- Pulmonary Research Group, Department of Medicine, University of Alberta, Edmonton, AB, Canada
Eosinophils derive from the bone marrow and circulate at low levels in the blood in healthy individuals. These granulated cells preferentially leave the circulation and marginate to tissues, where they are implicated in the regulation of innate and adaptive immunity. In diseases such as allergic inflammation, eosinophil numbers escalate markedly in the blood and tissues where inflammatory foci are located. Eosinophils possess a range of immunomodulatory factors that are released upon cell activation, including over 35 cytokines, growth factors, and chemokines. Unlike T and B cells, eosinophils can rapidly release cytokines within minutes in response to stimulation. While some cytokines are stored as pre-formed mediators in crystalloid granules and secretory vesicles, eosinophils are also capable of undergoing de novo synthesis and secretion of these immunological factors. Some of the molecular mechanisms that coordinate the final steps of cytokine secretion are hypothesized to involve binding of membrane fusion complexes comprised of soluble N-ethylmaleimide sensitive factor attachment protein receptors (SNAREs). These intracellular receptors regulate the release of granules and vesicles containing a range of secreted proteins, among which are cytokines and chemokines. Emerging evidence from both human and animal model-based research has suggested an active participation of eosinophils in several physiological/pathological processes such as immunomodulation and tissue remodeling. The observed eosinophil effector functions in health and disease implicate eosinophil cytokine secretion as a fundamental immunoregulatory process. The focus of this review is to describe the cytokines, growth factors, and chemokines that are elaborated by eosinophils, and to illustrate some of the intracellular events leading to the release of eosinophil-derived cytokines.
Introduction
Eosinophils are granulocytic white blood cells that are rare in healthy individuals, but become elevated in both blood and tissue compartments in helminthic parasite infection as well as allergic inflammation, particularly in late-onset persistent eosinophilic asthma (1). Typically, the number of eosinophils generated from the bone marrow in healthy individuals are low, resulting in relatively few cells circulating systemically. Eosinophils that are produced in the healthy bone marrow predominantly home to the gut mucosa, where they may be involved in maintenance of homeostasis with gut microbiota (2). The numbers of blood and tissue eosinophils are markedly altered in a range of specific inflammatory and allergic responses, where eosinophils may be found in high densities in mucosal tissues. The recruitment of eosinophils is thought to be orchestrated by a complex series of events involving antigen-presenting cells (APCs), mast cells, T cells, B cells, along with their released cytokines as immune signals.
Along with responding to immune signals, eosinophils themselves are a source of over 35 cytokines, chemokines, and growth factors (3, 4). These have profound effects on the progression of immune and inflammatory responses (Figure 1). The purpose of this review is to evaluate the role that eosinophil-derived cytokines, chemokines, and growth factors, and how these may contribute to the propagation of immune responses.
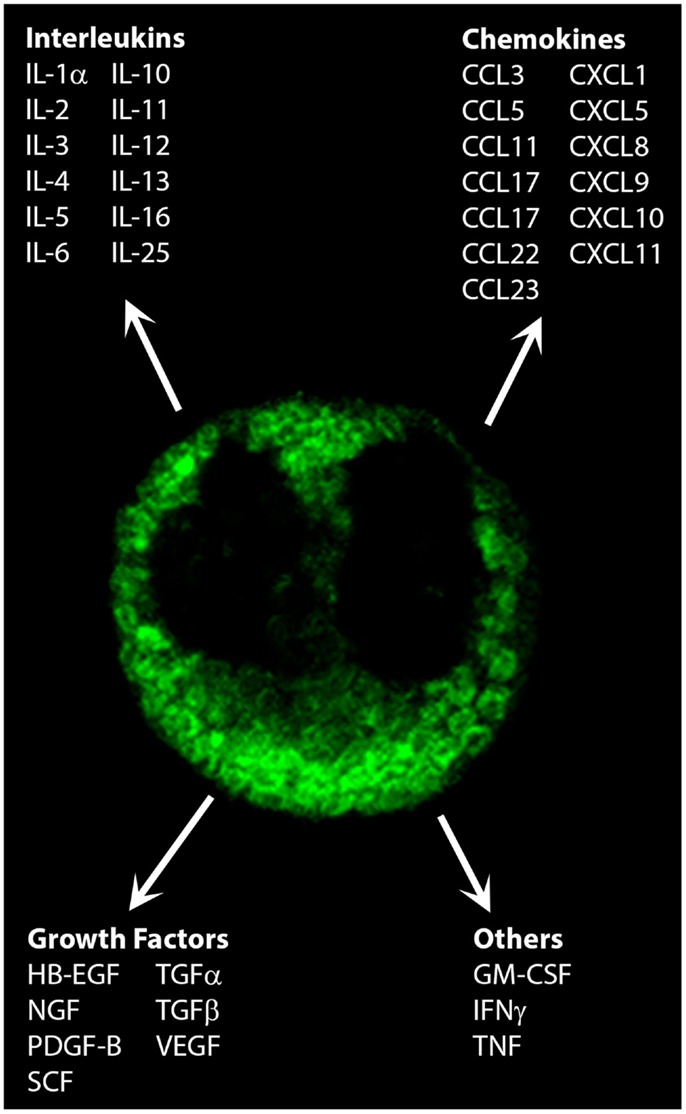
Figure 1. Cytokines, chemokines, and growth factors secreted by eosinophils. Shown is the immunofluorescent staining pattern obtained for CCL5/RANTES in a human peripheral blood eosinophil by confocal microscopy. Original magnification × 100.
Cytokines, Chemokines, and Growth Factors in Allergic Inflammation and Asthma
The role of eosinophils in immunity remains enigmatic. These granulated white blood cells are found, to varying degrees of similarity, in a wide range of invertebrates as well as vertebrates, including crustaceans, insects, mammals, fish, and birds (5, 6). Their expression in this wide variety of species suggests an important and evolutionarily conserved role in immunity. But what this role is, precisely, is still under intense scrutiny. While eosinophils have traditionally been implicated in maintenance of immunity against helminthic parasites, recent studies in transgenic mice that lack eosinophils suggest a more complex role for these cells than previously appreciated. In some cases, the absence of eosinophils actually inhibited parasitic growth (6), in contrast to the prevailing notion that eosinophils may be protective against helminthic parasites. More recent studies indicate that eosinophils may have a greater role in protection against viral infections, particularly respiratory viruses (7). Thus, the understanding of the role of eosinophils in immunity remains a fascinating and evolving area of research.
From a clinical perspective, eosinophils are widely known for their tendency to increase markedly during allergic inflammation in tissues that normally harbor very few eosinophils, such as the lungs and upper airways. A key feature of allergic inflammation and asthma is the acute or chronic inflammatory cell infiltration at sites of allergen exposure in atopic subjects. Eosinophils co-migrate with inflammatory cells and are thought to contribute to bronchoconstriction, mucus secretion, edema, and tissue injury in the airways. The inflammatory processes that underlie allergic responses are orchestrated by an elaborate network of cytokines and chemokines that regulate IgE responses, bone marrow progenitor cell differentiation, and adhesion molecule expression. Infiltration of inflammatory cells can further exacerbate inflammation in target tissues by further secretion of cytokines, chemokines, and growth factors from tissue-migrated cells. The allergic response is often manifested as a biphasic reaction in asthma, consisting of an early phase response that involves APC-mediated activation of T cells to a Th2 phenotype and mast cell degranulation, followed by the late-phase response in which a secondary infiltration of inflammatory cells occurs in affected sites. The role of eosinophils in the biphasic allergic response is thought to be mainly associated with the late-phase response. Eosinophils recruited to inflammatory sites frequently undergo degranulation, releasing a range of cationic cytotoxic molecules, including major basic protein (MBP) and eosinophil peroxidase (EPX), as well as producing numerous cytokines, chemokines, and growth factors.
Several regulatory cytokines have been defined as belonging to two classes of CD4+ T cells, which are involved in the initiation and maintenance of the allergic response. The first group of cytokines are those produced by T helper 1 (Th1) cells, which include interferon-γ (IFNγ), interleukin-2 (IL-2), and IL-12. The second group of cytokines are generated by Th2 cells, such as IL-4, IL-5, IL-9, and IL-13. More recent studies have suggested that Th17 and Treg cells are also important in the modulation of allergic responses by their production of immunosuppressive or regulatory cytokines including IL-10 and IL-17 (8). More recent studies have suggested an important role for IL-25 and IL-33 in the initiation of allergic responses, as these cytokines show a significant association with asthma in large cohort genome-wide association studies (9). In particular, IL-33 is important in the rapid induction of airway smooth muscle contraction by stimulating expansion of IL-13-producing type 2 innate lymphoid cells (10).
A substantial body of evidence from human and animal studies supports the hypothesis that allergic inflammation is an inappropriate response that arises from polarization of T cells toward a Th2 response, since greater expression of Th2 cytokines is seen in allergen-challenged individuals, along with a downregulation of Th1 cytokines. Enhanced expression of Th2 cytokines leads to the promotion of IgE switching of B cells, prevention of Th1 cytokine expression, increased tissue eosinophilia and eosinophil degranulation, and enhanced eosinophil survival. Prolongation of eosinophil survival, associated with delayed apoptosis, is thought to increase the amount of time that eosinophils actively release toxic mediators into tissues.
Against this background of Th2 responses in allergic inflammation, the cytokine network associated with allergy and asthma in humans is complex and not always associated with specific asthma phenotypes. Several studies suggest that although IL-4 triggers the polarization of T cells to a Th2 phenotype, it is not necessary for the manifestation of asthma (1). Moreover, the Th1 cytokine, IFNγ, may play a role in exacerbation of existing allergic inflammation as it is a potent activator of eosinophils in vitro (11–16). IFNγ has been found at elevated levels in the sera of patients with adult acute severe asthma (17, 18), and IFNγ+ cells become upregulated in correlation with eosinophil infiltration in allergic subjects (19, 20). Th1 and Th17 cytokines are associated with activation of innate immune cells in the recently characterized phenotype of non-Th2 asthma, which is a late-onset form of asthma that is seen in women, obese patients, smoking-associated asthma, and paucigranulocytic patients (1). Recent findings indicate that thymic stromal lymphopoietin (TSLP) may be a key target in airway hyperresponsiveness in allergic asthmatics (21). These observations suggest that Th2 cytokine responses alone are insufficient to promote asthmatic responses in the airways of human subjects.
However, the majority of asthma cases, although certainly not all, fit into the Th2 cytokine profile with varying degrees of eosinophilia (1). While the proportion of asthmatics exhibiting high numbers of eosinophils is not known, several studies of patients with mild to severe asthma suggest that it may be around 50% (1). Thus, eosinophils may be an important contributor to inflammatory responses at least half of asthma cases.
In summary, the substantial cytokine network underlying allergic inflammation is complex, with a Th2 cytokine profile and eosinophilia associating with some, but not all, asthma phenotypes. The way that eosinophil-derived cytokines contribute to immune defense or allergic diseases is not fully understood, although interestingly, recent discoveries have elucidated several novel functions for these cytokines in immunity and metabolism.
Eosinophils and Their Degranulation Responses
Eosinophils contain unique secretory granules known as crystalloid granules. These are so-called because of their characteristic crystalline cores, which appear electron-dense upon imaging by transmission electron microscopy. The crystalline core consists of highly concentrated, crystallized MBP, a cationic protein, which has cytotoxic effects on tissues upon its release (22). In addition to the MBP-rich crystalline core, crystalloid granules contain a matrix that is enriched in at least three other cationic proteins, which are EPX, eosinophil cationic protein (ECP), and eosinophil-derived neurotoxin (EDN). The liquid phase of the matrix also contains many other enzymes and proteins, including cytokines, chemokines, and growth factors (Figure 2).
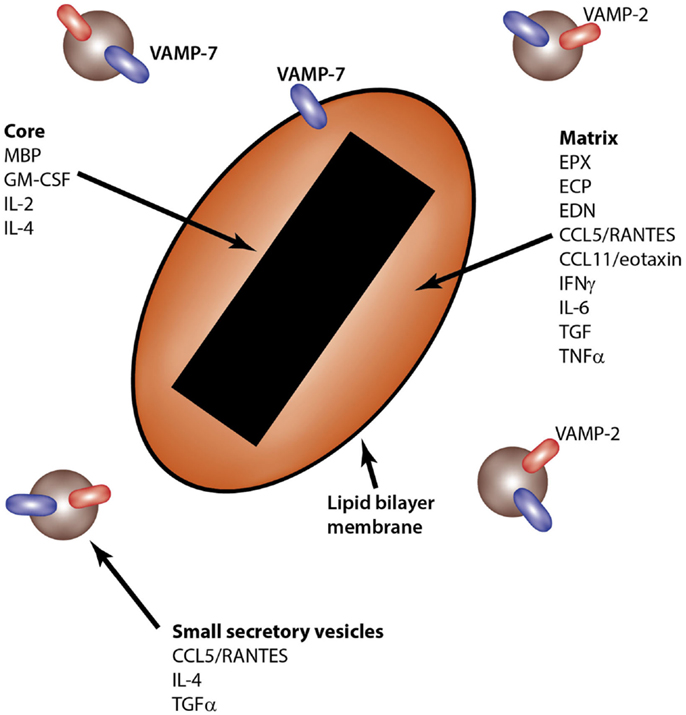
Figure 2. Intragranular sites of storage for eosinophil-derived cytokines. The eosinophil crystalloid granule consists of two internal compartments: the core, enriched in MBP, and the matrix, which contains EPX, ECP, and EDN, among other granule components. Small secretory vesicles also transport cytokines, including CCL5/RANTES, IL-4, and TGFα. SNARE molecules are shown in the lipid bilayer membranes of granules and secretory vesicles.
