Abstract
Breast cancer is the most prevalent malignancy among women worldwide and is a major contributor to cancer-related mortality. The tumor microenvironment (TME), composed of tumor cells, immune infiltrates, fibroblasts, and vascular components, is critically involved in tumor initiation, metastatic progression, and therapeutic response. In recent years, therapies targeting the TME have undergone rapid advancements, with the objective of enhancing antitumor immunity. Concurrently, mounting evidence underscores the pivotal role of the gut microbiota and its metabolites in modulating host immunity, influencing metabolic homeostasis, inflammation, and immune equilibrium. The composition and diversity of the gut microbiome influence breast cancer progression and patients’ responses to immunotherapy. Therefore, modulating the gut microbiota is a promising strategy to enhance the clinical outcomes of TME-targeted immunotherapies. In this review, we discuss the influence of gut microbiota and its derived metabolites on breast cancer progression and immunotherapy prognosis and explore potential strategies to optimize immunotherapy through gut microbiota modulation.
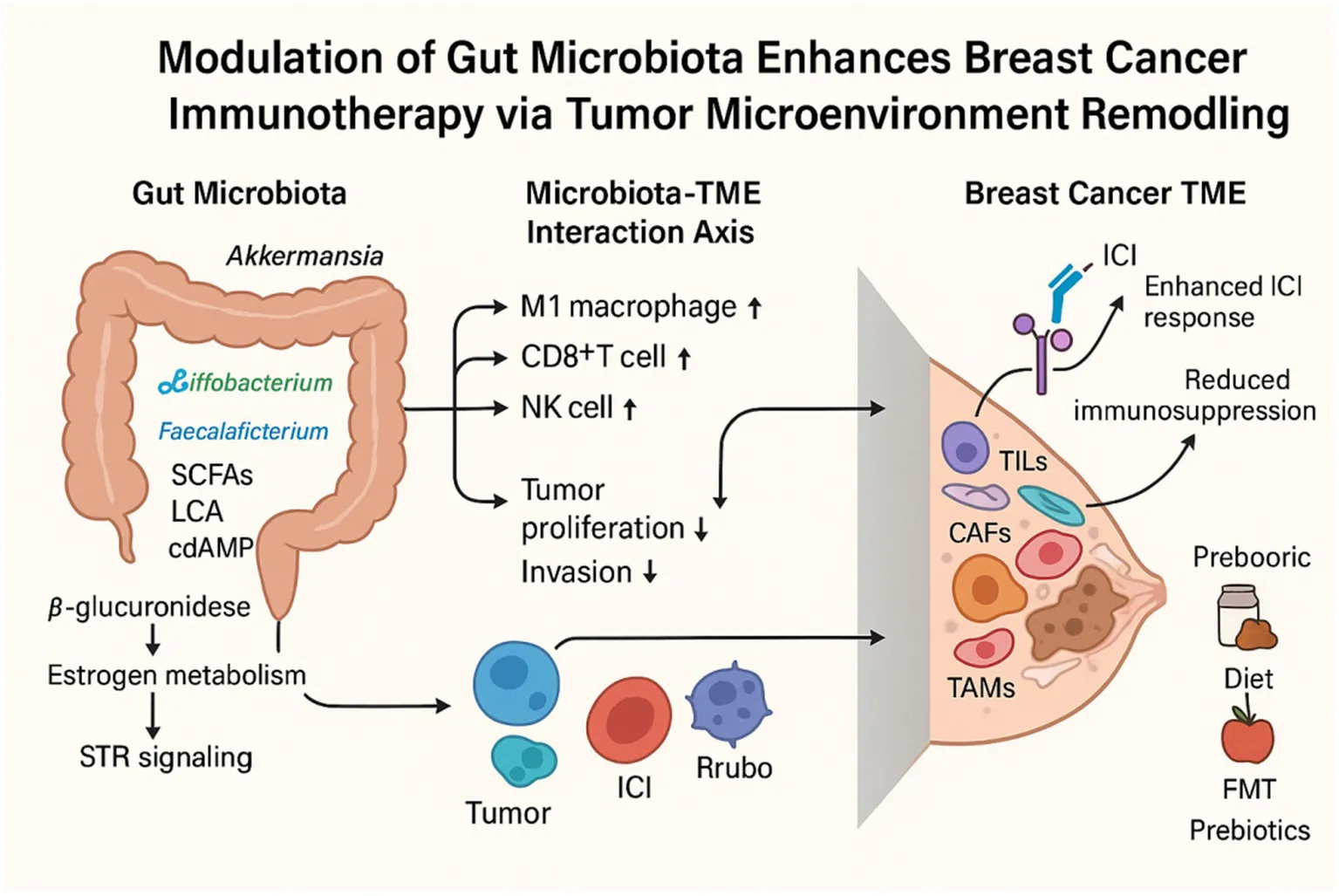
1 Introduction
Breast cancer represents a significant global health challenge, impacting millions of individuals each year. Epidemiological data indicate consistently high incidence and mortality rates worldwide, making it the most prevalent malignancy among women and a leading cause of cancer-related deaths (Sung et al., 2021). The management of breast cancer imposes substantial socioeconomic and healthcare burdens globally, with diagnosis, treatment, and long-term care placing significant strain on healthcare infrastructures and households alike. There is an urgent need for innovative diagnostic and therapeutic approaches to improve therapeutic efficacy and prolong patient survival (Sung et al., 2021). These strategies must integrate innovations in early detection, personalized treatment regimens, and comprehensive supportive care services to address the diverse needs of breast cancer patients worldwide.
The tumor microenvironment (TME) is a complex and dynamic ecosystem composed of malignant cells, adjacent non-malignant cells, and the extracellular matrix (ECM). It is critically involved in tumor development, metastasis, and therapeutic responses (Jin and Jin, 2020; Deepak et al., 2020). The TME encompasses cancer cells, stromal cells, vasculature, nerve fibers, the ECM, and associated acellular components, which collectively engage in complex interactions with malignant cells. These interactions regulate key oncogenic processes, including tumor proliferation, angiogenesis, evasion of apoptosis, immune suppression, and escape from immune surveillance (Hanahan and Weinberg, 2011). Given its critical role in tumor progression and therapeutic modulation, therapeutic strategies targeting the TME have witnessed significant advancements in recent years. These approaches primarily aim to modulate tumor-associated macrophages (TAMs), dendritic cells (DCs), T cells, tumor vasculature, the ECM, and cancer-associated fibroblasts (CAFs) (Bejarano et al., 2021).
The gut microbiome produces a vast array of small molecules and metabolites that are essential for various physiological processes, including metabolism, inflammation, immune regulation, and neural function (Jugder et al., 2021; Zheng et al., 2020; Erny et al., 2021; Morais et al., 2021). Given its capacity to modulate both local and systemic immune responses, there is growing interest in the influence of gut microbiota on the immune system in breast cancer and its impact on responses to immune checkpoint inhibitors (ICIs) (He et al., 2021; Fehervari, 2021; McQuade et al., 2019). Dysbiosis of the gut microbiota has also been implicated as a key contributor to cancer progression and variability in responses to ICI therapy (Mallott and Amato, 2021). Consequently, the therapeutic potential of targeting the gut microbiome as a modulator of the TME is increasingly recognized.
In this review, we provide an overview of recent advances in understanding the gut microbiota’s role in targeting the tumor microenvironment and modulating immune responses. Our aim is to elucidate the intricate relationship between the composition and function of the gut microbiome and breast cancer progression, metastasis, and therapeutic response, with an emphasis on their dynamic interplay. This understanding offers new insights into the development of microbiota-based targeted interventions to improve breast cancer treatment outcomes.
2 TME and immunotherapy in breast cancer
Clinically, the treatment and prognosis of breast cancer are largely determined by its molecular subtype, which is defined by the expression profiles of human epidermal growth factor receptor 2 (HER2) and hormone receptors (Prat et al., 2010; Perou et al., 2000). In recent years, therapeutic strategies such as ICI have shown considerable promise by modulating the TME instead of directly attacking tumor cells. A comprehensive understanding of the complex cellular and molecular interactions within the TME is essential for optimizing the efficacy of immunotherapeutic interventions.
