Abstract
Objective:
To identify significant predictors and construct a validated nomogram for predicting post-stroke cognitive impairment no dementia (PSCIND) risk among first-ever mild ischemic stroke (MIS) patients.
Methods:
This retrospective cohort study analyzed 242 first-ever MIS patients categorized into normal cognitive (n = 137) and PSCIND (n = 105) groups. Comprehensive data encompassing demographic characteristics, laboratory parameters, cerebral small vessel disease (CSVD) imaging markers, neuropsychological assessments, and ischemic stroke lesion characteristics were collected. Predictor selection was conducted through least absolute shrinkage and selection operator (LASSO) regression analysis, followed by multivariable logistic regression for nomogram construction. Model performance was assessed through discrimination (area under the curve), calibration (calibration plots, Hosmer-Lemeshow test), and clinical utility (decision curve analysis).
Results:
Eight independent predictors were identified: age (OR = 1.060, 95% CI: 1.016–1.106), education level (OR = 0.917, 95% CI: 0.845–0.995), type 2 diabetes mellitus (OR = 9.407, 95% CI: 3.761–23.528), superoxide dismutase (OR = 0.951, 95% CI: 0.931–0.972), uric acid (OR = 1.006, 95% CI: 1.002–1.010), homocysteine (OR = 1.058, 95% CI: 1.027–1.091), strategic infarcts (OR = 4.566, 95% CI: 2.148–9.707), and severe CSVD burden (OR = 3.818, 95% CI: 1.842–7.911). The nomogram exhibited excellent discrimination (AUC = 0.886) accompanied by satisfactory calibration (Hosmer-Lemeshow χ2 = 14.542, p = 0.104). Decision curve analysis showed clinical utility across threshold probabilities of 6–100%.
Conclusion:
This validated nomogram incorporating clinical, biochemical, and neuroimaging biomarkers provides a robust tool for individualized PSCIND risk assessment in first-ever MIS patients, with potential to guide targeted interventions and cognitive monitoring.
Introduction
Stroke remains a foremost global health challenge (1), with post-stroke cognitive impairment (PSCI) emerging as a predominant debilitating disabling neuropsychiatric sequelae (2, 3). A multicenter study demonstrated that up to 44% of stroke survivors develop PSCI within the first 6 months post-event (4, 5), imposing a substantial burden on long-term functional outcomes. PSCI is defined as cognitive deficits persisting for 3–6 months post-stroke, categorized into two clinical subtypes: post-stroke cognitive impairment no dementia (PSCIND) and post-stroke dementia (PSD) (5). As an early stage of PSCI, PSCIND is characterized by measurable cognitive decline without fulfilling dementia diagnostic criteria, thereby serving as a critical window for early intervention.
Despite preserved basic activities of daily living, individuals with PSCIND exhibit significantly compromised quality of life metrics (5). A meta-analysis synthesizing 23 prospective cohorts revealed a pooled PSCIND prevalence of 38% during the first post-stroke year (6). Alarmingly, approximately 25% of PSCIND cases progress to PSD in the absence of timely interventions, underscoring the urgency of early identification and management (7). Nevertheless, validated predictive tools for PSCIND remain conspicuously absent in clinical practice.
Ischemic stroke, constituting 62% of global stroke incidence (8), presents unique diagnostic and therapeutic challenges in PSCI management. Mild ischemic stroke (MIS), operationally defined by National Institutes of Health Stroke Scale (NIHSS) scores ≤5 (9, 10), represents more than half of incident ischemic cerebrovascular events (11). Notably, the subtle neurological deficits characteristic of MIS frequently result in systematic underassessment of cognitive domains, despite neuropsychological evidence indicating high PSCI prevalence in MIS patients (5, 12). This clinical oversight stems from the misconception that mild neurological symptoms equate to negligible cognitive consequences. While current evidence highlights MIS populations as priority targets for PSCI prevention, critical knowledge gaps persist regarding PSCIND predictors in first-ever MIS patients, with a paucity of validated prediction models tailored to this subgroup.
To address these challenges, this retrospective study systematically evaluates demographic, biochemical, and neuroimaging predictors of PSCIND in first-ever MIS patients. We aim to develop and validate a clinically actionable, visualization-enhanced prediction tool. This model seeks to enable individualized PSCIND risk assessment, thereby informing targeted interventions to mitigate cognitive decline and improve long-term outcomes in this vulnerable population.
Methods
Patients
This prognostic modeling study employed a retrospective cohort design using data from the Neurology Department of Hebei General Hospital (August 2019–December 2022). Inclusion criteria comprised: (a) age ≥50 years; (b) first-ever MIS (NIHSS score ≤5) confirmed by neuroimaging; (c) availability of complete clinical variables and neuropsychological assessments. Exclusion criteria were rigorously applied to minimize confounding: (a) transient ischemic attack or pre-stroke cognitive impairment (documented by medical records or caregiver reports); (b) comorbidities potentially influencing cognitive assessments, such as active epilepsy/seizure disorders, traumatic brain injury history and major psychiatric disorders (e.g., schizophrenia, severe depression); (c) unavailable 3–6 month follow-up data in the institutional database; (d) recurrent cerebrovascular events during 3–6 month follow-up period; (e) diagnosis of PSD during the 3–6 month follow-up period; (f) anti-dementia medications were used during the 3–6 month follow-up period. The flow chart of participant selection is illustrated in Supplementary Figure S1. The diagnostic prediction model was developed and reported in full compliance with the Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD) statement, specifically following the Type 1b framework (development using existing data from a single-center retrospective cohort) (13). Conducted in compliance with the Declaration of Helsinki, this study obtained ethical clearance from the Ethics Committee of Hebei General Hospital (No.2025-LW-0124).
Evaluation of post-stroke cognitive impairment no dementia
Standardized neuropsychological evaluations were administered to all participants, incorporating three validated measures: the Beijing-adapted Montreal Cognitive Assessment,1 alongside assessments of basic (BADL) and instrumental (IADL) activities of daily living. Initial cognitive and functional evaluations were conducted within 14 days post-MIS onset, with follow-up assessments scheduled at 3–6 months. Cognitive impairment was defined using education-adjusted MoCA cutoff criteria based on Chinese normative data. Cognitive impairment thresholds were defined according to Chinese normative data with education-stratified cutoffs: ≤13 for illiterate individuals, ≤19 for those with 1–6 years of educational attainment, and ≤24 for individuals with 7 or more years of education (14). PSCIND diagnosis required: (a) MoCA scores below education-adjusted thresholds at 3–6 month follow-up; (b) no or mild impairment in both BADL and IADL, thereby excluding dementia (2, 15, 16). Based on these criteria, first-ever MIS patients were stratified into two groups: PSCIND and normal cognitive group.
