- 1Division of Veterinary Public Health, ICAR-Indian Veterinary Research Institute, Izatnagar, India
- 2Department of Veterinary Pathology, Nagpur Veterinary College, Nagpur, India
- 3ICAR-National Research Centre on Meat, Hyderabad, India
The study evaluated the in vitro antimicrobial and antibiofilm efficacy of an antimicrobial peptide (AMP), lactoferricin (17–30) [Lfcin (17–30)], against biofilm-forming multi-drug-resistant (MDR) strains of enteroaggregative Escherichia coli (EAEC), and subsequently, the in vivo antimicrobial efficacy was assessed in a Galleria mellonella larval model. Initially, minimum inhibitory concentration (MIC; 32 μM), minimum bactericidal concentration (MBC; 32 μM), and minimum biofilm eradication concentration (MBEC; 32 μM) of Lfcin (17–30) were determined against MDR-EAEC field isolates (n = 3). Lfcin (17–30) was tested stable against high-end temperatures (70 and 90°C), physiological concentration of cationic salts (150 mM NaCl and 2 mM MgCl2), and proteases (proteinase-K and lysozyme). Further, at lower MIC, Lfcin (17–30) proved to be safe for sheep RBCs, secondary cell lines (HEp-2 and RAW 264.7), and beneficial gut lactobacilli. In the in vitro time-kill assay, Lfcin (17–30) inhibited the MDR-EAEC strains 3 h post-incubation, and the antibacterial effect was due to membrane permeation of Lfcin (17–30) in the inner and outer membranes of MDR-EAEC. Furthermore, in the in vivo experiments, G. mellonella larvae treated with Lfcin (17–30) exhibited an increased survival rate, lower MDR-EAEC counts (P < 0.001), mild to moderate histopathological changes, and enhanced immunomodulatory effect and were safe to larval cells when compared with infection control. Besides, Lfcin (17–30) proved to be an effective antibiofilm agent, as it inhibited and eradicated the preformed biofilm formed by MDR-EAEC strains in a significant (P < 0.05) manner both by microtiter plate assay and live/dead bacterial quantification-based confocal microscopy. We recommend further investigation of Lfcin (17–30) in an appropriate animal model before its application in target host against MDR-EAEC strains.
Introduction
In recent times, enteroaggregative Escherichia coli (EAEC) has been regarded as an emerging foodborne pathogen, and it has been frequently associated with the epidemic as well as endemic diarrheal episodes (Lima et al., 2018). In human infants, EAEC damages the intestinal epithelium, leading to poor nutritional status and thereby intellectual deficits (Lima et al., 2018), whereas in animals, it produces intestinal changes and diarrheal episodes (Kolenda et al., 2015). Recently, Verotoxin-producing EAEC strains (O104: H4) identified from the German foodborne outbreak of 2011 have evidenced massive economic losses (Boll et al., 2020). Generally, EAEC is regarded as a heterogeneous pathogen; the pathogenicity of EAEC is described initially by its surface adherence to the intestinal mucosa, followed by biofilm formation and toxin release, which often ends up with an inflammatory response (Lima et al., 2018). The biofilm formation by EAEC is correlated well with the persistence of infection and recalcitrance to empirical antimicrobial treatment (Lin et al., 2017; Petro et al., 2020). This persistent colonization of EAEC leads to carrier status, enabling antibiotic pressure, which results in frightening levels of multi-drug resistance. Globally, drug resistance among EAEC strains toward first-line antibiotics (beta-lactams and fluoroquinolones) has been well evident (Lima et al., 2018). Moreover, multi-drug-resistant (MDR)-EAEC strains were recovered from food handlers (Oundo et al., 2008), diarrheal children, travelers’ diarrhea (Hebbelstrup Jensen et al., 2018), mangrove estuaries (Ghaderpour et al., 2015), and surface water (Canizalez-Roman et al., 2019).
Currently, with the advent of AMR crisis (Ghosh et al., 2019; Molnar, 2019) and limited discovery of newer antibiotics, the focus has now been shifted toward identifying effective alternative therapeutics (Haney et al., 2019). Many promising approaches have been reported for addressing bacterial resistance, which includes antibodies targeting specific pathogens, phage therapy, exolysins, endolysins including enzybiotics, vaccines, prebiotic and probiotic strains, antimicrobial peptides (AMPs), and phytochemicals (Ghosh et al., 2019). Of late, cationic AMPs have gained considerable attention concerning their antimicrobial and antibiofilm technology solutions (Haney et al., 2019). AMPs are evolutionarily conserved entities present in a wide range of organisms and have been heralded as a promising alternative to antibiotics (Mookherjee et al., 2020). The complex and multi-modal action of AMPs enables recalcitrance to develop perdurable microbial resistance that offers yet another advantage over conventional antibiotics (Kumar et al., 2018; Haney et al., 2019). However, long-chain peptides increase their cost for production and thereby investigation (de la Fuente-Núñez et al., 2015); hence, short-chain synthetic peptides have been attempted (de Zoysa et al., 2015). In particular, a 14-amino-acid residue of the Lactoferricin protein, namely, Lfcin (17–30), was proposed to exhibit broad-spectrum antimicrobial activities predominantly by way of membrane permeabilization compared to that of the naive milk protein, lactoferrin (Silva et al., 2017). Lfcin (17–30) has been identified to act against bacteria, fungi, amoeba, stimulants of bio-warfare agents, and even bacterial biofilms (van der Kraan et al., 2005; Xu et al., 2010; Sijbrandij et al., 2017; Acosta-Smith et al., 2018; Díaz-Godínez et al., 2019). Nevertheless, its use against MDR pathogens, such as EAEC, remains unrevealed, barring a few systematic studies against pathogens like Klebsiella pneumoniae, Pseudomonas aeruginosa, and Acinetobacter baumannii (Reyes-Cortes et al., 2016; Pollini et al., 2017). Even studies addressing the antimicrobial and antibiofilm potential of Lfcin (17–30) against MDR-EAEC are lacking.
Furthermore, in vivo manifestations of EAEC have been established in various mammalian models, which involve logistical, ethical, and budgetary constraints (Philipson et al., 2013; Kumar et al., 2016). Therefore, as an alternative in vivo model, Galleria mellonella larvae have recently been explored to evaluate the therapeutic potential of novel candidates including AMPs against various bacterial pathogens, including MDR-EAEC (McCloskey et al., 2019; Vergis et al., 2019). Moreover, the short life span of larvae and their ability to simulate humans while exploring pathogens of public health significance (Wojda, 2017; Cutuli et al., 2019; Blasco et al., 2020) extends their importance as an excellent in vivo model to screen novel therapeutics including AMPs. However, studies addressing in vivo antibacterial efficacy of Lfcin (17–30) against MDR-EAEC have never been explored in the G. mellonella larvae model. The objectives of the study were to assess in vitro antimicrobial as well as antibiofilm efficacy of Lfcin (17–30) against biofilm-forming MDR-EAEC strains and later to evaluate its antimicrobial efficacy in a G. mellonella larval model. Simultaneously, in vitro stability, safety, and mechanism of action of Lfcin (17–30) against MDR-EAEC were also evaluated to explore its possible utility as a therapeutic candidate.
Materials and Methods
Bacterial Strains
The typical EAEC isolates with NCBI GenBank accession numbers KY941936.1 (MDR 1), KY941937.1 (MDR 2), and KY941938.1 (MDR 3) maintained in the laboratory repository of Division of Veterinary Public Health, Indian Veterinary Research Institute, Izatnagar, were re-validated by PCR (Vijay et al., 2015), confirmed using HEp-2 adherence assay (Cravioto et al., 1979) and subjected to antibiotic susceptibility testing (Clinical and Laboratory Standards Institute [CLSI], 2018). E. coli ATCC 25922 was used as the quality control strain for antibiotic susceptibility testing.
Antimicrobial Peptide
Lfcin (17–30) evaluated in this study was retrieved from BaAMPs (Di Luca et al., 2015), commercially synthesized from Shanghai Science Peptide Biological Technology, China, resuspended in phosphate-buffered saline (PBS; pH 7.40) with a final stock concentration of 10 mg/ml and stored at -20°C until further use (Supplementary Table S1). To compare the results of Lfcin (17–30), meropenem was used as an antibiotic treatment control throughout this study.
Characterization of Lfcin (17–30)
Initially, characterization of Lfcin (17–30) was performed by determining its minimum inhibitory concentration (MIC), minimum bactericidal concentration (MBC), in vitro stability assays (high-end temperatures, the physiological concentration of cationic salts and proteases), and in vitro safety assays (sheep erythrocyte hemolysis and secondary cell line cytotoxicity) (Supplementary Files S1–S3). The membrane permeabilization effect of Lfcin (17–30) was assessed by flow cytometry, whereas outer and inner membrane permeability of MDR-EAEC strains treated with 1 × and 2 × MIC levels of Lfcin (17–30) was carried out based on the nitrocefin activity as well as the release of cytoplasmic β-galactosidase activity, respectively (Supplementary File S4). Further, Lfcin (17–30) was evaluated for its antibacterial activity against beneficial lactobacilli (L. acidophilus MTCC 10307 and L. rhamnosus MTCC 1408) (Supplementary File S5).
In vitro Time-Kill Kinetics of MDR-EAEC With Lfcin (17–30)
The in vitro dose- and time-kill kinetics was assessed by co-incubating the log-phase cultures of each of MDR-EAEC isolates (ca. 1 × 107 CFU/ml) in cation-adjusted Mueller Hinton (CA-MH) broth with MIC (1 × and 2 × ) of Lfcin (17–30) (Supplementary Information S6). The desired inoculum for each MDR-EAEC strain and Lfcin (17–30) suspended in CA-MH broth was as follows: Group I, 107 CFU of MDR-EAEC (50 μl) with 1 × MIC of Lfcin (17–30) (50 μl); Group II, 107 CFU of MDR-EAEC (50 μl) with 2 × MIC of Lfcin (17–30) (50 μl); Group III, 107 CFU of MDR-EAEC (50 μl) with 10 μg/ml of Meropenem (50 μl); and Group IV, 107 CFU of MDR-EAEC (50 μl) in CA-MH broth (50 μl). The MDR-EAEC counts were enumerated (Miles et al., 1938) at 0, 30, 60, 90, 120, 150, and 180 min, and 24, 48, and 72 h post co-incubation.
