Abstract
Giardia duodenalis is a protozoan parasite responsible for giardiasis, a disease characterized by intestinal malabsorption, diarrhea and abdominal pain in a large number of mammal species. Giardiasis is one of the most common intestinal parasitic diseases in the world and thus a high veterinary, and public health concern. It is well-established that some probiotic bacteria may confer protection against this parasite in vitro and in vivo and we recently documented the implication of bile-salt hydrolase (BSH)-like activities from strain La1 of Lactobacillus johnsonii as mediators of these effects in vitro. We showed that these activities were able to generate deconjugated bile salts that were toxic to the parasite. In the present study, a wide collection of lactobacilli strains from different ecological origins was screened to assay their anti-giardial effects. Our results revealed that the anti-parasitic effects of some of the strains tested were well-correlated with the expression of BSH-like activities. The two most active strains in vitro, La1 and Lactobacillus gasseri CNCM I-4884, were then tested for their capacity to influence G. duodenalis infection in a suckling mice model. Strikingly, only L. gasseri CNCM I-4884 strain was able to significantly antagonize parasite growth with a dramatic reduction of the trophozoites load in the small intestine. Moreover, this strain also significantly reduced the fecal excretion of Giardia cysts after 5 days of treatment, which could contribute to blocking the transmission of the parasite, in contrast of La1 where no effect was observed. This study represents a step toward the development of new prophylactic strategies to combat G. duodenalis infection in both humans and animals.
Introduction
Giardia duodenalis (also known as G. lamblia or G. intestinalis) is the etiologic agent of the zoonotic disease giardiasis, one of the most common waterborne parasitic infections globally. G. duodenalis is a flagellate protozoan (Excavata, Diplomonad) that infects a broad diversity of animals such as humans, mammals, reptiles and birds (Thompson and Monis, 2004, 2012). Transmission can occur by ingestion of viable cysts from contaminated water and soil, or directly by contact with an infected animal’s feces (Gardner and Hill, 2001). After ingestion, exposure to gastric acid and proteases in the stomach leads to excystation and liberation of replicative trophozoite stages that adhere transiently to the proximal small intestine and persist for several days (for up to several months in some cases) (Reiner et al., 2003). After this replicative and pathogenic stage, trophozoites are carried into the colon and progressively encyst. Cysts, released through host feces, may then survive in the environment for several months and remain infectious at low doses (Thompson et al., 1993).
The main clinical symptoms of giardiasis are acute or chronic diarrhea, abdominal pain, intestinal malabsorption, steatorrhea, and weight loss (Farthing, 1996; Buret, 2007). Considered as a public health threat and a veterinary concern worldwide, giardiasis is responsible for many waterborne diarrhea outbreaks in developed countries (Roxstrom-Lindquist et al., 2006; Buret, 2007). In humans, it affects mainly children, and undernourished or immunosuppressed individuals. Even though giardiasis can be self-limited, standard antibiotic therapies including 5-nitroimidazole and benzimidazole drugs are needed to treat long-term infections (Savioli et al., 2006). In mammals, G. duodenalis infections are common in companion animals, small ruminants and cattle, for which alternative treatments are very limited (Harris et al., 2001).
The increasing number of clinical trial failures and the emergence of resistant Giardia strains in both medical and veterinary applications have encouraged the development of new therapeutic strategies (Harris et al., 2001; Upcroft and Upcroft, 2001). In this context, it has been established that gut microbiota plays a pivotal role in the protection against enteropathogens through the production of antimicrobial compounds and by competition for nutrients and attachment to the mucosal surface (Travers et al., 2011; Bengmark, 2013). In particular, probiotic bacteria such as some strains of lactobacilli have been shown to confer host health benefits by enhancing innate and adaptive immune responses (Sanders, 2008; Martin et al., 2013; Sokol, 2014). Moreover, pre-clinical studies suggest that colonization of the small intestine by G. duodenalis trophozoites depends on the composition of host gut microbiota (Singer and Nash, 2000; Barash et al., 2017; Bartelt et al., 2017). In recent years, probiotic-based therapies have been explored to treat giardiasis (Allain et al., 2017). For instance, some lactobacilli strains such Lactobacillus johnsonii La1 (also known as NCC533 but hereafter named La1), Lactobacillus casei MTCC1423 and Lactobacillus rhamnosus GG (LGG), have been shown to display anti-giardial properties by (i) antagonizing the proliferation of trophozoites and, (ii) reducing the severity of infection in several murine models (Humen et al., 2005; Shukla et al., 2008; Goyal et al., 2011; Travers et al., 2011). While the molecular mechanisms remain poorly understood, we have recently discovered the involvement of deconjugated bile salts, generated by the hydrolysis of conjugated bile salts from bile by La1 strain, as one of the mechanisms contributing to the inhibition of Giardia trophozoite growth in vitro (Perez et al., 2001; Travers et al., 2016). Our hypothesis was that these deconjugated bile salts were produced by bile salt hydrolase (BSH)-like enzymes released or secreted by this strain (Travers et al., 2016).
In the current study, we screened a wide collection of lactobacilli strains from various environmental origins in order to determine their ability to display an anti-giardial effect. In vitro analysis from supernatant-lactobacilli samples led to the identification of several strains having a strong inhibitory activity against G. duodenalis growth (up to the level previously reported for La1). In parallel, we showed that the in vitro anti-Giardia activity displayed by some of these lactobacilli strains is clearly correlated with their BSH-like activities in vitro. The two strains displaying the strongest inhibitory effects: La1 and Lactobacillus gasseri CNCM I-4884, were then evaluated in vivo in a suckling mice model challenged with the WB6 strain of G. duodenalis. Our results showed that L. gasseri CNCM I-4884 displayed a higher anti-giardial effect in vivo than La1, with an almost complete clearance of Giardia infection in suckling mice. These findings show that a screening based on the detection of BSH activities is a promising tool to identify new anti-Giardia lactobacilli strains. Overall, our study opens new therapeutic strategies for both preventing and treating giardiasis in humans.
Materials and Methods
Giardia duodenalis Culture Conditions
Giardia duodenalis was grown in vitro as recently described (Travers et al., 2016). Trophozoites of G. duodenalis strain WB clone 6 (WB6) assemblage A1 (ATCC 50803) were grown in Keiser’s modified TYI-S-33 medium (KM) (Morrison et al., 2007). KM medium was supplemented with 10% heat-inactivated fetal calf serum (FCS, reference A15-101, PAA Laboratories, GE healthcare), adjusted to pH 6.0, and sterilized with a 0.22 μm filter. G. duodenalis WB6 trophozoites were subcultured in anaerobic conditions at 5 × 104 cells per ml after chilling on ice for 10 min and centrifuged at 700 ×g, 5 min. Aliquots were frozen in liquid nitrogen and stored at -80°C until further use. For in vitro and in vivo experiments, 48 h old cultures of confluent trophozoites were pelleted at 700 × g, 5 min after chilling on ice for 10 min, and resuspended at the needed concentrations.
