- 1Division of Hematology and Oncology, Department of Medicine, Vanderbilt University Medical Center, Nashville, TN, United States
- 2Division of Cardiovascular Medicine, Brigham and Women's Hospital, Harvard Medical School, Boston, MA, United States
- 3Cardio-Oncology Program, Vanderbilt University Medical Center, Nashville, TN, United States
- 4Vanderbilt Institute for Clinical and Translational Research, Nashville, TN, United States
Immunomodulatory drugs (IMiDs), including thalidomide, lenalidomide, and pomalidomide, have improved survival of patients with multiple myeloma (MM). However, these therapies are associated with an increased risk of venous thromboembolism (VTE). Apixaban has been approved for treatment of acute VTE and for risk reduction of recurrent VTE following initial therapy. In this phase IV single-arm study (NCT02958969), we aim to prospectively evaluate the safety and efficacy of apixaban for primary prevention of VTE in patients with MM. The primary efficacy objective of this trial is to determine the rate of symptomatic VTE, including deep vein thrombosis (DVT) and pulmonary embolism (PE), over 6 months. The primary safety objective is to determine the rate of major bleeding in MM patients receiving apixaban prophylaxis. If proven safe and effective, apixaban will emerge as a promising option for oral VTE prophylaxis in MM patients.
Introduction
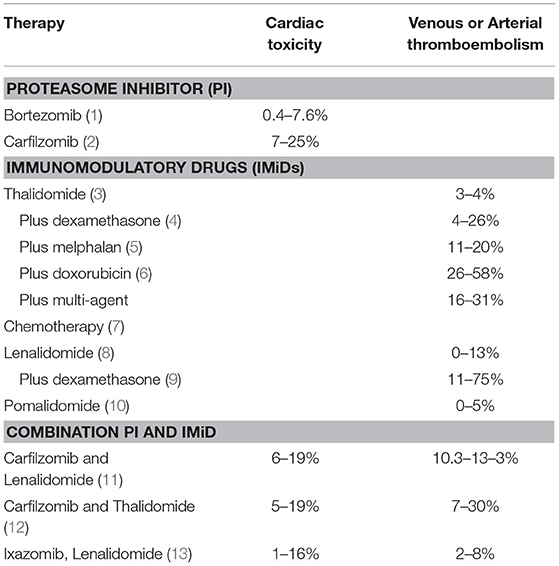
Multiple myeloma (MM) is a plasma cell malignancy characterized by clonal proliferation of malignant plasma cells in the bone marrow microenvironment, monoclonal protein in the blood or urine, and associated organ dysfunction (1). The 5-year survival rates of MM have nearly doubled during the past 3 decades (24.6% in 1975 vs. 52.4% in 2008) due to treatment advancements such as immunomodulatory/cereblon-binding drugs (IMiDs: thalidomide, lenalidomide, pomalidomide), proteasome inhibitors (PIs: bortezomib, carfilzomib, ixazomib), monoclonal antibodies targeting CD38 (daratumumab) and SLAMF7 (elotuzumab), among other emerging therapies (2).
Multiple Myeloma and Thrombosis
MM has been associated with an increased risk of venous thromboembolism (VTE) (3, 4). Compared with the general population, two large population-based studies found the incidence of VTE to be higher in patients with MM (5, 6). In a study of over 4 million US veterans, VTE risk was 9-fold higher in patients with MM compared all other patients. In a Swedish study of over 18,000 patients with MM and matched controls, the risk of VTE was 7.5-, 4.6-, and 4.1-fold higher at 1, 5, and 10 years, respectively, in patients with MM.
The risk of VTE is further increased in the setting of IMiDs (Table 1) (3, 7). However, the mechanism by which IMiDs increase thromboembolic events is unknown (3, 7). A meta-analysis of >3,000 MM patients found the risk of VTE increased 2.6-fold with thalidomide and 8-fold when thalidomide was prescribed with dexamethasone (8). In newly diagnosed MM, patients treated with lenalidomide plus high-dose dexamethasone a high rate (26%) of VTE was observed (9). These early associations of increased VTE risk with IMiDs have led to the common practice of thromboprophylaxis in MM. Due to the increasing use of IMiD-based regimens, prevention of VTE has become a major management challenge during MM treatment.
