- Department of Rheumatology, Endocrinology and Nephrology, Faculty of Medicine and Graduate School of Medicine, Hokkaido University, Sapporo, Japan
Immune cell metabolism is essential for regulating immune responses, including activation, differentiation, and function. Through glycolysis and oxidative phosphorylation (OXPHOS), metabolism supplies energy and key intermediates for cell growth and proliferation. Importantly, some metabolites generated during these processes act as signaling molecules that influence immune activity. Autoimmune diseases such as rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE) involve multiple immune cell types, and recent research in immunometabolism has revealed that disrupted metabolic pathways in these cells contribute to disease progression. Effector T cells, for instance, undergo metabolic reprogramming, particularly increased glycolysis, to meet the demands of proliferation and function during autoimmune responses. Targeting metabolic enzymes has shown therapeutic potential. In addition, metabolites themselves, termed immunometabolites, can directly modulate immune responses. These include both intracellularly generated and secreted molecules. Itaconate is a key immunometabolite and is derived from the TCA cycle by aconitate decarboxylase 1 (ACOD1) in activated macrophages. It inhibits the NLRP3 inflammasome and pro-inflammatory cytokines, such as IL-1β and IL-6. Beyond macrophages, itaconate alters metabolism and epigenetics in T cells by reducing 2-hydroxyglutarate and the S-adenosyl-L-methionine (SAM)/S-adenosyl-L-homocysteine (SAH) ratio, thereby suppressing Th17 differentiation and enhancing Foxp3 expression in Tregs. Itaconate ameliorates disease in experimental autoimmune encephalomyelitis, RA, SLE, and others. It also exhibits antimicrobial effects by blocking bacterial isocitrate lyase and viral replication. Despite increasing interest, reviews focusing specifically on immunometabolites remain limited. This review highlights emerging insights into metabolites involved in glycolysis, the TCA cycle, glutaminolysis, one-carbon metabolism, and lipid metabolism that influence autoimmune pathophysiology.
1 Introduction
Metabolism plays a crucial role in regulating the function of immune cells, influencing their activation, differentiation, and overall immune responses (1). It is essential for producing adenosine triphosphate (ATP) via several pathways, such as glycolysis and oxidative phosphorylation (OXPHOS). Metabolism also provides essential intermediates for nucleotide, amino acid, and lipid synthesis during cell proliferation and activation. Importantly, metabolites produced through intracellular metabolic processes can act as signaling molecules that regulate various cellular activities (2, 3).
Recently, immunometabolism, the study of metabolism in immune cells, has revealed that dysregulated immune cell metabolism contributes to the pathophysiology of autoimmune diseases (4). Autoimmune diseases, such as rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE), involve multiple different types of immune cells. Autoimmune diseases are characterized by a loss of tolerance to self-antigens, resulting in chronic inflammation and tissue damage. This breakdown of self-tolerance involves multiple immune cell subsets, including autoreactive effector T cells, such as Th1 and Th17, dysfunctional Tregs, activated B cells, and pro-inflammatory macrophages (5). These immune cells contribute to the excessive production of cytokines, autoantibodies, and tissue-infiltrating inflammatory mediators, thereby promoting disease progression (5). For instance, T cells rely on metabolic reprogramming, particularly glycolysis, for their energetic and biosynthetic demands during proliferation and differentiation into effector T cells (6). This metabolic shift is crucial for their activation and subsequent functions, especially in the context of autoimmune diseases (7). Therefore, the targeting of specific enzymes in immune cells is gaining traction as a potential therapeutic strategy (8). Moreover, recent research has also demonstrated that certain metabolites themselves influence immune cell responses by either accumulating within immune cells or by being secreted by non-immune cells to modulate immune responses (Table 1). These metabolites are defined as immunometabolites, and their modulation or supplementation could also be a new therapeutic approach.
While many review papers focusing on enzymes in immunometabolism have been published, those focused on immunometabolites are limited. The present review discusses the latest research on representative immunometabolites in glycolysis, the tricarboxylic acid (TCA) cycle, glutaminolysis, and lipid metabolism, which regulate the pathophysiology of autoimmune diseases, including systemic lupus erythematosus (SLE) and rheumatoid arthritis (RA).
2 Glycolysis
2.1 Lactate
Lactate is a metabolite generated from pyruvate by lactate dehydrogenase (LDH) through anaerobic glycolysis (Figure 1) (9). This pathway is activated under conditions of high energy demand or limited oxygen availability, such as during infection, inflammation, or cancer (10). In RA, lactate levels are markedly elevated in the hypoxic synovial microenvironment compared to osteoarthritis, primarily due to increased production and secretion by fibroblast-like synoviocytes (11). Elevated serum lactate levels have been reported in patients with multiple sclerosis and Sjögren’s syndrome (12, 13). Lactate exerts diverse immunomodulatory effects, functioning as both an immunosuppressive and pro-inflammatory mediator.
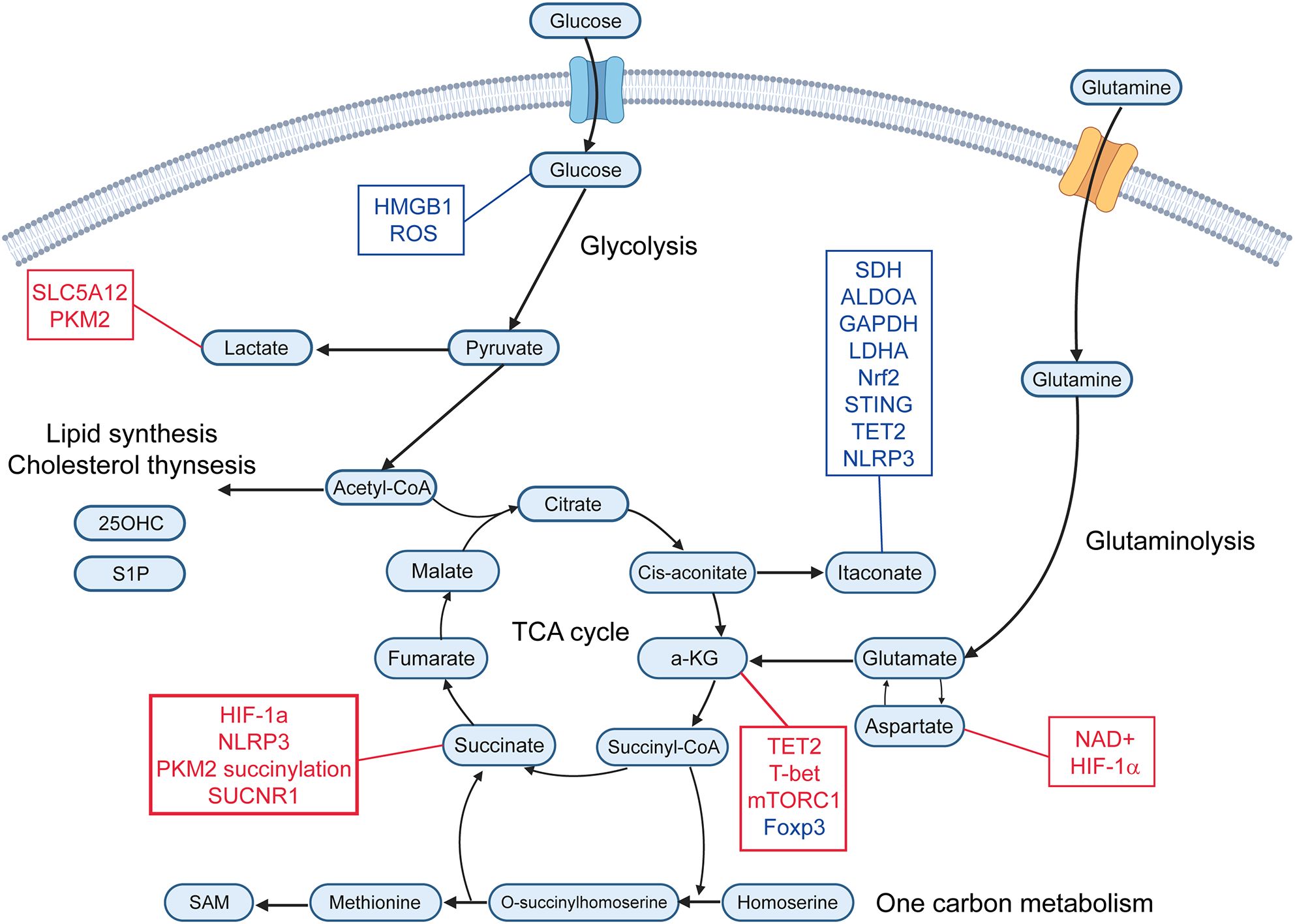
Figure 1. Immunometabolites and their molecular targets. This schematic illustrates key immunometabolites that regulate immune responses. Metabolites are organized according to their primary metabolic pathways. Immunomodulatory metabolites are connected to their downstream molecular targets. Red text represents upregulation or activation of the indicated target, while blue text represents downregulation or inhibition. α-KG, alpha-ketoglutarate; SAM, S-adenosyl-L-methionine; 25-OHC, 25-Hydroxycholeesterol; S1P, Sphingosine-1-phosphate; ROS, reactive oxygen species; PKM2, pyruvate kinase M2; SDH, succinate dehydrogenase; ALDOA, aldolase A; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; LDHA, lactate dehydrogenase; Nrf2, nuclear factor erythroid 2-related factor 2; TET2, ten-eleven translocation 2.
