- Department of Rheumatology and Clinical Immunology, Peking University First Hospital, Beijing, China
Objective: The debate persists regarding whether patients with psoriatic arthritis (PsA) face an increased risk of mortality. We aimed to ascertain the magnitude of all-cause mortality risk in patients with PsA compared to the general population through a systematic review and meta-analysis.
Methods: We conducted a comprehensive search of PubMed, EMBASE, and the Cochrane Library for studies published from inception to June 2025. STATA meta-analysis software was used to calculate the pooled risk estimates for mortality, represented as the standardized mortality ratio (SMR).
Results: Among the 4,502 articles identified in our research, 20 studies were included in the analysis. Overall, our findings revealed a 1.12-fold increased risk of death among patients with PsA compared to the general population (meta-SMR: 1.12, 95% CI 1.09-1.15). Subgroup analyses showed that mortality risks were elevated in Asian countries (meta-SMR: 1.28, 95% CI 1.04-1.57), in population-based studies (meta-SMR: 1.13, 95% CI 1.02-1.25), and among studies including over 1,000 patients (meta-SMR: 1.12, 95% CI 1.01-1.25). Malignancy, cardiovascular and cerebrovascular diseases, and infection/respiratory diseases emerged as the most frequent causes of mortality.
Conclusion: Our analysis suggested modestly elevated mortality in patients with PsA compared to the general population, though heterogeneity warrants cautious interpretation. Malignancy, cardiovascular and cerebrovascular diseases, and infection/respiratory disease were frequent causes of mortality and warrant further investigation.
Systematic review registration: https://www.crd.york.ac.uk/prospero/, identifier CRD42021275209.
Introduction
Psoriatic arthritis (PsA) is a chronic inflammatory disease associated with psoriasis (1). It is characterized by diverse clinical features, including enthesitis, arthritis, dactylitis, and axial involvement. In addition to musculoskeletal manifestations, PsA is frequently associated with obesity, type 2 diabetes, hypertension, and metabolic syndrome (2). Nevertheless, whether patients with PsA face a heightened risk of mortality compared to the general population remains debated.
While numerous studies have investigated the prognosis of PsA, their findings remain inconsistent. A cohort study of 9,572 patients with PsA documented 682 deaths (7.1%) during a median follow-up of 6.46 years, revealing an adjusted standardized mortality ratio (SMR) of 1.47 versus general population controls (3). Several other studies reported similar findings, with SMRs ranging from 1.11 to 2.19 (4–9). Conversely, publications from other regions have found no significant association between PsA and increased mortality rates (10–12). A previous meta-analysis of 11 studies indicated that all-cause mortality was not increased among patients with PsA (13). However, several recent studies with large sample sizes have presented conflicting findings, prompting a re-evaluation of this important issue (9, 14–18). This meta-analysis aimed to quantify the risk of mortality in patients with PsA through systematic synthesis of observational studies.
This article is a revised and expanded version of a paper presented at the 69th Annual Scientific Meeting of the Japan College of Rheumatology (JCR 2025), held in Fukuoka, Japan (19).
Methods
This systematic review and meta-analysis adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (20), with a prospectively registered protocol (registration number: PROSPERO CRD42021275209). Three independent investigators (HH, WX, and YF) performed literature screening, data extraction, and quality assessment.
Strategies of literature search and inclusion criteria
PubMed, EMBASE, and the Cochrane Library were systematically searched through June 2025 using comprehensive terms for “psoriatic arthritis” and “mortality,” without date/language restrictions. We also searched major annual meetings—specifically the annual meetings of the American College of Rheumatology (ACR) and the European Congress of Rheumatology (EULAR)—from 2016 to 2024. Supplementary hand-searching included screening the reference lists of relevant reviews and eligible articles. No language restrictions were applied.
We included only population-based and cohort studies that reported all-cause mortality rates among patients with PsA and either compared these rates with those of the general population or matched controls, or directly reported the SMR.
In instances where duplicate data appeared in different studies, preference was given to the most up-to-date data. After eliminating duplicates, two independent researchers (HH and WX) conducted a thorough screening of all titles and abstracts, followed by a comprehensive evaluation of the complete texts of potentially relevant articles. Case reports, case series, reviews, commentaries, and editorials were excluded. The detailed search protocol is available in Supplementary Appendix S1.
Data extraction and quality assessment
Two investigators (HH and FY) independently performed data extraction using a standardized template. Extracted data included study characteristics such as authorship, publication year, geographic origin, research design, clinical setting, date source, enrollment period, and follow-up duration; cohort attributes such as sample size, sex distribution, mean age, diagnostic criteria, clinical manifestations, and serological parameters; and outcomes including all-cause mortality rates and reported mortality risk factors.
Discrepancies in data extraction were adjudicated by a third investigator (ZZ). Two independent assessors evaluated study quality using the Newcastle–Ottawa Scale (NOS), with quality stratification defined as: high (7–9 stars), moderate (4–6 stars), or low (0–3 stars) (21).
Data synthesis and statistical analysis
Meta-analytic computations were conducted using Stata version 15.1 (StataCorp LP), employing DerSimonian–Laird random-effects models to derive SMR with 95% confidence intervals (CIs). Statistical significance was determined using two-sided tests with α=0.05. Gender-stratified meta-SMRs were calculated for male and female subgroups.
Heterogeneity was quantified using the I² statistic and interpreted as low (≤25%), moderate (26%–50%), or substantial (>50%). Sources of heterogeneity were explored through predefined subgroup meta-analyses and sensitivity assessments. All included studies underwent prespecified stratification by sex, study design (prospective/retrospective cohort), geographic region, population setting (population-based vs. tertiary-care referral cohorts), and cohort size (≥1,000 vs. <1,000 patients). The primary outcome was all-cause mortality, expressed as SMR among patients with PsA relative to the general population.
