- 1School of Biomedical Sciences, Institute of Health and Biomedical Innovation, Queensland University of Technology, Kelvin Grove, QLD, Australia
- 2Translational Research Institute, Brisbane, QLD, Australia
- 3School of Biomedical Engineering, University of Technology Sydney, Sydney, NSW, Australia
- 4Institute of Molecular Medicine, I.M. Sechenov First Moscow State Medical University, Moscow, Russia
- 5Department of Urology, Princess Alexandra Hospital, Woolloongabba, QLD, Australia
- 6Australian Prostate Cancer Research Centre, Institute of Health and Biomedical Innovation, Translational Research Institute, Queensland University of Technology, Princess Alexandra Hospital, Brisbane, QLD, Australia
- 7School of Medicine, University of Queensland, Royal Brisbane and Women's Hospital, Central Integrated Regional Cancer Service, Queensland Health, Brisbane, QLD, Australia
- 8Princess Alexandra Hospital, Queensland Health, Brisbane, QLD, Australia
Lung cancer affects over 1. 8 million people worldwide and is the leading cause of cancer related mortality globally. Currently, diagnosis of lung cancer involves a combination of imaging and invasive biopsies to confirm histopathology. Non-invasive diagnostic techniques under investigation include “liquid biopsies” through a simple blood draw to develop predictive and prognostic biomarkers. A better understanding of circulating tumor cell (CTC) dissemination mechanisms offers promising potential for the development of techniques to assist in the diagnosis of lung cancer. Enumeration and characterization of CTCs has the potential to act as a prognostic biomarker and to identify novel drug targets for a precision medicine approach to lung cancer care. This review will focus on the current status of CTCs and their potential diagnostic and prognostic utility in this setting.
Introduction
Lung cancer is the leading cause of cancer-related mortality among men and women worldwide (1). In 2012, the incidence was estimated at 1.8 million new cases, accounting for 12.9% of all new cancers diagnosed globally (2). There is an estimated 18% survival rate beyond 5 years for all stages combined, with poor outcomes largely due to late diagnosis (1, 3). The majority of patients present with locally advanced or metastatic disease, with ~20–30% of patients presenting with early stage disease (3, 4). Late diagnosis is a major underlying cause for this advanced disease presentation (5). The annual mortality rate for lung cancer is higher than for colon, breast, and prostate cancers combined (6). The majority of patients presenting with advanced stage at diagnosis contributes to this poor outcome (4).
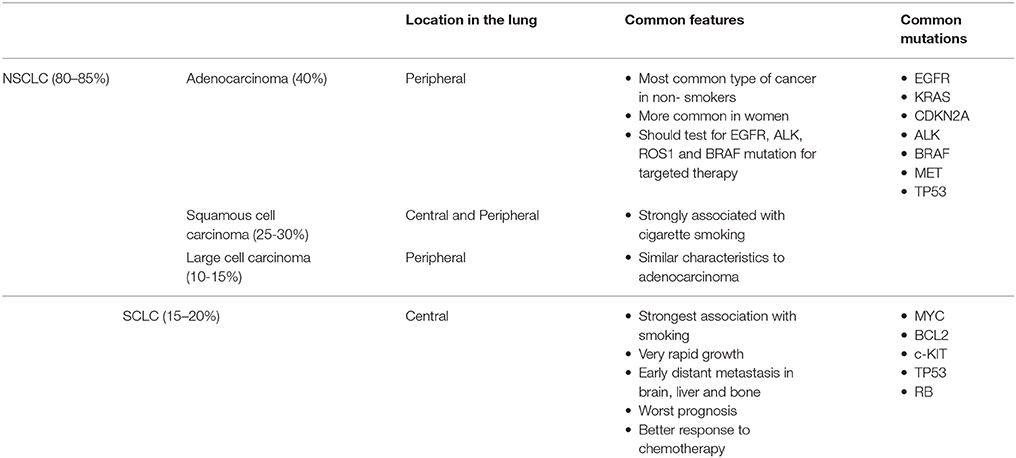
There are two main types of lung cancers, small cell lung carcinoma (SCLC) and non-small cell carcinoma (NSCLC). NSCLC is the most common, accounting for 80% of cases (7). NSCLC has three main histological subtypes: adenocarcinoma, squamous cell (epidermoid) carcinoma, and large cell undifferentiated carcinoma. Adenocarcinoma accounts for ~40% of cases although is increasing in relative incidence, and usually starts in mucus secreting epithelial cells (167). The prognosis of NSCLC subtypes depends on the stage of the tumor and the treatment availability.
Small cell lung cancer (SCLC) accounts for about 15% of all lung cancers diagnosed annually and up to 25% of lung cancer deaths. SCLC is characterized by a more aggressive clinical phenotype than NSCLC with progression to metastatic disease earlier in the disease course (8).
SCLC and NSCLC arise from different cell types and demonstrate varying clinical features as shown in Table 1.
Lung cancer may be initiated through exposure to carcinogens. The main risk factor for lung cancer is the use of tobacco. Tobacco is known to initiate and promote carcinogenesis and accounts for 85% of lung cancer cases (9). Additional known risks include exposure to pollutants such as asbestos, tar and metals including arsenic, and chromium. Common symptoms include persistent cough, worsening breathing, pneumonia that fails to resolve, chest discomfort, wheezing, blood in the sputum, and hoarseness (3, 10). A minority are asymptomatic, detected by chance through investigation of other illnesses or in screening programs (11).
Treatment options depend on the intent of treatment and may include loco-regional treatment such as surgery, image guided ablation including radical chemo-radiotherapy, stereotactic ablative radiation treatment, thermal ablation or cryotherapy, or systemic treatment such as chemotherapy, targeted agents, and immunotherapy, alongside novel agents under current investigation in clinical trials (11). An example of the power of targeted therapies in a precision medicine approach was demonstrated in 2004 by Lynch et al. (12) and Paez et al. (13) who demonstrated that patients with EGFR mutations present in the tumors of patients with non-small cell lung cancer exhibited a dramatic response to getfitinib, the epidermal growth factor (EGFR) tyrosine kinase inhibitor (TKI), bringing personalized medicine to reality for a subset of NSCLC patients (12, 13).
Utilization of expensive systemic targeted therapies, however, has traditionally required invasive biopsies in order to assess for targetable tumoral aberrations. This presents a challenge for the monitoring of lung cancers due to the requirement for longitudinal sampling of tumors (14).
Metastasis and Epithelial-Mesenchymal Transition
Metastasis is an extremely complex, multistep process. Cells must gain the ability to intravasate into the blood from the bulk tumor, travel through the blood undergoing sheer stressors and immune evasion, and extravasate to favorable metastatic sites such as bone, brain and liver (15–17). In order to detach from the primary tumor and disseminate into the blood, cells must undergo a cellular process known as epithelial-mesenchymal transition (EMT) (18). EMT enables tumor cells to become motile and enhances migratory capabilities which in effect allows cells to penetrate into the lymph vasculature and circulate as single or clusters of circulating tumor cells (CTCs) (19). Whilst in blood, CTCs exist in a dynamic EMT state (20). CTCs extravasate having undergone the reverse process known as mesenchymal to epithelial transition (MET) and colonize at distant organs, (21). EMT is thought to support cell invasiveness but restrict proliferation, thereby maintaining cancer cell survival in metastatic sites whereas MET re-activates proliferative potential (22). The famous “seed and soil” hypothesis proposed by Stephen Pagent in the Nineteenth century suggesting that tumor cells (the “seed”) have a preference to metastasize in certain organs (the ‘soil) (23). This hypothesis has since been revisited by Fidler and Langly, still holding significance in cancer research today (24, 25).
Circulating Tumor Cells in Lung Cancer
CTCs were first described by an Australian physician, Thomas Ashworth in 1869, where cancer cells in the blood were observed which resembled the cells of the primary tumor (26). CTCs play a central role in the metastatic spread of lung cancer, that is ultimately responsible for patient morbidity and mortality from the disease (27). While the concept of CTCs were described over one hundred years ago, it is only recently that they have been utilized in cancer diagnosis and prognosis (28).
Evidence has shown that the presence of CTCs in the blood correlates with poor overall survival in patients with metastatic prostate, breast and colon cancers (29–31). Patients with SCLC have on average 10 times more CTCs than patients with any other tumor type (32–34).
Molecular targeted therapies such as tyrosine kinase inhibitors (TKIs) in epidermal growth factor receptor (EGFR) mutants and anaplastic lymphoma kinase (ALK) inhibitors in ALK rearranged NSCLC patients have recently advanced the management of lung cancer for a limited proportion of patients (35–39). To determine eligibility for such targeted therapies, tumor biopsies have traditionally been necessary, increasing the likelihood of biopsy-related complications (40). Even in patients developing resistance to first line EGFR TKIs, liquid biopsies using circulating tumor DNA plasma only detect T790M mutations in ~80% of cases, particularly in low volume disease, making a repeat biopsy necessary. Tumor heterogeneity within the primary site or between primary and metastatic sites, can also create potential sampling bias, which may mask the true genetic profile of the cancer. The prospect of longitudinal sampling in order to monitor for the development of therapeutic resistance to treatments is likewise limited if invasive biopsies are essential (41, 42).