Bacterial Strains and Culture Conditions
The bacteria collection used in this study comprises 29 lactobacilli isolated from different biotopes (Table 1). The strains were grown in Man Rogosa Sharpe (MRS, Difco) medium on agar plates over night at 37°C in anaerobic conditions (GasPack Plus, BBL). Bacteria were then subcultured for 16 h at 37°C in MRS broth and were subsequently grown in Keiser’s modified TYI-S-33 medium supplemented with 10% heat-inactivated FCS (see above) to stationary phase. An intermediary subculture in modified TYI-S-33 medium was used for some strains as previously described (Perez et al., 2001). Bacterial supernatants were then collected after centrifugation at 10,000 ×g for 10 min, sterilized with a 0.22 μm filter and the pH was adjusted to 6.2 with 5N NaOH.
Table 1
| Lactobacillus species | Strain1 | Origin | TDCA (0.5%) hydrolase activity | TDCA hydrolase activity score2 | GDCA (0.5%) hydrolase activity | GDCA hydrolase activity score2 |
|---|---|---|---|---|---|---|
| L. acidophilus | ATCC4356 | Feces, human | P | + | N | - |
| L. brevis | CNRZ1845 | Malt (beer) | P | + | P | + |
| L. casei | ATCC393 | Cheese | N | - | N | - |
| L. crispatus | CIP103606 | Unknown | N | - | P | ++ |
| L. curvatus | CNRZ1335 | Fermented sausage/mea | N | - | N | - |
| L. gasseri | ATCC33323 | Vaginal tract, human | N | - | N | - |
| L. gasseri | CNCM I-4884 ATCC29601 | Cariouth tooth, human | P | +++ | P | ++ |
| L. helveticus | CNRZ1109 | Lactic starter | N | - | N | - |
| L. helveticus | ATCC11977 | Lactic starter (cheese) | N | - | P | ++ |
| L. johnsonii | CNRZ217 | Feces, rat | N | - | P | + |
| L. johnsonii | CNRZ218 | Feces, rat | P | + | P | ++ |
| L. johnsonii | CNRZ1897 | Fermented milk | P | +++ | ND | ND |
| L. johnsonii | CIP103614 | Vaginal tract, human | P | ++ | N | - |
| L. johnsonii | CIP103786 | Cheese | P | + | P | ++ |
| L. johnsonii | CIP103652 | Unknown | P | ++ | N | - |
| L. johnsonii | CIP103653 | Unknown | P | ++ | N | - |
| L. johnsonii | CIP103654 | Pharmaceutical preparation | P | + | P | + |
| L. johnsonii | CIP103781 | Unknown | P | +++ | P | ++ |
| L. johnsonii | CIP103782 | Urethran, human | P | + | N | - |
| L. johnsonii | ATCC33200 | Blood, human | P | ++ | P | ++ |
| L. johnsonii | La1 (NCC533) | Feces, human | P | +++ | P | ++ |
| L. paracasei | CNRZ315 | Unknown | N | - | N | - |
| L. pentosus | CNRZ1218 | Cheese | N | - | N | - |
| L. plantarum | CNRZ738 | Silage | N | - | P | + |
| L. rhamnosus | CNRZ317 | Unknown | N | - | N | - |
| L. rhamnosus | CNRZ2084 | Fermented milk | N | - | N | - |
| L. reuteri | CNRZ431 | Sourdough | N | - | N | - |
| L. sakei | CNRZ1332 | Moto, starter of sake | N | - | N | - |
| L. zeae | CNRZ2269 | Corn steep liquor | N | - | N | - |
List of tested lactobacilli strains with their characteristics and bile salt hydrolase activity specificities.
P, positive; N, negative; ND, non-determined. 1Strains were obtained from different collections: CIP, Collection de l’Institut Pasteur, France; CNRZ/CIRM, Centre International de Ressources Microbiennes; CNCM, Collection Nationale de Cultures de Microorganismes, Institut Pasteur, France; ATCC, American Type Culture Collection, United States. 2TDCA/GDCA hydrolase activity score were determined on the size of the halo zone (diameter): (-): no detected halo; (+): <8 mm; (++): 9 mm to 12 mm; (+++): >13 mm.
Bile Salt Hydrolase Assays
The bacterial strains were tested for Tauro-deoxycholic acid (TDCA) and Glyco-deoxycholic acid (GDCA) hydrolase activities on MRS-agar plates supplemented with either 0.5% TDCA (Sigma–Aldrich) or 0.5% GDCA (Merck) following the protocol described by Moser and Savage (2001). MRS plates with 0.5% TDCA were first incubated at 37°C in anaerobic conditions for 24 h prior to inoculation. Strains were grown anaerobically for 16 h at 37°C in MRS broth, then streaked on MRS-agar plates, supplemented or not, with 0.5% TDCA or 0.5% GDCA and incubated at 37°C in anaerobic conditions for 48–72 h. BSH activity can be easily detected when unconjugated deoxycholic acid precipitates in the MRS-agar plate, forming an iridescent halo below and around active colonies. These assays were performed in duplicates, and included two biological replicates. BSH activity score was determined as indicated in Table 1.
In Vitro Anti-giardial Assays
Filtered bacterial supernatants (500 μl) were co-incubated with fresh cultures of Giardia trophozoites (1.33 × 105 parasites/ml in KM medium, pH 6.0, supplemented with 10% heat-inactivated FCS) at a volumetric ratio of 1–3, in the presence or absence of bovine bile (0.6 g/L) at 37°C in anaerobic conditions for 22 h. BSH from Clostridium perfringens (1 U) (reference C4018, Sigma–Aldrich) was used as a positive control for Giardia growth inhibition as previously described (Travers et al., 2016). Samples were then ice-chilled for 10 min and trophozoite load was determined using hemocytometer (flagella mobility was used to screen parasite viability). The inhibition rates were determined by counting the number of living trophozoites and establishing ratios compared to the controls (in percentage). Experiments were conducted in duplicates and included three biological replicates.