Sensitivity analysis was performed using a leave-one-out meta-analysis to assess the influence of individual studies on the pooled estimates and to evaluate result robustness (22). Mortality risk factors in PsA were analyzed by pooling extracted risk estimates (95% CIs) using fixed-effects models. Given the low frequency of events, hazard ratios (HRs) were treated as risk ratios (RRs) to ensure consistency of effect size metrics. Forest plots were used to visualize pooled estimates, while funnel plot inspection and Egger’s/Begg’s tests quantitatively evaluated small-study effects (23).
Results
Study selection, characteristics, and quality assessment
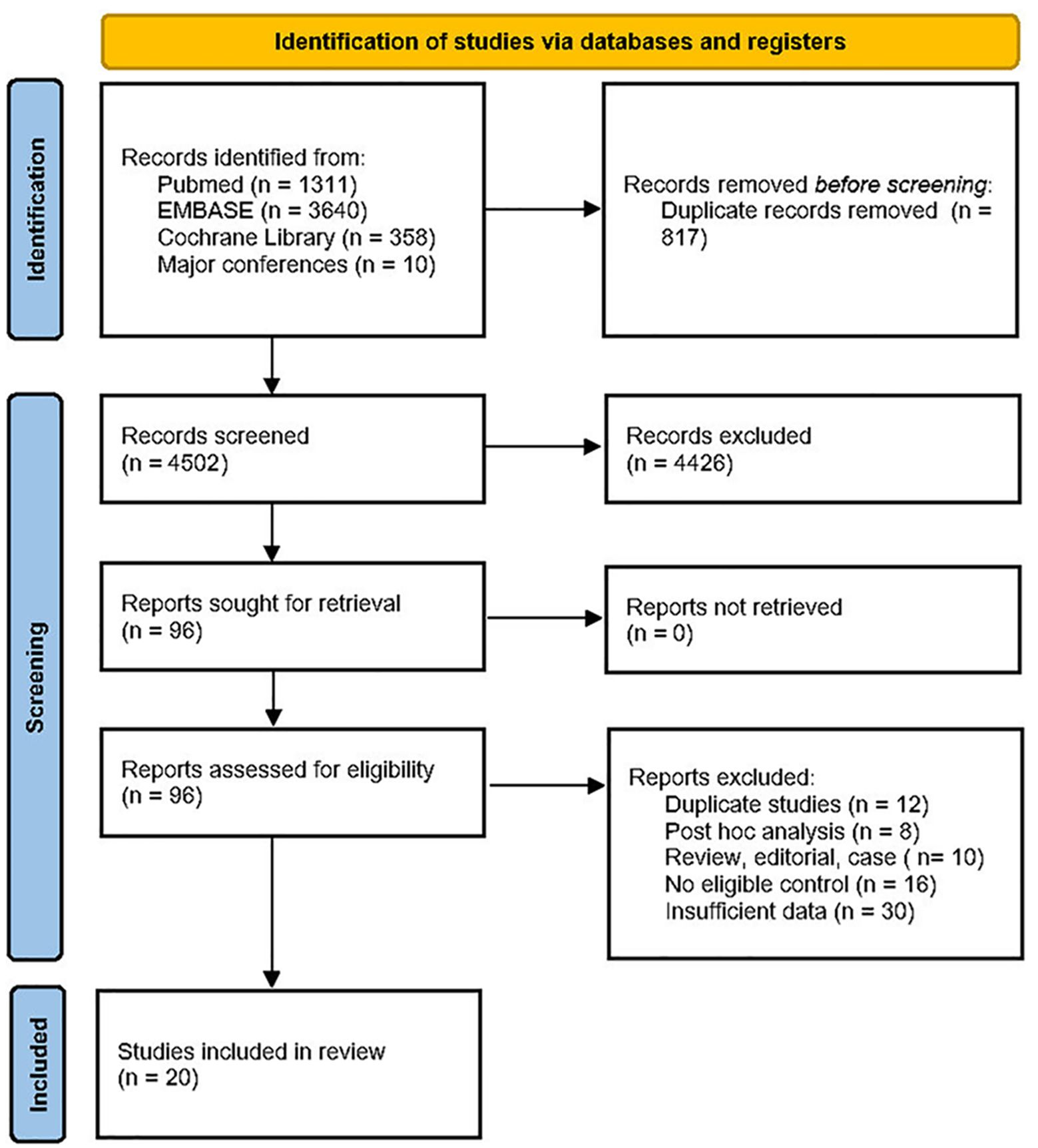
Initially, 4,502 articles were identified through the comprehensive search. After removing duplicates and screening titles and abstracts, 96 full-text articles were thoroughly assessed for eligibility. Ultimately, 20 studies involving more than 130,000 patients were deemed eligible for data extraction and analysis (3–12, 14–18, 24–28). The flowchart illustrating the study selection process is shown in Figure 1.