Use of CTCs as a liquid biopsy is promising for serial assessment of tumor evolution during the course of the disease and during systemic treatment in a less invasive, real-time manner, by a simple blood draw (19, 43). This liquid biopsy also provides potential for the early diagnosis of cancer and valuable insights into tumor heterogeneity and genomic diversity for the early diagnosis of cancer and guidance of clinical treatment (44, 45). A sensitive and unbiased isolation method to capture CTCs is therefore essential to provide tumoral material for analysis and potentially drive treatment decisions (46, 47).
Circulating Tumor Cell Detection Methods in Lung Cancer
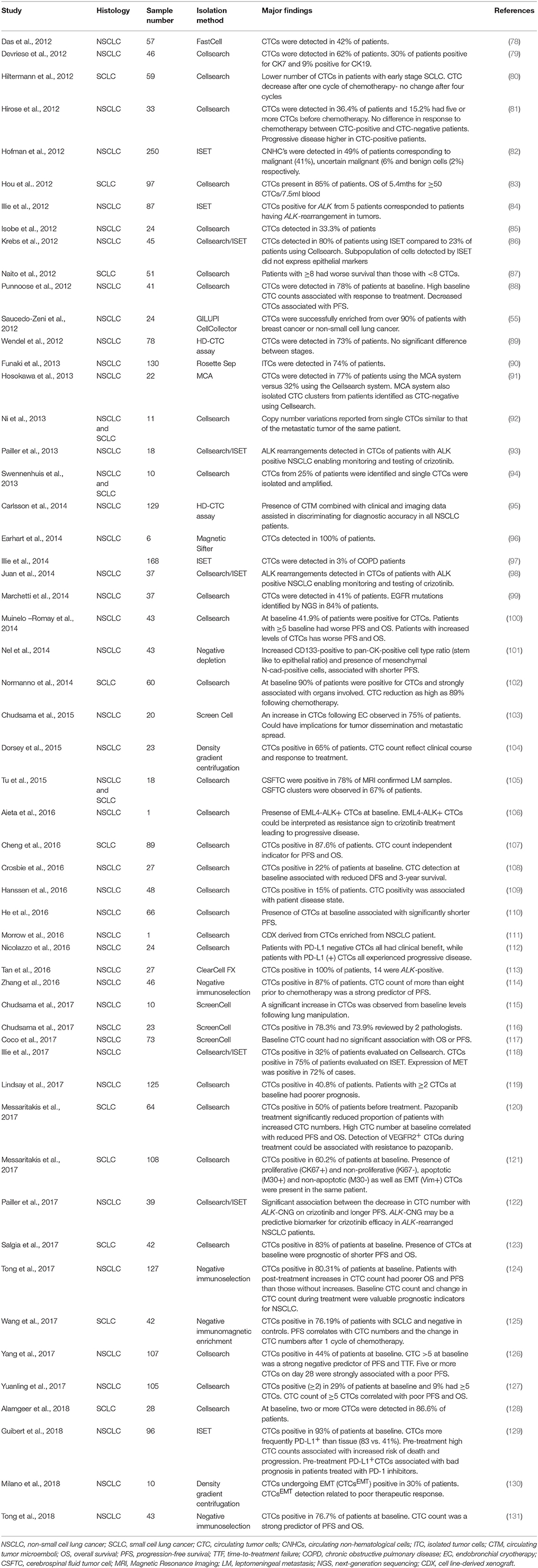
CTCs have the potential to accompany standard screening tests and be used for molecular characterization of a tumor (48). Detection of CTCs in NSCLC has been challenging due to the rarity in circulation (a few CTCs per billion normal blood cells) and the presence of non-epithelial characteristics (49). It is therefore imperative that sensitive and specific CTC detection methods are developed and optimized to assist in better patient monitoring and management (50–54). The advantages and disadvantages of the isolation methods in lung cancer are discussed and summarized in the Table 2. A summary of the CTC lung cancer studies are highlighted in Table 3.
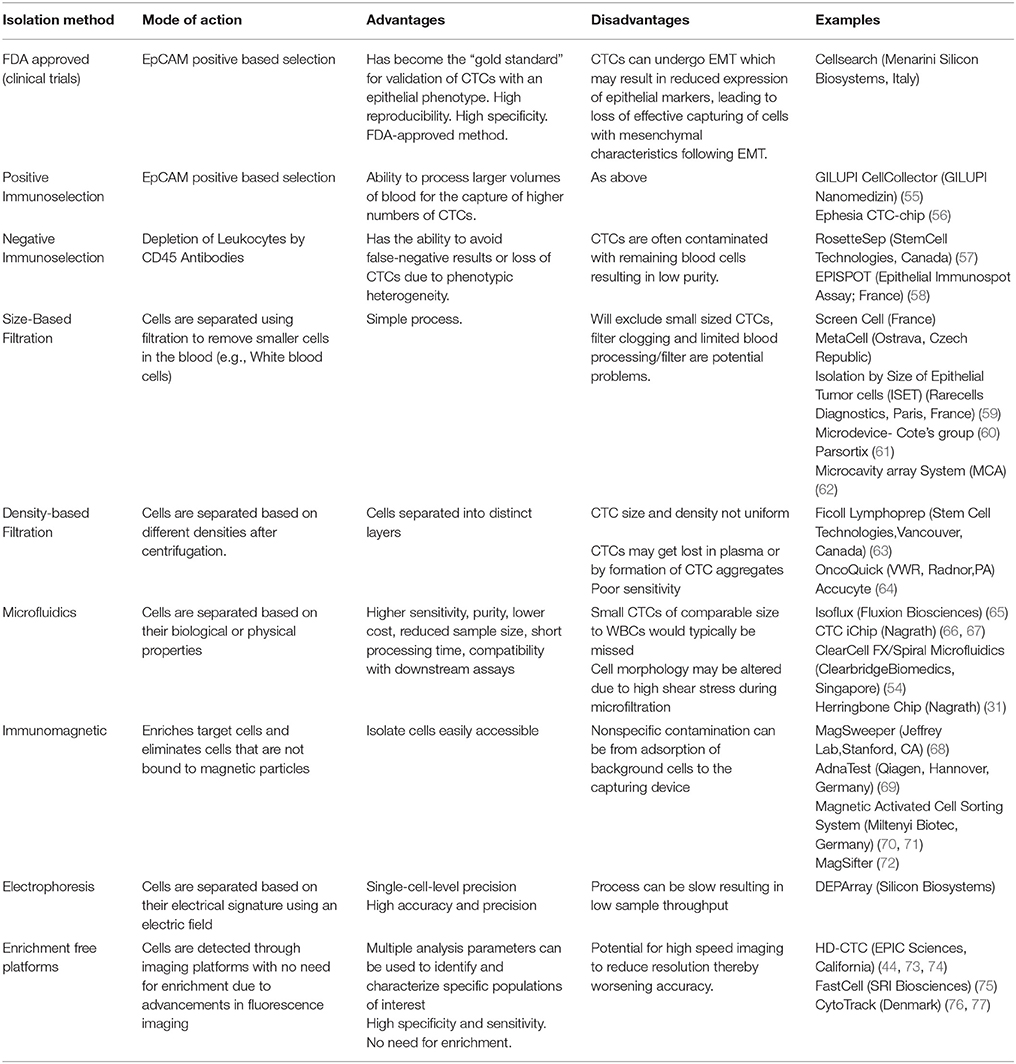
Table 2. The Summary of different Circulating Tumor Cell isolation methods currently used in research.
Ex-vivo Expansion of Circulating Tumor Cells
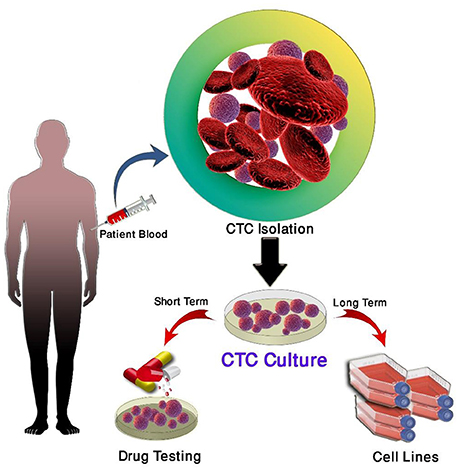
Despite limitations of current CTC isolation techniques, these cells have been detected in a number of cancers, including breast, head, and neck cancer, lung, prostate, colon and gastric cancer (21, 50, 53, 109, 132, 133–135). Successful ex-vivo culture of CTCs represents a “Holy Grail” in the study of cancer metastasis as it allows for in depth characterization of metastasis initiating cells as well as the testing of functional assays (136).