Predictor selection and data acquisition
Candidate predictors were retrospectively collected from electronic medical records and structured clinical assessments. Demographic variables included age, sex, educational attainment, and body mass index. Clinical parameters comprised smoking/alcohol use history, type 2 diabetes mellitus (T2DM) diagnosis, hypertension status, coronary heart disease history, and admission systolic/diastolic blood pressure (SBP/DBP). Fasting venous blood samples (≥8 h) were analyzed for metabolic, inflammatory, oxidative stress, and coagulation biomarkers, including fasting plasma glucose (FPG), lipid profile components [triglycerides, total cholesterol, high-density, low-density and very low density lipoprotein cholesterol, apolipoprotein A, apolipoprotein B, and lipoprotein (a)], superoxide dismutase (SOD), homocysteine (Hcy), and uric acid (UA), and fibrinogen. Neurological evaluations incorporated NIHSS for acute stroke severity and the modified Rankin Scale (mRS) for functional outcomes. Ischemic stroke subtypes were classified into three categories based on neuroimaging characteristics: (1) multiple infarcts (≥2 distinct vascular territories), (2) strategic infarcts (functionally critical regions including the thalamus, caudate nucleus, frontal cortex, medial temporal lobe, and angular gyrus), or (3) small-artery occlusion (subcortical lesions <20 mm). Cerebral small vessel disease (CSVD) burden was quantified using a validated total CSVD score (range: 0–4) derived from four neuroimaging markers, as detailed in our prior studies (17). A threshold score >2 defined severe CSVD burden.
Statistical analysis
Statistical analyses were performed using SPSS 26.0 (IBM Corp., Armonk, NY) and R 4.2.3 (R Foundation for Statistical Computing, Vienna, Austria). Normally distributed continuous variables were summarized as mean ± standard deviation, non-normally distributed variables as median (interquartile range), and categorical variables as frequencies (%). Group comparisons employed independent t-tests for parametric data, Mann–Whitney U tests for nonparametric data, and χ2 tests for categorical variables, based on distributional assumptions. To mitigate multicollinearity and overfitting, least absolute shrinkage and selection operator (LASSO) regression (“glmnet” package) identified optimal predictors for PSCIND in first-ever MIS patients. Variables retained through LASSO regression were entered into multivariable logistic regression to develop the final predictive model, which was graphically represented through a static nomogram (“rms” package) and an interactive web-based visualization tool (“DynNom” and “shiny” packages) deployed as a Shiny application. Model evaluation encompassed four key assessments: (1) discrimination capacity measured by receiver operating characteristic (ROC) curve analysis [quantified by area under the curve (AUC)]; (2) calibration accuracy evaluated through calibration plots with Hosmer-Lemeshow testing; (3) clinical applicability determined via decision curve analysis (DCA); and (4) predictive overall performance quantified by the Brier score (<0.25 indicating superior performance). Internal validation with 2000 bootstrap resamples enhanced model reliability, with statistical significance set at two-tailed p < 0.05.
Results
Participants characteristics
The study cohort comprised 242 first-ever MIS patients with a median age of 65 years (male predominance: 67.8%). Participants were stratified into normal cognitive (n = 137) and PSCIND (n = 105) groups. Comparative analysis demonstrated that PSCIND patients were significantly older, had lower educational attainment, and exhibited higher prevalence of hypertension and T2DM history. Biochemical analyses identified distinct biomarker patterns, with the PSCIND group displaying reduced SOD levels concomitant with elevated UA, fibrinogen, and Hcy levels relative to cognitively normal subjects. Neuroimaging assessments revealed significant between-group differences in lesion distribution: PSCIND patients demonstrated greater prevalence of multiple infarcts (34.3% vs. 21.6%), strategic infarcts (63.8% vs. 39.4%), and severe CSVD burden (57.1% vs. 24.8%), alongside reduced incidence of small artery occlusion subtype (44.8% vs. 61.3%) compared to cognitively normal counterparts. All intergroup comparisons reached statistical significance (p < 0.05), with comprehensive comparative data detailed in Table 1.
Table 1
| Variable | Total (n = 242) |
Normal cognitive group (n = 137) |
PSCIND group (n = 105) | p value |
|---|---|---|---|---|
| Age, median (IQR), year | 65 (58–73) | 63 (56–71) | 68 (63–75) | < 0.001* |
| Sex (male), n (%) | 164 (67.8) | 88 (64.2) | 76 (72.4) | 0.179 |
| Education, median (IQR), year | 9 (6–14) | 12 (9–15) | 9 (6–12) | 0.001* |
| Body mass index, median (IQR), kg/m2 | 25.0 (22.9–27.6) | 25.0 (23.0–27.4) | 25.0 (22.9–27.7) | 0.895 |
| Smoking, n (%) | 108 (44.6) | 61 (44.5) | 47 (44.8) | 0.971 |
| Alcohol use, n (%) | 71 (29.3) | 38 (27.7) | 33 (31.4) | 0.532 |
| Hypertension, n (%) | 148 (61.2) | 75 (54.7) | 73 (69.5) | 0.019* |
| SBP, median (IQR), mmHg | 148 (136–162) | 147 (136–162) | 148 (136–163) | 0.976 |
| DBP, mean ± SD, mmHg | 84.6 ± 11.9 | 85.2 ± 11.8 | 83.8 ± 12.1 | 0.366 |
| T2DM, n (%) | 70 (28.9) | 32 (23.4) | 38 (36.2) | 0.029* |
| Coronary heart disease, n (%) | 29 (12.0) | 16 (11.7) | 13 (12.4) | 0.868 |
| FPG, median (IQR), mmol/L | 5.25 (4.72–6.75) | 5.29 (4.74–6.76) | 5.16 (4.72–6.71) | 0.678 |
| TG, median (IQR), mmol/L | 1.29 (0.96–1.78) | 1.28 (0.95–1.73) | 1.31 (0.96–1.89) | 0.795 |
| TC, median (IQR), mmol/L | 4.57 (3.79–5.23) | 4.59 (3.92–5.22) | 4.38 (3.64–5.26) | 0.234 |
| HDL-C, median (IQR), mmol/L | 1.06 (0.91–1.24) | 1.07 (0.91–1.26) | 1.03 (0.91–1.20) | 0.229 |
| LDL-C, median (IQR), mmol/L | 2.99 (2.40–3.50) | 3.05 (2.50–3.51) | 2.85 (2.35–3.48) | 0.145 |
| VLDL-C, median (IQR), mmol/L | 0.47 (0.28–0.65) | 0.46 (0.28–0.63) | 0.48 (0.28–0.70) | 0.608 |
| ApoA1, median (IQR), g/L | 1.18 (1.03–1.36) | 1.20 (1.04–1.37) | 1.17 (1.01–1.36) | 0.297 |
| ApoB, median (IQR), g/L | 0.81 (0.69–0.96) | 0.83 (0.70–0.97) | 0.79 (0.68–0.94) | 0.252 |
| Lp (a), median (IQR), mg/L | 178 (100–313) | 171 (95–317) | 186 (103–310) | 0.648 |
| SOD, median (IQR), U/mL | 142 (129–156) | 150 (137–162) | 133 (120–143) | <0.001* |
| UA, median (IQR), μmol/L | 296 (244–352) | 281 (225–338) | 308 (267–372) | 0.001* |
| Fibrinogen, median (IQR), g/L | 2.85 (2.40–3.34) | 2.72 (2.26–3.16) | 3.02 (2.66–3.52) | <0.001* |
| Hcy, median (IQR), umol/L | 14.3 (11.8–19.5) | 12.9 (10.6–17.7) | 16.8 (13.3–22.8) | <0.001* |
| NIHSS, median (IQR), score | 2 (1–3) | 2 (1–3) | 2 (1–3) | 0.514 |
| MRS, median (IQR), score | 2 (1–3) | 2 (1–3) | 2 (2–3) | 0.099 |
| Multiple infarcts, n (%) | 66 (27.3) | 30 (21.6) | 36 (34.3) | 0.032* |
| Strategic infarcts, n (%) | 121 (50.0) | 54 (39.4) | 67 (63.8) | <0.001* |
| Small-artery occlusion, n (%) | 131 (54.1) | 84 (61.3) | 47 (44.8) | 0.010* |
| Severe CSVD burden, n (%) | 94 (38.8) | 34 (24.8) | 60 (57.1) | <0.001* |
Characteristics of the study patients with first-ever MIS between normal cognitive group and PSCIND group.