The contents of the crystalloid granule in eosinophils are released to the outside of the cell by at least four distinct mechanisms. These are (1) classical exocytosis (23); (2) compound exocytosis (24); (3) piecemeal degranulation (25), which is a form of exocytosis involving the fusion of small, rapidly mobilized secretory vesicles with the cell membrane; and (4) necrotic disintegration of the cell or “cytolysis,” where whole, intact granules are released upon cell membrane rupture (26, 27). Piecemeal degranulation and cytolysis are most commonly observed in tissues obtained from patients with allergic inflammation (28, 29). Tissue damage associated with eosinophilic asthma and allergic inflammation is thought to be related to excessive release and tissue deposition of eosinophil granule proteins, particularly MBP, EPX, and ECP (22).
Several physiological agonists induce the release of eosinophil granule proteins by exocytosis, including platelet-activating factor [PAF; (30, 31)], opsonized surfaces (32), complement factors [C5a, (33)], immunoglobulin complexes (34), and cytokines and chemokines including granulocyte/macrophage colony-stimulating factor (GM-CSF), IFNγ, IL-3, IL-5, and CCL11/eotaxin (16, 35–37). Many of these factors are present in allergic inflammation and would be expected to contribute to activation of eosinophil degranulation responses.
Human Eosinophils as a Source of Cytokines, Chemokines, and Growth Factors in Blood and Tissues
Over 35 cytokines, chemokines, and growth factors have been characterized in eosinophils (Table 1). In the majority of cases, messenger RNA and protein for each product has been identified. Evidence for the synthesis and expression of nearly all eosinophil-derived cytokines, chemokines, and growth factors has been obtained from peripheral blood eosinophils purified from non-atopic as well as atopic subjects. A number of these have been found as stored, pre-formed mediators in crystalloid granules, giving eosinophils the ability to release these potent immunoregulatory factors rapidly (<1 h) into the surrounding milieu in response to activation.
In confirmation of observations with peripheral blood eosinophils, tissue eosinophils have also been characterized for their ability to synthesize and secrete cytokines, chemokines, and growth factors. Studies of tissue eosinophils from nasal polyps, bronchial biopsies, bronchoalveolar lavage (BAL) fluid, sputum samples, celiac mucosal biopsies, and skin biopsies from atopic individuals have shown that these cells are also capable of elaborating these immunomodulatory factors. For example, a significant percentage of eosinophils from subjects with allergic rhinitis express GM-CSF (41), IL-4 (50), IL-5 (108), CCL3/macrophage inflammatory protein-1α (MIP-1α) (71), CCL5/regulated on activation, normal T cell expressed and secreted (RANTES) (109, 110), and transforming growth factor-β1 (TGFβ1) (98). As many as 44% of eosinophils present in nasal polyp tissues have been shown to be positive for IL-4 (50). Moreover, the majority of tissue-infiltrating eosinophils (84%) were found to be IL-4+ during allergen-induced cutaneous late-phase reactions at 6 h (50). In another study, around 20% of tissue eosinophils were positive for IL-4 and IL-5 mRNA in skin biopsies of allergic individuals 24 h following challenge, which increased to 50–60% for protein expression of IL-4 and IL-5 (111). Eosinophils have also been shown to express IL-4 and IL-5 mRNA and protein in bronchial biopsies of atopic asthmatics as well as normal non-atopic subjects (112).
Other cytokines and chemokines have been shown to increase in tissue eosinophils during allergic inflammation. Eosinophils have been shown to exhibit greater expression of TGFβ1 than those of normal control subjects in bronchial biopsy tissue sections (101, 102). Nasal mucosal biopsies from seasonal rhinitis patients were found to contain elevated CCL5/RANTES+ eosinophils, making up around 15% of the total CCL5/RANTES+ population of cells (109). Eosinophils found in late-phase cutaneous reactions following allergen challenge in atopic subjects also express increased CCL5/RANTES mRNA and protein (75).
Endobronchial or segmental challenge with allergen consistently results in elevated eosinophil numbers in BAL fluid samples. Eosinophils accumulating in the airways following allergen challenge have been shown to express GM-CSF and IL-5 (42), as well as CXCL8/IL-8 (87), and CCL11/eotaxin (76). BAL-derived eosinophils that were recruited to the airways upon allergen challenge secrete significantly increased levels of CXCL8/IL-8 compared with those of normal controls during in vitro incubation (87). Supporting these observations is the study showing that sputum eosinophils also express GM-CSF as determined by immunocytochemistry (113).
The discovery of expression of cytokines by tissue eosinophils is not restricted to those found in the skin and airways. Eosinophils in the gut mucosa have also been found to express cytokines. In patients with active celiac disease, eosinophils from the gut mucosa were shown to be positive for IL-5 mRNA, and following treatment with a gluten-free diet, the numbers of IL-5+ eosinophils declined (54). However, IL-5+ eosinophils have not been detected in all gastrointestinal disorders, as intestinal mucosal eosinophils in Crohn’s disease do not appear to be positive for IL-5 (56).
Other diseases also exhibit cytokine expression by eosinophils. IL-5+ eosinophils have been detected in blood and tissue samples of individuals with eosinophilic cystitis, hypereosinophilic syndrome, and eosinophilic heart disease (55, 56).
Taken together, these and other ex vivo studies in humans have demonstrated that eosinophils derived from both the peripheral blood and tissue sources are capable of synthesizing, and in some cases releasing, cytokines, chemokines, and growth factors in eosinophilic diseases.
Storage and Secretion of Eosinophil-Derived Cytokines, Chemokines, and Growth Factors
As many as 10 cytokines, chemokines, and growth factors have been identified as pre-formed mediators that are stored within the crystalloid granules of eosinophils. Those found within granules are CCL5/RANTES (16), CCL11/eotaxin (77), GM-CSF (44), IL-2 (48), IL-4 (51, 52), IL-5 (56, 57), IL-6 (15), IL-13 (4, 63), transforming growth factor-α (TGFα) (114), and tumor necrosis factor-α (TNF) (70). The most abundant cytokine in eosinophils appears to be IL-13, followed by IFNγ and TNF (4). The techniques used to determine intracellular sites of cytokine storage include immunocytochemistry, subcellular fractionation, immunogold labeling, and immunofluorescence using confocal microscopy analysis.
Most cytokines that have been identified in eosinophil crystalloid granules appear to be stored within the matrix compartment surrounding the crystalline core, although a few may colocalized with the MBP-containing core. These are GM-CSF (44), IL-2 (48), and IL-4 (51). Some cytokines have not had their precise intragranular location determined, such as IL-5. These fascinating observations indicate that eosinophils are capable of storing pre-formed cytokines that may be released rapidly in response to inflammatory events.
Eosinophils appear to use a specialized tubulovesicular system to transport some of these cytokines and chemokines from the crystalloid granule to the cell membrane. This membrane transport mechanism, also known as piecemeal degranulation, allows the shuttling of granule contents to the cell surface through rapidly mobilizable secretory vesicles that bud from the surface of the crystalloid granule (115–118). This mechanism of cytokine transport was first identified with CCL5/RANTES, in which at least two intracellular compartments store pre-formed cytokine. The first is the crystalloid granule, and the second is a pool of small secretory vesicles that sediment at a higher buoyant density than granules when analyzed by subcellular fractionation (16).
Other studies have demonstrated that these small secretory vesicles are important in cytokine trafficking, particularly for TGFα (114) and more recently, IL-4 (119–122). The membrane trafficking mechanisms associated with piecemeal degranulation of cytokines and chemokines are described in more detail elsewhere in this issue (123).
Small secretory vesicles increase in numbers as well as in their content of cytokines and chemokines upon stimulation of eosinophils by immunoregulatory cytokines. For example, stimulation of peripheral blood eosinophils in vitro by IFNγ induces intracellular mobilization of IL-6 and CCL5/RANTES prior to their release (15, 16). IFNγ specifically induces the redistribution of CCL5/RANTES within eosinophils from crystalloid granules to small secretory vesicles within 10 min of stimulation (16). Intriguingly, the pool of CCL5/RANTES-containing small secretory vesicles was mobilized to the cell periphery within minutes of stimulation, leaving MBP+ crystalloid granules behind in the cytoplasmic regions of cells, while crystalloid granule-associated CCL5/RANTES followed afterward. These findings suggest that granule proteins and cytokines/chemokines are selectively and differentially released in line with specific types of inflammatory responses in eosinophils.
Supernatants retrieved from IFNγ-stimulated eosinophils contained increased CCL5/RANTES and other crystalloid granule products (EPX), confirming the occurrence of degranulation (16). Following 16 h of stimulation by IFNγ, eosinophils were able to replenish their stores of CCL5/RANTES in their crystalloid granules. These findings have implications for sustained release of eosinophil-derived cytokines and chemokines in immune responses. An interesting possibility is that the eosinophil may have the potential to generate fine-tuned responses to immunological stimuli, with the release of cytokines/chemokines occurring separately from granule proteins.
CCL5/RANTES+ small secretory vesicles were subsequently found to colocalize with the soluble N-ethylmaleimide sensitive factor attachment protein receptor (SNARE), VAMP-2 (vesicle-associated membrane protein-2) (124). SNARE proteins are universal fusion proteins that regulate the attachment (docking) of lipid bilayer-surrounded granules or vesicles to target membranes such as the inner leaflet of the plasma membrane (125). The fusion of CCL5/RANTES+ vesicles upon docking with the inner leaflet of the plasma membrane is hypothesized to be dependent on binding to cognate target membrane-bound SNAREs, SNAP-23 and syntaxin-4 (117, 118, 126). Eosinophils depend on VAMP-7 for the release of granule proteins in response to intracellular secretagogues, GTPγS, and Ca2+ (127). However, we were unable to demonstrate whether CCL5/RANTES release was dependent on VAMP-7 as well, since negligible CCL5/RANTES was detected in supernatants of permeabilized eosinophils stimulated with these secretagogues. This suggests that the permeabilization process (using streptolysin-O) and/or the secretagogues used (GTPγS and Ca2+) may not be optimal for inducing piecemeal degranulation, leading to cytokine secretion, in eosinophils.
Our recent findings indicate that eosinophil secretion may be evoked by the addition of PAF, a potent secretagogue for both human and mouse eosinophils (31). Degranulation was assessed by an EPX ELISA that was optimized in-house (128). Previously, mouse eosinophils were considered to be poor degranulators (129–132). Using these novel parameters for the assessment of mouse eosinophil degranulation, we determined that the guanosine triphosphatases (GTPases) Rac2 and Rab27a contribute to the secretion of EPX from eosinophils (133, 134). Rac2 regulates the assembly of the actin cytoskeleton network, which is essential for granule movement through the cytoplasm, while Rab27a acts through a family of Sec/Munc proteins that regulate SNARE binding (135, 136). The role of GTPases in regulating cytokine secretion from eosinophils has not yet been defined.
Taken together, our studies suggest that cytokine trafficking and release in eosinophils may be mediated by VAMP-2 binding through its cognate SNAREs, SNAP-23, and syntaxin-4. Additional studies are required to understand how VAMP-2-mediated cytokine secretion may be regulated by GTPases.
Potential Immunological Roles for Eosinophil-Derived Cytokines, Chemokines, and Growth Factors
The ability of eosinophils to synthesize and secrete a large number of cytokines, chemokines, and growth factors suggest that these cells have the potential to regulate numerous immune responses, including allergic inflammation. The diversity of immunomodulators produced by eosinophils suggests that they may be able to orchestrate inflammatory processes in an exacerbating or modulatory manner. Many of the factors elaborated by eosinophils are likely to regulate immune responses, particularly CCL3/MIP-1α, CCL5/RANTES, CCL11/eotaxin, CXCL8/IL-8, GM-CSF, IL-1α, IL-2, IL-3, IL-4, IL-5, IL-6, IL-9, IL-10, IL-13, TNF, and various growth factors. Other eosinophil-derived cytokines, chemokines, and growth factors, shown in Table 1, are likely involved in other types of reactions that apply to unique situations, such as atopic dermatitis, which may involve IL-12 released from tissue eosinophils following an initial phase of a Th2 response during cutaneous allergen challenge.
Eosinophil-derived chemokines may support the recruitment and maintenance of tissue eosinophils and lymphocyte infiltration during allergic inflammation. Eosinophils generate numerous chemokines including CCL3/MIP-1α and CCL5/RANTES (16, 71, 74, 75), both of which are major regulators of local inflammatory responses and chemoattractants for circulating leukocytes (137, 138). CCL5/RANTES exerts direct effects on eosinophils by elevating intracellular Ca2+, triggering degranulation, and promoting superoxide release concurrently with enhanced chemotaxis (139–141).
CCL11/eotaxin is an important eosinophil-specific chemokine that is involved in chemotaxis of eosinophils into tissues, and is a highly potent agonist for inducing an influx of eosinophils during allergic responses (142). Gene knockout of CCL11/eotaxin markedly diminishes the tissue presence of eosinophils, which subsequently decreases allergic inflammation in the gut, skin, and airways (143). Like CCL5/RANTES, CCL11/eotaxin activates intracellular Ca2+ mobilization, degranulation, and respiratory burst in eosinophils, suggesting that it may act in an autocrine manner (144–146). Eosinophils express CCL11/eotaxin constitutively, apparently in association with their granules (77). This suggests a potential role for eosinophil-derived CCL11/eotaxin to function in a paracrine/autocrine manner for further recruitment of eosinophils at sites of inflammation.
Eosinophils release the neutrophil chemokine CXCL8/IL-8 (84), suggesting a role for eosinophils in recruitment of neutrophils at sites of inflammation. This chemokine is highly chemotactic and stimulatory for neutrophils and T cells (147), and eosinophils also have chemotactic responses to CXCL8/IL-8 following incubation with GM-CSF or IL-3 (148). However, IL-4 and IL-5 production in allergic inflammation may downregulate CXCL8/IL-8 expression, since these cytokines inhibit CXCL8/IL-8 release from monocytes (149). The overall role of CXCL8/IL-8 derived from eosinophils in inflammatory conditions is yet to be determined.
