Abstract
High mortality and hospitalization rates have seen Listeria monocytogenes as a foodborne pathogen of public health importance for many years and of particular concern for high-risk population groups. Food manufactures face an ongoing challenge in preventing the entry of L. monocytogenes into food production environments (FPEs) due to its ubiquitous nature. In addition to this, the capacity of L. monocytogenes strains to colonize FPEs can lead to repeated identification of L. monocytogenes in FPE surveillance. The contamination of food products requiring product recall presents large economic burden to industry and is further exacerbated by damage to the brand. Poor equipment design, facility layout, and worn or damaged equipment can result in Listeria hotspots and biofilms where traditional cleaning and disinfecting procedures may be inadequate. Novel biocontrol methods may offer FPEs effective means to help improve control of L. monocytogenes and decrease cross contamination of food. Bacteriophages have been used as a medical treatment for many years for their ability to infect and lyse specific bacteria. Endolysins, the hydrolytic enzymes of bacteriophages responsible for breaking the cell wall of Gram-positive bacteria, are being explored as a biocontrol method for food preservation and in nanotechnology and medical applications. Antibacterial proteins known as bacteriocins have been used as alternatives to antibiotics for biopreservation and food product shelf life extension. Essential oils are natural antimicrobials formed by plants and have been used as food additives and preservatives for many years and more recently as a method to prevent food spoilage by microorganisms. Competitive exclusion occurs naturally among bacteria in the environment. However, intentionally selecting and applying bacteria to effect competitive exclusion of food borne pathogens has potential as a biocontrol application. This review discusses these novel biocontrol methods and their use in food safety and prevention of spoilage, and examines their potential to control L. monocytogenes within biofilms in food production facilities.
Introduction
Listeria monocytogenes is a Gram-positive, rod shaped, facultative anaerobe capable of causing food borne illnesses particularly in high-risk population groups including the elderly, immune compromised, pregnant women, and neonates (Farber and Peterkin, 1991). While L. monocytogenes associated illness is not as common as that of other food borne pathogens like Salmonella, Campylobacter, or Escherichia coli, its mortality rate can be considered the highest. Approximately 30 % of invasive listeriosis cases lead to mortalities with most requiring hospitalization, and therefore demanding L. monocytogenes can be considered as a food borne pathogen of public health importance (Lomonaco et al., 2015; Véghová et al., 2016). Due to its ubiquitous nature, L. monocytogenes poses a food safety risk as it is frequently introduced into the processing environment through raw ingredients. L. monocytogenes can adhere to a variety of abiotic surfaces with some strains persisting for numerous years and acting as a source of continuous cross contamination (Fox E. et al., 2011; Coughlan et al., 2016; Colagiorgi et al., 2017).
Due to significant food safety risks, the control of L. monocytogenes has become a regulatory requirement that food business operators must adhere to. Regular cleaning, disinfecting, and sanitizing of food contact and non-food contact surfaces are required as part of a sanitation plan that also incorporates maintenance of equipment and buildings, pest control, and general hygiene. In addition, the implementation of good manufacturing practices and effective hazard analysis critical control point plan aids in reducing the risk of food borne illness (Drew and Clydesdale, 2015). However, L. monocytogenes is a difficult organism to eradicate and its presence still occurs even with the best management plans (Tompkin, 2002; Drew and Clydesdale, 2015).
While the exact mechanisms can be unclear for how L. monocytogenes is able to persist in food production environments (FPEs) so successfully, researchers have proposed that there are numerous factors at play. Poorly maintained equipment, surfaces, and unhygienic factory design can result in niches containing adequate nutrients, water, and protection from cleaning allowing bacteria to survive and grow while also introducing bacteria to subinhibitory levels of disinfectants (Carpentier and Cerf, 2011; Fox E.M. et al., 2011; Ibba et al., 2013; Møretrø et al., 2017). Typically disinfectants, when applied correctly, can sufficiently inhibit the colonization of introduced planktonic cells; however, dosing failures and applying disinfectants to wet surfaces can result in equipment being inadequately disinfected and bacteria being exposed to subinhibitory chemical levels (Martinez-Suarez et al., 2016; Møretrø et al., 2017). Incorporating desiccation processes has been shown to increase the effectiveness of disinfections procedures (Overney et al., 2017); however, an ample amount of drying time is difficult when continuous or even daily production runs are required. It is also important to note the difference between resistance, an increase in concentration or time required to exert the same reduction, and tolerance, an adaptation in a microbe’s susceptibility potentially the result of exposure to subinhibitory levels (Cerf et al., 2010; Ortega Morente et al., 2013). For example, some L. monocytogenes strains are known to carry genes for disinfectant chemical efflux pumps, such as qacH and bcrABC. The distribution of these genes tends to vary on a strain by strain basis instead of being unique to a specific lineage or subtype (Dutta et al., 2013; Ortiz et al., 2015; Møretrø et al., 2017). Although it has been reported that these genes only result in tolerance to quaternary ammonium compounds at levels far below the concentrations actually used in the food industry (Tezel and Pavlostathis, 2015), the ability to form biofilms is also a crucial factor in the survival of L. monocytogenes. Biofilms are composed of numerous cells attached to each other or an abiotic surface surrounded by an extracellular matrix containing a mixture of polysaccharides, proteins, and extracellular DNA (da Silva Fernandes et al., 2015; Fagerlund et al., 2017). This extracellular matrix provides a protective barrier around the internalized microbial cells from desiccation and heat, contributes to increased adhesion, and is a reservoir of nutrients (Colagiorgi et al., 2016). In addition, biofilms can impede the activity of antimicrobial agents as the matrix limits their diffusion potential and contains cells with differing susceptibility while also allowing for the acquisition of new genetic traits like those mentioned above through horizontal gene transfer. Further, biofilms typically consist of multiple species that can allow for the colonization of transient strains or provide increased attachment and survival to strains not typically good biofilm formers (Coughlan et al., 2016).
The Biocontrol Methods Movement
While tolerance to disinfectants and sanitizers is not considered as significant an issue as antibiotic resistance, their continued use and potential ineffectiveness against biofilms warrant new strategies for the control of L. monocytogenes. As consumers become more conscious of food safety significance, the use of novel biocontrol methods is gaining further interest. This return to biocontrol methods of microbes and plants has the potential to relieve some of the tolerance to disinfectants and decrease some of the selective pressures that their overuse has on maintaining resistance markers (Coughlan et al., 2016). Biocontrol methods with potential to act against listerial biofilms include bacteriophages, their endolysins, competitive bacterial species and their antimicrobial products, bacteriocins, and plant-derived products and will be discussed in this review.
Bacteriophages
The most abundant microorganism on earth, bacteriophages (phages) are viruses that infect bacteria for propagation, live naturally in the environment, and anywhere host bacteria are found (Bai et al., 2016; Pérez Pulido et al., 2016). Phages are classified based upon their morphology (head and tail, either contractile or non-contractile, or no tail), nucleic acid (double stranded or single stranded; deoxyribonucleic or ribonucleic acid), and life cycle, which is of most relevance for biocontrol. There are two types of life cycles phages can undergo after entering the bacterial cell: the lysogenic cycle (temperate phages) or the lytic cycle (Figure 1). Phages may be capable of a lysogenic cycle that converts to the lytic cycle in unfavorable conditions, or undergo a solely lytic life cycle. Temperate phages are not suitable as a biocontrol agent as integration into the host genome may result in increased pathogenicity through horizontal gene transfer (Hagens and Loessner, 2007; Salmond and Fineran, 2015). In comparison, lytic phages are ideal as a biocontrol agent due to their fast-lytic action.
