- 1Department of Dermatology, College of Medicine, The Ohio State University Wexner Medical Center, Columbus, OH, United States
- 2Division of Hematology/Oncology, Nationwide Children’s Hospital, Columbus, OH, United States
Natural killer (NK) cells are lymphoid-derived cells that play a critical role in bridging innate and adaptive immunity. Given their ability to recognize and directly kill targets possessing missing or altered self-proteins and to induce indirect killing via recruitment of adaptive immunity, they are in a unique position to modulate host immunologic responses. These complex immune sentinels typically circulate in the peripheral blood and/or reside in lymphoid tissues. As the largest organ, human skin functions in front line immunological defense, though it has not historically been categorized as lymphoid tissue. Whether tissue-resident ILC populations originally derive from conventional circulating NK cells, or whether they interface as developmentally distinct entities with phenotypic overlap within particular inflammatory contexts remains a subject of ongoing investigation. This review seeks to consolidate the currently available literature regarding NK cell and ILC skin homing and innate immune function in healthy vs. lesional human skin (including infection, inflammatory/autoimmune conditions, and cutaneous malignancy). Importantly, we elucidate significant gaps in the understanding of the complex role for NK cells in skin homeostasis and pathology, and posit unique opportunities the accessibility of this secondary lymphoid organ provides for translational studies to improve our understanding of cutaneous immunity.
Background
NK cells are immune effectors of the ILC family that play critical roles in both innate and adaptive immunity. They are generally found circulating in peripheral blood, but are recruited to specific tissues in response to stress signals initiated following host recognition of pathogenic cells, especially viruses and tumors (1). With their unique ability to recognize missing or altered self-proteins, or overexpressed non-self-proteins, NK cells play a critical role in eliminating pathologic cell populations through both direct and indirect mechanisms. These lethal cells eliminate their targets through both direct and indirect means. NK cells mediate direct killing via cytotoxic granule exocytosis, and indirectly by inducing death-receptor-mediated apoptosis via Fas or tumor necrosis factor (TNF) ligands (2). They also mediate antibody-dependent cellular toxicity, induced by NK cell expression of the Fc receptor CD16 (3). Additionally, NK cells secrete interferon (IFN)-γ and TNFα, which activate other immune targets crucial for initiating the adaptive immune response (4).
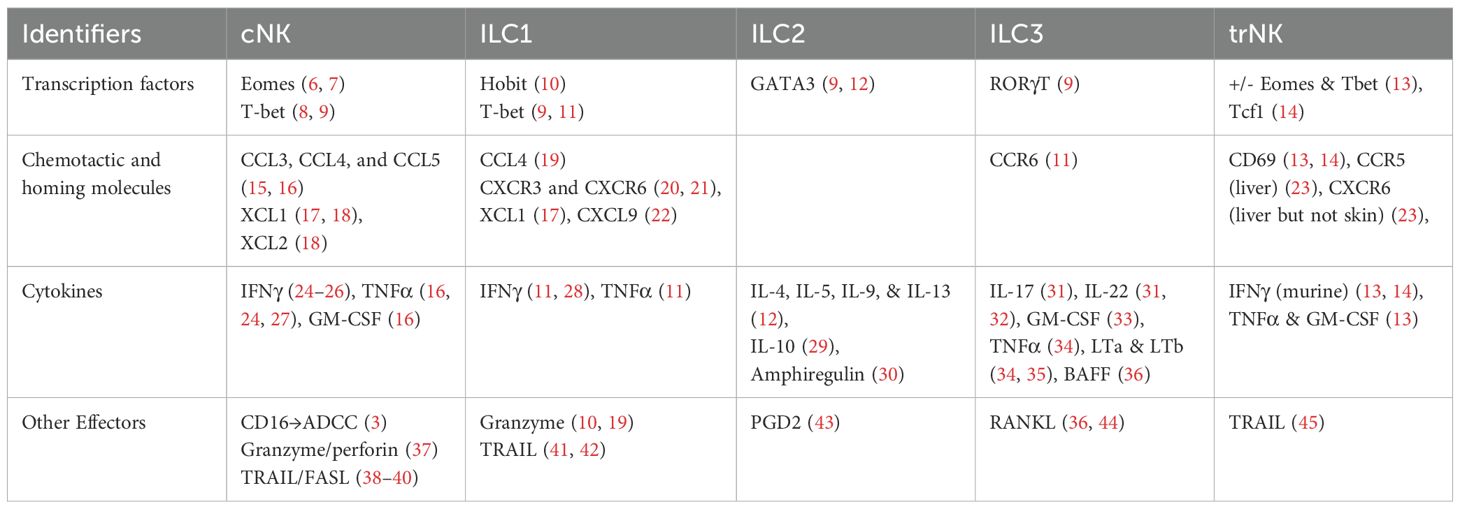
Members of the ILC family are typically differentiated by their cytokine secretion signature and transcription factor response (5) (Table 1) (6–45). For example, NK cells circulating in the peripheral blood are dependent on the transcription factor eomesodermin (Eomes) and secrete TNFα, IFNγ, perforin, and granzyme B upon activation (6, 7, 46).While these “conventional” NK cell (cNK) populations circulate in the blood, the remainder of the ILC subsets (ILC1, ILC2, ILC3, and lymphoid tissue inducer (LTi) cells) tend to be tissue-resident and demonstrate differential transcription factor dependencies associated with particular tissue localization (17). cNK cells are defined by expression of NK1.1, Nkp46, and CD49b in mice (47), whereas tissue-resident ILC1 cells express CD49a, which promotes lymphocyte homing to non-lymphoid tissues, and CD69, which retains lymphocytes within the tissue (6, 19, 48–50). A recent study demonstrated remarkably conserved expression of CD56brightTcf7hiCD69hi tissue resident NK cells in both murine and human skin (14), making murine models a helpful and generally translatable means of studying the role of NK cells in skin pathology. While CD69 is a marker of tissue residency, Tcf7 heralds stemness and is typically down-regulated in tissue-resident T-cells (51). Similarly, CD56dim NK cell populations also appear to be conserved between mice and humans and are typically isolated from circulation (14, 52).
A recent hallmark study made great strides in this classification dilemma via integration of robust scRNAseq and CITEseq data from healthy human blood, tissues (specifically lungs, intestinal epithelium, and tonsils), and a number of tumors (53). Their data identified three distinct NK cell subsets within human blood: NK1, NK2, and NK3 cells, each representing a different state of “maturity” and distinct capacity for the various inherent NK cell functions (cytotoxicity, cytokine secretion/proliferative capacity, and adaptability, respectively). In the majority of the tumors analyzed, the proportion of NK2 cells (CD56brightCD16neg) were increased at the tumor bed compared to peripheral blood, with a bias in their innate transcriptional profile toward expressing genes implicated in cell migration and tissue homing (53). As discussed by Chaudhry et al., NK cells entering tissues appear to take on distinct phenotypes and adopt responsiveness to tissue-specific transcriptional programs upon entry. This plasticity can make it difficult to distinguish NK from tissue-resident ILC1 cells in the different tissue types, such as in a subset of NK cells in human lung that express the classically associated transcription factor Hobit (54). In contrast, while Hobit is also expressed by ILC1s in mouse lamina propria (10, 55), intestinal ILC1 cells in humans instead express Prdm1 (Blimp1) (56). These findings are yet to be characterized in skin.
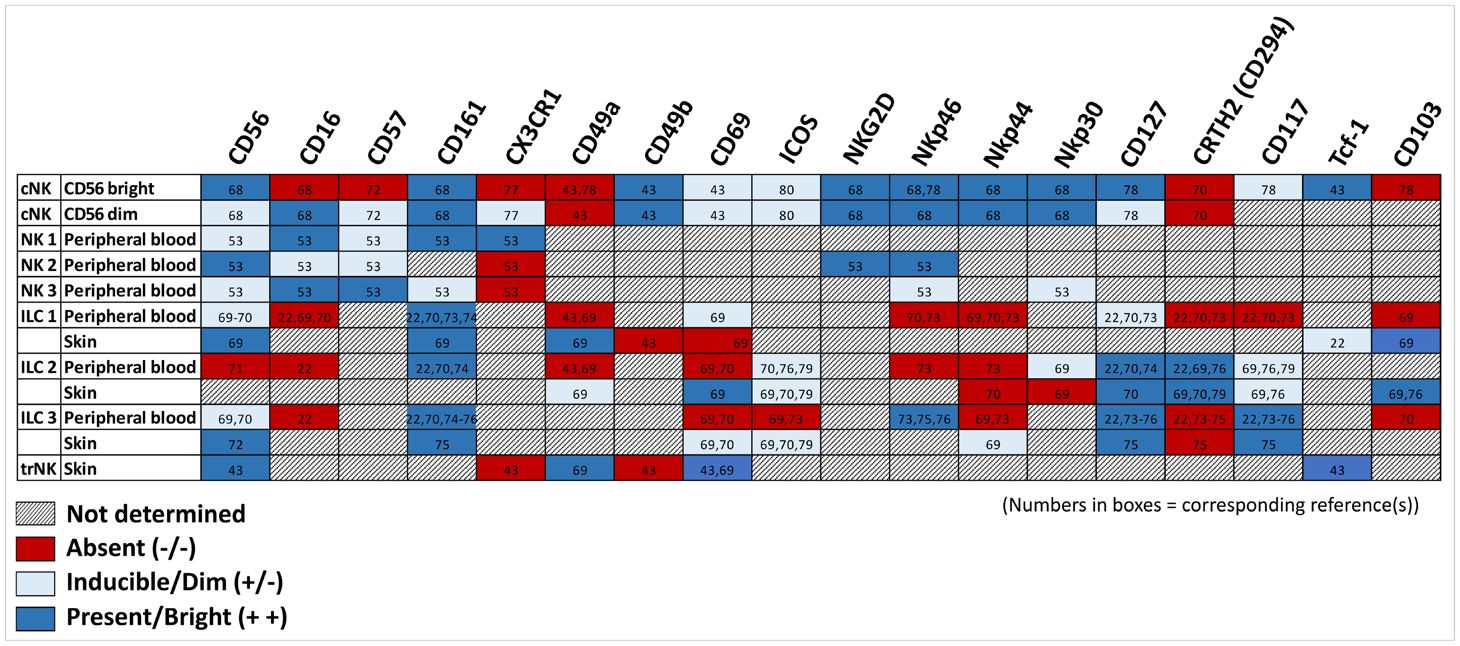
NK cells are typically subdivided into two groups based on their expression of CD56 and CD16. Generally, CD56brightCD16neg NK cells are thought to have an immunoregulatory phenotype (57–60). These cells are commonly found in secondary lymphoid organs and display poor cytolytic and ADCC capacity, but rapidly secrete cytokines and chemokines; these are also highly responsive to cytokine secretion within the tissue microenvironment [reviewed in 61]. In contrast, CD56dimCD16+ NK cells comprise 90% of all NK cells in circulation and are characterized by potent cytolytic function and the capacity to robustly mediate ADCC, but with the tradeoff of a more limited cytokine secretion repertoire (61–63). Though controversial, trNK cells are regarded by some as a subset of group 1 ILCs, which includes ILC1 cells and conventional/peripheral NK cells. In other models, resident NK cells exist on a spectrum with ILC1 cells. Some studies suggest cNK cells can differentiate into ILC1-like cells via IL-12 and TGFβ signaling following suppression of Eomes expression, with a simultaneous increase in T-bet expression (46). This transdifferentiation process appears to provide at least one strategy for tumor immunoevasion, mediated by tumor-secreted TGFβ inducing NK signaling via a non-canonical Smad4 independent pathway (64, 65).
Human skin consists of three distinct layers—the outermost epidermis, the dermis, and the underlying subcutis. The epidermis primarily provides a physical barrier function, while the dermis is traversed by blood vessels and lymphatics, allowing recruitment of both innate and adaptive immune cells that facilitate its participation in cell-mediated immunity (66). Additionally, the dermis contains complex networks of collagen and elastin fibers, collectively forming the extracellular matrix (ECM). This critical scaffold affects both recruitment and migration of immune cells into specific tissues (67). Much of the investigation to date regarding innate immunity in the skin focuses on identifying specific ILC subsets, with more recent studies evaluating tissue resident NK (trNK) cells. Key questions to address include whether trNK cells arise during skin development, or do they migrate from circulation in response to pathogen-specific exposures? Also, what is the role of trNK cells in specific cutaneous pathologies?
Skin trNK cell origin—current understanding
To date, most studies of NK cell populations in the skin have focused on ILC subsets (usually categorized as ILC1, ILC2, and ILC3 cells; see Table 2 (68–80)). Though found in every tissue, ILCs are especially prevalent in barrier tissues, such as the skin, lung, and intestines, where they are thought to be important for maintenance of tissue homeostasis, as well as mediating initial inflammation in response to pathology such as infection (81). A number of insightful review articles have outlined the role and contribution to skin pathology of these ILC subsets (46, 81–83). Notably, a recent review of trNK cells included a discussion of their presence and function in every organ known to house NK cells except the skin (84). Single cell transcriptomic profiling of developing fetal and adult healthy human skin compared with skin from adult patients with atopic dermatitis and psoriasis demonstrated an enrichment of ILCs within the skin during the first trimester for fetal tissue, as well as clonal expansion of disease-associated lymphocytes in diseased skin (85). In mice, NK 1.1+ ILC1 cells are highly enriched in fetal and neonatal skin, but rapidly decline within 6 weeks of life (86). These cells are believed to play a significant role in regulating early cutaneous microbiota colonization. Another study in mice demonstrated a population of invariant natural killer T (iNKT) cells that enter the skin early in the post-natal period, and play a crucial role in skin tissue development and homeostasis through transferrin-mediated regulation of iron metabolism (87). Given nomenclature variability, it is unclear whether these iNKT cells are related to a population of “epidermal lymphoid cells” that express NK cell markers and secrete IFNγ upon stimulation that have been identified seeding the epidermis during late embryogenesis in both wild type and T-cell deficient mice (88).
All three ILC subsets are present in healthy human skin (89, 90); however, they are differentially expressed in disease-specific states (e.g., ILC3s are significantly increased in lesional psoriasis) (91). scRNA-seq of lesional vs non-lesional skin identified four unique clusters of innate lymphocytes in skin that were CD161(KLRB1)+/CD3(CD3D/CD3G)neg. These subgroups included ILC1/3, ILC2, ILC1/NK cells, and NK (KLRD1+, GNLY+, PRF1+, GZMB+, and FCGR3A+) cells (85). In healthy human and murine skin, most effector ILCs are of the ILC2 subset, which produces IL-5 and IL-13 and are increased in cutaneous inflammatory disease (43, 92). These cells have also been implicated in wound healing (93). Some propose that ILC2 and ILC3 cells are predominantly tissue resident in healthy homeostatic states, whereas ILC1 cells continuously traffic between circulation and lymph nodes in a CD62L- and CCR7-dependent manner (94).
Healthy human dermis contains NK cells that are CD56+ and uniformly CD16anegCCR7neg, in contrast to circulating NK cells, which mostly express CD16a and frequently CCR7 as well (95). CD16a is an IgG Fc receptor expressed by NK cells that is essential for inducing antibody-dependent cellular toxicity (3). Upon NK cell activation, this receptor is cleaved by metalloproteinase 17 (ADAM-17) (96–98). ADAM-17, a critical upstream regulator of epidermal growth factor receptor (EGFR) signaling that is vital for human development, is ubiquitously expressed, including by keratinocytes (99). Keratinocyte-specific knock out of ADAM-17 results in mice born with a normal epidermal barrier; however, they quickly (within days of birth) develop compromised epidermal integrity and demonstrate chronic dermatitis as adults (100). ADAM17 inhibition in co-culture experiments prevents CD16a shedding by NK cells and increases their cytolytic function against breast cancer cells (101–103). These studies suggest that upon skin entry, trNK cells derived from circulation lose CD16a expression, likely mediated by ADAM-17 cleavage. It is unclear whether this loss may contribute to the identification of NK cells within the dermis, gut lamina, and cryopatches that demonstrate reduced ability to produce IFNγ or to degranulate (1).
Skin parabiont experiments demonstrate that the majority of trNK cells in the skin are very similar to those found in the liver and uterus, possessing CD49a+DX5neg markers (13). T-bet knock out mice lack both skin and liver trNK cells, suggesting a common precursor cell that is T-bet dependent, whereas uterine trNK cells are still present in T-bet deficient mice (13). Skin trNK cell development is also independent of HOBIT/ZNF683 (14), a transcription factor known for regulating differentiation of human CD34+ progenitor lymphocytic cells into CD56+ NK cells (104). Additionally, this differentiation does not appear to require the TGFβ receptor, which plays a central role in mediating development of skin-localized CD8+ resident memory T-cells (14).
