- 1School of Medicine, Case Western Reserve University, Cleveland, OH, United States
- 2Department of Veterinary Physiology and Pharmacology, Texas A&M University, College Station, TX, United States
- 3Department of Medicine, Brigham and Women's Hospital and Harvard Medical School, Boston, MA, United States
- 4Division of Surgical Research, Department of Surgery, Jefferson Pancreas, Biliary and Related Cancer Center, Jefferson Medical College, Thomas Jefferson University, Philadelphia, PA, United States
- 5Department of Surgery and Division of Surgical Oncology, University Hospitals Cleveland Medical Center, Cleveland, OH, United States
A Corrigendum on
Metabolic Dependencies in Pancreatic Cancer
by Vaziri-Gohar, A., Zarei, M., Brody, J. R., and Winter, J. M. (2018). Front. Oncol. 8:617. doi: 10.3389/fonc.2018.00617
In the original article, all references for in Tables 1, 2 were incorrectly listed. The corrected references for both Table 1 and Table 2 have been corrected and provided below.
In the original article, references in Table 2 were not provided in the reference list. The references have now been inserted.
The authors apologize for these errors and state that they do not change the scientific conclusions of the article in any way. The original article has been updated.
References
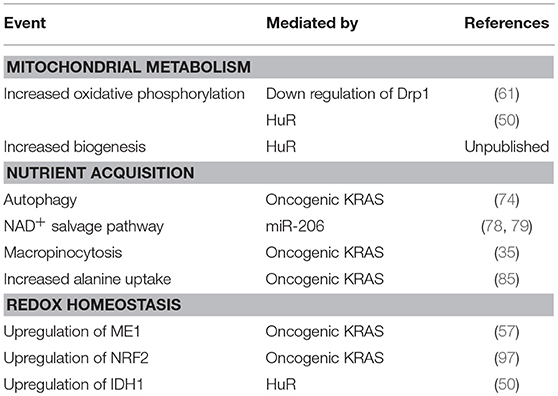
35. Commisso C, Davidson SM, Soydaner-Azeloglu RG, Parker SJ, Kamphorst JJ, Hackett S, et al. Macropinocytosis of protein is an amino acid supply route in Ras-transformed cells. Nature (2013) 497:633–7. doi: 10.1038/nature12138
50. Zarei M, Lal S, Parker SJ, Nevler A, Vaziri-Gohar A, Dukleska K, et al. Posttranscriptional upregulation of IDH1 by HuR establishes a powerful survival phenotype in pancreatic cancer cells. Cancer Res. (2017) 77:4460–71. doi: 10.1158/0008-5472.CAN-17-0015
57. Son J, Lyssiotis CA, Ying H, Wang X, Hua S, Ligorio M, et al. Glutamine supports pancreatic cancer growth through a KRAS-regulated metabolic pathway. Nature (2013) 496:101–5. doi: 10.1038/nature12040
61. Rambold AS, Kostelecky B, Elia N, Lippincott-Schwartz J. Tubular network formation protects mitochondria from autophagosomal degradation during nutrient starvation. Proc Natl Acad Sci USA. (2011) 108:10190–5. doi: 10.1073/pnas.1107402108
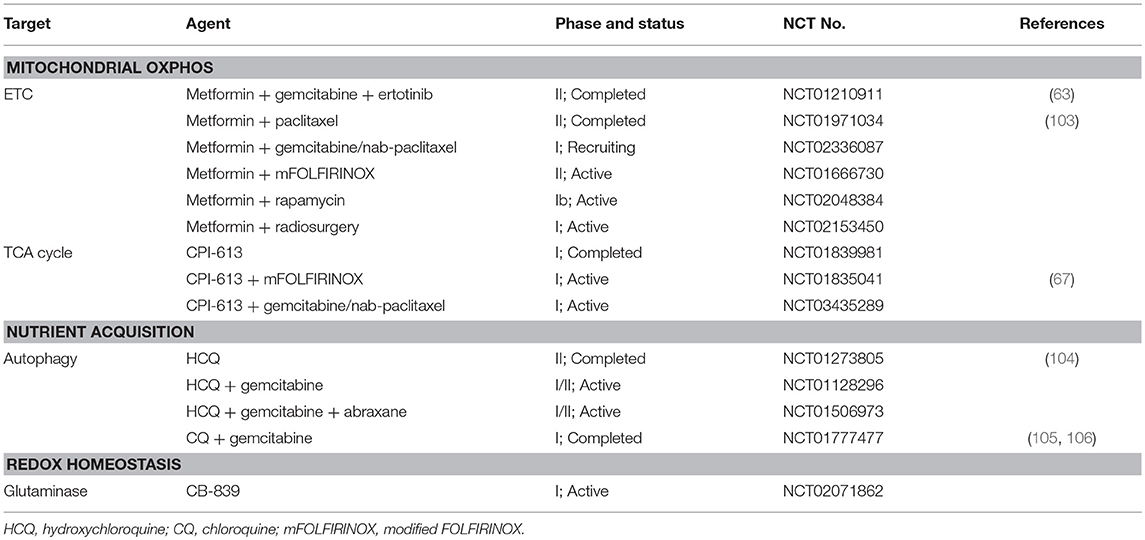
63. Kordes S, Pollak MN, Zwinderman AH, Mathot RA, Weterman MJ, Beeker A, et al. Metformin in patients with advanced pancreatic cancer: a double-blind, randomised, placebo-controlled phase 2 trial. Lancet Oncol. (2015) 16:839–47. doi: 10.1016/S1470-2045(15)00027-3
