- 1Department of Biochemistry and Genetics, Barkatullah University, Bhopal, India
- 2Division of Pathology, ICAR-Indian Veterinary Research Institute, Bareilly, India
- 3Immunology Section, ICAR-Indian Veterinary Research Institute, Bareilly, India
- 4Central University Laboratory, Tamil Nadu Veterinary and Animal Sciences University, Chennai, India
- 5Department of Veterinary Microbiology and Immunology, College of Veterinary Sciences, UP Pandit Deen Dayal Upadhayay Pashu Chikitsa Vigyan Vishwavidyalay Evum Go-Anusandhan Sansthan, Mathura, India
- 6Division of Biological Standardization, ICAR-Indian Veterinary Research Institute, Bareilly, India
- 7Division of Veterinary Biotechnology, ICAR-Indian Veterinary Research Institute, Bareilly, India
- 8ICAR-Indian Veterinary Research Institute, Bareilly, India
- 9School of Engineering and Science, Tecnologico de Monterrey, Campus Monterrey, Monterrey, Mexico
- 10Cellular Immunology Lab, Frank Reidy Research Center of Bioelectrics, Old Dominion University, Norfolk, VA, United States
Zika virus (ZIKV) remained largely quiescent for nearly six decades after its first appearance in 1947. ZIKV reappeared after 2007, resulting in a declaration of an international “public health emergency” in 2016 by the World Health Organization (WHO). Until this time, ZIKV was considered to induce only mild illness, but it has now been established as the cause of severe clinical manifestations, including fetal anomalies, neurological problems, and autoimmune disorders. Infection during pregnancy can cause congenital brain abnormalities, including microcephaly and neurological degeneration, and in other cases, Guillain-Barré syndrome, making infections with ZIKV a substantial public health concern. Genomic and molecular investigations are underway to investigate ZIKV pathology and its recent enhanced pathogenicity, as well as to design safe and potent vaccines, drugs, and therapeutics. This review describes progress in the design and development of various anti-ZIKV therapeutics, including drugs targeting virus entry into cells and the helicase protein, nucleosides, inhibitors of NS3 protein, small molecules, methyltransferase inhibitors, interferons, repurposed drugs, drugs designed with the aid of computers, neutralizing antibodies, convalescent serum, antibodies that limit antibody-dependent enhancement, and herbal medicines. Additionally, covalent inhibitors of viral protein expression and anti-Toll-like receptor molecules are discussed. To counter ZIKV-associated disease, we need to make rapid progress in developing novel therapies that work effectually to inhibit ZIKV.
Introduction
Zika virus (ZIKV) is a mosquito-borne virus belonging to the Spondweni serocomplex in the genus Flavivirus of the family Flaviviridae that has become a new threat following the Ebola virus epidemic (Singh et al., 2016). The expanding ZIKV epidemic was declared an emergency by the World Health Organization on February 1, 2016 (Fajardo et al., 2016; WHO, 2016). ZIKV is a single-stranded RNA virus that encodes a single polyprotein that is cleaved to form mature proteins, i.e., the capsid, envelope (E), and precursor of membrane and non-structural proteins. Other flaviviruses such as dengue virus (DENV), yellow fever virus (YFV), and West Nile virus (WNV) are closely related to ZIKV. In the last six decades since its discovery, ZIKV has been considered a mild human pathogen, but recently it has emerged as threat to global health, showing increased virulence, rapid spread, and an association with microcephaly and grave neurological complications like Guillain-Barré syndrome (GBS) (Cao-Lormeau et al., 2016; Carteaux et al., 2016; Mlakar et al., 2016; Sarno et al., 2016).
Zika virus has a wide tissue tropism in an experimental rhesus macaque model, infecting the hemolymphatic system, lymph nodes, spleen, cardiopulmonary, gastrointestinal, integument, and genitourinary tissues, along with the adrenal gland, spinal cord, and cerebrospinal fluid (Coffey et al., 2017). Additionally, it has been reported in muscles, kidneys, bladders, and in excreted urine (Gourinat et al., 2015). In males, ZIKV can infect testes (Govero et al., 2016), prostate and seminal vesicles, explaining the long-term persistence of viremia in semen, even after virus is no longer detectable in blood. In the female reproductive system, virus can be maintained in the vagina, uterus (Hirsch et al., 2017), vaginal epithelium (mice), and in uterine fibroblasts (Miner and Diamond, 2017). Miner and Diamond (2017) demonstrated the wide tissue tropism of the virus in Hofbauer cells, trophoblasts, and endothelial cells from the placenta. In addition, ZIKV was found to infect the cornea, neurosensory retina, optic nerve, aqueous humor, and tears. ZIKV infection in eyes results in uveitis (Furtado et al., 2016), and the persistence of the virus in cerebrospinal fluid and lymph nodes appears to enhance activity of rapamycin (mTOR), proinflammatory, and anti-apoptotic signaling pathways and reduce extracellular matrix signaling (Aid et al., 2017).
Zika virus adapts to human hosts by altering NS1 codon usage to facilitate viral replication and to increase viral titers (de Melo Freire et al., 2015). Furthermore, ZIKV placental transfer and its ability to infect neuronal tissue of growing fetuses is evident (Martines et al., 2016; Mlakar et al., 2016).
The complications of ZIKV infection are intensified by the unavailability of effective prophylactics, vaccines, or therapeutics. The spread of ZIKV, which, earlier, was limited to small geographical areas, has been facilitated by globalization, unplanned urbanization, poor sanitation, inadequate health services, and the emergence of insecticide resistance in mosquito vectors. Mosquitoes, mainly Aedes aegypti and Ae. albopictus, play a primary role in ZIKV transmission (Musso and Gubler, 2016). In addition, sexual transmission; male-to-female, female-to-male, and male-to-male transmissions have been reported by Hamer et al. (2017). A mathematical modeling study conducted by Gao et al. (2016) indicated that sexual activity contributed to 3.044% of transmission. During the typical incubation period of 2–7 days, despite the relatively low viral loads in people, infected human patients serve as a source of ZIKV (Foy et al., 2011). After viremia declines, convalescence begins, during which time a person is no longer infectious to a mosquito; however, they remain infective to other human hosts, with a low infection rate. The convalescent stage ends with establishment of long lasting immunity (Gao et al., 2016).
Zika virus vaccines in development include inactivated virus, nucleic acid-based vaccines (DNA or RNA), live vector vaccines, subunit vaccines, virus-like particles, and recombinant ZIKV. Because of its devastating effects, effective therapeutic agents and a vaccine are urgently needed. Presently, there are several drugs reported to be useful in treating ZIKV, a few of which are repurposed drugs. Efforts to develop effective drugs have increased worldwide, and a few compounds are in phase I trials (Alam et al., 2017; Ali et al., 2017). The present review discusses recent advances in and prospects for the design and development of various anti-viral drugs and therapeutics for ZIKV infection, including the identification of novel drug targets. The updated information compiled here will contribute to the design and development of additional effective drugs and pharmaceuticals to curtain the ill effects of ZIKV.
Advances in the Design and Development of Anti-Zikv Drugs and Therapies
Specific anti-viral drugs are not yet available to combat ZIKV. Acetaminophen is used to control fever and pain, anti-histamines are used for pruritic rashes, and fluids are administered to prevent dehydration in ZIKV-infected patients. However, certain drugs such as acetylsalicylic acid (aspirin) and non-steroidal anti-inflammatory drugs (NSAIDs) are contraindicated because they increase the risk of internal bleeding, and other flaviviral infections, including DENV and chikungunya virus, can cause hemorrhage (; Mukherjee and Era, 2016; Musso and Gubler, 2016). ZIKV actively replicates in and causes death of neurons. Research for developing anti-viral drugs for Zika is going on fast and compounds like Sofosbuvir, 7-DMA, BCX4450, and NITD008 are currently entering a phase I trial (Ali et al., 2017).
Recently, several drugs and therapeutic candidates have been explored to determine the most effective treatment regimens to combat ZIKV and its severe clinical complications, which are being described in the following sections.
Interferons as Anti-virals
Activation of the innate immune system by viruses leads to the release of interferons (IFNs), which are responsible for the elimination of viruses and for immune regulation. In an in vitro cell culture system developed for ZIKV cultivation, IFN-α, IFN-β, and IFN-γ have been shown to inhibit viral replication (Contreras and Arumugaswami, 2016). Type I interferons have shown dose-dependent inhibition of ZIKV replication in a cell culture study that used quantitative RT-PCR (Goebel et al., 2016). The inverse has been documented by Bowen et al. (2017); they demonstrated ZIKV’s ability to evade in the presence of type I interferon responses by degrading STAT2 signaling molecules. Trophoblastic cells secrete IFN-λ1, which exhibits anti-viral activities against single-stranded RNA viruses. In an in vitro model, conditioned medium obtained from PHT cells has been found to inhibit ZIKV growth in trophoblastic and non-trophoblastic cells by stimulating the secretion of IFN-λ1 (Bayer et al., 2016).
Inhibition of Virus Entry into Cells
Inhibition of viral entry into a cell can serve as the first line of defense against ZIKV infection. ZIKV first binds to cell receptors, including AXL (Nowakowski et al., 2016), DC-SIGN, Tyro3, TIM, and TAM (Hamel et al., 2015), and then enters cells by clathrin-dependent endocytosis. ZIKV entry is severely hampered in human microglial cell line (CHME3) by silencing the clathrin heavy chain (a component essential for clathrin-coated vesicle formation) and dynamin-2 (a GTPase, required to pinch off endocytic vesicle from the plasma membrane). TIM receptors mediate viral entry after binding with viral phosphatidylserine and phosphatidylethanolamine (Jemielity et al., 2013). TIM1-mediated entry of DENV-2, WNV, and EBOV is inhibited by duramycin-biotin, which has less profound hemolytic effects and does not exhibit cellular cytotoxicity (Richard et al., 2015). The same TIM1 receptors are also involved in ZIKV entry; therefore, these drugs can be evaluated for the prevention of ZIKV entry. Peptide (GQASNGVFVIHWGKFDSFGIAV) derived from the Japanese encephalitis virus (JEV) E protein stem is able to prevent ZIKV infection with IC50 even at the nanomolar scale (3.93 nM). It also decreases the viral load and prevents histopathological damages in brain and testes in AG6 mouse, and attenuates the inflammatory response (Chen et al., 2017).
Modes of entry of ZIKV and various drugs inhibiting viral entry and replication have been depicted in Figure 1.
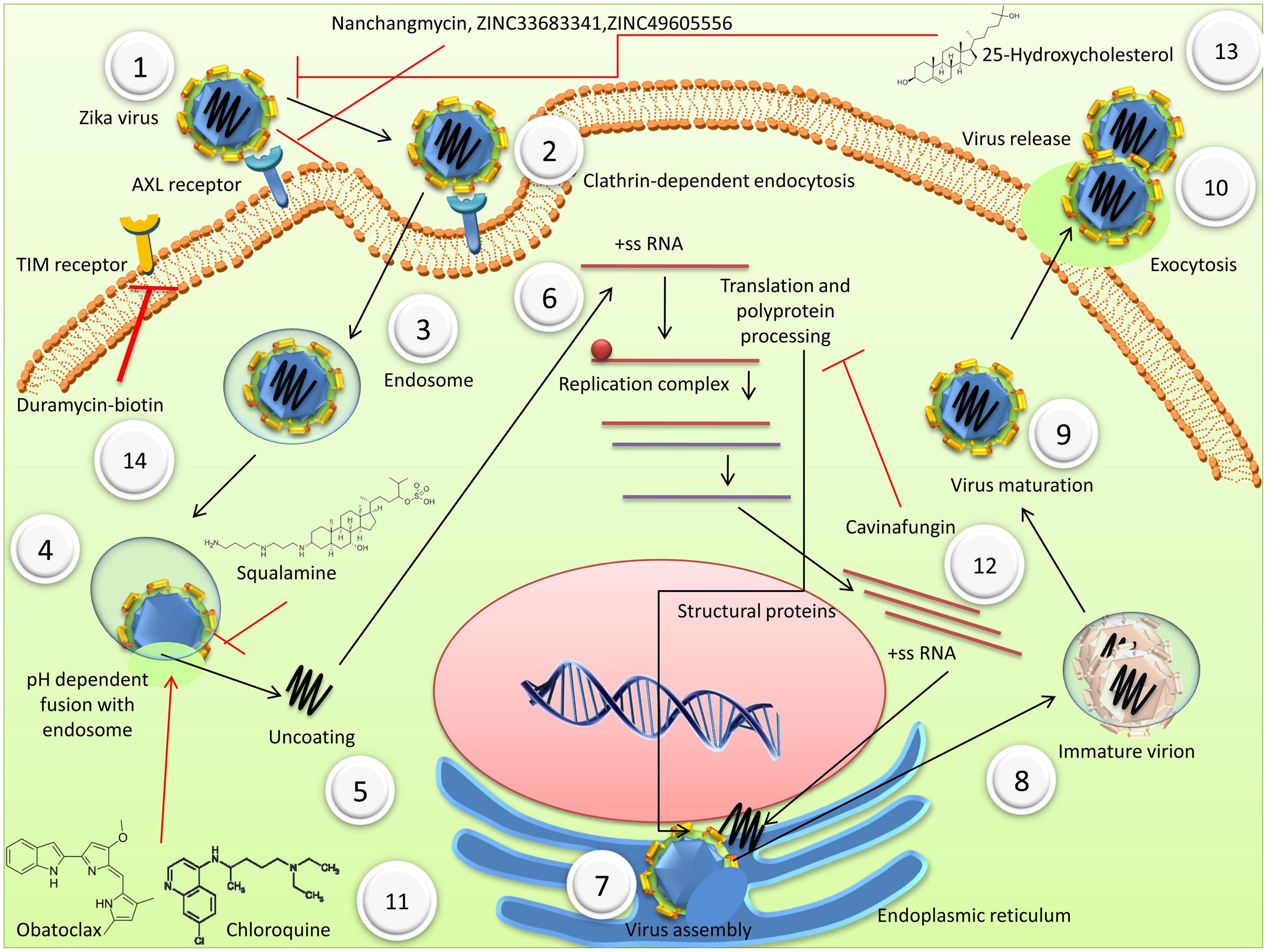
FIGURE 1. Mode of entry of Zika virus (ZIKV) and various drugs inhibiting viral entry and replication (1) ZIKV binds to cell receptors including AXL, DC-SIGN, Tyro3, TIM, and TAM. (2) Clathrin-dependent endocytosis. (3) Endosome mediated transport of ZIKV. (4) Fusion of virus membrane with host endosomal membrane, which depends on the pH. (5) Uncoating (6) The positive-sense genomic ssRNA is translated into a polyprotein, which is cleaved into all structural and non-structural proteins. Replication occurs at the surface of endoplasmic reticulum in cytoplasmic viral factories. A dsRNA genome is synthesized from the genomic ssRNA(+) (7) Virus assembly takes place at the endoplasmic reticulum. (8) At the endoplasmic reticulum, virions bud and are transported to the golgi apparatus. (9) In the golgi, prM protein is cleaved and maturation of the virion takes places. (10) Virions are released by exocytosis. (11) Obatoclax and chloroquineinhibit the acidic environment of endolysosomal vesicles. Squalamine, a cationic chemical, disturbs the electrostatic interaction between virus and host membranes during fusion and budding. (12) Cavinafungin, an alaninal-containing lipopeptide of fungal origin, inhibits ZIKV polyprotein processing and also the cleavage of signal peptide of host proteins. (13) Nanchangmycin, a polyether obtained from Streptomyces nanchangensis; small drug-like molecules, ZINC33683341 and ZINC49605556 block the receptor thus inhibiting the ZIKV entry. (14) TIM1 mediated entry is inhibited by Duramycin-biotin.