PSCIND, post-stroke cognitive impairment no dementia; IQR, interquartile range; SD, standard deviation; SBP, systolic blood pressure; DBP, diastolic blood pressure; T2DM, type 2 diabetes mellitus; FPG, fasting plasma glucose; TG, triglyceride; TC, total cholesterol; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; VLDL-C, very low density lipoprotein cholesterol; ApoA1, apolipoprotein A1; ApoB, apolipoprotein B; Lp (a), lipoprotein (a); SOD, superoxide dismutase; UA, uric acid; Hcy, Homocysteine; NIHSS, National Institutes of Health Stroke Scale; mRS, modified Rankin Scale. *Denotes significance at a p value of <0.05.
Predictor selection for predictive model
All 30 candidate variables listed in Table 1 were subjected to LASSO regression analysis for PSCIND predictor identification. Figure 1A illustrates the coefficient trajectories across varying regularization parameters, demonstrating progressive feature shrinkage as λ increases. The optimal regularization parameter was determined through 10-fold cross-validation (Figure 1B), where model selection occurred at the λ value corresponding to the minimum deviance plus one standard error (right vertical dashed line). This analytical approach ultimately selected eight predictors retaining non-zero coefficients: age, education level, T2DM status, SOD levels, UA levels, Hcy levels, strategic infarcts, and severe CSVD burden.
Figure 1

Variable selection by LASSO regression model. (A) LASSO coefficient profiles of the 30 variables. (B) Eight variables with nonzero coefficients were selected by optimal λ. The two dotted vertical lines were drawn at the optimal scores by minimum criteria and 1 standard error criteria.
Predictive model development and visualization
A multivariablelogistic regression model incorporating eight LASSO-selected predictors (age, education level, T2DM, SOD, UA, Hcy, strategic infarcts, and severe CSVD burden) was established to assess PSCIND risk in first-ever MIS patients. The analysis demonstrated significant associations between these predictors and PSCIND in Table 2: age (OR = 1.060, 95% CI: 1.016–1.106; p = 0.007), education level (OR = 0.917, 95% CI: 0.845–0.995; p = 0.037), T2DM (OR = 9.407, 95% CI: 3.761–23.528; p < 0.001), SOD (OR = 0.951, 95% CI: 0.931–0.972; p < 0.001), UA (OR = 1.006, 95% CI: 1.002–1.010; p = 0.005), Hcy (OR = 1.058, 95% CI: 1.027–1.091; p = 0.013), strategic infarcts (OR = 4.566, 95% CI: 2.148–9.707; p < 0.001), and severe CSVD burden (OR = 3.818, 95% CI: 1.842–7.911; p < 0.001).
Table 2
| Variable | β | SE | Wald χ2 | p value | OR | 95% CI |
|---|---|---|---|---|---|---|
| Age | 0.059 | 0.022 | 7.350 | 0.007 | 1.060 | 1.016–1.106 |
| Education | −0.087 | 0.041 | 4.355 | 0.037 | 0.917 | 0.845–0.995 |
| T2DM | 2.241 | 0.468 | 22.967 | <0.001 | 9.407 | 3.761–23.528 |
| SOD | −0.050 | 0.011 | 20.651 | <0.001 | 0.951 | 0.931–0.972 |
| UA | 0.006 | 0.002 | 7.861 | 0.005 | 1.006 | 1.002–1.010 |
| Hcy | 0.057 | 0.015 | 13.602 | 0.013 | 1.058 | 1.027–1.091 |
| Strategic infarcts | 1.519 | 0.385 | 15.578 | <0.001 | 4.566 | 2.148–9.707 |
| Severe CSVD burden | 1.340 | 0.372 | 12.988 | <0.001 | 3.818 | 1.842–7.911 |
Multivariate logistic regression analysis of the associated predictors for the risk of PSCIND in patients with first-ever MIS.
A clinical nomogram incorporating these predictors was constructed to estimate individualized PSCIND risk (Figure 2). Each predictor is assigned points on the upper axis, with total scores corresponding to predicted probabilities on the lower scale through vertical alignment. To enhance clinical utility, we developed an interactive web-based nomogram.2 For instance, a 60-year-old patient with 12-year education, T2DM history, Hcy 14 μmol/L, SOD 128 U/mL, UA 286 μmol/L, thalamic infarct, and CSVD score of 2 would have a 78.4% predicted probability of developing PSCIND (Figure 3).
