- Department of Respiratory & Critical Care Medicine, Hospital 2, Jilin University, Changchun, China
Asthma is a heterogeneous disease characterized by chronic airway inflammation, heightened reactivity, and structural remodeling. The responses of different phenotypes to traditional corticosteroid therapy vary significantly, with steroid resistance in low T-helper type 2 asthma remaining an urgent clinical challenge. In recent years, mesenchymal stem cells (MSCs) and their exosomes—mesenchymal stem cell-derived extracellular vesicles (MSC-EVs)—have emerged as promising therapeutic agents due to their potent immunomodulatory properties. In this review, we systematically explain how MSCs and MSC-EVs inhibit airway inflammation in asthma through multi-target immunoregulation, highlight their therapeutic potential in steroid-resistant asthma, and outline the challenges and optimization strategies involved in clinical translation, thereby providing a theoretical foundation for the development of novel therapies.
1 Introduction
Asthma is a common respiratory disorder characterized by chronic airway inflammation, heightened airway responsiveness, and structural remodeling (1–5). Its pathophysiology involves numerous inflammatory cells and mediators (6–10). Based on inflammatory cell profiles in induced sputum, asthma can be classified into four types: eosinophilic asthma, neutrophilic asthma, mixed granulocytic asthma and paucigranulocytic asthma (11). T-helper type 2 (Th2)-type asthma is characterized by activation of type 2 inflammatory pathways and is characterized by eosinophilic inflammation (12). In contrast, non-Th2 asthma lacks markers of type 2 inflammation and is characterized by neutrophilic, paucigranulocytic, or mixed inflammation (12). Corticosteroids are essential in asthma management; however, low Th2 asthma often exhibits steroid resistance and remains difficult to treat (13–17). Managing low Th2 asthma remains a significant clinical challenge in respiratory medicine. Several new biologic therapies have recently emerged. Anti-immunoglobulin E (anti-IgE) monoclonal antibodies (mAbs), such as omalizumab, reduce exacerbations by lowering free IgE levels and may delay disease progression (18). Anti-interleukin-5/interleukin-5 receptor alpha (anti-IL-5/IL-5Rα) agents, such as mepolizumab and benralizumab, effectively eliminate eosinophils, reduce acute exacerbation rates, and improve lung function (19, 20). IL-4Rα inhibitors, such as dupilumab, significantly improve mucus secretion and airway remodeling by blocking IL-4/IL-13 signaling. These are suitable for moderate to severe asthma (21). Anti-thymic stromal lymphopoietin mAbs, such as tezepelumab, inhibit upstream alarmin pathways and demonstrate efficacy across various asthma phenotypes (22). However, challenges remain in the clinical application of these drugs. Certain asthma subtypes—such as neutrophilic and mixed granulocytic asthma—respond poorly to treatment (23–25). Other concerns include disease rebound after discontinuation, complications associated with long-term use, and high treatment costs (26–29). Recent studies have found that mesenchymal stem cells (MSCs) have significant therapeutic effects in asthma, offering new avenues for treatment development.
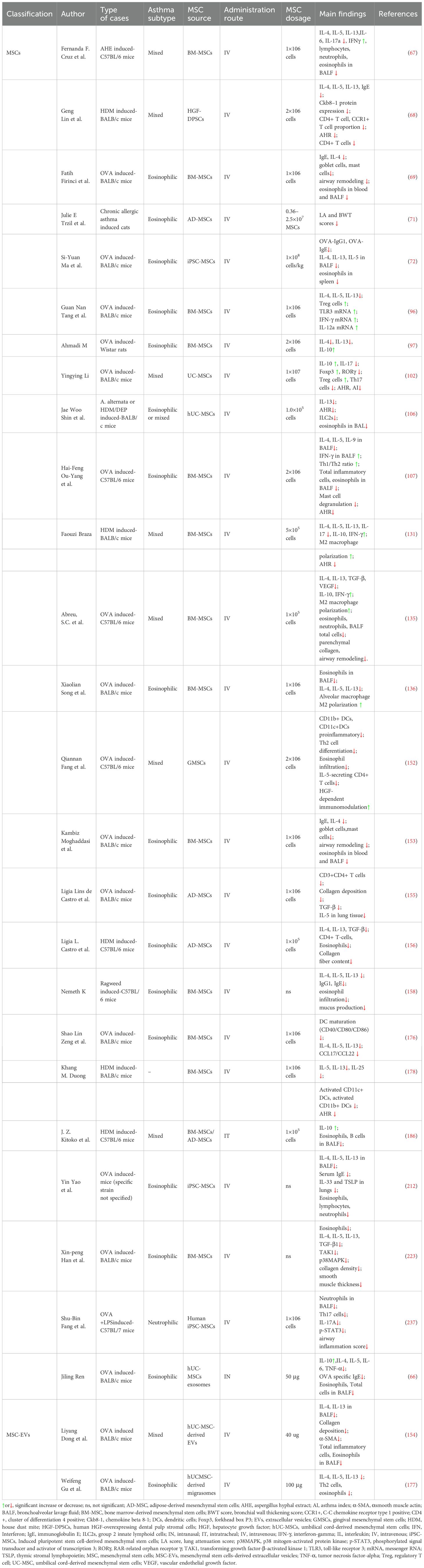
MSCs are adult stem cells capable of self-renewal and multi-lineage differentiation (30–33). First identified in 1976, MSCs have since been found in nearly all human tissues, including bone marrow-derived MSCs (BM-MSCs), adipose-derived MSCs, and umbilical cord blood-derived MSCs (UC-MSCs) (34–38) (Figure 1). Originating from the mesoderm, MSCs exhibit a strong capacity for self-renewal and diverse differentiation potential (39–41). Extensive studies have demonstrated that MSCs possess potent immunomodulatory properties and can migrate to sites of inflammation and tumors (42). MSCs have shown remarkable therapeutic benefits for numerous diseases due to their ability to differentiate into various cell types (43–47). In vitro-expanded MSCs have been applied in treating a range of immune and inflammatory conditions, including autoimmune diseases and organ failure, such as Crohn’s disease of the bowel and graft-versus-host disease (38).
Figure 1. The overview of multipotentiality and multi-lineage differentiation of MSCs. MSC, mesenchymal stem cells.
Recently, the potential role of MSCs in asthma has attracted increasing interest from researchers (48). However, the clinical application of MSCs faces several challenges, including the need for large cell doses, variability in cell quality, low post-transplantation survival rates, unintended effects following intravenous injection, and limited ability to cross physiological barriers such as the blood–brain barrier (49–53). Therefore, increasing attention has been directed toward extracellular vesicles (EVs) derived from MSCs and their ability to modulate normal and pathological cellular processes, including inflammation and tissue injury responses (54). MSC-derived extracellular vesicles (MSC-EVs) are lipid-based vesicles secreted by MSCs and include exosomes, microvesicles, and apoptotic bodies (55–57). Exosomes are formed through the endosomal-multivesicular body pathway, microvesicles are generated via direct budding from the plasma membrane resulting from localized membrane remodeling and shedding, while apoptotic bodies are produced through programmed cell apoptosis (58–60). Several studies have investigated the mechanisms by which MSC-EVs influence immune cell function (54, 61, 62). These vesicles carry a wide array of signaling molecules from their parent cells, such as proteins, lipids, surface receptors, enzymes, cytokines, metabolites, and nucleic acids (63, 64). By modulating innate and adaptive immune responses, MSC-EVs help regulate immunity by suppressing the activity of T cells, B cells, natural killer cells, innate lymphoid cells, macrophages, and dendritic cells (DCs), thereby influencing various asthma phenotypes (65, 66).
2 MSCs/EVs and asthma
MSCs and MSC-EVs exert significant effects on asthma and have emerged as promising therapeutic agents for its treatment (67–69). The therapeutic potential of MSCs in asthma largely stems from their immunomodulatory properties, which help regulate the balance between Th1 and Th2 cytokines, thereby reducing airway inflammation and remodeling (53, 70–72). Numerous studies have confirmed the immunomodulatory effects of MSCs (52, 73, 74). MSCs can reduce T cell proliferation (75, 76), inhibit the differentiation of monocytes into pro-inflammatory macrophages and DCs (77, 78), suppress the cytotoxicity and proliferation of natural killer cells (79), and limit B cell maturation and antibody production (80). These findings suggest that MSCs may influence asthma progression by modulating multiple immune cell types. MSC-EVs are increasingly recognized for their similar immunoregulatory roles and their advantages in transport and application (81). Table 1 categorizes and summarizes asthma animal models using MSCs or MSC-EVs as a therapeutic approach.
3 MSCs and immune and non-immune cells in asthma
MSCs are involved in the pathogenesis of asthma through their effects on a variety of immune and non-immune cells (Figure 2). In this review, we summarize the molecular mechanisms of MSCs as well as the role of MSC-EVs in asthma and their therapeutic potential.
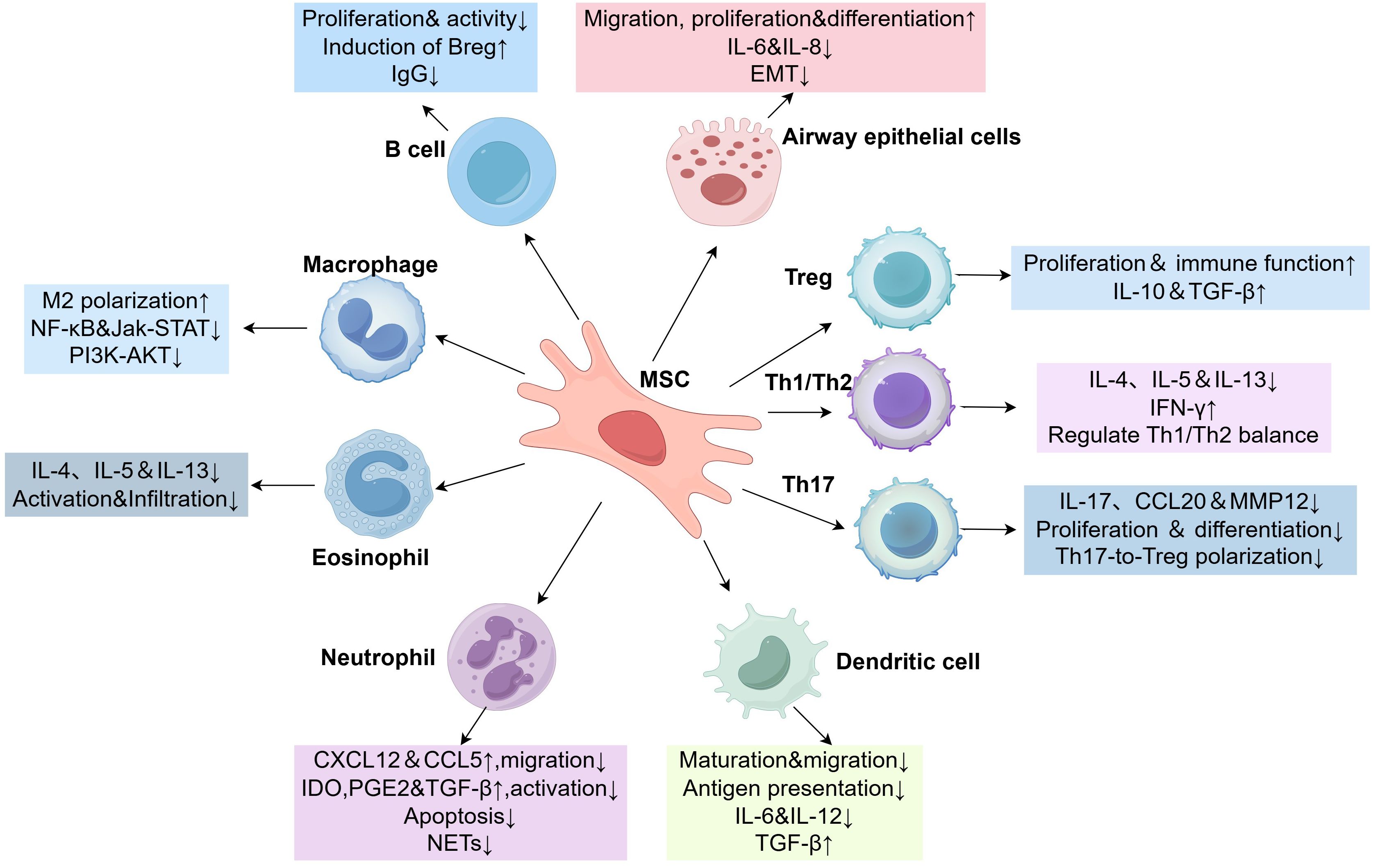
Figure 2. MSCs crosstalk with immune and non-immune cells. ↑or↓, significant increase or decrease; Breg, regulatory B cells; CCL, C-C motif chemokine ligand; CXCL, C-X-C motif chemokine ligand; EMT, epithelial-mesenchymal transition; IL, interleukin; Jak-STAT, janus kinase-signal transducer and activator of transcription; Treg, regulatory T cells; MSC, mesenchymal stem cells; MMP12, matrix metalloproteinase-12; NF-κB, nuclear factor kappa-B; IFN-γ, interferon-γ.
3.1 T cells
T lymphocyte subsets play a key role in the pathogenesis of asthma. The differentiation of various T cell subtypes and the cytokines they secrete are critical in driving different forms of asthma-related inflammation. MSC-EVs exert strong immunomodulatory effects and can regulate T cell metabolism, further influencing distinct asthma phenotypes (75, 82). Th2 cells are major drivers of eosinophilic asthma. They secrete type 2 cytokines such as IL-4, IL-5, and IL-13, which promote eosinophil proliferation and cause airway inflammation and damage (83). In contrast, Th17 cells are implicated in neutrophilic asthma. Cytokines such as IL-17, secreted by Th17 cells, strongly promote the recruitment and activation of neutrophils, resulting in increased neutrophilic infiltration in the airway, the release of inflammatory mediators and proteases, and consequent airway epithelial injury and remodeling (84, 85). MSCs can regulate various T cell states—including immature, naive, mature, effector, and memory T cells—through multiple mechanisms (86). For example, indoleamine 2, 3-dioxygenase (IDO), a rate-limiting enzyme in tryptophan catabolism induced by inflammatory cytokines, plays a role in inhibition of intracellular pathogen replication and immune regulation (87). IDO suppresses T cell proliferation and promotes regulatory T cell (Treg) differentiation by converting tryptophan to kynurenine (88). BM-MSCs have been shown to significantly upregulate IDO expression, inhibit Th17 cell differentiation, and reduce IL-17 secretion in inflammatory environments (88).