2.1 Composition of the TME in breast cancer
TME is a highly heterogeneous and dynamically evolving milieu comprising immune cells, fibroblasts, vascular endothelial cells, and tumor-associated stroma. Different cell types within the tumor microenvironment (TME) engage in intricate crosstalk through the secretion and expression of factors that either drive tumor proliferation and immune evasion or, alternatively, support adaptive immune responses to suppress tumor growth, thereby modulating disease progression (Swanton et al., 2024).
2.1.1 Macrophages
Tumor-associated macrophages (TAMs) play a pivotal role in the breast cancer TME, comprising up to 25% of immune cells in untreated triple-negative breast cancer (TNBC) (Keren et al., 2018). Within tumors, TAMs do not fit neatly into the classical dichotomy of pro-inflammatory M1 and immunosuppressive M2 macrophages; instead, they often exhibit a spectrum of polarization states, exhibiting concurrent immune-activating and immune-suppressive properties (Cassetta et al., 2019). Notably, FOLR2+ macrophages are positively associated with CD8+ T cell-mediated antitumor activity (Nalio Ramos et al., 2022). In contrast, CADM1+TREM2+ monocyte-derived macrophages become dominant as tumors progress and are strongly associated with poor prognosis in TNBC (Nalio Ramos et al., 2022). Therapeutic strategies aimed at targeting these cellular subsets have emerged as a focal point of intensive research.
2.1.2 Tumor-infiltrating lymphocytes
Tumor-infiltrating lymphocytes (TILs) within the breast cancer TME consist primarily of CD3+ T cells. The abundance of TILs varies significantly across breast cancer subtypes, with triple-negative breast cancer (TNBC) demonstrating the greatest TIL density at the time of diagnosis. This phenomenon correlates strongly with TNBC’s elevated tumor mutational burden, high TP53 mutation frequency, and genomic instability (Luen et al., 2016). These tissue-resident memory-like (TRM-like) cells are marked by dual expression of CD103 and CD69 and often exhibit elevated levels of PD-1, CTLA-4, TIM-3, and LAG-3, indicating a dual role in cytotoxic activity and partial functional exhaustion (Savas et al., 2018). Among CD4+ T cells, dysfunctional TCF1− subsets are found alongside helper T cells, such as Th1 and follicular helper T cells, as well as regulatory T cells (Tregs). Although Tregs constitute a relatively minor fraction, their immunosuppressive function is strongly associated with poor clinical prognosis (Keren et al., 2018; Wu et al., 2021).
In the breast cancer TME, tumor-infiltrating B lymphocytes primarily localize within clustered aggregates and are frequently associated with the formation of tertiary lymphoid structures (TLS), which resemble secondary lymphoid organs (Danenberg et al., 2022). Tumor-infiltrating B cells are more prevalent in TNBC and HER2+ breast cancer, and their presence is strongly associated with enhanced immunotherapy efficacy and improved clinical outcomes (Pal et al., 2021).
2.1.3 Non-immune cell
As a major component of the tumor stroma, CAFs exhibit substantial heterogeneity and play a crucial role in tumor progression by remodeling the extracellular matrix, facilitating angiogenesis, and secreting cytokines that drive cancer cell proliferation, migration, and invasion (Costa et al., 2018; Kieffer et al., 2020; Cords et al., 2023; Wu et al., 2020). The CAF-S1 subset promotes Treg survival and suppresses CD8+ T cell activity, whereas CAF-S4 is associated with an immune-activating phenotype (Costa et al., 2018). Vascular endothelial cells regulate the recruitment and infiltration of immune cells; high endothelial venules (HEVs) enhance antitumor immunity, while FASL expression may induce apoptosis of CD8+ T cells (Motz et al., 2014; Goveia et al., 2020; Geldhof et al., 2022). Although often underrepresented in single-cell analyses, adipocytes exert immunomodulatory effects in the TME by secreting bioactive mediators.
2.2 Targeting TME
2.2.1 Immune checkpoint blockade
Recent advances have achieved considerable success in immune checkpoint blockade (ICB) therapy targeting PD-1 and PD-L1 in breast cancer. The KEYNOTE trials have shown that incorporating immune checkpoint inhibitors into chemotherapy regimens markedly enhances recurrence-free survival (Cortes et al., 2022; Schmid et al., 2020). However, the heterogeneity of PD-L1 expression and interpatient variability in therapeutic response constrain the generalizability of this strategy and highlight the pressing need for reliable predictive biomarkers.
2.2.2 Targeting TAMs
Recent studies have investigated approaches to modulate TAMs through key immunosuppressive pathways, such as inhibiting TREM2 or CSF1R, to enhance antitumor immune responses (Haishan Lin et al., 2008; Timperi et al., 2022). However, a key challenge is the selective depletion or reprogramming of tumor-promoting TAMs while preserving the essential functions of resident macrophages in normal tissues. Overcoming these obstacles is a central focus of ongoing research efforts.
2.2.3 Targeting CAFs
Emerging therapeutic strategies aim to selectively target tumor-promoting CAFs by exploiting fibroblast activation protein (FAP) or other specific markers to alleviate tumor-induced immunosuppression within the TME (Ao et al., 2015). However, the inherent heterogeneity of CAFs and their overlapping characteristics with normal fibroblasts present major obstacles.
2.2.4 Targeting angiogenesis
Therapeutic strategies targeting tumor vasculature are primarily directed at normalizing dysfunctional tumor vasculature through agents such as anti-vascular endothelial growth factor (VEGF) therapies. This approach aims to facilitate immune cell infiltration and optimize drug delivery within the TME. However, the therapeutic window for vascular normalization is relatively narrow, and responses to anti-angiogenic treatments exhibit considerable interpatient variability.
3 TME of breast cancer and gut microbiome
Approximately 1014 microorganisms inhabit the gastrointestinal tract, playing essential roles in host physiology and maintaining dynamic interactions with the host (Domenica Rea et al., 2018). Breast cancer development and progression are shaped by a multitude of factors, including age, hormonal levels, menopausal status, inflammation, and immune regulation (Wu et al., 2022). The gut microbiota plays a pivotal role in the crosstalk between these risk factors and breast cancer, influencing both tumor staging and patient prognosis (Sommer et al., 2017). Accumulating evidence indicates that the gut microbiome modulates tumor cells and their microenvironment through multiple mechanisms, including immune system modulation, alterations in estrogen metabolism, and the production of bacterial metabolites. These findings underscore the microbiota’s role in breast cancer pathophysiology and highlight its potential as a therapeutic target.
3.1 Estrogen metabolism and gut microbiota in breast cancer
Breast cancer, particularly hormone receptor-positive (HR+) subtypes, is highly dependent on estrogen signaling (Russo, 2006). Estrogen is metabolized in the liver by the cytochrome P450 enzyme system into various metabolites, which are then excreted into the intestine through bile secretion. Within the gut, specific microbial taxa—such as Clostridia, Ruminococcaceae families, and Escherichia/Shigella species—produce β-glucuronidase, an enzyme responsible for deconjugating estrogen metabolites and restoring them to their biologically active free form. These free estrogens are then reabsorbed into circulation through the portal circulation, reaching multiple organs, including the breast (Zhu and Conney, 1998; Cullin et al., 2021). This enterohepatic recycling process, known as the estrobolome, is essential in regulating systemic free estrogen levels, thereby influencing breast cancer risk and progression.
The ratio of estrogen metabolites to free estrogen exhibits an inverse correlation with breast cancer risk (Falk et al., 2013), and the gut microbiota plays a crucial role in regulating systemic estrogen levels. Gut microbial dysbiosis can lead to increased circulating estrogen levels, thereby elevating the risk of postmenopausal breast cancer. The deconjugation of estrogen in circulation is largely dependent on the composition and diversity of the gut microbiome, underscoring its mechanistic role in estrogen metabolism (Fuhrman et al., 2012). Long-term antibiotic use may disrupt the normal microbial balance, resulting in excessive β-glucuronidase activity. This, in turn, increases free estrogen concentrations, thereby promoting breast cancer progression (Iizumi et al., 2017). Additionally, increased adipose tissue, as seen in obesity, may also modulate estrogen levels via similar mechanisms, further heightening breast cancer risk (Keum et al., 2015). Although existing studies have established a link between estrogen metabolism and gut microbiota, the specific microbial species that play dominant roles in estrogen processing, as well as their intricate interactions, are not yet fully elucidated. The development of clinical strategies aimed at modulating the gut microbiota to regulate estrogen levels and ultimately prevent or treat breast cancer remains in its nascent stages.