In vivo Assays Using G. mellonella Model
The final instar of G. mellonella larvae was utilized for performing the in vivo assays (Morgan et al., 2014). The larvae (ca. 200–250 mg) inoculated with aliquots of MDR-EAEC suspensions (10 μl) by injection using Hamilton syringe (26 gauge) via the last right pro-leg were incubated at 37°C and the observations were noted. The larvae kept in a sterile environment were provided with ad lib food during the experiment.
The determined LD50 dose for each MDR-EAEC strain in the larvae was validated and employed further in the in vivo experiments to evaluate the antibacterial efficacy of Lfcin (17–30).
In vivo Antimicrobial Efficacy of Lfcin (17–30)
Galleria mellonella larvae were grouped 40 per group for in vivo antimicrobial testing of Lfcin (17–30) as follows: group I (MDR-EAEC infection control), groups II and III (infection + treatment groups), group IV (PBS control), and group V [Lfcin(17–30) control]. Optimized LD50 dose of MDR-EAEC (a cocktail of three strains) was administered to larvae of groups I to III, while MIC dose of Lfcin (17–30) and Meropenem was administered 3 h post-infection (pi) in groups II and III, respectively. Larval group IV was injected with sterile PBS, whereas MIC dose of Lfcin (17–30) was administered in group V.
The larvae were then observed for their survival rate, melanization (Supplementary Information S7), MDR-EAEC counts (Supplementary Information S8), hemocyte density (Supplementary Information S9) (Wand et al., 2013), lactate dehydrogenase (LDH) cytotoxicity assay (Supplementary Information S10) (Gibreel and Upton, 2013), and histopathology (Supplementary Information S11) (Perdoni et al., 2014), at an interval of 6 h till 24 h, followed by 24-h interval up to 120 h pi.
Biofilm Formation by MDR-EAEC Isolates
The biofilm formation by MDR-EAEC strains was qualitatively assessed by Congo red binding assay and hydrophobicity index (Microbial adhesion to Solvents; MATS), while the time-dependent biofilm-forming ability was assessed in different media (Supplementary Information S12) as a semi-qualitative method.
Determination of Minimum Biofilm Eradication Concentration (MBEC) of Lfcin (17–30) Against MDR-EAEC Biofilm Formation
The MBEC of Lfcin (17–30) was determined against the preformed (48 h) MDR-EAEC biofilm (Ceri et al., 1999). MBEC value is the lowest concentration of Lfcin (17–30) that prevented the MDR-EAEC re-growth. This MBEC value of Lfcin (17–30) was further used to study the eradication of biofilm formed by MDR-EAEC strains.
Inhibition and Elimination of MDR-EAEC Biofilm by Lfcin (17–30)
The efficacy of Lfcin (17–30) to inhibit and eliminate the biofilms of MDR-EAEC strains was evaluated by crystal violet (CV) staining as well as by the live/dead staining method (de Zoysa et al., 2015). Further, to quantify live and dead cells, the confocal images obtained were analyzed using Fiji ImageJ software ver. 1.51s (Schindelin et al., 2012). The varying bio-volume proportions of live and dead bacteria obtained after deducting the background score were plotted in intensity histogram and interpreted as a function of Red-Green intensity.
Statistical Analysis
All the experiments were repeated individually and independently thrice; the data obtained were reflected as mean ± standard deviation for each assay using GraphPad Prism 5.01 software (GraphPad Software Inc., San Diego, CA). A one-way analysis of variance (ANOVA) with Bonferroni multiple comparison post-test was used to compare the differences observed in the in vitro cell line cytotoxicity assay and to analyze biofilm data, while paired two-tailed “t” test was used to analyze the antibacterial effect of Lfcin (17–30) on beneficial lactobacilli. A two-way (repeated measures) ANOVA with Bonferroni multiple comparison post-test was used to analyze the in vitro and in vivo time-dependent antimicrobial assays [inner and outer membrane permeability and killing kinetic assays of Lfcin (17–30), in vivo bacterial burden, melanization, hemocyte enumeration, and LDH assays]. The probit regression model was used to determine the LD50 dose of MDR-EAEC strains, while the log-rank (Mantel-Cox) test and log-rank test for trends were used for in vivo G. mellonella larval survival curves. A P value of ≤0.01 was considered highly significant, while a P value ≤0.05 was considered statistically significant.
Results
In this study, all the three typical MDR-EAEC wild strains employed were found to be resistant to four or more classes of antibiotics and were ESBL producers (Supplementary Table S2).
Characterization of Lfcin (17–30)
The MIC and MBC values of Lfcin (17–30) against the tested MDR-EAEC strains were found to be equal (32 μM).
Lfcin (17–30) was found stable at high-end temperatures (70 and 90°C; Supplementary Table S3A), protease (proteinase-K and trypsin; Supplementary Table S3B), and physiological concentration of cationic salts (150 mM NaCl and 2 mM MgCl2; Supplementary Table S3C), as MIC and MBC values of Lfcin (17–30) were found unaltered.
Lfcin (17–30) was found to be non-hemolytic at lower (1 × and 2 × MIC) concentrations; however, at 4 × MIC, minimal hemolysis (<5%) was noted (Supplementary Table S4). Similarly, Lfcin (17–30) marginally reduced the viability of secondary cell lines (HEp-2 and RAW 264.7) tested in a concentration-dependent manner. At lower concentrations (1 × and 2 × ), remarkable morphological changes were not evident; however, at 4 × MIC, moderate to higher cytotoxicity was evident in HEp-2 and RAW 264.7 cells (Supplementary Figure S1).
Also, a non-significant (P > 0.05) antimicrobial effect was observed against beneficial lactobacilli (L. acidophilus and L. rhamnosus; Supplementary Figure S2), suggesting the safety of Lfcin (17–30) against beneficial lactobacilli.
Lfcin (17–30), at MIC (1 × ), exhibited remarkable damage to the cell membrane of MDR-EAEC strains (PI-positive cells >50%), as evidenced by flow cytometry (Supplementary Figure S3). Moreover, Lfcin (17–30) permeated the inner as well as outer membrane of all the MDR-EAEC strains in the uptake of ONPG (Figure 1A) and nitrocefin (Figure 1B), respectively, in a concentration- and time-dependent manner. Moreover, the onset and progress of both inner and outer membrane permeabilization by Lfcin (17–30) was significantly quicker (P < 0.01) than the antibiotic control, meropenem.
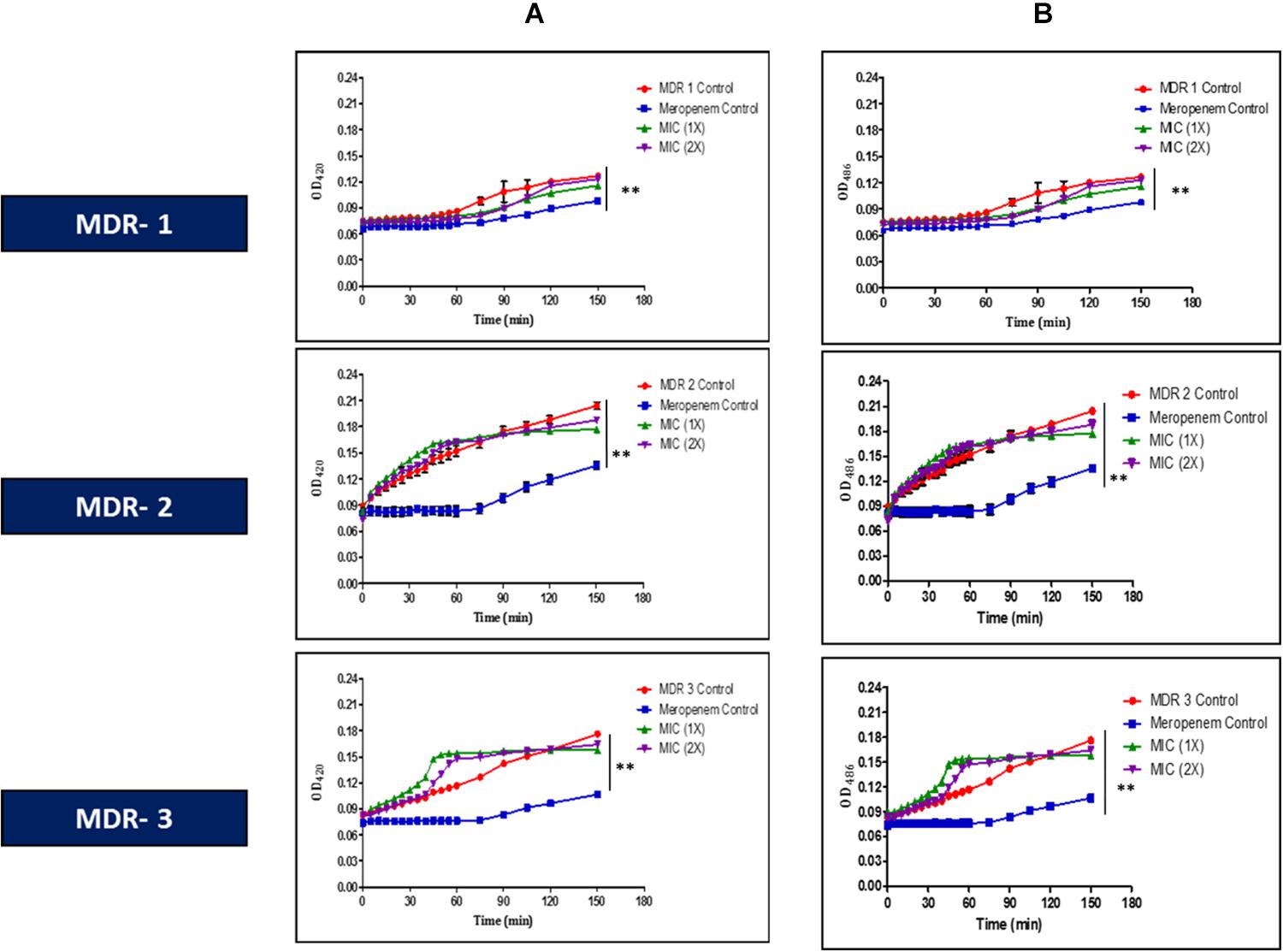
Figure 1. In vitro membrane permeability assay of Lfcin (17–30) in MDR-EAEC strains. MIC (1 × and 2 × ) concentrations of Lfcin (17–30) against MDR-EAEC strains employing ONPG (A) and nitrocefin (B), expressed as a function of time at 37°C. Hydrolysis of ONPG by β-galactosidase was used to monitor inner membrane permeabilization by absorbance at 420 nm, while hydrolysis of nitrocefin by β-lactamase was used to monitor outer membrane permeabilization by absorbance at 486 nm (***P < 0.001; **P < 0.01).
