- 1Department of General Surgery, Zigong Fourth People's Hospital, Zigong, Sichuan, China
- 2Division of Abdominal Tumor Multimodality Treatment, Cancer Center, West China Hospital, Sichuan University, Chengdu, Sichuan, China
Pancreatic cancer is a leading cause of cancer-related mortality, and unfortunately, the prognosis for this stubborn tumor type has remained bleak for an extended period. Current treatment options are limited and often ineffective, highlighting the need for innovative therapeutic strategies. Research in tumor immunotherapy has advanced significantly over the past few decades, leading to numerous studies progressing to clinical evaluation. Cancer vaccines are regarded as a promising therapeutic approach for treating pancreatic cancers. Cancer vaccines offer distinct advantages, especially for patients who are unresponsive to other treatments, as they can elicit broad and long-lasting T cell responses. This review offers a thorough examination of recent preclinical and clinical results of therapeutic cancer vaccines in pancreatic cancer therapy, highlighting the recent advancements in vaccine delivery platforms. Despite ongoing clinical efforts to develop cancer vaccines, there has not been a breakthrough in clinical outcomes, primarily due to substantial challenges including the low mutation burdens, the assessment of immune responses in preclinical models, and the intrinsic resistances to cancer vaccines. By tackling these challenges, cancer vaccines can expand the possibilities of personalized immunotherapy for pancreatic cancer.
1 Introduction
Pancreatic cancer is typically a fatal disease characterized by resistant and chronic nature (1). According to the global cancer statistics, there were 511,000 new cases of pancreatic cancer and approximately 467,000 deaths, making it the sixth leading cause of cancer-related mortality worldwide (2). The incidence and mortality rates of pancreatic cancer are influenced by various risk factors, including long-term smoking, obesity, chronic pancreatitis, and concurrent diabetes (3). Pancreatic ductal adenocarcinoma (PDAC) is the main type of pancreatic cancer, and approximately 50-60% of PDAC patients present with distant metastases, and 5-year survival rate is approximately 13%, which is attributed to its early systemic dissemination and aggressive local growth (4). The current treatment strategy for advanced pancreatic cancer consists of standard chemotherapy; however, it only presents a minor survival advantage due to the desmoplastic and chemo-resistant features (5, 6).
Immunotherapy leverages the patient’s immune system to target and destroy tumor cells, leading to a transformative effect on the treatment of human cancers (7). Immunomodulators, immune checkpoint inhibitor (ICI), and adoptive cell therapy provide promising strategies for solider tumors, including melanoma, lung cancer, colorectal cancer, and liver cancer (8–11). Early-phase trials assessing single-agent CTLA-4 or PD-1 blockade in PDAC showed little clinical activity, with no objective responses observed (12). Adoptive cell therapy (ACT) represents a novel type of immunotherapy that involves isolating a patient’s immune cells and genetically engineering them to mount tumor−specific responses (13). In pancreatic cancer, active investigations include chimeric antigen receptor T cells (CAR−T), CAR−NK cells, tumor−infiltrating lymphocytes (TILs), T−cell receptor (TCR)–engineered T cells, and cytokine−induced killer (CIK) cells. However, translation to solid tumors, particularly pancreatic cancer, remains early, and clinical outcomes to date have been limited (13–15). These unfavorable results are likely driven by PDAC’s profoundly immunosuppressive tumor microenvironment and intrinsically poor immunogenicity that limit the efficacy of immunotherapies (16, 17). Therefore, there is growing interest in investigating innovative immunotherapeutic strategies, such as therapeutic cancer vaccines, to tackle these challenges posed by the hostile TME. The objective of therapeutic cancer vaccines is to stimulate the generation of effector T cells, leading to the establishment of a lasting immune response against specific tumor antigens and eradicating cancerous cells (18, 19). In contrast to conventional monoclonal antibodies and small molecule inhibitors, cancer vaccines provide various benefits, such as reduced nonspecific effects, a wide therapeutic index, and the durable immunological memory (20, 21). Additionally, cancer vaccines enable precise targeting tailored to the specific traits of individual tumors (22, 23). As a result, vaccination represents a promising strategy for personalized treatment of pancreatic cancer, addressing the complexities introduced by tumor heterogeneity.
In this review, we offer an extensive overview of the current landscape of therapeutic cancer vaccines and their clinical applications in pancreatic cancer. Furthermore, we discuss the resistance mechanisms that tumors present and assess the potential benefits of combination therapies, which may help to enhance the effectiveness of cancer vaccines in the treatment of pancreatic cancer. Moreover, we outline their advantages, limitations, and challenges posed by vaccine technology and provide future perspectives on developing therapeutic vaccine in pancreatic cancer.
2 Clinical landscape of cancer vaccine in pancreatic cancer immunotherapy
After vaccination, innate immune cells such as natural killer (NK) cells, neutrophils, and macrophages rapidly identify foreign substances through pattern recognition receptors (PRRs), initiating specific immune responses (24). Distinct dendritic cells (DCs) subsets differentially prime defined T-cell lineages. With maturation, DCs traffic to secondary lymphoid tissues, particularly the draining lymph nodes (25). DCs capture and process peptides in peripheral tissues and present them via major histocompatibility complex (MHC) molecules. Endogenous proteins activate CD8+ T cells, while exogenous proteins are presented to CD4+ T cells as antigen-MHC-II complexes (26). APC-mediated activation of CD8+ cytotoxic T lymphocytes (CTLs) exit lymphoid organs, infiltrate tumors, and exert anti-tumor effects by inducing apoptosis in tumor cells through granule exocytosis (involving perforin and granzymes) and engagement of death receptors (such as Fas ligand and tumor necrosis factor-related apoptosis-inducing ligand) (27, 28). Therefore, the intricate interactions among immune components significantly influence the efficacy of cancer vaccines (Figure 1). Established delivery platforms for vaccines include, DC, tumor cell, nucleic acid and peptide vaccines, while emerging technologies are also under investigation (29, 30). In this section, we summarize current landscape of cancer vaccines in pancreatic cancer (Table 1).
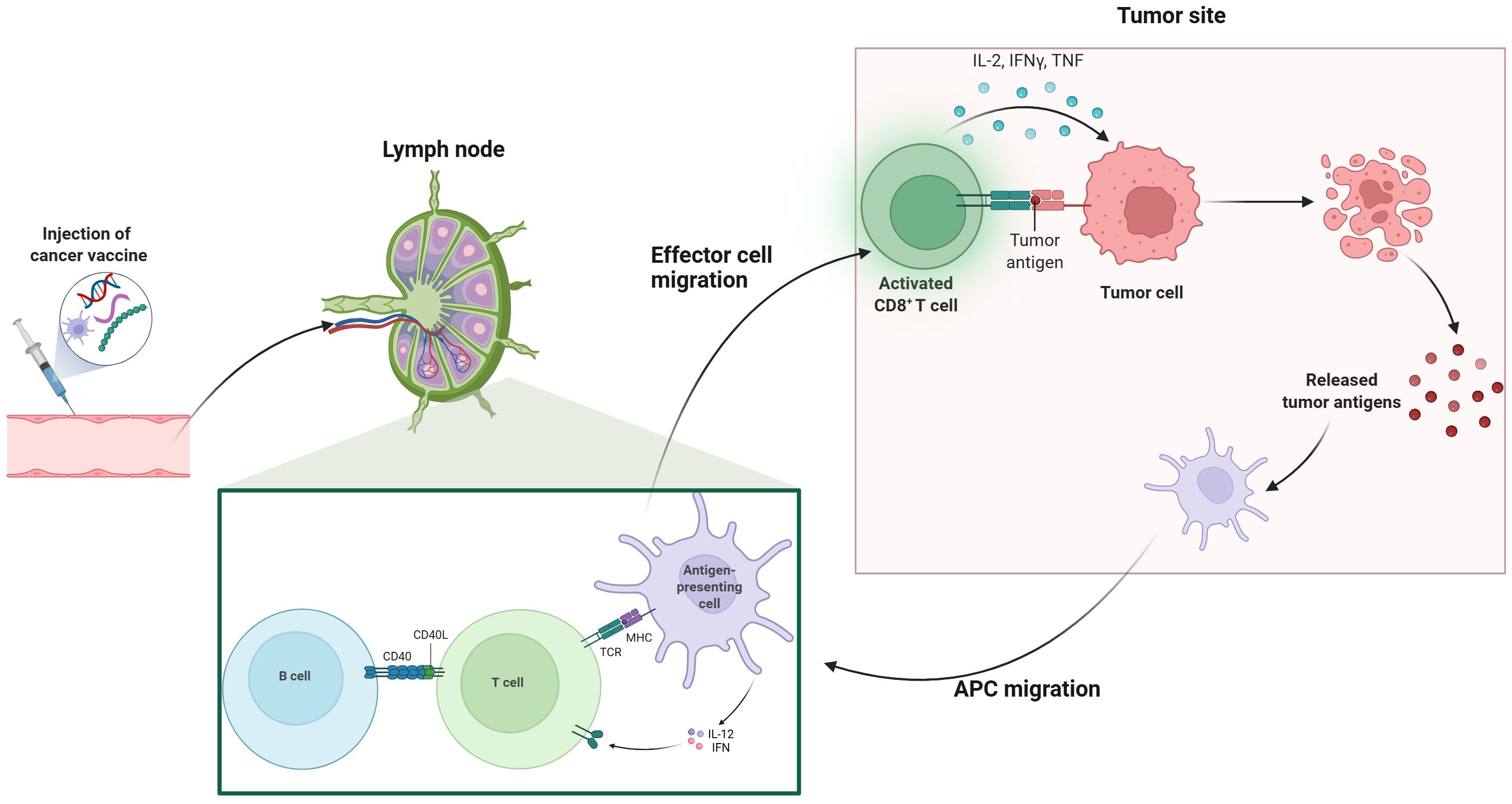
Figure 1. Mechanism of cancer vaccines. This illustration depicts the process of cancer vaccination and subsequent immune response activation. The series begins with the injection of a cancer vaccine, which then leads to the activation of T cells in the lymph nodes. Effector T cells migrate to the tumor site, where they recognize and target tumor antigens on cancer cells. Activated CD8+ T cells release cytokines such as IL-2, IFN-γ, and TNF, which enhance the immune response. Antigen-presenting cells (APCs) also migrate to the tumor site, facilitating further activation of B cells and T cells through interactions with CD40 and MHC-TCR complexes. This coordinated immune response aims to eliminate tumor cells and promote the release of additional tumor antigens, potentially enhancing the overall effectiveness of cancer immunotherapy.