Eosinophils have been shown to synthesize and release abundant GM-CSF, which promotes degranulation and mediator release from these cells (37, 40, 44, 148). The activation of eosinophils by ionomycin, a calcium ionophore, induces GM-CSF release, which prolongs their own survival in vitro (40). Thus, GM-CSF is likely a critical eosinophil-derived cytokine that is important in maintaining the viability and effector function of eosinophils at inflammatory foci in allergic and immune responses.
The production of IL-1α from human eosinophils has been associated with cytokine induction of human leukocyte antigen DR (HLA-DR) expression (45). This suggests that the eosinophil has the capacity to function as an APC, as demonstrated in mouse models of allergic inflammation (150–153). Eosinophils have also been demonstrated to function as APCs during Strongyloides stercoralis infection (154). Secretion of IL-1α from eosinophils may be an important factor in allergen presentation to T cells in subjects with established eosinophilia during Th2-deviated immune responses to specific allergens.
A role in allergic inflammation is also implicated for eosinophil-derived IL-2, an essential growth factor for T cells that is likely to be critical for the initiation of the allergic phenotype following an early phase involving IL-4 stimulation (155). IL-2 also induces eosinophil chemotaxis via its receptor (CD25) expressed on a proportion of them (156).
IL-3 is a pluripotent growth factor required for the generation of a wide range of myelocytic cells and granulocytes, and is an important autocrine factor produced and used by eosinophils when stimulated (148, 157). Thus, tissue eosinophils that are actively secreting IL-3 are likely to prolong their own survival by autocrine signaling.
The role of IL-4 in allergic inflammation has been extensively studied and is among the most clearly defined of all known cytokines, with biologics development targeting the function of this cytokine (158). This cytokine is firstly important in the maintenance of Th2 responses (159, 160), and secondly, it is a critical factor in initiating the switch of B cells to IgE isotype production (161). IL-4 also has many other stimulatory roles in allergic inflammation, by inducing chemotaxis in eosinophils and enhancing the capacity of eosinophils to release granule proteins (35, 162). Eosinophil-derived IL-4 may be important in promotion of inflammation by increasing local IgE production, as well as upregulating vascular cell adhesion molecule (VCAM) expression on endothelial cells, which would increase leukocyte adhesion and transmigration into affected tissues (163). This would, in turn, increase eosinophil-specific migration into tissues by expression of very late antigen 4 (VLA-4) ligand (164).
Similarly to IL-4, IL-5 is also extensively investigated and well-defined for its role in allergic inflammation. While IL-5 is not necessary for skewing immune responses toward a Th2 phenotype, it is important in the downstream events that are typical of Th2 responses to allergens. IL-5 is essential for the terminal differentiation of eosinophils from CD34+ progenitors present in the bone marrow (165, 166). It also has numerous effects on eosinophils including prolongation of survival, induction of chemotaxis, priming, and degranulation so that their responses to agonists are enhanced (35, 167, 168). Finally, IL-5 prolongs the survival of eosinophils in vitro (169). These observations suggest that eosinophil-derived IL-5 is involved in exacerbations of the local allergic response following its release.
An additional cytokine derived from eosinophils that is likely to contribute to allergic responses is IL-6. This pleiotropic cytokine, generated during acute phase reactions, is important in regulating T and B cell function, as well as priming of granulocytes. IL-6 is an essential cofactor with IL-4 in isotype switching of B cells toward IgE production (170). In asthmatics, IL-6 is elevated in the serum and BAL in both baseline conditions and following allergen challenge along with IL-4 and IL-5 (171, 172). Whether IL-6 expressed from eosinophils is important in allergic inflammation is not known.
Eosinophils may also augment Th2 responses by secretion of IL-9, a potent T cell, and mast cell growth factor (173, 174). The expression of IL-9 mRNA and protein products were demonstrated in eosinophils along with mRNA encoding the IL-9 receptor α subunit, suggesting an autocrine role for this cytokine in eosinophils.
Eosinophils have the capacity to express and release the immunosuppressive cytokine IL-10 (53). A role for eosinophil-derived IL-10 is likely to enhance allergic inflammation, since this cytokine acts in concert with IL-4 to mediate the growth, differentiation, and isotype switching of activated B cells (175). However, IL-10 is classically known for its immunosuppressive role in immunity by decreasing cytokine secretion from inflammatory cells and preventing allergic inflammation (176, 177). A recent study showed that eosinophil-derived IL-10 has a novel immunoregulatory role for eosinophils in helminth infection (60). IL-10 generated from eosinophils induced the proliferation of myeloid dendritic cells and CD4+ T lymphocytes, which inhibited expression of inducible nitric oxide synthase (iNOS), and protected intracellular Trichinella spiralis larvae. This striking observation suggests a protective role for eosinophils for intracellular T. spiralis larvae against NO-mediated killing, and further, that IL-10 derived from eosinophils drives this protective response. Further, it appears that T. spiralis exploits eosinophils to maintain its long-term survival in muscle tissue. These findings indicate a significant functional diversity of eosinophils that has not previously been appreciated until the advent of transgenic eosinophil-deficient mouse strains.
Among the more important cytokines released by eosinophils is IL-13, which is also stored in crystalloid granules as a pre-formed mediator (4, 63). IL-13 has many roles in the establishment of airway disease in asthma as well as pulmonary fibrosis, and also activates matrix metalloproteases in the airways (178). The activation of matrix metalloproteases by IL-13 is thought to protect against excessive allergic inflammation. IL-13 is also able to induce isotype switching of B cells to produce IgE (179), and has an important role in allergic inflammation (180). The expulsion of helminthic parasites from the gut of mice is also dependent on IL-13 (181). Eosinophils express IL-13 in inflammatory diseases (64), and this may have a role in the development of allergic inflammation, as discussed below.
Growth factors have an important role in inflammatory conditions, and those derived from eosinophils are likely to promote an inflammatory phenotype. Among these are heparin-binding epidermal growth factor (HB-EGF), which is a potent smooth muscle cell mitogen, and may contribute to pulmonary hypertension (89). Nerve growth factor (NGF) is elevated in subjects with allergic asthma, allergic rhinitis, and allergic urticarial–angioedema, with the largest increases observed in asthma (182). Eosinophils express NGF mRNA and protein, and this may contribute to the elevated levels seen in allergic inflammation (90). The most likely source of elevated serum NGF in allergy is from IgE-stimulated mast cells in tissues, since mast cells synthesize and secrete NGF (183). NGF stimulates T cells, B cell proliferation and differentiation, and eosinophil differentiation from peripheral progenitors (184), and is thus implicated in the pathogenesis of allergic inflammation.
Eosinophils also express the mast cell cytokine stem cell factor (SCF) (92). SCF may be associated with a positive feedback loop in tissue mast cells to maintain or exacerbate allergic inflammation, as well as inducing the growth and differentiation of mast cell progenitors residing in tissues (185).
Other growth factors produced by eosinophils include TGFα and TGFβ, which may modulate wound-healing and tissue remodeling (93, 102). TGFβ is specifically recognized for its role in chronic inflammation and fibrosis, and eosinophil-derived TGFβ may exert a role in tissue repair by inducing fibroblast growth and differentiation into myofibroblasts (186). Thus, TGFβ released from eosinophils may have a role in extracellular matrix protein deposition, particularly collagen, which contributes to structural abnormalities observed in severe allergic inflammation, including stromal fibrosis and basement membrane thickening.
Purified peripheral blood eosinophils from atopic individuals have been demonstrated to spontaneously release the pro-inflammatory cytokine TNF upon culture, and normal eosinophils stimulated with immobilized immunoglobulins or TNF express mRNA for this cytokine (53). TNF is a highly potent activator of monocytes, T cells, neutrophils, and endothelial cells, and enhances eosinophil adhesion and cytotoxicity (187, 188). This particular cytokine has numerous roles in inflammatory conditions, as well as helminthic infection and neoplasia associated with eosinophilic infiltration.
The Th1 cytokines, IL-12 and IFNγ, are also expressed in eosinophils. These are typically associated with inflammatory conditions that are distinct from the Th2 profile in allergic inflammation, and usually downregulate allergic inflammation following their release (189). However, Th2 cytokines can paradoxically promote Th1 responses in immune cells. IL-12 produced from eosinophils treated with IL-4 has been demonstrated to promote IFNγ mRNA expression in human Th1 cells (62). Allergen patch test reactions in atopic patients have shown that a Th1-like T cell activation occurs following the initial phase of increased local expression of Th2 cytokines associated with eosinophil infiltration after allergen challenge (190). Eosinophil-derived IL-12 was proposed to induce a switch from Th2 to Th1 responses commonly seen in late-phase allergic skin reactions (191). Eosinophils also express IFNγ in normal and atopic individuals (4, 69). Stimulation of eosinophils by IFNγ has been postulated to enhance allergic inflammation during viral infections, suggesting an autocrine role for this cytokine (192).
Taken together, these findings show that eosinophils have the capacity to generate numerous immunoregulatory cytokines, chemokines, and growth factors. However, while the discovery that eosinophils can synthesize and secrete these immunomodulatory factors is important, it is essential to determine whether eosinophil-derived cytokines, chemokines, and growth factors have bioactive roles in immunity. In this way, we may learn more about the specific immunological function of eosinophils in the regulation of inflammatory processes.
Do Eosinophil-Derived Cytokines, Chemokines, and Growth Factors have Bioactive Roles in the Immune System?
Although the studies described above have presented evidence for the expression and release of a plethora of cytokines, chemokines, and growth factors from eosinophils, around a third of these have been shown to have a direct bioactive role. These include a proliferation-inducing ligand (APRIL), CCL5/RANTES, GM-CSF, IL-1β, IL-4, IL-10, IL-12, IL-13, IL-16, TNF, and TGFβ, which have been shown to have direct bioactive effects on other cells or in mouse models.
Recent studies indicate that eosinophils express APRIL, along with IL-6, which promoted the survival of plasma cells in the bone marrow in mice (38). This important finding suggests that eosinophils may have a role in enhancing and maintaining immunoglobulin production from plasma cells, which was shown to be true in a later study where eosinophils increased IgA+ plasma cell numbers and the secretion of IgA in the gut mucosa (193). These studies suggest that eosinophils are essential for maintaining the integrity of the gut mucosal immunity, which is in agreement with their usual tissue homing under homeostatic conditions.
Other studies have shown that eosinophil-derived CCL5/RANTES and IL-16 have direct effects on lymphocytes in culture by inducing chemotactic activities (62). Thus, human eosinophils have the ability to alter the function of CD4+ lymphocytes and memory T cells (194, 195). Finally, eosinophil-derived CCL5/RANTES has been shown to exert bioactive effects in eosinophil chemotactic assays as determined by the inhibitory effects of antibody to CCL5/RANTES, suggesting a role for this chemokine in autocrine signaling to enhance eosinophil migration.
The prolongation of eosinophil survival by GM-CSF is sensitive to inhibition by cyclosporin A, suggesting that this drug may modulate allergic inflammation by preventing autocrine cytokine signaling in eosinophils (40). Thus, eosinophil-derived GM-CSF is likely to have biological effects on survival of tissue eosinophils as well as newly recruited cells.
A recent study has demonstrated a role for eosinophil-derived IL-1β in inducing synthesis and secretion of IL-17 in activated CD4+ T cells (47). As Th17 cells are implicated in the pathogenesis of allergic airway inflammation (196), this may be a key mechanism by which eosinophils promote Th17 cell differentiation.
Perhaps the most well studied cytokine elaborated by eosinophils is IL-4. Eosinophils are among the most abundant IL-4-expressing non-T, non-B cell populations in the lung and spleen of mice infected with the helminthic parasite Nippostrongylus brasiliensis (197). In the IL-4 reporter 4get mice, eosinophils were the most prevalent IL-4-producing cells infiltrating the lungs of mice infected with N. brasiliensis, with an up to 1000-fold increase (198). IL-4 expression in eosinophils is constitutive and programmed at an early stage of ontogeny (199). Moreover, instillation of IL-4 led to expansion of IL-4-producing eosinophils in vivo, suggesting that IL-4 is a potent factor in promoting the differentiation of bone marrow progenitor cells into Th2 cytokine-producing eosinophils, using mice expressing IL-4 with GFP (4get) (200). The production of IL-4 by eosinophils also occurs in other infections; when mice are infected with the fungal Cryptococcus neoformans, the majority of cells expressing IL-4 in the airways are eosinophils (201).
Adjuvant stimulation of B cell responses has been linked to IL-4 production from eosinophils in the spleen, based on findings in Δdbl-GATA eosinophil-deficient mice that were administered with alum (153). When alum was injected intraperitoneally into eosinophil-deficient animals, early B cell priming and IgM production was attenuated. This suggests a pivotal and previously unrecognized role for eosinophils in modulating the adaptive immune response to vaccines.
Interestingly, recent findings have indicated that eosinophil-derived IL-4 is also essential for the biogenesis of beige fat, a type of brown adipose tissue that is found in abundance in newborns that promotes non-shivering thermogenesis (202). Eosinophil-derived IL-4 was demonstrated to switch monocytes to the alternatively activated macrophage phenotype (203, 204), leading to the conversion of beige fat precursor cells to beige adipocytes (205). In a separate study, the satiety hormone leptin was found to be a potent secretagogue for eosinophils, and induced the expression and secretion of IL-1β, IL-6, and CXCL8/IL-8, as well as other chemokines, from eosinophils (206). This observation suggests that eosinophils may have an important role in improving the metabolic phenotype, by promoting insulin responsiveness and decreasing the incidence of obesity.
Eosinophil-derived IL-4 may be important in liver regeneration by promoting hepatocyte proliferation (207). In this study, liver injury induced by partial hepatectomy or carbon tetrachloride (CCl4) in 4get mice resulted in the rapid recruitment of eosinophils, which secreted IL-4 and induced the proliferation of hepatocytes and liver growth.