In vitro Time-Kill Kinetics of MDR-EAEC With Lfcin (17–30)
The antimicrobial effect of Lfcin (17–30) at 1 × and 2 × MIC was highly significant (P < 0.001) at 30 min post-coincubation (Figure 2). Further, none of the MDR-EAEC isolates exhibited any visible growth at 180 min post-coincubation. In contrast, an increased growth pattern was observed in the untreated control group at 30, 60, 90, 120, 150, and 180 min post-coincubation (Figure 2). Additionally, a highly significant (P < 0.001) reduction in bacterial counts was observed in the meropenem treatment group at 30 min post-coincubation, and after 60 min post-coincubation, complete inhibition of MDR-EAEC isolates was observed (Figure 2). Since no significant difference (P > 0.05) was observed between the MIC and MBC values of Lfcin (17–30) in inhibiting all three MDR-EAEC strains (Figure 2), further in vivo studies in G. mellonella larvae were carried out using the MIC value of Lfcin (17–30).
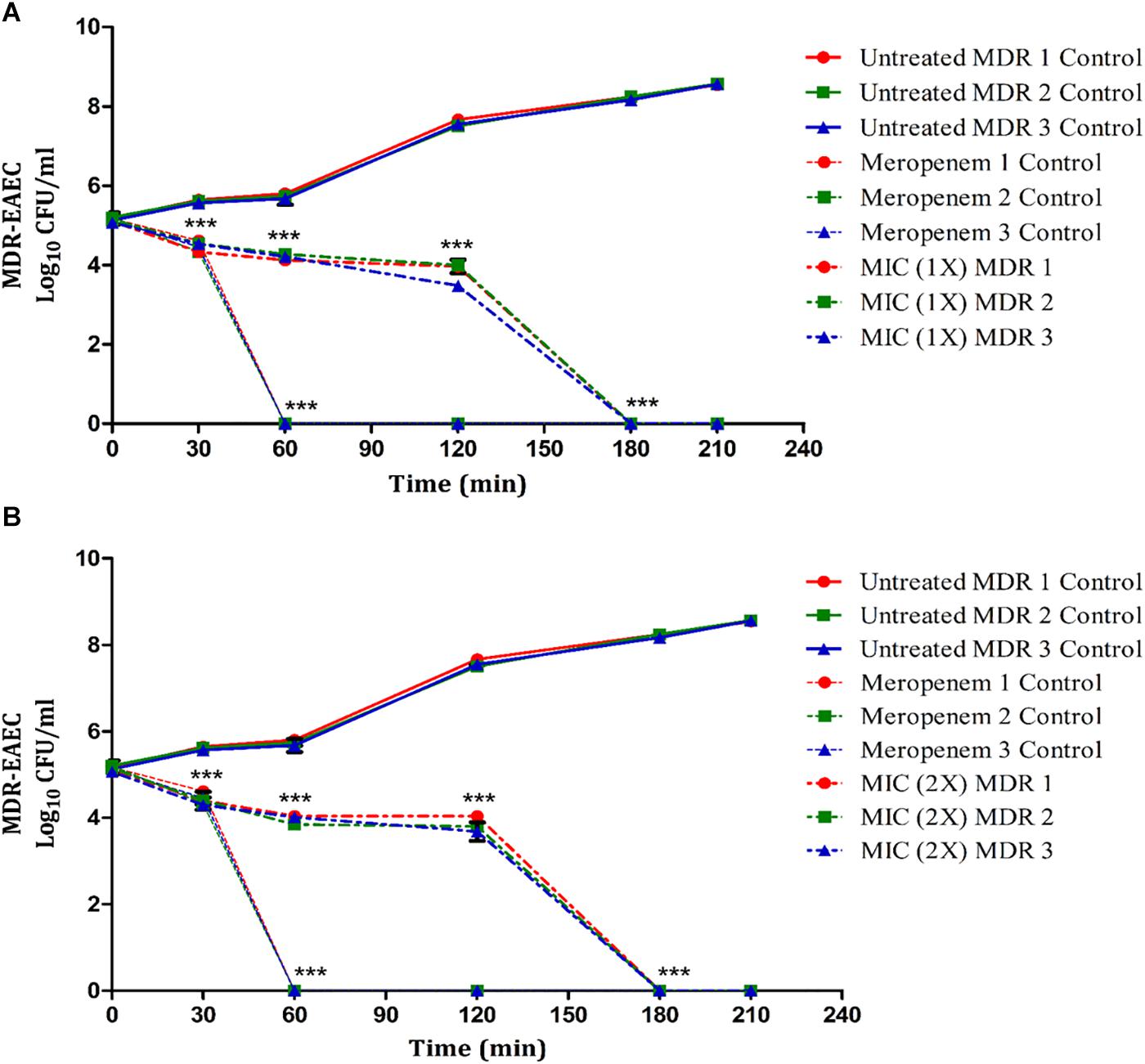
Figure 2. Dose- and time-dependent time-kill assay of MDR-EAEC isolates co-cultured with Lfcin (17–30). MDR-EAEC isolates (n = 3) were co-cultured with 1 × MIC of Lfcin (17–30) (A) and 2 × MIC of Lfcin (17–30) (B) in CA-MH broth at 37°C under static conditions with respective controls of MDR-EAEC isolates (untreated and meropenem-treated). Data expressed as the mean ± standard deviation (log10CFU/ml) of three independent experiments (***P < 0.001).
LD50 of MDR-EAEC Strains in the G. mellonella Larval Model
While optimizing the LD50 dose of MDR-EAEC, a concentration-dependent mortality rate was observed in the G. mellonella larvae survival plots (Supplementary Figure S4). An inoculum of 106 CFU/larvae was determined as the optimized LD50 dose of MDR-EAEC strains (Supplementary Figure S4).
In vivo Antimicrobial Efficacy of Lfcin (17–30)
In the MDR-EAEC infection control larval group, a survival rate of 52.50% was observed, while an enhanced survival rate (85%) was observed in the meropenem-treated group up to 120 h pi (Figure 3A). Further improved survival rate (90%) was noticed in the Lfcin (17–30)-treated infected larval group that corresponded to a significant log-rank Mantel–Cox test (P < 0.001) and log-rank test for trend (P < 0.05) (Figure 3A). All the uninfected larval control groups [PBS control and Lfcin (17–30) control] were found to be healthy with a 100% survival rate up to 120 h pi (Figure 3A).
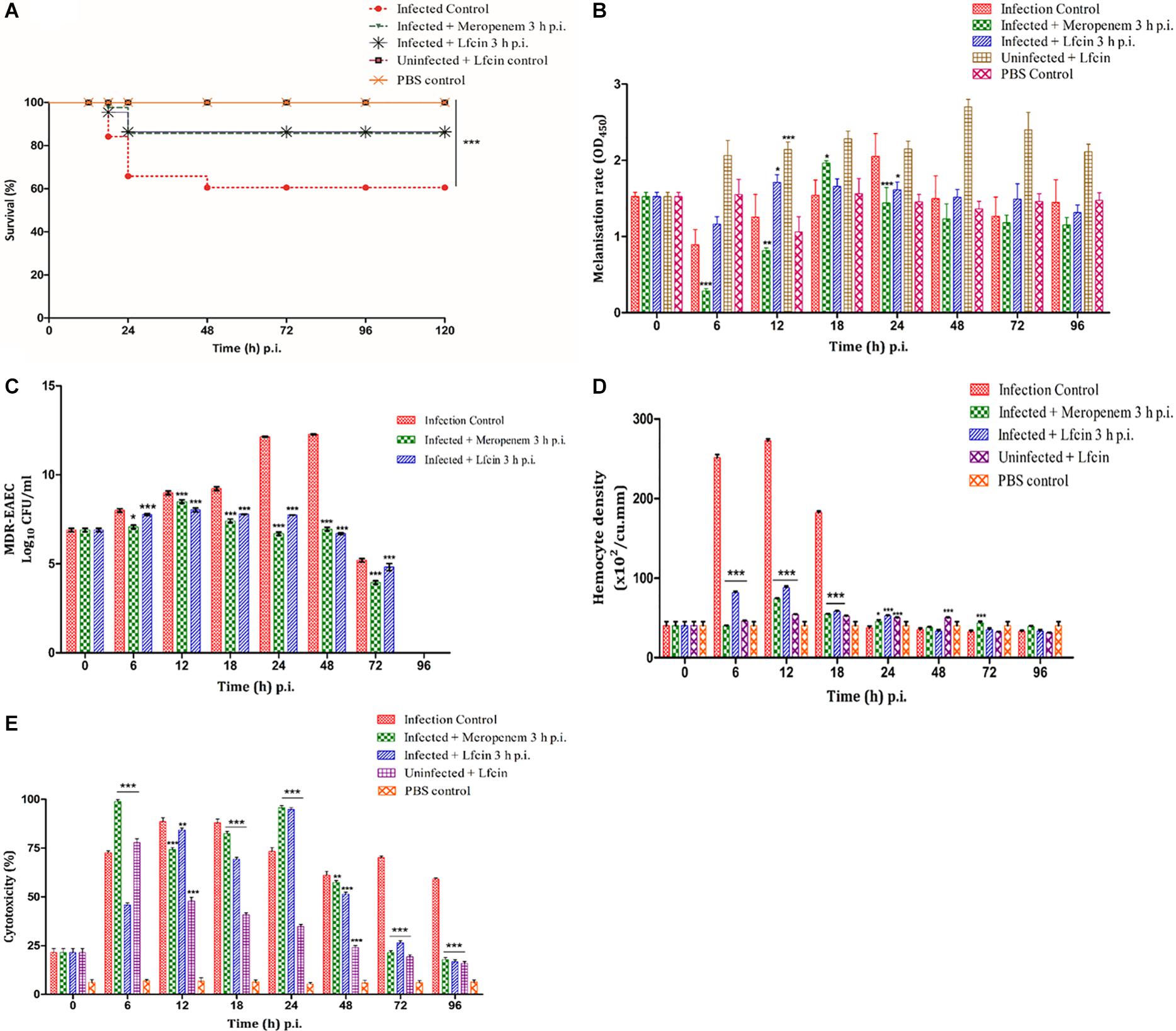
Figure 3. In vivo assays using G. mellonella model. Survival plot (A), melanization rate (B), MDR-EAEC counts (C), hemocyte density (D), and LDH cytotoxicity assay (E) of G. mellonella larvae infected with LD50 dose (106 CFU) of MDR-EAEC strains and treated with MIC (1 × ) of Lfcin (17–30) 3 h pi. MDR-EAEC-induced infection was treated with MIC of Lfcin (17–30), keeping respective controls [infection control, meropenem treatment, Lfcin (17–30) control, PBS control]. Data expressed as the mean ± standard deviation of three independent experiments (*P < 0.05; **P < 0.01; ***P < 0.001). Melanization rate was assessed by absorbance monitored at 450 nm, MDR-EAEC counts as log10CFU/ml of hemolymph on EMB agar plates supplemented with ampicillin (100 μg/plate), hemocyte density as cells/ml of hemolymph, and LDH cytotoxicity assay as the cytotoxicity (%) of larval hemolymph.