2.1 Cell-based vaccine
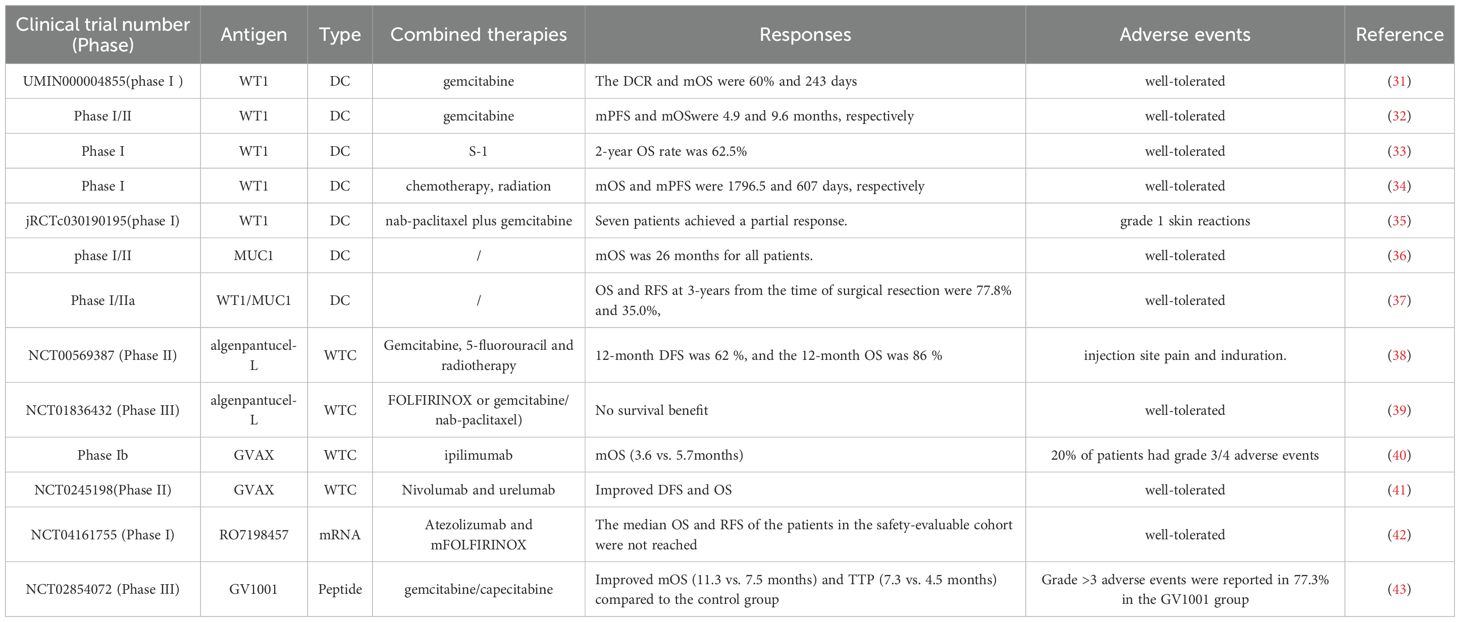
Cell-based cancer vaccines utilize the patient’s own immune cells, particularly DCs, to process both soluble and particulate antigens (44). Additionally, tumor cells are recognized as valuable antigen sources for vaccines, presented in multiple formats, including whole tumor cells (WTC) and tumor cell lysates (45). Both DC and WTC-based vaccines are evaluated in the clinical trials of pancreatic cancer. DCs play a crucial role in the immune system by effectively presenting both exogenous and endogenous antigens to T cells, thereby triggering adoptive immune responses (46). By showcasing tumor-specific antigens, DCs can trigger the generation of cytotoxic T lymphocytes that specifically recognize and destroy cancer cells (47). DCs are specialized APCs that are highly effective in generating robust immune responses and maintaining tolerance to self or benign foreign antigens, making DCs an appealing tool for developing immunotherapeutic strategies (48). Since the study has assessed the efficacy of DCs pulsed with melanoma-associated antigen (MAGE) 1 for treating melanoma patients, emerging evidence has demonstrated treatments based on DCs are safe, even in patients with advanced cancer (49). Additionally, DC-based therapies can enhance immune responses that lead to sustained remissions, providing optimism for enduring outcomes (50). The Wilms’ tumor 1 (WT1) antigen has been recognized as a highly effective target in various cancer types, including pancreatic cancer (51). WT1-peptide pulsed dendritic cell (WT1-DC) vaccine combined with gemcitabine chemotherapy resulted in durable specific T cell immune responses, which were associated with significant improvements in survival in advanced PDAC patients (31–35). Further studies broadened the insights into the potential clinical application of WT1-DC vaccines as evidenced by vaccine injection combined with chemotherapy exhibited safety and effectiveness in resected PDAC patients (52). And WT1-DC vaccine in conjunction with multimodal treatments including chemotherapy, irradiation and surgery was related with longer overall survival time (53). Moreover, the chemoimmunotherapy regimen consisted of nab-paclitaxel plus gemcitabine combined with WT1-DC vaccination regulated TME and facilitated conversion surgery for advanced pancreatic cancer patients (54). A chemoimmunotherapy approach combining a WT1−DC vaccine with multi−agent chemotherapy was observed to reprogram the TME toward an immunostimulatory phenotype, permitting conversion surgery in 7 of 9 patients with unresectable pancreatic ductal adenocarcinoma (55). Although early observations are encouraging and informative, definitive clarification of the treatment effect attributable to WT1−targeted chemoimmunotherapy will require large, well−controlled studies with appropriate comparators (55).
Mucin 1 (MUC1) was another tumor-associated antigen presented in pancreatic cancer cells, considering a promising antigen for immunotherapy (56, 57). The MUC1 peptide-loaded DC vaccine was elevated in patients after the surgical resection, which extended the medial survival time of patients to 26 months and exhibited favorable safety (58, 59). Pan et al. further modified the DC vaccines by loading the MUC1-PD-L1 immunogen (MUC1-Vax) that consist of a fusion gene that incorporated the extracellular domain of human MUC1 along with the programmed cell death ligand 1 (PD-L1), which demonstrated robust therapeutic effects against tumors in mice, indicating a promising strategy for treating pancreatic cancer by double-targeting MUC1 and PD-L1 (36). This innovation vaccine strategy should be further assessed in the clinical settings, further validating the safety and efficiencies in cancer patients. Similarly, Nagai et al. revealed that WT1/MUC1-DC vaccination extended the 3-year overall survival rate to 77.8% in the adjuvant setting of pancreatic cancer (37), which awaits to be assessed its clinical advantages in a large-scale trial. There are various approaches to load tumor antigens into DC vaccines, with earlier studies primarily focusing on the use of mRNA for this purpose (60). Utilizing circular RNA (circRNA) for antigen loading represents a promising alternative, as its inherent stability prolongs the duration of protein translation and enhances protein production compared to linear mRNA (61). Recent study utilized circRNA loaded FAPα and surviving(circRNAFS) and then transfected circRNAFS into DCs for vaccination (62). This vaccine combined with chemotherapy elicited significant immunogenic cell deaths and overcame immune evasion in Panc02 tumor model, which provided a novel insight into the clinical management of pancreatic cancer (62).
