- 1College of Veterinary Medicine, Huazhong Agricultural University, Wuhan, China
- 2Institute of Animal Sciences, Chinese Academy of Agricultural Sciences, Beijing, China
- 3State Key Laboratory for Animal Disease Control and Prevention, Harbin Veterinary Research Institute, Chinese Academy of Agricultural Sciences, Harbin, China
- 4Faculty of Pharmacy, Gomal University, Dera Ismail Khan, Khyber Pukhtunkhwa, Pakistan
- 5Department of Clinical Laboratory Sciences, College of Applied Medical Science, King Khalid University, Abha, Saudi Arabia
- 6Department of Clinical Laboratories Sciences, College of Applied Medical Sciences, Taif University, Taif, Saudi Arabia
- 7Department of Cell Biology, School of Life Sciences, Central South University, Changsha, China
- 8School of Medicine, College of Medicine, National Sun Yat-sen University, Kaohsiung, Taiwan
- 9Institute of Clinical Medicine, College of Medicine, National Cheng Kung University, Tainan, Taiwan
- 10Department of Pathology, Chi Mei Medical Center, Tainan, Taiwan
- 11Department of Pathology, Ditmanson Medical Foundation Chia-Yi Christian Hospital, Chiayi, Taiwan
- 12Department of Cosmetic Science, Chia Nan University of Pharmacy and Science, Tainan, Taiwan
- 13Doctoral Program in Translational Medicine, National Chung Hsing University, Taichung, Taiwan
- 14Department of Biotechnology and Bioindustry Sciences, College of Bioscience and Biotechnology, National Cheng Kung University, Tainan, Taiwan
Regulatory T cells (Tregs) are a unique subset of T cells vital for maintaining immune balance, preventing autoimmune diseases, and controlling immune responses. First identified in the early 1990s, Tregs are now well recognized for their role in suppressing excessive immune reactions and promoting tolerance to the body’s tissues. Among the broader Treg population, Tissue regulatory T cells (Tissue Tregs) are distinct as they do more than suppress immunity; they actively contribute to tissue repair and regeneration. Studies in both mice and humans have highlighted the important role of in aiding tissue repair and maintaining tissue integrity. Recent research reveals that Tregs participate in wound healing and tissue regeneration across various organs, including the heart, liver, kidneys, muscles, lungs, bones, and central nervous system. These discoveries emphasize the wide-ranging and significant influence of Tregs in fostering recovery and healing in different tissues throughout the body. These cells are characterized by their ability to produce a variety of growth factors, cytokines, and signaling molecules that support the repair and regeneration of damaged tissues. In this review, we present an overview of the emerging understanding of Treg-mediated repair processes in damaged tissues and organs.
Introduction
The worldwide rise in organ dysfunction caused by acute injuries, chronic diseases, or aging is increasing the need for organ transplants. However, the limited availability of donor organs and the use of immunosuppressive drugs pose significant challenges (1), leading to the search for alternative therapies. Recent advances in human pluripotent stem cell research, known for their ability to self-renew and differentiate into various cell types, offer a potentially limitless source of therapeutic cells for transplantation (2, 3). Despite this potential, there is limited clinical evidence of their long-term survival after transplant, possibly due to issues like poor cell viability and ongoing immune rejection (4, 5). Additionally, regenerative therapies focused on natural tissue repair, such as growth factor-based methods, have shown mixed results in clinical trials because of safety and cost concerns (6, 7). Therefore, it is crucial to develop strategies to improve tissue repair and regeneration.
Traditionally, the immune system has been seen mainly as a defense against pathogens, a view that has shaped how it appears in textbooks for over a century (8). However, beyond its protective role, the immune system is now understood as a crucial part of maintaining tissue homeostasis and supporting physiological processes like development, reproduction, wound healing, and tissue regeneration (9). Recent studies have emphasized the important role of innate immunity, especially the different polarization states of macrophages, in coordinating the complex events needed for tissue repair and regeneration (10, 11). Still, new evidence suggests that the adaptive immune system, particularly tissue Tregs, also has a key role in these processes (12, 13).
Forkhead box P3-expressing (Foxp3+) Tregs have long been recognized for their role in modulating immune responses and maintaining immune homeostasis (14). These cells, traditionally seen as suppressors of excessive immune activation, are now understood to have non-traditional functions that go beyond immune regulation (15, 16). Recent studies using transgenic mouse models, in which Tregs were specifically removed, have revealed their important roles in influencing non-immunological processes, including tissue repair and regeneration (15, 17). This new evidence indicates that Tregs are crucial in promoting repair and regeneration within injured tissues (18). A particularly intriguing aspect of Treg biology is their organ-specific functionality, with recent studies showing how tissue-resident Tregs respond to injury tailored to each organ’s distinct microenvironment (19, 20). This organ-specific behavior of Tregs highlights their role in tissue repair, not only as suppressors of inflammation but also as active contributors to the regeneration of damaged tissues (9, 11, 21). Treg cells not only suppress immune responses but also promote tissue repair by secreting pro-repair mediators. These factors act directly on resident structural and parenchymal cells within a tissue-specific context (22).
In this review, we will focus on the emerging roles of Tregs in tissue repair and regeneration, emphasizing their ability to modulate inflammation, promote healing, and interact with other immune cells. By exploring recent advancements in our understanding of tissue-resident Tregs and their functions, this review aims to shed light on the complex interplay between tissue Tregs and tissue regeneration, offering insights that could lead to new therapeutic strategies for enhancing tissue repair and restoring function more effectively.
Treg diversity
Tregs are essential for immune regulation and exhibit considerable diversity. Besides the well-known CD25 marker, they express a range of activation markers, including both co-stimulatory and co-inhibitory molecules such as PD-1, ICOS, LAG-3, CD27, CD69, LAP, and CTLA-4. They can also express members of the TNF receptor superfamily, like GITR and OX40, along with adhesion-related markers such as CD62L and CD49b. Additionally, they possess receptors involved in guiding their migration to peripheral tissues, including CCR7, CCR4, CCR6, CD103, and CCR5 (23). Foxp3 is a key transcription factor primarily expressed in CD4+ CD25+ Tregs in mice. It is present at minimal levels in CD4+ CD25- effector T cells and is mainly absent in CD8+ T cells (24, 25). Foxp3 is crucial for Treg development and their capacity to suppress immune responses (24–27). In mice, Tregs are typically identified as CD4+ CD25high Foxp3+. However, in humans, not all Foxp3-expressing cells display high CD25 levels (23), nor do they all possess immunosuppressive functions (28).
In humans, the expression of the IL-7 receptor alpha chain (CD127) is inversely related to Foxp3 expression. CD4+ CD127low T cells in humans exhibit similar suppressive functions to CD4+ CD25high T cells observed in laboratory settings (29). Therefore, human Tregs are more accurately defined as CD4+ CD25high CD127low/- Foxp3+. While this constitutes the core phenotype of Tregs, studies utilizing flow cytometry and RNA sequencing have demonstrated that both human and mouse Tregs are highly diverse (30). Their origin can also be used to categorize Tregs. Thymic Tregs (tTregs) are derived from CD4+ CD25high thymocytes that undergo positive selection after their T-cell receptors (TCRs) engage with high-affinity self-peptides presented by antigen-presenting cells and medullary thymic epithelial cells on MHC class II molecules. This process, combined with signals from cytokines such as IL-2 or IL-15 and TGF-β, promotes the differentiation of these cells into fully committed tTregs (31, 32). Peripheral Tregs (pTregs) develop from naive CD4+ Foxp3- T cells in secondary lymphoid organs and peripheral tissues, in response to cytokines like TGF-β upon encountering antigens (33).
Moreover, Tregs can be further classified by their differentiation stage (e.g., naive Tregs marked by CD45RA+ or activated Tregs) and their activation status (e.g., quiescent central Tregs characterized by CD44low CD62Lhigh or effector Tregs with CD44high CD62Llow). They can also be identified based on the expression of transcription factors that define specific T helper cell lineages, such as T-bet for Th1, Gata3 for Th2, and RoRγt for Th17 (30, 34). In recent years, the role of Tregs localized in non-lymphoid tissues has garnered increasing interest, extending beyond their traditional immune-suppressive functions. Tregs are present in various tissues under steady-state conditions and often accumulate following tissue injury. They are essential for maintaining tissue homeostasis and supporting repair through interactions with local tissue cells. These Tregs, called “tissue Tregs,’ share common features across different tissues but also display tissue-specific characteristics that suit the needs of each environment (35). Tissue-resident Tregs possess a limited T cell receptor (TCR) repertoire, likely recognizing self-antigens unique to the tissues they inhabit, such as fat tissue, muscle tissue, skin, lung tissue, intestines, and other non-lymphoid tissues. They exhibit distinct phenotypes compared to those in lymphoid tissues (18, 36).
Tregs in skeletal muscle repair and regeneration
The role of Tregs in skeletal muscle repair and regeneration has become an increasingly important topic in immunology and regenerative biology. Originally known for their ability to suppress immune responses and maintain self-tolerance, Tregs are now seen as active players in tissue repair processes, including those in skeletal muscle (10, 11, 37, 38). Their regenerative functions have been documented in organs such as the skin (21, 39–41), lungs (42), heart (43–45), and intestines (46), and it is becoming clearer that Tregs also have a vital role in skeletal muscle regeneration after both acute and chronic injuries. Skeletal muscle has a notable capacity to regenerate following injury, mainly due to the presence of muscle stem cells, or satellite cells (MuSCs), located between the basal lamina and sarcolemma of muscle fibers (47–49). The process of muscle regeneration involves several precisely coordinated steps, including inflammation (50, 51), stem cell activation (52), differentiation (52, 53), and tissue remodeling (47). The immune system is crucial to this regenerative process, with immune cells not only helping clear debris and fight infection but also regulating MuSC activity through cytokines, growth factors, and direct cell interactions (53–55).
Following muscle injury, there is a rapid and temporary infiltration of various immune cells. Among these, a distinct subset of CD4+Foxp3+ Tregs accumulates at the injury site. Murine models of muscle damage, such as cardiotoxin-induced injury, show a significant rise in Treg frequency in the injured tissue within the first few days after injury (11). These muscle resident Tregs are phenotypically and functionally different from those found in lymphoid tissues, with unique transcriptional and epigenetic signatures compared to lymphoid organ Tregs. Single-cell RNA sequencing (scRNA-seq) studies have revealed that muscle Tregs form a distinct cluster enriched for genes involved in tissue repair, such as amphiregulin (AREG), Il1rl1 (encoding ST2), and Gata3, as well as increased expression of BATF and IRF4 transcription factors linked to tissue adaptation and repair (11, 56, 57). Muscle-resident Tregs are not only present in experimental injury models but have also been identified in chronic muscle disorders. In conditions like Duchenne muscular dystrophy (modeled by mdx mice) and myotonic dystrophy in humans, an accumulation of Tregs with similar gene expression patterns has been observed (56, 58). This supports the idea that skeletal muscle contains a reparative Treg population that reacts dynamically to injury and ongoing degeneration.
The role of Tregs in skeletal muscle regeneration is complex. One key function is controlling the innate immune response, especially the activity of neutrophils and macrophages. Neutrophils quickly respond to muscle injury and are crucial for removing dead tissue (38). However, they also release reactive oxygen species and proteases that can cause additional tissue damage (59). Tregs help regulate neutrophil activity and infiltration. In laboratory experiments, activated Tregs have been shown to boost the anti-inflammatory nature of neutrophils by encouraging the release of IL-10, TGF-β, haem oxygenase-1, and indoleamine 2,3-dioxygenase (9). These substances help resolve inflammation and create an environment that supports healing. The interaction between Tregs and macrophages is equally important. In skeletal muscle, macrophages go through a well-understood change in their behavior during regeneration. At first, they display a pro-inflammatory M1-like phenotype, producing TNF-α, IL-1β, and other cytokines that promote inflammation and activate MuSCs (60). As healing advances, macrophages switch to an anti-inflammatory M2-like phenotype, which secretes IL-10 and TGF-β to help tissue remodeling and reduce inflammation (60, 61). Tregs are crucial in encouraging this change by releasing IL-4, IL-10, and IL-13 (62, 63). This cytokine-driven shift not only dampens excessive inflammation but also supports the growth and development of MuSCs, directly aiding muscle repair.
Studies have also shown that Tregs help maintain the homeostasis of macrophage subsets in injured muscle. For example, Treg depletion causes a decrease in MHCII+ macrophages and an overexpansion of MHCII+ macrophages, which are linked to increased antigen presentation and interferon-γ (IFN-γ) responses (64). This imbalance boosts inflammation and disrupts the regenerative process. Conversely, Tregs help stabilize macrophage populations and reduce excessive IFN-γ signaling, thus creating a favorable environment for muscle regeneration (64). In addition to their role in modulating the innate immune response, Tregs also influence adaptive immunity during skeletal muscle repair. After injury, effector T cells, including Th1, Th2, Th17, and CD8+ T cells, infiltrate the damaged muscle and contribute to local immune responses (62). Tregs suppress excessive effector T cell activation through various mechanisms. One important pathway involves the downregulation of costimulatory molecules CD80 and CD86 on dendritic cells via contact-dependent interactions involving CTLA-4 and LAG-3 (62). This process induces indoleamine 2,3-dioxygenase in dendritic cells, which limits T cell proliferation and inflammatory cytokine production, ultimately supporting tissue repair (62).
Tregs also exert direct suppressive effects on effector T cells. They can downregulate IFN-γ production from Th1 and CD8+ T cells, which is crucial because IFN-γ drives macrophage activation toward a pro-inflammatory state by inducing CIITA and promoting MHCII expression (38, 43, 65–67). By limiting IFN-γ levels, Tregs help maintain a favorable balance of immune activation that encourages regeneration rather than chronic inflammation (64). This immunoregulatory loop involving Tregs, macrophages, and effector T cells is central to coordinating muscle repair. One of the most compelling mechanisms by which Tregs contribute to skeletal muscle regeneration is through the secretion of AREG, an EGFR ligand that acts directly on muscle satellite cells. AREG binds to the EGFR expressed on MuSCs, activating downstream signaling pathways that support satellite cell proliferation and survival (68). Burzyn et al. (2013) demonstrated that myogenic precursors exposed to AREG show enhanced regenerative potential (11, 69). This direct crosstalk between Tregs and muscle progenitor cells highlights a non-immune mechanism by which Tregs influence tissue regeneration (Figure 1). The importance of AREG has been supported in both injury and disease models. For example, mice subjected to cryoinjury show increased Treg numbers and elevated expression of AREG, IL-10, and TGF-β at the injury site, supporting the idea that Tregs promote tissue regeneration through a combination of immunomodulation and direct action on satellite cells (70). Similarly, in models of influenza-induced lung damage, Treg-specific deletion of AREG impairs tissue repair despite a normal antiviral immune response, indicating that the role of AREG is specifically regenerative (70). AREG treatment also improves muscle function and reduces Th1-driven inflammation, partly through effects on the transcription factor T-bet in Tregs and the promotion of CD206hi Ly6Clow macrophages, further supporting its therapeutic potential (71).
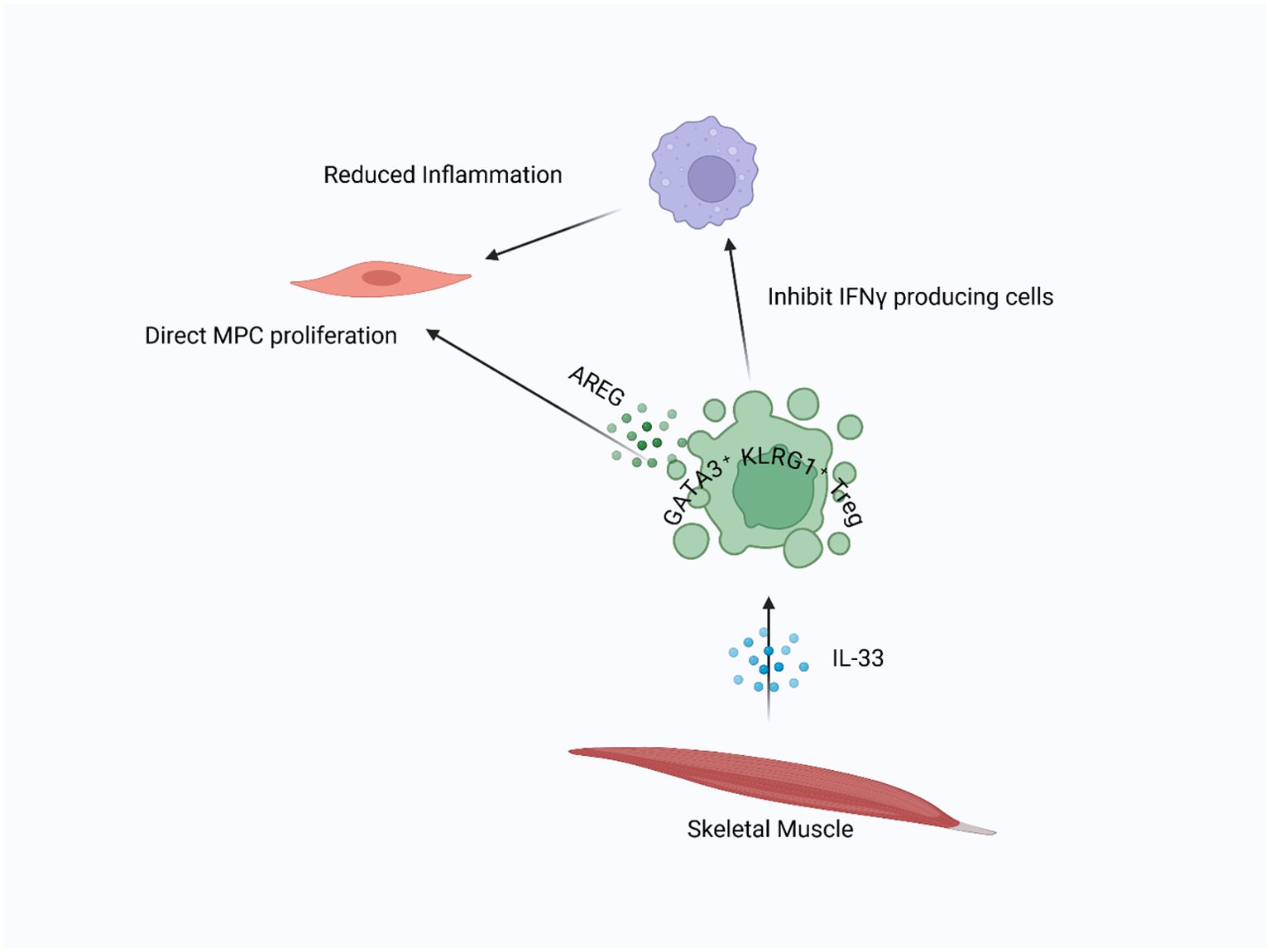
Figure 1. Skeletal muscle resident Tregs in injury repair: In injured skeletal muscle, IL-33 attracts Tregs to the damaged area. Once present, Tregs suppress inflammation driven by M1 macrophages and facilitate the transition to tissue repair. Additionally, Tregs directly stimulate satellite cell proliferation and differentiation by releasing AREG, supporting muscle regeneration.