An older proposed classification for tissue-derived NK cell lineages included two circulating populations and two tissue resident populations: 1) cNK cells in spleen, blood, and other organs, 2) thymic cNK cells, 3) trNK cells in liver and skin, and 4) uterine trNK cells (13). These “liver-resident” NK cells (CD49aEomes-NK1.1+KNp46+), however, are now generally classified as ILC1s, reflecting their distinct developmental origin and functional identity (10, 55). Klose et al., 2014 and Robinette et al. were first to delineate ILC subsets across tissues, highlighting the distinction between ILC1s and NK cells, which derive from different progenitors (11, 42). More recently, Nixon et al. described a population of CD49aEomes+ trNK cells in the salivary gland (“SG-ILC1”) that are phenotypically and developmentally distinct from both liver ILC1s and cNK cells (105). Together, these findings support the existence of at least three distinct Group 1 ILC populations: 1) ILC1, trNK, and cNK cells. Interestingly, trNK cells appear to represent a hybrid lineage with features derived from both ILC1 and cNK cell pathways. Torcellan et al. also recently described a group of skin-resident NK cells that acquire both long-term residency and memory properties in response to but then sustained following resolution of murine skin CMV infection. These were notably distinct from the previously identified ILC1, salivary trNK and cNK cell populations (14). In addition to providing a model by which circulating NK cells may establish tissue residency in the skin, these and prior findings importantly highlight tissue-specific diversity within the Group 1 ILC compartment. Overall, differentiating when and how circulating NK cell populations establish residency versus transiently infiltrate tissue(s) in response to infectious stimuli, remains a topic of ongoing investigation (106).
NK cells identified within skin and other peripheral tissues (unlike those in circulation) lack direct cytotoxic function (107). This absence may be altered/blocked upon exposure to ECM proteins within the dermis, as previously discussed. Further evidence for this alteration was demonstrated through a murine cytomegalovirus (mCMV) model (m157), which acts as an NK cell-specific activating ligand. Bunting et al. demonstrated that mCMV skin grafted onto the back of C57BL/6 WT mice recruited circulating cNK cells, giving rise to trNK cells in donor skin. Both CD49b+ cNK and CD49a+ trNK cells found in the m157-expressing skin grafts were recipient derived (107). Entry into the skin resulted in a drastic change of NK cell function, including down-regulation of their cytotoxic program, while simultaneously boosting chemokine and inflammatory cytokine production (107). Interestingly, trNK cells isolated from donor skin that were then tested in vitro regained their cytotoxic ability to fully degranulate outside of the skin. In an elegant parabiosis experiment, this group further demonstrated that NKp46negGFP+ NK cells injected intravenously into WT mice one day following skin transplantation were found in the recipient liver as well as in the skin graft twenty days later. These findings suggest the NK cells that set up tissue residency in skin grafts are recruited from circulation, then adopt skin tissue resident properties after migration to the dermis/epidermis (107). It also further supports an as yet undefined relationship between skin and liver trNK cells (108).
NK cell migration to the skin
As mentioned above, innate immune cells are among the most common lymphocytes found within skin in the early post-natal period, consisting predominantly of innate T-cell subtypes (109–111). Importantly, skin colonization with normal bacterial flora during early neonatal development invites an influx of activated regulatory T (Treg) cells into the skin that are critical for establishing immune tolerance to commensal organisms (111). Transcriptomic profiling of immune cells in mouse skin at different post-natal time points demonstrates that iNKT cells are especially dominant and play critical roles in skin development and homeostasis during this early period (87). These innate-like αβ T cells present in infant and young mice are gradually replaced in the early post-natal period by more adaptive immune populations in a microbiota-driven process (87).
T-cell lymphocyte trafficking and entry into the skin is well-characterized (112); however, data regarding NK cells in this arena is limited at best. In general, leukocytes homing to the skin adhere to the endothelium of dermal vasculature via β2 integrins such as LFA-1 (CD11a/CD18, αLβ2) or Mac-1 (CD11b/CD18, αMβ2) binding to ICAM-1 (CD54) (113, 114) or via β1 integrins binding to VCAM-1. Proinflammatory cytokines, such as IFNγ, TNFα, and IL-1, trigger ICAM-1 and VCAM-1 activation to facilitate this T-cell localization to sites of skin inflammation (114–117). The complex structural fortification of the ECM makes further lymphocyte localization to the epidermis difficult (107). Though many chemokines are involved, the CCL17-CCR-4 and CCL27-CCR10 axes appear to be the most influential in recruitment of T cells to active sites of skin inflammation, such as in psoriasis and atopic dermatitis (AD) [reviewed by 112]. Additionally, CCR6 is a key chemokine for directing skin homing of regulatory T cell (Tregs) in psoriasis pathogenesis (112). In contrast, trNK cells in the skin express a variety of chemokine receptors, including high levels of CXCR3 and CCR5, and infiltrate the skin in response to disease-specific chemokines, such as CLL5 in psoriasis (118). Though the phenotypic tissue and memory profiles for recruited T and NK cells generally mirror each other, it is important to characterize their cell-specific recruitment to and maturation within skin tissue, since they maintain distinct programs of differentiation (61).
While CCR10 expression is especially prevalent on skin memory-like resident T cells (119), the majority of ILCs in human skin also express CCR10 (43), which plays a role in recruiting activated lymphocytes from skin-draining lymph nodes to help regulate the homeostasis of T helper (Th) cells and Tregs (120). Irradiated CCR10-/- mice demonstrated lower skin ILC percentages and increased cutaneous damage compared with CCR10+/- controls (121). Specifically, ILC cells found in skin draining lymph nodes of CCR10 knock out mice demonstrate defective migration to the skin and instead divert to other sites lacking expression of CCR10 ligands, such as the lungs (120). CCL27, the ligand for CCR10, is highly and specifically expressed on epidermal basal keratinocytes in both healthy mouse and human skin, and is upregulated during inflammation (122). This ligand is also critical for the establishment of resident lymphocytes in skin and mucosal tissues, independent of stimulation by local commensal microbial populations (123). Another critical receptor in this homing process is cutaneous lymphocyte associated antigen (CLA). CLA is a glycoprotein expressed on the surface of various lymphocyte populations that serves as a homing receptor for skin-infiltrating NK cells (124, 125). It binds to E-selectin, which is expressed in endothelial cells and other tissues during acute inflammation (126). Intra-dermal injection of recombinant human CCL27 immediately recruits CLA+ homing lymphocytes from circulation (122). Additionally, dermal microvascular endothelial cells, fibroblasts, and circulating CLA+ T cells demonstrate surface expression of CCR10, all of which suggests a critical role for the CCR10-CCL27 axis in recruitment and migration of lymphocyte populations directly to skin (122). While skin infiltration also appears to be CCR10 dependent (120), CCR8 likely contributes to initial skin tissue homing. Skin resident NK cells specifically express CCR8 (95, 118), and CCR8 expression on specific ILC cells (discussed below in greater detail) is critical for skin homing (127). Though its ligand CCL1 is not expressed by keratinocytes or dermal fibroblasts, low levels of CCL1 on microvascular endothelial cells may initially recruit circulating NK cells (128). CCR8 is also critical for regulating the migration of inflammatory cutaneous dendritic cells from skin to draining lymph nodes in contact hypersensitivity reactions (129). These then trigger skin homing of CCR10+ ILCs from circulation to skin-draining lymph nodes (120). Further work is needed to delineate the role of CCL27-CCR10 axis in mediating adhesion and initiating transepidermal migration of cNK cells into skin.
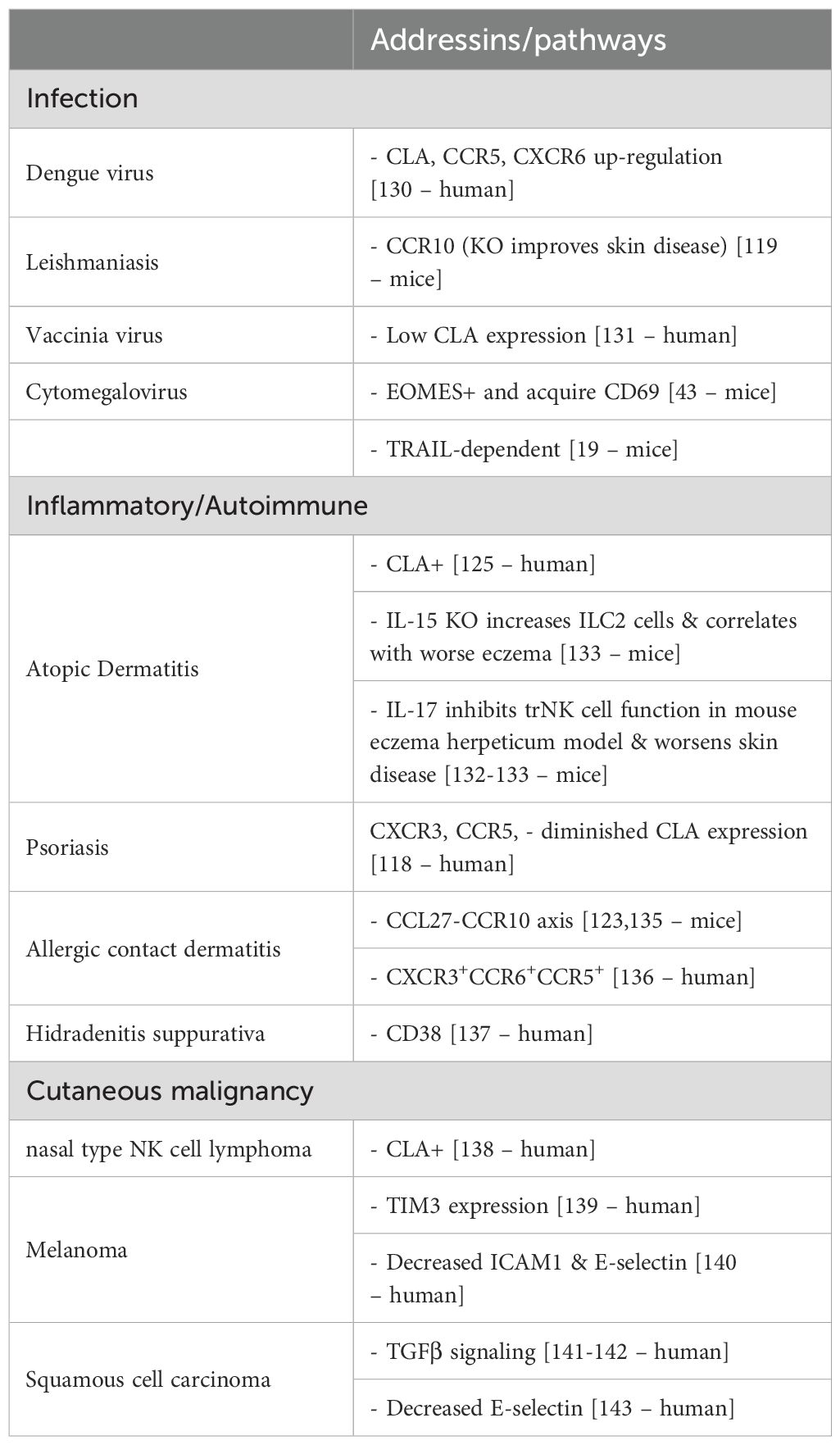
As most of current understanding of NK cell recruitment and migration to and effector function within skin has been garnered from models of cutaneous tissue perturbed by various pathologies, further discussion of this subject will be described through highlighting prior research findings in three general areas of human and murine disease: infection, inflammatory/autoimmune disease, and cutaneous malignancy (Table 3) (130–143).
NK cells in lesional skin
Infection
As just discussed, leukocytes found in the skin possess a repertoire of homing receptors, including CLA and a number of chemokine receptors (CCR4, CCR6, and CCR10), that facilitate trafficking to sites of inflammation (128). Many ILC2 cells derived from healthy donors also express baseline levels of CLA, CCR10, and CCR4 (43). Chemokine receptor CCR8 facilitates ILC2 migration from the bone marrow to the skin upon trauma to skin tissue (82). During acute infection, CLA expression on circulating vs trNK cells appears to be variably expressed, suggesting a crucial role for CLA in homing to and infiltration of skin in response to specific pathogens. In cutaneous leishmaniasis, for example, CD8+ T cells expressed higher levels of CLA within the skin, while circulating NK cells demonstrated reduced CLA expression, compared to those of age-matched controls (131). In contrast, infection with Dengue virus results in increased expression of CLA and other homing receptor (CCR5 and CXCR6) by circulating NK cells (130). It is unclear whether NK-cell mediated response to certain pathogens within the skin plays a role in the resultant tissue damage. For example, accumulation of circulating NK cells with higher cytotoxic capacity (CD56dim/CD57bright) positively correlated with skin lesion size in patients with cutaneous leishmaniasis (131), although the tissue pathology was thought to be more CD8+ T cell driven. Furthermore, CCR10 knock-out mice demonstrated robust and protracted innate immune responses in the skin that phenotypically manifested as increased clearance of cutaneous Leishmaniasis (119).
Are these NK cells recruited to the skin in response to particular infectious agents, already present based on prior cutaneous microbe sensitization, or some combination of the two? Although relatively unexplored in skin, recent works have revealed that tissue resident ILC cells can be mobilized in response to specific inflammatory cues and migrate to other organs (94, 144–146). A study in which CD45.1+ and CD45.2+ parabiotic mice were surgically connected for 30–40 days to achieve complete blood chimerism demonstrated that >95% of ILC cells (regardless of subset) within these tissues were of host origin (147). Notably, the one exception was ILC1 cells (Eomes-Nk1.1+) in lung and peripheral blood, which originated from both parabionts. Again, though, none of these experiments evaluated the skin.
A few studies have, however, looked at NK cell homing in response to local cutaneous infection. In murine models, following intradermal infection with vaccinia virus or Staphylococcus aureus, NK cells were immediately recruited to the site of skin infection and retained for up to 80 days post-infection (14). While cNK cells are phenotypically Eomes+/CD49a-, a number of recent studies in various tissues (uterus, salivary glands, etc) have demonstrated establishment of a Eomes+CD49a+ tissue-resident NK cell population (65, 148, 149). These are phenotypically and ontogenically distinct from resident ILC1 Eomes-CD49a+ cells within these tissues (149, 150). Similarly, Torcellan et al. demonstrated in skin that cNK cells are recruited to sites of cutaneous infection and subsequently undergo transcriptional reprogramming that render their expression profile similar to that of trNK cells (Eomes+CD49a+), with some evidence of memory-like features (CD69+) (14). As expected, when challenged near the previous site of infection with the same pathogen, a more robust effector response was mounted than that observed during the initial infection (14). Similarly, a population of trNK cells was recruited and expanded within murine salivary glands following CMV infection (151). Unexpectedly, these trNK cells were shown to eliminate anti-viral CD4+ cells in a TRAIL-dependent fashion (38), ultimately preventing the development of Sjogren’s syndrome, as compared with the control group. This observation suggests a more immunoregulatory role for mucocutaneous trNK cells in this context. Notably, Schuster et al. demonstrated that the trNK cell population that developed in these salivary glands in response to CMV infection was distinct from the populations present in infection-naïve mice (151). Given the complex relationship of human skin to its innumerable commensal and pathogenic microbes, it is unsurprising that immune cells responsible for bridging innate and adaptive immunity would be highly responsive and adaptable on a pathogen-specific basis. Construction of this highly variable adaptive programming likely begins during pre- and early post-natal development (87).
Finally, the critical role of NK cells in combating infections with cutaneous manifestations is abundantly evident in patients with low NK cell levels and/or NK cell deficiencies. NK cells especially play a role in eradication of the herpesvirus family, including herpes simplex virus (HSV), varicella zoster virus (VZV), Cytomegalovirus (CMV), Epstein-Barr virus (EBV), and human papilloma virus (HPV) (152–159). Patients with DOCK8 immunodeficiency syndrome are particularly susceptible to HPV infections, a common cause of cutaneous warts and increased HPV-driven SCC (160). EBV infections with mucosal and skin findings are increased in patients with Chediak Higashi syndrome (161). In addition to skin, teeth and hair abnormalities, patients with NEMO (NFκB) deficiency syndrome, who have both low NK cell and antibody function, are unable to mount a sufficient response to numerous bacterial and viral infections (162). Herpesvirus infections are also common in other immunodeficiency syndromes with low NK cell levels, including Wiskott-Aldrich (163), familial hemophagocytic lymphohistiocytosis (164–166), and Hermansky-Pudlack syndrome (167, 168). NK cell deficiency in humans is also associated with an increase in disseminated HSV and VZV infections (153, 169, 170). VZV infection in particular triggers CCR4 and CLA activation in infected NK cells, initiating their migration directly to the skin (154). Viruses like human immunodeficiency virus (HIV) and Kaposi’s sarcoma-associated herpesvirus down-regulate MHC class I molecules on the surface of infected cells to evade cytotoxic T cells, making them more susceptible to NK cell-mediated responses (171), but unfortunately NK cell activity in HIV infection is often defective (172, 173). The innate anti-viral function of NK cells presents a promising therapeutic avenue for exploitation of direct cellular therapy in cutaneous infectious disease.
