- 1Department of Dermatology, The First Affiliated Hospital of Jinan University & Jinan University Institute of Dermatology, Guangzhou, China
- 2Jinan University Institute of Dermatology, Guangzhou, China
- 3Department of Dermatology, The Fifth Affiliated Hospital of Jinan University, Heyuan, China
- 4Department of Basic Medical Sciences and Public Health, Jinan University, Guangzhou, China
- 5Department of Clinical Research, The First Affiliated Hospital of Jinan University, Guangzhou, China
- 6Guangdong Provincial Key Laboratory of Traditional Chinese Medicine Informatization, Guangzhou, China
Background: Increasing evidence suggests that metabolic syndrome (MetS) may contribute to the development of psoriasis. However, the mediating role of accelerated aging in this association remains unclear.
Methods: This study utilized data from 319,263 participants in the UK Biobank. Cox proportional hazards models were used to assess the associations between MetS, genetic predisposition, and psoriasis risk. Mediation analysis examined the role of accelerated aging (PhenoAgeAccel) in the relationship between MetS, its components, and psoriasis.
Results: MetS was associated with a 30% increased risk of psoriasis (HR: 1.30; 95% CI: 1.20–1.40). Among its components, abdominal obesity, low HDL cholesterol, high triglycerides, and hyperglycemia were each independently linked to higher risk. Individuals with both MetS and high genetic susceptibility had a substantially increased risk (HR: 2.93; 95% CI: 2.51–3.43). PhenoAgeAccel significantly mediated 28.8% of the MetS–psoriasis association.
Conclusions: MetS and its components play a key role in psoriasis development, especially in genetically susceptible individuals. Accelerated aging may partially explain this link, suggesting a potential biological pathway and underscoring the importance of early MetS identification.
Introduction
Psoriasis is a frequently occurring, chronic, and recurrent inflammatory skin condition characterized by erythematous plaques, scaling, and thickened lesions (1). The multifactorial pathogenesis of psoriasis is associated with abnormal keratinocyte proliferation driven by dysregulated immune responses (2, 3). Approximately 2% to 3% of the global population is affected, and both genders are at risk (4). Besides psoriasis’ prevalence and chronic state, it can result in considerable cosmetic disfigurement and disability (5, 6). These physical impacts often contribute to social isolation, psychological distress, and an increased risk of mental health disorders, including depression (7, 8).
Metabolic syndrome (MetS), a prevalent metabolic disorder, is characterized by a set of interrelated clinical manifestations, including central obesity, hypertension, hyperglycemia, and dyslipidemia, like elevated triglycerides and low high-density lipoprotein cholesterol levels (7, 9, 10). Studies have pointed out that the coexistence of these conditions leads to a twofold increase in cardiovascular disease risk and a fivefold increase in diabetes risk (11). Recent decades have witnessed a rise in the global prevalence of MetS, affecting around 25% of the population and accounting for 7% of total mortality (12). Transformations in modern living modes have put MetS as a major public health challenge across the world (13).
More attention is being paid to the relationship between psoriasis and MetS, and emerging evidence implies that MetS may stand as a novel risk factor for psoriasis (14, 15). The two conditions share several metabolic risk factors, including insulin resistance, obesity, hypertension, and dyslipidemia, which could collectively raise the risk of psoriasis through complex pathological mechanisms (16). Besides, chronic inflammation in psoriasis and metabolic abnormalities are closely associated, potentially contributing to disease progression via inflammatory pathways (17, 18).
Genetic factors significantly sway the onset of psoriasis, with the interaction between genetic susceptibility and environmental factors crucial for developing and spreading the disease (17). The reciprocal influence of MetS and genetic predisposition could hasten psoriasis development via immune or metabolic pathways. Further inquiry into these interactions is vital for enhancing the knowledge of psoriasis pathophysiology and formulating personalized treatment strategies.
Meanwhile, recent studies have demonstrated a close association between biological aging and metabolic syndrome (19). Biological aging has also been identified as a risk factor for the development of psoriasis (20, 21). However, few prospective studies have explored the potential mediating role of accelerated biological aging, limiting our understanding of the pathways through which this mechanism may influence psoriasis risk.
This study seeks to examine the associations of metabolic syndrome, its components, and genetic risk with psoriasis, and to assess the potential mediating role of accelerated biological aging in the MetS–psoriasis relationship.
Methods
Data source
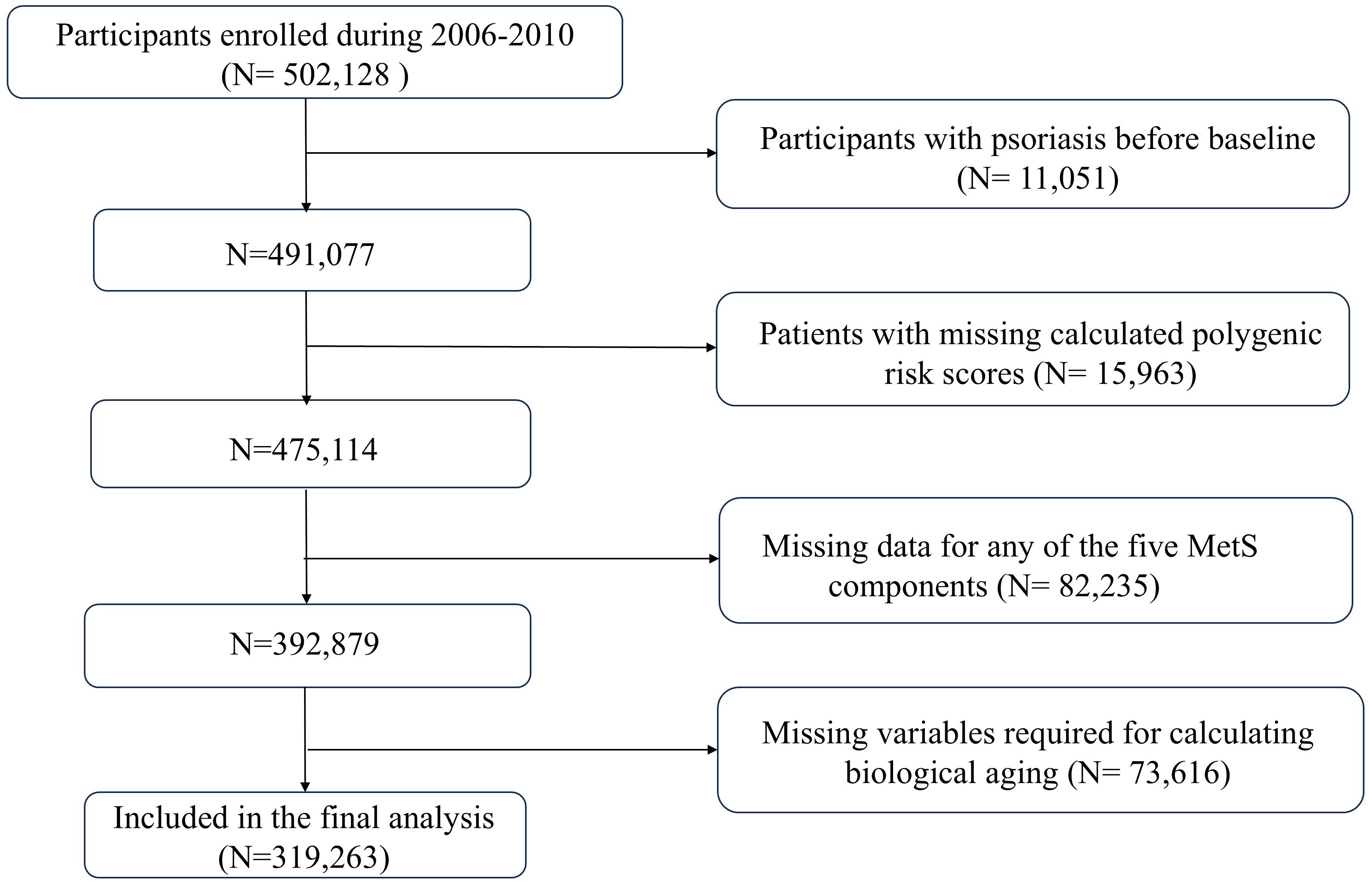
The data from the UK Biobank was applied in this prospective cohort study, which enrolled above 500,000 participants aged 37 to 73 years from April 2006 to December 2010, with follow-up concluding in July 2023. The participants were invited to take part in an assessment at one of 22 centers distributed across the UK, having sites in England, Scotland, and Wales. Detailed information regarding the UK Biobank was published previously (22). Participants were excluded under the condition that they (1) had psoriasis at baseline, (2) had data missing for any of the five components of metabolic syndrome or other relevant covariates, (3) lacked data on polygenic risk scores (PRS), or (4) had data missing for calculating biological aging. The sample choosing process is shown in Figure 1.