Despite its regenerative benefits, AREG does not directly increase Treg proliferation or survival. Instead, it affects the post-translational regulation of Foxp3, leading to its degradation via EGFR–GSK-3β signaling pathways (72, 73). This feedback mechanism may serve as a regulatory checkpoint to prevent prolonged or excessive Treg activation. Additionally, AREG upregulates key myogenic markers such as Pax7, MyoD, and myogenin, which promote MuSC activation and differentiation during regeneration (71). Various external signals regulate Treg accumulation and activation in injured muscle. The IL-33–ST2 axis has been identified as a crucial regulator of Treg expansion at the injury site. IL-33, an alarmin released by stressed or damaged cells, enhances ST2 expression on Tregs, promoting their activation and retention (57, 74, 75). Mice lacking ST2 exhibit delayed Treg recruitment and impaired muscle regeneration, confirming the importance of this axis in tissue repair (57).
Furthermore, IL-33 may interact with neuro-immune pathways, as IL-33+ muscle mesenchymal stem cells have been shown to communicate with nerves and stromal elements, influencing Treg accumulation through CGRP signaling (76, 77). Importantly, IL-33 is not limited to Tregs; it can also affect other IL1RL1+ cells, such as type 2 innate lymphoid cells (ILC2s) (78), eosinophils, and alternatively activated macrophages (79), all of which assist in muscle regeneration. While Tregs are the primary responders to IL-33 during tissue repair, it remains crucial to study its diverse effects carefully for therapeutic purposes. Other regulatory molecules, such as PD-1 and sex hormones, also regulate Treg function in muscle. PD-1 is vital for the formation of peripherally induced Tregs, and its absence reduces Treg accumulation and delays repair (80, 81). Estrogen, on the other hand, promotes Treg recruitment and suppresses Th1-mediated inflammation, further supporting the reparative environment (82, 83). Whether reparative Tregs in skeletal muscle are thymus-derived or peripherally induced still needs investigation. However, studies using neuropilin-1 (Nrp1) expression as a surrogate marker suggest that most tissue-resident Tregs, including those in skeletal muscle, are thymus-derived (84). This distinction has therapeutic implications because thymus-derived Tregs may provide more stable lineage characteristics, while peripherally induced Tregs can potentially be generated in vitro from conventional CD4+ T cells for adoptive cell therapy.
Role of Tregs in CNS repair and regeneration
Tregs are extensively studied for their vital role in maintaining immune balance and controlling inflammatory responses. Their role in tissue repair and regeneration has attracted significant interest, especially in the context of central nervous system (CNS) injuries. The CNS, known for its limited ability to regenerate, poses a unique challenge for repair processes. However, recent research highlights the potential of Tregs in supporting CNS repair and regeneration. In models of ischemic brain injury, Tregs have been found to infiltrate the CNS during the sub-acute to chronic phases, where they gather around the lesion site. Their presence is linked to better neurological outcomes, indicating a reparative role (85). One way Tregs may assist CNS repair is by modulating the inflammatory environment. Tregs release anti-inflammatory cytokines like IL-10 and TGF-β, which can inhibit the activity of pro-inflammatory cells such as microglia and astrocytes (86, 87). This inhibition is essential for minimizing secondary damage and creating a supportive environment for neural repair.
The specific mechanisms by which Tregs promote CNS repair are still being clarified, but several key processes have been identified. One important factor is the secretion of AREG, a molecule known for its tissue-protective and regenerative properties (35, 85). AREG produced by Tregs has been shown to reduce the activation of astrocytes, a type of glial cell that, when overly activated, can inhibit neural repair by forming a glial scar (85). By suppressing astrocyte activation, Tregs help prevent the formation of inhibitory scar tissue, thus supporting neuronal survival and axonal regrowth. In addition to AREG, Tregs in the CNS also express unique molecules that may contribute to their reparative functions. For instance, brain-infiltrating Tregs have been found to express high levels of serotonin receptor 7 (Htr7) (35), which enhances cAMP levels (88). cAMP is known to improve the proliferation of Tregs and boost their functional capabilities (89). Activation of Htr7 by serotonin or selective serotonin reuptake inhibitors (SSRIs) has been shown to increase Treg numbers in the brain, further supporting their role in CNS repair (35). This finding opens the door to potential therapeutic strategies that involve modulating Treg activity through pharmacological agents.
Tregs also interact with other CNS cells to promote repair. For instance, they influence the behavior of oligodendrocyte precursor cells (OPCs), which are responsible for remyelination. In experimental autoimmune encephalomyelitis models, transferring Tregs into Treg-deficient mice restores impaired remyelination (90), partly by secreting cell communication network factor 3 (CCN3), a protein that encourages OPC differentiation and myelination (90). This interaction between Tregs and OPCs highlights the diverse role of Tregs in CNS repair, extending beyond their traditional immune-regulatory functions. Another key interaction is between Tregs and resident CNS immune cells (macrophages, microglia, and γδT cells). Tregs can influence these macrophages (microglia), shifting them from a pro-inflammatory M1 phenotype to a more reparative M2 phenotype (35). This shift is crucial for clearing debris and supporting tissue regeneration after injury. Additionally, Tregs may also regulate the infiltration and activity of other immune cells, such as γδT cells and microglia, which participate in the inflammatory response after CNS injury (35). By modulating the immune response, aiding remyelination, and interacting with essential CNS cells, Tregs help create a supportive environment for tissue repair (Figure 2).
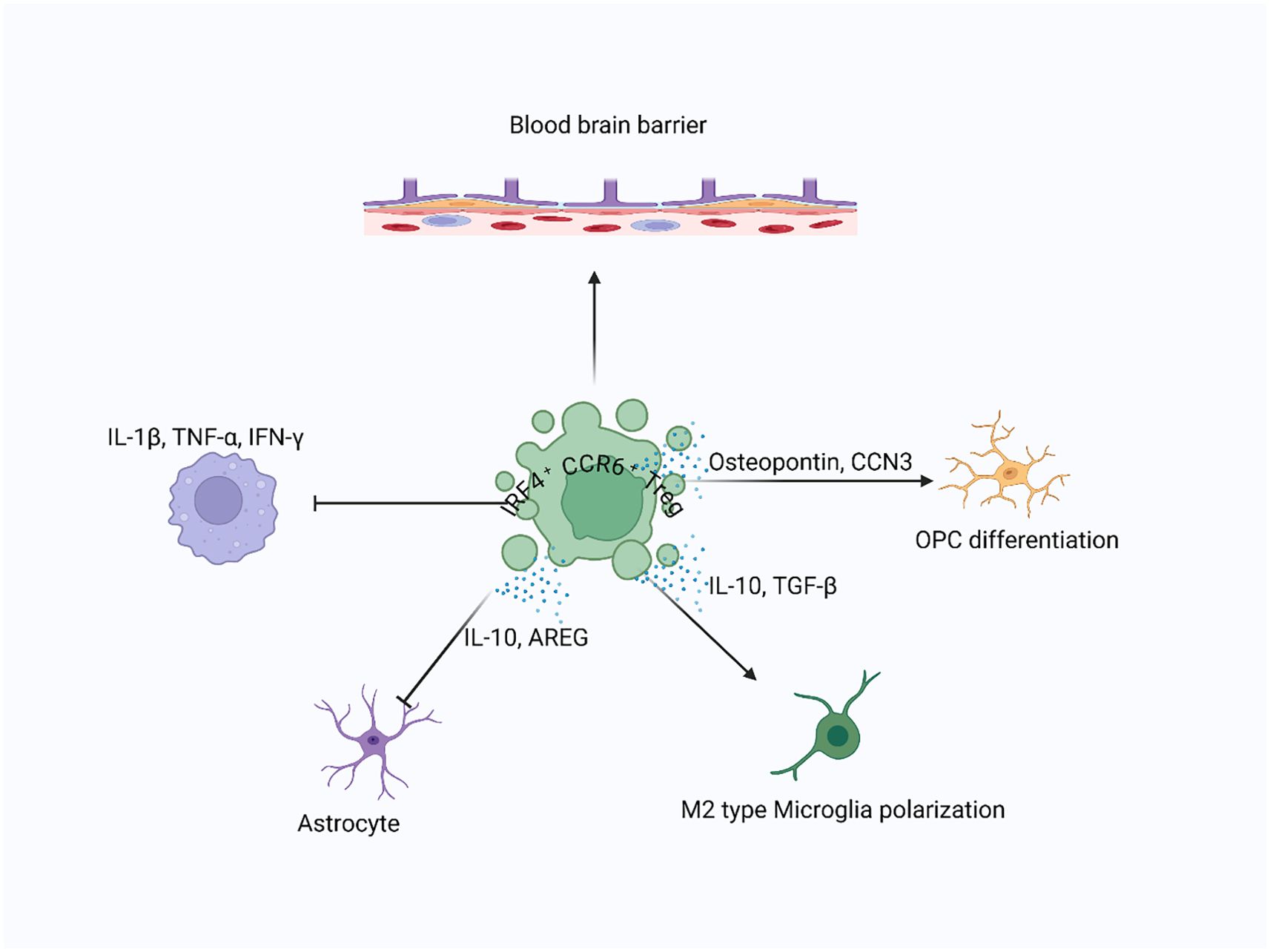
Figure 2. Treg-mediated recovery in CNS injury: Following CNS injury, Tregs infiltrate the brain parenchyma and interact with resident cells, including neurons, astrocytes, microglia, and oligodendrocyte progenitor cells (OPCs), to promote tissue recovery. Tregs suppress proinflammatory cytokines and stimulate the release of neuroprotective factors such as AREG, IL-10, and TGF-β, supporting remyelination, limiting glial scar formation, and enhancing debris clearance.
Tregs in cardiomyocyte repair and regeneration
In lower vertebrates like zebrafish, Tregs are known to promote heart regeneration (91). However, in mammals, adult heart regeneration is limited due to the minimal proliferation of cardiomyocytes and is insufficient to repair large necrotic areas (92). Unlike the neonatal heart, which can regenerate during the first week of life—though the extent depends on injury size—this ability is absent in adults (93, 94). Despite this limitation, Tregs have been shown to contribute to heart repair in adults and support neonatal heart regeneration after myocardial injuries through various mechanisms (95, 96). For example, when Tregs are ablated using diphtheria toxin in FOXP3DTR mice or depleted with an anti-CD25 antibody, adult mice experience significant heart deterioration after myocardial infarction (MI), including increased dilation of the left ventricles and impaired cardiac function (95, 96).
Furthermore, the absence of Tregs raises the risk of severe complications like apical aneurysm and cardiac rupture in adult mice post-MI (97). Similarly, depleting Tregs in neonatal mice results in increased fibrosis and reduced cardiomyocyte proliferation after injury, while adoptive transfer of Tregs promotes regeneration of neonatal heart cells by encouraging their proliferation (96). In adults, adoptive transfer of Tregs has been shown to improve cardiac function and reduce adverse remodeling after injury (96, 98). Additionally, activating Tregs through the administration of a super-agonistic anti-CD28 antibody (95). Collectively, these findings emphasize the essential role of Tregs in protecting the heart by minimizing adverse remodeling in adults and promoting regeneration in neonates.
Mechanistically, Tregs support heart tissue repair and improve cardiac function by regulating macrophage activity. In vitro studies have shown that Tregs encourage macrophages to adopt an M2-like phenotype (95), which is typically linked to tissue repair. However, this relationship depends on the context. In the neonatal heart, increased M2 macrophages have been seen in non-regenerating tissue after Treg depletion, while adoptive transfer of Tregs lowers M2 macrophages and enhances regeneration (96). Conversely, in the adult heart, Treg depletion reduces M2 markers and raises M1 cytokines after MI, indicating that M2 polarization benefits adult repair (95). These findings emphasize the dual role of M2 macrophages—pro-regenerative in adults but possibly pro-fibrotic in neonates—highlighting the need for a nuanced understanding of their function in cardiac recovery. This points to the importance of reconciling the pro-repair versus pro-fibrotic roles of M2 macrophages, especially when evaluating regenerative therapies across age groups.
In addition to modulating macrophages, Tregs can also suppress the pro-inflammatory responses of other immune cells, such as CD4+ and CD8+ T cells. Intriguingly, CD4+ T cells have a developmentally distinct role in heart repair and regeneration, where they inhibit heart regeneration in juvenile mice but support repair in adults (99). Unlike CD4+ T cells, CD8+ T cells are mostly unresponsive to heart injury in juvenile mice (99). Th1 and Th17 cells, known for producing pro-inflammatory cytokines like TNF-α, IFN-γ, and IL-17A, can directly inhibit cardiomyocyte proliferation and induce apoptosis in neonatal cardiomyocytes in vitro. Tregs likely contribute to heart regeneration by inhibiting these pro-inflammatory activities after myocardial injury.
Tregs also help reduce cardiomyocyte apoptosis through a mechanism involving CD39. In vivo studies have shown that transferring CD39-deficient Tregs does not decrease infarct size after MI as effectively as transferring normal Tregs (100). CD39 is known to catalyze the production of adenosine, which replicates the protective effects of Tregs on cardiomyocytes (101). The adenosine produced via CD39/CD73 signaling activates the adenosine A2A receptor, promoting mitochondrial stabilization, reducing oxidative stress, and preserving cellular ATP levels—effects critical for cardiomyocyte survival and function during ischemic stress (102). Furthermore, Tregs help prevent apoptosis of heart cells via the CD39/adenosine/reperfusion injury salvage kinase (RISK) pathway. The RISK pathway includes kinases such as Akt and ERK1/2, which collectively promote cardiomyocyte survival by preventing mitochondrial permeability transition pore opening and activating anti-apoptotic cascades (103). It is also important to distinguish between the reparative and anti-fibrotic functions of Tregs, as these represent distinct processes with separate molecular regulators. Reparative functions typically involve paracrine factors that support cardiomyocyte proliferation and survival (104). At the same time, anti-fibrotic effects stem from Treg-mediated suppression of fibroblast activation and ECM deposition through IL-10, AREG, and adenosine signaling (20, 105). Tregs play a crucial role in the regeneration of heart cells during the neonatal period by enhancing their proliferation. The limited ability of adult cardiomyocytes to regenerate can be linked to factors such as their binucleated state, which leads to cell cycle arrest, and the unique immune response observed in adults after injury. For instance, neonates exhibit a less dominant Th1 and Th17 response, instead favoring Th2-type immunity. The differentiation of Tregs from naive CD4+ T cells appears to be a natural default process in this context (106). Consequently, the impact of Treg-driven cardiomyocyte regeneration is more significant in neonates. The paracrine effects of Tregs seem to be a key mechanism, as the culture medium of Tregs alone can stimulate neonatal cardiomyocyte proliferation (96). Further analysis through single-cell RNA sequencing of Tregs after myocardial injury identified factors like CST7, MATN2, CCL24, GAS6, TNFSF11, FGL2, IGF2, AREG, IL33, as potential candidates that promote cardiomyocyte proliferation (96, 97). One important factor, AREG, secreted by Tregs, plays a key role in cardiac repair following myocardial infarction. Although it remains unclear whether AREG acts directly on cardiac progenitor cells, endothelial cells, or through intermediaries, its mitogenic and tissue-protective functions suggest broad regenerative roles. Treg-derived AREG has been shown to enhance cardiomyocyte proliferation and modulate macrophage polarization (96), and may also support endothelial cell function and neovascularization (104), highlighting its multifaceted contributions to heart regeneration. These findings expand the understanding of Tregs’ reparative abilities through their interactions with local cells like cardiomyocytes, enhancing tissue repair and regeneration. Overall, Tregs are crucial for both adult heart repair and neonatal heart regeneration, working through immune modulation, fibrosis reduction, prevention of apoptosis, and stimulation of cardiomyocyte proliferation (Figure 3).
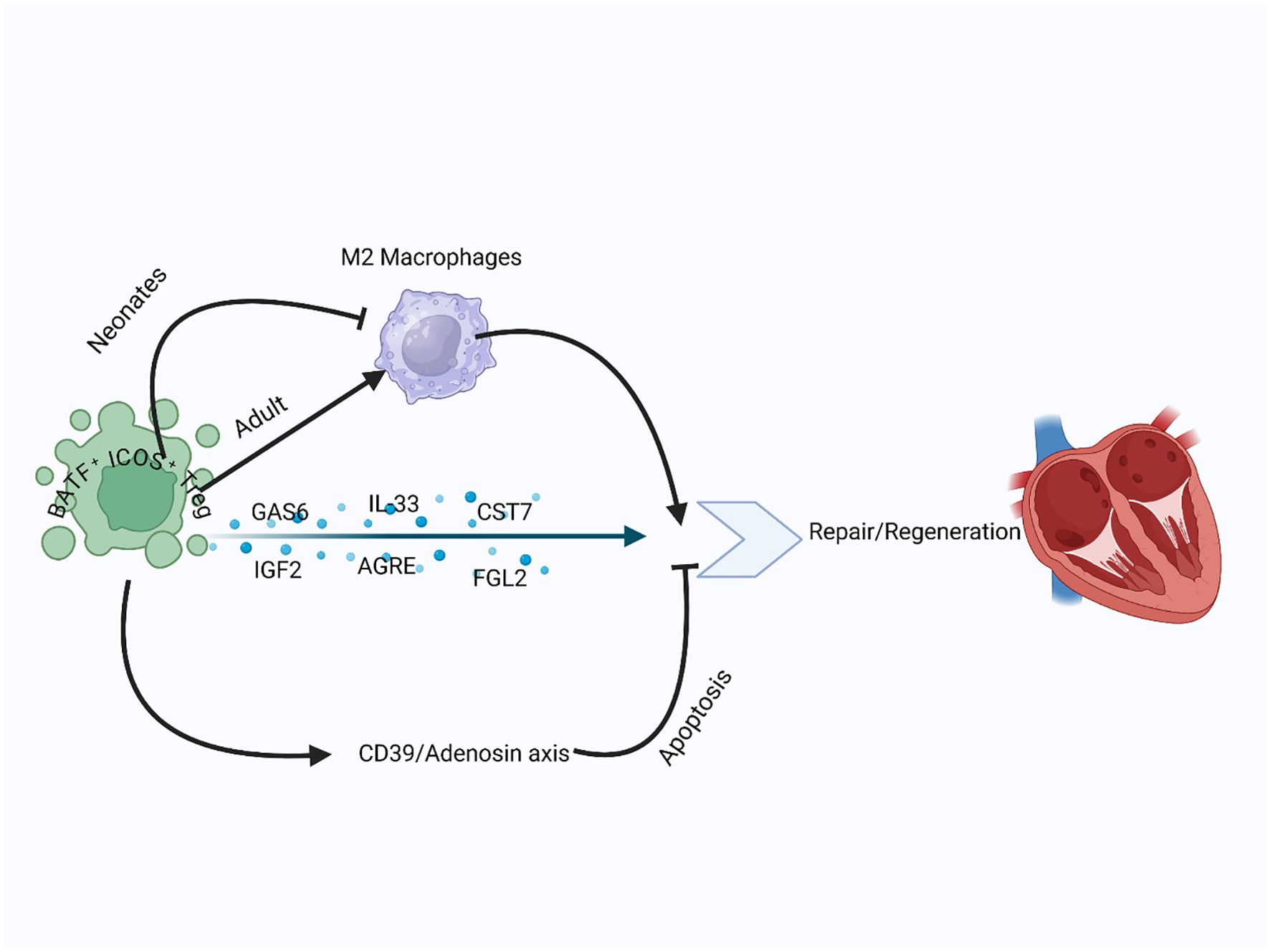
Figure 3. Treg role in cardiac repair and remodeling: In both neonatal and adult hearts, Tregs reduced adverse remodeling and enhanced cardiomyocyte proliferation. Tregs modulate M2 macrophage polarization, promoting repair in adults and limiting fibrosis in neonates. They reduce inflammation, inhibit apoptosis through the CD39–adenosine axis, and secrete paracrine factors like IGF2, CST7, and AREG to stimulate myocardial regeneration.