WTC vaccines represent a form of cancer immunotherapy that employs whole or lysed tumor cells, whether unaltered or modified, as a source of immunogenic components to elicit an anti-tumor response (63). Algenpantucel-L is an allogeneic pancreatic cancer vaccine composed of two human PDAC cell lines that express α-galactosyl (αGal) through retroviral transfer of the mouse αGT gene (38). This vaccine was evaluated as an adjunct to chemotherapy and radiotherapy in a study involving 70 patients who underwent surgical resection for PDAC and the inclusion of Algenpantucel-L resulted in improved disease-free survival (DFS) and overall survival (OS) (38). However, subsequent phase III trials did not validate the previous findings (39). To stimulate T-cell immune responses targeting different tumor antigens, researchers developed a pancreatic cancer vaccine known as GVAX, which was categorized as allogeneic and was composed of WTCs that secrete human GM-CSF (40, 64, 65). Furthermore, researchers revealed that the combination of GVAX, nivolumab, and urelumab (anti-CD137 agonist antibody) significantly elevated the levels of intratumoral cytotoxic T cells, demonstrating efficacy in PDAC patients undergoing neoadjuvant and adjuvant therapy (41). Recently, a clinical trial offered new insights into GVAX therapy, as the combination of the adjuvant GVAX vaccine and chemoradiotherapy showed good tolerance and moderate survival outcomes in PDAC patients, pending further validation in future studies (66). VIReST vaccine was developed by using pancreatic tumor cells derived from gene-edited induced pluripotent stem cells that were infected by viruses, which stimulated T cell immune responses, leading to a delay in malignant onset and progression in KPC transgenic mice (67), offering an innovation technological foundation for creating personalized cancer vaccines in high-risk population.
2.2 Nucleic acid-based vaccine
Nucleic acid-based cancer vaccines, encompassing both DNA and RNA formulations, signify a groundbreaking advancement in vaccination technology (68). These vaccines have garnered significant interest due to their accuracy, adaptability, and ease of production, positioning them as promising candidates for the development of personalized cancer vaccines (69). α-Enolase, known as ENO1, serves as an enzyme in the glycolytic pathway that was overexpressed in several cancers, including pancreatic cancer (70). Targeting ENO1 with monoclonal antibodies or silencing its expression could inhibit the migration of PDAC cells (71, 72). Researchers have developed ENO1 DNA vaccines that could prolong the survival of genetically engineered mice by eliciting cellular immune responses against ENO1 (73). However, the ENO1 DNA vaccine has no effect on the elimination of tumor; especially in the presence of phosphoinositide-3-kinaseγ (PI3Kγ) -mediated myeloid-derived suppressor cells (MDSC) recruitment within the TME (74, 75). Based on these findings, targeting MDSC through PI3Kγ inhibition in conjunction with ENO1 DNA vaccination could work synergistically to combat tumor growth in a B-cell-dependent immune response (76). Moreover, administering gemcitabine before ENO1 DNA vaccination activated significantly hindered tumor progression compared to mice that received either the vaccine or chemotherapy treatment alone, indicating the potential role of chemoimmunotherapy in pancreatic cancer therapy (77). Melanoma associated antigen A isoforms (MAGEA) played an essential role in modulating the interactions between tumor cells and stromal cells in PDAC (78, 79). Notably, vaccination with a MAGEA DNA vaccine targeting MAGEA2 and MAGEA10, could induce a robust immune response in chemotherapy-resistant mice (80). Administration of OsFS DNA vaccine that targeted human FAPα and survivin, significantly remodeling the immunosuppressive TME to impede tumor growth in PDAC (81). Furthermore, low-dose gemcitabine treatment could amplify the anti-tumor responses of OsFS, representing a promising approach for PDAC therapy (81). MUC1 DNA vaccines also exhibited potent anti-tumor effects on pancreatic cancer experimental models, which await further investigation in the clinical setting (82, 83). VVL−DD is a tumor−selective viral vaccine that demonstrated marked tumor selectivity in vitro and showed antitumor efficacy in a murine pancreatic cancer model in vivo (84).
Although advancements in DNA vaccines, however, DNA must first enter the nucleus, enabling extended production of target proteins, which arising a safe concern that integrating into the host genome (85, 86). Conversely, mRNA can be directly translated in the cytoplasm using the host cell’s machinery, which hold promise for developing cancer vaccine (87). The application of lipid nanoparticle (LNP) systems for mRNA vaccines, especially in the context of targeting personalized neoantigens, has emerged as a key area of interest (88, 89). Autogene cevumeran was an innovative mRNA vaccine that included RO7198457 and featured up to 20 patient-specific neoantigens intravenously delivered by LNPs (90), in combination with atezolizumab, and mFOLFIRINOX chemotherapy promoted significant T cell-mediated immune responses and linked to a delay in the recurrence of PDAC in the adjuvant setting (42). Follow-up results demonstrated that autogene cevumeran induced the generation of novel CD8+ T cells post-vaccination, which exhibited lasting effector functions and a memory-like T cell state for up to three years, potentially delaying the recurrence of PDAC (91). Currently, a global randomized trial (NCT05968326) is underway. Consequently, these results revealed that adjuvant mRNA-LNP neoantigen vaccines may address a significant challenge in pancreatic cancer vaccination.
2.3 Peptide-based vaccine
The peptide vaccine GV1001 consists of 16 amino acids sourced from the catalytic subunit of human telomerase reverse transcriptase (hTERT) (92). GV1001 elicited robust CD4+ and CD8+ T cell responses, as well as recognition by APCs (93). In earlier Phase I/II trials, T cell responses specific to GV1001 were observed in 50-80% of patients with PDAC who had an improved median survival (94). Notably, in patients with advanced PDAC who have high levels of eotaxin, the combination of GV1001 with chemotherapy resulted in improved OS and time to progression compared to patients treated with chemotherapy alone (43). A vaccine therapy employing three HLA-A2402-restricted peptides has shown promising effectiveness in clinical settings for treating patients with advanced PDAC (95). Recently, Cai et al. illustrated that targeting mesothelin (MSLN) could enhance the immune efficacy of neoantigen vaccines by diminishing cancer-associated fibroblasts (CAFs), which interrupted the conversion of naive CD4+ T cells into regulatory T cells, ultimately boosting t anti-tumor immunity (96). In line with this, VASH2-peptide vaccine, TM4SF5 peptide vaccine, and TGF-β- multipeptide vaccination inhibited PDAC progression in the preclinical studies (97–99), which await further validation in clinical trials. Moreover, ELI−002 2P vaccine employed Amph−modified mutant KRAS peptides (G12D, G12R; Amph−Peptides−2P) plus the CpG adjuvant Amph−CpG−7909 to optimize lymph−node delivery and heighten immunogenicity (100). In the phase 1 AMPLIFY-201 study, 84% of vaccinated pancreatic cancer patients mounted T-cell responses, tumor biomarkers declined in six individuals, and the median relapse-free survival (RFS) reached 16.33 months. T-cell reactivity predicted decreases in tumor biomarkers and ctDNA clearance, and was strongly associated with an 86% reduction in the risk of recurrence or death (101). ELI−002 exhibited potent therapeutic activity; however, further investigation is warranted.
3 Challenges in developing cancer vaccines
3.1 Low mutation burdens
Various factors related to the biology of PDAC that are unresponsive to immunotherapy, with hostile TME considered a key player in this process (102). This environment is highly immunosuppressive, marked by a low mutational burden and lack enough neoantigens (103). PDAC exhibit modern burdens in on-synonymous neo-antigenic mutations, leading to an absence of effective neoantigens (104). Neoantigens hold potential for enhancing personalized immunotherapy approaches for pancreatic cancer. A significant portion of neoantigens has been found to originate from atypical translation processes, which require further investigation to identify shared epitopes (105). Advancements in next-generation sequencing and developments in computational algorithms could uncover those antigens capable of effectively activating patients’ T cells, enabling their assessment in clinical studies, which may help overcome these limitations (106, 107). Moreover, immunosuppressive cells and factors present in the TME can hinder the anti-tumor immune response. Thus, developing effective cancer vaccines necessitates the implementation of strategies aimed at overcoming these immunosuppressive elements.
