Abstract
Endophytic actinobacteria are one of the important pharmaceutical resources and well known for producing different types of bioactive substances. Nevertheless, detection of the novelty, diversity, and bioactivity on endophytic actinobacteria isolated from mangrove plants are scarce. In this study, five different mangrove plants, Avicennia marina, Aegiceras corniculatum, Kandelia obovota, Bruguiera gymnorrhiza, and Thespesia populnea, were collected from Beilun Estuary National Nature Reserve in Guangxi Zhuang Autonomous Region, China. A total of 101 endophytic actinobacteria strains were recovered by culture-based approaches. They distributed in 7 orders, 15 families, and 28 genera including Streptomyces, Curtobacterium, Mycobacterium, Micrococcus, Brevibacterium, Kocuria, Nocardioides, Kineococcus, Kytococcus, Marmoricola, Microbacterium, Micromonospora, Actinoplanes, Agrococcus, Amnibacterium, Brachybacterium, Citricoccus, Dermacoccus, Glutamicibacter, Gordonia, Isoptericola, Janibacter, Leucobacter, Nocardia, Nocardiopsis, Pseudokineococcus, Sanguibacter, and Verrucosispora. Among them, seven strains were potentially new species of genera Nocardioides, Streptomyces, Amnibacterium, Marmoricola, and Mycobacterium. Above all, strain 8BXZ-J1 has already been characterized as a new species of the genus Marmoricola. A total of 63 out of 101 strains were chosen to screen antibacterial activities by paper-disk diffusion method and inhibitors of ribosome and DNA biosynthesis by means of a double fluorescent protein reporter. A total of 31 strains exhibited positive results in at least one antibacterial assay. Notably, strain 8BXZ-J1 and three other potential novel species, 7BMP-1, 5BQP-J3, and 1BXZ-J1, all showed antibacterial bioactivity. In addition, 21 strains showed inhibitory activities against at least one “ESKAPE” resistant pathogens. We also found that Streptomyces strains 2BBP-J2 and 1BBP-1 produce bioactive compound with inhibitory activity on protein biosynthesis as result of translation stalling. Meanwhile, Streptomyces strain 3BQP-1 produces bioactive compound inducing SOS-response due to DNA damage. In conclusion, this study proved mangrove plants harbored a high diversity of cultivable endophytic actinobacteria, which can be a promising source for discovery of novel species and bioactive compounds.
Introduction
The increased prevalence of “ESKAPE” pathogens, along with the rapid development of multidrug resistances became the driving force in new antibiotics discovery (Spellberg et al., 2008; Bassetti et al., 2013; Pendleton et al., 2013; Singh et al., 2017). New types of antibacterial drugs are so extremely limited that clinicians are forced to the situation as “Bad Bugs, No Drugs,” which made novel antibiotic discovery become a very important and urgent issue (Talbot et al., 2006; Boucher et al., 2009; Khan and Khan, 2016). It is well known that actinobacteria, especially the genus Streptomyces, are major producers of bioactive compounds, it account for nearly 45% of the total bioactive metabolites produced by microorganisms (Bérdy, 2012; Genilloud, 2017). However, after excavation for many decades, the discovery of new species and new antibiotics from common enviroments is becoming increasingly difficult. On the contrary, it is also becoming increasingly evident that un- and under-explored habitats are rich and new sources of actinobacteria for interesting novel bioactive metabolites, including antibiotics (Rateb et al., 2011; Manivasagana et al., 2014; Jiang et al., 2015; Mohammadipanah and Wink, 2015; Axenov-Gibanov et al., 2016; Hassan and Shaikh, 2017). Microbes have to adapt and evolve in metabolite and genetic level to resist the stress from their habitats, thus, have the capability to synthesis of novel chemicals to carry out special biofunctions and bioactivities (Wilson and Brimble, 2009). In fact, a large number of new bioactive compounds produced by actinobacterial strains from special enviroments have been discovered in recent years. (Wilson and Brimble, 2009; Rateb et al., 2011; Xu et al., 2014; Hassan and Shaikh, 2017).
Mangroves locate in the intertidal zone of tropical and subtropical coastlines and possess a unique environment with highly productive ecosystems, it harbors many kinds of microorganisms including actinobacteria (Jensen et al., 1991; Hong et al., 2009; Xu et al., 2014; Azman et al., 2015; Sun et al., 2017). Since 2007, 66 new species and 8 novel genera of actinobacteria have been isolated and identified from mangrove environments (Biswas et al., 2017; Hamada et al., 2017; Jiang et al., 2017; Law et al., 2017; Li F. et al., 2017; Liu et al., 2017; Sun et al., 2017; Qu et al., 2018). Furthermore, at least 84 new compounds including some “hot molecules,” such as salinosporamides, xiamycins and novel indolocarbazoles, were discovered from mangrove actinobacteria. (Kyeremeh et al., 2014; Xu et al., 2014; Ding et al., 2015; Fu et al., 2016; Han et al., 2016; Mangamuri et al., 2016a,b; Chen et al., 2017; Ye et al., 2017).
Endophytic actinobacteria have become a hot spot with increasing actinobacteria prospecting from a range of plant types (Coombs and Franco, 2003; Tian et al., 2007; Qin et al., 2009; Gohain et al., 2015; Passari et al., 2015; Trujillo et al., 2015; Nalini and Prakash, 2017). Moreover, novel endophytic actinobacteria from various tissues of plants have been increasingly reported, and some produced bioactive metabolites with new chemical structures (Qin et al., 2009; Golinska et al., 2015; Matsumoto and Takahashi, 2017; Sun et al., 2017). Until the present, some studies have implemented to isolate endophytic actinobacteria and their secondary metabolites from mangrove plants (Lin et al., 2005; Hong et al., 2009; Wang et al., 2010; Xie et al., 2011; Ding et al., 2012, 2015; Li et al., 2013, 2016; Xu et al., 2014, 2016; Li F. et al., 2017; Li F.N. et al., 2017). According to Xu et al. (2014), 29 of 34 compounds isolated from mangrove endophytic actinobacteria had novel structures, suggesting that mangrove endophytic actinobacteria have the ability to produce new bioactive metabolites. Even though, more than 10 new endophytic actinobacteria species have been characterized from mangrove plants (Jiang et al., 2017; Liu et al., 2017; Sun et al., 2017), studies on mangrove plants endophytic actinobacteria are still rather scarce when compared to actinobacteria prospecting for mangrove soil or sediment. Our results presented in this article revealed that actinobacteria isolated from mangrove plants are important sources for new species and diverse bioactive compounds, and researches on this area should draw much more attention (Yan et al., 2010; Liu et al., 2016, 2017; Tuo et al., 2016a,b; Jiang et al., 2017).
Beilun River is the boundary river between China and Vietnam and the Chinese part is located in Guangxi Zhuang Autonomous Region. The Beilun Estuary National Nature Reserve was estabished to protect mangrove plants in the year of 2000. The reserve contains rich mangrove flora composed of 14 plant species and is mostly unexplored (Liang et al., 2004). To our knowledge, few studies have focused on the endophytic actinobacterial communities of Beilun Estuary mangrove plants. In this study, the diversity and novelty of cultivable actinobacteria from mangrove plants of the Beilun Estuary National Nature Reserve was investigated and their ability to produce antimicrobial activity against “ESKAPE” was evaluated. Meanwhile, a high-throughput screening model based on double fluorescent protein reporter was also implemented to find strains producing secondary metabolites as ribosome and DNA biosynthesis inhibitors.
Materials and Methods
Sampling of Mangrove Plants
Mangrove plants were collected from Beilun Estuary National Nature Reserve (21°36’N, 108°12’E) in Guangxi Zhuang Autonomous Region, China, in July, 2015. A total of 19 tissues including leaves, branches, barks, roots, also flowers, and fruits (if present) were collected from five plants: Avicennia marina, Aegiceras corniculatum, Kandelia obovota, Bruguiera gymnorrhiza, and Thespesia populnea. After flame sterilization, the cut ends of root and branch were sealed with parafilm, and then all samples were transported to the laboratory and processed within 48 h.
Isolation of Endophytic Actinobacteria
All plant samples were washed thoroughly with tap water for a few minutes to remove organic debris and soil. After air-drying, the samples were processed according to the five-step sterilization method described by Qin et al. (2009), and ground into powder by using micromill, and distributed on plates containing different isolation media. The plates were incubated at 28°C for 2–5 weeks. Colonies that displayed differentiable morphologies were transferred onto ISP2 agar plates and repeatedly isolated and incubated until pure isolates were obtained. The purified cultures were maintained on ISP2 medium slants at 4°C and stored in 20% (v/v) glycerol suspensions at -80°C. A total of 10 media were used for the isolation of mangrove endophytic actinobacteria (Supplementary Table S1), and actidione (40 mg L-1), nystatin (40 mg L-1), and nalidixic acid (25 mg L-1) were added to the media to inhibit the growth of fungi and Gram-negative bacteria.