VTE Prophylaxis Strategies in MM
VTE risk assessment in MM includes individual risk factors, such as obesity, previous VTE, immobilization, disease status, and therapeutic regimen (3). Several VTE prophylactic strategies have been evaluated in MM (10, 11). However, prospective clinical trials are rare, and the optimal thromboprophylactic regimen remains uncertain. A phase III trial of 667 patients with newly diagnosed MM (NDMM) treated with thalidomide-based regimens indicated that aspirin (100 mg daily) or low-dose warfarin (1.25 mg daily) showed efficacies similar to LMWH (enoxaparin, 40 mg daily) in reducing VTE. However, patients with high risk of VTE were excluded from this study (12). Another prospective study included 342 patients randomized to either low dose aspirin (100 mg daily) or LWMH (enoxaparin 40 mg daily) with NDMM who were undergoing treatment with lenalidomide and low-dose dexamethasone. Aspirin was as effective as LMWH in this study; however, patients in this trial were relatively young (<65 years), lacked risk factors for cardiovascular disease or thrombosis, and had no history of VTE (13). Based on these limited studies and the consensus opinion of experts in the field, the International Myeloma Working Group has made recommendations regarding VTE risk assessment and prevention of IMiD-associated thrombosis in patients with MM. While aspirin (81–325 mg/day) is recommended for low risk patients, low molecular weight heparin (LMWH) is suggested for patients determined to be at high-risk for VTE (3).
LMWH, such as dalteparin, enoxaparin, and tinzaparin, have been well-studied for secondary prevention of cancer-related VTEs. However, the use of LMWH is complicated by injection site discomfort and consequent poor adherence to the prescribed regimen (14). In addition, LMWH is contraindicated in patients with a history of heparin-induced thrombocytopenia (HIT) and introduces a small risk of inducing HIT in patients (15).
A growing body of literature supports the use of direct oral anticoagulants (DOACs), including apixaban, for VTE prophylaxis (16). Apixaban directly blocks Factor Xa, which inhibits coagulation by interfering with conversion of prothrombin to thrombin, and is largely metabolized in the liver (17). Compared with injectable thromboprophylactic regimens, such as enoxaparin, apixaban offers the advantages of oral administration and does not require laboratory monitoring (18).
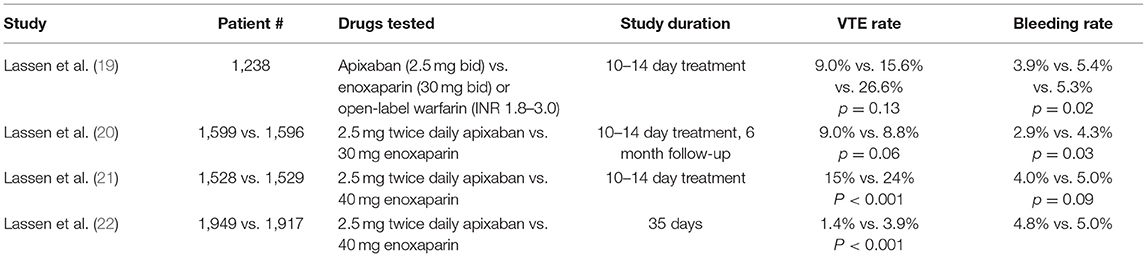
Apixaban prophylactic strategies have been evaluated for prevention of post-operative VTE after hip and knee replacement (Table 2). Studies of arthroplasty patients suggest that 2.5 mg twice-daily apixaban is superior to enoxaparin for VTE prevention with similar bleeding event rates. Apixaban is currently FDA-approved for treatment of acute VTE and prophylactic use following hip and knee replacement surgery and for reducing the risk of stroke in patients with non-valvular atrial fibrillation (23, 24).
