- 1Department of Neurology, Kangdong Sacred Heart Hospital, Hallym University College of Medicine, Seoul, South Korea
- 2Department of Neurology, Chuncheon Sacred Heart Hospital, Hallym University College of Medicine, Chuncheon, South Korea
- 3Department of Neurology, Dongtan Sacred Heart Hospital, Hallym University College of Medicine, Hwaseong, South Korea
- 4Department of Neurology, Kangnam Sacred Heart Hospital, Hallym University College of Medicine, Seoul, South Korea
- 5Hallym Neurological Institute, Department of Neurology, Hallym University Sacred Heart Hospital, Hallym University College of Medicine, Anyang-si, South Korea
Background: Although controversial, homocysteine (Hcy) and lipid parameters have been associated with particular stroke subtypes. However, there are limited studies concerning the relationship between Hcy and lipid levels in acute ischemic stroke (AIS). We evaluated the impact of Hcy levels on lipid profiles in terms of specific stroke subtypes.
Methods: A total of 2,324 patients with first-ever AIS were recruited from two hospitals in South Korea. The exclusion criteria were as follows: (a) pre-stroke modified Rankin scale (mRS) ≥ 1, (b) undetermined or other stroke etiology, and (c) absence of Hcy data. Among the 1,580 eligible patients, the Hcy level was divided into tertile groups. Logistic regression was used to assess association of Hcy levels with lipid levels by stroke subtypes.
Results: Significant downward trends in total cholesterol, low-density lipoprotein (LDL), and high-density lipoprotein (HDL) were only observed in patients with small vessel occlusion (SVO) as Hcy increased. In logistic regression analysis, while in patients with SVO subtype, the highest level of Hcy tertiles (OR = 1.648, 95% CI = 1.047–2.594) was associated with the lower HDL level (≤40 mg/dL), the significance disappeared in patients with LAA and CE subtypes.
Conclusion: Although our study does not demonstrate causal relationship, we suggest that Hcy might play a mediating role between HDL and SVO stroke development. To clarify the role of Hcy on AIS, this study will provide academic support for designing future research.
Introduction
Homocysteine (Hcy) is a sulfur-containing, toxic intermediate of the methionine metabolic pathway (1). Total Hcy levels are known to be associated with vascular events, mortality and morbidity after stroke (2). However, the mechanism by which Hcy affects stroke outcome is still unknown.
Although still controversial, among several stroke subtypes, Hcy levels have been shown to be associated with various small vessel disease (SVD) components, including white matter hyperintensities (WMH), lacunes, and cerebral microbleeds (3, 4). Previous studies reported a possible linkage between hyperhomocysteinemia and dyslipidemia (5–7), but studies have suggested mixed conclusions. In this regard, a recent Mendelian randomization analysis published an interesting result that genetically predicted Hcy was related to SVD but was not related to large artery atherosclerosis (LAA) or cardioembolic (CE) stroke (8). Furthermore, 1-standard deviation (SD) increase in high-density lipoprotein (HDL) was associated with a significantly decreased risk of SVD (9). Considering these results, Hcy may play an important role in a particular stroke subtype. However, there are limited academic data concerning the relationship between Hcy and lipid profiles, especially in particular stroke subtypes. The current guidelines do not recommend aggressive treatment for Hcy level. However, if the role of Hcy in a particular stroke subtype changes, to control Hcy may be the treatment option for stroke prevention.
Therefore, the aim of this study was to evaluate the impact of serum Hcy levels on lipid profiles in patients with acute ischemic stroke (AIS) by different stroke subtypes.