3.2 Impact of gut microbiota on immunity
In recent years, numerous studies have revealed a complex and bidirectional regulatory interplay between the gut microbiota and the host immune system, a dynamic interaction that plays a crucial role in the initiation and progression of breast cancer. When the gut microbiota maintains homeostasis, it contributes to tissue equilibrium and suppresses tumorigenesis by modulating both innate and adaptive immune responses (Dethlefsen et al., 2007). Specifically, dysbiosis triggers the upregulation of inflammatory mediators such as IL-6 and TNF-α, creating a pro-inflammatory milieu conducive to breast cancer cell proliferation and invasion (Willing et al., 2011). Moreover, a reduction in gut microbial diversity is strongly associated with reduced survival outcomes in breast cancer patients, often accompanied by alterations in the immune cell compartment, further underscoring the microbiota’s role in shaping tumor immunology (Bernardo et al., 2023).
In mouse models of hormone receptor-positive (HR+) breast cancer, antibiotic-induced dysbiosis induces an expansion of tumor-infiltrating myeloid cells, particularly those expressing high levels of arginase-1, while also significantly promoting the infiltration of M2-like macrophages (Buchta Rosean et al., 2019). As potent immunosuppressive cells, M2-like macrophages are closely linked to immune suppression and adverse clinical outcomes (Gwak et al., 2015). Beyond macrophages, the gut microbiota plays a pivotal role in modulating T cell recruitment and activation. For instance, Fusobacterium nucleatum can translocate to breast tissue via the bloodstream and selectively colonize tumor sites through Fap2-dependent interactions with cancer cells. This targeted colonization inhibits the infiltration of TILs and facilitates tumor progression and metastatic dissemination (Parhi et al., 2020). Nevertheless, the precise alterations in immune cell subpopulations across different breast cancer molecular subtypes and their causal relationship with microbiota regulation are not yet well characterized. A major challenge for future therapeutic strategies is identifying optimal strategies for microbiota modulation to restore or enhance antitumor immunity.
3.3 The influence of gut metabolites on breast cancer
Microbial metabolites systemically distributed within the host play a pivotal role in breast cancer progression. These metabolites are either directly produced through microbial fermentation—such as short-chain fatty acids (SCFAs), lactate, and pyruvate—or result from microbial biotransformation of host-derived molecules, including secondary bile acids, aromatic hydrocarbon metabolites, and redox-modified steroids (Zhang et al., 2021). These bioactive molecules modulate host gene expression and intracellular signaling cascades. Studies have shown that early-stage breast cancer patients exhibit reduced serum levels of lithocholic acid (LCA), implicating LCA deficiency as a potential contributor to tumor progression (Miko et al., 2018). Secondary bile acids, particularly LCA, produced by Clostridia spp., have demonstrated inhibitory effects on breast cancer cell proliferation by activating the G protein-coupled receptor TGR5. Additionally, LCA perturbs intracellular redox equilibrium by altering the balance of antioxidant and pro-oxidant enzymes (Sampsell et al., 2020). SCFAs—such as butyrate, propionate, and acetate—constitute major microbial fermentation products of dietary fiber, primarily generated by specific gut bacterial taxa, including Eubacterium rectale, Clostridium leptum, and Faecalibacterium prausnitzii (Jaye et al., 2022). SCFAs are essential for regulating cellular energy metabolism, inducing cell cycle arrest, and promoting apoptosis in breast cancer cells. Notably, butyrate has been shown to independently suppress breast cancer cell growth and, when combined with anti-HER2 monoclonal antibodies such as trastuzumab, synergistically enhances therapeutic efficacy (Garmpis et al., 2017; Chen et al., 2011). While no definitive tumor-promoting microbial metabolites have been identified in breast cancer, emerging evidence suggests that microbial-derived metabolites play dual roles in tumor pathophysiology. On one hand, they directly modulate cancer cell proliferation and apoptosis. Certain microbial metabolites, such as trimethylamine N-oxide (TMAO), have been recognized for their antitumor properties. TMAO enhances CD8+ T cell–mediated antitumor immunity and induces ferroptosis in tumor cells, thereby contributing to tumor suppression (Dovrolis et al., 2024). On the other, they shape the local immune microenvironment, indirectly influencing tumor progression. For example, lactate accumulation within the tumor microenvironment has been strongly linked to immunosuppressive effects. Acting as a signaling molecule, lactate signals through G protein–coupled receptors (GPRs) via both autocrine and paracrine pathways, leading to enhanced angiogenesis and immune evasion in the tumor microenvironment (Zhou et al., 2024). A key challenge involves accurately characterizing and quantifying these metabolites in breast cancer patients, elucidating their spatial and temporal distribution. Furthermore, the mechanisms by which interspecies microbial interactions shape the metabolic landscape remain largely undefined. Translating these molecular insights into clinical interventions—such as targeted probiotics, prebiotics, or metabolic modulators—necessitates rigorous validation in prospective clinical studies.
3.4 The influence of tissue-resident microbiota within the breast tumor microenvironment
Emerging evidence indicates that tumor-resident microbiota (TRM) within breast cancer tissues play a crucial role in modulating the efficacy of immunotherapy (Ma et al., 2021). TRM may colonize tumors through migration from adjacent tissues or by translocating across compromised vasculature during systemic circulation (Fu et al., 2022). The immunosuppressive nature of the tumor microenvironment (TME) creates a permissive niche for these microbial communities (Nejman et al., 2020). Through interactions with pattern recognition receptors such as Toll-like receptors (TLRs) and G protein–coupled receptors (GPCRs), TRM regulate T cell activation and differentiation, thereby influencing therapeutic outcomes. Notably, bacterial eradication using Au–Ag₂Se nanoassemblies, in conjunction with multimodal antitumor therapy, has been shown to restore immune surveillance and potentiate antitumor immunity, resulting in recurrence-free survival extending beyond 700 days (Wang et al., 2024). However, studies targeting TRM in combination with immunotherapy remain scarce, and the precise impact of TRM on breast cancer immunotherapeutic efficacy warrants further investigation. Elucidating the composition, functional roles, and immunomodulatory mechanisms of TRM may inform the development of novel therapeutic strategies aimed at refining and enhancing breast cancer immunotherapy.
4 The impact of gut microbiota on immunotherapy in breast cancer
The variability in breast cancer patients’ responses to immunotherapy has posed a substantial challenge to the broad implementation of these therapies. Given the profound impact of the gut microbiota on breast cancer prognosis, it represents a key factor in personalizing immunotherapeutic strategies (Nandi et al., 2023). Emerging evidence suggests that the gut microbiome plays a pivotal role in modulating immunotherapeutic efficacy by engaging in complex interactions with the host immune system, modulating systemic inflammation, and shaping immune cell function. High-throughput sequencing and metabolomic analyses have revealed pronounced gut microbiota dysbiosis in breast cancer patients, a phenomenon closely linked to alterations in the distribution and activity of immune cells within the tumor microenvironment. These findings offer a strong scientific basis for advancing microbiota-targeted immunotherapeutic approaches in breast cancer. Table 1 summarizes the known microbial species implicated in modulating the response to immunotherapy in breast cancer.
Table 1
| Microbial species | Impact |
|---|---|
| Faecalibacterium prausnitzii | Produces butyrate with anti-inflammatory properties, potentially enhancing the efficacy of ICIs |
| Akkermansia muciniphila | Associated with favorable response to ICI therapy, potentially enhancing treatment efficacy by augmenting antitumor immune responses |
| Bifidobacterium longum | Associated with patients exhibiting favorable responses to ICI therapy, potentially enhancing therapeutic efficacy through modulation of the immune system |
| Enterococcus hirae | Stimulates antigen-specific immune responses and enhances antitumor immunity |
| Barnesiella intestinihominis | Activates the immune system and potentiates antitumor immune responses |
| Lactobacillus genus | Produces lactate, modulates the immune system, and enhances antitumor immune responses |
| Clostridiales order | Produces short-chain fatty acids, modulates the immune system, and enhances antitumor immune responses |
Impact of key microbial species on immunotherapeutic response in breast cancer.