Melanization Assay
The rate of melanization was lower in the MDR-EAEC infection control larval group at 6 h pi, increased thereafter and reached its peak at 24 h pi, and was found to reduce at 48 h pi (Figure 3B). In the meropenem-treated infected group, the melanization rate was found to be lower at 6 h pi, peaked at 18 h pi, and gradually declined thereafter in a highly significant (P < 0.001) manner (Figure 3B). However, in the infected group treated with Lfcin (17–30), melanization was found to increase at 12 to 24 h pi; thereafter, the intensity of melanization was gradually declined (Figure 3B). In uninoculated larval groups treated with Lfcin (17–30), a slight increase in the melanization rate was observed at 6 h pi; thereafter, the melanization rate was observed at an increased intensity at 12 to 96 h pi (Figure 3B).
Enumeration of MDR-EAEC Counts
A significant reduction (P < 0.001) of MDR-EAEC counts was observed in the infected larval groups treated with Lfcin (17–30) at 24 h pi (mean 4.40 log reduction) and 48 h pi (mean 5.50 log reduction) as compared to the infected control group (Figure 3C). Further, MDR-EAEC was not detected in the hemolymph obtained from the uninoculated larval groups until 96 h pi (Figure 3C).
Enumeration of Hemocytes
The inoculated and the treatment groups of G. mellonella larvae revealed significantly (P < 0.001) increased hemocyte density at 6 h pi; later, hemocyte density peaked by 12 and reduced thereafter in a significant (P < 0.001) manner (Figure 3D); however, a significant difference (P > 0.05) in the hemocyte density was not noticed between all the larval groups at 72 to 96 h pi (Figure 3D).
LDH Assay
In the infected control group, LDH cytotoxicity increased in a highly significant manner (P < 0.001) at 6 h pi, peaked at 12 to 18 h pi, and retained cytotoxicity up to 96 h pi (Figure 3E). A significant (P < 0.001) increase in the cytotoxicity was noticed in the Lfcin (17–30)-treated infected larval group at 6 h pi and the cytotoxicity remained increased up to 48 h pi and thereafter a progressive reduction in cytotoxicity was observed (Figure 3E). Almost similar observations were noted in the infected group treated with meropenem (Figure 3E). In the uninoculated larval group treated with Lfcin (17–30), significantly increased cytotoxicity was observed at 6 to 18 h pi, and later it declined progressively (Figure 3E).
Histopathological Examination
All the groups, except for the infected control group, revealed no histopathological alteration at 6 and 12 h pi, while hemocytes were sparsely distributed with bare melanization at 12 to 18 h pi. However, hemocytes were more pronounced in the sub-cuticular area of the infected control group, at 12 to 18 h pi exhibiting bacterial phagocytic reaction with pronounced melanization and EAEC load surrounding the tubular organs. Further, cross-sections of the infected control larvae revealed clusters of hemocytes in the sub-cuticular area at 18 h pi exhibiting bacterial phagocytosis, which was reflected as finely stippled blue dots along with melanization; besides, bacterial load was observed around tubular organs. Nevertheless, Lfcin (17–30)-treated infected larvae and uninoculated control groups (groups IV and V) appeared healthy with sparse distribution of hemocytes exhibiting minimal melanization.
The inoculated control larvae exhibited increased pathological abnormalities at 24 and 48 h pi, which declined progressively at 72 h pi, while histopathological changes were mild to moderate in the Lfcin (17–30)-treated group at 24 h pi (Supplementary Figure S5), which declined progressively at later time points (48 and 72 h pi). Interestingly, pathological changes could not be appreciated in inoculated groups treated with Lfcin (17–30), the uninoculated control group, and the PBS control group.
Biofilm Formation by MDR-EAEC Isolates
Congo red binding assay revealed moderate to strong biofilm formation by MDR-2 and MDR-3 EAEC strains, while MDR-1 revealed weak biofilm formation (Supplementary Figure S6). Moreover, the hydrophobicity index indicated that all three MDR-EAEC strains employed were strongly basic and weakly acidic (Supplementary Figure S7). Further, the biofilm formation was highly significant (P < 0.001) in Dulbecco’s minimal essential medium (DMEM) containing 0.45% D-glucose on polystyrene surface at 48 h of incubation (Supplementary Figure S8). All the MDR-EAEC strains employed in this study were found to be moderate biofilm producers by microtiter plate assay (Wakimoto et al., 2004), as evidenced by OD595 values ranging from 0.375 to 0.537.
Determination of MBEC of Lfcin (17–30)
The MBEC values observed for Lfcin (17–30) were equal to the MBC values (32 μM) of Lfcin (17–30) against biofilm formed by the MDR-EAEC strains.
In vitro Efficacy of Lfcin (17–30) Against MDR-EAEC Biofilm
As evidenced by CV staining, the biofilm biomass of MDR-EAEC strains was reduced significantly at 24 h (P < 0.001) and 48 h (P < 0.01) on treatment with Lfcin (17–30) as compared to their respective controls (Figures 4A,B). Likewise, confocal microscopy also revealed a highly significant reduction in the biofilm biomass formed by MDR-EAEC isolates; however, significant inhibition in the biofilm biomass was observed at 24 h (P < 0.05) (Supplementary Figure S9) and 48 h (P < 0.01) (Supplementary Figure S9).
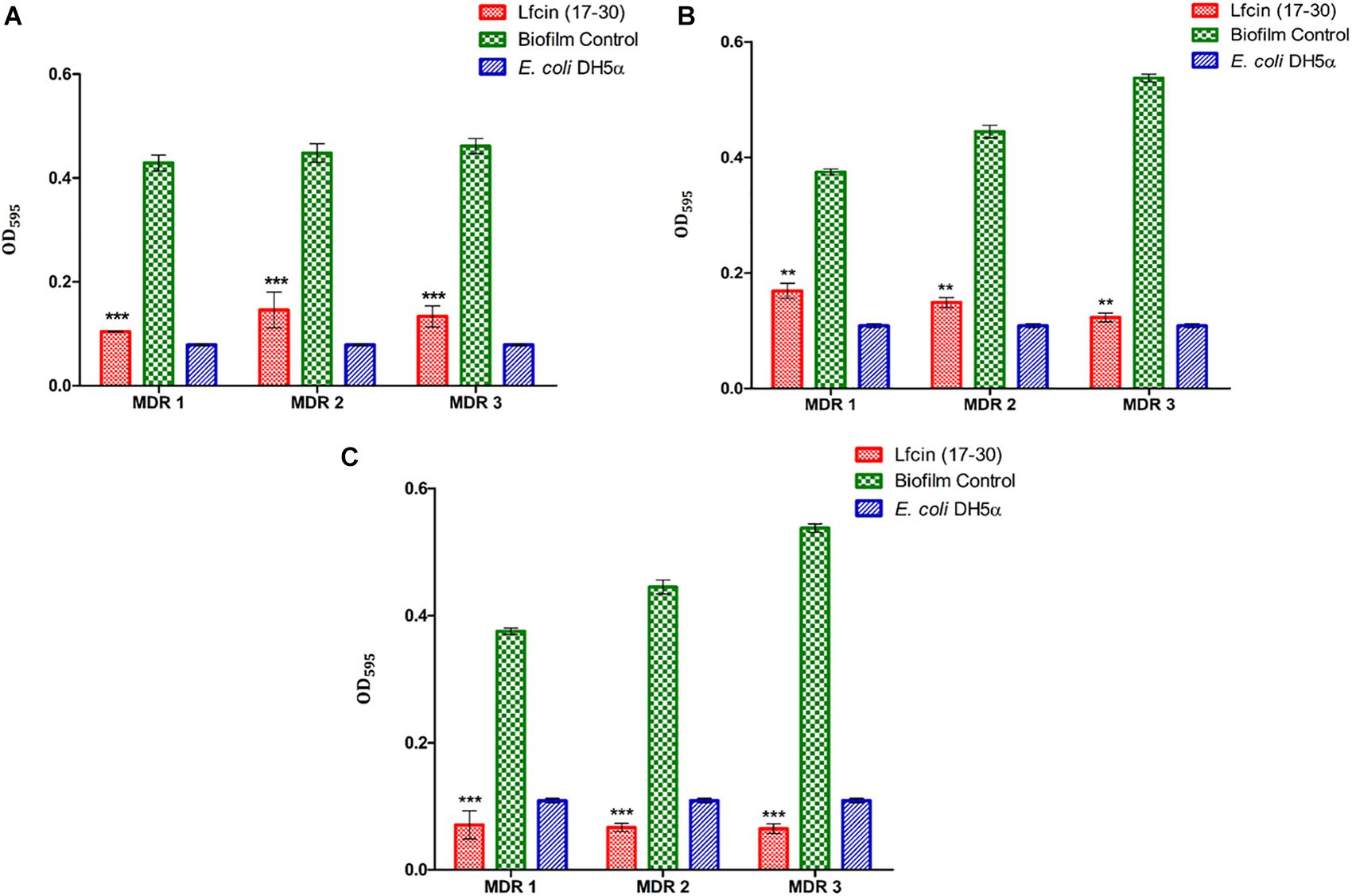
Figure 4. Inhibition and eradication of MDR-EAEC biofilm by Lfcin (17–30) by CV staining. Inhibition of MDR-EAEC biofilm by Lfcin (17–30) at 24 h (A), 48 h (B), and effect of treating MDR-EAEC preformed biofilms (48 h) with Lfcin (17–30) for an additional 24 h (C) by CV staining. Error bars indicate the standard deviation between strains. Control bars indicate corresponding MDR-EAEC strains (biofilm control) and E. coli DH5α biofilms, respectively, without AMP (***P < 0.001; **P < 0.01).