Assessment of psoriasis
The diagnosis of psoriasis was primarily confirmed through hospital admission records obtained from the England Hospital Episode Statistics, Scotland Incidence Records, and the Wales Patient Event Database. Confirmation criteria included events recorded for the first time (field numbers 131742 and 131743), including primary care data, ICD codes in hospital inpatient records, ICD codes in death registration records, and self-reported health status codes validated by nurses after a doctor’s diagnosis. The follow-up period was defined as starting from the day each participant was evaluated up to July 6, 2023. For full-scale details regarding psoriasis assessment in the UK Biobank, access the UK Biobank’s official site (https://www.biobank.c.k/).
Ascertainment of MetS
As per the 2009 standards developed by the American Heart Association, the National Heart, Lung, and Blood Institute, and the International Diabetes Federation, MetS is typified by having at least three of these elements: high blood pressure (systolic ≥130 mm Hg or diastolic ≥85 mm Hg); raised triglycerides (≥1.7 mmol/L); HDL cholesterol undergoes a reduction (<1 To men, the value reads 0 mmol/L, <1.3 mmol/L in women); escalated blood - sugar quantity, demonstrated by a raised HbA1c (≥42 mmol/mol [6.0%]); and elevated waist size.
Genetic information
The research platform of the UK Biobank offers a uniform polygenic risk score (PRS), created by meta-analyzing data from various genome-wide association studies (GWAS). Prior research has enhanced the understanding of the PRS methodology and the standard PRS sets used in GWAS data (23). In brief, the UK Biobank employs standardized definitions for subgroups, diseases, and quantitative traits to assess PRS consistently. The PRS algorithm is based on a meta-analysis of specific traits, utilizing the Bastion method to integrate data across diverse ancestral populations and related traits. A higher PRS reflects increased genetic susceptibility to the disease. For psoriasis (Field ID, 26269), participants were categorized into low (quintile 1), medium (quintiles 2-4), and high (quintile 5) genetic risk categories according to the PRS quintiles (24).
Covariates
Baseline sociodemographic and lifestyle data were collected via a touchscreen questionnaire. Sociodemographic variables comprised age (< 60 vs ≥ 60 years), race (white vs non-white), sex, Townsend Deprivation Index (divided into five quintiles), education level (degree, professional qualification, or secondary school), and household income (< £18,000, £18,000-£30,999, £31,000-£51,999, £52,000-£100,000, or > £100,000). Lifestyle factors included elements of smoking status (never, former, or current), alcohol consumption habit (never, former, or current), fasting time length (1–6 hours), activity (low, medium, high), and self-reported history of cardiovascular disease (CAD). A no-report category was included for participants who were uncertain or declined to respond.
Phenotypic age and PhenoAgeAccel calculation
Biological aging in this study was assessed using the PhenoAge algorithm, which estimates all-cause mortality risk based on a Gompertz proportional hazards model incorporating chronological age and nine clinical biomarkers: albumin, alkaline phosphatase, creatinine, C-reactive protein, glucose, mean corpuscular volume, red cell distribution width, white blood cell count, and lymphocyte percentage. These biomarkers were initially identified through a Cox penalized regression model using data from the NHANES III cohort (25). PhenoAge acceleration (PhenoAgeAccel), reflecting deviations in biological aging relative to chronological age, was calculated as the residual from regressing PhenoAge on chronological age (26). To address the skewed distribution of certain biomarkers, we applied percentile-based winsorization, setting values below the 1st percentile and above the 99th percentile to the corresponding cutoff points to minimize the influence of outliers (27). All calculations were performed using the “BioAge” package in R.
Statistical analysis
Descriptive statistical methods were employed to contrast the fundamental traits of participants who had Metabolic Syndrome (MetS) with those who did not. Variables of a categorical nature are denoted in percentage terms, whereas continuous variables are shown as average values with standard deviations (SD). The comparison of continuous and categorical variables across groups utilized T-tests, Wilcoxon rank-sum tests, and Chi-square (χ2) tests. Responses or missing data, such as “do not know/prefer not to answer,” were classified. The duration of follow-up was determined from the date of participant enrollment until either psoriasis diagnosis, death, dropout, or the study’s conclusion. The link between MetS, its elements, genetic vulnerability, and the risk of psoriasis was assessed using a Cox regression model, resulting in the computation of hazard ratios (HRs). The Schoenfeld residual-based test was used to confirm no significant deviations from proportional hazards in all models. Model 1 was adjusted for age, gender, household income, education level, Townsend deprivation index, and race. In Model 2, we further adjusted for polygenic risk score, cardiovascular disease history, smoking status, alcohol consumption, and fasting duration. To examine the joint exposure effects of MetS and genetic susceptibility on psoriasis risk, participants were categorized into six groups based on genetic risk (low, medium, high) and MetS (yes/no). Multicollinearity was assessed, and no significant issues were found. Additionally, the P-value for the trend was computed by coding joint exposure as a continuous variable. Stratified analyses were performed by gender (male, female) and age (< 60 years and ≥ 60 years).
Mediation analysis was conducted using the “mediation” package in R. First, a linear regression model was fitted to assess the association between the exposure (metabolic syndrome and its components) and the mediator (standardized PhenoAgeAccel), adjusting for potential confounders including age, sex, ethnicity, education level, TDI, income, smoking status, alcohol consumption, CAD history, fasting time, and physical activity. Next, a logistic regression model was constructed to evaluate the association between the exposure and the outcome (psoriasis), with both the exposure and mediator included in the model, adjusting for the same set of covariates. The average causal mediation effect (ACME) and the average direct effect (ADE) were estimated using the “mediate” function, with 1,000 bootstrap simulations to calculate confidence intervals.
Multiple sensitivity tests were performed to evaluate the solidity of the results, such as (1) omitting participants who had less than two years of follow-up to reduce differential bias; (2) excluding patients with fasting times less than 3 hours to reduce potential bias from serum metabolic biomarkers; (3) excluding patients with missing data of MetS components and covariates. Each P-value was bidirectional, establishing statistical significance at P<0.05. Every analysis was performed utilizing RStudio version 4.3.3.
Results
Baseline characteristics
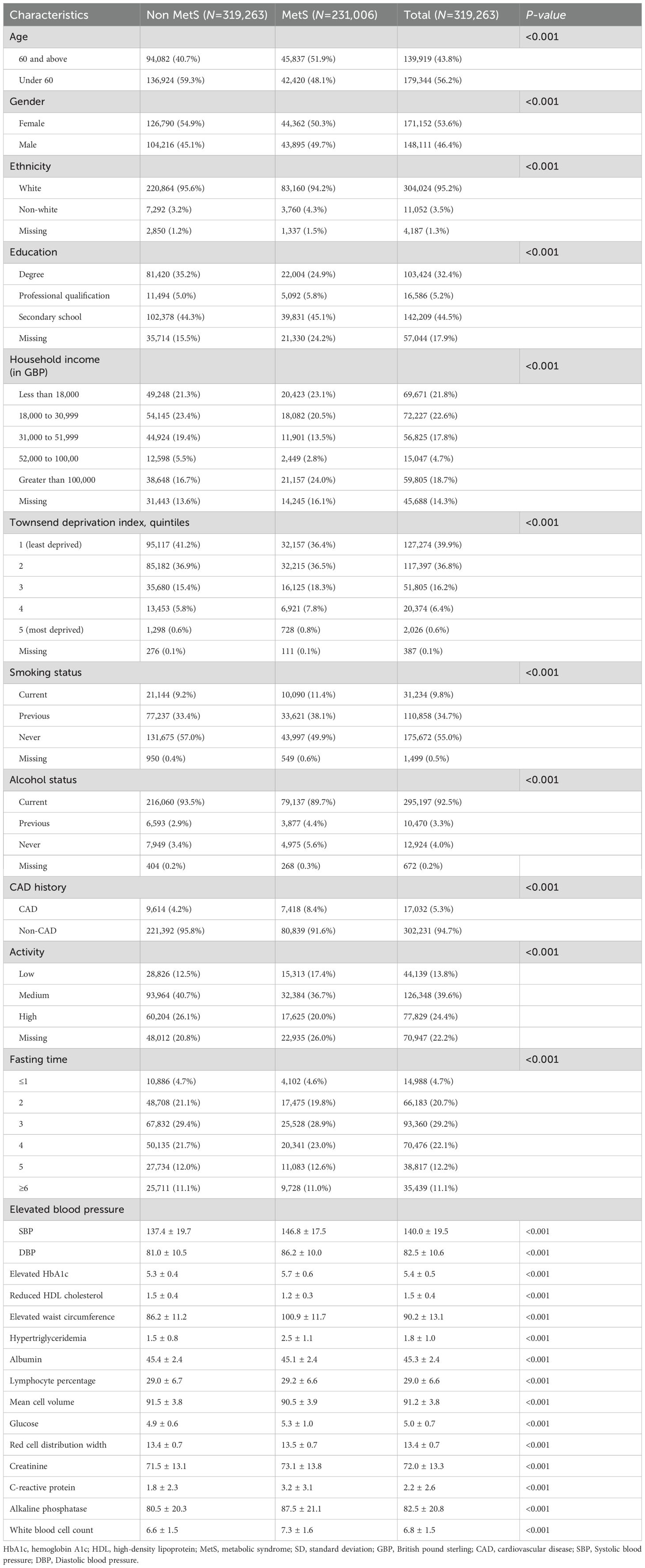
This study included 319,263 participants, of whom 231,006 (72.4%) did not have metabolic syndrome and 88,257 (27.6%) had metabolic syndrome. Table 1 displays initial traits categorized according to MetS status. Compared to those without MetS, individuals with a MetS diagnosis were more likely to be older, male, of non-white background, have lower levels of education, live in socioeconomically disadvantaged areas, earn less income, have a history of smoking, abstain from alcohol or be former drinkers, have a medium activity and report a prior diagnosis of coronary artery disease. At baseline, patients with MetS also manifested higher prevalences of hypertension, hyperglycemia, elevated triglycerides, central obesity, and low HDL cholesterol.
