- 1Department of Medical Genetics, Shahid Beheshti University of Medical Sciences, Tehran, Iran
- 2Department of Anatomical Sciences, Faculty of Medicine, Birjand University of Medical Sciences, Birjand, Iran
- 3Urogenital Stem Cell Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran
Endometriosis is a disorder characterized by the presence of endometrial glands and stroma like lesions outside of the uterus. Although several hypothesis have tried to explain the underlying cause of endometriosis, yet the main cause remained obscure. Recent studies have shown contribution of non-coding RNAs in the pathogenesis of endometriosis. Two classes of these transcripts namely long non-coding RNAs (lncRNAs) and microRNAs (miRNAs) have mostly attracted attention of researchers. Several studies have reported aberrant expression of these transcripts in affected tissues from patients as well as animal models. Modulation of important signaling pathways such as PI3K/AKT, P38-MAPK, ERK1/2-MAPK and Wnt-β catenin by miRNAs and lncRNAs have potentiated these molecules as biomarkers or therapeutic agents in endometriosis. Single nucleotide polymorphisms with miR-126, miR-143 and miR-146b have been associated with risk of endometriosis. Moreover, miRNAs and lncRNAs control inflammatory responses, cell proliferation, angiogenesis and tissue remodeling, thus understanding the role of these transcripts in endometriosis is a possible way to develop novel diagnostic tests and therapeutic targets for this disorder.
Introduction
Endometriosis is a condition that endometrial glands and stroma like lesions are detected in organs outside of the uterus (1). These lesions can involve the peritoneum or being presented as superficial implants or cysts on the ovary, or deep infiltrating lesions (2). Although the main etiology of endometriosis is not clear, numerous hypotheses have tried to explain the development of this disorder. Among the most appreciated hypotheses is the retrograde menstruation which can be accompanied by possible hematogenous or lymphatic circulation, thus leading to seeding of endometrial tissue in ectopic places. Yet, this phenomenon is much more prevalent than the occurrence of endometriosis. Hence, other hormonal or immune-related factors contribute in implantation and persistence of lesions in the pelvic cavity (3). Imperfect differentiation or migration of Müllerian residues during fetal period or transdifferentiate of circulating blood cells are other popular hypotheses regarding development of endometriosis (3). Notably, several genomics studies have shown remarkable alterations in gene profile in endometriosis (4). The genetics basis of this condition is complex and has not been explored yet, though, most studies have reported a polygenic/multifactorial mode for its inheritance (4). Most recently, non-coding RNAs have been demonstrated to contribute in the pathogenesis of endometriosis (5). These transcripts have regulatory roles on expression of protein-coding genes, thus regulate several signaling pathways. They are classified into two main classes according to their length: long non-coding RNAs (lncRNAs) with sizes more than 200 nucleotides and microRNAs (miRNAs) with sizes about 20 nucleotides. LncRNAs can regulate the genetic information flow, through modulating chromatin structure, transcription, splicing, mRNA stability, mRNA accesibility, and post-translational alterations. They have interaction domains for DNA, mRNAs, miRNAs, and proteins which are specified by nucleotide sequence and secondary structure (6). NONCODE database has indicated the presence of at least 100,000 lncRNAs in the human genome (7) which significantly surpluses the number of protein coding genes. There are complex interaction networks between lncRNAs and miRNAs. While certain miRNAs can regulate the stability and half-life of lncRNA, lncRNAs can compete with miRNAs for binding with the mRNA target sites (6). Being mostly located in the cytoplasm, miRNAs constitute critical regulators of gene expression. They mostly exert their effects at post-transcriptional level through binding with their targets and subsequent mRNA degradation and/or translational repression. In addition, miRNAs have been shown to exert specific nuclear functions being emphasized by the miRNA-guided transcriptional regulation of gene expression (8). The regulatory roles of miRNAs and lncRNAs in the expression of genes indicate their participation on the pathogenesis of human disorders. In the current study, we summarize the role of these transcripts in the pathophysiology of endometriosis.
MiRNAs and Endometriosis
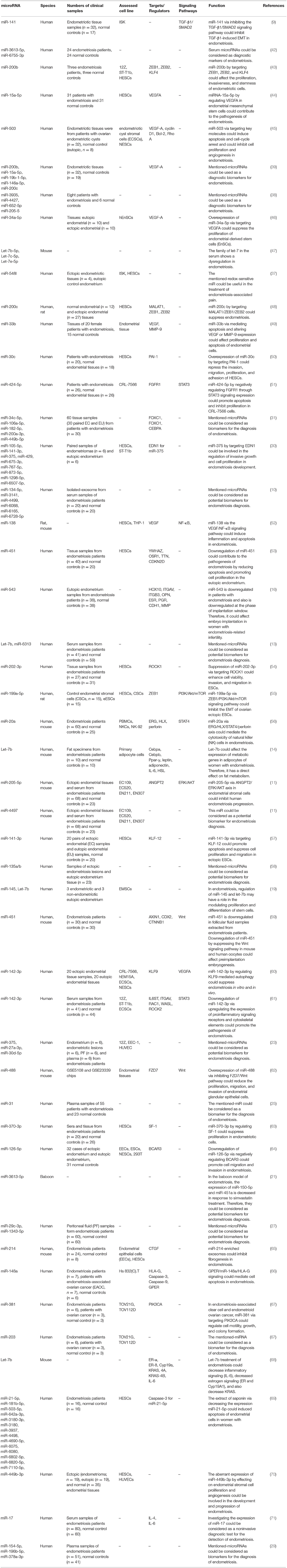
Several studies have reported aberrant expression of miRNAs in affected tissues or peripheral blood samples of patients. Zhang et al. have extracted exosomes from the serum of patients with endometriosis and healthy subjects, then assessed expression miRNAs by miRNA microarrays. They reported differential expression of 24 miRNAs between these two sets of samples. As confirmed by qPCR, expression of miR-22-3p and miR-320a was increased in serum exosomes of patients compared with controls (9). Another study has shown that exosomal miR-22-3p isolated from peritoneal macrophages increases proliferation, migration, and invasion of ectopic endometrial stromal cells via modulation of the SIRT1/NF-κB signaling pathway (10). Others have assessed expression profile of miRNAs peritoneal or tissue samples obtained from these patients. For instance, Zhou et al. have used miRNA microarray technique to identify miRNA signature in the ectopic endometrium samples. They reported over-expression of miR-3154 and miR-3926 in theses tissues compared with normal endometrium (11). Zhang et al. have isolated mononuclear cells from peritoneal fluid of patients with endometriosis and assessed expression of miRNAs in the supernatant of peritoneal fluid. They also purified human endometrial stromal cells from both endometrial and endometriotic tissues of these patients. They reported up-regulation of miR-146b peritoneal fluid supernatant and CD14 + monocytes/Macrophages of peritoneal fluid in endometriosis patients. This miRNA could inhibit the M1 polarization of endometrial stromal cells co-cultured macrophages (12). Table 1 shows the list of up-regulated miRNAs in samples obtained from patients with endometriosis.
Several miRNAs have been shown to be down-regulated during the pathogenic process of endometriosis. Rekker et al. have used fluorescence-activated cell sorting to endometrial stromal cells from paired endometrial and endometrioma biopsies. Subsequently, they profiled miRNAs in endometriotic stroma using high-throughput sequencing method. They reported downregulation of miR-375 in these cells compared to eutopic cells. This miRNA has been shown to regulate expression of the endothelin 1 (EDN1) gene (30). Yang et al. have shown down-regulation of miR-200b, miR-15a-5p, miR-19b-1-5p, miR-146a-5p, and miR-200c while up-regulation of VEGFA in endometriotic tissues. They have speculated that the higher angiogenic and proteolytic activities observed in the eutopic endometrium could assist the implantation of these cells at ectopic regions (39). Table 2 summarizes the function and characteristics of miRNAs that are down-regulated in samples obtained from patients with endometriosis.