Assessment of Anti-giardial Activityin Vivo
Cultures of G. duodenalis trophozoites WB6 were grown in Keister’s modified TYI-S-33 medium with 10% heat-inactivated FCS. Lactobacilli strains (La1, L. gasseri CNCM I-4884 and L. curvatus CNRZ1335) were cultured in MRS broth for 16 h at 37°C in anaerobic conditions from precultures. Bacteria were then harvested by centrifugation at 7000 ×g for 15 min, washed two times and resuspended in a corresponding volume of sterile PBS/Glycerol 15% to obtain a final concentration of 2.5 × 1010 CFU/ml for intragastric administrations (5 × 108 CFU per neonatal mouse). Mice used for reproduction were non-inbred mice of the OF1 strain (Charles River, Saint-Germain-Nuelles, France). All experiments were conducted in a filtered air chamber and manipulations were performed under a laminar flow hood to prevent contamination of the environment by cysts of G. duodenalis and other pathogens.
Lactobacilli strains were administered daily by intragastric gavage in a volume of 20 μl (5 × 108 CFU) to 5 days old neonatal mice from day 5 to day 15 (n = 9–12 per group) (Figure 2). Control animals (n = 8) received PBS with glycerol 15% instead of bacterial suspensions. Neonatal mice were then challenged with trophozoites at day 10 by intragastric gavage in a volume of 100 μl (105 trophozoites). Mice were sacrificed by cervical dislocation at day 16. The small intestine and colon contents were examined for the presence of trophozoites and cysts, by counting the parasite burden, using a hemocytometer. The counting of trophozoites was performed from the small intestine (resuspended in 5 ml of ice-chilled sterile PBS), while cysts were enumerated in the colon and caecum (resuspended in 5 ml of ice-chilled 2.5% Potassium dichromate). The minimum threshold of counting values for statistical analysis was set at 103 trophozoites.
Statistical Analysis
Results were expressed as means ± standard error of the mean (SEM). Comparison between groups was assessed by one or two-way analysis of variance (ANOVA), t-test, Mann–Whitney and Kruskal–Wallis. Correlation tests were performed using Spearman’s Rank Correlation Test. Statistical significance was calculated at p-values under 0.05 at 95% confidence interval.
Ethics Statement
All protocols were carried out in accordance with the institutional ethical guidelines of the ethics committee ANSES’s Animal Health Laboratory at Maisons-Alfort on the campus of the French National Veterinary School of Alfort (ENVA), which approved this study.
Results
BSH Activities of Lactobacilli Strains
The 29 strains tested in this study were able to grow in MRS-agar supplemented with 0.5% TDCA whereas only 25 grew in MRS-agar supplemented with 0.5% GDCA, supporting that GDCA has a higher bactericidal activity than TDCA in vivo (Begley et al., 2006). TDCA/GDCA hydrolase activity is a well-recognized indicator of BSH activity in bacteria (Bustos et al., 2012). A semi-quantitative method was used to monitor the BSH activity by measuring the halo zone of positive strains after 48–72 h of incubation. Among the 29 strains, 6 exhibited TDCA hydrolase activity, 4 exhibited GDCA hydrolase activity and 8 displayed both TDCA and GDCA hydrolase activities (Table 1). No halo was detected for 11 strains. Interestingly, all L. johnsonii strains tested displayed at least one type of BSH activity. Among the strains harboring both tauro and glyco-specific BSH activities, La1, L. gasseri CNCM I-4884 and L. johnsonii CIP103782 exhibited the highest BSH scores (Table 1 and Figure 1A).
FIGURE 1
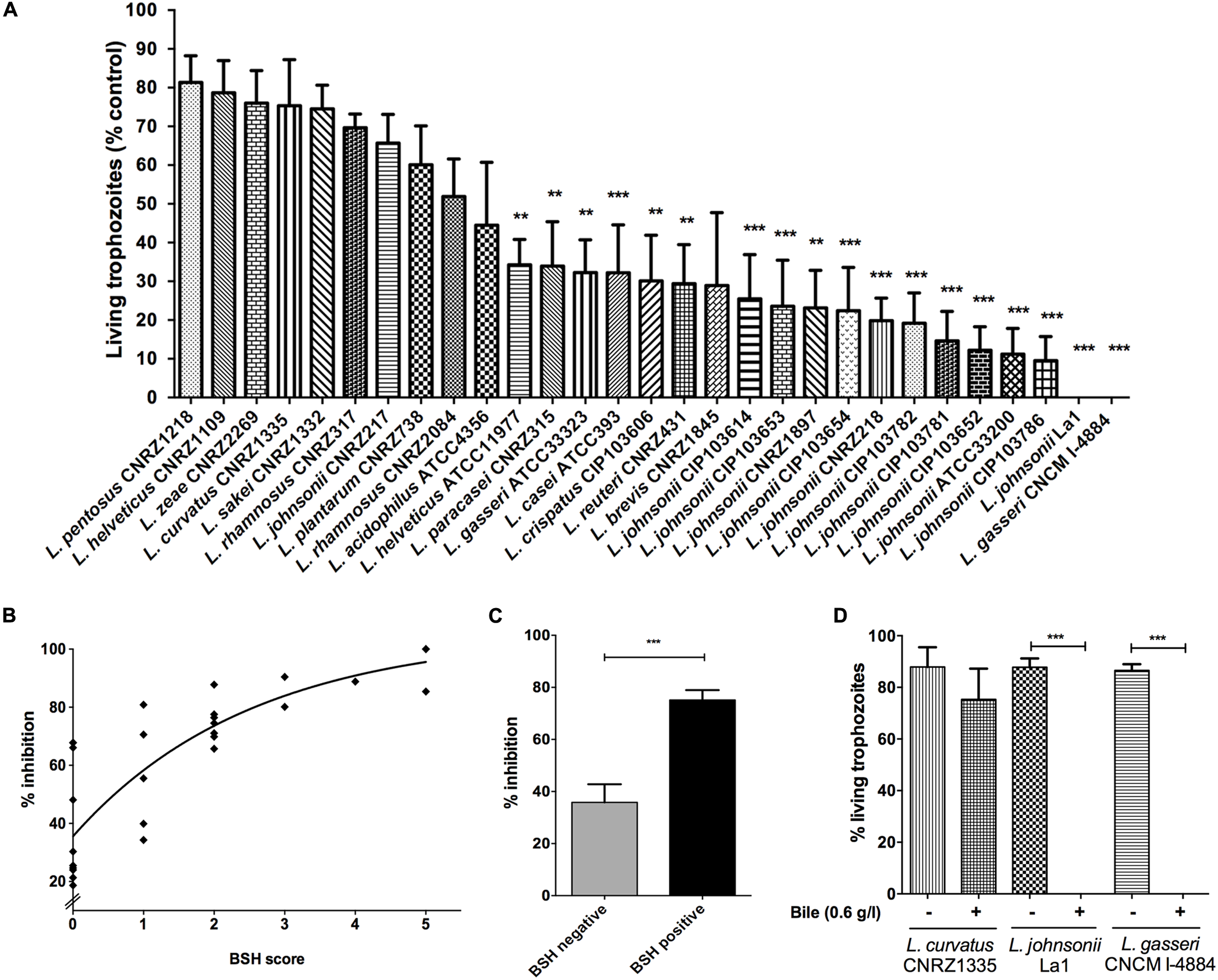
Bile-salt hydrolase (BSH) and anti-Giardia in vitro activities of different lactobacilli strains. (A) Percentage of living Giardia duodenalis trophozoites when cultivated 22 h with lactobacilli supernatants. G. duodenalis trophozoites were enumerated after 22 h of co-incubation at 37°C in anaerobic conditions (values are represented with bile supplementation). Values are mean ± SEM. (B) Spearman’s rank correlation test between percentage of inhibition of lactobacilli strains and their BSH activity score. BSH score was determined depending on the size of the halo zone (+ = 1; ++ = 2; +++ = 3, Table 1) and the ability to deconjugate either tauro-conjugated bile salts or glyco-conjugated bile salts or both (addition of BSH score for each substrate specificity). A positive correlation is observed (r = 0.86; p < 0.0001). (C) Inhibition assays according to Bile Salt Hydrolase activities. Lactobacilli strains were divided into two groups: (i) BSH negative (strains with no detected BSH activity), (ii) BSH positive (strains exhibiting either TDCA, GDCA, or both BSH activities). G. duodenalis trophozoites were enumerated after 22 h of co-incubation at 37°C in anaerobic conditions (values are represented with bile supplementation). Values are in mean ± SEM. (D) Percentage of living G. duodenalis trophozoites when co-cultivated 22 h with lactobacilli strains supernatants, with or without bile supplementation (bovine bile 0.6 g/L). G. duodenalis trophozoites were enumerated after 22 h of co-incubation at 37°C in anaerobic conditions. Values are mean ± SEM. ∗∗p ≤ 0.01; ∗∗∗p ≤ 0.001.