MetS status, PRS, and risk of incident psoriasis
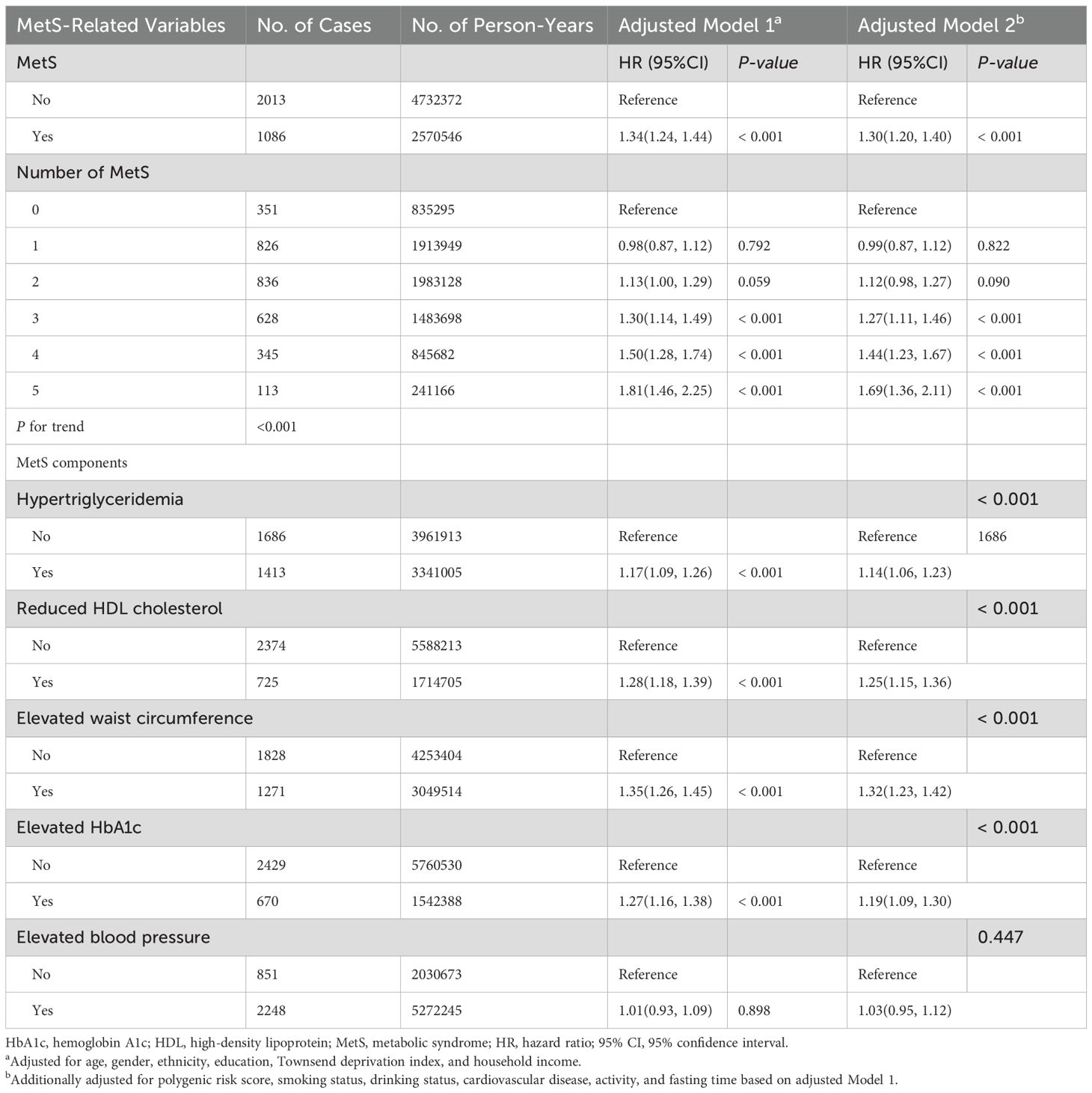
Exploring the link between metabolic syndrome and the occurrence of psoriasis, we performed a multivariable-adjusted Cox regression analysis, controlling for demographic variables in Model 1. Results in Table 2 reveal a notable link between metabolic syndrome and a heightened likelihood of psoriasis. Taking Model 1 as a base, Model 2 was additionally adapted for factors like polygenic risk score, smoking status, alcohol status, fasting time, activity, and history of CAD, according to previous findings (Table 2). People diagnosed with MetS showed a 30% increased likelihood of developing psoriasis in comparison to those free of the condition (HR 1.30; 95% CI, 1.20-1.40). Particularly speaking, the elements of metabolic syndrome—hypertriglyceridemia (HR 1.14 95% CI, 1.06-1.23), hyperglycemia (HR 1.19; 95% CI, 1.09-1.30), magnified waist size (HR 1.32; 95% CI, 1.23-1.42), and was accompanied by a drop in HDL cholesterol (HR 1.25; 95% CI, 1.15-1.36) — were respectively related to a greater possibility of psoriasis. Yet, there was no obvious link between hypertension (HR 1.03; 95% CI, 0.95-1.12) coupled with the risk of psoriasis. Besides, the likelihood of psoriasis grew in proportion to the number of metabolic abnormalities.
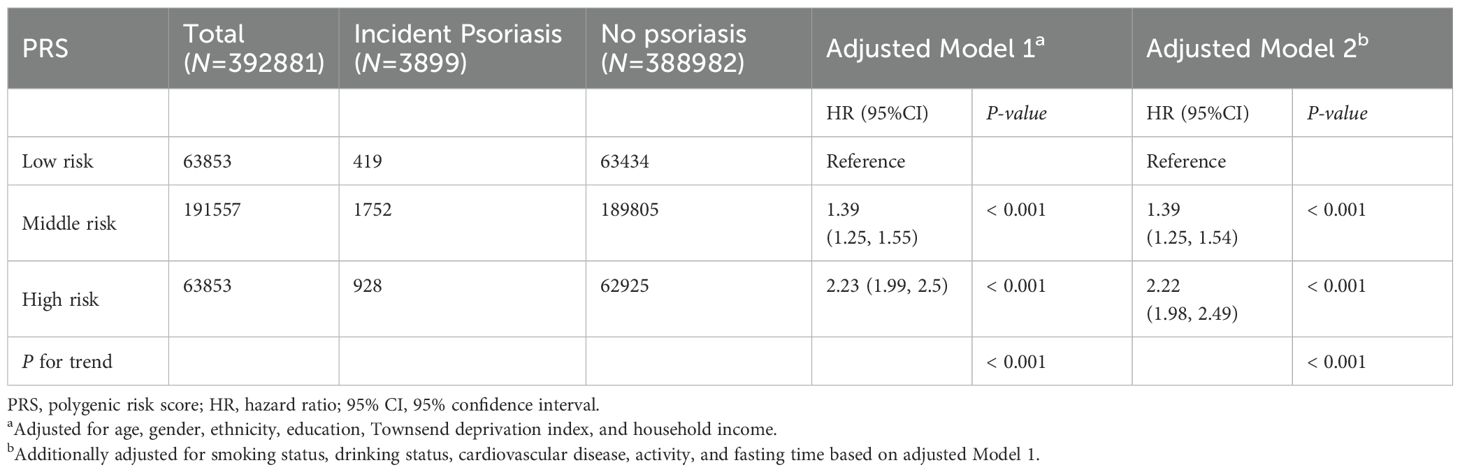
Table 3 shows how genetic risk affects psoriasis development. We measured genetic risk using polygenic risk scores. After adjustment, higher genetic risk groups had progressively higher psoriasis risk. The medium-risk group had 39% higher risk than the low-risk group (HR=1.39; 95%CI=1.25-1.54; p<0.001). The high-risk group had 2.22 times higher risk than the low-risk group (HR=2.22; 95%CI=1.98-2.49; p<0.001). The risk increased significantly with higher genetic risk scores (p-trend<0.001).