The efficacy of the sterilization process was confirmed using methods described by Qin et al. (2009). Briefly, the surface-sterilized tissues were washed three times in sterile distilled water, soaked in sterile water for 1 min with continuous stirring, and then, a 0.1 milliliter aliquot of the last washed water was inoculated onto ISP2 agar plates and incubated at 28°C. Meanwhile, the surface-sterilized tissues were imprinted onto ISP2 agar plates and incubated at 28°C. If no microbial growth was observed on the surface of the media, the sterilization was considered as effective.
Molecular Identification and Phylogenetic Analysis of Isolates
Genomic DNA was extracted from pure isolates as described by Zhou et al. (2010). Universal primers 27F and 1492R (Lane, 1991) were used for amplification of 16S rRNA gene fragments. Cycling conditions were as follows: initial denaturation at 95°C for 5 min, 35 cycles of 94°C for 1 min, 55°C for 1 min, and 72°C for 2 min, and a final extension of 10 min at 72°C. The PCR products were purified and sequenced on the ABI PRISM 3730XL DNA Analyzer from Life Sciences Solutions Group, Thermo Fisher Scientific (Beijing). The genus-level affiliation of the sequences was validated using available 16S rRNA gene sequences from the EzTaxon-e server1 (Kim et al., 2012). Sequence alignment and phylogenetic analysis were carried out using MEGA version 5 (Tamura et al., 2011). Phylogenetic trees were constructed by using the neighbor-joining method (Saitou and Nei, 1987) with bootstrap values based on 1000 replications (Felsenstein, 1985) using MEGA5 program. The 16S rRNA gene sequences of the potential novel isolates were deposited in GenBank under the accession numbers: MG563365–563372, and the partial 16S rRNA gene sequences of the remaining isolates were deposited in GenBank with the following accession numbers: MG563311–563364.
Antibacterial Activity Screening
Based on phylogenetic and phenotypic characteristics analysis, 63 strains were selected for antimicrobial assay. Each strain was transferred to 500 mL Erlenmeyer flasks containing 100 mL of YIM 38 medium (Jiang et al., 2015) and cultivated for 7 days at 28°C with 180 rpm orbital shaking. The 600 mL fermentation broth obtained from each of the isolates was separated from the mycelium by centrifugation at 4500 rpm at 20°C for 20 min. The supernatants were extracted twice with ethyl acetate (1:1, v/v), and the whole organic layer and 50 mL of water layer were concentrated under vacuum and freeze-dried, respectively, to obtain dried samples. The mycelium were soaked in acetone for 12 h and then filtered, the filtration were concentrated under vacuum to obtain dried samples. Finally, each of three kinds of dried samples was dissolved in 3 mL HPLC grade methanol and used in the antibacterial assay by agar disk diffusion method. The methanol sample (60 μL) was dripped on paper disk (diameter, 5 mm). Meanwhile, 60 μL methanol without sample was used as the negative control, and levofloxacin (10 μL, 0.1 mg/mL) was used as the positive control. After being dried in a hood, the paper disks were transferred to agar plates containing pathogenic bacteria and incubated at 37°C for 24 h. The diameters of the inhibition zones were measured by vernier caliper. The indicator bacteria used for antimicrobial assay were: Enterococcus faecalis (E. faecalis) (ATCC 29212, ATCC 51299), Staphylococcus aureus (S. aureus) (ATCC 25923, 2641), Klebsiella pneumoniae (K. pneumoniae) (ATCC 10031, ATCC 700603), Acinetobacter baumannii (A. baumannii) (ATCC19606, 2799), Pseudomonas aeruginosa (P. aeruginosa) (ATCC 27853, 2774), and Escherichia coli (E. coli) (ATCC 25922, 2800). Isolate 2774 is resistant to carbapenem and quinolone, isolates ATCC 51299, 2641, and 2799 are resistant to vancomycin, methicillin, and carbapenem, and strains 2800 and ATCC 700603 are both ESLB-producing species, respectively.
Mechanism of Action Determination
Reporter strain JW5503-pDualrep2 was used as previously described (Osterman et al., 2016). Briefly, 1 ml of the extracts from ethyl acetate layer was dried in hood and 100 μl DMSO was added to each sample, and a total of 63 samples were prepared. A total of 2 μl of each sample were applied to agar plate containing a lawn of the reporter strain. After overnight incubation at 37°C, the plate was scanned by ChemiDoc (Bio-Rad): “Cy3-blot” for RFP and “Cy5-blot” for Katushka2S. Erythromycin and levofloxacin were used as positive controls for ribosome and DNA biosynthesis inhibitors, respectively.
Results
Isolation and Diversity of Actinobacteria From Mangrove Plants of Beilun Estuary
A total of 318 strains of endophytic microbes were isolated from 19 tissues collected from 5 mangrove plants. A total of 158 presumed endophytic actinobacteria were selected on the basis of colonial morphology and were further identified by their 16S rRNA gene sequences. A total of 101 isolates were confirmed as actinobacteria and phylogenetic analysis based on the partial 16S rRNA genes sequences (approximately 700 bp) revealed that the 101 endophytic actinobacterial strains were assigned to 28 genera in 7 orders of 15 families: Streptomyces, Curtobacterium, Mycobacterium, Micrococcus, Brevibacterium, Kocuria, Nocardioides, Kineococcus, Kytococcus, Marmoricola, Microbacterium, Micromonospora, Actinoplanes, Agrococcus, Amnibacterium, Brachybacterium, Citricoccus, Dermacoccus, Glutamicibacter, Gordonia, Isoptericola, Janibacter, Leucobacter, Nocardia, Nocardiopsis, Pseudokineococcus, Sanguibacter, and Verrucosispora (Figure 1). Relative abundance of endophytic actinobacteria at the genus level revealed that Streptomyces and Curtobacterium were most abundant with 33.0 and 14.0%, respectively (Figure 2A). All endophytic actinobacterial strains were isolated from 10 isolation media, which had a major influence on the number of isolates recovered. The ISP2-M medium was the most effective as regards the number and diversity of isolates obtained; the TWYE medium produced the second-highest numbers and diversities. On the contrary, the TP medium yielded the lowest numbers (Figure 2B). All tissues produced at least one isolate, which indicated that endophytic actinobacterial isolates can colonize different tissues throughout the plants (Figure 2C). Out of 101 isolates, the majority (n = 35, 34.65%) were isolated from barks, followed by stem (n = 24, 23.76%), leaf (n = 22, 21.78%), fruits (n = 10, 9.90%), roots (n = 9, 8.91%), and flower (n = 1, 0.99%).