The secretion of IL-4 from eosinophils in local tissues during allergic reactions may serve as a major initial source of IL-4 required for switching tissue-infiltrating naïve T cells to the Th2 phenotype. Several mouse models of Th2 inflammation support this notion. Mice infected intraperitoneally with eggs from the trematode Schistosoma mansoni, which generates a strong Th2 phenotype, exhibited early IL-4 increases derived from degranulating peritoneal eosinophils. Eosinophil-derived IL-4 induced priming of naïve T cells and activation of mast cell IL-5 release (154). The significance of IL-4 generation from eosinophils in human studies is yet to be discovered.
Stimulation of eosinophils with the Th2 cytokine IL-4 can also promote the development of Th1 responses. As described in Section “Potential Immunological Roles for Eosinophil-Derived Cytokines, Chemokines, and Growth Factors” above, IL-4-treated eosinophils release the Th1 cytokine IL-12, which in turn induced the expression of IFNγ from Th1 cells during the culture of T cells with eosinophil-conditioned media (62). The finding that eosinophils may release Th1 cytokines suggests a more nuanced control of cytokine responses in allergic inflammation that these cells may possess. In summary, IL-4 production from eosinophils may be important in a variety of immune and metabolic responses that are only just beginning to be understood. These observations will continue to shape our understanding of the biological role of eosinophils in immunity.
A fascinating recent study using a computational model demonstrated that eosinophil-derived IL-13 is required for allergic airway responses (208). This was determined by the use of IL-13−/− eosinophils that were adoptively transferred by intravenous injection into Δdbl-GATA mice, which exhibited low airway hyperresponsiveness. IL-13 is important in inducing many of the characteristics of allergic airway disease, including airway hyperresponsiveness, goblet cell hyperplasia, and mucus secretion (180). In the computational model, it was found that eosinophil-derived IL-13 could not sustain an allergic asthma response in the absence of T cell (or other cell type)-derived IL-13, but that IL-13 production by eosinophils was integral to the development of allergic asthma (208).
Co-culture of human eosinophils and their conditioned media with the colon carcinoma cell line Colo-205 led to the production of TNF, which was involved in tumor cell killing along with granzyme A (73). This suggests a novel function for eosinophil-derived TNF in regulating tumor cell growth, and that tumor cells may be directly recognized by eosinophils.
Eosinophil-derived TGFβ has been shown to regulate fibroblast proliferation and differentiation (209). This is indicative of a potential role for eosinophils in wound-healing. Indeed, eosinophils were found to express both TGFβ and IL-13 following intradermal allergen challenge, which resulted in increased repair and remodeling events in human atopic skin (210).
Other Physiological or Pathological Roles for Eosinophil-Derived Cytokines
Although earlier studies in mice suggested that eosinophil-derived IL-4 was required for mounting a Th2 response during immune reactions to intraperitoneally injected Schistosoma mansoni eggs (211), more recent studies have shown that ablation of eosinophils from mice had negligible outcomes, or even inhibitory effects, on helminth larval or egg expulsion in Δdbl-GATA or PHIL mice (212). These experimental findings imply that eosinophil-derived cytokines, chemokines, and growth factors have no specific role in the regulation of parasitic worm diseases, in contrast to the classically held notion that eosinophils are important in parasitic worm expulsion.
In other studies, certain types of lymphoid and solid tumors have been associated with the infiltration of eosinophils into cancerous tissues (213), particularly specific lymphomas and Hodgkin’s disease. Although tumor-related eosinophilia was considered to be an epiphenomenon arising from the spontaneous elaboration of IL-5 from tumor cells, or overproduction of T cells during chemotherapy with IL-2 (214), there is evidence of eosinophil activation as a result of IL-2 anticancer therapy (215, 216). In some cases, tissue eosinophilia is considered to be a positive prognosis for head and neck cancers (217) and advanced bladder cancer (218). Specifically, in oral squamous cancer, eosinophils are a positive prognosis for early stages of disease (Stages II and III), but an unfavorable prognosis for advanced cases (Stages III–IV) (219, 220). The involvement of eosinophil-derived cytokines, chemokines, and growth factors in neoplasias associated with eosinophilic infiltration is partially understood. In oral squamous cell carcinoma with tumor-associated tissue eosinophilia (TATE), the source of CCL11/eotaxin is apparently eosinophils (221). The cultured oral squamous cell carcinoma cell line SCC9 secretes chemotactic prostaglandin D2, which promotes eosinophil transmigration, thus providing a signal to recruit eosinophils into tumor masses (222). Eosinophils that infiltrate into tumors/lymphomas also express IL-6, TGFβ, and CCL24/eotaxin-2 (223).
However, while numerous studies have highlighted a role for eosinophil recruitment and activation in many types of cancers, and that eosinophils express receptors and mediators shared with cytotoxic T cells (224), there are few that implicate eosinophil-derived cytokines, chemokines, and growth factors in the regulation of cancer growth. Interestingly, as described above, a recent study showed that human eosinophils possessed tumoricidal activity toward a colon cancerous cell lines in culture by releasing TNF (73). The tumor-killing effects of eosinophil-derived TNF were the first description of a cytokine, chemokine, or growth factor elaborated by eosinophils implicating a role for these immunomodulators in cancer.
Injection of IL-17E increases the efficiency of chemotherapy and results in eosinophilia (225). Eosinophil-derived IL-17 may be implicated in antitumor activity, since IL-17E (IL-25) exerts antitumor activity against many types of cancerous cell lines, at least in xenograft models using human cancer cells in mice (226). Eosinophils express IL-17 as determined by immunocytochemistry and Western blot analysis (66). Thus, the production of IL-17 from eosinophils may be important in protection of the host against cancers.
Moreover, since eosinophils produce numerous growth factors such as vascular endothelial growth factor (VEGF), as well as a number of other factors that promote angiogenesis, it is possible that eosinophilic inflammation is implicated in tumor neovascularization (227). Hypothetically, eosinophil-derived cytokines, chemokines, and growth factors may be involved in enhancing T cell-mediated tumor killing, particularly at the level of the local tissue environment where large numbers of infiltrating eosinophils accumulate and are actively degranulating onto tumor cells.
Taken together, these findings suggest that eosinophils may serve as important components of natural immunity, and their cytokines, chemokines, and growth factors may contribute to augment inflammatory responses in allergy and other conditions. Further studies are awaited to understand the involvement of eosinophil-derived immunomodulatory factors in the regulation of allergic inflammation and other inflammatory conditions, as well as the extent to which the release of these factors may be manipulated for therapeutic benefit.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Wenzel SE. Asthma phenotypes: the evolution from clinical to molecular approaches. Nat Med (2012) 18:716–25. doi: 10.1038/nm.2678
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
2. Jung Y, Rothenberg ME. Roles and regulation of gastrointestinal eosinophils in immunity and disease. J Immunol (2014) 193:999–1005. doi:10.4049/jimmunol.1400413
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
4. Spencer LA, Szela CT, Perez SA, Kirchhoffer CL, Neves JS, Radke AL, et al. Human eosinophils constitutively express multiple Th1, Th2, and immunoregulatory cytokines that are secreted rapidly and differentially. J Leukoc Biol (2009) 85:117–23. doi:10.1189/jlb.0108058
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
5. Lee JJ, Jacobsen EA, Mcgarry MP, Schleimer RP, Lee NA. Eosinophils in health and disease: the LIAR hypothesis. Clin Exp Allergy (2010) 40:563–75. doi:10.1111/j.1365-2222.2010.03484.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
6. Rosenberg HF, Dyer KD, Foster PS. Eosinophils: changing perspectives in health and disease. Nat Rev Immunol (2013) 13:9–22. doi:10.1038/nri3341
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
7. Percopo CM, Dyer KD, Ochkur SI, Luo JL, Fischer ER, Lee JJ, et al. Activated mouse eosinophils protect against lethal respiratory virus infection. Blood (2013) 123:743–52. doi:10.1182/blood-2013-05-502443
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
8. Palomares O, Yaman G, Azkur AK, Akkoc T, Akdis M, Akdis CA. Role of Treg in immune regulation of allergic diseases. Eur J Immunol (2010) 40:1232–40. doi:10.1002/eji.200940045
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
9. Moffatt MF, Gut IG, Demenais F, Strachan DP, Bouzigon E, Heath S, et al. A large-scale, consortium-based genomewide association study of asthma. N Engl J Med (2010) 363:1211–21. doi:10.1056/NEJMoa0906312
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
10. Barlow JL, Peel S, Fox J, Panova V, Hardman CS, Camelo A, et al. IL-33 is more potent than IL-25 in provoking IL-13-producing nuocytes (type 2 innate lymphoid cells) and airway contraction. J Allergy Clin Immunol (2013) 132:933–41. doi:10.1016/j.jaci.2013.05.012
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
11. Valerius T, Repp R, Kalden JR, Platzer E. Effects of IFN on human eosinophils in comparison with other cytokines. A novel class of eosinophil activators with delayed onset of action. J Immunol (1990) 145:2950–8.
12. Hartnell A, Kay AB, Wardlaw AJ. IFN-gamma induces expression of Fc gamma RIII (CD16) on human eosinophils. J Immunol (1992) 148:1471–8.
13. Hartnell A, Robinson DS, Kay AB, Wardlaw AJ. CD69 is expressed by human eosinophils activated in vivo in asthma and in vitro by cytokines. Immunology (1993) 80:281–6.
14. Ishihara C, Ochiai K, Kagami M, Takashahi H, Matsuyama G, Yoshida S, et al. Human peripheral eosinophils express functional interferon-gamma receptors (IFN-gammaR). Clin Exp Immunol (1997) 110:524–9. doi:10.1046/j.1365-2249.1997.4511469.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
15. Lacy P, Levi-Schaffer F, Mahmudi-Azer S, Bablitz B, Hagen SC, Velazquez J, et al. Intracellular localization of interleukin-6 in eosinophils from atopic asthmatics and effects of interferon γ. Blood (1998) 91:2508–16.
16. Lacy P, Mahmudi-Azer S, Bablitz B, Hagen SC, Velazquez JR, Man SF, et al. Rapid mobilization of intracellularly stored RANTES in response to interferon-γ in human eosinophils. Blood (1999) 94:23–32.
17. Corrigan CJ, Kay AB. CD4 T-lymphocyte activation in acute severe asthma. Relationship to disease severity and atopic status. Am Rev Respir Dis (1990) 141:970–7. doi:10.1164/ajrccm/141.4_Pt_1
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
18. Ten Hacken NH, Oosterhoff Y, Kauffman HF, Guevarra L, Satoh T, Tollerud DJ, et al. Elevated serum interferon-gamma in atopic asthma correlates with increased airways responsiveness and circadian peak expiratory flow variation. Eur Respir J (1998) 11:312–6. doi:10.1183/09031936.98.11020312
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
19. Kleinjan A, Dijkstra MD, Boks SS, Severijnen LA, Mulder PG, Fokkens WJ. Increase in IL-8, IL-10, IL-13, and RANTES mRNA levels (in situ hybridization) in the nasal mucosa after nasal allergen provocation. J Allergy Clin Immunol (1999) 103:441–50. doi:10.1016/S0091-6749(99)70469-0
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
20. Cho SH, Stanciu LA, Holgate ST, Johnston SL. Increased interleukin-4, interleukin-5, and interferon-gamma in airway CD4+ and CD8+ T cells in atopic asthma. Am J Respir Crit Care Med (2005) 171:224–30. doi:10.1164/rccm.200310-1416OC
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
21. Gauvreau GM, O’byrne PM, Boulet LP, Wang Y, Cockcroft D, Bigler J, et al. Effects of an anti-TSLP antibody on allergen-induced asthmatic responses. N Engl J Med (2014) 370:2102–10. doi:10.1056/NEJMoa1402895
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
22. Hogan SP, Rosenberg HF, Moqbel R, Phipps S, Foster PS, Lacy P, et al. Eosinophils: biological properties and role in health and disease. Clin Exp Allergy (2008) 38:709–50. doi:10.1111/j.1365-2222.2008.02958.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
23. Nusse O, Lindau M, Cromwell O, Kay AB, Gomperts BD. Intracellular application of guanosine-5’-O-(3-thiotriphosphate) induces exocytotic granule fusion in guinea pig eosinophils. J Exp Med (1990) 171:775–86. doi:10.1084/jem.171.3.775
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
24. Scepek S, Moqbel R, Lindau M. Compound exocytosis and cumulative degranulation by eosinophils and their role in parasite killing. Parasitol Today (1994) 10:276–8. doi:10.1016/0169-4758(94)90146-5
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
25. Dvorak AM, Furitsu T, Letourneau L, Ishizaka T, Ackerman SJ. Mature eosinophils stimulated to develop in human cord blood mononuclear cell cultures supplemented with recombinant human interleukin-5. Part I. Piecemeal degranulation of specific granules and distribution of Charcot-Leyden crystal protein. Am J Pathol (1991) 138:69–82.
26. Erjefalt JS, Greiff L, Andersson M, Matsson E, Petersen H, Linden M, et al. Allergen-induced eosinophil cytolysis is a primary mechanism for granule protein release in human upper airways. Am J Respir Crit Care Med (1999) 160:304–12. doi:10.1164/ajrccm.160.1.9809048
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
27. Saffari H, Hoffman LH, Peterson KA, Fang JC, Leiferman KM, Pease LF III, et al. Electron microscopy elucidates eosinophil degranulation patterns in patients with eosinophilic esophagitis. J Allergy Clin Immunol (2014) 133(1728–1734):e1721. doi:10.1016/j.jaci.2013.11.024
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
28. Erjefalt JS, Andersson M, Greiff L, Korsgren M, Gizycki M, Jeffery PK, et al. Cytolysis and piecemeal degranulation as distinct modes of activation of airway mucosal eosinophils. J Allergy Clin Immunol (1998) 102:286–94. doi:10.1016/S0091-6749(98)70098-3
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
29. Erjefalt JS, Persson CG. New aspects of degranulation and fates of airway mucosal eosinophils. Am J Respir Crit Care Med (2000) 161:2074–85. doi:10.1164/ajrccm.161.6.9906085
30. Kroegel C, Yukawa T, Dent G, Venge P, Chung KF, Barnes PJ. Stimulation of degranulation from human eosinophils by platelet-activating factor. J Immunol (1989) 142:3518–26.
