- Department of Biological Science, Center for Applied Biotechnology Studies, College of Natural Sciences and Mathematics, California State University Fullerton, Fullerton, CA, USA
Enzymatic modification is a prevalent mechanism by which bacteria defeat the action of antibiotics. Aminoglycosides are often inactivated by aminoglycoside modifying enzymes encoded by genes present in the chromosome, plasmids, and other genetic elements. The AAC(6′)-Ib (aminoglycoside 6′-N-acetyltransferase type Ib) is an enzyme of clinical importance found in a wide variety of gram-negative pathogens. The AAC(6′)-Ib enzyme is of interest not only because of his ubiquity but also because of other characteristics, it presents significant microheterogeneity at the N-termini and the aac(6′)-Ib gene is often present in integrons, transposons, plasmids, genomic islands, and other genetic structures. Excluding the highly heterogeneous N-termini, there are 45 non-identical AAC(6′)-Ib related entries in the NCBI database, 32 of which have identical name in spite of not having identical amino acid sequence. While some variants conserved similar properties, others show dramatic differences in specificity, including the case of AAC(6′)-Ib-cr that mediates acetylation of ciprofloxacin representing a rare case where a resistance enzyme acquires the ability to utilize an antibiotic of a different class as substrate. Efforts to utilize antisense technologies to turn off expression of the gene or to identify enzymatic inhibitors to induce phenotypic conversion to susceptibility are under way.
Aminoglycosides and Resistance
Aminoglycosides are bactericidal antibiotics that affect translation fidelity and, according to recent data, they may also stimulate the production of highly deleterious hydroxyl radicals (Vakulenko and Mobashery, 2003; Magnet and Blanchard, 2005; Jana and Deb, 2006; Kohanski et al., 2007; Majumder et al., 2007). Aminoglycosides are used to treat infections caused by gram-negative bacilli and, in combination with β-lactams or vancomycin, to treat some gram-positive pathogens, mainly staphylococci (Yao and Moellering, 2007). Since a step in the uptake process requires functional respiration, the spectrum of action of aminoglycosides is limited to aerobic bacteria (Muir et al., 1984). In addition to their most common uses, aminoglycosides can be utilized to treat diseases such as tuberculosis (Menzies et al., 2009; Brossier et al., 2010), plague, tularemia, brucellosis, endocarditis caused by enterococci, and others (Vakulenko and Mobashery, 2003; Yao and Moellering, 2007; Ramirez and Tolmasky, 2010). The fact that aminoglycosides also cause a decrease in eukaryotic translational fidelity permitted to initiate efforts to developed them as drugs to treat nonsense mutation related genetic disorders such as cystic fibrosis and Duchenne muscular dystrophy (Rich et al., 1990; Kellermayer, 2006; Hermann, 2007; Kondo et al., 2007; Zingman et al., 2007; Bidou et al., 2012; Kandasamy et al., 2012). A chemical labyrinthectomy using intratympanic injection of aminoglycosides is used when most treatments of Ménière's disease fail (Huon et al., 2012; Pacheu-Grau et al., 2012). Aminoglycoside-based drugs are also inhibitors of reproduction of the HIV virus, a property that could result in their utilization in the treatment of AIDS patients (Houghton et al., 2010).
The basic chemical structure of aminoglycosides is characterized by the presence of an aminocyclitol nucleus (streptamine, 2-deoxystreptamine, or streptidine) linked to amino sugars through glycosidic bonds. However, other compounds with different basic structures are also included within the aminoglycosides family, e.g., spectinomycin, an aminocyclitol not linked to amino sugars or compounds containing the aminocyclitol fortamine (Bryskier, 2005). They reach the cytoplasm of the bacterial cell in a three-step process, of which the first one is energy-independent and the following two are energy-dependent (Taber et al., 1987; Vakulenko and Mobashery, 2003; Ramirez and Tolmasky, 2010). At the molecular level, the action of aminoglycosides is characterized by interactions between the antibiotic molecule and the 16S rRNA. Although for all aminoglycosides the effect of this interaction is a change of conformation of the decoding A site producing one that resembles the closed state induced by interaction between cognate tRNA and mRNA, it must be noted that not all aminoglycosides seem to bind the same sites of the 16S rRNA. The consequence of the conformational change induced by the interaction 16S rRNA-aminoglycoside is the reduction of the proofreading capabilities of the ribosome, which in turns results in high levels of mistranslation (Bakker, 1992; Busse et al., 1992; Vakulenko and Mobashery, 2003; Vicens and Westhof, 2003; Magnet and Blanchard, 2005; Majumder et al., 2007; Zaher and Green, 2009; Ramirez and Tolmasky, 2010). Other molecular effects of some aminoglycosides have been described but it is not clear if some of them are not secondary to protein mistranslation. They include inhibition of 30S ribosomal subunit assembly, induction of RNA cleavage, or interference with the action of RNase P (Mikkelsen et al., 1999; Mehta and Champney, 2003; Belousoff et al., 2009).
Aminoglycosides are powerful tools against infections (Labaune et al., 2001; Avent et al., 2011) but unfortunately the levels of resistance are growing and in consequence failure of treatments with aminoglycosides is becoming more common (Galani et al., 2002; van ‘t Veen et al., 2005; Tolmasky, 2007a; Soler Bistue et al., 2008). Bacteria have developed numerous mechanisms to resist the action of aminoglycosides and cells can possess the genetic determinants for several of them enhancing the levels of resistance and making it very difficult to overcome all of them. Enzymatic inactivation by acetylation, adenylylation, or phosphorylation at different locations of the aminoglycoside molecule is among the most clinically relevant strategies bacteria use to resist the action of these antibiotics (Shaw et al., 1993; Vakulenko and Mobashery, 2003; Tolmasky, 2007a; Ramirez and Tolmasky, 2010; Chen et al., 2011; Chiang et al., 2013). The enzymes that catalyze these reactions are collectively known as aminoglycoside modifying enzymes. Other well studied mechanisms are: (1) mutation of the 16S rRNA or ribosomal proteins modify the target eliminating or reducing the interaction with the antibiotic molecule (O'Connor et al., 1991); (2) methylation of 16S rRNA, a mechanism found in most aminoglycoside-producing organisms and in clinical strains (Schmitt et al., 2009; Wachino and Arakawa, 2012); (3) reduced permeability to the antibiotic molecule by modification of the permeability of the outer membrane or diminished inner membrane transport (Hancock, 1981; Taber et al., 1987; Macleod et al., 2000; Over et al., 2001); (4) export outside the cell by active efflux pumps (Hocquet et al., 2003; Morita et al., 2012; Wachino and Arakawa, 2012); (5) sequestration by tight binding to a low active aminoglycoside acetyltransferase (Magnet et al., 2003); and (6) extracellular DNA shielding in biofilms (Chiang et al., 2013).
The general characteristics of all known aminoglycoside modifying enzymes have been recently reviewed (Ramirez and Tolmasky, 2010). This review will focus on the aminoglycoside 6′-N-acetyltransferase type Ib [AAC(6′)-Ib], which is of great clinical relevance and it is found in over 70% of AAC(6′)-I-producing gram-negative clinical isolates (Vakulenko and Mobashery, 2003), and has been the subject of numerous studies (Tolmasky, 2007a; Cambray and Mazel, 2008; Ramirez and Tolmasky, 2010).