Methods
Study Population
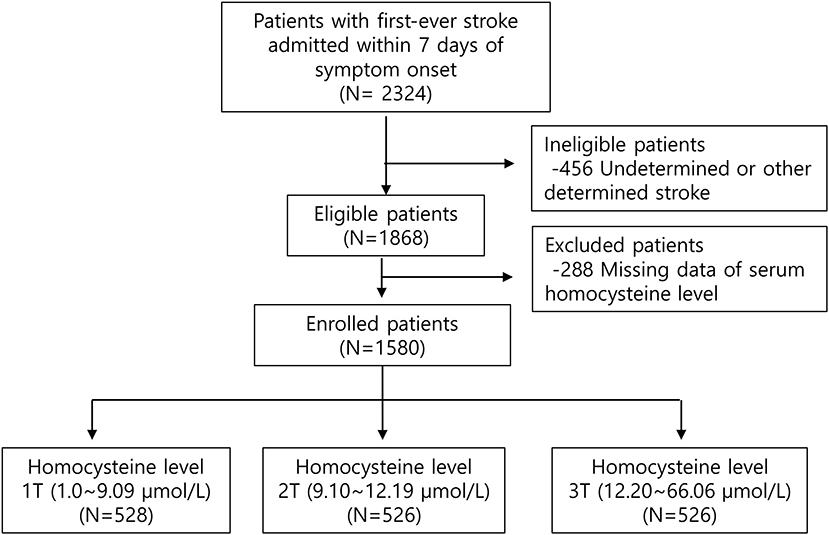
Patients with first-ever ischemic stroke (IS) within 7 days after symptom onset who were admitted to the two tertiary university hospitals from July 2007 to January 2015 were screened. Exclusion criteria was as follows: (1) patients with previous morbidities; (2) patients with undetermined or other determined stroke etiologies; (3) subjects without acute phase Hcy levels (Figure 1).
This study was approved by the Institutional Review Board (IRB No. KANGDONG 2020-02-005).
Stroke Subtyping
Investigators classified all stroke subtypes based on a review of original brain imaging. Trans thoracic echocardiogram was performed to evaluate the cardioembolic source in all patients. Extensive work-ups including head/neck angiography, 24-h Holter monitoring and transesophageal echocardiogram was performed. A modification of the Trial of Org 10172 (TOAST) classification was used (10). Large artery atherosclerosis was defined as symptomatic stroke with index arterial stenosis >50% or occlusion, presumably due to atherosclerosis. Cortical or cerebellar lesions and brainstem or subcortical lesions >1.5 cm in diameter on imaging are considered to be of LAA origin. Diagnostic work-ups should exclude potential sources of cardioembolism (10). According to the previous reports, CE stroke was defined as the presence of a potential moderate or high risk source of cardioembolism based on the evidence of their relative propensities for an embolus (10). To define CE etiology, we performed extensive work-ups: head/neck angiography, 24-h Holter monitoring. Transthoracic echocardiography was performed in almost all patients. Transesophageal echocardiography was administered when standard cardiac evaluation (Transthoracic echocardiography or Holter monitoring) did not reveal any CE source in high clinical suspicion of cardioembolism. Small vessel occlusion (SVO) was defined as a clinical lacunar infarction with a diameter <15 mm (11). Potential CE sources should not be present, and evaluation of the index large arteries should not demonstrate a stenosis of >50%. Other determined etiology includes subjects with rare causes of strokes, such as hematologic disorders, non-atherosclerotic stenosis, or hypercoagulable status. Undetermined etiology was defined as the cause of a stroke cannot be determined despite an extensive evaluations (negative). In others, this category includes patients with 2 or more etiologies (2 or more) or patients with incomplete evaluations (incomplete).
Biomarkers
Serum Hcy was measured at basal level within 7 days after stroke onset using a competitive immunoassay (Immulite 2000, DPC Biermann GmbH, Bad Nauheim, Germany). Since elevated blood Hcy concentration is known to increases the risk of stroke, it is included in etiology work-ups in many tertiary hospitals. In this regard, three hospitals in this study performed this Hcy-test on all IS patients.
Levels of Hcy were divided into tertiles (1T, 1.0–9.09 μmol/L; 2T, 9.10–12.19 μmol/L; and 3T, 12.20–66.06 μmol/L). Levels of lipid profiles were measured by using enzymatic and chromatometric methods. The serum obtained was used to estimate total cholesterol, triglyceride, high-density lipoprotein (HDL), and low-density lipoprotein (LDL) by using serum separate tube.
According to the general dyslipidemia diagnosis criteria, LDL and HDL were classified into 2 groups (LDL; ≤130 vs. >130 mg/dL and HDL; >40 vs. ≤40 mg/dL).
Stroke Scale
Initial stroke severity was assessed by the National Institutes of Health Stroke Scale (NIHSS) at admission. Functional outcome was measured at 3 months after stroke onset by using the modified Rankin Scale (mRS).
