Abstract
An unprecedented global social and economic impact as well as a significant number of fatalities have been brought on by the coronavirus disease 2019 (COVID-19), produced by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Acute SARS-CoV-2 infection can, in certain situations, cause immunological abnormalities, leading to an anomalous innate and adaptive immune response. While most patients only experience mild symptoms and recover without the need for mechanical ventilation, a substantial percentage of those who are affected develop severe respiratory illness, which can be fatal. The absence of effective therapies when disease progresses to a very severe condition coupled with the incomplete understanding of COVID-19’s pathogenesis triggers the need to develop innovative therapeutic approaches for patients at high risk of mortality. As a result, we investigate the potential contribution of promising combinatorial cell therapy to prevent death in critical patients.
1 Introduction
Severe acute respiratory syndrome coronavirus 2, or SARS-Cov-2, is a new coronavirus originally discovered after an outbreak of respiratory illness called COVID-19 in the Chinese city of Wuhan, Hubei (1). The infection of this virus quickly transitioned from being an isolated epidemic in a Chinese region to becoming a global health emergency of global alarm, and eventually a worldwide pandemic, due to the accelerated number of infections and fatalities that happened first in China and afterwards over the world. On March 11, 2020, given the rapid and progressive expansion of the epidemic internationally, the World Health Organization (WHO) declared a state of pandemic.
Coronaviruses belong to a broad virus family and happen to be the primary cause of common cold and some other diseases such as Middle East respiratory syndrome (MERS-CoV) and severe acute respiratory syndrome (SARS-CoV) (2, 3). They are encapsulated, and exhibit a spherical shape with the longest single-strand positive-sense RNA genome amid RNA viruses (4). Their name is due to a spike protein in their surface that resembles a crown.
SARS-CoV-2 is mostly spread through respiratory particles that are emitted when an infected individual sneezes, speaks or coughs. These particles, both small and large in size, tend to be concentrated within a short distance, the probability of transmission decreases with barrier methods as masks, physical separation and increased ventilation (5, 6).
Initial studies showed that SARS-CoV-2 infection can manifest across a wide clinical spectrum, ranging from asymptomatic infection to critical illness; for individuals who experience symptoms, the median incubation period of SARS-CoV-2 infection is typically around 4 to 5 days, and within an 11-day timeframe following infection, approximately 97% of individuals are likely to experience symptoms (7). Obesity, cardiovascular disease, chronic lung disease, diabetes, and advanced age are among the main risk factors associated with the progression of severe COVID-19 (6, 8, 9). Depending on the degree of severity of the clinical manifestations, the picture ranges from medium/moderate to severe and critical (Table 1). Clinical or radiographic evidence of lower respiratory tract disease, combined with a blood oxygen saturation level of 94% or above while the patient is breathing ambient air, indicates moderate disease. Severe disease can be identified by lung infiltrates (affecting more than 50% of the lung field within 24 to 48 hours), tachypnea (respiratory rate of 30 breaths per minute), hypoxemia (oxygen saturation of 93% or lower; ratio of partial pressure of arterial oxygen to fraction of inspired oxygen of 300), and tachycardia (heart rate exceeding 100 beats per minute) (6, 10) and patients can progress to Adult Respiratory Distress Syndrome (ARDS), which is a common immunopathological feature of severe COVID-19, SARS-1-CoV and MERS-CoV and is caused by an aggressive inflammatory response that can lead to respiratory difficulties and death (Figure 1).
Table 1
| Mild to moderate | Severe to critical |
|---|---|
| Fever | Immune Dysfunction |
| Dry cough | Lymphopenia |
| Smell/taste loss | Sustained inflammation |
| Headache | Secondary bacterial/fungi infection |
| Dizziness | ARDS (Acute respiratory distress syndrome |
| Nausea | Thrombosis |
| Diarrhea | Multiorganic damage (liver, kidney, myocardial) |
| Muscle/Joint pain Respiratory distress |
Clinical spectrum of infection severity.
Figure 1

Flow Chart of clinical outcome and epidemiology distribution of COVID-19 patients. Percentages are dependent on variants, country, age and other morbidities.
Throughout initial phases of the pandemic, approximately 17% to 35% of COVID-19 hospitalized patients were treated in an Intensive Care Unit (ICU), most likely due to hypoxemic respiratory worsening (11). Typically, children experience less severe symptoms primarily affecting the upper respiratory system, and hospitalization is rarely necessary, accounting for 2% to 5% of individuals who have been confirmed to have COVID-19 through laboratory testing exhibit an unclear susceptibility to the virus, as it is not well understood why they are less prone to contracting it, plausible causes include partial immunity from preceding viral exposures, lower exposure rates, and less strong immune responses, such as no cytokine storm (CS). Though the large percentage of pediatric cases are mild, a small proportion (7%) of hospitalized children, develop severe disease that requires mechanical ventilation (12).
2 High mortality risk COVID-19 patients: size of the niche
The evaluation of COVID-19 is based upon disease severity. Although severe sickness can affect anyone, most individuals with serious illness have at least one risk factor, several comorbidities and underlying diseases that eventually will progress into admission to the ICU, intubation, mechanical ventilation and death). Elder patients with comorbidities typically have severe to critical COVID-19 (13, 14). In a report from the Chinese Centre for Disease Control and Prevention (15), rates of case fatalities were respectively, 8% for those who age 70 to 79 year old and 15% for those aged 80 or older. This data contrasts to the 2.3% overall cohort case fatality rate (10), indicating that while symptoms of infection in kids and teenagers are often moderate, a tiny percentage do have severe and even fatal illness (16).
In a study of approximately 300,000 confirmed COVID-19 cases recorded in the United States, patients with documented co-morbidities exhibited a mortality rate that was 12-fold higher compared to those without such underlying conditions (17). Furthermore, the COVID-19 vaccine significantly decreases the risk of developing a severe disease and is linked to a lower fatality rate. Roughly, clinical features and distribution of COVID-19 population can be seen on Figure 1. Since a high proportion of SARS-CoV-2 infections are asymptomatic and mild infections go mostly undetected (18), the infection mortality rate has been estimated in many studies to be between 0.15% and 1%, with substantial variation by region and among different risk categories (19, 20).
As of May 31st 2023, 689,549,946 people have been diagnosed with COVID-19, either by PCR assessment or any other available assay. Total death reported rises up to 6,884,636, close to 1% (21).
The most prevalent clinical signs of COVID-19 include dyspnea, dry cough, and fever. Typically, 2 days to 2 weeks after viral contact, symptoms start to appear (22). Previous studies conducted on 181 COVID-19 confirmed cases outside Wuhan, China showed that 5 days after initial exposure, symptoms started to occur, and 97% of people began to experience symptoms 11 days after infection (7). Additional signs experienced by some patients include ageusia, anosmia, myalgia, sore throat, fatigue, diarrhea, and headache (23, 24). While patients may initially present with chills and respiratory symptoms without fever, in later stages of the disease, dyspnea can progress to ARDS or multiple organ failure (8).
Since the initial phases of the pandemic, it was clear that the diversity in symptoms, age, genetics, geographic location and morbidity play dissimilar roles in viral transmission and disease spectrum. Complex biochemical and immunological studies are required to understand the genetic implications underlying severe COVID-19. Similar to other RNA viruses, SARS-CoV-2 undergoes continuous evolution through random mutations and therefore, those new mutations could affect its infectivity and virulence. Moreover, the ability of the virus to elude adaptive immune responses from previous SARS-CoV-2 infections, specific antivirals and antibodies may also amend the efficacy of vaccines or increase reinfection risk (25).
According to seroprevalence surveys (26–28), more than 33%, and even more than 50% of the global population will be infected with SARS-CoV-2 by 2022 (29, 30). With so many people still infected, understanding the duration and effectiveness of immunity is crucial for future considerations regarding protection against reinfections and severe disease.
Several variants of SARS-CoV-2 mutants, which display certain characteristics, such as increased virulence or transmissibility, have been acknowledged so far, Alpha (B.1.1.7), which was first seen in the United Kingdom, Beta (B.1.351), initially discovered in South Africa; Gamma (P.1), discovered in Manaus, Brazil; and Delta, first discovered in India and became dominant in July 2021 (31, 32).
In November 2021, the Omicron (B.1.1.529) variant was classified as a Variant of Concern (VOC) and rapidly emerged as the predominant variant worldwide. This VOC is more contagious than previous variants which, in fact, have largely disappeared worldwide (33). The clinical relevance, emergence and transmission of these new variants evolves quickly, especially for how it may affect the effectiveness of vaccines and the rates of transmission as well as effectiveness of current therapeutics (34). With the introduction of the Omicron variant, the fraction of completely asymptomatic cases may increase even further. Due to its increased transmissibility and the occurrence of steep epidemic waves, it is anticipated that the number of infected individuals will rise significantly with the emergence of the Omicron variant (35, 36).
Moreover, those individuals with a higher death rate (WHO grade 8), are typically male, older than 65, and show concomitant comorbidities such as obesity, high blood pressure, cardiovascular illnesses, cancer, type 2 diabetes, smokers, etc. Characteristics that can be used as early warning signs of a High Mortality Risk (37), as well as non-vaccinated people or immunocompromised patients that might result in a poor response to the COVID-19 vaccination. Even more, a previous study has identified three phenotypes among COVID-19 patients who were admitted to the hospital based on their age and sex, underlying medical conditions, clinical and laboratory data, as well as radiological features at the time of presentation. The phenotypes have therapeutic implications despite not being intended for use in mortality prediction, and relationships with patient prognosis were identified, leading to the development of a simplified probabilistic model that may be relevant to other cohorts. Phenotype A was typical for young women with mild respiratory symptoms and normal inflammatory parameters, phenotype B included mainly obese patients, lymphopenia and moderately high inflammatory values. Finally, phenotype C patients were older, with higher inflammatory parameters and more comorbidities (38). Furthermore, the International Severe Acute Respiratory and Emerging Infection Consortium (ISARIC) has published an assessment of patients’ mortality risk based on a cohort of more than 35.000 patients (39), with 8 elements readily available at admission (age, respiratory rate, peripheral oxygen saturation, sex, level of consciousness, number of comorbidities, C reactive protein and urea level).
2.1 SARS-CoV-2 virus entry and effects
Coronaviruses have an enveloped, positive sense single-stranded RNA genome. The five main proteins are the spike protein (S), membrane glycoprotein (M), an additional membrane glycoprotein (HE), nucleocapsid protein (N), and a small membrane protein (SM). The S protein belongs to the group I fusion glycoproteins. It has a homotrimeric structure with a single top conformation and two lower conformations. The spike is made up of two subunits, the N′-terminal S1 and the C′-terminal S2, which are each in charge of a specific function, such as interacting with the host cell or engulfing a virion, respectively. RNA genome is hold within the nucleocapsid while the S-protein, membrane proteins and envelope constitute together the viral envelope (40).
Coronavirus spike proteins are class I fusion proteins that resemble the envelopes of other viruses such as human immunodeficiency virus (HIV) or hemagglutinin of influenza species (41). The SARS-CoV-2 virus uses the angiotensin-converting enzyme II (ACE2) receptor as a cellular entry (42), which is widely distributed in type II alveolar cells and as well as a small proportion of monocytes and macrophages, the capillary endothelium of the lungs and many other organs, including the hepatic, cardiovascular, gastrointestinal, and renal systems which also express ACE2 (43). ACE2 variant N720D may also enhance SARS-CoV-2 infectivity (44), and show more affinity of Furin to the mutated D614G S-protein of the virus (45). As soon as the viral spike protein attaches to the ACE2 receptor of the host cell membrane, the virus enters into host cells. Changes on the S-protein after docking grants the virus entrance to the endosomal pathway, virus RNA is then translated into viral components (46) in such a way that structural proteins are produced by the infected cell and their fragments exposed extracellularly by the Human Leukocyte Antigen (HLA)-II to the Antigen Presenting Cell (APC) (Figure 2).
Figure 2
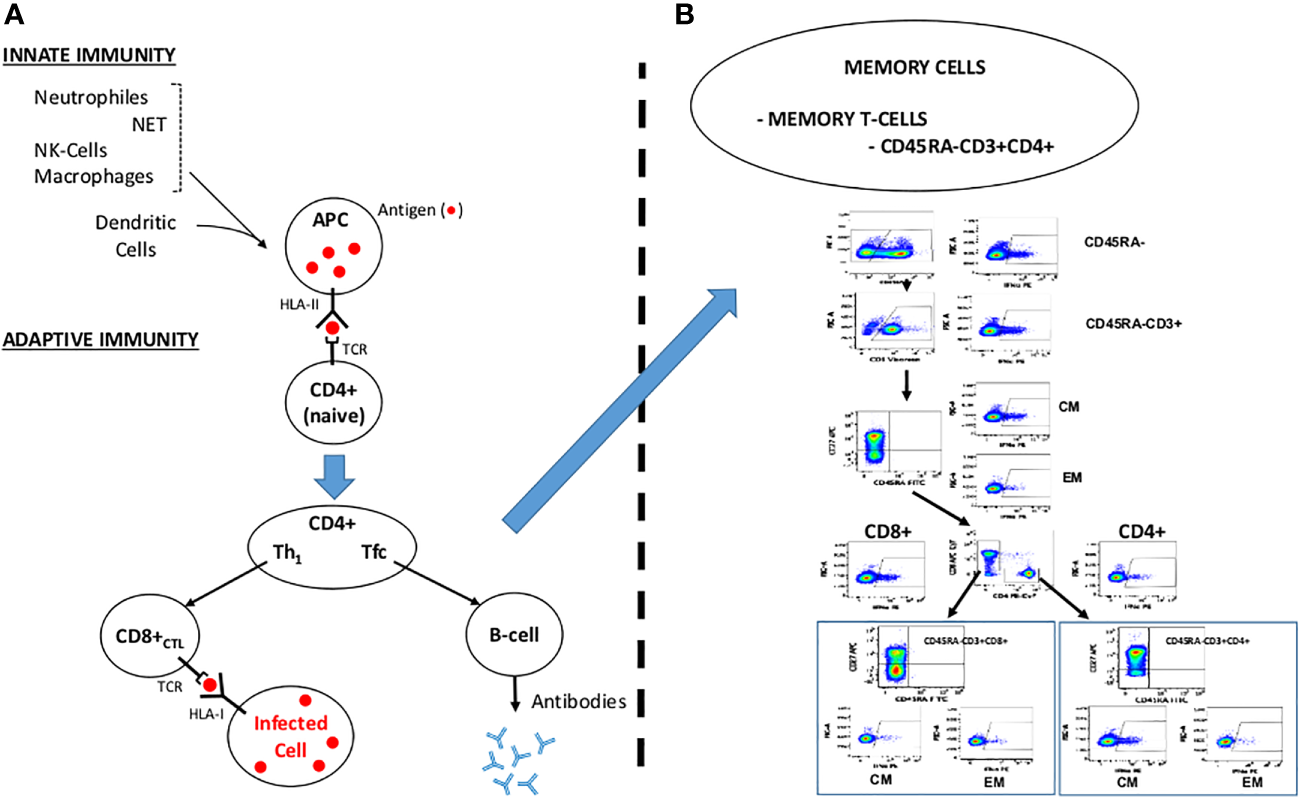
Overview of the Immune System Response. NET: Neutrophile Extracellular Traps. APC: Antigen Presenting Cell. NK: Natural Killer. TCR: T-cell Receptor. HLA-I/HLA-II: Human Leukocyte Antigen I and II. Th1: T Helper 1 Lymphocyte. Tfc: Follicular T Helper Cell. CD8+ CTL: Cytotoxic T Lymphocyte. (A) Course for a normal immune response engulfing innate and adaptive immunity. (B) Memory T-cells (modified from Ferreras et al, 2021). CD45RA-: CD45RA isoform depleted cells. CM: Central Memory T-cells. EM: Effector Memory T-cells.