Lactate modulates T cell function, contributing to inflammatory processes. The accumulation of lactate leads to upregulation of the lactate transporter SLC5A12 in human CD4 T cells, resulting in lactate uptake into CD4 T cells (14). This induces nuclear translocation of dimeric pyruvate kinase M2 (PKM2), activates acetyl-CoA carboxylase, thereby suppressing glycolysis and promoting fatty acid synthesis (14, 15). Human CD4 T cells treated with lactate increased IL-17A production (14). Inhibition of PKM2 reduces Th17 cell differentiation and attenuates disease activity of experimental autoimmune encephalomyelitis (EAE) (9). Furthermore, blockade of SLC5A12 has been shown to alleviate disease severity in a murine model of arthritis (14). However, other studies have shown that lactate suppresses the Th17 cell differentiation and promotes Treg cell differentiation (16, 17). These discrepancies should be further investigated in future studies.
Recent studies have shown that aging B cells shift their cellular metabolism toward glycolysis, secrete increased amounts of lactate, and adopt a proinflammatory phenotype via the SLC5A12, characterized by senescence-associated profile mediators and the production of autoantibodies, such as anti-dsDNA (18).
On the other hand, in macrophages, recent research has revealed that lactate serves as a fuel to promote histone H3K27 acetylation, leading to epigenetic changes, enabling the expression of immunosuppressive genes, such as Nr4A1 (19) (Figure 2). This process transcriptionally represses pro-inflammatory functions in macrophages and sustains long-term immunosuppression (19). These differences may reflect cell type-specific responses or variations in disease models.
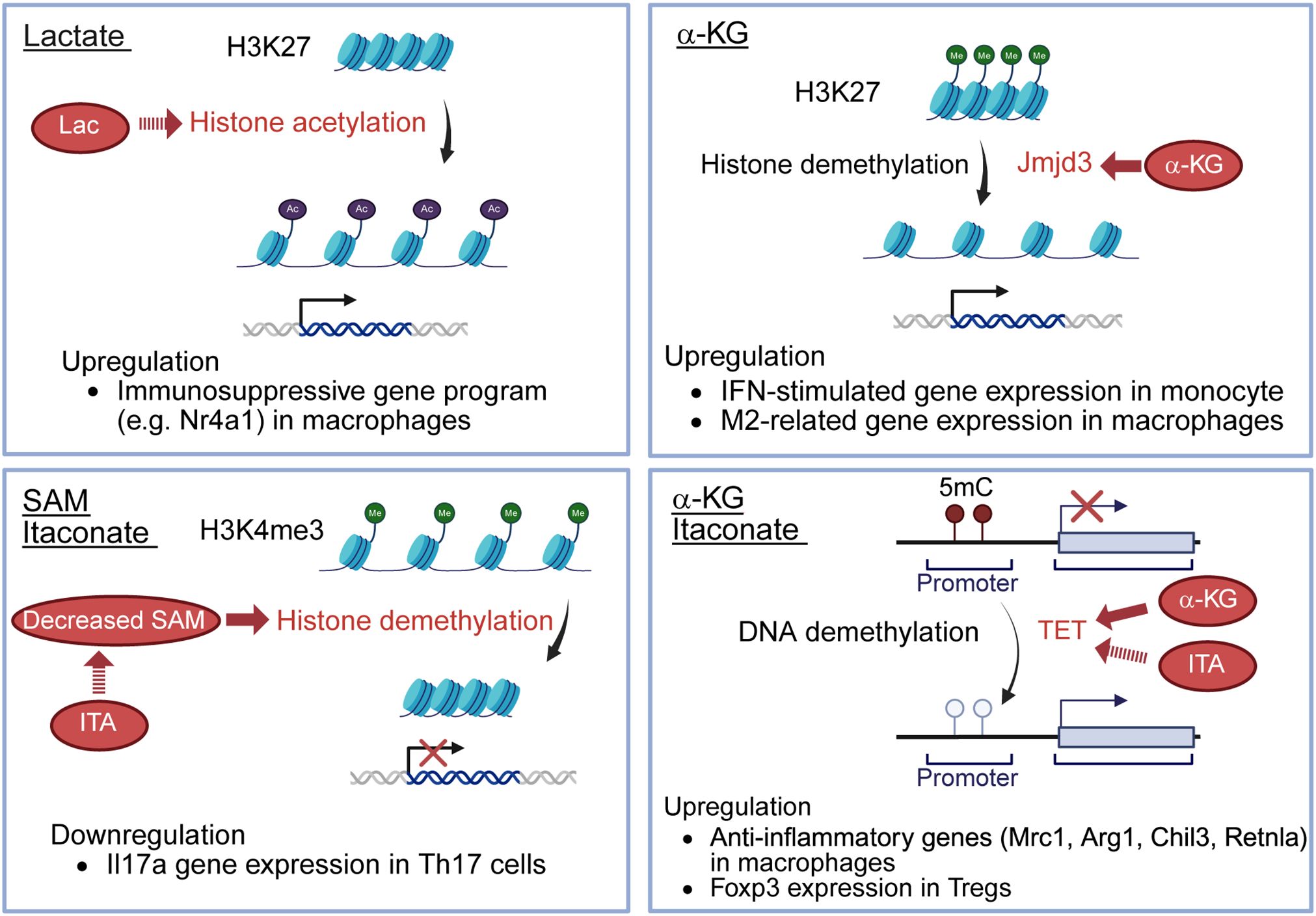
Figure 2. Effects of immunometabolites for epigenetics. Lactate serves as a fuel to promote histone H3K27 acetylation, resulting in the upregulation of immunosuppressive genes, such as Nr4a1, in macrophages. Itaconate decreases SAM/SAH ratio, thereby limiting histone demethylation at loci of the Il17a promoter, resulting in the downregulation of Il17a gene expression in Th17 cells. α-KG enhances the activity of Jmjd3, a histone demethylase, promoting histone H3K27 demethylation and inducing the upregulation of IFN-stimulated gene expression in monocytes and M2-related gene expression in macrophages. α-KG also serves as a cofactor for TET2, a DNA demethylase, leading to upregulated anti-inflammatory genes (Mrc1, Arg1, Chil3, Retnla) in macrophages. Itaconate indirectly promotes TET by inhibiting isocitrate dehydrogenases 1/2 in Tregs, decreasing 2-HG, known to inhibit TET enzymes. The increased TET activity allows TET-mediated DNA demethylation, leading to the upregulation of Foxp3 expression in Tregs. SAM, S-adenosyl-L-methionine; SAH, S-adenosyl-L-homocysteine; α-KG, alpha-ketoglutarate; TET2, ten-eleven translocation 2; 2-HG, 2-hydroxyglutarate; Lac, lactate; ITA, itaconate; 5mC, 5-methylcytosine.