Blocking of Receptor Binding
After screening, more than 2000 molecules for their ability to inhibit ZIKV replication, nanchangmycin, a polyether obtained from Streptomyces nanchangensis that possesses insecticidal and anti-bacterial activity has been shown to block ZIKV replication in U2OS cells in in vitro. It is considered to act by targeting AXL receptors and blocking clathrin-mediated endocytosis. However, the exact mechanism of action of nanchangmycin is unknown (Rausch et al., 2017). Two small drug-like molecules, ZINC33683341 and ZINC49605556, both identified through homology modeling in silico, have been reported to inhibit ZIKV E protein by binding the viral receptors. Antiviral activities of ZINC33683341 have been confirmed in in vitro test. Thus, such viral inhibitors may be candidate molecules for ZIKV drugs after further research and clinical validation (Fernando et al., 2016).
Inhibition of Endosomal Fusion
Fusion of the endosome to lysosome is a critical step in releasing virus from endosomes. Obatoclax is a potential anti-neoplastic and pro-apoptotic synthetic small molecule Bcl-2 inhibitor. Its mesylate salt is reported to reduce the acidity of endolysosomal vesicles in in vitro model. Bcl-2 antagonists are effective only against viruses that require a low pH for fusion and entry, such as ZIKV, WNV, YFV, and others. Despite this limitation, Obatoclax works as a broad-spectrum anti-viral agent (Varghese et al., 2016). However, in clinical phase I and II trials while treating hematological and myeloid malignancies, Obatoclax did not produce satisfactory results, possibly due to inadequate inhibition of Bcl-2 family proteins. Chloroquine, which is an anti-malarial drug, raises endolysosomal pH and inhibits ZIKV infection in human brain microvascular endothelial cells, human neural stem cells, and mouse neurospheres (Delvecchio et al., 2016). Similarly, SaliPhe, a molecule under pre-clinical study and vATPase inhibitor, was tested as an inhibitor of endocytosis to obstruct ZIKV infection (Adcock et al., 2017). Griffithsin, a lectin isolated from the red alga Griffithsia sp., is a potent flaviviral entry inhibitor. It can cross-link high-mannose oligosaccharides present on the viral E glycoproteins and has shown wide anti-viral activity against HIV (Alexandre et al., 2011), HPV (Levendosky et al., 2015), HSV (Nixon et al., 2013), HCV (Takebe et al., 2013), and SARS (O’Keefe et al., 2010). Squalamine, a FDA approved cationic chemical, which act by disturbing electrostatic interactions between the virus and host membranes during fusion and budding (Zasloff et al., 2011), has been found well tolerated as component of eye drop in clinical studies conducted on human participants. Therefore, such potent drugs can be used as an anti-viral agent against ZIKV too.
Inhibition of Virus Replication
The single-stranded RNA genome encodes a polyprotein, which is proteolytically cleaved into three structural proteins (C, prM, and E) and seven non-structural proteins (NS1, NS2A, NS2B, NS3, NS4A, NS4B, and NS5). The NS5 protein, an RNA-dependent RNA polymerase, plays an important role in viral RNA synthesis and inhibits IFN signaling by binding to STAT2 (Grant et al., 2016). ZIKV NS3 protein exhibits helicase activity that is essential for viral replication. The helicase domain of NS3 is activated by GTPγS (triphosphate), which facilitates the unwinding and translocation of RNA at the time of replication. The ZIKV helicase, along with NS5, is an attractive target for ZIKV drug development. Small membrane-associated interferon-inducible transmembrane proteins (IFITMs) are intrinsic immune system defenses that are able to inhibit replication of several pathogenic viruses. Both IFITM1 and IFITM3 have been reported to inhibit early stages of infection and replication of ZIKV in HeLa cells with the predominant role played by IFITM3 (Savidis et al., 2016). Cavinafungin, an alaninal-containing lipopeptide of fungal origin, has recently been found to inhibit ZIKV polyprotein processing and cleavage of host protein signal peptides through inhibition of host endoplasmic reticulum signal peptidase in in vitro model (Estoppey et al., 2017). Synthetic 25-hydroxycholesterol has been shown to inhibit ZIKV entry into the host in an in vivo assay using mouse and rhesus macaque models (Li et al., 2017). An in vitro study conducted in Vero cells using compounds such as ribavirin, CMX001, T-705, and T-1105 showed that T-705 (favipiravir) and T-1105 were able to reduce cell death caused by ZIKV (Cai et al., 2017). Thus, these compounds that inhibit ZIKV replication in cell culture need to be explored further so that they can be used safely against ZIKV.
Numerous drugs involved in inhibition of virus replicationhave been portrayed in Figure 2.
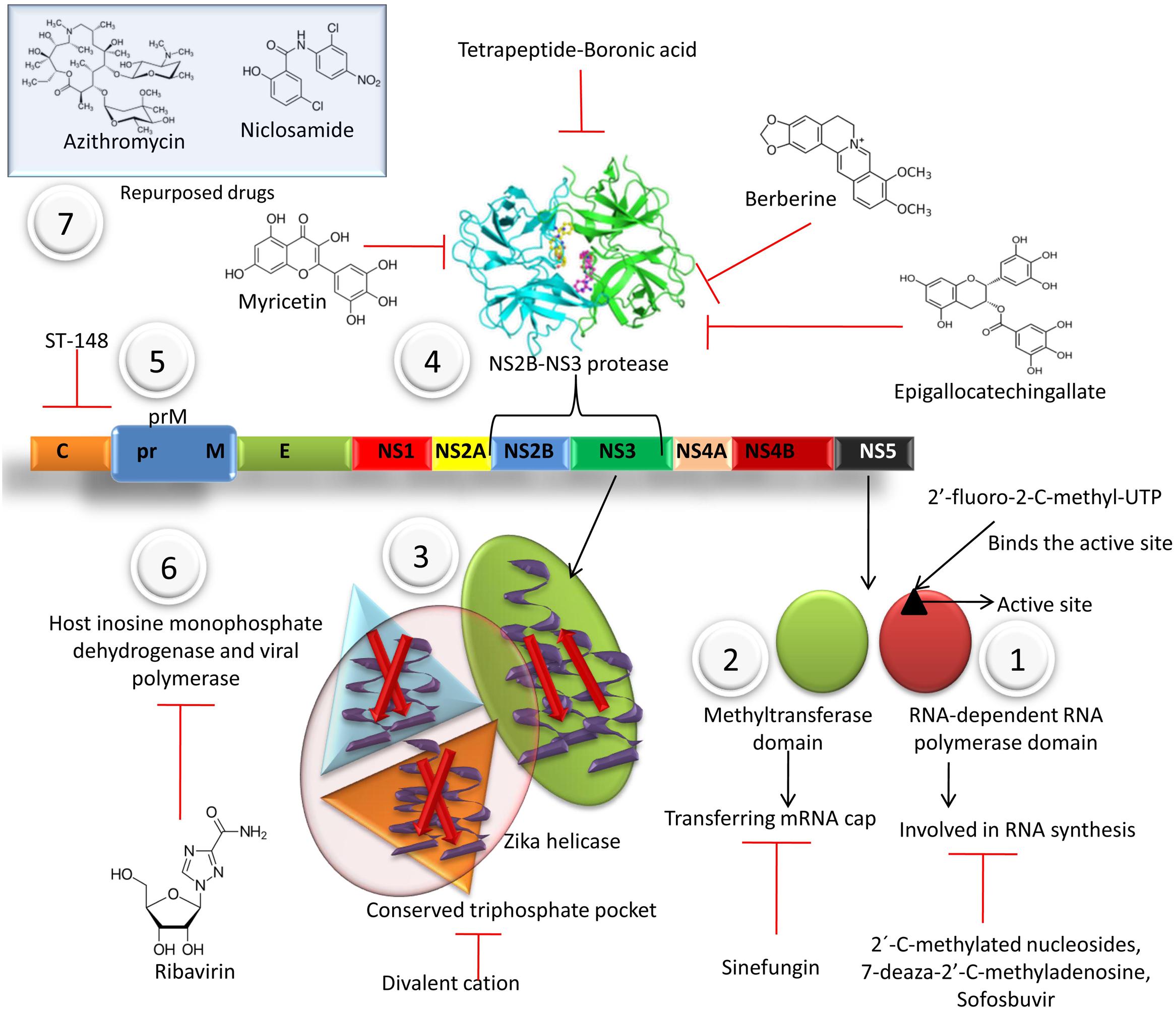
FIGURE 2. Various drugs involved in inhibition of virus replication at different stages. (1) Flaviviral NS5 has two major catalytic domains: RNA-dependent RNA polymerase (RdRp) and methyltransferase domain. Nucleoside analogs like 2′-C-methylated nucleosides, 7-deaza-2′-C-methyladenosine, Sofosbuvir may incorporate during the polymerase activity of RdRp in the viral nascent RNA chain and cause premature termination of RNA synthesis. The 2′-fluoro-2-C-methyl-UTP binds to the active site on NS5. (2) Methyltransferase domain is responsible for transferring mRNA cap. Sinefungin, an adenosine derivative, isolated from Streptomyces griseoleus, inhibit S-adenosyl-1-methionine (SAM), the natural substrate for methyltransferases and inhibit the methyltransferase activity. (3) Helicase crystal structure reveals a conserved triphosphate pocket and a positively charged tunnel for the accommodation of RNA. The helicase-activation is inhibited in the presence of divalent cation, due to extended conformation adopted by GTPγS in such conditions. (4) Tetrapeptide-Boronic acid is a potent inhibitor of NS2B-NS3 protease. Berberine, Myricetin, Epigallocatechingallate binds with affinity to NS3 protease and also inhibit the ZIKV replication. (5) Small-molecule inhibitor ST-148 inhibits capsid. (6) Ribavirin inhibits host inosine monophosphate dehydrogenase and viral polymerase. (7) Repurposed drugs like Chloroquine, azithromycin, niclosomide are used to treat ZIKV infection.
Inhibition of NS5
The flaviviral NS5 protein has two major catalytic domains. The first domain is the RNA-dependent RNA polymerase (RdRp), and the second one is a methyltransferase domain (Egloff et al., 2002). Conserved regions in ZIKV NS5 MTase and RdRp structures have been identified, so that current anti-virals targeting these regions in flaviviruses like DENV, WNV, and YFV may be used against ZIKV (Duan et al., 2017). NS5 is also involved in IFN antagonism. Mutant ZIKV, which is unable to prevent IFN-stimulated signaling can grow in IFN-deficient cell lines and has vaccine potential. Structural analysis of NS5 will provide information to design small molecule inhibitors that may prevent NS5-mediated interferon inhibition (Dar et al., 2017).
Inhibition of the RdRp domain
Nucleoside analogs may be incorporated into the viral nascent RNA chain during RdRp activity, causing premature termination of RNA synthesis (Kok, 2016). After screening a library of nucleoside analogs, 2′-C-methylated nucleosides were found to be most efficient at terminating synthesis because they were selective for ZIKV, and there was no cytotoxicity or adverse effects on cellular proliferation observed (Eyer et al., 2016). These nucleosides do not target the active RdRp site; rather, they terminate elongation of the nascent viral RNA chain. Another nucleoside analog, 7-deaza-2′-C-methyladenosine, developed initially as an HCV replication inhibitor, was also evaluated for inhibition of ZIKV replication in Vero cells and a mouse model. Using a mouse model deficient in IFN-α/β and the IFN-γ receptor (AG129 strain), intraperitoneal inoculation of ZIKV with approximately 2 plaque-forming units (pfu)/animal resulted in disease. Inoculated animals were treated with 7-deaza-2′-C-methyladenosine at a dose of 50 mg/kg/day. The drug delayed the onset of viremia and virus-induced morbidity and mortality in infected mice (Zmurko et al., 2016). Despite demonstrating efficacy in animal models, the drug remained unsuccessful in the phase I clinical trial conducted. Adenosine analog NITD008 was found to be effective against flaviviruses including ZIKV in both in vitro and in vivo studies and exhibited reduced viremia in mice (Deng Y.Q. et al., 2016). Unfortunately, in pre-clinical animal testing, it was found to be too toxic to be suitable for human trials.
Sofosbuvir (Sovaldi) is a nucleotide analog inhibitor that is commercially available for the treatment of chronic HCV infection. Its active metabolite is 2′-fluoro-2-C-methyl-UTP, which binds to the active site of NS5 (Reznik and Ashby, 2017). It has also shown the ability to inhibit ZIKV infection in human tumor cell lines and human fetal-derived neuronal stem cells (Bullard-Feibelman et al., 2017; Reznik and Ashby, 2017). In clinical phase I and II studies, the regimen containing sofosbuvir was found to be clinically safe and efficacious (Mangia and Piazzolla, 2014). It is a class B drug and can be used in men and non-pregnant women to prevent tissue damage. The 2′-C-ethynyl and 2′-C-methyl analog of 5′-triphosphates were found to be incorporated by the RdRp of ZIKV and therefore to efficiently terminate the elongating RNA chain (Lu et al., 2016); hence, they may be candidate for the design of better anti-virals against ZIKV.