In breast cancer immunotherapy, Toll-like receptors (TLRs) have emerged as key mediators that bridge innate and adaptive immune responses. Among them, TLR5 is expressed in breast cancer cells and exerts antitumor effects via the MyD88-dependent NF-κB signaling pathway. Activation of TLR5 induces the secretion of proinflammatory cytokines such as IL-8 and TNF-α, which subsequently suppresses tumor cell proliferation and migration (Chen et al., 2022). The expression level of TLR5 has been strongly correlated with tumor aggressiveness and patient prognosis. Downregulation of TLR5 has been shown to promote proliferation, metastasis, and epithelial-to-mesenchymal transition (EMT) in triple-negative breast cancer (TNBC), whereas high TLR5 expression is linked to diminished metastatic potential and favorable clinical outcomes (Shi et al., 2019). Moreover, TLR5 activation has been reported to induce the expression of the autophagy-associated protein MAP1S, thereby promoting autophagic activity that reinforces tumor suppression (Shi et al., 2014). The gut microbiota potentiates antitumor immune responses through Toll-like receptor (TLR) upregulation and downstream activation of inflammatory signaling pathways, thereby promoting T and B cell activation and improving the tumor immune microenvironment (Routy et al., 2018). For instance, Salmonella typhimurium flagellin has been shown to activate the TLR5 signaling pathway, triggering a pro-inflammatory cascade that suppresses breast cancer cell proliferation while promoting immune cell recruitment and activation, thereby augmenting immunotherapeutic efficacy (Cai et al., 2011). Studies have demonstrated that in murine models of TNBC, TLR5 agonists—such as flagellin or its derivative CBLB502—markedly extend survival and elicit durable immunological memory when administered in combination with anti–PD-1 and anti–CTLA-4 antibodies. This combinatorial strategy overcomes resistance to ICIs by activating DCs and CD8+ T cells, thereby enhancing antitumor immune responses (Gonzalez et al., 2023). This mechanistic insight highlights a potential therapeutic avenue for breast cancer immunotherapy, suggesting that modulation of TLR-associated signaling pathways may enhance treatment efficacy.
Activation of the STING pathway exerts both direct antiproliferative effects on tumor cells and reprograms the tumor microenvironment to enhance the efficacy of ICIs (Wang et al., 2025). This pathway is primarily initiated by cytosolic DNA sensing by cyclic GMP-AMP synthase (cGAS), which catalyzes the production of cyclic GMP-AMP (cGAMP). cGAMP subsequently activates STING, leading to robust type I interferon production, maturation of dendritic cells (DCs), and enhanced antigen presentation, ultimately driving the activation and cytotoxic activity of CD8+ T cells (Shen et al., 2025). In addition, STING activation upregulates chemokines such as CXCL9 and CXCL10, thereby promoting T cell infiltration into the tumor microenvironment and further augmenting antitumor immunity (de Mingo Pulido et al., 2021). Notably, the gut microbiota and its metabolites have emerged as critical modulators of STING pathway activation. Recent studies have identified Akkermansia muciniphila as a crucial regulator of the STING signaling pathway through the production of cyclic di-AMP (cdAMP). By activating the type I interferon (IFN-I) signaling cascade, A. muciniphila not only reprograms macrophages toward an antitumor phenotype but also facilitates NK cell–DC interactions, thereby orchestrating a multilayered antitumor immune response (Lam et al., 2021). Certain gut microbial taxa, such as Bifidobacterium species, can activate the host STING pathway through their metabolic products, thereby inducing type I interferon production and establishing an antiviral state in the tumor microenvironment. This, in turn, promotes the activation and infiltration of CD8+T cells, which enhances the efficacy of ICIs (Jia et al., 2024). However, the role of STING signaling in breast cancer remains a subject of ongoing debate. Some studies suggest that, in certain contexts, activation of the STING pathway may paradoxically promote immune evasion and therapeutic resistance (Hong et al., 2022). Moreover, the clinical application of STING agonists remains limited due to poor pharmacokinetic stability, suboptimal bioavailability, and the risk of adverse effects. Consequently, the precise modulation of STING signaling to harness its antitumor potential while minimizing unintended consequences represents a critical avenue for future research.
Activation of G protein–coupled receptor (GPCR) signaling—particularly via gut microbiota–derived SCFAs—has emerged as a key mechanism for modulating the TME and potentiating immunotherapeutic efficacy (Perez Escriva et al., 2025). SCFAs such as butyrate, propionate, and acetate signal through specific GPCRs, including GPR41 (FFAR3), GPR43 (FFAR2), and GPR109A, thereby modulating T cell function, inflammatory cytokine secretion, and tumor-associated signaling pathways, all of which contribute critically to breast cancer immunotherapy (Li et al., 2025). Studies have shown that SCFAs enhance the efficacy of ICIs by activating GPR43 and GPR41 to regulate T cell differentiation, promote Treg development, and enhance the cytotoxic activity of CD8+ T cells (Di Modica et al., 2022). Despite these promising findings, the role of GPCR signaling in breast cancer remains contentious. Some reports suggest that GPCR activation may, in certain contexts, promote immune escape and therapeutic resistance (Al-Qadami et al., 2022). Furthermore, the clinical application of GPCR agonists is limited by challenges such as limited receptor specificity, poor bioavailability, and adverse effects. Therefore, the precise modulation of GPCR pathways to harness their antitumor potential while mitigating deleterious outcomes constitutes a critical avenue for future research.
Despite significant progress, numerous challenges remain in leveraging gut microbiota for breast cancer immunotherapeutic interventions. First, substantial heterogeneity exists in the composition and functional profiles of the gut microbiome across different breast cancer molecular subtypes, presenting a significant barrier to the clinical translation of microbiota-targeted therapies, given the considerable interpatient variability. Second, differences in immune regulatory mechanisms between animal models and human patients limit the direct applicability of preclinical findings. Additionally, exogenous factors such as antibiotic-induced microbiome disruption can compromise immunotherapeutic efficacy. Thus, strategies for restoring or modulating beneficial microbial populations without inducing adverse effects require extensive validation in large-scale prospective clinical trials and mechanistic investigations. Future research should focus on elucidating the precise molecular interactions between the gut microbiota and breast cancer immunotherapy, establishing a conceptual and mechanistic basis for the advancement of personalized, microbiota-informed immunotherapeutic strategies.
5 Modulating gut microbiota as a potential therapeutic approach in breast cancer treatment
Given the critical role of the gut microbiota in breast cancer progression and its impact on immunotherapy efficacy, current research is dedicated to unraveling its therapeutic implications. Modulating the gut microbiome is gaining recognition as a promising strategy to curb disease progression and improve patient prognosis, offering a potential avenue for advancing breast cancer treatment.
5.1 Dietary intervention
Long-term dietary habits play a crucial role in shaping the composition and activity of the gut microbiota. Studies have shown that the gut microbiome can rapidly respond to dietary changes, even over short periods (David et al., 2014). Notably, a high-fat, high-sugar diet has been linked to adverse immune therapy outcomes, whereas sufficient dietary fiber and salt consumption has been associated with improved clinical prognosis (Szczyrek et al., 2021). In postmenopausal women, a daily fiber intake exceeding 30 g is significantly associated with a reduced risk of breast cancer while simultaneously promoting the proliferation of Bifidobacterium and Faecalibacterium prausnitzii, both of which exert anti-inflammatory and antitumor effects (Chen J. et al., 2019). The ketogenic diet, characterized by high fat, moderate protein, and low carbohydrate intake, does not appear to increase circulating estrogen levels, despite its high lipid content. Notably, when implemented in combination with multiple complementary therapies, this dietary approach resulted in complete and sustained remission in a patient with end-stage metastatic ERα-positive breast cancer (Hilakivi-Clarke et al., 2022). Additionally, polyphenols can selectively inhibit pathogenic bacteria while promoting the growth of beneficial gut microbiota. In breast cancer mouse models, polyphenols have been shown to modulate gut microbiota composition and significantly inhibit tumor progression (Nandi et al., 2023). While existing data support the beneficial role of dietary and lifestyle interventions in modulating the gut microbiota, the standardization and integration of these strategies into clinical management necessitate rigorous validation and integration into clinical practice.
