- College of Life and Environmental Sciences, University of Exeter, Penryn, UK
Interest in using bacteriophages to control the growth and spread of bacterial pathogens is being revived in the wake of widespread antibiotic resistance. However, little is known about the ecological effects that high concentrations of phages in the environment might have on natural microbial communities. We review the current evidence suggesting phage-mediated environmental perturbation, with a focus on agricultural examples, and describe the potential implications for human health and agriculture. Specifically, we examine the known and potential consequences of phage application in certain agricultural practices, discuss the risks of evolved bacterial resistance to phages, and question whether the future of phage therapy will emulate that of antibiotic treatment in terms of widespread resistance. Finally, we propose some basic precautions that could preclude such phenomena and highlight existing methods for tracking bacterial resistance to phage therapeutic agents.
Introduction
The selection for and subsequent evolution of antibiotic resistance in bacterial populations, both in the wider environment and during clinical treatment, presents a serious challenge to human health (Kåhrström, 2013). Although still controversial, it is increasingly clear that agricultural use of antibiotics has played a role in the continued selection for resistance genes and that the movement of these genes into pathogens of clinical relevance is possible (see Smith et al., 2009; van Cleef et al., 2010; Heuer et al., 2011; Zhu et al., 2013). Nosocomial environments also act as significant sources of antibiotic resistance and the transfer of antibiotic resistance genes into the agricultural environment has recently been demonstrated, suggesting the genetic exchange between environments works both ways (Price et al., 2012). As a result, alternative antimicrobial strategies are being sought. One such strategy is to utilize naturally occurring viral predators of bacteria: bacteriophages (phages). Lytic phages are capable of killing bacteria by invading and propagating within the host cell and then lysing open the cell to “burst” out, thus killing the bacteria. This is in contrast to temperate phages, which integrate into the genome of their hosts and can be transmitted vertically, serving as a refuge for phages in harsh environments (Svircev et al., 2011). The latter, although important to the ecology and evolution of bacterial populations, are not commonly considered for use as biocontrol and therefore will not be covered further in this review (but see Hyman and Abedon, 2010 for review of the effects of lysogeny on bacterial resistance). Despite being discovered as potential therapeutic agents over 80 years ago (d’Herelle, 1929), and their continual use in Russia and Georgia ever since (Kutter et al., 2010), few clinical trials of so-called “phage therapy” have been conducted in Western medicine (Wright et al., 2009; Sarker et al., 2012). Accordingly, no clinical phage therapy products are currently available in the West, and regulatory burdens may have dampened pharmaceutical interest, as years of research and clinical trials can cost millions of euros, presenting a formidable hurdle (Pirnay et al., 2011; Brüssow, 2012).
A more viable route to market has been provided by use of phages in agriculture and aquaculture (Jones et al., 2012; Martínez-Díaz and Hipólito-Morales, 2013). Recent review suggests that although phage therapy will not be the panacea that broad-spectrum antibiotics once were, phages could nonetheless play an important role in treating infections and maintaining food yields (Allen et al., 2013). Indeed, phage usage in agriculture has shown promise for treating numerous plant pathogens (Frampton et al., 2012), and some formulations of phages have been sold for large-scale environmental application (e.g., AgriPhageTM, Omnilytics). Likewise, the Food and Drug Administration in the USA has approved a product for the treatment of food products prior to market (ListshieldTM, Intralytix) and classed the use of phages in this specific context as “generally recognized as safe” (Food and Drug Administration (FDA), 2013). Another fruitful avenue for phage therapy may be aquaculture, an industry that has increased globally by over 10-fold in the last 30 years (Food and Agriculture Organization [FAO], 2012). Microbial diseases represent a severe threat to aquaculture productivity; accordingly, phages capable of lysing pathogens such as Flavobacterium psychrophilum, the causative agent of bacterial cold-water disease, have been isolated and tested as therapeutic agents (Kim et al., 2010). Combined, the scope for phage therapy to fill food production and clinical niches left vacant by redundant antibiotics seems vast. However, whilst phage therapy as a biopesticide could prove a useful tool, it also presents a risk for repeating the mistakes made with overuse of antibiotics, and the subsequently high levels of evolved antimicrobial resistance observed both in the environment and in hospitals (Zhu et al., 2013). For example, it is unclear whether introducing high concentrations of diverse phage types into the environment will select for broad resistance, making future treatments less likely to succeed. Furthermore, the role of phage-mediated selection in shaping bacterial growth rates and virulence to their hosts remains poorly understood.
Despite a number of reviews highlighting the need for increased understanding of environmental perturbations from anthropogenic antibiotic input (Martínez, 2008; Ding and He, 2010;Allen et al., 2013), the impact of antibiotic use on microbial communities has rarely been taken into account when designing treatment or application. This is particularly surprising given the known natural importance of these chemicals in shaping competition among bacterial strains (D’Costa et al., 2011). As phages are also known to select for resistant bacteria (Buckling and Rainey, 2002) and to mediate competition among strains (Bohannan and Lenski, 2000; Koskella et al., 2012), the same risks should apply to this alternative treatment. Unfortunately, little is currently known about the effects of applying high titers of phages to natural microbial communities. Most importantly, it is possible that with uncontrolled application of phages in the environment the future efficacy of phage therapy in a clinical setting could be reduced – a mistake we cannot afford given the need for new antimicrobial therapies as a result of drug-resistant pathogens (Levy and Marshall, 2004). Phage therapy in agriculture could serve as a testing ground for clinical use (Stone, 2002; Levin and Bull, 2004). However, there could also be a conflict of interest if cross-resistance to phage treatments is possible and if these resistant bacteria can spread from agricultural to clinical settings, as has been observed for antibiotic resistance (van Cleef et al., 2010; Perry and Wright, 2013). If phage therapy treatments fail, or improper use of phages in the environment goes unchecked, the use of widespread phage biocontrol in agriculture could jeopardize the future of phage therapy in hospitals. Fortunately, our understanding of phage-mediated selection is growing at a rapid pace and a new era of genomic investigation should allow monitoring of microbial communities following phage therapy. In this review, we will discuss the status of the field of phage therapy and consider the implications of phage host range and bacterial resistance. We suggest that with a few precautions phage therapy may be effective for treating bacterial infections in agriculture, aquaculture, healthcare, food production and food safety.
The Risks of Antimicrobial Use in Agriculture
The argument against using antibiotics as standard agricultural practice, both to improve growth rates and prevent disease, is not new (Witte, 1998) and has been extensively reviewed previously (Singer et al., 2003). However, unequivocally demonstrating increased resistance as a consequence of agricultural usage has proved elusive (Perry and Wright, 2013). A wave of new data supporting both direct and indirect routes of antibiotic resistance genes between agricultural and human populations suggests a bidirectional zoonotic exchange (Price et al., 2012). For example, recent studies have found diverse and abundant resistance genes in manure prior to disposal in the environment (Zhu et al., 2013) and a high prevalence of resistance to multiple antibiotics in enterobacteria isolated from tomato farms (Micallef et al., 2013) and in bacteria from manure-amended soils (Popowska et al., 2012). Furthermore, methicillin-resistant Staphylococcus aureus (MRSA) rates in workers on swine farms have been shown to be higher than for the average population in both North America and Europe (Voss et al., 2005;Khanna et al., 2008; Smith et al., 2009; van Cleef et al., 2010). Finally, calves treated with antibiotics are also more likely to carry MRSA and there is a direct association between intensity of animal contact and human MRSA carriage (Graveland et al., 2010). A similar trend is seen in aquaculture where bacteria nearer to farms were found to have higher levels of antibiotic resistance than nearby coastal regions in Italy (Labella et al., 2013). The increasing number of studies supporting the hypothesis that environmental use of antibiotics has contributed to selection for antibiotic resistance suggests that non-prudent use of antibiotics in healthcare and agriculture may reduce the effectiveness of antibiotic strategies as an essential treatment for disease.