Statistics
All statistical analyses were performed using SPSS version 21.0 (SPSS Inc., Chicago, Illinois, USA). The distribution of demographic, clinical, and laboratory data was analyzed using a χ2-test, Kruskal-Wallis-test, as appropriate. Since data was not normally distributed, Kruskal-Wallis-test was performed to compare whether there is a significance among groups. Values for the continuous variables are expressed as the means ± standard deviations (SDs). *P for trend was calculated using linear regression. Associations between variables were assessed by using multivariable binary logistic regression analysis. Odds ratios (ORs) and 95% confidence interval (95% CI) were expressed for the results and probability values. A probability value of ≤0.05 was considered statistically significant.
Results
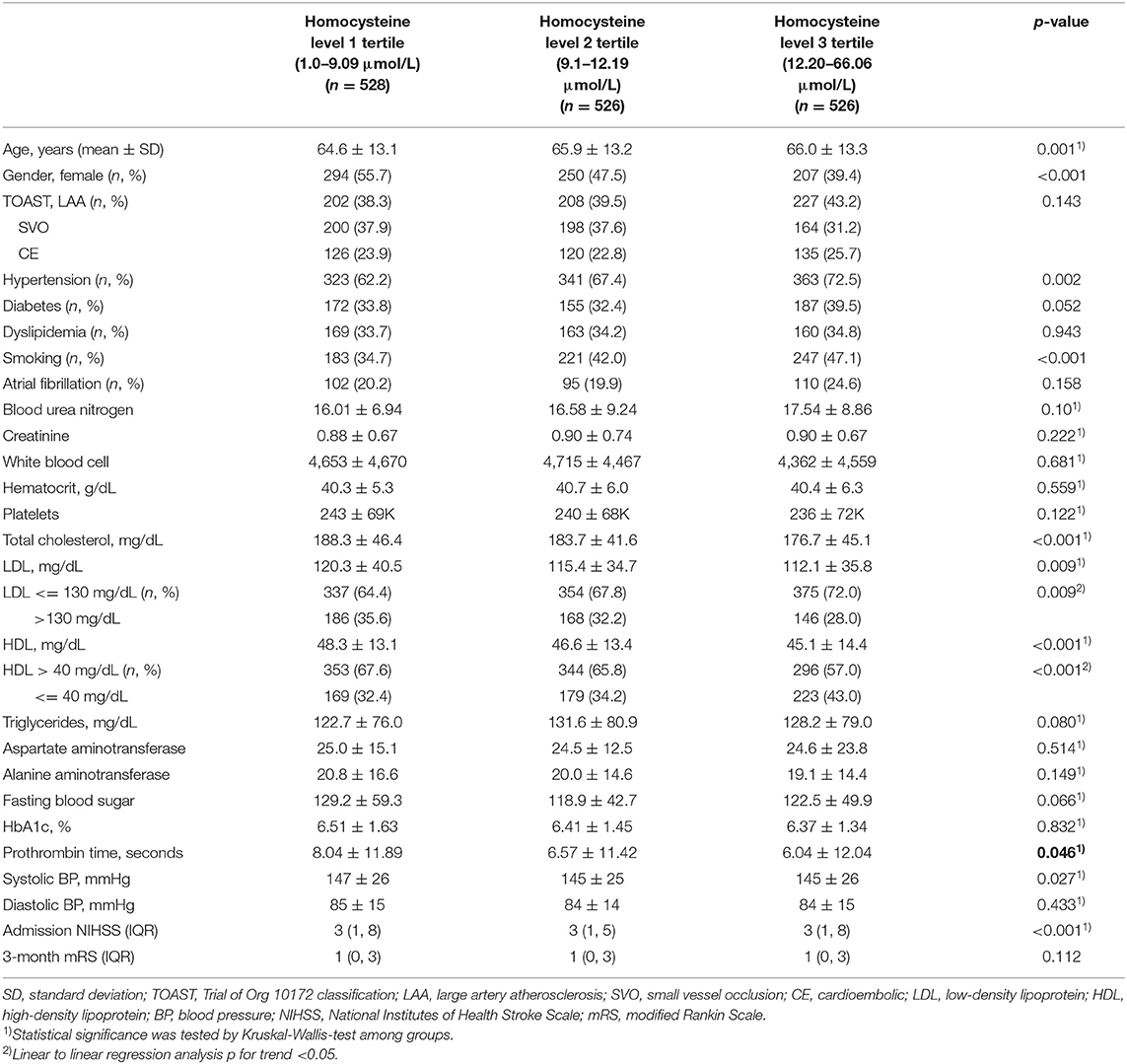
Among a total of 2,324 patients without previous morbidities (previous mRS = 0), patients with undetermined or other determined stroke etiologies (n = 456) were excluded. Of the 1,868 eligible subjects, patients were excluded because their acute phase Hcy concentrations (n = 288) were not measured. Finally, a total of 1,580 patients were included in this analysis. Patients were classified as tertiles according to their Hcy level (Figure 1). Hcy levels were 1.0–9.09 μmol/L (T1), 9.1–12.19 μmol/L (T2), and 12.20–66.06 μmol/L (T3). Patients with a higher homocysteine level were older, more likely to have hypertension, a smoking habit, and higher levels of blood urea nitrogen (Table 1). Total cholesterol, LDL, and HDL levels showed a downward trend across the Hcy tertile groups. There was no difference in stroke severity or short-term functional outcome according to Hcy level.