5.2 Fecal microbiota transplantation (FMT)
Fecal microbiota transplantation (FMT) involves the transfer of gut microbiota from a healthy donor to a recipient via the upper or lower gastrointestinal tract, aiming to reestablish microbial diversity and improve immunotherapeutic outcomes (Chen D. et al., 2019). An initial clinical trial has established the feasibility and safety of FMT in immunotherapy, showing promising outcomes in patients with anti-PD-1-refractory metastatic melanoma. Among 10 patients who had previously failed to respond to PD-1 blockade, three demonstrated renewed responsiveness to PD-1 blockade post-FMT. Notably, no severe immune-related adverse events (irAEs) were reported among the patients during the treatment period (Baruch et al., 2020). FMT has not yet been investigated as a therapeutic approach for breast cancer. Additional studies are needed to assess its therapeutic potential in breast cancer treatment and to determine whether microbiota modulation through FMT could enhance treatment efficacy in this setting.
5.3 Prebiotics and probiotics
Prebiotics are nondigestible dietary carbohydrates, including monosaccharides, polysaccharides, and fibers, that selectively promote the growth and activity of beneficial gut microbiota (Wu et al., 2022). Studies have demonstrated that prebiotics can inhibit breast cancer progression in both in vitro and in vivo models. Beyond commonly recognized prebiotics such as dietary fiber, emerging research has highlighted the gut microbiota-modulating properties of dietary phytochemicals, including polyphenols and flavonoids (Sun et al., 2018). Although the prebiotic properties of these phytochemicals require further investigation, they have been shown to promote the expansion of microbial taxa implicated in prebiotic-mediated gut health benefits. For instance, polyphenol intake has been associated with increased abundance of Bifidobacterium and Lactobacillus, as well as enhanced production of SCFAs, highlighting their significance in gut microbiota regulation. Probiotics, including species from the Lactobacillus and Bifidobacterium genera, are live microorganisms that confer health benefits to the host by actively modulating host physiological functions (Badgeley et al., 2021). Both preclinical and clinical studies have demonstrated that probiotic interventions—such as Lactobacillus casei BL23, Lactobacillus plantarum A, or a combination of Bifidobacterium lactis Bl-04 and Lactobacillus acidophilus NCFM—not only reestablish gut microbial homeostasis but also exhibit significant antitumor effects (Jacouton et al., 2017; Hibberd et al., 2017). In breast cancer survivors, administration of a probiotic blend containing Bifidobacterium longum BB536 and Lactobacillus rhamnosus HN001 has been shown to enhance microbial diversity while substantially lowering the Bacteroidetes-to-Firmicutes ratio, an indicator of gut microbiota dysbiosis. Additionally, probiotic supplementation has been linked to decreased body mass index (BMI) and enhanced insulin sensitivity (Pellegrini et al., 2020). These findings suggest that probiotics hold promise as an adjunctive approach for regulating metabolic and immune functions in breast cancer care.
Although these strategies have shown substantial antitumor efficacy in preclinical studies, significant interpatient variability and the intrinsic heterogeneity of gut microbiota composition pose substantial challenges to their broad clinical application. Tailored strategies based on individual microbiome profiles may be essential to optimize the therapeutic efficacy of microbiota-based interventions in breast cancer treatment.
6 Conclusion and perspectives
The role of the gut microbiota in shaping the breast cancer tumor microenvironment has attracted growing interest. Current research indicates that the microbiota exerts both direct and indirect effects on breast cancer initiation, progression, and metastasis through its regulation of estrogen metabolism, inflammatory signaling, and immune cell function. Moreover, its composition is strongly associated with the efficacy of immune checkpoint inhibitors and other therapeutic strategies. Although preclinical studies and emerging clinical data suggest that microbiota modulation represents a promising avenue for improving immunotherapy efficacy, several challenges remain in translating these findings into clinical applications. The causal relationships among the gut microbiota, immune system, and tumor development remain incompletely defined, and the underlying mechanisms warrant further in-depth investigation. The substantial heterogeneity in microbiota composition and function across different breast cancer molecular subtypes and among individuals poses a significant challenge to 389the standardization of microbiota-targeted therapies. Additionally, the precise mechanisms by which gut microbiota modulates immune responses through pathways such as TLR and STING, as well as the spatiotemporal dynamics of these interactions within the tumor microenvironment, are not yet fully elucidated. Comprehensive mechanistic studies to bridge these knowledge gaps will be essential for advancing microbiota-based strategies in breast cancer treatment. Future research should focus on identifying reliable biomarkers that can predict which subsets of breast cancer patients are most likely to respond to microbiota-targeted interventions. Moreover, a critical challenge in translating microbiota-targeted strategies from animal models to clinical applications is ensuring targeted and safe modulation while preserving microbial homeostasis in patients. Exogenous factors such as antibiotics, dietary interventions, and other environmental influences can perturb the gut microbiota, thereby potentially compromising immunotherapeutic efficacy. Therefore, well-designed large-scale prospective clinical trials are critical for rigorously assessing the safety and effectiveness of microbiota-modulating interventions. Future research should integrate multi-omics approaches, utilizing advanced technologies, including single-cell sequencing, spatial transcriptomics, and metabolomics, to systematically decipher the intricate interactions between the gut microbiota and the tumor microenvironment. Such comprehensive analyses will establish a solid conceptual framework for advancing personalized and precision-based therapeutic strategies in breast cancer treatment. Standardized and systematic clinical trial designs are critically needed to rigorously assess the efficacy and safety of various microbiota-based interventions—such as probiotics, FMT, and targeted metabolite supplementation—across distinct breast cancer subtypes. In parallel, the development of personalized microbial therapies, including customized probiotic formulations tailored to individual microbiota profiles and immunological features, represents a promising avenue for precision medicine. However, most current evidence is largely restricted to preclinical animal models, raising critical questions about the translatability of these findings to human settings. Challenges include interspecies differences in immune architecture and microbial ecology, the stability and reproducibility of microbiota interventions in humans, and the high degree of interindividual heterogeneity in immunotherapeutic responses. Therefore, future efforts should emphasize interdisciplinary collaboration and large-scale human cohort studies to establish clinically relevant model systems and accelerate the translation of microbiome research into therapeutic applications.
Overall, modulating the gut microbiota as a potential strategy for enhancing breast cancer immunotherapy represents a promising avenue. The integration of multimodal therapeutic strategies, including dietary interventions, prebiotic/probiotic supplementation, and microbiota transplantation, in conjunction with the continuous refinement of immunotherapeutic regimens, may help address existing challenges, facilitate clinical translation, and ultimately improve patient outcomes.