31. Dyer KD, Percopo CM, Xie Z, Yang Z, Kim JD, Davoine F, et al. Mouse and human eosinophils degranulate in response to platelet-activating factor (PAF) and lysoPAF via a PAF-receptor-independent mechanism: evidence for a novel receptor. J Immunol (2010) 184:6327–34. doi:10.4049/jimmunol.0904043
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
32. Winqvist I, Olofsson T, Olsson I. Mechanisms for eosinophil degranulation; release of the eosinophil cationic protein. Immunology (1984) 51:1–8.
33. Zeck-Kapp G, Kroegel C, Riede UN, Kapp A. Mechanisms of human eosinophil activation by complement protein C5a and platelet-activating factor: similar functional responses are accompanied by different morphologic alterations. Allergy (1995) 50:34–47. doi:10.1111/j.1398-9995.1995.tb02481.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
34. Abu-Ghazaleh RI, Fujisawa T, Mestecky J, Kyle RA, Gleich GJ. IgA-induced eosinophil degranulation. J Immunol (1989) 142:2393–400.
35. Fujisawa T, Abu-Ghazaleh R, Kita H, Sanderson CJ, Gleich GJ. Regulatory effect of cytokines on eosinophil degranulation. J Immunol (1990) 144:642–6.
36. Fujisawa T, Fukuda S, Atsuta J, Ichimi R, Kamiya H, Sakurai M. Interferon-gamma induces interleukin-3 release from peripheral blood eosinophils. Int Arch Allergy Immunol (1994) 104(Suppl 1):41–3. doi:10.1159/000236748
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
37. Horie S, Gleich GJ, Kita H. Cytokines directly induce degranulation and superoxide production from human eosinophils. J Allergy Clin Immunol (1996) 98:371–81. doi:10.1016/S0091-6749(96)70161-6
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
38. Chu VT, Frohlich A, Steinhauser G, Scheel T, Roch T, Fillatreau S, et al. Eosinophils are required for the maintenance of plasma cells in the bone marrow. Nat Immunol (2011) 12:151–9. doi:10.1038/ni.1981
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
39. Moqbel R, Hamid Q, Ying S, Barkans J, Hartnell A, Tsicopoulos A, et al. Expression of mRNA and immunoreactivity for the granulocyte/macrophage colony-stimulating factor in activated human eosinophils. J Exp Med (1991) 174:749–52. doi:10.1084/jem.174.3.749
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
40. Kita H, Ohnishi T, Okubo Y, Weiler D, Abrams JS, Gleich GJ. Granulocyte/macrophage colony-stimulating factor and interleukin 3 release from human peripheral blood eosinophils and neutrophils. J Exp Med (1991) 174:745–8. doi:10.1084/jem.174.3.745
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
41. Ohno I, Lea R, Finotto S, Marshall J, Denburg J, Dolovich J, et al. Granulocyte/macrophage colony-stimulating factor (GM-CSF) gene expression by eosinophils in nasal polyposis. Am J Respir Cell Mol Biol (1991) 5:505–10. doi:10.1165/ajrcmb/5.6.505
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
42. Broide DH, Paine MM, Firestein GS. Eosinophils express interleukin 5 and granulocyte macrophage-colony-stimulating factor mRNA at sites of allergic inflammation in asthmatics. J Clin Invest (1992) 90:1414–24. doi:10.1172/JCI116008
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
43. Takanaski S, Nonaka R, Xing Z, O’byrne P, Dolovich J, Jordana M. Interleukin 10 inhibits lipopolysaccharide-induced survival and cytokine production by human peripheral blood eosinophils. J Exp Med (1994) 180:711–5. doi:10.1084/jem.180.2.711
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
44. Levi-Schaffer F, Lacy P, Severs NJ, Newman TM, North J, Gomperts B, et al. Association of granulocyte-macrophage colony-stimulating factor with the crystalloid granules of human eosinophils. Blood (1995) 85:2579–86.
45. Weller PF, Rand TH, Barrett T, Elovic A, Wong DT, Finberg RW. Accessory cell function of human eosinophils. HLA-DR-dependent, MHC-restricted antigen-presentation and IL-1 alpha expression. J Immunol (1993) 150:2554–62.
46. Del Pozo V, De Andres B, Martin E, Maruri N, Zubeldia JM, Palomino P, et al. Murine eosinophils and IL-1: alpha IL-1 mRNA detection by in situ hybridization. Production and release of IL-1 from peritoneal eosinophils. J Immunol (1990) 144:3117–22.
47. Esnault S, Kelly EA, Nettenstrom LM, Cook EB, Seroogy CM, Jarjour NN. Human eosinophils release IL-1ss and increase expression of IL-17A in activated CD4+ T lymphocytes. Clin Exp Allergy (2012) 42:1756–64. doi:10.1111/j.1365-2222.2012.04060.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
48. Levi-Schaffer F, Barkans J, Newman TM, Ying S, Wakelin M, Hohenstein R, et al. Identification of interleukin-2 in human peripheral blood eosinophils. Immunology (1996) 87:155–61.
49. Bosse M, Audette M, Ferland C, Pelletier G, Chu HW, Dakhama A, et al. Gene expression of interleukin-2 in purified human peripheral blood eosinophils. Immunology (1996) 87:149–54.
50. Nonaka M, Nonaka R, Woolley K, Adelroth E, Miura K, Okhawara Y, et al. Distinct immunohistochemical localization of IL-4 in human inflamed airway tissues. IL-4 is localized to eosinophils in vivo and is released by peripheral blood eosinophils. J Immunol (1995) 155:3234–44.
51. Moqbel R, Ying S, Barkans J, Newman TM, Kimmitt P, Wakelin M, et al. Identification of messenger RNA for IL-4 in human eosinophils with granule localization and release of the translated product. J Immunol (1995) 155:4939–47.
52. Moller GM, De Jong TA, Van Der Kwast TH, Overbeek SE, Wierenga-Wolf AF, Thepen T, et al. Immunolocalization of interleukin-4 in eosinophils in the bronchial mucosa of atopic asthmatics. Am J Respir Cell Mol Biol (1996) 14:439–43. doi:10.1165/ajrcmb.14.5.8624248
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
53. Nakajima H, Gleich GJ, Kita H. Constitutive production of IL-4 and IL-10 and stimulated production of IL-8 by normal peripheral blood eosinophils. J Immunol (1996) 156:4859–66.
54. Desreumaux P, Janin A, Colombel JF, Prin L, Plumas J, Emilie D, et al. Interleukin 5 messenger RNA expression by eosinophils in the intestinal mucosa of patients with coeliac disease. J Exp Med (1992) 175:293–6. doi:10.1084/jem.175.1.293
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
55. Desreumaux P, Janin A, Dubucquoi S, Copin MC, Torpier G, Capron A, et al. Synthesis of interleukin-5 by activated eosinophils in patients with eosinophilic heart diseases. Blood (1993) 82:1553–60.
56. Dubucquoi S, Desreumaux P, Janin A, Klein O, Goldman M, Tavernier J, et al. Interleukin 5 synthesis by eosinophils: association with granules and immunoglobulin-dependent secretion. J Exp Med (1994) 179:703–8. doi:10.1084/jem.179.2.703
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
57. Moller GM, De Jong TA, Overbeek SE, Van Der Kwast TH, Postma DS, Hoogsteden HC. Ultrastructural immunogold localization of interleukin 5 to the crystalloid core compartment of eosinophil secondary granules in patients with atopic asthma. J Histochem Cytochem (1996) 44:67–9. doi:10.1177/44.1.8543784
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
58. Hamid Q, Barkans J, Meng Q, Ying S, Abrams JS, Kay AB, et al. Human eosinophils synthesize and secrete interleukin-6, in vitro. Blood (1992) 80:1496–501.
59. Melani C, Mattia GF, Silvani A, Care A, Rivoltini L, Parmiani G, et al. Interleukin-6 expression in human neutrophil and eosinophil peripheral blood granulocytes. Blood (1993) 81:2744–9.
60. Huang L, Gebreselassie NG, Gagliardo LF, Ruyechan MC, Lee NA, Lee JJ, et al. Eosinophil-derived IL-10 supports chronic nematode infection. J Immunol (2014) 193:4178–87. doi:10.4049/jimmunol.1400852
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
61. Minshall E, Chakir J, Laviolette M, Molet S, Zhu Z, Olivenstein R, et al. IL-11 expression is increased in severe asthma: association with epithelial cells and eosinophils. J Allergy Clin Immunol (2000) 105:232–8. doi:10.1016/S0091-6749(00)90070-8
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
62. Grewe M, Czech W, Morita A, Werfel T, Klammer M, Kapp A, et al. Human eosinophils produce biologically active IL-12: implications for control of T cell responses. J Immunol (1998) 161:415–20.
63. Woerly G, Lacy P, Younes AB, Roger N, Loiseau S, Moqbel R, et al. Human eosinophils express and release IL-13 following CD28-dependent activation. J Leukoc Biol (2002) 72:769–79.
64. Schmid-Grendelmeier P, Altznauer F, Fischer B, Bizer C, Straumann A, Menz G, et al. Eosinophils express functional IL-13 in eosinophilic inflammatory diseases. J Immunol (2002) 169:1021–7. doi:10.4049/jimmunol.169.2.1021
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
65. Lim KG, Wan HC, Bozza PT, Resnick MB, Wong DT, Cruikshank WW, et al. Human eosinophils elaborate the lymphocyte chemoattractants. IL-16 (lymphocyte chemoattractant factor) and RANTES. J Immunol (1996) 156:2566–70.
66. Molet S, Hamid Q, Davoine F, Nutku E, Taha R, Page N, et al. IL-17 is increased in asthmatic airways and induces human bronchial fibroblasts to produce cytokines. J Allergy Clin Immunol (2001) 108:430–8. doi:10.1067/mai.2001.117929
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
67. Terrier B, Bieche I, Maisonobe T, Laurendeau I, Rosenzwajg M, Kahn JE, et al. Interleukin-25: a cytokine linking eosinophils and adaptive immunity in Churg-Strauss syndrome. Blood (2010) 116:4523–31. doi:10.1182/blood-2010-02-267542
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
68. Tang W, Smith SG, Beaudin S, Dua B, Howie K, Gauvreau G, et al. IL-25 and IL-25 receptor expression on eosinophils from subjects with allergic asthma. Int Arch Allergy Immunol (2014) 163:5–10. doi:10.1159/000355331
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
69. Lamkhioued B, Gounni AS, Aldebert D, Delaporte E, Prin L, Capron A, et al. Synthesis of type 1 (IFN gamma) and type 2 (IL-4, IL-5, and IL-10) cytokines by human eosinophils. Ann N Y Acad Sci (1996) 796:203–8. doi:10.1111/j.1749-6632.1996.tb32582.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
70. Beil WJ, Weller PF, Tzizik DM, Galli SJ, Dvorak AM. Ultrastructural immunogold localization of tumor necrosis factor-α to the matrix compartment of eosinophil secondary granules in patients with idiopathic hypereosinophilic syndrome. J Histochem Cytochem (1993) 41:1611–5. doi:10.1177/41.11.8409368
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
71. Costa JJ, Matossian K, Resnick MB, Beil WJ, Wong DT, Gordon JR, et al. Human eosinophils can express the cytokines tumor necrosis factor-α and macrophage inflammatory protein-1α. J Clin Invest (1993) 91:2673–84. doi:10.1172/JCI116506
72. Tan X, Hsueh W, Gonzalez-Crussi F. Cellular localization of tumor necrosis factor (TNF)-alpha transcripts in normal bowel and in necrotizing enterocolitis. TNF gene expression by Paneth cells, intestinal eosinophils, and macrophages. Am J Pathol (1993) 142:1858–65.
73. Legrand F, Driss V, Delbeke M, Loiseau S, Hermann E, Dombrowicz D, et al. Human eosinophils exert TNF-alpha and granzyme A-mediated tumoricidal activity toward colon carcinoma cells. J Immunol (2010) 185:7443–51. doi:10.4049/jimmunol.1000446
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
74. Izumi S, Hirai K, Miyamasu M, Takahashi Y, Misaki Y, Takaishi T, et al. Expression and regulation of monocyte chemoattractant protein-1 by human eosinophils. Eur J Immunol (1997) 27:816–24. doi:10.1002/eji.1830270404
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
75. Ying S, Meng Q, Taborda-Barata L, Corrigan CJ, Barkans J, Assoufi B, et al. Human eosinophils express messenger RNA encoding RANTES and store and release biologically active RANTES protein. Eur J Immunol (1996) 26:70–6. doi:10.1002/eji.1830260111
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
76. Lamkhioued B, Renzi PM, Abi-Younes S, Garcia-Zepada EA, Allakhverdi Z, Ghaffar O, et al. Increased expression of eotaxin in bronchoalveolar lavage and airways of asthmatics contributes to the chemotaxis of eosinophils to the site of inflammation. J Immunol (1997) 159:4593–601.