2.2 Pyruvate
Pyruvate is a central metabolite produced primarily through glycolysis (Figure 1), wherein glucose is converted to pyruvate by pyruvate kinase under conditions of high energy demand, such as immune cell activation. Additional sources include lactate via LDH and alanine via alanine aminotransferase. Mitochondrial pyruvate uptake via the mitochondrial pyruvate carrier links glycolysis to the TCA cycle, supporting ATP production and biosynthetic pathways (20, 21). Ethyl pyruvate (EP), a stable derivative of pyruvate, has shown efficacy in experimental models of autoimmune diseases such as myocarditis, encephalomyelitis, and type 1 diabetes (22–24). EP reduces immune cell infiltration and inflammation by inhibiting HMGB1 signaling and promoting regulatory immune cells, including Tregs and tolerogenic dendritic cells (22, 24). In macrophages, pyruvate has anti-inflammatory effects by reducing mitochondrial reactive oxygen species (ROS) and pro-inflammatory cytokines, such as IL-1β, IL-6, and TNFα during viral infections (25).
3 TCA cycle
3.1 Succinate
Succinate, a key intermediate in the TCA cycle (Figure 1), is accumulated in human cells under conditions of hypoxia, inflammatory activation, and metabolic stress (26). In hypoxic microenvironments, macrophages shift toward glycolysis, leading to succinate accumulation due to impaired mitochondrial oxidation and reverse electron transport (27). Lipopolysaccharide (LPS) activation of Toll-like receptor 4 (TLR4) in macrophages drives succinate accumulation by inhibiting succinate dehydrogenase (SDH) and upregulating glutamine-dependent anaplerosis (27, 28). Inflammatory stimuli promote glycolytic flux, thereby increasing succinate production through the TCA cycle. Hypoxia-inducible factor-1α (HIF-1α) stabilizes under low oxygen, further enhancing glycolysis and succinate generation. Mitochondrial dysfunction (ROS overproduction) or mutations in SDH subunits can lead to pathological succinate accumulation (29).
Succinate accumulation in activated macrophages stabilizes HIF-1α by inhibiting prolyl hydroxylases, leading to increased transcription of IL-1β and NLRP3 inflammasome activation (27). High alpha-ketoglutarate (α-KG)/succinate ratios favor anti-inflammatory macrophage polarization, while low ratios drive pro-inflammatory responses (30). Cytosolic succinate can modify proteins, such as PKM2 via succinylation, enabling its nuclear translocation and transcriptional activation of pro-inflammatory genes in macrophages (31). Extracellular succinate binds the G-protein-coupled receptor SUCNR1 on anti-inflammatory macrophages, triggering calcium signaling and ERK phosphorylation to promote tissue repair and prostaglandin E2 secretion (32). In T cells, succinate directly influences their differentiation into pro-inflammatory Th1 and Th17 cells, especially under the condition of impaired SDH function and intracellular succinate accumulates. Elevated succinate/α-KG ratios in T cells upregulate pro-inflammatory gene expression and cytokine production, including IFNγ and IL-17A (33). In a mouse model of experimental autoimmune uveitis, succinate administration exacerbate disease severity by promoting neutrophil extracellular trap formation and increasing Th1/Th17 cell populations (34). Succinate can also suppress T cell degranulation and cytokine secretion in CD4 and CD8 T cells, particularly affecting the production of IFNγ (35). In dendritic cells, succinate promotes their maturation and migration to lymph nodes via SUCNR1, enhancing antigen presentation and IL-1β secretion, which facilitates T cell activation (36). In experimental arthritis models, succinate exposure increases Th17 cell frequencies and disease severity through SUCNR1-expressing dendritic cells (37).
3.2 Itaconate
ITA is one of the most important endogenous immunometabolites derived from the mitochondrial TCA cycle primarily in activated macrophages. ITA is produced from cis-aconitate (Figure 1), the TCA cycle intermediate, by aconitate decarboxylase 1 (ACOD1) which is induced during inflammatory responses in macrophages (38). ITA is also known to be produced in macrophages in response to glucocorticoid stimulation, and interestingly, this pathway is essential for the immunosuppressive action of glucocorticoids (39). ITA inhibits SDH, leading to reduced mitochondrial ROS, and suppression of pro-inflammatory cytokines, including IL-1β and IL-6 in macrophages (40). ITA and its derivatives also directly inhibit key glycolytic enzymes such as aldolase A (ALDOA), glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and lactate dehydrogenase A (LDHA), thereby reducing glycolysis and inflammatory responses (41). Moreover, ITA modulates transcription factors, such as nuclear factor erythroid 2-related factor 2 (Nrf2), STING, ten-eleven translocation 2 (TET2) and NLRP3, reducing inflammasome activation and oxidative stress (42, 43). Recent research indicates that ITA affects not only macrophages but also other immune cells and non-immune cells. ITA suppresses glycolysis and oxidative phosphorylation in T cells, leading to metabolic and epigenetic reprogramming of T cells (44). ITA decreases Th17 cell differentiation and increases Treg cell differentiation (44). In contrast, ITA does not affect Th1 or Th2 cell differentiation (44). Following treatment with ITA, the S-adenosyl-L-methionine (SAM)/S-adenosylhomocysteine (SAH) ratio is decreased, reducing histone methylation at loci of the Il17a promoter and limiting RORγt binding at the Il17a promoter in Th17 cells (44) (Figure 2). ITA also inhibits isocitrate dehydrogenases 1/2 in Tregs, decreasing 2-hydroxyglutarate (44), known to inhibit TET enzymes (45). The upregulation of TET allows TET-mediated DNA demethylation, increasing Foxp3 expression in Tregs (44) (Figure 2). Moreover, ITA can ameliorate disease activity in an EAE model (44). ITA also reduces the proliferation and migration of fibroblast-like synoviocytes (FLS) and ameliorates arthritis by inhibiting glycolysis and mitochondrial OXPHOS (46). It has been reported that ITA and its derivatives also ameliorate mouse models of SLE (47), inflammatory bowel diseases (48), and lung fibrosis (49). ITA also exerts antimicrobial effects by direct inhibition of bacterial isocitrate lyase and suppressing viral replication (50). Therefore, ITA could be considered a novel therapeutic target for autoimmune diseases with a low risk of infection.