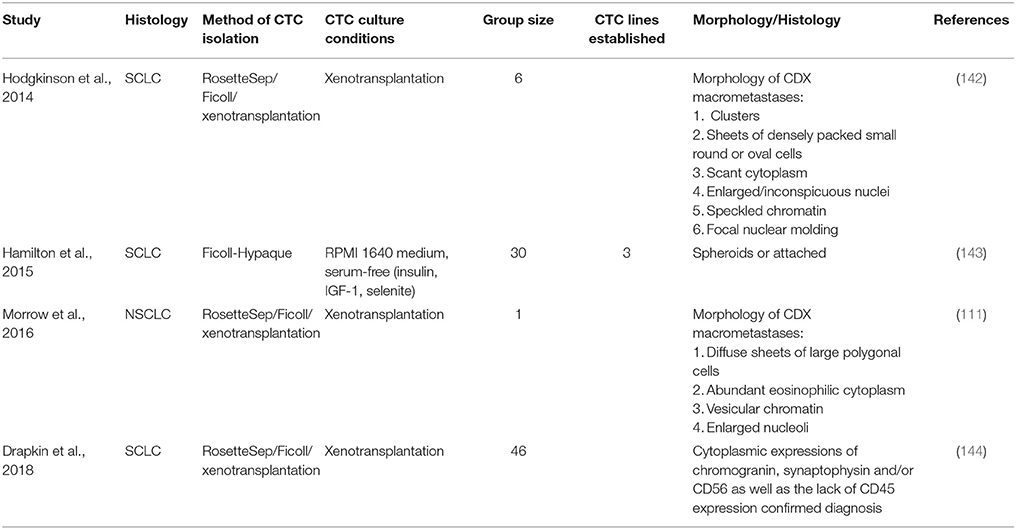
Short-term CTC culture (3–14 days) has been achieved in a number of cancer types, even from early stage cancers (137–139). This allows for the recapitulation of the disease in an ex vivo/in vivo setting for the testing of therapies and functional analysis (140). A summary of this is in Table 4. In comparison, long-term cultures have only been established in advanced metastatic cases where a large number of CTCs have been isolated (111, 142, 143) (Table 5). Long-term culture studies have shown that some CTCs in patient blood are immortalized and can be cultured ex vivo into stable cell lines (Figure 1) (139). There are only a few reports of successful long-term culture, notably, in patients with advanced stages of disease (136, 145, 146). CTC-expansion has been limited due to the influence of CTC enrichment. Certain cancers also require specific culture conditions for primary and metastatic samples (136). The successful culture of CTCs long-term holds great promise in developing personalized cancer treatment for testing of therapeutic efficacy using drug screening (140). This approach could assist in determining the choice of therapeutic regimen beneficial for patients and hence holds significance in advancement of precision medicine and personalized oncology (139).
Three main strategies are used for the propagation of CTCs in culture; two-dimensional (2D) culture, very commonly used for expansion of CTCs short-term, three-dimensional (3D) culture used for long-term expansion and xenotransplantation and four dimensional (4D) shown to mimic the process of metastasis (137, 147–150).
The expansion of CTCs in-vivo to generate patient derived xenografts (PDXs) may also be used to comprehensively analyse advanced disease biology and present a valuable model to understand cancer metastasis. The use of PDX's have been shown to mimic patient's disease and mirror response to chemotherapy (e.g., Platinum agents) (142, 151). However, PDXs have been challenging due to CTC heterogeneity causing unreliability of these models to translate clinically. PDX model development also takes 4–8 months and therefore are not optimal for rapid studies necessary for patients with advanced disease (151). In an ideal world cancer cell lines would be routinely generated from each cancer patient but this is not realistic at present (136, 139, 152).
Clinical Significance
The immediate need for early detection of lung cancer recurrence and monitoring treatment response is essential to facilitate improved survival of patients. Previous studies have shown computerized tomography (CT) screening has helped to reduce mortality, however CT has risks such as radiation exposure, leading to an increased risk of long-term cancer (153). This signifies the need for less invasive techniques for the early detection of metastasis and aid the personalized treatment of lung cancer. The use of CTCs as a liquid biopsy has the potential to accompany standard screening tests and also allow for molecular and genetic characterization of the tumor (48).
Enumeration of CTCs could provide a biomarker for cancer surveillance following treatment of early, locally advanced and advanced lung cancer and provided a better understanding on the mechanisms of metastasis (33). Although chemotherapy, targeted small molecules and immune checkpoint inhibitor therapies have shown significant benefits, the occurrence of acquired drug resistance and disease relapse are very common. Through serial sampling a longitudinal analysis of CTCs for identification of tumor evolution could provide valuable insights into mechanisms underlying resistance (154).
Detection of CTCs in lung cancer has been challenging, as CTCs usually present with non-epithelial characteristics (49). This emphasizes the need for more sensitive technologies to better capture CTCs for in-depth characterization and functional studies using cell culture and xenograft models. This will then ultimately assist in optimizing personalized therapies for lung cancer patients, with CTCs potentially being a prognostic biomarker.
Conclusion
The clinical significance of CTCs is yet to be established, however, advances in CTC detection and single-cell profiling have significantly improved our knowledge of underlying mechanisms of the evolution and dissemination of cancer and is progressively being translated to clinical studies. With lung cancer being the largest cause of cancer mortality worldwide, one of the biggest challenges for managing and treating patients is the lack of early screening/diagnostic methods (4). The isolation of CTCs from cerebrospinal fluid (CSF), may represent a unique subpopulation CTCs with ability to survive the journey in blood circulation and subsequent invasion of the CNS (105, 155). CTCs hold great promise as biomarkers for the early diagnosis and treatment selection of patients as well as broadening the current knowledge of metastasis (154).
Recurrence and progress of the disease, severity of symptoms and side-effects dramatically decrease patient's quality of life (QoL) (156). Therefore there is a vital need to monitor tumor evolution and understand mechanisms underlying development of therapeutic resistance.
Challenges for the field to address include the low sensitivity and specificity of current technologies prohibiting their use in current clinical settings, the large number of CTCs required for the development of CTC lines and patient xenografts for downstream functional analyses and the limited number of CTCs frequently found in patients with early stage disease (157). CTCs have demonstrated prognostic clinical utility is breast, lung and prostate cancers using the CellSearch technology (158, 159). Recent studies have demonstrated renewed interest in the FDA-approved Cellsearch platform for CTC PD-L1 analysis (160–162). These studies demonstrate how CTCs could be used to identify patients for anti PD-1/PD-L1 therapy (immunotherapy). Cellsearch relies on CTC enrichment using EpCAM (when CTCs undergo EMT, EpCAM is downregulated). As such the field is moving toward label-free technologies for CTC isolation. Currently, there are a number of technologies to enrich CTCs (i.e., Rarecyte, iChip, ISET, DEPArray, EPISPOT etc). The current label-free technologies are being validated for a number of cancers in larger clinical trials (163, 164). This is highlighted by the Cancer-ID network consortium in standardizing CTC/ctDNA and exosome isolation, analysis and reporting (165). The current gold standard in isolating CTCs from patient blood relies on the EpCAM status of these cells, thereby excluding a large majority of CTCs present in the blood of metastatic patients. Furthermore, Cellsearch does not allow for subsequent culture as the cells are fixed (166). CTCs as a liquid biopsy have valuable potential to improve early diagnosis, monitoring of disease, and direct treatment of lung cancer, however a better understanding of CTC biology is crucial for the field to move forward.
Author Contributions
JK, AK, KO, CP: Idea. JK, AK, MW: Preparation of figures and tables. All authors were involved in the preparation, review and editing of the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors would like to thank Mr. Sadegh Ghorbani for assisting in the schematic. This study was supported by the Queensland Centre for Head and Neck funded by Atlantic Philanthropies, the Queensland Government and the Translational Research Institute (TRI) Spore grant. QUT VC Fellowship for CP. QUT postgraduate research scholarship for JK.
References
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. (2017) 67:7–30. doi: 10.3322/caac.21387
2. Wong MCS, Lao XQ, Ho K-F, Goggins WB, Tse SLA. Incidence and mortality of lung cancer: global trends and association with socioeconomic status. Sci Rep. (2017) 7:14300. doi: 10.1038/s41598-017-14513-7
3. Insitute NNC. Cancer Stat Facts: Lung and Bronchus Cancer [Online]. NIH- National Cancer Institute (2017). Available online at: https://seer.cancer.gov/statfacts/html/lungb.html (Accessed July 17, 2017).
4. Ellis PM, Vandermeer R. Delays in the diagnosis of lung cancer. J Thorac Dis. (2011) 3:183–8. doi: 10.3978/j.issn.2072-1439.2011.01.01
5. Birring SS, Peake MD. Symptoms and the early diagnosis of lung cancer. Thorax (2005) 60:268–9. doi: 10.1136/thx.2004.032698
6. Malik PS, Raina V. Lung cancer: prevalent trends & emerging concepts. Indian J Med Res. (2015) 141:5–7.
7. Larsen JE, Minna JD. Molecular biology of lung cancer: clinical implications. Clin Chest Med. (2011) 32:703–40. doi: 10.1016/j.ccm.2011.08.003
8. Australia CC. Lung Cancer [Online] (2017). Available online at: http://www.cancer.org.au/about-cancer/types-of-cancer/lung-cancer.html#jump_9 (Accessed June 27, 2017).