FIGURE 1
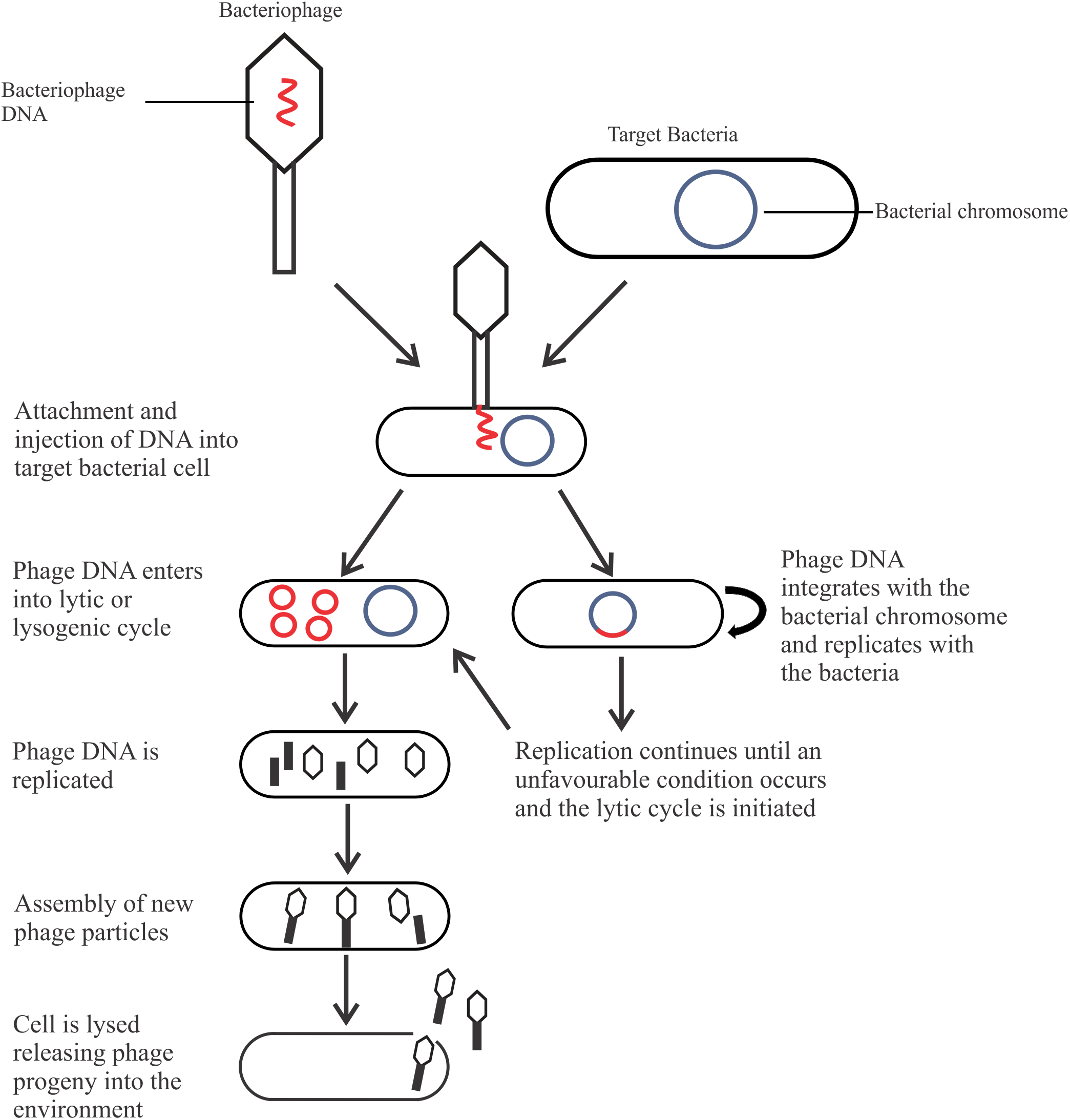
The life cycles of a bacteriophage.
Although identified over a hundred years ago, interest in phages has only recently been reignited with the rise of antibiotic resistance among bacteria (Hagens and Loessner, 2007). The utility of phages has included the treatment of diseases in humans and animals, typing of bacterial strains, decontaminating meat carcasses after slaughter, and targeted inactivation of pathogenic and spoilage bacteria on food contact and non-contact surfaces as well as surfaces of ready to eat products and during packaging and storage (Hagens and Loessner, 2007; Strauch et al., 2007). The application of phages as an innovative approach to control biofilms in the FPE is also beginning to be explored. While there has been great achievement in the use of phages from a therapeutic perspective, their success in the FPE is not as simple. Factors like the composition and structure of the biofilm, temperature, the metabolic state of the bacteria in the biofilm, the extracellular matrix in general, food components, and nutrients all provide additional challenges to the effectiveness of phage application (Parasion et al., 2014). While there have been some reports of phage resistance (Fister et al., 2016), it occurs more gradually than the development of antibiotic resistance as phages are able to mutate continuously, like bacteria, and resistance is further slowed by using a combination of phages active against the one bacterial species (Hagens and Loessner, 2007; Sadekuzzaman et al., 2017). There is a substantial amount of research conducted on phages’ ability to protect food from Listeria, with two commercial Listeria phage products, ListShieldTM and ListexTM P100 approved as food preservatives with the generally recognized as safe status since 2006. However, studies investigating the efficacy of these products and other Listeria phages against biofilms are few, with most having focused on ListexTM P100.
Biofilm maturity has the potential to reduce the efficacy of phage treatment, as well as any control method. Various studies have examined this concept utilizing preformed biofilms at various maturity levels, ranging from 24 h to 2 weeks, with most studies reporting a minimum 1-log reduction. Most studies to date have utilized stainless steel as the surface to form L. monocytogenes biofilms and examine the efficacy of bacteriophage treatments. This reflects the widespread presence of these surfaces, both food contact and non-contact in food processing environments. The success of bacteriophage treatments at inactivating L. monocytogenes biofilms on these surfaces, however, has shown mixed results. A number of studies demonstrated promising results when ListexTM P100 was applied to L. monocytogenes biofilms on stainless steel, with reductions in the order of 5-log being achieved (Soni and Nannapaneni, 2010; Montañez-Izquierdo et al., 2012). Both of these studies used an application treatment of 24 h at ambient room temperature. Iacumin et al. (2016) also applied ListexTM P100 for 24 h at 20°C onto stainless steel wafers and report the complete elimination of L. monocytogenes biofilm. This prolonged treatment application, however, in many cases is not practical in an FPE. In addition, Iacumin et al. (2016) pressed the stainless steel wafer onto an agar plate to replicate the process of cross-contamination in the FPE; however, it did not take into consideration the phage products ability or inability to act on biofilms in the crevices or corners where these might be thicker than a flat surface.
A shorter treatment time of 2 h was applied by Sadekuzzaman et al. (2017) when running a similar inactivation test with ListShieldTM; however, this was associated with a much lower inactivation of just 2-log when applied to L. monocytogenes biofilm on stainless steel. This was even less effective on a rubber surface, achieving a 1-log reduction in L. monocytogenes cell numbers. The results of Sadekuzzaman et al. (2017) also reflect those observed by Gutiérrez et al. (2017) who saw a similarly low inactivation achieved by a 4 h ListShieldTM treatment, typically 1-log or less. Although the latter study did show greater inactivation with ListexTM P100 under the same treatment conditions, the ListexTM P100 commercial phage preparation showed a reduced activity range, only capable of infecting 7 of the 11 strains tested. An important aspect in phage application is the ratio of phage to bacteria known as the multiplicity of infectivity. To increase the likelihood the phage will infect the bacterium, the phage needs to be at a higher ratio than the number of target bacterial cells (Montañez-Izquierdo et al., 2012). High multiplicity of infectivity has been reported to result in efficient phage treatment with one study recommending a multiplicity of infectivity around five was required for adequate reductions by ListexTM P100 (Montañez-Izquierdo et al., 2012).
Apart from temperature, multiplicity of infectivity, and treatment time, other factors may influence efficacy of biocontrol treatments, notably the presence of organic matter such as the food matrix. A further parameter which must be considered when examining efficacy of treatment on surfaces is the surface architecture itself, which may range from a smooth rendered surface to a scored surface with associated crevices which may be colonized by bacteria and their biofilms. Chaitiemwong et al. (2014) considered both surface crevices and food matrices (diluted food residues of ham, salmon, endive, or milk) when measuring the efficacy of ListexTM P100 treatment. Results suggested deeper crevice features on the surface decreased the treatment efficacy, with inactivation in the magnitude of > 3-log achieved on 0.2 mm crevices compared to the max 1.4-log CFU/mL observed in crevice depth of 5 mm. Of particular note was the difference seen when comparing the food matrix, with lower inactivation observed for milk and vegetable when compared with meat or fish. Ganegama Arachchi et al. (2013) mimicked conditions in fish processing and demonstrated the presence of fish protein led to a lower associated biofilm density compared to control stainless steel experiments when a fish protein matrix was added to the cultivation of L. monocytogenes biofilm on stainless steel. This highlights the complex role the food matrix may play in both biofilm formation and subsequent efficacy of bacteriophage treatment, demonstrating the need for further studies to understand the significance of food matrix on bacteriophage treatment efficacy.
Taken together, current literature detailing phage biocontrol studies directed at L. monocytogenes, such as those detailed above, shows differing success in their ability to decrease established biofilms. The often low reductions achieved demonstrate the challenges biofilms pose for not only bacteriophages but all control methods, but this is not to say that there is no place for phages as a potential biocontrol method. As with many disinfection regimes, additional interventions such as steps to loosen biofilm or remove organic matter can increase the success of phage treatments (Ganegama Arachchi et al., 2013). Further research considering multi-species biofilms and in-facility application will help determine the true potential of this biocontrol approach.
Endolysins
Endolysins (lysins) are hydrolytic enzymes required for bacteriophage dissemination from the host bacterial cell. They occur at the end of the lytic cycle to release the phage virions by breaking down peptidoglycan in the bacterial cell wall in what is termed lysis from within (Chan and Abedon, 2015; Schmelcher and Loessner, 2016). Researchers have harnessed lysins through protein expression production systems, generally in E. coli. Following purification of the lysin, it can be applied externally to the cell wall, thus not requiring phage infection, for biopreservation and biocontrol application (García et al., 2010). Lysins are grouped based upon the cell wall component they attack with the five main classes being N-acetylglucosaminidases, endo-β-N-acetlyglucosaminidases, lytic transglycosylases, endopeptidases, and N-acetylmuramoyl-L-alanine amidases (García et al., 2010; Schmelcher and Loessner, 2016). Lysins are highly specific with a narrow spectrum of activity making them host specific with some lysins only being active on the bacterial strain the phage was isolated from (Oliveira et al., 2012). In addition, they are fast acting and no development of resistance has been reported to date (Schmelcher and Loessner, 2016). Most research has occurred on Gram-positive bacteria using the lysis from without approach as the peptidoglycan layer is exposed. Although limited, antimicrobial activity of lysins on Gram-negative bacteria has been reported when used in conjunction with EDTA, a membrane permeablizer (Chan and Abedon, 2015).