The AAC(6′)-Ib Protein
The aminoglycoside N-acetyltransferases (AAC) belong to the GCN5-related N-acetyltransferase superfamily, also known as GNAT. This is a large group of enzymes that includes about 10,000 proteins from all kinds of organisms that share the property to catalyze the acetylation of a primary amine in numerous acceptor molecules using acetyl CoA as donor substrate (Neuwald and Landsman, 1997; Dyda et al., 2000; Vetting et al., 2005). The AACs are subdivided in groups based on the position where the acetyl group is transferred in the acceptor aminoglycoside molecule. Known AACs catalyze acetylation at the 1 [AAC(1)], 3 [AAC(3)], 2′ [AAC(2′)], or 6′ [AAC(6′)] positions (Shaw et al., 1993; De Pascale and Wright, 2010; Ramirez and Tolmasky, 2010). AAC(6′) enzymes are the most numerous group of AACs, more than 40 have been described, and can be found in gram-negatives as well as gram-positives (Shaw et al., 1993; Miller et al., 1997; Wright, 1999; Tolmasky, 2007a; Ramirez and Tolmasky, 2010). AAC(6′) enzymes are subdivided in two groups, AAC(6′)-I and AAC(6′)-II, which are differentiated by the profile of the aminoglycosides inactivated. With a few exceptions, AAC(6′)-I enzymes specify resistance to several aminoglycosides plus amikacin and gentamicin C1a and C2 but not to gentamicin C1 (Shaw et al., 1993). On the other hand, AAC(6′)-II enzymes catalyze acetylation of all forms of gentamicin but not of amikacin (Rather et al., 1992). In addition, enzymes with extended spectrum that may merit addition of new subclasses of AAC(6′)-I enzymes have been recently described (Casin et al., 2003; Robicsek et al., 2006; Strahilevitz et al., 2009). Phylogenetic analyses divided the AAC(6′) enzymes into three clades. However, with the information available it is still not clear if all AAC(6′) enzymes evolved from a single origin or the three groups are less related and the 6′ acetylating activity has evolved independently at least three times (Salipante and Hall, 2003). According to the phylogenetic analyses recently communicated by Salipante and Hall (Salipante and Hall, 2003) the AAC(6′)-Ib is most closely related to AAC(6′)-IIa AAC(6′)-IIb, AAC(6′)-IIc, and AAC(6′)-IId.
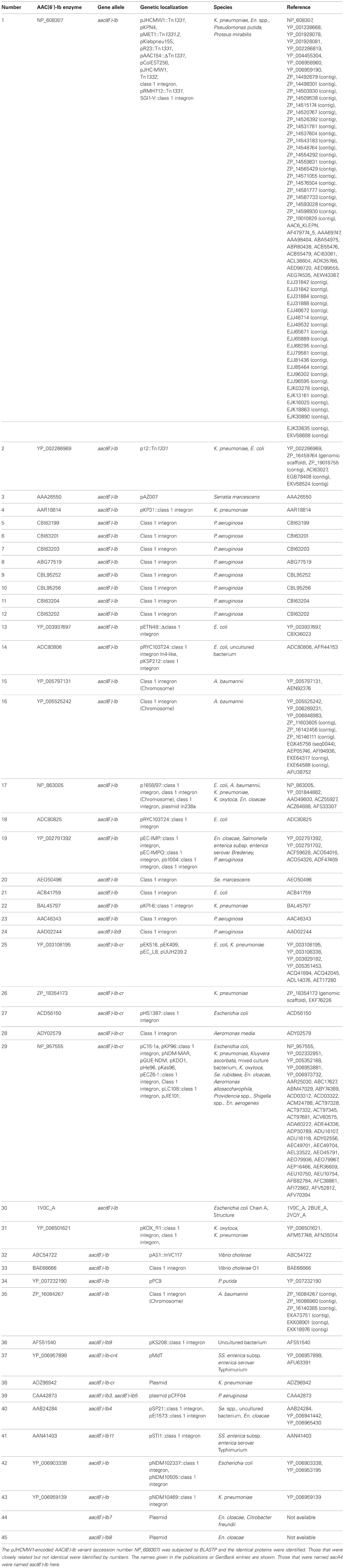
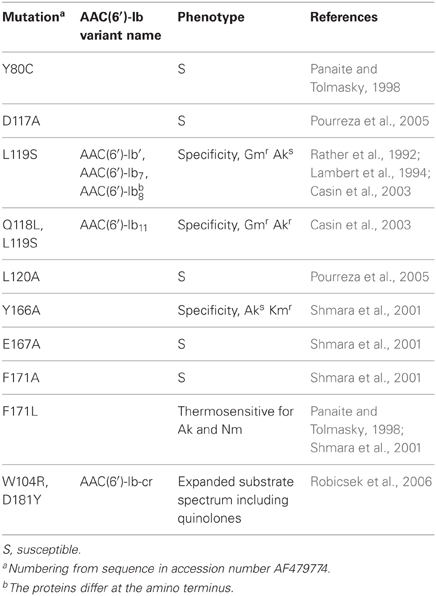
There are numerous variants of AAC(6′)-Ib, many of them identified by modifications in the name of the enzyme such as the addition of subscripts or a prime symbol superscript (Cambray and Mazel, 2008; Ramirez and Tolmasky, 2010). However, a large number of versions of the protein, or predicted protein, have all been named AAC(6′)-Ib, which can be a source of confusion or indetermination. These variants mainly differ at the N-terminus, however one should be careful when considering these differences because not in all of them the N-terminus has been experimentally determined (Dery et al., 1997; Casin et al., 2003; Soler Bistue et al., 2006; Maurice et al., 2008). Table 1 shows a list of the aac(6′)-Ib gene versions found in different genetic environments and bacterial species. Some variants differing at the N-termini such as AAC(6′)-Ib3, AAC(6′)-Ib4, AAC(6′)-Ib6, and AAC(6′)-Ib7 have been compared and it was found that they have similar behavior (Casin et al., 1998) but variations as small as one or two amino acids at key positions proved to be of high relevance (Table 2). For example, the AAC(6′)-Ib11 found in S. Typhimurium has L and S residues at positions 118 and 119 as opposed to Q and L or Q and S, the amino acids present at these positions in all previously described enzymes, acquired an extended resistance spectrum that includes all three gentamicin forms (Casin et al., 2003). (Amino acid numbers throughout the text are based on the sequence corresponding to accession number AF479774.) Another example worth mentioning is the AAC(6′)-Ib', originally found in Pseudomonas fluorescens BM2687, but previously generated by site-directed mutagenesis in the laboratory (Table 1). This protein has a L to S substitution at amino acid 119 that confers the enzyme an AAC(6′)-II profile, i.e., the enzyme confers resistance to gentamicin but not amikacin (Rather et al., 1992; Lambert et al., 1994). A highly surprising effect occurred in the natural variant known as AAC(6′)-Ib-cr, which has the modifications W104R and N181Y (Tables 1, 2). The substrate spectrum was expanded to include quinolone antibiotics, crossing the barrier from the aminoglycosides (Robicsek et al., 2006). Since the first detection of the AAC(6′)-Ib-cr variant there have been numerous reports of its presence, and variants of it, across the world in different genetic environments suggesting an extraordinary ability to disseminate (Quiroga et al., 2007; Cattoir and Nordmann, 2009; Strahilevitz et al., 2009; Rodriguez-Martinez et al., 2011; Ruiz et al., 2012; De Toro et al., 2013). Furthermore, there have been cases where a strain was found to simultaneously include genes coding for AAC(6′)-Ib and AAC(6′)-Ib-cr (Kim et al., 2011). AAC(6′)-Ib is also found fused to the C-terminal end of AAC(3)-Ib protein within a class I integron found in a Pseudomonas aeruginosa strain (Dubois et al., 2002) and to the C-terminus of the AAC(6′)-30 also within a P. aeruginosa class I integron (Mendes et al., 2004).
Subcellular localization studies of the AAC(6′)-Ib enzyme encoded by Tn1331 showed that the enzyme is homogeneously distributed in the cytoplasmic compartment (Dery et al., 2003). AAC(6′)-Ib was one of three aminoglycoside modifying enzymes used in a study consisting of molecular dynamics simulations of the enzymes and aminoglycoside ribosomal RNA binding site, unliganded, and complexed with an aminoglycoside, kanamycin A. These studies concluded that the enzymes efficiently mimic the nucleic acid environment of the ribosomal RNA binding cleft (Romanowska et al., 2013). Extensive studies using mutagenesis showed some interesting phenotypes such as modifications in specificity, enhanced activity, or selective thermosensitivity (Table 2) (Panaite and Tolmasky, 1998; Chavideh et al., 1999; Shmara et al., 2001; Casin et al., 2003; Pourreza et al., 2005; Kim et al., 2007; Maurice et al., 2008). In addition, alanine scanning showed that several amino acid substitutions by A had minor effects. These mutagenesis studies together with structural and enzymatic analyses led to a deep understanding of features and characteristics of AAC(6′)-Ib proteins (Rather et al., 1992; Vetting et al., 2004; Maurice et al., 2008; Vetting et al., 2008; Ramirez and Tolmasky, 2010). The three dimensional structure of AAC(6′)-Ib and AAC(6′)-Ib11 have been experimentally determined in various conditions. AAC(6′)-Ib was crystallized in complex with coenzyme A and also in complex with both coenzyme A and kanamycin. The structures were solved to 1.8 Å and 2.4 Å resolution, respectively (Maurice et al., 2008). The broad spectrum variant AAC(6′)-Ib11 was crystallized in the absence of substrate and the structure was solved to 2.1 Å resolution (Maurice et al., 2008). These studies concluded that AAC(6′)-Ib exists as a monomer while AAC(6′)-Ib11 shows monomer/dimer equilibrium (Maurice et al., 2008). This was a somewhat surprising finding considering that previous studies had shown that two other acetyltransferases, AAC(6′)-Ii and AAC(6′)-Iy, exist as dimers (Wright and Ladak, 1997; Wybenga-Groot et al., 1999; Draker et al., 2003; Vetting et al., 2004; Wright and Berghuis, 2007; Vong et al., 2012). Interestingly, analysis of these crystal structures showed the presence of a flexible flap in AAC(6′)-Ib11 that may be the basis for its ability to utilize amikacin as well as gentamicin as substrates (Maurice et al., 2008). In another study a molecular model of AAC(6′)-Ib-cr has been generated (Maurice et al., 2008; Vetting et al., 2008), which led to postulate that the D181Y substitution is mainly responsible for modification in the strength of binding of the antibiotic substrate and that the substitution W104R stabilizes the positioning of Y181 (Robicsek et al., 2006; Strahilevitz et al., 2009).