Figure 2
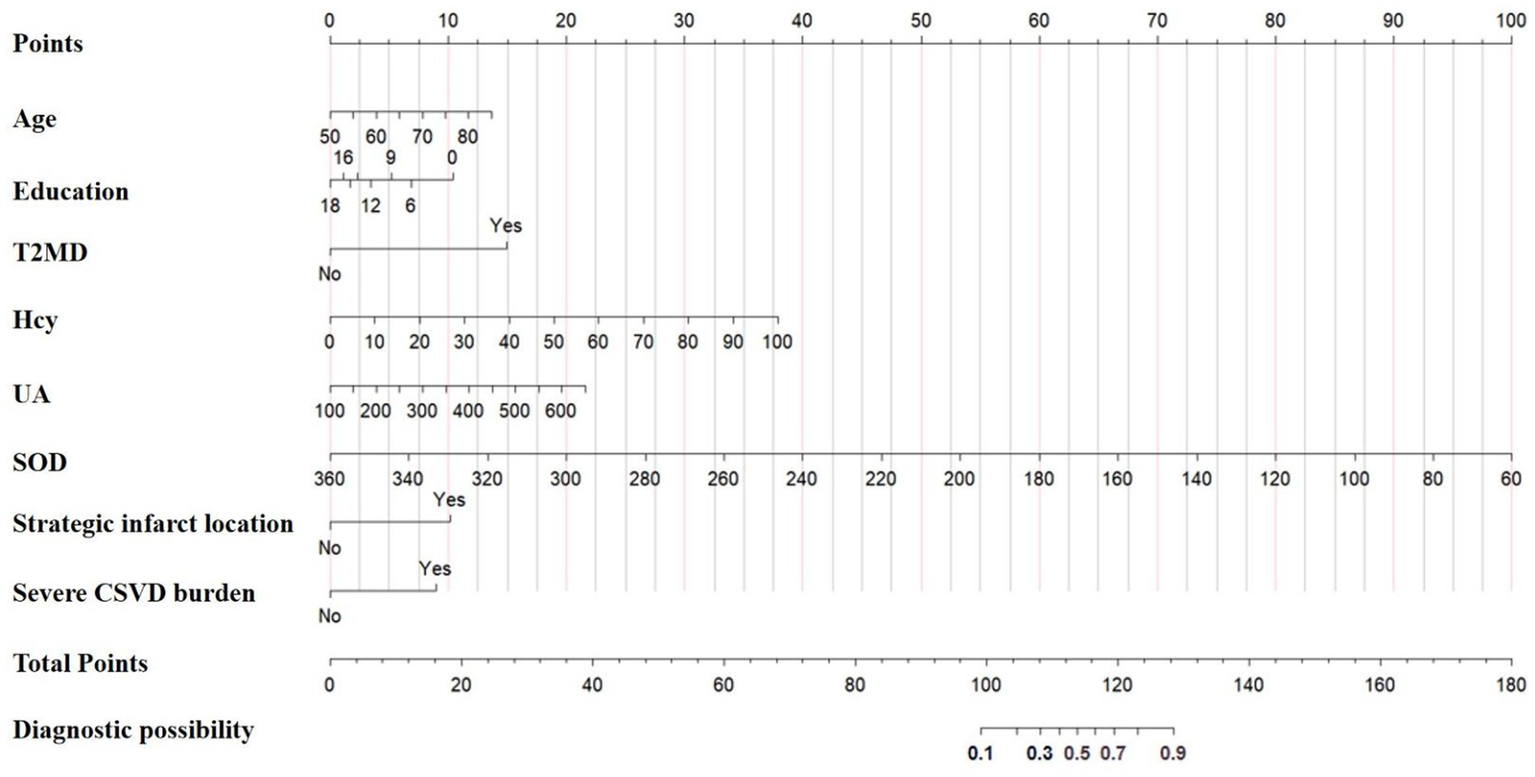
Nomogram for the prediction of the probability of PSCIND risk in patients with first-ever MIS.
Figure 3

Dynamic nomogram for the prediction of the probability of PSCIND risk in patients with first-ever MIS. An example for predicting the probability of having PSCIND risk in a first-ever MIS with online dynamic nomogram. A 60-year-old patient with 12-year education, T2DM history, hcy 14 μmol/L, SOD 128 U/mL, UA 286 μmol/L, thalamic infarct, and CSVD score of 2 demonstrates a predicted 78.4% probability of developing PSCIND.
Predictive model validation
The model demonstrated excellent discriminative capacity through ROC curve analysis with 2000 bootstrap resampling validations, achieving an AUC of 0.886 (95% CI: 0.844–0.929) (Figure 4). Calibration accuracy was confirmed via bootstrap-corrected calibration curves (2000 iterations) and Hosmer-Lemeshow test (χ2 = 14.542, p = 0.104), demonstrating strong concordance between predicted and observed probabilities (Figure 5). The model achieved a Brier score of 0.135 (<0.25 threshold), confirming superior overall predictive accuracy. DCA demonstrated enhanced clinical utility across 6–100% threshold probabilities, outperforming alternative strategies in net clinical benefit (Figure 6).
Figure 4
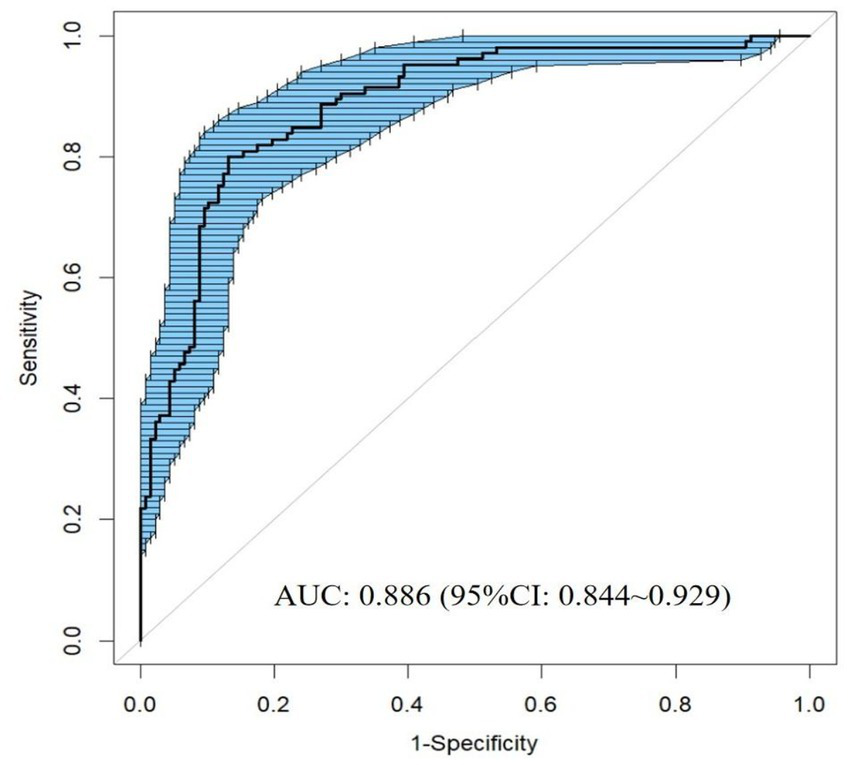
ROC curve of the nomogram for predicting PSCIND in patients with first-ever MIS.