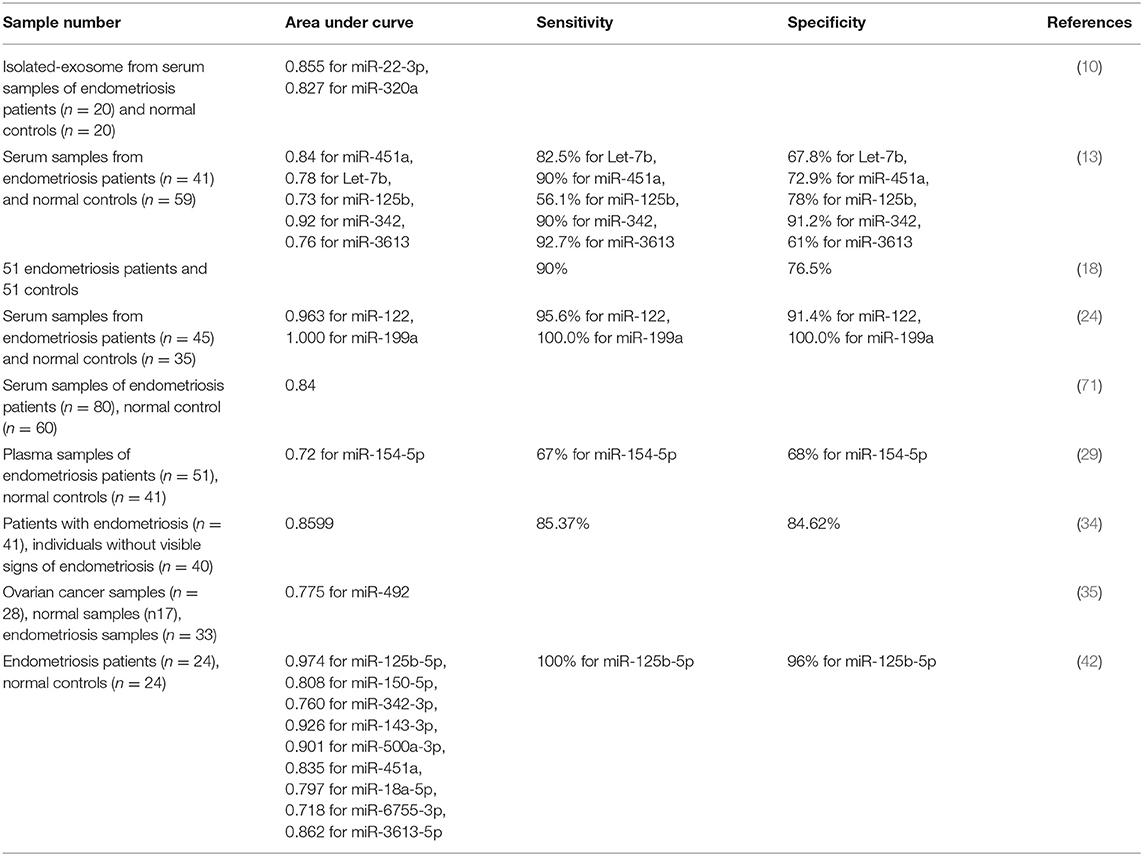
Diagnostic power of several miRNAs has been assessed in endometriosis. Maged et al. have shown that serum miR-122 and miR-199a had a sensitivity of 95.6 and 100.0% and a specificity of 91.4 and 100%, respectively, for diagnosis of disease status in women. Thus, these miRNAs are putative serum biomarkers for endometriosis (24). Moustafa et al. have shown up-regulation of miR-125b-5p, miR-150-5p, miR-342-3p, and miR-451a, while down-regulation of miR-3613-5p and let-7b in serum samples of patients with endometriosis compared with controls. The area under curve (AUC) values in receiver operating characteristic (ROC) curves ranged from 0.68 to 0.92 for these miRNAs. Notably, a classifier combining these miRNAs provided an AUC of 0.94 as verified in the independent set of individuals not included in the training set. Importantly, neither phase of menstrual cycle nor use of hormonal medicines affected the expression levels in these miRNAs. Thus, authors concluded the potential of the miRNAs panel in detection of endometriosis in clinical setting (13). Table 3 summarizes the results of studies which reported diagnostic value of miRNAs in endometriosis.
Few studies have reported association between single nucleotide polymorphisms (SNPs) within miRNA coding genes and risk of endometriosis. For instance, Sepahi et al. have genotyped the rs4636297 of miR-126 in 157 endometriosis patients and 252 healthy subjects. G allele of this SNP has been shown to protect against endometriosis. Moreover, significant association has also been detected between the A allele and severity of endometriosis (72). Zhang et al. have shown association between the CT/CC genotypes of miR-146b rs1536309 and the risk of pain symptom of endometriosis. Moreover, they detected lower levels of the miR-146b and higher pro-inflammatory functions in macrophages from CT/CC genotype carriers (12). Nimi-Hoveidi et al. have genotyped miR-143 rs41291957 and rs4705342 SNPs in infertile women with endometriosis and matched healthy subjects. They reported association between the C allele of rs4705342 and increased risk of endometriosis. In addition, the A allele of rs41291957 polymorphism was associated with susceptibility to endometriosis (73). Table 4 shows the results of studies which assessed association between miRNA SNPs and endometriosis.
LncRNAs and Endometriosis
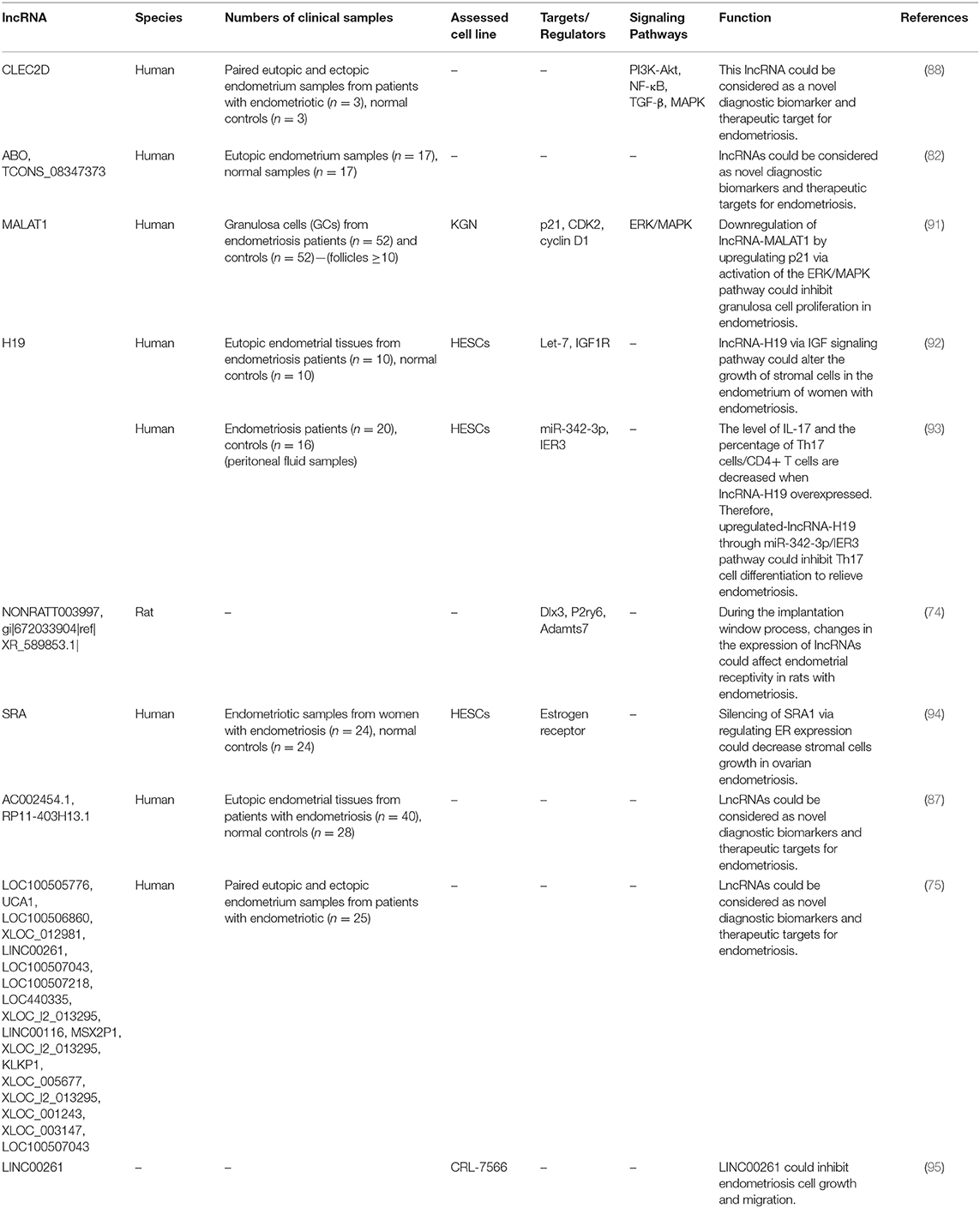
Expression levels of lncRNAs have been assessed in different samples obtained from patients with endometriosis or animal models of endometriosis. Cai et al. have assessed expression profiles of these transcripts in the uterus of rats with endometriosis and reported differential expression of a number of lncRNAs between endometriosis group and controls. They concluded that differentially expressed genes influence endometrial receptivity in rats with endometriosis during the implantation window which results in implantation failure (74). Using a high throughput method, Sun et al. have reported dysregulation of 948 lncRNA and 4,088 mRNA transcripts in ectopic endometrial tissue compared with paired eutopic endometrial tissue. These lncRNAs were mostly enriched in biological pathways associated with endometriosis, thus were thought to regulate expression of associated