3.1.1 Regulatory T cells
Tregs are a subset of T cells with significant immunosuppressive functions, characterized by the expression of forkhead box P3 (FOXP3), CD25, and CD4 (89, 90). Tregs play a critical role in asthma by suppressing excessive immune responses and reducing airway inflammation. Studies have shown that MSC-EVs can promote the differentiation of CD4+, CD25+, and FOXP3+ Tregs (91–93). This effect is mediated by specific microRNAs (miRNAs) contained in the vesicles, including miR-1470 and miR-21-5p. miR-1470 upregulates the expression of cyclin-dependent kinase inhibitor 1B (p27Kip1), while miR-21-5p participates in regulating Treg differentiation and function via the phosphoinositide 3-kinase (PI3K)/AKT pathway (91, 94–97). In addition, MSC-EVs can enhance the expression of miR-146a, a miRNA with significant immunomodulatory activity (98). miR-146a promotes the production of anti-inflammatory cytokines, such as IL-10 and transforming growth factor-beta (TGF-β), thereby enhancing the inhibitory function of Tregs (98). miR-126a also contributes to MSC-EV-mediated promotion of Treg differentiation. Studies have shown that MSCs can convert conventional T cells into Tregs by modulating miR-126a expression (99). miR-126a also plays a role in the stabilization of FOXP3 expression, which is a key transcription factor for Treg development and function (99). MSC-EVs can further upregulate p27Kip1 and down-regulate cyclin-dependent kinase 2 levels, leading to T cell cycle arrest and reduced T cell proliferation—an important immunosuppressive mechanism (100). Studies demonstrated that MSC-EVs modulate cytokine secretion in T cells. In a synovitis model, EV treatment reduced IL-17 levels and increased IL-10 and TGF-β expression (101, 102). It is important to note that MSC sources vary greatly in their ability to induce Tregs. An in vitro study comparing BM-MSCs and UC-MSCs found that UC-MSCs ability to induce Tregs was significantly greater than that of BM-MSCs (91). In terms of treatment, Zhang et al. reported that MSC-EVs improved disease symptoms by inhibiting T cell proliferation and enhancing Treg differentiation, leading to a decrease in proinflammatory cytokines and elevated anti-inflammatory cytokines (103).
3.1.2 Th1/Th2
The imbalance between Th1 and Th2 cells is a key factor in the pathogenesis of eosinophilic asthma. By secreting TGF-β1, MSCs suppress the expression of GATA-binding protein 3 and inhibit Th2 cell proliferation (104, 105). At the same time, some studies have shown that the therapeutic effect of MSCs on severe asthma by inhibiting Th2 cells depends on the route of administration, and the dose and time need to be optimized (106, 107). Cho et al. demonstrated that MSCs reduce Th2-related cytokines, including IL-4, IL-5, and IL-13, while increasing the Th1-related cytokine interferon-gamma (IFN-γ), thereby inhibiting Th2 differentiation and restoring the Th1/Th2 balance (108, 109). Another study found that systemic injection of BM-MSCs during antigen sensitization promoted a Th1 phenotype in antigen-specific CD4 T lymphocytes, suppressing Th2-mediated allergic airway inflammation through an IFN-γ-dependent mechanism (110). Interestingly, Chen et al. reported that MSC-EVs may promote the transformation of Th1 to Th2 cells, contrary to the findings above. However, that study primarily noted that the proportion of Th1 cells was significantly decreased and that of Th2 cells was significantly increased following MSC-EV treatment, without investigating the underlying molecular mechanisms in detail (111). In addition, studies have shown that MSCs can inhibit the phosphorylation of STAT3 by upregulating the expression of protein tyrosine phosphatase non-receptor type 2, thereby reducing the production of IFN-γ and IL-4 by Th1 and Th2 cells, and suppressing their differentiation and proliferation (112, 113). Combined with the findings of Cho et al., the effect of MSCs on IFN-γ appears to be bidirectional—inhibiting its production and signaling and promoting its expression and functional enhancement. The exact mechanism remains unclear and may depend on the specific biological environment and regulatory pathways involved.
3.1.3 Th17
Th17 cells secrete cytokines such as IL-17 and IL-12, promoting inflammatory responses and airway remodeling, and are thought to contribute to glucocorticoid-insensitive asthma subtypes, such as neutrophilic asthma (84, 114, 115). It has been demonstrated that treating rats with experimental autoimmune uveitis using MSCs significantly reduced the production of Th17-related cytokines, such as IL-17, lowered the proportion of Th17 cells among lymphocytes, and inhibited interleukin differentiation and activity (116). Guan et al. suggested that adipose-derived stem cells (ADSCs) directly inhibit Th17 cell proliferation through contact mediated by programmed death-ligand 1 (PD-L1) on their cell surface, and also secrete TGF-β and prostaglandin E2 (PGE2) to suppress Th17 cells (117). RNA sequencing analysis revealed that ADSCs significantly downregulated Th17-associated genes, including IL17A, CCL20, and MMP12 (117). Pathway analysis revealed that ADSC treatment significantly inhibited the activation of IL-17 signaling pathway (117). Other studies reported that gingiva-derived MSC-EVs reduced NF-κB phosphorylation by suppressing IKKB expression via miR-148a-3p, thereby inhibiting the IKKB/NF-κB signaling pathway, reducing IL-17A levels, increasing IL-10 production, and ultimately suppressing Th17 cell proliferation and differentiation while alleviating inflammation (118, 119). MSCs also regulate Th17 cell metabolism through mitochondrial transfer. Studies have shown that tunneling nanotubes and EVs can transfer mitochondria to Th17 cells, leading to metabolic reprogramming. After MSC reprogramming, Th17 cells exhibited increased oxidative phosphorylation and reduced glycolysis through the mTORC1 pathway, which inhibited the proliferation and function of Th17 cells. Moreover, the expression of FOXP3 in Th17 cells was significantly increased following mitochondrial transfer, suggesting a transition toward Treg cell phenotype (120, 121).
3.2 Monocytes and macrophages
In asthma, macrophages are key immune cells involved in phagocytosis, the release of inflammatory mediators, antigen presentation, and the regulation of immune responses (122, 123). Macrophages can be classified into two main phenotypes: classically activated (M1) and alternatively activated (M2) macrophages (124). The balance between M1 and M2 macrophages is particularly important in asthma pathogenesis. M2 macrophages are often associated with the promotion of airway inflammation and tissue remodeling (125). M1 macrophages are activated by Th1 cytokines, such as IFN-γ, and by toll-like receptor (TLR) signaling. Upon activation, M1 macrophages produce proinflammatory cytokines including IL-6 and IL-1β, which promote neutrophilic inflammation (126). In contrast, M2 macrophages are induced by Th2-derived cytokines, particularly IL-4 and IL-13, to produce eosinophil-promoting cytokines such as IL-10 and TGF-β (127). Paucigranulocytic asthma, a milder subtype of asthma, is characterized by a predominance of macrophages; however, its molecular mechanisms remain poorly understood (128, 129). Macrophage polarization induces Th1 and Th2 cytokine production, thereby contributing to the pathogenesis of eosinophilic asthma and neutrophilic asthma (130).
Several studies have shown that BM-MSCs can influence macrophages through different mechanisms, potentially affecting different asthma phenotypes (131). In the pathogenesis of asthma, macrophage polarization plays an important role in modulating the inflammatory response. Numerous studies have demonstrated that MSCs can promote M2 polarization (132–136). MSCs and MSC-EVs can induce the shift from pro-inflammatory M1 macrophages to anti-inflammatory M2 macrophages by secreting anti-inflammatory factors such as tumor necrosis factor-stimulated gene-6 (TSG-6) and IL-10, thereby reducing the inflammatory response (137–139). Furthermore, several studies have demonstrated that miRNAs and proteins carried by MSC-EVs—including miR-21-5p, miR-146a-5p, miR-451, miR-16, and galectin-1—can promote M2 polarization by inhibiting inflammatory pathways involving NF-κB, Jak-STAT, and PI3K-AKT, thus reducing inflammation (138, 140–142). In addition, MSC-derived mitochondria have been found to transfer to macrophages and promote M2 polarization by improving mitochondrial function (143). Li et al. demonstrated that MSCs can inhibit CD4+ T cell proliferation and promote M2 macrophage polarization in a TSG-6-dependent manner, thereby reducing inflammation (144). Through miRNA sequencing and proteomic analysis of mesenchymal stem cell-derived exosomes (MSC-Exos) and MRC-5-derived exosomes, Liu et al. proposed that MSC-Exos may inhibit pyroptosis via miRNA-mediated regulation of the caspase-1 pathway or through proteins with immunomodulatory functions. However, the specific miRNAs and proteins responsible remain unidentified (145).
When M1-type macrophages were co-cultured with MSCs, CD54-specific upregulation and enrichment were detected at the contact interface, and this specific interaction induced calcium signaling and enhanced the CD54-mediated immunosuppressive capacity of MSCs, which may represent an additional mechanism by which MSCs inhibit the inflammatory response (146).
Regarding monocytes, Du Rocher et al. demonstrated that MSCs induced phenotypic changes in monocytes, including downregulation of major histocompatibility complex class I and II, CD11c, and CCR5, and upregulation of CD14 and CD64 expression (147). These changes were accompanied by a reduction in IL-1β and IL-6 production by the monocytes. Overall, MSCs reduce the number of monocyte-derived macrophages and promote the polarization of macrophages from the M1 to the M2 phenotype, thereby alleviating airway inflammation.
3.3 Eosinophils
Eosinophilic asthma is a subtype of asthma characterized by elevated levels of eosinophils in the airway. It is associated with several cytokines, such as IL-4, IL-5, and IgE, and several novel immune agents have also been proposed for this asthma subtype (148–151). In recent years, numerous studies have confirmed that MSCs have significant effects on eosinophils and related cytokines (152, 153). Experiments have shown that human UCMSC-EVs treated with hypoxia can reduce eosinophilic infiltration in allergic airway inflammation and decrease levels of Th2 cytokines such as IL-4, IL-5, and IL-13 (154, 155). Ligia et al. also demonstrated that multiple infusions of fat-derived MSCs can reduce eosinophil-mediated inflammation in asthmatic models through immunosuppression (156). Interestingly, a case report suggested that intravenous MSC infusion significantly improved eosinophilic infiltrating gangrene of the lower extremity in patients with eosinophilic granulomatosis with polyangiitis, inhibiting local eosinophilic degranulation (157). Overall, MSCs can inhibit eosinophilic inflammation, and the specific mechanisms involved have been explored in recent years. For example, gingiva-derived MSCs inhibit eosinophil activation by secreting hepatocyte growth factor (152). BM-MSCs can inhibit eosinophil-related inflammation through TGF-β signaling pathway (158). In addition, cord blood MSCs have been shown to alleviate eosinophilic inflammation in atopic dermatitis and asthma by downregulating IgE and eosinophilic cationic protein levels (159). A study demonstrated that tonsil-derived MSCs may reduce allergic symptoms and eosinophilic infiltration by inhibiting the induction of cytokines such as IL-25 and IL-33, as well as chemokines such as CCL11 and CCL24 (160). Another study confirmed that MSCs can reduce total IgE, IL-5, and eosinophilic inflammatory cells through the expression and transduction of the IL10 gene (161). Notably, MSCs can also indirectly affect eosinophils by influencing other cells. Placenta-derived MSCs can reduce eosinophil-mediated airway hyperreactivity and inflammation in asthmatic models by inhibiting Th17 cell differentiation and promoting Treg cell expansion (102). Although MSCs inhibit eosinophilic inflammation, their specific roles and mechanisms in eosinophilic asthma require further investigation.
3.4 Neutrophils
Neutrophilic asthma is a subtype of asthma characterized by neutrophil-dominant airway inflammation and is closely associated with refractory, severe, and fatal asthma (162). It is often difficult to treat because of resistance to glucocorticoid therapy and the lack of other effective treatments (130). The pathogenesis of neutrophilic asthma involves the recruitment and activation of neutrophils in the airway (163). Neutrophils release a variety of pro-inflammatory mediators, including cytokines, chemokines, and neutrophil extracellular traps (NETs). These mediators contribute to airway inflammation, hyperresponsiveness, and remodeling, which underlie the clinical symptoms of asthma (164–166). By secreting chemokines such as CXCL12 and CCL5, MSCs inhibit the over-recruitment of neutrophils to inflammatory sites, thereby reducing tissue damage (167). Another study demonstrated that intratracheal administration of MSCs reduced CXCL1 expression via secretion of TSG-6, which inhibited neutrophil recruitment (168). Furthermore, MSCs secrete molecules such as IDO, PGE2, and TGF-β, which directly inhibit excessive neutrophil activation, reduce pro-inflammatory cytokine release, and alleviate inflammation (169, 170). BM-MSCs have been shown to inhibit neutrophil apoptosis and reduce reactive oxygen species (ROS) production (171). Moreover, MSC treatment can reduce chemotaxis, ROS production, and NADPH oxidase activity by upregulating CD24 expression, shifting activated neutrophils toward a senescent neutrophil phenotype and thereby suppressing inflammation (172). Another possible mechanism involves NETs. NETs are web-like structures composed of decondensed chromatin and antimicrobial proteins released by neutrophils to trap and kill pathogens (173). MSCs can regulate many neutrophil functions, including migration, ROS production, and NET formation. The direction of these effects depends closely on the specific environment and the source of MSCs (174). Under non-pathological conditions, MSCs enhance neutrophil migration and ROS production, whereas in inflammatory states, MSCs suppress these functions as well as NETosis (174). EVs secreted by MSCs carry miRNAs such as miR-199, which inhibit NET formation and reduce the release of pro-inflammatory mediators, thereby attenuating inflammation (167). Wang et al. demonstrated that MSCs can reduce NET formation by inhibiting the MEK/ERK signaling pathway (175). In addition, the source of MSCs influences their effect on neutrophils: MSCs derived from healthy donors block neutrophil infiltration and NETosis, whereas MSCs derived from patients with cancer enhance these functions (174). Hypoxia-challenged MSC-derived EVs have been shown to reduce excessive NET formation, promoting diabetic wound healing (175). This effect is mediated by the transfer of miR-17-5p, which targets the TLR4/ROS/MAPK pathway (175). Targeting NETs with MSCs may represent a novel therapeutic strategy for asthma.