As an alternative to antibiotic use, the application of phages in agriculture is being trialed as a biopesticide to control plant pathogens of tomato (Jones et al., 2012), citrus (Balogh et al., 2008), and onion (Lang et al., 2007) among others (reviewed in Svircev et al., 2011). For example, Erwinia amylovora (the causative agent of fire blight) infections are affecting a number of crop species in orchards across North America and Europe (see Malnoy et al., 2012 for review). Although antibiotics have traditionally been employed to control this disease, the emergence of streptomycin resistant strains (McManus et al., 2002) and a desire to reduce antibiotic use in the environment has led to the use of phages as an alternative. Phage biocontrol clearly has the potential to control fire blight infections, as lytic phages have been isolated that are highly infective to the pathogen, but definitive field trials are currently lacking. Given the evidenced risks of movement of antibiotic resistance genes between agricultural to human pathogens, we should ask whether the large-scale application of phages is likely to repeat these past mistakes. Until appropriate studies are conducted, the subsequent consequences of applying phages in agriculture for the spread of antibiotic resistance, the evolution of the pathogen, and the community of microbes within the plants and soil remain unknown.
Design and Implementation of Phage Therapy and Biocontrol
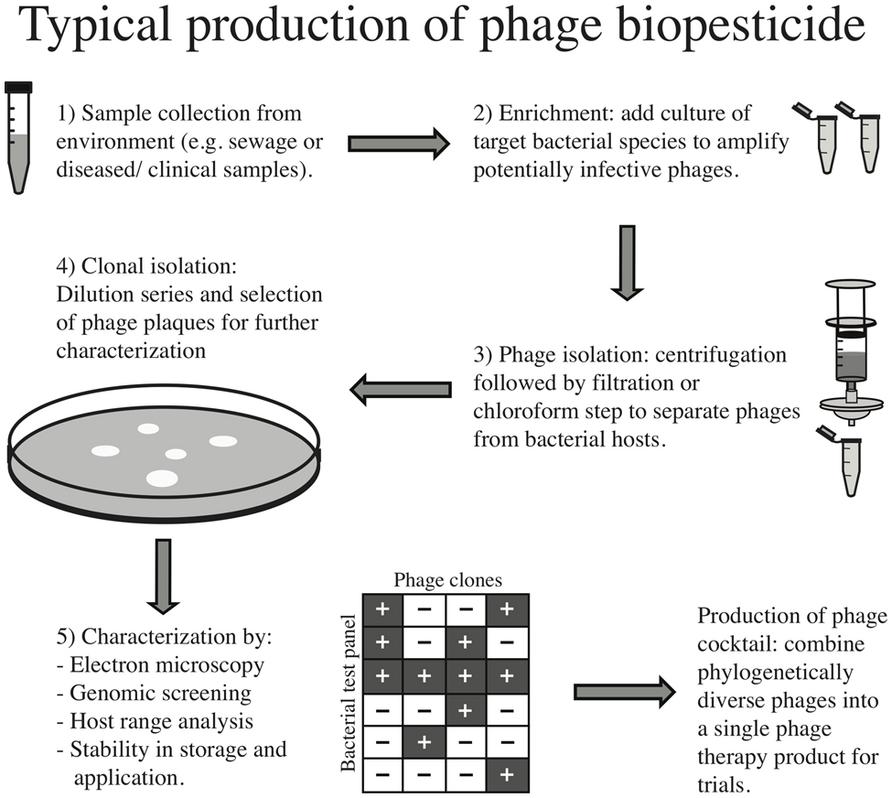
The process of preparing a phage therapy product for clinical use has been thoroughly described (Merabishvili et al., 2009; Gill and Hyman, 2010). Figure 1 also describes this process for clinical and environmental samples. Briefly, environmental samples such as sewage or clinical samples from infected wounds are collected. The next step normally employs an “enrichment” process whereby the target bacterial species is added to the sample to increase the titer of phages infective to this strain. The sample is either filtered or chloroform is added to separate phages from bacteria, and individual phage “plaques” (i.e., the localized absence of bacterial growth in a lawn due to lysis) are chosen for further characterization. Transmission electron microscopy may be employed to assign family level phylogeny and genetic sequencing for finer scale taxonomic assignment, and screening of virulence factors is typically conducted. Other properties such as stability across a range of environmental conditions may be tested for optimal storage and production. Importantly, phage host range is typically tested to ensure the selected isolates have high efficacy against the pathogen of interest. However, this screening is most often done using a reference panel of laboratory stocks, rather than a large subset of bacteria from the local environment in which the phages will be applied, leading to a biased host range description. Therefore one way to reduce the possible community-level effects of applying phages would be to perform large-scale host range analyses across a biologically meaningful panel of isolates (i.e., those bacterial strains and species with which the phages are likely to interact once applied), as has been done successfully in the field of microbial ecology (Flores et al., 2011; Koskella and Meaden, 2013).
Once individual phages have been isolated and characterized, phage cocktails are produced by combining multiple, usually phylogenetically diverse, phages into one formulation. The idea behind these combined treatments is twofold: first, the use of multiple phages should increase the breadth of efficacy of the treatment to include most circulating strains of a pathogen; and second, the evolution of bacterial resistance should be slowed relative to single phage treatment. Whilst this approach could select for broadly resistant bacterial hosts, a number of studies suggest that broad resistance will carry a larger cost and therefore will not spread as rapidly (Bohannan and Lenski, 2000; Hall et al., 2012; Koskella et al., 2012). For example, strains of the plant pathogen, Pseudomonas syringae, evolved in the presence of three phage types were just as likely to evolve resistance against all three as those strains evolved in the presence of a single phage type. Furthermore, bacteria treated with multiple phages were no more likely to be cross-resistant to novel phages but were found to have paid a greater cost for their resistance than bacteria treated with a single phage type (Koskella et al., 2012). A similar result was observed for Pseudomonas aeruginosa strains treated with one versus four phages (Hall et al., 2012). This emulates the common practice of using combined antibiotic treatment to decrease the likelihood of evolved antibiotic resistance (Traugott et al., 2011; Vardakas et al., 2013). Overall, a greater understanding of the costs of resistance to phage predation and of synergistic effects among phages in controlling bacterial pathogens will allow for a more informed development and application of treatment, and ideally the prevention of widespread resistance.