In the randomized trial, Apixaban after Initial Management of Pulmonary Embolism and Deep Vein Thrombosis (AMPLIFY-EXT), apixaban (2.5 or 5 mg twice daily) was evaluated vs. placebo after patients completed 6–12 months of anticoagulation (25). The rate of recurrent VTE was reduced from 8.8 to 1.7% in patients receiving placebo or apixaban, respectively. No differences were found between the two dose levels of apixaban for recurrence of VTE (1.7 vs. 1.7%). There were no significant differences in the risk of major hemorrhage between dose levels (0.2 vs. 0.1%) and this risk was similar to that seen in the placebo group (0.5%). Apixaban was also effective in preventing thromboembolism in patients receiving chemotherapy for advanced metastatic cancer (26). In this phase II trial, patients were randomized to receive 5, 10, or 20 mg once daily or placebo over 12 weeks. The rate of major and non-major bleeding was 3.1, 3.1, and 3.4% in the 5, 10 mg, and placebo groups, respectively. Risk of VTE was higher in the placebo group compared to all groups treated with apixaban (10.3 vs. 0%). Ten patients with metastatic MM were included in the apixaban cohorts (26).
Here, we describe our phase IV clinical trial (NCT02958969) that will further investigate the safety and efficacy of apixaban for VTE prophylaxis for patients with MM.
Main Study Objectives
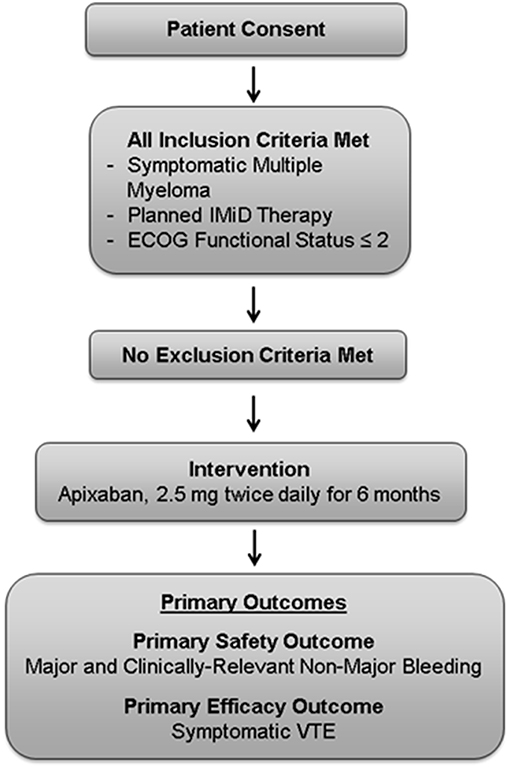
Our primary efficacy outcome is to assess the rate of symptomatic VTE over 6 months in patients with MM receiving IMiDs who are prescribed apixaban 2.5 mg orally twice daily for primary prevention of VTE. Accounting for patients receiving IMiDs at all phases of treatment, including maintenance, we hypothesize that the 6-month rate of symptomatic VTE will be <5% (27–30).
Our primary safety objective is to quantitatively assess the 6-month rate of major and clinically-relevant, non-major bleeding. We hypothesize that the 6-month rate of major and clinically-relevant, non-major bleeding in these patients will be ≤3% (31). We will also quantify the 6-month rate of myocardial infarction and stroke, which are known to occur in patients with MM receiving IMiDs (32).