Functions and specialization of skin-resident Tregs
Skin-resident Tregs can differentiate into subsets similar to helper T cells (TH1, TH2, TH17) through specific transcription factors. Specifically, “Type 2” or “repair type” Tregs, marked by IRF4, GATA-3, and BATF, are essential for managing type 2 immune responses and supporting tissue repair (21, 107, 108). GATA-3 acts as a master regulator of type 2 immunity, while BATF (basic leucine zipper transcription factor ATF-like) further refines the reparative phenotype by promoting IL-10 and AREG production (109, 110). GATA-3+ Tregs not only suppress type 2 inflammation but also perform reparative functions associated with TH2 and ILC2 cells. These cells detect tissue damage via receptors for alarmins such as IL-33, IL-18, TSLP, and IL-25, which are released by stressed cells (111). In various tissues, including the skin (36), wounded muscle (11), and visceral fat (112), GATA-3+ Tregs express the IL-33 receptor ST2, enabling them to engage in repair mechanisms triggered by IL-33 from damaged cells, a process independent of TCR stimulation. ST2+ Tregs are more effective at promoting wound healing by regulating inflammation and enhancing epithelial regeneration (113). Similarly, TSLP, released by keratinocytes during inflammation, helps activate skin Tregs (114). IL-18, another key cytokine, might also influence skin Tregs’ reparative function, though the presence of its receptor on skin Tregs needs further confirmation (115, 116). Thus, skin-resident type 2 Tregs are specialized for utilizing type 2 immune mechanisms in tissue repair (Figure 4).
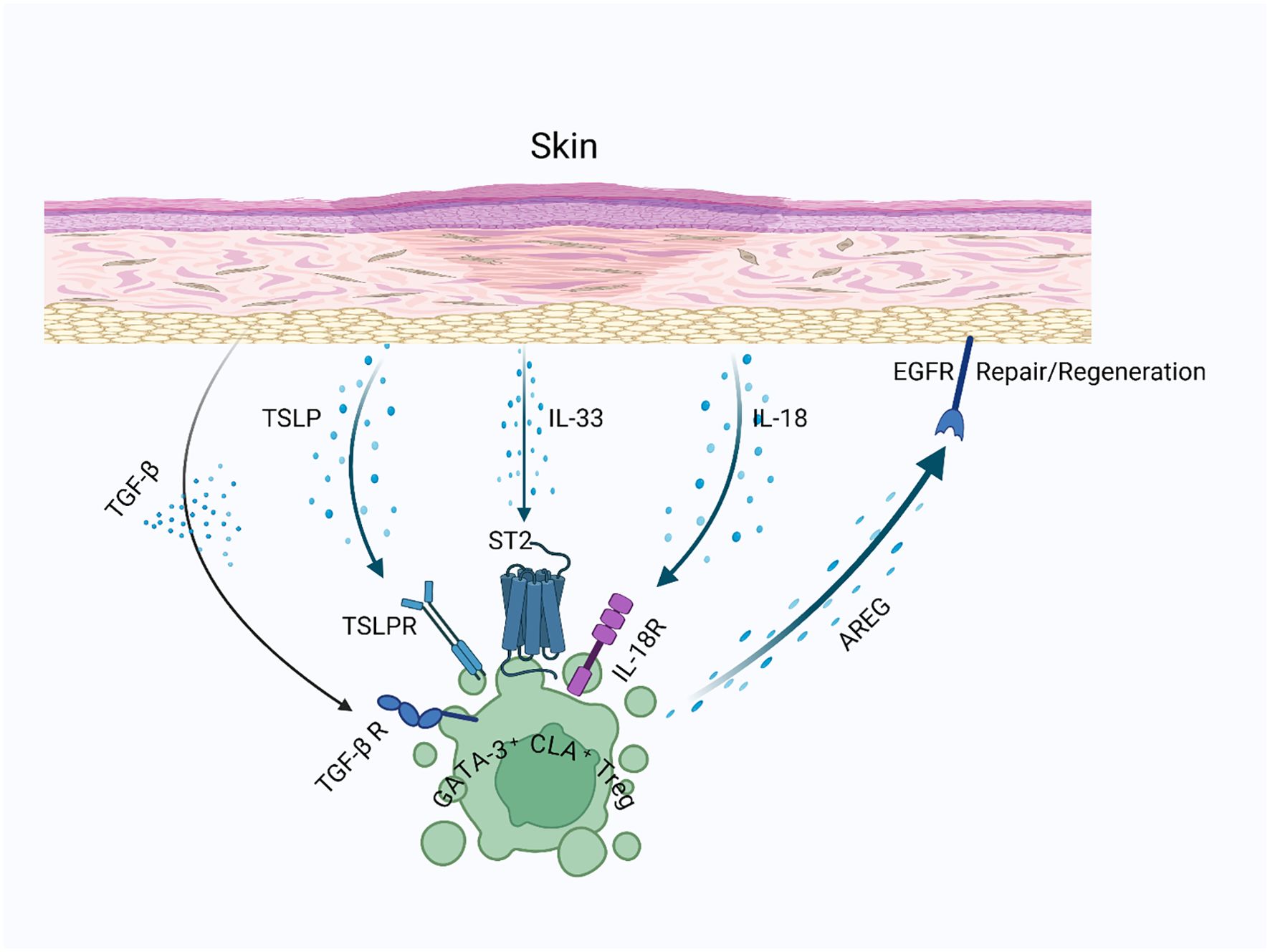
Figure 4. Skin-resident Tregs in wound healing: Skin-resident GATA-3-expressing Tregs express receptors for alarmins such as TSLP, IL-33, and potentially IL-18, which are released upon tissue injury. These signals enable Tregs to detect local damage and induce AREG production, promoting keratinocyte proliferation and facilitating the regeneration of stromal components in injured skin.
Skin injury triggers a strong inflammatory response that attracts immune cells, including Tregs, from the bloodstream to the damaged area. This response occurs not only in the skin but also in tissues that generally lack resident Tregs, such as the brain and muscle, emphasizing their role in healing (11, 85). Tregs gather in the skin after both minor and severe wounds, reaching their peak around one week post-injury (117, 118). The precise mechanisms behind Treg accumulation—whether through blood recruitment or local proliferation—are not entirely understood. Inflammation typically involves both processes and the movement of cells into the lymphatic system (119). Skin-resident Tregs respond promptly to injury, while circulating Tregs need time to activate and migrate, potentially supporting the repair process initiated by resident Tregs (21).
AREG, which interacts with epidermal growth factor receptor (EGFR), is essential for tissue repair and is produced by various type 2 immune cells, including GATA-3+ Tregs (120). The production of AREG in Tregs is stimulated by interleukins IL-18 and IL-33 (20). When bound to EGFR, AREG promotes the proliferation and differentiation of target cells, aiding in the repair and regeneration of damaged tissue. Interestingly, autocrine EGFR signaling in Tregs has been primarily described in skin wound healing (117), but emerging evidence suggests similar mechanisms operate in lung and muscle repair (11, 20). For example, in muscle injuries, AREG from Tregs supports the regeneration of myofibers from satellite cell progenitors (11). While several cell types, such as basophils, ILC2s, TH2 cells, and macrophages, can produce AREG, its expression by Tregs is particularly significant (20). Mice lacking AREG specifically in Tregs show increased mortality following influenza infection and extensive lung damage, highlighting the critical role of Tregs in AREG-mediated tissue repair (20).
Although research on Treg-derived AREG in skin injury is limited, evidence indicates its importance. Skin-resident Tregs express AREG (36), which promotes keratinocyte proliferation (120), similar to its effects on other epithelial tissues. AREG, along with keratinocyte growth factor (KGF) and fibroblast growth factor 2 (FGF2), synergistically enhances re-epithelialization by stimulating keratinocyte migration and proliferation (121). Additionally, these growth factors contribute to stromal remodeling by activating fibroblasts and promoting extracellular matrix reorganization (122). AREG also helps maintain vascular integrity by activating TGF-β signaling in pericytes (123), which may support stromal repair following skin injury. In wounded skin, Tregs express the EGFR receptor for AREG, and removing EGFR specifically from Tregs leads to fewer Tregs and slower wound healing, suggesting that AREG may have an autocrine effect in boosting Treg numbers during skin injury (117). Besides AREG, Tregs secrete other growth factors that aid repair by acting directly on non-hematopoietic cells. For example, Tregs release keratinocyte growth factor (KGF) to support alveolar regeneration after lung damage (124) and fibroblast growth factor 2 (FGF2) to enhance intestinal epithelial growth during colitis (125). In the skin, KGF and FGF2 likely contribute to epidermal repair by stimulating keratinocyte proliferation and migration. In zebrafish, Tregs help regenerate various organs after injury by secreting growth factors such as neuregulin 1 (NRG1) in the heart, nerve growth factor (NGF) in the spinal cord, and insulin-like growth factor 1 (IGF-1) in the retina (91). Both KGF and IGF-1 are also produced by dendritic epidermal T cells (DETCs) to support epidermal regeneration after skin injury (126). This suggests that skin-resident Tregs may employ similar mechanisms to facilitate repair processes following skin injury. The roles of IL-10 and TGF-β in skin repair seem to be both sequential and context-dependent. Early after injury, IL-10 predominates to suppress excessive inflammation, while TGF-β becomes more prominent in later stages to regulate fibrosis and promote matrix remodeling (127, 128). Tregs dynamically adjust their cytokine production in response to the wound microenvironment, ensuring a balanced healing response (117).
Tregs in lung repair and regeneration
The lung’s ability to maintain homeostasis despite constant exposure to airborne pathogens and particulates reflects its complex immune-regulatory networks (129, 130). In disease states like infection-induced acute respiratory distress syndrome (ARDS), this balance is disrupted, requiring not only pathogen clearance and inflammation control but also regeneration of damaged tissue (131). This regenerative process involves carefully coordinated interactions among epithelial, endothelial, and mesenchymal compartments (132). Tregs, beyond their typical anti-inflammatory functions, gather in injured alveolar spaces in both mice and humans and actively aid in lung repair (133, 134). Among epithelial cells, alveolar type II (AT2) cells are crucial for regeneration because of their progenitor role—they proliferate, secrete surfactant, and differentiate into alveolar type I (AT1) cells to restore the gas exchange surface (130). Tregs support AT2 proliferation and survival through various mechanisms, including secreting keratinocyte growth factor (KGF) and engaging in direct cell interactions. Specifically, Mock et al. (2014) showed that Tregs expressing CD103 (αE integrin) physically interact with AT2 cells via E-cadherin, a key adhesion molecule on epithelial cells (135). This CD103/E-cadherin axis helps retain Tregs within the alveolar niche and enables localized communication with AT2 cells. Co-culture experiments further support this interaction. In vitro studies using isolated Tregs and primary AT2 cells show that CD103+ Tregs promote AT2 proliferation and maintain their stem-like transcriptional profile, partly through E-cadherin-dependent adhesion (135, 136). When CD103 or E-cadherin is disrupted in these systems, Tregs’ ability to promote epithelial regeneration decreases, indicating that this interaction is not just structural but also essential for Treg-driven epithelial repair. Importantly, this adhesion-dependent mechanism likely allows close-range delivery of trophic signals, such as KGF and AREG, enhancing repair pathways in a context-specific manner (124, 135).
AREG, an EGFR ligand, has become an important mediator of Treg-driven tissue protection and repair in the lungs and other tissues (11, 20, 137, 138). In a mouse model of influenza-induced lung injury, IL-18 and IL-33 signaling promote Treg-derived AREG production, which helps preserve alveolar structure and gas exchange (136). Interestingly, the IL-18/IL-33-induced AREG expression seen in lung Tregs mirrors similar mechanisms in skin and gut tissue-resident Tregs. In the skin, AREG production by GATA3+ Tregs promotes keratinocyte proliferation, while in the gut, AREG supports maintaining the epithelial barrier during colitis (20). However, the lung microenvironment, rich with alveolar macrophages and surfactant-secreting epithelial cells, may uniquely boost IL-18/IL-33 signaling, making lung-resident Tregs especially powerful producers of AREG after infection or injury (68). This tissue-specific response could explain the strong repair response observed in the lungs. NOTCH4 is a key transcriptional regulator of IL-18–induced AREG expression in Tregs, and its levels are inversely related to serum AREG levels and COVID-19 severity, linking this pathway to clinical outcomes (138). Furthermore, a mesenchymal cell population expressing Col14a1 has been identified as a target of Treg-derived AREG signals, supporting alveolar epithelial organoid growth and aiding tissue repair in vivo (139). Deletion of EGFR in these stromal cells worsens hypoxia after influenza infection, indicating that Treg-derived AREG acts on mesenchymal targets as well.
However, mesenchymal cell populations are heterogeneous. While some subsets support epithelial regeneration, others (e.g., AXIN2+ myogenic precursors) can worsen injury by promoting fibrosis through myofibroblast activation (140). Tregs help control this maladaptive response. During the fibroproliferative phase of acute lung injury, Tregs decrease the recruitment of collagen-producing bone marrow–derived cells by modulating CXCL12/CXCR4 signaling (141). Pharmacologic inhibition of CXCR4 with AMD3100 reduces fibrosis independently of CXCL12, indicating CXCR4 as a potential therapeutic target. While epithelial repair has been a primary focus, endothelial regeneration is equally important for restoring lung homeostasis. Coordinated signaling between epithelium and endothelium is crucial for regenerating the alveolar-capillary interface (142). CAR4+ endothelial cells, which depend on VEGFA signaling from epithelial cells, play a vital role in this process during both development and post-viral injury (143, 144). These cells are enriched in tip-cell markers (Vegfr2, Nrp1, Apln) and are located near AT1 cells, suggesting their involvement in vascular regeneration.
Beyond acute injury, Tregs also have roles in chronic lung diseases. In pulmonary fibrosis models, Tregs regulate fibroblast activity and reduce excessive collagen buildup through TGF-β-dependent and independent pathways (145). In chronic obstructive pulmonary disease (COPD), lower Treg numbers and impaired function are linked to ongoing inflammation and alveolar damage (146). These findings indicate that improving or restoring Treg function could be important for preventing or treating chronic lung remodeling and dysfunction. Tregs have multiple roles in lung repair, including secreting trophic factors (KGF, AREG), directly interacting with epithelial cells via CD103/E-cadherin, controlling fibrotic responses through the CXCL12/CXCR4 axis, and supporting endothelial regeneration (Figure 5).
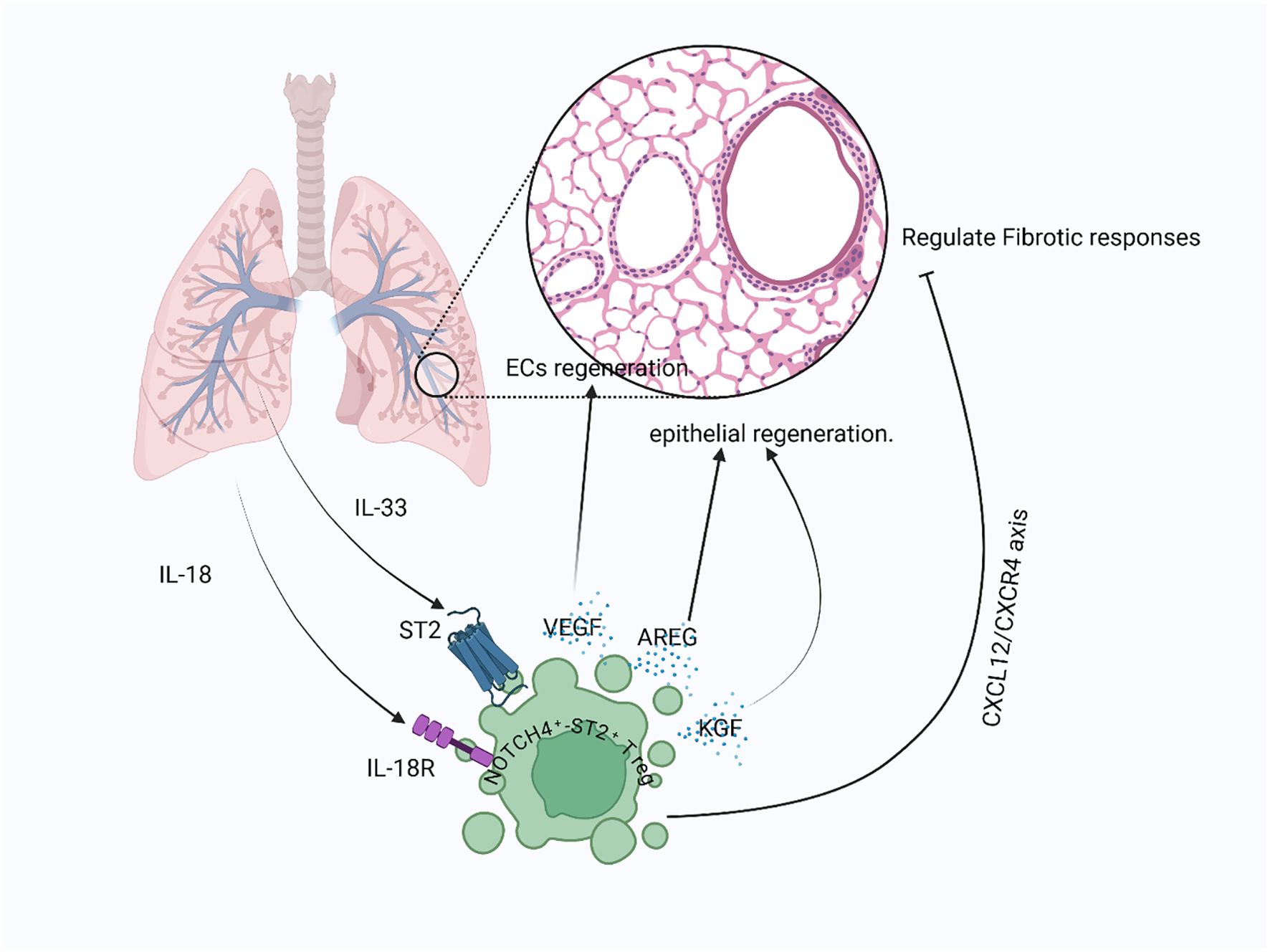
Figure 5. Treg-mediated lung repair post-injury: After lung injury, signals like IL-18 and IL-33 released from damaged epithelial cells and activated immune cells interact with their respective receptors on Tregs, stimulating the production of epithelial-repair-promoting factors such as AREG and KGF. Tregs also contribute to vascular repair by releasing VEGF to support alveolar capillary endothelial cell recovery, while limiting fibrotic responses through the CXCL12/CXCR4 signaling axis.
Tregs in bone repair and regeneration
Tregs have proven to be key players in bone healing and regeneration. Unlike many tissues that heal with scar tissue, bone has a remarkable ability to regenerate without scarring through the combined action of osteoblasts and osteoclasts (9). Osteoblasts, originating from mesenchymal stem cells (MSCs), build new bone, while osteoclasts, derived from bone marrow cells, break down old bone. Tregs help regulate this process by influencing both bone formation and breakdown [131]. Mechanistically, Tregs suppress osteoclastogenesis mainly by inhibiting receptor activator of nuclear factor kappa-B ligand (RANKL) signaling, which is crucial for osteoclast differentiation and activation. This suppression occurs via secretion of anti-inflammatory cytokines like IL-10, which inhibit osteoclast precursor maturation and function (147, 148). Tregs also produce TGF-β, contributing to osteoblast differentiation and supporting bone formation (149). Although direct interactions of Tregs with osteoblast precursors are less well defined, recent evidence suggests Tregs may promote osteoblast maturation indirectly by modulating the inflammatory environment to favor bone growth (150). MSCs can induce Tregs to develop from naive T-cells and help them multiply, partly through a protein called haeme oxygenase-1 (HO-1) (151). Notably, this MSC-induced Treg differentiation appears to be largely antigen-independent, relying instead on paracrine signals like TGF-β and prostaglandin E2, which create an immunosuppressive environment conducive to Treg expansion (152). Moreover, CD3+ T-cells aid in the differentiation of other blood cells into osteoclasts, and Tregs intervene to inhibit this process, using signaling molecules such as TGF-β and IL-4 (148, 153) (Figure 6). This inhibition is important because higher levels of Tregs in the blood are associated with lower levels of markers indicating bone loss, a trend observed in both healthy individuals and those with rheumatoid arthritis. Animal studies also support the idea that Tregs help prevent bone destruction. For instance, Tregs can protect against bone loss caused by TNF-α and reduce bone damage in ovariectomized mice (154, 155). Using specific antibodies to enhance Treg activity has also shown promise in reducing arthritis and increasing bone density in mice, likely by limiting osteoclast activity (9, 156).