Lfcin (17–30) also eliminated the preformed biofilm of MDR-EAEC strains significantly (P < 0.001) as shown by CV staining (Figure 4C) and confocal microscopy (Supplementary Figure S10).
Discussion
The emergence of multi-drug resistance as a result of “selection pressure” enables the pathogens to tolerate various antibiotic classes suggested for empirical therapy (Davies and Davies, 2010). With the limited availability of effective antibiotics against drug-resistant pathogens, the focus has now been targeted toward alternative strategies for treating infections. Of late, owing to their multi-faceted antimicrobial, antibiofilm, as well as immunomodulatory potential, studies employing AMPs have attracted additional momentum. Besides, AMPs have not been widely reported to gain resistance, enabling them to be chosen for treating drug-resistant, chronic, as well as persistent infections (Ghosh et al., 2019). EAEC causes chronic and persistent diarrhea due to its inherent biofilm-forming capability, which often ends up damaging the intestinal epithelium of human infants and young animals (Lima et al., 2018). Of late, emerging trends in AMR, particularly resurgence of MDR-EAEC and its rapid dissemination, have been reported from various sources (Kong et al., 2015; Lima et al., 2018), which represent an alarming public health threat and clinical challenge.
In this study, we attempted to assess the in vitro antimicrobial as well as antibiofilm efficacy of the Lfcin (17–30), a short-chain cationic AMP, from the regularly updated biofilm active AMPs (BaAMPs) database (Xu et al., 2010) against biofilm-forming MDR-EAEC field isolates and later to evaluate its antimicrobial efficacy in a G. mellonella larval model. Short-chain peptides (12–50 amino acids), preferably with cationic amino acids and a high proportion of hydrophobic residues (∼50%), were testified to be operative against biofilm-forming bacterial pathogens (de la Fuente-Núñez et al., 2015). Lfcin (17–30), a tetradecapeptide, has been reported to produce depolarization, loss of cytoplasmic inner membrane integrity, and pH gradient, thereby exerting a bactericidal effect, particularly on E. coli and also affect intra-cellular activities (Haukland and Vorland, 2001).
Despite having an excellent antibacterial activity, the clinical translation of AMPs involves certain limitations such as thermostability, proteolytic degradation, and salt inactivation within the body (Mohamed et al., 2016). In the present study, Lfcin (17–30) could withstand high-end temperatures (70 and 90°C). Generally, such high-end temperatures are often required for food processing, especially during the pelleting process to reduce the risk of microbial contaminants (Ebbensgaard et al., 2015). Based on this result, the intended incorporation of Lfcin (17–30) in the feed supplement is possible. Further, AMPs could be susceptible to proteolytic degradation by bacterial proteases together with gastrointestinal enzymes and serum proteases; however, in this study, Lfcin (17–30) was found to be protease-stable. Also, Lfcin (17–30) was found to be stable in the physiological concentration of cationic salts. Normally, the stability of AMP is highly dependent on amino acid residues such as tryptophan and arginine, which have previously shown to improve the antimicrobial activity of AMP under challenging salt conditions (Mohamed et al., 2016).
Cationic AMPs, like Lfcin (17–30), as employed in this study, may exhibit durable electrostatic interactions with the negatively charged phospholipids on the outer leaflet of the bacterial cell membrane and the least against eukaryotic cell membranes (Kumar et al., 2018). In our study, negligible hemolysis was caused by Lfcin (17–30) in sheep erythrocytes; nevertheless, the obtained findings need to be reasoned in the light of other cytotoxicity assays before ascertaining its utility as a potential therapeutic candidate. Additionally, Lfcin (17–30) reduced the viability of secondary cell lines (HEp-2 and RAW 264.7) in a concentration-dependent manner. Besides, typical cytotoxic effects such as detachment of confluent monolayer and cytoplasmic vacuolization were noticeably witnessed at higher concentrations (4 × MIC) of peptide, which might be attributed to its mechanism of action; however, the exact mechanism by which cytotoxicity varied was not understood absolutely (Vaucher et al., 2010). Furthermore, as part of the innate immune mechanism, AMPs may shape the composition of beneficial microbiota in maintaining intestinal homeostasis and assist in increasing the epithelial cells to avert bacterial uptake. Regardless of treatment with Lfcin (17–30), a non-significant effect was noticed on the tested beneficial lactobacilli, thereby proving its safety toward beneficial gut lactobacilli (Muniz et al., 2012).
The time-kill kinetic assay of Lfcin (17–30) exhibited a complete elimination of MDR-EAEC in 180 min, while similar inhibition was observed in meropenem after 60 min. Such complete bacterial inhibition by cationic peptides represents better treatment outcomes over antibiotics. Apart from the density of positively charged residues and an optimal balance between the hydrophilic and hydrophobic peptide surfaces, amphiphilic peptide confirmation is also responsible for the ability of AMPs to kill MDR-EAEC by disrupting bacterial membranes (Kumar et al., 2018). The membrane lipid-bilayer partition ability and optimum hydrophobicity might have led to the membrane damaging ability of Lfcin (17–30), as witnessed in flow cytometry, nitrocefin, and β-galactosidase activity (Marri et al., 1996; Epand et al., 2010). The antimicrobial activity of Lfcin (17–30) might be the result of its ability to stimulate the unfettered passage of small polar molecules through the cytoplasmic membrane. The time-kill kinetic assay in connection with the membrane permeabilization assay suggested the role of compromised and disrupted membrane integrity and pore formation as the main mechanism of cell killing by Lfcin (17–30).
High-throughput screening of antimicrobial agents require reliable and ergonomic in vivo laboratory models that could simulate humans. Though mammalian models are widely exploited, the budgetary, ethical, as well as logistic complications create hurdle in large-scale settings. These concerns in lab animal welfare warrants the possibility of utilizing invertebrate models in expediting the efficacy of antimicrobial agents for preliminary in vivo screening to reduce the candidate molecules for its further evaluation in ethical mammalian models. Having shared the innate immune response to the microbes (Wojda, 2017; Cutuli et al., 2019; Blasco et al., 2020), insects could be selected as suitable alternative models for extrapolating in vitro laboratory findings for probing the potential of Lfcin (17–30) against MDR-EAEC strains. The innate immune system of G. mellonella comprises a progressive cellular as well as humoral response compared to other existing invertebrate models. Although earlier reports regarding the in vivo efficacy of various antimicrobial agents against pathogens such as carbapenem-resistant Enterobacteriaceae, MRSA, P. aeruginosa, and K. pneumonia have explored the G. mellonella larvae model (Gibreel and Upton, 2013; Betts et al., 2014; Benthall et al., 2015; Cutuli et al., 2019), studies addressing the use of AMPs against such pathogens are scarcely documented in the literature. In the present study, dose-dependent mortality was reported in the larvae wherein the survival decreased with an increasing inoculum of MDR-EAEC. The determined LD50 dose (1 × 106 CFU/larvae) differed from an earlier study (1.11 × 104 CFU/larvae) (Jønsson et al., 2017), which could probably be due to varying levels in the virulence of EAEC strains.
The efficacy of AMPs against MDR-EAEC was assessed in comparison with an effective antibiotic, meropenem. The significant survival rate observed among meropenem-treated larvae, as reported in earlier studies (Hill et al., 2014; Benthall et al., 2015), might be due to the varied pharmacokinetic factors as compared to human, with a better antibiotic bioavailability in the larvae or with the synergistic effect of carbapenem with the natural AMPs present in the hemolymph. A significant increase observed in the survival rate of MDR-EAEC-infected larvae treated with Lfcin (17–30) in this study correlates with the earlier findings wherein different antimicrobials were tried against MRSA, A. baumanii, F. tularensis, and B. multivora (Ahmad et al., 2010; Brackman et al., 2011; Hill et al., 2014). The complete survival along with the lack of melanization of uninfected larval groups suggests the in vivo safety of Lfcin (17–30) tested and that the data need to be correlated with in vitro cytotoxicity assays. The result of the present study suggests that Lfcin (17–30) has an almost identical antibacterial potential to or a far better antibacterial potential than the antibiotic control used in this study; however, the exact AMP–host interaction remains uncertain.
The significant reduction in MDR-EAEC counts recovered from the infected larval hemolymph treated with Lfcin (17–30) over 24 and 48 h pi might be due to the bactericidal effect of AMP and/or metabolites produced during melanization (Hornsey and Wareham, 2011). The activation of the prophenoloxidase (PPO) cascade pathway in invertebrates is responsible for melanization wherein enhanced superoxide production in the larvae leads to the release of host AMPs, which in turn might be responsible for the clearance of pathogenic microbes (Wand et al., 2011). The bacterial clearance observed at 96 h pi could be achieved either by hemocyte-mediated aggregation or bacterial phagocytosis, which would have resulted in the secretion of larval AMPs leading to hemocyte degradation and larval melanization (Zhao et al., 2007; Desbois and Coote, 2011).
MDR-EAEC-stimulated hemocytes might successfully phagocytose the bacterial agents during the initial phase of infection at 6 to 18 h pi that has correlated fairly with the findings of MDR-EAEC enumeration wherein no significant difference in the MDR-EAEC counts could be noticed between the infection control group and the Lfcin (17–30)- and meropenem-treated infected larval groups. However, the MDR-EAEC, which evades phagocytosis and is located within the hemocytes, starts to replicate 24 h pi in the larvae, which is evident in the bacterial enumeration assay. A reduction in the circulating hemocytes noticed in all the larval groups at 72 and 96 h pi might be the result of cytotoxicity of MDR-EAEC strains on the larval cells (Amorim-Vaz et al., 2015). This depletion in hemocyte density could probably be ascribed to the infected hemocyte degradation and/or hemocyte sequestration in the nodules. However, upon any bacterial infection, the depletion of circulating hemocytes triggers the release of PPO components into the hemolymph, leading to the activation of phenoloxidase (PO). The activity of PO induces the formation of quinones and melanin that act as key components for defense reactions against the invading microbes (Barnoy et al., 2017).