The Implications of Evolved Resistance to Phages
Despite the promise of many phage therapy trials, the use of phages to control bacterial pathogen begs the question: could the evolution of phage resistance mirror the evolution and spread of antibiotic resistance? Numerous studies have shown that natural phages are well-adapted to their local bacterial populations (Vos et al., 2009; Koskella et al., 2011) and that bacteria in turn adapt to resist their local phages (Kunin et al., 2008; Koskella, 2013). However, a recent review of phage resistance as a result of prolonged phage therapy (Ormälä and Jalasvuori, 2013) concludes that, as it is possible to isolate phages infective to bacteria from different geographical locations and evolutionary histories (e.g., Flores et al., 2011), long-term resistance need not be a concern as a diverse set of phages capable of infecting newly resistant strains will always be available. Local phage diversity is often high (Breitbart and Rohwer, 2005), so infective phages should be easy to isolate from just a few environmental samples. However, this parallels the problems of antibiotic discovery – the process from discovery to a useable product is arduous and expensive, so despite a ready source of infective phages few companies are investing in treatments (Brüssow, 2012). If bacterial resistance to phage infection emerges rapidly and production is slow, redundancy of treatments seems likely. As pointed out by Pirnay et al. (2011) a reactive phage therapy program that is capable of rapidly isolating, screening, and applying infective phages will be better placed to respond to phage resistance than the slow and expensive process of approval and licensing for each phage type.
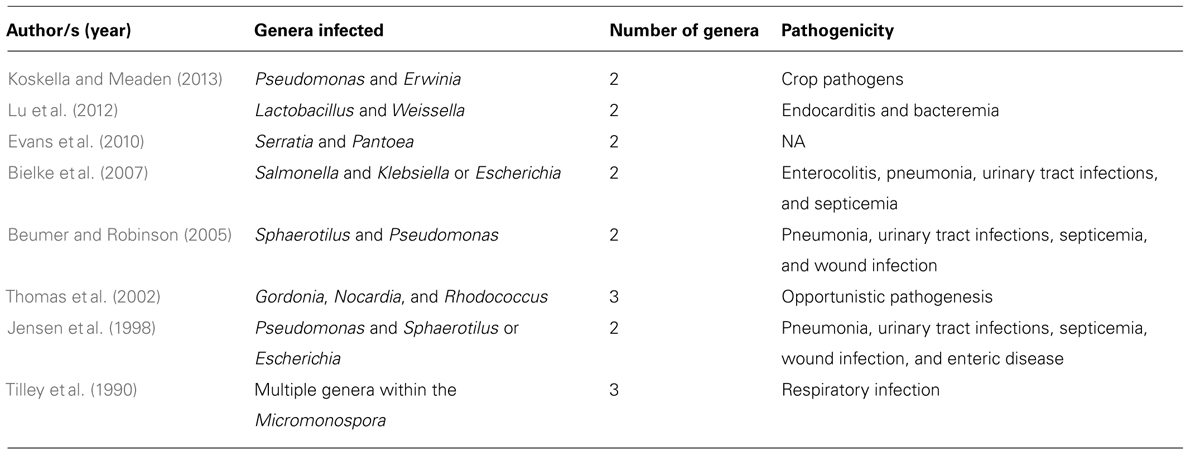
Currently the maximum breadth of bacterial resistance to phage (i.e., the number of phage types a single bacterium is capable of resisting) remains largely unknown, as novel genera of phages are continually being discovered (Holmfeldt et al., 2013). For example, the ubiquitous marine bacterial clade SAR116 was thought to be so abundant as a result of escaping phage predation, but a recent finding shows that it is indeed infected by phages, and that these phages are likely to be the most abundant species on the planet (Kang et al., 2013). Our knowledge of phage ecology and evolution is still in its infancy; the exact mechanisms of infection, and in turn resistance, are often unknown and could be simultaneously diverse among strains and yet largely conserved across genera (Koskella and Meaden, 2013). There are a number of published cases of phages that are capable of infecting bacterial hosts across genera (Table 1), suggesting the potential for shared resistance mechanisms. Even if unlikely, evolved resistance to the few phage therapy products available to clinicians would severely impair treatment potential. This problem may be exacerbated by the more stringent control of phage products for clinical use, and thus the slow pipeline from isolation to delivery, relative to the approval of cocktails for use in agriculture. As such, rapidly responding regulation, like the measures in place for seasonal flu vaccines (Verbeken et al., 2012), could be a more effective way of countering phage therapy product redundancy.
Finally, the combined use of phages and antibiotics has shown great promise due to the negative pleiotropic effects of phage resistance and antibiotic resistance. Experimental evolution has demonstrated that phages applied to populations of Pseudomonas fluorescens that had evolved antibiotic resistance reduced population densities to a greater degree than when applied to sensitive strains (Escobar-Páramo et al., 2012). In addition, combined treatment was shown to drastically hinder the evolution of bacterial resistance over time compared to antibiotic treatment alone (Zhang and Buckling, 2012). In poultry, the combination of phages and enrofloxacin resulted in lower mortality in infected birds than either treatment individually (Huff et al., 2004). Therefore, one potential step forward in controlling the spread of both antibiotic resistance and phage resistance in the environment and/or under clinical settings could be the carefully planned combination treatments of the two.
Phage-Mediated Attenuation of Bacterial Virulence
A common refrain of phage therapy and biocontrol is that even if resistance does emerge, such resistance is likely to be costly, and as such would attenuate bacterial virulence in a eukaryotic host (Inal, 2003; Hagens et al., 2004). Phage resistance does seem to be correlated with a reduction in metabolic fitness both in the lab (Bohannan and Lenski, 2000; Koskella et al., 2012) and the soil (Gómez and Buckling, 2011), however the effect this may have on virulence in a eukaryotic host is largely unknown. Of the few examples, phage-resistant strains of Yersinia pestis have been shown to have attenuated virulence in a mouse model system, resulting in a significant increase in time to death, and in some cases complete attenuation (Filippov et al., 2011). The same phenomenon has been observed in aquaculture with a direct correlation observed between phage resistance and complete attenuation of F. columnare in a zebrafish system, such that one phage-sensitive phenotype resulted in 100% mortality compared to 0% in the phage-resistant phenotypes (Laanto et al., 2012).
In plant pathogens, there is also evidence to suggest that phage-mediated selection might alter the infectivity and/or virulence of bacterial pathogens. Phages infecting the bacterium, Erwinia amylovora, were found to be most efficient at infecting strains that produced either high or low levels of exopolysaccharides (depending on the phage family examined), suggesting strong and context-dependent selection on a trait that is also known to play a key role in virulence on the plant host (Roach et al., 2013). Furthermore, Hosseinidoust et al. (2013) found that in tissue culture, phage-resistant variants of Pseudomonas aeruginosa actually secrete higher levels of virulence factors and caused more damage to cultured mammalian cells. With no general pattern yet emerging, phage-mediated attenuation of virulence seems hard to predict and certainly not guaranteed. It is possible that a bacterial pathogen could evolve resistance to a phage therapy product and maintain or even attain high virulence levels. Furthermore, the role of compensatory mutations to phage resistance is unknown; in the case of antibiotics such mutations can rapidly ameliorate the costs paid for resistance (Levin et al., 2000; Brandis and Hughes, 2013) and the same may be true of resistance to phages. If this turns out to be the case, phage therapy treatments that rely on the loss of costly resistance (or interactions among costly mutations to multiple phages) will be at constant risk of bacterial escape.