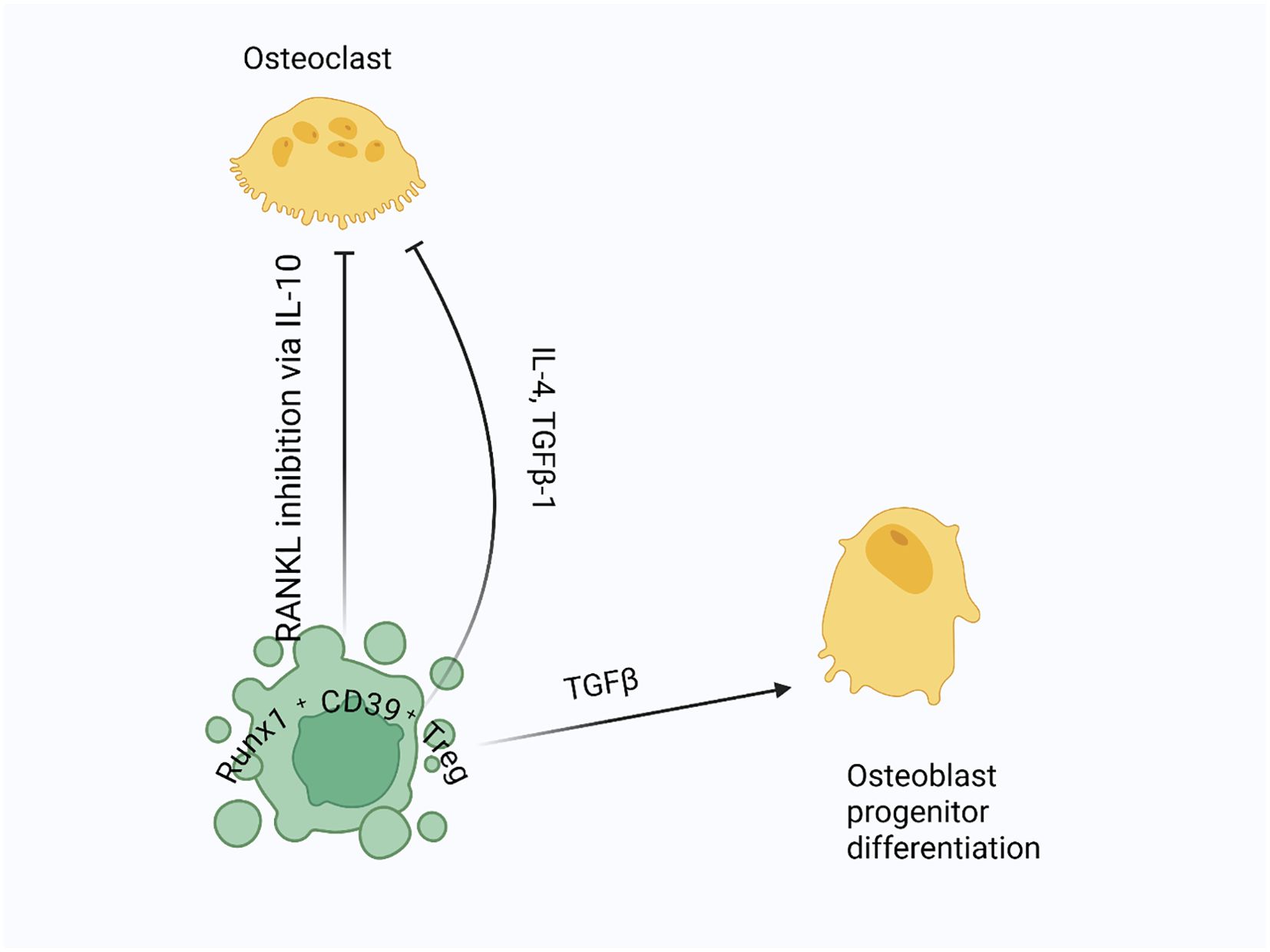
Figure 6. Treg regulation of osteoclastogenesis and bone formation: Tregs suppress osteoclastogenesis by inhibiting RANKL signaling, crucial for osteoclast differentiation and activation. They secrete anti-inflammatory cytokines like IL-10 to block osteoclast precursor maturation, and they release TGF-β and IL-4 to further inhibit osteoclast differentiation. Additionally, Tregs produce TGF-β to support osteoblast differentiation and bone formation.
Tregs may also directly aid in the maturation of bone-forming cells. In a bacterial-induced bone injury model, Tregs were drawn to the injury site by a signaling molecule called CCL22. There, they helped prevent bone loss by reducing inflammation (156). Additionally, Tregs support MSC-based bone repair by suppressing CD4+ T-cells that produce inflammatory cytokines like IFN-γ and TNF-α, which can impede bone formation (157, 158). Therefore, Tregs are vital for bone repair and regeneration. They not only help regulate bone resorption but also promote new bone growth, making them essential for maintaining bone health and facilitating healing.
Role of Tregs in liver diseases
When chronic wounds persist and cause ongoing inflammation, they can trigger a series of events that lead to liver fibrosis and eventually cirrhosis (159). Different liver inflammatory diseases, such as non-alcoholic fatty liver disease (NAFLD), non-alcoholic steatohepatitis (NASH), hepatitis B (HBV), and hepatitis C (HCV), contribute to liver fibrosis (160, 161). Reduced activity of Treg cells (indicated by low FoxP3 mRNA levels) has been observed in the NAFLD mouse model of the liver. This decline negatively affects NAFLD progression by disrupting the balance between Tregs and Th17 cells, resulting in an increased Th17-driven inflammatory response (162). An association has also been noted between an increased number of Foxp3+ lymphocytes in liver lobules and NAFLD-driven hepatic inflammation, further indicating the role of Tregs in hepatic disease (163). Similarly, fewer Tregs was observed in mice with steatohepatitis, but adoptively transferring Tregs into these mice helped reduce liver injury and slow disease progression (164). In autoimmune liver diseases, the number of infiltrating Tregs in the liver surpasses that circulating in the bloodstream (165). In these conditions, persistent inflammation causes hepatocyte apoptosis and necrosis. Consequently, the anti-inflammatory potential of Tregs helps mitigate this damage, providing a protective effect against liver inflammation (166). Studies also suggest that Tregs can contribute to liver fibrosis by influencing Kupffer cells through the TGF-β pathway (167, 168). Treg-derived TGF-β appears to act mainly through an indirect mechanism — modulating Kupffer cell polarization rather than acting directly — thereby shaping the immune microenvironment toward a more fibrotic or anti-fibrotic state (167, 169–171). Recent research proposes a dual role for Tregs in liver fibrosis: initially offering anti-inflammatory effects that protect hepatocytes, but later contributing to fibrotic progression (172, 173). In early NAFLD/NASH, Tregs suppress Th17-driven inflammation and inhibit hepatic stellate cell (HSC) activation (172, 173). However, in advanced fibrosis, they may switch roles (Figure 7). For example, AREG-expressing Tregs have been shown to directly stimulate EGFR signaling in HSCs, promoting fibrosis in both human and mouse models of NASH (171). This underscores a stage- and context-dependent shift from protective to pathogenic Treg activity. Single-cell RNA sequencing from mouse models and spatial transcriptomics from human liver tissue both reveal the presence of Tregs with a profibrotic gene signature, such as Areg and CTLA-4, which closely interact with hepatic stellate cells during fibrosis. This supports the idea that Tregs in fibrotic livers not only suppress inflammation but also actively contribute to fibrosis through cellular interactions and signaling (174, 175).
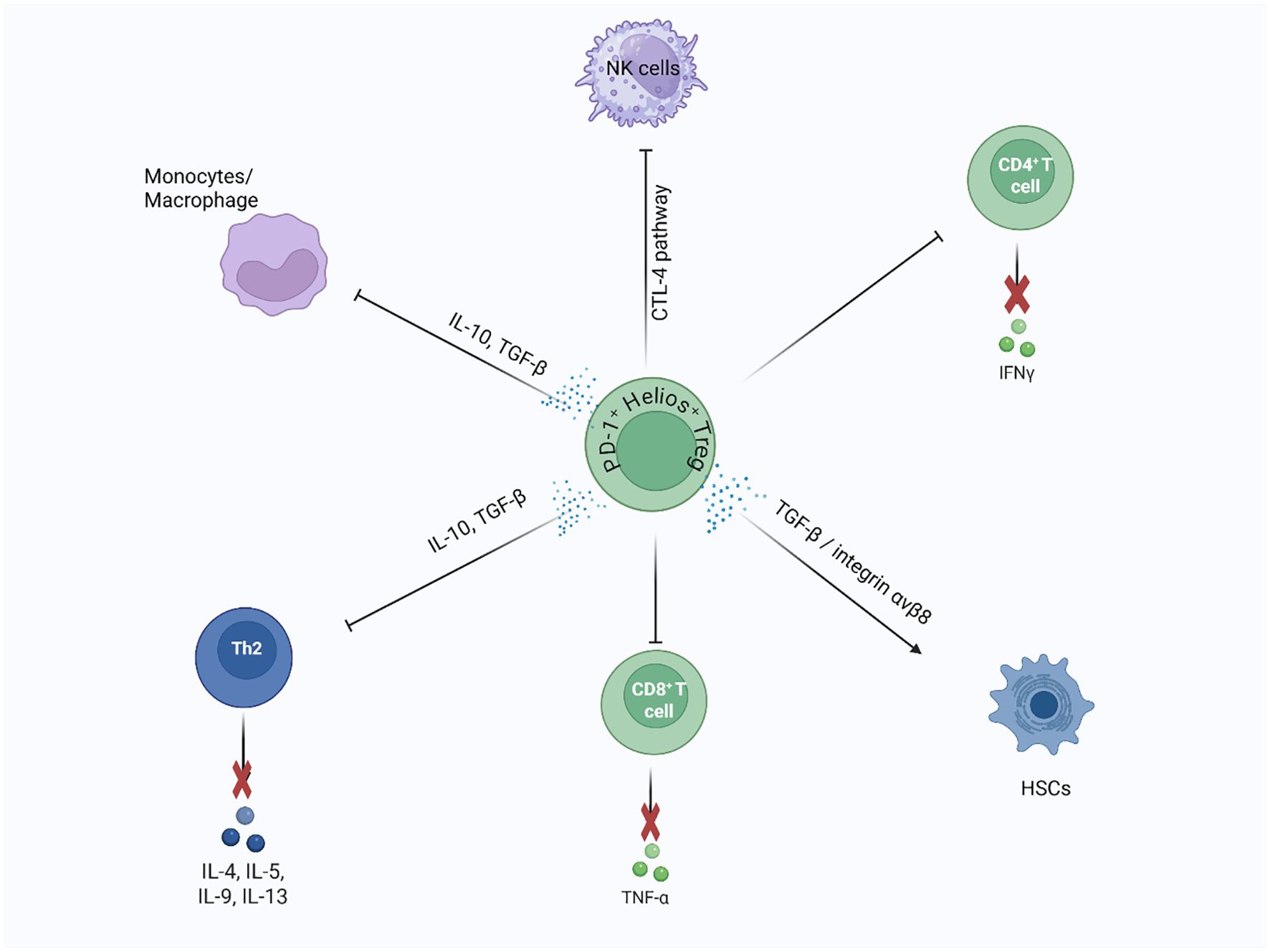
Figure 7. Dual role of liver Tregs in fibrosis regulation and repair: Liver Tregs (Helios+, ST2+) play a dual role in liver fibrosis and regeneration. During repair, hepatic stellate cells (HSCs) promote Treg regeneration via MMP 9/13-dependent TGF-β activation, supporting wound healing. In contrast, liver Tregs suppress pro-fibrotic immune cells, such as Th2 and Ly-6Chigh monocytes/macrophages, through IL-10 and TGF-β secretion. They also inhibit CD8+ T cell proliferation and modulate HSC activity by suppressing NK cells, influencing fibrosis progression.
Tregs play a vital role in wound healing and reducing inflammation. However, some studies indicate that lowering Tregs could help reverse fibrosis. Activated HSCs release IL-2, which increases Tregs in fibrotic tissue. In a healthy liver, Kupffer cells (KCs) secrete matrix metalloproteinases (MMPs) that break down and regulate the liver’s ECM. Tregs produce TGF-β, which can inhibit KCs-mediated MMP release, potentially hindering fibrosis reversal (172). Research using DEREG transgenic mice, which lack Tregs, showed that removing these cells led to increased liver fibrosis, mainly due to an increase in CD8+ and IL-17A+ T cells and higher secretion of inflammatory factors (161). TGF-β is an essential cytokine that influences whether T cells develop into Th17 cells or Tregs. When TGF-β is low along with IL-6, it promotes Th17 cell differentiation. Conversely, high levels of TGF-β cause naïve CD4+ T cells to develop into Tregs. During liver fibrosis, both IL-6 and TGF-β levels increase, stimulating HSCs to produce more ECM proteins. This process heightens Th17 cell numbers and creates an imbalance between Tregs and Th17 cells (160).
Tregs in kidney repair and regeneration
Tregs, known for their immune-modulatory functions, have increasingly been recognized for their key role in renal injury repair and regeneration, especially after ischemia-reperfusion injury (IRI). Studies show that Tregs infiltrate the kidneys during the healing process and are crucial in reducing damage and aiding recovery. In a murine model of ischemic acute kidney injury, Tregs were observed to migrate into the kidneys within 3 to 10 days following the injury. Their presence was linked to less renal tubular damage, increased tubular proliferation, and lower production of pro-inflammatory cytokines (176), all of which contributed to better renal function and lower mortality. Tregs primarily protect by suppressing excessive immune responses that can worsen renal injury. For example, depleting Tregs in animal models before inducing renal IRI resulted in more kidney damage, with higher blood urea nitrogen (BUN) and serum creatinine (Scr) levels, along with increased tubular necrosis scores. Conversely, infusing Tregs after IRI onset significantly enhanced renal repair by decreasing inflammatory cytokine production and supporting the recovery of renal function (177).
In addition to their immunosuppressive functions, Tregs also support tissue regeneration. In models of renal fibrosis, Tregs induced by mesenchymal stem cells (MSCs) pretreated with interferon-gamma (IFN-γ) were found to significantly decrease fibrosis and improve kidney function. This effect was mediated by the enzyme indoleamine 2,3-dioxygenase (IDO), whose expression was increased by IFN-γ (178). IDO played a key role in the anti-fibrotic effects of Tregs, indicating a potential therapeutic approach for preventing renal fibrosis. Further evidence emphasizes the role of Tregs in chronic kidney disease and transplantation. Foxp3+ Tregs have been shown to limit autoimmune kidney disease and aid in transplant tolerance. In kidney transplant models, depleting Tregs disrupted transplant tolerance, while their presence was linked to higher levels of immunosuppressive cytokines like TGF-β and IL-10 (179). This indicates that Tregs are not only vital for repairing acute injury but also crucial for maintaining long-term kidney health. It is important to distinguish the role of Tregs in acute versus chronic kidney conditions. In acute settings such as IRI, Tregs mainly work to suppress inflammation and protect renal tubular epithelial cells, primarily within the peritubular interstitium, thus promoting regeneration (180, 181). In contrast, in chronic conditions like renal fibrosis or transplantation, Tregs display functional plasticity and phenotypic diversity, driven by the inflammatory and alloimmune microenvironment. These cells may show altered FOXP3 stability, demethylation patterns, or cytokine expression—shifting between protective immune regulation, promotion of fibrotic processes, or tolerance induction after transplantation (181–183).
The mechanisms by which Tregs contribute to renal repair involve several pathways. One key process is suppressing innate immune responses, especially those mediated by neutrophils and macrophages (180), which are often elevated after IRI and contribute to ongoing tissue damage. Tregs accomplish this by secreting anti-inflammatory cytokines such as IL-10 and producing adenosine via CD73, which activates the adenosine A2A receptor (A2AR) on immune cells to further reduce inflammation. The CD73/adenosine A2A receptor (A2AR) axis plays a crucial role in controlling immune cell infiltration and supporting tubular epithelial cell survival. During injury, Tregs express CD73, converting extracellular AMP into adenosine. The adenosine then binds to A2AR on neutrophils and macrophages in the peritubular interstitium, decreasing their recruitment and cytokine production, thus creating a microenvironment (184).
Furthermore, activation of A2AR on renal epithelial cells and immune cells enhances tubular epithelial cell resistance to apoptosis and encourages proliferation, aiding kidney repair (185). Animal studies demonstrate the significance of this pathway: in models of ischemic kidney injury, Tregs lacking CD73 or A2AR fail to provide protection, whereas pharmacological activation of A2AR restores their protective function (184). Additionally, recent research emphasizes the role of chemokine receptors like CXCR3 in recruiting and functioning of Tregs during renal injury. CXCR3 expression on Tregs is linked to their migration into the kidney and subsequent improvement of IRI. Notably, CXCR3 is mainly found on activated effector Tregs, which helps them home to inflamed renal tissues. Evidence from human T-cell studies suggests that a small subset of CXCR3+ naïve-like T cells, which have ready effector potential, may upregulate CXCR3 after injury—indicating that even naïve or central-memory Tregs can gain homing ability during inflammation (186, 187). Mice overexpressing CXCR3 showed increased Treg infiltration, reduced inflammation, and better renal function (177, 186), highlighting the importance of chemokine signaling in Treg-mediated protection during renal IRI. Therefore, Tregs are essential for renal injury repair and regeneration by modulating immune responses, reducing inflammation, and promoting tissue regeneration. Their therapeutic potential is evident in both acute and chronic kidney diseases, making them a promising target for future therapies aimed at enhancing kidney repair and preventing fibrosis (Figure 8).