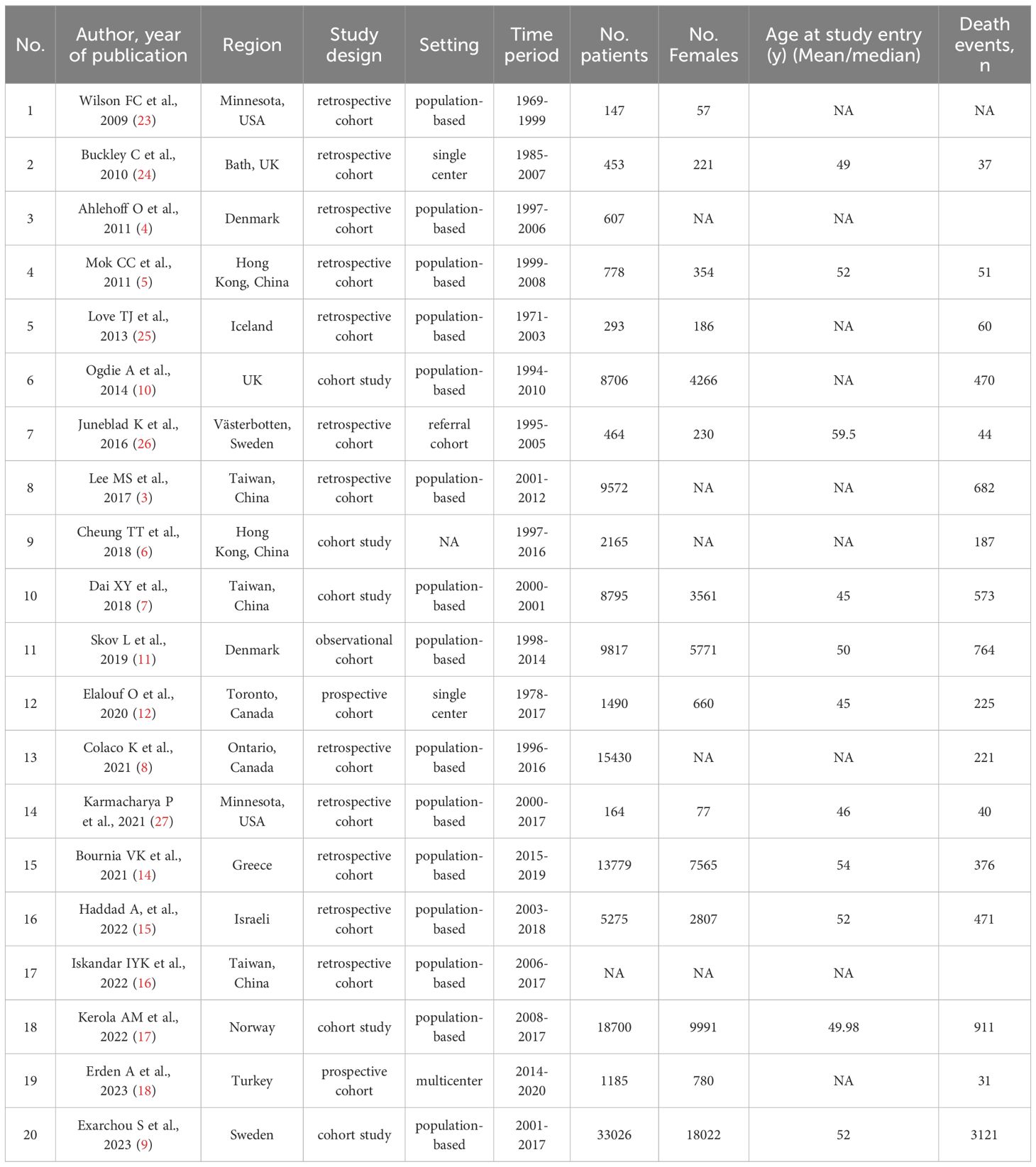
A summary of the main characteristics of the included studies is provided in Table 1. Of the 20 studies, 9 were conducted in European countries (4, 9–11, 14, 17, 25–27), 4 in North America (8, 12, 24, 28) and 7 in Asian countries (3, 5–7, 15, 16, 18). The mean age of patients at study entry ranged from 45 to 59 years. The proportion of female patients ranged from 39% to 66%.
Mortality in PsA patients
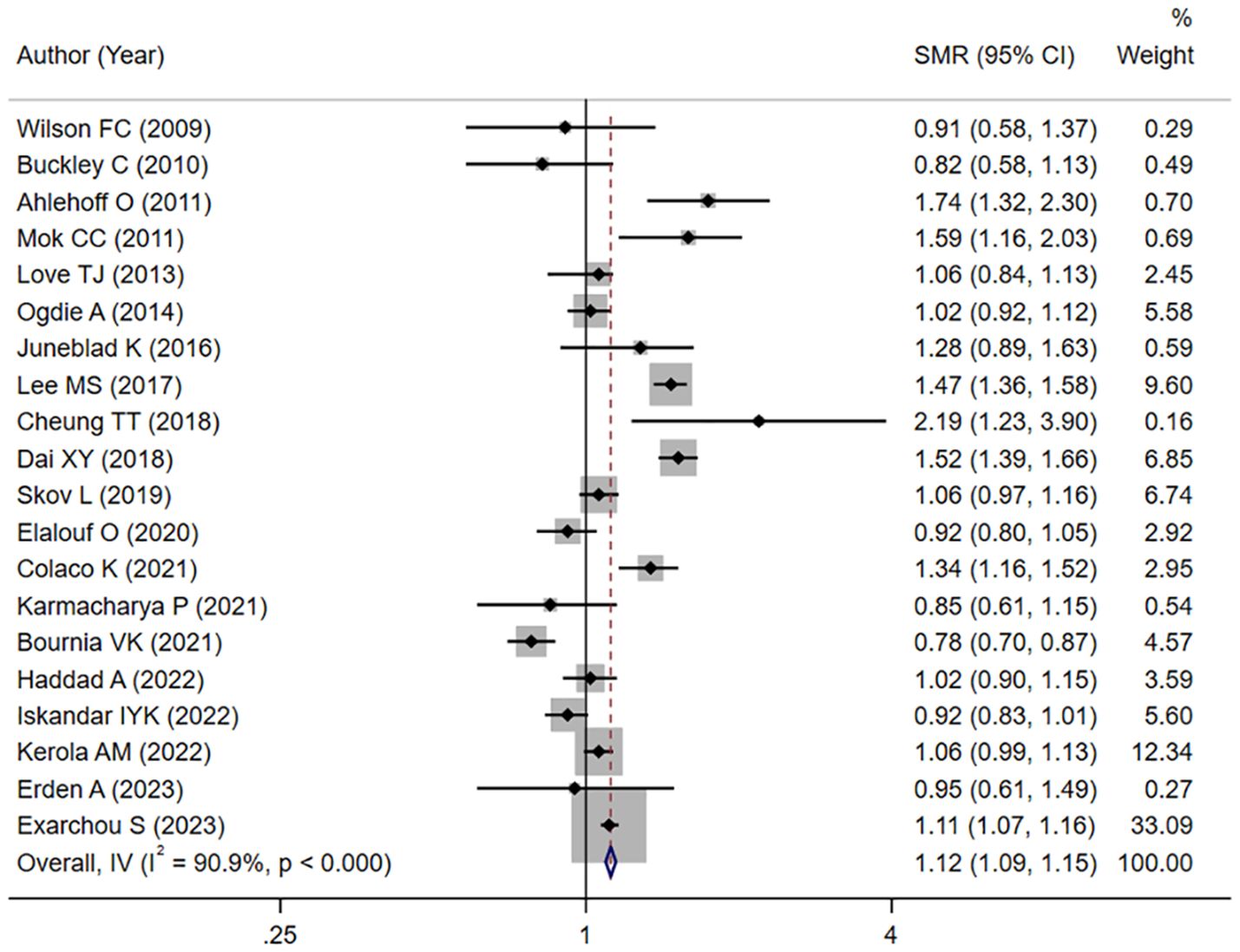
Overall, the mortality hazard among patients with PsA exhibited a significant elevation compared to the general population (meta-SMR: 1.12; 95% CI: 1.09–1.15), with substantial heterogeneity across the included studies (I2 = 90.9%, Figure 2).