9. Dela Cruz CS, Tanoue LT, Matthay RA. Lung cancer: epidemiology, etiology, and prevention. Clin Chest Med. (2011). 32:605–44. doi: 10.1016/j.ccm.2011.09.001
10. Cooley ME. Symptoms in adults with lung cancer: a systematic research review. J Pain Symptom Manage (2000) 19:137–53. doi: 10.1016/S0885-3924(99)00150-5
11. Institute NNC. Lung Cancer [Online]. (2017). Available online at: https://www.cancer.gov/types/lung/patient/non-small-cell-lung-treatment-pdq (Accessed July 17, 2017).
12. Lynch TJ, Bell DW, Sordella R, Gurubhagavatula S, Okimoto RA, Brannigan BW, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non–small-cell lung cancer to gefitinib. N Engl J Med. (2004) 350:2129–39. doi: 10.1056/NEJMoa040938
13. Paez JG, Jänne PA, Lee JC, Tracy S, Greulich H, Gabriel S, et al. Mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science (2004) 304:1497. doi: 10.1126/science.1099314
14. Hanssen A, Loges S, Pantel K, Wikman H. Detection of circulating tumor cells in non-small cell lung cancer. Front Oncol. (2015) 5:207. doi: 10.3389/fonc.2015.00207
15. Weiss L. Metastasis of cancer: a conceptual history from antiquity to the 1990s. Cancer Metastasis Rev. (2000) 19:I–XI, 193–383.
16. Hess KR, Varadhachary GR, Taylor SH, Wei W, Raber MN, Lenzi R, et al. Metastatic patterns in adenocarcinoma. Cancer (2006) 106:1624–33. doi: 10.1002/cncr.21778
17. Maheswaran S, Haber DA. Circulating tumor cells: a window into cancer biology and metastasis. Curr Opin Genet Dev. (2010) 20:96–9. doi: 10.1016/j.gde.2009.12.002
18. Thiery JP. Epithelial-mesenchymal transitions in tumour progression. Nat Rev Cancer (2002) 2:442–54. doi: 10.1038/nrc822
19. Nurwidya F, Zaini J, Putra AC, Andarini S, Hudoyo A, Syahruddin E, et al. Circulating tumor cell and cell-free circulating tumor DNA in lung cancer. Chonnam Med J. (2016) 52:151–8. doi: 10.4068/cmj.2016.52.3.151
20. Yu M, Bardia A, Wittner BS, Stott SL, Smas ME, Ting DT, et al. Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science (2013) 339:580–4. doi: 10.1126/science.1228522
21. Kulasinghe A, Kenny L, Perry C, Thiery J-P, Jovanovic L, Vela I, et al. Impact of label-free technologies in head and neck cancer circulating tumour cells. Oncotarget (2016) 7:71223–34. doi: 10.18632/oncotarget.12086.
22. Ocaña OH, Córcoles R, Fabra Á, Moreno-Bueno G, Acloque H, Vega S, et al. Metastatic colonization requires the repression of the epithelial-mesenchymal transition inducer Prrx1. Cancer Cell (2012) 22, 709–724. doi: 10.1016/j.ccr.2012.10.012
23. Paget S. The distribution of secondary growths in cancer of the breast. 1889. Cancer Metastasis Rev. (1989) 8:98–101.
24. Fidler IJ. The pathogenesis of cancer metastasis: theseed and soil hypothesis revisited. Nat Rev Cancer (2003) 3:453. doi: 10.1038/nrc1098
25. Langley RR, Fidler IJ. The seed and soil hypothesis revisited–the role of tumor-stroma interactions in metastasis to different organs. Int J Cancer (2011) 128:2527–35. doi: 10.1002/ijc.26031
26. Ashworth TR. A case of cancer in which cells similar to those in the tumours were seen in the blood after death. Med J Aust. (1869) 14:146–7.
27. Mehlen P, Puisieux A. Metastasis: a question of life or death. Nat Rev Cancer (2006) 6:449–58. doi: 10.1038/nrc1886
28. Hong Y, Fang F, Zhang Q. Circulating tumor cell clusters: what we know and what we expect (Review). Int J Oncol. (2016) 49:2206–16. doi: 10.3892/ijo.2016.3747
29. Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Miller MC, et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med. (2004) 351:781–91. doi: 10.1056/NEJMoa040766
30. Scher HI, Jia X, de Bono JS, Fleisher M, Pienta KJ, Raghavan D, et al. Circulating tumour cells as prognostic markers in progressive, castration-resistant prostate cancer: a reanalysis of IMMC38 trial data. Lancet Oncol. (2009) 10:233–9. doi: 10.1016/s1470-2045(08)70340-1
31. Stott SL, Hsu CH, Tsukrov DI, Yu M, Miyamoto DT, Waltman BA, et al. Isolation of circulating tumor cells using a microvortex-generating herringbone-chip. Proc Natl Acad Sci USA. (2010) 107:18392–7. doi: 10.1073/pnas.1012539107
32. Tanaka F, Yoneda K, Hasegawa S. Circulating tumor cells (CTCs) in lung cancer: current status and future perspectives. Lung Cancer (2010) 1:77–84.
33. Krebs MG, Sloane R, Priest L, Lancashire L, Hou JM, Greystoke A, et al. Evaluation and prognostic significance of circulating tumor cells in patients with non-small-cell lung cancer. J Clin Oncol. (2011) 29:1556–63. doi: 10.1200/JCO.2010.28.7045
34. Alix-Panabieres C, Pantel K. Characterization of single circulating tumor cells. FEBS Lett. (2017) 591:2241–50. doi: 10.1002/1873-3468.12662
35. Crinò L, Kim D, Riely GJ, Janne PA, Blackhall FH, Camidge DR, et al. Initial phase II results with crizotinib in advanced ALK-positive non-small cell lung cancer (NSCLC): PROFILE 1005. J Clin Oncol. (2011) 29(15_suppl.):7514. doi: 10.1200/jco.2011.29.15_suppl.7514
36. Camidge DR, Bang Y-J, Kwak EL, Iafrate AJ, Varella-Garcia M, Fox SB, et al. Activity and safety of crizotinib in patients with ALK-positive non-small-cell lung cancer: updated results from a phase 1 study. Lancet Oncol. (2012) 13:1011–9. doi: 10.1016/S1470-2045(12)70344-3
37. Rosell R, Carcereny E, Gervais R, Vergnenegre A, Massuti B, Felip E, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. (2012) 13:239–46. doi: 10.1016/s1470-2045(11)70393-x
38. Shaw AT, Kim DW, Nakagawa K, Seto T, Crinó L, Ahn MJ, et al. Crizotinib versus Chemotherapy in advanced ALK-positive lung cancer. N Engl J Med. (2013) 368:2385–94. doi: 10.1056/NEJMoa1214886
39. Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa K, Mekhail T, et al. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N Engl J Med. (2014) 371:2167–77. doi: 10.1056/NEJMoa1408440
40. Lindeman NI, Cagle PT, Beasley MB, Chitale DA, Dacic S, Giaccone G, et al. Molecular testing guideline for selection of lung cancer patients for EGFR and ALK tyrosine kinase inhibitors: guideline from the College of American Pathologists, International Association for the Study of Lung Cancer, and Association for Molecular Pathology. J Thorac Oncol. (2013) 8:823–59. doi: 10.1097/JTO.0b013e318290868f
41. Gerlinger M, Rowan AJ, Horswell S, Larkin J, Endesfelder D, Gronroos E, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. (2012) 366:883–92. doi: 10.1056/NEJMoa1113205
42. Moreira AL, Thornton RH. Personalized medicine for non–small-cell lung cancer: implications of recent advances in tissue acquisition for molecular and histologic testing. Clin Lung Cancer (2012) 13:334–9. doi: 10.1016/j.cllc.2012.01.004
43. Alix-Panabieres C, Pantel K. Clinical applications of circulating tumor cells and circulating tumor DNA as liquid biopsy. Cancer Discov. (2016) 6:479–91. doi: 10.1158/2159-8290.CD-15-1483
44. Kuhn P, Bethel K. A fluid biopsy as investigating technology for the fluid phase of solid tumors. Phys Biol. (2012) 9:010301. doi: 10.1088/1478-3975/9/1/010301
45. Alberter B, Klein CA, Polzer B. Single-cell analysis of CTCs with diagnostic precision: opportunities and challenges for personalized medicine. Expert Rev Mol Diagn. (2016) 16:25–38. doi: 10.1586/14737159.2016.1121099
46. Alix-Panabieres C, Pantel K. Circulating tumor cells: liquid biopsy of cancer. Clin Chem. (2013) 59:110–8. doi: 10.1373/clinchem.2012.194258
47. Alix-Panabieres C, Pantel K. Challenges in circulating tumour cell research. Nat Rev Cancer (2014) 14:623–31. doi: 10.1038/nrc3820
48. Murlidhar V, Ramnath N, Nagrath S, Reddy RM. Optimizing the detection of circulating markers to aid in early lung cancer detection. Cancers (2016) 8:61. doi: 10.3390/cancers8070061
49. Lianidou ES, Markou A, Strati A. The role of CTCs as tumor biomarkers. Adv Exp Med Biol. (2015) 867:341–67. doi: 10.1007/978-94-017-7215-0_21
50. Yu M, Stott S, Toner M, Maheswaran S, Haber DA. Circulating tumor cells: approaches to isolation and characterization. J Cell Biol. (2011) 192:373–82. doi: 10.1083/jcb.201010021
51. Harouaka R, Kang Z, Zheng SY, Cao L. Circulating tumor cells: advances in isolation and analysis, and challenges for clinical applications. Pharmacol Ther. (2014) 141:209–21. doi: 10.1016/j.pharmthera.2013.10.004