Increasing Horizontal Gene Flow Through Phage Application
In addition to the direct effects of phage application on the densities and relative frequencies of bacterial pathogens, we must also be aware of the potential dangers of phage-mediated horizontal gene transfer (HGT) among pathogenic and non-pathogenic bacterial species. This is particularly relevant as HGT is an important driver of antibiotic resistance evolution (Courvalin, 1994). Given that phages facilitate horizontal gene flow through the process of transduction and that beta-lactam resistance genes have been isolated from environmental phage genomes (Colomer-Lluch et al., 2011), there is clear need to be cautious in our application of phages in the environment. Moreover it has recently been shown that antibiotic treatment itself expands the number of genes that confer drug resistance in phage metagenomes (Modi et al., 2013). Environmental perturbation with antibiotics also expanded the ecological network of phages, suggesting that drug treatment selects for greater phage-mediated transfer of resistance genes (Modi et al., 2013). Finally, antibiotic treatment of swine increased the induction of prophage – another key mechanism of HGT (Allen et al., 2011).
Phage-mediated transfer of virulence factors is also a key concern, and has been well characterized in the Vibrio cholerae system whereby CTX Φ phage (among others) shapes the severity of cholera pandemics through the transfer of toxin producing virulence factors and environmental fitness benefits (Faruque and Mekalanos, 2012). Additionally, V. cholerae has been shown to become naturally competent on a chitin surface (similar to its environmental niche on crustacean exoskeletons), facilitating uptake of exogenous DNA from an array of sources (Meibom et al., 2005), which could be important if phages are lysing nearby pathogenic bacterial strains. Environmental perturbation through unnaturally high titers of phages could lead to high levels of transduction and horizontal gene flow with unintended outcomes, exacerbating antibiotic resistance and moving virulence factors and toxins among genomes. However, a better understanding of phage host range and host range expansion could help mitigate the spread of genes among bacterial hosts.
Impact of Phage Application on Natural Microbial Communities
The importance of the microbial flora to the fitness of human hosts has become clear in recent years, with microbes playing a proposed role in obesity (Greenblum et al., 2012), oral health (Belda-Ferre et al., 2012), Crohn’s disease (Manichanh et al., 2006), AIDS (Saxena et al., 2012), and even mental health (Foster and McVey Neufeld, 2013). Although less well studied, a similar role of microbiota in shaping fitness is likely to be true for agricultural plants (Peiffer and Ley, 2013) and livestock (McFall-Ngai et al., 2013). Whilst antibiotics can disrupt a large proportion of the microbial community (Dethlefsen and Relman, 2011), phage therapy may facilitate the targeted elimination of a pathogenic strain without disruption of the normal microbiota of a patient (Carlton, 1999) or a crop plant. Indeed, this argument has been a focal one to support phage therapy over antibiotic use (Loc-Carrillo and Abedon, 2011). The term “dysbiosis” has been introduced to describe a microbial flora that has become “unhealthy,” typically in human disease (Tamboli, 2004). The same phenomenon is likely to hold true for natural microbial communities both within and outside of the host environments, and thus the addition of foreign phages in the form of a biopesticide could destabilize such communities, causing “dysbiosis” and potentially having subsequent effects on disease and nutrient cycling. For example, in soil where ratios of phages to bacteria are expected to be near 1:1 (Reyes et al., 2010) an influx of applied phages could well disrupt stable ecological communities important in nutrient cycling. Conversely, in the marine environment, where this ratio is closer to 1:100 (Reyes et al., 2010), an influx of applied phages is unlikely to adversely disrupt a normal microbial community. Unfortunately, by the same logic, aquaculture may suffer from lower chances of success from phage biocontrol, as the concentrations of phages needed to influence bacterial density is likely to be a limiting factor.
Unpredictability of Infection Kinetics
Unlike antibiotic usage, where an effective dosage can be determined for different species and corresponding quantities used, the infection kinetics of phage therapy are less predictable (Payne and Jansen, 2003). This knowledge gap presents a challenge for effective use balanced with environmental safety, as an individual phage persists in the environment for relatively short timescales, but a lineage can reproduce indefinitely when hosts are plentiful. This continued replication makes calculations of dosage difficult. In antibiotic treatment the minimum inhibitory concentration (MIC) is crucial to informing proper antibiotic dosages; however, finding the right balance between a high enough titer of phages to be effective and not introducing excessive levels into the environment is difficult. Potentially the application of a single phage could lead to the continued replication of an infective phage, thus perpetuating not only treatment but also persistence in the environment. If there are unwanted effects of phage biopesticide there are no tools currently available to selectively remove such viruses from the environment. Also, the timing of treatment as a function of bacterial density can be crucial for a successful outcome (Payne and Jansen, 2003). Indeed, in a trial controlling V. parahaemolyticus infections in shrimp timing was crucial whereas changes in dosage had no effect. Reducing the multiplicity of infection made no difference whereas when treatment was delayed it was ineffective in controlling mortality (Martínez-Díaz and Hipólito-Morales, 2013), possibly as a result of non-linear infection kinetics. Such infection kinetics of phage therapy were tested in vitro with Campylobacter jejuni, a common poultry pathogen, and found to fit a non-linear model with a density-dependent proliferation threshold (Cairns et al., 2009). These studies highlight the nuances involved in effective phage therapy use and as such the use of phages cannot be analogous to that of antibiotics. Provided policy makers accept this and approach the field balancing the risks of widespread environmental use with the pressing need for a clinical alternative to antibiotics, phage biocontrol could be an integral tool in controlling bacterial diseases.
Future Outlook
Our understanding of the biology, ecology, and evolution of microbial pathogens has improved immeasurably since the advent of widespread antibiotic use. If we can learn the lessons from our mistakes with antibiotic use, phage therapy and biocontrol could form an integral tool in the fight against bacterial infections that threaten human health and food production (Pirnay et al., 2012; Allen et al., 2013). For example, the falling costs of whole genome sequencing (Kisand and Lettieri, 2013) should make tracking the evolution and spread of resistance genes in a clinical setting easier and more accurate (Didelot et al., 2012). Furthermore, advances in metagenomics may make monitoring the effects of environmental perturbations on microbial communities feasible and allow researchers to track changes over long-time periods. An attractive avenue of research for pharmaceutical companies may be the use of phage lysins – enzymes that are capable of bursting bacterial cells open “from without.” Such an approach avoids the problems of infection kinetics mentioned previously and can have a broad-spectrum encompassing multiple strains of antibiotic resistant pathogens, such as MRSA and vancomycin intermediate Staphylococcus aureus (Gilmer et al., 2013). The downside is that lysins, unlike phages, lack the ability to counter evolve to pathogens.