The antimicrobial activity of lysins has mostly focused on infection control of staphylococcal bacteria. Other applications that have been considered include use in agriculture to prevent plant disease by either intense application of cell lysates expressing a chosen lysin or development of transgenic plants by incorporation of the lysin gene into the plant genome (Düring et al., 1993; Kim et al., 2004); as a rapid detection and imaging method of pathogenic bacteria (Schmelcher et al., 2010; Bai et al., 2016); and transformation of listerial bacteriophage endolysin encoding genes into dairy starter cultures as a biopreservation method (Gaeng et al., 2000). Antilisterial lysins isolated to date have predominately focused on the control of planktonic cells in vitro with promising results although further validation is required (Table 1). Only a few antilisterial lysins have been assessed in food products and the food matrix and environment have been found to affect the antimicrobial activity (Oliveira et al., 2012).
Table 1
| Endolysin | Reported findings | Use | Reference |
|---|---|---|---|
| Ply118 | Rapidly lysed all Listeria strains tested and against three Bacillus species. | BC, IC | Loessner et al., 1995 |
| Ply500 | BC, IC | ||
| Ply511 | Rapidly lysed all Listeria strains tested against. | BC, IC | |
| PlyP35 | Determined optimal temperature, NaCl, pH, and various ions conditions. | BC, IC | Schmelcher et al., 2012 |
| PlyP40 | Lysed L. monocytogenes strain and L. innocua. | BC, IC | Loessner and Schmelcher, 2010 |
| PlyP825 | Inhibited all growth in L. monocytogenes strains used. | BC, IC | Grallert et al., 2012 |
| PlyPSA | Determined crystalized structure | RMD | Korndörfer et al., 2006 |
| PlyP100 | Lysed all L. monocytogenes, Listeria strains in cheese, and a Bacillus subtilus strain tested against | BC, BP | Van Tassell et al., 2017 |
| LysZ5 | Lysed L. monocytogenes, L. innocua, and Listeria welshimeri; reduced L. monocytogenes numbers in soya milk. | BC, BP | Zhang et al., 2012 |
| PlyLM | Lysed all L. monocytogenes and L. innocua strains tested against; digested L. monocytogenes biofilms when combine with a protease. | BC | Simmons et al., 2012 |
Antilisterial lysins reported in literature, key summary, and application.
BC, biocontrol; IC, infection control; RMD, rapid multiplex detection; BP, biopreservation.
To date there is only one lysin, PlyLM, which has been tested against L. monocytogenes biofilms after 100 % susceptibility on planktonic L. monocytogenes and Listeria innocua cells was achieved (Simmons et al., 2012). PlyLM reduced the monolayer biofilm to the same level as the application of lysozyme and proteinase K. When used in combination with proteinase K, or both proteinase K and lysozyme, synergistic effects were observed, and the biofilm was effectively digested. However, biofilms were only grown for 24 h at 37°C, and therefore the efficacy of these enzymes under other conditions merits further investigation, for example, performance at lower temperatures which are more reflective of those of most FPEs. More research has been undertaken on staphylococcal biofilms, predominantly monospecies biofilms, which have achieved reductions in biofilm mass. Of interest is their efficacy against persister cells. Persister cells are metabolically inactive subpopulations of cells, which are “super-resistant” to antimicrobial agents such as antibiotics (Brooun et al., 2000; Wood, 2017). Studies have shown these persister cells occur as a subpopulation of bacterial biofilms, and as such can present a significant obstacle to biofilm inactivation by antimicrobials (Brooun et al., 2000; Singh et al., 2009). Several studies have shown a promising role for lysins to inactivate persister cells in biofilms (Gutiérrez et al., 2014; Schuch et al., 2017). The success being reported against staphylococcal biofilms suggests that the potential lysins may have against biofilms in a food production context, particularly in targeting Listeria biofilms, which are a significant problem in FPEs. Another phage enzyme, extracellular polysaccharide depolymerase, has also be shown to degrade biofilm EPS; however, they are highly specific to the strains the phage infects (Chan and Abedon, 2015). A similar approach targeting L. monocytogenes in biofilms could also present an alternative control measure.
Competitive Bacterial Species
Competitive exclusion is where one bacterial species competes with another species over resources and/or space in a habitat, successfully reducing the number of cells or excluding that species (Hibbing et al., 2010). This competitive exclusion can be the result of the production of antimicrobials such as bacteriocins, organic acids either acting directly against the species it is competing with or acting on the environment altering the pH, or alternatively physically outcompeting other bacterial species for nutrients and/or space and limiting normal survival or proliferation of those competitive species. This strategy is typically categorized into three components: competition, where planktonic cells of both species are co-cultured for a period of time; exclusion, where the antagonistic species are grown to a biofilm cell density prior to the addition of planktonic cells of the target species; or displacement, in which the target species are grown to biofilm cell density prior to addition of planktonic antagonists (Woo and Ahn, 2013; Pérez-Ibarreche et al., 2016). As biofilms protect microorganisms from chemical cleaners and disinfectants, the use of non-pathogenic microorganisms may assist sanitation approaches in controlling, preventing, or eradicating unwanted species like food borne pathogens.
Competitive exclusion studies typically pit planktonic cells of the antagonist species (i.e., the species which will exert a competitive exclusion effect) against planktonic cells of the target species in a competition assay, grown together for a period of time facilitating biofilm formation. Daneshvar Alavi and Truelstrup Hansen (2013) used a short incubation time of 72 h which resulted in a 1-log decrease in L. monocytogenes cell density after application of Serratia proteamaculans. A similar reduction was also reported by Fox et al. (2014) of L. monocytogenes biofilm cell density after 96 h when grown in co-culture with Janthinobacterium lividum. However, greater reductions have been reported when cells were incubated for longer periods with results around log 4.5 and 5.5 on stainless steel coupons and polytetrafluoroethylene, respectively (Pérez-Ibarreche et al., 2016). Zhao et al. (2004) also reported higher magnitude reductions of 7.8-log reduction over 28 days at 15°C by two bacterial isolates, Lactococcus lactis (Lc. lactis) and Enterococcus durans. In another experiment performed at 8°C for 28 days, four isolates, including the previous two isolates were also capable of reductions around 7-log units. However, the higher reductions reported by Zhao et al. (2004) and Pérez-Ibarreche et al. (2016) were produced by lactic acid bacteria (LAB) whose inhibitory activity has been studied extensively for many years, particularly as probiotics (Jeong and Frank, 1994).
The inhibitory effect of LAB was further explored by Guerrieri et al. (2009) and Gómez et al. (2016) as a preformed biofilm preventing L. monocytogenes biofilm formation as part of the exclusion strategy. Gómez et al. (2016) tested a variety of LAB strains and found reductions ranged from 4- to 7-log units over 24 and 48 h; however, by 72 h, L. monocytogenes growth had increased by almost half fold of the control indicating that these strains were only capable of exclusion within the first 24–48 h. However, Lc. lactis 368 strain was able to completely exclude the growth of L. monocytogenes for the entire period, although it should be noted that all experiments were performed at a relatively elevated temperature and as such lower temperatures reflective of many FPEs require further consideration. In comparison, Guerrieri et al. (2009) showed the potential of LAB bacteria at refrigeration temperatures with a Lactobacillus plantarum (Lb. plantarum) strain capable of a 4-log reduction over a 10-day period. Mariani et al. (2011) used the native biofilm microflora of wooden cheese ripening shelves to achieve a 1- to 2-log reduction over a 12-day period, although this reduction was less than that observed in Guerrieri et al. (2009) and Gómez et al. (2016).
The third strategy displacement, as reviewed by Woo and Ahn (2013), demonstrated that the use of planktonic antagonist LAB strains as a post-treatment control method targeting L. monocytogenes was less effective compared to pre-treatment, although two strains (Lactobacillus paracasei and Lactobacillus rhamnosus) were capable of a 3-log reduction in L. monocytogenes biofilm cell density over 24 h when incubated at 37°C.
While most studies are performed in laboratories, Zhao et al. (2006, 2013) took the concept of competitive exclusion a step further and looked at its applicability in poultry processing facilities. In a fresh poultry facility, two LAB strains (Lc. lactis and E. durans) were added to two enzyme-based cleaners and applied as a foam to selected drains four times in the first week and then two times for the following 3 weeks. Sampling continued for 18 weeks after the last treatment. Most drains experienced significant reductions within the first week after only four applications and all drains maintained lower levels of Listeria throughout the sampling period (Zhao et al., 2006). Importantly, two drains reported significant reductions 16 weeks after treatments finished. Similar parameters were applied to the application of the same strains at a ready to eat poultry processing facility. By the end of the first week of application, Listeria was not detected in five of the six drains with all drains reporting negative results between weeks 8 and 13 (Zhao et al., 2013). It should also be noted that the strains utilized were known to either possess nisin or other forms of antimicrobials; however, it was not elucidated if the inhibition was the result of the production of antimicrobials.