Methyltransferase domain inhibition
The NS5 protein contains methyltransferase, which is responsible for transferring the mRNA cap. An NS5 methyltransferase null mutant was found to be lethal for the virus. Targeting the NS5 methyltransferase structural domains may prevent ZIKV propagation (Zhang C. et al., 2016). Structural analysis of ZIKV-NS5 aided in the identification of methyltransferase inhibitors based on in silico analysis, which could lead to the identification of hotspots for drug designing and development of anti-ZIKV drugs (Coutard et al., 2016; Stephen et al., 2016). Furthermore, sinefungin, an adenosine derivative originally isolated from Streptomyces griseoleus, is a potential anti-fungal and anti-parasitic compound that competitively inhibits S-adenosyl-1-methionine (SAM), the natural substrate for methyltransferases, as well as methyltransferase activity. On the basis of structural computational analysis, sinefungin was able to attach to GTP and GDP analogs and might be useful in enhancing their affinity toward the enzyme for greater selectivity and inhibition of ZIKV replication (Hercik et al., 2017). Severe toxicity was observed with this drug in animal studies conducted in dogs and goats while its usage as an anti-parasitic agent (Robert-Gero et al., 1989), which has hampered its clinical use. Therefore, less toxic and better tolerated derivatives must be obtained and tested against ZIKV.
Inhibition of NS3 (Helicase)
An NS3 inhibitor can be used to impede ZIKV infection. The helicase action of NS3 is inhibited by divalent cations that cause GTPγS to adopt an extended conformation (Cao et al., 2016). An understanding of the interactions between NS3 and GTPγS has led to the identification of small molecules that inhibit ZIKV. The ZIKV helicase crystal structure has revealed a conserved triphosphate pocket and a positively charged tunnel that accommodates the RNA. This critical substrate-binding pocket may be a good target for anti-virals. Tian et al. (2016) reported that the helicase of ZIKV is closely related to those of other members of the family Flaviviridae. Therefore, drugs that target the helicase of these viruses could also be explored for the control of ZIKV.
Inhibition of the NS2B-NS3 Protease
The crystallographic structure of the NS2B-NS3 protease of ZIKV indicated that tetrapeptide-boronic acid would be a potent inhibitor of the NS2B-NS3 protease (Lei et al., 2016). Based on studies of surface plasmon resonance and the kinetics of protease inhibition, Lee et al. (2016) identified several small molecular inhibitors of the protease. In addition, the group identified a “pre-open conformation” for the ZIKV NS2B-NS3 protease by X-ray crystallographic analysis. A molecular docking analysis revealed that berberine, an FDA-approved quaternary ammonium salt used against DENV, has a high binding affinity to the NS3 protease of ZIKV (Sahoo et al., 2016). Further, a ‘Hybrid Combinatorial Substrate Library’ approach has also been used to determine the substrate specificity of the NS2B-NS3 protease for the design of phosphonate-containing protease inhibitors (Rut et al., 2016). Lim et al. (2017) tested 22 plant polyphenolic compounds for their ability to inhibit Escherichia coli-expressed ZIKV NS2B-NS3 pro-protease. Inhibition of protease activity was evaluated by a fluorescence resonance energy transfer-based assay. Among all compounds tested, myricetin showed the strongest inhibition of the NS2B-NS3 protease, followed by luteolin, epicatechin gallate, gallocatechin gallate, and epigallocatechin gallate. CN-716, a capped peptidomimetic boronic-acid compound, has been found to form ZIKV NS2B-NS3 protease inhibitor complex, which might have biological importance in inhibiting ZIKV replication (Lei et al., 2016). Structural and functional insights gained through crystallographic techniques will accelerate the discovery of structure-based anti-ZIKV compounds.
Nucleoside Biosynthesis Inhibitors
Nucleoside biosynthesis inhibitors have broad anti-viral activities, but limited numbers of such compounds are available for clinical use. Inhibition of host inosine monophosphate dehydrogenase (IMPDH) and viral polymerase is the key to the anti-viral activity of ribavirin against flaviviruses (Crance et al., 2003). Mycophenolic acid (MPA) is an IMPDH inhibitor and exhibited potent dose-dependent anti ZIKV activity (EC50 of <0.32 μM) in cell culture experiments that were confirmed using qRT-PCR (Goebel et al., 2016). Contrary to the above finding, Adcock et al. (2017) found MPA to be not very effective (EC50 >50 μM) with observation of significant cytotoxicity and cytopathogenic effects in a high-throughput assay. Dihydroorotate dehydrogenase (DHODH), a host enzyme which is responsible for pyrimidine biosynthesis, is a possible enzyme target for antiviral research. Brequinar, an inhibitor of this enzyme, exhibited anti-ZIKV activity with an EC50 (half maximal effective concentration, which is a common measure of a drug’s potency; the lower the EC50, the more effective the drug is) at submicromolar levels, but a low therapeutic index for Brequinar has restricted its clinical use.
Capsid inhibition
In comparison with targeting E protein, the capsid has gained less research attention. It is a dimeric protein with a positively charged surface and hydrophobic core pocket. A single small-molecule inhibitor (ST-148) has been identified, which, despite poor oral bioavailability, was shown to decrease the viremias and viral loads of DENV-1–4, Modoc virus, YFV, and HCV. ST-148 was screened in a panel of 20,000 chemically diverse molecules using a high-throughput assay. It is non-mutagenic and selectively inhibits flaviviruses (Byrd et al., 2013). ST-148 mediates the self-interaction of capsid proteins and imposes structural rigidity, disturbing the assembly and disassembly of DENV particles (Scaturro et al., 2014). The concept of capsid protein stabilization may also be applicable to ZIKV.
Computer-Aided Drug Design
Before clinical studies, there are three essential phases of research: high-throughput computer or in silico drug design, medium-throughput in vitro drug testing, and low-throughput in vivo drug testing (Basak and Nandy, 2016). Understanding the ZIKV structure would aid in designing anti-viral therapies to curtail ZIKV infections (Cox et al., 2016). ZINC64717952 and ZINC39563464 have been found to block MTase and RdRp, respectively, based on a computational docking analysis (Ramharack and Soliman, 2017). The NS5 polymerase was inhibited by an andrographolide from Andrographis, whereas bisabolol and levomenol from Matricaria recutita and Myoporum crassifolium, respectively, blocked NS3 protease in virtual screening (Feranchuk et al., 2016). In silico techniques to identify enzyme blockers allow simultaneous assessments of various compounds with limits financial or experimental resource costs. A virtual screen of 36 million compounds from the MCULE database led to the selection of two molecules, MCULE-8830369631-0-1 and MCULE-9236850811-0-1, with inhibition constant (Ki) values of 0.08 and 0.30 μm, respectively (Onawole et al., 2017).
OpenZika, an IBM world community grid project, was used to identify drug molecule docking for various ZIKV structures. This platform allows data to be shared with researchers worldwide to facilitate the speedy discovery of anti-ZIKV drugs (Ekins et al., 2016). To elucidate therapeutically essential components like siRNAs, miRNAs, and sgRNAs (CRISPR/Cas9 targets) for ZIKV, an integrative multi-omics platform, ZikaVR1, is available. This platform offers other functions, including whole-genome alignment, codon information and bias assessments, phylogenetic deduction, and information regarding glycosylation sites and primer design (Gupta et al., 2016). The therapeutics based on enzyme, nucleoside, and capsid inhibitors are currently in their infancy and much more work needs to be carried out to bring these to clinical grounds. Computational analysis allows high throughput screening of potentially active molecules, however, in vivo validation is a prior requisite to move them from bench to bedside.
Drug Repurposing
Drugs take decades to develop and test for efficacy and safety. Since there is presently no approved vaccine or drug available for ZIKV, the major focus of researchers, therefore, is on attempting drug repurposing. Scientists are evaluating repurposing of several FDA approved drugs against ZIKV infections. In this direction, a few promising drug candidates have been shortlisted by adapting various screening methodologies. For example, chloroquine, a 4-aminoquinoline, readily increases the pH of acidic vesicles (Akpovwa, 2016) and inhibits a conformational change essential for fusion between the virus envelope and endosomal membrane (Smit et al., 2011). In vitro studies revealed that chloroquine decreases the number of ZIKV-infected neural cells in different cell models and protects cellular death (Delvecchio et al., 2016). Other anti-malarial drugs such as quinacrine, mefloquine, and GSK369796 also demonstrate anti-ZIKV activity by inhibiting autophagy (Balasubramanian et al., 2016). During the screening of a library of FDA-approved drugs, both established anti-virals like bortezomib and mycophenolic acid and compounds with no previously reported anti-viral activity (e.g., daptomycin) were found to inhibit ZIKV replication in human cervical, placental, neural stem, and primary human amniotic cells (Barrows et al., 2016). Xu M. et al. (2016) screened a panel of compounds containing FDA-approved drugs, drugs in clinical trials, and pharmacologically active compounds to suppress infection-induced caspase activity. Human neural progenitor cells and glial SNB-19 cells infected with ZIKV were used as models to quantify ZIKV-induced caspase-3 activity. Of these compounds, a pro-caspase inhibitor, emricasan, successfully protected both neural cell monolayers and three-dimensional organoid cultures of neural cells by decreasing ZIKV-induced caspase-3. Similarly, screening of 725 FDA-approved chemically diverse compounds in ZIKV-infected Huh7 cells at a 20-μM concentration led to the selection of lovastatin, a drug used to reduce cholesterol; 5-fluorouracil used as a cancer treatment; 6-azauridine, a broad-spectrum antimetabolite; palonosetron, which is used to treat chemotherapy-induced nausea and vomiting; and kitasamycin, a macrolide antibiotic. The selection criteria included a selectivity index, maximum activity, and the EC50 of compounds (Pascoalino et al., 2016).
Niclosamide, clinically given to treat helminths inefction, can protect ZIKV-infected cells and inhibit virus replication (Xu M. et al., 2016). Bortezomib and sorafenib are anti-cancer drugs possessing anti-viral activity and have been well tolerated in phase I clinical trials (Cheng et al., 2016). Azithromycin, a commercially available antibiotic, was also found to inhibit ZIKV proliferation in cultured brain cells, suggesting a possibly drug to prevent GBS and microcephaly (Retallack et al., 2016). Recently, bromocriptine, a drug indicated for the treatment of pituitary tumors, Parkinson’s disease, and type 2 diabetes, was shown to inhibit ZIKV replication in vitro, possibly by occupying the active site of the ZIKV-NS2B-NS3 protein. A fluorescence-based enzymatic assay also revealed that bromocriptine inhibits the activity of the ZIKV-NS2B-NS3 protease, possibly by occupying the active site pocket. In addition, bromocriptine, along with type I interferon, exhibits synergistic anti-ZIKV activity (Chan et al., 2017). Suramin is an approved drug used to treat trypanosomal human sleeping sickness and is available for prophylactic and therapeutic uses in children. It inhibits the early steps of ZIKV binding/entry and decreases the number of infectious ZIKV progeny virions (Albulescu et al., 2017; Tan et al., 2017). The safe pediatric anti-protozoan and anti-viral drug nitazoxanide was found to affect post-attachment steps of ZIKV infection at or below a 10 μM dose (Cao et al., 2017). The anti-ZIKV activity of nitazoxanide is not less than that of niclosamide (Xu M. et al., 2016), but poor absorption of niclosamide might reduce its utility.
Hyperactivation of the N-methyl-D-aspartate receptor (NMDAR), mediated by enhanced glutamate release, may result in the accumulation of high levels of Ca2+ in neurons, and this may further lead to apoptosis or necrosis of neural cells. Neurodegeneration in ZIKV disease possibly occurs due to the excitotoxicity of glutamate. FDA approved NMDAR antagonistic drugs to treat Alzheimer’s disease (namely memantine, MK-801, agmatine, and ifenprodil) were found to prevent neuronal cell death caused by ZIKV under in vitro conditions without reducing viral titers (Costa et al., 2017). Memantine was found to bind non-competitively with NMDAR, while blocking only the pathologically active NMDAR and leaving its physiological activity unaffected (Sirohi and Kuhn, 2017). Memantine is also listed in pregnancy category B drugs by the FDA, hence it could be used safely to reduce neurological complications associated with ZIKV infection (Sirohi and Kuhn, 2017). Practical usage of repurposing earlier approved drugs could help in formulating fascinating approaches to counter ZIKV and its associated complications for which purpose more research work is needed before moving into clinical trials.
Development of Pregnancy-Safe Drugs
The ability of ZIKV to infect fetuses and cause severe disease requires the development of drugs that function during pregnancy and that are safe for both the pregnant mother and fetus. The drugs must be able to cross the placental barrier to reach the fetus and to cross the blood-brain barrier to reach neural cells, the main targets of ZIKV. Khandia et al. (2017) summarized FDA-approved category B drugs (adequate animal study data shows no risk to fetuses, but controlled studies on pregnant women are unavailable) and category C drugs (animal studies revealed few teratogenic effects on fetuses, but control studies on pregnant women are unavailable; however, the potential benefits of using the drug may outweigh the risks). The list contains several drugs including the FDA category B drugs sofosbuvir (Sacramento et al., 2017), azithromycin (Retallack et al., 2016), niclosamide (Xu M. et al., 2016), palonosetron (Pascoalino et al., 2016), mefloquine (Balasubramanian et al., 2016), and daptomycin B (Barrows et al., 2016), category C drugs chloroquine (Delvecchio et al., 2016), amodiaquine, quinacrine hydrochloride (Balasubramanian et al., 2016), auranofin, clofazimine, deferasirox, methoxsalen, micafungin, sertraline-HCl, fingolimod, ivermectin, digoxin (Barrows et al., 2016), and seliciclib (Xu M. et al., 2016), which could be repurposed for treating ZIKV infection.