Inflammatory/autoimmune
Skin ILCs are felt to significantly contribute to the maintenance of tissue homeostasis, and their imbalance is implicated in a number of autoimmune and inflammatory diseases, such as atopic dermatitis (AD) and psoriasis (174). As previously mentioned, each of the ILC subgroups within the skin (as in other tissues) appear to mirror their T helper cell counterparts (175). For example, ILC1 cells are considered the innate counterparts of CD4+ Th1 cells, ILC2 cells have a role in atopic dermatitis (synonymous with eczema) akin to the Th2 subset, and ILC3 cells, which produce IL-17, may be relevant for psoriasis (91, 92, 176, 177). ILC2 is normally expressed in the dermis and subcutis, whereas ILC3/LTi-related genes are found primarily in the epidermis (178–180). ILC2 and ILC3 cells are mostly considered tissue resident, though as previously discussed, can be induced to migrate under inflammatory conditions. ILC1 cells traffic back and forth between circulation and the lymph nodes, dependent on CD62L and CCR7 (94) (Figure 1).
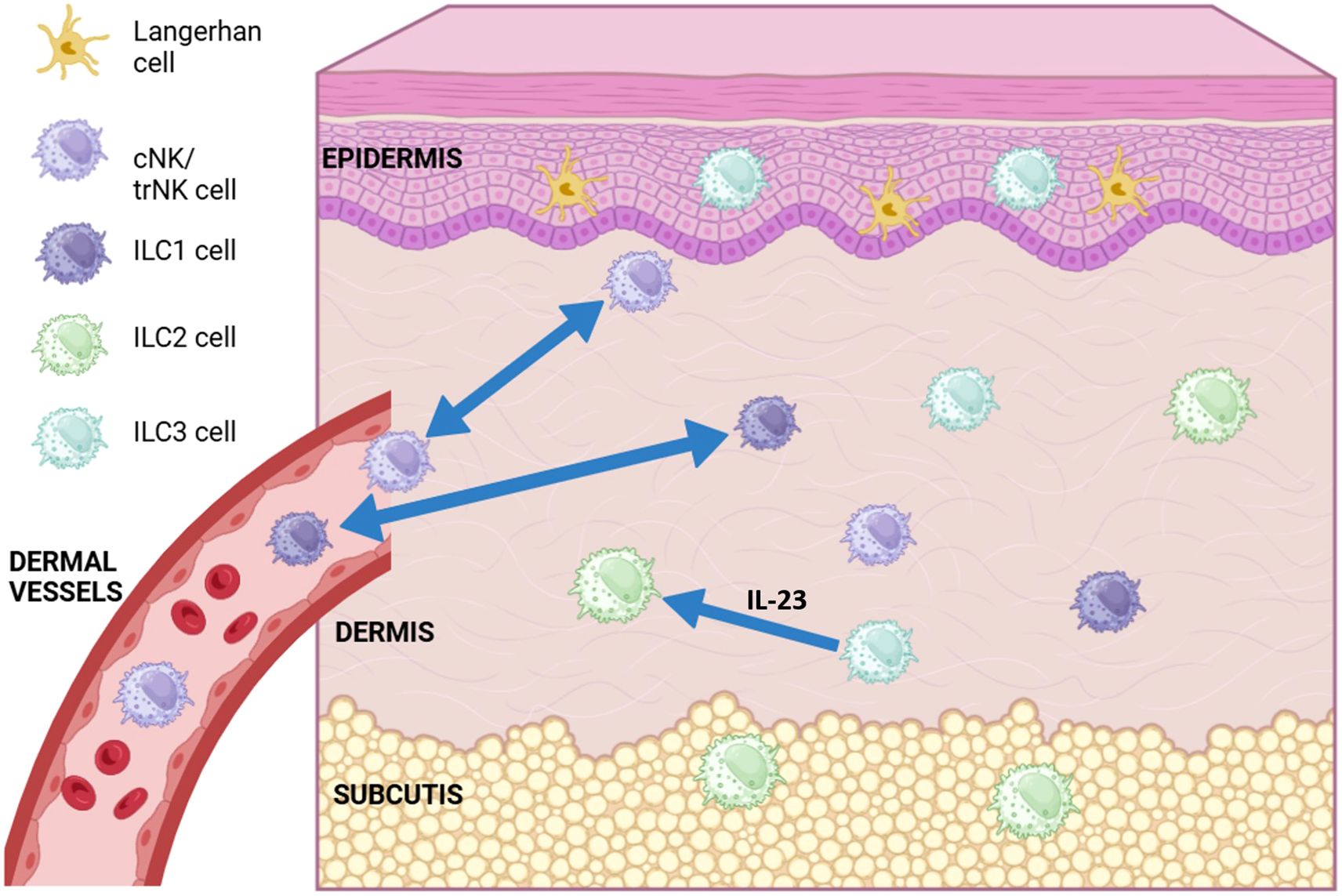
Figure 1. Diagram of conventional (cNK), tissue resident (trNK) NK cell and ILC (ILC1, ILC2, ILC3) subsets found in different layers of the skin. ILC1 and cNK cells have been shown to migrate back and forth between the dermis and dermal vasculature (blue double-headed arrows). ILC3 cells have been show to transdifferentiate into ILC2-like cells in the presence of IL-23 (blue arrow). Langerhans cells are the most abundant immune cell type found in the skin, most commonly in the epidermis. (Figure created using Biorender.com).
Interestingly, the basal level and subtype of ILCs within skin may be directly influenced by environmental exposures, as discussed above with cutaneous infection-triggered immune responses. For example, ILC2 cells produce type 2 cytokines (IL-4, IL-5, IL-9, IL-13) (181–184) and are overexpressed in both mouse and human atopic lesional skin (43). Moreover, evidence exists of additional ILC2 recruitment to these inflammatory sites, mediated by skin-specific expression of IL-33 (185). Notch signaling may play a significant role in this ILC2 plasticity in its conversion from a resting to pro-inflammatory state (186). Blocking Notch signaling via γ-secretase (a pan-Notch inhibitor) in a mouse model treated with recombinant IL-25 to induce inflammatory ILC2 populations abrogated their cytokine responsiveness and ability to transdifferentiate into an ILC3-like state (186). Addition of Notch ligands to isolated resting ILC2 cells in vitro rescued this phenotype of maintaining ILC2 expression of GATA3 while acquiring an ILC3-like ability to secrete IL-17 in its pro-inflammatory state. Atopic lesional skin specifically demonstrates an influx of granzyme positive NK cells in the dermis, with homing receptor CLA+ expression being especially prevalent in patients with severe disease (125). It is unclear, however, whether cNK cells recruited to the skin contribute to inflammatory pathogenesis, or instead help limit type 2 inflammatory responses mediated by local/previously established tissue resident ILC populations. IL-15 knock-out mice, which are NK cell deficient and clinically manifest a chronic eczematous dermatitis phenotype, demonstrate an increase in ILC2 cells and eosinophils within both lesional skin and skin-draining lymph nodes (133). Further, peripheral blood and lesional skin of patients with AD have a global reduction in NK cells, but also demonstrate decreased total CD56dim mature effector NK cells, which recovered following patient treatment with Dupilumab (IL-4 and IL-13 inhibitor) (133). This suggests a distinct role for cNK cell recruitment and skin infiltration in attempting to limit Th2 inflammatory response within disease-specific contexts.
Eczema herpeticum (EH) is one of the gravest and most feared complications in patients with atopic dermatitis. Eczematous mice intradermally injected with HSV1 virus rapidly develop EH; lesional skin from these mice was found to have reduced NK cell activity compared to non-eczematous mice injected with the virus (134). Adoptive transfer of healthy NK cells into these eczematous mice the day prior to viral infection, however, decreased their EH scores. Interestingly, skin lesions of eczematous mice infected with the vaccinia virus have increased numbers of NK cells compared to non-eczematous mice infected with the same virus; however, NK cell cytotoxic activity (measured by expression of granzyme B, perforin, and IFNγ) is significantly reduced in the eczematous mice (134). Adoptive transfer of healthy NK cells via intravenous injection into eczematous mice prevented development of skin lesions following vaccinia infection, and antibody-mediated IL-17A neutralization in eczematous mice delayed skin lesion onset after virus infection (134). Furthermore, this reduction in development of skin lesions was abrogated by αAGM1 antibody-mediated depletion of NK cells prior to IL-17A neutralization, suggesting IL-17A directly affects NK cell activity in virally-infected eczematous mice.
Parabiotic mouse experiments have shown that ILCs in the gut, lung, and skin are maintained and expanded locally in physiologic and pathogenic conditions with only minor contributions from circulating progenitors (147). What then, is the degree of plasticity between the various trNK and ILC subsets? Although ILC2 cells are the predominant ILC subtype found within healthy dermis, ILC3 cell populations are predominant in this layer in human and mouse psoriatic skin (92). Psoriatic plaques on human skin have also demonstrated an infiltrating immune cell population comprised of CD3-CD56+CD16-CD158b- NK cells, all of which are localized to the mid and papillary dermis (118). These skin-infiltrating NK cells express high levels of CXCR3 and CCR5 chemokine receptors, release abundant IFNγ following stimulation, and fail to express the skin-homing CLA antigen (118). Absence of the chemokine CCL27 in a knock-out mouse model results in an overactive cutaneous inflammatory response in the well-established model of imiquimod-induced psoriasis (123).
While much of the current literature regarding NK cells within inflammatory cutaneous conditions focuses on psoriasis and atopic dermatitis, where their numbers are greatly reduced and/or skewed to one specific ILC phenotype, there have also been documented in studies of immune subpopulations in several other diseases, where NK cells seem to play a more inflammatory role. Lichen planus (LP), whose damage is hallmarked by lymphocyte-induced vacuolization of basal keratinocytes at the junction between the epidermis and dermis, demonstrates a high number of CD3-CD56highCD16- NK cells in early LP lesions (136). Skin NK cells isolated from these lesions were negative for inhibitory receptors (KIR receptors CD158a and b), but highly positive for perforin and the activating receptors NKG2D and NKp44; additionally, these cells possessed the ability to secrete IFNγ (136).
Another common inflammatory cutaneous disease is allergic contact dermatitis (ACD). While Rag -/- mice (lacking lymphocytes) are unable to mount a contact hypersensitivity response, adoptive transfer of NK cells from sensitized donors restores this response, independent of B or T cells (135). NK cells accumulate abundantly in lesional skin of patients with ACD and secrete IFNγ in the presence of hapten-driven T cells in vitro (136). Interestingly, ILC1 cells sensitized in the skin draining lymph nodes in a mouse ACD model were then recruited to the liver, where they were retained and could be reactivated upon hapten re-exposure (20). Direct skin hapten challenge after this pre-requisite sensitization then mediated early recruitment (within the first 24 hours) of NK cells to the skin and skin draining lymph nodes (187). ILC1, ILC2, and ILC3 cell numbers then increased after this initial response, with subsequent decreases in cNK cells within the skin. Localization of this hapten-memory specific NK cell subpopulation to the liver suggests that some NK cells possess memory-like properties generally thought to be more associated with adaptive immune cell populations (135). In contrast to T-cell mediated hypersensitivity responses, however, NK cell induced inflammation is more transient and less inflammatory than that mediated by CD8+ T-cells (188).
Specific autoimmune inflammatory skin conditions associated with reduced numbers of NK cells include hidradenitis suppurativa (HS), lupus, and systemic sclerosis (189–191). Expression of CD38, a glycoprotein found on the surface of NK, B, and T cells that is an important mediator of inflammation, is often dysregulated in HS (137). ILC1 and 3 subsets are increased and ILC2 decreased in peripheral blood of patients with systemic lupus erythematosus (SLE) and in patients with HS compared to normal controls (192–194). While these studies do not directly examine the role of skin-infiltrating NK cells within these cutaneous disease states, cNK cell dysregulation within SLE is well-established. Not only are NK cell populations decreased in peripheral blood of patients with SLE compared to controls (195), but co-culture of serum of patients with SLE with NK cells from healthy controls actually inhibits NK cell activity (196). Interestingly, the degree of NK cell functional inhibition directly correlates with SLE disease activity (196). Better characterization of cNK cell recruitment and contribution to inflammatory and autoimmune skin diseases may further our understanding of disease-specific pathogenesis.
Cutaneous malignancy
NK cells are programmed to immediately and aggressively respond to viruses and tumor cells, so better characterization of the role of NK cells within cutaneous malignancy provides a promising potential area for therapeutic intervention. In general, impaired NK cell function correlates with high tumor stage and worse prognosis in a number of solid tumors [reviewed by 57]. For example, NK cell tumor infiltration is an independent predictor of progression free survival in GI stromal tumors and pulmonary adenocarcinoma (197, 198). Most of the work investigating the role of NK cells in cutaneous malignancy to date has understandably focused on melanoma, the most deadly skin cancer. Patients with melanoma have an enriched population of CD56brightCD16dim NK cells (199), which correlates with reduced overall and progression free survival (200). Some studies, however, have shown enrichment of CD56dimKIR+CD57+ highly cytotoxic NK cells in tumor-infiltrated lymph nodes in melanoma patients (201, 202), suggesting that arrest of NK cell cytotoxic efficacy likely occurs after skin tumor infiltration. It appears that trNK cells within the skin inherently lack direct tumor killing properties without cytokine-mediated licensing. Further support for this hypothesis comes from the finding that peripheral blood-isolated NK cells display an innate ability to kill both murine and human melanoma cells in vitro (203, 204), yet NK cells isolated directly from healthy human skin require culture in the presence of IL-2 to induce cytolytic activity against melanoma cells (95). It was also recently demonstrated that IL-2-expanded NK cells suppress cSCC cell survival and tumor growth both in 3D cell culture models as well as xenograft cSCC tumors in NSG mice (205). Treatment with these expanded NK cells resulted in a dose-dependent reduction in spheroid growth and Matrigel invasion assays, with an associated increase in apoptosis signaling pathways (205). Additionally, expression of leukocyte homing receptors and subsequent adhesion receptors (especially E-selectin and ICAM1) is reduced and/or dysregulated in malignant melanoma (140) and cutaneous squamous cell carcinoma (cSCC) (143). Interestingly, RNAseq data gene set scoring of tumors from patients with metastatic melanoma demonstrated that improved survival/prognosis was associated with increased NK cell tumor infiltration (206).
In contrast, cutaneous NK cell lymphomas appear to pose the opposite threat: too many activated skin-infiltrating NK cells. A number of case series and smaller studies on skin tissue from patients with this rare cutaneous lymphoma have shed an intriguing light on NK cell homing to and effector function within the skin. In particular, two cases of aggressive non-nasal extranodal NK cell lymphomas associated with EBV infection demonstrated early dissemination to the skin, liver, spleen, and BM (207). These tumors were CD56+CD16+, and expressed CLA, granzyme B, and TIA-1. In a study of 52 cases of NK-cell lymphoma, CLA expression was highly expressed in cases with a cutaneous component, compared to their non-cutaneous counterparts (138). The CLA+ group also had a significantly worse prognosis compared to the CLA- group, regardless of the tumor’s primary site or clinical staging (138). High CLA expression levels are also seen in NK-like T-cell lymphoma (208). Given its role in recruitment of circulating lymphocytes during active inflammation (including NK and T cells), these findings likely reflect a CLA mediated increase in NK cell infiltration and retention within cutaneous tumor tissue. Better characterization of this homing mechanism may provide contextual insights into when and why NK cells are recruited to and retained within inflammatory vs malignant skin tissue, and how their phenotype and function are affected upon arrival.
While great success has been achieved with the use of NK cell adjunct therapy in hematologic malignancies (209–219), it is well established that NK cells are rapidly rendered dysfunctional upon entry to the tumor microenvironment (TME) in several solid tumor malignancies (220). Melanoma-associated fibroblasts reduce NK cell cytotoxic capacity and function (221, 222), so it is unsurprising that a small clinical trial testing adoptive transfer of NK cells infused into patients with progressive stage IV melanoma saw no clinical response (223). A number of studies have demonstrated a role for immunotherapy in restoring NK cell function within the TME, with the degree of responsiveness to immunotherapy directly associated with baseline cNK cell presence before and during treatment. Survival in a cohort of patients with malignant melanoma treated with anti-CTLA-4 correlated with both low expression of the inhibitory receptor TIM-3 on circulating T and NK cells during and prior to treatment, as well as increased frequency of mature NK cells (CD3-CD56dimCD16+) during therapy (224). Treatment of patients with melanoma with ipilimumab induced a significant increase in CD56dim, but not CD56bright NK cells in the periphery (225). Importantly, there appears to be a role for circulating NK cells in limiting pulmonary seeding of melanoma metastasis and in recruitment of T cells directly to metastatic foci (226). Ongoing efforts include a Phase I/II clinical trial infusing ex-vivo expanded allogeneic universal donor NK cells in combination with Temozolomide as a lymphodepleting agent in patients with melanoma and brain metastasis (NCT 05588453).