3.5 Dendritic cells
DCs play an important role in various types of asthma by regulating immune cell activation and the inflammatory response. MSCs can inhibit the maturation, migration, and antigen-presenting functions of DCs through cytokine secretion, EVs, and direct cell contact, thereby exerting therapeutic effects in asthma (176–178). A study by Saeidi et al. suggested that UC-MSCs and BM-MSCs inhibit the differentiation, maturation, and endocytosis of monocyte-derived DCs under in vitro conditions via secreted factors (78). Peng et al. found that MSC-EVs can inhibit the differentiation of human monocytes into DCs and downregulate the expression of CD40, CD80, CD86, and HLA-DR, although they do not affect mature DCs (mDCs) (179). Reis et al. also demonstrated that MSC-EV treatment impaired antigen uptake by immature DCs and prevented their maturation, resulting in decreased expression of CD83, CD38, and CD80, reduced secretion of IL-6 and IL-12p70 and increased TGF-β production. The study also proposed a novel mechanism whereby MSC-EVs regulate DC function by targeting the CCR7 gene for degradation via miRNAs such as miR-21-5p, thereby significantly reducing their migration toward CCL21 (94). Another study showed that MSCs suppressed DC function by inhibiting IL-10/STAT3-mediated CST7 gene transcription (180). Another study suggested that MSCs may regulate the MAPK signaling pathway in DCs, secrete galectin-1 (Gal-1) to upregulate Gal-1 expression in DCs, and induce the development of tolerogenic DC immunophenotypes, thereby inhibiting DC function (181). Furthermore, Xiao-Qing et al. demonstrated that small MSC-EVs inhibited the activation of mDCs on type 2 innate lymphoid cells (ILC2s) in patients with allergic rhinitis (182). Mechanistically, the PGE2-EP2/4 axis plays a key role in the immunomodulatory effects of EV-modified mDCs on ILC2s. The study also confirmed the ability of MSC-EVs to modulate DC function and inhibit the Th2 immune response in allergic conditions (182). By increasing the production of IL-10, EV-treated DCs can suppress the production of Th2 cytokines such as IL-4, IL-9, and IL-13, thereby alleviating allergic reactions (182). These findings suggest that MSC-EVs may have therapeutic potential for treating allergic airway inflammation by targeting DCs.
3.6 B cells
B cells play pro-inflammatory and anti-inflammatory roles in asthma by producing IgE antibodies, regulating immune responses, and participating in airway inflammation. They are key players in the immunopathogenesis of asthma. Studies by Gauvreau et al. have found elevated levels of IgE+ B cells in the airways of patients with asthma, suggesting their involvement in allergic inflammation (183). Beyond antibody production, B cells also act as antigen-presenting cells that regulate T cell responses. In asthma, B cells can influence the differentiation of Th2 cells, thereby contributing to allergic inflammatory processes (184). Studies have shown that MSCs can regulate B cell proliferation and differentiation while also inhibiting B cell apoptosis (80, 185, 186). Increased PD-L1 in lipopolysaccharide-treated human adipose-derived MSCs was found to inhibit B cell proliferation and IgG secretion via the NF-κB pathway (187). Guo et al. reported that MSC treatment downregulated Th17 cells and upregulated CD1dhighCD5+ regulatory B (Breg) cell activity, thereby reducing the severity of experimental autoimmune encephalomyelitis (188). Epstein-Barr virus-induced gene 3 (EBI3), a β-subunit of the IL-12 cytokine family, is induced by Epstein-Barr virus infection and forms various immune-regulating cytokines by pairing with different α-subunits and play an important role in immune regulation (189, 190). Tonsil-derived MSCs have been shown to alleviate B cell-mediated immune responses and increase the population of IL-10-producing Breg cells by upregulating EBI3 expression (190). Similar to T cells, miRNAs contained in MSC-EVs also exert regulatory effects on B cells. miR-125b, derived from labial gland MSC exosomes, have been shown to influence primary Sjögren’s syndrome by directly targeting PRDM1. As a result, the proportion of CD19+CD20-CD27+CD38+ plasma cells decreased significantly (191). EGR2 plays a key role in B cell development, particularly during their survival and differentiation stages (192, 193). A study on neonatal hypoxia-ischemia brain injury suggested that MSC-EV-derived miR-410 may upregulate EGR2 and BCL2 expression by downregulating HDAC1. These findings suggest that miR-410 may have a regulatory role in B cell function, although further studies are needed (194). In addition, MSCs may influence asthma by modulating Bregs, a heterogeneous group of B cell subsets with immunosuppressive properties similar to those of Tregs (195). Chao et al. demonstrated that human UC-MSCs protected against experimental colitis by increasing the number of CD5+ Bregs and correcting imbalances between Treg/Th17/Th1 cell subsets (196). Another study also confirmed that MSCs promote the survival and proliferation of CD5+ Bregs (197). Multiple studies have shown that MSCs can enhance Breg production. Given the pivotal role of B cells in adaptive immune responses, further research is warranted to elucidate how MSCs modulate asthma pathogenesis through B cell-mediated mechanisms.
3.7 Mast cells
Mast cells (MCs) have long been recognized as key effector cells in asthma because of their increased abundance in the airways of patients with asthma compared with healthy individuals (197–199). MC activation influences the phenotype of asthma. A study by Tiotiu et al. demonstrated that IL-33-stimulated MC signatures were associated with severe neutrophilic asthma, while IgE-activated MCs were associated with eosinophilic phenotypes (200). Regarding corticosteroid responsiveness, a study by Alzahrani et al. suggested that MC-derived mediators may contribute to glucocorticoid insensitivity in severe asthma (201). MSC-derived exosomes have been shown to inhibit TLR7-mediated MC activation and reduce pro-inflammatory cytokine release (202). In addition, another study demonstrated that MSC-derived microvesicles suppressed MC activation by upregulating PGE2 production and E-prostanoid 4 (EP4) receptor expression (203). Brown et al. reported that co-culturing MSCs with MCs resulted in reduced MC degranulation, pro-inflammatory cytokine production, and chemotaxis. These effects were mediated via upregulation of COX-2 and enhanced by the activation of EP4 receptors on MCs (204). Moreover, MSCs have been shown to reduce levels of IL-5 in patients with asthma, a key cytokine for MC activation (205). Reduced IL-5 levels can lead to decreased MC activity, thereby reducing asthma symptoms (206, 207). Overall, MSCs can suppress MC function and attenuate inflammatory responses through various pathways, offering potential benefit across asthma phenotypes. Nevertheless, further studies are needed to confirm these effects.
3.8 Epithelial cells
Airway epithelial cells contribute to the formation and development of airway inflammation and hyperresponsiveness in asthma through their barrier function and the release of various inflammatory mediators (208, 209). MSCs can modulate epithelial cell function via the secretion of paracrine factors that promote repair by enhancing migration, proliferation, and differentiation (210). Placenta-derived MSCs have been shown to reduce the expression of inflammatory cytokines such as IL-6 and IL-8 in epithelial cells exposed to particulate matter, exerting anti-inflammatory effects that may aid asthma control (211). Another study demonstrated that MSCs reduce inflammation by forming tunneling nanotubes to transfer mitochondria into airway epithelial cells with mitochondrial dysfunction (212). Connexin 43 (CX43) is a key molecule regulating mitochondrial transfer, and inhibiting CX43 weakens the therapeutic effect (212). The epithelial-mesenchymal transition (EMT) plays an important role in airway remodeling in asthma. During EMT, epithelial cells lose their polarity and adhesion properties, acquire mesenchymal characteristics, and exhibit increased migratory and invasive capabilities (213). In patients with asthma, exposure to allergens, contaminants, and infections activates the TGF-β1/SMAD signaling pathway, inducing EMT. This results in increased TGF-β1 expression, reduced levels of epithelial markers such as E-cadherin, and upregulation of mesenchymal markers such as vimentin and alpha-smooth muscle actin (214, 215). Song et al. demonstrated that MSCs and their exosomes significantly inhibit airway remodeling and the EMT by suppressing the Wnt/β-catenin signaling pathway (216). Additional studies have provided direct or indirect evidence supporting the ability of MSCs to inhibit the EMT in bronchial epithelial cells, suggesting promising therapeutic potential for MSCs in treating airway diseases characterized by excessive EMT and fibrosis (217–219). Further research is needed to explore the role of MSCs in modulating epithelial cell responses in asthma.
3.9 Airway smooth muscle cells
A hallmark of asthma is airway remodeling, which is closely associated with airway smooth muscle cell (ASMC) proliferation and migration (220). Experimental studies have demonstrated that extracellular miR-301a-3p from ADSCs can significantly reduce remodeling and the inflammatory response in PDGF-BB-stimulated ASMCs by targeting the STAT3 pathway (221). Several studies have shown that MSCs or their EVs can reduce airway smooth muscle thickness and improve airway remodeling in asthmatic mouse models (222–224). However, the precise signaling pathways remain unclear to be fully elucidated. Findings from Lin et al. suggest that the PI3K/AKT signaling pathway may play a role (225). Further investigation is needed to confirm this mechanism.
4 Effect of MSCs/MSC-EVs on asthma subtypes
As mentioned above, asthma can be divided into four inflammatory subtypes based on induced sputum examination. The proportion of eosinophils in sputum is significantly increased in patients with eosinophilic asthma, which is closely related to Th2 immune response and responds well to glucocorticoid treatment (12, 226). The percentage of neutrophils in sputum of patients with neutrophilic asthma is significantly increased, which is related to Th1 and Th17 inflammatory pathways. Neutrophilic asthma is often seen in patients with severe asthma or patients who smoke, and shows poor response to glucocorticoids, and is often accompanied by airway remodeling (227, 228). Patients with mixed granulocytic asthma have both increased eosinophils and neutrophils in sputum, and their clinical manifestations and pathological features are more complex and their symptoms are more severe (229). The proportion of eosinophils and neutrophils in sputum of patients with paucigranulocytic asthma is not significantly increased, and the related mechanism is not clear, but may relate to airway mucus hypersecretion or epithelial barrier dysfunction; the symptoms are persistent but there is a lack of typical inflammatory markers (230–232).
MSCs and MSC-EVs influence various asthma subtypes by impacting both immune and non-immune cells. In eosinophilic asthma, MSCs and MSC-EVs can inhibit Th2 cells differentiation by blocking GATA3 expression through TGF-β1 (104, 105), and activate PI3K/AKT pathway through miR-1470 and miR-21-5p to promote Treg expansion, reducing airway inflammation (91, 97). At the same time, they also regulate macrophages, induce their polarization to the M2 phenotype by TSG-6 and IL-10 (137–139), and inhibit the NF-κB inflammatory pathway through miR-21-5p and miR-146a-5p (140–142), exerting anti-inflammatory effects by regulating macrophages. In addition, they can directly inhibit eosinophil activation through hepatocyte growth factor secretion and TGF-β signaling (152, 158). They can also block mast cell degranulation by COX-2 up-regulation and EP4 receptor activation (204), repair barrier function by CX43-mediated mitochondrial transfer (212). However, it should be noted that the curative effects of MSCs from different sources are significantly different. For example, UC-MSCs have a significantly stronger ability to induce Treg than BM-MSCs (91), and MSC-EVs may promote Th2 transformation through unknown mechanisms (111).
For neutrophilic asthma, MSCs and MSC-EVs inhibit Th17 cells differentiation through IDO-mediated tryptophan metabolism (88), block Th17 cells proliferation by PD-L1 contact (117), and promote their transformation into Treg through mitochondrial reprogramming (120, 121). They can also block neutrophil recruitment by down-regulating CXCL1 by TSG-6 (168), inhibit NET formation by miR-199 (167), and inhibit NETosis by targeting TLR4/ROS/MAPK pathway by miR-17-5p (175). This reduces neutrophilic inflammation. Activated by TLR4, M1 macrophages can highly express CD86 and other markers, secrete IL-6, IL-1β and TNF-α, and directly recruit neutrophils to infiltrate the airway (227). MSCs can regulate macrophages, inhibit M1 polarization by activating calcium signal through CD54 interaction (146), and block cell pyroptosis by caspase-1 pathway inhibition (145). Activated MCs can directly recruit neutrophils to the airway by releasing chemokines such as LTB4, or even capture and internalize neutrophils by forming an intracellular mast cell trap, using its undigested substances to enhance its pro-inflammatory function and further recruit more neutrophils (233). MSCs and MSC-EVs can block IL-33-activated MCs and attenuate the activation of MCs by inhibiting TLR7 signaling pathway (202). It is worth noting that MSCs in the inflammatory state significantly inhibit the function of neutrophils, but different sources of MSCs have opposite effects; healthy donor MSCs inhibit NETosis, while MSCs derived from patients with cancer enhance this effect (174).
In patients with mixed granulocyte asthma, not only the percentage of neutrophils in induced sputum, but also the level of exhaled nitric oxide, was significantly increased, suggesting the coexistence of type 2 and non-type 2 inflammation (229). According to the above, MSCs and MSC-EVs can inhibit mixed granulocytic asthma by inhibiting eosinophilic inflammation and neutrophilic inflammation simultaneously by different immune cells. In paucigranulocytic asthma, MSCs and MSC-EVs mainly play a role by regulating epithelial barrier function and mucus secretion. Studies have shown that the characteristic mucus hypersecretion and epithelial barrier damage of this subtype of asthma are related to the abnormal release of epithelial-derived alarmins (230–232). MiR-146a and other molecules carried by MSC-EVs can inhibit the NF-κB pathway in airway epithelial cells and reduce the expression of MUC5AC mucin (210, 216). At the same time, MSCs repair epithelial tight junction proteins and restore barrier integrity through CX43-mediated mitochondrial transfer (212). In addition, HGF secreted by MSCs can down-regulate IL-25 and IL-33 expression (152), indirectly inhibit ILC2 activation, and block the vicious cycle of non-Th2 type inflammation, thereby improving airway hypersecretion and persistent symptoms of paucigranulocytic asthma (160, 230).
5 MSCs and asthma treatment
Corticosteroids play a central role in asthma treatment, with inhaled corticosteroids being essential for maintenance therapy in some patients (234). However, some patients develop resistance, and those with low Th2 asthma are not sensitive to steroids (235). In recent years, studies have suggested that MSCs can be used to treat asthma by controlling airway inflammation (205, 236, 237). Numerous animal studies have shown that MSCs can regulate Th1/Th2 balance, inhibit the maturation and function of DCs, suppress B cell maturation and antibody production, reduce the infiltration and activation of eosinophils and neutrophils, and inhibit M2 macrophage polarization. These mechanisms contribute to reduced airway inflammation and hyperresponsiveness, thereby alleviating asthma symptoms. In clinical research, several MSC-based therapies have been or are currently being evaluated. However, no MSC-related asthma treatments have passed phase 3 clinical trials. One discontinued phase I clinical trial (NCT03137199) evaluated the efficacy of intravenously administered BM-MSCs in two groups of patients with asthma, assessing lung volume and function, peripheral eosinophil levels, dyspnea, and quality of life every 4 weeks. Unfortunately, this trial was terminated in 2020. Notably, in a separate phase I clinical trial, a 68-year-old male patient with asthma received an intravenous infusion of human UC-MSCs cultured in vitro. Follow-up at 2 and 6 months post-treatment revealed a marked reduction in asthma attack frequency and a decreased dependence on inhalers and supplemental oxygen. No treatment-related adverse events were reported, indicating that MSC therapy significantly improved the patient’s quality of life (238). Regarding the safety of MSC therapy, a phase 1 clinical study is currently being conducted to evaluate the safety, toxicity, and potential mechanisms of interferon-γ-induced mesenchymal stromal cells (γMSCs) in the treatment of moderate-to-severe persistent asthma. Participants are divided into low- and high-dose groups, and the primary outcome measures include the number of adverse events and severe adverse events post-interventions, as well as the incidence of grade ≥3 adverse reactions attributed to the γMSC product. This trial is ongoing (NCT05035862). In another ongoing clinical experiment, airway epithelial cells were obtained from participants via bronchoscopy to evaluate the effects of adult stem cell products on asthmatic airway epithelium in vitro. The study aims to explore the utility of MSC secretome in eliminating IgE- and type-2 cytokine-driven immune response and reversing airway remodeling (NCT04883320). MSCs show therapeutic promise for asthma; however, challenges remain. These include difficulties in storage and transportation, the risk of incomplete differentiation, and limited lifespan (239–241). Therefore, MSC-EVs may represent a more practical alternative. In a currently ongoing phase II clinical trial (NCT04602104), patients are assigned to low-, medium-, and high-dose groups to receive either aerosolized human MSC exosomes or saline for seven consecutive days. Other trials are evaluating the safety of exosomes derived from healthy volunteers (NCT04313647). To date, however, no clinical trials evaluating MSC-EV therapy for asthma have been completed, and further research is needed.