Use of Convalescent Serum
Recently, neutralizing activity of human convalescent serum against ZIKV has been demonstrated in a standard plaque reduction neutralization test (Li et al., 2016). Further, a decreased number of ZIKV-infected brain cells in ICR albino fetal mice were observed after treating pregnant mice intraperitoneally with convalescent serum. Furthermore, ZIKV-mediated caspase activity was reduced, indicating the utility of convalescent serum in limiting ZIKV infection and cell death. Convalescent serum also reversed thinning of the cortical plate (CP) and ventricular zone (VZ)/subventricular zone (SVZ) observed in the brains of ZIKV-infected fetal mice. Therefore, the use of convalescent serum for the treatment of ZIKV-infected pregnant women and whether it can protect against brain abnormalities in fetuses should be assessed. A study by Wang S. et al. (2016) demonstrated the suppression of ZIKV infection in pregnant mice with a reduction in caspase-3-activated cells using convalescent serum with high amounts of neutralizing antibodies. Convalescent serum also inhibited progenitor cell death in infected fetal brain tissue, thereby preventing microcephaly. ZIKV-confirmed convalescent human serum was able to neutralize multiple strains of infectious ZIKV or ZIKV RVPs, indicating that ZIKV is circulating as a single serotype (Wang S. et al., 2016). Additionally, antibodies present in convalescent serum can cross the placental, as well as the blood-brain barrier, of fetuses; thus, it is a good candidate for the treatment of infected pregnant women.
The convalescent serum should be free from ZIKV and other blood-borne pathogens prior to transfer, therefore heat treatment at 58.0 ± 1.0°C for 590 ± 10 min or solvent/detergent (S/D) treatment is commonly employed. S/D treatment with 1% (wt/wt) tri-n-butyl phosphate (TBP) and 1% (wt/wt) octoxynol-9 at 30.0 ± 1.0°C and pH 6.9–7.4 for 60 min completely inactivated ZIKV (Kühnel et al., 2017). A photochemical, amotosalen, quickly intercalates into DNA and RNA strands and forms covalent adducts with pyrimidine, thereby inhibiting replication and transcription of the virus. Plasma samples treated with amotosalen and UVA-light are safe for use in patients, as they are free from viable ZIKV particles, even in the presence of a detectable amount of ZIKV viral RNA (Aubry et al., 2016).
Use of Neutralizing Antibodies
A human monoclonal (mAb) antibody against DENV named C10 has been found to neutralize ZIKV E protein. Using an electron microscope, C10-ZIKV interactions were studied in extracellular (pH 8), early (pH 6.5), and late endosomal (pH 5.0) stages. At all of the tested pHs, C10 bound ZIKV E protein at different positions. At pH 8.0, it bound at the intradimer interface; at pH 6.5, it bound the virus surface; and at pH 5.0, it blocks raft structure. Of note, different structural rearrangements of the virus were blocked by C10 antibodies as depicted in visualization under electron microscope (Zhang S. et al., 2016), suggesting its broad applicability at different stage of infection. Out of the panel of human mAbs derived from patients previously infected with ZIKV, ZIKV-117 mAb was found to broadly neutralize the African and Asian-American lineages of ZIKV. The mAb recognized the unique quaternary epitope on the E protein dimer-dimer interface and effectively reduced ZIKV infection, maternal-to-fetal transfer, and tissue pathology and mortality (Sapparapu et al., 2016).
Three-dimensional cryo-electron microscopy showed that ZIKV-117 Fabs cross-link with monomers in surface E glycoprotein dimers and between neighboring dimers, thereby preventing the structural reorganization of E protein monomers and requiring the formation of fusogenic E protein trimers (Hasan et al., 2017). Neutralizing antibody 2A10G6, which targets the highly conserved fusion loop region of flavivirus E proteins, binds with high affinity and can neutralize ZIKV in a mouse model (Dai et al., 2016). Moreover, of 13 human mAbs from a single ZIKV patient, two mAbs (Z23 and Z3L1) potently bound and neutralized ZIKV, but it did not cross react with any DENV strains (Wang Q. et al., 2016).
Strategies to Limit Antibody-Dependent Enhancement (ADE)
Studies regarding the phenomenon of antibody-dependent enhancement (ADE), in which viremia is increased in the presence of pre-existing cross-reactive, poorly neutralizing antibodies against a heterologous flavivirus strain, are controversial. In few cases, pre-existing cross-reactive antibodies have shown to increase ADE, while in some experiments such antibodies exhibited therapeutic potential. For instance, preincubation of human myeloid cells (U937), which are poorly permissive for ZIKV, with convalescent serum obtained from subjects who resolved DENV infections, resulted in increased ZIKV infectivity (Dejnirattisai et al., 2016). These results were confirmed by Charles and Christofferson (2016), who used a DENV serotype 2-derived mAb (4G2) to demonstrate ADE in ZIKV infections. A contradictory finding is reported by Pantoja et al. (2017) who reported reduction in viremia in DENV-exposed rhesus macaques, when compared with naïve animals. Antibodies to WNV have also shown cross-reactivity with ZIKV E protein, resulting in viremias that were at least 35-fold higher than those of the controls, and studies to determine the role of anti-WNV antibodies in enhancing ZIKV revealed a pattern like that observed with anti-DENV antibodies. WNV may enhance ZIKV in vitro as well as in vivo; however, the amplitude of enhancement is less than that of DENV (Bardina et al., 2017). Because 4G2 is widely used as an anti-flavivirus mAb, the possibility of using this mAb for other flaviviruses such as JEV and YFV or to limit WNV mediated ADE of ZIKV should be considered.
The two EDI/II cross-reactive mAbs developed against ZIKV (ZKA78) and DENV (DV82) were tested for their capacity for ADE of DENV and ZIKV infection in animal models. Further, in an AG129 mouse model, wild-type mAbs ZKA78 and DV82 (without the LALA mutation), when administered prior to DENV-2 infection, resulted in severe disease and death of mice on the 5th day post-infection, suggesting that the DENV infections were affected by the presence of pre-existing ZIKV antibodies (Stettler et al., 2016). However, antibodies against the envelope dimer epitope 1 (EDE1) region were shown to neutralize ZIKV, in addition to all four DENV serotypes, indicating their potential immune-therapeutic potential in ZIKV infections (Swanstrom et al., 2016). The mouse mAb 2A10G6 reported to bind the conserved 98DRXW101 motif of the FL loop is a broadly neutralizing antibody. It not only neutralizes DENV1-4, but also protected A129 mice against ZIKV infection (Dai et al., 2016). Hence, it seems that the antigenic epitopes against which the antibody is generated is the deciding factor in developing/not developing ADE. The epitopes resulting in poor neutralization lead to ADE, while the strongly neutralizing antibodies have therapeutic potential. Information regarding common epitopes may be useful in determining strategies to limit ADE (Xu X. et al., 2016).
To develop therapeutic mAb candidates, LALA mutations in the Fc region of antibodies have been examined (Figure 3). The LALA mutations are a leucine (L)-to-alanine (A) substitution at positions 234 and 235 (LALA) in the Fc region of IgG antibody. The binding of the Fc region of an antibody with gamma receptors (FcγRs), expressed on various immune cells, triggers their effector function. In the case of ZIKV or DENV, the virus-antibody immune complex is internalized by FcγRs and can result in ADE. Introduction of the LALA mutations into the Fc region abolishes its ability to bind to FcγRs (Arduin et al., 2015). Such engineered antibodies are incapable of interaction with FcγRs and hence eliminate ADE from prior DENV infections in vitro and in vivo (Williams et al., 2013).
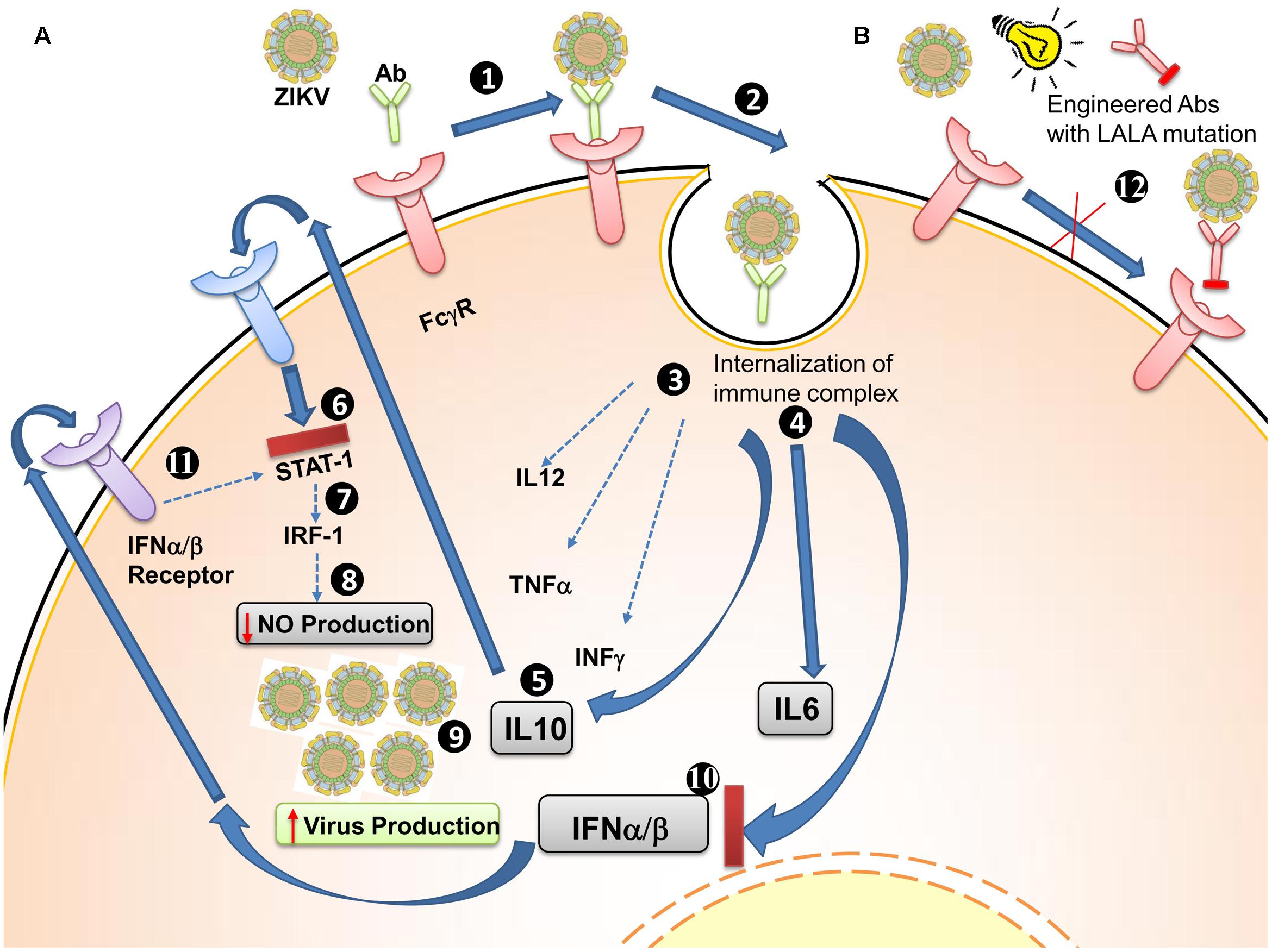
FIGURE 3. Antibody-dependent enhancement (ADE) of Zika virus (ZIKV) and strategies to limit it. (A) General ADE mechanism. (1) FcγRs are receptors present on the surface of various immune cells. Binding of antibody along with ZIKV results in (2) internalization of immune complexes into cells, (3) a reduction of IL12, TNFα, and IFNγ expression levels, and (4) increased levels of IL6 and IL12 (5). IL10 acts in an autocrine manner and binds to its own receptor (6), inhibiting the JAK-STAT pathway (7), which leads to reduced IRF1 production (8) and reduced IFN-stimulated response element (IRF1) production, resulting in decreased nitric oxide production. Nitric oxide, a diffusible radical antimicrobial and anti-viral, is reduced (9), which results in an increase in the number of infectious virus particles (10) IFNα/β are inhibited (11), diffuse out from the cell (12), and bind to their own receptors to reduce JAK-STAT signaling. (B) Antibodies engineered to prevent ADE (12) Engineered antibodies having LALA mutations in their Fc region are able to neutralize virus but fail to bind to FcγRs, thus preventing ADE.
Anti-ZIKV mAbs with the LALA mutation have been engineered for therapeutic and prophylactic purposes. The EDIII-specific neutralizing mAb ZKA64, which has the LALA mutations in the Fc region, blocked ADE in ZIKV infections in K652 cells in the presence of convalescent serum and completely protected A129 mice from lethal ZIKV challenge when administered one day prior to or post challenge (Stettler et al., 2016). Given the high potency and in vitro and in vivo efficacy of LALA mutant neutralizing antibodies, creation of such mutants appears to be a safe and promising approach to inhibit ADE in patients living in areas which co-circulating flaviviruses. The LALA mutations and substitution at amino acid position 297 (N297A) in the Fc region of an antibody reduces binding with FcγRs and C1q complement. Thus, such engineered antibodies may play an important role in therapeutics and prophylaxis (Arduin et al., 2015). DENV human mAb SIgN-3C, which strongly neutralizes ZIKV when the LALA mutations are introduced, did not induce ADE, showed a reduced viral load in fetal organs, and prevented virus-induced fetal growth retardation. This indicates the prophylactic potential of the antibody (Kam et al., 2017). In addition, mAbs such as Z23 and Z3L1 (mentioned in above section), which specifically neutralize ZIKV only and neither bind to nor neutralize any of the four DENV serotypes, are also of great importance in limiting ZIKV-associated ADE. In fact, a patent has been granted to Baehner et al. (2015), who modified the Fc region of human antibody, reducing its affinity for FcγRs by 1.15 to 100 folds and resulting in the inhibition of signaling cascades that lead to downstream immune response such as ADE in the case of DENV and ZIKV infection.