52. Caceres G, Puskas JA, Magliocco AM. Circulating tumor cells: a window into tumor development and therapeutic effectiveness. Cancer Control. (2015) 22:167–76. doi: 10.1177/107327481502200207
53. Yang MH, Imrali A, Heeschen C. Circulating cancer stem cells: the importance to select. Chin J Cancer Res. (2015) 27:437–49. doi: 10.3978/j.issn.1000-9604.2015.04.08
54. Warkiani ME, Khoo BL, Wu L, Tay AK, Bhagat AA, Han J, et al. Ultra-fast, label-free isolation of circulating tumor cells from blood using spiral microfluidics. Nat Protoc. (2016) 11:134–48. doi: 10.1038/nprot.2016.003
55. Saucedo-Zeni N, Mewes S, Niestroj R, Gasiorowski L, Murawa D, Nowaczyk P, et al. A novel method for the in vivo isolation of circulating tumor cells from peripheral blood of cancer patients using a functionalized and structured medical wire. Int J Oncol. (2012) 41:1241–50. doi: 10.3892/ijo.2012.1557
56. Saliba A-E, Saias L, Psychari E, Minc N, Simon D, Bidard F-C, et al. Microfluidic sorting and multimodal typing of cancer cells in self-assembled magnetic arrays. Proc Natl Acad Sci USA. (2010) 107:14524–9. doi: 10.1073/pnas.1001515107
57. He W, Kularatne SA, Kalli KR, Prendergast FG, Amato RJ, Klee GG, et al. Quantitation of circulating tumor cells in blood samples from ovarian and prostate cancer patients using tumor-specific fluorescent ligands. Int J Cancer (2008) 123:1968–73. doi: 10.1002/ijc.23717
58. Alix-Panabieres C. EPISPOT assay: detection of viable DTCs/CTCs in solid tumor patients. Recent Results Cancer Res. (2012) 195:69–76. doi: 10.1007/978-3-642-28160-0_6
59. Hofman V, Bonnetaud C, Ilie MI, Vielh P, Vignaud JM, Flejou JF, et al. Preoperative circulating tumor cell detection using the isolation by size of epithelial tumor cell method for patients with lung cancer is a new prognostic biomarker. Clin Cancer Res. (2011) 17:827–35. doi: 10.1158/1078-0432.CCR-10-0445
60. Lin HK, Zheng S, Williams AJ, Balic M, Groshen S, Scher HI, et al. Portable filter-based microdevice for detection and characterization of circulating tumor cells. Clin Cancer Res. (2010) 16:5011–8. doi: 10.1158/1078-0432.ccr-10-1105
61. Obermayr E, Maritschnegg E, Speiser P, Singer C, Schuster EM, Danzinger S, et al. Circulating rare cells enable highly efficient cancer detection. Cancer Res. (2015) 75(15 Suppl.). doi: 10.1158/1538-7445.am2015-lb-197.
62. Hosokawa M, Yoshikawa T, Negishi R, Yoshino T, Koh Y, Kenmotsu H, et al. Microcavity array system for size-based enrichment of circulating tumor cells from the blood of patients with small-cell lung cancer. Anal Chem. (2013) 85:5692–8. doi: 10.1021/ac400167x
63. Weitz J, Kienle P, Lacroix J, Willeke F, Benner A, Lehnert T, et al. Dissemination of tumor cells in patients undergoing surgery for colorectal cancer. Clin Cancer Res. (1998) 4:343–8.
64. Campton DE, Ramirez AB, Nordberg JJ, Drovetto N, Clein AC, Varshavskaya P, et al. High-recovery visual identification and single-cell retrieval of circulating tumor cells for genomic analysis using a dual-technology platform integrated with automated immunofluorescence staining. BMC Cancer (2015) 15:360. doi: 10.1186/s12885-015-1383-x
65. Harb W, Fan A, Tran T, Danila DC, Keys D, Schwartz M, et al. Mutational analysis of circulating tumor cells using a novel microfluidic collection device and qPCR assay. Transl Oncol. (2013) 6:528–38. doi: 10.1593/tlo.13367
66. Nagrath S, Sequist LV, Maheswaran S, Bell DW, Irimia D, Ulkus L, et al. Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature (2007) 450:1235–9. doi: 10.1038/nature06385
67. Sequist LV, Nagrath S, Toner M, Haber DA, Lynch TJ. The CTC-chip: an exciting new tool to detect circulating tumor cells in lung cancer patients. J Thorac Oncol. (2009) 4:281–3. doi: 10.1097/JTO.0b013e3181989565
68. Talasaz AH, Powell AA, Huber DE, Berbee JG, Roh K-H, Yu W, et al. Isolating highly enriched populations of circulating epithelial cells and other rare cells from blood using a magnetic sweeper device. Proc Natl Acad Sci USA. (2009) 106:3970–5. doi: 10.1073/pnas.0813188106
69. Zieglschmid V, Hollmann C, Gutierrez B, Albert W, Strothoff D, Gross E, et al. Combination of immunomagnetic enrichment with multiplex RT-PCR analysis for the detection of disseminated tumor cells. Anticancer Res. (2005) 25:1803–10.
70. Miltenyi S, Muller W, Weichel W, Radbruch A. High gradient magnetic cell separation with MACS. Cytometry (1990) 11:231–8. doi: 10.1002/cyto.990110203
71. Mayo C, Ortega FG, Giménez-Capitán A, Molina-Vila MA, Serrano MJ, Viteri S, et al. CK-coated magnetic-based beads as a tool to isolate circulating tumor cells (CTCs) in human tumors. Transl Lung Cancer Res. (2013) 2:65–71. doi: 10.3978/j.issn.2218-6751.2013.02.06
72. Earhart CM, Hughes CE, Gaster RS, Ooi CC, Wilson RJ, Zhou LY, et al. Isolation and mutational analysis of circulating tumor cells from lung cancer patients with magnetic sifters and biochips. Lab Chip. (2014) 14:78–88. doi: 10.1039/c3lc50580d
73. Bazhenova L, Nieva JJ, Kolatkar A, Luttgen M, Marinucci D, Bethel K, et al. Performance of the high-definition circulating tumor cells (HD-CTC) assay in patients with non-small cell lung cancer (NSCLC). J Clin Oncol. (2012) 30(15_suppl):e21074. doi: 10.1200/jco.2012.30.15_suppl.e21074
74. Marrinucci D, Bethel K, Kolatkar A, Luttgen MS, Malchiodi M, Baehring F, et al. Fluid biopsy in patients with metastatic prostate, pancreatic and breast cancers. Phys Biol. (2012) 9:016003. doi: 10.1088/1478-3975/9/1/016003
75. Krivacic RT, Ladanyi A, Curry DN, Hsieh HB, Kuhn P, Bergsrud DE, et al. A rare-cell detector for cancer. Proc Natl Acad Sci USA. (2004) 101:10501–4. doi: 10.1073/pnas.0404036101
76. Hillig T, Horn P, Nygaard AB, Haugaard AS, Nejlund S, Brandslund I, et al. In vitro detection of circulating tumor cells compared by the CytoTrack and CellSearch methods. Tumour Biol. (2015) 36:4597–601. doi: 10.1007/s13277-015-3105-z
77. Ferreira MM, Ramani VC, Jeffrey SS. Circulating tumor cell technologies. Mol Oncol. (2016) 10:374–94. doi: 10.1016/j.molonc.2016.01.007
78. Das M, Riess JW, Frankel P, Schwartz E, Bennis R, Hsieh HB, et al. ERCC1 expression in circulating tumor cells (CTCs) using a novel detection platform correlates with progression-free survival (PFS) in patients with metastatic non-small-cell lung cancer (NSCLC) receiving platinum chemotherapy. Lung Cancer (2012) 77:421–6. doi: 10.1016/j.lungcan.2012.04.005
79. Devriese LA, Bosma AJ, van de Heuvel MM, Heemsbergen W, Voest EE, Schellens JHM. Circulating tumor cell detection in advanced non-small cell lung cancer patients by multi-marker QPCR analysis. Lung Cancer (2012) 75:242–7. doi: 10.1016/j.lungcan.2011.07.003
80. Hiltermann TJ, Pore MM, van den Berg A, Timens W, Boezen HM, Liesker JJ, et al. Circulating tumor cells in small-cell lung cancer: a predictive and prognostic factor. Ann Oncol. (2012) 23:2937–42. doi: 10.1093/annonc/mds138
81. Hirose T, Murata Y, Oki Y, Sugiyama T, Kusumoto S, Ishida H, et al. Relationship of circulating tumor cells to the effectiveness of cytotoxic chemotherapy in patients with metastatic non-small-cell lung cancer. Oncol Res. (2012) 20:131–7. doi: 10.3727/096504012X13473664562583
82. Hofman V, Long E, Ilie M, Bonnetaud C, Vignaud JM, Flejou JF, et al. Morphological analysis of circulating tumour cells in patients undergoing surgery for non-small cell lung carcinoma using the isolation by size of epithelial tumour cell (ISET) method. Cytopathology (2012) 23:30–8. doi: 10.1111/j.1365-2303.2010.00835.x
83. Hou JM, Krebs MG, Lancashire L, Sloane R, Backen A, Swain RK, et al. Clinical significance and molecular characteristics of circulating tumor cells and circulating tumor microemboli in patients with small-cell lung cancer. J Clin Oncol. (2012) 30:525–32. doi: 10.1200/JCO.2010.33.3716
84. Ilie M, Long E, Butori C, Hofman V, Coelle C, Mauro V, et al. ALK-gene rearrangement: a comparative analysis on circulating tumour cells and tumour tissue from patients with lung adenocarcinoma. Ann Oncol. (2012) 23:2907–13. doi: 10.1093/annonc/mds137
85. Isobe K, Hata Y, Kobayashi K, Hirota N, Sato K, Sano G, et al. Clinical significance of circulating tumor cells and free DNA in non-small cell lung cancer. Anticancer Res. (2012) 32:3339–44.