77. Nakajima T, Yamada H, Iikura M, Miyamasu M, Izumi S, Shida H, et al. Intracellular localization and release of eotaxin from normal eosinophils. FEBS Lett (1998) 434:226–30. doi:10.1016/S0014-5793(98)00863-1
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
78. Gounni Abdelilah S, Wellemans V, Agouli M, Guenounou M, Hamid Q, Beck LA, et al. Increased expression of Th2-associated chemokines in bullous pemphigoid disease. Role of eosinophils in the production and release of these chemokines. Clin Immunol (2006) 120:220–31. doi:10.1016/j.clim.2006.03.014
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
79. Liu LY, Bates ME, Jarjour NN, Busse WW, Bertics PJ, Kelly EA. Generation of Th1 and Th2 chemokines by human eosinophils: evidence for a critical role of TNF-α. J Immunol (2007) 179:4840–8. doi:10.4049/jimmunol.179.8.5604-c
80. Jacobsen EA, Ochkur SI, Pero RS, Taranova AG, Protheroe CA, Colbert DC, et al. Allergic pulmonary inflammation in mice is dependent on eosinophil-induced recruitment of effector T cells. J Exp Med (2008) 205:699–710. doi:10.1084/jem.20071840
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
81. Matsumoto K, Fukuda S, Hashimoto N, Saito H. Human eosinophils produce and release a novel chemokine, CCL23, in vitro. Int Arch Allergy Immunol (2011) 155(Suppl 1):34–9. doi:10.1159/000327263
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
82. Persson-Dajotoy T, Andersson P, Bjartell A, Calafat J, Egesten A. Expression and production of the CXC chemokine growth-related oncogene-alpha by human eosinophils. J Immunol (2003) 170:5309–16. doi:10.4049/jimmunol.170.10.5309
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
83. Persson T, Monsef N, Andersson P, Bjartell A, Malm J, Calafat J, et al. Expression of the neutrophil-activating CXC chemokine ENA-78/CXCL5 by human eosinophils. Clin Exp Allergy (2003) 33:531–7. doi:10.1046/j.1365-2222.2003.01609.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
84. Braun RK, Franchini M, Erard F, Rihs S, De Vries IJ, Blaser K, et al. Human peripheral blood eosinophils produce and release interleukin-8 on stimulation with calcium ionophore. Eur J Immunol (1993) 23:956–60. doi:10.1002/eji.1830230429
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
85. Miyamasu M, Hirai K, Takahashi Y, Iida M, Yamaguchi M, Koshino T, et al. Chemotactic agonists induce cytokine generation in eosinophils. J Immunol (1995) 154:1339–49.
86. Simon HU, Yousefi S, Weber M, Simon D, Holzer C, Hartung K, et al. Human peripheral blood eosinophils express and release interleukin-8. Int Arch Allergy Immunol (1995) 107:124–6. doi:10.1159/000236950
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
87. Yousefi S, Hemmann S, Weber M, Holzer C, Hartung K, Blaser K, et al. IL-8 is expressed by human peripheral blood eosinophils. Evidence for increased secretion in asthma. J Immunol (1995) 154:5481–90.
88. Dajotoy T, Andersson P, Bjartell A, Lofdahl CG, Tapper H, Egesten A. Human eosinophils produce the T cell-attracting chemokines MIG and IP-10 upon stimulation with IFN-gamma. J Leukoc Biol (2004) 76:685–91. doi:10.1189/jlb.0803379
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
89. Powell PP, Klagsbrun M, Abraham JA, Jones RC. Eosinophils expressing heparin-binding EGF-like growth factor mRNA localize around lung microvessels in pulmonary hypertension. Am J Pathol (1993) 143:784–93.
90. Solomon A, Aloe L, Pe’er J, Frucht-Pery J, Bonini S, Levi-Schaffer F. Nerve growth factor is preformed in and activates human peripheral blood eosinophils. J Allergy Clin Immunol (1998) 102:454–60. doi:10.1016/S0091-6749(98)70135-6
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
91. Ohno I, Nitta Y, Yamauchi K, Hoshi H, Honma M, Woolley K, et al. Eosinophils as a potential source of platelet-derived growth factor B-chain (PDGF-B) in nasal polyposis and bronchial asthma. Am J Respir Cell Mol Biol (1995) 13:639–47. doi:10.1165/ajrcmb.13.6.7576701
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
92. Hartman M, Piliponsky AM, Temkin V, Levi-Schaffer F. Human peripheral blood eosinophils express stem cell factor. Blood (2001) 97:1086–91. doi:10.1182/blood.V97.4.1086
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
93. Wong DT, Weller PF, Galli SJ, Elovic A, Rand TH, Gallagher GT, et al. Human eosinophils express transforming growth factor alpha. J Exp Med (1990) 172:673–81. doi:10.1084/jem.172.3.673
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
94. Todd R, Donoff BR, Chiang T, Chou MY, Elovic A, Gallagher GT, et al. The eosinophil as a cellular source of transforming growth factor alpha in healing cutaneous wounds. Am J Pathol (1991) 138:1307–13.
95. Elovic A, Wong DT, Weller PF, Matossian K, Galli SJ. Expression of transforming growth factors-alpha and beta 1 messenger RNA and product by eosinophils in nasal polyps. J Allergy Clin Immunol (1994) 93:864–9. doi:10.1016/0091-6749(94)90379-4
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
96. Elovic AE, Ohyama H, Sauty A, Mcbride J, Tsuji T, Nagai M, et al. IL-4-dependent regulation of TGF-alpha and TGF-beta1 expression in human eosinophils. J Immunol (1998) 160:6121–7.
97. Wong DT, Elovic A, Matossian K, Nagura N, Mcbride J, Chou MY, et al. Eosinophils from patients with blood eosinophilia express transforming growth factor beta 1. Blood (1991) 78:2702–7.
98. Ohno I, Lea RG, Flanders KC, Clark DA, Banwatt D, Dolovich J, et al. Eosinophils in chronically inflamed human upper airway tissues express transforming growth factor beta 1 gene (TGF beta 1). J Clin Invest (1992) 89:1662–8. doi:10.1172/JCI115764
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
99. Kadin M, Butmarc J, Elovic A, Wong D. Eosinophils are the major source of transforming growth factor-beta 1 in nodular sclerosing Hodgkin’s disease. Am J Pathol (1993) 142:11–6.
100. Wong DT, Donoff RB, Yang J, Song BZ, Matossian K, Nagura N, et al. Sequential expression of transforming growth factors alpha and beta 1 by eosinophils during cutaneous wound healing in the hamster. Am J Pathol (1993) 143:130–42.
101. Ohno I, Nitta Y, Yamauchi K, Hoshi H, Honma M, Woolley K, et al. Transforming growth factor beta 1 (TGF beta 1) gene expression by eosinophils in asthmatic airway inflammation. Am J Respir Cell Mol Biol (1996) 15:404–9. doi:10.1165/ajrcmb.15.3.8810646
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
102. Minshall EM, Leung DY, Martin RJ, Song YL, Cameron L, Ernst P, et al. Eosinophil-associated TGF-beta1 mRNA expression and airways fibrosis in bronchial asthma. Am J Respir Cell Mol Biol (1997) 17:326–33. doi:10.1165/ajrcmb.17.3.2733
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
103. Shen ZJ, Esnault S, Rosenthal LA, Szakaly RJ, Sorkness RL, Westmark PR, et al. Pin1 regulates TGF-beta1 production by activated human and murine eosinophils and contributes to allergic lung fibrosis. J Clin Invest (2008) 118:479–90. doi:10.1172/JCI32789
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
104. Horiuchi T, Weller PF. Expression of vascular endothelial growth factor by human eosinophils: upregulation by granulocyte macrophage colony-stimulating factor and interleukin-5. Am J Respir Cell Mol Biol (1997) 17:70–7. doi:10.1165/ajrcmb.17.1.2796
105. Hoshino M, Takahashi M, Aoike N. Expression of vascular endothelial growth factor, basic fibroblast growth factor, and angiogenin immunoreactivity in asthmatic airways and its relationship to angiogenesis. J Allergy Clin Immunol (2001) 107:295–301. doi:10.1067/mai.2001.115626
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
106. Simson L, Ellyard JI, Dent LA, Matthaei KI, Rothenberg ME, Foster PS, et al. Regulation of carcinogenesis by IL-5 and CCL11: a potential role for eosinophils in tumor immune surveillance. J Immunol (2007) 178:4222–9. doi:10.4049/jimmunol.178.7.4222
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
107. Tedeschi A, Asero R, Marzano AV, Lorini M, Fanoni D, Berti E, et al. Plasma levels and skin-eosinophil-expression of vascular endothelial growth factor in patients with chronic urticaria. Allergy (2009) 64:1616–22. doi:10.1111/j.1398-9995.2009.02069.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
108. Durham SR, Ying S, Varney VA, Jacobson MR, Sudderick RM, Mackay IS, et al. Cytokine messenger RNA expression for IL-3, IL-4, IL-5, and granulocyte/macrophage-colony-stimulating factor in the nasal mucosa after local allergen provocation: relationship to tissue eosinophilia. J Immunol (1992) 148:2390–4.
109. Rajakulasingam K, Hamid Q, O’brien F, Shotman E, Jose PJ, Williams TJ, et al. RANTES in human allergen-induced rhinitis: cellular source and relation to tissue eosinophilia. Am J Respir Crit Care Med (1997) 155:696–703. doi:10.1164/ajrccm.155.2.9032215
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
110. Allen JS, Eisma R, Lafreniere D, Leonard G, Kreutzer D. Characterization of the eosinophil chemokine RANTES in nasal polyps. Ann Otol Rhinol Laryngol (1998) 107:416–20. doi:10.1177/000348949810700510
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
111. Barata LT, Ying S, Meng Q, Barkans J, Rajakulasingam K, Durham SR, et al. IL-4- and IL-5-positive T lymphocytes, eosinophils, and mast cells in allergen-induced late-phase cutaneous reactions in atopic subjects. J Allergy Clin Immunol (1998) 101:222–30. doi:10.1016/S0091-6749(98)70387-2
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
112. Ying S, Humbert M, Barkans J, Corrigan CJ, Pfister R, Menz G, et al. Expression of IL-4 and IL-5 mRNA and protein product by CD4+ and CD8+ T cells, eosinophils, and mast cells in bronchial biopsies obtained from atopic and nonatopic (intrinsic) asthmatics. J Immunol (1997) 158:3539–44.
113. Girgis-Gabardo A, Kanai N, Denburg JA, Hargreave FE, Jordana M, Dolovich J. Immunocytochemical detection of granulocyte-macrophage colony-stimulating factor and eosinophil cationic protein in sputum cells. J Allergy Clin Immunol (1994) 93:945–7. doi:10.1016/0091-6749(94)90390-5
114. Egesten A, Calafat J, Knol EF, Janssen H, Walz TM. Subcellular localization of transforming growth factor-alpha in human eosinophil granulocytes. Blood (1996) 87:3910–8.
115. Logan MR, Odemuyiwa SO, Moqbel R. Understanding exocytosis in immune and inflammatory cells: the molecular basis of mediator secretion. J Allergy Clin Immunol (2003) 111:923–32. doi:10.1067/mai.2003.1573
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
116. Melo RC, Weller PF. Piecemeal degranulation in human eosinophils: a distinct secretion mechanism underlying inflammatory responses. Histol Histopathol (2010) 25:1341–54.