Methods
Study Design
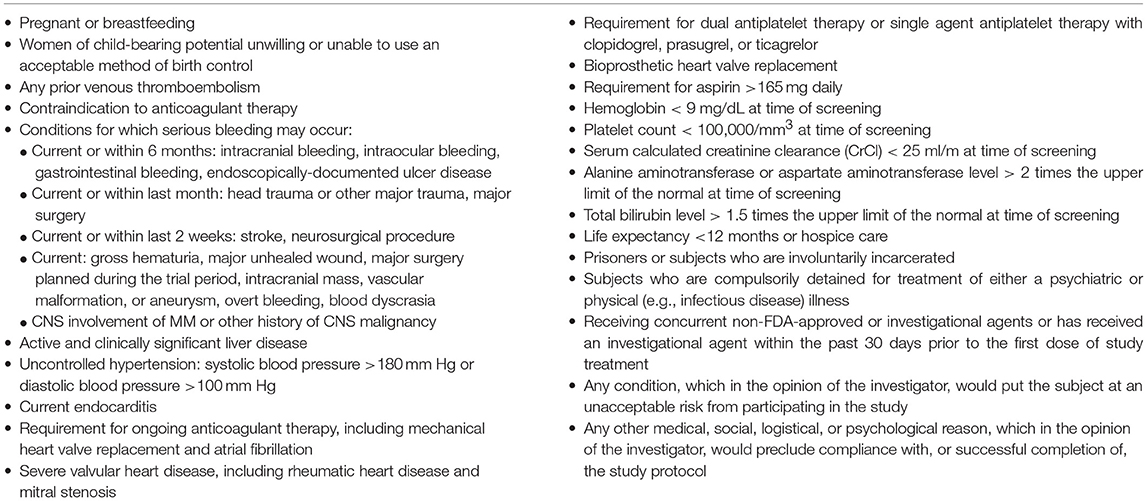
This is a phase 4, investigator-initiated U.S.-based, single-center, single-arm, open-label, proof-of-concept study (NCT02958969; Figure 1). Fifty patients will receive apixaban 2.5 mg orally twice daily for primary prevention of VTE for a planned 6 months. We project that this study will be completed in 24 months. The study population will include both male and female patients who are 18 years of age or older with MM defined according to the International Myeloma Working Group (IMWG) guidelines, and receiving IMiD-based therapy (33). Patients must have an Eastern Cooperative Oncology Group (ECOG) functional status ≤2. Patients will be receiving or starting IMiD therapy, with planned IMiD therapy for a minimum of 6 months. Patients will begin prophylactic anticoagulation within 3 weeks of enrollment. Patients will be instructed to stop aspirin prophylaxis while receiving apixaban. Inclusion and exclusion criteria are detailed in Tables 3, 4.
Primary and Secondary Study Outcomes
Efficacy Outcomes
As our primary efficacy outcome, we will assess the rate of symptomatic VTE, including deep vein thrombosis (DVT) and pulmonary embolism (PE), over 6 months. DVT will be diagnosed by ultrasonography as a non-compressible venous segment or segments or as a filling defect detected by computed tomographic (CT) venography, magnetic resonance (MR) venography, or contrast venography of the lower extremities from the popliteal vein or higher and all upper extremity DVTs will be captured including catheter-related. PE, subsegmental or greater, will be diagnosed by the presence of mismatched perfusion defects during ventilation perfusion scan, the presence of a pulmonary artery filling defect detected by contrast-enhanced chest CT, an intraluminal filling defect detected by invasive pulmonary angiography, or confirmation of PE at autopsy (25). Accordingly, as a secondary efficacy outcome, we will investigate the rate of MI and stroke at 6 months. Acute MI will be defined according to current standards (34). All strokes during the study will be assessed by imaging or autopsy and classified as primary hemorrhagic, non-hemorrhagic, infarction with hemorrhagic conversion, or unknown, as defined by the AHA/ASA (35).
Safety Outcomes
As our primary safety outcome, we will assess the rate of major and clinically relevant non-major bleeding over 6 months (Table 5). According to the International Society on Thrombosis (ISTH) classification, major bleeding is defined as overt bleeding that is associated with a decrease in hemoglobin of 2 g/dL or more, requiring the transfusion of 2 or more units of blood, occurring in a critical site, or contributing to death (36). ISTH classification defines clinically relevant non-major bleeding as overt bleeding that does not meeting the criteria for major bleeding, but is associated with medical intervention, surgical intervention, or interruption of the study drug. Secondarily, we will record all mortality at 6 months. Cause of death will be classified as related to cancer, myocardial infarction, pulmonary embolism, other cardiovascular causes, or due to other disease states.