Most NK cell research in cSCC has been performed on head and neck SCC (HNSCC), which is commonly HPV-driven (227). Cetuximab inhibits EGFR pathway signaling and activates innate immunity, partially via stimulation of NK cells in HNSCC (228). Co-treatment using PD1 blockade with stimulated NK cells (via monoclonal antibody-mediated blocking on the NK inhibitor receptor NKG2A) in a phase II trial treating head and neck SCC has shown encouraging results (229). Additionally, the PD-1 blocking antibody cemiplimab, which enhances NK cell intra-tumoral activity, has demonstrated promising treatment responses in metastatic cSCC (230–232).
Significantly, all of the studies discussed above regarding melanoma and cSCC have focused on NK cell levels circulating in the blood or in tumor-infiltrated lymph nodes. While little has been done to investigate the role of trNK cells directly within the TME of these cutaneous malignancies, a few studies demonstrate the positive effect of functional NK cells within skin tumors, as previously discussed (206, 233). Mechanistically, NK cell production of Flt3lg within the TME of melanoma tumors directly affects the level of intra-tumoral stimulatory dendritic cells (antigen presenting cells that are important for infection and tumor defense within skin). This NK cell infiltration not only correlated with increased overall survival in patients with melanoma, but also predicted positive responsiveness to anti-PDL1 immunotherapy (233). As mentioned previously, NK cells typically circulate in peripheral blood, but can be recruited via stress signals from pathogenic cells, such as directly to tissue (i.e., skin) (68). Though infiltrated with an abundance of NK cells, human cSCC-associated NK cells have impaired cytolytic function and IFN-γ production, which can partially be restored upon in vitro stimulation (234).
So, why are cNK cells homing to the skin for anti-tumoral response losing their cytolytic capacity? In addition to the prior discussion regarding a potential contribution of CD16 shedding in this process, a number of studies demonstrate a critical role for TGFβ signaling in direct suppression of NK cell activity and anti-tumor function within the TME. TGFβ1, a cytokine critical for wound healing in the skin (235), inhibits NK cell-mediated killing of dendritic cells through selective inhibition of NKp30 expression on NK cells (236). There is also evidence for a role of TGFβ signaling in mediating conversion from cNK cells (CD49a-CD49b+Eomes+) into a tissue-resident ILC1-like cell population in salivary glands and intestinal mucosa (65, 237). Suppression of this TGFβ signaling in NK cells enhances their ability to limit metastases in multiple tumor models in mice (including melanoma) through disinhibition of the mTOR pathway (238). In contrast, absence of SMAD4 in ILC1 and NK cells in mice caused NK cells to acquire an ILC1-like gene signature; these cells were unable to control tumor metastasis or viral infection (237). These findings suggest a role for canonical SMAD4 in restraining non-canonical TGFβ signaling via cytokine receptor TGFβR1 in NK cells (237).
We previously described a method for propagating large numbers of clinical-grade NK cells in vivo with IL-2 and irradiated K562 feeder cells expressing membrane-bound IL-21 and 4-1BBL (239, 240). More recently, we reported a modification of that method that enhances NK cell function and overcomes TGFβ-induced suppression (referred to as TGFβ “imprinting” by serially stimulating the NK cells with TGFβ during the expansion process (241). The addition of TGFβ during NK cell propagation impairs neither fold expansion nor viability of the final expanded NK cell product, but the resulting TGFβ imprinted (TGFβi) NK cells exhibit high cytotoxicity and a pro-inflammatory hypersecretion of IFN-γ and TNFα in response to tumor targets (241). Moreover, these cells significantly downregulate SMAD3 at the transcriptional level, resulting in resistance to suppression by TGFβ. Importantly, this cytokine hypersecretion persists for one month after removal of TGFβ (241), suggesting that TGFβi NK cells may retain their enhanced cytokine secretion in vivo, where IFNγ and TNFα secretion can then stimulate adaptive immunity and sensitize tumors to NK cell killing (27). Given the previously discussed evidence for TGFβ imprinting in mice to an ILC1-like phenotype (65, 237), we postulate that the unique characteristics of this modified NK cell product would enable these ex-vivo imprinted cells to persist following direct injection into skin tumors with an ILC1-like or skin tissue resident phenotype. Their acquired resistance to TGFβ and enhanced cytotoxic properties would then enable these cells to overcome the inherently suppressive TME of the cutaneous malignancy, and also mediate robust anti-tumoral activity. To this end, further pre-clinical and exploratory clinical studies are currently underway.
Discussion
Missing pieces/next steps/harnessing NK cells
One driving challenge to cohesion within the NK cell literature is the complex heterogeneity in defining NK cells dependent on the specific tissue and disease of interest. It appears universally accepted that NK cells are broadly defined as CD56+CD3- (differentiating them from T lymphocytic cell populations). However, until recently, there has been no standardized panel for describing NK cells. Some groups have previously described them in terms of being CD56brightCD16- vs CD56dimCD16+, having lytic or IFNγ secretory functions, circulating vs being tissue resident, or even based on their secretion of and response to specific cytokines (242, 243). For example, NK-22 cells were identified as a subpopulation found in mucosa-associated lymphoid tissues (i.e., Peyer’s patches and tonsils) that secrete IL-22 in response to IL-23 (32). Activated NK-22 cells in vitro stimulated epithelial cells to secrete IL-10 and to proliferate (32). Interestingly, a skin homing population of memory T-cells (CLA+CCR10+CCR6+CCR4+), referred to as “Th22,” comprise the majority of T lymphocytes isolated from psoriatic skin lesions (244). Given their known pathogenic role of secreting IL-22 in response to IL-23, NK-22 cells are likely also implicated in psoriasis (245, 246), though this has yet to be confirmed. Do these NK-22 cells truly represent a distinct subpopulation of NK cells? Or is this evidence that NK cells are marked by a degree of plasticity that enables immense adaptability in a pathogen-driven and microenvironment-specific context?
As previously discussed, NK cells circulating in peripheral blood were most recently characterized into three distinct subsets: NK1, NK2, and NK3 cells, respectively, that are each characterized by a unique portfolio of protein and transcriptional programing (53). The range of plasticity of ILC subsets and trNK cell populations within skin is still unclear: do NK cells entering tissues “transform” into ILC1-like cells, or do these ILC and trNK subsets represent distinct immune cell entities? Park et al. demonstrated NK-to-ILC1-like transdifferentiation in response to Toxoplasma gondii infection, and this population expanded within both affected tissues as well peripheral circulation (247). Conversely, in the skin, there is evidence for adaptation of circulating Eomes+ NK cells into CD69+Eomes+ resident NK cells, without loss of Eomes or conversion into ILC1s (14). These findings suggest that tissue-resident NK cells in the skin are a distinct population arising from circulating cNK cells, which at times may take on a ILC1-like phenotype, but are ontogenically distinct from ILC1 cells.
Additional questions to be answered within the skin tissue NK cell literature include understanding the two-faced role for NK cells in mediating host immune response, while also preventing host T cells from excessive activation and causing autoimmune damage to the specific tissues it should be protecting from pathogens. Many of the studies investigating cutaneous inflammatory and autoimmune processes have focused on T cell populations/subsets, with more recent data identifying ILC subsets within the skin that “mirror” their T helper cell counterparts, including in response to specific transcription factors (i.e., Th2 and ILC2 predominance in lesional skin of human and murine atopic dermatitis) (79, 92, 122, 244, 248). Given the critical role for NK cells as a bridge between innate and adaptive immunity, there is a need to further elucidate the role for disequilibrium in NK cell recruitment and function in the skin as it pertains to the pathogenesis of cutaneous autoimmune and inflammatory processes.
From a clinical or translational standpoint, what further implications do these findings carry? NK cells play a negligible role in graft rejection in skin and other organs and tend not to mediate graft-vs.-host disease (GvHD), making them an ideal effector tool for cellular therapy (249, 250). Additionally, NK cells are not thought to be involved in GvHD in stem cell transplants, with the exception of a single paper reporting significant GvHD following NK cell infusions (251). Understanding how trNK cells are recruited, activated, and regulated within different contexts and how they communicate with microorganisms and other tissue-resident immune cells, including surrounding stroma (epithelial cells, fibroblasts, neurons), may provide insights that would enable development of unique therapeutic strategies. For example, a recently published study describes a unique strategy to co-op innate immunity as a topical therapeutic approach in psoriasis to modulate pathologic inflammatory skin ILCs (252). Additionally, Mack et al. observed NK cell deficiency with an associated increase in type 2 cutaneous inflammation in patients with atopic dermatitis [Mack 2020]. The AD-like phenotype in their mouse model was significantly improved via treatment with an IL-15 superagonist, which promotes NK cell proliferation and survival. Direct NK cellular therapy also presents exciting opportunities for intervention within cutaneous malignancy, including accompaniment of a markedly improved side effect profile compared with that currently experienced with immune checkpoint inhibitor immunotherapies (253). Overall, these and other emerging strategies to enhance NK cell function within various skin pathologies offer promising therapeutic potential for a wide range of cutaneous diseases.
Conclusion
As early as the pre-natal period, human skin serves as barrier tissue that is confronted with and must adapt to a unique and highly individualized array of pathogens, sensitizing agents, and environmental exposures. Though the largest and most accessible organ providing this critical defense, skin is frequently overlooked in the NK cell literature, likely in part due to the heterogeneity in defining trNK cell origin and subsets. We submit that skin represents one of the secondary lymphoid organs of the human body, and that further characterization of skin trNK cells will reveal a highly plastic and adaptable immune cell population that is critical not only for skin immune development and maintenance of homeostasis, but also with the potential to provide unique therapeutic avenues for treatment of cutaneous infection and inflammatory and malignant diseases.
Author contributions
KJ: Writing – original draft, Writing – review & editing, Conceptualization. DL: Funding acquisition, Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Acknowledgments
We thank Clarity in Science Editing and Writing (clarityinscience.com) for English language scientific editing.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Luci C and Tomasello E. Natural killer cells: detectors of stress. Int J Biochem Cell Biol. (2008) 40:2335–40. doi: 10.1016/j.biocel.2008.05.001
2. Grudzien M and Rapak A. Effect of natural compounds on NK cell activation. J Immunol Res. (2018) 2018:4868417. doi: 10.1155/2018/4868417
3. Lanier LL, Ruitenberg JJ, and Phillips JH. Functional and biochemical analysis of CD16 antigen on natural killer cells and granulocytes. J Immunol. (1988) 141:3478–85. doi: 10.4049/jimmunol.141.10.3478
4. Moseman JE, Foltz JA, Sorathia K, Heipertz EL, and Lee DA. Evaluation of serum-free media formulations in feeder cell-stimulated expansion of natural killer cells. Cytotherapy. (2020) 22:322–8. doi: 10.1016/j.jcyt.2020.02.002
5. Tikoo S, Jain R, Kurz AR, and Weninger W. The lymphoid cell network in the skin. Immunol Cell Biol. (2018) 96:485–96. doi: 10.1111/imcb.12026
6. Daussy C, Faure F, Mayol K, Viel S, Gasteiger G, Charrier E, et al. T-bet and Eomes instruct the development of two distinct natural killer cell lineages in the liver and in the bone marrow. J Exp Med. (2014) 211:563–77. doi: 10.1084/jem.20131560
7. Pikovskaya O, Chaix J, Rothman NJ, Collins A, Chen YH, Scipioni AM, et al. Cutting edge: Eomesodermin is sufficient to direct type 1 innate lymphocyte development into the conventional NK lineage. J Immunol. (2016) 196:1449–54. doi: 10.4049/jimmunol.1502396
8. Gordon SM, Chaix J, Rupp LJ, Wu J, Madera S, Sun JC, et al. The transcription factors T-bet and Eomes control key checkpoints of natural killer cell maturation. Immunity. (2012) 36:55–67. doi: 10.1016/j.immuni.2011.11.016
9. Yudanin NA, Schmitz F, Flamar AL, Thome JJC, Tait Wojno E, Moeller JB, et al. Spatial and temporal mapping of human innate lymphoid cells reveals elements of tissue specificity. Immunity. (2019) 50:505–19.e4. doi: 10.1016/j.immuni.2019.01.012
10. Friedrich C, Taggenbrock RLRE, Doucet-Ladevèze R, Golda G, Moenius R, Arampatzi P, et al. Effector differentiation downstream of lineage commitment in ILC1s is driven by Hobit across tissues. Nat Immunol. (2021) 22:1256–67. doi: 10.1038/s41590-021-01013-0
11. Klose CSN, Flach M, Möhle L, Rogell L, Hoyler T, Ebert K, et al. Differentiation of type 1 ILCs from a common progenitor to all helper-like innate lymphoid cell lineages. Cell. (2014) 157:340–56. doi: 10.1016/j.cell.2014.03.030