2.2 Dysfunctional immune response
In the case of SARS-CoV-2 infection, severe disease is partially generated by the trigger of an unbalanced immune response. Innate immune cells must identify the virus’s invasion in order to stop the viral attack. This is done by pathogen-associated molecular patterns (PAMPs). Following the identification of viral genetic material, type I interferon (IFN) production is stimulated, and its signaling cascade activates important genes to inhibit viral replication and the development of a potent adaptive immune response (Figure 2A).
After an infection occurs, nasal epithelial cells increase the production of secreted immunoglobulins. In cases of severe COVID-19 infection, this natural mucosal defense mechanism can be exploited to harm the host by boosting the expression of pro-inflammatory cytokines (CS). Viral infections stimulate the enhancement of IFN regulatory genes (IRF3 and IRF7), which subsequently elevate the synthesis of type I IFN (47), in fact, nasal epithelial cells exhibit robust upregulation of both interferon I and III as their primary antiviral reaction (48). Type I IFNs play a vital role in regulating viral replication and bolstering the innate immune response (49).
The first barrier, the innate immunity, for any infection includes macrophages, neutrophils and NK-cells, mostly on the oropharyngeal mucosa. In 60-80% of the cases (asymptomatic) (18, 50–56), this barrier stops the virus and kills the virion-infected cell transmitting the message to the adaptive immune system. The HLA-II molecules of the antigen-presenting cells (APCs, mostly macrophages and dendritic cells) presents to the lymphocyte T-receptor (TCR) of the CD4-helper cells the antigens (“peptides” product of the hydrolysis of the S, M and N proteins) and activate into T-helper cells (Th): Th1 (in peripheral tissues) and follicular T-helper cells (Tfc, secondary lymphoid organs) which will, in turn, activate both the B-cells, which produce neutralizing antibodies and CD8+ cytotoxic cells, that kill virion infected cells. In convalescent patients recovered from COVID-19, memory NK-cells and B-cells remain in the body (in peripheral blood and lymphoid organs) ready to respond when these “foreign” antigens enter into the body (57).
Following the attachment of SARS-CoV-2 to the target cell, there is a possibility of over activation of the innate immune system and its associated cells, such as dendritic cells, macrophages, and granulocytes. The humoral and cellular immune system is subsequently activated by the complex of pro-inflammatory cytokines secreted by these cells. The immune system overreacts as a result of B-cell activation and antibody hypersecretion, causing tissue damage. Cytokines secreted from macrophages also recruit neutrophils and monocytes that penetrate the infection site too severely, damaging lung tissue and aggravating clinical symptoms. Figure 2 summarizes a simplified example of the physiological response of the immune system. Memory T-cells can be subdivided into Central memory T-Cells CDRA-CCR7+, characteristic of the homing of T cells to the lymph nodes and mucosal tissues and effector memory T-Cells CD45RA-CCR7- that migrate to peripheral tissues. Our group has previously identified SARS-Cov-2 specific memory T-Cells, Figure 2B, modified from Ferreras et al. (58).
Figure 3A depicts the dysfunctional SARS-Cov-2 induced immune response with CD8+ and CD4+ exhaustion (lymphopenia) and Macrophage over activation (Tumour Necrosis Factor-α and CS). Other effects of the virus on the immune system, endothelia and tissue damage are thoroughly described.
Figure 3
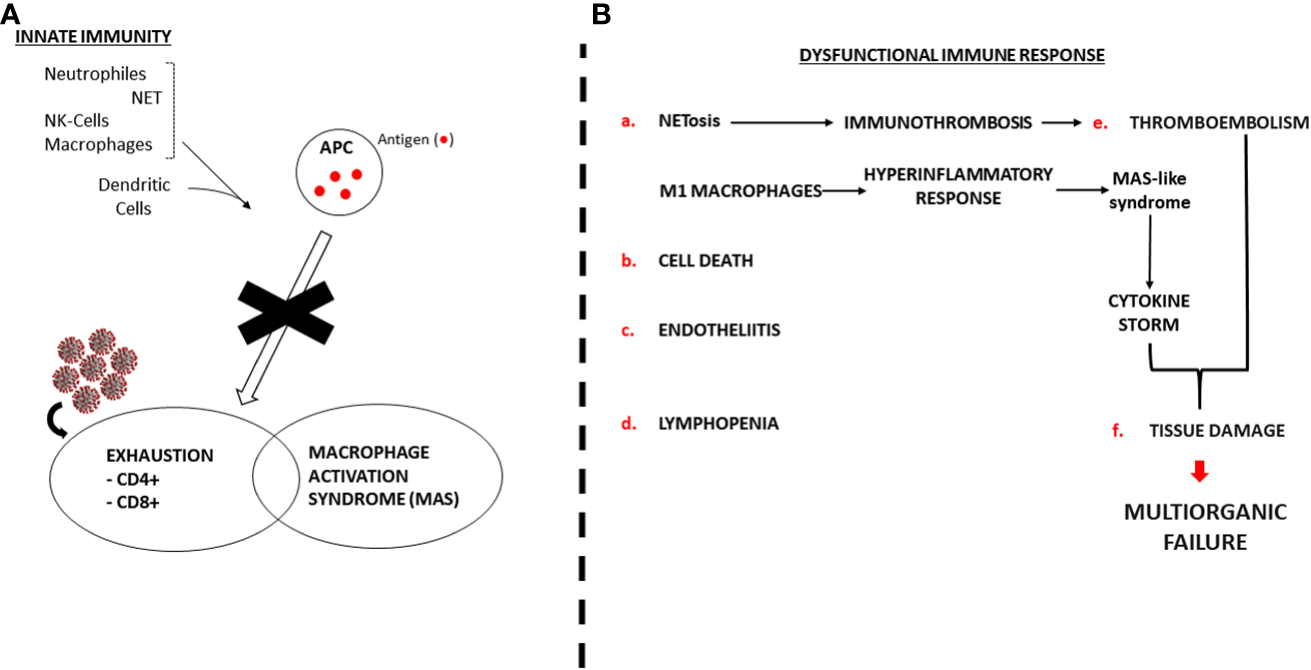
Dysfunctional SARS-Cov-2 induced immune response. NET: Neutrophile Extracellular Traps. APC: Antigen Presenting Cell. MAS: Macrophage Activation Syndrome. NK: Natural Killer. (A) SARS-Cov-2 blocks normal immune response. (B) Pathogenic mechanism of the dysregulated immune response.
2.2.1 NETosis and pyroptosis
In addition to engulfing bacteria, producing reactive oxygen species, degranulating, and secreting antimicrobials, neutrophils can also destroy invading pathogens, including viruses, by forming Neutrophile Extracellular Traps (NETs). NETs are extracellular fiber networks made mostly of neutrophil DNA that attach to and eliminate extracellular pathogens with little harm to the host cell. By harming endothelium cells, one of SARS-CoV-2 primary targets, neutrophils can also contribute to systemic viral dissemination (59–62). T-helper (Th) 1 cells and intermediate CD14+, CD16+ monocytes produce proinflammatory cytokine profiles. This is followed by neutrophil and macrophage infiltration into lung tissue, resulting in a CS (63). Rapid activation of pathogenic Th1 cells induces secretion of proinflammatory cytokines, such as interleukin-6 (IL-6) and granulocyte-macrophage colony-stimulating factor (GM-CSF). GM-CSF also activates CD14+ CD16+ inflammatory monocytes, causing them to produce vast quantities of IL-6, Tumour Necrosis Factor-α (TNF-α), and other cytokines (64, 65). IL-6, which is primarily linked to macrophages and dendritic cells (DCs), is one of the deleterious cytokines in COVID-19 linked with severe clinical conditions (66). This affiliation not only highlights the significant role of APCs in the development of the infection but also the widespread involvement of the innate immune system (67).
Once SARS-CoV-2 has entered the cell, rapid viral replication may result in significant vascular leakage and endothelial and epithelial cell death, which in turn triggers the production of a large amount of proinflammatory cytokines and chemokines and, as a direct result, cell pyroptosis (68).
Pyroptosis, a extremely inflammatory and Caspase-1-dependent form of programmed cell death, is a common reaction to intracellular pathogen infection and is a component of the immune system’s fight against infection. Interleukin (IL)-1β, which is released during pyroptosis, is raised in SARS-CoV-2 patients (8). Along with its beneficial effects on immune cell migration to inflamed tissues, IL-1β also increases on SARS-Cov2 and is largely released by macrophages through apoptosis and pyroptosis. In addition to IL-1βs beneficial effects on initiating and maintaining inflammation, NLRP3-IL-1 signaling has also been revealed to play a role in the COVID-19 CS. IL-1β, primarily released by macrophages through apoptosis and pyroptosis is elevated during SARS-Cov2 infection. It contributes positively to the recruitment of immune cells to inflamed tissues. Additionally, IL-1β is involved in initiating and sustaining inflammation, it has been hypothesized that NLRP3-IL-1β signaling also plays a role in the development of CS observed in COVID-19 (69).
NLRP3 is an innate immune system component that behaves as a pattern recognition receptor (PRR) that acknowledges PAMPs. There are several studies that suggest that IL-1β may contribute to COVID-19 CS in coronavirus infections (70, 71). In addition, alveolar epithelial cells and macrophages recognize pathogen-associated molecular patterns (PAMPs), damage-associated molecular patterns (DAMPs), and antibody-secreting cell (ASC) oligomers. This recognition leads to the release of proinflammatory chemokines, interferons, and cytokines, which attract immune cells, particularly T-cells and monocytes, from the bloodstream into the lungs affected by the infection (72), which might clarify the lymphopenia detected in the majority of the patients with SARS-CoV-2 infection (73). Additionally, SARS-CoV-2 induced pyroptosis among lymphocytes and macrophages exacerbates the lymphopenia in most of the observed patients (68).
In addition, SARS-CoV-2 infection, cell pyroptosis, and the resulting hyperinflammation might well induce NETosis, a controlled cell death process caused by neutrophils releasing NETs (74) that substantially lure and kill microbes as a mechanism of the innate immune response (75). However, if NETosis becomes dysregulated as it happens in acute and chronic inflammatory diseases (76), it can contribute to the pathogenesis of sepsis and ARDS, with NETs exacerbating multiorgan failure, microthrombi and vascular tissue damage (77). Preceding studies have described in severe cases of COVID-19, pathogenic immunothrombosis resulting from a dysregulated NET formation (59). COVID-19-related ARDS has been found to be linked to increased NET formation, and constitutes a potential marker of disease severity (78, 79).
Furthermore, the transmission risk is highly determined by the viral load (80) and viral antigens may well lead to severe disease and therefore induce a tougher antibody response (81). Previous studies in mice have showed that viral exposure dose defines likelihood of SARS-CoV-1 infection (80, 82) and SARS-CoV-2 research in golden hamsters were indicative of dose-dependent infection (83) (Figure 3). However, it is important to note that although patients admitted to ICU may exhibit a slower decline in viral load, the peak viral load of SARS-CoV-2 typically occurs within 5-6 days after the onset of symptoms (84), and does not vary between patients with mild and severe disease (85). A possible explanation could be that antibodies might exacerbate disease severity by antibody-dependent enhancement (ADE) as it happened with SARS-CoV outbreak in 2002 (86). Still, there is no present evidence backing this for SARS-Cov-2 (87). Moreover, SARS-CoV-2 RNA has been found in patients’ samples up until death, suggesting a clear correlation between poor disease outcome and viral load persistence (15).
2.2.2 Cell death and Sars-Cov-2
It is widely recognized that cellular apoptosis and autophagy are fundamental processes within cells, serving pivotal roles in upholding balance and influencing disease development (88). A growing body of research suggests that cell death and autophagy induced by coronaviruses could hold significance in virus infection and the development of disease. The occurrence of lymphopenia has been linked with various indicators of severity, including the presence of the death ligand, FasL (88). An increasing body of evidence highlights the dual nature of these processes in the context of viral infections (89, 90). On one side, these processes hinder the virus’s replication and transmission by clearing infected cells through cell death. Conversely, dysregulated cell death leads to uncontrolled cellular damage and disruption in immune responses. Concurrently, viruses exploit cell autophagy to their advantage, utilizing it for replication niches, immune evasion, and extracellular release (91).
Infection with SARS-CoV-2 in lung epithelial cells triggers both cell death and an inflammatory response (92), to counteract this phenomenon, the application of caspase inhibitors can inhibit apoptosis, leading to the preservation of CD4+ T-cells and reverse the process of lymphocyte apoptosis (93).While early inflammatory responses are essential for constraining viral replication (94), coronaviruses have developed tactics to elude detection by the innate immune system by evading activation of pattern recognition receptors (PRRs) and disrupting downstream interferon (IFN) responses (95). Conversely, severe cases of COVID-19 are marked by an excessive inflammatory reaction both in the lungs and bloodstream (8, 96). In such cases, a blend of cytokines, especially TNF-α and interferon gamma (IFN-γ), trigger a form of inflammatory cell death termed PANoptosis, subsequently resulting in a CS (96, 97).
2.2.3 Endotheliitis
Recent research indicates that endothelial damage and the resulting morphological and functional alterations in the endothelium are significant contributors to COVID-19-induced hyperinflammation. COVID-19 is expected to have a greater effect on the lungs, as they are the first organs to become infected and regenerate very slowly. Previous research suggest that SARS-CoV-2 infection causes a disproportionate immune response, known as a COVID-19 CS in severe COVID-19 cases (98, 99). Post-mortem histology from three patients with late-stage COVID-19 showed viral inclusions in microvascular lymphocytic endotheliitis and endothelial apoptotic cells, with infiltration of inflammatory cells around the vessels and endothelial cells, as well as signs of endothelial apoptotic cell death in the kidney, lung, heart and small bowel (100). Furthermore, recent research has found indications of endothelial glycocalyx disruption in the plasma and serum of 19 critically ill COVID-19 patients (101). This specific result is noteworthy because the integrity of the endothelial glycocalyx, which covers the luminal surface of endothelial cells, is essential for the preservation of vascular homeostasis (102). According to these results, SARS-CoV-2 infection has been observed to directly contribute to the development of endotheliitis in various organs. This involvement is evidenced by the presence of viral bodies within endothelial cells and the accompanying inflammatory response of the host. Additionally, in individuals with COVID-19, activation of apoptosis and pyroptosis may play a significant role in the damage to endothelial cells. The systemic decreased microcirculatory performance in various arterial beds and its clinical consequences in COVID-19 patients might be stated through COVID-19-endotheliitis (100).
The stabilization of the endothelium while preventing viral replication, primarily with anti-inflammatory anti-cytokine drugs, ACE inhibitors, and statins, this aspect of SARS-CoV-2 infection and the development of endotheliitis becomes particularly significant for vulnerable patients who already have pre-existing endothelial dysfunction. Factors such as male sex, hypertension, smoking, obesity, established cardiovascular disease, and diabetes, which are associated with endothelial dysfunction, are also known to be linked with unfavourable outcomes in COVID-19 (103–105).
2.2.4 Lymphopenia
Previous clinical findings indicate that lymphocyte measurements including B cells, natural killer (NK), CD8+ cytotoxic T and CD4+ T cells were all reduced in COVID-19 patients (106, 107). A thorough study of B and T cell populations in COVID-19 patients found a link between increased disease severity and lower numbers of CD8+ and CD4+ T cells. Furthermore, the drop in CD8+ T cells was greater in cases with milder illness than the decrease in CD4+ T cells (108). Mechanisms involved in lymphocyte depletion induced by SARS-CoV-2 might be due to direct T-cell infection via ACE2 receptor, resulting in T-cell death (109). The depletion and exhaustion of T-cells with their respective function can be speeded by a number of pro-inflammatory or anti-inflammatory cytokines, and the virus may kill secondary lymphoid tissues like lymph nodes and the spleen (110).