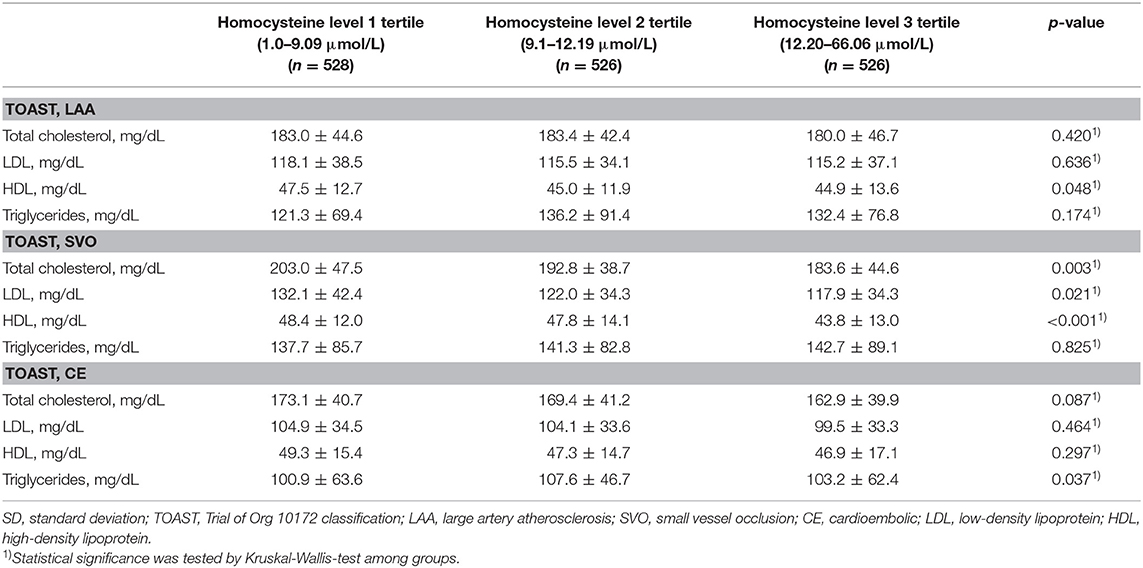
According to the stroke subtypes, a downward trend of total cholesterol (p = 0.003), LDL (p = 0.021), and HDL (p < 0.001) was significant only in patients with SVO (Table 2 and Figure 2). A downward trend of HDL (p = 0.048) was significant in patients with LAA.
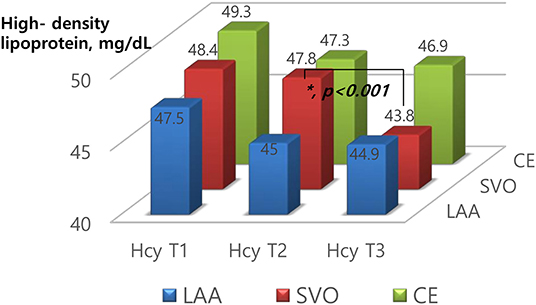
Figure 2. Levels of high-density lipoprotein according to homocysteine tertiles based on stroke subtypes.