FIGURE 1
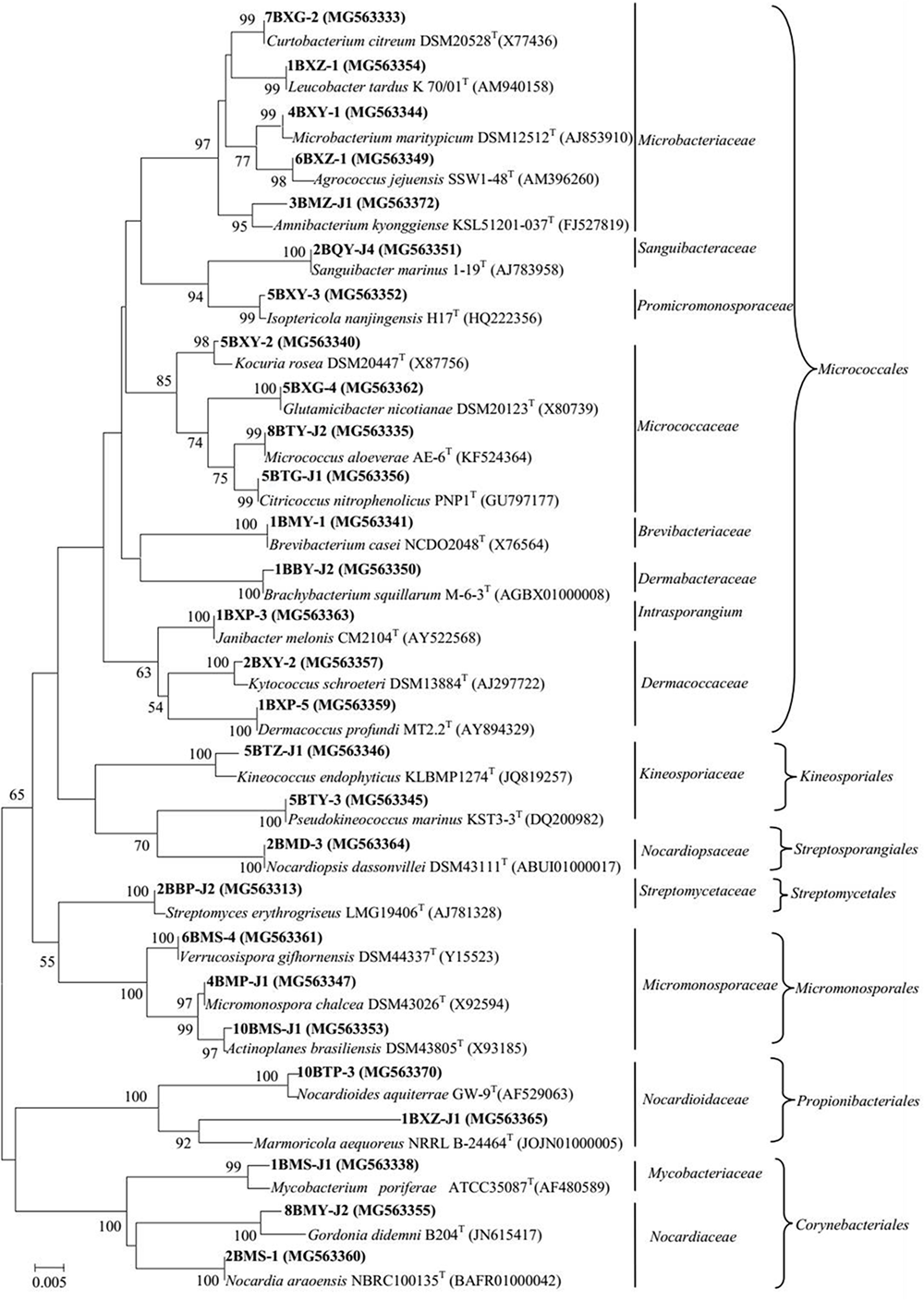
Phylogenetic tree of actinobacterial isolates from mangrove plants of Beilun Estuary that belong to the orders Micrococcales, Kineosporiales, Streptosporangiales, Streptomycetales, Micromonosporales, Propionibacteriales, Corynebacteriales, and closely related representative species. Neighbor-joining phylogenetic tree based on 16S rRNA gene sequences of the strains and closely related species of the genera. Numbers at nodes indicate the level of bootstrap support (> 50%) based on 1000 replications. Bar, 5 nt substitutions per 1000 nt.
FIGURE 2
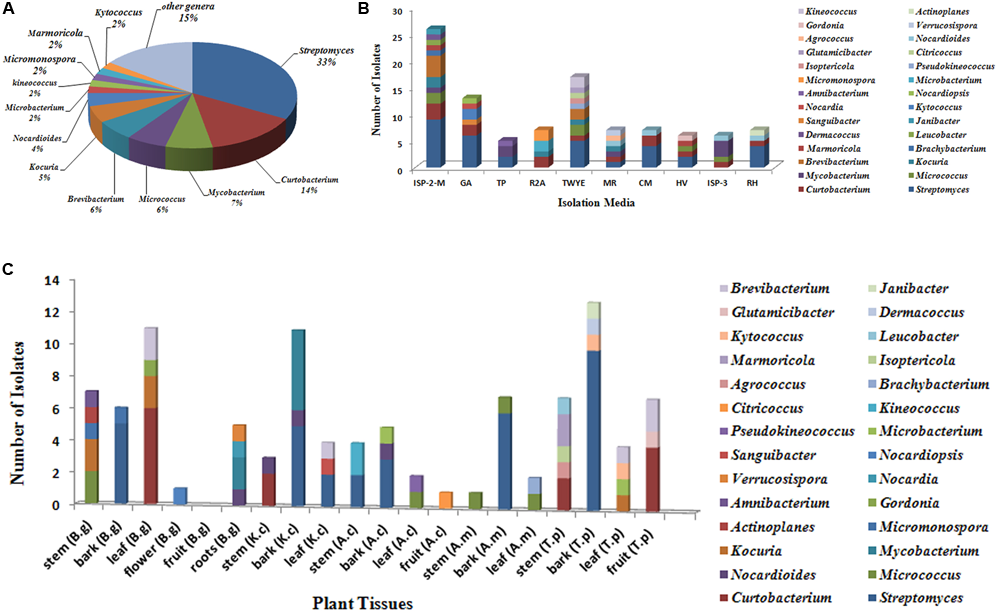
Diversity of culturable actinobacteria from mangrove plants of Beilun Estuary. (A) Pie chart representation of the percentage frequency of actinobacterial genera within the total number of isolates. (B) Number of actinobacterial isolates recovered from the different culture media used. (C) Number of actinobacterial isolates from different tissues of mangrove plants (B.g: Bruguiera gymnorrhiza; K.c: Kandelia candel; A.c: Aegiceras corniculatum; A.m: Avicennia marina; T.p: Thespesia populnea).
Phylogenetic Novelty of Isolated Actinobacteria
A total of 7 strains exhibited low sequence similarities (< 98.65%) with validly described species based on a 16S rRNA gene sequence search via the EzTaxon server, suggesting that these strains could represent novel taxons within the phylum Actinobacteria (Kim et al., 2014). These putative novel isolates belong to genera Marmoricola and Nocardioides in family Nocardioidaceae, genus Streptomyces in family Streptomycetaceae, genus Mycobacterium in family Mycobacteriaceae, and genus Amnibacterium in family Microbacteriaceae. In addition, strain 8BXZ-J1 has been characterized as a new species of the genus Marmoricola (Jiang et al., 2017). The almost complete sequencing (> 1375bp) of the 16S rRNA gene was performed in 7 potential novel strains and the new species. The phylogenetic tree based on 16S rRNA gene sequences generated by using the neighbor-joining method indicated that strain 1BXZ-J1 was clustered within the genus Marmoricola, strains 10BTP-3 and 6BMS-J1 were clustered within the genus Nocardioides, strains 5BQP-J3 and 7BMP-1 were clustered within the genus Streptomyces, strain 3BMZ-J1 was clustered within the genus Amnibacterium, and strain 3BMS-J1 was clustered within the genus Mycobacterium (Figure 3). The 16S rRNA gene sequences of strain 1BXZ-J1, 10BTP-3, 6BMS-J1, 5BQP-J3, 7BMP-1, 3BMZ-J1, and 3BMS-J1 showed highest similarities with Marmoricola aequoreus NRRLB-24464T (96.3%), Nocardioides soli mbc-2T (97.0%), Nocardioides aquiterrae GW-9T (97.3%), Streptomyces yogyakartensis NBRC 100779T (97.9%), Streptomyces phaeoluteichromatogenes NRRL 5799T (98.2%), Amnibacterium kyonggiense KSL51201-037T (98.1%), and Mycobacterium peregrinum ATCC 14467T (98.6%), respectively. These isolates will be further characterized in a polyphasic approach to determine their taxonomic positions. The phylogenetic analysis presented implied a considerable cultivable actinobacteria with novelty in mangrove plants from Beilun Estuary.