Figure 5
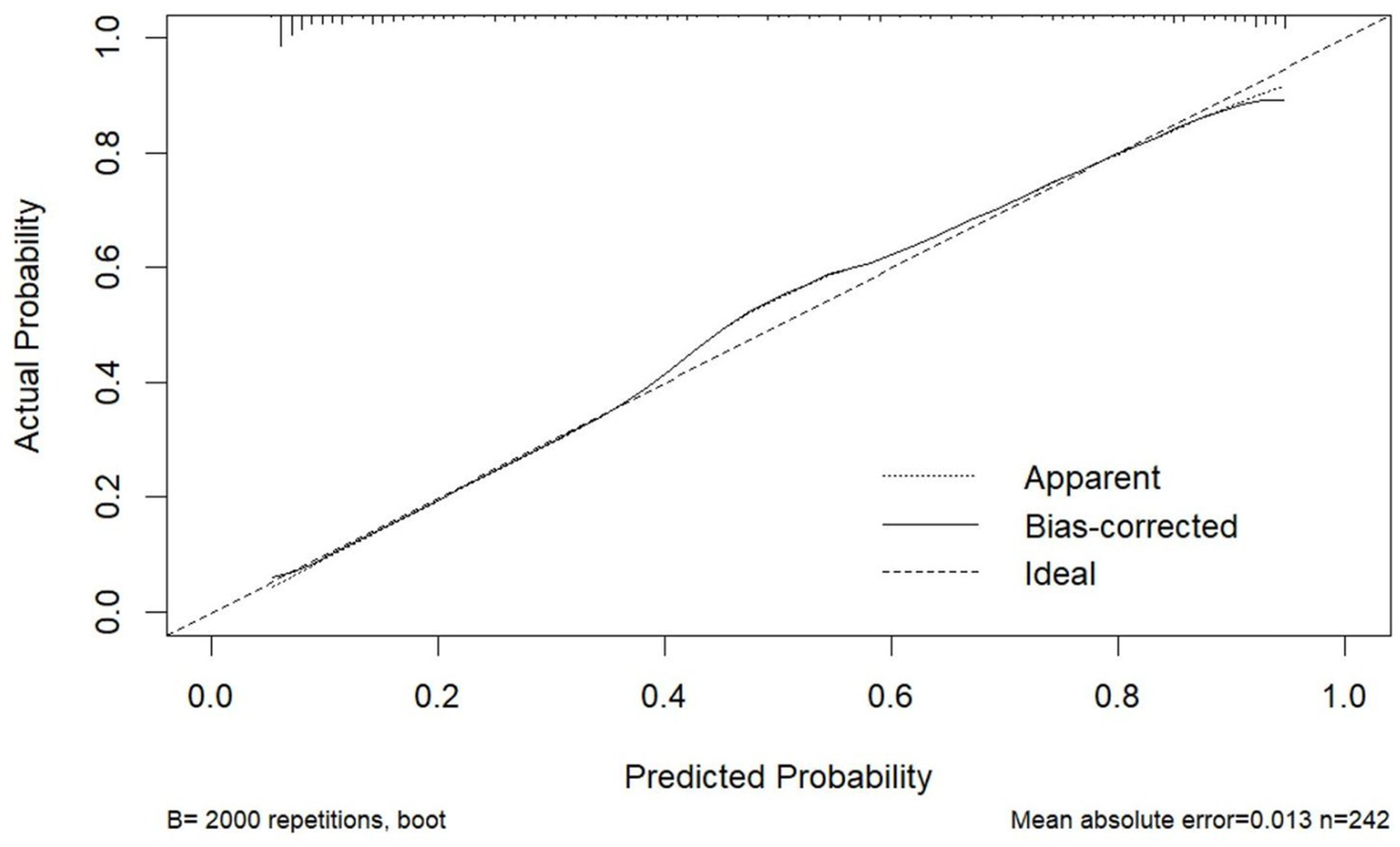
Calibration curve of the nomogram for predicting PSCIND in patients with first-ever MIS.
Figure 6
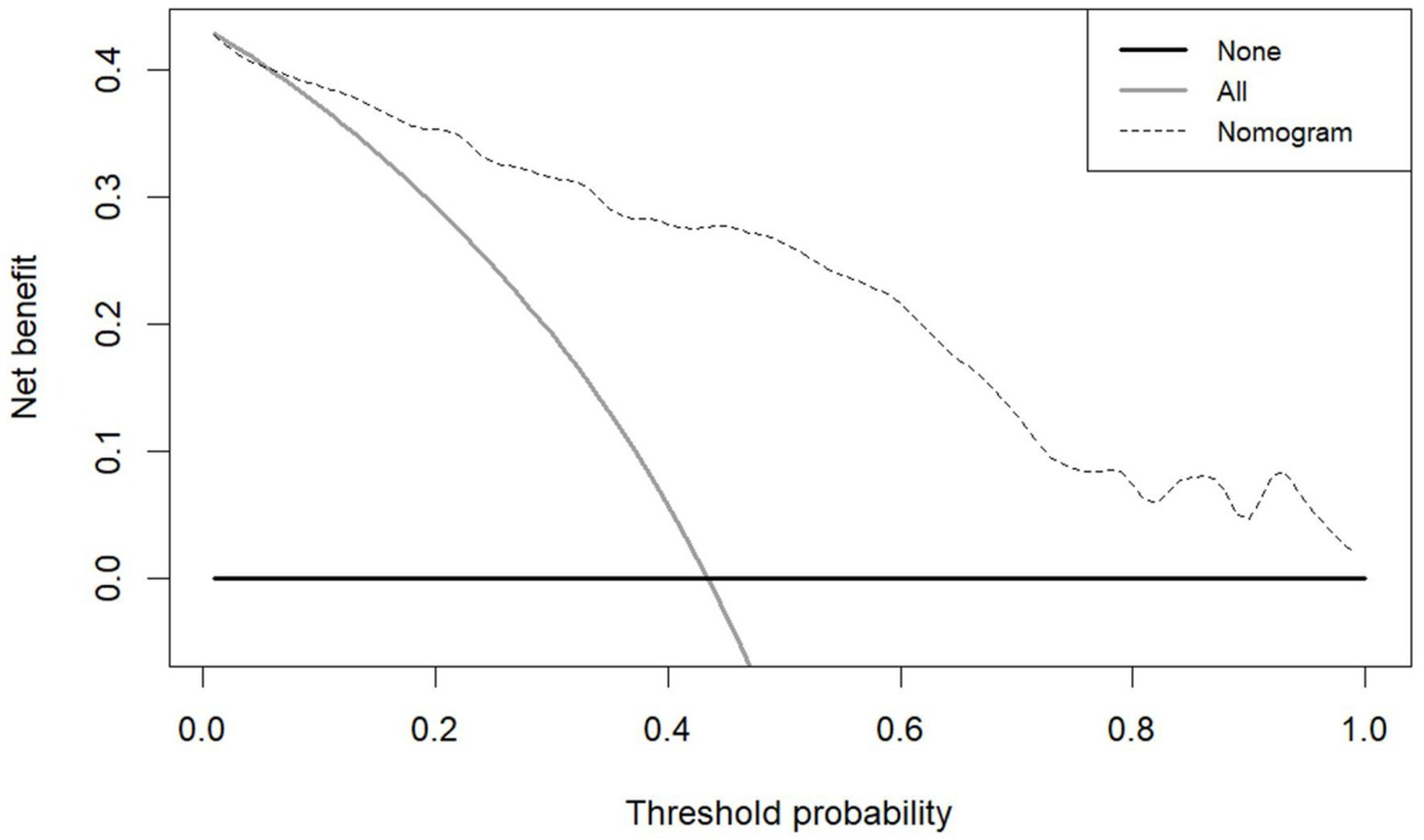
DCA of the nomogram for predicting PSCIND in patients with first-ever MIS.
Discussion
This study revealed a 43.4% prevalence of PSCIND in first-ever MIS patients and developed a validated clinical prediction model incorporating eight predictors: age, education level, T2DM, SOD, UA, Hcy, strategic infarcts, and severe CSVD burden. The model demonstrated robust discriminative capacity, excellent calibration, and clinical applicability, providing clinicians with a practical tool for early risk stratification in this population.