Seven studies reported sex-specific mortality estimates. A higher summary meta-SMR was observed among female patients (meta-SMR: 1.18; 95% CI: 1.06–1.31; I²=63.4%), while no significant difference was observed among male patients (meta-SMR: 1.01; 95% CI: 0.82–1.25; I²=68.5%) (Supplementary Figure S1).
Subgroup analysis
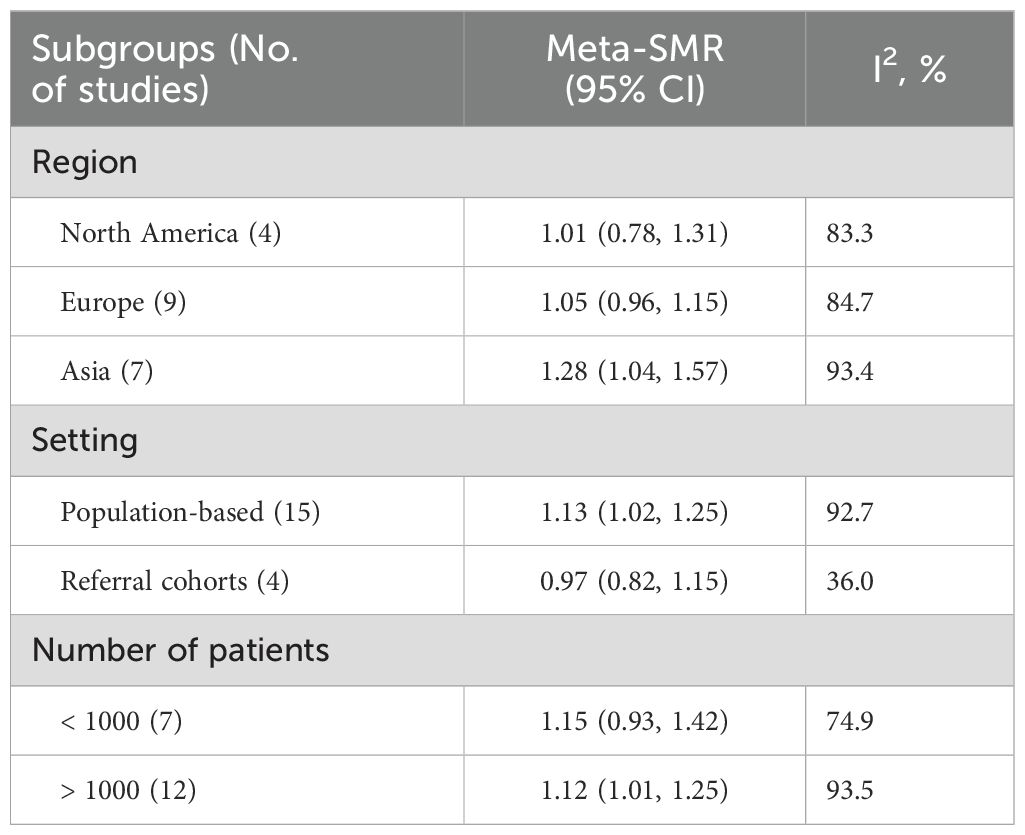
Subgroup analyses revealed that multiple factors may influence mortality risk. Elevated meta-SMRs were observed in studies from Asian countries (meta-SMR: 1.28; 95% CI: 1.04–1.57), population-based studies (meta-SMR: 1.13; 95% CI: 1.02–1.25), and studies involving more than 1,000 patients (meta-SMR: 1.12; 95% CI: 1.01–1.25). Notably, heterogeneity was reduced in analyses stratified by geographic region, study setting, and cohort size (Table 2 and Supplementary Figure S2).
Study quality assessment analysis and sensitivity
The median NOS score among the included studies was 7 (range: 5–8) (Supplementary Table S1). A study from Hong Kong involving more severe patients reported a particularly high SMR (meta-SMR: 2.19; 95% CI: 1.23–3.90) (6). When this study was excluded, the pooled meta-SMR was changed to 1.10 (95% CI: 1.01–1.21), with a corresponding I2 of 91.2%.