FIGURE 3
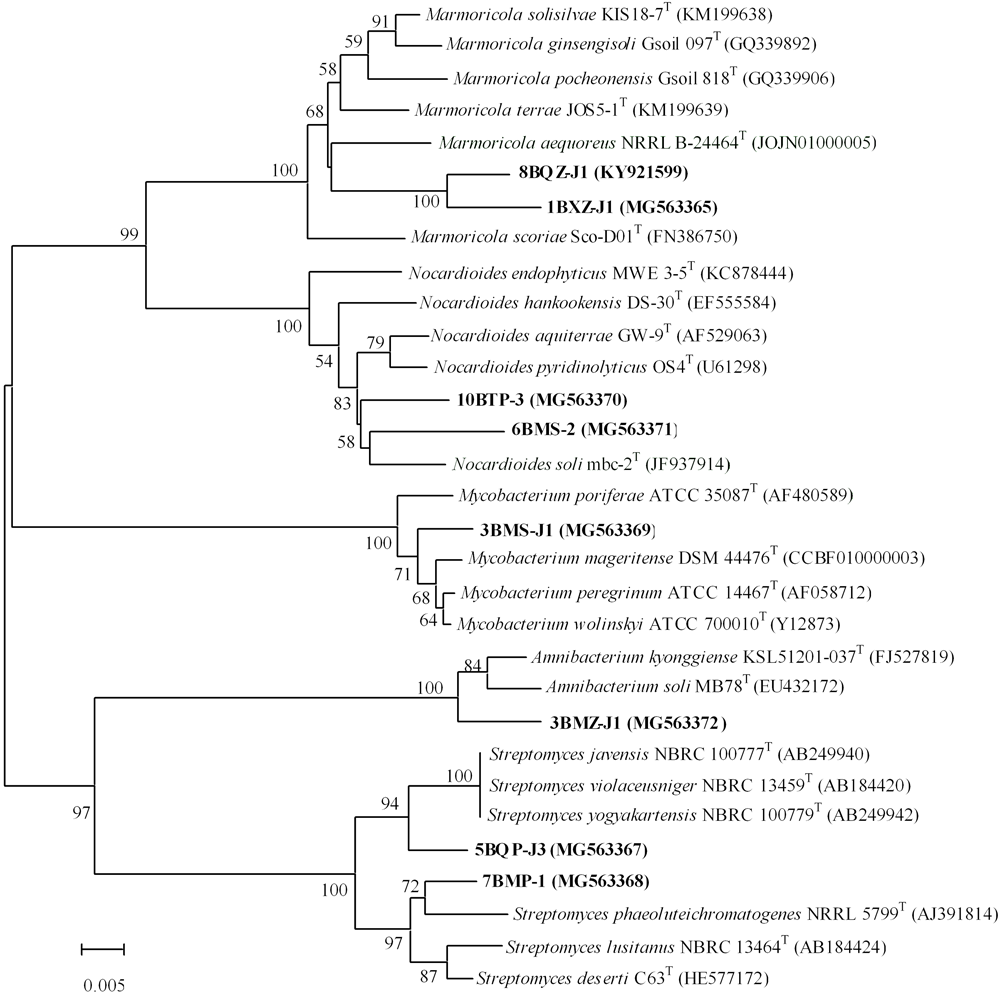
Phylogenetic tree of 7 potential novel strains and 1 new species from mangrove plants of Beilun Estuary and closely related representative species. Neighbor-joining phylogenetic tree based on 16S rRNA gene sequences of the strains and related species of the genera. Numbers at nodes indicate the level of bootstrap support (> 50%) based on 1000 replications. Bar, 5 nt substitutions per 1000 nt.
Antibacterial Activity of Actinobaterial Isolates
Antimicrobial activity was evaluated against a set of pathogenic bacteria. Out of 63 isolates, 31 (49.2%) exhibited antagonistic activity against at least one of the tested pathogens (Supplementary Table S2). They were affiliated to 15 different genera, i.e., Streptomyces (14), Curtobacterium (2), Micromonospora (2), Marmoricola (2), Micrococcus (1), Nocardioides (1), Mycobacterium (1), Kocuria (1), Microbacterium (1), Leucobacter (1), Gordonia (1), Citricoccus (1), Kytococcus (1), Nocardia (1), and Nocardiopsis (1). The antimicrobial profile of the actinobacteria against different pathogenic bacteria was shown in Figure 4. Regarding the sensitive pathogenic strains tested, activity against P. aeruginosa was clearly the most frequent (21 isolates, 33.3%), and activity against E. coli was the least frequent (7, 11.1%), while 27.0% (17), 19.0% (12), 14.3 (9), and 12.7% (8) of the isolates were active against S. aureus, E. faecalis, K. pneumonia, and A. baumannii, respectively. Concerning the resistant pathogenic strains tested, activity against S. aureus was the most frequent (12 isolates, 19.0%), and activity against K. pneumonia was the least frequent (2, 3.2%), while 12.7% (8), 9.5% (6), 7.9% (5), and 4.8% (3) of the isolates were active against P. aeruginosa, E. faecalis, A. baumannii, and E. coli, respectively. In all, 21 strains showed inhibitory activities against at least one “ESKAPE” resistant pathogens, suggesting that these strains might represent a valuable source of biologically active compounds with inhibitory activities against “ESKAPE” resistant pathogens.
FIGURE 4
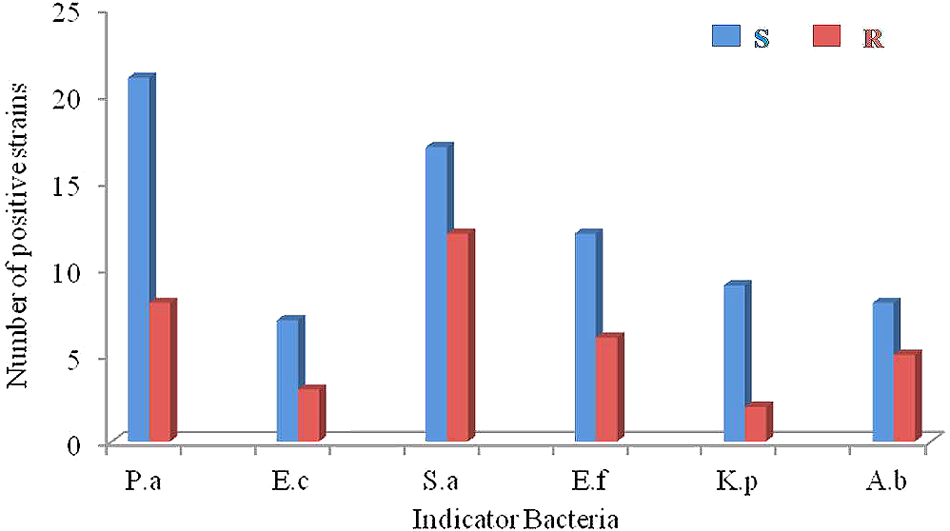
The antimicrobial profiles of the actinobacteria against ESKAPE (P.a: Pseudomonas aeruginosa; E.c: Escherichia coli; S.a: Staphyloccocus aureus; E.f: Enterococcus faecalis; K.p: Klebsiella peneumoniae; A.b: Acinetobacter baumannii). S: sensitive pathogenic strains; R: drug-resistant pathogenic strains.
Notably, three of the seven potential novel strains and new species (8BXZ-J1) showed activity against at least one of the tested pathogenic bacteria. Strain 7BMP-1 exhibited strong antimicrobial activities against S. aureus ATCC 25923 (19.1 mm), S. aureus 2641 (16.1 mm), E. faecalis ATCC 29212 (16.0 mm), P. aeruginosa ATCC 27853 (14.0 mm), E. faecalis ATCC 33186 (13.8 mm), and A. baumannii ATCC 19606 (9.7 mm). Strain 5BQP-J3 showed strong inhibitory activity against P. aeruginosa ATCC 27853 (19.1 mm), which is resistant to carbapenem and quinolone. Both strains 1BXZ-J1 and 8BXZ-J1 showed strong inhibitory activity against P. aeruginosa ATCC 27853, and the inhibition zones were 19.4 and 17.1 mm, respectively.
The 63 ethyl acetate extracts were assayed by a double fluorescent protein reporter (pDualrep2 reporter system) screening model and the results are shown in Figure 5. Strains 2BBP-J2 and 1BBP-1 induced Katushka2S expression, which demonstrated strong translation inhibition activity as erythromycin did, meanwhile, strain 3BQP-1-induced RFP expression and SOS-response because of DNA-damage as levofloxacin did.
FIGURE 5

Induction of a two-color dual reporter system sensitive to inhibitors of the ribosome progression or inhibitors of DNA replication, respectively. Spots of erythromycin (ERY), levofloxacin (LEV), and tested compounds were placed on the surface of an agar plate containing E. coli tolC cells transformed with the pDualrep2 reporter plasmid. Shown is the fluorescence of the lawn of E. coli cells scanned at 553/574 nm (green pseudocolor) for RFP fluorescence and 588/633 nm (red pseudocolor) for Katushka2S fluorescence. Induction of expression of Katushka2S is triggered by translation inhibitors, while RFP is upregulated by induction of DNA damage SOS response.