6 Prospects and conclusion
Overall, current evidence suggests that MSCs exert diverse and beneficial effects in asthma. MSCs can inhibit airway inflammation and remodeling—two hallmark features of asthma—indicating their promise as a potential therapeutic approach. Given the limitations of existing therapies and the urgent need for development of novel treatments, MSC-based approaches represent an area of growing interest. MSCs appear to regulate immune responses across various inflammatory subtypes of asthma by modulating the proliferation, differentiation, and cytokine secretion of several key cell types involved in the pathogenesis of asthma, such as B cells, T cells, macrophages, MCs, neutrophils, and eosinophils. However, the specific mechanisms and signaling pathways of MSCs regulation in asthma remain unknown, and warrant further investigations.
The available evidence suggests that MSCs and their derived EVs have demonstrated significant therapeutic potential in asthma by modulating immune and non-immune cell functions. They inhibit the proliferation and activation of T cells, B cells, eosinophils, neutrophils, and DCs; promote Tregs and Bregs; and induce the polarization of macrophages from pro-inflammatory M1 to anti-inflammatory M2 phenotypes. Collectively, these actions contribute to the reduction of airway inflammation and hyperresponsiveness. Although the therapeutic potential of MSCs has shown positive results in different studies as well as in clinical trials, the specific mechanisms and signaling pathways of MSCs in patients with different phenotypes of asthma are still under investigation.
MSC therapy has the potential to transform asthma treatment, particularly for patients with steroid-resistant subtypes such as neutrophilic asthma. However, one of the key limitations of MSC therapy is the relatively low survival rate of the cells and high cost because of variability in cell quality when compared with existing biologic agents and corticosteroid-based therapies. Future development of MSC-based therapies should focus on improving cell quality and survival, enhancing immunomodulatory capacity, and minimizing production costs. Pretreatment strategies—such as hypoxia or cytokine priming—may enhance the therapeutic efficacy of MSCs while simultaneously reducing the associated costs. Compared to MSCs, MSC-EVs offer greater therapeutic promise due to their enhanced safety, improved stability, and more flexible delivery options.
Research into the role of MSC-EVs in regulating immune and structural cells in asthma is still in its early stages, and more clinical trials and studies on the specific mechanism of action need to be carried out. Further investigations are needed to establish the safety, efficacy, and mechanism of action of MSCs and MSC-EVs in asthma therapy. These efforts will be critical in paving the way for novel therapeutic strategies for this chronic inflammatory disease.
Author contributions
Z-YH: Writing – original draft, Writing – review & editing. Y-QH: Writing – review & editing, Writing – original draft. LZ: Writing – review & editing, Writing – original draft. WL: Writing – review & editing, Writing – original draft. PG: Writing – review & editing, Writing – original draft.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. This review was supported by the R&D project on key technologies for social development (20240304055SF and 20240304201SF).
Acknowledgments
Figure support was provided by Figdraw.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. Author contributions.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Whitehead GS, Thomas SY, Shalaby KH, Nakano K, Moran TP, Ward JM, et al. TNF is required for TLR ligand-mediated but not protease-mediated allergic airway inflammation. J Clin Invest. (2017) 127:3313–26. doi: 10.1172/JCI90890
2. Melén E, Zar HJ, Siroux V, Shaw D, Saglani S, Koppelman GH, et al. Asthma Inception: Epidemiologic Risk Factors and Natural History Across the Life Course. Am J Respir Crit Care Med. (2024) 210(6):737–54. doi: 10.1164/rccm.202312-2249SO
3. Barnig C, Cernadas M, Dutile S, Liu X, Perrella MA, Kazani S, et al. Lipoxin A4 regulates natural killer cell and type 2 innate lymphoid cell activation in asthma. Sci Transl Med. (2013) 5:174ra26. doi: 10.1126/scitranslmed.3004812
4. Thompson EE, Dang Q, Mitchell-Handley B, Rajendran K, Ram-Mohan S, Solway J, et al. Cytokine-induced molecular responses in airway smooth muscle cells inform genome-wide association studies of asthma. Genome Med. (2020) 12:64. doi: 10.1186/s13073-020-00759-w
5. Moon TC, St Laurent CD, Morris KE, Marcet C, Yoshimura T, Sekar Y, et al. Advances in mast cell biology: new understanding of heterogeneity and function. Mucosal Immunol. (2010) 3:111–28. doi: 10.1038/mi.2009.136
6. Altman MC, Lai Y, Nolin JD, Long S, Chen CC, Piliponsky AM, et al. Airway epithelium-shifted mast cell infiltration regulates asthmatic inflammation via IL-33 signaling. J Clin Invest. (2019) 129:4979–91. doi: 10.1172/JCI126402
7. Holgate ST, Wenzel S, Postma DS, Weiss ST, Renz H, and Sly PD. Asthma. Nat Rev Dis Primers. (2015) 1:15025. doi: 10.1038/nrdp.2015.25
8. Pérez-Losada M, Authelet KJ, Hoptay CE, Kwak C, Crandall KA, and Freishtat RJ. Pediatric asthma comprises different phenotypic clusters with unique nasal microbiotas. Microbiome. (2018) 6:179. doi: 10.1186/s40168-018-0564-7
9. Modena BD, Tedrow JR, Milosevic J, Bleecker ER, Meyers DA, Wu W, et al. Gene expression in relation to exhaled nitric oxide identifies novel asthma phenotypes with unique biomolecular pathways. Am J Respir Crit Care Med. (2014) 190:1363–72. doi: 10.1164/rccm.201406-1099OC
10. Papanicolaou A, Wang H, Satzke C, Vlahos R, Wilson N, Bozinovski S, et al. Therapies for pneumonia-associated severe asthma phenotypes. Trends Mol Med. (2020) 26:1047–58. doi: 10.1016/j.molmed.2020.07.006
11. Papi A, Brightling C, Pedersen SE, and Reddel HK. Asthma. Lancet. (2018) 391:783–800. doi: 10.1016/S0140-6736(17)33311-1
12. Venkatesan P. 2025 GINA report for asthma. Lancet Respir Med. (2025) 13(8):e41–2. doi: 10.1016/S2213-2600(25)00242-5
13. Pang Z, Ran N, Yuan Y, Wang C, Wang G, Lin H, et al. Phenotype-Specific Therapeutic Effect of Rhodiola wallichiana var. cholaensis Combined with Dexamethasone on Experimental Murine Asthma and Its Comprehensive Pharmacological Mechanism. Int J Mol Sci. (2019) 20:4216. doi: 10.3390/ijms20174216
14. Wang Y, Wu M, Li Y, Yuen HH, and He ML. The effects of SARS-CoV-2 infection on modulating innate immunity and strategies of combating inflammatory response for COVID-19 therapy. J BioMed Sci. (2022) 29:27. doi: 10.1186/s12929-022-00811-4
15. He Y, Shi J, Nguyen QT, You E, Liu H, Ren X, et al. Development of highly potent glucocorticoids for steroid-resistant severe asthma. Proc Natl Acad Sci U.S.A. (2019) 116:6932–7. doi: 10.1073/pnas.1816734116
16. Patel R, Naqvi SA, Griffiths C, and Bloom CI. Systemic adverse effects from inhaled corticosteroid use in asthma: a systematic review. BMJ Open Respir Res. (2020) 7:e000756. doi: 10.1136/bmjresp-2020-000756
17. Pavord ID, Mathieson N, Scowcroft A, Pedersini R, Isherwood G, and Price D. The impact of poor asthma control among asthma patients treated with inhaled corticosteroids plus long-acting β(2)-agonists in the United Kingdom: a cross-sectional analysis. NPJ Prim Care Respir Med. (2017) 27:17. doi: 10.1038/s41533-017-0014-1
18. Thomas D, McDonald VM, Stevens S, Harvey ES, Baraket M, Bardin P, et al. Biologics (mepolizumab and omalizumab) induced remission in severe asthma patients. Allergy. (2024) 79:384–92. doi: 10.1111/all.15867
19. Schleich F, Graff S, Nekoee H, Moermans C, Henket M, Sanchez C, et al. Real-word experience with mepolizumab: Does it deliver what it has promised? Clin Exp Allergy. (2020) 50:687–95. doi: 10.1111/CEA.13601
20. Jackson DJ, Burhan H, Rupani H, Pfeffer PE, Clifton IJ, Faruqi S, et al. Overcoming barriers to remission in severe eosinophilic asthma: two-year real-world data with benralizumab. Clin Exp Allergy. (2024) 54:734–46. doi: 10.1111/cea.v54.10
21. Maspero JF, Antila MA, Deschildre A, Bacharier LB, Altincatal A, Laws E, et al. Dupilumab efficacy in children with type 2 asthma receiving high- to medium-dose inhaled corticosteroids (VOYAGE). J Allergy Clin Immunol Pract. (2024) 12:3303–12. doi: 10.1016/j.jaip.2024.08.038
22. Caminati M, Vatrella A, Rogliani P, Carpagnano E, Spanevello A, and Senna G. Tezepelumab for severe asthma: elevating current practice to recognize epithelial driven profiles. Respir Res. (2024) 25:367. doi: 10.1186/s12931-024-02998-6
23. Ediger D, Günaydın FE, Erbay M, and Pekbak G. Can omalizumab be an alternative treatment for non-atopic severe asthma? A real-life experience with omalizumab. Tuberk Toraks. (2023) 71:24–33. doi: 10.5578/tt.20239904
24. Özden Ş., Tepetam FM, Örçen C, and Yakut T. The phenotypic heterogeneity of obese and nonobese patients with severe asthma and comparison of omalizumab-mepolizumab treatment efficiency in these patients. Med (Baltimore). (2023) 102:e35247. doi: 10.1097/MD.000000000000035247
25. Clavenna MJ, Turner JH, Samuelson M, Tanner SB, Duncavage J, and Chandra RK. Differential effect of omalizumab on pulmonary function in patients with allergic asthma with and without chronic rhinosinusitis. Allergy Asthma Proc. (2016) 37:23–6. doi: 10.2500/aap.2016.37.3923
26. Vennera MDC, Sabadell C, and Picado C. Duration of the efficacy of omalizumab after treatment discontinuation in 'real life' severe asthma. Thorax. (2018) 73:782–4. doi: 10.1136/thoraxjnl-2017-210017
27. Cutroneo PM, Arzenton E, Furci F, Scapini F, Bulzomì M, Luxi N, et al. Safety of biological therapies for severe asthma: an analysis of suspected adverse reactions reported in the WHO pharmacovigilance database. BioDrugs. (2024) 38:425–48. doi: 10.1007/s40259-024-00653-6
28. Briegel I, Felicio-Briegel A, Mertsch P, Kneidinger N, Haubner F, and Milger K. Hypereosinophilia with systemic manifestations under dupilumab and possibility of dual benralizumab and dupilumab therapy in patients with asthma and CRSwNP. J Allergy Clin Immunol Pract. (2021) 9:4477–9. doi: 10.1016/j.jaip.2021.07.049
29. Mareque M, Climente M, Martinez-Moragon E, Padilla A, Oyagüez I, Touron C, et al. Cost-effectiveness of benralizumab versus mepolizumab and dupilumab in patients with severe uncontrolled eosinophilic asthma in Spain. J Asthma. (2023) 60:1210–20. doi: 10.1080/02770903.2022.2139718
30. Ji R, Zhang X, Qian H, Gu H, Sun Z, Mao F, et al. miR-374 mediates the Malignant transformation of gastric cancer-associated mesenchymal stem cells in an experimental rat model. Oncol Rep. (2017) 38:1473–81. doi: 10.3892/or.2017.5831