To clarify the relationship between Hcy and lipid profiles by stroke subtypes, we performed multivariable binary logistic regression analysis. After adjusting for age, gender, smoking, Hcy levels, prothrombin time, systolic BP, and admission NIHSS, in patients with LAA subtype, 1-elevated systolic BP (OR = 0.993, 95% CI = 0.986–1.000) and smoking (OR = 1.536, 95% CI = 1.086–2.173) was associated with HDL ≤ 40 mg/dL. In patients with SVO subtype, the highest level of Hcy tertiles (Hcy T3) (OR = 1.648, 95% CI = 1.047–2.594) and smoking (OR = 1.548, 95% CI = 1.050–2.282) was associated with the lower HDL level (HDL ≤ 40 mg/dL) (Table 3). However, the significance disappeared in patients with LAA and CE subtypes.
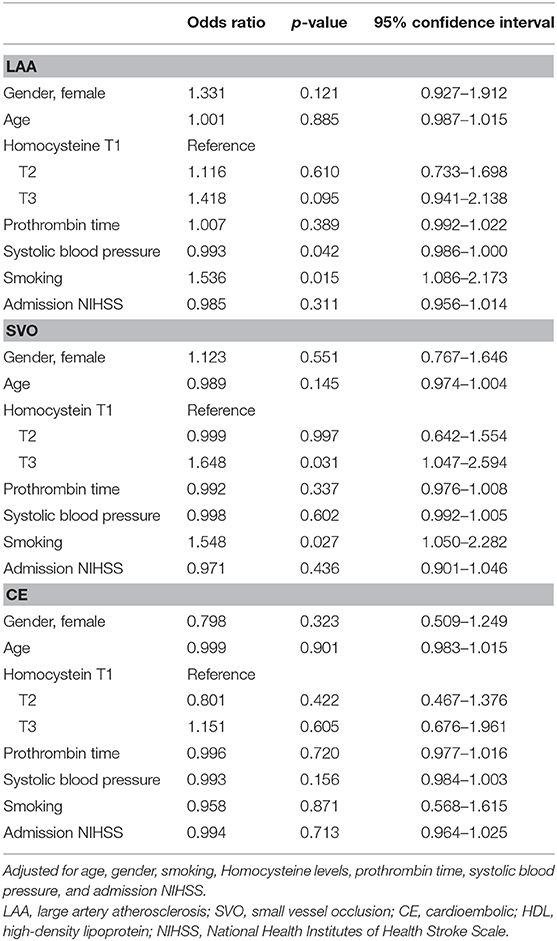
Table 3. Multivariate analysis of the associations between homocysteine levels and the lower level of high-density lipoprotein (HDL ≤ 40 mg/dL compared to HDL > 40 mg/dL).
Additionally, after adjusting for multiple variables, Hcy levels did not show significant associations with LDL in all stroke subtypes (Supplementary Table).
Discussion
Our study demonstrated that elevated Hcy levels during AIS are significantly related to decreased total cholesterol, decreased LDL and decreased HDL. According to stroke subtypes, interactions between HDL and Hcy levels were only observed in patients with SVO etiologies. To the best of our knowledge, this is the first report demonstrating relationship between Hcy and HDL considering stroke etiology.
Associations Between Hcy and Stroke
According to previous retrospective studies, Hcy levels were significantly higher in patients with coronary artery disease and stroke (12, 13). The exact pathological mechanisms by which Hcy might contribute to stroke have not yet been elucidated. However, prothrombotic effects, pro-oxidant effects, and genetic modifications, have been suggested as hypotheses. Hcy stimulates platelet activation and affects fibrinolytic cascades (14). Vascular smooth muscle proliferation, which is directly stimulated by Hcy, is a key component of atherosclerosis (14). Furthermore, Hcy oxidizes LDL to produce thiolated-LDL, inhibits intracellular antioxidant enzymes, and reduces the bioavailability of nitric oxide. In our study, patients with higher Hcy levels had lower LDL levels. Although we cannot explain the reason, this oxidization theory may partially support the mechanism. In addition, Hcy levels were not associated with short-term functional outcome (mRS at 3 month) in this study. We think that more potent predictors such as stroke severity (NIHSS) or conventional risk factors (HTN, DM, and AF) may affect this final result.