Study Monitoring
This study has been approved by the Institutional Review Board and is registered through ClinicalTrials.gov (NCT02958969). This study was designed in accordance with good clinical practice guidelines. The Vanderbilt University Medical Center Data Monitoring Committee is responsible for monitoring the progress and safety throughout the study. Adherence to the protocol will be assessed by pill count and calculated as the number of pills taken divided by the number of pills prescribed. Furthermore, subjects will be given a study diary to fill out at home each day. Patients will be asked to write down the time study drug is taken and any side effects. Study diaries will be collected at a regularly scheduled office visit at conclusion of the study period.
Statistical Methods and Sample Size Estimation
A formal hypothesis test will not be conducted in this proof-of-concept single arm study. Sample size estimation will be based on the precision of the measurements using the 95% confidence interval (CI) method. A sample size of 50 patients generates a half-width of CI <10%. Based on the estimated 5% VTE incidence rate, the half-width CI will be <6%. Therefore, the precision will be effectively measured with the estimated CI.
Statistical Analysis Plan
For this proof-of-concept study, we will calculate the 6-month rates of symptomatic VTE and major and clinically relevant non-major bleeding. We will also calculate 6-month rates of myocardial infarction and stroke. The primary analysis will be intention-to-treat.
Means, medians, and frequency distributions will be calculated for continuous variables. Number and percentages will be reported for binary and categorical variables. Differences between subgroups of interest will be examined using the chi-square or Fisher's exact test for binary and categorical variables and t-test or Wilcoxon Rank Sum for continuous variables, depending on the number of patients in each group. Subgroup analysis will include a comparison of newly diagnosed patients vs. those with recurrent or relapsed disease. Addition studies will compare patients based on the use of other concomitant medications (e.g., dexamethasone). All tests will be two-tailed and a p < 0.05 assumed to represent statistical significance. All analyses will be performed using SAS software.
Conclusion and Discussion
Immunomodulator/cereblon-binding agents comprise the therapeutic foundation of therapy for MM at all phases of therapy. While these agents are generally well tolerated, their increased risk of VTE presents a clinical challenge. Further compounding this challenge is the lack of rigorous, prospective trials of thromboprophylactic regimens for VTE prevention in these vulnerable patients (16, 37). The prophylactic efficacy of apixaban in cancer is under evaluation in several clinical trials (i.e., NCT02048865, NCT02366871). This study will specifically assess the efficacy and safety of apixaban for primary prevention in MM. If successful, this regimen has the potential to greatly improve VTE prophylaxis options for patients with MM.
Ethics Statement
This study was carried out in accordance with the recommendations of the Vanderbilt University Medical Center Institutional Review Board with written informed consent from all subjects. All subjects gave written informed consent in accordance with the Declaration of Helsinki. The protocol was approved by the Vanderbilt University Medical Center Institutional Review Board and Scientific Review Committee.
Author Contributions
RC, SG, JM, and GP developed the Research Concept, designed, and wrote the protocol. RC, SG, JM, GP, MJ, DP, SH, RH, BE, and HW critically reviewed the manuscript at all steps of developed. RC, JM, MJ, DP, SH, RH, BE, and HW enrolled and treated patients and completed all study required training and protocol requirements.