31. White JL, Walker NJ, Hu JC, Borjesson DL, and Athanasiou KA. A comparison of bone marrow and cord blood mesenchymal stem cells for cartilage self-assembly. Tissue Eng Part A. (2018) 24:1262–72. doi: 10.1089/ten.tea.2017.0424
32. Jiang M, Wang X, Wang P, Peng W, Zhang B, and Guo L. Inhibitor of RAGE and glucose−induced inflammation in bone marrow mesenchymal stem cells: Effect and mechanism of action. Mol Med Rep. (2020) 22:3255–62. doi: 10.3892/mmr.2020.11422
33. Lin H, Sohn J, Shen H, Langhans MT, and Tuan RS. Bone marrow mesenchymal stem cells: Aging and tissue engineering applications to enhance bone healing. Biomaterials. (2019) 203:96–110. doi: 10.1016/j.biomaterials.2018.06.026
34. Kim SW, Lee YK, Hong JH, Park JY, Choi YA, Lee DU, et al. Mutual destruction of deep lung tumor tissues by nanodrug-conjugated stealth mesenchymal stem cells. Adv Sci (Weinh). (2018) 5:1700860. doi: 10.1002/advs.201700860
35. Wan C, He Q, McCaigue M, Marsh D, and Li G. Nonadherent cell population of human marrow culture is a complementary source of mesenchymal stem cells (MSCs). J Orthop Res. (2006) 24:21–8. doi: 10.1002/jor.20023
36. Islam MS, Stemig ME, Takahashi Y, and Hui SK. Radiation response of mesenchymal stem cells derived from bone marrow and human pluripotent stem cells. J Radiat Res. (2015) 56:269–77. doi: 10.1093/jrr/rru098
37. Mohyeldin A, Garzón-Muvdi T, and Quiñones-Hinojosa A. Oxygen in stem cell biology: a critical component of the stem cell niche. Cell Stem Cell. (2010) 7:150–61. doi: 10.1016/j.stem.2010.07.007
38. Wang Y, Fang J, Liu B, Shao C, and Shi Y. Reciprocal regulation of mesenchymal stem cells and immune responses. Cell Stem Cell. (2022) 29:1515–30. doi: 10.1016/j.stem.2022.10.001
39. Li N, Gao J, Mi L, Zhang G, Zhang L, Zhang N, et al. Synovial membrane mesenchymal stem cells: past life, current situation, and application in bone and joint diseases. Stem Cell Res Ther. (2020) 11:381. doi: 10.1186/s13287-020-01885-3
40. Qian HB, Zou G, Li C, He QF, and Liu J. Bone marrow mesenchymal stem cells attenuate LPS-induced acute lung injury in mice by promoting RvE1/ProD1 and modulating Treg/Th17 balance. Turk J Biol. (2022) 46:173–85. doi: 10.55730/1300-0152.2584
41. Liu J, Wu M, Feng G, Li R, Wang Y, and Jiao J. Downregulation of LINC00707 promotes osteogenic differentiation of human bone marrow−derived mesenchymal stem cells by regulating DKK1 via targeting miR−103a−3p. Int J Mol Med. (2020) 46:1029–38. doi: 10.3892/ijmm.2020.4672
42. De Becker A and Riet IV. Homing and migration of mesenchymal stromal cells: How to improve the efficacy of cell therapy? World J Stem Cells. (2016) 8:73–87. doi: 10.4252/wjsc.v8.i3.73
43. Yang R, Huang H, Cui S, Zhou Y, Zhang T, and Zhou Y. IFN-γ promoted exosomes from mesenchymal stem cells to attenuate colitis via miR-125a and miR-125b. Cell Death Dis. (2020) 11:603. doi: 10.1038/s41419-020-02788-0
44. Zha K, Sun Z, Yang Y, Chen M, Gao C, Fu L, et al. Recent developed strategies for enhancing chondrogenic differentiation of MSC: impact on MSC-based therapy for cartilage regeneration. Stem Cells Int. (2021) 2021:8830834. doi: 10.1155/2021/8830834
45. Sima Y and Chen Y. MSC-based therapy in female pelvic floor disorders. Cell Biosci. (2020) 10:104. doi: 10.1186/s13578-020-00466-4
46. Boland L, Bitterlich LM, Hogan AE, Ankrum JA, and English K. Translating MSC therapy in the age of obesity. Front Immunol. (2022) 13:943333. doi: 10.3389/fimmu.2022.943333
47. Goetz MJ, Kremer S, Behnke J, Staude B, Shahzad T, Holzfurtner L, et al. MSC based therapies to prevent or treat BPD-A narrative review on advances and ongoing challenges. Int J Mol Sci. (2021) 22:1138. doi: 10.3390/ijms22031138
48. Discher DE, Mooney DJ, and Zandstra PW. Growth factors, matrices, and forces combine and control stem cells. Science. (2009) 324:1673–7. doi: 10.1126/science.1171643
49. Trounson A and McDonald C. Stem cell therapies in clinical trials: progress and challenges. Cell Stem Cell. (2015) 17:11–22. doi: 10.1016/j.stem.2015.06.007
50. Qiao Y, Xu Z, Yu Y, Hou S, Geng J, Xiao T, et al. Single cell derived spheres of umbilical cord mesenchymal stem cells enhance cell stemness properties, survival ability and therapeutic potential on liver failure. Biomaterials. (2020) 227:119573. doi: 10.1016/j.biomaterials.2019.119573
51. Miethe S, Potaczek DP, Bazan-Socha S, Bachl M, Schaefer L, Wygrecka M, et al. The emerging role of extracellular vesicles as communicators between adipose tissue and pathologic lungs with a special focus on asthma. Am J Physiol Cell Physiol. (2023) 324:C1119–c1125. doi: 10.1152/ajpcell.00057.2023
52. Abbaszadeh H, Ghorbani F, Abbaspour-Aghdam S, Kamrani A, Valizadeh H, Nadiri M, et al. Chronic obstructive pulmonary disease and asthma: mesenchymal stem cells and their extracellular vesicles as potential therapeutic tools. Stem Cell Res Ther. (2022) 13:262. doi: 10.1186/s13287-022-02938-5
53. Tynecka M, Moniuszko M, and Eljaszewicz A. Old friends with unexploited perspectives: current advances in mesenchymal stem cell-based therapies in asthma. Stem Cell Rev Rep. (2021) 17:1323–42. doi: 10.1007/s12015-021-10137-7
54. Hu Q, Zhang S, Yang Y, Yao JQ, Tang WF, Lyon CJ, et al. Extracellular vesicles in the pathogenesis and treatment of acute lung injury. Mil Med Res. (2022) 9:61. doi: 10.1186/s40779-022-00417-9
55. Hahm J, Kim J, and Park J. Strategies to enhance extracellular vesicle production. Tissue Eng Regener Med. (2021) 18:513–24. doi: 10.1007/s13770-021-00364-x
56. Debbi L, Guo S, Safina D, and Levenberg S. Boosting extracellular vesicle secretion. Biotechnol Adv. (2022) 59:107983. doi: 10.1016/j.biotechadv.2022.107983
57. Wei W, Pan Y, Yang X, Chen Z, Heng Y, Yang B, et al. The emerging role of the interaction of extracellular vesicle and autophagy-novel insights into neurological disorders. J Inflammation Res. (2022) 15:3395–407. doi: 10.2147/JIR.S362865
58. Hu Q, Lyon CJ, Fletcher JK, Tang W, Wan M, and Hu TY. Extracellular vesicle activities regulating macrophage- and tissue-mediated injury and repair responses. Acta Pharm Sin B. (2021) 11:1493–512. doi: 10.1016/j.apsb.2020.12.014
59. Sidhom K, Obi PO, and Saleem A. A review of exosomal isolation methods: is size exclusion chromatography the best option? Int J Mol Sci. (2020) 21:6466. doi: 10.3390/ijms21186466
60. Lai C, Tan SS, Yeo RW, Choo AB, Reiner AT, Su Y, et al. MSC secretes at least 3 EV types each with a unique permutation of membrane lipid, protein and RNA. J Extracell Vesicles. (2016) 5:29828. doi: 10.3402/jev.v5.29828
61. Hade MD, Suire CN, Mossell J, and Suo Z. Extracellular vesicles: Emerging frontiers in wound healing. Med Res Rev. (2022) 42:2102–25. doi: 10.1002/med.21918
62. Wu R, Fan X, Wang Y, Shen M, Zheng Y, Zhao S, et al. Mesenchymal stem cell-derived extracellular vesicles in liver immunity and therapy. Front Immunol. (2022) 13:833878. doi: 10.3389/fimmu.2022.833878
63. He X, Yang L, Dong K, Zhang F, Liu Y, Ma B, et al. Biocompatible exosome-modified fibrin gel accelerates the recovery of spinal cord injury by VGF-mediated oligodendrogenesis. J Nanobiotechnology. (2022) 20:360. doi: 10.1186/s12951-022-01541-3
64. Puhka M, Thierens L, Nicorici D, Forsman T, Mirtti T, Af Hällström T, et al. Exploration of Extracellular Vesicle miRNAs, Targeted mRNAs and Pathways in Prostate Cancer: Relation to Disease Status and Progression. Cancers (Basel). (2022) 14:532. doi: 10.3390/cancers14030532
65. Peng YQ, Deng XH, Xu ZB, Wu ZC, and Fu QL. Mesenchymal stromal cells and their small extracellular vesicles in allergic diseases: From immunomodulation to therapy. Eur J Immunol. (2023) 53:e2149510. doi: 10.1002/eji.202149510
66. Ren J, Liu Y, Yao Y, Feng L, Zhao X, Li Z, et al. Intranasal delivery of MSC-derived exosomes attenuates allergic asthma via expanding IL-10 producing lung interstitial macrophages in mice. Int Immunopharmacol. (2021) 91:107288. doi: 10.1016/j.intimp.2020.107288
67. Cruz FF, Borg ZD, Goodwin M, Sokocevic D, Wagner D, McKenna DH, et al. Freshly thawed and continuously cultured human bone marrow-derived mesenchymal stromal cells comparably ameliorate allergic airways inflammation in immunocompetent mice. Stem Cells Transl Med. (2015) 4:615–24. doi: 10.5966/sctm.2014-0268
68. Lin G, Tao M, Sun H, Deng X, Zhang L, Sun G, et al. HGF-DPSCs ameliorate asthma by regulating CCR1(+) Th2 cells responses in mice pulmonary mucosa. Cytotherapy. (2025) 27(6):709–22. doi: 10.1016/j.jcyt.2025.02.005
69. Firinci F, Karaman M, Baran Y, Bagriyanik A, Ayyildiz ZA, Kiray M, et al. Mesenchymal stem cells ameliorate the histopathological changes in a murine model of chronic asthma. Int Immunopharmacol. (2011) 11:1120–6. doi: 10.1016/j.intimp.2011.03.009
70. Yuan JY, Wang XY, Tong ZY, Dong YC, Zhao JY, Zhang Y, et al. Promising therapeutic functions of bone marrow mesenchymal stem cells derived-exosome in asthma. Can Respir J. (2022) 2022:1485719. doi: 10.1155/2022/1485719
71. Trzil JE, Masseau I, Webb TL, Chang CH, Dodam JR, Cohn LA, et al. Long-term evaluation of mesenchymal stem cell therapy in a feline model of chronic allergic asthma. Clin Exp Allergy. (2014) 44:1546–57. doi: 10.1111/cea.2014.44.issue-12
72. Ma SY, Zhao N, Cui L, Li Y, Zhang H, Wang J, et al. Therapeutic effects of human pluripotent stem cell-derived mesenchymal stem cells on a murine model of acute type-2-dominated airway inflammation. Stem Cell Rev Rep. (2022) 18:2939–51. doi: 10.1007/s12015-022-10389-x
73. Zhang LB and He M. Effect of mesenchymal stromal (stem) cell (MSC) transplantation in asthmatic animal models: A systematic review and meta-analysis. Pulm Pharmacol Ther. (2019) 54:39–52. doi: 10.1016/j.pupt.2018.11.007
74. Mohammadian M, Sadeghipour HR, Kashani IR, Jahromi GP, Omidi A, Nejad AK, et al. Evaluation of simvastatin and bone marrow-derived mesenchymal stem cell combination therapy on airway remodeling in a mouse asthma model. Lung. (2016) 194:777–85. doi: 10.1007/s00408-016-9884-1
75. Niu J, Yue W, Le-Le Z, Bin L, and Hu X. Mesenchymal stem cells inhibit T cell activation by releasing TGF-β1 from TGF-β1/GARP complex. Oncotarget. (2017) 8:99784–800. doi: 10.18632/oncotarget.21549
76. Castro-Manrreza ME and Montesinos JJ. Immunoregulation by mesenchymal stem cells: biological aspects and clinical applications. J Immunol Res. (2015) 2015:394917. doi: 10.1155/2015/394917
77. Liu J, Xing F, Fu Q, He B, Jia Z, Du J, et al. hUC-MSCs exosomal miR-451 alleviated acute lung injury by modulating macrophage M2 polarization via regulating MIF-PI3K-AKT signaling pathway. Environ Toxicol. (2022) 37:2819–31. doi: 10.1002/tox.v37.12
78. Saeidi M, Masoud A, Shakiba Y, Hadjati J, Mohyeddin Bonab M, Nicknam MH, et al. Immunomodulatory effects of human umbilical cord Wharton's jelly-derived mesenchymal stem cells on differentiation, maturation and endocytosis of monocyte-derived dendritic cells. Iran J Allergy Asthma Immunol. (2013) 12:37–49.