Table 1 shows that there are 45 non-identical AAC(6′)-Ib related entries in the NCBI database, 32 of which have identical name in spite of not having identical amino acid sequence. The N-termini of these proteins show the highest degree of heterogeneity with high variations in length stretching up to 60 amino acids, but these differences were suggested to be irrelevant (Casin et al., 1998; Maurice et al., 2008). Therefore, we defined a highly conserved central region composed of 181 amino acids shared by all proteins, which were compared using the MAFFT alignment algorithm (Katoh and Standley, 2013). Pairwise comparisons show that the sequences have 1 to 8 amino acid differences and a total of 24 positions showed amino acid variations. Moreover, clustering using the UPGMA algorithm (Sneath and Sokal, 1973) defined 18 sequence clusters, 14 of which consist of a singleton, and 4 of which include 2–16 proteins (Figure 1). Different clusters can exhibit similar properties while others show substantial differences in their characteristics such as those cases in which there are significant specificity variations like extended substrate spectrum as described in the above paragraphs.
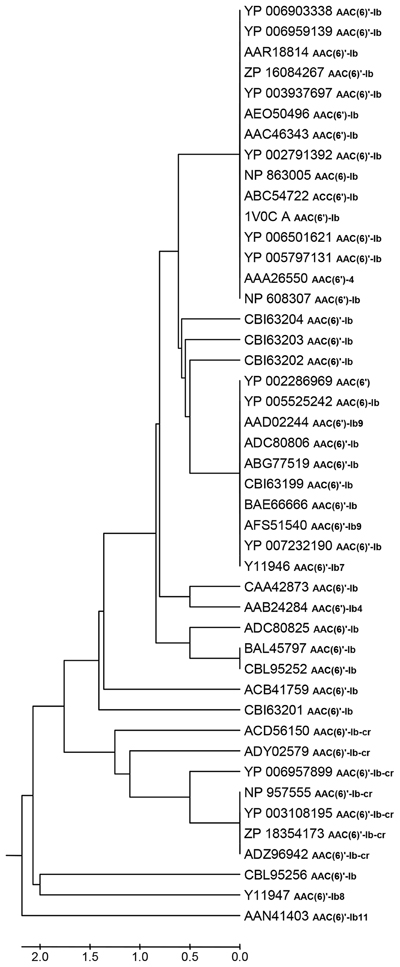
Figure 1. UPGMA clustering analyses of 45 AAC(6′)-Ib protein sequences. The optimal tree with the sum of branch length = 20.70628249 is shown. The evolutionary distances were computed using the number of differences method and are in the units of the number of amino acid differences per sequence. All positions containing gaps and missing data were eliminated. There were a total of 181 positions in the final dataset. Evolutionary analyses were conducted in MEGA5.
The aac(6′)-Ib Gene
The aac(6′)-Ib genes are usually found as fully functional or deficient gene cassettes associated to class 1 integrons, insertion sequences such as IS26, and truncated or disrupted integrons (Figure 2 and Table 1) (Sarno et al., 2002; Woodford et al., 2009; Ramirez and Tolmasky, 2010). These genetic elements may be part of plasmids, transposons, genomic islands, or other structures such as the KQ element (Rice et al., 2008), which together contribute to the gene's ability to disseminate at the cellular and molecular levels (Tolmasky, 2007b). When present in integrons, aac(6′)-Ib gene cassettes can be found located adjacent to the 5′-conserved region, i.e., flanked by attI and attC, or internal to the variable portion containing attC loci at both ends (Figure 2). In both cases, as expected, the gene cassette can be mobilized by the integrase IntI1 (Figure 2) (Cambray et al., 2010; Hall, 2012). In addition, a gene cassette-like structure containing aac(6′)-Ib, composed of a copy of attI1* at the beginning of the structural gene and a regular attC downstream of it (see Figure 2), was found as part of a region resembling the variable portion of integrons in Tn1331 (Woloj et al., 1986; Tolmasky et al., 1988; Tolmasky, 1990; Tolmasky and Crosa, 1993; Sarno et al., 2002), Tn1331.2 (Tolmasky and Crosa, 1991), Tn1332 (Poirel et al., 2006), the KQ element (Rice et al., 2008), a Tn1331 derivative recently isolated from a clinical Klebsiella pneumoniae strain belonging to the ST512, which derived from the ST258, known to be spread worldwide (Chen et al., 2012; Garcia-Fernandez et al., 2012; Warburg et al., 2012), and a complex mosaic region present in the chromosome of Proteus mirabilis JIE273 (Zong et al., 2009). Assays overexpressing IntI1 in cells containing Tn1331 were unable to detect any excision of the aac(6′)-Ib gene cassette-like structure, suggesting that it is nonfunctional or it excises at an extremely low efficiency (Ramirez et al., 2008). Interestingly, despite the IntI1-mediated lack of mobility of this DNA region, the gene could be mobilized by means of a mechanism recently proposed by Zong et al. that occurs through homologous recombination between 520-bp direct repeats located upstream and downstream of the gene cassette-like structure (Figure 2) (Zong et al., 2009).
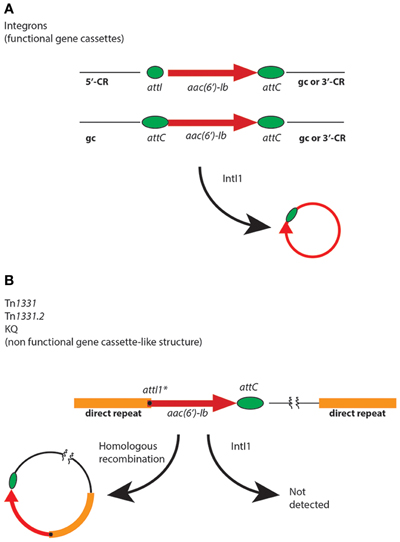
Figure 2. Mobilization of aac(6′)-Ib. (A) Generic genetic maps of integrons in which an aac(6′)-Ib gene cassette is located immediately following the 5′ conserved region (5′-CR) (top map) or following one or more gene cassettes (gc) inside the variable portion, and followed by other gene cassettes or the 3′ conserved region (3′-CR) (bottom map). The small green ellipse represents attI and the big green ellipses represent attC. (B) Relevant portion of the Tn1331, Tn1331.2, and KQ elements (Tolmasky and Crosa, 1987; Tolmasky et al., 1988; Sarno et al., 2002; Rice et al., 2008). For clarity Tn1332, which has a more complicated structure in its direct repeats (Poirel et al., 2006), is not shown, but it could experience mobilization by homologous recombination as shown. The black dot represents attI1*. The homologous recombination pathway for generation of an aac(6′)-Ib-containing circular molecule has been proposed by Zong et al. (2009).
These multiple locations taken together with the ability of the genetic elements to spread at the molecular and cellular level provide aac(6′)-Ib genes with the capability to reach virtually all gram-negatives and other undetermined bacteria such as those that are still unculturable. The gene has also been found in plasmids harboring resistance genes of high importance such as the recently described ndm-1 (Yong et al., 2009; Bonnin et al., 2012; Villa et al., 2012).