Funding
This research was funded by a Bristol-Myers Squibb/Pfizer Alliance research grant.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Palumbo A, Anderson K. Multiple myeloma. New Eng J Med. (2011) 364:1046–60. doi: 10.1056/NEJMra1011442
2. Gandhi UH, Senapedis W, Baloglu E, Unger TJ, Chari A, Vogl D, et al. Clinical implications of targeting XPO1-mediated nuclear export in multiple myeloma. Clin Lymph Myeloma Leukemia. (2018) 18:335–45. doi: 10.1016/j.clml.2018.03.003
3. Li W, Cornell RF, Lenihan D, Slosky D, Jagasia M, Piazza G, et al. Cardiovascular complications of novel multiple myeloma treatments. Circulation (2016) 133:908–12. doi: 10.1161/CIRCULATIONAHA.115.018351
4. Li W, Garcia D, Cornell RF, Gailani D, Laubach J, Maglio ME, et al. Cardiovascular and thrombotic complications of novel multiple myeloma therapies: a review. JAMA Oncol. (2017) 3:980–8. doi: 10.1001/jamaoncol.2016.3350
5. Piazza G. Venous thromboembolism and cancer. Circulation (2013) 128:2614–8. doi: 10.1161/CIRCULATIONAHA.113.002702
6. Piazza G, Rao AF, Nguyen TN, Seger AC, Hohlfelder B, Fanikos J, et al. Venous thromboembolism in hospitalized patients with active cancer. Clin Appl Thromb Hemost. (2013) 19:469–75. doi: 10.1177/1076029613481844
7. Fradley MG, Groarke JD, Laubach J, Alsina M, Lenihan DJ, Cornell RF, et al. Recurrent cardiotoxicity potentiated by the interaction of proteasome inhibitor and immunomodulatory therapy for the treatment of multiple myeloma. Br J Haematol. (2018) 180:271–5. doi: 10.1111/bjh.14970
8. Hicks LK, Haynes AE, Reece DE, Walker IR, Herst JA, Meyer RM, et al. A meta-analysis and systematic review of thalidomide for patients with previously untreated multiple myeloma. Cancer Treat Rev. (2008) 34:442–52. doi: 10.1016/j.ctrv.2008.02.003
9. Rajkumar SV, Jacobus S, Callander NS, Fonseca R, Vesole DH, Williams ME, et al. Lenalidomide plus high-dose dexamethasone versus lenalidomide plus low-dose dexamethasone as initial therapy for newly diagnosed multiple myeloma: an open-label randomised controlled trial. Lancet Oncol. (2010) 11:29–37. doi: 10.1016/S1470-2045(09)70284-0
10. Mikhael J, Rajkumar V, Roy V, Hayman SR, Fonseca R, Short KD, et al. Efficacy of pomalidomide plus low-dose dexamethasone in multiple myeloma patients despite previous use of lenalidomide. J Clin Oncol. (2011) 29(Suppl. 15):8067. doi: 10.1200/jco.2011.29.15_suppl.8067
11. Zonder JA, Crowley J, Hussein MA, Bolejack V, Moore DF Sr., Whittenberger BF, et al. Lenalidomide and high-dose dexamethasone compared with dexamethasone as initial therapy for multiple myeloma: a randomized Southwest Oncology Group trial (S0232). Blood (2010) 116:5838–41. doi: 10.1182/blood-2010-08-303487
12. Palumbo A, Cavo M, Bringhen S, Zamagni E, Romano A, Patriarca F, et al. Aspirin, warfarin, or enoxaparin thromboprophylaxis in patients with multiple myeloma treated with thalidomide: a phase III, open-label, randomized trial. J Clin Oncol. (2011) 29:986–93. doi: 10.1200/JCO.2010.31.6844
13. Larocca A, Cavallo F, Bringhen S, Di Raimondo F, Falanga A, Evangelista A, et al. Aspirin or enoxaparin thromboprophylaxis for patients with newly diagnosed multiple myeloma treated with lenalidomide. Blood (2012) 119:933–9; quiz 1093. doi: 10.1182/blood-2011-03-344333
14. Fanikos J, Stevens LA, Labreche M, Piazza G, Catapane E, Novack L, et al. Adherence to pharmacological thromboprophylaxis orders in hospitalized patients. Am J Med. (2010) 123:536–41. doi: 10.1016/j.amjmed.2009.11.017
15. Martel N, Lee J, Wells PS. Risk for heparin-induced thrombocytopenia with unfractionated and low-molecular-weight heparin thromboprophylaxis: a meta-analysis. Blood (2005) 106:2710–5. doi: 10.1182/blood-2005-04-1546
16. Storrar NPF, Mathur A, Johnson PRE, Roddie PH. Safety and efficacy of apixaban for routine thromboprophylaxis in myeloma patients treated with thalidomide- and lenalidomide-containing regimens. Br J Haematol. (2018). doi: 10.1111/bjh.15392 [Epub ahead of print].