Statements
Author contributions
XR: Data curation, Investigation, Methodology, Writing – original draft. LZ: Conceptualization, Funding acquisition, Writing – review & editing. LH: Data curation, Investigation, Validation, Writing – review & editing. JZ: Conceptualization, Formal analysis, Supervision, Validation, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the Jiangxi Provincial Key Research and Development Program (nos. 20232ACB206053 and 20203BBGL73205).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that no Gen AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1
Al-QadamiG. H.SecombeK. R.SubramaniamC. B.WardillH. R.BowenJ. M. (2022). Gut microbiota-derived short-Chain fatty acids: impact on Cancer treatment response and toxicities. Microorganisms10:2048. doi: 10.3390/microorganisms10102048
2
AoZ.ShahS. H.MachlinL. M.ParajuliR.MillerP. C.RawalS.et al. (2015). Identification of Cancer-associated fibroblasts in circulating blood from patients with metastatic breast Cancer. Cancer Res.75, 4681–4687. doi: 10.1158/0008-5472.CAN-15-1633
3
BadgeleyA.AnwarH.ModiK.MurphyP.LakshmikuttyammaA. (2021). Effect of probiotics and gut microbiota on anti-cancer drugs: mechanistic perspectives. Biochim. Biophys. Acta Rev. Cancer1875:188494. doi: 10.1016/j.bbcan.2020.188494
4
BaruchE. N.Ben-BetzalelG.OrtenbergR.LahatA.KatzL.AdlerK.et al. (2020). Fecal microbiota transplant promotes response in immunotherapy-refractory melanoma patients. Science371, 602–609. doi: 10.1126/science.abb5920
5
BejaranoL.JordaoM. J. C.JoyceJ. A. (2021). Therapeutic targeting of the tumor microenvironment. Cancer Discov.11, 933–959. doi: 10.1158/2159-8290.CD-20-1808
6
BernardoG.Le NociV.Di ModicaM.MontanariE.TriulziT.PupaS. M.et al. (2023). The emerging role of the microbiota in breast Cancer progression. Cells12:945. doi: 10.3390/cells12151945
7
Buchta RoseanC.BosticR. R.FereyJ. C. M.FengT. Y.AzarF. N.TungK. S.et al. (2019). Preexisting commensal Dysbiosis is a host-intrinsic regulator of tissue inflammation and tumor cell dissemination in hormone receptor-positive breast Cancer. Cancer Res.79, 3662–3675. doi: 10.1158/0008-5472.CAN-18-3464
8
CaiZ.SanchezA.ShiZ.ZhangT.LiuM.ZhangD. (2011). Activation of toll-like receptor 5 on breast cancer cells by flagellin suppresses cell proliferation and tumor growth. Cancer Res.71, 2466–2475. doi: 10.1158/0008-5472.CAN-10-1993
9
CassettaL.FragkogianniS.SimsA. H.SwierczakA.ForresterL. M.ZhangH.et al. (2019). Human tumor-associated macrophage and monocyte transcriptional landscapes reveal Cancer-specific reprogramming, biomarkers, and therapeutic targets. Cancer Cell35:e10. doi: 10.1016/j.ccell.2019.02.009
10
ChenJ.DouglassJ.PrasathV.NeaceM.AtrchianS.ManjiliM. H.et al. (2019). The microbiome and breast cancer: a review. Breast Cancer Res. Treat.178, 493–496. doi: 10.1007/s10549-019-05407-5
11
ChenW.WeiF.XuJ.WangY.ChenL.WangJ.et al. (2011). Trastuzumab enhances the anti-tumor effects of the histone deacetylase inhibitor sodium butyrate on a Her2-overexpressing breast cancer cell line. Int. J. Mol. Med.28, 985–991. doi: 10.3892/ijmm.2011.790
12
ChenD.WuJ.JinD.WangB.CaoH. (2019). Fecal microbiota transplantation in cancer management: current status and perspectives. Int. J. Cancer145, 2021–2031. doi: 10.1002/ijc.32003
13
ChenX.ZhangY.FuY. (2022). The critical role of toll-like receptor-mediated signaling in cancer immunotherapy. Med. Drug Disc.14:100122. doi: 10.3389/fimmu.2018.01509
14
CordsL.TietscherS.AnzenederT.LangwiederC.ReesM.De SouzaN.et al. (2023). Cancer-associated fibroblast classification in single-cell and spatial proteomics data. Nat. Commun.14:4294. doi: 10.1038/s41467-023-39762-1
15
CortesJ.RugoH. S.CesconD. W.ImS. A.YusofM. M.GallardoC.et al. (2022). Pembrolizumab plus chemotherapy in advanced triple-negative breast Cancer. N. Engl. J. Med.387, 217–226. doi: 10.1056/NEJMoa2202809
16
CostaA.KiefferY.Scholer-DahirelA.PelonF.BourachotB.CardonM.et al. (2018). Fibroblast heterogeneity and immunosuppressive environment in human breast Cancer. Cancer Cell33:e10. doi: 10.1016/j.ccell.2018.01.011
17
CullinN.Azevedo AntunesC.StraussmanR.Stein-ThoeringerC. K.ElinavE. (2021). Microbiome and cancer. Cancer Cell39, 1317–1341. doi: 10.1016/j.ccell.2021.08.006
18
DanenbergE.BardwellH.ZanotelliV. R. T.ProvenzanoE.ChinS. F.RuedaO. M.et al. (2022). Breast tumor microenvironment structures are associated with genomic features and clinical outcome. Nat. Genet.54, 660–669. doi: 10.1038/s41588-022-01041-y
19
DavidL. A.MauriceC. F.CarmodyR. N.GootenbergD. B.ButtonJ. E.WolfeB. E.et al. (2014). Diet rapidly and reproducibly alters the human gut microbiome. Nature505, 559–563. doi: 10.1038/nature12820
20
De Mingo PulidoA.HanggiK.CeliasD. P.GardnerA.LiJ.Batista-BittencourtB.et al. (2021). The inhibitory receptor Tim-3 limits activation of the CGAS-Sting pathway in intra-tumoral dendritic cells by suppressing extracellular DNA uptake. Immunity54, 1154–1167. doi: 10.1016/j.immuni.2021.04.019
21
DeepakK. G. K.NagarajuG. P.DasariV. R.NaginiS.RaoD. N.MallaR. R. (2020). Tumor microenvironment: challenges and opportunities in targeting metastasis of triple negative breast cancer. Pharmacol. Res.153:104683. doi: 10.1016/j.phrs.2020.104683
22
DethlefsenL.Mcfall-NgaiM.RelmanD. A. (2007). An ecological and evolutionary perspective on human-microbe mutualism and disease. Nature449, 811–818. doi: 10.1038/nature06245
23
Di ModicaM.ArlottaV.SfondriniL.TagliabueE.TriulziT. (2022). The link between the microbiota and Her2+ breast Cancer: the new challenge of precision medicine. Front. Oncol.12:947188. doi: 10.3389/fonc.2022.947188
24
Domenica ReaG. C.PalmaG.BarbieriA.LucianoA.Del PreteP.RossettiS.et al. (2018). Microbiota effects on cancer: from risks to therapies. Oncotarget9, 17915–17927. doi: 10.18632/oncotarget.24681
25
DovrolisN.SpathakisM.CollinsA. R.PandeyV. K.UddinM. I.AndersonD. D.et al. (2024). Pan-Cancer insights: A study of microbial metabolite receptors in malignancy dynamics. Cancers16:4178. doi: 10.3390/cancers16244178
26
ErnyD.DokalisN.MezoC.CastoldiA.MossadO.StaszewskiO.et al. (2021). Microbiota-derived acetate enables the metabolic fitness of the brain innate immune system during health and disease. Cell Metab.33:e7. doi: 10.1016/j.cmet.2021.10.010
27
FalkR. T.BrintonL. A.DorganJ. F.FuhrmanB. J.VeenstraT. D.XuX.et al. (2013). Relationship of serum estrogens and estrogen metabolites to postmenopausal breast cancer risk: a nested case-control study. Breast Cancer Res.15:R34. doi: 10.1186/bcr3416
28
FehervariZ. (2021). Microbiota shape tumor immunity. Nat. Immunol.22:1469. doi: 10.1038/s41590-021-01082-1
29
FuA.YaoB.DongT.ChenY.YaoJ.LiuY.et al. (2022). Tumor-resident intracellular microbiota promotes metastatic colonization in breast cancer. Cell185:e26. doi: 10.1016/j.cell.2022.02.027
30
FuhrmanB. J.SchairerC.GailM. H.Boyd-MorinJ.XuX.SueL. Y.et al. (2012). Estrogen metabolism and risk of breast cancer in postmenopausal women. J. Natl. Cancer Inst.104, 326–339. doi: 10.1093/jnci/djr531