79. Zhou Y, Cheng L, Liu L, and Li X. NK cells are never alone: crosstalk and communication in tumour microenvironments. Mol Cancer. (2023) 22:34. doi: 10.1186/s12943-023-01737-7
80. Liu J, Liu Q, and Chen X. The immunomodulatory effects of mesenchymal stem cells on regulatory B cells. Front Immunol. (2020) 11:1843. doi: 10.3389/fimmu.2020.01843
81. Mirershadi F, Ahmadi M, Rezabakhsh A, Rajabi H, Rahbarghazi R, and Keyhanmanesh R. Unraveling the therapeutic effects of mesenchymal stem cells in asthma. Stem Cell Res Ther. (2020) 11:400. doi: 10.1186/s13287-020-01921-2
82. Mansourabadi AH, Mohamed Khosroshahi L, Noorbakhsh F, and Amirzargar A. Cell therapy in transplantation: A comprehensive review of the current applications of cell therapy in transplant patients with the focus on Tregs, CAR Tregs, and Mesenchymal stem cells. Int Immunopharmacol. (2021) 97:107669. doi: 10.1016/j.intimp.2021.107669
83. Oppenheimer J, Hoyte FCL, Phipatanakul W, Silver J, Howarth P, and Lugogo NL. Allergic and eosinophilic asthma in the era of biomarkers and biologics: similarities, differences and misconceptions. Ann Allergy Asthma Immunol. (2022) 129:169–80. doi: 10.1016/j.anai.2022.02.021
84. Xie Y, Abel PW, Casale TB, and Tu Y. T(H)17 cells and corticosteroid insensitivity in severe asthma. J Allergy Clin Immunol. (2022) 149:467–79. doi: 10.1016/j.jaci.2021.12.769
85. Jin Y, Pan Z, Zhou J, Wang K, Zhu P, Wang Y, et al. Hedgehog signaling pathway regulates Th17 cell differentiation in asthma via IL-6/STAT3 signaling. Int Immunopharmacol. (2024) 139:112771. doi: 10.1016/j.intimp.2024.112771
86. Krampera M, Glennie S, Dyson J, Scott D, Laylor R, Simpson E, et al. Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood. (2003) 101:3722–9. doi: 10.1182/blood-2002-07-2104
87. Fujiwara Y, Kato S, Nesline MK, Conroy JM, DePietro P, Pabla S, et al. Indoleamine 2,3-dioxygenase (IDO) inhibitors and cancer immunotherapy. Cancer Treat Rev. (2022) 110:102461. doi: 10.1016/j.ctrv.2022.102461
88. Munn DH and Mellor AL. IDO in the tumor microenvironment: inflammation, counter-regulation, and tolerance. Trends Immunol. (2016) 37:193–207. doi: 10.1016/j.it.2016.01.002
89. Scheinecker C, Göschl L, and Bonelli M. Treg cells in health and autoimmune diseases: New insights from single cell analysis. J Autoimmun. (2020) 110:102376. doi: 10.1016/j.jaut.2019.102376
90. Mattar P and Bieback K. Comparing the immunomodulatory properties of bone marrow, adipose tissue, and birth-associated tissue mesenchymal stromal cells. Front Immunol. (2015) 6:560. doi: 10.3389/fimmu.2015.00560
91. Zhuansun Y, Du Y, Huang F, Lin L, Chen R, Jiang S, et al. MSCs exosomal miR-1470 promotes the differentiation of CD4(+)CD25(+)FOXP3(+) Tregs in asthmatic patients by inducing the expression of P27KIP1. Int Immunopharmacol. (2019) 77:105981. doi: 10.1016/j.intimp.2019.105981
92. Chiu M, Taurino G, Bianchi MG, and Bussolati O. The role of amino acids in the crosstalk between mesenchymal stromal cells and neoplastic cells in the hematopoietic niche. Front Cell Dev Biol. (2021) 9:714755. doi: 10.3389/fcell.2021.714755
93. Leyendecker A Jr., Pinheiro CCG, Amano MT, and Bueno DF. The use of human mesenchymal stem cells as therapeutic agents for the in vivo treatment of immune-related diseases: A systematic review. Front Immunol. (2018) 9:2056. doi: 10.3389/fimmu.2018.02056
94. Reis M, Mavin E, Nicholson L, Green K, Dickinson AM, and Wang XN. Mesenchymal stromal cell-derived extracellular vesicles attenuate dendritic cell maturation and function. Front Immunol. (2018) 9:2538. doi: 10.3389/fimmu.2018.02538
95. Wang K, Jiang Z, Webster KA, Chen J, Hu H, Zhou Y, et al. Enhanced cardioprotection by human endometrium mesenchymal stem cells driven by exosomal microRNA-21. Stem Cells Transl Med. (2017) 6:209–22. doi: 10.5966/sctm.2015-0386
96. Tang GN, Li CL, Yao Y, Xu ZB, Deng MX, Wang SY, et al. MicroRNAs involved in asthma after mesenchymal stem cells treatment. Stem Cells Dev. (2016) 25:883–96. doi: 10.1089/scd.2015.0339
97. Ahmadi M, Rahbarghazi R, Shahbazfar AA, Baghban H, and Keyhanmanesh R. Bone marrow mesenchymal stem cells modified pathological changes and immunological responses in ovalbumin-induced asthmatic rats possibly by the modulation of miRNA155 and miRNA133. Gen Physiol Biophys. (2018) 37:263–74. doi: 10.4149/gpb_2017052
98. Tavasolian F, Hosseini AZ, Soudi S, and Naderi M. miRNA-146a improves immunomodulatory effects of MSC-derived exosomes in rheumatoid arthritis. Curr Gene Ther. (2020) 20:297–312. doi: 10.2174/1566523220666200916120708
99. Khosravi M, Karimi MH, Hossein Aghdaie M, Kalani M, Naserian S, and Bidmeshkipour A. Mesenchymal stem cells can induce regulatory T cells via modulating miR-126a but not miR-10a. Gene. (2017) 627:327–36. doi: 10.1016/j.gene.2017.06.012
100. Lee S, Kim S, Chung H, Moon JH, Kang SJ, and Park CG. Mesenchymal stem cell-derived exosomes suppress proliferation of T cells by inducing cell cycle arrest through p27kip1/Cdk2 signaling. Immunol Lett. (2020) 225:16–22. doi: 10.1016/j.imlet.2020.06.006
101. Xu K, Ma D, Zhang G, Gao J, Su Y, Liu S, et al. Human umbilical cord mesenchymal stem cell-derived small extracellular vesicles ameliorate collagen-induced arthritis via immunomodulatory T lymphocytes. Mol Immunol. (2021) 135:36–44. doi: 10.1016/j.molimm.2021.04.001
102. Li Y, Li H, Cao Y, Wu F, Ma W, Wang Y, et al. Placenta−derived mesenchymal stem cells improve airway hyperresponsiveness and inflammation in asthmatic rats by modulating the Th17/Treg balance. Mol Med Rep. (2017) 16:8137–45. doi: 10.3892/mmr.2017.7605
103. Yang FY, Chen R, Zhang X, Huang B, Tsang LL, Li X, et al. Preconditioning enhances the therapeutic effects of mesenchymal stem cells on colitis through PGE2-mediated T-cell modulation. Cell Transplant. (2018) 27:1352–67. doi: 10.1177/0963689718780304
104. Lin Z, Cai W, Sun Y, Han B, Hu Y, He Z, et al. Mechanism and application of mesenchymal stem cells and their secreting extracellular vesicles in regulating CD4(+)T cells in immune diseases. Biophys Rep. (2024) 10:403–15. doi: 10.52601/bpr.2024.240005
105. Mo Y, Kang SY, Bang JY, Kim Y, Jeong J, Jeong EM, et al. Intravenous mesenchymal stem cell administration modulates monocytes/macrophages and ameliorates asthmatic airway inflammation in a murine asthma model. Mol Cells. (2022) 45:833–45. doi: 10.14348/molcells.2022.0038
106. Shin JW, Ryu S, Ham J, Jung K, Lee S, Chung DH, et al. Mesenchymal stem cells suppress severe asthma by directly regulating th2 cells and type 2 innate lymphoid cells. Mol Cells. (2021) 44:580–90. doi: 10.14348/molcells.2021.0101
107. Ou-Yang HF, Huang Y, Hu XB, and Wu CG. Suppression of allergic airway inflammation in a mouse model of asthma by exogenous mesenchymal stem cells. Exp Biol Med (Maywood). (2011) 236:1461–7. doi: 10.1258/ebm.2011.011221
108. Cho KS, Park MK, Kang SA, Park HY, Hong SL, Park HK, et al. Adipose-derived stem cells ameliorate allergic airway inflammation by inducing regulatory T cells in a mouse model of asthma. Mediators Inflammation. (2014) 2014:436476. doi: 10.1155/2014/436476
109. Cho KS, Park HK, Park HY, Jung JS, Jeon SG, Kim YK, et al. IFATS collection: Immunomodulatory effects of adipose tissue-derived stem cells in an allergic rhinitis mouse model. Stem Cells. (2009) 27:259–65. doi: 10.1634/stemcells.2008-0283
110. Goodwin M, Sueblinvong V, Eisenhauer P, Ziats NP, LeClair L, Poynter ME, et al. Bone marrow-derived mesenchymal stromal cells inhibit Th2-mediated allergic airways inflammation in mice. Stem Cells. (2011) 29:1137–48. doi: 10.1002/stem.v29.7
111. Chen W, Huang Y, Han J, Yu L, Li Y, Lu Z, et al. Immunomodulatory effects of mesenchymal stromal cells-derived exosome. Immunol Res. (2016) 64:831–40. doi: 10.1007/s12026-016-8798-6
112. Fang D, Cui K, Cao Y, Zheng M, Kawabe T, Hu G, et al. Differential regulation of transcription factor T-bet induction during NK cell development and T helper-1 cell differentiation. Immunity. (2022) 55:639–655.e7. doi: 10.1016/j.immuni.2022.03.005
113. Gao J, Hu J, Li P, Che K, Wang F, and Yan S. Human umbilical cord mesenchymal stem cells alleviate the imbalance of CD4(+) T cells via protein tyrosine phosphatase non-receptor type 2/signal transducer and activator of transcription 3 signaling in ameliorating experimental autoimmune thyroiditis in rats. Endocr J. (2022) 69:613–25. doi: 10.1507/endocrj.EJ20-0695
114. Newcomb DC and Peebles RS Jr. Th17-mediated inflammation in asthma. Curr Opin Immunol. (2013) 25:755–60. doi: 10.1016/j.coi.2013.08.002
115. Cosmi L, Liotta F, Maggi E, Romagnani S, and Annunziato F. Th17 cells: new players in asthma pathogenesis. Allergy. (2011) 66:989–98. doi: 10.1111/j.1398-9995.2011.02576.x
116. Mu Y, Xu W, Liu J, Wang Y, Chen J, and Zhou Q. Mesenchymal stem cells moderate experimental autoimmune uveitis by dynamic regulating Th17 and Breg cells response. J Tissue Eng Regener Med. (2022) 16:26–35. doi: 10.1002/term.v16.1
117. Guan J, Li Y, Lu F, and Feng J. Adipose-derived stem cells ameliorate atopic dermatitis by suppressing the IL-17 expression of Th17 cells in an ovalbumin-induced mouse model. Stem Cell Res Ther. (2022) 13:98. doi: 10.1186/s13287-022-02774-7
118. Zou Y, Xiao W, Liu D, Li X, Li L, Peng L, et al. Human umbilical cord mesenchymal stem cells improve disease characterization of Sjogren's syndrome in NOD mice through regulation of gut microbiota and Treg/Th17 cellular immunity. Immun Inflammation Dis. (2024) 12:e1139. doi: 10.1002/iid3.v12.1
119. Chen J, Shi X, Deng Y, Dang J, Liu Y, Zhao J, et al. miRNA-148a-containing GMSC-derived EVs modulate Treg/Th17 balance via IKKB/NF-κB pathway and treat a rheumatoid arthritis model. JCI Insight. (2024) 9:e177841. doi: 10.1172/jci.insight.177841
120. Akhter W, Nakhle J, Vaillant L, Garcin G, Saout C, Simon M, et al. Transfer of mesenchymal stem cell mitochondria to CD4(+) T cells contributes to repress Th1 differentiation by downregulating T-bet expression. Stem Cell Res Ther. (2023) 14:12. doi: 10.1186/s13287-022-03219-x
121. Luz-Crawford P, Hernandez J, Djouad F, Luque-Campos N, Caicedo A, Carrère-Kremer S, et al. Mesenchymal stem cell repression of Th17 cells is triggered by mitochondrial transfer. Stem Cell Res Ther. (2019) 10:232. doi: 10.1186/s13287-019-1307-9
122. Huang D, Sun C, Chen M, Bai S, Zhao X, Wang W, et al. Bergenin ameliorates airway inflammation and remodeling in asthma by activating SIRT1 in macrophages to regulate the NF-κB pathway. Front Pharmacol. (2022) 13:994878. doi: 10.3389/fphar.2022.994878
123. Menzel M, Kosinski J, Uller L, and Akbarshahi H. Rhinovirus-induced IFNβ expression is NFκB-dependent and regulated by the macrophage microenvironment. Sci Rep. (2019) 9:13394. doi: 10.1038/s41598-019-50034-1
124. Feketea G, Bocsan CI, Popescu C, Gaman M, Stanciu LA, and Zdrenghea MT. A review of macrophage microRNAs' Role in human asthma. Cells. (2019) 8:420. doi: 10.3390/cells8050420
125. Britt RD Jr., Ruwanpathirana A, Ford ML, and Lewis BW. Macrophages orchestrate airway inflammation, remodeling, and resolution in asthma. Int J Mol Sci. (2023) 24:10451. doi: 10.3390/ijms241310451
126. Wang S, Chen Y, Hong W, Li B, Zhou Y, and Ran P. Chronic exposure to biomass ambient particulate matter triggers alveolar macrophage polarization and activation in the rat lung. J Cell Mol Med. (2022) 26:1156–68. doi: 10.1111/jcmm.17169
127. Yuan G, Yang S, and Yang S. RGS12 represses oral squamous cell carcinoma by driving M1 polarization of tumor-associated macrophages via controlling ciliary MYCBP2/KIF2A signaling. Int J Oral Sci. (2023) 15:11. doi: 10.1038/s41368-023-00216-5
128. Tliba O and Panettieri RA Jr. Paucigranulocytic asthma: Uncoupling of airway obstruction from inflammation. J Allergy Clin Immunol. (2019) 143:1287–94. doi: 10.1016/j.jaci.2018.06.008
129. Hudey SN, Ledford DK, and Cardet JC. Mechanisms of non-type 2 asthma. Curr Opin Immunol. (2020) 66:123–8. doi: 10.1016/j.coi.2020.10.002
130. Dong H, Yang W, Li W, Zhu S, Zhu L, Gao P, et al. New insights into autophagy in inflammatory subtypes of asthma. Front Immunol. (2023) 14:1156086. doi: 10.3389/fimmu.2023.1156086
131. Braza F, Dirou S, Forest V, Sauzeau V, Hassoun D, Chesné J, et al. Mesenchymal stem cells induce suppressive macrophages through phagocytosis in a mouse model of asthma. Stem Cells. (2016) 34:1836–45. doi: 10.1002/stem.2344
132. Fang SB, Zhang HY, Meng XC, Wang C, He BX, Peng YQ, et al. Small extracellular vesicles derived from human MSCs prevent allergic airway inflammation via immunomodulation on pulmonary macrophages. Cell Death Dis. (2020) 11:409. doi: 10.1038/s41419-020-2606-x
133. Li H, Wang W, and Chang J. Calcium silicate enhances immunosuppressive function of MSCs to indirectly modulate the polarization of macrophages. Regener Biomater. (2021) 8:rbab056. doi: 10.1093/rb/rbab056
134. Xu Y, Liu X, Li Y, Dou H, Liang H, and Hou Y. SPION-MSCs enhance therapeutic efficacy in sepsis by regulating MSC-expressed TRAF1-dependent macrophage polarization. Stem Cell Res Ther. (2021) 12:531. doi: 10.1186/s13287-021-02593-2