Effect of interaction between MetS status and PRS on psoriasis risk
According to the data presented in Table 4, the link between metabolic syndrome and the risk of psoriasis varies based on genetic vulnerability levels. In individuals with moderate genetic susceptibility, those with metabolic syndrome had a 27% increased risk of psoriasis (HR, 1.27; 95% CI, 1.15-1.41; P < 0.001). In individuals with high genetic susceptibility, the association was even stronger, with a 41% increased risk (HR, 1.41; 95% CI, 1.23-1.62; P < 0.001). No significant interaction between metabolic syndrome and genetic susceptibility was observed regarding psoriasis risk (P = 0.143). Further analysis of psoriasis risk revealed that all components of metabolic syndrome– such as increased waist circumference, lower HDL cholesterol, higher fasting blood glucose, and higher triglyceride levels– are associated with increased psoriasis risk. This association was more pronounced among individuals with high genetic susceptibility. However, elevated blood pressure did not demonstrate a significant association with psoriasis risk. Additionally, a significant interaction was found between elevated triglycerides and genetic risk (P = 0.021).
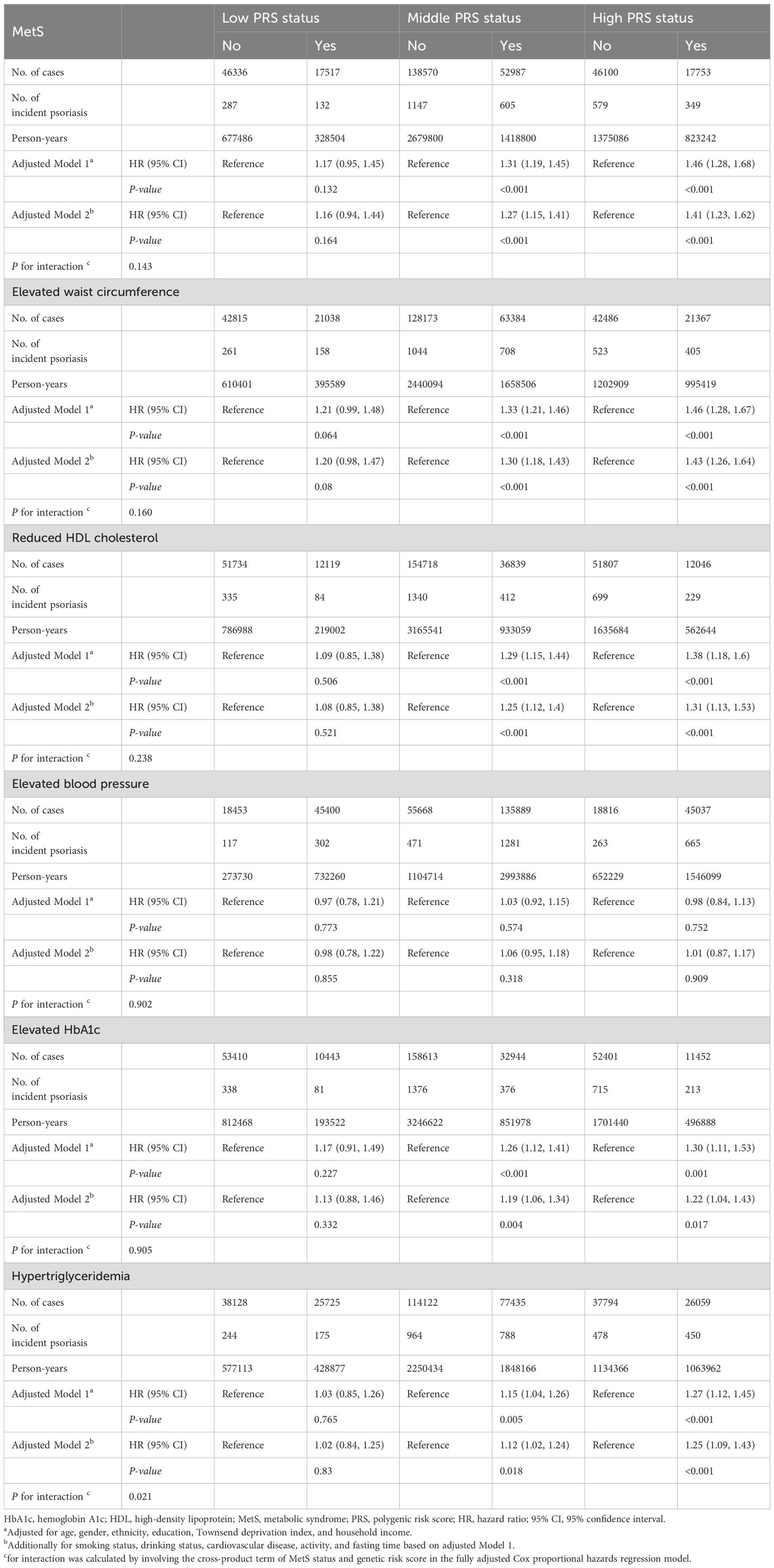
Table 4. Association between metabolic syndrome status and risk of psoriasis according to genetic predisposition.
Association between MetS status and risk of psoriasis according to PRS
In Figure 2, we examined the joint association of metabolic syndrome status and PRS with the risk of psoriasis development. In the fully adjusted model, participants with both metabolic syndrome and high genetic susceptibility exhibited a significantly elevated risk of psoriasis compared to those without metabolic syndrome and with low genetic susceptibility, with an HR of 2.93 (95% CI, 2.51-3.43). When evaluating specific metabolic components, the HRs were as follows: 2.68 (95% CI, 2.25-3.19) for elevated fasting glucose, 2.83 (95% CI, 2.39-3.36) for reduced high-density lipoprotein cholesterol, 2.52 (95% CI, 2.15-2.94) for elevated triglycerides, 2.21 (95% CI, 1.81-2.69) for elevated blood pressure, and 2.95 (95% CI, 2.52-3.45) for increased waist circumference. These findings suggest that the presence of components of metabolic syndrome, particularly associated with high genetic susceptibility, significantly increases the risk of developing psoriasis.