3.2 Technological obstacles in developed cancer vaccines
The technologies in developing pancreatic cancer vaccines are still in the early stages that pose multiple challenges. Compared to personalized neoantigen vaccines, public neoantigen vaccines can be manufactured in large quantities for immediate use, which reduces production time and lowers costs (108). However, a significant challenge is that the low number of shared neo-antigens among PDAC patients complicates the implementation of relevant treatment strategies, making them cumbersome and expensive (19, 109). To overcome the challenges, it is essential to implement standardized synthetic vaccine technologies that enable fully automated production and quality control, facilitating the establishment of efficient, miniaturized production lines (110). This approach enhances the overall efficiency and accessibility of vaccines for clinical applications in pancreatic cancer.
3.3 Lack of optimal preclinical models
Exploring the interactions and evaluating changes in immunological phenotype following anti-tumor treatments are considerably challenges due to the complexity and heterogeneity present in the TME of pancreatic tumors. Additionally, the weak immunogenicity of PDAC and its immunosuppressive characteristics complicate the development of animal models (111, 112). Genetically engineered preclinical models, created through gene modification and the introduction of mutations, represent the disease most accurately. However, these models cannot replicate the gradual accumulation of mutations seen in human pancreatic cancers, resulting in tumors that are relatively stable and less responsive to cancer vaccines (113, 114). These shortcomings highlight the necessity of integrating findings from animal models with clinical investigations to achieve a thorough understanding of immunotherapeutic approaches. Leveraging high-throughput methodologies to advance pancreatic cancer animal models will enable more faithful recapitulation of human disease features, thereby expediting the translation of preclinical discoveries into clinical practice.
3.4 Immunotherapeutic resistance mechanisms
The interplay between cancer and the immune system is intricate and dynamic. Adaptive immune resistance mechanisms enable tumors to protect themselves by adapting to immune attacks, even though they are recognized by the immune system (115). In PDAC driven by mutant KRAS (mKRAS), the downstream signaling pathways play a crucial role in immune evasion (116). For example, mKRAS facilitated the accumulation of MDSCs, CAFs and macrophages that inhibited cytotoxic T cell activity, as evidenced by autochthonous humanized mouse models of PDAC (117). Additionally, certain oncogenic pathways could induce T cell exclusion, allowing pancreatic tumors to evade immune attack. The activation of Wnt signaling pathway facilitated tumor development via upregulating expression of checkpoint molecules on T cells and inducing T cell exclusion, which contributed to immune escape (118). Furthermore, the loss of PTEN was linked to the activation of the PI3K-AKT signaling pathway and was associated with poor clinical responses to immunotherapy (119). While cancer vaccines could regulate TME, they often do not completely eliminate tumors when administered as standalone treatments due to these resistance mechanisms in PDAC (120, 121). Importantly, cancer vaccines can initiate epitope spreading, thereby expanding T cell responses and potentially improving the effectiveness of combination therapies (122). By leveraging the advantages of various treatment approaches, combination strategies can address the shortcomings of monotherapy and create a holistic method for targeting pancreatic tumors (123, 124).
4 Conclusions and perspectives
In recent decades, the recognitions on molecular mechanism of tumor cells evading immune detection have significantly improved, leading to notable advancements in cancer vaccines by mimicking natural immunity. With the advancements of sequencing technologies, the development of personalized cancer vaccines is likely to progress rapidly, which may revolutionize the therapeutic landscape of PDAC (125). Additionally, the careful selection of delivery systems is crucial for enhancing the immunotherapeutic responses (126). Recent technological advancements have introduced promising carriers, such as vaccines based on nanomaterials, which offer safer and durable immune responses (127). The integration of innovative strategies, meticulous candidate selection, and enhanced administration protocols has the potential to revolutionize cancer treatment, heralding a new era of therapeutic cancer vaccines. This progress ultimately paves the way for the effective use of cancer vaccines in the management of pancreatic cancer.
Author contributions
YW: Formal Analysis, Investigation, Methodology, Project administration, Resources, Writing – original draft, Writing – review & editing. PH: Conceptualization, Methodology, Project administration, Validation, Writing – review & editing. CL: Conceptualization, Methodology, Validation, Visualization, Writing – review & editing. ST: Formal Analysis, Investigation, Supervision, Validation, Writing – review & editing. HY: Funding acquisition, Investigation, Methodology, Resources, Validation, Visualization, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. This research was supported by Science & Technology Department of Sichuan Province Funding Project (2025ZNSFSC0549).
Acknowledgments
We thank the BioRender drawing software (https://www.biorender.com/).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Stoop TF, Javed AA, Oba A, Koerkamp BG, Seufferlein T, Wilmink JW, et al. Pancreatic cancer. Lancet. (2025) 405:1182–202. doi: 10.1016/s0140-6736(25)00261-2
2. Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. (2024) 74:229–63. doi: 10.3322/caac.21834
3. Klein AP. Pancreatic cancer epidemiology: understanding the role of lifestyle and inherited risk factors. Nat Rev Gastroenterol Hepatol. (2021) 18:493–502. doi: 10.1038/s41575-021-00457-x
4. Siegel RL, Kratzer TB, Giaquinto AN, Sung H, and Jemal A. Cancer statistics, 2025. CA Cancer J Clin. (2025) 75:10–45. doi: 10.3322/caac.21871
5. Beutel AK and Halbrook CJ. Barriers and opportunities for gemcitabine in pancreatic cancer therapy. Am J Physiol Cell Physiol. (2023) 324:C540–c552. doi: 10.1152/ajpcell.00331.2022
6. Park SJ, Yoo HC, Ahn E, Luo E, Kim Y, Sung Y, et al. Enhanced Glutaminolysis Drives Hypoxia-Induced Chemoresistance in Pancreatic Cancer. Cancer Res. (2023) 83:735–52. doi: 10.1158/0008-5472.Can-22-2045
7. Bergman PJ. Cancer Immunotherapy. Vet Clin North Am Small Anim Pract. (2024) 54:441–68. doi: 10.1016/j.cvsm.2023.12.002
8. Baulu E, Gardet C, Chuvin N, and Depil S. TCR-engineered T cell therapy in solid tumors: State of the art and perspectives. Sci Adv. (2023) 9:eadf3700. doi: 10.1126/sciadv.adf3700
9. Chan JD, Lai J, Slaney CY, Kallies A, Beavis PA, and Darcy PK. Cellular networks controlling T cell persistence in adoptive cell therapy. Nat Rev Immunol. (2021) 21:769–84. doi: 10.1038/s41577-021-00539-6
10. Lahiri A, Maji A, Potdar PD, Singh N, Parikh P, Bisht B, et al. Lung cancer immunotherapy: progress, pitfalls, and promises. Mol Cancer. (2023) 22:40. doi: 10.1186/s12943-023-01740-y
11. Li J, Smalley I, Chen Z, Wu JY, Phadke MS, Teer JK, et al. Single-cell Characterization of the Cellular Landscape of Acral Melanoma Identifies Novel Targets for Immunotherapy. Clin Cancer Res. (2022) 28:2131–46. doi: 10.1158/1078-0432.Ccr-21-3145
12. Formelli MG, Palloni A, Tavolari S, Deiana C, Andrini E, Di Marco M, et al. Classic versus innovative strategies for immuno-therapy in pancreatic cancer. Adv Drug Delivery Rev. (2025) 225:115671. doi: 10.1016/j.addr.2025.115671
13. Sherpally D and Manne A. Advancing Immunotherapy in Pancreatic Cancer: A Brief Review of Emerging Adoptive Cell Therapies. Cancers (Basel). (2025) 17. doi: 10.3390/cancers17040589
14. Hegazi A, Rager LE, Watkins DE, and Su KH. Advancing Immunotherapy in Pancreatic Cancer. Int J Mol Sci. (2024) 25. doi: 10.3390/ijms252111560