There have been some encouraging results in the use of LAB against L. monocytogenes biofilm cells in laboratory-based experiments (Table 2); however, very few have been trialed in actual FPEs, apart from Zhao et al. (2006, 2013). The results from their two studies have shown promising results as an alternative control method utilizing E. durans and Lc. lactis; however, further longitudinal research surrounding the in-facility application is required. In addition, the application of other bacterial species identified in some of the studies mentioned above, for example, J. lividum and S. proteamaculans, warrants in-facility testing. However, it should be noted that the LAB strains utilized for in-facility application studies were isolated from the production environment indicating that specific strains may work best in the environment they were isolated from and these strains may vary depending on the food industry.
Table 2
| Bacterial species | Mode of action | Studies |
|---|---|---|
| S. proteamaculans | Sánchez et al. (2010) identified a bacteriocin-like substance was produced at low temperatures capable of inhibiting L. monocytogenes. Inhibition was suggested to be the result of Jameson effect. | Sánchez et al., 2010; Daneshvar Alavi and Truelstrup Hansen, 2013 |
| J. lividum | Specific strain utilized not tested for antimicrobial compounds. J. lividium are reported to have antibacterial compounds capable of inhibiting Gram-positive bacteria (O’Sullivan et al., 1990). | Fox et al., 2014 |
| Lc. lactis | Neither of the studies by Zhao et al. tested for production of a bacteriocin; however, this species has previously be reported to produce nisin. | Zhao et al., 2004, 2006, 2013 |
| E. durans | Neither of the studies tested for the bacteriocin; however, this species has previously be reported to produce enterocin. | Zhao et al., 2004, 2006, 2013 |
| Lb. plantarum 396/1 | Inhibition was attributed to production of an organic acid. | Guerrieri et al., 2009 |
| Lb. paracasei | May be the result of competition for sites and resources. As a probiotic strain it may produce bacteriocin, organic acid or hydrogen peroxide. | Woo and Ahn, 2013 |
| Lb. rhamnosus | May be the result of competition for sites and resources. As a probiotic strain, it may produce bacteriocin, organic acid or hydrogen peroxide. A previous study isolated an antilisterial bacteriocin from this species (Jeong and Moon, 2015). | Woo and Ahn, 2013 |
| Lb. sakei | Bacteriocin producing strain. | Pérez-Ibarreche et al., 2016 |
| LAB – Lc. lactis 368, Lb. helveticus 354, Lb. casei 40, and W. viridescens 113 | Not identified as bacteriocin-producing strains. May be result of biosurfactants, or exclusion by trapping (killing cells embedded in biofilm). | Gómez et al., 2016 |
| Native microbial flora of cheese ripening wooden shelves | Established biofilms on active cheese ripening wooden shelves were used. Inhibition may have been the result of competition for sites and nutrients. | Mariani et al., 2011 |
Bacterial species active against L. monocytogenes and purported mode of action.
Houry et al. (2012) reported the use of bacterial species in a novel biocontrol approach. In the study, they identified a subpopulation of bacilli known as bacterial swimmers which were capable of creating transient pores within the biofilm structure. By pre-treating Staphylococcus aureus biofilms with bacterial swimmers, which also produced an anti-stapylococcal bactericide, they achieved a greater inactivation of S. aureus in biofilm by facilitating access of toxic substances in the environment into the biofilm.
Bacteriocins
An important component of the competitive survival strategy of bacteria is the production of antimicrobial products. One group of ribosomally synthesized antimicrobials are the heat stable peptides known as bacteriocins (Cotter et al., 2005; Gálvez et al., 2008; Winkelströter et al., 2015). It has been suggested that most bacteria produce at least one bacteriocin and LAB are known to be prolific producers (Cotter et al., 2005). Most bacteriocins have a narrow spectrum of activity, that is, they are active against the same species that produces them but the producer is immune to them, while some have a broad spectrum of activity acting on members within the same genus as well as other genera and species (Cotter et al., 2005). The mode of activity varies depending on the particular class of bacteriocin and can include pore formation, or inhibition of key cellular processes such as peptidoglycan production, DNA replication, mRNA, or protein synthesis, to name a few (Cotter et al., 2005). There are two main groups: Class I (also known as lantibiotics), peptides that undergo post-translational changes, and Class II, which do not (Cotter et al., 2013). Among the most well-characterized and successful bacteriocins to date is nisin, a Class I bacteriocin from Lc. lactis which has been approved for use in food as a preservative/additive by the World Health Organization, European Union, and the United States Food and Drug Authority (Cotter et al., 2005). A great deal of research has gone into identifying more bacteriocins active against L. monocytogenes planktonic cells and biofilms, an important arena as nisin resistance is slowly being reported.
Most studies can be classified into two groups based upon how the bacteriocin is applied: either as whole bacterial cells known or suspected of bacteriocin production, or alternatively the bacteriocin extract itself, applied either as a crude or semi-purified product. Their utility against preformed L. monocytogenes biofilms of varying times has been the subject of numerous studies, with some reporting promising results. For example, Gómez et al. (2016) assessed Lc. lactis, Lactobacillus sakei, and Lactobacillus curvatus, all known to produce nisin Z, sakacine A, and sakacine P, respectively, against 48 h preformed biofilms. Lb. sakei and Lb. curvatus were capable of complete inactivation over 72 h whereas the two Lc. lactis strains provided a 6-log reduction by the end of the test period. Winkelströter et al. (2015), however, were unable to produce results of a similar magnitude when L. monocytogenes was co-cultured with Lb. paraplantarum, only achieving 2-log inactivation at 24 and 48 h before decreasing by 72 h. Guerrieri et al. (2009) took an alternative approach and reported that Lb. plantarum and Enterococcus casseliflavus were able to inactivate L. monocytogenes 7-day preformed biofilms by 3.9- and 3.7-logs over a 10 day-period. Importantly, the results could be associated with bacteriocin production, as no changes to the pH were observed.
Another technique is extracting the bacteriocin in the form of cell-free supernatant (CFS), as a crude bacteriocin fermentate or semi-purifying the product. The antimicrobial activity of CFS has shown mixed success in co-inoculation studies to prevent the formation of biofilms by L. monocytogenes, with Camargo et al. (2016) reporting significant reductions after 24 h, whereas Bolocan et al. (2017) only observed between 1.6- and 3.6-log CFU/cm2 reduction after 72 h depending on the media used. In the latter study, however, the CFS extract which produced the highest reduction was from an isolate known to also produce an organic acid which was not removed, and therefore this result may not be associated solely to the antimicrobial activity of the bacteriocin. When Camargo et al. (2016) applied the CFS to 24 h preformed biofilms for 2.5 h, they found biofilm formation continued in some isolates.
Other researchers have compared the two methods, bacterial cells and extracts again with varying results. García-Almendárez et al.’s (2008) analysis on 4-day preformed biofilms demonstrated a crude bacteriocin fermentate from Lc. lactis known to produce nisin A was capable of a 2.7-log reduction over 24 h. However, a greater reduction over 5-logs was achieved when the Lc. lactis was applied for 6 h, then rinsed, and placed in a desiccator for five days. Whereas, Winkelströter et al. (2011) co-inoculated L. monocytogenes with Lb. sakei or its CFS and found that any decreases observed in the first 24 h were diminished with time, as results at 48 h were comparable to the pure culture levels. A promising approach by Pérez-Ibarreche et al. (2016) involved the supplementation of Lb. sakei cells with a semi-purified bacteriocin for 6 h, which resulted in a twofold reduction in L. monocytogenes numbers on the stainless steel surface, or an additional 1-log reduction on polytetrafluoroethylene.
As mentioned previously, the bacteriocin nisin has been approved for commercial purposes and has paved the way as an alternative biocontrol method. Research into bacteriocins has been performed with comparable results to the other non-commercial bacteriocins discussed above. Minei et al. (2008) found that nisin was capable of inhibiting L. monocytogenes biofilm formation for 9 h on stainless steel coupons, and although cell growth did recommence after this time, a 3.5-log inactivation was still maintained by 48 h. On the other hand, Henriques and Fraqueza (2017) shortened the treatment time to 5 min and even at the highest concentration, no activity was recorded, although activity was defined as a ≥ 5-log decrease.