Predictors of mortality and causes of mortality
Risk factors associated with mortality were analyzed using demographic, clinical, and laboratory data from five studies with available data (Supplementary Table S2) (9, 12, 15, 18, 27). Individuals characterized by older age, male sex, elevated acute-phase reactants, and the presence of comorbidities appeared to be associated with increased mortality risk. However, due to limited data, meta-analysis of these predictors was not feasible.
A total of 11 studies reported the etiology of mortality among 6,063 individuals diagnosed with PsA. Malignancy (2,023 patients; 33.4%), cardiovascular and cerebrovascular diseases (1,743 patients; 28.8%), and infection/respiratory disease (543 patients; 9.0%) were the most frequent causes of death (Supplementary Table S2).
Publication bias
Funnel plot symmetry analysis using both the Mantel–Haenszel (M–H) fixed-effect method and the Peto method demonstrated no detectable publication bias across comparative analyses (Figure 3). Statistical evaluation using Egger’s linear regression test (p=0.966) and Begg’s rank correlation test (p=0.966) further confirmed the absence of significant asymmetry in the distribution of effect estimates.
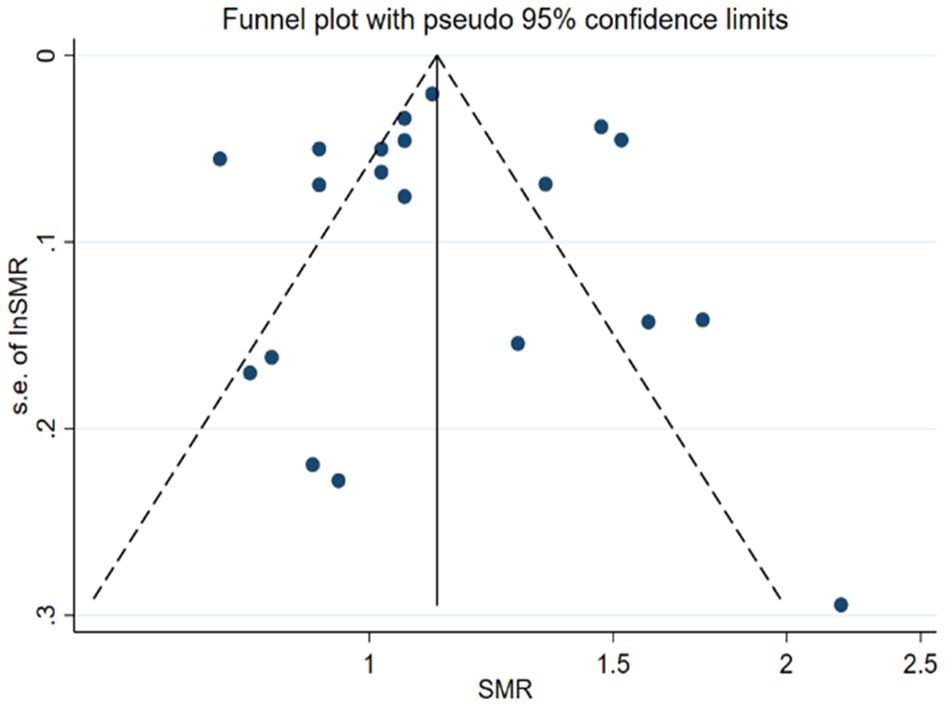
Figure 3. Funnel plot of 20 studies evaluating mortality of PsA patients compared with the general population.
Discussion
Substantial epidemiological evidence has established elevated mortality in several autoimmune conditions, notably systemic lupus erythematosus, Sjögren’s syndrome, and rheumatoid arthritis (29–31). However, the mortality risk associated with PsA remains a subject of debate. As a systemic autoimmune disorder, PsA involves multiple domains. Our meta-analysis revealed a 12% increase in mortality risk among patients with PsA compared to the general population.
Our analysis revealed a significantly elevated mortality risk in female patients with PsA relative to age-matched general population controls. A total of five studies reported that mortality or SMRs among female patients surpassed those of their male counterparts, indicating a potentially worse prognosis in women (5, 12, 17, 18, 25). However, this finding should be interpreted with caution due to the limited number of studies providing sex-specific SMRs. Further research is warranted.
Heterogeneity across studies was expected. Notably, compared to the general population, elevated mortality was observed among Asian patients with PsA, but not among patients in North America or Europe. This disparity could potentially be attributed to divergent treatment modalities, although the therapeutic strategies seemed similar across the studies.
We also noted a trend toward higher mortality risk in population-based studies compared with referral center cohorts. Additionally, the meta-analysis revealed elevated SMRs among studies involving over 1,000 patients. Given the publication of large-scale studies in recent years, this may explain the differences between our findings and those of earlier meta-analyses (13).
Our analysis demonstrated reduced overall life expectancy in patients with PsA compared to general population cohorts. Complications emerged as pivotal determinants contributing to the heightened mortality risk observed, particularly malignancy, cardiovascular and cerebrovascular diseases, and infection/respiratory disease, which were among the most frequently reported causes of death.
The association between PsA and cancer has been investigated in multiple studies. A systematic review and meta-analysis reported a significant association between psoriasis and overall cancer risk, but not between PsA and cancer (32). Conversely, a nationwide population-based cohort study in Korea found that individuals with PsA had a higher cancer risk than age- and sex-matched controls, especially for non-melanoma skin cancer (NMSC), lymphoma, and thyroid cancer (33). Similarly, a population-based study using the UK Clinical Practice Research Datalink reported an increased incidence of hematological cancers in patients with PsA (34). The chronic inflammatory nature of PsA may contribute to the elevated cancer risk by promoting the accumulation of somatic mutations and inducing various epigenetic changes during carcinogenesis (35). Lifestyle factors such as smoking and obesity—which are common risk factors for both PsA and cancer—may also play a role in increasing cancer risk among patients with PsA (36, 37). Notably, the use of biologic agents, including tumor necrosis factor inhibitors, has not been shown to increase cancer risk in PsA patients (32, 38). This calls for further investigation with longer follow-up and may serve as a reminder of the necessity of cancer screening in the management of PsA.