Discussion
Mangroves plants harbor a great diversity of culturable actinobacteria, and are proven to be a valuable microorganism source for discovery of new bioactive metabolites (Hong et al., 2009; Xu et al., 2014, 2016; Li et al., 2016; Li F.N. et al., 2017). In this study, a considerable diversity of endophytic actinobacteria was obtained from the mangrove plants collected in Beilun Estuary National Nature Reserve of Guangxi Zhuang Autonomous Region, China. In addition, to our knowledge, this is the first time strains that were rarely recovered in genera Sanguibacter and Citricoccus have been isolated and cultured from the inner parts of plants. Recently, Li F.N. et al. (2017) reported that 198 endophytic actinobacterial strains isolated from mangrove plants of Macao, China, were distributed in 30 genera affiliated to 8 orders. Li et al. (2016) isolated 146 endophytic actinobacteria belonging to 8 orders (18 genera) from mangrove plants of Dongzhaigang of Hainan Province, China. The 159 isolates of 19 actinobacteria genera belonging to 8 orders obtained from mangrove plants of Zhanjiang in Guangdong Province were reported in 2016 (Xu et al., 2016). Hong et al. (2009) identified 237 actinobacterial isolates belonging to five orders (13 different genera) from soil and plant samples of 8 mangrove sites in China. In our study, we identified 28 genera from 101 isolated actinobacterial strains, the diversity of endophytic actinobacteria is apparently higher than the previous studies. Therefore, it is clear that endophytic actinobacteria in mangrove plants are diverse, and that the variation in the diversity and richness of endophytic actinobacteria recovered from mangrove plants are closely related with the isolation media and different tissues.
Some novel endophytic actinobacteria have been discovered from mangrove plants, suggesting that they have potential as excellent sources of novel species with actinobacterial reagents (Liu et al., 2017; Sun et al., 2017). In this study, isolates from the genus Streptomyces were the most abundant, it is consistent with previous studies on actinobacterial communities in mangrove plants (Hong et al., 2009; Xu et al., 2014, 2016; Li et al., 2016; Li F.N. et al., 2017). Two Streptomyces strains (5BQP-J3 and 7BMP-1) are probably new species due to their relatively low gene sequence similarities to their closest type strains. Meanwhile, the gene sequence similarity between 5BQP-J3 and 7BMP-1 was 96.5%. In addition, five putative new rare actinobacteria species are proposed: Nocardioides sp. 10BTP-3, Nocardioides sp. 6BMS-2, Marmoricola sp. 1BXZ-J1, Mycobacterium sp. 3BMS-J1, and Amnibacterium sp. 3BMZ-J1, based on numerical thresholds related to 16S rRNA gene sequences (Kim et al., 2014; Supplementary Table S2). Further phylogenetic analysis of the isolates is shown in Figure 3. Pairwise comparison of the 16S rRNA gene sequences from the two Nocardioides strains (10BTP-3 and 6BMS-2) showed relatively low similarities (97.4 and 97.0%) to the type strains of Nocardioides aquiterrae and Nocardioides soli, respectively. The sequence similarity between 10BTP-3 and 6BMS-2 was 97.5%. Furthermore, the phylogenetic analysis suggested that the two isolates were diversely distributed within genus Nocardioides and clustered singly in the phylogenetic tree (Figure 3), indicating that isolates 10BTP-3 and 6BMS-2 might belong to new species. The Marmoricola strains 1BXZ-J1 and 8BXZ-J1 had lineages that were distinct from each other and from other members of the genus; and they also formed distinct subclades in the tree supported by high bootstrap values (Figure 3). The 16S rRNA gene sequences of 1BXZ-J1 and 8BXZ-J1 showed 96.3 and 96.9% identities to the nearest neighbors, Marmoricola aequoreus and Marmoricola solisilvae, respectively. The sequence similarity between 1BXZ-J1 and 8BXZ-J1 was 97.8%. Strain 8BXZ-J1 has been characterized as the first endophytic actinobacteria new species belonged to the genus Marmoricola (Jiang et al., 2017). Mycobacterium strains have been isolated from mangrove plants (Xu et al., 2016; Li F.N. et al., 2017) and other medicinal plants (Qin et al., 2009). However, novel species of the genus Mycobacterium from mangrove ecosystem have not yet been reported. 3BMS-J1 is probably a new Mycobacterium species because it presents relatively low 16S rRNA gene sequences similarity and forms a distinct phylogenetic branch when compared to strains from the genus Mycobacterium (Figure 3). Comparison of the 16S rRNA gene sequences of the isolate and its closest neighbors suggested that 3BMZ-J1 was closely related to the type strains of the genera Amnibacterium. To date, this genus comprises only two species that were isolated from Anyang stream and grass soil (Kim and Lee, 2011; Jin et al., 2013). Strain 8BXZ-J1 was isolated with HV medium, the potential novel strains 3BMS-J1 and 3BMZ-J1 were both isolated with TP medium, and the others potential novel strains 1BXZ-J1, 5BQP-J3, 6BMS-2, 7BMP-1, and 10BTP-3 were isolated with ISP-2-M medium, TWYE medium, MR medium, CM medium, and RH medium, respectively. This result demonstrated that it is still worthwhile to use traditional cultivating methods for isolating new actinobacterial species.
Analysis of composition in genera level of 31 positive strains from 63 tested strains indicated that Streptomyces strains predominated. In 63 tested strains, there are 15 Streptomyces strains, among them, 14 Streptomyces strains showed positive result. It is quite reasonable, since Streptomyces is the largest antibiotic-producing genus in the microbial world discovered, and it is well documented that mangrove Streptomyces are able to produce bioactive metabolites with a wide-range of activities, including antibacterial, antifungal, anti-HIV, and anticancer (Ding et al., 2010; Yuan et al., 2013; Khieu et al., 2015; Tan et al., 2015; Xu et al., 2015; Ser et al., 2017; Wang et al., 2017). Curtobacterium as the second most dominant genus in 63 tested strains, showed very low positive rate, only 2 of 11 displayed antibacterial activities against tested pathogens. Curtobacterium traditionally is viewed as a plant pathogen, and most studies focused on its role as an economically important plant pathogen (Huang et al., 2009; Buonaurio et al., 2015; Osdaghi et al., 2015). Recently, Undabarrena et al. (2016) reported Curtobacterium isolates showing potent antimicrobial bioactivity against more than one tested pathogens, and their antimicrobial activity was dependent on the culturing media.
It is noteworthy to highlight that the two rare actinobacterial strains, Kytococcus strain 2BXY-2 and Leucobacter strain 1BXZ-1, showed the strong inhibitory activity against Pseudomonas aeruginosa as shown in Supplementary Table S2. To our knowledge, antibacterial activities from these genera have been received scarce attention for potential development of novel antimicrobial reagents.
It is extensively accepted that novel microorganisms are a good source for the discovery of new secondary bioactive metabolites, as exemplified by salinosporamide A, a potent cytotoxic activity compound produced by Salinispora strain CNB-392 (Feling et al., 2003) and teixobactin, a new cell wall inhibitor with exceptional activity against Clostridium difficile and Bacillus anthracis produced by Eleftheria terrae (Ling et al., 2015). In the study, two potential new Streptomyces spieces (5BQP-J3 and 7BMP-1) displayed very strong antibacterial activities (Supplementary Table S2), thus it would be interesting to see if new antibiotics can be identified from these strains. In addition, antibacterial activities of Marmoricola isolates (1BXZ-J1 and 8BXZ-J1) against P. aeruginosa and S. aureus are highlighted by their novelty. Few have reported the antimicrobial activity potential of Marmoricola so far. Marmoricola isolates (1BXZ-J1 and 8BXZ-J1) may also be good candidates for production of bioactive compounds. Further experiments will be carried out to deepen our knowledge on the antibacterial activities of these novel isolates.
In the antibacterial assay, pDualrep2 reporter system was implemented to indicate existence of antibiotics in the culture broth, the system can distinguish simultaneously between antibiotics that induce the SOS response, a major general stress response caused by inhibitors of DNA biosynthesis and those that cause ribosome stalling. The existence of ribosome inhibitor such as erythromycin will lead Katushka2S expression, and the existence of inhibitor of DNA biosynthesis such as levofloxacin will lead RFP expression. Both 2BBP-J2 and 1BBP-1 fermentation broth demonstrated ribosome inhibition activities in their ethyl acetate extractions, indicating that Katushka2S expression had been induced. Meanwhile, strain 3BQP-1 demonstrated inhibition activity against DNA biosynthesis in its fermentation broth extract as shown by the induced RFP expression.
The diversity, novelty, and antimicrobial activity of endophytic actinobacteria from mangrove plants of Beilun Estuary were first comprehensively investigated in this study. The 101 selected isolates were assigned to 7 orders, 15 families, and 28 genera. Notably, 31 isolates belonging to 15 different genera show inhibitory activities against at least one “ESKAPE” pathogens, and 7 isolates affiliated with 5 genera are below the 98.65% sequence identity threshold and therefore may be potential candidates of new taxons. In addition, a new species of the genus Marmoricola isolated from the mangrove plants has been published. These results indicated that mangrove plants are potentially unique sources of novel actinobacteria with promising potential to produce highly bioactive metabolites.
