reaction, with a median interval of 31 days. Conversely, in 10 episodes, the infection occurred subsequent to the pustular reaction, with a median interval of 18 days following the onset of the dermatologic manifestation.
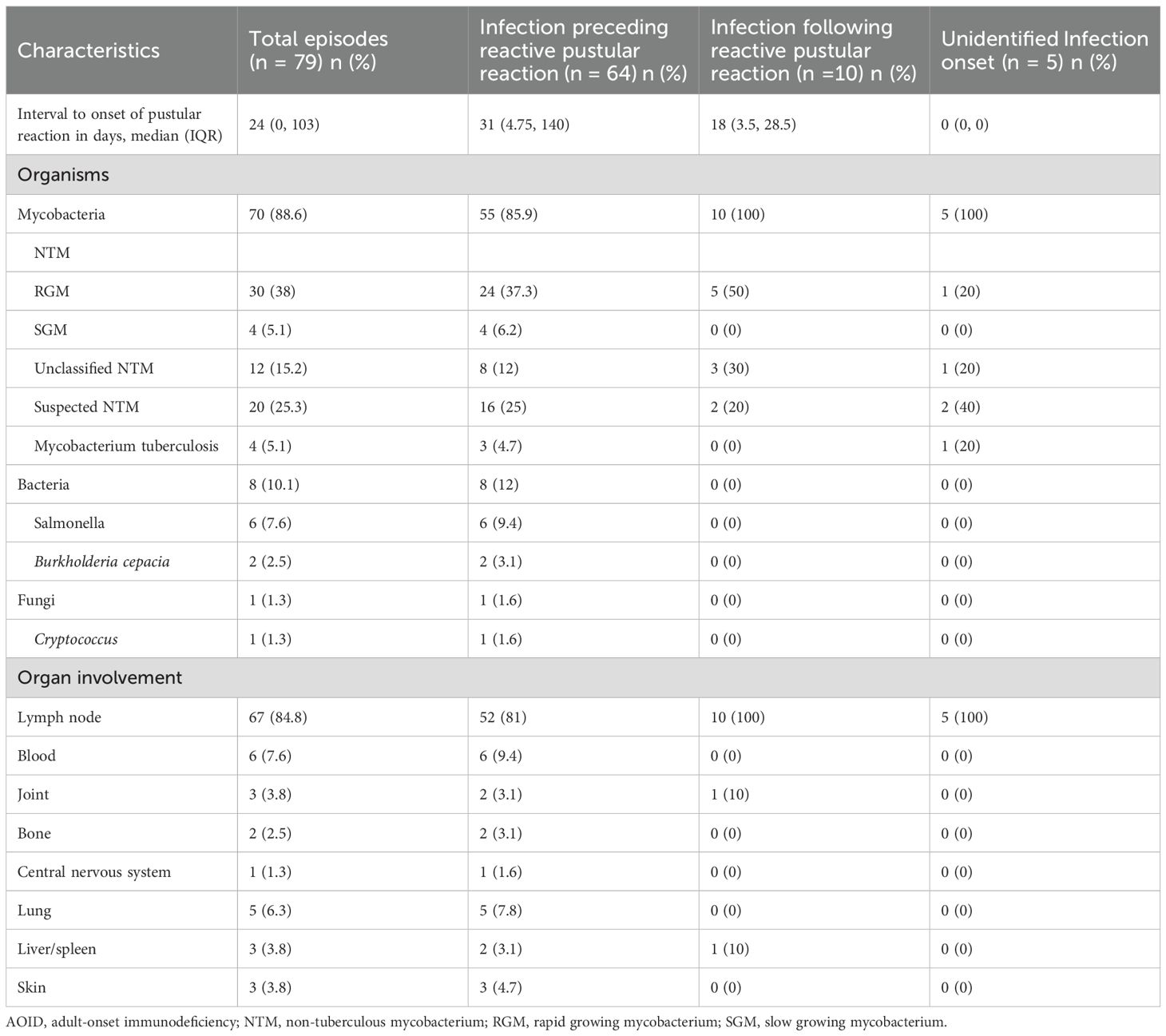
Table 3. Concomitant infections related to onset of pustular reaction in AOID patients (all episodes).
Among all documented episodes, the most frequently identified concomitant infections were caused by rapidly growing mycobacteria. The three most commonly involved organ systems were the lymph nodes, bloodstream, and lungs.