Use of Herbal Drugs
Herbal drugs are of increasing interest because of the development of anti-microbial resistance in microbes and owing to their cost effectiveness. Curcumin, a common food additive, is able to reduce ZIKV infectivity by hindering the virus binding to host cell in a dose- and time-dependent manner, without having adverse effects on cellular viability (Mounce et al., 2017). Recently, a Chinese semi-synthetic formulation from Andrographis paniculata named xiyanping, in combination with other antiviral and symptomatic treatments was administered to treat Zika fever in a patient admitted in Ganxian People’s Hospital. The patient recovered within 7 days after starting treatment that included this medication (Deng Y. et al., 2016). Quercetin, a flavonoid present in fruits, vegetables, leaves, and grains, has been found to inhibit Zika NS2B-NS3pro enzymatic activity in a dose-dependent manner. Commercially available quercetin has been reported to inhibit the ZIKV protease with an IC50 of 26.0 ± 0.1 μM (Roy et al., 2016).
In vitro studies with a polyphenol, (_)-epigallocatechingallate (EGCG), found in green tea have shown inhibition of ZIKV (Carneiro et al., 2016). The structures of ZIKV NS2B-NS3 protease, NS3 helicase, NS5 methyltransferase, and NS5-RdRp were generated by homology modeling using the BLOSUM80 scoring matrix by Byler et al. (2016) and molecular docking with virtual library of phytochemicals was carried out. Out of 2263 plant-derived secondary metabolites tested, 43 compounds docked with at least one of the ZIKV enzymatic proteins. Some of these include balsacone B from Populus balsamifera, kanzonol V from Glycyrrhiza glabra, cinnamoylechinaxanthol from Echinacea, cimiphenol from Actaea racemosa, and rosemarinic acid from Rosmarinus officinalis. Such common medicinal plants may serve as a source of herbal anti-virals. Such studies encourage the findings of structure-based drug (Byler et al., 2016). Several bioactive components, including alkaloids, flavonoids, saponins, tannins, terpenoids, essential oils, and herbs, such as Azidarachta indica and Tinospora cordifolia, have shown anti-flaviviral activities against DENV, JEV, and YFV infections. Such antiviral agents can also be explored for their efficacy against ZIKV and be used as complementary alternative medicine (Parida et al., 2002; Kiat et al., 2006; Meneses et al., 2009; Tang et al., 2012; Roy et al., 2015; Ching et al., 2016; Gómez-Calderón et al., 2017).
Compounds used for the treatment of various flaviviral diseases that are readily adaptable to ZIKV are presented in Table 1.
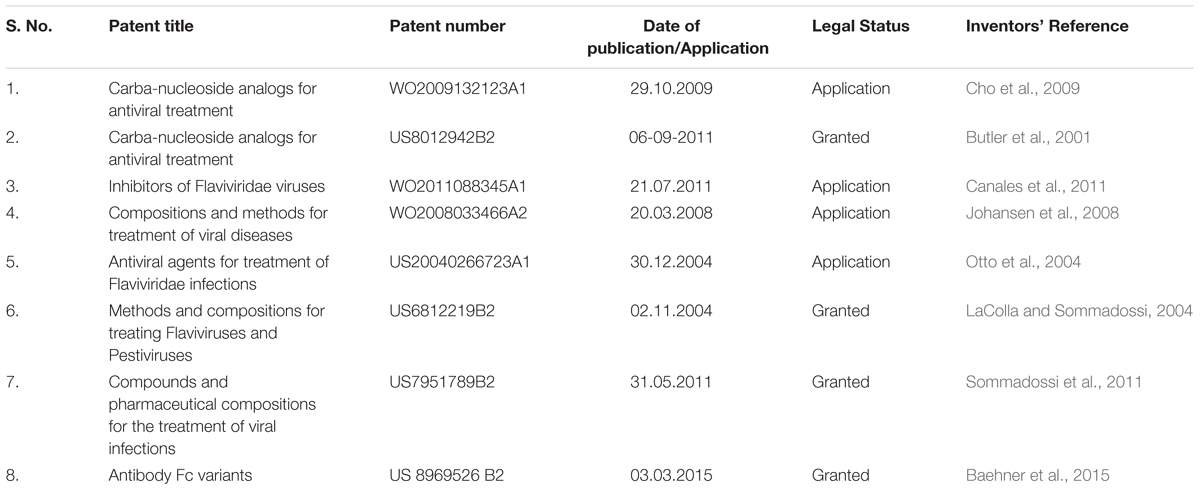
TABLE 1. Patents of novel innovations useful for the treatment of various flaviviral diseases [readily adaptable for Zika virus (ZIKV) treatment].
Other Strategies
Certain compounds such as QL-XII-47 and QL-XII-54 are quinolines (covalent inhibitors of DENV) that act by inhibiting viral E and NS5 protein expression without significantly affecting the host housekeeping protein GAPDH. Due to structural similarities between flaviviral E and NS5 proteins, similar compounds can be employed to combat ZIKV infection (de Wispelaere et al., 2016). Host caspases have been found to mediate the lethality of multiple pathogenic agents, based on the HapMap Project of B lymphoblastoid cells from a cohort of persons of African, European, and Asian ancestry. Bithionol (an FDA-approved drug) inhibits caspases and has been found to be effective in reducing the negative effects of ZIKV and bacterial and plant toxins. Thus, elucidation of such host proteins that further disease can be used to design drugs for ZIKV (Leonardi et al., 2016). Nordihydroguaiaretic acid (NDGA) alters the lipid metabolism of a host by intervening in the sterol regulatory element binding protein (SREBP) pathway. More recently, inhibitors of the SREBP pathway, including NDGA and its methylated derivative tetra-O-methyl nordihydroguaiaretic (M4N), PF-429242, and fatostatin, have also been found to reduce WNV and ZIKV replication. These drug candidates may serve as effective anti-viral agents against ZIKV (Merino-Ramos et al., 2017).
After ZIKV binds to nerve cells, Toll-like receptor (TLR)-3 is activated, leading to the dysregulation of genes participating in neurogenesis, axon guidance, and differentiation. TLR3 agonist poly (I:C) and thiophenecarboxamidopropionate compounds act as high-affinity competitive inhibitors of TLR3, and prevented a reduction in the size of ZIKV-treated neurospheres (Dang et al., 2016). Metadichol, a nanoemulsion of policosanols, binds to the Vit D receptor and stimulates the immune system. It can displace viruses bound to the Vit D receptor, thereby blocking viral entry into host cells. Metadichol has shown activity against ZIKV, EBOV, SARS coronavirus, JEV, WNV, and YFV. It is sold as a nutritional supplement in some Asian countries and is well tolerated; hence, it can be used as a safe and broad-spectrum anti-viral agent (Raghavan, 2016). Similarly, testing of other broad-spectrum anti-viral agents for anti-ZIKV activity will aid in rapid drug discovery to combat this virus.
An overview of recent advances in the design of drugs and therapies for ZIKV is presented in Table 2.
A few recent therapies include cytokines, TLRs, siRNA, RNA interference, probiotics, immunomodulatory interventions, and nanodrug delivery. These approaches have gained momentum and are being examined for optimum benefit and safety in humans and their companion animals. Prospective aspects of these valuable therapies could be given a due focus for designing and developing effective drugs, medicines, therapeutics and immunomodulatory pharmaceuticals for the treatment of ZIKV infections. Molecular and genetic analyses for a more in-depth understanding of ZIKV pathogenesis would facilitate the identification of novel targets and development of safer and more effective drugs to counter ZIKV effectively.
Conclusion and Future Perspectives
Zika virus, an arbovirus, shares several characteristic features with other members of the Flavivirus family Recent evidences of autoimmune complications (GBS) and maternal-to-fetal transmission of virus leading to microcephaly has accumulated. Using state-of-the-art methods to formulate effective diagnostics, anti-viral drugs, therapeutics, vaccines, and prevention and control strategies would aid in addressing this emergent virus. Some recent therapies have shown promise in inhibiting ZIKV infections and associated disease. These therapies include limiting viral entry into cells, targeting the ZIKV helicase protein, use of nucleoside analogs like 2′-C-methylated nucleosides and 7-deaza-2′-C-methyladenosine to terminate nascent RNA strand formation, and use of antibodies that bind to ZIKV but do not neutralize it, reducing the risk of ADE. ADE is of major concern in the application of ZIKV therapies in geographical regions where other flaviviruses are endemic. Thus, to limit ADE, antibodies are being engineering to contain a modified Fc region. Modification of the Fc region of antibodies not only hampers their attachment to FcγRs to inhibit internalization of the immune complex, but also reduces complement binding, preventing ADE. In the future, several such mutations may be identified, and humanized mAbs can be genetically engineered to prevent ADE. Combination use of such engineered antibodies might be evaluated for synergistic effects in other therapeutic and prophylactic regimens.
Encouraging results with repurposed drugs, as shown by the use of chloroquine, a malaria drug, has led to the screening of several other FDA-approved drugs, including niclosamide, emricasan, and daptomycin, palonosetron, kitasamycin, and many more, for ZIKV treatment. Another valuable strategy for the discovery of ZIKV preventives and anti-virals is the use of computational analysis.
More insights into genetic and molecular mechanisms associated with the recent increase in virulence of ZIKV could aid in the design and development of safer and more potent drugs and therapeutics against ZIKV. Along with identifying novel drug targets, therapeutics, and vaccines, strengthening of appropriate prevention and control measures, including mosquito control, could help in limiting ZIKV infections, its associated complications, and its potential for further spread. It is time for researchers, pharmaceutical companies, policy makers, regulators, and funding agencies to identify and implement strategies to counter ZIKV globally.
What We Are Still Lacking
Since the declaration of the Zika epidemic as an international public health emergency by WHO in 2016, research on ZIKV has increased many fold. However, there are areas that still need to be addressed.
(1) The percent contribution of each route of ZIKV infection is not precisely understood. Presently, based on mathematical modeling study, sexual transmission has been estimated to account for upto 3% of transmission, but contributions by other routes of infection are yet to be studied. This knowledge may be helpful in designing precisely targeted inhibitory molecules to block infection at site of entry.
(2) Many FDA approved drugs have been tested for efficacy against ZIKV; which can be repurposed for treating ZIKV infection in human. However, to date, no FDA category A drug has been identified clinically safe for use in mothers and fetuses.
(3) For engineered mAb, only two mutations that prevent internalization of immune complexes, i.e., LALA and N297A substitutions, have been identified. More such mutations must be identified for optimal efficacy and synergism.
Author Contributions
All the authors substantially contributed to the conception, design, analysis and interpretation of data, checking and approving final version of manuscript, and agree to be accountable for its contents. AM and RK initiated this review compilation; KD reviewed, analyzed, and edited; RK designed tables; AM, RK, and KK designed the figures; SS and RT covered critical aspects on drug and vaccine development; YM and RS reviewed virological aspects and analyzed data; DK reviewed biotechnological and bioinformatics advances; HI and SK overviewed immunotherapeutic aspects and drug development.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgment
All the authors acknowledge and thank their respective Institutes and Universities.
Footnotes
References
Adcock, R. S., Chu, Y. K., Golden, J. E., and Chung, D. H. (2017). Evaluation of anti-Zika virus activities of broad-spectrum antivirals and NIH clinical collection compounds using a cell-based, high-throughput screen assay. Antiviral Res. 138, 47–56. doi: 10.1016/j.antiviral.2016.11.018
Aid, M., Abbink, P., Larocca, R. A., Boyd, M., Nityanandam, R., Nanayakkara, O., et al. (2017). Zika virus persistence in the central nervous system and lymph nodes of rhesus monkeys. Cell 169, 610–620.e14. doi: 10.1016/j.cell.2017.04.008
Akpovwa, H. (2016). Chloroquine could be used for the treatment of filoviral infections and other viral infections that emerge or emerged from viruses requiring an acidic pH for infectivity. Cell Biochem. Funct. 34, 191–196. doi: 10.1002/cbf.3182
Alam, A., Imam, N., Farooqui, A., Ali, S., Malik, M. Z., and Ishrat, R. (2017). Recent trends in ZikV research: a step away from cure. Biomed. Pharmacother. 91, 1152–1159. doi: 10.1016/j.biopha.2017.05.045
Albulescu, I. C., Kovacikova, K., Tas, A., Snijder, E. J., and van Hemert, M. J. (2017). Suramin inhibits Zika virus replication by interfering with virus attachment and release of infectious particles. Antiviral Res. 143, 230–236. doi: 10.1016/j.antiviral.2017.04.016
Alexandre, K. B., Gray, E. S., Pantophlet, R., Moore, P. L., McMahon, J. B., Chakauya, E., et al. (2011). Binding of the mannose-specific lectin, griffithsin, to HIV-1 gp120 exposes the CD4-binding site. J. Virol. 85, 9039–9050. doi: 10.1128/JVI.02675-10
Ali, A., Wahid, B., Rafique, S., and Idrees, M. (2017). Advances in research on Zika virus. Asian Pac. J. Trop. Med. 10, 321–331. doi: 10.1016/j.apjtm.2017.03.020
Apostol, J. G., Gan, J. V. A., Raynes, R. J. B., Sabado, A. A. S., Carigma, A. Q., Santiago, L. A., et al. (2012). Platelet-increasing effects of Euphorbia hirta Linn. (Euphorbiaceae) in ethanol-induced thrombocytopenic rat models. Int. J. Pharm. Front. Res. 2, 1–11.
Arduin, E., Arora, S., Bamert, P. R., Kuiper, T., Popp, S., Geisse, S., et al. (2015). Highly reduced binding to high and low affinity mouse Fc gamma receptors by L234A/L235A and N297A Fc mutations engineered into mouse IgG2a. Mol. Immunol. 63, 456–463. doi: 10.1016/j.molimm.2014.09.017
Aubry, M., Richard, V., Green, J., Broult, J., and Musso, D. (2016). Inactivation of Zika virus in plasma with amotosalen and ultraviolet A illumination. Transfusion 56, 33–40. doi: 10.1111/trf.13271
Baehner, M., Jenewein, S., Kubbies, M., Moessner, E., and Schlothaue, T. (2015). Antibody Fc variants. US 8969526 B2. Washington, DC: U.S. Patent and Trademark Office.