Characterization of Anti-giardial Lactobacilli Strains
We screened the 29 lactobacilli strains for their inhibitory abilities against G. duodenalis. For this, bacterial supernatants were co-incubated for 22 h at 37°C with Giardia trophozoite cultures in KM growth medium supplemented, or not, with bovine bile (0.6 g/L). Living trophozoites were enumerated using hemocytometer. Parasite cultures that were grown without bile did not display major differences when compared to trophozoites grown in KM supplemented with bile, showing that cholesterol and lipids from fetal bovine serum are sufficient to fulfill the lipid uptake requirements of Giardia and that bile components are not toxic for Giardia (Travers et al., 2016).
When co-incubated with Giardia in KM supplemented with bovine bile, 19 bacterial supernatants exhibited significant antagonistic effects on Giardia growth, with a wide range of inhibition levels (Figure 1A). Six strains showed growth inhibition levels from 15 to 30%, 10 exhibited growth inhibition levels from 30 to 70%, and 12 strains displayed a strong reduction of trophozoites by 70–100% (p < 0.001). Only supernatant of L. gasseri CNCM I-4884 were as efficient as that of La1 to antagonize Giardia growth in vitro (100%). Using Spearman’s Rank Correlation Test, we observed a positive correlation between inhibition levels and BSH activity score (r = 0.86; p < 0.0001) of bacterial strains (Figure 1B). Inhibition assays were then divided into two groups: (i) BSH negative (strains with no detected BSH activity) and (ii) BSH positive (strains displaying either TDCA, GDCA, or both BSH activities). We observed that Giardia growth inhibitory activity of BSH positive strains were twofold higher relative to BSH negative strains (Student’s t-test; p < 0.0001) (Figure 1C). Moreover, the cytotoxic effect on Giardia was lost when trophozoites were co-incubated with bacterial supernatant in the absence of bile (Figure 1D). However, a very weak inhibition was still observed for a majority of the strains (range between 5 and 15%), which can be explained by the presence of other extracellular compounds released by lactobacilli (e.g., bacteriocins) that are also potentially deleterious to Giardia growth. Taken together, these in vitro results strongly suggest that the anti-giardial effect displayed by lactobacilli strains tested in this study is mostly bile-dependent.
In Vivo Effect of Orally Administered Lactobacilli against G. duodenalis
In vivo experiments were performed in order to assess the potential of newly identified anti-giardial lactobacilli strains to antagonize G. duodenalis in vivo. Bacterial suspensions, or PBS/glycerol were daily administered by intragastric gavage (5 × 108 CFU) to neonatal mice from day 5 to day 15 (Figure 2). The persistence of lactobacilli in suckling mice and administration route were determined in previous experiments (Supplementary Figure S1). Mice were challenged with G. duodenalis WB6 trophozoites at day 10 by intragastric gavage (1 × 105 trophozoites) and sacrificed at day 16 (Figure 2). In preliminary experiments, we showed that this parasite strain was able to persist and colonize in OF1 suckling mice model, the peak of trophozoite load being reached at day 16 (i.e., 6 days post-inoculation) (Supplementary Figure S2). On the other hand, we demonstrated that lactobacilli strains can persist in the gut up to 3–4 days after intragastric gavage.
FIGURE 2
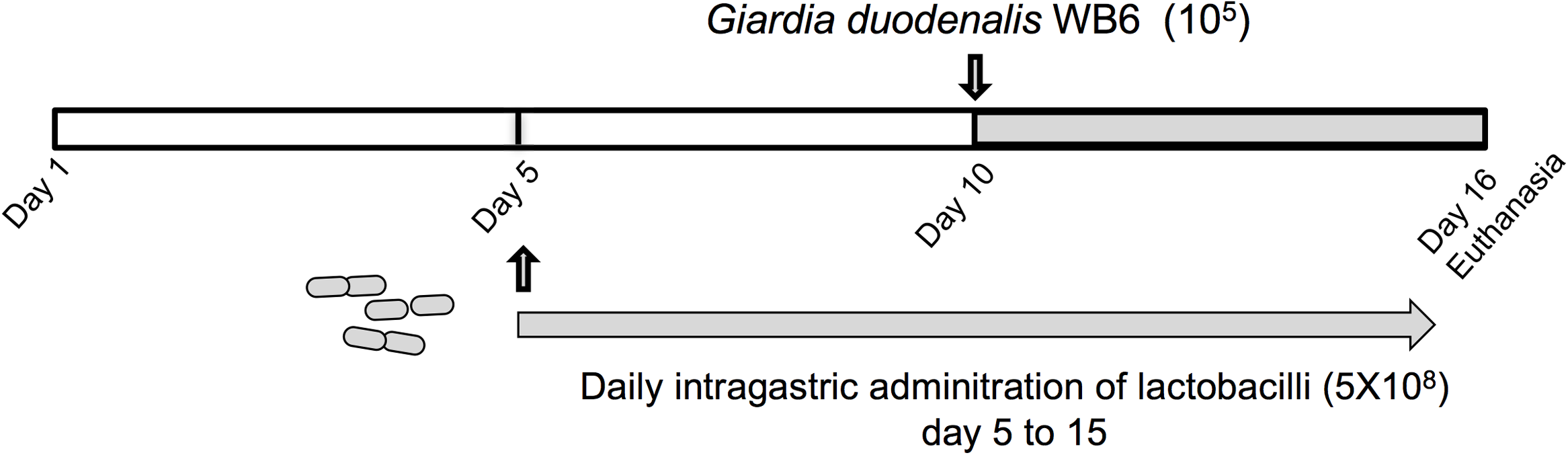
Experimental design of G. duodenalis infection in OF1 suckling mice model. Lactobacilli strains were administered daily by intragastric gavage (5 × 108 CFU) to 5 days old OF1 suckling mice from day 5 to day 15. Control animals received PBS/glycerol 15%. Mice were challenged with G. duodenalis WB6 trophozoites (105) at day 10 by intragastric gavage. Mice were sacrificed by cervical dislocation at day 16.