Inhibition
The rise in drug resistance affects all known antibiotics and has been identified as one of the greatest threats to human health. Therefore, there is an immediate need for new agents with activity against multiresistant bacteria and at the present moment there is no evidence that this need will be fully met in the near future (Boucher et al., 2009). A group of pathogens that cause the majority of hospital infections, named ESKAPE (Enterococcus faecium, Staphylococcus aureus, K. pneumoniae, Acinetobacter baumannii, P aeruginosa, and Enterobacter), is becoming highly resistant to antibiotics including aminoglycosides (Rice, 2008). They carry aminoglycoside modifying enzymes genes, and one of the most common in the gram-negative members is aac(6′)-Ib (Ramirez and Tolmasky, 2010; Shaul et al., 2011; Herzog et al., 2012). An obvious solution to this problem would be the development of new aminoglycosides, a strategy that is being pursued using numerous approaches (Green et al., 2010, 2011; Houghton et al., 2010). A variety of new aminoglycoside derivatives including chemical modification of existing aminoglycosides, aminoglycoside dimers, or aminoglycoside-small molecule conjugated are being produced and tested (reviewed in Houghton et al., 2010). In particular plazomicin (ACHN-490), a novel neoglycoside derived from sisomicin that carries a hydroxymethyl group at position 6′, has shown enhanced activity against multiresistance gram-negatives and gram-positives including strains carrying aac(6′)-Ib (Endimiani et al., 2009; Landman et al., 2011).
Others are approaching the problem in such a way that the existing aminoglycosides continue to be effective by designing enzymatic inhibitors that can act in combination with the antibiotic, mimicking the strategy successfully used to curb resistance to β-lactams (Williams and Northrop, 1979; Daigle et al., 1997; Haddad et al., 1999; Liu et al., 2000; Burk and Berghuis, 2002; Boehr et al., 2003; Draker et al., 2003; Gao et al., 2005, 2006; Welch et al., 2005; Lombes et al., 2008; Magalhaes et al., 2008; De Pascale and Wright, 2010; Drawz and Bonomo, 2010; Green et al., 2012; Vong et al., 2012). However, these efforts are still scarce when one compares them to those invested to discover and design β-lactamase inhibitors. Furthermore, the attempts to find inhibitors of AAC(6′)-Ib have only yielded a compound, synthesized using non-aminoglycoside-like fragments, with a rather modest level of inhibition of AAC(6′)-Ib (Lombes et al., 2008).
An alternative approach that is being explored is silencing expression of the resistance gene. Early attempts at interfering with expression of aac(6′)-Ib consisted of identifying regions available for interaction with antisense oligonucleotides in a monocistronic in vitro synthesized mRNA by RNase H mapping in combination with computer prediction of its secondary structure (Sarno et al., 2003). The selected sites were used as targets for a collection of oligodeoxynucleotides, of which some had the ability to induce RNase H-mediated in vitro degradation of the mRNA, inhibited in vitro synthesis of the enzyme in coupled transcription/translation assays, and upon delivery by electroporation significantly reduced the number of cells surviving after exposure to amikacin (Sarno et al., 2003). The mechanism of this in vivo inhibition is most probably through RNase H digestion of the mRNA, but other possibilities such as steric hindrance cannot be discarded at this time. Alternatively, modest but significant inhibition of expression of aac(6′)-Ib was achieved by applying EGS technology, in which short antisense RNA molecules, known as external guide sequences, are used to elicit RNase P-mediated degradation of a target mRNA (Guerrier-Takada et al., 1997; Lundblad and Altman, 2010). Initially, E. coli harboring aac(6′)-Ib were transformed with recombinant clones specifying the appropriate RNA oligonucleotide sequences under an inducible promoter. The transformed derivatives were then cultured in the presence of amikacin under conditions of expression of the external guide sequences. The results showed that in a few cases the external guide sequences induced a reduction of the minimal inhibitory concentration of amikacin (Soler Bistue et al., 2007). These results were considered proof of concept, but the strategy was not viable because antisense oligonucleotides must be added from the milieu and find their way inside the cells without being degraded. Thus, nuclease resistant oligonucleotide analogs that still induce inhibition of gene expression by RNase P activation had to be found. Out of a variety of oligoribonucleotide analogs including 2′-O-methyl oligoribonucleotides, phosphorodiamidate morpholino oligomers, phosphorothioate oligodeoxynucleotides, or locked nucleic acids (LNA)/DNA co-oligomers that were tested, LNA/DNA co-oligomers with certain configurations were found to be capable of eliciting RNase P-mediated cleavage of mRNA in vitro (Soler Bistue et al., 2009). Following this finding, a selected LNA/DNA co-oligomer was added to the hyperpermeable E. coli AS19 harboring aac(6′)-Ib and it was found that growth was inhibited in the presence of amikacin, indicating that the compound may have induced RNase P-mediated inhibition of expression of the gene (Soler Bistue et al., 2009). These results were encouraging but it must be noted that inhibition of expression of aac(6′)-Ib is still far from being a viable option to overcome aminoglycoside resistance in the clinical setting. Several problems remain to be solved like inducing penetration of the oligonucleotide analogs inside wild type cells in enough quantities to exert the biological activity or achieve enough inhibition levels in spite of the usual presence of multiple copies of the gene due to its inclusion in high copy number plasmids. Toward finding solutions to these problems, recent experiments suggest that LNA/DNA co-oligomers may be able to reach the cytoplasm of untreated cells at low efficiency (Traglia et al., 2012). Strategies will have to be developed to increase the efficiency of delivery inside bacterial cells.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors' work cited in this review article was funded by Public Health Service grant 2R15AI047115 (to Marcelo E. Tolmasky) from the National Institutes of Health. María S. Ramirez is a research career member of C.O.N.I.C.E.T.
References
Avent, M. L., Rogers, B. A., Cheng, A. C., and Paterson, D. L. (2011). Current use of aminoglycosides: indications, pharmacokinetics and monitoring for toxicity. Intern. Med. J. 41, 441–449.
Bakker, E. P. (1992). Aminoglycoside and aminocyclitol antibiotics: hygromycin B is an atypical bactericidal compound that exerts effects on cells of Escherichia coli characteristics for bacteriostatic aminocyclitols. J. Gen. Microbiol. 138, 563–569.
Belousoff, M. J., Graham, B., Spiccia, L., and Tor, Y. (2009). Cleavage of RNA oligonucleotides by aminoglycosides. Org. Biomol. Chem. 7, 30–33.
Bidou, L., Allamand, V., Rousset, J. P., and Namy, O. (2012). Sense from nonsense: therapies for premature stop codon diseases. Trends Mol. Med. 18, 679–688.
Boehr, D. D., Draker, K. A., Koteva, K., Bains, M., Hancock, R. E., and Wright, G. D. (2003). Broad-spectrum peptide inhibitors of aminoglycoside antibiotic resistance enzymes. Chem. Biol. 10, 189–196.
Bonnin, R. A., Poirel, L., Carattoli, A., and Nordmann, P. (2012). Characterization of an IncFII plasmid encoding NDM-1 from Escherichia coli ST131. PLoS ONE 7:e34752. doi: 10.1371/journal.pone.0034752
Boucher, H. W., Talbot, G. H., Bradley, J. S., Edwards, J. E., Gilbert, D., Rice, L. B., et al. (2009). Bad bugs, no drugs: no ESKAPE! An update from the Infectious Diseases Society of America. Clin. Infect. Dis. 48, 1–12.
Brossier, F., Veziris, N., Aubry, A., Jarlier, V., and Sougakoff, W. (2010). Detection by Geno- type MTBDRsl test of complex resistance mechanisms to second-line drugs and ethambutol in multidrug-resistant Mycobacterium tuberculosis complex isolates. J. Clin. Microbiol. 48, 1683–1689.
Bryskier, A. (2005). “Antibiotics and antibacterial agents: classifications and structure- activity relationships,” in Antimicrobial Agents, ed A. Bryskier (Washington, DC: ASM Press), 13–38.
Burk, D. L., and Berghuis, A. M. (2002). Protein kinase inhibitors and antibiotic resistance. Pharmacol. Ther. 93, 283–292.
Busse, H. J., Wostmann, C., and Bakker, E. P. (1992). The bactericidal action of streptomycin: membrane permeabilization caused by the insertion of mistranslated proteins into the cytoplasmic membrane of Escherichia coli and subsequent caging of the antibiotic inside the cells due to degradation of these proteins. J. Gen. Microbiol. 138, 551–561.