A major contributing component to the observed lymphopenia is probably due to the inflammatory CS. TNF-α and IL-6 levels in the serum have been found to be strongly linked with lymphopenia, whereas levels in the serum of healed patients are nearly normal. Massive lymphocyte death was discovered during autopsy examinations on lymphoid organs taken from numerous individuals who passed away from the condition; this death was ascribed to high levels of IL-6. Tocilizumab, an IL-6 receptor antagonist, treatment increased the number of circulating lymphocytes, further indicating that an increase in IL-6 is a major factor in the development of lymphopenia (111). Even more, treatment of critical ICU patients with intubation, mechanical ventilation and extracorporeal membrane oxygenation (ECMO) with adipose-tissue derived Mesenchymal Stromal Cells (MSCs) increase CD4+, CD8+ and B-cells, suggesting that inflammation synergizes with other mechanisms causing lymphopenia (112). This sequence of inflammatory events originates from the liberation of pro-inflammatory cytokines, notably IL-18 and IL-1β. This phenomenon elucidates the notable feature of commonly observed neutrophilia and leukopenia (113). Overall, the harshness of the disease has been closely linked to lymphopenia and hyper-inflammatory response.
2.2.5 Thromboembolism
Early findings indicated greater incidence of venous thromboembolism (VTE) in individuals with severe COVID-19 disease compared to data from similar patients not afflicted by SARS-CoV-2 in addition to respiratory problems. Patients with COVID-19 usually experience coagulation problems, and those with severe sickness frequently have high levels of coagulation markers such D-dimer and fibrinogen degradation products. Along with disseminated intravascular coagulation, severe thrombocytopenia and lymphopenia have also been linked to worse outcomes (114). As a result of the thromboinflammation brought on by COVID-19, medical research has also shown that patients with COVID-19 have an increased risk of developing pulmonary embolism (PE). In COVID-19, thromboinflammation appears as increased levels of procoagulants (like von Willebrand factor) and endothelial dysfunction, which reduces the endothelium’s protective antithrombotic action (115, 116).
2.2.6 Tissue damage
It has also been shown that SARS-CoV-2 principally affects the lung by generating diffuse alveolar destruction with ARDS, yet the recent research has revealed that the virus also has a negative influence on further important organs and tissue, such as the heart, brain, large intestine, kidneys, and spleen (117–120). In COVID-19 patients, there is a link between inflammation and serious organ damage. Diffuse alveolar injury, including damage to hyaline membranes, is the main pathophysiology in ARDS. Pneumocytes’ viral cytopathic effect suggests that there has been direct viral harm (121).
Emerging evidence suggests that SARS-CoV-2 has the capability to efficiently propagate and replicate in various organs, including the lungs, brain, heart, spleen, liver, and gastrointestinal tract. This is supported by findings on the virus’s route of entry and distribution, as well as the presence of infective RNA in these organs. These findings indicate the need to explore alternative mechanisms of cellular entry, such as non-receptor mediated endocrine entry, in addition to the conventional understanding of cellular entry through the ACE2 receptor.
2.3 T-cell and B-cell disorders
The resolution of a viral infection encompasses both cellular and humoral immune reactions. The humoral immune response, too, assumes a vital role in eradicating the virus within the host. The antibody responses specific to SARS-CoV-2 exhibit differing levels and characteristics between individuals with asymptomatic infections and those experiencing severe disease.
B cells play a crucial role by producing antibodies and facilitating the humoral immune response, rendering them highly significant in establishing protective immunity against SARS-CoV-2 (122), in addition T cells differentiate into cytotoxic T lymphocytes (CD8+) and helper T-cells (CD4+) engaging with the COVID-19 infection through distinct interactions (123).
Sustained defense hinges on the expansion of memory T cells and B cells tailored to SARS-CoV-2. Notably, follicular T-helper cells drive the humoral immune reaction, fostering a reservoir of specialized memory B cells primed to respond swiftly if confronted with a potential reinfection (124). The cytotoxic program in CD8+ T cells persists, type 1 cytokines and interleukin-17 (IL-17) production rise in T cells from patients who are in the recovery phase. It’s interesting to note that B cells from people with acute COVID-19 showed an imbalance between the cytokines IL-6 and IL-10 in response to the activation of Toll-like receptors, leaning toward a pro-inflammatory profile. Regardless of the clinical results, the frequency of IL-6+ B cells reverted to normal in convalescent patients, but the recovery of IL-10+ B cells was associated with the resolution of lung problems (125).
3 COVID-19 time-course
COVID-19 shows several phases. During the early infection phase, usually mild symptomatology is reported such as, fever, cough, headache, etc. Laboratory abnormalities are lymphopenia, D fragments of the fibrin protein (D-dimer), low LDH, etc. It is also characterized by an increased viral load, which encourages a conservative treatment based on antiviral agents. In a second phase, the inflammatory response would predominate, leading to pulmonary inflammation or fibrosis, coagulopathies, and tissue damage and where anti-inflammatory drugs would have greater importance (Figure 4).
Figure 4
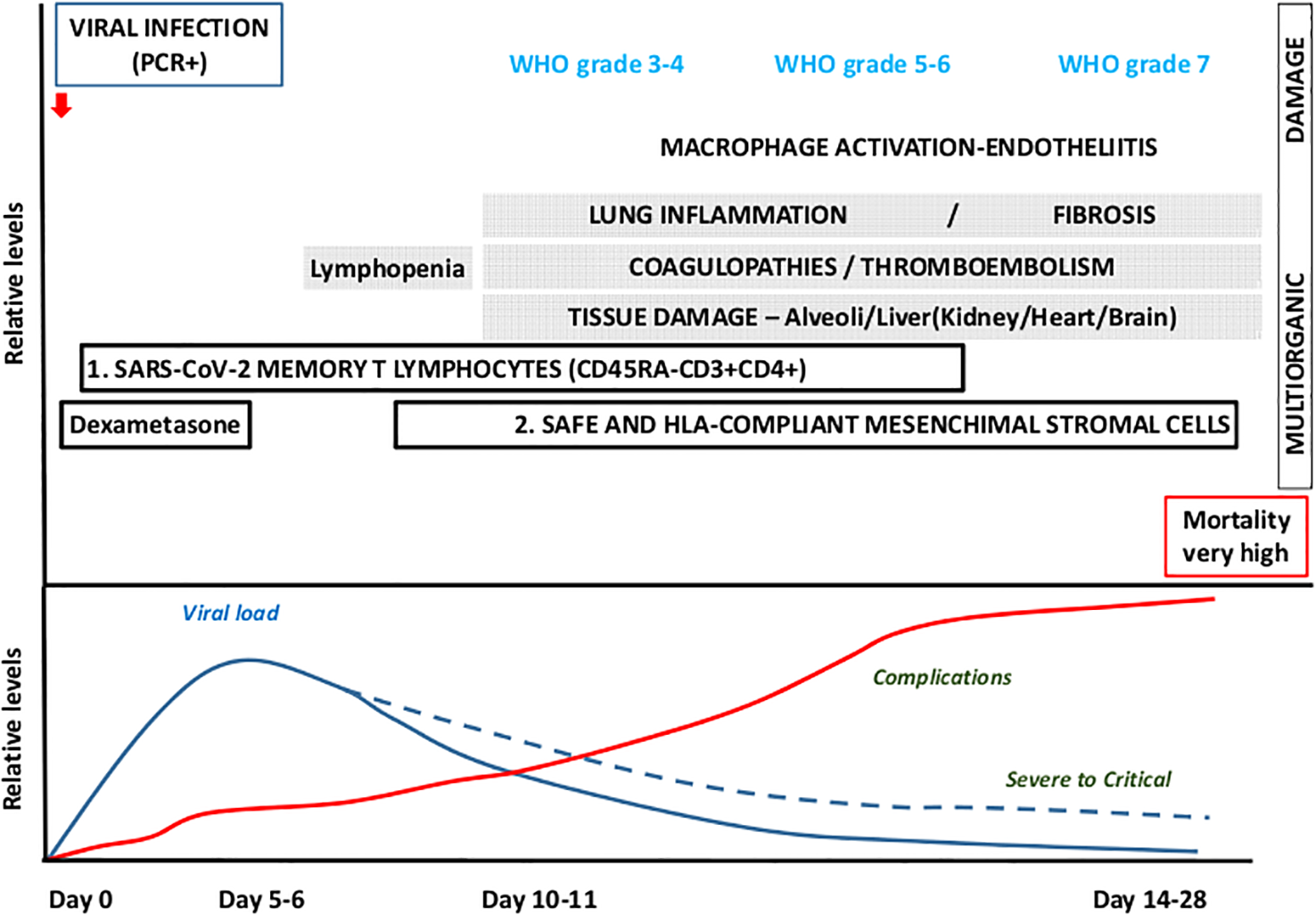
Time-course and clinical outcomes for COVID-19. Conservative treatment is being used for the first infection phase, which lasts up to 3–5 days and progresses with mild symptomatology and increased viral load, whereas in a small proportion of patients the second phase, 7 day onwards, yields extensive inflammatory response and an anti-inflammatory approach is recommended. Therapeutic proposal: Use SARS-Cov-2 memory CD45RA-CD3+CD4+ during the first phase (viral) and safe and HLA-compliant mesenchymal stromal cells for the second phase (complications).
SARS-CoV-2 clearance from the lungs and upper respiratory tract should be effective in the majority of COVID-19 patients as a result of the first cytokine release, activation of the antiviral interferon response, and recruitment of immune cells; nevertheless, an abnormal immune response could lead to a severe disease outcome. Certainly, the excessive production of inflammatory cytokines and chemical mediators, along with macrophage activation, endotheliitis, lung hyper-inflammation, coagulopathies, thromboembolism, and tissue damage, collectively contribute to a life-threatening response. This response is widely regarded as the primary cause of the severity of COVID-19 and can lead to fatalities in affected patients (126, 127). Furthermore, these life-threatening responses are associated with elevated levels of circulating cytokines, severe lymphopenia, thrombosis, and extensive infiltration of mononuclear cells in multiple organs (73, 128, 129).
We could roughly discuss the physiopathology and viral characteristics as follows; early infection, in which a viral infection results in an initial immune system depression that frequently results in significant lymphopenia (130). The virus infects ACE-2 receptor-containing cells (endothelial, alveoli, gut epithelia, kidney, etc.), causing a systemic endothelial inflammation that manifests clinically as fever, dry cough, and lymphopenia. This inflammation is more severe in patients with prior chronic endotheliitis (old age, obesity, atherosclerosis, hypertension, diabetes, etc.). In around 10–20% of infections, the virus is not stopped by the body’s first line of defense (NK-cells, neutrophils, macrophages, IFN-γ, etc.), the viral load rises, and pneumonia with lymphopenia occurs, requiring hospitalization (Figure 1). A hyper-inflammatory picture that mimics Macrophage Activation Syndrome (MAS), Graft-versus-Host Syndrome (GvHD), or secondary hemophagocytic lymphohistiocytosis is present in hospitalized patients along with symptoms such as respiratory distress, fever, hypoxia, etc.
Thromboembolism, tissue damage, and inflammation, a very severe systemic inflammatory phase requiring an intensive care unit develops in about 5% of infected patients. Unlike the SARS-1 coronavirus, the host reaction is highly robust and varied. The development of a major inflammatory phase (131), which leads to ARDS, is in fact one of the most obvious features of the physiopathology of pneumonia in COVID-19 illness (132) and a condition that is similar to the MAS (126, 133) and injury to tissues that might possibly require ECMO. Additionally, a progressive endothelial thromboinflammatory syndrome (with raised D-dimers levels) uncommon in other viral infections, aggravates the disease’s prognosis (107, 134).
4 Combinational therapies
Along with vaccine research and other strategies that obstruct viral entry or directly target the virus, therapies that treat the immunopathology of the illness are increasingly being prioritized. The use of monoclonal antibodies such as Sotrovimab or Tixagevimab-cilgavimab for modulating the inflammatory response can constitute an alternative therapeutic strategy. IFN, IL-1, IL-6, and complement factor 5a are all mediators that target the extreme inflammatory response that occurs after SARS-CoV-2 infection with the aim of avoiding organ damage (135–137) thus monoclonal antibodies aiming to block these factors, together with tyrosine kinase inhibitors are being used to block the cytokine storm-like response or their ability to avert pulmonary vascular leakage in people with COVID-19.
4.1 Convalescent plasma from donors
The earliest reports of treatments using plasma from patients who had recovered from viral illnesses were published during the 1918 flu pandemic. Clinical improvement was seen in all participants both before and after receiving convalescent plasma transfusion status, according to a preliminary study of 5 critically sick COVID-19 patients treated with convalescent plasma encapsulating neutralizing antibodies (138). Nevertheless a multicenter randomized open-label clinical trial for convalescent plasma in patients hospitalized with COVID-19 pneumonia (139) did not result in a clear protection.
4.2 Cytokine inhibitors
Alongside with the clinical symptoms linked to viral invasion, the CS has attracted the most interest, raising the hypothesis that anti-inflammatory treatments aimed at lowering interleukin-6 (IL-6), IL-1, or even TNF-α may be helpful. It has been established that increased plasma levels of cytokines and chemokines, IL-6 in particular, are significantly higher in severe than in mild to moderate disease allowing to predict COVID-19 severity and survival (140–142) and correlate with the harshness of the disease. Early reports showed augmented plasma concentrations of IL-6, therefore providing the introduction of anti-IL-6 therapies in randomized clinical trials (143, 144) therefore, novel cytokine inhibitors, including Baricitinib, Anakinra, and Tocilizumab, were likely options for treating severe COVID-19 which found appropriate niches (145–147).
Baricitinib, an inhibitor of Janus-Associated Kinases (JAKs), which belong to a family of intracellular, non-receptor tyrosine kinases that transduce cytokine-mediated signals via the JAK-STAT pathway and since and JAKs mediate actions of many pro-inflammatory cytokines, combines both antiviral and anti-inflammatory effects. JAKs inhibitors block the signal transduction pathways that activate the immune system and cause inflammation, therefore, constitute an attractive treatment approach to stop the development of more serious disorders (148), considering the significant role that this transduction channel had in the onset of the CS in COVID-19 (149).
It is clear that therapeutic approaches targeting cytokine responses, in addition to anti-viral medications, deserve special attention to reduce morbidity and mortality in COVID-19 patients, even though the mechanisms by which SARS-CoV-2 infection induces cytokine overproduction are not yet fully understood (150, 151).
4.3 Corticosteroids, dexamethasone
Patients with autoimmune and inflammatory conditions such asthma, systemic lupus erythematous, Crohn’s disease, and rheumatoid arthritis are given prescriptions for glucocorticoids. Systemic corticosteroids (such as hydrocortisone, dexamethasone, and methylprednisolone) are given orally or intravenously and are effectively dispersed throughout the body, showing a variable degree of relative anti-inflammatory effectiveness and mineralocorticoid effects. However, high doses inhibit the immune response.
Dexamethasone, a synthetic corticosteroid that has the ability of dampening the immune system, which discouraged its use during early phases of COVID-19 infection. RECOVERY Trial showed a beneficial outcome using low dose (6 mg/day) in severe patients, defined as those with oxygen saturation lower than 94% (152). Dexamethasone reduced deaths by one-fifth in other hospitalized patients receiving only oxygen, but there was no benefit seen in COVID-19 patients who did not require respiratory support. Patients who had symptoms for more than 7 days and needed mechanical ventilation benefited the most. On the other hand, there was no benefit (and potential harm) amid patients who had shorter symptom duration and no need for supplementary oxygen. Furthermore, it could be extremely dangerous during recovery because not only will the virus persevere, but the body will be prevented from producing protective antibodies (153, 154).
In summary, numerous randomized trials have found that systemic corticosteroid therapy improves clinical outcomes and drops mortality in COVID-19 hospitalized patients who need supplementary oxygen (155, 156), perhaps by dampening the systemic inflammatory response induced by COVID-19, which can result in lung injury and multisystem organ dysfunction.
4.4 Cell-based therapies
4.4.1 Memory T lymphocytes CD45RA-
The role of adaptive immunity in COVID-19 and the protective immunity conferred by T-cells and the role of memory T-cells in providing protection against SARS-CoV-2 has not yet been properly defined (58, 157, 158). Specific memory T-cells for other coronavirus has been found up to 11 years after infection (159), particularly in the context of allogeneic hematopoietic stem cell transplantation (HSCT). This immunological memory assigns a swift and fast secondary immune response that is decisive and forms the basis of adoptive cell treatment for viral infections in immunosuppressed patients. Infusion of CD45RA- memory T-cells reduces induced mortality and CD45RA- T-cells alloreactivity of GvHD in bone-marrow transplantation (160, 161).