Further, the results of melanization assay correlated fairly well with the hemocyte enumeration assay wherein with the decrease in hemocyte density, an increase in melanization intensity (12–24 h pi) was observed. The elevated melanization intensity observed in treatment groups infected with MDR-EAEC (12–24 h pi) could be interrelated with the activation of PO melanization cascade by stimulating hemocytes, which in turn might have led to the secretion of AMP within the fat body of insects, which is analogous to a liver of mammals (Barnoy et al., 2017). This would have activated specific signaling pathways leading to the generation of reactive oxygen species that might have destroyed the invading microbes (Desbois and Coote, 2011). It could also be inferred that Lfcin (17–30) improved the immunomodulation of larvae, as the melanization intensity was retained up to a later time point (96 h pi) in both uninfected and Lfcin (17–30)-treated infected larval groups. These observations suggest Lfcin (17–30) as a potential candidate for the development and re-purposing of drugs (Kumar et al., 2018).
Upon inoculation of MDR-EAEC, an elevated LDH production was observed in the inoculated larval group due to the increase in damaged and apoptotic host cells. The increasing trend in LDH production was observed in the meropenem-treated group wherein earlier studies reported the use of ampicillin as an effective therapeutic candidate for P. aeruginosa infection in G. mellonella larvae (Benthall et al., 2015). An initial increase in LDH production in all the inoculated groups could be attributed to the early host cell damage caused by pathogen before EAEC interacted with AMP. Besides, the minor trauma, while infecting the larvae and/or while administering the AMP could not be ignored, for the initial elevation of LDH. Furthermore, had Lfcin (17–30) been toxic to the larvae, the survival rate would have been reduced in all the groups treated with Lfcin (17–30), suggesting that Lfcin (17–30) was not cytotoxic to the larval cells.
Histopathological examination of the whole larvae was performed to divulge the sequence of events with host–pathogen interaction, pathogen migration, as well as recruitment of hemocytes (Pereira et al., 2015). It was observed that in the control infected group, the phagocytosis of MDR-EAEC mediated by hemocytes occurs promptly with the enrolment of hemocytes toward the heart region, where the hemocytes bind to the muscular architecture of heart and lead to phagocytosis of MDR-EAEC during 24 and 48 h pi. Later at 72 h pi, the hemocytes were evident in the heart and surrounding organs (pericardial cells and fat body), which, in turn, might have resulted in reducing the load of MDR-EAEC, melanization rate, as well as the circulating hemocyte density. Such sessile nature of larval hemocytes plays a vital role in controlling EAEC infection and pathogen recruitment.
Further, this fact reinforces the postulation that a coordinated interaction between the open circulatory system and the innate immune mechanism of the larvae is pivotal for operative immune responses (Lu et al., 2014; Pereira et al., 2015; Sheehan and Kavanagh, 2018). Nevertheless, a mild bacterial accumulation observed around the organelle without the pronounced aggregation of hemocytes or melanization observed at 24 and 48 h pi in the inoculated larval groups treated with Lfcin (17–30) and meropenem could serve as an indication of hemocyte-mediated phagocytosis that sequestered the pathogen within the internal organs and phagocytose them. None of the healthy larval controls exhibited appreciable histopathological changes with AMP treatment, which suggested the in vivo safety potential of Lfcin (17–30) in the G. mellonella larval model. The histopathology of the larval model was found closely interrelated with the MDR-EAEC burden, with estimation of various immune markers (melanization, hemocyte density), and with the in vitro time-kill kinetic assays. Additionally, it was concluded that those factors permitting the larval survival could be well pertinent to similar infections in humans (Sheehan and Kavanagh, 2018; Blasco et al., 2020).
Further, regarding microbial biofilm, nearly 80% of microbial infections in the living system involve biofilm formation (Batoni et al., 2016). Biofilm constitutes a syntrophic association of microbes enclosed within extracellular polymeric substance matrix that prevents microorganisms from adverse external influences, particularly antimicrobial agents (Lin et al., 2017; Petro et al., 2020). The adhesion of MDR-EAEC strains by MATS in this study was found to be comparatively higher to the tested monopolar solvent, chloroform, than to the apolar solvent, n-hexadecane, which bears almost identical van der Waal’s properties. Hence, the tested MDR-EAEC strains were deduced as strongly basic and weakly acidic. MDR-EAEC isolates employed in this study revealed significant hydrodynamic growth in DMEM supplemented with 0.45% D-glucose on the PS surface at 48 h. Besides, based on the grading criterion, moderate biofilm-forming ability (absorbance at 595 nm: 0.325–0.648) was observed for all the MDR-EAEC strains tested (Vijay et al., 2015).
The CV staining and confocal microscopy-based live/dead cell quantification revealed a significant biofilm biomass inhibition of MDR-EAEC strains by Lfcin (17–30) at 24 and 48 h. It could be speculated that for such initial inhibition of biofilm formation, Lfcin (17–30) could act as a coating agent covering either the surface of bacteria or biomaterials or both. The similar concentrations obtained for MBEC as well as MBC could be due to the moderate biofilm-forming ability of bacterial strains tested. Further, the elimination of preformed MDR-EAEC biofilms by peptides could be attained either by way of its direct antibacterial activity or detachment of live bacteria from the biofilms (Mataraci and Dosler, 2012; Silva et al., 2016; Zhang et al., 2016). However, the detachment and release of live bacterial cells from the preformed microbial biofilms by cationic peptides have not yet been documented.
The AMPs may be subjected to further clinical trials in suitable mammalian models before being translated as effective therapeutic candidates in either humans or animals. Many of the identified AMPs reported to have failed prior to or even during clinical trials. To date, only seven peptides were approved by the Food and Drug Administration (FDA) for therapeutic purpose. Though peptides are widely reported to treat skin infections, wounds, and pink eye as topical agents, some of them are also administered parentrally by oral route and direct injection (Chen and Lu, 2020). Moreover, conjugation of peptides with active molecules such as antibodies and/or nanoparticles, computational predictions, and high-throughput screening could be employed to make AMPs less toxic for target species while maintaining or improving their efficacy to eliminate pathogens, and further clinical studies in this regard is highly necessitated.
Conclusion
The antimicrobial and antibiofilm potential of Lfcin (17–30) was evaluated for the first time against the MDR-EAEC field isolates. Interestingly, Lfcin (17–30) was found to be stable (at high-end temperatures, proteases, and cationic salts), safe for eukaryotic cells, and beneficial to lactobacilli. Additionally, Lfcin (17–30) elicited a pronounced immunomodulatory effect and proved to be non-cytotoxic to the larval cells; overall, Lfcin (17–30) was found to be highly efficacious in the G. mellonella-EAEC infection model. Additionally, Lfcin (17–30) could inhibit the initial biofilm formation as well as eliminate the preformed biofilm of MDR-EAEC strains. Further studies using Lfcin (17–30) in mammalian models (mice/piglets) are warranted. Moreover, for the judicious application of Lfcin (17–30), the AMP can be coupled with targeted drug-delivery systems to prove its efficacy against MDR-EAEC in the infected mice or piglet models.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Author Contributions
DR, SM, and SB contributed to the conception and design of the study. JV and RP organized the experiments. MK and SR performed the statistical analysis. JV and MK wrote the first draft of the manuscript. JV, RP, MK, NK, and SR wrote sections of the manuscript. DR, SM, NK, and SB edited the manuscript. All authors contributed to manuscript revision and read and approved the submitted version.
Funding
This research work was supported by grants received from CAAST-ACLH (NAHEP/CAAST/2018-19) of ICAR-World Bank-funded National Agricultural Higher Education Project (NAHEP).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors thank the Director of the ICAR-Indian Veterinary Research Institute, Izatnagar, India, for providing facilities for the research. They are grateful to Dr. Chobi Debroy and Bhushan Jayarao, Pennsylvania State University, State College, PA, United States, for providing EAEC DNA. We thank Dr. Ajay Kumar, Scientist, and Dr. (Mrs.) Meeta Saxena, Senior Technical Officer, Division of Biochemistry, for their expertise in flow cytometry. They also thank Dr. Indira Devi, Director (Research), and Dr. Gavas Ragesh, Assistant Professor, KAU, Thrissur, Kerala, for providing G. mellonella larvae.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmicb.2020.575917/full#supplementary-material
References
Acosta-Smith, E., Viveros-Jiménez, K., Canizalez-Román, A., Reyes-Lopez, M., Bolscher, J. G., Nazmi, K., et al. (2018). Bovine lactoferrin and lactoferrin-derived peptides inhibit the growth of Vibrio cholerae and other Vibrio species. Front. Microbiol. 8:2633. doi: 10.3389/fmicb.2017.02633
Ahmad, S., Hunter, L., Qin, A., Mann, B. J., and van Hoek, M. L. (2010). Azithromycin effectiveness against intracellular infections of Francisella. BMC Microbiol. 10:123. doi: 10.1186/1471-2180-10-123
Amorim-Vaz, S., Delarze, E., Ischer, F., Sanglard, D., and Coste, A. T. (2015). Examining the virulence of Candida albicans transcription factor mutants using Galleria mellonella and mouse infection models. Front. Microbiol. 6:367. doi: 10.3389/fmicb.2015.00367
Barnoy, S., Gancz, H., Zhu, Y., Honnold, C. L., Zurawski, D. V., and Venkatesan, M. M. (2017). The Galleria mellonella larvae as an in vivo model for evaluation of Shigella virulence. Gut Microb. 8, 335–350. doi: 10.1080/19490976.2017.1293225
Batoni, G., Maisetta, G., and Esin, S. (2016). Antimicrobial peptides and their interaction with biofilms of medically relevant bacteria. Biochim. Biophys. Acta Biomembr. 1858, 1044–1060. doi: 10.1016/j.bbamem.2015.10.013
Benthall, G., Touzel, R. E., Hind, C. K., Titball, R. W., Sutton, J. M., Thomas, R. J., et al. (2015). Evaluation of antibiotic efficacy against infections caused by planktonic or biofilm cultures of Pseudomonas aeruginosa and Klebsiella pneumoniae in Galleria mellonella. Int. J. Antimicrob. Agents 46, 538–545. doi: 10.1016/j.ijantimicag.2015.07.014
Betts, J. W., Phee, L. M., Hornsey, M., Woodford, N., and Wareham, D. W. (2014). In vitro and in vivo activities of tigecycline-colistin combination therapies against carbapenem-resistant Enterobacteriaceae. Antimicrob. Agents Chemother. 58, 3541–3546. doi: 10.1128/AAC.02449-14
Blasco, L., Ambroa, A., Trastoy, R., Bleriot, I., Moscoso, M., Fernández-Garcia, L., et al. (2020). In vitro and in vivo efficacy of combinations of colistin and different endolysins against clinical strains of multi-drug resistant pathogens. Sci. Rep. 10, 1–12. doi: 10.1038/s41598-020-64145-7
Boll, E. J., Overballe-Petersen, S., Hasman, H., Roer, L., Ng, K., Scheutz, F., et al. (2020). Emergence of enteroaggregative Escherichia coli within the ST131 lineage as a cause of extraintestinal infections. mBio 11:e0353-20.