Cambray, G., and Mazel, D. (2008). Synonymous genes explore different evolutionary landscapes. PLoS Genet. 4:e1000256. doi: 10.1371/journal.pgen.1000256
Casin, I., Bordon, F., Bertin, P., Coutrot, A., Podglajen, I., Brasseur, R., et al. (1998). Aminoglycoside 6′-N-acetyltransferase variants of the Ib type with altered substrate profile in clinical isolates of Enterobacter cloacae and Citrobacter freundii. Antimicrob. Agents Chemother. 42, 209–215.
Casin, I., Hanau-Bercot, B., Podglajen, I., Vahaboglu, H., and Collatz, E. (2003). Salmonella enterica serovar Typhimurium bla(PER-1)-carrying plasmid pSTI1 encodes an extended-spectrum aminoglycoside 6′-N-acetyltransferase of type Ib. Antimicrob. Agents Chemother. 47, 697–703.
Cattoir, V., and Nordmann, P. (2009). Plasmid-mediated quinolone resistance in gram-negative bacterial species: an update. Curr. Med. Chem. 16, 1028–1046.
Chavideh, R., Sholly, S., Panaite, D., and Tolmasky, M. E. (1999). Effects of F171 mutations in the 6′-N-acetyltransferase type Ib [AAC(6′)-Ib] enzyme on susceptibility to aminoglycosides. Antimicrob. Agents Chemother. 43, 2811–2812.
Chen, L., Chavda, K. D., Mediavilla, J. R., Zhao, Y., Fraimow, H. S., Jenkins, S. G., et al. (2012). Multiplex real-time PCR for detection of an epidemic KPC-producing Klebsiella pneumoniae ST258 clone. Antimicrob. Agents Chemother. 56, 3444–3447.
Chen, W., Biswas, T., Porter, V. R., Tsodikov, O. V., and Garneau-Tsodikova, S. (2011). Unusual regioversatility of acetyltransferase Eis, a cause of drug resistance in XDR-TB. Proc. Natl. Acad. Sci. U.S.A. 108, 9804–9808.
Chiang, W. C., Nilsson, M., Jensen, P. O., Hoiby, N., Nielsen, T. E., Givskov, M., et al. (2013). Extracellular DNA shields against aminoglycosides in Pseudomonas aeruginosa biofilms. Antimicrob. Agents Chemother. 57, 2352–2361.
Daigle, D. M., McKay, G. A., and Wright, G. D. (1997). Inhibition of aminoglycoside antibiotic resistance enzymes by protein kinase inhibitors. J. Biol. Chem. 272, 24755–24758.
De Pascale, G., and Wright, G. D. (2010). Antibiotic resistance by enzyme inactivation: from mechanisms to solutions. Chembiochem 11, 1325–1334.
Dery, K. J., Chavideh, R., Waters, V., Chamorro, R., Tolmasky, L. S., and Tolmasky, M. E. (1997). Characterization of the replication and mobilization regions of the multiresistance Klebsiella pneumoniae plasmid pJHCMW1. Plasmid 38, 97–105.
Dery, K. J., Soballe, B., Witherspoon, M. S., Bui, D., Koch, R., Sherratt, D. J., et al. (2003). The aminoglycoside 6′-N-acetyltransferase type Ib encoded by Tn1331 is evenly distributed within the cell's cytoplasm. Antimicrob. Agents Chemother. 47, 2897–2902.
De Toro, M., Rodriguez, I., Rojo-Bezares, B., Helmuth, R., Torres, C., Guerra, B., et al. (2013). pMdT1, a small ColE1-like plasmid mobilizing a new variant of the aac(6′)-Ib-cr gene in Salmonella enterica serovar Typhimurium. J. Antimicrob. Chemother. doi: 10.1093/jac/dkt001. [Epub ahead of print].
Draker, K. A., Northrop, D. B., and Wright, G. D. (2003). Kinetic mechanism of the GCN5-related chromosomal aminoglycoside acetyltransferase AAC(6′)-Ii from Enterococcus faecium: evidence of dimer subunit cooperativity. Biochemistry 42, 6565–6574.
Drawz, S. M., and Bonomo, R. A. (2010). Three decades of beta-lactamase inhibitors. Clin. Microbiol. Rev. 23, 160–201.
Dubois, V., Poirel, L., Marie, C., Arpin, C., Nordmann, P., and Quentin, C. (2002). Molecular characterization of a novel class 1 integron containing bla(GES-1) and a fused product of aac3-Ib/aac6′-Ib′ gene cassettes in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 46, 638–645.
Dyda, F., Klein, D. C., and Hickman, A. B. (2000). GCN5-related N-acetyltransferases: a structural overview. Annu. Rev. Biophys. Biomol. Struct. 29, 81–103.
Endimiani, A., Hujer, K. M., Hujer, A. M., Armstrong, E. S., Choudhary, Y., Aggen, J. B., et al. (2009). ACHN-490, a neoglycoside with potent in vitro activity against multidrug-resistant Klebsiella pneumoniae isolates. Antimicrob. Agents Chemother. 53, 4504–4507.
Galani, I., Xirouchaki, E., Kanellakopoulou, K., Petrikkos, G., and Giamarellou, H. (2002). Transferable plasmid mediating resistance to multiple antimicrobial agents in Klebsiella pneumoniae isolates in Greece. Clin. Microbiol. Infect. 8, 579–588.
Gao, F., Yan, X., Baettig, O. M., Berghuis, A. M., and Auclair, K. (2005). Regio- and chemoselective 6′-N-derivatization of aminoglycosides: bisubstrate inhibitors as probes to study aminoglycoside 6′-N-acetyltransferases. Angew. Chem. Int. Ed. Engl. 44, 6859–6862.
Gao, F., Yan, X., Shakya, T., Baettig, O. M., Ait-Mohand-Brunet, S., Berghuis, A. M., et al. (2006). Synthesis and structure-activity relationships of truncated bisubstrate inhibitors of aminoglycoside 6′-N-acetyltransferases. J. Med. Chem. 49, 5273–5281.
Garcia-Fernandez, A., Villa, L., Carta, C., Venditti, C., Giordano, A., Venditti, M., et al. (2012). Klebsiella pneumoniae ST258 producing KPC-3 identified in italy carries novel plasmids and OmpK36/OmpK35 porin variants. Antimicrob. Agents Chemother. 56, 2143–2145.
Green, K. D., Chen, W., and Garneau-Tsodikova, S. (2011). Effects of altering aminoglycoside structures on bacterial resistance enzyme activities. Antimicrob. Agents Chemother. 55, 3207–3213.
Green, K. D., Chen, W., and Garneau-Tsodikova, S. (2012). Identification and characterization of inhibitors of the aminoglycoside resistance acetyltransferase Eis from Mycobacterium tuberculosis. ChemMedChem 7, 73–77.
Green, K. D., Chen, W., Houghton, J. L., Fridman, M., and Garneau-Tsodikova, S. (2010). Exploring the substrate promiscuity of drug-modifying enzymes for the chemoenzymatic generation of N-acylated aminoglycosides. Chembiochem 11, 119–126.
Guerrier-Takada, C., Salavati, R., and Altman, S. (1997). Phenotypic conversion of drug-resistant bacteria to drug sensitivity. Proc. Natl. Acad. Sci. U.S.A. 94, 8468–8472.
Haddad, J., Vakulenko, S. B., and Mobashery, S. (1999). An antibiotic cloaked by its own resistance enzyme. J. Am. Chem. Soc. 121, 11922–11923.
Hall, R. M. (2012). Integrons and gene cassettes: hotspots of diversity in bacterial genomes. Ann. N.Y. Acad. Sci. 1267, 71–78.
Hancock, R. E. (1981). Aminoglycoside uptake and mode of action–with special reference to streptomycin and gentamicin. I. Antagonists and mutants. J. Antimicrob. Chemother. 8, 249–276.
Hermann, T. (2007). Aminoglycoside antibiotics: old drugs and new therapeutic approaches. Cell. Mol. Life Sci. 64, 1841–1852.
Herzog, I. M., Green, K. D., Berkov-Zrihen, Y., Feldman, M., Vidavski, R. R., Eldar-Boock, A., et al. (2012). 6′-Thioether tobramycin analogues: towards selective targeting of bacterial membranes. Angew. Chem. Int. Ed. Engl. 51, 5652–5656.