12. Mjösberg J, Bernink J, Golebski K, Karrich JJ, Peters CP, Blom B, et al. The transcription factor GATA3 is essential for the function of human type 2 innate lymphoid cells. Immunity. (2012) 37:649–59. doi: 10.1016/j.immuni.2012.08.015
13. Sojka DK, Plougastel-Douglas B, Yang L, Pak-Wittel MA, Artyomov MN, Ivanova Y, et al. Tissue-resident natural killer (NK) cells are cell lineages distinct from thymic and conventional splenic NK cells. Elife. (2014) 3:e01659. doi: 10.7554/eLife.01659
14. Torcellan T, Friedrich C, Doucet-Ladevèze R, Ossner T, Solé VV, Riedmann S, et al. Circulating NK cells establish tissue residency upon acute infection of skin and mediate accelerated effector responses to secondary infection. Immunity. (2024) 57:124–140.e7. doi: 10.1016/j.immuni.2023.11.018
15. Fehniger TA, Herbein G, Yu H, Para MI, Bernstein ZP, O'Brien WA, et al. Natural killer cells from HIV-1+ patients produce C-C chemokines and inhibit HIV-1 infection. J Immunol. (1998) 161:6433–8. doi: 10.4049/jimmunol.161.11.6433
16. Fehniger TA, Shah MH, Turner MJ, VanDeusen JB, Whitman SP, Cooper MA, et al. Differential cytokine and chemokine gene expression by human NK cells following activation with IL-18 or IL-15 in combination with IL-12: implications for the innate immune response. J Immunol. (1999) 162:4511–20. doi: 10.4049/jimmunol.162.8.4511
17. McFarland AP, Yalin A, Wang SY, Cortez VS, Landsberger T, Sudan R, et al. Multi-tissue single-cell analysis deconstructs the complex programs of mouse natural killer and type 1 innate lymphoid cells in tissues and circulation. Immunity. (2021) 54:1320–37.e4. doi: 10.1016/j.immuni.2021.03.024
18. Yang C, Siebert JR, Burns R, Gerbec ZJ, Bonacci B, Rymaszewski A, et al. Heterogeneity of human bone marrow and blood natural killer cells defined by single-cell transcriptome. Nat Commun. (2019) 10:3931. doi: 10.1038/s41467-019-11947-7
19. Fuchs A, Vermi W, Lee JS, Lonardi S, Gilfillan S, Newberry RD, et al. Intraepithelial type 1 innate lymphoid cells are a unique subset of IL-12- and IL-15-responsive IFN-γ-producing cells. Immunity. (2013) 38:769–81. doi: 10.1016/j.immuni.2013.02.010
20. Wang X, Peng H, Cong J, Wang X, Lian Z, Wei H, et al. Memory formation and long-term maintenance of IL-7Rα+ ILC1s via a lymph node-liver axis. Nat Commun. (2018) 9:4854. doi: 10.1038/s41467-018-07405-5
21. Di Censo C, Marotel M, Mattiola I, Müller L, Scarno G, Pietropaolo G, et al. Granzyme A and CD160 expression delineates ILC1 with graded functions in the mouse liver. Eur J Immunol. (2021) 51:2568–75. doi: 10.1002/eji.202149209
22. Krueger PD, Narayanan S, Surette FA, Brown MG, Sung SJ, and Hahn YS. Murine liver-resident group 1 innate lymphoid cells regulate optimal priming of anti-viral CD8+ T cells. J Leukoc Biol. (2017) 101:329–38. doi: 10.1189/jlb.3A0516-225R
23. Hudspeth K, Donadon M, Cimino M, Pontarini E, Tentorio P, Preti M, et al. Human liver-resident CD56(bright)/CD16(neg) NK cells are retained within hepatic sinusoids via the engagement of CCR5 and CXCR6 pathways. J Autoimmun. (2016) 66:40–50. doi: 10.1016/j.jaut.2015.08.011
24. Saudemont A, Jouy N, Hetuin D, and Quesnel B. NK cells that are activated by CXCL10 can kill dormant tumor cells that resist CTL-mediated lysis and can express B7-H1 that stimulates T cells. Blood. (2005) 105:2428–35. doi: 10.1182/blood-2004-09-3458
25. Tato CM, Martins GA, High FA, DiCioccio CB, Reiner SL, and Hunter CA. Cutting edge: innate production of IFN-γ by NK cells is independent of epigenetic modification of the IFN-γ promoter. J Immunol. (2004) 173:1514–7. doi: 10.4049/jimmunol.173.3.1514
26. Arase H, Arase N, and Saito T. Interferon gamma production by natural killer (NK) cells and NK1.1+ T cells upon NKR-P1 cross-linking. J Exp Med. (1996) 183:2391–6. doi: 10.1084/jem.183.5.2391
27. Wang R, Jaw JJ, Stutzman NC, Zou Z, and Sun PD. Natural killer cell-produced IFN-γ and TNF-α induce target cell cytolysis through up-regulation of ICAM-1. J Leukoc Biol. (2012) 91:299–309. doi: 10.1189/jlb.0611308
28. Weizman OE, Adams NM, Schuster IS, Krishna C, Pritykin Y, Lau C, et al. ILC1 confer early host protection at initial sites of viral infection. Cell. (2017) 171:795–808.e12. doi: 10.1016/j.cell.2017.09.052
29. Seehus CR, Kadavallore A, Torre B, Yeckes AR, Wang Y, Tang J, et al. Alternative activation generates IL-10 producing type 2 innate lymphoid cells. Nat Commun. (2017) 8:1900. doi: 10.1038/s41467-017-02023-z
30. Salimi M, Barlow JL, Saunders SP, Xue L, Gutowska-Owsiak D, Wang X, et al. A role for IL-25 and IL-33-driven type-2 innate lymphoid cells in atopic dermatitis. J Exp Med. (2013) 210:2939–50. doi: 10.1084/jem.20130351
31. Cupedo T, Crellin NK, Papazian N, Weijer K, Grogan JL, et al. Human fetal lymphoid tissue–inducer cells are interleukin 17–producing precursors to RORC+ CD127+ natural killer–like cells. Nature Immunology (2009) 10:66–74. doi: 10.1038/ni.1668
32. Cella M, Fuchs A, Vermi W, Facchetti F, Otero K, Lennerz JKM, et al. A human natural killer cell subset provides an innate source of IL-22 for mucosal immunity. Nature. (2008) 457:722–5. doi: 10.1038/nature07537
33. Pearson C, Thornton EE, McKenzie B, Schaupp AL, Huskens N, Griseri T, et al. ILC3 GM-CSF production and mobilisation orchestrate acute intestinal inflammation. Elife. (2016) 5:e10066. doi: 10.7554/eLife.10066
34. Gogoleva VS, Kuprash DV, Grivennikov SI, Tumanov AV, Kruglov AA, and Nedospasov SA. LTα, TNF, and ILC3 in peyer's patch organogenesis. Cells. (2022) 11:1970. doi: 10.3390/cells11121970
35. Gury-BenAri M, Thaiss CA, Serafini N, Winter DR, Giladi A, Lara-Astiaso D, et al. The spectrum and regulatory landscape of intestinal innate lymphoid cells are shaped by the microbiome. Cell. (2016) 166:1231–46. doi: 10.1016/j.cell.2016.07.043
36. Cella M, Otero K, and Colonna M. Expansion of human NK-22 cells with IL-7, IL-2, and IL-1beta reveals intrinsic functional plasticity. Proc Natl Acad Sci U S A. (2010) 107:10961–6. doi: 10.1073/pnas.1005641107
37. Burkhardt JK, Hester S, Lapham CK, and Argon Y. The lytic granules of natural killer cells are dual-function organelles combining secretory and pre-lysosomal compartments. J Cell Biol. (1990) 111:2327-2340. doi: 10.1083/jcb.111.6.2327
38. Schuster IS, Wikstrom ME, Brizard G, Coudert JD, Estcourt MJ, Manzur M, et al. TRAIL+ NK cells control CD4+ T cell responses during chronic viral infection to limit autoimmunity. Immunity. (2014) 41:646–56. doi: 10.1016/j.immuni.2014.09.013
39. Bradley M, Zeytun A, Rafi-Janajreh A, Nagarkatti PS, and Nagarkatti M. Role of spontaneous and interleukin-2-induced natural killer cell activity in the cytotoxicity and rejection of Fas+ and Fas- tumor cells. Blood. (1998) 92:4248–55. doi: 10.1182/blood.V92.11.4248
40. Kayagaki N, Yamaguchi N, Nakayama M, Takeda K, Akiba H, Tsutsui H, et al. Expression and function of TNF-related apoptosis-inducing ligand on murine activated NK cells. J Immunol. (1999) 163:1906–13. doi: 10.4049/jimmunol.163.4.1906
41. Picarda G, Ghosh R, McDonald B, Verma S, Thiault N, El Morabiti R, et al. Cytomegalovirus evades TRAIL-mediated innate lymphoid cell 1 defenses. J Virol. (2019) 93:e00617–19. doi: 10.1128/JVI.00617-19
42. Robinette ML, Fuchs A, Cortez VS, Lee JS, Wang Y, Durum SK, et al. Transcriptional programs define molecular characteristics of innate lymphoid cell classes and subsets. Nat Immunol. (2015) 16:306–17. doi: 10.1038/ni.3094
43. Xue L, Salimi M, Panse I, Mjösberg JM, McKenzie AN, Spits H, et al. Prostaglandin D2 activates group 2 innate lymphoid cells through chemoattractant receptor-homologous molecule expressed on TH2 cells. J Allergy Clin Immunol. (2014) 133:1184–94. doi: 10.1016/j.jaci.2013.10.056
44. Bando JK, Gilfillan S, Song C, McDonald KG, Huang SC, Newberry RD, et al. The tumor necrosis factor superfamily member RANKL suppresses effector cytokine production in group 3 innate lymphoid cells. Immunity. (2018) 48:1208–1219.e4. doi: 10.1016/j.immuni.2018.04.012
45. Stegmann KA, Robertson F, Hansi N, Gill U, Pallant C, Christophides T, et al. CXCR6 marks a novel subset of T-bet(lo)Eomes(hi) natural killer cells residing in human liver. Sci Rep. (2016) 6:26157. doi: 10.1038/srep26157
46. Srivastava RK, Sapra L, Bhardwaj A, Mishra PK, Verma B, and Baig Z. Unravelling the immunobiology of innate lymphoid cells (ILCs): Implications in health and disease. Cytokine Growth Factor Rev. (2023) 74:56–75. doi: 10.1016/j.cytogfr.2023.09.002
47. Riggan L, Freud AG, and O'Sullivan TE. True detective: unraveling group 1 innate lymphocyte heterogeneity. Trends Immunol. (2019) 40:909–21. doi: 10.1016/j.it.2019.08.005
48. Meharra EJ, Schön M, Hassett D, Parker C, Havran W, and Gardner H. Reduced gut intraepithelial lymphocytes in VLA1 null mice. Cell Immunol. (2000) 201:1–5. doi: 10.1006/cimm.2000.1630
49. Shiow LR, Rosen DB, Brdicková N, Xu Y, An J, Lanier LL, et al. CD69 acts downstream of interferon-alpha/beta to inhibit S1P1 and lymphocyte egress from lymphoid organs. Nature. (2006) 440:540–4. doi: 10.1038/nature04606
50. Peng H, Jiang X, Chen Y, Sojka DK, Wei H, Gao X, et al. Liver-resident NK cells confer adaptive immunity in skin-contact inflammation. J Clin Invest. (2013) 123:1444–56. doi: 10.1172/JCI66381
51. Mackay LK, Minnich M, Kragten NA, Liao Y, Nota B, Seillet C, et al. Hobit and Blimp1 instruct a universal transcriptional program of tissue residency in lymphocytes. Science. (2016) 352:459–63. doi: 10.1126/science.aad2035
52. Crinier A, Milpied P, Escalière B, Piperoglou C, Galluso J, Balsamo A, et al. High-dimensional single-cell analysis identifies organ-specific signatures and conserved NK cell subsets in humans and mice. Immunity. (2018) 49:971–986.e5. doi: 10.1016/j.immuni.2018.09.009
53. Rebuffet L, Melsen JE, Escalière B, Basurto-Lozada D, Bhandoola A, Björkström NK, et al. High-dimensional single-cell analysis of human natural killer cell heterogeneity. Nat Immunol. (2024) 25:1474–88. doi: 10.1038/s41590-024-01883-0
54. Chaudhry MZ and Belz GT. Chameleon impersonation of NK cells and ILC1s. Nat Immunol. (2024) 25:1313–5. doi: 10.1038/s41590-024-01886-x
55. Yomogida K, Bigley TM, Trsan T, Gilfillan S, Cella M, Yokoyama WM, et al. Hobit confers tissue-dependent programs to type 1 innate lymphoid cells. Proc Natl Acad Sci U S A. (2021) 118:e2117965118. doi: 10.1073/pnas.2117965118
56. Jaeger N, Antonova AU, Kreisel D, Roan F, Lantelme E, Ziegler SF, et al. Diversity of group 1 innate lymphoid cells in human tissues. Nat Immunol. (2024) 25:1460–73. doi: 10.1038/s41590-024-01885-y
57. Nayyar G, Chu Y, and Cairo MS. Overcoming resistance to natural killer cell based immunotherapies for solid tumors. Front Oncol. (2019) 9:51. doi: 10.3389/fonc.2019.00051
58. Frey M, Packianathan NB, Fehniger TA, Ross ME, Wang WC, Stewart CC, et al. Differential expression and function of L-selectin on CD56bright and CD56dim natural killer cell subsets. J Immunol. (1998) 161:400–8. doi: 10.4049/jimmunol.161.1.400
59. Nagler A, Lanier LL, and Phillips JH. Constitutive expression of high affinity interleukin 2 receptors on human CD16-natural killer cells in vivo. J Exp Med. (1990) 171:1527–33. doi: 10.1084/jem.171.5.1527
60. Caligiuri MA, Murray C, Robertson MJ, Wang E, Cochran K, Cameron C, et al. Selective modulation of human natural killer cells in vivo after prolonged infusion of low dose recombinant interleukin 2. J Clin Invest. (1993) 91:123–32. doi: 10.1172/JCI116161
61. Schuster IS, Andoniou CE, and Degli-Esposti MA. Tissue-resident memory NK cells: Homing in on local effectors and regulators. Immunol Rev. (2024) 323:54–60. doi: 10.1111/imr.13332
62. Michel T, Poli A, Cuapio A, Briquemont B, Iserentant G, Ollert M, et al. Human CD56bright NK cells: an update. J Immunol. (2016) 196:2923–31. doi: 10.4049/jimmunol.1502570
63. Freud AG, Mundy-Bosse BL, Yu J, and Caligiuri MA. The broad spectrum of human natural killer cell diversity. Immunity. (2017) 47:820–33. doi: 10.1016/j.immuni.2017.10.008
64. Lopes N, Galluso J, Escalière B, Carpentier S, Kerdiles YM, and Vivier E. Tissue-specific transcriptional profiles and heterogeneity of natural killer cells and group 1 innate lymphoid cells. Cell Rep Med. (2022) 3:100812. doi: 10.1016/j.xcrm.2022.100812
65. Cortez VS, Cervantes-Barragan L, Robinette ML, Bando JK, Wang Y, Geiger TL, et al. Transforming growth factor-β Signaling guides the differentiation of innate lymphoid cells in salivary glands. Immunity. (2016) 44:1127–39. doi: 10.1016/j.immuni.2016.03.007
66. Kabashima K, Honda T, Ginhoux F, and Egawa G. The immunological anatomy of the skin. Nat Rev Immunol. (2019) 19:19–30. doi: 10.1038/s41577-018-0084-5
67. Wolf K, Ibrahim SA, Schneiderman S, Riehl V, Dambaeva S, and Beaman K. Conventional natural killer cells control vascular remodeling in the uterus during pregnancy by acidifying the extracellular matrix with a2V. Biol Reprod. (2023) 108:121–32. doi: 10.1093/biolre/ioac184
68. Ran GH, Lin YQ, Tian L, Zhang T, Yan DM, Yu JH, et al. Natural killer cell homing and trafficking in tissues and tumors: from biology to application. Signal Transduct Target Ther. (2022) 7:205. doi: 10.1038/s41392-022-01058-z
69. Meininger I, Carrasco A, Rao A, Soini T, Kokkinou E, and Mjösberg J. Tissue-specific features of innate lymphoid cells. Trends Immunol. (2020) 41:902–17. doi: 10.1016/j.it.2020.08.009
70. Hazenberg MD and Spits H. Human innate lymphoid cells. Blood. (2014) 124:700–9. doi: 10.1182/blood-2013-11-427781
71. Chen L, Youssef Y, Robinson C, Ernst GF, Carson MY, Young KA, et al. CD56 expression marks human group 2 innate lymphoid cell divergence from a shared NK cell and group 3 innate lymphoid cell developmental pathway. Immunity. (2018) 49:464–476.e4. doi: 10.1016/j.immuni.2018.08.010
72. Lopez-Vergès S, Milush JM, Pandey S, York VA, Arakawa-Hoyt J, Pircher H, et al. CD57 defines a functionally distinct population of mature NK cells in the human CD56dimCD16+ NK-cell subset. Blood. (2010) 116:3865–74. doi: 10.1182/blood-2010-04-282301