The elevated risk of cardiovascular disease (CVD) in PsA has been recognized for decades and has emerged as a major contributor to mortality, as evidenced by our findings and previous research (4, 17, 39). Chronic inflammation is known to consistently accelerate atherosclerosis, and systemic inflammatory markers have been strongly linked to cardiovascular events (40). Besides, as an important cytokine in the pathogenesis of PsA, IL-17 can contribute to the increased risk of cardiovascular events by affecting vascular and cardiac cells (41). Meng et al. demonstrated that systemic inflammatory markers independently increase the risk of major adverse cardiovascular events (MACE), underscoring the crucial role of inflammation in cardiovascular morbidity among PsA patients (42). Additionally, their research indicated that treatment with methotrexate and non-steroidal anti-inflammatory drugs (NSAIDs) was associated with a reduced risk of MACE, whereas the use of glucocorticoids appears to be harmful. Consequently, it is imperative to incorporate cardiovascular risk assessment in the therapeutic strategies for PsA patients.
In addition to malignancies and cardiovascular-related mortality, we also observed an increased mortality risk attributed to infections or respiratory diseases in patients with PsA. The expanding array of therapeutics available for PsA in recent years has prompted increased scrutiny regarding their safety profiles. Chiu et al. reported a heightened susceptibility to infections associated with targeted therapies compared to placebo in patients with PsA (43).
While previous studies have indicated that serious infections are rare among PsA patients treated with biologic or targeted disease-modifying antirheumatic drugs (DMARDs), it is noteworthy that most of these studies were based on randomized controlled trials, which are often characterized by strict participant selection criteria and relatively short follow-up durations (44, 45). Consequently, it is imperative to acknowledge the inherent limitations of such analyses in demonstrating an elevated risk of a rare side effect. Therefore, further real-world data are needed in the future.
Undoubtedly, several limitations of this meta-analysis warrant consideration. First, the included studies exhibited heterogeneity across multiple dimensions. Although most studies were of moderate to high quality, variability in study design and execution may have contributed to residual heterogeneity. We addressed this, to some extent, through prespecified subgroup analyses, sensitivity testing, and exclusion of an outlier study with a substantially elevated SMR. Importantly, the core finding of increased mortality risk in PsA patients relative to the general population remained consistent.
Second, the limited number of studies examining risk factors and causes of mortality warrants cautious interpretation of these findings. Finally, the lack of treatment-specific data across studies precluded any analysis of medication effects on disease progression and mortality outcomes.
Conclusion
Our investigation revealed a 1.12-fold increase in mortality risk among patients with PsA compared to the general population, although the presence of heterogeneity warrants cautious interpretation. Predominant causes of mortality included malignancies, cardiovascular and cerebrovascular diseases, as well as infection/respiratory conditions. Additional population-based studies—particularly those from diverse geographical regions and with extended follow-up durations—are needed to further elucidate mortality risk and its determinants in PsA.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Author contributions
HH: Data curation, Writing – original draft, Methodology, Investigation, Formal Analysis, Project administration. WX: Writing – original draft, Data curation, Formal Analysis. YG: Data curation, Formal Analysis, Writing – original draft. YF: Data curation, Writing – original draft, Formal Analysis. ZZ: Conceptualization, Project administration, Methodology, Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2025.1622159/full#supplementary-material
References
1. FitzGerald O, Ogdie A, Chandran V, Coates LC, Kavanaugh A, Tillett W, et al. Psoriatic arthritis. Nat Rev Dis Primers. (2021) 7:59. doi: 10.1038/s41572-021-00293-y
2. Ritchlin CT, Colbert RA, and Gladman DD. Psoriatic arthritis. New Engl J Med. (2017) 376:957–70. doi: 10.1056/NEJMra1505557
3. Lee MS, Yeh YC, Chang YT, and Lai MS. All-cause and cause-specific mortality in patients with psoriasis in Taiwan: A nationwide population-based study. J Invest Dermatol. (2017) 137:1468–73. doi: 10.1016/j.jid.2017.01.036
4. Ahlehoff O, Gislason GH, Charlot M, Jørgensen CH, Lindhardsen J, Olesen JB, et al. Psoriasis is associated with clinically significant cardiovascular risk: a Danish nationwide cohort study. J Internal Med. (2011) 270:147–57. doi: 10.1111/j.1365-2796.2010.02310.x
5. Mok CC, Kwok CL, Ho LY, Chan PT, and Yip SF. Life expectancy, standardized mortality ratios, and causes of death in six rheumatic diseases in Hong Kong, China. Arthritis rheumatism. (2011) 63:1182–9. doi: 10.1002/art.30277