31
GarmpisN.DamaskosC.GarmpiA.KalampokasE.KalampokasT.SpartalisE.et al. (2017). Histone deacetylases as new therapeutic targets in triple-negative breast Cancer: Progress and promises. Cancer Genomics Proteomics14, 299–313. doi: 10.21873/cgp.20041
32
GeldhofV.De RooijL.SokolL.AmersfoortJ.De SchepperM.RohlenovaK.et al. (2022). Single cell atlas identifies lipid-processing and immunomodulatory endothelial cells in healthy and malignant breast. Nat. Commun.13:5511. doi: 10.1038/s41467-022-33052-y
33
GonzalezC.WilliamsonS.GammonS. T.GlazerS.RheeJ. H.Piwnica-WormsD. (2023). Tlr5 agonists enhance anti-tumor immunity and overcome resistance to immune checkpoint therapy. Commun. Biol.6:31. doi: 10.1038/s42003-022-04403-8
34
GoveiaJ.RohlenovaK.TavernaF.TrepsL.ConradiL. C.PircherA.et al. (2020). An integrated gene expression landscape profiling approach to identify lung tumor endothelial cell heterogeneity and Angiogenic candidates. Cancer Cell37:e13. doi: 10.1016/j.ccell.2019.12.001
35
GwakJ. M.JangM. H.KimD. I.SeoA. N.ParkS. Y. (2015). Prognostic value of tumor-associated macrophages according to histologic locations and hormone receptor status in breast cancer. PLoS One10:e0125728. doi: 10.1371/journal.pone.0125728
36
Haishan LinE. L.HestirK.LeoC.HuangM.BoschE.HalenbeckR.et al. (2008). Discovery of a cytokine and its receptor by functional screening of the extracellular proteome. Science320, 807–811. doi: 10.1126/science.1154370
37
HanahanD.WeinbergR. A. (2011). Hallmarks of cancer: the next generation. Cell144, 646–674. doi: 10.1016/j.cell.2011.02.013
38
HeY.FuL.LiY.WangW.GongM.ZhangJ.et al. (2021). Gut microbial metabolites facilitate anticancer therapy efficacy by modulating cytotoxic Cd8(+) T cell immunity. Cell Metab.33:e7. doi: 10.1016/j.cmet.2021.03.002
39
HibberdA. A.LyraA.OuwehandA. C.RolnyP.LindegrenH.CedgardL.et al. (2017). Intestinal microbiota is altered in patients with colon cancer and modified by probiotic intervention. BMJ Open Gastroenterol.4:e000145. doi: 10.1136/bmjgast-2017-000145
40
Hilakivi-ClarkeL.VermaV.McdermottM.KoakP.AndradeF. D. O. (2022). Foods may modify responsiveness to cancer immune checkpoint blockers by altering both the gut microbiota and activation of estrogen receptors in immune cells. Front. Microbiomes1:1049688. doi: 10.3389/frmbi.2022.1049688
41
HongC.SchubertM.TijhuisA. E.RequesensM.RoordaM.Van Den BrinkA.et al. (2022). CGAS-Sting drives the Il-6-dependent survival of chromosomally instable cancers. Nature607, 366–373. doi: 10.1038/s41586-022-04847-2
42
IizumiT.BattagliaT.RuizV.Perez PerezG. I. (2017). Gut microbiome and antibiotics. Arch. Med. Res.48, 727–734. doi: 10.1016/j.arcmed.2017.11.004
43
JacoutonE.ChainF.SokolH.LangellaP.Bermudez-HumaranL. G. (2017). Probiotic strain Lactobacillus casei Bl23 prevents colitis-associated colorectal Cancer. Front. Immunol.8:1553. doi: 10.3389/fimmu.2017.01553
44
JayeK.LiC. G.ChangD.BhuyanD. J. (2022). The role of key gut microbial metabolites in the development and treatment of cancer. Gut Microbes14:2038865. doi: 10.1080/19490976.2022.2038865
45
JiaD.WangQ.QiY.JiangY.HeJ.LinY.et al. (2024). Microbial metabolite enhances immunotherapy efficacy by modulating T cell stemness in pan-cancer. Cell187:e21. doi: 10.1016/j.cell.2024.02.022
46
JinM. Z.JinW. L. (2020). The updated landscape of tumor microenvironment and drug repurposing. Signal Trans. Targeted Ther.5:166. doi: 10.1038/s41392-020-00280-x
47
JugderB. E.KamareddineL.WatnickP. I. (2021). Microbiota-derived acetate activates intestinal innate immunity via the Tip60 histone acetyltransferase complex. Immunity54:e3. doi: 10.1016/j.immuni.2021.05.017
48
KerenL.BosseM.MarquezD.AngoshtariR.JainS.VarmaS.et al. (2018). A structured tumor-immune microenvironment in triple negative breast Cancer revealed by multiplexed ion beam imaging. Cell174:e19. doi: 10.1016/j.cell.2018.08.039
49
KeumN.GreenwoodD. C.LeeD. H.KimR.AuneD.JuW.et al. (2015). Adult weight gain and adiposity-related cancers: a dose-response meta-analysis of prospective observational studies. J. Natl. Cancer Inst.107:djv088. doi: 10.1093/jnci/djv088
50
KiefferY.HocineH. R.GentricG.PelonF.BernardC.BourachotB.et al. (2020). Single-cell analysis reveals fibroblast clusters linked to immunotherapy resistance in Cancer. Cancer Discov.10, 1330–1351. doi: 10.1158/2159-8290.CD-19-1384
51
LamK. C.ArayaR. E.HuangA.ChenQ.Di ModicaM.RodriguesR. R.et al. (2021). Microbiota triggers Sting-type I Ifn-dependent monocyte reprogramming of the tumor microenvironment. Cell184:e21. doi: 10.1016/j.cell.2021.09.019
52
LiS.DuanY.LuoS.ZhouF.WuQ.LuZ. (2025). Short-chain fatty acids and cancer. Trends Cancer11, 154–168. doi: 10.1016/j.trecan.2024.11.003
53
LuenS.VirassamyB.SavasP.SalgadoR.LoiS. (2016). The genomic landscape of breast cancer and its interaction with host immunity. Breast29, 241–250. doi: 10.1016/j.breast.2016.07.015
54
MaJ.HuangL.HuD.ZengS.HanY.ShenH. (2021). The role of the tumor microbe microenvironment in the tumor immune microenvironment: bystander, activator, or inhibitor?J. Exp. Clin. Cancer Res.40:327. doi: 10.1186/s13046-021-02128-w
55
MallottE. K.AmatoK. R. (2021). Host specificity of the gut microbiome. Nat. Rev. Microbiol.19, 639–653. doi: 10.1038/s41579-021-00562-3
56
McquadeJ. L.DanielC. R.HelminkB. A.WargoJ. A. (2019). Modulating the microbiome to improve therapeutic response in cancer. Lancet Oncol.20, e77–e91. doi: 10.1016/S1470-2045(18)30952-5
57
MikoE.VidaA.KovacsT.UjlakiG.TrencsenyiG.MartonJ.et al. (2018). Lithocholic acid, a bacterial metabolite reduces breast cancer cell proliferation and aggressiveness. Biochim. Biophys. Acta Bioenerg.1859, 958–974. doi: 10.1016/j.bbabio.2018.04.002
58
MoraisL. H.SchreiberH. L. T.MazmanianS. K. (2021). The gut microbiota-brain axis in behaviour and brain disorders. Nat. Rev. Microbiol.19, 241–255. doi: 10.1038/s41579-020-00460-0
59
MotzG. T.SantoroS. P.WangL. P.GarrabrantT.LastraR. R.HagemannI. S.et al. (2014). Tumor endothelium FasL establishes a selective immune barrier promoting tolerance in tumors. Nat. Med.20, 607–615. doi: 10.1038/nm.3541
60
Nalio RamosR.Missolo-KoussouY.Gerber-FerderY.BromleyC. P.BugattiM.NunezN. G.et al. (2022). Tissue-resident Folr2(+) macrophages associate with Cd8(+) T cell infiltration in human breast cancer. Cell185:e25. doi: 10.1016/j.cell.2022.02.021
61
NandiD.ParidaS.SharmaD. (2023). The gut microbiota in breast cancer development and treatment: the good, the bad, and the useful!Gut Microbes15:2221452. doi: 10.1080/19490976.2023.2221452
62
NejmanD.LivyatanI.FuksG.GavertN.ZwangY.GellerL. T.et al. (2020). The human tumor microbiome is composed of tumor type-specific intracellular bacteria. Science368, 973–980. doi: 10.1126/science.aay9189
63