Hocquet, D., Vogne, C., El Garch, F., Vejux, A., Gotoh, N., Lee, A., et al. (2003). MexXY-OprM efflux pump is necessary for a adaptive resistance of Pseudomonas aeruginosa to aminoglycosides. Antimicrob. Agents Chemother. 47, 1371–1375.
Houghton, J. L., Green, K. D., Chen, W., and Garneau-Tsodikova, S. (2010). The future of aminoglycosides: the end or renaissance? Chembiochem 11, 880–902.
Huon, L. K., Fang, T. Y., and Wang, P. C. (2012). Outcomes of intratympanic gentamicin injection to treat Meniere's disease. Otol. Neurotol. 33, 706–714.
Jana, S., and Deb, J. K. (2006). Molecular understanding of aminoglycoside action and resistance. Appl. Microbiol. Biotechnol. 70, 140–150.
Kandasamy, J., Atia-Glikin, D., Shulman, E., Shapira, K., Shavit, M., Belakhov, V., et al. (2012). Increased selectivity toward cytoplasmic versus mitochondrial ribosome confers improved efficiency of synthetic aminoglycosides in fixing damaged genes: a strategy for treatment of genetic diseases caused by nonsense mutations. J. Med. Chem. 55, 10630–10643.
Katoh, K., and Standley, D. (2013). MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30, 772–780.
Kellermayer, R. (2006). Translational readthrough induction of pathogenic nonsense mutations. Eur. J. Med. Genet. 49, 445–450.
Kim, C., Villegas-Estrada, A., Hesek, D., and Mobashery, S. (2007). Mechanistic characterization of the bifunctional aminoglycoside-modifying enzyme AAC(3)-Ib/AAC(6′)-Ib' from Pseudomonas aeruginosa. Biochemistry 46, 5270–5282.
Kim, Y. T., Jang, J. H., Kim, H. C., Kim, H., Lee, K. R., Park, K. S., et al. (2011). Identification of strain harboring both aac(6′)-Ib and aac(6′)-Ib-cr variant simultaneously in Escherichia coli and Klebsiella pneumoniae. BMB Rep. 44, 262–266.
Kohanski, M. A., Dwyer, D. J., Hayete, B., Lawrence, C. A., and Collins, J. J. (2007). A common mechanism of cellular death induced by bactericidal antibiotics. Cell 130, 797–810.
Kondo, J., Hainrichson, M., Nudelman, I., Shallom-Shezifi, D., Barbieri, C. M., Pilch, D. S., et al. (2007). Differential selectivity of natural and synthetic aminoglycosides towards the eukaryotic and prokaryotic decoding A sites. Chembiochem 8, 1700–1709.
Labaune, J. M., Bleyzac, N., Maire, P., Jelliffe, R. W., Boutroy, M. J., Aulagner, G., et al. (2001). Once-a-day individualized amikacin dosing for suspected infection at birth based on population pharmacokinetic models. Biol. Neonate 80, 142–147.
Lambert, T., Ploy, M. C., and Courvalin, P. (1994). A spontaneous point mutation in the aac(6′)-Ib' gene results in altered substrate specificity of aminoglycoside 6′-N-acetyltransferase of a Pseudomonas fluorescens strain. FEMS Microbiol. Lett. 115, 297–304.
Landman, D., Kelly, P., Backer, M., Babu, E., Shah, N., Bratu, S., et al. (2011). Antimicrobial activity of a novel aminoglycoside, ACHN-490, against Acinetobacter baumannii and Pseudomonas aeruginosa from New York City. J. Antimicrob. Chemother. 66, 332–334.
Liu, M., Haddad, J., Azucena, E., Kotra, L. P., Kirzhner, M., and Mobashery, S. (2000). Tethered bisubstrate derivatives as probes for mechanism and as inhibitors of aminoglycoside 3′-phosphotransferases. J. Org. Chem. 65, 7422–7431.
Lombes, T., Begis, G., Maurice, F., Turcaud, S., Lecourt, T., Dardel, F., et al. (2008). NMR-guided fragment-based approach for the design of AAC(6′)-Ib ligands. Chembiochem 9, 1368–1371.
Lundblad, E. W., and Altman, S. (2010). Inhibition of gene expression by RNase P. N. Biotechnol. 27, 212–221.
Macleod, D. L., Nelson, L. E., Shawar, R. M., Lin, B. B., Lockwood, L. G., Dirk, J. E., et al. (2000). Aminoglycoside-resistance mechanisms for cystic fibrosis Pseudomonas aeruginosa isolates are unchanged by long-term, intermittent, inhaled tobramycin treatment. J. Infect. Dis. 181, 1180–1184.
Magalhaes, M. L., Vetting, M. W., Gao, F., Freiburger, L., Auclair, K., and Blanchard, J. S. (2008). Kinetic and structural analysis of bisubstrate inhibition of the Salmonella enterica aminoglycoside 6′-N-acetyltransferase. Biochemistry 47, 579–584.
Magnet, S., and Blanchard, J. S. (2005). Molecular insights into aminoglycoside action and resistance. Chem. Rev. 105, 477–498.
Magnet, S., Smith, T. A., Zheng, R., Nordmann, P., and Blanchard, J. S. (2003). Aminoglycoside resistance resulting from tight drug binding to an altered aminoglycoside acetyltransferase. Antimicrob. Agents Chemother. 47, 1577–1583.
Majumder, K., Wei, L., Annedi, S., and Kotra, L. P. (2007). “Aminoglycoside antibiotics,” in Enzyme-Mediated Resistance to Antibiotics: Mechanisms, Dissemination, and Prospects for Inhibition, eds R. A. Bonomo and M. E. Tolmasky (Washington, DC: ASM Press), 7–20.
Maurice, F., Broutin, I., Podglajen, I., Benas, P., Collatz, E., and Dardel, F. (2008). Enzyme structural plasticity and the emergence of broad-spectrum antibiotic resistance. EMBO Rep. 9, 344–349.
Mehta, R., and Champney, W. S. (2003). Neomycin and paromomycin inhibit 30S ribosomal subunit assembly in Staphylococcus aureus. Curr. Microbiol. 47, 237–243.
Mendes, R., Toleman, M., Ribeiro, J., Sader, H., Jones, R., and Walsh, T. (2004). Integron carrying a novel metallo-b-lactamase gene, blaIMP-16, and a fused form of aminoglycoside-resistance gene aac(6′)-30/aac(6′)-Ib: report from the SENTRY antimicrobial surveillance program. Antimicrob. Agents Chemother. 48, 4693–4702.
Menzies, D., Benedetti, A., Paydar, A., Martin, I., Royce, S., Pai, M., et al. (2009). Effect of duration and intermittency of rifampin on tuberculosis treatment outcomes: a systematic review and meta-analysis. PLoS Med. 6:e1000146. doi: 10.1371/journal.pmed.1000146
Mikkelsen, N. E., Brannvall, M., Virtanen, A., and Kirsebom, L. A. (1999). Inhibition of RNase P RNA cleavage by aminoglycosides. Proc. Natl. Acad. Sci. U.S.A. 96, 6155–6160.
Miller, G. H., Sabatelli, F. J., Hare, R. S., Glupczynski, Y., Mackey, P., Shlaes, D., et al. (1997). The most frequent aminoglycoside resistance mechanisms–changes with time and geographic area: a reflection of aminoglycoside usage patterns? Aminoglycoside Resistance Study Groups. Clin. Infect. Dis. 24(Suppl. 1), S46–S62.
Morita, Y., Tomida, J., and Kawamura, Y. (2012). MexXY multidrug efflux system of Pseudomonas aeruginosa. Front. Microbiol. 3:408. doi: 10.3389/fmicb.2012.00408
Muir, M. E., Van Heeswyck, R. S., and Wallace, B. J. (1984). Effect of growth rate on streptomycin accumulation by Escherichia coli and Bacillus megaterium. J. Gen. Microbiol. 130, 2015–2022.
Neuwald, A. F., and Landsman, D. (1997). GCN5-related histone N-acetyltransferases belong to a diverse superfamily that includes the yeast SPT10 protein. Trends Biochem. Sci. 22, 154–155.
O'Connor, M., De Stasio, E. A., and Dahlberg, A. E. (1991). Interaction between 16S ribosomal RNA and ribosomal protein S12: differential effects of paromomycin and streptomycin. Biochimie 73, 1493–1500.
Over, U., Gur, D., Unal, S., Miller, G. H., and Aminoglycoside Resistance Study Group. (2001). The changing nature of aminoglycoside resistance mechanisms and prevalence of newly recognized resistance mechanisms in Turkey. Clin. Microbiol. Infect. 7, 470–478.