Associations Between Hcy and Stroke Subtypes
The relationship between hyperhomocysteinemia and stroke subtypes is inconsistent. Because IS is a heterogeneous disease, Hcy may play a specific role in different stroke subtypes. A recent study demonstrated that serum Hcy is correlated with SVO indicators, including WMH volume, cerebral microbleeds, and enlarged perivascular spaces (3). In 377 Chinese IS patients, patients with SVO etiologies with severe leukoaraiosis had the highest Hcy levels (15). In the UK black stroke population, the highest levels of Hcy were found in patients with lacunar stroke with leukoaraiosis (4). A recent meta-analysis including 18 studies with 5,088 participants found that patients with SVD subtypes have significantly elevated Hcy levels compared with the levels in controls (16). However, some studies have reported different results (4). In young and middle-aged IS patients, hyper-Hcy was more common in LAA, especially intracranial arterial stenosis, than in extracranial arterial stenosis (17). Among a total of 3,799 AIS patients, although patients with the highest Hcy levels had a 1.6-fold increased risk of mortality, this result was only significant in patients with LAA etiology (2). Despite that finding, a recent Mendelian randomization study supported that a genetically predicted 1 SD increase in Hcy was associated with SVO [odds ratio (OR): 1.34, 95% confidence interval (CI): 1.13–1.58] but was not associated with LAA or CE stroke (9). In our study, we found that elevated Hcy levels are significantly associated with lower HDL level only in patients with SVO etiology. Although we cannot explain the biologic linkage among them, we suggest that Hcy might play a key role in pathologic impact of HDL on SVO development.
Associations Between Hcy and Lipids
It then remains to be determined why Hcy is related to HDL in patients with AIS in our study. Previous studies have shown how Hcy is associated with lipid profiles in various populations, including a community-based population (5), a coronary artery disease population (6, 18), a thyroid disease population (7) and an intensive care condition population (19). However, the exact mechanism by which Hcy affects the lipid profile in stroke patients is not well-understood. Although dyslipidemia is a well-known risk factor for stroke, the relationship between lipid profiles and IS subtypes remains controversial. In a total of 4,660 community-based Chinese people, hyperhomocysteinemia was independently related to elevated triglyceride levels and decreased HDL levels (5). In 126 patients with myocardial infarction, there was a negative correlation between HDL and Hcy and a positive correlation between LDL and Hcy (6). In 94 patients who were admitted to the intensive care unit, a negative relationship between Hcy and HDL was found with Pearson's correlation coefficient (19). However, studies examining the associations between Hcy and lipid profiles have shown mixed conclusions (20, 21). Despite that, the most consistent consensus indicates that elevated Hcy levels are associated with decreased HDL (5). This is consistent with our findings, but to the best of our knowledge, no studies have analyzed Hcy and lipid profiles in the context of stroke subtypes.
Associations Between Lipids and Stroke Subtypes
Previous observational studies have reported discrepant conclusions concerning the relationship of lipid profiles with IS (22–24). In addition, most studies have not performed stroke subtyping. Recently, the Stroke Genetics Network found that a 1 SD genetic increase in HDL was associated with a decreased risk of SVO (OR 0.79, 95% CI 0.67–0.90) in a multivariable Mendelian randomization study (8). In this regard, although our study does not provide the causality, we suggest that Hcy might play a mediating role between HDL and SVO stroke development.
In addition to the large number of enrolled patients with MRI documentation, a strength of this study is that it is the first study to investigate which lipid parameters are associated with Hcy, considering stroke subtypes. Since stroke etiology is heterogeneous, it is very helpful to determine which biomarkers should be treated. Our findings may have novel implications that increased Hcy may be a useful biomarker linked to HDL, at least in SVO subtypes. Despite this novel strength, there are some limitations to this study. First, this is a retrospective observational study, so it does not demonstrate causal relationships between Hcy, lipids, and stroke subtypes. However, this study will provide academic support for designing future research. Second, the timing of Hcy and lipid measurements is important because plasma levels can change. Although the measurement time was not the same, the blood sample was collected the day after admission within 1 week of the AIS. Third, in patients with LAA etiology, patients with higher Hcy levels had lower LDL levels. According to prior report, Hcy can oxidize LDL and combine with LDL particles to form Hcy-thiolactone (14, 18). It may affect the ambiguous result, but a lower LDL level might be due to previous statin use. Unfortunately, we did not evaluate the medication history. Finally, the result cannot be generalized to other cohorts.