135. Abreu SC, Antunes MA, Xisto DG, Cruz FF, Branco VC, Bandeira E, et al. Bone marrow, adipose, and lung tissue-derived murine mesenchymal stromal cells release different mediators and differentially affect airway and lung parenchyma in experimental asthma. Stem Cells Transl Med. (2017) 6:1557–67. doi: 10.1002/sctm.16-0398
136. Song X, Xie S, Lu K, and Wang C. Mesenchymal stem cells alleviate experimental asthma by inducing polarization of alveolar macrophages. Inflammation. (2015) 38:485–92. doi: 10.1007/s10753-014-9954-6
137. Nakao Y, Fukuda T, Zhang Q, Sanui T, Shinjo T, Kou X, et al. Corrigendum to "Exosomes from TNF-α-treated human gingiva-derived MSCs enhance M2 macrophage polarization and inhibit periodontal bone loss" [Acta Biomaterialia 2021, 122, 306-324. Acta Biomater. (2025) 191:428–9. doi: 10.1016/j.actbio.2024.11.029
138. Arabpour M, Saghazadeh A, and Rezaei N. Anti-inflammatory and M2 macrophage polarization-promoting effect of mesenchymal stem cell-derived exosomes. Int Immunopharmacol. (2021) 97:107823. doi: 10.1016/j.intimp.2021.107823
139. Zhang M, Johnson-Stephenson TK, Wang W, Wang Y, Li J, Li L, et al. Mesenchymal stem cell-derived exosome-educated macrophages alleviate systemic lupus erythematosus by promoting efferocytosis and recruitment of IL-17(+) regulatory T cell. Stem Cell Res Ther. (2022) 13:484. doi: 10.1186/s13287-022-03174-7
140. Zhao J, Li X, Hu J, Chen F, Qiao S, Sun X, et al. Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovasc Res. (2019) 115:1205–16. doi: 10.1093/cvr/cvz040
141. Ren W, Hou J, Yang C, Wang H, Wu S, Wu Y, et al. Extracellular vesicles secreted by hypoxia pre-challenged mesenchymal stem cells promote non-small cell lung cancer cell growth and mobility as well as macrophage M2 polarization via miR-21-5p delivery. J Exp Clin Cancer Res. (2019) 38:62. doi: 10.1186/s13046-019-1027-0
142. Li J, Pan Y, Yang J, Wang J, Jiang Q, Dou H, et al. Tumor necrosis factor-α-primed mesenchymal stem cell-derived exosomes promote M2 macrophage polarization via Galectin-1 and modify intrauterine adhesion on a novel murine model. Front Immunol. (2022) 13:945234. doi: 10.3389/fimmu.2022.945234
143. Yuan Y, Yuan L, Li L, Liu F, Liu J, Chen Y, et al. Mitochondrial transfer from mesenchymal stem cells to macrophages restricts inflammation and alleviates kidney injury in diabetic nephropathy mice via PGC-1α activation. Stem Cells. (2021) 39:913–28. doi: 10.1002/stem.3375
144. Li Y, Zhang D, Xu L, Dong L, Zheng J, Lin Y, et al. Cell-cell contact with proinflammatory macrophages enhances the immunotherapeutic effect of mesenchymal stem cells in two abortion models. Cell Mol Immunol. (2019) 16:908–20. doi: 10.1038/s41423-019-0204-6
145. Liu P, Yang S, Shao X, Li C, Wang Z, Dai H, et al. Mesenchymal stem cells-derived exosomes alleviate acute lung injury by inhibiting alveolar macrophage pyroptosis. Stem Cells Transl Med. (2024) 13:371–86. doi: 10.1093/stcltm/szad094
146. Espagnolle N, Balguerie A, Arnaud E, Sensebé L, and Varin A. CD54-mediated interaction with pro-inflammatory macrophages increases the immunosuppressive function of human mesenchymal stromal cells. Stem Cell Rep. (2017) 8:961–76. doi: 10.1016/j.stemcr.2017.02.008
147. Du Rocher B, Mencalha AL, Gomes BE, and Abdelhay E. Mesenchymal stromal cells impair the differentiation of CD14(++) CD16(-) CD64(+) classical monocytes into CD14(++) CD16(+) CD64(++) activate monocytes. Cytotherapy. (2012) 14:12–25. doi: 10.3109/14653249.2011.594792
148. Zhang Y, Yan B, Shen S, Song X, Jiang Y, Shi L, et al. Efficacy and safety of CM310 in severe eosinophilic chronic rhinosinusitis with nasal polyps (CROWNS-1): a multicentre, randomised, double-blind, placebo-controlled phase 2 clinical trial. EClinicalMedicine. (2023) 61:102076. doi: 10.1016/j.eclinm.2023.102076
149. Cardet JC and Israel E. Update on reslizumab for eosinophilic asthma. Expert Opin Biol Ther. (2015) 15:1531–9. doi: 10.1517/14712598.2015.1090972
150. Deeks ED and Brusselle G. Reslizumab in eosinophilic asthma: A review. Drugs. (2017) 77:777–84. doi: 10.1007/s40265-017-0740-2
151. Humbert M, Busse W, Hanania NA, Lowe PJ, Canvin J, Erpenbeck VJ, et al. Omalizumab in asthma: an update on recent developments. J Allergy Clin Immunol Pract. (2014) 2:525–36.e1. doi: 10.1016/j.jaip.2014.03.010
152. Fang Q, Wu W, Xiao Z, Zeng D, Liang R, Wang J, et al. Gingival-derived mesenchymal stem cells alleviate allergic asthma inflammation via HGF in animal models. iScience. (2024) 27:109818. doi: 10.1016/j.isci.2024.109818
153. Moghaddasi K, Hesaraki S, Arfaee F, and Athari SS. Investigating the effect of mesenchymal stem cells on the rate of clinical and pathological improvement of asthmatic lung in mouse model. Regener Ther. (2024) 25:157–61. doi: 10.1016/j.reth.2023.12.013
154. Dong L, Wang Y, Zheng T, Pu Y, Ma Y, Qi X, et al. Hypoxic hUCMSC-derived extracellular vesicles attenuate allergic airway inflammation and airway remodeling in chronic asthma mice. Stem Cell Res Ther. (2021) 12:4. doi: 10.1186/s13287-020-02072-0
155. de Castro LL, Xisto DG, Kitoko JZ, Cruz FF, Olsen PC, Redondo PAG, et al. Human adipose tissue mesenchymal stromal cells and their extracellular vesicles act differentially on lung mechanics and inflammation in experimental allergic asthma. Stem Cell Res Ther. (2017) 8:151. doi: 10.1186/s13287-017-0600-8
156. Castro LL, Kitoko JZ, Xisto DG, Olsen PC, Guedes HLM, Morales MM, et al. Multiple doses of adipose tissue-derived mesenchymal stromal cells induce immunosuppression in experimental asthma. Stem Cells Transl Med. (2020) 9:250–60. doi: 10.1002/sctm.19-0120
157. Wang H, Zhang Q, Wu S, Pan D, Ning Y, Wang C, et al. Mesenchymal stem cell therapy in eosinophilic granulomatosis with polyangiitis-related lower limb gangrene: a case report. Stem Cell Res Ther. (2024) 15:307. doi: 10.1186/s13287-024-03924-9
158. Nemeth K, Keane-Myers A, Brown JM, Metcalfe DD, Gorham JD, Bundoc VG, et al. Bone marrow stromal cells use TGF-beta to suppress allergic responses in a mouse model of ragweed-induced asthma. Proc Natl Acad Sci U.S.A. (2010) 107:5652–7. doi: 10.1073/pnas.0910720107
159. Kim HS, Lee JH, Roh KH, Jun HJ, Kang KS, and Kim TY. Clinical trial of human umbilical cord blood-derived stem cells for the treatment of moderate-to-severe atopic dermatitis: phase I/IIa studies. Stem Cells. (2017) 35:248–55. doi: 10.1002/stem.2401
160. Samivel R, Kim EH, Chung YJ, and Mo JH. Immunomodulatory effect of tonsil-derived mesenchymal stem cells in a mouse model of allergic rhinitis. Am J Rhinol Allergy. (2015) 29:262–7. doi: 10.2500/ajra.2015.29.4216
161. Hou C, Sun F, Liang Y, Nasab EM, and Athari SS. Effect of transduced mesenchymal stem cells with IL-10 gene on control of allergic asthma. Allergol Immunopathol (Madr). (2023) 51:45–51. doi: 10.15586/aei.v51i2.789
162. Gao H, Ying S, and Dai Y. Pathological roles of neutrophil-mediated inflammation in asthma and its potential for therapy as a target. J Immunol Res. (2017) 2017:3743048. doi: 10.1155/2017/3743048
163. Nair P and Prabhavalkar KS. Neutrophilic asthma and potentially related target therapies. Curr Drug Targets. (2020) 21:374–88. doi: 10.2174/1389450120666191011162526
164. Varricchi G, Modestino L, Poto R, Cristinziano L, Gentile L, Postiglione L, et al. Neutrophil extracellular traps and neutrophil-derived mediators as possible biomarkers in bronchial asthma. Clin Exp Med. (2022) 22:285–300. doi: 10.1007/s10238-021-00750-8
165. Grunwell JR, Stephenson ST, Tirouvanziam R, Brown LAS, Brown MR, and Fitzpatrick AM. Children with neutrophil-predominant severe asthma have proinflammatory neutrophils with enhanced survival and impaired clearance. J Allergy Clin Immunol Pract. (2019) 7:516–525.e6. doi: 10.1016/j.jaip.2018.08.024
166. Chen X, Li Y, Qin L, He R, and Hu C. Neutrophil extracellular trapping network promotes the pathogenesis of neutrophil-associated asthma through macrophages. Immunol Invest. (2021) 50:544–61. doi: 10.1080/08820139.2020.1778720
167. Mahmoudi M, Taghavi-Farahabadi M, Namaki S, Baghaei K, Rayzan E, Rezaei N, et al. Exosomes derived from mesenchymal stem cells improved function and survival of neutrophils from severe congenital neutropenia patients in vitro. Hum Immunol. (2019) 80:990–8. doi: 10.1016/j.humimm.2019.10.006
168. Ding Y, Gong P, Jiang J, Feng C, Li Y, Su X, et al. Mesenchymal stem/stromal cells primed by inflammatory cytokines alleviate psoriasis-like inflammation via the TSG-6-neutrophil axis. Cell Death Dis. (2022) 13:996. doi: 10.1038/s41419-022-05445-w
169. de Castro LL, Lopes-Pacheco M, Weiss DJ, Cruz FF, and Rocco PRM. Current understanding of the immunosuppressive properties of mesenchymal stromal cells. J Mol Med (Berl). (2019) 97:605–18. doi: 10.1007/s00109-019-01776-y
170. Dehnavi S, Sadeghi M, Tavakol Afshari J, and Mohammadi M. Interactions of mesenchymal stromal/stem cells and immune cells following MSC-based therapeutic approaches in rheumatoid arthritis. Cell Immunol. (2023) 393-394:104771. doi: 10.1016/j.cellimm.2023.104771
171. Salami F, Tavassoli A, Mehrzad J, and Parham A. Immunomodulatory effects of mesenchymal stem cells on leukocytes with emphasis on neutrophils. Immunobiology. (2018) 223:786–91. doi: 10.1016/j.imbio.2018.08.002
172. Feng B, Feng X, Yu Y, Xu H, Ye Q, Hu R, et al. Mesenchymal stem cells shift the pro-inflammatory phenotype of neutrophils to ameliorate acute lung injury. Stem Cell Res Ther. (2023) 14:197. doi: 10.1186/s13287-023-03438-w
173. Mutua V and Gershwin LJ. A review of neutrophil extracellular traps (NETs) in disease: potential anti-NETs therapeutics. Clin Rev Allergy Immunol. (2021) 61:194–211. doi: 10.1007/s12016-020-08804-7
174. Wang LT, Lee W, Liu KJ, Sytwu HK, Yen ML, and Yen BL. Mesenchymal stromal/stem cells know best: the remarkable complexities of its interactions with polymorphonuclear neutrophils. Stem Cells. (2024) 42:403–15. doi: 10.1093/stmcls/sxae011
175. Wang T, Zhang Z, Deng Z, Zeng W, Gao Y, Hei Z, et al. Mesenchymal stem cells alleviate sepsis-induced acute lung injury by blocking neutrophil extracellular traps formation and inhibiting ferroptosis in rats. PeerJ. (2024) 12:e16748. doi: 10.7717/peerj.16748
176. Zeng SL, Wang LH, Li P, Wang W, and Yang J. Mesenchymal stem cells abrogate experimental asthma by altering dendritic cell function. Mol Med Rep. (2015) 12:2511–20. doi: 10.3892/mmr.2015.3706
177. Gu W, Zheng T, Li W, Luo X, Xu X, Wang Y, et al. Migrasomes derived from human umbilical cord mesenchymal stem cells: a new therapeutic agent for ovalbumin-induced asthma in mice. Stem Cell Res Ther. (2025) 16:26. doi: 10.1186/s13287-025-04145-4
178. Duong KM, Arikkatt J, Ullah MA, Lynch JP, Zhang V, Atkinson K, et al. Immunomodulation of airway epithelium cell activation by mesenchymal stromal cells ameliorates house dust mite-induced airway inflammation in mice. Am J Respir Cell Mol Biol. (2015) 53:615–24. doi: 10.1165/rcmb.2014-0431OC
179. Peng YQ, Wu ZC, Xu ZB, Fang SB, Chen DH, Zhang HY, et al. Mesenchymal stromal cells-derived small extracellular vesicles modulate DC function to suppress Th2 responses via IL-10 in patients with allergic rhinitis. Eur J Immunol. (2022) 52:1129–40. doi: 10.1002/eji.202149497
180. Ghosh T, Barik S, Bhuniya A, Dhar J, Dasgupta S, Ghosh S, et al. Tumor-associated mesenchymal stem cells inhibit naïve T cell expansion by blocking cysteine export from dendritic cells. Int J Cancer. (2016) 139:2068–81. doi: 10.1002/ijc.v139.9
181. Zhang Y, Ge XH, Guo XJ, Guan SB, Li XM, Gu W, et al. Bone marrow mesenchymal stem cells inhibit the function of dendritic cells by secreting galectin-1. BioMed Res Int. (2017) 2017:3248605. doi: 10.1155/2017/3248605
182. Liu XQ, Peng YQ, Huang LX, Li CG, Kuang PP, Chen DH, et al. Dendritic cells mediated by small extracellular vesicles derived from MSCs attenuated the ILC2 activity via PGE2 in patients with allergic rhinitis. Stem Cell Res Ther. (2023) 14:180. doi: 10.1186/s13287-023-03408-2
183. Oliveria JP, Salter BM, Phan S, Obminski CD, Munoz CE, Smith SG, et al. Asthmatic subjects with allergy have elevated levels of IgE+ B cells in the airways. J Allergy Clin Immunol. (2017) 140:590–593.e9. doi: 10.1016/j.jaci.2016.12.981
184. Drake LY, Iijima K, Hara K, Kobayashi T, Kephart GM, and Kita H. B cells play key roles in th2-type airway immune responses in mice exposed to natural airborne allergens. PloS One. (2015) 10:e0121660. doi: 10.1371/journal.pone.0121660
185. Corcione A, Benvenuto F, Ferretti E, Giunti D, Cappiello V, Cazzanti F, et al. Human mesenchymal stem cells modulate B-cell functions. Blood. (2006) 107:367–72. doi: 10.1182/blood-2005-07-2657
186. Kitoko JZ, de Castro LL, Nascimento AP, Abreu SC, Cruz FF, Arantes AC, et al. Therapeutic administration of bone marrow-derived mesenchymal stromal cells reduces airway inflammation without up-regulating Tregs in experimental asthma. Clin Exp Allergy. (2018) 48:205–16. doi: 10.1111/cea.2018.48.issue-2
187. Sun J, Zhong H, Kang B, Lum T, Liu D, Liang S, et al. Roles of PD-L1 in human adipose-derived mesenchymal stem cells under inflammatory microenvironment. J Cell Biochem. (2024) 125:e30544. doi: 10.1002/jcb.v125.4