Advanced age and lower education emerged as critical demographic predictors, with each additional year of age (50–85 years) increasing PSCIND risk by 6% and each year of education reducing risk by 8%. These findings align with global evidence on socioeconomic and biological determinants of post-stroke cognitive decline (2, 18). Notably, T2DM exhibited the strongest predictive value (OR = 9.407), aligning with meta-analyses identifying diabetes as a principal risk modulator for PSCI (4).
Blood-based biomarker research has significantly improved PSCI diagnostic accuracy (19–21). Hcy has garnered significant attention as a readily measurable biomarker linked to neurological disorders, with emerging evidence supporting its role in PSCI and other cognitive impairments (22). A meta-analysis highlighted elevated Hcy as a potential early diagnostic marker for PSCI (23), aligning with our findings that higher Hcy levels significantly predicted PSCIND risk in first-ever MIS. However, a multicenter prospective study in MIS (NIHSS≤3) and transient ischemic attack patients reported no association between Hcy and 3-month PSCI incidence, though a gender-specific risk emerged at 12 months (24). These discrepancies may stem from cohort heterogeneity (e.g., stroke severity thresholds) and inadequate stratification of PSCI subtypes (e.g., PSCIND vs. dementia), underscoring the need for extended follow-up and subgroup analyses by sex/age in future studies.
The prognostic utility of UA exhibits ongoing controversy: while multiple studies have identified elevated serum UA levels as a sensitive predictor for PSCI development (25, 26), others paradoxically suggest a protective effect of hyperuricemia against cognitive decline (27). Data from the China National Stroke Registry-III revealed a U-shaped relationship between UA levels and PSCI risk in males (28), yet a meta-analysis found no diagnostic utility for UA in PSCI prediction (29). Notably, prior studies often neglected PSCI subtype stratification, ischemic lesion assessment, and critical imaging markers like CSVD burden. By systematically accounting for these factors, our study identified elevated UA as a significant PSCIND predictor in first-ever MIS patients. This finding underscores the necessity of contextualizing UA measurements within broader cerebrovascular evaluations, though large-scale multicenter validation is warranted.
SOD, a pivotal antioxidant enzyme, indirectly reflects the systemic capacity to eliminate oxygen free radicals and is crucial in mitigating oxidative stress (30, 31). Experimental and clinical data demonstrate that reduced SOD activity contributes to cognitive disorder pathogenesis through chronic oxidative damage, vascular endothelial impairment, and blood–brain barrier compromise (32–34). Despite its pathophysiological significance, SOD has been insufficiently investigated in PSCI research. Although a previous study identified an association between low SOD levels and PSCI risk in MIS patients (NIHSS≤8), the investigation omitted critical exclusion criteria (e.g., recurrent strokes potentially confounding cognitive trajectories) and neglected PSCI subtype stratification (34). Our study newly establishes diminished SOD activity as an independent predictor (OR = 0.951), mechanistically implicating oxidative stress in PSCIND pathogenesis while highlighting antioxidant supplementation as a plausible therapeutic strategy-an innovative dimension previously absent in predictive models.
The neuroanatomical distribution of ischemic stroke lesions significantly influences the risk of PSCI (35). Strategic infarcts, defined as lesions affecting brain regions critical for higher cortical functions-including the thalamus, caudate nucleus, frontal cortex, medial temporal lobe (including the hippocampus), and angular gyrus-have been identified as independent PSCI risk factors (36, 37). Previous studies have demonstrated the particular vulnerability of left middle frontal gyrus, anterior thalamic nuclei, and left angular gyrus in PSCI pathogenesis (37, 38). A study synthesizing data from 12 acute ischemic stroke cohorts further highlighted the strong association between PSCI and infarcts in the left frontotemporal lobe, left thalamus, and right parietal lobe (36). Our findings corroborate this evidence, identifying strategic infarcts as significant predictors of PSCIND in MIS patients (OR = 4.566). Nevertheless, the lack of granular anatomical stratification of infarct locations in our analysis highlights the need for future investigations to delineate region-specific contributions to cognitive outcomes.
Compared to isolated CSVD imaging markers, CSVD burden assessment provides a more comprehensive evaluation of cerebrovascular injury and better aligns with cognitive diagnostic frameworks (39, 40). Sung et al. (41) demonstrated that severe baseline CSVD burden (total score ≥3) independently predicts 1-year PSCI incidence in first-ever MIS patients-a finding paralleling our observed association between severe CSVD burden and elevated PSCIND risk (OR = 3.818). However, Du et al. (42) proposed an indirect relationship, where CSVD burden influences PSCI via disrupted brain network connectivity rather than direct effects. A multicenter prospective study substantiated the prognostic value of CSVD burden, correlating baseline scores with impaired executive function, attention, and visuospatial abilities at 12-month follow-up (43). These collective findings underscore the necessity of incorporating CSVD burden-rather than isolated markers-into PSCIND prediction models for first-ever MIS patients.
This study introduces three principal advancements: (1) The first dedicated model for first-ever MIS patients; (2) Incorporation of novel oxidative stress (SOD) and neuroimaging (CSVD burden) biomarkers; (3) Development of an interactive web-based nomogram enhancing clinical translation. However, several limitations warrant consideration. First, its retrospective design and single-center data may introduce selection bias and limit generalizability. Second, the moderate sample size restricts statistical power for subgroup analyses. Third, potential attrition bias must be acknowledged, as 76 patients (23.9% of eligible cohort) were lost to follow-up. Fourth, although CSVD burden was incorporated as a composite metric, constituent markers were not individually assessed. Fifth, Broad “strategic infarct” categorization obscures region-specific effects, necessitating finer anatomical stratification in future work. Prospective multi-center studies with larger cohorts and extended follow-up periods are needed to validate these findings and explore temporal biomarker dynamics.
Conclusion
We developed and validated an eight-predictor nomogram integrating demographic, biochemical, and neuroimaging parameters to assess PSCIND risk in first-ever MIS patients. The model’s discriminative accuracy and dynamic visualization interface facilitate early identification of high-risk individuals, enabling targeted interventions to mitigate cognitive decline. While demonstrating immediate clinical value, future large-scale multicenter studies integrating multi-omics approaches and neuroimaging biomarkers are warranted to enhance clinical implementation potential and elucidate underlying pathophysiological mechanisms.
Statements
Data availability statement
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding author/s.
Ethics statement
The studies involving humans were approved by the Ethics Committee of Hebei General Hospital (Approval number: 2025-LW-0124). The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation was not required from the participants or the participants' legal guardians/next of kin in accordance with the national legislation and institutional requirements.