15. Aznar MA, Good CR, Barber-Rotenberg JS, Agarwal S, Wilson W, Watts A, et al. Clinical and molecular dissection of CAR T cell resistance in pancreatic cancer. Cell Rep Med. (2025) 6:102301. doi: 10.1016/j.xcrm.2025.102301
16. Yang H, Zhang X, Zhang S, Yang Y, Chen Y, Jiang Y, et al. Targeting spermine metabolism to overcome immunotherapy resistance in pancreatic cancer. Nat Commun. (2025) 16:7827. doi: 10.1038/s41467-025-63146-2
17. Xie W, Yu X, Yang Q, Ke N, Wang P, Kong H, et al. The Immunomechanical Checkpoint PYK2 Governs Monocyte-to-Macrophage Differentiation in Pancreatic Cancer. Cancer Discovery. (2025) 15:1740–65. doi: 10.1158/2159-8290.Cd-24-1712
18. Duan Y, Zhan W, Yao J, Zhang L, Ge Y, Zhou Y, et al. RAE1-armoured DC vaccine boosts NKG2D-CAR-T cells elicited anti-solid tumour treatment. Pharmacol Res. (2025) 219:107867. doi: 10.1016/j.phrs.2025.107867
19. Peng K, Zhao X, Fu YX, and Liang Y. Eliciting antitumor immunity via therapeutic cancer vaccines. Cell Mol Immunol. (2025). doi: 10.1038/s41423-025-01316-4
20. Liu R, Chen Y, Xu X, Xue Q, and Gu H. Identification of esophageal cancer tumor antigens and immune subtypes for guiding vaccine development. J Thorac Dis. (2025) 17:3380–99. doi: 10.21037/jtd-2025-233
21. Ren C, He D, and Wang Q. Identification and Experimental Validation of Tumor Antigens and Hypoxia Subtypes of Osteosarcoma for Potential mRNA Vaccine Development. Curr Med Chem. (2025). doi: 10.2174/0109298673348041250407062346
22. Sandstad VBB, Modvig S, and Holmström MO. Neo-antigen specific cancer vaccines for acute lymphoblastic leukemia-challenges, opportunities, and future directions. Cancer Immunol Immunother. (2025) 74:248. doi: 10.1007/s00262-025-04107-y
23. Zaidi N, Jaffee EM, and Yarchoan M. Recent advances in therapeutic cancer vaccines. Nat Rev Cancer. (2025) 25:517–33. doi: 10.1038/s41568-025-00820-z
24. Pishesha N, Harmand TJ, and Ploegh HL. A guide to antigen processing and presentation. Nat Rev Immunol. (2022) 22:751–64. doi: 10.1038/s41577-022-00707-2
25. Allan RS, Waithman J, Bedoui S, Jones CM, Villadangos JA, Zhan Y, et al. Migratory dendritic cells transfer antigen to a lymph node-resident dendritic cell population for efficient CTL priming. Immunity. (2006) 25:153–62. doi: 10.1016/j.immuni.2006.04.017
26. Liu J, Song X, Guo W, Liu W, Xu S, Yang Y, et al. Unlock the code of MHC II-enabled Cancer immunotherapy. Crit Rev Oncol Hematol. (2025) 214:104813. doi: 10.1016/j.critrevonc.2025.104813
27. Chopp L, Redmond C, O'Shea JJ, and Schwartz DM. From thymus to tissues and tumors: A review of T-cell biology. J Allergy Clin Immunol. (2023) 151:81–97. doi: 10.1016/j.jaci.2022.10.011
28. Farhood B, Najafi M, and Mortezaee K. CD8(+) cytotoxic T lymphocytes in cancer immunotherapy: A review. J Cell Physiol. (2019) 234:8509–21. doi: 10.1002/jcp.27782
29. Yu Y, Lyu J, Muhadaisi Y, Shi C, and Wang D. Peptide based vesicles for cancer immunotherapy: design, construction and applications. Front Immunol. (2025) 16:1609162. doi: 10.3389/fimmu.2025.1609162
30. Owliaee I, Shoushtari M, Karimi Z, and Shojaeian A. Natural carrier systems in cancer vaccines and immunotherapy. Hum Vaccin Immunother. (2025) 21:2535787. doi: 10.1080/21645515.2025.2535787
31. Tiwari A, Alcover K, Carpenter E, Thomas K, Krum J, Nissen A, et al. Utility of cell-based vaccines as cancer therapy: Systematic review and meta-analysis. Hum Vaccin Immunother. (2024) 20:2323256. doi: 10.1080/21645515.2024.2323256
32. Chiang CL, Coukos G, and Kandalaft LE. Whole Tumor Antigen Vaccines: Where Are We? Vaccines (Basel). (2015) 3:344–72. doi: 10.3390/vaccines3020344
33. Redkin T and Turubanova V. Dendritic cell-derived exosomes as anti-cancer cell-free agents: new insights into enhancing immunogenic effects. Front Immunol. (2025) 16:1586892. doi: 10.3389/fimmu.2025.1586892
34. Nikfarjam S, Rezaie J, Kashanchi F, and Jafari R. Dexosomes as a cell-free vaccine for cancer immunotherapy. J Exp Clin Cancer Res. (2020) 39:258. doi: 10.1186/s13046-020-01781-x
35. Anguille S, Smits EL, Lion E, van Tendeloo VF, and Berneman ZN. Clinical use of dendritic cells for cancer therapy. Lancet Oncol. (2014) 15:e257–267. doi: 10.1016/s1470-2045(13)70585-0
36. Constantino J, Gomes C, Falcão A, Cruz MT, and Neves BM. Antitumor dendritic cell-based vaccines: lessons from 20 years of clinical trials and future perspectives. Transl Res. (2016) 168:74–95. doi: 10.1016/j.trsl.2015.07.008
37. Zanotta S, Galati D, De Filippi R, and Pinto A. Enhancing Dendritic Cell Cancer Vaccination: The Synergy of Immune Checkpoint Inhibitors in Combined Therapies. Int J Mol Sci. (2024) 25. doi: 10.3390/ijms25147509
38. Koido S, Okamoto M, Kobayashi M, Shimodaira S, and Sugiyama H. Significance of Wilms' tumor 1 antigen as a cancer vaccine for pancreatic cancer. Discovery Med. (2017) 24:41–9.
39. Koido S, Homma S, Okamoto M, Takakura K, Gong J, Sugiyama H, et al. Chemoimmunotherapy targeting Wilms' tumor 1 (WT1)-specific cytotoxic T lymphocyte and helper T cell responses for patients with pancreatic cancer. Oncoimmunology. (2014) 3:e958950. doi: 10.4161/21624011.2014.958950
40. Nishida S, Koido S, Takeda Y, Homma S, Komita H, Takahara A, et al. Wilms tumor gene (WT1) peptide-based cancer vaccine combined with gemcitabine for patients with advanced pancreatic cancer. J Immunother. (2014) 37:105–14. doi: 10.1097/cji.0000000000000020
41. Katsuda M, Miyazawa M, Ojima T, Katanuma A, Hakamada K, Sudo K, et al. A double-blind randomized comparative clinical trial to evaluate the safety and efficacy of dendritic cell vaccine loaded with WT1 peptides (TLP0-001) in combination with S-1 in patients with advanced pancreatic cancer refractory to standard chemotherapy. Trials. (2019) 20:242. doi: 10.1186/s13063-019-3332-5
42. Mayanagi S, Kitago M, Sakurai T, Matsuda T, Fujita T, Higuchi H, et al. Phase I pilot study of Wilms tumor gene 1 peptide-pulsed dendritic cell vaccination combined with gemcitabine in pancreatic cancer. Cancer Sci. (2015) 106:397–406. doi: 10.1111/cas.12621
43. Ogasawara M, Miyashita M, Yamagishi Y, and Ota S. Dendritic cell vaccination combined with a conventional chemotherapy for patients with relapsed or advanced pancreatic ductal adenocarcinoma: a single-center phase I/II trial. Ther Apher Dial. (2021) 25:415–24. doi: 10.1111/1744-9987.13659
44. Yanagisawa R, Koizumi T, Koya T, Sano K, Koido S, Nagai K, et al. WT1-pulsed Dendritic Cell Vaccine Combined with Chemotherapy for Resected Pancreatic Cancer in a Phase I Study. Anticancer Res. (2018) 38:2217–25. doi: 10.21873/anticanres.12464
45. Hanada S, Tsuruta T, Haraguchi K, Okamoto M, Sugiyama H, and Koido S. Long-term survival of pancreatic cancer patients treated with multimodal therapy combined with WT1-targeted dendritic cell vaccines. Hum Vaccin Immunother. (2019) 15:397–406. doi: 10.1080/21645515.2018.1524238
46. Koido S, Taguchi J, Shimabuku M, Kan S, Bito T, Misawa T, et al. Dendritic cells pulsed with multifunctional Wilms' tumor 1 (WT1) peptides combined with multiagent chemotherapy modulate the tumor microenvironment and enable conversion surgery in pancreatic cancer. J Immunother Cancer. (2024) 12. doi: 10.1136/jitc-2024-009765
47. Koido S, Taguchi J, Shimabuku M, Bito T, Morimoto S, Oji Y, et al. Predictors of patients with advanced pancreatic cancer undergoing conversion surgery via chemoimmunotherapy with a multifunctional Wilms' tumor 1 (WT1) peptide cocktail-pulsed dendritic cell vaccine. J Immunother Cancer. (2025) 13. doi: 10.1136/jitc-2024-011426
48. Murthy D, Attri KS, Suresh V, Rajacharya GH, Valenzuela CA, Thakur R, et al. The MUC1-HIF-1α signaling axis regulates pancreatic cancer pathogenesis through polyamine metabolism remodeling. Proc Natl Acad Sci U.S.A. (2024) 121:e2315509121. doi: 10.1073/pnas.2315509121
49. Shukla SK, Purohit V, Mehla K, Gunda V, Chaika NV, Vernucci E, et al. MUC1 and HIF-1alpha Signaling Crosstalk Induces Anabolic Glucose Metabolism to Impart Gemcitabine Resistance to Pancreatic Cancer. Cancer Cell. (2017) 32:71–87.e77. doi: 10.1016/j.ccell.2017.06.004
50. Lepisto AJ, Moser AJ, Zeh H, Lee K, Bartlett D, McKolanis JR, et al. A phase I/II study of a MUC1 peptide pulsed autologous dendritic cell vaccine as adjuvant therapy in patients with resected pancreatic and biliary tumors. Cancer Ther. (2008) 6:955–64.