PalB.ChenY.VaillantF.CapaldoB. D.JoyceR.SongX.et al. (2021). A single-cell RNA expression atlas of normal, preneoplastic and tumorigenic states in the human breast. EMBO J.40:e107333. doi: 10.15252/embj.2020107333
64
ParhiL.Alon-MaimonT.SolA.NejmanD.ShhadehA.Fainsod-LeviT.et al. (2020). Breast cancer colonization by Fusobacterium nucleatum accelerates tumor growth and metastatic progression. Nat. Commun.11:3259. doi: 10.1038/s41467-020-16967-2
65
PellegriniM.IppolitoM.MongeT.VioliR.CappelloP.FerrocinoI.et al. (2020). Gut microbiota composition after diet and probiotics in overweight breast cancer survivors: a randomized open-label pilot intervention trial. Nutrition74:110749. doi: 10.1016/j.nut.2020.110749
66
Perez EscrivaP.BernardinoC. T.LetellierE. (2025). De-coding the complex role of microbial metabolites in cancer. Cell Rep.44:115358. doi: 10.1016/j.celrep.2025.115358
67
PerouC. M.EisenM. B.Van De RijnM.JeffreykS. S.ReesC. A.PollackJ. R.et al. (2000). Molecular portraits of human breast tumours. Nature406, 747–752. doi: 10.1038/35021093
68
PratA.ParkerJ. S.KarginovaO.FanC.LivasyC.HerschkowitzJ. I.et al. (2010). Phenotypic and molecular characterization of the claudin-low intrinsic subtype of breast cancer. Breast Cancer Res.12:R68. doi: 10.1186/bcr2635
69
RoutyB.GopalakrishnanV.DaillereR.ZitvogelL.WargoJ. A.KroemerG. (2018). The gut microbiota influences anticancer immunosurveillance and general health. Nat. Rev. Clin. Oncol.15, 382–396. doi: 10.1038/s41571-018-0006-2
70
RussoJ. R. A. I. H. (2006). The role of estrogen in the initiation of Brest Cancer. J. Steroid Biochem. Mol. Biol.102, 89–96. doi: 10.1016/j.jsbmb.2006.09.004
71
SampsellK.HaoD.ReimerR. A. (2020). The gut microbiota: A potential gateway to improved health outcomes in breast Cancer treatment and survivorship. Int. J. Mol. Sci.21:239. doi: 10.3390/ijms21239239
72
SavasP.VirassamyB.YeC.SalimA.MintoffC. P.CaramiaF.et al. (2018). Single-cell profiling of breast cancer T cells reveals a tissue-resident memory subset associated with improved prognosis. Nat. Med.24, 986–993. doi: 10.1038/s41591-018-0078-7
73
SchmidP.CortesJ.PusztaiL.McarthurH.KummelS.BerghJ.et al. (2020). Pembrolizumab for early triple-negative breast Cancer. N. Engl. J. Med.382, 810–821. doi: 10.1056/NEJMoa1910549
74
ShenM.JiangX.PengQ.OyangL.RenZ.WangJ.et al. (2025). The cgas–Sting pathway in cancer immunity: mechanisms, challenges, and therapeutic implications. J. Hematol. Oncol.18:40. doi: 10.1186/s13045-025-01691-5
75
ShiM.YaoY.HanF.LiY.LiY. (2014). Map1S controls breast cancer cell Tlr5 signaling pathway and promotes Tlr5 signaling-based tumor suppression. PLoS One9:e86839. doi: 10.1371/journal.pone.0086839
76
ShiD.ZhaoS.JiangW.ZhangC.LiangT.HouG. (2019). Tlr5: A prognostic and monitoring indicator for triple-negative breast cancer. Cell Death Dis.10:954. doi: 10.1038/s41419-019-2187-8
77
SommerF.AndersonJ. M.BhartiR.RaesJ.RosenstielP. (2017). The resilience of the intestinal microbiota influences health and disease. Nat. Rev. Microbiol.15, 630–638. doi: 10.1038/nrmicro.2017.58
78
SunH.ChenY.ChengM.ZhangX.ZhengX.ZhangZ. (2018). The modulatory effect of polyphenols from green tea, oolong tea and black tea on human intestinal microbiota in vitro. J. Food Sci. Technol.55, 399–407. doi: 10.1007/s13197-017-2951-7
79
SungH.FerlayJ.SiegelR. L.LaversanneM.SoerjomataramI.JemalA.et al. (2021). Global Cancer statistics 2020: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin.71, 209–249. doi: 10.3322/caac.21660
80
SwantonC.BernardE.AbboshC.AndreF.AuwerxJ.BalmainA.et al. (2024). Embracing cancer complexity: hallmarks of systemic disease. Cell187, 1589–1616. doi: 10.1016/j.cell.2024.02.009
81
SzczyrekM.BitkowskaP.ChunowskiP.CzuchrytaP.KrawczykP.MilanowskiJ. (2021). Diet, microbiome, and Cancer immunotherapy-A comprehensive review. Nutrients13:2217. doi: 10.3390/nu13072217
82
TimperiE.GueguenP.MolgoraM.MagagnaI.KiefferY.Lopez-LastraS.et al. (2022). Lipid-associated macrophages are induced by Cancer-associated fibroblasts and mediate immune suppression in breast Cancer. Cancer Res.82, 3291–3306. doi: 10.1158/0008-5472.CAN-22-1427
83
WangY.HanY.YangC.BaiT.ZhangC.WangZ.et al. (2024). Long-term relapse-free survival enabled by integrating targeted antibacteria in antitumor treatment. Nat. Commun.15:4194. doi: 10.1038/s41467-024-48662-x
84
WangS.QinL.LiuF.ZhangZ. (2025). Unveiling the crossroads of Sting signaling pathway and metabolic reprogramming: the multifaceted role of the Sting in the TME and new prospects in cancer therapies. Cell Commun. Signal23:171. doi: 10.1186/s12964-025-02169-0
85
WillingB. P.RussellS. L.FinlayB. B. (2011). Shifting the balance: antibiotic effects on host-microbiota mutualism. Nat. Rev. Microbiol.9, 233–243. doi: 10.1038/nrmicro2536
86
WuS. Z.Al-EryaniG.RodenD. L.JunankarS.HarveyK.AnderssonA.et al. (2021). A single-cell and spatially resolved atlas of human breast cancers. Nat. Genet.53, 1334–1347. doi: 10.1038/s41588-021-00911-1
87
WuH.GangulyS.TollefsbolT. O. (2022). Modulating microbiota as a new strategy for breast Cancer prevention and treatment. Microorganisms10:727. doi: 10.3390/microorganisms10091727
88
WuS. Z.RodenD. L.WangC.HollidayH.HarveyK.CazetA. S.et al. (2020). Stromal cell diversity associated with immune evasion in human triple-negative breast cancer. EMBO J.39:e104063. doi: 10.15252/embj.2019104063
89
ZhangJ.XiaY.SunJ. (2021). Breast and gut microbiome in health and cancer. Genes Dis8, 581–589. doi: 10.1016/j.gendis.2020.08.002
90
ZhengD.LiwinskiT.ElinavE. (2020). Interaction between microbiota and immunity in health and disease. Cell Res.30, 492–506. doi: 10.1038/s41422-020-0332-7
91
ZhouY.HanW.FengY.WangY.SunT.XuJ. (2024). Microbial metabolites affect tumor progression, immunity and therapy prediction by reshaping the tumor microenvironment (review). Int. J. Oncol.65:5661. doi: 10.3892/ijo.2024.5661
92
ZhuB. T.ConneyA. H. (1998). Functional role of estrogen metabolism in target cells: review and perspectives. Carcinogenesis19, 1–27. doi: 10.1093/carcin/19.1.1
Summary
Keywords
breast cancer, tumor microenvironment, immunotherapy, gut microbiota, dietary intervention, fecal microbiota transplantation, prebiotics
Citation
Ren X, Zheng L, Huang L and Zhao J (2025) The role of the gut microbiota in shaping the tumor microenvironment and immunotherapy of breast cancer. Front. Microbiol. 16:1591745. doi: 10.3389/fmicb.2025.1591745
Received
11 March 2025
Accepted
13 May 2025
Published
27 May 2025
Volume
16 - 2025
Edited by
Maria Gazouli, National and Kapodistrian University of Athens, Greece
Reviewed by
Gratiela Gradisteanu Pircalabioru, University of Bucharest, Romania
Sheetal Parida, Johns Hopkins University, United States
Nikolas Dovrolis, Democritus University of Thrace, Greece
Updates
Copyright
© 2025 Ren, Zheng, Huang and Zhao.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jiangbo Zhao, jxsetyyzjb@163.com
†These authors have contributed equally to this work
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.