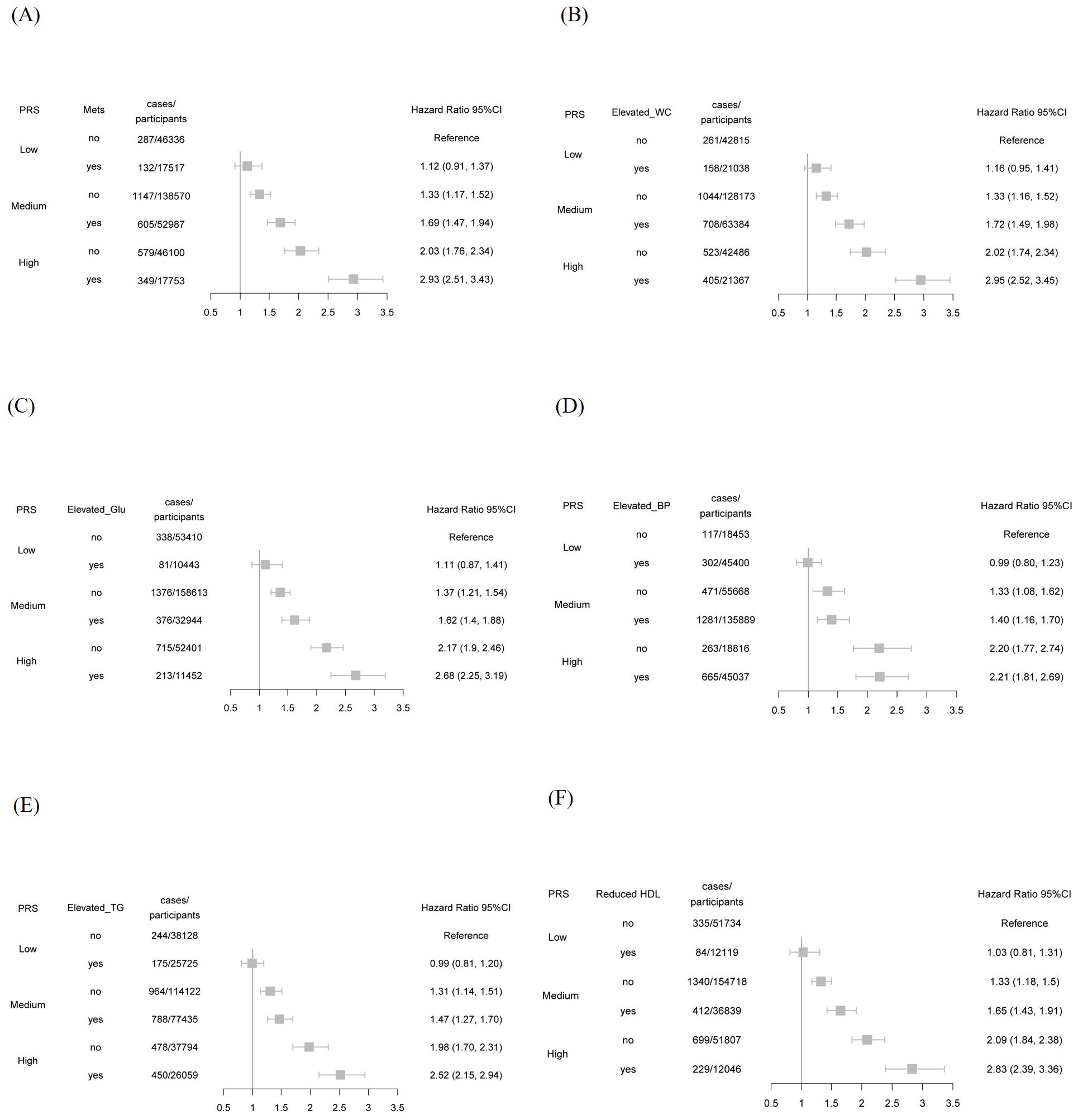
Figure 2. The risk of developing psoriasis was assessed according to metabolic syndrome (MetS) status and genetic susceptibility. (A) Metabolic syndrome (MetS); (B) Elevated waist circumference (WC); (C) Elevated glucose (Glu); (D) Elevated blood pressure (BP); (E) Elevated triglyceride (TG); (F) Reduced high-density lipoprotein (HDL).
Sensitivity analyses and subgroup
Sensitivity analyses were performed after excluding participants with missing MetS component and covariate data, less than 2 years of follow-up, or fasting times under 3 hours. As reported in Supplementary File 1, the presence of MetS and its components was significantly associated with an increased risk of psoriasis, and the dose-response relationship was significant with an increase in the number of MetS components (P represents trend < 0.001). Furthermore, Supplementary File 2 revealed that the presence of MetS continued to be associated with a higher risk of psoriasis across different PRS categories. These results enhance the performance of MetS, its components, and the risk of psoriasis under varying analytical conditions.
The analysis categorized results based on age, gender, and genetic predisposition. Findings indicated that metabolic syndrome was linked to a 46% higher risk of psoriasis in individuals younger than 60 years (HR: 1.46, 95% CI: 1.31-1.62) and a 17% increase in those aged 60 and above (HR: 1.17, 95% CI: 1.05-1.30). Among participants with a high polygenic risk score, MetS further amplified the risk, with HRs of 2.48 (95% CI: 1.98-3.12) for individuals over 60 and 3.49 (95% CI: 2.81-4.34) for those under 60. Gender-based analysis showed that MetS raised psoriasis risk by 36% in women (HR: 1.36, 95% CI: 1.22-1.51) and 25% in men (HR: 1.25, 95% CI: 1.12-1.39). Additionally, individuals with both high PRS and MetS exhibited a more than threefold risk increase, with HRs of 2.93 (95% CI: 2.34-3.67) in women and 2.96 (95% CI: 2.37-3.70) in men. Additional findings are available in Supplementary Files 3-6.
The mediating role of PhenoAgeAccel in the association between MetS and psoriasis risk
Mediation analysis indicated that PhenoAgeAccel acted as a mediator between metabolic syndrome and the risk of psoriasis (IE = 0.0007; DE = 0.0018; proportion mediated = 28.8%; P < 0.001) (Figure 3A).
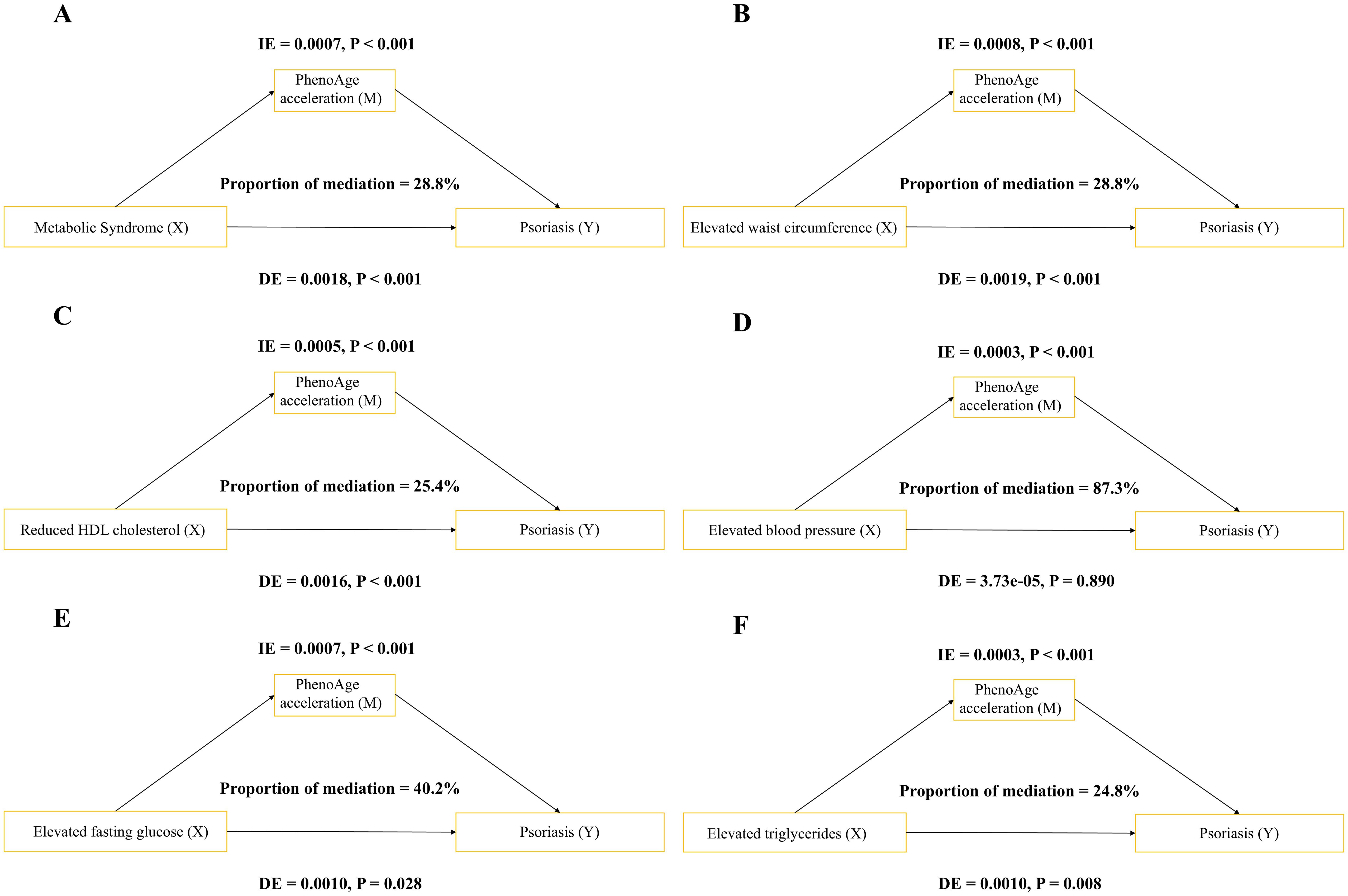
Figure 3. PhenoAge acceleration mediated the association between metabolic syndrome (MetS) components and the occurrence of psoriasis. (A) Metabolic syndrome (MetS); (B) Elevated waist circumference (WC); (C) Reduced high-density lipoprotein (HDL); (D) Elevated blood pressure (BP); (E) Elevated fasting glucose (Glu); (F) Elevated triglyceride (TG).