From the above, it is obvious that results vary significantly depending on if bacteriocin producing bacterial cells or the bacteriocin extracts is used. Results from bacteriocin extracts can be correlated to the antimicrobial action of the bacteriocin with greater certainty; however, additional analysis is required particularly when whole cells are used to help ensure that the measured inhibition is not the result of competitive exclusion or the production of other antimicrobials such as organic acids. The co-inoculation and preformed biofilm studies reflect the ability of the bacteriocin to either prevent the formation or affect the removal of established biofilms in the FPE; however, the length of time the biofilms are grown for prior to the bacteriocin being applied also affects the antimicrobial activity as mature biofilms may provide better resistance. Although several studies show that promising results most require additional analysis at temperatures and other environmental conditions mirroring the FPE to identify potential candidates suitable for further testing. With the potential resistance to nisin arising, the identification of other bacteriocins is essential. In addition, the application of synergistic antimicrobials to further combat the development of resistance should be considered.
Plant-Derived Antimicrobial Products – Essential Oils
An alternative to the use of chemicals, microorganisms, or their derivatives is the use of plant-derived antimicrobial products such as essential oils (EOs). Herbs and spices are commonly known to exhibit antimicrobial activity and have been used by various cultures for flavoring, as a food preservative or for medicinal purposes. EOs play a key role in protecting plants from bacteria, fungi, viruses, insects, and animals (Perricone et al., 2015). Traditional distillation, cold press/expressing, solvent extractions, and enfleurage methods have been used to extract EOs from plant-derived materials; more recently, modern techniques including microwave or ultra sound assisted extraction, pressurized extractions, and super critical fluid extraction have been used to obtain EOs from a variety of plant sources (including roots, wood, bark, twigs, leaves, seeds, buds, flowers, and fruits). However, the constituents and compositions of EOs vary significantly from high concentrations to trace amounts based upon the plant part, plant age, and extraction method used, in turn influencing their antimicrobial activity (Lemberkovics et al., 2004; Reyes-Jurado et al., 2014; Perricone et al., 2015; Xia et al., 2017). Key molecules in EOs with the most effective antibacterial activity are typically from aldehyde and phenol chemical classes which include compounds such as cinnamaldehyde, carvacrol, eugenol, or thymol (Bakkali et al., 2008; Perricone et al., 2015). EOs are able to permeabilize the cell membrane resulting in the leakage of ions or other cell content, and may also disrupt key genetic functions and/or cellular components like proteins, polysaccharides, phospholipids, fatty acids, and essential enzymes due to the lipophilic nature of EOs (Bakkali et al., 2008; de Oliveira et al., 2010, 2012a; Perricone et al., 2015).
While there are thousands of EOs described, it is reported around 300 of these have generally recognized as safe approval and are used commercially for flavoring or fragrance; however, more detailed information is required for their use as a biocontrol agent (Burt, 2004; Reyes-Jurado et al., 2014). Most research surrounding the antimicrobial activity of EOs focuses on their effects on planktonic cells of food spoilage and pathogenic bacteria either in standard laboratory conditions or in their application on food items. This application on food as a biocide has major limitations as higher concentrations are required potentially interfering with the sensory attributes of the food (Burt, 2004; Chorianopoulos et al., 2008). In addition, some components of food items, mainly fats, proteins, carbohydrates, water, salt, antioxidants, pH, and other preservatives or additives used may impact upon the activity of the EOs (Perricone et al., 2015). Further research is required to understand the impact EOs have on bacterial pathogens and in particular their ability to prevent or eradicate biofilms in FPEs. Some research is occurring within this space; however, there is limited research against L. monocytogenes biofilms with a few studies looking at the extracted EOs, the active components of specific EOs, or altering the EO chemical composition. de Oliveira et al. (2010) assessed the EOs from fresh citronella (Cymbopogon nardus) and lemongrass (Cymbopogon citratus) leaves applied alone or in combination; however, it was the Citronella EO which demonstrated the highest reductions against both the 3 and 240 h preformed biofilms with complete reduction after 60 min of application. Similar results reported in another study by de Oliveira et al. (2012b) found 2% (vol/vol) Chinese cinnamon extract (Cinnamomum cassia) was capable of reducing a 48 h preformed biofilm to below the detection limit (2.84-log CFU/cm2) after 20 min; however, both of these studies applied the EOs at temperatures above 20°C.
Essential oils contain a mixture of major and minor molecules responsible for their antimicrobial activity with some of the major components being explored further. The active components of clove (eugenol) and spearmint (carvone) EOs were tested on a 6 h preformed L. monocytogenes biofilm but were found to increase biofilm mass by Leonard et al. (2010). Citral and nerol, in contrast, both major components from lemongrass (C. citratus) and Lippia rehmannii (nerol only), displayed a similar reduction as the positive control ciprofloxacin.
Additional microbial species can also impact upon the activity of the EO or active component. For example, Leonard et al.’s (2010) study as mentioned above was on L. monocytogenes monospecies biofilms and reported a mixture of results among the EO and the various active components tested, whereas de Oliveira et al. (2012a) looked at the activity of Chinese cinnamon and its active component, Cinnamaldehyde, on a mixed biofilm of L. monocytogenes and enteropathogenic E. coli on stainless steel coupons dipped in reconstituted whole milk. The EO and cinnamaldehyde were both capable of reducing the mixed biofilm to below the detection limit of 2.84-log CFU/cm2 whereas the EO and active components only provided reductions just over 2-logs on the L. monocytogenes biofilm. Chorianopoulos et al. (2008) examined the EO and hydrosol (by-product of the steam distillation) of Satureja thymbra (Pink Savory) and showed similar results when grown in a mixed biofilm with a food borne pathogen (L. monocytogenes and Salmonella enterica) and a spoilage bacterium (Pseudomonas putida). It was noted that the optimized application time was 60 min and any increase in time provided no additional reduction. The impact other microbial players may have on the activity of EOs requires further exploration in order to gain insights into the various relationships at play.
A common problem for the use of EOs as a biocontrol method on food products is the associated impacts on taste at concentrations required for appropriate antimicrobial effect. A process to concentrate the EOs for application at a lower volume with the same potentially high antimicrobial activity may be required in the case of some EOs. Krogsgård Nielsen et al. (2017) looked at emulsifying and encapsulating isoeugenol oil to increase the antimicrobial effectiveness at a smaller volume with the addition of electrostatic forces to attract negatively charged bacteria to positively charged EOs. Although the concept of emulsification and encapsulation sounds promising, the minimal biofilm eradication concentrations (MBECs) for the coated and uncoated emulsified products were only half a log lower than the pure isoeugenol when tested in standard laboratory medium at three temperatures (6, 12, and 25°C) and no difference was observed in carrot juice. This observation requires further exploration as the reductions in the MBEC did not correlate to observations under confocal microscopy. Of note was the morphological changes observed in the mixed biofilms of Pseudomonas fluorescens and S. aureus from uniform layers to clusters of numerous cells, which requires further research to determine if there are any implications.
As mentioned previously, the use of EOs at concentrations to exhibit sufficient antimicrobial activity has the potential to impart undesirable flavor and when applied in an FPE may also result in an excessive sensorial impact. In addition, the interactions of EOs with components of the food matrix from food debris may also impact on the applicability of EOs in food environments. Only a few studies have investigated the application of EOs to disrupt or prevent the formation of biofilms. Further research on parameters specific to industry will allow a better decision on the application of EOs as an alternative or supplementary biocontrol method.
Concluding Remarks
While current sanitation processes are effective against planktonic cells, the potential for tolerant strains to increase due to interactions at subinhibitory levels and the potential reliance on them as antimicrobials, as the case in the health industry, is a cause for concern. The ability to eradicate established biofilms and prevent new biofilms from being formed is a challenging task which food production managers are charged with, as biofilms can present increased food safety risks. A useful tool in understanding the microbial community is metagenomics analysis of the FPE. By understanding the FPE microbiome, valuable information can be gained regarding persistence or transience of strains. This facilitates source tracking of persistent strains, can identify other microbial species that may provide either a positive or negative effect on the target strain, and can identify strains surviving the disinfection processes (Dass and Anandappa, 2017; Doyle et al., 2017). From this information, the appropriate biocontrol method can then be determined. There have been some significant advances in the development of biocontrol methods, particularly bacteriophages that have progressed to commercial products with the results of some studies validating their progression to commercialization. The use of competitive bacterial species has also showed some promising results with the concept of utilizing antagonist strains isolated from the production environment providing individualized treatment options. Bacteriocins and endolysins have also shown their ability to significantly reduce established biofilms; however, they typically require some form of purification process to achieve these results. The sensory implications of EOs at concentrations required to exert antimicrobial effects are a limiting factor in their use as a sole biocontrol method, and therefore they may find more appropriate utility as a supplementary method targeting non-food contact surfaces. However, like all biocontrol methods, efficacy can be impacted by a variety of factors including temperature or time the control method was applied for, the use of one species or multiple species biofilms, biofilm growth method, or surface matrix composition. Standardization in the assessment of novel biocontrol methods against biofilms is required, in addition to assessment under conditions reflective of FPEs before appropriate comparisons can be made.
Statements
Author contributions
JG and EF conceived the study and drafted the manuscript. All authors corrected and approved the manuscript.
Funding
This work was supported by University of Tasmania and CSIRO joint funding. JG was a recipient of University of Tasmania and CSIRO Ph.D. Scholarship.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The reviewer GP and handling Editor declared their shared affiliation.