Balasubramanian, A., Teramoto, T., Kulkarni, A. A., Bhattacharjee, A. K., and Padmanabhan, R. (2016). Antiviral activities of selected antimalarials against dengue virus type 2 and Zika virus. Antiviral Res. 137, 141–150. doi: 10.1016/j.antiviral.2016.11.015
Bardina, S. V., Bunduc, P., Tripathi, S., Duehr, J., Frere, J. J., Brown, J. A., et al. (2017). Enhancement of Zika virus pathogenesis by preexisting antiflavivirus immunity. Science 356, 175–180. doi: 10.1126/science.aal4365
Barreyro, F. J., Holod, S., Finocchietto, P. V., Camino, A. M., Aquino, J. B., Avagnina, A., et al. (2015). The pan-caspase inhibitor Emricasan (IDN-6556) decreases liver injury and fibrosis in a murine model of non-alcoholic steatohepatitis. Liver Int. 35, 953–966. doi: 10.1111/liv.12570
Barrows, N. J., Campos, R. K., Powell, S. T., Reddisiva Prasanth, K., Schott-Lerner, G., Soto-Acosta, R., et al. (2016). A screen of FDA-approved drugs for inhibitors of Zika virus infection. Cell Host Microbe 20, 259–270. doi: 10.1016/j.chom.2016.07.004
Basak, S. C., and Nandy, A. (2016). Computer-assisted approaches as decision support systems in the overall strategy of combating emerging diseases: some comments regarding drug design, vaccinomics, and genomic surveillance of the Zika virus. Curr. Comput. Aided Drug Des. 12, 2–4. doi: 10.2174/1573409912999160315115502
Bayer, A., Lennemann, N. J., Ouyang, Y., Bramley, J. C., Morosky, S., Marque, S. E. T. Jr., et al. (2016). Type III interferons produced by human placental trophoblasts confer protection against Zika virus infection. Cell Host Microbe 19, 705–712. doi: 10.1016/j.chom.2016.03.008
Bowen, J. R., Quicke, K. M., Maddur, M. S., O’Neal, J. T., McDonald, C. E., Fedorova, N. B., et al. (2017). Zika virus antagonizes type I interferon responses during infection of human dendritic cells. PLoS Pathog. 13:e1006164. doi: 10.1371/journal.ppat.1006164
Bullard-Feibelman, K. M., Govero, J., Zhu, Z., Salazar, V., Veselinovic, M., Diamond, M. S., et al. (2017). The FDA-approved drug sofosbuvir inhibits Zika virus infection. Antiviral Res. 137, 134–140. doi: 10.1016/j.antiviral.2016.11.023
Butler, T., Cho, A., Kim, C. U., and Xu, J. (2001). Carba-nucleoside analogs for antiviral treatment. US8012942B2. Washington, DC: U.S. Patent and Trademark Office.
Byler, K. G., Ogungbe, I. V., and Setzer, W. N. (2016). In-silico screening for anti-Zika virus phytochemicals. J. Mol. Graph. Model. 69, 78–91. doi: 10.1016/j.jmgm.2016.08.011
Byrd, C. M., Dai, D., Grosenbach, D. W., Berhanu, A., Jones, K. F., Cardwell, K. B., et al. (2013). A novel inhibitor of dengue virus replication that targets the capsid protein. Antimicrob. Agents Chemother. 57, 15–25. doi: 10.1128/AAC.01429-12
Cai, L., Sun, Y., Song, Y., Xu, L., Bei, Z., Zhang, D., et al. (2017). Viral polymerase inhibitors T-705 and T-1105 are potential inhibitors of Zika virus replication. Arch. Virol. doi: 10.1007/s00705-017-3436-8 [Epub ahead of print].
Canales, E., Clarke, M. O. H., Lazerwith, S. E., Lew, W., Morganelli, P. A., and Watkins, W. J. (2011). Inhibitors of flaviviridae viruses. WO2011088345A1. Washington, DC: U.S. Patent and Trademark Office.
Cao, R. Y., Xu, Y. F., Zhang, T. H., Yang, J. J., Yuan, Y., Hao, P., et al. (2017). Pediatric drug nitazoxanide: a potential choice for control of Zika. Open Forum Infect. Dis. 4, ofx009. doi: 10.1093/ofid/ofx009
Cao, X., Li, Y., Jin, X., Li, Y., Guo, F., and Jin, T. (2016). Molecular mechanism of divalent-metal-induced activation of NS3 helicase and insights into Zika virus inhibitor design. Nucleic Acids Res. 44, 10505–10514. doi: 10.1093/nar/gkw941
Cao-Lormeau, V. M., Blake, A., Mons, S., Lastère, S., Roche, C., Vanhomwegen, J., et al. (2016). Guillain-Barré Syndrome outbreak associated with Zika virus infection in French Polynesia: a case-control study. Lancet 387, 1531–1539. doi: 10.1016/S0140-6736(16)00562-6
Carneiro, B. M., Batista, M. N., Braga, A. C. S., Nogueira, M. L., and Rahal, P. (2016). The green tea molecule EGCG inhibits Zika virus entry. Virology 496, 215–218. doi: 10.1016/j.virol.2016.06.012
Carteaux, G., Maquart, M., Bedet, A., Contou, D., Brugières, P., Fourati, S., et al. (2016). Zika Virus Associated with Meningoencephalitis. N. Engl. J. Med. 374, 1595–1596. doi: 10.1056/NEJMc1602964
Chan, J. F. W., Chik, K. K. H., Yuan, S., Yip, C. C. Y., Zhu, Z., Tee, K. M., et al. (2017). Novel antiviral activity and mechanism of bromocriptine as a Zika virus NS2B-NS3 protease inhibitor. Antiviral Res. 141, 29–37. doi: 10.1016/j.antiviral.2017.02.002
Charles, A. S., and Christofferson, R. C. (2016). Utility of a dengue-derived monoclonal antibody to enhance Zika infection in vitro. PLoS Curr. Outbreaks. doi: 10.1371/currents.outbreaks.4ab8bc87c945eb41cd8a49e127082620
Chen, L., Liu, Y., Wang, S., Sun, J., Wang, P., Xin, Q., et al. (2017). Antiviral activity of peptide inhibitors derived from the protein E stem against Japanese encephalitis and Zika viruses. Antiviral Res. 141, 140–149. doi: 10.1016/j.antiviral.2017.02.009
Cheng, F., Murray, J. L., and Rubin, D. H. (2016). Drug repurposing: new treatments for Zika virus infection? Trends Mol. Med. 22, 919–921. doi: 10.1016/j.molmed.2016.09.006
Ching, S., Ramachandran, V., Gew, L. T., Lim, S. M. S., Sulaiman, W. A. W., Foo, Y. L., et al. (2016). Complementary alternative medicine use among patients with dengue fever in the hospital setting: a cross-sectional study in Malaysia. BMC Complement. Altern. Med. 16(1), 37. doi: 10.1186/s12906-016-1017-0
Cho, A., Choung, U., Kim, C. U., Parrish, J., and Xu, J. (2009). Carba-nucleoside analogs for antiviral treatment. WO2009132123A1. Washington, DC: U.S. Patent and Trademark Office.
Coffey, L. L., Pesavento, P. A., Keesler, R. I., Singapuri, A., Watanabe, J., Watanabe, R., et al. (2017). Zika virus tissue and blood compartmentalization in acute infection of rhesus macaques. PLoS ONE 12:e0171148. doi: 10.1371/journal.pone.0171148
Contreras, D., and Arumugaswami, V. (2016). Zika virus infectious cell culture system and the in vitro prophylactic effect of interferons. J. Vis. Exp. 114, e54767. doi: 10.3791/54767
Costa, V. V., Sarto, J. L. D., Rocha, R. F., Silva, F. R., Doriac, J. G., Olmo, I. G., et al. (2017). N-Methyl-d-Aspartate (n.d.) receptor blockade prevents neuronal death induced by zika virus infection. MBio 8, e00350–17. doi: 10.1128/mBio.00350-17
Coutard, B., Barral, K., Lichière, J., Selisko, B., Martin, B., Aouadi, W., et al. (2016). The Zika virus methyltransferase: structure and functions for drug design perspectives. J. Virol. 91, e02202–16. doi: 10.1128/JVI.02202-16
Cox, B. D., Stanton, R. A., and Schinazi, R. F. (2016). Predicting Zika virus structural biology: challenges and opportunities for intervention. Antivir. Chem. Chemother. 24, 118–126. doi: 10.1177/2040206616653873
Crance, J. M., Scaramozzino, N., Jouan, A., and Garin, D. (2003). Interferon, ribavirin, 6-azauridine and glycyrrhizin: antiviral compounds active against pathogenic flaviviruses. Antiviral Res. 58, 73–79. doi: 10.1016/S0166-3542(02)00185-7
Dai, L., Song, J., Lu, X., Deng, Y. Q., Musyoki, A. M., Cheng, H., et al. (2016). Structures of the Zika virus envelope protein and its complex with a flavivirus broadly protective antibody. Cell Host Microbe 19, 696–704. doi: 10.1016/j.chom.2016.04.013
Dang, J., Tiwari, S. K., Lichinchi, G., Qin, Y., Patil, V. S., Eroshkin, A. M., et al. (2016). Zika virus depletes neural progenitors in human cerebral organoids through activation of the innate immune receptor TLR3. Cell Stem Cell 19, 258–265. doi: 10.1016/j.stem.2016.04.014
Dar, H. A., Zaheer, T., Paracha, R. Z., and Ali, A. (2017). Structural analysis and insight into Zika virus NS5 mediated interferon inhibition. Infect. Genet. Evol. 51, 143–152. doi: 10.1016/j.meegid.2017.03.027
de Melo Freire, C. C., Iamarino, A., de Lima Neto, D. F., and de Andrade Zanotto, P. M. (2015). Spread of the pandemic Zika virus lineage is associated with NS1 codon usage adaptation in humans. bioRxiv. doi: 10.1101/032839
de Wispelaere, M., Carocci, M., Liang, Y., Liu, Q., Sun, E., Vetter, M. L., et al. (2016). Discovery of host-targeted covalent inhibitors of dengue virus. Antiviral Res. 139, 171–179. doi: 10.1016/j.antiviral.2016.12.017
Dejnirattisai, W., Supasa, P., Wongwiwat, W., Rouvinski, A., Barba-Spaeth, G., Duangchinda, T., et al. (2016). Dengue virus sero-cross-reactivity drives antibody-dependent enhancement of infection with zika virus. Nat. Immunol. 17, 1102–1108. doi: 10.1038/ni.3515
Delvecchio, R., Higa, L. M., Pezzuto, P., Valadão, A. L., Garcez, P. P., Monteiro, F. L., et al. (2016). Chloroquine, an endocytosis blocking agent, inhibits Zika virus infection in different cell models. Viruses 8:322. doi: 10.3390/v8120322
Deng, Y. Q., Zhang, N. N., Li, C. F., Tian, M., Hao, J. N., Xie, X. P., et al. (2016). Adenosine analog NITD008 is a potent inhibitor of Zika virus. Open Forum Infect. Dis. 3, ofw175. doi: 10.1093/ofid/ofw175
Deng, Y., Zeng, L., Bao, W., Xu, P., and Zhong, G. (2016). Experience of integrated traditional Chinese and Western medicine in first case of imported Zika virus disease in China. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue 28, 106–109. doi: 10.3760/cma.j.issn.2095-4352.2016.02.005
Doller, A., Akool, E. S., Müller, R., Gutwein, P., Kurowski, C., Pfeilschifter, J., et al. (2007). Molecular mechanisms of cyclosporin A inhibition of the cytokine-induced matrix metalloproteinase-9 in glomerular mesangial cells. J. Am. Soc. Nephrol. 18, 581–592. doi: 10.1681/ASN.2006060568
Duan, W., Song, H., Wang, H., Chai, Y., Su, C., Qi, J., et al. (2017). The crystal structure of Zika virus NS5 reveals conserved drug targets. EMBO J. 36, 919–933. doi: 10.15252/embj.201696241
Egloff, M. P., Benarroch, D., Selisko, B., Romette, J. L., and Canard, B. (2002). An RNA cap (nucleoside-2′-O)-methyltransferase in the flavivirus RNA polymerase NS5: crystal structure and functional characterization. EMBO J. 21, 2757–2768. doi: 10.1093/emboj/21.11.2757
Ekins, S., Perryman, A. L., and Andrade, C. H. (2016). OpenZika: an IBM world community grid project to accelerate Zika virus drug discovery. PLoS Negl. Trop. Dis. 10:e0005023. doi: 10.1371/journal.pntd.0005023
Estoppey, D., Lee, C. M., Janoschke, M., Lee, B. H., Wan, K. F., Dong, H., et al. (2017). The natural product Cavinafungin selectively interferes with Zika and Dengue virus replication by inhibition of the host signal peptidase. Cell Rep. 19, 451–460. doi: 10.1016/j.celrep.2017.03.071
Eyer, L., Nencka, R., Huvarová, I., Palus, M., Alves, M. J., Gould, E. A., et al. (2016). Nucleoside inhibitors of Zika virus. J. Infect. Dis. 214, 707–711. doi: 10.1093/infdis/jiw226
Fajardo, A., Cristina, J., and Moreno, P. (2016). Emergence and spreading potential of Zika virus. Front. Microbiol. 7:1667. doi: 10.3389/fmicb.2016.01667
Feranchuk, S., Potapova, U., and Belikov, S. (2016). Virtual Screening of Inhibitors for the Zika Virus Proteins. Available at: http://biorxiv.org/content/biorxiv/early/2016/06/27/060798.full.pdf. doi: 10.1101/060798
Fernando, S., Fernando, T., Stefanik, M., Eyer, L., and Ruzek, D. (2016). An approach for Zika virus inhibition using homology structure of the envelope protein. Mol. Biotechnol. 58, 801–806. doi: 10.1007/s12033-016-9979-1
Foy, B. D., Kobylinski, K. C., Chilson Foy, J. L., Blitvich, B. J., Travassos da Rosa, A., Haddow, A. D., et al. (2011). Probable non-vector-borne transmission of Zika virus, Colorado, USA. Emerg. Infect. Dis. 17, 880–882. doi: 10.3201/eid1705.101939
Furtado, J. M., Espósito, D. L., Klein, T. M., Teixeira-Pinto, T., and da Fonseca, B. A. (2016). Uveitis associated with Zika virus infection. N. Engl. J. Med. 375, 394–396. doi: 10.1056/NEJMc1603618
Gao, D., Lou, Y., He, D., Porco, T. C., Kuang, Y., Chowell, G., et al. (2016). Prevention and control of Zika as a mosquito-borne and sexually transmitted disease: a mathematical modeling analysis. Sci. Rep. 6:28070. doi: 10.1038/srep28070
Goebel, S., Snyder, B., Sellati, T., Saeed, M., Ptak, R., Murray, M., et al. (2016). A sensitive virus yield assay for evaluation of Antivirals against Zika Virus. J. Virol. Methods 238, 13–20. doi: 10.1016/j.jviromet.2016.09.015
Gómez-Calderón, C., Mesa-Castro, C., Robledo, S., Gómez, S., Bolivar-Avila, S., Diaz-Castillo, F., et al. (2017). Antiviral effect of compounds derived from the seeds of Mammea americana and Tabernaemontana cymosa on Dengue and Chikungunya virus infections. BMC Complement. Altern. Med. 17:57. doi: 10.1186/s12906-017-1562-1
Gourinat, A. C., O’Connor, O., Calvez, E., Goarant, C., and Dupont-Rouzeyrol, M. (2015). Detection of Zika Virus in Urine. Emerg. Infect. Dis. 21, 84–86. doi: 10.3201/eid2101.140894
Govero, J., Esakky, P., Scheaffer, S. M., Fernandez, E., Drury, A., Platt, D. J., et al. (2016). Zika virus infection damages the testes in mice. Nature 540, 438–442. doi: 10.1038/nature20556
Grant, A., Ponia, S. S., Tripathi, S., Balasubramaniam, V., Miorin, L., Sourisseau, M., et al. (2016). Zika virus targets human STAT2 to inhibit type I interferon signaling. Cell Host Microbe 19, 882–890. doi: 10.1016/j.chom.2016.05.009
Gupta, A. K., Kaur, K., Rajput, A., Dhanda, S. K., Sehgal, M., Khan, M. S., et al. (2016). ZikaVR: an integrated Zika virus resource for genomics, proteomics, phylogenetic and therapeutic analysis. Sci Rep. 6:32713. doi: 10.1038/srep32713
Hamel, R., Dejarnac, O., Wichit, S., Ekchariyawat, P., Neyret, A., Luplertlop, N., et al. (2015). Biology of Zika virus infection in human skin cells. J. Virol. 89, 8880–8896. doi: 10.1128/JVI.00354-15
Hamer, D. H., Wilson, M. E., Jean, J., and Chen, L. H. (2017). Epidemiology, prevention, and potential future treatments of sexually transmitted Zika virus infection. Curr. Infect. Dis. Rep. 19, 16. doi: 10.1007/s11908-017-0571-z
Hasan, S. S., Miller, A., Sapparapu, G., Fernandez, E., Klose, T., Long, F., et al. (2017). A human antibody against Zika virus crosslinks the E protein to prevent infection. Nat. Commun. 8:14722. doi: 10.1038/ncomms14722
Hercik, K., Brynda, J., Nencka, R., and Boura, E. (2017). Structural basis of Zika virus methyltransferase inhibition by sinefungin. Arch. Virol. 162, 2091–2096. doi: 10.1007/s00705-017-3345-x
Hirsch, A. J., Smith, J. L., Haese, N. N., Broeckel, R. M., Parkins, C. J., Kreklywich, C., et al. (2017). Zika Virus infection of rhesus macaques leads to viral persistence in multiple tissues. PLoS Pathog. 13:e1006219. doi: 10.1371/journal.ppat.1006219
Jemielity, S., Wang, J. J., Chan, Y. K., Ahmed, A. A., Li, W., Monahan, S., et al. (2013). TIM-family proteins promote infection of multiple enveloped viruses through virion-associated phosphatidylserine. PLoS Pathog. 9:e1003232. doi: 10.1371/journal.ppat.1003232
Jiang, W. L., Luo, X. L., and Kuang, S. J. (2005). Effects of Alternanthera philoxeroides Griseb against dengue virus in vitro. Di Yi Jun Yi Da Xue Xue Bao 25, 454–456.