When examining individual components of metabolic syndrome, PhenoAge acceleration mediated 28.8% of the association between increased waist circumference and psoriasis risk (IE = 0.0008; DE = 0.0019; P < 0.001) (Figure 3B) and 25.4% of the association between reduced HDL cholesterol and psoriasis risk (IE = 0.0005; DE = 0.0016) (Figure 3C). Additionally, PhenoAgeAccel mediated 40.2% of the association between elevated fasting glucose and psoriasis risk (IE = 0.0007; DE = 0.0010) (Figure 3E) and 24.8% of the association between elevated triglycerides and psoriasis risk (IE = 0.0003; DE = 0.0010) (Figure 3F). In the elevated blood pressure group, although the indirect effect was significant (IE = 0.0003; P < 0.001), the direct effect and the proportion mediated were not significant (Figure 3D).
To further verify the mediating role of PhenoAgeAccel, several sensitivity analyses were conducted, and the results remained largely consistent with the primary findings (Supplementary Figures 1-3).
Discussion
In this extensive prospective cohort study involving 319,263 UK Biobank participants, MetS was linked to a 30% higher risk of developing psoriasis. Genetic susceptibility, assessed using a polygenic risk score, further modified this association, with MetS significantly amplifying psoriasis risk among individuals with high genetic predisposition. Among the components of MetS, elevated fasting glucose, increased triglyceride levels, larger waist circumference, and lower high-density lipoprotein cholesterol were each linked to a higher risk of psoriasis. In contrast, no significant association was found for elevated blood pressure. Mediation analysis showed that biological aging significantly mediated the association between metabolic syndrome and the risk of psoriasis, with a mediation proportion of 28.8%. To our knowledge, this is the first large-scale cohort study to explore the relationship between metabolic syndrome, genetic susceptibility, and the risk of psoriasis, while also examining the mediating role of accelerated biological aging.
For instance, a Norwegian cohort study reported that MetS was associated with a higher risk of psoriasis (RR: 1.66, 95% CI: 1.30-2.14) (28). However, most studies have primarily examined the potential mechanisms underlying the development of metabolic disorders following psoriasis rather than examining whether MetS precedes psoriasis onset. A meta-analysis reported a higher incidence of psoriasis compared to the general population (RR, 2.26; CI, 95%, 1.70-3.01) (29). Similarly, a cross-sectional study identified an increased prevalence of MetS in individuals with psoriasis (30). Findings from the present cohort study further support these associations, showing a significant 30% increased risk of psoriasis.
Psoriasis is a chronic inflammatory condition characterized by systemic inflammation (1), with elevated levels of proinflammatory cytokines, including interleukin-1β, interleukin-6, and tumor necrosis factor α. These inflammatory mediators promote oxidative damage and impair insulin signaling throughout the body, which are fundamental characteristics of metabolic syndrome (31, 32). Metabolic abnormalities may further exacerbate inflammation, promoting the onset and progression of psoriasis (1, 33).
In this study, several components of MetS were linked with an increased risk of psoriasis. Increased waist circumference was positively associated with psoriasis, which is consistent with previous findings. An American prospective cohort study reported that larger waist circumference was associated with a higher risk of psoriasis (multivariable relative risk [RR], highest vs lowest tertile, 1.50; 95% CI, 1.24-1.82) (34). This association may be attributed to abdominal fat accumulation, which promotes the secretion of proinflammatory cytokines such as tumor necrosis factor α, interleukin 6, interleukin 17, and interleukin 23 (35, 36). These cytokines are directly involved in psoriasis pathogenesis through the activation of Th17 and Th1 cells, promoting abnormal keratinocyte proliferation and cutaneous inflammation (37, 38).
Lower HDL cholesterol levels and higher triglyceride levels also increase the risk of psoriasis. This finding is consistent with a cross-sectional analysis from the UK Biobank, which reported that HDL deficiency and elevated triglycerides were associated with 16.6% and 10.6% increased psoriasis risk, respectively (39). Additionally, an observational and Mendelian randomization analysis reported an association between elevated plasma triglycerides and psoriasis risk (40).
Elevated fasting glucose levels were linked with a 22% increased risk of psoriasis, consistent with findings from a prior cohort study (41). That study estimated that psoriasis patients accounted for approximately 125,650 additional new cases of type 2 diabetes annually worldwide compared with individuals without psoriasis. Hyperglycemia is known to promote systemic inflammation through the activation of multiple inflammatory pathways, including NF-κB and JAK-STAT, which are closely linked to psoriasis pathogenesis (42, 43).
Although previous studies have shown an increased risk of hypertension, particularly resistant hypertension, among patients with severe psoriasis (44), the research we conducted found no notable link between increased blood pressure and the risk of psoriasis.
Finally, a positive correlation was noted between the quantity of MetS elements and the risk of psoriasis (P for trend < 0.001). The results of this study offer a significant understanding of the specific MetS components that may contribute to psoriasis development and highlight the potential benefit of managing these metabolic factors to reduce psoriasis risk.
This study found that the combination of genetic factors and metabolic syndrome plays a significant role in the onset of psoriasis. Participants in the group with high PRS faced a risk of psoriasis that was 2.93 times higher than those in the low PRS group. As far as we are aware, this research is pioneering in assessing whether the link between metabolic syndrome and the risk of psoriasis differs based on genetic vulnerability.
An analysis segmented by age and gender revealed a notable link between MetS and a heightened risk of psoriasis among all subgroups. This correlation was more noticeable in people under 60 years old (HR, 1.46; 95% CI, 1.31-1.62), aligning with results from earlier research (45). The link between MetS and the risk of psoriasis was marginally more pronounced in women (HR, 1.36; 95% CI, 1.22-1.51) compared to men (HR, 1.25; 95% CI, 1.12-1.39). This difference may be related to hormonal fluctuations in women, linked to a rise in visceral fat build-up and higher levels of chronic inflammation, potentially contributing to greater metabolic risk (46–48). Additional sensitivity studies reinforced the strong link between MetS and the risk of psoriasis.
Mediation analysis revealed that accelerated biological aging significantly mediated the relationship between metabolic syndrome and psoriasis. Specifically, biological aging mediated the association between metabolic abnormalities, including increased waist circumference, reduced HDL cholesterol, elevated fasting glucose, and elevated triglycerides, and the risk of developing psoriasis. This suggests that biological aging is not only a consequence of metabolic syndrome but also plays a crucial role in driving the onset of psoriasis. A recent study has suggested that biological aging could be a potential risk factor for late-onset psoriasis (21). Moreover, accelerated biological aging may activate chronic low-grade inflammation pathways, particularly NF-κB and JAK-STAT signaling, leading to elevated systemic inflammation (49, 50). This indicates that biological aging may exacerbate metabolic abnormalities by influencing immune system function and promoting systemic inflammation, thereby contributing to the development of psoriasis. These findings not only provide new insights into the association between metabolic syndrome and psoriasis but also suggest that biological aging could be a potential intervention target for psoriasis, offering a novel approach for the prevention and treatment of the disease. The relationship between metabolic syndrome and biological aging may be bidirectional. Although the current study highlights the mediating role of biological aging in the association between metabolic syndrome and psoriasis, prior evidence suggests that metabolic dysfunction may itself accelerate biological aging through mechanisms involving oxidative stress, mitochondrial dysfunction, and chronic systemic inflammation (51–53). This raises the possibility of a feedback loop in which metabolic disturbances promote biological aging, which in turn enhances inflammatory processes that contribute to psoriasis pathogenesis. Further research using longitudinal and interventional study designs is warranted to clarify the directionality and mechanistic underpinnings of this relationship. The research presents multiple advantages. It included a large sample size, a prospective design, and independently obtained data on MetS, demonstrating a longitudinal association between metabolic syndrome and its components with the risk of psoriasis in real-life settings. The study also incorporated large-scale genetic risk score data, enabling extensive stratification of gene susceptibility and allowing for an evaluation of whether the association between MetS and psoriasis varies across different levels of genetic risk. This study has several limitations. First, the participants were predominantly middle-aged and older adults of European descent, which may limit the generalizability of the findings to other racial and ethnic populations. Second, the absence of clinical data, including Psoriasis Area and Severity Index scores (PASI) and treatment information, precluded assessment of associations between metabolic syndrome and disease severity. Third, the observational design limits causal inference, and residual confounding cannot be excluded. Longitudinal studies or Mendelian randomization approaches may help clarify causal relationships. Finally, psychological stress, which may influence metabolic and immune pathways, was not assessed. Future research should incorporate validated stress measures or biomarkers (e.g., cortisol) to better evaluate its potential role.