Previous research suggested that CD45RA- memory T-cells from the blood of convalescent donors include a SARS-CoV-2 specific T-cell population that is easy, efficient, and quick to isolate and subsequently, may be able to clear virally infected cells and confer T-cell immunity (Figure 2) (58). CD45RA- memory T cells from convalescent donors harboring SARS-CoV-2 specific T-cells were infused intravenously in a Phase 1 research clinical trial to assess the safety and feasibility of adoptive cell treatment for moderate to severe COVID-19 cases (NCT04578210). Nine patients with lymphopenia and pneumonia who had at least one HLA Class I match with the donor were enrolled (Figure 5A). The CD45RA- memory T cells were administered to the first 3 patients at a low dose (1x105 cells/kg), the following 3 patients at an intermediate dose (5x105 cells/kg), and the last 3 patients at a high dose (1x106 cells/kg). There were no documented severe negative effects. Six days following the infusion, the patient’s clinical state improved as measured by the National Early Warning Score (NEWS) and a 7-category point ordinal. Following infusion, the median length of stay in the hospital was 8 days for the low dose group, 7 days for the intermediate dose group, and 4 days for the high dose group. Two weeks following the infusion, the inflammatory markers normalized and all displayed lymphocyte recovery. This study provides early evidence that the use of allogeneic CD45RA- memory T-cells from convalescent donors for the treatment of COVID-19 patients with moderate to severe symptoms is possible, safe, and related with rapid clinical improvement and brief hospital stays (162). Adoptive T-cell therapy has essentially no side effects, and the cell product can be preserved in a lymphocyte biobank to allow ready access in the event of future viral pandemics (58). Repeated infusion of CD45RA- cells from family donors have also been used to refractory viral and fungi infections in transplanted individuals (163).
Figure 5
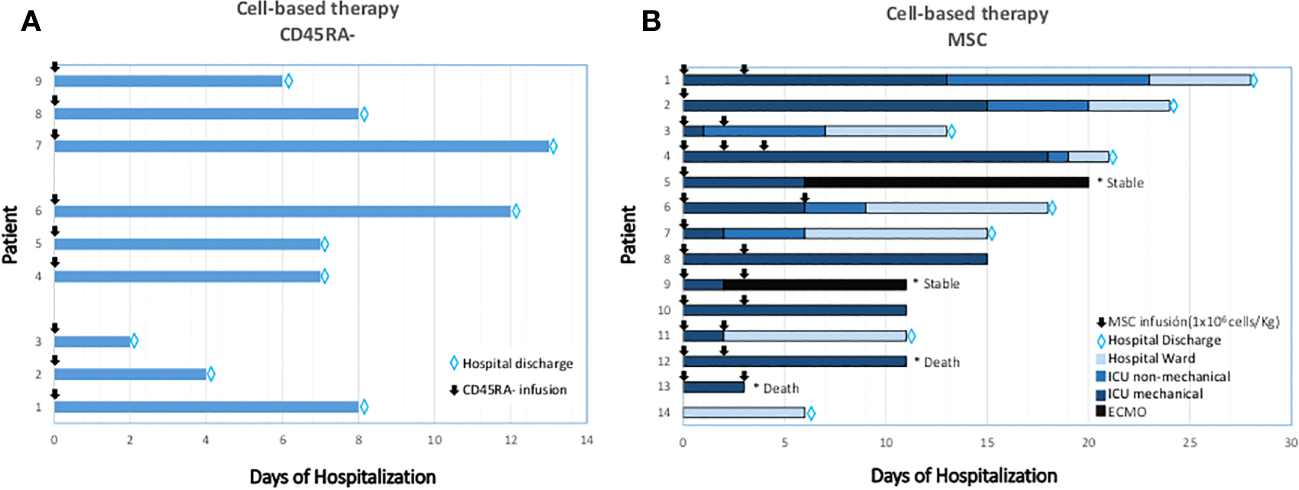
Clinical outcomes after CD45RA- (A) and MSC (B) treated patients. (A) T-cell memory based therapy (CD45RA depleted infused). Time-course and clinical outcome. (B) Cell-based therapy. Mesenchymal Stromal-stem Cell (MSC) infusions and timing are represented in arrows. In the X axis, days from the first MSC infusion are specified. Type of ventilation support is graded in colors through each row. MSC: Mesenchymal Stromal-stem Cells; ICU: Intensive Care Unit; ECMO: Extra Corporeal Membrane Oxygenation.
4.4.2 Mesenchymal stem-stromal cells
Advanced Therapy Medicinal Products (ATMPs) are cutting-edge therapies that include gene therapy, somatic cell therapy, and tissue-engineered products. Mesenchymal Stem-stromal cells (MSCs) are adult stem cells that have immunomodulatory, regenerative, and differentiation capabilities. Among them MSC have demonstrated to be safe in more than 400 clinical trials (Clinicaltrials.gov). As for October 31, 2022, MSC were proposed to be used in 37 clinical trials to treat COVID-19: 8 of them completed, 6 still recruiting, 7 active not recruiting, 4 suspended or withdrawn and the rest unknown.
MSC are multipotent non-hematopoietic progenitor cells with different degrees of stemness, derived from the mesodermal germ layer and resident in most of tissues (164). MSCs can be sourced from different types of tissues, including adipose tissue, Wharton’s jelly tissue, bone marrow, and amniotic fluid. They are able to differentiate in a wide range of cell types such as chondrocytes, osteocytes, neural cells, myocytes, and epithelial cells (165). The International Society for Cellular Therapy (ISCT) proposed the minimum criteria for the characterization of human MSC: (i) adherence to the plastic of the culture plate, (ii) adipogenic, chondrogenic and osteogenic differentiation capacity, and (iii) a specific profile of surface markers CD45+, CD90+ and CD73+, CD105+, HLADR-, CD11b- or CD14-, CD19- or CD79a- and CD34 (166, 167). Due to its unique biological properties including adhesion to plastic, easy expansion and culture, MSCs are the cell type mostly used in Cellular Therapy.
MSCs were first employed in humans as a cellular therapy in 1995, and they have since been used in basic research and clinical applications (168). The use of allogeneic MSCs in COVID-19 might prove useful due to its capabilities to diminish this clinical and biological picture of massive inflammation, and there are evidences of efficacy in GvHD (169, 170). Bone marrow-derived MSC appeared to be highly successful as a therapeutic option for ARDS on a phase I-II trial (171).
MSCs from the umbilical cord (UC-MSC) exhibit minimal immunogenicity and both immunoregulation and tissue-fixing abilities. They are an outstanding option for allogeneic adoptive transfer treatment because of this. It may be used to treat acute lung damage brought on by the H5N1 infection which shared a comparable inflammatory cytokine profile with COVID-19 (172–174). Mesenchymal Stromal-stem cell therapy might have the ability to avoid the release of cytokines via immune system, endorsing endogenous repair because of the regenerative properties of the stem cells. The MSC cells may aid in restoring lung microenvironment, protect alveolar epithelial cells, and treat pulmonary dysfunction and COVID-19 associated pneumonia (175). Furthermore, it has been shown that intravenous infusion UC-MSC as an adjuvant therapy to critically ill patients increases their chance of survival by inducing an anti-inflammatory state (176). The main reason for improvement of COVID-19 disease is the higher anti-inflammatory features of the MSCs after its intravenous provision. Furthermore, previous studies suggest that direct cell to cell mitochondrial transmission from MSCs to the alveolar epithelium and immune cells restricts the inflammatory response (177, 178). Among all stromal cells, Placenta-derived decidua stromal cells (DSCs) have been found to show a more robust immunosuppressive effect than other sources of MSCs. These DSCs display significantly stronger immunomodulatory and anti-inflammatory properties, making them successful in treating and managing steroid-refractory acute GvHD (179).
MSC therapy is thought to have the potential to prevent the excessive release of cytokines during an immune system response known as a cytokine storm. Additionally, MSCs possess reparative properties that can stimulate the body’s natural healing processes. These stem cells are capable of suppressing the proliferation of immune cells and modulating the functions of both innate and adaptive immune cells (180). Proinflammatory cytokines, such as TNF-α, IFN-γ, IL-1β, IL-2, IL-6, IL-8 and IL-17, signal over their receptors in MSC surface and stimulate biosynthesis of HGF, LIF, TSG-6, TGF-β, IL-10, and expression of superoxide dismutase (SOD), prostaglandin-E2 (PGE2), indoleamine-pyrrole 2,3-dioxygenase (IDO), nitric oxide synthase (iNOS, produced by murine cells) and cyclooxygenase-2 (COX-2) produced by human cells (181, 182). It has been shown that these molecules mediate the immunomodulatory and immunosuppressive properties of MSCs (180).
Our group was the first to use allogeneic MSC to treat critical mechanically ventilated COVID-19 patients (Figure 5B, Patients 1-13). Preliminary results of the BALMYS trial (NCT 04348461, suspended by lack of funding), indicate that critically ill patients with COVID-19 pneumonia can be safely administered with MSC derived from adipose tissue (AT-MSC) in and that administration was followed by clinical improvement and changes in inflammatory markers and recovery of immune populations (B-cells, CD4+ and CD8+ cells), these results suggest a potential biological effect of the cells. Only steroids were given concurrently when the cells were delivered. During their time in the ICU, the majority of patients received supportive care. This comprised normal treatments such as sedation and mechanical ventilation in addition to the use of vasopressors or inotropic medications, enteral or parenteral feeding, diuretics, and/or antibiotics. The findings imply that early adipose tissue-derived allogeneic MSC therapy during mechanical intubation may enhance the outcome (112). We also showed the absence of side effects following cell administration in these critically ill patients with respiratory failure, extensive inflammation, and prothrombotic risk. Allogeneic MSC was used to treat 13 adult COVID-19 patients who were receiving invasive mechanical ventilation and had previously taken antiviral and/or anti-inflammatory medications (including hydroxychloroquine, steroids, Tocilizumab, and/or Lopinavir/Ritonavir, among others) (112). Patient 14 was a Grade 4 (no-intubated, no-mechanical ventilation, that received bone-marrow derived mesenchymal stromal cells infused intravenously (183).
Nonetheless, pre-clinical evidence of the potential of MSCs is still limited and, even debatable (184, 185). MSCs are particularly useful because in addition of being anti-inflammatory and promoters of tissue regeneration they lacked ACE-2 Receptor (175) and the transmembrane protease serine 2 making them resistant to SARS-CoV-2 infection.
MSCs role plays through a paracrine mechanism, releasing biologically active substances known as the secretome (186) which is composed of soluble proteins, including a diverse range of growth factors, chemokines, and cytokines, as well as extracellular vesicles (EVs) (187) that are capable to interact with the target cells and modulate cellular responses. Because of its immunomodulatory, pro-angiogenic, anti-protease, regenerative, and anti-inflammatory properties, MSC-secretome could prove as a promising cell-free therapeutic tool both for acute and chronic lung diseases.
MSC therapies act by modulating responses in inflammatory diseases (188), migrate to sites of injury and inflammation in which they can exert their immunomodulatory effects, through a paracrine mode of action (189). Its immunomodulatory activity derives from their capability to in-vitro suppress proliferation and action of B, T and NK-cells, as well as dendritic cells, differentiation of monocytes to anti-inflammatory macrophages, differentiation of effector T cells to regulatory T cells and modulation of cytokine secretion (190).
Several clinical trials have assessed the numerous MSC sources used in COVID-19 to date (191, 192). MSC therapies have shown improved clinical symptomatology when comparing to conventional treatment (175, 193–195), and have also improved the survival of severe/critical patients even of those who developed ARDS (196–198).
One issue that must be taken into account is the proliferative activity of the MSC that might be affected by its origin as well as donor age (199, 200), therefore MSCs from older donors may have limited therapeutic efficacy. Unlike Embryonic Stem Cells (ESCs), MSCs are considered adult stem cells, and have a limited proliferative capacity. When cultured in vitro, they age, affecting their therapeutic properties, particularly after long-term culture (199). As very large numbers of MSCs are required for therapeutic applications, in vitro expansion is required.
Overall, MSC treatment is mostly safe and well tolerated, however some MSC-related adverse events have still been recorded, being the most predominant fever (201), chills (196), headache (202), allergic rash, and liver dysfunction (203). Nevertheless, in both MSC-treated patients and controls, adverse events were recorded, indicating that the side effects might be infusion-related (195, 204). Moreover, the baseline status of patients with co-existing illnesses and the speed of infusion may be associated with the appearance of adverse events (195).
Treatment with MSCs has several benefits (205) such as: a) can quickly expand to a clinical volume at the appropriate time, b) allogeneic mesenchymal stem cells have not yet been associated with any negative side effects in clinical trials using mesenchymal cells, c) feasible storage for future therapeutic use, d) mesenchymal stem cells are multipotent stem cells, e) suitability and efficacy of mesenchymal stem cells have been known in many clinical trials, f) easy availability as can be obtained from various tissues such as adipose tissues and bone marrow (205).
4.5 Vaccination
While the development of vaccines has taken typically around 8 to 15 years, the current development of multiple vaccines has been unraveled in less than 12 months (206). The remarkable effectiveness of vaccines against SARS-CoV-2, particularly 95% for BNT162b2 (BioNTech/Pfizer) and 94.1% for mRNA-1273 (Moderna), has already been discussed and described extensively (207, 208). Numerous studies have shown that natural immunity seems to persist for a reasonable amount of time (209–211). According to epidemiological research, natural immunity provides protection against reinfections for longer than a year with little, if any, deterioration throughout this period (212–216). The majority of patients’ persistence of anti-SARS-CoV-2 antibodies and cellular immunity over more than a year supports the idea of long-term protection against reinfections. It is worth mentioning that some studies indicate that protection against reinfection reaches its highest level around 4 to 5 months following the initial infection, and then gradually declines. This observation could potentially be attributed to persistent viral shedding or the misclassification of prolonged SARS-CoV-2 infections as reinfections (215–217). In comparison to those who get two doses of an mRNA vaccine, observational studies suggest that natural immunity provides equivalent or higher protection against SARS-CoV-2 infections. However, the data are not entirely consistent, since the loss of natural immunity appears to be very moderate, but there is substantial evidence supporting the notion that protection against SARS-CoV-2 infections conferred by vaccination, tends to diminish relatively quickly over time (218–220). People who have previously contracted SARS-CoV-2 but were afterwards immunized against it, or vice versa, are said to have “hybrid immunity” (219–222). Although the data from the SARS-CoV-2 vaccine randomized controlled trials (RCTs) on hybrid immunity is conflicting, it does appear to be superior to either vaccine-induced (without a booster) or natural immunity alone. Hybrid immunization suggests a higher level of protection when infection and subsequent immunization occur at least 6 months apart as opposed to a shorter gap (223).
Low vaccination rates in Africa, with a large number of countries with less than 10% of the population with vaccination coverage, the lifting or end of restrictions in much of Western countries, the possible appearance of new variants (as it is happening with BQ1.1XBB variants with higher viral scape to previous immunity) and the burst of cases in Asia, which is experiencing the worst situation since the beginning of 2020, is a cause for concern. Therefore, the development of novel alternative treatments is mandatory.
5 Rationale for use of MSC and combinatorial cell therapy in severe to critical COVID-19 patients
In both animal models and human therapeutic trials, MSC therapy inhibited the aberrant immune-mediated inflammatory response brought on by influenza virus infection (1, 224). There have been several clinical studies for stem cell treatment since the outbreak of the COVID-19 pandemic and quite a few have shown that MSCs not only lead to noteworthy reduction of lung damage and recovery time, but also increased patient survival and early-stage tolerance (173, 201, 203, 204).
For patients with high mortality risk from COVID-19, a combination of safe MSC and memory T-cells specific for SARS-CoV-2 (CD45RA-) might be the best course of action. CD45RA- will provide the anti-viral effect during the first days, while MSCs will inhibit the complications of the second phase.
