- 1Laboratory of Gene Regulation and Signal Transduction, Department of Pharmacology, University of California San Diego School of Medicine, La Jolla, CA, USA
- 2Laboratory of Gene Regulation and Signal Transduction, Department of Pathology, University of California San Diego School of Medicine, La Jolla, CA, USA
It is becoming increasingly clear that inflammation influences prostate cancer (PCa) development and that immune cells are among the primary drivers of this effect. This information has launched numerous clinical trials testing immunotherapy drugs in PCa patients. The results of these studies are promising but have yet to generate a complete response. Importantly, the precise immune profile that determines clinical outcome remains unresolved. Individual immune cell types are divided into various functional subsets whose effects on tumor development may differ depending on their particular phenotype and functional status, which is often shaped by the tumor microenvironment. Thus, this review aims to examine the current knowledge regarding the role of inflammation and specific immune cell types in mediating PCa progression to assist in directing and optimizing immunotherapy targets, regimens, and responses and to uncover areas in which further research is needed. Finally, a summary of ongoing immunotherapy clinical trials in PCa is provided.
Introduction
Prostate cancer (PCa) is the most common non-cutaneous cancer and second leading cancer-related cause of death of men in the United States. (1). Localized PCa can be controlled with surgery or radiation over 80% of the time. If prostate tumors are metastatic at presentation, neither surgery nor radiation will confer significant clinical benefit and androgen deprivation therapy (ADT) represents the sole therapeutic option for such patients. Unfortunately, resistance to hormonal targeting develops in most patients within 2 years, a state that is termed metastatic castration-resistant PCa (mCRPC) and is virtually untreatable (2, 3). Until recently, Docetaxel was the lone accepted remedy for mCRPC and was associated with a 2- to 3-month enhancement of survival (4–6). The advent of newer chemotherapeutic agents such as cabazitaxel and approval of next generation drugs abiraterone and enzalutamide have improved upon this effect (7–10), yet resistance ultimately ensues, possibly via mutations in the androgen receptor (AR) gene (11).
Relative to other forms of cancer, the discovery of valuable predictive markers of disease progression has been particularly challenging for PCa. Genetic markers capable of identifying clinically aggressive disease remain elusive (12, 13). In addition, PCa is unique among solid tumors in that imaging techniques are insufficient for definitive identification and diagnosis of malignancy (12, 14). Instead, the pre-biopsy serum prostate specific antigen (PSA) value is used as an indicator of potential underlying disease, but the utility of this screening method has been intensely scrutinized (15). In fact, the United States Preventative Services Task Force (USPSTF) recently recommended against routine PSA screening (16). Importantly, the ramifications of this ruling may be an increase in the proportion of men diagnosed at later stages of PCa (17). Thus, the identification of novel therapeutic targets for and biomarkers of mCRPC is severely needed.
A multitude of studies investigating alternative PCa treatment interventions or predictive markers have focused on components of the inflammatory response, with promising results. A relationship between inflammation and cancer was first proposed by Virchow in 1863 (18) and chronic inflammation is now well accepted as a driving force behind many malignancies, such as gastric, colon, esophageal and lung cancers, and hepatocellular carcinoma (19). While the role of the immune system in PCa is less clear, it has been implicated both as a possible etiologic agent and a tumor promoter. A recent report from the REDUCE study suggesting that aspirin and/or NSAID use decreased risk of PCa in men with a negative biopsy supports this notion (20). This review will explore the evidence regarding immune infiltration as a potential mediator and indicator of aggressive PCa, as well as a target for PCa therapeutics. Because this topic has been comprehensively evaluated elsewhere (21–26), only recent literature will be addressed here.
Prostatitis and Prostate Cancer: The Evidence
As with many other cancers, PCa arises from accumulation of acquired genetic and epigenetic alterations. Currently, age, ethnicity, and family history are the only known risk factors for development of prostate neoplasia. These genetic traits may partially explain the geographical variance in PCa incidence and mortality, with rates in Southeast and East Asia being much lower than those in the United States and Western Europe (22, 27). By contrast, the elevated rate of PCa observed in Chinese and Japanese men within one generation of relocation to the West indicates that environmental factors, particularly lifestyle factors such as diet, also contribute to the emergence of PCa (28). One likely link between environmental stimuli and PCa occurrence is chronic inflammation. Support for this hypothesis comes from reports of focal atrophic lesions in the prostate containing a relatively high percentage of proliferating epithelial cells and acute or chronic inflammatory infiltrate. These areas, designated proliferative inflammatory atrophy (PIA), comprise an extensive portion of the peripheral zone of the prostate, the primary site where PCa originates, and have been suggested to be precursors of prostatic intraepithelial neoplastic (PIN) lesions (22, 24, 29), although one recent retrospective epidemiological study refuted this concept (30). Additional evidence comes from a recent meta-analysis assessing 20 case–control studies conducted between 1990 and 2012 that revealed a significant positive relationship between self-reported prostatitis and PCa. However, the authors were unable to demonstrate a causal link between the two due to the absence of cohort studies in this analysis (31). Numerous other epidemiological investigations have examined the connection between prostatitis and subsequent PCa risk and while early studies (32–35) described an elevated risk of malignancy or recurrence due to presence of inflammation, subsequent studies have produced conflicting results (29, 30, 36–40). In addition to differences in study populations and potential selection and other forms of bias, plausible explanations for these inconsistencies include tissue sample size and composition, definition and measurement of inflammation, tissue regions assessed, and categories of prostatitis examined. In order to obtain a more accurate reflection of the relationship between prostatitis and PCa, improved standardization of these measurements, incorporation of asymptomatic inflammatory prostatitis, and a prospective design in future epidemiological studies are needed. Importantly, many investigations defined inflammation with discrete variables, such as present or absent or severe, moderate or mild rather than with a detailed analysis of the cellular composition and distribution of the infiltrate. Although this classification system may be obligatory for certain statistical analyses, it is a major limitation of these investigations and may be masking true associations that exist. Furthermore, these studies may benefit from experimental research designed to provide insight regarding the kinetics, distribution, type, and components of inflammation that are important for prostate tumorigenesis.