In patients with GPP, concomitant infections were observed in 6 of 77 individuals. When analyzed by episode, infections were implicated as potential triggers in 11 of 149 episodes of GPP flares. Identified infectious triggers included respiratory viral infections (such as common cold and SARS-CoV-2), bacterial infections (including erysipelas and Staphylococcus aureus pyoderma in 2 cases), and chronic hepatitis C virus infection.
3.4 Predictive scoring system
After removal of pathognomonic variables, the rest variables with significant association (p< 0.05) from univariable logistic regression were added into multivariable logistic regression as a full model (Table 4). After stepwise backward elimination, the remaining significant variables in final reduced model were concomitant infection (OR 57.98; 95% CI 12.45, 270.04), globulin level in g/dL (OR 6.39; 95% CI 2.16, 18.91) and each 10 U/L increment of alkaline phosphatase level (OR 1.24; 95% CI 1.07, 1.45). The formula to predict probability of pustular reaction is as follows:
Where CI is presence of concomitant infection, GLOB is globulin level (g/dL) and ALP is alkaline phosphatase level (U/L). A calculator for this formula is available at the following link: https://cmu.to/pustular-risk.
When applying this equation to all 141 patients, we found the outstanding discriminative ability with AuROC 0.97; 95% CI 0.94, 1.00 and almost perfect calibration curve (Figure 4). The best cut point for the predicted risk was 60%, with a sensitivity of 92% and a specificity of 95%. In comparison, a model including only concomitant infection yielded a lower AuROC of 0.88 (95% CI: 0.83–0.94), indicating that the inclusion of globulin and alkaline phosphatase improved the model’s predictive accuracy (P<0.001).
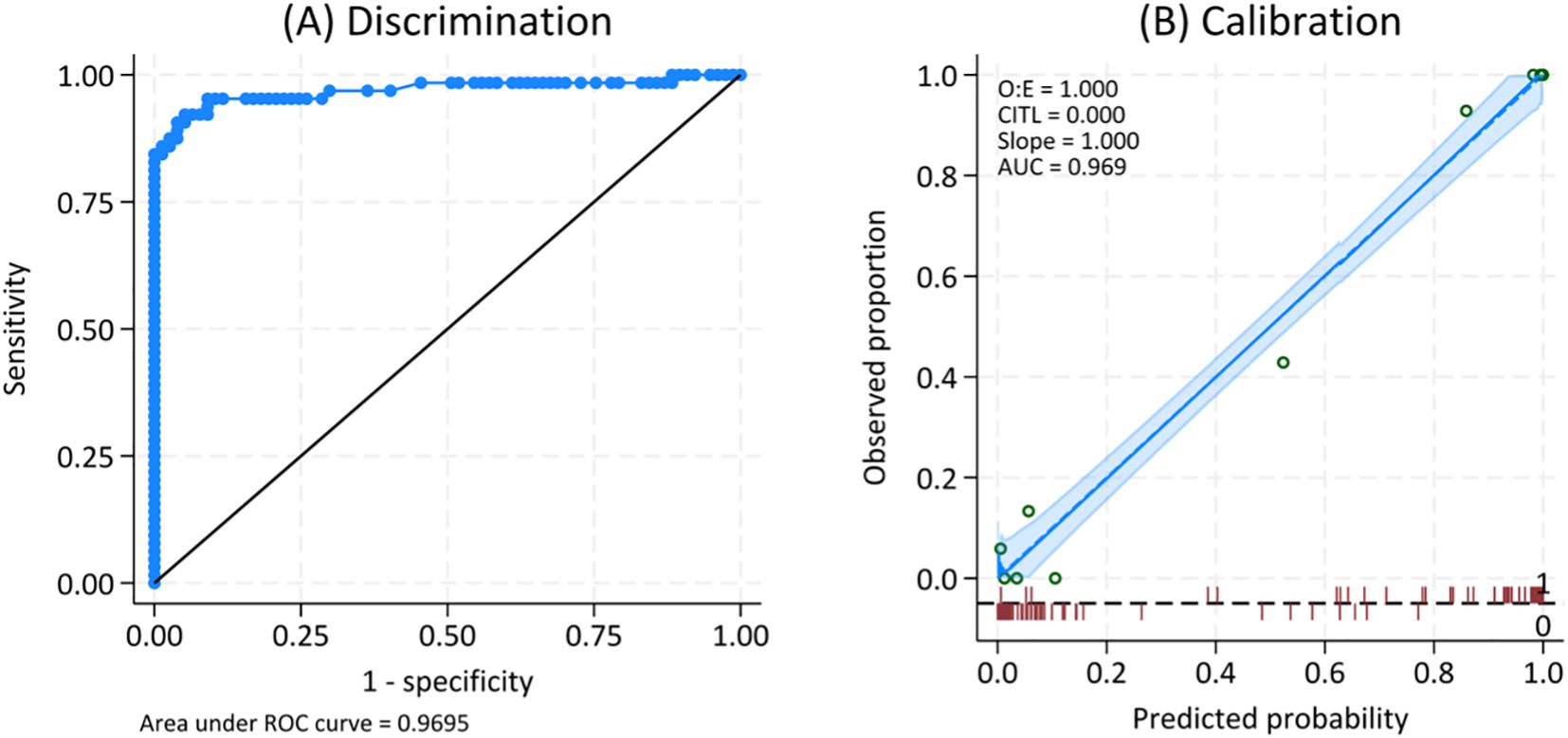
Figure 4. ROC (A) and calibration curve (B) for the predicted probability of pustular reaction in adult-onset immunodeficiency due to anti-interferon-gamma autoantibody.