86. Krebs MG, Hou J-M, Sloane R, Lancashire L, Priest L, Nonaka D, et al. Analysis of circulating tumor cells in patients with non-small cell lung cancer using epithelial marker-dependent and -independent approaches. J Thorac Oncol. (2012) 7:306–15. doi: 10.1097/JTO.0b013e31823c5c16
87. Naito T, Tanaka F, Ono A, Yoneda K, Takahashi T, Murakami H, et al. Prognostic impact of circulating tumor cells in patients with small cell lung cancer. J Thorac Oncol. (2012) 7:512–9. doi: 10.1097/JTO.0b013e31823f125d
88. Punnoose EA, Atwal S, Liu W, Raja R, Fine BM, Hughes BG, et al. Evaluation of circulating tumor cells and circulating tumor DNA in non-small cell lung cancer: association with clinical endpoints in a phase II clinical trial of pertuzumab and erlotinib. Clin Cancer Res. (2012) 18:2391–401. doi: 10.1158/1078-0432.CCR-11-3148
89. Wendel M, Bazhenova L, Boshuizen R, Kolatkar A, Honnatti M, Cho EH, et al. Fluid biopsy for circulating tumor cell identification in patients with early and late stage non-small cell lung cancer; a glimpse into lung cancer biology. Phys Biol. (2012) 9:016005. doi: 10.1088/1478-3967/9/1/016005
90. Funaki S, Sawabata N, Abulaiti A, Nakagiri T, Shintani Y, Inoue M, et al. Significance of tumour vessel invasion in determining the morphology of isolated tumour cells in the pulmonary vein in non-small-cell lung cancer. Eur J Cardiothorac Surg. (2013) 43:1126–30. doi: 10.1093/ejcts/ezs553
91. Hosokawa M, Kenmotsu H, Koh Y, Yoshino T, Yoshikawa T, Naito T, et al. Size-based isolation of circulating tumor cells in lung cancer patients using a microcavity array system. PLoS ONE (2013) 8:e67466. doi: 10.1371/journal.pone.0067466
92. Ni X, Zhuo M, Su Z, Duan J, Gao Y, Wang Z, et al. Reproducible copy number variation patterns among single circulating tumor cells of lung cancer patients. Proc Natl Acad Sci USA. (2013) 110:21083–8. doi: 10.1073/pnas.1320659110
93. Pailler E, Adam J, Barthelemy A, Oulhen M, Auger N, Valent A, et al. Detection of circulating tumor cells harboring a unique ALK rearrangement in ALK-positive non-small-cell lung cancer. J Clin Oncol. (2013) 31:2273–81. doi: 10.1200/JCO.2012.44.5932
94. Swennenhuis JF, Reumers J, Thys K, Aerssens J, Terstappen LW. Efficiency of whole genome amplification of single circulating tumor cells enriched by CellSearch and sorted by FACS. Genome Med. (2013) 5:106. doi: 10.1186/gm510
95. Carlsson A, Nair VS, Luttgen MS, Keu KV, Horng G, Vasanawala M, et al. Circulating tumor microemboli diagnostics for patients with non-small-cell lung cancer. J Thorac Oncol. (2014) 9:1111–9. doi: 10.1097/JTO.0000000000000235
96. Earhart CM, Hughes CE, Gaster RS, Ooi CC, Wilson RJ, Zhou LY, et al. Isolation and mutational analysis of circulating tumor cells from lung cancer patients with magnetic sifters and biochips. Lab Chip (2014) 14:78–88. doi: 10.1039/C3LC50580D
97. Ilie M, Hofman V, Long-Mira E, Selva E, Vignaud JM, Padovani B, et al. “Sentinel” circulating tumor cells allow early diagnosis of lung cancer in patients with chronic obstructive pulmonary disease. PLoS ONE (2014) 9:e111597. doi: 10.1371/journal.pone.0111597
98. Juan O, Vidal J, Gisbert R, Munoz J, Macia S, Gomez-Codina J. Prognostic significance of circulating tumor cells in advanced non-small cell lung cancer patients treated with docetaxel and gemcitabine. Clin Transl Oncol. (2014) 16:637–43. doi: 10.1007/s12094-013-1128-8
99. Marchetti A, Del Grammastro M, Felicioni L, Malatesta S, Filice G, Centi I, et al. Assessment of EGFR mutations in circulating tumor cell preparations from NSCLC patients by next generation sequencing: toward a real-time liquid biopsy for treatment. PLoS ONE (2014) 9:e103883. doi: 10.1371/journal.pone.0103883
100. Muinelo-Romay L, Vieito M, Abalo A, Nocelo MA, Baron F, Anido U, et al. Evaluation of circulating tumor cells and related events as prognostic factors and surrogate biomarkers in advanced NSCLC patients receiving first-line systemic treatment. Cancers (2014) 6:153–65. doi: 10.3390/cancers6010153
101. Nel I, Jehn U, Gauler T, Hoffmann AC. Individual profiling of circulating tumor cell composition in patients with non-small cell lung cancer receiving platinum based treatment. Transl Lung Cancer Res. (2014) 3:100–6. doi: 10.3978/j.issn.2218-6751.2014.03.05
102. Normanno N, Rossi A, Morabito A, Signoriello S, Bevilacqua S, Di Maio M, et al. Prognostic value of circulating tumor cells' reduction in patients with extensive small-cell lung cancer. Lung Cancer (2014) 85:314–9. doi: 10.1016/j.lungcan.2014.05.002
103. Chudasama D, Rice A, Soppa G, Anikin V. Circulating tumour cells in patients with lung cancer undergoing endobronchial cryotherapy. Cryobiology (2015) 71:161–3. doi: 10.1016/j.cryobiol.2015.06.001
104. Dorsey JF, Kao GD, MacArthur KM, Ju M, Steinmetz D, Wileyto EP, et al. Tracking viable circulating tumor cells (CTCs) in the peripheral blood of non-small cell lung cancer patients undergoing definitive radiation therapy: pilot study results. Cancer (2015) 121:139–49. doi: 10.1002/cncr.28975
105. Tu Q, Wu X, Le Rhun E, Blonski M, Wittwer B, Taillandier L, et al. CellSearch technology applied to the detection and quantification of tumor cells in CSF of patients with lung cancer leptomeningeal metastasis. Lung Cancer (2015) 90:352–7. doi: 10.1016/j.lungcan.2015.09.008
106. Aieta M, Facchinetti A, De Faveri S, Manicone M, Tartarone A, Possidente L, et al. Monitoring and characterization of circulating tumor cells (ctCs) in a patient with EML4-ALK–positive non–small cell lung cancer (NSCLC). Clin Lung Cancer (2016) 17:e173–7. doi: 10.1016/j.cllc.2016.05.002
107. Cheng Y, Liu XQ, Fan Y, Liu YP, Liu Y, Liu Y, et al. Circulating tumor cell counts/change for outcome prediction in patients with extensive-stage small-cell lung cancer. Future Oncol. (2016) 12:789–99. doi: 10.2217/fon.15.346
108. Crosbie PAJ, Shah R, Krysiak P, Zhou C, Morris K, Tugwood J, et al. Circulating tumor cells detected in the tumor-draining pulmonary vein are associated with disease recurrence after surgical resection of NSCLC. J Thorac Oncol. (2016) 11:1793–7. doi: 10.1016/j.jtho.2016.06.017
109. Hanssen A, Wagner J, Gorges TM, Taenzer A, Uzunoglu FG, Driemel C, et al. Characterization of different CTC subpopulations in non-small cell lung cancer. Sci Rep. (2016) 6:28010. doi: 10.1038/srep28010
110. Zhou J, Dong F, Cui F, Xu R, Tang X. The role of circulating tumor cells in evaluation of prognosis and treatment response in advanced non-small-cell lung cancer. Cancer Chemother Pharmacol. (2017) 79:825–33. doi: 10.1007/s00280-017-3269-x
111. Morrow CJ, Trapani F, Metcalf RL, Bertolini G, Hodgkinson CL, Khandelwal G, et al. Tumourigenic non-small-cell lung cancer mesenchymal circulating tumour cells: a clinical case study. Ann Oncol. (2016) 27:1155–60. doi: 10.1093/annonc/mdw122
112. Nicolazzo C, Raimondi C, Mancini M, Caponnetto S, Gradilone A, Gandini O, et al. Monitoring PD-L1 positive circulating tumor cells in non-small cell lung cancer patients treated with the PD-1 inhibitor Nivolumab. Sci Rep. (2016) 6:31726. doi: 10.1038/srep31726
113. Tan CL, Lim TH, Lim TKH, Tan DS-W, Chua YW, Ang MK, et al. Concordance of anaplastic lymphoma kinase (ALK) gene rearrangements between circulating tumor cells and tumor in non-small cell lung cancer. Oncotarget (2016) 7:23251–62. doi: 10.18632/oncotarget.8136
114. Zhang Z, Xiao Y, Zhao J, Chen M, Xu Y, Zhong W, et al. Relationship between circulating tumour cell count and prognosis following chemotherapy in patients with advanced non-small-cell lung cancer. Respirology (2016) 21:519–25. doi: 10.1111/resp.12696
115. Chudasama D, Burnside N, Beeson J, Karteris E, Rice A, Anikin V. Perioperative detection of circulating tumour cells in patients with lung cancer. Oncol Lett. (2017) 14:1281–6. doi: 10.3892/ol.2017.6366.