Despite the abundance of data evaluating the connection between prostatitis and PCa, the sources of prostatic inflammation have yet to be fully elucidated. Proposed causes can be divided into three broad categories: (1) microorganism induced, including E. coli, Propionibacterium acnes, and those associated with urine reflux and sexually transmitted diseases, (2) non-microorganism induced, such as uric acid crystals formed during urine reflux, estrogen, dietary factors, especially heterocyclic amines (HCA) 2-amino-1-methyl-6-phenylimidazo[4,5-b] pyridine (PhIP) generated from cooking meats at high temperatures (41), obesity, and physical trauma such as corpora amylacea (23–25, 42–44), and (3) treatment- or tumor-elicited inflammation (45, 46).
Some of these hypotheses were tested using mouse models. For example, Shinohara et al. reported that intraurethral inoculation of mice with a prostatectomy-derived strain of P. acnes elicited prolonged and lobe-specific severe acute and chronic inflammation that was associated with increased proliferation and reduced Nkx3.1 and AR expression. Inflammatory infiltrate consisted of luminal neutrophils and stromal mononuclear cells (44). Similar observations were noted in experiments using E. coli. Introduction of this bacterium into mouse urogenital tracts provoked a dramatic decline in Nkx3.1 and AR expression and a corresponding enhancement of proliferation as well as the basal cell marker p63. These changes were pursuant to an early influx of neutrophils into prostatic lumen and stroma followed by a surge in stromal mononuclear cells and later a marked increase in plasma cells (43). Upregulation of pro-inflammatory genes, including IL-6 and COX-2, was also detected using a comparable system (42). The effects of these events on PCa pathogenesis were examined by combining bacteria CP9-induced prostatitis and a mouse PCa model and revealed that tissue damage resulting from acute prostatitis facilitates accumulation of stromal α-smooth muscle actin-positive myofibroblasts and the conversion of basal cells into luminal cells, which accelerates PCa progression (47).
Non-bacterial inflammatory stimuli were also evaluated with respect to PCa development. A novel model of prostatitis involving ubiquitous overexpression of aromatase (AROM+) in mice exhibited early and persistent prostatic mast cell infiltration with subsequent accumulation of neutrophils, T lymphocytes, and macrophages, as well as increased expression of numerous chemokines. This chronic inflammation preceded formation of PIN lesions containing and surrounded by immune cells (48). Finally, Birbach et al. expressed a constitutively active form of the inflammatory mediator IκB kinase 2 in prostate epithelium of mice harboring a heterozygous Pten deletion and detected a robust inflammatory response, primarily encompassing neutrophils and macrophages in the prostate, that was accompanied by increased tumor size due to enhanced epithelial and stromal proliferation, generation of cribiform structures and stromal remodeling, but no invasion or metastasis (49).
Together, these data corroborate the ability of inflammatory stimuli to generate an immune response that modulates the local environment into one that is conducive to malignant transformation and progression and implicate chronic inflammation as a causative factor in PCa. More studies combining prostatitis and murine PCa models are needed to confirm these effects and determine the mechanisms by which these agents promote disease. Of course, these results also must be confirmed in humans.
Immune Cells and Prostate Cancer: Friends or Foes?
As previously described, epidemiological studies exploring the relationship between prostatitis and PCa development may be limited by the absence of characterization of cellular phenotypes displayed by tumor-infiltrating leukocytes (TIL). However, many studies assessing the association between specific immune cells and PCa are now available and those conducted between 2008 and 2014 are summarized according to cell type in Table 1. Most of these investigated CD3+, CD4+, or CD8+ cells in prostatectomy or biopsy tissue of PCa patients and found such cells to be pro-tumorigenic. For example, a cross-sectional study of lymphocyte infiltration in tumor tissue microarrays from patients with PSA recurrence reported that extremely high or low CD3+ cell counts were correlated with reduced PSA recurrence-free survival (RFS) (50). Importantly, CD3 was the only T-cell marker included in this study and no evaluations of T-cell subsets were performed. Thus, the variation in clinical outcomes across CD3+ cell quartiles may at least partially reflect differences in T-cell populations. In fact, the authors mentioned unpublished data indicating that higher numbers of regulatory T-cells (Treg) were associated with more advanced tumor stage and PSA-RFS. A likely assumption is that the CD3+ cells whose low numbers conferred poor prognoses were protective in nature. Thus, it is critical to determine not only the cell type but also the subtype associated with different outcomes in order to better direct therapeutic efforts. By contrast, the spontaneous TRAMP mouse PCa model was used to illustrate that the trafficking of a naturally arising clonally expanded PCa histone H4-specific population of CD44+CD8+ T-cells to prostate tissue lessened tumor burden but failed to improve survival (51). Several possible explanations for the lack of survival impact exist. First, it may be that the anti-tumorigenic CTL were initially successful in eliminating tumor cells but were eventually rendered inactive by a suppressive tumor microenvironment. Alternatively, it is conceivable that the antigen-specific destruction of H4-expressing tumor cells selected for less immunogenic and possibly more adept H4-negative tumor cells, or instead, that this response exerted pressure on cancer cells to downregulate MHC or undergo immunoediting of tumor antigens and thereby escape immune recognition and destruction. In line with the former hypothesis, several investigations demonstrated that tumor-infiltrating CTL expressed PD-1, one of several indicators of T-cell exhaustion, in PCa patients (52, 53). This is an essential consideration for immunotherapeutic efforts and emphasizes the importance of evaluating both the cellular subtype and functional status when assessing TIL.
Another cell type that has received much attention and shown relatively consistent results in the context of PCa progression is the tumor-associated macrophage (TAM). All studies analyzed demonstrated a pro-tumorigenic effect of TAM, although one report (54) specified that this was true only for M2 type macrophages. However, only a fraction of the literature displayed a link between TAM and recurrence (55, 56). In particular, Nonomura et al. (56) determined TAM count to be a significant prognostic factor of clinical outcome in PCa patients. A second study that found TAM to be related to recurrence indicated this to be true only in the control but not ADT treated group. Thus, TAM count may be a good biomarker for disease progression, but further research is needed to identify the specific subset of macrophages and PCa patients for which this is applicable. TAM may also represent a viable therapeutic target for PCa, as demonstrated by Escamilla et al., who showed a reduction in castration-induced recruitment of pro-tumorigenic TAM and delayed emergence of CRPC in a mouse Myc-CaP PCa model following CSF1R inhibition (57).