Mice were divided into four groups with a minimum of eight animals per group. The parasite burden in mice treated with PBS/glycerol was 20-fold higher than the initial dose showing that trophozoites were able to colonize, multiply, and persist in the small intestine. As shown in Figure 3A, mice treated with L. curvatus CNRZ1335 were not protected against G. duodenalis challenge as they present a similar profile in trophozoite colonization and proliferation. In contrast, mice treated with L. gasseri CNCM I-4884 exhibited an 18-fold reduction in the trophozoites load in the small intestine (∼93%) (p < 0.001) compared to the PBS/glycerol group (Figure 3A). Mice fed with the probiotic strain La1 exhibited a twofold reduction in the trophozoites load compared to controls (∼43%). Interestingly, L. gasseri CNCM I-4884 was more efficient than La1 in preventing G. duodenalis proliferation in vivo.
FIGURE 3
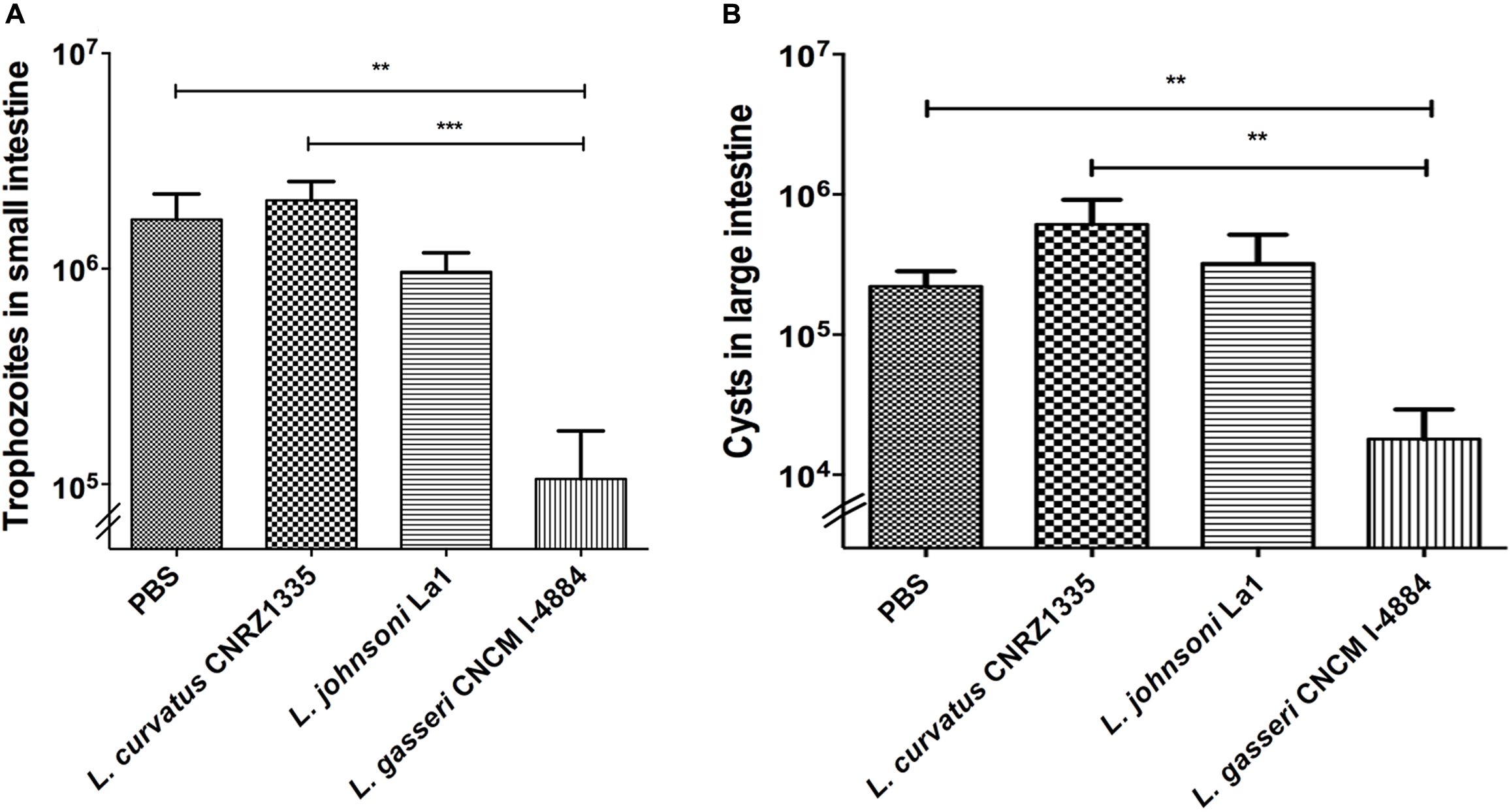
In vivo activities of Lactobacillus gasseri CNCM I-4884 and La1 lactobacilli strains against G. duodenalis.(A)G. duodenalis trophozoites enumeration after administration to animals. Suckling mice received either PBS (n = 8), La1 (n = 12), L. gasseri CNCM I-4884 (n = 10) or L. curvatus CNRZ1335 (n = 9) by intragastric gavage (5 × 108 CFU/mice) daily from day 5, before inoculation with G. duodenalis WB6 trophozoites (105 trophozoites per animal) at day 10. Gavages were performed until day 15. Small intestines were resuspended in PBS and trophozoites were counted using a hemocytometer. Values are mean ± SEM; p < 0.05. (B)G. duodenalis cysts counting in colon and caecum. Suckling mice received either PBS/glycerol 15%, La1, L. gasseri CNCM I-4884 or L. curvatus CNRZ1335 by intragastric gavage (5 × 108 CFU/mice) daily from day 5 before inoculation with G. duodenalis WB6 trophozoites (105 trophozoites per animal) at day 10. Gavages were performed until day 15 (n = 8–12/group). Colons and caeca were resuspended in 5 ml of ice-chilled 2.5% potassium dichromate and cysts were counted using a hemocytometer. Values are mean ± SEM. ∗∗p ≤ 0.01; ∗∗∗p ≤ 0.001.