Statements
Author contributions
Z-kJ carried out the sampling, the selective isolation, the primary identification of the isolates, the screening of antimicrobial activities, analyzed the data, and prepared the manuscript. LT and D-lH carried out the sampling, the selective isolation, and edited the manuscript. S-wL and F-nL prepared the samples for screening. OD, AT, DL, PS, IO, and VK sorted out the antibiotics’ mechanisms of action of the extracts by means of a double fluorescent protein reporter for high-throughput screening of ribosome and DNA biosynthesis inhibitors and edited the manuscript. C-hS conceived and designed the experiments, performed the sampling, and edited the manuscript.
Funding
The research work was supported by joint grant from NSFC and RFBR (National Natural Science Foundation of China, Grant No. 81611530716; and Russian Foundation for Basic Research, Grant No. 17-53-53130). C-hS was supported by CAMS Innovation Fund for Medical Sciences, Grant Nos. CAMS 2017-12M-B&R-08 and 2017-I2M-1-012 and NSFC, Grant Nos. 81621064 and 81373308. LT, D-lH, and S-wL was supported by NSFC, Grant Nos. 81603079, 81460537, and 81402834, respectively. IO was supported by the Russian Science Foundation, Grant No. 14-24-00061. F-nL was supported by PUMC Fund of the Fundamental Research Funds for the Central Universities, Grant No. 2017350022.
Acknowledgments
We thank Dr. Rongfeng Li from Johns Hopkins University, United States, for the helpful comments and discussion of the paper.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmicb.2018.00868/full#supplementary-material
Footnotes
References
1
Axenov-Gibanov D. V. Voytsekhovskaya I. V. Tokovenko B. T. Protasov E. S. Gamaiunov S. V. Rebets Y. V. et al (2016). Actinobacteria isolated from an underground lake and moonmilk speleothem from the biggest conglomeratic karstic cave in Siberia as sources of novel biologically active compounds.PLoS One11:e0149216. 10.1371/journal.pone.0149216
2
Azman A. S. Othman I. Velu S. S. Chan K. G. Lee L. H. (2015). Mangrove rare actinobacteria: taxonomy, natural compound, and discovery of bioactivity.Front. Microbiol.6:856. 10.3389/fmicb.2015.00856
3
Bassetti M. Merelli M. Temperoni C. Astilean A. (2013). New antibiotics for bad bugs: where are we?Ann. Clin. Microbiol. Antimicrob.121–15. 10.1186/1476-0711-12-22
4
Bérdy J. (2012). Thoughts and facts about antibiotics: where we are now and where we are heading.J. Antibiot.651–11. 10.1038/ja.2012.54
5
Biswas K. Choudhury J. D. Mahansaria R. Saha M. Mukherjee J. (2017). Streptomyces euryhalinus sp. nov., a new actinomycete isolated from a mangrove forest.J. Antibiot.70747–753. 10.1038/ja.2017.3
6
Boucher H. W. Talbot G. H. Bradley J. S. Edwards J. E. Gilbert J. D. Rice L. B. et al (2009). Bad bugs, no drugs: no ESKAPE! an update from the infectious diseases society of America.Clin. Infect. Dis.481–12. 10.1086/595011
7
Buonaurio R. Moretti C. da Silva D. P. Cortese C. Ramos C. Venturi V. (2015). The olive knot disease as a model to study the role of interspecies bacterial communities in plant disease.Front. Plant Sci.6:434. 10.3389/fpls.2015.00434
8
Chen L. Chai W. Y. Wang W. L. Song T. F. Lian X. Y. Zhang Z. Z. (2017). Cytotoxic bagremycins from mangrove-derived Streptomyces sp. Q22.J. Nat. Prod.801450–1456. 10.1021/acs.jnatprod.6b01136
9
Coombs J. T. Franco C. M. M. (2003). Isolation and identification of actinobacteria from surface-sterilized wheat roots.Appl. Environ. Microbiol.695603–5608. 10.1128/AEM.69.9.5603-5608.2003
10
Ding L. Goerls H. Dornblut K. Lin W. Maier A. Fiebig H. H. et al (2015). Bacaryolanes A-C, rare bacterial caryolanes from a mangrove endophyte.J. Nat. Prod.782963–2967. 10.1021/acs.jnatprod.5b00674
11
Ding L. Maier A. Fiebig H. H. Lin W. H. Peschel G. Hertweck C. (2012). Kandenols A-E, eudesmenes from an endophytic Streptomyces sp. of the mangrove tree Kandelia candel.J. Nat. Prod.752223–2227. 10.1021/np300387n
12
Ding L. Münch J. Goerls H. Maier A. Fiebig H. H. Lin W. H. et al (2010). Xiamycin, a pentacyclic indolosesquiterpene with selective anti-HIV activity from a bacterial mangrove endophyte.Bioorg. Med. Chem. Lett.206685–6687. 10.1016/j.bmcl.2010.09.010
13
Feling R. H. Buchanan G. O. Mincer T. J. Kauffman C. A. Jensen P. R. Fenical W. (2003). Salinosporamide A: a highly cytotoxic proteasome inhibitor from a novel microbial source, a marine bacterium of the new genus Salinospora.Angew. Chem. Int. Ed.42355–357. 10.1002/anie.200390115
14
Felsenstein J. (1985). Confidence limits on phylogenies: an approach using the bootstrap society.Evolution39783–791. 10.1111/j.1558-5646.1985.tb00420.x
15
Fu S. Wang F. Li H. Bao Y. Yang Y. Shen H. et al (2016). Secondary metabolites from marine-derived Streptomyces antibioticus strain H74-21.Nat. Prod. Res.302460–2467. 10.1080/14786419.2016.1201668
16
Genilloud O. (2017). Actinomycetes: still a source of novel antibiotics.Nat. Prod. Rep.341203–1232. 10.1039/c7np00026j
17
Gohain A. Gogoi A. Debnath R. Yadav A. Singh B. P. Gupta V. K. et al (2015). Antimicrobial biosynthetic potential and genetic diversity of endophytic actinomycetes associated with medicinal plants.FEMS. Microbiol. Lett.3621–10. 10.1093/femsle/fnv158
18
Golinska P. Wypij M. Agarkar G. Rathod D. Dahm H. Rai M. (2015). Endophytic actinobacteria of medicinal plants: diversity and bioactivity.Antonie Van Leeuwenhoek108267–289. 10.1007/s10482-015-0502-7
19
Hamada M. Saitou S. Tamura T. (2017). Arenivirga flava gen. nov., sp. nov., a new member of the family Microbacteriaceae isolated from a mangrove tidal flat.Int. J. Syst. Evol. Microbiol.673318–3322. 10.1099/ijsem.0.002113
20
Han Y. Tian E. Xu D. B. Ma M. Deng Z. X. Hong K. (2016). Halichoblelide D, a new elaiophylin derivative with potent cytotoxic activity from mangrove-derived Streptomyces sp. 219807.Molecules21:E970. 10.3390/molecules21080970
21
Hassan S. S. Shaikh A. L. (2017). Marine actinobacteria as a drug treasure house.Biomed. Pharmacother.8746–57. 10.1016/j.biopha.2016.12.086
22
Hong K. Gao A. H. Xie Q. Y. Gao H. Zhuang L. Lin H. P. et al (2009). Actinomycetes for marine drug discovery isolated from mangrove soils and plants in China.Mar. Drugs724–44. 10.3390/md7010024
23
Huang H. Erickson R. Balasubramanian P. Hsieh T. Conner R. (2009). Resurgence of bacterial wilt of common bean in North America.Can. J. Plant Pathol.31290–300. 10.1080/07060660909507603
24
Jensen P. R. Dwight R. Fenical W. (1991). Distribution of actinomycetes in near-shore tropical marine sediments.Appl. Environ. Microbiol.571102–1108. Available at: http://aem.asm.org/content/57/4/1102.full.pdf+html
25
Jiang Z. K. Guo L. Chen C. Liu S. W. Zhang L. Dai S. J. et al (2015). Xiakemycin A, a novel pyranonaphthoquinone antibiotic, produced by the Streptomyces sp. CC8-201 from the soil of a karst cave.J. Antibiot.68771–774. 10.1038/ja.2015.70
26
Jiang Z. K. Pan Z. Li F. N. Li X. J. Liu S. W. Tuo L. et al (2017). Marmoricola endophyticus sp. nov., an endophytic actinobacterium isolated from Thespesia populnea.Int. J. Syst. Evol. Microbiol.674379–4384. 10.1099/ijsem.0.002297
27
Jin L. Lee H. G. Kim H. S. Ahn C. Y. Oh H. M. (2013). Amnibacterium soli sp. nov., an actinobacterium isolated from grass soil.Int. J. Syst. Evol. Microbiol.634750–4753. 10.1099/ijs.0.052506-0
28
Khan S. N. Khan A. U. (2016). Breaking the spell: combating multidrug resistant ‘superbugs.Front. Microbiol.7:174. 10.3389/fmicb.2016.00174
29
Khieu T. N. Liu M. J. Nimaichand S. Quach N. T. Ky S. C. Phi Q. T. et al (2015). Characterization and evaluation of antimicrobial and cytotoxic effects of Streptomyces sp. HUST012 isolated from medicinal plant Dracaena cochinchinensis Lour.Front. Microbiol.6:574. 10.3389/fmicb.2015.00574
30
Kim M. Oh H. S. Park S. C. Chun J. (2014). Towards a taxonomic coherence between average nucleotide identity and 16S rRNA gene sequence similarity for species demarcation of prokaryotes.Int. J. Syst. Evol. Microbiol.64346–351. 10.1099/ijs.0.059774-0
31
Kim O. S. Cho Y. J. Lee K. Yoon S. H. Kim M. Na H. et al (2012). Introducing EzTaxone: a prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species.Int. J. Syst. Evol. Microbiol.62716–721. 10.1099/ijs.0.038075-0
32
Kim S. J. Lee S. S. (2011). Amnibacterium kyonggiense gen. nov., sp. nov., a new member of the family Microbacteriaceae.Int. J. Syst. Evol. Microbiol.61155–159. 10.1099/ijs.0.018788-0
33
Kyeremeh K. Acquah K. S. Camas M. Tabudravu J. Houssen W. Deng D. et al (2014). Butrepyrazinone, a new pyrazinone with an unusual methylation pattern from a Ghanaian Verrucosispora sp. K51G.Mar. Drugs125197–5208. 10.3390/md12105197
34
Lane D. J. (1991). “16S/23S rRNA sequencing,” inNucleic Acid Techniques in Bacterial SystematicsedsStackebrandtE.GoodfellowM. (New York, NY: John Wiley and Sons) 115–175.