As expected, presence of immunosuppressive immune cells, such as Treg (52, 58), myeloid derived suppressor cells (MDSC) (59–61), and according to one report, TH17 cells (62), in prostate tissue was suggestive of more aggressive disease, but surprisingly few studies were able to equate this with clinical prognosis. Neutrophils and mast cells have also been fairly well studied, but investigations involving both cell types have generated conflicting results concerning their influence on PCa. One source of contention may concern the area of tissue evaluated. This point is nicely illustrated by Johannson et al. (63), who implicated peritumoral but not intratumoral mast cells as independent prognostic variables of PCa progression. In addition, work from Pittoni et al. (64) emphasized that care must be taken to evaluate potential negative outcomes when manipulating immune cell populations with therapies. In the TRAMP model of murine PCa, targeting mast cells prevented growth of well-differentiated tumors but simultaneously stimulated progression of more aggressive, neuroendocrine type tumors. Similarly, Tang et al. observed a late IL-2-induced expansion of Treg following castration and immunization that negated the initial anti-tumorigenic CD8+ T-cell response (65).
Relative to other immune cells, B cells have been generally overlooked in terms of PCa. However, the importance of these cells in PCa progression was established in a landmark and subsequent papers (45, 66, 67) using subcutaneous Myc-CaP and spontaneous TRAMP mouse PCa models to show that castration-induced cell death and resulting hypoxia stimulated CXCL13-mediated infiltration of lymphotoxin β (LTb)-expressing B lymphocytes into regressing tumors. These cells subsequently activated IKKα, STAT3 and BMI1 in cancer cells to promote CRPC progression. Notably, interference with any aspect of this pathway delayed emergence of hormone-resistant tumors. These initial findings were extended to reveal that the identical mechanism operates in androgen-dependent regeneration of normal prostate progenitor cells (67). Interestingly, an independent investigation reported significant reductions in human and murine PCa tumor growth following combination therapy with HIF-1α and phospho-STAT3 inhibitors (68). Together, these data suggest that the B cell response to tissue injury or death following androgen depletion drives restorative or aberrant proliferation in the prostate. In addition, a recent report implicates immunosuppressive IgA+PD-L1+IL-10+ plasma cells in impeding response to low-dose chemotherapy in a T-cell-dependent manner (69). The presence of CD20+ B cells has been documented in human PCa as well, and the density of these cells was shown to be higher in malignant than benign tissue (70). Furthermore, a case study of an advanced PCa patient treated with the B cell depleting antibody Rituximab produced fairly encouraging results (71). A Phase 0 clinical trial evaluating Rituximab as neoadjuvant therapy in patients prior to radical prostatectomy is currently ongoing. Thus, although research is still in preliminary stages, B cells may represent an emerging target for PCa immunotherapeutics. However, it is likely that a damage response (45, 67) and/or an immunogenic stimulus (69) is needed to make these cells relevant in prostate carcinogenesis or that only particular subsets, such as those expressing LTβ, or IgA are crucial.
Prostate Cancer and Immunotherapy
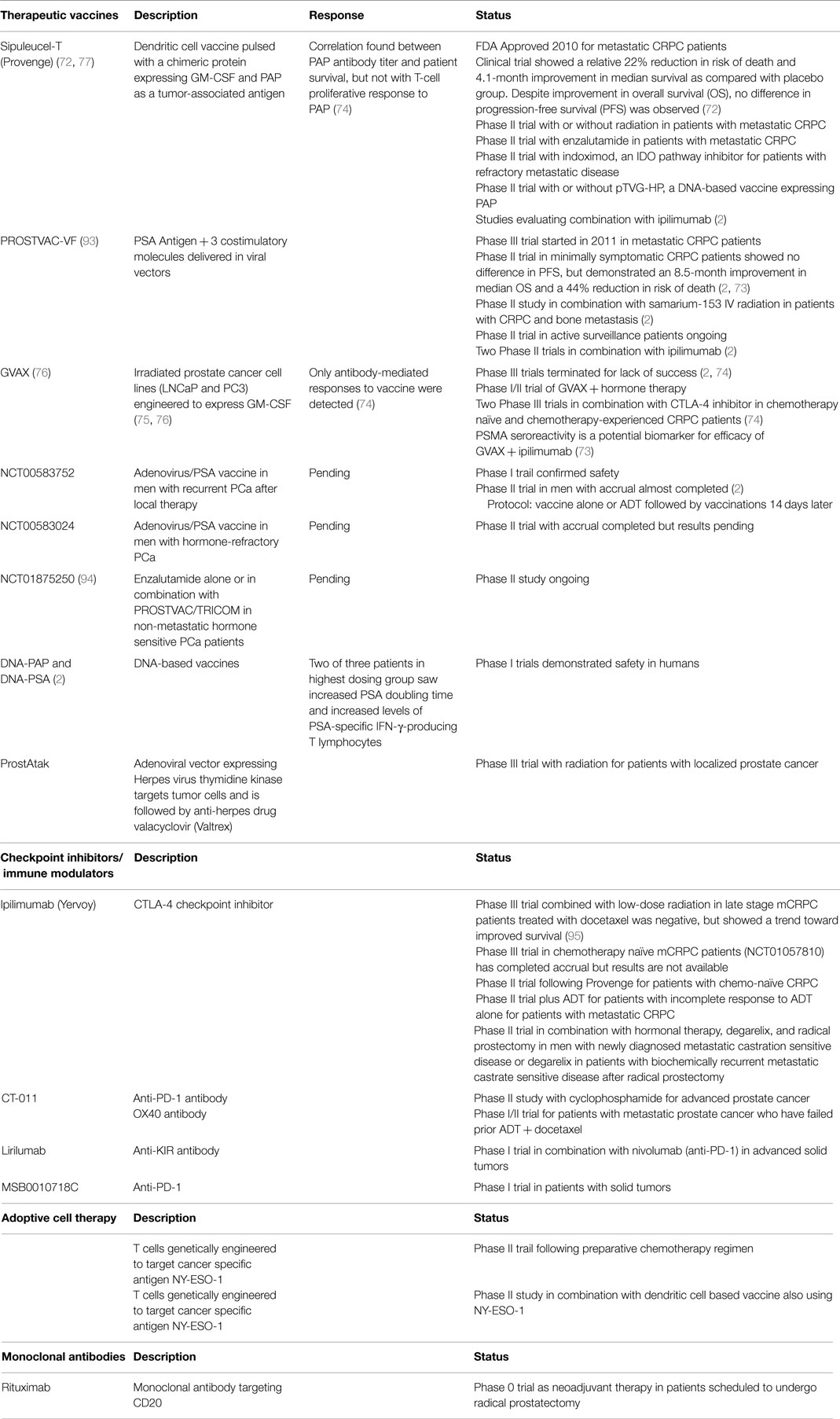
The emergence of immune cells as important mediators of PCa progression has spawned a multitude of clinical trials targeting these cells via different approaches, including therapeutic vaccines, checkpoint inhibitors/immune modulators, adoptive T-cell therapy, and monoclonal antibodies. In theory, PCa should be particularly conducive to this type of treatment due to its long latency period. However, because PCa is commonly diagnosed late in life, therapeutic vaccines may not be as efficacious as desired due to the age-related decline in immune responsiveness. Despite this fact, Sipuleucel-T was the first immunotherapy approved by the FDA for PCa and has shown an overall survival advantage in mCRPC patients, although no effect on progression-free survival was noted (72). The lack of short-term response observed in this study may at least partially be partially attributed to the fact that immunotherapeutic agents are not directly cytotoxic, as are more standard treatments. Due to space limitations, individual studies will not be discussed in detail here but are summarized in Table 2 (2, 72–77). One important message from the combined studies is that, while extremely promising, these immunotherapeutic methodologies still need to be optimized as no individual treatment has yet elicited a complete response. Complete and durable responses will likely require combinations of current immunotherapy and/or more standard approaches, such as chemotherapy, radiation, or ADT, which induce an immunogenic stimulus or enhance T-cell responses. In addition, ideal target patient populations and treatment protocols still need to be identified. For example, a study combining PROSTVAC and ipilimumab discovered that patients with lower PD-1+Tim-3NEGCD4EM, higher PD-1NEGTim-3+CD8+, and more CTLA-4NEG Treg at baseline survived longer. Furthermore, an increase in Tim-3+ natural killer cells following vaccination conferred survival benefits (78). Similarly, Klyushnenkova et al. (79) illustrated the importance of breaking immunological tolerance to “self” antigen by targeting CD25+ Treg during CTLA-4 blockade in mice. Although this research has several shortcomings, it highlights the potential value of such combination therapies.