References
1
Bai J. Kim Y. T. Ryu S. Lee J. H. (2016). Biocontrol and rapid detection of food-borne pathogens using bacteriophages and endolysins.Front. Microbiol.7:474. 10.3389/fmicb.2016.00474
2
Bakkali F. Averbeck S. Averbeck D. Idaomar M. (2008). Biological effects of essential oils - A review.Food Chem. Toxicol.46446–475. 10.1016/j.fct.2007.09.106
3
Bolocan A. S. Pennone V. O’connor P. M. Coffey A. Nicolau A. I. Mcauliffe O. et al (2017). Inhibition of Listeria monocytogenes biofilms by bacteriocin-producing bacteria isolated from mushroom substrate.J. Appl. Microbiol.122279–293. 10.1111/jam.13337
4
Brooun A. Liu S. Lewis K. (2000). A dose-response study of antibiotic resistance in Pseudomonas aeruginosa biofilms.Antimicrob. Agents Chemother.44640–646. 10.1128/AAC.44.3.640-646.2000
5
Burt S. (2004). Essential oils: their antibacterial properties and potential applications in foods - A review.Int. J. Food Microbiol.94223–253. 10.1016/j.ijfoodmicro.2004.03.022
6
Camargo A. C. de Paula O. A. Todorov S. D. Nero L. A. (2016). In vitro evaluation of bacteriocins activity against Listeria monocytogenes biofilm formation.Appl. Biochem. Biotechnol.1781239–1251. 10.1007/s12010-015-1941-3
7
Carpentier B. Cerf O. (2011). Review - Persistence of Listeria monocytogenes in food industry equipment and premises.Int. J. Food Microbiol.1451–8. 10.1016/j.ijfoodmicro.2011.01.005
8
Cerf O. Carpentier B. Sanders P. (2010). Tests for determining in-use concentrations of antibiotics and disinfectants are based on entirely different concepts: “resistance” has different meanings.Int. J. Food Microbiol.136247–254. 10.1016/j.ijfoodmicro.2009.10.002
9
Chaitiemwong N. Hazeleger W. C. Beumer R. R. (2014). Inactivation of Listeria monocytogenes by disinfectants and bacteriophages in suspension and stainless steel carrier tests.J. Food Prot.772012–2020. 10.4315/0362-028X.JFP-14-151
10
Chan B. K. Abedon S. T. (2015). Bacteriophages and their enzymes in biofilm control.Curr. Pharm. Des.2185–99. 10.2174/1381612820666140905112311
11
Chorianopoulos N. G. Giaouris E. D. Skandamis P. N. Haroutounian S. A. Nychas G. J. E. (2008). Disinfectant test against monoculture and mixed-culture biofilms composed of technological, spoilage and pathogenic bacteria: bactericidal effect of essential oil and hydrosol of Satureja thymbra and comparison with standard acid-base sanitizers.J. Appl. Microbiol.1041586–1596. 10.1111/j.1365-2672.2007.03694.x
12
Colagiorgi A. Bruini I. Di Ciccio P. A. Zanardi E. Ghidini S. Ianieri A. (2017). Listeria monocytogenes biofilms in the wonderland of food industry.Pathogens6:E41. 10.3390/pathogens6030041
13
Colagiorgi A. Di Ciccio P. Zanardi E. Ghidini S. Ianieri A. (2016). A look inside the Listeria monocytogenes biofilms extracellular matrix.Microorganisms4:22. 10.3390/microorganisms4030022
14
Cotter P. D. Hill C. Ross R. P. (2005). Bacteriocins: developing innate immunity for food.Nat. Rev. Microbiol.3777–788. 10.1038/nrmicro1273
15
Cotter P. D. Ross R. P. Hill C. (2013). Bacteriocins - a viable alternative to antibiotics?Nat. Rev. Microbiol.1195–105. 10.1038/nrmicro2937
16
Coughlan L. M. Cotter P. D. Hill C. Alvarez-Ordóñez A. (2016). New weapons to fight old enemies: novel strategies for the (bio)control of bacterial biofilms in the food industry.Front. Microbiol.7:1641. 10.3389/fmicb.2016.01641
17
da Silva Fernandes M. Kabuki D. Y. Kuaye A. Y. (2015). Behavior of Listeria monocytogenes in a multi-species biofilm with Enterococcus faecalis and Enterococcus faecium and control through sanitation procedures.Int. J. Food Microbiol.2005–12. 10.1016/j.ijfoodmicro.2015.01.003
18
Daneshvar Alavi H. E. Truelstrup Hansen L. (2013). Kinetics of biofilm formation and desiccation survival of Listeria monocytogenes in single and dual species biofilms with Pseudomonas fluorescens, Serratia proteamaculans or Shewanella baltica on food-grade stainless steel surfaces.Biofouling291253–1268. 10.1080/08927014.2013.835805
19
Dass S. C. Anandappa A. (2017). Food factory genomics: where big data drives quality and food safety.Food Prot. Trends37368–374.
20
de Oliveira M. M. M. Brugnera D. F. Cardoso M. D. G. Alves E. Piccoli R. H. (2010). Disinfectant action of Cymbopogon sp. essential oils in different phases of biofilm formation by Listeria monocytogenes on stainless steel surface.Food Control21549–553. 10.1016/j.foodcont.2009.08.003
21
de Oliveira M. M. M. Brugnera D. F. Do Nascimento J. A. Batista N. N. Piccoli R. H. (2012a). Cinnamon essential oil and cinnamaldehyde in the control of bacterial biofilms formed on stainless steel surfaces.Eur. Food Res. Technol.234821–832. 10.1007/s00217-012-1694-y
22
de Oliveira M. M. M. Brugnera D. F. Do Nascimento J. A. Piccoli R. H. (2012b). Control of planktonic and sessile bacterial cells by essential oils.Food Bioprod. Process.90809–818. 10.1016/j.fbp.2012.03.002
23
Doyle C. J. O’toole P. W. Cotter P. D. (2017). Metagenome-based surveillance and diagnostic approaches to studying the microbial ecology of food production and processing environments.Environ. Microbiol.194382–4391. 10.1111/1462-2920.13859
24
Drew C. A. Clydesdale F. M. (2015). New food safety law: effectiveness on the ground.Crit. Rev. Food Sci. Nutr.55689–700. 10.1080/10408398.2011.654368
25
Düring K. Porsch P. Fladung M. Lörz H. (1993). Transgenic potato plants resistant to the phytopathogenic bacterium Erwinia carotovora.Plant J.3587–598. 10.1046/j.1365-313X.1993.03040587.x
26
Dutta V. Elhanafi D. Kathariou S. (2013). Conservation and distribution of the benzalkonium chloride resistance cassette bcrABC in Listeria monocytogenes.Appl. Environ. Microbiol.796067–6074. 10.1128/AEM.01751-13
27
Fagerlund A. Møretrø T. Heir E. Briandet R. Langsruda S. (2017). Cleaning and disinfection of biofilms composed of Listeria monocytogenes and background microbiota from meat processing surfaces.Appl. Environ. Microbiol.10.1128/AEM.01046-17[Epub ahead of print].
28
Farber J. M. Peterkin P. I. (1991). Listeria monocytogenes, a food-borne pathogen.Microbiol. Rev.55476–511.
29
Fister S. Robben C. Witte A. K. Schoder D. Wagner M. Rossmanith P. (2016). Influence of environmental factors on phage–bacteria interaction and on the efficacy and infectivity of phage P100.Front. Microbiol.7:1152. 10.3389/fmicb.2016.01152
30
Fox E. Hunt K. O’brien M. Jordan K. (2011). Listeria monocytogenes in Irish farmhouse cheese processing environments.Int. J. Food Microbiol.145(Suppl. 1), S39–S45. 10.1016/j.ijfoodmicro.2010.10.012
31
Fox E. M. Leonard N. Jordan K. (2011). Physiological and transcriptional characterization of persistent and nonpersistent Listeria monocytogenes isolates.Appl. Environ. Microbiol.776559–6569. 10.1128/AEM.05529-11
32
Fox E. M. Solomon K. Moore J. E. Wall P. G. Fanning S. (2014). Phylogenetic profiles of in-house microflora in drains at a food production facility: comparison and biocontrol implications of Listeria-positive and -negative bacterial populations.Appl. Environ. Microbiol.803369–3374. 10.1128/AEM.00468-14
33
Gaeng S. Scherer S. Neve H. Loessner M. J. (2000). Gene cloning and expression and secretion of Listeria monocytogenes bacteriophage-lyric enzymes in Lactococcus lactis.Appl. Environ. Microbiol.662951–2958. 10.1128/AEM.66.7.2951-2958.2000
34
Gálvez A. López R. L. Abriouel H. Valdivia E. Omar N. B. (2008). Application of bacteriocins in the control of foodborne pathogenic and spoilage bacteria.Crit. Rev. Biotechnol.28125–152. 10.1080/07388550802107202
35
Ganegama Arachchi G. J. Cridge A. G. Dias-Wanigasekera B. M. Cruz C. D. Mcintyre L. Liu R. et al (2013). Effectiveness of phages in the decontamination of Listeria monocytogenes adhered to clean stainless steel, stainless steel coated with fish protein, and as a biofilm.J. Ind. Microbiol. Biotechnol.401105–1116. 10.1007/s10295-013-1313-3
36
García P. Rodríguez L. Rodríguez A. Martínez B. (2010). Food biopreservation: promising strategies using bacteriocins, bacteriophages and endolysins.Trends Food Sci. Technol.21373–382. 10.1016/j.tifs.2010.04.010
37
García-Almendárez B. E. Cann I. K. O. Martin S. E. Guerrero-Legarreta I. Regalado C. (2008). Effect of Lactococcus lactis UQ2 and its bacteriocin on Listeria monocytogenes biofilms.Food Control19670–680. 10.1016/j.foodcont.2007.07.015
38
Gómez N. C. Ramiro J. M. P. Quecan B. X. V. de Melo Franco B. D. G. (2016). Use of potential probiotic lactic acid bacteria (LAB) biofilms for the control of Listeria monocytogenes, Salmonella Typhimurium, and Escherichia coli O157: H7 biofilms formation.Front. Microbiol.7:863. 10.3389/fmicb.2016.00863
39
Grallert H. Molinario S. Kaps J. (2012). Novel Listeria bacteriophage p825n and uses thereof. U.S. Patent No. WO 2012159774 A1.