Microbial biofilms present a continued risk to healthcare as they may harbor bacteria in a less metabolically active state that survive antibiotic treatment (Oliver, 2010). Phages targeting biofilms in synchrony with antibiotics may form a novel strategy, although the inherent risks of HGT still remain. It may also be possible to circumvent this cycle of treatment, selection for resistance and re-infection through the use of “social disruption” treatments that reduce bacterial virulence without selecting for resistance (Boyle et al., 2013). Phages could play a role in the reduction of biofilm and public good production – one example is an engineered phage that expresses a biofilm-degrading enzyme (Lu and Collins, 2007). Whilst this is likely to reduce the fitness of bacterial populations, selection should be weaker than that of an antibiotic. Given the difficulties faced by clinicians in treating antibiotic-resistant infections and the urgency of finding alternative therapies, the prudent use of phages should be a priority. Nevertheless we have the tools to track resistance and simple measures such as providing a diverse set of phages for treatment could help. A seasonal-vaccine-like scheme could create a treatment program that is responsive to the evolution of resistance. In short, a very different model to that of our use of antibiotics.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
Allen, H. K., Levine, U. Y., Looft, T., Bandrick, M., and Casey, T. A. (2013). Treatment, promotion, commotion: antibiotic alternatives in food-producing animals. Trends Microbiol. 21, 114–119. doi: 10.1016/j.tim.2012.11.001
Allen, H. K., Looft, T., Bayles, D. O., Humphrey, S., Levine, U. Y., Alt, D., et al. (2011). Antibiotics in feed induce prophages in swine fecal microbiomes. mBio 2, e00260-11. doi: 10.1128/mBio.00260-11
Balogh, B., Canteros, B. I., Stall, R. E., and Jones, J. B. (2008). Control of citrus canker and citrus bacterial spot with bacteriophages. Plant Dis. 92, 1048–1052. doi: 10.1094/PDIS-92-7-1048
Belda-Ferre, P., Alcaraz, L. D., Cabrera-Rubio, R., Romero, H., Simón-Soro, A., Pignatelli, M., et al. (2012). The oral metagenome in health and disease. ISME J. 6, 46–56. doi: 10.1038/ismej.2011.85
Beumer, A., and Robinson, J. B. (2005). A broad-host-range, generalized transducing phage (SN-T) acquires 16S rRNA genes from different genera of bacteria. Appl. Environ. Microbiol. 71, 8301–8304. doi: 10.1128/AEM.71.12.8301-8304.2005
Bielke, L., Higgins, S., Donoghue, A., Donoghue, D., and Hargis, B. M. (2007). Salmonella host range of bacteriophages that infect multiple genera. Poult. Sci. 86, 2536–2540. doi: 10.3382/ps.2007-00250
Bohannan, B. J. M., and Lenski, R. E. (2000). Linking genetic change to community evolution: insights from studies of bacteria and bacteriophage. Ecol. Lett. 3, 362–377. doi: 10.1046/j.1461-0248.2000.00161.x
Boyle, K. E., Heilmann, S., van Ditmarsch, D., and Xavier, J. B. (2013). Exploiting social evolution in biofilms. Curr. Opin. Microbiol. 16, 207–212. doi: 10.1016/j.mib.2013.01.003
Brandis, G., and Hughes, D. (2013). Genetic characterization of compensatory evolution in strains carrying rpoB Ser531Leu, the rifampicin resistance mutation most frequently found in clinical isolates. J. Antimicrob. Chemother. 68, 2493–2497. doi: 10.1093/jac/dkt224
Breitbart, M., and Rohwer, F. (2005). Here a virus, there a virus, everywhere the same virus? Trends Microbiol. 13, 278–284. doi: 10.1016/j.tim.2005.04.003
Brüssow, H. (2012). What is needed for phage therapy to become a reality in Western medicine? Virology 434, 138–142. doi: 10.1016/j.virol.2012.09.015
Buckling, A., and Rainey, P. B. (2002). Antagonistic coevolution between a bacterium and a bacteriophage. Proc. Biol. Sci. 269, 931–936. doi: 10.1098/rspb.2001.1945
Cairns, B. J., Timms, A. R., Jansen, V. A. A., Connerton, I. F., and Payne, R. J. H. (2009). Quantitative models of in vitro bacteriophage–host dynamics and their application to phage therapy. PLoS Pathog. 5:e1000253. doi: 10.1371/journal.ppat.1000253
Carlton, R. M. (1999). Phage therapy: past history and future prospects. Arch. Immunol. Ther. Exp. 47, 267–274.
Colomer-Lluch, M., Jofre, J., and Muniesa, M. (2011). Antibiotic resistance genes in the bacteriophage DNA fraction of environmental samples. PLoS ONE 6:e17549. doi: 10.1371/journal.pone.0017549
Courvalin, P. (1994). Transfer of antibiotic resistance genes between gram-positive and gram-negative bacteria. Antimicrob. Agents Chemother. 38, 1447–1451. doi: 10.1128/AAC.38.7.1447
D’Costa, V. M., King, C. E., Kalan, L., Morar, M., Sung, W. W. L., Schwarz, C., et al. (2011). Antibiotic resistance is ancient. Nature 477, 457–461. doi: 10.1038/nature10388
Dethlefsen, L., and Relman, D. A. (2011). Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc. Natl. Acad. Sci. U.S.A. 108(Suppl.), 4554–4561. doi: 10.1073/pnas.1000087107
Didelot, X., Bowden, R., Wilson, D. J., Peto, T. E. A., and Crook, D. W. (2012). Transforming clinical microbiology with bacterial genome sequencing. Nat. Rev. Genet. 13, 601–612. doi: 10.1038/nrg3226
Ding, C., and He, J. (2010). Effect of antibiotics in the environment on microbial populations. Appl. Microbiol. Biotechnol. 87, 925–941. doi: 10.1007/s00253-010-2649-5
Escobar-Páramo, P., Gougat-Barbera, C., and Hochberg, M. E. (2012). Evolutionary dynamics of separate and combined exposure of Pseudomonas fluorescens SBW25 to antibiotics and bacteriophage. Evol. Appl. 5, 583–592. doi: 10.1111/j.1752-4571.2012.00248.x
Evans, T. J., Crow, M. A., Williamson, N. R., Orme, W., Thomson, N. R., Komitopoulou, E., et al. (2010). Characterization of a broad-host-range flagellum-dependent phage that mediates high-efficiency generalized transduction in, and between, Serratia and Pantoea. Microbiology 156, 240–247. doi: 10.1099/mic.0.032797-0
Faruque, S. M., and Mekalanos, J. J. (2012). Phage–bacterial interactions in the evolution of toxigenic Vibrio cholerae. Virulence 3, 556–565. doi: 10.4161/viru.22351
Filippov, A. A., Sergueev, K. V., He, Y., Huang, X.-Z., Gnade, B. T., Mueller, A. J., et al. (2011). Bacteriophage-resistant mutants in Yersinia pestis: identification of phage receptors and attenuation for mice. PLoS ONE 6:e25486. doi: 10.1371/journal.pone.0025486
Flores, C. O., Meyer, J. R., Valverde, S., Farr, L., and Weitz, J. S. (2011). Statistical structure of host–phage interactions. Proc. Natl. Acad. Sci. U.S.A. 108, E288–E297. doi: 10.1073/pnas.1101595108
Food and Agriculture Organization [FAO]. (2012). The State of World Fisheries and Aquaculture - 2012 (SOFIA). Available at: http://www.fao.org/docrep/016/i2727e/i2727e00.htm (accessed August 30, 2013).