3.3 α-KG
α-KG is primarily produced in mitochondria via the TCA cycle (Figure 1), where isocitrate dehydrogenase catalyzes the conversion of isocitrate to α-KG, generating NADH. Additionally, α-KG is produced from glutamine through glutaminase and glutamate dehydrogenase or transaminases. This pathway, called anaplerosis, is especially active in proliferating cells. α-KG plays an important role in modulating macrophage polarization. It has been shown to attenuate the pro-inflammatory phenotype of pro-inflammatory (M1) macrophages and to facilitate the establishment of endotoxin tolerance. This immunomodulatory effect is partly attributed to the intracellular α-KG/succinate ratio, where a lower ratio inhibits prolyl hydroxylase (PHD)-dependent hydroxylation of IKK-β, suppressing NF-κB nuclear translocation (51). α-KG also promotes M2 macrophage activation through demethylation of H3K27 and activation of M2-related genes by enhancing the activity of Jmjd3, a histone demethylase (30) (Figure 2). Additionally, α-KG also serves as a cofactor for TET2, the main enzyme catalyzing the hydroxylation of 5-methylcytosine to 5-hydroxymethylcytosine (5hmC) in DNA. This TET2-mediated DNA hydroxymethylation activates transcription of anti-inflammatory genes such as Mrc1, Arg1, Chil3, and Retnla, thereby promoting the polarization of lung interstitial macrophages toward an anti-inflammatory phenotype (52) (Figure 2). In CD4 T cells, α-KG acts as a metabolic regulator that promotes Th1 cell differentiation by enhancing T-bet expression and activating mTORC1 signaling (53). Although α-KG enhances TET2 activity and promotes DNA demethylation at the Foxp3 locus, it paradoxically destabilizes Foxp3 expression. This leads to increased OXPHOS and lipid synthesis, thereby suppressing Treg differentiation (54). Conversely, in RA models, α-KG administered via polymeric nanoparticles has been reported to exert anti-inflammatory effects by suppressing pathogenic Th17 responses and promoting Treg differentiation, reducing joint inflammation (55). In monocytes from patients with SLE, elevated α-KG levels, driven by increased isocitrate dehydrogenase activity, act as cofactors for histone demethylases KDM6A/B, promoting H3K27 demethylation at interferon-stimulated gene (ISG) promoters and sustaining IFNα-induced gene expression (56).
4 Amino acid metabolism
4.1 Glutamine
Glutamine is an immunoregulatory nutrient and is the most abundant amino acid in the serum (57). SLC1A5, also known as alanine-serine-cysteine transporter 2 (ASCT2), is a transporter for neutral amino acids, including glutamine. Slc1a5-/-CD4 T cells do not show impaired T-cell receptor-mediated activation, however, these T cells exhibit impaired Th1 and Th17 cell differentiation (58). Glutamine has the ability to regulate Th1 and Th2 balance. The addition of glutamine to the diet has been shown to favor Th1 cell responses over Th2 cells (59). Conversely, the addition of high concentrations of glutamine in vitro impairs Th2 cell differentiation, inhibiting inflammatory mediators such as leukotrienes, prostaglandins and platelet-activating factor, which are important for Th2 functions (60–62).
4.2 Glutamate
Glutamate is produced from glutamine by glutaminase (Figure 1). Glutaminase 1 expression is increased in Th17 cells, and the inhibition of glutaminase 1 reduces Th17 cell differentiation and ameliorates EAE and lupus-prone mice, MRL/lpr (63, 64). The inhibition of glutaminase 1 induces epigenetic changes in Th17 cells; however, these changes are not restored by a cell-permeable α-KG analog, dimethyl 2-ketoglutarate, suggesting a distinct mechanism of regulation of Th17 cells by glutaminase 1 (65). The inhibition of glutaminase 1 also reduced plasmablast differentiation in MRL/lpr mice (66). The glutaminase 1 inhibitor also reduced the number of activated B cells and Tfh cells in MRL/lpr mice (66, 67).
4.3 Aspartate
Aspartate is a non-essential amino acid that serves as a key intermediate linking cellular metabolism with immune cell function. It is primarily produced in the mitochondria by the transamination of oxaloacetate by glutamate (Figure 1), catalyzed by mitochondrial aspartate aminotransferase, and exported to the cytosol via the SLC25A12/13 transporters (68). Aspartate plays a central role in regulating TNF production in RA CD4 T cells (69). A deficiency in the production of mitochondrial aspartate disrupts NAD+ regeneration and ER stability, thereby promoting TNF translation (69). Supplementation with aspartate or NAD+ suppressed TNF release and tissue inflammation (69). In macrophages, aspartate acts as a metabolic regulator of inflammatory responses by promoting HIF-1α stabilization and promoting IL-1β production (70).
4.4 Tryptophan
Tryptophan is an essential amino acid and a precursor to kynurenine, serotonin, melatonin, and vitamin B3 (3). Kynurenine is a metabolite of tryptophan pathway used in the production of vitamin B3 (3). Metabolomic screening has identified an altered distribution of tryptophan metabolites in the feces of lupus-prone B6.Sle1.Sle2.Sle3 mice, including increased kynurenine levels, which are alleviated following antibiotic treatment. Interestingly, low dietary tryptophan intake ameliorates disease activity in these lupus-prone mice, whereas high dietary tryptophan intake exacerbates it.
5 One-carbon metabolism
5.1 Methionine
One-carbon metabolism serves as a crucial mechanism for cellular energy and production of vital signaling molecules, including single-carbon moieties (71). One-carbon metabolism includes folate and methionine metabolism. It also has important roles in DNA and RNA methylation, histone modification and redox homeostasis regulation (71). Intermediate metabolites in one-carbon metabolism, including methionine and SAM, mediate the T cell immune response.
Restriction of methionine inhibits Th17 cell differentiation and improves disease activity of EAE (72). Methionine is rapidly taken up by activated T cells and serves as the major substrate for the biosynthesis of SAM (72). Methylenetetrahydrofolate dehydrogenase 2 (MTHFD2), an enzyme of one-carbon metabolism, regulates de novo purine synthesis (73). In pathogenic Th17 cells, MTHFD2 prevents aberrant upregulation of Foxp3 and MTHFD2 deficiency promotes Treg cell differentiation (73).
5.2 SAM
SAM is the primary methyl donor for numerous cellular methylation reactions. Intracellular SAM levels are maintained within an optimal range by many homeostatic mechanisms (74). SAM is synthesized from methionine by methionine adenosyltransferase (MAT) (Figure 1), and MAT is inhibited by itaconate in Th17 cells (44). Reduced SAM levels decrease histone methylation at the Il17a promoter and reduce Th17 cell differentiation (72) (Figure 2). The SAM/SAH ratio is used as a methylation index, which means a key indicator of a cell’s methylation capacity (44). Thus some metabolites influence epigenetic changes.