40
Guerrieri E. De Niederhäusern S. Messi P. Sabia C. Iseppi R. Anacarso I. et al (2009). Use of lactic acid bacteria (LAB) biofilms for the control of Listeria monocytogenes in a small-scale model.Food Control20861–865. 10.3389/fmicb.2016.00863
41
Gutiérrez D. Rodríguez-Rubio L. Fernández L. Martínez B. Rodríguez A. García P. (2017). Applicability of commercial phage-based products against Listeria monocytogenes for improvement of food safety in Spanish dry-cured ham and food contact surfaces.Food Control731474–1482. 10.1016/j.foodcont.2016.11.007
42
Gutiérrez D. Ruas-Madiedo P. Martínez B. Rodríguez A. García P. (2014). Effective removal of staphylococcal biofilms by the endolysin LysH5.PLoS One9:e107307. 10.1371/journal.pone.0107307
43
Hagens S. Loessner M. J. (2007). Application of bacteriophages for detection and control of foodborne pathogens.Appl. Microbiol. Biotechnol.76513–519. 10.1007/s00253-007-1031-8
44
Henriques A. R. Fraqueza M. J. (2017). Biofilm-forming ability and biocide susceptibility of Listeria monocytogenes strains isolated from the ready-to-eat meat-based food products food chain.LWT Food Sci. Technol.81180–187. 10.1016/j.lwt.2017.03.045
45
Hibbing M. E. Fuqua C. Parsek M. R. Peterson S. B. (2010). Bacterial competition: surviving and thriving in the microbial jungle.Nat. Rev. Microbiol.815–25. 10.1038/nrmicro2259
46
Houry A. Gohar M. Deschamps J. Tischenko E. Aymerich S. Gruss A. et al (2012). Bacterial swimmers that infiltrate and take over the biofilm matrix.Proc. Natl. Acad. Sci. U.S.A.10913088–13093. 10.1073/pnas.1200791109
47
Iacumin L. Manzano M. Comi G. (2016). Phage inactivation of Listeria monocytogenes on San Daniele dry-cured ham and elimination of biofilms from equipment and working environments.Microorganisms4:E4. 10.3390/microorganisms4010004
48
Ibba M. Cossu F. Spanu V. Virdis S. Spanu C. Scarano C. et al (2013). Listeria monocytogenes contamination in dairy plants: evaluation of Listeria monocytogenes environmental contamination in two cheese-making plants using sheeps milk.Ital. J. Food Saf.2109–112. 10.4081/ijfs.2013.e31
49
Jeong D. K. Frank J. F. (1994). Growth of Listeria monocytogenes at 10°C in biofilms with microorganisms isolated from meat and dairy processing environments.J. Food Prot.57576–586. 10.4315/0362-028X-57.7.576
50
Jeong Y.-J. Moon G.-S. (2015). Antilisterial bacteriocin from Lactobacillus rhamnosus CJNU 0519 presenting a narrow antimicrobial spectrum.Korean J. Food Sci. Anim. Resour.35137–142. 10.5851/kosfa.2015.35.1.137
51
Kim W. S. Salm H. Geider K. (2004). Expression of bacteriophage phiEa1h lysozyme in Escherichia coli and its activity in growth inhibition of Erwinia amylovora.Microbiology1502707–2714. 10.1099/mic.0.27224-0
52
Korndörfer I. P. Danzer J. Schmelcher M. Zimmer M. Skerra A. Loessner M. J. (2006). The crystal structure of the bacteriophage PSA endolysin reveals a unique fold responsible for specific recognition of Listeria cell walls.J. Mol. Biol.364678–689. 10.1016/j.jmb.2006.08.069
53
Krogsgård Nielsen C. Kjems J. Mygind T. Snabe T. Schwarz K. Serfert Y. et al (2017). Antimicrobial effect of emulsion-encapsulated isoeugenol against biofilms of food pathogens and spoilage bacteria.Int. J. Food Microbiol.2427–12. 10.1016/j.ijfoodmicro.2016.11.002
54
Lemberkovics É. Kéry Á. Simándi B. Kakasy A. Balázs A. Héthelyi É. et al (2004). Influence of extraction methods on the composition of essential oils.Acta Pharm. Hung.74166–170.
55
Leonard C. M. Virijevic S. Regnier T. Combrinck S. (2010). Bioactivity of selected essential oils and some components on Listeria monocytogenes biofilms.South Afr. J. Bot.76676–680. 10.1016/j.sajb.2010.07.002
56
Loessner M. Schmelcher M. (2010). New endolysin plyp40. U.S. Patent No. US20140079727A1.
57
Loessner M. J. Wendlinger G. Scherer S. (1995). Heterogeneous endolysins in Listeria monocytogenes bacteriophages: a new class of enzymes and evidence for conserved holin genes within the siphoviral lysis cassettes.Mol. Microbiol.161231–1241. 10.1111/j.1365-2958.1995.tb02345.x
58
Lomonaco S. Nucera D. Filipello V. (2015). The evolution and epidemiology of Listeria monocytogenes in Europe and the United States.Infect. Genet. Evol.35172–183. 10.1016/j.meegid.2015.08.008
59
Mariani C. Oulahal N. Chamba J. F. Dubois-Brissonnet F. Notz E. Briandet R. (2011). Inhibition of Listeria monocytogenes by resident biofilms present on wooden shelves used for cheese ripening.Food Control221357–1362. 10.1016/j.ijfoodmicro.2008.06.028
60
Martinez-Suarez J. V. Ortiz S. Lopez-Alonso V. (2016). Potential impact of the resistance to quaternary ammonium disinfectants on the persistence of Listeria monocytogenes in food processing environments.Front. Microbiol.7:638. 10.3389/fmicb.2016.00638
61
Minei C. C. Gomes B. C. Ratti R. P. D’angelis C. E. M. De Martinis E. C. P. (2008). Influence of peroxyacetic acid and nisin and coculture with Enterococcus faecium on Listeria monocytogenes biofilm formation.J. Food Prot.71634–638. 10.4315/0362-028X-71.3.634
62
Montañez-Izquierdo V. Y. Salas-Vázquez D. I. Rodríguez-Jerez J. J. (2012). Use of epifluorescence microscopy to assess the effectiveness of phage P100 in controlling Listeria monocytogenes biofilms on stainless steel surfaces.Food Control23470–477. 10.1016/j.foodcont.2011.08.016
63
Møretrø T. Schirmer B. C. T. Heir E. Fagerlund A. Hjemli P. Langsrud S. (2017). Tolerance to quaternary ammonium compound disinfectants may enhance growth of Listeria monocytogenes in the food industry.Int. J. Food Microbiol.241215–224. 10.1016/j.ijfoodmicro.2016.10.025
64
Oliveira H. Azeredo J. Lavigne R. Kluskens L. D. (2012). Bacteriophage endolysins as a response to emerging foodborne pathogens.Trends Food Sci. Technol.28103–115. 10.1016/j.tifs.2012.06.016
65
Ortega Morente E. Fernandez-Fuentes M. A. Grande Burgos M. J. Abriouel H. Perez Pulido R. Galvez A. (2013). Biocide tolerance in bacteria.Int. J. Food Microbiol.16213–25. 10.1016/j.ijfoodmicro.2012.12.028
66
Ortiz S. Lopez-Alonso V. Rodriguez P. Martinez-Suarez J. V. (2015). The connection between persistent, disinfectant-resistant Listeria monocytogenes strains from two geographically separate Iberian pork processing plants: evidence from comparative genome analysis.Appl. Environ. Microbiol.82308–317. 10.1128/AEM.02824-15
67
O’Sullivan J. Mccullough J. Johnson J. H. Bonner D. P. Clark J. C. Dean L. et al (1990). Janthinocins A, B and C, novel peptide lactone antibiotics produced by Janthinobacterium lividum: I. taxonomy, fermentation, isolation, physico-chemical and biological characterization.J. Antibiot.43913–919. 10.7164/antibiotics.43.913
68
Overney A. Jacques-André-Coquin J. Ng P. Carpentier B. Guillier L. Firmesse O. (2017). Impact of environmental factors on the culturability and viability of Listeria monocytogenes under conditions encountered in food processing plants.Int. J. Food Microbiol.24474–81. 10.1016/j.ijfoodmicro.2016.12.012
69
Parasion S. Kwiatek M. Gryko R. Mizak L. Malm A. (2014). Bacteriophages as an alternative strategy for fighting biofilm development.Pol. J. Microbiol.63137–145.