188. Guo Y, Chan KH, Lai WH, Siu CW, Kwan SC, Tse HF, et al. Human mesenchymal stem cells upregulate CD1dCD5(+) regulatory B cells in experimental autoimmune encephalomyelitis. Neuroimmunomodulation. (2013) 20:294–303. doi: 10.1159/000351450
189. Scott EN, Ye C, Yano H, Lipatova Z, Brunazzi E, Vignali KM, et al. Ebi3 binding to IFN-γ and IL-10 limits their function. J Immunol. (2024) 213:1115–24. doi: 10.4049/jimmunol.2400236
190. Cho KA, Lee JK, Kim YH, Park M, Woo SY, and Ryu KH. Mesenchymal stem cells ameliorate B-cell-mediated immune responses and increase IL-10-expressing regulatory B cells in an EBI3-dependent manner. Cell Mol Immunol. (2017) 14:895–908. doi: 10.1038/cmi.2016.59
191. Wang M, Yang G, Jiang X, Lu D, Mei H, and Chen B. Peroxisome proliferator-activated receptor-γ Coactivator-1α (PGC-1α) regulates the expression of B-cell lymphoma/leukemia-2 (Bcl-2) and promotes the survival of mesenchymal stem cells (MSCs) via PGC-1α/ERRα Interaction in the absence of serum. Hypoxia High Glucose Conditions Med Sci Monit. (2017) 23:3451–60. doi: 10.12659/MSM.902183
192. Li S, Miao T, Sebastian M, Bhullar P, Ghaffari E, Liu M, et al. The transcription factors Egr2 and Egr3 are essential for the control of inflammation and antigen-induced proliferation of B and T cells. Immunity. (2012) 37:685–96. doi: 10.1016/j.immuni.2012.08.001
193. Miao T, Symonds ALJ, Singh R, Symonds JD, Ogbe A, Omodho B, et al. Egr2 and 3 control adaptive immune responses by temporally uncoupling expansion from T cell differentiation. J Exp Med. (2017) 214:1787–808. doi: 10.1084/jem.20160553
194. Han J, Yang S, Hao X, Zhang B, Zhang H, Xin C, et al. Extracellular vesicle-derived microRNA-410 from mesenchymal stem cells protects against neonatal hypoxia-ischemia brain damage through an HDAC1-dependent EGR2/bcl2 axis. Front Cell Dev Biol. (2020) 8:579236. doi: 10.3389/fcell.2020.579236
195. Mickael ME, Bieńkowska I, and Sacharczuk M. An update on the evolutionary history of bregs. Genes (Basel). (2022) 13. doi: 10.3390/genes13050890
196. Chao K, Zhang S, Qiu Y, Chen X, Zhang X, Cai C, et al. Human umbilical cord-derived mesenchymal stem cells protect against experimental colitis via CD5(+) B regulatory cells. Stem Cell Res Ther. (2016) 7:109. doi: 10.1186/s13287-016-0376-2
197. Peng Y, Chen X, Liu Q, Zhang X, Huang K, Liu L, et al. Mesenchymal stromal cells infusions improve refractory chronic graft versus host disease through an increase of CD5+ regulatory B cells producing interleukin 10. Leukemia. (2015) 29:636–46. doi: 10.1038/leu.2014.225
198. Amin K, Janson C, Boman G, and Venge P. The extracellular deposition of mast cell products is increased in hypertrophic airways smooth muscles in allergic asthma but not in nonallergic asthma. Allergy. (2005) 60:1241–7. doi: 10.1111/j.1398-9995.2005.00823.x
199. Yoo JM, Yang JH, Kim YS, Yang HJ, Cho WK, and Ma JY. Inhibitory effects of viscum coloratum extract on igE/antigen-activated mast cells and mast cell-derived inflammatory mediator-activated chondrocytes. Molecules. (2016) 22. doi: 10.3390/molecules22010037
200. Tiotiu A, Badi Y, Kermani NZ, Sanak M, Kolmert J, Wheelock CE, et al. Association of differential mast cell activation with granulocytic inflammation in severe asthma. Am J Respir Crit Care Med. (2022) 205:397–411. doi: 10.1164/rccm.202102-0355OC
201. Alzahrani A, Hakeem J, Biddle M, Alhadian F, Hussain A, Khalfaoui L, et al. Human lung mast cells impair corticosteroid responsiveness in human airway smooth muscle cells. Front Allergy. (2021) 2:785100. doi: 10.3389/falgy.2021.785100
202. Cho KA, Cha JE, Kim J, Kim YH, Ryu KH, and Woo SY. Mesenchymal stem cell-derived exosomes attenuate TLR7-mediated mast cell activation. Tissue Eng Regener Med. (2022) 19:117–29. doi: 10.1007/s13770-021-00395-4
203. Liu J, Kuwabara A, Kamio Y, Hu S, Park J, Hashimoto T, et al. Human mesenchymal stem cell-derived microvesicles prevent the rupture of intracranial aneurysm in part by suppression of mast cell activation via a PGE2-dependent mechanism. Stem Cells. (2016) 34:2943–55. doi: 10.1002/stem.2448
204. Brown JM, Nemeth K, Kushnir-Sukhov NM, Metcalfe DD, and Mezey E. Bone marrow stromal cells inhibit mast cell function via a COX2-dependent mechanism. Clin Exp Allergy. (2011) 41:526–34. doi: 10.1111/j.1365-2222.2010.03685.x
205. Lin SC, Liou YM, Ling TY, Chuang YH, and Chiang BL. Placenta-derived mesenchymal stem cells reduce the interleukin-5 level experimentally in children with asthma. Int J Med Sci. (2019) 16:1430–8. doi: 10.7150/ijms.33590
206. Dougan M, Dranoff G, and Dougan SK. GM-CSF, IL-3, and IL-5 family of cytokines: regulators of inflammation. Immunity. (2019) 50:796–811. doi: 10.1016/j.immuni.2019.03.022
207. Calhoun KH. Asthma treatments: new and emerging therapies. Int Forum Allergy Rhinol. (2015) 5 Suppl 1:S76–81. doi: 10.1002/alr.2015.5.issue-S1
208. Raby KL, Michaeloudes C, Tonkin J, Chung KF, and Bhavsar PK. Mechanisms of airway epithelial injury and abnormal repair in asthma and COPD. Front Immunol. (2023) 14:1201658. doi: 10.3389/fimmu.2023.1201658
209. Chatziparasidis G, Bush A, Chatziparasidi MR, and Kantar A. Airway epithelial development and function: A key player in asthma pathogenesis? Paediatr Respir Rev. (2023) 47:51–61. doi: 10.1016/j.prrv.2023.04.005
210. Halim NS, Aizat WM, and Yahaya BH. The effect of mesenchymal stem cell-secreted factors on airway epithelial repair. Regener Med. (2019) 14:15–31. doi: 10.2217/rme-2018-0020
211. Kim SY, Oh SH, Lee JH, Suh MW, and Park MK. Effects of placenta-derived mesenchymal stem cells on the particulate matter-induced damages in human middle ear epithelial cells. Stem Cells Int. (2019) 2019:4357684. doi: 10.1155/2019/4357684
212. Yao Y, Fan XL, Jiang D, Zhang Y, Li X, Xu ZB, et al. Connexin 43-mediated mitochondrial transfer of iPSC-MSCs alleviates asthma inflammation. Stem Cell Rep. (2018) 11:1120–35. doi: 10.1016/j.stemcr.2018.09.012
213. Loffredo LF, Abdala-Valencia H, Anekalla KR, Cuervo-Pardo L, Gottardi CJ, and Berdnikovs S. Beyond epithelial-to-mesenchymal transition: Common suppression of differentiation programs underlies epithelial barrier dysfunction in mild. moderate Severe asthma Allergy. (2017) 72:1988–2004. doi: 10.1111/all.2017.72.issue-12
214. Berair R and Brightling CE. Asthma therapy and its effect on airway remodelling. Drugs. (2014) 74:1345–69. doi: 10.1007/s40265-014-0250-4
215. Hackett TL. Epithelial-mesenchymal transition in the pathophysiology of airway remodelling in asthma. Curr Opin Allergy Clin Immunol. (2012) 12:53–9. doi: 10.1097/ACI.0b013e32834ec6eb
216. Song J, Zhu XM, and Wei QY. MSCs reduce airway remodeling in the lungs of asthmatic rats through the Wnt/β-catenin signaling pathway. Eur Rev Med Pharmacol Sci. (2020) 24:11199–211. doi: 10.26355/eurrev_202011_23608
217. Li X, Yan X, Wang Y, Wang J, Zhou F, Wang H, et al. NLRP3 inflammasome inhibition attenuates silica-induced epithelial to mesenchymal transition (EMT) in human bronchial epithelial cells. Exp Cell Res. (2018) 362:489–97. doi: 10.1016/j.yexcr.2017.12.013
218. Al-Ruweidi M, Ali FH, Shurbaji S, Popelka A, and Yalcin HC. Dexamethasone and transdehydroandrosterone significantly reduce pulmonary epithelial cell injuries associated with mechanical ventilation. J Appl Physiol (1985). (2021) 130:1143–51. doi: 10.1152/japplphysiol.00574.2020
219. Xu L, Xiang X, Ji X, Wang W, Luo M, Luo S, et al. Effects and mechanism of dehydroepiandrosterone on epithelial-mesenchymal transition in bronchial epithelial cells. Exp Lung Res. (2014) 40:211–21. doi: 10.3109/01902148.2013.879966
220. Camoretti-Mercado B and Lockey RF. Airway smooth muscle pathophysiology in asthma. J Allergy Clin Immunol. (2021) 147:1983–95. doi: 10.1016/j.jaci.2021.03.035
221. Feng CY, Bai SY, Li ML, Zhao JY, Sun JM, Bao HJ, et al. Adipose-derived mesenchymal stem cell-derived exosomal miR-301a-3p regulates airway smooth muscle cells during asthma by targeting STAT3. J Asthma Allergy. (2022) 15:99–110. doi: 10.2147/JAA.S335680
222. Mariñas-Pardo L, Mirones I, Amor-Carro O, Fraga-Iriso R, Lema-Costa B, Cubillo I, et al. Mesenchymal stem cells regulate airway contractile tissue remodeling in murine experimental asthma. Allergy. (2014) 69:730–40. doi: 10.1111/all.2014.69.issue-6
223. Han XP, Zhang FQ, Tan XS, Liu L, Ma WX, Ou-Yang HF, et al. EPO modified MSCs can inhibit asthmatic airway remodeling in an animal model. J Cell Biochem. (2018) 119:1008–16. doi: 10.1002/jcb.v119.1
224. Ogulur I, Gurhan G, Aksoy A, Duruksu G, Inci C, Filinte D, et al. Suppressive effect of compact bone-derived mesenchymal stem cells on chronic airway remodeling in murine model of asthma. Int Immunopharmacol. (2014) 20:101–9. doi: 10.1016/j.intimp.2014.02.028
225. Lin HY, Xu L, Xie SS, Yu F, Hu HY, Song XL, et al. Mesenchymal stem cells suppress lung inflammation and airway remodeling in chronic asthma rat model via PI3K/Akt signaling pathway. Int J Clin Exp Pathol. (2015) 8:8958–67.
226. Agache I. Severe asthma phenotypes and endotypes. Semin Immunol. (2019) 46:101301. doi: 10.1016/j.smim.2019.101301
227. Ray A and Kolls JK. Neutrophilic inflammation in asthma and association with disease severity. Trends Immunol. (2017) 38:942–54. doi: 10.1016/j.it.2017.07.003
228. Shao JS, Lai AC, Huang WC, Wu KC, Chi PY, Chang YM, et al. Pulmonary fibroblast-derived stem cell factor promotes neutrophilic asthma by augmenting IL-17A production from ILC3s. J Clin Invest. (2025) 27(16):e187372. doi: 10.1172/JCI187372
229. Tang H, Li Z, Yang C, Fu L, Ji X, Chen Z, et al. CTSS contributes to airway neutrophilic inflammation in mixed granulocytic asthma. Respir Res. (2024) 25:441. doi: 10.1186/s12931-024-03077-6
230. Deng K, Zhang X, Liu Y, Zhang L, Wang G, Feng M, et al. Heterogeneity of paucigranulocytic asthma: A prospective cohort study with hierarchical cluster analysis. J Allergy Clin Immunol Pract. (2021) 9:2344–55. doi: 10.1016/j.jaip.2021.01.004
231. Huang H, Jiang X, Du Q, Liao H, Cai S, and Dong H. Circadian clock disruption and non-type 2 asthma: A hypothesis-driven perspective on immune, epithelial, and steroid response. Clin Rev Allergy Immunol. (2025) 68:72. doi: 10.1007/s12016-025-09088-5
232. Serajeddini H, Thakar A, Kjarsgaard M, Huang C, Ragunayakam N, Sandhu E, et al. Sputum cellularity and MRI ventilation defects in severe asthma. Chest. (2025). doi: 10.1016/j.chest.2025.05.013
233. Mihlan M, Wissmann S, Gavrilov A, Kaltenbach L, Britz M, Franke K, et al. Neutrophil trapping and nexocytosis, mast cell-mediated processes for inflammatory signal relay. Cell. (2024) 187:5316–5335.e28. doi: 10.1016/j.cell.2024.07.014
234. Baiardini I, Braido F, Brandi S, Tarantini F, Bonini S, Bousquet PJ, et al. The impact of GINA suggested drugs for the treatment of asthma on Health-Related Quality of Life: a GA(2)LEN review. Allergy. (2008) 63:1015–30. doi: 10.1111/j.1398-9995.2008.01823.x
235. Schleich FN, Zanella D, Stefanuto PH, Bessonov K, Smolinska A, Dallinga JW, et al. Exhaled volatile organic compounds are able to discriminate between neutrophilic and eosinophilic asthma. Am J Respir Crit Care Med. (2019) 200:444–53. doi: 10.1164/rccm.201811-2210OC
236. Zulueta A, Colombo M, Peli V, Falleni M, Tosi D, Ricciardi M, et al. Lung mesenchymal stem cells-derived extracellular vesicles attenuate the inflammatory profile of Cystic Fibrosis epithelial cells. Cell Signal. (2018) 51:110–8. doi: 10.1016/j.cellsig.2018.07.015
237. Fang SB, Zhang HY, Jiang AY, Fan XL, Lin YD, Li CL, et al. Human iPSC-MSCs prevent steroid-resistant neutrophilic airway inflammation via modulating Th17 phenotypes. Stem Cell Res Ther. (2018) 9:147. doi: 10.1186/s13287-018-0897-y
238. Sharan J, Barmada A, Band N, Liebman E, and Prodromos C. First report in a human of successful treatment of asthma with mesenchymal stem cells: A case report with review of literature. Curr Stem Cell Res Ther. (2023) 18:1026–9. doi: 10.2174/1574888X18666221115141022
239. Wang S, Lei B, Zhang E, Gong P, Gu J, He L, et al. Targeted therapy for inflammatory diseases with mesenchymal stem cells and their derived exosomes: from basic to clinics. Int J Nanomedicine. (2022) 17:1757–81. doi: 10.2147/IJN.S355366
240. Baker KF and Isaacs JD. Prospects for therapeutic tolerance in humans. Curr Opin Rheumatol. (2014) 26:219–27. doi: 10.1097/BOR.0000000000000029
Keywords: asthma, inflammation, mesenchymal stem cells, exosomes, immunomodulation, cell-based therapy
Citation: Hou Z-y, Hao Y-q, Zhang L, Li W and Gao P (2025) New insights into mesenchymal stem cells in inflammatory subtypes of asthma. Front. Immunol. 16:1649597. doi: 10.3389/fimmu.2025.1649597
Received: 18 June 2025; Accepted: 07 August 2025;
Published: 26 August 2025.
Edited by:
Frédéric Velard, Université de Reims Champagne-Ardenne, FranceReviewed by:
Liyang Dong, Affiliated Hospital of Jiangsu University, ChinaNaila Arif Cheema, University of Verona, Italy
Copyright © 2025 Hou, Hao, Zhang, Li and Gao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Peng Gao, Z2FvcGVuZzEyMzRAc2luYS5jb20=
†These authors have contributed equally to this work
 Zheng-yan Hou
Zheng-yan Hou Yu-qiu Hao†
Yu-qiu Hao† Wei Li
Wei Li Peng Gao
Peng Gao