6 Lipid metabolism
6.1 25-Hydroxycholesterol
25-OHC, produced by the enzyme cholesterol 25-hydroxylase (Ch25h) in activated CD4 T cells under IL-27 and TGF-β stimulation, acts as an immunomodulator. 25-OHC suppresses cholesterol biosynthesis in T cells by downregulating key enzymes such as HMG-CoA reductase via inhibition of the sterol regulatory element binding proteins (SREBP) 2 pathway, leading to selective apoptosis of proliferating T cells while sparing resting cells. Exogenous administration of 25-OHC by intraperitoneal injection reduces the severity of skin inflammation in a contact hypersensitivity model (75). Ch25h deficiency leads to excessive IL-1β production from macrophages, as 25-OHC normally suppresses IL-1β transcription and inflammasome activation (76). Ch25h-deficient mice show heightened sensitivity to septic shock and exacerbated EAE due to unrestrained AIM2-dependent inflammasome activity (77).
6.2 Sphingosine-1-phosphate
S1P biosynthesis involves sphingomyelinase-mediated ceramide generation from sphingomyelin, ceramidase conversion to sphingosine, and ATP-dependent phosphorylation by sphingosine kinases, localized to distinct subcellular compartments (78). This pathway bridges membrane lipid turnover to S1P’s immunomodulatory and pro-inflammatory signaling. S1P, via S1P receptor 1, is essential for T cell egress from the thymus and lymphoid organs, thereby controlling their circulation and tissue infiltration. S1P receptor modulators, such as fingolimod and siponimod, treat multiple sclerosis by binding S1PR1 on lymphocytes, prevent them from egress from lymph nodes and reducing their migration into the central nervous system (79). S1P levels were elevated in both SLE patients with active nephritis involvement and in the murine lupus models (80, 81). Sphingosine analogs ameliorate a murine lupus nephritis model, reducing T cell infiltration into the kidney (82). In RA-FLS, S1P stimulation promotes proliferation, migration, and production of IL-6 and MMP-3 (83).
7 Conclusion
The field of immunometabolism has illuminated the critical role of intracellular metabolites in modulating immune cell function and driving autoimmune pathogenesis. This review has highlighted representative immunometabolites from key metabolic pathways and their various effects on immune cells relevant to diseases such as RA and SLE. Targeting these immunometabolites could be an effective therapeutic strategy, as supported by findings from the animal models described above.
However, significant challenges remain. Given that certain metabolites such as lactate and succinate, have dual roles, careful consideration of the specific disease context and target cell type is essential. Technical hurdles include developing selective inhibitors of metabolic enzymes and designing delivery systems to achieve optimal metabolite concentrations within target immune cells. Furthermore, potential off-target effects and long-term consequences of metabolic interventions require thorough investigation.
Future research should focus on elucidating the complex interactions between immunometabolites and immune cell signaling. A more comprehensive understanding of the mechanisms underlying immunometabolite action could facilitate the development of highly specific and effective therapeutic strategies.
Author contributions
MT: Writing – review & editing, Writing – original draft. MK:Writing – original draft, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. This work was supported by the Japan Agency for Medical Research and Development (AMED) under grant numbers [JP20ek0410078 (to M. Kono), JP23ek0109607 (to M. Kono), and JP23ek0410111 (to M. Kono)]. We thank BioRender.com for the creation of Figure 1 and 2.
Conflict of interest
MK reports grants and/or speaking fees from AbbVie Inc., Asahi-Kasei Co., Astellas Pharma Inc., AstraZeneca Plc., Ayumi Pharmaceutical Co., Ltd., Bristol-Myers Squibb Co., Ltd., Chugai Pharmaceutical Co., Ltd., Daiichi Sankyo Co., Ltd., Eisai Co. Ltd., Eli Lilly Japan K.K., Gilead Sciences K.K., GlaxoSmithKline K.K., Janssen Pharmaceutical K.K., Kowa Co. Ltd., Kyocera Co., Ltd., Lotte CO., LTD., Nippon Boehringer Ingelheim Co., Ltd., Nippon Shinyaku CO., LTD., Mitsubishi Tanabe Pharma Co., Mochida Pharmaceutical CO., LTD., Pfizer Inc., Sandoz., Taiju Life Social Welfare Foundation, Taisho Pharmaceutical, Takeda Pharmaceutical Co., Ltd., Terumo Co., Ltd., UCB Japan Co. Ltd. and Yamazaki Baking CO., LTD., outside the submitted work.
The remaining author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that Generative AI was used in the creation of this manuscript. This manuscript was developed with the support of AI-assisted tools, such as Claude 3.7 Sonet (Anthropic, CA, USA), which facilitated grammar correction, refinement of expression, and overall improvement in the quality of the manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Hisada R and Kono M. Recent advances in immunometabolism in rheumatic diseases. Curr Opin Rheumatol. (2025) 37:142–8. doi: 10.1097/bor.0000000000001071
2. Kono M. New insights into the metabolism of Th17 cells. Immunol Med. (2023) 46:15–24. doi: 10.1080/25785826.2022.2140503
3. Hisada R and Kono M. Potential therapies targeting metabolic pathways in systemic lupus erythematosus. Clin Immunol. (2024) 263:110224. doi: 10.1016/j.clim.2024.110224
4. Galgani M, Bruzzaniti S, and Matarese G. Immunometabolism and autoimmunity. Curr Opin Immunol. (2020) 67:10–7. doi: 10.1016/j.coi.2020.07.002
5. Song Y, Li J, and Wu Y. Evolving understanding of autoimmune mechanisms and new therapeutic strategies of autoimmune disorders. Signal Transduction Targeted Ther. (2024) 9:263. doi: 10.1038/s41392-024-01952-8
6. Mizui M and Kono M. Novel therapeutic strategies targeting abnormal T-cell signaling in systemic lupus erythematosus. Clin Immunol. (2024) 262:110182. doi: 10.1016/j.clim.2024.110182
7. Kono M, Yoshida N, and Tsokos GC. Metabolic control of T cells in autoimmunity. Curr Opin Rheumatol. (2020) 32:192–9. doi: 10.1097/bor.0000000000000685