6 Conclusion
MSCs have important immunoregulatory effects on inflammation. Since the effects of SARS-CoV-2 on patients’ bodies are widespread, wherein both the release of pro-inflammatory cytokines, severe damage of alveolar epithelial cells (225) together with systemic effects suggest that the use of MSC might be beneficial in severe-to-critically ill patients, promoting their recovery period and as a result of their build-up in the lung capillary network and repairing the damaged tissue (226). Furthermore, the lack of TMPRSS2 and ACE2 receptors on the MSC makes them resistant to SARS-CoV-2 infection.
Even more, the preliminary proof demonstrating the feasibility and safety of treating COVID-19 patients with moderate to severe symptoms using convalescent CD45RA- memory T-cells, and due to the fact that memory T-cells can swiftly tackle the infection and offer ongoing immune support to lessen the severity of the COVID-19 symptoms, this strategy might eradicate virally infected cells and provide T-cell immunity to prevent recurrent reinfections. Finally, Baricitinib and dexamethasone have proven effective while reducing inflammation and effectively treat COVID-19 patients who are hospitalized (227) however, Baricitinib had less side effects than dexamethasone, and this will help clinicians choose which immunomodulatory medication to administer to patients based on their particular risks for dexamethasone-related side effects (228).
Despite existence of antivirals and neutralizing antibodies specifically developed for SARS-CoV-2 with good results in clinical trials, there is still a huge margin of improvement: problems such as new variants of concern which scape to antibodies or appearance of resistance to antivirals (as already described for Paxlovid) demand the development of new treatments as well as focusing in efficient combinational therapies that show better treatment outcomes when administered together.
Statements
Author contributions
BS Conception and design, collection and assembly of data, data analysis and interpretation, manuscript writing and editing and financial support. BS, AG, EA, LH-B and CS: Collection and assembly of data, data analysis and interpretation, write the first draft. BS-J: writing the first draft of the clinical trial; CS, AM-Q, AP-M and SQ: reviewed the first version. All authors are members of the research team of the DECODE project (ICI21/0016: It is mandatory to decrease COVID-19 deaths). All authors contributed to the article and approved the submitted version.
Funding
This research was funded by the financial support to BS from: Institute of Health Carlos III (Co-funded by Fondos FEDER), Project ICI21/00016 and Agencia Valenciana de Innovacion Projects AVI-GVA COVID-19-68 and GVA-COVI19/2021/047 and Project JDRF 2-SRA-2019-837. BS strongly appreciates the support of his family and friends. The authors declare that this study received funding from BS's family funds. The funder was not involved in the study design, collection, analysis, interpretation of data, the writing of this article, or the decision to submit it for publication.
Conflict of interest
AP-M, CF and BS filed patents on this topic.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1
Organization WH . International Health Regulations Emergency Committee on Novel Coronavirus in China. (2020) Geneva: WHO, World Health Organization.
2
de Groot RJ Baker SC Baric RS Brown CS Drosten C Enjuanes L et al . Middle East respiratory syndrome coronavirus (MERS-CoV): announcement of the Coronavirus Study Group. J Virol (2013) 87(14):7790–2. doi: 10.1128/JVI.01244-13
3
Drosten C Günther S Preiser W van der Werf S Brodt HR Becker S et al . Identification of a novel coronavirus in patients with severe acute respiratory syndrome. New Engl J Med (2003) 348(20):1967–76. doi: 10.1056/NEJMoa030747
4
Belouzard S Millet JK Licitra BN Whittaker GR . Mechanisms of coronavirus cell entry mediated by the viral spike protein. Viruses (2012) 4(6):1011–33. doi: 10.3390/v4061011
5
Wang CC Prather KA Sznitman J Jimenez JL Lakdawala SS Tufekci Z et al . Airborne transmission of respiratory viruses. Sci (New York NY) (2021) 373(6558). doi: 10.1126/science.abd9149
6
Gandhi RT Lynch JB Del Rio C . Mild or moderate Covid-19. New Engl J Med (2020) 383(18):1757–66. doi: 10.1056/NEJMcp2009249
7
Lauer SA Grantz KH Bi Q Jones FK Zheng Q Meredith HR et al . The InCubation period of coronavirus disease 2019 (COVID-19) from publicly reported confirmed cases: estimation and application. Ann Internal Med (2020) 172(9):577–82. doi: 10.7326/M20-0504
8
Huang C Wang Y Li X Ren L Zhao J Hu Y et al . Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet (London England) (2020) 395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5
9
Wu C Chen X Cai Y Xia J Zhou X Xu S et al . Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Internal Med (2020) 180(7):934–43. doi: 10.1001/jamainternmed.2020.0994
10
Wu Z McGoogan JM . Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the chinese center for disease control and prevention. Jama (2020) 323(13):1239–42. doi: 10.1001/jama.2020.2648
11
Grasselli G Zangrillo A Zanella A Antonelli M Cabrini L Castelli A et al . Baseline characteristics and outcomes of 1591 patients infected with SARS-coV-2 admitted to ICUs of the Lombardy Region, Italy. Jama (2020) 323(16):1574–81. doi: 10.1001/jama.2020.5394
12
Götzinger F Santiago-García B Noguera-Julián A Lanaspa M Lancella L Calò Carducci FI et al . COVID-19 in children and adolescents in Europe: a multinational, multicentre cohort study. Lancet Child Adolesc Health (2020) 4(9):653–61. doi: 10.1016/S2352-4642(20)30177-2
13
World Health Organization . World Health Organization and R&D Blueprint strategy for COVID-19. (2022) Geneva: WHO, World Health Organization.
14
Zhang S Yang Z Li ZN Chen ZL Yue SJ Fu RJ et al . Are older people really more susceptible to SARS-coV-2? Aging Dis (2022) 13(5):1336–47. doi: 10.14336/AD.2022.0130
15
Zhou F Yu T Du R Fan G Liu Y Liu Z et al . Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet (London England) (2020) 395(10229):1054–62. doi: 10.1016/S0140-6736(20)30566-3
16
Forrest CB Burrows EK Mejias A Razzaghi H Christakis D Jhaveri R et al . Severity of acute COVID-19 in children <18 years old march 2020 to december 2021. Pediatrics (2022) 149(4). doi: 10.1542/peds.2021-055765
17
Stokes EK Zambrano LD Anderson KN Marder EP Raz KM El Burai Felix S et al . Coronavirus disease 2019 case surveillance - United States, january 22-may 30, 2020. MMWR Morbid Mortal Weekly Rep (2020) 69(24):759–65. doi: 10.15585/mmwr.mm6924e2
18
Oran DP Topol EJ . The proportion of SARS-coV-2 infections that are asymptomatic: A systematic review. Ann Internal Med (2021) 174(5):655–62. doi: 10.7326/M20-6976
19
Ioannidis JPA . Reconciling estimates of global spread and infection fatality rates of COVID-19: An overview of systematic evaluations. Eur J Clin Invest (2021) 51(5):e13554. doi: 10.1111/eci.13554
20
Meyerowitz-Katz G Merone L . A systematic review and meta-analysis of published research data on COVID-19 infection fatality rates. Int J Infect Dis (2020) 101:138–48. doi: 10.1016/j.ijid.2020.09.1464
21
John Hopkins Coronavirus Resource Center . (2022).
22
Seyed Hosseini E Riahi Kashani N Nikzad H Azadbakht J Hassani Bafrani H Haddad Kashani H . The novel coronavirus Disease-2019 (COVID-19): Mechanism of action, detection and recent therapeutic strategies. Virology (2020) 551:1–9. doi: 10.1016/j.virol.2020.08.011
23
Chen N Zhou M Dong X Qu J Gong F Han Y et al . Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet (London England) (2020) 395(10223):507–13. doi: 10.1016/S0140-6736(20)30211-7
24
Hui DS IA E Madani TA Ntoumi F Kock R Dar O et al . The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health - The latest 2019 novel coronavirus outbreak in Wuhan, China. Int J Infect Dis (2020) 91:264–6. doi: 10.1016/j.ijid.2020.01.009
25
Walensky RP Walke HT Fauci AS . SARS-coV-2 variants of concern in the United States-challenges and opportunities. Jama (2021) 325(11):1037–8. doi: 10.1001/jama.2021.2294
26
Rostami A Sepidarkish M Leeflang MMG Riahi SM Nourollahpour Shiadeh M Esfandyari S et al . SARS-CoV-2 seroprevalence worldwide: a systematic review and meta-analysis. Clin Microbiol Infect (2021) 27(3):331–40. doi: 10.1016/j.cmi.2020.10.020
27
Arora RK Joseph A Van Wyk J Rocco S Atmaja A May E et al . SeroTracker: a global SARS-CoV-2 seroprevalence dashboard. Lancet Infect Dis (2021) 21(4):e75–e6. doi: 10.1016/S1473-3099(20)30631-9
28
Jahan N Brahma A Kumar MS Bagepally BS Ponnaiah M Bhatnagar T et al . Seroprevalence of IgG antibodies against SARS-CoV-2 in India, March 2020 to August 2021: a systematic review and meta-analysis. Int J Infect Dis (2022) 116:59–67. doi: 10.1016/j.ijid.2021.12.353
29
Hotez PJ Batista C Amor YB Ergonul O Figueroa JP Gilbert S et al . Global public health security and justice for vaccines and therapeutics in the COVID-19 pandemic. EClinicalMedicine (2021) 39:101053. doi: 10.1016/j.eclinm.2021.101053
30
McIntyre PB Aggarwal R Jani I Jawad J Kochhar S MacDonald N et al . COVID-19 vaccine strategies must focus on severe disease and global equity. Lancet (London England) (2022) 399(10322):406–10. doi: 10.1016/S0140-6736(21)02835-X
31
Leung K Shum MH Leung GM Lam TT Wu JT . Early transmissibility assessment of the N501Y mutant strains of SARS-CoV-2 in the United Kingdom, October to November 2020. Eur Communicable Dis Bull (2021) 26(1). doi: 10.2807/1560-7917.ES.2020.26.1.2002106
32
Murugan NA Javali PS Pandian CJ Ali MA Srivastava V Jeyaraman J . Computational investigation of the increased virulence and pathogenesis of SARS-CoV-2 lineage B.1.1.7. Phys Chem Chem Phys (2022) 24(34):20371–80. doi: 10.1039/d2cp00469k
33
Chen J Wei GW . Omicron BA.2 (B.1.1.529.2): high potential to becoming the next dominating variant. ArXiv (2022). doi: 10.21203/rs.3.rs-1362445/v1
34
COVID-19 Treatment Guidelines Panel . Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Available at https://www.covid19treatmentguidelines.nih.gov/. Accessed 15/03/2003
35
Christie B . Covid-19: Early studies give hope omicron is milder than other variants. BMJ (Clin Res ed) (2021) 375:n3144. doi: 10.1136/bmj.n3144
36
Del Rio C Omer SB Malani PN . Winter of omicron-the evolving COVID-19 pandemic. Jama (2022) 327(4):319–20. doi: 10.1001/jama.2021.24315
37
Fang L Karakiulakis G Roth M . Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med (2020) 8(4):e21. doi: 10.1016/S2213-2600(20)30116-8
38
Gutiérrez-Gutiérrez B Del Toro MD Borobia AM Carcas A Jarrín I Yllescas M et al . Identification and validation of clinical phenotypes with prognostic implications in patients admitted to hospital with COVID-19: a multicentre cohort study. Lancet Infect Dis (2021) 21(6):783–92. doi: 10.1016/S1473-3099(21)00019-0
39
Knight SR Ho A Pius R Buchan I Carson G Drake TM et al . Risk stratification of patients admitted to hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: development and validation of the 4C Mortality Score. BMJ (Clin Res ed) (2020) 370:m3339. doi: 10.1136/bmj.m3339
40
Wu C Liu Y Yang Y Zhang P Zhong W Wang Y et al . Analysis of therapeutic targets for SARS-CoV-2 and discovery of potential drugs by computational methods. Acta Pharm Sin B (2020) 10(5):766–88. doi: 10.1016/j.apsb.2020.02.008
41
Whittaker GR Millet JK . Biochemical characterization of Middle East respiratory syndrome coronavirus spike protein proteolytic processing. Methods Mol Biol (Clifton NJ) (2020) 2099:21–37. doi: 10.1007/978-1-0716-0211-9_3
42
Zhou P Yang XL Wang XG Hu B Zhang L Zhang W et al . A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature (2020) 579(7798):270–3. doi: 10.1038/s41586-020-2012-7
43
Zhu N Zhang D Wang W Li X Yang B Song J et al . A novel coronavirus from patients with pneumonia in China, 2019. New Engl J Med (2020) 382(8):727–33. doi: 10.1056/NEJMoa2001017
44
Mohammad A Marafie SK Alshawaf E Abu-Farha M Abubaker J Al-Mulla F . Structural analysis of ACE2 variant N720D demonstrates a higher binding affinity to TMPRSS2. Life Sci (2020) 259:118219. doi: 10.1016/j.lfs.2020.118219
45
Mohammad A Alshawaf E Marafie SK Abu-Farha M Abubaker J Al-Mulla F . Higher binding affinity of furin for SARS-CoV-2 spike (S) protein D614G mutant could be associated with higher SARS-CoV-2 infectivity. Int J Infect Dis (2021) 103:611–6. doi: 10.1016/j.ijid.2020.10.033
46
Alanagreh L Alzoughool F Atoum M . The human coronavirus disease COVID-19: its origin, characteristics, and insights into potential drugs and its mechanisms. Pathog (Basel Switzerland) (2020) 9(5). doi: 10.3390/pathogens9050331
47
Bowie AG Unterholzner L . Viral evasion and subversion of pattern-recognition receptor signalling. Nat Rev Immunol (2008) 8(12):911–22. doi: 10.1038/nri2436
48
Hatton CF Botting RA Dueñas ME Haq IJ Verdon B Thompson BJ et al . Delayed induction of type I and III interferons mediates nasal epithelial cell permissiveness to SARS-CoV-2. Nat Commun (2021) 12(1):7092. doi: 10.1038/s41467-021-27318-0
49
Müller U Steinhoff U Reis LF Hemmi S Pavlovic J Zinkernagel RM et al . Functional role of type I and type II interferons in antiviral defense. Sci (New York NY) (1994) 264(5167):1918–21. doi: 10.1126/science.8009221
50
Alene M Yismaw L Assemie MA Ketema DB Mengist B Kassie B et al . Magnitude of asymptomatic COVID-19 cases throughout the course of infection: A systematic review and meta-analysis. PloS One (2021) 16(3):e0249090. doi: 10.1371/journal.pone.0249090
51
Chen X Huang Z Wang J Zhao S Wong MC Chong KC et al . Ratio of asymptomatic COVID-19 cases among ascertained SARS-CoV-2 infections in different regions and population groups in 2020: a systematic review and meta-analysis including 130 123 infections from 241 studies. BMJ Open (2021) 11(12):e049752. doi: 10.1136/bmjopen-2021-049752
52
Li C Zhu Y Qi C Liu L Zhang D Wang X et al . Estimating the prevalence of asymptomatic COVID-19 cases and their contribution in transmission - using henan province, China, as an example. Front Med (2021) 8:591372. doi: 10.3389/fmed.2021.591372
53
Ma Q Liu J Liu Q Kang L Liu R Jing W et al . Global percentage of asymptomatic SARS-coV-2 infections among the tested population and individuals with confirmed COVID-19 diagnosis: A systematic review and meta-analysis. JAMA Network Open (2021) 4(12):e2137257. doi: 10.1001/jamanetworkopen.2021.37257
54
Sah P Fitzpatrick MC Zimmer CF Abdollahi E Juden-Kelly L Moghadas SM et al . Asymptomatic SARS-CoV-2 infection: A systematic review and meta-analysis. Proc Natl Acad Sci United States America (2021) 118(34). doi: 10.1073/pnas.2109229118
55