116. Chudasama D, Barr J, Beeson J, Beddow E, Mcgonigle N, Rice A, et al. Detection of circulating tumour cells and survival of patients with non-small cell lung cancer. Anticancer Res. (2017) 37:169–73. doi: 10.21873/anticanres.11302
117. Coco S, Alama A, Vanni I, Fontana V, Genova C, Dal Bello MG, et al. Circulating cell-free DNA and circulating tumor cells as prognostic and predictive biomarkers in advanced non-small cell lung cancer patients treated with first-line chemotherapy. Int J Mol Sci. (2017) 18:1035. doi: 10.3390/ijms18051035
118. Ilie M, Szafer-Glusman E, Hofman V, Long-Mira E, Suttmann R, Darbonne W, et al. Expression of MET in circulating tumor cells correlates with expression in tumor tissue from advanced-stage lung cancer patients. Oncotarget (2017) 8:26112–21. doi: 10.18632/oncotarget.15345
119. Lindsay CR, Faugeroux V, Michiels S, Pailler E, Facchinetti F, Ou D, et al. A prospective examination of circulating tumor cell profiles in non-small-cell lung cancer molecular subgroups. Ann Oncol. (2017) 28:1523–31. doi: 10.1093/annonc/mdx156
120. Messaritakis I, Politaki E, Plataki M, Karavassilis V, Kentepozidis N, Koinis F, et al. Heterogeneity of circulating tumor cells (CTCs) in patients with recurrent small cell lung cancer (SCLC) treated with pazopanib. Lung Cancer (2017) 104:16–23. doi: 10.1016/j.lungcan.2016.12.008
121. Messaritakis I, Politaki E, Kotsakis A, Dermitzaki E-K, Koinis F, Lagoudaki E, et al. Phenotypic characterization of circulating tumor cells in the peripheral blood of patients with small cell lung cancer. PLoS ONE (2017) 12:e0181211. doi: 10.1371/journal.pone.0181211
122. Pailler E, Oulhen M, Borget I, Remon J, Ross K, Auger N, et al. Circulating tumor cells with aberrant ALK copy number predict progression-free survival during crizotinib treatment in ALK-rearranged non-small cell lung cancer patients. Cancer Res. (2017) 77:2222–30. doi: 10.1158/0008-5472.can-16-3072
123. Salgia R, Weaver RW, McCleod M, Stille JR, Yan SB, Roberson S, et al. Prognostic and predictive value of circulating tumor cells and CXCR4 expression as biomarkers for a CXCR4 peptide antagonist in combination with carboplatin-etoposide in small cell lung cancer: exploratory analysis of a phase II study. Invest New Drugs (2017) 35:334–44. doi: 10.1007/s10637-017-0446-z
124. Tong B, Xu Y, Zhao J, Chen M, Xing J, Zhong W, et al. Prognostic significance of circulating tumor cells in non-small cell lung cancer patients undergoing chemotherapy. Oncotarget (2017) 8:86615–24. doi: 10.18632/oncotarget.21255
125. Wang YL, Liu CH, Li J, Ma XP, Gong P. Clinical significance of circulating tumor cells in patients with small-cell lung cancer. Tumori J. (2017) 103:242–8. doi: 10.5301/tj.5000601
126. Yang B, Qin A, Zhang K, Ren H, Liu S, Liu X, et al. Circulating tumor cells predict prognosis following tyrosine kinase inhibitor treatment in EGFR-mutant non-small cell lung cancer patients. Oncol Res. (2017) 25:1601–6. doi: 10.3727/096504017X14928634401178
127. Qi Y, Wang W. Clinical significance of circulating tumor cells in squamous cell lung cancer patients. Cancer Biomark (2017) 18:161–7. doi: 10.3233/cbm-160090
128. Alamgeer M, Neil Watkins D, Banakh I, Kumar B, Gough DJ, Markman B, et al. A phase IIa study of HA-irinotecan, formulation of hyaluronic acid and irinotecan targeting CD44 in extensive-stage small cell lung cancer. Investig New Drugs (2018) 36:288–98. doi: 10.1007/s10637-017-0555-8
129. Guibert N, Delaunay M, Lusque A, Boubekeur N, Rouquette I, Clermont E, et al. PD-L1 expression in circulating tumor cells of advanced non-small cell lung cancer patients treated with nivolumab. Lung Cancer (2018) 120:108–12. doi: 10.1016/j.lungcan.2018.04.001
130. Milano A, Mazzetta F, Valente S, Ranieri D, Leone L, Botticelli A, et al. Molecular detection of EMT markers in circulating tumor cells from metastatic non-small cell lung cancer patients: potential role in clinical practice. Anal Cell Pathol. (2018) 2018:3506874. doi: 10.1155/2018/3506874
131. Tong B, Xu Y, Zhao J, Chen M, Zhong W, Xing J, et al. Prognostic role of circulating tumor cells in patients with EGFR-mutated or ALK-rearranged non-small cell lung cancer. Thorac Cancer (2018) 9:640–5. doi: 10.1111/1759-7714.12631
132. Hristozova T, Konschak R, Stromberger C, Fusi A, Liu Z, Weichert W, et al. The presence of circulating tumor cells (CTCs) correlates with lymph node metastasis in nonresectable squamous cell carcinoma of the head and neck region (SCCHN). Ann Oncol. (2011) 22:1878–85. doi: 10.1093/annonc/mdr130
133. Baccelli I, Schneeweiss A, Riethdorf S, Stenzinger A, Schillert A, Vogel V, et al. Identification of a population of blood circulating tumor cells from breast cancer patients that initiates metastasis in a xenograft assay. Nat Biotechnol. (2013) 31:539–44. doi: 10.1038/nbt.2576
134. Kulasinghe A, Perry C, Warkiani ME, Blick T, Davies A, O'Byrne K, et al. Short term ex-vivo expansion of circulating head and neck tumour cells. Oncotarget (2016) 7:60101–9. doi: 10.18632/oncotarget.11159
135. Kulasinghe A, Tran TH, Blick T, O'Byrne K, Thompson EW, Warkiani ME, et al. Enrichment of circulating head and neck tumour cells using spiral microfluidic technology. Sci Rep. (2017) 7:42517. doi: 10.1038/srep42517
136. Maheswaran S, Haber DA. Ex Vivo Culture of CTCs: an emerging resource to guide cancer therapy. Cancer Res. (2015) 75:2411–5. doi: 10.1158/0008-5472.CAN-15-0145
137. Gao D, Vela I, Sboner A, Iaquinta PJ, Karthaus WR, Gopalan A, et al. Organoid cultures derived from patients with advanced prostate cancer. Cell (2014) 159:176–87. doi: 10.1016/j.cell.2014.08.016
138. Alix-Panabieres C, Bartkowiak K, Pantel K. Functional studies on circulating and disseminated tumor cells in carcinoma patients. Mol Oncol. (2016) 10:443–9. doi: 10.1016/j.molonc.2016.01.004
139. Wang R, Chu GCY, Mrdenovic S, Annamalai AA, Hendifar AE, Nissen NN, et al. Cultured circulating tumor cells and their derived xenografts for personalized oncology. Asian J Urol. (2016) 3:240–53. doi: 10.1016/j.ajur.2016.08.005
140. Khoo BL, Grenci G, Jing T, Lim YB, Lee SC, Thiery JP, et al. Liquid biopsy and therapeutic response: Circulating tumor cell cultures for evaluation of anticancer treatment. Sci Adv. (2016) 2:e1600274. doi: 10.1126/sciadv.1600274
141. Zhang Z, Shiratsuchi H, Lin J, Chen G, Reddy RM, Azizi E, et al. Expansion of CTCs from early stage lung cancer patients using a microfluidic co-culture model. Oncotarget (2014) 5:12383–97. doi: 10.18632/oncotarget.2592
142. Hodgkinson CL, Morrow CJ, Li Y, Metcalf RL, Rothwell DG, Trapani F, et al. Tumorigenicity and genetic profiling of circulating tumor cells in small-cell lung cancer. Nat Med. (2014) 20:897–903. doi: 10.1038/nm.3600
143. Hamilton G, Burghuber O, Zeillinger R. Circulating tumor cells in small cell lung cancer: ex vivo expansion. Lung (2015) 193:451–2. doi: 10.1007/s00408-015-9725-7
144. Drapkin BJ, George J, Christensen CL, Mino-Kenudson M, Dries R, Sundaresan T, et al. Genomic and functional fidelity of small cell lung cancer patient-derived xenografts. Cancer Discov. 8:600–15. doi: 10.1158/2159-8290.CD-17-0935
145. Pantel K, Alix-Panabières C. Cell lines from circulating tumor cells. Oncoscience (2015) 2:815–6. doi: 10.18632/oncoscience.195
146. Pantel K, Alix-Panabieres C. Functional studies on viable circulating tumor cells. Clin Chem. (2016) 62:328–34. doi: 10.1373/clinchem.2015.242537
147. Khoo BL, Lee SC, Kumar P, Tan TZ, Warkiani ME, Ow SG, et al. Short-term expansion of breast circulating cancer cells predicts response to anti-cancer therapy. Oncotarget (2015) 6:15578–93. doi: 10.18632/oncotarget.3903
148. Kolostova K, Spicka J, Matkowski R, Bobek V. Isolation, primary culture, morphological and molecular characterization of circulating tumor cells in gynecological cancers. Am J Transl Res. (2015) 7:1203–13.