6. Cheung TT, Tsoi SMF, Cheung BMY, and Lau CS. Mortality and causes of death in patients with inflammatory arthritis in Hong Kong over 20 years. Arthritis Rheumatol. (2018) 70:223–4. doi: 10.1002/art.40700
7. Dai YX, Hsu MC, Hu HY, Chang YT, Chen TJ, Li CP, et al. The risk of mortality among psoriatic patients with varying severity: A nationwide population-based cohort study in Taiwan. Int J Environ Res Public Health. (2018) 15:2622. doi: 10.3390/ijerph15122622
8. Colaco K, Widdifield J, Luo J, Rosen CF, Alhusayen R, Paterson JM, et al. Trends in mortality and cause-specific mortality among patients with psoriasis and psoriatic arthritis in Ontario, Canada. J Am Acad Dermatol. (2021) 84:1302–9. doi: 10.1016/j.jaad.2020.10.031
9. Exarchou S, Di Giuseppe D, Klingberg E, Sigurdardottir V, Wedrén S, Lindström U, et al. Mortality in patients with psoriatic arthritis in Sweden: a nationwide, population-based cohort study. Ann rheumatic Dis. (2023) 83:446–56. doi: 10.1136/ard-2023-224965
10. Ogdie A, Haynes K, Troxel AB, Love TJ, Hennessy S, Choi H, et al. Risk of mortality in patients with psoriatic arthritis, rheumatoid arthritis and psoriasis: a longitudinal cohort study. Ann rheumatic Dis. (2014) 73:149–53. doi: 10.1136/annrheumdis-2012-202424
11. Skov L, Thomsen SF, Kristensen LE, Dodge R, Hedegaard MS, and Kjellberg J. Cause-specific mortality in patients with psoriasis and psoriatic arthritis. Br J Dermatol. (2019) 180:100–7. doi: 10.1111/bjd.16919
12. Elalouf O, Muntyanu A, Polachek A, Pereira D, Ye JY, Lee KA, et al. Mortality in psoriatic arthritis: Risk, causes of death, predictors for death. Semin Arthritis rheumatism. (2020) 50:571–5. doi: 10.1016/j.semarthrit.2020.04.001
13. Chaudhary H, Bohra N, Syed K, Donato A, Murad MH, and Karmacharya P. All-cause and cause-specific mortality in psoriatic arthritis and ankylosing spondylitis: A systematic review and meta-analysis. Arthritis Care Res. (2023) 75:1052–65. doi: 10.1002/acr.24820
14. Bournia VK, Fragoulis GE, Mitrou P, Mathioudakis K, Tsolakidis A, Konstantonis G, et al. All-cause mortality in systemic rheumatic diseases under treatment compared with the general population, 2015-2019. RMD Open. (2021) 7:e001694. doi: 10.1136/rmdopen-2021-001694
15. Haddad A, Saliba W, Lavi I, Batheesh A, Kasem S, Gazitt T, et al. The association of psoriatic arthritis with all-cause mortality and leading causes of death in psoriatic arthritis. J Rheumatol. (2022) 49:165–70. doi: 10.3899/jrheum.210159
16. Iskandar IYK, Chen TC, Chen LC, Lee MS, Yang YY, Wang TC, et al. Incidence, prevalence, and mortality of people with psoriasis and psoriatic arthritis in Taiwan: A nationwide cohort study. Acta dermato-venereologica. (2022) 102:adv00807. doi: 10.2340/actadv.v102.1962
17. Kerola AM, Kazemi A, Rollefstad S, Lillegraven S, Sexton J, Wibetoe G, et al. All-cause and cause-specific mortality in rheumatoid arthritis, psoriatic arthritis and axial spondyloarthritis: a nationwide registry study. Rheumatol (Oxford England). (2022) 61:4656–66. doi: 10.1093/rheumatology/keac210
18. Erden A, Ayan G, Kilic L, Solmaz D, Bakirci S, Kimyon G, et al. Mortality in psoriatic arthritis patients, changes over time, and the impact of COVID-19: results from a multicenter Psoriatic Arthritis Registry (PsART-ID). Clin Rheumatol. (2023) 42:385–90. doi: 10.1007/s10067-022-06492-6
19. Hong Huang ZZ. Mortality in patients with psoriatic arthritis: A systematic review and meta-analysis. In: The 69th Annual Scientific Meeting of the Japan College of Rheumatology. Fukuoka, Japan (2025). p. S218.
20. Moher D, Liberati A, Tetzlaff J, and Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PloS Med. (2009) 6:e1000097. doi: 10.1371/journal.pmed.1000097
21. Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. (2010) 25:603–5. doi: 10.1007/s10654-010-9491-z
22. Li L, Shen J, Bala MM, Busse JW, Ebrahim S, Vandvik PO, et al. Incretin treatment and risk of pancreatitis in patients with type 2 diabetes mellitus: systematic review and meta-analysis of randomised and non-randomised studies. BMJ (Clinical Res ed.). (2014) 348:g2366. doi: 10.1136/bmj.g2366
23. Egger M, Davey Smith G, Schneider M, and Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ (Clinical Res ed.). (1997) 315:629–34. doi: 10.1136/bmj.315.7109.629
24. Wilson FC, Icen M, Crowson CS, McEvoy MT, Gabriel SE, and Kremers HM. Time trends in epidemiology and characteristics of psoriatic arthritis over 3 decades: a population-based study. J Rheumatol. (2009) 36:361–7. doi: 10.3899/jrheum.080691
25. Buckley C, Cavill C, Taylor G, Kay H, Waldron N, Korendowych E, et al. Mortality in psoriatic arthritis - a single-center study from the UK. J Rheumatol. (2010) 37:2141–4. doi: 10.3899/jrheum.100034
26. Love TJ, Aspelund T, Ogdie A, Gelfand JM, Choi HK, Gudnason V, et al. Psoriatic arthritis and mortality-a nationwide study. Arthritis rheumatism. (2013) 65:S441. doi: 10.1002/art.38216
27. Juneblad K, Rantapää-Dahlqvist S, and Alenius GM. Disease activity and increased risk of cardiovascular death among patients with psoriatic arthritis. J Rheumatol. (2016) 43:2155–61. doi: 10.3899/jrheum.160070
28. Karmacharya P, Crowson CS, Bekele D, Achenbach SJ, Davis JM 3rd, Ogdie A, et al. The epidemiology of psoriatic arthritis over five decades: A population-based study. Arthritis Rheumatol (Hoboken N.J.). (2021) 73:1878–85. doi: 10.1002/art.41741