73. Del Zotto G, Vacca P, Moretta L, and Quatrini L. CPHEN-15: Comprehensive phenotyping of human peripheral blood helper-ILCs by flow cytometry. Cytometry A. (2023) 103:378–82. doi: 10.1002/cyto.a.24717
74. Bernink JH, Mjösberg J, and Spits H. Human ILC1: to be or not to be. Immunity. (2017) 46:756–7. doi: 10.1016/j.immuni.2017.05.001
75. Koprivica I, Stanisavljević S, Mićanović D, Jevtić B, Stojanović I, and Miljković Đ. ILC3: a case of conflicted identity. Front Immunol. (2023) 14:1271699. doi: 10.3389/fimmu.2023.1271699
76. Qin M, Fang Y, Zheng Q, Peng M, Wang L, Sang X, et al. Tissue microenvironment induces tissue specificity of ILC2. Cell Death Discov. (2024) 10:324. doi: 10.1038/s41420-024-02096-y
77. Hamann I, Unterwalder N, Cardona AE, Meisel C, Zipp F, Ransohoff RM, et al. Analyses of phenotypic and functional characteristics of CX3CR1-expressing natural killer cells. Immunology. (2011) 133:62–73. doi: 10.1111/j.1365-2567.2011.03409.x
78. Melsen JE, Lugthart G, Lankester AC, and Schilham MW. Human circulating and tissue-resident CD56(bright) natural killer cell populations. Front Immunol. (2016) 7:262. doi: 10.3389/fimmu.2016.00262
79. Kobayashi T and Moro K. Tissue-specific diversity of group 2 innate lymphoid cells in the skin. Front Immunol. (2022) 13:885642. doi: 10.3389/fimmu.2022.885642
80. Rojas-Diaz JM, Solorzano-Ibarra F, Garcia-Barrientos NT, Klimov-Kravtchenko K, Guitron-Aviña MS, Cruz-Ramos JA, et al. Uncovering the expression pattern of the costimulatory receptors ICOS, 4-1BB, and OX-40 in exhausted peripheral and tumor-infiltrating natural killer cells from patients with cervical cancer. Int J Mol Sci. (2024) 25:8775. doi: 10.3390/ijms25168775
81. Yang J, Zhao L, Xu M, and Xiong N. Establishment and function of tissue-resident innate lymphoid cells in the skin. Protein Cell. (2017) 8:489–500. doi: 10.1007/s13238-017-0388-4
82. Sun Z, Vattepu R, and Zhang S. Chemokines and innate lymphoid cells in skin inflammation. Cells. (2021) 10:3074. doi: 10.3390/cells10113074
83. Spits H, Bernink JH, and Lanier L. NK cells and type 1 innate lymphoid cells: partners in host defense. Nat Immunol. (2016) 17:758–64. doi: 10.1038/ni.3482
84. Hashemi E and Malarkannan S. Tissue-resident NK cells: development, maturation, and clinical relevance. Cancers (Basel). (2020) 12:1553. doi: 10.3390/cancers12061553
85. Reynolds G, Vegh P, Fletcher J, Poyner EFM, Stephenson E, Goh I, et al. Developmental cell programs are co-opted in inflammatory skin disease. Science. (2021) 371:eaba6500. doi: 10.1126/science.aba6500
86. Yang J, Restori KH, Xu M, Song EH, Zhao L, Hu S, et al. Preferential perinatal development of skin-homing NK1.1+ Innate lymphoid cells for regulation of cutaneous microbiota colonization. iScience. (2020) 23:101014. doi: 10.1016/j.isci.2020.101014
87. Wang WB, Lin YD, Zhao L, Liao C, Zhang Y, Davila M, et al. Developmentally programmed early-age skin localization of iNKT cells supports local tissue development and homeostasis. Nat Immunol. (2023) 24:225–38. doi: 10.1038/s41590-022-01399-5
88. Almeida FF, Tenno M, Brzostek J, Li JL, Allies G, Hoeffel G, et al. Identification of a novel lymphoid population in the murine epidermis. Sci Rep. (2015) 5:12554. doi: 10.1038/srep12554
89. Dyring-Andersen B, Geisler C, Agerbeck C, Lauritsen JP, Gúdjonsdottir SD, Skov L, et al. Increased number and frequency of group 3 innate lymphoid cells in nonlesional psoriatic skin. Br J Dermatol. (2014) 170:609–16. doi: 10.1111/bjd.12658
90. Teunissen MBM, Munneke JM, Bernink JH, Spuls PI, Res PCM, Te Velde A, et al. Composition of innate lymphoid cell subsets in the human skin: enrichment of NCR(+) ILC3 in lesional skin and blood of psoriasis patients. J Invest Dermatol. (2014) 134:2351–60. doi: 10.1038/jid.2014.146
91. Villanova F, Flutter B, Tosi I, Grys K, Sreeneebus H, Perera GK, et al. Characterization of innate lymphoid cells in human skin and blood demonstrates increase of NKp44+ ILC3 in psoriasis. J Invest Dermatol. (2014) 134:984–91. doi: 10.1038/jid.2013.477
92. Bielecki P, Riesenfeld SJ, Hütter JC, Torlai Triglia E, Kowalczyk MS, Ricardo-Gonzalez RR, et al. Skin-resident innate lymphoid cells converge on a pathogenic effector state. Nature. (2021) 592:128–32. doi: 10.1038/s41586-021-03188-w
93. Rak GD, Osborne LC, Siracusa MC, Kim BS, Wang K, Bayat A, et al. IL-33-dependent group 2 innate lymphoid cells promote cutaneous wound healing. J Invest Dermatol. (2016) 136:487–96. doi: 10.1038/JID.2015.406
94. Dutton EE, Gajdasik DW, Willis C, Fiancette R, Bishop EL, Camelo A, et al. Peripheral lymph nodes contain migratory and resident innate lymphoid cell populations. Sci Immunol. (2019) 4:eaau8082. doi: 10.1126/sciimmunol.aau8082
95. Ebert LM, Meuter S, and Moser B. Homing and function of human skin gammadelta T cells and NK cells: relevance for tumor surveillance. J Immunol. (2006) 176:4331–6. doi: 10.4049/jimmunol.176.7.4331
96. Jing Y, Ni Z, Wu J, Higgins L, Markowski TW, Kaufman DS, et al. Identification of an ADAM17 cleavage region in human CD16 (FcγRIII) and the engineering of a non-cleavable version of the receptor in NK cells. PloS One. (2015) 10:e0121788. doi: 10.1371/journal.pone.0121788
97. Oliviero B, Mantovani S, Varchetta S, Mele D, Grossi G, Ludovisi S, et al. Hepatitis C virus-induced NK cell activation causes metzincin-mediated CD16 cleavage and impaired antibody-dependent cytotoxicity. J Hepatol. (2017) 66:1130–7. doi: 10.1016/j.jhep.2017.01.032
98. Romee R, Foley B, Lenvik T, Wang Y, Zhang B, Ankarlo D, et al. NK cell CD16 surface expression and function is regulated by a disintegrin and metalloprotease-17 (ADAM17). Blood. (2013) 121:3599–608. doi: 10.1182/blood-2012-04-425397
99. Peschon JJ, Slack JL, Reddy P, Stocking KL, Sunnarborg SW, Lee DC, et al. An essential role for ectodomain shedding in mammalian development. Science. (1998) 282:1281–4. doi: 10.1126/science.282.5392.1281
100. Franzke CW, Cobzaru C, Triantafyllopoulou A, Löffek S, Horiuchi K, Threadgill DW, et al. Epidermal ADAM17 maintains the skin barrier by regulating EGFR ligand-dependent terminal keratinocyte differentiation. J Exp Med. (2012) 209:1105–19. doi: 10.1084/jem.20112258
101. Pham DH, Kim JS, Kim SK, Shin DJ, Uong NT, Hyun H, et al. Effects of ADAM10 and ADAM17 inhibitors on natural killer cell expansion and antibody-dependent cellular cytotoxicity against breast cancer cells in vitro. Anticancer Res. (2017) 37:5507–13. doi: 10.21873/anticanres.11981
102. Peng L, Cook K, Xu L, Cheng L, Damschroder M, Gao C, et al. Molecular basis for the mechanism of action of an anti-TACE antibody. MAbs. (2016) 8:1598–605. doi: 10.1080/19420862.2016.1226716
103. Mishra HK, Pore N, Michelotti EF, and Walcheck B. Anti-ADAM17 monoclonal antibody MEDI3622 increases IFNγ production by human NK cells in the presence of antibody-bound tumor cells. Cancer Immunol Immunother. (2018) 67:1407–16. doi: 10.1007/s00262-018-2193-1
104. Post M, Cuapio A, Osl M, Lehmann D, Resch U, Davies DM, et al. The transcription factor ZNF683/HOBIT regulates human NK-cell development. Front Immunol. (2017) 8:535. doi: 10.3389/fimmu.2017.00535
105. Nixon BG, Chou C, Krishna C, Dadi S, Michel AO, Cornish AE, et al. Cytotoxic granzyme C-expressing ILC1s contribute to antitumor immunity and neonatal autoimmunity. Sci Immunol. (2022) 7:eabi8642. doi: 10.1126/sciimmunol.abi8642
106. Friedrich C, Torcellan T, and Gasteiger G. Differentiation of tissue-resident NK cells: Why origin and context matter. Immunity. (2025) 58:517–9. doi: 10.1016/j.immuni.2025.02.020
107. Bunting MD, Vyas M, Requesens M, Langenbucher A, Schiferle EB, Manguso RT, et al. Extracellular matrix proteins regulate NK cell function in peripheral tissues. Sci Adv. (2022) 8:eabk3327. doi: 10.1126/sciadv.abk3327
108. Peng H and Sun R. Liver-resident NK cells and their potential functions. Cell Mol Immunol. (2017) 14:890–4. doi: 10.1038/cmi.2017.72
109. Havran WL, Grell S, Duwe G, Kimura J, Wilson A, Kruisbeek AM, et al. Limited diversity of T-cell receptor gamma-chain expression of murine Thy-1+ dendritic epidermal cells revealed by V gamma 3-specific monoclonal antibody. Proc Natl Acad Sci U S A. (1989) 86:4185–9. doi: 10.1073/pnas.86.11.4185
110. Gray EE, Suzuki K, and Cyster JG. Cutting edge: Identification of a motile IL-17-producing gammadelta T cell population in the dermis. J Immunol. (2011) 186:6091–5. doi: 10.4049/jimmunol.1100427
111. Scharschmidt TC, Vasquez KS, Truong HA, Gearty SV, Pauli ML, Nosbaum A, et al. A wave of regulatory T cells into neonatal skin mediates tolerance to commensal microbes. Immunity. (2015) 43:1011–21. doi: 10.1016/j.immuni.2015.10.016
112. Richmond JM, Strassner JP, Essien KI, and Harris JE. T-cell positioning by chemokines in autoimmune skin diseases. Immunol Rev. (2019) 289:186–204. doi: 10.1111/imr.12762
113. Dustin ML, Rothlein R, Bhan AK, Dinarello CA, and Springer TA. Induction by IL 1 and interferon-gamma: tissue distribution, biochemistry, and function of a natural adherence molecule (ICAM-1). J Immunol. (1986) 137:245–54. doi: 10.4049/jimmunol.137.1.245
114. Griffiths CE, Voorhees JJ, and Nickoloff BJ. Characterization of intercellular adhesion molecule-1 and HLA-DR expression in normal and inflamed skin: modulation by recombinant gamma interferon and tumor necrosis factor. J Am Acad Dermatol. (1989) 20:617–29. doi: 10.1016/s0190-9622(89)70073-6
115. Barker JN, Sarma V, Mitra RS, Dixit VM, and Nickoloff BJ. Marked synergism between tumor necrosis factor-alpha and interferon-gamma in regulation of keratinocyte-derived adhesion molecules and chemotactic factors. J Clin Invest. (1990) 85:605–8. doi: 10.1172/JCI114481
116. Groves RW, Ross EL, Barker JN, and MacDonald DM. Vascular cell adhesion molecule-1: expression in normal and diseased skin and regulation in vivo by interferon gamma. J Am Acad Dermatol. (1993) 29:67–72. doi: 10.1016/0190-9622(93)70154-l
117. Petzelbauer P, Pober JS, Keh A, and Braverman IM. Inducibility and expression of microvascular endothelial adhesion molecules in lesional, perilesional, and uninvolved skin of psoriatic patients. J Invest Dermatol. (1994) 103:300–5. doi: 10.1111/1523-1747.ep12394720
118. Ottaviani C, Nasorri F, Bedini C, de Pità O, Girolomoni G, and Cavani A. CD56brightCD16(-) NK cells accumulate in psoriatic skin in response to CXCL10 and CCL5 and exacerbate skin inflammation. Eur J Immunol. (2006) 36:118–28. doi: 10.1002/eji.200535243
119. Xia M, Hu S, Fu Y, Jin W, Yi Q, Matsui Y, et al. CCR10 regulates balanced maintenance and function of resident regulatory and effector T cells to promote immune homeostasis in the skin. J Allergy Clin Immunol. (2014) 134:634–644.e10. doi: 10.1016/j.jaci.2014.03.010
120. Yang J, Hu S, Zhao L, Kaplan DH, Perdew GH, and Xiong N. Selective programming of CCR10(+) innate lymphoid cells in skin-draining lymph nodes for cutaneous homeostatic regulation. Nat Immunol. (2016) 17:48–56. doi: 10.1038/ni.3312
121. Mao Y, Tao R, Cao X, Bao Q, Wang D, and Zhao Y. Innate lymphoid cells regulate radiation-induced skin damage via CCR10 signaling. Int J Radiat Biol. (2020) 96:1157–64. doi: 10.1080/09553002.2020.1793013
122. Homey B, Alenius H, Müller A, Soto H, Bowman EP, Yuan W, et al. CCL27-CCR10 interactions regulate T cell-mediated skin inflammation. Nat Med. (2002) 8:157–65. doi: 10.1038/nm0202-157
123. Davila ML, Xu M, Huang C, Gaddes ER, Winter L, Cantorna MT, et al. CCL27 is a crucial regulator of immune homeostasis of the skin and mucosal tissues. iScience. (2022) 25:104426. doi: 10.1016/j.isci.2022.104426
124. Tsuchiyama J, Yoshino T, Toba K, Harada N, Nishiuchi R, Akagi T, et al. Induction and characterization of cutaneous lymphocyte antigen on natural killer cells. Br J Haematol. (2002) 118:654–62. doi: 10.1046/j.1365-2141.2002.03608.x
125. de Lima JF, Teixeira FME, Ramos YÁL, de Carvalho GC, Castelo Branco ACC, Pereira NV, et al. Outlining the skin-homing and circulating CLA+NK cells in patients with severe atopic dermatitis. Sci Rep. (2024) 14:2663. doi: 10.1038/s41598-024-53224-8
126. Ferran M, Romeu ER, Rincón C, Sagristà M, Giménez Arnau AM, Celada A, et al. Circulating CLA+ T lymphocytes as peripheral cell biomarkers in T-cell-mediated skin diseases. Exp Dermatol. (2013) 22:439–42. doi: 10.1111/exd.12154
127. Sun Z, Sen H, Zhu X, and Islam SA. Cutting edge: CCR8 signaling regulates IL-25- and IL-33-responsive skin group 2 innate lymphoid cell migration and function. J Immunol. (2023) 211:1751–5. doi: 10.4049/jimmunol.2200829
128. McCully ML and Moser B. The human cutaneous chemokine system. Front Immunol. (2011) 2:33. doi: 10.3389/fimmu.2011.00033
129. Yabe R, Shimizu K, Shimizu S, Azechi S, Choi BI, Sudo K, et al. CCR8 regulates contact hypersensitivity by restricting cutaneous dendritic cell migration to the draining lymph nodes. Int Immunol. (2015) 27:169–81. doi: 10.1093/intimm/dxu098
130. Zimmer CL, Cornillet M, Solà-Riera C, Cheung KW, Ivarsson MA, Lim MQ, et al. NK cells are activated and primed for skin-homing during acute dengue virus infection in humans. Nat Commun. (2019) 10. doi: 10.1038/s41467-019-11878-3
131. Covre LP, Devine OP, Garcia de Moura R, Vukmanovic-Stejic M, Dietze R, Ribeiro-Rodrigues R, et al. Compartmentalized cytotoxic immune response leads to distinct pathogenic roles of natural killer and senescent CD8+ T cells in human cutaneous leishmaniasis. Immunology. (2020) 159:429–40. doi: 10.1111/imm.13173
132. Kawakami Y, Ando T, Lee JR, Kim G, Kawakami Y, Nakasaki T, et al. Defective natural killer cell activity in a mouse model of eczema herpeticum. J Allergy Clin Immunol. (2017) 139:997–1006.e10. doi: 10.1016/j.jaci.2016.06.034
133. Mack MR, Brestoff JR, Berrien-Elliott MM, Trier AM, Yang TB, McCullen M, et al. Blood natural killer cell deficiency reveals an immunotherapy strategy for atopic dermatitis. Sci Transl Med. (2020) 12:eaay1005. doi: 10.1126/scitranslmed.aay1005