Summary
The presence of inflammatory cells in the prostate is well documented. Evidence suggests that these cells may drive PCa development. Additionally, growing tumors can induce recruitment of immune cells into the prostate microenvironment and initiate a reciprocal interaction that promotes disease progression. Surprisingly, information regarding precise profiles of prostate tumor-infiltrating leukocytes is extremely limited. Individual cells and the overall immune response have the potential to be anti- or pro-tumorigenic, depending on cellular phenotypes, combinations, localization and the tumor microenvironment. Thus, the identities, traits, functional status, distribution, kinetics, and interactions of inflammatory cells in the prostate and PCa must be fully characterized in order to elevate and combine the already promising current immunotherapies. Furthermore, efforts must be directed at elucidating better indicators of potential response to facilitate identification of ideal target populations, as well as biomarkers to assess therapeutic efficacy. Progress in these areas will strengthen the existing optimism that immunotherapy will become part of a successful standard care regimen for future PCa patients.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
MK is an American Cancer Society Research Professor and research in his lab is supported by the NIH and the American Association for Cancer Research. The authors apologize to those whose papers were not cited due to space restrictions.
References
1. Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin (2014) 64:9–29. doi:10.3322/caac.21208
2. Uhlman MA, Bing MT, Lubaroff DM. Prostate cancer vaccines in combination with additional treatment modalities. Immunol Res (2014) 59:236–42. doi:10.1007/s12026-014-8532-1
3. Kantoff PW, Carroll PR, D’Amico AV, editors. Prostate Cancer: Principles and Practice. Philadelphia, PA: Lippincott William & Wilkins (2002).
4. Berthold DR, Pond GR, Soban F, de Wit R, Eisenberger M, Tannock IF. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer: updated survival in the TAX 327 study. J Clin Oncol (2008) 26:242–5. doi:10.1200/JCO.2007.12.4008
5. Petrylak DP, Tangen CM, Hussain MH, Lara PN Jr, Jones JA, Taplin ME, et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med (2004) 351:1513–20. doi:10.1056/NEJMoa041318
6. Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Chi KN, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med (2004) 351:1502–12. doi:10.1056/NEJMoa040720
7. Basch E, Autio K, Ryan CJ, Mulders P, Shore N, Kheoh T, et al. Abiraterone acetate plus prednisone versus prednisone alone in chemotherapy-naive men with metastatic castration-resistant prostate cancer: patient-reported outcome results of a randomised phase 3 trial. Lancet Oncol (2013) 14:1193–9. doi:10.1016/S1470-2045(13)70424-8
8. de Bono JS, Logothetis CJ, Molina A, Fizazi K, North S, Chu L, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med (2011) 364:1995–2005. doi:10.1056/NEJMoa1014618
9. Scher HI, Fizazi K, Saad F, Taplin ME, Sternberg CN, Miller K, et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med (2012) 367:1187–97. doi:10.1056/NEJMoa1207506
10. Schrader AJ, Boegemann M, Ohlmann CH, Schnoeller TJ, Krabbe LM, Hajili T, et al. Enzalutamide in castration-resistant prostate cancer patients progressing after docetaxel and abiraterone. Eur Urol (2014) 65:30–6. doi:10.1016/j.eururo.2013.06.042
11. Culig Z. Targeting the androgen receptor in prostate cancer. Expert Opin Pharmacother (2014) 15:1427–37. doi:10.1517/14656566.2014.915313
12. Taverna G, Giusti G, Seveso M, Hurle R, Colombo P, Stifter S, et al. Mast cells as a potential prognostic marker in prostate cancer. Dis Markers (2013) 35:711–20. doi:10.1155/2013/478303
13. Xu J, Sun J, Zheng SL. Prostate cancer risk-associated genetic markers and their potential clinical utility. Asian J Androl (2013) 15:314–22. doi:10.1038/aja.2013.42
14. Morris MJ, Autio KA, Basch EM, Danila DC, Larson S, Scher HI. Monitoring the clinical outcomes in advanced prostate cancer: what imaging modalities and other markers are reliable? Semin Oncol (2013) 40:375–92. doi:10.1053/j.seminoncol.2013.04.008
15. Ryan CJ, Shah S, Efstathiou E, Smith MR, Taplin ME, Bubley GJ, et al. Phase II study of abiraterone acetate in chemotherapy-naive metastatic castration-resistant prostate cancer displaying bone flare discordant with serologic response. Clin Cancer Res (2011) 17:4854–61. doi:10.1158/1078-0432.CCR-11-0815
16. Moyer VA. Screening for prostate cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med (2012) 157:120–34. doi:10.7326/0003-4819-157-2-201207170-00459
17. Hall MD, Schultheiss TE, Farino G, Wong JYC. Increase in higher risk prostate cancer cases following new screening recommendation by the US preventive services task force (USPSTF). J Clin Oncol (2015) 33(7 Suppl):143.
18. Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature (2008) 454:436–44. doi:10.1038/nature07205
19. Kuraishy A, Karin M, Grivennikov SI. Tumor promotion via injury- and death-induced inflammation. Immunity (2011) 35:467–77. doi:10.1016/j.immuni.2011.09.006
20. Vidal AC, Howard LE, Moreira DM, Castro-Santamaria R, Andriole GL, Freedland SJ. Aspirin, NSAIDs, and risk of prostate cancer: results from the REDUCE study. Clin Cancer Res (2015) 21:756–62. doi:10.1158/1078-0432.CCR-14-2235
21. Bardan R, Dumache R, Dema A, Cumpanas A, Bucuras V. The role of prostatic inflammation biomarkers in the diagnosis of prostate diseases. Clin Biochem (2014) 47:909–15. doi:10.1016/j.clinbiochem.2014.02.008
22. De Marzo AM, Platz EA, Sutcliffe S, Xu J, Gronberg H, Drake CG, et al. Inflammation in prostate carcinogenesis. Nat Rev Cancer (2007) 7:256–69. doi:10.1038/nrc2090
23. Elkahwaji JE. The role of inflammatory mediators in the development of prostatic hyperplasia and prostate cancer. Res Rep Urol (2012) 5:1–10. doi:10.2147/RRU.S23386
24. Nakai Y, Nonomura N. Inflammation and prostate carcinogenesis. Int J Urol (2013) 20:150–60. doi:10.1111/j.1442-2042.2012.03101.x
25. Sfanos KS, De Marzo AM. Prostate cancer and inflammation: the evidence. Histopathology (2012) 60:199–215. doi:10.1111/j.1365-2559.2011.04033.x
26. Vasto S, Carruba G, Candore G, Italiano E, Di Bona D, Caruso C. Inflammation and prostate cancer. Future Oncol (2008) 4:637–45. doi:10.2217/14796694.4.5.637
27. Hsing AW, Tsao L, Devesa SS. International trends and patterns of prostate cancer incidence and mortality. Int J Cancer (2000) 85:60–7. doi:10.1002/(SICI)1097-0215(20000101)85:1<60::AID-IJC11>3.0.CO;2-B
28. Peto J. Cancer epidemiology in the last century and the next decade. Nature (2001) 411:390–5. doi:10.1038/35077256
29. Davidsson S, Fiorentino M, Andren O, Fang F, Mucci LA, Varenhorst E, et al. Inflammation, focal atrophic lesions, and prostatic intraepithelial neoplasia with respect to risk of lethal prostate cancer. Cancer Epidemiol Biomarkers Prev (2011) 20:2280–7. doi:10.1158/1055-9965.EPI-11-0373
30. Vral A, Magri V, Montanari E, Gazzano G, Gourvas V, Marras E, et al. Topographic and quantitative relationship between prostate inflammation, proliferative inflammatory atrophy and low-grade prostate intraepithelial neoplasia: a biopsy study in chronic prostatitis patients. Int J Oncol (2012) 41:1950–8. doi:10.3892/ijo.2012.1646
31. Jiang J, Li J, Yunxia Z, Zhu H, Liu J, Pumill C. The role of prostatitis in prostate cancer: meta-analysis. PLoS One (2013) 8:e85179. doi:10.1371/journal.pone.0085179
32. Dennis LK, Lynch CF, Torner JC. Epidemiologic association between prostatitis and prostate cancer. Urology (2002) 60:78–83. doi:10.1016/S0090-4295(02)01637-0
33. Irani J, Goujon JM, Ragni E, Peyrat L, Hubert J, Saint F, et al. High-grade inflammation in prostate cancer as a prognostic factor for biochemical recurrence after radical prostatectomy. Pathologist multi center study group. Urology (1999) 54:467–72. doi:10.1016/S0090-4295(99)00152-1
34. MacLennan GT, Eisenberg R, Fleshman RL, Taylor JM, Fu P, Resnick MI, et al. The influence of chronic inflammation in prostatic carcinogenesis: a 5-year followup study. J Urol (2006) 176:1012–6. doi:10.1016/j.juro.2006.04.033
35. Roberts RO, Bergstralh EJ, Bass SE, Lieber MM, Jacobsen SJ. Prostatitis as a risk factor for prostate cancer. Epidemiology (2004) 15:93–9. doi:10.1097/01.ede.0000101022.38330.7c
36. Engelhardt PF, Brustmann H, Seklehner S, Riedl CR. Chronic asymptomatic inflammation of the prostate type IV and carcinoma of the prostate: is there a correlation? Scand J Urol (2013) 47:230–5. doi:10.3109/00365599.2012.733961
37. Gurel B, Lucia MS, Thompson IM Jr, Goodman PJ, Tangen CM, Kristal AR, et al. Chronic inflammation in benign prostate tissue is associated with high-grade prostate cancer in the placebo arm of the prostate cancer prevention trial. Cancer Epidemiol Biomarkers Prev (2014) 23:847–56. doi:10.1158/1055-9965.EPI-13-1126
38. Moreira DM, Nickel JC, Gerber L, Muller RL, Andriole GL, Castro-Santamaria R, et al. Baseline prostate inflammation is associated with a reduced risk of prostate cancer in men undergoing repeat prostate biopsy: results from the REDUCE study. Cancer (2014) 120:190–6. doi:10.1002/cncr.28349
39. Pepe P, Aragona F. Does an inflammatory pattern at primary biopsy suggest a lower risk for prostate cancer at repeated saturation prostate biopsy? Urol Int (2011) 87:171–4. doi:10.1159/000328043