Cysts enumerations were performed in both colon and caecum. We observed a significant reduction (-81%) (p < 0.01) in cyst formation in groups treated with L. gasseri CNCM I-4884 compared to both PBS/glycerol and L. curvatus-treated groups. No significant reduction was observed in the group treated with either La1 or L. curvatus CNRZ1335 compared to control groups treated with PBS/glycerol (Figure 3B).
Discussion
Several studies have demonstrated that probiotics might play key roles against enteropathogens including intestinal prototozan parasites (see Travers et al., 2011, for a review). In the last 10 years, several lactobacilli strains have been studied for their ability to prevent the establishment of G. duodenalis in vivo and to reduce the severity of giardial infections (Humen et al., 2005; Shukla et al., 2008; Goyal et al., 2011; Goyal and Shukla, 2013). In some instances, in vitro assays were designed to decipher the molecular mechanisms involved in these protective effects. For the probiotic strain La1 of L. johnsonii, previously known to antagonize Giardia growth in vitro and in vivo, Perez and collaborators established the presence of an active principle in the supernatant of this probiotic strain (Perez et al., 2001; Humen et al., 2005). More recently, we undertook the molecular characterization of this active principal from La1 and discovered at least one possible mechanism of action: the involvement of bacterial BSH-like activities, that would mediate anti-giardial effect by generating deconjugated bile salts (toxic for the parasite) from non-toxic conjugated bile salts (Travers et al., 2016). BSH are common in Lactobacillus and Bifidobacterium, playing an important role for colonization and persistence in the gut (Tanaka et al., 1999; Begley et al., 2006; Denou et al., 2008). Therefore, we looked for other probiotic strains (closely related or not to La1) exhibiting anti-giardial activities. We thus selected 29 lactobacilli strains from diverse origins and screened their supernatants for their putative anti-giardial activities in vitro while, in parallel, we analyzed their BSH activities and properties. Among the supernatants issued from the 29 analyzed strains, 19 were found to display anti-parasitic activities on fresh trophozoites Giardia cultures. Interestingly, these 19 strains also displayed the highest BSH scores, indicating a correlation between these two properties. These results thereby suggest that a screening method based on BSH activities may predict potential anti-giardial activity in lactobacilli. Interestingly, L. gasseri CNCM I-4884 and La1 strains (which killed 100% of trophozoites in vitro) displayed both TDCA and GDCA specific BSH activities. We therefore propose that this dual or combined BSH activity would contribute to reinforce the release of deconjugated bile salt toxic for Giardia, from a larger panel of substrates. However, no clear relationships appeared between the observed anti-giardial activity and the environmental origins of the lactobacilli isolates (Table 1).
Lactobacillus gasseri CNCM I-4884 and La1 were subsequently administrated to OF1 suckling mice to assess their antagonistic effects in vivo. L. curvatus CNRZ1335, which displayed neither anti-giardial activity nor BSH activities in vitro, was used as a negative control. High trophozoites burden in controls showed efficient colonization and proliferation of the G. duodenalis WB6 in the OF1 mice (Shukla et al., 2008). Mice treated with L. gasseri CNCM I-4884 exhibited a dramatic reduction of viable trophozoites in their small intestines, 6 days post-challenge. La1 strain supplementation also lead to a reduction of trophozoites in mice intestines, but to a much lesser extent than L. gasseri CNCM I-4884. Previous studies have reported anti-giardial properties of lactobacilli strains. For instance, L. casei MTCC1423 and LGG strains reduced the duration and the severity of G. duodenalis infection (Portland strain) in C57BL/6 mice in 7–14 days (Shukla et al., 2008; Goyal et al., 2011) and La1 antagonized Giardia (WB6) in Mongolian gerbils in 7–21 days (Humen et al., 2005). However, no correlation between the anti-giardial effects and a BSH-like activity was established in these studies. Strikingly, in the current study, administration of L. gasseri CNCM I-4884 led to a stronger reduction of trophozoites load in a short period of time. Cyst formation is another important indication of the control of infectious parasite development (Lujan et al., 1997). A treatment capable of preventing trophozoite differentiation into cysts is indeed essential to minimize their spreading in the environment via animals’ feces. We observed that mice fed with L. gasseri CNCM I-4884 exhibited a dramatic reduction of cyst excretion 6 days post-challenge compared to untreated infected mice. This is in concordance with previous results showing that other lactobacilli such as L. casei MTCC 1423 and LGG were effective in eliminating cysts after 7 days of treatment. In contrast, no cyst reduction was observed in mice treated with La1 although its ability to eliminate cysts has been previously reported in mongolian gerbils (Humen et al., 2005). The differences in terms of in vivo efficiency between L. gasseri CNCM I-4884 and La1, both BSH-positive strains, can be explained by either a better hydrolysis of conjugated bile salts in vivo, a higher ability for competing for biological niches in the small intestine, and a better persistence in the gut.
To date, the underlying mechanisms involved in the antagonistic effect of lactobacilli against Giardia is an emerging field. Besides the role of BSH-like activities that we have previously reported for La1 (Travers et al., 2016), and now for other Lactobacillus strains, other mechanisms have been identified. Among them, the role of anti-microbial peptides in the anti-giardial activity of lactobacilli has been explored. Indeed, when administered orally, P106, a derived bacteriocin isolated from L. acidophilus, was shown to reduce the trophozoite burden in mice at high concentrations (Amer et al., 2014). However, the ability to produce anti-microbial peptides is strain dependent and highly variable and this cannot be extrapolated to all anti-giardial strains. Also, probiotic lactobacilli are known to enhance the mucosal immune system, which participate in clearing enteropathogens from the gut (Howarth and Wang, 2013). The immunomodulatory properties of lactobacilli have also been proposed to explain their anti-giardial properties (Goyal and Shukla, 2013; Allain et al., 2017; Barash et al., 2017; Bartelt et al., 2017; Fink and Singer, 2017). In our study, the local and systemic response to Giardia infection has not been assessed due to the fact that suckling mice have an immature immune system (Basha et al., 2014).