Additionally, when this equation was applied to 61 patients lacking pathognomonic variables, it demonstrated a discriminative ability with AuROC 0.78; 95% CI: 0.55–1.00. The optimal cut-off point for the predicted risk was 32%, yielding a sensitivity of 62% and a specificity of 91%.
4 Discussion
To date, pustular reaction in AOID has not been the focus of a dedicated investigation, in contrast to Sweet syndrome, which is more extensively documented. This study represents the first attempt to delineate the clinical features, systemic associations, and diagnostic considerations of this distinct neutrophilic dermatosis within the AOID spectrum.
The pustular reaction observed in AOID is clinically characterized by an acute eruption of pinhead-sized pustules on an erythematous base. Histopathological examination typically reveals subcorneal and/or intraepidermal pustules (9). To date, the pathogenesis of pustular eruptions in the context of AOID remains poorly elucidated. However, it is plausible that these cutaneous manifestations represent a subset within the broader spectrum of reactive neutrophilic dermatoses. Such eruptions are likely driven by aberrant crosstalk between the innate and adaptive immune systems, with particular involvement of the T helper 1 and T helper 17 pathways. The signature cytokines of these respective axes are IFN-γ and interleukin-17 (16). Despite increasing recognition of this entity, a standardized nomenclature has yet to be established. Various terms have been employed in prior literature to describe this reaction, including exanthematous pustulosis, acute generalized exanthematous pustulosis (AGEP), pustular psoriasis, and generalized pustular reaction (1, 6, 7, 9, 11, 17).
However, the term AGEP may be suboptimal for describing pustular reaction in the context of AOID for several reasons. Firstly, in our cohort, only 51 of 94 episodes (54.3%) exhibited a truly generalized distribution, thereby challenging the appropriateness of the term “generalized” in this clinical setting. Furthermore, over 90% of AGEP cases reported in the literature are attributed to drug-induced etiologies, with viral infections representing the predominant cause among the non-drug-related cases. Importantly, to date, no instances of AGEP secondary to Mycobacterium infection have been documented, further underscoring the potential limitations of applying this nomenclature in AOID-associated presentations.
We observed that a considerable number of AOID cases presenting with concomitant infection were subsequently treated with antibiotics or anti-tuberculosis agents, after which pustular eruptions developed. These eruptions were frequently diagnosed as drug-induced AGEP. However, a significant proportion of such cases may represent pustular reaction intrinsic to AOID rather than true drug-induced AGEP. This diagnostic ambiguity likely contributes to the under-recognition and underreporting of pustular reaction associated with AOID.
Differentiating the pustular reaction associated with AOID from GPP remains a clinical challenge. Dermatologic assessment traditionally emphasizes features such as annular configuration and the presence of a “lake of pus” as suggestive of GPP. However, these features are not pathognomonic. In our cohort, one case of AOID-associated pustular reaction exhibited an annular configuration, and two cases demonstrated a lake of pus, indicating that these findings may also occur in the context of AOID. Thus, reliance on these clinical signs alone may be insufficient for accurate diagnostic distinction between the two entities. Recently, mutations in SERPINA1, SERPINB3, and TGFBR2 have been identified in both patients GPP and those with AOID-associated pustular reaction, suggesting a potentially shared pathogenic mechanism between the two conditions (18–20) Based on these findings, we propose that GPP-like lesions may represent a clinical variant of the pustular reaction observed in AOID.