40. Yli-Hemminki TH, Laurila M, Auvinen A, Maattanen L, Huhtala H, Tammela TL, et al. Histological inflammation and risk of subsequent prostate cancer among men with initially elevated serum prostate-specific antigen (PSA) concentration in the Finnish prostate cancer screening trial. BJU Int (2013) 112:735–41. doi:10.1111/bju.12153
41. Li G, Wang H, Liu AB, Cheung C, Reuhl KR, Bosland MC, et al. Dietary carcinogen 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine-induced prostate carcinogenesis in CYP1A-humanized mice. Cancer Prev Res (Phila) (2012) 5:963–72. doi:10.1158/1940-6207.CAPR-12-0023
42. Boehm BJ, Colopy SA, Jerde TJ, Loftus CJ, Bushman W. Acute bacterial inflammation of the mouse prostate. Prostate (2012) 72:307–17. doi:10.1002/pros.21433
43. Khalili M, Mutton LN, Gurel B, Hicks JL, De Marzo AM, Bieberich CJ. Loss of Nkx3.1 expression in bacterial prostatitis: a potential link between inflammation and neoplasia. Am J Pathol (2010) 176:2259–68. doi:10.2353/ajpath.2010.080747
44. Shinohara DB, Vaghasia AM, Yu SH, Mak TN, Bruggemann H, Nelson WG, et al. A mouse model of chronic prostatic inflammation using a human prostate cancer-derived isolate of Propionibacterium acnes. Prostate (2013) 73:1007–15. doi:10.1002/pros.22648
45. Ammirante M, Luo JL, Grivennikov S, Nedospasov S, Karin M. B-cell-derived lymphotoxin promotes castration-resistant prostate cancer. Nature (2010) 464:302–5. doi:10.1038/nature08782
46. Mercader M, Bodner BK, Moser MT, Kwon PS, Park ES, Manecke RG, et al. T cell infiltration of the prostate induced by androgen withdrawal in patients with prostate cancer. Proc Natl Acad Sci U S A (2001) 98:14565–70. doi:10.1073/pnas.251140998
47. Kwon OJ, Zhang L, Ittmann MM, Xin L. Prostatic inflammation enhances basal-to-luminal differentiation and accelerates initiation of prostate cancer with a basal cell origin. Proc Natl Acad Sci U S A (2014) 111:E592–600. doi:10.1073/pnas.1318157111
48. Ellem SJ, Wang H, Poutanen M, Risbridger GP. Increased endogenous estrogen synthesis leads to the sequential induction of prostatic inflammation (prostatitis) and prostatic pre-malignancy. Am J Pathol (2009) 175:1187–99. doi:10.2353/ajpath.2009.081107
49. Birbach A, Eisenbarth D, Kozakowski N, Ladenhauf E, Schmidt-Supprian M, Schmid JA. Persistent inflammation leads to proliferative neoplasia and loss of smooth muscle cells in a prostate tumor model. Neoplasia (2011) 13:692–703. doi:10.1593/neo.11524
50. Flammiger A, Bayer F, Cirugeda-Kuhnert A, Huland H, Tennstedt P, Simon R, et al. Intratumoral T but not B lymphocytes are related to clinical outcome in prostate cancer. APMIS (2012) 120:901–8. doi:10.1111/j.1600-0463.2012.02924.x
51. Savage PA, Vosseller K, Kang C, Larimore K, Riedel E, Wojnoonski K, et al. Recognition of a ubiquitous self antigen by prostate cancer-infiltrating CD8+ T lymphocytes. Science (2008) 319:215–20. doi:10.1126/science.1148886
52. Ebelt K, Babaryka G, Frankenberger B, Stief CG, Eisenmenger W, Kirchner T, et al. Prostate cancer lesions are surrounded by FOXP3+, PD-1+ and B7-H1+ lymphocyte clusters. Eur J Cancer (2009) 45:1664–72. doi:10.1016/j.ejca.2009.02.015
53. Sfanos KS, Bruno TC, Meeker AK, De Marzo AM, Isaacs WB, Drake CG. Human prostate-infiltrating CD8+ T lymphocytes are oligoclonal and PD-1+. Prostate (2009) 69:1694–703. doi:10.1002/pros.21020
54. Lanciotti M, Masieri L, Raspollini MR, Minervini A, Mari A, Comito G, et al. The role of M1 and M2 macrophages in prostate cancer in relation to extracapsular tumor extension and biochemical recurrence after radical prostatectomy. Biomed Res Int (2014) 2014:486798. doi:10.1155/2014/486798
55. Gannon PO, Poisson AO, Delvoye N, Lapointe R, Mes-Masson AM, Saad F. Characterization of the intra-prostatic immune cell infiltration in androgen-deprived prostate cancer patients. J Immunol Methods (2009) 348:9–17. doi:10.1016/j.jim.2009.06.004
56. Nonomura N, Takayama H, Nakayama M, Nakai Y, Kawashima A, Mukai M, et al. Infiltration of tumour-associated macrophages in prostate biopsy specimens is predictive of disease progression after hormonal therapy for prostate cancer. BJU Int (2011) 107:1918–22. doi:10.1111/j.1464-410X.2010.09804.x
57. Escamilla J, Schokrpur S, Liu C, Priceman SJ, Moughon D, Jiang Z, et al. CSF1 receptor targeting in prostate cancer reverses macrophage-mediated resistance to androgen blockade therapy. Cancer Res (2015) 75:950–62. doi:10.1158/0008-5472.CAN-14-0992
58. Valdman A, Jaraj SJ, Comperat E, Charlotte F, Roupret M, Pisa P, et al. Distribution of Foxp3-, CD4- and CD8-positive lymphocytic cells in benign and malignant prostate tissue. APMIS (2010) 118:360–5. doi:10.1111/j.1600-0463.2010.02604.x