17. Agrawal R, Jain P, Dikshit SN. Apixaban: a new player in the anticoagulant class. Curr Drug Targets. (2012) 13:863–75. doi: 10.2174/138945012800564059
18. Poulsen BK, Grove EL, Husted SE. New oral anticoagulants: a review of the literature with particular emphasis on patients with impaired renal function. Drugs (2012) 72:1739–53. doi: 10.2165/11635730-000000000-00000
19. Lassen MR, Davidson BL, Gallus A, Pineo G, Ansell J, Deitchman D. The efficacy and safety of apixaban, an oral, direct factor Xa inhibitor, as thromboprophylaxis in patients following total knee replacement. J Thromb Haemost (2007) 5:2368–75. doi: 10.1111/j.1538-7836.2007.02764.x
20. Lassen MR, Raskob GE, Gallus A, Pineo G, Chen D, Portman RJ. Apixaban or enoxaparin for thromboprophylaxis after knee replacement. N Engl J Med. (2009) 361:594–604. doi: 10.1056/NEJMoa0810773
21. Lassen MR, Raskob GE, Gallus A, Pineo G, Chen D, Hornick P. Apixaban versus enoxaparin for thromboprophylaxis after knee replacement (ADVANCE-2): a randomised double-blind trial. Lancet (2010) 375:807–15. doi: 10.1016/S0140-6736(09)62125-5
22. Lassen MR, Gallus A, Raskob GE, Pineo G, Chen D, Ramirez LM. Apixaban versus enoxaparin for thromboprophylaxis after hip replacement. N Engl J Med. (2010) 363:2487–98. doi: 10.1056/NEJMoa1006885
23. Granger CB, Alexander JH, McMurray JJ, Lopes RD, Hylek EM, Hanna M, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. (2011) 365:981–2. doi: 10.1056/NEJMoa1107039
24. Raskob GE, Gallus AS, Pineo GF, Chen D, Ramirez LM, Wright RT, et al. Apixaban versus enoxaparin for thromboprophylaxis after hip or knee replacement: pooled analysis of major venous thromboembolism and bleeding in 8464 patients from the ADVANCE-2 and ADVANCE-3 trials. J Bone Joint Surg Br. (2012) 94:257–64. doi: 10.1302/0301-620X.94B2.27850
25. Agnelli G, Buller HR, Cohen A, Curto M, Gallus AS, Johnson M, et al. Apixaban for extended treatment of venous thromboembolism. N Engl J Med. (2013) 368:699–708. doi: 10.1056/NEJMoa1207541
26. Levine MN, Gu C, Liebman HA, Escalante CP, Solymoss S, Deitchman D, et al. A randomized phase II trial of apixaban for the prevention of thromboembolism in patients with metastatic cancer. J Thromb Haemost. (2012) 10:807–14. doi: 10.1111/j.1538-7836.2012.04693.x
27. Osman K, Comenzo R, Rajkumar SV. Deep venous thrombosis and thalidomide therapy for multiple myeloma. N Engl J Med. (2001) 344:1951–2. doi: 10.1056/NEJM200106213442516
28. Brandenburg NA, Knight T, Anastassopoulos KP, Minton N, Knight RD. Incidence of venous thromboembolic events (VTE) in multiple myeloma patients receiving lenalidomide (LEN) with and without dexamethasone (DEX). J Clin Oncol. (2012) 30(Suppl. 15):e18559-e. doi: 10.1200/jco.2012.30.15_suppl.e18559
29. Attal M, Lauwers-Cances V, Hulin C, Leleu X, Caillot D, Escoffre M, et al. Lenalidomide, bortezomib, and dexamethasone with transplantation for Myeloma. N Engl J Med. (2017) 376:1311–20. doi: 10.1056/NEJMoa1611750