51. Pecher G, Häring A, Kaiser L, and Thiel E. Mucin gene (MUC1) transfected dendritic cells as vaccine: results of a phase I/II clinical trial. Cancer Immunol Immunother. (2002) 51:669–73. doi: 10.1007/s00262-002-0317-z
52. Pan J, Zeng W, Jia J, Shi Y, Wang D, Dong J, et al. A Novel Therapeutic Tumor Vaccine Targeting MUC1 in Combination with PD-L1 Elicits Specific Anti-Tumor Immunity in Mice. Vaccines (Basel). (2022) 10. doi: 10.3390/vaccines10071092
53. Nagai K, Adachi T, Harada H, Eguchi S, Sugiyama H, and Miyazaki Y. Dendritic Cell-based Immunotherapy Pulsed With Wilms Tumor 1 Peptide and Mucin 1 as an Adjuvant Therapy for Pancreatic Ductal Adenocarcinoma After Curative Resection: A Phase I/IIa Clinical Trial. Anticancer Res. (2020) 40:5765–76. doi: 10.21873/anticanres.14593
54. Beck JD, Reidenbach D, Salomon N, Sahin U, Türeci Ö, Vormehr M, et al. mRNA therapeutics in cancer immunotherapy. Mol Cancer. (2021) 20:69. doi: 10.1186/s12943-021-01348-0
55. Li H, Peng K, Yang K, Ma W, Qi S, Yu X, et al. Circular RNA cancer vaccines drive immunity in hard-to-treat malignancies. Theranostics. (2022) 12:6422–36. doi: 10.7150/thno.77350
56. Cai Z, Wuri Q, Song Y, Qu X, Hu H, Cao S, et al. CircRNA-loaded DC vaccine in combination with low-dose gemcitabine induced potent anti-tumor immunity in pancreatic cancer model. Cancer Immunol Immunother. (2025) 74:68. doi: 10.1007/s00262-024-03924-x
57. Pérez-Baños A, Gleisner MA, Flores I, Pereda C, Navarrete M, Araya JP, et al. Whole tumour cell-based vaccines: tuning the instruments to orchestrate an optimal antitumour immune response. Br J Cancer. (2023) 129:572–85. doi: 10.1038/s41416-023-02327-6
58. Hardacre JM, Mulcahy M, Small W, Talamonti M, Obel J, Krishnamurthi S, et al. Addition of algenpantucel-L immunotherapy to standard adjuvant therapy for pancreatic cancer: a phase 2 study. J Gastrointest Surg. (2013) 17:94–100. doi: 10.1007/s11605-012-2064-6
59. Hewitt DB, Nissen N, Hatoum H, Musher B, Seng J, Coveler AL, et al. A Phase 3 Randomized Clinical Trial of Chemotherapy With or Without Algenpantucel-L (HyperAcute-Pancreas) Immunotherapy in Subjects With Borderline Resectable or Locally Advanced Unresectable Pancreatic Cancer. Ann Surg. (2022) 275:45–53. doi: 10.1097/sla.0000000000004669
60. Jaffee EM, Hruban RH, Biedrzycki B, Laheru D, Schepers K, Sauter PR, et al. Novel allogeneic granulocyte-macrophage colony-stimulating factor-secreting tumor vaccine for pancreatic cancer: a phase I trial of safety and immune activation. J Clin Oncol. (2001) 19:145–56. doi: 10.1200/jco.2001.19.1.145
61. Le DT, Lutz E, Uram JN, Sugar EA, Onners B, Solt S, et al. Evaluation of ipilimumab in combination with allogeneic pancreatic tumor cells transfected with a GM-CSF gene in previously treated pancreatic cancer. J Immunother. (2013) 36:382–9. doi: 10.1097/CJI.0b013e31829fb7a2
62. Laheru D, Lutz E, Burke J, Biedrzycki B, Solt S, Onners B, et al. Allogeneic granulocyte macrophage colony-stimulating factor-secreting tumor immunotherapy alone or in sequence with cyclophosphamide for metastatic pancreatic cancer: a pilot study of safety, feasibility, and immune activation. Clin Cancer Res. (2008) 14:1455–63. doi: 10.1158/1078-0432.Ccr-07-0371
63. Heumann T, Judkins C, Li K, Lim SJ, Hoare J, Parkinson R, et al. A platform trial of neoadjuvant and adjuvant antitumor vaccination alone or in combination with PD-1 antagonist and CD137 agonist antibodies in patients with resectable pancreatic adenocarcinoma. Nat Commun. (2023) 14:3650. doi: 10.1038/s41467-023-39196-9
64. Hill CS, Parkinson R, Jaffee EM, Sugar E, Zheng L, Onners B, et al. Phase 1 Study of Adjuvant Allogeneic Granulocyte-Macrophage Colony-Stimulating Factor-Transduced Pancreatic Tumor Cell Vaccine, Low-Dose Cyclophosphamide, and Stereotactic Body Radiation Therapy Followed by FOLFIRINOX in High-Risk Resected Pancreatic Ductal Adenocarcinoma. Int J Radiat Oncol Biol Phys. (2025) 121:930–41. doi: 10.1016/j.ijrobp.2024.10.039
65. Lu S, Zhang Z, Du P, Chard LS, Yan W, El Khouri M, et al. A Virus-Infected, Reprogrammed Somatic Cell-Derived Tumor Cell (VIReST) Vaccination Regime Can Prevent Initiation and Progression of Pancreatic Cancer. Clin Cancer Res. (2020) 26:465–76. doi: 10.1158/1078-0432.Ccr-19-1395
66. Liao HC and Liu SJ. Advances in nucleic acid-based cancer vaccines. J BioMed Sci. (2025) 32:10. doi: 10.1186/s12929-024-01102-w
67. Harisa GI, Faris TM, Sherif AY, Alzhrani RF, Alanazi SA, Kohaf NA, et al. Coding Therapeutic Nucleic Acids from Recombinant Proteins to Next-Generation Vaccines: Current Uses, Limitations, and Future Horizons. Mol Biotechnol. (2024) 66:1853–71. doi: 10.1007/s12033-023-00821-z
68. Aziguli T, Xiao SY, Yang Y, Mutailifu M, Li XQ, Yin SQ, et al. ENO1 promotes PDAC progression by inhibiting CD8(+) T cell infiltration through upregulating PD-L1 expression via HIF-1α signaling. Transl Oncol. (2025) 52:102261. doi: 10.1016/j.tranon.2024.102261
69. Principe M, Borgoni S, Cascione M, Chattaragada MS, Ferri-Borgogno S, Capello M, et al. Alpha-enolase (ENO1) controls alpha v/beta 3 integrin expression and regulates pancreatic cancer adhesion, invasion, and metastasis. J Hematol Oncol. (2017) 10:16. doi: 10.1186/s13045-016-0385-8