59. Di Mitri D, Toso A, Chen JJ, Sarti M, Pinton S, Jost TR, et al. Tumour-infiltrating Gr-1 myeloid cells antagonize senescence in cancer. Nature (2014) 515(7525):134–7. doi:10.1038/nature13638
60. Garcia AJ, Ruscetti M, Arenzana TL, Tran LM, Bianci-Frias D, Sybert E, et al. Pten null prostate epithelium promotes localized myeloid-derived suppressor cell expansion and immune suppression during tumor initiation and progression. Mol Cell Biol (2014) 34:2017–28. doi:10.1128/MCB.00090-14
61. Idorn M, Kollgaard T, Kongsted P, Sengelov L, Thor Straten P. Correlation between frequencies of blood monocytic myeloid-derived suppressor cells, regulatory T cells and negative prognostic markers in patients with castration-resistant metastatic prostate cancer. Cancer Immunol Immunother (2014) 63(11):1177–87. doi:10.1007/s00262-014-1591-2
62. Derhovanessian E, Adams V, Hahnel K, Groeger A, Pandha H, Ward S, et al. Pretreatment frequency of circulating IL-17+ CD4+ T-cells, but not Tregs, correlates with clinical response to whole-cell vaccination in prostate cancer patients. Int J Cancer (2009) 125:1372–9. doi:10.1002/ijc.24497
63. Johansson A, Rudolfsson S, Hammarsten P, Halin S, Pietras K, Jones J, et al. Mast cells are novel independent prognostic markers in prostate cancer and represent a target for therapy. Am J Pathol (2010) 177:1031–41. doi:10.2353/ajpath.2010.100070
64. Pittoni P, Tripodo C, Piconese S, Mauri G, Parenza M, Rigoni A, et al. Mast cell targeting hampers prostate adenocarcinoma development but promotes the occurrence of highly malignant neuroendocrine cancers. Cancer Res (2011) 71:5987–97. doi:10.1158/0008-5472.CAN-11-1637
65. Tang S, Moore ML, Grayson JM, Dubey P. Increased CD8+ T-cell function following castration and immunization is countered by parallel expansion of regulatory T cells. Cancer Res (2012) 72:1975–85. doi:10.1158/0008-5472.CAN-11-2499
66. Ammirante M, Shalapour S, Kang Y, Jamieson CA, Karin M. Tissue injury and hypoxia promote malignant progression of prostate cancer by inducing CXCL13 expression in tumor myofibroblasts. Proc Natl Acad Sci U S A (2014) 111:14776–81. doi:10.1073/pnas.1416498111
67. Ammirante M, Kuraishy AI, Shalapour S, Strasner A, Ramirez-Sanchez C, Zhang W, et al. An IKKalpha-E2F1-BMI1 cascade activated by infiltrating B cells controls prostate regeneration and tumor recurrence. Genes Dev (2013) 27:1435–40. doi:10.1101/gad.220202.113
68. Reddy KR, Guan Y, Qin G, Zhou Z, Jing N. Combined treatment targeting HIF-1alpha and Stat3 is a potent strategy for prostate cancer therapy. Prostate (2011) 71:1796–809. doi:10.1002/pros.21397
69. Shalapour S, Font-Burgada J, Di Caro G, Zhong Z, Wang K, Ammirante M, et al. Immunosuppressive plasma cells impede T cell-dependent immunogenic chemotherapy. Nature (2015) 521(7550):94–8. doi:10.1038/nature14395
70. Woo JR, Liss MA, Muldong MT, Palazzi K, Strasner A, Ammirante M, et al. Tumor infiltrating B-cells are increased in prostate cancer tissue. J Transl Med (2014) 12:30. doi:10.1186/1479-5876-12-30
71. Dalgleish A, Featherstone P, Vlassov V, Rogosnitzky M. Rituximab for treating CD20+ prostate cancer with generalized lymphadenopathy: a case report and review of the literature. Invest New Drugs (2014) 32:1048–52. doi:10.1007/s10637-014-0063-z
72. Kantoff PW, Higano CS, Shore ND, Berger ER, Small EJ, Penson DF, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med (2010) 363:411–22. doi:10.1056/NEJMoa1001294
73. Gerritsen WR. The evolving role of immunotherapy in prostate cancer. Ann Oncol (2012) 23(Suppl 8):viii22–7. doi:10.1093/annonc/mds259
74. Rigamonti N, Bellone M. Prostate cancer, tumor immunity and a renewed sense of optimism in immunotherapy. Cancer Immunol Immunother (2012) 61:453–68. doi:10.1007/s00262-012-1216-6
75. Simons JW, Mikhak B, Chang JF, DeMarzo AM, Carducci MA, Lim M, et al. Induction of immunity to prostate cancer antigens: results of a clinical trial of vaccination with irradiated autologous prostate tumor cells engineered to secrete granulocyte-macrophage colony-stimulating factor using ex vivo gene transfer. Cancer Res (1999) 59:5160–8.
76. Simons JW, Sacks N. Granulocyte-macrophage colony-stimulating factor-transduced allogeneic cancer cellular immunotherapy: the GVAX vaccine for prostate cancer. Urol Oncol (2006) 24:419–24. doi:10.1016/j.urolonc.2005.08.021
77. Small EJ, Fratesi P, Reese DM, Strang G, Laus R, Peshwa MV, et al. Immunotherapy of hormone-refractory prostate cancer with antigen-loaded dendritic cells. J Clin Oncol (2000) 18:3894–903.