Johansen, L. M., Owens, C. M., Mawhinney, C., Chappell, T. W., Brown, A. T., Frank, M. G., et al. (2008). Compositions and methods for treatment of viral diseases. WO2008033466A2. Washington, DC: U.S. Patent and Trademark Office.
Kam, Y. W., Lee, C. Y., Teo, T. H., Howland, S. W., Amrun, S. N., Lum, F. M., et al. (2017). Cross-reactive dengue human monoclonal antibody prevents severe pathologies and death from Zika virus infections. JCI Insight 2:e92428. doi: 10.1172/jci.insight.92428
Khandia, R., Munjal, A., and Dhama, K. (2017). Consequences of Zika virus infection during fetal stage and pregnancy safe drugs: an update. Int. J. Pharmacol. 14, 370–377. doi: 10.3923/ijp.2017.370.377
Kiat, T. S., Pippen, R., Yusof, R., Ibrahim, H., Khalid, N., and Rahman, N. A. (2006). Inhibitory activity of cyclohexenyl chalcone derivatives and flavonoids of fingerroot, Boesenbergia rotunda (L.), towards dengue-2 virus NS3 protease. Bioorg. Med. Chem. Lett. 16, 3337–3340. doi: 10.1016/j.bmcl.2005.12.075
Kok, W. M. (2016). New developments in flavivirus drug discovery. Expert Opin. Drug Discov. 11, 433–445. doi: 10.1517/17460441.2016.1160887
Kouznetsova, J., Sun, W., Martínez-Romero, C., Tawa, G., Shinn, P., Chen, C. Z., et al. (2014). Identification of 53 compounds that block Ebola virus-like particle entry via a repurposing screen of approved drugs. Emerg. Microbes Infect. 3:e84. doi: 10.1038/emi.2014.88
Kühnel, D., Müller, S., Pichotta, A., Radomski, K. U., Volk, A., and Schmidt, T. (2017). Inactivation of Zika virus by solvent/detergent treatment of human plasma and other plasma-derived products and pasteurization of human serum albumin. Transfusion 57, 802–810. doi: 10.1111/trf.13964
LaColla, P., and Sommadossi, J. P. (2004). Methods and compositions for treating flaviviruses and pestiviruses. US6812219B2. Washington, DC: U.S. Patent and Trademark Office.
Lee, H., Ren, J., Nocadello, S., Rice, A. J., Ojeda, I., Light, S., et al. (2016). Identification of novel small molecule inhibitors against NS2B/NS3 serine protease from Zika virus. Antiviral Res. 139, 49–58. doi: 10.1016/j.antiviral.2016.12.016
Lei, J., Hansen, G., Nitsche, C., Klein, C. D., Zhang, L., and Hilgenfeld, R. (2016). Crystal structure of Zika virus NS2B-NS3 protease in complex with a boronate inhibitor. Science 353, 503–505. doi: 10.1126/science.aag2419
Lennard, L. (1992). The clinical pharmacology of 6-mercaptopurine. Eur. J. Clin. Pharmacol. 43, 329–339. doi: 10.1007/BF02220605
Leonardi, W., Zilbermintz, L., Cheng, L. W., Zozaya, J., Tran, S. H., Elliott, J. H., et al. (2016). Bithionol blocks pathogenicity of bacterial toxins, ricin, and Zika virus. Sci. Rep. 6:34475. doi: 10.1038/srep34475
Levendosky, K., Mizenina, O., Martinelli, E., Jean-Pierre, N., Kizima, L., Rodriguez, A., et al. (2015). Griffithsin and carrageenan combination to target Herpes Simplex Virus 2 and Human Papillomavirus. Antimicrob. Agents Chemother. 59, 7290–7298. doi: 10.1128/AAC.01816-15
Li, C., Deng, Y. Q., Wang, S., Ma, F., Aliyari, R., Huang, X. Y., et al. (2017). 25-Hydroxycholesterol protects host against Zika virus infection and its associated microcephaly in a mouse model. Immunity 46, 446–456. doi: 10.1016/j.immuni.2017.02.012
Li, H., Saucedo-Cuevas, L., Regla-Nava, J. A., Chai, G., Sheets, N., Tang, W., et al. (2016). Zika virus infects neural progenitors in the adult mouse brain and alters proliferation. Cell Stem Cell 19, 593–598. doi: 10.1016/j.stem.2016.08.005
Lim, H. J., Nguyen, T. T., Kim, N. M., Park, J. S., Jang, T. S., and Kim, D. (2017). Inhibitory effect of flavonoids against NS2B-NS3 protease of ZIKA virus and their structure activity relationship. Biotechnol. Lett. 39, 415–421. doi: 10.1007/s10529-016-2261-6
Lim, L., Roy, A., and Song, J. (2016). Identification of a Zika NS2B-NS3pro pocket susceptible to allosteric inhibition by small molecules including qucertin rich in edible plants. bioRxiv. doi: 10.1101/078543
Lu, G., Bluemling, G. R., Collop, P., Hager, M., Kuiper, D., Gurale, B. P., et al. (2016). Analysis of Ribonucleotide 5′ -Triphosphate analogs as potential inhibitors of Zika virus RNA-dependent RNA polymerase using non-radioactive polymerase assays. Antimicrob. Agents Chemother. 61, e01967–16. doi: 10.1128/AAC.01967-16AAC.01967-16
Mangia, A., and Piazzolla, V. (2014). Overall efficacy and safety results of sofosbuvir-based therapies in Phase II and III studies. Dig. Liver Dis. 46, 179–185. doi: 10.1016/j.dld.2014.09.026
Martines, R. B., Bhatnagar, J., Keating, M. K., Silva-Flannery, L., Muehlenbachs, A., Gary, J., et al. (2016). Notes from the Field: evidence of Zika virus infection in brain and placental tissues from two congenitally infected newborns and two fetal losses - Brazil, 2015. MMWR Morb. Mortal. Wkly. Rep. 65, 159–160. doi: 10.15585/mmwr.mm6506e1
Mastrangelo, E., Pezzullo, M., De Burghgraeve, T., Kaptein, S., Pastorino, B., Dallmeier, K., et al. (2012). Ivermectin is a potent inhibitor of flavivirus replication specifically targeting NS3 helicase activity: new prospects for an old drug. J. Antimicrob. Chemother. 67, 1884–1894. doi: 10.1093/jac/dks147
Meneses, R., Ocazionez, R. E., Martínez, J. R., and Stashenko, E. E. (2009). Inhibitory effect of essential oils obtained from plants grown in Colombia on yellow fever virus replication in vitro. Ann. Clin. Microbiol. Antimicrob. 8:8. doi: 10.1186/1476-0711-8-8
Merino-Ramos, T., Jiménez de Oya, N., Saiz, J. C., and Martín-Acebes, M. A. (2017). Antiviral activity of nordihydroguaiaretic acid and its derivative tetra-O-methyl nordihydroguaiaretic acid against West Nile virus and Zika virus. Antimicrob. Agents Chemother. doi: 10.1128/AAC.00376-17
Miner, J. J., and Diamond, M. S. (2017). Zika virus pathogenesis and tissue tropism. Cell Host Microbe 21, 134–142. doi: 10.1016/j.chom.2017.01.004
Mlakar, J., Korva, M., Tul, N., Popović, M., Poljšak-Prijatelj, M., Mraz, J., et al. (2016). Zika virus associated with microcephaly. N. Engl. J. Med. 374, 951–958. doi: 10.1056/NEJMoa1600651
Mounce, B. C., Cesaro, T., Carrau, L., Vallet, T., and Vignuzzi, M. (2017). Curcumin inhibits Zika and chikungunya virus infection by inhibiting cell binding. Antiviral Res. 142, 148–157. doi: 10.1016/j.antiviral.2017.03.014
Mukherjee, S., and Era, N. (2016). Zika virus disease: global concerns and making way through it. Communit. Acquir. Infect. 3, 31. doi: 10.4103/2225-6482.184908
Musso, D., and Gubler, D. J. (2016). Zika virus. Clin. Microbiol. Rev. 29, 487–524. doi: 10.1128/CMR.00072-15
Nixon, B., Stefanidou, M., Mesquita, P. M., Fakioglu, E., Segarra, T., Rohan, L., et al. (2013). Griffithsin protects mice from genital herpes by preventing cell-to-cell spread. J. Virol. 87, 6257–6269. doi: 10.1128/JVI.00012-13
Nowakowski, T. J., Pollen, A. A., Di Lullo, E., Sandoval-Espinos, A. C., Bershteyn, M., and Kriegstein, A. R. (2016). Expression analysis highlights AXL as a candidate Zika virus entry receptor in neural stem cells. Cell Stem Cell 18, 591–596. doi: 10.1016/j.stem.2016.03.012
O’Keefe, B. R., Giomarelli, B., Barnard, D. L., Shenoy, S. R., Chan, P. K., McMahon, J. B., et al. (2010). Broad-spectrum in vitro activity and in vivo efficacy of the antiviral protein griffithsin against emerging viruses of the family Coronaviridae. J. Virol. 84, 2511–2521. doi: 10.1128/JVI.02322-09
Onawole, A. T., Sulaiman, K. O., Adegoke, R. O., and Kolapo, T. U. (2017). Identification of potential inhibitors against the Zika virus using consensus scoring. J. Mol. Graph. Model. 73, 54–61. doi: 10.1016/j.jmgm.2017.01.018
Otto, M., Watanabe, K., Patterson, S., Pankiewicz, K., and Stuyver, L. (2004). Antiviral agents for treatment of flaviviridae infections. US20040266723A1. Washington, DC: U.S. Patent and Trademark Office.
Pantoja, P., Pérez-Guzmán, E. X., Rodríguez, I. V., White, L. J., González, O., Serrano, C., et al. (2017). Zika virus pathogenesis in rhesus macaques is unaffected by pre-existing immunity to dengue virus. Nat. Commun. 8:15674. doi: 10.1038/ncomms15674
Parida, M. M., Upadhyay, C., Pandya, G., and Jana, A. M. (2002). Inhibitory potential of neem (Azadirachta indica Juss) leaves on dengue virus type-2 replication. J. Ethnopharmacol. 79, 273–278. doi: 10.1016/S0378-8741(01)00395-6
Pascoalino, B. S., Courtemanche, G., Cordeiro, M. T., Gil, L. H., and Freitas-Junior, L. (2016). Zika antiviral chemotherapy: identification of drugs and promising starting points for drug discovery from an FDA-approved library. F1000Res. 5, 2523. doi: 10.12688/f1000research.9648.1
Raghavan, P. R. (2016). In vitro inhibition of Zika virus by Metadichol, a novel Nano emulsion lipid. J. Immunol. Tech. Infect. Dis. 5, 4.