protein coding genes in cis- and/or trans (75). Huang et al. have assessed expression of the lncRNA UCA1 in ectopic and eutopic endometrium tissues of ovarian endometriosis patients and controls. They reported over-expression of this lncRNA in ectopic endometrium tissues compared with paired eutopic endometrium tissues in the majority of patients. They also demonstrated higher serum levels of this lncRNA after treatment. Notably, serum levels of UCA1 on the day of discharge were remarkably lower in patients with recurrence compared with patients without recurrence. Based on these results, authors concluded that UCA1 participates in the pathogenesis of ovarian endometriosis and may be a putative diagnostic and prognostic marker for this condition (76). Tables 5, 6 show up- and down-regulated lncRNAs in the endometriotic samples, respectively.
Among down-regulated lncRNAs is H19 whose role in the pathogenesis of endometriosis has been shown in Figure 1.
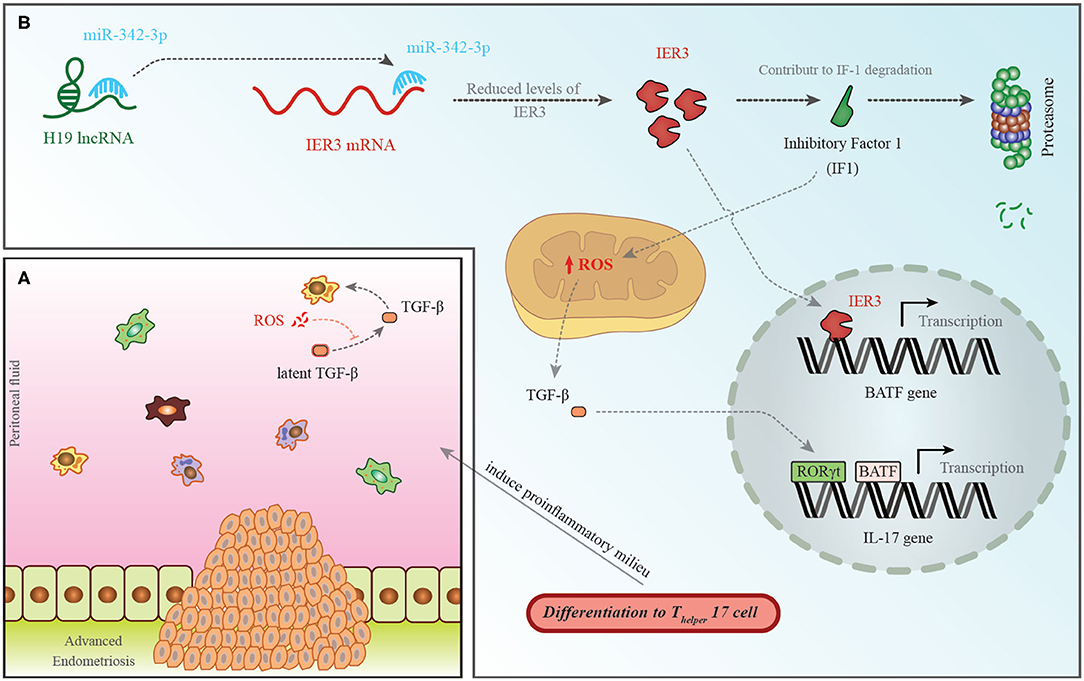
Figure 1. (A) A pro-endometriotic microenvironment produced by an existing endometriotic lesion provides the appropriate micro-environment for the progression of this disorder. After the buildup of cells by a previously established lesion, these elements show distinctive features that destroy immune surveillance (96). (B) H19 levels have been shown to be decreased in the PBMCs of patients with endometriosis. This is accompanied with an increase in miR-342-3p levels. This miRNA binds with the 3′ UTR of IER3, thus inhibiting its expression (93). IER3 participates in proteasomal degradation of IF-1. Decrease in the levels of IF-1 leads to increase in reactive oxygen species (ROS) levels (97). ROS increases active extracellular TGF-β levels. This cytokine influences RORγt, thus activating transcription of IL-17 and leading to differentiation of TH17 cells (98).
The expression pattern of MALAT1 has been assessed in a number of studies among them is the study by Liang et al. that reported down-regulation of this lncRNA in the endometriosis (48). Figure 2 depicts the molecular mechanism of involvement of MALAT1 in this disorder.
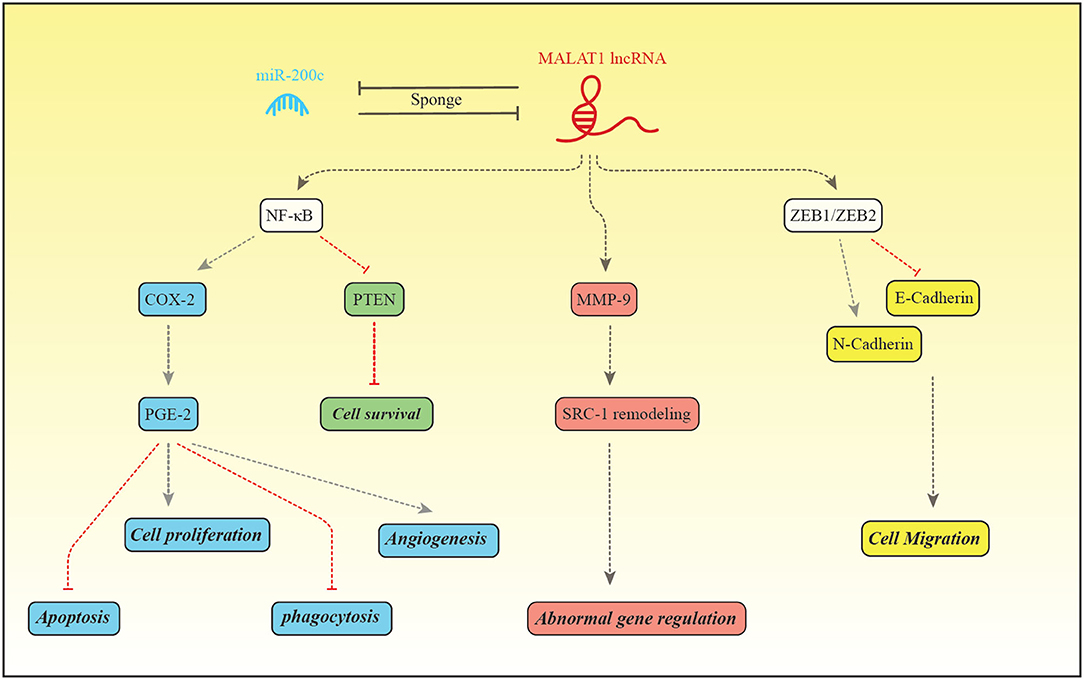
Figure 2. MALAT1 and miR-200c regulate expression of each other through the sponging mechanism. Liang et al. have reported down-regulation of MALAT1 and up-regulation of miR-200c in patients with endometriosis (48). MALAT1 increase expression of NF-κB which in turn binds with PGE-2 to enhance its expression (99). PGE-2 activates BCL2/BAX through interaction with EP2/EP4 receptor and suppresses intrinsic apoptotic pathway (100). PGE2 also activates cell proliferation through EP2/EP3 (101). In addition, PGE-2 suppresses MMP2, CD36 and annexin A2 in macrophages, thus inhibits phagocytic activity of macrophages. These effects facilitate implantation and growth of endometrial tissue in the peritoneum (101). PGE-2 influences angiogenic activity and cell cycle progression through increasing expression of VEGF and inhibiting PTEN, respectively (99). MALAT1 can enhance MMP9 levels. MMP9 increases production of the truncated isoform of Steroid receptor coactivator-1. MALAT1 also increases transcription of ZEB1/ZEB2, therefore induces mesenchymal cell phenotype which is accompanied by enhancement of cell migration (48).