8. Vukelic M, Kono M, and Tsokos GC. T cell metabolism in lupus. Immunometabolism. (2020) 2(2):e200009. doi: 10.20900/immunometab20200009
9. Kono M, Yoshida N, Maeda K, Skinner NE, Pan W, Kyttaris VC, et al. Pyruvate dehydrogenase phosphatase catalytic subunit 2 limits Th17 differentiation. Proc Natl Acad Sci U.S.A. (2018) 115:9288–93. doi: 10.1073/pnas.1805717115
10. Rabinowitz JD and Enerbäck S. Lactate: the ugly duckling of energy metabolism. Nat Metab. (2020) 2:566–71. doi: 10.1038/s42255-020-0243-4
11. Garcia-Carbonell R, Divakaruni AS, Lodi A, Vicente-Suarez I, Saha A, Cheroutre H, et al. Critical role of glucose metabolism in rheumatoid arthritis fibroblast-like synoviocytes. Arthritis Rheumatol. (2016) 68:1614–26. doi: 10.1002/art.39608
12. Amorini AM, Nociti V, Petzold A, Gasperini C, Quartuccio E, Lazzarino G, et al. Serum lactate as a novel potential biomarker in multiple sclerosis. Biochim Biophys Acta. (2014) 1842:1137–43. doi: 10.1016/j.bbadis.2014.04.005
13. Pucino V, Bombardieri M, Pitzalis C, and Mauro C. Lactate at the crossroads of metabolism, inflammation, and autoimmunity. Eur J Immunol. (2017) 47:14–21. doi: 10.1002/eji.201646477
14. Pucino V, Certo M, Bulusu V, Cucchi D, Goldmann K, Pontarini E, et al. Lactate buildup at the site of chronic inflammation promotes disease by inducing CD4(+) T cell metabolic rewiring. Cell Metab. (2019) 30:1055–74.e8. doi: 10.1016/j.cmet.2019.10.004
15. Haas R, Smith J, Rocher-Ros V, Nadkarni S, Montero-Melendez T, D’Acquisto F, et al. Lactate regulates metabolic and pro-inflammatory circuits in control of T cell migration and effector functions. PloS Biol. (2015) 13:e1002202. doi: 10.1371/journal.pbio.1002202
16. Zhang YT, Xing ML, Fang HH, Li WD, Wu L, and Chen ZP. Effects of lactate on metabolism and differentiation of CD4(+)T cells. Mol Immunol. (2023) 154:96–107. doi: 10.1016/j.molimm.2022.12.015
17. Pradenas C, Luque-Campos N, Oyarce K, Contreras-Lopez R, Bustamante-Barrientos FA, Bustos A, et al. Lactate: an alternative pathway for the immunosuppressive properties of mesenchymal stem/stromal cells. Stem Cell Res Ther. (2023) 14:335. doi: 10.1186/s13287-023-03549-4
18. Romero M, Miller K, Gelsomini A, Garcia D, Li K, Suresh D, et al. Immunometabolic effects of lactate on humoral immunity in healthy individuals of different ages. Nat Commun. (2024) 15:7515. doi: 10.1038/s41467-024-51207-x
19. Shi W, Cassmann TJ, Bhagwate AV, Hitosugi T, and Ip WKE. Lactic acid induces transcriptional repression of macrophage inflammatory response via histone acetylation. Cell Rep. (2024) 43:113746. doi: 10.1016/j.celrep.2024.113746
20. Gray LR, Tompkins SC, and Taylor EB. Regulation of pyruvate metabolism and human disease. Cell Mol Life Sci. (2014) 71:2577–604. doi: 10.1007/s00018-013-1539-2
21. Kim MJ, Lee H, Chanda D, Thoudam T, Kang HJ, Harris RA, et al. The role of pyruvate metabolism in mitochondrial quality control and inflammation. Mol Cells. (2023) 46:259–67. doi: 10.14348/molcells.2023.2128
22. Gajić D, Despotović S, Koprivica I, Miljković Đ, and Saksida T. Ethyl pyruvate ameliorates experimental autoimmune myocarditis. Biomolecules. (2021) 11(12):1768. doi: 10.3390/biom11121768
23. Djedović N, Stanisavljevic S, Jevtić B, Momčilović M, Lavrnja I, and Miljković D. Anti-encephalitogenic effects of ethyl pyruvate are reflected in the central nervous system and the gut. BioMed Pharmacother. (2017) 96:78–85. doi: 10.1016/j.biopha.2017.09.110
24. Koprivica I, Vujičić M, Gajić D, Saksida T, and Stojanović I. Ethyl pyruvate stimulates regulatory T cells and ameliorates type 1 diabetes development in mice. Front Immunol. (2019) 9:3130. doi: 10.3389/fimmu.2018.03130
25. Abusalamah H, Reel JM, and Lupfer CR. Pyruvate affects inflammatory responses of macrophages during influenza A virus infection. Virus Res. (2020) 286:198088. doi: 10.1016/j.virusres.2020.198088
26. Huang H, Li G, He Y, Chen J, Yan J, Zhang Q, et al. Cellular succinate metabolism and signaling in inflammation: implications for therapeutic intervention. Front Immunol. (2024) 15:1404441. doi: 10.3389/fimmu.2024.1404441
27. Tannahill GM, Curtis AM, Adamik J, Palsson-McDermott EM, McGettrick AF, Goel G, et al. Succinate is an inflammatory signal that induces IL-1β through HIF-1α. Nature. (2013) 496:238–42. doi: 10.1038/nature11986
28. Wise DR, Ward PS, Shay JE, Cross JR, Gruber JJ, Sachdeva UM, et al. Hypoxia promotes isocitrate dehydrogenase-dependent carboxylation of α-ketoglutarate to citrate to support cell growth and viability. Proc Natl Acad Sci U.S.A. (2011) 108:19611–6. doi: 10.1073/pnas.1117773108
29. Eniafe J and Jiang S. The functional roles of TCA cycle metabolites in cancer. Oncogene. (2021) 40:3351–63. doi: 10.1038/s41388-020-01639-8
30. Liu PS, Wang H, Li X, Chao T, Teav T, Christen S, et al. α-ketoglutarate orchestrates macrophage activation through metabolic and epigenetic reprogramming. Nat Immunol. (2017) 18:985–94. doi: 10.1038/ni.3796
31. Wang F, Wang K, Xu W, Zhao S, Ye D, Wang Y, et al. SIRT5 desuccinylates and activates pyruvate kinase M2 to block macrophage IL-1β Production and to prevent DSS-induced colitis in mice. Cell Rep. (2017) 19:2331–44. doi: 10.1016/j.celrep.2017.05.065
32. Peruzzotti-Jametti L, Bernstock JD, Vicario N, Costa ASH, Kwok CK, Leonardi T, et al. Macrophage-derived extracellular succinate licenses neural stem cells to suppress chronic neuroinflammation. Cell Stem Cell. (2018) 22:355–68.e13. doi: 10.1016/j.stem.2018.01.020
33. Chen X, Sunkel B, Wang M, Kang S, Wang T, Gnanaprakasam JNR, et al. Succinate dehydrogenase/complex II is critical for metabolic and epigenetic regulation of T cell proliferation and inflammation. Sci Immunol. (2022) 7:eabm8161. doi: 10.1126/sciimmunol.abm8161
34. Li H, Tan H, Liu Z, Pan S, Tan S, Zhu Y, et al. Succinic acid exacerbates experimental autoimmune uveitis by stimulating neutrophil extracellular traps formation via SUCNR1 receptor. Br J Ophthalmol. (2023) 107:1744–9. doi: 10.1136/bjophthalmol-2021-320880