134. Kawakami Y, Tomimori Y, Yumoto K, Hasegawa S, Ando T, Tagaya Y, et al. Inhibition of NK cell activity by IL-17 allows vaccinia virus to induce severe skin lesions in a mouse model of eczema vaccinatum. J Exp Med. (2009) 206:1219–25. doi: 10.1084/jem.20082835
135. O’Leary JG, Goodarzi M, Drayton DL, and von Andrian UH. T cell– and B cell–independent adaptive immunity mediated by natural killer cells. Nat Immunol. (2006) 7:507–16. doi: 10.1038/ni1332
136. Carbone T, Nasorri F, Pennino D, Eyerich K, Foerster S, Cifaldi L, et al. CD56highCD16-CD62L- NK cells accumulate in allergic contact dermatitis and contribute to the expression of allergic responses. J Immunol. (2010) 184:1102–10. doi: 10.4049/jimmunol.0902518
137. Mi QS, Dimitrion P, Hamzavi I, Yin C, Loveless I, Toor J, et al. Dysregulated CD38 expression in blood and skin immune cells of patients with hidradenitis suppurativa. Res Sq. (2023) 184, rs.3.rs–2609421. doi: 10.21203/rs.3.rs-2609421/v1
138. Yoshino T, Nakamura S, Suzumiya J, Niitsu N, Ohshima K, Tsuchiyama J, et al. Expression of cutaneous lymphocyte antigen is associated with a poor outcome of nasal-type natural killer-cell lymphoma. Br J Haematol. (2002) 118:482–7. doi: 10.1046/j.1365-2141.2002.03607.x
139. da Silva IP, Gallois A, Jimenez-Baranda S, Khan S, Anderson AC, Kuchroo VK, et al. Reversal of NK-cell exhaustion in advanced melanoma by Tim-3 blockade. Cancer Immunol Res. (2014) 2:410–22. doi: 10.1158/2326-6066.CIR-13-0171
140. Weishaupt C, Munoz KN, Buzney E, Kupper TS, and Fuhlbrigge RC. T-cell distribution and adhesion receptor expression in metastatic melanoma. Clin Cancer Res. (2007) 13:2549–56. doi: 10.1158/1078-0432.CCR-06-2450
141. Glick AB. The role of TGFβ Signaling in squamous cell cancer: lessons from mouse models. J Skin Cancer. (2012) 2012:249063. doi: 10.1155/2012/249063
142. Klöss S, Chambron N, Gardlowski T, Weil S, Koch J, Esser R, et al. Cetuximab reconstitutes pro-inflammatory cytokine secretions and tumor-infiltrating capabilities of sMICA-inhibited NK cells in HNSCC tumor spheroids. Front Immunol. (2015) 6:543. doi: 10.3389/fimmu.2015.00543
143. Clark RA, Huang SJ, Murphy GF, Mollet IG, Hijnen D, Muthukuru M, et al. Human squamous cell carcinomas evade the immune response by down-regulation of vascular E-selectin and recruitment of regulatory T cells. J Exp Med. (2008) 205:2221–34. doi: 10.1084/jem.20071190
144. Huang Y, Mao K, Chen X, Sun MA, Kawabe T, Li W, et al. S1P-dependent interorgan trafficking of group 2 innate lymphoid cells supports host defense. Science. (2018) 359:114–9. doi: 10.1126/science.aam5809
145. van der Ploeg EK, Golebski K, van Nimwegen M, Fergusson JR, Heesters BA, Martinez-Gonzalez I, et al. Steroid-resistant human inflammatory ILC2s are marked by CD45RO and elevated in type 2 respiratory diseases. Sci Immunol. (2021) 6:eabd3489. doi: 10.1126/sciimmunol.abd3489
146. Liang Z, Tang Z, Zhu C, Li F, Chen S, Han X, et al. Intestinal CXCR6+ ILC3s migrate to the kidney and exacerbate renal fibrosis via IL-23 receptor signaling enhanced by PD-1 expression. Immunity. (2024) 57:1306–1323.e8. doi: 10.1016/j.immuni.2024.05.004
147. Gasteiger G, Fan X, Dikiy S, Lee SY, and Rudensky AY. Tissue residency of innate lymphoid cells in lymphoid and nonlymphoid organs. Science. (2015) 350:981–5. doi: 10.1126/science.aac9593
148. Doisne JM, Balmas E, Boulenouar S, Gaynor LM, Kieckbusch J, Gardner L, et al. Composition, development, and function of uterine innate lymphoid cells. J Immunol. (2015) 195:3937–45. doi: 10.4049/jimmunol.1500689
149. Filipovic I, Chiossone L, Vacca P, Hamilton RS, Ingegnere T, Doisne JM, et al. Molecular definition of group 1 innate lymphoid cells in the mouse uterus. Nat Commun. (2018) 9:4492. doi: 10.1038/s41467-018-06918-3
150. Sparano C, Solís-Sayago D, Zangger NS, Rindlisbacher L, Van Hove H, Vermeer M, et al. Autocrine TGF-β1 drives tissue-specific differentiation and function of resident NK cells. J Exp Med. (2025) 222:e20240930. doi: 10.1084/jem.20240930
151. Schuster IS, Sng XYX, Lau CM, Powell DR, Weizman OE, Fleming P, et al. Infection induces tissue-resident memory NK cells that safeguard tissue health. Immunity. (2023) 56:531–546.e6. doi: 10.1016/j.immuni.2023.01.016
152. Rodenberg RR, Spadafora D, Fitzpatrick S, Daly G, Lausch R, and Barrington RA. γδ T17 cells regulate the acute antiviral response of NK cells in HSV-1-infected corneas. Invest Ophthalmol Vis Sci. (2024) 65:16. doi: 10.1167/iovs.65.13.16
153. Biron CA, Byron KS, and Sullivan JL. Severe herpesvirus infections in an adolescent without natural killer cells. N Engl J Med. (1989) 320:1731–5. doi: 10.1056/NEJM198906293202605
154. Campbell TM, McSharry BP, Steain M, Ashhurst TM, Slobedman B, and Abendroth A. Varicella zoster virus productively infects human natural killer cells and manipulates phenotype. PloS Pathog. (2018) 14:e1006999. doi: 10.1371/journal.ppat.1006999
155. de Vries E, Koene HR, Vossen JM, Gratama JW, von dem Borne AE, Waaijer JL, et al. Identification of an unusual Fc gamma receptor IIIa (CD16) on natural killer cells in a patient with recurrent infections. Blood. (1996) 88:3022–7. doi: 10.1182/blood.V88.8.3022.bloodjournal8883022
156. Sun JC, Beilke JN, and Lanier LL. Adaptive immune features of natural killer cells. Nature. (2009) 457:557–61. doi: 10.1038/nature07665
157. Cohen JI, Dropulic L, Hsu AP, Zerbe CS, Krogmann T, Dowdell K, et al. Association of GATA2 deficiency with severe primary epstein-barr virus (EBV) infection and EBV-associated cancers. Clin Infect Dis. (2016) 63:41–7. doi: 10.1093/cid/ciw160
158. Yang F, Yuan C, Chen F, Qin ZS, Schmitt NC, Lesinski GB, et al. Combined IL6 and CCR2 blockade potentiates antitumor activity of NK cells in HPV-negative head and neck cancer. J Exp Clin Cancer Res. (2024) 43:76. doi: 10.1186/s13046-024-03002-1
159. Dancy E, Stratton P, Pichard DC, Marciano BE, Cowen EW, McBride AA, et al. Human papillomavirus disease in GATA2 deficiency: a genetic predisposition to HPV-associated female anogenital Malignancy. Front Immunol. (2024) 15:1445711. doi: 10.3389/fimmu.2024.1445711
160. Mizesko MC, Banerjee PP, Monaco-Shawver L, Mace EM, Bernal WE, Sawalle-Belohradsky J, et al. Defective actin accumulation impairs human natural killer cell function in patients with dedicator of cytokinesis 8 deficiency. J Allergy Clin Immunol. (2013) 131:840–8. doi: 10.1016/j.jaci.2012.12.1568
161. Gil-Krzewska A, Wood SM, Murakami Y, Nguyen V, Chiang SCC, Cullinane AR, et al. Chediak-Higashi syndrome: Lysosomal trafficking regulator domains regulate exocytosis of lytic granules but not cytokine secretion by natural killer cells. J Allergy Clin Immunol. (2016) 137:1165–77. doi: 10.1016/j.jaci.2015.08.039
162. Orange JS, Ramesh N, Remold-O’Donnell E, Sasahara Y, Koopman L, Byrne M, et al. Wiskott–Aldrich syndrome protein is required for NK cell cytotoxicity and colocalizes with actin to NK cell-activating immunologic synapses. Proc Natl Acad Sci. (2002) 99:11351–6. doi: 10.1073/pnas.162376099
163. Lanzi G, Moratto D, Vairo D, Masneri S, Delmonte O, Paganini T, et al. A novel primary human immunodeficiency due to deficiency in the WASP-interacting protein WIP. J Exp Med. (2012) 209:29–34. doi: 10.1084/jem.20110896
164. Risma KA, Frayer RW, Filipovich AH, and Sumegi J. Aberrant maturation of mutant perforin underlies the clinical diversity of hemophagocytic lymphohistiocytosis. J Clin Investig. (2006) 116:182–92. doi: 10.1172/JCI26217
165. Feldmann J, Callebaut I, Raposo G, Certain S, Bacq D, Dumont C, et al. Munc13–4 is essential for cytolytic granules fusion and is mutated in a form of familial hemophagocytic lymphohistiocytosis (FHL3). Cell. (2003) 115:461–73. doi: 10.1016/s0092-8674(03)00855-9
166. Bryceson YT, Rudd E, Zheng C, Edner J, Ma D, Wood SM, et al. Defective cytotoxic lymphocyte degranulation in syntaxin-11 deficient familial hemophagocytic lymphohistiocytosis 4 (FHL4) patients. Blood. (2007) 110:1906–15. doi: 10.1182/blood-2007-02-074468
167. Badolato R and Parolini S. Novel insights from adaptor protein 3 complex deficiency. J Allergy Clin Immunol. (2007) 120:735–41; quiz 742-3. doi: 10.1016/j.jaci.2007.08.039
168. Badolato R, Prandini A, Caracciolo S, Colombo F, Tabellini G, Giacomelli M, et al. Exome sequencing reveals a pallidin mutation in a Hermansky-Pudlak-like primary immunodeficiency syndrome. Blood. (2012) 119:3185–7. doi: 10.1182/blood-2012-01-404350
169. Habu S, Akamatsu K, Tamaoki N, and Okumura K. In vivo significance of NK cell on resistance against virus (HSV-1) infections in mice. J Immunol. (1984) 133:2743–7. doi: 10.4049/jimmunol.133.5.2743
170. Ching C and Lopez C. Natural killing of herpes simplex virus type 1-infected target cells: normal human responses and influence of antiviral antibody. Infect Immun. (1979) 26:49–56. doi: 10.1128/iai.26.1.49-56.1979
171. Petersen JL, Morris CR, and Solheim JC. Virus evasion of MHC class I molecule presentation. J Immunol. (2003) 171:4473–8. doi: 10.4049/jimmunol.171.9.4473
172. Bonavida B, Katz J, and Gottlieb M. Mechanism of defective NK cell activity in patients with acquired immunodeficiency syndrome (AIDS) and AIDS-related complex. I. Defective trigger on NK cells for NKCF production by target cells, and partial restoration by IL 2. J Immunol. (1986) 137:1157–63. doi: 10.4049/jimmunol.137.4.1157
173. Katz JD, Mitsuyasu R, Gottlieb MS, Lebow LT, and Bonavida B. Mechanism of defective NK cell activity in patients with acquired immunodeficiency syndrome (AIDS) and AIDS-related complex. II. Normal antibody-dependent cellular cytotoxicity (ADCC) mediated by effector cells defective in natural killer (NK) cytotoxicity. J Immunol. (1987) 139:55–60. doi: 10.4049/jimmunol.139.1.55
174. Kobayashi T, Ricardo-Gonzalez RR, and Moro K. Skin-resident innate lymphoid cells - cutaneous innate guardians and regulators. Trends Immunol. (2020) 41:100–12. doi: 10.1016/j.it.2019.12.004
175. Eberl G, Di Santo JP, and Vivier E. The brave new world of innate lymphoid cells. Nat Immunol. (2015) 16:1–5. doi: 10.1038/ni.3059
176. Bernink JH, Ohne Y, Teunissen MBM, Wang J, Wu J, Krabbendam L, et al. c-Kit-positive ILC2s exhibit an ILC3-like signature that may contribute to IL-17-mediated pathologies. Nat Immunol. (2019) 20:992–1003. doi: 10.1038/s41590-019-0423-0
177. Batista MD, Ho EL, Kuebler PJ, Milush JM, Lanier LL, Kallas EG, et al. Skewed distribution of natural killer cells in psoriasis skin lesions. Exp Dermatol. (2013) 22:64–6. doi: 10.1111/exd.12060
178. Roediger B, Kyle R, Yip KH, Sumaria N, Guy TV, Kim BS, et al. Cutaneous immunosurveillance and regulation of inflammation by group 2 innate lymphoid cells. Nat Immunol. (2013) 14:564–73. doi: 10.1038/ni.2584
179. Li Z, Hodgkinson T, Gothard EJ, Boroumand S, Lamb R, Cummins I, et al. Epidermal Notch1 recruits RORγ(+) group 3 innate lymphoid cells to orchestrate normal skin repair. Nat Commun. (2016) 7:11394. doi: 10.1038/ncomms11394
180. Kobayashi T, Voisin B, Kim DY, Kennedy EA, Jo JH, Shih HY, et al. Homeostatic control of sebaceous glands by innate lymphoid cells regulates commensal bacteria equilibrium. Cell. (2019) 176:982–997.e16. doi: 10.1016/j.cell.2018.12.031
181. Liu S, Sirohi K, Verma M, McKay J, Michalec L, Sripada A, et al. Optimal identification of human conventional and nonconventional (CRTH2-IL7Rα-) ILC2s using additional surface markers. J Allergy Clin Immunol. (2020) 146:390–405. doi: 10.1016/j.jaci.2020.01.038
182. Pelly VS, Kannan Y, Coomes SM, Entwistle LJ, Rückerl D, Seddon B, et al. IL-4-producing ILC2s are required for the differentiation of TH2 cells following Heligmosomoides polygyrus infection. Mucosal Immunol. (2016) 9:1407–17. doi: 10.1038/mi.2016.4
183. Neill DR, Wong SH, Bellosi A, Flynn RJ, Daly M, Langford TK, et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature. (2010) 464:1367–70. doi: 10.1038/nature08900
184. Licona-Limón P, Henao-Mejia J, Temann AU, Gagliani N, Licona-Limón I, Ishigame H, et al. Th9 cells drive host immunity against gastrointestinal worm infection. Immunity. (2013) 39:744–57. doi: 10.1016/j.immuni.2013.07.020
185. Imai Y, Yasuda K, Sakaguchi Y, Haneda T, Mizutani H, Yoshimoto T, et al. Skin-specific expression of IL-33 activates group 2 innate lymphoid cells and elicits atopic dermatitis-like inflammation in mice. Proc Natl Acad Sci U S A. (2013) 110:13921–6. doi: 10.1073/pnas.1307321110
186. Zhang K, Xu X, Pasha MA, Siebel CW, Costello A, Haczku A, et al. Cutting edge: notch signaling promotes the plasticity of group-2 innate lymphoid cells. J Immunol. (2017) 198:1798–803. doi: 10.4049/jimmunol.1601421
187. Rafei-Shamsabadi DA, van de Poel S, Dorn B, Kunz S, Martin SF, Klose CSN, et al. Lack of type 2 innate lymphoid cells promotes a type I-driven enhanced immune response in contact hypersensitivity. J Invest Dermatol. (2018) 138:1962–72. doi: 10.1016/j.jid.2018.03.001
188. Rouzaire P, Luci C, Blasco E, Bienvenu J, Walzer T, Nicolas JF, et al. Natural killer cells and T cells induce different types of skin reactions during recall responses to haptens. Eur J Immunol. (2012) 42:80–8. doi: 10.1002/eji.201141820
189. Zhou S, Li Q, Wu H, and Lu Q. The pathogenic role of innate lymphoid cells in autoimmune-related and inflammatory skin diseases. Cell Mol Immunol. (2020) 17:335–46. doi: 10.1038/s41423-020-0399-6
190. Clottu AS, Humbel M, Fluder N, Karampetsou MP, and Comte D. Innate lymphoid cells in autoimmune diseases. Front Immunol. (2022) 12:789788. doi: 10.3389/fimmu.2021.789788
191. Kucuksezer UC, Aktas Cetin E, Esen F, Tahrali I, Akdeniz N, Gelmez MY, et al. The role of natural killer cells in autoimmune diseases. Front Immunol. (2021) 12:622306. doi: 10.3389/fimmu.2021.622306
192. Petrasca A, Hambly R, Molloy O, Kearns S, Moran B, Smith CM, et al. Innate lymphoid cell (ILC) subsets are enriched in the skin of patients with hidradenitis suppurativa. PloS One. (2023) 18:e0281688. doi: 10.1371/journal.pone.0281688
193. Hou M and Liu S. Innate lymphoid cells are increased in systemic lupus erythematosus. Clin Exp Rheumatol. (2019) 37:676–9.