117. Stanley AC, Lacy P. Pathways for cytokine secretion. Physiology (2010) 25:218–29. doi:10.1152/physiol.00017.2010
118. Lacy P, Stow JL. Cytokine release from innate immune cells: association with diverse membrane trafficking pathways. Blood (2011) 118:9–18. doi:10.1182/blood-2010-08-265892
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
119. Bandeira-Melo C, Sugiyama K, Woods LJ, Weller PF. Cutting edge: eotaxin elicits rapid vesicular transport-mediated release of preformed IL-4 from human eosinophils. J Immunol (2001) 166:4813–7. doi:10.4049/jimmunol.166.8.4813
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
120. Melo RC, Perez SA, Spencer LA, Dvorak AM, Weller PF. Intragranular vesiculotubular compartments are involved in piecemeal degranulation by activated human eosinophils. Traffic (2005) 6:866–79. doi:10.1111/j.1600-0854.2005.00322.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
121. Spencer LA, Melo RC, Perez SA, Bafford SP, Dvorak AM, Weller PF. Cytokine receptor-mediated trafficking of preformed IL-4 in eosinophils identifies an innate immune mechanism of cytokine secretion. Proc Natl Acad Sci U S A (2006) 103:3333–8. doi:10.1073/pnas.0508946103
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
122. Melo RC, Spencer LA, Dvorak AM, Weller PF. Mechanisms of eosinophil secretion: large vesiculotubular carriers mediate transport and release of granule-derived cytokines and other proteins. J Leukoc Biol (2008) 83:229–36. doi:10.1189/jlb.0707503
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
123. Spencer LA, Bonjour LA, Melo RC, Weller PF. Eosinophil secretion of granule-derived cytokines. Front Immunol (2014) 5:496. doi:10.3389/fimmu.2014.00496
124. Lacy P, Logan MR, Bablitz B, Moqbel R. Fusion protein vesicle-associated membrane protein 2 is implicated in IFNγ-induced piecemeal degranulation in human eosinophils from atopic individuals. J Allergy Clin Immunol (2001) 107:671–8. doi:10.1067/mai.2001.113562
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
125. Sudhof TC, Rothman JE. Membrane fusion: grappling with SNARE and SM proteins. Science (2009) 323:474–7. doi:10.1126/science.1161748
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
126. Logan MR, Lacy P, Bablitz B, Moqbel R. Expression of eosinophil target SNAREs as potential cognate receptors for vesicle-associated membrane protein-2 in exocytosis. J Allergy Clin Immunol (2002) 109:299–306. doi:10.1067/mai.2002.121453
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
127. Logan MR, Lacy P, Odemuyiwa SO, Steward M, Davoine F, Kita H, et al. A critical role for vesicle-associated membrane protein-7 in exocytosis from human eosinophils and neutrophils. Allergy (2006) 61:777–84. doi:10.1111/j.1398-9995.2006.01089.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
128. Ochkur SI, Kim JD, Protheroe CA, Colbert D, Moqbel R, Lacy P, et al. The development of a sensitive and specific ELISA for mouse eosinophil peroxidase: assessment of eosinophil degranulation ex vivo and in models of human disease. J Immunol Methods (2012) 375:138–47. doi:10.1016/j.jim.2011.10.002
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
129. Eum SY, Haile S, Lefort J, Huerre M, Vargaftig BB. Eosinophil recruitment into the respiratory epithelium following antigenic challenge in hyper-IgE mice is accompanied by interleukin 5-dependent bronchial hyperresponsiveness. Proc Natl Acad Sci U S A (1995) 92:12290–4. doi:10.1073/pnas.92.26.12290
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
130. Korsgren M, Erjefalt JS, Korsgren O, Sundler F, Persson CG. Allergic eosinophil-rich inflammation develops in lungs and airways of B cell-deficient mice. J Exp Med (1997) 185:885–92. doi:10.1084/jem.185.5.885
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
131. Haile S, Lefort J, Joseph D, Gounon P, Huerre M, Vargaftig BB. Mucous-cell metaplasia and inflammatory-cell recruitment are dissociated in allergic mice after antibody- and drug-dependent cell depletion in a murine model of asthma. Am J Respir Cell Mol Biol (1999) 20:891–902. doi:10.1165/ajrcmb.20.5.3446
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
132. Malm-Erjefalt M, Persson CG, Erjefalt JS. Degranulation status of airway tissue eosinophils in mouse models of allergic airway inflammation. Am J Respir Cell Mol Biol (2001) 24:352–9. doi:10.1165/ajrcmb.24.3.4357
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
133. Lacy P, Willetts L, Kim JD, Lo AN, Lam B, Maclean EI, et al. Agonist activation of F-actin-mediated eosinophil shape change and mediator release is dependent on Rac2. Int Arch Allergy Immunol (2011) 156:137–47. doi:10.1159/000322597
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
134. Kim JD, Willetts L, Ochkur S, Srivastava N, Hamburg R, Shayeganpour A, et al. An essential role for Rab27a GTPase in eosinophil exocytosis. J Leukoc Biol (2013) 94:1265–74. doi:10.1189/jlb.0812431
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
135. Stow JL, Manderson AP, Murray RZ. SNAREing immunity: the role of SNAREs in the immune system. Nat Rev Immunol (2006) 6:919–29. doi:10.1038/nri1980
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
136. Sun CX, Magalhaes MA, Glogauer M. Rac1 and Rac2 differentially regulate actin free barbed end formation downstream of the fMLP receptor. J Cell Biol (2007) 179:239–45. doi:10.1083/jcb.200705122
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
137. Schall TJ, Bacon K, Toy KJ, Goeddel DV. Selective attraction of monocytes and T lymphocytes of the memory phenotype by cytokine RANTES. Nature (1990) 347:669–71. doi:10.1038/347669a0
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
138. Taub DD, Conlon K, Lloyd AR, Oppenheim JJ, Kelvin DJ. Preferential migration of activated CD4+ and CD8+ T cells in response to MIP-1 alpha and MIP-1 beta. Science (1993) 260:355–8. doi:10.1126/science.7682337
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
139. Rot A, Krieger M, Brunner T, Bischoff SC, Schall TJ, Dahinden CA. RANTES and macrophage inflammatory protein 1 alpha induce the migration and activation of normal human eosinophil granulocytes. J Exp Med (1992) 176:1489–95. doi:10.1084/jem.176.6.1489
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
140. Alam R, Stafford S, Forsythe P, Harrison R, Faubion D, Lett-Brown MA, et al. RANTES is a chemotactic and activating factor for human eosinophils. J Immunol (1993) 150:3442–8.
141. Kapp A, Zeck-Kapp G, Czech W, Schopf E. The chemokine RANTES is more than a chemoattractant: characterization of its effect on human eosinophil oxidative metabolism and morphology in comparison with IL-5 and GM-CSF. J Invest Dermatol (1994) 102:906–14. doi:10.1111/1523-1747.ep12383399
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
142. Jose PJ, Griffiths-Johnson DA, Collins PD, Walsh DT, Moqbel R, Totty NF, et al. Eotaxin: a potent eosinophil chemoattractant cytokine detected in a guinea pig model of allergic airways inflammation. J Exp Med (1994) 179:881–7. doi:10.1084/jem.179.3.881
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
143. Rothenberg ME, Maclean JA, Pearlman E, Luster AD, Leder P. Targeted disruption of the chemokine eotaxin partially reduces antigen-induced tissue eosinophilia. J Exp Med (1997) 185:785–90. doi:10.1084/jem.185.4.785
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
144. Elsner J, Hochstetter R, Kimmig D, Kapp A. Human eotaxin represents a potent activator of the respiratory burst of human eosinophils. Eur J Immunol (1996) 26:1919–25. doi:10.1002/eji.1830260837
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
145. Tenscher K, Metzner B, Schopf E, Norgauer J, Czech W. Recombinant human eotaxin induces oxygen radical production, Ca(2+)-mobilization, actin reorganization, and CD11b upregulation in human eosinophils via a pertussis toxin-sensitive heterotrimeric guanine nucleotide-binding protein. Blood (1996) 88:3195–9.
146. El-Shazly A, Masuyama K, Nakano K, Eura M, Samejima Y, Ishikawa T. Human eotaxin induces eosinophil-derived neurotoxin release from normal human eosinophils. Int Arch Allergy Immunol (1998) 117(Suppl 1):55–8. doi:10.1159/000053573
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
147. Baggiolini M, Walz A, Kunkel SL. Neutrophil-activating peptide-1/interleukin 8, a novel cytokine that activates neutrophils. J Clin Invest (1989) 84:1045–9. doi:10.1172/JCI114265
148. Warringa RA, Koenderman L, Kok PT, Kreukniet J, Bruijnzeel PL. Modulation and induction of eosinophil chemotaxis by granulocyte-macrophage colony-stimulating factor and interleukin-3. Blood (1991) 77:2694–700.
149. Standiford TJ, Kunkel SL, Kasahara K, Milia MJ, Rolfe MW, Strieter RM. Interleukin-8 gene expression from human alveolar macrophages: the role of adherence. Am J Respir Cell Mol Biol (1991) 5:579–85. doi:10.1165/ajrcmb/5.6.579
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
150. Shi HZ, Humbles A, Gerard C, Jin Z, Weller PF. Lymph node trafficking and antigen presentation by endobronchial eosinophils. J Clin Invest (2000) 105:945–53. doi:10.1172/JCI8945
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
151. Shi HZ. Eosinophils function as antigen-presenting cells. J Leukoc Biol (2004) 76:520–7. doi:10.1189/jlb.0404228
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
152. Wang HB, Ghiran I, Matthaei K, Weller PF. Airway eosinophils: allergic inflammation recruited professional antigen-presenting cells. J Immunol (2007) 179:7585–92. doi:10.4049/jimmunol.179.11.7585
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
153. Akuthota P, Wang HB, Spencer LA, Weller PF. Immunoregulatory roles of eosinophils: a new look at a familiar cell. Clin Exp Allergy (2008) 38:1254–63. doi:10.1111/j.1365-2222.2008.03037.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
154. Padigel UM, Lee JJ, Nolan TJ, Schad GA, Abraham D. Eosinophils can function as antigen-presenting cells to induce primary and secondary immune responses to Strongyloides stercoralis. Infect Immun (2006) 74:3232–8. doi:10.1128/IAI.02067-05
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
155. Blanchard D, Gaillard C, Hermann P, Banchereau J. Role of CD40 antigen and interleukin-2 in T cell-dependent human B lymphocyte growth. Eur J Immunol (1994) 24:330–5. doi:10.1002/eji.1830240209
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
156. Rand TH, Silberstein DS, Kornfeld H, Weller PF. Human eosinophils express functional interleukin 2 receptors. J Clin Invest (1991) 88:825–32. doi:10.1172/JCI115383
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
157. Rothenberg ME, Owen WF Jr, Silberstein DS, Woods J, Soberman RJ, Austen KF, et al. Human eosinophils have prolonged survival, enhanced functional properties, and become hypodense when exposed to human interleukin 3. J Clin Invest (1988) 81:1986–92. doi:10.1172/JCI113547
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
158. Steinke JW, Borish L. Th2 cytokines and asthma. Interleukin-4: its role in the pathogenesis of asthma, and targeting it for asthma treatment with interleukin-4 receptor antagonists. Respir Res (2001) 2:66–70. doi:10.1186/rr40
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
159. Abbas AK, Murphy KM, Sher A. Functional diversity of helper T lymphocytes. Nature (1996) 383:787–93. doi:10.1038/383787a0
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
160. Lohning M, Stroehmann A, Coyle AJ, Grogan JL, Lin S, Gutierrez-Ramos JC, et al. T1/ST2 is preferentially expressed on murine Th2 cells, independent of interleukin 4, interleukin 5, and interleukin 10, and important for Th2 effector function. Proc Natl Acad Sci U S A (1998) 95:6930–5. doi:10.1073/pnas.95.12.6930
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
161. Shapira SK, Jabara HH, Thienes CP, Ahern DJ, Vercelli D, Gould HJ, et al. Deletional switch recombination occurs in interleukin-4-induced isotype switching to IgE expression by human B cells. Proc Natl Acad Sci U S A (1991) 88:7528–32. doi:10.1073/pnas.88.17.7528
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
162. Dubois GR, Bruijnzeel-Koomen CA, Bruijnzeel PL. IL-4 induces chemotaxis of blood eosinophils from atopic dermatitis patients, but not from normal individuals. J Invest Dermatol (1994) 102:843–6. doi:10.1111/1523-1747.ep12382362
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
163. Schleimer RP, Sterbinsky SA, Kaiser J, Bickel CA, Klunk DA, Tomioka K, et al. IL-4 induces adherence of human eosinophils and basophils but not neutrophils to endothelium. association with expression of VCAM-1. J Immunol (1992) 148:1086–92.
164. Walsh GM, Mermod JJ, Hartnell A, Kay AB, Wardlaw AJ. Human eosinophil, but not neutrophil, adherence to IL-1-stimulated human umbilical vascular endothelial cells is alpha 4 beta 1 (very late antigen-4) dependent. J Immunol (1991) 146:3419–23.
165. Yamaguchi Y, Suda T, Suda J, Eguchi M, Miura Y, Harada N, et al. Purified interleukin 5 supports the terminal differentiation and proliferation of murine eosinophilic precursors. J Exp Med (1988) 167:43–56. doi:10.1084/jem.167.1.43
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
166. Shalit M, Sekhsaria S, Malech HL. Modulation of growth and differentiation of eosinophils from human peripheral blood CD34+ cells by IL5 and other growth factors. Cell Immunol (1995) 160:50–7. doi:10.1016/0008-8749(95)80008-7
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
167. Yamaguchi Y, Hayashi Y, Sugama Y, Miura Y, Kasahara T, Kitamura S, et al. Highly purified murine interleukin 5 (IL-5) stimulates eosinophil function and prolongs in vitro survival. IL-5 as an eosinophil chemotactic factor. J Exp Med (1988) 167:1737–42. doi:10.1084/jem.167.5.1737
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
168. Kita H, Weiler DA, Abu-Ghazaleh R, Sanderson CJ, Gleich GJ. Release of granule proteins from eosinophils cultured with IL-5. J Immunol (1992) 149:629–35.
169. Yamaguchi Y, Suda T, Ohta S, Tominaga K, Miura Y, Kasahara T. Analysis of the survival of mature human eosinophils: interleukin-5 prevents apoptosis in mature human eosinophils. Blood (1991) 78:2542–7.
170. Vercelli D, Jabara HH, Arai K, Yokota T, Geha RS. Endogenous interleukin 6 plays an obligatory role in interleukin 4-dependent human IgE synthesis. Eur J Immunol (1989) 19:1419–24. doi:10.1002/eji.1830190811
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
171. Yokoyama A, Kohno N, Fujino S, Hamada H, Inoue Y, Fujioka S, et al. Circulating interleukin-6 levels in patients with bronchial asthma. Am J Respir Crit Care Med (1995) 151:1354–8. doi:10.1164/ajrccm.151.5.7735584
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
172. Deetz DC, Jagielo PJ, Quinn TJ, Thorne PS, Bleuer SA, Schwartz DA. The kinetics of grain dust-induced inflammation of the lower respiratory tract. Am J Respir Crit Care Med (1997) 155:254–9. doi:10.1164/ajrccm.155.1.9001321
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
173. Gounni AS, Gregory B, Nutku E, Aris F, Latifa K, Minshall E, et al. Interleukin-9 enhances interleukin-5 receptor expression, differentiation, and survival of human eosinophils. Blood (2000) 96:2163–71.
174. Gounni AS, Nutku E, Koussih L, Aris F, Louahed J, Levitt RC, et al. IL-9 expression by human eosinophils: regulation by IL-1beta and TNF-alpha. J Allergy Clin Immunol (2000) 106:460–6. doi:10.1067/mai.2000.109172
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
175. Jeannin P, Lecoanet S, Delneste Y, Gauchat JF, Bonnefoy JY. IgE versus IgG4 production can be differentially regulated by IL-10. J Immunol (1998) 160:3555–61.