35. Gudgeon N, Munford H, Bishop EL, Hill J, Fulton-Ward T, Bending D, et al. Succinate uptake by T cells suppresses their effector function via inhibition of mitochondrial glucose oxidation. Cell Rep. (2022) 40:111193. doi: 10.1016/j.celrep.2022.111193
36. Rubic T, Lametschwandtner G, Jost S, Hinteregger S, Kund J, Carballido-Perrig N, et al. Triggering the succinate receptor GPR91 on dendritic cells enhances immunity. Nat Immunol. (2008) 9:1261–9. doi: 10.1038/ni.1657
37. Saraiva AL, Veras FP, Peres RS, Talbot J, de Lima KA, Luiz JP, et al. Succinate receptor deficiency attenuates arthritis by reducing dendritic cell traffic and expansion of Th17 cells in the lymph nodes. FASEB J. (2018) 32:fj201800285. doi: 10.1096/fj.201800285
38. Michelucci A, Cordes T, Ghelfi J, Pailot A, Reiling N, Goldmann O, et al. Immune-responsive gene 1 protein links metabolism to immunity by catalyzing itaconic acid production. Proc Natl Acad Sci U.S.A. (2013) 110:7820–5. doi: 10.1073/pnas.1218599110
39. Auger JP, Zimmermann M, Faas M, Stifel U, Chambers D, Krishnacoumar B, et al. Metabolic rewiring promotes anti-inflammatory effects of glucocorticoids. Nature. (2024) 629:184–92. doi: 10.1038/s41586-024-07282-7
40. Lampropoulou V, Sergushichev A, Bambouskova M, Nair S, Vincent E, Loginicheva E, et al. Itaconate links inhibition of succinate dehydrogenase with macrophage metabolic remodeling and regulation of inflammation. Cell Metab. (2016) 24:158–66. doi: 10.1016/j.cmet.2016.06.004
41. Liao ST, Han C, Xu DQ, Fu XW, Wang JS, and Kong LY. 4-Octyl itaconate inhibits aerobic glycolysis by targeting GAPDH to exert anti-inflammatory effects. Nat Commun. (2019) 10:5091. doi: 10.1038/s41467-019-13078-5
42. Ryan DG, Knatko EV, Casey AM, Hukelmann JL, Dayalan Naidu S, Brenes AJ, et al. Nrf2 activation reprograms macrophage intermediary metabolism and suppresses the type I interferon response. iScience. (2022) 25:103827. doi: 10.1016/j.isci.2022.103827
43. Hooftman A, Angiari S, Hester S, Corcoran SE, Runtsch MC, Ling C, et al. The immunomodulatory metabolite itaconate modifies NLRP3 and inhibits inflammasome activation. Cell Metab. (2020) 32:468–78.e7. doi: 10.1016/j.cmet.2020.07.016
44. Aso K, Kono M, Kanda M, Kudo Y, Sakiyama K, Hisada R, et al. Itaconate ameliorates autoimmunity by modulating T cell imbalance via metabolic and epigenetic reprogramming. Nat Commun. (2023) 14:984. doi: 10.1038/s41467-023-36594-x
45. Xu W, Yang H, Liu Y, Yang Y, Wang P, Kim SH, et al. Oncometabolite 2-hydroxyglutarate is a competitive inhibitor of α-ketoglutarate-dependent dioxygenases. Cancer Cell. (2011) 19:17–30. doi: 10.1016/j.ccr.2010.12.014
46. Tada M, Kudo Y, Kono M, Kanda M, Takeyama S, Sakiyama K, et al. Itaconate reduces proliferation and migration of fibroblast-like synoviocytes and ameliorates arthritis models. Clin Immunol. (2024) 264:110255. doi: 10.1016/j.clim.2024.110255
47. Blanco LP, Patino-Martinez E, Nakabo S, Zhang M, Pedersen HL, Wang X, et al. Modulation of the itaconate pathway attenuates murine lupus. Arthritis Rheumatol. (2022) 74:1971–83. doi: 10.1002/art.42284
48. Yang W, Wang Y, Wang T, Li C, Shi L, Zhang P, et al. Protective effects of IRG1/itaconate on acute colitis through the inhibition of gasdermins-mediated pyroptosis and inflammation response. Genes Dis. (2023) 10:1552–63. doi: 10.1016/j.gendis.2022.05.039
49. Ogger PP, Albers GJ, Hewitt RJ, O’Sullivan BJ, Powell JE, Calamita E, et al. Itaconate controls the severity of pulmonary fibrosis. Sci Immunol. (2020) 5(52):eabc1884. doi: 10.1126/sciimmunol.abc1884
50. Rittenhouse JW and McFadden BA. Inhibition of isocitrate lyase from Pseudomonas indigofera by itaconate. Arch Biochem Biophys. (1974) 163:79–86. doi: 10.1016/0003-9861(74)90456-1
51. Jha AK, Huang SC, Sergushichev A, Lampropoulou V, Ivanova Y, Loginicheva E, et al. Network integration of parallel metabolic and transcriptional data reveals metabolic modules that regulate macrophage polarization. Immunity. (2015) 42:419–30. doi: 10.1016/j.immuni.2015.02.005
52. Zhou B, Magana L, Hong Z, Huang LS, Chakraborty S, Tsukasaki Y, et al. The angiocrine Rspondin3 instructs interstitial macrophage transition via metabolic-epigenetic reprogramming and resolves inflammatory injury. Nat Immunol. (2020) 21:1430–43. doi: 10.1038/s41590-020-0764-8
53. Klysz D, Tai X, Robert PA, Craveiro M, Cretenet G, Oburoglu L, et al. Glutamine-dependent α-ketoglutarate production regulates the balance between T helper 1 cell and regulatory T cell generation. Sci Signal. (2015) 8:ra97. doi: 10.1126/scisignal.aab2610
54. Matias MI, Yong CS, Foroushani A, Goldsmith C, Mongellaz C, Sezgin E, et al. Regulatory T cell differentiation is controlled by αKG-induced alterations in mitochondrial metabolism and lipid homeostasis. Cell Rep. (2021) 37:109911. doi: 10.1016/j.celrep.2021.109911
55. Mangal JL, Inamdar S, Suresh AP, Jaggarapu M, Esrafili A, Ng ND, et al. Short term, low dose alpha-ketoglutarate based polymeric nanoparticles with methotrexate reverse rheumatoid arthritis symptoms in mice and modulate T helper cell responses. Biomater Sci. (2022) 10:6688–97. doi: 10.1039/d2bm00415a
56. Montano EN, Bose M, Huo L, Tumurkhuu G, De Los Santos G, Simental B, et al. α-ketoglutarate-dependent KDM6 histone demethylases and interferon-stimulated gene expression in lupus. Arthritis Rheumatol. (2024) 76:396–410. doi: 10.1002/art.42724
57. Kono M, Yoshida N, and Tsokos GC. Amino acid metabolism in lupus. Front Immunol. (2021) 12:623844. doi: 10.3389/fimmu.2021.623844
58. Nakaya M, Xiao Y, Zhou X, Chang JH, Chang M, Cheng X, et al. Inflammatory T cell responses rely on amino acid transporter ASCT2 facilitation of glutamine uptake and mTORC1 kinase activation. Immunity. (2014) 40:692–705. doi: 10.1016/j.immuni.2014.04.007
59. Caris AV, Lira FS, de Mello MT, Oyama LM, and dos Santos RV. Carbohydrate and glutamine supplementation modulates the Th1/Th2 balance after exercise performed at a simulated altitude of 4500 m. Nutrition. (2014) 30:1331–6. doi: 10.1016/j.nut.2014.03.019
60. Chang WK, Yang KD, and Shaio MF. Effect of glutamine on Th1 and Th2 cytokine responses of human peripheral blood mononuclear cells. Clin Immunol. (1999) 93:294–301. doi: 10.1006/clim.1999.4788