While the anti-giardial properties of lactobacilli appear to be multifactorial, there is a clear contribution of BSH-activities. However, further experiments are necessary to investigate the potential of BSH in treating giardiasis, including the direct effect of BSHs in vitro and in vivo. Knockout mutant strains for all BSH genes (several BSH genes are commonly found in lactobacilli) are still needed to evaluate the relative contribution of each one of these enzymes. To summarize, this study shows BSH activity as a key screening parameter to identify anti-Giardia lactobacilli strains. In addition, L. gasseri CNCM I-4884, which displayed both high anti-giardial and BSH activities in vitro, antagonizes Giardia survival in OF1 mice. This study represents a significant step toward the development of new prophylactic strategies, with both human and veterinary applications.
Statements
Ethics statement
All protocols were carried out in accordance with the institutional ethical guidelines of the ethics committee ANSES’s Animal Health Laboratory at Maisons-Alfort on the campus of the French National Veterinary School of Alfort (ENVA), which approved this study.
Author contributions
IF, BP, PG, TA, and LB-H conceived and designed the study. TA, BP, SC, and MT performed all the experiments. M-AT, IV, and PL discussed the experiments and results. TA, IF, and LB-H wrote the manuscript.
Funding
This work was partially funded by Région Île-de-France-DIM (Maladies Infectieuses, Parasitaires et Nosocomiales Émergentes, program no. 120092).
Acknowledgments
The authors thank Dr. Cissé Sow (MHNH) and Yasmine Kirati (Mett) for their technical help in preliminary experiments. They also thank Dr. Saulius Kulakauskas (INRA, Jouy-en-Josas) who provided some strains of lactic acid bacteria.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmicb.2018.00089/full#supplementary-material
FIGURE S1Persistence of L. johnsonii La1 strain (Eryr) in OF1 suckling mice. Each mouse received a single administration of 5 × 108 CFU of L. johnsonii La1 Eryr either by oral gavage (days 1, 2, and 3; n = 4) or intragastric gavage (days 1, 2, 3, and 4; n = 4). L. johnsonii La1 was transformed with a plasmid harboring an erythromycin (Ery)-resistance gene as described previously (Allain et al., 2016). Values are in mean ± SEM.
FIGURE S2Kinetics of infection of G. duodenalis strain WB6 in OF1 suckling mice. (A)G. duodenalis trophozoites enumeration in small intestine after single gavage (day 0). Trophozoite burden was measured at days 3, 6, 8, 10, 12, 15, and 18 (n = 10). Small intestines were resuspended in PBS and trophozoites were counted using a hemocytometer (B)G. duodenalis cysts enumeration in large intestine after single gavage (day 0). Cysts were measured at days 3, 6, 8, 10, 12, 15, and 18 (n = 10). Large intestines were resuspended in 2.5% Potassium dichromate and cysts were counted using a hemocytometer. Values are in mean ± SEM.
References
1
AllainT.AmatC. B.MottaJ. P.MankoA.BuretA. G. (2017). Interactions of Giardia sp. with the intestinal barrier: epithelium, mucus, and microbiota.Tissue Barriers5:e1274354. 10.1080/21688370.2016.1274354
2
AllainT.MansourN. M.BahrM. M.MartinR.FlorentI.LangellaP.et al (2016). A new lactobacilli in vivo expression system for the production and delivery of heterologous proteins at mucosal surfaces.FEMS Microbiol. Lett.363:fnw117. 10.1093/femsle/fnw117
3
AmerE. I.MossallamS. F.MahrousH. (2014). Therapeutic enhancement of newly derived bacteriocins against Giardia lamblia.Exp. Parasitol.14652–63. 10.1016/j.exppara.2014.09.005
4
BarashN. R.MaloneyJ. G.SingerS. M.DawsonS. C. (2017). Giardia alters commensal microbial diversity throughout the murine gut.Infect. Immun.85e00948-e16. 10.1128/IAI.00948-16
5
BarteltL. A.BolickD. T.Mayneris-PerxachsJ.KollingG. L.MedlockG. L.ZaenkerE. I.et al (2017). Cross-modulation of pathogen-specific pathways enhances malnutrition during enteric co-infection with Giardia lamblia and enteroaggregative Escherichia coli.PLOS Pathog.13:e1006471. 10.1371/journal.ppat.1006471
6
BashaS.SurendranN.PichicheroM. (2014). Immune responses in neonates.Expert Rev. Clin. Immunol.101171–1184. 10.1586/1744666X.2014.942288
7
BegleyM.HillC.GahanC. G. (2006). Bile salt hydrolase activity in probiotics.Appl. Environ. Microbiol.721729–1738. 10.1128/AEM.72.3.1729-1738.2006
8
BengmarkS. (2013). Gut microbiota, immune development and function.Pharmacol. Res.6987–113. 10.1016/j.phrs.2012.09.002
9
BuretA. G. (2007). Mechanisms of epithelial dysfunction in giardiasis.Gut56316–317. 10.1136/gut.2006.107771
10
BustosA. Y.SaavedraL.De ValdezG. F.RayaR. R.TarantoM. P. (2012). Relationship between bile salt hydrolase activity, changes in the internal pH and tolerance to bile acids in lactic acid bacteria.Biotechnol. Lett.341511–1518. 10.1007/s10529-012-0932-5
11
DenouE.PridmoreR. D.BergerB.PanoffJ. M.ArigoniF.BrussowH. (2008). Identification of genes associated with the long-gut-persistence phenotype of the probiotic Lactobacillus johnsonii strain NCC533 using a combination of genomics and transcriptome analysis.J. Bacteriol.1903161–3168. 10.1128/JB.01637-07
12
FarthingM. J. (1996). Giardiasis.Gastroenterol. Clin. North Am.25493–515. 10.1016/S0889-8553(05)70260-0
13
FinkM. Y.SingerS. M. (2017). The intersection of immune responses, microbiota, and pathogenesis in giardiasis.Trends Parasitol.33901–913. 10.1016/j.pt.2017.08.001
14
GardnerT. B.HillD. R. (2001). Treatment of giardiasis.Clin. Microbiol. Rev.14114–128. 10.1128/CMR.14.1.114-128.2001
15
GoyalN.ShuklaG. (2013). Probiotic Lactobacillus rhamnosus GG modulates the mucosal immune response in Giardia intestinalis-infected BALB/c mice.Dig. Dis. Sci.581218–1225. 10.1007/s10620-012-2503-y
16
GoyalN.TiwariR. P.ShuklaG. (2011). Lactobacillus rhamnosus GG as an effective probiotic for murine giardiasis.Interdiscip. Perspect. Infect. Dis.2011:795219. 10.1155/2011/795219
17
HarrisJ. C.PlummerS.LloydD. (2001). Antigiardial drugs.Appl. Microbiol. Biotechnol.57614–619. 10.1007/s002530100720
18
HowarthG. S.WangH. (2013). Role of endogenous microbiota, probiotics and their biological products in human health.Nutrients558–81. 10.3390/nu5010058
19
HumenM. A.De AntoniG. L.BenyacoubJ.CostasM. E.CardozoM. I.KozubskyL.et al (2005). Lactobacillus johnsonii La1 antagonizes Giardia intestinalis In Vivo.Infect. Immun.731265–1269. 10.1128/IAI.73.2.1265-1269.2005
20
LujanH. D.MowattM. R.NashT. E. (1997). Mechanisms of Giardia lamblia differentiation into cysts.Microbiol. Mol. Biol. Rev.61294–304.