Author contributions
ZT: Funding acquisition, Formal analysis, Methodology, Software, Writing – original draft. JF: Writing – original draft, Formal analysis, Software, Data curation, Investigation, Conceptualization. XX: Methodology, Formal analysis, Supervision, Writing – original draft. TG: Conceptualization, Methodology, Data curation, Writing – original draft. JX: Methodology, Writing – review & editing. XJ: Writing – original draft, Visualization, Conceptualization, Validation. YC: Supervision, Methodology, Writing – review & editing. PL: Funding acquisition, Validation, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the 2023 Medical Scientific Research Project of Hebei Provincial Health Commission (No. 20230380), the Scientific and Technological Innovation 2030-Major Project Subject of “Brain Science and Brain-inspired Research” (No. 2021ZD0201807), and the 2023 Hebei Province Government-funded Excellent Talents Project in Clinical Medicine (No. ZF2023177).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that no Gen AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fneur.2025.1618953/full#supplementary-material
Footnotes
1.^ MoCA; www.mocatest.org.
2.^ Accessible at https://tengzhenjie.shinyapps.io/DynNomapp4/.
References
1.
GBD 2021 Stroke Risk Factor Collaborators . Global, regional, and national burden of stroke and its risk factors, 1990-2021: a systematic analysis for the global burden of disease study 2021. Lancet Neurol. (2024) 23:973–1003. doi: 10.1016/S1474-4422(24)00369-7
2.
Quinn TJ Richard E Teuschl Y Gattringer T Hafdi M O'Brien JT et al . European stroke organisation and European academy of neurology joint guidelines on post-stroke cognitive impairment. Eur J Neurol. (2021) 28:3883–920. doi: 10.1111/ene.15068
3.
Gallucci L Sperber C Guggisberg AG Kaller CP Heldner MR Monsch AU et al . Post-stroke cognitive impairment remains highly prevalent and disabling despite state-of-the-art stroke treatment. Int J Stroke. (2024) 19:888–97. doi: 10.1177/17474930241238637
4.
Lo JW Crawford JD Desmond DW Godefroy O Jokinen H Mahinrad S et al . Profile of and risk factors for poststroke cognitive impairment in diverse ethnoregional groups. Neurology. (2019) 93:e2257–71. doi: 10.1212/WNL.0000000000008612
5.
El HN Katzan IL Rost NS Blake ML Byun E Pendlebury ST et al . Cognitive impairment after ischemic and hemorrhagic stroke: a scientific statement from the American Heart Association/American Stroke Association. Stroke. (2023) 54:e272–91. doi: 10.1161/STR.0000000000000430
6.
Sexton E McLoughlin A Williams DJ Merriman NA Donnelly N Rohde D et al . Systematic review and meta-analysis of the prevalence of cognitive impairment no dementia in the first year post-stroke. Eur Stroke J. (2019) 4:160–71. doi: 10.1177/2396987318825484
7.
Merriman NA Sexton E McCabe G Walsh ME Rohde D Gorman A et al . Addressing cognitive impairment following stroke: systematic review and meta-analysis of non-randomised controlled studies of psychological interventions. BMJ Open. (2019) 9:e24429. doi: 10.1136/bmjopen-2018-024429
8.
Feigin VL Brainin M Norrving B Martins S Sacco RL Hacke W et al . Global stroke fact sheet 2022. Int J Stroke. (2022) 17:18–29. doi: 10.1177/17474930211065917
9.
Dawson J Merwick A Webb A Dennis M Ferrari J Fonseca AC . European stroke organisation expedited recommendation for the use of short-term dual antiplatelet therapy early after minor stroke and high-risk TIA. Eur Stroke J. (2021) 6:CLXXXVII–CXCI. doi: 10.1177/23969873211000877
10.
Roberts PS Krishnan S Burns SP Ouellette D Pappadis MR . Inconsistent classification of mild stroke and implications on health services delivery. Arch Phys Med Rehabil. (2020) 101:1243–59. doi: 10.1016/j.apmr.2019.12.013
11.
Wang Y Jing J Meng X Pan Y Wang Y Zhao X et al . The third China National Stroke Registry (CNSR-III) for patients with acute ischaemic stroke or transient ischaemic attack: design, rationale and baseline patient characteristics. Stroke Vasc Neurol. (2019) 4:158–64. doi: 10.1136/svn-2019-000242
12.
Aam S Einstad MS Munthe-Kaas R Lydersen S Ihle-Hansen H Knapskog AB et al . Post-stroke cognitive impairment-impact of follow-up time and stroke subtype on severity and cognitive profile: the nor-COAST study. Front Neurol. (2020) 11:699. doi: 10.3389/fneur.2020.00699
13.
Moons KG Altman DG Reitsma JB Ioannidis JP Macaskill P Steyerberg EW et al . Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): explanation and elaboration. Ann Intern Med. (2015) 162:W1–W73. doi: 10.7326/M14-0698
14.
Lu J Li D Li F Zhou A Wang F Zuo X et al . Montreal cognitive assessment in detecting cognitive impairment in Chinese elderly individuals: a population-based study. J Geriatr Psychiatry Neurol. (2011) 24:184–90. doi: 10.1177/0891988711422528
15.
Pedrosa H De Sa A Guerreiro M Maroco J Simoes MR Galasko D et al . Functional evaluation distinguishes MCI patients from healthy elderly people-the ADCS/MCI/ADL scale. J Nutr Health Aging. (2010) 14:703–9. doi: 10.1007/s12603-010-0102-1
16.
Skrobot OA Black SE Chen C DeCarli C Erkinjuntti T Ford GA et al . Progress toward standardized diagnosis of vascular cognitive impairment: guidelines from the vascular impairment of cognition classification consensus study. Alzheimers Dement. (2018) 14:280–92. doi: 10.1016/j.jalz.2017.09.007
17.
Teng Z Feng J Dong Y Xu J Jiang X Chen H et al . Triglyceride glucose index is associated with cerebral small vessel disease burden and cognitive impairment in elderly patients with type 2 diabetes mellitus. Front Endocrinol (Lausanne). (2022) 13:970122. doi: 10.3389/fendo.2022.970122
18.
Mtambo ML Ragunathan T Mohan D Warren N Su TT Quek KF . Post-stroke cognitive impairment and dementia research in Southeast Asia: a scoping review. Asian J Psychiatr. (2025) 107:104460. doi: 10.1016/j.ajp.2025.104460
19.
Kim KY Shin KY Chang KA . Potential biomarkers for post-stroke cognitive impairment: a systematic review and Meta-analysis. Int J Mol Sci. (2022) 23:602. doi: 10.3390/ijms23020602
20.
Lu Q Yu A Pu J Chen D Zhong Y Bai D et al . Post-stroke cognitive impairment: exploring molecular mechanisms and omics biomarkers for early identification and intervention. Front Mol Neurosci. (2024) 17:1375973. doi: 10.3389/fnmol.2024.1375973
21.
Li H Ke X Feng B Tian H Cai Z Zhang A et al . Research progress on the mechanism and markers of metabolic disorders in the occurrence and development of cognitive dysfunction after ischemic stroke. Front Endocrinol (Lausanne). (2025) 16:1500650. doi: 10.3389/fendo.2025.1500650
22.
Smith AD Refsum H . Homocysteine - from disease biomarker to disease prevention. J Intern Med. (2021) 290:826–54. doi: 10.1111/joim.13279
23.