61. Ko HM, Kang NI, Kim YS, Lee YM, Jin ZW, Jung YJ, et al. Glutamine preferentially inhibits T-helper type 2 cell-mediated airway inflammation and late airway hyperresponsiveness through the inhibition of cytosolic phospholipase A(2) activity in a murine asthma model. Clin Exp Allergy. (2008) 38:357–64. doi: 10.1111/j.1365-2222.2007.02900.x
62. Stark JM, Tibbitt CA, and Coquet JM. The metabolic requirements of th2 cell differentiation. Front Immunol. (2019) 10:2318. doi: 10.3389/fimmu.2019.02318
63. Kono M, Yoshida N, Maeda K, and Tsokos GC. Transcriptional factor ICER promotes glutaminolysis and the generation of Th17 cells. Proc Natl Acad Sci U.S.A. (2018) 115:2478–83. doi: 10.1073/pnas.1714717115
64. Kono M, Yoshida N, Maeda K, Suárez-Fueyo A, Kyttaris VC, and Tsokos GC. Glutaminase 1 inhibition reduces glycolysis and ameliorates lupus-like disease in MRL/lpr mice and experimental autoimmune encephalomyelitis. Arthritis Rheumatol. (2019) 71:1869–78. doi: 10.1002/art.41019
65. Johnson MO, Wolf MM, Madden MZ, Andrejeva G, Sugiura A, Contreras DC, et al. Distinct regulation of th17 and th1 cell differentiation by glutaminase-dependent metabolism. Cell. (2018) 175:1780–95.e19. doi: 10.1016/j.cell.2018.10.001
66. Sumikawa MH, Iwata S, Zhang M, Miyata H, Ueno M, Todoroki Y, et al. An enhanced mitochondrial function through glutamine metabolism in plasmablast differentiation in systemic lupus erythematosus. Rheumatol (Oxford). (2022) 61:3049–59. doi: 10.1093/rheumatology/keab824
67. Zhang X, Wang G, Bi Y, Jiang Z, and Wang X. Inhibition of glutaminolysis ameliorates lupus by regulating T and B cell subsets and downregulating the mTOR/P70S6K/4EBP1 and NLRP3/caspase-1/IL-1β pathways in MRL/lpr mice. Int Immunopharmacol. (2022) 112:109133. doi: 10.1016/j.intimp.2022.109133
68. Chen J, Cui L, Lu S, and Xu S. Amino acid metabolism in tumor biology and therapy. Cell Death Dis. (2024) 15:42. doi: 10.1038/s41419-024-06435-w
69. Wu B, Zhao TV, Jin K, Hu Z, Abdel MP, Warrington KJ, et al. Mitochondrial aspartate regulates TNF biogenesis and autoimmune tissue inflammation. Nat Immunol. (2021) 22:1551–62. doi: 10.1038/s41590-021-01065-2
70. Wang H, Zheng X, Liu B, Xia Y, Xin Z, Deng B, et al. Aspartate metabolism facilitates IL-1β Production in inflammatory macrophages. Front Immunol. (2021) 12:753092. doi: 10.3389/fimmu.2021.753092
71. Qiu Y, Xie E, Xu H, Cheng H, and Li G. One-carbon metabolism shapes T cell immunity in cancer. Trends Endocrinol Metab. (2024) 35:967–80. doi: 10.1016/j.tem.2024.05.010
72. Roy DG, Chen J, Mamane V, Ma EH, Muhire BM, Sheldon RD, et al. Methionine metabolism shapes T helper cell responses through regulation of epigenetic reprogramming. Cell Metab. (2020) 31:250–66.e9. doi: 10.1016/j.cmet.2020.01.006
73. Sugiura A, Andrejeva G, Voss K, Heintzman DR, Xu X, Madden MZ, et al. MTHFD2 is a metabolic checkpoint controlling effector and regulatory T cell fate and function. Immunity. (2022) 55:65–81.e9. doi: 10.1016/j.immuni.2021.10.011
74. Xing Z and Tu BP. Mechanisms and rationales of SAM homeostasis. Trends Biochem Sci. (2025) 50:242–54. doi: 10.1016/j.tibs.2024.12.009
75. Takahashi H, Nomura H, Iriki H, Kubo A, Isami K, Mikami Y, et al. Cholesterol 25-hydroxylase is a metabolic switch to constrain T cell-mediated inflammation in the skin. Sci Immunol. (2021) 6:eabb6444. doi: 10.1126/sciimmunol.abb6444
76. Reboldi A, Dang EV, McDonald JG, Liang G, Russell DW, and Cyster JG. Inflammation. 25-Hydroxycholesterol suppresses interleukin-1-driven inflammation downstream of type I interferon. Science. (2014) 345:679–84. doi: 10.1126/science.1254790
77. Dang EV, McDonald JG, Russell DW, and Cyster JG. Oxysterol restraint of cholesterol synthesis prevents AIM2 inflammasome activation. Cell. (2017) 171:1057–71.e11. doi: 10.1016/j.cell.2017.09.029
78. Proia RL and Hla T. Emerging biology of sphingosine-1-phosphate: its role in pathogenesis and therapy. J Clin Invest. (2015) 125:1379–87. doi: 10.1172/jci76369
79. Mao-Draayer Y, Sarazin J, Fox D, and Schiopu E. The sphingosine-1-phosphate receptor: A novel therapeutic target for multiple sclerosis and other autoimmune diseases. Clin Immunol. (2017) 175:10–5. doi: 10.1016/j.clim.2016.11.008
80. Patyna S, Büttner S, Eckes T, Obermüller N, Bartel C, Braner A, et al. Blood ceramides as novel markers for renal impairment in systemic lupus erythematosus. Prostaglandins Other Lipid Mediat. (2019) 144:106348. doi: 10.1016/j.prostaglandins.2019.106348
81. Strasser DS, Froidevaux S, Sippel V, Gerossier E, Grieder U, Pierlot GM, et al. Preclinical to clinical translation of cenerimod, a novel S1P(1) receptor modulator, in systemic lupus erythematosus. RMD Open. (2020) 6(2):e001262. doi: 10.1136/rmdopen-2020-001261
82. Ando S, Amano H, Amano E, Minowa K, Watanabe T, Nakano S, et al. FTY720 exerts a survival advantage through the prevention of end-stage glomerular inflammation in lupus-prone BXSB mice. Biochem Biophys Res Commun. (2010) 394:804–10. doi: 10.1016/j.bbrc.2010.03.078
83. Inoue T, Kohno M, Nagahara H, Murakami K, Sagawa T, Kasahara A, et al. Upregulation of sphingosine-1-phosphate receptor 3 on fibroblast-like synoviocytes is associated with the development of collagen-induced arthritis via increased interleukin-6 production. PloS One. (2019) 14:e0218090. doi: 10.1371/journal.pone.0218090
Keywords: cellular metabolism, metabolite, itaconate, glycolysis, glutaminolysis
Citation: Tada M and Kono M (2025) Metabolites as regulators of autoimmune diseases. Front. Immunol. 16:1637436. doi: 10.3389/fimmu.2025.1637436
Received: 29 May 2025; Accepted: 07 August 2025;
Published: 21 August 2025.
Edited by:
Juliana Lauar Gonçalves, Centro Universitário Una, BrazilReviewed by:
Bingdong Zhu, Lanzhou University, ChinaCopyright © 2025 Tada and Kono. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Michihito Kono, bS1rb25vQG1lZC5ob2t1ZGFpLmFjLmpw
†These authors have contributed equally to this work
 Maria Tada†
Maria Tada† Michihito Kono
Michihito Kono