70. Cappello P, Tonoli E, Curto R, Giordano D, Giovarelli M, and Novelli F. Anti-α-enolase antibody limits the invasion of myeloid-derived suppressor cells and attenuates their restraining effector T cell response. Oncoimmunology. (2016) 5:e1112940. doi: 10.1080/2162402x.2015.1112940
71. Cappello P, Rolla S, Chiarle R, Principe M, Cavallo F, Perconti G, et al. Vaccination with ENO1 DNA prolongs survival of genetically engineered mice with pancreatic cancer. Gastroenterology. (2013) 144:1098–106. doi: 10.1053/j.gastro.2013.01.020
72. Kaneda MM, Cappello P, Nguyen AV, Ralainirina N, Hardamon CR, Foubert P, et al. Macrophage PI3Kγ Drives Pancreatic Ductal Adenocarcinoma Progression. Cancer Discovery. (2016) 6:870–85. doi: 10.1158/2159-8290.Cd-15-1346
73. Cappello P, Curcio C, Mandili G, Roux C, Bulfamante S, and Novelli F. Next Generation Immunotherapy for Pancreatic Cancer: DNA Vaccination is Seeking New Combo Partners. Cancers (Basel). (2018) 10. doi: 10.3390/cancers10020051
74. Curcio C, Mucciolo G, Roux C, Brugiapaglia S, Scagliotti A, Guadagnin G, et al. PI3Kγ inhibition combined with DNA vaccination unleashes a B-cell-dependent antitumor immunity that hampers pancreatic cancer. J Exp Clin Cancer Res. (2024) 43:157. doi: 10.1186/s13046-024-03080-1
75. Mandili G, Curcio C, Bulfamante S, Follia L, Ferrero G, Mazza E, et al. In pancreatic cancer, chemotherapy increases antitumor responses to tumor-associated antigens and potentiates DNA vaccination. J Immunother Cancer. (2020) 8. doi: 10.1136/jitc-2020-001071
76. Bert T, Lubomierski N, Gangsauge S, Münch K, Printz H, Prasnikar N, et al. Expression spectrum and methylation-dependent regulation of melanoma antigen-encoding gene family members in pancreatic cancer cells. Pancreatology. (2002) 2:146–54. doi: 10.1159/000055905
77. Cogdill AP, Frederick DT, Cooper ZA, Garber HR, Ferrone CR, Fiedler A, et al. Targeting the MAGE A3 antigen in pancreatic cancer. Surgery. (2012) 152:S13–18. doi: 10.1016/j.surg.2012.05.031
78. Qin H, Chen J, Bouchekioua-Bouzaghou K, Meng YM, Griera JB, Jiang X, et al. Immunization with a multi-antigen targeted DNA vaccine eliminates chemoresistant pancreatic cancer by disrupting tumor-stromal cell crosstalk. J Transl Med. (2023) 21:702. doi: 10.1186/s12967-023-04519-3
79. Geng F, Dong L, Bao X, Guo Q, Guo J, Zhou Y, et al. CAFs/tumor cells co-targeting DNA vaccine in combination with low-dose gemcitabine for the treatment of Panc02 murine pancreatic cancer. Mol Ther Oncolytics. (2022) 26:304–13. doi: 10.1016/j.omto.2022.07.008
80. Rong Y, Jin D, Wu W, Lou W, Wang D, Kuang T, et al. Induction of protective and therapeutic anti-pancreatic cancer immunity using a reconstructed MUC1 DNA vaccine. BMC Cancer. (2009) 9:191. doi: 10.1186/1471-2407-9-191
81. Wu WC, Jin DY, Lou WH, Wang DS, and Qin XY. Induction of antigen-specific CTL and antibody responses in mice by a novel recombinant tandem repeat DNA vaccine targeting at mucin 1 of pancreatic cancer. J Cancer Res Clin Oncol. (2010) 136:1861–8. doi: 10.1007/s00432-010-0845-4
82. Xuan Y, Yan W, Wang R, Wang X, Guo Y, et al. and IL-21-armed oncolytic vaccinia virus significantly enhances anti-tumor activity and synergizes with anti-PD1 immunotherapy in pancreatic cancer. Front Immunol. (2024) 15:1506632. doi: 10.3389/fimmu.2024.1506632
83. Kutzler MA and Weiner DB. DNA vaccines: ready for prime time? Nat Rev Genet. (2008) 9:776–88. doi: 10.1038/nrg2432
84. Ulmer JB. DNA vaccines forum: how to enhance DNA vaccines safety and potency? IDrugs. (2005) 8:357–9.
85. Guasp P, Reiche C, Sethna Z, and Balachandran VP. RNA vaccines for cancer: Principles to practice. Cancer Cell. (2024) 42:1163–84. doi: 10.1016/j.ccell.2024.05.005
86. Vishweshwaraiah YL and Dokholyan NV. mRNA vaccines for cancer immunotherapy. Front Immunol. (2022) 13:1029069. doi: 10.3389/fimmu.2022.1029069
87. Cai W, Cai M, Wu M, and Gao X. mRNA vaccines: Emerging opportunities for herpesvirus prevention and therapeutic intervention. Int J Biol Macromol. (2025) 321. doi: 10.1016/j.ijbiomac.2025.146204
88. Braiteh F, LoRusso P, Balmanoukian A, Klempner S, Camidge DR, Hellmann M, et al. Abstract CT169: A phase Ia study to evaluate RO7198457, an individualized Neoantigen Specific immunoTherapy (iNeST), in patients with locally advanced or metastatic solid tumors. Cancer Res. (2020) 80:CT169–9. doi: 10.1158/1538-7445.AM2020-CT169
89. Rojas LA, Sethna Z, Soares KC, Olcese C, Pang N, Patterson E, et al. Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer. Nature. (2023) 618:144–50. doi: 10.1038/s41586-023-06063-y
90. Sethna Z, Guasp P, Reiche C, Milighetti M, Ceglia N, Patterson E, et al. RNA neoantigen vaccines prime long-lived CD8(+) T cells in pancreatic cancer. Nature. (8056) 2025:1042–51:639. doi: 10.1038/s41586-024-08508-4
91. Kyte JA. Cancer vaccination with telomerase peptide GV1001. Expert Opin Investig Drugs. (2009) 18:687–94. doi: 10.1517/13543780902897631
92. Nava-Parada P and Emens LA. GV-1001, an injectable telomerase peptide vaccine for the treatment of solid cancers. Curr Opin Mol Ther. (2007) 9:490–7.