Shang W Kang L Cao G Wang Y Gao P Liu J et al . Percentage of asymptomatic infections among SARS-coV-2 omicron variant-positive individuals: A systematic review and meta-analysis. Vaccines (2022) 10(7). doi: 10.3390/vaccines10071049
56
Subramanian R He Q Pascual M . Quantifying asymptomatic infection and transmission of COVID-19 in New York City using observed cases, serology, and testing capacity. Proc Natl Acad Sci United States America (2021) 118(9). doi: 10.1073/pnas.2019716118
57
Eguizabal C Herrera L Inglés-Ferrándiz M Izpisua Belmonte JC . Treating primary immunodeficiencies with defects in NK cells: from stem cell therapy to gene editing. Stem Cell Res Ther (2020) 11(1):453. doi: 10.1186/s13287-020-01964-5
58
Ferreras C Pascual-Miguel B Mestre-Durán C Navarro-Zapata A Clares-Villa L Martín-Cortázar C et al . SARS-coV-2-specific memory T lymphocytes from COVID-19 convalescent donors: identification, biobanking, and large-scale production for adoptive cell therapy. Front Cell Dev Biol (2021) 9:620730. doi: 10.3389/fcell.2021.620730
59
Barnes BJ Adrover JM Baxter-Stoltzfus A Borczuk A Cools-Lartigue J Crawford JM et al . Targeting potential drivers of COVID-19: Neutrophil extracellular traps. J Exp Med (2020) 217(6). doi: 10.1084/jem.20200652
60
Hemmat N Derakhshani A Bannazadeh Baghi H Silvestris N Baradaran B De Summa S . Neutrophils, crucial, or harmful immune cells involved in coronavirus infection: A bioinformatics study. Front Genet (2020) 11:641. doi: 10.3389/fgene.2020.00641
61
Soy M Keser G Atagündüz P Tabak F Atagündüz I Kayhan S . Cytokine storm in COVID-19: pathogenesis and overview of anti-inflammatory agents used in treatment. Clin Rheumatol (2020) 39(7):2085–94. doi: 10.1007/s10067-020-05190-5
62
Vardhana SA Wolchok JD . The many faces of the anti-COVID immune response. J Exp Med (2020) 217(6). doi: 10.1084/jem.20200678
63
Hussman JP . Cellular and molecular pathways of COVID-19 and potential points of therapeutic intervention. Front Pharmacol (2020) 11:1169. doi: 10.3389/fphar.2020.01169
64
Haiming W Xiaoling X Yonggang Z Binqing F Xiaohu Z Dongsheng W et al . Aberrant pathogenic GM-CSF+ T cells and inflammatory CD14+CD16+ monocytes in severe pulmonary syndrome patients of a new coronavirus. bioRxiv (2020), 945576. doi: 10.1101/2020.02.12.945576
65
Hu B Huang S Yin L . The cytokine storm and COVID-19. J Med Virol (2021) 93(1):250–6. doi: 10.1002/jmv.26232
66
Farzi R Aghbash PS Eslami N Azadi A Shamekh A Hemmat N et al . The role of antigen-presenting cells in the pathogenesis of COVID-19. Pathol Res Practice (2022) 233:153848. doi: 10.1016/j.prp.2022.153848
67
McKechnie JL Blish CA . The innate immune system: fighting on the front lines or fanning the flames of COVID-19? Cell Host Microbe (2020) 27(6):863–9. doi: 10.1016/j.chom.2020.05.009
68
Guan WJ Ni ZY Hu Y Liang WH Ou CQ He JX et al . Clinical characteristics of coronavirus disease 2019 in China. New Engl J Med (2020) 382(18):1708–20. doi: 10.1056/NEJMoa2002032
69
Yang L Xie X Tu Z Fu J Xu D Zhou Y . The signal pathways and treatment of cytokine storm in COVID-19. Signal Transduction Targeted Ther (2021) 6(1):255. doi: 10.1038/s41392-021-00679-0
70
He L Ding Y Zhang Q Che X He Y Shen H et al . Expression of elevated levels of pro-inflammatory cytokines in SARS-CoV-infected ACE2+ cells in SARS patients: relation to the acute lung injury and pathogenesis of SARS. J Pathol (2006) 210(3):288–97. doi: 10.1002/path.2067
71
Lau SKP Lau CCY Chan KH Li CPY Chen H Jin DY et al . Delayed induction of proinflammatory cytokines and suppression of innate antiviral response by the novel Middle East respiratory syndrome coronavirus: implications for pathogenesis and treatment. J Gen Virol (2013) 94(Pt 12):2679–90. doi: 10.1099/vir.0.055533-0
72
Tian S Hu W Niu L Liu H Xu H Xiao SY . Pulmonary pathology of early-phase 2019 novel coronavirus (COVID-19) pneumonia in two patients with lung cancer. J Thorac Oncol (2020) 15(5):700–4. doi: 10.1016/j.jtho.2020.02.010
73
Qin C Zhou L Hu Z Zhang S Yang S Tao Y et al . Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in wuhan, China. Clin Infect Dis (2020) 71(15):762–8. doi: 10.1093/cid/ciaa248
74
Fuchs TA Abed U Goosmann C Hurwitz R Schulze I Wahn V et al . Novel cell death program leads to neutrophil extracellular traps. J Cell Biol (2007) 176(2):231–41. doi: 10.1083/jcb.200606027
75
Schönrich G Raftery MJ . Neutrophil extracellular traps go viral. Front Immunol (2016) 7:366. doi: 10.1083/jcb.200606027
76
Bonaventura A Liberale L Carbone F Vecchié A Diaz-Cañestro C Camici GG et al . The pathophysiological role of neutrophil extracellular traps in inflammatory diseases. Thromb Haemostasis (2018) 118(1):6–27. doi: 10.1160/TH17-09-0630
77
Czaikoski PG Mota JM Nascimento DC Sônego F Castanheira FV Melo PH et al . Neutrophil extracellular traps induce organ damage during experimental and clinical sepsis. PloS One (2016) 11(2):e0148142. doi: 10.1371/journal.pone.0148142
78
Middleton EA He XY Denorme F Campbell RA Ng D Salvatore SP et al . Neutrophil extracellular traps contribute to immunothrombosis in COVID-19 acute respiratory distress syndrome. Blood (2020) 136(10):1169–79. doi: 10.1182/blood.2020007008
79
Hasan A Al-Ozairi E Al-Baqsumi Z Ahmad R Al-Mulla F . Cellular and humoral immune responses in covid-19 and immunotherapeutic approaches. ImmunoTargets Ther (2021) 10:63–85. doi: 10.2147/ITT.S280706
80
Watanabe T Bartrand TA Weir MH Omura T Haas CN . Development of a dose-response model for SARS coronavirus. Risk Anal (2010) 30(7):1129–38. doi: 10.1111/j.1539-6924.2010.01427.x
81
Seow J Graham C Merrick B Acors S Pickering S Steel KJA et al . Longitudinal observation and decline of neutralizing antibody responses in the three months following SARS-CoV-2 infection in humans. Nat Microbiol (2020) 5(12):1598–607. doi: 10.1038/s41564-020-00813-8
82
Goyal A Reeves DB Cardozo-Ojeda EF Schiffer JT Mayer BT . Viral load and contact heterogeneity predict SARS-CoV-2 transmission and super-spreading events. eLife (2021) 10. doi: 10.7554/eLife.63537.sa2
83
Sia SF Yan LM Chin AWH Fung K Choy KT Wong AYL et al . Pathogenesis and transmission of SARS-CoV-2 in golden hamsters. Nature (2020) 583(7818):834–8. doi: 10.1038/s41586-020-2342-5
84
Kim JY Ko JH Kim Y Kim YJ Kim JM Chung YS et al . Viral load kinetics of SARS-coV-2 infection in first two patients in Korea. J Korean Med Sci (2020) 35(7):e86. doi: 10.3346/jkms.2020.35.e86
85
Lucas C Wong P Klein J Castro TBR Silva J Sundaram M et al . Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature (2020) 584(7821):463–9. doi: 10.1038/s41586-020-2588-y
86
Peiris JS Chu CM Cheng VC Chan KS Hung IF Poon LL et al . Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet (London England) (2003) 361(9371):1767–72. doi: 10.1016/S0140-6736(03)13412-5
87
Iwasaki A Yang Y . The potential danger of suboptimal antibody responses in COVID-19. Nat Rev Immunol (2020) 20(6):339–41. doi: 10.1038/s41577-020-0321-6
88
Li X Zhang Z Wang Z Gutiérrez-Castrellón P Shi H . Cell deaths: Involvement in the pathogenesis and intervention therapy of COVID-19. Signal Transduction Targeted Ther (2022) 7(1):186. doi: 10.1038/s41392-022-01043-6
89
Demarco B Chen KW Broz P . Cross talk between intracellular pathogens and cell death. Immunol Rev (2020) 297(1):174–93. doi: 10.1111/imr.12892
90
Imre G . The involvement of regulated cell death forms in modulating the bacterial and viral pathogenesis. Int Rev Cell Mol Biol (2020) 353:211–53. doi: 10.1016/bs.ircmb.2019.12.008
91
Zhao Z Lu K Mao B Liu S Trilling M Huang A et al . The interplay between emerging human coronavirus infections and autophagy. Emerg Microbes Infect (2021) 10(1):196–205. doi: 10.1080/22221751.2021.1872353
92
Li S Zhang Y Guan Z Li H Ye M Chen X et al . SARS-CoV-2 triggers inflammatory responses and cell death through caspase-8 activation. Signal Transduction Targeted Ther (2020) 5(1):235. doi: 10.1038/s41392-020-00334-0
93
Cizmecioglu A Akay Cizmecioglu H Goktepe MH Emsen A Korkmaz C Esenkaya Tasbent F et al . Apoptosis-induced T-cell lymphopenia is related to COVID-19 severity. J Med Virol (2021) 93(5):2867–74. doi: 10.1002/jmv.26742
94
Park A Iwasaki A . Type I and type III interferons - induction, signaling, evasion, and application to combat COVID-19. Cell Host Microbe (2020) 27(6):870–8. doi: 10.1016/j.chom.2020.05.008
95
Vabret N Britton GJ Gruber C Hegde S Kim J Kuksin M et al . Immunology of COVID-19: current state of the science. Immunity (2020) 52(6):910–41. doi: 10.1016/j.immuni.2020.05.002
96
Karki R Kanneganti TD . The ‘cytokine storm’: molecular mechanisms and therapeutic prospects. Trends Immunol (2021) 42(8):681–705. doi: 10.1016/j.it.2021.06.001
97
Karki R Sharma BR Tuladhar S Williams EP Zalduondo L Samir P et al . Synergism of TNF-α and IFN-γ Triggers inflammatory cell death, tissue damage, and mortality in SARS-coV-2 infection and cytokine shock syndromes. Cell (2021) 184(1):149–68.e17. doi: 10.1016/j.cell.2020.11.025
98
Feng Y Ling Y Bai T Xie Y Huang J Li J et al . COVID-19 with different severities: A multicenter study of clinical features. Am J Respir Crit Care Med (2020) 201(11):1380–8. doi: 10.1164/rccm.202002-0445OC
99
Calabretta E Moraleda JM Iacobelli M Jara R Vlodavsky I O’Gorman P et al . COVID-19-induced endotheliitis: emerging evidence and possible therapeutic strategies. Br J Haematol (2021) 193(1):43–51. doi: 10.1111/bjh.17240
100
Varga Z Flammer AJ Steiger P Haberecker M Andermatt R Zinkernagel AS et al . Endothelial cell infection and endotheliitis in COVID-19. Lancet (London England) (2020) 395(10234):1417–8. doi: 10.1016/S0140-6736(20)30937-5
101
Yuan L Cheng S Sol W van der Velden AIM Vink H Rabelink TJ et al . Heparan sulfate mimetic fucoidan restores the endothelial glycocalyx and protects against dysfunction induced by serum of COVID-19 patients in the intensive care unit. ERJ Open Res (2022) 8(2). doi: 10.1183/23120541.00652-2021
102
Stahl K Gronski PA Kiyan Y Seeliger B Bertram A Pape T et al . Injury to the endothelial glycocalyx in critically ill patients with COVID-19. Am J Respir Crit Care Med (2020) 202(8):1178–81. doi: 10.1164/rccm.202007-2676LE
103
Anderson TJ Meredith IT Yeung AC Frei B Selwyn AP Ganz P . The effect of cholesterol-lowering and antioxidant therapy on endothelium-dependent coronary vasomotion. New Engl J Med (1995) 332(8):488–93. doi: 10.1056/NEJM199502233320802
104
Feldmann M Maini RN Woody JN Holgate ST Winter G Rowland M et al . Trials of anti-tumour necrosis factor therapy for COVID-19 are urgently needed. Lancet (London England) (2020) 395(10234):1407–9. doi: 10.1016/S0140-6736(20)30858-8
105
Flammer AJ Sudano I Hermann F Gay S Forster A Neidhart M et al . Angiotensin-converting enzyme inhibition improves vascular function in rheumatoid arthritis. Circulation (2008) 117(17):2262–9. doi: 10.1161/CIRCULATIONAHA.107.734384
106
Yang L Liu S Liu J Zhang Z Wan X Huang B et al . COVID-19: immunopathogenesis and immunotherapeutics. Signal Transduction Targeted Ther (2020) 5(1):128. doi: 10.1038/s41392-020-00243-2
107
Zhang JJ Dong X Cao YY Yuan YD Yang YB Yan YQ et al . Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy (2020) 75(7):1730–41. doi: 10.1111/all.14238
108
Mathew D Giles JR Baxter AE Oldridge DA Greenplate AR Wu JE et al . Deep immune profiling of COVID-19 patients reveals distinct immunotypes with therapeutic implications. Sci (New York NY) (2020) 369(6508). doi: 10.1126/science.abc8511
109
Tan YX Tan TH Lee MJ Tham PY Gunalan V Druce J et al . Induction of apoptosis by the severe acute respiratory syndrome coronavirus 7a protein is dependent on its interaction with the Bcl-XL protein. J Virol (2007) 81(12):6346–55. doi: 10.1128/JVI.00090-07
110
Tan L Wang Q Zhang D Ding J Huang Q Tang YQ et al . Lymphopenia predicts disease severity of COVID-19: a descriptive and predictive study. Signal Transduction Targeted Ther (2020) 5(1):33. doi: 10.1038/s41392-020-0148-4
111
Moosazadeh M Mousavi T . Combination therapy of tocilizumab and steroid for COVID-19 patients: A meta-analysis. J Med Virol (2022) 94(4):1350–6. doi: 10.1002/jmv.27489
112
Sánchez-Guijo F García-Arranz M López-Parra M Monedero P Mata-Martínez C Santos A et al . Adipose-derived mesenchymal stromal cells for the treatment of patients with severe SARS-CoV-2 pneumonia requiring mechanical ventilation. A Proof Concept Study EClinMed (2020) 25:100454. doi: 10.1016/j.eclinm.2020.100454
113
Ahmed-Hassan H Sisson B Shukla RK Wijewantha Y Funderburg NT Li Z et al . Innate immune responses to highly pathogenic coronaviruses and other significant respiratory viral infections. Front Immunol (2020) 11:1979. doi: 10.3389/fimmu.2020.01979
114
Angelini DE Kaatz S Rosovsky RP Zon RL Pillai S Robertson WE et al . COVID-19 and venous thromboembolism: A narrative review. Res Pract Thromb Haemostasis (2022) 6(2):e12666. doi: 10.1002/rth2.12666
115
Kaminetzky M Moore W Fansiwala K Babb JS Kaminetzky D Horwitz LI et al . Pulmonary embolism at CT pulmonary angiography in patients with COVID-19. Radiol Cardiothoracic Imag (2020) 2(4):e200308. doi: 10.1148/ryct.2020200308
116
Suh YJ Hong H Ohana M Bompard F Revel MP Valle C et al . Pulmonary embolism and deep vein thrombosis in COVID-19: A systematic review and meta-analysis. Radiology (2021) 298(2):E70–e80. doi: 10.1148/radiol.2020203557
117
Bradley BT Maioli H Johnston R Chaudhry I Fink SL Xu H et al . Histopathology and ultrastructural findings of fatal COVID-19 infections in Washington State: a case series. Lancet (London England) (2020) 396(10247):320–32. doi: 10.1016/S0140-6736(20)31305-2
118
Unudurthi SD Luthra P Bose RJC McCarthy JR Kontaridis MI . Cardiac inflammation in COVID-19: Lessons from heart failure. Life Sci (2020) 260:118482. doi: 10.1016/j.lfs.2020.118482
119
Wichmann D Sperhake JP Lütgehetmann M Steurer S Edler C Heinemann A et al . Autopsy findings and venous thromboembolism in patients with COVID-19: A prospective cohort study. Ann Internal Med (2020) 173(4):268–77. doi: 10.7326/M20-2003
120
Shah MD Sumeh AS Sheraz M Kavitha MS Venmathi Maran BA Rodrigues KF . A mini-review on the impact of COVID 19 on vital organs. Biomed Pharmacother (2021) 143:112158. doi: 10.1016/j.biopha.2021.112158
121
Xu Z Shi L Wang Y Zhang J Huang L Zhang C et al . Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med (2020) 8(4):420–2. doi: 10.1016/S2213-2600(20)30076-X
122
Chen S Guan F Candotti F Benlagha K Camara NOS Herrada AA et al . The role of B cells in COVID-19 infection and vaccination. Front Immunol (2022) 13:988536. doi: 10.3389/fimmu.2022.988536
123
Mohammed RN Tamjidifar R Rahman HS Adili A Ghoreishizadeh S Saeedi H et al . Correction: A comprehensive review about immune responses and exhaustion during coronavirus disease (COVID-19). Cell Communication Signaling (2022) 20(1):139. doi: 10.1186/s12964-022-00967-4
124