67. Alistar A, Morris BB, Desnoyer R, Klepin HD, Hosseinzadeh K, Clark C, et al. Safety and tolerability of the first-in-class agent CPI-613 in combination with modified FOLFIRINOX in patients with metastatic pancreatic cancer: a single-centre, open-label, dose-escalation, phase 1 trial. Lancet Oncol. (2017) 18:770–8. doi: 10.1016/S1470-2045(17)30314-5
74. Yang S, Wang X, Contino G, Liesa M, Sahin E, Ying H, et al. Pancreatic cancers require autophagy for tumor growth. Genes Dev. (2011) 25:717–29. doi: 10.1101/gad.2016111
78. Ju HQ, Zhuang ZN, Li H, Tian T, Lu YX, Fan XQ, et al. Regulation of the Nampt-mediated NAD salvage pathway and its therapeutic implications in pancreatic cancer. Cancer Lett. (2016) 379:1–11. doi: 10.1016/j.canlet.2016.05.024
79. Chini CC, Guerrico AM, Nin V, Camacho-Pereira J, Escande C, Barbosa MT, et al. Targeting of NAD metabolism in pancreatic cancer cells: potential novel therapy for pancreatic tumors. Clin Cancer Res. (2014) 20:120–30. doi: 10.1158/1078-0432.CCR-13-0150
85. Sousa CM, Biancur DE, Wang X, Halbrook CJ, Sherman MH, Zhang L, et al. Pancreatic stellate cells support tumour metabolism through autophagic alanine secretion. Nature (2016) 536:479–83. doi: 10.1038/nature19084
97. DeNicola GM, Karreth FA, Humpton TJ, Gopinathan A, Wei C, Frese K, et al. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature (2011) 475:106–9. doi: 10.1038/nature10189
103. Braghiroli MI, de Celis Ferrari AC, Pfiffer TE, Alex AK, Nebuloni D, Carneiro AS, et al. Phase II trial of metformin and paclitaxel for patients with gemcitabine-refractory advanced adenocarcinoma of the pancreas. Ecancermedicalscience (2015) 9:563. doi: 10.3332/ecancer.2015.563
104. Wolpin BM, Rubinson DA, Wang X, Chan JA, Cleary JM, Enzinger PC, et al. Phase II and pharmacodynamic study of autophagy inhibition using hydroxychloroquine in patients with metastatic pancreatic adenocarcinoma. Oncologist (2014) 19:637–8. doi: 10.1634/theoncologist.2014-0086
105. Balic A, Sørensen MD, Trabulo SM, Sainz B, Cioffi M, Vieira CR, et al. Chloroquine targets pancreatic cancer stem cells via inhibition of CXCR4 and hedgehog signaling. Mol Cancer Ther. (2014) 13:1758–71. doi: 10.1158/1535-7163.MCT-13-0948
Keywords: pancreatic cancer, metabolism, redox homeostasis, metabolic dependencies, targeting metabolism
Citation: Vaziri-Gohar A, Zarei M, Brody JR and Winter JM (2019) Corrigendum: Metabolic Dependencies in Pancreatic Cancer. Front. Oncol. 8:672. doi: 10.3389/fonc.2018.00672
Received: 19 December 2018; Accepted: 20 December 2018;
Published: 31 January 2019.
Copyright © 2019 Vaziri-Gohar, Zarei, Brody and Winter. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jordan M. Winter, am9yZGFuLndpbnRlckBVSGhvc3BpdGFscy5vcmc=
 Ali Vaziri-Gohar
Ali Vaziri-Gohar Mahsa Zarei
Mahsa Zarei Jonathan R. Brody
Jonathan R. Brody Jordan M. Winter1,5*
Jordan M. Winter1,5*