In patients presenting with a pustular eruption, following the exclusion of drug-induced eruptions and pyogenic skin infections, several distinguishing clinical clues may aid in differentiating the pustular reaction associated with AOID from GPP. Findings from this study underscore the importance of a thorough physical examination. Specifically, targeted evaluation of the nails, tongue, lymph nodes, liver, and spleen is recommended, as nail abnormalities and geographic tongue are more indicative of GPP, whereas the presence of lymphadenopathy and hepatosplenomegaly favors a diagnosis of AOID-associated pustular reaction. In cases where these clinical features are absent or inconclusive, a predictive scoring system may be utilized to estimate the likelihood of AOID-associated pustular disease, thereby aiding diagnostic decision-making. The presence of concomitant infection, along with elevated serum globulin and alkaline phosphatase levels, further strengthens the clinical suspicion of AOID-associated pustular reaction. Moreover, recently published studies have identified specific human leukocyte antigen (HLA) alleles—most notably HLA-DPB105:01 and HLA-DRB115:02—as potential genetic biomarkers capable of distinguishing pustular eruptions associated with AOID from GPP (21).
Involvement of the nails and tongue in GPP reflects systemic inflammation characteristic of mucocutaneous psoriasis. Geographic tongue, in particular, shares histopathological features with cutaneous psoriasis. Notably, the higher prevalence of pustules of Kogoj observed in geographic tongue further supports its association with pustular psoriasis and underscores its potential as a clinical marker in differentiating GPP from other pustular dermatoses (22).
The involvement of the reticuloendothelial system, including lymphadenopathy and hepatosplenomegaly, correlates with the presence of concomitant infections in AOID-associated pustular reaction, emphasizing its role as a critical indicator. In conjunction with elevated globulin and alkaline phosphatase levels, this systemic involvement supports the use of these markers as key predictors in predictive models for identifying AOID-related pustular reaction. Previous studies have demonstrated significantly higher alkaline phosphatase levels in AOID patients compared to both healthy individuals and HIV-infected patients. Moreover, within the same cohort of AOID patients, alkaline phosphatase levels are notably higher during active infections compared to periods without infection, further underscoring its utility as a dynamic biomarker of disease activity (5).
Our findings regarding concomitant infection in AOID-associated pustular reaction are consistent with previous studies on AOID-associated Sweet syndrome, which also identified rapidly growing mycobacteria as the most prevalent pathogens (23). Based on the shared clinical and immunological context—namely, the occurrence of neutrophilic dermatoses in the setting of AOID—we propose a potential interaction between AOID, NTM infection, and the development of pustular reaction. This suggests a common pathogenic mechanism linking the presence of AIGA, microbial triggers, and neutrophilic skin inflammation.
Regarding the temporal relationship between concomitant infection onset and pustular reaction, most pustular reactions in AOID occurred after the onset of concomitant infection; however, a substantial number of cases presented before active infection. This finding highlights the importance of clinicians actively screening for concomitant infection in AOID patients experiencing skin flare-ups.
Currently, no established guidelines exist for the treatment of reactive dermatoses in this syndrome. The majority of reported treatments primarily rely on corticosteroids (9). Additionally, acitretin has emerged as an alternative therapeutic option for pustular reaction, demonstrating satisfactory outcomes in affected patients (24).
This study has some limitations. First, the study is inherently retrospective in nature, which may introduce recall bias or misinterpretation of data. This could affect the accuracy of patient histories and clinical assessments. Additionally, the prevalence of pustular reaction in AOID may be underestimated, as some cases may have been misdiagnosed as drug-induced AGEP, particularly in those with a history of drug exposure. However, in this study, we excluded cases with a clear history of drug-induced AGEP based on drug exposure and temporal associations. Future prospective studies focusing on distinguishing pustular reaction in AOID from AGEP may provide valuable insights. Thirdly, detailed histopathological data were not available, and such features may offer additional clues to aid in differentiating these two conditions. Finally, the algorithm developed in this study was derived exclusively from a cohort of patients with AOID and GPP. Its performance and specificity in distinguishing AOID-associated pustular reactions from other pustular dermatoses or pustular eruptions occurring in immunodeficiencies of different etiologies (e.g., HIV infection, primary immunodeficiency syndromes, or iatrogenic immunosuppression) have not yet been validated.