Conclusion
In conclusion, we observed that elevated Hcy levels were significantly associated with lower HDL levels only in patients with SVO subtype. Consequently, the findings in the present study might suggest that Hcy may play a mediating role between HDL and SVO stroke development. The current guidelines do not recommend aggressive treatment for Hcy level. However, if the role of Hcy in a particular stroke subtype changes, to control Hcy may be the treatment option for stroke prevention. Future studies are needed to define the role of Hcy on lipid metabolism in stroke development.
Data Availability Statement
Data cannot be shared because there are embargoes on datasets. Anonymized data will be shared by request from any qualified investigator.
Ethics Statement
The studies involving human participants were reviewed and approved by IRB No. KANGDONG 2020-02-005. Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements.
Author Contributions
YK: study design, clinical and image data acquisition, analysis and interpretation, and primary responsibility for writing the manuscript. B-CL and J-HL: study design, data interpretation, critical revision of the manuscript for important intellectual content, and supervision of the study. S-HL, YJK, CK, MUJ, SJ, J-SL, MSO, and K-HY: data acquisition and critical. All authors contributed to the article and approved the submitted version.
Funding
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (NRF- 2018R1C1B5086320) and supported by a Grant no. 2019-08 from the Kangdong Sacred Heart Hospital Fund (YK) and funded by Research of Korea Centers for Disease Control and Prevention (2020-ER6303-00) (K-HY).
Conflict of Interest
B-CL reports the following relationships: Boehringer-Ingelheim, Bayer, Daiichi-Sankyo, Esai, and AstraZeneca.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fneur.2020.565506/full#supplementary-material
References
1. Lehotsky J, Tothova B, Kovalska M, Dobrota D, Benova A, Kalenska D, et al. Role of homocysteine in the ischemic stroke and development of ischemic tolerance. Front Neurosci. (2016) 10:538. doi: 10.3389/fnins.2016.00538
2. Shi Z, Guan Y, Huo YR, Liu S, Zhang M, Lu H, et al. Elevated total homocysteine levels in acute ischemic stroke are associated with long-term mortality. Stroke. (2015) 46:2419–25. doi: 10.1161/STROKEAHA.115.009136
3. Nam KW, Kwon HM, Jeong HY, Park JH, Kwon H, Jeong SM. Serum homocysteine level is related to cerebral small vessel disease in a healthy population. Neurology. (2019) 92:e317–25. doi: 10.1212/WNL.0000000000006816
4. Khan U, Crossley C, Kalra L, Rudd A, Wolfe CD, Collinson P, et al. Homocysteine and its relationship to stroke subtypes in a UK black population: the south London ethnicity and stroke study. Stroke. (2008) 39:2943–9. doi: 10.1161/STROKEAHA.107.513416
5. Momin M, Jia J, Fan F, Li J, Dou J, Chen D, et al. Relationship between plasma homocysteine level and lipid profiles in a community-based Chinese population. Lipids Health Dis. (2017) 16:54. doi: 10.1186/s12944-017-0441-6
6. Qujeq D, Omran TS, Hosini L. Correlation between total homocysteine, low-density lipoprotein cholesterol and high-density lipoprotein cholesterol in the serum of patients with myocardial infarction. Clin Biochem. (2001) 34:97–101. doi: 10.1016/S0009-9120(01)00187-4
7. Saleh AAS. Lipid profile and levels of homocysteine and total antioxidant capacity in plasma of rats with experimental thyroid disorders. J Basic Appl Zool. (2015) 72:173–8. doi: 10.1016/j.jobaz.2015.01.001
8. Hindy G, Engstrom G, Larsson SC, Traylor M, Markus HS, Melander O, et al. Role of blood lipids in the development of ischemic stroke and its subtypes: a mendelian randomization study. Stroke. (2018) 49:820–7. doi: 10.1161/STROKEAHA.117.019653
9. Larsson SC, Traylor M, Markus HS. Homocysteine and small vessel stroke: a mendelian randomization analysis. Ann Neurol. (2019) 85:495–501. doi: 10.1002/ana.25440
10. Adams HP Jr, Bendixen BH, Kappelle LJ, Biller J, Love BB, Gordon DL, et al. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in acute stroke treatment. Stroke. (1993) 24:35–41. doi: 10.1161/01.STR.24.1.35
11. Chung JW, Park SH, Kim N, Kim WJ, Park JH, Ko Y, et al. Trial of ORG 10172 in acute stroke treatment (TOAST) classification and vascular territory of ischemic stroke lesions diagnosed by diffusion-weighted imaging. J Am Heart Assoc. (2014) 3:e001119. doi: 10.1161/JAHA.114.001119
12. Nurk E, Tell GS, Vollset SE, Nygard O, Refsum H, Ueland PM. Plasma total homocysteine and hospitalizations for cardiovascular disease: the Hordaland homocysteine study. Arch Intern Med. (2002) 162:1374–81. doi: 10.1001/archinte.162.12.1374
13. Stampfer MJ, Malinow MR, Willett WC, Newcomer LM, Upson B, Ullmann D, et al. A prospective study of plasma homocyst(e)ine and risk of myocardial infarction in US physicians. JAMA. (1992) 268:877–81. doi: 10.1001/jama.268.7.877
14. Tayal D, Goswami B, Ch B, Mallika V. Role of homocysteine and lipoprotein (A) in atherosclerosis: an update. Biomed Res. (2011) 22:391–405.