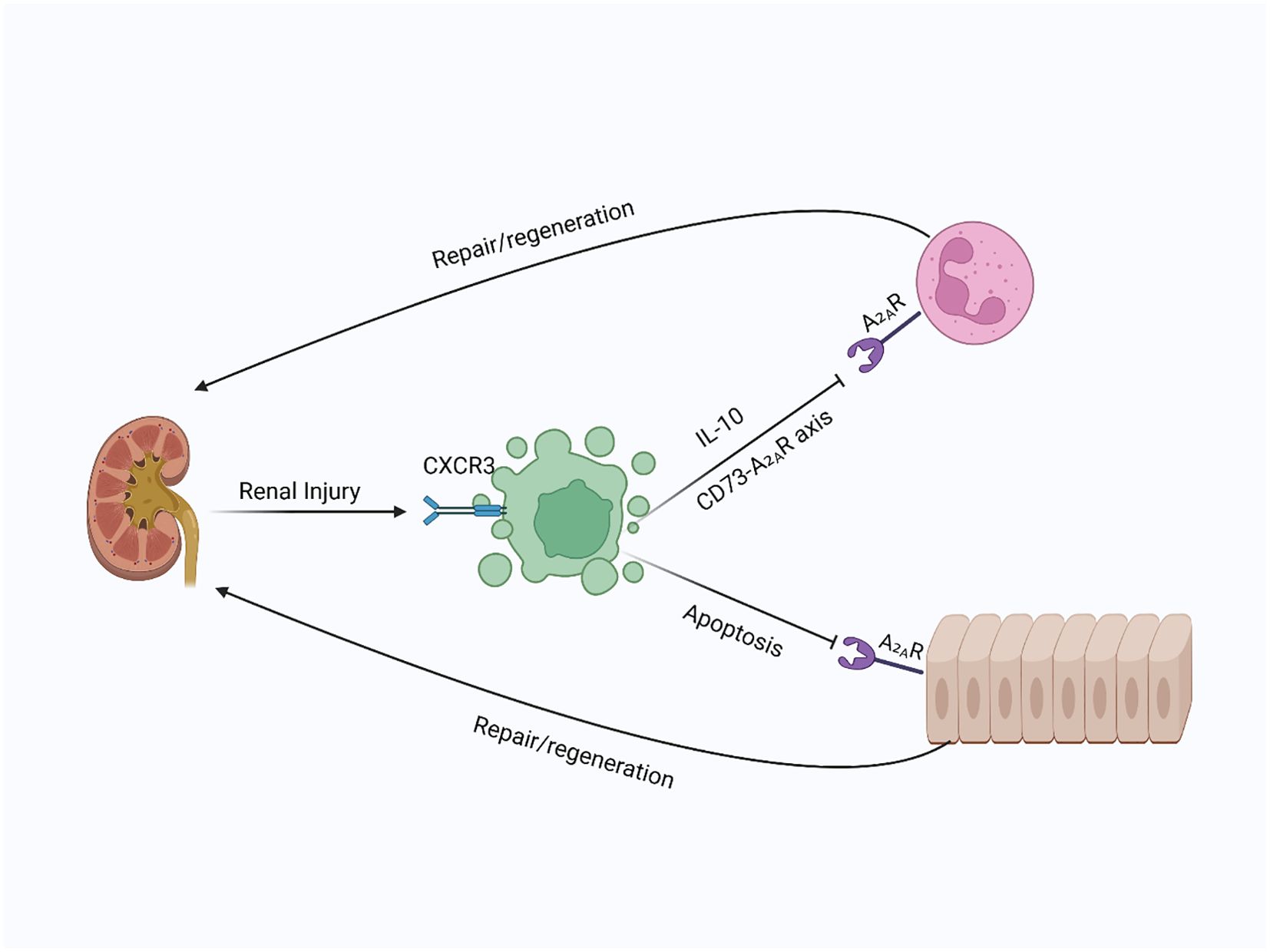
Figure 8. Tregs in kidney injury and repair: In kidney injury, Tregs are recruited to the site of damage, where they suppress immune responses and promote repair. By secreting IL-10 and producing adenosine via CD73, Tregs activate A2A receptors on immune and epithelial cells, inhibiting immune infiltration and apoptosis, leading to enhanced kidney regeneration.
Role of Treg in the gastrointestinal tract
Tregs are increasingly recognized not only for their immunosuppressive abilities but also for their active role in gastrointestinal (GI) tissue repair and regeneration (188). Instead of passively maintaining tolerance, Tregs dynamically adjust to environmental cues in the inflamed gut, where they play key roles in managing damage, preserving epithelial integrity, and supporting tissue renewal (46). During mucosal injury or infection, Tregs are recruited to affected sites, where they help control excessive immune activation. By secreting anti-inflammatory cytokines like IL-10 and TGF-β, they reduce collateral tissue damage by modulating both innate and adaptive immune responses (189, 190). Importantly, their functions go beyond immune suppression. In the intestinal mucosa, Tregs support the survival of intestinal epithelial cells (IECs) (22), promoting barrier stability and preventing microbial translocation. These effects are partly mediated through IL-10-dependent pathways that strengthen tight junctions and prevent IEC apoptosis (191, 192).
In response to inflammatory signals and epithelial-derived alarmins such as IL-33, a subset of Tregs expressing the transcription factor GATA3 becomes functionally active. These GATA3+ IL-33R+ Tregs are essential for reducing immunopathology and aiding tissue recovery during infection and inflammation (78, 193). Their accumulation is vital for maintaining mucosal balance, especially in the small intestine, where losing GATA3 expression impairs Treg localization and function during enteric infections (193). These cells show transcriptional stability even in germ-free conditions, indicating a thymic origin and a specialized adaptation to the GI environment (194, 195). Tregs also reprogram the inflammatory microenvironment to support regeneration. One key process involves shifting macrophage phenotypes from pro-inflammatory M1 states to reparative M2 profiles (196, 197). This shift improves debris clearance and fosters conditions favorable for tissue remodeling (112). Additionally, Tregs produce several paracrine mediators, including AREG (198, 199), insulin-like growth factor 2 (IGF-2) (200), and CST7, all of which aid epithelial repair and functional recovery (188). The environment in which Tregs operate is crucial. While they protect against inflammation-related damage during infection or injury, their presence in the tumor microenvironment, such as in colorectal cancer (CRC), can have different effects. Depending on the cytokine environment and subset type, Tregs may either suppress tumor-promoting inflammation or facilitate immune evasion by reducing anti-tumor immunity (201, 202). Collectively, Tregs are increasingly recognized as critical mediators of gastrointestinal tissue regeneration, functioning at the interface between immune regulation and epithelial restoration.
Role of Treg in oral mucosa
The oral mucosa represents a unique immunological niche, continuously exposed to dietary antigens, commensal microbes, and mechanical stress. Within this dynamic environment, Tregs play a crucial role in maintaining immune balance and preserving epithelial integrity (203). Unlike other mucosal sites where local Treg induction is prominent, the oral mucosa is mainly populated by circulating Tregs that are recruited and acquire tissue-specific phenotypes, notably expressing CD103 and high levels of CTLA-4 to support retention and function within the oral tissue microenvironment (203). Mechanistically, Tregs suppress local effector T cell functions (Th1 and Th17). In Foxp3 knockout or IL-2 knockout mice (both of which lack functional Tregs), researchers observed a significant increase in Th1 (IFNγ+) and Th17 (IL-17A+) cells in the cervical draining lymph nodes, along with histological evidence of severe mucosal inflammation and damage, compared to wild-type controls (204). These findings emphasize that Tregs actively limit pathogenic T cell expansion to prevent mucosal injury.
Furthermore, the oral microenvironment is unique in that it minimally induces local peripheral Treg development; instead, the mucosa is mainly populated by recruited peripheral Tregs that adopt a tissue-resident phenotype (CD103+, CTLA-4 high) in situ, thereby maintaining homeostasis despite continuous exposure to food antigens, commensals, and mechanical stress (203). Tregs also influence microbiome-driven mucosal immunopathology. In murine models of oropharyngeal candidiasis, antibiotic disruption of resident bacteria resulted in decreased Foxp3+ Tregs and IL-17A+ cells, leading to worsened oral mucosal pathology (205, 206). Supplementing with short-chain fatty acids restored Treg frequencies (including Foxp3+IL-17A+ “Treg17” cells) and reduced tissue damage—showing that microbiota-derived metabolites support Treg-mediated protection of oral barrier tissues (206). The balance between Treg suppression and Th17 immunity is especially important. Some evidence indicates Tregs in the oral mucosa can promote protective Th17 responses by consuming IL-2 (a negative regulator of Th17 differentiation) and by producing TGF-β, thus allowing controlled IL-17 production that supports barrier immunity without causing pathological inflammation (207). This suggests that Tregs manage a finely tuned balance between tolerance and defense.
In disease contexts such as oral lichen planus (OLP), the role of Tregs appears complex. Immunohistochemical analysis of FOXP3+ Tregs in OLP lesions revealed correlations with disease activity, suggesting that Tregs may be either insufficient or dysregulated in chronic inflammatory conditions of the oral mucosa (208). Functional studies further support this, showing that FOXP3+ Tregs in OLP patients express reduced levels of immunosuppressive cytokines like TGF-β and demonstrate impaired suppressive capacity, indicating a loss of regulatory efficiency despite elevated cell counts (209). While CD8+ cytotoxic T cells mediate keratinocyte apoptosis in OLP, inadequate Treg suppression likely contributes to unchecked local immune activation. In contrast, evidence from celiac disease further illustrates the dynamic role of Tregs in oral mucosal injury. Sanchez-Solares et al. (2021) demonstrated that celiac disease disrupts oral epithelial integrity, shown by reduced E-cadherin and claudin-1 expression. This epithelial injury was accompanied by an increased infiltration of Foxp3+ Tregs and elevated expression of AREG, a key tissue repair molecule. The positive correlation between Treg presence and Areg expression suggests a compensatory, pro-repair function of Tregs in response to mucosal damage (210). Together, these findings indicate that Tregs may play dual roles in oral mucosal diseases—being insufficient or dysfunctional in chronic inflammation like OLP, while adopting a reparative phenotype in barrier-restoration settings such as celiac disease.
Role of Treg in genitourinary
The genitourinary (GU) tract, including urinary and reproductive organs, requires a finely tuned immune balance to tolerate beneficial agents like sperm and commensals while defending against pathogens such as sexually transmitted infections. Tregs have emerged as central mediators in orchestrating this immune balance, modulating immune responses to favor tissue protection and homeostasis within the GU tract (46). For a successful and healthy pregnancy, the maternal immune system must accept both sperm and the fetus, which expresses paternal antigens and is therefore partially foreign. Tregs present in maternal blood and at the maternal–fetal interface has been shown to expand during gestation, playing a crucial role in preventing fetal rejection, spontaneous abortion, and complications like preeclampsia in both humans and animal models (211–214). In mice, Treg accumulation begins shortly after first contact with seminal antigens, even before embryo implantation, with recruitment seen in the uterus and draining lymph nodes (215–217). During pregnancy, the number of CD25+ T cells doubles in iliac and inguinal lymph nodes, and Tregs make up roughly 30% of uterine CD4+ T cells. Additionally, uterine Foxp3 expression increases nearly 1,000-fold compared to non-pregnant counterparts, indicating a significant surge in local Treg populations (211). Treg deficiency early in gestation has been linked to impaired uterine artery remodeling, supporting their role in preventing hypertensive disorders like preeclampsia (218). Immune suppression observed during the luteal phase of the reproductive cycle and after exposure to seminal extracellular vesicles suggests that Tregs in the uterus and vagina may facilitate early pregnancy through localized immunosuppression (219–223). Growing interest in Treg-based therapies highlights their potential to address pregnancy-related complications. However, further clinical research is needed to develop diagnostic tools for early pregnancy Treg profiling and to identify optimal therapeutic windows (224).
Cervical and vaginal Tregs
The healthy vaginal ecosystem is usually dominated by Lactobacillus species, which help prevent colonization by harmful bacteria and fungi. Disruption of this balance (dysbiosis) can cause overgrowth of damaging microorganisms, leading to bacterial vaginosis, candidiasis, urinary tract infections, and a disrupted vaginal pH—factors that collectively increase the risk of STIs and infertility (225, 226). Like the gastrointestinal system, vaginal immunity must tolerate beneficial microbes while remaining able to respond properly to infections. Growing evidence shows that Tregs support this balance. For example, Lactobacillus crispatus, a common commensal species, has been shown to induce Treg differentiation from naive CD4+ T cells in vitro (227). Additionally, microbial imbalance is often linked with increased inflammatory cytokines and decreased Treg levels in peripheral blood, suggesting that loss of microbial harmony may shift immune responses from regulation to inflammation (226, 228, 229). These findings highlight the need for further research on how vaginal Tregs mediate the balance between tolerance and immunity based on microbial signals. Tregs in the uterine and vaginal mucosa not only support homeostasis and pregnancy but also help in immune responses to infections. In mouse models of vaginal HSV-2 infection, Tregs in the draining lymph nodes are crucial for proper movement of antigen-presenting dendritic cells from the infection site. When these Tregs are absent, virus-specific immune responses are delayed, emphasizing their vital role in antiviral defense (46, 214).
Future prospective
The increasing focus on the immune system’s role in tissue repair and regeneration reflects a shift in regenerative medicine. However, a significant knowledge gap remains about how the adaptive immune system—especially Treg cells—affects tissue healing. Important questions include whether neoantigens produced during injury can trigger specific repair responses, and how T cells distinguish between self- and non-self-antigens in this process. The mechanisms behind T cell recruitment, activation, and function during tissue injury are still not fully understood. To address these issues, future research should utilize advanced models such as antigen tracing in transgenic reporter mice and mass spectrometry to discover neoantigens specific to injury that may induce Treg recruitment and activation. At the same time, combining lineage tracing with single-cell transcriptomics can reveal Treg clonal behavior and antigen specificity in their local environments, shedding light on how individual clones contribute to repair in specific tissues. Progress in immune-based regenerative therapies is also slowed by the lack of specific markers for immune cell subsets involved in tissue repair. Advances in single-cell genomics and multi-omics profiling may identify lineage- and function-specific markers that differentiate reparative immune cells from non-reparative ones. Additionally, the difference between tissues that regenerate without scarring (such as bone) and those prone to fibrosis (like the heart and liver) remains unclear. Gaining a deeper understanding of how immune cells, tissue-resident progenitors, and myofibroblasts interact could reveal mechanisms that promote regeneration over fibrosis and help develop targeted repair strategies.
Another important aspect is the impact of aging on immune-mediated tissue repair. The decline in immune function with age may impair Treg recruitment, function, or stability, ultimately reducing regenerative capacity in older individuals. However, most current studies on Treg function in tissue repair rely on the Foxp3DTR mouse model, which has limitations due to the development of systemic autoimmunity with prolonged Treg depletion. To address this, future studies should include gain-of-function approaches, such as adoptive transfer of purified Tregs into Rag1+/+ mice, to assess Treg function without the confounding effects of autoimmunity. Emerging conditional genetic models, like Foxp3-CreERT2 mice, offer better temporal control over Treg depletion and avoid long-term systemic effects. When combined with adoptive transfer into lymphopenic hosts, these models enable precise examination of Treg stability, plasticity, and reparative functions over time in vivo. These tools are crucial for moving beyond correlative findings toward mechanistic understanding. From a therapeutic standpoint, modulating tissue-resident Tregs to enhance local repair is a promising strategy. Treg populations in tissues such as the lung, skeletal muscle, and skin help maintain local immune homeostasis. Notably, IL-33 signaling expands tissue-resident Tregs and boosts their reparative ability, partly through the induction of AREG, a key factor in tissue repair. However, the full functional diversity among these Tregs and their direct roles in regeneration are still not fully understood.
To translate preclinical findings into human applications, advanced organoid systems and humanized mouse models serve as powerful platforms. Organoids derived from lung, skin, and gut tissues that include immune components can mimic tissue-specific microenvironments, enabling detailed mechanistic and therapeutic screening. Similarly, humanized mice with reconstituted human immune systems provide an in vivo environment to study human Treg trafficking, stability, and antigen specificity during tissue injury and repair. Despite their therapeutic potential, Treg-based regenerative treatments encounter key obstacles. These include maintaining the phenotypic stability of Tregs after transfer, ensuring effective trafficking and retention in injured tissues, and selecting antigen-specific Tregs to prevent broad immunosuppression. Overcoming these hurdles is essential for fully realizing the therapeutic potential of Treg-based strategies for tissue repair and regeneration.
Author contributions
AR: Writing – original draft. IK: Writing – original draft. IA: Writing – original draft. AW: Writing – original draft. MA: Writing – review & editing. FA: Writing – review & editing. KA: Writing – review & editing. AQ: Writing – review & editing. I-CL: Writing – review & editing. C-CC: Writing – review & editing.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. The authors are thankful to the Deanship of Research and Graduate Studies, King Khalid University, Abha, Saudi Arabia, for financially supporting this work through the Large Research Group Project under Grant No. R.G.P.2/724/46.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Lozano R, Naghavi M, Foreman K, Lim S, Shibuya K, Aboyans V, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the global burden of disease study 2010. Lancet. (2012) 380:2095–128. doi: 10.1016/s0140-6736(12)61728-0
2. Schwartz SD, Hubschman JP, Heilwell G, Franco-Cardenas V, Pan CK, Ostrick RM, et al. Embryonic stem cell trials for macular degeneration: A preliminary report. Lancet. (2012) 379:713–20. doi: 10.1016/s0140-6736(12)60028-2
3. Schwartz SD, Regillo CD, Lam BL, Eliott D, Rosenfeld PJ, Gregori NZ, et al. Human embryonic stem cell-derived retinal pigment epithelium in patients with age-related macular degeneration and stargardt’s macular dystrophy: follow-up of two open-label phase 1/2 studies. Lancet. (2015) 385:509–16. doi: 10.1016/s0140-6736(14)61376-3
4. Lui KO, Fairchild PJ, and Waldmann H. Prospects for ensuring acceptance of es cell-derived tissues. In: Lui KO and Paul J, editors. Stembook. Harvard Stem Cell Institute Copyright, Cambridge (MA (2010). Fairchild and Herman Waldmann. (2008).