5 Conclusion
This is the first study to specifically focus on pustular reaction in AOID. Clinically, pustular reaction in AOID may be difficult to distinguish from GPP, as both conditions can present with confluent pustules—often referred to as “lakes of pus”—and annular configurations, which are considered signature features of GPP. It must be emphasized, however, that such hallmark morphologies are only rarely and inconsistently observed in AOID-associated pustular reactions. We identified several pathognomonic clinical clues that may help differentiate AOID-associated pustular reaction from GPP, including lymphadenopathy, hepatomegaly, and splenomegaly. Nevertheless, in the absence of these features, a combination of concomitant infections, elevated globulin levels, and alkaline phosphatase levels can be utilized to estimate the likelihood of pustular reaction in AOID.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding authors.
Ethics statement
This study protocol was reviewed and approved by the Ethics Committee of Faculty of Medicine, Chiang Mai University (MED-2566-09405) and Khon Kaen University Ethics Committee for Human Research, approval number HE661088. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation was not required from the participants or the participants’ legal guardians/next of kin in accordance with the national legislation and institutional requirements. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author contributions
NJ: Validation, Writing – original draft, Data curation, Visualization, Resources, Conceptualization, Formal Analysis, Methodology. PD: Writing – original draft, Investigation, Data curation, Formal Analysis, Conceptualization, Methodology. CC: Writing – review & editing, Data curation, Conceptualization, Investigation, Methodology, Resources. SuC: Resources, Visualization, Validation, Writing – review & editing, Conceptualization, Investigation, Methodology. SA: Resources, Writing – review & editing, Investigation, Conceptualization, Methodology. PC: Investigation, Validation, Conceptualization, Methodology, Resources, Writing – review & editing. SK: Resources, Methodology, Conceptualization, Writing – review & editing, Investigation. RR: Conceptualization, Resources, Writing – review & editing, Investigation, Methodology. NT: Investigation, Resources, Methodology, Writing – review & editing, Conceptualization. SiC: Resources, Writing – review & editing, Conceptualization, Methodology, Investigation. RC: Resources, Writing – review & editing, Conceptualization, Investigation, Methodology. PP: Software, Writing – review & editing, Conceptualization, Visualization, Data curation, Formal Analysis, Methodology, Resources, Validation. MC: Resources, Writing – review & editing, Funding acquisition, Investigation, Methodology, Formal Analysis, Writing – original draft, Validation, Data curation, Visualization, Project administration, Conceptualization, Supervision.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. This work was supported by the Faculty of Medicine Research Fund, grant No.031-2567.
Acknowledgments
We would like to express our sincere appreciation to Assistant Professor Dr. Arnone Nithichanon, Department of Microbiology, Faculty of Medicine, Khon Kaen University, for his invaluable contributions and dedication to the diagnostic investigations related to anti-interferon-gamma autoantibodies. The authors acknowledge the use of ChatGPT, based on OpenAI’s GPT-4 model, accessed via OpenAI’s platform (https://chat.openai.com), for assistance with improving the clarity, academic tone, and style of the manuscript. This tool was employed solely for language enhancement; all scientific content, analysis, and conclusions were generated by the authors.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2025.1619832/full#supplementary-material
Supplementary Figure 1 | Number of participants from each institution.