15. Ma Y, Zhao X, Zhang W, Liu L, Wang Y, Fang R, et al. Homocysteine and ischemic stroke subtype: a relationship study in Chinese patients. Neurol Res. (2010) 32:636–41. doi: 10.1179/016164109X12445616596445
16. Piao X, Wu G, Yang P, Shen J, De A, Wu J, et al. Association between homocysteine and cerebral small vessel disease: a meta-analysis. J Stroke Cerebrovasc Dis. (2018) 27:2423–30. doi: 10.1016/j.jstrokecerebrovasdis.2018.04.035
17. Gungor L, Polat M, Ozberk MB, Avci B, Abur U. Which ischemic stroke subtype is associated with hyperhomocysteinemia? J Stroke Cerebrovasc Dis. (2018) 27:1921–9. doi: 10.1016/j.jstrokecerebrovasdis.2018.02.033
18. Yadav A, Bhagwat V, Rathod I. Relationship of plasma homocysteine with lipid profile parameters in ischemic heart disease. Indian J Clin Biochem. (2006) 21:106–10. doi: 10.1007/BF02913076
19. Samara I, Karikas GA, Kalkani E, Tzimogianni A, Bournousouzis N, Fytou-Pallikari A. Negative correlations of serum total-homocysteine and HDL-c levels in ICU patients. Health Sci J. (2010) 4:189–92.
20. Laloux P, Galanti L, Jamart J. Lipids in ischemic stroke subtypes. Acta Neurologica Belgica. (2004) 104:13–9.
21. Yuan BB, Luo GG, Gao JX, Qiao J, Yang JB, Huo K, et al. Variance of serum lipid levels in stroke subtypes. Clin Lab. (2015) 61:1509–14. doi: 10.7754/Clin.Lab.2015.150118
22. Yaghi S, Elkind MS. Lipids and cerebrovascular disease: research and practice. Stroke. (2015) 46:3322–8. doi: 10.1161/STROKEAHA.115.011164
23. Kurth T, Everett BM, Buring JE, Kase CS, Ridker PM, Gaziano JM. Lipid levels and the risk of ischemic stroke in women. Neurology. (2007) 68:556–62. doi: 10.1212/01.wnl.0000254472.41810.0d
Keywords: HDL, cholesterol, lipid metabolism, homocysteine, brain ischemia, stroke
Citation: Kim Y, Lee J-H, Lee S-H, Kim YJ, Kim C, Jang MU, Jung S, Lim J-S, Oh MS, Yu K-H and Lee B-C (2020) Serum Homocysteine Is Associated With HDL Only in Stroke Patients With Small Vessel Occlusion. Front. Neurol. 11:565506. doi: 10.3389/fneur.2020.565506
Received: 26 May 2020; Accepted: 19 October 2020;
Published: 02 December 2020.
Edited by:
Phyo Kyaw Myint, University of Aberdeen, United KingdomReviewed by:
Wen-Jun Tu, Chinese Academy of Medical Sciences and Peking Union Medical College, ChinaAlexander Tsiskaridze, Tbilisi State University, Georgia
Copyright © 2020 Kim, Lee, Lee, Kim, Kim, Jang, Jung, Lim, Oh, Yu and Lee. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Byung-Chul Lee, c3NicmFpbkBoYWxseW0uYWMua3I=
 Yerim Kim
Yerim Kim Ju-Hun Lee1
Ju-Hun Lee1