Interaction Between miRNAs and lncRNAs in the Pathogenesis of Endometriosis
Based on the significant roles of lncRNAs and miRNAs in the pathogenesis of endometriosis and the presence of functional interactions between these two sets of transcripts, it is expected that lncRNA/miRNA pairs could regulate certain aspects of endometriosis. LncRNAs can act as a competing endogenous RNA (ceRNA) for miRNAs to affect their bioavailability of these transcripts. Assessments in the endometrial tissues have led to identification of a number of miRNAs that are inhibited by the lncRNA H19. For instance, Ghazal et al. have shown that H19 serves as a molecular sponge to decrease the availability of let-7. This interaction leads to over-expression of the downstream target of let-7, IGF1R, thus increasing the proliferation of endometrial stroma cells. They also demonstrated down-regulation of H19 in the eutopic endometrium of patients with endometriosis and speculated that the subsequent decrease in the IGF1R activity might diminish endometrial stromal cell proliferation and negatively influence the endometrial receptivity for pregnancy (92). In addition, Xu et al. have demonstrated the role of the estrogen-modulated H19/ACTA2/miR-216a-5p axis in the regulation of invasion and migration of eutopic endometrial stromal cells in subjects with endometriosis (78). Liu et al. have reported the significance of H19/miR-342-3p/IER3 axis in suppression of Th17 cell differentiation and decreasing the risk of endometriosis (93). A recent high throughput study of RNA profile of the ectopic and eutopic endometrium of patients has led to construction of the ceRNA network. Assessment of the RNA interaction network in endometriosis has resulted to identification of the role of miRNAs and lncRNAs that are associated with cyclin-dependent kinase 1 (CDK1) and proliferating cell nuclear antigen (PCNA). These genes regulate the growth and apoptosis of endometrial stromal cells, thus are involved in the pathophysiology of endometriosis. Taken together, the RNA interactive network has critical role in this disorder (102).
Discussion
Several studies have assessed expression profile of lncRNAs and miRNAs in tissues/blood samples obtained from patients with endometriosis. Association between genomic variants of miRNAs and endometriosis has also been another research avenue. However, the latter field has been less explored for lncRNAs. Considering the presence of myriads of SNPs within lncRNA coding genes that modulate their expression and regulatory functions on their targets, assessment of their association with the risk of endometriosis is a necessary step for identification of the role of these transcripts. Non-coding RNAs have fundamental roles in the development of endometriosis. Their role in this process has been highlighted not only by the studies which reported their aberrant expression in patients' samples, but also by the investigations which showed modulation of their expression by therapeutic agents. For instance, Quercetin (3,3′,4′,5,7-pentahydroxyflavone) as a phytochemical agent with antioxidant, anti-inflammatory and antiangiogenic characteristics has been shown to suppress the proliferation and induce cell cycle arrest in VK2/E6E7 and End1/E6E7 cells. Moreover, it has exerted antiproliferative and anti-inflammatory impacts on endometriosis autoimplanted mouse models. This effect has been accompanied by induction of miR-503-5p, miR-1283, miR-3714 and miR-6867-5p in both cell lines and stimulation of miR-503-5p and miR-546 expression in the animal model (103). Saponin extract, as another natural therapeutic agent has been shown to reduce expression of miR-21-5p in the human endometrial stromal cells from patients with endometriosis. Suppression of this miRNA could induce apoptosis in these cells. These results imply that he therapeutic effect of saponin is exerted through modulation of specific miRNAs (69).
Expressions of miRNAs have been assessed in different samples from patients with endometriosis such as endometrium, peripheral blood and peritoneal fluid. There are some cases of inconsistency between these studies. For instance, expression of miR-451a has been shown to be up-regulated in serum (13), exosomes (20) and endometriosis lesions of patients with endometriosis (23) as well as samples obtained from mouse models of endometriosis (22). However, another study has reported downregulation of miR-451 in the eutopic endometrial tissues of patients with endometriosis compared with control tissues (53). The lncRNA UCA1 has been reported to be up-regulated in ectopic endometrium tissues compared with paired eutopic endometrium tissues in the majority of patients using qRT-PCR (76). On the other hand, a microarray analysis showed down-regulated of this lncRNA in ovarian ectopic endometrial tissue compared with paired eutopic endometrial tissue (75). Similar discrepancy has been observed for MALAT1. While it has been upregulated in endometrial tissues from patients with endometriosis compared with normal controls (78), it was downregulated in granulosa cells from endometriosis patients compared with controls (91). The heterogeneity of samples and the method of expression analysis can partly explain the inconsistency of these results.
Mechanistically, lncRNAs can sponge miRNAs, regulate expression of inflammatory factors, alter cell proliferation, migration and apoptosis of endometrial cells. They might also affect implantation process (104, 105). Several transcription factors and signaling pathways have been regulated by lncRNAs in the endometrial tissues. Examples are HOX genes, N-cadherin, snail, slug, TCF8/ZEB1, matrix metalloproteinase, apoptosis related genes such as caspases and autophagy-related genes such as Beclin1.
The advent of next generation sequencing has enhanced the pace of identification of dysregulated non-coding RNAs in all human diseases including endometriosis. This technique has been applied by Khalaj et al. to identify signature of these transcripts in extracellular vesicles (EVs) obtained from endometriosis patient tissues and plasma samples compared with controls. Authors have demonstrated the presence of distinctive signatures of miRNAs and lncRNAs indicating their participation in the pathogenesis of endometriosis. Dysregulated transcripts were enriched in the pathways related to immune and metabolic functions. Their results indicated that endometriosis-associated EVs transport distinctive cargo and influence the disease course by modulation of inflammation, angiogenesis and proliferation (23). Moreover, exosomal miRNAs isolated from peritoneal macrophages have been shown to increase proliferation, migration, and invasion of ectopic endometrial stromal cells (10). Thus, these transcripts have fundamental roles in the pathogenesis of endometriosis. Taken together, these studies have opened a new research era for identification of the pathophysiology of endometriosis. Another technical development which has facilitated identification of this process has been the cell sorting technique. This technique has paved the way for cell-type-specific analysis of ectopic tissues to recognize the interactions between different cell types during the course of disease (30).