21
MartinR.MiquelS.UlmerJ.KechaouN.LangellaP.Bermudez-HumaranL. G. (2013). Role of commensal and probiotic bacteria in human health: a focus on inflammatory bowel disease.Microb. Cell Fact.12:71. 10.1186/1475-2859-12-71
22
MorrisonH. G.McarthurA. G.GillinF. D.AleyS. B.AdamR. D.OlsenG. J.et al (2007). Genomic minimalism in the early diverging intestinal parasite Giardia lamblia.Science3171921–1926. 10.1126/science.1143837
23
MoserS. A.SavageD. C. (2001). Bile salt hydrolase activity and resistance to toxicity of conjugated bile salts are unrelated properties in lactobacilli.Appl. Environ. Microbiol.673476–3480. 10.1128/AEM.67.8.3476-3480.2001
24
PerezP. F.MinnaardJ.RouvetM.KnabenhansC.BrassartD.De AntoniG. L.et al (2001). Inhibition of Giardia intestinalis by extracellular factors from Lactobacilli: an In Vitro study.Appl. Environ. Microbiol.675037–5042. 10.1128/AEM.67.11.5037-5042.2001
25
ReinerD. S.HetskoM. L.MeszarosJ. G.SunC. H.MorrisonH. G.BruntonL. L.et al (2003). Calcium signaling in excystation of the early diverging eukaryote, Giardia lamblia.J. Biol. Chem.2782533–2540. 10.1074/jbc.M208033200
26
Roxstrom-LindquistK.PalmD.ReinerD.RingqvistE.SvardS. G. (2006). Giardia immunity–an update.Trends Parasitol.2226–31. 10.1016/j.pt.2005.11.005
27
SandersM. E. (2008). Probiotics: definition, sources, selection, and uses.Clin. Infect. Dis.46(Suppl. 2)S58–S61. 10.1086/523341
28
SavioliL.SmithH.ThompsonA. (2006). Giardia and Cryptosporidium join the ‘Neglected Diseases Initiative’.Trends Parasitol.22203–208. 10.1016/j.pt.2006.02.015
29
ShuklaG.DeviP.SehgalR. (2008). Effect of Lactobacillus casei as a probiotic on modulation of giardiasis.Dig. Dis. Sci.532671–2679. 10.1007/s10620-007-0197-3
30
SingerS. M.NashT. E. (2000). The role of normal flora in Giardia lamblia infections in mice.J. Infect. Dis.1811510–1512. 10.1086/315409
31
SokolH. (2014). Probiotics and antibiotics in IBD.Dig. Dis.32(Suppl. 1)10–17. 10.1159/000367820
32
TanakaH.DoesburgK.IwasakiT.MierauI. (1999). Screening of lactic acid bacteria for bile salt hydrolase activity.J. Dairy Sci.822530–2535. 10.3168/jds.S0022-0302(99)75506-2
33
ThompsonR. C.MonisP. (2012). Giardia–from genome to proteome.Adv. Parasitol.7857–95. 10.1016/B978-0-12-394303-3.00003-7
34
ThompsonR. C.MonisP. T. (2004). Variation in Giardia: implications for taxonomy and epidemiology.Adv. Parasitol.5869–137. 10.1016/S0065-308X(04)58002-8
35
ThompsonR. C.ReynoldsonJ. A.MendisA. H. (1993). Giardia and giardiasis.Adv. Parasitol.3271–160. 10.1016/S0065-308X(08)60207-9
36
TraversM. A.FlorentI.KohlL.GrellierP. (2011). Probiotics for the control of parasites: an overview.J. Parasitol. Res.2011:610769. 10.1155/2011/610769
37
TraversM. A.SowC.ZirahS.DeregnaucourtC.ChaouchS.QueirozR. M.et al (2016). Deconjugated bile salts produced by extracellular bile-salt hydrolase-like activities from the probiotic Lactobacillus johnsonii La1 Inhibit Giardia duodenalis In vitro Growth.Front. Microbiol.7:1453. 10.3389/fmicb.2016.01453
38
UpcroftP.UpcroftJ. A. (2001). Drug targets and mechanisms of resistance in the anaerobic protozoa.Clin. Microbiol. Rev.14150–164. 10.1128/CMR.14.1.150-164.2001
Summary
Keywords
Giardia duodenalis, lactobacilli, Lactobacillus johnsonii, Lactobacillus gasseri, probiotics, bile salt hydrolases
Citation
Allain T, Chaouch S, Thomas M, Travers M-A, Valle I, Langella P, Grellier P, Polack B, Florent I and Bermúdez-Humarán LG (2018) Bile Salt Hydrolase Activities: A Novel Target to Screen Anti-Giardia Lactobacilli?. Front. Microbiol. 9:89. doi: 10.3389/fmicb.2018.00089
Received
26 September 2017
Accepted
15 January 2018
Published
08 February 2018
Volume
9 - 2018
Edited by
Andrea Gomez-Zavaglia, Centro de Investigación y Desarrollo en Criotecnología de Alimentos (CIDCA), Argentina
Reviewed by
Siddhartha Das, University of Texas at El Paso, United States; Paula Carasi, Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Argentina
Updates
Copyright
© 2018 Allain, Chaouch, Thomas, Travers, Valle, Langella, Grellier, Polack, Florent and Bermúdez-Humarán.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Luis G. Bermúdez-Humarán, luis.bermudez@inra.fr Isabelle Florent, isabelle.florent@mnhn.fr
†Present address: Marie-Agnès Travers, Laboratoire de Génétique et Pathologie des Mollusques Marins, SG2M-LGPMM, Ifremer, La Tremblade, France
This article was submitted to Food Microbiology, a section of the journal Frontiers in Microbiology
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.