194. Blokland SLM, van den Hoogen LL, Leijten EFA, Hartgring SAY, Fritsch R, Kruize AA, et al. Increased expression of Fas on group 2 and 3 innate lymphoid cells is associated with an interferon signature in systemic lupus erythematosus and Sjögren's syndrome. Rheumatol (Oxford). (2019) 58:1740–5. doi: 10.1093/rheumatology/kez116
195. Thangjam N, Dey B, Khonglah Y, Tiewsoh I, and Ksoo R. Natural killer cell count in systemic lupus erythematosus patients: A flow cytometry-based study. Cureus. (2023) 15:e46885. doi: 10.7759/cureus.46885
196. Ytterberg SR and Schnitzer TJ. Inhibition of natural killer cell activity by serum from patients with systemic lupus erythematosus: roles of disease activity and serum interferon. Ann Rheum Dis. (1984) 43:457–61. doi: 10.1136/ard.43.3.457
197. Gras Navarro A, Björklund AT, and CheKenya M. Therapeutic potential and challenges of natural killer cells in treatment of solid tumors. Front Immunol. (2015) 6:202. doi: 10.3389/fimmu.2015.00202
198. Takanami I, Takeuchi K, and Giga M. The prognostic value of natural killer cell infiltration in resected pulmonary adenocarcinoma. J Thorac Cardiovasc Surg. (2001) 121:1058–63. doi: 10.1067/mtc.2001.113026
199. Levi I, Amsalem H, Nissan A, Darash-Yahana M, Peretz T, Mandelboim O, et al. Characterization of tumor infiltrating natural killer cell subset. Oncotarget. (2015) 6:13835–43. doi: 10.18632/oncotarget.3453
200. de Jonge K, Ebering A, Nassiri S, Maby-El Hajjami H, Ouertatani-Sakouhi H, Baumgaertner P, et al. Circulating CD56bright NK cells inversely correlate with survival of melanoma patients. Sci Rep. (2019) 9:4487. doi: 10.1038/s41598-019-40933-8
201. Ali TH, Pisanti S, Ciaglia E, Mortarini R, Anichini A, Garofalo C, et al. Enrichment of CD56(dim)KIR + CD57 + highly cytotoxic NK cells in tumour-infiltrated lymph nodes of melanoma patients. Nat Commun. (2014) 5:5639. doi: 10.1038/ncomms6639
202. Messaoudene M, Fregni G, Fourmentraux-Neves E, Chanal J, Maubec E, Mazouz-Dorval S, et al. Mature cytotoxic CD56(bright)/CD16(+) natural killer cells can infiltrate lymph nodes adjacent to metastatic melanoma. Cancer Res. (2014) 74:81–92. doi: 10.1158/0008-5472.CAN-13-1303
203. Lakshmikanth T, Burke S, Ali TH, Kimpfler S, Ursini F, Ruggeri L, et al. NCRs and DNAM-1 mediate NK cell recognition and lysis of human and mouse melanoma cell lines in vitro and in vivo. J Clin Invest. (2009) 119:1251–63. doi: 10.1172/JCI36022
204. Pietra G, Manzini C, Vitale M, Balsamo M, Ognio E, Boitano M, et al. Natural killer cells kill human melanoma cells with characteristics of cancer stem cells. Int Immunol. (2009) 21:793–801. doi: 10.1093/intimm/dxp047
205. Adhikary G, Heipertz EL, Preradovic M, Chen X, Xu W, Newland JJ, et al. Natural killer cells suppress human cutaneous squamous cell carcinoma cancer cell survival and tumor growth. Mol Carcinog. (2023) 62:845–54. doi: 10.1002/mc.23528
206. Cursons J, Souza-Fonseca-Guimaraes F, Foroutan M, Anderson A, Hollande F, Hediyeh-Zadeh S, et al. A gene signature predicting natural killer cell infiltration and improved survival in melanoma patients. Cancer Immunol Res. (2019) 7:1162–74. doi: 10.1158/2326-6066.CIR-18-0500
207. Chang ST, Liao YL, Lin SH, and Chuang SS. NK-cell lymphoma with nodal presentation and expression of cutaneous lymphocyte-associated antigen. Pathol Res Pract. (2010) 206:463–6. doi: 10.1016/j.prp.2009.07.001
208. Magro CM and Dyrsen ME. Cutaneous lymphocyte antigen expression in benign and neoplastic cutaneous B- and T-cell lymphoid infiltrates. J Cutan Pathol. (2008) 35:1040–9. doi: 10.1111/j.1600-0560.2007.00971.x
209. Miller JS, Soignier Y, Panoskaltsis-Mortari A, McNearney SA, Yun GH, Fautsch SK, et al. Successful adoptive transfer and in vivo expansion of human haploidentical NK cells in patients with cancer. Blood. (2005) 105:3051–7. doi: 10.1182/blood-2004-07-2974
210. Savani BN, Mielke S, Adams S, Uribe M, Rezvani K, Yong AS, et al. Rapid natural killer cell recovery determines outcome after T-cell-depleted HLA-identical stem cell transplantation in patients with myeloid leukemias but not with acute lymphoblastic leukemia. Leukemia. (2007) 21:2145–52. doi: 10.1038/sj.leu.2404892
211. Lee DA, Denman CJ, Rondon G, Woodworth G, Chen J, Fisher T, et al. Haploidentical natural killer cells infused before allogeneic stem cell transplantation for myeloid Malignancies: A phase I trial. Biol Blood Marrow Transplant. (2016) 22:1290–8. doi: 10.1016/j.bbmt.2016.04.009
212. Ciurea SO, Schafer JR, Bassett R, Denman CJ, Cao K, Willis D, et al. Phase 1 clinical trial using mbIL21 ex vivo-expanded donor-derived NK cells after haploidentical transplantation. Blood. (2017) 130:1857–68. doi: 10.1182/blood-2017-05-785659
213. Ciurea SO, Kongtim P, Soebbing D, Trikha P, Behbehani G, Rondon G, et al. Decrease post-transplant relapse using donor-derived expanded NK-cells. Leukemia. (2022) 36:155–64. doi: 10.1038/s41375-021-01349-4
214. Ciurea SO, Kongtim P, Srour S, Chen J, Soebbing D, Shpall E, et al. Results of a phase I trial with Haploidentical mbIL-21 ex vivo expanded NK cells for patients with multiply relapsed and refractory AML. Am J Hematol. (2024) 99:890–9. doi: 10.1002/ajh.27281
215. Rubnitz JE, Inaba H, Ribeiro RC, Pounds S, Rooney B, Bell T, et al. NKAML: a pilot study to determine the safety and feasibility of haploidentical natural killer cell transplantation in childhood acute myeloid leukemia. J Clin Oncol. (2010) 28:955–9. doi: 10.1200/JCO.2009.24.4590
216. Shapiro RM, Birch GC, Hu G, Vergara Cadavid J, Nikiforow S, Baginska J, et al. Expansion, persistence, and efficacy of donor memory-like NK cells infused for posttransplant relapse. J Clin Invest. (2022) 132:e154334. doi: 10.1172/JCI154334
217. Shapiro RM, Sheffer M, Booker MA, Tolstorukov MY, Birch GC, Sade-Feldman M, et al. First-in-human evaluation of memory-like NK cells with an IL-15 super-agonist and CTLA-4 blockade in advanced head and neck cancer. J Hematol Oncol. (2025) 18:17. doi: 10.1186/s13045-025-01669-3
218. Romee R, Rosario M, Berrien-Elliott MM, Wagner JA, Jewell BA, Schappe T, et al. Cytokine-induced memory-like natural killer cells exhibit enhanced responses against myeloid leukemia. Sci Transl Med. (2016) 8:357ra123. doi: 10.1126/scitranslmed.aaf2341
219. Berrien-Elliott MM, Foltz JA, Russler-Germain DA, Neal CC, Tran J, Gang M, et al. Hematopoietic cell transplantation donor-derived memory-like NK cells functionally persist after transfer into patients with leukemia. Sci Transl Med. (2022) 14:eabm1375. doi: 10.1126/scitranslmed.abm1375
220. Dean I, Lee CYC, Tuong ZK, Li Z, Tibbitt CA, Willis C, et al. Rapid functional impairment of natural killer cells following tumor entry limits anti-tumor immunity. Nat Commun. (2024) 15:683. doi: 10.1038/s41467-024-44789-z
221. Balsamo M, Scordamaglia F, Pietra G, Manzini C, Cantoni C, Boitano M, et al. Melanoma-associated fibroblasts modulate NK cell phenotype and antitumor cytotoxicity. Proc Natl Acad Sci U S A. (2009) 106:20847–52. doi: 10.1073/pnas.0906481106
222. Pietra G, Manzini C, Rivara S, Vitale M, Cantoni C, Petretto A, et al. Melanoma cells inhibit natural killer cell function by modulating the expression of activating receptors and cytolytic activity. Cancer Res. (2012) 72:1407–15. doi: 10.1158/0008-5472.CAN-11-2544
223. Parkhurst MR, Riley JP, Dudley ME, and Rosenberg SA. Adoptive transfer of autologous natural killer cells leads to high levels of circulating natural killer cells but does not mediate tumor regression. Clin Cancer Res. (2011) 17:6287–97. doi: 10.1158/1078-0432.CCR-11-1347
224. Tallerico R, Cristiani CM, Staaf E, Garofalo C, Sottile R, Capone M, et al. IL-15, TIM-3 and NK cells subsets predict responsiveness to anti-CTLA-4 treatment in melanoma patients. Oncoimmunology. (2016) 6:e1261242. doi: 10.1080/2162402X.2016.1261242
225. Tietze JK, Angelova D, Heppt MV, Ruzicka T, and Berking C. Low baseline levels of NK cells may predict a positive response to ipilimumab in melanoma therapy. Exp Dermatol. (2017) 26:622–9. doi: 10.1111/exd.13263
226. Vyas M, Requesens M, Nguyen TH, Peigney D, Azin M, and Demehri S. Natural killer cells suppress cancer metastasis by eliminating circulating cancer cells. Front Immunol. (2023) 13:1098445. doi: 10.3389/fimmu.2022.1098445
227. Sabatini ME and Chiocca S. Human papillomavirus as a driver of head and neck cancers. Br J Cancer. (2020) 122:306–14. doi: 10.1038/s41416-019-0602-7
228. Dong S, Zhao M, Zhu J, Li T, Yan M, Xing K, et al. Natural killer cells: a future star for immunotherapy of head and neck squamous cell carcinoma. Front Immunol. (2024) 15:1442673. doi: 10.3389/fimmu.2024.1442673
229. André P, Denis C, Soulas C, Bourbon-Caillet C, Lopez J, Arnoux T, et al. Anti-NKG2A mAb is a checkpoint inhibitor that promotes anti-tumor immunity by unleashing both T and NK cells. Cell. (2018) 175:1731–1743.e13. doi: 10.1016/j.cell.2018.10.014
230. Migden MR, Khushalani NI, Chang ALS, Lewis KD, Schmults CD, Hernandez-Aya L, et al. Cemiplimab in locally advanced cutaneous squamous cell carcinoma: results from an open-label, phase 2, single-arm trial. Lancet Oncol. (2020) 21:294–305. doi: 10.1016/S1470-2045(19)30728-4
231. Gross ND, Miller DM, Khushalani NI, Divi V, Ruiz ES, Lipson EJ, et al. Neoadjuvant cemiplimab for stage II to IV cutaneous squamous-cell carcinoma. N Engl J Med. (2022) 387:1557–68. doi: 10.1056/NEJMoa2209813
232. Rischin D, Migden MR, Lim AM, Schmults CD, Khushalani NI, Hughes BGM, et al. Phase 2 study of cemiplimab in patients with metastatic cutaneous squamous cell carcinoma: primary analysis of fixed-dosing, long-term outcome of weight-based dosing. J Immunother Cancer. (2020) 8:e000775. doi: 10.1136/jitc-2020-000775
233. Barry KC, Hsu J, Broz ML, Cueto FJ, Binnewies M, Combes AJ, et al. A natural killer-dendritic cell axis defines checkpoint therapy-responsive tumor microenvironments. Nat Med. (2018) 24:1178–91. doi: 10.1038/s41591-018-0085-8
234. Luci C, Bihl F, Bourdely P, Khou S, Popa A, Meghraoui-Kheddar A, et al. Cutaneous squamous cell carcinoma development is associated with a temporal infiltration of ILC1 and NK cells with immune dysfunctions. J Invest Dermatol. (2021) 141:2369–79. doi: 10.1016/j.jid.2021.03.018
235. Ramirez H, Patel SB, and Pastar I. The role of TGFβ Signaling in wound epithelialization. Adv Wound Care (New Rochelle). (2014) 3:482–91. doi: 10.1089/wound.2013.0466
236. Castriconi R, Cantoni C, Della Chiesa M, Vitale M, Marcenaro E, Conte R, et al. Transforming growth factor beta 1 inhibits expression of NKp30 and NKG2D receptors: consequences for the NK-mediated killing of dendritic cells. Proc Natl Acad Sci U S A. (2003) 100:4120–5. doi: 10.1073/pnas.0730640100
237. Cortez VS, Ulland TK, Cervantes-Barragan L, Bando JK, Robinette ML, Wang Q, et al. SMAD4 impedes the conversion of NK cells into ILC1-like cells by curtailing non-canonical TGF-β signaling. Nat Immunol. (2017) 18:995–1003. doi: 10.1038/ni.3809
238. Viel S, Marçais A, Guimaraes FS, Loftus R, Rabilloud J, Grau M, et al. TGF-β inhibits the activation and functions of NK cells by repressing the mTOR pathway. Sci Signal. (2016) 9:ra19. doi: 10.1126/scisignal.aad1884
239. Denman CJ, Senyukov V, Somanchi SS, Phatarpekar PV, Kopp LM, Johnson JL, et al. Membrane-bound IL-21 promotes sustained ex vivo proliferation of human natural killer cells. PloS One. (2012) 7:e30264–4. doi: 10.1371/journal.pone.0030264
240. Somanchi SS, Senyukov VV, Denman CJ, and Lee DA. Expansion, purification, and functional assessment of human peripheral blood NK cells. J Vis Exp. (2011) 48):2540. doi: 10.3791/2540
241. Foltz JA, Moseman JE, Thakkar A, Chakravarti N, and Lee DA. TGFβ Imprinting during activation promotes natural killer cell cytokine hypersecretion. Cancers (Basel). (2018) 10:423. doi: 10.3390/cancers10110423
242. Gottschalk LR, Bray RA, Kaizer H, and Gebel HM. Two populations of CD56 (Leu-19)+/CD16+ cells in bone marrow transplant recipients. Bone Marrow Transplant. (1990) 5:259–64.
243. Cooper MA, Fehniger TA, and Caligiuri MA. The biology of human natural killer-cell subsets. Trends Immunol. (2001) 22:633–40. doi: 10.1016/s1471-4906(01)02060-9
244. Duhen T, Geiger R, Jarrossay D, Lanzavecchia A, and Sallusto F. Production of interleukin 22 but not interleukin 17 by a subset of human skin-homing memory T cells. Nat Immunol. (2009) 10:857–63. doi: 10.1038/ni.1767
245. Dunphy S and Gardiner CM. NK cells and psoriasis. J BioMed Biotechnol. (2011) 2011:248317. doi: 10.1155/2011/248317
246. Sato Y, Ogawa E, and Okuyama R. Role of innate immune cells in psoriasis. Int J Mol Sci. (2020) 21:6604. doi: 10.3390/ijms21186604
247. Park E, Patel S, Wang Q, Andhey P, Zaitsev K, Porter S, et al. Toxoplasma gondii infection drives conversion of NK cells into ILC1-like cells. Elife. (2019) 8:e47605. doi: 10.7554/eLife.47605
248. Kim BS, Siracusa MC, Saenz SA, Noti M, Monticelli LA, Sonnenberg GF, et al. TSLP elicits IL-33-independent innate lymphoid cell responses to promote skin inflammation. Sci Transl Med. (2013) 5:170ra16. doi: 10.1126/scitranslmed.3005374
249. Manilay JO and Sykes M. Natural killer cells and their role in graft rejection. Curr Opin Immunol. (1998) 10:532–8. doi: 10.1016/s0952-7915(98)80219-7
250. Olson JA, Zeiser R, Beilhack A, Goldman JJ, and Negrin RS. Tissue-specific homing and expansion of donor NK cells in allogeneic bone marrow transplantation. J Immunol. (2009) 183:3219–28. doi: 10.4049/jimmunol.0804268
251. Shah NN, Baird K, Delbrook CP, Fleisher TA, Kohler ME, Rampertaap S, et al. Acute GVHD in patients receiving IL-15/4-1BBL activated NK cells following T-cell-depleted stem cell transplantation. Blood. (2015) 125:784–92. doi: 10.1182/blood-2014-07-592881
252. Xu Y, Gan Y, Qi F, Lu X, Zhang X, Zhang J, et al. Innate lymphoid cell-based immunomodulatory hydrogel microspheres containing Cutibacterium acnes extracellular vesicles for the treatment of psoriasis. Acta Biomater. (2024) 184:296–312. doi: 10.1016/j.actbio.2024.06.006
Keywords: natural killer cells, innate lymphoid cells, cutaneous disease, skin, innate immunity
Citation: Johnson KM and Lee DA (2025) Natural killer cells in skin: a unique opportunity to better characterize the many facets of an overlooked secondary lymphoid organ. Front. Immunol. 16:1646719. doi: 10.3389/fimmu.2025.1646719
Received: 13 June 2025; Accepted: 15 July 2025;
Published: 12 August 2025.
Edited by:
Laurent Brossay, Brown University, United StatesReviewed by:
Negre Olivier, Smart Immune, FranceTommaso Torcellan, Translationale Onkologie an der Universitätsmedizin der Johannes Gutenberg-Universität Mainz, Germany
Copyright © 2025 Johnson and Lee. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Kirsten M. Johnson, a2lyc3Rlbi5qb2huc29uQG9zdW1jLmVkdQ==
†ORCID: Kirsten M. Johnson, orcid.org/0000-0003-2388-0621
Dean A. Lee, orcid.org/0000-0001-6693-5392
 Kirsten M. Johnson
Kirsten M. Johnson Dean A. Lee
Dean A. Lee