5. Lui KO, Waldmann H, and Fairchild PJ. Embryonic stem cells: overcoming the immunological barriers to cell replacement therapy. Curr Stem Cell Res Ther. (2009) 4:70–80. doi: 10.2174/157488809787169093
6. Martino MM, Briquez PS, Maruyama K, and Hubbell JA. Extracellular matrix-inspired growth factor delivery systems for bone regeneration. Adv Drug Delivery Rev. (2015) 94:41–52. doi: 10.1016/j.addr.2015.04.007
7. Briquez PS, Hubbell JA, and Martino MM. Extracellular matrix-inspired growth factor delivery systems for skin wound healing. Adv Wound Care (New Rochelle). (2015) 4:479–89. doi: 10.1089/wound.2014.0603
8. Sattler S. The role of the immune system beyond the fight against infection. Adv Exp Med Biol. (2017) 1003:3–14. doi: 10.1007/978-3-319-57613-8_1
9. Li J, Tan J, Martino MM, and Lui KO. Regulatory T-cells: potential regulator of tissue repair and regeneration. Front Immunol. (2018) 9:585. doi: 10.3389/fimmu.2018.00585
10. Gama JFG, Romualdo RD, de Assis ML, de Oliveira LM, Quírico-Santos T, Alves LA, et al. Role of regulatory T cells in skeletal muscle regeneration: A systematic review. Biomolecules. (2022) 12. doi: 10.3390/biom12060817
11. Burzyn D, Kuswanto W, Kolodin D, Shadrach JL, Cerletti M, Jang Y, et al. A special population of regulatory T cells potentiates muscle repair. Cell. (2013) 155:1282–95. doi: 10.1016/j.cell.2013.10.054
12. Forbes SJ and Rosenthal N. Preparing the ground for tissue regeneration: from mechanism to therapy. Nat Med. (2014) 20:857–69. doi: 10.1038/nm.3653
13. Bosurgi L, Cao YG, Cabeza-Cabrerizo M, Tucci A, Hughes LD, Kong Y, et al. Macrophage function in tissue repair and remodeling requires il-4 or il-13 with apoptotic cells. Science. (2017) 356:1072–6. doi: 10.1126/science.aai8132
14. Kanamori M, Nakatsukasa H, Okada M, Lu Q, and Yoshimura A. Induced regulatory T cells: their development, stability, and applications. Trends Immunol. (2016) 37:803–11. doi: 10.1016/j.it.2016.08.012
15. Burzyn D, Benoist C, and Mathis D. Regulatory T cells in nonlymphoid tissues. Nat Immunol. (2013) 14:1007–13. doi: 10.1038/ni.2683
16. Liston A and Gray DH. Homeostatic control of regulatory T cell diversity. Nat Rev Immunol. (2014) 14:154–65. doi: 10.1038/nri3605
17. Lei H, Schmidt-Bleek K, Dienelt A, Reinke P, and Volk HD. Regulatory T cell-mediated anti-inflammatory effects promote successful tissue repair in both indirect and direct manners. Front Pharmacol. (2015) 6:184. doi: 10.3389/fphar.2015.00184
18. Panduro M, Benoist C, and Mathis D. Tissue tregs. Annu Rev Immunol. (2016) 34:609–33. doi: 10.1146/annurev-immunol-032712-095948
19. Zhou X, Tang J, Cao H, Fan H, and Li B. Tissue resident regulatory T cells: novel therapeutic targets for human disease. Cell Mol Immunol. (2015) 12:543–52. doi: 10.1038/cmi.2015.23
20. Arpaia N, Green JA, Moltedo B, Arvey A, Hemmers S, Yuan S, et al. A distinct function of regulatory T cells in tissue protection. Cell. (2015) 162:1078–89. doi: 10.1016/j.cell.2015.08.021
21. Boothby IC, Cohen JN, and Rosenblum MD. Regulatory T cells in skin injury: at the crossroads of tolerance and tissue repair. Sci Immunol. (2020) 5. doi: 10.1126/sciimmunol.aaz9631
22. Loffredo LF, Savage TM, Ringham OR, and Arpaia N. Treg-tissue cell interactions in repair and regeneration. J Exp Med. (2024) 221. doi: 10.1084/jem.20231244
23. Chen X and Oppenheim JJ. Resolving the identity myth: key markers of functional cd4+Foxp3+ Regulatory T cells. Int Immunopharmacol. (2011) 11:1489–96. doi: 10.1016/j.intimp.2011.05.018
24. Fontenot JD, Gavin MA, and Rudensky AY. Foxp3 programs the development and function of cd4+Cd25+ Regulatory T cells. Nat Immunol. (2003) 4:330–6. doi: 10.1038/ni904
25. Hori S, Nomura T, and Sakaguchi S. Control of regulatory T cell development by the transcription factor foxp3. Science. (2003) 299:1057–61. doi: 10.1126/science.1079490
26. Khattri R, Cox T, Yasayko SA, and Ramsdell F. An essential role for scurfin in cd4+Cd25+ T regulatory cells. Nat Immunol. (2003) 4:337–42. doi: 10.1038/ni909
27. Williams LM and Rudensky AY. Maintenance of the foxp3-dependent developmental program in mature regulatory T cells requires continued expression of foxp3. Nat Immunol. (2007) 8:277–84. doi: 10.1038/ni1437
28. Miyara M, Yoshioka Y, Kitoh A, Shima T, Wing K, Niwa A, et al. Functional delineation and differentiation dynamics of human cd4+ T cells expressing the foxp3 transcription factor. Immunity. (2009) 30:899–911. doi: 10.1016/j.immuni.2009.03.019
29. Liu W, Putnam AL, Xu-Yu Z, Szot GL, Lee MR, Zhu S, et al. Cd127 expression inversely correlates with foxp3 and suppressive function of human cd4+ T reg cells. J Exp Med. (2006) 203:1701–11. doi: 10.1084/jem.20060772
30. Shevyrev D and Tereshchenko V. Treg heterogeneity, function, and homeostasis. Front Immunol. (2019) 10:3100. doi: 10.3389/fimmu.2019.03100
31. Liu Y, Zhang P, Li J, Kulkarni AB, Perruche S, and Chen W. A critical function for tgf-beta signaling in the development of natural cd4+Cd25+Foxp3+ Regulatory T cells. Nat Immunol. (2008) 9:632–40. doi: 10.1038/ni.1607
32. Lio CW and Hsieh CS. A two-step process for thymic regulatory T cell development. Immunity. (2008) 28:100–11. doi: 10.1016/j.immuni.2007.11.021
33. Curotto de Lafaille MA and Lafaille JJ. Natural and adaptive foxp3+ Regulatory T cells: more of the same or a division of labor? Immunity. (2009) 30:626–35. doi: 10.1016/j.immuni.2009.05.002
34. Fang D and Zhu J. Dynamic balance between master transcription factors determines the fates and functions of cd4 T cell and innate lymphoid cell subsets. J Exp Med. (2017) 214:1861–76. doi: 10.1084/jem.20170494
35. Ito M, Komai K, Nakamura T, Srirat T, and Yoshimura A. Tissue regulatory T cells and neural repair. Int Immunol. (2019) 31:361–9. doi: 10.1093/intimm/dxz031
36. Delacher M, Imbusch CD, Weichenhan D, Breiling A, Hotz-Wagenblatt A, Träger U, et al. Genome-wide DNA-methylation landscape defines specialization of regulatory T cells in tissues. Nat Immunol. (2017) 18:1160–72. doi: 10.1038/ni.3799
37. Becker M, Joseph SS, Garcia-Carrizo F, Tom RZ, Opaleva D, Serr I, et al. Regulatory T cells require il6 receptor alpha signaling to control skeletal muscle function and regeneration. Cell Metab. (2023) 35:1736–51.e7. doi: 10.1016/j.cmet.2023.08.010
38. Wu J, Ren B, Wang D, and Lin H. Regulatory T cells in skeletal muscle repair and regeneration: recent insights. Cell Death Dis. (2022) 13:680. doi: 10.1038/s41419-022-05142-8
39. Knoedler S, Knoedler L, Kauke-Navarro M, Rinkevich Y, Hundeshagen G, Harhaus L, et al. Regulatory T cells in skin regeneration and wound healing. Mil Med Res. (2023) 10:49. doi: 10.1186/s40779-023-00484-6
40. Luan J, Truong C, Vuchkovska A, Guo W, Good J, Liu B, et al. Cd80 on Skin Stem Cells Promotes Local Expansion of Regulatory T cells Upon Injury to Orchestrate Repair within an Inflammatory Environment. Immunity. (2024) 57:1071–86.e7. doi: 10.1016/j.immuni.2024.04.003
41. Nayer B, Tan JL, Alshoubaki YK, Lu YZ, Legrand JMD, Lau S, et al. Local administration of regulatory T cells promotes tissue healing. Nat Commun. (2024) 15:7863. doi: 10.1038/s41467-024-51353-2
42. Jiang JF, Lu HY, Wang MY, He LY, Zhu Y, and Qiao Y. Role of regulatory T cells in mouse lung development. Exp Biol Med (Maywood). (2024) 249:10040. doi: 10.3389/ebm.2024.10040
43. Fung THW, Yang KY, and Lui KO. An emerging role of regulatory T-cells in cardiovascular repair and regeneration. Theranostics. (2020) 10:8924–38. doi: 10.7150/thno.47118
44. de la Vega Gallardo N, Dittmer M, Dombrowski Y, and Fitzgerald DC. Regenerating cns myelin: emerging roles of regulatory T cells and ccn proteins. Neurochem Int. (2019) 130:104349. doi: 10.1016/j.neuint.2018.11.024
45. Hu W, Li J, and Cheng X. Regulatory T cells and cardiovascular diseases. Chin (Engl). (2023) 136:2812–23. doi: 10.1097/cm9.0000000000002875
46. Traxinger BR, Richert-Spuhler LE, and Lund JM. Mucosal tissue regulatory T cells are integral in balancing immunity and tolerance at portals of antigen entry. Mucosal Immunol. (2022) 15:398–407. doi: 10.1038/s41385-021-00471-x
47. Ahmad K, Shaikh S, Chun HJ, Ali S, Lim JH, Ahmad SS, et al. Extracellular matrix: the critical contributor to skeletal muscle regeneration-a comprehensive review. Inflammation Regener. (2023) 43:58. doi: 10.1186/s41232-023-00308-z
48. Hung M, Lo HF, Jones GEL, and Krauss RS. The muscle stem cell niche at a glance. J Cell Sci. (2023) 136. doi: 10.1242/jcs.261200
49. Byun WS, Lee J, and Baek JH. Beyond the bulk: overview and novel insights into the dynamics of muscle satellite cells during muscle regeneration. Inflammation Regener. (2024) 44:39. doi: 10.1186/s41232-024-00354-1
50. Tu H and Li YL. Inflammation balance in skeletal muscle damage and repair. Front Immunol. (2023) 14:1133355. doi: 10.3389/fimmu.2023.1133355
51. Yang W and Hu P. Skeletal muscle regeneration is modulated by inflammation. J Orthop Translat. (2018) 13:25–32. doi: 10.1016/j.jot.2018.01.002
52. Fu X, Wang H, and Hu P. Stem cell activation in skeletal muscle regeneration. Cell Mol Life Sci. (2015) 72:1663–77. doi: 10.1007/s00018-014-1819-5
53. Koike H, Manabe I, and Oishi Y. Mechanisms of cooperative cell-cell interactions in skeletal muscle regeneration. Inflammation Regener. (2022) 42:48. doi: 10.1186/s41232-022-00234-6
54. Waldemer-Streyer RJ, Kim D, and Chen J. Muscle cell-derived cytokines in skeletal muscle regeneration. FEBS J. (2022) 289:6463–83. doi: 10.1111/febs.16372
55. Fang J, Feng C, Chen W, Hou P, Liu Z, Zuo M, et al. Redressing the interactions between stem cells and immune system in tissue regeneration. Biol Direct. (2021) 16:18. doi: 10.1186/s13062-021-00306-6
56. Villalta SA, Rosenthal W, Martinez L, Kaur A, Sparwasser T, Tidball JG, et al. Regulatory T cells suppress muscle inflammation and injury in muscular dystrophy. Sci Transl Med. (2014) 6:258ra142. doi: 10.1126/scitranslmed.3009925
57. Kuswanto W, Burzyn D, Panduro M, Wang KK, Jang YC, Wagers AJ, et al. Poor repair of skeletal muscle in aging mice reflects a defect in local, interleukin-33-dependent accumulation of regulatory T cells. Immunity. (2016) 44:355–67. doi: 10.1016/j.immuni.2016.01.009
58. Castiglioni A, Corna G, Rigamonti E, Basso V, Vezzoli M, Monno A, et al. Foxp3+ T cells recruited to sites of sterile skeletal muscle injury regulate the fate of satellite cells and guide effective tissue regeneration. PloS One. (2015) 10:e0128094. doi: 10.1371/journal.pone.0128094
59. Dort J, Fabre P, Molina T, and Dumont NA. Macrophages are key regulators of stem cells during skeletal muscle regeneration and diseases. Stem Cells Int. (2019) 2019:4761427. doi: 10.1155/2019/4761427
60. Chen Z, Lan H, Liao Z, Huang J, Jian X, Hu J, et al. Regulatory T cells-centered regulatory networks of skeletal muscle inflammation and regeneration. Cell Biosci. (2022) 12:112. doi: 10.1186/s13578-022-00847-x
61. Lemos DR, Babaeijandaghi F, Low M, Chang CK, Lee ST, Fiore D, et al. Nilotinib reduces muscle fibrosis in chronic muscle injury by promoting tnf-mediated apoptosis of fibro/adipogenic progenitors. Nat Med. (2015) 21:786–94. doi: 10.1038/nm.3869
62. D’Alessio FR, Kurzhagen JT, and Rabb H. Reparative T lymphocytes in organ injury. J Clin Invest. (2019) 129:2608–18. doi: 10.1172/jci124614
63. Tiemessen MM, Jagger AL, Evans HG, van Herwijnen MJ, John S, and Taams LS. Cd4+Cd25+Foxp3+ Regulatory T cells induce alternative activation of human monocytes/macrophages. Proc Natl Acad Sci U.S.A. (2007) 104:19446–51. doi: 10.1073/pnas.0706832104
64. Panduro M, Benoist C, and Mathis D. T(Reg) cells limit ifn-Γ Production to control macrophage accrual and phenotype during skeletal muscle regeneration. Proc Natl Acad Sci U.S.A. (2018) 115:E2585–e93. doi: 10.1073/pnas.1800618115
65. Muñoz-Rojas AR and Mathis D. Tissue regulatory T cells: regulatory chameleons. Nat Rev Immunol. (2021) 21:597–611. doi: 10.1038/s41577-021-00519-w
66. Ding M, Huang T, Zhu R, Gu R, Shi D, Xiao J, et al. Immunological behavior analysis of muscle cells under ifn-Γ Stimulation in vitro and in vivo. Anat Rec (Hoboken). (2018) 301:1551–63. doi: 10.1002/ar.23834
67. Steeg PS, Moore RN, Johnson HM, and Oppenheim JJ. Regulation of murine macrophage ia antigen expression by a lymphokine with immune interferon activity. J Exp Med. (1982) 156:1780–93. doi: 10.1084/jem.156.6.1780
68. Zaiss DM, van Loosdregt J, Gorlani A, Bekker CP, Gröne A, Sibilia M, et al. Amphiregulin enhances regulatory T cell-suppressive function via the epidermal growth factor receptor. Immunity. (2013) 38:275–84. doi: 10.1016/j.immuni.2012.09.023
69. Burzyn D, Wagers A, Mathis D, and Cerletti M. Methods of isolation and analysis of treg immune infiltrates from injured and dystrophic skeletal muscle. Methods Mol Biol. (2019) 1899:229–37. doi: 10.1007/978-1-4939-8938-6_16
70. Zhang C, Qiao Y, Huang L, Li F, Zhang Z, Ping Y, et al. Regulatory T cells were recruited by ccl3 to promote cryo-injured muscle repair. Immunol Lett. (2018) 204:29–37. doi: 10.1016/j.imlet.2018.10.004
71. Jin RM, Warunek J, and Wohlfert EA. Therapeutic administration of il-10 and amphiregulin alleviates chronic skeletal muscle inflammation and damage induced by infection. Immunohorizons. (2018) 2:142–54. doi: 10.4049/immunohorizons.1800024
72. Dai K, Huang L, Sun X, Yang L, and Gong Z. Hepatic cd206-positive macrophages express amphiregulin to promote the immunosuppressive activity of regulatory T cells in hbv infection. J Leukoc Biol. (2015) 98:1071–80. doi: 10.1189/jlb.4A0415-152R
73. Wang S, Zhang Y, Wang Y, Ye P, Li J, Li H, et al. Amphiregulin confers regulatory T cell suppressive function and tumor invasion via the egfr/gsk-3β/foxp3 axis. J Biol Chem. (2016) 291:21085–95. doi: 10.1074/jbc.M116.717892
74. Delacher M, Imbusch CD, Hotz-Wagenblatt A, Mallm JP, Bauer K, Simon M, et al. Precursors for nonlymphoid-tissue treg cells reside in secondary lymphoid organs and are programmed by the transcription factor batf. Immunity. (2020) 52:295–312.e11. doi: 10.1016/j.immuni.2019.12.002
75. Lam AJ, MacDonald KN, Pesenacker AM, Juvet SC, Morishita KA, Bressler B, et al. Innate control of tissue-reparative human regulatory T cells. J Immunol. (2019) 202:2195–209. doi: 10.4049/jimmunol.1801330
76. Peine M, Marek RM, and Löhning M. Il-33 in T cell differentiation, function, and immune homeostasis. Trends Immunol. (2016) 37:321–33. doi: 10.1016/j.it.2016.03.007
77. Wang K, Yaghi OK, Spallanzani RG, Chen X, Zemmour D, Lai N, et al. Neuronal, stromal, and T-regulatory cell crosstalk in murine skeletal muscle. Proc Natl Acad Sci U.S.A. (2020) 117:5402–8. doi: 10.1073/pnas.1922559117
78. Schiering C, Krausgruber T, Chomka A, Fröhlich A, Adelmann K, Wohlfert EA, et al. The alarmin il-33 promotes regulatory T-cell function in the intestine. Nature. (2014) 513:564–8. doi: 10.1038/nature13577
79. Knipper JA, Willenborg S, Brinckmann J, Bloch W, Maaß T, Wagener R, et al. Interleukin-4 receptor α Signaling in myeloid cells controls collagen fibril assembly in skin repair. Immunity. (2015) 43:803–16. doi: 10.1016/j.immuni.2015.09.005
80. Shou J, Shi X, Liu X, Chen Y, Chen P, and Xiao W. Programmed death-1 promotes contused skeletal muscle regeneration by regulating treg cells and macrophages. Lab Invest. (2021) 101:719–32. doi: 10.1038/s41374-021-00542-4
81. Chen X, Fosco D, Kline DE, Meng L, Nishi S, Savage PA, et al. Pd-1 regulates extrathymic regulatory T-cell differentiation. Eur J Immunol. (2014) 44:2603–16. doi: 10.1002/eji.201344423
82. Liao ZH, Huang T, Xiao JW, Gu RC, Ouyang J, Wu G, et al. Estrogen signaling effects on muscle-specific immune responses through controlling the recruitment and function of macrophages and T cells. Skelet Muscle. (2019) 9:20. doi: 10.1186/s13395-019-0205-2
83. Mohammad I, Starskaia I, Nagy T, Guo J, Yatkin E, Väänänen K, et al. Estrogen receptor α Contributes to T cell-mediated autoimmune inflammation by promoting T cell activation and proliferation. Sci Signal. (2018) 11. doi: 10.1126/scisignal.aap9415
84. Malko D, Elmzzahi T, and Beyer M. Implications of regulatory T cells in non-lymphoid tissue physiology and pathophysiology. Front Immunol. (2022) 13:954798. doi: 10.3389/fimmu.2022.954798
85. Ito M, Komai K, Mise-Omata S, Iizuka-Koga M, Noguchi Y, Kondo T, et al. Brain regulatory T cells suppress astrogliosis and potentiate neurological recovery. Nature. (2019) 565:246–50. doi: 10.1038/s41586-018-0824-5
86. Yoshimura A, Wakabayashi Y, and Mori T. Cellular and molecular basis for the regulation of inflammation by tgf-beta. J Biochem. (2010) 147:781–92. doi: 10.1093/jb/mvq043