93. Bernhardt SL, Gjertsen MK, Trachsel S, Møller M, Eriksen JA, Meo M, et al. Telomerase peptide vaccination of patients with non-resectable pancreatic cancer: A dose escalating phase I/II study. Br J Cancer. (2006) 95:1474–82. doi: 10.1038/sj.bjc.6603437
94. Jo JH, Kim YT, Choi HS, Kim HG, Lee HS, Choi YW, et al. Efficacy of GV1001 with gemcitabine/capecitabine in previously untreated patients with advanced pancreatic ductal adenocarcinoma having high serum eotaxin levels (KG4/2015): an open-label, randomised, Phase 3 trial. Br J Cancer. (2024) 130:43–52. doi: 10.1038/s41416-023-02474-w
95. Shindo Y, Hazama S, Suzuki N, Iguchi H, Uesugi K, Tanaka H, et al. Predictive biomarkers for the efficacy of peptide vaccine treatment: based on the results of a phase II study on advanced pancreatic cancer. J Exp Clin Cancer Res. (2017) 36:36. doi: 10.1186/s13046-017-0509-1
96. Cai Z, Li Z, Zhong W, Lin F, Dong X, Ye H, et al. Targeting Mesothelin Enhances Personalized Neoantigen Vaccine Induced Antitumor Immune Response in Orthotopic Pancreatic Cancer Mouse Models. Adv Sci (Weinh). (2025) 12:e2407976. doi: 10.1002/advs.202407976
97. Perez-Penco M, Weis-Banke SE, Schina A, Siersbæk M, Hübbe ML, Jørgensen MA, et al. TGFβ-derived immune modulatory vaccine: targeting the immunosuppressive and fibrotic tumor microenvironment in a murine model of pancreatic cancer. J Immunother Cancer. (2022) 10. doi: 10.1136/jitc-2022-005491
98. Suzuki Y and Sato Y. Vasohibin-2-Targeting Therapies for the Treatment of Pancreatic Ductal Adenocarcinoma. Tohoku J Exp Med. (2024) 262:163–71. doi: 10.1620/tjem.2023.J109
99. Park S, Kim D, Wu G, Jung H, Park JA, Kwon HJ, et al. A peptide-CpG-DNA-liposome complex vaccine targeting TM4SF5 suppresses growth of pancreatic cancer in a mouse allograft model. Onco Targets Ther. (2018) 11:8655–72. doi: 10.2147/ott.S186606
100. Pant S, Wainberg ZA, Weekes CD, Furqan M, Kasi PM, Devoe CE, et al. Lymph-node-targeted, mKRAS-specific amphiphile vaccine in pancreatic and colorectal cancer: the phase 1 AMPLIFY-201 trial. Nat Med. (2024) 30:531–42. doi: 10.1038/s41591-023-02760-3
101. Wainberg ZA, Weekes CD, Furqan M, Kasi PM, Devoe CE, Leal AD, et al. Lymph node-targeted, mKRAS-specific amphiphile vaccine in pancreatic and colorectal cancer: phase 1 AMPLIFY-201 trial final results. Nat Med. (2025). doi: 10.1038/s41591-025-03876-4
102. Minaei E, Ranson M, Aghmesheh M, Sluyter R, and Vine KL. Enhancing pancreatic cancer immunotherapy: Leveraging localized delivery strategies through the use of implantable devices and scaffolds. J Control Release. (2024) 373:145–60. doi: 10.1016/j.jconrel.2024.07.023
103. Yu B, Shao S, and Ma W. Frontiers in pancreatic cancer on biomarkers, microenvironment, and immunotherapy. Cancer Lett. (2025) 610:217350. doi: 10.1016/j.canlet.2024.217350
104. Bear AS, Vonderheide RH, and O'Hara MH. Challenges and Opportunities for Pancreatic Cancer Immunotherapy. Cancer Cell. (2020) 38:788–802. doi: 10.1016/j.ccell.2020.08.004
105. Katsikis PD, Ishii KJ, and Schliehe C. Challenges in developing personalized neoantigen cancer vaccines. Nat Rev Immunol. (2024) 24:213–27. doi: 10.1038/s41577-023-00937-y
106. Castro A, Zanetti M, and Carter H. Neoantigen Controversies. Annu Rev BioMed Data Sci. (2021) 4:227–53. doi: 10.1146/annurev-biodatasci-092820-112713
107. Pearlman AH, Hwang MS, Konig MF, Hsiue EH, Douglass J, DiNapoli SR, et al. Targeting public neoantigens for cancer immunotherapy. Nat Cancer. (2021) 2:487–97. doi: 10.1038/s43018-021-00210-y
108. Blass E, Keskin DB, Tu CR, Forman C, Vanasse A, Sax HE, et al. A multi-adjuvant personal neoantigen vaccine generates potent immunity in melanoma. Cell. (2025). doi: 10.1016/j.cell.2025.06.019
109. Halbrook CJ, Lyssiotis CA, Pasca di Magliano M, and Maitra A. Pancreatic cancer: Advances and challenges. Cell. (2023) 186:1729–54. doi: 10.1016/j.cell.2023.02.014
110. Pallerla S, Pires IS, Melo MB, Yun D, Wagner A, Budai M, et al. Scale-up and cGMP manufacturing of next-generation vaccine adjuvant saponin/MPLA nanoParticles (SMNP). J Pharm Sci. (2025) 114:103913. doi: 10.1016/j.xphs.2025.103913
111. Vemulapalli V, Natha C, and Thosani N. Endoscopic Immuno-Oncology: A New Frontier in Treatment of Pancreatic Cancer. Cancers (Basel). (2025) 17. doi: 10.3390/cancers17132091
112. Zhou J, Zhang K, Liu C, Long J, Gu H, Dong H, et al. Utidelone suppresses PDAC growth and enhances gemcitabine therapy by inducing immunogenic cell death. iScience. (2025) 28:112509. doi: 10.1016/j.isci.2025.112509
113. Urban W, Kropacz M, Łach M, and Jankowska A. CRISPR-Cas9 in the Tailoring of Genetically Engineered Animals. Curr Issues Mol Biol. (2025) 47. doi: 10.3390/cimb47050330
114. Whitaker WR, Russ ZN, Stanley Shepherd E, Popov LM, Louie A, Lam K, et al. Controlled colonization of the human gut with a genetically engineered microbial therapeutic. Science. (2025) 389:303–8. doi: 10.1126/science.adu8000
115. Yang M, Cui M, Sun Y, Liu S, and Jiang W. Mechanisms, combination therapy, and biomarkers in cancer immunotherapy resistance. Cell Commun Signal. (2024) 22:338. doi: 10.1186/s12964-024-01711-w
116. Cheng NC and Vonderheide RH. Immune vulnerabilities of mutant KRAS in pancreatic cancer. Trends Cancer. (2023) 9:928–36. doi: 10.1016/j.trecan.2023.07.004
117. Hingorani SR, Petricoin EF, Maitra A, Rajapakse V, King C, Jacobetz MA, et al. Preinvasive and invasive ductal pancreatic cancer and its early detection in the mouse. Cancer Cell. (2003) 4:437–50. doi: 10.1016/s1535-6108(03)00309-x
118. Hosein AN, Dangol G, Okumura T, Roszik J, Rajapakshe K, Siemann M, et al. Loss of Rnf43 Accelerates Kras-Mediated Neoplasia and Remodels the Tumor Immune Microenvironment in Pancreatic Adenocarcinoma. Gastroenterology. (2022) 162:1303–1318.e1318. doi: 10.1053/j.gastro.2021.12.273
119. Zhong Y, Tian Y, Wang Y, Bai J, Long Q, Yan L, et al. Small Extracellular Vesicle piR-hsa-30937 Derived from Pancreatic Neuroendocrine Neoplasms Upregulates CD276 in Macrophages to Promote Immune Evasion. Cancer Immunol Res. (2024) 12:840–53. doi: 10.1158/2326-6066.Cir-23-0825
120. Gugulothu KN, Anvesh Sai P, Suraparaju S, Karuturi SP, Pendli G, Kamma RB, et al. WT1 Cancer Vaccine in Advanced Pancreatic Cancer: A Systematic Review. Cureus. (2024) 16:e56934. doi: 10.7759/cureus.56934
121. Wu J and Cai J. Dilemma and Challenge of Immunotherapy for Pancreatic Cancer. Dig Dis Sci. (2021) 66:359–68. doi: 10.1007/s10620-020-06183-9
122. Kaczmarek M, Poznańska J, Fechner F, Michalska N, Paszkowska S, Napierała A, et al. Cancer Vaccine Therapeutics: Limitations and Effectiveness-A Literature Review. Cells. (2023) 12. doi: 10.3390/cells12172159
123. Gössling GCL, Zhen DB, Pillarisetty VG, and Chiorean EG. Combination immunotherapy for pancreatic cancer: challenges and future considerations. Expert Rev Clin Immunol. (2022) 18:1173–86. doi: 10.1080/1744666x.2022.2120471
124. Sarfraz Z, Sarfraz A, Farooq MD, Khalid M, Cheema K, Javad F, et al. The Current Landscape of Clinical Trials for Immunotherapy in Pancreatic Cancer: A State-of-the-Art Review. J Gastrointest Cancer. (2024) 55:1026–57. doi: 10.1007/s12029-024-01078-8
125. Huang X, Zhang G, Tang TY, Gao X, and Liang TB. Personalized pancreatic cancer therapy: from the perspective of mRNA vaccine. Mil Med Res. (2022) 9:53. doi: 10.1186/s40779-022-00416-w
126. Liu N, Wang X, Wang Z, Kan Y, Fang Y, Gao J, et al. Nanomaterials-driven in situ vaccination: a novel frontier in tumor immunotherapy. J Hematol Oncol. (2025) 18:45. doi: 10.1186/s13045-025-01692-4
Keywords: pancreatic cancer, vaccine, immunotherapy, tumor antigen, clinical application
Citation: Wang Y, Huang P, Li C, Tu S and Yang H (2025) Therapeutic cancer vaccines in pancreatic cancer. Front. Immunol. 16:1674743. doi: 10.3389/fimmu.2025.1674743
Received: 28 July 2025; Accepted: 29 September 2025;
Published: 20 October 2025.
Edited by:
Mariam Mathew George, Cornell University, United StatesReviewed by:
Kuo-Hui Su, The University of Toledo, United StatesCopyright © 2025 Wang, Huang, Li, Tu and Yang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hua Yang, eWFuZ2h1YV9kY0AxMjYuY29t
 YuHan Wang1
YuHan Wang1 Peng Huang
Peng Huang Hua Yang
Hua Yang