Food and Drug Administration [FDA]. (2013). Center for Food Safety and Applied Nutrition GRAS Notice Inventory – Agency Response Letter GRAS Notice No. GRN 000435. Available at: http://www.fda.gov/Food/IngredientsPackagingLabeling/GRAS/NoticeInventory/ucm345473.htm
Foster, J. A., and McVey Neufeld, K.-A. (2013). Gut–brain axis: how the microbiome influences anxiety and depression. Trends Neurosci. 36, 305–312. doi: 10.1016/j.tins.2013.01.005
Frampton, R. A., Pitman, A. R., and Fineran, P. C. (2012). Advances in bacteriophage-mediated control of plant pathogens. Int. J. Microbiol. 2012, 326452. doi: 10.1155/2012/326452
Gill, J. J., and Hyman, P. (2010). Phage choice, isolation, and preparation for phage therapy. Curr. Pharm. Biotechnol. 11, 2–14. doi: 10.2174/138920110790725311
Gilmer, D. B., Schmitz, J. E., Euler, C. W., and Fischetti, V. A. (2013). Novel bacteriophage lysin with broad lytic activity protects against mixed infection by Streptococcus pyogenes and methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 57, 2743–2750. doi: 10.1128/AAC.02526-12
Gómez, P., and Buckling, A. (2011). Bacteria-phage antagonistic coevolution in soil. Science 332, 106–109. doi: 10.1126/science.1198767
Graveland, H., Wagenaar, J. A., Heesterbeek, H., Mevius, D., van Duijkeren, E., and Heederik, D. (2010). Methicillin resistant Staphylococcus aureus ST398 in veal calf farming: human MRSA carriage related with animal antimicrobial usage and farm hygiene. PLoS ONE 5:e10990. doi: 10.1371/journal.pone.0010990
Greenblum, S., Turnbaugh, P. J., and Borenstein, E. (2012). Metagenomic systems biology of the human gut microbiome reveals topological shifts associated with obesity and inflammatory bowel disease. Proc. Natl. Acad. Sci. U.S.A. 109, 594–599. doi: 10.1073/pnas.1116053109
Hagens, S., Habel, A., von Ahsen, U., von Gabain, A., and Bläsi, U. (2004). Therapy of experimental pseudomonas infections with a nonreplicating genetically modified phage. Antimicrob. Agents Chemother. 48, 3817–3822. doi: 10.1128/AAC.48.10.3817-3822.2004
Hall, A. R., De Vos, D., Friman, V.-P., Pirnay, J.-P., and Buckling, A. (2012). Effects of sequential and simultaneous applications of bacteriophages on populations of Pseudomonas aeruginosa in vitro and in wax moth larvae. Appl. Environ. Microbiol. 78, 5646–5652. doi: 10.1128/AEM.00757-12
Heuer, H., Schmitt, H., and Smalla, K. (2011). Antibiotic resistance gene spread due to manure application on agricultural fields. Curr. Opin. Microbiol. 14, 236–243. doi: 10.1016/j.mib.2011.04.009
Holmfeldt, K., Solonenko, N., Shah, M., Corrier, K., Riemann, L., Verberkmoes, N. C., et al. (2013). Twelve previously unknown phage genera are ubiquitous in global oceans. Proc. Natl. Acad. Sci. U.S.A. 110, 12798–12803. doi: 10.1073/pnas.1305956110
Hosseinidoust, Z., Van De Ven, T. G. M., and Tufenkji, N. (2013). Evolution of Pseudomonas aeruginosa virulence as a result of phage predation. Appl. Environ. Microbiol. 79, 6110–6116. doi: 10.1128/AEM.01421-13
Huff, W., Huff, G., Rath, N., Balog, J., and Donoghue, A. (2004). Therapeutic efficacy of bacteriophage and Baytril (enrofloxacin) individually and in combination to treat colibacillosis in broilers. Poult. Sci. 83, 1944–1947.
Hyman, P., and Abedon, S. T. (2010). Bacteriophage host range and bacterial resistance. Adv. Appl. Microbiol. 70, 217–248. doi: 10.1016/S0065-2164(10)70007-1
Inal, J. M. (2003). Phage therapy: a reappraisal of bacteriophages as antibiotics. Arch. Immunol. Ther. Exp. 51, 237–244.
Jensen, E. C., Schrader, H. S., Rieland, B., Thompson, T. L., Lee, K. W., Nickerson, K. W., et al. (1998). Prevalence of broad-host-range lytic bacteriophages of Sphaerotilus natans, Escherichia coli, and Pseudomonas aeruginosa. Appl. Environ. Microbiol. 64, 575–580.
Jones, J. B., Vallad, G. E., Iriarte, F. B., Obradović, A., Wernsing, M. H., Jackson, L. E., et al. (2012). Considerations for using bacteriophages for plant disease control. Bacteriophage 2, 208–214. doi: 10.4161/bact.23857
Kåhrström, C. T. (2013). Entering a post-antibiotic era? Nat. Rev. Microbiol. 11, 146–146. doi: 10.1038/nrmicro2983
Kang, I., Oh, H.-M., Kang, D., and Cho, J.-C. (2013). Genome of a SAR116 bacteriophage shows the prevalence of this phage type in the oceans. Proc. Natl. Acad. Sci. U.S.A. 110, 12343–12348. doi: 10.1073/pnas.1219930110
Khanna, T., Friendship, R., Dewey, C., and Weese, J. S. (2008). Methicillin resistant Staphylococcus aureus colonization in pigs and pig farmers. Vet. Microbiol. 128, 298–303. doi: 10.1016/j.vetmic.2007.10.006
Kim, J. H., Gomez, D. K., Nakai, T., and Park, S. C. (2010). Isolation and identification of bacteriophages infecting ayu Plecoglossus altivelis altivelis specific Flavobacterium psychrophilum. Vet. Microbiol. 140, 109–115. doi: 10.1016/j.vetmic.2009.07.002
Kisand, V., and Lettieri, T. (2013). Genome sequencing of bacteria: sequencing, de novo assembly and rapid analysis using open source tools. BMC Genomics 14:211. doi: 10.1186/1471-2164-14-211
Koskella, B. (2013). Phage-mediated selection on microbiota of a long-lived host. Curr. Biol. 23, 1256–1260. doi: 10.1016/j.cub.2013.05.038
Koskella, B., Lin, D. M., Buckling, A., and Thompson, J. N. (2012). The costs of evolving resistance in heterogeneous parasite environments. Proc. Biol. Sci. 279, 1896–1903. doi: 10.1098/rspb.2011.2259
Koskella, B., and Meaden, S. (2013). Understanding bacteriophage specificity in natural microbial communities. Viruses 5, 806–823. doi: 10.3390/v5030806
Koskella, B., Thompson, J. N., Preston, G. M., and Buckling, A. (2011). Local biotic environment shapes the spatial scale of bacteriophage adaptation to bacteria. Am. Nat. 177, 440–451. doi: 10.1086/658991
Kunin, V., He, S., Warnecke, F., Peterson, S. B., Garcia Martin, H., Haynes, M., et al. (2008). A bacterial metapopulation adapts locally to phage predation despite global dispersal. Genome Res. 18, 293–297. doi: 10.1101/gr.6835308
Kutter, E., De Vos, D., Gvasalia, G., Alavidze, Z., Gogokhia, L., Kuhl, S., et al. (2010). Phage therapy in clinical practice: treatment of human infections. Curr. Pharm. Biotechnol. 11, 69–86. doi: 10.2174/138920110790725401
Laanto, E., Bamford, J. K. H., Laakso, J., and Sundberg, L.-R. (2012). Phage-driven loss of virulence in a fish pathogenic bacterium. PLoS ONE 7:e53157. doi: 10.1371/journal.pone.0053157
Labella, A., Gennari, M., Ghidini, V., Trento, I., Manfrin, A., Borrego, J. J., et al. (2013). High incidence of antibiotic multi-resistant bacteria in coastal areas dedicated to fish farming. Mar. Pollut. Bull. 70, 197–203. doi: 10.1016/j.marpolbul.2013.02.037
Lang, J. M., Gent, D. H., and Schwartz, H. F. (2007). Management of Xanthomonas leaf blight of onion with bacteriophages and a plant activator. Plant Dis. 91, 871–878. doi: 10.1094/PDIS-91-7-0871
Levin, B. R., and Bull, J. J. (2004). Population and evolutionary dynamics of phage therapy. Nat. Rev. Microbiol. 2, 166–173. doi: 10.1038/nrmicro822
Levin, B. R., Perrot, V., and Walker, N. (2000). Compensatory mutations, antibiotic resistance and the population genetics of adaptive evolution in bacteria. Genetics 154, 985–997.