Brackman, G., Cos, P., Maes, L., Nelis, H. J., and Coenye, T. (2011). Quorum sensing inhibitors increase the susceptibility of bacterial biofilms to antibiotics in vitro and in vivo. Antimicrob. Agents Chemother. 55, 2655–2661. doi: 10.1128/AAC.00045-11
Canizalez-Roman, A., Velazquez-Roman, J., Valdez-Flores, M. A., Flores-Villaseñor, H., Vidal, J. E., Muro-Amador, S., et al. (2019). Detection of antimicrobial-resistance diarrheagenic Escherichia coli strains in surface water used to irrigate food products in the northwest of Mexico. Int. J. Food Microbiol. 304, 1–10. doi: 10.1016/j.ijfoodmicro.2019.05.017
Ceri, H., Olson, M. E., Stremick, C., Read, R. R., Morck, D., and Buret, A. (1999). The calgary biofilm device: new technology for rapid determination of antibiotic susceptibilities of bacterial biofilms. J. Clin. Microbiol. 37, 1771–1776. doi: 10.1128/jcm.37.6.1771-1776.1999
Chen, C. H., and Lu, T. K. (2020). Development and challenges of antimicrobial peptides for therapeutic applications. Antibiotics 9:24. doi: 10.3390/antibiotics9010024
Clinical, and Laboratory Standards Institute [CLSI] (2018). Performance Standards for Antimicrobial Susceptibility Testing, 28th Edn, Wayne, PE: CLSI.
Cravioto, A., Gross, R. J., Scotland, S. M., and Rowe, B. (1979). An adhesive factor found in strains of Escherichia coli belonging to the traditional infantile enteropathogenic serotypes. Curr. Microbiol. 3, 95–99. doi: 10.1007/bf02602439
Cutuli, M. A., Petronio, G., Vergalito, F., Magnifico, I., Pietrangelo, L., Venditti, N., et al. (2019). Galleria mellonella as a consolidated in vivo model hosts: new developments in antibacterial strategies and novel drug testing. Virulence 10, 527–541. doi: 10.1080/21505594.2019.1621649
Davies, J., and Davies, D. (2010). Origins and evolution of antibiotic resistance. Microbiol. Mol. Biol. Rev. 74, 417–433. doi: 10.1128/MMBR.00016-10
de la Fuente-Núñez, C., Reffuveille, F., Mansour, S. C., Reckseidler-Zenteno, S. L., Hernández, D., Brackman, G., et al. (2015). D-enantiomeric peptides that eradicate wild-type and multidrug-resistant biofilms and protect against lethal Pseudomonas aeruginosa infections. Chem. Biol. 22, 196–205. doi: 10.1016/j.chembiol.2015.01.002
de Zoysa, G. H., Cameron, A. J., Hegde, V. V., Raghothama, S., and Sarojini, V. (2015). Antimicrobial peptides with potential for biofilm eradication: synthesis and structure activity relationship studies of battacin peptides. J. Med. Chem. 58, 625–639. doi: 10.1021/jm501084q
Desbois, A. P., and Coote, P. J. (2011). Wax moth larva (Galleria mellonella): an in vivo model for assessing the efficacy of antistaphylococcal agents. J. Antimicrob. Chemoth. 66, 1785–1790. doi: 10.1093/jac/dkr198
Di Luca, M., Maccari, G., Maisetta, G., and Batoni, G. (2015). BaAMPs: the database of biofilm-active antimicrobial peptides. Biofouling 31, 193–199. doi: 10.1080/08927014.2015.1021340
Díaz-Godínez, C., González-Galindo, X., Meza-Menchaca, T., Bobes, R. J., de la Garza, M., León-Sicairos, N., et al. (2019). Synthetic bovine lactoferrin peptide Lfampin kills Entamoeba histolytica trophozoites by necrosis and resolves amoebic intracecal infection in mice. Biosci. Rep. 39:BSR20180850. doi: 10.1042/BSR20180850
Ebbensgaard, A., Mordhorst, H., Overgaard, M. T., Nielsen, C. G., Aarestrup, F. M., and Hansen, E. B. (2015). Comparative evaluation of the antimicrobial activity of different antimicrobial peptides against a range of pathogenic bacteria. PLoS One 10:e0144611. doi: 10.1371/journal.pone.0144611
Epand, R. F., Pollard, J. E., Wright, J. O., Savage, P. B., and Epand, R. M. (2010). Depolarization, bacterial membrane composition, and the antimicrobial action of ceragenins. Antimicrob. Agents Chemother. 54, 3708–3713. doi: 10.1128/AAC.00380-10
Ghaderpour, A., Ho, W. S., Chew, L. L., Bong, C. W., Chong, V. C., Thong, K. L., et al. (2015). Diverse and abundant multi-drug resistant E. coli in Matang mangrove estuaries Malaysia. Front. Microbiol. 6:977. doi: 10.3389/fmicb.2015.00977
Ghosh, C., Sarkar, P., Issa, R., and Haldar, J. (2019). Alternatives to conventional antibiotics in the era of antimicrobial resistance. Trends Microbiol. 27, 323–338. doi: 10.1016/j.tim.2018.12.010
Gibreel, T. M., and Upton, M. (2013). Synthetic epidermicin NI01 can protect Galleria mellonella larvae from infection with Staphylococcus aureus. J. Antimicrob. Chem. 68, 2269–2273. doi: 10.1093/jac/dkt195
Haney, E. F., Straus, S. K., and Hancock, R. E. (2019). Reassessing the host defense peptide landscape. Front. Chem. 7:43. doi: 10.3389/fchem.2019.00043
Haukland, H. H., and Vorland, L. H. (2001). Post-antibiotic effect of the antimicrobial peptide lactoferricin on Escherichia coli and Staphylococcus aureus. J. Antimicrob. Chemother. 48, 569–571. doi: 10.1093/jac/48.4.569
Hebbelstrup Jensen, B., Adler Sørensen, C., Hebbelstrup, R. R. S., Rejkjær Holm, D., Friis-Møller, A., Engberg, J., et al. (2018). Characterization of diarrheagenic enteroaggregative Escherichia coli in danish adults—antibiotic treatment does not reduce duration of diarrhea. Front. Cell Infect. Microbiol. 8:306. doi: 10.3389/fcimb.2018.00306
Hill, L., Veli, N., and Coote, P. J. (2014). Evaluation of Galleria mellonella larvae for measuring the efficacy and pharmacokinetics of antibiotic therapies against Pseudomonas aeruginosa infection. Int. J. Antimicrob. Agents 43, 254–261. doi: 10.1016/j.ijantimicag.2013.11.001
Hornsey, M., and Wareham, D. W. (2011). In vivo efficacy of glycopeptide-colistin combination therapies in a Galleria mellonella model of Acinetobacter baumannii infection. Antimicrob. Agents Chemother. 55, 3534–3537. doi: 10.1128/AAC.00230-11
Jønsson, R., Struve, C., Jenssen, H., and Krogfelt, K. A. (2017). The wax moth Galleria mellonella as a novel model system to study enteroaggregative Escherichia coli pathogenesis. Virulence 8, 1894–1899. doi: 10.1080/21505594.2016.1256537
Kolenda, R., Burdukiewicz, M., and Schierack, P. (2015). A systematic review and meta-analysis of the epidemiology of pathogenic Escherichia coli of calves and the role of calves as reservoirs for human pathogenic E. coli. Front. Cell Infect. Microbiol. 5:23. doi: 10.3389/fcimb.2015.00023
Kong, H., Hong, X., and Li, X. (2015). Current perspectivesin pathogenesis and antimicrobial resistance of enteroaggregative Escherichia coli. Microb. Pathog. 85, 44–49. doi: 10.1016/j.micpath.2015.06.002
Kumar, M., Dhaka, P., Vijay, D., Vergis, J., Mohan, V., Kumar, A., et al. (2016). Antimicrobial effects of Lactobacillus plantarum and Lactobacillus acidophilus against multidrug-resistant enteroaggregative Escherichia coli. Int. J. Antimicrob. Agents 48, 265–270. doi: 10.1016/j.ijantimicag.2016.05.014
Kumar, P., Kizhakkedathu, J., and Straus, S. (2018). Antimicrobial peptides: diversity, mechanism of action and strategies to improve the activity and biocompatibility in vivo. Biomolecules 8:4. doi: 10.3390/biom8010004
Lima, A. A., Medeiros, P. H., and Havt, A. (2018). Enteroaggregative Escherichia coli subclinical and clinical infections. Curr. Op. Infect. Dis. 31, 433–439. doi: 10.1097/QCO.0000000000000477
Lin, S., Yang, L., Chen, G., Li, B., Chen, D., Li, L., et al. (2017). Pathogenic features and characteristics of food borne pathogens biofilm: biomass, viability and matrix. Microb. Pathog. 111, 285–291. doi: 10.1016/j.micpath.2017.08.005
Lu, A., Zhang, Q., Zhang, J., Yang, B., Wu, K., Xie, W., et al. (2014). Insect prophenoloxidase: the view beyond immunity. Front. Physiol. 5:252. doi: 10.3389/fcimb.2015.00252
Marri, L., Dallai, R., and Marchini, D. (1996). The novel antibacterial peptide ceratotoxin A alters permeability of the inner and outer membrane of Escherichia coli K-12. Curr. Microbiol. 33, 40–43. doi: 10.1007/s002849900071
Mataraci, E., and Dosler, S. (2012). In vitro activities of antibiotics and antimicrobial cationic peptides alone and in combination against methicillin-resistant Staphylococcus aureus biofilms. Antimicrob. Agents Chemother. 56, 6366–6371. doi: 10.1128/AAC.01180-1
McCloskey, A. P., Lee, M., Megaw, J., McEvoy, J., Coulter, S. M., Pentlavalli, S., et al. (2019). Investigating the in vivo antimicrobial activity of a self-assembling peptide hydrogel using a Galleria mellonella infection model. ACS Omega 4, 2584–2589. doi: 10.1021/acsomega.8b03578