Considering the unavailability of affected tissues in the endometriosis except through invasive methods, identification of biomarkers in the serum of patients has a practical significance. Recent studies have demonstrated appropriate diagnostic power and sensitivity and specificity values for several miRNAs in this regard. Several miRNAs panels are expected to be applied in the clinical settings with high diagnostic power values. In spite of the presence of these supporting results, there is no consensus on a panel for the diagnosis of endometriosis, since most of studies have been conducted in small samples sizes of patients and their results have not been verified in independent samples. Besides, based on the differences in the source of controls, the applied techniques and the biological sources, meta-analysis of the obtained data is complicated. The diagnostic power of lncRNAs in the endometriosis has been less studied. Thus, future studies are needed to assess this aspect as well.
Taken together, based on the results of human and animal investigation, both miRNAs and lncRNAs participate in the pathogenesis of endometriosis. A more comprehensive assessment of these transcripts using the high throughput methods and identification of the functional links between these two sets of transcripts would facilitate identification of the pathogenesis of endometriosis and recognition of possible therapeutic targets in this regard.
Author Contributions
HS performed the data collection. MT and SG-F wrote the draft and revised it. MT designed the hypothesis. All authors contributed to the article and approved the submitted version.
Funding
This study was financially supported by Shahid Beheshti University of Medical Sciences.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
2. Nisolle M, Donnez J. Peritoneal endometriosis, ovarian endometriosis, and adenomyotic nodules of the rectovaginal septum are three different entities. Fertil Steril. (1997) 68:585–96. doi: 10.1016/S0015-0282(97)00191-X
3. Parasar P, Ozcan P, Terry KL. Endometriosis: epidemiology, diagnosis and clinical management. Curr Obstet Gynecol Rep. (2017) 6:34–41. doi: 10.1007/s13669-017-0187-1
4. Hansen KA, Eyster KM. Genetics and genomics of endometriosis. Clin Obstet Gynecol. (2010) 53:403–12. doi: 10.1097/GRF.0b013e3181db7ca1
5. Panir K, Schjenken JE, Robertson SA, Hull ML. Non-coding RNAs in endometriosis: a narrative review. Human Reprod Update. (2018) 24:497–515. doi: 10.1093/humupd/dmy014
6. Fernandes JCR, Acuña SM, Aoki JI, Floeter-Winter LM, Muxel SM. Long non-coding RNAs in the regulation of gene expression: physiology and disease. Noncoding RNA. (2019) 5:17. doi: 10.3390/ncrna5010017
7. Zhao Y, Li H, Fang S, Kang Y, Wu W, Hao Y, et al. NONCODE 2016: an informative and valuable data source of long non-coding RNAs. Nucleic Acids Res. (2016) 44(D1):D203–8. doi: 10.1093/nar/gkv1252
8. Catalanotto C, Cogoni C, Zardo G. MicroRNA in control of gene expression: an overview of nuclear functions. Int J Mol Sci. (2016) 17:1712. doi: 10.3390/ijms17101712
9. Zhang L, Li H, Yuan M, Li D, Sun C, Wang G. Serum exosomal microRNAs as potential circulating biomarkers for endometriosis. Dis Mark. (2020) 2020:2456340. doi: 10.1155/2020/2456340
10. Zhang L, Li H, Yuan M, Li D, Wang G. Exosomal miR-22-3p derived from peritoneal macrophages enhances proliferation, migration, and invasion of ectopic endometrial stromal cells through regulation of the SIRT1/NF-κB signaling pathway. Eur Rev Med Pharmacol Sci. (2020) 24:571–80. doi: 10.26355/eurrev_202001_20033
11. Zhou C-F, Liu M-J, Wang W, Wu S, Huang Y-X, Chen G-B, et al. miR-205-5p inhibits human endometriosis progression by targeting ANGPT2 in endometrial stromal cells. Stem Cell Res Ther. (2019) 10:287. doi: 10.1186/s13287-019-1388-5
12. Zhang Z, Li H, Zhao Z, Gao B, Meng L, Feng X. miR-146b level and variants is associated with endometriosis related macrophages phenotype and plays a pivotal role in the endometriotic pain symptom. Taiw J Obstetr Gynecol. (2019) 58:401–8. doi: 10.1016/j.tjog.2018.12.003
13. Moustafa S, Burn M, Mamillapalli R, Nematian S, Flores V, Taylor HS. Accurate diagnosis of endometriosis using serum microRNAs. Am J Obstetr Gynecol. (2020). doi: 10.1016/j.ajog.2020.02.050
14. Zolbin MM, Mamillapalli R, Nematian SE, Goetz L, Taylor HS. Adipocyte alterations in endometriosis: reduced numbers of stem cells and microRNA induced alterations in adipocyte metabolic gene expression. Reprod Biol Endocrinol. (2019) 17:36. doi: 10.1186/s12958-019-0480-0
15. Zhang H, Li G, Sheng X, Zhang S. Upregulation of miR-33b promotes endometriosis via inhibition of Wnt/β-catenin signaling and ZEB1 expression. Mol Med Rep. (2019) 19:2144–52. doi: 10.3892/mmr.2019.9870
16. Yang P, Wu Z, Ma C, Pan N, Wang Y, Yan L. Endometrial miR-543 is downregulated during the implantation window in women with endometriosis-related infertility. Reprod Sci. (2019) 26:900–8. doi: 10.1177/1933719118799199
17. Dai Y, Lin X, Xu W, Lin X, Huang Q, Shi L, et al. MiR-210-3p protects endometriotic cells from oxidative stress-induced cell cycle arrest by targeting BARD1. Cell Death Dis. (2019) 10:1–15. doi: 10.1038/s41419-019-1395-6
18. Nabiel Y, ELshahawy H, Mosbah A. Intrauterine bacterial colonization and endometrial microRNA-17-5p levels in association to endometriosis: a study in an egyptian population. Immunol Investig. (2019). doi: 10.1080/08820139.2019.1693592. [Epub ahead of print].
19. Mashayekhi P, Noruzinia M, Zeinali S, Khodaverdi S. Endometriotic mesenchymal stem cells epigenetic pathogenesis: deregulation of miR-200b, miR-145, and let7b in a functional imbalanced epigenetic disease. Cell J. (2019) 21:179. doi: 10.22074/cellj.2019.5903
20. Chen Y, Wang K, Xu Y, Guo P, Hong B, Cao Y, et al. Alteration of myeloid-derived suppressor cells, chronic inflammatory cytokines, and exosomal miRNA contribute to the peritoneal immune disorder of patients with endometriosis. Reprod Sci. (2019) 26:1130–8. doi: 10.1177/1933719118808923
21. Cosar E, Mamillapalli R, Moridi I, Duleba A, Taylor HS. Serum microRNA biomarkers regulated by simvastatin in a primate model of endometriosis. Reprod Sci. (2019) 26:1343–50. doi: 10.1177/1933719118765971
22. Li M, Zhou Y, Taylor HS. miR-451a inhibition reduces established endometriosis lesions in mice. Reprod Sci. (2019) 26:1506–11. doi: 10.1177/1933719119862050
23. Khalaj K, Miller JE, Lingegowda H, Fazleabas AT, Young SL, Lessey BA, et al. Extracellular vesicles from endometriosis patients are characterized by a unique miRNA-lncRNA signature. JCI Insight. (2019) 4:e128846. doi: 10.1172/jci.insight.128846
24. Maged AM, Deeb WS, El Amir A, Zaki SS, El Sawah H, Al Mohamady M, et al. Diagnostic accuracy of serum miR-122 and miR-199a in women with endometriosis. Int J Gynecol Obstetr. (2018) 141:14–9. doi: 10.1002/ijgo.12392
25. Bashti O, Noruzinia M, Garshasbi M, Abtahi M. miR-31 and miR-145 as potential non-invasive regulatory biomarkers in patients with endometriosis. Cell J. (2018) 20:1–131. doi: 10.22074/cellj.2018.4915
26. Cheng F, Lu L, Wang H, Cheng H, Zhang D. Expression and significance of miR-126 and miR-145 in infertility due to endometriosis. J Coll Phys Surg Pak. (2019) 29:585–7. doi: 10.29271/jcpsp.2019.06.585
27. Marí-Alexandre J, Barceló-Molina M, Belmonte-López E, García-Oms J, Estellés A, Braza-Boïls A, et al. Micro-RNA profile and proteins in peritoneal fluid from women with endometriosis: their relationship with sterility. Fertil Steril. (2018) 109:675–84. e2. doi: 10.1016/j.fertnstert.2017.11.036
28. Pei T, Liu C, Liu T, Xiao L, Luo B, Tan J, et al. miR-194-3p represses the progesterone receptor and decidualization in eutopic endometrium from women with endometriosis. Endocrinology. (2018) 159:2554–62. doi: 10.1210/en.2018-00374
29. Pateisky P, Pils D, Szabo L, Kuessel L, Husslein H, Schmitz A, et al. hsa-miRNA-154-5p expression in plasma of endometriosis patients is a potential diagnostic marker for the disease. Reprod Biomed Online. (2018) 37:449–66. doi: 10.1016/j.rbmo.2018.05.007
30. Rekker K, Tasa T, Saare M, Samuel K, Kadastik Ü, Karro H, et al. Differentially-expressed miRNAs in ectopic stromal cells contribute to endometriosis development: the plausible role of miR-139-5p and miR-375. Int J Mol Sci. (2018) 19:3789. doi: 10.3390/ijms19123789
31. Zhao L, Gu C, Ye M, Zhang Z, Fan W, Meng Y. Integration analysis of microRNA and mRNA paired expression profiling identifies deregulated microRNA-transcription factor-gene regulatory networks in ovarian endometriosis. Reprod Biol Endocrinol. (2018) 16:4. doi: 10.1186/s12958-017-0319-5
32. Shu X-M. Assessment of the expression of mir-29c, mir-200a and mir-145 in endometrial tissue and the downstream molecules in infertile patients with endometriosis. J Hain Med Univ. (2017) 23:97–100.