176. Grunig G, Corry DB, Leach MW, Seymour BW, Kurup VP, Rennick DM. Interleukin-10 is a natural suppressor of cytokine production and inflammation in a murine model of allergic bronchopulmonary aspergillosis. J Exp Med (1997) 185:1089–99. doi:10.1084/jem.185.6.1089
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
177. Kosaka S, Tamauchi H, Terashima M, Maruyama H, Habu S, Kitasato H. IL-10 controls Th2-type cytokine production and eosinophil infiltration in a mouse model of allergic airway inflammation. Immunobiology (2011) 216:811–20. doi:10.1016/j.imbio.2010.12.003
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
178. Zheng T, Zhu Z, Wang Z, Homer RJ, Ma B, Riese RJ Jr., et al. Inducible targeting of IL-13 to the adult lung causes matrix metalloproteinase- and cathepsin-dependent emphysema. J Clin Invest (2000) 106:1081–93. doi:10.1172/JCI10458
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
179. Punnonen J, Aversa G, Cocks BG, Mckenzie AN, Menon S, Zurawski G, et al. Interleukin 13 induces interleukin 4-independent IgG4 and IgE synthesis and CD23 expression by human B cells. Proc Natl Acad Sci U S A (1993) 90:3730–4. doi:10.1073/pnas.90.8.3730
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
180. Wills-Karp M, Luyimbazi J, Xu X, Schofield B, Neben TY, Karp CL, et al. Interleukin-13: central mediator of allergic asthma. Science (1998) 282:2258–61. doi:10.1126/science.282.5397.2258
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
181. Urban JF Jr, Noben-Trauth N, Donaldson DD, Madden KB, Morris SC, Collins M, et al. IL-13, IL-4Ralpha, and Stat6 are required for the expulsion of the gastrointestinal nematode parasite Nippostrongylus brasiliensis. Immunity (1998) 8:255–64. doi:10.1016/S1074-7613(00)80477-X
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
182. Bonini S, Lambiase A, Bonini S, Angelucci F, Magrini L, Manni L, et al. Circulating nerve growth factor levels are increased in humans with allergic diseases and asthma. Proc Natl Acad Sci U S A (1996) 93:10955–60. doi:10.1073/pnas.93.20.10955
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
183. Levi-Montalcini R, Skaper SD, Dal Toso R, Petrelli L, Leon A. Nerve growth factor: from neurotrophin to neurokine. Trends Neurosci (1996) 19:514–20. doi:10.1016/S0166-2236(96)10058-8
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
184. Matsuda H, Coughlin MD, Bienenstock J, Denburg JA. Nerve growth factor promotes human hemopoietic colony growth and differentiation. Proc Natl Acad Sci U S A (1988) 85:6508–12. doi:10.1073/pnas.85.17.6508
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
186. Wynn TA. Integrating mechanisms of pulmonary fibrosis. J Exp Med (2011) 208:1339–50. doi:10.1084/jem.20110551
187. Slungaard A, Vercellotti GM, Walker G, Nelson RD, Jacob HS. Tumor necrosis factor alpha/cachectin stimulates eosinophil oxidant production and toxicity towards human endothelium. J Exp Med (1990) 171:2025–41. doi:10.1084/jem.171.6.2025
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
188. Tonnel AB, Gosset P, Molet S, Tillie-Leblond I, Jeannin P, Joseph M. Interactions between endothelial cells and effector cells in allergic inflammation. Ann N Y Acad Sci (1996) 796:9–20. doi:10.1111/j.1749-6632.1996.tb32562.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
189. Holt PG, Macaubas C, Stumbles PA, Sly PD. The role of allergy in the development of asthma. Nature (1999) 402:B12–7. doi:10.1038/46890
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
190. Grewe M, Walther S, Gyufko K, Czech W, Schopf E, Krutmann J. Analysis of the cytokine pattern expressed in situ in inhalant allergen patch test reactions of atopic dermatitis patients. J Invest Dermatol (1995) 105:407–10. doi:10.1111/1523-1747.ep12321078
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
191. Grewe M, Bruijnzeel-Koomen CA, Schopf E, Thepen T, Langeveld-Wildschut AG, Ruzicka T, et al. A role for Th1 and Th2 cells in the immunopathogenesis of atopic dermatitis. Immunol Today (1998) 19:359–61. doi:10.1016/S0167-5699(98)01285-7
192. Herz U, Lacy P, Renz H, Erb K. The influence of infections on the development and severity of allergic disorders. Curr Opin Immunol (2000) 12:632–40. doi:10.1016/S0952-7915(00)00155-2
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
193. Chu VT, Beller A, Rausch S, Strandmark J, Zanker M, Arbach O, et al. Eosinophils promote generation and maintenance of immunoglobulin-A-expressing plasma cells and contribute to gut immune homeostasis. Immunity (2014) 40:582–93. doi:10.1016/j.immuni.2014.02.014
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
194. Cruikshank WW, Center DM, Nisar N, Wu M, Natke B, Theodore AC, et al. Molecular and functional analysis of a lymphocyte chemoattractant factor: association of biologic function with CD4 expression. Proc Natl Acad Sci U S A (1994) 91:5109–13. doi:10.1073/pnas.91.11.5109
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
195. Cruikshank WW, Lim K, Theodore AC, Cook J, Fine G, Weller PF, et al. IL-16 inhibition of CD3-dependent lymphocyte activation and proliferation. J Immunol (1996) 157:5240–8.
196. Cosmi L, Liotta F, Maggi E, Romagnani S, Annunziato F. Th17 cells: new players in asthma pathogenesis. Allergy (2011) 66:989–98. doi:10.1111/j.1398-9995.2011.02576.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
197. Conrad DH, Ben-Sasson SZ, Le Gros G, Finkelman FD, Paul WE. Infection with Nippostrongylus brasiliensis or injection of anti-IgD antibodies markedly enhances Fc-receptor-mediated interleukin 4 production by non-B, non-T cells. J Exp Med (1990) 171:1497–508. doi:10.1084/jem.171.5.1497
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
198. Voehringer D, Shinkai K, Locksley RM. Type 2 immunity reflects orchestrated recruitment of cells committed to IL-4 production. Immunity (2004) 20:267–77. doi:10.1016/S1074-7613(04)00026-3
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
199. Gessner A, Mohrs K, Mohrs M. Mast cells, basophils, and eosinophils acquire constitutive IL-4 and IL-13 transcripts during lineage differentiation that are sufficient for rapid cytokine production. J Immunol (2005) 174:1063–72. doi:10.4049/jimmunol.174.2.1063
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
200. Chen L, Grabowski KA, Xin JP, Coleman J, Huang Z, Espiritu B, et al. IL-4 induces differentiation and expansion of Th2 cytokine-producing eosinophils. J Immunol (2004) 172:2059–66. doi:10.4049/jimmunol.172.4.2059
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
201. Piehler D, Stenzel W, Grahnert A, Held J, Richter L, Kohler G, et al. Eosinophils contribute to IL-4 production and shape the T-helper cytokine profile and inflammatory response in pulmonary cryptococcosis. Am J Pathol (2011) 179:733–44. doi:10.1016/j.ajpath.2011.04.025
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
202. Qiu Y, Nguyen KD, Odegaard JI, Cui X, Tian X, Locksley RM, et al. Eosinophils and type 2 cytokine signaling in macrophages orchestrate development of functional beige fat. Cell (2014) 157:1292–308. doi:10.1016/j.cell.2014.03.066
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
203. Maizels RM, Allen JE. Immunology. Eosinophils forestall obesity. Science (2011) 332:186–7. doi:10.1126/science.1205313
204. Wu D, Molofsky AB, Liang HE, Ricardo-Gonzalez RR, Jouihan HA, Bando JK, et al. Eosinophils sustain adipose alternatively activated macrophages associated with glucose homeostasis. Science (2011) 332:243–7. doi:10.1126/science.1201475
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
205. Lee SD, Tontonoz P. Eosinophils in fat: pink is the new brown. Cell (2014) 157:1249–50. doi:10.1016/j.cell.2014.05.025
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
206. Wong CK, Cheung PF, Lam CW. Leptin-mediated cytokine release and migration of eosinophils: implications for immunopathophysiology of allergic inflammation. Eur J Immunol (2007) 37:2337–48. doi:10.1002/eji.200636866
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
207. Goh YP, Henderson NC, Heredia JE, Red Eagle A, Odegaard JI, Lehwald N, et al. Eosinophils secrete IL-4 to facilitate liver regeneration. Proc Natl Acad Sci U S A (2013) 110:9914–9. doi:10.1073/pnas.1304046110
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
208. Walsh ER, Thakar J, Stokes K, Huang F, Albert R, August A. Computational and experimental analysis reveals a requirement for eosinophil-derived IL-13 for the development of allergic airway responses in C57BL/6 mice. J Immunol (2011) 186:2936–49. doi:10.4049/jimmunol.1001148
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
209. Levi-Schaffer F, Garbuzenko E, Rubin A, Reich R, Pickholz D, Gillery P, et al. Human eosinophils regulate human lung- and skin-derived fibroblast properties in vitro: a role for transforming growth factor beta (TGF-beta). Proc Natl Acad Sci U S A (1999) 96:9660–5. doi:10.1073/pnas.96.17.9660
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
210. Phipps S, Ying S, Wangoo A, Ong YE, Levi-Schaffer F, Kay AB. The relationship between allergen-induced tissue eosinophilia and markers of repair and remodeling in human atopic skin. J Immunol (2002) 169:4604–12. doi:10.4049/jimmunol.169.8.4604
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
211. Sabin EA, Kopf MA, Pearce EJ. Schistosoma mansoni egg-induced early IL-4 production is dependent upon IL-5 and eosinophils. J Exp Med (1996) 184:1871–8. doi:10.1084/jem.184.5.1871
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
212. Fabre V, Beiting DP, Bliss SK, Gebreselassie NG, Gagliardo LF, Lee NA, et al. Eosinophil deficiency compromises parasite survival in chronic nematode infection. J Immunol (2009) 182:1577–83. doi:10.4049/jimmunol.182.3.1577
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
214. Macdonald D, Gordon AA, Kajitani H, Enokihara H, Barrett AJ. Interleukin-2 treatment-associated eosinophilia is mediated by interleukin-5 production. Br J Haematol (1990) 76:168–73. doi:10.1111/j.1365-2141.1990.tb07867.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
215. Van Haelst Pisani C, Kovach JS, Kita H, Leiferman KM, Gleich GJ, Silver JE, et al. Administration of interleukin-2 (IL-2) results in increased plasma concentrations of IL-5 and eosinophilia in patients with cancer. Blood (1991) 78:1538–44.
216. Sosman JA, Bartemes K, Offord KP, Kita H, Fisher SG, Kefer C, et al. Evidence for eosinophil activation in cancer patients receiving recombinant interleukin-4: effects of interleukin-4 alone and following interleukin-2 administration. Clin Cancer Res (1995) 1:805–12.
217. Goldsmith MM, Belchis DA, Cresson DH, Merritt WD III, Askin FB. The importance of the eosinophil in head and neck cancer. Otolaryngol Head Neck Surg (1992) 106:27–33.
218. Huland E, Huland H. Tumor-associated eosinophilia in interleukin-2-treated patients: evidence of toxic eosinophil degranulation on bladder cancer cells. J Cancer Res Clin Oncol (1992) 118:463–7. doi:10.1007/BF01629431
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
219. Horiuchi K, Mishima K, Ohsawa M, Sugimura M, Aozasa K. Prognostic factors for well-differentiated squamous cell carcinoma in the oral cavity with emphasis on immunohistochemical evaluation. J Surg Oncol (1993) 53:92–6. doi:10.1002/jso.2930530209
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
220. Dorta RG, Landman G, Kowalski LP, Lauris JR, Latorre MR, Oliveira DT. Tumour-associated tissue eosinophilia as a prognostic factor in oral squamous cell carcinomas. Histopathology (2002) 41:152–7. doi:10.1046/j.1365-2559.2002.01437.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
221. Lorena SC, Oliveira DT, Dorta RG, Landman G, Kowalski LP. Eotaxin expression in oral squamous cell carcinomas with and without tumour associated tissue eosinophilia. Oral Dis (2003) 9:279–83. doi:10.1034/j.1601-0825.2003.00958.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
222. Davoine F, Sim A, Tang C, Fisher S, Ethier C, Puttagunta L, et al. Eosinophils in human oral squamous carcinoma; role of prostaglandin D2. J Inflamm (2013) 10:4. doi:10.1186/1476-9255-10-4
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
223. Roth N, Stadler S, Lemann M, Hosli S, Simon HU, Simon D. Distinct eosinophil cytokine expression patterns in skin diseases – the possible existence of functionally different eosinophil subpopulations. Allergy (2011) 66:1477–86. doi:10.1111/j.1398-9995.2011.02694.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
224. Gatault S, Legrand F, Delbeke M, Loiseau S, Capron M. Involvement of eosinophils in the anti-tumor response. Cancer Immunol Immunother (2012) 61:1527–34. doi:10.1007/s00262-012-1288-3
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
225. Benatar T, Cao MY, Lee Y, Li H, Feng N, Gu X, et al. Virulizin induces production of IL-17E to enhance antitumor activity by recruitment of eosinophils into tumors. Cancer Immunol Immunother (2008) 57:1757–69. doi:10.1007/s00262-008-0502-9
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
226. Benatar T, Cao MY, Lee Y, Lightfoot J, Feng N, Gu X, et al. IL-17E, a proinflammatory cytokine, has antitumor efficacy against several tumor types in vivo. Cancer Immunol Immunother (2010) 59:805–17. doi:10.1007/s00262-009-0802-8
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
227. Stockmann C, Schadendorf D, Klose R, Helfrich I. The impact of the immune system on tumor: angiogenesis and vascular remodeling. Front Oncol (2014) 4:69. doi:10.3389/fonc.2014.00069
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Keywords: chemokines, growth factors, allergy, asthma, inflammation, helminths, cancer
Citation: Davoine F and Lacy P (2014) Eosinophil cytokines, chemokines, and growth factors: emerging roles in immunity. Front. Immunol. 5:570. doi: 10.3389/fimmu.2014.00570
Received: 29 August 2014; Accepted: 24 October 2014;
Published online: 10 November 2014.
Edited by:
Uday Kishore, Brunel University, UKReviewed by:
Giovanna Schiavoni, Istituto Superiore di Sanità, ItalyJaya Talreja, Wayne State University, USA
Copyright: © 2014 Davoine and Lacy. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Paige Lacy, Department of Medicine, 559 Heritage Medical Research Centre, University of Alberta, Edmonton, AB T6G 2S2, Canada e-mail:cGFpZ2UubGFjeUB1YWxiZXJ0YS5jYQ==
 Francis Davoine
Francis Davoine Paige Lacy
Paige Lacy