35
Law J. W. Ser H. L. Duangjai A. Saokaew S. Bukhari S. I. Khan T. M. et al (2017). Streptomyces colonosanans sp. nov., a novel actinobacterium isolated from Malaysia mangrove soil exhibiting antioxidative activity and cytotoxic potential against human colon cancer cell lines.Front. Microbiol.8:877. 10.3389/fmicb.2017.00877
36
Li F. Tuo L. Su Z. W. Wei X. Q. Zhang X. Y. Gao C. H. et al (2017). Nocardioides sonneratiae sp. nov., an endophytic actinomycete isolation from a branch of Sonneratia apetala.Int. J. Syst. Evol. Microbiol.672592–2597. 10.1099/ijsem.0.001979
37
Li F. N. Pan Z. Tuo L. Liu S. W. Zuo X. Chen L. et al (2017). Studies on the diversity and novelty of endophytic actinobacteria isolated from mangrove plants collected in Macao.Chin. J. Antibiot.4226–34. 10.13461/j.cnki.cja.005906
38
Li J. Dai S. J. Tuo L. Jiang Z. K. Liu S. W. Jiang M. G. et al (2016). Diversity and antimicrobial activity of endophytic actinobacteria isolated from eumangroves collected in DongzhaiGang of Hainan Province.Microbiol. China431753–1765. 10.13344/j.microbiol.china.150680
39
Li L. Tang Y. L. Wei B. Xie Q. Y. Deng Z. X. Hong K. (2013). Micromonospora sonneratiae sp. nov., isolated from a root of Sonneratia apetala.Int. J. Syst. Evol. Microbiol.632383–2388. 10.1099/ijs.0.043570-0
40
Liang S. C. Liu J. F. Liang M. Z. (2004). Ecological study on the mangrove communities in beilun hekou national nature reserve.J. Guangxi Normal Univ.2270–76.
41
Lin W. H. Li L. Y. Fu H. Z. Sattler I. Huang X. S. Grabley S. (2005). New cyclopentenone derivatives from an endophytic Streptomyces sp. Isolated from the mangrove plant Aegiceras comiculatum.J. Antibiot.58594–598. 10.1038/ja.2005.81
42
Ling L. L. Schneider T. Peoples A. J. Spoering A. L. Engels I. Conlon B. P. et al (2015). A new antibiotic kills pathogens without detectable resistance.Nature517455–459. 10.1038/nature14098
43
Liu S. W. Tuo L. Li X. J. Li F. N. Li J. Jiang M. G. et al (2017). Mangrovihabitans endophyticus gen. nov., sp. nov., a new member of the family Micromonosporaceae isolated from Bruguiera sexangula.Int. J. Syst. Evol. Microbiol.671629–1636. 10.1099/ijsem.0.001764
44
Liu S. W. Xu M. Tuo L. Li X. J. Hu L. Chen L. et al (2016). Phycicoccus endophyticus sp. nov., an endophytic actinobacterium isolated from Bruguiera gymnorhiza.Int. J. Syst. Evol. Microbiol.661105–1111. 10.1099/ijsem.0.000842
45
Mangamuri U. K. Muvva V. Poda S. Naragani K. Munaganti R. K. Chitturi B. et al (2016a). Bioactive metabolites produced by Streptomyces cheonanensis VUK-A from Coringa mangrove sediments: isolation, structure elucidation and bioactivity.3 Biotech6:63. 10.1007/s13205-016-0398-6
46
Mangamuri U. K. Vijayalakshmi M. Poda S. Manavathi B. Chitturi B. Yenamandra V. (2016b). Isolation and biological evaluation of N-(4-aminocyclooctyl)-3 5-dinitrobenzamide, a new semisynthetic derivative from the Mangrove-associated actinomycete Pseudonocardia endophytica VUK-10.3 Biotech6:158. 10.1007/s13205-016-0472-0
47
Manivasagana P. Kanga K. H. Sivakumarb K. Eunice C. Y. Chanc L. Oha H. M. et al (2014). Marine actinobacteria: an important source of bioactive natural products.Environ. Toxicol. Pharmacol.38172–188. 10.1016/j.etap.2014.05.014
48
Matsumoto A. Takahashi Y. (2017). Endophytic actinomycetes: promising source of novel bioactive compounds.J. Antibiot.70514–519. 10.1038/ja.2017.20
49
Mohammadipanah F. Wink J. (2015). Actinobacteria from arid and desert habitats: diversity and biological activity.Front. Microbiol.6:1541. 10.3389/fmicb.2015.01541
50
Nalini M. S. Prakash H. S. (2017). Diversity and bioprospecting of actinomycete endophytes from the medicinal plants.Lett. Appl. Microbiol.64261–270. 10.1111/lam.12718
51
Osdaghi E. Taghavi S. M. Fazliarab A. Elahifard E. Lamichhane J. R. (2015). Characterization, geographic distribution and host range of Curtobacterium flaccumfaciens: an emerging bacterial pathogen in Iran.Crop Prot.78185–192. 10.1016/j.cropro.2015.09.015
52
Osterman I. A. Komarova E. S. Shiryaev D. I. Korniltsev I. A. Khven I. M. Lukyanov D. A. et al (2016). Sorting out antibiotics’ mechanisms of action: a double fluorescent protein reporter for high throughput screening of ribosome and DNA biosynthesis inhibitors.Antimicrob. Agents Chemother.607481–7489.10.1128/AAC.02117-16
53
Passari A. K. Mishra V. K. Saikia R. Gupta V. K. Singh B. P. (2015). Isolation, abundance and phylogenetic affiliation of endophytic actinomycetes associated with medicinal plants and screening for their in vitro antimicrobial biosynthetic potential.Front. Microbiol.6:273. 10.3389/fmicb.2015.00273
54
Pendleton J. N. Gorman S. P. Gilmore B. F. (2013). Clinical relevance of the ESKAPE pathogens.Expert Rev. Anti Infect. Ther.11297–308. 10.1586/eri.13.12
55
Qin S. Li J. Chen H. H. Zhao G. Z. Zhu W. Y. Jiang C. L. et al (2009). Isolation, diversity, and antimicrobial activity of rare Actinobacteria from medicinal plants of tropical rain forests in Xishuangbanna, China.Appl. Environ. Microbiol.756176–6186. 10.1128/AEM.01034-09
56
Qu Z. Bao X. D. Xie Q. Y. Zhao Y. X. Yan B. Dai H. F. et al (2018). Actinoplanes sediminis sp. nov., isolated from marine sediment.Int. J. Syst. Evol. Microbiol.6871–75. 10.1099/ijsem.0.002451
57
Rateb M. E. Houssen W. E. Arnold M. Abdelrahman M. H. Deng H. Harrison W. T. A. et al (2011). Chaxamycins A-D, bioactive ansamycins from a hyper-arid desert Streptomyces sp.J. Nat. Prod.741491–1499. 10.1021/np200320u
58
Saitou N. Nei M. (1987). The neighbor-joining method: a new method for reconstructing phylogenetic trees.Mol. Biol. Evol.4406–425. 10.1093/oxfordjournals.molbev.a040454
59
Ser H. L. Tan L. T. H. Law J. W. F. Chan K. G. Duangjai A. Saokaew S. et al (2017). Focused review: cytotoxic and antioxidant potentials of mangrove-derived Streptomyces.Front. Microbiol.8:2065. 10.3389/fmicb.2017.02065
60
Singh S. B. Young K. Silver L. L. (2017). What is an “ideal” antibiotic? Discovery challenges and path forward.Biochem. Pharmacol.13363–73. 10.1016/j.bcp.2017.01.003