33. Joshi NR, Miyadahira EH, Afshar Y, Jeong J-W, Young SL, Lessey BA, et al. Progesterone resistance in endometriosis is modulated by the altered expression of microRNA-29c and FKBP4. J Clin Endocrinol Metab. (2017) 102:141–9. doi: 10.1210/jc.2016-2076
34. Nothnick WB, Falcone T, Joshi N, Fazleabas AT, Graham A. Serum miR-451a levels are significantly elevated in women with endometriosis and recapitulated in baboons (Papio anubis) with experimentally-induced disease. Reprod Sci. (2017) 24:1195–202. doi: 10.1177/1933719116681519
35. Braicu O-L, Budisan L, Buiga R, Jurj A, Achimas-Cadariu P, Pop LA, et al. miRNA expression profiling in formalin-fixed paraffin-embedded endometriosis and ovarian cancer samples. OncoTargets Ther. (2017) 10:4225. doi: 10.2147/OTT.S137107
36. Kim MK, Lee SK, Park JH, Lee JH, Yun BH, Park JH, et al. Ginsenoside Rg3 decreases fibrotic and invasive nature of endometriosis by modulating miRNA-27b: in vitro and in vivo studies. Sci Rep. (2017) 7:1–14. doi: 10.1038/s41598-017-17956-0
37. Wright KR, Mitchell B, Santanam N. Redox regulation of microRNAs in endometriosis-associated pain. Redox Biol. (2017) 12:956–66. doi: 10.1016/j.redox.2017.04.037
38. Xu X, Li Z, Liu J, Yu S, Wei Z. MicroRNA expression profiling in endometriosis-associated infertility and its relationship with endometrial receptivity evaluated by ultrasound. J X Ray Sci Technol. (2017) 25:523–32. doi: 10.3233/XST-17286
39. Yang R, Teng H, Xu X, Liu S, Wang Y, Guo F, et al. Microarray analysis of microRNA deregulation and angiogenesis-related proteins in endometriosis. Genet Mol Res. (2016) 15:1–8. doi: 10.4238/gmr.15027826
40. Xu T, Zhao S, Dong M, Yu X. Hypoxia responsive miR-210 promotes cell survival and autophagy of endometriotic cells in hypoxia. Eur Rev Med Pharmacol Sci. (2016) 20:399–406.
41. Falih ES, Aubaid SH, Yousif WaT. The role of microRNA 122, MCP-1 and TGF-β1 as diagnostic biomarkers for endometriosis. Indian J Public Health Res Dev. (2019) 10:2652–6. doi: 10.5958/0976-5506.2019.03267.4
42. Cosar E, Mamillapalli R, Ersoy GS, Cho S, Seifer B, Taylor HS. Serum microRNAs as diagnostic markers of endometriosis: a comprehensive array-based analysis. Fertil Steril. (2016) 106:402–9. doi: 10.1016/j.fertnstert.2016.04.013
43. Eggers JC, Martino V, Reinbold R, Schäfer SD, Kiesel L, Starzinski-Powitz A, et al. microRNA miR-200b affects proliferation, invasiveness and stemness of endometriotic cells by targeting ZEB1, ZEB2 and KLF4. Reprod Biomed Online. (2016) 32:434–45. doi: 10.1016/j.rbmo.2015.12.013
44. Liu X, Bai X, Teng Y, Song L, Lu N, Yang R. miRNA-15a-5p regulates VEGFA in endometrial mesenchymal stem cells and contributes to the pathogenesis of endometriosis. Eur Rev Med Pharmacol Sci. (2016) 20:3319–26.
45. Hirakawa T, Nasu K, Abe W, Aoyagi Y, Okamoto M, Kai K, et al. miR-503, a microRNA epigenetically repressed in endometriosis, induces apoptosis and cell-cycle arrest and inhibits cell proliferation, angiogenesis, and contractility of human ovarian endometriotic stromal cells. Human Reprod. (2016) 31:2587–97. doi: 10.1093/humrep/dew217
46. Ma Y, Huang YX, Chen YY. miRNA-34a-5p downregulation of VEGFA in endometrial stem cells contributes to the pathogenesis of endometriosis. Mol Med Rep. (2017) 16:8259–64. doi: 10.3892/mmr.2017.7677
47. Seifer BJ, Su D, Taylor HS. Circulating miRNAs in murine experimental endometriosis: decreased abundance of let-7a. Reprod Sci. (2017) 24:376–81. doi: 10.1177/1933719116667228
48. Liang Z, Chen Y, Zhao Y, Xu C, Zhang A, Zhang Q, et al. miR-200c suppresses endometriosis by targeting MALAT1 in vitro and in vivo. Stem Cell Res Ther. (2017) 8:251. doi: 10.1186/s13287-017-0706-z
49. Yang W, Hong L, Xu X, Wang Q, Huang J, Jiang L. Regulation of miR-33b on endometriosis and expression of related factors. Eur Rev Med Pharmacol Sci. (2017) 21:2027−33.
50. Chen X, Jiang Y, Pan D. miR-30c may serve a role in endometriosis by targeting plasminogen activator inhibitor-1. Exp Ther Med. (2017) 14:4846–52. doi: 10.3892/etm.2017.5145
51. Yang W, Hong L, Xu X, Wang Q, Huang J, Jiang L. MiR-424-5p regulates proliferation and apoptosis by targeting FGFR1 in endometriosis cells. Int J Clin Exp Med. (2017) 10:666–74.