87. Wing JB and Sakaguchi S. Multiple treg suppressive modules and their adaptability. Front Immunol. (2012) 3:178. doi: 10.3389/fimmu.2012.00178
88. Lenglet S, Louiset E, Delarue C, Vaudry H, and Contesse V. Activation of 5-ht(7) receptor in rat glomerulosa cells is associated with an increase in adenylyl cyclase activity and calcium influx through T-type calcium channels. Endocrinology. (2002) 143:1748–60. doi: 10.1210/endo.143.5.8817
89. Klein M and Bopp T. Cyclic amp represents a crucial component of treg cell-mediated immune regulation. Front Immunol. (2016) 7:315. doi: 10.3389/fimmu.2016.00315
90. Dombrowski Y, O’Hagan T, Dittmer M, Penalva R, Mayoral SR, Bankhead P, et al. Regulatory T cells promote myelin regeneration in the central nervous system. Nat Neurosci. (2017) 20:674–80. doi: 10.1038/nn.4528
91. Hui SP, Sheng DZ, Sugimoto K, Gonzalez-Rajal A, Nakagawa S, Hesselson D, et al. Zebrafish regulatory T cells mediate organ-specific regenerative programs. Dev Cell. (2017) 43:659–72.e5. doi: 10.1016/j.devcel.2017.11.010
92. Lázár E, Sadek HA, and Bergmann O. Cardiomyocyte renewal in the human heart: insights from the fall-out. Eur Heart J. (2017) 38:2333–42. doi: 10.1093/eurheartj/ehx343
93. Porrello ER, Mahmoud AI, Simpson E, Hill JA, Richardson JA, Olson EN, et al. Transient regenerative potential of the neonatal mouse heart. Science. (2011) 331:1078–80. doi: 10.1126/science.1200708
94. Lam NT and Sadek HA. Neonatal heart regeneration: comprehensive literature review. Circulation. (2018) 138:412–23. doi: 10.1161/circulationaha.118.033648
95. Weirather J, Hofmann UD, Beyersdorf N, Ramos GC, Vogel B, Frey A, et al. Foxp3+ Cd4+ T cells improve healing after myocardial infarction by modulating monocyte/macrophage differentiation. Circ Res. (2014) 115:55–67. doi: 10.1161/circresaha.115.303895
96. Li J, Yang KY, Tam RCY, Chan VW, Lan HY, Hori S, et al. Regulatory T-cells regulate neonatal heart regeneration by potentiating cardiomyocyte proliferation in a paracrine manner. Theranostics. (2019) 9:4324–41. doi: 10.7150/thno.32734
97. Zacchigna S, Martinelli V, Moimas S, Colliva A, Anzini M, Nordio A, et al. Paracrine effect of regulatory T cells promotes cardiomyocyte proliferation during pregnancy and after myocardial infarction. Nat Commun. (2018) 9:2432. doi: 10.1038/s41467-018-04908-z
98. Tang TT, Yuan J, Zhu ZF, Zhang WC, Xiao H, Xia N, et al. Regulatory T cells ameliorate cardiac remodeling after myocardial infarction. Basic Res Cardiol. (2012) 107:232. doi: 10.1007/s00395-011-0232-6
99. Li J, Liang C, Yang KY, Huang X, Han MY, Li X, et al. Specific ablation of cd4(+) T-cells promotes heart regeneration in juvenile mice. Theranostics. (2020) 10:8018–35. doi: 10.7150/thno.42943
100. Xia N, Jiao J, Tang TT, Lv BJ, Lu YZ, Wang KJ, et al. Activated regulatory T-cells attenuate myocardial ischaemia/reperfusion injury through a cd39-dependent mechanism. Clin Sci (Lond). (2015) 128:679–93. doi: 10.1042/cs20140672
101. Borsellino G, Kleinewietfeld M, Di Mitri D, Sternjak A, Diamantini A, Giometto R, et al. Expression of ectonucleotidase cd39 by foxp3+ Treg cells: hydrolysis of extracellular atp and immune suppression. Blood. (2007) 110:1225–32. doi: 10.1182/blood-2006-12-064527
102. Borg N, Alter C, Görldt N, Jacoby C, Ding Z, Steckel B, et al. Cd73 on T cells orchestrates cardiac wound healing after myocardial infarction by purinergic metabolic reprogramming. Circulation. (2017) 136:297–313. doi: 10.1161/circulationaha.116.023365
103. Yellon DM, Beikoghli Kalkhoran S, and Davidson SM. The risk pathway leading to mitochondria and cardioprotection: how everything started. Basic Res Cardiol. (2023) 118:22. doi: 10.1007/s00395-023-00992-5
104. Wang Y, Li J, Zhang Y, He P, Liu W, Zeng W, et al. Areg(+) regulatory T cells mediating myocardial repair and neovascularization after myocardial infarction. Mol Med. (2025) 31:229. doi: 10.1186/s10020-025-01281-8
105. Sziksz E, Pap D, Lippai R, Béres NJ, Fekete A, Szabó AJ, et al. Fibrosis related inflammatory mediators: role of the il-10 cytokine family. Mediators Inflammation. (2015) 2015:764641. doi: 10.1155/2015/764641
106. Debock I and Flamand V. Unbalanced neonatal cd4(+) T-cell immunity. Front Immunol. (2014) 5:393. doi: 10.3389/fimmu.2014.00393
107. Josefowicz SZ, Lu LF, and Rudensky AY. Regulatory T cells: mechanisms of differentiation and function. Annu Rev Immunol. (2012) 30:531–64. doi: 10.1146/annurev.immunol.25.022106.141623
108. Levine AG, Mendoza A, Hemmers S, Moltedo B, Niec RE, Schizas M, et al. Stability and function of regulatory T cells expressing the transcription factor T-bet. Nature. (2017) 546:421–5. doi: 10.1038/nature22360
109. Cipolletta D, Feuerer M, Li A, Kamei N, Lee J, Shoelson SE, et al. Ppar-Γ Is a major driver of the accumulation and phenotype of adipose tissue treg cells. Nature. (2012) 486:549–53. doi: 10.1038/nature11132
110. Vasanthakumar A, Moro K, Xin A, Liao Y, Gloury R, Kawamoto S, et al. The transcriptional regulators irf4, batf and il-33 orchestrate development and maintenance of adipose tissue-resident regulatory T cells. Nat Immunol. (2015) 16:276–85. doi: 10.1038/ni.3085
111. Lloyd CM and Snelgrove RJ. Type 2 immunity: expanding our view. Sci Immunol. (2018) 3. doi: 10.1126/sciimmunol.aat1604
112. Li C, DiSpirito JR, Zemmour D, Spallanzani RG, Kuswanto W, Benoist C, et al. Tcr transgenic mice reveal stepwise, multi-site acquisition of the distinctive fat-treg phenotype. Cell. (2018) 174:285–99.e12. doi: 10.1016/j.cell.2018.05.004
113. Spath S, Roan F, Presnell SR, Höllbacher B, and Ziegler SF. Profiling of tregs across tissues reveals plasticity in st2 expression and hierarchies in tissue-specific phenotypes. iScience. (2022) 25:104998. doi: 10.1016/j.isci.2022.104998
114. Kashiwagi M, Hosoi J, Lai JF, Brissette J, Ziegler SF, Morgan BA, et al. Direct control of regulatory T cells by keratinocytes. Nat Immunol. (2017) 18:334–43. doi: 10.1038/ni.3661
115. Ricardo-Gonzalez RR, Van Dyken SJ, Schneider C, Lee J, Nussbaum JC, Liang HE, et al. Tissue signals imprint ilc2 identity with anticipatory function. Nat Immunol. (2018) 19:1093–9. doi: 10.1038/s41590-018-0201-4
116. Harrison OJ, Linehan JL, Shih HY, Bouladoux N, Han SJ, Smelkinson M, et al. Commensal-specific T cell plasticity promotes rapid tissue adaptation to injury. Science. (2019) 363. doi: 10.1126/science.aat6280
117. Nosbaum A, Prevel N, Truong HA, Mehta P, Ettinger M, Scharschmidt TC, et al. Cutting edge: regulatory T cells facilitate cutaneous wound healing. J Immunol. (2016) 196:2010–4. doi: 10.4049/jimmunol.1502139
118. Mathur AN, Zirak B, Boothby IC, Tan M, Cohen JN, Mauro TM, et al. Treg-cell control of a cxcl5-il-17 inflammatory axis promotes hair-follicle-stem-cell differentiation during skin-barrier repair. Immunity. (2019) 50:655–67.e4. doi: 10.1016/j.immuni.2019.02.013
119. Masopust D and Schenkel JM. The integration of T cell migration, differentiation and function. Nat Rev Immunol. (2013) 13:309–20. doi: 10.1038/nri3442
120. Zaiss DMW, Gause WC, Osborne LC, and Artis D. Emerging functions of amphiregulin in orchestrating immunity, inflammation, and tissue repair. Immunity. (2015) 42:216–26. doi: 10.1016/j.immuni.2015.01.020
121. Werner S and Grose R. Regulation of wound healing by growth factors and cytokines. Physiol Rev. (2003) 83:835–70. doi: 10.1152/physrev.2003.83.3.835
122. Barrientos S, Stojadinovic O, Golinko MS, Brem H, and Tomic-Canic M. Growth factors and cytokines in wound healing. Wound Repair Regener. (2008) 16:585–601. doi: 10.1111/j.1524-475X.2008.00410.x
123. Minutti CM, Modak RV, Macdonald F, Li F, Smyth DJ, Dorward DA, et al. A macrophage-pericyte axis directs tissue restoration via amphiregulin-induced transforming growth factor beta activation. Immunity. (2019) 50:645–54.e6. doi: 10.1016/j.immuni.2019.01.008
124. Dial CF, Tune MK, Doerschuk CM, and Mock JR. Foxp3(+) regulatory T cell expression of keratinocyte growth factor enhances lung epithelial proliferation. Am J Respir Cell Mol Biol. (2017) 57:162–73. doi: 10.1165/rcmb.2017-0019OC
125. Song X, Dai D, He X, Zhu S, Yao Y, Gao H, et al. Growth factor fgf2 cooperates with interleukin-17 to repair intestinal epithelial damage. Immunity. (2015) 43:488–501. doi: 10.1016/j.immuni.2015.06.024
126. Nielsen MM, Witherden DA, and Havran WL. Γδ T cells in homeostasis and host defence of epithelial barrier tissues. Nat Rev Immunol. (2017) 17:733–45. doi: 10.1038/nri.2017.101
127. Peranteau WH, Zhang L, Muvarak N, Badillo AT, Radu A, Zoltick PW, et al. Il-10 overexpression decreases inflammatory mediators and promotes regenerative healing in an adult model of scar formation. J Invest Dermatol. (2008) 128:1852–60. doi: 10.1038/sj.jid.5701232
128. Ferguson MW and O’Kane S. Scar-free healing: from embryonic mechanisms to adult therapeutic intervention. Philos Trans R Soc Lond B Biol Sci. (2004) 359:839–50. doi: 10.1098/rstb.2004.1475
129. López-Otín C and Kroemer G. Hallmarks of health. Cell. (2021) 184:33–63. doi: 10.1016/j.cell.2020.11.034
130. Basil MC, Katzen J, Engler AE, Guo M, Herriges MJ, Kathiriya JJ, et al. The cellular and physiological basis for lung repair and regeneration: past, present, and future. Cell Stem Cell. (2020) 26:482–502. doi: 10.1016/j.stem.2020.03.009
131. Kotton DN and Morrisey EE. Lung regeneration: mechanisms, applications and emerging stem cell populations. Nat Med. (2014) 20:822–32. doi: 10.1038/nm.3642
132. Konkimalla A, Tata A, and Tata PR. Lung regeneration: cells, models, and mechanisms. Cold Spring Harb Perspect Biol. (2022) 14. doi: 10.1101/cshperspect.a040873
133. Mock JR, Tune MK, Dial CF, Torres-Castillo J, Hagan RS, and Doerschuk CM. Effects of ifn-Γ on immune cell kinetics during the resolution of acute lung injury. Physiol Rep. (2020) 8:e14368. doi: 10.14814/phy2.14368
134. Jovisic M, Mambetsariev N, Singer BD, and Morales-Nebreda L. Differential roles of regulatory T cells in acute respiratory infections. J Clin Invest. (2023) 133. doi: 10.1172/jci170505
135. Mock JR, Garibaldi BT, Aggarwal NR, Jenkins J, Limjunyawong N, Singer BD, et al. Foxp3+ Regulatory T cells promote lung epithelial proliferation. Mucosal Immunol. (2014) 7:1440–51. doi: 10.1038/mi.2014.33
136. Mock JR, Dial CF, Tune MK, Gilmore RC, O’Neal WK, Dang H, et al. Impact of regulatory T cells on type 2 alveolar epithelial cell transcriptomes during resolution of acute lung injury and contributions of ifn-Γ. Am J Respir Cell Mol Biol. (2020) 63:464–77. doi: 10.1165/rcmb.2019-0399OC
137. Monticelli LA, Sonnenberg GF, Abt MC, Alenghat T, Ziegler CG, Doering TA, et al. Innate lymphoid cells promote lung-tissue homeostasis after infection with influenza virus. Nat Immunol. (2011) 12:1045–54. doi: 10.1031/ni.2131
138. Harb H, Benamar M, Lai PS, Contini P, Griffith JW, Crestani E, et al. Notch4 signaling limits regulatory T-cell-mediated tissue repair and promotes severe lung inflammation in viral infections. Immunity. (2021) 54:1186–99.e7. doi: 10.1016/j.immuni.2021.04.002
139. Kaiser KA, Loffredo LF, Santos-Alexis KL, Ringham OR, and Arpaia N. Regulation of the alveolar regenerative niche by amphiregulin-producing regulatory T cells. J Exp Med. (2023) 220. doi: 10.1084/jem.20221462
140. Zepp JA, Zacharias WJ, Frank DB, Cavanaugh CA, Zhou S, Morley MP, et al. Distinct mesenchymal lineages and niches promote epithelial self-renewal and myofibrogenesis in the lung. Cell. (2017) 170:1134–48.e10. doi: 10.1016/j.cell.2017.07.034
141. Garibaldi BT, D’Alessio FR, Mock JR, Files DC, Chau E, Eto Y, et al. Regulatory T cells reduce acute lung injury fibroproliferation by decreasing fibrocyte recruitment. Am J Respir Cell Mol Biol. (2013) 48:35–43. doi: 10.1165/rcmb.2012-0198OC
142. Ding BS, Nolan DJ, Guo P, Babazadeh AO, Cao Z, Rosenwaks Z, et al. Endothelial-derived angiocrine signals induce and sustain regenerative lung alveolarization. Cell. (2011) 147:539–53. doi: 10.1016/j.cell.2011.10.003
143. Niethamer TK, Stabler CT, Leach JP, Zepp JA, Morley MP, Babu A, et al. Defining the role of pulmonary endothelial cell heterogeneity in the response to acute lung injury. Elife. (2020) 9. doi: 10.7554/eLife.53072
144. Vila Ellis L, Cain MP, Hutchison V, Flodby P, Crandall ED, Borok Z, et al. Epithelial vegfa specifies a distinct endothelial population in the mouse lung. Dev Cell. (2020) 52:617–30.e6. doi: 10.1016/j.devcel.2020.01.009
145. D’Alessio FR, Tsushima K, Aggarwal NR, West EE, Willett MH, Britos MF, et al. Cd4+Cd25+Foxp3+ Tregs resolve experimental lung injury in mice and are present in humans with acute lung injury. J Clin Invest. (2009) 119:2898–913. doi: 10.1172/jci36498
146. Hou J, Wang X, Su C, Ma W, Zheng X, Ge X, et al. Reduced frequencies of foxp3(+)Garp(+) regulatory T cells in copd patients are associated with multi-organ loss of tissue phenotype. Respir Res. (2022) 23:176. doi: 10.1186/s12931-022-02099-2
147. Bozec A and Zaiss MM. T regulatory cells in bone remodelling. Curr Osteoporos Rep. (2017) 15:121–5. doi: 10.1007/s11914-017-0356-1
148. Kim YG, Lee CK, Nah SS, Mun SH, Yoo B, and Moon HB. Human cd4+Cd25+ Regulatory T cells inhibit the differentiation of osteoclasts from peripheral blood mononuclear cells. Biochem Biophys Res Commun. (2007) 357:1046–52. doi: 10.1016/j.bbrc.2007.04.042
149. Wei E, Hu M, Wu L, Pan X, Zhu Q, Liu H, et al. Tgf-β Signaling regulates differentiation of mscs in bone metabolism: disputes among viewpoints. Stem Cell Res Ther. (2024) 15:156. doi: 10.1186/s13287-024-03761-w
150. Li J, Xia T, Zhao Q, Wang C, Fu L, Zhao Z, et al. Biphasic calcium phosphate recruits tregs to promote bone regeneration. Acta Biomater. (2024) 176:432–44. doi: 10.1016/j.actbio.2024.01.001
151. Mougiakakos D, Jitschin R, Johansson CC, Okita R, Kiessling R, and Le Blanc K. The impact of inflammatory licensing on heme oxygenase-1-mediated induction of regulatory T cells by human mesenchymal stem cells. Blood. (2011) 117:4826–35. doi: 10.1182/blood-2010-12-324038
152. Negi N and Griffin MD. Effects of mesenchymal stromal cells on regulatory T cells: current understanding and clinical relevance. Stem Cells. (2020) 38:596–605. doi: 10.1002/stem.3151
153. Horwood NJ, Kartsogiannis V, Quinn JM, Romas E, Martin TJ, and Gillespie MT. Activated T lymphocytes support osteoclast formation in vitro. Biochem Biophys Res Commun. (1999) 265:144–50. doi: 10.1006/bbrc.1999.1623
154. Zaiss MM, Frey B, Hess A, Zwerina J, Luther J, Nimmerjahn F, et al. Regulatory T cells protect from local and systemic bone destruction in arthritis. J Immunol. (2010) 184:7238–46. doi: 10.4049/jimmunol.0903841
155. Zaiss MM, Sarter K, Hess A, Engelke K, Böhm C, Nimmerjahn F, et al. Increased bone density and resistance to ovariectomy-induced bone loss in foxp3-transgenic mice based on impaired osteoclast differentiation. Arthritis Rheum. (2010) 62:2328–38. doi: 10.1002/art.27535
156. Glowacki AJ, Yoshizawa S, Jhunjhunwala S, Vieira AE, Garlet GP, Sfeir C, et al. Prevention of inflammation-mediated bone loss in murine and canine periodontal disease via recruitment of regulatory lymphocytes. Proc Natl Acad Sci U.S.A. (2013) 110:18525–30. doi: 10.1073/pnas.1302829110
157. Liu Y, Yang R, and Shi S. Systemic infusion of mesenchymal stem cells improves cell-based bone regeneration via upregulation of regulatory T cells. Tissue Eng Part A. (2015) 21:498–509. doi: 10.1089/ten.TEA.2013.0673
158. Liu Y, Wang L, Kikuiri T, Akiyama K, Chen C, Xu X, et al. Mesenchymal stem cell-based tissue regeneration is governed by recipient T lymphocytes via ifn-Γ and tnf-α. Nat Med. (2011) 17:1594–601. doi: 10.1038/nm.2542
159. Koyama Y and Brenner DA. Liver inflammation and fibrosis. J Clin Invest. (2017) 127:55–64. doi: 10.1172/jci88881
160. Sun XF, Gu L, Deng WS, and Xu Q. Impaired balance of T helper 17/T regulatory cells in carbon tetrachloride-induced liver fibrosis in mice. World J Gastroenterol. (2014) 20:2062–70. doi: 10.3748/wjg.v20.i8.2062
161. Roh YS, Park S, Lim CW, and Kim B. Depletion of foxp3+ Regulatory T cells promotes profibrogenic milieu of cholestasis-induced liver injury. Dig Dis Sci. (2015) 60:2009–18. doi: 10.1007/s10620-014-3438-2
162. He B, Wu L, Xie W, Shao Y, Jiang J, Zhao Z, et al. The imbalance of th17/treg cells is involved in the progression of nonalcoholic fatty liver disease in mice. BMC Immunol. (2017) 18:33. doi: 10.1186/s12865-017-0215-y