29. Yurkovich M, Vostretsova K, Chen W, and Aviña-Zubieta JA. Overall and cause-specific mortality in patients with systemic lupus erythematosus: a meta-analysis of observational studies. Arthritis Care Res. (2014) 66:608–16. doi: 10.1002/acr.22173
30. Huang H, Xie W, Geng Y, Fan Y, and Zhang Z. Mortality in patients with primary Sjögren’s syndrome: a systematic review and meta-analysis. Rheumatol (Oxford England). (2021) 60:4029–38. doi: 10.1093/rheumatology/keab364
31. Dadoun S, Zeboulon-Ktorza N, Combescure C, Elhai M, Rozenberg S, Gossec L, et al. Mortality in rheumatoid arthritis over the last fifty years: systematic review and meta-analysis. Joint Bone Spine. (2013) 80:29–33. doi: 10.1016/j.jbspin.2012.02.005
32. Vaengebjerg S, Skov L, Egeberg A, and Loft ND. Prevalence, incidence, and risk of cancer in patients with psoriasis and psoriatic arthritis: A systematic review and meta-analysis. JAMA Dermatol. (2020) 156:421–9. doi: 10.1001/jamadermatol.2020.0024
33. Eun Y, Hwang J, Seo GH, and Ahn JK. Risk of cancer in Korean patients with psoriatic arthritis: a nationwide population-based cohort study. RMD Open. (2023) 9:e002874. doi: 10.1136/rmdopen-2022-002874
34. Hagberg KW, Li L, Peng M, Paris M, Shah K, and Jick SS. Rates of cancers and opportunistic infections in patients with psoriatic arthritis compared with patients without psoriatic arthritis. J Clin rheumatology: Pract Rep rheumatic musculoskeletal Dis. (2016) 22:241–7. doi: 10.1097/RHU.0000000000000364
35. Greten FR and Grivennikov SI. Inflammation and cancer: triggers, mechanisms, and consequences. Immunity. (2019) 51:27–41. doi: 10.1016/j.immuni.2019.06.025
36. Nguyen UDT, Zhang Y, Lu N, Louie-Gao Q, Niu J, Ogdie A, et al. Smoking paradox in the development of psoriatic arthritis among patients with psoriasis: a population-based study. Ann rheumatic Dis. (2018) 77:119–23. doi: 10.1136/annrheumdis-2017-211625
37. Love TJ, Zhu Y, Zhang Y, Wall-Burns L, Ogdie A, Gelfand JM, et al. Obesity and the risk of psoriatic arthritis: a population-based study. Ann rheumatic Dis. (2012) 71:1273–7. doi: 10.1136/annrheumdis-2012-201299
38. Hellgren K, Dreyer L, Arkema EV, Glintborg B, Jacobsson LT, Kristensen LE, et al. Cancer risk in patients with spondyloarthritis treated with TNF inhibitors: a collaborative study from the ARTIS and DANBIO registers. Ann rheumatic Dis. (2017) 76:105–11. doi: 10.1136/annrheumdis-2016-209270
39. Polachek A, Touma Z, Anderson M, and Eder L. Risk of cardiovascular morbidity in patients with psoriatic arthritis: A meta-analysis of observational studies. Arthritis Care Res. (2017) 69:67–74. doi: 10.1002/acr.22926
40. Ridker PM, Buring JE, Cook NR, and Rifai N. C-reactive protein, the metabolic syndrome, and risk of incident cardiovascular events: an 8-year follow-up of 14–719 initially healthy American women. Circulation. (2003) 107:391–7. doi: 10.1161/01.CIR.0000055014.62083.05
41. Beringer A and Miossec P. Systemic effects of IL-17 in inflammatory arthritis. Nat Rev Rheumatol. (2019) 15:491–501. doi: 10.1038/s41584-019-0243-5
42. Meng H, Lam SH, So H, and Tam LS. Incidence and risk factors of major cardiovascular events in rheumatoid arthritis and psoriatic arthritis: A population-based cohort study. Semin Arthritis rheumatism. (2024) 65:152416. doi: 10.1016/j.semarthrit.2024.152416
43. Chiu HY, Hung YT, and Huang YH. Comparative short-term risks of infection and serious infection in patients receiving biologic and small-molecule therapies for psoriasis and psoriatic arthritis: a systemic review and network meta-analysis of randomized controlled trials. Ther Adv chronic Dis. (2023) 14:20406223231206225. doi: 10.1177/20406223231206225
44. Séauve M, Auréal M, Laplane S, Lega JC, Cabrera N, and Coury F. Risk of infections in psoriatic arthritis or axial spondyloarthritis patients treated with targeted therapies: A meta-analysis of randomized controlled trials. Joint Bone Spine. (2023) 91:105673. doi: 10.1016/j.jbspin.2023.105673
45. Aureal M, Seauve M, Laplane S, Lega JC, Cabrera N, and Coury F. Incidence of infections in patients with psoriatic arthritis and axial spondyloarthritis treated with biological or targeted disease-modifying agents: a systematic review and meta-analysis of randomised controlled trials, open-label studies and observational studies. RMD Open. (2023) 9:e003064. doi: 10.1136/rmdopen-2023-003064
Keywords: psoriatic arthritis, systematic review, meta-analysis, mortality, causes of mortality
Citation: Huang H, Xie W, Geng Y, Fan Y and Zhang Z (2025) Mortality in patients with psoriatic arthritis: a systematic review and meta-analysis. Front. Immunol. 16:1622159. doi: 10.3389/fimmu.2025.1622159
Received: 02 May 2025; Accepted: 21 July 2025;
Published: 18 August 2025.
Edited by:
James Cheng-Chung Wei, Chung Shan Medical University Hospital, TaiwanReviewed by:
Chun-Ting Lin, Chung Shan Medical University Hospital, TaiwanQianru Zhang, Beijing Tsinghua Changgung Hospital, China
Copyright © 2025 Huang, Xie, Geng, Fan and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhuoli Zhang, emh1b2xpLnpoYW5nQDEyNi5jb20=
 Hong Huang
Hong Huang Wenhui Xie
Wenhui Xie Yan Geng
Yan Geng Yong Fan
Yong Fan Zhuoli Zhang
Zhuoli Zhang