Levy, S. B., and Marshall, B. (2004). Antibacterial resistance worldwide: causes, challenges and responses. Nat. Med. 12, 122–129. doi: 10.1038/nm1145
Loc-Carrillo, C., and Abedon, S. T. (2011). Pros and cons of phage therapy. Bacteriophage 1, 111–114. doi: 10.4161/bact.1.2.14590
Lu, T. K., and Collins, J. J. (2007). Dispersing biofilms with engineered enzymatic bacteriophage. Proc. Natl. Acad. Sci. U.S.A. 104, 11197–11202. doi: 10.1073/pnas.0704624104
Lu, Z., Pérez-Díaz, I. M., Hayes, J. S., and Breidt, F. (2012). Bacteriophage ecology in a commercial cucumber fermentation. Appl. Environ. Microbiol. 78, 8571–8578. doi: 10.1128/AEM.01914-12
Malnoy, M., Martens, S., Norelli, J. L., Barny, M.-A., Sundin, G. W., Smits, T. H. M., et al. (2012). Fire blight: applied genomic insights of the pathogen and host. Annu. Rev. Phytopathol. 50, 475–494. doi: 10.1146/annurev-phyto-081211-172931
Manichanh, C., Rigottier-Gois, L., Bonnaud, E., Gloux, K., Pelletier, E., Frangeul, L., et al. (2006). Reduced diversity of faecal microbiota in Crohn’s disease revealed by a metagenomic approach. Gut 55, 205–211. doi: 10.1136/gut.2005.073817
Martínez, J. L. (2008). Antibiotics and antibiotic resistance genes in natural environments. Science 321, 365–367. doi: 10.1126/science.1159483
Martínez-Díaz, S. F., and Hipólito-Morales, A. (2013). Efficacy of phage therapy to prevent mortality during the vibriosis of brine shrimp. Aquaculture 400–401, 120–124. doi: 10.1016/j.aquaculture.2013.03.007
McFall-Ngai, M., Hadfield, M. G., Bosch, T. C. G., Carey, H. V., Domazet-Lošo, T., Douglas, A. E., et al. (2013). Animals in a bacterial world, a new imperative for the life sciences. Proc. Natl. Acad. Sci. U.S.A. 110, 3229–3236. doi: 10.1073/pnas.1218525110
McManus, P. S., Stockwell, V. O., Sundin, G. W., and Jones, A. L. (2002). Antibiotic use in plant agriculture. Annu. Rev. Phytopathol. 40, 443–465. doi: 10.1146/annurev.phyto.40.120301.093927
Meibom, K. L., Blokesch, M., Dolganov, N. A., Wu, C.-Y., and Schoolnik, G. K. (2005). Chitin induces natural competence in Vibrio cholerae. Science 310, 1824–1827. doi: 10.1126/science.1120096
Merabishvili, M., Pirnay, J.-P., Verbeken, G., Chanishvili, N., Tediashvili, M., Lashkhi, N., et al. (2009). Quality-controlled small-scale production of a well-defined bacteriophage cocktail for use in human clinical trials. PLoS ONE 4:e4944. doi: 10.1371/journal.pone.0004944
Micallef, S. A., Rosenberg Goldstein, R. E., George, A., Ewing, L., Tall, B. D., Boyer, M. S., et al. (2013). Diversity, distribution and antibiotic resistance of Enterococcus spp. recovered from tomatoes, leaves, water and soil on U.S. Mid-Atlantic farms. Food Microbiol. 36, 465–474. doi: 10.1016/j.fm.2013.04.016
Modi, S. R., Lee, H. H., Spina, C. S., and Collins, J. J. (2013). Antibiotic treatment expands the resistance reservoir and ecological network of the phage metagenome. Nature 499, 219–222. doi: 10.1038/nature12212
Oliver, J. D. (2010). Recent findings on the viable but nonculturable state in pathogenic bacteria. FEMS Microbiol. Rev. 34, 415–425.