Cox RJ Brokstad KA . Not just antibodies: B cells and T cells mediate immunity to COVID-19. Nat Rev Immunol (2020) 20(10):581–2. doi: 10.1038/s41577-020-00436-4
125
Shuwa HA Shaw TN Knight SB Wemyss K McClure FA Pearmain L et al . Alterations in T and B cell function persist in convalescent COVID-19 patients. Med (New York NY) (2021) 2(6):720–35.e4. doi: 10.2139/ssrn.3720301
126
Mehta P McAuley DF Brown M Sanchez E Tattersall RS Manson JJ . COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet (London England) (2020) 395(10229):1033–4. doi: 10.1016/S0140-6736(20)30628-0
127
Teijaro JR Walsh KB Rice S Rosen H Oldstone MB . Mapping the innate signaling cascade essential for cytokine storm during influenza virus infection. Proc Natl Acad Sci United States America (2014) 111(10):3799–804. doi: 10.1073/pnas.1400593111
128
Merad M Martin JC . Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat Rev Immunol (2020) 20(6):355–62. doi: 10.1038/s41577-020-0331-4
129
Wang F Nie J Wang H Zhao Q Xiong Y Deng L et al . Characteristics of peripheral lymphocyte subset alteration in COVID-19 pneumonia. J Infect Dis (2020) 221(11):1762–9. doi: 10.1093/infdis/jiaa150
130
Zheng Y Zhang Y Chi H Chen S Peng M Luo L et al . The hemocyte counts as a potential biomarker for predicting disease progression in COVID-19: a retrospective study. Clin Chem Lab Med (2020) 58(7):1106–15. doi: 10.1515/cclm-2020-0377
131
Moore JB June CH . Cytokine release syndrome in severe COVID-19. Sci (New York NY) (2020) 368(6490):473–4. doi: 10.1126/science.abb8925
132
Tay MZ Poh CM Rénia L MacAry PA Ng LFP . The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol (2020) 20(6):363–74. doi: 10.1038/s41577-020-0311-8
133
Shi M Liu ZW Wang FS . Immunomodulatory properties and therapeutic application of mesenchymal stem cells. Clin Exp Immunol (2011) 164(1):1–8. doi: 10.1111/j.1365-2249.2011.04327.x
134
Connors JM Levy JH . Thromboinflammation and the hypercoagulability of COVID-19. J Thromb Haemostasis (2020) 18(7):1559–61. doi: 10.1111/jth.14849
135
Alzghari SK Acuña VS . Supportive treatment with tocilizumab for COVID-19: A systematic review. J Clin Virol (2020) 127:104380. doi: 10.1016/j.jcv.2020.104380
136
Sanders JM Monogue ML Jodlowski TZ Cutrell JB . Pharmacologic treatments for coronavirus disease 2019 (COVID-19): A review. Jama (2020) 323(18):1824–36. doi: 10.1001/jama.2020.6019
137
Scavone C Brusco S Bertini M Sportiello L Rafaniello C Zoccoli A et al . Current pharmacological treatments for COVID-19: What’s next? Br J Pharmacol (2020) 177(21):4813–24. doi: 10.1111/bph.15072
138
Shen C Wang Z Zhao F Yang Y Li J Yuan J et al . Treatment of 5 critically ill patients with COVID-19 with convalescent plasma. Jama (2020) 323(16):1582–9. doi: 10.1001/jama.2020.4783
139
Avendaño-Solá C Ramos-Martínez A Muñez-Rubio E Ruiz-Antorán B Malo de Molina R Torres F et al . A multicenter randomized open-label clinical trial for convalescent plasma in patients hospitalized with COVID-19 pneumonia. J Clin Invest (2021) 131(20). doi: 10.1172/JCI152740
140
Bautista-Vargas M Bonilla-Abadía F Cañas CA . Potential role for tissue factor in the pathogenesis of hypercoagulability associated with in COVID-19. J Thromb Thrombolysis (2020) 50(3):479–83. doi: 10.1007/s11239-020-02172-x
141
Del Valle DM Kim-Schulze S Huang HH Beckmann ND Nirenberg S Wang B et al . An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat Med (2020) 26(10):1636–43. doi: 10.1038/s41591-020-1051-9
142
Mazzoni A Salvati L Maggi L Annunziato F Cosmi L . Hallmarks of immune response in COVID-19: Exploring dysregulation and exhaustion. Semin Immunol (2021) 55:101508. doi: 10.1016/j.smim.2021.101508
143
Luo P Liu Y Qiu L Liu X Liu D Li J . Tocilizumab treatment in COVID-19: A single center experience. J Med Virol (2020) 92(7):814–8. doi: 10.1002/jmv.25801
144
Zhang C Wu Z Li JW Zhao H Wang GQ . Cytokine release syndrome in severe COVID-19: interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality. Int J Antimicrobial Agents (2020) 55(5):105954. doi: 10.1016/j.ijantimicag.2020.105954
145
Cortegiani A Ippolito M Greco M Granone V Protti A Gregoretti C et al . Rationale and evidence on the use of tocilizumab in COVID-19: a systematic review. Pulmonology (2021) 27(1):52–66. doi: 10.1016/j.pulmoe.2020.07.003
146
Kyriazopoulou E Poulakou G Milionis H Metallidis S Adamis G Tsiakos K et al . Early treatment of COVID-19 with anakinra guided by soluble urokinase plasminogen receptor plasma levels: a double-blind, randomized controlled phase 3 trial. Nat Med (2021) 27(10):1752–60. doi: 10.1038/s41591-021-01499-z
147
Marconi VC Ramanan AV de Bono S Kartman CE Krishnan V Liao R et al . Efficacy and safety of baricitinib for the treatment of hospitalised adults with COVID-19 (COV-BARRIER): a randomised, double-blind, parallel-group, placebo-controlled phase 3 trial. Lancet Respir Med (2021) 9(12):1407–18. doi: 10.1016/S2213-2600(21)00331-3
148
Satarker S Tom AA Shaji RA Alosious A Luvis M Nampoothiri M . JAK-STAT pathway inhibition and their implications in COVID-19 therapy. Postgrad Med (2021) 133(5):489–507. doi: 10.1080/00325481.2020.1855921
149
Grant AH Estrada A 3rd Ayala-Marin YM Alvidrez-Camacho AY Rodriguez G Robles-Escajeda E et al . The many faces of JAKs and STATs within the COVID-19 storm. Front Immunol (2021) 12:690477. doi: 10.3389/fimmu.2021.690477
150
Ragab D Salah Eldin H Taeimah M Khattab R Salem R . The COVID-19 cytokine storm; what we know so far. Front Immunol (2020) 11:1446. doi: 10.3389/fimmu.2020.01446
151
Barilli A Visigalli R Ferrari F Recchia Luciani G Soli M Dall’Asta V et al . The JAK1/2 inhibitor baricitinib mitigates the spike-induced inflammatory response of immune and endothelial cells in vitro. Biomedicines (2022) 10(9). doi: 10.3390/biomedicines10092324
152
Recovery Trial . (2022).
153
Isidori AM Arnaldi G Boscaro M Falorni A Giordano C Giordano R et al . COVID-19 infection and glucocorticoids: update from the Italian Society of Endocrinology Expert Opinion on steroid replacement in adrenal insufficiency. J Endocrinol Invest (2020) 43(8):1141–7. doi: 10.1007/s40618-020-01266-w
154
Theoharides TC Conti P . Dexamethasone for COVID-19? Not so fast. J Biol Regulators Homeostatic Agents (2020) 34(3):1241–3. doi: 10.23812/20-EDITORIAL_1-5
155
Li H Yan B Gao R Ren J Yang J . Effectiveness of corticosteroids to treat severe COVID-19: A systematic review and meta-analysis of prospective studies. Int Immunopharmacol (2021) 100:108121. doi: 10.1016/j.intimp.2021.108121
156
FakhriRavari A Jin S Kachouei FH Le D Lopez M . Systemic corticosteroids for management of COVID-19: Saving lives or causing harm? Int J Immunopathol Pharmacol (2021) 35:20587384211063976. doi: 10.1177/20587384211063976
157
Grifoni A Weiskopf D Ramirez SI Mateus J Dan JM Moderbacher CR et al . Targets of T cell responses to SARS-coV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell (2020) 181(7):1489–501.e15. doi: 10.1016/j.cell.2020.05.015
158
Leung W Soh TG Linn YC Low JG Loh J Chan M et al . Rapid production of clinical-grade SARS-CoV-2 specific T cells. Adv Cell Gene Ther (2020) 3(4):e101. doi: 10.1002/acg2.101
159
Ng OW Chia A Tan AT Jadi RS Leong HN Bertoletti A et al . Memory T cell responses targeting the SARS coronavirus persist up to 11 years post-infection. Vaccine (2016) 34(17):2008–14. doi: 10.1016/j.vaccine.2016.02.063
160
Teschner D Distler E Wehler D Frey M Marandiuc D Langeveld K et al . Depletion of naive T cells using clinical grade magnetic CD45RA beads: a new approach for GVHD prophylaxis. Bone Marrow Transplantation (2014) 49(1):138–44. doi: 10.1038/bmt.2013.114
161
Triplett BM Shook DR Eldridge P Li Y Kang G Dallas M et al . Rapid memory T-cell reconstitution recapitulating CD45RA-depleted haploidentical transplant graft content in patients with hematologic Malignancies. Bone Marrow Transplantation (2015) 50(7):968–77. doi: 10.1038/bmt.2014.324
162
Pérez-Martínez A Mora-Rillo M Ferreras C Guerra-García P Pascual-Miguel B Mestre-Durán C et al . Phase I dose-escalation single centre clinical trial to evaluate the safety of infusion of memory T cells as adoptive therapy in COVID-19 (RELEASE). EClinicalMedicine (2021) 39:101086. doi: 10.1016/j.eclinm.2021.101086
163
Al-Akioui Sanz K Echecopar Parente C Ferreras C Menéndez Ribes M Navarro A Mestre C et al . CD45RA- T cells to treat severe refractory infections in immunocompromised patients. Front Med (Lausanne) (2023) 10:1083215. doi: 10.3389/fmed.2023.1083215
164
Soria-Juan B Escacena N Capilla-González V Aguilera Y Llanos L Tejedo JR et al . Cost-effective, safe, and personalized cell therapy for critical limb ischemia in type 2 diabetes mellitus. Front Immunol (2019) 10:1151. doi: 10.3389/fimmu.2019.01151
165
Han Y Li X Zhang Y Han Y Chang F Ding J . Mesenchymal stem cells for regenerative medicine. Cells (2019) 8(8). doi: 10.3390/cells8080886
166
Pittenger MF Mackay AM Beck SC Jaiswal RK Douglas R Mosca JD et al . Multilineage potential of adult human mesenchymal stem cells. Sci (New York NY) (1999) 284(5411):143–7. doi: 10.1126/science.284.5411.143
167
Prockop DJ . Marrow stromal cells as stem cells for nonhematopoietic tissues. Sci (New York NY) (1997) 276(5309):71–4. doi: 10.1126/science.276.5309.71
168
Rogers CJ Harman RJ Bunnell BA Schreiber MA Xiang C Wang FS et al . Rationale for the clinical use of adipose-derived mesenchymal stem cells for COVID-19 patients. J Trans Med (2020) 18(1):203. doi: 10.1186/s12967-020-02380-2
169
Hashmi S Ahmed M Murad MH Litzow MR Adams RH Ball LM et al . Survival after mesenchymal stromal cell therapy in steroid-refractory acute graft-versus-host disease: systematic review and meta-analysis. Lancet Haematol (2016) 3(1):e45–52. doi: 10.1016/S2352-3026(15)00224-0
170
Le Blanc K Frassoni F Ball L Locatelli F Roelofs H Lewis I et al . Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet (London England) (2008) 371(9624):1579–86. doi: 10.1016/S0140-6736(08)60690-X
171
Grégoire C Layios N Lambermont B Lechanteur C Briquet A Bettonville V et al . Bone marrow-derived mesenchymal stromal cell therapy in severe COVID-19: preliminary results of a phase I/II clinical trial. Front Immunol (2022) 13:932360. doi: 10.3389/fimmu.2022.932360
172
Darwish I Mubareka S Liles WC . Immunomodulatory therapy for severe influenza. Expert Rev Anti-infective Ther (2011) 9(7):807–22. doi: 10.1586/eri.11.56
173
Liang B Chen J Li T Wu H Yang W Li Y et al . Clinical remission of a critically ill COVID-19 patient treated by human umbilical cord mesenchymal stem cells: A case report. Medicine (2020) 99(31):e21429. doi: 10.1097/MD.0000000000021429
174
Shi L Huang H Lu X Yan X Jiang X Xu R et al . Effect of human umbilical cord-derived mesenchymal stem cells on lung damage in severe COVID-19 patients: a randomized, double-blind, placebo-controlled phase 2 trial. Signal Transduction Targeted Ther (2021) 6(1):58. doi: 10.1038/s41392-021-00488-5
175
Leng Z Zhu R Hou W Feng Y Yang Y Han Q et al . Transplantation of ACE2(-) mesenchymal stem cells improves the outcome of patients with COVID-19 pneumonia. Aging Dis (2020) 11(2):216–28. doi: 10.14336/AD.2020.0228
176
Dilogo IH Aditianingsih D Sugiarto A Burhan E Damayanti T Sitompul PA et al . Umbilical cord mesenchymal stromal cells as critical COVID-19 adjuvant therapy: A randomized controlled trial. Stem Cells Trans Med (2021) 10(9):1279–87. doi: 10.1002/sctm.21-0046
177
Court AC Le-Gatt A Luz-Crawford P Parra E Aliaga-Tobar V Bátiz LF et al . Mitochondrial transfer from MSCs to T cells induces Treg differentiation and restricts inflammatory response. EMBO Rep (2020) 21(2):e48052. doi: 10.15252/embr.201948052
178
Islam MN Das SR Emin MT Wei M Sun L Westphalen K et al . Mitochondrial transfer from bone-marrow-derived stromal cells to pulmonary alveoli protects against acute lung injury. Nat Med (2012) 18(5):759–65. doi: 10.1038/nm.2736
179
Sadeghi B Remberger M Gustafsson B Winiarski J Moretti G Khoein B et al . Long-term follow-up of a pilot study using placenta-derived decidua stromal cells for severe acute graft-versus-host disease. Biol Blood Marrow Transplant (2019) 25(10):1965–9. doi: 10.1016/j.bbmt.2019.05.034
180
Naji A Eitoku M Favier B Deschaseaux F Rouas-Freiss N Suganuma N . Biological functions of mesenchymal stem cells and clinical implications. Cell Mol Life Sci (2019) 76(17):3323–48. doi: 10.1007/s00018-019-03125-1
181
Leyendecker A Jr. Pinheiro CCG Amano MT Bueno DF . The use of human mesenchymal stem cells as therapeutic agents for the in vivo treatment of immune-related diseases: A systematic review. Front Immunol (2018) 9:2056. doi: 10.3389/fimmu.2018.02056
182
Zhou Y Yamamoto Y Xiao Z Ochiya T . The immunomodulatory functions of mesenchymal stromal/stem cells mediated via paracrine activity. J Clin Med (2019) 8(7). doi: 10.3390/jcm8071025
183
Soler Rich R Rius Tarruella J Melgosa Camarero MT . Expanded mesenchymal stem cells: a novel therapeutic approach for SARS-CoV-2 pneumonia (COVID-19). Concepts regarding a first case in Spain. Med Clin (English ed) (2020) 155(7):318–9. doi: 10.1016/j.medcli.2020.06.018
184
Khoury M Cuenca J Cruz FF Figueroa FE Rocco PRM Weiss DJ . Current status of cell-based therapies for respiratory virus infections: applicability to COVID-19. Eur Respir J (2020) 55(6). doi: 10.1183/13993003.00858-2020
185
Yen BL Yen ML Wang LT Liu KJ Sytwu HK . Current status of mesenchymal stem cell therapy for immune/inflammatory lung disorders: Gleaning insights for possible use in COVID-19. Stem Cells Trans Med (2020) 9(10):1163–73. doi: 10.1002/sctm.20-0186
186
Caplan AI . Mesenchymal stem cells: time to change the name! Stem Cells Trans Med (2017) 6(6):1445–51. doi: 10.1002/sctm.17-0051
187
Crivelli B Chlapanidas T Perteghella S Lucarelli E Pascucci L Brini AT et al . Mesenchymal stem/stromal cell extracellular vesicles: From active principle to next generation drug delivery system. J Controlled Release (2017) 262:104–17. doi: 10.1016/j.jconrel.2017.07.023
188
Qu W Wang Z Hare JM Bu G Mallea JM Pascual JM et al . Cell-based therapy to reduce mortality from COVID-19: Systematic review and meta-analysis of human studies on acute respiratory distress syndrome. Stem Cells Trans Med (2020) 9(9):1007–22. doi: 10.1002/sctm.20-0146
189
Kallmeyer K Pepper MS . Homing properties of mesenchymal stromal cells. Expert Opin Biol Ther (2015) 15(4):477–9. doi: 10.1517/14712598.2015.997204
190
Qu W Wang Z Engelberg-Cook E Yan D Siddik AB Bu G et al . Efficacy and safety of MSC cell therapies for hospitalized patients with COVID-19: A systematic review and meta-analysis. Stem Cells Trans Med (2022) 11(7):688–703. doi: 10.1093/stcltm/szac032
191
Sengul F Ozturk B Vatansev H . Mesenchymal stem cell therapy for COVID-19. Am J Stem Cells (2021) 10(5):79–89.