163. Cairoli V, De Matteo E, Rios D, Lezama C, Galoppo M, Casciato P, et al. Hepatic lymphocytes involved in the pathogenesis of pediatric and adult non-alcoholic fatty liver disease. Sci Rep. (2021) 11:5129. doi: 10.1038/s41598-021-84674-z
164. Rau M, Schilling AK, Meertens J, Hering I, Weiss J, Jurowich C, et al. Progression from nonalcoholic fatty liver to nonalcoholic steatohepatitis is marked by a higher frequency of th17 cells in the liver and an increased th17/resting regulatory T cell ratio in peripheral blood and in the liver. J Immunol. (2016) 196:97–105. doi: 10.4049/jimmunol.1501175
165. Almeida J, Polvorosa MA, Gonzalez-Quintela A, Marcos M, Pastor I, Hernandez Cerceño ML, et al. Decreased peripheral blood cd4+/cd25+ Regulatory T cells in patients with alcoholic hepatitis. Alcohol Clin Exp Res. (2013) 37:1361–9. doi: 10.1111/acer.12095
166. Czaja AJ. Hepatic inflammation and progressive liver fibrosis in chronic liver disease. World J Gastroenterol. (2014) 20:2515–32. doi: 10.3748/wjg.v20.i10.2515
167. Zhang X, Feng M, Liu X, Bai L, Kong M, Chen Y, et al. Persistence of cirrhosis is maintained by intrahepatic regulatory T cells that inhibit fibrosis resolution by regulating the balance of tissue inhibitors of metalloproteinases and matrix metalloproteinases. Transl Res. (2016) 169:67–79. doi: 10.1016/j.trsl.2015.10.008
168. Ajith A, Merimi M, Arki MK, Hossein-Khannazer N, Najar M, Vosough M, et al. Immune regulation and therapeutic application of T regulatory cells in liver diseases. Front Immunol. (2024) 15:1371089. doi: 10.3389/fimmu.2024.1371089
169. Lu L, Feng M, Gu J, Xia Z, Zhang H, Zheng S, et al. Restoration of intrahepatic regulatory T cells through mmp-9/13-dependent activation of tgf-β Is critical for immune homeostasis following acute liver injury. J Mol Cell Biol. (2013) 5:369–79. doi: 10.1093/jmcb/mjt042
170. Zhang X, Lou J, Bai L, Chen Y, Zheng S, and Duan Z. Immune regulation of intrahepatic regulatory T cells in fibrotic livers of mice. Med Sci Monit. (2017) 23:1009–16. doi: 10.12659/msm.899725
171. Savage TM, Fortson KT, de Los Santos-Alexis K, Oliveras-Alsina A, Rouanne M, Rae SS, et al. Amphiregulin from regulatory T cells promotes liver fibrosis and insulin resistance in non-alcoholic steatohepatitis. Immunity. (2024) 57:303–18.e6. doi: 10.1016/j.immuni.2024.01.009
172. Wu KJ, Qian QF, Zhou JR, Sun DL, Duan YF, Zhu X, et al. Regulatory T cells (Tregs) in liver fibrosis. Cell Death Discov. (2023) 9:53. doi: 10.1038/s41420-023-01347-8
173. Cebi M and Yilmaz Y. Immune system dysregulation in the pathogenesis of non-alcoholic steatohepatitis: unveiling the critical role of T and B lymphocytes. Front Immunol. (2024) 15:1445634. doi: 10.3389/fimmu.2024.1445634
174. Su Q, Kim SY, Adewale F, Zhou Y, Aldler C, Ni M, et al. Single-cell rna transcriptome landscape of hepatocytes and non-parenchymal cells in healthy and nafld mouse liver. iScience. (2021) 24:103233. doi: 10.1016/j.isci.2021.103233
175. Watson BR, Paul B, Rahman RU, Amir-Zilberstein L, Segerstolpe Å, Epstein ET, et al. Spatial transcriptomics of healthy and fibrotic human liver at single-cell resolution. Nat Commun. (2025) 16:319. doi: 10.1038/s41467-024-55325-4
176. Gandolfo MT, Jang HR, Bagnasco SM, Ko GJ, Agreda P, Satpute SR, et al. Foxp3+ Regulatory T cells participate in repair of ischemic acute kidney injury. Kidney Int. (2009) 76:717–29. doi: 10.1038/ki.2009.259
177. Jun C, Qingshu L, Ke W, Ping L, Jun D, Jie L, et al. Protective effect of cxcr3+Cd4+Cd25+Foxp3+ Regulatory T cells in renal ischemia-reperfusion injury. Mediators Inflammation. (2015) 2015:360973. doi: 10.1155/2015/360973
178. Kurawaki S, Nakashima A, Ishiuchi N, Kanai R, Maeda S, Sasaki K, et al. Mesenchymal stem cells pretreated with interferon-gamma attenuate renal fibrosis by enhancing regulatory T cell induction. Sci Rep. (2024) 14:10251. doi: 10.1038/s41598-024-60928-4
179. Hu M, Wang C, Zhang GY, Saito M, Wang YM, Fernandez MA, et al. Infiltrating foxp3(+) regulatory T cells from spontaneously tolerant kidney allografts demonstrate donor-specific tolerance. Am J Transplant. (2013) 13:2819–30. doi: 10.1111/ajt.12445
180. Kinsey GR, Sharma R, Huang L, Li L, Vergis AL, Ye H, et al. Regulatory T cells suppress innate immunity in kidney ischemia-reperfusion injury. J Am Soc Nephrol. (2009) 20:1744–53. doi: 10.1681/asn.2008111160
181. Dousdampanis P, Trigka K, and Mouzaki A. Tregs and kidney: from diabetic nephropathy to renal transplantation. World J Transplant. (2016) 6:556–63. doi: 10.5500/wjt.v6.i3.556
182. do Valle Duraes F, Lafont A, Beibel M, Martin K, Darribat K, Cuttat R, et al. Immune cell landscaping reveals a protective role for regulatory T cells during kidney injury and fibrosis. JCI Insight. (2020) 5. doi: 10.1172/jci.insight.130651
183. Li XC and Turka LA. An update on regulatory T cells in transplant tolerance and rejection. Nat Rev Nephrol. (2010) 6:577–83. doi: 10.1038/nrneph.2010.101
184. Kinsey GR, Huang L, Jaworska K, Khutsishvili K, Becker DA, Ye H, et al. Autocrine adenosine signaling promotes regulatory T cell-mediated renal protection. J Am Soc Nephrol. (2012) 23:1528–37. doi: 10.1681/asn.2012010070
185. Kinsey G, Huang L, Khutsishvili K, Ye H, Lobo P, and Okusa M. Protective action of regulatory T cells in ischemic kidney injury requires adenosine 2a receptor signaling and pd-1 (178.15). J Immunol. (2012) 188:178.15–.15. doi: 10.4049/jimmunol.188.Supp.178.15
186. Xian W, Wu J, Li Q, Du X, Wang N, Chen D, et al. Cxcr3 alleviates renal ischemia−Reperfusion injury via increase of tregs. Mol Med Rep. (2021) 24. doi: 10.3892/mmr.2021.12180
187. De Simone G, Mazza EMC, Cassotta A, Davydov AN, Kuka M, Zanon V, et al. Cxcr3 identifies human naive cd8(+) T cells with enhanced effector differentiation potential. J Immunol. (2019) 203:3179–89. doi: 10.4049/jimmunol.1901072
188. Zhang P, Wang J, Miao J, and Zhu P. The dual role of tissue regulatory T cells in tissue repair: return to homeostasis or fibrosis. Front Immunol. (2025) 16:1560578. doi: 10.3389/fimmu.2025.1560578
189. Izcue A, Coombes JL, and Powrie F. Regulatory T cells suppress systemic and mucosal immune activation to control intestinal inflammation. Immunol Rev. (2006) 212:256–71. doi: 10.1111/j.0105-2896.2006.00423.x
190. Figliuolo da Paz VR, Jamwal DR, and Kiela PR. Intestinal regulatory T cells. Adv Exp Med Biol. (2021) 1278:141–90. doi: 10.1007/978-981-15-6407-9_9
191. Cosovanu C and Neumann C. The many functions of foxp3(+) regulatory T cells in the intestine. Front Immunol. (2020) 11:600973. doi: 10.3389/fimmu.2020.600973
192. Biton M, Haber AL, Rogel N, Burgin G, Beyaz S, Schnell A, et al. T helper cell cytokines modulate intestinal stem cell renewal and differentiation. Cell. (2018) 175:1307–20.e22. doi: 10.1016/j.cell.2018.10.008
193. Wohlfert EA, Grainger JR, Bouladoux N, Konkel JE, Oldenhove G, Ribeiro CH, et al. Gata3 controls foxp3+ Regulatory T cell fate during inflammation in mice. J Clin Invest. (2011) 121:4503–15. doi: 10.1172/jci57456
194. Sefik E, Geva-Zatorsky N, Oh S, Konnikova L, Zemmour D, McGuire AM, et al. Mucosal immunology. Individual intestinal symbionts induce a distinct population of rorγ+ Regulatory T cells. Science. (2015) 349:993–7. doi: 10.1126/science.aaa9420
195. Ohnmacht C, Park JH, Cording S, Wing JB, Atarashi K, Obata Y, et al. Mucosal immunology. The microbiota regulates type 2 immunity through rorγt+ T cells. Science. (2015) 349:989–93. doi: 10.1126/science.aac4263
196. Jiang Z and Wu C. Reciprocal interactions between regulatory T cells and intestinal epithelial cells. Front Immunol. (2022) 13:951339. doi: 10.3389/fimmu.2022.951339
197. Monticelli LA, Osborne LC, Noti M, Tran SV, Zaiss DM, and Artis D. Il-33 promotes an innate immune pathway of intestinal tissue protection dependent on amphiregulin-egfr interactions. Proc Natl Acad Sci U.S.A. (2015) 112:10762–7. doi: 10.1073/pnas.1509070112
198. Shao J and Sheng H. Amphiregulin promotes intestinal epithelial regeneration: roles of intestinal subepithelial myofibroblasts. Endocrinology. (2010) 151:3728–37. doi: 10.1210/en.2010-0319
199. Wang L, Wang S, Lin J, Li J, Wang M, Yu J, et al. Treg and intestinal myofibroblasts-derived amphiregulin induced by tgf-β Mediates intestinal fibrosis in crohn’s disease. J Transl Med. (2025) 23:452. doi: 10.1186/s12967-025-06413-6
200. Yang G, Geng XR, Song JP, Wu Y, Yan H, Zhan Z, et al. Insulin-like growth factor 2 enhances regulatory T-cell functions and suppresses food allergy in an experimental model. J Allergy Clin Immunol. (2014) 133:1702–8.e5. doi: 10.1016/j.jaci.2014.02.019
201. Sun B, Liu M, Cui M, and Li T. Granzyme B-expressing treg cells are enriched in colorectal cancer and present the potential to eliminate autologous T conventional cells. Immunol Lett. (2020) 217:7–14. doi: 10.1016/j.imlet.2019.10.007
202. Osman A, Yan B, Li Y, Pavelko KD, Quandt J, Saadalla A, et al. Tcf-1 controls T(Reg) cell functions that regulate inflammation, cd8(+) T cell cytotoxicity and severity of colon cancer. Nat Immunol. (2021) 22:1152–62. doi: 10.1038/s41590-021-00987-1
203. Park JY, Chung H, DiPalma DT, Tai X, and Park JH. Immune quiescence in the oral mucosa is maintained by a uniquely large population of highly activated foxp3(+) regulatory T cells. Mucosal Immunol. (2018) 11:1092–102. doi: 10.1038/s41385-018-0027-2
204. Xue N, Wang Y, Cheng H, Liang H, Fan X, Zuo F, et al. Regulatory T cell therapy suppresses inflammation of oral mucosa. Front Immunol. (2022) 13:1009742. doi: 10.3389/fimmu.2022.1009742
205. Pandiyan P, Bhaskaran N, Zou M, Schneider E, Jayaraman S, and Huehn J. Microbiome dependent regulation of T(Regs) and th17 cells in mucosa. Front Immunol. (2019) 10:426. doi: 10.3389/fimmu.2019.00426
206. Bhaskaran N, Quigley C, Paw C, Butala S, Schneider E, and Pandiyan P. Role of short chain fatty acids in controlling T(Regs) and immunopathology during mucosal infection. Front Microbiol. (2018) 9:1995. doi: 10.3389/fmicb.2018.01995
207. Bergmeier LA, Dutzan N, Smith PC, and Kraan H. Editorial: immunology of the oral mucosa. Front Immunol. (2022) 13:877209. doi: 10.3389/fimmu.2022.877209
208. Tao XA, Xia J, Chen XB, Wang H, Dai YH, Rhodus NL, et al. Foxp3 T regulatory cells in lesions of oral lichen planus correlated with disease activity. Oral Dis. (2010) 16:76–82. doi: 10.1111/j.1601-0825.2009.01608.x
209. Zhou L, Cao T, Wang Y, Yao H, Du G, Chen G, et al. Frequently increased but functionally impaired cd4+Cd25+ Regulatory T cells in patients with oral lichen planus. Inflammation. (2016) 39:1205–15. doi: 10.1007/s10753-016-0356-9
210. Sanchez-Solares J, Sanchez L, Pablo-Torres C, Diaz-Fernandez C, Sørensen P, Barber D, et al. Celiac disease causes epithelial disruption and regulatory T cell recruitment in the oral mucosa. Front Immunol. (2021) 12:623805. doi: 10.3389/fimmu.2021.623805
211. Aluvihare VR, Kallikourdis M, and Betz AG. Regulatory T cells mediate maternal tolerance to the fetus. Nat Immunol. (2004) 5:266–71. doi: 10.1038/ni1037
212. Quinn KH, Lacoursiere DY, Cui L, Bui J, and Parast MM. The unique pathophysiology of early-onset severe preeclampsia: role of decidual T regulatory cells. J Reprod Immunol. (2011) 91:76–82. doi: 10.1016/j.jri.2011.05.006
213. Santner-Nanan B, Peek MJ, Khanam R, Richarts L, Zhu E, Fazekas de St Groth B, et al. Systemic increase in the ratio between foxp3+ and il-17-producing cd4+ T cells in healthy pregnancy but not in preeclampsia. J Immunol. (2009) 183:7023–30. doi: 10.4049/jimmunol.0901154
214. Sasaki Y, Darmochwal-Kolarz D, Suzuki D, Sakai M, Ito M, Shima T, et al. Proportion of peripheral blood and decidual cd4(+) cd25(Bright) regulatory T cells in pre-eclampsia. Clin Exp Immunol. (2007) 149:139–45. doi: 10.1111/j.1365-2249.2007.03397.x
215. Guerin LR, Moldenhauer LM, Prins JR, Bromfield JJ, Hayball JD, and Robertson SA. Seminal fluid regulates accumulation of foxp3+ Regulatory T cells in the preimplantation mouse uterus through expanding the foxp3+ Cell pool and ccl19-mediated recruitment. Biol Reprod. (2011) 85:397–408. doi: 10.1095/biolreprod.110.088591
216. Moldenhauer LM, Diener KR, Thring DM, Brown MP, Hayball JD, and Robertson SA. Cross-presentation of male seminal fluid antigens elicits T cell activation to initiate the female immune response to pregnancy. J Immunol. (2009) 182:8080–93. doi: 10.4049/jimmunol.0804018
217. Robertson SA, Guerin LR, Bromfield JJ, Branson KM, Ahlström AC, and Care AS. Seminal fluid drives expansion of the cd4+Cd25+ T regulatory cell pool and induces tolerance to paternal alloantigens in mice. Biol Reprod. (2009) 80:1036–45. doi: 10.1095/biolreprod.108.074658
218. Care AS, Bourque SL, Morton JS, Hjartarson EP, Robertson SA, and Davidge ST. Reduction in regulatory T cells in early pregnancy causes uterine artery dysfunction in mice. Hypertension. (2018) 72:177–87. doi: 10.1161/hypertensionaha.118.10858
219. Vojtech L, Zhang M, Davé V, Levy C, Hughes SM, Wang R, et al. Extracellular vesicles in human semen modulate antigen-presenting cell function and decrease downstream antiviral T cell responses. PloS One. (2019) 14:e0223901. doi: 10.1371/journal.pone.0223901
220. Wira CR and Fahey JV. A new strategy to understand how hiv infects women: identification of a window of vulnerability during the menstrual cycle. Aids. (2008) 22:1909–17. doi: 10.1097/QAD.0b013e3283060ea4
221. Wira CR, Fahey JV, Ghosh M, Patel MV, Hickey DK, and Ochiel DO. Sex hormone regulation of innate immunity in the female reproductive tract: the role of epithelial cells in balancing reproductive potential with protection against sexually transmitted pathogens. Am J Reprod Immunol. (2010) 63:544–65. doi: 10.1111/j.1600-0897.2010.00842.x
222. Wira CR, Fahey JV, Rodriguez-Garcia M, Shen Z, and Patel MV. Regulation of mucosal immunity in the female reproductive tract: the role of sex hormones in immune protection against sexually transmitted pathogens. Am J Reprod Immunol. (2014) 72:236–58. doi: 10.1111/aji.12252
223. Hughes SM, Pandey U, Johnston C, Marrazzo J, Hladik F, and Micks E. Impact of the menstrual cycle and ethinyl estradiol/etonogestrel contraceptive vaginal ring on granulysin and other mucosal immune mediators. Am J Reprod Immunol. (2021) 86:e13412. doi: 10.1111/aji.13412
224. Robertson SA, Green ES, Care AS, Moldenhauer LM, Prins JR, Hull ML, et al. Therapeutic potential of regulatory T cells in preeclampsia-opportunities and challenges. Front Immunol. (2019) 10:478. doi: 10.3389/fimmu.2019.00478
225. Ma B, Forney LJ, and Ravel J. Vaginal microbiome: rethinking health and disease. Annu Rev Microbiol. (2012) 66:371–89. doi: 10.1146/annurev-micro-092611-150157
226. Campisciano G, Zanotta N, Licastro D, De Seta F, and Comar M. In vivo microbiome and associated immune markers: new insights into the pathogenesis of vaginal dysbiosis. Sci Rep. (2018) 8:2307. doi: 10.1038/s41598-018-20649-x
227. Eslami S, Hadjati J, Motevaseli E, Mirzaei R, Farashi Bonab S, Ansaripour B, et al. Lactobacillus crispatus strain sj-3c-us induces human dendritic cells (Dcs) maturation and confers an anti-inflammatory phenotype to dcs. Apmis. (2016) 124:697–710. doi: 10.1111/apm.12556
228. Schellenberg JJ, Card CM, Ball TB, Mungai JN, Irungu E, Kimani J, et al. Bacterial vaginosis, hiv serostatus and T-cell subset distribution in a cohort of east african commercial sex workers: retrospective analysis. Aids. (2012) 26:387–93. doi: 10.1097/QAD.0b013e32834ed7f0
Keywords: regulatory T cells, tissue, injury, repair, regeneration
Citation: Raheem A, Khan I, Ahmad I, Wajid A, Alshahrani MY, Alzahrani FM, Alzahrani KJ, Qadeer A, Liao I-C and Chen C-C (2025) The emerging role of tissue regulatory T cells in tissue repair and regeneration. Front. Immunol. 16:1640113. doi: 10.3389/fimmu.2025.1640113
Received: 03 June 2025; Accepted: 15 August 2025;
Published: 29 August 2025.
Edited by:
Karina Pino-Lagos, University of the Andes, ChileReviewed by:
Carolina Isabel Rojas, University of the Andes, ChileAquib Ehtram, La Jolla Institute for Immunology (LJI), United States
Copyright © 2025 Raheem, Khan, Ahmad, Wajid, Alshahrani, Alzahrani, Alzahrani, Qadeer, Liao and Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Abdul Qadeer, cWFkZWVya3RrODQ4QHlhaG9vLmNvbQ==; I-Chuang Liao, aWNodWFuZ2xpYW9AZ21haWwuY29t; Chien-Chin Chen, aGxtYXJrY0BnbWFpbC5jb20=
 Abdul Raheem
Abdul Raheem Ilyas Khan
Ilyas Khan Iqbal Ahmad
Iqbal Ahmad Abdul Wajid4
Abdul Wajid4 Khalid J. Alzahrani
Khalid J. Alzahrani Abdul Qadeer
Abdul Qadeer I-Chuang Liao
I-Chuang Liao Chien-Chin Chen
Chien-Chin Chen