61
Spellberg B. Guidos R. Gilbert D. Bradley J. Boucher H. W. Scheld W. M. et al (2008). The epidemic of antibiotic-resistant infections: a call to action for the medical community from the infectious diseases society of America.Clin. Infect. Dis.46155–164. 10.1086/524891
62
Sun C. H. Li F. N. Cai Z. Wu L. (2017). Brief introduction of mangrove researches including drug discovery granted by National Natural Science Foundation of China from 1986 to 2016.Chin. J. Antibiot.42241–248. 10.13461/j.cnki.cja.005904
63
Talbot G. H. Bradley J. Edwards J. J. E. Gilbert D. Scheld M. Bartlett J. G. (2006). Bad bugs need drugs: an update on the development pipeline from the antimicrobial availability task force of the infectious diseases society of America.Clin. Infect. Dis.42657–668. 10.1086/499819
64
Tamura K. Peterson D. Peterson N. Stecher G. Nei M. Kumar S. (2011). MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods.Mol. Biol. Evol.282731–2739. 10.1093/molbev/msr121
65
Tan L. T. H. Ser H. L. Yin W. F. Chan K. G. Lee L. H. Goh B. H. (2015). Investigation of antioxidative and anticancer potentials of Streptomyces sp. MUM256 isolated from Malaysia mangrove soil.Front. Microbiol.6:1316. 10.3389/fmicb.2015.01316
66
Tian X. L. Cao L. X. Tan H. M. Han W. Q. Chen M. Liu Y. H. et al (2007). Diversity of cultivated and uncultivated actinobacterial endophytes in the stems and roots of rice.Microb. Ecol.53700–707. 10.1007/s00248-006-9163-4
67
Trujillo M. E. Riesco R. Benito P. Carro L. (2015). Endophytic actinobacteria and the interaction of Micromonospora and nitrogen fixing plants.Front. Microbiol.6:1341. 10.3389/fmicb.2015.01341
68
Tuo L. Li F. N. Pan Z. Lou I. Guo M. Lee S. M. Y. et al (2016a). Nakamurella endophytica sp. nov., a novel endophytic actinobacterium isolated from the bark of Kandelia candel.Int. J. Syst. Evol. Microbiol.661577–1582. 10.1099/ijsem.0.000923
69
Tuo L. Li J. Liu S. W. Liu Y. Hu L. Chen L. et al (2016b). Microlunatus endophyticus sp. nov., an endophytic actinobacterium isolated from bark of Bruguiera sexangula.Int. J. Syst. Evol. Microbiol.66481–486. 10.1099/ijsem.0.000779
70
Undabarrena A. Beltrametti F. Claverías F. P. González M. Moore E. R. Seeger M. et al (2016). Exploring the diversity and antimicrobial potential of marine actinobacteria from the comau fjord in Northern Patagonia, Chile.Front. Microbiol.7:1135. 10.3389/fmicb.2016.01135
71
Wang F. F. Xu M. J. Li Q. S. Sattler I. Lin W. H. (2010). p-Aminoacetophenonic acids produced by a mangrove endophyte Streptomyces sp. (strain HK10552).Molecules152782–2790. 10.3390/molecules15042782
72
Wang W. L. Song T. F. Chai W. Y. Chen L. Chen L. Lian X. Y. et al (2017). Rare polyene-polyol macrolides from mangrove-derived Streptomyces sp. ZQ4BG.Sci. Rep.7:1703. 10.1038/s41598-017-01912-z
73
Wilson Z. E. Brimble M. A. (2009). Molecules derived from the extremes of life.Nat. Prod. Rep.2644–71. 10.1039/b800164m
74
Xie Q. Y. Wang C. Wang R. Qu Z. Lin H. P. Goodfellow M. et al (2011). Jishengella endophytica gen. nov., sp. nov., a new member of the family Micromonosporaceae.Int. J. Syst. Evol. Microbiol.611153–1159. 10.1099/ijs.0.025288-0
75
Xu D. B. Ye W. W. Han Y. Deng Z. Y. Hong K. (2014). Natural products from mangrove actinomycetes.Mar. Drugs122590–2613. 10.3390/md12052590
76
Xu M. Li J. Dai S. J. Gao C. Y. Liu J. M. Tuo L. et al (2016). Study on diversity and bioactivity of actinobacteria isolated from mangrove plants collected from Zhanjiang in Guangdong Province.Chin. J. Antibiot.4126–34. 10.13461/j.cnki.cja.005662
77
Xu M. J. Liu X. J. Xu J. Zhang Z. G. (2015). New anti-fibrotic drug candidates from mangrove-derived Streptomyces xiamenensis suppressing local inflammation and mechanical stress in Hypertrophic Scars.J. Investig. Dermatol. Symp. Proc.1740–41. 10.1038/jidsymp.2015.16
78
Yan L. L. Han N. N. Zhang Y. Q. Yu L. Y. Chen J. Wei Y. Z. et al (2010). Antimycin A18 produced by an endophytic Streptomyces albidoflavus isolated from a mangrove plant.J. Antibiot.63259–261. 10.1038/ja.2010.21
79
Ye X. Chai W. Lian X. Y. Zhang Z. (2017). Novel propanamide analogue and antiproliferative diketopiperazines from mangrove Streptomyces sp. Q24.Nat. Prod. Res.311390–1396. 10.1080/14786419.2016.1253079
80
Yuan G. Hong K. Lin H. P. She Z. G. Li J. (2013). New azalomycin F analogs from mangrove Streptomyces sp. 211726 with activity against microbes and cancer cells.Mar. Drugs11817–829. 10.3390/md11030817
81
Zhou S. Q. Huang X. L. Huang D. Y. Hu X. W. Chen J. L. (2010). A rapid method for extracting DNA from actinomycetes by Chelex-100.BiotechnologyBulletin2123–125.
Summary
Keywords
mangrove plants, endophytic actinobacteria, diversity, antimicrobial activity, novel species
Citation
Jiang Z-k, Tuo L, Huang D-l, Osterman IA, Tyurin AP, Liu S-w, Lukyanov DA, Sergiev PV, Dontsova OA, Korshun VA, Li F-n and Sun C-h (2018) Diversity, Novelty, and Antimicrobial Activity of Endophytic Actinobacteria From Mangrove Plants in Beilun Estuary National Nature Reserve of Guangxi, China. Front. Microbiol. 9:868. doi: 10.3389/fmicb.2018.00868
Received
26 December 2017
Accepted
16 April 2018
Published
04 May 2018
Volume
9 - 2018
Edited by
Wen-Jun Li, Sun Yat-sen University, China
Reviewed by
Isao Yumoto, National Institute of Advanced Industrial Science and Technology (AIST), Japan; Learn-Han Lee, Monash University Malaysia, Malaysia; Virginia Helena Albarracín, Center for Electron Microscopy (CIME), Argentina; Zhiyong Li, Shanghai Jiao Tong University, China
Updates
Copyright
© 2018 Jiang, Tuo, Huang, Osterman, Tyurin, Liu, Lukyanov, Sergiev, Dontsova, Korshun, Li and Sun.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Cheng-hang Sun, chenghangsun@hotmail.com; sunchenghang@imb.pumc.edu.cn
This article was submitted to Extreme Microbiology, a section of the journal Frontiers in Microbiology
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.