Pacheu-Grau, D., Perez-Delgado, L., Gomez-Diaz, C., Fraile-Rodrigo, J., Montoya, J., and Ruiz-Pesini, E. (2012). Mitochondrial ribosome and Meniere's disease: a pilot study. Eur. Arch. Otorhinolaryngol. 269, 2003–2008.
Panaite, D. M., and Tolmasky, M. E. (1998). Characterization of mutants of the 6′-N-acetyltransferase encoded by the multiresistance transposon Tn1331: effect of Phen171-to-Leu171 and Tyr80-to-Cys80 substitutions. Plasmid 39, 123–133.
Poirel, L., Cabanne, L., Collet, L., and Nordmann, P. (2006). Class II transposon-borne structure harboring metallo-beta-lactamase gene blaVIM-2 in Pseudomonas putida. Antimicrob. Agents Chemother. 50, 2889–2891.
Pourreza, A., Witherspoon, M., Fox, J., Newmark, J., Bui, D., and Tolmasky, M. E. (2005). Mutagenesis analysis of a conserved region involved in acetyl coenzyme A binding in the aminoglycoside 6′-N-acetyltransferase type Ib encoded by plasmid pJHCMW1. Antimicrob. Agents Chemother. 49, 2979–2982.
Quiroga, M. P., Andres, P., Petroni, A., Soler Bistue, A. J., Guerriero, L., Vargas, L. J., et al. (2007). Complex class 1 integrons with diverse variable regions, including aac(6′)-Ib-cr, and a novel allele, qnrB10, associated with ISCR1 in clinical enterobacterial isolates from Argentina. Antimicrob. Agents Chemother. 51, 4466–4470.
Ramirez, M. S., Parenteau, T. R., Centron, D., and Tolmasky, M. E. (2008). Functional characterization of Tn1331 gene cassettes. J. Antimicrob. Chemother. 62, 669–673.
Ramirez, M. S., and Tolmasky, M. E. (2010). Aminoglycoside modifying enzymes. Drug Resist. Updat. 13, 151–171.
Rather, P. N., Munayyer, H., Mann, P. A., Hare, R. S., Miller, G. H., and Shaw, K. J. (1992). Genetic analysis of bacterial acetyltransferases: identification of amino acids determining the specificities of the aminoglycoside 6′-N-acetyltransferase Ib and IIa proteins. J. Bacteriol. 174, 3196–3203.
Rice, L. B. (2008). Federal funding for the study of antimicrobial resistance in nosocomial pathogens: no ESKAPE. J. Infect. Dis. 197, 1079–1081.
Rice, L. B., Carias, L. L., Hutton, R. A., Rudin, S. D., Endimiani, A., and Bonomo, R. A. (2008). The KQ element, a complex genetic region conferring transferable resistance to carbapenems, aminoglycosides, and fluoroquinolones in Klebsiella pneumoniae. Anti-microb. Agents Chemother. 52, 3427–3429.
Rich, D. P., Anderson, M. P., Gregory, R. J., Cheng, S. H., Paul, S., Jefferson, D. M., et al. (1990). Expression of cystic fibrosis transmembrane conductance regulator corrects defective chloride channel regulation in cystic fibrosis airway epithelial cells. Nature 347, 358–363.
Robicsek, A., Strahilevitz, J., Jacoby, G. A., Macielag, M., Abbanat, D., Park, C. H., et al. (2006). Fluoroquinolone-modifying enzyme: a new adaptation of a common aminoglycoside acetyltransferase. Nat. Med. 12, 83–88.
Rodriguez-Martinez, J. M., Cano, M. E., Velasco, C., Martinez-Martinez, L., and Pascual, A. (2011). Plasmid-mediated quinolone resistance: an update. J. Infect. Chemother. 17, 149–182.
Romanowska, J., Reuter, N., and Trylska, J. (2013). Comparing aminoglycoside binding sites in bacterial ribosomal RNA and aminoglycoside modifying enzymes. Proteins 81, 63–80.
Ruiz, J., Pons, M. J., and Gomes, C. (2012). Transferable mechanisms of quinolone resistance. Int. J. Antimicrob. Agents 40, 196–203.
Salipante, S. J., and Hall, B. G. (2003). Determining the limits of the evolutionary potential of an antibiotic resistance gene. Mol. Biol. Evol. 20, 653–659.
Sarno, R., Ha, H., Weinsetel, N., and Tolmasky, M. E. (2003). Inhibition of aminoglycoside 6′-N-acetyltransferase type Ib-mediated amikacin resistance by antisense oligodeoxynucleotides. Antimicrob. Agents Chemother. 47, 3296–3304.
Sarno, R., McGillivary, G., Sherratt, D. J., Actis, L. A., and Tolmasky, M. E. (2002). Complete nucleotide sequence of Klebsiella pneumoniae multiresistance plasmid pJHCMW1. Antimicrob. Agents Chemother. 46, 3422–3427.
Schmitt, E., Galimand, M., Panvert, M., Courvalin, P., and Mechulam, Y. (2009). Structural bases for 16 s rRNA methylation catalyzed by ArmA and RmtB methyltransferases. J. Mol. Biol. 388, 570–582.
Shaul, P., Green, K. D., Rutenberg, R., Kramer, M., Berkov-Zrihen, Y., Breiner-Goldstein, E., et al. (2011). Assessment of 6′- and 6′”-N-acylation of aminoglycosides as a strategy to overcome bacterial resistance. Org. Biomol. Chem. 9, 4057–4063.
Shaw, K. J., Rather, P. N., Hare, R. S., and Miller, G. H. (1993). Molecular genetics of aminoglycoside resistance genes and familial relationships of the aminoglycoside-modifying enzymes. Microbiol. Rev. 57, 138–163.
Shmara, A., Weinsetel, N., Dery, K. J., Chavideh, R., and Tolmasky, M. E. (2001). Systematic analysis of a conserved region of the aminoglycoside 6′-N-acetyltransferase type Ib. Antimicrob. Agents Chemother. 45, 3287–3292.
Sneath, P. H., and Sokal, R. R. (1973). Numerical Taxonomy. The Principles and Practice of Numerical Classification. San Francisco, CA: W. H. Freeman.
Soler Bistue, A. J., Birshan, D., Tomaras, A. P., Dandekar, M., Tran, T., Newmark, J., et al. (2008). Klebsiella pneumoniae multiresistance plasmid pMET1: similarity with the Yersinia pestis plasmid pCRY and integrative conjugative elements. PLoS ONE 3:e1800. doi: 10.1371/journal.pone.0001800
Soler Bistue, A. J., Ha, H., Sarno, R., Don, M., Zorreguieta, A., and Tolmasky, M. E. (2007). External guide sequences targeting the aac(6′)-Ib mRNA induce inhibition of amikacin resistance. Antimicrob. Agents Chemother. 51, 1918–1925.
Soler Bistue, A. J., Martin, F. A., Petroni, A., Faccone, D., Galas, M., Tolmasky, M. E., et al. (2006). Vibrio cholerae InV117, a class 1 integron harboring aac(6′)-Ib and blaCTX-M-2, is linked to transposition genes. Antimicrob. Agents Chemother. 50, 1903–1907.
Soler Bistue, A. J., Martin, F. A., Vozza, N., Ha, H., Joaquin, J. C., Zorreguieta, A., et al. (2009). Inhibition of aac(6′)-Ib-mediated amikacin resistance by nuclease-resistant external guide sequences in bacteria. Proc. Natl. Acad. Sci. U.S.A. 106, 13230–13235.
Strahilevitz, J., Jacoby, G. A., Hooper, D. C., and Robicsek, A. (2009). Plasmid-mediated quinolone resistance: a multifaceted threat. Clin. Microbiol. Rev. 22, 664–689.
Taber, H. W., Mueller, J. P., Miller, P. F., and Arrow, A. S. (1987). Bacterial uptake of aminoglycoside antibiotics. Microbiol. Rev. 51, 439–457.
Tolmasky, M. (2007a). “Aminogylcoside-modifying enzymes: characteristics, localization, and dissemination,” in Enzyme-Mediated Resistance to Antibiotics: Mechanisms, Dissemination, and Prospects for Inhibition, eds R. Bonomo and M. Tolmasky (Washington, DC: ASM Press), 35–52.