References
1. Browne SK, Burbelo PD, Chetchotisakd P, Suputtamongkol Y, Kiertiburanakul S, Shaw PA, et al. Adult-onset immunodeficiency in Thailand and Taiwan. N Engl J Med. (2012) 367:725–34. doi: 10.1056/NEJMoa1111160
2. Browne SK. Anticytokine autoantibody-associated immunodeficiency. Annu Rev Immunol. (2014) 32:635–57. doi: 10.1146/annurev-immunol-032713-120222
3. Pithukpakorn M, Roothumnong E, Angkasekwinai N, Suktitipat B, Assawamakin A, Luangwedchakarn V, et al. HLA-DRB1 and HLA-DQB1 are associated with adult-onset immunodeficiency with acquired anti-interferon-gamma autoantibodies. PloS One. (2015) 10:e0128481. doi: 10.1371/journal.pone.0128481
4. Zhang B, Fan J, Huang C, Fan H, Chen J, Huang X, et al. Characteristics and outcomes of anti-interferon gamma antibody-associated adult onset immunodeficiency. J Clin Immunol. (2023) 43:1660–70. doi: 10.1007/s10875-023-01537-0
5. Wongkulab P, Wipasa J, Chaiwarith R, and Supparatpinyo K. Autoantibody to interferon-gamma associated with adult-onset immunodeficiency in non-HIV individuals in Northern Thailand. PloS One. (2013) 8:e76371. doi: 10.1371/journal.pone.0076371
6. Chan JFW, Trendell-Smith NJ, Chan JCY, Hung IFN, Tang BSF, Cheng VCC, et al. Reactive and infective dermatoses associated with adult-onset immunodeficiency due to anti-interferon-gamma autoantibody: Sweet’s syndrome and beyond. Dermatol Basel Switz. (2013) 226:157–66. doi: 10.1159/000347112
7. Chetchotisakd P, Kiertiburanakul S, Mootsikapun P, Assanasen S, Chaiwarith R, and Anunnatsiri S. Disseminated nontuberculous mycobacterial infection in patients who are not infected with HIV in Thailand. Clin Infect Dis Off Publ Infect Dis Soc Am. (2007) 45:421–7. doi: 10.1086/520030
8. Choonhakarn C, Chetchotisakd P, Jirarattanapochai K, and Mootsikapun P. Sweet’s syndrome associated with non-tuberculous mycobacterial infection: a report of five cases. Br J Dermatol. (1998) 139:107–10. doi: 10.1046/j.1365-2133.1998.02324.x
9. Jutivorakool K, Sittiwattanawong P, Kantikosum K, Hurst CP, Kumtornrut C, Asawanonda P, et al. Skin manifestations in patients with adult-onset immunodeficiency due to anti-interferon-gamma autoantibody: A relationship with systemic infections. Acta Derm Venereol. (2018) 98:742–7. doi: 10.2340/00015555-2959
10. Chaowattanapanit S, Choonhakarn C, Chetchotisakd P, Sawanyawisuth K, and Julanon N. Clinical features and outcomes of Sweet’s syndrome associated with non-tuberculous mycobacterial infection and other associated diseases. J Dermatol. (2016) 43:532–6. doi: 10.1111/1346-8138.13167
11. Tungphaisal V, Phinyo P, Rujiwetpongstorn R, Kiratikanon S, Tovanabutra N, Chaiwarith R, et al. Reactive neutrophilic dermatoses in adult-onset immunodeficiency due to interferon-gamma autoantibody and their associated factors. Dermatol Basel Switz. (2023) 239:248–54. doi: 10.1159/000528064
12. Mengesha YM and Bennett ML. Pustular skin disorders: diagnosis and treatment. Am J Clin Dermatol. (2002) 3:389–400. doi: 10.2165/00128071-200203060-00003
13. Rattanathammethee K, Chawansuntati K, Chaiwarith R, Praparattanapan J, Supparatpinyo K, and Wipasa J. Dot enzyme-linked immunosorbent assay strip as a screening tool for detection of autoantibody to interferon gamma in sera of suspected cases of adult-onset immunodeficiency. J Clin Lab Anal. (2018) 32:e22460. doi: 10.1002/jcla.22460
14. Nithichanon A, Chetchotisakd P, Matsumura T, Takahashi Y, Ato M, Sakagami T, et al. Diagnosis of NTM active infection in lymphadenopathy patients with anti-interferon-gamma auto-antibody using inhibitory ELISA vs. indirect ELISA. Sci Rep. (2020) 10:8968. doi: 10.1038/s41598-020-65933-x
15. Navarini AA, Burden AD, Capon F, Mrowietz U, Puig L, Köks S, et al. European consensus statement on phenotypes of pustular psoriasis. J Eur Acad Dermatol Venereol JEADV. (2017) 31:1792–9. doi: 10.1111/jdv.14386