149. Mishra DK, Creighton CJ, Zhang Y, Chen F, Thrall MJ, Kim MP. Ex vivo four-dimensional lung cancer model mimics metastasis. Ann Thorac Surg. (2015) 99:1149–56. doi: 10.1016/j.athoracsur.2014.08.085
150. Mishra DK, Miller RA, Pence KA, Kim MP. Small cell and non small cell lung cancer form metastasis on cellular 4D lung model. BMC Cancer (2018) 18:441. doi: 10.1186/s12885-018-4358-x
151. Hidalgo M, Amant F, Biankin AV, Budinska E, Byrne AT, Caldas C, et al. Patient-derived xenograft models: an emerging platform for translational cancer research. Cancer Discov. (2014) 4:998–1013. doi: 10.1158/2159-8290.CD-14-0001
152. Dive C, Brady G. Snapshot: circulating tumor cells. Cell (2017) 168:742.e741. doi: 10.1016/j.cell.2017.01.026
153. Albert JM. Radiation risk from CT: implications for cancer screening. Am J Roentgenol. (2013) 201:W81–7. doi: 10.2214/AJR.12.9226
154. Krebs MG, Metcalf RL, Carter L, Brady G, Blackhall FH, Dive C. Molecular analysis of circulating tumour cells-biology and biomarkers. Nat Rev Clin Oncol. (2014) 11:129–44. doi: 10.1038/nrclinonc.2013.253
155. Faltas B. Circulating tumor cells in the cerebrospinal fluid: “tapping” into diagnostic and predictive potential. Oncotarget (2011) 2:822. doi: 10.18632/oncotarget.349
156. Polanski J, Jankowska-Polanska B, Rosinczuk J, Chabowski M, Szymanska-Chabowska A. Quality of life of patients with lung cancer. Onco Targets Ther. (2016) 9:1023–8. doi: 10.2147/OTT.S100685
157. Man Y, Wang Q, Kemmner W. Currently used markers for CTC isolation - advantages, limitations and impact on cancer prognosis. J Clin Exp Path. (2011) 1:102. doi: 10.4172/2161-0681.1000102
158. Allard WJ, Matera J, Miller MC, Repollet M, Connelly MC, Rao C, et al. Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin Cancer Res. (2004) 10:6897–904. doi: 10.1158/1078-0432.ccr-04-0378
159. Bidard FC, Michiels S, Riethdorf S, Mueller V, Esserman LJ, Lucci A, et al. Circulating tumor cells in breast cancer patients treated by neoadjuvant chemotherapy: a meta-analysis. J Natl Cancer Inst. (2018) 110:560–7. doi: 10.1093/jnci/djy018
160. Mazel M, Jacot W, Pantel K, Bartkowiak K, Topart D, Cayrefourcq L, et al. Frequent expression of PD-L1 on circulating breast cancer cells. Mol Oncol. (2015) 9:1773–82. doi: 10.1016/j.molonc.2015.05.009
161. Kulasinghe A, Perry C, Kenny L, Warkiani ME, Nelson C, Punyadeera C. PD-L1 expressing circulating tumour cells in head and neck cancers. BMC Cancer (2017) 17:333. doi: 10.1186/s12885-017-3316-3
162. Strati A, Koutsodontis G, Papaxoinis G, Angelidis I, Zavridou M, Economopoulou P, et al. Prognostic significance of PD-L1 expression on circulating tumor cells in patients with head and neck squamous cell carcinoma. Ann Oncol. (2017) 28:1923–33. doi: 10.1093/annonc/mdx206
163. Pinzani P, Salvadori B, Simi L, Bianchi S, Distante V, Cataliotti L, et al. Isolation by size of epithelial tumor cells in peripheral blood of patients with breast cancer: correlation with real-time reverse transcriptase–polymerase chain reaction results and feasibility of molecular analysis by laser microdissection. Hum Pathol. (2006) 37:711–8. doi: 10.1016/j.humpath.2006.01.026
164. Kaifi JT, Kunkel M, Das A, Harouaka RA, Dicker DT, Li G, et al. Circulating tumor cell isolation during resection of colorectal cancer lung and liver metastases: a prospective trial with different detection techniques. Cancer Biol Ther. (2015) 16:699–708. doi: 10.1080/15384047.2015.1030556
165. Schlange T, Stoecklein N, Neves RP, Pleier S, Bender S, Brychta N, et al. Abstract 513: Standardization of technologies for CTC, ctDNA and miRNA enrichment, isolation and analysis for liquid biopsies during the first year of IMI's CANCER-ID. Cancer Res. (2016) 76:513. doi: 10.1158/1538-7445.AM2016-513
166. Neumann MH, Schneck H, Decker Y, Schomer S, Franken A, Endris V, et al. Isolation and characterization of circulating tumor cells using a novel workflow combining the CellSearch((R)) system and the CellCelector(). Biotechnol Prog. (2017) 33:125–32. doi: 10.1002/btpr.2294
167. American, Cancer Society [Online],. Available online at: https://www.cancer.org/cancer/non-small-cell-lung-cancer.html (Accessed October 11, 2017).
Keywords: lung cancer, NSCLC, SCLC, Circulating tumor cells, liquid biopsy
Citation: Kapeleris J, Kulasinghe A, Warkiani ME, Vela I, Kenny L, O'Byrne K and Punyadeera C (2018) The Prognostic Role of Circulating Tumor Cells (CTCs) in Lung Cancer. Front. Oncol. 8:311. doi: 10.3389/fonc.2018.00311
Received: 21 May 2018; Accepted: 23 July 2018;
Published: 14 August 2018.
Edited by:
Karen L. Reckamp, Irell & Manella Graduate School of Biological Sciences, City of Hope, United StatesReviewed by:
Shadia I. Jalal, Indiana University Bloomington, United StatesTimothy F. Burns, University of Pittsburgh Cancer Institute, United States
Copyright © 2018 Kapeleris, Kulasinghe, Warkiani, Vela, Kenny, O'Byrne and Punyadeera. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chamindie Punyadeera, Y2hhbWluZGllLnB1bnlhZGVlcmFAcXV0LmVkdS5hdQ==
 Joanna Kapeleris
Joanna Kapeleris Arutha Kulasinghe
Arutha Kulasinghe Majid E. Warkiani3,4
Majid E. Warkiani3,4 Liz Kenny
Liz Kenny Kenneth O'Byrne
Kenneth O'Byrne Chamindie Punyadeera
Chamindie Punyadeera