Miles, A. A., Misra, S. S., and Irwin, J. O. (1938). The estimation of the bactericidal power of the blood. Epidemiol. Infect. 38, 732–749. doi: 10.1017/S002217240001158X
Mohamed, M. F., Abdelkhalek, A., and Seleem, M. N. (2016). Evaluation of short synthetic antimicrobial peptides for treatment of drug-resistant and intracellular Staphylococcus aureus. Sci. Rep. 6, 1–14. doi: 10.1038/srep29707
Molnar, A. (2019). Antimicrobial resistance awareness and games. Trends Microbiol. 27, 1–3. doi: 10.1016/j.tim.2018.09.007
Mookherjee, N., Anderson, M. A., Haagsman, H. P., and Davidson, D. J. (2020). Antimicrobial host defence peptides: functions and clinical potential. Nat. Rev. Drug Discov. 19, 311–332. doi: 10.1038/s41573-019-0058-8
Morgan, J. K., Ortiz, J. A., and Riordan, J. T. (2014). The role for TolA in enterohemorrhagic Escherichia coli pathogenesis and virulence gene transcription. Microb. Pathog. 77, 42–52. doi: 10.1016/j.micpath.2014.10.010
Muniz, L. R., Knosp, C., and Yeretssian, G. (2012). Intestinal antimicrobial peptides during homeostasis, infection, and disease. Front. Immunol. 3:310. doi: 10.3389/fimmu.2012.00310
Oundo, J. O., Kariuki, S. M., Boga, H. I., Muli, F. W., and Iijima, Y. (2008). High incidence of enteroaggregative Escherichia coli among food handlers in three areas of Kenya: a possible transmission route of travelers’ diarrhea. J. Travel Med. 15, 31–38. doi: 10.1111/j.1708-8305.2007.00174.x
Perdoni, F., Falleni, M., Tosi, D., Cirasola, D., Romagnoli, S., Braidotti, P., et al. (2014). A histological procedure to study fungal infection in the wax moth Galleria mellonella. Eur. J. Histochem. 58:2428. doi: 10.4081/ejh.2014.2428
Pereira, M. F., Rossi, C. C., de Queiroz, M. V., Martins, G. F., Isaac, C., Bosse, J. T., et al. (2015). Galleria mellonella is an effective model to study Actinobacillus pleuropneumoniae infection. Microbiol. 161, 387–400. doi: 10.1099/mic.0.083923-0
Petro, C. D., Duncan, J. K., Seldina, Y. I., Allué-Guardia, A., Eppinger, M., Riddle, M. S., et al. (2020). Genetic and virulence profiles of Enteroaggregative Escherichia coli (EAEC) isolated from deployed military personnel (DMP) with travelers’ diarrhea. Front. Cell Infect. Microbiol. 10:200. doi: 10.3389/fcimb.2020.00200
Philipson, C. W., Bassaganya-Riera, J., and Hontecillas, R. (2013). Animal models of enteroaggregative Escherichia coli infection. Gut Microb. 4, 281–291. doi: 10.4161/gmic.24826
Pollini, S., Brunetti, J., Sennati, S., Rossolini, G. M., Bracci, L., Pini, A., et al. (2017). Synergistic activity profile of an antimicrobial peptide against multidrugresistant and extensively drug-resistant strains of Gram-negative bacterial pathogens. J. Pept. Sci. 23, 329–333. doi: 10.1002/psc.2978
Reyes-Cortes, R., Acosta-Smith, E., Mondragón-Flores, R., Nazmi, K., Bolscher, J. G., Canizalez-Roman, A., et al. (2016). Antibacterial and cell penetrating effects of LFcin17-30, LFampin265-284, and LF chimera on enteroaggregative Escherichia coli. Int. J. Biochem. Cell Biol. 95, 76–81. doi: 10.1139/bcb-2016-0088
Schindelin, J., Arganda-Carreras, I., Frise, E., Kaynig, V., Longair, M., Pietzsch, T., et al. (2012). Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682. doi: 10.1038/nmeth.2019
Sheehan, G., and Kavanagh, K. (2018). Analysis of the early cellular and humoral responses of Galleria mellonella larvae to infection by Candida albicans. Virulence 9, 163–172. doi: 10.1080/21505594.2017.1370174
Sijbrandij, T., Ligtenberg, A. J., Nazmi, K., Veerman, E. C., Bolscher, J. G., and Bikker, F. J. (2017). Effects of lactoferrin derived peptides on simulants of biological warfare agents. World J. Microbiol. Biotechnol. 33:3. doi: 10.1007/s11274-016-2171-8
Silva, O. N., Alves, E. S. F., de la Fuente-Nuñez, C., Ribiero, S. M., Mandal, S. M., Gaspar, D., et al. (2016). Structural studies of a lipid-binding peptide from tunicate hemocytes with anti-biofilm activity. Sci. Rep. 6, 1–14. doi: 10.1038/srep27128
Silva, T., Moreira, A. C., Nazmi, K., Moniz, T., Vale, N., Rangel, M., et al. (2017). Lactoferricin peptides increase macrophages’ capacity to kill Mycobacterium avium. mSphere 2, e301–e317. doi: 10.1128/mSphere.00301-17
van der Kraan, M. I., van Marle, J., Nazmi, K., Groenink, J., van’t Hof, W., Veerman, E. C., et al. (2005). Ultrastructural effects of antimicrobial peptides from bovine lactoferrin on the membranes of Candida albicans and Escherichia coli. Peptides 26, 1537–1542. doi: 10.1016/j.peptides.2005.02.011
Vaucher, R. A., De da Motta, S. A., and Brandelli, A. (2010). Evaluation of the in vitro cytotoxicity of the antimicrobial peptide P34. Cell Biol. Int. 34, 317–323. doi: 10.1042/CBI20090025
Vergis, J., Malik, S. V. S., Pathak, R., Kumar, M., Ramanjaneya, S., Kurkure, N. V., et al. (2019). Antimicrobial efficacy of Indolicidin against multi-drug resistant enteroaggregative Escherichia coli in a Galleria mellonella model. Front. Microbiol. 10:2723. doi: 10.3389/fmicb.2019.02723
Vijay, D., Dhaka, P., Vergis, J., Negi, M., Mohan, V., Kumar, M., et al. (2015). Characterization and biofilm forming ability of diarrhoeagenic enteroaggregative Escherichia coli isolates recovered from human infants and young animals. Comp. Immunol. Microbiol. Infect. Dis. 38, 21–31. doi: 10.1016/j.cimid.2014.11.004
Wakimoto, N., Nishi, J., Sheikh, J., Nataro, J. P., Sarantuya, J. A. V., Iwashita, M., et al. (2004). Quantitative biofilm assay using a microtiter plate to screen for enteroaggregative Escherichia coli. Am. J. Trop. Med. Hyg. 71, 687–690. doi: 10.4269/ajtmh.2004.71.687
Wand, M. E., McCowen, J. W., Nugent, P. G., and Sutton, J. M. (2013). Complex interactions of Klebsiella pneumoniae with the host immune system in a Galleria mellonella infection model. J. Med. Microbiol. 62, 1790–1798. doi: 10.1099/jmm.0.063032-0
Wand, M. E., Müller, C. M., Titball, R. W., and Michell, S. L. (2011). Macrophage and Galleria mellonella infection models reflect the virulence of naturally occurring isolates of B. pseudomallei, B. thailandensis and B. oklahomensis. BMC Microbiol. 11:11. doi: 10.1186/1471-2180-11-11
Wojda, I. (2017). Immunity of the greater wax moth Galleria mellonella. Ins. Sci. 24, 342–357. doi: 10.1111/1744-7917.12325
Xu, G., Xiong, W., Hu, Q., Zuo, P., Shao, B., Lan, F., et al. (2010). Lactoferrin-derived peptides and Lactoferricin chimera inhibit virulence factor production and biofilm formation in Pseudomonas aeruginosa. J. Appl. Microbiol. 109, 1311–1318. doi: 10.1111/j.1365-2672.2010.04751.x
Zhang, T., Wang, Z., Hancock, R. E. W., de la Fuente-Nuñez, C., and Haapasalo, M. (2016). Treatment of oral biofilms by a D-enantiomeric peptide. PLoS One 11:e0166997. doi: 10.1371/journal.pone.0166997
Keywords: antimicrobial peptide, biofilm, confocal microscopy, enteroaggregative E. coli, Galleria mellonella, lactoferricin (17–30)
Citation: Vergis J, Malik SS, Pathak R, Kumar M, Ramanjaneya S, Kurkure NV, Barbuddhe SB and Rawool DB (2020) Exploiting Lactoferricin (17–30) as a Potential Antimicrobial and Antibiofilm Candidate Against Multi-Drug-Resistant Enteroaggregative Escherichia coli. Front. Microbiol. 11:575917. doi: 10.3389/fmicb.2020.575917
Received: 24 June 2020; Accepted: 17 August 2020;
Published: 18 September 2020.
Edited by:
Elisabeth Grohmann, Beuth Hochschule für Technik Berlin, GermanyReviewed by:
Cesar de la Fuente-Nunez, University of Pennsylvania, United StatesDébora Coraça-Huber, Innsbruck Medical University, Austria
Copyright © 2020 Vergis, Malik, Pathak, Kumar, Ramanjaneya, Kurkure, Barbuddhe and Rawool. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Deepak Bhiwa Rawool, ZGVlcGFrLnJhd29vbEB5YWhvby5jb20=
†Present address: Jess Vergis, Department of Veterinary Public Health, College of Veterinary and Animal Sciences, KVASU, Pookode, India
 Jess Vergis
Jess Vergis Satyaveer Singh Malik
Satyaveer Singh Malik Richa Pathak
Richa Pathak Manesh Kumar
Manesh Kumar Sunitha Ramanjaneya
Sunitha Ramanjaneya Nitin Vasantrao Kurkure
Nitin Vasantrao Kurkure Sukhadeo Baliram Barbuddhe
Sukhadeo Baliram Barbuddhe Deepak Bhiwa Rawool
Deepak Bhiwa Rawool