16. Chieosilapatham P, Daroontum T, Suwansirikul S, Chaiwarith R, Phinyo P, Chaowattanapanit S, et al. Comparative immunohistochemical analysis of inflammatory cytokines in distinct subtypes of Sweet syndrome. Front Immunol. (2024) 15:1355681/full. doi: 10.3389/fimmu.2024.1355681/full
17. Ogawa Y, Hasebe R, Ohnuma T, Sano S, Mitsui H, Shimada S, et al. Acute generalized exanthematous pustulosis associated with anti-interferon-γ neutralizing autoantibody-positive disseminated nontuberculous mycobacterial infection. Eur J Dermatol. (2019) 29:339–41. doi: 10.1684/ejd.2019.3587
18. Kantaputra P, Chaowattanapanit S, Kiratikanon S, Chaiwarith R, Choonhakarn C, Intachai W, et al. SERPINA1, generalized pustular psoriasis, and adult-onset immunodeficiency. J Dermatol. (2021) 48:1597–601. doi: 10.1111/1346-8138.16081
19. Kantaputra P, Daroontum T, Chuamanochan M, Chaowattanapanit S, Kiratikanon S, Choonhakarn C, et al. SERPINB3, adult-onset immunodeficiency, and generalized pustular psoriasis. Genes. (2023) 14:266. doi: 10.3390/genes14020266
20. Kantaputra P, Daroontum T, Chuamanochan M, Chaowattanapanit S, Intachai W, Olsen B, et al. Loss of function TGFBR2 variant as a contributing factor in generalized pustular psoriasis and adult-onset immunodeficiency. Genes. (2022) 14:103. doi: 10.3390/genes14010103
21. Sangphukieo A, Thongkumkoon P, Noisagul P, Lo Piccolo L, O’Brien TE, Chaowattanapanit S, et al. Human leukocyte antigen markers for distinguishing pustular psoriasis and adult-onset immunodeficiency with pustular reaction. Genes. (2024) 15:278. doi: 10.3390/genes15030278
22. Picciani BLS, Domingos TA, Teixeira-Souza T, dos Santos V de CB, Gonzaga HF de S, Cardoso-Oliveira J, et al. Geographic tongue and psoriasis: clinical, histopathological, immunohistochemical and genetic correlation - a literature review. Bras Dermatol. (2016) 91:410–21. doi: 10.1590/abd1806-4841.20164288
23. Kiratikanon S, Phinyo P, Rujiwetpongstorn R, Patumanond J, Tungphaisal V, Mahanupab P, et al. Adult-onset immunodeficiency due to anti-interferon-gamma autoantibody-associated Sweet syndrome: A distinctive entity. J Dermatol. (2022) 49:133–41. doi: 10.1111/1346-8138.16202
Keywords: acute generalized exanthematous pustulosis, adult-onset immunodeficiency, anti-interferongamma autoantibodies, pustular psoriasis, pustular reaction
Citation: Julanon N, Danpanichkul P, Choonhakarn C, Chaowattanapanit S, Anunnatsiri S, Chetchotisakd P, Kiratikanon S, Rujiwetpongstorn R, Tovanabutra N, Chiewchanvit S, Chaiwarith R, Phinyo P and Chuamanochan M (2025) Pustular reaction in adult-onset immunodeficiency due to anti-interferon-gamma autoantibodies. Front. Immunol. 16:1619832. doi: 10.3389/fimmu.2025.1619832
Received: 29 April 2025; Accepted: 06 August 2025;
Published: 22 August 2025.
Edited by:
Manoj Kumar Tembhre, All India Institute of Medical Sciences, IndiaReviewed by:
Nasikarn Angkasekwinai, Mahidol University, ThailandEmmanouil Karampinis, University of Thessaly, Greece
Mohammed Nasser Al-Abdulla, Hamad General Hospital, Qatar
Copyright © 2025 Julanon, Danpanichkul, Choonhakarn, Chaowattanapanit, Anunnatsiri, Chetchotisakd, Kiratikanon, Rujiwetpongstorn, Tovanabutra, Chiewchanvit, Chaiwarith, Phinyo and Chuamanochan. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Phichayut Phinyo, cGhpY2hheXV0cGhpbnlvQGdtYWlsLmNvbQ==; Mati Chuamanochan, bWF0aS5jQGNtdS5hYy50aA==
 Narachai Julanon
Narachai Julanon Pojsakorn Danpanichkul
Pojsakorn Danpanichkul Charoen Choonhakarn1
Charoen Choonhakarn1 Suteeraporn Chaowattanapanit
Suteeraporn Chaowattanapanit Salin Kiratikanon
Salin Kiratikanon Rujira Rujiwetpongstorn
Rujira Rujiwetpongstorn Siri Chiewchanvit
Siri Chiewchanvit Phichayut Phinyo
Phichayut Phinyo Mati Chuamanochan
Mati Chuamanochan