30. Attal M, Lauwers-Cances V, Marit G, Caillot D, Moreau P, Facon T, et al. Lenalidomide maintenance after stem-cell transplantation for multiple myeloma. N Engl J Med. (2012) 366:1782–91. doi: 10.1056/NEJMoa1114138
31. Agnelli G, Buller HR, Cohen A, Curto M, Gallus AS, Johnson M, et al. Oral apixaban for the treatment of acute venous thromboembolism. N Engl J Med. (2013) 369:799–808. doi: 10.1056/NEJMoa1302507
32. Leebeek FW, Kruip MJ, Sonneveld P. Risk and management of thrombosis in multiple myeloma. Thromb Res. (2012) 129 (Suppl. 1):S88–92. doi: 10.1016/S0049-3848(12)70024-5
33. Rajkumar SV. Updated diagnostic criteria and staging system for multiple myeloma. Am Soc Clin Oncol Educ Book. (2016) 35:e418–23. doi: 10.1200/EDBK_159009
34. Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD, et al. Third universal definition of myocardial infarction. Nat Rev Cardiol. (2012) 9:620–33. doi: 10.1038/nrcardio.2012.122
35. Sacco RL, Kasner SE, Broderick JP, Caplan LR, Connors JJ, Culebras A, et al. An updated definition of stroke for the 21st century: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke (2013) 44:2064–89. doi: 10.1161/STR.0b013e318296aeca
36. Schulman S, Angerås U, Bergqvist D, Eriksson B, Lassen MR, Fisher W; Subcommittee on Control of Anticoagulation of the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non-surgical patients. J Thromb Haemost. (2005) 3:692–4. doi: 10.1111/j.1538-7836.2005.01204.x
37. Pegourie B, Pernod G, Karlin L, Benboubker L, Orsini F, Tiab M, et al. Evaluation of an oral direct anti-Xa anticoagulant, apixaban, for the prevention of venous thromboembolism in patients with myeloma treated with IMiD* compounds: a pilot study (MYELAXAT). J Clin Oncol. (2017) 35(Suppl. 15):8019. doi: 10.1200/JCO.2017.35.15_suppl.509
Keywords: Apixaban (Eliquis®), myeloma, anticoagulation (AC), immunomodualtors, venous thomboembolism
Citation: Cornell RF, Goldhaber SZ, Engelhardt BG, Moslehi J, Jagasia M, Patton D, Harrell S, Hall R, Wyatt H and Piazza G (2019) Apixaban for Primary Prevention of Venous Thromboembolism in Patients With Multiple Myeloma Receiving Immunomodulatory Therapy. Front. Oncol. 9:45. doi: 10.3389/fonc.2019.00045
Received: 08 October 2018; Accepted: 17 January 2019;
Published: 26 February 2019.
Edited by:
Onder Alpdogan, Thomas Jefferson University, United StatesReviewed by:
Ehsan Malek, Case Western Reserve University, United StatesRehan Khan, Mayo Clinic, United States
Adam Finn Binder, Thomas Jefferson University, United States
Copyright © 2019 Cornell, Goldhaber, Engelhardt, Moslehi, Jagasia, Patton, Harrell, Hall, Wyatt and Piazza. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Robert Frank Cornell, cm9iZXJ0LmYuY29ybmVsbEB2dW1jLm9yZw==
 Robert Frank Cornell
Robert Frank Cornell Samuel Z. Goldhaber2
Samuel Z. Goldhaber2 Daryl Patton
Daryl Patton