52. Zhang A, Wang G, Jia L, Su T, Zhang L. Exosome-mediated microRNA-138 and vascular endothelial growth factor in endometriosis through inflammation and apoptosis via the nuclear factor-κB signaling pathway. Int J Mol Med. (2019) 43:358–70. doi: 10.3892/ijmm.2018.3980
53. Gao S, Liu S, Gao Z-M, Deng P, Wang D-B. Reduced microRNA-451 expression in eutopic endometrium contributes to the pathogenesis of endometriosis. World J Clin Cases. (2019) 7:2155. doi: 10.12998/wjcc.v7.i16.2155
54. Zhang M, Zhang Y, Li L, Ma L, Zhou C. Dysregulation of miR-202-3p affects migration and invasion of endometrial stromal cells in endometriosis via targeting ROCK1. Reprod Sci. (2020) 27:731–42. doi: 10.1007/s43032-019-00079-4
55. Liu Y, Lu C, Fan L, Wang J, Li T, Liu Z, et al. MiR-199a-5p targets ZEB1 to inhibit the epithelial-mesenchymal transition of ovarian ectopic endometrial stromal cells via PI3K/Akt/mTOR signal pathway in vitro and in vivo. Reprod Sci. (2020) 27:110–8. doi: 10.1007/s43032-019-00016-5
56. Chen L-J, Hu B, Han Z-Q, Ni J, Zhou Y-M, Chen X-X, et al. MicroRNA-20a mediates the cytotoxicity of natural killer cells in endometriosis via ERG/HLX/STAT4/perforin axis. Preprint. (2020). doi: 10.21203/rs.2.17459/v1
57. Zhang Y, Yan J, Pan X. miR-141-3p affects apoptosis and migration of endometrial stromal cells by targeting KLF-12. Pflügers Archiv Eur J Physiol. (2019) 471:1055–63. doi: 10.1007/s00424-019-02283-2
58. Petracco R, Dias ACdO, Taylor HS, Petracco Á, Badalotti M, Michelon JdR, et al. Evaluation of miR-135a/b expression in endometriosis lesions. Biomed Rep. (2019) 11:181–7. doi: 10.3892/br.2019.1237
59. Li X, Zhang W, Fu J, Xu Y, Gu R, Qu R, et al. MicroRNA-451 is downregulated in the follicular fluid of women with endometriosis and influences mouse and human embryonic potential. Reprod Biol Endocrinol. (2019) 17:96. doi: 10.1186/s12958-019-0538-z
60. Ma L, Li Z, Li W, Ai J, Chen X. MicroRNA-142-3p suppresses endometriosis by regulating KLF9-mediated autophagy in vitro and in vivo. RNA Biol. (2019) 16:1733–48. doi: 10.1080/15476286.2019.1657352
61. Kästingschäfer CS, Schäfer SD, Kiesel L, Götte M. miR-142-3p is a novel regulator of cell viability and proinflammatory signalling in endometrial stroma cells. Reprod Biomed Online. (2015) 30:553–6. doi: 10.1016/j.rbmo.2015.01.002
62. Zhu H, Cao XX, Liu J, Hua H. MicroRNA-488 inhibits endometrial glandular epithelial cell proliferation, migration, and invasion in endometriosis mice via Wnt by inhibiting FZD7. J Cell Mol Med. (2019) 23:2419–30. doi: 10.1111/jcmm.14078
63. Hu Z, Mamillapalli R, Taylor HS. Increased circulating miR-370-3p regulates steroidogenic factor 1 in endometriosis. Am J Physiol Endocrinol Metab. (2019) 316:E373–82. doi: 10.1152/ajpendo.00244.2018
64. Meng X, Liu J, Wang H, Chen P, Wang D. MicroRNA-126-5p downregulates BCAR3 expression to promote cell migration and invasion in endometriosis. Mol Cell Endocrinol. (2019) 494:110486. doi: 10.1016/j.mce.2019.110486
65. Wu D, Lu P, Mi X, Miao J. Exosomal miR-214 from endometrial stromal cells inhibits endometriosis fibrosis. MHR Basic Sci Reprod Med. (2018) 24:357–65. doi: 10.1093/molehr/gay019
66. He SZ, Li J, Bao HC, Wang MM, Wang XR, Huang X, et al. G protein-coupled estrogen receptor/miR-148a/human leukocyte antigen-G signaling pathway mediates cell apoptosis of ovarian endometriosis. Mol Med Rep. (2018) 18:1141–8. doi: 10.3892/mmr.2018.9039
67. Hsu CY, Hsieh TH, Er TK, Chen HS, Tsai CC, Tsai EM. MiR-381 regulates cell motility, growth and colony formation through PIK3CA in endometriosis-associated clear cell and endometrioid ovarian cancer. Oncol Rep. (2018) 40:3734–42. doi: 10.3892/or.2018.6779
68. Sahin C, Mamillapalli R, Yi KW, Taylor HS. micro RNA Let-7b: a Novel treatment for endometriosis. J Cell Mol Med. (2018) 22:5346–53. doi: 10.1111/jcmm.13807
69. Park JH, Lee SK, Kim MK, Lee JH, Yun BH, Park JH, et al. Saponin extracts induced apoptosis of endometrial cells from women with endometriosis through modulation of miR-21-5p. Reprod Sci. (2018) 25:292–301. doi: 10.1177/1933719117711263
70. Liu Y, Chen J, Zhu X, Tang L, Luo X, Shi Y. Role of miR-449b-3p in endometriosis via effects on endometrial stromal cell proliferation and angiogenesis. Mol Med Rep. (2018) 18:3359–65. doi: 10.3892/mmr.2018.9341
71. Wang F, Wang H, Jin D, Zhang Y. Serum miR-17, IL-4, and IL-6 levels for diagnosis of endometriosis. Medicine. (2018) 97:e10853. doi: 10.1097/MD.0000000000010853
72. Sepahi N, Kohan L, Jahromi AR, Daneshbod Y, Hoveidi EN. mir-126 rs4636297 and TGF β RI rs334348 functional gene variants are associated with susceptibility to endometriosis and its severity. Gynecol Endocrinol. (2017) 33:429–32. doi: 10.1080/09513590.2017.1290064
73. Nimi-Hoveidi E, Kohan L, Hashemi SS. Association of miR-143 rs41291957 and rs4705342 genetic variants with endometriosis risk in infertile women. KAUMS J. (2016) 20:441−6.