Tolmasky, M. E. (2007b). “Overview of dissemination mechanisms of genes coding for resistance to antibiotics,” in Enzyme-Mediated Resistance to Antibiotics: Mechanisms, Dissemination, and Prospects for Inhibition, eds R. Bonomo and M. E. Tolmasky (Washington, DC: ASM Press), 267–270.
Tolmasky, M. E. (1990). Sequencing and expression of aadA, bla, and tnpR from the multiresistance transposon Tn1331. Plasmid 24, 218–226.
Tolmasky, M. E., Chamorro, R. M., Crosa, J. H., and Marini, P. M. (1988). Transposon-mediated amikacin resistance in Klebsiella pneumoniae. Antimicrob. Agents Chemother. 32, 1416–1420.
Tolmasky, M. E., and Crosa, J. H. (1987). Tn1331, a novel multiresistance transposon encoding resistance to amikacin and ampicillin in Klebsiella pneumoniae. Antimicrob. Agents Chemother. 31, 1955–1960.
Tolmasky, M. E., and Crosa, J. H. (1991). Regulation of plasmid-mediated iron transport and virulence in Vibrio anguillarum. Biol. Met. 4, 33–35.
Tolmasky, M. E., and Crosa, J. H. (1993). Genetic organization of antibiotic resistance genes (aac(6′)-Ib, aadA, and oxa9) in the multiresistance transposon Tn1331. Plasmid 29, 31–40.
Traglia, G., Davies Sala, C., Fuxman Bass, J., Soler Bistué, A., Zorreguieta, A., Ramirez, M. S., et al. (2012). Internalization of locked nucleic acids/DNA hybrid oligomers into Escherichia coli. Biores. Open Access 1, 260–263.
Vakulenko, S. B., and Mobashery, S. (2003). Versatility of aminoglycosides and prospects for their future. Clin. Microbiol. Rev. 16, 430–450.
van ‘t Veen, A., Van Der Zee, A., Nelson, J., Speelberg, B., Kluytmans, J. A., and Buiting, A. G. (2005). Outbreak of infection with a multiresistant Klebsiella pneumoniae strain associated with contaminated roll boards in operating rooms. J. Clin. Microbiol. 43, 4961–4967.
Vetting, M. W., Lp, S. D. C., Yu, M., Hegde, S. S., Magnet, S., Roderick, S. L., et al. (2005). Structure and functions of the GNAT superfamily of acetyltransferases. Arch. Biochem. Biophys. 433, 212–226.
Vetting, M. W., Magnet, S., Nieves, E., Roderick, S. L., and Blanchard, J. S. (2004). A bacterial acetyltransferase capable of regioselective N-acetylation of antibiotics and histones. Chem. Biol. 11, 565–573.
Vetting, M. W., Park, C. H., Hegde, S. S., Jacoby, G. A., Hooper, D. C., and Blanchard, J. S. (2008). Mechanistic and structural analysis of aminoglycoside N-acetyltransferase AAC(6′)-Ib and its bifunctional, fluoroquinolone-active AAC(6′)-Ib-cr variant. Biochemistry 47, 9825–9835.
Vicens, Q., and Westhof, E. (2003). Molecular recognition of aminoglycoside antibiotics by ribosomal RNA and resistance enzymes: an analysis of x-ray crystal structures. Biopolymers 70, 42–57.
Villa, L., Poirel, L., Nordmann, P., Carta, C., and Carattoli, A. (2012). Complete sequencing of an IncH plasmid carrying the blaNDM-1, blaCTX-M-15 and qnrB1 genes. J. Antimicrob. Chemother. 67, 1645–1650.
Vong, K., Tam, I. S., Yan, X., and Auclair, K. (2012). Inhibitors of aminoglycoside resistance activated in cells. ACS Chem. Biol. 7, 470–475.
Wachino, J., and Arakawa, Y. (2012). Exogenously acquired 16S rRNA methyltransferases found in aminoglycoside-resistant pathogenic Gram-negative bacteria: an update. Drug Resist. Updat. 15, 133–148.
Warburg, G., Hidalgo-Grass, C., Partridge, S. R., Tolmasky, M. E., Temper, V., Moses, A. E., et al. (2012). A carbapenem-resistant Klebsiella pneumoniae epidemic clone in Jerusalem: sequence type 512 carrying a plasmid encoding aac(6′)-Ib. J. Antimicrob. Chemother. 67, 898–901.
Welch, K. T., Virga, K. G., Whittemore, N. A., Ozen, C., Wright, E., Brown, C. L., et al. (2005). Discovery of non-carbohydrate inhibitors of aminoglycoside-modifying enzymes. Bioorg. Med. Chem. 13, 6252–6263.
Williams, J. W., and Northrop, D. B. (1979). Synthesis of a tight-binding, multisubstrate analog inhibitor of gentamicin acetyltransferase I. J. Antibiot. 32, 1147–1154.
Woloj, M., Tolmasky, M. E., Roberts, M. C., and Crosa, J. H. (1986). Plasmid-encoded amikacin resistance in multiresistant strains of Klebsiella pneumoniae isolated from neonates with meningitis. Antimicrob. Agents Chemother. 29, 315–319.
Woodford, N., Carattoli, A., Karisik, E., Underwood, A., Ellington, M. J., and Livermore, D. M. (2009). Complete nucleotide sequences of plasmids pEK204, pEK499, and pEK516, encoding CTX-M enzymes in three major Escherichia coli lineages from the United Kingdom, all belonging to the international O25:H4-ST131 clone. Antimicrob. Agents Chemother. 53, 4472–4482.
Wright, G. D., and Berghuis, A. M. (2007). “Structural aspects of aminoglycoside modifying enzymes,” in Enzyme-Mediated Resistance to Antibiotics: Mechanisms, Dissemination, and Prospects for Inhibition, eds R. Bonomo and M. Tolmasky (Washington, DC: ASM Press), 21–33.
Wright, G. D., and Ladak, P. (1997). Overexpression and characterization of the chromosomal aminoglycoside 6′-N-acetyltransferase from Enterococcus faecium. Antimicrob. Agents Chemother. 41, 956–960.
Wybenga-Groot, L. E., Draker, K., Wright, G. D., and Berghuis, A. M. (1999). Crystal structure of an aminoglycoside 6′-N-acetyltransferase: defining the GCN5-related N-acetyltransferase superfamily fold. Structure 7, 497–507.
Yao, J., and Moellering, R. (2007). “Antibacterial agents,” in Manual of Clinical Microbiology, eds P. Murray, E. Baron, J. Jorgensen, M. Landry, and M. Pfaller (Washington, DC: American Society for Microbiology Press), 1077–1113.
Yong, D., Toleman, M. A., Giske, C. G., Cho, H. S., Sundman, K., Lee, K., et al. (2009). Characterization of a new metallo-beta-lactamase gene, bla(NDM-1), and a novel erythromycin esterase gene carried on a unique genetic structure in Klebsiella pneumoniae sequence type 14 from India. Antimicrob. Agents Chemother. 53, 5046–5054.
Zaher, H. S., and Green, R. (2009). Fidelity at the molecular level: lessons from protein synthesis. Cell 136, 746–762.
Zingman, L. V., Park, S., Olson, T. M., Alekseev, A. E., and Terzic, A. (2007). Aminoglycoside-induced translational read-through in disease: overcoming nonsense mutations by pharmacogenetic therapy. Clin. Pharmacol. Ther. 81, 99–103.
Keywords: antibiotic resistance, aminoglycoside, inhibition, acetyltransferase, mobile elements, integron, transposon
Citation: Ramirez MS, Nikolaidis N and Tolmasky ME (2013) Rise and dissemination of aminoglycoside resistance: the aac(6′)-Ib paradigm. Front. Microbiol. 4:121. doi: 10.3389/fmicb.2013.00121
Received: 20 March 2013; Accepted: 29 April 2013;
Published online: 17 May 2013.
Edited by:
Marilyn C. Roberts, University of Washington, USACopyright © 2013 Ramirez, Nikolaidis and Tolmasky. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in other forums, provided the original authors and source are credited and subject to any copyright notices concerning any third-party graphics etc.
*Correspondence: Marcelo E. Tolmasky, Department of Biological Science, Center for Applied Biotechnology Studies, College of Natural Sciences and Mathematics, California State University Fullerton, 800 N State College Boulevard, Fullerton, CA 92831, USA. e-mail:bXRvbG1hc2t5QGZ1bGxlcnRvbi5lZHU=