Conclusion
In this large-scale prospective cohort study, metabolic syndrome was significantly associated with an increased risk of developing psoriasis, with the association being even stronger among individuals with higher genetic susceptibility. Specific components of MetS, including increased waist circumference, elevated fasting glucose, higher triglyceride levels, and reduced high-density lipoprotein cholesterol, were each independently linked to a greater risk of psoriasis. Mediation analysis further revealed that accelerated biological aging played a significant mediating role in the relationship between MetS and psoriasis risk, suggesting that biological aging is not merely a consequence of metabolic disturbances but also an important driver in the development of psoriasis. These findings enhance our understanding of the interplay between metabolic health, genetic predisposition, and psoriasis risk and highlight biological aging as a potential target for future prevention and intervention strategies against psoriasis.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The studies involving humans were approved by Northwest Multi-Center Research Ethics Committee (REC reference number: 11/NW/03820). The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
Author contributions
RT: Data curation, Validation, Visualization, Conceptualization, Methodology, Formal Analysis, Resources, Investigation, Funding acquisition, Project administration, Writing – review & editing, Supervision, Software, Writing – original draft. SQ: Writing – original draft, Conceptualization, Investigation, Software. JZ: Conceptualization, Writing – review & editing, Methodology, Data curation. MC: Conceptualization, Writing – review & editing. HY: Writing – original draft, Investigation, Conceptualization. JL: Software, Writing – review & editing. WL: Data curation, Conceptualization, Writing – original draft. LD: Conceptualization, Writing – review & editing, Methodology.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. The study was supported by Key Scientific Problems and Medical Technical Problems Research Project of the China Medical Education Association (2022KTZ009; 2024KTZ014).
Acknowledgments
The authors express their sincere appreciation to all participants and staff involved in data collection and management at the UK Biobank. This study was conducted using data from the UK Biobank under application number 206581.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2025.1620027/full#supplementary-material
References
1. Rendon A and Schäkel K. Psoriasis pathogenesis and treatment. Int J Mol Sci. (2019) 20(6):1475. doi: 10.3390/ijms20061475
2. Zhou G, Gan L, Zhao B, Fang F, Liu H, Chen X, et al. Adding salt to foods and risk of psoriasis: A prospective cohort study. J Autoimmun. (2024) 147:103259. doi: 10.1016/j.jaut.2024.103259
3. Griffiths CE and Barker JN. Pathogenesis and clinical features of psoriasis. Lancet (Lond Engl). (2007) 370:263–71. doi: 10.1016/S0140-6736(07)61128-3
4. Christophers E. Psoriasis–epidemiology and clinical spectrum. Clin Exp Dermatol. (2001) 26:314–20. doi: 10.1046/j.1365-2230.2001.00832.x
5. Boehncke WH and Schön MP. Psoriasis. Lancet (Lond Engl). (9997) 2015:386. doi: 10.1016/S0140-6736(14)61909-7
6. Ouyang F, Yang H, Di Z, Hu J, Ding Y, Ji C, et al. Life’s Essential 8, genetic susceptibility and the risk of psoriatic disease: a prospective cohort study. Br J Dermatol. (2024) 191:897–905. doi: 10.1093/bjd/ljae268
7. Alberti KGMM, Zimmet P, and Shaw J. The metabolic syndrome–a new worldwide definition. Lancet (Lond Engl). (2005) 366:1059–62. doi: 10.1016/S0140-6736(05)67402-8
8. Langley RGB, Krueger GG, and Griffiths CEM. Psoriasis: epidemiology, clinical features, and quality of life. Ann Rheum Dis. (2005) 64:ii18–23; discussion ii24-5. doi: 10.1136/ard.2004.033217
9. Eckel RH, Grundy SM, and Zimmet PZ. The metabolic syndrome. Lancet (Lond Engl). (2005) 365:1415–28. doi: 10.1016/S0140-6736(05)66378-7
10. Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, et al. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. (2005) 112:2735–52. doi: 10.1161/CIRCULATIONAHA.105.169404
11. Grundy SM. Pre-diabetes, metabolic syndrome, and cardiovascular risk. J Am Coll Cardiol. (2012) 59:635–43. doi: 10.1016/j.jacc.2011.08.080
12. Moura ARDS, Paz SMRSD, Frota KMG, and Carvalho CMRG. Lifestyle associated with risk of metabolic syndrome in adults and the elderly. Nutrition. (2022) 99-100:111647. doi: 10.1016/j.nut.2022.111647
13. Ford ES, Giles WH, and Dietz WH. Prevalence of the metabolic syndrome among US adults: findings from the third National Health and Nutrition Examination Survey. JAMA. (2002) 287:356–9. doi: 10.1001/jama.287.3.356
14. Gisondi P, Fostini AC, Fossà I, Girolomoni G, and Targher G. Psoriasis and the metabolic syndrome. Clin Dermatol. (2018) 36:21–8. doi: 10.1016/j.clindermatol.2017.09.005
15. Mohamed Haris NH, Krishnasamy S, Chin KY, Mariappan V, and Arumugam M. Metabolic syndrome screening and nutritional status of patients with psoriasis: A scoping review. Nutrients. (2023) 15(12):2707. doi: 10.3390/nu15122707
16. Hao Y, Zhu YJ, Zou S, Zhou P, Hu YW, Zhao QX, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. (2021) 12:711060. doi: 10.3389/fimmu.2021.711060
17. Wu JJ, Kavanaugh A, Lebwohl MG, Gniadecki R, and Merola JF. Psoriasis and metabolic syndrome: implications for the management and treatment of psoriasis. J Eur Acad Dermatol Venereol. (2022) 36:797–806. doi: 10.1111/jdv.18044
18. Secchiero P, Rimondi E, Marcuzzi A, Longo G, Papi C, Manfredini M, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. (2024) 25:1–14. doi: 10.3390/ijms25158098
19. Dominguez LJ and Barbagallo M. The biology of the metabolic syndrome and aging. Curr Opin Clin Nutr Metab Care. (2016) 19:5–11. doi: 10.1097/MCO.0000000000000243
20. Lin Z, Wang HF, Yu LY, Chen J, Kong CC, Zhang B, et al. The relationship between biological aging and psoriasis: evidence from three observational studies. Immun Ageing. (2025) 22:6. doi: 10.1186/s12979-025-00500-4
21. Yang Z, Wang F, Zhang T, Mao R, and Li J. Association of aging and the risk of late-onset psoriasis: Evidence from the UK Biobank cohort. J Am Acad Dermatol. (2025) 92(6):1375–7. doi: 10.1016/j.jaad.2025.01.070
22. Sudlow C, Gallacher J, Allen N, Beral V, Burton P, Danesh J, et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PloS Med. (2015) 12:e1001779. doi: 10.1371/journal.pmed.1001779
23. Bycroft C, Freeman C, Petkova D, Band G, Elliott LT, Sharp K, et al. The UK Biobank resource with deep phenotyping and genomic data. Nature. (2018) 562:203–9. doi: 10.1038/s41586-018-0579-z
24. Ye YX, Chen JX, Li Y, Lai YW, Lu Q, Xia PF, et al. Adherence to a planetary health diet, genetic susceptibility, and incident cardiovascular disease: a prospective cohort study from the UK Biobank. Am J Clin Nutr. (2024) 120:648–55. doi: 10.1016/j.ajcnut.2024.06.014