74. Cai H, Zhu X, Li Z, Zhu Y, Lang J. lncRNA/mRNA profiling of endometriosis rat uterine tissues during the implantation window. Int J f Mol Med. (2019) 44:2145–60. doi: 10.3892/ijmm.2019.4370
75. Sun P-r, Jia S-z, Lin H, Leng J-H, Lang J-H. Genome-wide profiling of long noncoding ribonucleic acid expression patterns in ovarian endometriosis by microarray. Fertil Steril. (2014) 101:1038-46. e7. doi: 10.1016/j.fertnstert.2013.12.035
76. Huang H, Zhu Z, Song Y. Downregulation of lncrna uca1 as a diagnostic and prognostic biomarker for ovarian endometriosis. Rev Assoc Méd Bras. (2019) 65:336–41. doi: 10.1590/1806-9282.65.3.336
77. Qiu J-J, Lin Y-Y, Tang X-Y, Ding Y, Yi X-F, Hua K-Q. Extracellular vesicle-mediated transfer of the lncRNA-TC0101441 promotes endometriosis migration/invasion. Exp Cell Res. (2020) 388:111815. doi: 10.1016/j.yexcr.2020.111815
78. Xu Z, Zhang L, Yu Q, Zhang Y, Yan L, Chen Z-J. The estrogen-regulated lncRNA H19/miR-216a-5p axis alters stromal cell invasion and migration via ACTA2 in endometriosis. Mol Hum Reprod. (2019) 25:550–61. doi: 10.1093/molehr/gaz040
79. Liu H, Zhang Z, Xiong W, Zhang L, Du Y, Liu Y, et al. Long non-coding RNA MALAT 1 mediates hypoxia-induced pro-survival autophagy of endometrial stromal cells in endometriosis. J Cell Mol Med. (2019) 23:439–52. doi: 10.1111/jcmm.13947
80. Zhang C, Wu W, Zhu H, Yu X, Zhang Y, Ye X, et al. Knockdown of long noncoding RNA CCDC144NL-AS1 attenuates migration and invasion phenotypes in endometrial stromal cells from endometriosis. Biol Reprod. (2019) 100:939–49. doi: 10.1093/biolre/ioy252
81. Zhu M, Chen L, Hu M, Shi Z, Liu Y. Effects of lncRNA BANCR on endometriosis through ERK/MAPK pathway. Eur Rev Med Pharmacol Sci. (2019) 23:6806–12. doi: 10.26355/eurrev_201908_18719
82. Cui D, Ma J, Liu Y, Lin K, Jiang X, Qu Y, et al. Analysis of long non-coding RNA expression profiles using RNA sequencing in ovarian endometriosis. Gene. (2018) 673:140–8. doi: 10.1016/j.gene.2018.06.046
83. Lin D, Huang Q, Wu R, Dai S, Huang Z, Ren L, et al. Long non-coding RNA AFAP1-AS1 promoting epithelial-mesenchymal transition of endometriosis is correlated with transcription factor ZEB1. Am J Reprod Immunol. (2019) 81:e13074. doi: 10.1111/aji.13074
84. Liu J, Wang Q, Zhang R, Zhang C, Lin J, Huang X. Identification of LINC01279 as a cell cycle-associated long non-coding RNA in endometriosis with GBA analysis. Mol Med Rep. (2018) 18:3850–8. doi: 10.3892/mmr.2018.9387
85. Zhang C, Wu W, Ye X, Ma R, Luo J, Zhu H, et al. Aberrant expression of CHL1 gene and long non-coding RNA CHL1-AS1, CHL1-AS2 in ovarian endometriosis. Eur J Obstetr Gynecol Reprod Biol. (2019) 236:177–82. doi: 10.1016/j.ejogrb.2019.03.020
86. Wang M, Hao C, Huang X, Bao H, Qu Q, Liu Z, et al. Aberrant expression of lncRNA (HOXA11-AS1) and homeobox A (HOXA9, HOXA10, HOXA11, and HOXA13) genes in infertile women with endometriosis. Reprod Sci. (2018) 25:654–61. doi: 10.1177/1933719117734320
87. Wang Y, Li Y, Yang Z, Liu K, Wang D. Genome-wide microarray analysis of long non-coding RNAs in eutopic secretory endometrium with endometriosis. Cell Physiol Biochem. (2015) 37:2231–45. doi: 10.1159/000438579
88. Liu S-P, Tian X, Cui H-Y, Zhang Q, Hua K-Q. The messenger RNA and long non-coding RNA expression profiles in ectopic and eutopic endometrium provide novel insights into endometriosis. Reprod Dev Med. (2019) 3:11. doi: 10.4103/2096-2924.255992
89. Qiu J, Hua K. Exosomal long noncoding RNA-NONHAT076754 faciliates endometriosis invasion and predicts endometriosis recurrence. J Minim Invasive Gynecol. (2019) 26:S12. doi: 10.1016/j.jmig.2019.09.044
90. Qiu J-J, Lin X-J, Zheng T-T, Tang X-Y, Zhang Y, Hua K-Q. The exosomal long noncoding RNA aHIF is upregulated in serum from patients with endometriosis and promotes angiogenesis in endometriosis. Reprod Sci. (2019) 26:1590–602. doi: 10.1177/1933719119831775
91. Li Y, Liu Y-D, Chen S-L, Chen X, Ye D-S, Zhou X-Y, et al. Down-regulation of long non-coding RNA MALAT1 inhibits granulosa cell proliferation in endometriosis by up-regulating P21 via activation of the ERK/MAPK pathway. MHR Basic Sci Reprod Med. (2019) 25:17–29. doi: 10.1093/molehr/gay045
92. Ghazal S, McKinnon B, Zhou J, Mueller M, Men Y, Yang L, et al. H19 lncRNA alters stromal cell growth via IGF signaling in the endometrium of women with endometriosis. EMBO Mol Med. (2015) 7:996–1003. doi: 10.15252/emmm.201505245
93. Liu Z, Liu L, Zhong Y, Cai M, Gao J, Tan C, et al. LncRNA H19 over-expression inhibited Th17 cell differentiation to relieve endometriosis through miR-342-3p/IER3 pathway. Cell Bioscience. (2019) 9:84. doi: 10.1186/s13578-019-0346-3
94. Lin K, Zhan H, Ma J, Xu K, Wu R, Zhou C, et al. Silencing of SRA1 regulates ER expression and attenuates the growth of stromal cells in ovarian endometriosis. Reprod Sci. (2017) 24:836–43. doi: 10.1177/1933719116670036
95. Sha L, Huang L, Luo X, Bao J, Gao L, Pan Q, et al. Long non-coding RNA LINC00261 inhibits cell growth and migration in endometriosis. J Obstetr Gynaecol Res. (2017) 43:1563–9. doi: 10.1111/jog.13427
96. Liang Y, Wu J, Wang W, Xie H, Yao S. Pro-endometriotic niche in endometriosis. Reprod Biomed Onl. (2019) 38:549–59. doi: 10.1016/j.rbmo.2018.12.025
97. García-Bermúdez J, Cuezva JM. The ATPase inhibitory factor 1 (IF1): A master regulator of energy metabolism and of cell survival. Biochim Biophys Acta Bioenerget. (2016) 1857:1167–82. doi: 10.1016/j.bbabio.2016.02.004
98. Zhang D, Jin W, Wu R, Li J, Park S-A, Tu E, et al. High glucose intake exacerbates autoimmunity through reactive-oxygen-species-mediated TGF-β cytokine activation. Immunity. (2019) 51:671–81. e5. doi: 10.1016/j.immuni.2019.08.001
99. Kaponis A, Iwabe T, Taniguchi F, Ito M, Deura I, Decavalas G, et al. The role of NF-kappaB in endometriosis. Front Biosci. (2012) 4:1213–34. doi: 10.2741/s327
100. Cho YJ, Lee SH, Park JW, Han M, Park MJ, Han SJ. Dysfunctional signaling underlying endometriosis: current state of knowledge. J Mol Endocrinol. (2018) 60:R97–113. doi: 10.1530/JME-17-0227
101. Hsiao K-Y, Wu M-H, Tsai S-J. Roles of Prostaglandin E 2 in Endometriosis. Endometriosis: Tokyo: Springer (2014). p. 125–46.
102. Zhang M, Li J, Duan S, Fang Z, Tian J, Yin H, et al. Comprehensive characterization of endometrial competing endogenous RNA network in infertile women of childbearing age. Aging. (2020) 12:4204–21. doi: 10.18632/aging.102874
103. Park S, Lim W, Bazer FW, Whang K-Y, Song G. Quercetin inhibits proliferation of endometriosis regulating cyclin D1 and its target microRNAs in vitro and in vivo. J Nutr Biochem. (2019) 63:87–100. doi: 10.1016/j.jnutbio.2018.09.024
104. Gao W-l, Liu M, Yang Y, Yang H, Liao Q, Bai Y, et al. The imprinted H19 gene regulates human placental trophoblast cell proliferation via encoding miR-675 that targets Nodal Modulator 1 (NOMO1). RNA Biol. (2012) 9:1002–10. doi: 10.4161/rna.20807
Keywords: miRNA, lncRNA, endometriosis, non-coding RNA, inflammation
Citation: Ghafouri-Fard S, Shoorei H and Taheri M (2020) Role of Non-coding RNAs in the Pathogenesis of Endometriosis. Front. Oncol. 10:1370. doi: 10.3389/fonc.2020.01370
Received: 03 April 2020; Accepted: 29 June 2020;
Published: 04 August 2020.
Edited by:
Jawed Akhtar Siddiqui, University of Nebraska Medical Center, United StatesReviewed by:
Felice Petraglia, University of Florence, ItalyKai Fu, Central South University, China
Copyright © 2020 Ghafouri-Fard, Shoorei and Taheri. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Mohammad Taheri, bW9oYW1tYWRfODIzQHlhaG9vLmNvbQ==
 Soudeh Ghafouri-Fard1
Soudeh Ghafouri-Fard1 Hamed Shoorei
Hamed Shoorei Mohammad Taheri
Mohammad Taheri