70
Pérez Pulido R. Grande Burgos M. J. Gálvez A. Lucas López R. (2016). Application of bacteriophages in post-harvest control of human pathogenic and food spoiling bacteria.Crit. Rev. Biotechnol.36851–861. 10.3109/07388551.2015.1049935
71
Pérez-Ibarreche M. Castellano P. Leclercq A. Vignolo G. (2016). Control of Listeria monocytogenes biofilms on industrial surfaces by the bacteriocin-producing Lactobacillus sakei CRL1862.FEMS Microbiol. Lett.3631–6. 10.1093/femsle/fnw118
72
Perricone M. Arace E. Corbo M. R. Sinigaglia M. Bevilacqua A. (2015). Bioactivity of essential oils: a review on their interaction with food components.Front. Microbiol.6:76. 10.3389/fmicb.2015.00076
73
Reyes-Jurado F. Franco-Vega A. Ramírez-Corona N. Palou E. López-Malo A. (2014). Essential oils: antimicrobial activities, extraction methods, and their modeling.Food Eng. Rev.7275–297. 10.1007/s12393-014-9099-2
74
Sadekuzzaman M. Yang S. Mizan M. F. R. Kim H. S. Ha S. D. (2017). Effectiveness of a phage cocktail as a biocontrol agent against L. monocytogenes biofilms.Food Control78256–263. 10.1016/j.foodcont.2016.10.056
75
Salmond G. P. C. Fineran P. C. (2015). A century of the phage: past, present and future.Nat. Rev. Microbiol.13777–786. 10.1038/nrmicro3564
76
Sánchez L. A. Hedström M. Delgado M. A. Delgado O. D. (2010). Production, purification and characterization of serraticin A, a novel cold-active antimicrobial produced by Serratia proteamaculans 136.J. Appl. Microbiol.109936–945. 10.1111/j.1365-2672.2010.04720.x
77
Schmelcher M. Loessner M. J. (2016). Bacteriophage endolysins: applications for food safety.Curr. Opin. Biotechnol.3776–87. 10.1016/j.copbio.2015.10.005
78
Schmelcher M. Shabarova T. Eugster M. R. Eichenseher F. Tchang V. S. Banz M. et al (2010). Rapid multiplex detection and differentiation of Listeria cells by use of fluorescent phage endolysin cell wall binding domains.Appl. Environ. Microbiol.765745–5756. 10.1128/AEM.00801-10
79
Schmelcher M. Waldherr F. Loessner M. J. (2012). Listeria bacteriophage peptidoglycan hydrolases feature high thermoresistance and reveal increased activity after divalent metal cation substitution.Appl. Microbiol. Biotechnol.93633–643. 10.1007/s00253-011-3372-6
80
Schuch R. Khan B. K. Raz A. Rotolo J. A. Wittekind M. (2017). Bacteriophage lysin CF-301, a potent antistaphylococcal biofilm agent.Antimicrob. Agents Chemother.61:e02666-16. 10.1128/AAC.02666-16
81
Simmons M. Morales C. A. Oakley B. B. Seal B. S. (2012). Recombinant expression of a putative amidase cloned from the genome of Listeria monocytogenes that lyses the bacterium and its monolayer in conjunction with a protease.Probiot. Antimicrob. Proteins41–10. 10.1007/s12602-011-9084-5
82
Singh R. Ray P. Das A. Sharma M. (2009). Role of persisters and small-colony variants in antibiotic resistance of planktonic and biofilm-associated Staphylococcus aureus: an in vitro study.J. Med. Microbiol.581067–1073. 10.1099/jmm.0.009720-0
83
Soni K. A. Nannapaneni R. (2010). Removal of Listeria monocytogenes biofilms with bacteriophage P100.J. Food Prot.731519–1524. 10.4315/0362-028X-73.8.1519
84
Strauch E. Hammerl J. A. Hertwig S. (2007). Bacteriophages: new tools for safer food?J. Verbrauchersch. Lebensm.2138–143. 10.4067/S0716-10182015000700010
85
Tezel U. Pavlostathis S. G. (2015). Quaternary ammonium disinfectants: microbial adaptation, degradation and ecology.Curr. Opin. Biotechnol.33296–304. 10.1016/j.copbio.2015.03.018
86
Tompkin R. B. (2002). Control of Listeria monocytogenes in the food-processing environment.J. Food Prot.65709–725. 10.4315/0362-028X-65.4.709
87
Van Tassell M. L. Ibarra-Sanchez L. A. Hoepker G. P. Miller M. J. (2017). Hot topic: antilisterial activity by endolysin PlyP100 in fresh cheese.J. Dairy Sci.1002482–2487. 10.3168/jds.2016-11990
88
Véghová A. Minarovièová J. Koreňová J. Drahovská H. Kaclíková E. (2016). Prevalence and tracing of persistent Listeria monocytogenes strains in meat processing facility production chain.J. Food Saf.37:e12315. 10.1111/jfs.12315
89
Winkelströter L. K. Gomes B. C. Thomaz M. R. S. Souza V. M. De Martinis E. C. P. (2011). Lactobacillus sakei 1 and its bacteriocin influence adhesion of Listeria monocytogenes on stainless steel surface.Food Control221404–1407. 10.1016/j.foodcont.2011.02.021
90
Winkelströter L. K. Tulini F. L. De Martinis E. C. P. (2015). Identification of the bacteriocin produced by cheese isolate Lactobacillus paraplantarum FT259 and its potential influence on Listeria monocytogenes biofilm formation.LWT Food Sci. Technol.64586–592. 10.1016/j.lwt.2015.06.014
91
Woo J. Ahn J. (2013). Probiotic-mediated competition, exclusion and displacement in biofilm formation by food-borne pathogens.Lett. Appl. Microbiol.56307–313. 10.1111/lam.12051
92
Wood T. K. (2017). Strategies for combating persister cell and biofilm infections.Microb. Biotechnol.101054–1056. 10.1111/1751-7915.12774
93
Xia X. Li G. Luo D. Xia J. Zheng J. Kan J. (2017). Analysis of essential oil extracted by different methods from Osmanthus fragrans.J. Chin. Cereals Oils Assoc.3267–73.
94
Zhang H. Bao H. Billington C. Hudson J. A. Wang R. (2012). Isolation and lytic activity of the Listeria bacteriophage endolysin LysZ5 against Listeria monocytogenes in soya milk.Food Microbiol.31133–136. 10.1016/j.fm.2012.01.005
95
Zhao T. Doyle M. P. Zhao P. (2004). Control of Listeria monocytogenes in a biofilm by competitive-exclusion microorganisms.Appl. Environ. Microbiol.703996–4003. 10.1128/AEM.70.7.3996-4003.2004
96
Zhao T. Podtburg T. C. Zhao P. Chen D. Baker D. A. Cords B. et al (2013). Reduction by competitive bacteria of Listeria monocytogenes in biofilms and Listeria bacteria in floor drains in a ready-to-eat poultry processing plant.J. Food Prot.76601–607. 10.4315/0362-028X.JFP-12-323
97
Zhao T. Podtburg T. C. Zhao P. Schmidt B. E. Baker D. A. Cords B. et al (2006). Control of Listeria spp. by competitive-exclusion bacteria in floor drains of a poultry processing plant.Appl. Environ. Microbiol.723314–3320. 10.1128/AEM.72.5.3314-3320.2006
Summary
Keywords
Listeria monocytogenes , biofilms, biocontrol, bacteriophages, bacteriocins, endolysins, competitive exclusion, essential oils
Citation
Gray JA, Chandry PS, Kaur M, Kocharunchitt C, Bowman JP and Fox EM (2018) Novel Biocontrol Methods for Listeria monocytogenes Biofilms in Food Production Facilities. Front. Microbiol. 9:605. doi: 10.3389/fmicb.2018.00605
Received
22 December 2017
Accepted
15 March 2018
Published
03 April 2018
Volume
9 - 2018
Edited by
Giovanna Suzzi, Università di Teramo, Italy
Reviewed by
Laurent Guillier, Agence Nationale de Sécurité Sanitaire de l’Alimentation, de l’Environnement et du Travail (ANSES), France; Giorgia Perpetuini, Università di Teramo, Italy; Arun K. Bhunia, Purdue University, United States
Updates
Copyright
© 2018 Gray, Chandry, Kaur, Kocharunchitt, Bowman and Fox.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Edward M. Fox, edward.fox@csiro.au
This article was submitted to Food Microbiology, a section of the journal Frontiers in Microbiology
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.