25. Levine ME, Lu AT, Quach A, Chen BH, Assimes TL, Bandinelli S, et al. An epigenetic biomarker of aging for lifespan and healthspan. Aging (Albany NY). (2018) 10:573–91. doi: 10.18632/aging.101414
26. Liu Z, Kuo PL, Horvath S, Crimmins E, Ferrucci L, and Levine M. A new aging measure captures morbidity and mortality risk across diverse subpopulations from NHANES IV: A cohort study. PloS Med. (2018) 15:e1002718. doi: 10.1371/journal.pmed.1002718
27. Davidov O, Jelsema CM, and Peddada S. Testing for inequality constraints in singular models by trimming or winsorizing the variance matrix. J Am Stat Assoc. (2018) 113:906–18. doi: 10.1080/01621459.2017.1301258
28. Snekvik I, Nilsen TIL, Romundstad PR, and Saunes M. Metabolic syndrome and risk of incident psoriasis: prospective data from the HUNT Study, Norway. Br J Dermatol. (2019) 180:94–9. doi: 10.1111/bjd.16885
29. Armstrong AW, Harskamp CT, and Armstrong EJ. Psoriasis and metabolic syndrome: a systematic review and meta-analysis of observational studies. J Am Acad Dermatol. (2013) 68:654–62. doi: 10.1016/j.jaad.2012.08.015
30. Teklu M, Zhou W, Kapoor P, Patel N, Dey AK, Sorokin AV, et al. Metabolic syndrome and its factors are associated with noncalcified coronary burden in psoriasis: An observational cohort study. J Am Acad Dermatol. (2021) 84:1329–38. doi: 10.1016/j.jaad.2020.12.044
31. Shoelson SE, Lee J, and Goldfine AB. Inflammation and insulin resistance. J Clin Invest. (2006) 116:1793–801. doi: 10.1172/JCI29069
32. Coto-Segura P, Eiris-Salvado N, González-Lara L, Queiro-Silva R, Martinez-Camblor P, Maldonado-Seral C, et al. Psoriasis, psoriatic arthritis and type 2 diabetes mellitus: a systematic review and meta-analysis. Br J Dermatol. (2013) 169:783–93. doi: 10.1111/bjd.12473
33. Silveira Rossi JL, Barbalho SM, Reverete de Araujo R, Bechara MD, Sloan KP, and Sloan LA. Metabolic syndrome and cardiovascular diseases: Going beyond traditional risk factors. Diabetes Metab Res Rev. (2022) 38:e3502. doi: 10.1002/dmrr.3502
34. Kumar S, Han J, Li T, and Qureshi AA. Obesity, waist circumference, weight change and the risk of psoriasis in US women. J Eur Acad Dermatol Venereol. (2013) 27:1293–8. doi: 10.1111/jdv.12001
35. Guo L, Kircik L, and Armstrong AW. INDIVIDUAL ARTICLE: psoriasis and obesity: optimizing pharmacologic treatment and lifestyle interventions. J Drugs Dermatol. (2025) 24:491722s4–491722s14.
36. Liang Y, Wang Y, Peng A, Li J, and Zhang K. Molecular mechanisms and drug therapy of metabolism disorders in psoriasis. J Dermatolog Treat. (2024) 35:2375580. doi: 10.1080/09546634.2024.2375580
37. Guttman-Yassky E, Krueger JG, and Lebwohl MG. Systemic immune mechanisms in atopic dermatitis and psoriasis with implications for treatment. Exp Dermatol. (2018) 27:409–17. doi: 10.1111/exd.13336
38. Lockshin B, Balagula Y, and Merola JF. Interleukin 17, inflammation, and cardiovascular risk in patients with psoriasis. J Am Acad Dermatol. (2018) 79:345–52. doi: 10.1016/j.jaad.2018.02.040
39. Xiao Y, Jing D, Tang Z, Peng C, Yin M, Liu H, et al. Serum lipids and risk of incident psoriasis: A prospective cohort study from the UK biobank study and mendelian randomization analysis. J Invest Dermatol. (2022) 142:3192–3199.e12. doi: 10.1016/j.jid.2022.06.015
40. Greve AM, Wulff AB, Bojesen SE, and Nordestgaard BG. Elevated plasma triglycerides increase the risk of psoriasis: a cohort and Mendelian randomization study. Br J Dermatol. (2024) 191:209–15. doi: 10.1093/bjd/ljae089
41. Wan MT, Shin DB, Hubbard RA, Noe MH, Mehta NN, and Gelfand JM. Psoriasis and the risk of diabetes: A prospective population-based cohort study. J Am Acad Dermatol. (2018) 78:315–322.e1. doi: 10.1016/j.jaad.2017.10.050
42. Shao Z, Li X, Xu X, and Chen P. DPP-4 inhibitor linagliptin ameliorates imiquimod-induced psoriasis-like skin alterations in type 2 diabetic mice by inhibiting the MAPK/NF-κB inflammatory pathway. Drug Dev Res. (2022) 83:1373–82. doi: 10.1002/ddr.21966
43. Mamizadeh M, Tardeh Z, and Azami M. The association between psoriasis and diabetes mellitus: A systematic review and meta-analysis. Diabetes Metab Syndr. (2019) 13:1405–12. doi: 10.1016/j.dsx.2019.01.009
44. Hu MY, Yang Q, and Zheng J. The association of psoriasis and hypertension: focusing on anti-inflammatory therapies and immunological mechanisms. Clin Exp Dermatol. (2020) 45:836–40. doi: 10.1111/ced.14327
45. Wang X, Zeng Z, Wang X, Zhao P, Xiong L, Liao T, et al. Magnesium depletion score and metabolic syndrome in US adults: analysis of NHANES 2003 to 2018. J Clin Endocrinol Metab. (2024) 109:e2324–33. doi: 10.1210/clinem/dgae075
46. Jeong HG and Park H. Metabolic disorders in menopause. Metabolites. (2022) 12:1–15. doi: 10.3390/metabo12100954
47. Han A and Choi YJ. Comparison of metabolic syndrome (MetS) risk and nutritional status according to menopause age and the impact of socioeconomic status on metS prevalence in postmenopausal women: A cross-sectional study based on the 8th korea national health and nutrition. Nutrients. (2024) 16:1–13. doi: 10.3390/nu16070967
48. Pu D, Tan R, Yu Q, and Wu J. Metabolic syndrome in menopause and associated factors: a meta-analysis. Climacteric. (2017) 20:583–91. doi: 10.1080/13697137.2017.1386649
49. Wang Y, Wang L, Wen X, Hao D, Zhang N, He G, et al. NF-κB signaling in skin aging. Mech Ageing Dev. (2019) 184:111160. doi: 10.1016/j.mad.2019.111160
50. Onorati A, Havas AP, Lin B, Rajagopal J, Sen P, Adams PD, et al. Upregulation of PD-L1 in senescence and aging. Mol Cell Biol. (2022) 42:e0017122. doi: 10.1128/mcb.00171-22
51. Masenga SK, Kabwe LS, Chakulya M, and Kirabo A. Mechanisms of oxidative stress in metabolic syndrome. Int J Mol Sci. (2023) 24:e0017122. doi: 10.3390/ijms24097898
52. Prasun P. Mitochondrial dysfunction in metabolic syndrome. Biochim Biophys Acta Mol Basis Dis. (2020) 1866:165838. doi: 10.1016/j.bbadis.2020.165838
Keywords: metabolic syndrome, psoriasis, genetic susceptibility, biological aging, mediation analysis
Citation: Tian R, Qiu S, Zhang J, Chen M, Yu H, Lau W, Lyu J and Deng L (2025) Accelerated biological aging as a potential mediator mediates the relationship between metabolic syndrome and the risk of psoriasis: a prospective analysis from the UK biobank. Front. Immunol. 16:1620027. doi: 10.3389/fimmu.2025.1620027
Received: 29 April 2025; Accepted: 16 July 2025;
Published: 11 August 2025.
Edited by:
Manoj Kumar Tembhre, All India Institute of Medical Sciences, IndiaReviewed by:
Ramune Sepetiene, Abbott, United StatesVinh Ngo, Pham Ngoc Thach University of Medicine, Vietnam
Copyright © 2025 Tian, Qiu, Zhang, Chen, Yu, Lau, Lyu and Deng. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jun Lyu, bHl1anVuMjAyMEBqbnUuZWR1LmNu; Liehua Deng, TGllaHVhZGVuZ0AxMjYuY29t
†These authors have contributed equally to this work and share first authorship
 Rongqian Tian
Rongqian Tian Shaona Qiu
Shaona Qiu Jinrong Zhang
Jinrong Zhang Ming Chen
Ming Chen Hai Yu
Hai Yu Waichi Lau1,2,4
Waichi Lau1,2,4 Jun Lyu
Jun Lyu Liehua Deng
Liehua Deng