Ormälä, A.-M., and Jalasvuori, M. (2013). Phage therapy: should bacterial resistance to phages be a concern, even in the long run? Bacteriophage 3, e24219. doi: 10.4161/bact.24219
Payne, R. J. H., and Jansen, V. A. A. (2003). Pharmacokinetic principles of bacteriophage therapy. Clin. Pharmacokinet. 42, 315–325. doi: 10.2165/00003088-200342040-00002
Peiffer, J. A., and Ley, R. E. (2013). Exploring the maize rhizosphere microbiome in the field: a glimpse into a highly complex system. Commun. Integr. Biol. 6, e25177. doi: 10.4161/cib.25177
Perry, J. A., and Wright, G. D. (2013). The antibiotic resistance “mobilome”: searching for the link between environment and clinic. Front. Microbiol. 4:138. doi: 10.3389/fmicb.2013.00138
Pirnay, J.-P., De Vos, D., Verbeken, G., Merabishvili, M., Chanishvili, N., Vaneechoutte, M., et al. (2011). The phage therapy paradigm: prêt-à-porter or sur-mesure? Pharm. Res. 28, 934–937. doi: 10.1007/s11095-010-0313-5
Pirnay, J.-P., Verbeken, G., Rose, T., Jennes, S., Zizi, M., Huys, I., et al. (2012). Introducing yesterday’s phage therapy in today’s medicine. Future Virol. 7, 379–390. doi: 10.2217/fvl.12.24
Popowska, M., Rzeczycka, M., Miernik, A., Krawczyk-Balska, A., Walsh, F., and Duffy, B. (2012). Influence of soil use on prevalence of tetracycline, streptomycin, and erythromycin resistance and associated resistance genes. Antimicrob. Agents Chemother. 56, 1434–1443. doi: 10.1128/AAC.05766-11
Price, L. B., Stegger, M., Hasman, H., Aziz, M., Larsen, J., Andersen, P. S., et al. (2012). Staphylococcus aureus CC398: host adaptation and emergence of methicillin resistance in livestock. mBio 3, e00305-11. doi: 10.1128/mBio.00305-11
Reyes, A., Haynes, M., Hanson, N., Angly, F. E., Heath, A. C., Rohwer, F., et al. (2010). Viruses in the faecal microbiota of monozygotic twins and their mothers. Nature 466, 334–338. doi: 10.1038/nature09199
Roach, D. R., Sjaarda, D. R., Castle, A. J., and Svircev, A. M. (2013). Host exopolysaccharide quantity and composition impact Erwinia amylovora bacteriophage pathogenesis. Appl. Environ. Microbiol. 79, 3249–3256. doi: 10.1128/AEM.00067-13
Sarker, S. A., McCallin, S., Barretto, C., Berger, B., Pittet, A.-C., Sultana, S., et al. (2012). Oral T4-like phage cocktail application to healthy adult volunteers from Bangladesh. Virology 434, 222–232. doi: 10.1016/j.virol.2012.09.002
Saxena, D., Li, Y., Yang, L., Pei, Z., Poles, M., Abrams, W. R., et al. (2012). Human microbiome and HIV/AIDS. Curr. HIV/AIDS Rep. 9, 44–51. doi: 10.1007/s11904-011-0103-7
Singer, R. S., Finch, R., Wegener, H. C., Bywater, R., Walters, J., and Lipsitch, M. (2003). Antibiotic resistance – the interplay between antibiotic use in animals and human beings. Lancet Infect. Dis. 3, 47–51. doi: 10.1016/S1473-3099(03)00490-0
Smith, T. C., Male, M. J., Harper, A. L., Kroeger, J. S., Tinkler, G. P., Moritz, E. D., et al. (2009). Methicillin-resistant Staphylococcus aureus (MRSA) strain ST398 is present in Midwestern U.S. swine and swine workers. PLoS ONE 4:e4258. doi: 10.1371/journal.pone.0004258
Stone, R. (2002). Bacteriophage therapy. Food and agriculture: testing grounds for phage therapy. Science 298, 730. doi: 10.1126/science.298.5594.730
Svircev, A. M., Lehman, S. M., Sholberg, P., Roach, D., and Castle, A. J. (2011). “Phage biopesticides and soil bacteria: multilayered and complex interactions,” in Soil Biology, Vol. 23, Biocommunication in Soil Microorganisms, ed. G. Witzany (Berlin: Springer), 215–236.
Tamboli, C. P. (2004). Dysbiosis in inflammatory bowel disease. Gut 53, 1–4. doi: 10.1136/gut.53.1.1
Thomas, J. A., Soddell, J. A., and Kurtböke, D. I. (2002). Fighting foam with phages? Water Sci. Technol. 46, 511–518.
Tilley, B. C., Meyertons, J. L., and Lechevalier, M. P. (1990). Characterization of a temperate actinophage, MPphiWR-1, capable of infecting Micromonospora purpurea ATCC15835. J. Ind. Microbiol. 5, 167–182. doi: 10.1007/BF01573867
Traugott, K. A., Echevarria, K., Maxwell, P., Green, K., and Lewis, J. S. (2011). Monotherapy or combination therapy? The Pseudomonas aeruginosa conundrum. Pharmacotherapy 31, 598–608. doi: 10.1592/phco.31.6.598
van Cleef, B. A., Verkade, E. J. M., Wulf, M. W., Buiting, A. G., Voss, A., Huijsdens, X. W., et al. (2010). Prevalence of livestock-associated MRSA in communities with high pig-densities in The Netherlands. PLoS ONE 5:e9385. doi: 10.1371/journal.pone.0009385
Vardakas, K. Z., Tansarli, G. S., Bliziotis, I. A., and Falagas, M. E. (2013). β-Lactam plus aminoglycoside or fluoroquinolone combination versus β-lactam monotherapy for Pseudomonas aeruginosa infections: a meta-analysis. Int. J. Antimicrob. Agents 41, 301–310. doi: 10.1016/j.ijantimicag.2012.12.006
Verbeken, G., Pirnay, J.-P., De Vos, D., Jennes, S., Zizi, M., Lavigne, R., et al. (2012). Optimizing the European regulatory framework for sustainable bacteriophage therapy in human medicine. Arch. Immunol. Ther. Exp. 60, 161–172. doi: 10.1007/s00005-012-0175-0
Vos, M., Birkett, P. J., Birch, E., Griffiths, R. I., and Buckling, A. (2009). Local adaptation of bacteriophages to their bacterial hosts in soil. Science 325, 833. doi: 10.1126/science.1174173
Voss, A., Loeffen, F., Bakker, J., Klaassen, C., and Wulf, M. (2005). Methicillin-resistant Staphylococcus aureus in pig farming. Emerg. Infect. Dis. 11, 1965–1966. doi: 10.3201/eid1112.050428
Witte, W. (1998). BIOMEDICINE: medical consequences of antibiotic use in agriculture. Science 279, 996–997. doi: 10.1126/science.279.5353.996
Wright, A., Hawkins, C. H., Anggård, E. E., and Harper, D. R. (2009). A controlled clinical trial of a therapeutic bacteriophage preparation in chronic otitis due to antibiotic-resistant Pseudomonas aeruginosa; a preliminary report of efficacy. Clin. Otolaryngol. 34, 349–357. doi: 10.1111/j.1749-4486.2009.01973.x
Zhang, Q.-G., and Buckling, A. (2012). Phages limit the evolution of bacterial antibiotic resistance in experimental microcosms. Evol. Appl. 5, 575–582. doi: 10.1111/j.1752-4571.2011.00236.x
Keywords: phage therapy, antibiotic resistance, coevolution, phage resistance, microbial communities
Citation: Meaden S and Koskella B (2013) Exploring the risks of phage application in the environment. Front. Microbiol. 4:358. doi: 10.3389/fmicb.2013.00358
Received: 06 September 2013; Accepted: 12 November 2013;
Published online: 29 November 2013.
Edited by:
Heather K. Allen, National Animal Disease Center, USAReviewed by:
Andres Moya, University of Valencia, SpainNatalia Ivanova, Lawrence Berkeley National Laboratory, USA
Copyright © 2013 Meaden and Koskella. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sean Meaden, College of Life and Environmental Sciences, University of Exeter, Penryn Campus, Treliever Road, Penryn, TR10 9EZ Penryn, UK e-mail:c20zNDFAZXhldGVyLmFjLnVr
 Meaden Sean
Meaden Sean Britt Koskella
Britt Koskella