78. Jochems C, Tucker JA, Tsang KY, Madan RA, Dahut WL, Liewehr DJ, et al. A combination trial of vaccine plus ipilimumab in metastatic castration-resistant prostate cancer patients: immune correlates. Cancer Immunol Immunother (2014) 63:407–18. doi:10.1007/s00262-014-1524-0
79. Klyushnenkova EN, Riabov VB, Kouiavskaia DV, Wietsma A, Zhan M, Alexander RB. Breaking immune tolerance by targeting CD25+ regulatory T cells is essential for the anti-tumor effect of the CTLA-4 blockade in an HLA-DR transgenic mouse model of prostate cancer. Prostate (2014) 74:1423–32. doi:10.1002/pros.22858
80. Fujii T, Shimada K, Asai O, Tanaka N, Fujimoto K, Hirao K, et al. Immunohistochemical analysis of inflammatory cells in benign and precancerous lesions and carcinoma of the prostate. Pathobiology (2013) 80:119–26. doi:10.1159/000342396
81. Yuan H, Hsiao YH, Zhang Y, Wang J, Yin C, Shen R, et al. Destructive impact of T-lymphocytes, NK and Mast cells on basal cell layers: implications for tumor invasion. BMC Cancer (2013) 13:258. doi:10.1186/1471-2407-13-258
82. Sfanos KS, Bruno TC, Maris CH, Xu L, Thoburn CJ, DeMarzo AM, et al. Phenotypic analysis of prostate-infiltrating lymphocytes reveals TH17 and Treg skewing. Clin Cancer Res (2008) 14:3254–61. doi:10.1158/1078-0432.CCR-07-5164
83. Gollapudi K, Galet C, Grogan T, Zhang H, Said JW, Huang J, et al. Association between tumor-associated macrophage infiltration, high grade prostate cancer, and biochemical recurrence after radical prostatectomy. Am J Cancer Res (2013) 3:523–9.
84. Craig M, Ying C, Loberg RD. Co-inoculation of prostate cancer cells with U937 enhances tumor growth and angiogenesis in vivo. J Cell Biochem (2008) 103:1–8. doi:10.1002/jcb.21379
85. Fleischmann A, Schlomm T, Kollermann J, Sekulic N, Huland H, Mirlacher M, et al. Immunological microenvironment in prostate cancer: high mast cell densities are associated with favorable tumor characteristics and good prognosis. Prostate (2009) 69:976–81. doi:10.1002/pros.20948
86. Sonpavde G, Pond GR, Armstrong AJ, Clarke SJ, Vardy JL, Templeton AJ, et al. Prognostic impact of the neutrophil-to-lymphocyte ratio in men with metastatic castration-resistant prostate cancer. Clin Genitourin Cancer (2014) 12:317–24. doi:10.1016/j.clgc.2014.03.005
87. Fujita K, Imamura R, Tanigawa G, Nakagawa M, Hayashi T, Kishimoto N, et al. Low serum neutrophil count predicts a positive prostate biopsy. Prostate Cancer Prostatic Dis (2012) 15:386–90. doi:10.1038/pcan.2012.27
88. Sumbul AT, Sezer A, Abali H, Kose F, Gultepe I, Mertsoylu H, et al. Neutrophil-to-lymphocyte ratio predicts PSA response, but not outcomes in patients with castration-resistant prostate cancer treated with docetaxel. Int Urol Nephrol (2014) 46:1531–5. doi:10.1007/s11255-014-0664-7
89. Templeton AJ, Pezaro C, Omlin A, McNamara MG, Leibowitz-Amit R, Vera-Badillo FE, et al. Simple prognostic score for metastatic castration-resistant prostate cancer with incorporation of neutrophil-to-lymphocyte ratio. Cancer (2014) 120(21):3346–52. doi:10.1002/cncr.28890
90. Nuhn P, Vaghasia AM, Goyal J, Zhou XC, Carducci MA, Eisenberger MA, et al. Association of pretreatment neutrophil-to-lymphocyte ratio (NLR) and overall survival (OS) in patients with metastatic castration-resistant prostate cancer (mCRPC) treated with first-line docetaxel. BJU Int (2013) 114:E11–7. doi:10.1111/bju.12531
91. Shafique K, Proctor MJ, McMillan DC, Qureshi K, Leung H, Morrison DS. Systemic inflammation and survival of patients with prostate cancer: evidence from the Glasgow inflammation outcome study. Prostate Cancer Prostatic Dis (2012) 15:195–201. doi:10.1038/pcan.2011.60
92. Sadeghi N, Badalato GM, Hruby G, Grann V, McKiernan JM. Does absolute neutrophil count predict high tumor grade in African-American men with prostate cancer? Prostate (2012) 72:386–91. doi:10.1002/pros.21440
93. DiPaola RS, Plante M, Kaufman H, Petrylak DP, Israeli R, Lattime E, et al. A phase I trial of pox PSA vaccines (PROSTVAC-VF) with B7-1, ICAM-1, and LFA-3 co-stimulatory molecules (TRICOM) in patients with prostate cancer. J Transl Med (2006) 4:1. doi:10.1186/1479-5876-4-1
94. Burotto M, Singh N, Heery CR, Gulley JL, Madan RA. Exploiting synergy: immune-based combinations in the treatment of prostate cancer. Front Oncol (2014) 4:351. doi:10.3389/fonc.2014.00351
95. Kwon ED, Drake CG, Scher HI, Fizazi K, Bossi A, van den Eertwegh AJ, et al. Ipilimumab versus placebo after radiotherapy in patients with metastatic castration-resistant prostate cancer that had progressed after docetaxel chemotherapy (CA184-043): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol (2014) 15:700–12. doi:10.1016/S1470-2045(14)70189-5
Keywords: prostate cancer, immune cells, inflammation, immunotherapy, B cells
Citation: Strasner A and Karin M (2015) Immune infiltration and prostate cancer. Front. Oncol. 5:128. doi: 10.3389/fonc.2015.00128
Received: 02 April 2015; Accepted: 22 May 2015;
Published: 08 July 2015
Edited by:
Shidong Jia, Genentech Inc, USAReviewed by:
Rosemary O’Connor, University College Cork, IrelandClaire Perks, University of Bristol, UK
Copyright: © 2015 Strasner and Karin. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Michael Karin, Laboratory of Gene Regulation and Signal Transduction, Departments of Pharmacology and Pathology, University of California San Diego School of Medicine, 9500 Gilman Drive Mail Code 0723, La Jolla, CA 92093, USA,a2FyaW5vZmZpY2VAdWNzZC5lZHU=
 Amy Strasner
Amy Strasner Michael Karin
Michael Karin