192
Zanirati G Provenzi L Libermann LL Bizotto SC Ghilardi IM Marinowic DR et al . Stem cell-based therapy for COVID-19 and ARDS: a systematic review. NPJ Regenerative Med (2021) 6(1):73. doi: 10.1038/s41536-021-00181-9
193
Iglesias M Butrón P Torre-Villalvazo I Torre-Anaya EA Sierra-Madero J Rodriguez-Andoney JJ et al . Mesenchymal stem cells for the compassionate treatment of severe acute respiratory distress syndrome due to COVID 19. Aging Dis (2021) 12(2):360–70. doi: 10.14336/AD.2020.1218
194
Tang L Jiang Y Zhu M Chen L Zhou X Zhou C et al . Clinical study using mesenchymal stem cells for the treatment of patients with severe COVID-19. Front Med (2020) 14(5):664–73. doi: 10.1007/s11684-020-0810-9
195
Xu R Feng Z Wang FS . Mesenchymal stem cell treatment for COVID-19. EBioMedicine (2022) 77:103920. doi: 10.1016/j.ebiom.2022.103920
196
Hashemian SR Aliannejad R Zarrabi M Soleimani M Vosough M Hosseini SE et al . Mesenchymal stem cells derived from perinatal tissues for treatment of critically ill COVID-19-induced ARDS patients: a case series. Stem Cell Res Ther (2021) 12(1):91. doi: 10.1186/s13287-021-02165-4
197
Xu X Jiang W Chen L Xu Z Zhang Q Zhu M et al . Evaluation of the safety and efficacy of using human menstrual blood-derived mesenchymal stromal cells in treating severe and critically ill COVID-19 patients: An exploratory clinical trial. Clin Trans Med (2021) 11(2):e297. doi: 10.1002/ctm2.297
198
Sadeghi B Roshandel E Pirsalehi A Kazemi S Sankanian G Majidi M et al . Conquering the cytokine storm in COVID-19-induced ARDS using placenta-derived decidua stromal cells. J Cell Mol Med (2021) 25(22):10554–64. doi: 10.1111/jcmm.16986
199
Escacena N Quesada-Hernández E Capilla-Gonzalez V Soria B Hmadcha A . Bottlenecks in the efficient use of advanced therapy medicinal products based on mesenchymal stromal cells. Stem Cells Int (2015) 2015:895714. doi: 10.1155/2015/895714
200
Stenderup K Justesen J Clausen C Kassem M . Aging is associated with decreased maximal life span and accelerated senescence of bone marrow stromal cells. Bone (2003) 33(6):919–26. doi: 10.1016/j.bone.2003.07.005
201
Meng F Xu R Wang S Xu Z Zhang C Li Y et al . Human umbilical cord-derived mesenchymal stem cell therapy in patients with COVID-19: a phase 1 clinical trial. Signal Transduction Targeted Ther (2020) 5(1):172. doi: 10.1038/s41392-020-00286-5
202
Saleh M Vaezi AA Aliannejad R Sohrabpour AA Kiaei SZF Shadnoush M et al . Cell therapy in patients with COVID-19 using Wharton’s jelly mesenchymal stem cells: a phase 1 clinical trial. Stem Cell Res Ther (2021) 12(1):410. doi: 10.1186/s13287-021-02483-7
203
Häberle H Magunia H Lang P Gloeckner H Körner A Koeppen M et al . Mesenchymal stem cell therapy for severe COVID-19 ARDS. J Intensive Care Med (2021) 36(6):681–8. doi: 10.1177/0885066621997365
204
Lanzoni G Linetsky E Correa D Messinger Cayetano S Alvarez RA Kouroupis D et al . Umbilical cord mesenchymal stem cells for COVID-19 acute respiratory distress syndrome: A double-blind, phase 1/2a, randomized controlled trial. Stem Cells Trans Med (2021) 10(5):660–73. doi: 10.1002/sctm.20-0472
205
Golchin A Farahany TZ Khojasteh A Soleimanifar F Ardeshirylajimi A . The clinical trials of mesenchymal stem cell therapy in skin diseases: an update and concise review. Curr Stem Cell Res Ther (2019) 14(1):22–33. doi: 10.2174/1574888X13666180913123424
206
Dutta AK . Vaccine against covid-19 disease - present status of development. Indian J Pediatrics (2020) 87(10):810–6. doi: 10.1007/s12098-020-03475-w
207
Rotshild V Hirsh-Raccah B Miskin I Muszkat M Matok I . Comparing the clinical efficacy of COVID-19 vaccines: a systematic review and network meta-analysis. Sci Rep (2021) 11(1):22777. doi: 10.1038/s41598-021-02321-z
208
Sharif N Alzahrani KJ Ahmed SN Dey SK . Efficacy, immunogenicity and safety of COVID-19 vaccines: A systematic review and meta-analysis. Front Immunol (2021) 12:714170. doi: 10.3389/fimmu.2021.714170
209
Chemaitelly H Bertollini R Abu-Raddad LJ . Efficacy of natural immunity against SARS-coV-2 reinfection with the beta variant. New Engl J Med (2021) 385(27):2585–6. doi: 10.1056/NEJMc2110300
210
Hansen CH Michlmayr D Gubbels SM Mølbak K Ethelberg S . Assessment of protection against reinfection with SARS-CoV-2 among 4 million PCR-tested individuals in Denmark in 2020: a population-level observational study. Lancet (London England) (2021) 397(10280):1204–12. doi: 10.1016/S0140-6736(21)00575-4
211
Pilz S Chakeri A Ioannidis JP Richter L Theiler-Schwetz V Trummer C et al . SARS-CoV-2 re-infection risk in Austria. Eur J Clin Invest (2021) 51(4):e13520. doi: 10.1111/eci.13520
212
Dehgani-Mobaraki P Zaidi AK Yadav N Floridi A Floridi E . Longitudinal observation of antibody responses for 14 months after SARS-CoV-2 infection. Clin Immunol (Orlando Fla) (2021) 230:108814. doi: 10.1016/j.clim.2021.108814
213
Gussarow D Bonifacius A Cossmann A Stankov MV Mausberg P Tischer-Zimmermann S et al . Long-lasting immunity against SARS-coV-2: dream or reality? Front Med (2021) 8:770381. doi: 10.3389/fmed.2021.770381
214
Rosati M Terpos E Ntanasis-Stathopoulos I Agarwal M Bear J Burns R et al . Sequential analysis of binding and neutralizing antibody in COVID-19 convalescent patients at 14 months after SARS-coV-2 infection. Front Immunol (2021) 12:793953. doi: 10.3389/fimmu.2021.793953
215
Kim P Gordon SM Sheehan MM Rothberg MB . Duration of severe acute respiratory syndrome coronavirus 2 natural immunity and protection against the delta variant: A retrospective cohort study. Clin Infect Dis (2022) 75(1):e185–e90. doi: 10.1093/cid/ciab999
216
Sheehan MM Reddy AJ Rothberg MB . Reinfection rates among patients who previously tested positive for coronavirus disease 2019: A retrospective cohort study. Clin Infect Dis (2021) 73(10):1882–6. doi: 10.1093/cid/ciab234
217
Pilz S Theiler-Schwetz V Trummer C Krause R Ioannidis JPA . SARS-CoV-2 reinfections: Overview of efficacy and duration of natural and hybrid immunity. Environ Res (2022) 209:112911. doi: 10.1016/j.envres.2022.112911
218
Bozio CH Grannis SJ Naleway AL Ong TC Butterfield KA DeSilva MB et al . Laboratory-confirmed COVID-19 among adults hospitalized with COVID-19-like illness with infection-induced or mRNA vaccine-induced SARS-coV-2 immunity - nine states, january-september 2021. MMWR Morbid Mortal Weekly Rep (2021) 70(44):1539–44. doi: 10.15585/mmwr.mm7044e1
219
Lumley SF Rodger G Constantinides B Sanderson N Chau KK Street TL et al . An observational cohort study on the incidence of severe acute respiratory syndrome coronavirus 2 (SARS-coV-2) infection and B.1.1.7 variant infection in healthcare workers by antibody and vaccination status. Clin Infect Dis (2022) 74(7):1208–19. doi: 10.1093/cid/ciab608
220
Shrestha NK Burke PC Nowacki AS Terpeluk P Gordon SM . Necessity of coronavirus disease 2019 (COVID-19) vaccination in persons who have already had COVID-19. Clin Infect Dis (2022) 75(1):e662–e71. doi: 10.1093/cid/ciac022
221
Aiello A Accardi G Candore G Carruba G Davinelli S Passarino G et al . Nutrigerontology: a key for achieving successful ageing and longevity. Immun Ageing (2016) 13:17. doi: 10.1186/s12979-016-0071-2
222
Cavanaugh AM Spicer KB Thoroughman D Glick C Winter K . Reduced risk of reinfection with SARS-coV-2 after COVID-19 vaccination - kentucky, may-june 2021. MMWR Morbid Mortal Weekly Rep (2021) 70(32):1081–3. doi: 10.15585/mmwr.mm7032e1
223
Abu-Raddad LJ Chemaitelly H Ayoub HH Yassine HM Benslimane FM Al Khatib HA et al . Association of prior SARS-coV-2 infection with risk of breakthrough infection following mRNA vaccination in Qatar. Jama (2021) 326(19):1930–9. doi: 10.1001/jama.2021.19623
224
Chen J Hu C Chen L Tang L Zhu Y Xu X et al . Clinical study of mesenchymal stem cell treatment for acute respiratory distress syndrome induced by epidemic influenza A (H7N9) infection: A hint for COVID-19 treatment. Eng (Beijing China) (2020) 6(10):1153–61. doi: 10.1016/j.eng.2020.02.006
225
Gallelli L Zhang L Wang T Fu F . Severe acute lung injury related to COVID-19 infection: A review and the possible role for escin. J Clin Pharmacol (2020) 60(7):815–25. doi: 10.1002/jcph.1644
226
da Silva KN Gobatto ALN Costa-Ferro ZSM Cavalcante BRR Caria ACI de Aragão França LS et al . Is there a place for mesenchymal stromal cell-based therapies in the therapeutic armamentarium against COVID-19? Stem Cell Res Ther (2021) 12(1):425. doi: 10.1186/s13287-021-02502-7
227
Raman R Patel KJ Ranjan K . COVID-19: unmasking emerging SARS-coV-2 variants, vaccines and therapeutic strategies. Biomolecules (2021) 11(7). doi: 10.3390/biom11070993
228
Wolfe CR Tomashek KM Patterson TF Gomez CA Marconi VC Jain MK et al . Baricitinib versus dexamethasone for adults hospitalised with COVID-19 (ACTT-4): a randomised, double-blind, double placebo-controlled trial. Lancet Respir Med (2022) 10(9):888–99. doi: 10.1016/S2213-2600(22)00088-1
Summary
Keywords
COVID-19, cytokine storm, immunomodulation, mesenchymal stromal cells, SARS-CoV-2, advanced therapies
Citation
Gonzaga A, Andreu E, Hernández-Blasco LM, Meseguer R, Al-Akioui-Sanz K, Soria-Juan B, Sanjuan-Gimenez JC, Ferreras C, Tejedo JR, Lopez-Lluch G, Goterris R, Maciá L, Sempere-Ortells JM, Hmadcha A, Borobia A, Vicario JL, Bonora A, Aguilar-Gallardo C, Poveda JL, Arbona C, Alenda C, Tarín F, Marco FM, Merino E, Jaime F, Ferreres J, Figueira JC, Cañada-Illana C, Querol S, Guerreiro M, Eguizabal C, Martín-Quirós A, Robles-Marhuenda Á, Pérez-Martínez A, Solano C and Soria B (2023) Rationale for combined therapies in severe-to-critical COVID-19 patients. Front. Immunol. 14:1232472. doi: 10.3389/fimmu.2023.1232472
Received
31 May 2023
Accepted
22 August 2023
Published
11 September 2023
Volume
14 - 2023
Edited by
Francisco J. Medrano, Virgen del Rocío University Hospital, Spain
Reviewed by
Gang Xu, Anhui Medical University, China; Olle Thor,Hans Ringden, Karolinska Institutet (KI), Sweden
Updates
Copyright
© 2023 Gonzaga, Andreu, Hernández-Blasco, Meseguer, Al-Akioui-Sanz, Soria-Juan, Sanjuan-Gimenez, Ferreras, Tejedo, Lopez-Lluch, Goterris, Maciá, Sempere-Ortells, Hmadcha, Borobia, Vicario, Bonora, Aguilar-Gallardo, Poveda, Arbona, Alenda, Tarín, Marco, Merino, Jaime, Ferreres, Figueira, Cañada-Illana, Querol, Guerreiro, Eguizabal, Martín-Quirós, Robles-Marhuenda, Pérez-Martínez, Solano and Soria.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Bernat Soria, bernat.soria@umh.es
†These authors have contributed equally to this work and share first authorship
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.