Rainey, M. M., Korostyshevsky, D., Lee, S., and Perlstein, E. O. (2010). The antidepressant sertraline targets intracellular vesiculogenic membranes in yeast. Genetics 185, 1221–1233. doi: 10.1534/genetics.110.117846
Ramharack, P., and Soliman, M. E. S. (2017). Zika virus NS5 protein potential inhibitors: an enhanced in silico approach in drug discovery. J. Biomol. Struct. Dyn. 17, 1–16. doi: 10.1080/07391102.2017.1313175
Rausch, K., Hackett, B., Weinbrenm, N., Reeder, S., Sadovsky, Y., Hunter, C., et al. (2017). Screening bioactives reveals nanchangmycin as a broad spectrum antiviral active against Zika virus. Cell Rep. 18, 804–815. doi: 10.1016/j.celrep.2016.12.068
Retallack, H., Di Lullo, E., Arias, C., Knopp, K. A., Laurie, M. T., Sandoval-Espinosa, C., et al. (2016). Zika virus cell tropism in the developing human brain and inhibition by azithromycin. Proc. Natl. Acad. Sci. U.S.A. 113, 14408–14413. doi: 10.1073/pnas.1618029113
Reznik, S. E., and Ashby, J. C. (2017). Sofosbuvir: an anti-viral drug with potential efficacy against Zika infection. Int. J. Infect. Dis. 55, 29–30. doi: 10.1016/j.ijid.2016.12.011
Richard, A. S., Zhang, A., Park, S. J., Farzan, M., Zong, M., and Choe, H. (2015). Virion-associated phosphatidylethanolamine promotes TIM1-mediated infection by Ebola, dengue, and West Nile viruses. Proc. Natl Acad. Sci. U.S.A. 112, 14682–14687. doi: 10.1073/pnas.1508095112
Robert-Gero, M., Lawrence, F., and Lederer, E. (1989). “Potential clinical use of Sinefungin: reduction of toxicity and enhancement of activity,” in Leishmaniasis. NATO ASI Series (Series A: Life Sciences), Vol. 171, ed. D. T. Hart (Boston, MA: Springer). doi: 10.1007/978-1-4613-1575-9_110
Roy, A., Lim, L., and Song, J. (2016). Identification of quercetin from fruits to immediately fight Zika. bioRxiv. doi: 10.1101/074559
Roy, S., Chaurvedi, P., and Chowdhary, A. (2015). Evaluation of antiviral activity of essential oil of Trachyspermum ammi against Japanese encephalitis virus. Pharmacognosy Res. 7, 263–267. doi: 10.4103/0974-8490.157977
Rut, W., Zhang, L., Kasperkiewicz, P., Poreba, M., Hilgenfeld, R., and Drąg, M. (2016). Extended substrate specificity and first potent irreversible inhibitor/activity-based probe design for Zika virus NS2B-NS3 protease. Antiviral Res. 139, 88–94. doi: 10.1016/j.antiviral.2016.12.018
Sacramento, C. Q., de Melo, G. R., de Freitas, C. S., Rocha, N., Hoelz, L. V., Miranda, M., et al. (2017). The clinically approved antiviral drug sofosbuvir inhibits Zika virus replication. Sci. Rep. 7:40920. doi: 10.1038/srep40920
Sahoo, M., Jena, L., Daf, S., and Kumar, S. (2016). Virtual screening for potential inhibitors of ns3 protein of Zika virus. Genomics Inform. 14, 104–111. doi: 10.5808/GI.2016.14.3.104
Saiz, J. C., and Martín-Acebes, M. A. (2017). The race to find antivirals for Zika virus. Antimicrob. Agents Chemother. 61:e00411–17. doi: 10.1128/AAC.00411-17
Sapparapu, G., Fernandez, E., Kose, N., Cao, B., Fox, J. M., Bombardi, R. G., et al. (2016). Neutralizing human antibodies prevent Zika virus replication and fetal disease in mice. Nature 540, 443–447. doi: 10.1038/nature20564
Sarno, M., Sacramento, G. A., Khouri, R., do Rosario, M. S., Costa, F., Archanjo, G., et al. (2016). Zika virus infection and stillbirths: a case of hydrops fetalis, hydranencephaly and fetal demise. PLoS Negl. Trop. Dis. 10:e0004517. doi: 10.1371/journal.pntd.0004517
Savidis, G., Perreira, J. M., Portmann, J. M., Meraner, P., Guo, Z., Green, S., et al. (2016). The IFITMs inhibit Zika virus replication. Cell Rep. 15, 2323–2330. doi: 10.1016/j.celrep.2016.05.074
Saxena, S. K., Elahi, A., Gadugu, S., and Prasad, A. K. (2016). Zika virus outbreak: an overview of the experimental therapeutics and treatment. Virus Dis. 27, 111–115. doi: 10.1007/s13337-016-0307-y
Scaturro, P., Trist, I. M., Paul, D., Kumar, A., Acosta, E. G., Byrd, C. M., et al. (2014). Characterization of the mode of action of a potent dengue virus capsid inhibitor. J. Virol. 88, 11540–11555. doi: 10.1128/JVI.01745-14
Schneider, W. M., Chevillotte, M. D., and Rice, C. M. (2014). Interferon-stimulated genes: a complex web of host defenses. Annu. Rev. Immunol. 32, 513–545. doi: 10.1146/annurev-immunol-032713-120231
Silverman, J. A., Perlmutter, N. G., and Shapiro, H. M. (2003). Correlation of daptomycin bactericidal activity and membrane depolarization in Staphylococcus aureus. Antimicrob. Agents Chemother. 47, 2538–2544. doi: 10.1128/AAC.47.8.2538-2544.2003
Singh, R. K., Dhama, K., Malik, Y. S., Ramakrishnan, M. A., Karthik, K., Tiwari, R., et al. (2016). Zika virus – emergence, evolution, pathology, diagnosis, and control: current global scenario and future perspectives – a comprehensive review. Vet. Q. 36, 150–175. doi: 10.1080/01652176.2016.1188333
Sirohi, D., and Kuhn, R. J. (2017). Can an FDA-Approved Alzheimer’s drug be repurposed for alleviating neuronal symptoms of Zika virus? MBio 8:e00916–17. doi: 10.1128/mBio.00916-17
Smit, J. M., Moesker, B., Rodenhuis-Zybert, I., and Wilschut, J. (2011). Flavivirus cell entry and membrane fusion. Viruses 3, 160–171. doi: 10.3390/v3020160
Sommadossi, J. P., Gosselin, G., Pierra, C., Perigaud, C., and Peyrottes, S. (2011). Compounds and pharmaceutical compositions for the treatment of viral infections. US7951789B2. Washington, DC: U.S. Patent and Trademark Office.
Sriwilaijaroen, N., Fukumoto, S., Kumagai, K., Hiramatsu, H., Odagiri, T., Tashiro, M., et al. (2012). Antiviral effects of Psidium guajava Linn. (guava) tea on the growth of clinical isolated H1N1 viruses: its role in viral hemagglutination and neuraminidase inhibition. Antiviral Res. 94, 139–146. doi: 10.1016/j.antiviral.2012.02.013
Stephen, P., Baz, M., Boivin, G., and Lin, S. X. (2016). Structural insight into NS5 of Zika virus leading to the discovery of MTase inhibitors. J. Am. Chem. Soc. 138, 16212–16215. doi: 10.1021/jacs.6b10399
Stettler, K., Beltramello, M., Espinosa, D. A., Graham, V., Cassotta, A., Bianchi, S., et al. (2016). Specificity, cross-reactivity, and function of antibodies elicited by Zika virus infection. Science 353, 823–826. doi: 10.1126/science.aaf8505
Swanstrom, J. A., Plante, J. A., Plante, K. S., Young, E. F., McGowan, E., Gallichotte, E. N., et al. (2016). Dengue virus envelope dimer epitope monoclonal antibodies isolated from dengue patients are protective against Zika virus. MBio 7:e001123–16. doi: 10.1128/mBio.01123-16
Takebe, Y., Saucedo, C. J., Lund, G., Uenishi, R., Hase, S., Tsuchiura, T., et al. (2013). Antiviral lectins from red and blue-green algae show potent in vitro and in vivo activity against hepatitis C virus. PLoS ONE 8:e64449. doi: 10.1371/journal.pone.0064449
Tan, C. W., Sam, I. C., Chong, W. L., Lee, V. S., and Chan, Y. F. (2017). Polysulfonate suramin inhibits Zika virus infection. Antiviral Res. 143, 186–194. doi: 10.1016/j.antiviral.2017.04.017
Tang, L. I., Ling, A. P., Koh, R. Y., Chye, S. M., and Voon, K. G. (2012). Screening of anti-dengue activity in methanolic extracts of medicinal plants. BMC Complement. Altern. Med. 12:3. doi: 10.1186/1472-6882-12-3
Tian, H., Ji, X., Yang, X., Xie, W., Yang, K., Chen, C., et al. (2016). The crystal structure of Zika virus helicase: basis for antiviral drug design. Protein Cell 7, 450–454. doi: 10.1007/s13238-016-0275-4
Varghese, F. S., Rausalu, K., Hakanen, M., Saul, S., Kümmerer, B. M., Susi, P., et al. (2016). Obatoclax inhibits alphavirus membrane fusion by neutralizing the acidic environment of endocytic compartments. Antimicrob. Agents Chemother. 61:e02227–16. doi: 10.1128/AAC.02227-16
Wang, Q., Yang, H., Liu, X., Dai, L., Ma, T., Qi, J., et al. (2016). Molecular determinants of human neutralizing antibodies isolated from a patient infected with Zika virus. Sci. Transl. Med. 8:369ra179. doi: 10.1126/scitranslmed.aai8336
Wang, S., Hong, S., Deng, Y. Q., Ye, Q., Zhao, L.-Z., Zhang, F.-C., et al. (2016). Transfer of convalescent serum to pregnant mice prevents Zika virus infection and microcephaly in offspring. Cell Res. 2016, 158–160. doi: 10.1038/cr.2016.144
WHO (2016). Available at. http://www.who.int/emergencies/zika-virus/timeline/en/
Williams, K. L., Sukupolvi-Petty, S., Beltramello, M., Johnson, S., Sallusto, F., Lanzavecchia, A., et al. (2013). Therapeutic efficacy of antibodies lacking Fc receptor binding against lethal dengue virus infection is due to neutralizing potency and blocking of enhancing antibodies. PLoS Pathog. 9:e1003157. doi: 10.1371/journal.ppat.1003157
Xu, M., Lee, E. M., Wen, Z., Cheng, Y., Huang, W. K., Qian, X., et al. (2016). Identification of small-molecule inhibitors of Zika virus infection and induced neural cell death via a drug repurposing screen. Nat Med. 22:1101–1107. doi: 10.1038/nm.4184
Xu, X., Vaughan, K., Weiskopf, D., Grifon, A., Diamond, M. S., Sette, A., et al. (2016). Identifying candidate targets of immune responses in Zika virus based on homology to epitopes in other flavivirus species. PLoS Curr. 15, 8. doi: 10.1371/currents.outbreaks.9aa2e1fb61b0f632f58a098773008c4b
Zasloff, M., Adams, A. P., Beckerman, B., Campbell, A., Han, Z., Luijten, E., et al. (2011). Squalamine as a broad-spectrum systemic antiviral agent with therapeutic potential. Proc. Natl. Acad. Sci. U.S.A. 108, 15978–15983. doi: 10.1073/pnas.1108558108
Zhang, C., Feng, T., Cheng, J., Li, Y., Yin, X., Zeng, W., et al. (2016). Structure of the NS5 methyltransferase from Zika virus and implications in inhibitor design. Biochem. Biophys. Res. Commun. doi: 10.1016/j.bbrc.2016.11.098 [Epub ahead of print].
Zhang, S., Kostyuchenko, V. A., Ng, T. S., Lim, X. N., Ooi, J. S., Lambert, S., et al. (2016). Neutralization mechanism of a highly potent antibody against Zika virus. Nat. Commun. 7:13679. doi: 10.1038/ncomms13679
Zmurko, J., Marques, R. E., Schols, D., Verbeken, E., Kaptein, S. J., and Neyts, J. (2016). The viral polymerase inhibitor 7-Deaza-2′-C-Methyladenosine is a potent inhibitor of in vitro Zika virus replication and delays disease progression in a robust mouse infection model. PLoS Negl. Trop. Dis. 10:e0004695. doi: 10.1371/journal.pntd.0004695
Keywords: Zika virus, drugs, therapies, microcephaly, Guillain-Barré Syndrome
Citation: Munjal A, Khandia R, Dhama K, Sachan S, Karthik K, Tiwari R, Malik YS, Kumar D, Singh RK, Iqbal HMN and Joshi SK (2017) Advances in Developing Therapies to Combat Zika Virus: Current Knowledge and Future Perspectives. Front. Microbiol. 8:1469. doi: 10.3389/fmicb.2017.01469
Received: 13 June 2017; Accepted: 20 July 2017;
Published: 03 August 2017.
Edited by:
Rubén Bueno-Marí, Universitat de València, SpainReviewed by:
A. Arturo Leis, Methodist Rehabilitation Center, United StatesJuan-Carlos Saiz, Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria, Spain
Copyright © 2017 Munjal, Khandia, Dhama, Sachan, Karthik, Tiwari, Malik, Kumar, Singh, Iqbal and Joshi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Kuldeep Dhama, a2RoYW1hQHJlZGlmZm1haWwuY29t
 Ashok Munjal
Ashok Munjal Rekha Khandia
Rekha Khandia Kuldeep Dhama
Kuldeep Dhama Swati Sachan
Swati Sachan Kumaragurubaran Karthik
Kumaragurubaran Karthik Ruchi Tiwari
Ruchi Tiwari Yashpal S. Malik
Yashpal S. Malik Deepak Kumar
Deepak Kumar Raj K. Singh
Raj K. Singh Hafiz M. N. Iqbal
Hafiz M. N. Iqbal Sunil K. Joshi
Sunil K. Joshi