Yang Y Chen Y Yang Y Yang T Wu T Chen J et al . Prediction models for post-stroke cognitive impairment: a systematic review and Meta-analysis. Public Health Nurs. (2025) 42:1375–88. doi: 10.1111/phn.13509
24.
Li R Weng H Pan Y Meng X Liao X Wang M et al . Relationship between homocysteine levels and post-stroke cognitive impairment in female and male population: from a prospective multicenter study. J Transl Int Med. (2021) 9:264–72. doi: 10.2478/jtim-2021-0035
25.
Sun J Lv X Gao X Chen Z Wei D Ling Y et al . The association between serum uric acid level and the risk of cognitive impairment after ischemic stroke. Neurosci Lett. (2020) 734:135098. doi: 10.1016/j.neulet.2020.135098
26.
Ma Y Chen Y Yang T He X Yang Y Chen J et al . Blood biomarkers for post-stroke cognitive impairment: a systematic review and meta-analysis. J Stroke Cerebrovasc Dis. (2024) 33:107632. doi: 10.1016/j.jstrokecerebrovasdis.2024.107632
27.
Ran F Liu F Zhang Y Chen L . Serum uric acid and high-sensitivity C-reactive protein as predictors of cognitive impairment in patients with cerebral infarction. Dement Geriatr Cogn Disord. (2020) 49:235–42. doi: 10.1159/000507806
28.
Liu Q Liao X Pan Y Jin A Zhang Y . Association between serum uric acid levels and cognitive function in patients with ischemic stroke and transient ischemic attack (TIA): a 3-month follow-up study. Neuropsychiatr Dis Treat. (2021) 17:991–9. doi: 10.2147/NDT.S300893
29.
Jiang Z Li M Wang K Duan H Zhang B Fang S . Potential biomarkers of post-stroke cognitive impairment in Chinese population: a systematic review and Meta-analysis. Mol Neurobiol. (2025) 62:8670–94. doi: 10.1007/s12035-025-04792-x
30.
Fukai T Ushio-Fukai M . Superoxide dismutases: role in redox signaling, vascular function, and diseases. Antioxid Redox Signal. (2011) 15:1583–606. doi: 10.1089/ars.2011.3999
31.
Fanoudi S Alavi MS Hosseini M Sadeghnia HR . Nigella sativa and thymoquinone attenuate oxidative stress and cognitive impairment following cerebral hypoperfusion in rats. Metab Brain Dis. (2019) 34:1001–10. doi: 10.1007/s11011-019-00394-4
32.
Fracassi A Marcatti M Zolochevska O Tabor N Woltjer R Moreno S et al . Oxidative damage and antioxidant response in frontal cortex of demented and nondemented individuals with Alzheimer's neuropathology. J Neurosci. (2021) 41:538–54. doi: 10.1523/JNEUROSCI.0295-20.2020
33.
Zhu S Wei X Yang X Huang Z Chang Z Xie F et al . Plasma lipoprotein-associated phospholipase a2 and superoxide dismutase are independent predicators of cognitive impairment in cerebral small vessel disease patients: diagnosis and assessment. Aging Dis. (2019) 10:834–46. doi: 10.14336/AD.2019.0304
34.
Zhang MS Liang JH Yang MJ Ren YR Cheng DH Wu QH et al . Low serum superoxide dismutase is associated with a high risk of cognitive impairment after mild acute ischemic stroke. Front Aging Neurosci. (2022) 14:834114. doi: 10.3389/fnagi.2022.834114
35.
Pantoni L Salvadori E . Location of infarcts and post-stroke cognitive impairment. Lancet Neurol. (2021) 20:413–4. doi: 10.1016/S1474-4422(21)00107-1
36.
Weaver NA Kuijf HJ Aben HP Abrigo J Bae HJ Barbay M et al . Strategic infarct locations for post-stroke cognitive impairment: a pooled analysis of individual patient data from 12 acute ischaemic stroke cohorts. Lancet Neurol. (2021) 20:448–59. doi: 10.1016/S1474-4422(21)00060-0
37.
Zhao L Biesbroek JM Shi L Liu W Kuijf HJ Chu WW et al . Strategic infarct location for post-stroke cognitive impairment: a multivariate lesion-symptom mapping study. J Cereb Blood Flow Metab. (2018) 38:1299–311. doi: 10.1177/0271678X17728162
38.
Puy L Barbay M Roussel M Canaple S Lamy C Arnoux A et al . Neuroimaging determinants of post stroke cognitive performance. Stroke. (2018) 49:2666–73. doi: 10.1161/STROKEAHA.118.021981
39.
Duering M Biessels GJ Brodtmann A Chen C Cordonnier C de Leeuw FE et al . Neuroimaging standards for research into small vessel disease-advances since 2013. Lancet Neurol. (2023) 22:602–18. doi: 10.1016/S1474-4422(23)00131-X
40.
Li X Yuan J Qin W Yang L Yang S Li Y et al . Higher total cerebral small vessel disease burden was associated with mild cognitive impairment and overall cognitive dysfunction: a propensity score-matched case-control study. Front Aging Neurosci. (2021) 13:695732. doi: 10.3389/fnagi.2021.695732
41.
Sung PS Lee KP Lin PY Su HC Yu RL Tsai KJ et al . Factors associated with cognitive outcomes after first-ever ischemic stroke: the impact of small vessel disease burden and neurodegeneration. J Alzheimer's Dis. (2021) 83:569–79. doi: 10.3233/JAD-210587
42.
Du J Wang Y Zhi N Geng J Cao W Yu L et al . Structural brain network measures are superior to vascular burden scores in predicting early cognitive impairment in post stroke patients with small vessel disease. Neuroimage Clin. (2019) 22:101712. doi: 10.1016/j.nicl.2019.101712
43.
Georgakis MK Fang R During M Wollenweber FA Bode FJ Stosser S et al . Cerebral small vessel disease burden and cognitive and functional outcomes after stroke: a multicenter prospective cohort study. Alzheimers Dement. (2023) 19:1152–63. doi: 10.1002/alz.12744
Summary
Keywords
post-stroke cognitive impairment, mild ischemic stroke, cerebral small vessel disease, nomogram, prediction model
Citation
Teng Z, Feng J, Xie X, Guan T, Xu J, Jiang X, Chang Y and Lv P (2025) A nomogram for predicting post-stroke cognitive impairment no dementia in patients with first-ever mild ischemic stroke. Front. Neurol. 16:1618953. doi: 10.3389/fneur.2025.1618953
Received
27 April 2025
Accepted
08 August 2025
Published
19 August 2025
Volume
16 - 2025
Edited by
Robert T. Mallet, University of North Texas Health Science Center, United States
Reviewed by
Mohamed G. Zeinhom, Kafrelsheikh University, Egypt
Mohamed Derhab, Mayo Clinic, United States
Updates
Copyright
© 2025 Teng, Feng, Xie, Guan, Xu, Jiang, Chang and Lv.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Peiyuan Lv, peiyuanlu2@163.comYanzhong Chang, yzchang@hebtu.edu.cn
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.